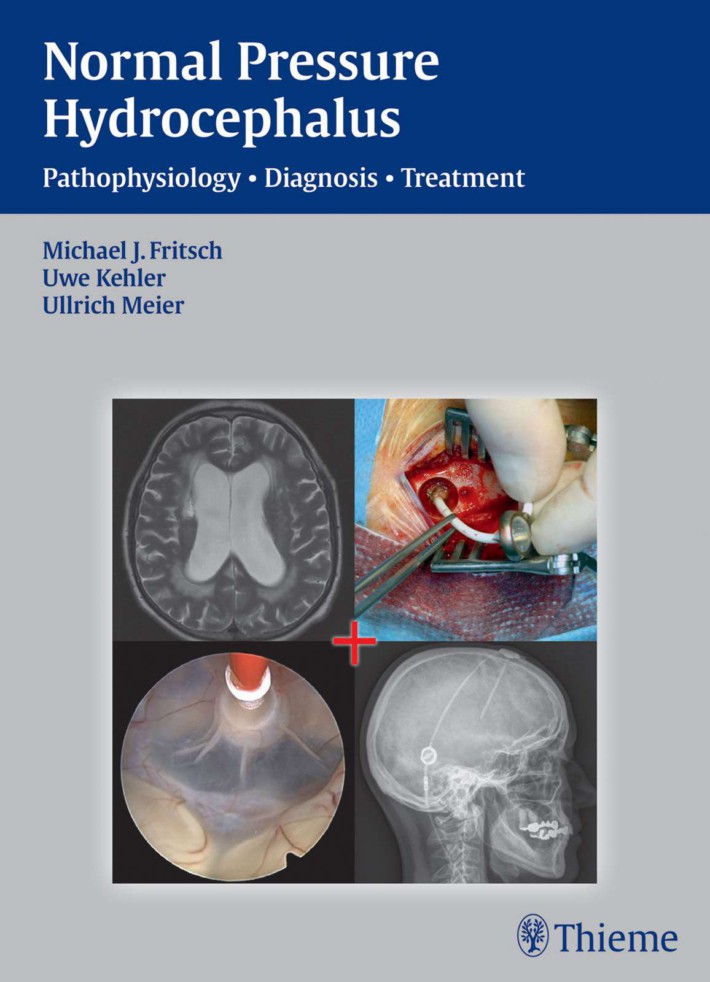
Normal Pressure Hydrocephalus
Pathophysiology • Diagnosis • Treatment
Michael J. Fritsch, MD, PhD
Chairman
Department of Neurosurgery
Dietrich-Bonhoeffer-Klinikum
Neubrandenburg, Germany
Uwe Kehler, MD, PhD
Professor and Chairman
Department of Neurosurgery
Asklepios Klinik Altona
Hamburg, Germany
Ullrich Meier, MD, PhD
Professor and Chairman
Department of Neurosurgery
Unfallkrankenhaus Berlin
Berlin, Germany
With contributions by
Johannes Lemcke, Christoph Miethke
144 illustrations
Thieme
Stuttgart New York Delhi Rio
Library of Congress Data
Fritsch, Michael J., author.
Important note: Medicine is an ever-changing science
Normal pressure hydrocephalus : pathophysiology,
undergoing continual development. Research and clinical
diagnosis, treatment / Michael J. Fritsch, Uwe Kehler,
experience are continually expanding our knowledge, in
Ullrich Meier ; with the collaboration of Johannes Lemcke,
particular our knowledge of proper treatment and drug
Christoph Miethke.
therapy. Insofar as this book mentions any dosage or
p. ; cm.
application, readers may rest assured that the authors,
Includes bibliographical references.
editors, and publishers have made every effort to ensure
ISBN 978-3-13-164601-9 (hardback)
that such references are in accordance with the state of
I. Kehler, Uwe, author. II. Meier, Ullrich, author.
knowledge at the time of production of the book.
III. Lemcke, Johannes (Neurosurgeon), author.
Nevertheless, this does not involve, imply, or express any
IV. Miethke, Christoph, author. V. Title.
guarantee or responsibility on the part of the publishers in
[DNLM: 1. Hydrocephalus, Normal
respect to any dosage instructions and forms of applications
Pressure-physiopathology. 2. Hydrocephalus, Normal
stated in the book. Every user is requested to examine
Pressure-diagnosis. 3. Hydrocephalus, Normal
carefully the manufacturers’ leaflets accompanying each
Pressure-therapy. WL 203]
drug and to check, if necessary in consultation with a
RC391
physician or specialist, whether the dosage schedules men-
616.85'8843-dc23
tioned therein or the contraindications stated by the man-
2013041564
ufacturers differ from the statements made in the present
book. Such examination is particularly important with
Author C. Miethke is founder, CEO and part-owner
drugs that are either rarely used or have been newly
of Christoph Miethke GmbH & Co., developer and
released on the market. Every dosage schedule or every
manufacturer of some of the products presented in
form of application used is entirely at the user’s own risk
and responsibility. The authors and publishers request
this book.
every user to report to the publishers any discrepancies
or in accuracies noticed.
If errors in this work are found after publication, errata
description page.
© 2014 Georg Thieme Verlag KG
Thieme Publishers Stuttgart
Rüdigerstrasse 14, 70469 Stuttgart, Germany,
þ49 [0]711 8931 421
Thieme Publishers New York
333 Seventh Avenue, New York, NY 10001 USA,
1-800-782-3488
Some of the product names, patents, and registered
Thieme Publishers Delhi
designs referred to in this book are infact registered
A-12, Second Floor, Sector -2, NOIDA -201301,
trademarks or proprietary names even though specific
Uttar Pradesh, India, +91 120 45 566 00
reference to this fact is not always made in the text.
Therefore, the appearance of a name without designation
Thieme Publishers Rio, Thieme Publicações Ltda.
as proprietary is not to be construed as a representation
Argentina Building 16th floor, Ala A, 228 Praia do Botafogo
by the publisher that it is in the public domain.
Rio de Janeiro 22250-040 Brazil, þ55 21 3736-3631
This book, including all parts thereof, is legally protected
Cover design: Thieme Publishing Group
by copyright. Any use, exploitation, or commercialization
Typesetting by Thomson Digital, India
outside the narrow limits set by copyright legislation,
Printed in China by Everbest Printing co. Ltd.
without the publisher’s consent, is illegal and liable to
ISBN 978-3-13-164601-9
prosecution. This applies in particular to photostat repro-
Also available as e-book:
duction, copying, mimeographing, preparation of micro-
eISBN 978-3-13-164611-8
films, and electronic data processing and storage.
Contributors
Michael J. Fritsch, MD, PhD
Uwe Kehler, MD, PhD
Chairman
Professor and Chairman
Department of Neurosurgery
Department of Neurosurgery
Dietrich-Bonhoeffer-Klinikum
Asklepios Klinik Altona
Neubrandenburg, Germany
Hamburg, Germany
Johannes Lemcke, MD
Ullrich Meier, MD, PhD
Senior Neurosurgeon
Professor and Chairman
Department of Neurosurgery
Department of Neurosurgery
Unfallkrankenhaus Berlin
Unfallkrankenhaus Berlin
Berlin, Germany
Berlin, Germany
Christoph Miethke, MEng
President and Director of Research and
Development
Christoph Miethke GmbH & Co. KG
Potsdam, Germany
V
Preface
developments. In Thieme Publishers, we have found
How This Book Came To Be
apartner with whomwe could carryout thisproject;
The beginnings of this book go back to a course that
the result is the book that you now hold in your
the authors held on the diagnosis and therapy of
hands.
normal pressure hydrocephalus (NPH).
A first meeting to prepare for this course took
What Unites Us
place in 2008. At that time, we were of the opinion
that the clinical picture of idiopathic NPH was under-
The authors are united in their extraordinary clinical
represented, both in the neurosurgical literature and
and scientific interest in hydrocephalus, in particular
in the awareness of colleagues and the public. It was
NPH. We view this topic from different perspectives:
our intention to change this situation.
one from the perspective of developer and engineer
At the beginning there was skepticism. Most of the
(C.M.) and the other from the perspective of the
neurosurgical courses that are carried out today are
practitioners (M.J.F., U.K., J.L., U.M.). This productive
done with an emphasis on hands-on experience. The
cooperation over many years, and the close dialog
question was: Who would be interested in NPH as a
with respect to product development (what do our
disease, and in the surgical techniques for implanting
patients need?) and clinical use (what treatment
shunts? Would we even have enough participants to
results can we expect and attain?), is a notable
justify such a course?
strength of this author group and we are convinced
After appropriate preparation, a first course was
that this cooperation will result in further improve-
organized in 2009, with the support of the Aesculap
ment in the treatment of patients with hydrocepha-
Academy in Berlin. The course was quickly booked out,
lus. The cooperatively published SVASONA study is
and was successful with respect to its contents and
just one example of this.
organization. After a repetition in 2010, we began to
Finally, we wrote this book to document the coop-
hold two courses annually in 2011 in Berlin, with
eration within our own group, to strengthen it, and to
guests and participants from Europe and the USA;
provide impetus for further progress. We would
and in Bangkok, with participants from Asia and the
enjoy hearing from readers with criticism, tips, and
Pacific. The courses remain well attended; the listeners
ideas, and we are committed to maintaining the
are highly interested and the discussions are lively.
cooperation within the author group and beyond
Following successful establishment of the courses,
in the coming years.
the question arose as to what sort of accompanying
literature could be provided to research and review
Michael J. Fritsch
the disease and its diagnosis and therapy. In answer-
Uwe Kehler
ing this question, the idea formed to write a book on
Johannes Lemcke
the topic that would cover the scientific studies, our
Christoph Miethke
clinical experience, and the newest technological
Ullrich Meier
VI
Contents
1
Introduction
2
Michael J. Fritsch
1.1
Why a Book about NPH?
2
2
Epidemiology of Idiopathic Normal Pressure Hydrocephalus
6
Michael J. Fritsch
2.1
Epidemiologic Studies
6
2.1.7
Tanaka et al (2009)
7
2.1.1
Trenkwalder et al (1995)
6
2.1.8
Iseki et al (2009)
7
2.1.2
Tisell et al (2005)
6
2.1.9
Klassen and Ahlskog (2011)
7
2.1.3
Marmarou et al (2007)
6
2.1.4
Hiraoka et al (2008)
6
2.2
Discussion
7
2.1.5
Brean and Eide (2008)
6
2.1.6
Brean et al (2009)
7
2.3
Summary
8
3
History
10
Michael J. Fritsch
3.1
Early History
10
3.4
Hakim and the Discovery of Normal
Pressure Hydrocephalus
12
3.2
Hydrocephalus and its Treatment in
the 19th and 20th Centuries
10
3.5
Modern Shunt Technology and
Perspectives
13
3.3
Development of Modern Shunt
Concepts
11
4
Clinical Characteristics and Differential Diagnosis
16
Uwe Kehler
4.1
Natural History
16
4.3.2
Parkinson Disease
19
4.3.3
Alzheimer Disease
20
4.2
Symptoms of iNPH
16
4.3.4
Binswanger Disease (Subcortical Vascular
4.2.1
Main Symptoms
16
Dementia)
20
4.2.2
Other Symptoms
17
4.3.5
Brain Atrophy
20
4.3.6
Cervical Myelopathy
21
4.3
Differential Diagnosis
18
4.3.7
Lumbar Spinal Canal Stenosis
21
4.3.1
Secondary NPH and Chronic Obstructive
Hydrocephalus
18
4.4
Summary
21
5
Pathophysiology
24
Johannes Lemcke, Ullrich Meier
5.1
What Causes iNPH? Theories Proposed
5.2
Inner Contradictions
25
by the Discoverers and the Bulk Flow
Theory
24
VII
Contents
6
Noninvasive Diagnostic Work-up
28
Uwe Kehler
6.1
Evaluation of the Patient’s
6.2.2
Evaluating Incontinence
29
Medical History
28
6.2.3
Neuropsychological Testing
29
6.2
Clinical Examination
28
6.3
Summary
33
6.2.1
Evaluating Gait Disturbance
28
7
Imaging
36
Johannes Lemcke
7.1
Computed Tomography
36
7.6
Multifrequency Magnetic Resonance
Elastography
39
7.2
Subjective Assessment of Ventricular
Size and Shape
36
7.7
Direct Postoperative Imaging
39
7.3
Indices
36
7.8
Imaging for Routine Follow-Up
39
7.3.1
Evans Index
36
7.3.2
Other Indices
36
7.9
Native Radiography/Valve Setting
39
7.3.3
Third Ventricle Diameter
37
7.10
Using Imaging for Troubleshooting
41
7.4
MRI/Phase-Contrast MRI
38
7.10.1
Overdrainage
41
7.10.2
Underdrainage
42
7.5
Diffusion Tensor Imaging
38
8
Invasive Diagnostic Work-up
46
Johannes Lemcke, Ullrich Meier
8.1
Intracranial Pressure Monitoring
46
8.3
CSF Tap Test
51
8.2
Lumbar Infusion Test
46
8.4
External Lumbar Drainage
52
8.2.1
Outflow Resistance
47
8.2.2
Calculation Procedure
48
8.5
CSF and Serum Biochemical Tests
52
8.2.3
Dynamic Infusion Test Within the
Investigation Chain
50
8.6
Diagnostic Pathway
53
9
Shunt and Valve Technology
58
Christoph Miethke
9.1
Definition
58
9.6
Ventriculoatrial Shunts
61
9.2
Types
58
9.7
Classification of Valves
61
9.3
Physical Basics
58
9.8
Adjustable Differential Pressure
Valves
64
9.4
Hydrostatic Pressure
58
9.9
Hydrostatic Devices
67
9.5
Physics of Ventriculoperitoneal
9.9.1
Adjustable Hydrostatic Devices
77
Shunts
59
VIII
Contents
10
Shunt and Valve Settings
86
Michael J. Fritsch, Uwe Kehler, Johannes Lemcke, Ullrich Meier
10.1
Shunt Settings
86
10.2.7
The “Optimal” Shunt Setting
89
According to M.F.
10.3
Shunt Settings
89
10.2
Shunt Settings
86
According to U.M. and J.L.
According to U.K.
10.3.1
Why Use Gravitational Valves?
89
10.2.1
Ventriculoatrial versus
10.3.2
Does Valve Opening Pressure of
Ventriculoperitoneal Shunt
86
Hydrostatic Valves Have an Influence
10.2.2
Frontal versus Parietal/Occipital
on the Course of the Disease?
90
Bore Hole
87
10.3.3
Conclusion regarding Clinical
10.2.3
Selection of Valve Opening Pressure
88
Practice
91
10.2.4
Should the Valve and/or the Gravitational
Device be Programmable?
88
10.4
Efficiency of Gravitational Valves
91
10.2.5
Shunt Configuration (with or without a
Cerebrospinal Fluid Reservoir)
88
10.4.1
Conclusion
92
10.2.6
Selecting Catheter Material
89
11
Surgical Technique
94
Michael J. Fritsch
11.1
Settings in the Operating Room
94
11.5.1
Ventricular Catheter
95
11.5.2
Valve
96
11.2
Positioning
94
11.5.3
Peritoneal Catheter
98
11.3
Shaving and Disinfection
94
11.6
Alternatives to the VP Shunt
99
11.4
Draping
94
11.5
Surgical Procedure
95
12
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
102
Uwe Kehler
12.1
Rationale for ETV in Communicating
12.2.3
Surgical Technique
104
Hydrocephalus and NPH
102
12.3
Complications
106
12.1.1
ETV in Shunt Failure
103
12.4
Outcomes
107
12.2
Technique
103
12.2.1
Preoperative Planning
103
12.5
Summary
107
12.2.2
Positioning of the Patient
104
13
Scales and Scores
110
Ullrich Meier
13.1
Black Grading Scale
110
13.5
NPH Recovery Rate
111
13.5.1
Conclusion
112
13.2
Index for Postoperative Improvement .
110
13.6
Comorbidity Index
112
13.3
Stein-Langfitt Scale
111
13.4
Kiefer Grading Scale
111
IX
Contents
14
Follow-up Management of Idiopathic Normal Pressure Hydrocephalus
116
Ullrich Meier
14.1
Organizing Follow-up Examinations . . .
116
14.2.2
Six Months
116
14.2.3
Twelve Months
116
14.2
Follow-up Intervals
116
14.2.1
One/Three Months
116
14.3
Should Follow-ups End?
117
15
Complications
120
Michael J. Fritsch, Uwe Kehler, Johannes Lemcke, Ullrich Meier
15.1
Intraoperative Complications
120
15.2.7
How Can Shunt Infections Be Avoided in
iNPH?
125
15.2
Infections
121
15.2.8
How Can Shunt-Related Infections Be
15.2.1
What is a Shunt Infection?
121
Treated?
126
15.2.2
Shunt Colonization
122
15.2.3
Shunt-Related CSF Infection/Meningitis . .
123
15.3
Postoperative Complications
128
15.2.4
Shunt-Related Peritonitis/Abdominal
15.3.1
Underdrainage
128
Abscess
123
15.3.2
Overdrainage
128
15.2.5
Epidemiology of Shunt Infections in
Patients With iNPH
123
15.4
Shunt Malfunction: Shunt Revisions . . .
131
15.2.6
Does My Patient Have a Shunt
15.4.1
When to Revise a Shunt?
131
Infection?
124
15.4.2
How to Revise a Shunt?
132
16
Prognosis
142
Ullrich Meier
16.1
Predictors
142
16.2
Improvement of Prognosis
142
17
Summary and Future Perspectives
146
Michael J. Fritsch
17.1
Educating Medical Personnel
146
17.4
Diagnostic Tools and Prediction of
Shunt Responsiveness
147
17.2
Educating the Public
146
17.5
Shunt Technology Improvement
147
17.3
Role of Biomarkers
147
Index
149
X
1.1
Why a Book about NPH?
2
Chapter 1
Introduction
Michael J. Fritsch
1
Introduction
1 Introduction
Michael J. Fritsch
Normal pressure hydrocephalus (NPH) is a neurologic
or general practitioner specializing in NPH will have a
condition that is characterized by an enlargement of
busy practice for many years to come. This book will
the ventricles and the Hakim triad.1,2 The Hakim triad
provide much of the information needed for the man-
is named after Salomón Hakim, who first described
agement of NPH.
the clinical picture in his degree thesis in 1957, and
Third, since we do not know enough about the true eti-
consists of gait impairment (a broad-based, shuffling,
ology and pathophysiology of hydrocephalus—and, in
magnetic gait), mental deterioration
(dementia), and
particular, NPH—there is an increasing need for labora-
urinary incontinence.3,4
tory research (e.g., biomarkers for neurodegenerative dis-
Further signs of NPH include headaches and imbalance.
eases), better prediction of who will benefit from shunt
The clinical symptoms can be improved by the shunting
surgery (based on imaging studies or clinical tests), clini-
of cerebral spinal fluid, which is temporarily achieved via
cal studies to test the efficiency and efficacy of treatment,
lumbar puncture or external lumbar drain, or perma-
and, last but not the least, scientific discussion about the
nently achieved via shunt implantation.
aforementioned topics.9
NPH can be divided into two subtypes: primary or
With this book, we would like to contribute to the sci-
idiopathic NPH (iNPH) and secondary NPH (sNPH). sNPH
entific literature on hydrocephalus in general and, more
has a known underlying cause for hydrocephalus. The
specifically, on NPH.
most common etiologies for secondary NPH are sub-
Fourth, effective treatment
(shunt placement) for
arachnoid hemorrhage, intracerebral or intraventricular
patients with NPH can significantly contribute to an
hemorrhage, severe traumatic brain injury, meningitis,
improvement in their quality of life, as well as to the
and ventriculitis.
quality of life of the entire family.10
iNPH is a disease of the elderly population, with an
The patient population with NPH is mostly over
increasing incidence beginning between the ages of 65
65 years of age. Mobility improvement (ability to walk),
and 70 years. In primary or iNPH, the cause of hydroceph-
cognitive function (short-term memory and orientation
alus remains unclear (idiopathic), at least when following
in space, time, and situations), and social function (uri-
the currently established theories. Some of the modern
nary incontinence) can make a difference to quality of
concepts of etiology and pathophysiology of iNPH will be
life, even if the duration of the benefit is limited (typi-
presented in this book (see Chapter 5).
cally 3-5 years). Shunt placement in patients with NPH
may be one of the few neurosurgical procedures where
function is restored and, for example, is comparable to
1.1 Why a Book about NPH?
deep brain stimulation. Although there is no such thing
as surgery without risk, shunt placement does have a
There are several answers to why a book about NPH is
low complication rate. The risk-benefit ratio strongly
needed. The diagnostic work-up of patients with NPH
favors surgical treatment if the appropriate diagnosis
is quite challenging, the daily routine management
has been made.
of elderly patients with dementia is demanding, and
there are some much more interesting and rewarding
neurosurgical procedures than placement of a shunt.
References
First, iNPH is underdiagnosed. It is estimated that 80%
of NPH cases remain unrecognized. One of the main
[1] Adams RD, Fisher CM, Hakim S, Ojemann RG, Sweet WH. Sympto-
reasons is the difficulty in differentiating iNPH from
matic occult hydrocephalus with “normal” cerebrospinal fluid pres-
sure: a treatable syndrome. N Engl J Med 1965; 273: 117-126
other neurodegenerative diseases (Alzheimer disease,
[2] Hakim S, Adams RD. The special clinical problem of symptomatic
Parkinson disease, vascular dementia, and lumbar spinal
hydrocephalus with normal cerebrospinal fluid pressure. Obser-
canal stenosis).5,6
With this book, we would like to
vations on cerebrospinal fluid hydrodynamics. J Neurol Sci 1965;
share our knowledge with medical professionals about
2: 307-327
the signs and symptoms of NPH, diagnostic tests, and
[3] Hakim CA, Hakim R, Hakim S. Normal-pressure hydrocephalus.
Neurosurg Clin N Am 2001; 12: 761-773
available treatment options. The most important point
[4] Wallenstein MB, McKhann GM. Salomón Hakim and the discovery of
is to consider NPH as a differential diagnosis if patients
normal-pressure hydrocephalus. Neurosurgery 2010; 67: 155-159,
present with gait impairment, dementia, and urinary
discussion 159
incontinence.7,8
[5] Tisell M, Höglund M, Wikkelsø C. National and regional incidence of
surgery for adult hydrocephalus in Sweden. Acta Neurol Scand 2005;
Second, the number of patients requiring diagnosis
112: 72-75
and treatment is rising. Therefore, NPH treatment and
[6] Kiefer M, Unterberg A. The differential diagnosis and treatment of
the follow-up of treated patients will be increasingly
normal-pressure hydrocephalus. Dtsch Arztebl Int 2012; 109: 15-25,
needed in the future. The neurosurgeon, neurologist,
quiz 26
2
Introduction
[7] Conn HO. Normal pressure hydrocephalus: a case report by a physi-
[9] Aschoff A, Kremer P, Hashemi B, Kunze S. The scientific history of
cian who is the patient. Clin Med 2007; 7: 296-299
hydrocephalus and its treatment. Neurosurg Rev 1999; 22: 67-93,
[8] Conn HO, Lobo FM. What do physicians know about normal pressure
discussion 94-95
hydrocephalus and when did they know it? A survey of 284 physi-
[10] Hebb AO, Cusimano MD. Idiopathic normal pressure hydrocephalus:
cians. Yale J Biol Med 2008; 81: 19-29
a systematic review of diagnosis and outcome. Neurosurgery 2001;
49: 1166-1184, discussion 1184-1186
3
2.1
Epidemiologic Studies
6
Chapter 2
2.2
Discussion
7
Epidemiology of Idiopathic
2.3
Summary
8
Normal Pressure
Hydrocephalus
Michael J. Fritsch
2
Epidemiology of Idiopathic Normal Pressure Hydrocephalus
2 Epidemiology of Idiopathic Normal Pressure
Hydrocephalus
Michael J. Fritsch
between clinical findings and
(attempted) imaging,
2.1 Epidemiologic Studies
according to the authors, suggests that iNPH symptoms
are often overlooked.
2.1.1 Trenkwalder et al (1995)
There are limitations to this study including the almost
In 1995, Trenkwalder et al conducted one of the first and
negligible number of patients who underwent imaging
most quoted studies.1 The aim of the study was to investi-
and further clinical evaluation. Among a subset of 17
gate the prevalence of different types of Parkinson dis-
patients who underwent a standard iNPH diagnostic and
ease (PD) in elderly patients. The study was conducted by
management protocol, 11 received a shunt.
going door-to-door in two Bavarian villages and survey-
ing 982 participants. Individuals older than 65 years were
2.1.4 Hiraoka et al (2008)
included. Following a screening questionnaire and motor
testing, all individuals suspected of having PD were rein-
Hiraoka et al examined the prevalence of iNPH in the eld-
vestigated by a neurologist and underwent a computed
erly population of a Japanese rural community.4 They
tomography (CT) scan. The prevalence of PD in this study
selected 2,053 residents aged 65 years or older to com-
group was 0.71% and—as an unexpected finding—the
plete a health questionnaire. From this group, 240 people
prevalence of normal pressure hydrocephalus (NPH) was
were randomly selected for an MRI examination. Of these
0.41% (4/982). No further diagnostic work-up or treat-
240 people, only 200 underwent MRI, and 170 under-
ment was attempted.
went a neurologic examination and neuropsychological
The study was performed 18 years ago. Magnetic reso-
testing. The 40 participants who did not undergo MRI
nance imaging (MRI) has brought further insight into the
were unable to do so because of diseases, immobility, or
diagnosis of hydrocephalus, and the age distribution of
other physical problems. Patients with an Evans index of
the German population has changed—one would expect a
0.3 or greater and narrowing of the cerebrospinal fluid
higher prevalence of NPH if this study were repeated
(CSF) space at the convexity and midline area were con-
today.
sidered to be positive for ventricular enlargement. Those
who underwent MRI were screened for clinical signs and
symptoms of iNPH. Within the group of 170 who were
2.1.2 Tisell et al (2005)
examined, 5 (2.9%) demonstrated ventricular enlarge-
Tisell et al determined that the annual incidence of sur-
ment and cognitive impairment (5/5), and 1 of these five
gery for adult hydrocephalus in Sweden was 3.4 per
showed gait disturbance (1/5), and 1 showed urinary
100,000 between the years 1996 and 1998.2 The most
incontinence (1/5). The authors concluded that there was
common indication for surgery was NPH (47%). The
a 2.9% prevalence of iNPH in the elderly population aged
resulting incidence for NPH was 1.598 (47% of 3.4) per
65 years or older.
100,000 each year.
This study has several limitations. First, the majority of
the 40 participants not examined were 80 years of age or
older and in poor physical condition. Among these 40,
2.1.3 Marmarou et al (2007)
there were probably more people who would have ful-
Marmarou et al wanted to estimate the prevalence of
filled the criteria for iNPH. Second, no CSF tap test, lum-
idiopathic NPH (iNPH) among residents of two assisted-
bar drainage test, or CSF shunting was performed in any
living and two extended-care facilities.3
Patients who
of the patients. Therefore, there is no clinical confirma-
were 85 years or older were excluded. In a retrospective
tion of the diagnosis of iNPH. Third, the distribution of
chart review, the analysis included 147 patients. Overall,
symptoms (five participants with dementia, including
14.7% of the patients had symptoms of gait disturbance
one with gait disturbance, and one with urinary
and incontinence, and 9.4% had symptoms of gait distur-
incontinence) is atypical for iNPH. One would expect to
bance and dementia. A total of 11 of the 147 patients
find gait disturbances in almost all of these patients.
(7.5%) had a complete Hakim triad. The authors assumed
that gait disturbance plus incontinence, dementia, or both
2.1.5 Brean and Eide (2008)
was necessary for a clinical diagnosis of iNPH; therefore,
they concluded that the overall prevalence of the disease
Brean and Eide examined the prevalence of iNPH in
ranged from approximately 9% to 14%.
Norway in 2008.5 In a population of 220,000 inhabitants,
However, of the 147 patients included in the survey,
intense efforts were directed to the public and to health
only 5 had undergone MRI or CT imaging. The discrepancy
care professionals to recruit and refer patients with iNPH
6
Epidemiology of Idiopathic Normal Pressure Hydrocephalus
during a 12-month period. The task was supported by the
The overall prevalence of (possible) iNPH was considered
fact that the population of 220,000 was served by one
to be 1.4%.
single neurological department and one single neuro-
Again, the distribution of symptoms appears atypical
surgical department. Within the 12-month period, 86
for iNPH and is similar to that of another study from
patients were referred for evaluation of NPH. Out of
Japan.4 One would expect gait disturbances in almost all
those, 48 patients fulfilled the criteria for NPH based on
of these patients. In contrast, it is quite possible that the
imaging and clinical examination.
symptomatology for iNPH in Japan is different from that
Based on these data, the minimum prevalence of iNPH
in Europe, and this would be interesting to evaluate.
in the Norwegian population was calculated as 21.9 per
Another limitation of this study is the nonparticipation
100,000 cases. The incidence for the 12-month period
of 70 randomly selected inhabitants. One can assume that
was found to be 5.5 per 100,000 cases per year. The
participation of this subgroup would have increased the
authors concluded that these numbers represent the
prevalence (elderly people, immobility) and that further
minimum estimates.
clinical testing and treatment would have decreased the
The limitation of this study—which is similar to that of
prevalence (since not all suspected people will test posi-
Tisell et al’s study in Sweden—is that only referred
tive or benefit from treatment). Notwithstanding these
patients were evaluated.2 This excludes the population
limitations, this study seems to be one of the best designs
that was simply not evaluated (probably the majority) as
published so far.
well as part of the population that was treated outside of
the area.
2.1.8 Iseki et al (2009)
In 2009, Iseki et al investigated the clinical and preclinical
2.1.6 Brean et al (2009)
stage of iNPH in a general population.8 All residents of
In a follow-up study in 2009, Brean et al retrospectively
two communities aged either 61 years or 70 to 72 years
collected data on patients who were hospitalized from
(N = 1,142) were asked to undergo an MRI examination,
2002 to 2006 with a diagnosis of iNPH in one of five neu-
and, of those,
790
(69.2%) participated in the study.
rosurgical centers in Norway.6 During the 5-year period,
A total of 12 residents (1.52%) displayed typical features
252 patients underwent operations for iNPH.
of iNPH on MRI, including an Evans index of > 0.3 and nar-
The total incidence of iNPH was 1.09 per 100,000 cases
rowing of the subarachnoid space and cortical sulci at the
per year. This number must be put in perspective with
high convexity of the cerebrum. These individuals were
regard to the estimated incidence of 5.5 per 100,000 cases
categorized as having “possible iNPH.” Of the 12 individu-
per year from the 2008 study.
als,
8 were asymptomatic and 4 had gait disturbances,
The yearly incidence ranged from a minimum of 0.84
dementia, or both. The prevalence was determined to be
per 100,000 per year to a maximum of 1.7 per 100,000
0.51% (4/790) among the Japanese elderly population
per year. The incidence was age dependent and was high-
(> 61 years of age).
est in those aged 70 to 79 years. No regional differences
regarding incidence, sex, and age were found.
2.1.9 Klassen and Ahlskog (2011)
The data from the 2009 study suggest that there are
In a retrospective study of records, Klassen and Ahlskog
too few patients being evaluated and treated for iNPH.
determined the community incidence of clinically sus-
One can estimate that roughly 20% of the patients who
pected NPH in Olmsted County
(Minnesota, USA).9
fulfilled the diagnostic criteria for iNPH based on imaging
Between 1995 and 2003, 41 patients underwent an inva-
and clinical examination were eventually treated.
sive diagnostic procedure for evaluation of suspected
NPH. Out of those, 13 received a shunt. The authors calcu-
2.1.7 Tanaka et al (2009)
lated, based on the number of inhabitants in the county,
Tanaka et al investigated the prevalence of (possible)
an incidence of 1.19 per 100,000 per year. However, one
iNPH using a random sample database.7 Among 1,654
can again assume that these numbers are underestimated
members of the population of the Tajiri area aged 65
because only those patients who were referred and those
years or older, 567 were randomly selected, of whom 497
who were treated were counted.
underwent MRI. Participants were classified as having
possible iNPH if they had ventricular enlargement (Evans
2.2 Discussion
index ≥ 0.3), at least one clinical finding of the Hakim
triad, and no obvious cause for hydrocephalus. Seven par-
These studies show a wide variety of prevalence and inci-
ticipants met the aforementioned criteria. None of them
dence for iNPH (▶ Table 2.1). Studies that determine the
presented with the full clinical triad and only three par-
prevalence based only on clinical findings3 overestimate,
ticipants met two criteria. Cognitive impairment was the
and studies that determine the prevalence based only on
most common symptom (n = 6), followed by gait distur-
treated patients2,6,9
clearly underestimate the
“true”
bance (n = 3). No urinary incontinence was observed.
prevalence. The same would apply to the incidence of iNPH.
7
Epidemiology of Idiopathic Normal Pressure Hydrocephalus
Table 2.1 Summary of publications determining the prevalence and incidence of NPH
Authors
Year
Population
Age (years) Prevalence (%)a
Incidence (%)b
Trenkwalder et al
1995
N=982
> 65
0.41
Tisell et al
2005
0.00159
(1.598/100,000/y)
Marmarou et al
2007
N=147
< 85
9.4-14.7
Hiraoka et al
2008
N=170
≥ 65
2.9
Brean and Eide
2008
220,000
(estimated)
0.0219
(21.9/100,000)
0.0055
(5.5/100,000/y)
Brean et al
2009
220,000
(estimated)
0.00109
(1.09/100,000/y)
Tanaka et al
2009
N=497
≥ 65
1.4
Iseki et al
2009
N=790
≥ 61
0.51
Klassen and Ahlskog
2011
N=41
0.00119
(1.19/100,000/y)
Abbreviations: NPH, normal pressure hydrocephalus; y, year.
aNumber of (suspected) NPH-positive patients/number of people evaluated.
bNumber of (suspected) NPH-positive patients/number of people evaluated/time period (1 y).
The most accurate determinations are, most likely, those
2.3 Summary
from the Tanaka et al and Iseki et al studies.7,8 This is
because both studies are population-based with
(ran-
The prevalence of iNPH is not currently well defined but,
domly) selected people who underwent radiographic and
in developed countries, is most likely between 0.5% and
clinical examinations; however—and this is their limitation
1.5% among the population aged 61 years and older.
—no further diagnostic work-up or treatment was initiated
The prevalence and incidence of iNPH are increasing for
in either study.7,8
several reasons, including age pyramid distribution, diag-
We can assume that the number of people in the eld-
nostic tools, and disease awareness. Those aged between
erly population (≥ 65 years) in developed countries (from
70 and 79 years should receive the most attention for
studies in Germany, Sweden, Norway, the United States,
iNPH from medical professionals.
and Japan) displaying symptoms of the Hakim triad and
ventricular enlargement is increasing. Reasons for this
are: change of the age distribution pyramid in developed
References
countries toward the older population; better diagnostic
tools (MRI) and clinical tests (higher sensitivity and speci-
[1] Trenkwalder C, Schwarz J, Gebhard J et al. Starnberg trial on epidemiol-
ogy of Parkinsonism and hypertension in the elderly. Prevalence of
ficity); and increased awareness among physicians, care-
Parkinson’s disease and related disorders assessed by a door-to-door sur-
givers, and the general population about iNPH.
vey of inhabitants older than 65 years. Arch Neurol 1995; 52: 1017-1022
The incidence of iNPH increases with age, and the high-
[2] Tisell M, Höglund M, Wikkelsø C. National and regional incidence of
est incidence appears to be seen in those between the
surgery for adult hydrocephalus in Sweden. Acta Neurol Scand 2005;
112: 72-75
ages of 70 and 79 years.6 Members of this population
[3] Marmarou A, Young HF, Aygok GA. Estimated incidence of normal pres-
should raise the highest suspicion for being positive for
sure hydrocephalus and shunt outcome in patients residing in assisted-
the disease among physicians.
living and extended-care facilities. Neurosurg Focus 2007; 22: E1
Most likely, the population of those aged 80 years and
[4] Hiraoka K, Meguro K, Mori E. Prevalence of idiopathic normal-pressure
older has an even higher incidence than is reported.
hydrocephalus in the elderly population of a Japanese rural commu-
nity. Neurol Med Chir (Tokyo) 2008; 48: 197-199, discussion 199-200
There are several reasons why this population does not
[5] Brean A, Eide PK. Prevalence of probable idiopathic normal pressure
receive the most attention with regard to the diagnosis of
hydrocephalus in a Norwegian population. Acta Neurol Scand 2008;
and treatment for iNPH. First, persons older than 80 years
118: 48-53
represent a small part of the elderly population. Second,
[6] Brean A, Fredø HL, Sollid S, Müller T, Sundstrøm T, Eide PK. Five-year
patients of this age present less frequently with NPH
incidence of surgery for idiopathic normal pressure hydrocephalus in
Norway. Acta Neurol Scand 2009; 120: 314-316
symptoms, either because patients do not visit to their
[7] Tanaka N, Yamaguchi S, Ishikawa H, Ishii H, Meguro K. Prevalence of
primary care physician or neurologist at all
(perhaps
possible idiopathic normal-pressure hydrocephalus in Japan: the
due to immobility or for other various reasons) or they
Osaki-Tajiri project. Neuroepidemiology 2009; 32: 171-175
present with “more significant diseases” such as stroke,
[8] Iseki C, Kawanami T, Nagasawa H et al. Asymptomatic ventriculome-
galy with features of idiopathic normal pressure hydrocephalus on
cardiovascular emergencies, and malignancies. Third, as
MRI (AVIM) in the elderly: a prospective study in a Japanese popula-
several authors have pointed out, this subgroup of
tion. J Neurol Sci 2009; 277: 54-57
patients has a negative risk-benefit ratio for diagnostic
[9] Klassen BT, Ahlskog JE. Normal pressure hydrocephalus: how often
and surgical measures related to hydrocephalus.3,9
does the diagnosis hold water? Neurology 2011; 77: 1119-1125
8
3.1
Early History
10
Chapter 3
3.2
Hydrocephalus and its
History
Treatment in the 19th and 20th
Centuries
10
Michael J. Fritsch
3.3
Development of Modern Shunt
Concepts
11
3.4
Hakim and the Discovery of
Normal Pressure Hydrocephalus
12
3.5
Modern Shunt Technology and
Perspectives
13
History
3 History
Michael J. Fritsch
Thomas Willis
(1621-1675), an English anatomist,
3.1 Early History
neurologist, and psychiatrist, is today best known for the
Hydrocephalus, which is derived from the Greek words
“Circle of Willis.” He was the first to number the order of
hydro (water) and cephalon (head, brain), is a condition
the cranial nerves in the way in which they are numbered
that has been known for more than 2,000 years.
today. Based on the studies of his coworker Richard Lower
Hippocrates (ca. 460-370 BC) gave one of the earliest
(1631-1691), who had shown that the cribriform plate is
scientific descriptions of hydrocephalus. In the Corpus
watertight and, therefore, cannot be the exit of CSF from
Hippocraticum, which is a collection of around 70 medical
the brain, Willis postulated that the circulation of CSF must
works from ancient Greece (probably written not only by
occur within the brain. He described the communication of
Hippocrates but also by his followers and students), the
the surface of the brain (sulci, subarachnoid space) with
term hydrocephalus was first used. The term referred to
the cavities beneath the fornix (lateral ventricles).
“fluid collection” in and around the brain. The disease was
Antonio Pacchioni (1655-1726) was an Italian anato-
explained by liquefaction of the brain caused by epileptic
mist who described the arachnoid granulations that are
seizures. The symptoms correlated to hydrocephalus were
now named after him. However, he assumed that they
described as headaches, vomiting, and visual disturbances.1
were sites of CSF secretion.1
Galen of Pergamon (ca. 129-199) acquired anatomic
Claude-Nicolas Le Cat (1700-1768), a French surgeon,
knowledge by dissecting living (vivisection) and dead
on October 15, 1744 introduced a specially invented can-
animals. The law at that time prohibited the dissection of
nula into the lateral ventricle of a newborn with hydro-
human cadavers. Galen gave descriptions of ventricular
cephalus. The cannula was used as a tap and was left in
anatomy, the choroid plexus, as well as of the cerebrospi-
place for 5 days until the child died. Le Cat had developed
nal fluid
(CSF) as watery clear liquid.1
Galen also
the device for the purpose of repeatedly draining extrac-
described the difference between motor and sensory
ranial CSF to treat congenital hydrocephalus. The proce-
nerves, the concept of muscle tone, and the concept of
dure is considered to be the first use of a device for the
muscle agonists and antagonists.
repeated therapeutic removal of CSF from the ventricular
Leonardo da Vinci (1452-1519) completed the first
system, inaugurating the concept of external ventricular
detailed drawing of the ventricular system in 1510. New
drainage.2,3
discoveries in human anatomy and physiology became
Robert Whytt
(1714-1766) performed scientific
possible when dissection of the human body was toler-
clinical studies on patients, especially in children with
ated and later legalized.1 As an artist, da Vinci was given
suspected hydrocephalus. For example, Whytt described
permission to dissect human bodies at the Hospital of
the difference in the clinical course depending on
Santa Maria Nuova in Florence, Italy. Based on these stud-
whether an infant had open or closed cranial sutures.
ies, Leonardo created illustrations of the human skeleton,
Francois Magendie (1783-1855) described the caudal
muscles, heart, and vascular system, and a fetus in utero.
opening of the fourth ventricle and postulated that an
Da Vinci’s illustrations depicted anatomical details in a
occlusion of the CSF pathway may cause hydrocephalus.
then-unknown realism.
Magendie inaugurated CSF pressure measurement by
Andreas Vesalius (1514-1564), a Flemish anatomist
performing a suboccipital puncture in a dog in 1841.1
and physician, was the author of one of the most influen-
tial books of his time, entitled De Humani Corporis
Fabrica (On the Structure of the Human Body). Vesalius is
3.2 Hydrocephalus and its
often referred to as the founder of modern anatomy.
He acquired his knowledge through the anatomical
Treatment in the 19th and
dissection of human bodies.
20th Centuries
Vesalius gave the first scientific description of hydro-
cephalus based on clinical and anatomical data. He
Axel Hendrick Key (1832-1901) and Magnus Gustav
observed the disease in a 2-year-old girl with a growing
Retzius (1842-1919) developed and proved a new mod-
head circumference. Vesalius examined the patient while
ern concept of CSF physiology of their time, which, for
she was still alive; after her death, he found that the
the most part, is still valid today. They proposed that CSF
head enlargement came from fluid collection inside the
is produced in the choroid plexus, flows through and out
ventricles (9 lb [4.08 kg] of water) rather than from fluid
of the ventricular system, and is reabsorbed through the
accumulation around the brain. This opened the way for
subarachnoid villi.1
further thoughts and studies about CSF, its pathway, and
Emil Theodor Kocher (1841-1917) was a Swiss physi-
the related disease, hydrocephalus.1
cian and is best known for his work in thyroid surgery.
10
History
He received the Nobel Prize for Physiology and Medicine
from meningitis 7 months later. An autopsy revealed that
in 1909. A number of instruments and surgical approaches
both of the venous shunts had healed well in place with
are named after him; one of these is the frontal precoronal
patent lumens. No blood was found in the ventricles, con-
bur-hole approach for ventricular puncture.
firming the concept of unidirectional flow directed by the
Heinrich Irenaeus Quincke
(1842-1922) was a
venous valves.
German internist and surgeon. He worked at the univer-
One year later, Payr implanted a formalin-fixed
sities of Vienna, Berlin (Charité), Bern, and Kiel. His main
paraffin-impregnated bovine artery into a patient
16
contribution to today’s understanding of hydrocephalus
years of age that connected the lateral ventricle with the
was the lumbar puncture (at the time, it was called
interhemispheric fissure. According to clinical observa-
“Quincke puncture”), which he performed for diagnostic
tions, the shunt functioned for 11 years.
and therapeutic purposes. His main interest was the diag-
William Jason Mixter
(1880-1958), an American
nosis and treatment of meningitis and multiple sclerosis.
neurosurgeon practicing at the Massachusetts General
In 1893, he described a condition of “increased intra-
Hospital in Boston, was the first to perform a purely
cranial pressure,” a disease he called “meningitis serosa,”
endoscopic third ventriculostomy in
1923. Mixter
which is now referred to as pseudotumor cerebri.
inspected the ventricles and perforated the floor of the
Carl Wernicke (1848-1905) was a German psychiatrist
third ventricle during the surgery. He performed this
and neuropathologist. In 1873, he examined a patient
procedure on only one patient and did not pursue this
who had experienced a stroke. Although the man was
kind of operation any further.1
able to speak and his hearing was unimpaired, he could
Walter Edward Dandy (1886-1946), a neurosurgeon,
not understand what was said to him. After the patient
and Kenneth Daniel Blackfan (1883-1941), a pediatri-
died, Wernicke dissected his brain and found a lesion in
cian, studied hydrocephalus at Johns Hopkins Hospital in
the left temporal parietal region. Wernicke concluded
Baltimore, Maryland. They injected dye into the ventricle
that this region was an important part of speech compre-
of a dog and described the pathway of CSF. The knowl-
hension and named the syndrome “sensory aphasia.”
edge they gained enabled them to induce hydrocephalus
With regard to hydrocephalus, Wernicke is known for
in laboratory animals by blocking the sylvian aqueduct.
performing punctures of the ventricular trigone by using
They utilized small pieces of cotton for obstruction. Later
a trocar from a lateral approach, under aseptic condi-
on, Dandy and Blackfan occluded the foramen of Monro
tions.4 Until recently, the trigonal approach had certain
and made the observation that animals treated this way
applications and was mainly used by pediatric surgeons
would not develop hydrocephalus once the choroid
and neurosurgeons for shunt placement in infant
plexus had also been removed.5-7 Dandy also described
hydrocephalus.
the occipital puncture, and inaugurated pneumoencepha-
Jan Mikulicz-Radecki (1850-1905) was a Polish sur-
lography in 1918 for diagnostic purposes.
geon who worked in Krakau, Königsberg, and Breslau.
Mikulicz contributed mainly to the field of abdominal
surgery. In 1893, he implanted a wick made of glass wool
3.3 Development of Modern
into the lateral ventricle of an infant aged 6 months; the
Shunt Concepts
wick extended into the subarachnoid and subgaleal
spaces. This is considered to be the first permanently
Arne Torkildsen
(1899-1968), a Norwegian neuro-
implanted CSF drainage. The clinical course of the patient
surgeon, performed the first ventriculocisternostomy
was documented for the following 2 years and displayed
(a shunt from the occipital horn of the lateral ventricle to
a long-lasting reduction of head circumference.1
the cisterna magna, also called “Torkildsen’s operation”;
Erwin Payr
(1871-1946) was Chief of Surgery in
Lundar and Nakstad, 1990).8 The Torkildsen shunt was a
Greifswald, Germany, from 1907 to 1910. In 1910, he was
popular method to treat obstructive hydrocephalus until
appointed Professor of Surgery at the University of
the 1980s. The concept of connecting the ventricles with
Königsberg and relocated to Leipzig, Germany, in 1911
the subarachnoid space was later repopularized by the
where he remained until his retirement in 1937.
endoscopic ventriculocisternostomy.
During his time in Greifswald in 1907, Payr created the
Robert H. Pudenz (1911-1998), a neurosurgeon, and
first drainage from the ventricular system into the supe-
Ted Heyer, an engineer, spent 3 years at the Huntington
rior sagittal sinus of a girl aged 9 years with hydrocepha-
Medical Research Institutes in Pasadena to research how
lus. He utilized the autologous saphenous vein using pre-
to construct a Teflon valve with a transverse slit mecha-
served valves as shunt material, thereby directing the CSF
nism. Pudenz implanted it in 1955 into a child with
flow. The patient’s symptoms of elevated intracranial
hydrocephalus.9 The ventriculoatrial shunt functioned for
pressure improved to a certain extent. Six weeks later,
2 years. In 1958, Rudi Schulte, a young watchmaker from
Payr implanted a second shunt on the opposite side in
Germany, joined the two. He improved the slit-valve
the same patient. However, poor wound healing compli-
mechanism by adding further multiple longitudinal slits.
cated the surgery. The child initially recovered but died
Pudenz became the medical director of the Heyer-Schulte
11
History
Corporation and was a partner in the Pudenz-Schulte
As part of his fellowship, Hakim conducted autopsies
Corporation, which later became part of Medtronic.
on patients with neurodegenerative diseases. Some of the
John D. Holter (1916-2003) was a toolmaker. His son
cases demonstrated enlargement of the ventricular sys-
Charles was born in 1955 with spina bifida; soon after his
tem. At the time, it was unclear whether the enlargement
birth, he contracted meningitis and subsequently devel-
was due to cerebral atrophy or the increased ventricular
oped hydrocephalus.10 Holter was astonished and chal-
volume driven by pressure.
lenged by the inability of medical technology to correct
Three years later, in 1957, Hakim returned to Bogotá,
what appeared to be a simple hydraulic problem. In an
Colombia. He took a position at the Hospital San Juan de
attempt to save his son, Holter developed a silicone one-
Dios. During his first year back in Bogotá, Hakim was con-
way valve in his workshop at home. After finding a medi-
fronted with the case of a 16-year-old boy with a severe
cally suitable grade of Silastic (Dow Corning Corporation,
traumatic brain injury from a car accident.
Midland, Michigan, USA), the device was patented and
The patient was operated on for a subdural hema-
Holter founded a company to manufacture shunts.
toma and the surgery was considered to be successful.
Although Holter was unable to save his son, the design he
However, the patient remained in an impaired level of
developed together with the neurosurgeon Eugene
consciousness. As a diagnostic procedure, pneumoen-
Bernard Spitz (called the Spitz-Holter shunt) has been
cephalography was performed and the study indicated
used throughout the world since 1956.11
ventricular enlargement. The pressure readings taken at
Eugene Bernard Spitz (1919-2006) was a pediatric
the same time displayed a rather normal intracranial
neurosurgeon at the Children’s Hospital of Philadelphia,
pressure.
Pennsylvania. In May 1949, and still a neurosurgery resi-
Hakim removed 15 mL CSF for further laboratory
dent, he implanted a valve constructed by his colleague,
investigation. After the CSF removal, the patient’s level of
the neurosurgeon Frank E. Nulsen (1916-1994), that
consciousness improved the next day. His alertness
consisted of two ball-in-cone valves in series with a
subsequently declined over the following days and then
rubber pumping chamber in between them.12 Spitz and
improved again after a second lumbar puncture. Hakim
Nulsen used a 1.7-mm polyethylene catheter placed in
decided to implant a ventricular atrial shunt. The
the superior vena cava, which slightly protruded into the
patient improved significantly and the treatment was
atrium, as the distal part of the shunt. Clinical follow-up
long lasting. The case was presented in Hakim’s doctoral
for 2.5 years, serial pneumoencephalograms, and diag-
thesis in 1964.14
nostic shunt occlusions proved the function of the first
Hakim asked the question: why does a patient with
implanted shunt valve.10 Spitz later continued to implant
normal intracranial pressure benefit from the removal of
shunt valves constructed by John D. Holter (see above).
(a small amount of) CSF? His answer was simple and
Ayub Khan Ommaya (1930-2008) was a Pakistani
scientifically sophisticated at the same time: what was
neurosurgeon and the inventor of the subgaleal reservoir
considered to be “normal pressure” was too high for this
named after him. The first report of the Ommaya reser-
particular individual. Hakim took the well-known physics
voir was given in 1963. The reservoir was the first medi-
equation to define pressure: pressure equals force per
cal device to use silicone. It is connected to a ventricular
unit area (P = F/A). At the same time, force equals pressure
catheter, originally used for applying intrathecal chemo-
multiplied by unit area (F = P × A).
therapy. The concept was later adapted for use in the
Since this particular patient had enlarged ventricles,
treatment of hydrocephalus, allowing it to be integrated
his unit area would be enlarged compared with a healthy
into a shunt system or to be applied as a stand-alone
person. If one applies the same pressure (so far consid-
solution for repeated transcutaneous puncture.1
ered to be normal) to a larger area, then the force must be
increased to keep the equation in balance. Hakim con-
cluded that this increased force would impact the brain
causing damage, or at least impair the level of conscious-
3.4 Hakim and the Discovery
ness. Hakim further concluded that the pressure in small
of Normal Pressure
and large ventricles may be equal, but the force in the
larger ventricles is increased due to larger surface area
Hydrocephalus
(volume).
Salomón Hakim (1922-2011) was born in Barranquilla,
After publishing his thesis in 1964, another patient
Colombia, to a family of emigrants from Lebanon. He
presented to Hakim. This woman was an American citi-
started medical school in 1944 and afterward enrolled in
zen and she displayed the symptoms of normal pressure
neurosurgical residency training in Bogotá, Colombia.
hydrocephalus. Hakim recommended a spinal tap test
After being awarded a fellowship in 1950 in Boston,
and, if successful, shunt placement. The family of the
Massachusetts (United States), Hakim returned 4 years
patient was afraid to undergo treatment in Colombia and
later as a research fellow in neuropathology to the
wanted to go back to the United States. Since Hakim knew
Massachusetts General Hospital.13
that no one in the United States at the time was aware of
12
History
the clinical condition—and, therefore, no one would treat
New theories and concepts of the etiology and patho-
the patient appropriately—he offered to accompany the
physiology of hydrocephalus will be covered in Chapter 5,
patient to Boston, where he and the patient met with
while the different valve concepts, valve adjustability,
Dr. Raymond Adams.
overdrainage protection, and modern concepts of pres-
At the Massachusetts General Hospital, the patient
sure measurement will be reviewed in Chapter 9.
underwent placement of a temporary spinal drainage
and, consequently, shunting. This case was summarized
in a 1965 article that appeared in the New England Jour-
References
nal of Medicine.15 Hakim was considered to be only one of
[1]
Aschoff A, Kremer P, Hashemi B, Kunze S. The scientific history of
the coauthors. The first author was Raymond Adams,
hydrocephalus and its treatment. Neurosurg Rev 1999; 22: 67-93,
who, at the time, was the chief of service of the
discussion 94-95
Department of Neurosurgery. The other coauthors
[2]
Kompanje EJ, Delwel EJ. The first description of a device for repeated
external ventricular drainage in the treatment of congenital
included C. Miller-Fisher (Neurology), Robert Ojemann
hydrocephalus, invented in 1744 by Claude-Nicolas Le Cat. Pediatr
(Neurosurgery), and William Sweet
(Neurosurgery).
Neurosurg 2003; 39: 10-13
According to a comment from Dr. Marvin Bergsneider
[3]
Missori P, Paolini S, Domenicucci M. The origin of the cannula for
in Neurosurgery, the order of the authors was chosen
ventriculostomy in pediatric hydrocephalus. J Neurosurg Pediatr
alphabetically.16
2011; 7: 290-294
[4]
Wernicke C. Lehrbuch der Gehirnkrankheiten. Fischer, Kassel; 1981,
After Hakim’s publication in 1965, the spinal tap test
pp. 377-378
became—and has remained since then—the standard
[5]
Dandy WE, Blackfan KD. An experimental and clinical study of inter-
diagnostic test. Further tools to predict shunt respon-
nal hydrocephalus. JAMA 1913; 61: 2216-2217
siveness include lumbar spinal drainage, a lumbar (and
[6]
Dandy WE, Blackfan KD. Internal hydrocephalus: an experimental,
ventricular) infusion test, and intracranial pressure
clinical, and pathological study. Am J Dis Child 1914; 8: 406-482
[7]
Dandy WE. Extirpation of the choroid plexus of the lateral ventricle
monitoring.
in communicating hydrocephalus. Am Surg 1918; 68: 569-578
Initial pneumoencephalography was later replaced by
[8]
Lundar T, Nakstad P. [Torkildsen’s operation—50 years later] Tidsskr
computed tomography and magnetic resonance imaging.
Nor Laegeforen 1990; 110: 584-586
Alternatives to shunting include choroid plexus cauteri-
[9]
Pudenz RH. The surgical treatment of hydrocephalus—an historical
review. Surg Neurol 1981; 15: 15-26
zation or removal, medical treatment with acetazol-
[10]
Boockvar JA, Loudon W, Sutton LN. Development of the Spitz-Holter
amide, and, later, endoscopic third ventriculostomy.
valve in Philadelphia. J Neurosurg 2001; 95: 145-147
Hakim was further involved in laboratory and clinical
[11]
Carrington KW. Ventriculo-venous shunt using the Holter valve as a
research as well as the development of shunting
treatment of hydrocephalus. J Mich State Med Soc 1959; 58: 373-
systems.17 In 1966, Hakim presented a unidirectional,
376, passim
[12]
Nulsen FE, Spitz EB. Treatment of hydrocephalus by direct shunt from
pressure-regulated valve that was more efficient and
ventricle to jugular vein. Surg Forum 1952; 2: 399-403
reliable than previous slit mechanism valves. The Hakim
[13]
Wallenstein MB, McKhann GM. Salomón Hakim and the discovery of
system consisted of a spring and ball-in-cone mechanism
normal-pressure hydrocephalus. Neurosurgery 2010; 67: 155-159,
that controlled pressure.
discussion 159
[14]
Hakim S. Some observations on CSF pressure: hydrocephalic syn-
drome in adults with “normal” CSF pressure. Thesis 957, Javeriana
University School of Medicine, Bogotá, Colombia, 1964
3.5 Modern Shunt Technology
[15]
Adams RD, Fisher CM, Hakim S, Ojemann RG, Sweet WH. Sympto-
matic occult hydrocephalus with
“normal” cerebrospinal fluid
and Perspectives
pressure: a treatable syndrome. N Engl J Med 1965; 273: 117-126
[16]
Bergsneider M. Comment to: Wallenstein M, McKhann GM. Salomón
Since the 1950s, probably more than 200 shunt designs
Hakim and the discovery of normal pressure hydrocephalus.
have been developed.1 Neuroendoscopy has evolved over
Neurosurgery 2010; 67: 159
the last 15 years and has replaced shunt implantation as
[17]
Hakim CA, Hakim R, Hakim S. Normal-pressure hydrocephalus.
the treatment of choice for obstructive hydrocephalus.
Neurosurg Clin N Am 2001; 12: 761-773, ix
13
4.1
Natural History
16
Chapter 4
4.2
Symptoms of iNPH
16
Clinical Characteristics and
4.3
Differential Diagnosis
18
Differential Diagnosis
4.4
Summary
21
Uwe Kehler
4
Clinical Characteristics and Differential Diagnosis
4 Clinical Characteristics and Differential Diagnosis
Uwe Kehler
The main symptoms of idiopathic normal pressure hydro-
(▶Fig. 4.1). Several authors deny the diagnosis of iNPH if
cephalus (iNPH) were described by Hakim and Adams1
gait disturbance is not present.1 A list of the symptoms of
and included gait disturbance, incontinence, and mental
iNPH is given in Box Symptoms of iNPH.
disorder. Because iNPH is a disease of the elderly, several
different conditions are often superimposed. Older peo-
Symptoms of iNPH
ple may experience hip and knee problems that interfere
with gait. Older men might have prostate adenomas with
Main symptoms
bladder problems, and women might have stress
● Gait disturbance
incontinence due to pelvic floor insufficiency—both of
● Incontinence
which interfere with bladder function. Dementia may
● Dementia
also be caused by cerebrovascular disease, Alzheimer dis-
ease (AD), and others. Cerebrovascular disease and AD
Additional symptoms
occur in up to 70% of patients with iNPH .2 Symptoms of
● Headache
iNPH develop over months and even years and they may
● Psychiatric syndromes
be very subtle at the beginning and difficult to detect.
● Dizziness/vertigo
However, being aware of the symptoms is important to
● Extended need of sleep
provisionally diagnose early iNPH, to differentiate it from
● Large head circumference
other diseases, and to be able to detect them even if
● Impairment of sexual function
comorbidities obscure the typical signs.
● Comorbidities
4.1 Natural History
There is a great variation in the onset, severity, and pro-
4.2.1 Main Symptoms
gression of symptoms. The cardinal symptoms, which
include gait disturbance, dementia, and incontinence, all
Gait Disturbance
of which do not have to be present together, may actually
Gait disturbance is typically the first symptom noted and
appear at different times. If symptoms are present, then
is thought to be an apraxia of gait. The gait disturbance is
they will usually progress.3,4 The progression can vary
characterized by decreased gait velocity and cadence
considerably, seeing almost stable courses over many
with shorter and more variable strides (see Box Typical
years or severe deteriorations in several months.5 The
gait characteristics in iNPH (p. 17)). The step height is
often slow and uncharacteristic onset of symptoms makes
early diagnosis of iNPH difficult. Therefore, it is difficult in
many cases to estimate how long the disease has been
ongoing. In exceptional cases, spontaneous regression of
symptoms may occur.4 Evaluation of the patient’s medical
history might be difficult due to possible dementia,
11%
7%
0.3%
therefore, the help of relatives, life partners, or both is
Gait disturbance
Incontinence
mandatory.
The typical onset of iNPH occurs in the elderly popula-
48%
tion (older than 60 years), but it may appear earlier. The
patient's age should be at least 40 years to have a proba-
29%
3%
ble diagnosis of iNPH.3 Younger age does not rule out
iNPH, but it makes the diagnosis less likely.
2%
4.2 Symptoms of iNPH
Dementia
The typical triad of iNPH is gait disturbance, incontinence,
and dementia, but all three symptoms will present simul-
taneously in about only one-half of patients.6 Gait distur-
Fig. 4.1
Frequency of simultaneous occurrence of iNPH
bance is almost always present, whereas incontinence or
symptoms. After Dauch and Zimmermann.3
dementia alone caused by iNPH is extremely rare
16
Clinical Characteristics and Differential Diagnosis
reduced and the dorsal extension of the forefoot is insuf-
Dementia
ficient. The patient’s feet may shuffle on the floor; this
Dementia in iNPH corresponds to a frontal subcortical
has also been described as glued to the floor or as a mag-
dementia with psychomotoric slowing, apathy, affective
netic gait.7 The gait is atactic and broad-based; when per-
indifference, inattentiveness, and memory deficits (see
turbed, the patient may lose balance. Patients may also
Box Typical signs of mental disorders in iNPH).3 Intellec-
have problems when they begin walking or when turn-
tual abilities seem to be present, but in a sleepy, blurred
ing. In contrast to Parkinson disease (PD), patients with
10
condition with loss of spontaneity and communication.
normal pressure hydrocephalus (NPH) have a preserved
Patients may not be able to answer questions right away
reciprocal arm swing,8 with a backward-directed posture
(akinetic mutism), but, after insisting, there will be a
and a tendency to fall. In the late stage of the condition,
delayed answer that is often correct.10,11 Symptoms may
apraxia of the upper limbs may also occur.
range from subtle to severe.
Typical gait characteristics in iNPH
Typical signs of mental disorders
● Atactic
in iNPH
● Broad-based
● Psychomotor slowing
● Slow
● Apathy
● Short step size
● Affective indifference
● Magnetic gait, shuffling
● Inattentiveness
● Preserved reciprocal arm swing
● Memory impairment
In the early stages of iNPH, the gait disturbance is subtle,
Differentiating dementia seen in iNPH from other forms
difficult to detect, and may be difficult to classify as NPH
of dementia may be difficult because symptoms may be
gait. Patients often complain about gait disturbance or
blurred by comorbidities that often exist such as AD and
slight balance problems, which they describe as dizziness,
cerebrovascular disease.2
which cannot be recognized by others. If gait disturbance
has progressed and is distinct, then it may be easily rec-
ognized as gait disturbance related to iNPH. In its final
Pathophysiology of Dementia in iNPH
stages, patients can walk only with massive support and
The cause of dementia in iNPH is not understood, but the
may become bedridden.
frontostriatal system, subcortical structures, and periven-
tricular projection fibers may be involved.3
Incontinence
Increased urinary frequency and urgency is often found
4.2.2 Other Symptoms
toward the onset of the disease (see Box Typical bladder
symptoms in iNPH). The gait disturbance may accentuate
Headache
the urgency, because it impedes reaching the toilet in time.
