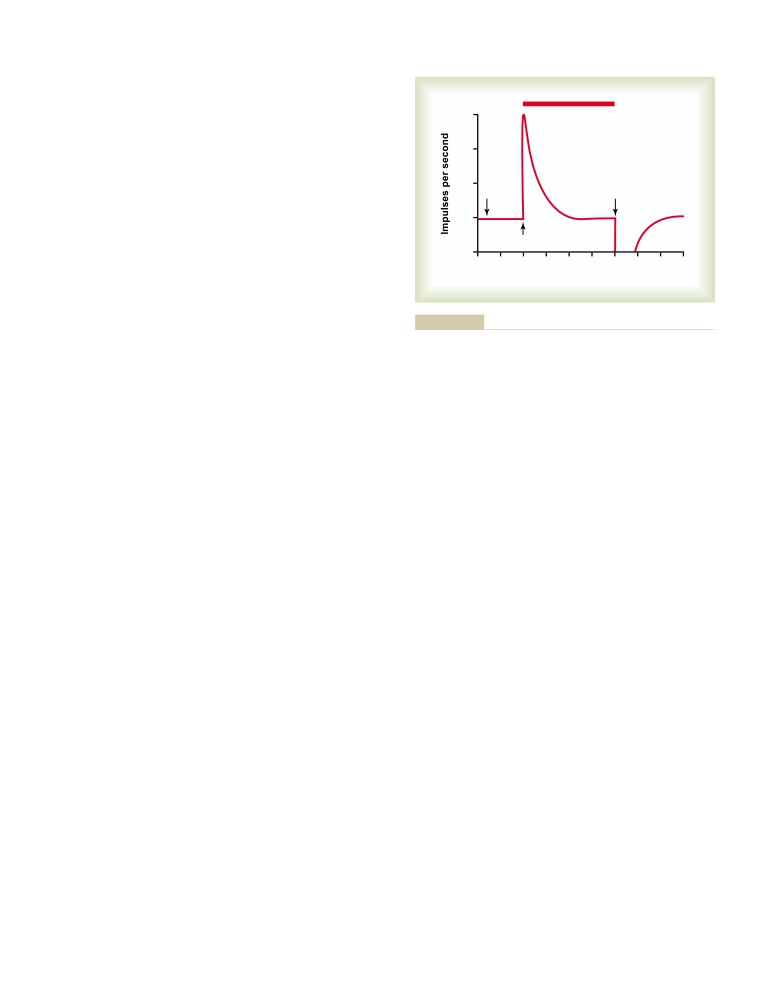
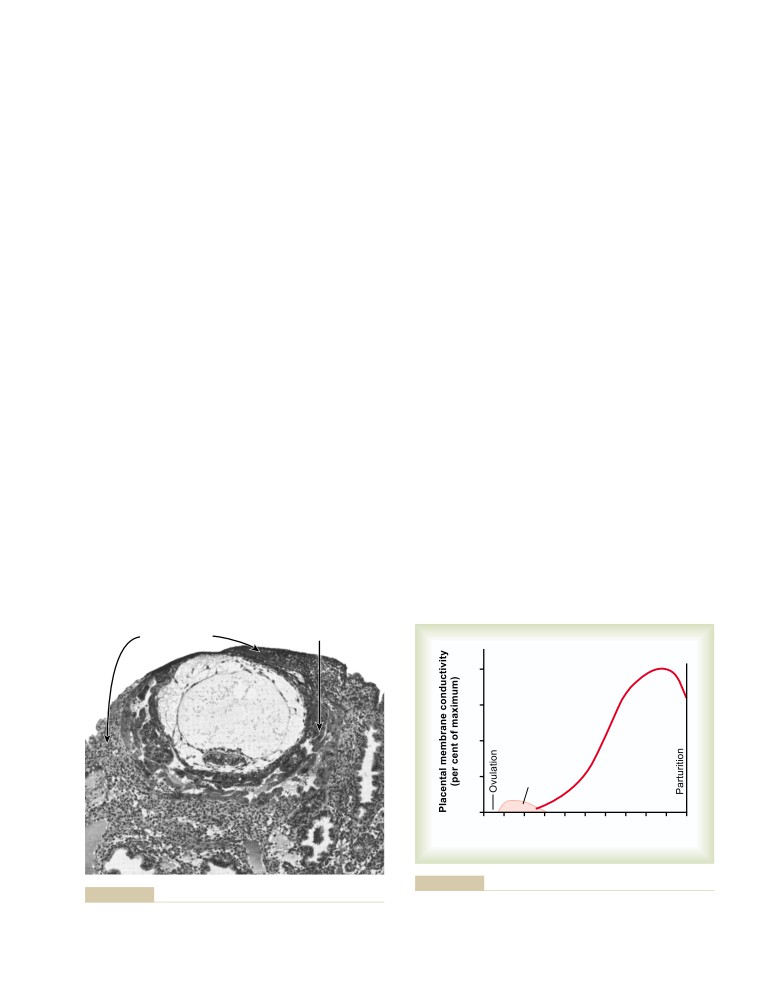
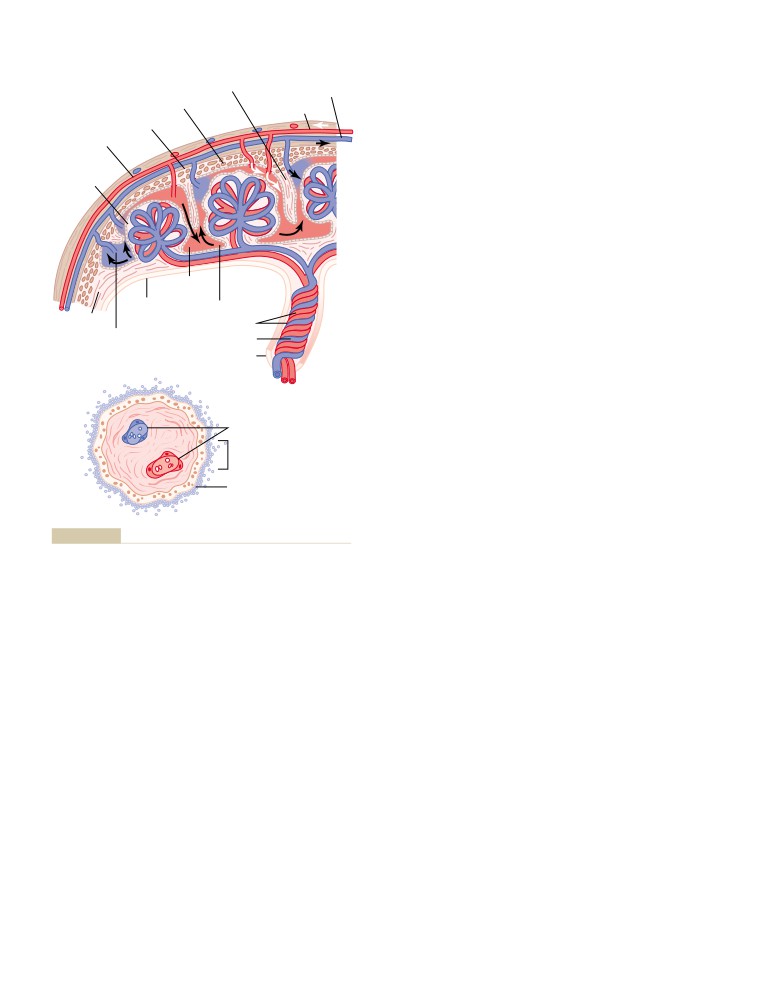
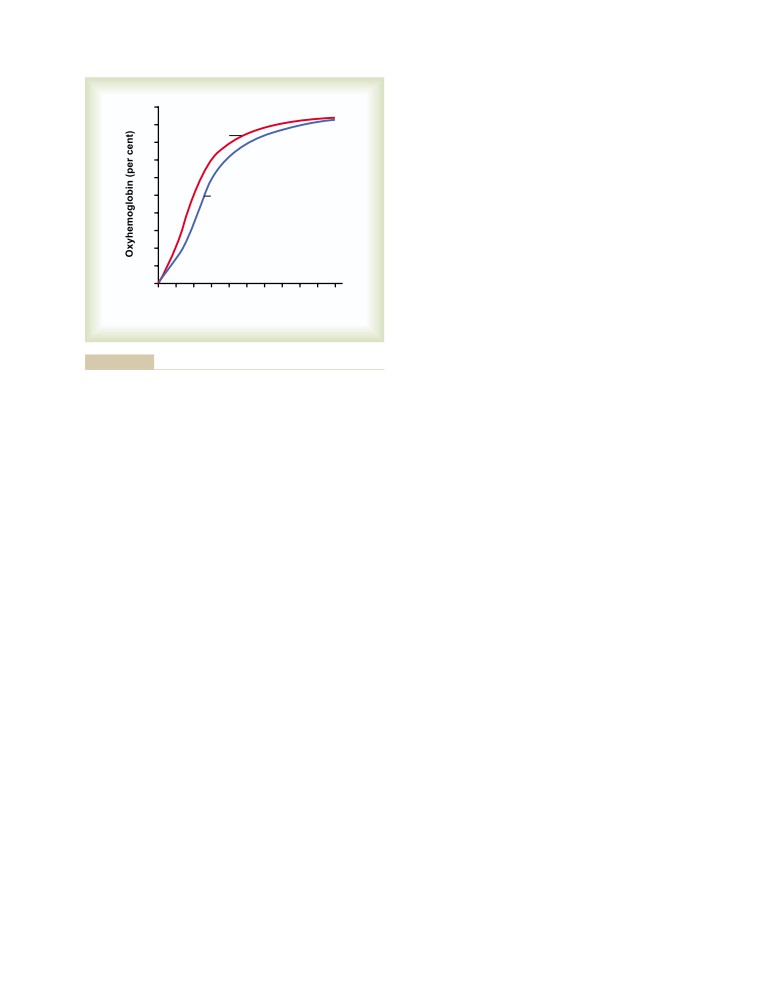
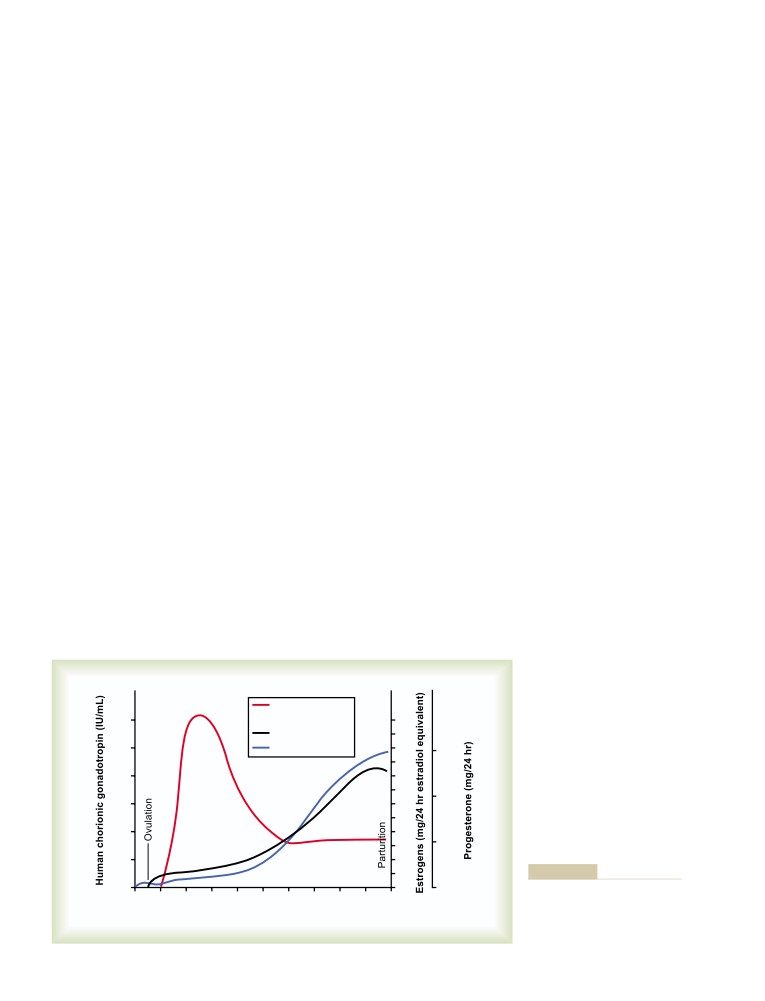
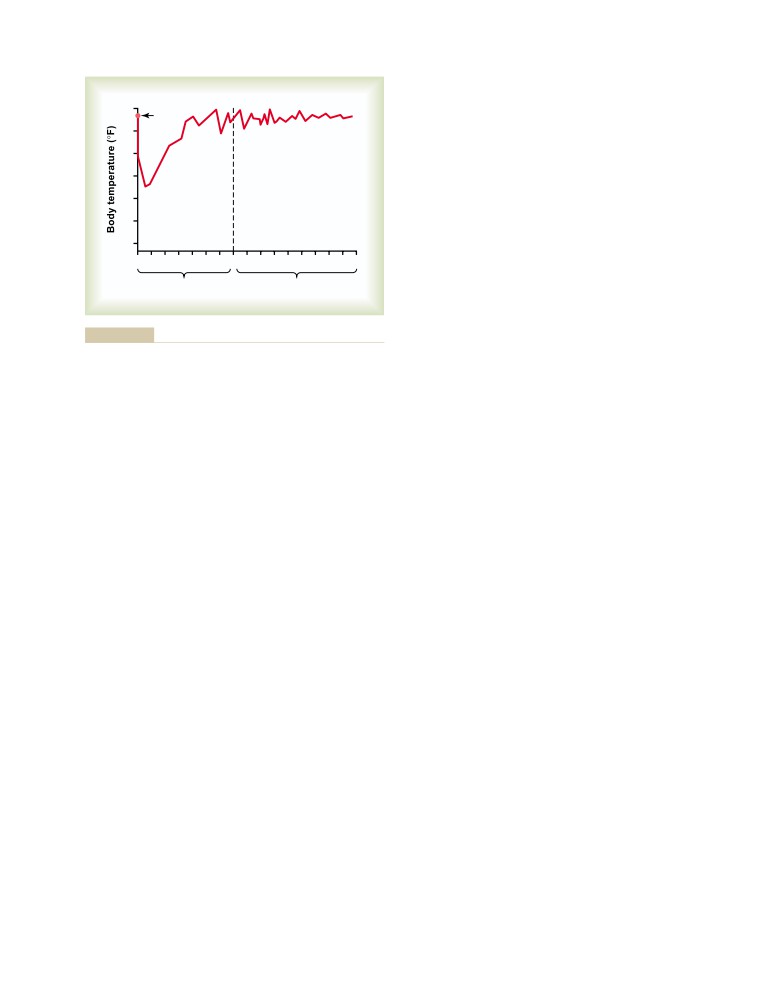
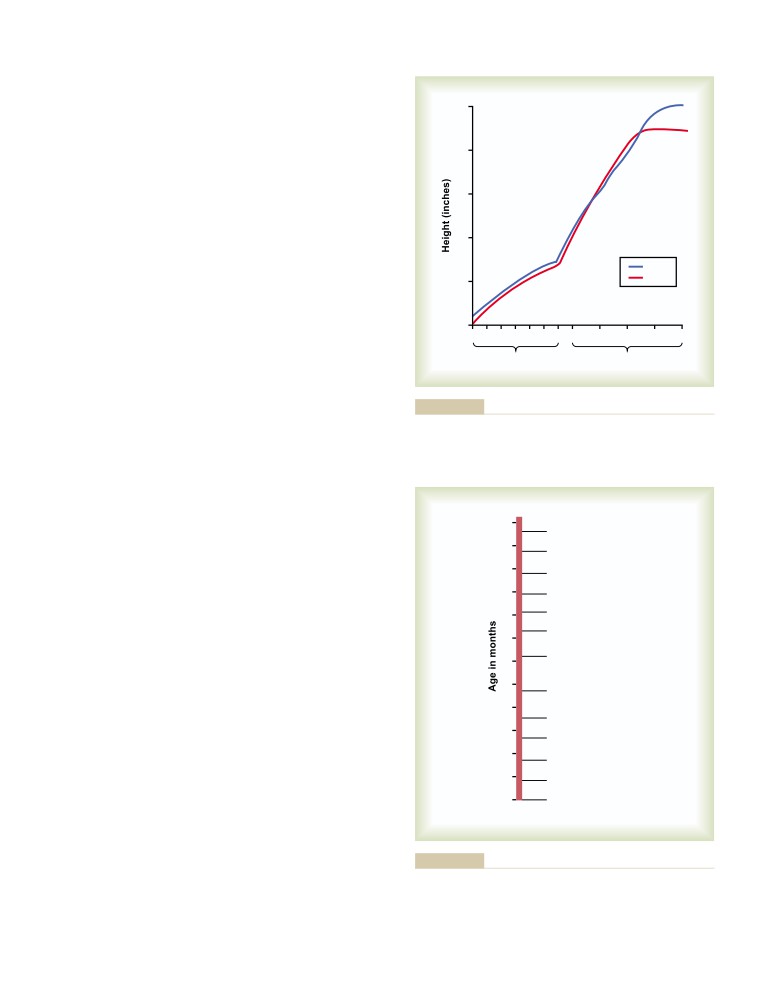
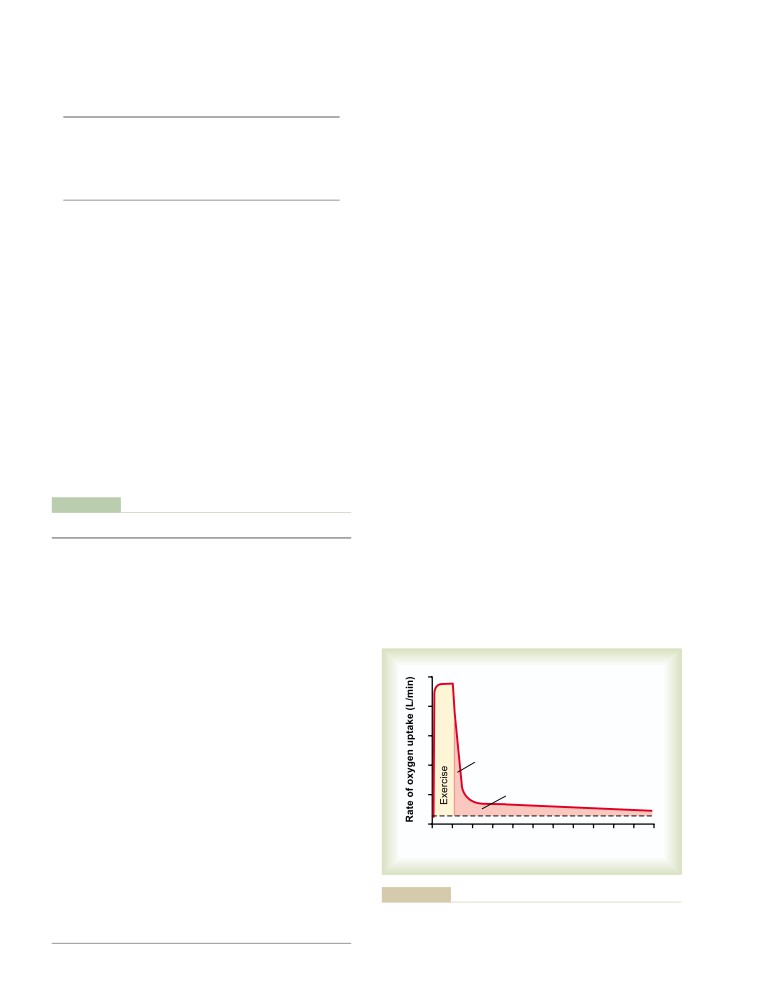
T E X T B O O K
of Medical
Physiology
T E X T B O O K
of Medical
Physiology
E L E V E N T H
E D I T I O N
Arthur C. Guyton, M.D.†
Professor Emeritus
Department of Physiology and Biophysics
University of Mississippi Medical Center
Jackson, Mississippi
†Deceased
John E. Hall, Ph.D.
Professor and Chairman
Department of Physiology and Biophysics
University of Mississippi Medical Center
Jackson, Mississippi
Elsevier Inc.
1600 John F. Kennedy Blvd., Suite 1800
Philadelphia, Pennsylvania 19103-2899
TEXTBOOK OF MEDICAL PHYSIOLOGY
ISBN 0-7216-0240-1
International Edition ISBN 0-8089-2317-X
Copyright © 2006, 2000, 1996, 1991, 1986, 1981, 1976, 1971, 1966, 1961, 1956 by Elsevier Inc.
All rights reserved. No part of this publication may be reproduced or transmitted in any form or by
any means, electronic or mechanical, including photocopying, recording, or any information storage
and retrieval system, without permission in writing from the publisher. Permissions may be sought
directly from Elsevier’s Health Sciences Rights Department in Philadelphia, PA, USA: phone: (+1)
215 239 3804, fax: (+1) 215 239 3805, e-mail: healthpermissions@elsevier.com. You may also complete
Support” and then “Obtaining Permissions”.
NOTICE
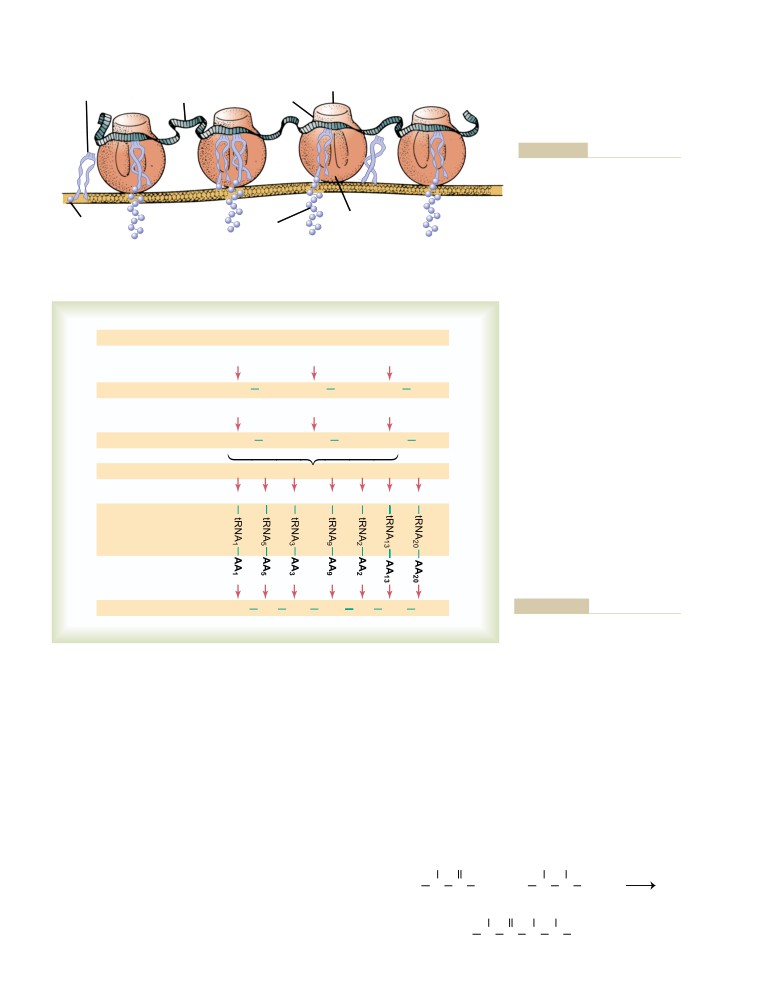
Knowledge and best practice in this field are constantly changing. As new research and experience
broaden our knowledge, changes in practice, treatment and drug therapy may become necessary
or appropriate. Readers are advised to check the most current information provided (i) on
procedures featured or (ii) by the manufacturer of each product to be administered, to verify the
recommended dose or formula, the method and duration of administration, and contraindications.
It is the responsibility of the practitioner, relying on their own experience and knowledge of the
patient, to make diagnoses, to determine dosages and the best treatment for each individual
patient, and to take all appropriate safety precautions. To the fullest extent of the law, neither the
Publisher nor the Author assumes any liability for any injury and/or damage to persons or
property arising out or related to any use of the material contained in this book.
Library of Congress Cataloging-in-Publication Data
Guyton, Arthur C.
Textbook of medical physiology / Arthur C. Guyton, John E. Hall.—11th ed.
p. ; cm.
Includes bibliographical references and index.
ISBN 0-7216-0240-1
1. Human physiology.
2. Physiology, Pathological. I. Title: Medical physiology. II. Hall,
John E. (John Edward) III. Title.
[DNLM:
1. Physiological Processes. QT 104 G992t 2006]
QP34.5.G9
2006
612—dc22
2004051421
Publishing Director: Linda Belfus
Acquisitions Editor: William Schmitt
Managing Editor: Rebecca Gruliow
Publishing Services Manager: Tina Rebane
Project Manager: Mary Anne Folcher
Design Manager: Steven Stave
Marketing Manager: John Gore
Cover illustration is a detail from Opus 1972 by Virgil Cantini, Ph.D., with permission of the artist and
Mansfield State College, Mansfield, Pennsylvania.
Chapter opener credits: Chapter 43, modified from © Getty Images 21000058038; Chapter 44, modified
from © Getty Images 21000044598; Chapter 84, modified from © Corbis.
Working together to grow
libraries in developing countries
Printed in China
Last digit is the print number:
9
8
7
6
5
4
3
2
1
To
My Family
For their abundant support, for their patience and
understanding, and for their love
To
Arthur C. Guyton
For his imaginative and innovative research
For his dedication to education
For showing us the excitement and joy of physiology
And for serving as an inspirational role model
Arthur C. Guyton, M.D.
1919-2003
I N M E M O R I A M
The sudden loss of Dr. Arthur C. Guyton in an automobile accident on April 3,
2003, stunned and saddened all who were privileged to know him. Arthur
Guyton was a giant in the fields of physiology and medicine, a leader among
leaders, a master teacher, and an inspiring role model throughout the world.
Arthur Clifton Guyton was born in Oxford, Mississippi, to Dr. Billy S.
Guyton, a highly respected eye, ear, nose, and throat specialist, who later
became Dean of the University of Mississippi Medical School, and Kate Small-
wood Guyton, a mathematics and physics teacher who had been a missionary
in China before marriage. During his formative years, Arthur enjoyed watching
his father work at the Guyton Clinic, playing chess and swapping stories with
William Faulkner, and building sailboats
(one of which he later sold to
Faulkner). He also built countless mechanical and electrical devices, which he
continued to do throughout his life. His brilliance shone early as he graduated
top in his class at the University of Mississippi. He later distinguished himself
at Harvard Medical School and began his postgraduate surgical training at
Massachusetts General Hospital.
His medical training was interrupted twice—once to serve in the Navy during
World War II and again in 1946 when he was stricken with poliomyelitis during
his final year of residency training. Suffering paralysis in his right leg, left arm,
and both shoulders, he spent nine months in Warm Springs, Georgia, recuper-
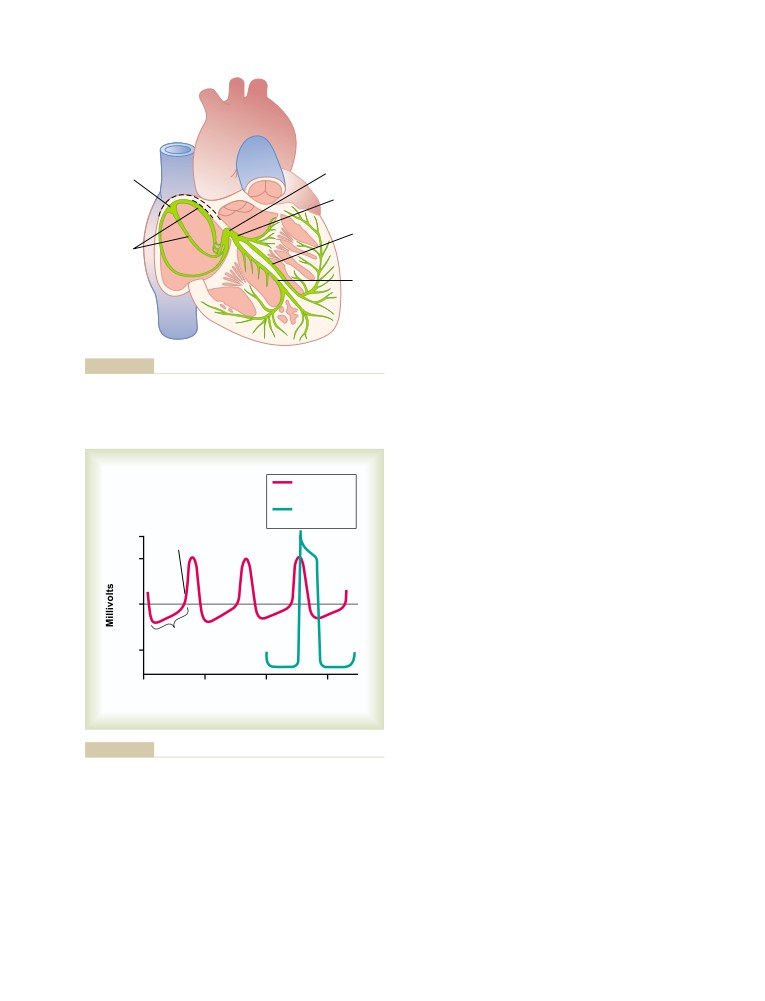
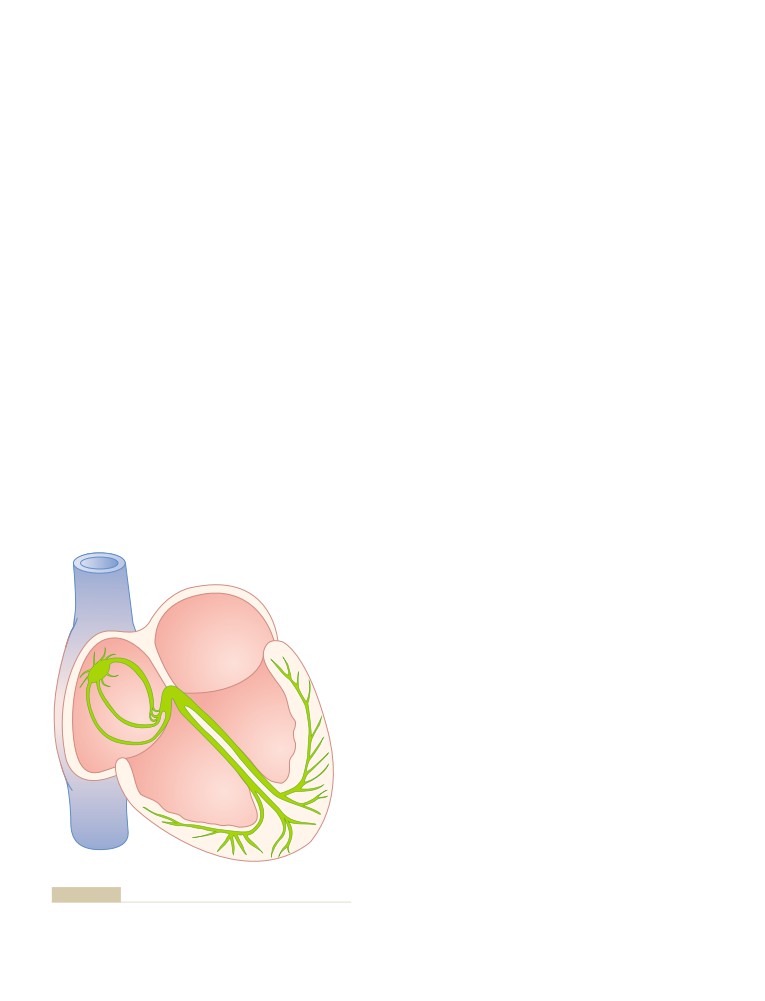
ating and applying his inventive mind to building the first motorized wheelchair
controlled by a “joy stick,” a motorized hoist for lifting patients, special leg
braces, and other devices to aid the handicapped. For those inventions he
received a Presidential Citation.
He returned to Oxford where he devoted himself to teaching and research
at the University of Mississippi School of Medicine and was named Chair of the
Department of Physiology in 1948. In 1951 he was named one of the ten out-
standing men in the nation. When the University of Mississippi moved its
Medical School to Jackson in 1955, he rapidly developed one of the world’s
premier cardiovascular research programs. His remarkable life as a scientist,
author, and devoted father is detailed in a biography published on the occasion
of his “retirement” in 1989.1
A Great Physiologist. Arthur Guyton’s research contributions, which include
more than 600 papers and 40 books, are legendary and place him among the
greatest physiologists in history. His research covered virtually all areas of car-
diovascular regulation and led to many seminal concepts that are now an inte-
gral part of our understanding of cardiovascular disorders, such as hypertension,
heart failure, and edema. It is difficult to discuss cardiovascular physiology
without including his concepts of cardiac output and venous return, negative
interstitial fluid pressure and regulation of tissue fluid volume and edema,
regulation of tissue blood flow and whole body blood flow autoregulation,
renal-pressure natriuresis, and long-term blood pressure regulation. Indeed, his
concepts of cardiovascular regulation are found in virtually every major text-
book of physiology. They have become so familiar that their origin is sometimes
forgotten.
One of Dr. Guyton’s most important scientific legacies was his application of
principles of engineering and systems analysis to cardiovascular regulation. He
used mathematical and graphical methods to quantify various aspects of circu-
latory function before computers were widely available. He built analog com-
puters and pioneered the application of large-scale systems analysis to modeling
the cardiovascular system before the advent of digital computers. As digital
computers became available, his cardiovascular models expanded dramatically
to include the kidneys and body fluids, hormones, and the autonomic nervous
system, as well as cardiac and circulatory functions.2 He also provided the first
comprehensive systems analysis of blood pressure regulation. This unique
approach to physiological research preceded the emergence of biomedical
vii
viii
In Memoriam
engineering—a field that he helped to establish and to
and 20/20 described the remarkable home environ-
promote in physiology, leading the discipline into a
ment that Arthur and Ruth Guyton created to raise
quantitative rather than a descriptive science.
their family. His devotion to family is beautifully
It is a tribute to Arthur Guyton’s genius that his
expressed in the dedication of his Textbook of Medical
concepts of cardiovascular regulation often seemed
Physiology5:
heretical when they were first presented, yet stimu-
To
lated investigators throughout the world to test them
My father for his uncompromising principles that
experimentally. They are now widely accepted. In fact,
guided my life
many of his concepts of cardiovascular regulation
My mother for leading her children into intellectual
are integral components of what is now taught in
pursuits
most medical physiology courses. They continue to
My wife for her magnificent devotion to her family
be the foundation for generations of cardiovascular
My children for making everything worthwhile
physiologists.
Dr. Guyton received more than 80 major honors
Dr. Guyton was a master teacher at the University
from diverse scientific and civic organizations and uni-
of Mississippi for over 50 years. Even though he was
versities throughout the world. A few of these that are
always busy with service responsibilities, research,
especially relevant to cardiovascular research include
writing, and teaching, he was never too busy to talk
the Wiggers Award of the American Physiological
with a student who was having difficulty. He would
Society, the Ciba Award from the Council for High
never accept an invitation to give a prestigious lecture
Blood Pressure Research, The William Harvey Award
if it conflicted with his teaching schedule.
from the American Society of Hypertension, the
His contributions to education are also far reach-
Research Achievement Award of the American Heart
ing through generations of physiology graduate
Association, and the Merck Sharp & Dohme Award
students and postdoctoral fellows. He trained over
of the International Society of Hypertension. It was
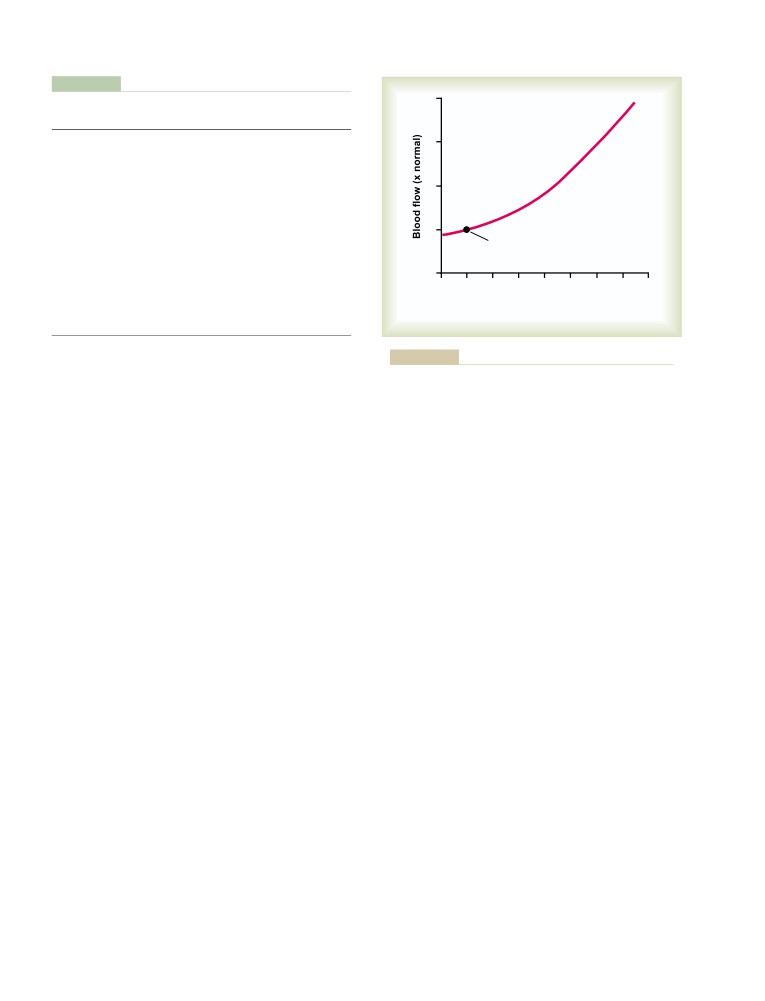
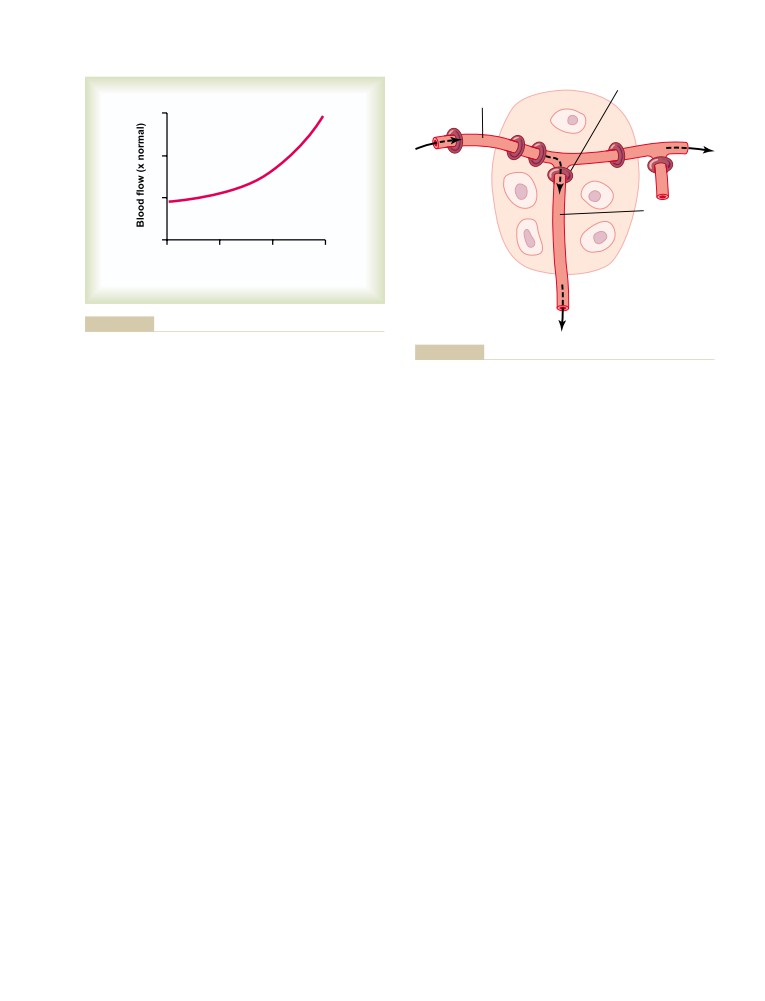
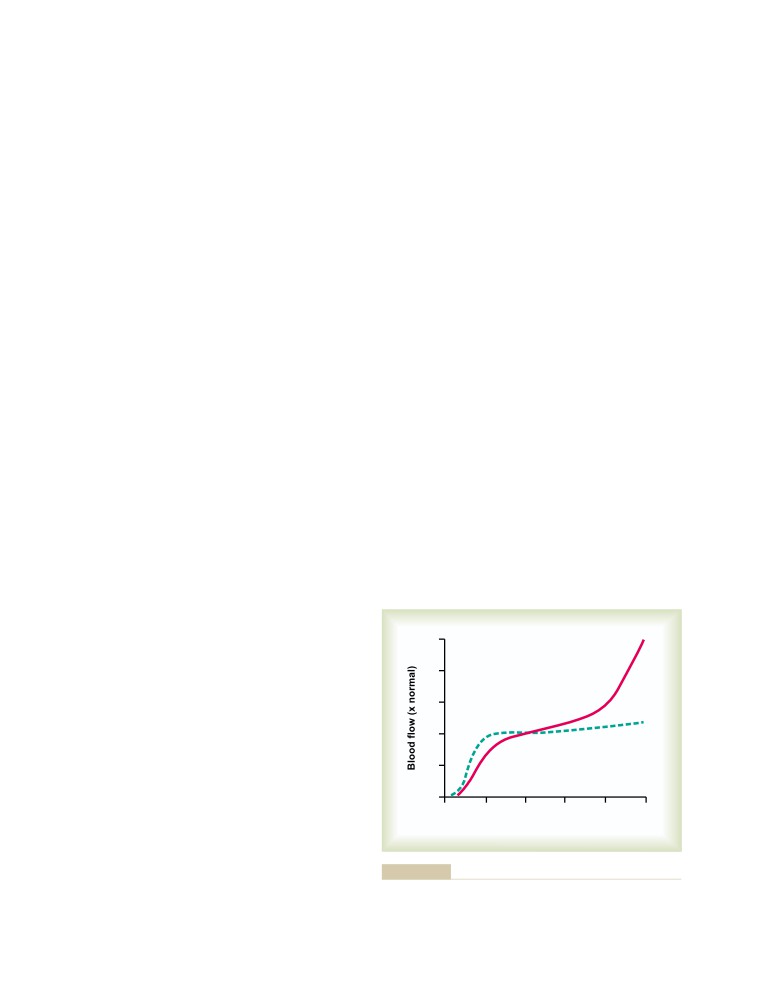
150 scientists, at least 29 of whom became chairs of
appropriate that in 1978 he was invited by the Royal
their own departments and six of whom became pres-
College of Physicians in London to deliver a special
idents of the American Physiological Society. He gave
lecture honoring the 400th anniversary of the birth of
students confidence in their abilities and emphasized
William Harvey, who discovered the circulation of the
his belief that “People who are really successful in the
blood.
research world are self-taught.” He insisted that his
Dr. Guyton’s love of physiology was beautifully
trainees integrate their experimental findings into a
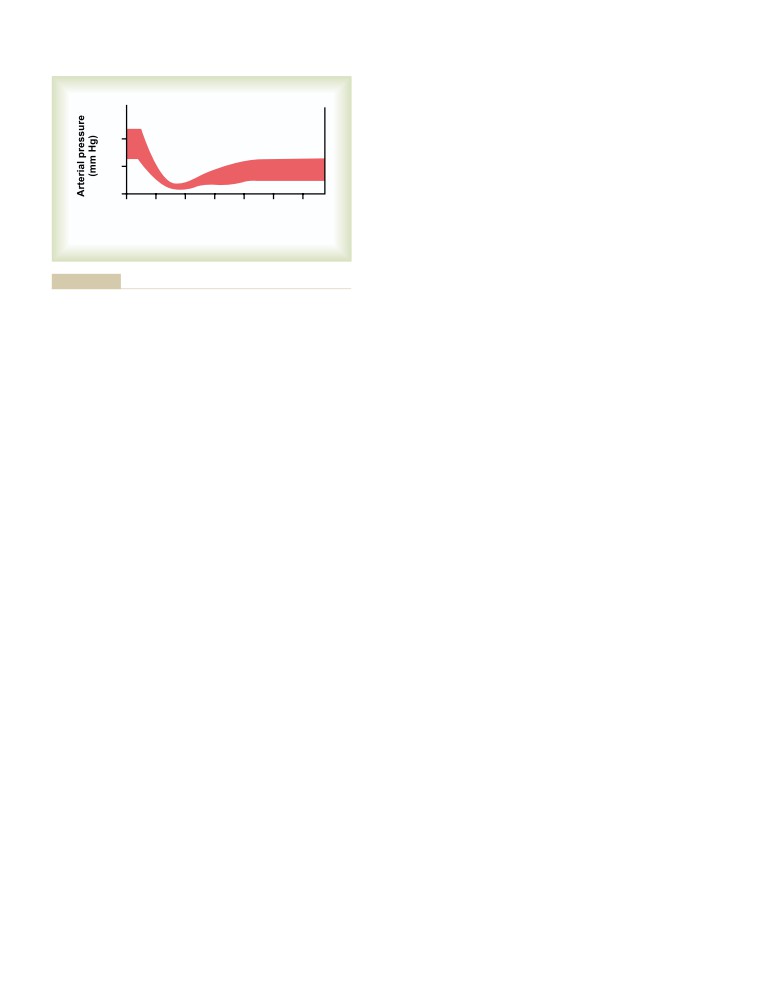
articulated in his president’s address to the American
broad conceptual framework that included other
Physiological Society in 1975,3 appropriately entitled
interacting systems. This approach usually led them
Physiology, a Beauty and a Philosophy. Let me quote
to develop a quantitative analysis and a better
just one sentence from his address: What other person,
understanding of the particular physiological systems
whether he be a theologian, a jurist, a doctor of medi-
that they were studying. No one has been more pro-
cine, a physicist, or whatever, knows more than you, a
lific in training leaders of physiology than Arthur
physiologist, about life? For physiology is indeed an
Guyton.
explanation of life. What other subject matter is more
Dr. Guyton’s Textbook of Medical Physiology, first
fascinating, more exciting, more beautiful than the
published in 1956, quickly became the best-selling
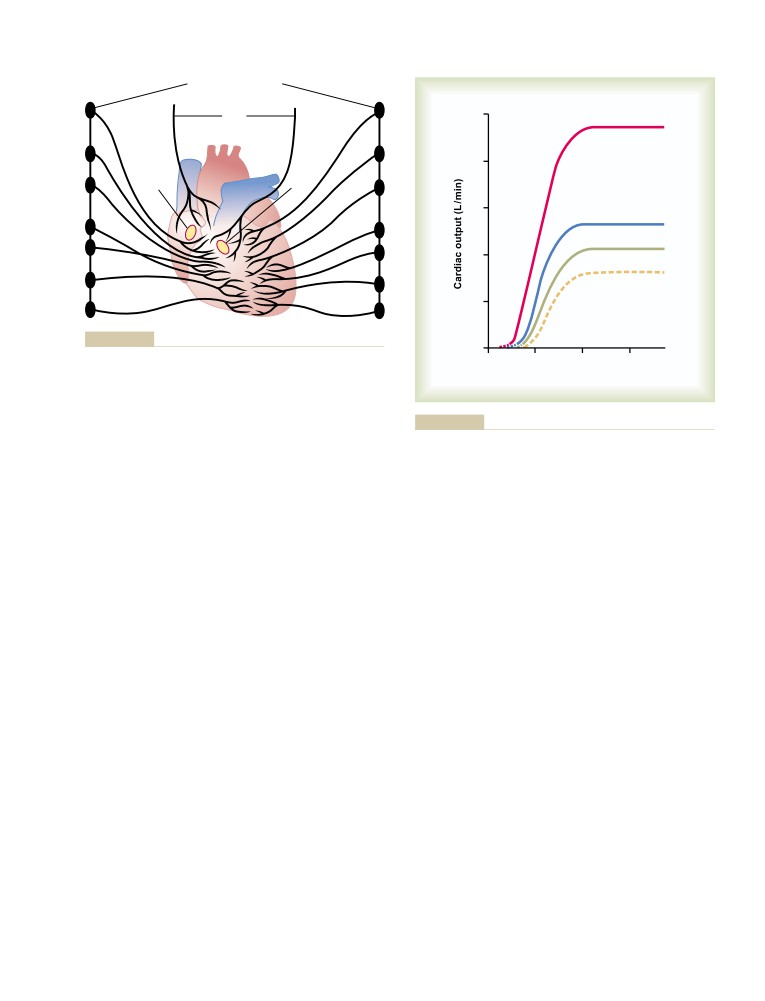
subject of life?
medical physiology textbook in the world. He had a
gift for communicating complex ideas in a clear and
A Master Teacher. Although Dr. Guyton’s research
interesting manner that made studying physiology fun.
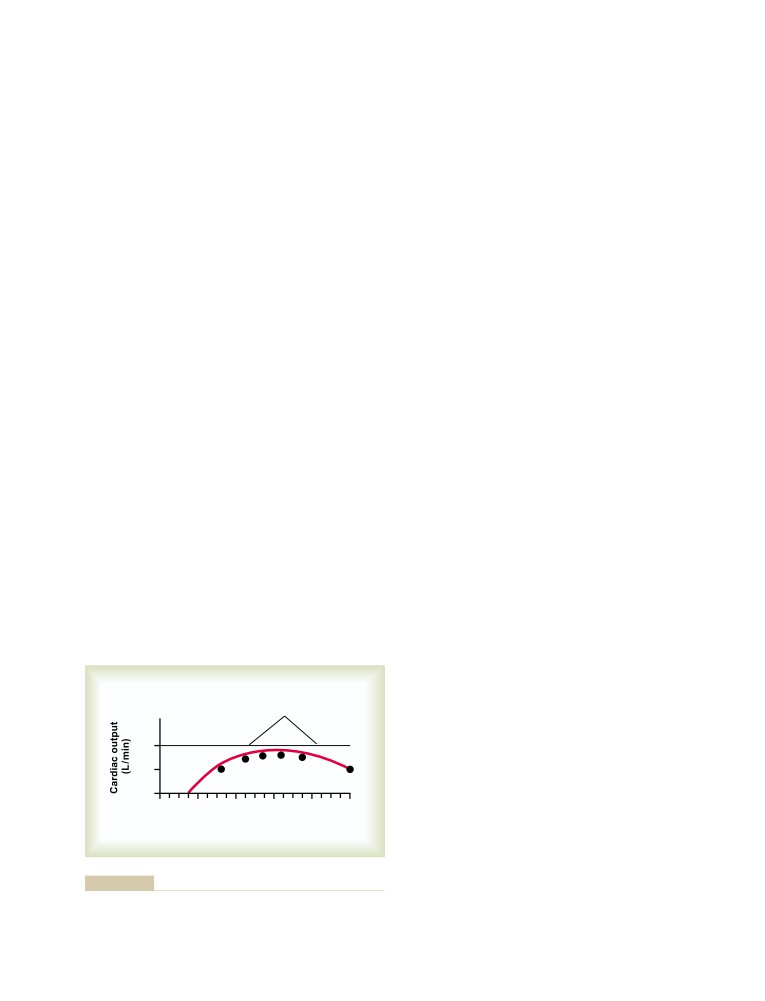
accomplishments are legendary, his contributions as an
He wrote the book to teach his students, not to impress
educator have probably had an even greater impact.
his professional colleagues. Its popularity with stu-
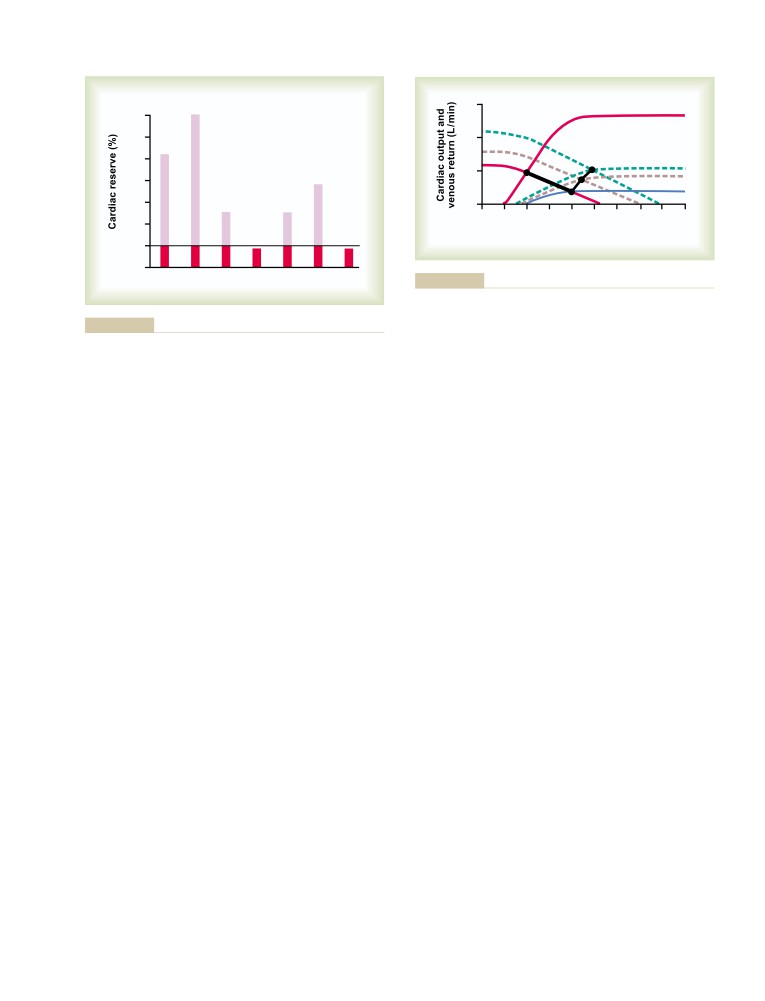
He and his wonderful wife Ruth raised ten children,
dents has made it the most widely used physiology
all of whom became outstanding physicians—a
textbook in history. This accomplishment alone was
remarkable educational achievement. Eight of the
enough to ensure his legacy.
Guyton children graduated from Harvard Medical
The Textbook of Medical Physiology began as
School, one from Duke Medical School, and one from
lecture notes in the early 1950s when Dr. Guyton was
The University of Miami Medical School after receiv-
teaching the entire physiology course for medical stu-
ing a Ph.D. from Harvard. An article published in
dents at the University of Mississippi. He discovered
Reader’s Digest in 1982 highlighted their extraordinary
that the students were having difficulty with the text-
family life.4
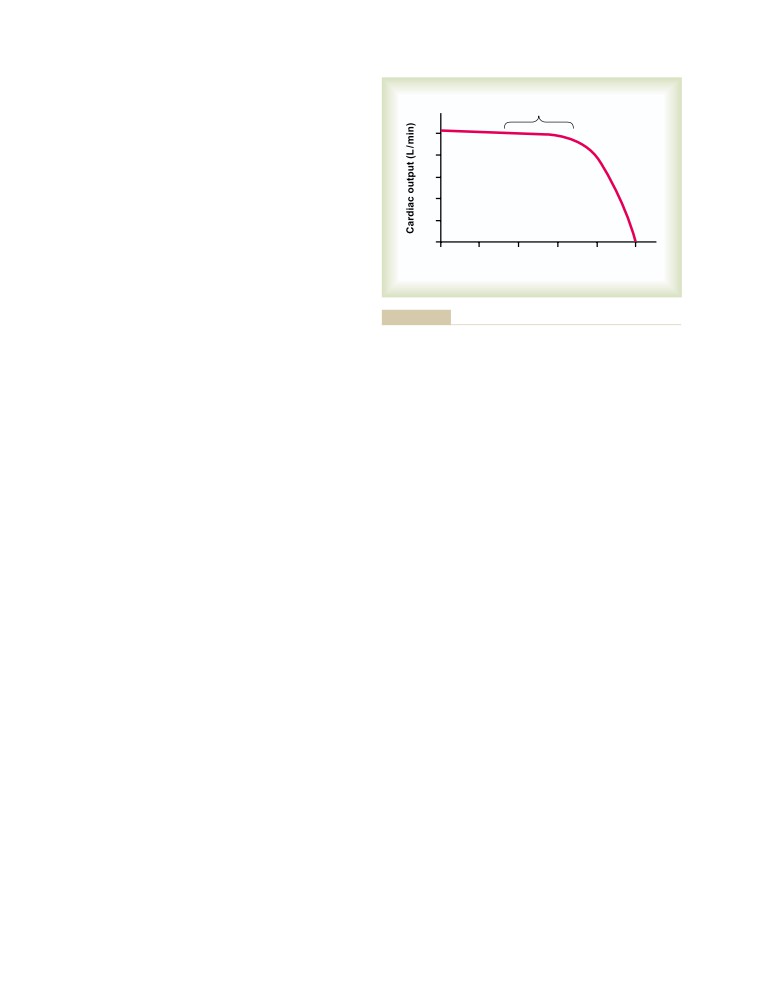
books that were available and began distributing
The success of the Guyton children did not occur by
copies of his lecture notes. In describing his experi-
chance. Dr. Guyton’s philosophy of education was to
ence, Dr. Guyton stated that “Many textbooks of
“learn by doing.” The children participated in count-
medical physiology had become discursive, written pri-
less family projects that included the design and
marily by teachers of physiology for other teachers of
construction of their home and its heating system,
physiology, and written in language understood by
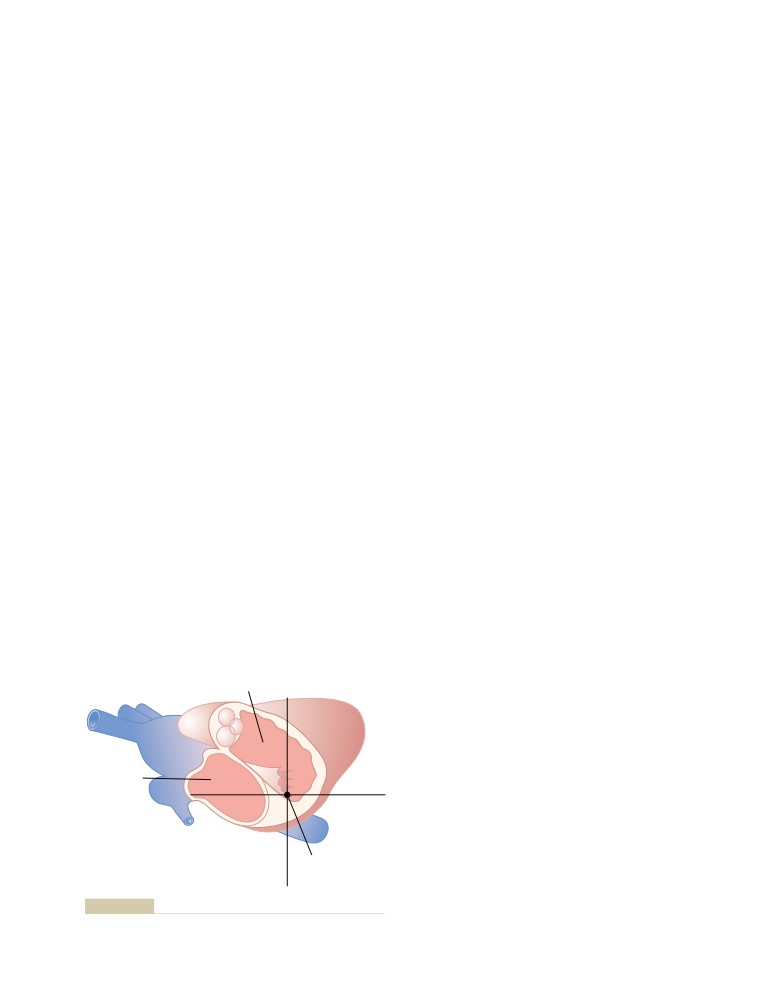
the swimming pool, tennis court, sailboats, go-carts
other teachers but not easily understood by the basic
and electrical cars, household gadgets, and electronic
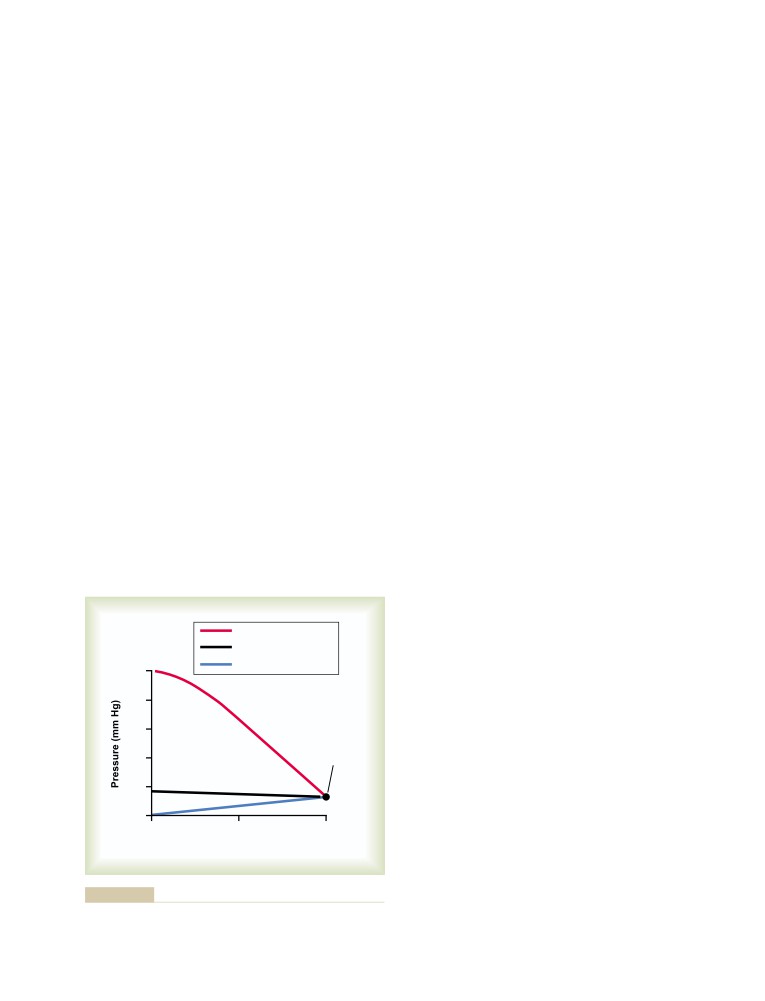
student of medical physiology.”6
instruments for their Oxford Instruments Company.
Through his Textbook of Medical Physiology, which
Television programs such as Good Morning America
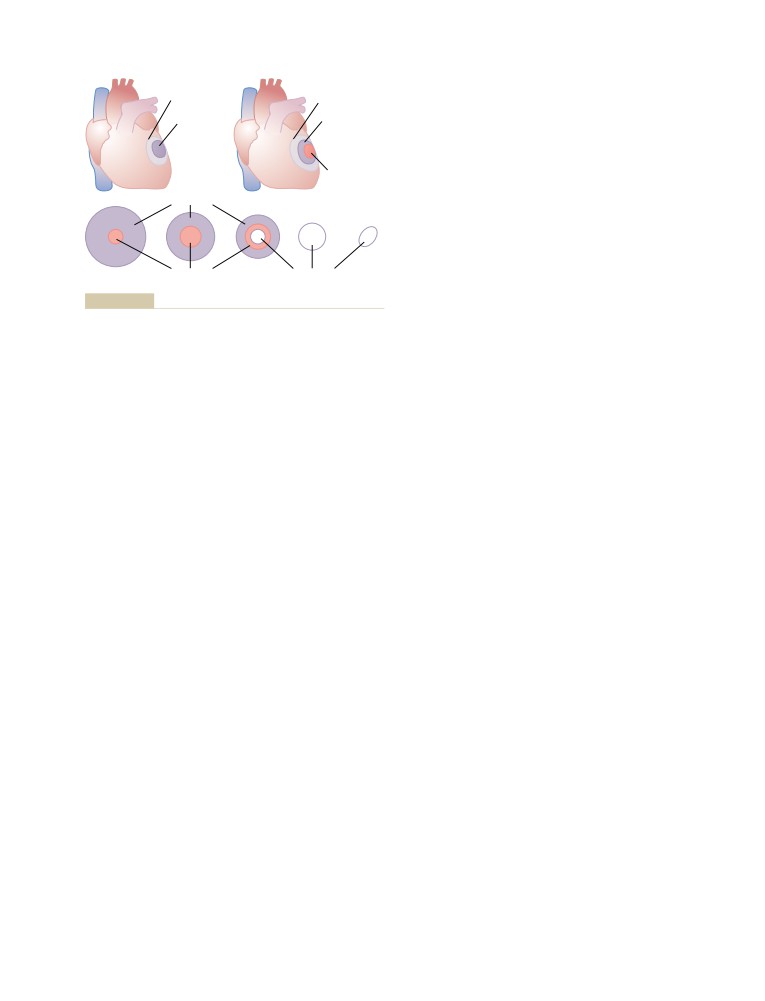
is translated into 13 languages, he has probably done
In Memoriam
ix
more to teach physiology to the world than any other
We celebrate the magnificent life of Arthur Guyton,
individual in history. Unlike most major textbooks,
recognizing that we owe him an enormous debt. He
which often have 20 or more authors, the first eight
gave us an imaginative and innovative approach to
editions were written entirely by Dr. Guyton—a feat
research and many new scientific concepts. He gave
that is unprecedented for any major medical textbook.
countless students throughout the world a means of
For his many contributions to medical education, Dr.
understanding physiology and he gave many of us
Guyton received the 1996 Abraham Flexner Award
exciting research careers. Most of all, he inspired us—
from the Association of American Medical Colleges
with his devotion to education, his unique ability to
(AAMC). According to the AAMC, Arthur Guyton
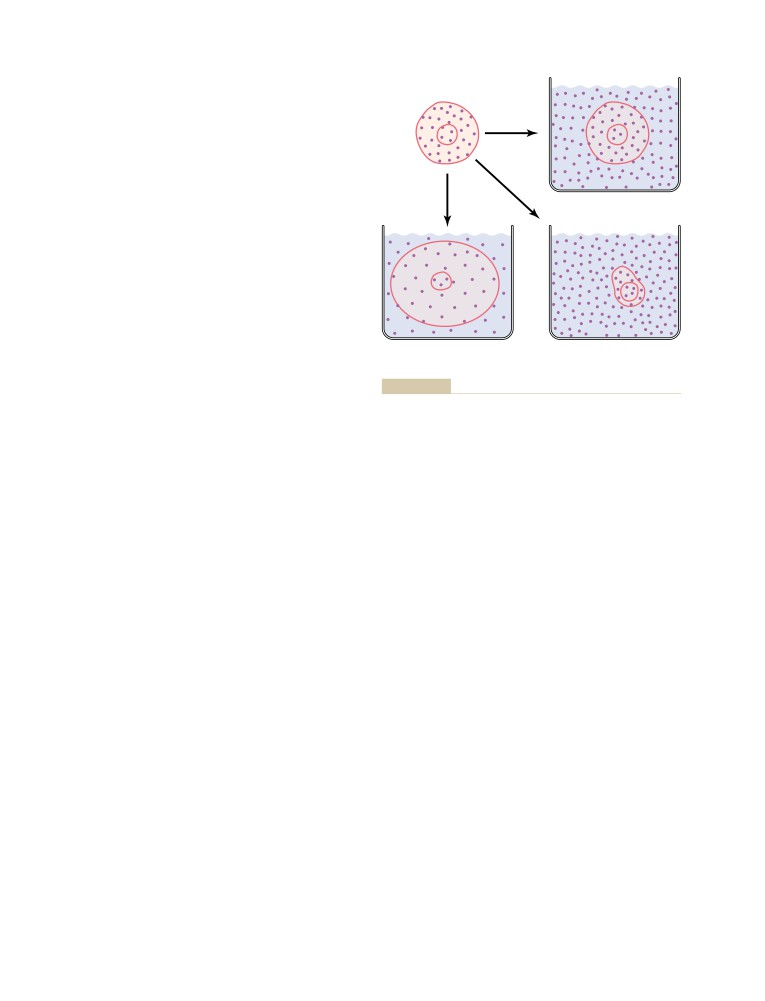
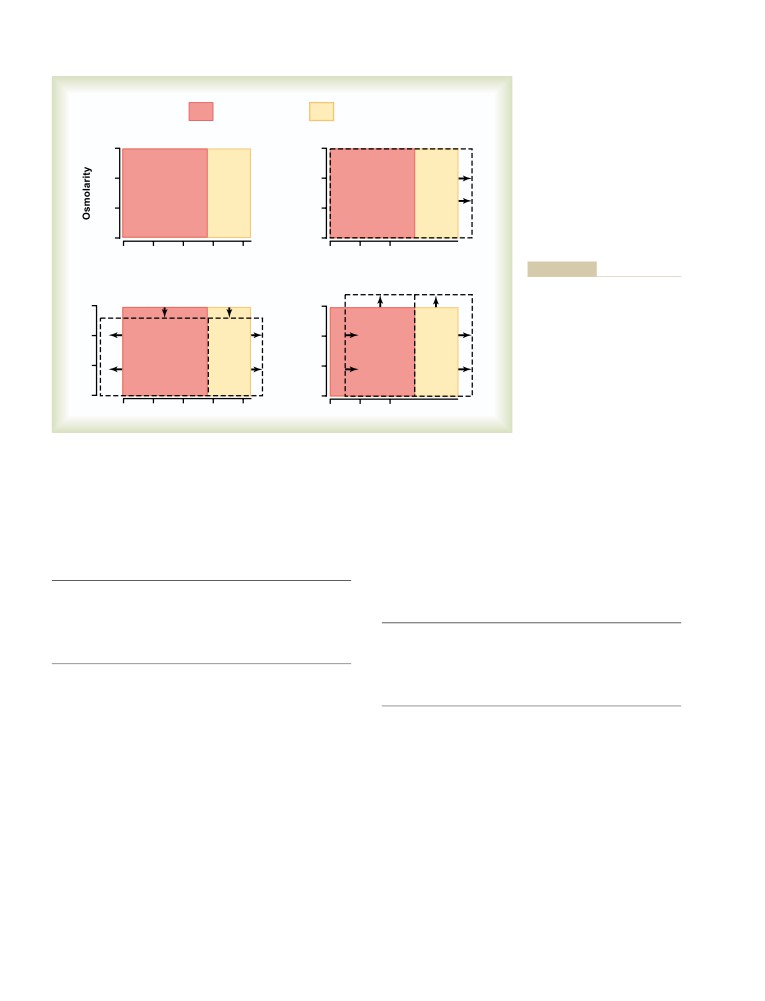
bring out the best in those around him, his warm and
“. . . for the past 50 years has made an unparalleled
generous spirit, and his courage. We will miss him
impact on medical education.” He is also honored each
tremendously, but he will remain in our memories as
year by The American Physiological Society through
a shining example of the very best in humanity. Arthur
the Arthur C. Guyton Teaching Award.
Guyton was a real hero to the world, and his legacy is
everlasting.
An Inspiring Role Model. Dr. Guyton’s accomplish-
ments extended far beyond science, medicine, and edu-
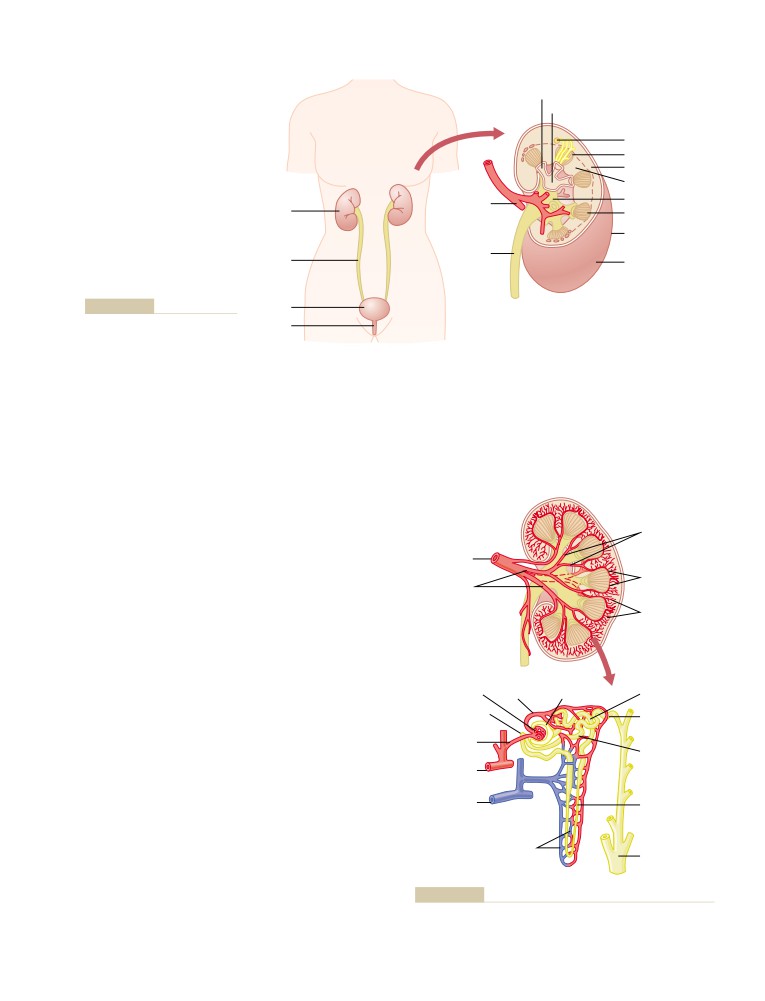
References
cation. He was an inspiring role model for life as well
as for science. No one was more inspirational or influ-
1. Brinson C, Quinn J: Arthur C. Guyton—His Life, His
ential on my scientific career than Dr. Guyton. He
Family, His Achievements. Jackson, MS, Hederman
taught his students much more than physiology—
Brothers Press, 1989.
he taught us life, not so much by what he said but by
2. Guyton AC, Coleman TG, Granger HJ: Circulation:
his unspoken courage and dedication to the highest
overall regulation. Ann Rev Physiol 34:13-46, 1972.
3. Guyton AC: Past-President’s Address. Physiology, a
standards.
