This page intentionally left blank
A-Z of Abdominal Radiology
A-Z of Abdominal
Radiology
Gabriel Conder, MRCP, FRCR
Consultant Radiologist,
Northwick Park and Central Middlesex Hospitals, North West London
Hospitals NHS Trust, London
John Rendle, FRCS (Eng), FRCR
Consultant Radiologist
Mayday University Hospital, Mayday Healthcare NHS Trust, Croydon
Sarah Kidd, MRCP, FRCR
Specialist Registrar in Radiology
St. Mary’s Hospital, Imperial College Healthcare NHS Trust, London
Rakesh R. Misra, BSc (Hons), FRCS (Eng.), FRCR
Consultant Radiologist
Wycombe Hospital, Buckinghamshire Hospital NHS Trust, High Wycombe
Series Editor
R. R. Misra
CAMBRIDGE UNIVERSITY PRESS
Cambridge, New York, Melbourne, Madrid, Cape Town, Singapore, São Paulo
Cambridge University Press
The Edinburgh Building, Cambridge CB2 8RU, UK
Published in the United States of America by Cambridge University Press, New York
© G. Conder, J. Rendle, S. Kidd and R. Misra 2009
This publication is in copyright. Subject to statutory exception and to the
provision of relevant collective licensing agreements, no reproduction of any part
may take place without the written permission of Cambridge University Press.
First published in print format
2009
ISBN-13
978-0-511-48087-4
eBook (NetLibrary)
ISBN-13
978-0-521-70014-6
paperback
Cambridge University Press has no responsibility for the persistence or accuracy
of urls for external or third-party internet websites referred to in this publication,
and does not guarantee that any content on such websites is, or will remain,
accurate or appropriate.
Every effort has been made in preparing this publication to provide accurate and
up-to-date information which is in accord with accepted standards and practice at
the
time of publication. Although case histories are drawn from actual cases, every
effort has
been made to disguise the identities of the individuals involved. Nevertheless, the
authors,
editors and publishers can make no warranties that the information contained
herein is
totally free from error, not least because clinical standards are constantly changing
through
research and regulation. The authors, editors and publishers therefore disclaim all
liability
for direct or consequential damages resulting from the use of material contained in
this
bli
i
R d
l
d i d
f l
i
i f
i
To my beautiful wife Rachel, and four inspirational
children - Rohan, Ela, Krishan and Maya. R. R. M.
To my mother and father - very different people, but
both inspirational. G. C.
CONTENTS
Acknowledgements
page ix
Preface
xi
List of abbreviations
xii
Abdominal trauma
2
Abscesses within the abdomen
14
Achalasia
20
Adnexal masses
22
Adrenal masses
28
Aortic aneurysm
38
Appendicitis
40
Ascites
46
Bezoar
50
Biliary gas
54
Biliary obstruction
56
Bladder calculi
64
Bowel obstruction
66
Calcifications
80
Chilaiditi’s sign/syndrome
86
Cholecystitis
88
Cirrhosis
92
Colitis
96
Colonic carcinoma
110
Colonic diverticulitis
116
Colonic polyps
120
Crohn’s disease
126
Dermoid tumour
134
Ectopic pregnancy
136
Endometrial carcinoma
138
vii
Familial polyposis coli
142
Fistulae
144
Foreign bodies
150
Free intra-abdominal gas
156
Gallstones
162
Hepatic masses
168
Herniae of the abdomen and pelvis
190
Intussusception
200
Lines and devices
208
Lymphadenopathy
220
Lymphoma: abdominal
224
Omental secondary deposits
232
Pancreatic carcinoma
234
Pancreatitis
240
Portal vein thrombosis
248
Portal venous gas
252
Portal venous hypertension
256
Pyelonephritis
262
Renal artery stenosis
270
Renal calcification
276
Renal developmental anomalies
282
Renal masses: benign
290
Renal masses: malignant
300
Renal tract calculi
308
Retroperitoneal fibrosis
314
Splenomegaly
318
Testicular neoplasms
322
Transitional cell carcinoma
324
Tuberculosis of the abdomen and pelvis
330
Uterine fibroids
340
Volvulus
346
viii
ACKNOWLEDGEMENTS
I would like to thank my consultant colleagues Carolyn Charlesworth,
Philip Cadman, Richard Hughes, Dinuke Warakaulle, Eric Woo and
Vipin Uthappa for their help in sourcing so many of the varied images
needed for this book. Your time and effort is greatly appreciated.
Thanks to my good friend Miles Berry, who helped to conceive the
idea for this book in a Parisian bistro in 2001.
R. R. M.
The authors would also like to thank Professor W. Gedroyc and Drs Jon
Ord and David Burling for contributing several important images.
ix
PREFACE
The A-to-Z of Abdominal Radiology is a valuable addition to the A-to-Z of
Radiology series, continuing the theme of concise introductions to specific
fields of radiology. The text is concisely laid out, following a similar format
to the other title in this series, using a bullet point format to allow rapid
assimilation of relevant facts. The text is richly illustrated with images from
modern scanners.
The book will be a useful aid to medical students, radiographers,
surgical trainees, physicians and emergency doctors who wish to gain a
greater understanding of abdominal and pelvic imaging and how it can
improve their clinical practice. Radiology trainees will also find this a
helpful aide-mémoire to consolidate their knowledge.
G. C.
J. R.
S. K.
R. R. M.
xi
ABBREVIATIONS
99mTc
technetium-99m
AAA
Abdominal aortic aneurysm
ACE
Angiotensin-converting-enzyme
AIDS
Acquired immunodeficiency syndrome
AP
Antero-posterior
ARDS
Adult respiratory distress syndrome
ATLS
Advanced trauma life support
AXR
Abdominal X-ray
CA19-9
Carbohydrate antigen 19-9
CECT
Contrast-enhanced computed tomography
CNS
Central nervous system
CT
Computed tomography
CXR
Chest X-ray
ERCP
Endoscopic retrograde cholangiopancreatography
FDG
Fluorodeoxyglucose
FIGO
Federation international de gynaecologie et obstetrique
FNH
Focal nodular hyperplasia
GI
Gastrointestinal
Gy
Gray
HCC
Hepatocellular carcinoma
HD
Hodgkin’s disease
HU
Hounsfield unit
IUCD
Intrauterine contraceptive device
IV
Intravenous
IVC
Inferior vena cava
IVU
Intravenous urogram
KUB
Kidney-ureter-bladder
LBO
Large-bowel obstruction
LIF
Left iliac fossa
LUQ
Left upper quadrant
MALT
Mucosa-associated lymphoid tissue
MIBG
Meta-iodobenzylguanidine
MR
Magnetic resonance
MRCP
Magnetic resonance cholangiopancreatography
MRI
Magnetic resonance imaging
NECT
Non-enhanced CT
xii
NHL
Non-Hodgkin lymphoma
NM
Nuclear medicine
PET
Positron emission tomography
PID
Pelvic inflammatory disease
PTC
Percutaneous trans-hepatic cholangiogram
RAS
Renal artery stenosis
RIF
Right iliac fossa
RLQ
Right lower quadrant
RPF
Retroperitoneal fibrosis
RTA
Road traffic accident
RUQ
Right upper quadrant
SBO
Small-bowel obstruction
T1W
T1 weighted
T2W
T2 weighted
TA
Trans-abdominal
TB
Tuberculosis
TCC
Transitional cell carcinoma
TIPS
Transjugular intrahepatic porto-systemic shunt
TURBT
Transurethral resection of bladder tumour
TURP
Transurethral resection of prostate
TV
Transvaginal
UC
Ulcerative colitis
UK
United Kingdom
US
Ultrasound
USS
Ultrasound scan
VIP
Vasoactive intestinal polypeptide
WBC
White blood cell count
xiii
A-Z OF ABDOMINAL
RADIOLOGY
A
Abdominal trauma
Clinical characteristics
• Ageneraldiscussion,followedbyorgan-specificsummaries,isgivenbelow.
• Abdominal trauma is managed as part of general trauma under the
ATLS (advanced trauma life support) algorithm, where basic care of
airway, breathing and circulation is followed by a secondary survey and
simultaneous management.
• Abdominal trauma is usually divided into blunt and penetrating trauma.
Penetrating trauma
•
Generally managed by surgical exploration and repair. Surgical repair
may be primary or secondary (delayed) according to a multitude of
factors, including degree of contamination, time since injury and gen-
eral health of the patient.
•
Where the patient is haemodynamically unstable, with signs of
obvious massive intra-abdominal haemorrhage, many of the investiga-
tions below are suspended, and the patient is managed surgically on
an emergency basis with coexistent haemodynamic monitoring and
management.
•
If the patient is haemodynamically stable, the usual radiological inves-
tigation is a contrast-enhanced computed tomography (CECT) scan.
This is usually performed following a radiographic primary survey
(lateral cervical spine, chest and pelvis radiographs).
•
It is now generally considered that diagnostic peritoneal lavage is rarely
indicated and has been superseded by CECT.
•
Emergency ultrasound (US) of the abdomen and pelvis is sometimes
performed and may be of use in determining the presence and location
of intra-abdominopelvic blood. Vascularity of abdominal viscera (e.g.
the kidneys) can also be assessed. US may also be used to monitor
patients who are being managed conservatively. However, CECT is
considered superior in the context of acute abdominal trauma.
Blunt trauma to the spleen
• Spleen is the most commonly injured solid intra-abdominal organ.
• Blunt trauma is the most common cause.
• Often (40%) associated with lower rib fractures and left renal injury.
• In 20% of patients with left rib fractures, there is a concomitant splenic
2
injury.
A
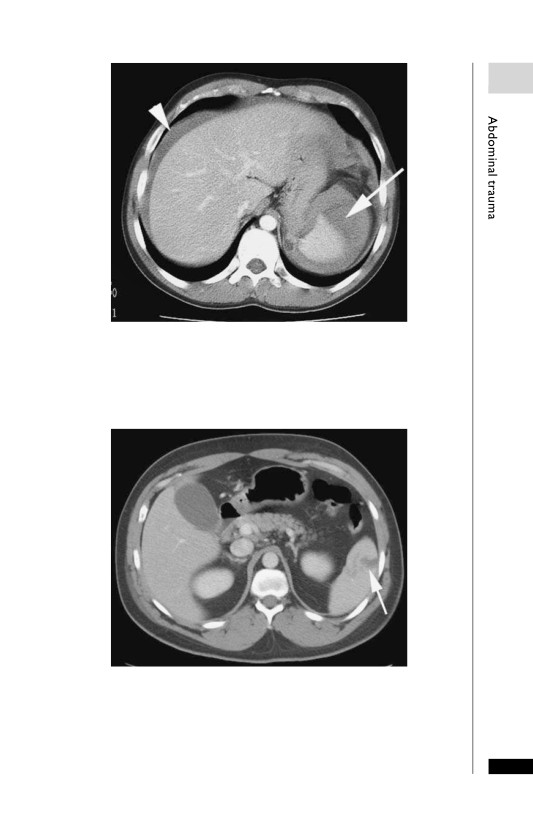
Rupture of the spleen. Rupture of the anterior half of the spleen
caused by blunt trauma in falling from a horse. Haemorrhage is seen
within the splenic bed (arrow) along with free blood around the liver
(arrowhead).
Splenic laceration (arrow).
•
25% of patients with left renal injuries also have splenic injuries.
• Damage ranges from subcapsular haematoma to total splenic laceration,
potentially leading to exsanguination.
3
Radiological features
A
•
Abdominal X-ray (AXR):
• Poor sensitivity in identifying both the site and presence of intra-
abdominal haemorrhage.
• May show associated fractures, displaced air-filled bowel caused by
intra-abdominal blood or free air from associated bowel perforation.
•
CECT:
• In nearly all cases of abdominal trauma, a CECT of the abdomen and
pelvis is performed to ensure adequate coverage of injuries.
• Mottled splenic parenchymal enhancement may represent contusion.
NB. The spleen in the arterial phase of a CECT may normally appear
mottled.
• Splenic fracture may present as complete separation of unenhancing
splenic fragments.
• Subcapsular haematoma is shown by a crescentic region of low
attenuation compressing normal parenchyma.
• Intracapsular haematoma is demonstrated by a hypodense inhomoge-
neous region within the spleen.
• Splenic laceration is revealed as a hypoattenuating line connecting
opposite visceral surfaces. Associated with perisplenic fluid.
• Multiple lacerations represent a shattered spleen.
• Disruption of the splenic capsule with high-density fluid within
abdomen represents splenic rupture with free intraperitoneal
haemorrhage.
Complications
• Splenic pseudocyst formation.
• Delayed rupture - up to 10 days later.
• Infected subcapsular haematoma.
• Splenic artery pseudoaneurysm.
Blunt trauma to the liver
• Second most frequently injured intra-abdominal organ.
• Associated with splenic injury in 45%.
• Right lobe more frequently injured.
• When left lobe is involved, there may be associated injury to the
duodenum, pancreas and transverse colon.
Radiological features
• AXR:
• Increased density in RUQ, displacement of neighbouring organs, e.g.
4
right kidney displaced downwards and medially.
A
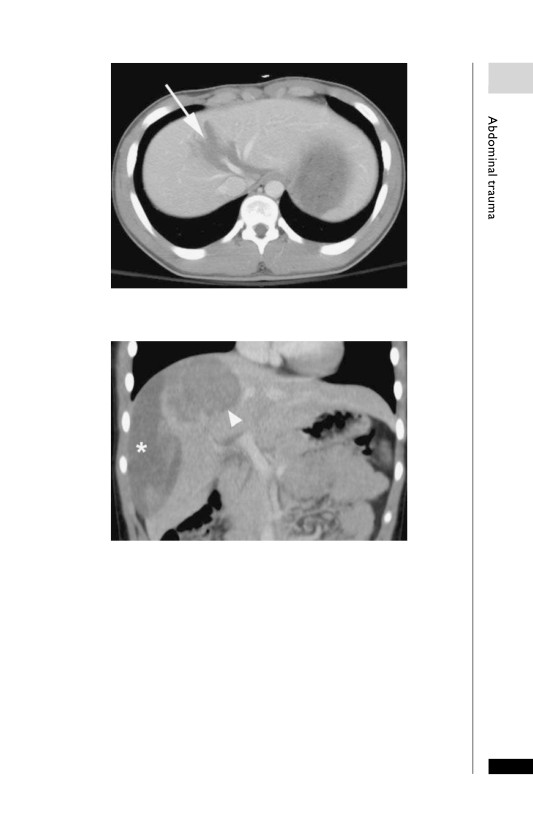
Large liver laceration (arrow).
Liver haematoma. Large haematoma within the superior aspect of the
right lobe of liver (arrowhead) with an additional subcapsular haematoma
(asterisk).
• CECT:
• Subcapsular haematoma - hypoattenuating lenticular configuration,
usually resolves in 6-8 weeks and generally managed conservatively.
• Hepatic laceration - irregular linear branching, single or multiple
stellate configurations of low density relative to enhancing
parenchyma.
• Focal hepatic devascularisation
- focal hypodense wedge lesion
extending to liver surface.
• Active haemorrhage - focal hyperdense area.
• Hepatic necrosis - intrahepatic or subcapsular gas.
5
Complications
A
• Occur in up to 20%.
• Haemobilia.
• Pseudoaneurysm/arteriovenous fistula formation.
• Biloma.
• Infection/necrosis.
• Delayed hepatic rupture - unusual.
Blunt trauma to the kidneys
• Occurs in 10% of blunt abdominal injury.
• Often caused by a direct blow (80%).
• Usually caused by laceration by lower ribs or devascularisation of the
renal pedicle in acceleration-deceleration injuries.
• Associated with other abdominal organ injury in 20%.
• Almost always presents with some degree of haematuria (over 95%).
• Main exception is with renal pedicle injuries, where 25% have no
haematuria due to devascularisation of kidney.
• Not usually accompanied by lower renal tract injury.
• Four degrees of injury, ranging from contusion and corticomedullary
laceration (grade I) to renal pedicle avulsion (grade IV).
Radiological features
• AXR: Plain film findings commonly seen in renal trauma include:
• Absent psoas shadow.
• Enlarged/distorted kidney and pelvicalyceal system
(following
administration of IV contrast).
• Fractures of the 10th, 11th and 12th ribs.
• Fractures of the transverse processes of the 1st, 2nd or 3rd lumbar vertebra.
• Scoliosis, concave towards the injured side, due to associated muscle
spasm.
• Localised small-bowel/colonic ileus.
• US:
• May show devascularisation, renal swelling from a diffuse haematoma,
peri- or pararenal haematoma, renal laceration or ureteric obstruction
from a ureteric clot.
• CECT:
• Renal contusion - focal areas of decreased contrast enhancement or
striated nephrogram.
• Renal laceration - irregular linear hypodense parenchymal areas.
• Renal fracture - laceration connecting two cortical surfaces.
• Shattered kidney - multiple separated renal fragments.
• Subcapsular haematoma - crescentic superficial hypodense area.
6
• Wedge infarction - wedge-shaped perfusion defect.
A
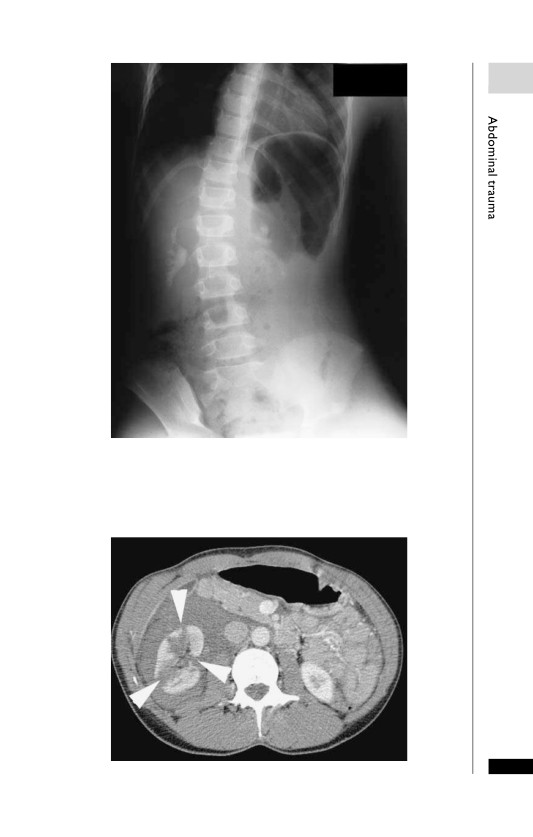
Closed blunt right renal trauma. There is asymmetry between the two
renal outlines, with distortion of the left pelvicalyceal system and left renal
outline. In addition there is a scoliosis of the thoracolumbar spine, concave
towards the injured side, and a localised ileus of the splenic flexure. Normal
contrast excretion seen from the right kidney.
7
Kidney trauma. Multiple fractures of the right kidney (arrowheads)
caused by blunt trauma from a kick by a horse. Extensive surrounding
perinephric haemorrhage.
• Renal vein thrombosis - persistent delayed nephrogram.
A
• Delayed scans are also often performed to check for a ureteric leak.
Blunt trauma to pancreas
• Usually caused by compression against the vertebral column; often
associated with seat belt compression injuries.
• Usually associated with upper abdominal visceral injury.
• Graded from minor contusion and capsular haematoma (grade I) to
severe devascularising crush injury (grade IV).
• Usually damage occurs at most vulnerable segment of pancreas: the
junction of the body and tail.
Radiological features
• CECT:
• Laceration - area of intrapancreatic low attenuation, often difficult to see.
• Direct evidence of haemorrhage - fluid around superior mesenteric
artery and pancreas.
• Indirect evidence - thickening of anterior pararenal fascia.
• Often requires delayed scans to exclude/monitor complications of
pancreatitis and devascularised pancreas.
Complications
• Post-traumatic pancreatitis, with:
• peripancreatic fat stranding
• diffuse or focal pancreatic enlargement
• irregular pancreatic contour.
• Splenic vessel fistula or arterial pseudoaneurysm.
• Pancreatic abscess.
• Pancreatic pseudocyst.
Blunt Trauma to the gastrointestinal tract
• The proximal jejunum is most commonly affected, followed by the
duodenum and ascending colon at the ileocaecal valve region. The
descending colon is only rarely involved.
Radiological features
• CECT:
• Appearances range from mesenteric or intramural haematoma to
8
frank colonic laceration and perforation.
A
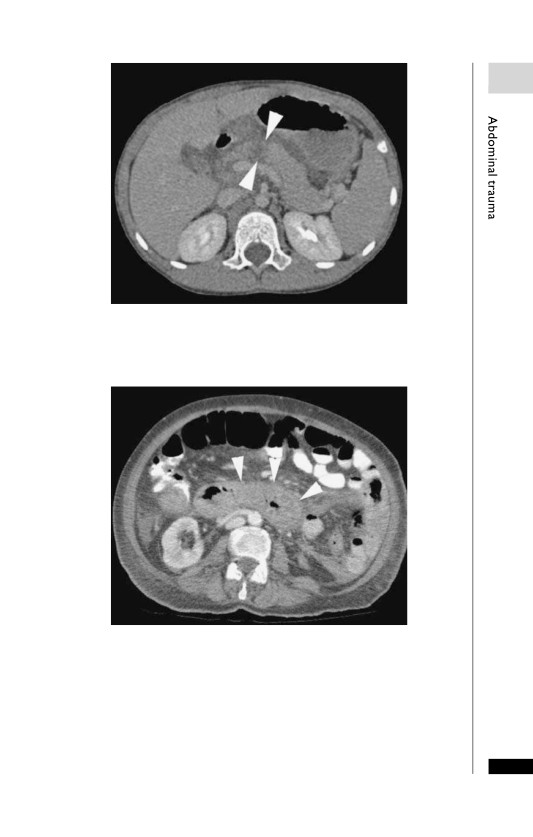
Pancreatic trauma. Laceration of the pancreas within the proximal
body (arrowheads).
Duodenal haematoma. Diffuse thickening of the third part of
duodenum secondary to a post-traumatic duodenal haematoma
(arrowheads).
• Subtle helpful signs include streaky hyperdense mesentery, sentinel clot
sign adjacent to local bowel injury, and hyperdense enhancement of
bowel wall in delayed venous phase caused by mesenteric vascular
damage.
9
Less-common abdominal trauma injuries
A
• Gallbladder.
• Ureter.
• Stomach.
• Adrenals.
Trauma to the bladder
Aetiology and frequency
The probability of bladder injury varies according to the degree of bladder
distension; a full bladder is more likely to be injured than an empty one.
• Approximately 10-25% of patients with a pelvic fracture also have
urethral trauma. Conversely, 10-29% of patients with posterior urethral
disruption have an associated bladder rupture.
• External trauma: 80%.
• Blunt injury: 60-85%:
- road traffic accident (RTA), 85%.
- fall, 10%.
- assault, 5%.
• Penetrating trauma: 15-40%:
- gunshot wound, 85%.
- stabbing, 15%.
• Iatrogenic: 15%.
• Includes gynaecological (post-hysterectomy), urological (after trans-
urethral resection of prostate (TURP) or bladder tumour (TURBT))
and orthopaedic (post-fixation of pelvic fractures) procedures.
• Intoxication: 4%.
• Spontaneous: <1%.
Classification of bladder rupture
• Extraperitoneal rupture: 50-75%.
• Intraperitoneal rupture: 25-45%.
• Incidence higher in children because of the predominantly intra-
abdominal location of the bladder prior to puberty.
• The bladder descends into the pelvis usually by the age of 20 years.
• Combined extraperitoneal and intraperitoneal rupture: 5-10%.
Extraperitoneal bladder rupture
• Traumatic extraperitoneal ruptures are usually associated with pelvic
fractures in up to
90% of patients. Conversely, approximately
10% of patients with pelvic fractures also have significant bladder
10
injuries.
A
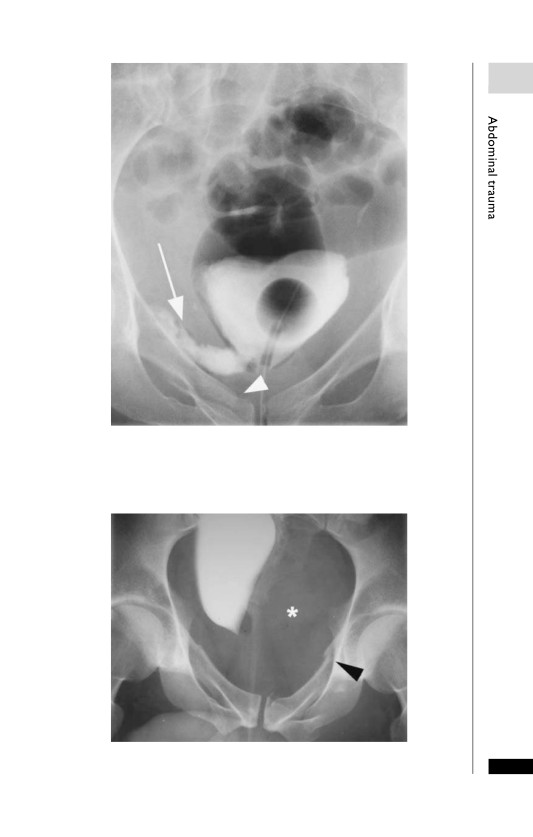
Extraperitoneal bladder rupture following a fall. Cystogram
demonstrates extravasation of contrast in to the right hemipelvis, tracking
along the peritoneal reflection (arrow). Note the fracture of the right
superior pubic ramus (arrowhead).
Large left pelvic haematoma (asterisk) secondary to a pelvic fracture
11
(arrowhead). Cystogram reveals that the bladder is elevated and compressed
to the right, producing a so-called ‘tear drop’ shape to the bladder.
• Results from the shearing force of the deforming bony pelvic ring,
A
which causes a ‘burst’ injury.
• Bony fragments may also cause direct laceration of the bladder and
the degree of bladder injury is directly related to the severity of the
fracture.
Intraperitoneal bladder rupture
• Occurs as the result of a direct blow to a distended urinary bladder. The
resulting increased intravesical pressure causes a horizontal tear along the
intraperitoneal portion of the bladder wall. This type of injury is
common amongst alcoholic patients or those sustaining blunt trauma
following a RTA.
Clinical characteristics
• Clinical signs of bladder injury are relatively non-specific; however, a
triad of symptoms is often present:
• gross haematuria
• suprapubic pain or tenderness
• difficulty or inability to void.
• Abdominal examination may reveal distension, guarding or rebound
tenderness.
• Absent bowel sounds and signs of peritoneal irritation indicate a possible
intraperitoneal bladder rupture.
• If blood is present at the urethral meatus, suspect a urethral injury.
Radiological features
• CT:
• Often the first test performed in patients with blunt abdominal trauma.
• Provides information on the status of the pelvic organs and bony
pelvis.
• CT cystography:
- The most sensitive test for evaluating a bladder perforation.
- Contrast is instilled into the bladder via a urethral catheter (or supra-
pubic catheter if contraindicated) followed by an abdominopelvic CT
scan.
- Intraperitoneal and extraperitoneal nature of a rupture can be
accurately assessed.
• Cystogram:
• Standard imaging for a suspected bladder injury.
- Consists of an initial plain radiograph of the kidney-ureter-bladder
(KUB).
- Followed by AP and oblique views of the contrast-filled bladder.
- Further AP film obtained after drainage.
12
A
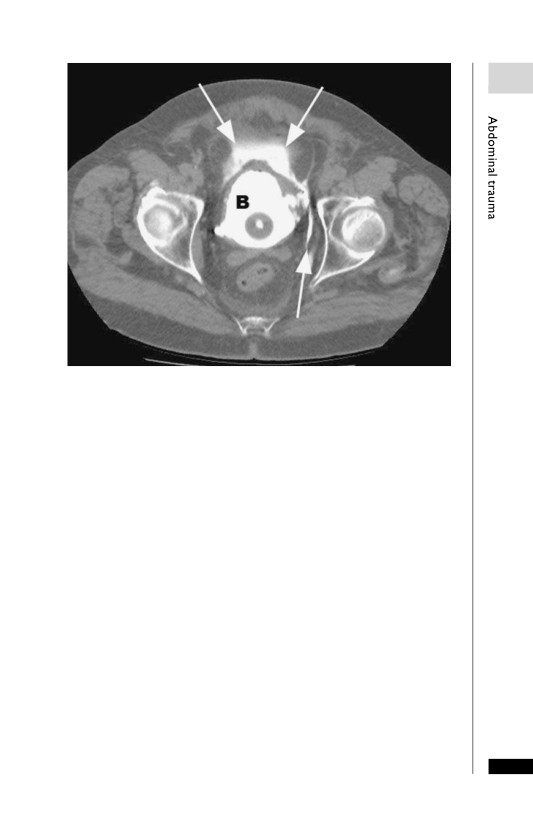
Extraperitoneal bladder perforation following a transurethral resection of
a bladder tumour. This CT cystogram clearly demonstrates contrast
extravasation into the perivesical soft tissues (arrows). Note the presence of
several bladder diverticula and a Foley balloon catheter within the bladder (B).
• Extraperitoneal rupture:
- Contrast extravasation around the base of the bladder is confined to
the perivesical space. The bladder may assume a tear-drop shape from
compression by a pelvic hematoma.
- With more complex injuries contrast may extend into the thigh,
penis, perineum or anterior abdominal wall.
• Intraperitoneal rupture:
- Contrast extravasation into the peritoneal cavity, often outlining
loops of bowel and the paracolic gutters.
• Combination of intra- and extraperitoneal rupture:
- Contrast outlines the abdominal viscera and perivesical space.
13
A
Abscesses within the abdomen
Peritoneal abcesses
Clinical characteristics
• Accumulation of collections of pus in the peritoneal spaces.
• Subphrenic abcesses are usually secondary to surgery.
• Paracolic abcesses are usually local to their cause, such as diverticulitis,
appendicitis or anastomotic failure.
• They present with raised inflammatory markers, swinging pyrexia, pain
and malaise.
Radiological features
• AXR: abscesses are often of soft tissue density and, therefore, difficult to
see. The presence of gas makes the identification easier. The finding
of gas pockets outside the bowel lumen, particularly if its appearance
does not change over time, is highly suggestive. A subphrenic gas-fluid
level on an erect chest X-ray (CXR) or erect AXR is suggestive of a
collection in this region.
• USS: an effective test for abdominal collections, being sensitive for fluid
collections or gas-fluid collections. It can also be used for guided
percutaneous drainage. Occasionally deep collections may be obscured
by overlying bowel gas.
• CECT: very sensitive for collections and may identify those obscured on
US. CT guidance can be utilised for percutaneous drainage.
• NM: using111indium labelled white cell scans are very sensitive for
detecting peritoneal abscesses but are infrequently used as the use of CT
increases.
14
A
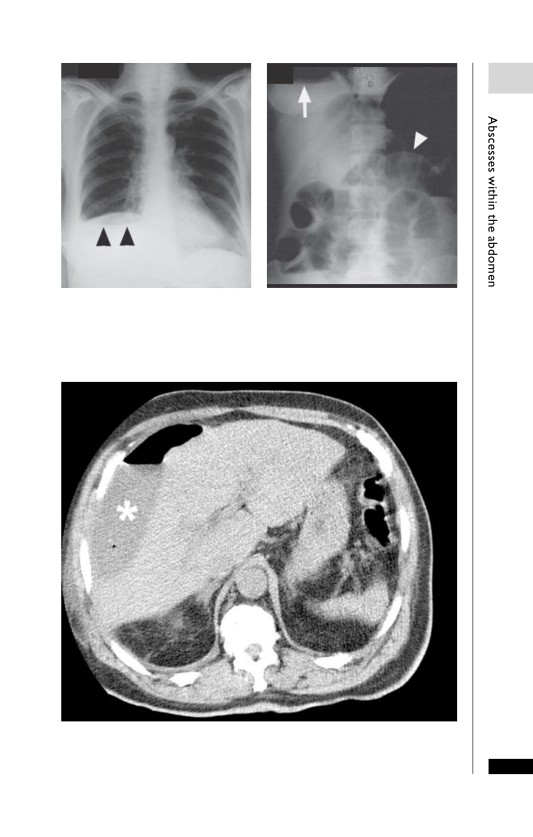
Right subphrenic abscess and free air. Air-fluid level below the right
hemidiaphragm (black arrowhead and white arrow) and a Riggler’s sign
(white arrowhead).
Large hepatic subcapsular abscess containing air (asterisk). Note the
effacement of the adjacent right lobe of liver, confirming the subcapsular
position.
15
A
Perirenal and renal abscesses
Clinical characteristics
• A complication of renal infection (see Pyelonephritis).
•
80% of cases result from ascending infection; may be related to obstruction.
• Haematogenous spread causes 20%.
• Urine culture may be negative.
• A renal abscess that extends through the renal capsule becomes a
perinephric abscess.
Radiological features
• AXR: provides clues as to the presence of a perinephric abscess. These
include loss of psoas shadow, focal mass seen in the renal outline,
displacement of the renal outline and gas in the renal bed. Calculi
may also be seen.
• USS: a hypo- or anechoic irregular mass, ± gas bubbles, ± perinephric
extension, may be demonstrated. Guided drainage can be performed.
• Intravenous urogram (IVU): a renal abscess may distort the pelvic-
alyceal system, but any mass lesion, such as a simple cyst, can have this
effect and hence an IVU is poorly specific.
• CT: may demonstrate a mass lesion with a low-density centre, and an
enhancing wall. Perinephric spread will be demonstrated and causal
factors, such as calculi, identified. Guided drainage can be performed.
• NM: inflammatory change in the renal bed may be demonstrated with a
radiolabelled white cell scan.
Hepatic abscesses
Clinical characteristics
• Most commonly secondary to biliary sepsis.
• Other causes include sepsis arriving via the portal venous system (i.e.
portal pyaemia, e.g. appendicitis), direct spread, indwelling arterial lines
and direct contamination associated with trauma.
• Pyogenic organisms (88%), amoebic (10%), fungal (2%).
• Presentation may include pyrexia, vomiting, abdominal pain, jaundice
and positive blood cultures.
16
A
Post-appendicectomy abscess. Note the complex multiloculated
collection deep to the incision (arrow).
Liver abscess. Heterogeneous collection within the right lobe of the liver
17
(arrow).
Radiological features
A
• USS: may demonstrate an irregular collection containing thick debris ±
trough enhancement, ± gas bubbles. Amoebic abscesses rarely contain
gas and may demonstrate internal septations. US-guided drainage may
be performed.
• CT: heterogeneous hypodense lesion with mural enhancement.