Headache is typically found in high-pressure hydrocepha-
In advanced stages, complete urinary incontinence and
lus; however, it is invariably present in iNPH as well.
even fecal incontinence may be present.3 Less commonly,
Exact data about the frequency of headache in NPH are
voiding difficulty is found; 14% of patients with iNPH have
not available, and headache as a symptom of iNPH is
a postvoid residual of more than 100 mL.9
controversial. Although some authors almost neglect
headache, others consider headache to be an important
Typical bladder symptoms in iNPH
NPH symptom, much like Kiefer et al12 who assigned
● High urinary frequency
headaches the same importance as complete urinary
● Urinary urgency
incontinence or severe gait disturbance in the grading
● Complete urinary incontinence
score named after him. Headache is usually felt as “pres-
● Fecal incontinence (rare)
sure in the head.” If headache is present in NPH, then it is
also important to evaluate its improvement after a spinal
tap test as well as after shunting.
Pathophysiologically, the bladder dysfunction in iNPH is
the sequela of a neurogenic bladder dysfunction with
Psychiatric Syndromes
detrusor overactivity, which was noted in 95% of patients.9
Psychiatric syndromes may appear in parallel with iNPH,
The incontinence in iNPH is not as well described in
but, in some cases, they may appear as a symptom of
the literature as compared with the other symptoms of
iNPH, as described in several case reports.3,13-15 Depres-
gait disturbance and dementia.
sion, mania, paranoia, and others were described as
17
Clinical Characteristics and Differential Diagnosis
resolving with shunt treatment; however, the patho-
4.3 Differential Diagnosis
genesis is not clear. Knowledge of psychiatric syndromes
is important, though, because typical symptoms seen in
Many other diagnoses may mimic iNPH (see Box Differen-
NPH may be obscured by psychiatric disorders and ade-
tial diagnosis of iNPH). Knowledge of the differential
quate therapy may be withheld.
diagnoses is essential for making the correct diagnosis as
well as for initiating the correct treatment. To achieve
this, the most important differential diagnoses are
Dizziness/Vertigo
described below, with their differentiation from iNPH.
Often patients complain about dizziness/vertigo, but it is
difficult to objectively assess this uncomfortable feeling.
Differential diagnosis of iNPH
Many clinicians suggest dizziness is the subjective sensa-
● Secondary normal pressure hydrocephalus
tion of gait ataxia and the fear of falling. In fact, it often
● Chronic obstructive hydrocephalus
disappears simultaneously with gait improvement after
● Parkinson disease
shunting. Dizziness is also addressed in the Kiefer grading
● Alzheimer disease
scale of iNPH, reflecting that dizziness is at least one of the
more or less important subjective symptoms of iNPH.12
● Binswanger disease/cerebrovascular disease
● Brain atrophy
However, dizziness may also be a sign of the comorbidities
● Cervical myelopathy
of NPH, particularly cerebral microangiopathy.
● Lumbar spinal canal stenosis
Extended Need of Sleep
Patients with iNPH usually sleep more than healthy peo-
ple, as reported by patients and their relatives, although
4.3.1 Secondary NPH and Chronic
reliable data are missing. Dementia with missing
impulses, interest, and—hence—boredom may be the
Obstructive Hydrocephalus
cause. The raised need for sleep decreases after shunt-
If there are clinical signs of NPH along with a preexisting
ing.16,17
condition that could interfere with cerebrospinal fluid
(CSF) resorption, such as meningitis, subarachnoid hem-
Large Head Circumference
orrhage (SAH), traumatic brain injury (TBI), then we are
referring to secondary NPH (sNPH). However, the causa-
Patients with iNPH often have a significantly larger head
tive factor for NPH remains unclear and is controversial if
circumference compared with the normal population.18
these events were only of a moderate severity and/or
This observation supports the assumption that congenital
occurred many years or even decades ago. Therefore, no
asymptomatic hydrocephalus plays a role in the develop-
clear limit can be defined between iNPH and sNPH.
ment of iNPH at least in some patients.18
In iNPH and sNPH, no direct obstruction of the CSF
pathways can be seen in imaging. This assumes that there
Impairment of Sexual Function
is a communicating hydrocephalus, which is in contrast
Sexual dysfunction in patients with iNPH is often not
to chronic obstructive hydrocephalus, where often an
evaluated and, if present, it is assumed to be a normal
aqueductal stenosis or fourth ventricle outlet obstruction
complaint of an older person. However, in some cases, a
is seen.
regained variable sexual desire may be observed after
treatment.19
Symptoms
Clinical signs are identical in iNPH, sNPH, and other forms
Comorbidities
of chronic communicating hydrocephalus, as also in
Nearly 90% of patients with iNPH have comorbidities such
chronic obstructive hydrocephalus. Gait disturbance,
as cardiovascular diseases, diabetes mellitus, PD, and AD.2
dementia, and incontinence are the leading symptoms in
Sometimes it is difficult to differentiate between the
all these conditions, and they do not allow any differenti-
comorbidity and the primary disease (i.e., AD, cerebro-
ation between the entities.
vascular disease, PD). Frequent coexistence also raises the
question of similar pathogenetic pathways of NPH, AD,
Diagnosis
and cerebrovascular disease. The importance of comor-
bidities was recognized by Kiefer who developed and
Exploration of the patient’s medical and family history, a
introduced the comorbidity index, which suggests a clear
clinical neurologic evaluation, and imaging are necessary.
impact on the outcome after shunting.20,21 Therefore,
Differentiation is only possible by exploring the patient’s
comorbidities of NPH must be recognized because they
history and imaging
(▶ Table
4.1). Patients must be
play a role in further treatment indications.
actively asked for any previous incidence of subarachnoid
18
Clinical Characteristics and Differential Diagnosis
mainly on the patient’s history and neurologic examina-
Table 4.1
Differentiation between iNPH, sNPH, and chronic
obstructive hydrocephalus
tion, and imaging and laboratory tests can serve to rule
out other diseases.24
Symptoms:
History of
Imaging:
gait distur-
SAH, TBI,
CSF pathway
bance,
meningitis,
obstruction
Symptoms
incontinence,
brain surgery
dementia
Cardinal symptoms include tremor, bradykinesia, rigidity,
and postural instability. Tremor is by far the most familiar
iNPH
+
-
-
symptom. With a gradual progression, the symptoms
sNPH
+
+
-
interfere with daily activities and manifest as difficulties
Chronic
+
+ /-
+
in walking, talking, and coordination. Additional symp-
obstructive
hydrocephalus
toms can be depression, difficulty in swallowing, urinary
disorders, and others.
Abbreviations: CSF, cerebral spinal fluid; iNPH, idiopathic normal
pressure hydrocephalus; SAH, subarachnoid hemorrhage; sNPH,
secondary normal pressure hydrocephalus; TBI, traumatic brain
Diagnosis
injury.
The diagnosis of PD relies mainly on the patient’s history
and neurologic examination, and imaging and laboratory
hemorrhage, brain injury, SAH, TBI, brain surgery, menin-
tests can serve to rule out other diseases. Imaging (cranial
gitis, or other diseases that could interfere with CSF
computed tomography, MRI, or both) should help to
resorption capacity.
differentiate PD from NPH, reveal any frontal space
Magnetic resonance imaging
(MRI) should be per-
occupying lesions, and cerebrovascular disease. However,
formed to see the size of the ventricles as well as the CSF
advances in neuroimaging with functional imaging
pathways. Finding aqueductal stenosis, fourth ventricle
approaches such as positron emission tomography (PET)
outlet occlusions, or other CSF pathway obstructions
and single-photon emission computerized tomography
suggests late-onset or chronic obstructive hydrocephalus.
(SPECT) have been successfully employed to detect
A spinal tap test can be performed in all communicat-
25
dopaminergic dysfunction in PD.
ing hydrocephalus subtypes, but the test should be
avoided in cases of clear obstructive hydrocephalus.
Treatment
Treatment
Currently, there is no cure for PD, but a variety of medica-
tions provide substantial symptom relief. The first type of
There is no difference in the treatment of patients with
treatment is levodopa with a decarboxylase inhibitor. Its
sNPH and iNPH. Hydrocephalus shunting is the treatment
effect is not uniform to all symptoms; bradykinesia and
of choice in patients who are symptomatic. However,
rigidity respond best, while tremor may only be margin-
shunting of patients with sNPH seems to have a slightly
ally reduced. Problems with balance and other symptoms
better prognosis than for those with iNPH.22
may not be alleviated at all. Anticholinergics may help
In the NPH subgroup of patients with infratentorial
control tremor and rigidity. In late drug-refractory stages,
intracisternal obstructive hydrocephalus, endoscopic
deep brain stimulation is an option.24
third ventriculostomy (ETV) can be an alternative treat-
ment (see Chapter 12). In chronic obstructive hydroceph-
Differentiating PD from NPH
alus (if the obstruction is distal to the third ventricle),
ETV is the treatment of choice.
If tremor and rigidity are the leading symptoms for PD,
then differentiating between the two conditions seems
easy. If walking difficulties dominate, the arm movement
4.3.2 Parkinson Disease
should be observed; in patients with PD, there is a lack of
PD is a slowly progressing neurodegenerative disorder,
swinging arms during walking, whereas patients with
especially in the elderly population, with a prevalence of
iNPH have preserved their reciprocal arm swing during
1.8% in people older than 65 years.23 It affects movements
gait. Sometimes a virtually paddling action of the arms
or the control of movements, including speech and body
during gait may be observed. Large ventricles in imaging
language. Parkinson-like symptoms (Parkinsonism) may
studies and a positive response to a spinal tap test may
be idiopathic, genetic, or secondary to other entities (e.g.,
make NPH more probable. Alternatively, a good response
brain trauma, cerebrovascular disease, NPH, multiple
to levodopa makes PD more likely; however, because of
system atrophy). The underlying pathomechanism is the
frequent comorbidities, a definite answer can never be
degeneration of dopamine-containing cells of the sub-
achieved through these evaluations alone. The sequela of
stantia nigra, resulting in a lack of dopamine. At this
these difficulties in the differential diagnosis is that sev-
point, treatment with levodopa combined with carbidopa
eral patients with PD get shunt treatment and several
can intervene to improve symptoms. The diagnosis relies
patients with NPH are on medication for PD.
19
Clinical Characteristics and Differential Diagnosis
borderline impairment can be seen in attention, concen-
4.3.3 Alzheimer Disease
tration, and executive functions.3
AD is the most frequent cause of dementia.26 AD is
primarily a degenerative disease of the brain with an
4.3.4 Binswanger Disease
unknown etiology that has characteristic neuropathologic
and neurochemical properties. It usually affects elderly
(Subcortical Vascular Dementia)
people (older than 60 years), and develops subtly and
Binswanger disease is caused by microvascular degenera-
progresses slowly over time.27
tion, which, in turn, causes deep white matter and peri-
ventricular damage. Changes in the white matter lead
Symptoms
to a disruption of the subcortical neural circuits that
control executive cognitive functioning. Risk factors
Loss of cognitive functioning with memory disturbance,
disorientation, and other symptoms in varying degrees
of Binswanger disease include arterial hypertension,
are characteristic of AD. Usually the symptoms are pro-
diabetes, and smoking. It is a disease seen in the elderly
gressive, beginning at the preclinical stage, at which point
population and is usually progressive.32
they are normally not detected, and then they progress
into mild cognitive impairment and dementia, resulting
Symptoms
in a complete dependency on others to perform basic
Psychomotor slowness is the most characteristic feature
activities of daily living.
of Binswanger disease. Disturbance of short-term mem-
ory, mood, and attention is often seen.32 Unsteady gait
Diagnosis
and urinary symptoms, not caused by urologic diseases,
History and psychometric tests, especially those that
can appear.32
focus on memory, attention, counting, and language, may
help to substantiate the suspicion of AD. Lumbar punc-
Diagnosis
ture with protein detection can help ensure the diagno-
Diagnosis is made by the combination of the described
sis: the level of tau protein in CSF is increased and the
symptoms, with white matter and periventricular changes
level of β-amyloid is decreased in patients with AD.28
best shown in MRI. Imaging is essential for the diagnosis.
Imaging (MRI, CT, or both) may help to rule out other
Other diseases such as NPH should be ruled out.
causes of dementia, such as cerebrovascular disease, NPH,
chronic subdural hematomas, or brain tumors. SPECT will
reveal any metabolic changes in the temporal region.
Treatment
Combining clinical and neuropsychological symptoms,
There is no specific treatment available for Binswanger
and CSF concentration of tau and β-amyloid proteins with
disease. Treatment goals must focus on alleviation of
imaging, especially SPECT (which reveals temporal meta-
symptoms. Memantine may delay the progression by reg-
bolic changes), can predict AD with high reliability29;
ulating the glutamatergic system. Ventriculoperitoneal
histopathologically, it can be proven through the pres-
shunting may be effective with concomitant hydrocepha-
ence of amyloid plaques and neurofibrillary tangle.30
lus.33 To prevent Binswanger disease, risk factors such as
hypertension, diabetes and smoking should be controlled
Treatment
as early as possible.32
There is no current treatment that can cure or substan-
tially delay the disease over the long term. Therefore, cur-
Differentiating Binswanger Disease
rent approaches focus on helping people maintain mental
from NPH
function, manage behavioral symptoms, and slow or
delay the symptoms of the disease.26 Medical treatment
The presence of large ventricles makes iNPH the more
for AD is aimed at inhibiting cholinesterase or glutamate;
probable diagnosis, while predominant white matter
however, these effects are limited. Ventriculoperitoneal
changes make Binswanger disease more probable. However,
shunting has been tried, to wash out accumulated pro-
the high-level coexistence of iNPH with cerebrovascular
teins in CSF; however, a significant effect on the course
changes makes the differentiation extremely difficult.
of AD could not be achieved.31
The spinal tap test and CSF dynamics are helpful, but not
absolutely reliable.33
Differentiating AD from NPH
4.3.5 Brain Atrophy
In AD, cognitive impairment affects mainly memory,
learning, orientation, attention, concentration, executive
Brain atrophy is described as a loss of brain tissue or neu-
function, and writing. In iNPH, psychomotor slowing is
ron cells that may be generalized or partial (focal). Quite
the most prominent psychological symptom, whereas
often it is the sequela of a brain disease such as AD or
20
Clinical Characteristics and Differential Diagnosis
Binswanger disease, or chronic alcohol abuse, or others.
bladder dysfunction with incontinence may develop.
Brain atrophy is a description of the imaging findings and
When a spinal puncture in a patient with iNPH is difficult,
not a diagnosis per se. The underlying cause must be
lumbar spinal canal stenosis must be ruled out.
determined. The neuroradiologic
“diagnosis” of brain
atrophy and the uncritical use of this term may mislead
Symptoms
physicians to reject further investigation, thus causing
patients with a treatable disease such as iNPH to have
Typically, patients present with spinal (neurogenic) clau-
beneficial therapy withheld.
dication, leg pain, and numbness or weakness in the legs
after walking a long distance. Step length can be reduced,
but shuffling is not usually present. Patients recover
4.3.6 Cervical Myelopathy
when bending over or changing position (sitting or lying
Gait disturbance often occurs in the elderly and may
down). In contrast to walking problems, cycling is usually
coexist with “senile” dementia. Cervical myelopathy with
not impaired. In advanced cases, walking may be
impossible and incontinence may be present due to cauda
gait ataxia and urinary problems is frequently seen in
equina compression.
degenerative cervical spondylosis with spinal canal ste-
nosis. The disease is usually progressive and can be easily
confused with iNPH, with predominant gait and urinary
Diagnosis
disturbances. However, in combination with imaging and
Imaging of the spine (MRI or CT) will show the spinal
dynamic CSF tests, a diagnosis can be confirmed in most
canal stenosis. In individual cases with concomitant ven-
cases.
triculomegaly, a spinal tap test can reveal the predomi-
nant diagnosis.
Symptoms
Frequently, patients with cervical myelopathy have a his-
Differentiating Lumbar Spinal Canal
tory of neck pain. Gait disturbance is characterized by spi-
Stenosis from iNPH
nal ataxia, which is an ataxia that substantially deteriorates
in the dark or with the eyes closed. In advanced cases, a
Symptoms of iNPH normally do not depend on walking
spasticity with elevated muscle tendon reflexes can be
distance. In severe cases where patients are already
found. Urinary problems are often described, with urgency
immobile, inquiring about the medical history of a
and incontinence occurring in progressive disease.
patient may reveal a relationship with walking distance
in previous stages of the disease. A lack of improvement
after a spinal tap test—preferably in a higher level than
Diagnosis
the stenosis—may indicate a symptomatic spinal canal
Imaging of the spine (MRI or CT) will show cervical spon-
stenosis.
dylosis with spinal canal stenosis, with eventual edema
or gliosis of the spinal cord. However, in individual cases,
4.4 Summary
especially with concomitant ventriculomegaly, the differ-
entiation may be difficult. A spinal tap test may reveal
Becoming aware and knowledgeable of the symptoms of
elevated protein levels found in spinal CSF obstructions
iNPH is essential to diagnose the condition. Of course,
and there may be no improvement after tapping.
iNPH must be differentiated from other conditions that
have similar symptoms, and imaging must be interpreted
Differentiating Cervical Myelopathy
cautiously. However, suspicion of iNPH should never be
carelessly neglected even if other conditions are present.
from iNPH
Health care professionals must be aware of the diseases
In cervical myelopathy, gait ataxia substantially deterio-
that frequently coexist with iNPH. Only with this attitude
rates with the eyes closed. Muscle tendon reflexes and
will we not wrongly refer patients for shunting, and
muscle tone of the lower limbs are usually elevated; in
conversely, we will not shunt inappropriate patients.
addition, pyramidal signs (Babinski reflex) may be pres-
ent, which is usual for iNPH. A spinal tap test will show
no improvement in myelopathy.
References
[1] Hakim S, Adams RD. The special clinical problem of symptomatic
hydrocephalus with normal cerebrospinal fluid pressure. Observa-
4.3.7 Lumbar Spinal Canal Stenosis
tions on cerebrospinal fluid hydrodynamics. J Neurol Sci
1965;
2: 307-327
Gait disturbance in lumbar spinal canal stenosis usually
[2] Bech-Azeddine R, Høgh P, Juhler M, Gjerris F, Waldemar G. Idiopathic
deteriorates with walking distance and is normally associ-
normal-pressure hydrocephalus: clinical comorbidity correlated with
ated with back and leg pain. Lumbar spinal canal stenosis
cerebral biopsy findings and outcome of cerebrospinal fluid shunting.
is often found in the elderly population. In severe stenosis,
J Neurol Neurosurg Psychiatry 2007; 78: 157-161
21
Clinical Characteristics and Differential Diagnosis
[3]
Relkin N, Marmarou A, Klinge P, Bergsneider M, Black PM. Diagnosing
only a subset of patients. J Neurol Neurosurg Psychiatry 2007;
idiopathic normal-pressure hydrocephalus. Neurosurgery 2005; 57
78: 508-511
(Suppl): S4-S16, discussion ii-v
[19]
Missori P, Scollato A, Formisano R et al. Restoration of sexual activity
[4]
Toma AK, Stapleton S, Papadopoulos MC, Kitchen ND, Watkins LD.
in patients with chronic hydrocephalus after shunt placement. Acta
Natural history of idiopathic normal-pressure hydrocephalus.
Neurochir (Wien) 2009; 151: 1241-1244
Neurosurg Rev 2011; 34: 433-439
[20]
Kiefer M, Eymann R, Steudel WI. Outcome predictors for normal-
[5]
Hughes CP, Siegel BA, Coxe WS et al. Adult idiopathic communicating
pressure hydrocephalus. Acta Neurochir Suppl
(Wien) 2006; 96
hydrocephalus with and without shunting. J Neurol Neurosurg
(Suppl): 364-367
Psychiatry 1978; 41: 961-971
[21]
Meier U, Lemcke J. Co-morbidity as a predictor of outcome in patients
[6]
Dauch WA, Zimmermann R. [Normal pressure hydrocephalus. An
with idiopathic normal-pressure hydrocephalus. Acta Neurochir
evaluation 25 years following the initial description]. Fortschr Neurol
Suppl (Wien) 2010; 106: 127-130
Psychiatr 1990; 58: 178-190
[22]
Børgesen SE. Conductance to outflow of CSF in normal pressure
[7]
Stolze H, Kuhtz-Buschbeck JP, Drücke H et al. Gait analysis in idio-
hydrocephalus. Acta Neurochir (Wien) 1984; 71: 1-45
pathic normal pressure hydrocephalus—which parameters respond
[23]
to the CSF tap test? Clin Neurophysiol 2000; 111: 1678-1686
[8]
Kuba H, Inamura T, Ikezaki K et al. Gait disturbance in patients with
[24]
low pressure hydrocephalus. J Clin Neurosci 2002; 9: 33-36
[9]
Sakakibara R, Uchiyama T, Kanda T, Uchida Y, Kishi M, Hattori T. [Uri-
on April 15, 2013
nary dysfunction in idiopathic normal pressure hydrocephalus].
[25]
Pavese N, Brooks DJ. Imaging neurodegeneration in Parkinson’s dis-
Brain Nerve 2008; 60: 233-239
ease. Biochim Biophys Acta 2009; 1792: 722-729
[10]
Hakim CA, Hakim R, Hakim S. Normal-pressure hydrocephalus. Neu-
[26]
rosurg Clin N Am 2001; 12: 761-773, ix
[11]
AWMF Guidelines Normal Pressure Hydrocephalus. http://www.
[27]
[12]
Kiefer M, Eymann R, Komenda Y, Steudel WI. [A grading system for
[28]
Sunderland T, Linker G, Mirza N et al. Decreased beta-amyloid1-42
chronic hydrocephalus]. Zentralbl Neurochir 2003; 64: 109-115
and increased tau levels in cerebrospinal fluid of patients with
[13]
Pinner G, Johnson H, Bouman WP, Isaacs J. Psychiatric manifestations
Alzheimer disease. JAMA 2003; 289: 2094-2103
of normal-pressure hydrocephalus: a short review and unusual case.
[29]
Schmidt D, Zimmermann R, Lewczuk P et al. Confirmation rate of
Int Psychogeriatr 1997; 9: 465-470
blinded (99m)Tc-SPECT compared to neurochemical dementia bio-
[14]
Price TR, Tucker GJ. Psychiatric and behavioral manifestations of nor-
markers in CSF in patients with Alzheimer disease. J Neural Transm
mal pressure hydrocephalus. A case report and brief review. J Nerv
2010; 117: 1111-1114
Ment Dis 1977; 164: 51-55
[30]
Tiraboschi P, Hansen LA, Thal LJ, Corey-Bloom J. The importance of
[15]
Rosen H, Swigar ME. Depression and normal pressure hydrocephalus.
neuritic plaques and tangles to the development and evolution of AD.
A dilemma in neuropsychiatric differential diagnosis. J Nerv Ment Dis
Neurology 2004; 62: 1984-1989
1976; 163: 35-40
[31]
Silverberg GD, Mayo M, Saul T, Fellmann J, Carvalho J, McGuire D.
[16]
Hellström P, Edsbagge M, Blomsterwall E et al. Neuropsychological
Continuous CSF drainage in AD: results of a double-blind, random-
effects of shunt treatment in idiopathic normal pressure hydrocepha-
ized, placebo-controlled study. Neurology 2008; 71: 202-209
lus. Neurosurgery 2008; 63: 527-535, discussion 535-536
[32]
National Institute of Neurological Disorders and Stroke. http://www.
[17]
Tisell M, Hellström P, Ahl-Börjesson G et al. Long-term outcome in
109 adult patients operated on for hydrocephalus. Br J Neurosurg
April 15, 2013
2006; 20: 214-221
[33]
Tisell M, Tullberg M, Hellström P, Edsbagge M, Högfeldt M, Wikkelsö
[18]
Wilson RK, Williams MA. Evidence that congenital hydrocephalus
C. Shunt surgery in patients with hydrocephalus and white matter
is a precursor to idiopathic normal pressure hydrocephalus in
changes. J Neurosurg 2011; 114: 1432-1438
22
5.1
What Causes iNPH? Theories
Chapter 5
Proposed by the Discoverers and
the Bulk Flow Theory
24
Pathophysiology
5.2
Inner Contradictions
25
Johannes Lemcke, Ullrich Meier
5
Pathophysiology
5 Pathophysiology
Johannes Lemcke, Ullrich Meier
This … leads to the conclusion that the pressure produced
5.1 What Causes iNPH?
by retention of fluid was not the major force causing
ventricular enlargement, and that some other local force
Theories Proposed by the
must have been responsible for the marked variation in
Discoverers and the Bulk
ventricular size.
(Edgar A. Bering Jr., 1962)
Flow Theory
When Hakim and Adams1,2 described normal pressure
hydrocephalus (NPH) for the first time in 1965 and
The oldest ideas regarding the pathophysiology of iNPH
developed the first theories regarding its patho-
can be traced back to the discoverers of its clinical pic-
physiology, they could not have suspected that a clini-
ture: Adams1 and Hakim.14 The underlying concept of the
cal picture of reversible dementia in old age would
hydraulic press theory proposed by these authors is
continually gain in importance simply because of the
based on the proposal that the physiologic sites of CSF
demographic development in Western societies. Today,
production and reabsorption are to be equated with the
just as then, fundamental questions with regard to the
choroid plexus or Pacchionian granulations. This so-
precise pathogenic mechanism of idiopathic NPH
called bulk flow theory was disseminated at that time
(iNPH) have still not been answered. From the view-
almost without exception. It can be traced back to, among
point of the clinician, iNPH still constitutes a black
others, Walter E. Dandy
(1886-1946) who published
box. Although certain input and output variables are
experiments in 1914 in which he obstructed the easily
known and, with the implantation of a cerebrospinal
accessible cerebral aqueduct in dogs with cotton fila-
fluid
(CSF) shunt or third ventriculostomy, there are
ments and then observed an expansion of the ventricular
also certain control mechanisms, the true pathogenetic
system.15 He came to the obvious conclusion from his
process and the precise point of attack for therapy
observations that the production of CSF must be intra-
have not been determined. Even though model systems
ventricular and its reabsorption must be extraventricular.
of known mechanisms have been proposed—for exam-
Therefore, according to these considerations, the site of
ple, that of Egnor et al3—none of the working groups
production is the choroid plexus and the site of
currently publishing worldwide has offered a regula-
reabsorption is the Pacchionian granulations in the vicin-
tory circuit with a precise identification of the control
ity of the apex. Contemporaries of Hakim and Adams also
and regulatory factors. On the contrary, fundamental
provided support for this idea with highly modernized
questions about iNPH remain largely unanswered,
experiments (for the time) using radioisotopes. Accord-
namely:
ingly, among others, Di Chiro and Grove16,17 and Kieffer et
● Is it an entity in its own right, or do we include symp-
al18
observed an accumulation of cisternally injected
toms and secondary pathogenic mechanisms with dif-
radioisotopes (99Tc or198Au) in the region of the Pacchio-
ferent geneses under the term iNPH4,5?
nian granulations after approximately 24 hours.
● Is iNPH only a form of expression or degree of severity
The bulk flow theory postulates a CSF flow from the
of a more basic superordinate disease6-13?
choroid plexus in the lateral ventricle over the foramina
● Does the essential therapeutic effect of a CSF shunt
of Monro in the third ventricle, via the aqueduct in the
or third ventriculostomy really lie in the effective
fourth ventricle and, from there, over the foramina of
outflow of CSF from the ventricles, or is it in fact
Luschka in the complete subarachnoid space. Then, via
produced by the composition of the CSF because of
the arachnoid granulations, reabsorption takes place in
an increased turnover or a pulse-synchronous tran-
the sinus. Based on the bulk flow theory, it is possible to
sient outplacement of a proportion of the CSF from
clarify all obstructive forms of hydrocephalus without dif-
the ventricular system?
ficulty. However, the explanation of nonobstructive forms
of hydrocephalus with this model is complicated.
The authors are unable to provide an unreservedly
Hakim1 and Adams,14 who, as mentioned before, based
provable answer to any of these questions. However, if
their hydraulic press theory on the bulk flow theory, had
we try to understand, step by step, the theoretical
to take a conceptual circumlocution to explain the iNPH
considerations from past decades, then a network of
discovered by them within the scope of the model. They
factors results, which brings us much further in our
assumed a disturbance of CSF reabsorption in the region
search for an understanding of the pathophysiology of
of the arachnoid granulation that, first of all, results in an
iNPH.
increase in drainage resistance (Rout) and, with that, an
24
Pathophysiology
initial concomitant increase in intracranial pressure.1,14
circulation of CSF in 1962 in an article published in the
Thereupon, there should be a pressure-dependent pro-
Journal of Neurosurgery.21
gression of the ventricle width in an “unobserved time
With the possibility of using noninvasive magnetic res-
window.” The subsequent normal intracranial pressure is,
onance tagging, and relatively and precisely recording
according to Adams1 and Hakim,14 explained by Pascal’s
blood and CSF movements, the invasive considerations
law. From this, they concluded that the force with which
from animal experiments that were carried out in the
a fluid acts on the surface is a direct pressure; however,
1960s and 1970s were translated into the modern reper-
the force is indirectly disproportional to this surface and
toire. Pioneering thoughts regarding these techniques
correspondingly declines. They graphically portrayed this
were developed by Greitz25-27 who may be considered to
in their work as air balloons and manometers, and the
be the founder of the hydrodynamic concept. The signifi-
pressure in the already dilated ventricles then declines.1,14
cant moment of this concept was the circumstance that,
The result is known clinically as an expanded ventricle
in healthy individuals, because of the elasticity of the
system with normal intracranial pressure.
walls of the basilar arteries, a so-called Windkessel effect
arises.
During the flow of blood in the systole, the amplitude
5.2 Inner Contradictions
of the pulsatile blood flow exceeding the average flow is
The concept of the hydraulic press theory based on the
absorbed by a pressure-induced widening of the vessel's
bulk flow theory was, however, not satisfactory even at
cross-section and is momentarily “stored.” This absorbed
that time because it possessed a few very unlikely
blood volume is then driven forth in the systole. From
implications. Consequently, it was difficult for clinicians
this, after entry of the vessels into the cerebral paren-
to understand why the phase of greatly increased intra-
chyma in the capillary bed, an almost laminar perfusion
cranial pressure, which results in ventricular expansion
of the cerebral parenchyma with blood is produced. The
and should take place free of symptoms and the com-
blood pulse-dependent changes in volume of the entire
mencement of complaints, should first occur in the
parenchyma are kept within limits in healthy individuals,
phase of renewed normalized intracranial pressure. In
meaning that only a very small discharge of CSF of
addition, the concepts intersect with experimental
0.03 mL/systole through the aqueduct is needed because
observations that had already been investigated by con-
of the systolic increase in volume of the parenchyma.27
temporaries of the discoverers of the clinical picture
The volume requirement for the CSF, which is caused by
and in which dynamic—not static—pressure is in the
the systolic widening of the cerebral arteries, is, in con-
foreground.
trast, compensated via a CSF outflow over the foramen
Accordingly, in an experiment using dogs in 1962,
magnum, as well as a momentary venous compression of
Bering19-21 undertook a unilateral plexectomy following
the bridging veins. If now, because of arteriosclerotic ves-
the induction of a communicating hydrocephalus using a
sel disease, the Windkessel effect no longer occurs as a
Kaolin installation, without limiting the communication
result of the rigid vessel walls in the basilar arteries, then
between the ventricles in any form. In the subsequent
the systolic pulse wave will flow unimpeded into the cap-
measurements, he found identical average pressures in
illary bed in the parenchyma. At the same time, the con-
both lateral ventricles; however, he also recorded pulse
secutive CSF pulse in the subarachnoid space fails to
amplitudes that massively differed from one another in
appear, which, in the physiologic case, results in a com-
the CSF pressure curves. Only the lateral ventricle dilated
pression of the bridging veins, so that a momentary
with an intact choroid plexus.
reduction in the venous discharge from the parenchyma
In fact, plexectomies were already being contemplated
occurs.28,29
Therefore, the arterial pulse wave flows
in the early days of neurosurgery as a therapeutic option
unimpeded through the entire parenchyma and leads to
for hydrocephalic constellations. But, at that time, the
a short-term increase in volume of the cerebral paren-
simple idea behind this was to disrupt CSF production
chyma during the systole. In the subarachnoid space, this
using this maneuver.22
can be compensated by a discharge of CSF through the
In contrast, Bering’s experiment opened the door to a
foramen magnum so that there is no significantly
completely new approach to explaining the effect of pul-
increased counterpressure for the cortical areas of the
sating pressure on ventricle size. In an experiment pub-
brain. This does not apply to the parenchyma region near
lished in 1967 by Wilson and Bertan,23,24 the concept was
the ventricle.
further deepened in this direction. Likewise, they instilled
In accordance with the Hagen-Poiseuille law, volume
carbon black and thereby induced a communicating
flow into narrow tubes is inversely proportional to the
hydrocephalus in dogs. They did not undertake a plexec-
fourth power of the radius. A sufficient volume of CSF
tomy, but they did unilaterally obliterate the choroid
cannot escape during the short duration of the systole,
artery. Here a unilateral hydrocephalus was also manifest.
and this results in a situation in which the parenchyma
The two experimenters drew on work from Bering,
region near the ventricle adds to the incompressible ven-
much cited at the time, who initiated a rethinking of the
tricle volume once more with every pulse wave. In this
25
Pathophysiology
manner, a degeneration of the parenchyma near the ven-
[8]
Golomb J, Wisoff J, Miller DC et al. Alzheimer’s disease comorbidity
in normal pressure hydrocephalus: prevalence and shunt response. J
tricle, and consequently a “passive” widening of the ven-
Neurol Neurosurg Psychiatry 2000; 68: 778-781
tricle without the need for a static increase in pressure,
[9]
Kapaki EN, Paraskevas GP, Tzerakis NG et al. Cerebrospinal fluid tau,
can be explained.
phospho-tau181 and beta-amyloid1-42 in idiopathic normal pres-
Egnor3 has drawn attention to the fact that the effect of
sure hydrocephalus: a discrimination from Alzheimer’s disease. Eur J
the pulsating choroid plexus, which was already known
Neurol 2007; 14: 168-173
[10]
Kudo T, Mima T, Hashimoto R et al. Tau protein is a potential biologi-
from experiments carried out in the 1960s, must be
cal marker for normal pressure hydrocephalus. Psychiatry Clin
added. As the first outlet of the internal carotid
Neurosci 2000; 54: 199-202
artery, the anterior choroid artery, in the absence of a
[11]
Laske C, Stransky E, Leyhe T et al. BDNF serum and CSF concentra-
Windkessel effect, is naturally also perfused by a strongly
tions in Alzheimer’s disease, normal pressure hydrocephalus and
healthy controls. J Psychiatr Res 2007; 41: 387-394
pulsating blood current. The amplitude of the pulse
[12]
Silverberg GD, Mayo M, Saul T, Rubenstein E, McGuire D. Alzheimer’s
reaches the choroid plexus at approximately the same
disease, normal-pressure hydrocephalus, and senescent changes in
time as the pulse amplitude, which already makes up the
CSF circulatory physiology: a hypothesis. Lancet Neurol 2003; 2:
increase in volume in the parenchyma so that the paren-
506-511
chyma near the ventricle not only impinges on the
[13]
Silverberg GD, Levinthal E, Sullivan EV et al. Assessment of low-flow
CSF drainage as a treatment for AD: results of a randomized pilot
incompressible cerebrospinal volumes in the ventricles
study. Neurology 2002; 59: 1139-1145
but also, at the same time, increases this volume as a
[14]
Hakim S, Adams RD. The special clinical problem of symptomatic
result of the pathologic pulsating perfusion of the choroid
hydrocephalus with normal cerebrospinal fluid pressure. Observations
plexus with the same rhythm. Consequently, the latest
on cerebrospinal fluid hydrodynamics. J Neurol Sci 1965; 2: 307-327
deliberations bridge the divide to the experimental
[15]
Dandy WE. Experimental hydrocephalus. Ann Surg
1919;
70:
129-142
findings that had already been obtained when iNPH was
[16]
Di Chiro G, Grove AS. Evaluation of surgical and spontaneous cerebro-
discovered. The theory is compatible with clinical experi-
spinal fluid shunts by isotope scanning. J Neurosurg 1966; 24: 743-748
ence that teaches us that iNPH is a disease occurring in
[17]
Di Chiro G. Observations on the circulation of the cerebrospinal fluid.
the third phase of life and that cardiovascular disease,
Acta Radiol Diagn (Stockh) 1966; 5: 988-1002
[18]
Kieffer SA, Stadlan EM, D’Angio GJ. Anatomic studies of the distribu-
diabetes, and stroke are the most frequent comorbidities
tion and effects of intrathecal radioactive gold. Acta Radiol Ther Phys
of the condition. However, it also leaves some questions
Biol 1969; 8: 27-37
unanswered. Consequently, the precise cerebral damage
[19]
Bering EA. Cerebrospinal fluid. Prog Neurol Psychiatry 1966; 21:
mechanism that results in the still clinically relevant
358-373
Hakim triad, with gait disorders, urinary incontinence,
[20]
Bering EA. Choroid plexus and arterial pulsation of cerebrospinal
fluid; demonstration of the choroid plexuses as a cerebrospinal fluid
and dementia, has been clarified neither at the topo-
pump. AMA Arch Neurol Psychiatry 1955; 73: 165-172
graphical level nor at the cellular level.
[21]
Bering EA. Circulation of the cerebrospinal fluid. Demonstration of
the choroid plexuses as the generator of the force for flow of fluid
and ventricular enlargement. J Neurosurg 1962; 19: 405-413
References
[22]
Dandy WE. Extirpation of the choroid plexus of the lateral ventricles
in communicating hydrocephalus. Ann Surg 1918; 68: 569-579
[1] Adams RD, Fisher CM, Hakim S, Ojemann RG, Sweet WH. Sympto-
[23]
Wilson CB, Bertan V. Interruption of the anterior choroidal artery in
matic occult hydrocephalus with “normal” cerebrospinal-fluid pres-
experimental hydrocephalus. Arch Neurol 1967; 17: 614-619
sure. A treatable syndrome. N Engl J Med 1965; 273: 117-126
[24]
Wilson CB, Bertan V. Role of the anterior choroidal artery in hydro-
[2] Hakim CA, Hakim R, Hakim S. Normal-pressure hydrocephalus.
cephalus. Surg Forum 1965; 16: 438-440
Neurosurg Clin N Am 2001; 12: 761-773, ix
[25]
Greitz D. The hydrodynamic hypothesis versus the bulk flow hypoth-
[3] Egnor M, Zheng L, Rosiello A, Gutman F, Davis R. A model of pulsa-
esis. Neurosurg Rev 2004; 27: 299-300
tions in communicating hydrocephalus. Pediatr Neurosurg 2002; 36:
[26]
Greitz D. Paradigm shift in hydrocephalus research in legacy of
281-303
Dandy’s pioneering work: rationale for third ventriculostomy in com-
[4] Brecknell JE, Brown JI. Is idiopathic normal pressure hydrocephalus
municating hydrocephalus. Childs Nerv Syst 2007; 23: 487-489
an independent entity? Acta Neurochir (Wien) 2004; 146: 1003-1006,
[27]
Greitz D. Radiological assessment of hydrocephalus: new theories
discussion 1006-1007
and implications for therapy. Neurosurg Rev 2004; 27: 145-165,
[5] Brown JI, Brecknell JE. Is idiopathic normal pressure hydrocephalus
discussion 166-167
an independent entity? Acta Neurochir (Wien) 2005; 147: 803-804
[28]
Bateman GA. The reversibility of reduced cortical vein compliance
[6] Chakravarty A. Unifying concept for Alzheimer’s disease, vascular
in normal-pressure hydrocephalus following shunt insertion.
dementia and normal pressure hydrocephalus - a hypothesis. Med
Neuroradiology 2003; 45: 65-70
Hypotheses 2004; 63: 827-833
[29]
Bateman GA, Levi CR, Schofield P, Wang Y, Lovett EC. The venous
[7] George AE, Holodny A, Golomb J, de Leon MJ. The differential diagno-
manifestations of pulse wave encephalopathy: Windkessel dys-
sis of Alzheimer’s disease. Cerebral atrophy versus normal pressure
function in normal aging and senile dementia. Neuroradiology 2008;
hydrocephalus. Neuroimaging Clin N Am 1995; 5: 19-31
50: 491-497
26
6.1
Evaluation of the Patient’s
Chapter 6
Medical History
28
Noninvasive Diagnostic
6.2
Clinical Examination
28
Work-up
6.3
Summary
33
Uwe Kehler
6
Noninvasive Diagnostic Work-up
6 Noninvasive Diagnostic Work-up
Uwe Kehler
A diagnostic work-up always begins with the medical his-
The diagnostic work-up is based on the patient’s medi-
tory and a clinical examination of the patient. However,
cal history, and complete clinical examination with an
the latter remains very subjective and is very much
assessment of gait disorders, incontinence, and mental
dependent on the examining physician. Gait and neuro-
impairment.
psychological tests can objectify and grade the symptoms
and make these comparable during follow-up as well as
6.1 Evaluation of the Patient’s
with other patients. Of course, imaging is also essential in
idiopathic normal pressure hydrocephalus (iNPH) and is
Medical History
described in Chapter 7.
“There is no accepted standard for this topic” is a state-
As with any disease, a complete medical history is neces-
ment from guidelines that address iNPH,1 and it is also
sary. If normal pressure hydrocephalus (NPH) is suspected,
valid for the diagnostic work-up of iNPH. Multiple tests
then questioning must go into more detail providing exact
for walking, balance, and neuropsychological evaluation
description of the symptoms. Patients and/or their rela-
(fewer for incontinence) are available; however, there is
tives should describe the symptoms and complaints. Ask
no common agreement about which test is most relevant
about any urinary problems because patients often do not
for diagnostic purposes and which test is best for deter-
mention them because of a sense of shame. In addition,
mining treatment efficacy. Because gait disturbances
the neuropsychological situation should be evaluated; it is
react more extensively and more quickly to spinal tap
here that an interview with the patient’s relatives is of
tests and shunting than do incontinence and dementia,
great importance because they might perceive behavioral
gait tests are of high clinical importance when evaluating
changes, memory, and concentration-attention distur-
the efficacy of diagnostic spinal tap tests and shunting.
bances that are different from those reported by the
Various tests are applied in different departments, thus
patient. As the disease becomes more advanced, especially
making comparisons very difficult. Many tests are
when dementia develops, further information from rela-
extremely time-consuming, which result in restrained
tives is very important. Ask about minor symptoms of
performance in daily routines. However, for scientific
NPH such as headache, dizziness, and an increased need
evaluations, it is worthwhile to perform them because
of sleep, among others. The physician must also check dif-
they will enable us to learn more about their relevance in
ferential diagnoses and comorbidities.
iNPH as well as about the condition itself. Some tests
seem to show only general deviations from normal,
6.2 Clinical Examination
whereas others may show iNPH-specific changes. Several
tests can be performed only in patients who are able to
The clinical examination of the patient should concen-
collaborate sufficiently. It is obvious that gait tests require
trate on the typical NPH triad, with an exact description
certain mobility and neuropsychological tests require cer-
of the clinical findings. These should be completed by gait
tain cooperation, thus excluding the testing of patients
and neuropsychological tests to quantify the deficits.
with severe dementia.
A selection of widely used tests, with short comments
Normal values for diagnostic tests are often critical
relating to their clinical value and practicability, is
because they are for otherwise healthy patients
presented below.
without comorbidities: What is the normal walking
speed for a patient aged 80 years with coxarthrosis
6.2.1 Evaluating Gait Disturbance
or Parkinson disease? What are the normal values of
psychometric tests if the patient also has Binswanger
Gait disturbance is the most important clinical feature of
disease? Even with these tests, it is still important
iNPH and the symptom with the best prognosis after
to follow up with individual patients to evaluate
treatment. Substantial improvement is often observed
diagnostic and therapeutic efforts.
only hours after a spinal tap test. Therefore, gait is not
Symptoms are often subtle and differences are not eas-
only a sensitive marker of the severity of NPH, but it also
ily detected even on using sophisticated tests. Therefore,
serves as a sensitive marker of the efficacy of the spinal
reports from the patients, and from their relatives who
tap test and shunting. Deteriorations in gait during fol-
witness the everyday life of the patients, must be consid-
low-up may trigger a new investigation for suspected
ered. This information plays a special role during follow-
shunt failure. Thus, this emphasizes the importance of a
up when objective measures cannot demonstrate any
good description of the gait disturbance as well as quanti-
substantial difference.
fication and scoring.
28
Noninvasive Diagnostic Work-up
It is worthwhile to perform a video recording, espe-
Gait Description
cially for detecting subtle differences in gait after a spinal
The physician may describe the gait as slow, broad-based,
tap test or after treatment. It is reproducible and provides
atactic, and shuffling (see Section 6.2.1). A description
an opportunity to demonstrate the effect of a spinal tap
such as this should raise suspicion of NPH and a descrip-
test or shunting to others, independent of the physician.
tion should always be present in the patient chart. How-
The disadvantage is that it is a time-consuming proce-
ever, a description alone is subjective and not sufficient to
dure; however, it is very reliable.
detect slight differences (i.e., after a spinal tap test). In
The routine video recording of a timed up-and-go test
addition, it is very much dependent on the examining
is desirable for all patients. However, if no sufficient time
physician and is difficult to compare with descriptions
is available in daily clinical routine, the turn, gait speed,
from other examiners. Therefore, it must be described
and the timed up-and-go tests are valuable and reliable
using tests that make the gait disturbance measurable
tests for gait evaluation.
and comparable. A selection of tests and gait discriptions
are presented below.
6.2.2 Evaluating Incontinence
Step Length
The task of the physician is to obtain an adequate medical
history of urinary problems despite the fact that the
One way of measuring step length is a description of
patient might feel a sense of shame. In particular, the
the length of steps compared with the length of a
physician must ask about pollakiuria, urgency, and
patient’s foot (e.g., step length = one-half of foot length).
incontinence, as well as fecal incontinence, as information
It is a more objective parameter than the verbal
about these is necessary to detect bladder and bowel
description. However, it may vary substantially, and
problems.
therefore it can be accepted only as a supplement for
Neurological and urological examinations should rule
other tests.
out other causes of incontinence such as cauda equina
syndrome, urinary tract infection, and others.
180°/360° Turn
The patient is asked to turn 180° or 360° with as few
6.2.3 Neuropsychological Testing
steps as possible; normal values: 2 to 3 steps for 180°, 4
to 5 steps for 360° turn. If the patient understands this
There are a variety of neuropsychological tests availa-
task, then it is an easily reproducible, quickly performed
ble, and there are many modifications of the original
test that is good for use during follow-up, and is indepen-
tests performed in clinical work. However, there is
dent of the physician.
no general agreement as to which is the best test
for identifying, measuring, and following up mental
Gait Speed (10 m)
disorders. Intensive neuropsychological testing is time-
consuming and not practical for routine use in a hos-
The time (and steps) needed to cover a distance of 10 m
pital. However, for scientific purposes, it is worthwhile
(marked on the floor) with normal walking is measured.
to perform extensive testing with the aim of learning
It is a good reproducible parameter, but it is difficult to
more about the neuropsychological pathology of
perform in some places (e.g., there is not always enough
iNPH. In addition, relevant and suitable items of
space).
neuropsychological tests could be developed for
shorter routine tests.
Timed Up-and-Go Test
For routine testing in a multicultural/multilinguistic
The sitting patient is asked to get up from his or her chair,
environment, language-independent tests or multi-
walk 3 m, turn, go back to the chair, and sit down again.
lingual tests are needed, so that some patients are not
Normal time: < 10 seconds. This is a complex task with
excluded. A neuropsychological expert is often not avail-
different actions; it is reproducible, and superior to deter-
able; therefore, testing should be sufficiently easy for
mining gait speed only.2
general physicians to perform the neuropsychological
work-up.
The widely used mini mental state examination
Video Recording of Normal Gait, Turn, or
(MMSE; see below) is often performed in patients with
Timed Up-and-Go Test
iNPH; however, it “measures” cortical dementias rather
Walking and different tasks are recorded on video, so that
than subcortical dementias (as is the case in iNPH) and is,
determinations can be repeated indefinitely, and analyses
therefore, less specific for iNPH. Nevertheless, patients
can be performed later, as well as time measurements for
with NPH perform significantly worse in the MMSE than
the different recorded tasks.
healthy individuals and show significant improvement
29
Noninvasive Diagnostic Work-up
after shunting.3,4 Other psychometric tests may be even
Grooved Pegboard Test
more selective when detecting deficits in iNPH (e.g., sim-
The grooved pegboard test (Lafayette Instrument Co.,
ple reaction time, the grooved pegboard test, the Stroop
Lafayette, IN, USA) measures eye-to-hand coordination,
test, the digit span test, the trail-making test). For the
motor speed,6 and concentration (▶ Fig. 6.2). The grooved
MMSE, reaction time, grooved pegboard, digit span, Rey
pegboard is a board with 25 holes and differently posi-
auditory verbal learning, and Stroop tests, patients with
tioned slots. The pegs/needles have a ridge on one side
iNPH perform significantly worse compared with healthy
and fit in the holes only when rotated in the correct
individuals, and they show a significant improvement
orientation. The patient should insert the pegs as quickly
after treating iNPH with a shunt.3,4 Indeed, there are also
as possible in a line from left to right using the dominant
many other and potentially more suitable tests; however,
hand. The time taken to complete the task is measured.
the aforementioned tests represent examples of tests that
The test can also be performed with the nondominant
are relatively easy to perform and have the opportunity
hand, but this additional test does not seem to be as
to be widely used in patients with iNPH. Below is a
important in assessing patients with iNPH. (The patient
(small) selection of tests for NPH that can be performed
should wear his or her glasses during the test.) The
by health care professionals; descriptions of these tests
performance is age dependent, and unfortunately there
can be found in ▶ Table 6.1.
are no exact normal values for very old patients (older
than 70 years).
Mini Mental State Examination
Normal values are given by the manufacturer. The
The MMSE is probably the most popular neuro-
time required for completion of this test, including
psychological test, and is described by Folstein et al.5 It
patient instruction and performance (with the domi-
screens cognitive impairment and assesses orientation in
nant hand only), is 5 to 10 minutes. The patient’s per-
time and place, attention, concentration, calculation, lan-
formance time should be documented for follow-up,
guage, short-term memory, and the ability to perform
and performance is significantly improved in patients
easy tasks (▶ Fig. 6.1). The test is also available in a num-
who have had a shunt inserted.4 The simplicity and
ber of different languages.6
quick accomplishment of this test makes it suitable
The test scores range from 0 to 30. Scores in the range
in the routine assessment of patients, as well as in
of 25 to 27 are indicative of mild cognitive impairment,
follow-up after shunting.
while scores of 24 or lower represent a high probability
of cognitive impairment.
Rey Auditory Verbal Learning Test
The MMSE test is performed with little effort and takes
about 10 to 15 minutes only. It can be used for screening
The RAVLT assesses verbal learning and memory. In the
as well as for follow-up of patients with iNPH, even
variant most used,6 15 words are read aloud. The patient
though it is not specially designed for subcortical demen-
must recall the spoken words. The task is repeated in five
tia such as that seen in iNPH. Shunt treatment produces a
identical trials, reading the words again in the same order
significant improvement in performance of the MMSE.4
followed by a free recall test. After the fifth trial (RAVL1-5),
Despite the criticism of its use in iNPH (“alibi for neuro-
15 different words are read aloud, followed by a recall of
psychological testing”), the MMSE holds a firm place in
this second word list. Then the first list must be recalled
iNPH diagnosis and follow-up.
without presenting the first list. After
20 minutes have
passed, the patient must recall the first list again.
The test looks particularly for memory and learning
Table 6.1 Selection of neuropsychological tests applicable for
patients with NPH and their suitability for routine use
disabilities, and it is quite easy to perform. However, it is
time consuming (> 30 min); therefore, it is not suitable for
Test
Time required for
Validity
Feasibility in
routine use.
completion (min)a
for NPH
daily routine
MMSE
10-15
+
++
Grooved peg-
5-10
+
++
Digit Span Test
board test
The digit span test is a subtest of the Wechsler Adult
RAVLT
> 30
+
-
Intelligence Scale III
(WAIS-III) and mostly assesses
Digit span test
5-10
+
±
short-term and working memory.3 A series of digits
TMT
10
±
+
must be repeated forward and backward. Scores corre-
Stroop test
10
+
+
spond to the number of digits in the longest series for
Abbreviations: MMSE, mini mental state examination; NPH,
forward and backward repetitions.3 This test can be
normal pressure hydrocephalus; RAVLT, Rey auditory verbal
combined with other subtests of the WAIS-III, but then
learning test; TMT, trail-making test.
it should be performed by professionals in neuro-
aIncluding time required for patient instruction.
psychological testing.
30
Noninvasive Diagnostic Work-up
Max.
Fig. 6.1 Mini Mental State Examination.
Task
points
A score of 30 indicates normal cogni-
tion, a low score indicates severe
1.
What is the: Year? Season?
cognitive impairment. The performance
5
Date? Day? Month?
of the MMSE test may be influenced by
other diseases, especially by depression.
2.
Where are we: Country? State? Town?
Modified from Strauss et al.6
5
Hospital? Floor?
3.
Name 3 objects (apple, penny, table), taking
1 second to say each, then ask the patient to repeat
the 3 words. Repeat the task until the patient leams
3
all 3, up to 6 trials. The score is based on the first
trial.
4.
Serial sevens: Subtract 7 from 100, then subtract 7
from the result, etc., continuing through five
5
subtractions. Score the number of correct answers.
5.
Ask to recall the three objects named in test item 3.
3
6.
Point at a pencil and a watch. Have the patient
2
name them as you point.
7.
Ask the patient to repeat ̏No ifs, ands, or buts̋—
1
only 1 attempt.
8.
Have the patient follow a 3-stage command:
̏Take the paper in your right hand.
3
Fold the paper in half.
Put the paper on the floor.̋
9.
Have patient read and obey the following:
1
̏Close your eyes.̋
Have patient write a sentence of his/her
10.
1
own choice.
Have patient copy the overlapping pentagon design
11.
1
below.
Sum
30
Design for task 11:
31
Noninvasive Diagnostic Work-up
correct order by drawing a line with a pencil. In Task 2,
he or she must connect numbers and letters in alternat-
ing order. The time taken to complete the task is meas-
ured. The performance time is age dependent; the 70th
percentile for the age group 60 to 80 years is between 26
and 39 seconds for Task 1, and is between 62 and
96 seconds for Task 2.6 This test is easy and quick to
perform; therefore, it is suitable for routine use.
Stroop Test
There are many variations of the Stroop test, and this
makes direct comparisons difficult. In the Swedish
Stroop test, there are two parts: first, the patient must
name the color of
100
rectangles8
(color naming;
▶ Fig. 6.4); in the second part, he or she must read the
Fig. 6.2 Grooved pegboard test. The patient is advised to
name of the printed color of 100 incongruent color
position all 25 pegs as quickly as possible from left to right using
words (interference condition;
▶ Fig.
6.5). The time
the dominant hand only. The time is measured.
needed (in seconds) to complete each task is measured
and the difference between the two tasks is deter-
mined, thus “reflecting the extra time needed to select
Trail-Making Test
the correct response in the interference condition”.4 As
The trail-making test measures attention, speed, and
reference: Healthy individuals need for color naming
mental flexibility (▶ Fig. 6.3).6,7
In Task 1, the patient
67 seconds, for interference task
128 seconds, and
should connect 25 randomly positioned numbers in their
“response selection” 62 seconds.
Fig. 6.3 Trail-making test. Task 1: connect
the numbers with a line from 1 to 2, from 2 to
3, and so on until the end. Do not lift the
pencil from the paper. The time is measured.
Task 2: connect the numbers and letters from
1 to A, from A to 2, from 2 to B, and so on
until the end. Do not lift the pencil from the
paper. The time is measured.
32
Noninvasive Diagnostic Work-up
Fig. 6.4 Stroop test (color naming). The
color of each rectangle has to be named by
the patient. The time for completing the
task is registered.
Fig. 6.5 Stroop test (interference condi-
tion). The color of each word (not the word
itself) must be named by the patient. The
time for completing the task is registered.
The Stroop test is easy to perform and lasts less than
routinely, in addition to a detailed patient medical his-
10 minutes. Therefore, it is a practical test for routine use.
tory and clinical examination. In the author’s experi-
ence, the
360° turn, the timed up-and-go test, the
MMSE, and the grooved pegboard tests have proved
6.3 Summary
their feasibility; of course, other tests may be of simi-
lar value. The neuropsychological tests described
Physicians should have effective and objective tests that
above should serve only as a short overview of some
they can perform routinely without extensive effort to
tests that can be performed by a physician and not
identify and follow up patients who have iNPH. All tests
necessarily by a neuropsychologist.
for gait disturbance and mental disorders are valuable
because of their ability to measure disabilities in patients
with iNPH and for their use during patient follow-up.
References
However, gait reacts more rapidly and distinctly to cere-
[1] Relkin N, Marmarou A, Klinge P, Bergsneider M, Black PM. Diagnosing
brospinal fluid tapping and to shunting; therefore, gait
idiopathic normal-pressure hydrocephalus. Neurosurgery 2005; 57
evaluation plays a more important clinical role, particu-
Suppl: S4-S16, discussion ii-v
larly in the diagnosis and evaluation of shunt function.
[2] Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic func-
However, all symptoms should be assessed to describe a
tional mobility for frail elderly persons. J Am Geriatr Soc 1991; 39:
142-148
complete picture of the patient with iNPH.
[3] Hellström P, Edsbagge M, Archer T, Tisell M, Tullberg M, Wikkelsø C.
To adequately evaluate a patient with iNPH before
The neuropsychology of patients with clinically diagnosed idiopathic
and after treatment, a diagnostically conclusive
normal pressure hydrocephalus. Neurosurgery 2007; 61: 1219-1226,
gait and neuropsychological test should be performed
discussion 1227-1228
33
Noninvasive Diagnostic Work-up
[4] Hellström P, Edsbagge M, Blomsterwall E et al. Neuropsychological
[6] Strauss E, Sherman EMS, Spreen O. A compendium of neuropsychological
effects of shunt treatment in idiopathic normal pressure hydrocepha-
tests. 3rd edition, Oxford University Press, 2006
lus. Neurosurgery 2008; 63: 527-535, discussion 535-536
[7] Partington JE, Leiter RG. Partington’s Pathway test. The Psychological
[5] Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical
Service Center Bulletin 1949; 1: 9-20
method for grading the cognitive state of patients for the clinician.
[8] Hellström P, Scharin M. Stroop, the bewildering effect. [in Swedish]
J Psychiatr Res 1975; 12: 189-198
Svensk Neuropsykologi 2001; 13: 12-15
34
7.1
Computed Tomography
36
Chapter 7
7.2
Subjective Assessment of
Imaging
Ventricular Size and Shape
36
Johannes Lemcke
7.3
Indices
36
7.4
MRI/Phase-Contrast MRI
38
7.5
Diffusion Tensor Imaging
38
7.6
Multifrequency Magnetic
Resonance Elastography
39
7.7
Direct Postoperative Imaging
39
7.8
Imaging for Routine Follow-Up
39
7.9
Native Radiography/Valve
Setting
39
7.10
Using Imaging for
Troubleshooting
41
Imaging
7 Imaging
Johannes Lemcke
In the early days, an invasive method had to be used even
prediction for which patients would become respond-
for the evaluation of ventricle size in idiopathic normal
ers or nonresponders.2,3
pressure hydrocephalus (iNPH). Now, we try to perform
the complete diagnosis without having to resort to inva-
sive methods. Although promising approaches have been
7.3 Indices
found, completely noninvasive diagnostics for iNPH have
The radiologic assessment of patients with iNPH
not become a reality and the same difficulties remain in
requires that different scans in the history of one patient
the follow-up of patients with iNPH, as well as during the
are comparable when carried out by different exam-
treatment of complications.
iners. Consequently, an index must be used to document
The aim of this chapter is to describe the range of alter-
ventricular size.
natives available so as to avoid invasive diagnostics when
The indices used in the assessment of patients with
possible.
iNPH have advantages, but they also have pitfalls. Consist-
ency in the use of the same index by all examiners is
essential.
7.1 Computed Tomography
Native computed tomography (CT) remains the norm for
7.3.1 Evans Index
diagnostic and follow-up examinations of iNPH. Even an
old, one-slice CT scanner can provide all of the informa-
The Evans index was introduced by Detroit (Michigan,
tion needed about the configuration of the ventricular
United States) radiologist William A. Evans in 1942.4
system. A CT scan requires only a few minutes and CT
At that time, the standard diagnostic method was ence-
scanners are universally available.
phalography. The present day definition of the index is
The primary use of a CT scan in the diagnosis of iNPH is
an adaptation: in the cella media slice of a CT scan, the
to detect an enlarged ventricular system, which is the key
widest distance in the frontal horns is divided by the
feature in iNPH. The secondary objective is to exclude the
widest transverse distance between the tabulae inter-
possibility that
nae. The index is widespread, easy to calculate, and does
● Other features could produce the symptoms and not
not require any special data acquisition. There is consen-
the suspected iNPH
sus that an Evans index > 0.3 is indicative of hydrocepha-
● Other features may have produced hydrocephalus
lus. The Evans index directly indicates the width of the
(secondary hydrocephalus)
frontal horns, which, in our opinion, is the most specific
● A contraindication for the surgical treatment of iNPH
change in the shape of the ventricles in patients with
exists.
iNPH. The disadvantages of the index are that values
vary significantly depending on the CT slice within the
same CT scan and the lack of representation of ventricu-
7.2 Subjective Assessment of
lar volume.5 A pitfall of the Evans index is that CT scans
with different gantry angles cannot be compared
Ventricular Size and Shape
because the transverse distance between the tabulae
A variety of assessment methods are widely available.
internae changes according to the position of the slice.
Indeed, any experienced neurosurgeon could claim that
The same problem results from different standards in
he or she can identify a hydrocephalic constellation in a
the angle of transverse CT and magnetic resonance
CT scan without the use of a measuring instrument.
imaging (MRI) scans.
Although this approach is not objective, it does appear
that the results of subjective assessment are not com-
7.3.2 Other Indices
pletely unreliable.1
In our experience, the typical CT scan of a patient
Many indices have been published with regard to differ-
with iNPH shows enlarged, cloddy ventricles. The fron-
ent forms of hydrocephalus. None of these alternative
tal horns are more dilated than the occipital horns.
indices is widespread for the evaluation of patients with
The third ventricle is dilated, but it still retains an oval
iNPH. All indices have advantages and disadvantages.
shape. Massive enlargements of the extracerebral
Most important for communication within a clinic or a
arachnoid space and the sulci are not typical. Some of
department is that all collaborators use the same index
these criteria have been subjected to systematic
and are familiar with that index. For scientific publica-
examination, but they have failed to provide a reliable
tions, use of the Evans index is obligatory.