Beauty and a Philosophy. The Physiologist 8:495-501,
He had a special ability to motivate people through
1975.
his indomitable spirit. Although he was severely chal-
4. Bode R: A Doctor Who’s Dad to Seven Doctors—So Far!
lenged by polio, those of us who worked with him
Readers’ Digest, December, 1982, pp. 141-145.
never thought of him as being handicapped. We were
5. Guyton AC: Textbook of Medical Physiology. Philadel-
too busy trying to keep up with him! His brilliant
phia, Saunders, 1956.
mind, his indefatigable devotion to science, education,
6. Guyton AC: An author’s philosophy of physiology text-
and family, and his spirit captivated students and
book writing. Adv Physiol Ed 19: s1-s5, 1998.
trainees, professional colleagues, politicians, business
leaders, and virtually everyone who knew him. He
John E. Hall
would not succumb to the effects of polio. His courage
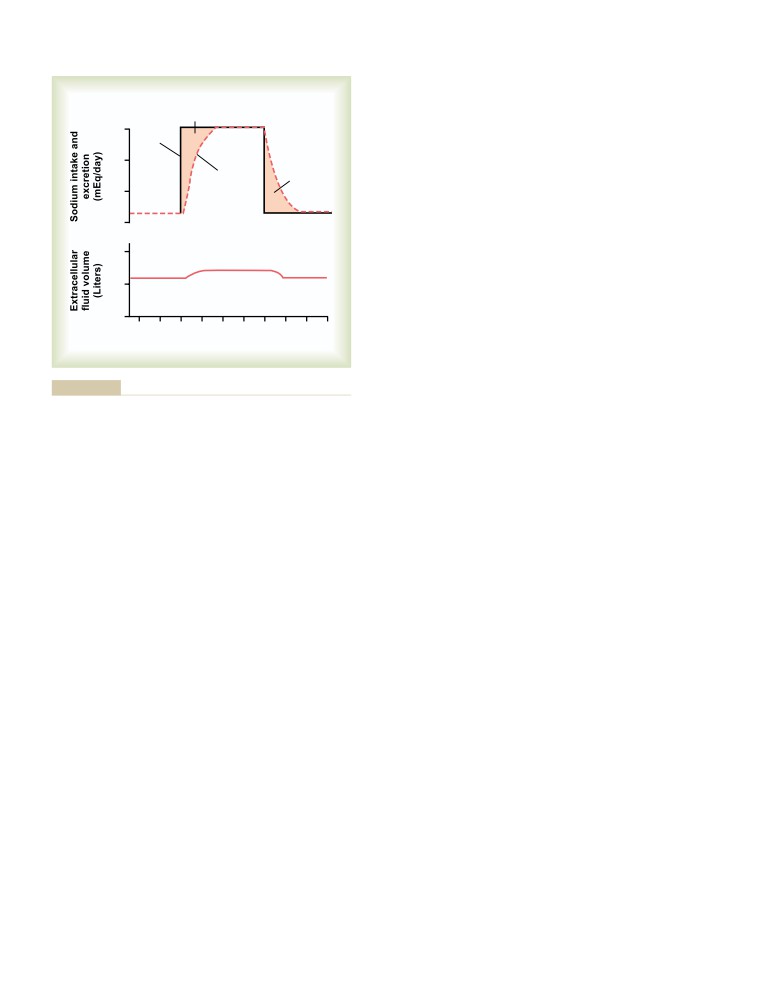
Jackson, Mississippi
challenged and inspired us. He expected the best and
somehow brought out the very best in people.
P R E F A C E
The first edition of the Textbook of Medical Phys-
iology was written by Arthur C. Guyton almost 50
years ago. Unlike many major medical textbooks,
which often have 20 or more authors, the first
eight editions of the Textbook of Medical Physi-
ology were written entirely by Dr. Guyton with
each new edition arriving on schedule for nearly
40 years. Over the years, Dr. Guyton’s textbook
became widely used throughout the world and was translated into 13 languages.
A major reason for the book’s unprecedented success was his uncanny ability
to explain complex physiologic principles in language easily understood by stu-
dents. His main goal with each edition was to instruct students in physiology,
not to impress his professional colleagues. His writing style always maintained
the tone of a teacher talking to his students.
I had the privilege of working closely with Dr. Guyton for almost 30 years
and the honor of helping him with the 9th and 10th editions. For the 11th
edition, I have the same goal as in previous editions—to explain, in language
easily understood by students, how the different cells, tissues, and organs of the
human body work together to maintain life. This task has been challenging and
exciting because our rapidly increasing knowledge of physiology continues to
unravel new mysteries of body functions. Many new techniques for learning
about molecular and cellular physiology have been developed. We can present
more and more the physiology principles in the terminology of molecular and
physical sciences rather than in merely a series of separate and unexplained bio-
logical phenomena. This change is welcomed, but it also makes revision of each
chapter a necessity.
In this edition, I have attempted to maintain the same unified organization
of the text that has been useful to students in the past and to ensure that
the book is comprehensive enough that students will wish to use it in later life
as a basis for their professional careers. I hope that this textbook conveys
the majesty of the human body and its many functions and that it stimulates
students to study physiology throughout their careers. Physiology is the link
between the basic sciences and medicine. The great beauty of physiology is
that it integrates the individual functions of all the body’s different cells, tissues,
and organs into a functional whole, the human body. Indeed, the human
body is much more than the sum of its parts, and life relies upon this total func-
tion, not just on the function of individual body parts in isolation from the
others.
This brings us to an important question: How are the separate organs and
systems coordinated to maintain proper function of the entire body? Fortu-
nately, our bodies are endowed with a vast network of feedback controls that
achieve the necessary balances without which we would not be able to live.
Physiologists call this high level of internal bodily control homeostasis. In
disease states, functional balances are often seriously disturbed and homeosta-
sis is impaired. And, when even a single disturbance reaches a limit, the whole
body can no longer live. One of the goals of this text, therefore, is to emphasize
the effectiveness and beauty of the body’s homeostasis mechanisms as well as
to present their abnormal function in disease.
Another objective is to be as accurate as possible. Suggestions and critiques
from many physiologists, students, and clinicians throughout the world have
been sought and then used to check factual accuracy as well as balance in the
text. Even so, because of the likelihood of error in sorting through many thou-
sands of bits of information, I wish to issue still a further request to all readers
to send along notations of error or inaccuracy. Physiologists understand the
importance of feedback for proper function of the human body; so, too, is feed-
back important for progressive improvement of a textbook of physiology. To
the many persons who have already helped, I send sincere thanks.
xi
xii
Preface
A brief explanation is needed about several features
wish to study particular physiologic mechanisms more
of the 11th edition. Although many of the chapters
deeply.
have been revised to include new principles of physi-
The material in large print constitutes the funda-
ology, the text length has been closely monitored
mental physiologic information that students will
to limit the book size so that it can be used effec-
require in virtually all their medical activities and
tively in physiology courses for medical students and
studies.
health care professionals. Many of the figures have
I wish to express my thanks to many other persons
also been redrawn and are now in full color. New
who have helped in preparing this book, including
references have been chosen primarily for their pres-
my colleagues in the Department of Physiology &
entation of physiologic principles, for the quality of
Biophysics at the University of Mississippi Medical
their own references, and for their easy accessibility.
Center who provided valuable suggestions. I am also
Most of the selected references are from recently
grateful to Ivadelle Osberg Heidke, Gerry McAlpin,
published scientific journals that can be freely
and Stephanie Lucas for their excellent secretarial
accessed from the PubMed internet site at http://
services, and to William Schmitt, Rebecca Gruliow,
Mary Anne Folcher, and the rest of the staff of
Use of these references, as well as cross-references
Elsevier Saunders for continued editorial and produc-
from them, can give the student almost complete cov-
tion excellence.
erage of the entire field of physiology.
Finally, I owe an enormous debt to Arthur Guyton
Another feature is that the print is set in two sizes.
for an exciting career in physiology, for his friendship,
The material in small print is of several different kinds:
for the great privilege of contributing to the Textbook
first, anatomical, chemical, and other information that
of Medical Physiology, and for the inspiration that he
is needed for immediate discussion but that most stu-
provided to all who knew him.
dents will learn in more detail in other courses; second,
physiologic information of special importance to
certain fields of clinical medicine; and, third, informa-
John E. Hall
tion that will be of value to those students who may
Jackson, Mississippi
TABLE OF CONTENTS
The DNA Code in the Cell Nucleus Is
U N I T I
Transferred to an RNA Code in the
Introduction to Physiology: The
Cell Cytoplasm—The Process
of Transcription
30
Cell and General Physiology
Synthesis of RNA
30
Assembly of the RNA Chain from Activated
Nucleotides Using the DNA Strand
C H A P T E R
1
as a Template—The Process of
Functional Organization of the
“Transcription”
31
Messenger RNA—The Codons
31
Human Body and Control of the
Transfer RNA—The Anticodons
32
“Internal Environment”
3
Ribosomal RNA
33
Cells as the Living Units of the Body
3
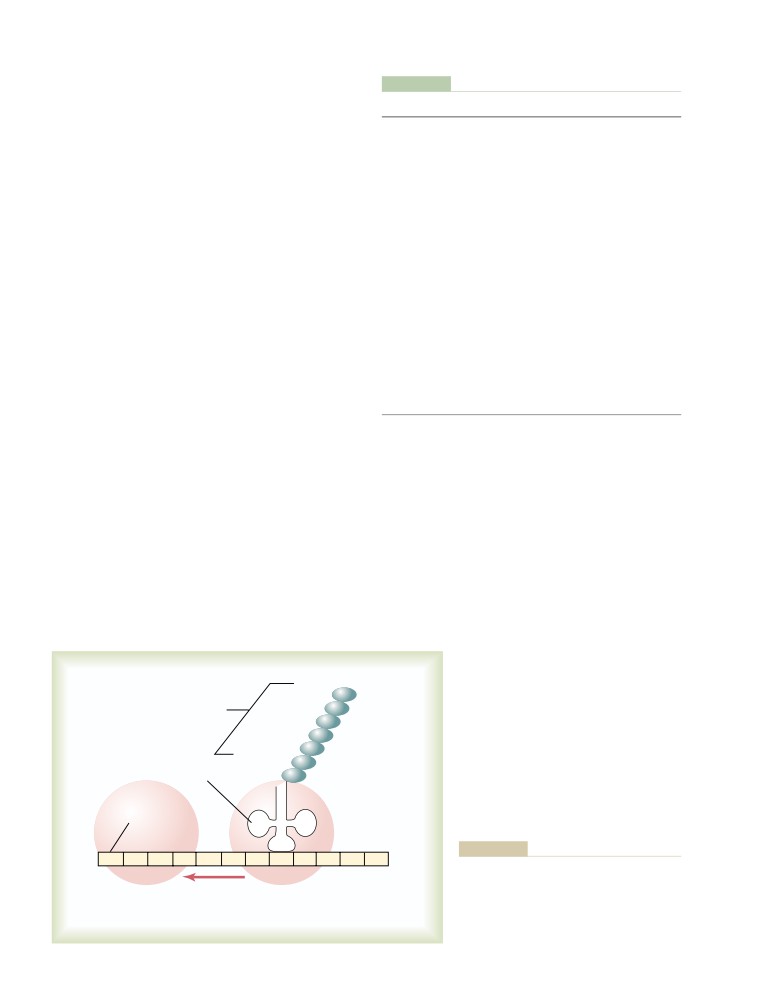
Formation of Proteins on the Ribosomes—
Extracellular Fluid—The “Internal
The Process of “Translation”
33
Environment”
3
Synthesis of Other Substances in the
“Homeostatic” Mechanisms of the Major
Cell
35
Functional Systems
4
Control of Gene Function and
Homeostasis
4
Biochemical Activity in Cells
35
Extracellular Fluid Transport and Mixing
Genetic Regulation
35
System—The Blood Circulatory System
4
Control of Intracellular Function by
Origin of Nutrients in the Extracellular Fluid
5
Enzyme Regulation
36
Removal of Metabolic End Products
5
The DNA-Genetic System Also Controls
Regulation of Body Functions
5
Cell Reproduction
37
Reproduction
6
Cell Reproduction Begins with Replication
Control Systems of the Body
6
of DNA
37
Examples of Control Mechanisms
6
Chromosomes and Their Replication
38
Characteristics of Control Systems
7
Cell Mitosis
38
Summary—Automaticity of the Body
9
Control of Cell Growth and Cell
Reproduction
39
Cell Differentiation
40
C H A P T E R
2
Apoptosis—Programmed Cell Death
40
Cancer
40
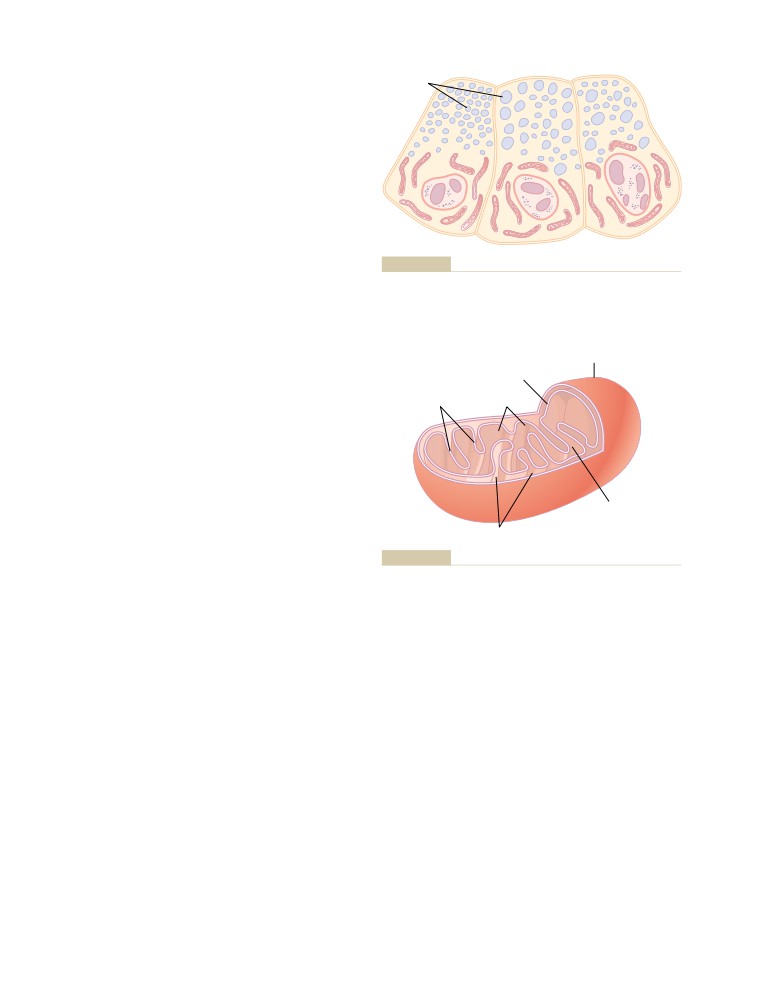
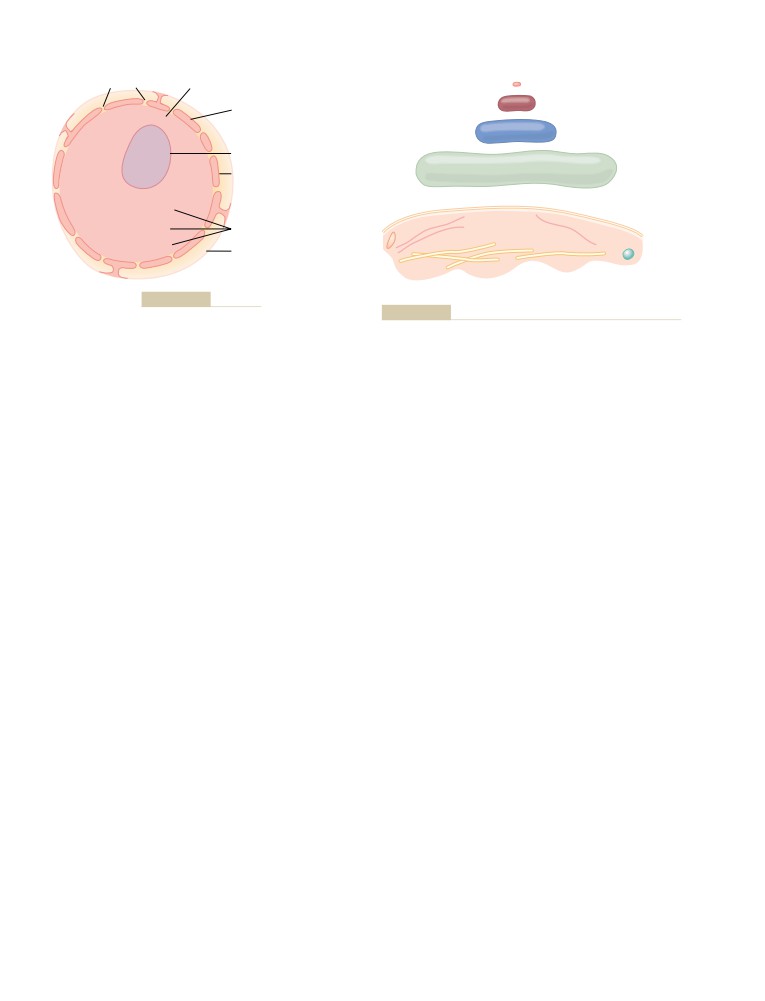
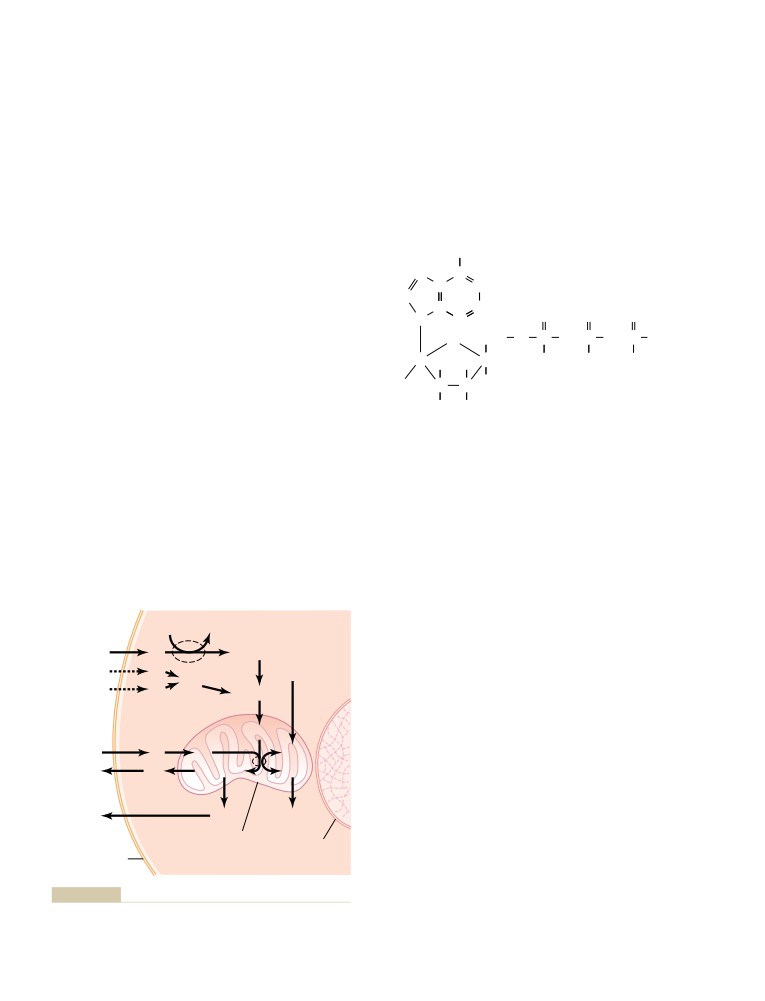
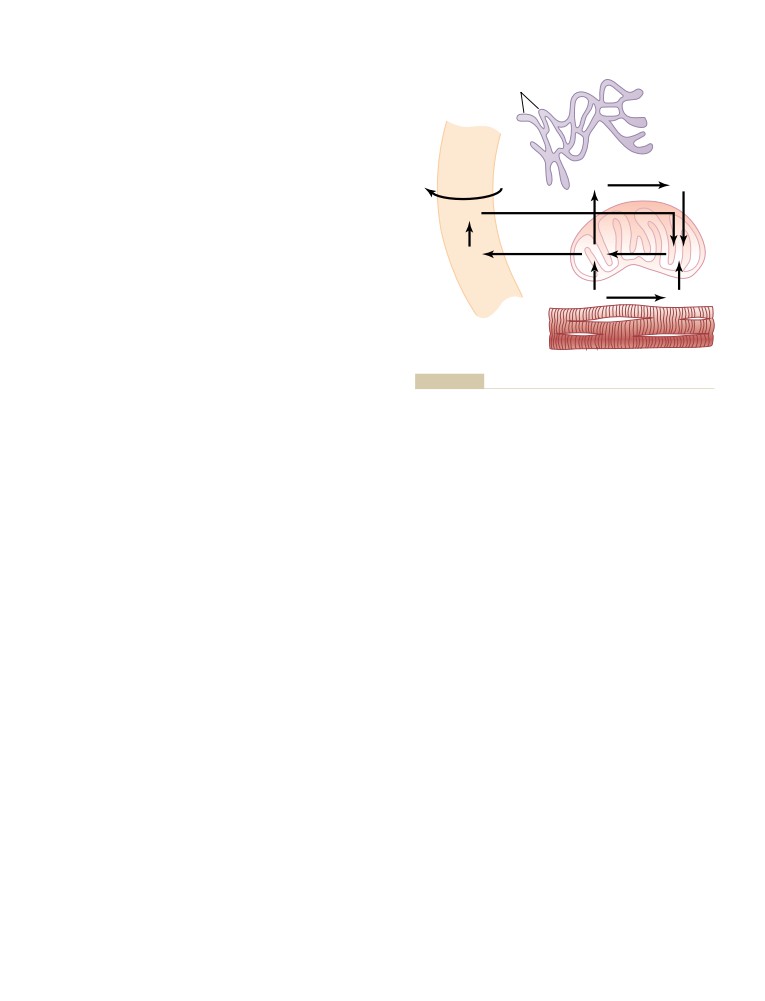
The Cell and Its Functions
11
Organization of the Cell
11
Physical Structure of the Cell
12
Membranous Structures of the Cell
12
U N I T I I
Cytoplasm and Its Organelles
14
Nucleus
17
Membrane Physiology, Nerve,
Nuclear Membrane
17
Nucleoli and Formation of Ribosomes
18
and Muscle
Comparison of the Animal Cell with
Precellular Forms of Life
18
Functional Systems of the Cell
19
C H A P T E R
4
Ingestion by the Cell—Endocytosis
19
Transport of Substances Through
Digestion of Pinocytotic and Phagocytic
Foreign Substances Inside the Cell—
the Cell Membrane
45
Function of the Lysosomes
20
The Lipid Barrier of the Cell Membrane,
Synthesis and Formation of Cellular
and Cell Membrane Transport
Structures by Endoplasmic Reticulum
Proteins
45
and Golgi Apparatus
20
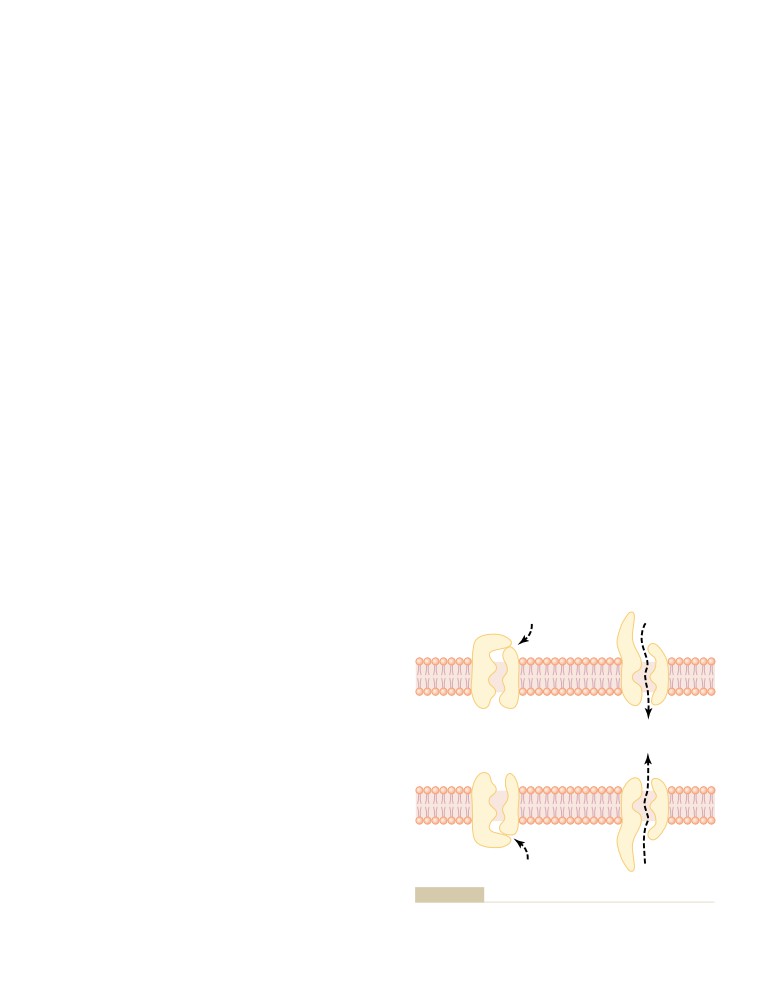
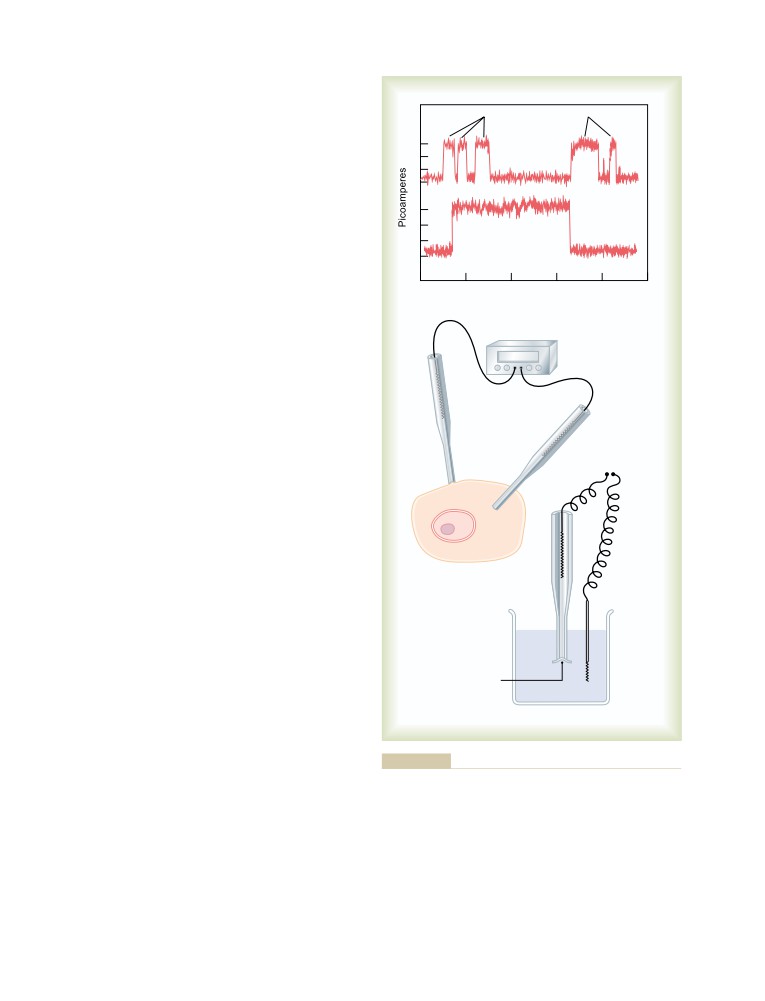
Diffusion
46
Extraction of Energy from Nutrients—
Diffusion Through the Cell Membrane
46
Function of the Mitochondria
22
Diffusion Through Protein Channels, and
Locomotion of Cells
24
“Gating” of These Channels
47
Ameboid Movement
24
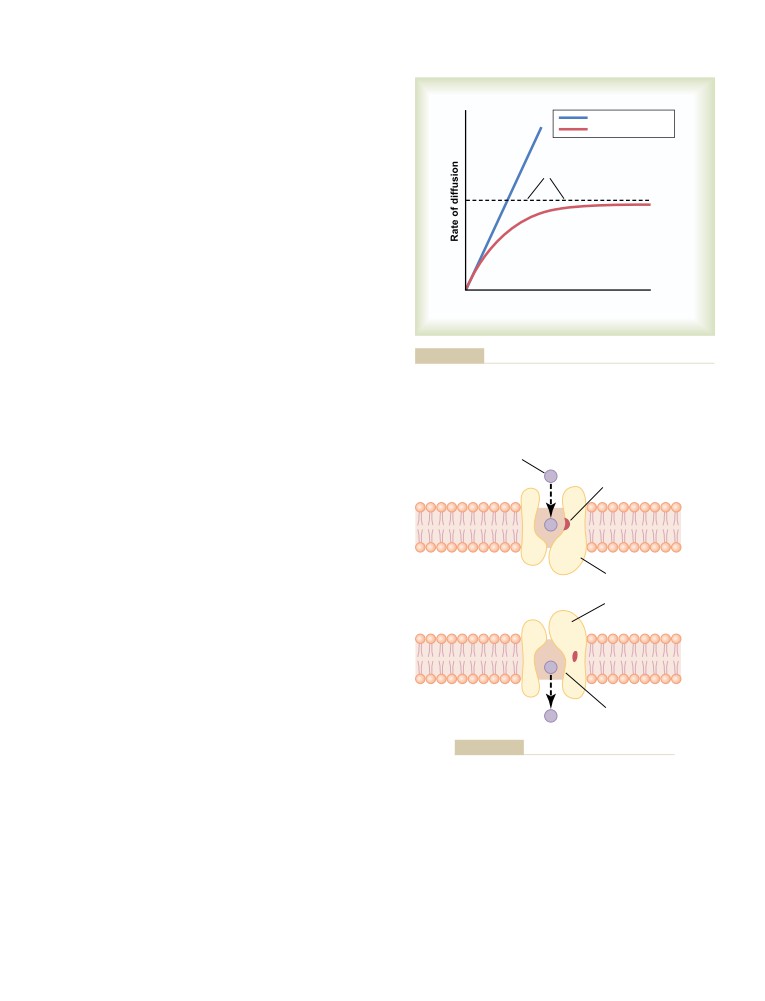
Facilitated Diffusion
49
Cilia and Ciliary Movement
24
Factors That Affect Net Rate of Diffusion
50
Osmosis Across Selectively Permeable
Membranes—“Net Diffusion” of Water
51
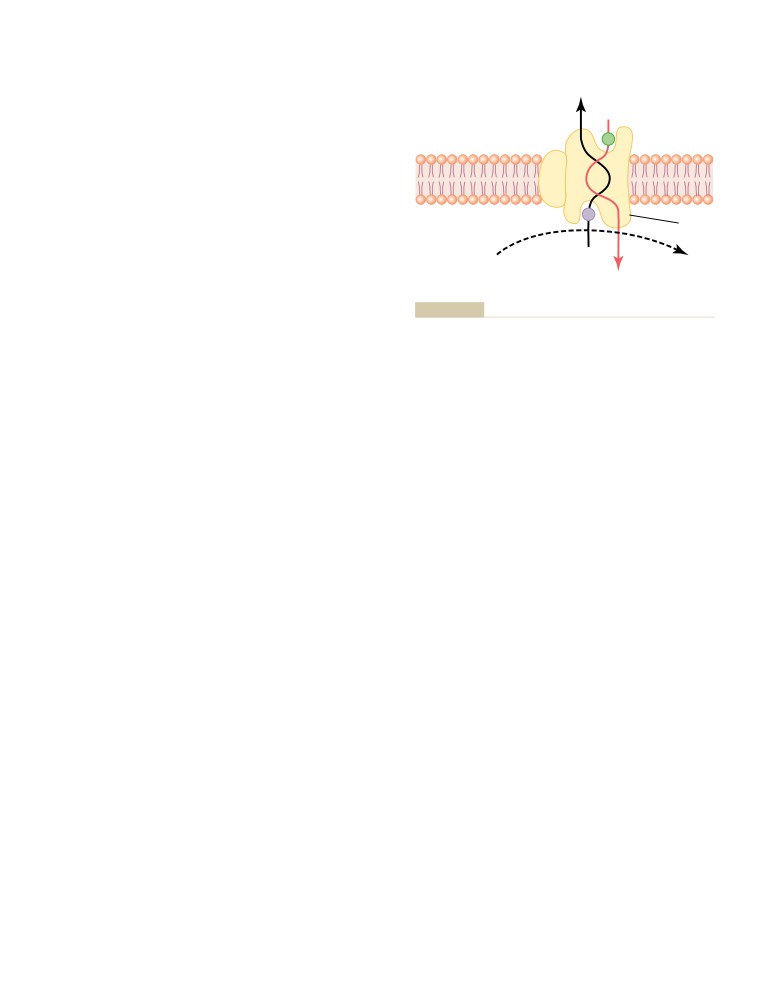
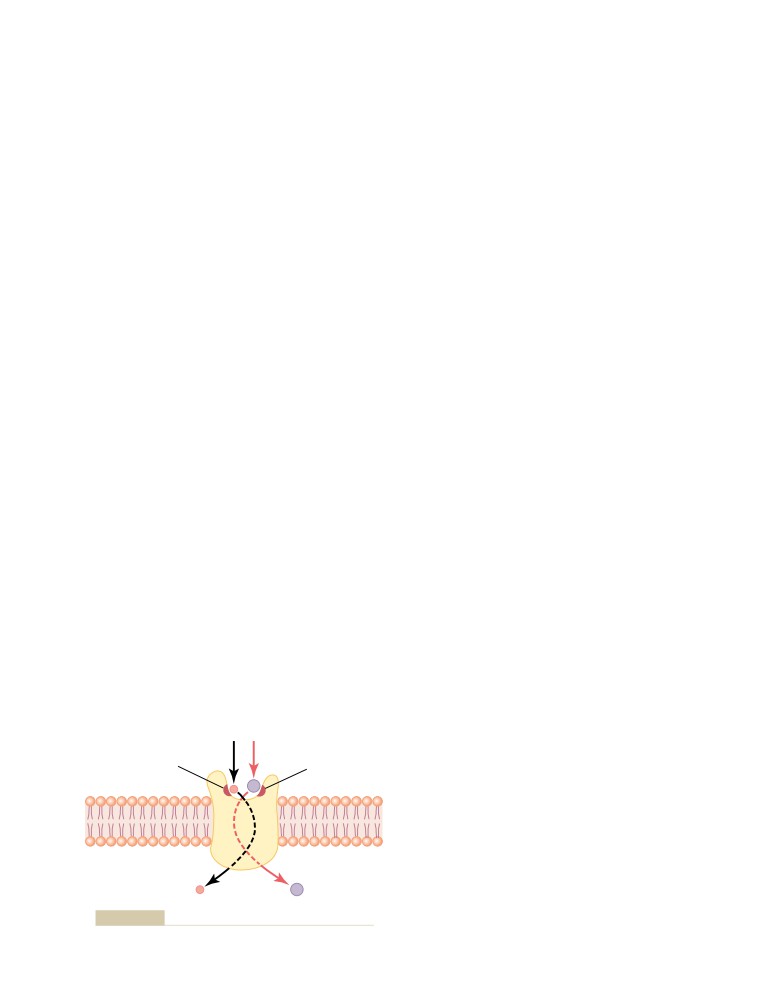
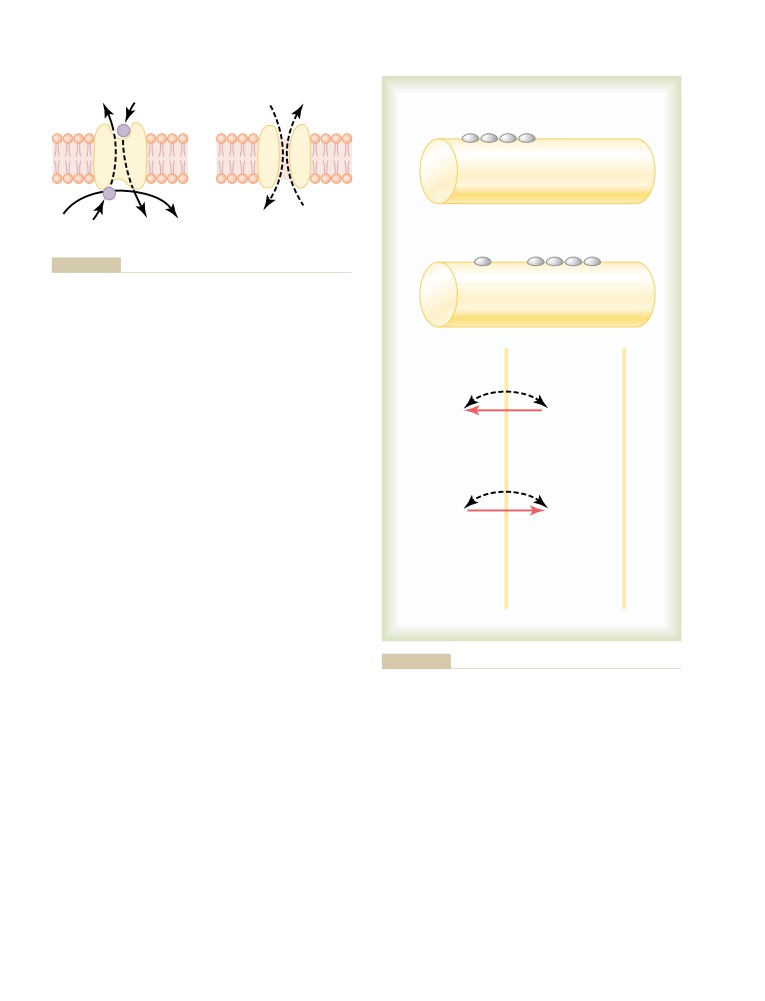
“Active Transport” of Substances
C H A P T E R
3
Through Membranes
52
Genetic Control of Protein Synthesis,
Primary Active Transport
53
Cell Function, and Cell Reproduction
27
Secondary Active Transport—Co-Transport
Genes in the Cell Nucleus
27
and Counter-Transport
54
Genetic Code
29
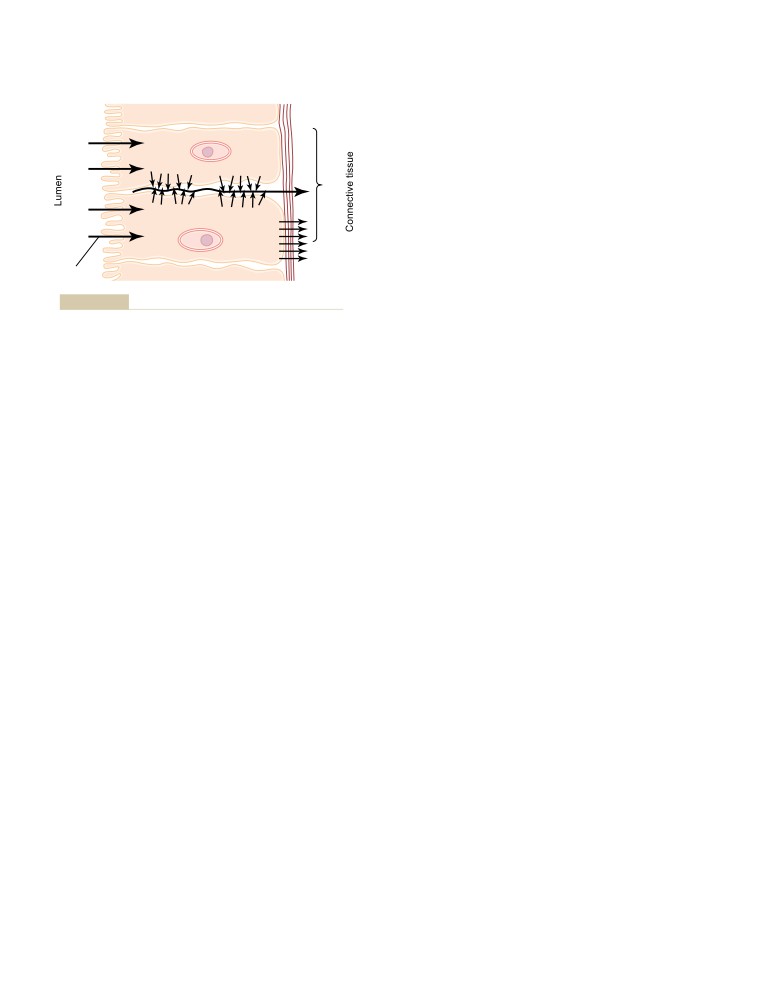
Active Transport Through Cellular Sheets
55
xiii
xiv
Table of Contents
C H A P T E R
5
C H A P T E R
7
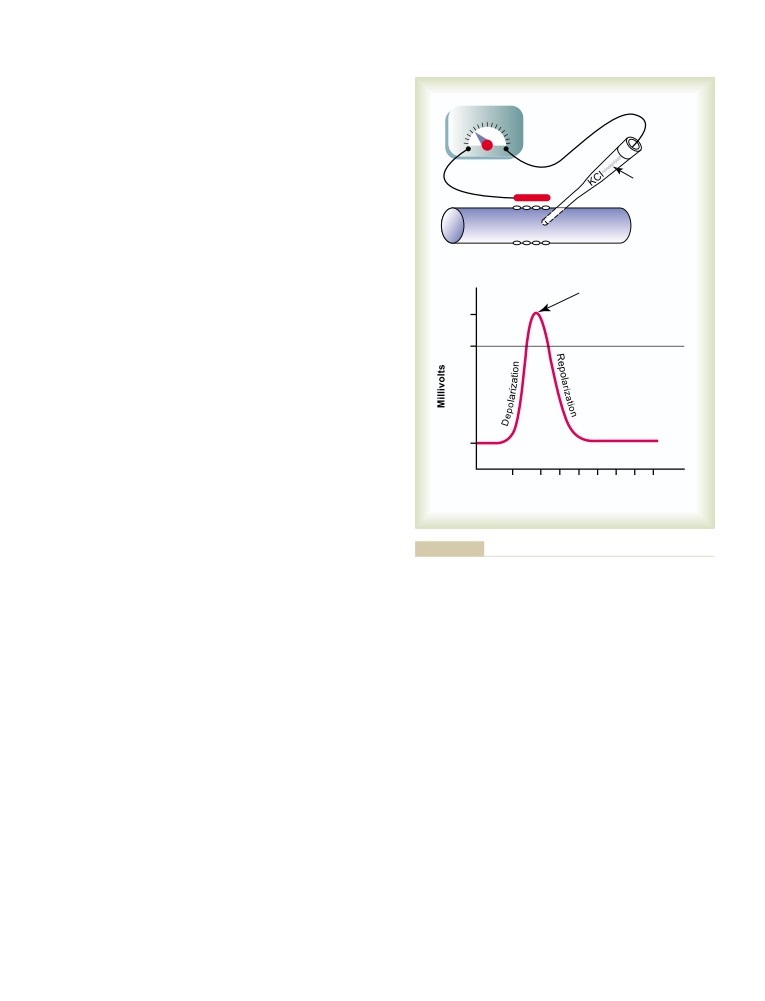
Membrane Potentials and Action
Excitation of Skeletal Muscle:
Potentials
57
Neuromuscular Transmission and
Basic Physics of Membrane
Excitation-Contraction Coupling
85
Potentials
57
Transmission of Impulses from Nerve
Membrane Potentials Caused by
Endings to Skeletal Muscle Fibers:
Diffusion
57
The Neuromuscular Junction
85
Measuring the Membrane Potential
58
Secretion of Acetylcholine by the Nerve
Resting Membrane Potential of Nerves
59
Terminals
85
Origin of the Normal Resting Membrane
Molecular Biology of Acetyline
Potential
60
Formation and Release
88
Nerve Action Potential
61
Drugs That Enhance or Block
Voltage-Gated Sodium and Potassium
Transmission at the Neuromuscular
Channels
62
Junction
88
Summary of the Events That Cause the
Myasthenia Gravis
89
Action Potential
64
Muscle Action Potential
89
Roles of Other Ions During the Action
Spread of the Action Potential to the
Potential
64
Interior of the Muscle Fiber by Way of
Initiation of the Action Potential
65
“Transverse Tubules”
89
Propagation of the Action Potential
65
Excitation-Contraction Coupling
89
Re-establishing Sodium and Potassium
Transverse Tubule-Sarcoplasmic Reticulum
Ionic Gradients After Action Potentials
System
89
Are Completed—Importance of Energy
Release of Calcium Ions by the
Metabolism
66
Sarcoplasmic Reticulum
90
Plateau in Some Action Potentials
66
Rhythmicity of Some Excitable Tissues—
Repetitive Discharge
67
C H A P T E R
8
Special Characteristics of Signal
Contraction and Excitation of
Transmission in Nerve Trunks
68
Smooth Muscle
92
Excitation—The Process of Eliciting
Contraction of Smooth Muscle
92
the Action Potential
69
Types of Smooth Muscle
92
“Refractory Period” After an Action
Contractile Mechanism in Smooth Muscle
93
Potential
70
Regulation of Contraction by Calcium Ions
95
Recording Membrane Potentials and
Nervous and Hormonal Control of
Action Potentials
70
Smooth Muscle Contraction
95
Inhibition of Excitability—“Stabilizers”
Neuromuscular Junctions of Smooth
and Local Anesthetics
70
Muscle
95
Membrane Potentials and Action Potentials
in Smooth Muscle
96
Effect of Local Tissue Factors and
Hormones to Cause Smooth Muscle
C H A P T E R
6
Contraction Without Action Potentials
98
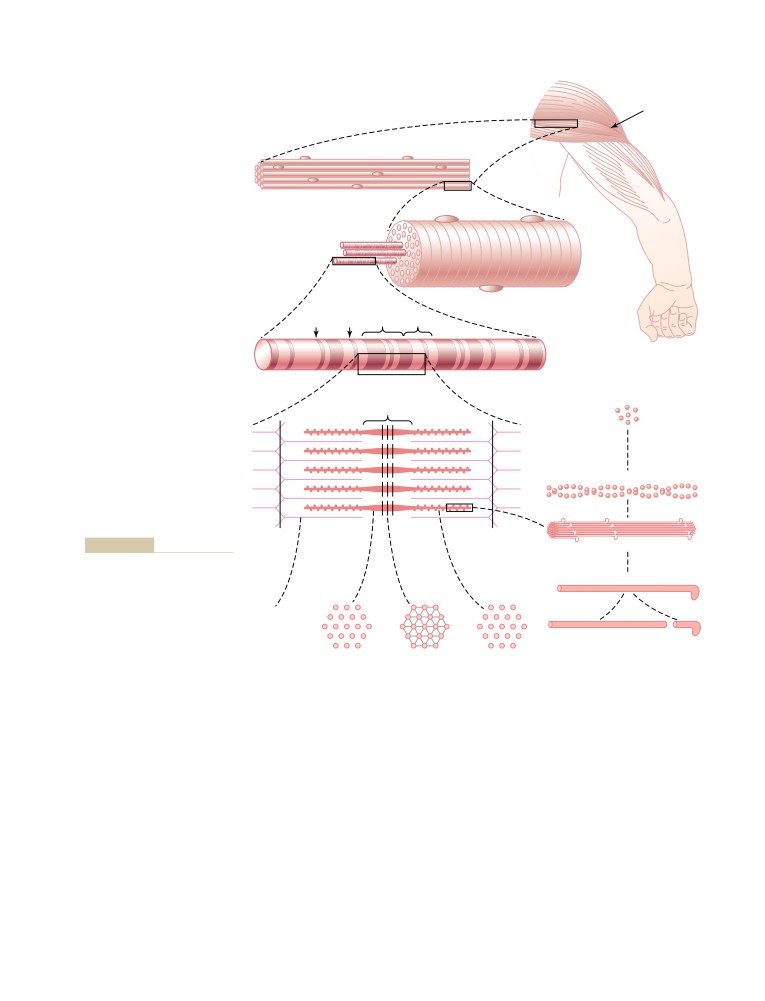
Contraction of Skeletal Muscle
72
Source of Calcium Ions That Cause
Contraction (1) Through the Cell
Physiologic Anatomy of Skeletal
Membrane and (2) from the Sarcoplasmic
Muscle
72
Reticulum
99
Skeletal Muscle Fiber
72
General Mechanism of Muscle
Contraction
74
Molecular Mechanism of Muscle
U N I T I I I
Contraction
74
Molecular Characteristics of the
The Heart
Contractile Filaments
75
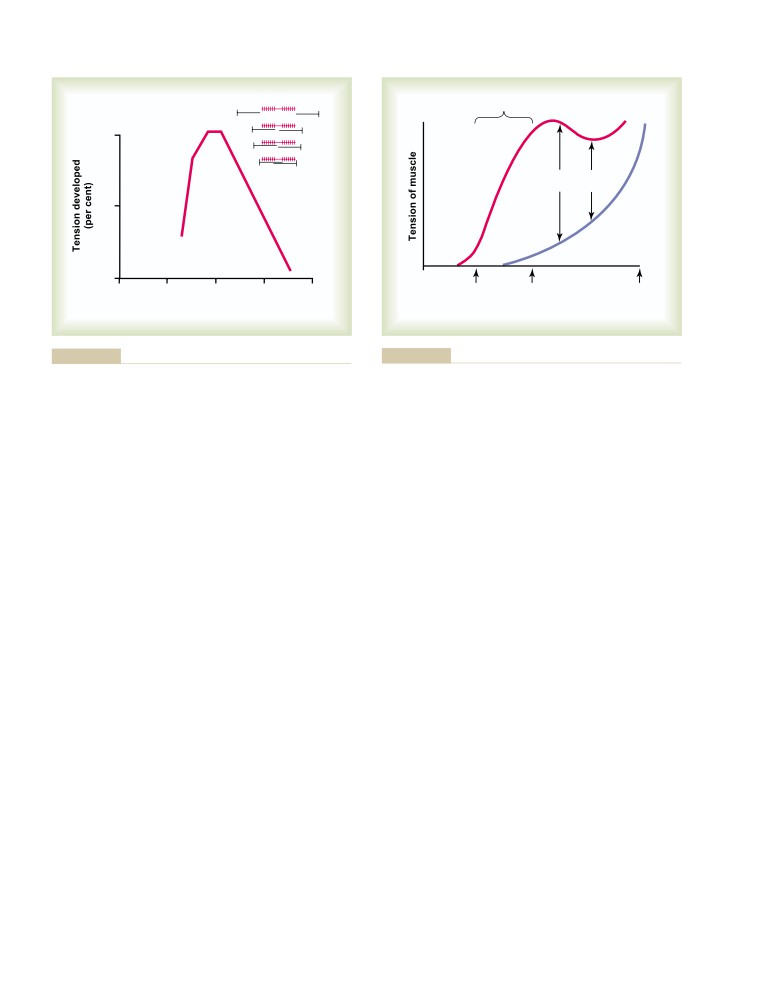
Effect of Amount of Actin and Myosin
C H A P T E R
9
Filament Overlap on Tension Developed
Heart Muscle; The Heart as a Pump
by the Contracting Muscle
77
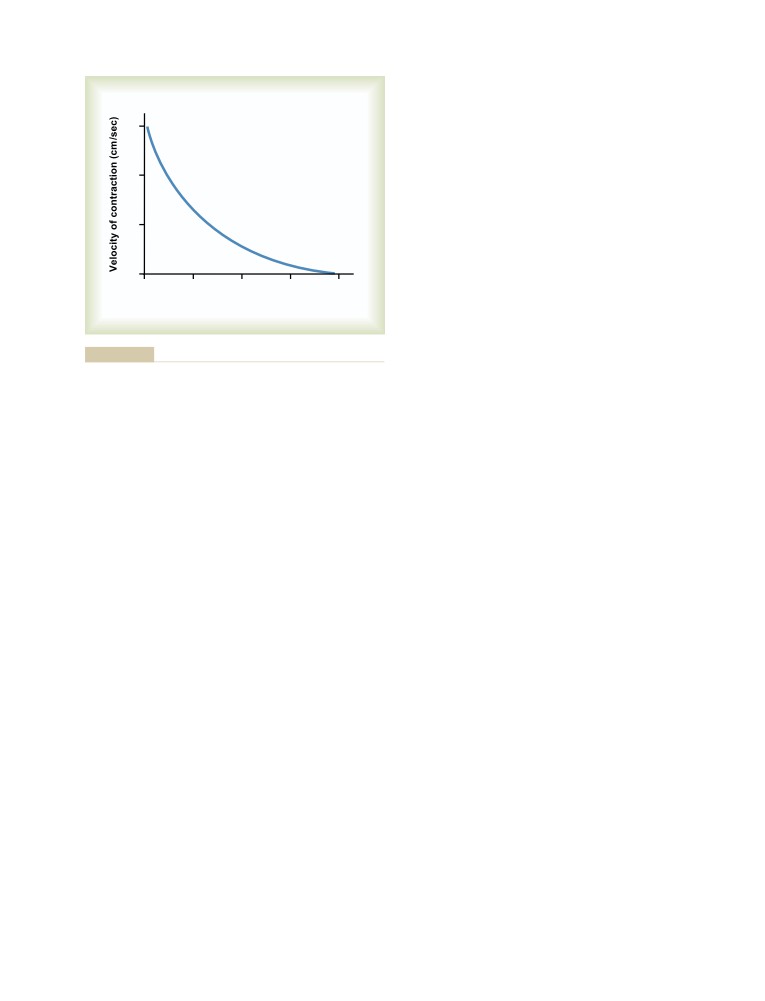
Relation of Velocity of Contraction to
and Function of the Heart Valves
103
Load
78
Physiology of Cardiac Muscle
103
Energetics of Muscle Contraction
78
Physiologic Anatomy of Cardiac Muscle
103
Work Output During Muscle Contraction
78
Action Potentials in Cardiac Muscle
104
Sources of Energy for Muscle Contraction
79
The Cardiac Cycle
106
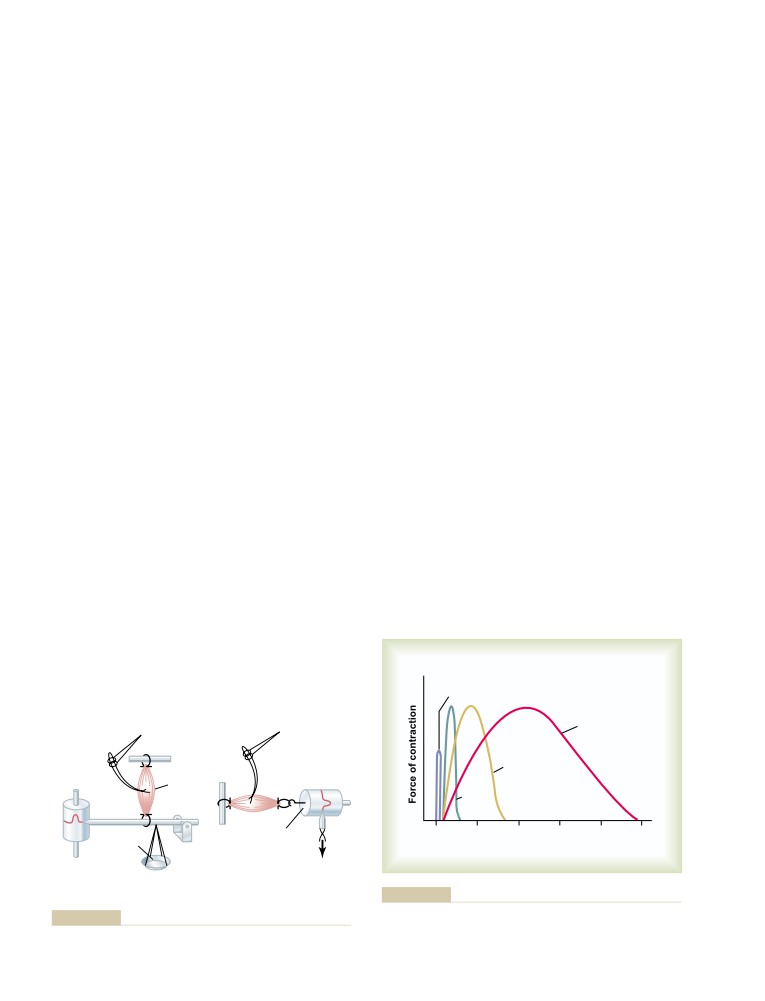
Characteristics of Whole Muscle
Diastole and Systole
106
Contraction
80
Relationship of the Electrocardiogram to
Mechanics of Skeletal Muscle Contraction
81
the Cardiac Cycle
107
Remodeling of Muscle to Match Function
82
Function of the Atria as Primer Pumps
107
Rigor Mortis
83
Function of the Ventricles as Pumps
108
Table of Contents
xv
Function of the Valves
109
Flow of Electrical Currents in the Chest
Aortic Pressure Curve
109
Around the Heart
126
Relationship of the Heart Sounds to
Electrocardiographic Leads
127
Heart Pumping
109
Three Bipolar Limb Leads
127
Work Output of the Heart
110
Chest Leads (Precordial Leads)
129
Graphical Analysis of Ventricular Pumping
110
Augmented Unipolar Limb Leads
129
Chemical Energy Required for Cardiac
Contraction: Oxygen Utilization by
the Heart
111
C H A P T E R
1
2
Regulation of Heart Pumping
111
Intrinsic Regulation of Heart Pumping—
Electrocardiographic Interpretation
The Frank-Starling Mechanism
111
of Cardiac Muscle and Coronary
Effect of Potassium and Calcium Ions on