18
A
Pelvic abscess. Complex collection (C) deep within the pelvis. B,
bladder.
Pelvic abscess (same patient). large fluid collection, containing pockets
of air, deep within the pelvis (asterisk).
19
A
Achalasia
Clinical characteristics
• This is a motility disorder that results in failure of relaxation of the lower
oesophageal sphincter.
• The sphincter will only relax when the hydrostatic pressure of the
column of food, or liquid, in the oesophagus exceeds that in the
stomach, more usually in the erect position.
• May be of primary or secondary aetiology; most commonly primary.
Secondary achalasia occurs rarely in diseases such as malignancy, dia-
betes mellitus and Chagas’ disease.
• Peak age of incidence is 20-40 years, but it has been diagnosed in children.
• Most common presentation is dysphagia. Patients may also complain of
retrosternal chest pain from oesophageal distension and effortless regur-
gitation of undigested food.
• Complications include aspiration pneumonia and an increased inci-
dence of oesophageal cancer.
Radiological features
•
Plain X ray:
• An air-fluid level may be seen within the dilated oesphagus on the
chest radiograph.
• A small or absent gastric bubble may be present on the abdominal
radiograph, but this is non-specific and generally an AXR is not
helpful.
•
Barium studies:
• The barium swallow is the radiological investigation of choice.
• Findings include:
- Failure of oesophageal peristalsis to clear the oesphagus of barium.
- Failure of relaxation of the lower oesophageal sphincter.
- Poorly coordinated peristaltic activity.
- Oesophageal dilatation is a late feature.
- Narrowing of the oesophagus at the lower sphincter resulting in
characteristic ‘bird’s beak’ tapering.
• The barium study may be normal, particularly in the early stages of
the disease.
• Radiological findings should be confirmed by endoscopy and mano-
metry and, if necessary, biopsies to exclude a secondary cause such as
malignancy, or to exclude malignancy as a complication of long-
standing achalasia.
20
A
Achalasia. Note the large volume of debris within the dilated oesophagus
and the characteristic ‘bird’s beak’ tapering at the lower sphincter.
• The radiological findings characteristic of achalasia, with normal mano-
metric readings, are a feature of pseudoachalasia, a condition that occurs
in tumours of the distal oesophagus or lower oesophageal sphincter.
• CT: demonstrates the dilated oesophagus but this is non-specific.
• NM, MRI and USS: these modalities do not have a role in the
diagnosis of achalasia.
21
A
Adnexal masses
Clinical characteristics
•
In anatomical terms the adnexa are considered to include the uterine
(fallopian) tubes and ovaries. Each ovary lies posterolaterally on either
side of the uterus, attached to the broad ligaments by its own mesentery,
the mesovarium, and to the uterus by the ovarian ligament. The fallopian
tubes lie in the superior aspect of the broad ligament. Each tube is
approximately 10cm long and from the uterus runs posteriorly, laterally
and then inferiorly.
•
The presentation of ovarian disease includes pain, menstrual irregular-
ities, infertility, dyspareunia or abdominal distension, or it may be an
incidental finding.
•
Ovarian pathology can be divided into the following categories:
• Benign physiological cysts:
- follicular cysts
- corpus luteal cysts.
• Benign mixed solid/cystic lesions
- mucinous cystadenoma
- serous cystadenoma
- teratoma/dermoid
- endometriosis.
• Benign solid tumours
- granulosa cell tumour
- fibroma
- Brenner tumour.
•
Malignancy: primary/secondary.
•
Pathology of the uterine tubes can be caused by pelvic inflammatory
disease (PID), ectopic pregnancy or rarely carcinoma.
•
Acute PID will present with pain and fever and possibly a palpable mass
in the adnexa.
•
The chronic form is often asymptomatic and may only be diagnosed
during investigation for infertility or pelvic pain.
•
Non-gynaecological causes of adnexal masses include an appendix mass
or diverticular disease.
•
Imaging features of ovarian neoplasias are rarely definitively diagnostic.
22
A
Ovarian cyst. Follicular cyst within the right ovary (arrow).
Large anechoic right ovarian cyst (c) indenting the superior aspect
of the bladder (b).
23
Radiological features
A
•
AXR: plain films do not have a role in the diagnosis of adnexal
pathology. Any abnormality on an AXR is likely to be an incidental
finding (e.g. calcification within an ovarian dermoid cyst).
•
Contrast studies:
• The hysterosalpingogram is used to demonstrate the uterine tubes
using x-ray screening following the instillation of iodinated contrast
into the uterine cavity via the cervical os.
• It forms part of the investigation of infertility to assess tubal patency.
• The ovaries will not be visible on x-ray screening.
•
USS:
•
Trans-abdominal (TA) and transvaginal (TV) US is the modality of
choice for initial gynaecological imaging.
•
Because of the anatomy of the adnexa, the positions of the ovaries can
be very variable and dependent on the degree of distension of the
urinary bladder. Pregnancy also affects laxity of the ligaments.
•
The TA US is usually done with a full bladder and TV US with an
empty bladder.
•
For both TA and TV US, a good anatomical landmark for the ovaries
is the internal iliac artery.
•
Ovarian cysts are very common and are usually considered normal
unless >20-25mm in diameter (cut off varies between institutions) or
the patient is prepubertal, pregnant or postmenopausal.
•
Cysts
> 20-25mm in women of reproductive age are usually
rescanned at a different stage of the menstrual cycle, for example
2 weeks later, to document their physiological nature.
•
Malignant ovarian masses are usually more complex than benign
lesions and US is used to assess ovarian morphology.
•
Indicators of malignancy include larger size, thick cyst wall, internal
septations, solid components and mixed echogenicity cyst fluid.
Blood flow assessment and pulsed Doppler are also of use.
•
The normal non-dilated fallopian tube is not visible ultrasonically but
can be seen following the instillation of an US micro-bubble contrast
medium via the cervical os. This technique, known as hysterosalpingo-
contrast sonography (HyCoSy), is used as an alternative to the hystero-
salpingogram to assess tubal patency.
•
The abnormal dilated tube (a hydrosalpinx), in chronic PID for
example, appears as a serpiginous structure in the pelvis.
•
An important adnexal mass is that of ectopic pregnancy, of which
97% are tubal. The US findings in ectopic pregnancy are absence of
a uterine pregnancy, an adnexal mass and free intraperitoneal fluid.
24
A
Ovarian abscess. Complex multiseptated right ovarian mass with
eccentric wall thickening (callipers).
Ovarian carcinoma. Complex septated ovarian mass
25
(asterisk) containing a large enhancing solid component posteriorly.
•
CT:
A
• CT is of some use in diagnosing adnexal masses but US and MRI
provide more internal detail, owing to better soft tissue differentia-
tion, without the radiation dose.
• CT is sensitive for fat or calcification within a mass, for example in a
dermoid tumour.
•
MRI:
• Can provide excellent resolution of the pelvis and is very useful in the
assessment of gynaecological pathologies.
• Used with fat suppression, it is sensitive for the fat seen in dermoid
tumours.
• Sensitivity for different blood breakdown products means MRI is
sensitive for endometrial deposits where fluid-fluid levels, caused by
haemorrhage of different ages, are seen within endometriotic cysts.
•
MRI features that suggest malignancy in an ovarian cyst:
• size >4cm.
• soft tissue, non-fatty mural nodules.
• large soft tissue component.
• mural or septal thickness of >3mm.
• mural or septal irregularity.
• metastatic deposits or direct spread.
• presence of ascites and peritoneal nodules.
26
A
Bilateral ovarian carcinomata (asterisks). The right ovarian mass
is predominantly solid while the left is largely cystic with eccentric
wall thickening.
Right ovarian carcinoma: axial T1W MRI pre- and postcontrast. Large
enhancing solid right adnexal mass (arrow).
27
A
Adrenal masses
Clinical characteristics
• The adrenal glands have a ‘Y’ configuration with an anteromedial body
and two posterior limbs.
• The right adrenal gland lies between the right crus of the diaphragm and
IVC at the level of the upper pole of the kidney. The left adrenal
extends from the upper pole of the left kidney almost to the hilum. It
lies in front of the left crus of the diaphragm.
• Usually the limbs are 3-6mm thick and the width of the entire gland is
<1cm.
• The adrenal cortex produces glucocorticoids (cortisol), mineralocorti-
coids (aldosterone) and androgens. The adrenal medulla produces adre-
naline and noradrenaline.
Causes of adrenal masses
•
Functional:
• adenoma causing Conn’s or Cushing’s syndrome.
• phaeochromocytoma.
• adrenal carcinoma.
•
Malignant:
• metastases.
• carcinoma.
• lymphoma.
• neuroblastoma.
•
Benign:
• non-functioning adenoma.
• angiomyolipoma.
• cyst.
• haemorrhage.
•
The most common neoplasms are adenomata and metastastic disease.
A common indication for adrenal imaging is to differentiate between
these.
•
The presentation of adrenal masses depends upon whether the mass is
functional or not.
•
Non-functioning tumours such as adenoma are usually an incidental
finding when the patient is imaged for another reason.
28
(A)
(B)
A
Adrenal adenoma. ‘In-phase’ (A) and ‘out-of-phase’ (B) MRI confirms
loss of signal in a lipid-rich left adrenal adenoma (arrows) on the
out-of-phase image compared with the corresponding in-phase imaging.
Adrenal adenoma. Low-density non-functioning right adrenal adenoma
(arrow).
• With a functional adenoma, the presentation varies with the type of
hormone being produced in excess.
• Conn’s syndrome is caused by excess aldosterone and results in hyper-
tension, hypokalaemia and alkalosis.
• Cushing’s syndrome is caused by excess glucocorticoid and results in
myopathy, osteoporosis, water retention, obesity of the trunk, head
and neck, hypertension, predisposition to infection, easy bruising, and
hyperglycaemia.
• Phaeochromocytoma is a neuroendocrine tumour that secretes cat-
echolamines, resulting in hypertension, cardiomyopathy, weight loss
and hyperglycaemia. The ‘rule of 10s’ applies to phaeochromocy-
29
toma: 10% are adrenal, 10% are bilateral, 10% are malignant.
Radiological features
A
Adrenal adenomas versus metastatic deposits
•
USS:
• Although the adult adrenal glands may be visualised on US, it is
usually the surrounding fat that is visible.
• The adrenals appear hypoechoic relative to the fat but may only be
seen when abnormally enlarged.
• By contrast the neonatal adrenal is readily seen because of its larger
size relative to the other intra-abdominal organs, smaller patient size
and less retroperitoneal fat.
• Adrenal masses usually appear hypoechoic, but haemorrhage or
necrosis results in a heterogeneous appearance.
• It is important to differentiate adrenal masses from masses arising from
the adjacent liver, spleen, kidneys and pancreas.
•
CT:
• CT is a well-established modality for investigating adrenal masses
and uses the fact that the majority of adenomata have high lipid
content.
• A non-contrast CT is performed and the CT number (Hounsfield
units (HU)) of the mass measured. If the HU is ≤10 it is considered to
be a benign adenoma on the basis of its fat content.
• If the HU is >10, an enhanced 80-s and a 15-min delayed postcontrast
CT are obtained, and the enhancement washout calculated. The
washout is a measure of the percentage decrease between the
enhanced and delayed images.
• A large decrease is a high percentage washout and a small decrease is a
low percentage washout.
• If the enhancement washout is > 50%, the diagnosis of a benign lipid-
poor adenoma is made.
• If the washout is <50%, the mass is considered indeterminate, and a
biopsy may be necessary to make a diagnosis, particularly in a patient
with a new extra-adrenal primary neoplasm.
• Metastatic deposits are usually larger and more heterogeneous than
adenomata and do not have intracellular fat. Even in patients with
known malignant primaries, half of all adrenal masses will be benign.
30
A
Adrenal adenoma. Low-density bilateral non-functioning adrenal
adenomata (arrows).
Adrenal adenoma. Low-density functioning left adrenal adenoma,
producing Conn’s syndrome (arrowhead).
31
• MRI:
A
• MRI also makes use of the lipid content of adenomata.
• In-phase and out-of-phase (also known as chemical shift) imaging
demonstrates a loss of signal in a lipid-rich adenoma, on the out-of-
phase image compared with the corresponding in-phase imaging.
• A metastatic deposit does not demonstrate this loss of signal.
• Chemical shift imaging only provides the same information about an
adrenal mass as CT by identifying the lipid-rich subset of adenomata.
Intervention
• Adrenal venous sampling can be performed to identify whether the
abnormal hormone production is unilateral or bilateral: most com-
monly in primary hyperaldosteronism (Conn’s syndrome).
Phaeochromocytoma
Radiological features
• US:
• Phaeochromocytoma may be visible as a well-defined mass, which
may be solid or cystic to variable degrees.
• Echogenicity will be variable.
• CT:
• Phaeochromocytomas are usually large with a homogeneous density,
although larger masses may appear heterogeneous owing to haemor-
rhage or necrosis.
• Show strong contrast enhancement.
• Some authors believe that IV contrast administration may precipitate
a hypertensive crisis and recommend alpha and beta blockade prior to
IV contrast.
• MRI:
• Phaeochromocytoma have very high signal intensity on T2-weighted
(T2W) images, higher than adenoma or metastasis, and usually iso- or
hypointense on T1-weighted (T1W) imaging.
• Strong enhancement which may be heterogeneous depending on the
degree of cystic change.
32
A
Left phaeochromocytoma. Large heterogeneously enhancing
left adrenal mass.
Phaeochromocytoma: Axial T1W and T2W MRI. The lesion is
isointense on T1W and markedly hyperintense on T2W.
33
• NM:
A
• NM has a role in the diagnosis of phaeochromocytoma.131I-MIBG
and123I-MIBG are concentrated in the sympathomedullary system
and focal uptake is seen in phaeochromocytoma. Whole-body imag-
ing will demonstrate extra-adrenal tumours.
Adrenocortical carcinoma
• A rare carcinoma of the 4th to 7th decade.
• May present with Cushing’s syndrome if hyperfunctioning.
• Other presentations include pain and a mass.
• Usually >5cm in diameter at presentation.
• One-third show calcification.
• Can invade adjacent structures including the IVC.
• May metastasise to nodes, bones and lungs.
Radiological features
• USS:
• Tumour necrosis and haemorrhage result in heterogeneous
echogenicity.
• CT:
• Central area is of low density owing to necrosis.
• May be calcification and areas of haemorrhage.
• Irregular peripheral enhancement following contrast.
• MRI:
• The mix of haemorrhage and necrosis results in heterogeneous signal on
both T1W and T2W imaging.
• Irregular peripheral enhancement.
Myelolipoma
• A benign tumour composed of haematopoietic tissue and fat. Unlike
adenomas, the fat is discrete fat rather than intracellular.
• Can present with painful haemorrhage.
34
A
Phaeochromocytoma whole-body MIBG isotope scan. Uptake seen
within a left adrenal phaeochromocytoma (arrow).
Note physiological uptake in the liver and salivary glands, and excretion in
the bladder. The uptake over the right wrist corresponds to the injection
site.
35
Radiological Features
A
• AXR: not generally helpful but may demonstrate a discrete, lucent,
fatty mass or calcification caused by previous haemorrhage.
• USS: heterogeneous mass is a mix of fat and haematopoietic tissue.
• CT: discrete fat interspersed with fine bands of soft tissue.
• MRI: discrete fat is demonstrated by high signal in T1W that is sup-
pressed on fat-suppressed sequences.
Adrenal haemorrhage
• May be caused by trauma, physiological stress (such as sepsis), a bleeding
diathesis or haemorrhage into an underlying neoplasia.
Radiological features
• USS:
• Especially sensitive in neonates.
• Initially a solid mass that becomes heterogeneous through liquefac-
tion. No signal on Doppler interrogation.
• Chronically may have cystic appearance with echogenic calcific foci.
• CT:
• Oval mass with peri-adrenal fat stranding.
• Chronically, cystic change and calcification can occur.
• MRI: signal will depend on the age of haematoma and the associated
blood breakdown products.
36
A
Adrenal carcinoma. Large heterogeneous right adrenal mass (arrow).
The central low attenuation is secondary to necrosis.
(A)
(B)
Right myelolipoma. Axial (A) and coronal (B) reformatted CECT: a large
37
fatty mass replacing the right adrenal gland (arrows).
A
Aortic aneurysm
Clinical characteristics
• Afocalwideningoftheabdominalaortaof>3cm,involvingalllayersof
the vessel wall.
• Usual caused by atherosclerosis but may be secondary to trauma, infec-
tion, vasculitis or connective tissue disorders.
• Often asymptomatic.
• May present with a pulsitile mass, vessel rupture or an embolic event.
• Rupture classically presents with hypovolaemic shock, a pulsatile mass
and back pain.
Radiological features
•
AXR:
• Calcification of the aortic wall is a common finding in atherosclerosis.
• Loss of parallelism of the aortic wall suggests aneurysmal dilatation.
• Rarely vertebral body erosions may be seen with long-standing
aneurysms.
• In the acute scenario, loss of the psoas outline is associated with
retroperitoneal rupture.
•
USS:
• Is useful in both diagnosis and monitoring the size of abdominal aortic
aneurysms (AAA).
• In the acute abdomen, US can confirm the presence of an AAA, and
the presence of free abdominal fluid suggests rupture.
• However as CT is more sensitive and specific, US is most useful in the
case of a patient too unstable to be transferred to CT.
•
CECT:
• CT is used as part of elective surgical planning in determining the
anatomy of the AAA, particularly in relation to visceral vessels such as
the renal arteries.
• Retroperitoneal fibrosis associated with an AAA may be seen as a
surrounding soft tissue mass.
• In the acute setting, CT is the investigation of choice, often demon-
strating the precise site of rupture and is very sensitive to intraperi-
toneal and retroperitoneal haemorrhage.
• Rarer complications such as aorto-caval or aorto-enteric fistulae, and
occlusion, can be detected.
38
A
Aortic aneurysm. Calcification in the left lateral wall of the aneurysm
(arrowheads).
Ruptured aortic aneurysm. The arrowheads denote the breach in the wall of
the aneurysm (A), with extensive associated retroperitoneal haemorrhage (H).
39
A
Appendicitis
Clinical characteristics
• A common cause of an acute abdomen with a peak incidence in the
2nd and 3rd decades.
• The aetiology is probably related to luminal obstruction, often by
lymphoid hyperplasia or a faecolith.
• Typically presents with RIF pain, nausea, vomiting, fever and evidence
of inflammation such as raised WBC and CRP.
• However, one-third may have an atypical presentation.
• Complications include localised perforation, abscess formation and gen-
eralised peritonitis. Rarely an obstructed appendix becomes distended by
abnormal accumulation of mucus, forming an appendix mucocoele.
Radiological features
• AXR:
• Is neither sensitive nor specific but can provide clues.
• The presence of a calcified appendicolith in the RLQ, combined with
abdominal pain, has a high positive predictive value for acute
appendicitis.
• Other signs are less specific and include caecal wall thickening, small-
bowel ileus and decreased small-bowel gas in the RIF.
• Free peritoneal fluid can lead to loss of the psoas outline, loss of the fat
planes around the bladder and loss of definition of the inferior liver
outline.
40
(A)
A
(B)
Appendicolith. AXR (A) and coronal CT reformat (B) demonstrate the
presence of a RIF appendicolith (arrows). The CT demonstrates free fluid
within abdomen and pelvis and several dilated loops of small bowel,
41
secondary to ruptured appendicitis with an associated ileus.
• USS:
A
• Is the initial imaging of choice if there is diagnostic uncertainty.
• Can identify other causes of RIF pain such as ovarian torsion and
mesenteric adenitis.
• US findings that suggest appendicitis include:
- visualisation of a blind-ending, non-peristaltic, non-compressible
appendix.
- a diameter of ≥6mm.
- presence of an appendicolith, and distension of lumen.
- peri-appendiceal free fluid.
• NB A negative US does not exclude appendicitis; if there is a high
degree of clinical suspicion this should not preclude further imaging
or laparoscopy.
42
A
Ruptured appendicitis (arrowheads). Note the presence of an
ill-defined mottled gas pattern within the RIF, with an air-fluid level,
caused by the ruptured appendix.
43
• CECT:
A
• Is increasingly being used. However, it is not a first line investigation
owing to the radiation dose incurred by the patient.
• Tends to be used where there is diagnostic dilemma such as with an
atypical presentation.
• Findings include:
- a thickened appendix ± an appendicolith.
- inflammatory stranding in the adjacent fat.
- an inflammatory appendix mass.
- a local collection.
- local lymphadenopathy.
44
A
Appendicitis. Dilated tubular appendix containing an appendicolith
(arrow).
Appendicitis. Thickened tubular appendix, with inflammatory stranding
seen at its tip (arrow).
45
A
Ascites
Clinical characteristics
Intra-abdominal free fluid that may be classified as:
• Exudate: >30g/dl of protein; causes include peritoneal TB, pancreati-
tis, Meig’s syndrome and carcinomatosis.
• Transudate: <30g/dl of protein; causes include hypoalbuminaemia,
congestive cardiac failure, chronic renal failure, Budd-Chiari syndrome
and cirrhosis.
Radiological features
• AXR:
• Initial signs relate to the dependent accumulation of free fluid in the
pelvis and may be subtle and overlooked.
• Later signs are medial displacement of both the lateral border of the
liver and ascending and descending colon, bulging flanks, centralised
bowel loops and a generalised ‘greying’ of the abdominal film.
• USS:
• This is the investigation of choice to confirm the presence of ascites,
without the use of ionising radiation.
• US may provide additional information about the ascites such as
loculation or the presence of debris within the fluid.
• In addition, US allows the siting of diagnostic taps or therapeutic
drains.
• Evidence as to the aetiology of the ascites can also be gained, such as
the presence of cirrhosis.
46
A
Ascites. Generalised ‘greying’ of the abdominal film in AXR with
several centralised bowel loops.
Ascites: Abdominal US showing large-volume ascites (asterisk).
47
• CT:
A
• The radiation dose precludes this as an investigation to confirm the
presence of ascites, but CT often confirms the presence and extent of
ascites when performed for another reason.
• The cause may also be identified, such as evidence of pancreatitis.
• It is less sensitive than US in assessing for loculation or debris within
the ascitic fluid.
48
A
Ascites. Large-volume ascites (asterisk) and a small left basal pleural
effusion in CT image (arrow).
49
B
Bezoar
Clinical characteristics
• This is an intestinal mass caused by the accumulation of ingested
material.
• A phytobezoar is formed from poorly digested plant fibre.
• A trichobezoar is formed from ingested hair, almost always in females.
• Can lead to obstruction or ulceration.
50
B
Trichobezoar. Large ‘hair ball’ mass completely filling the stomach
(arrow).
51
Radiological features
B
• AXR:
• A mass may be seen within the stomach.
• May demonstrate bowel obstruction.
• Barium studies:
• May demonstrate an intraluminal filling defect that does not have a
fixed site of attachment to the bowel wall.
• Barium may flow into crevices within the bezoar.
• CT:
• This may demonstrate a low-density mass containing pockets of air.
• As on barium studies, oral contrast may intersperse with the mass
though gaps between the ingested materials.
52
B
Trichobezoar (same patient) in coronal CT reformat. Oral
contrast is seen outlining the huge trichobezoar.
53
B
Biliary gas
Clinical characteristics
• Gas within the biliary tree is often an incidental finding secondary to
medical intervention, such as a sphincterotomy or cholecystoenterostomy.
• Other causes include:
• a lax sphincter of Oddi in the elderly.
• passage of a gallstone.
• biliary fistulae caused by stones, neoplasia or duodenal ulceration.
• Biliary gas may result in gas within the gallbladder.
• Gas within the gallbladder may be secondary to emphysematous chol-
ecystitis. This is an infection with gas-forming organisms, seen in
diabetics and leading to mural and intraluminal gallbladder gas.
Radiological features
• AXR: branching radiolucencies are seen within the liver. These radio-
lucencies do not extend to the liver edge: a feature that helps to differ-
entiate biliary gas from gas in the portal vein. Gas in the gallbladder may
result in a gas-fluid level on an erect film.
• USS: linear echogenic shadows, paralleling the portal venous system,
are the characteristic appearance of biliary gas. Biliary calculi may be
identified.
• CT: branching air densities that parallel the portal system will be seen.
Calculi, fistulae or neoplastic masses may be identified.
54
B
Biliary gas Air in the biliary tree seen as linear echogenic shadows
paralleling the portal venous system (arrowheads).
Air in the biliary tree. CT elegantly demonstrates the air (arrows).
55
The relationship to the portal venous system is clear.
B
Biliary obstruction
Clinical characteristics
• Characterised by jaundice with a raised alkaline phosphatase and
.-glutamyl transferase.
• May be an acute or chronic presentation and may or may not be
accompanied by pain.
• In 75% of adult biliary obstruction, the cause is benign, including
calculi, strictures from previous trauma or surgery, pancreatitis and
sclerosing cholangitis.
• Malignant causes include pancreatic head carcinoma, ampullary and
duodenal malignancy, cholangiocarcinoma and metastases.
• The triad of a palpable, distended gallbladder, obstructive jaun-
dice and the absence of pain are highly suggestive of a malignant
cause.
• An obstructed, infected biliary tree is a medical emergency and occurs
more frequently with calculus than malignant obstruction (2:1).
56
B
Biliary obstruction. Abdominal US demonstrating dilated biliary
radicles (arrowheads) at the liver hilum.
Dilatation of the biliary tree. Note the fluid-filled ducts paralleling
57
the portal venous system (arrow).
Radiological features
B
• USS:
• Is the imaging modality of choice in establishing the mechanical cause
of biliary obstruction. The bile duct is usually easily visualised at the
hepatic hilum. Most normal ducts measure <5mm in diameter;
however, studies have shown that up to 4% of normal cases have a
duct that measures >7mm. Therefore a diameter of 6-8mm can be
non-specific and may require further investigation. Intrahepatic bili-
ary dilatation can be seen as dilated biliary radicals seen running along
the portal vessels.
• Will often establish the cause and level of biliary obstruction, with
visualisation of intraductal calculi or obstructing masses. Patient
factors (such as body mass index) or overlying bowel gas may obscure
the more distal biliary tree.
58
B
Obstructed common bile duct (arrow) caused by a carcinoma in the
head of pancreas demonstrated by ERCP.
59
• CT: is sensitive for biliary tree dilatation and, although less sensitive for
B
calculi than US, has the advantage of visualising the distal biliary tree
when it is obscured on US. It can also detect, and often accurately stage,
obstructing tumours.
• Magnetic resonance cholangiopancreatography (MRCP): use of
very heavily T2W imaging sequences to outline the fluid-filled bile
ducts can demonstrate the level of biliary obstruction very well. Stones
are demonstrated as filling defects. The addition of conventional axial
T1W and T2W images can give further information regarding extrinsic
causes of obstruction such as tumours.
60
B
Biliary obstruction. Tight hilar stricture (HS) caused by a
cholangiocarcinoma, resulting in dilatation of the right intrahepatic ducts
(RD) seen by MRCP. The left intrahepatic ducts (LD) are normal by
comparison but note that the tumour does cause tapering of the duct at the
hilum.
CD, common duct; PD, pancreatic duct. D2 and D3, second and third parts
of duodenum, respectively.
61
• Endoscopic retrograde cholangiopancreatography (ERCP): a
B
combined endoscopic/fluoroscopic procedure outlines the biliary tree
with similar effect to an MRCP, but it has the advantage of allowing
therapeutic procedures such as sphincterotomy with stone removal and
biliary stent placement.
• Percutaneous trans-hepatic cholangiography (PTC): the rise of
less-invasive techniques means that PTC is now performed less as a
diagnostic procedure. It is useful in patients who have had a failed
ERCP and in those who cannot have an MRCP. The use of PTC
has been extended to allow percutaneous trans-hepatic placement of
drainage catheters or stents, especially when this is not possible during
ERCP or in cases of an obstructed, infected system.
62
B
Gallstone impacted within the distal common bile duct (arrowhead)
on ERCP.
63
B
Bladder calculi
Clinical characteristics
• Bladder calculi may form around a foreign body such as prostatic
chippings, pubic hair, fragments of bone from penetrating injuries,
and pieces of Foley catheters.
• May migrate from the proximal urinary tract.
• May be caused by stasis such as outflow obstruction, neuropathic
bladders and vesicular diverticulae.
• Infection, especially with Proteus sp., can result in bladder calculi
formation.
Radiological features
• AXR: Seen as a calcified body projected over the bladder area. In
women, calcified fibroids may cause confusion although the latter
may be seen lying above, rather than over, the bladder area.
• USS: Confirms the position and nature of the calcified density.
64
B
Large bilobed bladder calculus (arrow).
65
B
Bowel obstruction
Although small- and large-bowel obstruction (SBO and LBO) have com-
mon clinical features of colicky abdominal pain, vomiting, absolute con-
stipation and abdominal distension, their aetiology and management are
quite different and, therefore, will be considered separately. Predominance
of certain signs depends on the level of the obstruction.
Small-bowel obstruction
Clinical characteristics
• SBO presents with colicky abdominal pain and vomiting, which occurs
earlier in more proximal obstruction and later in distal obstruction.
• The bowel distal to the obstruction empties by absorption or evacuation
of its contents, whereas the bowel proximal to the obstruction distends
with gas and fluid.
• This eventually results in fluid shifts into the bowel, causing hypovo-
laemia and electrolyte imbalances.
• If SBO is not reversed it may progress from a simple to strangulated
obstruction with compromise of bowel wall arterial supply with a risk of
perforation and peritonitis.
• The causes of SBO can be divided into intraluminal, luminal and
extrinsic causes:
• Intraluminal: foreign body, bezoar, parasites, gallstones, food bolus.
• Luminal: atresia, inflammatory stricture (Crohn’s disease, TB), hae-
matoma, tumour.
• Extrinsic: adhesions, congenital bands, malrotation, herniae,
intussusception.
• Adhesions, herniae and tumours cause the majority of SBO, with
postsurgical adhesions being the most common cause.
66
B
Acute small-bowel obstruction. Multiple dilated loops of small bowel
within the central abdomen.
67
Radiological features
B
•
AXR:
•
The jejunum lies mainly in the left hypochondrium and the ileum in
the pelvic midline.
•
In complete SBO, the small-bowel loops will distend in 3 to 5h and
are usually seen in the central abdomen.
•
An intraluminal width of >3cm on the AXR is abnormal.
•
Distended loops containing mainly fluid will appear as soft tissue
density. Occasionally, small pockets of gas trapped within the valvulae
conniventes create a ‘string of beads’ appearance.
•
A less frequent occurrence is gallstone ileus, when an intraluminal
calcified stone is seen, usually in the terminal ileum, with proximal
SBO and gas in the biliary tree owing to a cholecysto-duodenal fistula.
•
A closed-loop obstruction occurs when a loop of bowel is not
decompressed by the caudal passage of gas and fluid. This usually
occurs from adhesions and can result in a U-shaped distended loop of
small bowel that does not move over time. If it contains fluid, it
produces a pseudotumour appearance on the AXR because of its soft
tissue density; if it contains gas, it has a coffee-bean shape.
•
In children with an intussusception, a soft tissue mass may be seen on
the AXR and there may be signs of proximal SBO.
•
Chronic SBO may result in massively dilated bowel loops.
•
Intramural gas, as a result of bowel wall ischaemia, is usually associated
with a poor prognosis.
•
Usually a supine AXR is sufficient for assessment of a patient with
suspected SBO, but if it is normal, and there is a strong clinical
suspicion of obstruction, an erect AXR may help by demonstrating
air-fluid levels in dilated loops of small bowel.
•
A lateral decubitus AXR is indicated to show free gas if an erect CXR
cannot be performed.
68
B
Acute small-bowel obstruction secondary to an obstructed
right inguinal hernia (arrowhead).
Small-bowel obstruction secondary to a right femoral hernia. The
transition point is in the right femoral canal (arrow) with multiple dilated
69
proximal loops.
•
Small bowel enema (enteroclysis):
B
• This investigation is more appropriate for chronic SBO than acute
SBO and involves the placement of a nasojejunal tube and subsequent
instillation of a barium suspension.
• It will demonstrate a delay in the passage of barium and calibre change
at the site of obstruction.
•
USS:
• Distended fluid-filled loops of small bowel are clearly visible on US
and their peristaltic activity can be assessed.
• The distal bowel loses its muscular activity at the site of obstruction,
whereas proximally there is an initial increased peristalsis in an attempt
to overcome the obstruction. This distinguishes it from ileus, where
there is a generalised loss of peristalsis.
•
CT:
• When the AXR suggests SBO, CT can confirm the diagnosis,
indicate the level and may reveal a cause.
• IV contrast is given but oral contrast is not usually necessary as the
dilated loops contain fluid and gas, which act as intrinsic contrast.
• A small bowel diameter >2.5cm on CT is abnormal.
• A transition point, where the calibre of the bowel changes from
dilated to collapsed, indicates the level of obstruction.
• Adhesive bands are not visible on CT and the diagnosis is inferred on
the basis of a transition point and the lack of an identifiable cause.
• In closed-loop obstruction, CT will demonstrate a U-shaped loop of
bowel with a slightly twisted mesentery containing vessels.
• CT is also useful in assessing the presence of strangulation. The bowel
wall will appear thickened, with oedema and haemorrhage in the
mesentery. The use of IV contrast helps to assess bowel wall enhance-
ment, and in severe cases intramural gas will be present.
70
B
Small-bowel obstruction secondary to adhesions. Note the transition
point (arrowhead) in the RIF, with multiple dilated and fluid-filled proximal
small-bowel loops.
Small-bowel obstruction. Dilated fluid-filled small-bowel loops
71
secondary to a closed loop obstruction (arrow).
B
Large bowel obstruction
Clinical characteristics
• Abdominal distension predominates, with colicky abdominal pain, and
absolute constipation a feature of complete obstruction.
• Faeculent vomiting occurs if the ileocaecal valve is incompetent, when
the features of SBO are added to those of LBO.
• The most common causes of LBO are carcinoma of the colon, diver-
ticulitis and volvulus, with carcinoma being the most common.
• The clinical history may suggest the underlying cause: an abrupt onset of
symptoms suggests an acute event such as volvulus, a history of change
in bowel habit and weight loss suggests carcinoma, and a chronic history
of constipation suggests diverticulitis.
• Right-sided colonic lesions can become quite large before obstruction
develops owing to the soft stool consistency, whereas sigmoid and rectal
tumours obstruct earlier because of the narrower colonic calibre and
more solid stool.