36
Imaging
ventricle and to the proportions of the third and fourth
7.3.3 Third Ventricle Diameter
ventricles. Because these cases are rare, they must be
To differentiate between iNPH and some forms of func-
examined individually. In our view, an index that includes
tional aqueduct stenosis that are hardly detectable, it
the width of the third ventricle is not needed for patients
is important to pay attention to the width of the third
with iNPH (Table 7.1).
Table 7.1 Indices for measuring ventricular enlargement
Index
Image
Calculation
Advantages
Disadvantages/pitfalls
● Gantry inclination affects
● Widespread
Evans index4
Evans index = a/b
the index
● Easy to calculate
● CT and MRI not comparable
Fronto-occipital horn ratio
● Easy to calculate
● Less relevant for iNPH as
syn. frontal and occipital
FOHR = a + c/2b
● CT and MRI are com-
wide occipital horns are not
horn ratio6-8
parable
pathognomonic
(FOR syn. FOHR)
● Easy to calculate
Frontal horn ratio9
(FHR)
FHR = a/b
● CT and MRI are com-
● Very uncommon for iNPH
syn. bifrontal index (BFI)10
parable
● Easy to calculate
Bicaudate ratio (BCR)11,12
BCR = a/b
● CT and MRI are com-
● Very uncommon for iNPH
parable
● Easy to calculate
Third ventricular sylvian
3VSFR = (a + a′)/b
● CT and MRI are com-
● Very uncommon for iNPH
fissure ratio (3VSFR)9
parable
Third ventricular brain
● CT and MRI are com-
● Very uncommon for iNPH
VBR3=A*A′/B*B′
ratio (VBR3)9
parable
● Complicated calculation
Abbreviations: CT, computed tomography; iNPH, idiopathic normal pressure hydrocephalus; MRI, magnetic resonance imaging; syn.,
synonym.
37
Imaging
is forced out through the aqueduct with every heart-
7.4 MRI/Phase-Contrast MRI
beat. This CSF movement can be measured by functional
MRI has advantages compared with CT in relation to the
MRI (fMRI) either in milliliters per heartbeat or in milli-
following possibilities:
liters per second.6-21 Through clinical studies performed
● The detection of special conditions (e.g., membranes) in
by our own research group,22 we generated a threshold
the region of the aqueduct that could explain a func-
value of 24.5 mL/s to identify patients with iNPH, but
tional stenosis
we also found that this parameter is not appropriate for
● The measurement of aqueduct cerebrospinal fluid (CSF)
all patients. Currently, the measurement of aqueduct
flow
CSF flow is not a reliable method to establish an indica-
tion for shunt surgery (▶ Fig. 7.1).
Generally, a CT scan is sufficient as preoperative imaging
and has the advantage that postoperative CT scans are
comparable in relation to ventricular width.
7.5 Diffusion Tensor Imaging
A preoperative MRI should be performed if the CT scan
shows a mismatch between the size of the lateral ventri-
On the one hand, diffusion tensor imaging (DTI) is a very
cles and the third ventricle or between the third and
interesting area of fMRI that may potentially provide
fourth ventricles. In those cases, it may be of interest to
noninvasive diagnostics for iNPH in the future, and, on
detect structures causing functional stenosis and to open
the other, it may help us to elucidate the pathophysiology
up the possibility of performing endoscopic procedures.
of iNPH. However, at present, the method has not been
As it is not a mistake to shunt a patient with obstructive
established for diagnosing iNPH and cannot be the sole
hydrocephalus, MRI is not obligatory in patients showing
basis for the decision to carry out shunt surgery.
enlarged lateral and third ventricles but a normal fourth
Hattingen et al23 have reported specific microstructure
ventricle. An MRI is, of course, essential if there is a suspi-
changes in periventricular white matter structures.
cion of a “trapped” fourth ventricle that could worsen
Lenfeldt et al24 speculate that the changes in the anterior
with postshunt surgery.
frontal white matter could impair signaling between the
Aqueduct CSF flow seems to have a strong relation-
frontal cortex and the basal ganglia, thereby disturbing
ship with the pathophysiology of the iNPH; therefore,
motor-planning processes. Kim et al25 were able to differ-
several attempts have been made to develop a reliable
entiate between patients with a shunt-responsive iNPH
diagnostic tool based on this relationship. The rationale
and patients with Alzheimer disease using DTI and found
of increased aqueduct CSF flow is that, as a result of
specific changes (higher fractional anisotropy) in the pos-
rigid basal arteries, cerebral blood flow is not trans-
terior limb of the internal capsule that could possibly
formed into a laminar streaming before entering the
explain gait disturbances in patients with iNPH. Micro-
capillaries in patients with iNPH. Accordingly, the peri-
structural alterations of the hippocampus were detected
ventricular parenchyma pulsates with the frequency of
by Hong et al26 and could possibly be associated with
the blood. Balancing this parenchyma pulsation, the CSF
amnestic deficits in patients with iNPH.
Fig. 7.1 (a and b) Measurement of the aqueductal cerebrospinal fluid flow by phase-contrast magnetic resonance imaging.
38
Imaging
company, Raynham, Massachusetts, United States) has
7.6 Multifrequency Magnetic
been implanted, then the CT scout view will not allow
Resonance Elastography
a reliable reading of the valve setting. In such cases, a
native lateral radiography of the skull must be per-
Magnetic resonance elastography is another emerging
formed.
technique with a possible future benefit for diagnosing
Performing MRI directly after surgery as a routine con-
iNPH. It utilizes an MRI head cradle connected to a vibra-
trol examination provides no advantage, apart from the
tion generator that induces mechanical vibrations in the
decrease in radiation exposure, and it involves the risk of
head during MRI and so allows a visualization of the elas-
an unintended valve setting.
ticity of the brain.
The working groups of Sack27,28 were able to show a
significant decrease in viscoelastic properties
(shear
7.8 Imaging for Routine
modulus) in tissue areas near the ventricular system.
These findings are promising, but they are not yet of prac-
Follow-Up
tical use nor are they able to elucidate the patho-
Routine follow-up of patients with iNPH should be
physiology (▶ Fig. 7.2).
managed with CT scans in the first year following shunt
implantation. Proper follow-up periods are, for exam-
7.7 Direct Postoperative
ple, 3, 6, and 12 months after shunt implantation.29,30
Further follow-up should be arranged on an annual
Imaging
basis. For follow-up examinations more than 12 months
after shunt implantations, most authors state that CT
The imaging performed directly following surgery should
scans are not required as long as the patient does not
ensure the following:
show severe changes in clinical symptoms.31 In the era
● Correct placement of the ventricular catheter
of gravitational valves, constancy in the size of the
● Absence of intracerebral bleeding complications
ventricular system or only a minimal decrease in ven-
● Ventricular size after shunt implantation
tricular size does not constitute evidence that therapy
● Correct placement of the abdominal catheter
has failed. Meier et al32,33
were able to show that
● Initial setting of the valve, if a programmable valve was
patients with no or a minimal decrease in ventricular
used
size after shunt surgery with gravitational valves had bet-
● Vertical position of a gravitational unit, if one has been
ter outcomes than those with significantly decreasing
implanted
ventricles.
In the authors’ experience, these features can be
achieved with a native CT scan and native abdominal
two-plane radiography. The intra-abdominal place-
7.9 Native Radiography/Valve
ment of the abdominal catheter can also be verified by
Setting
ultrasonography. The advantage is the decrease in
radiation exposure, but the disadvantage is a doubtful
Programmable valves are widespread in the therapy of
result in many cases.
iNPH. The possibilities of definite control of the valve set-
If a small programmable valve (e.g., CODMAN MEDOS
ting are interesting in relation to many clinical situations
programmable valve, Codman, a Johnson & Johnson
(Table 7.2).
Fig. 7.2 Top row: wave images after Fourier
decomposition (real part). The vibration
frequency is given in the top left-hand
corner. Blue color scales vibrations toward
the reader and red to yellow color scales
motion beneath the image plane. (a)
Healthy volunteer (woman, 72 years of age).
(b) Patient with normal pressure hydro-
cephalus (woman, 70 years of age).27
39
Imaging
Table 7.2 Use of imaging modalities to adjust programmable valves
Valve type
MRI safe
CT scout
Native radiography Fluoroscopy
Adjustment scheme
Aesculap
Stable up to 3 T
MIETHKE proGAV
(new version)
Aesculap
Stable up to 3 T
MIETHKE proGAV
(old version)
Aesculap
Stable up to 3 T
MIETHKE
proSA
CODMAN MEDOS
Resetting after
programmable
MRI
valve
CODMAN CERTAS
Stable up to 3 T
40
Imaging
Table 7.2
continued
Valve type
MRI safe
CT scout
Native radiography Fluoroscopy
Adjustment scheme
Sophysa Polaris
Stable up to 3 T
Medtronic
Resetting after
STRATA
MRI
Abbreviations: CT, computed tomography; MRI, magnetic resonance imaging.
tissue, thus allowing the CSF to bypass the
7.10 Using Imaging for
valve.
Troubleshooting
2. Disintegration of a programmable valve due to heavy
magnetic fields or mechanical forces.
In cases wherein there is a suspicion of shunt failure, a vari-
3. Entrapment of CSF concrement in a ball-in-cone valve
ety of possible causes can be postulated. The experienced
that causes a permanent opening of the valve.
surgeon can often determine the precise failure mechanism
4. Implantation of a gravitational valve at an incorrect
with only a few examinations. The clinical management of
angle to the body axis that causes a permanent
shunt failure detection is described in Chapter 15.
opening of the valve (▶ Fig. 7.3).
Almost every noninfective shunt complication will
result in either overdrainage or underdrainage. Accord-
ingly, comparing a native cranial CT scan with a previous
one will establish whether too much or too little CSF is
transported by the shunt.
A CT scan of a patient with overdrainage shows a
decrease in the size of the ventricular system when com-
pared with a previous CT scan. Such a decrease must be
taken even more seriously if it appears in a patient with a
gravitational valve. Moreover, a subdural hygroma or
hematoma is a sign of overdrainage.
Underdrainage typically manifests as an enlargement of
the ventricular system compared with previous CT scans.
7.10.1 Overdrainage
There are four possibilities of shunt failure that cause
overdrainage that is detectable with radiologic methods:
1. Shunt disconnection proximal to the valve
Fig. 7.3 Implantation of a gravitational valve at incorrect angle
with a shunt surrounding sheath of connective
with respect to body axis.
41
Imaging
Fig. 7.4 Contrast medium shuntogram
showing patency of ventriculoperitoneal
shunt (a) or obstruction of same (b).
[5]
Toma AK, Holl E, Kitchen ND, Watkins LD. Evans’ index revisited:
7.10.2 Underdrainage
the need for an alternative in normal pressure hydrocephalus.
Neurosurgery 2011; 68: 939-944
There are innumerable possibilities of shunt failure caus-
[6]
Bateman GA. Magnetic resonance imaging quantification of compli-
ing underdrainage that can be comprehended with refer-
ance and collateral flow in late-onset idiopathic aqueductal stenosis:
ence to radiology. In principle, all kinds of obstruction,
venous pathophysiology revisited. J Neurosurg 2007; 107: 951-958
dislocation, disconnection, and kinking, among others,
[7]
Bateman GA, Loiselle AM. Can MR measurement of intracranial hydro-
can cause underdrainage complications.
dynamics and compliance differentiate which patient with idiopathic
normal pressure hydrocephalus will improve following shunt inser-
A dislocation of the ventricular catheter can be
tion? Acta Neurochir (Wien) 2007; 149: 455-462, discussion 462
detected easily by a CT scan. The integrity of the shunt
[8]
Bateman GA, Levi CR, Schofield P, Wang Y, Lovett EC. The patho-
system can be established by native radiography of the
physiology of the aqueduct stroke volume in normal pressure hydro-
course of the shunt.
cephalus: can co-morbidity with other forms of dementia be
Even a correctly placed shunt without any dis-
excluded? Neuroradiology 2005; 47: 741-748
[9]
Bateman GA. Pulse-wave encephalopathy: a comparative study of the
connection does not ensure that a shunt is functional;
hydrodynamics of leukoaraiosis and normal-pressure hydrocephalus.
therefore, invasive functional imaging may be indicated.
Neuroradiology 2002; 44: 740-748
There are two possibilities for performing a shunto-
[10]
Bateman GA, Brown KM. The measurement of CSF flow through the
gram: using radionuclides34
or a contrast medium.
aqueduct in normal and hydrocephalic children: from where does it
come, to where does it go? Childs Nerv Syst 2012; 28: 55-63
Although we personally have never seen a shunt infection
[11]
Bateman GA, Stevens SA, Stimpson J. A mathematical model of idio-
in a patient with iNPH caused by an invasive shuntogram,
pathic intracranial hypertension incorporating increased arterial
the danger is obvious. Therefore, shuntograms should be
inflow and variable venous outflow collapsibility. J Neurosurg 2009;
performed only under sterile conditions. When the ante-
110: 446-456
chamber or the reservoir of the valve is punctured with a
[12]
Bradley WG. MR prediction of shunt response in NPH: CSF morphol-
ogy versus physiology. AJNR Am J Neuroradiol 1998; 19: 1285-1286
small cannula, a contrast medium must be injected. The
[13]
Edelman RR, Wedeen VJ, Davis KR et al. Multiphasic MR imaging: a
contrast medium should reach the ventricle on the proxi-
new method for direct imaging of pulsatile CSF flow. Radiology 1986;
mal side and the intra-abdominal space on the distal side.
161: 779-783
One should take into account the fact that proof of
[14]
Mascalchi M, Arnetoli G, Inzitari D et al. Cine-MR imaging of aque-
“patency” in the shuntogram still leaves the possibility
ductal CSF flow in normal pressure hydrocephalus syndrome before
and after CSF shunt. Acta Radiol 1993; 34: 586-592
open for nonphysiologic resistance to CSF flow in the
[15]
Mase M, Yamada K, Banno T, Miyachi T, Ohara S, Matsumoto T. Quan-
shunt (e.g., due to an inadequate valve setting or partial
titative analysis of CSF flow dynamics using MRI in normal pressure
obstruction; ▶ Fig. 7.4).
hydrocephalus. Acta Neurochir Suppl (Wien) 1998; 71: 350-353
[16]
Miyati T, Mase M, Banno T et al. Frequency analyses of CSF flow on
cine MRI in normal pressure hydrocephalus. Eur Radiol 2003; 13:
1019-1024
References
[17]
Poca MA, Sahuquillo J, Busto M et al. Agreement between CSF flow
[1] Mann SA, Wilkinson JS, Fourney DR, Stoneham GW. Comparison of
dynamics in MRI and ICP monitoring in the diagnosis of normal pres-
computed tomography 3-dimensional volumetric analysis of ventric-
sure hydrocephalus. Sensitivity and specificity of CSF dynamics to
ular size to visual radiological assessment. J Comput Assist Tomogr
predict outcome. Acta Neurochir Suppl (Wien) 2002; 81: 7-10
2009; 33: 789-794
[18]
Schroth G, Klose U. MRI of CSF flow in normal pressure hydrocepha-
[2] Wikkelsö C, Andersson H, Blomstrand C, Matousek M, Svendsen P.
lus. Psychiatry Res 1989; 29: 289-290
Computed tomography of the brain in the diagnosis of and prognosis
[19]
Scollato A, Tenenbaum R, Bahl G, Celerini M, Salani B, Di Lorenzo N.
in normal pressure hydrocephalus. Neuroradiology 1989; 31: 160-165
Changes in aqueductal CSF stroke volume and progression of symp-
[3] Chatzidakis EM, Barlas G, Condilis N et al. Brain CT scan indexes in
toms in patients with unshunted idiopathic normal pressure hydro-
the normal pressure hydrocephalus: predictive value in the outcome
cephalus. AJNR Am J Neuroradiol 2008; 29: 192-197
of patients and correlation to the clinical symptoms. Ann Ital Chir
[20]
Sharma AK, Gaikwad S, Gupta V, Garg A, Mishra NK. Measurement of
2008; 79: 353-362
peak CSF flow velocity at cerebral aqueduct, before and after lumbar
[4] Evans WA. An encephalographic ratio for estimating ventricular
CSF drainage, by use of phase-contrast MRI: utility in the manage-
enlargement and cerebral atrophy. Arch Neurol Psychiatry 1942; 47:
ment of idiopathic normal pressure hydrocephalus. Clin Neurol
931-937
Neurosurg 2008; 110: 363-368
42
Imaging
[21] Yoshihara M, Tsunoda A, Sato K, Kanayama S, Calderon A. Differential
[28] Freimann FB, Streitberger KJ, Klatt D et al. Alteration of brain viscoe-
diagnosis of NPH and brain atrophy assessed by measurement of
lasticity after shunt treatment in normal pressure hydrocephalus.
intracranial and ventricular CSF volume with 3D FASE MRI. Acta
Neuroradiology 2012; 54: 189-196
Neurochir Suppl (Wien) 1998; 71: 371-374
[29] Lemcke J, Meier U. Improved outcome in shunted iNPH with a combi-
[22] Al-Zain FT, Rademacher G, Meier U, Mutze S, Lemcke J. The role of
nation of a Codman Hakim programmable valve and an Aesculap-
cerebrospinal fluid flow study using phase contrast MR imaging
Miethke ShuntAssistant. Cent Eur Neurosurg 2010; 71: 113-116
in diagnosing idiopathic normal pressure hydrocephalus. Acta
[30] Meier U, Lemcke J, Al-Zain F. Course of disease in patients with idio-
Neurochir Suppl (Wien) 2008; 102: 119-123
pathic normal pressure hydrocephalus (iNPH): a follow-up study 3, 4
[23] Hattingen E, Jurcoane A, Melber J et al. Diffusion tensor imaging in
and 5 years following shunt implantation. Acta Neurochir Suppl
patients with adult chronic idiopathic hydrocephalus. Neurosurgery
(Wien) 2008; 102: 125-127
2010; 66: 917-924
[31] Klinge P, Marmarou A, Bergsneider M, Relkin N, Black PM. Outcome
[24] Lenfeldt N, Larsson A, Nyberg L, Birgander R, Eklund A, Malm J. Diffu-
of shunting in idiopathic normal-pressure hydrocephalus and the
sion tensor imaging reveals supplementary lesions to frontal white
value of outcome assessment in shunted patients. Neurosurgery
matter in idiopathic normal pressure hydrocephalus. Neurosurgery
2005; 57 (Suppl): S40-S52, discussion ii-v
2011; 68: 1586-1593, discussion 1593
[32] Meier U, Mutze S. Correlation between decreased ventricular size and
[25] Kim MJ, Seo SW, Lee KM et al. Differential diagnosis of idiopathic
positive clinical outcome following shunt placement in patients with
normal pressure hydrocephalus from other dementias using diffusion
normal-pressure hydrocephalus. J Neurosurg 2004; 100: 1036-1040
tensor imaging. AJNR Am J Neuroradiol 2011; 32: 1496-1503
[33] Meier U, Paris S, Gräwe A, Stockheim D, Hajdukova A, Mutze S. Is
[26] Hong YJ, Yoon B, Shim YS et al. Differences in microstructural altera-
there a correlation between operative results and change in ventricu-
tions of the hippocampus in Alzheimer disease and idiopathic normal
lar volume after shunt placement? A study of 60 cases of idiopathic
pressure hydrocephalus: a diffusion tensor imaging study. AJNR Am J
normal-pressure hydrocephalus. Neuroradiology 2003; 45: 377-380
Neuroradiol 2010; 31: 1867-1872
[34] Kharkar S, Shuck J, Kapoor S, Batra S, Williams MA, Rigamonti D.
[27] Streitberger KJ, Wiener E, Hoffmann J et al. In vivo viscoelastic prop-
Radionuclide shunt patency study for evaluation of suspected ventri-
erties of the brain in normal pressure hydrocephalus. NMR Biomed
culoperitoneal shunt malfunction in adults with normal pressure
2011; 24: 385-392
hydrocephalus. Neurosurgery 2009; 64: 909-916, discussion 916-918
43
8.1
Intracranial Pressure Monitoring
46
Chapter 8
8.2
Lumbar Infusion Test
46
Invasive Diagnostic Work-up
8.3
CSF Tap Test
51
Johannes Lemcke, Ullrich Meier
8.4
External Lumbar Drainage
52
8.5
CSF and Serum Biochemical
Tests
52
8.6
Diagnostic Pathway
53
8
Invasive Diagnostic Work-up
8 Invasive Diagnostic Work-up
Johannes Lemcke, Ullrich Meier
The first steps carried out in the investigation of an indi-
referred to as B-waves. Gaab et al13 distinguished B-waves
vidual are the taking of a comprehensive history and a
from the sinusoidal type, which are physiologic and
physical-neurologic examination. To establish a diagnosis
appear during sleep, as well as B-waves from the ramp
of hydrocephalus, by definition there must be ventricle
type, which are considered to be pathologic. Increased
expansion. It is not possible to diagnose normal pressure
B-wave activity during continuous ICP measurement is
hydrocephalus (NPH) using computed tomography (CT)
judged by some authors to be a pathognomonic sign of
or nuclear magnetic resonance imaging (MRI) alone.1-3
NPH and should correlate with a good result following
According to Børgesen et al,4
as well as Tans and
shunt implantation. Kosteljanetz14 reported false-positive
Poortvliet,5 the size of the ventricle does not correlate
results during continuous ICP measurement. In 66% of his
with the pressure-volume index, flow resistance, and
patients he found B-waves, and in 31% he found an
resting pressure. For this reason, selection of patients for
increase in resistance in the subsequent infusion test.
shunt implantation based solely on the findings of
A total of 28% of patients had both B-waves and patholog-
CT should be rejected, nor can a CT cisternography6
ically increased flow resistance, and 38% of patients with
guarantee a diagnosis of NPH. Børgesen et al7 reported
B-waves had no increased flow resistance.
the proportion of false-positive results of CT cisternography
Brean and Eide15,16 reported increased ICP pulse ampli-
to be more than 60%.
tude during sleep at night in 90% of the patients with
The measurement of cerebrospinal fluid (CSF) flow
NPH. In addition, during the lumbar infusion test, there
speed in the cerebral aqueduct has been clinically
was an increase in ICP pulse amplitudes in patients with
evaluated using functional MRI (fMRI).8 Using the two-
NPH. Both authors15,16
postulated that increased ICP
dimensional phase-contrast technique, it was possible
pulse amplitudes
(quantification of pulsatility: Qpulse)
to demonstrate that a CSF flow rate of more than 24.5
have a higher predictive value for a shunt operation than
mL/min was correlated with NPH with a specificity of
the flow resistance
(Rout) measured in the lumbar
95%. However, this noninvasive method of investigation
infusion test. Pfisterer et al17 favored continuous intra-
had the disadvantage of having a low sensitivity (i.e.,
ventricular ICP measurement with the detection of
46%), so too many false-negative results were obtained.
B-waves (more than 50%). In the event of suspect findings
Other noninvasive diagnostic methods, such as single-
(B-waves:
10-50%), the same authors recommended
photon emission tomography (SPECT), positron emission
using the lumbar cerebrospinal tap test. A noninvasive
tomography (PET), and xenon CT9 can be helpful in
measurement of ICP using otoacoustic emissions was
the assessment of the course of the disease, but not in
favored by Frank et al18; however, use of this method has
diagnostic investigation.
not prevailed in clinical practice.
In the guidelines for the management of idiopathic
NPH (iNPH) of the Japanese Neurosurgery Society19 and
8.1 Intracranial Pressure
the guidelines of the U.S. iNPH Study Group,20 ICP moni-
toring and B-wave analysis take a secondary position in
Monitoring
the diagnostic chain of iNPH and are described with class
III evidence.
In the first description of the clinical picture of NPH,10 a
lumbar CSF pressure of
180
to
200 mm H2O was
described. However, the single or multiple measurement
8.2 Lumbar Infusion Test
of CSF pressure by means of a lumbar puncture has only
historical significance, because it varies greatly through-
In the infusion test, the ICP is measured during an
out the course of the day, not only interindividually but
infusion of artificial CSF. The flow is applied against the
also individually. Therefore, a short-term intracranial
ICP. The slope of the regression line is an expression of
pressure (ICP) measurement is not effective as a diagnos-
conductance, and the reciprocal value is resistance.21,22
tic method.
The principle of the constant-rate infusion test is the
Various flow diagrams used for the diagnostic investi-
infusion of artificial CSF in the lumbar subarachnoid
gation of NPH11,12 include the analysis and assessment of
space or in a lateral ventricle. The volume is injected with
the ICP signal following long-term measurement over at
the aid of an infusion pump at a constant rate between
least 24 to 48 hours, and also 72 hours. There are ICP
0.76 and 2 mL/min. The increase in pressure that is pro-
waves in addition to the pulse and breathing waves: the
voked is continuously registered and, after achieving
A-waves, the B-waves, and the C-waves. Oscillations of
the new steady state, the infusion is discontinued. In the
the ICP with a frequency of 0.5 to 3/min and amplitudes
condition of the newly set plateau, one can assume that
of less than a few mm Hg up to more than 50 mm Hg are
the reabsorption rate is equal to the infusion rate. In the
46
Invasive Diagnostic Work-up
procedure of Katzman and Hussey,23 one needs between
Table 8.1 Normal values for Rout (Rout)
90 and 120 minutes for the investigation, because it takes
Study
Normal value of R
out
about 40 to 60 minutes to achieve the plateau. In contrast
(mm Hg/mL/min)
to the conventional infusion test, the dynamic infusion
Boon et al42
< 18
test of Meier et al24,25 does not require a balanced state to
Børgesen and Gjerris45
< 12
calculate the parameter described; using regression anal-
Ekstedt40
< 6.6
ysis, it is calculated from a comparison of the increasing
Fuhrmeister28
< 3.8
and decreasing sides of the pressure curve.
The literature about positive predictors in the infusion
Kahlon et al27
< 14
test was reviewed and it was concluded that a high resist-
Meier et al43,44
< 13
ance (Rout > 12.5 mm Hg/mL/min), a sensitivity of 98.5%,
Shapiro et al41
< 2.8
and a specificity of 62% is achieved and should be consid-
Sklar74
< 10
ered to have a value of 86%, in total, as a predictor. The
Tans and Poortvliet47
< 13
sensitivity of CSF dynamics studies has been reported
with values between 56% and 100%, as well as specificity
between 50% and 90% in various publications with more
influence of pressure in the sinus sagittalis superior.
than 30 patients.26 Kahlon et al27 report that the infusion
Mostly, it has been assumed that sinus pressure is con-
test has an 80% positive predictive value (PPV), whereby
stant when there is an increase in ICP.32-36 In animal
false negatives of up to 16% are possible.
experiments, Love et al37 observed an increase in pres-
sure in the sinus sagittalis superior with increasing ICP.
8.2.1 Outflow Resistance
This increase in pressure can, in their opinion, be traced
back to a collapse of the veins. For this reason, the values
Outflow resistance Rout (Torr/mL/min) is a measure of
measured for resistance are too great because in the cal-
pressure decrease following a bolus injection. Outflow
culation, the resistance of the veins must have an influ-
resistance is calculated according to the Hagen-Poiseuille
ence. In the method of calculation of Meier et al,38,39 the
law. CSF flow Q (mL/min) is proportional to the pressure
sinus pressure is indirectly expressed in the nonlinear
gradient (delta) p between the subarachnoid space (po)
course of outflow resistance R(p). The normal values for
and the sinus sagittalis superior (pss) (dp = po - pss) and is
outflow resistance of Ekstedt,40 Fuhrmeister,28 as well as
inversely proportional to the frictional resistance R,
Shapiro et al,41 dating from 1978, 1985, and 1980, respec-
which arises at the arachnoidal villi and partly in the sub-
tively, are too low. These must be revised on the basis of
arachnoid space as well as other bottlenecks in the CSF28:
contemporary literature, so that the standard of assess-
ment of Boon et al,42 Kahlon et al,27 as well as Meier et
p
Q¼
ðmm Hg=mL=minÞ
ð1Þ
al,43,44
which correlate with the investigation results of
RðmL=minÞ;orR¼Q
Børgesen and Gjerris,45,46 as well as Tans and Poortvliet,47
Together with the measurement of ICP, the determi-
are confirmed. At the same time, it should be noted that
nation of flow resistance is the most important param-
the high normal values for outflow resistance Rout, as
eter in the analysis of the pathologic processes of CSF
described by Boon et al,42 do indeed have a high PPV, but
dynamics. By calculating outflow resistance, the quanti-
they also have a high negative predictive value. In this
tative extent of a disruption of reabsorption or disrup-
way, too many patients with iNPH in the initial stage will
tion of the passage of CSF can be determined. ICP
be excluded.
increases with increasing outflow resistance and this
The measurement of outflow resistance can be carried
correlation is nonlinear, according to the investigations
out using the isotope dilution method,48 the constant-
of Børgesen et al.29,30 The correlation expresses a reduc-
rate infusion test,23,49 the perfusion method,50,51 or the
tion in the rate of CSF production with increasing ICP.
bolus injection method.22,52 The bolus injection method
Tychmanowicz et al31 observed a nonlinear change in
of Marmarou et al53 is a technically simple procedure, but
resistance during an increase in ICP in animal experi-
the mathematical equation is complicated and the
ments. With low rates of infusion, resistance showed
threshold values given in the literature are too imprecise
its greatest increase, and, with higher rates of infusion
for a safe assessment of CSF dynamics.13
The bolus-
and an increase in plateau pressure, lower outflow
shaped intrathecal injection of a small volume results in a
resistance was observed (▶ Table 8.1).
short-term increase in ICP. After that, there is an expo-
Because of the invasive method of investigation used,
nential decline in pressure. The peak pressure must
the normal values for outflow resistance have been
exceed the resting pressure and the injection amount
obtained almost exclusively in the context of diagnostic
must be greater than the rate of CSF formation.49 Using
clarification in patients who have suspected disturbances
this method, there are numerous sources of error, as lin-
of CSF dynamics. A factor that has received little attention
ear conditions are assumed, in other words, constant val-
up to now in the determination of resistance has been the
ues for resistance and compliance.13,49,54
Comparisons
47
Invasive Diagnostic Work-up
between the bolus injection method and the infusion
into account the independence of pressure from the
methods have resulted in the realization that only a small
mathematical precepts of the two system components
correlation exists between the outflow resistance values
C(p) and R(p) results in a significantly more flexible model
obtained.4,36,55-58
The values measured with the bolus
concept. Our own conceptual model incorporates the
injection method were generally below those achieved
known model of Marmarou et al22,52 as a special case of
with the infusion methods. For this reason, the infusion
craniospinal pressure dynamics. At the same time, the
tests were used, in which either the rate of infusion
function of capacity C(p) adopts any desired constant
(constant-flow technique) or the pressure in the cranio-
nonlinear course as a function of pressure. It is not lim-
spinal system (constant-pressure technique) were kept
ited—as in the model known up to now—by the prede-
constant.23,28,59-63 The outflow resistance was calculated
fined function C = 1/(k × p). Consequently, difficulties in
from the quotients between the pressure achieved by the
interpretation dependent on the method, as in the bolus
infusion, the constant remaining pressure (steady state
test, can be avoided. The second change is produced by a
pressure pss) minus the starting pressure to the infusion
reduction in the number of unknown parameters in the
rate (IInf).13
model. Sinus pressure is not neglected in this model, but
So-called “gold standard” outflow resistance method of
is a part of the function R(p)54 (▶ Fig. 8.1).
calculation is
CSF dynamics are essentially determined by the influence
of compliance C and the outflow resistance Rout.53,59,60,68
At the same time, an increase in pressure can have an influ-
Rout ¼PSS Po
ð2Þ
ence on these two parameters. One cause of this is the elas-
I Inf
ticity of biologic tissues.66,69 Therefore, compliance is greatly
This method of calculation, which is also considered to
dependent on pressure.70
In pathologic cases, outflow
be noncontentious at the international level, is used as
resistance also shows characteristic changes as a function
the so-called “gold standard.” Schmidt et al64 examined
of pressure p.54 The basis for determining the functions C(p)
the reliability and reproducibility of the ventricular or the
and R(p) is the well-known infusion test used in constant-
lumbar infusion tests and the lumboventricular perfusion
flow technology.14 In our method of calculation, similar to
test. According to their results, there is a good correlation
the methods of calculation referred to above, the total pres-
between the two test procedures. The authors empha-
sure course p(t) observed over time t is processed during
sized the validity of the measurement of outflow resist-
and after the infusion. All formulae for calculating C(p)
ance in the diagnostic process for disorders of CSF
and R(p) are derived from a comparison of the increase in
dynamics.
pressure An(p) (positive slope) during the infusion and the
decrease Ab(p) (negative slope) after the infusion at the
same pressure level (▶ Fig. 8.2).
8.2.2 Calculation Procedure
The intrathecal applied infusion rate iInf(t) can be
In 1970, Katzman and Hussey23 described the basis for
divided into two separate currents, ir(t) and ic(t). It can in
the calculation of outflow resistance after carrying out
one part, ir(t), flow out or be reabsorbed and, in the other,
an intrathecal infusion test. In
1972, Marmarou and
ic(t), be stored in the spaces into which the CSF flows. The
Shulman65 extended this method of investigation by
same applies to CSF production Io, which is, at the same
introducing the bolus test for the determination of
time, assumed to be constant.
pressure-volume characteristics and compliance. Of all
the methods of investigating CSF dynamics, the infusion
iInf ðtÞ þ I0 ¼ icðtÞ þ irðtÞ
ð3Þ
test possesses the best prerequisites for further develop-
ment. The signal used in the infusion test corresponds to
the step function used in the technical field for system
The rate of storage ic(t) or the change in volume in
analysis. The fact that with the aid of the infusion test
the store per unit time, dv/dt, is determined by the
only a value for resistance R is determined is, however,
not satisfactory. The pressure course per unit time p(t)
provoked in the infusion test clearly shows the influence
of the craniospinal capacity C(p), as a result of the delayed
pressure response, known from the bolus test.
Starting from the model concept of CSF dynamics pro-
posed by Marmarou et al,52,66 comprising constant resist-
ance and a restricted pressure-dependent capacity, a new
method of calculation for the infusion test was derived
from a more precise interpretation of the modified
model. The model of CSF dynamics according to Meier et
al43,44,67 is characterized by two decisive changes in rela-
Fig. 8.1 Model of cerebrospinal fluid dynamics.43,44,67
tion to the models known up to that time. Thus, taking
48
Invasive Diagnostic Work-up
same time, this also applies to the proof of the inde-
pendence of both differential equations from one
another. Eq. (7) - Eq. (8)
I Inf
¼ CðpÞ½AnðpÞ þ AbðpÞ
T1 ¼ T2
ð9Þ
In accordance with the formulae for the equivalence of
equations, the two equations are equivalent only if the
left and the right terms of the second equation are
expanded in relation to the first additive by an identical
term T on both sides. Because T1 is not equal to T2, it follows
that the two differential equations are independent. The
terms of the functions we are looking for, R(p) and C(p), can
be deduced from the following transformation:
I Inf
Fig. 8.2
Investigation method for the infusion test.
C ðpÞ ¼
ð10Þ
AnðpÞ þ AbðpÞ
Ab = negative slope; An = positive slope.43,44,66
To determine the outflow resistance R(p), Eq. (10) is
compliance C(p) of the store and the temporal change in
used in Eq. (8).
pressure dp/dt.
-AbðpÞIInf
I0 ¼
ð11Þ
AnðpÞ þ AbðpÞ
RðpÞ
dv
dp
dv
icðtÞ ¼
¼ CðpÞ
C ðpÞ ¼
ð4Þ
dt
dt
dp
p
The rate of outflow ir(t) depends on the respective
RðpÞ ¼
ð12Þ
AbðpÞ
pressure p(t) and outflow resistance R(p).
I0 þ IInf
AnðpÞþAbðpÞ
pðtÞ
p
In this way, a nonlinear, first-order differential equation
irðtÞ ¼
irðpÞ ¼
ð5Þ
RðpÞ
RðpÞ
is appropriate for describing CSF dynamics as a model.
This contains two functions: outflow resistance R(p) and
By substituting these values into Eq. (3), one obtains a
compliance C(p). In contrast to the models of Marmarou
mathematical model for the CSF dynamics as a nonlinear,
et al,52,66 Shulman et al,71 and Charlton et al,68 intradural
first-order differential equation.
sinus pressure does not appear in the model equation pre-
sented here [Eq. (6)]. However, its influence is indirectly
iInf ðtÞ þ I0 ¼ CðpÞdp
ð6Þ
expressed in the nonlinear course of the outflow resist-
dt
RðpÞ
ance R(p). As a result of the model concept selected, the
outflow resistance is understood as being the sum of all
Here we sought for two parameter functions C(p) and R
successive CSF passage and reabsorption resistances. Like-
(p). For this purpose, we need to find two independent
differential equations based on the model formulation
wise, compliance should be viewed as being the ability to
expand all CSF spaces that can be reached from the inves-
[Eq.
(6)]. For the infusion test, this can be achieved
tigation site.
readily. At the same time, the first differential equation
The first-order differential equation can only describe
[Eq. (7)] describes the positive slope in the positive curve
[positive slope (p) = dp/dt] during the infusion with a con-
an increasing pressure course p(t) during a constant flow
infusion. In contrast, on discontinuation of an infusion, a
stant rate of infusion iInf (t) = IInf. The second differential
declining pressure course should be verified. There is a
equation [Eq. (8)] is characterized by the declining pres-
simple method for smoothing the time function p(t). The
sure course [negative slope (p) = -dp/dt] following the
time series of the discontinuously recorded pressure
infusion with iInf (t) = 0.
course p(t) is sorted according to the pressure value dur-
p
ð7Þ
ing the infusion in the ascending and, after the infusion,
I Inf þ I 0 ¼ C ðpÞ AnðpÞ þ
RðpÞ
descending sequence. In this way, variations in pressure
p
that can be caused by movements of the patient are ade-
I0 ¼ CðpÞ AbðpÞ þ
ð8Þ
RðpÞ
quately smoothed.54
Using the assumption that the outflow resistance (p) in
After differentiation of Eqs. (7) and (8), one obtains
the vicinity of p0
does not differ significantly from
the basis for the calculation of compliance C(p). At the
R(p0) = p0/I0, it is possible to calculate the rate of production
49
Invasive Diagnostic Work-up
of CSF (I0). In Eq. (12), R(p) is used for the ratio p0/I0. By
compliance at the site of the measurement if there is
transforming to I0, one obtains an equation for calculating
coupling occlusion.13,38,44 The ICP values are on average
CSF production. Naturally, this only applies to the pressure
3 Torr higher than lumbar pressures at ventricular mea-
p near the resting pressure p0.
surement sites. Otherwise, the investigation results from
the infusion test correlate in the case of communicating
I Inf AbðpÞ
cerebrospinal spaces independently of the infusion or
I0 ¼
ð13Þ
ðp
1ÞðAnðpÞ-AbðpÞÞ
measurement site. For clinical practice, the following
p0
conclusions are postulated11,25,72:
Because of the aid assumption, discussed above, this
● In the case of definite communicating CSF spaces in the
relationship should be used only when close to the rest-
imaging procedures of neuroradiology, the lumbar
ing pressure. Another problem is the high sensitivity of
infusion test is a patient friendly and valid method of
Eq. (7) close to the resting pressure p0. Therefore, it is
investigation.
appropriate to determine the rate of production of CSF
● If there is suspected obstructive hydrocephalus, or com-
before the actual infusion using an additional test. As a
munication between all cerebrospinal spaces cannot be
result of a defined volume intake -ΔV by the patient, the
verified, the indication for the ventricular infusion test
pressure -Δp is reduced below the resting pressure p0.
should be made.
Because hardly any reabsorption takes place below the
resting pressure, one can disregard the reabsorption rate
Tans and Poortvliet5
have reported a difference of
IR in relation to the store rate Ic and set I0 = Ic.53
4 mm Hg between lumbar and ventricular ICP values and
differences of mm Hg/min/mL in the case of outflow
resistances. Czosnyka et al73
judged the computer-
I0 ¼ΔVdp
ð14Þ
Δ p dt
supported lumbar infusion test to be a minimally
invasive, fast, and reliable method of investigation.
From the relationship of the volume intake -ΔV to the
The spinal infusion test developed by Fuhrmeister28
fast reduction in pressure -Δp and the subsequent tem-
followed—with a high infusion rate of 5 mL/min—and it is
porally slow increase again to dp/dt, the CSF production
essentially half-way between the bolus test and the con-
can now be determined. After determining CSF produc-
stant-rate infusion test. With this high infusion rate, there
tion I0 in Eq. (14) and logic control with the calculated
is no plateau pressure. The increase in pressure during
value in Eq. (7), the function outflow resistance R(p) can
the infusion and the fall in pressure to the resting pres-
be calculated using Eq. (12). When in doubt, for CSF
sure after ending the infusion are continually registered.
production, Eq. (14) can be used for calculating outflow
The data of the ICP time curves are evaluated at the end
resistance (▶ Fig. 8.3).
of the investigation using a computer. The adverse events
of the constant-rate infusion test are considered to be rel-
8.2.3 Dynamic Infusion Test Within
atively minor. The most frequently reported adverse
events are restlessness, agitation, sweating, headache,
the Investigation Chain
backache, transient paresthesia, pain in the legs and
Free communication between all CSF spaces is considered
seat region, muscle cramps, fecal and urinary urge
to be a prerequisite for lumbar pressure measurements
incontinence, tachycardia, and hypertension.14,50,74 These
since it is only possible to make a measurement of system
complaints occur almost exclusively in conjunction with
Fig. 8.3 Lumbar infusion test.
50
Invasive Diagnostic Work-up
Lumbar Infusion Test
Fig. 8.4 Intrathecal infusion test.
30 mm Hg
20 mm Hg
10 mm Hg
Opening
Infusion 120 mL/h
Recovery
pressure
5 min
10-10 min
10-15 min
increased ICP of more than
50 mm Hg induced by
investigation technique alone, but instead requires fur-
infusion. Following interruption of the infusion or the
ther diagnostic methods to be carried out.
administration of a volume, the symptoms quickly
In the guidelines for the management of iNPH of the
decline. The experience of Meier et al25,75,76 shows that
Japanese Neurosurgery Society19 and the guidelines of
limiting the investigation-dependent ICP increase to
the U.S. iNPH Study Group,20 the PPV of cerebrospinal
40 mm Hg, as well as a slower ICP increase with infusion
outflow resistance
(infusion test) lies somewhere
rates of 2 mL/min in adults, also has mild adverse events
between 75% and 92%. At the same time, the sensitivity of
as a consequence. Accordingly,
11% of patients com-
the infusion test is stated to be 58% to 100%, and the spec-
plained of postpuncture headache symptoms, and 1% of
ificity 44% to 92%.
patients had a refectory meningism without inflamma-
tory signs in the CSF (▶ Fig. 8.4).
8.3 CSF Tap Test
In the constant-pressure infusion test, the pressure
level to be achieved is predefined and the infusion rate
The CSF tap test (also called the cerebrospinal tap test
required to achieve this is set via an electronic control cir-
or simply tap test) is based on the principle that should
cuit. In doing so, the difference between CSF reabsorption
be achieved with the implantation of a CSF diverting
and production can be determined using a spectrum of
shunt. As a result of the intake of 40 to 60 mL CSF,
different pressure values. This method is considered to be
there is a reduction in the outflow resistance and an
complicated. The infusion pressure is electronically con-
improvement in the regional cerebral blood flow. Malm
trolled and generated by an air pump that pumps air into
et al80 and Wikkelsö et al81 reported both false-positive
the infusion bottle. The outflow resistance is calculated in
and false-negative results to be 5% to 10% in the cere-
the same way as in the constant-rate infusion test. In
brospinal tap test. In addition, Ishikawa et al82
and
addition, the calculation of the pressure in the sinus sag-
Sand et al83
reported false-negative investigation
ittalis superior and the pressure-volume relationship are
results following diagnostic CSF intake. As a cause for
achieved. The expenditure in terms of time for this inves-
this, the duration of the disease and the extent of the
tigation is approximately 3 hours, which allows adequate
brain breakdown process resulting from NPH need to
measurement results to be obtained for CSF reabsorption
be discussed. According to Kahlon et al,27 the PPV of
in the various pressure ranges. The criticism of some
the cerebrospinal tap test, with a value of
94%, is
research groups40,74,77 is the reason why the quantitative
indeed higher than that obtained with the infusion test.
evaluation of the CSF dynamics is a function of pressure.
However, with the cerebrospinal tap test alone, 58% of
For this reason, the constant-rate infusion method is not
patients with positive postoperative outcomes were not
suitable. The adverse events of the method can be com-
diagnosed. Therefore, the false-negative findings with
pared with those of the constant-rate infusion test.
the cerebrospinal tap test amount to 58%. For these rea-
Having carried out 2,256 CSF dynamics investigations
sons, establishing the indication for a shunt operation
over 17 years, Ekstedt and Fridén78 concluded that the
is suggested when either the infusion test or the cere-
outflow resistance can be determined with the greatest
brospinal tap test is positive.27 In contrast to the delib-
accuracy using the infusion method. Sprung et al79 under-
erations of Ravdin et al,84 we recommend the clinical
line the value of this method, but they state that the diag-
control of gait disturbances according to a fixed proce-
nosis of NPH is not achieved from the result of a special
dure, not only 2 to 4 hours after the tap test, but also
51
Invasive Diagnostic Work-up
We recommend a CSF effusion rate of 50 mL in 8 hours, and
this diagnostic method should only be used in patients after
negative findings have been obtained through the cerebro-
spinal tap test. In patients who have had a longer period
of anamnesis, external lumbar drainage results in fewer
false-negative findings than the cerebrospinal tap test.
According to Sharma et al,87
the positive effect of
lumbar drainage can be well demonstrated with phase-
contrast MRI with regard to the CSF change in the
aqueduct. At the same time, the hyperdynamic amplitude
of the CSF flow changes to physiologic amplitudes in the
majority of patients with iNPH. As is also the case with
Fig. 8.5 Spinal tap test.
the cerebrospinal tap test, free communication of the CSF
routes (no spinal stenoses) is a methodologic condition
with lumbar drainage.88 Governale et al46 reported a
after 24 and 48 hours, so that false-negative findings
complication rate of 3% with external lumbar CSF drain-
are not precipitated in patients who have had a longer
age over 3 to 5 days. A total of 1.7% of patients showed
history of iNPH.
symptomatic subdural or subarachnoid bleeding, 0.8%
In the guidelines for the management of iNPH from the
had meningitis, and 0.4% had dislocated catheters.46
Japanese Neurosurgery Society19 and the guidelines of the
In the guidelines for the management of iNPH of the
U.S. iNPH Study Group,20 the PPV of the tap test is described
Japanese Neurosurgery Society19 and the guidelines of
as having a value of 73% to 100%, as well as class II evidence.
the U.S. iNPH Study Group,20 the PPV of external lumbar
At the same time, the sensitivity of the cerebrospinal tap
drainage is reported as
80% to 100%, with class III
test amounts to 26% to 62%, with a specificity of 33% to
evidence. At the same time, the sensitivity of external
100%. Because of the low sensitivity, the combination of the
lumbar drainage is 50% to 100% and specificity is 60%
cerebrospinal tap test with the intrathecal infusion test or
to 100%. Because of the small number of false-negative
ICP monitoring is recommended (▶ Fig. 8.5).
findings, a combination of external lumbar drainage
with the intrathecal infusion test or ICP monitoring is
recommended (▶ Fig. 8.6).
8.4 External Lumbar Drainage
Some authors85,86 recommend external lumbar CSF drain-
8.5 CSF and Serum
age after the cerebrospinal tap test or as a single diagnostic
method. At the same time, it should be noted that the CSF
Biochemical Tests
effusion rate of 5 to 10 mL/h is not exceeded and the
patient is maintained in a horizontal
(lying) position.
The level of amyloid β-peptides 1 to 42 is reduced in the
The drainage must be closed before the patient gets up.
CSF of patients with NPH, but the level of tumor necrosis
Fig. 8.6 External lumbar drainage.
52
Invasive Diagnostic Work-up
factor-α is increased.19 Li et al89 consider the leucine-rich
8.6 Diagnostic Pathway
α-2-glycoprotein in CSF to be a specific biomarker
for patients with iNPH. The biochemical investigations of
The following diagnostic procedures, shown in ▶ Fig. 8.7,
disease-specific biomarkers are in the development
should be initiated when there is a clinical suspicion of
phase and require clinical evaluation. Consequently, the
NPH. At the pre-inpatient stage, the investigation should
biomarkers are not suitable for use in clinical practice in
be based on CT or nuclear MRI for verifying internal
the diagnosis of iNPH.
hydrocephalus in which contextual, slight cortical
Fig. 8.7 Clinical pathway of
Suspicion of
idiopathic normal pressure
iNPH
hydrocephalus (iNPH) diagno-
sis.12 CSF, cerebrospinal fluid,
CT, computed tomography;
ICP, intracranial pressure.
Gait ataxia
no
+
No shunt
other symptom of
Hakim triad
yes
no
CT:Evans
No shunt
≥ 0.3
yes
or
Lumbar
ICP monitoring
infusion test
24 h
CSF tap test
R
out>13 or
yes
improvement after CSF tap test
or B wave-frequency > 50%
Lumbar CSF drainage
Shunt
48-72 h
surgery
no
yes
Clinical
No shunt
improvement
53
Invasive Diagnostic Work-up
atrophies, and/or periventricular hypodensities do not
CJJ, Eijndhoven JHM, van Maas AIR, Tans JTJ, eds. Intracranial.
Pressure VIII. Berlin, Heidelberg, New York: Springer Verlag;
represent predictors. The advantage of nuclear MRI is its
1993, pp. 749-752
greater certainty in the diagnosis regarding the commu-
[6]
Bannister R, Gilford E, Kocen R. Isotope encephalography in the diag-
nication between all CSF spaces as a prerequisite for the
nosis of dementia due to communicating hydrocephalus. Lancet
lumbar infusion test. Accordingly, the patient should be
1967; 2: 1014-1017
admitted for an ICP measurement over 48 hours or for
[7]
Børgesen SE, Gyldensted C, Gjerris F, Lester J. Computed tomography
and pneumoencephalography compared to conductance to outflow
the intrathecal infusion test. The indication for the
of CSF in normal pressure hydrocephalus. Neuroradiology 1980; 20:
ventricular infusion test is fulfilled only when communi-
17-22
cation between the CSF outlet paths cannot be demon-
[8]
Al-Zain FT, Rademacher G, Meier U, Mutze S, Lemcke J. The role of
strated with absolute certainty using neuroradiological
cerebrospinal fluid flow study using phase contrast MR imaging
in diagnosing idiopathic normal pressure hydrocephalus. Acta
imaging methods, if there are stenoses in the region of
Neurochir Suppl (Wien) 2008; 102: 119-123
spinal canal, or if there are clear clinical symptoms that
[9]
Meier U, Reichmuth B, Zeilinger FS, Lehmann R. The importance of
the lumbar infusion test shows physiologic values.
xenon-computed tomography in the diagnosis of normal pressure
Following the ICP measurement or postinfusion, the
hydrocephalus. Intern. J. Neuroradiology 1996; 2: 153-160
cerebrospinal tap test, with an intake of 40 to 60 mL, is
[10]
Adams RD, Fisher CM, Hakim S, Ojemann RG, Sweet WH. Sympto-
matic occult hydrocephalus with
“normal” cerebrospinal fluid
recommended. Following the computer-supported
pressure. A treatable syndrome. N Engl J Med 1965; 273: 117-126
evaluation of B-wave activity or the parameters of the
[11]
Meier U, Kiefer M, Sprung C. Normal. Pressure Hydrocephalus:
constant-rate infusion test, if there is pathologically
Pathology, Pathophysiology, Diagnostics, Therapeutics and Clinical
increased B-wave activity or pathologic outflow resist-
Course. Erwitte: PVV Science Publications; 2003
ances, respectively, then the indication for a shunt opera-
[12]
Meier U, Lemcke J. Zur Diagnostik des idiopathischen Normaldruck-
hydrozephalus aus der Perspektive von Langzeitbeobachtungen
tion has been established. An improvement in the clinical
Schweizer Archiv für Neurologie und Psychiatrie 2007; 158: 139-149
symptoms, particularly in gait ataxia, following CSF intake
[13]
Gaab MR, Haubitz J, Brawanski A, Faulstich J, Heißler HE. Pressure-
would confirm this indication. If an improvement in the
volume diagram, pulse amplitude and intracranial pulse volume.
spectrum of complaints following the cerebrospinal tap
Analysis and significance. In: Ishii S, Nagai H, Brock M. Intracranial
Pressure V. Berlin, Heidelberg, New York: Springer Verlag; 1983, pp.
test does not take place in patients with pathologic out-
261-268
flow resistance and very large compliance, then a lumbar
[14]
Kosteljanetz M. Intracranial pressure: cerebrospinal fluid dynamics
CSF drainage over 72 hours is indicated. With improving
and pressure-volume relations. Acta Neurol Scand Suppl 1987; 111:
clinical symptoms, particularly with gait ataxia, a shunt
1-23
operation should take place at a time interval of at least
[15]
Brean A, Eide PK. Assessment of idiopathic normal pressure patients
in neurological practice: the role of lumbar infusion testing for refer-
14 days following the investigation. The time interval
ral of patients to neurosurgery. Eur J Neurol 2008; 15: 605-612
referred to between intrathecal diagnosis and surgical
[16]
Eide PK, Brean A. Cerebrospinal fluid pulse pressure amplitude dur-
therapy is generally recommended in patients with NPH,
ing lumbar infusion in idiopathic normal pressure hydrocephalus can
because these patients are exceptionally sensitive and
predict response to shunting. Cerebrospinal Fluid Res 2010; 7: 5
[17]
Pfisterer WK, Aboul-Enein F, Gebhart E, Graf M, Aichholzer M, Mühl-
can react to abrupt CSF drainage with a worsening in
bauer M. Continuous intraventricular pressure monitoring for diag-
their clinical condition. Conservative therapy is recom-
nosis of normal-pressure hydrocephalus. Acta Neurochir
(Wien)
mended with physiologic B-wave activity or physiologic
2007; 149: 983-990, discussion 990
outflow resistance. If the gait ataxia worsens within
[18]
Frank AM, Alexiou C, Hulin P, Janssen T, Arnold W, Trappe AE.
6 months in these patients, then repeating the diagnostic
Non-invasive measurement of intracranial pressure changes by otoa-
coustic emissions (OAEs)—a report of preliminary data. Zentralbl
process should be considered carefully.
Neurochir 2000; 61: 177-180
[19]
Ishikawa M, Hashimoto M, Kuwana N et al. Guidelines for manage-
ment of idiopathic normal pressure hydrocephalus. Neurol Med Chir
References
(Tokyo) 2008; 48 Suppl: S1-S23
[20]
Marmarou A, Bergsneider M, Relkin N, Klinge P, Black PM. Develop-
[1] Cardoso ER, Del Bigio MR. Age-related changes of cerebral ventricular
ment of guidelines for idiopathic normalpressure hydrocephalus:
size. Part II: Normalization of ventricular size following shunting.
introduction. Neurosurgery 2005; 57(3, Suppl): S1-S3,discussion ii-v
Acta Neurochir (Wien) 1989; 97: 135-138
[21]
Albeck MJ, Børgesen SE, Gjerris F, Schmidt JF, Sørensen PS. Intra-
[2] Casmiro M, D’Alessandro R, Cacciatore FM, Daidone R, Calbucci F,
cranial pressure and cerebrospinal fluid outflow conductance in
Lugaresi E. Risk factors for the syndrome of ventricular enlargement
healthy subjects. J Neurosurg 1991; 74: 597-600
with gait apraxia (idiopathic normal pressure hydrocephalus): a
[22]
Marmarou A, Shulman K, LaMorgese J. Compartmental analysis of
case-control study. J Neurol Neurosurg Psychiatry 1989; 52: 847-852
compliance and outflow resistance of the cerebrospinal fluid system.
[3] Meier U, Mutze P. Correlation between decreased ventricular
J Neurosurg 1975; 43: 523-534
size and positive clinical outcome following shunt placement in
[23]
Katzman R, Hussey F. A simple constant-infusion manometric test for
patients with normal-pressure hydrocephalus. J Neurosurg 2004;
measurement of CSF absorption. I. Rationale and method. Neurology
100: 1036-1040
1970; 20: 534-544
[4] Børgesen SE, Gjerris F, Sørensen SC. Cerebrospinal fluid conductance
[24]
Künzel B, Klages G, Kynast J, Meier U, Warnke JP. A mathematical
and compliance of the craniospinal space in normal-pressure hydro-
model of the cerebrospinal fluid dynamics and the simultaneous
cephalus. A comparison between two methods for measuring con-
determination of outflow resistance and compliance. In: Willems
ductance to outflow. J Neurosurg 1979; 51: 521-525
JL, Bemmel JH van, Michel J, eds. Progress in Computer-assisted
[5] Tans JTJ, Poortvliet DCJ. Comparison of lumbar and ventricular
Function Analysis. North-Holland: Elsevier Science;
1988, pp.
constant flow and bolus infusions in hydrocephalus. In: Avezaat
393-398
54
Invasive Diagnostic Work-up
[25]
Meier U. Der intrathekale Infusionstest als Entscheidungshilfe zur
[44]
Meier U, Bartels P. The importance of the intrathecal infusion test in
Shunt-Operation beim Normaldruckhydrozephalus. Akt Neurol
the diagnosis of normal pressure hydrocephalus. J Clin Neurosci
1997; 24: 24-34
2002; 9: 260-267
[26]
Hebb AO, Cusimano MD. Idiopathic normal pressure hydrocephalus:
[45]
Børgesen SE, Gjerris F. Relationships between intracranial pressure,
a systematic review of diagnosis and outcome. Neurosurgery 2001;
ventricular size, and resistance to CSF outflow. J Neurosurg 1987; 67:
49: 1166-1184, discussion 1184-1186
535-539
[27]
Kahlon B, Sundbärg G, Rehncrona S. Comparison between the lumbar
[46]
Governale LS, Fein N, Logsdon J, Black PM. Techniques and complica-
infusion and CSF tap tests to predict outcome after shunt surgery in
tions of external lumbar drainage for normal pressure hydrocephalus.
suspected normal pressure hydrocephalus. J Neurol Neurosurg
Neurosurgery 2008; 63 Suppl 2: 379-384, discussion 384
Psychiatry 2002; 73: 721-726
[47]
Tans JTJ, Poortvliet DCJ. Significance of compliance in adult hydro-
[28]
Fuhrmeister U. Liquorabflußwiderstand und intrakranielle Elastizität
cephalus. In: Gjerris F, Børgesen SE, Sørensen PS, eds. Outflow of
bei akuten und chronischen Erkrankungen des Subarachnoidalraums.
Cerebrospinal Fluid. Alfred Benzon Symposium
27. Copenhagen:
Würzburg: Habilitationsschrift: 1985
Munksgaard; 1989, pp. 272-279
[29]
Børgesen SE, Gjerris F. Relationships between intracranial pressure,
[48]
Pappenheimer JR, Heisey SR, Jordan EF, Downer JC. Perfusion of the
ventricular size, and resistance to CSF outflow. J Neurosurg 1987; 67:
cerebral ventricular system in unanesthetized goats. Am J Physiol
535-539
1962; 203: 763-774
[30]
Børgesen SE, Gjerris F, Schmidt J. Measurement of resistance to CSF
[49]
Kosteljanetz M. CSF dynamics and pressure-volume relationships in
outflow by subarachnoid perfusion. In: Gjerris F, Børgesen SE,
communicating hydrocephalus. J Neurosurg 1986; 64: 45-52
Sørensen PS, eds. Outflow of Cerebrospinal Fluid. Alfred Benzon
[50]
Gjerris F, Børgesen SE, Hoppe E, Boesen F, Nordenbo AM. The con-
Symposium 27. Copenhagen: Munksgaard; 1989, pp. 121-129
ductance to outflow of CSF in adults with high-pressure hydrocepha-
[31]
Tychmanowicz K, Czernicki Z, Pawłowski G, Stepińska G. ICP depen-
lus. Acta Neurochir (Wien) 1982; 64: 59-67
dent changes of CSF outflow resistance. Acta Neurochir (Wien) 1992;
[51]
Gjerris F, Børgesen SE, Schmidt J, Sørensen PS. Resistance to cerebro-
117: 44-47
spinal fluid outflow in patients with normal pressure hydrocephalus.
[32]
Black PM. Normal-pressure hydrocephalus: current understanding
In: Gjerris F, Børgesen SE, Sørensen PS, eds. Outflow of Cerebrospinal
of diagnostic tests and shunting. Postgrad Med 1982; 71: 57-61,
Fluid. Alfred Benzon Symposium
27. Copenhagen: Munksgaard;
65-67
1989, pp. 329-338
[33]
Butler AB, Mann JD, Maffeo CJ, Dacey RG Jr, Johnson RN, Bass NH.
[52]
Marmarou A, Shulman K, LaMorgese J. A compartmental analysis
Mechanisms of cerebrospinal fluid absorption in normal and
of compliance and outflow resistance and the effects of elevated
pathologically altered arachnoid villi. In: Woods JH, ed. Neuro-
blood pressure. In: Lundberg N, Pontén U, Brock M, eds. Intracranial
biology of Cerebrospinal Fluid. New York, NY: Plenum Press; 1983,
Pressure II. Berlin, Heidelberg, New York: Springer Verlag; 1975, pp.
pp. 707-726
86-88
[34]
Chazal J, Janny P, Georget AM, Colnet G. Benign intracranial hyper-
[53]
Marmarou A, Shulman K, Rosende RM. A nonlinear analysis of the
tension. A clinical evaluation of the CSF absorption mechanisms. Acta
cerebrospinal fluid system and intracranial pressure dynamics.
Neurochir Suppl (Wien) 1979; 28: 505-508
J Neurosurg 1978; 48: 332-344
[35]
Shapiro K, Fried A. Shunt dependent hydrocephalus: pressure volume
[54]
Künzel B. Mathematisch-physikalische Interpretation der kraniospi-
characterization and altered CSF outflow resistance. In: Miller JD,
nalen. Druckdynamik. [dissertation]. Berlin: Humboldt-Universität zu
Teasdale GM, Rowan JO, Galbraith SL, Mendelow AD, eds. Intracranial
Berlin; 1993
Pressure VI. Berlin, Heidelberg, New York: Springer Verlag; 1986, pp.
[55]
Chadduck WM, Seibert JJ, Adametz J, Glasier CM, Crabtree M, Stansell
118-122
CA. Cranial Doppler ultrasonography correlates with criteria for ven-
[36]
Sullivan HG, Miller JD, Griffith RL, Carter W, Rucker S. Bolous versus
triculoperitoneal shunting. Surg Neurol 1989; 31: 122-128
steady-state infusion for determination of CSF outflow resistance.