Blood Flow Abnormalities: Vectorial
Heart Function
113
Effect of Temperature on Heart Function
114
Analysis
131
Increasing the Arterial Pressure Load
Principles of Vectorial Analysis of
(up to a Limit) Does Not Decrease the
Electrocardiograms
131
Cardiac Output
114
Use of Vectors to Represent Electrical
Potentials
131
Direction of a Vector Is Denoted in Terms
C H A P T E R
1
0
of Degrees
131
Axis for Each Standard Bipolar Lead and
Rhythmical Excitation of the Heart
116
Each Unipolar Limb Lead
132
Specialized Excitatory and Conductive
Vectorial Analysis of Potentials Recorded
System of the Heart
116
in Different Leads
133
Sinus (Sinoatrial) Node
116
Vectorial Analysis of the Normal
Internodal Pathways and Transmission of
Electrocardiogram
134
the Cardiac Impulse Through the Atria
118
Vectors That Occur at Successive Intervals
Atrioventricular Node, and Delay of Impulse
During Depolarization of the Ventricles—
Conduction from the Atria to the Ventricles
118
The QRS Complex
134
Rapid Transmission in the Ventricular
Electrocardiogram During Repolarization—
Purkinje System
119
The T Wave
134
Transmission of the Cardiac Impulse in the
Depolarization of the Atria—The P Wave
136
Ventricular Muscle
119
Vectorcardiogram
136
Summary of the Spread of the Cardiac
Mean Electrical Axis of the Ventricular
Impulse Through the Heart
120
QRS—And Its Significance
137
Control of Excitation and Conduction
Determining the Electrical Axis from
in the Heart
120
Standard Lead Electrocardiograms
137
The Sinus Node as the Pacemaker of the
Abnormal Ventricular Conditions That Cause
Heart
120
Axis Deviation
138
Role of the Purkinje System in Causing
Conditions That Cause Abnormal
Synchronous Contraction of the
Voltages of the QRS Complex
140
Ventricular Muscle
121
Increased Voltage in the Standard Bipolar
Control of Heart Rhythmicity and Impulse
Limb Leads
140
Conduction by the Cardiac Nerves: The
Decreased Voltage of the Electrocardiogram
140
Sympathetic and Parasympathetic Nerves
121
Prolonged and Bizarre Patterns of the
QRS Complex
141
Prolonged QRS Complex as a Result of
C H A P T E R
1
1
Cardiac Hypertrophy or Dilatation
141
The Normal Electrocardiogram
123
Prolonged QRS Complex Resulting from
Characteristics of the Normal
Purkinje System Blocks
141
Electrocardiogram
123
Conditions That Cause Bizarre QRS
Depolarization Waves Versus
Complexes
141
Repolarization Waves
123
Current of Injury
141
Relationship of Atrial and Ventricular
Effect of Current of Injury on the QRS
Contraction to the Waves of the
Complex
141
Electrocardiogram
125
The J Point—The Zero Reference Potential
Voltage and Time Calibration of the
for Analyzing Current of Injury
142
Electrocardiogram
125
Coronary Ischemia as a Cause of Injury
Methods for Recording
Potential
143
Electrocardiograms
126
Abnormalities in the T Wave
145
Pen Recorder
126
Effect of Slow Conduction of the
Flow of Current Around the Heart
Depolarization Wave on the
During the Cardiac Cycle
126
Characteristics of the T Wave
145
Recording Electrical Potentials from a
Shortened Depolarization in Portions of
Partially Depolarized Mass of Syncytial
the Ventricular Muscle as a Cause of
Cardiac Muscle
126
T Wave Abnormalities
145
xvi
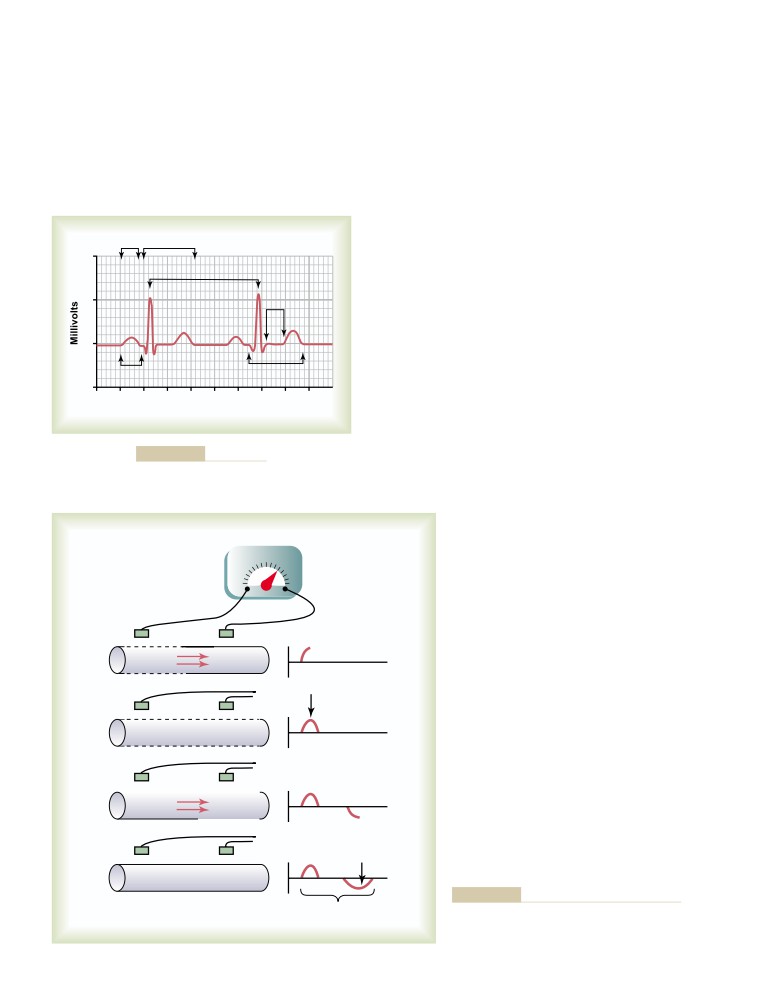
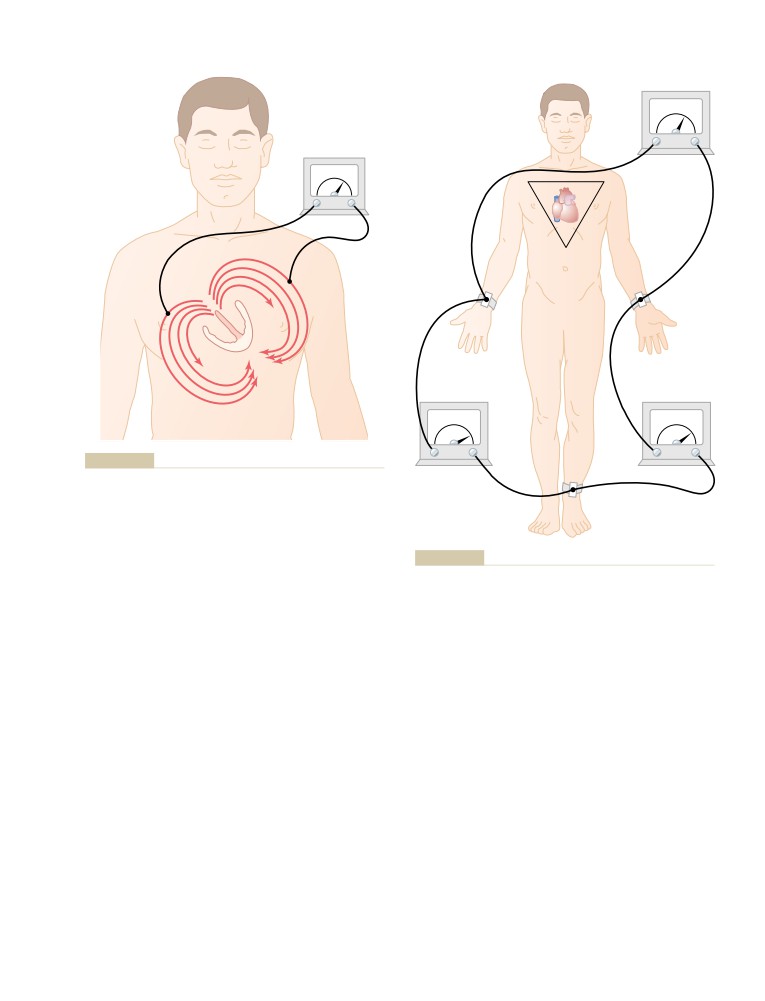
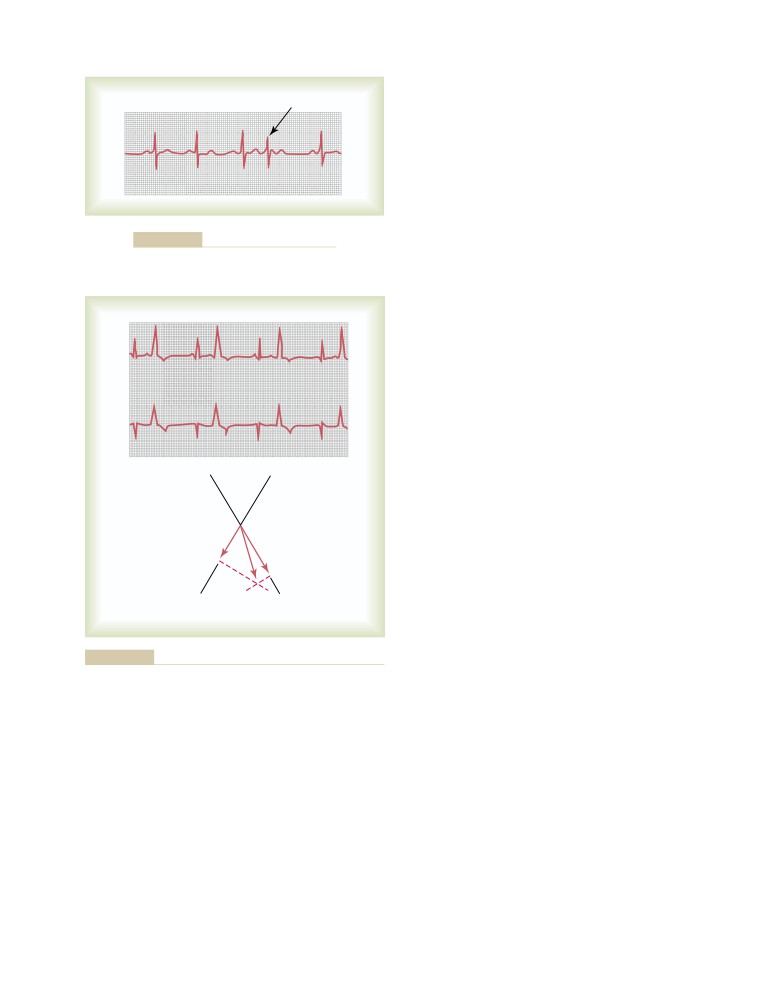
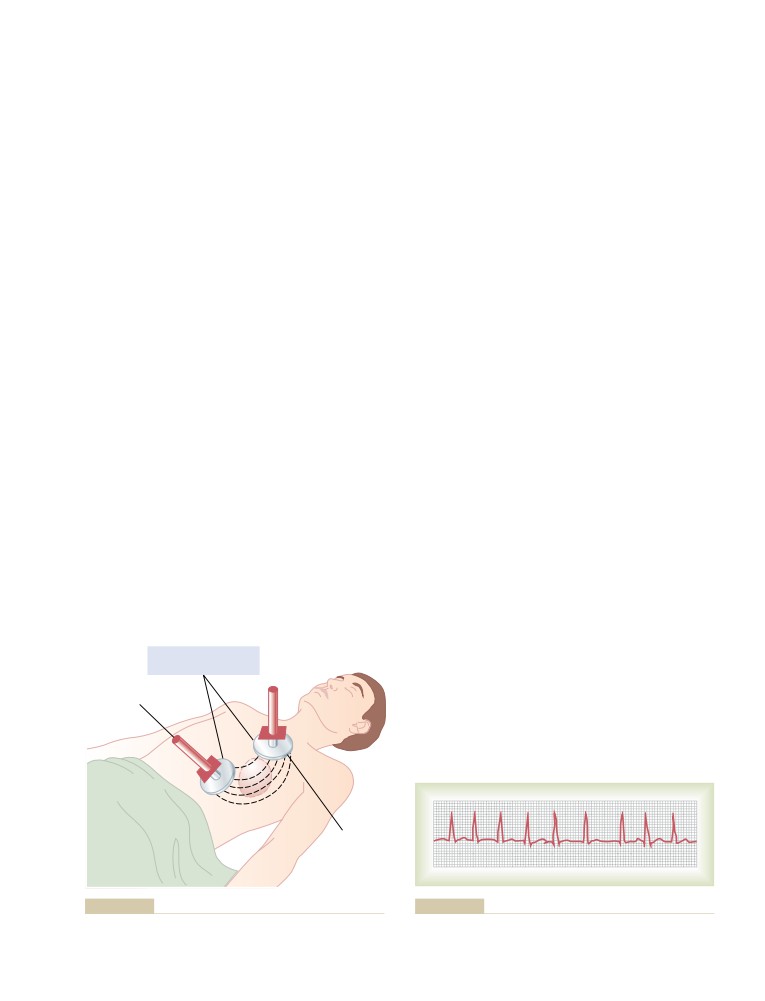
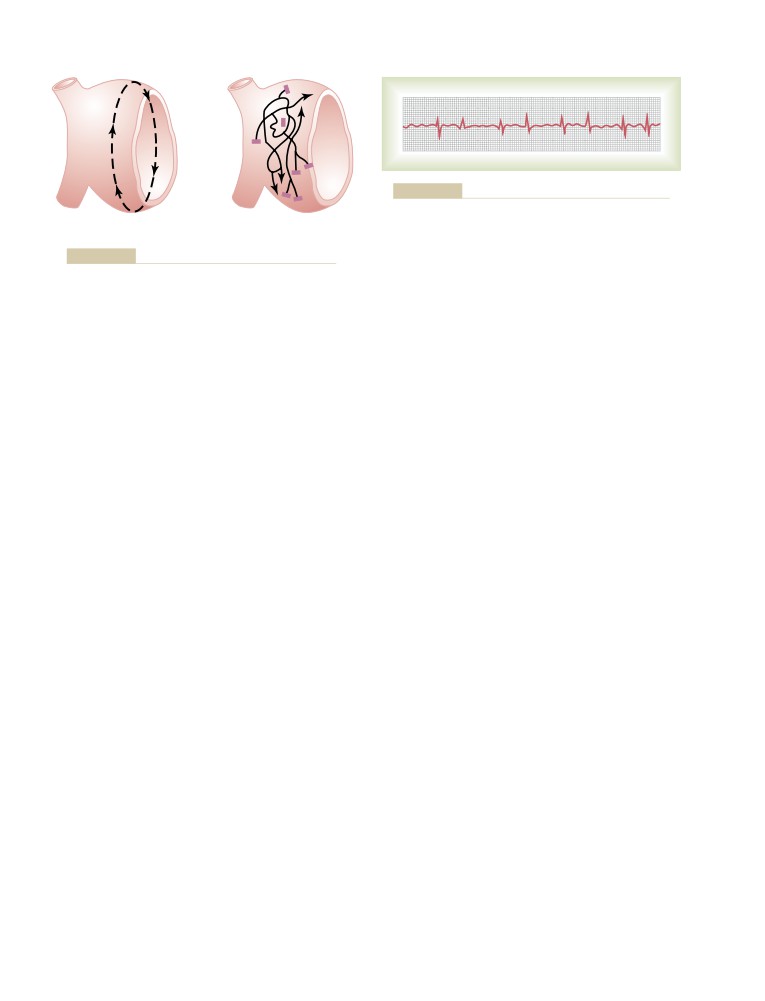
Table of Contents
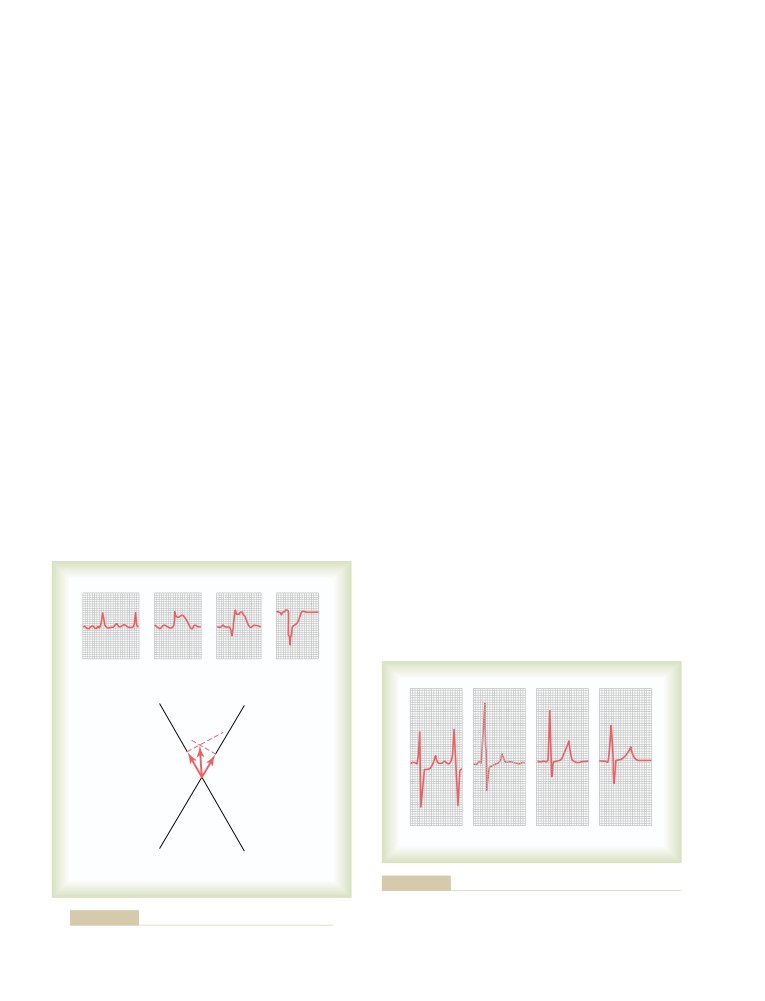
C H A P T E R
1
3
Volume-Pressure Curves of the Arterial
and Venous Circulations
172
Cardiac Arrhythmias and Their
Arterial Pressure Pulsations
173
Electrocardiographic Interpretation
147
Transmission of Pressure Pulses to the
Abnormal Sinus Rhythms
147
Peripheral Arteries
174
Tachycardia
147
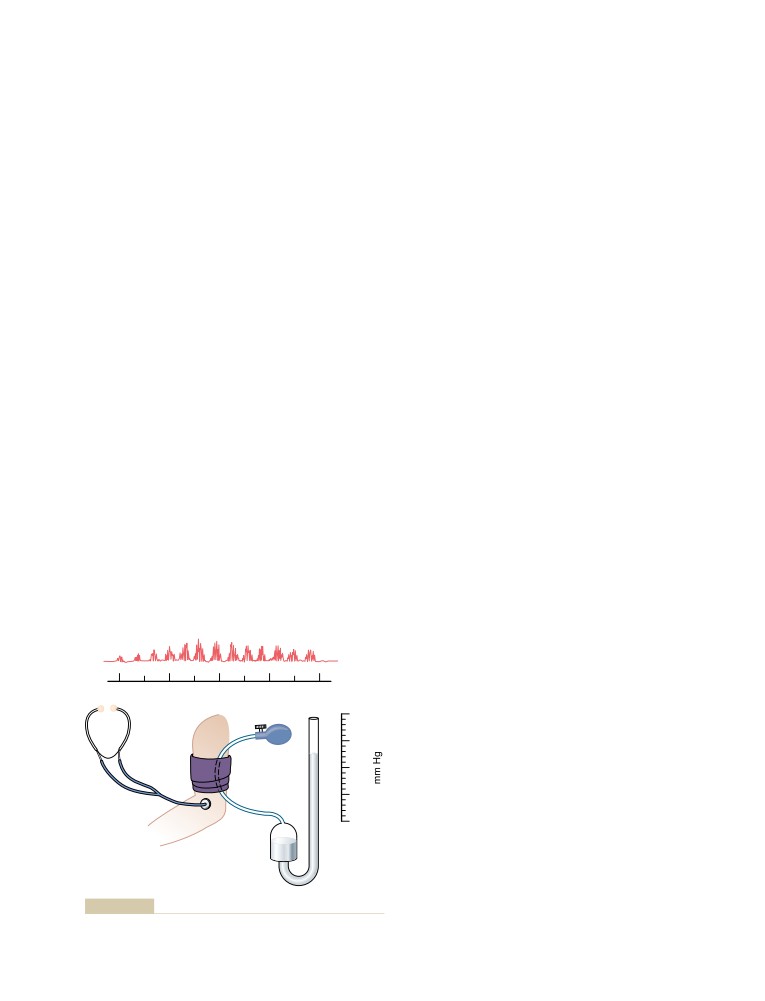
Clinical Methods for Measuring Systolic
Bradycardia
147
and Diastolic Pressures
175
Sinus Arrhythmia
148
Veins and Their Functions
176
Abnormal Rhythms That Result from
Venous Pressures—Right Atrial Pressure
Block of Heart Signals Within the
(Central Venous Pressure) and
Intracardiac Conduction Pathways
148
Peripheral Venous Pressures
176
Sinoatrial Block
148
Blood Reservoir Function of the Veins
179
Atrioventricular Block
148
Incomplete Atrioventricular Heart Block
149
Incomplete Intraventricular Block—
C H A P T E R
1
6
Electrical Alternans
150
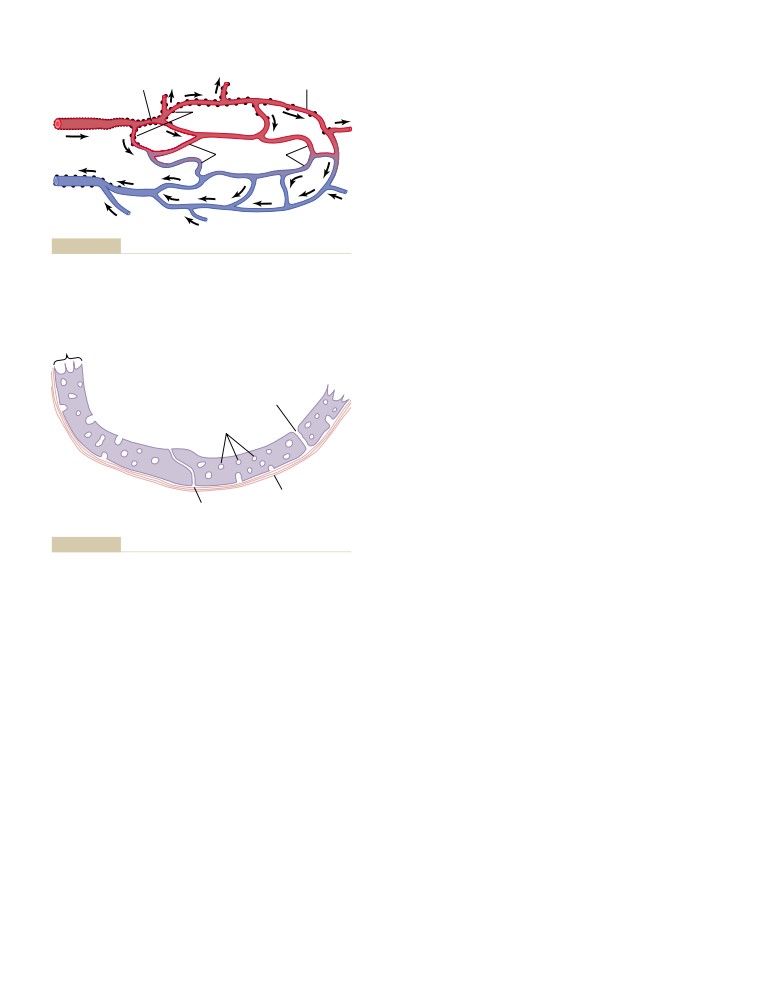
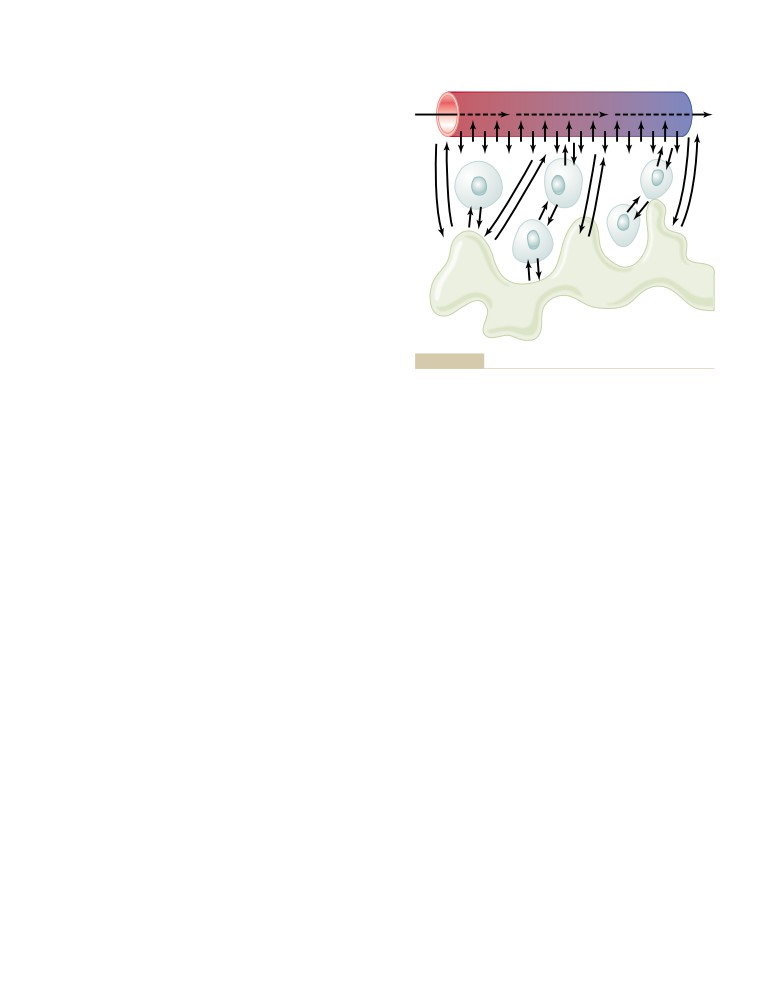
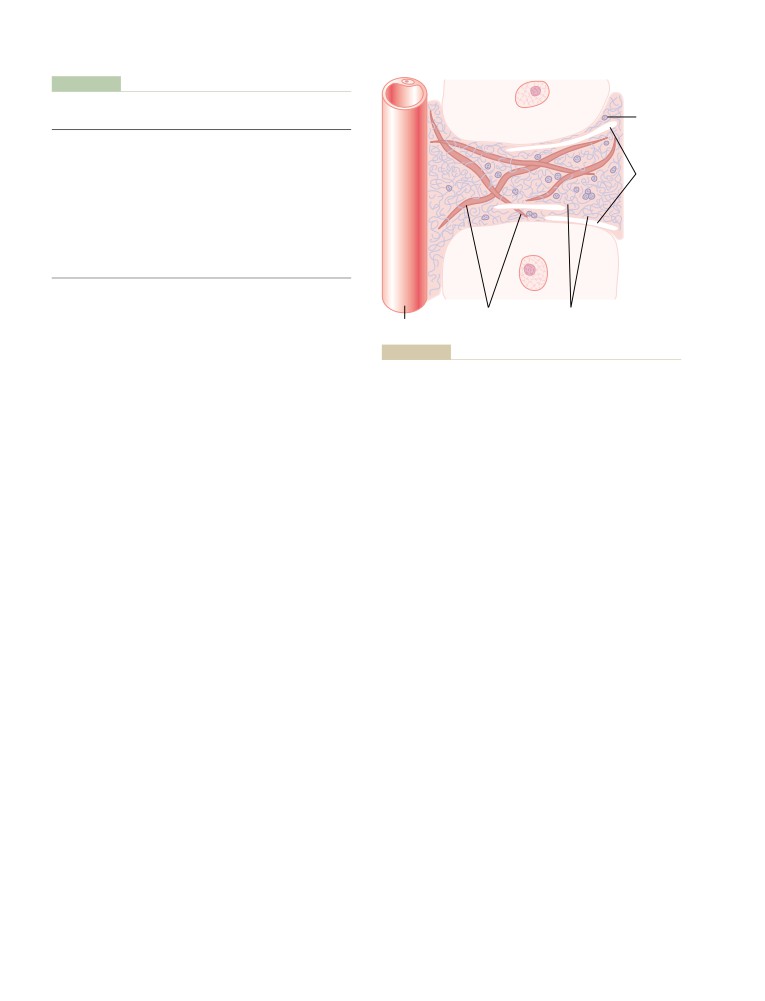
The Microcirculation and the
Premature Contractions
150
Lymphatic System: Capillary Fluid
Premature Atrial Contractions
150
A-V Nodal or A-V Bundle Premature
Exchange, Interstitial Fluid, and
Contractions
150
Lymph Flow
181
Premature Ventricular Contractions
151
Structure of the Microcirculation and
Paroxysmal Tachycardia
151
Capillary System
181
Atrial Paroxysmal Tachycardia
152
Flow of Blood in the Capillaries—
Ventricular Paroxysmal Tachycardia
152
Vasomotion
182
Ventricular Fibrillation
152
Average Function of the Capillary System
183
Phenomenon of Re-entry—“Circus
Exchange of Water, Nutrients, and
Movements” as the Basis for Ventricular
Other Substances Between the Blood
Fibrillation
153
and Interstitial Fluid
183
Chain Reaction Mechanism of Fibrillation
153
Diffusion Through the Capillary Membrane
183
Electrocardiogram in Ventricular Fibrillation
154
The Interstitium and Interstitial Fluid
184
Electroshock Defibrillation of the Ventricle
154
Fluid Filtration Across Capillaries Is
Hand Pumping of the Heart
Determined by Hydrostatic and
(Cardiopulmonary Resuscitation) as
Colloid Osmotic Pressures, and
an Aid to Defibrillation
155
Capillary Filtration Coefficient
185
Atrial Fibrillation
155
Capillary Hydrostatic Pressure
186
Atrial Flutter
156
Interstitial Fluid Hydrostatic Pressure
187
Cardiac Arrest
156
Plasma Colloid Osmotic Pressure
188
Interstitial Fluid Colloid Osmotic Pressure
188
Exchange of Fluid Volume Through the
Capillary Membrane
189
U N I T I V
Starling Equilibrium for Capillary Exchange
189
The Circulation
Lymphatic System
190
Lymph Channels of the Body
190
Formation of Lymph
191
C H A P T E R
1
4
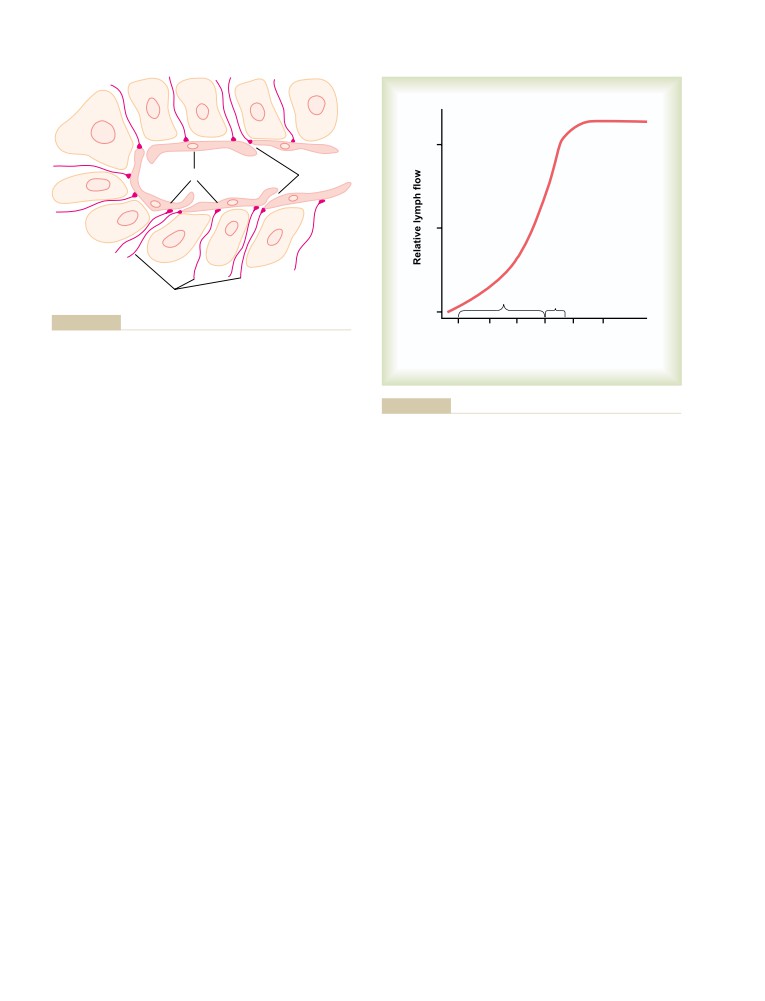
Rate of Lymph Flow
192
Role of the Lymphatic System in Controlling
Overview of the Circulation; Medical
Interstitial Fluid Protein Concentration,
Physics of Pressure, Flow, and
Interstitial Fluid Volume, and Interstitial
Resistance
161
Fluid Pressure
193
Physical Characteristics of the
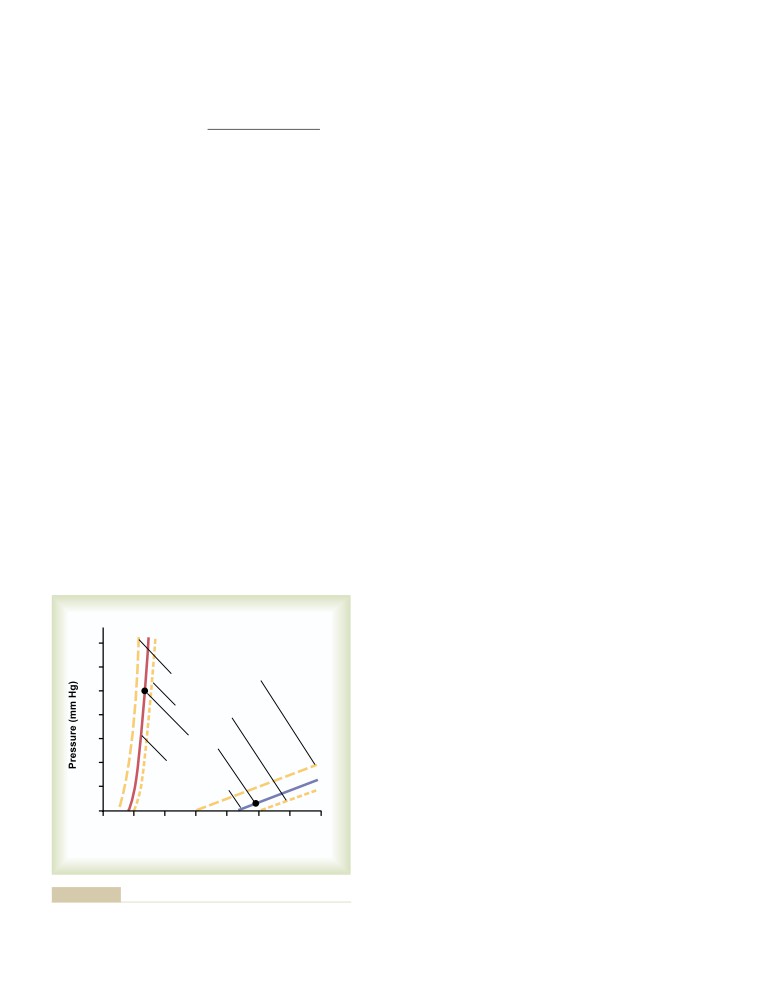
Circulation
161
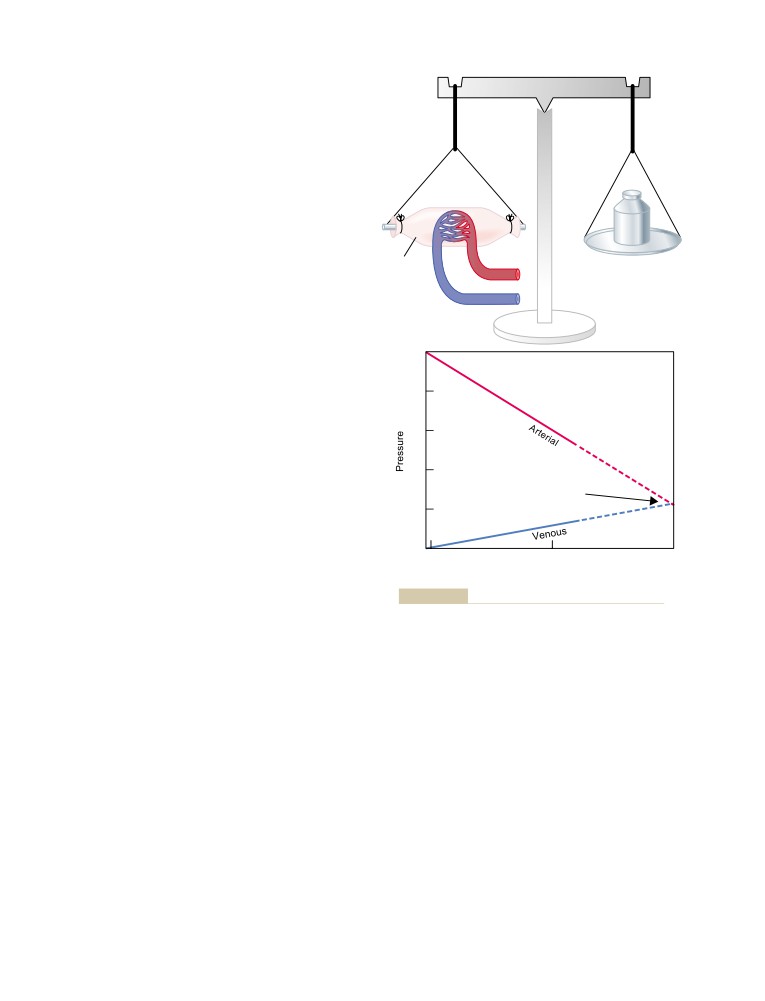
Basic Theory of Circulatory Function
163
C H A P T E R
1
7
Interrelationships Among Pressure,
Local and Humoral Control of Blood
Flow, and Resistance
164
Flow by the Tissues
195
Blood Flow
164
Local Control of Blood Flow in Response
Blood Pressure
166
to Tissue Needs
195
Resistance to Blood Flow
167
Mechanisms of Blood Flow Control
196
Effects of Pressure on Vascular Resistance
Acute Control of Local Blood Flow
196
and Tissue Blood Flow
170
Long-Term Blood Flow Regulation
200
Development of Collateral Circulation—A
Phenomenon of Long-Term Local Blood
C H A P T E R
1
5
Flow Regulation
201
Vascular Distensibility and Functions
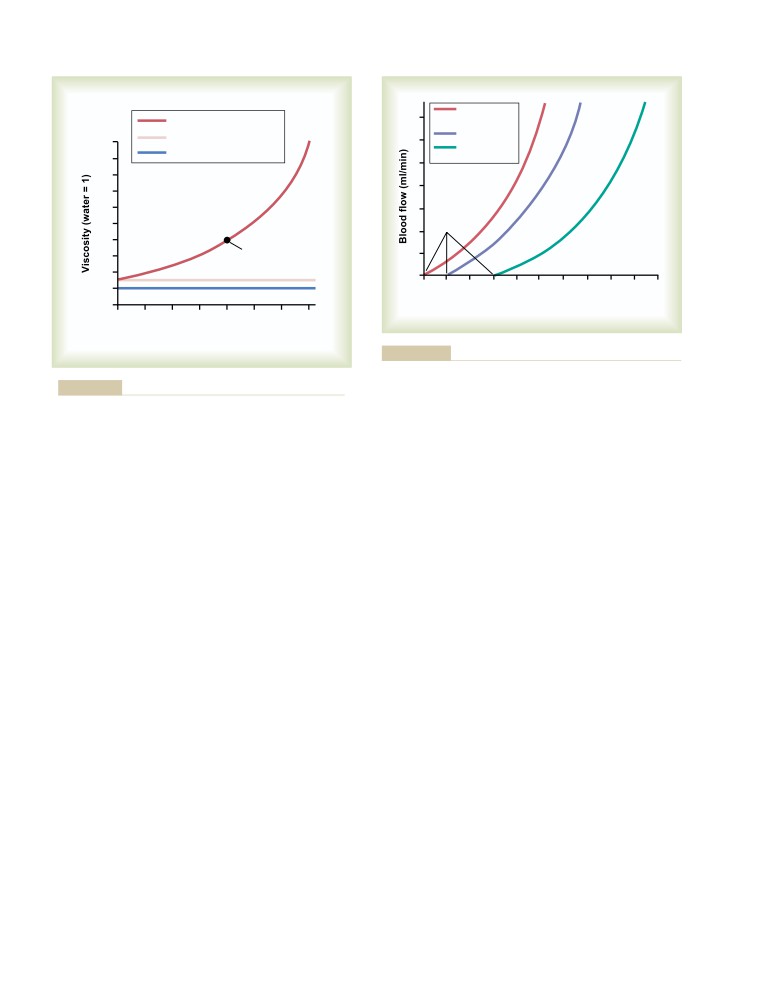
Humoral Control of the Circulation
201
of the Arterial and Venous Systems
171
Vasoconstrictor Agents
201
Vascular Distensibility
171
Vasodilator Agents
202
Vascular Compliance (or Vascular
Vascular Control by Ions and Other
Capacitance)
171
Chemical Factors
202
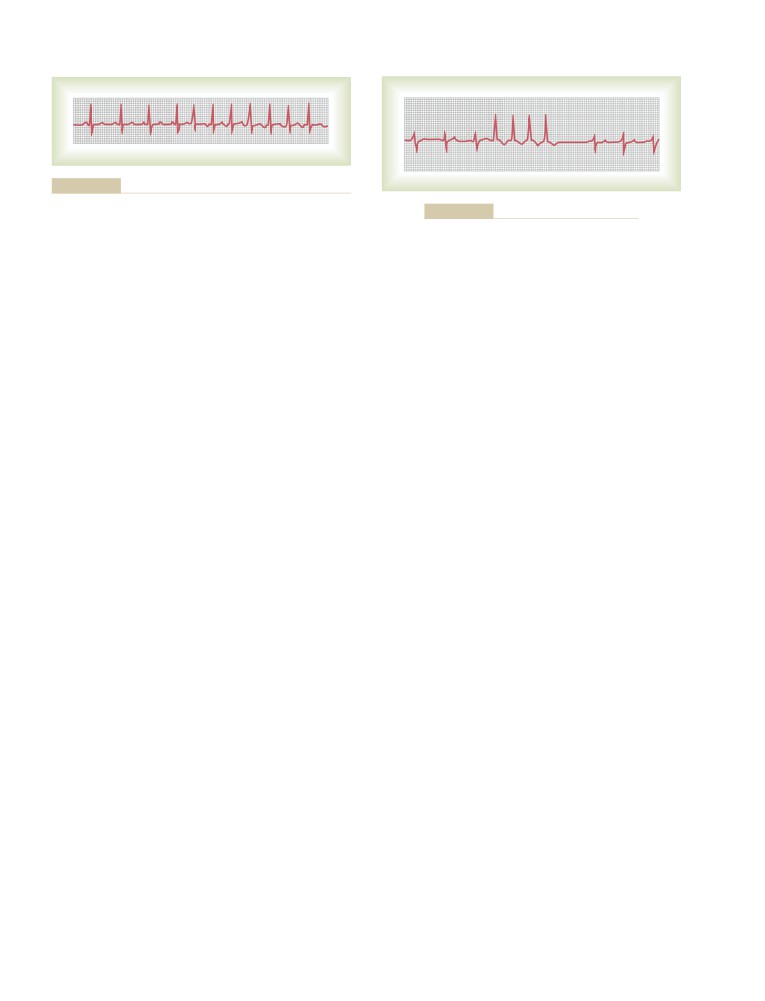
Table of Contents
xvii
C H A P T E R
1
8
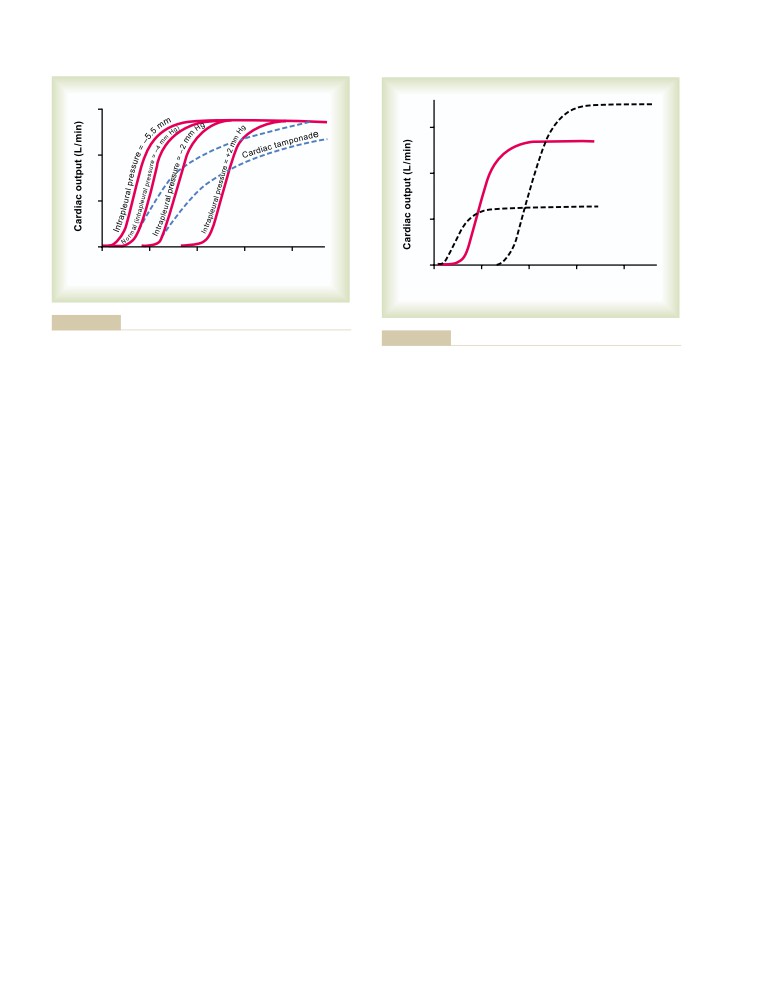
Cardiac Output Regulation Is the Sum of
Blood Flow Regulation in All the Local
Nervous Regulation of the Circulation,
Tissues of the Body—Tissue Metabolism
and Rapid Control of Arterial Pressure
204
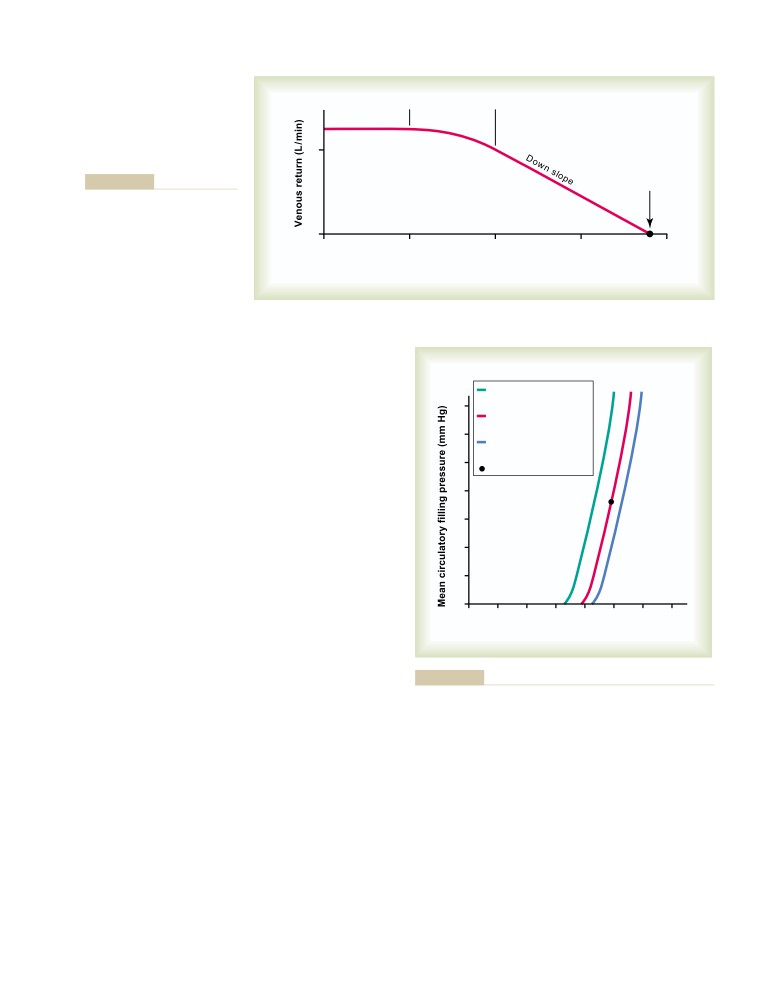
Regulates Most Local Blood Flow
233
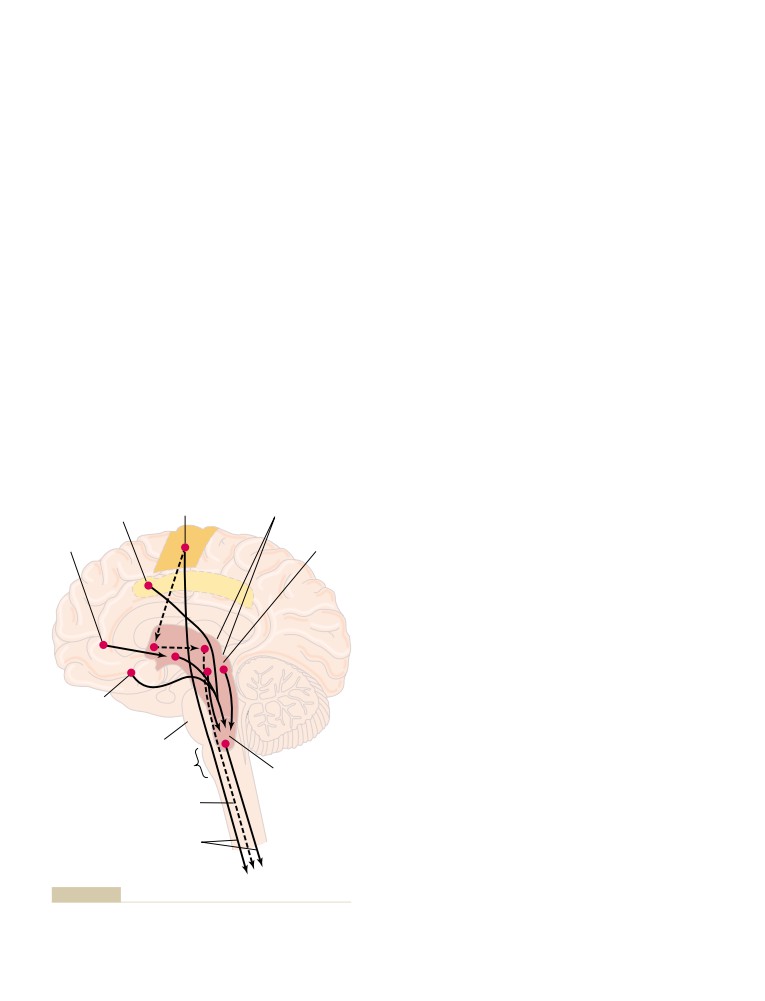
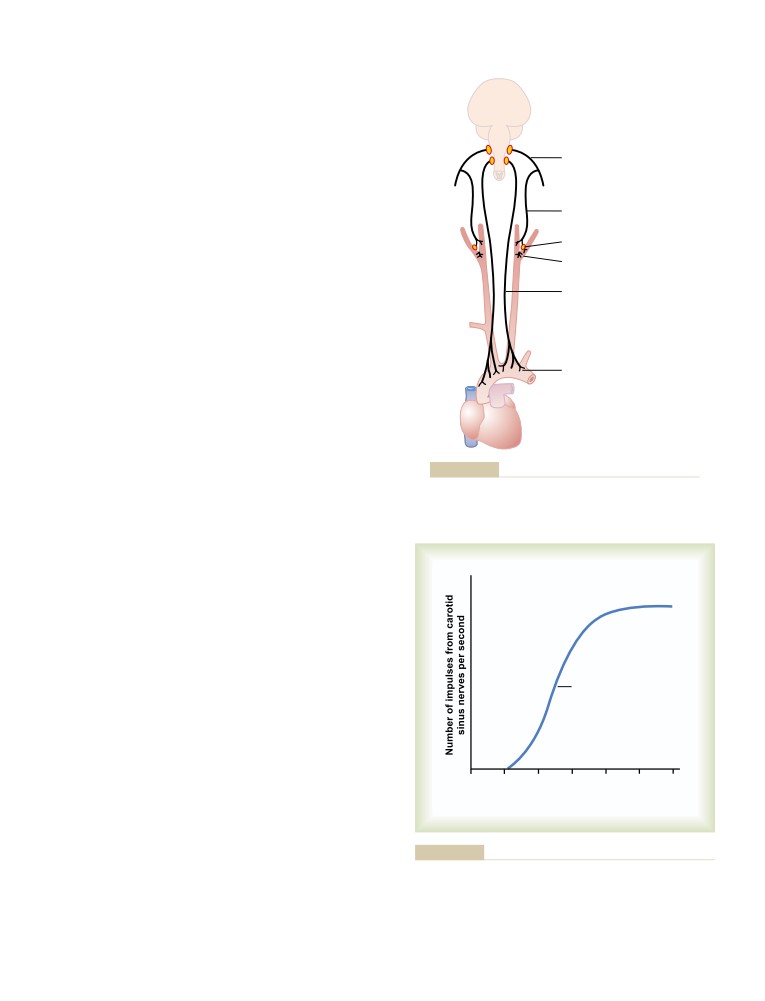
Nervous Regulation of the Circulation
204
The Heart Has Limits for the Cardiac Output
Autonomic Nervous System
204
That It Can Achieve
234
Role of the Nervous System in Rapid
What Is the Role of the Nervous System in
Control of Arterial Pressure
208
Controlling Cardiac Output?
235
Increase in Arterial Pressure During Muscle
Pathologically High and Pathologically
Exercise and Other Types of Stress
208
Low Cardiac Outputs
236
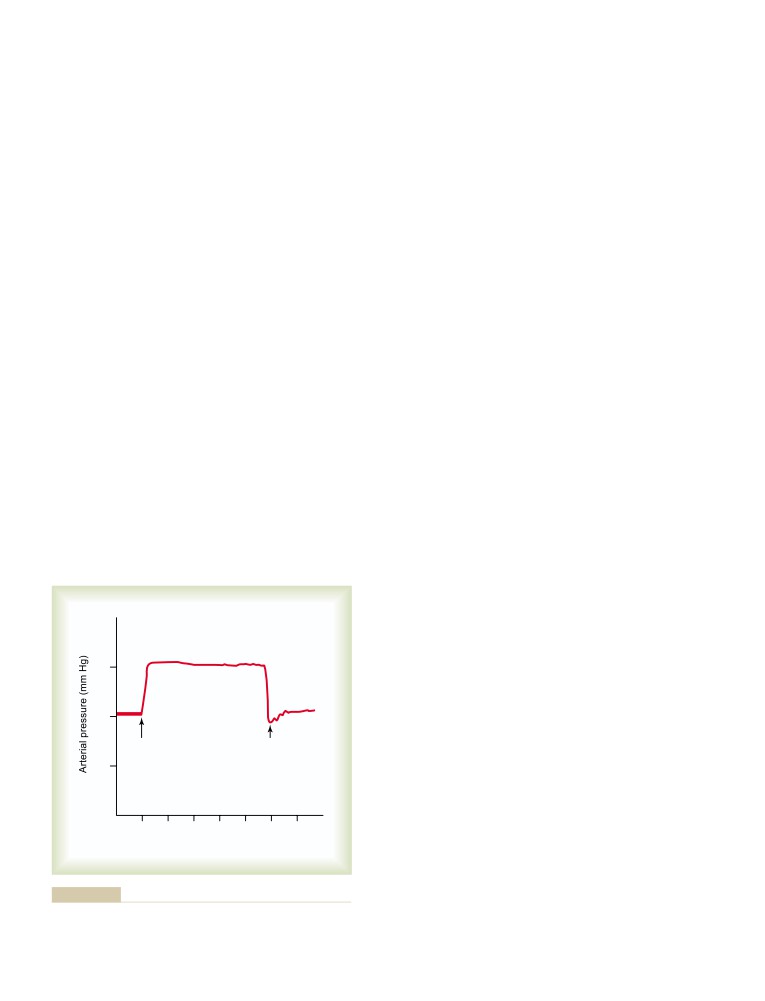
Reflex Mechanisms for Maintaining Normal
High Cardiac Output Caused by Reduced
Arterial Pressure
209
Total Peripheral Resistance
236
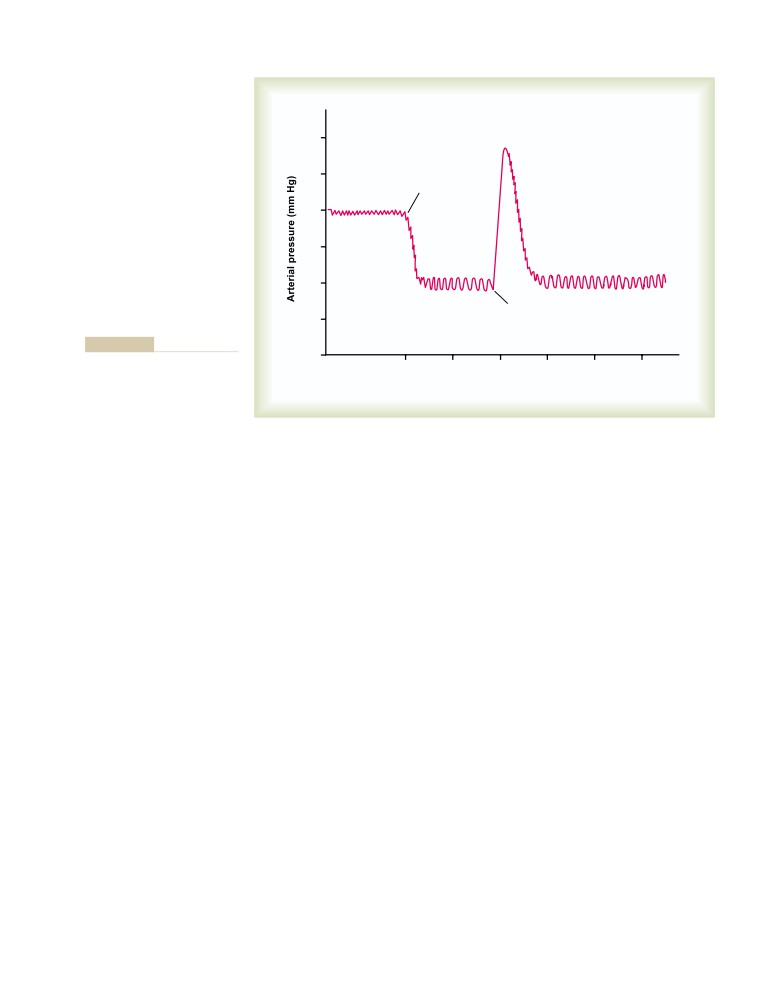
Central Nervous System Ischemic
Low Cardiac Output
237
Response—Control of Arterial Pressure
A More Quantitative Analysis of Cardiac
by the Brain’s Vasomotor Center in
Output Regulation
237
Response to Diminished Brain Blood
Cardiac Output Curves Used in the
Flow
212
Quantitative Analysis
237
Special Features of Nervous Control
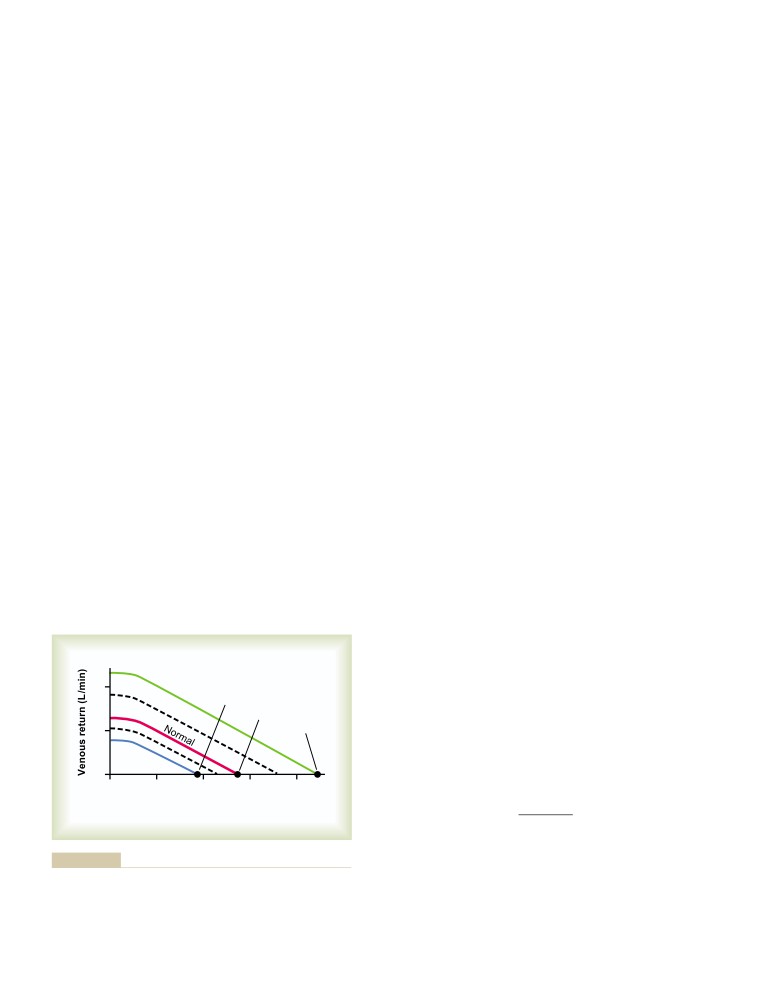
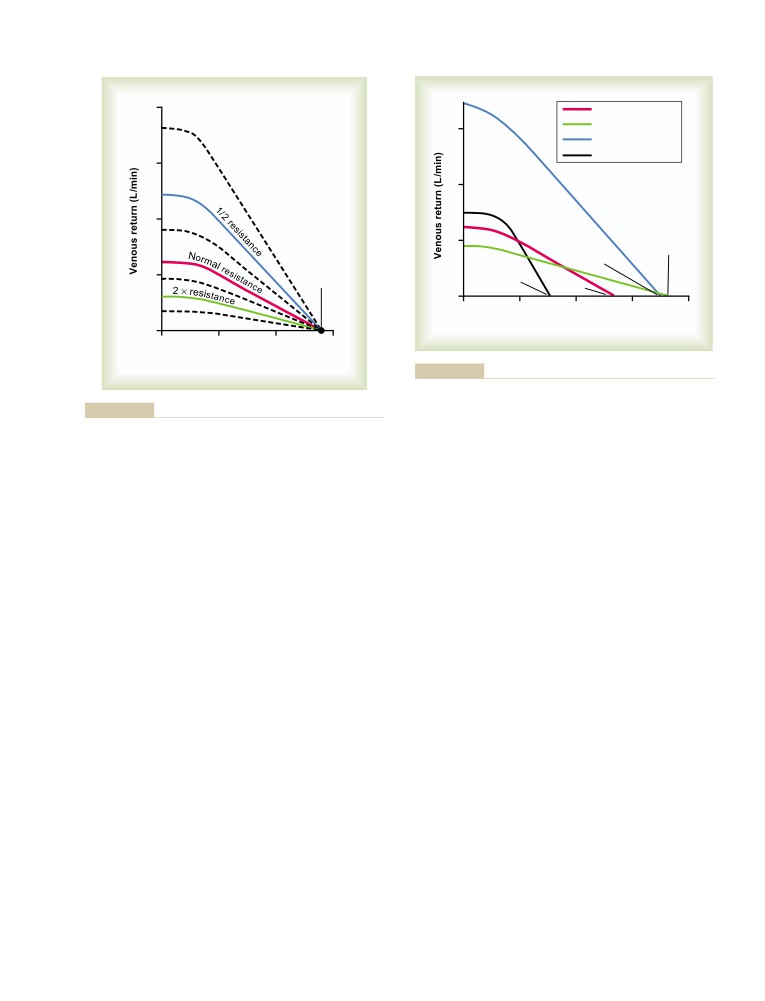
Venous Return Curves
238
of Arterial Pressure
213
Analysis of Cardiac Output and Right Atrial
Role of the Skeletal Nerves and Skeletal
Pressure, Using Simultaneous Cardiac
Muscles in Increasing Cardiac Output
Output and Venous Return Curves
241
and Arterial Pressure
213
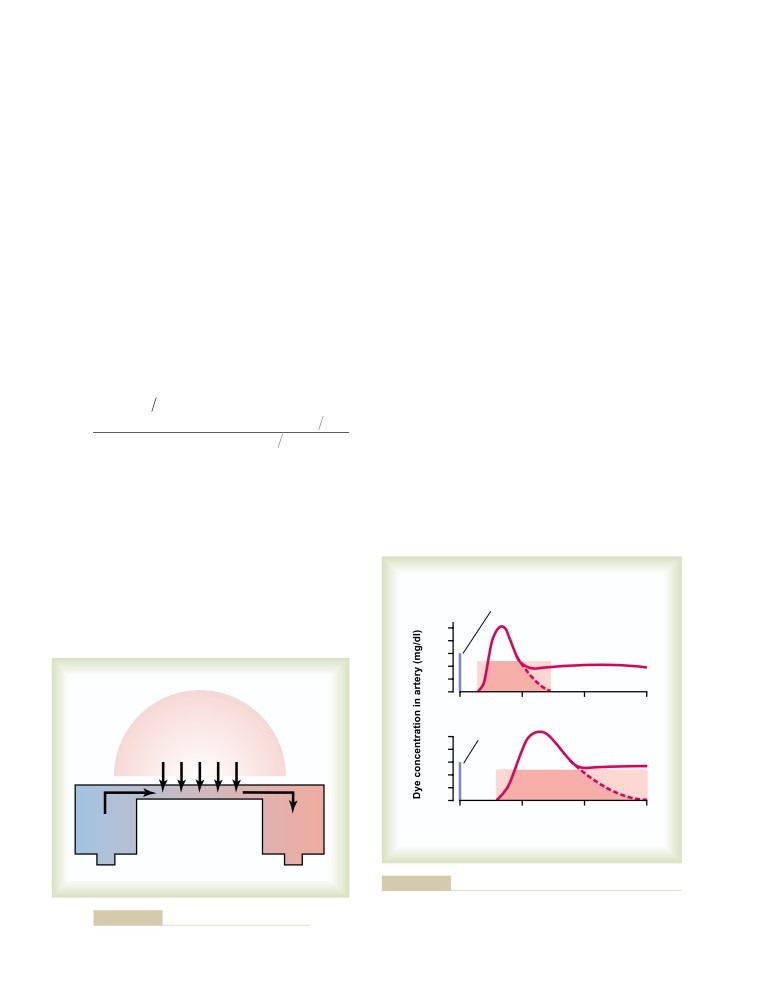
Methods for Measuring Cardiac
Respiratory Waves in the Arterial Pressure
214
Output
243
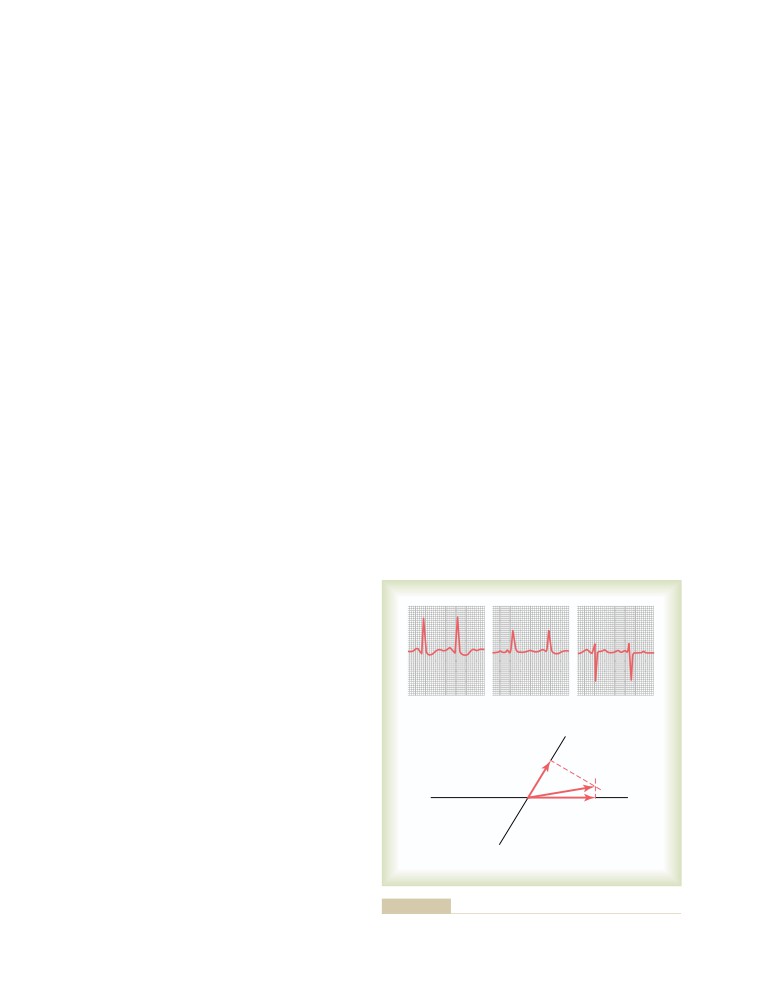
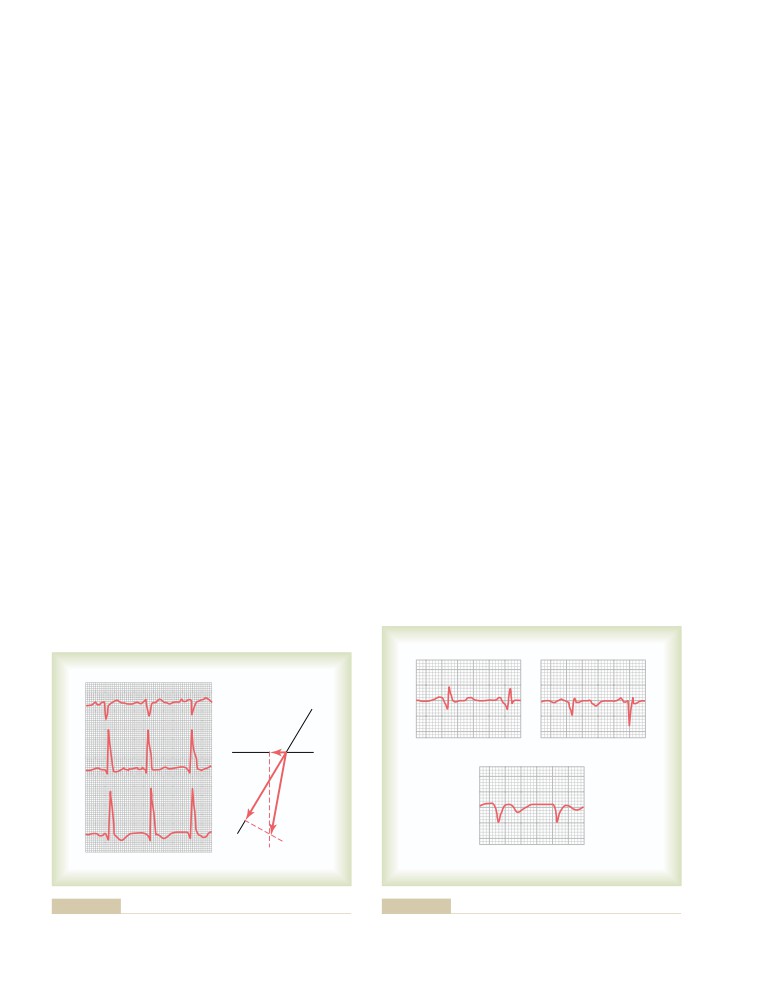
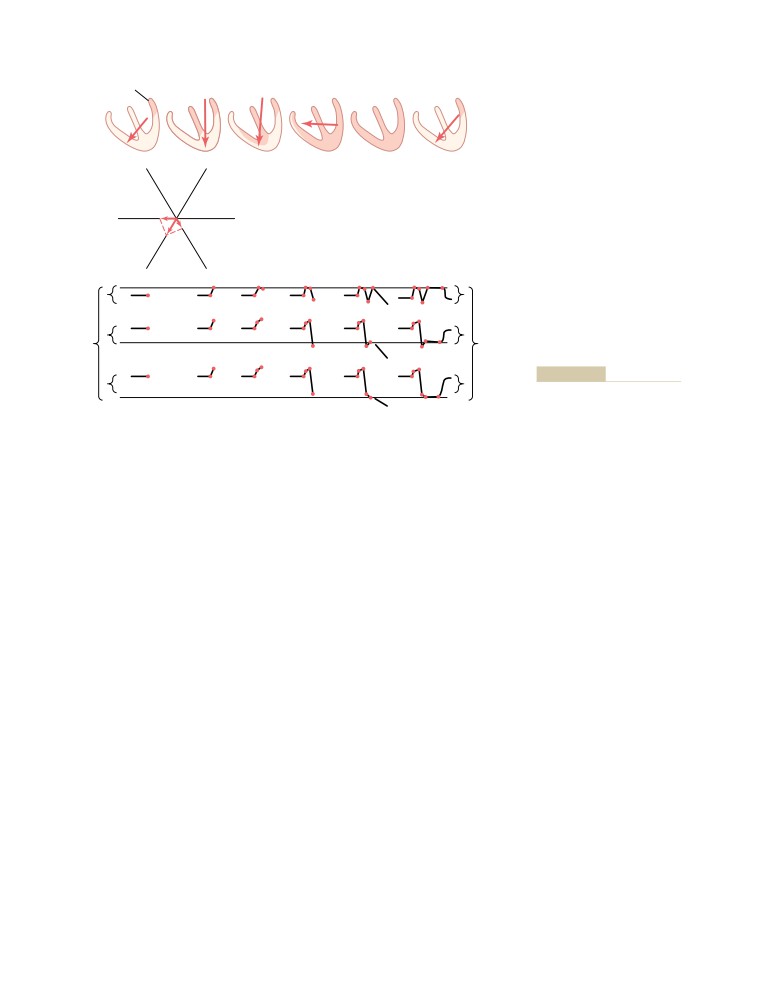
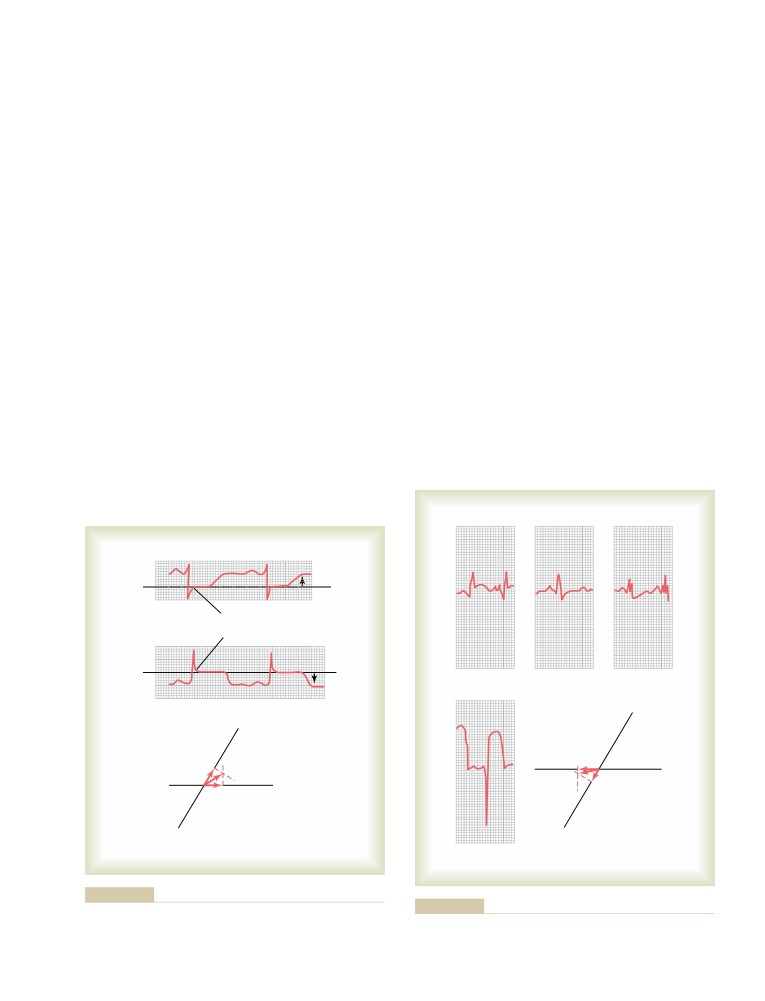
Arterial Pressure “Vasomotor” Waves—
Pulsatile Output of the Heart as Measured
Oscillation of Pressure Reflex Control
by an Electromagnetic or Ultrasonic
Systems
214
Flowmeter
243
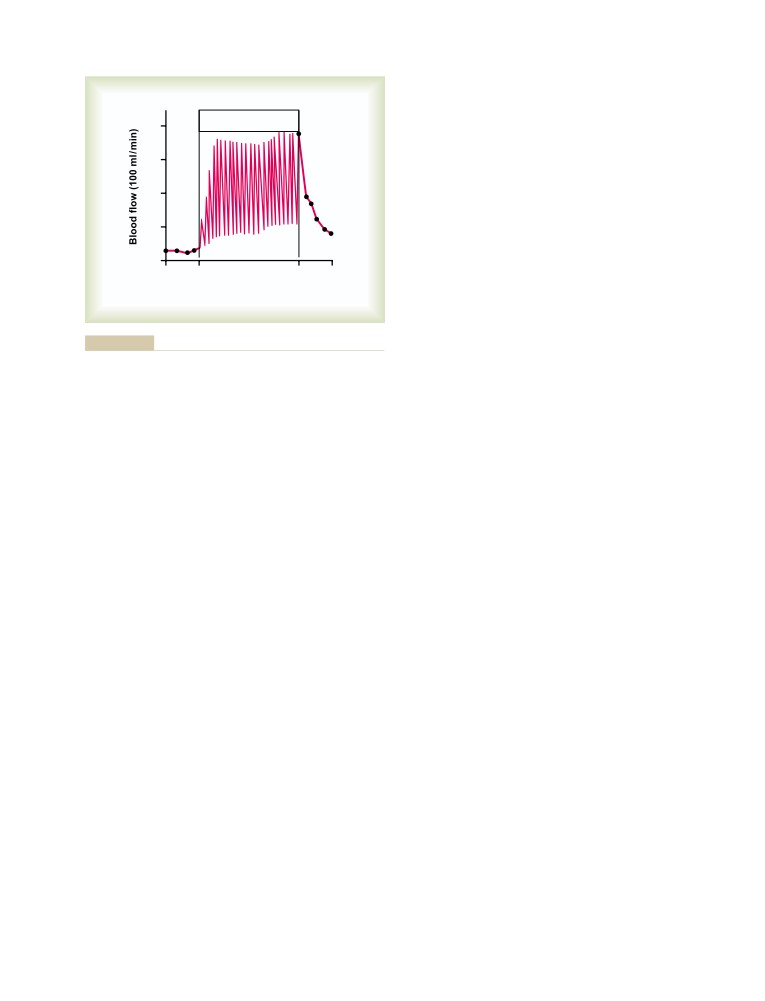
Measurement of Cardiac Output Using the
Oxygen Fick Principle
244
C H A P T E R
1
9
Indicator Dilution Method for Measuring
Cardiac Output
244
Dominant Role of the Kidney in Long-
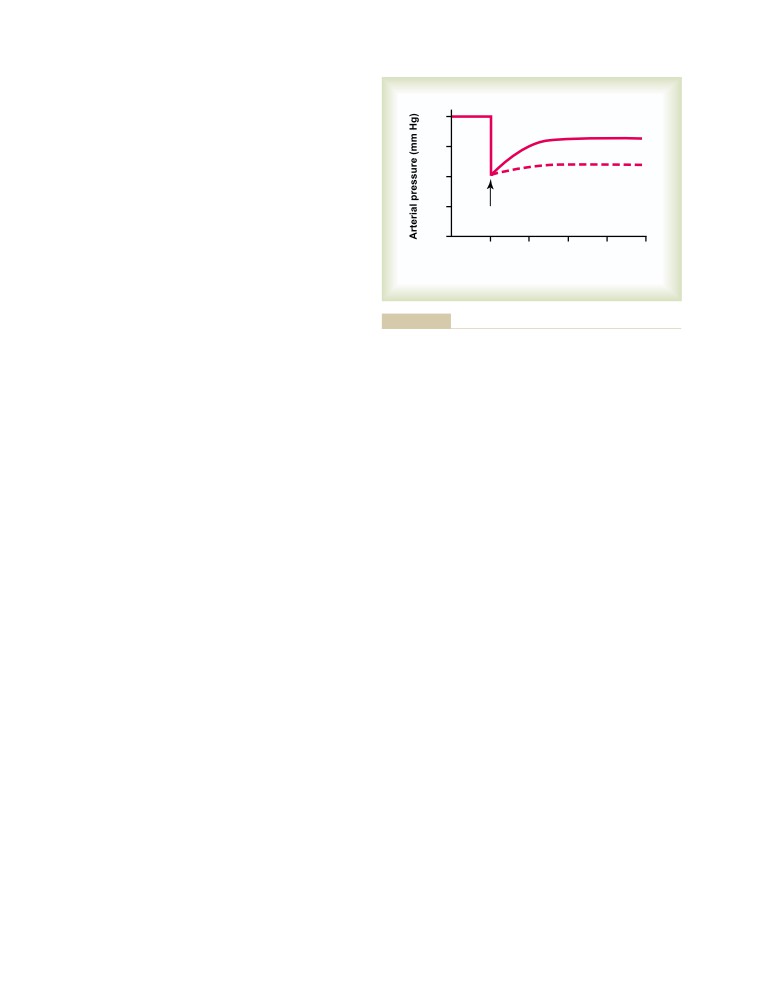
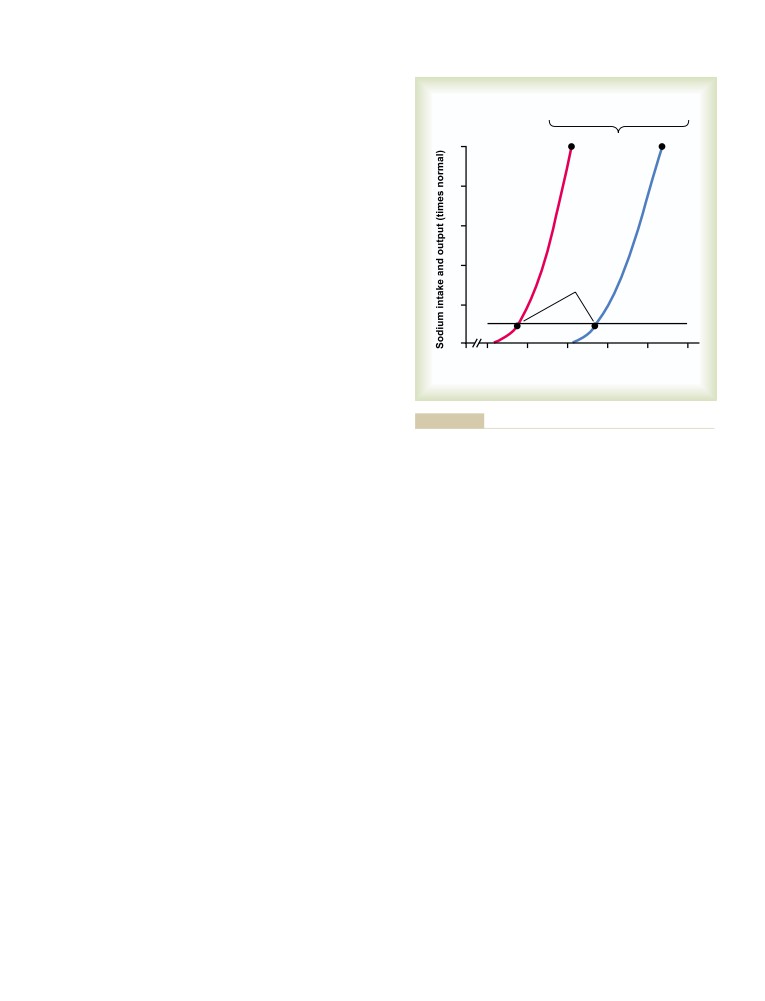
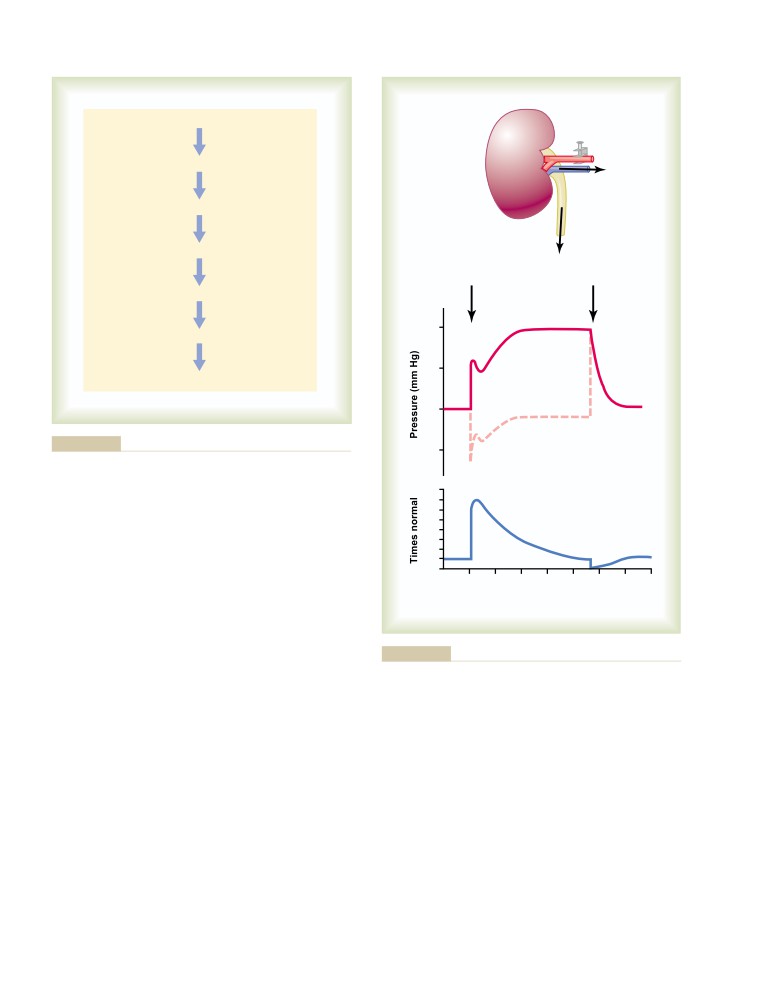
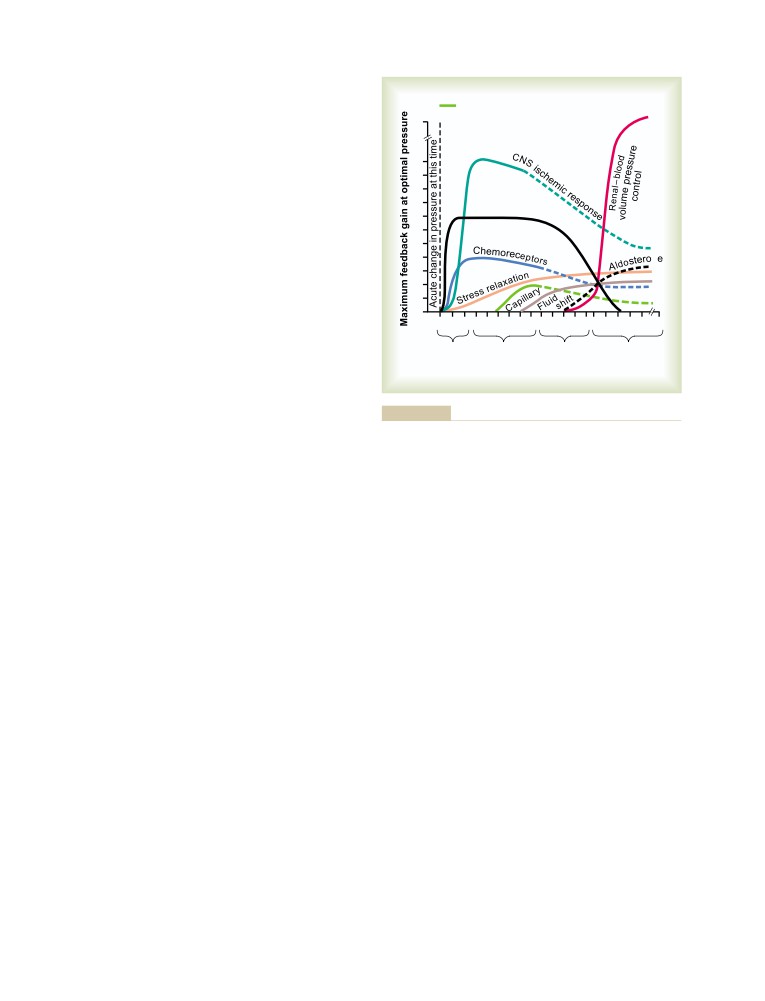
Term Regulation of Arterial Pressure
C H A P T E R
2
1
and in Hypertension: The Integrated
Muscle Blood Flow and Cardiac
System for Pressure Control
216
Renal-Body Fluid System for Arterial
Output During Exercise; the
Pressure Control
216
Coronary Circulation and Ischemic
Quantitation of Pressure Diuresis as a Basis
Heart Disease
246
for Arterial Pressure Control
217
Blood Flow in Skeletal Muscle
Chronic Hypertension (High Blood Pressure)
and Blood Flow Regulation
Is Caused by Impaired Renal Fluid
During Exercise
246
Excretion
220
Rate of Blood Flow Through the Muscles
246
The Renin-Angiotensin System:
Control of Blood Flow Through the Skeletal
Its Role in Pressure Control and in
Muscles
247
Hypertension
223
Total Body Circulatory Readjustments
Components of the Renin-Angiotensin
During Exercise
247
System
223
Coronary Circulation
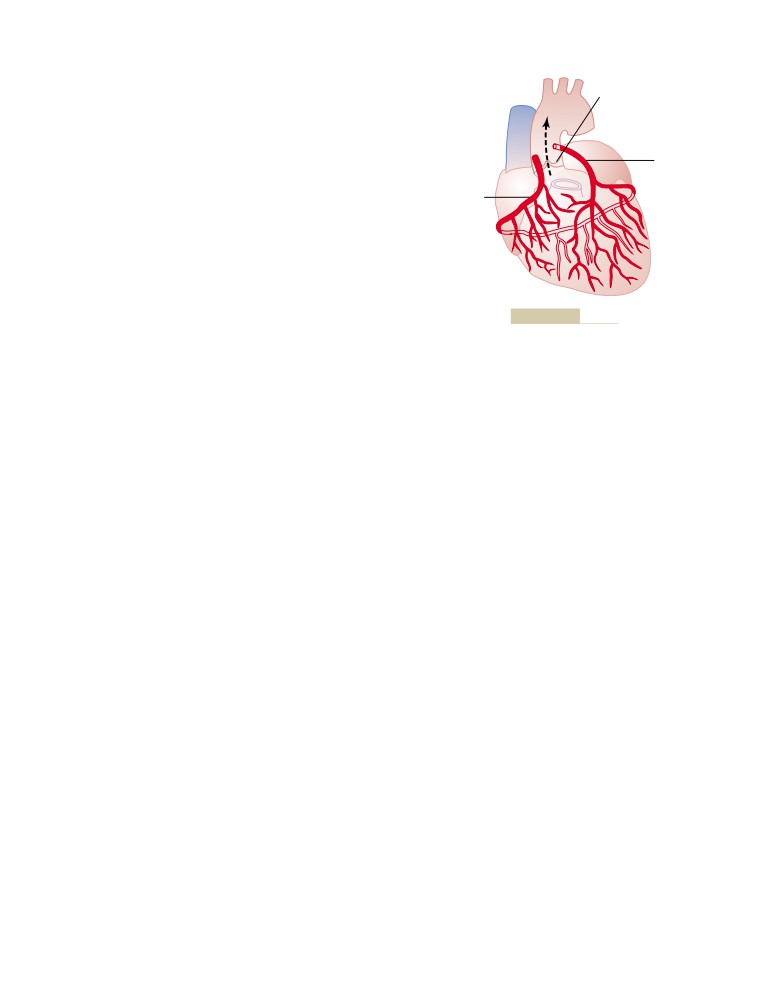
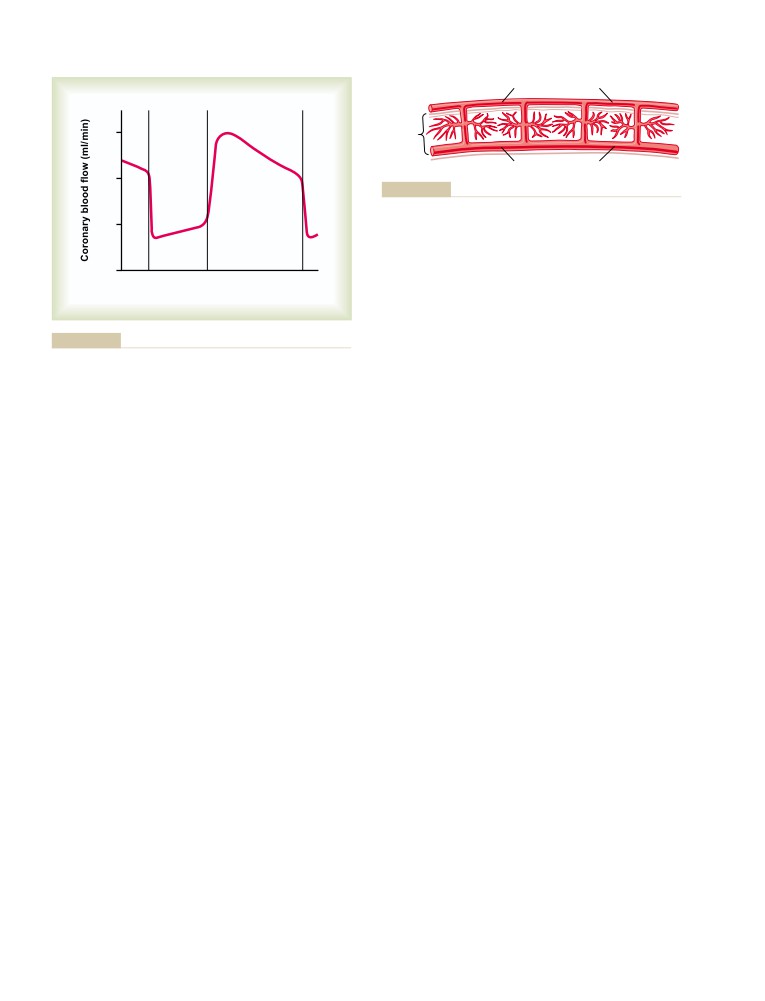
249
Types of Hypertension in Which Angiotensin
Physiologic Anatomy of the Coronary Blood
Is Involved: Hypertension Caused by a
Supply
249
Renin-Secreting Tumor or by Infusion
Normal Coronary Blood Flow
249
of Angiotensin II
226
Control of Coronary Blood Flow
250
Other Types of Hypertension Caused by
Special Features of Cardiac Muscle
Combinations of Volume Loading and
Metabolism
251
Vasoconstriction
227
Ischemic Heart Disease
252
“Primary (Essential) Hypertension”
228
Causes of Death After Acute Coronary
Summary of the Integrated,
Occlusion
253
Multifaceted System for Arterial
Stages of Recovery from Acute
Pressure Regulation
230
Myocardial Infarction
254
Function of the Heart After Recovery
from Myocardial Infarction
255
C H A P T E R
2
0
Pain in Coronary Heart Disease
255
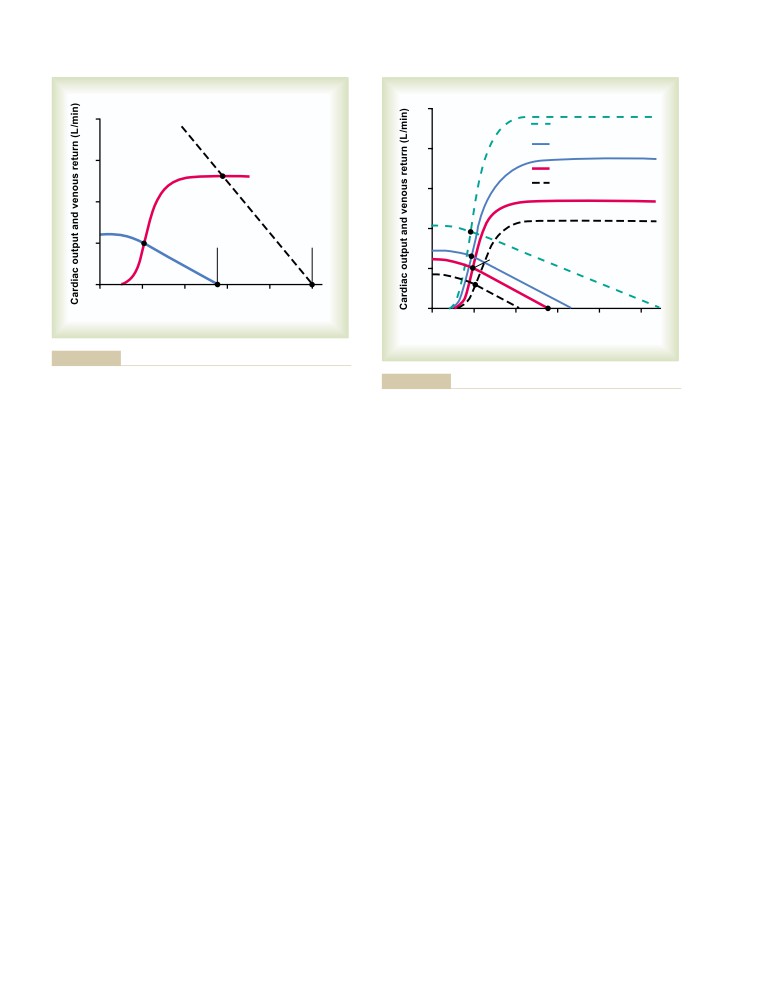
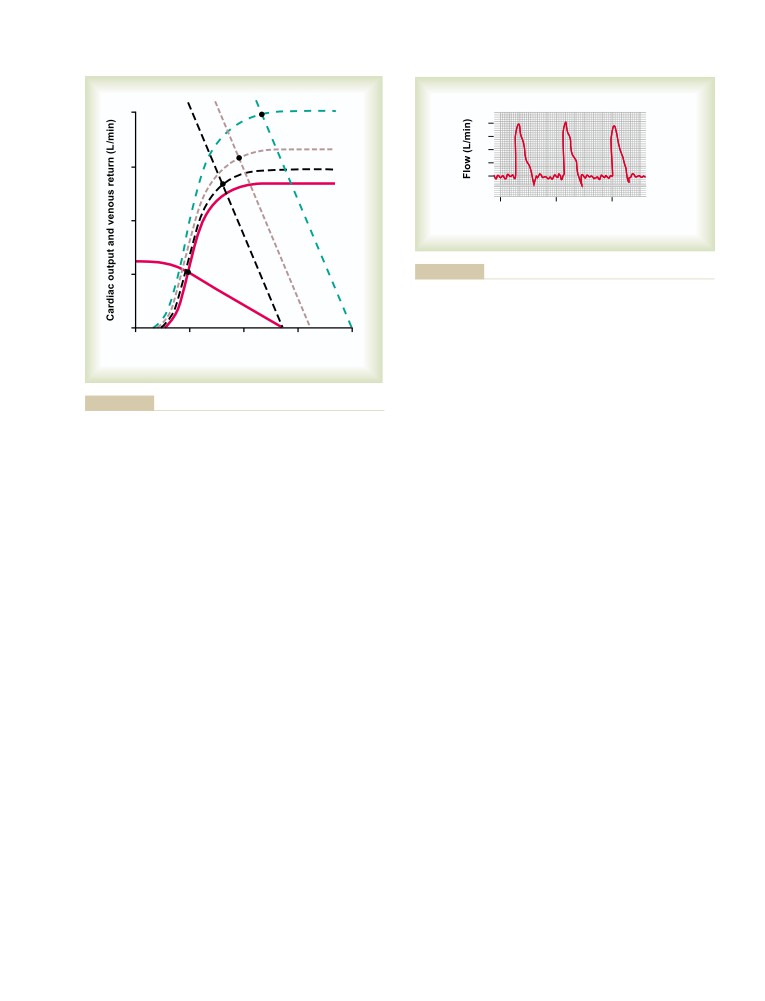
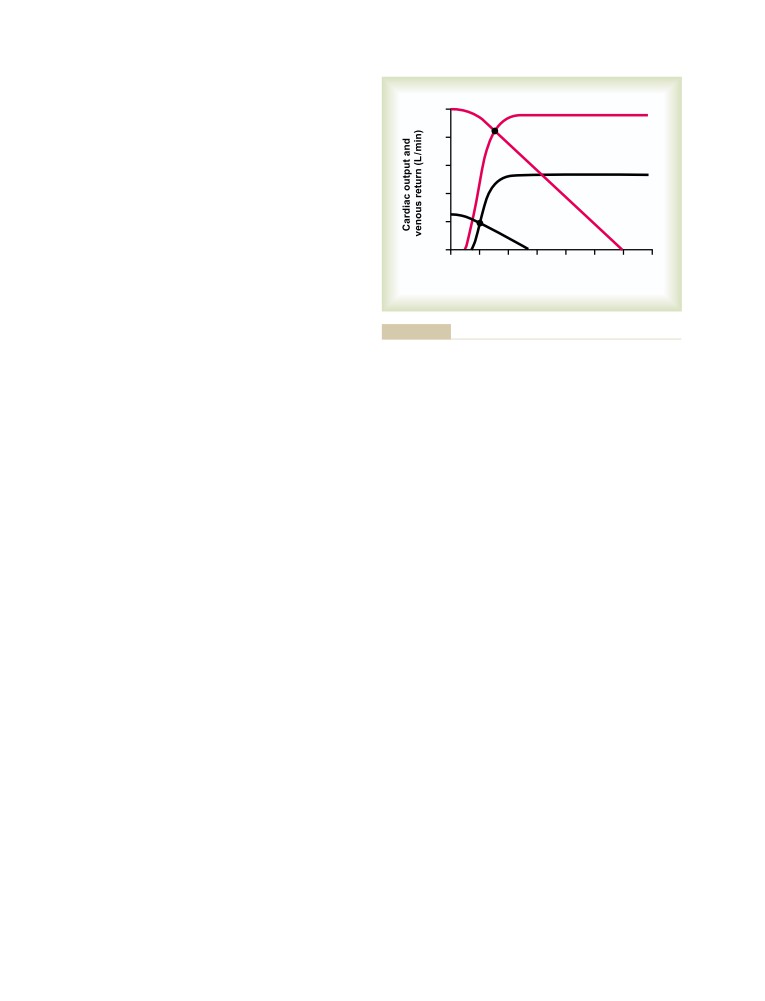
Cardiac Output, Venous Return,
Surgical Treatment of Coronary Disease
256
and Their Regulation
232
Normal Values for Cardiac Output at
C H A P T E R
2
2
Rest and During Activity
232
Cardiac Failure
258
Control of Cardiac Output by Venous
Return—Role of the Frank-Starling
Dynamics of the Circulation in
Mechanism of the Heart
232
Cardiac Failure
258
xviii
Table of Contents
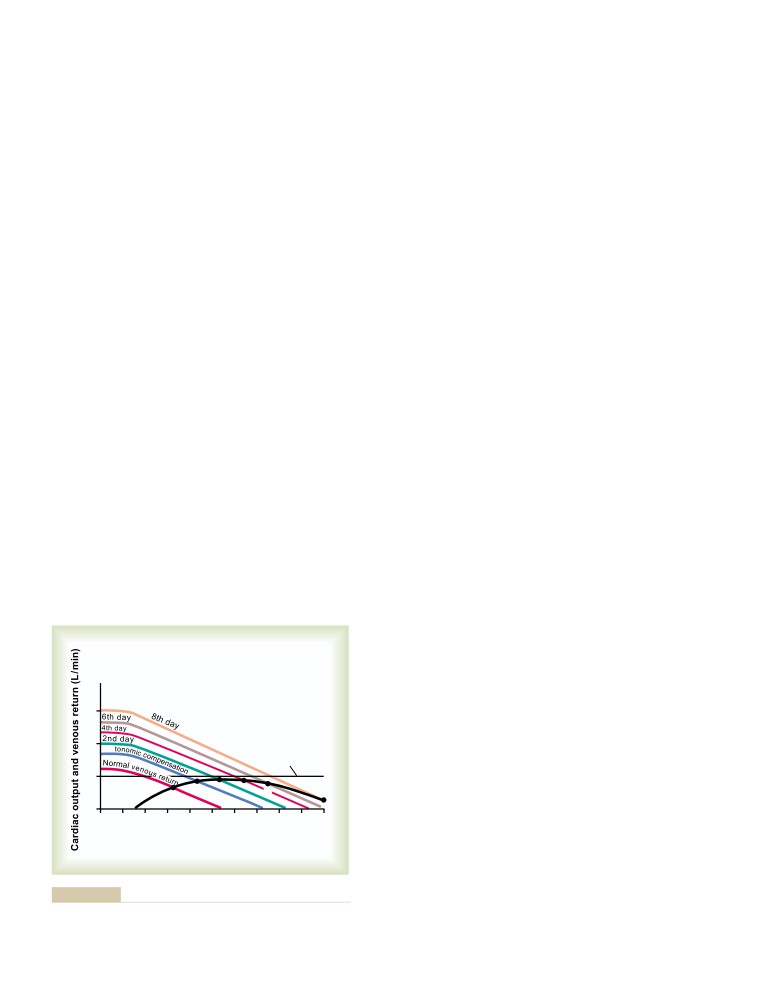
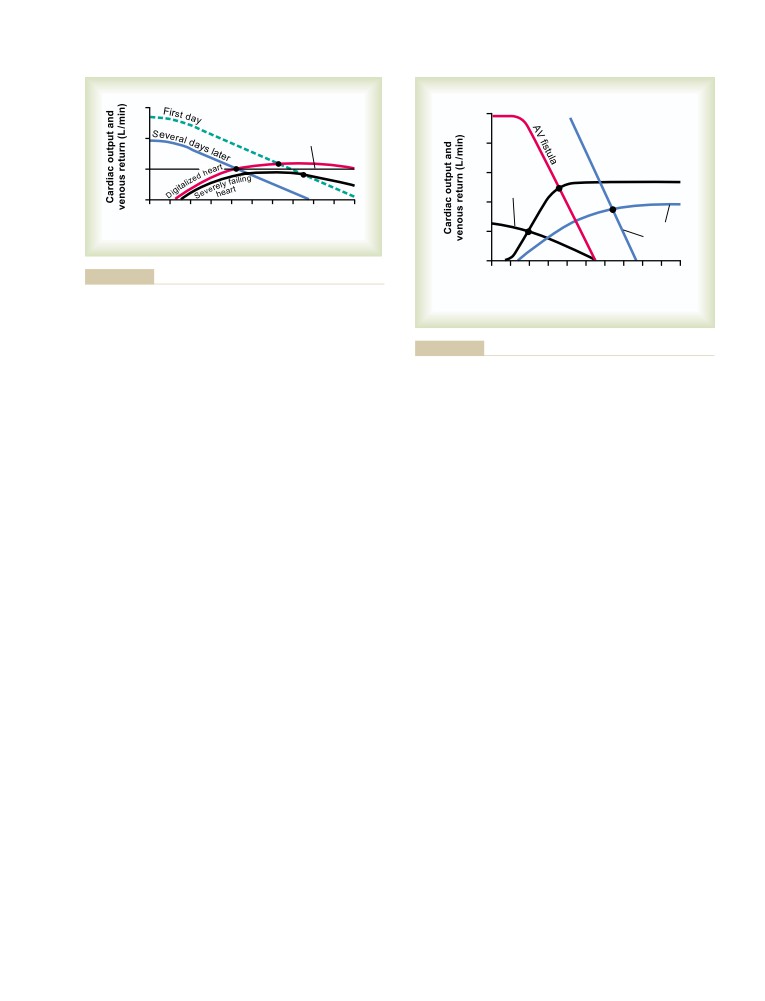
Acute Effects of Moderate Cardiac Failure
258
Neurogenic Shock—Increased Vascular
Chronic Stage of Failure—Fluid Retention
Capacity
285
Helps to Compensate Cardiac Output
259
Anaphylactic Shock and Histamine
Summary of the Changes That Occur After
Shock
285
Acute Cardiac Failure—“Compensated
Septic Shock
286
Heart Failure”
260
Physiology of Treatment in Shock
286
Dynamics of Severe Cardiac Failure—
Replacement Therapy
286
Decompensated Heart Failure
260
Treatment of Shock with Sympathomimetic
Unilateral Left Heart Failure
262
Drugs—Sometimes Useful, Sometimes
Low-Output Cardiac Failure—
Not
287
Cardiogenic Shock
262
Other Therapy
287
Edema in Patients with Cardiac Failure
263
Circulatory Arrest
287
Cardiac Reserve
264
Effect of Circulatory Arrest on the Brain
287
Quantitative Graphical Method for Analysis
of Cardiac Failure
265
U N I T V
C H A P T E R
2
3
The Body Fluids and Kidneys
Heart Valves and Heart Sounds;
Dynamics of Valvular and Congenital
C H A P T E R
2
5
Heart Defects
269
The Body Fluid Compartments:
Heart Sounds
269
Extracellular and Intracellular Fluids;
Normal Heart Sounds
269
Interstitial Fluid and Edema
291
Valvular Lesions
271
Fluid Intake and Output Are Balanced
Abnormal Circulatory Dynamics in
During Steady-State Conditions
291
Valvular Heart Disease
272
Daily Intake of Water
291
Dynamics of the Circulation in Aortic
Daily Loss of Body Water
291
Stenosis and Aortic Regurgitation
272
Body Fluid Compartments
292
Dynamics of Mitral Stenosis and Mitral
Intracellular Fluid Compartment
293
Regurgitation
273
Extracellular Fluid Compartment
293
Circulatory Dynamics During Exercise in
Blood Volume
293
Patients with Valvular Lesions
273
Constituents of Extracellular and
Abnormal Circulatory Dynamics in
Intracellular Fluids
293
Congenital Heart Defects
274
Ionic Composition of Plasma and
Patent Ductus Arteriosus—A Left-to-Right
Interstitial Fluid Is Similar
293
Shunt
274
Important Constituents of the Intracellular
Tetralogy of Fallot—A Right-to-Left Shunt
274
Fluid
295
Causes of Congenital Anomalies
276
Measurement of Fluid Volumes in the
Use of Extracorporeal Circulation
Different Body Fluid Compartments—
During Cardiac Surgery
276
The Indicator-Dilution Principle
295
Hypertrophy of the Heart in Valvular
Determination of Volumes of Specific
and Congenital Heart Disease
276
Body Fluid Compartments
295
Regulation of Fluid Exchange and
C H A P T E R
2
4
Osmotic Equilibrium Between
Intracellular and Extracellular Fluid
296
Circulatory Shock and Physiology of
Basic Principles of Osmosis and
Its Treatment
278
Osmotic Pressure
296
Physiologic Causes of Shock
278
Osmotic Equilibrium Is Maintained
Circulatory Shock Caused by Decreased
Between Intracellular and
Cardiac Output
278
Extracellular Fluids
298
Circulatory Shock That Occurs Without
Volume and Osmolality of Extracellular
Diminished Cardiac Output
278
and Intracellular Fluids in Abnormal
What Happens to the Arterial Pressure in
States
299
Circulatory Shock?