Radiological features
• AXR:
• This may be diagnostic. The distended colon lies around the periph-
ery of the abdomen and is distinguished from small bowel by haustral
markings that do not traverse the entire bowel lumen.
• Bowel distal to the obstruction is collapsed and the rectum does not
contain gas.
• If there is a tumour of the caecum and ascending colon, with an
incompetent ileocaecal valve, only the small bowel may be distended.
• The typical appearance of a caecal or sigmoid volvulus may be
apparent (this is covered elsewhere).
• Intramural or free intra-abdominal gas may be seen.
• Water-soluble contrast enema:
• Awater-solublecontrastrather thanbariumisusedbecauseofthe risk
of intraperitoneal contamination when perforation has occurred; the
contrast is instilled rectally.
• It may confirm the diagnosis and indicate the level of obstruction.
• In most instances, contrast enemas have been replaced by abdominal
and pelvic CT studies.
72
B
Large-bowel obstruction. Gaseous distension of the colon, with
relative paucity of air beyond the mid descending colon.
Large-bowel obstruction (same patient as above). Contrast enema
73
demonstrates an obstructing lesion (arrow) within the mid descending colon.
• USS: may demonstrate fluid-filled loops of bowel and thickened bowel
B
wall, but the findings are often non-specific and generally not helpful.
• CT:
• This has advantages over contrast enema especially in elderly or frail
patients.
• It is usually performed with IV contrast; oral contrast may be helpful
to outline small-bowel loops.
• CT will confirm the obstruction, with a colonic diameter of >6cm
(9cm in the caecum) being considered abnormal.
• The identification of a transition point indicates the level and the
cause may also be apparent.
• As stated above, the most common cause is colonic carcinoma,
followed by diverticulitis and volvulus.
• One of the limitations of CT is the difficulty in differentiating
diverticulitis from colonic cancer in certain patients.
• CT will also assess the presence of complications, such as strangula-
tion, as indicated by congestive changes or haemorrhage in the
mesentery, bowel wall thickening, and intramural gas.
Pseudo-obstruction
Clinical characteristics
• Also known as Ogilvie syndrome, this is a clinical syndrome with the
signs, symptoms and radiographic appearance of LBO but with no
identifiable mechanical cause.
• Recognised causes are:
• recent surgery.
• severe pulmonary or cardiovascular disease.
• severe electrolyte disturbance: hyponatraemia, hypokalaemia, hypo-
magnesaemia, hypo/hypercalcaemia.
• malignancy.
• systemic infection.
• severe constipation.
• medications: opioids, anticholinergic drugs, clonidine, amphetamines,
phenothiazines, steroids.
74
B
Large-bowel obstruction secondary to an obstructing sigmoid carcinoma.
Note the extensive colonic distension up to, and including, the sigmoid colon.
Large-bowel obstruction secondary to an obstructing sigmoid
carcinoma, with extensive colonic dilatation and gross faecal loading.
75
Radiological features
B
• AXR:
• This does not differentiate pseudo-obstruction from mechanical
obstruction but will demonstrate distended loops of large bowel.
• Serial films may be useful to document the clinical course and mon-
itor the colonic diameter.
• Water-soluble contrast enema: a water-soluble contrast enema will
not only help to exclude a mechanical obstruction, by demonstrating
free passage of contrast, but may also be therapeutic when Gastrografin
is used, as its hyperosmolality causes a fluid shift into the bowel,
stimulating colonic motility.
• CT: further imaging is not usually necessary unless a mechanical
obstruction has not yet been excluded.
76
B
Pseudo-obstruction. Significant gaseous distension of the ascending and
transverse colon. In contrast with mechanical large-bowel obstruction, no
obstructing lesion can be identified to account for the appearances.
77
B
Ileus
Clinical characteristics
• Paralytic ileus usually occurs after intra-abdominal surgery but may also
occur in several other settings:
• Postoperative: most common.
• Inflammatory bowel disease.
• Inflammatory: pancreatitis, appendicitis, cholecystitis, diverticulitis,
peritonitis.
• Metabolic: hypokalaemia, hypocalcaemia, hypomagnesaemia.
• Medication: opioids.
• It occurs due to cessation of peristalsis, resulting in a functional
obstruction.
• It differs from pseudo-obstruction in that both the small and large bowel
are involved.
• The patient presents with abdominal distension and vomiting, usually
postoperatively.
• Colicky pain is not a feature and bowel sounds are absent, in contrast to
the high-pitched sounds of obstruction.
Radiological features
• AXR:
• Loops of both small and large bowel will be dilated with no apparent
transition point.
• Further imaging is only necessary if there is difficulty excluding a
mechanical obstruction.
78
B
lleus. Postoperative gaseous distension of both small and large bowel.
Note the midline surgical staples and the positive Rigler’s sign (arrows).
79
C
Calcifications
Numerous structures can calcify within the abdomen and pelvis. Most of
these are of no importance, but raise clinical concern when the AXR is
viewed!
• Costochondral calcification: age-related calcification of the costo-
chondral junctions is a common finding in the lower ribs projecting
over the superior part of the AXR. Typically in men, the calcification is
parallel to the edge of the cartilage, while in women the calcification is
central.
• Fibroid calcification: degenerate uterine fibroids (leiomyomata) may
calcify, resulting in a rounded, lobulated calcific body projected over
the female pelvis.
• Haematoma: some haematomas heal leaving a residual area of calci-
fication. Haematomas that follow an intramuscular injection into the
buttocks often calcify and may project over the pelvis and lower abdo-
men, possibly leading to diagnostic concern. These haematomas are
usually well defined, rounded or oval with rim calcification.
80
C
Costochondral calcification (asterisk) and splenic artery (arrow)
calcification.
81
Large calcified uterine fibroid (arrowhead).
• Mesenteric lymph nodes: postinflammatory nodes may calcify. If
C
mesenteric nodes are affected this may result in lobulated calcification,
which typically lies medially within the abdomen. This may cause
diagnostic confusion particularly in gallstone ileus. Calcified mesenteric
nodes are not evidence of active lymph node disease.
• Phleboliths: phleboliths are benign venous calcifications that com-
monly occur in the pelvic veins and may simulate distal ureteric stones.
A typical phlebolith is round with a slightly radiolucent centre.
However, they may be projected over the course of the lower ureter
and those with an atypical appearance may be indistinguishable from
ureteric stones. If this is a clinical concern, further imaging, such as an
IVU or non-enhanced CT, may be necessary.
82
C
Calcification of multiple injection sites (arrow). Note the sterilisation
clips within the pelvis.
83
Large calcified mesenteric node (arrowhead).
• Transverse processes: clearly calcification is normal in the transverse
C
processes of the lumbar vertebrae. However, the tips often appear
denser on AXR than the remainder of the transverse processes and
can, on occasion, be misinterpreted as ureteric calculi, as the courses of
the ureters often overlie the lower four transverse processes.
• Vascular calcification: vascular calcification is a common finding,
particularly in the elderly. Typically is seen as parallel lines of calci-
fication running along the course of arterial structures, most commonly
the aorta and iliac arteries. Often in the elderly these vessels have an
ectatic course, but the parallel nature of the calcification remains.
If parallelism is lost, aneurysmal dilatation should be suspected. The
splenic artery is often seen as a calcified serpiginous vessel in the left
upper quadrant.
84
C
Multiple calcified phleboliths (arrowheads).
Splenic artery calcification (arrows). Note the incidental pacemaker
lead (arrowhead).
Vascular calcification (arrowheads) and seminal vesical (arrows)
85
calcification.
C
Chilaiditi’s sign/syndrome
Clinical characteristics
•
Pronounced ‘Ky-la-ditty’.
•
Refers to the usually asymptomatic interposition of bowel between
the liver and right hemidiaphragm; usually is hepatic flexure, less com-
monly small bowel.
•
Seen in up to 0.25% of chest CXRs.
•
Most frequently an incidental finding in males and almost always in
adults.
•
Contributing factors include:
• absence of normal suspensory ligaments of the transverse colon.
• abnormality or absence of the falciform ligament.
• redundant colon, as might be seen in chronic constipation.
• aerophagia (air ‘swallower’).
• paralysis or eventration of the right hemidiaphragm.
• chronic lung disease, cirrhosis and ascites.
•
Chilaiditi’s ‘sign’ refers to the asymptomatic presence of interposed
bowel.
•
Chilaiditi’s ‘syndrome’ may present with:
• abdominal pain.
• constipation.
• vomiting.
• respiratory distress.
• anorexia.
•
It is clinically important because it may simulate a pneumoperitoneum,
resulting in unnecessary surgery.
Radiological features
• CXR: the presence of haustral folds within the ‘air beneath the dia-
phragm’ confirms it is contained within large bowel.
• CT:
• May be required if there is diagnostic dilemma.
• Particularly useful if there is strong clinical suspicion of concomitant
pneumoperitoneum, as this may be more difficult to diagnose.
86
C
Chilaiditi’s sign. CXR shows interposition of hepatic flexure between the
liver and right hemidiaphragm (arrow). Note the presence of multiple
colonic haustrations.
Chilaiditi’s sign. CECT shows interposition of the hepatic flexure
between the liver and right hemidiaphragm (arrow).
87
C
Cholecystitis
Clinical characteristics
• Inflammation of the gallbladder that presents with RUQ pain.
• May be acute or chronic; the most common aetiology is gallstones in
both.
• In acute cholecystitis, gallstones impact in the neck of gallbladder
or within the cystic duct, with spontaneous disimpaction in 85%.
• Complications include perforation, gallbladder necrosis and gallblad-
der empyema.
• acalculous cholecystitis may be caused by decreased flow in the cystic
artery, occurs more common in seriously ill patients, can be complicated
by perforation and has a 6% mortality rate.
Radiological features
• USS: mainstay of imaging in cholecystitis.
• Primary finding is of a thickened gallbladder wall (>3mm), which
may be poorly defined, with impacted calculi.
• A gallstone is identified as an echogenic body with posterior acoustic
shadowing.
• The presence of inflammatory fluid or exudate surrounding the
gallbladder is often seen.
• Inflammatory debris may be identified as coarse non-dependent
echogenic material that, unlike stones, has no acoustic shadow.
• Positive ultrasound Murphy’s sign in 85% Focal gallbladder tender-
ness upon direct pressure with the US probe.
88
C
Acute cholecystitis. Large calculus (arrowhead) impacted in the
neck of the gallbladder. Associated thickened and oedematous gallbladder
wall.
Acalculus cholecystitis (asterisk). Markedly thickened and oedematous
89
gallbladder wall with surrounding pericholecystic free fluid.
• CT: should not be necessary in the investigation of cholecystitis but
C
may be utilised as part of the investigation of non-specific abdominal
pain.
• Again the primary finding is of gallbladder mural thickening.
Associated inflammatory changes may be seen, characterised by
stranding in the adjacent fat. Biliary calculi may be identified.
• CT may be used to assess for complications such as perforation or
necrosis.
• MRI: can be helpful in difficult cases.
• MRCP may demonstrate biliary calculi.
• Standard MRI sequences may demonstrate gallbladder wall thicken-
ing; T2W scans, with fat suppression, can show associated inflamma-
tory changes and pericholecystic fluid.
90
C
Acute cholecystitis; CT features. Markedly thickened enhancing
gallbladder wall (arrow), with associated surrounding inflammatory
stranding.
91
C
Cirrhosis
Clinical characteristics
•
Cirrhosis is defined as hepatic fibrosis with the formation of regener-
ative nodules lacking normal hepatic architecture; particularly lack of a
central vein.
•
The aetiology is varied but a common underlying factor is repeated or
chronic inflammation and tissue destruction, with subsequent disorgan-
ised repair and regeneration.
•
There is a huge variety of causative agents. In developed countries, the
most common are viral (hepatitis B) and toxic (chronic alcohol abuse).
In some cases, no cause is found (cryptogenic hepatic cirrhosis).
•
Other less-common causes include;
• infestation with Schistosomes.
• chronic biliary obstruction; inflammatory bowel disease, cystic fibrosis,
primary biliary cirrhosis.
• haemochromatosis.
• vascular insufficiency from hepatic veno-occlusive disease or congestive
heart failure.
• nutritional: intestinal bypass procedures, abetalipoproteinaemia and
steatosis.
• drug-induced: particularly with methotrexate, isoniazid and
methyldopa.
• genetic: Wilson’s disease, type IV glycogen storage disease and
,1-antitrypsin deficiency.
•
A pseudo-cirrhotic picture is seen in patients with treated hepatic
metastatic disease and should not be confused with real hepatic cirrhosis.
•
Morphologically there are two main types of cirrhosis:
• Chronic sclerosing cirrhosis involves minimal regeneration
of hepatocytes with little nodule formation leading to a small hard
liver.
• Nodular cirrhosis shows significant regeneration, leading to the
formation of multiple small nodules. In the early stages, the liver
may be enlarged.
•
Deranged intrahepatic venous flow results in bypassing of portal venous
flow directly into the systemic vasculature, leading to the development
of submucosal porto-venous varices, most commonly in the distal
oesophagus and rectal mucosa.
•
Patients present with signs and symptoms of chronic liver failure and
complications from portal hypertension.
•
Clinical features include jaundice, low-grade fever, anorexia and weight
loss, and nephrotic syndrome caused by low serum albumin (deficient
92
production by the liver).
C
Cirrhosis. Shrunken liver with an irregular margin and coarsened
echotexture. Note the presence of caudate lobe hypertrophy (arrowhead)
and ascites (asterisk).
• Clinical features of portal hypertension include hepatic encephalopathy
(from bypassing of the liver’s detoxifying capability secondary to porto-
systemic shunts) and risk of significant, often life-threatening, haemor-
rhage from oesophageal or rectal varices.
• There is an association with hypogonadism, anaemia, pancreatitis,
coagulopathy and cholelithiasis.
Radiological features
• Radiological features include changes visible within the liver, changes
owing to portal hypertension and changes from chronic hepatic
insufficiency.
• AXR: some of these changes, such as initial hepatic enlargement, ascites
and splenomegaly, may be seen on plain radiography but this is not
sensitive or specific.
• USS:
• US has an overall sensitivity of 80% in the detection of changes from
cirrhosis.
• On US, the liver may be enlarged initially with subsequent shrinkage
noted.
• Because of differential portal supply, the right lobe is often more
affected than the left, with subsequent shrinkage of the right lobe
(including the quadrate lobe), with hypertrophy of the left lobe and
caudate lobe (segment 1).
• Surface nodularity, increased parenchymal echogenicity, a coarse
heterogeneous texture and surface indentations are all seen on USS.
93
• Coexisting splenomegaly and signs of portal hypertension such as
C
portal varices and ascites are common.
• On duplex imaging, there is generally increased hepatic arterial
resistance following a meal and so-called ‘portalisation’ of the hepatic
venous waveform.
•
CT:
• On CECT, a shrunken liver with an irregular drawn-in surface is
generally seen. Liver density and enhancement following IV contrast
is variable.
• Fatty infiltration as seen in early cirrhosis may lead to a hypodense
enlarged liver. Generally there is non-uniform enhancement owing
to areas of fatty infiltration and areas of hepatic fibrosis.
• Hypertrophy of the caudate lobe may be seen.
• Liver density may, however, be normal. Splenomegaly and ascites are
easily detected by CECT.
•
MRI: is often used as a problem-solving tool in liver imaging, and
cirrhosis is no exception.
• An irregular area within the liver, as seen on US and CECT, may
represent a regenerating cirrhotic nodule, a dysplastic nodule or a
frankly malignant nodule (cirrhosis is a strong risk factor for the
development of hepatocellular carcinoma, particularly if caused by
viral infection).
• Regenerating nodules are hypointense on T1W and T2W sequences
because of iron deposits within them. Malignant nodules tend to be
variable on T1W and hyperintense on T2W images. They show
marked early contrast enhancement following gadolinium on T1W
sequences.
94
C
Cirrhosis. Axial scan in the portal phase. As on US, the liver has an
irregular in-drawn margin and there is hypertrophy of the caudate lobe.
Note that the liver is generally of reduced attenuation in comparison with
the spleen, consistent with diffuse fatty infiltration.
Cirrhosis. Shrunken liver, with an irregular margin (arrowhead), and
95
ascites (arrows).
C
Colitis
• Colitis is inflammation of the colon.
• Causes include inflammatory bowel disease, ischaemia and infection.
Ulcerative colitis
Clinical characteristics
• Ulcerative colitis (UC) is an idiopathic inflammatory bowel disease that
affects the colon and usually starts in the rectum. It causes circum-
ferential, continuous proximal inflammation of the colon and, unlike
Crohn’s disease, skip lesions are not a feature.
• The diagnosis is usually made using endoscopy and biopsy.
• There may be a backwash terminal ileitis but otherwise GI involvement
is limited to the colon.
• Presents with abdominal pain and bloody diarrhoea. May be accom-
panied by electrolyte imbalances and fevers.
• The usual pattern is of periods of disease activity interspersed with
periods of remission.
• There are extra-colonic manifestations, which include erythema nodo-
sum, iritis, primary sclerosing cholangitis, chronic active hepatitis,
peripheral arthritis and spondylitis.
• Complications include toxic megacolon, with its associated high risk of
perforation and death. More chronic complications include stricture
formation and markedly increased risk of colonic carcinoma.
Radiological features
• AXR: plain films may demonstrate hyperplastic ‘mucosal islands’; dif-
fuse colonic dilatation, with loss of the normal haustral pattern; and
paucity of faecal material owing to inflammation.
• Barium enema (acute phase): may demonstrate narrowing and
incomplete filling owing to spasm. Different types of ulcer may be
seen: undermining (collar stud) ulcers, shallow ulcers or longitudinal
submucosal ulceration, resulting in a double tract of barium. Fine
granularity of the mucosa may be seen owing to a combination of
mucosal oedema and fine ulceration. Oedematous haustra may result
in thumb printing. Pseudopolyps may be caused by oedematous areas of
mucosa separated by areas of denuded mucosa.
• Barium enemas (chronic stage): may demonstrate a featureless,
narrow-calibre lumen - ‘hose pipe’ colon. Malignant transformation
may be seen. Surveillance for malignant change is usually via endos-
copy, thus avoiding radiation in what is often a young population.
• CT: may demonstrate circumferential wall thickening >10mm. A
96
continuous distribution from the rectum is suggestive of UC.
C
Late-stage ulcerative colitis. Single contrast enema demonstrates an
extensive featureless colon with loss of haustrations, narrowing of the
lumen and associated shortening. There is also mucosal irregularity in the
transverse colon consistent with ulceration.
Ulcerative colitis. Diffuse circumferential colonic wall thickening
97
(asterixes) due to UC.
Differential diagnosis
C
One of the main differential diagnoses for UC is Crohn’s disease
Differences between UC and Crohn’s disease
Feature
Crohn’s disease
Ulcerative colitis
GI tract
Can affect any segment Colon and reflux terminal ileitis
only
Skip lesions
Present
None; continuous starting at
distal colon
Colonic shortening None
Yes, plus loss of haustra in
chronic cases
Ulcers
Deep, fissuring
Shallow confluent
Fistulae
A feature
None
Pseudodiverticulum Yes
None
Toxic megacolon Rare
Relatively common
Carcinoma
Slight increase in colonic Marked increase in rate of
carcinoma rate
colonic adenocarcinoma
98
C
Pseudomembranous colitis. Marked circumferential sigmoid colonic
wall thickening with mucosal enhancement.
Ischaemic colitis. Note the characteristic ‘thumb printing’ of thickened,
oedematous folds in the distal transverse colon (arrows).
99
C
Pseudomembranous colitis
Clinical characteristics
• Colitis induced by a toxin produced by Clostridium difficile.
• Predisposing factors include antibiotic therapy, shock, proximal LBO,
intestinal ischaemia and renal transplantation.
• Results in profuse watery diarrhoea, abdominal pain and tenderness.
• Endoscopy shows pseudomembranes.
• Diagnosed by visualisation of the pseudomembranes and stool assay for
the toxin.
Radiological features
• AXR: may demonstrate an ileus with mild gaseous large and small
bowel distension. Thumb printing may be a feature. The haustra may
appear thickened owing to oedema and the pseudomembranes may
cause an irregular, shaggy mucosal appearance.
• Barium enema: generally not required and is contraindicated in severe
cases. May demonstrate barium within clefts in the pseudomembranes.
The pseudomembranes may cause a nodular or shaggy appearance.
• USS: may demonstrate non-specific colonic wall thickening and the
presence of ascites.
• CT: may demonstrate circumferential colonic wall thickening with
mucosal enhancement.
100
C
Late-stage ischaemic colitis. Single-contrast barium enema
demonstrates a clear zone of transition between normal and abnormal
colon at the junction of the middle and distal thirds of the transverse colon.
The proximal colon has normal mucosa and haustral pattern while the distal
segment is featureless and abnormally narrowed.
Superior mesenteric artery (SMA) thrombosis. Normal enhancement
101
of the aorta (arrowhead). No enhancement seen in the SMA (arrow).
C
Ischaemic colitis
Clinical characteristics
• Ischaemic colitis is caused by interruption to the colonic blood supply.
• Aetiologies include thrombosis, bowel obstruction and trauma.
• Predisposing factors include age, oral contraceptives, sickle cell disease
and surgical ligation of the inferior mesenteric artery.
• Presents with acute lower abdominal pain and tenderness, usually out of
proportion to the clinical signs. There may be rectal bleeding or
diarrhoea.
• Most commonly affects the left side of the colon, especially at the
splenic flexure where there is a watershed between the territories of
the superior and inferior mesenteric arteries. The rectum is usually
spared.
• May be a transient condition with spontaneous resolution over a few
months. May lead to incomplete healing with smooth stricture forma-
tion. Severe disease can lead to colonic infarction, with a high associated
mortality.
Radiological features
• AXR: plain film is often normal; however, gas within the colon may
outline the characteristic thumb printing of thickened, oedematous
folds seen in this condition.
• Barium enema: single-contrast instant enema may demonstrate
thumb printing and ulceration associated with this condition. A double-
contrast enema shows these findings more reliably but should be used with
caution in an acutely ill patient. A smooth stricture may be demonstrated
on a delayed study.
• CT: contrast-enhanced spiral CT is the usual first-line investigation for
suspected ischaemic colitis. A dual phase scan, performed in the arterial
and portal phases, may demonstrate thrombus in both the mesenteric
arterial or venous systems. The affected colon may appear abnormally
circumferentially thickened and demonstrate poor contrast enhance-
ment. There may be a sharp cut off between normal and abnormal
colon at the boundary of vascular territories. Mural gas may be seen in
more advanced disease and, in severe cases, portal gas may be identified.
The latter is a poor prognostic factor.
• Angiography: a more limited role in the era of multislice spiral CT but
may demonstrate attenuated arterial flow or the presence of a thrombus.
102
C
Mesenteric venous thrombosis: extensive serosal and mucosal
enhancement caused by mesenteric venous thrombosis.
103
Bowel necrosis. Portal venous gas (arrowhead) secondary to necrosis.
C
Radiation colitis
Clinical characteristics
• A late complication of radiotherapy that may present many years after
therapy. Typically involves doses of >45Gy.
• The radiation causes an occlusive endarteritis.
• Most commonly occurs in the rectum following treatment of gynaeco-
logical malignancy.
• May cause rectal bleeding, tenesmus and diarrhoea.
Radiological features
• Changes are confined to the radiotherapy field.
• Barium enema: may show strictures; these will often be smooth and
symmetrical, but prior ulceration may lead to scarring and distortion of
colonic folds. Fistulae may also occur.
• CT: may demonstrate luminal narrowing and homogeneous colonic
wall thickening. There may be thickening of pararectal fascia by fibrosis.
The surrounding fat may show increased density, again from fibrosis.
104
C
Small-bowel infarction with the development of extensive mural gas
(arrows).
105
C
Toxic megacolon
Clinical characteristics
• This is an acute fulminant full-thickness colitis.
• There is loss of smooth muscle tone, with rapid development of exten-
sive colonic dilatation (>5.5cm).
• Most commonly seen in UC.
• Other causes include Crohn’s disease, ischaemic colitis, pseudomem-
branous colitis and infective colitis.
• Untreated, this results in colonic perforation, with an associated mortal-
ity of 20%.
106
C
Toxic megacolon. Oedematous mucosal ‘islands’ (arrowheads) seen in a
dilated transverse colon (TC).
107
Radiological features
C
• AXR: plain films demonstrate colonic distension/colonic ileus. This
may be localised or involve the whole colon. The loss of the normal
haustral pattern and development of mucosal islands suggests mucosal
ischaemia. Mural gas and free peritoneal gas are late complications.
• Serial AXR: may demonstrate increasing dilatation. Associated small-
bowel dilatation indicates poor prognosis for medical treatment.
• CT: demonstrates colonic dilatation, with distorted haustra and mucosal
islands. Late complications such as mural gas and free intraperitoneal air
are readily identifiable.
• Barium enema: contraindicated because of the risk of perforation.
108
C
Toxic megacolon. Extensive colonic distenstion with multiple mucosal
‘islands’ throughout, indicating developing mucosal ischaemia; this is
usually a precursor to perforation.
109
C
Colonic carcinoma
Clinical characteristics
• Most common GI carcinoma. Causes 15% of cancer deaths.
• Predisposing conditions include familial polyposis coli, Gardner’s syn-
drome and inflammatory bowel disease.
• Presentations include weight loss, iron-deficiency anaemia, abdominal
pain, rectal bleeding, perforation, intussusception and colonic
obstruction.
• Common sites for secondary spread include liver, lung, lymph nodes
and adrenals.
Radiological features
• AXR: useful in secondary large-bowel obstruction but otherwise AXR
has a non-specific role in diagnosing colonic carcinoma.
• Barium enema: double-contrast barium enema has, in the past, been
the radiological investigation of choice. May demonstrate the classic
annular ‘apple core’ lesion or a fungating polypoid lesion. Rarely,
longitudinal and circumferential spread (scirrhous carcinoma) is seen,
especially related to chronic UC.
110
C
Colonic carcinoma. ‘Apple core’ stricture of the mid descending colon
caused by a carcinoma.
Annular carcinoma at the splenic flexure (arrowheads) resulting in
proximal large-bowel obstruction; note the dilated fluid-filled proximal
colon.
111
• USS: can detect large carcinomas, nodal and hepatic spread, but is
C
relatively insensitive for primary colonic carcinoma.
• CT pneumocolon: excellent investigation for demonstrating the
abnormal mural thickening of colonic carcinoma. It has the advantage
over barium enemas of being able to demonstrate extra-colonic
involvement, and incidental non-colonic pathology.
• Standard CECT:
• Can demonstrate the larger carcinomas, and oral contrast 24h prior to
the scan increases the sensitivity. Psammomatous calcification may be
seen in mucinous adenocarcinoma.
• Can accurately stage colonic carcinoma, including intrathoracic
spread.
• Complications such as perforation, obstruction and abscess formation
can be assessed.
112
C
Sigmoid carcinoma. Large obstructing carcinoma (asterisk).
See the following three related images.
Sigmoid carcinoma. Gastrograffin enema confirms an obstructing
proximal sigmoid carcinoma (arrowheads). Only a thin ‘string’ of contrast
113
is seen traversing the carcinoma.
• MRI: important in local staging of rectal carcinoma, as it is more
C
sensitive than CT in staging pelvic malignancy. Contrast-enhanced
MRI is more sensitive than CT in detecting hepatic metastases.
• Fluoroscopic stenting: in inoperable disease, a palliative fluoro-
scopically placed expanding stent can be used to preempt or treat
LBO.
114
C
Fluoroscopic stenting. Following the insertion of an expandable metal
stent (asterisk) to relieve the obstruction, the proximal bowel is
decompressed.
Fluoroscopic stenting. The position of the colonic stent is clearly
assessable on CT.
115
C
Colonic diverticulitis
Clinical characteristics
• Diverticular disease of the colon occurs when overactive smooth muscle
causes herniation of the mucosal and submucosal layers of the colon
through the muscle layers. Predominantly seen within the sigmoid
colon, although diverticular disease may be present anywhere within
the colon.
• Colonic diverticular disease is very common in Western societies where
the diet is low in roughage. Approximately 50% of those over 60 will be
affected. Up to a quarter may suffer from diverticulitis.
• Diverticulitis occurs when a colonic diverticulum becomes inflamed,
often by a faecolith. This presents with abdominal pain, usually in the
LIF, fever and a raised WBC.
• Complications include perforation, abscess formation and fistula
formation into adjacent structures, such as the uterus or bladder.
• Treatment is often conservative with antibiotic therapy, but surgery or
radiological intervention, such as percutaneous abscess drainage, may be
required.
Radiological features
• AXR: plain films may demonstrate a localised ileus, gas in a fistula or
abscess, or occasionally a pneumoperitoneum.
• Barium enema: demonstrates colonic diverticula very well and may
also demonstrate complications such as fistulae, perforation and abscess-
eses. The lumen of the colon affected by diverticular disease is usually
narrowed owing to muscular hypertrophy.
• USS: detects thickened bowel and fluid collections, but the diverticula
themselves and small gas collections are very difficult to identify. US is
generally less sensitive than CT but can be used for percutaneous
drainage.
116
C
Extensive sigmoid diverticular disease.
117
• CECT: CT is the best imaging modality for visualising diverticulitis.
C
Diverticular disease is seen as small outpouchings arising from the affected
colon. These are usually gas filled, but high density within them may be
from a previous barium enema or a calcified faecolith. The affected colon
is often thickened by smooth muscle hypertrophy. In diverticulitis, the
pericolic fat becomes ‘stranded’ and indistinct as a result of inflammatory
oedema. Fluid- and gas-filled abscesses may be seen, as may pockets
of free gas. Fistulae may be directly demonstrated or inferred through
the presence of gas within adjacent involved organs such as the bladder
and uterus. CT can be used to guide percutaneous drainage of fluid
collections.
• It is important to remember that a carcinoma can occur within a
segment of colon affected by diverticular disease, and this can lead to
diagnostic confusion. Carcinoma often causes a shorter segment of
bowel thickening with heaped-up margins - the so-called ‘apple core’
lesion seen in barium enemas.
118
C
Caecal diverticulitis. Extensive inflammatory ‘stranding’ surrounding a
thickened caecum (arrow); note the localised posterior perforation
(arrowhead).
Acute sigmoid diverticulitis. Several sigmoid diverticula are present
(arrowheads) and the surrounding fat is ‘dirty’ owing to local inflammatory
change.
119
C
Colonic polyps
Adenomatous colonic polyps
Clinical characteristics
• Adenomatous colonic polyps are often asymptomatic but have the
potential to undergo malignant change. Adenomatous polyps of 5-9mm
have a 1% incidence of malignant change while those >2cm have an
incidence of almost 50%. Polyps >10mm need to be excised.
• Symptoms include diarrhoea, abdominal pain, rectal bleeding and
hypokalaemia.
Radiological features
• Endoscopic examination of the colon has the advantage of allowing
biopsy and excision of any polyps, but it is an invasive procedure, and
examination of the whole colon may be incomplete in a percentage of
patients.
• Barium enema:
• Double-contrast barium enemas have been the mainstay in colonic
imaging in the past, having a false-negative rate of 7% for polyps <10mm.
• Colonic polyps have characteristic appearances on barium enema, the
main cause of diagnostic confusion being differentiation from colonic
diverticula.
• The table shows the features of colonic polyps.
120
C
Pedunculated polyp (arrows) seen within the distal sigmoid colon.
Large rectal adenoma ‘carpeting’ the rectum. Contrast is seen outlining
121
irregular filling defects within the rectum.
Features of colonic polyps on barium enemas
C
Feature
Characteristics
Meniscus sign
Barium forms a meniscus around the base of a polyp. When
viewed en face, the inner border of the meniscus is well
defined and the outer less well defined. In the en face view
of a diverticulum, the outer border is the better defined.
Hat sign
The ‘hat sign’ results from an oblique viewing of a polyp. The
barium meniscus forms the rim of the ‘bowler hat’, and the
barium covered polyp the body of the hat
Target sign
An en face view of a pedunculated polyp will demonstrate a
‘bull’s eye’ appearance, caused by the meniscus around the
base superimposed on the view of the barium-covered head
of the polyp
Increased density
A polyp is an intraluminal soft tissue that is of increased density
sign
compared with the gas-filled lumen in a good-quality
double-contrast enema. When the polyp is coated in
barium, this can be seen as a focus of increased density
Negative filling
Barium pools dependently and if a polyp is sitting in this pool
defect
it may be visible as a negative filling defect
Stalk sign
An oblique view of a pedunculated polyp may show the
outline of the stalk as two parallel barium lines that flare
slightly where the stalk merges with the mucosa. The axis of
the stalk may change as the patient’s orientation does during
the study
• CT pneumocolonography:
• Increasingly is replacing barium enema as the radiological investiga-
tion of choice. With multislice CT scanners, polyps of 5mm can be
detected with high sensitivity.
• There are a number of advantages of CT pneumocolonography over
barium studies. Although the same bowel preparation is used, the
study is quicker and easier for patients. It involves a slightly higher
radiation dose but provides more diagnostic information, such as
staging information about a colonic carcinoma or demonstrating
incidental, but significant, pathology.
122
C
Prone CT pneumocolon. Large pedunculated polyp within the sigmoid
colon (arrowhead).
10mm polyp in the ascending colon (arrowhead).
123
C
Post-inflammatory colonic polyposis
Clinical characteristics
• This is non-neoplastic and results from the re-epithelisation of inflam-
matory polyps.
• Most commonly seen in UC but can be seen after ischaemic or infective
colitis.
• It has no malignant potential.
• The differential diagnosis includes familial polyposis.
Radiological features
• Barium enema: a characteristic appearance with filiform projections,
attached only at their bases.
124
C
Inflammatory polyps. Barium enema in a patient with long-standing UC.
Inflammatory pseudopolyps can be seen within the proximal sigmoid colon
(arrow) and an irregular shouldered carcinoma within the distal sigmoid
colon (arrowhead).
125
C
Crohn’s disease
Clinical characteristics
• An inflammatory condition that can affect any portion of the GI tract.
Skip lesions (normal areas between affected ones) are typical and help to
distinguish colonic Crohn’s disease from UC.
• The terminal ileum is the commonest site affected.
• The inflammation can affect all histological layers of the GI tract.
• Presentation may include weight loss, recurrent diarrhoea, anaemia,
abdominal pain, peri-anal fistulae, abscesses and malabsorption.
• Complications include fistulae and abscess formation, stenosis, SBO,
adenocarcinoma and lymphoma.
• The differential includes UC, enteric TB, Yersiniae infection, lymphoma
and radiation ileitis.