[56]
Kosteljanetz M, Nehen AM, Kaalund J. Cerebrospinal fluid outflow
Ann Neurol 1979; 5: 228-238
resistance measurements in the selection of patients for shunt sur-
[37]
Love JA, Ekstedt J, Fridén H. Labile sagittal sinus pressures in the cat.
gery in the normal pressure hydrocephalus syndrome. A controlled
In: Miller JD, Teasdale GM, Rowan JO, Galbraith SL, Mendelow AD,
trial. Acta Neurochir (Wien) 1990; 104: 48-53
eds. Intracranial Pressure VI. Berlin, Heidelberg, New York: Springer
[57]
Miller JD, Takizawa H. Validity of measurements of CSF outflow
Verlag; 1986, pp. 132-134
resistance estimated by the bolus injection method. In: Miller JD,
[38]
Meier U, Zeilinger FS, Kintzel D. Diagnostic in normal pressure hydro-
Teasdale GM, Rowan JO, Galbraith SL, Mendelow AD, eds. Intracranial
cephalus: a mathematical model for determination of the ICP-depen-
Pressure VI. Berlin, Heidelberg, New York: Springer Verlag; 1986, pp.
dent resistance and compliance. Acta Neurochir (Wien) 1999; 141:
105-107
941-947, discussion 947-948
[58]
Shapiro K. Influences of the skull and dura on the resistance to CSF
[39]
Meier U, Kiefer M, Bartels P. The ICP-dependency of resistance to
outflow. In: Gjerris F, Børgesen SE, Sørensen PS, eds. Outflow of Cere-
cerebrospinal fluid outflow: a new mathematical method for CSF-
brospinal Fluid. Alfred Benzon Symposium 27. Copenhagen: Munks-
parameter calculation in a model with H-TX rats. J Clin Neurosci
gaard; 1989, pp. 215-221
2002; 9: 58-63
[59]
Ekstedt J. CSF hydrodynamics studied by means of constant pressure
[40]
Ekstedt J. CSF hydrodynamic studies in man. 2. Normal hydro-
infusion technique. In: Lundberg N, Pontén U, Brock M, eds. Intra-
dynamic variables related to CSF pressure and flow. J Neurol Neuro-
cranial Pressure II. Berlin, Heidelberg, New York: Springer Verlag;
surg Psychiatry 1978; 41: 345-353
1975, pp. 35-41
[41]
Shapiro K, Marmarou A, Shulman K. A method for predicting PVI in
[60]
Ekstedt J. CSF hydrodynamic studies in man. 1. Method of constant
normal patients. In: Shulman K, Marmarou A, Miller JD, Becker DP,
pressure CSF infusion. J Neurol Neurosurg Psychiatry
1977; 40:
Hochwald GM, Brock M, eds. Intracranial Pressure IV. Berlin, Heidelberg,
105-119
New York: Springer Verlag; 1980, pp. 85-90
[61]
Fuhrmeister U, Ruether P, Dommasch D, Gaab M. Alterations of CSF
[42]
Boon AJ, Tans JT, Delwel EJ et al. Does CSF outflow resistance predict
hydrodynamics following meningitis and subarachnoid hemorrhage.
the response to shunting in patients with normal pressure hydro-
In: Shulman K, Marmarou A, Miller JD, Becker DP, Hochwald GM,
cephalus? Acta Neurochir Suppl (Wien) 1998; 71: 331-333
Brock M, eds. Intracranial Pressure IV. Berlin, Heidelberg, New York:
[43]
Meier U, Reichmuth B, Knopf W, Riederer A. Intrathecal infusion test:
Springer Verlag; 1980, pp. 241-244
an investigative method to treat malresorptive hydrocephalus by
[62]
Price DJ. The clinical value of measurement of CSF outflow resistance.
shunt operation. In: Lorenz R, Klinger M, Brock M, eds. Advances in
In: Gjerris F, Børgesen SE, Sørensen PS, eds. Outflow of Cerebrospinal
Neurosurgery 21. Berlin, Heidelberg, New York: Springer Verlag;
Fluid. Alfred Benzon Symposium
27. Copenhagen: Munksgaard;
1993, pp. 125-129
1989, pp. 390-398
55
Invasive Diagnostic Work-up
[63]
Sakomoto H, Nakamura T, Marmarou A, Becker DP. Comparison of
[76]
Meier U, König A, Miethke C. Predictors of outcome in patients with
CSF formation and outflow resistance measured by ventriculo-cister-
normal-pressure hydrocephalus. Eur Neurol 2004; 51: 59-67
nal perfusion and volume manipulation techniques. In: Miller JD,
[77]
Portnoy HD, Croissant PD. A practical method for measuring hydro-
Teasdale GM, Rowan JO, Galbraith SL, Mendelow AD, eds. Intracranial
dynamics of cerebrospinal fluid. Surg Neurol 1976; 5: 273-277
Pressure VI. Berlin, Heidelberg, New York: Springer Verlag; 1986, pp.
[78]
Ekstedt J, Fridén H. Estimation of CSF outflow resistance in humans:
108-110
infusion methods. In: Gjerris F, Børgesen SE, Sørensen PS, eds.
[64]
Schmidt JF, Fedders O, Børgesen SE, Gjerris F. Reproducibility of mea-
Outflow of Cerebrospinal Fluid. Alfred Benzon Symposium
27.
surements of resistance to CSF outflow. In: Gjerris F, Børgesen SE,
Copenhagen: Munksgaard; 1989, pp. 148-165
Sørensen PS, eds. Outflow of Cerebrospinal Fluid. Alfred Benzon Sym-
[79]
Sprung C, Collmann H, Fuchs FC, Suwito S, Duisberg R. Pre- and post-
posium 27. Copenhagen: Munksgaard; 1989, pp. 224-229
operative evaluation of hydrocephalus using the infusion test. In:
[65]
Marmarou A, Shulman K. Computer modelling of CSF pressure/vol-
Wüllenweber R, Brock M, Hamer J, Klinger M, Spoerri O, eds. Lumbar
ume and its relationship to hydrocephalus. In: Brock M, Dietz H, eds.
Disc. Adult Hydrocephalus. Berlin, Heidelberg, New York: Springer
Intracranial Pressure I. Berlin, Heidelberg, New York: Springer Verlag;
Verlag; 1977, pp. 161-167
1972, pp. 275-279
[80]
Malm J, Kristensen B, Fagerlund M, Koskinen LO, Ekstedt J. Cerebro-
[66]
Marmarou A, Shulman K. Pressure-volume relationships - basic
spinal fluid shunt dynamics in patients with idiopathic adult
aspects. In: Laurin RL, ed. Head Injuries. New York, NY: Grune and
hydrocephalus syndrome. J Neurol Neurosurg Psychiatry 1995; 58:
Strattton; 1976, pp. 233-236
715-723
[67]
Künzel B, Klages G, Meier U. Der intrathekale Infusionstest zur Unter-
[81]
Wikkelsö C, Andersson H, Blomstrand C, Lindqvist G, Svendsen P.
suchung der kraniospinalen Liquordynamik. Ein neues Auswertever-
Normal pressure hydrocephalus. Predictive value of the cerebrospinal
fahren [The intrathecal infusion test in the study of craniospinal cere-
fluid tap-test. Acta Neurol Scand 1986; 73: 566-573
brospinal fluid dynamics. A new assessment procedure]. Zentralbl
[82]
Ishikawa M, Kikuchi H. Lumbar CSF pressure measurement in idio-
Neurochir 1987; 48: 320-326
pathic normal pressure hydrocephalus in the aged. In: Nagai H,
[68]
Charlton JD, Johnson RN, Pederson NE, Mann JD. Assessment of cere-
Kamiya K, Ishii P, eds. Intracranial Pressure IX. Berlin, Heidelberg,
brospinal fluid compliance and outflow resistance: analysis of
New York: Springer Verlag; 1994, pp. 448-449
steady-state response to sinusoidal input. Ann Biomed Eng 1983; 11:
[83]
Sand T, Bovim G, Grimse R, Myhr G, Helde G, Cappelen J. Idiopathic
551-561
normal pressure hydrocephalus: the CSF tap-test may predict the
[69]
Marmarou A, Shapiro K, Pöll W, Shulmann K. Studies of kinetics of
clinical response to shunting. Acta Neurol Scand 1994; 89: 311-316
fluid movements within brain tissue. In: Beks JWF, Bosch AD, Brock
[84]
Ravdin LD, Katzen HL, Jackson AE, Tsakanikas D, Assuras S, Relkin NR.
M, eds. Intracranial Pressure III. Berlin, Heidelberg, New York:
Features of gait most responsive to tap test in normal pressure hydro-
Springer Verlag; 1976, pp. 1-4
cephalus. Clin Neurol Neurosurg 2008; 110: 455-461
[70]
Berman B, Agarwal G. An integrative approach to intracranial hydrau-
[85]
Haan J, Thomeer RTWM. Predictive value of temporary external lum-
lic physiology. I. Basic concepts, pressure-volume relationships, and
bar drainage in normal pressure hydrocephalus. Neurosurgery 1988;
infusion studies. Surg Neurol 1984; 22: 83-95
22: 388-391
[71]
Shulman K, Marmarou A. Weitz P. Gradients of brain interstitial
[86]
Walchenbach R, Geiger E, Thomeer RTWM, Vanneste JAL. The value
fluid pressure in experimental brain infusion and compression. In:
of temporary external lumbar CSF drainage in predicting the
Lundberg N, Pontén U, Brock M, eds. Intracranial Pressure II. Berlin,
outcome of shunting on normal pressure hydrocephalus. J Neurol
Heidelberg, New York: Springer Verlag; 1975, pp. 221-223
Neurosurg Psychiatry 2002; 72: 503-506
[72]
Kiefer M, Eymann R, Steudel WI. The dynamic infusion test in rats.
[87]
Sharma AK, Gaikwad S, Gupta V, Garg A, Mishra NK. Measurement of
Childs Nerv Syst 2000; 16: 451-456
peak CSF flow velocity at cerebral aqueduct, before and after lumbar
[73]
Czosnyka M, Gjerris F, Maksymowicz W, et al. Computerised lumbar
CSF drainage, by use of phase-contrast MRI: utility in the manage-
infusion test - Multicentre experience in clinical studies in hydro-
ment of idiopathic normal pressure hydrocephalus. Clin Neurol
cephalus. In: Nagai H, Kamiya K, Ishii P, eds. Intracranial Pressure IX.
Neurosurg 2008; 110: 363-368
Berlin, Heidelberg, New York: Springer Verlag; 1994, pp. 494-495
[88]
Komotar RJ, Zacharia BE, Mocco J, Kaiser MG, Frucht SJ, McKhann GM.
[74]
Sklar FH, Beyer CW, Ramanathan M, Elashvili I, Cooper PR, Clark WK.
Cervical spine disease may result in a negative lumbar spinal
Servo-controlled lumbar infusions: a clinical tool for the determina-
drainage trial in normal pressure hydrocephalus: case report.
tion of CSF dynamics as a function of pressure. Neurosurgery 1978;
Neurosurgery 2008; 63 (Suppl 2): 315
3: 170-175
[89]
Li X, Miyajima M, Mineki R, Taka H, Murayama K, Arai H. Analysis of
[75]
Meier U, Zeilinger FS, Kintzel D. Signs, symptoms and course of nor-
potential diagnostic biomarkers in cerebrospinal fluid of idiopathic
mal pressure hydrocephalus in comparison with cerebral atrophy.
normal pressure hydrocephalus by proteomics. Acta Neurochir
Acta Neurochir (Wien) 1999; 141: 1039-1048
(Wien) 2006; 148: 859-864
56
9.1
Definition
58
Chapter 9
9.2
Types
58
Shunt and Valve Technology
9.3
Physical Basics
58
Christoph Miethke
9.4
Hydrostatic Pressure
58
9.5
Physics of Ventriculoperitoneal
Shunts
59
9.6
Ventriculoatrial Shunts
61
9.7
Classification of Valves
61
9.8
Adjustable Differential Pressure
Valves
64
9.9
Hydrostatic Devices
67
Shunt and Valve Technology
9 Shunt and Valve Technology
Christoph Miethke
Scientific knowledge is based on independent research
5,000 m drops down to 540 mbar. Consequently, the
and observed evidence. The evidence for the superior-
importance of pressure depends on the location. This
ity of a specific valve design has been of interest since
is true within a gas environment, and—even more
the first shunt was implanted. Many publications have
importantly—within a fluid system.
reported on the clinical experience with different
However, the intracranial pressure (ICP) is defined
types of valves; however, prospective randomized tri-
as the difference between the absolute pressure within
als could not reveal significant differences.1-3 For the
the ventricular system and the outer atmospheric
first time ever, significant differences were shown by
pressure. Merritt and Fremont-Smith9 reported on ICP
Meier and co-workers4-6
who compared adjustable
measurements in 1,033 healthy persons and found an
differential pressure
(DP) valves with and without a
average value of 13 cm H2O with a peak at 15 cm H2O
gravitational unit. His results confirm the understanding
(▶ Fig. 9.1).
that the hydrostatic pressure within a shunt system is
In a healthy person, ICP is determined by the
the outstanding factor that must be taken into account
production of CSF and its absorption. The production
and is systematically controlled by the characteristics of
of CSF is independent of the pressure gradient
the valve.
between the ventricular system and the aterial blood
vessels. However, the absorption depends on the
gradient between the intraventricular and venous
9.1 Definition
pressures. As the venous pressure increases, the ICP
also increases.
A shunt is an artificial hydraulic connection between dif-
ferent compartments within the human body. In hydro-
cephalus, a shunt is used to connect a compartment filled
9.4 Hydrostatic Pressure
with cerebrospinal fluid (CSF) and a cavity within the
body where the drained fluid can be absorbed. In neuro-
Hydrostatic pressure is of utmost importance for the
surgical practice, the ventriculoperitoneal (VP) shunt is
physics of CSF drainage and shunts. It is defined as
used most often. Usually a shunt includes a valve, which
controls drainage throughout the shunt. Shunts without a
ph ¼
g h
ð15Þ
valve are very rarely implanted.
where ph is the hydrostatic pressure (cm H2O), δ is the
9.2 Types
specific weight of the fluid (kg/L), g is the acceleration
due to gravity (g = 9.80665 m/s2), and h is the height of
The history of the treatment of hydrocephalus reveals
the water column (WC; cm).
a wide variety of different approaches in draining CSF
into a compartment within the human body where it
can be resorbed. Most of these attempts have not been
introduced in clinical practice and are only chosen
today in cases when one of the already established
shunt types fails for any reason. Nowadays the implan-
tation of a VP shunt has become the standard
practice.7
Other options are the ventriculoatrial (VA),
the lumboperitoneal
(LP), or the rarely implanted
ventriculopleural shunt.8
9.3 Physical Basics
The absolute atmospheric pressure depends on sea-
sonal changes in the weather, and the altitude of the
location. At sea level, the normal atmospheric pressure
is defined as 10.24 m H2O (1,024 mbar). At high alti-
tudes, the atmospheric pressure decreases with
Fig. 9.1 Frequency-distribution curve for the intracranial pres-
increasing height. If the pressure at sea level is 1,024
sure in 1,033 normal human participants.9
mbar, then the atmospheric pressure at a height of
58
Shunt and Valve Technology
9.5 Physics of Ventriculoperito-
neal Shunts
Every shunt has an inlet and an outlet. The outlet is con-
nected to the absorption cavity (normally the abdomen
or the right atrium of the heart), while the inlet is con-
nected to the ventricular system of the brain. Two factors
determine how much CSF will be drained through the
shunt: the opening characteristic of the valve and the
pressure situation at the outlet of the valve.
A container filled with water has an outlet at its
Fig. 9.2 Negative pressure within (a) straw and in (b) tube.
bottom, where a tube is fixed with an integrated valve.
The opening pressure of the valve defines the level of the
water within the container, together with the pressure at
The hydrostatic pressure in a fluid increases with its
the outlet of the distal tubing. If the distal tubing is low-
depth. ▶ Fig. 9.2 demonstrates a simple experiment. The
ered, then the water level within the container is lowered
atmospheric pressure acts on the surface of the water.
accordingly. In this model, the container can be seen as
The absolute pressure under the surface increases with
the ventricular system, and the opening at the outlet
represents the pressure situation within the abdominal
the depth. If a straw is put into a glass of water and the
straw is closed by a finger and moved out of the fluid,
cavity. For this pressure not only is the value relevant (in
then the water remains within the straw; now the surface
this case, the atmospheric pressure), but the position is
of the water is below the column. The atmospheric pres-
also important (▶ Fig. 9.4). This phenomenon is indepen-
dent of whether or not the container is open or closed. If,
sure acts on the lower outlet of the straw, and the closing
for example, the container is covered with a thin mem-
finger at the upper outlet is pulled into the straw due to
the negative pressure within the straw. The higher the
brane, then the membrane will sink if the distance
point where the pressure is measured within the straw,
between the outlet level of the tubing and the zero-level
the lower the pressure. It is also clear that the hydrostatic
(an imaginary water level) is higher than the opening
pressure of the valve and allows flow throughout the
pressure is higher as one measures deeper in the water
reservoir. Within the straw, the pressure is the lowest at
shunt (▶ Fig. 9.5). The drainage stops at the moment
the top end of the straw, where the finger closes the
when the pressure is at the zero level.
straw. The finger is pulled into the straw because
The resulting pressure situation within the covered box
the pressure is lower than the surrounding atmospheric
is not affected by the characteristics of the membrane. If
the membrane is stiff, like bone, then the drainage and
pressure.
If the reference point is in the middle of the tube, then
the physics of the system can still be correctly described
the pressure becomes negative going up and positive
by this model (▶ Fig. 9.6). The pressure within the hard
going down the tube (▶ Fig. 9.3).
box (the ventricular system within the head) is deter-
Understanding hydrostatic pressure is also important
mined by the outlet pressure and the level of the outlet
together with the opening performance of the valve.
for the peritoneal cavity. Within the peritoneal cavity
exists a hydrostatic pressure that becomes positive going
Within the closed container there is obviously hydro-
down and negative going up. The important question is:
static pressure that is becoming lower at a higher point
Where is the reference point, the zero level?
within the hydraulic system and higher at a lower point.
Hence, the ICP of a shunted patient is first of all a result
of the pressure in the volume into which the fluid
is going to be drained. Currently, the abdomen is the
preferred site.
If the pressure within the abdomen is so important for
ICP, then how can this model be considered an open sys-
tem? The fluid being drained into the abdominal cavity is
normally not effecting the pressure situation within the
abdomen; it is easily absorbed. Normally, the peritoneum
is opened during surgery and the catheter is inserted
without any control of the placement of the distal end.
When the abdomen is opened, the fluid it contains can be
seen between the organs of the abdomen. This fluid nor-
Fig. 9.3 Pressure changes within a tube with the reference level
at one side.
mally can freely communicate between the organs within
the abdomen, and it establishes a hydrostatic pressure
59
Shunt and Valve Technology
Fig. 9.4 Function of differential pressure valves.
Fig. 9.5 Container with a shunt covered by a thin membrane.
within the abdomen. For this reason, it is not important
where the distal end of the peritoneal catheter is placed;
however, the reference point (zero level) within the peri-
toneal cavity is important. It is obvious that the pressure
within the peritoneal cavity depends on the activities
of daily life of each individual patient. The nutritional
condition, individual anatomic variations, movements, or
features due to illness have a definitive impact on the pres-
sure situation. However, on average, the widely accepted
assumption is that, for a person in a horizontal position on
his or her back, the zero level of the peritoneal pressure
is at the point of the upper abdominal skin (a study by
Freimann and Sprung) (▶ Fig. 9.7). This means that, for a
patient who is obese, this point should be generally con-
sidered to be higher than for a patient who is thin. In
terms of adequate shunting and avoidance of overdrainage,
Fig. 9.6 Hard box model of the ventricles within the head with
the horizontal position is logically less important than the
an integrated shunt.
upright position. Normally overdrainage in the upright
60
Shunt and Valve Technology
not change pressure. The zero-level reference remains
more or less at the same level regardless of the bulk flow
of the CSF.
9.6 Ventriculoatrial Shunts
In contrast to VP shunts, the placement of an atrial cathe-
ter on the distal tip of a VA catheter is very important for
Fig. 9.7 Model of a differential pressure valve in a patient lying
the resulting ICP. The pressure within the vena cava,
in a horizontal position.
the right atrium, or the right heart varies significantly.
Consequently, precise positioning of the tube is manda-
tory. In principle, the function of shunts as described
above is also true for VA shunts. The posture-dependent
changing values are different because the reference
points are different. Therefore, the physics of VA shunts
require a posture-dependent function as with VP shunts.
Depending on individual anatomy, the hydrostatic differ-
ence from the foramen of Monro to the right atrium is
lower than the distance to the diaphragm. Accordingly,
the hydrostatic compensation in the upright patient is
lower. Generally, the percentage of complications with
VP and VA shunts seems to be comparable, although
complications with VA shunts are more severe and more
Fig. 9.8 Shunt physics of a differential pressure valve in a
difficult to treat.11-14
patient in the upright position.
9.7 Classification of Valves
position is more challanging. However, more seldomly
All available valves can be classified into four subtypes.
underdrainage in the horizontal position in obese patients
▶ Table 9.1 gives an overview. The classification follows
might be a problem as well.
When the patient is in the upright position, the physics
mechanical characteristics. Basically, there are only two
groups, namely DP and hydrostatic valves. In principle,
within the abdomen changes because the direction of
the option of a noninvasive readjustment does not change
gravity is different. The abdomen is still a hydraulic com-
the hydraulic performance. The valve works following the
partment with hydrostatic pressure; however, the zero
adjustment, but it is still a DP or a hydrostatic valve. The
level in the upright patient moves toward the region of
the diaphragm (▶ Fig. 9.8).
adjustment simply avoids the otherwise necessary valve
change. However, once adjusted it works like a nonadjust-
For any position of the shunted patient, a “zero level”
able valve during the daily life of the patient.
exists as a reference point for the function of the shunt.
DP valves were the first valves to be used successfully
Essential for the flow throughout the shunt is how far,
in the treatment of hydrocephalus. Spitz, Holter, and Nul-
how often, and how long does this reference point (zero
level) shift. The average rate of production of CSF is
sen were the pioneers who introduced these valves in the
early 1950s.15-18 This was a definitive breakthrough in
known to be about 20 mL/h. A shunt allows a flow that is
the treatment of hydrocephalus and the beginning of
several times higher than this rate of production.10 There-
fore, in cases of temporarily increased intraperitoneal
pressure, the shunt can easily compensate for under-
Table 9.1 Classification of valves
drainage as soon the peritoneal pressure drops to normal
Valve
Fixed
Adjustable
values. For this reason, the risk of underdrainage is not
very relevant as long as the pressure shift within the
DP valves
abdomen does not occur for periods longer than an hour
Silicon slit
X
or two. This is different for the opposite situation: if the
Membrane
X
abdominal pressure decreases temporarily for any reason,
Ball-in-cone
X
X
this automatically leads to decreased ICP, which is not as
Hydrostatic valve principles
easily compensated as underdrainage because of the low
ASD technology
X
rate of production of CSF compared to the high flow rate
Flow reducing
X
of a shunt.
Gravitational
X
X
The absorption capacity of the peritoneum is so high
that the CSF, which is drained into the peritoneum, does
Abbreviations: ASD, antisiphon device; DP, differential pressure.
61
Shunt and Valve Technology
modern shunt therapy. Since then, many different models
that a subdural effusion does not always develop in all
have been proposed to improve the clinical outcome or
patients with a negative pressure in the upright position
the surgical treatment.
does not mean that the ICP is not significantly negative.
In patients receiving shunt therapy, ICP generally is
Regardless of whether or not there is a clinical complica-
controlled by the opening characteristic of the valve and
tion in all shunts with DP valves, the pressure in the
—even more importantly—by the pressure within the
upright position becomes negative. Therefore, a DP valve
peritoneal cavity. The valve simply transmits the refer-
is an insufficient solution in principle, even though it sol-
ential pressure within the peritoneal cavity to the ven-
ves the underlying problem of the pressure crisis due to
tricles in the brain by adding the value of the opening
hydrocephalus.
pressure. A noted drawback of DP valves is the fact that
Despite these considerations, various types of DP valves
they do not take into account the posture-dependent
have been developed and implanted with good clinical
physics of shunts in patients with hydrocephalus.
success. However, clinical studies have not confirmed one
Although the first proposals to overcome problems due
or other valve design as being superior.2 However, labora-
to overdrainage go back to the 1960s and 1970s,19 the
tory investigations have revealed significant mechanic
importance of a consequent hydraulic compensation of
and hydraulic differences.
the posture-dependent physics is still underestimated
The oldest design in the group of DP valves, the sili-
and is seen as a rare or not-too-important complication.
cone-slit valve, shows a flow-reducing performance
Some clinical reviews report a very low incidence of
that is somewhat similar to the performance of the so-
overdrainage, especially in children.2,20 However, on the
called flow-reducing devices.18,23,24 The membrane and
other hand, the list of overdrainage-related complica-
ball-in-cone valves have a similar hydraulic perform-
tions in children and in adults is long and well described
ance, whereas the ball-in-cone valves demonstrate the
in other papers.21,22
most reliable function and precision.25,26 The silicone-
DP valves, neither fixed pressure valves nor adjustable
slit valves get their characteristics from the stiffness of
devices, do not and cannot fulfill the requirements for
the silicone. The simplest model of a silicone-slit valve
both horizontal and upright position of patients. In the
is a catheter with a closed end and one or several slits
upright position, the reference point within the perito-
close to the end of the tube. The stiffer the material,
neum, on average, is the diaphragm. At this point, the
the thicker the wall of the tube, and the smaller the
inner pressure is more or less equal to the atmospheric
slit (incision) in the tube, the fewer slits there are, the
pressure. The implanted shunt, together with the ven-
higher the opening pressure becomes. More sophisti-
tricular system of the patient and the peritoneum, is a
cated models show a cross-incision at the closed end
hydraulic system of connected compartments. Again,
of the tubing (▶ Fig. 9.9). Silicone-slit valves are rarely
based on the pressure situation in the abdominal cav-
used today.
ity, the DP valve transmits the reference point, or the
The second type of nonadjustable DP valve is the
zero-level.
membrane valve. The opening performance of this
A typical DP valve with an opening pressure of 10cm
device is defined by the stiffness of a membrane, which
H2O, for example, can shift this zero level 10cm above the
closes the pathway by sealing against a valve seat. The
diaphragm. ICP within the ventricles at the foramen of
normally flat, rounded membrane is fixed at the valve
Monro of normal healthy individuals without a shunt is
housing in the way that the valve is closed; the mem-
about 0 cm H2O, which means that it is about equal to the
brane covers the valve seat. If the DP acting on the valve
atmospheric pressure at this point. Depending on the
seat exceeds the opening pressure, then the flexible sili-
height of the individual patient, the DP valve will have to
cone membrane is displaced from the valve seat at the
compensate the height difference from the diaphragm to
outer end and the valve opens. To achieve a different
the ventricles. In adult patients this typically is about 30
opening pressure for the different valve ranges, mem-
to 40 cm H2O; in children or newborns it is accordingly
branes of different thickness are used. The membrane
less. A DP valve with an opening pressure of only 10 cm
gets thicker and becomes stiffer as the opening pressure
H2O, which, for the horizontal position fulfills the
rises (▶ Fig. 9.10).
requirement of a physiological ICP, keeps ICP at negative
values in the upright position: -20 to -30 cm H2O in
adults, and -10 to -20 cm H2O in children. These values
are not physiological, neither in adults nor children.
Whether or not this fact leads to complications such as
subdural effusions, ventricular collapse, or headache
seemingly depends on individual structures and their
ability to withstand this suction. As ICP becomes more
negative, there is a higher risk of hematoma, for example,
Fig. 9.9 Typical silicone-slit valve.
as a consequence of a ruptured bridging vein. The fact
62
Shunt and Valve Technology
Fig. 9.10 Closed and open membrane valve.
Fig. 9.12 Ball-in-cone valve with different springs and valve seats.
but the seat is integrated into a movable membrane.
The ball is part of the fixed housing. If the DP at the
valve seat increases, then it overcomes the spring force
at a certain point, which defines the opening character-
istic of the valve, and the valve opens. If the area on
which the pressure is acting is more than 200 times
larger than in any ball-in-cone valve, then the force to
overcome possible sticking effects is 200 times larger
than in the other ball-in-cone valve. Clinical papers
reporting the experience with these devices have con-
firmed their reliability.28-37
Fig. 9.11 Ball-in-cone valve.
A different concept for lowering the likelihood of block-
age is the underlying concept of the smallest available DP
valve. The miniNAV has dimensions that are barely larger
Ball-in-cone valves represent the third group of DP
than the diameter of the catheter. The channels within
valves available on today’s market. A round valve seat is
the valve are smaller than the inner diameter of perito-
closed by a metallic or sapphire ball, and it is kept in the
neal tubes. At any place within the valve the flow is
seat by the force of a metallic spring (▶ Fig. 9.11).
always higher than within the catheter; this might lower
The spring can be a classical cylindrical or a kind of leaf
the risk of protein collection within the valve. However,
spring. The opening pressure of such a valve can be calcu-
there is no clinical evidence for the superiority of one DP
lated by
valve design over the other, neither in terms of blockage
nor in terms of function (▶ Fig. 9.13).
p¼F
ð16Þ
A
where p is the opening pressure of the device, F is the
force of the spring, and A is the area closed by the ball at
the valve seat.
Consequently, the spring force needed to establish a
certain opening pressure depends on the diameter of
the valve seat. For hydrocephalus valves, this aspect
offers the opportunity to decrease the influence of
sticking particles in the CSF on the valve function by
choosing large areas with consequently strong forces27
(▶ Fig. 9.12).
A combination of ball-in-cone and membrane valves
introduces the option to increase the opening or oper-
ating forces in a way that the sticking forces, which
influence the function of the valve, remain small in
comparison with the operating force (▶ Fig. 9.12, right).
This principle is established in the (hydrostatic) DUAL-
SWITCH valve27,28 and the MONOSTEP valve. In these
Fig. 9.13 The miniNAV concept in comparison with other
devices the ball-in-cone principle is inverted: the ball,
differential pressure valves.
which closes the valve at the valve seat, is not movable,
63
Shunt and Valve Technology
curve, which is the same for both adjustable and non-
9.8 Adjustable Differential
adjustable devices. However, this aspect alone does not
Pressure Valves
explain why the outcome of adjustable devices is not sig-
nificantly better. This can be explained by the limited
Adjustable DP valves represent the most popular and cur-
value of adjustable devices in accordance to their
rent type of valves. These valves allow the valve charac-
mechanical performance as a DP-valve. Adjustable devices
teristic to be adapted to the clinical situation of the
do not solve the problem. They only shift it.
patient.38 Nonadjustable DP valves are available with dif-
There are two reasons for adjusting the valve. The first
ferent opening characteristics. The opening pressures
reason is an unsatisfactory clinical outcome leading to
vary from manufacturer to manufacturer. Valves are
suspicion of underdrainage. In this case, the opening
offered with very low, low, medium, high, and very high
pressure of the valve is decreased. The other reason is the
opening pressures. Very low is equal to 2 to 4 cm H2O and
adjustment of the valve to a higher value to treat over-
very high is equal to 18 to 20 cm H2O. Depending on the
drainage. However, both actions are, at the same time,
experience, the surgeon chooses an opening pressure that
biased because of systematic drawbacks produced by the
he or she believes to be the best option for the patient.
change of the valve setting: increasing the opening pres-
This choice is not based on scientific clinical evidence,
sure leads to underdrainage in a patient in a horizontal
because it has not been shown that one opening pressure
position, while decreasing the opening pressure increases
offers a clinical outcome better than the other. As a gen-
the risk of overdrainage in an upright patient.
eral rule, it seems logical and acceptable that a high
The available adjustable DP valves offer the option of a
resistance (opening pressure) decreases the risk of over-
pressure setting between 0 and maximum 20 cm H2O. In
drainage and a low resistance decreases the risk of
cases of overdrainage, the valves are adjusted to 20 cm
underdrainage. In patients with an unsatisfactory clinical
H2O. For an average adult patient, this value is too low. As
outcome or complications, such as subdural effusion
the hydrostatic pressure within the silicone tube of a
(overdrainage) or remaining clinical symptoms (under-
shunted patient can easily be 40 cm H2O, the adjustment
drainage), the implanted DP valve is revised and changed
up to 20 cm H2O cannot counterbalance this value. This
to one with a lower or higher opening pressure. Alterna-
means that, even with the highest adjustment, the goal of
tively, based on the growing evidence for hydrostatic
physiological pressure values cannot be achieved. ICP
components, a siphon-compensating device is also
remains too negative at -20 cm H2O, so the subdural effu-
implanted (see Section 9.9).19,21,23-25,29,31,32,39-41
sion cannot be treated. At the same setting, the pressure
To avoid the additional revision, adjustable devices
situation in the horizontal position becomes too high. The
have been developed that allow noninvasive adjustment
normal range of ICP is about 10 to 15 cm H2O. With an
of the device. In cases of underdrainage, the opening
adjustment to 20 cm H2O, the ICP is too high, and the ICP
pressure can be decreased; in cases of overdrainage, the
grows to hydrocephalic values after longer periods in a
valve pressure is increased (▶ Fig. 9.14).
horizontal position. This is especially critical for patients
Adjustable devices are popular up to now although
with normal pressure hydrocephalus (NPH), because it is
their superiority versus nonadjustable DP valves has not
known that a low-pressure setting is beneficial for a good
been confirmed.42-44 This might be surprising because
clinical outcome.34,45 The advantage of a lower risk for
the avoidance of a further revision should improve the
severe overdrainage-related complications in the upright
survival of an implanted valve. One explanation might be
position is diminished by the drawback of underdrainage
the high influence of the infection rate on the survival
in the horizontal position (▶ Fig. 9.15).
a
b
c
d
Fig. 9.14 Principles for pressure adjustments in hydrocephalus shunts. (a) proGAV principle. (b) Sophysa principle. (c) CODMAN MEDOS
principle. (d) STRATA principle.
64
Shunt and Valve Technology
Fig. 9.15 High setting of an adjustable valve: there is still overdrainage in the upright position and underdrainage in the horizontal
position.
The opposite can occur in patients with unsatisfactory
the valve is the lowest if the active length of the spring is
clinical outcomes combined with suspected underdrain-
the longest. The long spring exerts a low force on the ball
age. In these patients, adjustment of very low opening
at the valve seat and the short spring exerts a high force
pressures dramatically increases the risk of overdrainage-
and consequently a high opening pressure of the valve. To
related complications when the patient is in an upright
decrease the risk of unintended readjustments, the non-
position (▶ Fig. 9.16).
relevant part of the spring is snapped elastically into a
There is no clinical evidence for the superiority of
groove in the outer housing (▶ Fig. 9.17).
adjustable valves, but there is mechanical evidence that
The adjustment of this device is performed by magnets
DP valves do not guarantee physiological pressure estab-
from the outside, which have to be placed in such a way
lishment in patients with NPH. However, this type of
that the rotor within the housing moves into the required
valve is far more expensive than nonadjustable devices,
position for the required opening pressure. The most
but they are well accepted and commonly used.41
important drawback of this device is the fact that it can be
The first adjustable valve, which was introduced into
easily readjusted by magnets routinely used in daily life.46
the market in the late 1980s, was the so-called Sophysa
The setting of the highest and lowest value is established
SU8. This valve offers eight pressure settings between 4
by turning the rotor in a range of about 90°. The position
and 20 cm H2O. The cylindrical flat housing incorporates
of the rotor can be determined from outside with the help
a rotor with two magnets, one at each end, and it is fixed
of a compass. The needle of the compass follows the posi-
on an axle in the middle of the housing. At one end of the
tion of the magnets inside the rotor. With the right orien-
rotor, an arc-shaped leaf spring is fixed on the rotor in
tation in parallel to the inlet connector of the implanted
such a way that, if the rotor is turning, then the spring
device, the position of the needle allows the reading of
changes the relevant length of its lever. Thus, the active
the pressure setting. Due to the fact that the rotor can be
length of the spring changes, defining the resulting open-
turned in an angle of about 90° only, there is a certain risk
ing pressure of the device. The pressure becomes higher
of a misreading. In cases of problems, the adjustment can
if the active length of the spring is short. The setting of
be controlled with radiography.47
Fig. 9.16 Adjustment to a low valve setting in patients with suspected underdrainage.
65
Shunt and Valve Technology
trolled magnetic fields placed above the device from out-
side. On one side, the spring lies on the ball at the valve
seat; on the other side the spring lies on the “stairs” of a
rotor, which can be turned through 360°. While turning,
the spring climbs (or goes down) the stairs and this
changes the opening pressure. To perform an adjustment,
the company Codman50 offers a special tool, which cre-
ates the magnetic fields above the device. The rotor incor-
porates eight small magnets, which are cylindrically
placed at the outer end of the rotor. The north-to-south
orientation of the magnets is in parallel to the rotor axis
and changes from neighbor to neighbor. The rotation of
the rotor during the adjustment process happens in steps
of 20°. The adjustment apparatus placed above the valve
first creates magnetic fields with a changing orientation
Fig. 9.17 (a and b) The first adjustable differential pressure
in a way that the valve is adjusted stepwise to the lowest
valve on the market, the Sophysa SU 8.
setting. This means that the spring lies on the lowest stair
of the rotor in this position, as the spring cannot jump to
the highest stair next to the lowest. From this point, the
To improve this device and avoid the risk of an
apparatus starts turning the rotor in the opposite direc-
unintended readjustment of the valve while it was
tion in steps of 20°. One step is equivalent to a pressure
implanted, the company Sophysa48 developed the Polaris
change of 1 cm H2O. The highest adjustment is 20 cm
valve. This device introduced the radial movement of the
H2O; the lowest is 3 cm H2O. Once the setting is achieved,
magnets within the rotor. Due to their orientation the
it cannot be controlled without radiography. This is a very
magnets are normally pulled toward the axle of the valve.
important disadvantage of this device (▶ Fig. 9.19).51
In this position the rotor cannot be turned around
A second important problem of the MEDOS HAKIM
because the movable parts snap into notches, which hin-
valve is the fact that it can be readjusted accidentally
ders the rotor turning around. To perform an adjustment,
whenever an external magnetic field acts on it. There are
magnets from outside, which are stronger than the mag-
several reports in the literature that confirm the rele-
nets within the valve, force the valve magnets to move
vance of this drawback.52-56 Serious complications are
toward the outer housing away from the axle in the mid-
reported especially in patients in whom the valves are
dle of the housing. In this position the rotor can move
unintentionally readjusted from the highest to the lowest
freely, and adjustment is easily possible (▶ Fig. 9.18).49
value. If the valve is adjusted to the highest setting (20 cm
The second adjustable valve brought into the market
H2O) and the rotor gets turned by 20° in the appropriate
was the CODMAN HAKIM valve, which is called the
direction, the setting switches down to 3 cm H2O, which
MEDOS CODMAN programmable valve by the manufac-
is the lowest setting. This is especially dangerous because
turer. A leaf spring exerts force on a ball into a valve seat,
the intended adjustment was an increase in opening
whereby the force of the spring can be adjusted by con-
pressure to the highest possible value.53
a
b
c
d
Fig. 9.18 Principle of the Polaris valve from Sophysa. (a) Rotor locked, no adjustment possible. (b) Rotor unlocked by external magnets,
adjustment possible. (c) Position of the unlocked rotor is changed, rotor still unlocked. (d) Rotor locked in the new position after the
readjustment.49 Printed with permission.
66
Shunt and Valve Technology
pulls the rotor up against the weaker spring force. In this
position, the rotor can be rotated following the rotation
of the outer magnet. The turning rotor changes the spring
force of the device that determines the opening pressure.
The valve is available with five different settings from 2
to 15 cm H2O (▶ Fig. 9.20).59,60
The setting of the valve can be determined without
radiography. Medtronic offers an electronic device with
magneto-sensitive components that show the position as
well as the pressure setting on a small screen. The valve is
affected by external magnetic fields that can be used to
Fig. 9.19 The principle of the adjustable MEDOS HAKIM valve
change the adjustment of the device.56,61-63 The valve is
(patent).51 Printed with permission.
available with or without the siphon control unit (see
Section 9.9).
The requirement that the adjustment must be con-
9.9 Hydrostatic Devices
firmed by radiography is especially important for the
pediatric population wherein X-rays generally should be
Problems with overdrainage became obvious very soon
avoided. For this reason, the manufacturer introduced a
after the first successful implantations of shunt systems
device that acoustically controls the correct setting of the
in the treatment of hydrocephalus. Since then, three dif-
valve. A microphone detects each single sound induced
ferent technologies have been developed and brought to
by the spring, as it climbs from one step to the next dur-
the market to improve clinical outcomes and to lower the
ing the adjustment procedure. If the device is not able to
rate of complications, especially those due to overdrain-
detect as many typical sounds as pressure steps that have
age. Currently, this type of complication is not seen as a
to be adjusted, the procedure must be repeated or the
systematical consequence of shunting but as an untypical
information is given that the adjustment was not success-
event depending on individual circumstances. For this
ful. Independent investigations of this device confirmed
reason, most hydrostatic devices are not regularly used in
the function in general; however, the function could not
combination with DP-valves but only in cases of compli-
be guaranteed in 100% of patients. Therefore, the device
cations due to overdrainage.
requires radiographical confirmation of the adjustment,
Hydrostatic devices should be defined as valves or
not just from a legal standpoint.57,58
valve components that aim to counterbalance or avoid
In the
1990s, PS-Medical
(Medtronic) introduced
overdrainage. The first device offering such an approach
another adjustable DP valve, the so-called STRATA valve.
was proposed by Portnoy et al who presented the so-
This valve is also readjusted by external magnets placed
called antisiphon device (ASD).19 The principle is smart
above the implant. The valve mechanism incorporates
and very effective: a thin membrane closes the distal
two different springs: one determines the opening pres-
pathway of the device, which has a circular outlet of
sure; the other keeps the rotor in the appropriate position
1 mm diameter, whereas the proximal pressure acts on
as long as there is no magnetic field around it, which
an area that is 18 times larger (▶ Fig. 9.21).
Fig. 9.20 The mechanism of the STRATA valve by Medtronic.59,60 Printed with permission.
67
Shunt and Valve Technology
Whether or not the membrane opens depends on the
proximal force acting against the distal force Fd. If Fp is
higher than Fd, then the membrane opens and allows the
drainage of CSF. As a result of this mechanism, the ICP
cannot become negative regardless of the position of the
patient. If the ICP becomes zero, then Fp becomes zero.
The negative pressure at the outlet does not influence the
ICP; the membrane continuously closes the device and
consequently the shunt. If the ICP increases due to further
production of CSF, then Fp starts to increase. If Fp becomes
higher than Fd, then the valve opens and allows the flow
of CSF. For the above example of a standing person with a
hydrostatic pressure of 40 cm H2O, the theoretical open-
ing pressure of the ASD would be 40 cm H2O divided by
18, which is 2.22 cm H2O. To achieve an ICP of 10 cm H2O,
the opening pressure of the additional DP valve should be
7.78 cm H2O. The ASD mechanism inverts the “siphon-
ing.” As pressure of the distal hanging tube becomes more
Fig. 9.21 Principle of an antisiphon device. The membrane
negative, the resulting opening pressure of the device
(yellow) closes the valve seat. The suction at the distal outlet
turns higher (▶ Fig. 9.22).19
(red) establishes a significantly lower force than the proximal
Even though the ASD has been successfully intro-
(ventricular) pressure acting on the large membrane (blue) to
duced into clinical practice, it does have one systemat-
open the device.
ical and very important drawback. The explanation
described above does not include the influence of sub-
cutaneous pressure. Several papers report problems of
underdrainage after the implantation of a valve with
ASD units.19,41,64-67
If the device is closed, then two different forces are act-
In addition to the hanging water column at the distal
ing on the membrane. Pressure is defined as force to area.
outlet of the device, the subcutaneous pressure controls the
Therefore, the forces acting on the membrane can be sep-
function. An implant is encapsulated by the surrounding
arately calculated for the distal and proximal part of the
device. The proximal force is determined by ICP and by
the size of the membrane, which is 18 times larger than
the part of the membrane covering the distal outlet. The
negative pressure resulting from the hanging water col-
umn within the tube distally to the valve, which causes
suction on the closing membrane, arises only when the
patient is upright. A typical value for an adult patient
could be -40 cm H2O. A typical ICP could be 10 cm H2O.
The forces determining the opening of the valve can be
calculated with the following formulas:
1
Fd ¼ pd
D2
ð17Þ
4
1
Fp ¼ pp
18
D2
ð18Þ
4
where Fd is the force induced by the distal water column;
Fig. 9.22 Opening and closing pressure (OP, CP) of a Hakim
Fp is the force established by the proximal water column
differential pressure valve and an anti-siphon-valve as depen-
or the proximal pressure at the closed membrane (includ-
dent on the negative distal pressure. ASD, antisiphon device;
ing the ICP); D is the cross section of the distal part of the
ASV, antisiphon valve; ASV-CP, antisiphon valve closing pres-
sure; ASV + HAKIM OP, combination of ASV and HAKIM opening
valve seat which is closed by the membrane; pd is the
pressure; ASV-OP, antisiphon valve opening pressure; HAKIM
proximal pressure at the closed valve seat; and pp is the
OP, HAKIM differential pressure valve opening pressure.
proximal pressure at the closed valve seat.
68
Shunt and Valve Technology
tissue, which results in an unpredictable force on the mem-
less constant. The Orbis Sigma valve (OSV) is a membrane
brane. Scars above the valve might even increase this factor.
valve that automatically decreases the opened area at the
If a patient is lying on the device, the pressure acting on the
valve seat as soon as the differential pressure acting on
membrane at the outer surface also increases unpredictably
the device increases. Normally in conventional DP valves,
(▶ Fig. 9.23). Therefore, ASDs can never establish ICP condi-
the increased intraventricular pressure results in an
tions that satisfactorily address the physiological require-
increased flow. In contrast to this, the OSV narrows its
ments.65 Nevertheless, different designs of this principle
pathways such that the increasing pressure does not
have been developed to improve the performance. Even
result in increased flow.
though the reasons for the unsatisfying clinical outcome
A sapphire plate with a hole in the middle is integrated
might be different, clinical data confirm that these devices
into a silicone membrane. The membrane is fixed at the
perform neither worse nor better than DP valves.2
outer housing of the device. A ruby pin is fixed in the
In addition to this important drawback, the function of
middle of the housing and closes the hole of the sapphire
devices with an antisiphon chamber—as used in the
plate. The device is closed as long as there is no differen-
DELTA68
and STRATA69-71
valves—depends greatly on
tial pressure between the inlet and the outlet (step 1). If
their precise placement. If the valve is implanted too
there is a positive pressure difference between the inlet
low, the resulting ICP becomes too negative leading to
and the outlet, then the membrane follows this pressure,
overdrainage symptoms; if it is implanted too high—for
opens the device, and allows a flow, which is equal to a
example, frontally next to the burr hole—the ICP might
normal average CSF production rate of 20 mL/h. Due to
stay significantly above physiological values.64 For lumbo-
the shape of the ruby pin, the opened area at the valve
peritoneal shunting, the mechanism cannot counter-
seat depends on the position of the membrane, which
balance overdrainage due to the location where the
again depends on the differential pressure. The pressure
device has to be placed.
becomes higher as the pin narrows the hole within the
The second hydrostatic principle was introduced in
membrane, so an increase in flow is hindered (step 2). At
1989 by Sainte-Rose et al,72 who proposed a valve that
a differential pressure of about 40 cm H2O, the membrane
controlled the flow throughout the shunt within the lim-
has reached the end of the pin, the hole becomes open
its of the production rate of CSF. The proposal was based
and the flow can rapidly increase (step 3) (▶ Fig. 9.24).
on the assumption that the production of CSF is more or
Similar to valves with ASD technology, clinical studies
have not been able to demonstrate a better or worse
clinical outcome for the OSV in comparison with DP
valves.2,73-75
There are several factors that might be
considered as a possible reason for this.
First, it is a questionable assumption that the CSF pro-
duction rate is constant. It is known that production
changes during life: children produce less CSF, whereas
adults produce more. Also, CSF production rate varies
from individual to individual and even intra-individually
during the day (▶ Fig. 9.25).76
Generally it is impossible to establish a flow control
within a shunt of hydrocephalic patients. All available
Fig. 9.23 Forces determining the function of antisiphon
devices. As long as the device is closed, no CSF is being drained.
The increasing ICP moves the plate (membrane) away from the
valve seat and hereby allows a flow throughout the device. The
opening of the device is nearly not affected by the strength of
the distal suction due to the small relevant area. The biggest
impact on the device is realized by the subcutaneous pressure
which is not predictable and consequently might lead to
unpredictable pressure condition within the shunt system.
Fig. 9.24 Principle of the Orbis Sigma valve (OSV).
69
Shunt and Valve Technology
Fig. 9.25 Cerebrospinal fluid production over 24 hours in a
shunted patient.
Fig. 9.27 The two principles of flow reduction. (a) Orbis Sigma
valve principle. (b) SIPHONGUARD principle.
valves are controlled by pressure. A flow control would
mean determining the actual production rate of CSF as
well as the actual absorption rate, calculating the balance,
or increased CSF production. In either case, the CSF drain-
and allowing drainage of the calculated flow. If there is
age is then limited by a narrow pathway in parallel to the
no absorption and the production rate is higher than (the
main valve seat leading to a significantly higher resist-
allowed) 20 mL/h, then the ICP will increase, resulting in
ance (▶ Fig. 9.27). The device does not react posture
underdrainage. In contrast, the valve leads to overdrain-
dependently, but instead the function depends on the dif-
age if the production rate is lower than 20 mL/h. If more
ferential pressure. When the hydrostatic pressure is at its
is drained than is produced, the ICP decreases and the
highest, and whenever the flow throughout the valve
patient may experience overdrainage (▶ Fig. 9.26). There-
becomes higher than the average CSF production, the
fore, the so-called flow-reducing devices might lead to
flow reduction is activated in the upright position. When
overdrainage as well as underdrainage. Reports in the lit-
there are high production rates (e.g., during deep rapid
erature regarding the clinical performance of these
eye movement sleep at night) the resistance might be too
devices confirm this hypothesis.34,73,77-81
high and lead to ICP crises.
Another principle of a so-called flow-reducing device
Generally it can be stated for flow-reducing devices
has been developed by Codman. The SIPHONGUARD hin-
that, in an upright position, when the hydrostatic pres-
ders the flow throughout the valve by pressing a ball
sure is high, a corresponding higher flow is effectively
against the force of a spring toward the valve seat
prevented so that overdrainage is delayed, at least
depending on the flow to be drained. If the flow exceeds
momentarily. Crucial shortcomings are that they are
a critical value, the ball closes the main pathway of the
more liable to congestion (compared with standard DP
valve. This might happen as a result of postural changes
valves) and mechanical problems, such as blockade,
because of the necessarily minuscule flow-through diam-
eters. In case of the OSV, the minimal flow-through cross
section is a very thin ring with an average radius of
approximately 690 μm and a thickness of only 15 μm. CSF
contains leukocytes, of which some types, the monocytes,
can achieve diameters of up to 20 μm. In general, the high
danger of clumping of biomaterial, cells, and proteins
contained in the CSF is the reason for one of the most
serious and frequent valve malfunctions, namely valve
occlusion. For the SIPHONGUARD, the helix-like, high-
resistance channel has a length of approximately 15 cm
and the diameter of the capillary tube is approximately
550 μm; there is also a lack of “security level” for very
high pressures, and this can be highly problematic.
Dangerous high-pressure crises such as A-waves or
Fig. 9.26 Theoretical concept of flow control within shunts.
B-waves—the latter are suspected to eventually lead to
70
Shunt and Valve Technology
normal pressure hydrocephalus82—cannot be absorbed or
the DSV switches between two different opening pres-
are only very slowly absorbed.
sures, one for the horizontal and one for the upright posi-
The final group of hydrostatic devices introduces grav-
tion, and, accordingly, it is known as a switcher-type
ity control of the posture-dependent physics within a
gravitational valve.
shunt. The principle was a proposal of Salomón Hakim,83
The titanium housing of the DSV incorporates two
as published in his 1974 patent. The weight of metallic
membranes with integrated titanium plates. A ball in
balls counterbalances the posture-dependent hydrostatic
the center of the valve is fixed in the housing. The
pressure. It is difficult to understand that this principle
membranes are pressed against this ball by two springs
has been favored for LP drainage only, and not for VP
of different strengths to produce the different posture-
drainage. However, Hakim developed different devices
dependent opening performance of the device. The
that use the weight of a metal ball to systematically
valve is designed to be implanted in the chest of adult
address the hydrostatic pressure within the shunt system
patients. It is important to implant the valve in parallel
when the patient is upright.
with the body axis to guarantee the intended function.
The dual-switch valve (DSV) was introduced in 1996
Implantation in a sloping position might lead to
and comprises two valve chambers in parallel within one
overdrainage-related complications (▶ Fig. 9.28).
housing. One chamber controls the pressure in the hori-
The DSV was the first gravity-controlled valve to be used
zontal position, while the other one controls it in the
clinically in patients with NPH. Several groups have
upright position.27 In the horizontal position, the opening
reported on a superior clinical outcome, but the results have
pressure of conventional DP valves is sufficient, while
been empirical and without clear evidence.29,31,33-37,84,85
high pressure is required for the upright position. This
The performance of the valve has not been investigated
high pressure is equal to the distance between the fora-
in an independent prospective randomized trial, and this
men of Monro and the diaphragm. The appropriate cham-
is also the case for most of the other available valves.
ber is activated by the position of a tantalum ball, which
However, clinical data confirm the concept of the valve
acts as a switch. In the horizontal position, the distal
and show that the overdrainage-related complication rate
pathway of the low-pressure chamber is open; in the
seems to be significantly lower than for DPVs.35,36
upright position, the tantalum ball closes the pathway at
One of the very first cases where a DSV was implanted
an angle of about 60° to 70°. In the position below this
was in a 40-year-old woman with NPH. A DSV with an
closing angle, the tantalum ball narrows the area at the
opening pressure of 13 cm H2O for the recumbent posi-
seat of the switch and the flow is reduced. Nevertheless,
tion and 40 cm H2O for the upright position was chosen
Fig. 9.28 The working principle of the DUALSWITCH valve: (a) the device is closed as the ICP is lower than the opening pressure of the
valve for the lying position. (b) The ICP is increased, the horizontal part of the valve opens and hereby keeps the the ICP within the
required limits. (c) In the upright position the tantalum switch (green) closes the pathway of the low-pressure-chamber, the valve
chamber for the horizontal position of the patient is closed. The opening pressure for the upright position is not reached yet. The valve
does not allow any drainage. (d) the pressure has reached the opening force of the high-pressure chamber. The valve opens and allows a
CSF flow which avoids a further increase of the ICP.
71
Shunt and Valve Technology
Fig. 9.29 Incorrect placement of the DUALSWITCH valve and the clinical consequence.
for this VP shunt. The valve was placed subcutaneously in
(▶Fig. 9.30). Although such case reports do not prove the
the upper part of the thoracic region. After 1 month, the
concept in general, they confirm the theory of gravita-
patient developed severe headache and deterioration of
tional valves. Cases such as this were the breakthrough
gait. Computed tomography revealed bilateral subdural
for gravitational valves being considered a serious tech-
hygromas, which required treatment (▶ Fig. 9.29), and so
nology for VP shunting of patients with hydrocephalus.86
the shunt was ligated. It was recognized that because of
Although all clinical papers reporting use of the DSV
the barrel-shaped thorax and the implantation of the
have presented good to very good clinical results
device in the subclavicular groove, the valve was in a
for shunted patients, the valve has never become very
position that was too inclined and therefore the high-
popular. One reason for this is the size of the valve and,
pressure chamber could not be activated (▶ Fig. 9.30).
consequently, the required placement of the valve in the
After the hematoma had resolved and the hydrocephalic
middle or lower thoracic region (e.g., on the sternum).
status of the patient continued to require a shunt, a valve
This is possible in adult patients only, and does not
of the same type (16/50) was implanted subcutaneously
correspond with the favored neurosurgical standard
at the lower thorax and positioned in a strict vertical
procedure. The second reason for the limited clinical use
position while the patient was upright. The improvement
is the growing popularity of adjustable devices.
in the patient was clinically significant, and there were
Another gravitational valve principle relies on a sugges-
no further complications. The ventricles became signifi-
tion by Hakim. He developed a valve system with two
cantly smaller, without any signs of subdural effusion
valves in series: one conventional ball-in-cone valve, and
Fig. 9.30 Correct placement of the dual-switch valve: small ventricles without overdrainage.
72
Shunt and Valve Technology
a second valve not controlled by a spring but by the effect
of gravity on three stainless steel balls. The housing of the
valve is U-shaped, and the inlet and outlet connectors are
arranged at a right angle to the valve axis. The horizontal-
vertical valve was designed for use in LP shunts only, as
here it was obvious that the spinal CSF column required a
higher opening pressure when the patient was upright.
Unfortunately, the vertical placement of the valve is cru-
cial for the correct function of the valve, but this is often
difficult to achieve in lumbo-peritoneal shunts.
In contrast to the switcher type this gravitational valve
principle is called the analogous type because at any
given angle the hydrostatic pressure is counterbalanced
by the weight of the balls accordingly. The relevant force
increases in an analogous manner to the sine function of
the angle.
Based on clinical data regarding the use of the DSV and
the idea of Hakim, the SHUNTASSISTANT (Miethke, Pots-
dam, Germany) as well as the gravity-compensating
accessory (GCA; Cordis, Biot, Valbonne, France) have been
Fig. 9.31 Function of the analogous type of gravitational valve
developed to introduce the option of a gravitational func-
in principle.
tion in combination with adjustable valves. In contrast to
Hakim’s horizontal vertical valve and the GCA, the SHUN-
TASSISTANT has no stainless steel balls to establish the
its maximum. The sine of 90° is equal to 1. For any posi-
posture-dependent gravitational function. The opening
tion of the device, the resulting opening pressure can be
pressure of the analogous type of valve is determined by
calculated by
the weight of the ball(s) and the position of the device.
The weight of the balls depends on the material used or,
pð Þ ¼ pmax sinð Þ
ð19Þ
better still, its specific weight. In combination with the
requirement of a slim and small housing, which, particu-
larly in children, can easily be implanted parallel to the
The maximum opening pressure of these devices is
body axis under the thin skin retroauricularly, the volume
chosen depending on the height of the patient. The taller
of the gravitational body should be as small as possible.
a person is, the higher the required gravitational compen-
Therefore, the SHUNTASSISTANT contains a tantalum ball
sation. Based on clinical experience with gravitational
for gravitational control. Tantalum has a specific weight
valves, the recommendation for choosing the appropriate
of
16.6 kg/L in contrast to stainless steel with about
valve can be concluded as follows:
7.8 kg/L. The small tantalum ball consequently allows a
● Patient shorter than 1.6 m (< 5' 3"): pmax = 15 cm H2O
small housing with a diameter of 4.6 mm and a length of
● Patient height 1.6 to 1.8 m (5' 3" to 5' 11"): pmax = 20 cm
23.7 mm, including the connectors. The pediatric version
H2O
with a lower gravitational force has a diameter of 4 mm,
● Patient taller than 1.8 m (> 5' 11"): pmax = 25 cm H2O.
which is nearly as thin as standard silicone tubing
(▶Fig. 9.31).
Until now, there has been no clinical evidence for these
When the patient is in a horizontal position, the weight
recommendations. The optimal opening pressure for each
of the tantalum ball does not add further resistance to
individual patient might be higher or lower than these
the opening pressure of the ball-in-cone valve, which is
recommendations.
only dependent on the force of the spring; when the
As mentioned above, correct vertical placement is very
patient is in an upright position, the weight of the gravi-
important for the analogous type of gravitational devices.
tational ball counterbalances the hydrostatic pressure in
Park et al87 reported a clear correlation between the valve
the shunt system. The mechanism of the SHUNTASSIST-
and body position and emphasized that anterior inclina-
ANT includes two different balls: one (smaller) sapphire
tion of the valve by more than 20° relative to the vertical
ball, which acts as the valve ball on the seat, and a (larger)
increases the likelihood of underdrainage.
gravitational tantalum ball. The weight of the tantalum
The SHUNTASSISTANT and the GCA are designed to be
ball acts against the sapphire ball, thereby defining the
implanted together with a DP valve.88 The two devices
opening performance of the device. The force of gravity
are implanted in series. Normally the SHUNTASSISTANT
can be calculated with the sine of the angle. When the
is implanted distally from the DP valve. The physics of
device is in a vertical position, the gravitational force is at
such a shunt is illustrated in ▶ Fig. 9.32.
73
Shunt and Valve Technology
Fig. 9.33 Comparison of the (a) switch-type (dual-switch valve)
and the (b) analogous type of gravitational valves (gravity-
assisted valve, GAV).
The switched-type valve, the DSV, offers large mem-
branes with strong spring forces and, consequently, a
very reliable and robust type of valve. The GAV with a
conventional ball-in-cone mechanism is theoretically less
robust; however, the relevance of this aspect has never
been proven clinically or in the laboratory. The benefit of
the GAV, the analogous valve type, can be seen in the pre-
Fig. 9.32 Function of a differential pressure valve in combina-
cise counterbalance of the hydrostatic pressure for each
tion with a SHUNTASSISTANT.
and any valve inclination. This can also be seen as a draw-
back because accuracy is required in terms of placement
of the valve and sufficient opening pressure (▶ Fig. 9.33).
Decisive for the function of VP shunts is the zero level
The risk of overdrainage for the DSV in a semireclined
within the peritoneal cavity. This level is the referential
position of around 45° to 60°, which has been shown to
point, which is transmitted to the ventricles by the valves
be low and hardly relevant, is accompanied by the theo-
integrated into the shunt. The gravitational part of the
retical benefit of a “flow reserve” at oblique angles. If the
system is posture dependent (changing between 0 and
opening pressure for the upright position is chosen high,
maximum opening pressure), whereas the DP valve does
then the GAV will underdrain at any angle, whereas the
not change depending on posture. If an adjustable DP-
DSV still acts like a DP valve allowing a substantial higher
valve is chosen, then the performance of this part of the
flow than the GAV. However, the flow throughout the
shunt can be adapted to the individual requirements. Any
DSV at an angle is in comparison to normal DP valves
adjustment of the DP valve always has the same conse-
reduced by the switching ball which significantly narrows
quence for the vertical and the horizontal position of the
the pathway, hereby on the other hand abating the risk of
patient, although the adjustment is necessary and benefi-
overdrainage. Regardless, the GAV (▶ Fig. 9.34) is an alter-
cial only for one of both options. For the other option, it
native to the DSV that can be conventionally integrated
might even worsen the condition. The opening pressures
into a shunt and accordingly be implanted retroauricu-
of both units must be added together to calculate the
larly like any other valve. The only important aspect that
overall characteristic of this combination. The sum repre-
must be taken into account is its required position paral-
sents the opening performance of the system. Thus this
lel to the body axis.
combination does not offer the frequently required
Critical feedback about gravitational valves is given in
option of changing just one part of the system; for exam-
the context of incorrect positioning of the device,87 under-
ple, adjusting the opening pressure for the upright posi-
drainage in bedridden patients,89 or the blockage of the
tion only, without changing the setting for the horizontal
device. Such complications are well known for any kind of
position.
shunt and not specifically worse for gravitational valves.
The DSV is implanted in the middle or lower thoracic
For these reasons, adjustable DP valves have been devel-
region (e.g., on the sternum), which limits its clinical use.
oped. The introduction of adjustability in gravitational
The gravity-assisted valve (GAV) is an alternative to the
shunts promises further potential for clinically relevant
DSV without the limitations due to size. Both valves reli-
treatment options, to further minimize complications
ably counterbalance the hydrostatic pressure changes due
such as underdrainage or overdrainage. Gravitational
to changes in the posture of the patient. Whether one of
shunts offer two options for adjustment: the opening
the two different principles is superior to the other has
pressure for the horizontal position, and, independently,
not been determined based on clinical performance.
the performance of the valve in the upright position.
74
Shunt and Valve Technology
a
Fig. 9.35 The mechanism and photograph of the adjustment
unit of the proGAV valve (on the right is the photograph of both
the adjustment and the gravitational unit).
able and precise. The valve should be implantable in neo-
nates and adults; the adjustment should allow the lowest
possible and highest reasonable pressure (▶ Fig. 9.35).