279
Effect of Adding Saline Solution to the
Tissue Deterioration Is the End Result of
Extracellular Fluid
299
Circulatory Shock, Whatever the Cause
279
Glucose and Other Solutions
Stages of Shock
279
Administered for Nutritive Purposes
301
Shock Caused by Hypovolemia—
Clinical Abnormalities of Fluid Volume
Hemorrhagic Shock
279
Regulation: Hyponatremia and
Relationship of Bleeding Volume to
Hypernatremia
301
Cardiac Output and Arterial Pressure
279
Causes of Hyponatremia: Excess Water or
Progressive and Nonprogressive
Loss of Sodium
301
Hemorrhagic Shock
280
Causes of Hypernatremia: Water Loss or
Irreversible Shock
284
Excess Sodium
302
Hypovolemic Shock Caused by Plasma
Edema: Excess Fluid in the Tissues
302
Loss
284
Intracellular Edema
302
Hypovolemic Shock Caused by Trauma
285
Extracellular Edema
302
Table of Contents
xix
Summary of Causes of Extracellular Edema
303
Importance of GFR Autoregulation in
Safety Factors That Normally Prevent
Preventing Extreme Changes in Renal
Edema
304
Excretion
323
Fluids in the “Potential Spaces” of
Role of Tubuloglomerular Feedback in
the Body
305
Autoregulation of GFR
323
Myogenic Autoregulation of Renal Blood
Flow and GFR
325
C H A P T E R
2
6
Other Factors That Increase Renal Blood
Flow and GFR: High Protein Intake and
Urine Formation by the Kidneys:
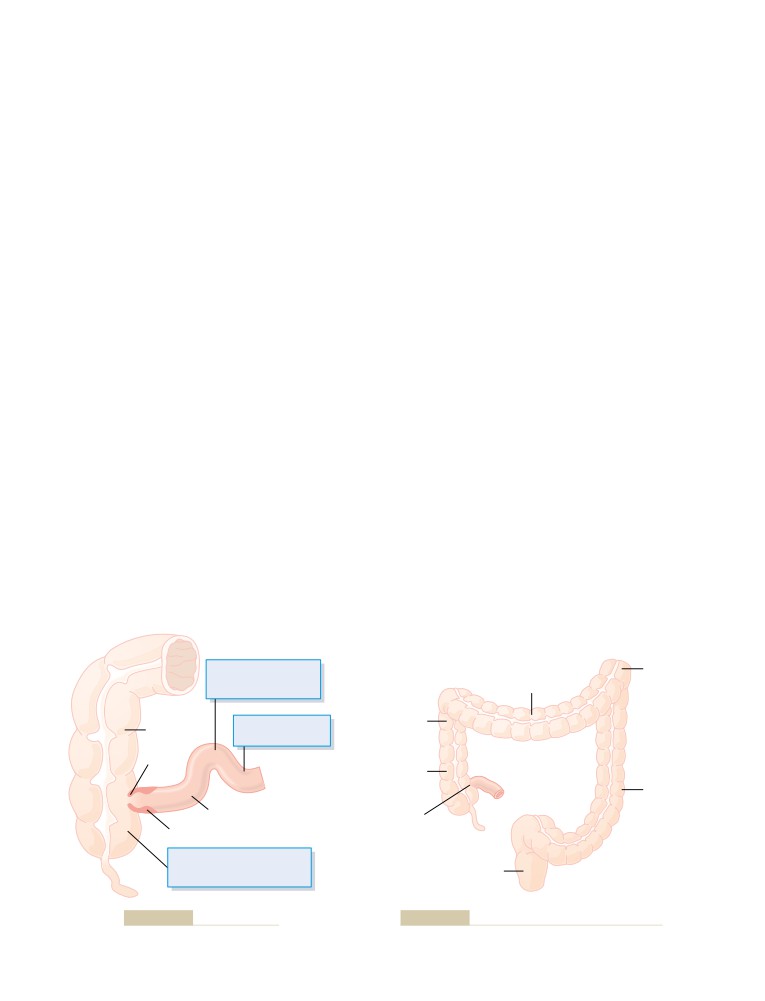
Increased Blood Glucose
325
I. Glomerular Filtration, Renal Blood
Flow, and Their Control
307
Multiple Functions of the Kidneys in
Homeostasis
307
C H A P T E R
2
7
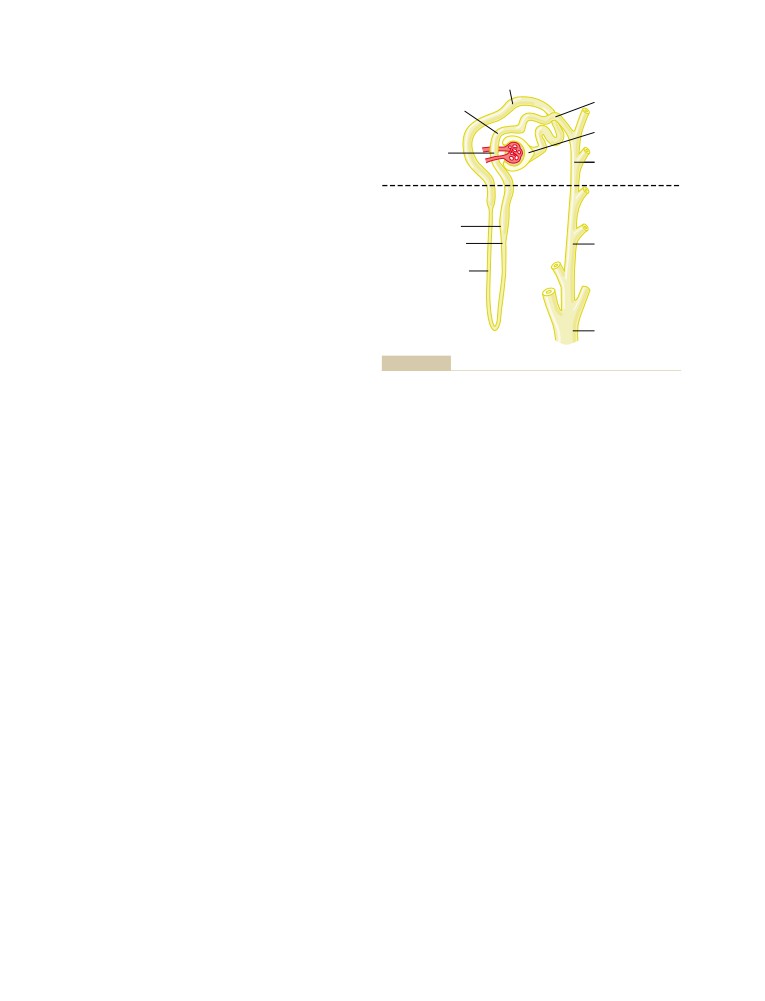
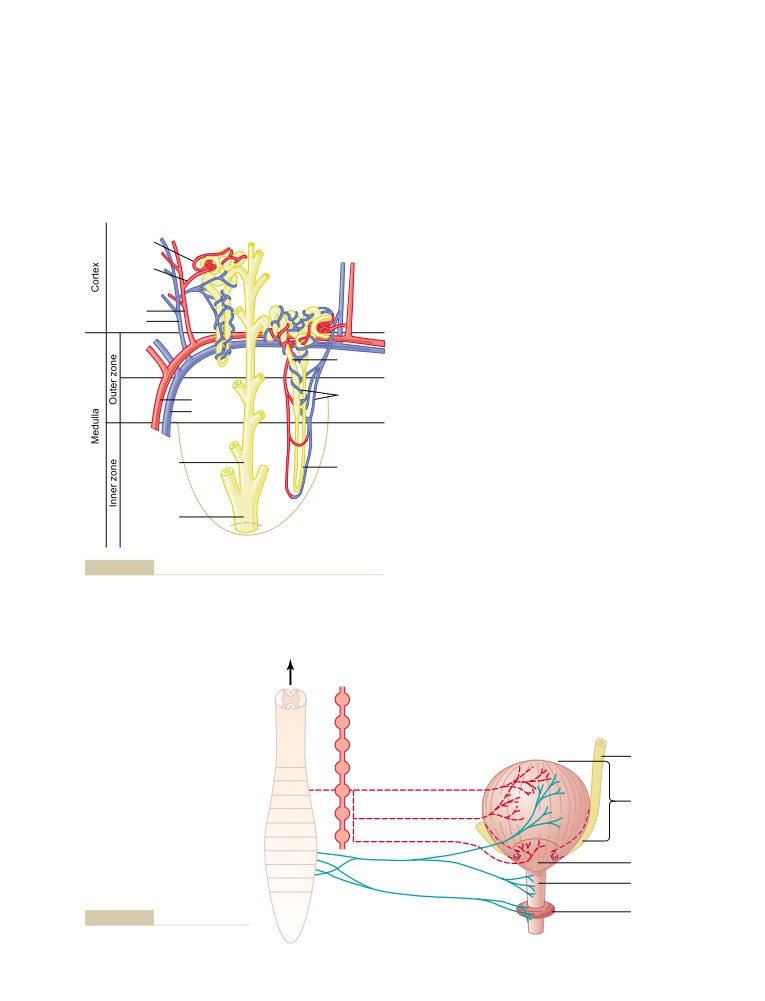
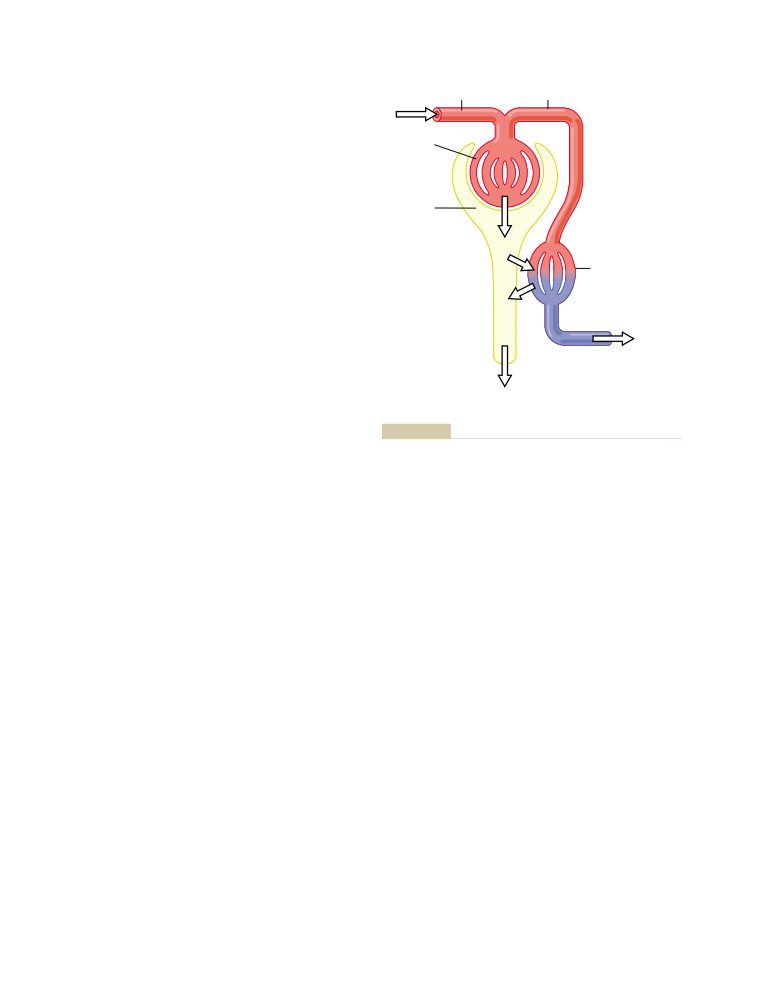
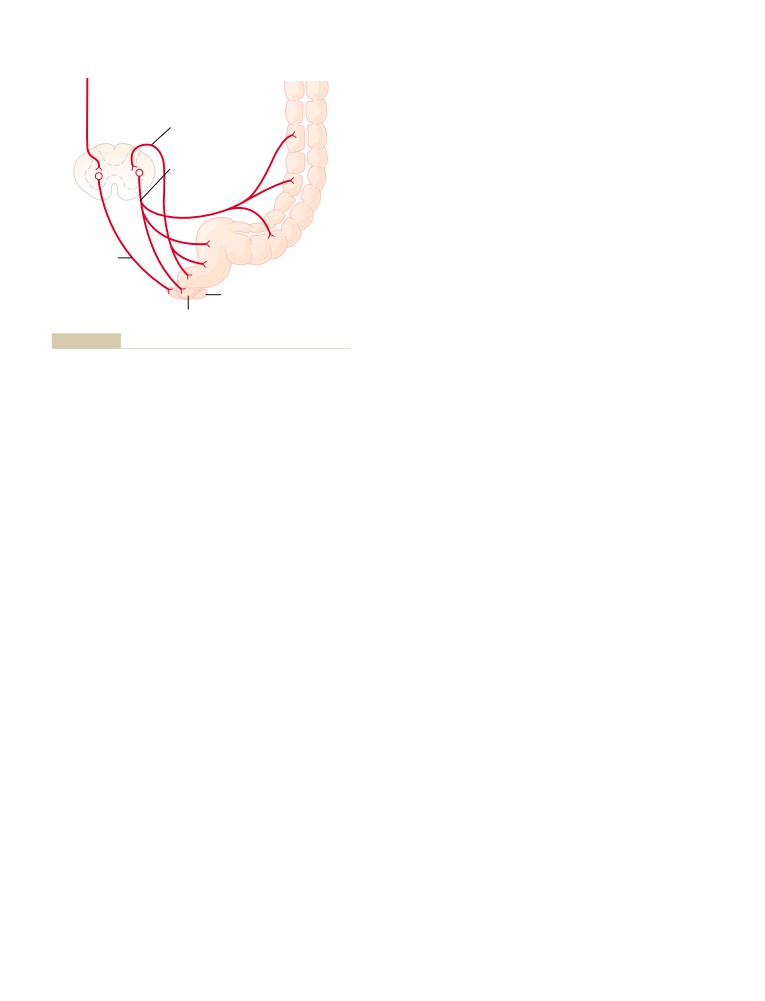
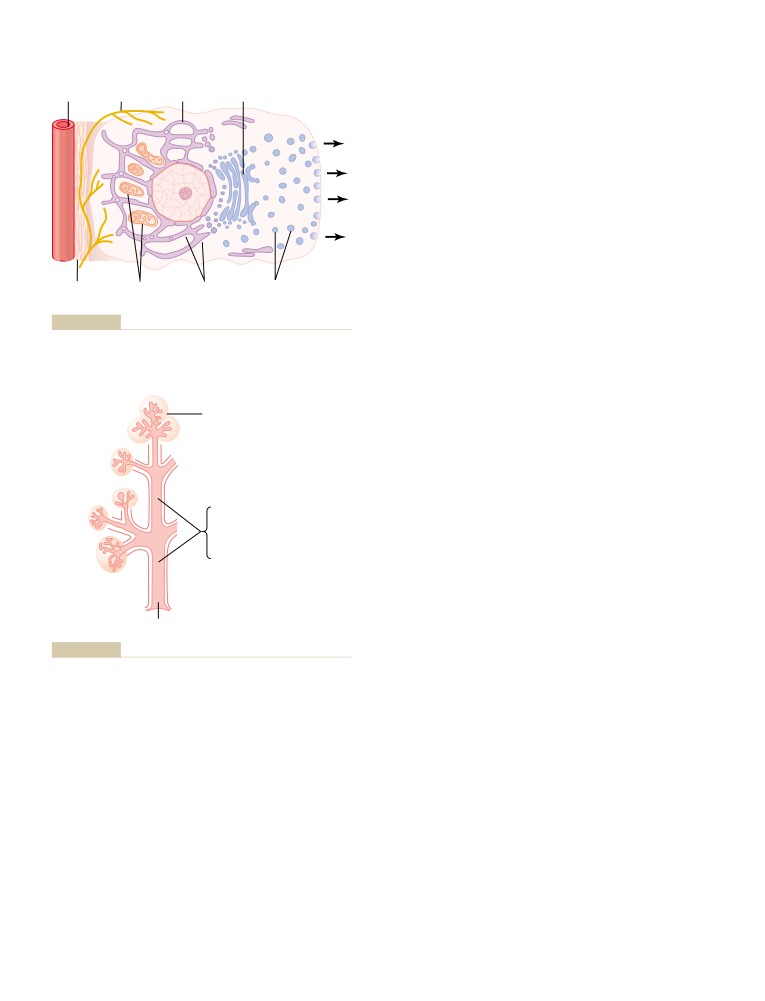
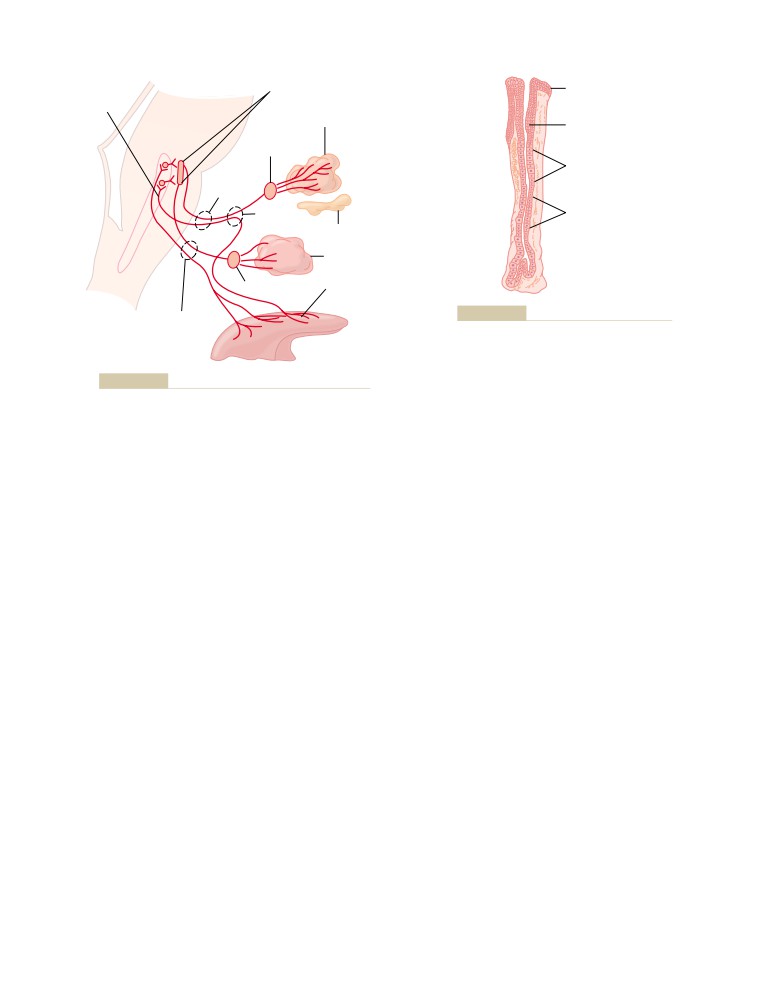
Physiologic Anatomy of the Kidneys
308
Urine Formation by the Kidneys:
General Organization of the Kidneys and
II. Tubular Processing of the
Urinary Tract
308
Renal Blood Supply
309
Glomerular Filtrate
327
The Nephron Is the Functional Unit of the
Reabsorption and Secretion by the
Kidney
310
Renal Tubules
327
Micturition
311
Tubular Reabsorption Is Selective and
Physiologic Anatomy and Nervous
Quantitatively Large
327
Connections of the Bladder
311
Tubular Reabsorption Includes
Transport of Urine from the Kidney
Passive and Active Mechanisms
328
Through the Ureters and into
Active Transport
328
the Bladder
312
Passive Water Reabsorption by Osmosis
Innervation of the Bladder
312
Is Coupled Mainly to Sodium
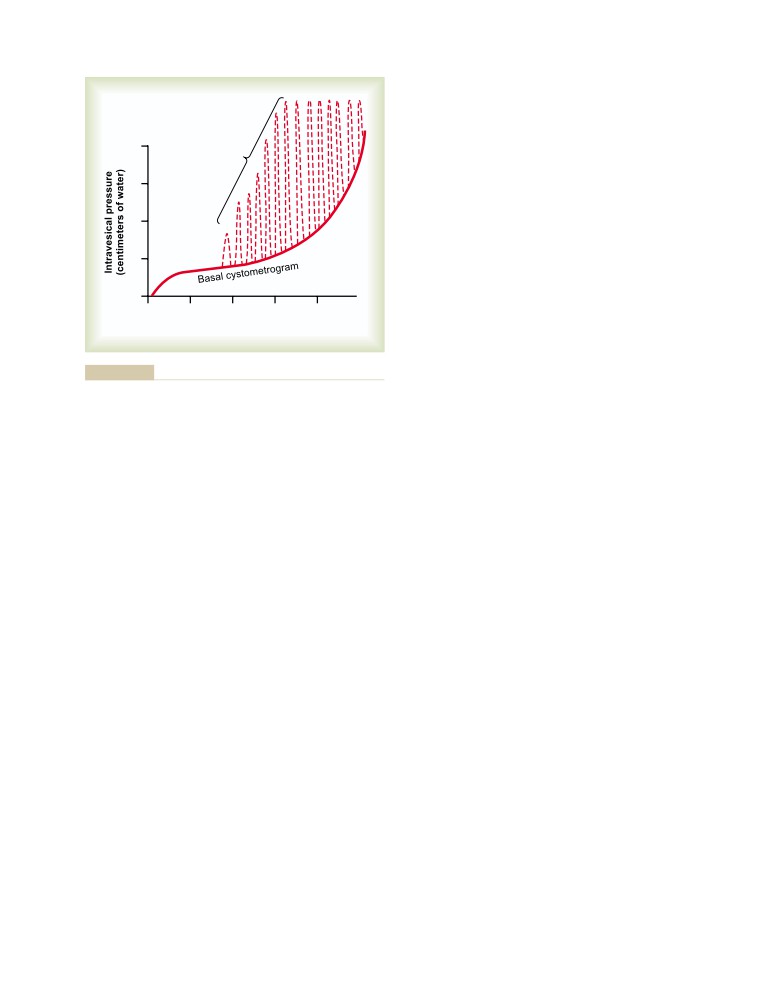
Filling of the Bladder and Bladder Wall
Reabsorption
332
Tone; the Cystometrogram
312
Reabsorption of Chloride, Urea, and Other
Micturition Reflex
313
Solutes by Passive Diffusion
332
Facilitation or Inhibition of Micturition
Reabsorption and Secretion Along
by the Brain
313
Different Parts of the Nephron
333
Abnormalities of Micturition
313
Proximal Tubular Reabsorption
333
Urine Formation Results from
Solute and Water Transport in the Loop
Glomerular Filtration, Tubular
of Henle
334
Reabsorption, and Tubular Secretion
314
Distal Tubule
336
Filtration, Reabsorption, and Secretion of
Late Distal Tubule and Cortical Collecting
Different Substances
315
Tubule
336
Glomerular Filtration—The First Step in
Medullary Collecting Duct
337
Urine Formation
316
Summary of Concentrations of Different
Composition of the Glomerular Filtrate
316
Solutes in the Different Tubular
GFR Is About 20 Per Cent of the Renal
Segments
338
Plasma Flow
316
Regulation of Tubular Reabsorption
339
Glomerular Capillary Membrane
316
Glomerulotubular Balance—The Ability
Determinants of the GFR
317
of the Tubules to Increase Reabsorption
Increased Glomerular Capillary Filtration
Rate in Response to Increased Tubular
Coefficient Increases GFR
318
Load
339
Increased Bowman’s Capsule Hydrostatic
Peritubular Capillary and Renal Interstitial
Pressure Decreases GFR
318
Fluid Physical Forces
339
Increased Glomerular Capillary Colloid
Effect of Arterial Pressure on Urine
Osmotic Pressure Decreases GFR
318
Output—The Pressure-Natriuresis and
Increased Glomerular Capillary Hydrostatic
Pressure-Diuresis Mechanisms
341
Pressure Increases GFR
319
Hormonal Control of Tubular Reabsorption
342
Renal Blood Flow
320
Sympathetic Nervous System Activation
Renal Blood Flow and Oxygen
Increases Sodium Reabsorption
343
Consumption
320
Use of Clearance Methods to Quantify
Determinants of Renal Blood Flow
320
Kidney Function
343
Blood Flow in the Vasa Recta of the Renal
Inulin Clearance Can Be Used to Estimate
Medulla Is Very Low Compared with Flow
GFR
344
in the Renal Cortex
321
Creatine Clearance and Plasma Creatinine
Physiologic Control of Glomerular
Clearance Can Be Used to Estimate
Filtration and Renal Blood Flow
321
GFR
344
Sympathetic Nervous System Activation
PAH Clearance Can Be Used to Estimate
Decreases GFR
321
Renal Plasma Flow
345
Hormonal and Autacoid Control of Renal
Filtration Fraction Is Calculated from GFR
Circulation
322
Divided by Renal Plasma Flow
346
Autoregulation of GFR and Renal
Calculation of Tubular Reabsorption or
Blood Flow
323
Secretion from Renal Clearance
346
xx
Table of Contents
C H A P T E R
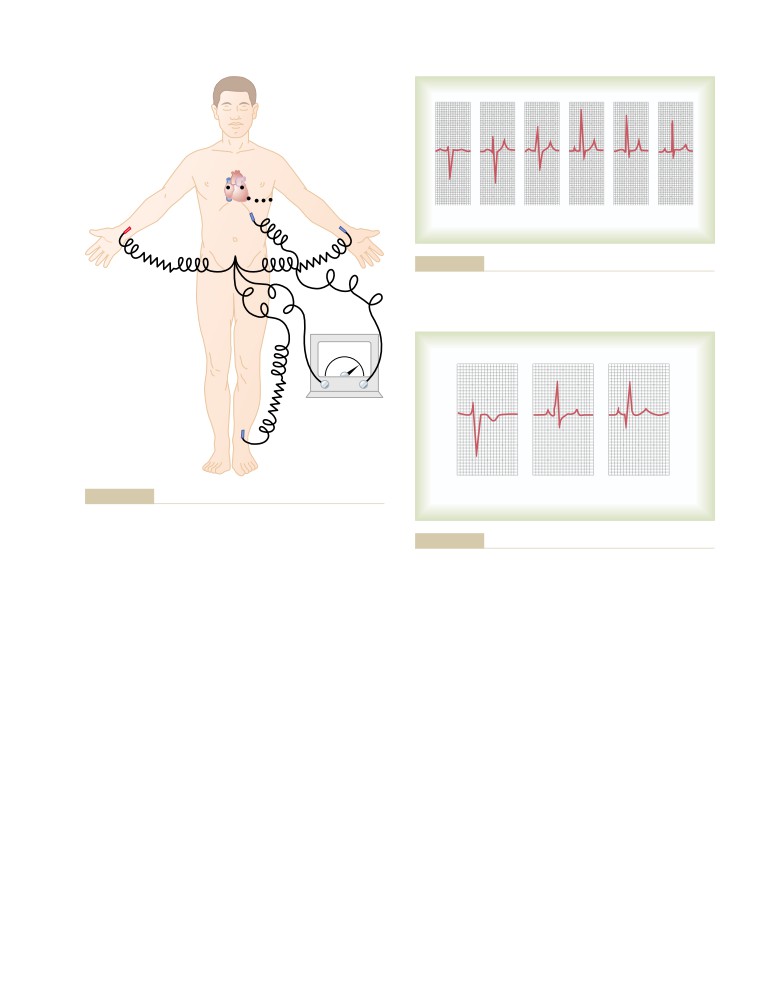
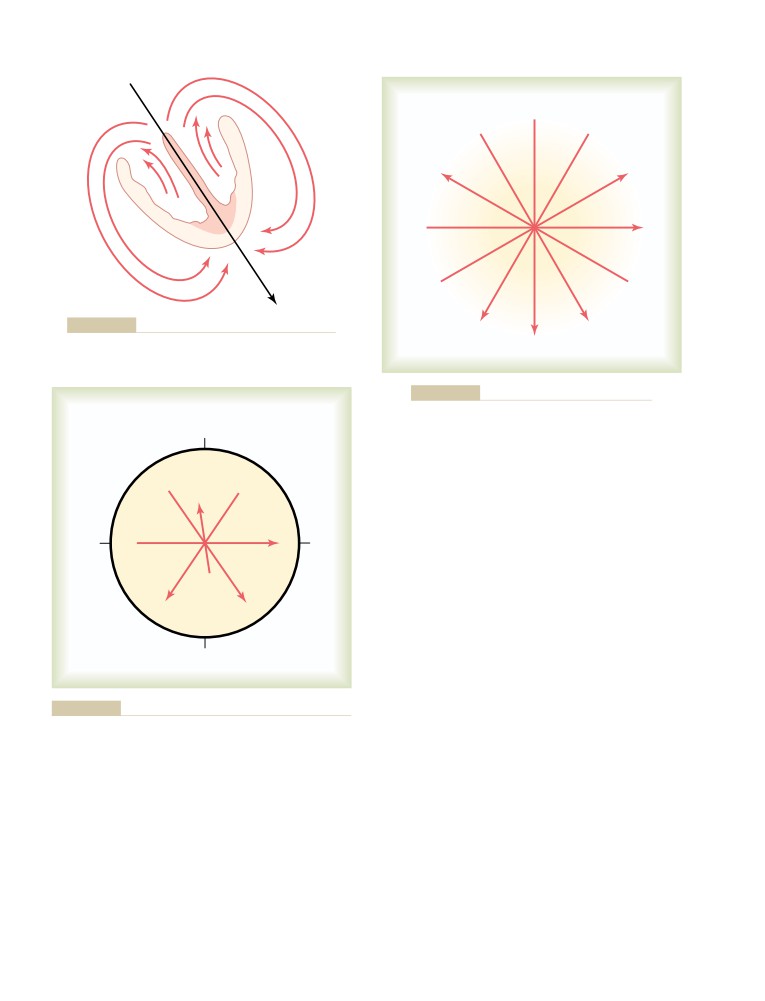
2
8
C H A P T E R
2
9
Regulation of Extracellular Fluid
Renal Regulation of Potassium,
Osmolarity and Sodium
Calcium, Phosphate, and Magnesium;
Concentration
348
Integration of Renal Mechanisms for
The Kidneys Excrete Excess Water
Control of Blood Volume and
by Forming a Dilute Urine
348
Extracellular Fluid Volume
365
Antidiuretic Hormone Controls Urine
Regulation of Potassium Excretion
Concentration
348
and Potassium Concentration in
Renal Mechanisms for Excreting a
Extracellular Fluid
365
Dilute Urine
349
Regulation of Internal Potassium
The Kidneys Conserve Water by
Distribution
366
Excreting a Concentrated Urine
350
Overview of Renal Potassium Excretion
367
Obligatory Urine Volume
350
Potassium Secretion by Principal Cells of
Requirements for Excreting a Concentrated
Late Distal and Cortical Collecting
Urine—High ADH Levels and Hyperosmotic
Tubules
367
Renal Medulla
350
Summary of Factors That Regulate
Countercurrent Mechanism Produces a
Potassium Secretion: Plasma Potassium
Hyperosmotic Renal Medullary Interstitium
351
Concentration, Aldosterone, Tubular Flow
Role of Distal Tubule and Collecting Ducts in
Rate, and Hydrogen Ion Concentration
368
Excreting a Concentrated Urine
352
Control of Renal Calcium Excretion
Urea Contributes to Hyperosmotic Renal
and Extracellular Calcium Ion
Medullary Interstitium and to a
Concentration
371
Concentrated Urine
353
Control of Calcium Excretion by the
Countercurrent Exchange in the Vasa Recta
Kidneys
372
Preserves Hyperosmolarity of the
Regulation of Renal Phosphate Excretion
372
Renal Medulla
354
Control of Renal Magnesium Excretion
Summary of Urine Concentrating Mechanism
and Extracellular Magnesium Ion
and Changes in Osmolarity in Different
Concentration
373
Segments of the Tubules
355
Integration of Renal Mechanisms for
Quantifying Renal Urine Concentration
Control of Extracellular Fluid
373
and Dilution: “Free Water” and Osmolar
Sodium Excretion Is Precisely Matched to
Clearances
357
Intake Under Steady-State Conditions
373
Disorders of Urinary Concentrating
Sodium Excretion Is Controlled by Altering
Ability
357
Glomerular Filtration or Tubular Sodium
Control of Extracellular Fluid Osmolarity
Reabsorption Rates
374
and Sodium Concentration
358
Importance of Pressure Natriuresis and
Estimating Plasma Osmolarity from Plasma
Pressure Diuresis in Maintaining Body
Sodium Concentration
358
Sodium and Fluid Balance
374
Osmoreceptor-ADH Feedback System
358
Pressure Natriuresis and Diuresis Are Key
ADH Synthesis in Supraoptic and
Components of a Renal-Body Fluid
Paraventricular Nuclei of the
Feedback for Regulating Body Fluid
Hypothalamus and ADH Release from
Volumes and Arterial Pressure
375
the Posterior Pituitary
359
Precision of Blood Volume and Extracellular
Cardiovascular Reflex Stimulation of ADH
Fluid Volume Regulation
376
Release by Decreased Arterial Pressure
Distribution of Extracellular Fluid
and/or Decreased Blood Volume
360
Between the Interstitial Spaces and
Quantitative Importance of Cardiovascular
Vascular System
376
Reflexes and Osmolarity in Stimulating
Nervous and Hormonal Factors Increase
ADH Secretion
360
the Effectiveness of Renal-Body Fluid
Other Stimuli for ADH Secretion
360
Feedback Control
377
Role of Thirst in Controlling Extracellular
Sympathetic Nervous System Control of
Fluid Osmolarity and Sodium
Renal Excretion: Arterial Baroreceptor and
Concentration
361
Low-Pressure Stretch Receptor Reflexes
377
Central Nervous System Centers for Thirst
361
Role of Angiotensin II In Controlling Renal
Stimuli for Thirst
361
Excretion
377
Threshold for Osmolar Stimulus of Drinking
362
Role of Aldosterone in Controlling Renal
Integrated Responses of Osmoreceptor-ADH
Excretion
378
and Thirst Mechanisms in Controlling
Role of ADH in Controlling Renal Water
Extracellular Fluid Osmolarity and Sodium
Excretion
379
Concentration
362
Role of Atrial Natriuretic Peptide in
Role of Angiotensin II and Aldosterone
Controlling Renal Excretion
378
in Controlling Extracellular Fluid
Integrated Responses to Changes in
Osmolarity and Sodium Concentration
362
Sodium Intake
380
Salt-Appetite Mechanism for
Conditions That Cause Large Increases
Controlling Extracellular Fluid
in Blood Volume and Extracellular
Sodium Concentration and Volume
363
Fluid Volume
380
Table of Contents
xxi
Increased Blood Volume and Extracellular
Renal Correction of Acidosis—Increased
Fluid Volume Caused by Heart Diseases
380
Excretion of Hydrogen Ions and
Increased Blood Volume Caused by
Addition of Bicarbonate Ions to the
Increased Capacity of Circulation
380
Extracellular Fluid
396
Conditions That Cause Large Increases
Acidosis Decreases the Ratio of HCO3-/H+ in
in Extracellular Fluid Volume but with
Renal Tubular Fluid
396
Normal Blood Volume
381
Renal Correction of Alkalosis—Decreased
Nephrotic Syndrome—Loss of Plasma
Tubular Secretion of Hydrogen Ions
Proteins in Urine and Sodium Retention
and Increased Excretion of
by the Kidneys
381
Bicarbonate Ions
396
Liver Cirrhosis—Decreased Synthesis of
Alkalosis Increases the Ratio of HCO3-/H+
Plasma Proteins by the Liver and
in Renal Tubular Fluid
396
Sodium Retention by the Kidneys
381
Clinical Causes of Acid-Base Disorders
397
Respiratory Acidosis Is Caused by
Decreased Ventilation and Increased PCO2
397
Respiratory Alkalosis Results from Increased
C H A P T E R
3
0
Ventilation and Decreased PCO2
397
Regulation of Acid-Base Balance
383
Metabolic Acidosis Results from Decreased
Hydrogen Ion Concentration Is
Extracellular Fluid Bicarbonate
Precisely Regulated
383
Concentration
397
Acids and Bases—Their Definitions
Treatment of Acidosis or Alkalosis
398
and Meanings
383
Clinical Measurements and Analysis of
Defenses Against Changes in Hydrogen
Acid-Base Disorders
398
Ion Concentration: Buffers, Lungs,
Complex Acid-Base Disorders and Use of
and Kidneys
384
the Acid-Base Nomogram for Diagnosis
399
Buffering of Hydrogen Ions in the Body
Use of Anion Gap to Diagnose Acid-Base
Fluids
385
Disorders
400
Bicarbonate Buffer System
385
Quantitative Dynamics of the Bicarbonate
C H A P T E R
3
1
Buffer System
385
Phosphate Buffer System
387
Kidney Diseases and Diuretics
402
Proteins: Important Intracellular
Diuretics and Their Mechanisms of
Buffers
387
Action
402
Respiratory Regulation of Acid-Base
Osmotic Diuretics Decrease Water
Balance
388
Reabsorption by Increasing Osmotic
Pulmonary Expiration of CO2 Balances
Pressure of Tubular Fluid
402
Metabolic Formation of CO2
388
“Loop” Diuretics Decrease Active
Increasing Alveolar Ventilation Decreases
Sodium-Chloride-Potassium Reabsorption
Extracellular Fluid Hydrogen Ion
in the Thick Ascending Loop of Henle
403
Concentration and Raises pH
388
Thiazide Diuretics Inhibit Sodium-Chloride
Increased Hydrogen Ion Concentration
Reabsorption in the Early Distal Tubule
404
Stimulates Alveolar Ventilation
389
Carbonic Anhydrase Inhibitors Block
Renal Control of Acid-Base Balance
390
Sodium-Bicarbonate Reabsorption in the
Secretion of Hydrogen Ions and
Proximal Tubules
404
Reabsorption of Bicarbonate Ions
Competitive Inhibitors of Aldosterone
by the Renal Tubules
390
Decrease Sodium Reabsorption from and
Hydrogen Ions Are Secreted by Secondary
Potassium Secretion into the Cortical
Active Transport in the Early Tubular
Collecting Tubule
404
Segments
391
Diuretics That Block Sodium Channels
Filtered Bicarbonate Ions Are Reabsorbed
in the Collecting Tubules Decrease
by Interaction with Hydrogen Ions in the
Sodium Reabsorption
404
Tubules
391
Kidney Diseases
404
Primary Active Secretion of Hydrogen Ions in
Acute Renal Failure
404
the Intercalated Cells of Late Distal and
Prerenal Acute Renal Failure Caused by
Collecting Tubules
392
Decreased Blood Flow to the Kidney
405
Combination of Excess Hydrogen Ions
Intrarenal Acute Renal Failure Caused by
with Phosphate and Ammonia Buffers
Abnormalities within the Kidney
405
in the Tubule—A Mechanism for
Postrenal Acute Renal Failure Caused by
Generating “New” Bicarbonate Ions
392
Abnormalities of the Lower Urinary
Phosphate Buffer System Carries Excess
Tract
406
Hydrogen Ions into the Urine and
Physiologic Effects of Acute Renal Failure
406
Generates New Bicarbonate
393
Chronic Renal Failure: An Irreversible
Excretion of Excess Hydrogen Ions and
Decrease in the Number of Functional
Generation of New Bicarbonate by the
Nephrons
406
Ammonia Buffer System
393
Vicious Circle of Chronic Renal Failure
Quantifying Renal Acid-Base Excretion
394
Leading to End-Stage Renal Disease
407
Regulation of Renal Tubular Hydrogen Ion
Injury to the Renal Vasculature as a Cause
Secretion
395
of Chronic Renal Failure
408
xxii
Table of Contents
Injury to the Glomeruli as a Cause of
C H A P T E R
3
4
Chronic Renal Failure—
Resistance of the Body to Infection: II.
Glomerulonephritis
408
Injury to the Renal Interstitium as a
Immunity and Allergy
439
Cause of Chronic Renal Failure—
Innate Immunity
439
Pyelonephritis
409
Acquired (Adaptive) Immunity
439
Nephrotic Syndrome—Excretion of Protein
Basic Types of Acquired Immunity
440
in the Urine Because of Increased
Both Types of Acquired Immunity Are
Glomerular Permeability
409
Initiated by Antigens
440
Nephron Function in Chronic Renal Failure
409
Lymphocytes Are Responsible for
Effects of Renal Failure on the Body
Acquired Immunity
440
Fluids—Uremia
411
Preprocessing of the T and B Lymphocytes
440
Hypertension and Kidney Disease
412
T Lymphocytes and B-Lymphocyte
Specific Tubular Disorders
413
Antibodies React Highly Specifically
Treatment of Renal Failure by Dialysis
Against Specific Antigens—Role of
with an Artificial Kidney
414
Lymphocyte Clones
442
Origin of the Many Clones of Lymphocytes
442
Specific Attributes of the B-Lymphocyte
System—Humoral Immunity and the
U N I T V I
Antibodies
443
Special Attributes of the T-Lymphocyte
Blood Cells, Immunity, and Blood
System-Activated T Cells and Cell-
Clotting
Mediated Immunity
446
Several Types of T Cells and Their Different
Functions
446
C H A P T E R
3
2
Tolerance of the Acquired Immunity
Red Blood Cells, Anemia, and
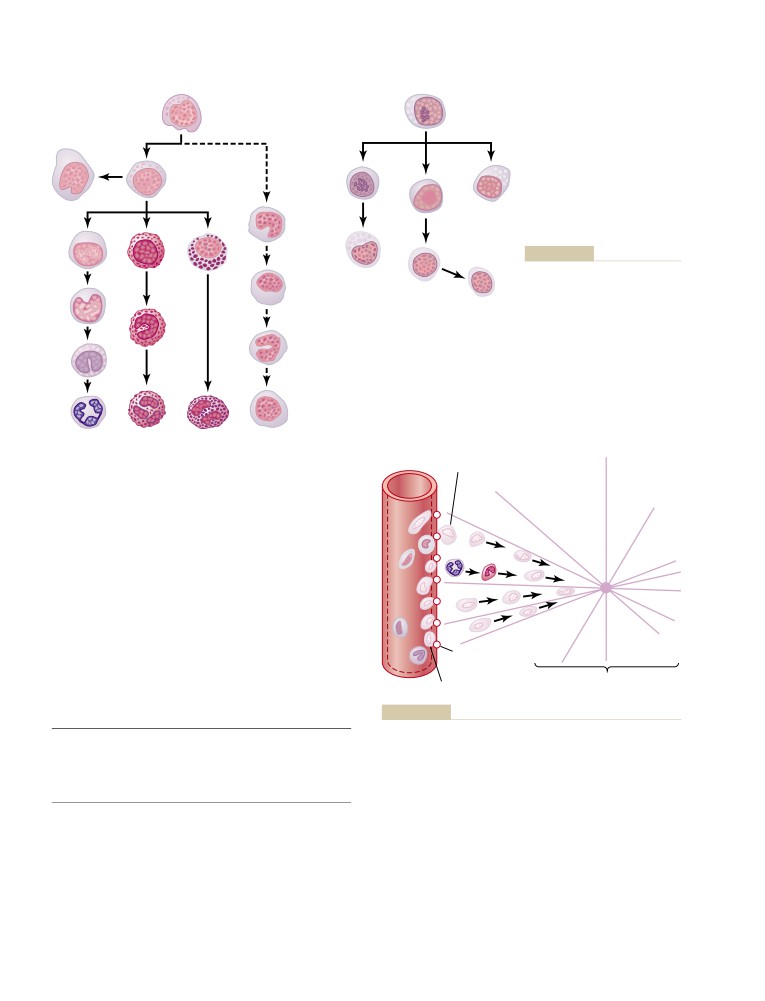
System to One’s Own Tissues—Role
Polycythemia
419
of Preprocessing in the Thymus and
Red Blood Cells (Erythrocytes)
419
Bone Marrow
448
Production of Red Blood Cells
420
Immunization by Injection of Antigens
448
Formation of Hemoglobin
424
Passive Immunity
449
Iron Metabolism
425
Allergy and Hypersensitivity
449
Life Span and Destruction of Red Blood
Allergy Caused by Activated T Cells:
Cells
426
Delayed-Reaction Allergy
449
Anemias
426
Allergies in the “Allergic” Person, Who Has
Effects of Anemia on Function of the
Excess IgE Antibodies
449
Circulatory System
427
Polycythemia
427
C H A P T E R
3
5
Effect of Polycythemia on Function of the
Blood Types; Transfusion; Tissue and
Circulatory System
428
Organ Transplantation
451
Antigenicity Causes Immune Reactions
C H A P T E R
3
3
of Blood
451
Resistance of the Body to Infection: I.