126
C
Early Crohn’s colitis. Multiple shallow aphthous ulcers within the caecum
(arrows).
127
Radiological features
C
• AXR: may demonstrate bowel wall thickening, related to inflammatory
change, as well as complications such as perforation, toxic megacolon and
abscess formation. However these changes are non-specific.
• USS: as Crohn’s disease is a chronic condition that often affects
young patients, repeated high-dose radiation studies, such as barium
follow-through, raises the risks of long-term complications such as
radiation-induced lymphoma. US can be used to assess for the mural
thickening (>8mm) associated with active disease, in patients with
known Crohn’s disease, without exposing them to ionising radiation.
US can also detect abscesses and guides the placement of drains.
128
C
Crohn’s colitis. Multiple deep ‘fissuring’ ulcers (arrows) seen within the
distal transverse colon.
129
• Barium studies:
C
• Early features include shallow aphthoid ulcers, enlargement of
lymphoid nodules and thickening and distortion of the valvulae
conniventes of the small bowel.
• Later features include cobble-stoning of the mucosa, from serpigi-
nous ulcers with intervening oedema, and rigid and straightened
small-bowel loops, caused by spasm and oedema. Small-bowel
loops may also appear separated owing to increased mesenteric fat
and nodes. Post-inflammatory pseudopolyps may be seen. Fistulae
may be demonstrated: entero-colic, entero-cutaneous or to other
viscera. Pseudodiverticula are caused by outpouchings of normal
bowel opposite fibrosed areas.
• In the end stages, stenotic sections are seen with ‘strings’ of barium
running through them. These are most common at the terminal
ileum. Proximal dilated loops may be seen.
130
C
Late-stage Crohn’s disease. Long irregular stricture within the terminal
ileum (arrows).
Crohn’s disease by CT enterography. Short segment stricture (arrow)
within the terminal ileum (arrow).
131
• CT: thickened bowel wall may show a double-halo configuration; the
C
halo is the lumen surrounded by low-density oedematous mucosa,
which in turn is surrounded by a higher density thickened and fibrotic
muscularis. The increase in mesenteric fat may also be seen. Oral
contrast outlines fistulae, strictures and abscesses; the latter can be drained
under CT guidance.
• MRI: demonstrates thickened, enhancing bowel loops. T2W fat-
suppressed MRI is especially useful in demonstrating the anatomy of
peri-anal fistulae prior to surgery. MR enteroclysis shows promise as a
radiation-free replacement for the barium follow-through and small-
bowel enema.
132
C
Crohn’s ileitis. Hyperaemic thickened terminal ileum (arrowheads).
Crohn’s terminal ileitis. Diffusely thickenened terminal ileum (arrow)
with surrounding inflammatory stranding (asterisks).
133
D
Dermoid tumour
Clinical characteristics
• Dermoid tumour is also known as a mature cystic teratoma.
• A benign ovarian tumour that contains mature ectodermal tissues, such
as fat, hair and teeth. Forms up to a quarter of ovarian tumours.
• May present as an abdominal mass or acute abdominal pain if torsion
occurs.
• Often an incidental finding on imaging.
• Malignant degeneration occurs in 1-2%.
Radiological features
• AXR: can be diagnostic when a soft-tissue-density mass is seen within
the pelvis containing focal areas of fat density, bone or teeth.
• CT: may show fat-fluid interfaces within the tumour. A soft-tissue
nodule (Rokitansky nodule) may project into the cyst. This is made up
of sebaceous material.
• USS: advantageous compared with CT as it incurs no radiation dose. It
may demonstrate a complex mass with echo-poor cystic areas, echo-
genic areas of fat, a fat-fluid level and highly echogenic areas from teeth
and other calcification.
• MRI: again has the advantage of no ionising radiation. Although not as
sensitive as CT for calcification, MRI is very sensitive for detecting the
diagnostic fat within a dermoid tumour.
134
D
Dermoid tumour. A tooth and additional calcific debris seen within
a dermoid tumour (arrow).
(A)
(B)
Dermoid tumour. Large echogenic fatty mass (asterisk) seen posterior
to the bladder on US (A). The low-density fat within the tumour is
confirmed on CT (arrow) (B).
135
E
Ectopic pregnancy
Clinical characteristics
• Ectopic pregnancy is implantation of an embryo outside the uterine
cavity.
• It is associated with a history of a previous ectopic pregnancy, tubal
surgery, pelvic inflammatory disease, endometriosis and use of an IUCD.
• Usually presents by the 7th week of pregnancy.
• Most (95%) occur in the fallopian tube.
• Other sites are ovarian, intra-abdominal and cervical.
• Symptoms include vaginal bleeding, abdominal pain and an adnexal
mass.
• May present with hypovolaemic shock in a ruptured ectopic pregnancy.
Radiological features
• May be diagnosed by TA or TV US. The latter is more sensitive but also
more invasive.
• USS: only specific finding by TA/TV US is visualisation of a live
ectopic embryo but this is seen in less than 20% of cases. Supportive
findings include no intrauterine pregnancy at 6 weeks of gestation,
pelvic free fluid or hyperechoic clot, hydro- or haematosalpinx or
a thickened endometrium. A pseudogestational sac may be seen -
consisting of endometrial thickening with an anechoic centre composed
of haemorrhage.
• Extensive intra-abdominal and pelvic haemorrhage may be seen in
women with a ruptured ectopic pregnancy. This can be assessed on
both US and CECT.
136
E
Ectopic pregnancy. Pseudogestational sac (arrow) with free fluid in the
pouch of Douglas (asterisk).
(A)
(B)
Ectopic pregnancy. The uterus (A) appears normal with no intra-uterine
pregnancy identified. However an extra-uterine gestation sac is seen in
the right adnexa (B).
Ruptured ectopic pregnancy. Axial and coronal CT show extra-uterine
gestation sac (arrow) with extensive haemoperitoneum (asterisk).
137
E
Endometrial carcinoma
•
Endometrial carcinoma is the most prevalent female cancer of the
genital tract and 4th most prevalent cancer in women.
•
Adenocarcinoma make up 90-95%.
•
Tumours of epithelial and mesenchymal origin
(sarcomata) form
5-10%; such as Leiomyosarcoma, malignant mixed müllerian tumours,
adenosarcomas, gestational trophoblastic tumours.
•
Incidence in UK is 4900 per annum, 990 deaths annually.
•
Disease of postmenopausal women; peak age 55-62 years, 75% over the
age of 50 years.
•
Risk factors:
• exposure to unopposed oestrogens, such as extrinsic oestrogen ther-
apy and tamoxifen, is common risk.
• obesity.
• nulliparity.
• ovarian malfunction in polycystic ovaries.
• late menopause.
•
Other associations are:
• diabetes mellitus.
• smoking.
• hypertension.
• association with breast cancer.
•
90% arise within uterine epithelium: 90% of these are well-differentiated
adenocarcinoma (grade I).
•
Endometrial cancer arises in the glandular component of endometrial
epithelium.
•
Grows as a polypoid mass within the endometrial cavity, producing
ulceration and vaginal bleeding.
•
Spread is by:
• direct invasion of the myometrium and extension through to the
serosa.
• direct invasion of parametrium with serosal seeding.
• direct extension down endocervical canal.
•
Uterus has a rich blood and lymphatic supply, a common route of
spread.
•
Lymph node and haematogeneous metastases are common:
• upper uterine body tumours spread to common iliac and para-aortic
nodes.
• mid and lower uterine body tumours spread to parametrial, para-
cervical and obturator nodes.
• metastases via round ligament and vaginal extension produce inguinal
adenopathy.
•
Distant spread is to bone, lungs, liver and brain.
138
E
Endometrial carcinoma. TA US of the pelvis demonstrating abnormal
endometrial thickening (arrow). Asterisk indicates the bladder.
Endometrial carcinoma. TV US of the pelvis demonstrating abnormal
endometrial thickening (arrow/calipers). Asterisk indicates the body of
uterus.
Clinical characteristics
• Usually presents with intermenstrual/postmenopausal vaginal bleeding.
• Occasionally in advanced disease, presents with sequelae of distant
spread to target organs or peritoneum.
• Rarely presents with an abdominal (uterine) mass.
139
Radiological features
E
•
FIGO classification used to stage uterine cancer:
Stage I: tumour confined to endometrium or myometrium (A-C).
Stage II: invasion of cervix (A-B).
Stage III: invasion of serosa, adnexae (A) or vagina (B) with nodal
metastases (IIIC).
Stage IV: invasion of bladder/bowel (IVA) or distant metastases (IVB).
•
Depth of myometrial invasion is the most important prognostic factor:
incidence of nodal mestastases rises from 3% (stage IB) to 40% (stage IC).
•
TV US, CT and MRI are all capable of assessing myometrial invasion.
•
Early endometrial disease best assessed by direct visualisation and biopsy.
•
Cross-sectional imaging best for more advanced disease.
•
USS (TV/TA):
• TV/US in the preferred modality due to greater sensitivity.
• Increase in endometrial thickness >5mm, usually echogenic, irregu-
lar poorly defined boundary.
• Myometrial invasion demonstrated as disruption of the normally
smooth interface between the endometrium and myometrium.
• Depth of invasion assessed by proportion of myometrium occupied
by echogenic tumour.
• Accuracy is 77-91%.
• Cervical involvement often detected.
• Usually superior to CT, equivalent to MRI for myometrial invasion
assessment.
• Extra-uterine and nodal spread not accurately determined.
• Diagnostic value of Doppler blood flow measurements is
controversial.
•
CECT:
• Demonstrates endometrial tumour as a hypodense mass in the endo-
metrial cavity or myometrium, or fluid-filled uterus caused by
endocervical canal obstruction by the tumour. Not used for local
staging.
• Capable of detecting deep myometrial invasion.
• May show cervical extension but less accurate than TV US or MRI.
• Accuracy of 58-76%.
• Often cannot differentiate from benign uterine masses, so less useful
in early disease.
• Most useful for advanced disease and detection of distant metastases,
pelvic extension and nodal mestastases.
•
MRI:
• Excellent for local staging.
• Widening or heterogeneity of signal within endometrial canal may be
only sign in stage IA carcinoma (confined to endometrium).
• Endometrial tumour has lower signal than endometrium, and higher
signal than myometrium.
140
(A)
(B)
E
Endometrial carcinoma. Axial (A) and sagittal (B) T1 MRI of the pelvis
after intravenous contrast. Abnormal enhancing soft tissue is seen
expanding the endometrial cavity (asterisk). The body of uterus is markedly
thinned (arrow). B, bladder.
• Disruption or absence of junctional zone implies myometrial inva-
sion; however, junctional zone may not be visible in some post-
menopausal women.
• Myometrial invasion shown as areas of relatively high signal within
the low-signal myometrium.
• Contrast-enhanced T1W images clearly define zonal anatomy and
improve accuracy.
• Endometrial cancer enhances more slowly than endometrium (bright
on T1W) or myometrium (dark on T1W).
• Relationship of tumour to cervix is important prognostically.
• Multiple planes used to assess both longitudinal and radial tumour
spread.
• Cervical epithelium is hyperintense on T2W and late post-gadolinium
T1W images; disrupted in cervical extension.
• MRI is superior to TV US and CT in predicting cervical stromal invasion.
• MRI is inferior to hysteroscopy in detecting mucosal involvement.
• Overall sensitivity of MRI is 82-94% in detecting deep myometrial
invasion.
141
F
Familial polyposis coli
Clinical characteristics
• An autosomal dominant condition with 80% penetrance that is charac-
terised by a myriad of (~1000) colonic adenomatous polyps.
• The polyps develop at the age of puberty.
• Symptoms include vague abdominal pain, bloody diarrhoea and
protein-losing enteropathy.
• The main complication is malignant transformation. By 20 years fol-
lowing diagnosis, almost 100% will have developed colonic carcinoma.
There is also a lesser increase in the incidence of gastric and small-bowel
malignancy.
• The treatment is total colectomy in the late teens or early twenties.
Radiological features
• Generally now diagnosed in family members by colonoscopy, but
sporadic cases may present at barium enema with a myriad of small
polyps forming a ‘carpet’ throughout the colon. There may be evidence
of carcinoma, often with more than one synchronous tumour.
• CT colonography can reliably detect polyps of 5mm and smaller but
represents a significant radiation dose in patients who are usually in their
teens at the time of investigation.
142
F
Familial polyposis coli. The colon is ‘carpeted’ in multiple small polyps.
Familial polyposis coli (same patient; enlarged view of the sigmoid
143
colon). Multiple polyps are clearly evident (arrowheads).
F
Fistulae
Clinical characteristics
• A fistula is an abnormal communication between two epithelialised
surfaces. Examples include biliary-enteric, entero-cutaneous, aorto-
enteric, entero-vesical, ano-rectal, vesico-colic.
• The causes include trauma, surgical complication, infection and inflam-
mation, for example secondary to inflammatory bowel disease and
diverticular disease.
• Clinical presentation will depend on the type of fistula.
144
F
Vesico-vaginal fistula. Film from an IVU series. Contrast within the
bladder (asterisk) is seen within the vaginal vault (arrow).
Colo-vesical fistula. Barium enema decubitus film (left side down) shows
diverticular stricture (arrowhead) and an air-barium level within the
bladder (arrows).
145
Radiological features
F
• Imaging of a fistula is aimed at identifying its exact path and any
underlying disease to aid surgical repair.
• AXR:
• Plain radiography generally is not helpful in diagnosing fistulae.
• Fluoroscopy following the instillation of iodinated contrast into a
cutaneous fistula (fistulogram) can be useful to demonstrate the tract.
• Barium: similarly barium in the bowel, either as a small bowel follow-
through or barium enema, or water-soluble contrast in the bladder as a
cystogram, may be used to demonstrate a fistula.
• Nonetheless, it is important to be aware that the fistulous tract may be
very small and, therefore, difficult to see on these studies.
• Despite this, such studies may be of use in delineating the extent of
underlying bowel disease.
146
(A)
F
(B)
Aorto-enteric fistula. Axial CT before (A) and after (B) contrast. On
the unenhanced scan, a small pocket of gas is seen along the anterior wall
of the aortic graft (arrow), caused by infection. Following contrast,
enhancement is seen within several small bowel loops (asterisk) from a fistula.
147
• US, CT and MRI:
F
• All these modalities may be of use in imaging fistulae.
• A mass may be seen associated with the fistula, particularly when it is
caused by an inflammatory condition such as diverticular disease or
inflammatory bowel disease.
• The tract may also be visible.
• Air may be seen within the bladder in a colo-vesical fistula.
• Usually CT is the most useful of these cross-sectional modalities.
• Angiography:
• Angiography may be necessary to help to diagnose an aorto-enteric
fistula between the aorta and bowel, most commonly the third or
fourth part of the duodenum.
• This usually occurs in a patient with a preexisting aortic graft who
presents with an upper GI bleed, pain or a pulsatile mass.
• Alternatively, an aorto-enteric fistula may be investigated with CT
pre- and postcontrast.
148
F
Entero-cutaneous fistula. Fistulogram where contrast is injected via a
cannula though the cutaneous ostium (arrow) demonstrates a fistula to both
small bowel and caecum (arrowheads). Note the presence of an IVC filter
adjacent to the L3 vertebral body.
149
F
Foreign bodies
Clinical characteristics
• Foreign bodies may appear in abdominal imaging following trauma,
iatrogenic intervention, and patient ingestion or insertion - either
intentionally or accidentally.
• There is a wide range of presentation, ranging from asymptomatic to in
extremis.
• Children tend to swallow coins, marbles, disc batteries and crayons.
• Adults tend to swallow dentures, chicken and fish bones or may obstruct
on a food bolus.
• Bizarre ingestions are more likely with psychiatric patients and a sub-
group of prison inmates.
• Within the oesophagus there are three points of narrowing where a
foreign body is likely to impact: the cricopharyngeus, at the level of the
aortic arch and at the lower oesophageal sphincter.
• Once a foreign body has passed in to the stomach, there is an 80-90%
chance of it passing through the GI tract.
• The main obstacle to passage through the small bowel is the ileocaecal
valve.
• Complications include laceration, perforation, associated peritonitis,
abscess formation and infection. Disc batteries may cause oesophageal
erosion.
150
F
Foreign body. Swallowed screw within the stomach (arrow).
151
Foreign body. Swallowed coin within the stomach (arrow).
Radiological features of ingested foreign bodies
F
• The imaging of an ingested foreign body consists of a CXR to assess
whether the body lies in the oesophagus.
• In children, this should include the neck.
• For fish and chicken bones, a soft tissue lateral radiograph of the neck is
indicated.
• In adults, a lateral CXR may also be needed if the frontal radiograph is
negative.
• If the foreign body has passed into the stomach, the patient can be
reassured.
• Generally, AXR is not justified because of radiation dose. Two excep-
tions are singestion of sharp or poisonous objects, such as open safety
pins or batteries.
152
F
Foreign body. Retained forceps following a laparotomy.
Foreign body. Mercury thermometer deliberately inserted into the
bladder.
153
Radiological features of iatrogenic/inserted
F
and incidental foreign bodies
• AXR:
• Detects radioopaque foreign bodies, such as metallic materials and
some types of glass.
• Can be useful for checking the position of medical devices, for
example the ‘lost IUCD’, as well as screening postoperative patients
when a surgical item is unaccounted for.
• If an unexpected foreign body is found, always consider that it may be
within the patient’s clothes.
• Remember not all foreign bodies are radioopaque, for example
wood.
• USS: initial investigation for checking whether an IUCD is within the
endometrial cavity, thereby avoiding the radiation dose of an AXR.
• CT: useful for assessing secondary complications of foreign bodies.
154
F
Foreign body. Retained surgical swab (arrow).
Foreign body. Large rectal foreign body.
155
F
Free intra-abdominal gas
Pneumoperitoneum
Clinical characteristics
• The aetiology of pneumoperitoneum includes perforation of a hollow
viscus, through trauma, iatrogenic intervention or from inherent bowel
disease, such as a perforated ulcer. Other causes include gas entering via
the peritoneal surface, such as trans-abdominal biopsy or catheter place-
ment, and via the female genital tract.
• Gas-forming peritonitis or the rupture of a gas-filled intraperitoneal
abscess can also lead to a pneumoperitoneum.
• Postoperative gas takes a variable amount of time to disappear but if it
remains for more than 3 days consider ongoing gas leakage.
Radiological features
• Erect CXR:
• Small amounts of free gas are generally seen first in the RUQ as a
single area of radiolucency lying between the right hemidiaphragm
and the liver.
• Volumes as small as 2ml may be identified.
• Gas may also be seen under the left hemidiaphragm.
• Be aware that the patient should be in the erect position for at least
5 min before the CXR is taken to allow free gas to accumulate under
the diaphragm.
• Decubitus AXR:
• On occasion, the patient may not be able to maintain an erect
position for long enough, in which case a left lateral decubitus
AXR (left side down) can be helpful.
• In this position, free gas will lie between the lateral aspect of the liver
and the diaphragm.
156
F
Free air under the diaphragm (asterisk).
157
Free intra-abdominal air (Supine AXR). Note Rigler’s sign (arrowheads).
• AXR:
F
• The supine AXR can demonstrate free gas, although greater quanti-
ties are usually needed than with the erect CXR.
• Free gas against a soft tissue surface will outline that tissue more clearly
than usually seen on AXR: structures so outlined include the falci-
form ligament, the liver edge, the umbilical ligaments and diaphrag-
matic slips.
•
“Riggler’s sign” (the double-wall) sign represents air within the bowel
lumen outlining the inner surface, while free air outlines the outer wall.
• Plain film pitfalls include Chilaiditi’s syndrome, subdiaphragmatic fat,
adjacent gas-filled bowel loops simulating Riggler’s sign, subdiaph-
ragmatic abscess, and diverticula of the stomach or duodenum.
• CT:
• This is very sensitive for detecting free gas, but the radiation dose
involved means that plain films remain the mainstay investigation.
• On CT, areas of gas need to be carefully examined to establish
whether they represent free or bowel gas.
• May demonstrate likely site of perforation.
• USS: much less sensitive than the above techniques as differentiating
free air from bowel gas is difficult.
158
F
Pneumoperitoneum. Left lateral decubitus film. Note the presence
of free air (arrow) between the lateral margin of the liver (L) and the rib
cage.
159
Pneumoperitoneum. Free air anterior to the liver (asterisk).
F
Pneumoretroperitoneum
Clinical characteristics
• Can be caused by duodenal perforation or trauma, urinary tract gas,
pancreatic abscesses or gas tracking down from a pneumomediastinum.
Radiological features
• AXR: the presence of gas in the retropertioneum may sharply outline
the kidney and psoas muscle, and streaks of gas may outline the muscle
bundles.
160
F
Peritoneal air (Supine AXR). Extensive intraperitoneal (asterisk) and
retroperitoneal free air following ERCP. Note a biliary stent in situ (black
arrowhead). The inferior margin of the liver is outlined in the intraperitoneal
compartment (white arrowhead), with the kidneys (arrows) and psoas
muscles (curved arrow) outlined in the retroperitoneal compartment.
161
Pneumoretroperitoneum. Free air within the retroperitoneum (arrows).
G
Gallstones
Clinical characteristics
•
Gallstones are common, affecting 10-20% of the population.
•
Two main components are cholesterol and calcium bilirubinate
•
There are three types of gallstone, based on their components: choles-
terol, pigment and mixed, of which mixed is the most common type.
• Cholesterol stones are caused by precipitation of supersaturated bile
and occur in the Western population.
• Pigment stones occur from excessive haemolysis, resulting in excess
unconjugated bilirubin and hence precipitation of calcium
bilirubinate.
•
Gallstones may be present for decades without causing symptoms or
complications, and usually do not require any treatment.
•
Biliary colic is the result of a gallstone impacting within the cystic duct
during gallbladder contraction, which then eases as the gallbladder
relaxes and the stone dislodges. The pain localizes to the RUQ and
epigastrium.
•
Acute cholecystitis occurs when persistent obstruction of the cystic duct
causes gallbladder distension and inflammation. This may progress to
pus collecting within the gallbladder, known as an empyema. If the
gallbladder perforates, a pericholecystic abscess may form.
•
Chronic cholecystitis occurs in the setting of long-standing gallstones
and results in a shrunken gallbladder with loss of function.
•
Gallbladder adenocarcinoma occurs in patients with gallstones, but gallstones are
not carcinogenic per se.
Radiological features
• AXR: 30% of gallstones are calcified and 10% are visible on AXR. This
is usually an incidental finding rather than a diagnostic one, owing to the
high false-negative rate.
• USS:
• The most important modality for imaging gallstones and their
complications.
• Patients must be fasted to avoid gallbladder contraction, thereby
reducing false-negative investigations.
• Gallstones appear as echogenic foci, with posterior acoustic shadow-
ing, that move within the gallbladder when the patient is turned.
162
G
Multiple small gallstones (arrow).
163
Solitary gallstone (arrow). Note the posterior acoustic shadowing.
•
Fluoroscopy: the oral cholecystogram has largely been replaced by US
G
for the investigation of gallstones. An oral cholecystographic agent is
given and AXRs are taken the following day. Gallstones appear as filling
defects in the opacified gallbladder.
•
CT:
• Calcified gallstones have a characteristic appearance on CT but are
not as clearly visualised when non-calcified.
• Complications such as biliary obstruction and cholecystitis may also
be seen, but US remains the first-line investigation.
•
ERCP:
• ERCP is performed by physicians or surgeons rather than radiologists.
A side-viewing endoscope is used and the ampulla of Vater
cannulated.
• Stones are visualised as filling defects within the contrast-filled bile
ducts.
• Therapeutic procedures, such as sphincterotomy and removal of
stones, may also be performed.
•
MRI:
• MRCP is an alternative when it is not possible to perform ERCP.
• MRCP utilises heavily T2W sequences to highlight the slow-moving
fluid in the biliary system.
• Gallstones will appear as defects of low signal intensity within the
high-signal-intensity bile.
164
G
Gallstone within the neck of gallbladder (arrow).
165
Calcified gallstones. Axial T2 MRI shows mutlpile hypointense foci seen
within the gallbladder (arrow) indicating the stones.
• NM:
G
• Iminodiacetic acid labelled with technetium-99m is used to assess
biliary excretion and gallbladder function (HIDA scan).
• Failure to visualise the gallbladder by 4h, with normal bowel activity,
indicates cystic duct obstruction in acute cholecystitis.
• Has now largely been replaced by US.
• Interventional radiology:
• Interventional procedures include drainage of a gallbladder empyema
and PTC.
• PTC is performed to access the biliary tree percutaneously, and
involves percutaneous cannulation of the intrahepatic bile ducts via
a transhepatic approach.
166
G
Large impacted gallstone within the distal common bile duct
(arrowhead).
167
H
Hepatic masses
The differentiation of clinically significant hepatic masses from incidental,
non-significant, hepatic masses is an important aspect of abdominal imaging.
Simple hepatic cysts
Clinical characteristics
• Very common incidental finding.
• Increasing incidence with advanced age.
• Associated with tuberose sclerosis and polycystic kidney disease.
• Rarely symptomatic.
Radiological features
• USS:
• Investigation of choice to confirm presence of a simple cyst.
• Well-defined, anechoic lesion with no visible wall.
• Haemorrhage or infection may result in internal echoes.
• CT: fluid attenuation
(0-10HU) lesion with no visible wall or
enhancement
• MRI: very high signal on T2W imaging, low signal on T1W and no
enhancement are the features of a simple cyst on MRI.
168
H
Hepatic simple cyst. Typical appearance of an anechoic lesion,
demonstrating through transmission (calipers).
169
Hepatic cyst (arrow).
H
Hepatic haemangioma
Clinical characteristics
• A common, benign, liver lesion made up of vascular channels with
slowly circulating blood.
• Most are clinically silent but larger ones may spontaneously haemor-
rhage or cause pain from haemangioma thrombosis.
• Kasabach-Merritt syndrome is the association of a large haemangioma
and thrombocytopenia.
Radiological features
•
USS:
• The majority of haemangiomas are uniformly hyperechoic on US.
The majority show through enhancement.
• Some may be heterogeneous owing to scarring, fibrosis or haemorrhage.
• Often have no Doppler signal. If detectable, the velocities are <50cm s-1
•
90% are unchanged in US appearance on follow-up.
•
CT:
• Many liver lesions are investigated with a three-phase CT scan.
• This comprises a precontrast series, an arterial phase series and a portal
phase series. Occasionally delayed series are required.
• Haemangiomas are low density on precontrast imaging. On early
postcontrast, there is nodular, peripheral enhancement. Delayed
imaging shows filling in from the periphery inwards. This results in
complete ‘in-fill’ in three-quarters of haemangiomas.
• Larger haemangiomas are more likely not to fill in completely owing
to increased incidence of central scaring.
• Small (<1cm) haemangiomas may not demonstrate classical periph-
eral, nodular enhancement, being seen as a homogeneous enhancing
lesion on early phase imaging.
• The differential includes hypervascular metastases but these wash out
on delayed imaging, and are then hypodense compared with normal
hepatic parenchyma. Haemangiomas remain hyperdense on delayed
images.
170
H
Haemangioma. US shows well-defined hyperechoic lesions.
171
• MRI:
H
• MRI is increasingly used to delineate hepatic masses. It has the
advantage over CT of not incurring a radiation dose, particularly
important when compared with multiple-phase CT examinations.
• Masses are well defined, sometimes with lobulated, margins. Smaller
lesions are homogeneous but larger ones may be inhomogeneous
because of scarring.
• T1W sequences show iso- or hypointense lesions.
• On T2W sequences, haemangiomas are very bright because of the
slow-flowing blood. The hyperintensity characteristically remains
with increasing echo times.
• The post-gadolinium enhancement pattern mirrors that seen on CECT.
172
H
Haemangioma. Arterial and delayed phases of CECT. Peripheral nodular
enhancement is seen on this arterial phase scan, with central ‘in-filling’ on
the delayed scan (asterisk). (See next image.)
173
H
Haemangioma. Arterial and delayed phases of CECT. Peripheral
nodular enhancement is seen on the arterial phase scan, with central
‘in-filling’ on this delayed scan (asterisk).
174
Hepatic adenomas
H
Clinical characteristics
• Benign proliferation of hepatocytes. Well defined with a pseudo-capsule
of compressed normal hepatic tissue.
• May undergo fatty change, haemorrhage and necrosis.
• A condition seen almost exclusively in young females; occurs in men
taking anabolic steroids.
• Hormone sensitive with an increased incidence with hormone
contraception.
• Diabetes mellitus is a further risk factor.
• Growth during pregnancy may result in rupture.
• One-fifth of patients are asymptomatic.
• May present with RUQ pain from bleeding or a mass effect. Hepatomegaly
may be clinically evident.
• Biopsy carries high bleeding risk.
175
Radiological features
H
•
USS:
• Well-defined mass of variable echogenicity.
• The pseudo-capsule may be seen as a hypoechoic rim.
• In larger examples, necrosis can lead to cystic regions.
•
CT:
• On precontrast series, adenomas are usually well defined.
• Necrosis may lead to decreased density, while haemorrhage may lead
to increased attenuation.
• Adenomas have a vascular supply from the hepatic artery and, on
arterial phase CT, show brief enhancement relative to normal hepatic
tissue. Often iso- or hypodense on portal phase so may be missed if
only a portal phase postcontrast series is performed.
•
MRI:
• As adenomas are largely composed of hepatocytes, they often have a
very similar signal intensity to normal liver parenchyma on standard
T1W and T2W sequences.
• The presence of haemorrhage and intracellular fat can lead to slight
hyperintensity on T1W and T2W series.
• The presence of intracellular fat means that T1W out-of-phase
sequences will lead to a drop in signal relative to liver parenchyma.
NB: Small (<2cm) hepatomas may also occasionally demonstrate this
feature.
• As with CECT, hepatic adenomas show transient arterial enhance-
ment and subsequent homogeneous washout.
176
H
Haemangioma Axial T2W MRI shows well-defined hyperintense lesion
(arrowhead); this remained hyperintense with increasing echo times.
Liver adenoma. Arterial enhancement (note the intense enhancement
of the aorta) of a lesion (arrowhead) within the posterior aspect of the
177
right lobe of liver.
H
Focal nodular hyperplasia
Clinical characteristics
• Focal nodular hyperplasia (FNH) is a benign congenital hamartoma of
the liver.
• Consists of multiple small lobules of hepatocytes. There is often a central
fibrous scar with an arteriovenous malformation, producing a spoke-
like pattern of radiating vessels. The lesion is non-encapsulated.
• Most are <5cm in diameter. Often subcapsular in position and some are
pedunculated.
• Not caused by oral contraceptives but may grow under the hormonal
influence.
• Can be seen at any age but most commonly in the 3rd and 4th decades.
• Usually an incidental finding and are asymptomatic, although discom-
fort from a mass effect can occur.
• Unlike adenomas, FNHs rarely undergo necrosis or haemorrhage, and
intracellular fat is rarely seen.
Radiological feature
• USS:
• US may demonstrate some specific features; the mass is usually
homogeneous and either hypoechoic or isoechoic to hyperechoic
relative to hepatic tissue. The central scar will only be seen in one-fifth
of patients.
• Doppler studies may demonstrate the central arterial supply and the
spoke-like pattern of draining vessels.
• Shunt vessels have high velocity and arterial pulsatility.
178
H
Focal nodular hyperplasia. Axial scan precontrast, arterial and
delayed phase. The lesion (arrows) is slightly hypodense precontrast,
with homogeneous avid enhancement during the arterial phase scan
and rapid washout on the delayed scan.
179
•
CT:
H
• Best assessed with three-phase enhanced CT.
• On non-enhanced CT (NECT), FNHs are slightly hypoattenuating
or isoattenuating to normal liver.
• Following contrast, there is peak enhancement at 30-60 seconds with
rapid subsequent washout.
• The central scar may enhance less on the early phase but may be
hyperattenuating on delayed imaging owing to delayed washout.
•
Nuclear imaging: sulphur colloid scans involve the uptake of isotope
by the Kupffer cells of the reticular endothelial system of the liver. FNH
are the only hepatic masses to contain enough of these cells to have
normal or increased uptake on this imaging.
•
MRI:
• FNH are usually homogeneously slightly hyper- or isointense on
T2W images, homogeneously hypo- or isointense on T1W images.
• The central scar is usually hyperintense on T2W and hypointense on
T1W images.
• Following gadolinium enhancement, there is intense, homogeneous,
arterial phase enhancement within the body of the FNH. This rapidly
washes out to reach portal phase isointensity.
• The scar shows late, long-lasting enhancement.
180
H
Multiple hypoechoic liver metastases (arrowheads).
Large solid metastasis (asterisk) of mixed echogenicity. Note the
presence of ascites posterior to the liver.
181
H
Hepatic metastatic deposits
Clinical characteristics
• Metastatic deposits are the most common malignant liver masses, and
the liver is the second most common site of metastatic deposits after
local lymph nodes.
• Common sites of origin include the colon, stomach, breast, pancreas
and lung.
• Often discovered during staging of known primary tumours, but they may
present with hepatomegaly, jaundice or deranged liver function tests.
Radiological features
•
These can be very variable owing to the many possible primary tumours
of origin.
•
Mucinous adenocarcinomas, classically from colon, breast, stomach or
ovary, may calcify.
•
Metastases from melanoma, pancreatic islet cell tumours, thyroid carci-
noma, carcinoid and renal cell carcinoma may be hypervascular.
•
Metastases from stomach, pancreas, breast, lung and colon are often
hypovascular.
•
Hyperechoic metastases are seen in colonic carcinoma and hepatoma.
•
Hypoechoic secondaries are seen in cervical carcinoma, lymphoma and
pancreatic adenocarcinoma.
•
CT:
• Very reliable at detecting metastases, especially those <10mm.
• Best performed with IV contrast, although a NECT may be helpful
when hypervascular deposits are suspected, as these may be obscured
postcontrast. NECT also helps to detect the fine calcification seen in
mucinous GI secondaries - classically colonic metastases.
• Dynamic contrast enhancement usually shows a pattern of initial,
peripheral rim enhancement with a relatively hypovascular centre,
leading to a ‘target’ appearance, with delayed washout. However
many patterns of enhancement may be seen, depending on type of
metastases, including non-enhancement.
182
H
Multiple hypodense liver metastases within both lobes of
the liver.
Large hypodense colonic carcinoma metastasis within the right lobe
183
of the liver.
•
MRI:
H
•
MRI is better than CT at both detecting and characterising liver
masses.