A sapphire ball (yellow) is pressed by a spring force (red)
b
into a valve seat. The relevant spring force is defined by
the adjustment of a rotor (blue), in which two magnets are
Fig. 9.34 (a) Photograph and (b) X-ray of the GAV.
integrated. The spring itself is a thin titanium wire with a
thickness of 0.1 mm. This wire is welded with a titanium
axis in a right angle. The axle is placed in two small drilling
The proGAV was introduced in 2004.90 The proGAV is a
holes, which allow the free rotation of the axis without
gravitational valve that offers the adjustment of the DP
substantial friction. At an angle of about 50° to the tita-
valve unit, which controls the pressure when the patient is
nium wire, a stiff titanium arrow-shaped rod is welded to
in a horizontal position. The development of the valve took
the axis, and this transfers the spring force to the valve ball
into account the criticisms regarding available adjustable
(yellow). The straight wire is the most reliable and easiest
valves such as the CODMAN HAKIM valve, the STRATA
form of a spring. The adjustment of the valve takes place
valve, and the Sophy SU8 valve. The valve should contain a
by turning the rotor (blue). This happens by the external
mechanism that avoids unintended readjustments. The
placement of magnets, which determine the position of
pressure setting should be detectable without X-ray, as a
the rotor (the magnets inside the rotor; ▶ Fig. 9.36).
specifically important requirement for neonates and chil-
To avoid unintended readjustment of the valve setting
dren. The adjustment should be as easy as possible and
due to magnetic fields during activities of daily life or
the reading of the adjusted pressure range should be reli-
during magnetic resonance imaging scans, the rotor is
Fig. 9.36 Different adjustments of the proGAV valve obtained by turning the rotor.
75
Shunt and Valve Technology
Fig. 9.37 Function of the proGAV valve brake to avoid unintended readjustment and the method to release it during adjustment
operations. The red arrow shows the part of the titanium housing, where the rotor is elastically pressed against the not movable wall of
valve. The friction between the rotor and the housing hinder any movement of the rotor (left). If the adjustment tool is placed right
above the valve and an appropriate (small) force is established on the titanium membrane, the friction annuled (red circles) and the
rotor is moving in the position directed by the adjustment tool.
blocked in the housing as long as there is no force acting
requires precise placement of the instrument above the
from the outside on the membrane of the titanium hous-
valve. If the instrument is placed more than 3 mm off cen-
ing of the valve. The rotor can only follow the force of the
ter from the valve, then the possible mistake in the read-
external magnetic field produced by the adjustment
ing can exceed 3 cm H2O. Another verification tool uses a
instruments if friction between the rotor and the housing
disk with magnets floating inside a round housing with
is removed. The externally applied force on the mem-
two glass windows. By passing along the area of the skin,
brane moves the axle of the valve, which moves up
where the valve is expected to be, the magnets of the
the rotor. The rotor no longer touches the housing of the
instrument and the valve magnets interfere with each
device, the friction is abolished, and it moves into the
other. By moving the housing above the valve in a circular
position determined by the magnetic field of the adjust-
motion, the corresponding magnets catch each other, the
ing instrument. The mechanism of the brake has been
rotating disk is hindered from turning, and the number on
confirmed to reliably avoid unintended readjustments
the disk in the direction of the inlet connector of the valve
due to magnetic fields up to 3 T (▶ Fig. 9.37).61
gives the adjusted pressure level. If needed, the adjust-
Verification of the valve setting is easily possible with-
ment can be determined by radiography also (▶ Fig. 9.38).
out radiography. The verification instrument
(e.g., the
Since 2004, which is when the valve was first intro-
“proGAV Verification Tool”) is placed immediately above
duced to the market, several papers have confirmed
the implanted valve. By pushing a button, the brake inside
the positive clinical results for gravitational shunts. The
the instrument is released and the position of the instru-
option of postoperative noninvasive readjustment of the
ment scale follows the position of the magnets of the fixed
valve seemingly improves the treatment options in com-
rotor in the valve. Reliable reading of the adjustment
parison with nonadjustable gravitational valves.40,61,91-95
Fig. 9.38 X-ray determination of the proGAV setting.
76
Shunt and Valve Technology
device. Within the outer ASD membrane, a cylindrical
9.9.1 Adjustable Hydrostatic
body with an inner thread is integrated that is moved by
Devices
turning a rotor with magnets. Turning the rotor (red) in
Three different types of hydrostatic devices are currently
one direction decreases and turning in the other direction
increases the operating pressure of the device by moving
in clinical use, namely valves with a so-called ASD mecha-
the cage with an integrated membrane up and down.
nism, flow-reducing devices, and gravitational valves. In
terms of adjustment, the most complicated device seems
Unfortunately, such a device is not currently available.
to be the ASD. A review of the literature in medical and
Flow-reducing devices represent the second hydro-
technical journals and in patent databases does not reveal
static concept. Considering the idea of a flow control of
about 20 mL/h, the requirement of an adjustment option
any reason for this. The well-known STRATA valve, which
incorporates an ASD chamber, allows adjustment of the DP
for such a device appears logical. The flow control is
valve unit. However, the adjustment of this valve part does
achieved by a narrow pathway at the valve seat that
not address the following: underdrainage or overdrainage
becomes even narrower as soon as the differential pres-
in patients in which there is not perfect placement of the
sure acting on the device increases. Changes in viscosity
of the CSF, cell particles within the CSF, or variations of
valve (too high or too low); increased subcutaneous pres-
sure or performance changes at the ASD membrane as a
average production rate in an individual, or even a chang-
result of tissue or body reactions; and changes in material
ing production rate during lifetime, might require an
behavior or mechanical performance. The option of func-
adaptation to the changed situation to avoid a revision
tional adjustment of ASDs could allow noninvasive inter-
caused by underdrainage or overdrainage. Although a
concept for an adjustable flow-reducing valve was
vention in patients with mismanaged CSF drainage, for
example, by influencing the characteristic of the thin
patented in 1985 (▶ Fig. 9.40), it has never been intro-
membrane of the ASD by adjusting the elasticity of the
duced into clinical practice.96
membrane, which could increase or decrease the point of
The only adjustable hydrostatic valve that has been
closing or opening. ▶ Fig. 9.39 offers a proposal for such a
introduced into routine clinical practice is the adjustable
Fig. 9.39 Concept of an adjustable antisiphon device. (a)
Fig. 9.40 (a—e) Concept of an adjustable flow-reducing device.96
Normal adjustment. (b) Adjustment to low setting. (c)
Printed with permission.
Adjustment to high setting.
77
Shunt and Valve Technology
Fig. 9.41 Principle of the adjustable gravitational valve proSA.
gravitational valve proSA.97 The basic principle of this
A sapphire ball ensures the precise closure of the valve so
device is the adjustment of a spring force, which is acting
that any reflux is effectively prevented. The opening pres-
against the gravitational force. The weight of a metallic
sure of the valve is determined by both a bar spring and a
ball (red) acts on a second (sapphire) ball (yellow) that
heavy tantalum weight. The bar spring and the weight
closes the outlet of a tube filled with water (▶ Fig. 9.41).
are attached to a cantilever, which is freely able to rotate
The weight of the ball counterbalances the hydrostatic
about a fixed axis and keeps the sapphire ball in its posi-
pressure within the tube. If the hydrostatic pressure
tion. The tension of the spring, and thus the opening
exceeds the pressure of the ball against the valve seat,
pressure, can be adjusted by turning an eccentric rotor; in
then the valve opens and allows flow. The flow stops at
other words, a cam. An increase in the tension of the
the equilibrium between the ball pressure and the water
spring results in a reduction of the effective weight of the
pressure (1). If there is a leaf spring fixed at a joint on one
tantalum piece (▶ Fig. 9.42).
side, which allows the turning of the spring around the
▶ Fig. 9.43 depicts the angle dependency of the con-
joint without friction and which is placed between the
struction. When the patient is in the supine position
two balls at the valve seat without adding any force to the
(α = 0°), the weight pulls down in the direction of the
mechanism, then the hydrostatic balance is not affected,
cantilever so that no effective force acts laterally on
and the ball compensates the same hydrostatic pressure
the ball. In this state the valve is always open. When
without the spring. The weight of the spring can be
the patient is in the upright position, the weight acts
neglected (2, 3). If the spring force is carefully adjusted in
perpendicular to the cantilever so that the opening
such a way that it is at work against the gravitational
pressure reaches its maximum. The maximum value
force of the metallic ball, then the compensated hydro-
can be modified between 0 and 40 cm H2O by adapting
static pressure is thereby decreased because the weight is
the tension of the spring.
now partially counterbalanced by the adjusted spring force.
Strong magnets are integrated into the central rotor
In case the adjustment of the spring force becomes equal
so that it can easily be adjusted in vivo in a post-
to or higher than the gravitational force, the weight of the
operative and noninvasive manner. Both edges of the
ball is completely compensated, and the water column
rotor rest firmly on a circular step so that unintended
within the tubing sinks to the level of the valve seat (4).
rotation is securely blocked by friction. This robust
The hydrostatic compensation of the metallic ball func-
brake can be disengaged by depressing the bottom
tions only in the vertical position. At an angle, the force
operating against the valve seat is calculated by the sine
of the angle multiplied by the maximum force of the ball.
The adjustable spring force is not posture dependent.
Horizontally, the ball is pushed away, even at the lowest
adjustment. With adjustment of the spring force to high,
the hydrostatic pressure might already be completely
compensated at an angle of 45° or even higher. Conse-
quently, the adjustment of the spring force does not only
allow the adjustment of the hydrostatic pressure to be
compensated, but it also allows adjustment of the angle
at which the gravitational mechanism is activated.
The real valve, how it is actualized, consists of a robust
titanium housing that safely protects the inner mecha-
nism from all environmental conditions, especially ten-
sion and pressure during physical (muscular) exertion.
This ensures the correct function of the sensitive mecha-
nism and also enhances stability against leakage or break-
Fig. 9.42 Front view of the proSA.
age. The proximal end contains a ball-in-cone valve.
78
Shunt and Valve Technology
Fig. 9.45 Sterile adjustment and verification tool for use during
surgery.
Germany).98 The small and simple device is placed on the
housing of the valve. Due to two integrated magnets,
Fig. 9.43 Angle dependency of the opening pressure of the
CHECK-MATE moves into a position on the valve to be
proSA valve.
adjusted; this position is determined by the magnets
inside the valve and CHECK-MATE. Possible adjustment
numbers are written by a laser in a circle on the housing
surface of the proSA with the adjustment tool so that
of the device. The number closest to the inlet connector
the base is slightly deformed, thus raising the rotor
discloses the actual setting of the valve. The setting can be
from the step (▶ Fig. 9.44).
changed with the same instrument by turning the device
For the adjustment and its verification, respective tools
over the valve bringing the required number to the inlet
are provided that are similar to the instruments of the
connector and pressing it down and decoupling the brake
proGAV. Like the rotor, the tools contain strong permanent
inside the valve. The rotor is released and moves into the
magnets. This means no electric energy; in other words,
desired position. The success of this maneuver can easily
no batteries or radiography are required for adjustment or
be tested afterward (▶ Fig. 9.45).
verification. Nevertheless, the verification can also be per-
For a gravitational device, the opening pressure is vari-
formed with radiography if necessary for any reason. A
able “by definition” because it changes with the posture
method to adjust the valve under sterile conditions during
of the patient. So, for instance, “adjustability” means the
surgery is possible with a simple device called CHECK-
possibility to select the maximal opening pressure in an
MATE (e.g., the proGAV CHECK-MATE Miethke, Potsdam,
upright position without influencing the pressure level of
the supine position. If the weight is completely neutral-
ized, then the opening pressure becomes zero. In this
way, the tension of the bar spring controls a “threshold”;
in other words, a certain inclination angle beyond which
the tantalum weight comes into operation. After crossing
the threshold, the force applied onto the ball behaves in
the well-known sinusoidal manner, but it is always
reduced by a fixed amount generated by the tension of
the spring (▶ Fig. 9.46).
If the proSA is adjusted to 10 cm H2O and has a maxi-
mum opening pressure in the vertical position, then the
gravitational mechanism is not activated any earlier than
at an angle of 45°. The benefit of this might be the oppor-
tunity to diminish the negative result of a nonoptimal
positioning of the valve in an oblique position. The
retarded activation of the gravitational mechanism
decreases the risk of underdrainage.
The inclination angle determining the opening pressure
of the proSA is defined as the angle between the flow
Fig. 9.44 Principle of the brake within the proGAV and proSA.
direction of the valve and its projection onto the ground
79
Shunt and Valve Technology
Fig. 9.48 X-ray of a patient with proGAV and proSA valves.
Fig. 9.46 Calculated (ideal) pressure-angle dependency for
different pressure settings.
only. If the proSA is implanted together with an adjusta-
ble DP valve (proGAV, Medos Hakim) and Strata (without
plan. The proSA has a twofold rotational symmetry
Siphon-Control Unit, Polaris), this type of shunt presents
around the axis of its flow direction: twisting it around
any adjustment which might be required to achieve an
180° makes it look (almost) identical. This implies that,
optimum setting. When both valves are adjusted to the
for every angle, two nonequivalent positions are possible.
lowest adjustment (0 cm H2O), the valve operates like a
In general, for any gravitational device it is essential that
tube without a valve; when both valves are adjusted to
it works independent of this orientation. In other words,
the maximum adjustment (20 cm H2O for the DP valve,
there is no functional difference if the patient straightens
and 40 cm H2O for the proSA), the valve has a resulting
up out of the lateral or out of the dorsal position. The
opening pressure in the horizontal position of 20 cm H2O,
opening characteristic is dependent on the adjustment as
and 60 cm H2O (20 + 40 cm H2O) in the upright position,
well as the angle between the valve axis along the con-
which is nearly closed. In between these settings, the
nectors and the horizontal position (▶ Fig. 9.47).
valve can be independently adjusted to the optimum
Until now, no clinical data have been published about
value for the horizontal and upright positions. This
the proSA. The option of the adjustment of a DP valve has
expensive solution offers a unique individual adjustment,
added the option of adjustment of the gravitational part
especially for complex cases (▶ Fig. 9.48).
90°
Feff = F0•sin(α)
90°
Feff = F0•sin(α)
45°
45°
Feff
Feff
F0
F
α
0
α
0°
α
0°
α
a
b
Fig. 9.47 Inclination of the proSA with different orientations. (a) Frontal view. (b) Lateral view.
80
Shunt and Valve Technology
preventing post-shunt subdural hematomas. J Neurosurg 1973; 38:
The combination of a nonadjustable DP valve with an
729-738
adjustable gravitational valve is chosen more often than a
[20]
Di Rocco C, Marchese E, Velardi F. A survey of the first complication of
combination of two adjustable valves. The miniNAV is the
newly implanted CSF shunt devices for the treatment of nontumoral
smallest available DP valve, which is slightly larger than
hydrocephalus. Cooperative survey of the
1991-1992 Education
the tube itself. In particular, it is used as an option in
Committee of the ISPN. Childs Nerv Syst 1994; 10: 321-327
[21]
Gruber R, Jenny P, Herzog B. Experiences with the anti-siphon device
combination with the proSA. It is offered with the lowest
(ASD) in shunt therapy of pediatric hydrocephalus. J Neurosurg 1984;
possible opening pressure of DP valves close to zero.
61: 156-162
[22]
Faulhauer K, Schmitz P. Overdrainage phenomena in shunt treated
hydrocephalus. Acta Neurochir (Wien) 1978; 45: 89-101
[23]
Aschoff A, Kremer P, Hashemi B, Kunze S. The scientific history of
References
hydrocephalus and its treatment. Neurosurg Rev 1999; 22: 67-93,
[1]
Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
discussion 94-95
lus Study: randomized comparison of low- and medium-pressure
[24]
Aschoff A, Kremer P, Hashemi B, Benesch C. Technical design of 130
shunts. J Neurosurg 1998; 88: 490-495
hydrocephalus valves. An overview on historical, available, and pro-
[2]
Drake JM, Kestle JR, Milner R et al. Randomized trial of cerebrospinal
totype valves. Childs Nerv Syst 1996; 12: 474-504
fluid shunt valve design in pediatric hydrocephalus. Neurosurgery
[25]
Aschoff A, Kremer P, Benesch C, Fruh K, Klank A, Kunze S. Overdrain-
1998; 43: 294-303, discussion 303-305
age and shunt technology. A critical comparison of programmable,
[3]
Pollack IF, Albright AL, Adelson PD Hakim-Medos Investigator Group.
hydrostatic and variable-resistance valves and flow-reducing devices.
A randomized, controlled study of a programmable shunt valve
Childs Nerv Syst 1995; 11: 193-202
versus a conventional valve for patients with hydrocephalus.
[26]
Czosnyka M, Czosnyka Z, Whitehouse H, Pickard JD. Hydrodynamic
Neurosurgery 1999; 45: 1399-1408, discussion 1408-1411
properties of hydrocephalus shunts: United Kingdom Shunt Evalua-
[4]
Lemcke J, Meier U, Müller C et al. Is it possible to minimize overdrain-
tion Laboratory. J Neurol Neurosurg Psychiatry 1997; 62: 43-50
age complications with gravitational units in patients with idiopathic
[27]
Miethke C, Affeld K. A new valve for the treatment of hydrocephalus.
normal pressure hydrocephalus? Protocol of the randomized con-
Biomed Tech (Berl) 1994; 39: 181-187
trolled SVASONA Trial
(ISRCTN51046698). Acta Neurochir Suppl
[28]
Sprung C, Miethke C, Trost HA, Lanksch WR, Stolke D. The dual-
(Wien) 2010; 106: 113-115
switch valve. A new hydrostatic valve for the treatment of hydro-
[5]
Lemcke J, Meier U, Müller C et al. On the method of a randomised
cephalus. Childs Nerv Syst 1996; 12: 573-581
comparison of programmable valves with and without gravitational
[29]
Kiefer M, Eymann R, Strowitzki M, Steudel W-I. Gravitational shunts
units: the SVASONA study. Acta Neurochir Suppl (Wien) 2012; 114:
in longstanding overt ventriculomegaly in adults. Neurosurgery
243-246
2005; 57: 109-119
[6]
Lemcke J, Meier U, Müller C et al. Safety and efficacy of gravitational
[30]
Kiefer M, Eymann R. Gravitational shunt complications after a five-
shunt valves in patients with idiopathic normal pressure hydroceph-
year follow-up. Acta Neurochir Suppl (Wien) 2010; 106: 107-112
alus: a pragmatic, randomised, open label, multicentre trial (SVA-
[31]
Hertel F, Züchner M, Decker C, Schill S, Bosniak I, Bettag M. The
SONA). J Neurol Neurosurg Psychiatry 2013; 84: 850-857
Miethke dual switch valve: experience in 169 adult patients with dif-
[7]
Sgouros P, Kombogiorgas D. Cerebrospinal fluid shunts. In: Mallucci
ferent kinds of hydrocephalus: an open field study. Minim Invasive
C, Sgouros P, eds. Cerebrospinal Fluid Disorders. New York, NY:
Neurosurg 2008; 51: 147-153
Informa Healthcare USA, Inc.; 2010, pp. 438-453
[32]
Sprung C, Miethke C, Schlosser HG, Brock M. The enigma of under-
[8]
Grote W. Neurochirurgie. Stuttgart: Thieme; 1986
drainage in shunting with hydrostatic valves and possible solutions.
[9]
Merritt & Fremont-Smith. The Cerebrospinal Fluid. Philadelphia: WB
Acta Neurochir Suppl (Wien) 2005; 95: 229-235
Saunders; 1937
[33]
Kiefer M, Meier U, Eymann R. Gravitational valves: relevant differ-
[10]
Aschoff A. In-vitro-Testung von Hydrocephalus-Ventilen. Heidelberg:
ences with different technical solutions to counteract hydrostatic
Habilitationsschrift; 1994
pressure. Acta Neurochir Suppl (Wien) 2006; 96: 343-347
[11]
Ignelzi RJ, Kirsch WM. Follow-up analysis of ventriculoperitoneal and
[34]
Meier U, Kintzel D. Clinical experiences with different valve systems
ventriculoatrial shunts for hydrocephalus. J Neurosurg 1975; 42:
in patients with normal-pressure hydrocephalus: evaluation of the
679-682
Miethke dual-switch valve. Childs Nerv Syst 2002; 18: 288-294
[12]
Keucher TR, Mealey J. Long-term results after ventriculoatrial
[35]
Trost HA, Sprung C, Lanksch W, Stolke D, Miethke C. Dual-switch
and ventriculoperitoneal shunting for infantile hydrocephalus. J
valve: clinical performance of a new hydrocephalus valve. Acta
Neurosurg 1979; 50: 179-186
Neurochir Suppl (Wien) 1998; 71: 360-363
[13]
Fernell E, von Wendt L, Serlo W, Heikkinen E, Andersson H. Ventricu-
[36]
Sprung C, Miethke C, Shakeri K, Lanksch WR. The importance of
loatrial or ventriculoperitoneal shunts in the treatment of hydro-
the dual-switch valve for the treatment of adult normotensive or
cephalus in children? Z Kinderchir 1985; 40 (Suppl 1): 12-14
hypertensive hydrocephalus. Eur J Pediatr Surg 1997; 7 (Suppl 1):
[14]
Olsen L, Frykberg T. Complications in the treatment of hydrocephalus
38-40
in children. A comparison of ventriculoatrial and ventriculoperito-
[37]
Udayakumaran S, Roth J, Kesler A, Constantini S. Miethke DualSwitch
neal shunts in a 20-year material. Acta Paediatr Scand 1983; 72:
Valve in lumboperitoneal shunts. Acta Neurochir (Wien) 2010; 152:
385-390
1793-1800
[15]
Boockvar JA, Loudon W, Sutton LN. Development of the Spitz-Holter
[38]
Zemack G, Romner B. Adjustable valves in normal-pressure hydro-
valve in Philadelphia. J Neurosurg 2001; 95: 145-147
cephalus: a retrospective study of 218 patients. Neurosurgery 2002;
[16]
Gilbertson MP. An appreciation of John Holter [SRHSB Web site]. June
51: 1392-1400, discussion 1400-1402
[39]
Rohde V, Haberl EJ, Ludwig H, Thomale UW. First experiences with
an adjustable gravitational valve in childhood hydrocephalus. J Neu-
[17]
Nulsen FE, Spitz EB. Treatment of hydrocephalus by direct shunt from
rosurg Pediatr 2009; 3: 90-93
ventricle to jugular vein. Surg Forum 1951; 2: 399-403
[40]
Sprung C, Schlosser HG, Lemcke J et al. The adjustable proGAV shunt:
[18]
Scarff JE. Treatment of hydrocephalus: an historical and critical
a prospective safety and reliability multicenter study. Neurosurgery
review of methods and results. J Neurol Neurosurg Psychiatry 1963;
2010; 66: 465-474
26: 1-26
[41]
Aschoff A, Benesch CPK, von Haken MS, Klank A, Osterloh M, Fruh K.
[19]
Portnoy HD, Schulte RR, Fox JL, Croissant PD, Tripp L. Anti-siphon
The solved and unsolved problems of hydrocephalus valves: a critical
and reversible occlusion valves for shunting in hydrocephalus and
comment. Adv Neurosurgery 1993; 21: 103-114
81
Shunt and Valve Technology
[42]
Tokoro K, Chiba Y, Abe H. Pitfalls of the Sophy programmable
[65]
Drake JM, da Silva MC, Rutka JT. Functional obstruction of an antisi-
pressure valve: Is it really better than a conventional valve and
phon device by raised tissue capsule pressure. Neurosurgery 1993;
an anti-siphon-device? In: Matsumoto P, ed. Hydrocephalus:
32: 137-139
Pathogenesis and Treatment. Tokyo, Japan: Springer Verlag; 1991,
[66]
da Silva MC, Drake JM. Complications of cerebrospinal fluid shunt
pp. 405-421
antisiphon devices. Pediatr Neurosurg 1991-1992; 17: 304-309
[43]
Richards H, Seeley H, Pickard J. Are adjustable valves effective? Data
[67]
Kremer P, Aschoff A, Kunze S. Therapeutic risks of anti-siphon
from the UK Shunt Registry. Cerebrospinal Fluid Res 2007: S30
devices. Eur J Pediatr Surg 1991; 1: 47-48
[44]
Ringel F, Schramm J, Meyer B. Comparison of programmable shunt
[68]
Watson DA. The Delta Valve: a physiologic shunt system. Childs Nerv
valves vs standard valves for communicating hydrocephalus of
Syst 1994; 10: 224-230
adults: a retrospective analysis of 407 patients. Surg Neurol 2005;
[69]
Kondageski C, Thompson D, Reynolds M, Hayward RD. Experience
63: 36-41, discussion 41
with the Strata valve in the management of shunt overdrainage. J
[45]
Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
Neurosurg 2007; 106 (Suppl): 95-102
lus Study: the role of cerebrovascular disease. J Neurosurg 1999; 90:
[70]
Kestle JR, Walker ML Strata Investigators. A multicenter prospective
221-226
cohort study of the Strata valve for the management of hydrocepha-
[46]
Schneider T, Knauff U, Nitsch J, Firsching R. Electromagnetic field haz-
lus in pediatric patients. J Neurosurg 2005; 102 Suppl: 141-145
ards involving adjustable shunt valves in hydrocephalus. J Neurosurg
[71]
Ahn ES, Bookland M, Carson BS, Weingart JD, Jallo GI. The Strata pro-
2002; 96: 331-334
grammable valve for shunt-dependent hydrocephalus: the pediatric
[47]
Bernard M. Valve for the Treatment of Hydrocephalus, S. D. Sophysa,
experience at a single institution. Childs Nerv Syst 2007; 23: 297-303
[72]
Sainte-Rose C, Hooven MD, Hirsch JF. A new approach in the treat-
ment of hydrocephalus. J Neurosurg 1987; 66: 213-226
[73]
Weiner HL, Constantini S, Cohen H, Wisoff JH. Current treatment of
[48]
Sophysa.
15,
normal-pressure hydrocephalus: comparison of flow-regulated and
2013
differential-pressure shunt valves. Neurosurgery 1995; 37: 877-884
[49]
Cabaud F, Coneau P, Negre P. [FR] Valve Sous-Cutanee, F. Sophysa SA,
[74]
Hanlo PW, Cinalli G, Vandertop WP et al. Treatment of hydrocephalus
determined by the European Orbis Sigma Valve II survey: a multicenter
prospective 5-year shunt survival study in children and adults in whom
15, 2013
a flow-regulating shunt was used. J Neurosurg 2003; 99: 52-57
[50]
[75]
Meier U, Zeilinger FS, Reyer T, Kintzel D. [Clinical experience with
depuy-divisions/codman-and-shurtleff. Accessed on April 15, 2013
various shunt systems in normal pressure hydrocephalus]. Zentralbl
[51]
Rosenberg Meir U. [EN] Self adjusting hydrocephalus valve, U. COD-
Neurochir 2000; 61: 143-149
MAN & SHURTLEFF, Patent no. EP000001491232A2, 25.06.2004
[76]
Hara M, Kadowaki C, Konishi Y, Ogashiwa M, Numoto M, Takeuchi K.
A new method for measuring cerebrospinal fluid flow in shunts. J
Neurosurg 1983; 58: 557-561
[52]
Akbar M, Stippich C, Aschoff A. Magnetic resonance imaging and
[77]
Serlo W. Experiences with flow-regulated shunts
(Orbis-Sigma
cerebrospinal fluid shunt valves. N Engl J Med 2005; 353: 1413-1414
valves) in cases of difficulty in managing hydrocephalus in children.
[53]
Anderson RC, Walker ML, Viner JM, Kestle JR. Adjustment and mal-
Childs Nerv Syst 1995; 11: 166-169
function of a programmable valve after exposure to toy magnets.
[78]
Schoener WF, Reparon C, Verheggen R, Markakis E. Evaluation of
Case report. J Neurosurg 2004; 101 (Suppl): 222-225
shunt failures by compliance analysis and inspection of shunt valves
[54]
Nomura S, Fujisawa H, Suzuki M. Effect of cell phone magnetic fields
and shunt materials, using microscopic or scanning electron micro-
on adjustable cerebrospinal fluid shunt valves. Surg Neurol 2005; 63:
scopic techniques. In: Matsumoto P, ed. Hydrocephalus: Pathogenesis
467-468
and Treatment. Tokyo, Japan Springer Verlag; 1991, pp. 452-472
[55]
Utsuki S, Shimizu S, Oka H, Suzuki S, Fujii K. Alteration of the pres-
[79]
Hoekstra A. Artificial shunting of cerebrospinal fluid. Int J Artif
sure setting of a Codman-Hakim programmable valve by a television.
Organs 1994; 17: 107-111
Neurol Med Chir (Tokyo) 2006; 46: 405-407
[80]
Zeilinger FS, Reyer T, Meier U, Kintzel D. Clinical experiences with the
[56]
Nakashima K, Nakajo T, Kawamo M et al. Programmable shunt valves:
dual-switch valve in patients with normal pressure hydrocephalus.
in vitro assessment of safety of the magnetic field generated by a por-
Acta Neurochir Suppl (Wien) 2000; 76: 559-562
table game machine. Neurol Med Chir (Tokyo) 2011; 51: 635-638
[81]
Sainte-Rose C. Shunt obstruction: a preventable complication?
[57]
Yamashita N, Kamiya K, Yamada K. Experience with a programmable
Pediatr Neurosurg 1993; 19: 156-164
valve shunt system. J Neurosurg 1999; 91: 26-31
[82]
Bradley WG. Normal pressure hydrocephalus: new concepts on etiol-
[58]
Shurtleff C. A Precision and Accuracy Study of the Codman Valve
ogy and diagnosis. AJNR Am J Neuroradiol 2000; 21: 1586-1590
Position Verification (VPV) System 2005; NCT00196196. Available at:
[83]
Hakim S. [EN] Ventricular Shunt Having a Variable Pressure Valve, H.
[59]
2013
on April 15, 2013
[60]
Bertrand WJ, Watson DA. [EN] Implantable adjustable fluid flow control
[84]
Meier U. Gravity valves for idiopathic normal-pressure hydrocepha-
valve, U. PUDENZ SCHULTE MED RES, Patent no. US000005637083A,
lus: a prospective study with 60 patients. Acta Neurochir Suppl
(Wien) 2005; 95: 201-205
[85]
Tsunoda A, Maruki C. Clinical experience with a dual switch valve
[61]
Lavinio A, Harding S, Van Der Boogaard F et al. Magnetic field interac-
(Miethke) for the management of adult hydrocephalus. Neurol Med
tions in adjustable hydrocephalus shunts. J Neurosurg Pediatr 2008;
Chir (Tokyo) 2007; 47: 403-408, discussion 408
2: 222-228
[86]
Sprung C, Miethke C, Shakeri K, Lanksch WR. Pitfalls in shunting of
[62]
Czosnyka ZH, Czosnyka M, Richards HK, Pickard JD. Evaluation of
hydrocephalus—clinical reality and improvement by the hydrostatic
three new models of hydrocephalus shunts. Acta Neurochir Suppl
dual-switch valve. Eur J Pediatr Surg 1998; 8 (Suppl 1): 26-30
(Wien) 2005; 95: 223-227
[87]
Park J, Kim GJ, Hwang SK. Valve inclination influences the performance
[63]
Strahle J, Selzer BJ, Muraszko KM, Garton HJ, Maher CO. Programma-
of gravity-assisted valve. Surg Neurol 2007; 68: 14-18, discussion 18
ble shunt valve affected by exposure to a tablet computer. J
[88]
Zachenhofer I, Donat M, Roessler K. The combination of a
Neurosurg Pediatr 2012; 10: 118-120
programmable valve and a subclavicular anti-gravity device in
[64]
Kurtom KH, Magram G. Siphon regulatory devices: their role in the
hydrocephalus patients at high risk for hygromas. Neurol Res
treatment of hydrocephalus. Neurosurg Focus 2007; 22: E5
2012; 34: 219-222
82
Shunt and Valve Technology
[89] Kaestner S, Kruschat T, Nitzsche N, Deinsberger W. Gravitational
[94] Mirzayan MJ, Klinge PM, Samii M, Goetz F, Krauss JK. MRI safety of a
shunt units may cause under-drainage in bedridden patients. Acta
programmable shunt assistant at 3 and 7 Tesla. Br J Neurosurg 2012;
Neurochir (Wien) 2009; 151: 217-221, discussion 221
26: 397-400
[90] Meier U, Lemcke J. First clinical experiences in patients with idio-
[95] Toma AK, Tarnaris A, Kitchen ND, Watkins LD. Use of the proGAV
pathic normal-pressure hydrocephalus with the adjustable gravity
shunt valve in normal-pressure hydrocephalus. Neurosurgery 2011;
valve manufactured by Aesculap (proGAV(Aesculap)). Acta Neurochir
68 Suppl Operative: 245-249
Suppl (Wien) 2006; 96: 368-372
[96] Hooven MD. [EN] Non-invasively adjustable valve, U. CORDIS CORP,
[91] Thomale UW, Gebert AF, Haberl H, Schulz M. Shunt survival rates by
using the adjustable differential pressure valve combined with a
gravitational unit (proGAV) in pediatric neurosurgery. Childs Nerv
Accessed on April 15, 2013
Syst 2013; 29: 425-431
[97] Czosnyka M, Czosnyka Z, Pickard JD. Programmable Shunt Assistant
[92] Freimann FB, Sprung C. Shunting with gravitational valves—can
tested in Cambridge shunt evaluation laboratory. Acta Neurochir
adjustments end the era of revisions for overdrainage-related
Suppl (Wien) 2012; 113: 71-76
events?: clinical article. J Neurosurg 2012; 117: 1197-1204
[98] Miethke C., Miethke GmbH & Co. KG.: Accessories. 2013, http://
[93] Weinzierl MR, Hans F-J, Stoffel M, Oertel MF, Korinth MC. Experience
with a gravitational valve in the management of symptomatic overdrain-
on April 15, 2013
age in children with shunts. J Neurosurg Pediatr 2012; 9: 468-472
83
10.1
Shunt Settings
86
Chapter 10
According to M.F.
Shunt and Valve Settings
10.2
Shunt Settings
86
According to U.K.
Michael J. Fritsch, Uwe Kehler,
Johannes Lemcke, Ullrich Meier
10.3
Shunt Settings
89
According to U.M. and J.L.
10.4
Efficiency of Gravitational Valves
91
Shunt and Valve Settings
10 Shunt and Valve Settings
Michael J. Fritsch, Uwe Kehler, Johannes Lemcke, Ullrich Meier
In this chapter, readers will find three different perspec-
H2O. Patients may benefit from the further reduction of
tives provided by three nonaffiliated neurosurgeons who
the opening pressure, at least for a certain amount of
have considerable experience treating patients with
time. Again, patients are likely to be protected from over-
normal pressure hydrocephalus (NPH). The differences
drainage by the gravitational unit or by another adequate
reflect the fact that—as is quite common in medicine—
device.
there is usually not only one standard solution to a partic-
In patients with iNPH, there is no need to adjust the
ular problem.
valve immediately after surgery. NPH is chronic and long
lasting, so improvement will take some time (usually
weeks or months). Therefore, it is recommended to per-
10.1 Shunt Settings
form the first valve adjustments—if any—3 months after
surgery.
Michael J. Fritsch
In summary, the optimal setting for the treatment of a
patient with iNPH is a VP shunt with an opening pressure
First, ventriculoperitoneal (VP) shunt placement is the
of 5 cm H2O and a gravitational unit (shunt assistant,
treatment of choice for patients with NPH.1 Alternatives,
proSA), which will prevent overdrainage, at 20 cm H2O.
such as endoscopic third ventriculostomy, have not been
Adjustability is beneficial for the patient, and the first
proven to match the clinical results achieved by shunt
adjustments should be performed at about 3 months
placement. Ventriculoatrial (VA) shunts may be used as
after surgery.
an alternative, but they should be used only in patients
with impairment of abdominal absorption capacity (e.g.,
following extensive abdominal surgery or peritonitis);
10.2 Shunt Settings
otherwise, the risk of surgery and the time consumed by
Uwe Kehler
the procedure favors the use of VP shunts.
Second, the initial opening pressure of the valve should
There are a number of shunts and valves with different
be low. In my practice, the valve is always set to 5 cm H2O
pressure settings. The literature gives no clear evidence-
opening pressure. Some evidence suggests that clinical
based answer about the best shunt system; however,
improvement after shunt implantation is significantly
there are many logical reflections and experiences that
better with a low opening pressure.2,3 This low opening
may help when choosing the appropriate valve. Naturally,
pressure is beneficial for the patient, and there is no rea-
a place for discussions and improvements will remain.
son for not providing this benefit to patients.
Currently, there is no exclusive “right way” to perform a
Third, if the initial opening pressure is low, then there
shunt or to select the shunt components, so there will be
is an increased risk for overdrainage. To reduce this risk,
more than one good solution. The following sections will
the shunt should carry a gravitational unit or another
explain how to choose the correct shunt and components
device that reliably prevents overdrainage. Therefore, a
and detail the reasons for my decisions. Besides theoreti-
standard gravitational unit with an opening pressure of
cal considerations, the author (U.K.) will also reflect upon
20 cm H2O is recommended for patients with idiopathic
economic aspects. The decision-making process will be
NPH (iNPH).
divided into these parts:
Fourth, adjustability plays an emerging role in today’s
1. Select:
shunt technology, and physicians and patients demand it.
a) VA versus VP shunt
Adjustability (note that “adjustability” is not the same as
b) Frontal or occipital bore hole
the term “programmable” here, because shunts today are
2. Valve:
not truly programmable, only adjustable) may prevent
a) Opening pressure
the need for revisional surgery if the shunt does not fit
b) Programmable or fixed valve
perfectly well. At the time of publication, there has
3. Shunt configuration
not been a randomized, controlled prospective study
4. Catheter material
confirming the advantage of an adjustable versus a
fixed-pressure valve. However, empirical data and clinical
experience favor adjustability over fixed-pressure valves.
10.2.1 Ventriculoatrial versus
In iNPH, adjustability plays a role at the
3-year or
Ventriculoperitoneal Shunt
4-year follow-up period when the condition of patients is
likely to have deteriorated compared with the improved
The selection between a VA and a VP shunt must be
status initially seen after the shunt was placed. At this
decided in favor of the VP shunt because both shunts
time, the opening pressure can be lowered to 0 to 3 cm
have about the same frequency of complications, but
86
Shunt and Valve Settings
complications in VA shunts are potentially more
severe.4-6 This is especially true in the elderly patient
population with chronic heart and lung disease,
chronic obstructive pulmonary disease, and pulmo-
nary hypertension; the presence of cardiac complica-
tions are even more dangerous. Of course, if there are
any contraindications (e.g., ascites, extensive perito-
neal scarring), then VA shunts are an appropriate
alternative.
10.2.2 Frontal versus Parietal/
Occipital Bore Hole
There are no clear data to support one approach or the
other for the ventricular catheter. However, there are
three main reasons why a frontal bore hole should be
used, namely:
1. The probability of hitting the ventricular system
with the ventricular catheter is higher, because the
tolerable diversion from the optimal angle where
the ventricle is still hit is wider in the frontal
region than in the occipital region (▶ Fig. 10.1,
▶ Fig. 10.2).
2. The tip of the catheter is—if approached from the
anterior—easy to locate in the frontal choroid plexus
free zone of the lateral ventricle. Plexus invasion is
a commonly described cause of shunt obstructions.7
Using the occipital approach, the ventricular cathe-
ter must be very long to reach with its perforations
in the frontal located free plexus zone; this makes it
more difficult.
3. Shunt revisions are associated with additional risk of
intraventricular hemorrhage due to adherence to the
Fig. 10.2
Magnetic resonance imaging (MRI) with optimal track
choroid plexus.8
for a occipital ventricular catheter (green line) with tolerance of
track deviation where the catheter still enters the ventricle
(yellow lines) in the (a) sagittal and (b) axial planes.
For these reasons, the frontal approach is preferable.
a
b
Fig. 10.1 Magnetic resonance imaging (MRI) with optimal track for a frontal ventricular catheter (green line) with tolerance of track
deviation where the catheter still enters the ventricle (yellow lines) in the (a) sagittal and (b) coronal planes. The wider the angle of the
extremes of the tolerated tracks, the lower the risk of failed ventricle puncture (compare also ▶ Fig. 10.2).
87
Shunt and Valve Settings
values of different parameters may change over time; for
10.2.3 Selection of Valve
example, intra-abdominal pressure with increasing
Opening Pressure
weight or constipation may influence the resulting ICP.
Boon et al have shown clearly that we should use a low-
In such cases, an adjustment of the valve would be desir-
able. However, adjusting a differential pressure valve has
pressure valve in patients with NPH,9 and, to counter-
an important shortcoming: downregulating the differen-
balance overdrainage, we should also use a gravitational
device (Section 10.4).10
tial valve not only results in improved drainage in the
The Boon study9 supports the theoretical consideration
horizontal position but also accelerates drainage in the
that normal intracranial pressure
(ICP) should be
upright position, threatening overdrainage (see Chapter 9).
achieved with the shunt. Calculating the resulting ICP in a
Increasing the VOP would improve the drainage in
VP-shunted patient, the hydrostatic pressure difference
the upright position but will worsen the drainage in the
between the head and the peritoneal cavity in an upright
horizontal position. Therefore, an adjustment of the
position, the abdominal pressure, and the valve opening
gravitational device would be advantageous: because
pressure (VOP) should be considered. In the horizontal
the programmable gravitational shunt assistant (proSA)
works in the upright position only, an adjustment can be
position, the hydrostatic pressure difference can be
neglected, being almost 0. According to the equation,
performed with an effect in the upright position only, and
not simultaneously interfere when in the horizontal posi-
tion. However, the high prices of adjustable valves make
ICP ¼ hydrostatic difference þ VOP
ð20Þ
routine use with low reimbursements difficult.
þ abdominal pressure
Theoretical considerations motivate us to use pro-
grammable shunt assistants rather than programmable
differential pressure valves. The economic situation
(see Box: Equation for calculating ICP and VOP in patients
affects the selection also: In uncomplicated cases, we
with VP shunts), the VOP should be around 0 to 5 cm
initially use
(the cheaper) nonprogrammable valves
H2O to achieve a physiologic ICP (of 5-10 cm H2O) in a
with low opening pressures and hydrostatic pressure
horizontal position. In the upright position, a gravita-
compensation around 30 to 35 cm H2O (see above). In
tional device should be implanted to counterbalance
revision surgeries or shunt implantations, when we
the difference in hydrostatic pressure. Usually a gravi-
expect problems, we select (the expensive) program-
tational device with a resistance of 30 to 35 cm H2O is
mable valves.
sufficient in patients with a body height of
160 to
190 cm (5.3-6.2 ft).
10.2.5 Shunt Configuration
Equation for calculating ICP and VOP in
(with or without a Cerebrospinal
patients with VP shunts
Fluid Reservoir)
If the shunt works well, no additional devices are needed.
ICP ¼ VOP þ GD HPD þ IAP
ð21Þ
However, unfortunately, not all shunts work as well as
⇒ VOP ¼ ICP þ HPD GD IAP
they should. In those cases, an additional cerebrospinal
fluid (CSF) reservoir is very helpful: CSF reservoirs permit
Abbreviations: GD, setting of gravitational device;
through palpation, pumping, and even puncturing a
HPD, hydrostatic pressure difference between ventricle
rough diagnosis of the cause of shunt malfunction (see
and abdomen; IAP, intra-abdominal pressure; ICP, intra-
also Fig. 15.6). Below are brief diagnoses of the causes of a
cranial pressure; VOP, valve opening pressure; VP, ventri-
shunt malfunction:
culoperitoneal.
1. If the reservoir can be pressed out by simultaneous
occlusion of the proximal catheter, then this means
that the valve and the distal catheter are not totally
Clinical experience supports a valve selection with a very
blocked. (However, a differentiation between mild,
low opening pressure (0-5 cm H2O) and a hydrostatic
moderate, or distinct elevated opening pressure is not
component of 30 to 35 cm H2O.
possible.)
2. If the reservoir does not refill promptly after lifting the
10.2.4 Should the Valve and/or
finger from the proximal catheter after the first proce-
the Gravitational Device be
dure, then the diagnosis of a proximal catheter
obstruction or slit ventricles is made. This provides a
Programmable?
hint of where to search further.
The equation for calculating the ICP and determining the
3. By “pumping” the reservoir (compressing the proximal
VOP suggests that the choice of the valve is clearly
catheter during pumping to avoid a reflux to the ven-
defined so that a fixed valve is sufficient. However, the
tricle), a fibrin clot or debris, which may have occluded
88
Shunt and Valve Settings
the valve or distal catheter, may be mobilized and the
sary and what is not. Responsible economic and medical
shunt function can be restored (in a few cases).
indications and the use of sophisticated shunt devices are
4. “Pumping” can be used as a noninvasive tap test:
all necessary for us to help as many patients as possible
instead of a spinal tap test for re-evaluating the situa-
without burdening the entire society.
tion, about 100 to 200 “pumps” can pull a substantial
volume (depending on the size of the pumping reser-
voir) from the ventricles.
10.3 Shunt Settings
5. In unclear cases, puncture of the CSF reservoir makes it
Ullrich Meier, Johannes Lemcke
possible to perform microbiologic examinations, ICP
measurements, and a shuntogram.
The implantation of a valve-regulated shunt as the ther-
apy of first choice is indisputable in patients with iNPH.
For additional information, see Section 15.4.
The VP shunt for adults has gained wide acceptance inter-
The advantages of a reservoir during shunt failure are
nationally. Following more than 50 years of experience
so convincing that the author (U.K.) almost always uses
worldwide with valve-regulated shunt therapy of internal
such a reservoir. He prefers the large Sprung Borehole
hydrocephalus, there are still substantial therapeutic
Reservoir from Miethke (Potsdam, Germany), which has
problems that derive from the nonphysiologic principles
an integrated antireflux valve that eases pumping in
of construction of these valves. These difficulties are
higher volumes
(the antireflux valve helps pumping
reflected by the fact that more than 200 different valves
because no additional occlusion of the proximal catheter
are in the market today, with different construction prin-
is necessary). The disadvantage is its size and, conse-
ciples, different characteristic curves for different valves,
quently, the cosmetic aspect in patients who are bald
and different modes of operation.15,16
(using a frontal approach).
Conventional differential pressure valves have the dis-
advantage, particularly for patients with iNPH, that when
10.2.6 Selecting Catheter Material
set up in a vertical position, they tend to open suddenly,
remaining open for too long while generating a suction
Usually the clinician has only a few choices for the catheter.
force on the CSF space of the already atrophic, previously
Currently, there are two main different catheters, namely,
damaged brain. The fundamental question then becomes
antibiotic impregnated
(BACTISEAL) or nonimpregnated
whether such suction phenomena, with consequent over-
catheters. Silver-impregnated catheters are expected soon
drainage complications, can be reduced or prevented by
for shunts. Presently, the shunt infection rates in patients
the use of gravitational valves.
with NPH are low, at around 3% to 4%.11,12 However, this
rate is still too high. The first studies with antibiotic-
impregnated shunts have reported promising results12,13;
10.3.1 Why Use Gravitational
however, there is still a worry about increasing resistance
Valves?
to antibiotics, and increasing resistance to antibiotics with
all their negative side effects.14 The next obstacle to the use
Effective therapy of disease-specific symptoms following
of these impregnated catheters in all patients is their high
the implantation of a differential pressure valve with a
cost. However, if the reduction of CSF shunt infections can
lower valve setting in patients with iNPH has the dis-
be proved without creating new problems with regard to
advantage of a significantly greater overdrainage rate.2 The
antibiotic resistance, their use will be a must. Currently,
fundamental problem that the pressure gradient is
antibiotic-impregnated catheters are used in only those
increased when positioning the body axis in the vertical
patients who are at risk of noncurable infections.
direction by the addition of a hydrostatic pressure compo-
nent has, in the past, been countered using a variety of
strategies. Early on, it was found that differential pressure
10.2.7 The “Optimal” Shunt Setting
valves with high opening pressures could prevent over-
The optimal shunt would be an antibiotic-impregnated
drainage, which, in particular, comes into effect in the ver-
shunt with a programmable differential pressure valve,
tical body position. However, this happens only at the
with a programmable gravitational device, and a CSF
expense of permanent underdrainage in the horizontal
reservoir with an integrated ICP teletransducer. The tele-
position when, because of the absent hydrostatic pressure
transducer could guide us on how to properly adjust the
component and the high opening pressure, drainage of CSF
valves. However, the costs would be higher than the
no longer takes place.17-19 Therefore, the occurrence of
reimbursement by most health insurance companies,
pressure peaks at night could not be compensated. The use
thus making hydrocephalus treatment a money-losing
of differential pressure valves does provide an effective
business. This could lead to an extreme conservative indi-
therapy for disease-specific symptoms; however, a subs-
cation for shunt surgery, thus withholding beneficial
tantially greater rate of overdrainage must be accepted.2
therapy from many patients. The future must show
With later constructions, such as the antisiphon device,
evidence-based results that detail what is really neces-
malfunctions resulted because of the surrounding growth
89
Shunt and Valve Settings
pressure range compared with differential pressure valves
in the intermediate pressure range. However, this advan-
tage resulted in a higher overdrainage rate of 73% versus
Fig. 10.3 proGAV (adjustable valve/gravitational unit).
34%.2 No statement was made regarding the clinical rele-
vance of this postoperative complication, which was
described as a subdural hygroma. These rates of complica-
of scar tissue.20 The development of adjustable valves did
tion should be considered to be exceptionally high. In
indeed open up the possibility of programming the pres-
addition, a clinical study carried out by the neurosurgical
sure to a value so high that overdrainage could no longer
clinics of the Unfallkrankenhaus Berlin and the University
result, but then a sufficient flow of CSF could not be guar-
of Homburg Saar in 200414 confirmed that the course of
anteed. Only when gravitational valves that are capable
the disease in patients with iNPH and implanted gravita-
of switching from one pressure level for the horizontal
tional valves in the low pressure range of 50 mm H2O or
position and another for a standing position were
lower produced results that were significantly better than
developed was the decisive step taken to eliminate the
that with gravitational valves in a range of 100 to 130 mm
problems previously referred to.21,22
H2O. The advantage of the implanted gravitational valves
The author (U.M.) has gained experience with gravita-
is that, with an overdrainage rate of 10% versus 4% com-
tional valves since 1996.23 In the beginning, static and
pared with the Dutch study, it was clearly smaller, but the
nonadjustable gravitational valves, such as the Miethke
complication was still present between the valve pressure
DUALSWITCH valve Aesculap, were implanted in patients
levels. This question was successfully investigated and
with iNPH.22,24 Since 2004, in the majority of cases, adjust-
resolved in 2013 in a prospective and randomized, multi-
able gravitational valves, mostly the proGAV Aesculap, but
center study carried out by German clinics (called the
also the combination of adjustable MEDOS CODMAN
SVASONA study; refer to Section 10.4).10
valves with the Miethke SHUNTASSISTANT Aesculap,19,24,25
On reviewing the international literature on under-
have been implanted (▶ Fig. 10.3). Both types of valves or
drainage or overdrainage following a shunt operation, it
valve combinations allowed good intraoperative handling
becomes apparent that there is a serious lack of agree-
and the possibility of easy adjustment. The crucial disadvan-
ment and that there are discrepancies with regard to the
tages of a combination of an adjustable MEDOS CODMAN
definition and subdivision of these postoperative compli-
valve with a Miethke SHUNTASSISTANT Aesculap are the
cations. Some authors counted the number of overall
unintentional maladjustment of the valve and the necessity
complications, while others counted the mechanical com-
of X-ray control after each valve adjustment. With both
plications only, leaving aside infections. Drake et al25
types of valve, care must be taken to achieve precise vertical
combined underdrainage as a complication together with
orientation of the gravitational valve to ensure trouble-free
occlusions, and defined overdrainage as subdural
operation of the gravitational unit and to minimize poten-
hygromas and the slit ventricle syndrome. Boon et al2 did
tial complications of overdrainage.
not define underdrainage and, likewise, did neither
Currently, the proGAV Aesculap is, in the view of the
describe subdural hygromas as overdrainage nor a clinical
author, the optimal type of valve for patients with iNPH.
symptom of overdrainage. Our view is that one should
In the Unfallkrankenhaus Berlin
(Accident and
speak only of a functional underdrainage when the pres-
Emergency Hospital, Berlin), adjustable gravitational
sure setting of the valve is too high, the actual value of
valves such as the proSA Aesculap are implanted in spe-
the valve pressure setting is increased in vivo in relation
cial cases only, or following complications, especially in
to the target value because of manufacturing defects or
patients who are overweight and those who are very
changes in valve function in vivo, or as a result of an
slim. Therefore, the adjustable gravitational valve proSA
increase in intraperitoneal pressure as a consequence of
Aesculap represents a secondary option.
adiposity or an insufficiency of CSF reabsorption or CSF
outflow being reduced (e.g., in a pseudocyst).
In the shunt registry of England,26 which had a large
10.3.2 Does Valve Opening Pressure
number of patients—more than 10,000 cases—under-
of Hydrostatic Valves Have an Influ-
drainage was in the foreground with a value of 52%. Over-
drainage, with a value of 3%, was stated to be very low as
ence on the Course of the Disease?
a postoperative cause of complications. In contrast, a
Even after a precisely determined diagnosis, surgical ther-
Scandinavian research group27 has reported that 80% of
apy is fraught with complications in patients with iNPH, a
all shunt complications result from overdrainage.
fact that significantly affects the clinical course and
As a rule, in the Unfallkrankenhaus Berlin, valves
the rate of improvement.15 The Dutch Normal Pressure
are implanted in patients with iNPH with a low pressure
Hydrocephalus Study2 in 1999 produced a significantly
setting of 70 mm H2O or lower. Only in the context of clin-
better improvement in the course of the disease when
ical studies, for example, the SVASONA study (refer to
differential pressure valves were implanted in the low
Section 10.4) was an implantation carried out at 100 mm
90
Shunt and Valve Settings
H2O. In those cases the valve was adjusted to 70 mm H2O
Although the construction principle was plausible and
and with all other patient groups to 50 mm H2O after
clinical experience appeared to corroborate the theory,
3 months. This valve adjustment strategy was undertaken
there was a lack of evidence for the efficiency of gravita-
from the perspective that, during the operation, as a result
tional valves.7 With this background, a randomized multi-
of a loss of CSF, a relative underdrainage is iatrogenically
center trial was designed to compare low-pressure valves
produced, which should be compensated by the valve
without gravitational units with low-pressure valves with
adjustment. In our experience, 70% of patients with iNPH
gravitational units.
show an optimal disease course with a valve adjustment
To compare low-pressure valves with or without gravi-
between 40 and 60 mm H2O.14,19A total of 15% of patients
tational units, patients were randomly allocated to two
require a valve adjustment of 20 or 30 mm H2O, 10% of
treatment groups. To increase patient safety, programma-
patients require an adjustment of 70 to 90 mm H2O, and
ble valves were used. The valves were implanted with an
5% of patients require 0 or 10 mm H2O. It should be noted
opening pressure of 100 mm H2O. Three months after
that these figures apply exclusively to implanted gravita-
surgery, the valves were adjusted into the low pressure
tional valves and not solely to differential pressure valves.
range (70 mm H2O). Because there is no precisely pro-
In contrast, for the selection of the gravitational unit
grammable valve commercially available with or without
(ShuntAssistant), the height of the patient is decisive for
a gravitational unit, group I received proGAV valves
the gravitational unit:
(Aesculap, Miethke) and group II received CODMAN
● Height < 160 cm = 200 mm H2O
MEDOS programmable valves
(Codman, Johnson &
● Height 160 to 180 cm = 250 mm H2O
Johnson, Raynham, Massachusetts, United States).30-32
● Height > 180 cm = 300 mm H2O.
To achieve significant results, a sample size of 250
patients with a 50/50 split into the treatment groups was
included in the calculations. An interim analysis was
10.3.3 Conclusion regarding
planned half-way. The study would be stopped if the
Clinical Practice
experimental device produced a statistically significant
According to international experience as well as our own
advantage over the control treatment.
experience regarding patients with iNPH,19,28,29
the
Clinical assessment, including the Kiefer score1 and the
adjustable gravitational valve currently represents the
SF-12 (short form health survey), was conducted before
gold standard for therapy. For most patients, the optimal
the study, and 6 and 12 months postoperatively. The NPH
VOP setting lies in the low range for the horizontal
recovery rate was calculated from the Kiefer score. Com-
position with values around 50 mm H2O.14 The pressure
puted tomography scans were performed preoperatively,
setting for the gravitational unit (high pressure range
at the time of discharge, and after 3, 6, and 12 months.
for sitting and standing positions) should be selected
The first end point of the SVASONA study10 was over-
according to the height of the patient.
drainage, which was defined as subdural hygromas or
subdural hematomas of at least 3 mm or clinical signs of
overdrainage that led to valve adjustment to 90 mm H2O
10.4 Efficiency of Gravitational
or more in both groups or back fitting of a gravitational
unit in patients of group II.
Valves
At the interim analysis,
145 of the
151 recruited
Ullrich Meier, Johannes Lemcke
patients had full data available at 6 months of follow-up.
The observed risk difference in overdrainage events
Overdrainage complications have always been a major
exceeded the presumed effect size; thus, the investigators
problem in the therapy of patients with iNPH. This prob-
decided to terminate the trial at that time.
lem became even larger when Boon et al9 demonstrated
A total of 71 patients in the treatment group without
that low-pressure valves may lead not only to a better out-
gravitational units and 74 patients in the treatment group
come of patients than medium-pressure valves but also to
with gravitational units in the intent-to-treat population
a much higher rate of overdrainage complications. Facing
had clinical follow-up data at 6 months.
this dilemma, gravitational valves were constructed.
The 6-month cumulative incidence of overdrainage
The underlying problem of overdrainage complications
complications was 26 of 74 patients in the treatment
is the different hydrostatic pressure in a VP shunt in the
group with gravitational valves, and 4 of 74 patients in
upright and the horizontal positions. Gravitational valves
the treatment group without gravitational valves. The dif-
must be able to switch between a low-pressure mode in
ference was highly significant statistically (P < 0.001).
the horizontal position and a high-pressure mode in the
Underdrainage complications occurred in 3 of 74 patients
upright position. Gravitational units are ball-in-cone
without gravitational valves and in 2 of 77 patients with
valves. The closing force is applied by slidable arranged
gravitational valves.
ball with a heavy weight that works on the valve ball if
According to the Kiefer score, a significant clinical
the device is moved into the upright position.22
improvement was recorded in the group without
91
Shunt and Valve Settings
gravitational valves and in the group with gravitational
[11]
Kehler U, Langer N, Gliemroth J et al. Reduction of shunt obstructions
by using a peel-away sheath technique? A multicenter prospective
valves. The difference between the treatment groups
randomized trial. Clin Neurol Neurosurg 2012; 114: 381-384
was not significant.
[12]
Farber SH, Parker SL, Adogwa O, McGirt MJ, Rigamonti D. Effect of
Whereas a significant difference between the compli-
antibiotic-impregnated shunts on infection rate in adult hydrocepha-
cation rates of the two treatment groups was evident,
lus: a single institution’s experience. Neurosurgery
2011;
69:
there was only a tendency in the difference between the
625-629, discussion 629
[13]
Parker SL, Anderson WN, Lilienfeld S, Megerian JT, McGirt MJ. Cere-
outcomes measured by specific iNPH scores. These find-
brospinal shunt infection in patients receiving antibiotic-impreg-
ings underscore the evidence that gravitational valves
nated versus standard shunts. J Neurosurg Pediatr 2011; 8: 259-265
prevent the risk of overdrainage but do not suppress the
[14]
Demetriades AK, Bassi S. Antibiotic resistant infections with antibi-
favorable outcomes achieved with low-pressure valves.
otic-impregnated Bactiseal catheters for ventriculoperitoneal shunts.
Br J Neurosurg 2011; 25: 671-673
[15]
Aschoff A, Kremer P, Benesch C, Fruh K, Klank A, Kunze S. Overdrain-
10.4.1 Conclusion
age and shunt technology. A critical comparison of programmable,
hydrostatic and variable-resistance valves and flow-reducing devices.
Gravitational valves are highly effective at avoiding over-
Childs Nerv Syst 1995; 11: 193-202
[16]
Czosnyka Z, Czosnyka M, Richards HK, Pickard JD. Laboratory testing of
drainage complications and do not show an unfavorable
hydrocephalus shunts — conclusion of the U.K. Shunt evaluation pro-
influence on other parameters over the course of disease.
gramme. Acta Neurochir (Wien) 2002; 144: 525-538, discussion 538
[17]
Frim DM, Goumnerova LC. In vivo intracranial pressure dynamics in
patients with hydrocephalus treated by shunt placement. J
Conflicts of Interest
Neurosurg 2000; 92: 927-932
The study was conducted as an investigator-initiated trial
[18]
de Jong DA, Delwel EJ, Avezaat CJ. Hydrostatic and hydrodynamic
considerations in shunted normal pressure hydrocephalus. Acta
and was monitored by the Center of Clinical Research
Neurochir (Wien) 2000; 142: 241-247
UKB SVASONA. All participants have received grants for
[19]
Meier U, Lemcke J, Al-Zain F. Clinical experience in the treatment
each enrolled patient from B. Braun Aesculap, Melsungen,
of idiopathic normal-pressure hydrocephalus using the program-
Germany. (Participants: Ullrich Meier, Johannes Lemcke,
mable gravity-assisted valve
(proGAV Aesculap). Neurosurg Q
Cornelia Müller, Michael J. Fritsch, Michael Kiefer, Regina
2007; 17: 52-55
[20]
Kiefer M, Eymann R, Meier U. Five years experience with gravitational
Eymann, Uwe Kehler, Niels Langer, Martin U. Schuhmann,
shunts in chronic hydrocephalus of adults. Acta Neurochir (Wien)
Andreas Speil, Friedrich Weber, Victor Remenez, Veit
2002; 144: 755-767, discussion 767
Rohde, Hans-Christoph Ludwig, Dirk Stengel.)
[21]
Meier U, Kiefer M, Sprung C. Normal Pressure Hydrocephalus: Pathol-
ogy, Pathophysiology, Diagnostics, Therapeutics and Clinical Course.