O-A-B Blood Types
451
Leukocytes, Granulocytes, the
A and B Antigens—Agglutinogens
451
Agglutinins
452
Monocyte-Macrophage System, and
Agglutination Process In Transfusion
Inflammation
429
Reactions
452
Leukocytes (White Blood Cells)
429
Blood Typing
453
General Characteristics of Leukocytes
429
Rh Blood Types
453
Genesis of the White Blood Cells
430
Rh Immune Response
453
Life Span of the White Blood Cells
431
Transfusion Reactions Resulting from
Neutrophils and Macrophages Defend
Mismatched Blood Types
454
Against Infections
431
Transplantation of Tissues and Organs
455
Phagocytosis
431
Attempts to Overcome Immune Reactions
Monocyte-Macrophage Cell System
in Transplanted Tissue
455
(Reticuloendothelial System)
432
Inflammation: Role of Neutrophils and
C H A P T E R
3
6
Macrophages
434
Inflammation
434
Hemostasis and Blood Coagulation
457
Macrophage and Neutrophil Responses
Events in Hemostasis
457
During Inflammation
434
Vascular Constriction
457
Eosinophils
436
Formation of the Platelet Plug
457
Basophils
436
Blood Coagulation in the Ruptured
Leukopenia
436
Vessel
458
The Leukemias
437
Fibrous Organization or Dissolution of the
Effects of Leukemia on the Body
437
Blood Clot
458
Table of Contents
xxiii
Mechanism of Blood Coagulation
459
C H A P T E R
3
8
Conversion of Prothrombin to Thrombin
459
Pulmonary Circulation, Pulmonary
Conversion of Fibrinogen to Fibrin—
Formation of the Clot
460
Edema, Pleural Fluid
483
Vicious Circle of Clot Formation
460
Physiologic Anatomy of the Pulmonary
Initiation of Coagulation: Formation of
Circulatory System
483
Prothrombin Activator
461
Pressures in the Pulmonary System
483
Prevention of Blood Clotting in the
Blood Volume of the Lungs
484
Normal Vascular System—Intravascular
Blood Flow Through the Lungs and
Anticoagulants
463
Its Distribution
485
Lysis of Blood Clots—Plasmin
464
Effect of Hydrostatic Pressure
Conditions That Cause Excessive
Gradients in the Lungs on Regional
Bleeding in Human Beings
464
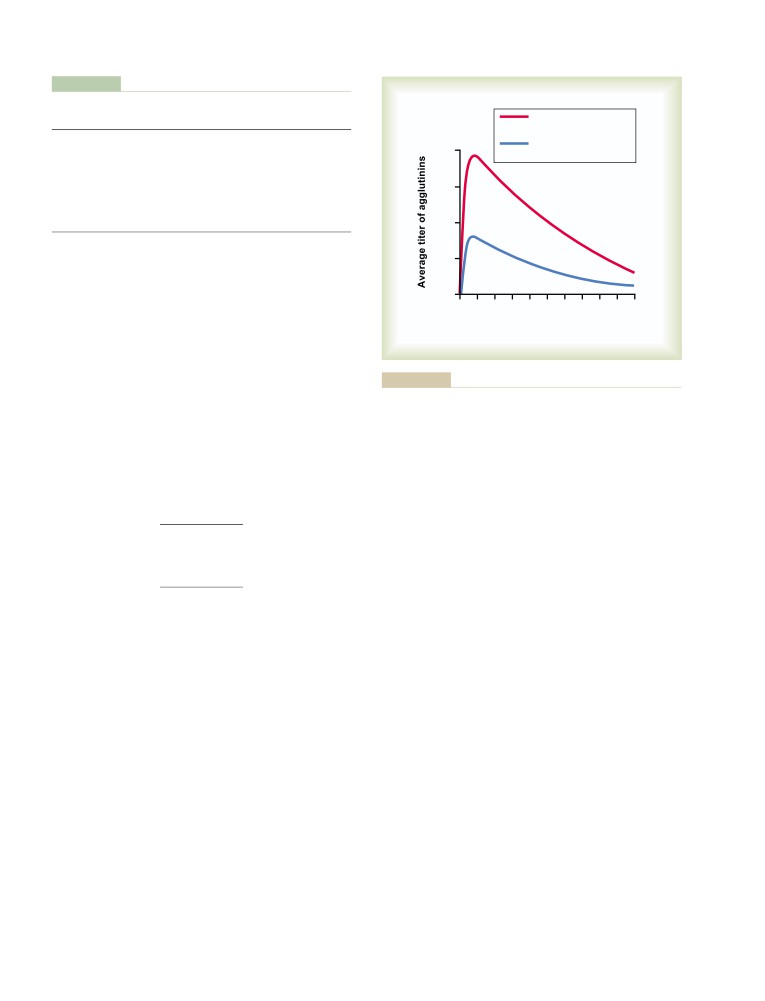
Pulmonary Blood Flow
485
Decreased Prothrombin, Factor VII,
Zones 1, 2, and 3 of Pulmonary Blood Flow
485
Factor IX,and Factor X Caused by
Effect of Increased Cardiac Output on
Vitamin K Deficiency
464
Pulmonary Blood Flow and Pulmonary
Hemophilia
465
Arterial Pressure During Heavy Exercise
486
Thrombocytopenia
465
Function of the Pulmonary Circulation
Thromboembolic Conditions in the
When the Left Atrial Pressure Rises as a
Human Being
465
Result of Left-Sided Heart Failure
487
Femoral Venous Thrombosis and Massive
Pulmonary Capillary Dynamics
487
Pulmonary Embolism
466
Capillary Exchange of Fluid in the Lungs,
Disseminated Intravascular Coagulation
466
and Pulmonary Interstitial Fluid Dynamics
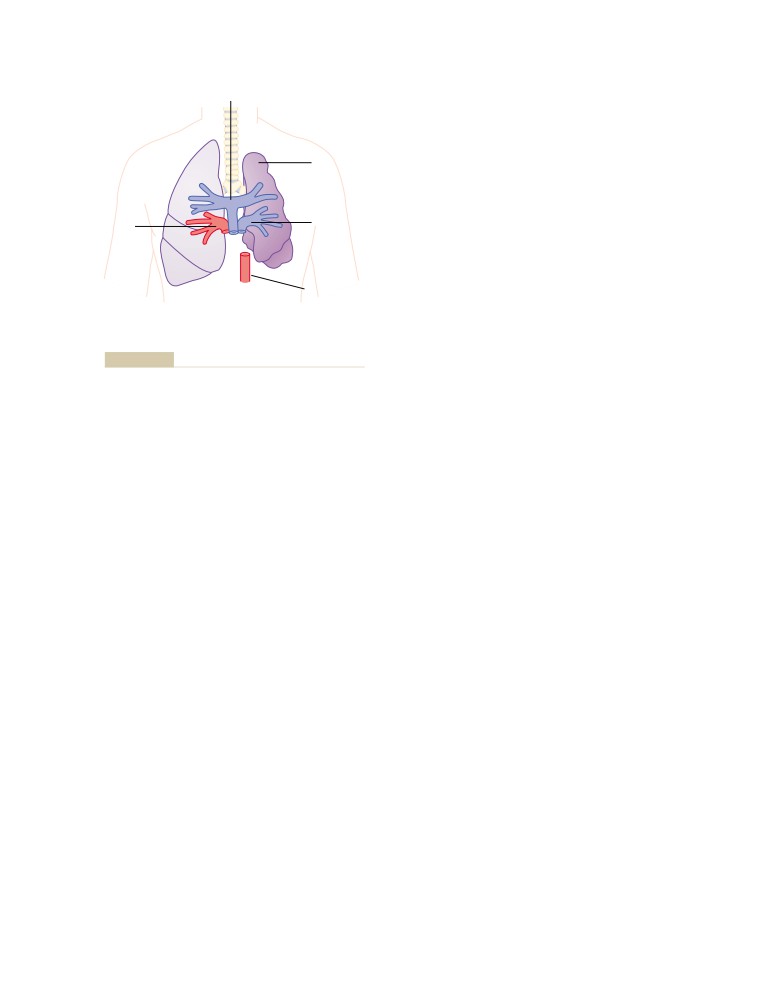
487
Anticoagulants for Clinical Use
466
Pulmonary Edema
488
Heparin as an Intravenous Anticoagulant
466
Fluid in the Pleural Cavity
489
Coumarins as Anticoagulants
466
Prevention of Blood Coagulation Outside
C H A P T E R
3
9
the Body
466
Blood Coagulation Tests
467
Physical Principles of Gas Exchange;
Bleeding Time
467
Diffusion of Oxygen and Carbon
Clotting Time
467
Dioxide Through the Respiratory
Prothrombin Time
467
Membrane
491
Physics of Gas Diffusion and Gas
U N I T V I I
Partial Pressures
491
Molecular Basis of Gas Diffusion
491
Respiration
Gas Pressures in a Mixture of Gases—
“Partial Pressures” of Individual Gases
491
C H A P T E R
3
7
Pressures of Gases Dissolved in Water
Pulmonary Ventilation
471
and Tissues
492
Mechanics of Pulmonary Ventilation
471
Vapor Pressure of Water
492
Muscles That Cause Lung Expansion and
Diffusion of Gases Through Fluids—
Contraction
471
Pressure Difference Causes Net
Movement of Air In and Out of the Lungs
Diffusion
493
and the Pressures That Cause the
Diffusion of Gases Through Tissues
493
Movement
472
Composition of Alveolar Air—Its Relation
Effect of the Thoracic Cage on Lung
to Atmospheric Air
493
Expansibility
474
Rate at Which Alveolar Air Is Renewed by
Pulmonary Volumes and Capacities
475
Atmospheric Air
494
Recording Changes in Pulmonary Volume—
Oxygen Concentration and Partial Pressure
Spirometry
475
in the Alveoli
494
Abbreviations and Symbols Used in
CO2 Concentration and Partial Pressure in
Pulmonary Function Tests
476
the Alveoli
495
Determination of Functional Residual
Expired Air
495
Capacity, Residual Volume, and Total
Diffusion of Gases Through the
Lung Capacity—Helium Dilution Method
476
Respiratory Membrane
496
Minute Respiratory Volume Equals
Factors That Affect the Rate of Gas
Respiratory Rate Times Tidal Volume
477
Diffusion Through the Respiratory
Alveolar Ventilation
477
Membrane
498
“Dead Space” and Its Effect on Alveolar
Diffusing Capacity of the Respiratory
Ventilation
477
Membrane
498
Rate of Alveolar Ventilation
478
Effect of the Ventilation-Perfusion
Functions of the Respiratory
Ratio on .Al.eolar Gas Concentration
499
Passageways
478
PO2-PCO2, VA/Q
Diagram
500
Trachea, Bronchi, and Bronchioles
478
Concept
Normal Respiratory Functions of the
(When VA/Q
Is Greater Than Normal)
500
Nose
480
Abnormalities of Ventilation-Perfusion Ratio
501
xxiv
Table of Contents
C H A P T E R
4
0
Chronic Breathing of Low Oxygen Stimulates
Respiration Even More—The Phenomenon
Transport of Oxygen and Carbon
of “Acclimatization”
519
Dioxide in Blood and Tissue Fluids
502
Composite Effects of PCO2, pH, and PO2 on
Transport of Oxygen from the Lungs to
Alveolar Ventilation
519
the Body Tissues
502
Regulation of Respiration During
Diffusion of Oxygen from the Alveoli to the
Exercise
520
Pulmonary Capillary Blood
502
Other Factors That Affect Respiration
521
Transport of Oxygen in the Arterial Blood
503
Sleep Apnea
522
Diffusion of Oxygen from the Peripheral
Capillaries into the Tissue Fluid
503
Diffusion of Oxygen from the Peripheral
C H A P T E R
4
2
Capillaries to the Tissue Cells
504
Respiratory Insufficiency—
Diffusion of Carbon Dioxide from the
Pathophysiology, Diagnosis, Oxygen
Peripheral Tissue Cells into the
Capillaries and from the Pulmonary
Therapy
524
Capillaries into the Alveoli
504
Useful Methods for Studying Respiratory
Role of Hemoglobin in Oxygen Transport
505
Abnormalities
524
Reversible Combination of Oxygen with
Study of Blood Gases and Blood pH
524
Hemoglobin
505
Measurement of Maximum Expiratory Flow
525
Effect of Hemoglobin to “Buffer” the
Forced Expiratory Vital Capacity and Forced
Tissue PO2
507
Expiratory Volume
526
Factors That Shift the Oxygen-Hemoglobin
Physiologic Peculiarities of Specific
Dissociation Curve—Their Importance for
Pulmonary Abnormalities
526
Oxygen Transport
507
Chronic Pulmonary Emphysema
526
Metabolic Use of Oxygen by the Cells
508
Pneumonia
527
Transport of Oxygen in the Dissolved State
509
Atelectasis
528
Combination of Hemoglobin with Carbon
Asthma
529
Monoxide—Displacement of Oxygen
509
Tuberculosis
530
Transport of Carbon Dioxide in the Blood
510
Hypoxia and Oxygen Therapy
530
Chemical Forms in Which Carbon Dioxide
Oxygen Therapy in Different Types of
Is Transported
510
Hypoxia
530
Carbon Dioxide Dissociation Curve
511
Cyanosis
531
When Oxygen Binds with Hemoglobin,
Hypercapnia
531
Carbon Dioxide Is Released (the Haldane
Dyspnea
532
Effect) to Increase CO2 Transport
511
Artificial Respiration
532
Change in Blood Acidity During Carbon
Dioxide Transport
512
Respiratory Exchange Ratio
512
U N I T V I I I
C H A P T E R
4
1
Aviation, Space, and Deep-Sea
Regulation of Respiration
514
Diving Physiology
Respiratory Center
514
Dorsal Respiratory Group of Neurons—Its
Control of Inspiration and of Respiratory
C H A P T E R
4
3
Rhythm
514
Aviation, High-Altitude, and Space
A Pneumotaxic Center Limits the Duration
Physiology
537
of Inspiration and Increases the
Respiratory Rate
514
Effects of Low Oxygen Pressure on the
Ventral Respiratory Group of Neurons—
Body
537
Functions in Both Inspiration and
Alveolar PO2 at Different Elevations
537
Expiration
515
Effect of Breathing Pure Oxygen on Alveolar
Lung Inflation Signals Limit Inspiration—
PO2 at Different Altitudes
538
The Hering-Breuer Inflation Reflex
515
Acute Effects of Hypoxia
538
Control of Overall Respiratory Center
Acclimatization to Low PO2
539
Activity
516
Natural Acclimatization of Native Human
Chemical Control of Respiration
516
Beings Living at High Altitudes
540
Direct Chemical Control of Respiratory
Reduced Work Capacity at High Altitudes
Center Activity by Carbon Dioxide and
and Positive Effect of Acclimatization
540
Hydrogen Ions
516
Acute Mountain Sickness and High-Altitude
Peripheral Chemoreceptor System for
Pulmonary Edema
540
Control of Respiratory Activity—Role
Chronic Mountain Sickness
541
of Oxygen in Respiratory Control
518
Effects of Acceleratory Forces on the
Effect of Low Arterial PO2 to Stimulate
Body in Aviation and Space Physiology
541
Alveolar Ventilation When Arterial Carbon
Centrifugal Acceleratory Forces
541
Dioxide and Hydrogen Ion Concentrations
Effects of Linear Acceleratory Forces on the
Remain Normal
519
Body
542
Table of Contents
xxv
“Artificial Climate” in the Sealed
C H A P T E R
4
6
Spacecraft
543
Sensory Receptors, Neuronal Circuits
Weightlessness in Space
543
for Processing Information
572
Types of Sensory Receptors and the
C H A P T E R
4
4
Sensory Stimuli They Detect
572
Physiology of Deep-Sea Diving and
Differential Sensitivity of Receptors
572
Transduction of Sensory Stimuli into
Other Hyperbaric Conditions
545
Nerve Impulses
573
Effect of High Partial Pressures of
Local Electrical Currents at Nerve Endings—
Individual Gases on the Body
545
Receptor Potentials
573
Nitrogen Narcosis at High Nitrogen
Adaptation of Receptors
575
Pressures
545
Nerve Fibers That Transmit Different
Oxygen Toxicity at High Pressures
546
Types of Signals, and Their
Carbon Dioxide Toxicity at Great Depths
Physiologic Classification
576
in the Sea
547
Transmission of Signals of Different
Decompression of the Diver After Excess
Intensity in Nerve Tracts—Spatial and
Exposure to High Pressure
547
Temporal Summation
577
Scuba (Self-Contained Underwater
Transmission and Processing of Signals
Breathing Apparatus) Diving
549
in Neuronal Pools
578
Special Physiologic Problems in
Relaying of Signals Through Neuronal
Submarines
550
Pools
579
Hyperbaric Oxygen Therapy
550
Prolongation of a Signal by a Neuronal
Pool—“Afterdischarge”
581
Instability and Stability of Neuronal
U N I T I X
Circuits
583
Inhibitory Circuits as a Mechanism for
The Nervous System: A. General
Stabilizing Nervous System Function
583
Synaptic Fatigue as a Means for Stabilizing
Principles and Sensory Physiology
the Nervous System
583
C H A P T E R
4
5
Organization of the Nervous System,
C H A P T E R
4
7
Basic Functions of Synapses,
Somatic Sensations: I. General
“Transmitter Substances”
555
Organization, the Tactile and
General Design of the Nervous System
555
Central Nervous System Neuron: The Basic
Position Senses
585
Functional Unit
555
CLASSIFICATION OF SOMATIC SENSES
585
Sensory Part of the Nervous System—
Detection and Transmission of Tactile
Sensory Receptors
555
Sensations
585
Motor Part of the Nervous System—
Detection of Vibration
587
Effectors
556
TICKLE AND ITCH
587
Processing of Information—“Integrative”
Sensory Pathways for Transmitting
Function of the Nervous System
556
Somatic Signals into the Central
Storage of Information—Memory
557
Nervous System
587
Major Levels of Central Nervous System
Dorsal Column-Medial Lemniscal System
588
Function
557
Anterolateral System
588
Spinal Cord Level
557
Transmission in the Dorsal Column—
Lower Brain or Subcortical Level
558
Medial Lemniscal System
588
Higher Brain or Cortical Level
558
Anatomy of the Dorsal Column—Medial
Comparison of the Nervous System
Lemniscal System
588
with a Computer
558
Somatosensory Cortex
589
Central Nervous System Synapses
559
Somatosensory Association Areas
592
Types of Synapses—Chemical and
Overall Characteristics of Signal
Electrical
559
Transmission and Analysis in the Dorsal
Physiologic Anatomy of the Synapse
559
Column-Medial Lemniscal System
592
Chemical Substances That Function as
Position Senses
594
Synaptic Transmitters
562
Interpretation of Sensory Stimulus Intensity
593
Electrical Events During Neuronal Excitation
564
Judgment of Stimulus Intensity
594
Electrical Events During Neuronal
Position Senses
594
Inhibition
566
Transmission of Less Critical Sensory
Special Functions of Dendrites for Exciting
Signals in the Anterolateral Pathway
595
Neurons
568
Anatomy of the Anterolateral Pathway
595
Relation of State of Excitation of the Neuron
Some Special Aspects of
to Rate of Firing
569
Somatosensory Function
596
Some Special Characteristics of
Function of the Thalamus in Somatic
Synaptic Transmission
570
Sensation
596
xxvi
Table of Contents
Cortical Control of Sensory Sensitivity—
Pupillary Diameter
618
“Corticofugal” Signals
597
Errors of Refraction
619
Segmental Fields of Sensation—The
Visual Acuity
621
Dermatomes
597
Determination of Distance of an Object
from the Eye—“Depth Perception”
621
Ophthalmoscope
622
C H A P T E R
4
8
Fluid System of the Eye—Intraocular
Somatic Sensations: II. Pain,
Fluid
623
Headache, and Thermal Sensations
598
Formation of Aqueous Humor by the Ciliary
Body
623
Types of Pain and Their Qualities—
Outflow of Aqueous Humor from the Eye
623
Fast Pain and Slow Pain
598
Intraocular Pressure
624
Pain Receptors and Their Stimulation
598
Rate of Tissue Damage as a Stimulus
for Pain
599
C H A P T E R
5
0
Dual Pathways for Transmission of Pain
The Eye: II. Receptor and Neural
Signals into the Central Nervous
Function of the Retina
626
System
600
Anatomy and Function of the
Dual Pain Pathways in the Cord and Brain
Structural Elements of the Retina
626
Stem—The Neospinothalamic Tract and
Photochemistry of Vision
628
the Paleospinothalamic Tract
600
Rhodopsin-Retinal Visual Cycle, and
Pain Suppression (“Analgesia”) System
Excitation of the Rods
629
in the Brain and Spinal Cord
602
Automatic Regulation of Retinal Sensitivity—
Brain’s Opiate System—Endorphins and
Light and Dark Adaptation
631
Enkephalins
602
Color Vision
632
Inhibition of Pain Transmission by
Tricolor Mechanism of Color Detection
632
Simultaneous Tactile Sensory Signals
603
Color Blindness
633
Treatment of Pain by Electrical Stimulation
603
Neural Function of the Retina
633
Referred Pain
603
Neural Circuitry of the Retina
633
Visceral Pain
603
Ganglion Cells and Optic Nerve Fibers
636
Causes of True Visceral Pain
604
Excitation of the Ganglion Cells
637
“Parietal Pain” Caused by Visceral Disease
604
Localization of Visceral Pain—“Visceral”
and the “Parietal” Pain Transmission
C H A P T E R
5
1
Pathways
604
The Eye: III. Central
Some Clinical Abnormalities of Pain
Neurophysiology of Vision
640
and Other Somatic Sensations
605
Hyperalgesia
605
Visual Pathways
640
Herpes Zoster (Shingles)
605
Function of the Dorsal Lateral Geniculate
Tic Douloureux
605
Nucleus of the Thalamus
640
Brown-Séquard Syndrome
606
Organization and Function of the Visual
Headache
606
Cortex
641
Headache of Intracranial Origin
606
Layered Structure of the Primary Visual
Thermal Sensations
607
Cortex
642
Thermal Receptors and Their Excitation
607
Two Major Pathways for Analysis of Visual
Transmission of Thermal Signals in the
Information—(1) The Fast “Position” and
Nervous System
609
“Motion” Pathway; (2) The Accurate Color
Pathway
643
Neuronal Patterns of Stimulation During
Analysis of the Visual Image
643
U N I T X
Detection of Color
644
Effect of Removing the Primary Visual
The Nervous System: B.
Cortex
644
The Special Senses
Fields of Vision; Perimetry
644
Eye Movements and Their Control
645
Fixation Movements of the Eyes
645
C H A P T E R
4
9
“Fusion” of the Visual Images from the
The Eye: I. Optics of Vision
613
Two Eyes
647
Physical Principles of Optics
613
Autonomic Control of Accommodation
Refraction of Light
613
and Pupillary Aperture
648
Application of Refractive Principles to
Control of Accommodation (Focusing the
Lenses
613
Eyes)
649
Focal Length of a Lens
615
Control of Pupillary Diameter
649
Formation of an Image by a Convex Lens
616
Measurement of the Refractive Power of a
C H A P T E R
5
2
Lens—“Diopter”
616
The Sense of Hearing
651
Optics of the Eye
617
The Eye as a Camera
617
Tympanic Membrane and the Ossicular
Mechanism of “Accommodation”
617
System
651
Table of Contents
xxvii
Conduction of Sound from the Tympanic
Reciprocal Inhibition and Reciprocal
Membrane to the Cochlea
651
Innervation
681
Transmission of Sound Through Bone
652
Reflexes of Posture and Locomotion
682
Cochlea
652
Postural and Locomotive Reflexes of
Functional Anatomy of the Cochlea
652
the Cord
682
Transmission of Sound Waves in the
Scratch Reflex
683
Cochlea—“Traveling Wave”
654
Spinal Cord Reflexes That Cause
Function of the Organ of Corti
655
Muscle Spasm
683
Determination of Sound Frequency—The
Autonomic Reflexes in the Spinal
“Place” Principle
656
Cord
683
Determination of Loudness
656
Spinal Cord Transection and Spinal
Central Auditory Mechanisms
657
Shock
684
Auditory Nervous Pathways
657
Function of the Cerebral Cortex in Hearing
658
Determination of the Direction from Which
C H A P T E R
5
5
Sound Comes
660
Cortical and Brain Stem Control of
Centrifugal Signals from the Central
Motor Function
685
Nervous System to Lower Auditory
Centers
660
MOTOR CORTEX AND CORTICOSPINAL
Hearing Abnormalities
660
TRACT
685
Types of Deafness
660
Primary Motor Cortex
685
Premotor Area
686
Supplementary Motor Area
686
C H A P T E R
5
3
Some Specialized Areas of Motor Control
The Chemical Senses—Taste and
Found in the Human Motor Cortex
686
Transmission of Signals from the Motor
Smell
663
Cortex to the Muscles
687
Sense of Taste
663
Incoming Fiber Pathways to the Motor
Primary Sensations of Taste
663
Cortex
688
Taste Bud and Its Function
664
Red Nucleus Serves as an Alternative
Transmission of Taste Signals into the
Pathway for Transmitting Cortical Signals
Central Nervous System
665
to the Spinal Cord
688
Taste Preference and Control of the Diet
666
“Extrapyramidal” System
689
Sense of Smell
667
Excitation of the Spinal Cord Motor Control
Olfactory Membrane
667
Areas by the Primary Motor Cortex and
Stimulation of the Olfactory Cells
667
Red Nucleus
689
Transmission of Smell Signals into the
Role of the Brain Stem in Controlling
Central Nervous System
668
Motor Function
691
Support of the Body Against Gravity—
Roles of the Reticular and Vestibular
Nuclei
691
U N I T X I
Vestibular Sensations and Maintenance
The Nervous System: C. Motor and
of Equilibrium
692
Vestibular Apparatus
692
Integrative Neurophysiology
Function of the Utricle and Saccule in the
Maintenance of Static Equilibrium
694
Detection of Head Rotation by the
C H A P T E R
5
4
Semicircular Ducts
695
Motor Functions of the Spinal Cord;
Vestibular Mechanisms for Stabilizing the
the Cord Reflexes
673
Eyes
696
Organization of the Spinal Cord for Motor
Other Factors Concerned with Equilibrium
696
Functions
673
Functions of Brain Stem Nuclei in
Muscle Sensory Receptors—Muscle
Controlling Subconscious, Stereotyped
Spindles and Golgi Tendon Organs—
Movements
697
And Their Roles in Muscle Control
675
Receptor Function of the Muscle Spindle
675
Muscle Stretch Reflex
676
C H A P T E R
5
6
Role of the Muscle Spindle in Voluntary
Contributions of the Cerebellum and
Motor Activity
678
Basal Ganglia to Overall Motor
Clinical Applications of the Stretch
Reflex
678
Control
698
Golgi Tendon Reflex
679
Cerebellum and Its Motor Functions
698
Function of the Muscle Spindles and Golgi
Anatomical Functional Areas of the
Tendon Organs in Conjunction with Motor
Cerebellum
699
Control from Higher Levels of the Brain
680
Neuronal Circuit of the Cerebellum
700
Flexor Reflex and the Withdrawal
Function of the Cerebellum in Overall
Reflexes
680
Motor Control
703
Crossed Extensor Reflex
681
Clinical Abnormalities of the Cerebellum
706
xxviii
Table of Contents
Basal Ganglia—Their Motor Functions
707
Hypothalamus, a Major Control
Function of the Basal Ganglia in Executing
Headquarters for the Limbic System
732
Patterns of Motor Activity—The Putamen
Vegetative and Endocrine Control
Circuit
708
Functions of the Hypothalamus
733
Role of the Basal Ganglia for Cognitive
Behavioral Functions of the Hypothalamus
Control of Sequences of Motor Patterns—
and Associated Limbic Structures
734
The Caudate Circuit
709
“Reward” and “Punishment” Function of
Function of the Basal Ganglia to Change
the Limbic System
735
the Timing and to Scale the Intensity of
Importance of Reward or Punishment in
Movements
709
Behavior
736
Functions of Specific Neurotransmitter
Specific Functions of Other Parts of
Substances in the Basal Ganglial
the Limbic System
736
System
710
Functions of the Hippocampus
736
Integration of the Many Parts of the
Functions of the Amygdala
737
Total Motor Control System
712
Function of the Limbic Cortex
738
Spinal Level
712
Hindbrain Level
712
C H A P T E R
5
9
Motor Cortex Level
712
What Drives Us to Action?
713
States of Brain Activity—Sleep, Brain
Waves, Epilepsy, Psychoses
739
Sleep
739
C H A P T E R
5
7
Slow-Wave Sleep
739
Cerebral Cortex, Intellectual Functions
REM Sleep (Paradoxical Sleep,
of the Brain, Learning and Memory
714
Desynchronized Sleep)
740
Basic Theories of Sleep
740
Physiologic Anatomy of the Cerebral
Physiologic Effects of Sleep
741
Cortex
714
Brain Waves
741
Functions of Specific Cortical Areas
715
Origin of Brain Waves
742
Association Areas
716
Effect of Varying Levels of Cerebral
Comprehensive Interpretative Function of
Activity on the Frequency of the EEG
743
the Posterior Superior Temporal Lobe—
Changes in the EEG at Different Stages of
“Wernicke’s Area” (a General
Wakefulness and Sleep
743
Interpretative Area)
718
Epilepsy
743
Functions of the Parieto-occipitotemporal
Grand Mal Epilepsy
743
Cortex in the Nondominant Hemisphere
719
Petit Mal Epilepsy
744
Higher Intellectual Functions of the
Focal Epilepsy
744
Prefrontal Association Areas
719
Psychotic Behavior and Dementia—
Function of the Brain in
Roles of Specific Neurotransmitter
Communication—Language Input
Systems
745
and Language Output
720
Depression and Manic-Depressive
Function of the Corpus Callosum and
Psychoses—Decreased Activity of the
Anterior Commissure to Transfer
Norepinephrine and Serotonin
Thoughts, Memories, Training, and
Neurotransmitter Systems
745
Other Information Between the Two
Schizophrenia—Possible Exaggerated
Cerebral Hemispheres
722
Function of Part of the Dopamine
Thoughts, Consciousness, and Memory
723
System
745
Memory—Roles of Synaptic Facilitation and
Alzheimer’s Disease—Amyloid Plaques
Synaptic Inhibition
723
and Depressed Memory
746
Short-Term Memory
724
Intermediate Long-Term Memory
724
Long-Term Memory
725
C H A P T E R
6
0
Consolidation of Memory
725
The Autonomic Nervous System and
the Adrenal Medulla
748
C H A P T E R
5
8
General Organization of the Autonomic
Behavioral and Motivational
Nervous System
748
Mechanisms of the Brain—The
Physiologic Anatomy of the Sympathetic
Nervous System
748
Limbic System and the
Preganglionic and Postganglionic
Hypothalamus
728
Sympathetic Neurons
748
Activating-Driving Systems of the Brain
728
Physiologic Anatomy of the
Control of Cerebral Activity by Continuous
Parasympathetic Nervous System
750
Excitatory Signals from the Brain Stem
728
Basic Characteristics of Sympathetic
Neurohormonal Control of Brain Activity
730
and Parasympathetic Function
750
Limbic System
731
Cholinergic and Adrenergic Fibers—
Functional Anatomy of the Limbic
Secretion of Acetylcholine or
System; Key Position of the
Norepinephrine
750
Hypothalamus
731
Receptors on the Effector Organs
752
Table of Contents
xxix
Excitatory and Inhibitory Actions of
Physiological Anatomy of the
Sympathetic and Parasympathetic
Gastrointestinal Wall
771
Stimulation
753
Neural Control of Gastrointestinal
Effects of Sympathetic and Parasympathetic
Function—Enteric Nervous System
773
Stimulation on Specific Organs
753
Differences Between the Myenteric and
Function of the Adrenal Medullae
755
Submucosal Plexuses
774
Relation of Stimulus Rate to Degree of
Types of Neurotransmitters Secreted by
Sympathetic and Parasympathetic Effect
756
Enteric Neurons
775
Sympathetic and Parasympathetic “Tone”
756
Hormonal Control of Gastrointestinal
Denervation Supersensitivity of Sympathetic
Motility
776
and Parasympathetic Organs after
Functional Types of Movements in the
Denervation
756
Gastrointestinal Tract
776
Autonomic Reflexes
757
Propulsive Movements—Peristalsis
776
Stimulation of Discrete Organs in Some
Mixing Movements
777
Instances and Mass Stimulation in
Gastrointestinal Blood Flow—
Other Instances by the Sympathetic
“Splanchnic Circulation”
777
and Parasympathetic Systems
757
Anatomy of the Gastrointestinal Blood
“Alarm” or “Stress” Response of the
Supply
778
Sympathetic Nervous System
758
Effect of Gut Activity and Metabolic
Medullary, Pontine, and Mesencephalic
Factors on Gastrointestinal Blood
Control of the Autonomic Nervous
Flow
778
System
758
Nervous Control of Gastrointestinal Blood
Pharmacology of the Autonomic
Flow
779
Nervous System
759
Drugs That Act on Adrenergic Effector
Organs—Sympathomimetic Drugs
759
C H A P T E R
6
3
Drugs That Act on Cholinergic Effector
Organs
759
Propulsion and Mixing of Food in the
Drugs That Stimulate or Block Sympathetic
Alimentary Tract
781
and Parasympathetic Postganglionic
Ingestion of Food
781
Neurons
759
Mastication (Chewing)
781
Swallowing (Deglutition)
782
C H A P T E R
6
1
Motor Functions of the Stomach
784
Storage Function of the Stomach
784
Cerebral Blood Flow, Cerebrospinal
Mixing and Propulsion Of Food in the
Fluid, and Brain Metabolism
761
Stomach—The Basic Electrical Rhythm
Cerebral Blood Flow
761
of the Stomach Wall
784
Normal Rate of Cerebral Blood Flow
761
Stomach Emptying
785
Regulation of Cerebral Blood Flow
761
Regulation of Stomach Emptying
785
Cerebral Microcirculation
763
Movements of the Small Intestine
786
Cerebral Stroke Occurs When Cerebral
Mixing Contractions (Segmentation
Blood Vessels are Blocked
763
Contractions)
786
Cerebrospinal Fluid System
763
Propulsive Movements
787
Cushioning Function of the Cerebrospinal
Function of the Ileocecal Valve
788
Fluid
763
Movements of the Colon
788
Formation, Flow, and Absorption of
Defecation
789
Cerebrospinal Fluid
764
Other Autonomic Reflexes That Affect
Cerebrospinal Fluid Pressure
765
Bowel Activity
790
Obstruction to Flow of Cerebrospinal Fluid
Can Cause Hydrocephalus
766
Blood-Cerebrospinal Fluid and Blood-Brain
Barriers
766
C H A P T E R
6
4
Brain Edema
766
Secretory Functions of the Alimentary
Brain Metabolism
767
Tract
791
General Principles of Alimentary Tract
Secretion
791
U N I T X I I
Anatomical Types of Glands
791
Basic Mechanisms of Stimulation of the
Gastrointestinal Physiology
Alimentary Tract Glands
791
Basic Mechanism of Secretion by Glandular
C H A P T E R
6
2
Cells
791
Lubricating and Protective Properties of
General Principles of Gastrointestinal
Mucus, and Importance of Mucus in the
Function—Motility, Nervous Control,
Gastrointestinal Tract
793
and Blood Circulation
771
Secretion of Saliva
793
General Principles of Gastrointestinal
Nervous Regulation of Salivary Secretion
794
Motility
771
Esophageal Secretion
795
xxx
Table of Contents
Gastric Secretion
794
Diarrhea
822
Characteristics of the Gastric Secretions
794
Paralysis of Defecation in Spinal Cord
Pyloric Glands—Secretion of Mucus and
Injuries
823
Gastrin
797
General Disorders of the
Surface Mucous Cells
797
Gastrointestinal Tract
823
Stimulation of Gastric Acid Secretion
797
Vomiting
823
Regulation of Pepsinogen Secretion
798
Nausea
824
Inhibition of Gastric Secretion by Other
Gastrointestinal Obstruction
824
Post-Stomach Intestinal Factors
798
Chemical Composition of Gastrin And Other
Gastrointestinal Hormones
799
Pancreatic Secretion
799
U N I T X I I I
Pancreatic Digestive Enzymes
799
Secretion of Bicarbonate Ions
800
Metabolism and Temperature
Regulation of Pancreatic Secretion
800
Regulation
Secretion of Bile by the Liver; Functions
of the Biliary Tree
802
Physiologic Anatomy of Biliary Secretion
802
Function of Bile Salts in Fat Digestion and
C H A P T E R
6
7
Absorption
804
Metabolism of Carbohydrates,
Liver Secretion of Cholesterol and
and Formation of Adenosine
Gallstone Formation
804
Secretions of the Small Intestine
805
Triphosphate
829
Secretion of Mucus by Brunner’s Glands in
Release of Energy from Foods, and the
the Duodenum
805
Concept of “Free Energy”
829
Secretion of Intestinal Digestive Juices by
Role of Adenosine Triphosphate in
the Crypts of Lieberkühn
805
Metabolism
829
Regulation of Small Intestine Secretion—
Central Role of Glucose in
Local Stimuli
806
Carbohydrate Metabolism
830
Secretions of the Large Intestine
806
Transport of Glucose Through the
Cell Membrane
831
Insulin Increases Facilitated Diffusion of
C H A P T E R
6
5
Glucose
831
Digestion and Absorption in the
Phosphorylation of Glucose
831
Gastrointestinal Tract
808
Glycogen Is Stored in Liver and
Muscle
831
Digestion of the Various Foods by
Glycogenesis—The Process of Glycogen
Hydrolysis
808
Formation
832
Digestion of Carbohydrates
809
Removal of Stored Glycogen—
Digestion of Proteins
810
Glycogenolysis
832
Digestion of Fats
811
Release of Energy from the Glucose
Basic Principles of Gastrointestinal
Molecule by the Glycolytic Pathway
832
Absorption
812
Summary of ATP Formation During the
Anatomical Basis of Absorption
812
Breakdown of Glucose
836
Absorption in the Small Intestine
813
Control of Energy Release from Stored
Absorption of Water
814
Glycogen When the Body Needs Additional
Absorption of Ions
814
Energy
836
Absorption of Nutrients
815
Anaerobic Release of Energy—“Anaerobic
Absorption in the Large Intestine:
Glycolysis”
836
Formation of Feces
817
Release of Energy from Glucose by the
Pentose Phosphate Pathway
837
C H A P T E R
6
6
Glucose Conversion to Glycogen or Fat
838
Formation of Carbohydrates from
Physiology of Gastrointestinal
Proteins and Fats—“Gluconeogenesis”
838
Disorders
819
Blood Glucose
839
Disorders of Swallowing and of the
Esophagus
819
Disorders of the Stomach
819
C H A P T E R
6
8
Peptic Ulcer
820
Specific Causes of Peptic Ulcer in the
Lipid Metabolism
840
Human Being
821
Transport of Lipids in the Body Fluids
840
Disorders of the Small Intestine
821
Transport of Triglycerides and Other Lipids
Abnormal Digestion of Food in the Small
from the Gastrointestinal Tract by
Intestine—Pancreatic Failure
821
Lymph—The Chylomicrons
840
Malabsorption by the Small Intestine
Removal of the Chylomicrons from the
Mucosa—Sprue
822
Blood
841
Disorders of the Large Intestine
822
“Free Fatty Acids” Are Transported in the
Constipation
822
Blood in Combination with Albumin
841
Table of Contents
xxxi
Lipoproteins—Their Special Function in
C H A P T E R
7
1
Transporting Cholesterol and
Dietary Balances; Regulation of
Phospholipids
841
Fat Deposits
842
Feeding; Obesity and Starvation;
Adipose Tissue
842
Vitamins and Minerals
865
Liver Lipids
842
Energy Intake and Output Are Balanced
Use of Triglycerides for Energy:
Under Steady-State Conditions
865
Formation of Adenosine Triphosphate
842
Dietary Balances
865
Formation of Acetoacetic Acid in the
Energy Available in Foods
865
Liver and Its Transport in the Blood
844
Methods for Determining Metabolic
Synthesis of Triglycerides from
Utilization of Proteins, Carbohydrates,
Carbohydrates
844
and Fats
866
Synthesis of Triglycerides from Proteins
845
Regulation of Food Intake and Energy
Regulation of Energy Release from
Storage
865
Triglycerides
846
Neural Centers Regulate Food Intake
867
Obesity
846
Factors That Regulate Quantity of Food
Phospholipids and Cholesterol
846
Intake
870
Phospholipids
846
Obesity
872
Cholesterol
847
Decreased Physical Activity and
Cellular Structural Functions of
Abnormal Feeding Regulation as
Phospholipids and Cholesterol—
Causes of Obesity
872
Especially for Membranes
848
Treatment of Obesity
873
Atherosclerosis
848
Inanition, Anorexia, and Cachexia
874
Basic Causes of Atherosclerosis—The
Starvation
874
Roles of Cholesterol and Lipoproteins
850
Vitamins
875
Other Major Risk Factors for
Vitamin A
875
Atherosclerosis
850
Thiamine (Vitamin B1)
875
Prevention of Atherosclerosis
850
Niacin
876
Riboflavin (Vitamin B2)
876
Vitamin B12
876
C H A P T E R
6
9
Folic Acid (Pteroylglutamic Acid)
877
Protein Metabolism
852
Pyridoxine (Vitamin B6)
877
Basic Properties
852
Pantothenic Acid
877
Amino Acids
852
Ascorbic Acid (Vitamin C)
877
Transport and Storage of Amino Acids
854
Vitamin D
878
Blood Amino Acids
854
Vitamin E
878
Storage of Amino Acids as Proteins in the
Vitamin K
878
Cells
854
Mineral Metabolism
878
Functional Roles of the Plasma
Proteins
855
C H A P T E R
7
2
Essential and Nonessential Amino Acids
855
Energetics and Metabolic Rate
881
Obligatory Degradation of Proteins
857
Adenosine Triphosphate (ATP)
Hormonal Regulation of Protein
Functions as an “Energy Currency”
Metabolism
857
in Metabolism
881
Phosphocreatine Functions as an
Accessory Storage Depot for Energy
C H A P T E R
7
0
and as an “ATP Buffer”
882
The Liver as an Organ
859
Anaerobic Versus Aerobic Energy
882
Physiologic Anatomy of the Liver
859
Summary of Energy Utilization by the
Hepatic Vascular and Lymph
Cells
883
Systems
859
Control of Energy Release in the Cell
884
Blood Flows Through the Liver from the
Metabolic Rate
884
Portal Vein and Hepatic Artery
860
Measurement of the Whole-Body Metabolic
The Liver Functions as a Blood Reservoir
860
Rate
885
The Liver Has Very High Lymph Flow
860
Energy Metabolism—Factors That
Regulation of Liver Mass—Regeneration
860
Influence Energy Output
885
Hepatic Macrophage System Serves a
Overall Energy Requirements for Daily
Blood-Cleansing Function
861
Activities
885
Metabolic Functions of the Liver
861
Basal Metabolic Rate (BMR)—The
Carbohydrate Metabolism
861
Minimum Energy Expenditure for the
Fat Metabolism
861
Body to Exist
886
Protein Metabolism
862
Energy Used for Physical Activities
887
Other Metabolic Functions of the Liver
862
Energy Used for Processing Food—
Measurement of Bilirubin in the Bile
Thermogenic Effect of Food
887
as a Clinical Diagnostic Tool
862
Energy Used for Nonshivering
Jaundice—Excess Bilirubin in the
Thermogenesis—Role of Sympathetic
Extracellular Fluid
863
Stimulation
887
xxxii
Table of Contents
C H A P T E R
7
3
Growth Hormone Promotes Growth of
Many Body Tissues
922
Body Temperature, Temperature
Growth Hormone Has Several Metabolic
Regulation, and Fever
889
Effects
922
Normal Body Temperatures
889
Growth Hormone Stimulates Cartilage and
Body Temperature Is Controlled by
Bone Growth
923
Balancing Heat Production Against
Growth Hormone Exerts Much of Its Effect
Heat Loss
889
Through Intermediate Substances Called
Heat Production
889
“Somatomedins” (Also Called “Insulin-Like
Heat Loss
890
Growth Factors”)
923
Regulation of Body Temperature—Role
Regulation of Growth Hormone Secretion
924
of the Hypothalamus
894
Abnormalities of Growth Hormone Secretion
926
Neuronal Effector Mechanisms That
Posterior Pituitary Gland and Its
Decrease or Increase Body Temperature
895
Relation to the Hypothalamus
927
Concept of a “Set-Point” for Temperature
Chemical Structures of ADH and Oxytocin
928
Control
896
Physiological Functions of ADH
928
Behavioral Control of Body Temperature
897
Oxytocic Hormone
929
Abnormalities of Body Temperature
Regulation
898
C H A P T E R
7
6
Fever
898
Exposure of the Body to Extreme Cold
900
Thyroid Metabolic Hormones
931
Synthesis and Secretion of the Thyroid
Metabolic Hormones
931
Iodine Is Required for Formation of
U N I T X I V
Thyroxine
931
Iodide Pump (Iodide Trapping)
932
Endocrinology and Reproduction
Thyroglobulin, and Chemistry of Thyroxine
and Triiodothyronine Formation
932
Release of Thyroxine and Triiodothyronine
C H A P T E R
7
4
from the Thyroid Gland
933
Introduction to Endocrinology
905
Transport of Thyroxine and Triiodothyronine
to Tissues
934
Coordination of Body Functions by
Physiologic Functions of the Thyroid
Chemical Messengers
905
Hormones
934
Chemical Structure and Synthesis of
Thyroid Hormones Increase the
Hormones
906
Transcription of Large Numbers of Genes
934
Hormone Secretion, Transport, and
Thyroid Hormones Increase Cellular
Clearance from the Blood
908
Metabolic Activity
934
Feedback Control of Hormone Secretion
909
Effect of Thyroid Hormone on Growth
936
Transport of Hormones in the Blood
909
Effects of Thyroid Hormone on Specific
“Clearance” of Hormones from the Blood
909
Bodily Mechanisms
936
Mechanisms of Action of Hormones
910
Regulation of Thyroid Hormone
Hormone Receptors and Their Activation
910
Secretion
938
Intracellular Signaling After Hormone
Anterior Pituitary Secretion of TSH Is
Receptor Activation
910
Regulated by Thyrotropin-Releasing
Second Messenger Mechanisms for
Hormone from the Hypothalamus
938
Mediating Intracellular Hormonal
Feedback Effect of Thyroid Hormone to
Functions
912
Decrease Anterior Pituitary Secretion
Hormones That Act Mainly on the Genetic
of TSH
939
Machinery of the Cell
915
Diseases of the Thyroid
940
Measurement of Hormone
Hyperthyroidism
940
Concentrations in the Blood
915
Symptoms of Hyperthyroidism
940
Radioimmunoassay
915
Hypothyroidism
941
Enzyme-Linked Immunosorbent Assay
Cretinism
942
(ELISA)
916
C H A P T E R
7
7
C H A P T E R
7
5
Adrenocortical Hormones
944
Pituitary Hormones and Their Control
Synthesis and Secretion of
by the Hypothalamus
918
Adrenocortical Hormones
944
Pituitary Gland and Its Relation to the
Functions of the Mineralocorticoids-
Hypothalamus
918
Aldosterone
947
Hypothalamus Controls Pituitary
Renal and Circulatory Effects of
Secretion
919
Aldosterone
948
Hypothalamic-Hypophysial Portal Blood
Aldosterone Stimulates Sodium and
Vessels of the Anterior Pituitary Gland
920
Potassium Transport in Sweat Glands,
Physiological Functions of Growth
Salivary Glands, and Intestinal Epithelial
Hormone
921
Cells
949
Table of Contents
xxxiii
Cellular Mechanism of Aldosterone Action
950
Calcium in the Plasma and Interstitial
Possible Nongenomic Actions of
Fluid
978
Aldosterone and Other Steroid Hormones
950
Inorganic Phosphate in the Extracellular
Regulation of Aldosterone Secretion
950
Fluids
979
Functions of the Glucocorticoids
950
Non-Bone Physiologic Effects of Altered
Effects of Cortisol on Carbohydrate
Calcium and Phosphate Concentrations
Metabolism
951
in the Body Fluids
979
Effects of Cortisol on Protein Metabolism
952
Absorption and Excretion of Calcium and
Effects of Cortisol on Fat Metabolism
952
Phosphate
980
Cortisol Is Important in Resisting Stress
Bone and Its Relation to Extracellular
and Inflammation
952
Calcium and Phosphate
980
Other Effects of Cortisol
954
Precipitation and Absorption of Calcium
Cellular Mechanism of Cortisol Action
954
and Phosphate in Bone—Equilibrium
Regulation of Cortisol Secretion by
with the Extracellular Fluids
981
Adrenocorticotropic Hormone from the
Calcium Exchange Between Bone and
Pituitary Gland
955
Extracellular Fluid
982
Adrenal Androgens
957
Deposition and Absorption of Bone—
Abnormalities of Adrenocortical
Remodeling of Bone
982
Secretion
957
Vitamin D
983
Hypoadrenalism-Addison’s Disease
957
Actions of Vitamin D
985
Hyperadrenalism-Cushing’s Syndrome
958
Parathyroid Hormone
985
Primary Aldosteronism (Conn’s Syndrome)
959
Effect of Parathyroid Hormone on Calcium
Adrenogenital Syndrome
959
and Phosphate Concentrations in the
Extracellular Fluid
986
Control of Parathyroid Secretion by
C H A P T E R
7
8
Calcium Ion Concentration
988
Insulin, Glucagon, and Diabetes
Calcitonin
988
Mellitus
961
Summary of Control of Calcium Ion
Concentration
989
Insulin and Its Metabolic Effects
961
Pathophysiology of Parathyroid
Effect of Insulin on Carbohydrate
Hormone, Vitamin D, and Bone
Metabolism
963
Disease
990
Effect of Insulin on Fat Metabolism
965
Primary Hyperparathyroidism
990
Effect of Insulin on Protein Metabolism
Secondary Parathyroidism
991
and on Growth
966
Rickets—Vitamin D Deficiency
991
Mechanisms of Insulin Secretion
967
Osteoporosis—Decreased Bone Matrix
991
Control of Insulin Secretion
968
Physiology of the Teeth
992
Other Factors That Stimulate Insulin
Function of the Different Parts of the
Secretion
969
Teeth
992
Role of Insulin (and Other Hormones) in
Dentition
993
“Switching” Between Carbohydrate and
Mineral Exchange in Teeth
993
Lipid Metabolism
969
Dental Abnormalities
994
Glucagon and Its Functions
970
Effects on Glucose Metabolism
970
Regulation of Glucagon Secretion
971
C H A P T E R
8
0
Somatostatin Inhibits Glucagon and
Reproductive and Hormonal
Insulin Secretion
971
Functions of the Male (and Function
Summary of Blood Glucose
of the Pineal Gland)
996
Regulation
971
Diabetes Mellitus
972
Physiologic Anatomy of the Male
Type I Diabetes—Lack of Insulin Production
Sexual Organs
996
by Beta Cells of the Pancreas
972
Spermatogenesis
996
Type II Diabetes—Resistance to the
Steps of Spermatogenesis
996
Metabolic Effects of Insulin
974
Function of the Seminal Vesicles
999
Physiology of Diagnosis of Diabetes
Function of the Prostate Gland
999
Mellitus
975
Semen
999
Treatment of Diabetes
976
Male Sexual Act
1001
Insulinoma—Hyperinsulinism
976
Abnormal Spermatogenesis and Male
Fertility
1001
Neuronal Stimulus for Performance of the
C H A P T E R
7
9
Male Sexual Act
1001
Parathyroid Hormone, Calcitonin,
Stages of the Male Sexual Act
1002
Calcium and Phosphate
Testosterone and Other Male Sex
Hormones
1003
Metabolism, Vitamin D, Bone,
Secretion, Metabolism, and Chemistry of
and Teeth
978
the Male Sex Hormone
1003
Overview of Calcium and Phosphate
Functions of Testosterone
1004
Regulation in the Extracellular Fluid
Basic Intracellular Mechanism of Action of
and Plasma
978
Testosterone
1006
xxxiv
Table of Contents
Control of Male Sexual Functions by
Response of the Mother’s Body to
Hormones from the Hypothalamus and
Pregnancy
1034
Anterior Pituitary Gland
1006
Changes in the Maternal Circulatory System
Abnormalities of Male Sexual Function
1008
During Pregnancy
1035
Prostate Gland and Its Abnormalities
1008
Parturition
1036
Hypogonadism in the Male
1008
Increased Uterine Excitability Near Term
1036
Testicular Tumors and Hypergonadism in
Onset of Labor—A Positive Feedback
the Male
1009
Mechanism for Its Initiation
1037
Pineal Gland—Its Function in Controlling
Abdominal Muscle Contractions During
Seasonal Fertility in Some Animals
1009
Labor
1037
Mechanics of Parturition
1037
Separation and Delivery of the Placenta
1038
C H A P T E R
8
1
Labor Pains
1038
Female Physiology Before Pregnancy
Involution of the Uterus After Parturition
1038
and Female Hormones
1011
Lactation
1038
Physiologic Anatomy of the Female
Development of the Breasts
1038
Sexual Organs
1011
Initiation of Lactation—Function of
Female Hormonal System
1011
Prolactin
1039
Monthly Ovarian Cycle; Function of the
Ejection (or “Let-Down”) Process in Milk
Gonadotropic Hormones
1012
Secretion—Function of Oxytocin
1040
Gonadotropic Hormones and Their Effects
Milk Composition and the Metabolic Drain
on the Ovaries
1012
on the Mother Caused by Lactation
1041
Ovarian Follicle Growth—The “Follicular”
Phase of the Ovarian Cycle
1013
C H A P T E R
8
3
Corpus Luteum—“Luteal” Phase of the
Fetal and Neonatal Physiology
1042
Ovarian Cycle
1014
Growth and Functional Development
Summary
1015
of the Fetus
1042
Functions of the Ovarian Hormones—
Development of the Organ Systems
1042
Estradiol and Progesterone
1016
Adjustments of the Infant to
Chemistry of the Sex Hormones
1016
Extrauterine Life
1044
Functions of the Estrogens—Their Effects
Onset of Breathing
1044
on the Primary and Secondary Female Sex
Circulatory Readjustments at Birth
1045
Characteristics
1017
Nutrition of the Neonate
1047
Functions of Progesterone
1018
Special Functional Problems in the
Monthly Endometrial Cycle and Menstruation
1018
Neonate
1047
Regulation of the Female Monthly
Respiratory System
1047
Rhythm—Interplay Between the
Circulation
1047
Ovarian and Hypothalamic-Pituitary
Fluid Balance, Acid-Base Balance, and
Hormones
1019
Renal Function
1048
Feedback Oscillation of the Hypothalamic-
Liver Function
1048
Pituitary-Ovarian System
1021
Digestion, Absorption, and Metabolism
Puberty and Menarche
1021
of Energy Foods; and Nutrition
1048
Menopause
1022
Immunity
1049
Abnormalities of Secretion by the
Endocrine Problems
1049
Ovaries
1023
Special Problems of Prematurity
1050
Female Sexual Act
1023
Immature Development of the Premature
Female Fertility
1024
Infant
1050
Instability of the Homeostatic Control
C H A P T E R
8
2
Systems in the Premature Infant
1050
Pregnancy and Lactation
1027
Danger of Blindness Caused by Excess
Oxygen Therapy in the Premature Infant
1051
Maturation and Fertilization of the Ovum
1027
Growth and Development of the Child
1051
Transport of the Fertilized Ovum in the
Behavioral Growth
1052
Fallopian Tube
1028
Implantation of the Blastocyst in the Uterus
1029
Early Nutrition of the Embryo
1029
Function of the Placenta
1029
U N I T X V
Developmental and Physiologic Anatomy
Sports Physiology
of the Placenta
1029
Hormonal Factors in Pregnancy
1031
Human Chorionic Gonadotropin and Its
Effect to Cause Persistence of the
C H A P T E R
8
4
Corpus Luteum and to Prevent
Sports Physiology
1055
Menstruation
1032
Secretion of Estrogens by the Placenta
1032
Muscles in Exercise
1055
Secretion of Progesterone by the Placenta
1033
Strength, Power, and Endurance of Muscles
1055
Human Chorionic Somatomammotropin
1033
Muscle Metabolic Systems in Exercise
1056
Other Hormonal Factors in Pregnancy
1034
Phosphocreatine-Creatine System
1057
Table of Contents
xxxv
Nutrients Used During Muscle Activity
1059
Body Fluids and Salt in Exercise
1065
Effect of Athletic Training on Muscles and
Drugs and Athletes
1065
Muscle Performance
1060
Body Fitness Prolongs Life
1066
Respiration in Exercise
1061
Cardiovascular System in Exercise
1062
Body Heat in Exercise
1065
Index
1067
U N I T I
Introduction to
Physiology:
The Cell and
General Physiology
1. Functional Organization of the Human
Body and Control of the “Internal Environment”
2. The Cell and Its Functions
3. Genetic Control of Protein Synthesis,
Cell Function, and Cell Reproduction
C H A P T E R
1
Functional Organization of the
Human Body and Control of the
“Internal Environment”
The goal of physiology is to explain the physical and
chemical factors that are responsible for the origin,
development, and progression of life. Each type of
life, from the simple virus to the largest tree or the
complicated human being, has its own functional
characteristics. Therefore, the vast field of physiol-
ogy can be divided into viral physiology, bacterial
physiology, cellular physiology, plant physiology,
human physiology, and many more subdivisions.
Human Physiology. In human physiology, we attempt to explain the specific char-
acteristics and mechanisms of the human body that make it a living being. The
very fact that we remain alive is almost beyond our control, for hunger makes
us seek food and fear makes us seek refuge. Sensations of cold make us look
for warmth. Other forces cause us to seek fellowship and to reproduce. Thus,
the human being is actually an automaton, and the fact that we are sensing,
feeling, and knowledgeable beings is part of this automatic sequence of life;
these special attributes allow us to exist under widely varying conditions.
Cells as the Living Units of the Body
The basic living unit of the body is the cell. Each organ is an aggregate of many
different cells held together by intercellular supporting structures.
Each type of cell is specially adapted to perform one or a few particular
functions. For instance, the red blood cells, numbering 25 trillion in each
human being, transport oxygen from the lungs to the tissues. Although the red
cells are the most abundant of any single type of cell in the body, there are
about 75 trillion additional cells of other types that perform functions different
from those of the red cell. The entire body, then, contains about 100 trillion
cells.
Although the many cells of the body often differ markedly from one another,
all of them have certain basic characteristics that are alike. For instance, in all
cells, oxygen reacts with carbohydrate, fat, and protein to release the energy
required for cell function. Further, the general chemical mechanisms for chang-
ing nutrients into energy are basically the same in all cells, and all cells deliver
end products of their chemical reactions into the surrounding fluids.
Almost all cells also have the ability to reproduce additional cells of their
own kind. Fortunately, when cells of a particular type are destroyed from one
cause or another, the remaining cells of this type usually generate new cells until
the supply is replenished.
Extracellular Fluid—The “Internal Environment”
About 60 per cent of the adult human body is fluid, mainly a water solution of
ions and other substances. Although most of this fluid is inside the cells and is
called intracellular fluid, about one third is in the spaces outside the cells and
3
4
Unit I Introduction to Physiology: The Cell and General Physiology
is called extracellular fluid. This extracellular fluid is in
Extracellular Fluid Transport and
constant motion throughout the body. It is transported
Mixing System—The Blood
rapidly in the circulating blood and then mixed
Circulatory System
between the blood and the tissue fluids by diffusion
through the capillary walls.
Extracellular fluid is transported through all parts of
In the extracellular fluid are the ions and nutrients
the body in two stages. The first stage is movement of
needed by the cells to maintain cell life. Thus, all cells
blood through the body in the blood vessels, and the
live in essentially the same environment—the extra-
second is movement of fluid between the blood capil-
cellular fluid. For this reason, the extracellular fluid is
laries and the intercellular spaces between the tissue
also called the internal environment of the body, or the
cells.
milieu intérieur, a term introduced more than 100 years
Figure 1-1 shows the overall circulation of blood.
ago by the great 19th-century French physiologist
All the blood in the circulation traverses the entire cir-
Claude Bernard.
culatory circuit an average of once each minute when
Cells are capable of living, growing, and performing
the body is at rest and as many as six times each minute
their special functions as long as the proper concen-
when a person is extremely active.
trations of oxygen, glucose, different ions, amino acids,
As blood passes through the blood capillaries,
fatty substances, and other constituents are available
continual exchange of extracellular fluid also occurs
in this internal environment.
between the plasma portion of the blood and the
Differences Between Extracellular and Intracellular Fluids.
The extracellular fluid contains large amounts of
Lungs
sodium, chloride, and bicarbonate ions plus nutrients
for the cells, such as oxygen, glucose, fatty acids, and
amino acids. It also contains carbon dioxide that is
being transported from the cells to the lungs to be
excreted, plus other cellular waste products that are
being transported to the kidneys for excretion.
The intracellular fluid differs significantly from
O2
CO2
the extracellular fluid; specifically, it contains large
amounts of potassium, magnesium, and phosphate ions
Right
Left
instead of the sodium and chloride ions found in the
heart
heart
pump
pump
extracellular fluid. Special mechanisms for transport-
ing ions through the cell membranes maintain the ion
concentration differences between the extracellular
Gut
and intracellular fluids. These transport processes are
discussed in Chapter 4.
“Homeostatic” Mechanisms of
Nutrition and excretion
the Major Functional Systems
Kidneys
Homeostasis
The term homeostasis is used by physiologists to mean
maintenance of nearly constant conditions in the inter-
nal environment. Essentially all organs and tissues of
Regulation
Excretion
the body perform functions that help maintain these
of
constant conditions. For instance, the lungs provide
electrolytes
oxygen to the extracellular fluid to replenish the
oxygen used by the cells, the kidneys maintain con-
Venous
Arterial
stant ion concentrations, and the gastrointestinal
end
end
system provides nutrients.
A large segment of this text is concerned with the
manner in which each organ or tissue contributes to
homeostasis. To begin this discussion, the different
functional systems of the body and their contributions
Capillaries
to homeostasis are outlined in this chapter; then we
briefly outline the basic theory of the body’s control
Figure 1-1
systems that allow the functional systems to operate in
support of one another.
General organization of the circulatory system.
Chapter 1
Functional Organization of the Human Body and Control of the “Internal Environment”
5
Arteriole
the gastrointestinal tract. Here different dissolved
nutrients, including carbohydrates, fatty acids, and
amino acids, are absorbed from the ingested food into
the extracellular fluid of the blood.
Liver and Other Organs That Perform Primarily Metabolic Func-
tions. Not all substances absorbed from the gastroin-
testinal tract can be used in their absorbed form by the
cells. The liver changes the chemical compositions of
many of these substances to more usable forms, and
other tissues of the body—fat cells, gastrointestinal
Venule
mucosa, kidneys, and endocrine glands—help modify
the absorbed substances or store them until they are
needed.
Musculoskeletal System. Sometimes the question is
asked, How does the musculoskeletal system fit into
Figure 1-2
the homeostatic functions of the body? The answer is
Diffusion of fluid and dissolved constituents through the capillary
obvious and simple: Were it not for the muscles, the
walls and through the interstitial spaces.
body could not move to the appropriate place at the
appropriate time to obtain the foods required for
nutrition. The musculoskeletal system also provides
motility for protection against adverse surroundings,
interstitial fluid that fills the intercellular spaces. This
without which the entire body, along with its homeo-
process is shown in Figure 1-2. The walls of the capil-
static mechanisms, could be destroyed instantaneously.
laries are permeable to most molecules in the plasma
of the blood, with the exception of the large plasma
protein molecules. Therefore, large amounts of fluid
Removal of Metabolic End Products
and its dissolved constituents diffuse back and forth
between the blood and the tissue spaces, as shown by
Removal of Carbon Dioxide by the Lungs. At the same time
the arrows. This process of diffusion is caused by
that blood picks up oxygen in the lungs, carbon dioxide
kinetic motion of the molecules in both the plasma and
is released from the blood into the lung alveoli; the res-
the interstitial fluid. That is, the fluid and dissolved
piratory movement of air into and out of the lungs
molecules are continually moving and bouncing in all
carries the carbon dioxide to the atmosphere. Carbon
directions within the plasma and the fluid in the inter-
dioxide is the most abundant of all the end products
cellular spaces, and also through the capillary pores.
of metabolism.
Few cells are located more than 50 micrometers from
a capillary, which ensures diffusion of almost any sub-
Kidneys. Passage of the blood through the kidneys
stance from the capillary to the cell within a few
removes from the plasma most of the other substances
seconds. Thus, the extracellular fluid everywhere in the
besides carbon dioxide that are not needed by the
body—both that of the plasma and that of the inter-
cells. These substances include different end products
stitial fluid—is continually being mixed, thereby
of cellular metabolism, such as urea and uric acid; they
maintaining almost complete homogeneity of the
also include excesses of ions and water from the food
extracellular fluid throughout the body.
that might have accumulated in the extracellular fluid.
The kidneys perform their function by first filtering
large quantities of plasma through the glomeruli into
Origin of Nutrients in the
the tubules and then reabsorbing into the blood those
Extracellular Fluid
substances needed by the body, such as glucose, amino
acids, appropriate amounts of water, and many of the
Respiratory System. Figure 1-1 shows that each time the
ions. Most of the other substances that are not needed
blood passes through the body, it also flows through
by the body, especially the metabolic end products
the lungs. The blood picks up oxygen in the alveoli,
such as urea, are reabsorbed poorly and pass through
thus acquiring the oxygen needed by the cells. The
the renal tubules into the urine.
membrane between the alveoli and the lumen of the
pulmonary capillaries, the alveolar membrane, is only
0.4 to 2.0 micrometers thick, and oxygen diffuses by
Regulation of Body Functions
molecular motion through the pores of this membrane
into the blood in the same manner that water and ions
Nervous System. The nervous system is composed of
diffuse through walls of the tissue capillaries.
three major parts: the sensory input portion, the central
nervous system (or integrative portion), and the motor
Gastrointestinal Tract. A large portion of the blood
output portion. Sensory receptors detect the state of
pumped by the heart also passes through the walls of
the body or the state of the surroundings. For instance,
6
Unit I Introduction to Physiology: The Cell and General Physiology
receptors in the skin apprise one whenever an object
concentration of carbon dioxide in the extracellular
touches the skin at any point. The eyes are sensory
fluid. The liver and pancreas regulate the concentra-
organs that give one a visual image of the surrounding
tion of glucose in the extracellular fluid, and the
area. The ears also are sensory organs. The central
kidneys regulate concentrations of hydrogen, sodium,
nervous system is composed of the brain and spinal
potassium, phosphate, and other ions in the extracel-
cord. The brain can store information, generate
lular fluid.
thoughts, create ambition, and determine reactions
that the body performs in response to the sensations.
Appropriate signals are then transmitted through the
Examples of Control Mechanisms
motor output portion of the nervous system to carry
out one’s desires.
Regulation of Oxygen and Carbon Dioxide Concentrations in the
A large segment of the nervous system is called the
Extracellular Fluid. Because oxygen is one of the major
autonomic system. It operates at a subconscious level
substances required for chemical reactions in the cells,
and controls many functions of the internal organs,
it is fortunate that the body has a special control
including the level of pumping activity by the heart,
mechanism to maintain an almost exact and constant
movements of the gastrointestinal tract, and secretion
oxygen concentration in the extracellular fluid. This
by many of the body’s glands.
mechanism depends principally on the chemical char-
acteristics of hemoglobin, which is present in all red
Hormonal System of Regulation. Located in the body are
blood cells. Hemoglobin combines with oxygen as the
eight major endocrine glands that secrete chemical
blood passes through the lungs. Then, as the blood
substances called hormones. Hormones are trans-
passes through the tissue capillaries, hemoglobin,
ported in the extracellular fluid to all parts of the body
because of its own strong chemical affinity for oxygen,
to help regulate cellular function. For instance, thyroid
does not release oxygen into the tissue fluid if too
hormone increases the rates of most chemical reac-
much oxygen is already there. But if the oxygen con-
tions in all cells, thus helping to set the tempo of bodily
centration in the tissue fluid is too low, sufficient
activity. Insulin controls glucose metabolism; adreno-
oxygen is released to re-establish an adequate con-
cortical hormones control sodium ion, potassium ion,
centration. Thus, regulation of oxygen concentration
and protein metabolism; and parathyroid hormone
in the tissues is vested principally in the chemical
controls bone calcium and phosphate. Thus, the hor-
characteristics of hemoglobin itself. This regulation is
mones are a system of regulation that complements
called the oxygen-buffering function of hemoglobin.
the nervous system. The nervous system regulates
Carbon dioxide concentration in the extracellular
mainly muscular and secretory activities of the body,
fluid is regulated in a much different way. Carbon
whereas the hormonal system regulates many meta-
dioxide is a major end product of the oxidative reac-
bolic functions.
tions in cells. If all the carbon dioxide formed in the
cells continued to accumulate in the tissue fluids, the
mass action of the carbon dioxide itself would soon
Reproduction
halt all energy-giving reactions of the cells. Fortu-
nately, a higher than normal carbon dioxide concen-
Sometimes reproduction is not considered a homeo-
tration in the blood excites the respiratory center,
static function. It does, however, help maintain home-
causing a person to breathe rapidly and deeply. This
ostasis by generating new beings to take the place of
increases expiration of carbon dioxide and, therefore,
those that are dying. This may sound like a permissive
removes excess carbon dioxide from the blood and
usage of the term homeostasis, but it illustrates that, in
tissue fluids. This process continues until the concen-
the final analysis, essentially all body structures are
tration returns to normal.
organized such that they help maintain the automatic-
ity and continuity of life.
Regulation of Arterial Blood Pressure. Several systems con-
tribute to the regulation of arterial blood pressure.
One of these, the baroreceptor system, is a simple and
Control Systems of the Body
excellent example of a rapidly acting control mecha-
nism. In the walls of the bifurcation region of the
The human body has thousands of control systems in
carotid arteries in the neck, and also in the arch of the
it. The most intricate of these are the genetic control
aorta in the thorax, are many nerve receptors called
systems that operate in all cells to help control intra-
baroreceptors, which are stimulated by stretch of the
cellular function as well as extracellular function. This
arterial wall. When the arterial pressure rises too high,
subject is discussed in Chapter 3.
the baroreceptors send barrages of nerve impulses to
Many other control systems operate within the
the medulla of the brain. Here these impulses inhibit
organs to control functions of the individual parts
the vasomotor center, which in turn decreases the
of the organs; others operate throughout the entire
number of impulses transmitted from the vasomotor
body to control the interrelations between the organs.
center through the sympathetic nervous system to the
For instance, the respiratory system, operating in
heart and blood vessels. Lack of these impulses causes
association with the nervous system, regulates the
diminished pumping activity by the heart and also
Chapter 1
Functional Organization of the Human Body and Control of the “Internal Environment”
7
dilation of the peripheral blood vessels, allowing
vast numbers of control systems that keep the body
increased blood flow through the vessels. Both of these
operating in health; in the absence of any one of these
effects decrease the arterial pressure back toward
controls, serious body malfunction or death can result.
normal.
Conversely, a decrease in arterial pressure below
Characteristics of Control Systems
normal relaxes the stretch receptors, allowing the
vasomotor center to become more active than usual,
The aforementioned examples of homeostatic control
thereby causing vasoconstriction and increased heart
mechanisms are only a few of the many thousands in
pumping, and raising arterial pressure back toward
the body, all of which have certain characteristics in
normal.
common. These characteristics are explained in this
section.
Normal Ranges and Physical Characteristics
of Important Extracellular Fluid Constituents
Negative Feedback Nature of Most
Table 1-1 lists the more important constituents and
Control Systems
physical characteristics of extracellular fluid, along
Most control systems of the body act by negative feed-
with their normal values, normal ranges, and maximum
back, which can best be explained by reviewing some
limits without causing death. Note the narrowness of
of the homeostatic control systems mentioned pre-
the normal range for each one. Values outside these
viously. In the regulation of carbon dioxide concen-
ranges are usually caused by illness.
tration, a high concentration of carbon dioxide in the
Most important are the limits beyond which abnor-
extracellular fluid increases pulmonary ventilation.
malities can cause death. For example, an increase
This, in turn, decreases the extracellular fluid carbon
in the body temperature of only 11°F (7°C) above
dioxide concentration because the lungs expire greater
normal can lead to a vicious cycle of increasing cellu-
amounts of carbon dioxide from the body. In other
lar metabolism that destroys the cells. Note also the
words, the high concentration of carbon dioxide initi-
narrow range for acid-base balance in the body, with
ates events that decrease the concentration toward
a normal pH value of 7.4 and lethal values only
normal, which is negative to the initiating stimulus.
about 0.5 on either side of normal. Another important
Conversely, if the carbon dioxide concentration falls
factor is the potassium ion concentration, because
too low, this causes feedback to increase the concen-
whenever it decreases to less than one third normal,
tration. This response also is negative to the initiating
a person is likely to be paralyzed as a result of the
stimulus.
nerves’ inability to carry signals. Alternatively, if
In the arterial pressure-regulating mechanisms, a
the potassium ion concentration increases to two or
high pressure causes a series of reactions that promote
more times normal, the heart muscle is likely to be
a lowered pressure, or a low pressure causes a series
severely depressed. Also, when the calcium ion con-
of reactions that promote an elevated pressure. In both
centration falls below about one half of normal, a
instances, these effects are negative with respect to the
person is likely to experience tetanic contraction of
initiating stimulus.
muscles throughout the body because of the sponta-
Therefore, in general, if some factor becomes exces-
neous generation of excess nerve impulses in the
sive or deficient, a control system initiates negative
peripheral nerves. When the glucose concentration
feedback, which consists of a series of changes that
falls below one half of normal, a person frequently
return the factor toward a certain mean value, thus
develops extreme mental irritability and sometimes
maintaining homeostasis.
even convulsions.
These examples should give one an appreciation for
“Gain” of a Control System. The degree of effectiveness
the extreme value and even the necessity of the
with which a control system maintains constant
Table 1-1
Important Constituents and Physical Characteristics of Extracellular Fluid
Normal Value
Normal Range
Approximate Short-Term
Unit
Nonlethal Limit
Oxygen
40
35-45
10-1000
mm Hg
Carbon dioxide
40
35-45
5-80
mm Hg
Sodium ion
142
138-146
115-175
mmol/L
Potassium ion
4.2
3.8-5.0
1.5-9.0
mmol/L
Calcium ion
1.2
1.0-1.4
0.5-2.0
mmol/L
Chloride ion
108
103-112
70-130
mmol/L
Bicarbonate ion
28
24-32
8-45
mmol/L
Glucose
85
75-95
20-1500
mg/dl
Body temperature
98.4
(37.0)
98-98.8 (37.0)
65-110 (18.3-43.3)
∞F (∞C)
Acid-base
7.4
7.3-7.5
6.9-8.0
pH
8
Unit I Introduction to Physiology: The Cell and General Physiology
conditions is determined by the gain of the negative
feedback. For instance, let us assume that a large
volume of blood is transfused into a person whose
5
baroreceptor pressure control system is not function-
Return to
ing, and the arterial pressure rises from the normal
4
normal
level of 100 mm Hg up to 175 mm Hg. Then, let us
Bled 1 liter
assume that the same volume of blood is injected into
the same person when the baroreceptor system is func-
3
tioning, and this time the pressure increases only
25 mm Hg. Thus, the feedback control system has
Bled 2 liters
2
caused a “correction” of -50 mm Hg—that is, from
175 mm Hg to 125 mm Hg. There remains an increase
in pressure of +25 mm Hg, called the “error,” which
1
means that the control system is not 100 per cent effec-
tive in preventing change. The gain of the system is
Death
0
then calculated by the following formula:
1
2
3
Hours
Correction
Gain
=
Error
Figure 1-3
Thus, in the baroreceptor system example, the correc-
tion is -50 mm Hg and the error persisting is +25 mm
Recovery of heart pumping caused by negative feedback after 1
Hg. Therefore, the gain of the person’s baroreceptor
liter of blood is removed from the circulation. Death is caused by
positive feedback when 2 liters of blood are removed.
system for control of arterial pressure is -50 divided
by +25, or -2. That is, a disturbance that increases or
decreases the arterial pressure does so only one third
as much as would occur if this control system were not
present.
Positive feedback is better known as a “vicious
The gains of some other physiologic control systems
cycle,” but a mild degree of positive feedback can be
are much greater than that of the baroreceptor
overcome by the negative feedback control mecha-
system. For instance, the gain of the system controlling
nisms of the body, and the vicious cycle fails to
internal body temperature when a person is exposed
develop. For instance, if the person in the aforemen-
to moderately cold weather is about -33. Therefore,
tioned example were bled only 1 liter instead of 2
one can see that the temperature control system is
liters, the normal negative feedback mechanisms for
much more effective than the baroreceptor pressure
controlling cardiac output and arterial pressure would
control system.
overbalance the positive feedback and the person
would recover, as shown by the dashed curve of
Positive Feedback Can Sometimes Cause
Figure 1-3.
Vicious Cycles and Death
One might ask the question, Why do essentially all
control systems of the body operate by negative feed-
Positive Feedback Can Sometimes Be Useful. In some
back rather than positive feedback? If one considers
instances, the body uses positive feedback to its advan-
the nature of positive feedback, one immediately sees
tage. Blood clotting is an example of a valuable use of
that positive feedback does not lead to stability but to
positive feedback. When a blood vessel is ruptured
instability and often death.
and a clot begins to form, multiple enzymes called clot-
Figure 1-3 shows an example in which death can
ting factors are activated within the clot itself. Some
ensue from positive feedback. This figure depicts the
of these enzymes act on other unactivated enzymes
pumping effectiveness of the heart, showing that the
of the immediately adjacent blood, thus causing
heart of a healthy human being pumps about 5 liters
more blood clotting. This process continues until the
of blood per minute. If the person is suddenly bled 2
hole in the vessel is plugged and bleeding no longer
liters, the amount of blood in the body is decreased to
occurs. On occasion, this mechanism can get out of
such a low level that not enough blood is available for
hand and cause the formation of unwanted clots.
the heart to pump effectively. As a result, the arterial
In fact, this is what initiates most acute heart
pressure falls, and the flow of blood to the heart muscle
attacks, which are caused by a clot beginning on the
through the coronary vessels diminishes. This results in
inside surface of an atherosclerotic plaque in a coro-
weakening of the heart, further diminished pumping,
nary artery and then growing until the artery is
a further decrease in coronary blood flow, and still
blocked.
more weakness of the heart; the cycle repeats itself
Childbirth is another instance in which positive
again and again until death occurs. Note that each
feedback plays a valuable role. When uterine contrac-
cycle in the feedback results in further weakening of
tions become strong enough for the baby’s head to
the heart. In other words, the initiating stimulus causes
begin pushing through the cervix, stretch of the cervix
more of the same, which is positive feedback.
sends signals through the uterine muscle back to the
Chapter 1
Functional Organization of the Human Body and Control of the “Internal Environment”
9
body of the uterus, causing even more powerful
Summary—Automaticity
contractions. Thus, the uterine contractions stretch
of the Body
the cervix, and the cervical stretch causes stronger
contractions. When this process becomes powerful
The purpose of this chapter has been to point out, first,
enough, the baby is born. If it is not powerful enough,
the overall organization of the body and, second, the
the contractions usually die out, and a few days pass
means by which the different parts of the body operate
before they begin again.
in harmony. To summarize, the body is actually a social
Another important use of positive feedback is
order of about 100 trillion cells organized into differ-
for the generation of nerve signals. That is, when
ent functional structures, some of which are called
the membrane of a nerve fiber is stimulated, this
organs. Each functional structure contributes its share
causes slight leakage of sodium ions through sodium
to the maintenance of homeostatic conditions in the
channels in the nerve membrane to the fiber’s interior.
extracellular fluid, which is called the internal envi-
The sodium ions entering the fiber then change
ronment. As long as normal conditions are maintained
the membrane potential, which in turn causes more
in this internal environment, the cells of the body con-
opening of channels, more change of potential,
tinue to live and function properly. Each cell benefits
still more opening of channels, and so forth. Thus, a
from homeostasis, and in turn, each cell contributes its
slight leak becomes an explosion of sodium entering
share toward the maintenance of homeostasis. This
the interior of the nerve fiber, which creates the
reciprocal interplay provides continuous automaticity
nerve action potential. This action potential in turn
of the body until one or more functional systems lose
causes electrical current to flow along both the outside
their ability to contribute their share of function. When
and the inside of the fiber and initiates additional
this happens, all the cells of the body suffer. Extreme
action potentials. This process continues again and
dysfunction leads to death; moderate dysfunction
again until the nerve signal goes all the way to the end
leads to sickness.
of the fiber.
In each case in which positive feedback is useful, the
positive feedback itself is part of an overall negative
feedback process. For example, in the case of blood
References
clotting, the positive feedback clotting process is a
negative feedback process for maintenance of normal
Adolph EF: Physiological adaptations: hypertrophies and
blood volume. Also, the positive feedback that causes
superfunctions. Am Sci 60:608, 1972.
nerve signals allows the nerves to participate in
Bernard C: Lectures on the Phenomena of Life Common to
thousands of negative feedback nervous control
Animals and Plants. Springfield, IL: Charles C Thomas,
systems.
1974.
Cabanac M: Regulation and the ponderostat. Int J Obes
Relat Metab Disord 25(Suppl 5):S7, 2001.
Cannon WB: The Wisdom of the Body. New York: WW
More Complex Types of Control Systems—
Norton, 1932.
Adaptive Control
Conn PM, Goodman HM: Handbook of Physiology: Cellu-
Later in this text, when we study the nervous system,
lar Endocrinology. Bethesda: American Physiological
we shall see that this system contains great numbers
Society, 1997.
of interconnected control mechanisms. Some are
Csete ME, Doyle JC: Reverse engineering of biological com-
simple feedback systems similar to those already
plexity. Science 295:1664, 2002.
discussed. Many are not. For instance, some move-
Danzler WH (ed): Handbook of Physiology, Sec 13: Com-
ments of the body occur so rapidly that there is not
parative Physiology. Bethesda: American Physiological
enough time for nerve signals to travel from the
Society, 1997.
peripheral parts of the body all the way to the brain
Dickinson MH, Farley CT, Full RJ, et al: How animals move:
an integrative view. Science 288:100, 2000.
and then back to the periphery again to control the
Garland T Jr, Carter PA: Evolutionary physiology. Annu Rev
movement. Therefore, the brain uses a principle called
Physiol 56:579, 1994.
feed-forward control to cause required muscle con-
Gelehrter TD, Collins FS: Principles of Medical Genetics.
tractions. That is, sensory nerve signals from the
Baltimore: Williams & Wilkins, 1995.
moving parts apprise the brain whether the movement
Guyton AC: Arterial Pressure and Hypertension. Philadel-
is performed correctly. If not, the brain corrects the
phia: WB Saunders, 1980.
feed-forward signals that it sends to the muscles the
Guyton AC, Jones CE, Coleman TG: Cardiac Output and Its
next time the movement is required. Then, if still
Regulation. Philadelphia: WB Saunders, 1973.
further correction is needed, this will be done again for
Guyton AC, Taylor AE, Granger HJ: Dynamics and
subsequent movements. This is called adaptive control.