•
This is important if resection or ablation of liver metastases is being
considered, as the presence of multiple deposits may preclude such
interventions. Conversely, MRI may establish that lesions seen on
other modalities are benign.
•
Classically, metastases are slightly hypointense in T1W and hyper-
intense on T2W.
•
Haemorrhagic metastases may have mixed T1W and T2W signal,
depending on the stage of blood product breakdown.
•
Melanin has paramagnetic properties, leading to variable hyper-and
hypointensity on both T1W and T2W images in melanoma
metastases.
•
Metastases from mucinous carcinomas, such as ovarian mucinous
cystadenocarcinoma or mucinous carcinoma of the pancreas, may
also be high on T1W images owing to their proteinaceous content.
•
Standard extracellular gadolinium enhancement patterns are similar
to those of iodinated contrast enhancement in CECT.
•
Some gadolinium- and manganese-based MRI contrast agents are
selectively taken up by hepatocytes, so increasing T1W parenchymal
hyperintensity in those hepatic neoplasia containing hepatocytes,
such as hepatic adenomas, FNHs and some well-differentiated hep-
atomas. However neoplasias not containing hepatocytes, such as
metastatic deposits and haemangiomas, do not take up such agents.
Therefore, on late (>10min) imaging, such lesions are hypointense
against the contrast uptake by hepatic parenchyma. This can be
helpful in detecting small deposits when liver resection or ablation is
being considered.
Hepatocellular carcinoma
Clinical characteristics
• Hepatocellular carcinoma (HCC) is also known as hepatoma.
• A primary malignancy of hepatocytes.
• In the developed world, this is largely associated with liver cirrhosis.
• Other risk factors include chronic viral hepatitis, carcinogens such as
aflatoxin, thorotrast, sex hormones and metabolic disorders such as
Wilson’s disease, haemachromatosis and ,1-antitrypsin deficiency.
• Much more common in Asia and sub-Saharan Africa.
• May present with RUQ pain, hepatomegaly, ascites, weight loss, fevers
and paraneoplastic symptoms.
• Often HCCs are detected on USS.
• HCCs are associated with elevated ,-fetoprotein, which is used as a
screening tool in at-risk patients.
184
H
Hepatocellular carcinoma. Large solid mass within the left lobe of liver.
Appearances in US are often non-specific; compare the appearance with the
large metastasis pictured above.
Hepatocellular carcinoma in a patient with haemochromatosis. Large
irregular enhancing mass within the left lobe of liver visible on CECT; the
central area of low attenuation (asterisk) represents necrosis. There is
associated invasion of the left portal venous branches. Note the presence of
ascites around the liver and spleen.
185
• Metastases may occur to the lungs, adrenal glands, bones and lymph
H
nodes.
• Invasion of the hepatic veins can result in a Budd-Chiari syndrome.
Tumour can track along the venous system, involving the IVC and right
atrium.
• Portal vein invasion is also a feature.
Radiological features
•
USS:
• US can be used as a screening tool in at-risk patients and as an initial
investigation in those with raised ,-fetoprotein, but it is not specific.
• HCCs may be focal, where a discrete mass can be seen. These may be
of variable echogenicity, especially in larger HCCs when haemor-
rhage and necrosis are seen.
• HCC is a vascular tumour and Doppler studies may demonstrate high
velocities.
• Invasion of venous structures can be seen.
• Diffuse HCC is poorly defined and may be difficult to detect on US,
as the subtle changes in echogenicity are difficult to differentiate
against the coarse echotexture often seen in cirrhosis.
• HCCs usually occur in cirrhotic livers where cirrhotic nodules are
common. However cirrhotic nodules are rarely seen on US and any
focal lesion should be considered highly suspicious.
•
CT:
• Detects two-thirds of HCCs in cirrhotic livers and more in non-
cirrhotic livers.
• May be focal, usually poorly defined or diffusely infiltrating.
Multifocal lesions are well recognised.
• Usually hypodense on pre-enhancement imaging; 10% may show
calcification.
• Arterial enhancement shows a heterogeneous pattern, although
lesions <1.5cm may show homogeneous enhancement.
• Necrotic, non-enhancing areas may be seen.
• On delayed portal phase scans, the tumour is usually of low attenu-
ation compared with the liver parenchyma.
• Involvement and occlusion of the portal and/or hepatic venous
systems may be demonstrated, as may areas of poorly perfused liver
secondary to venous compromise.
• Metastatic deposits, ascites and the extent of any vascular involvement
can all be assessed on CT.
186
H
Hepatocellular carcinoma. Axial T1 fat-saturated image postcontrast
from the same patient as on CT this demonstrates an irregular enhancing
necrotic mass within the left lobe (asterisk) with portal vein invasion (arrow).
Hepatocellular carcinoma pre-ablation. Portal phase T1W MR
post-gadolinium demonstrating a small mass (arrow) adjacent to the upper pole
187
of the right kidney (K). Note the irregular liver outline in keeping with cirrhosis.
•
MRI:
H
•
More sensitive than CT for the detection of HCCs.
•
The signal characteristics vary but classically the lesions are hypoin-
tense on T1W and mildly hyperintense on T2W. Hyperintensity on
T1W is a known feature, caused by the presence of intracellular fat,
and is associated with early, well-differentiated HCCs with a better
prognosis.
•
Arterial phase gadolinium enhancement is the most sensitive
sequence for hepatomas. They demonstrate diffuse heterogeneous
enhancement, although, as with CECT, small lesions may enhance
homogeneously. In contrast, metastatic deposits classically show
peripheral, rather than diffuse, early enhancement.
•
On delayed (2min post-injection) imaging, HCCs are usually hypo-
intense in comparison with hepatic tissue. There may be an enhanc-
ing psuedocapsule surrounding the HCC during the late
enhancement phase.
•
Diffusely infiltrating HCC is seen as mottled areas of T2W hyper-
intensity and early gadolinium enhancement.
•
MRI can detect metastatic deposits and assess the extent of venous
complications.
•
The presence of hepatocytes means HCCs take up liver-specific
contrast agents, unlike metastatic deposits.
•
There are a number of other mass lesions seen in cirrhotic livers.
- Regenerative nodules are not neoplastic but represent local hep-
atocyte proliferation. These tend to be <1.5cm in diameter, are
generally iso- or slightly hypointense in T1W and T2W sequences
and show no significant enhancement post-gadolinium.
- Dysplastic nodules represent adenomatous hyperplasia and are
premalignant, often developing into HCCs. Both mildly dysplastic
nodules and severely dysplastic ones are usually <1.5cm and may
be mildly hypo- or hyperintense on both T1W and T2W images.
The mildly dysplastic nodules demonstrate only minimal enhance-
ment, while severely dysplastic ones demonstrate intense, but
homogeneous enhancement.
188
Radiological interventions in hepatic masses
H
• As well as the use of cross-sectional imaging to obtain accurate tissue
samples from liver lesions, there has been an increase in radiologically
supported therapies.
• Percutaneous ablations of individual metastatic deposits and HCCs
using chemical or thermal ablation have had encouraging results com-
pared with hepatic resection.
• Ablations are performed under CT, US or MRI. One advantage of
using MRI combined with thermal ablation is that changes in the signal
of tissues, as their temperatures change, means real-time mapping of the
burn can be performed, resulting in a more accurate ablation.
• In the future, focused high-intensity US may be used to ablate hepatic
masses in a truly non-invasive manner.
Hepatocellular carcinoma post-ablation. The ablation film is a
real-time T1 image. The faint low signal line running obliquely through the
liver (arrow) is the sheath containing the laser fibre. The posteromedial area
of very low signal (asterisk) is the thermal burn in the carcinoma.
189
H
Herniae of the abdomen and pelvis
Hiatus hernia
Clinical characteristics
•
The oesphagus normally passes through the diaphragm at the level of
the 10th thoracic vertebra, and the phrenico-oesphageal membrane
fixes the oesophagus to the surrounding diaphragm.
•
When this membrane is deficient, a hiatus hernia occurs.
•
Hiatus hernia is common and the prevalence increases with age.
•
Although the majority of hiatus herniae are small, they can be very large
with the entire stomach lying in the thorax.
•
There are two types of hiatus hernia: sliding and paraoesophageal:
• Sliding is by far the most common type when the gastro-oesophageal
junction slides through the hiatus.
• In paraoesophageal herniae, the gastro-oesophageal junction is in the
normal position and the proximal stomach moves into the thorax,
where it usually lies to the left of the oesophagus in the posterior
mediastinum.
•
Sliding herniae are more likely to vary in their size and position and are
more commonly associated with gastro-oesophageal reflux.
•
Paraoesophageal herniae are more prone to incarceration and, rarely,
volvulus, and reflux is not always an association.
•
Hiatus herniae are frequently asymptomatic but associated gastro-
oesophageal reflux may cause dyspepsia.
•
Chronic gastro-oesophageal reflux can result in Barrett’s oesophagus,
which is the development of columnar epithelium in the lower oeso-
phagus; this results in a much higher risk of adenocarcinoma of the
oesophagus.
190
H
Sliding hiatus hernia (HH) with a prominent Schatzki ring (arrows).
191
Radiological features
H
•
Plain X-ray: hiatus hernia is often diagnosed on chest radiography by
the presence of a mass behind the heart, with or without an air-fluid
level dependent on the patient’s position.
•
Upper GI tract barium study:
•
This is the radiological study of choice for diagnosing a hiatus hernia.
•
A double-contrast study is performed using barium and gas to coat
and distend the oesophagus and stomach.
•
A sliding hiatus hernia is more easily diagnosed by endoscopy than
barium study as the key feature is identification of the squamocolum-
nar mucosal junction, which should normally lie at 40cm from the
incisor teeth. A sliding hiatus hernia is diagnosed if the squamoco-
lumnar junction is seen at 38cm or less (depending on patient height).
•
The mucosal junction is more difficult to visualise on barium study
and, therefore, secondary features are relied on. These features
include the Schatzki, or B, ring, which is seen as a circumferential
indentation in the distal oesophagus on the barium study and marks
the site of the squamocolumnar junction.
•
Other features are the incisural notch, which is an incomplete ring
marking the site of the mucosal junction, and the diameter of the
hiatus being larger than the distal thoracic oesophagus.
•
The less-common paraoesophageal hernia is more readily seen on
barium study as it does not depend upon the identification of the
mucosal junction. The key feature is that the gastro-oesophageal
junction lies in the abdomen.
•
When assessing for a sliding hiatus hernia, it is important to ensure
maximal distension of the distal oesphagus with the patient in the
prone position, and also to assess for associated gastro-oesophageal
reflux with the patient tilted to the right from the supine position.
•
CT: hiatus hernia is often an incidental finding on cross-sectional
imaging, but it may also provide more detailed information of the
hernial contents in a large hernia.
192
H
Sliding hiatus hernia (HH) with marked associated gastro-oesophageal
reflux (GOR).
Large hiatus hernia (arrowheads) demonstrated on CECT of the thorax.
193
H
Types of abdominal and pelvic herniae
•
Epigastric: a hernia through the fibres of the linea alba.
•
Femoral: a hernia through the femoral canal, medial and inferior to the
inguinal canal.
•
Incisional: an iatrogenic hernia, occuring in 2-10% of all abdominal
operations, secondary to breakdown of the surgical closure site from
prior surgery. Recurrence rates approach 20-45% following repair.
•
Inguinal: can be subdivided into indirect and direct. An indirect inguinal
hernia passes along the inguinal canal and the hernial sac may or may not
be limited to the inguinal canal. The neck of the sac lies lateral to the
inferior epigastric artery. A direct inguinal hernia passes directly forward
through the defect in the posterior wall of the inguinal canal, and the
neck of the hernia lies medial to the inferior epigastric artery.
•
Internal: herniation of bowel through defects in the peritoneum,
omentum, mesentery or band of adhesions.
•
Lumbar: hernia through the lumbar triangle. These tend to have a
wide neck.
•
Obturator: the hernia passes through the obturator foramen, following
the path of the obturator nerves and muscles. Because of its anatomic
position, this hernia presents more commonly as a bowel obstruction
than as a protrusion of bowel contents.
•
Peri-umbilical: herniation of abdominal contents through the peri-
umbilical tissues.
•
Richter: occurs when only the antemesenteric border of the bowel
herniates through a fascial defect, and may lead to incarceration or
strangulation of the focal herniated segment. It may occur with any of
the various abdominal herniae and is particularly dangerous, as the
strangulated bowel may be reduced spontaneously, leading to perfora-
tion and peritonitis.
•
Spigelian: this rare form of abdominal wall hernia occurs through the
semilunar line along the lateral aspect of the rectus sheath.
•
Umbilical: herniation of abdominal contents through the umbilicus.
194
H
Indirect inguinal hernia. T, testis.
Strangulated right inguinal hernia. Note the ‘stranding’ in the
195
subcutaneous fat (arrows) as a result of local ischaemia.
Clinical characteristics
H
• The clinical presentation of a hernia depends upon the site, size and
hernial sac contents. Complications include incarceration, irreducibi-
lity, obstruction and strangulation.
Radiological features
• Plain X-Ray: not usually the modality of choice for diagnosing her-
niae; however, it is of use when bowel obstruction, secondary to a
hernia, is suspected. Occasionally identified incidentally on a barium
study.
• USS: useful to localise herniae and for differentiating from other causes
of a palpable lump such as solid masses or a haematoma.
• CT and MRI: although US can provide good resolution of many
superficial herniae, CT may provide more information regarding inter-
nal herniae.
196
H
Epigastric hernia. Herniation of omental fat (arrowhead) through the
linea alba (arrow).
Large indirect left inguinal hernia containing multiple loops of small
bowel (arrow).
197
• Herniography/herniogram:
H
• This technique is less frequently used now as there are other less-
invasive modalities available.
• A spinal needle is introduced into the peritoneal cavity and radio-
graphic contrast injected.
• The patient is asked to cough and perform the valsalva manoeuvre to
demonstrate contrast in the hernial sac.
Herniogram following installation of water soluble contrast into the
peritoneal cavity. Peritoneal indentations and fossae of the anterior
abdominal wall and their relation to the sites of groin hernia.
1, median umbilical ligament (obliterated urachus); 2, medial umbilical
ligament (obliterated umbilical arteries); 3, lateral umbilical ligament
(containing inferior epigastric arteries). Sites of possible herniae: A, lateral
fossa (indirect inguinal hernia); B, medial fossa (direct inguinal hernia);
C, supravesical fossa (supravesical hernia).
198
H
Herniogram. Direct (arrowhead) and indirect (arrow) inguinal hernia.
Herniogram. Bilateral indirect inguinal hernia caused by the presence of
patent processus vaginalis bilaterally (arrows).
199
I
Intussusception
• Invagination or prolapse of a segment of intestinal tract (= intussusceptum)
into the lumen of the adjacent intestine (= intussuscipiens)
•
90% are ileocolic and ileoileocolic.
• In adults, the majority are caused by a pathological lead point.
Paediatric intussusception
Clinical characteristics
• In 90% of all paediatric intussusceptions there is no pathological lead
point and they are thought to be associated with lymphoid hyperplasia
in Peyer’s patches of the ileum.
• In the remaining 10%, the lead point is:
• Meckel’s diverticulum (most common lead point)
• polyp or other tumour
• duplication cyst
• Intussusception usually occurs in the first 2 years and rarely in neonates.
• The patient presents with severe colicky pain and vomiting. Initial stools
passed at the start of symptoms are unremarkable; blood and mucus
(‘redcurrant jelly’) stools are passed after 24h or so.
• On examination, there may be a palpable sausage-shaped mass, most
often in the upper abdomen.
200
Serosal surfaces
Peritoneal
in contact
I
transudate
Serosal surfaces
in contact
intussuscipiens (receptor)
Mucosal surfaces
returning limb of intussusceptum
in contact
mesentery
entering limb of intussusceptum
bowel lumen
Intussusception.
Radiological features
• AXR: film is normal in 50%. A soft-tissue mass or intraluminal filling
defect, in a partially air-filled bowel loop (commonly at hepatic flexure),
may be seen.
• USS: a ‘target sign’ may be seen, appearing as concentric alternating
echogenic and echo-poor rings, representing compressed mucosal and
serosal surfaces and oedematous bowel wall, respectively. Colour
Doppler shows blood vessels dragged in between the entering and
exiting layers of the intussusception. Absence of blood flow indicates
devitalised bowel segments.
• Antegrade barium study: contraindicated in perforation. Barium
examinations have been generally superseded by US. May show a
‘coiled spring’ appearance with a beak-like distal string of barium as it
runs through the central column.
• Barium enema: contraindicated in perforation. Water soluble contrast
enemas may demonstrate a convex intraluminal mass, representing the
intussusceptum, surrounded by contrast. The ‘coiled spring’ sign is seen as
contrast flows over the oedematous fold of the reflected intussusceptum.
201
Radiological management
I
• An intussusception can be reduced radiologically under fluoroscopic
guidance with barium, water-soluble contrast or air.
• Contraindications include peritonitis and shock.
• Prior to attempting reduction, the child should be resuscitated and both
surgical and anaesthetic cover available.
• The pneumatic technique is preferred as it is faster and if perforation
complicates the procedure the tears tend to be smaller, and hence the
degree of peritoneal contamination lessened.
• Radiological reduction can also be attempted under US guidance using
saline.
• The complication rate for radiological reduction is less than 3% and
includes perforation, the reduction of infarcted bowel and the non-
detection of a pathological lead point.
202
I
Intussusception. Soft tissue density mass within the upper abdomen
(arrowheads).
203
Intussusception. Classic US appearance of the ‘target sign’ in intussusception.
I
Adult intussusception
Clinical characteristrics
• Unlike in the paediatric situation, the majority, approximately 80%,
have a defined lead point such as a neoplasia, inverted diverticulum,
foreign body, chronic ulcers or postsurgical changes.
• Intussusceptions are the cause of approximately 15% of bowel obstructions.
• Other presentations include bloody diarrhoea, recurrent colicky abdo-
minal pain, palpable mass and change in bowel habit.
Radiological features
• CT:
• This is the imaging modality of choice. The intussusception is iden-
tified by a series of concentric rings. The central component repre-
sents the lumen and the intussusceptum wall. The middle ring is of
mesenteric fat in which mesenteric vessels are usually visible. The
peripheral ring is made up of the reflected intussusceptum and the
intussuscipiens.
• May also demonstrate the pathological lead point and its extent.
• Complications such as obstruction or perforation are also well
visualised.
204
I
Intussusception. Single-contrast enema shows ‘coiled spring’ appearance
(arrow) in the RUQ.
Ileocaecal intussusception. A further example that is secondary to a
carcinoma (asterisk).
205
• Barium studies and USS: no longer first-line investigation, but
I
chronic, intermittent intussusceptions may be uncovered during these
investigations. The findings are similar to those in children; however,
evidence of lead points is again more likely to be seen.
Management
In the adult group, management is surgical rather than radiological.
206
I
Adult intussusception. Axial CECT with coronal reformat. Large
207
intussuscepting carcinoma within the descending colon (arrows).
L
Lines and devices
• It is important to be able to recognise common lines and devices used in
the abdomen in order to confirm they are correctly positioned and to
avoid misinterpretation.
Radiological features
• Most lines and devices are visible on AXR; however, as this is only a
two-dimensional assessment, further imaging is sometimes necessary to
assess the position and patency of various lines and tubes.
• US can confirm the patency of ureteric and biliary stents by document-
ing the absence of hydronephrosis and biliary dilatation.
• US and/or CT may be needed to assess surgical drains.
• If there is concern regarding patency of a vascular line, radiographic
contrast can be directly injected into it under fluoroscopic guidance.
208
L
Endovascular thoraco-abdominal aortic stent seen in situ (arrows).
209
Ureteric stent. Right double ‘pigtail’ stent.
Types of line/device
L
Vascular lines
• In the neonate, umbilical arterial and venous lines are commonly used.
• The umbilical artery catheter is passed via the umbilical artery into
the internal iliac artery and the infrarenal aorta. On AXR, it is seen to
loop down into the pelvis before travelling in a cephalic direction to
the left of the midline. The tip of the line should lie either above or
below the renal arteries, between T8 and T12 or L3 and L4.
• The umbilical vein catheter should traverse the umbilical vein, the
portal sinus (junction of left and right portal vein), ductus venosus and
IVC and end in the right atrium. On the abdominal film the catheter
passes in a cephalic direction from its entry point and should lie above
the liver in the right atrium. The umbilical vein catheter is easily
misplaced in a portal or hepatic branch and the tip is then seen
projected over the liver on AXR.
• Femoral venous lines lie in the inguinal regions and pass in the iliac
veins to the IVC.
• Arterial stents within the abdominal aorta and iliac arteries have a
mesh-like appearance.
• IVC filters look like the spokes of an umbrella and lie to the right of the
midline on the abdominal film.
210
L
Umbilical lines. The asterisks indicate cardiac monitoring leads. NGT,
nasogastric tube; UVC, umbilical venous catheter; UAC, umbilical arterial
catheter.
211
Percutaneous endoscopic gastrostomy. IVC filter (arrowhead) and
tube (arrow) in situ.
Urinary tract lines
L
• The most commonly seen tube relating to the urinary tract on AXR is
the urinary catheter, placed either via the urethra or the suprapubic
route.
• Ureteric stents have curled ends and lie lateral to the transverse
processes of the vertebral bodies. Transplant kidneys may also have
ureteric stents that lie in the pelvis.
• Nephrostomy tubes placed percutaneously to drain an obstructed
kidney are seen in the upper abdomen, with a coiled end in the renal
angle and an external component.
• Patients with renal failure on peritoneal dialysis have an intra-
abdominal catheter that is tunnelled under the skin before entering
the abdomen and usually lies in the lower abdomen, although its
position is variable.
212
L
Cholecystectomy clips in the RUQ (arrowheads).
213
Gynaecological lines/devices
L
• Commonly identified devices include:
• intrauterine contraceptive devices (‘the coil’)
• sterilisation clips
• ring pessary: used as conservative treatment for uterine prolapse.
214
L
Intrauterine contraceptive device and sterilisation clips. The clip on
the right side has migrated superiorly.
Ring pessary in situ (arrow).
215
Gastrointestinal tract lines
L
• Tubes commonly seen within the GI tract are nasogastric and naso-
jejunal tubes placed for feeding. To aid identification, the tips of the
tubes are radioopaque and the tips of both should lie in the LUQ. The
nasojejunal tube lies more distally within the bowel and follows the
C-shaped loop of the duodenum, crossing the midline into the left side
of the abdomen, to lie just distal to the duodenojejunal junction.
• Stents placed within the biliary system, for example within the com-
mon bile duct, are visible on the abdominal film, lying in the RUQ.
• Stoma devices. The position of these is naturally dependent on the
procedure performed.
216
L
Nasojejunal tube in situ.
Stoma device seen in the LIF (arrows).
217
Surgical drains
L
Drainage tubes are often present in postoperative patients; the site obvi-
ously relating to the procedure performed, and hence clinical history is
important when looking at the AXR.
218
L
Postoperative abdomen. NG, nasogastric tube; D, surgical drain;
S, surgical staples
Expandable biliary stent (arrowhead) and percutaneous pigtail
219
drainage catheter (arrow) seen in situ within the RUQ.
L
Lymphadenopathy
Clinical characteristics
• Lymphadenopathy can have numerous causes, which can be broadly
divided into neoplastic, infective and inflammatory conditions:
• neoplastic: lymphoma, leukaemia, metastatic cancer.
• infective: TB, bacterial, viral, fungal.
• inflammatory and miscellaneous: Castleman’s disease, amyloid,
sarcoid, mesenteric lymphadenitis.
Radiological features
•
As part of the investigation of a patient with lymphadenopathy, which is
apparent elsewhere, abdominal imaging may be performed.
•
The main purpose is to assess the size and extent of the lymph node
enlargement (i.e. regional or widespread) as well as to identify an
underlying cause.
•
Imaging also plays an important role in the follow-up of treated patients.
Generally the smaller diameter (short axis diameter) of the node in the
axial plane is recorded and regional nodal groups have different upper
limits of normality. As a rule of thumb, a short axis diameter of <1cm is
considered within normal limits, although there is overlap between
benign and malignant adenopathy. However, using size as a criterion
of malignancy may overlook microscopic metastases when only part of a
node is involved. If a lower threshold is used to separate benign and
malignant, the sensitivity increases but the specificity decreases, and
when a higher threshold is applied the sensitivity drops and the specif-
icity rises.
•
AXR: may detect calcification within lymph nodes but overall is not
helpful.
•
USS:
• As well as confirming that a palpable swelling is caused by lymph node
enlargement, US can characterise and assess the extent of lymphaden-
opathy, although visualisation of the retroperitoneum can sometimes
be obscured by overlying bowel gas.
• Normal lymph nodes are usually oval and hypoechoic (dark) with
hilar vascularity. They often have an echogenic fatty hilum.
• Malignant disease alters the morphology of nodes so that they become
more rounded with peripheral vascularity.
• US is also useful for performing US-guided fine needle aspiration
cytology or biopsy.
220
L
Lymphoma. Multiple large-volume mesenteric lymph nodes (arrows),
with additional confluent retroperitoneal nodal masses (asterisk).
Necrotic ring-enhancing node secondary to a malignant gastric ulcer
(not shown). Extensive soft tissue stranding present in the left upper
quadrant from local tumour infiltration (asterisk).
• CT:
• CT is particularly useful in assessing the extent of lymphadenopathy,
identifying an underlying cause and any complications, for example
hydronephrosis secondary to enlarged lymph nodes.
• It is also useful as a method of following up patients known to have
lymphadenopathy, after treatment.
221
• IV contrast is routinely given to help to differentiate lymph nodes
L
from adjacent vessels and oral contrast or water to outline small-
bowel loops.
• As on US, benign nodes may also demonstrate a fatty hilum and
characteristic oval shape.
• Enlarged lymph nodes can be characterised further by their appear-
ance. For example, tuberculous nodes typically have a low-density
necrotic centre and rim enhancement after IV contrast.
• Squamous cell metastases are also typically necrotic. In contrast,
lymphomatous nodes have a homogeneous appearance with minimal
enhancement.
• The rare condition, Castleman’s disease demonstrates marked lymph
node enhancement.
•
MRI:
• Both normal and malignant lymph nodes have intermediate signal
intensity on T1W sequences, intermediate-high on T2W sequences
and enhance after IV gadolinium.
• The same size criteria as for CT are used with the same problems.
• There have been clinical trials using specific contrast agents for
lymphoid tissue, which demonstrate improved accuracy, but these
agents are not in widespread use.
•
Positron emission tomography (PET) and PET-CT:
• Both PET and PET-CT use the glucose analogue
18F-
Fluorodeoxyglucose (FDG), which is taken up by cells with an increased
rate of glycolysis, as in malignant lymph nodes.
• The disadvantage of PET is its lack of spatial resolution owing to
limited anatomical information.
• This has been improved by the combination of CT with PET to
create PET-CT. The patient is scanned sequentially by the PET and
CT scanners from skull base to mid-thigh and the images recon-
structed and coregistered for interpretation by the radiologist.
• PET-CT can now provide accurate anatomical information about a
region of increased uptake of isotope in the PET component.
• It is important to be aware that other conditions, such as infection,
inflammation and granulomatous disease, can also demonstrate
increased glycolysis. However, PET-CT has an important role in
the diagnosis and staging of malignant disease.
222
L
Nodal tuberculosis. Multiple abdominopelvic ring-enhancing necrotic
(caseous) nodes (arrows).
223
L
Lymphoma: abdominal
Clinical characteristics
•
Lymphomas are malignancies of the lymphoid tissues of the body and
are broadly divided into Hodgkin’s disease (HD) and non-Hodgkin
lymphoma (NHL). They are a diverse group of neoplasms with some
broad patterns of presentation and behaviour.
•
Lymphoma accounts for 5-6% of all adult and 10% of childhood
malignancies. NHL is more common than HD (60:40%); in the UK
in 2001, there were 4600 deaths from NHL and 260 deaths from HD.
•
HD classically shows a bimodal peak distribution: in the third decade
and subsequently in the 65-75 age group. NHL is a disease of children
and the elderly, with a median age at diagnosis of 65 years.
•
The different behaviour of HD and NHL is reflected in their clinical
staging (which is distinct from their pathological staging). While both
HD and NHL are essentially diseases of lymph nodes, both may present
as either localised nodal disease or as widely disseminated cancer.
•
Most patients with HD present with painless asymmetrical nodal
enlargement, accompanied by night sweats, fever, pruritus and weight
loss in 40% (B symptoms).
• The commonest site of nodal involvement is cervical, present in up to
80% of patients. Exclusive infradiaphragmatic disease occurs in under
10% of patients. Splenomegaly is present in about a third. HD spreads
contiguously from one nodal group to the next. Primary extra-nodal
HD is very rare.
•
Patients with NHL also present with nodal enlargement, though in this
case it is often non-contiguous and extra-nodal disease is common.
Associated B symptoms occur half as often as with HD.
•
HD disease is staged via the Cotswold classification, a modification of
the Ann Arbor classification. Early disease is treated with radiotherapy,
while more advanced disease is treated with radiotherapy and chemo-
therapy. Overall 10-year survival in patients treated with radiotherapy
for early-stage HD is >90%.
•
The prognosis in NHL is very variable. Unlike HD, histological subtype
(particularly the division into low- and high-grade disease) as well as
stage is the major determinant of treatment. Low-grade lymphoma is
often asymptomatic and surveillance or local radiotherapy may be
appropriate. In high-grade lymphoma, multidrug chemotherapy and
new antibody therapies are utilised. The 10-year survival for low-grade
NHL treated with radiotherapy approaches 90%.
224
L
Lymphomatous deposit within the liver (arrow) and several
enlarged retroperitoneal nodes (arrowheads).
Splenic lymphoma. Multiple hypodense lymphomatous deposits within
225
the spleen.
Radiological features
L
• Abdominal lymphoma may involve both nodal and extra-nodal sites.
Retroperitoneal nodal involvement is present in up to 35% of patients
with HD and up to 55% in NHL. Mesenteric nodes are involved in >50%
of patients with NHL but only 5% in HD. In HD, coeliac, splenic hilar and
porta hepatis nodes are involved in 30% of patients. Involvement of
splenic nodes is invariably associated with splenic infiltration. There
may be contiguous spread from mediastinal nodes in HD. In NHL,
nodal involvement is often non-contiguous and may be associated with
visceral involvement as well as diffuse mesenteric and omental involve-
ment. In the pelvis, all nodal groups may be involved in both NHL and
HD. Involved nodes are typically rounded, homogeneously enhancing
and may form a matted nodal group particularly in NHL.
• Extra-nodal abdominal involvement in both HD and NHL may
include the liver, spleen, GI tract, pancreas, genitourinary system and
adrenal glands.
Liver
• Liver and invariably splenic involvement occurs in 5% of patients with
HD, with 15% of patients with NHL similarly affected. Large focal areas
of involvement are easily seen on CT, MRI and US as well-defined,
large hypodense (on both NECT and CECT) or hypoechoic areas, but
are present in only 10% of hepatic involvement. On MRI, these areas
are low on T1W and high on T2W images. Imaging is insensitive to the
more common diffuse microscopic involvement seen in both HD and
NHL. Diffuse hepatomegaly is strongly suggestive of liver involvement.
Spleen
• The spleen is involved in 30-40% of patients with HD, usually with
coexistent supradiaphragmatic disease. In 10%, it may be the only
infradiaphragmatic site of disease.
• Splenomegaly, however, is an insensitive indicator of splenic involve-
ment, with 33% of normal-sized spleens found to contain tumour at
laparotomy. Splenic lymphoma may present as a solitary lesion, miliary
splenic nodules or multiple low-attenuation masses on CECT and US.
Optimum CECT scanning of the spleen is in the porto-venous phase of
contrast enhancement. US assessment of splenic involvement is insen-
sitive. Splenic involvement is seen in 40% of patients with NHL.
• Splenomegaly and splenic infarction are more common in NHL than HD.
226
L
Gastric MALToma (arrow). Diffuse gastric wall thickening.
Burkitt’s lymphoma. Axial section showing diffuse small-bowel wall
227
thickening (arrows).
Gastrointestinal tract
L
•
This is the commonest site of primary extra-nodal lymphoma in NHL.
•
Involvement in HD is very rare.
•
Primary lymphoma usually only affects one site. It develops from
mucosa-associated lymphoid tissue (MALT tumours) and has a bimodal
distribution of under 10 years and between 50 and 60 years.
•
Secondary lymphoma of the GI tract is common, with stomach
involvement being the most common (50%), followed by small bowel
(33%), large bowel (15%) and oesophagus (1%).
•
Almost 50% of patients demonstrate multiorgan abdominal disease.
•
Primary lymphoma of the stomach arises in MALT. It may present as
multiple nodules with central ulceration, a large fungating solitary lesion
or, in 10%, diffuse gastric fold thickening. Diagnosis is best made by
endoscopy but cross-sectional imaging is needed to assess the deep tissue
extent, extra-gastric nodal involvement and multiorgan disease.
•
Small bowel:
• Lymphoma accounts for 50% of all small-bowel tumours and usually
affects the terminal ileum.
• Patients with AIDS are particularly prone to disease in this region.
Multifocal disease is present in up to half.
• As the lymphoma develops and destroys the layers of the bowel,
involved areas initially produce mural thickening, leading to subse-
quent wall destruction, with segments of alternating constriction and
dilatation.
• Less commonly, involvement remains submucosal, leading to polyp
formation and intussusception. Indeed, lymphoma is the commonest
cause of intussusception in children after infancy.
• T-cell lymphoma associated with gluten-sensitive enteropathy and
,-chain disease affects the proximal small bowel and may lead to
bowel perforation.
• The small intestine may also be affected secondary to direct extension
from mesenteric adenopathy, leading to encasement and displace-
ment of affected bowel loops, peritoneal enhancement and occasion-
ally ascites in advanced disease, which may be indistinguishable from
peritoneal carcinomatosis.
•
Colon: primary lymphoma of the large bowel is rare (0.05% of all
colonic neoplasms) and usually affects the rectum and caecum, together
with terminal ileum, and may present as a large mass indistinguishable
from colonic carcinoma. Secondary involvement is more diffuse or
segmental.
•
Oesophagus: involvement is very unusual and when present involves
the distal oesophagus resulting in a typically smooth tapered
oesophagus.
228
(A)
(B)
L
Renal lymphoma before (A) and after (B) treatment. Large hypodense
masses expanding the right kidney prior to treatment, with significant
reduction in volume of the lesions post-treatment.