Erwitte: PVV Science Publications; 2003
References
[22]
Meier U, Kiefer M, Sprung C. Evaluation of the Miethke dual-switch
valve in patients with normal pressure hydrocephalus. Surg Neurol
[1] Kiefer M, Unterberg A. The differential diagnosis and treatment of nor-
2004; 61: 119-127, discussion 127-128
mal-pressure hydrocephalus. Dtsch Arztebl Int 2012; 109: 15-25, quiz 26
[23]
Meier U, Kintzel D. Clinical experiences with different valve systems
[2] Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
in patients with normal-pressure hydrocephalus: evaluation of the
lus Study: the role of cerebrovascular disease. J Neurosurg 1999; 90:
Miethke dual-switch valve. Childs Nerv Syst 2002; 18: 288-294
221-226
[24]
Meier U. Outcome of idiopathic normal-pressure hydrocephalus after
[3] Meier U, Kiefer M, Lemcke J. On the optimal opening pressure of
surgery with gravity valves. Neurosurg Q 2004; 14: 119-126
hydrostatic valves in cases of idiopathic normal-pressure hydroceph-
[25]
Drake JM, Kestle JR, Milner R et al. Randomized trial of cerebrospinal
alus: a prospective randomized study with 122 patients. Neurosurg
fluid shunt valve design in pediatric hydrocephalus. Neurosurgery
Q 2005; 15: 103-109
1998; 43: 294-303, discussion 303-305
[4] Ignelzi RJ, Kirsch WM. Follow-up analysis of ventriculoperitoneal and ven-
[26]
Richards HK, Seeley HM, Pickard JD. Shunt revisions: data from the
triculoatrial shunts for hydrocephalus. J Neurosurg 1975; 42: 679-682
UK shunt registry. Eur J Pediatr Surg 2000; 10 (Suppl.) I: 59
[5] Kluge S, Baumann HJ, Regelsberger J et al. Pulmonary hypertension
[27]
Børgesen SE. Conductance to outflow of CSF in normal pressure
after ventriculoatrial shunt implantation. J Neurosurg 2010; 113:
hydrocephalus. Acta Neurochir (Wien) 1984; 71: 1-45
1279-1283
[28]
Sprung C, Schlosser HG, Lemcke J et al. The adjustable proGAV shunt:
[6] Lam CH, Villemure JG. Comparison between ventriculoatrial and ven-
a prospective safety and reliability multicenter study. Neurosurgery
triculoperitoneal shunting in the adult population. Br J Neurosurg
2010; 66: 465-474
1997; 11: 43-48
[29]
Lemcke J, Meier U, Müller C et al. On the method of a randomized
[7] Collins P, Hockley AD, Woollam DH. Surface ultrastructure of tissues
comparison of programmable valves with and without gravitational
occluding ventricular catheters. J Neurosurg 1978; 48: 609-613
units: the SVASONA study. Acta Neurochir Suppl 2012; 114: 243-246
[8] Brownlee RD, Dold ONR, Myles ST. Intraventricular hemorrhage com-
[30]
Toma AK, Tarnaris A, Kitchen ND, Watkins LD. Use of the proGAV
plicating ventricular catheter revision: incidence and effect on shunt
shunt valve in normal-pressure hydrocephalus. Neurosurgery 2011;
survival. Pediatr Neurosurg 1995; 22: 315-320
68
(Suppl Operative): 245-249
[9] Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
[31]
Lemcke J, Meier U. Improved outcome in shunted iNPH with a combi-
lus Study: randomized comparison of low- and medium-pressure
nation of a Codman Hakim programmable valve and an Aesculap-
shunts. J Neurosurg 1998; 88: 490-495
Miethke ShuntAssistant. Cent Eur Neurosurg 2010; 71: 113-116
[10] Lemcke J, Meier U, Müller C et al. Safety and efficacy of gravitational
[32]
Meier U, Lemcke J. First clinical experiences in patients with idio-
shunt valves in patients with idiopathic normal pressure hydroceph-
pathic normal-pressure hydrocephalus with the adjustable gravity
alus: a pragmatic, randomised, open label, multicentre trial (SVA-
valve manufactured by Aesculap (proGAV(Aesculap)). Acta Neurochir
SONA). J Neurol Neurosurg Psychiatry 2013; 84: 850-857
Suppl (Wien) 2006; 96: 368-372
92
11.1
Settings in the Operating Room
94
Chapter 11
11.2
Positioning
94
Surgical Technique
11.3
Shaving and Disinfection
94
Michael J. Fritsch
11.4
Draping
94
11.5
Surgical Procedure
95
11.6
Alternatives to the VP Shunt
99
Surgical Technique
11 Surgical Technique
Michael J. Fritsch
Shunt surgery is considered to be an easy surgery.
The initial pressure of the valve is set to 5 cm opening
Among neurosurgical operations, shunt placement is
pressure; this can be adjusted in adjustable valves before
one of the least time-consuming procedures, and, with
surgery. Further details about shunt and valve settings
regard to surgical risk and microsurgical capability, it
can be found in Chapter 15, and a discussion about over-
is also one of the least challenging. However, as most
drainage and underdrainage and their related complica-
neurosurgeons are aware, shunt surgery has significant
tions can be found in Chapter 10.
potential for complications. There are many details
that may not be conducted in a perfect way and,
therefore, may contribute to a poor outcome or even
11.2 Positioning
to complications.
Positioning of the patient is essential for smooth-running
There is no particular or a single specific method of
of surgery and should be performed by the surgeon or an
performing shunt operation. Different surgical techniques
experienced assistant. If there is no reason to operate on
may lead to successful outcomes. The here presented sur-
the patient’s left side
(like previous surgeries or
gical technique for placement of a ventricular-peritoneal
implants), then we prefer to operate on the right side.
shunt (VP shunt) is based on personal experience and
The patient should be in a supine position. The head rests
communication with other neurosurgeons. It is also
either in a horseshoe-head holder or on the table in a gel
based on the training experience and the practice in
head ring. The head is turned to the opposite side, usually
different neurosurgical centers.
rotated 45° to 60°, and slightly tilted posteriorly. The right
shoulder is slightly elevated. There should be a straight
line between the chest, neck, and retroauricular region to
11.1 Settings in the
allow for easy tunneling.
Operating Room
We follow the “shunt rules” established by Maurice
11.3 Shaving and Disinfection
Choux, especially those implemented for pediatric
cases.1 Therefore, if it is an elective procedure, shunt
In adult patients, we shave the frontal, temporal, and ret-
surgery should be the first case in the morning. During
roauricular regions with clippers. In contrast, in children
surgery, the number of personnel in the operating
(not a topic of this book), we shave only a small strip fol-
room (OR) should be limited as much as possible (e.g.,
lowing the shunt path or, in newborns, we do not shave
the head at all. In our opinion, it is important to manually
surgeon, assistant, anesthesiologist, scrub nurse).
Shunt hardware must be in the OR before incision of
clean the skin before disinfection. The surgeon should
the skin. Doors should be closed and marked with a
then perform the disinfection so that he or she can recog-
sign that indicates shunt surgery is taking place and
nize the required borders of the surgical field. This is
there will be limited access. There is no movement of
especially important in revision cases or if there is a
potential change in the surgical plan during the proce-
staff in and out of the OR during surgery unless there
is an emergency. As a standard procedure—as in many
dure; the surgeon may need to think in advance about
centers- a single shot of antibiotic prophylaxis (cefur-
the extent of disinfection.
oxime 1.5 g intravenously) is administered 30 minutes
before incision of the skin. There is no routine use of
11.4 Draping
antibiotics postoperatively.
Successful shunt surgery begins with the indication.
The surgeon should also perform the draping. We start
The second most important step is to choose the appro-
with a surgical foil that covers nearly all of the disinfected
priate shunt hardware. Ambulatory patients
(i.e., the
skin. The foil should be applied when the patient’s skin
majority of patients with idiopathic normal pressure
has completely dried. Overlap is possible. The draping
hydrocephalus [iNPH]) are fitted with a gravity-assisted
is then applied from head to abdomen, starting with a
valve, while nonambulatory patients (i.e., the majority
U-shaped drape on the head, followed by two straight
of patients with secondary NPH) are fitted with a
drapes on each side, finishing with a large straight drape
differential pressure valve. Ambulatory patients have an
perpendicular to the latter drapes. Of course, application
increased risk of overdrainage. Patients who are nonam-
of the drapes first and the foil second is also possible.
bulatory, but remain in a horizontal position for most of
Once the draping is finished, the rest of the instruments
the day, have a lesser risk of overdrainage and may bene-
required for surgery are brought on to the table. After-
fit from a differential pressure valve.
ward, gloves are changed (▶ Fig. 11.1).
94
Surgical Technique
Fig. 11.1 Patient positioned and draped for
a right ventriculoperitoneal shunt.
bore hole should be placed
11 cm above the nasion
11.5 Surgical Procedure
(which, in Caucasian patients, is 2 cm anterior to the cor-
Two surgeons perform the surgery together. The hard-
onal suture); 3 cm paramedial to the midline; and the
ware is placed in a proximal-to-distal direction. We begin
proximal catheter should be advanced into the brain 6 cm
with a precoronal bore hole, implant the ventricular cath-
from the inner table of the skull. Anatomical landmarks
eter, followed by the valve, and then the distal peritoneal
(to puncture the ipsilateral frontal horn) are the median
catheter. Other surgeons prefer to operate from both
cantus of the ipsilateral eye and the tragus of the ipsi-
sides, one surgeon starting at the bore hole, the other at
lateral ear. Following these measurements, it is possible
the abdominal incision.
to place the ventricular catheter in the frontal horn, away
The shunt should be handled with instruments rather
from the foramen of Monro in nearly all patients, even
than the surgeon’s hands or gloves (if possible) so as to
if they have small ventricles. Different methods for opti-
avoid any contact with the patient’s skin, and the shunt
mal positioning of the ventricular catheter have been
should be taken out of the package as late as possible
published.2
before implantation. If, for any reason, the shunt is kept
After placement of the bore hole, first the dura is
outside the package, then it should be covered with
opened and then the arachnoid. Ventricular puncture
sterile fluid.
should not be performed without incising the arachnoid.
We connect the ventricular catheter, which has a length
of 6.5 cm (bringing it exactly 6 cm into the brain meas-
11.5.1 Ventricular Catheter
ured from the inner table of the skull), to a bore hole
The skin incision for the frontal approach is curved lin-
reservoir before its placement in the ventricular system.
early. This type of incision allows for the placement of a
From the bore hole reservoir, a distal catheter, leading to
bore hole reservoir completely under the skin and avoids
the valve, is placed in a subgaleal plane and it appears at
having to puncture the reservoir later through an incision
the second skin incision behind the ear (▶ Fig. 11.2).
in the skin.
After this part of the shunt has been placed under
The bore hole is placed at Kocher’s point, which is 2 cm
the skin, the ventricular system is punctured usually
precoronal and 3 cm paramedian. Use of the standard
using a Cushing or Scott cannula. After the puncture
measurements of 11-3-6 is highly recommended: the
and confirmation of cerebrospinal fluid (CSF) flow, the
Fig. 11.2 Patient operated on the left side.
The ventricular (proximal) catheter is con-
nected to the bore hole reservoir and the
distal catheter (coming from the reservoir)
is positioned in the subgaleal plane. The
valve and the peritoneal catheter have
already been positioned subcutaneously.
95
Surgical Technique
Fig. 11.3 Puncture of the ventricle utilizing a Scott cannula.
entire cannula is removed and—using bayoneted ana-
tomic forceps—the proximal catheter is advanced using
the same puncture tract into the ventricular system.
Once the catheter has reached the ventricle, CSF will
flow into the bore hole reservoir.
After mild pumping, distal CSF flow will occur. At this
a
point, CSF is taken for laboratory tests (e.g., glucose, sugar,
protein, cell count) and, if indicated, samples for gram
stain and culture are also taken. We avoid taking CSF
from the ventricular catheter directly after the puncture
because, in small ventricles (which are rare in patients
with NPH), the surgeon may lose the opportunity for a
second puncture, if needed, when the ventricle becomes
too small (▶ Fig. 11.3, ▶ Fig. 11.4, ▶ Fig. 11.5).
Other surgeons prefer to puncture the ventricle with
the ventricular catheter itself, which is armed with a
guidewire. The advantage is the single passage through
the brain parenchyma, but the disadvantage is that the
connection between the ventricular catheter and distal
shunt parts must be performed with the catheter already
b
in place. This usually requires manipulation with in-and-
out movements and carries a risk of displacement.
Fig. 11.4 (a and b) Removal of the Scott cannula and
positioning of the ventricular catheter bluntly within the same
puncture tract using bayoneted anatomic forceps. The tech-
11.5.2 Valve
nique is demonstrated on two different patients.
The second incision should be made posterior and
slightly superior to the pinna of the ear. For the past few
years we have been placing the skin incision longitudi-
nally, following the path of the arteries in the temporo-
occipital region. By doing so, we are more likely to avoid
damage to a major branch of the occipital or retroauricu-
lar artery. Before this, we used a transverse incision. From
either incision it is possible to easily dissect the subgaleal
plane behind the ear toward the direction of the mastoid
(▶Fig. 11.7, ▶Fig. 11.8, ▶ Fig. 11.9).
The ideal valve placement is behind the ear, far enough
away from the ear to prevent problems with the patient’s
spectacles, but close enough to allow a reliable positioning
of the valve parallel to the longitudinal axis of the body.
Later on, this positioning should be confirmed by lateral
Fig. 11.5 Cerebrospinal fluid flows into and is visible in the bore
hole reservoir.
skull radiography (▶ Fig. 11.10, ▶ Fig. 11.11, ▶ Fig. 11.12).
96
Surgical Technique
Fig. 11.6 At the distal end of the catheter (connecting bore
Fig. 11.7 Positioning of the valve (proGAV) behind the ear (left
hole reservoir and valve), cerebrospinal fluid can be taken for
side) coming from a transverse incision. It is important to place the
laboratory tests.
gravitational unit parallel to the longitudinal axis of the body and to
dissect a sufficiently large pouch in the subgaleal retroauricular
plane to allow a smooth positioning of the valve, the gravitational
unit, and the peritoneal catheter without kinking.
Fig. 11.9 The previously used retroauricular skin incision is
Fig. 11.8 Positioning of the valve (proGAV, proximal, adjusta-
reopened, and the (adjustable) gravitational unit is placed
ble) and gravitational unit (proSA, distal, adjustable) in a patient
parallel to the longitudinal axis of the body; none of the
requiring shunt revision.
implants will be positioned directly under the incision, but
between the ear and the incision.
Fig. 11.10 Palpation of the adjustable valve 6 months after
Fig. 11.11 Adjustment of the valve. The valve should be placed
surgery.
far enough from the ear to avoid problems with the patient’s
spectacles and to allow easy adjustment without compressing
or kinking the pinna.
97
Surgical Technique
Fig. 11.13 Cerebrospinal fluid aspiration from the distal end of
the peritoneal catheter, before implantation in a patient with
secondary (posthemorrhagic) normal pressure hydrocephalus.
skin incision behind the ear and the valve should be
between the ear and skin incisions in a well-prepared
pouch.
At this point during the operation, CSF should be
dripping out at the very distal end of the shunt
Fig. 11.12 Lateral radiograph of the skull shows correct
placement of a proGAV valve (round-shaped, proximal, adjust-
system. If not, it should be aspirated or advanced by
able) and the gravitational unit (distal part of the valve,
pumping the reservoir. This is confirmation that there
nonadjustable, placed parallel to the longitudinal axis of the
has been no obstruction thus far in the placement
patient’s body).
process (▶ Fig. 11.13).
Once this is confirmed, there are two ways of placing
the distal catheter in the abdominal cavity. One could use
At this point, in the surgery, the ventricular catheter
a trocar; this has been a safe and reliable procedure in
and the reservoir have been placed. The reservoir allows
our practice in recent years. Results for the use of the tro-
for safe CSF aspiration at any time and secures the
car technique in experienced hands have shown it to be
perpendicular right-angled positioning of the catheter.
safe and, as some authors report, even superior to the
Movement of the proximal catheter after it has been
open laparotomy technique (obstruction rate, infection
placed is almost completely excluded by this measure.
rate, time of surgery).3,4 In our experience, without per-
We close the galea at this point.
forming a clinical study, obstruction and infection rates
are comparable with the open technique; however, the
trocar saves time (10 min); therefore, it shows a lower
11.5.3 Peritoneal Catheter
infection rate, and is less invasive for the patient (smaller
The third incision will be at the umbilicus. Now tun-
skin incision, less dissection), and more convenient for
neling from the abdomen to the retroauricular region
the surgeon.
will be performed. In patients in whom this tunneling
Alternatively, one could use the standard open
cannot be carried out, a fourth incision slightly above
approach with preparation of the subcutaneous fat tis-
the clavicle is necessary. The tunneling is performed
sue and the anterior sheath of the rectus abdominis,
from the supraclavicular to the retroauricular direc-
preparation of the muscle—which we usually do bluntly
tion, and afterward from the supraclavicular region to
using two clamps—and preparation of the posterior
the retroauricular region. After positioning of the valve
fascia of the rectus abdominal muscle, which can be
and the distal catheter in the subcutaneous plane, the
opened using a knife or a pair of small scissors. It is
connection between the catheter coming from the
important that the peritoneum is opened appropriately.
reservoir and the proximal part of the valve can be
We always dissect with the blunt dissector intra-
performed and secured.
abdominally to make it sure that it is opened correctly,
It is of utmost importance to prepare the pouch behind
that the peritoneal cavity has been reached, and that
the ear in such a fashion (i.e., large and wide enough) that
there are no adhesions at that particular site of the
the valve fits into it smoothly, without much further
shunt placement. Afterward, the distal catheter is placed
preparation. The valve can be placed by a slight distal
in the peritoneal cavity. Usually the catheter is secured
pulling of the catheter in the abdominal region. After the
to the posterior wall of the abdominal rectus muscle
valve placement, there should be no hardware under the
using nonabsorbable sutures (▶ Fig. 11.14).
98
Surgical Technique
a
b
Fig. 11.14 (a) Placement of the peritoneal catheter in the peritoneal cavity. (b) Sutures close the peritoneal cavity and at the same time
fix the catheter in place.
In patients with intraperitoneal adhesions, it is benefi-
11.6 Alternatives to the
cial to place the peritoneal catheter under direct laparo-
scopic view. This requires cooperation with an abdominal
VP Shunt
surgeon. An increasing number of publications promote
Alternatives to the VP shunt are ventriculoatrial
(VA)
routine laparoscopic placement (▶ Fig. 11.15, ▶ Fig. 11.16,
shunts and lumboperitoneal
(LP) shunts. The latter
▶ Fig. 11.17).5-9
shunts are indicated and used widely in patients with
Fig. 11.15 Laparoscopic view: the trocar perforates the
Fig. 11.16 Laparoscopic view: the peritoneal catheter is pulled
abdominal wall.
through.
99
Surgical Technique
pseudotumor cerebri. However, some reviews have
reported unacceptably high rates of complications and
revisions for LP shunts.2
VA shunts are used in patients with severe abdominal
scarring following peritonitis or extensive abdominal
surgery. The proximal placement of these shunts is as
described above. The distal catheter can be placed by sur-
gical insertion into the facial vein, found 2 cm below the
mandibular angle, or by direct puncture of the internal
jugular vein. The disadvantage of VA shunts is the risk of
right atrial thrombosis (▶ Fig. 11.18).
References
[1] Choux M, Genitori L, Lang D, Lena G. Shunt implantation: reducing
the incidence of shunt infection. J Neurosurg 1992; 77: 875-880
[2] Kandasamy J, Hayhurst C, Clark S et al. Electromagnetic stereotac-
tic ventriculoperitoneal CSF shunting for idiopathic intracranial
Fig. 11.17 Laparoscopic view: the catheter is positioned in the
hypertension: a successful step forward? World Neurosurg 2011;
abdominal cavity.
75: 155-160, discussion 32-33
[3] Bani A, Telker D, Hassler W, Grundlach M. Minimally invasive
implantation of the peritoneal catheter in ventriculoperitoneal shunt
placement for hydrocephalus: analysis of data in 151 consecutive
adult patients. J Neurosurg 2006; 105: 869-872
[4] Wang GM, Fu SL, Ge PF et al. Use of a new type of trocar for the surgi-
cal treatment of hydrocephalus: a simple and effective technique. J
Int Med Res 2011; 39: 766-771
[5] Khaitan L, Brennan EJ. A laparoscopic approach to ventriculoperi-
toneal shunt placement in adults. Surg Endosc
1999;
13:
1007-1009
[6] Naftel RP, Argo JL, Shannon CN et al. Laparoscopic versus open
insertion of the peritoneal catheter in ventriculoperitoneal shunt
placement: review of 810 consecutive cases. J Neurosurg 2011;
115: 151-158
[7] Roth JS, Park AE, Gewirtz R. Minilaparoscopically assisted placement
of ventriculoperitoneal shunts. Surg Endosc 2000; 14: 461-463
[8] Roth J, Sagie B, Szold A, Elran H. Laparoscopic versus non-
laparoscopic-assisted ventriculoperitoneal shunt placement in
adults. A retrospective analysis. Surg Neurol 2007; 68: 177-184,
discussion 184
[9] Turner RD, Rosenblatt SM, Chand B, Luciano MG. Laparoscopic perito-
Fig. 11.18 Chest X-ray, the tip of the ventriculoatrial shunt can
neal catheter placement: results of a new method in 111 patients.
be seen approximately over the head of the fourth rib.
Neurosurgery 2007; 61 (Suppl): 167-172, discussion 172-174
100
12.1
Rationale for ETV in
Chapter 12
Communicating Hydrocephalus
and NPH
102
Endoscopic Third
Ventriculostomy in Normal
12.2
Technique
103
Pressure Hydrocephalus
12.3
Complications
106
Uwe Kehler
12.4
Outcomes
107
12.5
Summary
107
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
12 Endoscopic Third Ventriculostomy in Normal
Pressure Hydrocephalus
Uwe Kehler
Endoscopic third ventriculostomy
(ETV) is a well-
ous meningitis or infratentorial subarachnoid hemor-
established treatment for obstructive hydrocephalus. In
rhage, or they may be of unknown origin (idiopathic). In
patients with obstructive hydrocephalus, ETV may
these cases, there is still communication between the
replace a shunt system and thus help avoid shunt-related
ventricles and the subarachnoid space/great cistern,
complications, such as shunt infections, shunt disloca-
which means it is still a communicating hydrocephalus.
tions, obstructions, and overdrainage. The development
However, if no real communication exists between the
of endoscopes and instruments, as well as video cameras
great cistern and the prepontine cistern, CSF cannot reach
with high definition (HD) technology, has improved the
the supratentorial reabsorption sites. A perforation at the
safety of endoscopic procedures. However, for a long time
floor of the third ventricle bypasses this infratentorial
the indication was restricted to pure obstructive hydro-
intracisternal obstruction, and does so in exactly the
cephalus, such as aqueductal stenosis, posterior fossa
same way as in aqueductal stenosis. InfinOH is character-
tumors, and fourth ventricle outlet obstructions. The
ized by a downward bulging floor of the third ventricle,
advantages of shunt avoidance were so obvious that a
and a forward-pushed lamina terminalis as a sign of
search began for more indications for ETV. Following this,
pressure difference between the third ventricle and the
ETV was also performed in communicating forms of
surrounding subarachnoid space (▶ Fig. 12.1). The infra-
hydrocephalus, which led to an ongoing debate of
tentorial obstruction cannot be visualized directly, but
whether ETV could be helpful in communicating hydro-
indirect signs are clear, and similar to those of a dam: the
cephalus, even in idiopathic normal pressure hydroceph-
prestenotic/predam spaces (ventricles and great cistern)
alus (iNPH). Today, two extreme practices exist: some
are enlarged, and the poststenotic spaces (prepontine cis-
neurosurgeons simply indicate ETV as a first-line proce-
tern and supratentorial subarachnoid space) are of nor-
dure for all patients with hydrocephalus, while others
mal size or narrower. These signs can be seen clearly in
implant only shunts. However, even though ETV is a safe
thin sagittal T2-weighted magnetic resonance imaging
procedure, it is not completely risk-free
(see below);
(MRI) slices (see ▶ Fig. 12.1). The rationale is confirmed
therefore, a pathophysiologic rationale should explain
by resolution of clinical signs of NPH as well as by resolu-
why ETV could work in communicating forms of hydro-
tion of the bulging membranes after ETV (▶ Fig. 12.2).
cephalus. From the ethical point of view it is critical to
Criticism to this InfinOH subtype is that it is no idiopathic
offer a patient an ETV, without understanding why it
but a secondary NPH. It might be that the prepontine
could work. Presently, several rationales have been devel-
obstructions are caused by preceding events such as mild
oped that make an indication acceptable for ETV, at least
meningitis or even hemorrhage. However, in many
in some cases of communicating hydrocephalus and NPH.
patients these events were probably asymptomatic or not
remembered by the patient; so, from a clinical point of
view (if no cause is found), idiopathic NPH must be
12.1 Rationale for ETV in
assumed. However, the cases of InfinOH account for about
only 10% of iNPH2; thus, with this concept successful ETV
Communicating Hydrocephalus
can be explained only in 10% of iNPH patients (see
▶ Fig. 12.2).
and NPH
The hydrodynamic concept of reduced intracranial
The bulk flow theory provides a good explanation of why
compliance in the development of iNPH has a very differ-
ETV helps in obstructive hydrocephalus such as aqueduc-
ent approach to explaining successful ETV in iNPH. The
tal stenosis: by perforating the floor of the third ventricle
decrease of elasticity and capacitance of cerebral vessels
a communication between the ventricular system—where
leads to reduced cerebral blood volume. The consequence
cerebrospinal fluid
(CSF) is formed—and the sub-
is a reduction of cerebral blood flow and intracranial
arachnoid space—where CSF is reabsorbed—is estab-
compliance, followed by increased intracerebral pulse
lished. The obstruction/aqueductal stenosis is bypassed
pressure. The increased systolic pressure pushes the brain
through this communication.
toward the skull and compresses the periventricular
But, how can we explain a successful ETV in communi-
spaces; this explains the ventricular enlargement with
cating hydrocephalus and even in NPH? One explanation
narrowing of the subarachnoid space.3,4 Opening the floor
could be infratentorial intracisternal obstructions
(or
of the third ventricle allows flow out of the ventricular
infratentorial intracisternal obstructive hydrocephalus
system, reducing the water-hammer effect of the systolic
[InfinOH]).1 These obstructions may be a result of previ-
pulse pressure, and stopping the ongoing pathogenic
102
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
Fig. 12.1 Infratentorial intracisternal obstructive hydrocephalus
(InfinOH) pre-endoscopic third ventriculostomy. The floor of the
Fig. 12.2 Infratentorial intracisternal obstructive hydrocephalus
third ventricle bulges downward and the lamina terminal is pushed
(InfinOH) post-endoscopic third ventriculostomy. The floor of
forward. The normal position is shown with dotted lines. No
the third ventricle is no longer bulging downward nor is the
obstruction of the cerebrospinal fluid pathway can be seen directly.
lamina terminal pushed forward.
mechanism of iNPH. The same effect is realized with a
size of the foramen Monro, and anomalies of the anatomy
shunt, which is even pronounced if a slight overdrainage
of the floor of the third ventricle that could interfere with
is present. If this concept is correct, then ETV should be
a safe perforation (i.e., basilar artery aneurysms, elonga-
effective in iNPH as well as in chronic hydrocephalus,
tions of the vessels, arteriovenous malformations). The
which is thought to have a similar pathogenesis. How-
space between the pons and the clivus should also be
ever, this theory and the results are very controversial
observed to determine whether the space is large enough
(see below).
to perform the ETV.
Usually, a rigid endoscope is introduced in the lateral
ventricle via a frontal paramedian bore hole. The site of
12.1.1 ETV in Shunt Failure
the bore hole should be determined by extension of the
ETV is often discussed as an alternative to shunt revision,
line from the tuber cinereum to the foramen of Monro
but the outcome of ETV usually depends on the cause of
(▶Fig. 12.3). The tuber cinereum is situated between the
hydrocephalus. ETV is very successful in obstructive
mammillary bodies and the infundibulum recess/clivus
hydrocephalus after shunt malfunction and only partially
and represents the position for the perforation of the
successful in patients with communicating hydrocepha-
floor of the third ventricle. The upward extension of
lus.5 When ETV is successful in the latter group this might
the described line toward the skull indicates the site
be explained by the InfinOH and/or by the hydrodynamic
of the bore hole (▶ Fig. 12.3 and ▶ Fig. 12.4). The exact
concept.
position of the bore hole on the patient’s skin can be
determined by measuring the distance from the nasion
(see ▶ Fig. 12.3 and ▶ Fig. 12.4) or, of course, by neurona-
12.2 Technique
vigation. The bore hole should be about 2 cm from the
midline; when coming more from the lateral direction,
ETV is a well-established procedure, and the technique
the rigid endoscope will reach the floor of the third ven-
might differ in only a few minor details. ETV can be per-
tricle on the contralateral side, endangering structures
formed with a rigid or a flexible endoscope. For visualiza-
such as the hypothalamus, oculomotor nerve, and the
tion purposes, the endoscope is connected to a video
posterior communicating artery. With a more medial
camera and the images are seen on a screen located in a
bore hole, there is risk of lesion of a bridging vein or even
position that is comfortable for the surgeon.
of the sagittal sinus. The exact position of the bore hole is
especially important if the foramen of Monro is small.
Incorrect positioning of the bore hole must be compen-
12.2.1 Preoperative Planning
sated by stretching the foramen of Monro with the endo-
For planning the endoscopic procedure, MRI—if possible
scope to reach the above described perforation site at the
—should be available. Accurate planning is necessary to
floor of the third ventricle. If the delicate structures
minimize complications. Imaging should help to find the
around the foramen are pulled or pushed it may lead to
optimal entry point for the endoscope, should show the
its damage and cause neurologic deficits.
103
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
12.2.2 Positioning of the Patient
The patient is placed in a supine position with the head
tilted slightly forward so that the bore hole is almost at
the highest point of the head. This prevents air from
entering the ventricles, which could impair visualization.
12.2.3 Surgical Technique
After preoperative prophylactic antibiotics, skin dis-
infection, and sterile covering, a skin incision and bore
hole trepanation are performed. The dura is opened. The
arachnoid, pia, and cortex are coagulated and incised.
Depending on the endoscope design and size, the endo-
scope sheath, with or without the endoscope, is pushed
gently through the brain toward the ventricle. It is
directed toward the tragus in the sagittal plane and
toward the nasion in the coronal plane. Caution is needed
if the brain and the ventricle walls are stiff: the ventricu-
lar wall may be pushed forward by the blunt endoscope,
Fig. 12.3 Determination of the bore hole site for endoscopic
without the endoscope entering it. A prepuncture with a
third ventriculostomy (ETV). The site of ETV and the foramen of
Cushing needle opens the ventricle wall more easily and
Monro are marked on a sagittal magnetic resonance image
helps the endoscope enter into the ventricle without
(MRI) (black circles). The trajectory linking these structures,
pushing and dislocating the wall of the ventricle.
when extended upward to the skull, determines the entry point
for the endoscope. The distance from the nasion to the entry
Once in the ventricle, the surgery can be continued
point is measured on the sagittal MRI slice, which is then
under visual control. Preferably a 0° endoscope optic is
transferred to the patient’s head.
used. First, orientation is needed: if the foramen of Monro
is not directly in view, the tip of the endoscope must be
moved anteriorly if the plexus is visible. The choroid
plexus leads toward the foramen. If the plexus is not visi-
ble, then the endoscope is in the choroid plexus-free
frontal horn and must be moved posteriorly.
The foramen of Monro (▶ Fig. 12.5) is bounded ante-
riorly and medially by the fornix and laterally by the
Fig. 12.4 Localization of the bore hole for endoscopic third
ventriculostomy: 2 cm paramedial, approximately at the coronal
Fig. 12.5 Foramen of Monro with the choroid plexus in the
suture; the exact distance is measured from the nasion (orange
posterior part and the fornix, which together form the anterior
line), determined by the sagittal magnetic resonance image.
and medial borders of the foramen.
104
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
x
*
*
Fig. 12.6 Translucent floor of the third ventricle with infundib-
Fig. 12.7 Opaque floor of the third ventricle. Mammillary
ular recess (←), basilar artery bifurcation (▲), perforating vessels
bodies (*) and infundibular recess (x) can be identified. The
from the P1 segments and mammillary bodies (➧). The point of
structures at risk of damage, especially the basilar artery, cannot
planned endoscopic third ventriculostomy is marked with an
be seen.
asterisk.
thalamus. The posterior border is covered by the cho-
fied. If the floor is translucent, then it is easy to go just
roid plexus, which continues to the roof of the third
anterior to the basilar artery tip (▶ Fig. 12.6). If the floor
ventricle. At the lateral boundary, the thalamostriate
is opaque (▶ Fig. 12.7), then one has to be aware of the
vein can be identified, which drains toward the inter-
underlying anatomy
(▶Fig.
12.8). Just ahead of the
nal cerebral vein at the roof of the third ventricle. The
mamillary bodies, the basilar artery and the P1 segment
septal vein is visible at the medial site.
of the posterior cerebral artery are situated, with perfo-
If an optimal entry point has been chosen, the floor of
rating vessels traveling to the brainstem.6 In the lateral
the third ventricle with the mammillary bodies and the
part of the floor of the third ventricle, the oculomotor
infundibular recess can be seen through the foramen of
nerve and the posterior communicating artery are
Monro. The endoscope is then pushed through the fora-
located, and the infundibular recess with the pituitary
men of Monro to well inside the third ventricle. Next, the
gland is situated anteriorly. Going a little bit deeper
side for perforating into the prepontine cistern is identi-
along the clivus, the abducens nerve is located just some
Fig. 12.8 Floor of the third ventricle, with
structures at risk of damage during perfo-
Chiasma
ration. Post., posterior; comm., communi-
Infundibular recess
cating.
Carotid artery
Post. comm. artery
Oculomotor nerve
Clivus
Perforation site for ETV
Post. cerebral. artery
Basilar artery
Mammillary body
Perforators
105
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
millimeters beside the midline. The surgeon must be
aware of all the structures before perforating the floor of
the third ventricle because they could be damaged by the
perforating catheter. The optimal site of perforation is just
in front of the basilar artery tip and just behind the clivus,
clearly in the midline.
The perforation itself can often be done bluntly—for
instance, with a balloon catheter, just pushing it for-
ward along the clivus. In some cases the floor can be
very tight, so sharp instruments may be required for
the perforation.7
The little hole that has been made should be
enlarged to prevent reclosure. This can be performed
with a balloon catheter, which will be blown up
(▶ Fig. 12.9) or by splaying a forceps when introduced
in the little hole.
There should be an unobstructed view toward the basi-
lar artery, and the perforating vessels, as well as to the
brainstem itself. If not, then the perforation might have
been done in the subdural space just anterior to the
Fig. 12.10 Stoma achieved in an opaque floor of the third
arachnoid. In this case, a perforation through the arach-
ventricle.
noid/Liliequist membrane is necessary to achieve the
right connection between the ventricle and the sub-
arachnoid space.
The size of the stoma is not clear. From a practical point
12.3 Complications
of view, it should be as large as possible without taking
Experience with ETV has grown enormously in the last
any unnecessary risks. So, a size of 3 to 4 mm can be
decade and it has been shown to be a simple and safe
achieved safely in most cases (▶ Fig. 12.10). Rinsing usu-
procedure.8,9 However, the potential risks of the proce-
ally stops the bleeding.
dure are high and may lead to temporary or even perma-
The ETV is then finished by removing the endoscope
nent neurological deficits. Therefore, everything must be
and closing the bore hole with bone wax and skin
done to reduce these complications. Knowledge of spe-
sutures.
cific complications and the understanding of their causes
are necessary to avoid or at least reduce them. Currently,
there are no publications that focus specifically on com-
plications of ETV performed in patients with NPH. How-
ever, the frequency of complications of ETV is probably
related to the procedure itself rather than the diagnosis
(obstructive or communicating hydrocephalus). More
rigid arteriosclerotic vessels and a rigid floor of the
third ventricle, which appear to be more frequent in
patients with NPH than in young patients with aque-
ductal stenosis, may lead to a slightly higher number
of complications.
Moreover, in patients with small ventricles or even slit
ventricles—which are not seen in NPH—the risk is higher.
Complications might be a result of insufficient experi-
ence of the surgeon with the endoscopic techniques and
insufficient presurgical planning. Several authors have
demonstrated a reduction in the number of complica-
tions with increasing experience of the surgeon.8,9
Theoretically, all structures around the endoscope are at
risk of damage: Introducing the endoscope into the brain
toward the ventricle may be hindered by a rigid cortex—
which is found especially in older patients. The cortex may
Fig. 12.9 The hole in the floor of the third ventricle is dilated
be pushed downward by the endoscope stretching the
blowing up the balloon of a Fogarty catheter.
bridging veins and even tear them.
106
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
The route of the endoscope must be planned and
that “CSF shunting with a programmable valve is superior
checked throughout the ventricular puncture. An
to treatment with endoscopic third ventriculostomy with
inappropriate direction might prevent the surgeon from
choroid plexus coagulation in idiopathic NPH.”20
entering the ventricle in the correct place. If the direction
In conclusion, ETV for iNPH without selection of sub-
is too lateral it can damage the thalamus or even the
types, such as InfinOH, cannot be generally recom-
internal capsule, causing hemiparesis. If it is too medial it
mended. The success rate of ETV for iNPH is far worse
can damage the anterior cerebral artery and its branches,
than for obstructed hydrocephalus. The risk of recurrence
or it can lead to the contralateral ventricle.
seemed to be higher in patients with NPH.14,17 According
Inside the lateral ventricle, the endoscope must be
to the hydrodynamic theory a much higher success rate
pushed through the foramen of Monro. All structures of
would be expected. Alternatively, preselected patients
the foramen are potentially at risk of damage, particularly
with iNPH and infratentorial intracisternal obstructions
if the entry point of the bore hole was not adequate. The
may benefit from ETV; however, there should be a discus-
site of the bore and the foramen of Monro determine
sion as to whether these cases are really idiopathic cases.
where the rigid endoscope reach the floor of the third
These obstructions may be due to a former clinically not
ventricle. If the endoscope does not reach the planned
apparent infection or hemorrhage, which would classify
perforation site it must be moved around in the third
the NPH as secondary NPH.
ventricle. This causes stretching of the foramen of Monro
One should be extremely cautious not to be satisfied
with its sensitive structures like the fornix of the thala-
with partial recovery of a patient after ETV and withhold
mostriate vein. Lesions to these structures may lead to
potential further treatment that could improve the condi-
intraventricular hematoma or memory deficits.
tion of the patient. If the recovery after ETV is not substan-
The most dangerous part of ETV is perforating the floor
tial, then further diagnostic evaluation by a spinal tap test
of the third ventricle itself. The basilar artery and the per-
or external lumbar drainage is recommended to see if fur-
forating vessels might be injured, which might be life
ther improvement is possible. If so, then ETV has failed at
threatening. Of course, all structures on the floor of the
least partially, and shunt placement should be performed.
third ventricle are at risk (see ▶ Fig. 12.8).
Reported complications include venous and arterial
12.5 Summary
bleeding, with intracerebral and intraventricular hemato-
mas and subarachnoid hemorrhages, injuries of the thala-
The role of ETV in iNPH is yet to be determined. Although
mus with hemiparesis, fornix contusions,10 and lesions of
the hydrodynamic theory should explain at least partial
the hypothalamus with diabetes insipidus. CSF leaks (3.1%)
improvement after ETV in most patients, the results of
and meningitis (2.3%) are seen more often.9 Injuries of cra-
the first randomized trial contradict this theory and show
nial nerves have been described only rarely.8,11 Procedure-
that ETV has an insufficient effect in iNPH.20 The InfinOH
related complications are seen in about
9% to 10%,8,9
concept may explain the success of ETV in a subgroup of
whereas most complications are asymptomatic or transient;
patients only. The differentiation between chronic hydro-
permanent and severe complications are rare. Procedure-
cephalus (e.g., aqueductal stenosis) and iNPH remains
related mortality in trained hands is less than 1%.8,9
problematic and the two cannot be differentiated clini-
cally. NPH is a far too complex condition and not under-
stood well enough. It becomes difficult to determine who
12.4 Outcomes
will and who will not benefit from ETV. However, we
need to provide a rationale as to why and when ETV will
The outcome of ETV in obstructive hydrocephalus is
help in the treatment of iNPH, otherwise we will expose
favorable (up to 95%)12; nevertheless, successful ETV does
patients to unnecessary risk. The final results of the pro-
not necessarily mean cure—even in obstructive hydro-
spective randomized study20 will give us an answer as to
cephalus. There are various reports of late reclosure of
whether ETV is an acceptable option for treatment of
the stoma,13,14 and some with sudden deterioration.15
iNPH. Presently, ETV cannot be generally recommended
Therefore, similarly to a ventriculoperitoneal (VP) shunt,
as a first-line treatment for iNPH, but it is acceptable in
regular follow-up is recommended.
some subgroups (InfinOH) and in those patients who do
There are only a few papers reporting the success rate of
not consent to shunt therapy.
ETV in NPH, and the results are somewhat controversial,
varying from 21% to 69%.16-18 More studies are planned or
are in progress and are necessary before a final recommen-
References
dation either for or against ETV can be provided. The first
randomized controlled study that compared ETV with
[1] Kehler U, Gliemroth J. Extraventricular intracisternal obstructive hydro-
cephalus—a hypothesis to explain successful 3rd ventriculostomy in
plexus coagulation with VP shunts with a programmable
communicating hydrocephalus. Pediatr Neurosurg 2003; 38: 98-101
valve was stopped early due to the neuroendoscopic treat-
[2] Kehler U, Herzog J. Infratentorial intracisternal obstructive hydrocepha-
ment lacking efficacy in patients with iNPH.19,20 The con-
lus (InfinOH): how often is this subtype, which can be treated endo-
clusion of that well-designed and well-executed study was
scopically, among idiopathic normal pressure hydrocephalus (iNPH)?
107
Endoscopic Third Ventriculostomy in Normal Pressure Hydrocephalus
[13]
Dusick JR, McArthur DL, Bergsneider M. Success and complication
rates of endoscopic third ventriculostomy for adult hydrocephalus: a
series of 108 patients. Surg Neurol 2008; 69: 5-15
[3]
Greitz D. Radiological assessment of hydrocephalus: new theories
[14]
Fabiano AJ, Doyle K, Grand W. Delayed stoma failure in adult commu-
and implications for therapy. Neurosurg Rev 2004; 27: 145-165, dis-
nicating hydrocephalus after initial successful treatment by endo-
cussion 166-167
scopic third ventriculostomy: case report. Neurosurgery 2010; 66:
[4]
Greitz D. Paradigm shift in hydrocephalus research in legacy of
E1210-E1211, discussion E1211
Dandy’s pioneering work: rationale for third ventriculostomy in com-
[15]
Mobbs RJ, Vonau M, Davies MA. Death after late failure of endoscopic
municating hydrocephalus. Childs Nerv Syst 2007; 23: 487-489
third ventriculostomy: a potential solution. Neurosurgery 2003; 53:
[5]
O’Brien DF, Javadpour M, Collins DR, Spennato P, Mallucci CL. Endo-
384-385, discussion 385-386
scopic third ventriculostomy: an outcome analysis of primary cases
[16]
Gangemi M, Maiuri F, Naddeo M et al. Endoscopic third ventriculos-
and procedures performed after ventriculoperitoneal shunt mal-
tomy in idiopathic normal pressure hydrocephalus: an Italian multi-
function. J Neurosurg 2005; 103 (Suppl): 393-400
center study. Neurosurgery 2008; 63: 62-67, discussion 67-69
[6]
Fabiano AJ, Leonardo J, Grand W. Posterior cerebral artery P1 seg-
[17]
Hailong F, Guangfu H, Haibin T et al. Endoscopic third ventriculos-
ment at the stoma during endoscopic third ventriculostomy in adults.
tomy in the management of communicating hydrocephalus: a pre-
J Neurol Neurosurg Psychiatry 2010; 81: 374-378
liminary study. J Neurosurg 2008; 109: 923-930
[7]
Kehler U, Gliemroth J, Knopp U, Arnold H. How to perforate safely a
[18]
Longatti PL, Fiorindi A, Martinuzzi A. Failure of endoscopic third ven-
resistant floor of the 3rd ventricle? Technical note. Minim Invasive
triculostomy in the treatment of idiopathic normal pressure hydro-
Neurosurg 1998; 41: 198-199
cephalus. Minim Invasive Neurosurg 2004; 47: 342-345
[8]
Schroeder HWS, Niendorf WR, Gaab MR. Complications of endoscopic
[19]
Edwards R, Bunnage M, O'Brien D, Luciano M, Pople I. A prospective,
third ventriculostomy. J Neurosurg 2002; 96: 1032-1040
randomised, controlled trial to evaluate the efficacy and safety of
[9]
Sacko O, Boetto S, Lauwers-Cances V, Dupuy M, Roux FE. Endoscopic
endoscopic choroid plexus coagulation with third ventriculostomy in
third ventriculostomy: outcome analysis in
368
procedures. J
the treatment of idiopathic normal pressure hydrocephalus
Neurosurg Pediatr 2010; 5: 68-74
[ISRCTN29863839] Cerebrospinal Fluid Res 2004; 1 (Suppl 1): S58
[10]
Kehler U, Regelsberger J, Gliemroth J. The mechanism of fornix lesions
[20]
Edwards R, Bunnage M, O'Brien D, Luciano M, Pople I. Results of a
in 3rd ventriculostomy. Minim Invasive Neurosurg 2003; 46: 202-204
prospective, randomised, controlled trial to evaluate the efficacy and
[11]
Buelens E, Wilms G, van Loon J, van Calenbergh F. The oculomotor
safety of endoscopic choroid plexus coagulation with third ventricu-
nerve: anatomic relationship with the floor of the third ventricle.
lostomy in the treatment of idiopathic normal pressure hydrocepha-
Childs Nerv Syst 2011; 27: 943-948
lus [ISRCTN29863839], presented at the IFNE Interim Meeting, Tokyo,
[12]
Spennato P, Tazi S, Bekaert O, Cinalli G, Decq P. Endoscopic third ven-
triculostomy for idiopathic aqueductal stenosis. World Neurosurg
2013; 79 (Suppl): e13-e20
108
13.1
Black Grading Scale
110
Chapter 13
13.2
Index for Postoperative
Scales and Scores
Improvement
110
Ullrich Meier
13.3
Stein-Langfitt Scale
111
13.4
Kiefer Grading Scale
111
13.5
NPH Recovery Rate
111
13.6
Comorbidity Index
112
Scales and Scores
13 Scales and Scores
Ullrich Meier
Clinical classification of special clinical pictures should,
not consider the specific symptoms of the clinical
on the one hand, take into account the specific symptoms
picture of NPH. Taking into account these two
and, on the other, provide or enable an evaluation of the
negative attributes, the Black Grading Scale for shunt
individual course of the disease, while also allowing for
assessment1 can only be conditionally recommended
an interindividual comparison. An assessment of the indi-
for assessing the individual course in patients with
vidual symptoms, according to their importance for the
iNPH (▶ Table 13.1).8
patient and bearing in mind the pathophysiologic, occu-
pational medicine, and psychosocial aspects is, in this
context, indispensable. With regard to practicality in the
13.2 Index for Postoperative
context of everyday hospital routine, clinical grading
Improvement
must be constructed in a lucid manner, as far as possible
without inclusion or exclusion criteria, as well as being
From postoperative observations of the clinical course of
easy to use. From this perspective, we investigated three
50 patients with iNPH in whom, following a careful pre-
different scales for assessing the course of disease in
selection during 1989 to 1994, a shunt operation was car-
patients with normal pressure hydrocephalus (NPH): the
ried out in Freiburg, Germany, Krauss et al5 reported the
Black Grading Scale for shunt assessment,1 the clinical
calculation of an index intended to clarify the extent of
grading scheme of Kiefer et al.2-4 and the index for the
postoperative improvement in findings. In this, an index
postoperative improvement in findings for patients with
of 0 indicates no or minimal improvement, and an index
idiopathic normal pressure hydrocephalus
(iNPH) of
of 1 indicates an excellent improvement in findings for all
Krauss et al.5,6 The description of the Stein-Langfitt scale7
cardinal symptoms. To calculate the index, the post-
was taken from the literature.
operative changes in three cardinal symptoms are
evaluated separately on a three-point scale. The index is
produced by the quotients of the actual improvement in
13.1 Black Grading Scale
findings and the maximum possible improvement in
findings. In patients with two preoperative cardinal
The Black Grading Scale for shunt assessment1 grades
symptoms, an index between 0/4 and 4/4 is possible, and
different forms of clinical improvement following a
in patients with three cardinal symptoms, an index
shunt operation, but it does not allow any distinction
between 0/6 and 6/6 is possible.5,6 The scientific approach
between an unchanged course of the disease and clini-
to the calculation of the index for the postoperative
cal worsening. This fact has a negative consequence in
improvement in findings in patients with iNPH5 is similar
a comparative consideration of patient groups on dif-
to that of the clinical grading of Kiefer et al.4 In the latter,
ferentiating the results of follow-up. A positive attri-
in addition to the cardinal symptoms for NPH, headaches
bute of the Black Grading Scale for shunt assessment1
and symptoms of dizziness are also evaluated, as well as a
is a clear subdivision of the scale into six groups,
clear gradation of the individual symptoms, which is
which are defined as excellent, good, gradual, time-
undertaken according to their degree of severity. For this
limited improvement, bad, and death. In this way,
reason, we believe that the index for postoperative
the gradation is simple and can be used without any
improvement in findings for patients with iNPH proposed
inclusion or exclusion criteria.
8
by Krauss et al5 is unsuitable for clinical practice.
The Black Grading Scale for shunt assessment1
is
well suited for use in patients with pressure hydro-
cephalus. For this patient group, the individual symp-
?
toms do not so much dominate in the clinical course
Table 13.1 Black Grading Scale for shunt assessment1
following a shunt operation, but the results of the
Assessment
Level of activity following the shunt
operation as such are to be taken into account without
operation
consideration of the preoperative condition of the
Very good
Achieved the same level of activity as
patient. For interindividual comparison of patients in
before the illness, without restrictions
relation to the course of the disease with internal
Good
Achieved the same level of activity as
hydrocephalus, the Black Grading Scale for shunt
before the illness, with restrictions
assessment1
is, in our experience, well suited.8,9
In
Moderate
Improvement, but cannot carry out a job
addition to providing an undifferentiated summary
when the course of the disease is unchanged and
Moderate/temporary
Temporary improvement
when there is worsening of the clinical symptoms, the
Bad
No improvement or worsening
Black Grading Scale for shunt assessment1 also does
Death
Died within 6 weeks following the
operation or because of the operation
110
Scales and Scores
??
??
Table 13.2 Stein-Langfitt scale7
Table 13.3 Clinical grading in normal pressure hydrocephalus4
Grade
Clinical condition
Score
Symptoms
0
No neurological deficit; the patient is able to work
Mental
1
Slight neurological deficit; the patient is able to get
0
No deficits
home alone
1
Forgetful, disorders of concentration
2
At times, the patient requires to be cared for at home
4
Apathic or partly oriented
3
Care of the patient at home is necessary, despite the
6
Completely disoriented, impairment of higher
patient having some remaining residual ability
cortical functions
4
The patient is not able to take care of himself/herself
Gait disorder
0
No deficits or deficits demonstrable only with
13.3 Stein-Langfitt Scale
special test
2
Straddle-legged, ataxic, but certain gait disorders
With the Stein-Langfitt scale,7 patients are judged both
4
Difficulties in movement: mobility aids required
preoperatively as well as postoperatively according to
5
Only a few steps with the aid of a person possible
their clinical condition and, in particular, to their residual
abilities. The Stein-Langfitt scale7
does not take into
6
Unable to walk
account the specific symptoms of the clinical picture of
Incontinence
iNPH. Since most patients with iNPH are found to be in
0
Not present
stages 1 or 2, a substantial lack of discrimation power has
1
Urge incontinence
to be stated compared to other scales. In addition, there
3
Intermittent urinary incontinence (e.g., at night)
are problems with the allocation of grades. For example,
4
Long-term urinary incontinence
a patient with mild gait ataxia and urinary incontinence
6
Urinary and fecal incontinence
would be allocated to both grades 1 and 2. Consequently,
Headache
the Stein-Langfitt scale7 can only be conditionally recom-
mended for assessing the individual course in patients
0
Not present
with iNPH (▶ Table 13.2).8
1
Intermittent or permanent headache which does
not influence the activity of daily life
4
Permanent severe headaches
13.4 Kiefer Grading Scale
Dizziness
0
Not present
The clinical grading of NPH by Kiefer et al4
takes
into account the severity of the individual symptoms:
1
Intermittent dizziness appearing under stress or
spontaneous dizziness
dementia, gait disorders, incontinence, headache symp-
tomatology, and symptoms of dizziness (▶ Table 13.3).
Because there is an evaluation of symptoms of headache
incontinence, headache, and dizziness) interindividual
and dizziness, which appear specifically in the initial
comparisons should not be made. For interindividual
stage in patients with NPH, depending on the clinical
comparison of the disease courses, a separate assessment
triad
(Hakim triad: dementia, gait ataxia, and urinary
of individual symptoms or the NPH recovery rate accord-
incontinence), it yields additional information. At the
ing to Meier8-10 is recommended (Eq. 13.1).
same time, the symptoms of the Hakim triad—with up to
six points—are graded higher than the secondary symp-
toms of headache and dizziness, with up to three or four
13.5 NPH Recovery Rate
points. In this way, a patient who shows only a subtle
Using the results of clinical grading for NPH according to
gait disorder as an individual symptom can be allocated
Kiefer et al2,4, Meier8,9 suggested an NPH recovery rate for
zero points; in contrast, a patient with all five symptoms
use in interindividual comparisons:
and maximal expression of the symptoms will be allo-
cated 25 points. The clinical grading for NPH according to
Kiefer et al4 is, in our experience, well suited for the indi-
NPH recovery rate
vidual patient’s description of the disease course. Using
NPH gradingpostoperative NPH grading
preoperative
classification of the grade for the individual symptoms in
¼
10
NPH grading according to Kieferpreoperative
the sequence cited (e.g., M1-G2-I1-K1-S1), the course of
ð22Þ
the individual symptoms, even with catamnestic exami-
nations, can be described in addition to the complex
The NPH recovery rate shows good correlation with the
course of the disease (six points). Because it is assessing
Black Grading Scale for shunt assessment, from the spe-
five very different symptoms (dementia, gait disorder,
cific perspective of the clinical picture of iNPH with the
111
Scales and Scores
??
??
Table 13.4 Comparison of the Black Grading Scale for shunt
Table 13.5 Comorbidity index2
assessment1 and NPH recovery rate8
Risk factors
1 Point
2 Points
3 Points
Black Gra-
Description
NPH recovery
Vascular
Hypertension
Diabetes mellitus
ding Scale
rate (points)
Aortofemoral
Peripheral vascu-
Excellent
Same level of activity as before
≥ 7.5
bypass
lar occlusion
the disease
Stent
Good
Slight limitations
≥5
Cerebro-
Posterior circu-
Vascular ence-
Cerebral
vascular
lation insuffi-
phalopathy
infarction
Fair
Gradual improvement
≥3
ciency
Transient ische-
Transient
Temporary improvement
≥2
Internal carotid
mic attack/pro-
Poor
No change or worse
<2
artery stenosis
longed reversible
ischemic neuro-
logical deficit
Black Grading Scale for shunt assessment.1 Our experi-
Cardiac
Arrhythmia
ence with the NPH recovery rate confirms the practice-
Valvular disease
oriented approach, specifically when comparing different
Heart failure/
investigation groups of patients with iNPH or different
stent
research groups (▶ Table 13.4).8
Aortocoronary
bypass
Infarction
13.5.1 Conclusion
The clinical grading for NPH proposed by Kiefer et al4
The most frequent comorbidity in this con-
reported.11
is a practice-oriented score for assessment of the indi-
text, with a value of 78% (referred to all comorbidities
vidual course in patients with iNPH. The NPH recovery
observed), is vascular encephalopathy based on a cere-
rate proposed by Meier8 can be used for interindividual
brovascular insult; Parkinson disease or Parkinsonoid has
comparisons.
a value of 10%, as does nonverifiable but clinically sus-
pected Alzheimer disease.15 To be able to separate the
clinical picture of iNPH from other dementia syndromes,
13.6 Comorbidity Index
Golomb et al12
and Savolainen et al13
for example,
As with other medical specialist areas, there is a need for
initiated investigations of the coincidence of Alzheimer
interdisciplinary consideration of one’s own specialist
disease. Likewise, centrally determined disorders of
area for two important reasons: (1) the possibility must
movement represent an overlapping entity. In our
be excluded that the symptoms encountered can be
research group, we also found that 8% of patients had
traced back to other diseases, and so cannot be improved
Parkinson disease.9,10
Exclusion of advanced cerebro-
by the specific therapy; (2) it is important to recognize
vascular disease represents at least one of the main
situations in which the course of the disease is so
reasons for invasive diagnosis of iNPH.14
unfavorably influenced by secondary diseases that no
Quantification of comorbidities is important in the
improvement is to be expected from the specific therapy.
clinical context because of the risk factors involved. A
Both considerations represent a contraindication for sur-
systematic evaluation was performed by Kiefer et al2 in
gical therapy.
the CMI they presented in 2006, which assigns points to
Patients with iNPH are usually elderly individuals. For
frequently occurring secondary diseases and thus allows
this reason, there are often multiple morbidities. Conse-
the empirical determination of a threshold, and signifi-
quently, secondary diseases influence the quality of life in
cantly reduces the probability of a good to very good out-
these patients. Therefore, the question of whether using
come. The indirect correlation between outcome and CMI
comorbidity index (CMI) prognostic statements about the
has been clearly explained.10 Accordingly, among patients
course of the disease following a shunt operation in
with a CMI of 0 or 1 point, 67% showed an excellent out-
patients with iNPH is possible must be investigated, and
come; whereas of those with a CMI of 6 to 8 points, 45%
whether comorbidity has a predictive value.
showed an unsatisfactory course of the disease. Kiefer et
Kiefer et al2
introduced an evaluation of secondary
al2
established a CMI of 3 points as the cut-off point
diseases according to their clinical significance for NPH.
between patients with a statistically favorable outcome
By adding points according to the presence of secondary
and those with a tendency to a worse outcome. This value
diseases, a CMI of 0 to a maximum of 23 points can be
has also been confirmed in other publications.2,10 The
calculated (▶ Table 13.5).
proportion of patients with a favorable or very favorable
Various authors have reported an average of 43% of
outcome was 80% in the group with a CMI of 3 points.
patients with comorbidities in NPH.11-15 Accordingly, a
Only 10 of the 31 patients (32%) with a CMI of more than
cerebrovascular insufficiency in 45% of patients has been
3 points achieved such an improvement.
112
Scales and Scores
For patients with iNPH, comorbidity represents a
control group: a comparative study. Neurosurgery 1997; 40: 491-495,
discussion 495-496
statistically significant predictor
(sensitivity
80% and
[7]
Stein SC, Langfitt TW. Normal-pressure hydrocephalus. Predicting
specificity
68%) with reference to the course of the
the results of cerebrospinal fluid shunting. J Neurosurg 1974; 41:
disease following a shunt operation. A CMI of 0 to 3 points
463-470
indicates a good prognosis following a shunt operation.
[8]
Meier U. The grading of normal pressure hydrocephalus. Biomed
With a CMI of more than 3 points, the indication for a
Tech (Berl) 2002; 47: 54-58
[9]
Meier U, Kiefer M, Sprung C. Normal pressure hydrocephalus: pathol-
shunt operation should be established carefully. Successful
ogy, pathophysiology, diagnostics, therapeutics and clinical course.
therapy is not expected in those with a CMI greater than
Erwitte: PVV Science Publications; 2003
6 points, because of secondary diseases.
[10]
Meier U, Lemcke J. The influence of co-morbidity on the post-
operative outcome of patients with idiopathic normal-pressure
hydrocephalus (iNPH). Acta Neurochir Suppl 2008; 102: 141-144
References
[11]
Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
lus Study: the role of cerebrovascular disease. J Neurosurg 1999; 90:
[1] Black PM. Idiopathic normal-pressure hydrocephalus. Results of
221-226
shunting in 62 patients. J Neurosurg 1980; 52: 371-377
[12]
Golomb J, Wisoff J, Miller DC et al. Alzheimer’s disease comorbidity
[2] Kiefer M, Eymann R, Steudel WI. Outcome predictors for normal-
in normal pressure hydrocephalus: prevalence and shunt response. J
pressure hydrocephalus. Acta Neurochir Suppl (Wien) 2006; 96:
Neurol Neurosurg Psychiatry 2000; 68: 778-781
364-367
[13]
Savolainen S, Paljärvi L, Vapalahti M. Prevalence of Alzheimer’s dis-
[3] Kiefer M, Eymann R, Meier U. Five years experience with gravitational
ease in patients investigated for presumed normal pressure hydro-
shunts in chronic hydrocephalus of adults. Acta Neurochir (Wien)
cephalus: a clinical and neuropathological study. Acta Neurochir
2002; 144: 755-767, discussion 767
(Wien) 1999; 141: 849-853
[4] Kiefer M, Eymann R, Komenda Y, Steudel WI. Ein Graduierungssys-
[14]
Tullberg M, Mansson JE, Fredman P et al. CSF sulfatide distinguishes
tem für den chronischen Hydrozephalus. Zentralbl Neurochir 2003;
between NPH and subcortical ateriosclerotic encephalopathy. J
64: 109-115
Neurol Neurosurg Psychiatry 2000; 69: 74-81
[5] Krauss JK, Droste DW, Mergner T. Der idiopathische Normaldruckhy-
[15]
Tullberg M, Månsson JE, Fredman P et al. CSF sulfatide distin-
drozephalus. Dtsch Arztebl 1997; 94: A-589-A-595
guishes between normal pressure hydrocephalus and subcortical
[6] Krauss JK, Regel JP, Vach W et al. White matter lesions in patients with
arteriosclerotic encephalopathy. J Neurol Neurosurg Psychiatry
idiopathic normal pressure hydrocephalus and in an age-matched
2000; 69: 74-81
113
14.1
Organizing Follow-up
Chapter 14
Examinations
116
Follow-up Management
14.2
Follow-up Intervals
116
of Idiopathic Normal Pressure
14.3
Should Follow-ups End?
117
Hydrocephalus
Ullrich Meier
14
Follow-up Management of Idiopathic Normal Pressure Hydrocephalus
14 Follow-up Management of Idiopathic Normal
Pressure Hydrocephalus
Ullrich Meier
Following surgery, patients with idiopathic normal pressure
should be carried out to monitor significant changes
hydrocephalus (iNPH) constitute a very special clientele.
in ventricle size and signs of overdrainage, such as
The vast majority are limited in their mobility, orientation,
extracerebral efflorescence. The valve setting may be
self-perception, and outlook. To properly follow up with
adjusted at the
3-month follow-up period if the
patients who have iNPH, the physician providing care must
department has a schedule of planned readjustments;
deal with these problems.
for example, to compensate the intraoperative loss of
CSF or to allow the brain to make a stepwise adapta-
tion to the shunt.
14.1 Organizing Follow-up
In our department, we customarily reset gravitational
valves from an initial value of 70 to 50 mm H2O after
Examinations
3 months.
iNPH is a life-long disease. Shunt insertion can provide
permanent relief from symptoms, but it cannot produce
14.2.2 Six Months
healing in terms of eliminating the chain of causes of the
disease, which are currently unknown. Therefore, shunt
The 6-month follow-up milestone period should establish
surgery is the first step in management, not the first step
whether the initially selected opening pressure is appro-
in healing.
priate for the individual patient or not.
This management must be continued for the duration
If a patient with iNPH shows no improvement with
of the disease. After receiving a shunt, the patient should
a relatively high valve setting (e.g., ≥ 100 mm H2O), we
be included in a life-long, follow-up program. The follow-
would not expect an improvement in the further
up program should be organized in such a way that there
course of the disease without changing the opening
are appointments agreed to in advance, with the option
pressure.
of additional ones being available should changes in
Another CT scan should be carried out to detect over-
symptoms require them.
and underdrainage complications. The Evans index
should be measured and recorded at every follow-up
examination. A change in the Evans index with no corre-
14.2 Follow-up Intervals
sponding change in the clinical symptoms should never
lead to a resetting of the valve in patients with iNPH. The
Most authors recommend that follow-up examinations
clinical symptoms rank first!
should take place at 1 or 3, 6, and 12 months after shunt
In our department, we evaluate whether a valve
surgery. Our view is that three follow-up examinations
adjustment to 30 mm H2O could improve the patient’s
should take place within the first year.1,2
symptoms after 6 months. This should be carried out,
in particular, if the patient reports an improvement
14.2.1 One/Three Months
after resetting at 3 months which then disappears in
the following weeks.