Control of the Body Fluids. Philadelphia: WB Saunders,
1975.
Adaptive control, in a sense, is delayed negative
Hoffman JF, Jamieson JD: Handbook of Physiology: Cell
feedback.
Physiology. Bethesda: American Physiological Society,
Thus, one can see how complex the feedback
1997.
control systems of the body can be. A person’s life
Krahe R, Gabbiani F: Burst firing in sensory systems. Nat
depends on all of them. Therefore, a major share of
Rev Neurosci 5:13, 2004.
this text is devoted to discussing these life-giving
Lewin B: Genes VII. New York: Oxford University Press,
mechanisms.
2000.
10
Unit I Introduction to Physiology: The Cell and General Physiology
Masoro EJ (ed): Handbook of Physiology, Sec 11: Aging.
Smith HW: From Fish to Philosopher. New York: Doubleday,
Bethesda: American Physiological Society, 1995.
1961.
Milhorn HT: The Application of Control Theory to
Thomson RC: Biomaterials Regulating Cell Function and
Physiological Systems. Philadelphia: WB Saunders,
Tissue Development. Warrendale, PA: Materials Research
1966.
Society, 1998.
Orgel LE: The origin of life on the earth. Sci Am 271:76,
Tjian R: Molecular machines that control genes. Sci Am
1994.
272:54, 1995.
C H A P T E R
2
The Cell and Its Functions
Each of the 100 trillion cells in a human being is a
living structure that can survive for months or many
years, provided its surrounding fluids contain appro-
priate nutrients. To understand the function of
organs and other structures of the body, it is essen-
tial that we first understand the basic organization
of the cell and the functions of its component
parts.
Organization of the Cell
A typical cell, as seen by the light microscope, is shown in Figure 2-1. Its two
major parts are the nucleus and the cytoplasm. The nucleus is separated from
the cytoplasm by a nuclear membrane, and the cytoplasm is separated from the
surrounding fluids by a cell membrane, also called the plasma membrane.
The different substances that make up the cell are collectively called proto-
plasm. Protoplasm is composed mainly of five basic substances: water, elec-
trolytes, proteins, lipids, and carbohydrates.
Water. The principal fluid medium of the cell is water, which is present in most
cells, except for fat cells, in a concentration of 70 to 85 per cent. Many cellular
chemicals are dissolved in the water. Others are suspended in the water as solid
particulates. Chemical reactions take place among the dissolved chemicals or at
the surfaces of the suspended particles or membranes.
Ions. The most important ions in the cell are potassium, magnesium, phosphate,
sulfate, bicarbonate, and smaller quantities of sodium, chloride, and calcium.
These are all discussed in more detail in Chapter 4, which considers the inter-
relations between the intracellular and extracellular fluids.
The ions provide inorganic chemicals for cellular reactions. Also, they are
necessary for operation of some of the cellular control mechanisms. For
instance, ions acting at the cell membrane are required for transmission of elec-
trochemical impulses in nerve and muscle fibers.
Proteins. After water, the most abundant substances in most cells are proteins,
which normally constitute 10 to 20 per cent of the cell mass. These can be
divided into two types: structural proteins and functional proteins.
Structural proteins are present in the cell mainly in the form of long filaments
that themselves are polymers of many individual protein molecules. A promi-
nent use of such intracellular filaments is to form microtubules that provide the
“cytoskeletons” of such cellular organelles as cilia, nerve axons, the mitotic
spindles of mitosing cells, and a tangled mass of thin filamentous tubules that
hold the parts of the cytoplasm and nucleoplasm together in their respective
compartments. Extracellularly, fibrillar proteins are found especially in the col-
lagen and elastin fibers of connective tissue and in blood vessel walls, tendons,
ligaments, and so forth.
The functional proteins are an entirely different type of protein, usually com-
posed of combinations of a few molecules in tubular-globular form. These
11
12
Unit I Introduction to Physiology: The Cell and General Physiology
surrounding extracellular fluid so that it is readily
available to the cell. Also, a small amount of carbohy-
Cell
drate is virtually always stored in the cells in the form
membrane
of glycogen, which is an insoluble polymer of glucose
Cytoplasm
that can be depolymerized and used rapidly to supply
Nucleolus
the cells’ energy needs.
Nucleoplasm
Nucleus
Nuclear
membrane
Physical Structure of the Cell
The cell is not merely a bag of fluid, enzymes, and
chemicals; it also contains highly organized physical
Figure 2-1
structures, called intracellular organelles. The physical
nature of each organelle is as important as the cell’s
Structure of the cell as seen with the light microscope.
chemical constituents for cell function. For instance,
without one of the organelles, the mitochondria, more
than 95 per cent of the cell’s energy release from nutri-
ents would cease immediately. The most important
organelles and other structures of the cell are shown
proteins are mainly the enzymes of the cell and, in con-
in Figure 2-2.
trast to the fibrillar proteins, are often mobile in the
cell fluid. Also, many of them are adherent to mem-
branous structures inside the cell. The enzymes come
Membranous Structures of the Cell
into direct contact with other substances in the cell
fluid and thereby catalyze specific intracellular chem-
Most organelles of the cell are covered by membranes
ical reactions. For instance, the chemical reactions
composed primarily of lipids and proteins. These mem-
that split glucose into its component parts and then
branes include the cell membrane, nuclear membrane,
combine these with oxygen to form carbon dioxide
membrane of the endoplasmic reticulum, and mem-
and water while simultaneously providing energy for
branes of the mitochondria, lysosomes, and Golgi
cellular function are all catalyzed by a series of protein
apparatus.
enzymes.
The lipids of the membranes provide a barrier that
impedes the movement of water and water-soluble
substances from one cell compartment to another
Lipids. Lipids are several types of substances that are
because water is not soluble in lipids. However, protein
grouped together because of their common property
molecules in the membrane often do penetrate all the
of being soluble in fat solvents. Especially important
way through the membrane, thus providing specialized
lipids are phospholipids and cholesterol, which
pathways, often organized into actual pores, for
together constitute only about 2 per cent of the total
passage of specific substances through the membrane.
cell mass. The significance of phospholipids and cho-
Also, many other membrane proteins are enzymes that
lesterol is that they are mainly insoluble in water and,
catalyze a multitude of different chemical reactions,
therefore, are used to form the cell membrane and
discussed here and in subsequent chapters.
intracellular membrane barriers that separate the dif-
ferent cell compartments.
Cell Membrane
In addition to phospholipids and cholesterol, some
The cell membrane (also called the plasma mem-
cells contain large quantities of triglycerides, also
brane), which envelops the cell, is a thin, pliable,
called neutral fat. In the fat cells, triglycerides often
elastic structure only 7.5 to 10 nanometers thick. It
account for as much as 95 per cent of the cell mass. The
is composed almost entirely of proteins and lipids.
fat stored in these cells represents the body’s main
The approximate composition is proteins, 55 per cent;
storehouse of energy-giving nutrients that can later be
phospholipids, 25 per cent; cholesterol, 13 per cent;
dissoluted and used to provide energy wherever in the
other lipids, 4 per cent; and carbohydrates, 3 per cent.
body it is needed.
Lipid Barrier of the Cell Membrane Impedes Water Penetration.
Figure 2-3 shows the structure of the cell membrane.
Carbohydrates. Carbohydrates have little structural
Its basic structure is a lipid bilayer, which is a thin,
function in the cell except as parts of glycoprotein mol-
double-layered film of lipids—each layer only one
ecules, but they play a major role in nutrition of the
molecule thick—that is continuous over the entire cell
cell. Most human cells do not maintain large stores of
surface. Interspersed in this lipid film are large globu-
carbohydrates; the amount usually averages about 1
lar protein molecules.
per cent of their total mass but increases to as much
The basic lipid bilayer is composed of phospholipid
as 3 per cent in muscle cells and, occasionally, 6 per
molecules. One end of each phospholipid molecule is
cent in liver cells. However, carbohydrate in the
soluble in water; that is, it is hydrophilic. The other end
form of dissolved glucose is always present in the
is soluble only in fats; that is, it is hydrophobic. The
Chapter 2
The Cell and Its Functions
13
Chromosomes and DNA
Centrioles
Secretory
granule
Golgi
apparatus
Microtubules
Nuclear
Cell
membrane
membrane
Nucleolus
Glycogen
Ribosomes
Lysosome
Figure 2-2
Reconstruction of a typical
Mitochondrion
Granular
Smooth
Microfilaments
cell, showing the internal
endoplasmic
(agranular)
organelles in the cytoplasm
reticulum
endoplasmic
and in the nucleus.
reticulum
phosphate end of the phospholipid is hydrophilic, and
body fluids. Cholesterol controls much of the fluidity
the fatty acid portion is hydrophobic.
of the membrane as well.
Because the hydrophobic portions of the phospho-
lipid molecules are repelled by water but are mutually
Cell Membrane Proteins. Figure 2-3 also shows globular
attracted to one another, they have a natural tendency
masses floating in the lipid bilayer. These are mem-
to attach to one another in the middle of the mem-
brane proteins, most of which are glycoproteins. Two
brane, as shown in Figure 2-3. The hydrophilic phos-
types of proteins occur: integral proteins that protrude
phate portions then constitute the two surfaces of the
all the way through the membrane, and peripheral pro-
complete cell membrane, in contact with intracellular
teins that are attached only to one surface of the mem-
water on the inside of the membrane and extracellular
brane and do not penetrate all the way through.
water on the outside surface.
Many of the integral proteins provide structural
The lipid layer in the middle of the membrane is
channels (or pores) through which water molecules
impermeable to the usual water-soluble substances,
and water-soluble substances, especially ions, can
such as ions, glucose, and urea. Conversely, fat-soluble
diffuse between the extracellular and intracellular
substances, such as oxygen, carbon dioxide, and
fluids. These protein channels also have selective prop-
alcohol, can penetrate this portion of the membrane
erties that allow preferential diffusion of some sub-
with ease.
stances over others.
The cholesterol molecules in the membrane are also
Other integral proteins act as carrier proteins for
lipid in nature because their steroid nucleus is highly
transporting substances that otherwise could not pen-
fat soluble. These molecules, in a sense, are dissolved
etrate the lipid bilayer. Sometimes these even trans-
in the bilayer of the membrane. They mainly help
port substances in the direction opposite to their
determine the degree of permeability (or imperme-
natural direction of diffusion, which is called “active
ability) of the bilayer to water-soluble constituents of
transport.” Still others act as enzymes.
14
Unit I Introduction to Physiology: The Cell and General Physiology
Carbohydrate
Extracellular
fluid
Integral protein
Lipid
bilayer
Figure 2-3
Peripheral
protein
Structure of the cell membrane,
showing that it is composed
Intracellular
mainly of a lipid bilayer of phos-
fluid
pholipid molecules, but with large
numbers of protein molecules
Cytoplasm
protruding through the layer.
Also, carbohydrate moieties are
attached to the protein molecules
on the outside of the membrane
and to additional protein mole-
cules on the inside.
(Redrawn
Integral protein
from Lodish HF, Rothman JE: The
assembly of cell membranes. Sci
Am
240:48,
1979. Copyright
George V. Kevin.)
Integral membrane proteins can also serve as recep-
cell surface. Many other carbohydrate compounds,
tors for water-soluble chemicals, such as peptide hor-
called proteoglycans—which are mainly carbohydrate
mones, that do not easily penetrate the cell membrane.
substances bound to small protein cores—are loosely
Interaction of cell membrane receptors with specific
attached to the outer surface of the cell as well.
ligands that bind to the receptor causes conforma-
Thus, the entire outside surface of the cell often has a
tional changes in the receptor protein. This, in turn,
loose carbohydrate coat called the glycocalyx.
enzymatically activates the intracellular part of the
The carbohydrate moieties attached to the outer
protein or induces interactions between the receptor
surface of the cell have several important functions:
and proteins in the cytoplasm that act as second mes-
(1) Many of them have a negative electrical charge,
sengers, thereby relaying the signal from the extracel-
which gives most cells an overall negative surface
lular part of the receptor to the interior of the cell. In
charge that repels other negative objects. (2) The gly-
this way, integral proteins spanning the cell membrane
cocalyx of some cells attaches to the glycocalyx of
provide a means of conveying information about the
other cells, thus attaching cells to one another. (3)
environment to the cell interior.
Many of the carbohydrates act as receptor substances
Peripheral protein molecules are often attached to
for binding hormones, such as insulin; when bound,
the integral proteins. These peripheral proteins func-
this combination activates attached internal proteins
tion almost entirely as enzymes or as controllers of
that, in turn, activate a cascade of intracellular
transport of substances through the cell membrane
enzymes. (4) Some carbohydrate moieties enter into
“pores.”
immune reactions, as discussed in Chapter 34.
Membrane Carbohydrates—The Cell
“Glycocalyx.” Mem-
brane carbohydrates occur almost invariably in
Cytoplasm and Its Organelles
combination with proteins or lipids in the form of gly-
coproteins or glycolipids. In fact, most of the integral
The cytoplasm is filled with both minute and large dis-
proteins are glycoproteins, and about one tenth of the
persed particles and organelles. The clear fluid portion
membrane lipid molecules are glycolipids. The “glyco”
of the cytoplasm in which the particles are dispersed
portions of these molecules almost invariably protrude
is called cytosol; this contains mainly dissolved pro-
to the outside of the cell, dangling outward from the
teins, electrolytes, and glucose.
Chapter 2
The Cell and Its Functions
15
Dispersed in the cytoplasm are neutral fat globules,
endoplasmic reticulum are large numbers of minute
glycogen granules, ribosomes, secretory vesicles, and
granular particles called ribosomes. Where these are
five especially important organelles: the endoplasmic
present, the reticulum is called the granular endoplas-
reticulum, the Golgi apparatus, mitochondria, lyso-
mic reticulum. The ribosomes are composed of a
somes, and peroxisomes.
mixture of RNA and proteins, and they function to
synthesize new protein molecules in the cell, as dis-
Endoplasmic Reticulum
cussed later in this chapter and in Chapter 3.
Figure 2-2 shows a network of tubular and flat vesic-
ular structures in the cytoplasm; this is the endoplas-
Agranular Endoplasmic Reticulum. Part of the endoplasmic
mic reticulum. The tubules and vesicles interconnect
reticulum has no attached ribosomes. This part is
with one another. Also, their walls are constructed
called the agranular, or smooth, endoplasmic reticu-
of lipid bilayer membranes that contain large amounts
lum. The agranular reticulum functions for the syn-
of proteins, similar to the cell membrane. The total
thesis of lipid substances and for other processes of the
surface area of this structure in some cells—the liver
cells promoted by intrareticular enzymes.
cells, for instance—can be as much as 30 to 40 times
the cell membrane area.
Golgi Apparatus
The detailed structure of a small portion of endo-
The Golgi apparatus, shown in Figure 2-5, is closely
plasmic reticulum is shown in Figure 2-4. The space
related to the endoplasmic reticulum. It has mem-
inside the tubules and vesicles is filled with endoplas-
branes similar to those of the agranular endoplasmic
mic matrix, a watery medium that is different from the
reticulum. It usually is composed of four or more
fluid in the cytosol outside the endoplasmic reticulum.
stacked layers of thin, flat, enclosed vesicles lying near
Electron micrographs show that the space inside the
one side of the nucleus. This apparatus is prominent
endoplasmic reticulum is connected with the space
in secretory cells, where it is located on the side of
between the two membrane surfaces of the nuclear
the cell from which the secretory substances are
membrane.
extruded.
Substances formed in some parts of the cell enter
The Golgi apparatus functions in association with
the space of the endoplasmic reticulum and are then
the endoplasmic reticulum. As shown in Figure 2-5,
conducted to other parts of the cell. Also, the vast
small “transport vesicles” (also called endoplasmic
surface area of this reticulum and the multiple enzyme
reticulum vesicles, or ER vesicles) continually pinch off
systems attached to its membranes provide machinery
from the endoplasmic reticulum and shortly thereafter
for a major share of the metabolic functions of the cell.
fuse with the Golgi apparatus. In this way, substances
entrapped in the ER vesicles are transported from the
Ribosomes and the Granular Endoplasmic Reticulum.
endoplasmic reticulum to the Golgi apparatus. The
Attached to the outer surfaces of many parts of the
transported substances are then processed in the Golgi
apparatus to form lysosomes, secretory vesicles, and
other cytoplasmic components that are discussed later
in the chapter.
Golgi vesicles
Matrix
Golgi
apparatus
ER vesicles
Endoplasmic
Granular
reticulum
endoplasmic
Agranular
reticulum
endoplasmic
reticulum
Figure 2-4
Figure 2-5
Structure of the endoplasmic reticulum. (Modified from DeRober-
tis EDP, Saez FA, DeRobertis EMF: Cell Biology, 6th ed. Philadel-
A typical Golgi apparatus and its relationship to the endoplasmic
phia: WB Saunders, 1975.)
reticulum (ER) and the nucleus.
16
Unit I Introduction to Physiology: The Cell and General Physiology
Lysosomes
Secretory
Lysosomes, shown in Figure
2-2, are vesicular
granules
organelles that form by breaking off from the Golgi
apparatus and then dispersing throughout the cyto-
plasm. The lysosomes provide an intracellular digestive
system that allows the cell to digest (1) damaged cel-
lular structures,
(2) food particles that have been
ingested by the cell, and (3) unwanted matter such as
bacteria. The lysosome is quite different in different
types of cells, but it is usually 250 to 750 nanometers
in diameter. It is surrounded by a typical lipid bilayer
membrane and is filled with large numbers of small
granules 5 to 8 nanometers in diameter, which are
protein aggregates of as many as 40 different hydro-
Figure 2-6
lase
(digestive) enzymes. A hydrolytic enzyme is
Secretory granules
(secretory vesicles) in acinar cells of
the
capable of splitting an organic compound into two or
pancreas.
more parts by combining hydrogen from a water mol-
ecule with one part of the compound and combining
the hydroxyl portion of the water molecule with the
other part of the compound. For instance, protein
Outer membrane
is hydrolyzed to form amino acids, glycogen is
hydrolyzed to form glucose, and lipids are hydrolyzed
Inner membrane
to form fatty acids and glycerol.
Crests
Matrix
Ordinarily, the membrane surrounding the lysosome
prevents the enclosed hydrolytic enzymes from
coming in contact with other substances in the cell and,
therefore, prevents their digestive actions. However,
some conditions of the cell break the membranes of
some of the lysosomes, allowing release of the diges-
tive enzymes. These enzymes then split the organic
substances with which they come in contact into small,
Oxidative
highly diffusible substances such as amino acids and
phosphorylation
glucose. Some of the more specific functions of lyso-
Outer chamber
enzymes
somes are discussed later in the chapter.
Figure 2-7
Peroxisomes
Structure of a mitochondrion. (Modified from DeRobertis EDP,
Peroxisomes are similar physically to lysosomes, but
Saez FA, DeRobertis EMF: Cell Biology, 6th ed. Philadelphia: WB
they are different in two important ways. First, they are
Saunders, 1975.)
believed to be formed by self-replication (or perhaps
by budding off from the smooth endoplasmic reticu-
lum) rather than from the Golgi apparatus. Second,
they contain oxidases rather than hydrolases. Several
vesicles store protein proenzymes (enzymes that are
of the oxidases are capable of combining oxygen with
not yet activated). The proenzymes are secreted later
hydrogen ions derived from different intracellular
through the outer cell membrane into the pancreatic
chemicals to form hydrogen peroxide (H2O2). Hydro-
duct and thence into the duodenum, where they
gen peroxide is a highly oxidizing substance and is
become activated and perform digestive functions on
used in association with catalase, another oxidase
the food in the intestinal tract.
enzyme present in large quantities in peroxisomes, to
oxidize many substances that might otherwise be poi-
Mitochondria
sonous to the cell. For instance, about half the alcohol
The mitochondria, shown in Figures 2-2 and 2-7, are
a person drinks is detoxified by the peroxisomes of the
called the “powerhouses” of the cell. Without them,
liver cells in this manner.
cells would be unable to extract enough energy from
the nutrients, and essentially all cellular functions
Secretory Vesicles
would cease.
One of the important functions of many cells is secre-
Mitochondria are present in all areas of each cell’s
tion of special chemical substances. Almost all such
cytoplasm, but the total number per cell varies from
secretory substances are formed by the endoplasmic
less than a hundred up to several thousand, depending
reticulum-Golgi apparatus system and are then
on the amount of energy required by the cell. Further,
released from the Golgi apparatus into the cytoplasm
the mitochondria are concentrated in those portions
in the form of storage vesicles called secretory vesicles
of the cell that are responsible for the major share of
or secretory granules. Figure 2-6 shows typical secre-
its energy metabolism. They are also variable in size
tory vesicles inside pancreatic acinar cells; these
and shape. Some are only a few hundred nanometers
Chapter 2
The Cell and Its Functions
17
in diameter and globular in shape, whereas others are
elongated—as large as 1 micrometer in diameter and
7 micrometers long; still others are branching and
filamentous.
The basic structure of the mitochondrion, shown
in Figure
2-7, is composed mainly of two lipid
bilayer-protein membranes: an outer membrane and
an inner membrane. Many infoldings of the inner
membrane form shelves onto which oxidative enzymes
are attached. In addition, the inner cavity of the mito-
chondrion is filled with a matrix that contains large
quantities of dissolved enzymes that are necessary for
extracting energy from nutrients. These enzymes
operate in association with the oxidative enzymes on
the shelves to cause oxidation of the nutrients, thereby
forming carbon dioxide and water and at the same
time releasing energy. The liberated energy is used to
synthesize a “high-energy” substance called adenosine
Figure 2-8
triphosphate (ATP). ATP is then transported out of the
mitochondrion, and it diffuses throughout the cell to
Microtubules teased from the flagellum of a sperm.
(From
Wolstenholme GEW, O’Connor M, and The publisher, JA Churchill,
release its own energy wherever it is needed for per-
1967. Figure 4, page 314. Copyright the Novartis Foundation
forming cellular functions. The chemical details of ATP
formerly the Ciba Foundation.)
formation by the mitochondrion are given in Chapter
67, but some of the basic functions of ATP in the cell
are introduced later in this chapter.
Mitochondria are self-replicative, which means that
Nucleus
one mitochondrion can form a second one, a third one,
and so on, whenever there is a need in the cell for
The nucleus is the control center of the cell. Briefly, the
increased amounts of ATP. Indeed, the mitochondria
nucleus contains large quantities of DNA, which are
contain DNA similar to that found in the cell nucleus.
the genes. The genes determine the characteristics of
In Chapter 3 we will see that DNA is the basic chem-
the cell’s proteins, including the structural proteins, as
ical of the nucleus that controls replication of the cell.
well as the intracellular enzymes that control cyto-
The DNA of the mitochondrion plays a similar role,
plasmic and nuclear activities.
controlling replication of the mitochondrion itself.
The genes also control and promote reproduction of
the cell itself. The genes first reproduce to give two
Filament and Tubular Structures of the Cell
identical sets of genes; then the cell splits by a special
The fibrillar proteins of the cell are usually organized
process called mitosis to form two daughter cells, each
into filaments or tubules. These originate as precursor
of which receives one of the two sets of DNA genes.
protein molecules synthesized by ribosomes in the
All these activities of the nucleus are considered in
cytoplasm. The precursor molecules then polymerize
detail in the next chapter.
to form filaments. As an example, large numbers of
Unfortunately, the appearance of the nucleus under
actin filaments frequently occur in the outer zone of
the microscope does not provide many clues to the
the cytoplasm, called the ectoplasm, to form an elastic
mechanisms by which the nucleus performs its control
support for the cell membrane. Also, in muscle cells,
activities. Figure
2-9 shows the light microscopic
actin and myosin filaments are organized into a special
appearance of the interphase nucleus
(during the
contractile machine that is the basis for muscle con-
period between mitoses), revealing darkly staining
traction, as discussed in detail in Chapter 6.
chromatin material throughout the nucleoplasm.
A special type of stiff filament composed of poly-
During mitosis, the chromatin material organizes in
merized tubulin molecules is used in all cells to con-
the form of highly structured chromosomes, which can
struct very strong tubular structures, the microtubules.
then be easily identified using the light microscope, as
Figure
2-8 shows typical microtubules that were
illustrated in the next chapter.
teased from the flagellum of a sperm.
Another example of microtubules is the tubular
skeletal structure in the center of each cilium that radi-
Nuclear Membrane
ates upward from the cell cytoplasm to the tip of the
cilium. This structure is discussed later in the chapter
The nuclear membrane, also called the nuclear enve-
and is illustrated in Figure 2-17. Also, both the centri-
lope, is actually two separate bilayer membranes, one
oles and the mitotic spindle of the mitosing cell are
inside the other. The outer membrane is continuous
composed of stiff microtubules.
with the endoplasmic reticulum of the cell cytoplasm,
Thus, a primary function of microtubules is to act as
and the space between the two nuclear membranes is
a cytoskeleton, providing rigid physical structures for
also continuous with the space inside the endoplasmic
certain parts of cells.
reticulum, as shown in Figure 2-9.
18
Unit I Introduction to Physiology: The Cell and General Physiology
Pores
Nucleoplasm
15 nm — Small virus
150 nm — Large virus
Endoplasmic
reticulum
350 nm — Rickettsia
Nucleolus
1 mm Bacterium
Nuclear envelope -
outer and inner
membranes
Cell
Chromatin material (DNA)
Cytoplasm
5 - 10 mm +
Figure 2-9
Figure 2-10
Structure of the nucleus.
Comparison of sizes of precellular organisms with that of the
average cell in the human body.
The nuclear membrane is penetrated by several
thousand nuclear pores. Large complexes of protein
molecules are attached at the edges of the pores so
(4) a bacterium, and (5) a nucleated cell, demonstrat-
that the central area of each pore is only about 9
ing that the cell has a diameter about 1000 times that
nanometers in diameter. Even this size is large enough
of the smallest virus and, therefore, a volume about 1
to allow molecules up to 44,000 molecular weight to
billion times that of the smallest virus. Correspond-
pass through with reasonable ease.
ingly, the functions and anatomical organization of the
cell are also far more complex than those of the virus.
The essential life-giving constituent of the small
Nucleoli and Formation of Ribosomes
virus is a nucleic acid embedded in a coat of protein.
This nucleic acid is composed of the same basic nucleic
The nuclei of most cells contain one or more highly
acid constituents (DNA or RNA) found in mammalian
staining structures called nucleoli. The nucleolus,
cells, and it is capable of reproducing itself under
unlike most other organelles discussed here, does not
appropriate conditions. Thus, the virus propagates its
have a limiting membrane. Instead, it is simply an accu-
lineage from generation to generation and is therefore
mulation of large amounts of RNA and proteins of the
a living structure in the same way that the cell and the
types found in ribosomes. The nucleolus becomes con-
human being are living structures.
siderably enlarged when the cell is actively synthesiz-
As life evolved, other chemicals besides nucleic acid
ing proteins.
and simple proteins became integral parts of the
Formation of the nucleoli (and of the ribosomes in
organism, and specialized functions began to develop
the cytoplasm outside the nucleus) begins in the
in different parts of the virus. A membrane formed
nucleus. First, specific DNA genes in the chromosomes
around the virus, and inside the membrane, a fluid
cause RNA to be synthesized. Some of this is stored
matrix appeared. Specialized chemicals then devel-
in the nucleoli, but most of it is transported outward
oped inside the fluid to perform special functions;
through the nuclear pores into cytoplasm. Here, it is
many protein enzymes appeared that were capable of
used in conjunction with specific proteins to assemble
catalyzing chemical reactions and, therefore, deter-
“mature” ribosomes that play an essential role in
mining the organism’s activities.
forming cytoplasmic proteins, as discussed more fully
In still later stages of life, particularly in the rick-
in Chapter 3.
ettsial and bacterial stages, organelles developed inside
the organism, representing physical structures of
chemical aggregates that perform functions in a more
Comparison of the Animal Cell
efficient manner than can be achieved by dispersed
with Precellular Forms of Life
chemicals throughout the fluid matrix.
Finally, in the nucleated cell, still more complex
Many of us think of the cell as the lowest level of life.
organelles developed, the most important of which is
However, the cell is a very complicated organism that
the nucleus itself. The nucleus distinguishes this type
required many hundreds of millions of years to
of cell from all lower forms of life; the nucleus pro-
develop after the earliest form of life, an organism
vides a control center for all cellular activities, and it
similar to the present-day virus, first appeared on
provides for exact reproduction of new cells genera-
earth. Figure 2-10 shows the relative sizes of (1) the
tion after generation, each new cell having almost
smallest known virus, (2) a large virus, (3) a rickettsia,
exactly the same structure as its progenitor.
Chapter 2
The Cell and Its Functions
19
Functional Systems of the Cell
Proteins
Receptors
Coated pit
In the remainder of this chapter, we discuss several
Clathrin
representative functional systems of the cell that make
it a living organism.
Ingestion by the Cell—Endocytosis
A
B
If a cell is to live and grow and reproduce, it must
Actin and myosin
Dissolving clathrin
obtain nutrients and other substances from the sur-
rounding fluids. Most substances pass through the cell
membrane by diffusion and active transport.
Diffusion involves simple movement through the
membrane caused by the random motion of the mol-
ecules of the substance; substances move either
through cell membrane pores or, in the case of lipid-
C
D
soluble substances, through the lipid matrix of the
Figure 2-11
membrane.
Active transport involves the actual carrying of a
Mechanism of pinocytosis.
substance through the membrane by a physical pro-
tein structure that penetrates all the way through the
membrane. These active transport mechanisms are so
membrane change in such a way that the entire pit
important to cell function that they are presented in
invaginates inward, and the fibrillar proteins sur-
detail in Chapter 4.
rounding the invaginating pit cause its borders to close
Very large particles enter the cell by a specialized
over the attached proteins as well as over a small
function of the cell membrane called endocytosis. The
amount of extracellular fluid. Immediately thereafter,
principal forms of endocytosis are pinocytosis and
the invaginated portion of the membrane breaks away
phagocytosis. Pinocytosis means ingestion of minute
from the surface of the cell, forming a pinocytotic
particles that form vesicles of extracellular fluid and
vesicle inside the cytoplasm of the cell.
particulate constituents inside the cell cytoplasm.
What causes the cell membrane to go through the
Phagocytosis means ingestion of large particles, such
necessary contortions to form pinocytotic vesicles
as bacteria, whole cells, or portions of degenerating
remains mainly a mystery. This process requires energy
tissue.
from within the cell; this is supplied by ATP, a high-
energy substance discussed later in the chapter. Also,
Pinocytosis. Pinocytosis occurs continually in the cell
it requires the presence of calcium ions in the extra-
membranes of most cells, but it is especially rapid
cellular fluid, which probably react with contractile
in some cells. For instance, it occurs so rapidly in
protein filaments beneath the coated pits to provide
macrophages that about 3 per cent of the total macro-
the force for pinching the vesicles away from the cell
phage membrane is engulfed in the form of vesicles
membrane.
each minute. Even so, the pinocytotic vesicles are so
small—usually only 100 to 200 nanometers in diame-
Phagocytosis. Phagocytosis occurs in much the same
ter—that most of them can be seen only with the elec-
way as pinocytosis, except that it involves large
tron microscope.
particles rather than molecules. Only certain cells
Pinocytosis is the only means by which most large
have the capability of phagocytosis, most notably the
macromolecules, such as most protein molecules, can
tissue macrophages and some of the white blood cells.
enter cells. In fact, the rate at which pinocytotic vesi-
Phagocytosis is initiated when a particle such as a
cles form is usually enhanced when such macro-
bacterium, a dead cell, or tissue debris binds with
molecules attach to the cell membrane.
receptors on the surface of the phagocyte. In the case
Figure 2-11 demonstrates the successive steps of
of bacteria, each bacterium usually is already attached
pinocytosis, showing three molecules of protein
to a specific antibody, and it is the antibody that
attaching to the membrane. These molecules usually
attaches to the phagocyte receptors, dragging the bac-
attach to specialized protein receptors on the surface
terium along with it. This intermediation of antibodies
of the membrane that are specific for the type of
is called opsonization, which is discussed in Chapters
protein that is to be absorbed. The receptors generally
33 and 34.
are concentrated in small pits on the outer surface of
Phagocytosis occurs in the following steps:
the cell membrane, called coated pits. On the inside of
1. The cell membrane receptors attach to the surface
the cell membrane beneath these pits is a latticework
ligands of the particle.
of fibrillar protein called clathrin, as well as other pro-
2. The edges of the membrane around the points of
teins, perhaps including contractile filaments of actin
attachment evaginate outward within a fraction of
and myosin. Once the protein molecules have bound
a second to surround the entire particle; then,
with the receptors, the surface properties of the local
progressively more and more membrane receptors
20
Unit I Introduction to Physiology: The Cell and General Physiology
attach to the particle ligands. All this occurs
occurs in the uterus after pregnancy, in muscles during
suddenly in a zipper-like manner to form a closed
long periods of inactivity, and in mammary glands at
phagocytic vesicle.
the end of lactation. Lysosomes are responsible for
3. Actin and other contractile fibrils in the cytoplasm
much of this regression. The mechanism by which lack
surround the phagocytic vesicle and contract
of activity in a tissue causes the lysosomes to increase
around its outer edge, pushing the vesicle to the
their activity is unknown.
interior.
Another special role of the lysosomes is removal of
4. The contractile proteins then pinch the stem of
damaged cells or damaged portions of cells from
the vesicle so completely that the vesicle
tissues. Damage to the cell—caused by heat, cold,
separates from the cell membrane, leaving the
trauma, chemicals, or any other factor—induces lyso-
vesicle in the cell interior in the same way that
somes to rupture. The released hydrolases immedi-
pinocytotic vesicles are formed.
ately begin to digest the surrounding organic
substances. If the damage is slight, only a portion of
the cell is removed, followed by repair of the cell. If
Digestion of Pinocytotic and
the damage is severe, the entire cell is digested, a
Phagocytic Foreign Substances Inside
process called autolysis. In this way, the cell is com-
the Cell—Function of the Lysosomes
pletely removed, and a new cell of the same type ordi-
narily is formed by mitotic reproduction of an adjacent
Almost immediately after a pinocytotic or phagocytic
cell to take the place of the old one.
vesicle appears inside a cell, one or more lysosomes
The lysosomes also contain bactericidal agents that
become attached to the vesicle and empty their acid
can kill phagocytized bacteria before they can cause
hydrolases to the inside of the vesicle, as shown in
cellular damage. These agents include (1) lysozyme,
Figure 2-12. Thus, a digestive vesicle is formed inside
which dissolves the bacterial cell membrane; (2) lyso-
the cell cytoplasm in which the vesicular hydrolases
ferrin, which binds iron and other substances before
begin hydrolyzing the proteins, carbohydrates, lipids,
they can promote bacterial growth; and (3) acid at a
and other substances in the vesicle. The products of
pH of about 5.0, which activates the hydrolases and
digestion are small molecules of amino acids, glucose,
inactivates bacterial metabolic systems.
phosphates, and so forth that can diffuse through the
membrane of the vesicle into the cytoplasm. What is
left of the digestive vesicle, called the residual body,
Synthesis and Formation of Cellular
represents indigestible substances. In most instances,
Structures by Endoplasmic Reticulum
this is finally excreted through the cell membrane by
and Golgi Apparatus
a process called exocytosis, which is essentially the
opposite of endocytosis.
Specific Functions of the
Thus, the pinocytotic and phagocytic vesicles con-
Endoplasmic Reticulum
taining lysosomes can be called the digestive organs of
The extensiveness of the endoplasmic reticulum and
the cells.
the Golgi apparatus in secretory cells has already been
emphasized. These structures are formed primarily of
Regression of Tissues and Autolysis of Cells. Tissues of the
lipid bilayer membranes similar to the cell membrane,
body often regress to a smaller size. For instance, this
and their walls are loaded with protein enzymes that
catalyze the synthesis of many substances required by
Lysosomes
the cell.
Most synthesis begins in the endoplasmic reticulum.
The products formed there are then passed on to the
Golgi apparatus, where they are further processed
before being released into the cytoplasm. But first, let
us note the specific products that are synthesized in
Pinocytotic or
specific portions of the endoplasmic reticulum and the
phagocytic
Golgi apparatus.
vesicle
Digestive vesicle
Proteins Are Formed by the Granular Endoplasmic Reticulum.
The granular portion of the endoplasmic reticulum is
characterized by large numbers of ribosomes attached
to the outer surfaces of the endoplasmic reticulum
Residual body
membrane. As we discuss in Chapter 3, protein mole-
cules are synthesized within the structures of the ribo-
somes. The ribosomes extrude some of the synthesized
Excretion
protein molecules directly into the cytosol, but they
also extrude many more through the wall of the endo-
Figure 2-12
plasmic reticulum to the interior of the endoplasmic
Digestion of substances in pinocytotic or phagocytic vesicles by
vesicles and tubules, that is, into the endoplasmic
enzymes derived from lysosomes.
matrix.
Chapter 2
The Cell and Its Functions
21
Synthesis of Lipids by the Smooth Endoplasmic Reticulum.
Protein
Lipid
Secretory
The endoplasmic reticulum also synthesizes lipids,
Ribosomes
formation
formation
Lysosomes
vesicles
especially phospholipids and cholesterol. These are
rapidly incorporated into the lipid bilayer of the endo-
plasmic reticulum itself, thus causing the endoplasmic
reticulum to grow more extensive. This occurs mainly
in the smooth portion of the endoplasmic reticulum.
To keep the endoplasmic reticulum from growing
beyond the needs of the cell, small vesicles called ER
vesicles or transport vesicles continually break away
from the smooth reticulum; most of these vesicles then
migrate rapidly to the Golgi apparatus.
Other Functions of the Endoplasmic Reticulum. Other sig-
nificant functions of the endoplasmic reticulum, espe-
cially the smooth reticulum, include the following:
Transport
1. It provides the enzymes that control glycogen
Glycosylation
vesicles
breakdown when glycogen is to be used for
Granular
Smooth
Golgi
energy.
endoplasmic
endoplasmic
apparatus
2. It provides a vast number of enzymes that are
reticulum
reticulum
capable of detoxifying substances, such as drugs,
that might damage the cell. It achieves
Figure 2-13
detoxification by coagulation, oxidation,
Formation of proteins, lipids, and cellular vesicles by the endo-
hydrolysis, conjugation with glycuronic acid, and
plasmic reticulum and Golgi apparatus.
in other ways.
Specific Functions of the Golgi Apparatus
additional carbohydrate moieties are added to the
Synthetic Functions of the Golgi Apparatus. Although the
secretions. Also, an important function of the Golgi
major function of the Golgi apparatus is to provide
apparatus is to compact the endoplasmic reticular
additional processing of substances already formed
secretions into highly concentrated packets. As the
in the endoplasmic reticulum, it also has the capability
secretions pass toward the outermost layers of the
of synthesizing certain carbohydrates that cannot be
Golgi apparatus, the compaction and processing
formed in the endoplasmic reticulum. This is especially
proceed. Finally, both small and large vesicles contin-
true for the formation of large saccharide polymers
ually break away from the Golgi apparatus, carrying
bound with small amounts of protein; the most impor-
with them the compacted secretory substances, and in
tant of these are hyaluronic acid and chondroitin sulfate.
turn, the vesicles diffuse throughout the cell.
A few of the many functions of hyaluronic acid and
To give an idea of the timing of these processes:
chondroitin sulfate in the body are as follows: (1) they
When a glandular cell is bathed in radioactive amino
are the major components of proteoglycans secreted
acids, newly formed radioactive protein molecules can
in mucus and other glandular secretions; (2) they are
be detected in the granular endoplasmic reticulum
the major components of the ground substance outside
within 3 to 5 minutes. Within 20 minutes, newly formed
the cells in the interstitial spaces, acting as filler
proteins are already present in the Golgi apparatus,
between collagen fibers and cells; and (3) they are
and within 1 to 2 hours, radioactive proteins are
principal components of the organic matrix in both
secreted from the surface of the cell.
cartilage and bone.
Types of Vesicles Formed by the Golgi Apparatus—Secretory
Processing of Endoplasmic Secretions by the Golgi Apparatus—
Vesicles and Lysosomes. In a highly secretory cell, the
Formation of Vesicles. Figure
2-13 summarizes the
vesicles formed by the Golgi apparatus are mainly
major functions of the endoplasmic reticulum and
secretory vesicles containing protein substances that
Golgi apparatus. As substances are formed in the
are to be secreted through the surface of the cell mem-
endoplasmic reticulum, especially the proteins, they
brane. These secretory vesicles first diffuse to the cell
are transported through the tubules toward portions
membrane, then fuse with it and empty their sub-
of the smooth endoplasmic reticulum that lie nearest
stances to the exterior by the mechanism called exo-
the Golgi apparatus. At this point, small transport vesi-
cytosis. Exocytosis, in most cases, is stimulated by the
cles composed of small envelopes of smooth endo-
entry of calcium ions into the cell; calcium ions inter-
plasmic reticulum continually break away and diffuse
act with the vesicular membrane in some way that is
to the deepest layer of the Golgi apparatus. Inside these
not understood and cause its fusion with the cell mem-
vesicles are the synthesized proteins and other prod-
brane, followed by exocytosis—that is, opening of the
ucts from the endoplasmic reticulum.
membrane’s outer surface and extrusion of its contents
The transport vesicles instantly fuse with the Golgi
outside the cell.
apparatus and empty their contained substances into
Some vesicles, however, are destined for intracellu-
the vesicular spaces of the Golgi apparatus. Here,
lar use.
22
Unit I Introduction to Physiology: The Cell and General Physiology
Use of Intracellular Vesicles to Replenish Cellular Membranes.
all these digestive and metabolic functions are given
Some of the intracellular vesicles formed by the Golgi
in Chapters 62 through 72.
apparatus fuse with the cell membrane or with the
Briefly, almost all these oxidative reactions occur
membranes of intracellular structures such as the
inside the mitochondria, and the energy that is
mitochondria and even the endoplasmic reticulum.
released is used to form the high-energy compound
This increases the expanse of these membranes and
ATP. Then, ATP, not the original foodstuffs, is used
thereby replenishes the membranes as they are used
throughout the cell to energize almost all the subse-
up. For instance, the cell membrane loses much of its
quent intracellular metabolic reactions.
substance every time it forms a phagocytic or pinocy-
totic vesicle, and the vesicular membranes of the Golgi
Functional Characteristics of ATP
apparatus continually replenish the cell membrane.
In summary, the membranous system of the endo-
plasmic reticulum and Golgi apparatus represents a
NH2
highly metabolic organ capable of forming new intra-
N C
cellular structures as well as secretory substances to be
C
N
extruded from the cell.
Adenine
HC
C
CH
O
O
O
N
N
Extraction of Energy from Nutrients—
CH O P
O
~
P
O
~
P
O-
O
2
Function of the Mitochondria
C
C
O-
O-
O-
H
H
Phosphate
The principal substances from which cells extract
H
C C H
energy are foodstuffs that react chemically with
oxygen—carbohydrates, fats, and proteins. In the
OH OH
human body, essentially all carbohydrates are con-
Ribose
verted into glucose by the digestive tract and liver
Adenosine triphosphate
before they reach the other cells of the body. Similarly,
proteins are converted into amino acids and fats into
ATP is a nucleotide composed of (1) the nitrogenous
fatty acids. Figure 2-14 shows oxygen and the food-
base adenine, (2) the pentose sugar ribose, and (3)
stuffs—glucose, fatty acids, and amino acids—all enter-
three phosphate radicals. The last two phosphate rad-
ing the cell. Inside the cell, the foodstuffs react
icals are connected with the remainder of the molecule
chemically with oxygen, under the influence of
by so-called high-energy phosphate bonds, which are
enzymes that control the reactions and channel the
represented in the formula above by the symbol ~.
energy released in the proper direction. The details of
Under the physical and chemical conditions of the
body, each of these high-energy bonds contains about
12,000 calories of energy per mole of ATP, which is
many times greater than the energy stored in the
average chemical bond, thus giving rise to the term
2ADP
2ATP
high-energy bond. Further, the high-energy phosphate
bond is very labile, so that it can be split instantly on
Glucose
Gl
Pyruvic acid
demand whenever energy is required to promote
Fatty acids
FA Acetoacetic
36 ADP
other intracellular reactions.
acid
When ATP releases its energy, a phosphoric acid
Amino acids
AA
Acetyl-CoA
radical is split away, and adenosine diphosphate (ADP)
is formed. This released energy is used to energize vir-
tually all of the cell’s other functions, such as synthe-
Acetyl-CoA
sis of substances and muscular contraction.
O2
O2
O2
ADP
To reconstitute the cellular ATP as it is used up,
CO2
CO2
CO2+H2O
ATP
energy derived from the cellular nutrients causes ADP
and phosphoric acid to recombine to form new ATP,
and the entire process repeats over and over again. For
H2O
H2O
36 ATP
these reasons, ATP has been called the energy currency
of the cell because it can be spent and remade contin-
Mitochondrion
ually, having a turnover time of only a few minutes.
Nucleus
Cell
membrane
Chemical Processes in the Formation of ATP—Role of the
Mitochondria. On entry into the cells, glucose is sub-
Figure 2-14
jected to enzymes in the cytoplasm that convert it
into pyruvic acid (a process called glycolysis). A small
Formation of adenosine triphosphate (ATP) in the cell, showing
amount of ADP is changed into ATP by the energy
that most of the ATP is formed in the mitochondria. ADP, adeno-
sine diphosphate.
released during this conversion, but this amount
Chapter 2
The Cell and Its Functions
23
accounts for less than 5 per cent of the overall energy
Ribosomes
metabolism of the cell.
Membrane
By far, the major portion of the ATP formed in the
Endoplasmic
transport
cell, about 95 per cent, is formed in the mitochondria.
reticulum
The pyruvic acid derived from carbohydrates, fatty
acids from lipids, and amino acids from proteins are
eventually converted into the compound acetyl-CoA
Protein synthesis
in the matrix of the mitochondrion. This substance, in
Na+
Na+
ATP
ADP
turn, is further dissoluted (for the purpose of extract-
ing its energy) by another series of enzymes in the
ADP
mitochondrion matrix, undergoing dissolution in a
Mitochondrion
sequence of chemical reactions called the citric acid
cycle, or Krebs cycle. These chemical reactions are
ATP
ATP
ADP
so important that they are explained in detail in
Chapter 67.
In this citric acid cycle, acetyl-CoA is split into its
ATP
ADP
component parts, hydrogen atoms and carbon dioxide.
The carbon dioxide diffuses out of the mitochondria
and eventually out of the cell; finally, it is excreted
from the body through the lungs.
Muscle contraction
The hydrogen atoms, conversely, are highly reactive,
and they combine instantly with oxygen that has also
Figure 2-15
diffused into the mitochondria. This releases a tremen-
dous amount of energy, which is used by the mito-
Use of adenosine triphosphate (ATP) (formed in the mitochon-
drion) to provide energy for three major cellular functions: mem-
chondria to convert very large amounts of ADP to
brane transport, protein synthesis, and muscle contraction. ADP,
ATP. The processes of these reactions are complex,
adenosine diphosphate.
requiring the participation of large numbers of protein
enzymes that are integral parts of mitochondrial mem-
branous shelves that protrude into the mitochondrial
matrix. The initial event is removal of an electron from
the hydrogen atom, thus converting it to a hydrogen
phate ions, chloride ions, urate ions, hydrogen ions,
ion. The terminal event is combination of hydrogen
and many other ions and various organic substances.
ions with oxygen to form water plus the release of
Membrane transport is so important to cell function
tremendous amounts of energy to large globular pro-
that some cells—the renal tubular cells, for instance—
teins, called ATP synthetase, that protrude like knobs
use as much as 80 per cent of the ATP that they form
from the membranes of the mitochondrial shelves.
for this purpose alone.
Finally, the enzyme ATP synthetase uses the energy
In addition to synthesizing proteins, cells synthesize
from the hydrogen ions to cause the conversion of
phospholipids, cholesterol, purines, pyrimidines, and a
ADP to ATP. The newly formed ATP is transported
host of other substances. Synthesis of almost any
out of the mitochondria into all parts of the cell cyto-
chemical compound requires energy. For instance, a
plasm and nucleoplasm, where its energy is used to
single protein molecule might be composed of as many
energize multiple cell functions.
as several thousand amino acids attached to one
This overall process for formation of ATP is called
another by peptide linkages; the formation of each of
the chemiosmotic mechanism of ATP formation. The
these linkages requires energy derived from the break-
chemical and physical details of this mechanism are
down of four high-energy bonds; thus, many thousand
presented in Chapter 67, and many of the detailed
ATP molecules must release their energy as each
metabolic functions of ATP in the body are presented
protein molecule is formed. Indeed, some cells use as
in Chapters 67 through 71.
much as 75 per cent of all the ATP formed in the cell
simply to synthesize new chemical compounds, espe-
Uses of ATP for Cellular Function. Energy from ATP is
cially protein molecules; this is particularly true during
used to promote three major categories of cellular
the growth phase of cells.
functions: (1) transport of substances through multiple
The final major use of ATP is to supply energy for
membranes in the cell,
(2) synthesis of chemical
special cells to perform mechanical work. We see in
compounds throughout the cell, and (3) mechanical
Chapter 6 that each contraction of a muscle fiber
work. These uses of ATP are illustrated by examples
requires expenditure of tremendous quantities of ATP
in Figure 2-15: (1) to supply energy for the transport
energy. Other cells perform mechanical work in other
of sodium through the cell membrane, (2) to promote
ways, especially by ciliary and ameboid motion, which
protein synthesis by the ribosomes, and (3) to supply
are described later in this chapter. The source of
the energy needed during muscle contraction.
energy for all these types of mechanical work is ATP.
In addition to membrane transport of sodium,
In summary, ATP is always available to release its
energy from ATP is required for membrane transport
energy rapidly and almost explosively wherever in the
of potassium ions, calcium ions, magnesium ions, phos-
cell it is needed. To replace the ATP used by the cell,
24
Unit I Introduction to Physiology: The Cell and General Physiology
much slower chemical reactions break down carbohy-
remainder of the cell body is pulled forward toward the
drates, fats, and proteins and use the energy derived
point of attachment. This attachment is effected by
from these to form new ATP. More than 95 per cent
receptor proteins that line the insides of exocytotic vesi-
cles. When the vesicles become part of the pseudopodial
of this ATP is formed in the mitochondria, which
membrane, they open so that their insides evert to the
accounts for the mitochondria being called the “pow-
outside, and the receptors now protrude to the outside
erhouses” of the cell.
and attach to ligands in the surrounding tissues.
At the opposite end of the cell, the receptors pull
away from their ligands and form new endocytotic vesi-
Locomotion of Cells
cles. Then, inside the cell, these vesicles stream toward
By far the most important type of movement that occurs
the pseudopodial end of the cell, where they are used
in the body is that of the muscle cells in skeletal, cardiac,
to form still new membrane for the pseudopodium.
and smooth muscle, which constitute almost 50 per cent
The second essential effect for locomotion is to
of the entire body mass. The specialized functions of
provide the energy required to pull the cell body in the
these cells are discussed in Chapters 6 through 9. Two
direction of the pseudopodium. Experiments suggest
other types of movement—ameboid locomotion and
the following as an explanation: In the cytoplasm of all
ciliary movement—occur in other cells.
cells is a moderate to large amount of the protein actin.
Much of the actin is in the form of single molecules that
do not provide any motive power; however, these poly-
Ameboid Movement
merize to form a filamentous network, and the network
contracts when it binds with an actin-binding protein
Ameboid movement is movement of an entire cell in
such as myosin. The whole process is energized by the
relation to its surroundings, such as movement of white
high-energy compound ATP. This is what happens in
blood cells through tissues. It receives its name from the
the pseudopodium of a moving cell, where such
fact that amebae move in this manner and have pro-
a network of actin filaments forms anew inside the
vided an excellent tool for studying the phenomenon.
enlarging pseudopodium. Contraction also occurs in
Typically, ameboid locomotion begins with protrusion
the ectoplasm of the cell body, where a preexisting
of a pseudopodium from one end of the cell. The
actin network is already present beneath the cell
pseudopodium projects far out, away from the cell
membrane.
body, and partially secures itself in a new tissue area.
Then the remainder of the cell is pulled toward the
Types of Cells That Exhibit Ameboid Locomotion. The most
pseudopodium. Figure 2-16 demonstrates this process,
common cells to exhibit ameboid locomotion in the
showing an elongated cell, the right-hand end of which
human body are the white blood cells when they move
is a protruding pseudopodium. The membrane of this
out of the blood into the tissues in the form of tissue
end of the cell is continually moving forward, and the
macrophages. Other types of cells can also move by
membrane at the left-hand end of the cell is continually
ameboid locomotion under certain circumstances. For
following along as the cell moves.
instance, fibroblasts move into a damaged area to help
repair the damage, and even the germinal cells of the
Mechanism of Ameboid Locomotion. Figure 2-16 shows the
skin, though ordinarily completely sessile cells, move
general principle of ameboid motion. Basically, it results
toward a cut area to repair the rent. Finally, cell loco-
from continual formation of new cell membrane at the
motion is especially important in development of the
leading edge of the pseudopodium and continual
embryo and fetus after fertilization of an ovum. For
absorption of the membrane in mid and rear portions
instance, embryonic cells often must migrate long dis-
of the cell. Also, two other effects are essential for
tances from their sites of origin to new areas during
forward movement of the cell. The first effect is attach-
development of special structures.
ment of the pseudopodium to surrounding tissues so
Control of Ameboid Locomotion—Chemotaxis. The most
that it becomes fixed in its leading position, while the
important initiator of ameboid locomotion is the
process called chemotaxis. This results from the appear-
ance of certain chemical substances in the tissues.
Movement of cell
Any chemical substance that causes chemotaxis to
occur is called a chemotactic substance. Most cells
Endocytosis
that exhibit ameboid locomotion move toward the
source of a chemotactic substance—that is, from an area
of lower concentration toward an area of higher con-
Pseudopodium
centration—which is called positive chemotaxis. Some
cells move away from the source, which is called nega-
tive chemotaxis.
But how does chemotaxis control the direction of
Exocytosis
ameboid locomotion? Although the answer is not
certain, it is known that the side of the cell most exposed
to the chemotactic substance develops membrane
changes that cause pseudopodial protrusion.
Surrounding tissue
Receptor binding
Cilia and Ciliary Movements
Figure 2-16
A second type of cellular motion, ciliary movement, is a
Ameboid motion by a cell.
whiplike movement of cilia on the surfaces of cells. This
Chapter 2
The Cell and Its Functions
25
The flagellum of a sperm is similar to a cilium; in fact,
Tip
it has much the same type of structure and same type
of contractile mechanism. The flagellum, however, is
much longer and moves in quasi-sinusoidal waves
instead of whiplike movements.
In the inset of Figure 2-17, movement of the cilium is
shown. The cilium moves forward with a sudden, rapid
whiplike stroke 10 to 20 times per second, bending
Membrane
sharply where it projects from the surface of the cell.
Then it moves backward slowly to its initial position.
Cross section
The rapid forward-thrusting, whiplike movement
pushes the fluid lying adjacent to the cell in the direc-
tion that the cilium moves; the slow, dragging movement
Filament
in the backward direction has almost no effect on fluid
movement. As a result, the fluid is continually propelled
in the direction of the fast-forward stroke. Because most
ciliated cells have large numbers of cilia on their sur-
faces and because all the cilia are oriented in the same
direction, this is an effective means for moving fluids
Forward stroke
from one part of the surface to another.
Mechanism of Ciliary Movement. Although not all aspects of
ciliary movement are clear, we do know the following:
Basal plate
First, the nine double tubules and the two single tubules
are all linked to one another by a complex of protein
Cell
cross-linkages; this total complex of tubules and cross-
membrane
linkages is called the axoneme. Second, even after
Backward stroke
removal of the membrane and destruction of other ele-
Basal body
ments of the cilium besides the axoneme, the cilium can
still beat under appropriate conditions. Third, there are
two necessary conditions for continued beating of the
Rootlet
axoneme after removal of the other structures of the
cilium: (1) the availability of ATP and (2) appropriate
ionic conditions, especially appropriate concentrations
of magnesium and calcium. Fourth, during forward
motion of the cilium, the double tubules on the front
edge of the cilium slide outward toward the tip of the
Figure 2-17
cilium, while those on the back edge remain in place.
Fifth, multiple protein arms composed of the protein
Structure and function of the cilium. (Modified from Satir P: Cilia.
dynein, which has ATPase enzymatic activity, project
Sci Am 204:108, 1961. Copyright Donald Garber: Executor of the
from each double tubule toward an adjacent double
estate of Bunji Tagawa.)
tubule.
occurs in only two places in the human body: on the
Given this basic information, it has been determined
sufaces of the respiratory airways and on the inside
that the release of energy from ATP in contact with the
surfaces of the uterine tubes (fallopian tubes) of the
ATPase dynein arms causes the heads of these arms to
reproductive tract. In the nasal cavity and lower respi-
“crawl” rapidly along the surface of the adjacent double
ratory airways, the whiplike motion of cilia causes a
tubule. If the front tubules crawl outward while the back
layer of mucus to move at a rate of about 1 cm/min
tubules remain stationary, this will cause bending.
toward the pharynx, in this way continually clearing
The way in which cilia contraction is controlled is not
these passageways of mucus and particles that have
understood. The cilia of some genetically abnormal cells
become trapped in the mucus. In the uterine tubes, the
do not have the two central single tubules, and these
cilia cause slow movement of fluid from the ostium of
cilia fail to beat. Therefore, it is presumed that some
the uterine tube toward the uterus cavity; this move-
signal, perhaps an electrochemical signal, is transmitted
ment of fluid transports the ovum from the ovary to the
along these two central tubules to activate the dynein
uterus.
arms.
As shown in Figure 2-17, a cilium has the appearance
of a sharp-pointed straight or curved hair that projects
2 to 4 micrometers from the surface of the cell. Many
References
cilia often project from a single cell—for instance, as
many as 200 cilia on the surface of each epithelial cell
Alberts B, Johnson A, Lewis J, et al: Molecular Biology of
inside the respiratory passageways. The cilium is
the Cell. New York: Garland Science, 2002.
covered by an outcropping of the cell membrane, and it
Bonifacino JS, Glick BS: The mechanisms of vesicle budding
is supported by
11 microtubules—9 double tubules
and fusion. Cell 116:153, 2004.
located around the periphery of the cilium, and 2 single
Calakos N, Scheller RH: Synaptic vesicle biogenesis,
tubules down the center, as demonstrated in the cross
docking, and fusion: a molecular description. Physiol Rev
section shown in Figure 2-17. Each cilium is an out-
76:1, 1996.
growth of a structure that lies immediately beneath the
Danial NN, Korsmeyer SJ: Cell death: critical control points.
cell membrane, called the basal body of the cilium.
Cell 116:205, 2004.
26
Unit I Introduction to Physiology: The Cell and General Physiology
Deutsch C: The birth of a channel. Neuron 40:265, 2003.
Maxfield FR, McGraw TE: Endocytic recycling. Nat Rev
Dröge W: Free radicals in the physiological control of cell
Mol Cell Biol 5:121, 2004.
function. Physiol Rev 82:47, 2002.
Mazzanti M, Bustamante JO, Oberleithner H: Electrical
Duchen MR: Roles of mitochondria in health and disease.
dimension of the nuclear envelope. Physiol Rev 81:1, 2001.
Diabetes 53(Suppl 1):S96, 2004.
Perrios M: Nuclear Structure and Function. San Diego: Aca-
Edidin M: Lipids on the frontier: a century of cell-membrane
demic Press, 1998.
bilayers. Nat Rev Mol Cell Biol 4:414, 2003.
Ridley AJ, Schwartz MA, Burridge K, et al: Cell migration:
Gerbi SA, Borovjagin AV, Lange TS: The nucleolus: a site of
integrating signals from front to back. Science 302:1704,
ribonucleoprotein maturation. Curr Opin Cell Biol 15:318,
2003.
2003.
Scholey JM: Intraflagellar transport. Annu Rev Cell Dev
Hamill OP, Martinac B: Molecular basis of mechanotrans-
Biol 19:423, 2003.
duction in living cells. Physiol Rev 81:685, 2001.
Schwab A: Function and spatial distribution of ion channels
Lange K: Role of microvillar cell surfaces in the regulation
and transporters in cell migration. Am J Physiol Renal
of glucose uptake and organization of energy metabolism.
Physiol 280:F739, 2001.
Am J Physiol Cell Physiol 282:C1, 2002.
Vereb G, Szollosi J, Matko J, et al: Dynamic, yet structured:
Mattaj IW: Sorting out the nuclear envelope from the
the cell membrane three decades after the Singer-
endoplasmic reticulum. Nat Rev Mol Cell Biol
5:65,
Nicolson model. Proc Natl Acad Sci U S A 100:8053,
2004.
2003.
C H A P T E R
3
Genetic Control of Protein
Synthesis, Cell Function,
and Cell Reproduction
Virtually everyone knows that the genes, located in
the nuclei of all cells of the body, control heredity
from parents to children, but most people do not
realize that these same genes also control day-to-
day function of all the body’s cells. The genes
control cell function by determining which sub-
stances are synthesized within the cell—which struc-
tures, which enzymes, which chemicals.
Figure 3-1 shows the general schema of genetic control. Each gene, which is
a nucleic acid called deoxyribonucleic acid (DNA), automatically controls the
formation of another nucleic acid, ribonucleic acid (RNA); this RNA then
spreads throughout the cell to control the formation of a specific protein.
Because there are more than 30,000 different genes in each cell, it is theoreti-
cally possible to form a very large number of different cellular proteins.
Some of the cellular proteins are structural proteins, which, in association with
various lipids and carbohydrates, form the structures of the various intracellu-
lar organelles discussed in Chapter 2. However, by far the majority of the pro-
teins are enzymes that catalyze the different chemical reactions in the cells. For
instance, enzymes promote all the oxidative reactions that supply energy to the
cell, and they promote synthesis of all the cell chemicals, such as lipids, glyco-
gen, and adenosine triphosphate (ATP).
Genes in the Cell Nucleus
In the cell nucleus, large numbers of genes are attached end on end in extremely
long double-stranded helical molecules of DNA having molecular weights
measured in the billions. A very short segment of such a molecule is shown in
Figure 3-2. This molecule is composed of several simple chemical compounds
bound together in a regular pattern, details of which are explained in the next
few paragraphs.
Basic Building Blocks of DNA. Figure 3-3 shows the basic chemical compounds
involved in the formation of DNA. These include (1) phosphoric acid, (2) a
sugar called deoxyribose, and (3) four nitrogenous bases (two purines, adenine
and guanine, and two pyrimidines, thymine and cytosine). The phosphoric acid
and deoxyribose form the two helical strands that are the backbone of the DNA
molecule, and the nitrogenous bases lie between the two strands and connect
them, as illustrated in Figure 3-6.
Nucleotides. The first stage in the formation of DNA is to combine one mole-
cule of phosphoric acid, one molecule of deoxyribose, and one of the four bases
to form an acidic nucleotide. Four separate nucleotides are thus formed, one for
each of the four bases: deoxyadenylic, deoxythymidylic, deoxyguanylic, and
deoxycytidylic acids. Figure 3-4 shows the chemical structure of deoxyadenylic
acid, and Figure 3-5 shows simple symbols for the four nucleotides that form
DNA.
Organization of the Nucleotides to Form Two Strands of DNA Loosely Bound to Each Other.
Figure 3-6 shows the manner in which multiple numbers of nucleotides are
27
28
Unit I Introduction to Physiology: The Cell and General Physiology
Gene (DNA)
RNA formation
Protein formation
Cell structure
Cell enzymes
Figure 3-2
The helical, double-stranded structure of the gene. The outside
strands are composed of phosphoric acid and the sugar deoxyri-
bose. The internal molecules connecting the two strands of the
Cell function
helix are purine and pyrimidine bases; these determine the “code”
of the gene.
Figure 3-1
General schema by which the genes control cell function.
Phosphoric acid
O
H
O
P
O
H
O
H
Deoxyribose
H
H
H
O
C
O
H
H
O
C
C
C
C
H
H
O
H
Bases
H
H
N
H
O
N
C
N C
H
C
N
H
C
O
C
C
C
H
C
C
H
N
N
C
N
H
H
H
Adenine
Thymine
H
O
N
H
N
C
N
C
C
N
H
H
C
H
O
C
C
H
C
C
N
N
N
N
C
H
H
H
Guanine
Cytosine
Purines
Pyrimidines
Figure 3-3
The basic building blocks of DNA.
Chapter 3
Genetic Control of Protein Synthesis, Cell Function, and Cell Reproduction
29
H
H
N
Adenine
N
C
C
N
A
T
H
C
C
C
H
Phosphate
N
P
D
P
D
N
H
H
Deoxyadenylic acid
Deoxythymidylic acid
O
O
C
H
Deoxyribose
H
O
P
O
C
C
C
C
O
H
H
O
H
G
C
H
P
D
P
D
Deoxyguanylic acid
Deoxycytidylic acid
Figure 3-4
Deoxyadenylic acid, one of the nucleotides that make up DNA.
Figure 3-5
Symbols for the four nucleotides that combine to form DNA.
Each nucleotide contains phosphoric acid (P), deoxyribose (D),
and one of the four nucleotide bases: A, adenine; T, thymine;
G, guanine; or C, cytosine.
C
C
G
T
C
T
G
A
A
P
D
P
D P
D
P
D P
D P
D
P
D P
D P
D
Figure 3-6
Arrangement of deoxyribose nucleotides
in a double strand of DNA.
bound together to form two strands of DNA. The two
so many times during the course of their function in
strands are, in turn, loosely bonded with each other by
the cell.
weak cross-linkages, illustrated in Figure 3-6 by the
To put the DNA of Figure 3-6 into its proper phys-
central dashed lines. Note that the backbone of each
ical perspective, one could merely pick up the two ends
DNA strand is comprised of alternating phosphoric
and twist them into a helix. Ten pairs of nucleotides
acid and deoxyribose molecules. In turn, purine and
are present in each full turn of the helix in the DNA
pyrimidine bases are attached to the sides of the
molecule, as shown in Figure 3-2.
deoxyribose molecules. Then, by means of loose
hydrogen bonds (dashed lines) between the purine
and pyrimidine bases, the two respective DNA strands
are held together. But note the following:
Genetic Code
1. Each purine base adenine of one strand always
bonds with a pyrimidine base thymine of the other
The importance of DNA lies in its ability to control
strand, and
the formation of proteins in the cell. It does this by
2. Each purine base guanine always bonds with a
means of the so-called genetic code. That is, when the
pyrimidine base cytosine.
two strands of a DNA molecule are split apart, this
Thus, in Figure 3-6, the sequence of complementary
exposes the purine and pyrimidine bases projecting to
pairs of bases is CG, CG, GC, TA, CG, TA, GC, AT, and
the side of each DNA strand, as shown by the top
AT. Because of the looseness of the hydrogen bonds,
strand in Figure 3-7. It is these projecting bases that
the two strands can pull apart with ease, and they do
form the genetic code.
30
Unit I Introduction to Physiology: The Cell and General Physiology
DNA strand
C
C
G
U
C
U
G
Figure 3-7
P
R
P R
P R
P
R
P R
P R
P
R
Triphosphate
RNA molecule
Combination of ribose nucleotides
P
with a strand of DNA to form a
P
molecule of RNA that carries the
genetic code from the gene to the
RNA polymerase
cytoplasm. The RNA polymerase
enzyme moves along the DNA
strand and builds the RNA molecule.
C
C
G
U
C
U
G
A
A
Figure 3-8
P
R
P R
P R
P R
P R
P R
P R
P R
P R
Portion of an RNA molecule, showing three RNA
“codons”—CCG, UCU, and GAA—which control
Proline
Serine
Glutamic acid
attachment of the three amino acids proline,
serine, and glutamic acid, respectively, to the
growing RNA chain.
The genetic code consists of successive “triplets” of
The RNA, in turn, diffuses from the nucleus through
bases—that is, each three successive bases is a code
nuclear pores into the cytoplasmic compartment,
word. The successive triplets eventually control the
where it controls protein synthesis.
sequence of amino acids in a protein molecule that is
to be synthesized in the cell. Note in Figure 3-6 that
the top strand of DNA, reading from left to right, has
Synthesis of RNA
the genetic code GGC, AGA, CTT, the triplets being
separated from one another by the arrows. As we
During synthesis of RNA, the two strands of the DNA
follow this genetic code through Figures 3-7 and 3-8,
molecule separate temporarily; one of these strands is
we see that these three respective triplets are respon-
used as a template for synthesis of an RNA molecule.
sible for successive placement of the three amino
The code triplets in the DNA cause formation of com-
acids, proline, serine, and glutamic acid, in a newly
plementary code triplets (called codons) in the RNA;
formed molecule of protein.
these codons, in turn, will control the sequence of
amino acids in a protein to be synthesized in the cell
cytoplasm.
The DNA Code in the Cell
Basic Building Blocks of RNA. The basic building blocks of
Nucleus Is Transferred to
RNA are almost the same as those of DNA, except for
an RNA Code in the Cell
two differences. First, the sugar deoxyribose is not used
Cytoplasm—The Process
in the formation of RNA. In its place is another sugar
of Transcription
of slightly different composition, ribose, containing an
extra hydroxyl ion appended to the ribose ring struc-
Because the DNA is located in the nucleus of the cell,
ture. Second, thymine is replaced by another pyrimi-
yet most of the functions of the cell are carried out in
dine, uracil.
the cytoplasm, there must be some means for the DNA
genes of the nucleus to control the chemical reactions
Formation of RNA Nucleotides. The basic building blocks
of the cytoplasm. This is achieved through the inter-
of RNA form RNA nucleotides, exactly as previously
mediary of another type of nucleic acid, RNA, the for-
described for DNA synthesis. Here again, four sepa-
mation of which is controlled by the DNA of the
rate nucleotides are used in the formation of RNA.
nucleus. Thus, as shown in Figure 3-7, the code is trans-
These nucleotides contain the bases adenine, guanine,
ferred to the RNA; this process is called transcription.
cytosine, and uracil. Note that these are the same bases
Chapter 3
Genetic Control of Protein Synthesis, Cell Function, and Cell Reproduction
31
as in DNA, except that uracil in RNA replaces
c. When the RNA polymerase reaches the
thymine in DNA.
end of the DNA gene, it encounters a new
sequence of DNA nucleotides called the
chain-terminating sequence; this causes the
“Activation” of the RNA Nucleotides. The next step in
polymerase and the newly formed RNA chain
the synthesis of RNA is “activation” of the RNA
to break away from the DNA strand. Then the
nucleotides by an enzyme, RNA polymerase. This
polymerase can be used again and again to
occurs by adding to each nucleotide two extra phos-
form still more new RNA chains.
phate radicals to form triphosphates (shown in Figure
d. As the new RNA strand is formed, its weak
3-7 by the two RNA nucleotides to the far right during
hydrogen bonds with the DNA template break
RNA chain formation). These last two phosphates are
away, because the DNA has a high affinity for
combined with the nucleotide by high-energy phos-
rebonding with its own complementary DNA
phate bonds derived from ATP in the cell.
strand. Thus, the RNA chain is forced away
The result of this activation process is that
from the DNA and is released into the
large quantities of ATP energy are made available to
nucleoplasm.
each of the nucleotides, and this energy is used to
Thus, the code that is present in the DNA strand is
promote the chemical reactions that add each new
eventually transmitted in complementary form to the
RNA nucleotide at the end of the developing RNA
RNA chain. The ribose nucleotide bases always
chain.
combine with the deoxyribose bases in the following
combinations:
Assembly of the RNA Chain from
Activated Nucleotides Using the DNA
DNA Base
RNA Base
Strand as a Template—The Process
of “Transcription”
guanine
cytosine
cytosine
guanine
adenine
uracil
Assembly of the RNA molecule is accomplished in
thymine
adenine
the manner shown in Figure 3-7 under the influence
of an enzyme, RNA polymerase. This is a large protein
enzyme that has many functional properties necessary
Three Different Types of RNA. There are three different
for formation of the RNA molecule. They are as
types of RNA, each of which plays an independent and
follows:
entirely different role in protein formation:
1.
In the DNA strand immediately ahead of the
1. Messenger RNA, which carries the genetic code to
initial gene is a sequence of nucleotides called
the cytoplasm for controlling the type of protein
the promoter. The RNA polymerase has an
formed.
appropriate complementary structure that
2. Transfer RNA, which transports activated amino
recognizes this promoter and becomes attached to
acids to the ribosomes to be used in assembling
it. This is the essential step for initiating formation
the protein molecule.
of the RNA molecule.
3. Ribosomal RNA, which, along with about 75
2.
After the RNA polymerase attaches to the
different proteins, forms ribosomes, the physical
promoter, the polymerase causes unwinding of
and chemical structures on which protein
about two turns of the DNA helix and separation
molecules are actually assembled.
of the unwound portions of the two strands.
3.
Then the polymerase moves along the DNA
strand, temporarily unwinding and separating the
two DNA strands at each stage of its movement.