Pancreas
• Primary pancreatic lymphoma is responsible for just over 1% of pan-
creatic neoplasms and is more common in NHL than HD.
• A large pancreatic mass, with only mild pancreatic duct and common
bile duct obstruction, associated with retroperitoneal nodal involve-
ment is highly suspicious of lymphoma.
Genitourinary tract
• Although rarely involved in early disease, involvement rises to 50% in
advanced lymphoma.
• The testicle is most commonly involved, followed by kidney and
perirenal space. Involvement of the prostate, bladder, uterus, vagina
and ovaries is documented but rare.
• Testicular lymphoma is the commonest form of testicular neoplasm in
patients over 60 years.
• It is found in 1% of men with NHL, almost never in HD. Many cases are
clinically silent and the testis is an important site of residual disease
following treatment at other sites (similar to CNS disease). US demon-
strates non-specific findings of focal areas of hypoechogenicity or diffuse
decreased testicular echogenicity. It is bilateral in over 20% of patients.
229
•
Renal involvement is a relatively late event, with disease remaining
L
clinically silent in many patients.
•
Renal function usually remains normal and in 90% is caused by high-
grade NHL.
•
Involvement usually produces multiple intrarenal masses (60%). On
CT, these show typical density reversal patterns on both NECT and
CECT. In 10-20%, a solitary mass, indistinguishable from carcinoma, is
noted.
•
Renal involvement occurs in the absence of retroperitoneal adenopathy
in over half of those affected. Direct infiltration of the kidneys from the
retroperitoneum via the renal hilum is the second commonest mode of
involvement.
•
A soft tissue mass in the perirenal space is also commonly seen, encasing
the kidney without apparently invading it.
•
Diffuse renal involvement and subsequent enlargement and patchy
non-enhancement are less commonly seen.
•
Secondary involvement of the bladder is found in 10-15% of patients:
on CT and MRI this appears either as diffuse widespread bladder wall
thickening or as a large nodular mass. Both of these are indistinguishable
from transitional cell carcinoma and result in haematuria.
•
Primary bladder lymphoma is a MALT type and is more common in
women with a history of cystitis, presenting as solitary or multiple sessile
masses.
•
Involvement of the prostate is usually diffuse, with direct extension
from pelvic side wall nodes, in advanced disease, being the most common
form.
•
Female genital tract involvement in advanced lymphoma is common,
though primary involvement is rare (1% of extra-nodal NHL).
•
Most cases involve postmenopausal women, presenting with postme-
nopausal bleeding. Lymphoma may affect the cervix or uterine body,
where it produces diffuse enlargement with homogeneous signal return
or enhancement. Characteristically, the uterine mucosa is uninvolved
and the low-signal-intensity junctional zone, seen on MRI, remains
intact.
•
Ovarian lymphoma is rare but carries a poor prognosis because of the
advanced stage of disease at this point. Lesions are characteristically
bilateral and show homogeneous enhancement without haemorrhage,
necrosis or calcification.
Adrenal glands
• Primary involvement is rare; however in diffuse lymphoma, secondary
involvement occurs in 4% as assessed on CT, and in 24% of postmortem
examinations.
• Occurs more commonly with NHL than HD.
230
L
Bilateral adrenal lymphoma in a patient with HIV. The coronal T2W
MRI demonstrates large, heterogeneous, faintly hyperintense adrenal
masses (arrows). Following the administration of gadolinium (Post Gad),
only minimal enhancement is demonstrated (arrowhead).
• Bilateral involvement is seen in 50% of patients.
• Usually characterized as an area of low signal intensity on T1W images
and as an area of heterogeneous high signal intensity on T2W images,
with minimal progressive enhancement after contrast.
New developments
• The recent introduction of18F-FDG PET-CT has contributed signifi-
cantly to the detection and assessment of lymphoma.
• Cross-sectional imaging (CT, MRI and US) depends on the detection
of abnormalities in size, appearance and enhancement for the diagnosis
of organ involvement in lymphoma.
• Lymphomatous involvement often does not lead to anatomical changes
on imaging.
• This is particularly important in the assessment of post-treatment poten-
tial residual/recurrent disease. Functional imaging with18F-FDG PET-
CT enables a semiquantitative assessment of functionally abnormal
tissue and has become increasingly central to imaging in lymphoma.
231
O
Omental secondary deposits
Clinical characteristics
• The omentum is a fold of visceral peritoneum related to the stomach
and transverse colon, containing a large amount of fat and a rich vascular
network.
• Secondary neoplasms of the omentum are more common than primary
neoplasms. Ovarian carcinoma is the most common primary neoplasm
metastasising to omentum.
• Spread is either direct along the transverse mesocolon, the gastrosplenic
or gastrocolic ligaments or via peritoneal or haematogenous spread.
• The patient may be asymptomatic but there may be indicators of
progression of their underlying malignancy, with rising levels of tumour
markers.
Radiological features
• Omental deposits are visible on both US and CT. They may be detected
earlier on CT as irregular soft tissue density in the omental fat.
• In more advanced disease, the deposits range from discrete nodules to
confluent solid masses known as omental cakes. These may enhance.
Additional peritoneal deposits may be seen.
• MRI using gadolinium enhancement with fat suppression is also sensi-
tive for omental and peritoneal secondaries; the deposits show as
enhancing soft tissue lesions against the background of the dark sup-
pressed fat.
232
O
Multiple peritoneal nodules (arrowheads) in a patient with ovarian
carcinoma.
Extensive omental cake (arrowheads) in a patient with ovarian
233
carcinoma.
P
Pancreatic carcinoma
Clinical characteristics
•
Subtypes include carcinoma arising from ductal elements (ductal carci-
noma), carcinoma arising from the glandular elements (pancreatic acinar
carcinoma), tumours arising from the endocrine component of the
pancreas (islet cell cancer, glucagonoma, gastrinoma, VIPoma, soma-
tostatinoma and non-functioning islet cell tumour). Metastases to the
pancreas occur but are uncommon.
•
This discussion will focus on pancreatic ductal carcinoma.
•
Ductal carcinoma accounts for 80-90% of non-endocrine pancreatic
cancer and is the fifth leading cause of cancer death in Western countries.
•
There is increased risk with alcohol abuse, diabetes, heredity and
African ancestry.
•
A number of oncogene mutations have been identified including p53
and k-ras mutations, as well as overexpression of growth factors such as
epidermal growth factor and transforming growth factor.
•
There is an increasing incidence with age, ductal carcinoma being most
common in the 7th decade.
•
Over 50% of lesions are located in the head, 25% in the body and
approximately 10% in the tail.
•
Spread is through the pancreas into adjacent structures, with local
nodal and subsequent metastases to liver (30%), lungs, pleura and
bone. Peritoneal carcinomatosis with ascites occurs in up to 10% of
patients.
•
Local extension is predominantly posterior, involving visceral vessels
and the retroperitoneum. Extension anteriorly or laterally into the
splenic hilum also occurs.
•
Local extension may result in a tumour mass involving pancreas, duo-
denum, porta hepatis, spleen, stomach, root of small-bowel mesentery
and particularly the left adrenal gland.
•
In 85% of patients presenting with ductal adenocarcinoma, there will be
extra-pancreatic extension at the time of presentation and the carci-
noma is incurable.
•
At presentation, 40% have lymph node metastases, 50% have hepatic
metastases and 35% have peritoneal implants.
•
Median survival is 12-18 months. It has the worst 5-year survival of
more than 60 human malignancies.
•
Clinical features depend on the extent of disease and the site of origin.
•
In general, tumours in the head produce earlier symptoms than more
distal tumours, because of early involvement of the common bile duct as
it passes through the head of the pancreas to empty into the second part
234
of the duodenum.
P
Pancreatic carcinoma. Ill-defined hypoechoic mass within the head of
pancreas (arrow) on US. B, body of pancreas; SV, splenic vein.
Pancreatic carcinoma. Obstruction of the pancreatic duct owing to a
mass within the head of pancreas (arrow) shown on ERCP.
• Signs of head of pancreas lesions include obstructive jaundice.
Pancreatic carcinoma is the commonest cause of malignant biliary
obstruction (75%).
• In less central lesions, or in advanced local disease, symptoms include
severe central back pain (involvement of retroperitoneal nerves), portal
235
thrombophlebitis and thrombosis (direct extension into vein or para-
P
neoplastic syndrome ), diabetes (destruction of islet cells by tumour) and
fatigue, malaise and weight loss (pancreatic insufficiency and paraneo-
plastic disease).
Radiological features
•
The goals of imaging include accurate preoperative staging to identify
the small proportion of patients where curative pancreaticoduodenec-
tomy may be considered, and to diagnose mimics of ductal adenocarci-
noma that have a significantly better prognosis.
•
In patients with obvious extra-pancreatic extension (where curative
surgery is not possible), CT- or US-guided fine needle aspiration is
standard procedure to confirm imaging diagnosis. In small tumours,
endoscopic US-guided fine needle aspiration can be used to improve
diagnostic yield.
•
In general, MRI and CECT are the most widely used modalities for
imaging and are roughly equivalent in their accuracy for tumour iden-
tification, local staging and identification of distant metastases.
•
The role of18F-FDG PET is evolving and it may become more useful in
the differentiation of benign and malignant lesions, and the identifica-
tion of distant metastases.
•
Pancreatic ductal carcinomas are relatively avascular compared with
normal pancreatic tissue, while islet cell tumours are hypervascular.
These features are utilised by subsequent imaging techniques.
•
Patient preparation for CECT includes distension of stomach and
duodenum with 1000ml of water orally. Initial NECT of target area
is followed by a late arterial phase scan. This arterial phase improves
visualisation of ductal carcinoma through its relative hypoattenuation, a
result of its relatively hypovascular nature, compared with the normal
pancreatic tissue.
•
This phase also enhances visualisation of small hepatic metastases by
showing ring enhancement.
•
Islet cell tumours, which are hypervascular, readily show up against
pancreatic tissue during arterial phase.
•
Venous phase scanning at 70s serves to confirm liver metastases, portal
venous occlusion and venous collaterals in portal obstruction.
•
Curved planar reconstructions through the pancreas can demonstrate
the pancreatic duct, common bile duct and peripancreatic vasculature.
Of particular importance is involvement of the superior mesenteric
vasculature.
•
Arterial encasement by tumour is an absolute contraindication to sur-
gical resection.
•
Obliteration of fat planes around
50% of the circumference of a
vessel on both MRI and CECT is reliable in predicting vascular
236
involvement.
P
Carcinoma of the head of pancreas. Ill-defined mass replacing the
head of pancreas (arrow) seen on CECT, with extension into the adjacent
body and invasion of the second part of duodenum.
Pancreatic mucinous cystadenocarcinoma. Large, faintly septated,
cystic neoplasm (asterisk) effacing the body and tail of pancreas.
• Involvement of the portal venous system is controversial, with some
surgeons still opting for attempted curative surgery.
• Most tumours (except uncinate process tumours) lead to eventual
involvement and obstruction of the common bile duct. Tumours in
the head and uncinate process typically infiltrate along the superior
mesenteric artery and root of small-bowel mesentery, while tumours
within the tail extend into splenic hilar structures and coeliac and
hepatic vessels.
• Direct extension into bowel wall is often visualised as an infiltrating
hypovascular mass involving the strongly enhancing bowel wall.
237
•
Assessment of nodal involvement is difficult. Pancreatic carcinoma
P
drains to peripancreatic nodes, then to nodes in the porta hepatis and
root of small-bowel mesentery, and finally to peri-aortic and distal
superior mesenteric nodes. Nodes >1cm are considered involved.
However, there is a high propensity for micrometastases in pancreatic
carcinoma, with no subsequent nodal enlargement. In addition, reactive
adenopathy following coexistent pancreatitis or instrumentation
(ERCP) is common, and nodal evaluation by CT is poor.
•
Metastases to liver and peritoneal surfaces are the most frequently
observed intra-abdominal sites. Small hepatic metastases are detected
during late arterial enhancement by ring enhancement. Peritoneal
thickening, nodules and ascites are indications of peritoneal
involvement.
•
Fast MR techniques, such as echo planar imaging and three-dimensional
volumetric breath holding, have established MR as a CT equivalent tool
for initial diagnosis and staging. The most useful sequence is contrast-
enhanced T1W breath-hold gradient echo sequences. MRCP is useful
for visualising the pancreatic duct or common bile duct obstruction or
involvement.
•
Pancreatic ductal carcinoma generally has a lower signal intensity than
normal pancreatic tissue, particularly with fat-suppression techniques.
T2W images are utilised for detection of hepatic metastases, showing
little contrast difference within the pancreas itself. Fast spin-echo tech-
niques are utilised for pancreatic visualisation owing to their rapid
acquisition and gadolinium-enhanced T1W echo gradient sequences
are used to map vascular invasion.
•
Endoscopic US plays a useful role in local staging when there is no
CECT or MRI evidence of distant metastases. Carcinoma is typically
hypoechoic on US and ductal obstruction distal to the lesion is readily
visualised on endoscopic US. Where tumour is unresectable, a defini-
tive tissue diagnosis may be obtained by endoscopic US-guided fine
needle aspiration.
•
Use of18F-FDG PET is sensitive and specific for pancreatic malignancy,
with sensitivity of 88% and specificity of 83%. It is useful in differ-
entiating benign from malignant lesions, confirming possible liver and
peritoneal metastases as identified on CECT or MRI, and is useful in
the follow-up of patients in the context of a rising carbohydrate
antigen 19-9 (CA19-9) serology.
•
Upper GI tract contrast studies are currently less utilised for diagnosis
and generally are performed in the context of postsurgical or palliative
management. However, on occasion, they may be the first indicator of
pancreatic malignancy, which may be demonstrated as an irregular or
smooth nodular mass, in the case of an ampullary lesion, or spiculation
and traction of the duodenum, in the case of local extension from a head
or body cancer. Extrinsic indentation of the ampulla of the duodenum
may be seen.
238
P
Carcinoma of the head of pancreas. Coronal reformat CECT shows
an ill-defined hypodense mass within the head of pancreas (arrow) with a
fairly well-defined hypodense metastasis within the right lobe of liver
(arrowhead).
239
P
Pancreatitis
Clinical characteristics
• Classified as acute and chronic.
• Alcohol and cholelithiasis account for 60-70% of cases.
• Cholelithiasis responsible for 75% of cases of acute pancreatitis.
• Alcohol responsible for 70% of chronic pancreatitis cases.
• In
20-30% less common causes are involved, including metabolic
disorders (often with hypercalcaemia and/or hyperlipidaemia as com-
mon factors), multiple myeloma, amyloidosis and sarcoidosis.
• Hereditary pancreatitis is an autosomal dominant disorder producing
early-onset recurrent pancreatitis, with 20-40% developing pancreatic
carcinoma.
• Other causes include trauma, surgery, viral infections, such as measles
and mononucleosis, parasites, structural abnormalities, such as pancreas
divisum, and drugs such as steroids and thiazide diuretics.
• Up to 20% of cases are idiopathic.
• Complications include:
• pancreatic necrosis.
• acute peripancreatic fluid collections (phlegmon).
• pseudocyst formation.
• pancreatic abscess.
Clinical presentation
• Mild acute pancreatitis presents with epigastric abdominal pain, often
radiating through to the back, associated with nausea, vomiting and
general malaise.
• Occasionally progresses to acute severe pancreatitis, with profound
systemic upset including shock, adult respiratory distress syndrome
(ARDS) and multiorgan failure, flank ecchymosis (Grey-Turner sign)
and peri-umbilical ecchymosis (Cullen sign).
• Chronic pancreatitis presents with chronic intermittent abdominal pain
and symptoms and signs of pancreatic insufficiency, including malab-
sorption, diabetes mellitus and occasionally obstructive jaundice.
• Patients may also present with complications of acute pancreatitis, such
as jaundice from oedema of the common bile duct or calculi, pain and
upper GI obstructive symptoms (from pseudocyst formation) and dia-
betes from islet cell destruction.
240
P
Pancreatitis. Sentinel loops in the left upper quadrant (asterisks)
secondary to pancreatitis.
Acute pancreatitis
Radiological features
• AXR:
•
‘Sentinel loop’ - localised segment of gas-filled small bowel caused by
peritonism - is typically seen in the upper abdomen.
•
‘Colon cutoff sign’: dilated transverse colon with abrupt change to a
gasless descending colon, caused by local inflammation resulting in
spasm of the splenic flexure.
• Mottled appearance of pancreatic region owing to fat necrosis.
• Gas-less abdomen: multiple loops of fluid-filled bowel.
• Ascites.
• Intrapancreatic gas bubbles caused by necrosis and abscess formation.
• Gallstones may be identified if calcified.
• None of these findings is specific and usually CT and/or US are more
helpful.
• CXR:
• Left-sided pleural effusion.
• Left-sided hemidiaphragmatic elevation.
• Left-sided basal atelectasis.
241
• Pulmonary oedema or infiltrates in ARDS.
P
• Occasionally a pericardial effusion complicates pancreatitis.
• None of these findings are specific.
•
USS:
• Hypoechoic enlarged gland, caused by diffuse oedema.
• The pancreatic duct may be dilated (not normally seen in the normal
gland).
• Complications of acute pancreatitis (vide infra).
•
Barium meal and follow through:
• Enlarged tortuous rugal folds.
• Widening of retrogastric space (from pancreatic enlargement/inflam-
mation in lesser sac).
• Diminished duodenal peristalsis from surrounding inflammation.
• Oedematous swelling of papilla of Vater: Poppel sign.
• These studies have been largely superseded by CT and US.
•
CECT:
• Normal appearance of pancreas in nearly 30%.
• Enlarged and indistinct gland as a result of oedema.
• Thickening of anterior pararenal fascia.
• Hyperdense intrapancreatic portion can represent haemorrhagic
pancreatitis.
• Hypodense intrapancreatic portion may be seen, representing
phlegmonous pancreatitis.
• Non-enhancing areas indicate necrotic pancreatitis.
•
MRI:
• Inflammatory change in the peripancreatic fat is well demonstrated
on T2W fat-saturated images. Areas of non-enhancement indicate
necrosis.
• MRI is generally used to investigate possible aetiologies, using
MRCP to demonstrate gallstones not identified on US or CT.
Chronic pancreatitis
Radiological features
• AXR: multiple irregular pancreatic calcifications are virtually patho-
gnomonic. More commonly seen in alcohol-related disease.
• USS:
• Atrophic, bright pancreas, often with focal or extensive irregular duct
dilatation.
• Intraductal calcifications.
• Less commonly focal or diffuse glandular enlargement, mainly caused
by mild biliary duct dilatation.
242
P
Acute pancreatitis. Swollen enhancing pancreas with extensive
peripancreatic inflammatory soft tissue stranding (arrow).
Acute pancreatitis. Diffusely swollen pancreas with extensive
243
peripancreatic inflammatory free fluid (arrows).
• CT (pre- and postcontrast):
P
• Similar findings to those with US.
• NECT is excellent for demonstrating minimal degrees of pancreatic
calcification.
• CECT allows further characterisation of necrotic areas or intra/peri-
pancreatic fluid collections.
• Allows assessment of regional damage caused by recurrent inflamma-
tion (e.g. damage to the duodenum).
• MRI:
• Loss of signal intensity on T1W fat-suppressed images results from loss
of aqueous protein from pancreatic acini owing to fibrosis.
• Suboptimal contrast enhancement on post-gadolinium T1W images
results from destruction of the vascular tree by fibrosis.
• Cholangiopancreatography:
• Increasing use of MRCP has to a degree supplanted the use of ERCP.
• Key finding is irregular dilatation of pancreatic duct (grade I-III).
• Ranges from slight duct ectasia to beading of main duct with intra-
ductal protein plugs.
• Filling defects may represent calculi.
Complications of pancreatitis
Pancreatic necrosis
• Release of proteolytic and procoagulant enzymes within the pancreas
leads to diffuse necrosis.
• CECT is 90% accurate for diagnosis, demonstrating non-enhancement
of necrotic tissue.
• Often associated with other complications, for example ARDS, wide-
spread fat necrosis.
• Prognosis depends on degree of necrosis, rising to 50% mortality with
90% glandular necrosis.
Infected pancreatic necrosis
• Necrotic pancreatic tissue that becomes infected.
• May occur at any time following pancreatitis.
• CECT: gas-forming organisms produce pockets of gas within non-
enhancing necrotic pancreatic tissue.
• Open surgical debridement and drainage required.
• Prognosis worse than for pancreatic abscess.
244
P
Chronic pancreatitis. Secondary multifocal coarse pancreatic
calcification (arrows).
Necrotising pancreatitis. Non-enhancing necrotic tissue within the head
and proximal body of the pancreas (arrow).
245
Acute fluid collections (phlegmon)
P
• Collections of enzyme-rich fluid anterior to pancreas.
• Occurs in ~40% of patients with acute pancreatitis.
• Usually anterior and peripheral to gland.
• No fibrous capsule, as opposed to pseudocyst formation.
• Because of the proteolytic nature of the fluid, the collection can readily
spread into the mediastinum, pararenal fascia and neighbouring organs
(e.g. liver and spleen).
• Resolution is spontaneous in 50% and only requires serial follow-up.
• In the other 50% pseudocysts develop or complications of superadded
infection or haemorrhage occur.
• Often only distinguishing factor from pseudocyst is resolution on serial
imaging.
• On CECT, the collection presents as a hypodense area of water density
(<10HU) within the pancreatic bed, which may persist for months.
Pancreatic pseudocyst
•
A collection of pancreatic fluid that is encysted in a fibrous wall.
•
Occurs approximately 1 month after the acute onset of pancreatitis.
•
Usually oval or round in configuration.
•
Two-thirds are located within the pancreas itself but can occasionally be
intraperitoneal, retroperitoneal, intraparenchymal or mediastinal.
•
Caused by microperforation of the pancreatic duct, leading to an
encapsulated collection of fluid and enzymes.
•
Follows acute pancreatitis in up to 10% of patients. May also been seen
in those with chronic pancreatitis.
•
Usually sterile but may become secondarily infected, leading to abscess
formation.
•
Of pseudocysts, 50% resolve spontaneously and pose no problems, 20%
are stable and 30% lead to complications including:
• severe haemorrhage caused by erosion into superior mesenteric
vessels.
• splenic vein thrombosis from adjacent inflammation and pressure.
• peritonitis caused by rupture into the peritoneal cavity, lesser or
greater sac.
• dissection into adjacent organs such as liver, spleen or posterior wall of
stomach.
• pressure obstruction of duodenum or common bile duct, leading to
jaundice and cholangitis.
• superadded infection.
• increasing size through communication with pancreatic duct or
haemorrhage and osmosis.
246
P
Chronic pancreatitis with pseudocyst formation. Multiple
peripancreatic pseudocysts (asterisks). Note the pancreatic calcification
typical of chronic pancreatitis.
• AXR:
• Indentation of posterior stomach wall by cyst.
• Indirect evidence of pancreatic mass (e.g. displacement of the splenic
flexure, downward displacement of duodenojejunal junction.
• Gastric outlet obstruction.
• Not sensitive or specific.
• USS:
• Usually a unilocular single hypoechoic cyst is seen in relation to
pancreas.
• Multilocular in under 10%.
• May contain internal echoes owing to debris, haemorrhage, cellular
debris.
• CECT: fluid-filled peripancreatic cyst of 0-30HU attenuation.
• ERCP: communication with pancreatic duct in 70%.
Pancreatic abscess
• Fluid collection, often in relation to the pancreas, containing bacteria
and pus.
• Usually occurs 4 weeks after acute pancreatitis.
• Enhancement of abscess capsule on CECT.
• Managed by percutaneous drainage.
247
P
Portal vein thrombosis
• In the majority of cases, no cause for portal vein thrombosis is evident, in
spite of extensive investigation.
• More common in children and young adults.
• Secondary causes include:
• trauma: accidental, intraoperative or following umbilical vein
cannulation.
• tumour invasion, typically in hepatocellular carcinoma, cholangio-
carcinoma and pancreatic carcinoma.
• blood dyscrasias leading to hypercoagulable states, such as sickle cell
disease and severe dehydration (again more common in children).
• intra-abdominal sepsis.
• cirrhosis and portal hypertension, presumably owing to altered hae-
modynamics and venous stasis.
• May be isolated with relatively few effects or may result in:
• portal hypertension, with its associated complications.
• extension of thrombosis into mesenteric vessels, with subsequent
small-bowel infarction.
• hepatic infarction: two-thirds of the hepatic blood flow is derived
from the portal vein.
Clinical characteristics
• Most commonly seen in children.
• Hepatic encephalopathy as a result of diversion of portal blood through
the systemic circulation.
• Abdominal pain.
• Haematemesis secondary to oesophageal varices.
Radiological features
• AXR:
• Shows hepatosplenomegaly.
• Not very sensitive.
248
P
Portal vein thrombosis. Non-opacification of the porto-splenic venous
confluence (arrow) and distal splenic vein. Note the presence of ascites (asterisk).
249
•
USS:
P
• Best imaging modality for screening in high-risk cases (lack of radi-
ation is especially useful in the younger target age group).
• Portal thrombosis appears as intraluminal material, of variable echo-
genicity, in an enlarged portal vein.
• Presence of portal hypertension can be confirmed by direct Doppler
interrogation of the portal vein and presence of porto-systemic col-
laterals and ascites.
• Decrease in hepatic arterial resistive index - a value of <0.5 is highly
suggestive - as a result of a partial compensatory increase in arterial
flow. This may be helpful in differentiating acute from chronic portal
vein thrombosis, as a fall in the index is not seen in the chronic
condition or in non-occlusive thrombosis (e.g. malignant thrombus).
• Cavernous transformation may be evident in subacute or chronic
thrombosis: a thrombosed extrahepatic portal vein is seen, with
multiple cavernous channels at the porta hepatis.
•
CT: typical appearances of intraluminal thrombosis:
• hyperdense thrombus on non-enhancing CT.
• hypodense filling defect surrounded by contrast on CECT.
•
MRI:
• Collaterals appear as multiple tubular flow voids.
• Portal vein thrombus is bright on T1W images.
•
Angiography:
• Thrombus appears as a filling defect on mesenteric venography.
• Tumour thrombus may appear as streaky contrast-filled vessels within
the thrombus-filling defect.
250
P
Portal vein thrombosis. CECT coronal reformat shows filling defect
within the portal vein, representing thrombus (arrow), secondary to
pancreatitis. Note the large secondary hypodense pseudocysts (asterisks).
251
P
Portal venous gas
•
Bacterial overgrowth and invasion of the colonic submucosal venous
plexus can lead to gas in the portal system. This results in portal venous
flow carrying gas to the liver.
•
In the adult, it is usually a result of bowel wall infarction and impending
gangrene, and carries a very poor prognosis. Often a pre-terminal event.
•
The aetiology usually involves intestinal necrosis from a variety of
causes. The most common include:
• arterial or venous occlusion leading to bowel infarction.
• any severe inflammatory colonic condition such as severe UC, severe
diverticular disease or intra-abdominal abscess.
• severe necrotising pancreatitis can also result in portal gas from sub-
sequent adjacent colonic inflammation.
• necrotising enterocolitis.
• perforated peptic ulceration.
•
In children, often accompanies bowel obstruction, most commonly
from some form of congenital atresia such as oesophageal or duodenal
atresia.
•
Also seen in relation to umbilical vein catheterisation in sick neonates.
•
A relatively common finding in neonatal necrotising enterocolitis,
where, in contrast to adults, it does not carry the same poor prognosis.
•
Other, less-common, causes include iatrogenic injection of gas during
colonoscopy, air embolism during barium enema procedures, pneumo-
nia and diabetes.
Clinical characteristics
• There are no distinct clinical features.
• Often just part of the picture in a severely ill adult or neonate.
• Its significance is as an indicator of severe sepsis or bowel infarction.
• It is a worse prognostic sign in adults than in neonates.
Radiological features
• AXR:
• Often a subtle finding, which may be transient, especially in neonates
with necrotising enterocolitis.
• Typically presents as peripheral hepatic branching linear lucencies.
• Gas may occasionally be seen in the intestinal wall (pneumatosis
intestinalis) and less commonly in the mesenteric vessels themselves.
252
P
Portal venous gas caused by bowel necrosis. Note the branching
lucencies within the portal venules, extending peripherally to the liver edge.
253
• USS:
P
• Gas in the portal venules within the liver produces intensely echo-
genic foci, often described as a ‘snow storm’ appearance.
• Doppler US reveals sharp bidirectional spikes superimposed on the
normal portal vein signal, caused by almost total reflection of sound
waves from microscopic portal gas bubbles.
• CT:
• Both NECT and CECT are exceptionally sensitive to the presence
of air.
• Portal vein gas is often difficult to distinguish from pneumobilia. In
gas within the intrahepatic bile ducts, the gas is often seen in a more
central position within the liver. This is presumably because of the
central flow direction of bile, carrying gas bubbles towards the porta
hepatis, whereas the portal flow carries the gas towards the periphery
of the liver.
254
P
Portal venous gas. Extensive gas seen predominantly within the left lobe
of liver, secondary to bowel necrosis.
255
P
Portal venous hypertension
•
Defined as a portal venous pressure of more than 5-10mmHg.
•
Almost always caused by increased portal venous resistance.
•
Site of resistance may be pre-, intra- or post-hepatic.
•
Prehepatic causes include portal venous thrombosis and portal venous
compression from tumour, lymphadenopathy or pancreatic masses
including a pseudocyst.
•
Intrahepatic causes include obstruction at the pre-sinusoidal, sinusoidal
and post-sinusoidal levels:
• pre-sinusoidal: congenital hepatic fibrosis, idiopathic non-cirrhotic
fibrosis, primary biliary cirrhosis.
• sinusoidal: hepatitis, sickle cell disease.
• post-sinusoidal: cirrhosis and veno-occlusive hepatic disease.
•
Post-hepatic causes are increased resistance to hepatic venous outflow, as
in congestive heart failure, constrictive pericarditis and Budd-Chiari
syndrome.
•
The most common cause world wide is schistosomiasis. In Western
countries, it is probably alcohol-induced cirrhosis, followed by hepatitis
C-induced cirrhosis.
•
Less-common causes include hyperdynamic flow states through the
portal venous system. These are mainly caused by abnormal vascular
channels in either hepatocellular carcinoma, or congenital arteriovenous
fistulae in conditions such as Osler-Weber-Rendu syndrome or trau-
matic arteriovenous fistula formation.
•
Whatever the underlying aetiology, elevated portal venous pressure
leads to diversion of GI venous return from the portal system to the
systemic circulation via porto-systemic collateral vessels.
•
Porto-systemic collateral vessels include:
• Coronary vein to azygos or hemi-azygos system via lower oesopha-
geal veins.
• Superior and inferior mesenteric veins to iliac veins via mesenteric
varices.
• Para-umbilical and omental veins to superficial veins of chest and
anterior abdominal wall via caput medusae veins.
• Splenic vein to azygos system via gastric fundal veins in gastrosplenic
ligament, producing splenic hilar varices.
• Gastric veins to renal veins via gastro-renal anastomoses.
• Gastric veins to pulmonary veins via gastric and pericardiophrenic
collaterals.
• Inferior mesenteric veins to internal iliac veins via rectal haemorrhoi-
dal veins.
256
P
Large lower oesophageal varices (arrowhead) caused by portal
hypertension. Note the irregular outline of the liver owing to chronic liver
disease.
Portal hypertension with splenic hilar varices (arrowhead; same
patient as above). Evidence of chronic liver disease and splenomegaly
257
(asterisk). Gallstones in gallbladder.
• Splenic veins to retroperitoneal venous system via duodenal and
P
retroperitoneal varices.
• Intrahepatic shunt from portal to hepatic veins.
• Portal venous congestion also produces splenomegaly, ascites and GI
congestion.
Clinical characteristics
• Dilated porto-systemic collaterals (varices), particularly in the lower
oesophagus and lower rectum, are submucosal in position, structurally
weak and easily traumatised, and may undergo torrential haemorrhage.
• Drainage of blood from the GI system, directly into the systemic venous
circulation, bypasses the detoxifying function of the liver and may lead
to hepatic encephalopathy.
• Increased portal pressure produces local GI mucosal congestion, leading
to GI bleeding, malabsorption, splenomegaly and ascites.
• Underlying liver damage potentiates the effects of malabsorption and
haemorrhage.
Radiological features
•
USS:
• Enlarged portal vein (abnormal >13mm diameter).
• Enlarged superior mesenteric and splenic veins (less useful).
• Presence of ascites, splenomegaly, evidence of liver disease such as
cirrhosis or fibrosis.
• Presence of porto-systemic collaterals, especially at the splenic hilum,
falciform ligament, ligamentum teres of liver
(recanalised para-
umbilical veins) and lower gasto-oesophageal varices.
• Important to identify patency of portal and splenic veins as this will
influence management.
• Increased echogenicity and thickening of intrahepatic portal veins.
• Loss of the normal portal venous pressure fluctuation with respiration;
normally portal velocity will increase in expiration.
• Reduction in portal venous velocity, normally 12-30cm s−1.
• Reversal of the normal portal venous flow towards the liver (i.e.
hepatofugal flow).
• Portal hypertension results in an absolute increase in hepatic arterial
flow to the liver. This manifests as a dilated hepatic artery with an
elevated resistive index, >0.78.
•
Angiography:
• Elevated portal pressures (hepatic wedge pressure minus IVC pres-
sure): normal is <5mmHg.
• Portal flow towards the liver and porto-systemic collaterals as seen
with US.
• Corkscrew hepatic arteries are fairly indicative.
258
P
Portal hypertension. CECT (coronal reformat) shows multiple large
lower oesophageal varices (arrow), splenomegaly (S), irregular liver edge
and ascites (asterisk).
• CT and MRI:
• The presence of ascites, splenomegaly, collateral flow and varices can
all be identified on CECT and MRI if disease is advanced enough.
• Often the underlying liver damage (e.g. cirrhosis or fibrosis) is also
evident.
259
Radiological management
P
•
The most serious complications are hepatic encephalopathy and massive
GI haemorrhage from bleeding porto-systemic varices.
•
Management is directed towards halting bleeding and reducing portal
hypertension.
•
Transjugular intrahepatic porto-systemic shunt (TIPS):
• An intrahepatic porto-sytemic shunt to reduce the portal hyperten-
sion is formed under radiological control. The procedure involves:
- percutaneous approach via right internal jugular vein.
- insertion of an expandable metallic stent between the hepatic and
portal veins within the liver substance.
• Shunt surveillance is with US at regular intervals of 3-6 months.
• The shunt and collateral morphology, as well as haemodynamics, are
assessed at each US assessment.