The first examination should be arranged relatively soon
after the shunt implantation, for example, after 1
or
3 months. The aim of this follow-up examination is to iden-
14.2.3 Twelve Months
tify complications that may be linked to surgery, such as:
● Impaired healing
The last routine CT scan should be made 12 months after
● Mild overdrainage complications not warranting con-
surgery. The decision to carry out further CT scans should
sultation with a physician
depend on the patient’s clinical symptoms. If marked
● Dislocation of the abdominal catheter
changes in the clinical symptoms are not seen, then fur-
● Underdrainage complications
ther follow-up examinations can be limited to the clinical
● Other complications
examination.
Analogous to the follow-up at 6 months, a secondary
The intraoperative loss of cerebrospinal fluid
(CSF)
deterioration of the specific symptoms after an improve-
will certainly be compensated 3 months after surgery.
ment following the
6-month resetting will require
The first signs that the shunt is effective will then
another resetting
(e.g., to
10 mm H2O [gravitational
become apparent. A computed tomography (CT) scan
valves]; ▶ Table 14.1).
116
Follow-up Management of Idiopathic Normal Pressure Hydrocephalus
?
Table 14.1 Follow-up scheme for management of idiopathic normal pressure hydrocephalus
Intervention
Follow-up interval
1 or 3 months
6 months
12 months
> 12 months
Valve setting scheme
From 70 to
From 50 to 30 mm H2O
From 30 to 10 mm H2O
Resetting, conforming
(suggestion applies to
50 mm H2O
No resetting when there is
when there are persisting
to requirements
gravitational valves)
complete relief of the symptoms
symptoms
CT scan required
Yes
Yes
Yes
No
14.3 Should Follow-ups End?
References
We suggest further follow-up examinations on an annual
[1] Meier U, Lemcke J, Al-Zain F. Clinical experience in the treatment of idi-
opathic normal-pressure hydrocephalus using the programmable grav-
basis. CT scans are not necessary if the patient does not
ity-assisted valve (proGAV Aesculap®). Neurosurg Q 2007; 17: 52-55
show new symptoms. In our opinion, there is no reason
[2] Lemcke J, Meier U. Improved outcome in shunted iNPH with a combi-
to stop the follow-up program for patients at any point in
nation of a Codman Hakim programmable valve and an Aesculap-
the future course of the disease.
Miethke ShuntAssistant. Cent Eur Neurosurg 2010; 71: 113-116
117
15.1
Intraoperative Complications
120
Chapter 15
15.2
Infections
121
Complications
15.3
Postoperative Complications
128
Michael J. Fritsch, Uwe Kehler,
15.4
Shunt Malfunction: Shunt
Johannes Lemcke, Ullrich Meier
Revisions
131
15
Complications
15 Complications
Michael J. Fritsch, Uwe Kehler, Johannes Lemcke, Ullrich Meier
the surgical technique. Intraoperative radiography and
15.1 Intraoperative
positioning of the tip of the ventricular catheter in the
Complications
atrium, while strictly avoiding contact with the tricuspid
valve, should be standard. In this context, injuries and
Ullrich Meier
bleeding from the veins of the neck and arteries, as well
as the submandibular gland, the accessory nerves, and
A standard procedure in the surgical treatment of normal
the vagus nerve, are very uncommon. Inadvertent
pressure hydrocephalus (NPH) is the implantation of a
implantation in the thorax with the formation of a hydro-
valve-controlled, ventriculoperitoneal
(VP) shunt. The
thorax is rare.1
most frequent intraoperative complication during this
However, in addition to the incorrect positioning of the
procedure is incorrect positioning of the catheter. Older
ventricular catheter over the frontal bore hole anterior to
sources in the literature1 have reported a rate of up to
the coronal suture, there may be other intracranial com-
12% atypical positioning of the ventricular catheter
plications. Consequently, it is possible that intracranial
depending on the site of the implantation. The most fre-
bleeding may occur with a manual puncture as well as
quent incorrect positioning of the ventricular catheter
with a load-reducing-dependent puncture. The most fre-
was found for temporoparietal implantations, followed
quent location of intracranial bleeding in this context is
by implantations in the area of the central region
an intracerebral hematoma in the ventricular catheter.
and over the occipital lobes. The lowest rate of incorrect
Marmarou et al2 reported a rate of up to 3% of intra-
positioning was found for implantation of the ventricle
cerebral bleeding following shunt implantation. In our
catheter over the frontal brain. In contrast to the other
opinion, the rate of complications is around 1% to 2%. The
three sites of implantation described, there were also the
causes of this kind of intracerebral bleeding can be hered-
fewest neurological deficits as a result of incorrect posi-
itary
(e.g., Waldenström disease), as well as through
tioning of a ventricular catheter over the frontal brain.1
acquired bleeding disorders (e.g., alcohol abuse, coagula-
By definition, in the above study, only primary
tion-inhibiting drugs). Other forms of intracranial bleed-
incorrect positioning of the catheter was taken into
ing include subdural and epidural hematomas, which are
account with regard to intraoperative complications and
rarely encountered. In contrast to the hematomas found
not secondary dislocations of catheters—for example, as a
in children, subgaleal and intragaleal bleeding occurs
result of a gain in size or a secondary retraction of the
only rarely in adults. The risk of intraventricular bleeding
catheter material with evidence of regular postoperative
from the choroid plexus exists only with revision inter-
catheter positioning.
ventions, when the ventricular catheter grows closely
Based on the above investigation, ventricular catheter
together with plexus structures and is removed only with
implantation over a bore hole anterior to the coronal
substantial tension. At the same time, it is advisable to
suture will be taken as the standard procedure.
leave the ventricular catheter in situ so that there is dan-
Incorrect positioning in the subcutaneous course of the
ger of infection.
catheter can result in the formation of slings, breakages,
Obstruction of the ventricular catheter opening during
and disconnection, with consequential obstruction of
puncture, as a consequence of brain tissue detritus, has
cerebrospinal fluid
(CSF) drainage. These disturbances
also been investigated scientifically. Utilizing a multicen-
can be caused by intraoperative and primary incorrect
ter, prospective, and randomized study, researchers
positioning, and also as a result of movement by the
intended to demonstrate that a peel-away sheath could
patient and growth in size. As a rule, incorrect positioning
help eliminate this possibility of complications due to
in the abdomen is associated with extraperitoneal cathe-
primary ventricle obstruction.3
Unfortunately, the
ter positioning, with CSF congestion, subcutaneous CSF
“peel-away sheath” hydrocephalus study of contactless
accumulation, and signs of insufficient shunt function.
ventricular catheter insertion provided no statistically
Incorrect abdominal positioning as a result of perforation
significant results.3
Consequently, no advantage was
of the abdominal organs, the inguinal ring, and the navel
seen in the use of a peel-away sheath in the implanta-
is exceptionally rare, just as are injuries to the abdominal
tion of the ventricular catheter.3
vessels and intraoperative injury to the intestines.1
Meier et al4,5 reported an incorrect positioning rate of
In cases where it is not possible to drain into the peri-
3.1% in a sample of 128 patients. In that study, primary
toneum, conversion of atrial drainage represents a practi-
incorrect positioning of the ventricular catheter (1.6%)
cal solution. Incorrect positioning of a ventriculoatrial
occurred in two patients and, likewise, in two patients
(VA) shunt may be produced by a heart catheter that is
incorrect positioning of the abdominal catheter appeared
too short or too long. This is a problem associated with
in the abdominal skin or in an intra-abdominal cyst (1.6%).
120
Complications
because it can frequently block it. When shunt insuffi-
?
ciency has been demonstrated, the complete shunt,
Table 15.1 Rates of intraoperative complications
including the valve and the complete catheter, should be
Intraoperative complication
Rate (%)
removed and replaced with a new system. All intracranial
Catheter displacement
3-7
bleeding with an expansion effect and/or corresponding
● Ventricular catheter
1-6
acute symptoms of an increase in intracranial pressure
● Subcutaneous shunt
0-1
must be operated on immediately and be treated as a
cerebral emergency. For this purpose, there is a vital OP
● Peritoneal catheter
1-3
indication. In rare cases of injury to the intestines or, very
Intracranial bleeding (intracerebral hematoma)
0-3
rarely, injury to the large abdominal vessels, the neuro-
Intestinal injuries
Rare
surgeon should be advised to consult a visceral surgeon
Unsuccessful catheter installation in the ventricle
Rare
intraoperatively to clarify the situation as soon as possi-
system, in the veins of patients with intracranial
ble. At the same time, the prognosis for the patient
cysts, or in the peritoneum in patients with
accretions
depends on the type of injury, its recognition, and correct
specialist care. In this context, it should be assumed that,
Injury to the large vessels of the abdomen
Very rare
when there is an intestinal injury or injury to large
Acute intraoperative pulmonary embolism
Very rare
abdominal vessels, the correct treatment should be given
during the same procedure, and implantation of the
Sprung et al6 reported a rate of 6.9% incorrect positioning
peritoneal catheter should not take place in the abdomen.
of the shunt catheter in a population of 144 patients;
Consideration should be given as to whether the implanta-
eight ventricular catheters (5.6%) and two peritoneal cath-
tion of the peritoneal catheter should then take place in the
eters (1.4%) were affected.6 Regarding rare complications
interval or should be given precedence at another drainage
(< 0.1%) and very rare complications (< 0.01%), there are
site (atrium drainage). For patients who have already expe-
only case reports and rates of these types of complications
rienced revisional operations numerous times, this may
are not known (▶ Table 15.1).4-6
prove to be difficult. In this case, a small venous diam-
With intraoperative complications, the fundamental
eter, strong adhesions or cysts in the abdominal cavity,
principle of neurosurgery—to avoid the same—still
as well as intracranial cysts may result in the need to
applies. However, this is not entirely possible for the
use very infrequent drainage sites
(e.g., intrapleural,
incorrect positioning of a catheter. Some neurosurgeons7,8
intravesical). Guidelines from the various neurosurgical
tend to hold the view that the aid of neuronavigation or
professional associations2,8 give no recommendations
mechanical puncture can minimize the incorrect position-
regarding these rare and special drainage variant sites.
ing of ventricular catheters. In the region of the peritoneal
catheter, special trocars and/or laparoscopic implantation
techniques perform the same task. However, it can be
15.2 Infections
assumed that, by using these aids, a reduction in the rate
of complications for “incorrect catheter positioning” can
Johannes Lemcke
indeed be achieved, but it is unlikely to completely
exclude this intraoperative complication.
Ay, think so still, till experience change thy mind.
The authors suggest verifying the correct positioning of
(Mephastophilis in Christopher Marlowe's
the catheter using postoperative computed tomography
Doctor Faustus, c.1592)
(CT) or magnetic resonance imaging (MRI) control of the
head, which is considered to be an undisputed standard.
There are few data available in the literature on the subject
The intra-abdominal position of peritoneal catheters
of shunt infections in patients with idiopathic NPH (iNPH).
should be documented using radiographic control of the
As the epidemiologic parameters for these patients
abdomen at two levels when standing. In the event of
differ substantially from those for children who are the
incorrect positioning, the rule is to carry out a catheter
recipients of shunts, for whom many data are available, it
revision as soon as possible when the patient is in a stable
is difficult to give specific evidence-based guidelines for
general condition. This is because incorrect positioning
iNPH. Nonetheless, the available literature on iNPH has
leads to obstructions in the shunt course in the majority
been carefully reviewed and collated to describe the
of cases and can cause valve insufficiency, shunt insuffi-
microbiologic mechanisms of shunt colonization.
ciency, and a negative course of the disease. In patients
with bleeding or intracerebral hematoma with no clinical
15.2.1 What is a Shunt Infection?
indications of an increase in intracranial increase, con-
servative therapy should be pursued first. At the same
We propose that there are three categories of shunt-
time, it is important that following the reabsorption of
related infections, namely shunt colonization, shunt-related
the bleeding, control of valve or shunt functionality must
CSF infection/meningitis, and shut-related peritonitis/
take place to control the blood that has entered the shunt
abdominal abscess.
121
Complications
The ability to produce slime enables organisms to
15.2.2 Shunt Colonization
adhere to one another and to carry out an exchange of
Biofilm Development
substances. The slime film acts as a mechanical barrier,
protecting bacteria from host defense mechanisms and
The colonization of VP shunts in patients with iNPH fol-
15
systemic antibiotics (▶ Fig. 15.1).
lows the same general mechanisms underlying the colo-
nization of implanted biomaterials identified in the
Clinical Implications of Biofilm Development
1980s.10 To understand shunt infections, we also need to
understand these mechanisms.
Principles on how to avoid shunt colonization in patients
Once a biomaterial such as a silicone catheter with a
with iNPH can be derived from the following knowledge.
valve made of silicone or titan is implanted, it becomes an
The source of bacteria that causes shunt colonization
easy surface for colonization.11 The combination of trau-
is normal skin flora. The local accumulation of possible
matized tissue in the immediate vicinity of the implant
infectious organisms in the vicinity of the implanted
and the lack of a host defense mechanism provides the
shunt results from: (a) direct contact between the skin
ideal conditions for colonization by organisms. The initial
and the shunt material before or during the implanta-
attachment is determined by the physical characteristics
tion process, or (b) by immigration of the skin flora
of the cell, the fluid medium, and the surface of the mate-
via the wound margin into the sites of shunt implanta-
rial used, and can be characterized as reversible, non-
tion during the surgical operation.16 The former can be
specific adhesion. At present we do not know whether
avoided by using skin-covering film within the surgical
the docking cell is a host cell of the patient or a polymer-
wrap, while the latter can be avoided by increasing
ophil bacterium such as Staphylococcus epidermidis.12,13
the speed of the operation, using just a few small sur-
The time window for attachment has been referred to as
gical approaches to reduce surgical trauma, and adopt-
“the race for the surface.” Once the bacterial cell has
ing a “no touch policy”17 (i.e. opening the sterile wrap
adhered, short-range chemical interactions support the
of the shunt catheters and the device as late as possi-
process. Nonspecific fimbrial interactions on the part of
ble during the surgical procedure). A similar approach
the bacteria now proceed in a manner like that of interac-
has been introduced for handling external ventricular
tions with the glucoproteinaceous conditioning film that
drainages (EVDs).18
directly covers any artificial material brought into a mam-
The time window, during which it becomes critical
malian body. The adhesion by exopolysaccharide glycoca-
as to whether tissue cells or bacteria win the “race for
lyx polymers is irreversible. A continuous biofilm is now
the surface,”11 is known to be the few hours following
set up with microcolonies and single cells embedded in a
the surgical procedure. Since the time window is
highly hydrated, predominantly anionic matrix of bacte-
known, we can apply prophylactic systemic antibiotics
rial exopolymers and trapped macromolecules.14
for its duration.19,20
........ Attachment
....... Adhesion
Aggregation
........ Dispersion
10,000
Shear
15
10
2° minutes
Fimbrial
Microzone
adhesin
5
Repulsion
Attraction
Polymer
Ca +
Fe +
1° minute
Attraction
Mg
(-)
(-)
Al +
(-)
(-)
(-)
0
Surface oxide
Van der Waals
Hydrophobic
Receptor
Receptor
Chemical
forces
interaction
interaction
0 hours
3 hours
24 hours
48 hours
Substratum
Fig. 15.1 Molecular mechanisms of biofilm development.11
122
Complications
Staphylococcus epidermidis have been implicated as the
15.2.3 Shunt-Related CSF Infection/
22
organisms causing shunt-related abdominal abscesses.
Meningitis
Surgical revision is essential in the event of shunt-related
Patients with a VP shunt can be infected with meningitis
abscesses. The shunt has to be removed and externalized
until the infection has been eradicated with certainty.
in the same diverse ways that patients without shunts
can be infected. In patients with shunts, the shunt must
be viewed as a potential for secondary complications
15.2.5 Epidemiology of Shunt
because the infection is able to spread in an artificial way.
On the other hand, a CSF infection or meningitis can, in
Infections in Patients With iNPH
and of itself, be a secondary complication in shunt
The frequency with which shunt infections are
patients because infections can spread into the CSF via
detected and the organisms are identified depends to
the shunt.
a large extent on the methods used.23-26 Most authors
The most common manifestations of a CSF infection in
do not describe the pathway of infection in patients
patients with iNPH and shunts are slight or doubtful clini-
with suspected shunt infection when the focus of the
cal signs of meningitis, such as minimal nuchal stiffness,
paper is not shunt infections per se. Indeed, in some
slightly elevated temperatures, sleepiness, slightly ele-
publications, the rate of shunt infections has not been
vated C-reactive protein (CRP) values, slightly elevated
reported at all.
lactate values in the CSF, and slightly elevated protein val-
ues in the CSF. The microbiologic examination of the CSF
Frequency of Shunt Infections
will reveal the presence of an organism, which could pos-
sibly be a secondary contaminant, or it will show no con-
The rule of thumb is: the more conscientious authors are,
tamination at all.
and the longer the follow-up period is, the higher is the
Patients with iNPH can produce deceptive results.
reported rate of shunt infections. Otherwise, the more
Because of their age, they do not tend to produce fulmi-
well established the diagnostic pathway, the more pre-
nant reactions of their immune system and can mislead
cisely defined are the criteria to be met in diagnosing an
the clinician into neglecting the signs of shunt-related
iNPH, and the lower the rate of shunt infections.
CSF infections. Once they do show clear signs of such an
Reviewing the literature related to shunt infections in
infection, they have already been badly affected by the
patients with iNPH over the last
20 years, we have
infection.
become aware that a realistic rate of shunt infection in a
follow-up period of 2 years or less is about 3% to 6%. In
long-term follow-up periods of 5 years' duration or more,
15.2.4 Shunt-Related Peritonitis/
10% is a realistic rate. However, we have to consider the
Abdominal Abscess
fact that peritonitis and meningitis occur independently
Attention should be paid to abdominal symptoms in
of the presence of VP shunts. The incidence of these infec-
tions has been unavoidably included in the infection rates
patients with a VP shunt. Patients can complain of
given in publications reporting long-term follow-up
abdominal pain and distension, dysuria, constipation,
headache, and fever. Shunt-related abdominal infections
(▶Table 15.2, ▶Table 15.3).
can appear months or even years after the last shunt sur-
gery. Consequently, the chronology may differ from a pri-
Lumboperitoneal/Ventriculoatrial
mary shunt colonization. A shunt-related abdominal
Shunts in NPH
pseudocyst, abscess, or peritonitis can be diagnosed using
abdominal ultrasonography and/or CT scans. In patients
Chang et al27 reported on the management of 32 patients
with leukocytosis of the peripheral blood, no pleocytosis
with NPH by insertion of a lumboperitoneal (LP) shunt,
of the CSF and no clinical symptoms of meningitis, liga-
and found one patient
(3%) with a shunt infection
tion of the shunt, and administration of systemic antibi-
4 months after surgery.
otics can be performed.
There are almost no current data available regarding
Kariyattil et al21 reported that abdominal symptoms
infection rates in VA shunts for iNPH. Bret et al28 needed
may be the mode of presentation in patients with ascites,
to perform repeat operations as a result of septic compli-
whereas shunt-related abdominal pseudocysts are more
cations in
5% of a patient collective of
129 patients,
likely to present with shunt malfunction.
including 14 patients with a VA shunt, within a follow-up
Shunt-related abdominal abscesses can also appear with
period of 16.7 months.
a time delay of months or years. Abdominal pain and fever
associated with an elevated white cell count are typical.
Organisms
Ultrasonography studies and enhanced CT scans usually
show well-defined, lobular fluid collections. Methicillin-
The organisms found responsible for shunt colonization
resistant Staphylococcus aureus, Proteus mirabilis, and
originate, in the majority of cases, from skin flora.29
123
Complications
??
Table 15.2 Prospective studies on patients with iNPH
Author
Year of publication
No. of patients
Follow-up time
Infection rate (%)
Mirzayan et al80
2010
34a
80.9 ± 51.6 months
8a
Meier et al81
2008
148
12 months
3
McGirt et al82
2008
132
18 ± 13 months
12
Kahlon et al83
2007
27b
5.5 ± 1.4 years
3b
Marmarou et al84
2005
102c
12 months
3
Sorteberg et al85
2004
17
9
(5-15) months
6
Boon et al86
1998
96
12 months
3
Larsson et al30
1991
74
2.1 years
19
Greenberg et al35
1977
45d
16.7
(3-29) months
7
a34 patients with a long-term follow-up, out of a total of 51 treated patients; the infection rate was given for the total number of patients
(4 of 51).
b27 patients with a long-term follow-up, out of a total of 75 treated patients; the infection rate was given for the total number of 75
patients (1 patient with shunt infection, 1 patient with wound infection).
c102 patients of a total of 151 treated patients underwent shunt surgery.
d45 patients with follow-up > 12 months, out of a total of 73 treated patients.
??
Table 15.3 Retrospective studies on patients with iNPH
Author
Year of publication
No. of patients
Follow-up time
Infection rate (%)
Eide and Sorteberg87
2010
130
2
(0.3-6) years
9
Pujari et al88
2008
55
5.9 ± 2.5 years
10
Zemack and Romner89
2002
147
26.7 months
6.4a
Lund-Johanson et al40
1994
95
1-9 yearsb
8.4
aOf a total 218 patients, including 71 patients with secondary normal pressure hydrocephalus.
bNo mean follow-up time given.
Abbreviation: iNPH, idiopathic normal pressure hydrocephalus.
Larsson et al found that CSF cultures grew Staphy-
15.2.6 Does My Patient Have a
lococcus epidermidis in 68%, Propionibacterium acnes
Shunt Infection?
in 12%, and were negative in 18% of cases of clinical
infections in patients with NPH.30
Fan-Harvard
Clinical Diagnosis of Shunt Infections
assumed that infections of central nervous system
shunts are dominated by coagulase-negative staphy-
The clinical presentation of patients with shunt infections
lococci, with Staphylococcus epidermidis accounting
can occur with obvious symptoms leading to the diagno-
sis. Fever, along with rubor of the track of the shunt, will
for 50% to 75% of infections, followed by Staphylococcus
be relatively straightforward symptoms.
aureus.30
Walters et al assessed31
200 pediatric cases
with VP shunt infections and found gram-positive cocci
However, the vast majority of patients will present
distributed to Staphylococcus epidermidis in
47%,
with less straightforward symptoms or mild symptoms,
Staphylococcus aureus in
27%, Streptococcus faecalis
or they will present with symptoms that could allow
different diagnoses.
in 10%, and miscellaneous in 13%, as well as gram-
negative rods: Escherichia sp. 19%, Klebsiella sp. 19%,
Pseudomonas sp. 8%, and miscellaneous 4%. According
What Can We Learn From the Time of
to Bayston et al,32 Propionibacterium acnes, a normal
Outbreak of a Shunt Infection?
anaerobic skin inhabitant, causes up to 14% of infec-
tions. Livni et al33 isolated Staphylococcus epidermidis
The period of time between surgery and the occurrence
and Staphylococcus aureus from infected shunt
of primary shunt colonization is, in our experience,
material. Sandoe and Longshaw34 were able to show
between a few days to about 2 months.
that Staphylococcus lugdunensis caused a VP shunt
Shunt-related infections occurring beyond 2 months
infection.
after surgery almost always have other causes than the
124
Complications
surgery itself. In these cases, we have to look for a reason,
lactate and protein content, and number of cells change
which can fall into one of three categories:
relatively rapidly with changes in infectious events. Com-
1. Mechanical cause (e.g., secondary exposition of shunt
pared with CSF from the valve reservoir, these parameters
or valve due to permanent pressure of the temples of
may show changes for a long period of time after an
spectacles onto a retroauriculargly implanted valve)
infectious event has occurred if taken from a lumbar
2. Primary infection of any of the body compartments
puncture. The disadvantage of puncturing the valve pre-
penetrated by the shunt due to factors not dependent
chamber is the danger of a de novo infection caused by
on hydrocephalus and the shunt
the puncture. Every time a CSF sample is taken, material
3. Changes in the immunocompetence of the patient due
for microbiologic examination should also be taken.
to factors independent of hydrocephalus and shunt
The examination of parts of the shunt should not be
that result in an outbreak of a clinically nonevident
considered to be a regular part of the diagnostic process,
shunt colonization acquired during surgery
as it can only be performed if the surgeon has already
explanted the shunt as a result of a serious suspicion of
There do not appear to be any published studies in which
shunt-related infection. Several authors tend toward
a differentiation has been made between different types
making an “overdiagnosis” by examining parts of the
of shunt-related infections. Reports in the literature have
shunt apparatus. Bayston et al38,39 reported positive cul-
given a wide range of times between surgery and shunt-
tures on explanted shunt parts in the absence of clinical
related infections developing in patients with iNPH, for
infections. Walters et al31 compared the clinical presenta-
example, 1 to 30 weeks30 and 1 to 14 months.35 These
tion with microbiologic examination of CSF samples and
data suggest that several types of infections have been
microbiologic examination of the shunt apparatus. They
recorded.
found a precise match only in cases in which there were
A special subgroup of shunt infections consists of
meningeal signs of infection. In patients with fever alone
delayed infections caused by long-term changes in the
or peritoneal signs alone, CSF cultures were less sensitive
material used for the shunt system itself, such as mineral-
than cultures of parts of the shunt.
ization and biodegradation of silicone materials. These
changes are well known in pediatric patients but are not
15.2.7 How Can Shunt Infections Be
a serious problem in patients with iNPH.36
Avoided in iNPH?
Clinical Examination
Shunt infections are a source of frustration for the
In addition to the neurologic examination of patients sus-
surgeon and dangerous for the patient. Therefore, the
avoidance of shunt infections in patients with iNPH
pected of having a shunt-related infection, a local exami-
nation and palpation of the shunt are needed. Wound
should be a priority.
dehiscence, sutures that stick out, exposed parts of the
There are three possibilities of prevention associated
shunt, and rubor along the shunt trace, in particular,
with the surgeon, the patient, and the material.
should be looked for. The abdomen should be palpated
and auscultated.
The Surgeon
The neurologic examination should target symptoms of
Lund-Johansen et al40 found that the infection rate was
meningitis, as well as symptoms of shunt dysfunction,
higher among patients operated on by residents, whereas
especially underdrainage due to abdominal cysts or
the choice of the shunt type (Orbis-Sigma, Holter, Hakim)
obstruction by infectious concrement.
and the perioperative use of antibiotics were not corre-
lated with complication or failure rates. As the proviso to
Paraclinical Examination
let only experienced surgeons implant shunts would
There are types of sample to obtain for a paraclinical
work, at the very most, for one generation only, we have
examination: serum samples, CSF samples, and parts of
to further differentiate the factor “surgeon.”
the shunt
(e.g., catheter tips). Obtaining the latter
Risk factors for shunt infections associated with the
involves the explantation of the shunt.
surgeon are extended operation time, subcutaneous
The analysis of serum CRP and leukocytes has not been
hematoma as a culture medium for bacteria, provoking
reviewed for patients with iNPH, but it has for other
extensive skin contact with the shunt material, and sub-
patients with shunts. Schuhmann et al37
noted that
cutaneous sutures that are too long and cause suture
measuring CRP in blood serum significantly increased the
granuloma. The probability of contamination of the shunt
precision of diagnosis of a shunt infection.
material with organisms originating from the surgeon
CSF samples can be obtained from a valve reservoir or
seems to be extremely low; in a swab series, Bayston et al
via a lumbar puncture. The advantage of CSF samples
demonstrated that organisms involved in shunt coloniza-
from the valve reservoir is that they are taken from
tion were present on the patient preoperatively.41-45
an area with a high CSF turnover and, therefore, the CSF
Another risk factor associated with the surgeon is the
125
Complications
choice of an inadequate shunt system, which can result in
protein conditioning film and exposed to Staphylococcus
the need for revision surgery and thereby increase the
epidermidis, it was able to kill the adhering bacteria
risk of infection.
within 48 to 52 hours.43
Early fear that BACTISEAL catheters shows a risk of
epileptogenic potential has not been substantiated.55
The Patient
In our patients with iNPH, we found a correlation
SILVERLINE
between the rates of shunt infections and the presence of
diabetes mellitus, adiposity, decubitus ulcer, and raised
There is disagreement in the literature regarding the in
CRP or leukocytosis. Because the first two cannot be
vitro results for SILVERLINE catheters (Spiegelberg, Ham-
cured in a short period of time, these patients should
burg, Germany). Bayston et al52 reported that SILVERLINE
catheters do not show the ability to eradicate higher
be operated on by experienced surgeons to reduce other
inocula of contaminating bacteria during in vitro tests,
risks of shunt infections.
In cases of pre-existing infections, with increased levels
while other authors have reported low bacterial coloniza-
of CRP or leukocytosis, as well as clinically detectable
tion during in vitro experiments.50
infections without increased levels of these parameters,
Nonetheless, there is evidence that SILVERLINE cathe-
ters work in practice. Lackner et al56 found a significantly
shunt insertion should be postponed by at least 4 weeks.
Bayston et al46,47 were able to show that a high CSF pro-
lower rate of catheter-related ventriculitis in patients
tein content per se does not increase the risk of shunt
with occlusive hydrocephalus. Izci et al57 implanted VP
infections. Brydon speculated that the risk of CSF infec-
shunts with SILVERLINE ventricular catheters in seven
patients with a positive CSF culture and reported that
tions in patients who are hyperproteinorrhaghic might be
the CSF culture became negative within a period of 14
increased because they have a different skin flora, with
possibly more pathogenic organisms present.46,47
months. In our patient sample, we were able to prove the
effectiveness of SILVERLINE EVDs in relation to the
prevention of catheter-related CSF infections.
58
The Material
There is no evidence demonstrating a toxic risk of
SILVERLINE catheters, although silver sulfide deposits
The alternatives that can be used to prevent shunt infec-
tions in patients with iNPH by selecting an appropriate
have been identified in the tissue, and silver was released
implant material can be divided in two categories, which
into the CSF.59,60
are described below.
The first category includes all possibilities for avoid-
15.2.8 How Can Shunt-Related
ing later indications requiring revision of the shunt
Infections Be Treated?
system due to complications other than infections.
Programmable valves can help avoid reoperations due
The type of treatment suitable for shunt infections
to overdrainage or underdrainage and thereby avoid
depends on the type of shunt-related infection. As we dis-
infectious complications. CSF reservoirs enable the sur-
cussed in Section 15.2.1, three categories of shunt-related
geon to confirm the suspicion of a shunt infection and
infections can be differentiated: shunt colonization, shunt-
to avoid a needless explantation of noninfected shunts.
associated meningitis, and shunt-related peritonitis.
Therefore, we would recommend that programmable
valves with reservoirs should be the standard alterna-
Treatment of Shunt Colonization
tive to be used in the treatment of iNPH.
The second category involves the use of antibacterial or
Shunt colonization originates from biofilm development
bacteriostatic shunt materials. Materials using two differ-
following the contamination of the shunt material with
ent principles are available. One is the impregnation of
skin flora and the subsequent victory of the skin flora in
the silicone shunt material with antibiotic substances,
the “race for the surface” against the tissue cells. Shunt
while the other uses the impregnation of the silicone
colonization can be primary—on implantation of the
with silver nanoparticles.
shunt—or secondary—taking place at a specific event
(e.g., exposure of the shunt as the result of a skin defect).
In both cases, bacteria have already won the “race for the
BACTISEAL
surface” and there is no possibility of contesting the vic-
BACTISEAL
(Codman, Johnson & Johnson, Raynham,
tory. Therefore, any shunt that has become colonized must
Massachusetts, United States) was the first material avail-
be removed. If a shunt-related infection occurs within the
able. BACTISEAL is a silicone rubber impregnated with
first 2 months following implantation, then we have to
clindamycin and rifampicin. The effectiveness of the
assume that shunt colonization has taken place. If a
material has been shown both in vitro and in vivo.14-54
shunt-related infection occurs in the context of an expo-
Even when the antimicrobial-impregnated silicone shunt
sure of the shunt or valve—independent of the time that
material was experimentally covered with a plasma
has elapsed since the shunt was implanted—then we also
126
Complications
have to assume that colonization of the shunt has taken
leukocytes) indicate no systemic inflammation, then a
place. Despite the fact that there are reports that shunts
new shunt (SILVERLINE or BACTISEAL) can be implanted
remain exposed for a long period of time
(e.g.,
on the opposite side. It should be stressed once more that
15 months), every confirmed case of colonization of a
this situation is an exception in patients with iNPH.
shunt results in an infection of the CSF sooner or later.61
Adhesion-mediated infections develop that are notori-
Treatment of Meningitis in Patients
ously resistant to antibiotics and host defenses, and tend to
With a Shunt
persist until the biomaterial or foreign body is removed.11,
62 In the 1980s, during which time different treatment
It is very uncommon to find meningitis in a patient who
modalities were advocated in neurosurgery, James et al63
has had a VP shunt for iNPH. In such cases, the shunt
conducted a randomized study of patients with a shunt-
should be removed and an EVD (SILVERLINE or BACTI-
related infection using three treatment groups. Group A
SEAL) should be implanted on the opposite to the side of
underwent shunt removal and received systemic antibiot-
the shunt. Bacteriologic testing of a CSF sample taken
ics and external ventricular drainage for the administration
perioperatively should be carried out. Directed systemic
of antibiotics. Group B was treated with removal and
antibiotics can be started immediately and intrathecal
immediate replacement of the shunt, and intrashunt anti-
antibiotics should be initiated once the results of bacte-
biotic therapy. Group C received antibiotics without remov-
riologic tests have become available. Our experience is
ing or replacing the shunt. All patients in Group A and 90%
limited to the off-label use of vancomycin (2 × 5 mg intra-
of the patients in Group B were treated successfully,
thecal). We generally give this for 10 days. After 48 to
whereas only three patients in Group C responded to treat-
72 hours after intrathecal application of antibiotics has
ment. Walters et al confirmed these results in a retrospec-
ceased, we take a CSF sample for bacteriologic tests. If this
tive analysis of more than 200 patients.64
sample is sterile and serum parameters (CRP, leukocytes)
Therefore, we recommend removal of the shunt in
indicate no systemic inflammation, then the EVD is
patients in whom there is a reasonable suspicion of shunt
removed. A new shunt (SILVERLINE or BACTISEAL) can be
colonization, even if there are no signs of a CSF infection,
implanted on the opposite side after about 3 months.
meningitis, or generalized infection. In patients with
The development of a cerebral abscess around the ven-
iNPH where there is colonization of a shunt but no signs
tricular catheter of a VP shunt in patients with iNPH is
of CSF infection, and if the patient tolerates being without
very uncommon. We do not have experience of our own
a shunt for a period of about 3 months, an EVD is not
in this regard; however, in the literature, one can find rec-
implanted. The patient should be treated with intra-
ommendations for the neuroendoscopic removal of both
venous antibiotics, primarily to prevent the spread of the
the ventricular catheter and the abscess.66
infection to the CSF. With regard to the infection per se,
Recurrent pleural effusions have been reported in
the host of the bacterial load is removed with the shunt,
patients with shunt-related infections in ventriculo-
and the antibiotic therapy has, in some way, a “preven-
pleural shunts. These patients also show high lactate
tive” characteristic. Therefore, there is no need to take a
dehydrogenase levels and lymphocytosis in the pleu-
sledgehammer to
“crack a nut.” Recommendations
ral fluid. Shunt ligation has been recommended as
regarding modern broad-spectrum antibiotics (e.g., line-
treatment.67
zolid) for the treatment of shunt-related infections can be
found in the literature.65 In our opinion, second-genera-
Treatment of Peritonitis in Patients
tion cephalosporins that permeate the CSF are adequate
With a Shunt
for most patients. We would recommend application for
7 to 10 days after removing the shunt. The implantation
Unlike other patients with a shunt and peritonitis, those
of the new shunt should be carried out about 3 months
with iNPH will overcome in 4 to 6 weeks with no CSF
after the removal of the old one. We prefer to use the
drainage. Consequently, the procedure of the first choice
opposite side for implantation of the new shunt.
should be to remove the entire shunt system and reim-
If the hydrocephalic constellation of the patient neces-
plant a new shunt system after the completion of the
sitates permanent drainage after removal of the shunt,
treatment of peritonitis with antibiotics.
then we recommend the implantation of an EVD (SILVER-
In cases in which there is some doubt regarding an
LINE or BACTISEAL) on the same side of the shunt that
abdominal infection, or if sterile inflammation of a dislo-
has been removed. In addition, systemic antibiotics
cated abdominal catheter is more likely, one option may
should be given. Intrathecal administration of antibiotics
be to ligate the shunt on the downstream side of the valve
is not necessary in patients who do not have symptoms
(e.g., subclavicular) and remove only the abdominal cath-
of meningitis. Following systemic antibiotic therapy over
eter to replace it after antibiotic treatment. Most cases
a period of 7 to 10 days and a 3-day period with no anti-
that were tried to treat in this way, for whatever reason,
biotics, bacteriologic tests are carried out on a sample of
ended with the removal of the entire shunt at an earlier
CSF. If the sample is sterile and serum parameters (CRP,
or later stage.
127
Complications
The successful conversion of VP shunts into VA shunts
As valve-related or shunt-related complications, we see
in cases of clinically isolated abdominal shunt-related
underdrainage and overdrainage of the shunt system in
infections has been reported in the literature, but they
surgical complications.
have been of an experimental nature only.68
15.3.1 Underdrainage
15.3 Postoperative
In underdrainage, the amount of CSF that is drained per
time interval is too low, with respect to the individual
Complications
demand of the patient. This can be related to a hitherto
Michael J. Fritsch
unrecognized surgical complication (obstruction) or it
can be related to the valve setting and the shunt setting.
Complications in shunt surgery can be divided into surgi-
In underdrainage, there is no clinical improvement after
cal complications and valve-related or shunt-related
shunt surgery, and the condition of patients, who have so
complications. Surgical complications, which account for
far shown improvement after treatment, begins to deteri-
approximately 50% or more of all complications, include
orate based on the Hakim triad or the Kiefer index. The
early infection, wound dehiscence, all types of dis-
size of the ventricles on imaging studies does not
connection, and misplacement of shunt parts (e.g., proxi-
correspond with the clinical outcome of patients with
mal ventricular catheter, valve, distal peritoneal or atrial
iNPH; therefore, they are not helpful when evaluating
catheter; ▶ Fig. 15.2, ▶ Fig. 15.3a, ▶ Fig. 15.3b).
suboptimal shunt function.69
If obstruction, disconnection, or incorrect placement
is ruled out
(radiography shunt series, shuntogram),
then adjustment of the valve (if it is adjustable) should
be performed. Adjustment should be performed in steps
that are sufficiently large. We aim for an adjustment of
at least 2 cm H2O, or better 3 cm H2O, to see a change in
the clinical condition, but we also closely follow-up each
patient.
Another option to work-up underdrainage would be a
repeated tap test, which can be performed either via lum-
bar puncture or via shunt tap. After removing 40 mL CSF,
the patient should improve. If the valve is not adjustable
and the patient improved after the puncture, then we
would see the indication for a shunt revision with a lower
pressure valve.
Underdrainage presents complications of which most
neurosurgeons are aware.
15.3.2 Overdrainage
Overdrainage, which can also be shunt- or valve-related,
is a complication that, with only a few exceptions, is still
not known or recognized well enough in the medical
Fig. 15.2
Secondary dislocation of the distal (peritoneal)
community and in the public.70
catheter.
During overdrainage, clinical improvement is present,
but patients may also develop orthostatic headaches,
Fig. 15.3 (a) Incorrect placement of the
proximal (ventricular) catheter in the brain
parenchyma and (b) secondary obstruction
of the catheter tip.
a
b
128
Complications
Fig. 15.4 (a-d) Bilateral subdural hygroma
in a patient with iNPH; following initial
clinical improvement in the Hakim triad, the
patient developed orthostatic headaches
during the daytime, dizziness, and
imbalance.
a
b
c
d
imbalance, and new focal neurologic deficits. MRI or CT
with iNPH and an opening pressure of 5 cm H2O showed
may display relatively small ventricles and subdural
a statistically significant better outcome than patients
hygroma, either unilaterally or bilaterally (▶ Fig. 15.4).
with an opening pressure between 10 and 13 cm H2O.72
Both studies have demonstrated that a low-pressure
setting is better for the clinical improvement of the
Does Overdrainage Occur?
patient but, at the same time, the risk of overdrainage
Boon et al71 studied the optimal opening pressure of
increases. One could conclude that it is one of the choices
valves implanted in patients with NPH in the Dutch Nor-
that we must decide upon as surgeons (▶ Fig. 15.5).
mal-Pressure Hydrocephalus study.71 They examined 96
patients with iNPH who were randomized to receive
either a low-pressure valve with an opening pressure of
4 cm H2O or a medium- to high-pressure valve with an
opening pressure of 10 cm H2O. Patients were examined
before surgery and at 1, 3, 6, 9, and 12 months after sur-
gery. Clinical improvement was found in 74% of patients
with low-pressure shunts and in 53% of patients with
medium- to high-pressure shunts. The P value for this
difference was
0.06; therefore, the results were not
statistically significant. However, the authors con-
cluded that patients with iNPH should be treated with
a low-pressure shunt.
The second important point of the study is that sub-
dural effusions occurred in 71% of patients with a low-
Fig. 15.5 Low opening pressure is beneficial for the patient but
pressure shunt and in 34% of patients with a medium- to
increases the risk of overdrainage. The attempt to prevent
high-pressure shunt.71
overdrainage (with a higher opening pressure) is limiting the
The clinical results of the Dutch NPH study were con-
benefit of shunt placement.
firmed by Meier et al72 who demonstrated that patients
129
Complications
(shunt). Based on the laws of physics, these fluid-filled
How Does Overdrainage Happen?
spaces, which are connected to each other, have a pres-
In a shunted person, there are four pressure forces that
sure gradient depending on the position (difference in
must be taken into consideration.
height) of the two spaces in relation to each other
First, there is the pressure inside the ventricles (intra-
(▶Fig. 15.6a).
cranial pressure, ICP). The vector of this pressure is
If a person is standing, then the upper fluid-filled
directed parallel to CSF flow—in other words, from the
space
(ventricle) is approximately
40 cm above the
ventricle to the valve into the distal catheter.
lower fluid-filled space (abdominal cavity at the level
The second pressure is the opening pressure of the
of the diaphragm). The hydrostatic pressure equals a
valve, which is in opposition to the first pressure vector
gradient that, in the upright position, is directed from
and counteracts the ICP.
above to below; therefore, it is acting parallel to the
The third force is the pressure in the abdominal cavity
ICP, which is directing CSF flow from a proximal to
(of which there is not much known). The pressure in the
distal direction. Those two pressures are counteracted
abdominal cavity also counteracts the ICP. If the pressure
by the opening pressure of the valve and the pressure
in the abdominal cavity is high, then the CSF flow from
in the abdominal cavity.
the ventricular system to the abdomen would be reduced
If we assign numbers on the pressure forces (vectors),
or diminished. The abdominal pressure probably changes
we can calculate with the following equation: the pres-
from second to second depending on the body position
sure in the ventricular system (ICP) should be normal,
and the activity of the shunted person.
meaning around 0 cm H2O. The valve opening pressure
The fourth pressure force is the hydrostatic pressure.
could be set at 10 cm H2O. The pressure in the abdominal
The hydrostatic pressure is a pressure gradient between
cavity can be calculated at approximately 10 cm H2O. The
the ventricular system and the abdominal cavity. Two
hydrostatic pressure in an average person, who has a
fluid-filled cavities, one the ventricular system and the
body height of about 175 cm, would be approximately
other the abdominal cavity, are connected
via
a
tube
40 cm H2O (distance from the foramen of Monro to the
P (Ventricle) = ICP
P (Ventricle) = 0 cm H2O
P (Valve) = Opening pressure
P (Valve) = 10 cm
P (Hight) = Hydrostatic pressure
P (Hight) = Hydrostatic pressure
= - 40 cm
P (Abdomen)
P (Abdomen) = 10 cm
a
P total = 0 + 10 - 40 + 10 = - 20 cm
Overdrainage
b
P (Ventricle) = 0 cm
P (Ventricle) = 0 cm
P (Valve) = 10 cm
P (Valve) = 5 cm
P (Hight) = Hydrostatic pressure
P (Hight) = Hydrostatic pressure
= 0
= - 40 cm
P (Abdomen) = 10 cm
P (Abdomen) = 10 cm
P total = 0 + 5 - 40 + 10 = - 25 cm
P total = 0 + 10 - 0 + 10 = + 20 cm
Gravitational unit compensating 25 cm
c
Underdrainage
d
e. g. proGAV 05/25
Fig. 15.6 (a) P (Ventricle) = ICP; P (Valve) = opening pressure; P (Abdomen); P (Height) = hydrostatic pressure. (b) In the upright position
the patient would be drained at −20 cm of water and would be overdrained. (c) In the horizontal position (prone or supine) the
hydrostatic pressure is zero. Pressures in the ventricle and in the abdominal cavity remain unchanged. The patient would drain against
20 cm of water and would be underdrained. (d) The optimal setting for the patient example would be: normal ICP (pressure
ventricle = 0), low opening pressure of the valve (5 cm), abdominal pressure normal (10 cm), hydrostatic pressure in the upright position
(−40 cm) and compensation for the hydrostatic pressure with a gravitational unit (set at 25 cm).
130
Complications
diaphragm). If all four pressures were added together,
Shunt Function by Palpation
then a negative pressure of −20 cm H2O would result;
The technique to determine the valve/shunt function
thus, the patient would be overdrained (▶ Fig. 15.6b).
depends on the design of the shunt components, the pres-
In the horizontal position (prone or supine) the hydro-
ence of flushing/pump reservoirs, and their location (at
static pressure is zero. Pressures in the ventricle and the
the bore hole or more distally). Of course, different shunt
abdominal cavity remain unchanged. The patient would
designs may have their own particularities that cannot all
drain against 20 cm of H2O and would be underdrained
be described here. The following examination technique is
(▶Fig. 15.6c).
described for a shunt system with a flushing reservoir
The optimal setting for the patient would be: normal
between the bore hole and the valve (▶ Fig. 15.7):
ICP (pressure ventricle = 0), low opening pressure of the
● Testing the ventricular catheter: the membrane of the
valve
(5 cm H2O), abdominal pressure normal (10 cm
flushing reservoir is compressed with one finger, then
H2O), hydrostatic pressure in the upright position
the catheter between the reservoir and the valve is
(−40 cm H2O) and compensation for the hydrostatic pres-
occluded with a finger of the other hand. When remov-
sure with a gravitational unit
(set at
25 cm H2O)
ing the finger from the reservoir, the reservoir must be
(▶Fig. 15.6d).
promptly filled with CSF via the ventricular catheter
while the distal catheter is still occluded. The re-
15.4 Shunt Malfunction:
expanding membrane of the reservoir can be felt and
even seen. If it does not fill again, then the ventricular
Shunt Revisions
catheter may be partially or completely occluded or the
Uwe Kehler
ventricle may be collapsed due to overdrainage.
● Testing the valve together with the distal catheter:
occluding the proximal catheter (between the bore hole
15.4.1 When to Revise a Shunt?
and the flushing reservoir) with one finger, the reser-
No clinical improvement after shunt surgery or the
voir must be pressed out. If there is an enormous resist-
reappearance of clinical symptoms further in the course
ance or “compression” is not possible, then the valve or
of the disease strongly suggests a malfunction of the
shunt. If adjustable valves are used, then readjustments
of these valves should be performed. When there is no
change, a new diagnosis is indicated to detect the reason
for the shunt malfunction.
Diagnositics of Shunt Malfunction
In addition to clinical examination, imaging with CT or
MRI is indicated and a comparison with a preshunt imag-
ing is necessary. Larger ventricles indicate shunt mal-
function, while smaller ventricles indicate a functioning
shunt, but they can also be correlated with overdrainage.
Unchanged ventricles can only be interpreted together
with the clinical condition because ventricle size after
shunting with gravitational valves or after endoscopic
third ventriculostomy does not change substantially.
However, if clinical improvement is absent or clinical
deterioration is recognized, then a shunt malfunction
must be assumed.
Inspection
If shunt malfunction is suspected, then an examination of
the shunt is necessary. The valve and the course of the
Fig. 15.7 Examination of shunt function by palpation: the left
catheter should be inspected with additional palpation.
finger occludes the proximal catheter to avoid reflux to the
ventricles. The membrane of the flushing reservoir is now
Sometimes a dislocation of the shunt components can be
compressed pushing the cerebrospinal fluid out of the reservoir
palpated, or subcutaneous cysts/masses may be found.
and through the valve. Inability to push down the membrane
Pain around the shunt and flush along the shunt may
indicates a valve or distal catheter obstruction.
have been caused by inflammation.
131
Complications
distal catheter may be obstructed. If the chamber refills
planned. The timing of the revision surgery in infections
(with the proximal catheter still occluded), then the
must be regarded separately depending on the infection
valve mechanism may be defective.
status.
It is important to remember that shunt palpation is not
15.4.2 How to Revise a Shunt?
very precise—a complete blockage can be detected,
but determining an inadequate opening pressure is
Surgical strategy of shunt revision is easy if a clear cause
impossible.
is found such as catheter dislocation/misplacement, and
shunt disconnection (▶ Table 15.4).
Shunt Pumping
Repeated expression of the flushing reservoir with simul-
Ventricular Catheter Dislocation/Improper
taneous compression of the proximal catheter results in a
Placement
“pumping” function. Depending on the size of the reser-
In cases of intraparenchymal ventricular catheter place-
voir, 100 pumps may remove a substantial volume from
ment (▶ Fig. 15.8), the bore hole must be exposed and
the ventricles, thus constituting a “noninvasive tap test.”
catheter must be corrected. A similar procedure must be
If a malfunction of the shunt is present, clinical improve-
ment can be observed after this noninvasive tap test. A
permanent improvement will follow if the shunt is freed,
??
through this pumping maneuver, from a fibrin clot or if
Table 15.4 Shunt complications and their management
an occluding membrane around the distal end of the
Cause
Management
peritoneal catheter tears. A temporary improvement may
Underdrainage
be seen if these causes are not eliminated, if the valve
Shunt dislocation/mis-
Repositioning
opening pressure is too high, or if the peritoneal CSF
placement
resorption is insufficient. However, if an improvement is
Shunt disconnection
Reconnection
not seen after this pumping maneuver, it does not rule
Shunt obstruction
Flushing via the reservoir, replace-
out shunt malfunction.
ment of the obstructed part
If a CSF leak or a shunt disconnection is present during
False valve placement
Repositioning
“pumping,” then a subcutaneous mass will appear that
Mechanical valve failure
Replacement of the valve
will indicate as well as locate the problem.
Wrong valve selection
Replacement with adequate valve
setting, preferable with program-
Radiography
mable valve
Radiography can show the course of the catheter with all
Scar formation around the
Replacement at different position
its possible dislocations, migrations, incorrect position-
catheter tip
ing, and disconnections.
Impaired intraperitoneal/
Changing to atrial diversion
intrapleural cerebrospinal
fluid absorption
Shuntogram
High abdominal pressure/
Change to different
If no cause is found, then a shuntogram may be per-
high venous atrial pressure
formed. Radioisotopes or a radio-opaque contrast
If no cause is found
Revision with intraoperative test-
medium is injected into the shunt chamber and may
ing, change of malfunctioning
show an obstruction under the gamma camera or radio-
parts, eventually with cardial or
pleural shunt diversion
graph. However, a false-negative shuntogram occurs in
more than 30% of tests, and this must not be neglected in
Overdrainage
the interpretation.73
Wrong valve selection
Additional implantation of a grav-
If no reason
(e.g., dislocation, disconnection, or
itational device or flow limiter, or
replacing the existing valve with a
obstruction) is found for the malfunction, then there are
higher pressure setting
two other possibilities, namely that the shunt problem
Oblique placement of a
Repositioning the vertical position
has not been found or the shunt indication (and that
gravitational valve
means the diagnosis of iNPH) was incorrect. Therefore,
Valve failure
Replacement of the valve
new diagnostics, such as for the initial diagnosis of
iNPH with (repeated) tap test, is necessary to prove the
Infection
shunt indication itself and, with that, the indication
Infected shunt
Antibiotics, shunt removal and
for shunt revision.
new shunt implantation after
complete cure infection
With the reappearance of NPH-like symptoms or a lack
Wound dehiscence with
Antibiotics, shunt removal, new
in clinical improvement after shunt surgery (with a reas-
open exposed shunt parts
shunt preferably on the opposite
sessed indication for shunt surgery), revision should be
side, exceptional local revision—
132
Complications
Fig. 15.8 Computed tomography, sagittal reconstruction:
incorrect paraventricular intraparenchymal placement of ven-
tricular catheter.
Fig. 15.10 X-ray of shunt disconnection.
current imaging should be available because a disconnected
catheter might have migrated further. This can make it
difficult or even impossible to reach the migrated compo-
nents of the shunt from the restricted exposure.
Kinked Shunt Catheter
A kinked catheter can also be revised via local exposure
(▶Fig.
15.11); however, the exposure must be long
enough so that a local improvement does not lead to new
peripheral kinking.
Oblique Positioned Gravitational Valve
Gravitational valves must be orientated in a vertical posi-
tion when patients are walking or sitting to develop their
Fig. 15.9 Axial computed tomography scan: migration of
optimal action. If oblique implantation is detected as a
ventricular catheter with bore hole reservoir.
cause for the malfunction of the shunt, then repositioning
in a vertical position is necessary (▶ Fig. 15.12). However,
performed in cases of ventricular catheter migration
vertical positioning of the valve is not easy if the patient
(▶Fig. 15.9). If the ventricular catheter is torn and has
is lying on the operating table with a partially rotated and
migrated entirely into the ventricles, then it can be
even slightly flexed cervical spine covered with drapes in
retrieved by an endoscopic approach. In addition, leaving
the operating field. Therefore, the optimal position
the catheter in the ventricle could be an option if endo-
should be marked preoperatively on the skin so that the
scopic recovering seems to be difficult and is hazardous.
correct position can be recognized intraoperatively.
Disconnection
Dislocation of Abdominal Catheter
When disconnection occurs (▶ Fig. 15.10), local exposure of
Often, most incorrectly positioned peritoneal catheters
the shunt is usually sufficient to reconnect the components
are found in a subcutaneous pouch filled with CSF
and to secure the connections with sutures. However,
(▶Fig. 15.13, ▶Fig. 15.14). Local revision is necessary,
133
Complications
Fig. 15.12 X-ray of a gravitational-assisted valve implanted
obliquely (dotted line shows the optimal alignment of the
gravitation-driven valve).
Fig. 15.11 X-ray of a kinked catheter.
and the peritoneum is reopened and a long catheter
Obstructed Shunt Catheter or Valve
(> 30 cm) is placed in the peritoneal cavity. The cathe-
If an obstruction can be demonstrated, (e.g., through fill-
ter should be fixed with a purse-string suture at the
ing of the shunt with a contrast agent [“shuntogram”]),
level of the peritoneum to prevent a new dislocation.
then the revision should be performed by replacing the
Because some intraperitoneal scarring from the previ-
obstructed part. ▶ Fig. 15.15 shows an obstructed ventric-
ous shunt surgery may be present, reopening of the
ular catheter that has been removed. Special attention is
peritoneum should be some distance away from the
necessary if the obstruction has been caused by ingrowth
first opening.
of the choroid plexus into the ventricular catheter. The
A peritoneal catheter may be torn or it may have
catheter may be attached to the plexus and removing it
slipped intraperitoneally. If it does not disturb the
may provoke intraventricular bleeding. If the catheter
patient and does not irritate the bowels, then it may
adhere to the plexus or brain tissue, then it should not be
be left in place. Otherwise, a laparoscopic recovering is
pulled out with force; in this case, it should be left in
advised.
place and a new ventricular catheter should be inserted.
Fig. 15.13 X-rays of a dislocated peritoneal
catheter. (a) anteroposterior view, (b) lateral
view.
a
b
134
Complications
In some cases, an open revision can be avoided by
“pumping” the shunt via a flushing reservoir that can free
some particles that may have occluded the shunt.
Overdrainage
If overdrainage is symptomatic, then revision surgery can
be performed by implanting a gravity-driven device or a
flow limiter
(see also
▶ Table 15.4). Alternatively, the
valve could be replaced with another valve with a higher
pressure setting. However, it remains difficult to deter-
mine the exact setting of the device preoperatively. There-
fore, a programmable gravitational device is favorable.
If oblique positioning of the valve is the reason for
overdrainage (see ▶ Fig. 15.12), then, of course, the first
step should be the vertical repositioning of the gravity-
assisted device.
In cases of subdural hygromas/hematomas, implanta-
tion of a gravitational device or a flow limiter is an
option. Continuous clinical observation thereafter is man-
datory because it may not be sufficient to stop the grow-
ing of the subdural hygromas/hematomas, thus making a
hematoma evacuation necessary.
Fig. 15.14 Ultrasound of a dislocated peritoneal catheter, with a
subcutaneous pseudocyst filled with cerebrospinal fluid.
Shunt Infection
Wound-Healing Problems/Wound Infections/
a
Skin Dehiscence Over Shunt Material
If the shunt is infected, then the infection usually cannot
be treated sufficiently with antibiotics
(see also
▶ Table 15.4). The reason for this is the biofilm formation
of bacterial colonies on the shunt material; these protect
the microbes against antibodies, white blood cells, and
antibiotics.74-77
Therefore, the infected shunt must be
removed and should be reimplanted after the infection
has been eradicated completely. However, if the infection
is cured completely by antibiotic treatment, then a new
shunt can be implanted on the opposite site, with total
removal of the infected shunt.
If parts of the shunt are exposed and open in skin
ulcerations, then infection of the shunt must be assumed.
Therefore, it should be treated similar to a shunt infection
and the shunt removed and antibiotics used. In rare cases
(e.g., if the patient refuses replacement of the entire
shunt), then changing the exposed shunt parts, antibiotic
treatment, and wound revision could be tried and might
be successful (▶ Fig. 15.16).
In cases of untreatable persistent infections, antibiotic-
impregnated catheters, which generally seem to reduce
the shunt infection rate,78 could be helpful.
Underdrainage Without Obvious Cause
b
Shunt revision is more difficult if no cause of shunt mal-
Fig. 15.15
(a) and (b) Ventricular catheters completely
function is found. Reasons for this could be incorrect valve
obstructed by choroid plexus tissue and fibrin clots, in two
selection, high intraperitoneal pressure, reduced resorp-
different views.
tion capacity of the peritoneum, or scarring around the
135
Complications
For these special types of revisions, some principles
should be considered:
● Avoid unnecessary exposure of functioning shunt
parts.
● Prepare the whole shunt site, so the shunt revision can
be extended to all parts of the shunt.
● Be familiar with the valve used, so that you know how
to check it.
● Use the same sterile precautions as those for normal
shunt surgery.
Beginning the Shunt Revision
Most shunts consist of a ventricular catheter, a valve, and
a distal (usually peritoneal) catheter. To avoid unnecessary
Fig. 15.16 Skin ulceration due to repeated scratching with
shunt exposure during revision, the surgery should
exposed shunt parts 12 months after shunt implantation.
begin at the level of the valve. After the valve is exposed,
the proximal catheter is disconnected from the valve. If
no CSF is running, then the proximal part must be
revised, which is often done by replacing the old ven-
catheter perforations, among others. The latter may
tricular catheter.
appear if any allergic or foreign body reaction against the
shunt material has developed. But, in partial shunt
obstruction or valve failures, this may be difficult or
Blocked Ventricular Catheter
undetectable by imaging. All the potential reasons must
Attention must be paid when removing a “sticking”
be kept in mind when beginning shunt revision surgery.
ventricular catheter. No force should be used to pull
Repeated clinical improvement after spinal tap tests indi-
the catheter, which might stick to the choroid plexus
cates a revision even if the reason for the shunt mal-
or ventricle wall, otherwise intraventricular bleeding
function is not known. In these cases, the surgeons must
might be a risk. Eventually the catheter will remain
be flexible and able to react adequately to different intra-
in place and a new catheter should be placed in the
operative findings (▶ Fig. 15.17).
ventricle.
Fig. 15.17 Algorithm of shunt revision if no
Preperation of the valve, disconnection proximal to the valve
cause of shunt malfunction is found pre-
operatively. CSF, cerebrospinal fluid.
check ventricular catheter
no CSF running
OK
change ventricular
check valve with peritoneal catheter
catheter
defect
OK
check valve alone
defect
OK
check peritoneal catheter
defect
OK
change valve
change
reduce valve
peritoneal
setting,consider atrial
catheter
or pleural diversion
136
Complications
normal-pressure hydrocephalus. Neurosurgery 2005; 57 (Suppl):
Blocked Valve and/or the
S2-1-S2-3
Peritoneal Catheter
[3]
Kehler U, Langer N, Gliemroth J et al. The peel-away sheath hydro-
cephalus study “contactless” ventricular catheter insertion: Does it
If CSF is running out of the proximal catheter, the proxi-
reduce shunt obstructions? A multicenter prospective randomized
mal part of the shunt is fine and not the reason for the
trial. Clin Neurol Neurosurg 2011
shunt malfunction. The valve, together with the perito-
[4]
Meier U, Kiefer M, Sprung C. Normal Pressure Hydrocephalus: Pathol-
neal catheter, must be checked by measuring the opening
ogy, Pathophysiology, Diagnostics, Therapeutics and Clinical Course.
Erwitte: PVV Science Publications; 2003
pressure of the system. In cases of gravity-driven devices,
[5]
Meier U, Kiefer M, Sprung C. Evaluation of the Miethke dual-switch
it is necessary to determine the opening pressure in the
valve in patients with normal pressure hydrocephalus. Surg Neurol
horizontal and vertical positions of the valve to see if the
2004; 61: 119-127, discussion 127-128
gravitational component works properly. The opening
[6]
Sprung C, Schlosser HG, Lemcke J et al. The adjustable proGAV shunt:
a prospective safety and reliability multicenter study. Neurosurgery
pressure, which is intraoperatively determined, should be
2010; 66: 465-474
around 5 to 10 cm H2O higher than the valve setting,
[7]
Azeem SS, Origitano TC. Ventricular catheter placement with a frame-
while still taking into account the intra-abdominal pres-
less neuronavigational system: a 1-year experience. Neurosurgery
sure. Do not perform flushing before measuring the sys-
2007; 60 (Suppl 2): 243-247, discussion 247-248
tem because an obstructing clot may be flushed away or a
[8]
Hayhurst C, Beems T, Jenkinson MD et al. Effect of electromagnetic-
navigated shunt placement on failure rates: a prospective multicen-
membranous structure around the openings of the peri-
ter study. J Neurosurg 2010; 113: 1273-1278
toneal catheter may tear, thus making it impossible to
[9]
Ishikawa M, Hashimoto M, Kuwana N et al. Guidelines for manage-
detect the shunt problem afterwards. If an obstruction or
ment of idiopathic normal pressure hydrocephalus. Neurol Med Chir
a high opening pressure is evident, then the valve must
(Tokyo) 2008; 48 (Suppl): S1-S23
be disconnected from the peritoneal catheter and the
[10]
Drake JM, Sainte-Rose C. The Shunt Book. New York, NY: Blackwell
Science, Inc.; 1995
measurements must be repeated separately for the valve
[11]
Gristina AG. Biomaterial-centered infection: microbial adhesion ver-
and the peritoneal shunt.
sus tissue integration. Science 1987; 237: 1588-1595
If the valve does not work properly, it must be replaced
[12]
Hsieh YL, Merry J. The adherence of Staphylococcus aureus, Staphylo-
with a new one.
coccus epidermidis and Escherichia coli on cotton, polyester and their
blends. J Appl Bacteriol 1986; 60: 535-544
If the peritoneal catheter does not work properly, then
[13]
Liang X, Wang A, Cao T et al. Effect of cast molded rifampicin/silicone
it has to be renewed, preferably with a different perito-
on Staphylococcus epidermidis biofilm formation. J Biomed Mater Res
neal opening to avoid local scars.