Messenger RNA—The Codons
As it moves along, it adds at each stage a new
activated RNA nucleotide to the end of the newly
Messenger RNA molecules are long, single RNA
forming RNA chain by the following steps:
strands that are suspended in the cytoplasm. These
a. First, it causes a hydrogen bond to form
molecules are composed of several hundred to several
between the end base of the DNA strand and
thousand RNA nucleotides in unpaired strands, and
the base of an RNA nucleotide in the
they contain codons that are exactly complementary
nucleoplasm.
to the code triplets of the DNA genes. Figure 3-8
b. Then, one at a time, the RNA polymerase
shows a small segment of a molecule of messenger
breaks two of the three phosphate radicals
RNA. Its codons are CCG, UCU, and GAA. These are
away from each of these RNA nucleotides,
the codons for the amino acids proline, serine, and glu-
liberating large amounts of energy from the
tamic acid. The transcription of these codons from the
broken high-energy phosphate bonds; this
DNA molecule to the RNA molecule is shown in
energy is used to cause covalent linkage of the
Figure 3-7.
remaining phosphate on the nucleotide with
the ribose on the end of the growing RNA
RNA Codons for the Different Amino Acids. Table 3-1 gives
chain.
the RNA codons for the 20 common amino acids
32
Unit I Introduction to Physiology: The Cell and General Physiology
found in protein molecules. Note that most of the
Table 3-1
amino acids are represented by more than one codon;
RNA Codons for Amino Acids and for Start and Stop
also, one codon represents the signal “start manufac-
turing the protein molecule,” and three codons repre-
sent “stop manufacturing the protein molecule.” In
Amino Acid
RNA
Codons
Table 3-1, these two types of codons are designated CI
Alanine
GCU GCC GCA GCG
for “chain-initiating” and CT for “chain-terminating.”
Arginine
CGU CGC CGA CGG AGA
AGG
Asparagine
AAU AAC
Aspartic acid
GAU GAC
Cysteine
UGU UGC
Transfer RNA—The Anticodons
Glutamic acid GAA GAG
Glutamine
CAA CAG
Another type of RNA that plays an essential role in
Glycine
GGU GGC GGA GGG
protein synthesis is called transfer RNA, because it
Histidine
CAU CAC
Isoleucine
AUU AUC AUA
transfers amino acid molecules to protein molecules as
Leucine
CUU CUC CUA CUG UUA
UUG
the protein is being synthesized. Each type of transfer
Lysine
AAA AAG
RNA combines specifically with 1 of the 20 amino
Methionine
AUG
acids that are to be incorporated into proteins. The
Phenylalanine
UUU UUC
transfer RNA then acts as a carrier to transport its
Proline
CCU CCC CCA CCG
Serine
UCU UCC UCA UCG AGC
AGU
specific type of amino acid to the ribosomes, where
Threonine
ACU ACC ACA ACG
protein molecules are forming. In the ribosomes, each
Tryptophan
UGG
specific type of transfer RNA recognizes a particular
Tyrosine
UAU UAC
codon on the messenger RNA (described later) and
Valine
GUU GUC GUA GUG
Start (CI)
AUG
thereby delivers the appropriate amino acid to the
Stop (CT)
UAA UAG UGA
appropriate place in the chain of the newly forming
protein molecule.
CI, chain-initiating; CT, chain-terminating.
Transfer RNA, which contains only about
80
nucleotides, is a relatively small molecule in com-
parison with messenger RNA. It is a folded chain of
nucleotides with a cloverleaf appearance similar to
that shown in Figure 3-9. At one end of the molecule
messenger RNA. The specific code in the transfer
is always an adenylic acid; it is to this that the trans-
RNA that allows it to recognize a specific codon is
ported amino acid attaches at a hydroxyl group of the
again a triplet of nucleotide bases and is called an anti-
ribose in the adenylic acid.
codon. This is located approximately in the middle
Because the function of transfer RNA is to cause
of the transfer RNA molecule (at the bottom of the
attachment of a specific amino acid to a forming
cloverleaf configuration shown in Figure 3-9). During
protein chain, it is essential that each type of transfer
formation of the protein molecule, the anticodon bases
RNA also have specificity for a particular codon in the
combine loosely by hydrogen bonding with the codon
Alanine
Cysteine
Forming protein
Histidine
Alanine
Phenylalanine
Serine
Transfer RNA
Proline
Start codon
GGG
Figure 3-9
AUG
GCC
UGU
CAU
GCC
UUU
UCC
CCC
AAA
CAG
GAC
UAU
A messenger RNA strand is moving through two
ribosomes. As each “codon” passes through,
Messenger
Ribosome
Ribosome
an amino acid is added to the growing protein
RNA movement
chain, which is shown in the right-hand ribo-
some. The transfer RNA molecule transports
each specific amino acid to the newly forming
protein.
Chapter 3
Genetic Control of Protein Synthesis, Cell Function, and Cell Reproduction
33
bases of the messenger RNA. In this way, the respec-
bases called the “chain-initiating” codon. Then, as
tive amino acids are lined up one after another along
shown in Figure 3-9, while the messenger RNA travels
the messenger RNA chain, thus establishing the
through the ribosome, a protein molecule is formed—
appropriate sequence of amino acids in the newly
a process called translation. Thus, the ribosome reads
forming protein molecule.
the codons of the messenger RNA in much the same
way that a tape is “read” as it passes through the play-
back head of a tape recorder. Then, when a “stop” (or
Ribosomal RNA
“chain-terminating”) codon slips past the ribosome,
the end of a protein molecule is signaled and the
The third type of RNA in the cell is ribosomal RNA;
protein molecule is freed into the cytoplasm.
it constitutes about 60 per cent of the ribosome. The
remainder of the ribosome is protein, containing about
Polyribosomes. A single messenger RNA molecule can
75 types of proteins that are both structural proteins
form protein molecules in several ribosomes at the
and enzymes needed in the manufacture of protein
same time because the initial end of the RNA strand
molecules.
can pass to a successive ribosome as it leaves the first,
The ribosome is the physical structure in the cyto-
as shown at the bottom left in Figure 3-9 and in Figure
plasm on which protein molecules are actually syn-
3-10. The protein molecules are in different stages of
thesized. However, it always functions in association
development in each ribosome. As a result, clusters of
with the other two types of RNA as well: transfer RNA
ribosomes frequently occur, 3 to 10 ribosomes being
transports amino acids to the ribosome for incorpora-
attached to a single messenger RNA at the same time.
tion into the developing protein molecule, whereas
These clusters are called polyribosomes.
messenger RNA provides the information necessary
It is especially important to note that a messenger
for sequencing the amino acids in proper order for
RNA can cause the formation of a protein molecule
each specific type of protein to be manufactured.
in any ribosome; that is, there is no specificity of ribo-
Thus, the ribosome acts as a manufacturing plant in
somes for given types of protein. The ribosome is
which the protein molecules are formed.
simply the physical manufacturing plant in which the
chemical reactions take place.
Formation of Ribosomes in the Nucleolus. The DNA genes
Many Ribosomes Attach to the Endoplasmic Reticulum. In
for formation of ribosomal RNA are located in five
Chapter 2, it was noted that many ribosomes become
pairs of chromosomes in the nucleus, and each of these
attached to the endoplasmic reticulum. This occurs
chromosomes contains many duplicates of these par-
because the initial ends of many forming protein mol-
ticular genes because of the large amounts of riboso-
ecules have amino acid sequences that immediately
mal RNA required for cellular function.
attach to specific receptor sites on the endoplasmic
As the ribosomal RNA forms, it collects in the
reticulum; this causes these molecules to penetrate the
nucleolus, a specialized structure lying adjacent to the
reticulum wall and enter the endoplasmic reticulum
chromosomes. When large amounts of ribosomal RNA
matrix. This gives a granular appearance to those por-
are being synthesized, as occurs in cells that manufac-
tions of the reticulum where proteins are being formed
ture large amounts of protein, the nucleolus is a large
and entering the matrix of the reticulum.
structure, whereas in cells that synthesize little protein,
Figure 3-10 shows the functional relation of mes-
the nucleolus may not even be seen. Ribosomal RNA
senger RNA to the ribosomes and the manner in
is specially processed in the nucleolus, where it binds
which the ribosomes attach to the membrane of the
with “ribosomal proteins” to form granular condensa-
endoplasmic reticulum. Note the process of translation
tion products that are primordial subunits of ribo-
occurring in several ribosomes at the same time in
somes. These subunits are then released from the
response to the same strand of messenger RNA. Note
nucleolus and transported through the large pores of
also the newly forming polypeptide (protein) chains
the nuclear envelope to almost all parts of the cyto-
passing through the endoplasmic reticulum membrane
plasm. After the subunits enter the cytoplasm, they
into the endoplasmic matrix.
are assembled to form mature, functional ribosomes.
Yet it should be noted that except in glandular cells
Therefore, proteins are formed in the cytoplasm of the
in which large amounts of protein-containing secre-
cell, but not in the cell nucleus, because the nucleus
tory vesicles are formed, most proteins synthesized by
does not contain mature ribosomes.
the ribosomes are released directly into the cytosol
instead of into the endoplasmic reticulum. These pro-
teins are enzymes and internal structural proteins of
Formation of Proteins on
the cell.
the Ribosomes—The Process
of “Translation”
Chemical Steps in Protein Synthesis. Some of the chemical
events that occur in synthesis of a protein molecule are
When a molecule of messenger RNA comes in contact
shown in Figure 3-11. This figure shows representative
with a ribosome, it travels through the ribosome,
reactions for three separate amino acids, AA1, AA2,
beginning at a predetermined end of the RNA mole-
and AA20. The stages of the reactions are the follow-
cule specified by an appropriate sequence of RNA
ing: (1) Each amino acid is activated by a chemical
34
Unit I Introduction to Physiology: The Cell and General Physiology
Messenger
Small
Ribosome
Transfer RNA
RNA
subunit
Figure 3-10
Physical structure of the ribosomes,
as well as their functional relation to
messenger RNA, transfer RNA, and
the endoplasmic reticulum during
Large
the formation of protein molecules.
Amino acid
Polypeptide
subunit
(Courtesy of Dr. Don W. Fawcett,
chain
Montana.)
Amino acid
AA1
AA2
AA20
+
+
+
ATP
ATP
ATP
Activated amino acid
AMP
AA1
AMP
AA2
AMP
AA20
+
+
+
tRNA1
tRNA2
tRNA20
RNA-amino acyl complex
tRNA1
AA1
tRNA2
AA2
tRNA20
AA20
+
+
+
Messenger RNA
GCC
UGU
AAU
CAU
CGU
AUG
GUU
GCC
UGU
AAU
CAU
CGU
AUG
GUU
Complex between tRNA,
messenger RNA, and
amino acid
GTP
GTP
GTP
GTP
GTP
GTP
GTP
Protein chain
AA1
AA5
AA3
AA9
AA2
AA13
AA20
Figure 3-11
Chemical events in the formation of
a protein molecule.
process in which ATP combines with the amino acid
energy from two additional high-energy phosphate
to form an adenosine monophosphate complex with the
bonds, making a total of four high-energy bonds used
amino acid, giving up two high-energy phosphate
for each amino acid added to the protein chain. Thus,
bonds in the process. (2) The activated amino acid,
the synthesis of proteins is one of the most energy-con-
having an excess of energy, then combines with its
suming processes of the cell.
specific transfer RNA to form an amino acid-tRNA
complex and, at the same time, releases the adenosine
Peptide Linkage. The successive amino acids in the
monophosphate. (3) The transfer RNA carrying the
protein chain combine with one another according to
amino acid complex then comes in contact with the
the typical reaction:
messenger RNA molecule in the ribosome, where
the anticodon of the transfer RNA attaches tem-
porarily to its specific codon of the messenger RNA,
NH2
O
H
R
thus lining up the amino acid in appropriate sequence
to form a protein molecule. Then, under the influence
R C C OH + H N C COOH
of the enzyme peptidyl transferase (one of the proteins
in the ribosome), peptide bonds are formed between
NH2
O
H
R
the successive amino acids, thus adding progressively
R
C
C
N C COOH +
H2O
to the protein chain. These chemical events require
Chapter 3
Genetic Control of Protein Synthesis, Cell Function, and Cell Reproduction
35
In this chemical reaction, a hydroxyl radical (OH-)
There are basically two methods by which the bio-
is removed from the COOH portion of the first amino
chemical activities in the cell are controlled. One of
acid, and a hydrogen (H+) of the NH2 portion of the
these is genetic regulation, in which the degree of acti-
other amino acid is removed. These combine to form
vation of the genes themselves is controlled, and the
water, and the two reactive sites left on the two suc-
other is enzyme regulation, in which the activity levels
cessive amino acids bond with each other, resulting
of already formed enzymes in the cell are controlled.
in a single molecule. This process is called peptide
linkage. As each additional amino acid is added, an
additional peptide linkage is formed.
Genetic Regulation
The “Operon” of the Cell and Its Control of Biochemical Syn-
Synthesis of Other
thesis—Function of the Promoter. Synthesis of a cellular
Substances in the Cell
biochemical product usually requires a series of reac-
tions, and each of these reactions is catalyzed by a
Many thousand protein enzymes formed in the
special protein enzyme. Formation of all the enzymes
manner just described control essentially all the other
needed for the synthetic process often is controlled by
chemical reactions that take place in cells. These
a sequence of genes located one after the other on
enzymes promote synthesis of lipids, glycogen, purines,
the same chromosomal DNA strand. This area of the
pyrimidines, and hundreds of other substances. We
DNA strand is called an operon, and the genes respon-
discuss many of these synthetic processes in relation
sible for forming the respective enzymes are called
to carbohydrate, lipid, and protein metabolism in
structural genes. In Figure
3-12, three respective
Chapters 67 through 69. It is by means of all these
structural genes are shown in an operon, and it is
substances that the many functions of the cells are
demonstrated that they control the formation of three
performed.
respective enzymes that in turn cause synthesis of a
specific intracellular product.
Note in the figure the segment on the DNA strand
Control of Gene Function and
called the promoter. This is a group of nucleotides that
Biochemical Activity in Cells
has specific affinity for RNA polymerase, as already
discussed. The polymerase must bind with this pro-
From our discussion thus far, it is clear that the genes
moter before it can begin traveling along the DNA
control both the physical and the chemical functions
strand to synthesize RNA. Therefore, the promoter is
of the cells. However, the degree of activation of
an essential element for activating the operon.
respective genes must be controlled as well; otherwise,
some parts of the cell might overgrow or some chem-
Control of the Operon by a
“Repressor Protein”—The
ical reactions might overact until they kill the cell.
“Repressor Operator.” Also note in Figure 3-12 an addi-
Each cell has powerful internal feedback control
tional band of nucleotides lying in the middle of the
mechanisms that keep the various functional opera-
promoter. This area is called a repressor operator
tions of the cell in step with one another. For each gene
because a “regulatory” protein can bind here and
(more than 30,000 genes in all), there is at least one
prevent attachment of RNA polymerase to the pro-
such feedback mechanism.
moter, thereby blocking transcription of the genes of
Activator
Repressor
operator
operator
Operon
Structural
Structural
Structural
Promoter
Gene A
Gene B
Gene C
Enzyme A Enzyme B Enzyme C
Inhibition of
the operator
Figure 3-12
Substrates
Synthesized
Function of an operon to control synthesis of a non
product
protein intracellular product, such as an intra-
(Negative feedback)
cellular metabolic chemical. Note that the synthe-
sized product exerts negative feedback to inhibit
the function of the operon, in this way automatically
controlling the concentration of the product itself.
36
Unit I Introduction to Physiology: The Cell and General Physiology
this operon. Such a negative regulatory protein is
by still other proteins. As long as the DNA is in
called a repressor protein.
this compacted state, it cannot function to form
RNA. However, multiple control mechanisms are
Control of the Operon by an “Activator Protein”—The “Activa-
beginning to be discovered that can cause selected
tor Operator.” Note in Figure 3-12 another operator,
areas of chromosomes to become decompacted
called the activator operator, that lies adjacent to but
one part at a time so that partial RNA
ahead of the promoter. When a regulatory protein
transcription can occur. Even then, some specific
binds to this operator, it helps attract the RNA po-
“transcriptor factor” controls the actual rate of
lymerase to the promoter, in this way activating the
transcription by the separate operon in the
operon. Therefore, a regulatory protein of this type is
chromosome. Thus, still higher orders of control
called an activator protein.
are used for establishing proper cell function. In
addition, signals from outside the cell, such as
Negative Feedback Control of the Operon. Finally, note in
some of the body’s hormones, can activate specific
Figure 3-12 that the presence of a critical amount
chromosomal areas and specific transcription
of a synthesized product in the cell can cause negative
factors, thus controlling the chemical machinery
feedback inhibition of the operon that is responsible
for function of the cell.
for its synthesis. It can do this either by causing a
Because there are more than 30,000 different genes
regulatory repressor protein to bind at the repressor
in each human cell, the large number of ways in which
operator or by causing a regulatory activator protein
genetic activity can be controlled is not surprising. The
to break its bond with the activator operator. In either
gene control systems are especially important for con-
case, the operon becomes inhibited. Therefore, once
trolling intracellular concentrations of amino acids,
the required synthesized product has become abun-
amino acid derivatives, and intermediate substrates
dant enough for proper cell function, the operon
and products of carbohydrate, lipid, and protein
becomes dormant. Conversely, when the synthesized
metabolism.
product becomes degraded in the cell and its concen-
tration decreases, the operon once again becomes
Control of Intracellular Function
active. In this way, the desired concentration of the
by Enzyme Regulation
product is controlled automatically.
In addition to control of cell function by genetic
Other Mechanisms for Control of Transcription by the Operon.
regulation, some cell activities are controlled by intra-
Variations in the basic mechanism for control of the
cellular inhibitors or activators that act directly on spe-
operon have been discovered with rapidity in the past
cific intracellular enzymes. Thus, enzyme regulation
2 decades. Without giving details, let us list some of
represents a second category of mechanisms by which
them:
cellular biochemical functions can be controlled.
1. An operon frequently is controlled by a
regulatory gene located elsewhere in the genetic
Enzyme Inhibition. Some chemical substances formed in
complex of the nucleus. That is, the regulatory
the cell have direct feedback effects in inhibiting the
gene causes the formation of a regulatory protein
specific enzyme systems that synthesize them. Almost
that in turn acts either as an activator or as a
always the synthesized product acts on the first
repressor substance to control the operon.
enzyme in a sequence, rather than on the subsequent
2. Occasionally, many different operons are
enzymes, usually binding directly with the enzyme and
controlled at the same time by the same
causing an allosteric conformational change that inac-
regulatory protein. In some instances, the same
tivates it. One can readily recognize the importance of
regulatory protein functions as an activator
inactivating the first enzyme: this prevents buildup of
for one operon and as a repressor for another
intermediary products that are not used.
operon. When multiple operons are controlled
Enzyme inhibition is another example of negative
simultaneously in this manner, all the operons
feedback control; it is responsible for controlling
that function together are called a regulon.
intracellular concentrations of multiple amino acids,
3. Some operons are controlled not at the starting
purines, pyrimidines, vitamins, and other substances.
point of transcription on the DNA strand but
farther along the strand. Sometimes the control is
not even at the DNA strand itself but during the
Enzyme Activation. Enzymes that are normally inactive
processing of the RNA molecules in the nucleus
often can be activated when needed. An example of
before they are released into the cytoplasm;
this occurs when most of the ATP has been depleted
rarely, control might occur at the level of protein
in a cell. In this case, a considerable amount of cyclic
formation in the cytoplasm during RNA
adenosine monophosphate
(cAMP) begins to be
translation by the ribosomes.
formed as a breakdown product of the ATP; the pres-
4. In nucleated cells, the nuclear DNA is packaged
ence of this cAMP, in turn, immediately activates the
in specific structural units, the chromosomes.
glycogen-splitting enzyme phosphorylase, liberating
Within each chromosome, the DNA is wound
glucose molecules that are rapidly metabolized and
around small proteins called histones, which in
their energy used for replenishment of the ATP stores.
turn are held tightly together in a compacted state
Thus, cAMP acts as an enzyme activator for the
Chapter 3
Genetic Control of Protein Synthesis, Cell Function, and Cell Reproduction
37
enzyme phosphorylase and thereby helps control
Centromere
Chromosome
intracellular ATP concentration.
Nuclear
Another interesting instance of both enzyme inhi-
membrane
bition and enzyme activation occurs in the formation
of the purines and pyrimidines. These substances are
Nucleolus
needed by the cell in approximately equal quantities
for formation of DNA and RNA. When purines are
Centriole
formed, they inhibit the enzymes that are required for
Aster
formation of additional purines. However, they acti-
A
B
vate the enzymes for formation of pyrimidines. Con-
versely, the pyrimidines inhibit their own enzymes but
activate the purine enzymes. In this way, there is con-
tinual cross-feed between the synthesizing systems
for these two substances, resulting in almost exactly
equal amounts of the two substances in the cells at all
times.
C
D
Summary. In summary, there are two principal methods
by which cells control proper proportions and proper
quantities of different cellular constituents: (1) the
mechanism of genetic regulation and (2) the mecha-
nism of enzyme regulation. The genes can be either
activated or inhibited, and likewise, the enzyme
systems can be either activated or inhibited. These reg-
ulatory mechanisms most often function as feedback
E
F
control systems that continually monitor the cell’s
biochemical composition and make corrections as
needed. But on occasion, substances from without the
cell
(especially some of the hormones discussed
throughout this text) also control the intracellular
biochemical reactions by activating or inhibiting one
or more of the intracellular control systems.
G
H
Figure 3-13
The DNA-Genetic System Also
Stages of cell reproduction.
A,
B,
and
C, Prophase.
D,
Prometaphase. E, Metaphase. F, Anaphase. G and H, Telophase.
Controls Cell Reproduction
(From Margaret C. Gladbach, Estate of Mary E. and Dan Todd,
Kansas.)
Cell reproduction is another example of the ubiqui-
tous role that the DNA-genetic system plays in all life
processes. The genes and their regulatory mechanisms
determine the growth characteristics of the cells and
Except in special conditions of rapid cellular repro-
also when or whether these cells will divide to form
duction, inhibitory factors almost always slow or stop
new cells. In this way, the all-important genetic system
the uninhibited life cycle of the cell. Therefore, differ-
controls each stage in the development of the human
ent cells of the body actually have life cycle periods
being, from the single-cell fertilized ovum to the whole
that vary from as little as 10 hours for highly stimu-
functioning body. Thus, if there is any central theme to
lated bone marrow cells to an entire lifetime of the
life, it is the DNA-genetic system.
human body for most nerve cells.
Life Cycle of the Cell. The life cycle of a cell is the period
from cell reproduction to the next cell reproduction.
Cell Reproduction Begins
When mammalian cells are not inhibited and are repro-
with Replication of DNA
ducing as rapidly as they can, this life cycle may be as
little as 10 to 30 hours. It is terminated by a series of
As is true of almost all other important events in the
distinct physical events called mitosis that cause divi-
cell, reproduction begins in the nucleus itself. The first
sion of the cell into two new daughter cells. The events
step is replication (duplication) of all DNA in the chro-
of mitosis are shown in Figure 3-13 and are described
mosomes. Only after this has occurred can mitosis take
later. The actual stage of mitosis, however, lasts for
place.
only about 30 minutes, so that more than 95 per cent
The DNA begins to be duplicated some 5 to 10
of the life cycle of even rapidly reproducing cells is
hours before mitosis, and this is completed in 4 to 8
represented by the interval between mitosis, called
hours. The net result is two exact replicas of all DNA.
interphase.
These replicas become the DNA in the two new
38
Unit I Introduction to Physiology: The Cell and General Physiology
daughter cells that will be formed at mitosis. After
abnormal cellular function and sometimes even cell
replication of the DNA, there is another period of 1 to
death. Yet, given that there are 30,000 or more genes
2 hours before mitosis begins abruptly. Even during
in the human genome and that the period from one
this period, preliminary changes are beginning to take
human generation to another is about 30 years, one
place that will lead to the mitotic process.
would expect as many as 10 or many more mutations
in the passage of the genome from parent to child. As
Chemical and Physical Events of DNA Replication. DNA is
a further protection, however, each human genome is
replicated in much the same way that RNA is tran-
represented by two separate sets of chromosomes with
scribed in response to DNA, except for a few impor-
almost identical genes. Therefore, one functional gene
tant differences:
of each pair is almost always available to the child
1.
Both strands of the DNA in each chromosome
despite mutations.
are replicated, not simply one of them.
2.
Both entire strands of the DNA helix are
replicated from end to end, rather than small
Chromosomes and Their Replication
portions of them, as occurs in the transcription of
RNA.
The DNA helixes of the nucleus are packaged in chro-
3.
The principal enzymes for replicating DNA
mosomes. The human cell contains 46 chromosomes
are a complex of multiple enzymes called DNA
arranged in 23 pairs. Most of the genes in the two chro-
polymerase, which is comparable to RNA
mosomes of each pair are identical or almost identical
polymerase. It attaches to and moves along the
to each other, so it is usually stated that the different
DNA template strand while another enzyme,
genes also exist in pairs, although occasionally this is
DNA ligase, causes bonding of successive DNA
not the case.
nucleotides to one another, using high-energy
In addition to DNA in the chromosome, there is a
phosphate bonds to energize these attachments.
large amount of protein in the chromosome, composed
4.
Formation of each new DNA strand occurs
mainly of many small molecules of electropositively
simultaneously in hundreds of segments along
charged histones. The histones are organized into vast
each of the two strands of the helix until the
numbers of small, bobbin-like cores. Small segments of
entire strand is replicated. Then the ends of the
each DNA helix are coiled sequentially around one
subunits are joined together by the DNA ligase
core after another.
enzyme.
The histone cores play an important role in the reg-
5.
Each newly formed strand of DNA remains
ulation of DNA activity because as long as the DNA
attached by loose hydrogen bonding to the
is packaged tightly, it cannot function as a template for
original DNA strand that was used as its template.
either the formation of RNA or the replication of new
Therefore, two DNA helixes are coiled together.
DNA. Further, some of the regulatory proteins have
6.
Because the DNA helixes in each chromosome
been shown to decondense the histone packaging of
are approximately 6 centimeters in length and
the DNA and to allow small segments at a time to form
have millions of helix turns, it would be
RNA.
impossible for the two newly formed DNA helixes
Several nonhistone proteins are also major compo-
to uncoil from each other were it not for some
nents of chromosomes, functioning both as chromoso-
special mechanism. This is achieved by enzymes
mal structural proteins and, in connection with the
that periodically cut each helix along its entire
genetic regulatory machinery, as activators, inhibitors,
length, rotate each segment enough to cause
and enzymes.
separation, and then resplice the helix. Thus, the
Replication of the chromosomes in their entirety
two new helixes become uncoiled.
occurs during the next few minutes after replication of
the DNA helixes has been completed; the new DNA
DNA Repair, DNA “Proofreading,” and “Mutation.” During
helixes collect new protein molecules as needed. The
the hour or so between DNA replication and the
two newly formed chromosomes remain attached to
beginning of mitosis, there is a period of very active
each other (until time for mitosis) at a point called the
repair and “proofreading” of the DNA strands. That is,
centromere located near their center. These duplicated
wherever inappropriate DNA nucleotides have been
but still attached chromosomes are called chromatids.
matched up with the nucleotides of the original tem-
plate strand, special enzymes cut out the defective
areas and replace these with appropriate complemen-
Cell Mitosis
tary nucleotides. This is achieved by the same DNA
polymerases and DNA ligases that are used in repli-
The actual process by which the cell splits into two new
cation. This repair process is referred to as DNA
cells is called mitosis. Once each chromosome has
proofreading.
been replicated to form the two chromatids, in many
Because of repair and proofreading, the transcrip-
cells, mitosis follows automatically within 1 or 2 hours.
tion process rarely makes a mistake. But when a
mistake is made, this is called a mutation. The muta-
Mitotic Apparatus: Function of the Centrioles. One of the
tion causes formation of some abnormal protein in the
first events of mitosis takes place in the cytoplasm,
cell rather than a needed protein, often leading to
occurring during the latter part of interphase in or
Chapter 3
Genetic Control of Protein Synthesis, Cell Function, and Cell Reproduction
39
around the small structures called centrioles. As shown
chromosomes. One of these sets is pulled toward one
in Figure 3-13, two pairs of centrioles lie close to each
mitotic aster and the other toward the other aster as
other near one pole of the nucleus. (These centrioles,
the two respective poles of the dividing cell are pushed
like the DNA and chromosomes, were also replicated
still farther apart.
during interphase, usually shortly before replication of
the DNA.) Each centriole is a small cylindrical body
Telophase. In telophase (see Figure 3-13G and H), the
about 0.4 micrometer long and about 0.15 micrometer
two sets of daughter chromosomes are pushed com-
in diameter, consisting mainly of nine parallel tubular
pletely apart. Then the mitotic apparatus dissolutes,
structures arranged in the form of a cylinder. The two
and a new nuclear membrane develops around each
centrioles of each pair lie at right angles to each other.
set of chromosomes. This membrane is formed from
Each pair of centrioles, along with attached pericen-
portions of the endoplasmic reticulum that are already
triolar material, is called a centrosome.
present in the cytoplasm. Shortly thereafter, the cell
Shortly before mitosis is to take place, the two pairs
pinches in two, midway between the two nuclei. This is
of centrioles begin to move apart from each other. This
caused by formation of a contractile ring of micro-
is caused by polymerization of protein microtubules
filaments composed of actin and probably myosin (the
growing between the respective centriole pairs and
two contractile proteins of muscle) at the juncture of
actually pushing them apart. At the same time, other
the newly developing cells that pinches them off from
microtubules grow radially away from each of the cen-
each other.
triole pairs, forming a spiny star, called the aster, in
each end of the cell. Some of the spines of the aster
penetrate the nuclear membrane and help separate
Control of Cell Growth and
the two sets of chromatids during mitosis. The complex
Cell Reproduction
of microtubules extending between the two new cen-
triole pairs is called the spindle, and the entire set of
We know that certain cells grow and reproduce all the
microtubules plus the two pairs of centrioles is called
time, such as the blood-forming cells of the bone
the mitotic apparatus.
marrow, the germinal layers of the skin, and the epithe-
lium of the gut. Many other cells, however, such as
Prophase. The first stage of mitosis, called prophase, is
smooth muscle cells, may not reproduce for many
shown in Figure 3-13A, B, and C. While the spindle is
years. A few cells, such as the neurons and most stri-
forming, the chromosomes of the nucleus (which in
ated muscle cells, do not reproduce during the entire
interphase consist of loosely coiled strands) become
life of a person, except during the original period of
condensed into well-defined chromosomes.
fetal life.
In certain tissues, an insufficiency of some types of
Prometaphase. During this stage (see Figure 3-13D),
cells causes these to grow and reproduce rapidly until
the growing microtubular spines of the aster fragment
appropriate numbers of them are again available. For
the nuclear envelope. At the same time, multiple
instance, in some young animals, seven eighths of
microtubules from the aster attach to the chromatids
the liver can be removed surgically, and the cells of
at the centromeres, where the paired chromatids are
the remaining one eighth will grow and divide until the
still bound to each other; the tubules then pull one
liver mass returns almost to normal. The same occurs
chromatid of each pair toward one cellular pole and
for many glandular cells and most cells of the bone
its partner toward the opposite pole.
marrow, subcutaneous tissue, intestinal epithelium,
and almost any other tissue except highly differenti-
Metaphase. During metaphase (see Figure 3-13E), the
ated cells such as nerve and muscle cells.
two asters of the mitotic apparatus are pushed farther
We know little about the mechanisms that maintain
apart. This is believed to occur because the micro-
proper numbers of the different types of cells in
tubular spines from the two asters, where they inter-
the body. However, experiments have shown at least
digitate with each other to form the mitotic spindle,
three ways in which growth can be controlled. First,
actually push each other away. There is reason to
growth often is controlled by growth factors that come
believe that minute contractile protein molecules
from other parts of the body. Some of these circulate
called “motor molecules,” perhaps composed of the
in the blood, but others originate in adjacent tissues.
muscle protein actin, extend between the respective
For instance, the epithelial cells of some glands, such
spines and, using a stepping action as in muscle,
as the pancreas, fail to grow without a growth factor
actively slide the spines in a reverse direction along
from the sublying connective tissue of the gland.
each other. Simultaneously, the chromatids are pulled
Second, most normal cells stop growing when they
tightly by their attached microtubules to the very
have run out of space for growth. This occurs when
center of the cell, lining up to form the equatorial plate
cells are grown in tissue culture; the cells grow until
of the mitotic spindle.
they contact a solid object, and then growth stops.
Third, cells grown in tissue culture often stop growing
Anaphase. During this phase (see Figure 3-13F), the
when minute amounts of their own secretions are
two chromatids of each chromosome are pulled apart
allowed to collect in the culture medium. This, too,
at the centromere. All 46 pairs of chromatids are
could provide a means for negative feedback control
separated, forming two separate sets of 46 daughter
of growth.
40
Unit I Introduction to Physiology: The Cell and General Physiology
Regulation of Cell Size. Cell size is determined almost
affecting another part, and this part affecting still other
entirely by the amount of functioning DNA in the
parts.
nucleus. If replication of the DNA does not occur, the
Thus, although our understanding of cell differenti-
cell grows to a certain size and thereafter remains at
ation is still hazy, we know many control mechanisms
that size. Conversely, it is possible, by use of the chem-
by which differentiation could occur.
ical colchicine, to prevent formation of the mitotic
spindle and therefore to prevent mitosis, even though
replication of the DNA continues. In this event, the
Apoptosis—Programmed
nucleus contains far greater quantities of DNA than
it normally does, and the cell grows proportionately
Cell Death
larger. It is assumed that this results simply from
increased production of RNA and cell proteins, which
The 100 trillion cells of the body are members of a
in turn cause the cell to grow larger.
highly organized community in which the total number
of cells is regulated not only by controlling the rate of
cell division but also by controlling the rate of cell
Cell Differentiation
death. When cells are no longer needed or become a
threat to the organism, they undergo a suicidal pro-
A special characteristic of cell growth and cell division
grammed cell death, or apoptosis. This process involves
is cell differentiation, which refers to changes in
a specific proteolytic cascade that causes the cell to
physical and functional properties of cells as they pro-
shrink and condense, to disassemble its cytoskeleton,
liferate in the embryo to form the different bodily
and to alter its cell surface so that a neighboring
structures and organs. The description of an especially
phagocytic cell, such as a macrophage, can attach to
interesting experiment that helps explain these pro-
the cell membrane and digest the cell.
cesses follows.
In contrast to programmed death, cells that die as a
When the nucleus from an intestinal mucosal cell of
result of an acute injury usually swell and burst due to
a frog is surgically implanted into a frog ovum from
loss of cell membrane integrity, a process called cell
which the original ovum nucleus was removed, the
necrosis. Necrotic cells may spill their contents, causing
result is often the formation of a normal frog. This
inflammation and injury to neighboring cells. Apopto-
demonstrates that even the intestinal mucosal cell,
sis, however, is an orderly cell death that results in
which is a well-differentiated cell, carries all the
disassembly and phagocytosis of the cell before any
necessary genetic information for development of all
leakage of its contents occurs, and neighboring cells
structures required in the frog’s body.
usually remain healthy.
Therefore, it has become clear that differentiation
Apoptosis is initiated by activation of a family of
results not from loss of genes but from selective
proteases called caspases. These are enzymes that are
repression of different genetic operons. In fact, elec-
synthesized and stored in the cell as inactive procas-
tron micrographs suggest that some segments of DNA
pases. The mechanisms of activation of caspases are
helixes wound around histone cores become so con-
complex, but once activated, the enzymes cleave and
densed that they no longer uncoil to form RNA mol-
activate other procaspases, triggering a cascade that
ecules. One explanation for this is as follows: It has
rapidly breaks down proteins within the cell. The cell
been supposed that the cellular genome begins at a
thus dismantles itself, and its remains are rapidly
certain stage of cell differentiation to produce a regu-
digested by neighboring phagocytic cells.
latory protein that forever after represses a select
A tremendous amount of apoptosis occurs in tissues
group of genes. Therefore, the repressed genes never
that are being remodeled during development. Even
function again. Regardless of the mechanism, mature
in adult humans, billions of cells die each hour in
human cells produce a maximum of about 8000 to
tissues such as the intestine and bone marrow and
10,000 proteins rather than the potential 30,000 or
are replaced by new cells. Programmed cell death,
more if all genes were active.
however, is precisely balanced with the formation
Embryological experiments show that certain cells
of new cells in healthy adults. Otherwise, the body’s
in an embryo control differentiation of adjacent cells.
tissues would shrink or grow excessively. Recent
For instance, the primordial chorda-mesoderm is called
studies suggest that abnormalities of apoptosis may
the primary organizer of the embryo because it forms
play a key role in neurodegenerative diseases such as
a focus around which the rest of the embryo develops.
Alzheimer’s disease, as well as in cancer and auto-
It differentiates into a mesodermal axis that contains
immune disorders. Some drugs that have been used
segmentally arranged somites and, as a result of induc-
successfully for chemotherapy appear to induce apop-
tions in the surrounding tissues, causes formation of
tosis in cancer cells.
essentially all the organs of the body.
Another instance of induction occurs when the
developing eye vesicles come in contact with the ecto-
Cancer
derm of the head and cause the ectoderm to thicken
into a lens plate that folds inward to form the lens of
Cancer is caused in all or almost all instances by muta-
the eye. Therefore, a large share of the embryo devel-
tion or by some other abnormal activation of cellular
ops as a result of such inductions, one part of the body
genes that control cell growth and cell mitosis. The
Chapter 3
Genetic Control of Protein Synthesis, Cell Function, and Cell Reproduction
41
abnormal genes are called oncogenes. As many as 100
2.
Chemical substances of certain types also have a
different oncogenes have been discovered.
high propensity for causing mutations. It was
Also present in all cells are antioncogenes, which
discovered long ago that various aniline dye
suppress the activation of specific oncogenes. There-
derivatives are likely to cause cancer, so that
fore, loss of or inactivation of antioncogenes can allow
workers in chemical plants producing such
activation of oncogenes that lead to cancer.
substances, if unprotected, have a special
Only a minute fraction of the cells that mutate in the
predisposition to cancer. Chemical substances that
body ever lead to cancer. There are several reasons for
can cause mutation are called carcinogens. The
this. First, most mutated cells have less survival capa-
carcinogens that currently cause the greatest
bility than normal cells and simply die. Second, only
number of deaths are those in cigarette smoke.
a few of the mutated cells that do survive become
They cause about one quarter of all cancer
cancerous, because even most mutated cells still
deaths.
have normal feedback controls that prevent excessive
3.
Physical irritants also can lead to cancer, such as
growth.
continued abrasion of the linings of the intestinal
Third, those cells that are potentially cancerous are
tract by some types of food. The damage to
often, if not usually, destroyed by the body’s immune
the tissues leads to rapid mitotic replacement of
system before they grow into a cancer. This occurs in
the cells. The more rapid the mitosis, the greater
the following way: Most mutated cells form abnormal
the chance for mutation.
proteins within their cell bodies because of their
4.
In many families, there is a strong hereditary
altered genes, and these proteins activate the body’s
tendency to cancer. This results from the fact that
immune system, causing it to form antibodies or sen-
most cancers require not one mutation but two or
sitized lymphocytes that react against the cancerous
more mutations before cancer occurs. In those
cells, destroying them. In support of this is the fact
families that are particularly predisposed to
that in people whose immune systems have been sup-
cancer, it is presumed that one or more cancerous
pressed, such as in those taking immunosuppressant
genes are already mutated in the inherited
drugs after kidney or heart transplantation, the prob-
genome. Therefore, far fewer additional mutations
ability of a cancer’s developing is multiplied as much
must take place in such family members before a
as fivefold.
cancer begins to grow.
Fourth, usually several different activated onco-
5.
In laboratory animals, certain types of viruses can
genes are required simultaneously to cause a cancer.
cause some kinds of cancer, including leukemia.
For instance, one such gene might promote rapid
This usually results in one of two ways. In the case
reproduction of a cell line, but no cancer occurs
of DNA viruses, the DNA strand of the virus can
because there is not a simultaneous mutant gene to
insert itself directly into one of the chromosomes
form the needed blood vessels.
and thereby cause a mutation that leads to
But what is it that causes the altered genes? Con-
cancer. In the case of RNA viruses, some of these
sidering that many trillions of new cells are formed
carry with them an enzyme called reverse
each year in humans, a better question might be, Why
transcriptase that causes DNA to be transcribed
is it that all of us do not develop millions or billions of
from the RNA. The transcribed DNA then inserts
mutant cancerous cells? The answer is the incredible
itself into the animal cell genome, leading to
precision with which DNA chromosomal strands are
cancer.
replicated in each cell before mitosis can take place,
and also the proofreading process that cuts and repairs
Invasive Characteristic of the Cancer Cell. The major dif-
any abnormal DNA strand before the mitotic process
ferences between the cancer cell and the normal cell
is allowed to proceed. Yet, despite all these inherited
are the following: (1) The cancer cell does not respect
cellular precautions, probably one newly formed cell
usual cellular growth limits; the reason for this is that
in every few million still has significant mutant
these cells presumably do not require all the same
characteristics.
growth factors that are necessary to cause growth of
Thus, chance alone is all that is required for muta-
normal cells. (2) Cancer cells often are far less adhe-
tions to take place, so we can suppose that a large
sive to one another than are normal cells. Therefore,
number of cancers are merely the result of an unlucky
they have a tendency to wander through the tissues, to
occurrence.
enter the blood stream, and to be transported all
However, the probability of mutations can be
through the body, where they form nidi for numerous
increased manyfold when a person is exposed to
new cancerous growths. (3) Some cancers also produce
certain chemical, physical, or biological factors, includ-
angiogenic factors that cause many new blood vessels
ing the following:
to grow into the cancer, thus supplying the nutrients
1. It is well known that ionizing radiation, such as
required for cancer growth.
x-rays, gamma rays, and particle radiation from
radioactive substances, and even ultraviolet light
Why Do Cancer Cells Kill? The answer to this question
can predispose individuals to cancer. Ions formed
usually is simple. Cancer tissue competes with normal
in tissue cells under the influence of such
tissues for nutrients. Because cancer cells continue to
radiation are highly reactive and can rupture
proliferate indefinitely, their number multiplying day
DNA strands, thus causing many mutations.
by day, cancer cells soon demand essentially all the
42
Unit I Introduction to Physiology: The Cell and General Physiology
nutrition available to the body or to an essential part
Cooke MS, Evans MD, Dizdaroglu M, Lunec J: Oxidative
of the body. As a result, normal tissues gradually suffer
DNA damage: mechanisms, mutation, and disease.
nutritive death.
FASEB J 17:1195, 2003.
Cullen BR: Nuclear RNA export. J Cell Sci 116:587, 2003.
Fedier A, Fink D: Mutations in DNA mismatch repair genes:
implications for DNA damage signaling and drug sensi-
References
tivity. Int J Oncol 24:1039, 2004.
Hahn S: Structure and mechanism of the RNA polymerase
Alberts B, Johnson A, Lewis J, et al: Molecular Biology of
II transcription machinery. Nat Struct Mol Biol 11:394,
the Cell. New York: Garland Science, 2002.
2004.
Aranda A, Pascal A: Nuclear hormone receptors and gene
Hall JG: Genomic imprinting: nature and clinical relevance.
expression. Physiol Rev 81:1269, 2001.
Annu Rev Med 48:35, 1997.
Balmain A, Gray J, Ponder B: The genetics and genomics of
Jockusch BM, Hüttelmaier S, Illenberger S: From the nucleus
cancer. Nat Genet 33(Suppl):238, 2003.
toward the cell periphery: a guided tour for mRNAs. News
Bowen ID, Bowen SM, Jones AH: Mitosis and Apoptosis:
Physiol Sci 18:7, 2003.
Matters of Life and Death. London: Chapman & Hall,
Kazazian HH Jr: Mobile elements: drivers of genome
1998.
evolution. Science 303:1626, 2004.
Burke W: Genomics as a probe for disease biology. N Engl
Lewin B: Genes IV. Oxford: Oxford University Press, 2000.
J Med 349:969, 2003.
Nabel GJ: Genetic, cellular and immune approaches to
Caplen NJ, Mousses S: Short interfering RNA (siRNA)-
disease therapy: past and future. Nat Med 10:135, 2004.
mediated RNA interference (RNAi) in human cells. Ann
Pollard TD, Earnshaw WC: Cell Biology. Philadelphia:
N Y Acad Sci 1002:56, 2003.
Elsevier Science, 2002.
U N I T II
Membrane
Physiology, Nerve,
and Muscle
4. Transport of Substances Through the Cell Membrane
5. Membrane Potentials and Action Potentials
6. Contraction of Skeletal Muscle
7. Excitation of Skeletal Muscle: Neuromuscular
Transmission and Excitation-Contraction Coupling
8. Contraction and Excitation of Smooth Muscle
C H A P T E R
4
Transport of Substances Through
the Cell Membrane
Figure 4-1 gives the approximate concentrations of
important electrolytes and other substances in the
extracellular fluid and intracellular fluid. Note that
the extracellular fluid contains a large amount of
sodium but only a small amount of potassium.
Exactly the opposite is true of the intracellular fluid.
Also, the extracellular fluid contains a large amount
of chloride ions, whereas the intracellular fluid con-
tains very little. But the concentrations of phosphates and proteins in the intra-
cellular fluid are considerably greater than those in the extracellular fluid. These
differences are extremely important to the life of the cell. The purpose of this
chapter is to explain how the differences are brought about by the transport
mechanisms of the cell membranes.
The Lipid Barrier of the Cell Membrane,
and Cell Membrane Transport Proteins
The structure of the membrane covering the outside of every cell of the body
is discussed in Chapter 2 and illustrated in Figures 2-3 and 4-2. This membrane
consists almost entirely of a lipid bilayer, but it also contains large numbers of
protein molecules in the lipid, many of which penetrate all the way through the
membrane, as shown in Figure 4-2.
The lipid bilayer is not miscible with either the extracellular fluid or the intra-
cellular fluid. Therefore, it constitutes a barrier against movement of water
molecules and water-soluble substances between the extracellular and intracel-
lular fluid compartments. However, as demonstrated in Figure 4-2 by the left-
most arrow, a few substances can penetrate this lipid bilayer, diffusing directly
through the lipid substance itself; this is true mainly of lipid-soluble substances,
as described later.
The protein molecules in the membrane have entirely different properties for
transporting substances. Their molecular structures interrupt the continuity of
the lipid bilayer, constituting an alternative pathway through the cell mem-
brane. Most of these penetrating proteins, therefore, can function as transport
proteins. Different proteins function differently. Some have watery spaces all
the way through the molecule and allow free movement of water as well as
selected ions or molecules; these are called channel proteins. Others, called
carrier proteins, bind with molecules or ions that are to be transported; confor-
mational changes in the protein molecules then move the substances through
the interstices of the protein to the other side of the membrane. Both the
channel proteins and the carrier proteins are usually highly selective in the types
of molecules or ions that are allowed to cross the membrane.
“Diffusion” Versus “Active Transport.” Transport through the cell membrane, either
directly through the lipid bilayer or through the proteins, occurs by one of two
basic processes: diffusion or active transport.
Although there are many variations of these basic mechanisms, diffusion
means random molecular movement of substances molecule by molecule, either
through intermolecular spaces in the membrane or in combination with a carrier
45
46
Unit II
Membrane Physiology, Nerve, and Muscle
EXTRACELLULAR
INTRACELLULAR
FLUID
FLUID
Na+ ---------------
142 mEq/ L ---------10 mEq/ L
K+ -----------------
4 mEq/ L ------------
140 mEq/ L
Ca++ --------------
2.4 mEq/ L ----------0.0001 mEq/ L
Mg++ --------------1.2 mEq/ L ----------58 mEq/ L
Cl- ----------------
103 mEq/ L ---------4 mEq/ L
HCO3- -----------
28 mEq/ L -----------10 mEq/ L
Phosphates-----
4 mEq/ L -------------75 mEq/ L
SO4- -------------
1 mEq/L -------------2 mEq/L
Glucose ---------
90 mg/dl ------------
0 to 20 mg/dl
Amino acids ---- 30 mg/dl ------------ 200 mg/dl ?
Figure 4-3
Cholesterol
Diffusion of a fluid molecule during a thousandth of a second.
Phospholipids
0.5 g/dl-------------- 2 to 95 g/dl
Neutral fat
PO2 ---------------
35 mm Hg ---------
20 mm Hg ?
the basic physics and physical chemistry of these two
PCO2 -------------
46 mm Hg ---------
50 mm Hg ?
processes.
pH -----------------7.4 -------------------
7.0
Proteins ----------2 g/dl ----------------
16 g/dl
Diffusion
(5 mEq/ L)
(40 mEq/ L)
All molecules and ions in the body fluids, including
water molecules and dissolved substances, are in con-
stant motion, each particle moving its own separate
Figure 4-1
way. Motion of these particles is what physicists call
“heat”—the greater the motion, the higher the tem-
Chemical compositions of extracellular and intracellular fluids.
perature—and the motion never ceases under any
condition except at absolute zero temperature. When
a moving molecule, A, approaches a stationary mole-
Channel
Carrier proteins
cule, B, the electrostatic and other nuclear forces of
protein
molecule A repel molecule B, transferring some of the
energy of motion of molecule A to molecule B. Con-
sequently, molecule B gains kinetic energy of motion,
while molecule A slows down, losing some of its
kinetic energy. Thus, as shown in Figure 4-3, a single
molecule in a solution bounces among the other
molecules first in one direction, then another, then
Energy
another, and so forth, randomly bouncing thousands of
Simple
Facilitated
diffusion
diffusion
times each second. This continual movement of mole-
cules among one another in liquids or in gases is called
Diffusion
Active transport
diffusion.
Ions diffuse in the same manner as whole molecules,
Figure 4-2
and even suspended colloid particles diffuse in a
Transport pathways through the cell membrane, and the basic
similar manner, except that the colloids diffuse far
mechanisms of transport.
less rapidly than molecular substances because of their
large size.
protein. The energy that causes diffusion is the energy
of the normal kinetic motion of matter.
Diffusion Through the Cell Membrane
By contrast, active transport means movement of
ions or other substances across the membrane in com-
Diffusion through the cell membrane is divided into
bination with a carrier protein in such a way that the
two subtypes called simple diffusion and facilitated
carrier protein causes the substance to move against
diffusion. Simple diffusion means that kinetic move-
an energy gradient, such as from a low-concentration
ment of molecules or ions occurs through a membrane
state to a high-concentration state. This movement
opening or through intermolecular spaces without any
requires an additional source of energy besides kinetic
interaction with carrier proteins in the membrane.
energy. Following is a more detailed explanation of
The rate of diffusion is determined by the amount of
Chapter 4
Transport of Substances Through the Cell Membrane
47
substance available, the velocity of kinetic motion, and
diffusion directly along these channels from one side
the number and sizes of openings in the membrane
of the membrane to the other. The protein channels
through which the molecules or ions can move.
are distinguished by two important characteristics: (1)
Facilitated diffusion requires interaction of a carrier
they are often selectively permeable to certain sub-
protein. The carrier protein aids passage of the mole-
stances, and (2) many of the channels can be opened
cules or ions through the membrane by binding
or closed by gates.
chemically with them and shuttling them through the
membrane in this form.
Selective Permeability of Protein Channels. Many of the
Simple diffusion can occur through the cell mem-
protein channels are highly selective for transport of
brane by two pathways: (1) through the interstices of
one or more specific ions or molecules. This results
the lipid bilayer if the diffusing substance is lipid
from the characteristics of the channel itself, such as
soluble, and (2) through watery channels that pene-
its diameter, its shape, and the nature of the electrical
trate all the way through some of the large transport
charges and chemical bonds along its inside surfaces.
proteins, as shown to the left in Figure 4-2.
To give an example, one of the most important of the
protein channels, the so-called sodium channel, is only
Diffusion of Lipid-Soluble Substances Through the Lipid Bilayer.
0.3 by 0.5 nanometer in diameter, but more important,
One of the most important factors that determines
the inner surfaces of this channel are strongly nega-
how rapidly a substance diffuses through the lipid
tively charged, as shown by the negative signs inside
bilayer is the lipid solubility of the substance. For
the channel proteins in the top panel of Figure 4-4.
instance, the lipid solubilities of oxygen, nitrogen,
These strong negative charges can pull small dehy-
carbon dioxide, and alcohols are high, so that all these
drated sodium ions into these channels, actually pulling
can dissolve directly in the lipid bilayer and diffuse
the sodium ions away from their hydrating water
through the cell membrane in the same manner that
molecules. Once in the channel, the sodium ions
diffusion of water solutes occurs in a watery solution.
diffuse in either direction according to the usual laws
For obvious reasons, the rate of diffusion of each of
of diffusion. Thus, the sodium channel is specifically
these substances through the membrane is directly
selective for passage of sodium ions.
proportional to its lipid solubility. Especially large
Conversely, another set of protein channels is selec-
amounts of oxygen can be transported in this way;
tive for potassium transport, shown in the lower panel
therefore, oxygen can be delivered to the interior of
of Figure 4-4. These channels are slightly smaller than
the cell almost as though the cell membrane did not
the sodium channels, only 0.3 by 0.3 nanometer, but
exist.
they are not negatively charged, and their chemical
bonds are different. Therefore, no strong attrac-
Diffusion of Water and Other Lipid-Insoluble Molecules Through
tive force is pulling ions into the channels, and the
Protein Channels. Even though water is highly insoluble
potassium ions are not pulled away from the water
in the membrane lipids, it readily passes through chan-
nels in protein molecules that penetrate all the way
through the membrane. The rapidity with which water
molecules can move through most cell membranes is
astounding. As an example, the total amount of water
Outside
Gate
Na+
Na+
closed
that diffuses in each direction through the red cell
Gate open
membrane during each second is about 100 times as
-
-
-
-
great as the volume of the red cell itself.
-
-
-
-
Other lipid-insoluble molecules can pass through
-
-
-
-
the protein pore channels in the same way as water
-
-
-
–
molecules if they are water soluble and small enough.
Inside
However, as they become larger, their penetration falls
off rapidly. For instance, the diameter of the urea
molecule is only 20 per cent greater than that of water,
yet its penetration through the cell membrane pores is
about 1000 times less than that of water. Even so, given
Outside
the astonishing rate of water penetration, this amount
of urea penetration still allows rapid transport of urea
through the membrane within minutes.
Gate open
Diffusion Through Protein Channels,
Gate
and “Gating” of These Channels
Inside
closed
K+
K+
Computerized three-dimensional reconstructions of
Figure 4-4
protein channels have demonstrated tubular pathways
Transport of sodium and potassium ions through protein channels.
all the way from the extracellular to the intracellu-
Also shown are conformational changes in the protein molecules
lar fluid. Therefore, substances can move by simple
to open or close “gates” guarding the channels.
48
Unit II
Membrane Physiology, Nerve, and Muscle
molecules that hydrate them. The hydrated form of
the potassium ion is considerably smaller than the
hydrated form of sodium because the sodium ion
Open sodium channel
attracts far more water molecules than does potas-
sium. Therefore, the smaller hydrated potassium ions
3
can pass easily through this small channel, whereas the
larger hydrated sodium ions are rejected, thus provid-
ing selective permeability for a specific ion.
0
Gating of Protein Channels. Gating of protein channels
3
provides a means of controlling ion permeability of the
channels. This is shown in both panels of Figure 4-4
for selective gating of sodium and potassium ions. It is
0
believed that some of the gates are actual gatelike
extensions of the transport protein molecule, which
0
2
4
6
8
10
can close the opening of the channel or can be lifted
Milliseconds
away from the opening by a conformational change in
A
the shape of the protein molecule itself.
The opening and closing of gates are controlled in
Recorder
two principal ways:
1.
Voltage gating. In this instance, the molecular
conformation of the gate or of its chemical bonds
responds to the electrical potential across the cell
membrane. For instance, in the top panel of Figure
4-4, when there is a strong negative charge
on the inside of the cell membrane, this presumably
could cause the outside sodium gates to remain
tightly closed; conversely, when the inside of the
membrane loses its negative charge, these gates
To recorder
would open suddenly and allow tremendous
quantities of sodium to pass inward through the
sodium pores. This is the basic mechanism for
eliciting action potentials in nerves that are
responsible for nerve signals. In the bottom panel
of Figure 4-4, the potassium gates are on the
intracellular ends of the potassium channels, and
they open when the inside of the cell membrane
becomes positively charged. The opening of these
gates is partly responsible for terminating the
action potential, as is discussed more fully in
Chapter 5.
2.
Chemical (ligand) gating. Some protein channel
gates are opened by the binding of a chemical
substance (a ligand) with the protein; this causes a
conformational or chemical bonding change in the
protein molecule that opens or closes the gate. This
Membrane
is called chemical gating or ligand gating. One of
“patch”
the most important instances of chemical gating
B
is the effect of acetylcholine on the so-called
acetylcholine channel. Acetylcholine opens the gate
of this channel, providing a negatively charged pore
about 0.65 nanometer in diameter that allows
Figure 4-5
uncharged molecules or positive ions smaller
than this diameter to pass through. This gate is
A, Record of current flow through a single voltage-gated sodium
exceedingly important for the transmission of nerve
channel, demonstrating the “all or none” principle for opening and
signals from one nerve cell to another (see Chapter
closing of the channel. B, The “patch-clamp” method for record-
ing current flow through a single protein channel. To the left,
45) and from nerve cells to muscle cells to cause
recording is performed from a “patch” of a living cell membrane.
muscle contraction (see Chapter 7).
To the right, recording is from a membrane patch that has been
torn away from the cell.
Open-State Versus Closed-State of Gated Channels.
Figure 4-5A shows an especially interesting charac-
membrane. Note that the channel conducts current
teristic of most voltage-gated channels. This figure
either “all or none.” That is, the gate of the channel
shows two recordings of electrical current flowing
snaps open and then snaps closed, each open state
through a single sodium channel when there was an
lasting for only a fraction of a millisecond up to several
approximate 25-millivolt potential gradient across the
milliseconds. This demonstrates the rapidity with
Chapter 4
Transport of Substances Through the Cell Membrane
49
which changes can occur during the opening and
closing of the protein molecular gates. At one voltage
potential, the channel may remain closed all the time
Simple diffusion
or almost all the time, whereas at another voltage
Facilitated diffusion
level, it may remain open either all or most of the time.
At in-between voltages, as shown in the figure, the
gates tend to snap open and closed intermittently,
Vmax
giving an average current flow somewhere between
the minimum and the maximum.
Patch-Clamp Method for Recording Ion Current Flow Through
Single Channels. One might wonder how it is technically
possible to record ion current flow through single
protein channels as shown in Figure 4-5A. This has been
achieved by using the “patch-clamp” method illustrated
in Figure 4-5B. Very simply, a micropipette, having a tip
diameter of only 1 or 2 micrometers, is abutted against
Concentration of substance
the outside of a cell membrane. Then suction is applied
inside the pipette to pull the membrane against the tip
of the pipette. This creates a seal where the edges of the
pipette touch the cell membrane. The result is a minute
Figure 4-6
membrane “patch” at the tip of the pipette through
which electrical current flow can be recorded.
Effect of concentration of a substance on rate of diffusion through
Alternatively, as shown to the right in Figure 4-5B,
a membrane by simple diffusion and facilitated diffusion. This
the small cell membrane patch at the end of the pipette
shows that facilitated diffusion approaches a maximum rate called
can be torn away from the cell. The pipette with
the Vmax.
its sealed patch is then inserted into a free solution.
This allows the concentrations of ions both inside the
micropipette and in the outside solution to be altered
Transported
as desired. Also, the voltage between the two sides of
molecule
the membrane can be set at will—that is, “clamped” to
Binding point
a given voltage.
It has been possible to make such patches small
enough so that only a single channel protein is found
in the membrane patch being studied. By varying the
concentrations of different ions, as well as the voltage
across the membrane, one can determine the transport
characteristics of the single channel and also its gating
properties.
Carrier protein
and
conformational
change
Facilitated Diffusion
Facilitated diffusion is also called carrier-mediated dif-
fusion because a substance transported in this manner
diffuses through the membrane using a specific carrier
protein to help. That is, the carrier facilitates diffusion
of the substance to the other side.
Release
Facilitated diffusion differs from simple diffusion in
of binding
the following important way: Although the rate of
simple diffusion through an open channel increases
Figure 4-7
proportionately with the concentration of the diffus-
ing substance, in facilitated diffusion the rate of
Postulated mechanism for facilitated diffusion.
diffusion approaches a maximum, called Vmax, as the
concentration of the diffusing substance increases. This
difference between simple diffusion and facilitated dif-
pore large enough to transport a specific molecule
fusion is demonstrated in Figure 4-6. The figure shows
partway through. It also shows a binding “receptor” on
that as the concentration of the diffusing substance
the inside of the protein carrier. The molecule to be
increases, the rate of simple diffusion continues to
transported enters the pore and becomes bound. Then,
increase proportionately, but in the case of facilitated
in a fraction of a second, a conformational or chemi-
diffusion, the rate of diffusion cannot rise greater than
cal change occurs in the carrier protein, so that the
the Vmax level.
pore now opens to the opposite side of the membrane.
What is it that limits the rate of facilitated diffusion?
Because the binding force of the receptor is weak, the
A probable answer is the mechanism illustrated in
thermal motion of the attached molecule causes it to
Figure 4-7. This figure shows a carrier protein with a
break away and to be released on the opposite side of
50
Unit II
Membrane Physiology, Nerve, and Muscle
the membrane. The rate at which molecules can be
Outside
Inside
transported by this mechanism can never be greater
than the rate at which the carrier protein molecule
can undergo change back and forth between its two
states. Note specifically, though, that this mechanism
Co
Ci
allows the transported molecule to move—that is, to
“diffuse”—in either direction through the membrane.
Among the most important substances that cross
cell membranes by facilitated diffusion are glucose and
Membrane
most of the amino acids. In the case of glucose, the
A
carrier molecule has been discovered, and it has a
molecular weight of about 45,000; it can also transport
-
+
-
+
-
- -
-
-
-
several other monosaccharides that have structures
-
-
-
-
-
similar to that of glucose, including galactose. Also,
-
-
-
-
-
-
-
-
insulin can increase the rate of facilitated diffusion of
-
-
-
-
-
-
-
-
glucose as much as 10-fold to 20-fold. This is the prin-
-
-
-
-
-
-
- -
-
-
cipal mechanism by which insulin controls glucose use
-
-
-
-
-
in the body, as discussed in Chapter 78.
-
-
-
B
Factors That Affect Net Rate
of Diffusion
By now it is evident that many substances can diffuse
through the cell membrane. What is usually important
Piston
P1
P2
is the net rate of diffusion of a substance in the desired
direction. This net rate is determined by several
factors.
C
Effect of Concentration Difference on Net Diffusion Through a
Figure 4-8
Membrane. Figure 4-8A shows a cell membrane with a
substance in high concentration on the outside and low
Effect of concentration difference (A), electrical potential differ-
concentration on the inside. The rate at which the sub-
ence affecting negative ions (B), and pressure difference (C) to
cause diffusion of molecules and ions through a cell membrane.
stance diffuses inward is proportional to the con-
centration of molecules on the outside, because this
concentration determines how many molecules strike
the outside of the membrane each second. Conversely,
right, creating the condition shown in the right panel
the rate at which molecules diffuse outward is propor-
of Figure 4-8B, in which a concentration difference of
tional to their concentration inside the membrane.
the ions has developed in the direction opposite to the
Therefore, the rate of net diffusion into the cell is pro-
electrical potential difference. The concentration dif-
portional to the concentration on the outside minus
ference now tends to move the ions to the left, while
the concentration on the inside, or:
the electrical difference tends to move them to the
right. When the concentration difference rises high
Net diffusion µ (Co - Ci)
enough, the two effects balance each other. At normal
in which Co is concentration outside and Ci is concen-
body temperature (37°C), the electrical difference
tration inside.
that will balance a given concentration difference of
univalent ions—such as sodium (Na+) ions—can be
Effect of Membrane Electrical Potential on Diffusion of Ions—
determined from the following formula, called the
The
“Nernst Potential.” If an electrical potential is
Nernst equation:
applied across the membrane, as shown in Figure
4-8B, the electrical charges of the ions cause them to
EMF(in millivolts)=±
61 logC1
move through the membrane even though no concen-
C2
tration difference exists to cause movement. Thus, in
in which EMF is the electromotive force (voltage)
the left panel of Figure 4-8B, the concentration of
between side 1 and side 2 of the membrane, C1 is the
negative ions is the same on both sides of the mem-
concentration on side 1, and C2 is the concentration on
brane, but a positive charge has been applied to the
side 2. This equation is extremely important in under-
right side of the membrane and a negative charge to
standing the transmission of nerve impulses and is dis-
the left, creating an electrical gradient across the mem-
cussed in much greater detail in Chapter 5.
brane. The positive charge attracts the negative ions,
whereas the negative charge repels them. Therefore,
net diffusion occurs from left to right. After much time,
Effect of a Pressure Difference Across the Membrane. At
large quantities of negative ions have moved to the
times, considerable pressure difference develops
Chapter 4
Transport of Substances Through the Cell Membrane
51
between the two sides of a diffusible membrane.
Water
NaCl solution
This occurs, for instance, at the blood capillary mem-
brane in all tissues of the body. The pressure is about
20 mm Hg greater inside the capillary than outside.
Pressure actually means the sum of all the forces of
the different molecules striking a unit surface area at
a given instant. Therefore, when the pressure is higher
on one side of a membrane than on the other, this
means that the sum of all the forces of the molecules
striking the channels on that side of the membrane is
greater than on the other side. In most instances, this
is caused by greater numbers of molecules striking the
membrane per second on one side than on the other
side. The result is that increased amounts of energy are
available to cause net movement of molecules from
the high-pressure side toward the low-pressure side.
Osmosis
This effect is demonstrated in Figure 4-8C, which
shows a piston developing high pressure on one side
Figure 4-9
of a “pore,” thereby causing more molecules to strike
Osmosis at a cell membrane when a sodium chloride solution is
the pore on this side and, therefore, more molecules to
placed on one side of the membrane and water is placed on the
“diffuse” to the other side.
other side.
Osmosis Across Selectively
where there is pure water, than on the right side, where
Permeable Membranes—
the water concentration has been reduced. Thus, net
“Net Diffusion” of Water
movement of water occurs from left to right—that is,
osmosis occurs from the pure water into the sodium
By far the most abundant substance that diffuses
chloride solution.
through the cell membrane is water. Enough water
ordinarily diffuses in each direction through the red
Osmotic Pressure
cell membrane per second to equal about 100 times the
If in Figure 4-9 pressure were applied to the sodium
volume of the cell itself. Yet, normally, the amount that
chloride solution, osmosis of water into this solution
diffuses in the two directions is balanced so precisely
would be slowed, stopped, or even reversed. The exact
that zero net movement of water occurs. Therefore, the
amount of pressure required to stop osmosis is called
volume of the cell remains constant. However, under
the osmotic pressure of the sodium chloride solution.
certain conditions, a concentration difference for water
The principle of a pressure difference opposing
can develop across a membrane, just as concentration
osmosis is demonstrated in Figure 4-10, which shows
differences for other substances can occur. When this
a selectively permeable membrane separating two
happens, net movement of water does occur across the
columns of fluid, one containing pure water and the
cell membrane, causing the cell either to swell or to
other containing a solution of water and any solute
shrink, depending on the direction of the water move-
that will not penetrate the membrane. Osmosis of
ment. This process of net movement of water caused
water from chamber B into chamber A causes the
by a concentration difference of water is called
levels of the fluid columns to become farther and
osmosis.
farther apart, until eventually a pressure difference
To give an example of osmosis, let us assume the
develops between the two sides of the membrane great
conditions shown in Figure 4-9, with pure water on
enough to oppose the osmotic effect. The pressure dif-
one side of the cell membrane and a solution of
ference across the membrane at this point is equal to
sodium chloride on the other side. Water molecules
the osmotic pressure of the solution that contains the
pass through the cell membrane with ease, whereas
nondiffusible solute.
sodium and chloride ions pass through only with diffi-
culty. Therefore, sodium chloride solution is actually a
Importance of Number of Osmotic Particles (Molar Concentra-
mixture of permeant water molecules and nonperme-
tion) in Determining Osmotic Pressure. The osmotic pres-
ant sodium and chloride ions, and the membrane is
sure exerted by particles in a solution, whether they
said to be selectively permeable to water but much less
are molecules or ions, is determined by the number of
so to sodium and chloride ions. Yet the presence of the
particles per unit volume of fluid, not by the mass of
sodium and chloride has displaced some of the water
the particles. The reason for this is that each particle
molecules on the side of the membrane where these
in a solution, regardless of its mass, exerts, on average,
ions are present and, therefore, has reduced the con-
the same amount of pressure against the membrane.
centration of water molecules to less than that of pure
That is, large particles, which have greater mass (m)
water. As a result, in the example of Figure 4-9, more
than small particles, move at slower velocities (v). The
water molecules strike the channels on the left side,
small particles move at higher velocities in such a way
52
Unit II
Membrane Physiology, Nerve, and Muscle
osmolality of the extracellular and intracellular fluids
is about 300 milliosmoles per kilogram of water.
Relation of Osmolality to Osmotic Pressure. At normal body
temperature, 37°C, a concentration of 1 osmole per
liter will cause 19,300 mm Hg osmotic pressure in the
solution. Likewise, 1 milliosmole per liter concentra-
tion is equivalent to 19.3 mm Hg osmotic pressure.
Multiplying this value by the 300 milliosmolar con-
centration of the body fluids gives a total calculated
A
B
osmotic pressure of the body fluids of 5790 mm Hg.
The measured value for this, however, averages only
Non-
diffusible
about 5500 mm Hg. The reason for this difference is
solute
that many of the ions in the body fluids, such as sodium
Semipermeable
membrane
and chloride ions, are highly attracted to one another;
consequently, they cannot move entirely unrestrained
in the fluids and create their full osmotic pressure
Water
potential. Therefore, on average, the actual osmotic
pressure of the body fluids is about 0.93 times the cal-
culated value.
The Term “Osmolarity.” Because of the difficulty of meas-
uring kilograms of water in a solution, which is required
to determine osmolality, osmolarity, which is the
Figure 4-10
osmolar concentration expressed as osmoles per liter of
solution rather than osmoles per kilogram of water, is
Demonstration of osmotic pressure caused by osmosis at a semi-
used instead. Although, strictly speaking, it is osmoles
permeable membrane.
per kilogram of water (osmolality) that determines
osmotic pressure, for dilute solutions such as those in
the body, the quantitative differences between osmo-
that their average kinetic energies (k), determined by
larity and osmolality are less than 1 per cent. Because it
the equation
is far more practical to measure osmolarity than osmo-
lality, this is the usual practice in almost all physiologic
2
studies.
mv
k
=
2
“Active Transport” of
are the same for each small particle as for each large
Substances Through
particle. Consequently, the factor that determines the
osmotic pressure of a solution is the concentration of
Membranes
the solution in terms of number of particles (which is
the same as its molar concentration if it is a nondisso-
At times, a large concentration of a substance is
ciated molecule), not in terms of mass of the solute.
required in the intracellular fluid even though the
extracellular fluid contains only a small concentration.
“Osmolality”—The Osmole. To express the concentration
This is true, for instance, for potassium ions. Con-
of a solution in terms of numbers of particles, the unit
versely, it is important to keep the concentrations of
called the osmole is used in place of grams.
other ions very low inside the cell even though their
One osmole is 1 gram molecular weight of osmoti-
concentrations in the extracellular fluid are great. This
cally active solute. Thus, 180 grams of glucose, which
is especially true for sodium ions. Neither of these two
is 1 gram molecular weight of glucose, is equal to 1
effects could occur by simple diffusion, because simple
osmole of glucose because glucose does not dissociate
diffusion eventually equilibrates concentrations on the
into ions. Conversely, if a solute dissociates into two
two sides of the membrane. Instead, some energy
ions,
1 gram molecular weight of the solute will
source must cause excess movement of potassium ions
become 2 osmoles because the number of osmotically
to the inside of cells and excess movement of sodium
active particles is now twice as great as is the case for
ions to the outside of cells. When a cell membrane
the nondissociated solute. Therefore, when fully disso-
moves molecules or ions “uphill” against a concentra-
ciated, 1 gram molecular weight of sodium chloride,
tion gradient (or “uphill” against an electrical or pres-
58.5 grams, is equal to 2 osmoles.
sure gradient), the process is called active transport.
Thus, a solution that has 1 osmole of solute dissolved
Different substances that are actively transported
in each kilogram of water is said to have an osmolality
through at least some cell membranes include sodium
of 1 osmole per kilogram, and a solution that has
ions, potassium ions, calcium ions, iron ions, hydrogen
1/1000 osmole dissolved per kilogram has an osmo-
ions, chloride ions, iodide ions, urate ions, several dif-
lality of
1 milliosmole per kilogram. The normal
ferent sugars, and most of the amino acids.
Chapter 4
Transport of Substances Through the Cell Membrane
53
Primary Active Transport and Secondary Active Transport.
3Na+
Active transport is divided into two types according to
Outside
the source of the energy used to cause the transport:
2K+
primary active transport and secondary active trans-
port. In primary active transport, the energy is derived
directly from breakdown of adenosine triphosphate
(ATP) or of some other high-energy phosphate com-
pound. In secondary active transport, the energy is
derived secondarily from energy that has been stored
in the form of ionic concentration differences of
secondary molecular or ionic substances between the
ATPase
two sides of a cell membrane, created originally by
primary active transport. In both instances, transport
3Na+
depends on carrier proteins that penetrate through the
ATP
ADP
+
cell membrane, as is true for facilitated diffusion.
Inside
2K+
Pi
However, in active transport, the carrier protein func-
tions differently from the carrier in facilitated diffu-
Figure 4-11
sion because it is capable of imparting energy to the
transported substance to move it against the electro-
Postulated mechanism of the sodium-potassium pump. ADP,
adenosine diphosphate; ATP, adenosine triphosphate; Pi,
chemical gradient. Following are some examples
phosphate ion.
of primary active transport and secondary active
transport, with more detailed explanations of their
and three sodium ions bind on the inside, the ATPase
principles of function.
function of the protein becomes activated. This then
cleaves one molecule of ATP, splitting it to adenosine
diphosphate
(ADP) and liberating a high-energy
Primary Active Transport
phosphate bond of energy. This liberated energy is
then believed to cause a chemical and conformational
Sodium-Potassium Pump
change in the protein carrier molecule, extruding the
Among the substances that are transported by primary
three sodium ions to the outside and the two potas-
active transport are sodium, potassium, calcium,
sium ions to the inside.
hydrogen, chloride, and a few other ions.