• TIPS is a valuable management tool especially in patients with upper
GI variceal haemorrhage or refractory ascites and hepatorenal
syndrome.
•
Complications of TIPS include shunt obstruction, dislodgement and
hepatic vein stenosis.
•
Operative complications include vascular injury, particularly to the
hepatic artery, subcapsular haematoma or intraperitoneal haemorrhage
and bile duct damage.
•
Although the procedure can lessen portal hypertension, it can worsen
hepatic encephalopathy as it will bypass the detoxifying effect of the
liver.
260
P
Massive rectal varices secondary to chronic portal venous thrombosis
(arrowhead).
Portal venous thrombosis. Volume rendered image of huge portosystemic
261
collateral circulation, in a patient with chronic portal venous thrombosis.
P
Pyelonephritis
•
Consists of upper urinary tract infection with concomitant pelvicalyceal
and parenchymal inflammation.
•
Subtypes include emphysematous pyelonephritis, tuberculous pyelo-
nephritis and xanthogranulomatous pyelonephritis.
•
Cause is infected urine from lower tract in 80% of adult cases.
•
An underlying anatomical abnormality is responsible for 5%:
• obstruction.
• ureteric or renal calculus.
• urinary stasis or reflux.
•
Produces wedges of infection extending from papillary tip to cortical
surface in patchy distribution, sharply demarcated from adjacent spared
parenchyma.
•
Escherichia coli and Proteus, Klebsiella, Enterobacter and Pseudomonas spp.
responsible for most infections.
•
Haematogenous spread with Gram-positive cocci cause 20%.
•
Far more common in women, probably because they have shorter urethrae.
•
Risk factors include:
• urinary obstruction (e.g. prostatic hypertrophy in adults).
• pelviureteric junction obstruction and posterior urethral valve syn-
drome in children.
• calculi in any age group.
• vesicoureteric reflux in children.
• pregnancy, through hormonal relaxation of ureteral smooth muscle
and pressure effects on the ureters.
• diabetes mellitus.
• instrumentation of urinary tract.
• immunosuppression.
• possibly environmental factors such as dehydration.
•
Medical treatment is usually initiated without any imaging.
•
Indication for imaging in acute situation:
• diabetes mellitus.
• history of renal calculi.
• underlying renal tract abnormality: anatomical (e.g. scarred kidneys)
or functional (e.g. outflow obstruction).
• growth of atypical organisms.
• poor clinical response to appropriate antibiotic treatment.
• frequent recurrences.
• more than one confirmed episode in a male.
•
Management is aimed towards rapid elimination of infecting organism
with use of antibiotics and fluid replacement.
262
P
Right pyelonephritis. Large focal area of reduced echogenicity (arrow)
replacing the lower half of the right kidney.
Right pyelonephritis. ‘Striated nephrogram’ showing stellate
hypoattenuating areas from hypoperfusion and oedema.
263
• In the case of obstruction, this must be relieved as a matter of urgency
P
in the presence of infection, if scarring is to be prevented. This may involve
placement of percutaneous nephrostomy or surgical removal of calculi.
• Underlying contributing factors must be dealt with, for example benign
prostatic hypertrophy causing outflow obstruction or calculi.
• Medical contributing factors must be controlled (e.g. optimum blood
glucose control maintained).
Clinical characteristics
• Presents acutely with signs of generalised sepsis such as fever, chills,
leukocytosis.
• Signs of localised infection: flank pain, pyuria and sometimes haematuria,
typically microscopic.
Radiological features
•
Imaging is important to ensure resolution of acute changes and to
exclude anatomical predisposing factors. It is useful in:
• defining underlying contributing factors such as obstruction, calculi,
reflux and previous renal scarring.
• diagnosing complications including renal and perirenal abscesses and
emphysematous pyelonephritis.
• diagnosing late sequelae, for example scarring, reflux and obstruction.
•
Intravenous urogram:
• Renal tract stones ± obstruction may be identified.
• Normal in over 75% of those with acute uncomplicated pyelonephritis.
• Renal enlargement may be seen, but this is poorly sensitive.
• Immediate persistent dense nephrogram is seen, which may be
striated.
• Shows delayed opacification of a collecting system.
• Compression of collecting system occurs from pyramidal oedema.
• In severe disease, the kidney cannot be visualised: the aetiology is
multifactorial, including interstitial oedema, inflammation, vasculitis
and nephron function disruption.
•
USS:
•
Usually normal.
•
When abnormal, most common findings are calculi and evidence of
obstruction:
• enlarged kidney(s)
• decreased echogenicity: either focal or global.
• loss of central sinus echogenicity owing to fat infiltrated with inflam-
matory oedema.
• loss of corticomedullary differentiation.
• mild hydronephrosis and ureteral dilatation.
• perirenal fluid from localised perinephric exudates.
264
• abscesses or collections.
P
Left pyelonephritis. Swollen and hypoattenuating left kidney.
Bilateral pyelonephritis. Large hypoattenuating areas within the renal
265
cortices caused by hypoperfusion and oedema.
• CT:
P
• Often normal.
• Localised or generalised renal enlargement identified.
• Wedge areas of hypoattenuating cortex in early contrast phase owing
to hypoperfusion and oedema.
• Poor corticomedullary differentiation.
• Mild pelvicalyceal and ureteral dilatation.
• Thickening of Gerota’s fascia.
• Perirenal stranding, implying perirenal inflammatory changes.
• Persistent enhancement on delayed scans in areas of diminished
enhancement on early phase.
• Calculi may be seen, as may obstruction.
• MRI: wedge-shaped areas of high signal on T1W post-gadolinium
sequences.
• Renal cortical scintigraphy:
• Generalised or focal decreased uptake of tracer
(usually
99mTc-
labelled DMSA).
• Reflects decreased nephron function.
Complications of pyelonephritis
Abscesses
•
Renal tract abscesses:
• Usually caused by E. coli or Proteus spp.
• Cause is an ascending infection in 80%.
• Cause is haematogenous in 20%; there is a relatively high incidence in
IV drug users, endocarditis, blood sepsis and diabetics.
•
Renal abscess:
• IVU: Focal mass displacing collecting system.
• CECT: non-enhancing low-attenuation intrarenal lesion with enhan-
cing capsule.
• Gas bubbles may be present and are pathognomonic but not invariable.
• Perinephric fat obliteration.
• USS: hypoechoic mass with a thickened wall and septations.
• NM: Localised hot spot on galium-67 imaging.
•
Perinephric abscess:
• Caused by extension of a renal abscess through the renal capsule.
• High incidence in diabetics.
• CT: Focal renal mass extending through capsule on cross-sectional
imaging.
• Occasionally gas in renal fossa.
• AXR: shows loss of psoas muscle margin with ipsilateral scoliosis
caused by localised muscle spasm, concave towards involved side.
266
P
Thick-walled abscess (arrowhead) at the upper pole of the right kidney (K).
267
Large right renal abscess (asterisk) and smaller left renal abscesses (arrows).
Xanthogranulomatous pyelonephritis
P
• Chronic suppurative granulomatous infection in patients with chronic
obstruction.
• Usually patients have underlying diabetes mellitus.
• Characterised by parenchymal destruction and replacement with lipid-
laden macrophages.
• There is diffuse renal involvement in 90%, with 10% being localised.
• Females are more affected than males; 10% seen in diabetics.
• Large or staghorn calculus seen in 75%, but in 25% is caused by non-
calculus obstruction.
• Produces an enlarged non-functioning kidney.
• USS may show dilated calyces, low-echo masses, with internal echoes
replacing the renal parenchyma.
• CECT: the kidney is replaced by multiple non-enhancing low-
attenuation masses. The attenuation value of these masses varies
from −10 to 30HU, depending on the lipid content. Caluli are readily
apparent.
Emphysematous pyelonephritis
• Usually caused by Gram-negative infection in diabetics.
• Ranges from gas in collecting system alone (emphysematous pyelitis),
with a mortality of 20%, to gas in the renal parenchyma and collecting
system (emphysematous pyelonephritis), with a mortality of up to 80%.
• CT: most reliable in these cases and can detect small pockets of air
within the kidney, often extending beyond the capsule and into the
renal vein and retroperitoneum.
• USS:
• May demonstrate echogenic foci within the renal parenchyma and
sinus caused by gas. Occasionally gas in the perinephric space can
obscure the kidney.
• Treatment is with nephrectomy.
268
P
Xanthogranulomatous pyelonephritis of the left kidney. Chronic
obstructing calculi and several large non-enhancing masses are seen
expanding the left kidney.
269
R
Renal artery stenosis
Clinical characteristics
•
Renal artery stenosis (RAS) is responsible for up to 4% of cases of
hypertension, rising to one in four patients with resistant hypertension
and half of those with malignant hypertension.
•
Approximately 80% is caused by atherosclerosis; 90% of atherosclerotic
stenoses are in the renal artery trunk, usually within 2cm of its origin.
•
The majority of the remainder results from fibromuscular hyperplasia.
This is the most common cause of elevated blood pressure in children
and young adults and can result in renal insufficiency. This can lead to
beading of the artery through alternating stenoses and aneurysmal
dilatations. It may also cause a smooth, tubular narrowing. These find-
ings can be subtle.
•
Rarer causes include dissection, vasculitis, thromboembolic events and
retroperitoneal fibrosis.
•
Screening for RAS should be considered in:
• severe hypertension in young patients.
• a unilateral atrophic kidney.
• worsening of renal function following use of an angiotensin-converting
enzyme (ACE) inhibitor.
• hypertension with associated renal impairment of unknown aetiology.
• long-standing hypertension that rapidly deteriorates.
• refractory hypertension.
• presence of abdominal bruit and renal impairment and/or hypertension.
• vasculopathies with renal impairment and/or hypertension.
270
R
Renal artery stenosis. MR angiography demonstrates bilateral renal
artery stenoses (arrows).
271
Radiological features
R
•
USS:
• A useful imaging technique in assessing renal impairment and hyper-
tension as it can assess renal size, hydronephrosis, renal scarring and
masses. It is relatively insensitive for detecting RAS as visualising the
renal artery can be difficult in many patients. A further complicating
factor is the common occurrence of multiple renal arteries.
• US usually only helpful in children, very thin adults and in assessing
transplant arteries
(which are more accessible at US than native
arteries).
• US signs of RAS are:
- direct visualisation of the stenosis.
- apeaksystolicvelocityof>150cms−1withadopplerangleof<60°.
- peak renal artery velocity : peak aortic velocity >3.5.
- absent diastolic forward flow.
- spectral broadening, owing to turbulence.
- dampened waveforms: slow upstroke (pulsus tardus) and decreased
amplitude (pulsus parvus).
•
CECT:
• CT angiography using multislice scanners, fine collimation and arte-
rial phase IV enhancement can accurately delineate RAS, but at a
high radiation dose.
• Note that iodinated contrast is nephrotoxic and may cause deterio-
ration in the renal function. Patients should be well hydrated.
•
MR angiography:
• Now the first choice for assessing for RAS, with sensitivities of >95%
and specificities of >90%.
• Although phase contrast and time of flight studies have been used
in the past, generally gadolinium-enhanced MR angiography is now
used.
• Can be limited by the presence of a metallic stent. May have difficulty
assessing small arteries, either accessory vessels or more distal branches.
272
R
Renal artery stenosis. MR angiography demonstrates a severe right renal
artery stenosis (arrow). Note the markedly atrophic left renal artery
(arrowhead). See interventional digital subtraction images below.
273
• Conventional angiography:
R
• This allows direct visualisation of a RAS.
• Also allows pressure studies, which help to assess subtle stenosis.
• Percutaneous transluminal angioplasty and endovascular stent
placement are the first-line treatments for RAS.
• Complications of renovascular intervention include partial or total
arterial occlusion owing to thrombosis or dissection, with possible
consequent renal infarction. Renal artery spasm can also occur.
274
R
Renal artery digital subtraction angiography. Following the renal
MRA (MR angiography (above)), the patient underwent formal renal artery
digital subtraction angiography. This confirmed the severe right renal
artery stenosis (arrow) and atrophic left renal artery. This was treated with
275
balloon angioplasty and stent insertion, with an excellent end result.
R
Renal calcification
• Calcification of the renal system may be partial, total, regional, unilateral
or bilateral. It may involve just the kidney, part of the organ, or it may
be widespread and involve the associated ureter, bladder and seminal
vesicles in the male. The causes of calcification are varied but may be
classified according to the pattern of involvement.
• Renal calculi are dealt with in a separate section.
• Calcification caused by regional/local disease may involve either part of
the kidney or the retroperitoneum.
Perirenal retroperitoneal calcification
•
This is usually caused by trauma, infection or neoplasm.
•
Traumatic retroperitoneal haematomas often resorb only partially, leav-
ing an area of dystrophic calcification behind.
•
The most common neoplasms associated with retroperitoneal calcifica-
tion are Wilm’s tumour, neuroblastoma, teratomas and cavernous
haemangioma.
• Wilms’ tumour is the most common solid renal tumour of childhood
and may be bilateral in 10%, arising from nephroblastomatosis. The
tumour is usually large at presentation, arising from the renal cortex
and associated with calcification in about 15%.
• Neuroblastoma is the most common abdominal malignancy in
infancy (cf. Wilms’ tumour, which is more common in childhood).
It arises from neural crest tissue and presents as a large solid mass, often
extending across the midline, encasing the aorta and IVC. It typically
produces fine amorphous or stippled calcification (in 85%).
• Teratomas may occasionally extend to the level of the kidneys. They
characteristically produce distinctive calcified structures such as
teeth, cartilage and even digits. The appearance is radiologically
characteristic.
• Cavernous haemangiomas of the retroperitoneum demonstrate
characteristic calcified phleboliths within their capacious sinusoidal
network.
276
R
Calcified tuberculous right psoas abscess (asterisk). Note the expansion
and bowing of the psoas outline, with lateral displacement of the right kidney.
TB autonephrectomy. IVU demonstrates a densely calcified,
non-enhancing, small right kidney. The appearances are characteristic
277
of long-standing renal tuberculosis.
R
Calcification associated with a distinct
renal mass
•
The differential lies between a renal tumour, cyst, infection or vascular
calcification.
•
A calcified renal mass should always be investigated further as it may be
malignant in up to 75% of cases. The pattern of calcification is also
important. Peripheral calcification is usually non-malignant (20%) and is
usually calcification in a renal cyst. Non-peripheral calcification is
malignant in nearly 90% of occurrences.
•
Renal tumours associated with calcification are Wilms’ tumour in
childhood and renal cell carcinoma in adulthood, where the calcifica-
tion is often non-peripheral.
•
Renal infection with TB often results in calcification of the resulting
abscess (see Tuberculosis). Characteristic calcifications in a lobar distribu-
tion are often seen in end-stage TB: TB autonephrectomy.
•
Pyogenic abscesses rarely calcify.
•
Hydatid disease affects the kidneys in under 5% of patients, but when
present calcifies in over 50%.
•
Renal cysts demonstrate a spectrum of calcification. Simple cysts very
rarely calcify (1%) but intracystic infection or haemorrhage often results
in areas of intracystic calcification. In multiple renal cysts, as in adult
polycystic disease, peripheral egg-shell calcification is common and
much of the kidney becomes replaced by multiple areas of thin egg-
shell calcification.
•
Vascular calcification associated with a mass may result from either RAS
or a renal artery aneurysm, in which case a soft tissue mass is associated
with curvilinear or circular egg-shell calcification.
278
R
Peripheral calcification in a right renal cyst (arrow).
Hydatid disease of the left kidney. A large peripherally calcified cyst
(asterisk), containing internal septations, is seen expanding the left kidney.
279
R
Nephrocalcinosis
• This is renal parenchymal calcification, which may be cortical or medul-
lary in distribution. Overall incidence varies from under 1% to around 5%
of the population and it has a wide spectrum of causes: 95% is medullary
nephrocalcinosis with 5% presenting as cortical nephrocalcinosis.
Cortical nephrocalcinosis
• This presents as peripheral (cortical) calcification with sparing of both
the subcortical renal region and the medullary pyramids, producing
classical ‘tramline’ calcifications.
• On US the involved kidney demonstrates a highly echogenic cortex.
• The most common causes include:
• chronic glomerulonephritis.
• cortical necrosis, most commonly from pregnancy, renovascular
shock, infection and renal toxins such as ethylene glycol ingestion
(antifreeze).
• less common causes include Alport’s syndrome (hereditary nephritis
and deafness), rejected renal transplant and chronic hypercalcaemic
states (congenital, paraneoplastic).
Medullary nephrocalcinosis
• In medullary nephrocalcinosis, calcifications involve the distal convo-
luted tubules within the renal medullary regions.
• AXR: There is stippled calcifications of the medullary pyramids.
• USS: There is corresponding hyperechogenicity of the involved renal
pyramids. The earliest sign may be absence of the relatively hypoechoic
renal medulla.
• The most common causes include:
• hyperparathyroid states, producing chronic hypercalcaemia and
hypercalciuria.
• papillary necrosis (diabetes, cirrhosis, chronic renal obstruction, anal-
gesic abuse and sickle cell disease).
• medullary sponge kidney.
• chronic pyelonephritis.
• other causes of hypercalciuria such as milk alkali syndrome, hyper-
vitaminosis D, renal tubular acidosis, hyperthyroidism, Cushing’s
syndrome, diabetes mellitus and paraneoplastic syndrome.
• causes of hyperoxaluria such as hereditary hyperoxaluria and derange-
ments of bile acid metabolism.
280
R
Medullary sponge kidney. Hyperechoic medullary pyramids (arrows)
seen in US.
Nephrocalcinosis in medullary sponge kidney. Tomogram from an
IVU series. Extensive stippled calcification can be seen within several
281
medullary pyramids (arrows).
R
Renal developmental anomalies
Horseshoe kidney
• Most common renal fusion anomaly, with an incidence of up to 1%.
• Caused by fusion of lower renal poles in 90% and by upper pole fusion
in 10%.
• Associations include:
• Turner’s syndrome.
• trisomy 18.
• cardiovascular, CNS, anorectal, genitourinary and musculoskeletal
anomalies.
• vesicoureteric reflux.
• pelviureteric junction obstruction with secondary hydronephrosis.
• Complications include renal calculi formation and infection, possibly
caused by urinary stasis.
Radiological features
• May be imaged with US, IVU, CT or MRI.
• Plain AXR may demonstrate absence of normal renal outlines.
• Long axes of the kidneys are medially orientated.
• Kidneys lie more inferiorly than the normal renal position, at L4/5,
lying between the aorta and the inferior mesenteric artery origin.
• The renal pelves point anteriorly.
282
R
Horseshoe kidney by IVU (arrows).
283
Horseshoe kidney on CT (arrows).
R
Crossed renal ectopia
• The kidney is located on the opposite side from the insertion of its
ureter into the bladder.
• Usually the kidneys are fused (crossed fused renal ectopia).
• The crossed kidney lies more inferiorly.
• Associations include:
• obstruction.
• calculi.
• hypospadias.
• urethral valves.
Pelvic kidney
• Failure of renal ascent has an incidence of 1:750.
• Often associated with failure of rotation, with the renal pelvis facing
anteriorly.
• Associations include:
• contralateral renal agenesis.
• hydronephrosis due to a high PUJ.
• reflux and hypospadias.
• A common cause of ‘absent’ kidney as seen on USS.
Complete ureteral duplication
(duplex kidney)
• Duplex kidney is caused by two, rather than one, ureteral buds arising
from the mesonephric duct, resulting in ureteral duplication. Up to 40%
are bilateral.
• The lower moiety drains the lower pole and mid-kidney and distally
inserts into the trigone of the bladder. This moiety is prone to vesi-
coureteric reflux, owing to a short ureteral course through the bladder
wall, with an associated risk of secondary chronic pyelonephritis.
Pelviureteric junction obstruction is a further complication.
284
R
Crossed left renal ectopia. The ‘crossed’ left kidney lies inferior to
the right as seen in IVU, but the left ureter correctly inserts into the left
side of the bladder.
285
Pelvic kidney on CT (arrow).
• The upper pole moiety’s ureter has an ectopic distal insertion, more
R
inferior and medially to the lower pole moiety ureter. In men, the
insertion is always proximal to the external sphincter, so enuresis does
not occur. It can insert into the seminal vesicles, prostatic urethra and
ejaculatory ducts as well as low in the bladder. Men with duplex kidneys
are more at risk of epidymo-orchitis. Women with a duplex system may
have distal insertion below the external sphincter, leading to enuresis.
Sites include the distal urethra, genital tract and rectum.
• In men and women, the upper pole moiety is prone to obstruction
because of its ectopic distal insertion. An increased incidence of uretero-
coele is seen, further increasing the risk of distal obstruction.
Radiological features
• IVU:
• If both moieties are functional, then the ureteric duplication and the
sites of insertion distally will be demonstrated.
• The upper moiety may opacify poorly owing to obstruction. The
associated hydronephrosis may displace the lower moiety calyces
inferiorly - ‘drooping lily sign’.
• The lower pole may demonstrate scarring and atrophy, with secon-
dary loss of function through reflux nephropathy. The lower moiety
ureter may be tortuous and dilated because of reflux.
• USS:
• The kidney may be enlarged.
• Two renal pelves separated by renal parenchyma may be seen.
• Complications such as hydronephrosis or scarring can be identified.
Partial duplex system
• Partial duplex system is a result of a single ureteric bud branching. In
distinction to a full duplex system, there is a single distal ureter and
bladder insertion.
• It can be associated with obstruction of the lower pole pelviureteric
junction.
•
‘Yo-yo’ reflux, when urine refluxes from lower moiety into upper or
visa versa, may occur.
• There is an increased risk of urinary tract infection.
286
R
Pelvic kidney. Radioisotope study shows small left pelvic kidney (arrowheads).
Left duplex system. The lower moiety inserts into the bladder trigone
(not shown), whereas the upper moiety ureter usually has an ectopic distal
287
insertion (see text).
R
Renal agenesis
• If there is failure of induction, the ureteral bud does not lead to
formation of metanephric tissue. This results in a blind-ending ureter.
• In formation failure, the ureteral bud does not form, with resultant
absent trigone, ureter and kidney.
• Bilateral agenesis, Potter’s syndrome, is invariably fatal in the first week
of life owing to associated pulmonary hypoplasia.
• Unilateral agenesis is associated with trisomies, Turner’s syndrome and
Fanconi’s anaemia.
• Renal agenesis is associated with other urogenital tract anomalies. In
men, there may be agenesis or hypoplasia of the gonads and vas defer-
ens. In women, 40% of patients with renal agenesis have uterine or
vaginal anomalies.
288
R
Left partial duplex. The two ureters combine into a single ureter
opposite L4.
289
R
Renal masses: benign
Simple renal cysts
• The simple cyst is the most common renal mass, possibly caused by
tubular blockage.
• In those aged over 50 years, 50% have simple renal cysts.
• These cysts have no malignant potential.
Radiological features
• USS:
• Rounded outline.
• Smooth, well-defined walls.
• Anechoic contents with through enhancement.
• Accuracy of >90%.
• CT:
• Well-defined, thin walls with no enhancement.
• Fluid density <25HU.
• Accuracy of >90%.
• MRI:
• Well-defined lesion of water signal intensity.
• Low signal on T1W and high on T2W imaging.
• No solid components and no enhancement.
• IVU:
• Smooth displacement or indentation of collection system.
• Well-defined defect identified in the nephrogram.
• Well-defined, non-enhancing exophytic lesion.
• IVU lacks sensitivity and specificity.
Renal sinus cyst (parapelvic cyst)
• Rounded fluid-filled cyst within the renal pelvis, arising from the renal
parenchyma or renal sinus, but not connected to the collecting system.
• Can occasionally cause calyceal obstruction or rarely hydronephrosis.
Usually asymptomatic.
290
R
Simple cyst arising from the lower pole of left kidney (arrow). Note the
anechoic contents and through transmission.
Right renal cyst. Well-defined low-attenuation lesion within the right
291
kidney (arrowhead).
Radiological features
R
• USS: may be mis-diagnosed as hydronephrosis on US.
• On a contrast study, such as an IVU or CT urogram, the lack of contrast
within the parapelvic cyst will differentiate it from hydronephrosis.
• If symptomatic, they can be ablated with an ultrasound guided 95%
alcohol injection.
Atypical cysts
• Simple cysts may have atypical appearance following haemorrhage or
infection.
Radiological features
• USS:
• Rounded lesion but wall may appear thickened.
• Shifting debris seen within cyst.
• Internal septations may be identified.
• CT: increased density
(>25HU) but no enhancement, owing to
increased protein within cyst.
• MRI: the usual signal of a simple cyst, low on T1W and high on T2W,
is altered, with iso- or hyperintensity seen on T1W, as a result of the
increased protein or blood breakdown products. The T2W signal is
usually hyperintense but less so than in a simple cyst.
Adult polycystic kidney disease
Clinical characteristics
• Adult polycystic kidney disease is an autosomal dominant condition that
results in a collagen defect.
• Carrier frequency is 1:1000.
• Associations include:
• berry aneurysms.
• cysts in pancreas, liver and other organs.
• mitral valve prolapse.
• Presentation usually occurs in the 4th or 5th decade.
• Presents with loin masses, renal failure, hypertension, haematuria,
proteinuria and abdominal pain.
• Complications include:
• renal failure.
• cyst haemorrhage, a cause of abdominal pain.
• calculi, often urate.
• increased incidence of renal cell carcinoma.
292
R
Large left renal cyst. The cyst is of uniform high signal intensity
(arrow) on T2W MRI.
293
Right parapelvic cyst (arrow).
Radiological features
R
• AXR: not a first-line investigation but may show enlarged renal shad-
ows and curvilinear cyst calcification.
• USS:
• Multiple, bilateral cortical cysts.
• Enlarged kidneys.
• Renal outline is indistinct and as disease develops less cortical paren-
chyma is seen.
• If complicated by haemorrhage, debris may be seen within the cysts.
• IVU:
• Multiple areas of poor opacification on the nephrographic phase.
• Distortion of collecting systems.
• CT and MRI:
• Thinned parenchyma.
• Multiple, thin-walled, cysts. Mural thickening raises possibility of
complicating carcinoma.
• Low attenuation on CT.
• On MRI, they are low signal on T1W and high on T2W imaging.
• Following haemorrhage, cyst attenuation increases on CT. On MRI,
the T1W signal increases and the T2W signal is variable.
Acquired cystic kidney disease
Clinical characteristics
• Chronic renal cysts develop in patients on haemodialysis.
• There is a 90% incidence after 10 years of haemodialysis.
• Complications and associations include:
• haemorrhage into the cysts.
• increased incidence of renal cell carcinomas, with up to 5% of patients
developing these.
Radiological features
• The diagnosis is made by the presence of more than three cysts in a patient
on haemodialysis, without history of inherited cystic renal disease.
• The kidney will usually be atrophic. The cysts are usually <3cm, thin
walled and will have similar appearances to those seen in polycystic
kidney disease, including the findings of intracystic haemorrhage.
• The importance of imaging is to detect developing renal cell carcinomas.
294
R
Atypical left hyperdense cyst. The increased density may be caused
by haemorrhage or proteinaceous debris.
Adult polycystic kidney disease. Multilple thin-walled cysts are seen
replacing both kidneys, with no discernible normal parenchyma remaining.
295
Note the multifocal areas of cystic dystrophic calcification.
R
Angiomyolipomas
Clinical characteristics
• Angiomyolipoma is a benign renal mesenchymal tumour containing fat,
smooth muscle and thick-walled blood vessels.
• Incidence is up to 3%.
• The vessels do not have a complete elastic layer, predisposing to aneur-
ysms and haemorrhage. If the tumour is greater than >4cm, there is a
60% chance of haemorrhage.
• Angiomyolipoma is sporadic in 80% and associated with tuberose
sclerosis in
20%. There is an association with von Hippel-Lindau
syndrome and neurofibromatosis.
• These tumours are often incidental findings, but if haemorrhage occurs,
they may present with flank pain or haemodynamic shock.
Radiological features
• The presence of fat within a renal lesion is highly suggestive of angio-
myolipoma, although occasionally renal cell carcinoma contains fat, as
do renal lipomas and liposarcomas. All these differentials are rare and
interval follow-up scans should be considered.
• USS: characteristic fat appears as areas of increased echogenicity within
a mass, but this is non-specific as calcification can cause confusion.
• CT: demonstrates variable amounts of fat within the lesion and may
demonstrate the presence of haemorrhage.
• MRI: focal fat with high T1W and T2W signal, that show signal loss on
fat suppression sequences, is characteristic.
296
R
Acquired cystic kidney disease. Large bilateral renal cysts (asterisk).
Right angiomyolipoma. Large fatty echogenic mass seen within the
lower pole of the right kidney (arrow).
297
R
Oncocytomas
Clinical characteristics
• A well-defined tumour seen from the 3rd decade onwards with a peak
in the 7th.
• Contains oncoctyes and well-differentiated proximal tubular cells.
• Often has central scar.
• Usually asymptomatic and may be discovered incidentally; it can be up
to 10cm in diameter.
• Unfortunately, neither the radiological findings nor needle biopsy can
reliably differentiate oncocytomas from renal cell carcinomas, and
resection is usually necessary.
Radiological features
• USS:
• Well-defined mass, often with a hypoechoic central scar.
• No through enhancement.
• CT:
• Well-defined mass with homogeneous enhancement, but if there is a
central scar this will be seen as a stellate, poorly enhancing region.
• Pseudocapsule may be seen.
• MRI:
• Again a well-defined lesion.
• The pseudocapsule may be seen as a well-defined low-signal rim.
• Shows enhancement, although scar may not enhance if present, with
peripheral washout.
298
R
Angiomyolipoma. Bulky heterogeneous mass expanding the upper pole
of the right kidney (arrowheads). Note the low-attenuation fatty areas
within the tumour.
Left oncocytoma. Large enhancing left renal mass with a prominent
299
stellate central scar.
R
Renal masses: malignant
Renal cell carcinoma
Clinical characteristics
• Adenocarcinoma accounts for 85% of renal malignancies.
• However, 30% are incidental findings during imaging.
• Most commonly present in the 6th or 7th decade; rarely occur in
children.
• Presenting features include haematuria, weight loss, fevers, anaemia,
flank pain or an abdominal mass.
• Cystic change may occur through necrosis, involvement of preexisting
cysts or in the cystadenocarcinoma histological subtype.
• Predisposing factors:
• von Hippel-Lindau syndrome is associated with multiple renal cell
carcinomas, in a younger cohort than usually seen.
• smoking.
• haemodialysis (as well as acquired cystic disease of renal failure).
• Metastatic spread includes ‘cannon ball’ lesions to the lungs, secondary
lesions to the liver, adrenal glands and contralateral kidney, lytic skeletal
metastases and hypervascular metastases to the brain.
• Direct spread can occur through the capsule into the perinephric fat and
the liver.
• Tumour may extend along the renal vein and involve the IVC and right
atrium.
Radiological features
• IVU:
• Control film may demonstrate a distorted renal outline and occasional
renal calcification. Skeletal metastases may be evident.
• Following contrast, parenchymal and calyceal distortion may be
evident.
• If there is renal vein obstruction, the affected kidney may not
enhance.
• Generally IVUs are non-specific and, in particular, small tumours may
be indistinguishable from other renal masses, such as simple cysts.
• USS:
• Tumours may be hypoechoic, hyperechoic or isoechoic compared
with renal parenchyma; most are hyperechoic. Cystic areas may be
present.
300
• Well-differentiated tumours may appear well demarcated.
R
Renal cell carcinoma. IVU shows an ill-defined mass (asterisk)
expanding the upper pole of the right kidney with distortion of the
upper pole calyces.
Renal cell carcinoma. Large solid mass (M), seen by US, is replacing
the upper two thirds of the kidney (K) with tumour extension (arrow)
into IVC (arrowhead).
• Involvement of venous structures (renal vein and IVC) may be seen as
soft tissue distending the vessels.
• US is less sensitive than MRI or CECT.
• CECT:
• A three-phase (precontrast, arterial phase contrast and portal phase
contrast) CT is usually performed.
301
• On precontrast studies, the lesion may be homogeneous if <3cm.
R
They are of increased density (>20HU) compared with simple cysts.
When >3cm in size, they become increasingly heterogeneous owing
to the increased incidence of necrosis. Tumour calcification may be
seen.
• Following IV contrast, the tumour generally enhances less avidly than
adjacent normal parenchyma, enhancement may be heterogeneous
owing to necrosis and cystic elements.
• The boundary between tumour and normal parenchyma is often ill
defined.
• The delayed phase of the CECT is very accurate at determining
venous involvement; this is seen as low-density filling defects within
the renal vein, surrounded by contrast.
• CECT is first-line investigation for tumour staging. It is effective at
demonstrating local adenopathy and distant metastases. Direct exten-
sion through the capsule into the perinephric space is well
demonstrated.
•
MRI:
• MRI is the most accurate imaging technique for abdominal staging of
renal carcinoma.
• Renal cell carcinomas are generally hypointense compared with renal
parenchyma on T1W and mildly hyperintense on T2W. However,
the difference in the signal characteristics between the tumour and
parenchyma, on an unenhanced scan, may be small; consequently, as
with CECT, three-phase enhancement is required.
• The 85% of tumours that are hypervascular will show heterogeneous
enhancement. Small tumours may demonstrate homogeneous enhance-
ment and may be iso-intense with parenchyma on some phases of
enhancement, hence the need for more than one postcontrast phase.
• The hypovascular carcinomas are usually of the papillary subtype,
are often well defined, and on CT can be mistaken for cysts. MRI
will demonstrate the solid nature of the lesions, and the presence
of subtle areas of enhancement within such lesions is highly
suggestive.
Radiological therapies
• There is a growing interest in the use of radiologically guided percuta-
neous thermal ablation, to debulk or destroy renal cell carcinomas (small
tumours) and for patients not fit for conventional surgery.
• Thermal ablation has been performed using radiofrequency probes,
laser fibres, high-power focused US and cryotherapy. These can be
monitored using CT, US or MRI. MRI has an advantage as changes in
temperature affect the T1W relaxation times, which can be utilised to
make an accurate, real-time thermal map to assess the extent of thermal
injury.
302
R
Right renal cell carcinoma. Tumour extends into, and expands, the
right renal vein (arrow), with tumour thrombus seen in the IVC (arrowhead).
Left renal cell carcinoma. Heterogeneous, hypervascular mass replacing
303
the left kidney.
R
Renal lymphoma
Clinical characteristics
• Primary renal lymphoma and Hodgkin’s lymphoma of the kidney are
rare, but secondary renal involvement in NHL is not uncommon,
especially in late disease.