A 2006; 76: 580-588
If no mistake can be found intraoperatively, then the
[14]
Costerton JW, Cheng KJ, Geesey GG et al. Bacterial biofilms in nature
valve should be replaced with a new one with a lower
and disease. Annu Rev Microbiol 1987; 41: 435-464
[15]
von Eiff C, Peters G, Heilmann C. Pathogenesis of infections due
pressure setting, preferable with a programmable unit.
to coagulase-negative staphylococci. Lancet Infect Dis
2002; 2:
This would provide the opportunity to make further cor-
677-685
rections during follow-up without additional surgery. In
[16]
Raahave D. Bacterial density in operation wounds. Acta Chir Scand
the case of a repeated shunt malfunction, a different shunt
1974; 140: 585-593
[17]
Kehler U, Klöhn A, Heese O, Gliemroth J. Hydrocephalus therapy:
diverting area (e.g., atrial, pleura) must be considered.
reduction of shunt occlusions using a peel-away sheath. Clin Neurol
Following revision surgery, clinical follow-up with the
Neurosurg 2003; 105: 253-255
patient is mandatory to evaluate the effectiveness of the
[18]
Rivero-Garvía M, Márquez-Rivas J, Jiménez-Mejías ME, Neth O,
procedure and to determine whether further action (e.g.,
Rueda-Torres AB. Reduction in external ventricular drain infection
downprogramming of the valve) is necessary.
rate. Impact of a minimal handling protocol and antibiotic-impreg-
nated catheters. Acta Neurochir (Wien) 2011; 153: 647-651
If there is no clinical improvement, diagnosis of the
[19]
Bayston R, Bannister C, Boston V et al. A prospective randomised con-
shunt malfunction must be repeated. An additional revi-
trolled trial of antimicrobial prophylaxis in hydrocephalus shunt sur-
sion must be considered after a newly positive response
gery. Z Kinderchir 1990; 45 (Suppl 1): 5-7
to a spinal tap test. If still no cause can be found, a cardiac
[20]
Raahave D. Penetration of penicillin into laparotomy wounds to
shunt diverting to the right atrium, or even to the pleural
reduce bacterial contamination during intestinal operations. Scand J
Gastroenterol Suppl 1976; 37: 129-134
cavity, gallbladder, or other organs, may be necessary to
[21]
Kariyattil R, Steinbok P, Singhal A, Cochrane DD. Ascites and
solve a potential absorption deficit of the peritoneum. ICP
abdominal pseudocysts following ventriculoperitoneal shunt sur-
telemetric measurements79
may help to determine
gery: variations of the same theme. J Neurosurg
2007;
106
whether the absence of clinical improvement is corre-
(Suppl): 350-353
[22]
Liu KL, Lee TC, Lin MT, Chen SJ. Education and imaging. Gastro-
lated with ICP and shunt function (▶ Table 15.4).
intestinal: abdominal abscess associated with a ventriculoperitoneal
shunt. J Gastroenterol Hepatol 2007; 22: 757
[23]
Hampl J, Schierholz J, Jansen B, Aschoff A. In vitro and in vivo efficacy
References
of a rifampin-loaded silicone catheter for the prevention of CSF shunt
infections. Acta Neurochir (Wien) 1995; 133: 147-152
[1] Grumme T, Kolodziejczyk D. Komplikationen in der Neurochirurgie.
[24]
Bayston R. Ventriculoperitoneal shunt-associated infection. J Infect
Band 2: Kraniale, zerebrale und neuropädiatrische Chirurgie. Berlin,
1991; 23: 343
Wien: Blackwell Wissenschafts-Verlag; 1995
[25]
Bayston R. Serum C-reactive protein test in diagnosis of septic com-
[2] Marmarou A, Bergsneider M, Black PMcL, Klinge P, Relkin N.
plications of cerebrospinal fluid shunts for hydrocephalus. Arch Dis
Guidelines for the diagnosis and management of idiopathic
Child 1979; 54: 545-548
137
Complications
[26]
Bayston R. Epidemiology, diagnosis, treatment, and prevention of
[50]
Secer HI, Kural C, Kaplan M et al. Comparison of the efficacies of anti-
cerebrospinal fluid shunt infections. Neurosurg Clin N Am 2001; 12:
biotic-impregnated and silver-impregnated ventricular catheters on
703-708, viii
the prevention of infections. An in vitro laboratory study. Pediatr
[27]
Chang CC, Kuwana N, Ito S. Management of patients with normal-
Neurosurg 2008; 44: 444-447
pressure hydrocephalus by using lumboperitoneal shunt system
[51]
Eymann R, Chehab S, Strowitzki M, Steudel WI, Kiefer M. Clinical and
with the Codman Hakim programmable valve. Neurosurg Focus
economic consequences of antibiotic-impregnated cerebrospinal
1999; 7: e8
fluid shunt catheters. J Neurosurg Pediatr 2008; 1: 444-450
[28]
Bret P, Guyotat J, Ricci AC, Mottolese C, Jouanneau E. [Clinical experi-
[52]
Bayston R, Grove N, Siegel J, Lawellin D, Barsham S. Prevention of
ence with the Sophy adjustable valve in the treatment of adult hydro-
hydrocephalus shunt catheter colonisation in vitro by impregnation
cephalus. A series of 147 cases]. Neurochirurgie 1999; 45 (2): 98-
with antimicrobials. J Neurol Neurosurg Psychiatry
1989;
52:
108, discussion 108-109
605-609
[29]
Davis CP. Normal flora. In: Baron S, ed. Medical Microbiology. 4th ed.
[53]
Bayston R, Vera L, Mills A, Ashraf W, Stevenson O, Howdle SM. In
Galveston, TX: University of Texas Medical Branch at Galveston; 1996
vitro antimicrobial activity of silver-processed catheters for neuro-
[30]
Larsson A, Wikkelsö C, Bilting M, Stephensen H. Clinical parameters
surgery. J Antimicrob Chemother 2010; 65: 258-265
in 74 consecutive patients shunt operated for normal pressure hydro-
[54]
Bayston R, Milner RD. Antimicrobial activity of silicone rubber used
cephalus. Acta Neurol Scand 1991; 84: 475-482
in hydrocephalus shunts, after impregnation with antimicrobial sub-
[31]
Walters BC, Hoffman HJ, Hendrick EB, Humphreys RP. Cerebrospinal
stances. J Clin Pathol 1981; 34: 1057-1062
fluid shunt infection. Influences on initial management and subse-
[55]
Abed WT, Alavijeh MS, Bayston R, Shorvon SD, Patsalos PN. An evalu-
quent outcome. J Neurosurg 1984; 60: 1014-1021
ation of the epileptogenic properties of a rifampicin/clindamycin-
[32]
Bayston R, Vera L, Ashraf W. Activity of an antimicrobial hydrocepha-
impregnated shunt catheter. Br J Neurosurg 1994; 8: 725-730
lus shunt catheter against Propionibacterium acnes. Antimicrob
[56]
Lackner P, Beer R, Broessner G et al. Efficacy of silver nanoparticles-
Agents Chemother 2010; 54: 5082-5085
impregnated external ventricular drain catheters in patients with
[33]
Livni G, Yuhas Y, Ashkenazi S, Michowiz S. In vitro bacterial adherence
acute occlusive hydrocephalus. Neurocrit Care 2008; 8: 360-365
to ventriculoperitoneal shunts. Pediatr Neurosurg 2004; 40: 64-69
[57]
Izci Y, Secer H, Akay C, Gonul E. Initial experience with silver-impreg-
[34]
Sandoe JA, Longshaw CM. Ventriculoperitoneal shunt infection
nated polyurethane ventricular catheter for shunting of cerebrospinal
caused by Staphylococcus lugdunensis. Clin Microbiol Infect 2001; 7:
fluid in patients with infected hydrocephalus. Neurol Res 2009; 31:
385-387
234-237
[35]
Greenberg JO, Shenkin HA, Adam R. Idiopathic normal pressure
[58]
Lemcke J, Depner F, Meier U. The impact of silver nanoparticle-coated
hydrocephalus— a report of 73 patients. J Neurol Neurosurg Psychia-
and antibiotic-impregnated external ventricular drainage catheters
try 1977; 40: 336-341
on the risk of infections: a clinical comparison of 95 patients. Acta
[36]
Echizenya K, Satoh M, Murai H, Ueno H, Abe H, Komai T. Mineraliza-
Neurochir Suppl (Wien) 2012; 114: 347-350
tion and biodegradation of CSF shunting systems. J Neurosurg 1987;
[59]
Lansdown AB. Critical observations on the neurotoxicity of silver. Crit
67: 584-591
Rev Toxicol 2007; 37: 237-250
[37]
Schuhmann MU, Ostrowski KR, Draper EJ et al. The value of C-
[60]
Galiano K, Pleifer C, Engelhardt K et al. Silver segregation and bacte-
reactive protein in the management of shunt infections. J Neurosurg
rial growth of intraventricular catheters impregnated with silver
2005; 103 (Suppl): 223-230
nanoparticles in cerebrospinal fluid drainages. Neurol Res 2008; 30:
[38]
Bayston R, Leung TS, Wilkins BM, Hodges B. Bacteriological examina-
285-287
tion of removed cerebrospinal fluid shunts. J Clin Pathol 1983; 36:
[61]
Kouyialis AT, Stranjalis G, Korfias S, Sakas DE. Long-term air-exposed
987-990
functioning hydrocephalus valve with no infection. South Med J
[39]
Bayston R, Ashraf W, Barker-Davies R et al. Biofilm formation by Pro-
2006; 99: 1127-1129
pionibacterium acnes on biomaterials in vitro and in vivo: impact on
[62]
Gristina A. Biomaterial-centered infection: microbial adhesion versus
diagnosis and treatment. J Biomed Mater Res A 2007; 81: 705-709
tissue integration. 1987. Clin Orthop Relat Res 2004: 4-12
[40]
Lund-Johansen M, Svendsen F, Wester K. Shunt failures and complica-
[63]
James HE, Walsh JW, Wilson HD, Connor JD, Bean JR, Tibbs PA. Pro-
tions in adults as related to shunt type, diagnosis, and the experience
spective randomized study of therapy in cerebrospinal fluid shunt
of the surgeon. Neurosurgery 1994; 35: 839-844, discussion 844
infection. Neurosurgery 1980; 7: 459-463
[41]
Bayston R. Hydrocephalus shunt infections. J Antimicrob Chemother
[64]
Walters BC, Hoffman HJ, Hendrick EB, Humphreys RP. Cerebrospinal
1994; 34 (Suppl A): 75-84
fluid shunt infection. Influences on initial management and subse-
[42]
Bayston R. Hydrocephalus shunt infections and their treatment. J
quent outcome. J Neurosurg 1984; 60: 1014-1021
Antimicrob Chemother 1985; 15: 259-261
[65]
Gill CJ, Murphy MA, Hamer DH. Treatment of Staphylococcus epider-
[43]
Bayston R, Ashraf W, Bhundia C. Mode of action of an antimicrobial
midis ventriculo-peritoneal shunt infection with linezolid. J Infect
biomaterial for use in hydrocephalus shunts. J Antimicrob Chemother
2002; 45: 129-132
2004; 53: 778-782
[66]
Shin DS, Hwang SC, Kim BT, Shin WH. Delayed cerebral abscess as a
[44]
Bayston R, Bannister C, Boston V et al. A prospective randomised con-
shunt complication and endoscopic removal of the ventricular cathe-
trolled trial of antimicrobial prophylaxis in hydrocephalus shunt sur-
ter and abscess. J Korean Neurosurg Soc 2008; 43: 300-303
gery. Z Kinderchir 1990; 45 (Suppl 1): 5-7
[67]
Irani F, Elkambergy H, Okoli K, Abou DS. Recurrent symptomatic
[45]
Bayston R, Lari J. A study of the sources of infection in colonised
pleural effusion due to a ventriculopleural shunt. Respir Care 2009;
shunts. Dev Med Child Neurol 1974; 16 (Suppl 32): 16-22
54: 1112-1114
[46]
Brydon HL, Bayston R, Hayward R, Harkness W. Reduced bacterial
[68]
Yamashita K, Yonekawa Y, Kawano T et al. Intra-abdominal cyst fol-
adhesion to hydrocephalus shunt catheters mediated by cerebrospi-
lowing revision of ventriculoperitoneal shunt—case report. Neurol
nal fluid proteins. J Neurol Neurosurg Psychiatry 1996; 60: 671-675
Med Chir (Tokyo) 1990; 30: 748-752
[47]
Brydon HL, Bayston R, Hayward R, Harkness W. The effect of increas-
[69]
Meier U, Mutze S. Correlation between decreased ventricular size
ing CSF protein concentration upon shunt function. Eur J Pediatr Surg
and positive clinical outcome following shunt placement in patients
1993; 3 (Suppl 1): 33-34
with normal-pressure hydrocephalus. J Neurosurg 2004; 100: 1036-
[48]
Fan-Havard P, Nahata MC. Treatment and prevention of infections of
1040
cerebrospinal fluid shunts. Clin Pharm 1987; 6: 866-880
[70]
Aschoff A, Kremer P, Benesch C, Fruh K, Klank A, Kunze S. Overdrain-
[49]
Brydon HL, Hayward R, Harkness W, Bayston R. Does the cerebrospi-
age and shunt technology. A critical comparison of programmable,
nal fluid protein concentration increase the risk of shunt complica-
hydrostatic and variable-resistance valves and flow-reducing devices.
tions? Br J Neurosurg 1996; 10: 267-273
Childs Nerv Syst 1995; 11: 193-202
138
Complications
[71]
Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
[81]
Meier U, Lemcke J, Al-Zain F. Course of disease in patients with idio-
lus Study: the role of cerebrovascular disease. J Neurosurg 1999; 90:
pathic normal pressure hydrocephalus (iNPH): a follow-up study 3, 4
221-226
and 5 years following shunt implantation. Acta Neurochir Suppl
[72]
Meier U, Kiefer M, Lemcke J. On the optimal opening pressure of
(Wien) 2008; 102: 125-127
hydrostatic valves in cases of idiopathic normal-pressure hydroceph-
[82]
McGirt MJ, Woodworth G, Coon AL, Thomas G, Williams MA, Riga-
alus: a Prospective Randomized Study With 122 Patients. Neurosurg
monti D. Diagnosis, treatment, and analysis of long-term outcomes
Q 2005; 15: 103-109
in idiopathic normal-pressure hydrocephalus. Neurosurgery 2008;
[73]
Vassilyadi M, Tataryn ZL, Matzinger MA, Briggs V, Ventureyra EC.
62
(Suppl 2): 670-677
Radioisotope shuntograms at the Children’s Hospital of Eastern
[83]
Kahlon B, Sjunnesson J, Rehncrona S. Long-term outcome in patients
Ontario. Childs Nerv Syst 2006; 22: 43-49
with suspected normal pressure hydrocephalus. Neurosurgery 2007;
[74]
Nickel JC, Wright JB, Ruseska I, Marrie TJ, Whitfield C, Costerton JW.
60: 327-332, discussion 332
Antibiotic resistance of Pseudomonas aeruginosa colonizing a urinary
[84]
Marmarou A, Young HF, Aygok GA et al. Diagnosis and management
catheter in vitro. Eur J Clin Microbiol 1985; 4: 213-218
of idiopathic normal-pressure hydrocephalus: a prospective study in
[75]
Nickel JC, Ruseska I, Wright JB, Costerton JW. Tobramycin resistance
151 patients. J Neurosurg 2005; 102: 987-997
of Pseudomonas aeruginosa cells growing as a biofilm on urinary
[85]
Sorteberg A, Eide PK, Fremming AD. A prospective study on the clini-
catheter material. Antimicrob Agents Chemother 1985; 27: 619-624
cal effect of surgical treatment of normal pressure hydrocephalus:
[76]
Khoury AE, Lam K, Ellis B, Costerton JW. Prevention and control of
the value of hydrodynamic evaluation. Br J Neurosurg 2004; 18:
bacterial infections associated with medical devices. ASAIO J 1992;
149-157
38: M174-M178
[86]
Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
[77]
Drake JM, Sainte-Rose Ch. The Shunt Book. Cambridge, MA: Blackwell
lus Study: randomized comparison of low- and medium-pressure
Science; 1995
shunts. J Neurosurg 1998; 88: 490-495
[78]
Eymann R, Steudel WI, Kiefer M. Infection rate with application of an
[87]
Eide PK, Sorteberg W. Diagnostic intracranial pressure monitoring
antibiotic-impregnated catheter for shunt implantation in children -
and surgical management in idiopathic normal pressure hydro-
a retrospective analysis. Klin Padiatr 2009; 221: 69-73
cephalus: a 6-year review of 214 patients. Neurosurgery 2010;
[79]
Frischholz M, Sarmento L, Wenzel M, Aquilina K, Edwards R, Coak-
66: 80-91
ham HB. Telemetric implantable pressure sensor for short- and long-
[88]
Pujari S, Kharkar S, Metellus P, Shuck J, Williams MA, Rigamonti D.
term monitoring of intracranial pressure. Conf Proc IEEE Eng Med
Normal pressure hydrocephalus: long-term outcome after shunt sur-
Biol Soc 2007; 2007: 514
gery. J Neurol Neurosurg Psychiatry 2008; 79: 1282-1286
[80]
Mirzayan MJ, Luetjens G, Borremans JJ, Regel JP, Krauss JK. Extended
[89]
Zemack G, Romner B. Adjustable valves in normal-pressure hydro-
long-term (> 5 years) outcome of cerebrospinal fluid shunting in idio-
cephalus: a retrospective study of 218 patients. Neurosurgery 2008;
pathic normal pressure hydrocephalus. Neurosurgery 2010; 67: 295-301
62
(Suppl 2): 677-687
139
16.1
Predictors
142
Chapter 16
16.2
Improvement of Prognosis
142
Prognosis
Ullrich Meier
16
Prognosis
16 Prognosis
Ullrich Meier
The diagnostic evaluation for surgical intervention is a
According to Chang et al,7 advanced age and male gen-
central problem in the treatment of normal pressure
der constitute negative predictors in relation to an
hydrocephalus
(NPH). In this context, the statement
improvement in cognitive function following a shunt
“Results are good if the indications are correct” still
operation in patients with NPH. Other authors8-13 have
applies. There are quite substantial differences in reports
drawn attention to the importance of ataxic gait as the
of the results of treatments following shunt implantation.
characteristic symptom of NPH and its improvement fol-
The reasons for this are the different views regarding the
lowing a cerebrospinal tap test or lumbar cerebrospinal
diagnostic evaluation for shunt implantation. Therefore,
fluid drainage, as well as the symptom with the highest
predictors must be found that have a high degree of
rate of improvement in the further course of the illness
predictive value in relation to the postoperative course.1
following shunt operation. In contrast to all of the previ-
ously mentioned authors, Delwel et al14 found no individ-
ual predictors with regard to the postoperative course
16.1 Predictors
following shunt operation. Also, parameters of the intra-
thecal infusion test, such as drainage resistance (Rout),
Little or no expression of dementia or amnestic symp-
were not helpful in relation to the results of the previ-
toms and the presence of iNPH symptoms for less than
ously mentioned research groups with regard to prognos-
one year are positive predictors of the course of disease
tic evaluation. Other authors have described comorbidity
after shunt implantation. Patients with NPH in an early
(arterial hypertension, diabetes mellitus, peripheral and/
stage (with no atrophy of the brain) and a drainage resist-
or coronary large vessel diseases, cardiac diseases, cere-
ance (Rout) of more than 15 Torr/mL/min, or patients with
brovascular diseases, Parkinson disease) as a negative and
NPH in the late stage (with atrophy of the brain) and a
statistically significant predictor of the quality of the clin-
drainage resistance (Rout) of more than 20 Torr/mL/min as
ical course of the disease in patients with iNPH following
a result of the intrathecal infusion test, have a positive
10,15,16
shunt therapy.
prognosis. Because of better treatment results, the
implantation of a hydrostatic valve is recommended in
patients with NPH.2
Sorteberg et al3 showed that drainage resistance (Rout)
16.2 Improvement
using an intrathecal infusion test is a good predictor of
of Prognosis
the postoperative course following a shunt operation for
idiopathic NPH
(iNPH). In contrast, the authors
A meta-analysis within the scope of the guidelines for
described the intracranial pressure (ICP) measurement
the management of iNPH of the Japanese Neurosurgeons
as having no predictive value. Poca et al4 came to the
Society reported improvement in 31% to 100% of the
conclusion that, following a shunt operation in patients
patients between 3 months to 2 years after shunt sur-
with NPH, a good improvement in ataxic gait and uri-
gery. A longer follow-up period of 3 to 5 years produces
nary incontinence can be achieved. In contrast, it is not
an improvement rate of 61% to 91%.17 Depending on the
possible to achieve a significant improvement in mental
individual symptoms, there is an improvement in disor-
function with this surgical therapy. For this reason,
ders of gait in 58% to 90% and the amnestic symptoms or
those authors consider the presence of dementia to be a
the symptoms of dementia in 29% to 80% of patients,
negative predictor. Murakami et al5 consider the mea-
and the symptom of urinary incontinence in 20% to 78%
surement of regional cerebral blood flow using single-
of patients.17
Patients with the classical Hakim triad
photon emission computed tomography diagnostics to
show an improvement rate of 65% to 74%.17 In the guide-
have a positive predictive value in relation to the post-
lines of the U.S. iNPH Study Group,18 a rate of improve-
operative course of the illness. Gallia et al6 reported the
ment of 72% to 80% in iNPH following a shunt operation
cerebrospinal tap test, with an improvement in ataxic
has been reported. One year after surgical therapy, 30%
gait, to have a positive predictive value of 73% to 100%.
to 95% of the patients continued to show improvement.
However, there was a low sensitivity for this method of
The study reported an improvement rate of 78.9%, 2 to 3
investigation, with a value of 26% to 61%, whereas a neg-
years after the shunt operation, and a rate of 43%, 5
ative cerebrospinal tap test cannot exclude the presence
years after the shunt operation. In relation to the indi-
of NPH. For this reason, the authors favor lumbar CSF
vidual symptoms and a comparison of 5 years versus 1
drainage over a period of 48 hours with a positive
year after the operation, it has been reported that the
predictive value of 80% to 100%. With this method of
improvement in 76% of patients was reduced to 47% of
investigation, the authors quote a sensitivity of 50% to
patients and in those with urinary incontinence from
100% and a specificity of 60% to 100%.
58% to 29%.18
142
Prognosis
?
Table 16.1 Improvement ratea
Authors, year
No. of
Type of valve/valve pressure setting
Postinvestigation
No. of patients show-
patients
period (months)b
ing improvement in
with iNPH
clinical symptoms (%)
Black 198022
62
HAKIM medium pressure valve (DPV)
36.5
61
Boon et al 200023
95
HAKIM low and medium pressure valve (DPV)
12
76
Delwel et al 200514
66
Spitz-Holter medium pressure valve (DPV)
≥ 12
59
Dixon et al 200224
49
Unknown
10
(1-36)
44-86
(symptom-related)
Kahlon et al 200225
51
CMPV
6
84
Kiefer et al 200226
91
MDSV and CMPV with Miethke shunt assistant
26
85
Krauss and Regel 199727
41
DPV and CMPV
16
90
Marmarou et al 200528
151
Unknown
12
66.2
McConnell et al 200429
51
CMPV
11
(1-49)
68.8-86.3
(symptom-related)
Meier and Lemcke
117
MDSV, 64 patients proGAV, 34 patients pCHV
26.9
(12-60)
1 year: 88.9
20071,21
with MIETHKE shunt assistant, 20 patients
5 years: 60
Mori 200130
120
DPV versus CMPV
36
73.3
Poca et al
43
Low-pressure valve (DPV)
6
39.5-81.4
200431
(symptom-related)
Walchenbach et al
43
CMPV and CODMAN MEDOS valve (DPV)
2, 6, 12
50-91
200219
(symptom-related)
Zemack and Romner
147
CMPV
26.7
78.9
200232
aNumber of patients more than 40 and observation period 6 months or more.
bThe range is shown in parentheses.
Abbreviations: DPV, differential pressure valve; iNPH, idiopathic normal pressure hydrocephalus; CMPV, Codman Medos Programmable
▶ Table
16.1
summarizes the results of treatment
The efforts of the physicians providing treatment should
according to the available literature. In this context, only
be directed at verifying the diagnosis of NPH at an early
those citations in the literature that have patient groups
stage when there is an ataxic gait but no urinary
of more than 40 and whose periods of investigation were
incontinence and no dementia disorders, as well as no or
6 months or more have been listed. In summary, it can be
little marked comorbidity, and, in so doing, the results of
said that, in the case of patients with iNPH 1 year follow-
therapy following shunt implantation will clearly be bet-
ing the shunt operation, an improvement in the pre-
ter. A delay in diagnosis produces, in unfavorable cases,
operative symptoms was seen in
59% to
91% of
an irreversible reduction in cognitive potential. A loss of
patients.14,19 The rate of improvement 2 to 3 years after
time means also a loss of brain: “Time is brain!” For this
the shunt operation was 61% to 85%.20 Our own research
reason, sensitive diagnostics and the therapy resulting
results, which included 5 years of follow-up after the
from these are essential for the future course of NPH.
shunt operation, produced an improvement rate of 60%.21
A consideration of the medium-term course of the dis-
ease in patients with iNPH is difficult because of the sub-
References
stantial influence of comorbidity. In the case of our own
[1] Meier U, Lemcke J. Zur Diagnostik des idiopathischen Normaldruck-
population of patients,1 10% of the patients had died 2
hydrozephalus aus der Perspektive von Langzeitbeobachtungen.
years after operation. Other authors have reported a
Schweiz Arch Neurol Psychiatr 2007; 158: 139-149
[2] Meier U, König A, Miethke C. Predictors of outcome in patients with
death rate of 28% within a time period of 3 years.18
normal-pressure hydrocephalus. Eur Neurol 2004; 51: 59-67
Because of the advanced age of the patients with iNPH
[3] Sorteberg A, Eide PK, Fremming AD. A prospective study on the clini-
and the influence of comorbid factors, the future progno-
cal effect of surgical treatment of normal pressure hydrocephalus:
sis of the course of the disease 5 to 10 years following the
the value of hydrodynamic evaluation. Br J Neurosurg 2004; 18: 149-
shunt operation will also be difficult or impossible to
157
[4] Poca MA, Mataró M, Matarín M, Arikan F, Junqué C, Sahuquillo J.
make. In any case, it can be assumed that nontreated
Good outcome in patients with normal-pressure hydrocephalus and
patients with iNPH show a rapid increase in clinical
factors indicating poor prognosis. J Neurosurg 2005; 103: 455-463
symptoms and that the majority will end up as patients
[5] Murakami M, Hirata Y, Kuratsu JI. Predictive assessment of shunt effec-
requiring nursing care as well as dementia care.
tiveness in patients with idiopathic normal pressure hydrocephalus by
143
Prognosis
determining regional cerebral blood flow on 3D stereotactic surface
shunting on normal pressure hydrocephalus. J Neurol Neurosurg
projections. Acta Neurochir (Wien) 2007; 149: 991-997
Psychiatry 2002; 72: 503-506
[6]
Gallia GL, Rigamonti D, Williams MA. The diagnosis and treatment of
[20]
Meier U, Lemcke J. Clinical outcome of patients with idiopathic
idiopathic normal pressure hydrocephalus. Nat Clin Pract Neurol
normal pressure hydrocephalus three years after shunt implantation.
2006; 2: 375-381
Acta Neurochir Suppl (Wien) 2006; 96: 377-380
[7]
Chang S, Agarwal S, Williams MA, Rigamonti D, Hillis AE. Demo-
[21]
Meier U, Lemcke J, Al-Zain F. Course of disease in patients with idio-
graphic factors influence cognitive recovery after shunt for normal-
pathic normal pressure hydrocephalus (iNPH): a follow-up study 3, 4
pressure hydrocephalus. Neurologist 2006; 12: 39-42
and 5 years following shunt implantation. Acta Neurochir Suppl
[8]
Duinkerke A, Williams MA, Rigamonti D, Hillis AE. Cognitive recovery
(Wien) 2008; 102: 125-127
in idiopathic normal pressure hydrocephalus after shunt. Cogn Behav
[22]
Black PM. Idiopathic normal-pressure hydrocephalus. Results of
Neurol 2004; 17: 179-184
shunting in 62 patients. J Neurosurg 1980; 52: 371-377
[9]
Graff-Radford NR, Godersky JC. Normal-pressure hydrocephalus.
[23]
Boon AJ, Tans JT, Delwel EJ et al. The Dutch normal-pressure hydro-
Onset of gait abnormality before dementia predicts good surgical
cephalus study. How to select patients for shunting? An analysis of
outcome. Arch Neurol 1986; 43: 940-942
four diagnostic criteria. Surg Neurol 2000; 53: 201-207
[10]
Kiefer M, Eymann R, Steudel WI. Outcome predictors for normal-
[24]
Dixon GR, Friedman JA, Luetmer PH et al. Use of cerebrospinal fluid
pressure hydrocephalus. Acta Neurochir Suppl (Wien) 2006; 96:
flow rates measured by phase-contrast MR to predict outcome of
364-367
ventriculoperitoneal shunting for idiopathic normal-pressure hydro-
[11]
Sand T, Bovim G, Grimse R, Myhr G, Helde G, Cappelen J. Idiopathic
cephalus. Mayo Clin Proc 2002; 77: 509-514
normal pressure hydrocephalus: the CSF tap-test may predict the
[25]
Kahlon B, Sundbärg G, Rehncrona S. Comparison between the lumbar
clinical response to shunting. Acta Neurol Scand 1994; 89: 311-316
infusion and CSF tap tests to predict outcome after shunt surgery in
[12]
Savolainen S, Hurskainen H, Paljärvi L, Alafuzoff I, Vapalahti M. Five-
suspected normal pressure hydrocephalus. J Neurol Neurosurg
year outcome of normal pressure hydrocephalus with or without a
Psychiatry 2002; 73: 721-726
shunt: predictive value of the clinical signs, neuropsychological eval-
[26]
Kiefer M, Eymann R, Meier U. Five years experience with gravitational
uation and infusion test. Acta Neurochir (Wien) 2002; 144: 515-523,
shunts in chronic hydrocephalus of adults. Acta Neurochir (Wien)
discussion 523
2002; 144: 755-767, discussion 767
[13]
Vassilouthis J. Poor prognosis/good outcome in patients with NPH. J
[27]
Krauss JK, Regel JP. The predictive value of ventricular CSF removal in
Neurosurg 2006; 104: 986-988, author reply 986-988
normal pressure hydrocephalus. Neurol Res 1997; 19: 357-360
[14]
Delwel EJ, de Jong DA, Avezaat CJ. The prognostic value of clinical
[28]
Marmarou A, Young HF, Aygok GA et al. Diagnosis and management
characteristics and parameters of cerebrospinal fluid hydrodynamics
of idiopathic normal-pressure hydrocephalus: a prospective study in
in shunting for idiopathic normal pressure hydrocephalus. Acta
151 patients. J Neurosurg 2005; 102: 987-997
Neurochir (Wien) 2005; 147: 1037-1042, discussion 1042-1043
[29]
McConnell KA, Zou KH, Chabrerie AV, Bailey NO, Black PM. Decreases
[15]
Kiefer M, Meier U, Eymann R. Gravitational valves: relevant differ-
in ventricular volume correlate with decreases in ventricular pres-
ences with different technical solutions to counteract hydrostatic
sure in idiopathic normal pressure hydrocephalus patients who
pressure. Acta Neurochir Suppl (Wien) 2006; 96: 343-347
experienced clinical improvement after implantation with adjustable
[16]
Meier U, Lemcke J. Is it possible to optimize treatment of patients
valve shunts. Neurosurgery 2004; 55: 582-592, discussion 592-593
with idiopathic normal pressure hydrocephalus by implanting an
[30]
Mori K. Management of idiopathic normal-pressure hydrocephalus: a
adjustable Medos Hakim valve in combination with a Miethke shunt
multiinstitutional study conducted in Japan. J Neurosurg 2001; 95:
assistant? Acta Neurochir Suppl (Wien) 2006; 96: 381-385
970-973
[17]
Ishikawa M, Hashimoto M, Kuwana N et al. Guidelines for manage-
[31]
Poca MA, Mataró M, Del Mar Matarín M, Arikan F, Junqué C, Sahu-
ment of idiopathic normal pressure hydrocephalus. Neurol Med Chir
quillo J. Is the placement of shunts in patients with idiopathic nor-
(Tokyo) 2008; 48 (Suppl): S1-S23
mal-pressure hydrocephalus worth the risk? Results of a study based
[18]
Marmarou A, Bergsneider M, Relkin N, Klinge P, Black PM. Develop-
on continuous monitoring of intracranial pressure. J Neurosurg 2004;
ment of guidelines for idiopathic normal-pressure hydrocephalus:
100: 855-866
introduction. Neurosurgery 2005; 57 (Suppl): S1-S3, discussion ii-v
[32]
Zemack G, Romner B. Adjustable valves in normal-pressure hydro-
[19]
Walchenbach R, Geiger E, Thomeer RT, Vanneste JA. The value of tem-
cephalus: a retrospective study of 218 patients. Neurosurgery 2002;
porary external lumbar CSF drainage in predicting the outcome of
51: 1392-1400, discussion 1400-1402
144
17.1
Educating Medical Personnel
146
Chapter 17
17.2
Educating the Public
146
Summary and Future
17.3
Role of Biomarkers
147
Perspectives
17.4
Diagnostic Tools and Prediction
Michael J. Fritsch
of Shunt Responsiveness
147
17.5
Shunt Technology Improvement
147
Summary and Future Perspectives
17 Summary and Future Perspectives
Michael J. Fritsch
Normal pressure hydrocephalus (NPH) is a clinical condi-
broadened, not simply by establishing the diagnosis early,
tion of people aged 65 years and older, and it is character-
but by differentiating NPH from other findings (ventriculo-
ized by an enlargement of the ventricles
(Evans
megaly without clinical findings) or diseases (Alzheimer
index ≥ 0.3) and the Hakim triad
(gait impairment,
dementia, vascular dementia, Parkinson disease, Huntington
dementia, urinary incontinence).1 Gait impairment is the
disease, spongiform encephalopathy, multiple-system
primary symptom and the first symptom to improve after
atrophy, corticobasal atrophy) and following up patients
treatment. Dementia and urinary incontinence are two-
after surgical treatment (and possibly more and more with
fold less likely to improve.2
regard to valve adjustments to fit patient needs), all of
The diagnostic procedures of choice are the spinal tap
which have gained increasing importance.10
test or a lumbar drainage for 1 to 3 days.3 Additional tests
Interdisciplinary NPH clinics, staffed by neurologists,
are the lumbar infusion test and overnight intracranial
neurosurgeons, neuropsychologists, nurses, and social
pressure (ICP) monitoring. The treatment of choice is the
workers, may be one concept for the future to meet the
placement of a ventriculoperitoneal (VP) shunt. Cerebro-
increasing demand.
spinal fluid (CSF) shunting is safe and effective, with a
An excellent example that illustrates this topic is a study
long-term shunt response rate of approximately 75%.2,4
published by Harold O. Conn,11 who is a retired faculty
Outcome is improved with treatment early after the
member of the Yale University School of Medicine. Interest-
onset of symptoms, with low comorbidity at the time of
ingly enough, the author of the paper also had NPH. Soon
initial treatment, and low opening pressure on the
after retirement in 1992, he developed slowness and clumsi-
implanted valve.5,6 Modern shunt technology (adjustabil-
ness of gait, later urinary incontinence, short-term memory
ity, overdrainage protection, impregnated antimicrobial
loss, slow responsiveness, and decrease in reaction time and
catheters) enables an improvement of the benefit-to-risk
mental sharpness. He was diagnosed by one neurologist with
ratio for treated patients.
cerebral atrophy and by another colleague with Parkinson
On the one hand, NPH is underdiagnosed, while, on the
disease. He did not respond to pharmaceutical therapy.
other hand, the elderly population (those older than 65
The correct diagnosis of NPH was finally made in 2003.
years of age) is steadily increasing in industrialized coun-
By that time, he was virtually unable to walk and asked
tries. So, what are the future developments and demands
for authorization for an electric scooter. Since this request
of NPH?
was refused, he was referred to another neurologist who
established the diagnosis based on magnetic resonance
imaging (MRI) and significant improvement following a
17.1 Educating Medical
spinal tap test. A week later, a ventriculoperitoneal shunt
was implanted, virtually restoring him to normal health.
Personnel
Conn recognized the lack of awareness of NPH by many
physicians and initiated a survey to explore the situation.
Education of medical personnel is the key point in
Using a questionnaire, he interviewed
166 practicing
improving patient care and advancing scientific knowl-
physicians regarding their knowledge of NPH.11,12 The
edge-based treatment. This book is part of that effort.
interviewed candidates had graduated from 50 American
Idiopathic NPH (iNPH) is underdiagnosed, and an esti-
and 33 international medical schools. Nearly one-third of
mated 80% of NPH cases remain unrecognized.7 Family
the physicians had never heard of NPH, about
20%
physicians, neurologists, and psychiatrists are in a key
learned about NPH in medical school, and about 50%
position to differentiate NPH from alternative diagnoses
learned about it after medical school. One has to take into
and to refer patients for treatment to a neurosurgeon.8,9
consideration that about one-half of the physicians grad-
Of utmost importance is disease awareness.
uated from medical school before NPH was described in
Results of shunt treatment are better if patients are
the literature, which first occurred in 1965.
treated early in the course of the disease.2 A cut-off point
to achieve the best possible results is at about 1 year after
the onset of symptoms. This observation stresses the
17.2 Educating the Public
above-mentioned responsibility of family physicians,
neurologists, psychiatrists, and neurosurgeons.
Educating the public is as important as educating medical
NPH is a chronic condition, meaning that patients are
personnel. Similar to other endemic diseases in industri-
not “cured” after shunt surgery, and their condition will
alized countries
(e.g., hypertension, diabetes, stroke,
deteriorate again after a certain time period (usually within
cancer, Alzheimer disease), which are quite present in
3-5 years). Therefore, the potential role for neurologists
the public’s consciousness, there needs to be further
and family physicians in the long-term management has
education about NPH.
146
Summary and Future Perspectives
Low back pain catches public attention every other
Characteristic morphologic features on MRI in combi-
week in newspapers or the electronic media. Gait impair-
nation with positive clinical tests (tap test) have been
ment with an onset beyond 65 years of age needs to have
examined in several studies with regard to their positive
similar attention. “Is there a cure for dementia?” could be
predictive value to define shunt responders. Characteris-
one of the provocative slogans to promote awareness of
tic MRI findings for patients with NPH include the frontal
the Hakim triad.
and parietal narrowing of the subarachnoid space, the
“Public” not only means possible patients and their
upward bowing of the corpus callosum, and the marked
families but also nursing home staff or other inpatient or
dilatation of the sylvian fissure.14 In particular, the “mis-
outpatient care facilities for the elderly population.
match” between the narrowing of the subarachnoid space
In his publication about his personal experience with
at the high-convexity with the widening of the sylvian
the diagnosis and treatment of NPH, Conn discusses that
fissure on fluid-attenuated inversion recovery
(FLAIR)
the criteria used to define ventricular enlargement are dif-
sequences seems to have a strong positive predictive
ficult to establish.12 Even more difficult to establish is the
value.
physician’s response to what is considered mild or moder-
Single-photon emission computed tomography data
ate ventricular enlargement. The diagnosis may raise com-
before and after NPH shunt operations have compared
plex medical and socioeconomic issues about hospital
regional cerebral blood flow (rCBF) in responders and
stays, invasive diagnostic procedures, and even brain sur-
nonresponders (based on clinical examination). Respond-
gery. Educating the public as well as physicians should
ers manifested a significantly lower rCBF in the basal
lead to an adequate and competent specialist evaluation of
frontal lobes and cingulate gyrus.15
a patient with mild ventricular enlargement and moderate
symptoms. Elderly patients with symptoms, as well as
their families, should be given the opportunity to make an
17.5 Shunt Technology
informed decision if they want a diagnostic evaluation
Improvement
and, if indicated, shunt surgery or not, whatever the risk.
Conn points out that having experienced end-stage NPH
Further improvement on shunt technology will have an
and having been abandoned to his disease, he did not feel
impact on the treatment of patients with NPH. The main
that there was much to lose by choosing surgery.12
topics are adjustability of the valves while maintaining
patient safety (MRI safe valves), units or devices that
reduce or prevent overdrainage (and the adjustability of
17.3 Role of Biomarkers
such units), catheter impregnation, reducing infection
risk, and new telemetric pressure monitoring devices that
The selection of surgical candidates is one of the challenges
will allow health care professionals to obtain real-time
of NPH management. Appropriate diagnostic tools have
pressure data from patients, either before or after shunt
developed over the past 40 years. So far, biologic markers
surgery, for further optimal adjustment of the valve.
have been overlooked in this matter, which is in contrast to
Three different types of hydrostatic devices are cur-
other neurodegenerative or neuroinflammatory diseases.
rently in clinical use: valves with a so-called antisiphon
Biomarkers may be used to indicate the onset, the presence,
device mechanism, flow-reducing devices, and gravita-
or the progression of a clinical condition. In such a setting,
tional valves. Although patients shunted 16 hours per day
biomarkers may assist in the more appropriate selection of
(or even longer) with hydrocephalus can walk, move, or
patients for shunt surgery. This could work in combination
sit in an upright position, today the vast majority of cases
with other clinical tests, invasive or noninvasive.
have simple adjustable or nonadjustable differential-
Tumor necrosis factor, tau protein, lactate, sulfatide,
pressure valve implants. These systems normally address
and neurofilament triple protein are the most promising
only the horizontal position of the patient, establishing
CSF markers for chronic hydrocephalus. However, at pres-
nonphysiologic pressure conditions in the ventricular
ent, none of these biomarkers meets the criteria to justify
system of the patient during the day. Despite the fact that
routine use in clinical practice. Future studies will be
most of the patients benefit from being shunted with
needed to obtain substantial data.13
these systems without any (severe) complication, this is
very likely to be the reason why hydrostatic valves or
components are most often seen as additional implants,
17.4 Diagnostic Tools and
which are considered as a solution in case of difficulties.
Prediction of Shunt
The task of a consequently more physiologic control of
ICP is often neglected in light of the fact that shunt place-
Responsiveness
ment is one of the most successful neurosurgical opera-
Different diagnostic studies may gain future importance
tions. Nevertheless, the sequelae of overdrainage offer a
in the diagnostic work-up, as well as in the prediction of
wide range of serious complications for neonates, chil-
shunt responsiveness.
dren, and adults. Thickening of the skull as a consequence
147
Summary and Future Perspectives
of overshunting during early childhood is definitely a
cerebrospinal fluid outflow controlled by medium pressure valve, in
normal pressure hydrocephalus. Acta Neurochir (Wien) 2005; 147:
severe long-term complication that cannot be easily
953-958, discussion 958
treated. Patients with NPH sometimes experience sub-
[4]
Hebb AO, Cusimano MD. Idiopathic normal pressure hydrocephalus:
dural hematoma, which can worsen the quality of life or
a systematic review of diagnosis and outcome. Neurosurgery 2001;
the clinical status in comparison with the situation before
49: 1166-1184, discussion 1184-1186
surgery. Boon et al5 found subdural effusions in 53% and
[5]
Boon AJ, Tans JT, Delwel EJ et al. Dutch Normal-Pressure Hydrocepha-
lus Study: the role of cerebrovascular disease. J Neurosurg 1999; 90:
70% of patients with NPH and shunted with medium/
221-226
low-pressure and differential-pressure valves, respec-
[6]
Meier U, Kiefer M, Lemcke J. On the optimal opening pressure of
tively. Facts like these give credence to the systematic
hydrostatic valves in cases of idiopathic normal-pressure hydroceph-
importance of hydrostatic devices. Until now, there have
alus: A Prospective Randomized Study With 122 Patients. Neurosurg
Q 2005; 15: 103-109
been no signs reported that the methodic use of hydro-
[7]
Kiefer M, Unterberg A. The differential diagnosis and treatment of
static valves has any disadvantage.
normal-pressure hydrocephalus. Dtsch Arztebl Int 2012; 109: 15-25,
Against this backdrop, the adjustability of hydrostatic
quiz 26
components promises further progress in terms of
[8]
Bech-Azeddine R, Waldemar G, Knudsen GM et al. Idiopathic normal-
increased numbers of treatment options; this is especially
pressure hydrocephalus: evaluation and findings in a multidiscipli-
nary memory clinic. Eur J Neurol 2001; 8: 601-611
true for problematic cases. It seems obvious that the
[9]
Tisell M, Höglund M, Wikkelsø C. National and regional incidence of
adjustment of the hydrostatic component in a growing
surgery for adult hydrocephalus in Sweden. Acta Neurol Scand 2005;
child introduces a meaningful procedure to the treatment.
112: 72-75
Regardless of the scientific competition of the different
[10]
Wilson RK, Williams MA. The role of the neurologist in the longitudi-
hydrostatic approaches, it is worth considering whether
nal management of normal pressure hydrocephalus. Neurologist
2010; 16: 238-248
and how adjustment options are or could be offered.
[11]
Conn HO. Normal pressure hydrocephalus: a case report by a physi-
cian who is the patient. Clin Med 2007; 7: 296-299
[12]
Conn HO, Lobo FM. What do physicians know about normal pressure
References
hydrocephalus and when did they know it? A survey of 284 physi-
cians. Yale J Biol Med 2008; 81: 19-29
[1] Hakim S, Adams RD. The special clinical problem of symptomatic
[13]
Tarnaris A, Watkins LD, Kitchen ND. Biomarkers in chronic adult
hydrocephalus with normal cerebrospinal fluid pressure. Observa-
hydrocephalus. Cerebrospinal Fluid Res 2006; 3: 11
tions on cerebrospinal fluid hydrodynamics. J Neurol Sci 1965; 2:
[14]
Lee WJ, Wang SJ, Hsu LC, Lirng JF, Wu CH, Fuh JL. Brain MRI as a pre-
307-327
dictor of CSF tap test response in patients with idiopathic normal
[2] McGirt MJ, Woodworth G, Coon AL, Thomas G, Williams MA, Riga-
pressure hydrocephalus. J Neurol 2010; 257: 1675-1681
monti D. Diagnosis, treatment, and analysis of long-term outcomes
[15]
Murakami M, Hirata Y, Kuratsu JI. Predictive assessment of shunt
in idiopathic normal-pressure hydrocephalus. Neurosurgery 2005;
effectiveness in patients with idiopathic normal pressure hydro-
57: 699-705, discussion 699-705
cephalus by determining regional cerebral blood flow on
3D
[3] Panagiotopoulos V, Konstantinou D, Kalogeropoulos A, Maraziotis T.
stereotactic surface projections. Acta Neurochir
(Wien)
2007;
The predictive value of external continuous lumbar drainage, with
149: 991-997
148
Index
CHECK-MATE adjustment
drainage resistance, predictive
gravity-assisted valve (GAV) 74,
A
device 79, 79
value 142
74-75
abdominal cavity pressure 130
chronic obstructive hydrocephalus,
draping 94, 95
- See also gravitational valves;
abducens nerve 106
see obstructive hydrocephalus
DUALSWITCH valve (DSV) 63, 71,
proGAV valve
abscess
clinical examination 28
71, 74, 90
grooved pegboard test 30, 32
- abdominal 123
clinical grading scales, see scales
- correct placement 72, 72
- cerebral 127
CODMAN MEDOS valve 66, 67, 90
- incorrect placement 71, 72
adjustable valves 39, 64
H
- pressure adjustment princi-
- See also specific valves
ple 64
Hakim triad 2
- benefits of adjustability 64, 86,
comorbidity
E
- postoperative improvement 142
88
- idiopathic NPH 18
economic considerations 88-89
Hakim, Salomón 12
- differential pressure (DP)
- index (CMI) 112, 112
education
head circumference 18
valves 64, 64, 65, 74
- predictive value 142
- medical personnel 146
headache 17, 111
- gravitational valves 74, 90
- quantification 112
- public 146
- overdrainage and 128
- hydrostatic devices 77
complications, see infections; over-
Eide, PK. 6
hematoma 62, 120
- imaging modality use in adjust-
drainage; underdrainage; shunt
endoscopic third ventriculostomy
- endoscopic third ventriculos-
ment 39, 40
failure
(ETV) 19, 102
tomy complications 107
- reasons for adjustment 64
- endoscopic third ventriculos-
- See also third ventricle
- gravitational valve evaluation 91
- versus nonadjustable valves 64
tomy 106
- complications 106
- shunt revision 135
Ahlskog, JE. 7
- intraoperative 120
- in nonobstructive hydrocepha-
Heyer, Ted 11
Alzheimer disease 20
-- rates of 121
lus 102
Hippocrates 10
- coincidence 112
- postoperative 128, 148
- in shunt failure 103
Hiraoka, K. 6
antibiotic treatment, see infections
computed tomography (CT) 36
- outcomes 107
historical background 10
antibiotic-impregnated shunts 89,
Conn, Harold O. 146-147
- technique 103-104
- early history 10
126
-- borehole site 103, 104
- shunt concepts 11
antisiphon device (ASD) 67, 68-69
-- patient positioning 104
- treatment in the 19th and 20th
- adjustable 77
D
-- preoperative planning 103
Centuries 10
aqueductal CSF flow 38, 38, 46
da Vinci, Leonardo 10
epidemiologic studies 6, 8
Holter, John D. 12
atmospheric pressure 58
Dandy, Walter Edward 11
Evans index 36
hydraulic press theory 24-25
atrophy, brain 20
dementia 17
external lumbar drainage 52, 52
hydrocephalus, see normal
- Alzheimer disease 20
pressure hydrocephalus (NPH);
- pathophysiology 17
obstructive hydrocephalus
B
F
- postoperative improvement 142
- history 10
BACTISEAL 126
- predictive value 142
-- early history 10
flow control 69, 70
ball-in-cone valve 63, 63
diagnosis, see imaging
-- shunt concepts 11
flow-reducing devices 70, 77
basilar artery 105, 105
- biomarkers 147
-- treatment in the 19th and 20th
- adjustable 77, 77
Binswanger disease 20
- diagnostic pathway 53, 53
Centuries 10
follow-up management 116
biochemical tests 52
- differential diagnosis 18, 19
hydrostatic devices 67, 147
- follow-up intervals 116
biofilm development 122, 122
- invasive work-up 46
- See also specific devices
-- one/three months examina-
- clinical implications 122
-- biochemical tests 52
- adjustable 77
tion 116
biomarkers 147
-- CSF tap test 51, 52
- opening pressure impact 90
-- six months examination 116
Black Grading Scale for shunt
-- external lumbar drainage 52,
hydrostatic pressure 58, 59, 130
-- twelve months examina-
assessment 110, 110, 112
52
- changes within a tube 59
tion 116
Blackfan, Kenneth Daniel 11
-- intracranial pressure monitor-
- organizing follow-up examina-
brain atrophy 20
ing 46
tions 116
Brean, A. 6-7
I
-- lumbar infusion test 46
- scheme 117
bulk flow theory 24
- noninvasive work-up 28
foramen of Monro 103-104, 104
idiopathic NPH (iNPH) 2
-- clinical examination 28
- risk of damage 107
- comorbidities 18
-- medical history 28
frontal ventricular catheter 87, 87
- diagnostic pathway 53
C
- secondary NPH 18
- See also ventricular catheter
- differential diagnosis 18, 19
catheter, see peritoneal catheter;
- shunt responsiveness predic-
- natural history 16
ventricular catheter
tion 147
- pathophysiology 24
cerebrospinal fluid (CSF)
differential pressure (DP) valves 61
G
-- bulk flow theory 24
- aqueductal flow 38, 38, 46
- See also specific valves
- symptoms 16, 16
gait disturbance 16
- dynamics 48
- adjustable 64, 64, 65, 74
- treatment 19
- evaluation 28
- early studies 10
- overdrainage problem 89
- underdiagnosis 2, 146
Galen of Pergamon 10
- infection 123
- physics of 59, 60-61
gravitational valves 41, 71, 90
imaging 36
-- diagnosis 125
diffusion tensor imaging (DTI) 38
- See also specific imaging
- See also specific valves
- outflow resistance 47
digit span test 30
techniques
- adjustable 74, 90
-- calculation procedure 48, 49
disconnection 133, 133
- direct postoperative imaging 39
- analogous type 73, 73
- production rate 69, 70
disease awareness issues
- routine follow-up 39
- correct placement 72, 72
- reservoir 88, 96, 96-97, 98
- medical personnel 146
- efficiency 91
- troubleshooting applications 41
- tap test 51, 52, 128
- public 146
-- overdrainage 41
- incorrect placement 41, 71, 72,
-- predictive value 142
disinfection 94
-- underdrainage 42
134
cervical myelopathy 21
dizziness 18, 111
- valve setting 39, 40
–- repositioning 133
149
Index
incontinence 17
- multifrequency magnetic
- flow-reduction device 69
Rey auditory verbal learning test
- evaluation 29
resonance elastography 39, 39
- imaging 41
(RAVLT) 30
- postoperative improvement 142
Marmarou, A. 6
- mechanism 130, 130
index for postoperative improve-
medical history 28
- obese patients 61
S
ment 110
MEDOS CODMAN valve 66, 67, 90
- prevalence 90-91
infections 121
- pressure adjustment
- prevention 64, 86, 88
scales 110
- avoidance 125
principle 64
- shunt revision 135
- Black Grading Scale for shunt
- diagnosis 124
membrane valve 62, 63
- valve adjustment 64, 65
assessment 110, 110, 112
-- clinical examination 125
meningitis 123
- Kiefer grading scale 111, 111
-- paraclinical examination 125
- treatment 127
- Stein-Langfitt Scale 111, 111
-- timing of infection 124
Mikulicz-Radecki, Jan 11
P
Schulte, Rudi 11
- epidemiology 123, 124
Mini Mental State Examination
Pacchioni, Antonio 10
secondary NPH (sNPH) 2, 18
- organisms 123
(MMSE) 29, 30, 31
Parkinson disease 19
- diagnosis 18, 19
- shunt colonization 122, 126
miniNAV valve 63, 63, 81
- coincidence 112
- symptoms 18
-- biofilm development 122, 122
Minnesota study, USA 7
- prevalence 6
- treatment 19
- shunt-related CSF infection/
Mixter, William Jason 11
patient positioning 94
sexual dysfunction 18
meningitis 123, 127
MONOSTEP valve 63
- endoscopic third ventriculos-
shaving 94
- shunt-related peritonitis/
multifrequency magnetic
tomy 104
shunt 2, 19, 58
abdominal abscess 123, 127
resonance elastography 39, 39
Payr, Erwin 11
- See also ventriculoatrial (VA)
- treatment 126
peritoneal catheter
shunt; ventriculoperitoneal (VP)
-- shunt revision 135
- dislocation 133, 134-135
shunt; lumboperitoneal (LP)
infratentorial intracisternal ob-
N
- obstruction 137
shunt; valves
structive hydrocephalus (Infin-
neuropsychological testing 29
- placement 98, 98, 99-100
- assessment 110, 110, 112
OH) 102, 103
normal pressure hydrocephalus
peritonitis 123
- CSF reservoir 88, 96, 96-97, 98
infundibular recess 105, 105
(NPH) 2
- treatment 127
- definition 58
intracranial bleeding 120
- See also idiopathic NPH (iNPH);
phase-contrast MRI 38
- disconnection 133, 133
- See also hematoma
secondary NPH (sNPH)
plexectomy 25
- failure, see shunt failure; shunt
intracranial pressure (ICP) 58, 130
- discovery of 12
Polaris valve 66, 66
revisions
- calculation with VP shunt 88, 88
Norwegian studies 6-7
posterior cerebral artery 105, 105
- future technological improve-
- frequency distribution curve 58
NPH, see normal pressure
posterior communicating
ments 147
- monitoring 46
hydrocephalus (NPH)
artery 105, 105
- historical background 11
intraoperative complications 120
NPH recovery rate 111, 112
postoperative complications 128,
- infection, see infections
- rates of 121
Nulsen, Frank E.
12
148
- modern perspectives 13
intrathecal infusion test 142
- See also infections; overdrainage;
- optimal settings 86, 89
Iseki, C.
7
underdrainage; shunt failure
- placement, see surgical
O
postoperative improvement
technique
index 110
- responsiveness prediction 147
J
obese patients 61
prediction
- selection of 86, 86
obstructive hydrocephalus 18
Japanese studies 6-7
- diagnosis 19, 50
- postoperative outcome 142
- types of 58
- shunt responsiveness 147
shunt failure 41, 132
-- differential diagnosis 19
proGAV valve 75, 75-76, 80, 90, 90
- See also overdrainage; under-
- infratentorial intracisternal
K
- pressure adjustment
drainage; shunt revisions
(InfinOH) 102, 103
Key, Axel Hendrick 10
- symptoms 18
principle 64
- diagnostics of shunt malfunc-
Kiefer grading scale 111, 111
- treatment 19, 102
- setting verification 76, 76
tion 88, 131
- surgical placement 97-98
-- inspection 131
Klassen, BT. 7
occipital ventricular catheter 87,
prognosis 142
-- palpation 131, 131
Kocher, Emil Theodor 10
87
- postoperative improve-
-- pumping 132
Kocher’s point 95
- See also ventricular catheter
oculomotor nerve 105, 105
ment 142, 143
-- radiography 132
Ommaya, Ayub Khan 12
- predictors 142
-- shuntogram 132
L
programmable valves, see adjust-
- disconnection 133, 133
operating room 94
able valves
shunt revisions 131-132
- patient positioning 94, 104
Le Cat, Claude-Nicolas 10
proSA valve 77, 78-80, 90
- catheter dislocation/improper
Orbis Sigma valve (OSV) 69, 69, 70,
lumbar infusion test 46, 50-51
70
- adjustment verification 79
placement 132, 133
- outflow resistance 47, 47
- CHECK-MATE adjustment
-- peritoneal catheter 133, 134-
-- calculation procedure 48, 49
outcome
device 79, 79
135
lumbar puncture 46
- endoscopic third ventriculos-
- opening pressure-angle relation-
- disconnection 133, 133
tomy 107
lumbar spinal canal stenosis 21
ships 78-79, 79-80
- endoscopic third ventriculos-
- postoperative improve-
lumboperitoneal (LP) shunt 99
ment 142, 143
psychiatric symptoms 17
tomy role 103
- See also shunt
public awareness 146
- kinked catheter 133, 134
- infection 123
-- index 110
Pudenz, Robert H. 11
- oblique positioned gravitational
- predictors 142
valve 133, 134
outflow resistance 47, 47
M
- bolus injection measurement
- obstructed catheter/valve 134,
Q
method 47
135, 136-137
Magendie, Francois 10
- overdrainage 135
- calculation procedure 48, 49
Quincke, Heinrich Irenaeus 11
magnetic resonance imaging
- shunt infection 135
overdrainage 41, 62, 90-91, 128,
(MRI) 38
- underdrainage without obvious
129, 148
- diffusion tensor imaging
R
cause 135
- antisiphon device 69
(DTI) 38
-- algorithm 136
- definition 90
Retzius, Magnus Gustav 10
150
Index
- when to revise 131
- valve placement 96, 97-98
- evaluation 29
- material 89
SHUNTASSISTANT 73, 74, 90
- ventricular catheter 95, 95,
- postoperative improvement 142
- obstruction 120, 128, 135
- gravity-compensating accessory
96-97
-- shunt revision 134, 136
(GCA) 73
Swedish study 6
- occipital approach 87, 87
V
shuntogram 42, 42, 132
- surgical technique 95, 95, 96-97
silicone-slit valve 62, 62
ventricular infusion test 50
valves 58
SILVERLINE 126
T
ventricular size and shape assess-
- See also shunt; specific valves
SIPHONGUARD 70, 70, 71
ment 36
Tanaka, N. 7
- adjustment 97
skin ulceration 135, 136
tap test 51, 52
-- imaging modality use 39, 40
- indices 36, 37
sleep requirement 18
- predictive value 142
-- overdrainage 64, 65
- third ventricle diameter 37
Sophysa valves
ventriculoatrial (VA) shunt 61, 86,
third ventricle, see endoscopic third
-- underdrainage 64, 65
- Polaris 66, 66
100, 100
ventriculostomy (ETV)
- classification 61, 61
- pressure adjustment princi-
- See also shunt
- diameter 37
- differential pressure (DP)
ples 64
- floor 105
valves 61
- incorrect positioning 120
- SU8 64, 65, 66
-- anomalies 103
-- adjustable 64, 64, 65, 74
- infection 123
Spitz, Eugene Bernard 12
- right atrial thrombosis risk 100
-- opaque 105, 105-106
-- overdrainage problem 89
Stein-Langfitt Scale 111, 111
- versus ventriculoperitoneal (VP)
-- perforation 106, 106, 107
-- physics of 59, 60-61
step length 29
shunt 86
-- translucent 105, 105
- gravitational 41, 41, 71, 90
STRATA valve 67, 67, 77
timed up-and-go test 29
-- adjustable 74, 90
ventriculoperitoneal (VP) shunt 58,
- pressure adjustment princi-
86
Tisell, M. 6
-- analogous type 73, 73
ple 64
- See also shunt
Torkildsen, Arne 11
-- correct placement 72, 72
Stroop test 32, 33
- alternatives to 99
trail-making test 32, 32
-- efficiency 91
subcortical vascular dementia 20
Trenkwalder, C. 6
-- incorrect placement 41, 71, 72,
- incorrect positioning 120
subdural effusion 62, 64, 129
tuber cinereum 103
134
- infection 121
subdural hygroma 71, 90, 129
- physics of 59, 60
turn tests 29
- hydrostatic devices 67
- gravitational valve evaluation 91
-- overdrainage mechanism 130,
-- adjustable 77
- overdrainage association 41, 129
130
- opening pressure selection 86,
- shunt revision 135
U
88, 129, 129
- versus ventriculoatrial (VA)
surgical technique 94-95
-- influence on course of dis-
shunt 86
underdrainage 42, 89-90, 128
- disinfection 94
vertigo 18
- antisiphon device 68
ease 90
- draping 94, 95
Vesalius, Andreas 10
- selection of 89
- definition 90
- endoscopic third ventriculos-
video recording, gait 29
- surgical placement 96, 97-98
- flow-reduction device 70
tomy (ETV) 103-104
ventricular catheter 87
- imaging 41
- intraoperative complica-
- obese patients 61
- dislocation 128
tions 120
W
- prevalence 90-91
-- management 132
-- rates of 121
- frontal approach 87, 87, 95
Wernicke, Carl 11
- prevention 64
- operating room 94
- incorrect positioning 120, 128,
Whytt, Robert 10
- shunt revision 135
- patient positioning 94, 104
133
Willis, Thomas 10
-- algorithm 136
- peritoneal catheter 98, 98, 99-
- valve adjustment 64, 65, 128
-- avoidance 121
Windkessel effect 25
100
urinary incontinence 17
-- management 132
wound infections 135
- shaving 94
– kinked 133, 134
wound-healing problems 135
151