• Spread to the kidneys may be by direct extension or haematogenous.
• Presents with a renal mass, haematuria, weight loss and flank pain, but
many are asymptomatic. May cause renal impairment.
• Other signs of lymphoma, such as splenomegaly, may be seen.
Radiological features
•
Lymphoma is bilateral in 75%.
•
Solitary masses in 20%.
•
There may be renal enlargement owing to diffuse infiltration but the
renal contour is maintained. Renal sinus involvement is common.
•
May be perinephric in position, and a kidney surrounded by tumour
mass, with no compression or renal function loss, is highly suggestive of
lymphoma.
•
Renal vessels that remain patent despite being surrounded by tumour
are also suggestive.
•
USS:
• Focal lymphomatous lesions are generally hypoechoic or anechoic,
but without the through enhancement seen in true cysts.
• Loss of echogenic renal sinus fat is a common finding.
• General hypoechoic renal enlargement.
• Other evidence of abdominal lymphoma may be seen.
•
CT:
• Single or multiple nodules within the kidneys can be identified. They
are of lower density and enhance less avidly than normal renal
parenchyma.
• Other evidence of abdominal or pelvic involvement is seen.
•
MRI: renal lymphoma is hypointense to renal parenchyma on T1W
imaging, and isointense or slightly hypointense on T2W. It shows
relatively poor gadolinium enhancement.
304
R
Renal lymphoma. Large, poorly enhancing, low-density lymphomatous
deposits within the right kidney (arrowheads) and two smaller lesions within
the upper and lower poles of the left.
305
R
Metastases to the kidney
Clinical characteristics
• Usually a feature of advanced malignant disease and usually asympto-
matic. Does not change management.
• May originate in many types of malignancy but common primaries
include lung, breast and GI malignancies.
Radiological features
• Usually bilateral, small, masses in a patient with a known primary.
• May be solitary, especially from colonic carcinoma. Solitary lesions tend
to be more infiltrative and less exophytic than renal cell carcinoma.
• If confirmation of diagnosis is clinically necessary, a needle aspiration
under CT or US guidance can be performed.
Transitional cell carcinonma
See separate section.
306
R
Large renal metastasis (asterisk) from an ovarian primary tumour
(arrow). This replaces most of the right kidney and infiltrates into the
adjacent liver.
307
R
Renal tract calculi
•
Calculous disease of the renal tract may affect the kidney, ureter or
bladder. It affects men four times as often as women and affects 1-2
per 1000 population in developed countries. The underlying causes are
many, with 70-80% of symptomatic patients having an underlying
metabolic abnormality.
•
Theories as to the pathogenesis include chronically raised levels of
calcium excretion, abnormalities in oxalate, cystine, urate and xanthine
metabolism, urological sepsis and urinary stagnation, and chronic
dehydration.
•
Renal calculi may be clearly visible on plain radiography, or non-
opaque depending on their mineral content and type.
•
Opaque stones include calculi composed of calcium oxalate/phosphate
and magnesium ammonium phosphate/calcium phosphate. Poorly
opaque stones are composed of cystine while uric acid, xanthine and
mucoprotein calculi are non-opaque.
•
Calcium-containing calculi either result from a raised level of serum
calcium, as in hyperparathyroidism, milk alkali syndrome and sarcoido-
sis, or occur with normal levels of calcium, as in urinary obstruction,
infection, renal tubular acidosis and medullary sponge kidney.
•
Similarly, oxalate stones may be associated with primary oxaluria, which
is a rare autosomal recessive condition leading to diffuse nephrocalci-
nosis, vascular calcification, urolithiasis, osteopenia and dense or lucent
bands within the metaphyses. Oxalate stones may also form in the
presence of abnormal bile acid metabolism, as in patients with small
bowel disease, Crohn’s disease or large segment small-bowel resection
at or near the terminal ileum (site of reabsorption of bile salts in the
hepatic-enteric cycle).
•
Uric acid stones may form in the presence of chronically raised serum
urate, as in gout or myeloproliferative disorders (increased cell break-
down and urate metabolism), particularly during treatment of such
conditions. Urate stones may also form in patients with ileostomies or
following chronic dehydration (hot climates). Finally, matrix stones are
seen in chronically infected kidneys.
•
Symptoms, clinical signs and radiological appearances depend to a
degree on whether a renal calculus causes incomplete or complete
obstruction, or does not obstruct the renal tract.
308
R
Staghorn calculus within a hydronephrotic right kidney (arrow). Note
the posterior acoustic shadowing typical of calculi in general.
309
Large bilateral staghorn calculi. Note that this is a ‘control’ film from an
IVU series and not a postcontrast radiograph.
Clinical characteristics
R
• Renal tract calculi may be entirely asymptomatic. When a calculus
causes renal obstruction, this results in severe renal angle pain, typically
colicky in nature, radiating from loin to groin, and associated with
nausea and vomiting.
• Renal calculi predispose to urinary tract infection. The association of
obstruction and infection, unless relieved, rapidly leads to significant
irreversible renal damage and may lead to renal failure.
Radiological features
•
Plain AXR:
• Radiodense stones may be seen on plain films of the involved area. An
important differential within the pelvis is the presence of phleboliths
within pelvic venous plexuses. These may cause diagnostic difficulty,
particularly in the region of the ureterovesical angles. Hydronephrosis
from calculous obstruction may be seen as enlarged soft tissue renal
shadows on plain AXR.
•
IVU:
• IVU is an important investigative tool in suspected calculous disease.
The control film may show radiodensities along the renal tract. After
administration of IV contrast, subtotal obstruction leads to a pro-
longed, delayed, nephrogram and hydronephrosis, the degree of
which depends on the degree of obstruction.
• The site of obstruction may be identified by a standing column of
contrast within the ipsilateral ureter above the level involved.
• In the case of total obstruction, there is, classically, absence of a
nephrogram on the affected side with no contrast seen within the
collecting system.
• Occasionally, in cases of total obstruction, high pressure within the
renal cortex causes extravasation of contrast within the perinephric
tissues.
310
R
Obstructed right kidney caused by a calculus just beyond the
pelviureteric junction (arrow).
311
• USS:
R
• Hydronephrosis is easily identified on US; an important differential is
a parapelvic cyst. US may also identify upper and lower ureteric
dilatation, although it is less accurate at identifying the mid ureter.
• Identification of bilateral ureteric jets of urine is important to exclude
ureteric obstruction. Calculi may be identified within the bladder as
echogenic foci with posterior acoustic shadowing.
• CT:
• NECT is the most accurate method of identifying renal tract calculi;
these are seen as radio-opacities surrounded by a cuff of ureteric
soft tissue. In contrast to other modalities, CT identifies all types of
renal tract calculus, including xanthine- and mucoprotein-containing
calculi.
• CT is particularly useful in identification of small ureteric calculi and
may identify ureteric oedema, perinephric stranding and renal
enlargement.
• Following contrast administration, delayed and decreased cortical
enhancement of the affected kidney reflects altered vascular dynamics,
owing to increased pressure within the distal nephrons.
312
R
Left renal tract obstruction, with a standing column of contrast, caused
by a left vesicoureteric junction calculus (arrow).
Obstructed left kidney. Renal tract CT shows the left kidney is
hydronephrotic. The obstructing calculus is clearly identified, surrounded
by a cuff of ureteric wall, within the proximal ureter (arrowhead).
313
R
Retroperitoneal fibrosis
• Retroperitoneal fibrosis (RPF) is the pathological formation of dense
fibrotic tissue in the retroperiteum, that can lead to complications
involving the vasculature, lymphatics and renal tracts.
• Primary RPF (60%) is:
• probably an autoimmune process, leading to a generalised vasculitis.
• associated with fibrotic changes in other organs in 10%, including:
- fibrosing mediastinitis.
- fibrosing thyroiditis.
- sclerosing cholangitis.
• may respond to steroid treatment.
• Secondary RPF (40%); causes include:
• radiation therapy.
• desmoplastic responses to great vessel aneurysms or neoplasias.
• drugs, including beta-blockers and amphetamines.
• response to retroperitoneal fluid collections.
• connective tissue disorders.
Clinical characteristics
• Non-specific symptoms such as weight loss, nausea, pyrexia and general
malaise.
• Dull pain in the back and abdomen.
• Renal impairment and/or hypertension.
• Lower limb oedema.
• Usually the fibrosis starts at the bifurcation of the aorta and extends
proximally to eventually involve the renal hilum. More rarely, it
extends distally into the pelvis.
314
R
Retroperitoneal fibrosis. IVU demonstrates the classical features of
medially ‘pulled’ ureters (arrow), ureteric tapering (arrowheads) and
ureteric ectasia.
315
Radiological features
R
• IVU:
• Classical triad of features:
- bilateral ectasia of the ureters superior to L4/5 from impaired
peristalsis.
- ureters pulled medially by the fibrotic tissue.
- gradual tapering of the ureters under extrinsic pressure from fibrotic
tissue.
• Generally mild hydronephrosis.
• USS:
• Homogeneous hypoechoic retroperitoneal soft tissue.
• Renal tract dilation may be seen.
• CECT: can show peri-aortic soft tissue, which may enhance if active
inflammation is present.
• MRI:
• Low to medium signal intensity in T1W image.
• May be high intensity in T2W image if there is active inflammation,
and low signal if fibrotic.
• NM: there is uptake of gallium during active inflammation.
316
R
Retroperitoneal fibrosis. CECT demonstrates circumferential soft
tissue surrounding the aorta (arrowhead) and encasing the mid ureter
(not shown); this results in moderate right hydronephrosis (arrow).
317
S
Splenomegaly
•
Abnormally enlarged spleen.
•
Various methods for assessing size. Most radiologists ‘eyeball’ spleen for
size.
•
Inferior tip of spleen extends below the tip of the right lobe of liver.
•
Splenic length is longer than 12cm on transverse diameter.
•
Splenic index is most accurate assessment: multiply the three dimensions
and a value >140cm3 is significant.
•
In children, the spleen is more than 1.25 times the length of adjacent
kidney.
•
Spleen longer than 12-14 cm in superior-inferior dimension in an adult.
•
Diameter of AP spleen more than two-thirds the abdominal diameter.
•
Causes of splenomegaly can be divided according to the degree of sple-
nomegaly they cause.
•
Huge spleen:
• malaria.
• kala azar, leischmaniasis, syphilis.
• extramedullary haemopoiesis (e.g. myelofibrosis).
• chronic myeloid leukaemia.
• lymphoma.
• Gaucher’s disease.
•
Moderately large spleen:
• glycogen storage diseases, Gaucher disease, Neimann-Pick disease.
• haemolytic anaemias, haemochromatosis, sickle cell disease (early on
in sequestration crises).
• portal hypertension, splenic vein thrombosis.
• causes of huge spleen, in less advanced stages.
• leukaemia.
•
Borderline splenomegaly:
• sarcoidosis.
• amyloidosis.
• rheumatoid arthritis (Felty syndrome).
• systemic lupus erythematosus.
• haemodialysis.
• viral infections: hepatitis, glandular fever.
• bacterial infections: brucellosis, typhoid, tuberculosis, generalised
bacterial septicaemia.
• rickettsial infections: typhus.
• fungal infections: histoplasmosis.
•
Neoplasms of the spleen such as secondaries and angiosarcoma generally
replace splenic tissue rather than cause splenomegaly.
•
NB: sickle cell disease causes splenomegaly early on in the disease, followed
318
by auto-splenectomy following infarction, leading to a small spleen.
S
Splenomegaly in a patient with portal hypertension. The lower
pole of the spleen (arrowheads) clearly extends below the lower costal
margin. Furthermore, there is a generalised ‘haze’ over the abdomen,
caused by ascites, and thickening of valvulae conniventes secondary to
fold oedema.
Clinical characteristics
• Splenomegaly can often be detected on clinical examination, if mod-
erate to large.
• May present with dull LUQ pain.
• Presentation is more often with features of the underlying disease
process.
• If the spleen is very large, presentation can be with obstructive symp-
toms such as gastric obstruction or left renal hydronephrosis.
• An enlarged spleen is also at increased risk of infarction and trauma.
319
Radiological features
S
•
AXR:
• Enlarged spleen is often seen as large soft tissue density mass in the
LUQ.
• Spleen extends downwards and medially towards centre of abdomen.
• Enlarged spleen may be inferred from displacement of gas-filled small
bowel loops.
• May cause left renal hydronephrosis on IVU.
•
USS:
• Imaging modality of choice in initial confirmation of splenomegaly.
• Usually homogeneous texture on US.
• Coarse spleen, with similar echo properties to liver in haemochro-
matosis.
•
CT and MRI:
• Splenomegaly is easily identified on both CT and MRI, especially if
multiplanar reconstructions are used.
• Early arterial enhancement of spleen leads to patchy contrast
enhancement on CECT and can be mistaken for pathology.
• Associated pathology is often identified, for example portal varices
and cirrhotic liver in portal hypertension or a high-attenuation spleen
on NECT in haemochromatosis (from iron deposition).
320
S
Massive splenomegaly (asterisk) axial CECT.
321
Massive splenomegaly (asterisk) coronal reformat CECT.
T
Testicular neoplasms
•
Testicular neoplasms form 1-2 % of all male cancers.
•
Peak age of occurrence is 25-35 years and it is the most common
malignancy in age group 15-35 years.
•
Risk factors are Caucasian origin, testicular maldescent and positive family
history.
•
Metastases occur to liver, lung, brain and bones. Nodal metastases are
usually unilateral; bilateral in 8%.
•
The contralateral testis is at increased risk of developing a metachronous
tumour.
•
Most (95%) testicular neoplasms are germ cell tumours:
• seminoma (40%): very radiosensitive with a good prognosis.
• embryonal carcinoma (10%): more aggressive than seminomas.
• teratoma (10%): occur in a younger age group (10-20 years) and have
good prognosis.
• choriocarcinoma (1%): most aggressive form.
• mixed tumours: teratocarcinoma is the most common type; may
undergo spontaneous regression.
•
Sex cord-stromal tumours are usually benign and hormonally active.
•
Metastases: common primary sites include prostate, lung, kidney, GI
tract and melanoma. Metastases from prostatic carcinoma account for
75% of bilateral metastases to the testis. Routes of spread include
haematogenous, lymphatic, retrograde extension from the vas deferens
or direct invasion from an adjacent mass.
Clinical characteristics
• Chronic pain, sensation of heaviness.
• Acute scrotal pain in 10%, from intratumoural haemorrhage.
• Enlarging testicular mass.
• Secondary hormonal effects: gynaecomastia, virilisation.
Radiological features
• USS:
• Sensitivity of 95% for the detection of testicular tumours.
• Seminomas tend to be hypoechoic; the remainder are heterogeneous
in echotexture.
• Metastases usually present as multiple, bilateral hypoechoic masses.
• Lymphoma presents as diffuse or multifocal testicular enlargement.
• CECT: used for staging the disease.
322
T
Testicular neoplasm. Well-defined hypoechoic tumour within the
right testis. Normal left testis.
323
T
Transitional cell carcinoma
Clinical characteristics
• Approximately 85% of urothelial malignancies are transitional cell car-
cinomas (TCC).
• Risk factors include:
• smoking.
• aniline dyes.
• chronic urinary infection.
• analgesic abuse.
• cyclophosphamide therapy.
• high ingestion of coffee.
• Bladder involvement is much more common than the upper tracts.
• Synchronous and metachronous tumours relatively common.
• Symptoms include haematuria, flank pain and renal colic if obstruction
occurs.
• Spread is most commonly to local lymph nodes, the peritoneum or
liver.
Radiological features
• Because synchronous tumours are relatively common, the entire renal
tract should be investigated radiologically or urologically.
Bladder
Radiological features
• IVU: shows irregular filling defect; may have a sessile appearance.
• CECT and USS: both demonstrate focal mural thickening and
an exophytic mass projecting into the bladder lumen or within a
diverticulum.
• MRI:
• Most accurate for staging of bladder TCC.
• TCC are isointense on T1W sequences and hyperintense on T2W.
• TCC show early enhancement post-gadolinium injection, allowing
accurate assessment of bladder wall involvement.
324
T
Transitional cell carcinoma of the bladder. US shows solid
exophytic carcinoma within a bladder diverticulum (arrowhead).
Transitional cell carcinoma of the bladder. Corresponding CT
demonstrates the solid enhancing mass within the diverticulum
(arrowhead).
325
T
Kidneys
Radiological features
•
IVU:
• Occasionally punctate calcification may be seen on the control film.
• Sessile filling defects can be seen within the collecting systems.
Contrast trapped within the intersitium of a papilliform tumour
gives rise to a stippled appearance.
• Non-opacification of a calyx occurs when obstructed by tumour.
Renal obstruction may be from tumour at the pelviureteric junction.
This may result in renal atrophy.
• Tumour infiltrating the renal parenchyma results in decreased con-
trast excretion, with preservation of the normal renal outline; this is
often not the case in renal cell carcinoma.
•
USS:
• Tumour is usually isoechoic or hyperechoic in comparison with renal
parenchyma.
• Depending on tumour location, calyceal dilatation, with or without
renal pelvis dilatation, may be seen.
• Renal infiltration with preservation of renal outline may be seen.
•
CECT:
• CT urography is an excretory phase CT of the renal tract and is
increasingly replacing IVU. This results in opacification of the pelvi-
calyceal systems and ureters, allowing assessment of filling defects as
in IVU.
• Mass centred on the collecting system shows variable enhancement.
Associated thickening of collecting system wall may be visible.
• There may be punctuate calcification.
• Renal infiltration can be demonstrated.
• CECT is commonly used as a staging study.
•
MRI:
• MR urography, either using heavily T2W images, to utilise the hyper-
intensity of urine, or heavily T1W delayed images, post-gadolinium,
can give similar images to IVU but without ionizing radiation.
• TCC may show heterogeneous gadolinium enhancement, but usu-
ally less than the renal parenchyma.
326
T
Transitional cell carcinoma of the bladder. Film from an IVU series
shows an ill-defined filling defect (arrows) within the bladder secondary to
the carcinoma.
Transitional cell carcinoma of the kidney. Multiple carcinomas create
irregular filling defects within the left renal pelvis and upper ureter (arrows).
327
Transitional cell carcinoma of the kidney. Enhancing mass within
the right renal pelvis (arrow).
T
Ureters
Radiological features
• IVU:
• Variable number of irregular intraluminal filling defects can be seen.
• Irregular luminal stricturing.
• Variable degrees of obstruction can lead to hydroureter, hydroneph-
rosis or a non-functioning kidney.
• USS: may demonstrate features of renal tract obstruction but the ureter,
particularly the mid ureter, is often poorly visualised using this modality.
• CECT: intraluminal soft tissue mass can be seen. Local extension into
periureteric fat and beyond may be demonstrated.
• MRI: MR urography can again replicate the IVU findings, but addi-
tional sequences are required to stage the tumour.
328
T
Ureteric transitional cell carcinoma. Obstructing carcinoma of the
lower right ureter (arrowhead) with secondary hydronephrosis.
329
Tuberculosis of the abdomen
T
and pelvis
Clinical characteristics
• Caused by Mycobacterium tuberculosis, an acid-alkali-fast bacillus.
• Infections are most commonly pulmonary, but any organ system in the
body may be affected.
• Increasing incidence in the developed world as a result of increased
immigration from endemic areas and increased numbers of immuno-
compromised individuals.
• Presentation depends on affected organ systems. Commonly, there are
pulmonary symptoms including chronic cough and haemoptysis.
• Non specific symptoms such as weight loss, fevers and night sweats are
characteristic.
• Abdominal symptoms will include the above non-specific signs, with
abdominal pain, nausea and vomiting. Urinary tract involvement man-
ifests as non-specific symptoms as well as dysuria, haematuria and sterile
pyuria.
Abdominal tuberculous lymphadenopathy
Clinical characteristics
• Multiple lymph node groups are affected simultaneously, most com-
monly mesenteric and peri-pancreatic groups.
• The main differential is lymphoma, which does not tend to undergo
necrosis.
Radiological features
• In 40-70% of patients with lymphadenitis, there are enlarged nodes
with hypodense centres and peripheral hyperdense enhancing rims,
characteristic, but not pathognomic, of caseous necrosis.
• Other nodal patterns include conglomerate mixed density masses,
enlarged homogeneous-density nodes, and an increased number (>3)
of normal or mildly enlarged homogeneous nodes.
330
T
Tubercular lymphadenopathy. Caseous low-attenuation tuberculous
node (arrowheads).
Peritoneal tuberculosis (wet type). This demonstrates cystic loculated
fluid collections within the small bowel mesentery (arrows).
331
T
Tuberculous peritonitis
Clinical characteristics
• Usually occurs secondary to haematogenous spread or from the rupture
of a tuberculous lymph node.
Radiological features
• There are three types.
• Wet: most common, featuring large amounts of free loculated fluid
with a high protein density (20-45 HU).
• Fibrotic: large omental and mesenteric cake-like masses with matting
of bowel loops.
• Dry: mesenteric thickening, fibrous adhesions and caseous nodules.
• Varying degrees of omental and mesenteric involvement are seen.
• Omentum appears smudged, caked or thickened in equal frequency.
• CT: findings in TB peritonitis are non-specific, with disseminated
peritoneal malignancy, non-TB peritonitis and mesothelioma as differ-
ential diagnoses.
• One complication is small bowel obstruction, and evidence may be seen
on imaging.
Gastrointestinal tuberculosis
Clinical characteristics
• TB in the GI tract is rare, but when present usually (90%) involves the
ileocaecal region and/or right side of colon. The gastroduodenal
regions are affected less often. The oesophagus is the least common
GI location affected by TB.
Radiological features
• Skip areas of concentric mural thickening with luminal narrowing,
sometimes with proximal dilatation elsewhere in the small bowel, and
in the presence of ileocaecal involvement strongly suggests TB.
• Mucosal ulceration is a common feature. Fistulae may occur.
• CT:
• The most common CT finding is mural thickening, which is mostly
concentric.
• Commonly there is focal mural thickening involving the ileocaecal
valve and medial aspect of the caecum.
• Associated evidence of TB, such as necrotic lymphadenitis, may be seen.
332
T
Peritoneal tuberculosis (fibrotic type). Omental caking is
demonstrated (arrowheads) with thickening of the underlying small
bowel (asterisk).
Ileocaecal tuberculosis (barium enema). Marked retraction of the
333
ileocaecal area can be seen, with an incompetent ileocaecal valve.
• Barium studies:
T
• The terminal ileum may appear rigid with proximal dilatation. The
valve itself may be enlarged.
• The caecum may appear contracted owing to fibrosis and spasm. The
affected ileal segments tend to be shorter than those in Crohn’s disease
and cobble-stoning is not seen in TB.
• Gastroduodenal TB can result in pyloric stenosis, deep ulceration and
rugal hypertrophy as a result of the infiltration.
• The differential, on barium studies, includes inflammatory bowel
disease, lymphoma, carcinoma or other infections.
Hepatosplenic tuberculosis
Clinical characteristics
• Usually caused by haematogenous spread as part of miliary TB.
• Hepatosplenic involvement is categorised as:
• micronodular or miliary: characterised by innumerable nodules,
diameter 0.5-2.0mm (which may not be detected by CT)
• macronodular: associated with tuberculoma dissemination and very
rare.
Radiological features
• CT:
• When TB is present, CT shows an enlarged liver and spleen contain-
ing multiple non-specific, low-attenuation lesions.
• The presence of calcified granulomata on CT, in the absence of a
known primary tumour, in patients with known risk factors should
raise suspicion of TB. Calcified granulomata may represent old,
healed TB lesions.
334
T
Hepatosplenic tuberculosis. Multiple calcified granulomata are seen
within the liver, spleen, portal and peripancreatic nodes. The right kidney is
hydronephrotic and a small calculus is seen within the collecting system.
335
Adrenal tuberculosis. Bilateral adrenal enlargement (arrows).
T
Adrenal tuberculosis
Clinical characteristics
• Adrenal involvement is seen in up to 6% of patients with active TB.
• These patients almost always present with bilateral adrenal involvement
and an Addisonian type clinical picture.
Radiological features
• The CT signs of active tuberculous adrenalitis are enlarged glands
associated with large hypodense, necrotic areas, with or without dot-
like calcification.
• Old TB lesions can result in dense adrenal calcification visible on CT or
AXR.
Genitourinary tuberculosis
Clinical characteristics
• Genitourinary TB is the most common clinical manifestation of extrap-
ulmonary tuberculosis.
• M. tuberculosis reaches the prostate, seminal vesicles and, particularly, the
kidneys by hematogenous route from the lungs.
• All other genital organs, including the epididymis and bladder, become
involved by direct extension of infection.
• Clinical clues include history of TB, sterile pyuria, haematuria, fre-
quency and dysuria.
336
T
Tuberculosis. (film from IVU series). Infundibular strictures (arrows)
within the lower pole of the right kidney, with associated calycectasis.
337
Tuberculous prostatic abscess (arrowhead).
Radiological features
T
•
Renal TB involvement is unilateral in 75%.
•
The earliest urographic abnormality is a ‘moth-eaten’ calyx caused by
erosion. It may resemble papillary necrosis but the latter is more often
bilateral and symmetrical.
•
Renal parenchymal cavitation may be detected as irregular pools of
contrast material.
•
Dilated calyces with related infundibular stricture at one or more sites
within the collecting system may be seen.
•
Characteristic calcifications in a lobar distribution are often seen in end-
stage TB: known as TB autonephrectomy.
•
Ureteric manifestations, characterised by thickened ureteric wall or
strictures, occur in almost half of all those with renal TB, involvement
being most common in the distal third of the ureter. CT demonstrates
focal ureteric mural thickening and inflammatory change in the peri-
ureteric fat.
•
Sequential bladder involvement is manifested as interstitial cystitis with
wall thickening, ulceration and eventual scarring, with long-term loss of
cystic volume.
•
Genital tract TB almost always involves the fallopian tubes in women,
usually causing bilateral salpingitis. Tubo-ovarian abscesses may be seen
on US, CT or MRI but are non-specific.
•
Male involvement is confined to the seminal vesicles or prostate, which
are occasionally calcified.
•
CECT shows hypoattenuating prostatic lesions, which likely represent
foci of caseous necrosis and inflammation. However non-tuberculous
pyogenic prostatic abscesses have a similar CT appearance.
Musculoskeletal tuberculosis
• Musculoskeletal manifestation of TB may be identified. The most
common ones are infective discitis and psoas abscesses.
338
T
Renal tuberculosis. Intravenous urogram demonstrating a densely
calcified, non-enhancing, small right kidney. The appearances are
characteristic of long-standing renal tuberculosis; so-called tuberculosis
autonephrectomy. The left kidney (not shown) enhances normally.
339
U
Uterine fibroids
Clinical characteristics
•
Fibroids (uterine leiomyomata) are the most common gynaecological
tumour and are present in up to 50% of women over 40 years of age.
•
Fibroids are benign tumours composed of smooth muscle and fibrous
tissue that are hormone dependent, enlarging during pregnancy and
hormone-replacement therapy.
•
They usually present well after puberty and shrink after the menopause.
•
The clinical presentation depends on the position and size of the
fibroids:
• intramural fibroids are the most common.
• submucosal fibroids are least common; they lie adjacent to the uterine
cavity are most likely to cause symptoms such as menorrrhagia and
dysmenorrhoea by enlarging and distorting the uterine cavity.
• a third, subserosal group, lie on the outer uterine surface.
•
Symptoms and complications of fibroids include:
• pain.
• abnormal bleeding.
• torsion of pedunculated subserosal fibroids.
• subfertility.
• problems in pregnancy and labour, such as spontaneous abortion,
premature and obstructed labour.
• sarcomatous degeneration of fibroids (rare: < 1%).
Radiological features
• AXR:
• Not a modality of choice, but calcified fibroids may be seen in the
pelvis.
• Circumferential calcification tends to follow pregnancy; punctate
calcification is usually postmenopausal.
340
U
Calcified Fibroids. Two examples within the pelvis (arrowheads).
341
•
Hysterosalpingogram:
U
• Performed to investigate infertility.
• Submucosal and larger intramural fibroids will be seen as smooth
filling defects outlined by contrast in the inverted triangular-shaped
endometrial cavity.
•
USS:
• The echogenicity of fibroids is variable on US, with areas of calcifi-
cation as described above.
• Fatty, haemorrhagic and cystic areas may also be seen.
• The uterus can be focally or diffusely enlarged, with individual
fibroids measuring a few millimetres to over 20cm.
• The uterus may have an irregular outline if there are subserosal
fibroids. Large fibroids may compress the ureters, and it is important
to check for the presence of hydronephrosis.
•
CT: not particularly helpful at diagnosing fibroids other than detecting
uterine enlargement, as fibroids have the same density and contrast
enhancement as the uterine myometrium, unless calcified.
•
MRI:
• MRI provides excellent visualisation of the uterus and is used to
demonstrate the exact size and location of fibroids, which are of
lower signal intensity than the myometrium on T2W sequences.
• Approximately one-third of fibroids have a high-signal rim on T2W
images from peritumoral oedema, lymphatics or veins.
• Fibroids may degenerate as they enlarge, resulting in heterogeneous
signal.
• Contrast-enhanced MRI is obtained if information is required about
the vascular supply of the fibroids, such as prior to uterine artery
embolisation (see below).
• Advantages of MRI are the ability to delineate the relationship of
fibroids to the endometrial lining when planning surgery, and to
distinguish adenomyosis from fibroids.
• Adenomyosis is the presence of ectopic endometrium in the myome-
trium, which causes similar symptoms to fibroids. The diagnosis is
made on MRI if the junctional zone (the inner myometrium) is >11 mm
thick.
342
U
Large intramural fibroid (calipers) displacing the uterine body (arrow).
Intramural fibroid. Sagittal T2W MRI of the pelvis demonstrating a
343
hypointense intramural fibroid (arrow). The endometrial cavity (E) is of
uniform hyperintensity.
Radiological management
U
•
Medical treatment is only a temporary measure because hormonal
manipulation may shrink fibroids but it will not remove them. Hence
it is used only in patients awaiting surgery or who are close to the
menopause.
•
Surgical options include myomectomy and hysterectomy, which can be
performed by laparotomy or by operative hysteroscopy. Myomectomy
is performed to conserve fertility whereas hysterectomy is the definitive
treatment of fibroids.
•
Interventional radiological techniques include uterine artery embolisa-
tion, where the uterine artery is catheterised via a percutaneous femoral
artery approach, and bilateral embolisation of the uterine arteries is
performed with polyvinyl alcohol foam particles. Reasons for proce-
dure failure include an aberrant arterial supply to the uterus, incomplete
embolisation, very large fibroids or a coexisting disorder such as
adenomyosis.
•
Newer techniques include MRI-guided focused US and laser ablation,
which both use heat to cause tissue necrosis within the fibroids. The
former uses thermal heating by US and the latter uses a percutaneous
approach to insert laser fibres into the fibroids. In both cases the heating
of the fibroid tissue is monitored by detecting changes in MRI sequen-
ces to create a real-time thermal map.
344
U
Subserosal fibroid. Sagittal T2W MRI of the pelvis demonstrating a huge
degenerate, heterogeneous, subserosal fibroid (asterisk). Arrowhead
indicates the endometrial cavity; UB, uterine body.
345
V
Volvulus
• Volvulus results from torsion along the mesenteric axis of a segment of
the alimentary tract.
• It produces partial or complete intestinal obstruction.
• The ensuing ischaemia results in gangrene and perforation.
• The sigmoid colon is most commonly affected, followed by the cae-
cum, transverse colon and stomach.
• Predisposing factors include redundant bowel loops (e.g. in the chroni-
cally constipated), an elongated mesentery, malrotation and chronic
colonic distension.
Clinical characteristics
• Colonic volvulus presents with features of bowel obstruction: abdomi-
nal pain and distension, vomiting and constipation. In gastric volvulus,
there is severe epigastric pain, vigorous attempts to vomit, with little
result, and an inability to pass a nasogastric tube.
• The duration, type and severity of symptoms depend upon the location
of the obstruction, i.e. location of volvulus.
Gastric volvulus
Clinical characteristics
• Often occurs as a complication of a hiatus hernia
• Two types described:
• organo-axial rotation about a line extending from cardia to pylorus
• mesentero-axial rotation around an axis extending from the lesser to
greater curvature.
• Complications include intramural emphysema and gastric perforation.
Radiological features
• AXR: shows massively distended stomach in LUQ.
• Barium swallow/meal: shows incomplete or absent passage of con-
trast into the stomach.
346
V
Gastric volvulus. In a large hiatus hernia on CT.
Caecal volvulus. Gas-filled distended caecum rotates towards the left
upper quadrant.
347
V
Caecal volvulus
Clinical characteristics
• Predisposing factors are malrotation and a congenitally long mesentery.
• Peak age is 20-40 years, more commonly in males.
Radiological features
• AXR: ‘kidney-shaped’ distended caecum rotates centrally towards the
LUQ.
• Instant enema: the tapered end of the barium column points towards
the torsion.
Sigmoid volvulus
Clinical characteristics
• Typically occurs in elderly constipated patients. The sigmoid colon
twists on its mesenteric axis.
Radiological features
• AXR:
• Greatly distended loop, with fluid-fluid levels, mainly on the left side,
extending towards diaphragm.
• Produces a typical ‘coffee bean sign’, with a distinct midline crease,
representing the mesenteric root, surrounded by a gaseous distended loop.
• Associated with proximal bowel obstruction.
• Instant enema: ‘bird of prey sign’: tapered hook-like end can be seen
on the barium column.
• CT: tightly torted mesentery, produced by twisted afferent and efferent
loops, produces the so-called ‘swirl sign’.
348
V
Classic sigmoid volvulus. ‘Coffee bean’ sign.
Sigmoid volvulus. The torted segment (arrow) and faecal loading within
the proximal dilated sigmoid are clearly demonstrated on CT.
349
V
Small-bowel volvulus
Clinical characteristics
• Usually occurs in the ileum and is related to the presence of congenital
bands or adhesions.
Radiological features
• AXR: proximal small-bowel dilatation is seen.
• CT:
• U-shaped configuration of distended and fluid-filled loops of small
bowel converge towards the point of torsion.
• Tightly twisted mesentery can be seen around the point of torsion
(‘whirl sign’).
• There are fusiform tapering loops.
• There may be signs of bowel ischaemia or infarction.
350
V
Small-bowel volvulus. Closed loop obstruction and dilated loops
converging on the point of torsion (arrow).
351