As with other enzymes, the Na+-K+ ATPase pump
The active transport mechanism that has been
can run in reverse. If the electrochemical gradients for
studied in greatest detail is the sodium-potassium
Na+ and K+ are experimentally increased enough so
(Na+-K+) pump, a transport process that pumps
that the energy stored in their gradients is greater than
sodium ions outward through the cell membrane of all
the chemical energy of ATP hydrolysis, these ions will
cells and at the same time pumps potassium ions from
move down their concentration gradients and the Na+-
the outside to the inside. This pump is responsible for
K+ pump will synthesize ATP from ADP and phos-
maintaining the sodium and potassium concentration
phate. The phosphorylated form of the Na+-K+ pump,
differences across the cell membrane, as well as for
therefore, can either donate its phosphate to ADP to
establishing a negative electrical voltage inside the
produce ATP or use the energy to change its confor-
cells. Indeed, Chapter 5 shows that this pump is also
mation and pump Na+ out of the cell and K+ into the
the basis of nerve function, transmitting nerve signals
cell. The relative concentrations of ATP, ADP, and
throughout the nervous system.
phosphate, as well as the electrochemical gradients for
Figure 4-11 shows the basic physical components of
Na+ and K+, determine the direction of the enzyme
the Na+-K+ pump. The carrier protein is a complex of
reaction. For some cells, such as electrically active
two separate globular proteins: a larger one called the
nerve cells, 60 to 70 per cent of the cells’ energy
a subunit, with a molecular weight of about 100,000,
requirement may be devoted to pumping Na+ out of
and a smaller one called the b subunit, with a molec-
the cell and K+ into the cell.
ular weight of about 55,000. Although the function of
the smaller protein is not known (except that it might
Importance of the Na+-K+ Pump for Controlling Cell Volume.
anchor the protein complex in the lipid membrane),
One of the most important functions of the Na+-K+
the larger protein has three specific features that are
pump is to control the volume of each cell. Without
important for the functioning of the pump:
function of this pump, most cells of the body would
1. It has three receptor sites for binding sodium ions
swell until they burst. The mechanism for controlling
on the portion of the protein that protrudes to the
the volume is as follows: Inside the cell are large
inside of the cell.
numbers of proteins and other organic molecules that
2. It has two receptor sites for potassium ions on the
outside.
cannot escape from the cell. Most of these are nega-
3. The inside portion of this protein near the sodium
tively charged and therefore attract large numbers of
binding sites has ATPase activity.
potassium, sodium, and other positive ions as well. All
To put the pump into perspective: When two potas-
these molecules and ions then cause osmosis of water
sium ions bind on the outside of the carrier protein
to the interior of the cell. Unless this is checked, the
54
Unit II
Membrane Physiology, Nerve, and Muscle
cell will swell indefinitely until it bursts. The normal
In the renal tubules are special intercalated cells in
mechanism for preventing this is the Na+-K+ pump.
the late distal tubules and cortical collecting ducts that
Note again that this device pumps three Na+ ions to
also transport hydrogen ions by primary active trans-
the outside of the cell for every two K+ ions pumped
port. In this case, large amounts of hydrogen ions are
to the interior. Also, the membrane is far less perme-
secreted from the blood into the urine for the purpose
able to sodium ions than to potassium ions, so that
of eliminating excess hydrogen ions from the body
once the sodium ions are on the outside, they have a
fluids. The hydrogen ions can be secreted into the
strong tendency to stay there. Thus, this represents a
urine against a concentration gradient of about
net loss of ions out of the cell, which initiates osmosis
900-fold.
of water out of the cell as well.
If a cell begins to swell for any reason, this auto-
Energetics of Primary Active Transport
matically activates the Na+-K+ pump, moving still more
The amount of energy required to transport a sub-
ions to the exterior and carrying water with them.
stance actively through a membrane is determined by
Therefore, the Na+-K+ pump performs a continual sur-
how much the substance is concentrated during trans-
veillance role in maintaining normal cell volume.
port. Compared with the energy required to concen-
trate a substance 10-fold, to concentrate it 100-fold
Electrogenic Nature of the Na+-K+ Pump. The fact that the
requires twice as much energy, and to concentrate
Na+-K+ pump moves three Na+ ions to the exterior for
it
1000-fold requires three times as much energy.
every two K+ ions to the interior means that a net of one
In other words, the energy required is proportional
positive charge is moved from the interior of the cell to
to the logarithm of the degree that the substance
the exterior for each cycle of the pump. This creates pos-
is concentrated, as expressed by the following
itivity outside the cell but leaves a deficit of positive ions
formula:
inside the cell; that is, it causes negativity on the inside.
Therefore, the Na+-K+ pump is said to be electrogenic
because it creates an electrical potential across the cell
Energy(in calories per osmole)=
1400 logC1
membrane. As discussed in Chapter 5, this electrical
C2
potential is a basic requirement in nerve and muscle
fibers for transmitting nerve and muscle signals.
Thus, in terms of calories, the amount of energy
required to concentrate 1 osmole of substance 10-fold
Primary Active Transport of Calcium Ions
is about 1400 calories; or to concentrate it 100-fold,
Another important primary active transport mecha-
2800 calories. One can see that the energy expenditure
nism is the calcium pump. Calcium ions are normally
for concentrating substances in cells or for removing
maintained at extremely low concentration in the
substances from cells against a concentration gradient
intracellular cytosol of virtually all cells in the body, at
can be tremendous. Some cells, such as those lining the
a concentration about 10,000 times less than that in the
renal tubules and many glandular cells, expend as
extracellular fluid. This is achieved mainly by two
much as 90 per cent of their energy for this purpose
primary active transport calcium pumps. One is in the
alone.
cell membrane and pumps calcium to the outside of
the cell. The other pumps calcium ions into one or
more of the intracellular vesicular organelles of the
Secondary Active Transport—
cell, such as the sarcoplasmic reticulum of muscle cells
Co-Transport and Counter-Transport
and the mitochondria in all cells. In each of these
instances, the carrier protein penetrates the membrane
When sodium ions are transported out of cells by
and functions as an enzyme ATPase, having the same
primary active transport, a large concentration gradi-
capability to cleave ATP as the ATPase of the sodium
ent of sodium ions across the cell membrane usually
carrier protein. The difference is that this protein has
develops—high concentration outside the cell and
a highly specific binding site for calcium instead of for
very low concentration inside. This gradient represents
sodium.
a storehouse of energy because the excess sodium
outside the cell membrane is always attempting to
Primary Active Transport of Hydrogen Ions
diffuse to the interior. Under appropriate conditions,
At two places in the body, primary active transport of
this diffusion energy of sodium can pull other sub-
hydrogen ions is very important: (1) in the gastric
stances along with the sodium through the cell mem-
glands of the stomach, and (2) in the late distal tubules
brane. This phenomenon is called co-transport; it is one
and cortical collecting ducts of the kidneys.
form of secondary active transport.
In the gastric glands, the deep-lying parietal cells
For sodium to pull another substance along with it,
have the most potent primary active mechanism for
a coupling mechanism is required. This is achieved by
transporting hydrogen ions of any part of the body.
means of still another carrier protein in the cell mem-
This is the basis for secreting hydrochloric acid in the
brane. The carrier in this instance serves as an attach-
stomach digestive secretions. At the secretory ends of
ment point for both the sodium ion and the substance
the gastric gland parietal cells, the hydrogen ion con-
to be co-transported. Once they both are attached, the
centration is increased as much as a millionfold and
energy gradient of the sodium ion causes both the
then released into the stomach along with chloride
sodium ion and the other substance to be transported
ions to form hydrochloric acid.
together to the interior of the cell.
Chapter 4
Transport of Substances Through the Cell Membrane
55
In counter-transport, sodium ions again attempt to
Other important co-transport mechanisms in at
diffuse to the interior of the cell because of their large
least some cells include co-transport of chloride ions,
concentration gradient. However, this time, the sub-
iodine ions, iron ions, and urate ions.
stance to be transported is on the inside of the cell and
must be transported to the outside. Therefore, the
Sodium Counter-Transport of Calcium and
sodium ion binds to the carrier protein where it pro-
Hydrogen Ions
jects to the exterior surface of the membrane, while
Two especially important counter-transport mecha-
the substance to be counter-transported binds to the
nisms (transport in a direction opposite to the primary
interior projection of the carrier protein. Once both
ion) are sodium-calcium counter-transport and
have bound, a conformational change occurs, and
sodium-hydrogen counter-transport.
energy released by the sodium ion moving to the
Sodium-calcium counter-transport occurs through
interior causes the other substance to move to the
all or almost all cell membranes, with sodium ions
exterior.
moving to the interior and calcium ions to the exterior,
both bound to the same transport protein in a counter-
Co-Transport of Glucose and Amino Acids
transport mode. This is in addition to primary active
Along with Sodium Ions
transport of calcium that occurs in some cells.
Glucose and many amino acids are transported into
Sodium-hydrogen counter-transport occurs in
most cells against large concentration gradients; the
several tissues. An especially important example is in
mechanism of this is entirely by co-transport, as shown
the proximal tubules of the kidneys, where sodium ions
in Figure 4-12. Note that the transport carrier protein
move from the lumen of the tubule to the interior
has two binding sites on its exterior side, one for
of the tubular cell, while hydrogen ions are counter-
sodium and one for glucose. Also, the concentration of
transported into the tubule lumen. As a mechanism for
sodium ions is very high on the outside and very low
concentrating hydrogen ions, counter-transport is not
inside, which provides energy for the transport. A
nearly as powerful as the primary active transport of
special property of the transport protein is that a con-
hydrogen ions that occurs in the more distal renal
formational change to allow sodium movement to the
tubules, but it can transport extremely large numbers
interior will not occur until a glucose molecule also
of hydrogen ions, thus making it a key to hydrogen
attaches. When they both become attached, the con-
ion control in the body fluids, as discussed in detail in
formational change takes place automatically, and the
Chapter 30.
sodium and glucose are transported to the inside of the
cell at the same time. Hence, this is a sodium-glucose
co-transport mechanism.
Active Transport Through
Sodium co-transport of the amino acids occurs in the
Cellular Sheets
same manner as for glucose, except that it uses a dif-
ferent set of transport proteins. Five amino acid trans-
At many places in the body, substances must be trans-
port proteins have been identified, each of which is
ported all the way through a cellular sheet instead of
responsible for transporting one subset of amino acids
simply through the cell membrane. Transport of this
with specific molecular characteristics.
type occurs through the (1) intestinal epithelium, (2)
Sodium co-transport of glucose and amino acids
epithelium of the renal tubules, (3) epithelium of all
occurs especially through the epithelial cells of the
exocrine glands, (4) epithelium of the gallbladder, and
intestinal tract and the renal tubules of the kidneys to
(5) membrane of the choroid plexus of the brain and
promote absorption of these substances into the
other membranes.
blood, as is discussed in later chapters.
The basic mechanism for transport of a substance
through a cellular sheet is (1) active transport through
the cell membrane on one side of the transporting cells
in the sheet, and then (2) either simple diffusion or
facilitated diffusion through the membrane on the
Na+
Glucose
opposite side of the cell.
Figure
4-13 shows a mechanism for transport
Na-binding
Glucose-binding
of sodium ions through the epithelial sheet of the
site
site
intestines, gallbladder, and renal tubules. This figure
shows that the epithelial cells are connected together
tightly at the luminal pole by means of junctions called
“kisses.” The brush border on the luminal surfaces of
the cells is permeable to both sodium ions and water.
Therefore, sodium and water diffuse readily from the
lumen into the interior of the cell. Then, at the basal
and lateral membranes of the cells, sodium ions are
Na+
Glucose
actively transported into the extracellular fluid of
the surrounding connective tissue and blood vessels.
Figure 4-12
This creates a high sodium ion concentration gradient
Postulated mechanism for sodium co-transport of glucose.
across these membranes, which in turn causes osmosis
56
Unit II
Membrane Physiology, Nerve, and Muscle
Brush
Basement
References
border
membrane
Agre P, Kozono D: Aquaporin water channels: molecular
mechanisms for human diseases. FEBS Lett 555:72, 2003.
Benos DJ, Stanton BA: Functional domains within the
Na+
Na+
degenerin/epithelial sodium channel (Deg/ENaC) super-
Active
family of ion channels. J Physiol 520:631, 1999.
transport
Osmosis
Na+
Caplan MJ: Ion pump sorting in polarized renal epithelial
and
cells. Kidney Int 60:427, 2001.
O
H2
Decoursey TE: Voltage-gated proton channels and other
Active
Osmosis
proton transfer pathways. Physiol Rev 83:475, 2003.
transport
Active
De Weer P: A century of thinking about cell membranes.
Na
+
Na+
transport
Annu Rev Physiol 62:919, 2000.
Osmosis
Dolphin AC: G protein modulation of voltage-gated calcium
channels. Pharmacol Rev 55:607, 2003.
Diffusion
Jentsch TJ, Stein V, Weinreich F, Zdebik AA: Molecular
structure and physiological function of chloride channels.
Figure 4-13
Physiol Rev 82:503, 2002.
Kaupp UB, Seifert R: Cyclic nucleotide-gated ion channels.
Basic mechanism of active transport across a layer of cells.
Physiol Rev 82:769, 2002.
Kellenberger S, Schild L: Epithelial sodium channel/
degenerin family of ion channels: a variety of functions for
a shared structure. Physiol Rev 82:735, 2002.
MacKinnon R: Potassium channels. FEBS Lett 555:62, 2003.
Peres A, Giovannardi S, Bossi E, Fesce R: Electrophysiolog-
of water as well. Thus, active transport of sodium ions
ical insights into the mechanism of ion-coupled cotrans-
at the basolateral sides of the epithelial cells results in
porters. News Physiol Sci 19:80, 2004.
Philipson KD, Nicoll DA, Ottolia M, et al: The Na+/Ca2+
transport not only of sodium ions but also of water.
exchange molecule: an overview. Ann N Y Acad Sci 976:1,
These are the mechanisms by which almost all the
2002.
nutrients, ions, and other substances are absorbed into
Rossier BC, Pradervand S, Schild L, Hummler E: Epithelial
the blood from the intestine; they are also the way the
sodium channel and the control of sodium balance: inter-
same substances are reabsorbed from the glomerular
action between genetic and environmental factors. Annu
filtrate by the renal tubules.
Rev Physiol 64:877, 2002.
Throughout this text are numerous examples of the
Russell JM: Sodium-potassium-chloride cotransport. Physiol
different types of transport discussed in this chapter.
Rev 80:211, 2000.
C H A P T E R
5
Membrane Potentials and
Action Potentials
Electrical potentials exist across the membranes of
virtually all cells of the body. In addition, some cells,
such as nerve and muscle cells, are capable of gen-
erating rapidly changing electrochemical impulses
at their membranes, and these impulses are used to
transmit signals along the nerve or muscle mem-
branes. In still other types of cells, such as glandular
cells, macrophages, and ciliated cells, local changes
in membrane potentials also activate many of the cells’ functions. The present
discussion is concerned with membrane potentials generated both at rest and
during action by nerve and muscle cells.
Basic Physics of Membrane Potentials
Membrane Potentials Caused by Diffusion
“Diffusion Potential” Caused by an Ion Concentration Difference on the Two Sides of the
Membrane. In Figure 5-1A, the potassium concentration is great inside a nerve
fiber membrane but very low outside the membrane. Let us assume that the
membrane in this instance is permeable to the potassium ions but not to any
other ions. Because of the large potassium concentration gradient from inside
toward outside, there is a strong tendency for extra numbers of potassium ions
to diffuse outward through the membrane. As they do so, they carry positive
electrical charges to the outside, thus creating electropositivity outside the
membrane and electronegativity inside because of negative anions that remain
behind and do not diffuse outward with the potassium. Within a millisecond or
so, the potential difference between the inside and outside, called the diffusion
potential, becomes great enough to block further net potassium diffusion to the
exterior, despite the high potassium ion concentration gradient. In the normal
mammalian nerve fiber, the potential difference required is about 94 millivolts,
with negativity inside the fiber membrane.
Figure 5-1B shows the same phenomenon as in Figure 5-1A, but this time
with high concentration of sodium ions outside the membrane and low sodium
inside. These ions are also positively charged. This time, the membrane is highly
permeable to the sodium ions but impermeable to all other ions. Diffusion of
the positively charged sodium ions to the inside creates a membrane potential
of opposite polarity to that in Figure 5-1A, with negativity outside and positiv-
ity inside. Again, the membrane potential rises high enough within milliseconds
to block further net diffusion of sodium ions to the inside; however, this time,
in the mammalian nerve fiber, the potential is about 61 millivolts positive inside
the fiber.
Thus, in both parts of Figure 5-1, we see that a concentration difference of
ions across a selectively permeable membrane can, under appropriate condi-
tions, create a membrane potential. In later sections of this chapter, we show
that many of the rapid changes in membrane potentials observed during nerve
and muscle impulse transmission result from the occurrence of such rapidly
changing diffusion potentials.
57
58
Unit II
Membrane Physiology, Nerve, and Muscle
three factors: (1) the polarity of the electrical charge
of each ion, (2) the permeability of the membrane (P)
DIFFUSION POTENTIALS
to each ion, and (3) the concentrations (C) of the
Nerve fiber
Nerve fiber
(Anions)-
(Anions)-
respective ions on the inside (i) and outside (o) of
+
-
-
+
-
+
+
-
the membrane. Thus, the following formula, called the
(Anions)-
(Anions)-
+
-
–
+
–
+
+
-
Goldman equation, or the Goldman-Hodgkin-Katz
+
-
–
+
–
+
+
-
equation, gives the calculated membrane potential on
+
-
–
+
–
+
+
-
K+
K+
Na+
Na+
the inside of the membrane when two univalent posi-
+
-
–
+
–
+
+
-
+
-
–
+
–
+
+
-
tive ions, sodium (Na+) and potassium (K+), and one
+
-
–
+
–
+
+
-
univalent negative ion, chloride (Cl-), are involved.
(- 94 mV)
-
+
(+ 61 mV)
+
-
+
-
–
+
–
+
+
-
EMF (millivolts)
+
-
-
+
-
+
+
-
C
P
+
+C
P
+
+C
-
P
-
Na+i Na
K+i K
Cl
o
Cl
=-61◊log
A
B
C P ++C P ++C P
-
Na+o Na
K o K
Cl
i
Cl
Let us study the importance and the meaning of this
Figure 5-1
equation. First, sodium, potassium, and chloride ions
are the most important ions involved in the develop-
A, Establishment of a “diffusion” potential across a nerve fiber
ment of membrane potentials in nerve and muscle
membrane, caused by diffusion of potassium ions from inside the
fibers, as well as in the neuronal cells in the nervous
cell to outside through a membrane that is selectively permeable
system. The concentration gradient of each of these
only to potassium. B, Establishment of a “diffusion potential” when
the nerve fiber membrane is permeable only to sodium ions. Note
ions across the membrane helps determine the voltage
that the internal membrane potential is negative when potassium
of the membrane potential.
ions diffuse and positive when sodium ions diffuse because of
Second, the degree of importance of each of the ions
opposite concentration gradients of these two ions.
in determining the voltage is proportional to the mem-
brane permeability for that particular ion. That is, if the
Relation of the Diffusion Potential to the Concentration Differ-
membrane has zero permeability to both potassium
ence—The Nernst Potential. The diffusion potential level
and chloride ions, the membrane potential becomes
across a membrane that exactly opposes the net diffu-
entirely dominated by the concentration gradient of
sion of a particular ion through the membrane is called
sodium ions alone, and the resulting potential will be
the Nernst potential for that ion, a term that was intro-
equal to the Nernst potential for sodium. The same
duced in Chapter 4. The magnitude of this Nernst
holds for each of the other two ions if the membrane
potential is determined by the ratio of the concentra-
should become selectively permeable for either one of
tions of that specific ion on the two sides of the mem-
them alone.
brane. The greater this ratio, the greater the tendency
Third, a positive ion concentration gradient from
for the ion to diffuse in one direction, and therefore
inside the membrane to the outside causes electroneg-
the greater the Nernst potential required to prevent
ativity inside the membrane. The reason for this is that
additional net diffusion. The following equation, called
excess positive ions diffuse to the outside when their
the Nernst equation, can be used to calculate the
concentration is higher inside than outside. This carries
Nernst potential for any univalent ion at normal body
positive charges to the outside but leaves the nondif-
temperature of 98.6°F (37°C):
fusible negative anions on the inside, thus creating
electronegativity on the inside. The opposite effect
Concentration inside
EMF (millivolts) = ± 61log
occurs when there is a gradient for a negative ion. That
Concentration outside
is, a chloride ion gradient from the outside to the inside
where EMF is electromotive force.
causes negativity inside the cell because excess nega-
When using this formula, it is usually assumed that
tively charged chloride ions diffuse to the inside, while
the potential in the extracellular fluid outside the
leaving the nondiffusible positive ions on the outside.
membrane remains at zero potential, and the Nernst
Fourth, as explained later, the permeability of the
potential is the potential inside the membrane. Also,
sodium and potassium channels undergoes rapid
the sign of the potential is positive (+) if the ion dif-
changes during transmission of a nerve impulse,
fusing from inside to outside is a negative ion, and it
whereas the permeability of the chloride channels
is negative (-) if the ion is positive. Thus, when the con-
does not change greatly during this process. Therefore,
centration of positive potassium ions on the inside is
rapid changes in sodium and potassium permeability
10 times that on the outside, the log of 10 is 1, so that
are primarily responsible for signal transmission in
the Nernst potential calculates to be -61 millivolts
nerves, which is the subject of most of the remainder
inside the membrane.
of this chapter.
Calculation of the Diffusion Potential
Measuring the Membrane
When the Membrane Is Permeable to
Potential
Several Different Ions
When a membrane is permeable to several different
The method for measuring the membrane potential is
ions, the diffusion potential that develops depends on
simple in theory but often difficult in practice because
Chapter 5
Membrane Potentials and Action Potentials
59
Nerve fiber
- + - + - + - + - + - + - + -
+ - + + - - + - + - - + + - +
0
- + - + - + - + - + - + - + -
+ - + + - - + - + - - + + - +
– + - + - + - + - + - + - + -
—
+
+ - + + - - + - + - - + + - +
- + - + - + - + - + - + - + -
+ - + + - - + - + - - + + - +
- + - + - + - + - + - + - + -
+ - + + - - + - + - - + + - +
Silver-silver
chloride
+ + + + + + + + + + +
+ + + + +
0
- - - - - - - - - -
- - - - -
electrode
(- 90
– - - - - - - - -
- - - - - - -
+ + + + + + + + +
mV)+ + + + + + + +
-90
Figure 5-2
Figure 5-3
Measurement of the membrane potential of the nerve fiber using
a microelectrode.
Distribution of positively and negatively charged ions in the extra-
cellular fluid surrounding a nerve fiber and in the fluid inside the
fiber; note the alignment of negative charges along the inside
of the small size of most of the fibers. Figure 5-2 shows
surface of the membrane and positive charges along the outside
a small pipette filled with an electrolyte solution. The
surface. The lower panel displays the abrupt changes in mem-
brane potential that occur at the membranes on the two sides of
pipette is impaled through the cell membrane to
the fiber.
the interior of the fiber. Then another electrode, called
the “indifferent electrode,” is placed in the extracellular
fluid, and the potential difference between the inside
and outside of the fiber is measured using an appropri-
equally small number of positive ions moving from
ate voltmeter. This voltmeter is a highly sophisticated
outside to inside the fiber can reverse the potential from
electronic apparatus that is capable of measuring very
-90 millivolts to as much as +35 millivolts within as little
small voltages despite extremely high resistance to elec-
as 1/10,000 of a second. Rapid shifting of ions in this
trical flow through the tip of the micropipette, which
manner causes the nerve signals discussed in subse-
has a lumen diameter usually less than 1 micrometer
quent sections of this chapter.
and a resistance more than a million ohms. For record-
ing rapid changes in the membrane potential during
Resting Membrane Potential
transmission of nerve impulses, the microelectrode is
connected to an oscilloscope, as explained later in the
of Nerves
chapter.
The lower part of Figure 5-3 shows the electrical
The resting membrane potential of large nerve fibers
potential that is measured at each point in or near the
when not transmitting nerve signals is about
-90
nerve fiber membrane, beginning at the left side of the
millivolts. That is, the potential inside the fiber is 90
figure and passing to the right. As long as the electrode
millivolts more negative than the potential in the extra-
is outside the nerve membrane, the recorded potential
cellular fluid on the outside of the fiber. In the next few
is zero, which is the potential of the extracellular fluid.
Then, as the recording electrode passes through the
paragraphs, we explain all the factors that determine
voltage change area at the cell membrane (called the
the level of this resting potential, but before doing so,
electrical dipole layer), the potential decreases abruptly
we must describe the transport properties of the resting
to -90 millivolts. Moving across the center of the fiber,
nerve membrane for sodium and potassium.
the potential remains at a steady -90-millivolt level but
reverses back to zero the instant it passes through the
Active Transport of Sodium and Potassium Ions Through the
membrane on the opposite side of the fiber.
Membrane—The Sodium-Potassium (Na+-K+) Pump. First, let
To create a negative potential inside the membrane,
us recall from Chapter 4 that all cell membranes of the
only enough positive ions to develop the electrical
body have a powerful Na+-K+ that continually pumps
dipole layer at the membrane itself must be transported
sodium ions to the outside of the cell and potassium
outward. All the remaining ions inside the nerve fiber
ions to the inside, as illustrated on the left-hand side in
can be both positive and negative, as shown in the upper
Figure 5-4. Further, note that this is an electrogenic
panel of Figure 5-3. Therefore, an incredibly small
pump because more positive charges are pumped to
number of ions needs to be transferred through the
the outside than to the inside (three Na+ ions to the
membrane to establish the normal “resting potential” of
-90 millivolts inside the nerve fiber; this means that only
outside for each two K+ ions to the inside), leaving a
about 1/3,000,000 to 1/100,000,000 of the total positive
net deficit of positive ions on the inside; this causes a
charges inside the fiber needs to be transferred. Also, an
negative potential inside the cell membrane.
60
Unit II
Membrane Physiology, Nerve, and Muscle
Outside
3Na+
2K+
Na+
K+
K+
4 mEq/L
K+
140 mEq/L
(-94 mV)
(-94 mV)
Na+
K+
ATP
Na+
K+
ADP
A
K+ -Na+
Na+
K+
Na+-K+ pump
"leak" channels
142 mEq/L
4 mEq/L
Figure 5-4
Na+
K+
Functional characteristics of the Na+-K+ pump and of the K+-Na+
14 mEq/L
140 mEq/L
(-86 mV)
“leak” channels. ADP, adenosine diphosphate; ATP, adenosine
triphosphate.
(+61 mV)
(-94 mV)
B
+
-
-
+
The Na+-K+ also causes large concentration gradi-
+
-
-
+
ents for sodium and potassium across the resting nerve
Diffusion
-
+
membrane. These gradients are the following:
+
-
-
+
Na+
Na+
Na+ (outside):
142 mEq/L
pump
-
+
+
-
-
+
Na+ (inside):
14 mEq/L
+
-
-
+
142 mEq/L
14 mEq/L
K+ (outside):
4 mEq/L
+
-
-
+
+
-
-
+
K+ (inside):
140 mEq/L
-
+
Diffusion
The ratios of these two respective ions from the inside
-
+
to the outside are
+
-
-
+
K+
K+
pump
-
+
Na+inside/Na+outside = 0.1
+
-
-
+
4 mEq/L
140 mEq/L
K+inside/K+outside = 35.0
+
-
-
+
+
-
-
+
(-90 mV)
+
-
-
+
Leakage of Potassium and Sodium Through the Nerve Mem-
+
-
-
+
brane. The right side of Figure 5-4 shows a channel
(Anions)-
(Anions)-
+
-
-
+
protein in the nerve membrane through which potas-
C
sium and sodium ions can leak, called a potassium-
sodium (K+-Na+) “leak” channel. The emphasis is on
potassium leakage because, on average, the channels
are far more permeable to potassium than to sodium,
Figure 5-5
normally about 100 times as permeable. As discussed
later, this differential in permeability is exceedingly
Establishment of resting membrane potentials in nerve
fibers
under three conditions: A, when the membrane potential is caused
important in determining the level of the normal
entirely by potassium diffusion alone; B, when the membrane
resting membrane potential.
potential is caused by diffusion of both sodium and potassium
ions; and C, when the membrane potential is caused by diffusion
of both sodium and potassium ions plus pumping of both these
Origin of the Normal Resting
ions by the Na+-K+ pump.
Membrane Potential
Figure 5-5 shows the important factors in the estab-
potential corresponding to this ratio is -94 millivolts
lishment of the normal resting membrane potential of
because the logarithm of 35 is 1.54, and this times -61
-90 millivolts. They are as follows.
millivolts is -94 millivolts. Therefore, if potassium ions
were the only factor causing the resting potential, the
Contribution of the Potassium Diffusion Potential. In Figure
resting potential inside the fiber would be equal to -94
5-5A, we make the assumption that the only move-
millivolts, as shown in the figure.
ment of ions through the membrane is diffusion of
potassium ions, as demonstrated by the open channels
Contribution of Sodium Diffusion Through the Nerve Membrane.
between the potassium symbols
(K+) inside and
Figure 5-5B shows the addition of slight permeability
outside the membrane. Because of the high ratio of
of the nerve membrane to sodium ions, caused by the
potassium ions inside to outside, 35:1, the Nernst
minute diffusion of sodium ions through the K+-Na+
Chapter 5
Membrane Potentials and Action Potentials
61
leak channels. The ratio of sodium ions from inside to
outside the membrane is 0.1, and this gives a calculated
Nernst potential for the inside of the membrane of
0
+61 millivolts. But also shown in Figure 5-5B is the
Nernst potential for potassium diffusion of -94 milli-
—
+
volts. How do these interact with each other, and what
will be the summated potential? This can be answered
by using the Goldman equation described previously.
Silver-silver
Intuitively, one can see that if the membrane is highly
chloride
+ + + +
– - - -
+ + + + +
electrode
permeable to potassium but only slightly permeable to
– - - -
- - - - -
+ + + +
sodium, it is logical that the diffusion of potassium con-
tributes far more to the membrane potential than does
+ + + +
- - - -
- - - - - -
the diffusion of sodium. In the normal nerve fiber, the
+ + + +
- - - -
+ + + + + +
permeability of the membrane to potassium is about
100 times as great as its permeability to sodium. Using
this value in the Goldman equation gives a potential
Overshoot
inside the membrane of -86 millivolts, which is near
+35
the potassium potential shown in the figure.
Contribution of the Na+-K+ Pump. In Figure
5-5C, the
0
Na+-K+ pump is shown to provide an additional con-
tribution to the resting potential. In this figure, there
is continuous pumping of three sodium ions to the
outside for each two potassium ions pumped to the
inside of the membrane. The fact that more sodium
ions are being pumped to the outside than potassium
-90
to the inside causes continual loss of positive charges
Resting
from inside the membrane; this creates an additional
degree of negativity (about -4 millivolts additional) on
0
0.1 0.2 0.3 0.4 0.5 0.6 0.7
the inside beyond that which can be accounted for by
Milliseconds
diffusion alone. Therefore, as shown in Figure 5-5C,
the net membrane potential with all these factors
operative at the same time is about -90 millivolts.
In summary, the diffusion potentials alone caused by
Figure 5-6
potassium and sodium diffusion would give a mem-
brane potential of about -86 millivolts, almost all of
Typical action potential recorded by the method shown in the
upper panel of the figure.
this being determined by potassium diffusion. Then, an
additional -4 millivolts is contributed to the mem-
brane potential by the continuously acting electro-
genic Na+-K+ pump, giving a net membrane potential
explosive onset of the action potential and the
of -90 millivolts.
almost equally rapid recovery.
The successive stages of the action potential are as
follows.
Nerve Action Potential
Resting Stage. This is the resting membrane potential
Nerve signals are transmitted by action potentials,
before the action potential begins. The membrane is
which are rapid changes in the membrane potential
said to be “polarized” during this stage because of the
that spread rapidly along the nerve fiber membrane.
-90 millivolts negative membrane potential that is
Each action potential begins with a sudden change
present.
from the normal resting negative membrane potential
to a positive potential and then ends with an almost
Depolarization Stage. At this time, the membrane sud-
equally rapid change back to the negative potential.
denly becomes very permeable to sodium ions, allow-
To conduct a nerve signal, the action potential moves
ing tremendous numbers of positively charged sodium
along the nerve fiber until it comes to the fiber’s
ions to diffuse to the interior of the axon. The normal
end.
“polarized” state of -90 millivolts is immediately neu-
The upper panel of Figure 5-6 shows the changes
tralized by the inflowing positively charged sodium
that occur at the membrane during the action poten-
ions, with the potential rising rapidly in the positive
tial, with transfer of positive charges to the interior of
direction. This is called depolarization. In large nerve
the fiber at its onset and return of positive charges to
fibers, the great excess of positive sodium ions moving
the exterior at its end. The lower panel shows graphi-
to the inside causes the membrane potential to actu-
cally the successive changes in membrane potential
ally “overshoot” beyond the zero level and to become
over a few 10,000ths of a second, illustrating the
somewhat positive. In some smaller fibers, as well as in
62
Unit II
Membrane Physiology, Nerve, and Muscle
many central nervous system neurons, the potential
Activation
merely approaches the zero level and does not over-
gate
Na+
Na+
Na+
shoot to the positive state.
Repolarization Stage. Within a few 10,000ths of a second
after the membrane becomes highly permeable to
sodium ions, the sodium channels begin to close and
the potassium channels open more than normal.
Inactivation
Then, rapid diffusion of potassium ions to the exterior
gate
re-establishes the normal negative resting mem-
Resting
Activated
Inactivated
brane potential. This is called repolarization of the
(-90 mV)
(-90 to +35 mV)
(+35 to -90 mV,
membrane.
delayed)
To explain more fully the factors that cause both
depolarization and repolarization, we need to describe
the special characteristics of two other types of trans-
port channels through the nerve membrane: the
voltage-gated sodium and potassium channels.
K+
K+
Voltage-Gated Sodium and
Resting
Slow activation
(-90 mV)
(+35 to -90 mV)
Potassium Channels
Inside
Figure 5-7
The necessary actor in causing both depolarization
and repolarization of the nerve membrane during the
Characteristics of the voltage-gated sodium (top) and potassium
action potential is the voltage-gated sodium channel. A
(bottom) channels, showing successive activation and inactiva-
voltage-gated potassium channel also plays an impor-
tion of the sodium channels and delayed activation of the potas-
sium channels when the membrane potential is changed from the
tant role in increasing the rapidity of repolarization of
normal resting negative value to a positive value.
the membrane. These two voltage-gated channels are
in addition to the Na+-K+ pump and the K+-Na+ leak
channels.
Voltage-Gated Sodium Channel—Activation
conformational change that flips the inactivation gate
and Inactivation of the Channel
to the closed state is a slower process than the con-
The upper panel of Figure 5-7 shows the voltage-gated
formational change that opens the activation gate.
sodium channel in three separate states. This channel
Therefore, after the sodium channel has remained
has two gates—one near the outside of the channel
open for a few 10,000ths of a second, the inactivation
called the activation gate, and another near the inside
gate closes, and sodium ions no longer can pour to the
called the inactivation gate. The upper left of the figure
inside of the membrane. At this point, the membrane
depicts the state of these two gates in the normal
potential begins to recover back toward the resting
resting membrane when the membrane potential is
membrane state, which is the repolarization process.
-90 millivolts. In this state, the activation gate is closed,
Another important characteristic of the sodium
which prevents any entry of sodium ions to the inte-
channel inactivation process is that the inactivation
rior of the fiber through these sodium channels.
gate will not reopen until the membrane potential
returns to or near the original resting membrane
Activation of the Sodium Channel. When the membrane
potential level. Therefore, it usually is not possible for
potential becomes less negative than during the
the sodium channels to open again without the nerve
resting state, rising from -90 millivolts toward zero, it
fiber’s first repolarizing.
finally reaches a voltage—usually somewhere between
-70 and -50 millivolts—that causes a sudden confor-
Voltage-Gated Potassium Channel and
mational change in the activation gate, flipping it all
Its Activation
the way to the open position. This is called the acti-
The lower panel of Figure 5-7 shows the voltage-gated
vated state; during this state, sodium ions can pour
potassium channel in two states: during the resting
inward through the channel, increasing the sodium
state (left) and toward the end of the action potential
permeability of the membrane as much as 500- to
(right). During the resting state, the gate of the potas-
5000-fold.
sium channel is closed, and potassium ions are pre-
vented from passing through this channel to the
Inactivation of the Sodium Channel. The upper right panel
exterior. When the membrane potential rises from
of Figure
5-7 shows a third state of the sodium
-90 millivolts toward zero, this voltage change causes
channel. The same increase in voltage that opens the
a conformational opening of the gate and allows
activation gate also closes the inactivation gate. The
increased potassium diffusion outward through the
inactivation gate, however, closes a few 10,000ths of
channel. However, because of the slight delay in
a second after the activation gate opens. That is, the
opening of the potassium channels, for the most part,
Chapter 5
Membrane Potentials and Action Potentials
63
they open just at the same time that the sodium chan-
Figure 5-8 shows an experimental apparatus called a
nels are beginning to close because of inactivation.
voltage clamp, which is used to measure flow of ions
Thus, the decrease in sodium entry to the cell and the
through the different channels. In using this apparatus,
two electrodes are inserted into the nerve fiber. One of
simultaneous increase in potassium exit from the cell
these is to measure the voltage of the membrane poten-
combine to speed the repolarization process, leading
tial, and the other is to conduct electrical current into
to full recovery of the resting membrane potential
or out of the nerve fiber. This apparatus is used in the
within another few 10,000ths of a second.
following way: The investigator decides which voltage
he or she wants to establish inside the nerve fiber. The
Research Method for Measuring the Effect of Voltage on Opening
electronic portion of the apparatus is then adjusted to
and Closing of the Voltage-Gated Channels—The
“Voltage
the desired voltage, and this automatically injects either
Clamp.” The original research that led to quantitative
positive or negative electricity through the current elec-
understanding of the sodium and potassium channels
trode at whatever rate is required to hold the voltage,
was so ingenious that it led to Nobel Prizes for the sci-
as measured by the voltage electrode, at the level set by
entists responsible, Hodgkin and Huxley. The essence of
the operator. When the membrane potential is suddenly
these studies is shown in Figures 5-8 and 5-9.
increased by this voltage clamp from -90 millivolts to
zero, the voltage-gated sodium and potassium channels
open, and sodium and potassium ions begin to pour
through the channels. To counterbalance the effect of
these ion movements on the desired setting of the intra-
cellular voltage, electrical current is injected automati-
Amplifier
cally through the current electrode of the voltage clamp
to maintain the intracellular voltage at the required
steady zero level. To achieve this, the current injected
must be equal to but of opposite polarity to the net
Electrode
current flow through the membrane channels. To
in fluid
measure how much current flow is occurring at each
Voltage
Current
instant, the current electrode is connected to an oscillo-
electrode
electrode
scope that records the current flow, as demonstrated on
the screen of the oscilloscope in Figure 5-8. Finally, the
investigator adjusts the concentrations of the ions to
other than normal levels both inside and outside the
nerve fiber and repeats the study. This can be done
easily when using large nerve fibers removed from some
Figure 5-8
crustaceans, especially the giant squid axon, which in
“Voltage clamp” method for studying flow of ions through specific
some cases is as large as 1 millimeter in diameter. When
channels.
sodium is the only permeant ion in the solutions inside
and outside the squid axon, the voltage clamp measures
current flow only through the sodium channels. When
potassium is the only permeant ion, current flow only
through the potassium channels is measured.
Another means for studying the flow of ions through
an individual type of channel is to block one type of
channel at a time. For instance, the sodium channels can
Na+ channel
K+ channel
be blocked by a toxin called tetrodotoxin by applying it
to the outside of the cell membrane where the sodium
30
activation gates are located. Conversely, tetraethylam-
20
monium ion blocks the potassium channels when it is
applied to the interior of the nerve fiber.
10
Figure 5-9 shows typical changes in conductance of
the voltage-gated sodium and potassium channels when
0
the membrane potential is suddenly changed by use of
- 90 mV
+10 mV
- 90 mV
the voltage clamp from -90 millivolts to +10 millivolts
Membrane potential
and then, 2 milliseconds later, back to -90 millivolts.
Note the sudden opening of the sodium channels (the
0
1
2
3
activation stage) within a small fraction of a millisecond
Time (milliseconds)
after the membrane potential is increased to the posi-
tive value. However, during the next millisecond or so,
the sodium channels automatically close (the inactiva-
Figure 5-9
tion stage).
Note the opening (activation) of the potassium chan-
Typical changes in conductance of sodium and potassium ion
nels. These open slowly and reach their full open state
channels when the membrane potential is suddenly increased
only after the sodium channels have almost completely
from the normal resting value of -90 millivolts to a positive value
closed. Further, once the potassium channels open, they
of +10 millivolts for 2 milliseconds. This figure shows that the
remain open for the entire duration of the positive
sodium channels open (activate) and then close
(inactivate)
membrane potential and do not close again until after
before the end of the 2 milliseconds, whereas the potassium chan-
the membrane potential is decreased back to a negative
nels only open (activate), and the rate of opening is much slower
than that of the sodium channels.
value.
64
Unit II
Membrane Physiology, Nerve, and Muscle
Summary of the Events That Cause
close back to their original status, but again, only after
an additional millisecond or more delay.
the Action Potential
The middle portion of Figure 5-10 shows the ratio
of sodium conductance to potassium conductance at
Figure 5-10 shows in summary form the sequential
each instant during the action potential, and above this
events that occur during and shortly after the action
is the action potential itself. During the early portion
potential. The bottom of the figure shows the changes
of the action potential, the ratio of sodium to potas-
in membrane conductance for sodium and potassium
sium conductance increases more than
1000-fold.
ions. During the resting state, before the action
Therefore, far more sodium ions flow to the interior of
potential begins, the conductance for potassium ions is
the fiber than do potassium ions to the exterior. This
50 to 100 times as great as the conductance for sodium
is what causes the membrane potential to become
ions. This is caused by much greater leakage of potas-
positive at the action potential onset. Then the sodium
sium ions than sodium ions through the leak channels.
channels begin to close and the potassium channels to
However, at the onset of the action potential, the
open, so that the ratio of conductance shifts far in
sodium channels instantaneously become activated
favor of high potassium conductance but low sodium
and allow up to a 5000-fold increase in sodium con-
conductance. This allows very rapid loss of potassium
ductance. Then the inactivation process closes the
ions to the exterior but virtually zero flow of
sodium channels within another fraction of a millisec-
sodium ions to the interior. Consequently, the action
ond. The onset of the action potential also causes
potential quickly returns to its baseline level.
voltage gating of the potassium channels, causing them
to begin opening more slowly a fraction of a millisec-
ond after the sodium channels open. At the end of the
Roles of Other Ions During
action potential, the return of the membrane potential
to the negative state causes the potassium channels to
the Action Potential
Thus far, we have considered only the roles of sodium
and potassium ions in the generation of the action
potential. At least two other types of ions must be con-
sidered: negative anions and calcium ions.
Impermeant Negatively Charged Ions (Anions) Inside the Nerve
Axon. Inside the axon are many negatively charged ions
+ 60
Overshoot
that cannot go through the membrane channels. They
+ 40
+ 20
include the anions of protein molecules and of many
100
0
organic phosphate compounds, sulfate compounds, and
- 20
so forth. Because these ions cannot leave the interior of
10
- 40
the axon, any deficit of positive ions inside the mem-
- 60
brane leaves an excess of these impermeant negative
- 80
1
anions. Therefore, these impermeant negative ions are
-100
responsible for the negative charge inside the fiber
0.1
Positive
when there is a net deficit of positively charged potas-
afterpotential
sium ions and other positive ions.
0.01
0.001
Calcium Ions. The membranes of almost all cells of the
Action potential
100
body have a calcium pump similar to the sodium pump,
Ratio of conductances
and calcium serves along with (or instead of) sodium in
Na+
10
K+
some cells to cause most of the action potential. Like
the sodium pump, the calcium pump pumps calcium
1
ions from the interior to the exterior of the cell mem-
brane (or into the endoplasmic reticulum of the cell),
0.1
creating a calcium ion gradient of about 10,000-fold.
This leaves an internal cell concentration of calcium
0.01
0.005
ions of about 10-7 molar, in contrast to an external con-
0
0.5
1.0
1.5
centration of about 10-3 molar.
Milliseconds
In addition, there are voltage-gated calcium channels.
These channels are slightly permeable to sodium ions as
well as to calcium ions; when they open, both calcium
Figure 5-10
and sodium ions flow to the interior of the fiber. There-
fore, these channels are also called Ca++-Na+ channels.
Changes in sodium
and potassium conductance during
the
The calcium channels are slow to become activated,
course of the action potential. Sodium conductance increases
requiring 10 to 20 times as long for activation as the
several thousand-fold during the early stages of the action poten-
sodium channels. Therefore, they are called slow chan-
tial, whereas potassium conductance increases only about 30-fold
nels, in contrast to the sodium channels, which are called
during the latter stages of the action potential and for a short
fast channels.
period thereafter. (These curves were constructed from theory pre-
Calcium channels are numerous in both cardiac
sented in papers by Hodgkin and Huxley but transposed from
squid axon to apply to the membrane potentials of large mam-
muscle and smooth muscle. In fact, in some types of
malian nerve fibers.)
smooth muscle, the fast sodium channels are hardly
Chapter 5
Membrane Potentials and Action Potentials
65
present, so that the action potentials are caused almost
fiber from -90 millivolts up to about -65 millivolts
entirely by activation of slow calcium channels.
usually causes the explosive development of an action
potential. This level of -65 millivolts is said to be the
Increased Permeability of the Sodium Channels When
threshold for stimulation.
There Is a Deficit of Calcium Ions. The concentration of
calcium ions in the extracellular fluid also has a pro-
found effect on the voltage level at which the sodium
Propagation of the
channels become activated. When there is a deficit of
Action Potential
calcium ions, the sodium channels become activated
(opened) by very little increase of the membrane poten-
tial from its normal, very negative level. Therefore, the
In the preceding paragraphs, we discussed the action
nerve fiber becomes highly excitable, sometimes dis-
potential as it occurs at one spot on the membrane.
charging repetitively without provocation rather than
However, an action potential elicited at any one point
remaining in the resting state. In fact, the calcium ion
on an excitable membrane usually excites adjacent
concentration needs to fall only 50 per cent below
portions of the membrane, resulting in propagation of
normal before spontaneous discharge occurs in some
the action potential along the membrane. This mecha-
peripheral nerves, often causing muscle “tetany.” This is
nism is demonstrated in Figure 5-11. Figure 5-11A
sometimes lethal because of tetanic contraction of the
shows a normal resting nerve fiber, and Figure 5-11B
respiratory muscles.
shows a nerve fiber that has been excited in its mid-
The probable way in which calcium ions affect the
portion—that is, the midportion suddenly develops
sodium channels is as follows: These ions appear to bind
to the exterior surfaces of the sodium channel protein
increased permeability to sodium. The arrows show a
molecule. The positive charges of these calcium ions
“local circuit” of current flow from the depolarized
in turn alter the electrical state of the channel protein
areas of the membrane to the adjacent resting mem-
itself, in this way altering the voltage level required to
brane areas. That is, positive electrical charges are
open the sodium gate.
carried by the inward-diffusing sodium ions through
the depolarized membrane and then for several mil-
limeters in both directions along the core of the axon.
Initiation of the Action Potential
These positive charges increase the voltage for a dis-
tance of 1 to 3 millimeters inside the large myelinated
Up to this point, we have explained the changing
sodium and potassium permeability of the membrane,
as well as the development of the action potential
itself, but we have not explained what initiates the
action potential. The answer is quite simple.
+ + + + + + + + + + + + + + + + + + + + + + +
- - - - - - - - - - - - - - - - - - - - - - -
A Positive-Feedback Vicious Cycle Opens the Sodium Channels.
First, as long as the membrane of the nerve fiber
- - - - - - - - - - - - - - - - - - - - - - -
remains undisturbed, no action potential occurs in the
+ + + + + + + + + + + + + + + + + + + + + + +
normal nerve. However, if any event causes enough
A
initial rise in the membrane potential from -90 milli-
+ + + + + + + + + + + + - - + + + + + + + + +
volts toward the zero level, the rising voltage itself
- - - - - - - - - - - - + + - - - - - - - - -
causes many voltage-gated sodium channels to begin
opening. This allows rapid inflow of sodium ions, which
- - - - - - - - - - - - + + - - - - - - - - -
causes a further rise in the membrane potential, thus
+ + + + + + + + + + + + - - + + + + + + + + +
opening still more voltage-gated sodium channels and
B
allowing more streaming of sodium ions to the interior
of the fiber. This process is a positive-feedback vicious
+ + + + + + + + + + - - - - + + + + + + + +
cycle that, once the feedback is strong enough, contin-
- - - - - - - - - - + + + + - - - - - - - -
ues until all the voltage-gated sodium channels have
- - - - - - - - - - + + + + - - - - - - - -
become activated (opened). Then, within another frac-
+ + + + + + + + + + - - - - + + + + + + + +
tion of a millisecond, the rising membrane potential
C
causes closure of the sodium channels as well as
opening of potassium channels, and the action poten-
+ + - - - - - - - - - - - - - - - - - - + +
tial soon terminates.
- - + + + + + + + + + + + + + + + + + + - -
Threshold for Initiation of the Action Potential. An action
- - + + + + + + + + + + + + + + + + + + - -
potential will not occur until the initial rise in mem-
+ + - - - - - - - - - - - - - - - - - - + +
brane potential is great enough to create the vicious
D
cycle described in the preceding paragraph. This
occurs when the number of Na+ ions entering the fiber
becomes greater than the number of K+ ions leaving
Figure 5-11
the fiber. A sudden rise in membrane potential of 15
to 30 millivolts usually is required. Therefore, a sudden
Propagation of action potentials in both directions along a con-
increase in the membrane potential in a large nerve
ductive fiber.
66
Unit II
Membrane Physiology, Nerve, and Muscle
fiber to above the threshold voltage value for initiat-
ing an action potential. Therefore, the sodium channels
in these new areas immediately open, as shown in
Figure 5-11C and D, and the explosive action poten-
tial spreads. These newly depolarized areas produce
still more local circuits of current flow farther along
the membrane, causing progressively more and more
depolarization. Thus, the depolarization process
travels along the entire length of the fiber. This trans-
mission of the depolarization process along a nerve or
muscle fiber is called a nerve or muscle impulse.
At rest
Direction of Propagation. As demonstrated in Figure
5-11, an excitable membrane has no single direction
0
100
200
300
of propagation, but the action potential travels in all
Impulses per second
directions away from the stimulus—even along all
branches of a nerve fiber—until the entire membrane
has become depolarized.
Figure 5-12
All-or-Nothing Principle. Once an action potential has
Heat production in a nerve fiber at rest and at progressively
been elicited at any point on the membrane of a
increasing rates of stimulation.
normal fiber, the depolarization process travels over
the entire membrane if conditions are right, or it does
not travel at all if conditions are not right. This is called
their original state by the Na+-K+ pump. Because this
the all-or-nothing principle, and it applies to all normal
pump requires energy for operation, this “recharging”
excitable tissues. Occasionally, the action potential
of the nerve fiber is an active metabolic process, using
reaches a point on the membrane at which it does not
energy derived from the adenosine triphosphate
generate sufficient voltage to stimulate the next area
(ATP) energy system of the cell. Figure 5-12 shows
of the membrane. When this occurs, the spread of
that the nerve fiber produces excess heat during
depolarization stops. Therefore, for continued propa-
recharging, which is a measure of energy expenditure
gation of an impulse to occur, the ratio of action poten-
when the nerve impulse frequency increases.
tial to threshold for excitation must at all times be
A special feature of the Na+-K+ ATPase pump is that
greater than 1. This “greater than 1” requirement is
its degree of activity is strongly stimulated when excess
called the safety factor for propagation.
sodium ions accumulate inside the cell membrane. In
fact, the pumping activity increases approximately in
proportion to the third power of this intracellular
Re-establishing Sodium and
sodium concentration. That is, as the internal sodium
concentration rises from 10 to 20 mEq/L, the activity
Potassium Ionic Gradients
of the pump does not merely double but increases
After Action Potentials Are
about eightfold. Therefore, it is easy to understand
Completed—Importance
how the “recharging” process of the nerve fiber can be
of Energy Metabolism
set rapidly into motion whenever the concentration
differences of sodium and potassium ions across the
The transmission of each action potential along a
membrane begin to “run down.”
nerve fiber reduces very slightly the concentration
differences of sodium and potassium inside and
Plateau in Some Action
outside the membrane, because sodium ions diffuse to
the inside during depolarization and potassium ions
Potentials
diffuse to the outside during repolarization. For a
single action potential, this effect is so minute that
In some instances, the excited membrane does not
it cannot be measured. Indeed, 100,000 to 50 million
repolarize immediately after depolarization; instead,
impulses can be transmitted by large nerve fibers
the potential remains on a plateau near the peak of the
before the concentration differences reach the point
spike potential for many milliseconds, and only then
that action potential conduction ceases. Even so, with
does repolarization begin. Such a plateau is shown
time, it becomes necessary to re-establish the sodium
in Figure 5-13; one can readily see that the plateau
and potassium membrane concentration differences.
greatly prolongs the period of depolarization. This
This is achieved by action of the Na+-K+ pump in the
type of action potential occurs in heart muscle fibers,
same way as described previously in the chapter for
where the plateau lasts for as long as 0.2 to 0.3 second
the original establishment of the resting potential. That
and causes contraction of heart muscle to last for this
is, sodium ions that have diffused to the interior of the
same long period.
cell during the action potentials and potassium ions
The cause of the plateau is a combination of several
that have diffused to the exterior must be returned to
factors. First, in heart muscle, two types of channels
Chapter 5
Membrane Potentials and Action Potentials
67
+60
Rhythmical
Plateau
Potassium
action
+40
+60
conductance
potentials Threshold
+20
+40
0
+20
-20
0
-40
- 20
-60
- 40
- 60
-80
-100
0
1
2
3
Seconds
0
0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9
Hyperpolarization
Seconds
Figure 5-14
Figure 5-13
Rhythmical action potentials (in millivolts) similar to those recorded
Action potential (in millivolts) from a Purkinje fiber of the heart,
in the rhythmical control center of the heart. Note their relationship
showing a “plateau.”
to potassium conductance and to the state of hyperpolarization.
below a critical value, both of which increase sodium
enter into the depolarization process: (1) the usual
permeability of the membrane.
voltage-activated sodium channels, called fast chan-
nels, and (2) voltage-activated calcium-sodium chan-
Re-excitation Process Necessary for Spontaneous Rhythmicity.
nels, which are slow to open and therefore are called
For spontaneous rhythmicity to occur, the membrane
slow channels. Opening of fast channels causes the
even in its natural state must be permeable enough to
spike portion of the action potential, whereas the slow,
sodium ions (or to calcium and sodium ions through
prolonged opening of the slow calcium-sodium chan-
the slow calcium-sodium channels) to allow automatic
nels mainly allows calcium ions to enter the fiber,
membrane depolarization. Thus, Figure 5-14 shows
which is largely responsible for the plateau portion of
that the “resting” membrane potential in the rhythmi-
the action potential as well.
cal control center of the heart is only -60 to -70 milli-
A second factor that may be partly responsible for
volts. This is not enough negative voltage to keep the
the plateau is that the voltage-gated potassium chan-
sodium and calcium channels totally closed. Therefore,
nels are slower than usual to open, often not opening
the following sequence occurs: (1) some sodium and
very much until the end of the plateau. This delays the
calcium ions flow inward; (2) this increases the mem-
return of the membrane potential toward its normal
brane voltage in the positive direction, which further
negative value of -80 to -90 millivolts.
increases membrane permeability; (3) still more ions
flow inward; and (4) the permeability increases more,
and so on, until an action potential is generated. Then,
RHYTHMICITY OF SOME
at the end of the action potential, the membrane re-
EXCITABLE TISSUES—
polarizes. After another delay of milliseconds or
REPETITIVE DISCHARGE
seconds, spontaneous excitability causes depolariza-
tion again, and a new action potential occurs sponta-
Repetitive self-induced discharges occur normally in
neously. This cycle continues over and over and causes
the heart, in most smooth muscle, and in many of the
self-induced rhythmical excitation of the excitable
neurons of the central nervous system. These rhyth-
tissue.
mical discharges cause (1) the rhythmical beat of the
Why does the membrane of the heart control center
heart, (2) rhythmical peristalsis of the intestines, and
not depolarize immediately after it has become re-
(3) such neuronal events as the rhythmical control of
polarized, rather than delaying for nearly a second
breathing.
before the onset of the next action potential? The
Also, almost all other excitable tissues can discharge
answer can be found by observing the curve labeled
repetitively if the threshold for stimulation of the
“potassium conductance” in Figure 5-14. This shows
tissue cells is reduced low enough. For instance, even
that toward the end of each action potential, and con-
large nerve fibers and skeletal muscle fibers, which
tinuing for a short period thereafter, the membrane
normally are highly stable, discharge repetitively when
becomes excessively permeable to potassium ions. The
they are placed in a solution that contains the drug
excessive outflow of potassium ions carries tremen-
veratrine or when the calcium ion concentration falls
dous numbers of positive charges to the outside of the
68
Unit II
Membrane Physiology, Nerve, and Muscle
Axon
Myelin
sheath
Schwann cell
cytoplasm
Schwann cell
nucleus
Node of Ranvier
A
Figure 5-15
Unmyelinated axons
Cross section of a small nerve trunk containing both myelinated
and unmyelinated fibers.
Schwann cell nucleus
Schwann cell cytoplasm
membrane, leaving inside the fiber considerably more
negativity than would otherwise occur. This continues
B
for nearly a second after the preceding action poten-
Figure 5-16
tial is over, thus drawing the membrane potential
nearer to the potassium Nernst potential. This is a state
Function of the Schwann cell to insulate nerve fibers. A, Wrapping
called hyperpolarization, also shown in Figure 5-14. As
of a Schwann cell membrane around a large axon to form the
long as this state exists, self-re-excitation will not
myelin sheath of the myelinated nerve fiber. B, Partial wrapping of
the membrane and cytoplasm of a Schwann cell around multiple
occur. But the excess potassium conductance (and the
unmyelinated nerve fibers (shown in cross section). (A, Modified
state of hyperpolarization) gradually disappears, as
from Leeson TS, Leeson R: Histology. Philadelphia: WB Saunders,
shown after each action potential is completed in the
1979.)
figure, thereby allowing the membrane potential again
to increase up to the threshold for excitation. Then,
suddenly, a new action potential results, and the
The myelin sheath is deposited around the axon by
process occurs again and again.
Schwann cells in the following manner: The membrane
of a Schwann cell first envelops the axon. Then the
Schwann cell rotates around the axon many times,
Special Characteristics
laying down multiple layers of Schwann cell membrane
of Signal Transmission
containing the lipid substance sphingomyelin. This sub-
stance is an excellent electrical insulator that decreases
in Nerve Trunks
ion flow through the membrane about 5000-fold. At the
Myelinated and Unmyelinated Nerve Fibers. Figure
5-15
juncture between each two successive Schwann cells
shows a cross section of a typical small nerve, revealing
along the axon, a small uninsulated area only 2 to 3
many large nerve fibers that constitute most of the
micrometers in length remains where ions still can flow
cross-sectional area. However, a more careful look
with ease through the axon membrane between the
reveals many more very small fibers lying between
extracellular fluid and the intracellular fluid inside the
the large ones. The large fibers are myelinated, and the
axon. This area is called the node of Ranvier.
small ones are unmyelinated. The average nerve trunk
contains about twice as many unmyelinated fibers as
“Saltatory” Conduction in Myelinated Fibers from Node to Node.
myelinated fibers.
Even though almost no ions can flow through the thick
Figure 5-16 shows a typical myelinated fiber. The
myelin sheaths of myelinated nerves, they can flow with
central core of the fiber is the axon, and the membrane
ease through the nodes of Ranvier. Therefore, action
of the axon is the membrane that actually conducts the
potentials occur only at the nodes. Yet the action poten-
action potential. The axon is filled in its center with axo-
tials are conducted from node to node, as shown in
plasm, which is a viscid intracellular fluid. Surrounding
Figure 5-17; this is called saltatory conduction. That is,
the axon is a myelin sheath that is often much thicker
electrical current flows through the surrounding extra-
than the axon itself. About once every 1 to 3 milli-
cellular fluid outside the myelin sheath as well as
meters along the length of the myelin sheath is a node
through the axoplasm inside the axon from node to
of Ranvier.
node, exciting successive nodes one after another. Thus,
Chapter 5
Membrane Potentials and Action Potentials
69
Myelin sheath
Axoplasm
Node of Ranvier
+60
Action potentials
+40
+20
0
Acute
-20
subthreshold
potentials
-40
-60
Threshold
A
B
C
D
1
2
3
0
1
2
3
4
Milliseconds
Figure 5-17
Saltatory conduction along a myelinated axon. Flow of electrical
current from node to node is illustrated by the arrows.
Figure 5-18
Effect of stimuli of increasing voltages to elicit an action potential.
Note development of “acute subthreshold potentials” when the
stimuli are below the threshold value required for eliciting an
the nerve impulse jumps down the fiber, which is the
action potential.
origin of the term “saltatory.”
Saltatory conduction is of value for two reasons. First,
by causing the depolarization process to jump long
Excitation of a Nerve Fiber by a Negatively Charged Metal
intervals along the axis of the nerve fiber, this mecha-
Electrode. The usual means for exciting a nerve or
nism increases the velocity of nerve transmission in
muscle in the experimental laboratory is to apply elec-
myelinated fibers as much as 5- to 50-fold. Second,
tricity to the nerve or muscle surface through two small
saltatory conduction conserves energy for the axon
electrodes, one of which is negatively charged and
because only the nodes depolarize, allowing perhaps
the other positively charged. When this is done, the
100 times less loss of ions than would otherwise be nec-
excitable membrane becomes stimulated at the negative
essary, and therefore requiring little metabolism for re-
electrode.
establishing the sodium and potassium concentration
The cause of this effect is the following: Remember
differences across the membrane after a series of nerve
that the action potential is initiated by the opening of
impulses.
voltage-gated sodium channels. Further, these channels
Still another feature of saltatory conduction in
are opened by a decrease in the normal resting electri-
large myelinated fibers is the following: The excellent
cal voltage across the membrane. That is, negative
insulation afforded by the myelin membrane and the 50-
current from the electrode decreases the voltage on the
fold decrease in membrane capacitance allow repolar-
outside of the membrane to a negative value nearer to
ization to occur with very little transfer of ions.
the voltage of the negative potential inside the fiber.
This decreases the electrical voltage across the mem-
Velocity of Conduction in Nerve Fibers. The velocity of con-
brane and allows the sodium channels to open, result-
duction in nerve fibers varies from as little as 0.25 m/sec
ing in an action potential. Conversely, at the positive
in very small unmyelinated fibers to as great as 100 m/
electrode, the injection of positive charges on the
sec (the length of a football field in 1 second) in very
outside of the nerve membrane heightens the voltage
large myelinated fibers.
difference across the membrane rather than lessening
it. This causes a state of hyperpolarization, which actu-
ally decreases the excitability of the fiber rather than
Excitation—The Process
causing an action potential.
of Eliciting the Action
Threshold for Excitation, and “Acute Local Potentials.” A weak
Potential
negative electrical stimulus may not be able to excite a
Basically, any factor that causes sodium ions to begin to
fiber. However, when the voltage of the stimulus is
diffuse inward through the membrane in sufficient
increased, there comes a point at which excitation does
numbers can set off automatic regenerative opening of
take place. Figure 5-18 shows the effects of successively
the sodium channels. This can result from mechanical
applied stimuli of progressing strength. A very weak
disturbance of the membrane, chemical effects on the
stimulus at point A causes the membrane potential to
membrane, or passage of electricity through the mem-
change from -90 to -85 millivolts, but this is not a suffi-
brane. All these are used at different points in the body
cient change for the automatic regenerative processes
to elicit nerve or muscle action potentials: mechanical
of the action potential to develop. At point B, the stim-
pressure to excite sensory nerve endings in the skin,
ulus is greater, but again, the intensity is still not enough.
chemical neurotransmitters to transmit signals from one
The stimulus does, however, disturb the membrane
neuron to the next in the brain, and electrical current to
potential locally for as long as 1 millisecond or more
transmit signals between successive muscle cells in the
after both of these weak stimuli. These local potential
heart and intestine. For the purpose of understanding
changes are called acute local potentials, and when they
the excitation process, let us begin by discussing the
fail to elicit an action potential, they are called acute
principles of electrical stimulation.
subthreshold potentials.
70
Unit II
Membrane Physiology, Nerve, and Muscle
At point C in Figure 5-18, the stimulus is even
decrease excitability. For instance, a high extracellular
stronger. Now the local potential has barely reached the
fluid calcium ion concentration decreases membrane
level required to elicit an action potential, called the
permeability to sodium ions and simultaneously reduces
threshold level, but this occurs only after a short “latent
excitability. Therefore, calcium ions are said to be a
period.” At point D, the stimulus is still stronger, the
“stabilizer.”
acute local potential is also stronger, and the action
potential occurs after less of a latent period.
Local Anesthetics. Among the most important stabilizers
Thus, this figure shows that even a very weak stimu-
are the many substances used clinically as local anes-
lus causes a local potential change at the membrane, but
thetics, including procaine and tetracaine. Most of these
the intensity of the local potential must rise to a thresh-
act directly on the activation gates of the sodium chan-
old level before the action potential is set off.
nels, making it much more difficult for these gates to
open, thereby reducing membrane excitability. When
“Refractory Period” After an Action
excitability has been reduced so low that the ratio of
Potential, During Which a New
action potential strength to excitability threshold (called
the “safety factor”) is reduced below 1.0, nerve impulses
Stimulus Cannot Be Elicited
fail to pass along the anesthetized nerves.
A new action potential cannot occur in an excitable fiber
as long as the membrane is still depolarized from the pre-
ceding action potential. The reason for this is that shortly
Recording Membrane
after the action potential is initiated, the sodium channels
Potentials and Action
(or calcium channels, or both) become inactivated, and
Potentials
no amount of excitatory signal applied to these channels
at this point will open the inactivation gates. The only
Cathode Ray Oscilloscope. Earlier in this chapter, we noted
condition that will allow them to reopen is for the mem-
that the membrane potential changes extremely rapidly
brane potential to return to or near the original resting
during the course of an action potential. Indeed, most
membrane potential level. Then, within another small
of the action potential complex of large nerve fibers
fraction of a second, the inactivation gates of the chan-
takes place in less than 1/1000 second. In some figures
nels open, and a new action potential can be initiated.
of this chapter, an electrical meter has been shown
The period during which a second action potential
recording these potential changes. However, it must be
cannot be elicited, even with a strong stimulus, is
understood that any meter capable of recording most
called the absolute refractory period. This period for
action potentials must be capable of responding
large myelinated nerve fibers is about 1/2500 second.
extremely rapidly. For practical purposes, the only
Therefore, one can readily calculate that such a fiber
common type of meter that is capable of responding
can transmit a maximum of about 2500 impulses per
accurately to the rapid membrane potential changes is
second.
the cathode ray oscilloscope.
Figure 5-19 shows the basic components of a cathode
Inhibition of Excitability—
ray oscilloscope. The cathode ray tube itself is composed
“Stabilizers” and Local Anesthetics
basically of an electron gun and a fluorescent screen
against which electrons are fired. Where the electrons
In contrast to the factors that increase nerve excitabil-
hit the screen surface, the fluorescent material glows. If
ity, still others, called membrane-stabilizing factors, can
the electron beam is moved across the screen, the spot
Recorded
action potential
Horizontal
plates
Electron gun
Electron
beam
Plugs
Vertical
Stimulus
plates
Electronic
artifact
Electronic
amplifier
sweep circuit
Electrical
stimulator
Figure 5-19
Nerve
Cathode ray oscilloscope for recording transient
action potentials.
Chapter 5
Membrane Potentials and Action Potentials
71
of glowing light also moves and draws a fluorescent line
Hodgkin AL: The Conduction of the Nervous Impulse.
on the screen.
Springfield, IL: Charles C Thomas, 1963.
In addition to the electron gun and fluorescent
Hodgkin AL, Huxley AF: Quantitative description of
surface, the cathode ray tube is provided with two sets
membrane current and its application to conduction and
of electrically charged plates—one set positioned on the
excitation in nerve. J Physiol (Lond) 117:500, 1952.
two sides of the electron beam, and the other set posi-
Kleber AG, Rudy Y: Basic mechanisms of cardiac impulse
tioned above and below. Appropriate electronic control
propagation and associated arrhythmias. Physiol Rev
circuits change the voltage on these plates so that the
84:431, 2004.
electron beam can be bent up or down in response to
Lu Z: Mechanism of rectification in inward-rectifier K+
electrical signals coming from recording electrodes on
channels. Annu Rev Physiol 66:103, 2004.
nerves. The beam of electrons also is swept horizontally
Matthews GG: Cellular Physiology of Nerve and Muscle.
across the screen at a constant time rate by an internal
Malden, MA: Blackwell Science, 1998.
electronic circuit of the oscilloscope. This gives the
Perez-Reyes E: Molecular physiology of low-voltage-
record shown on the face of the cathode ray tube in
activated T-type calcium channels. Physiol Rev 83:117,
the figure, giving a time base horizontally and voltage
2003.
changes from the nerve electrodes shown vertically.
Poliak S, Peles E: The local differentiation of myelinated
Note at the left end of the record a small stimulus arti-
axons at nodes of Ranvier. Nat Rev Neurosci 12:968, 2003.
fact caused by the electrical stimulus used to elicit the
Pollard TD, Earnshaw WC: Cell Biology. Philadelphia:
nerve action potential. Then further to the right is
Elsevier Science, 2002.
the recorded action potential itself.
Ruff RL: Neurophysiology of the neuromuscular junction:
overview. Ann N Y Acad Sci 998:1, 2003.
Xu-Friedman MA, Regehr WG: Structural contributions to
References
short-term synaptic plasticity. Physiol Rev 84:69, 2004.
Alberts B, Johnson A, Lewis J, et al: Molecular Biology of
the Cell. New York: Garland Science, 2002.
Grillner S: The motor infrastructure: from ion channels to
neuronal networks. Nat Rev Neurosci 4:573, 2003.
C H A P T E R
6
Contraction of
Skeletal Muscle
About 40 per cent of the body is skeletal muscle,
and perhaps another 10 per cent is smooth and
cardiac muscle. Some of the same basic principles of
contraction apply to all these different types of
muscle. In this chapter, function of skeletal muscle
is considered mainly; the specialized functions of
smooth muscle are discussed in Chapter 8, and
cardiac muscle is discussed in Chapter 9.
Physiologic Anatomy of Skeletal Muscle
Skeletal Muscle Fiber
Figure 6-1 shows the organization of skeletal muscle, demonstrating that all
skeletal muscles are composed of numerous fibers ranging from 10 to 80
micrometers in diameter. Each of these fibers is made up of successively smaller
subunits, also shown in Figure 6-1 and described in subsequent paragraphs.
In most skeletal muscles, each fiber extends the entire length of the muscle.
Except for about 2 per cent of the fibers, each fiber is usually innervated by only
one nerve ending, located near the middle of the fiber.
Sarcolemma. The sarcolemma is the cell membrane of the muscle fiber. The sar-
colemma consists of a true cell membrane, called the plasma membrane, and an
outer coat made up of a thin layer of polysaccharide material that contains
numerous thin collagen fibrils. At each end of the muscle fiber, this surface layer
of the sarcolemma fuses with a tendon fiber, and the tendon fibers in turn collect
into bundles to form the muscle tendons that then insert into the bones.
Myofibrils; Actin and Myosin Filaments. Each muscle fiber contains several hundred
to several thousand myofibrils, which are demonstrated by the many small open
dots in the cross-sectional view of Figure 6-1C. Each myofibril (Figure 6-1D
and E) is composed of about 1500 adjacent myosin filaments and 3000 actin fil-
aments, which are large polymerized protein molecules that are responsible for
the actual muscle contraction. These can be seen in longitudinal view in the elec-
tron micrograph of Figure 6-2 and are represented diagrammatically in Figure
6-1, parts E through L. The thick filaments in the diagrams are myosin, and the
thin filaments are actin.
Note in Figure 6-1E that the myosin and actin filaments partially interdigi-
tate and thus cause the myofibrils to have alternate light and dark bands, as
illustrated in Figure 6-2. The light bands contain only actin filaments and are
called I bands because they are isotropic to polarized light. The dark bands
contain myosin filaments, as well as the ends of the actin filaments where they
overlap the myosin, and are called A bands because they are anisotropic to
polarized light. Note also the small projections from the sides of the myosin
filaments in Figure 6-1E and L. These are cross-bridges. It is the interaction
between these cross-bridges and the actin filaments that causes contraction.
Figure 6-1E also shows that the ends of the actin filaments are attached to a
so-called Z disc. From this disc, these filaments extend in both directions to
72
Chapter 6
Contraction of Skeletal Muscle
73
SKELETAL MUSCLE
Muscle
A
Muscle fasciculus
B
C
Muscle fiber
H
Z
A
I
band disc band
band
Myofibril
Z
Sarcomere Z
D
G-Actin molecules
H
J
Myofilaments
F-Actin filament
K
L
E
Figure 6-1
Myosin filament
Organization of skeletal muscle,
from the gross to the molecular
Myosin molecule
level. F, G, H, and I are cross
M
sections at the levels indicated.
(Drawing by Sylvia Colard
Keene. Modified from Fawcett
N
DW: Bloom and Fawcett: A Text-
Light
Heavy
book of Histology. Philadelphia:
meromyosin
meromyosin
WB Saunders, 1986.)
F
G
H
I
interdigitate with the myosin filaments. The Z disc,
beginning to overlap one another. We will see later
which itself is composed of filamentous proteins dif-
that, at this length, the muscle is capable of generating
ferent from the actin and myosin filaments, passes
its greatest force of contraction.
crosswise across the myofibril and also crosswise from
myofibril to myofibril, attaching the myofibrils to one
What Keeps the Myosin and Actin Filaments in Place?
another all the way across the muscle fiber. Therefore,
Titin Filamentous Molecules. The side-by-side rela-
the entire muscle fiber has light and dark bands, as do
tionship between the myosin and actin filaments is dif-
the individual myofibrils. These bands give skeletal and
ficult to maintain. This is achieved by a large number
cardiac muscle their striated appearance.
of filamentous molecules of a protein called titin. Each
The portion of the myofibril (or of the whole muscle
titin molecule has a molecular weight of about 3
fiber) that lies between two successive Z discs is called
million, which makes it one of the largest protein mol-
a sarcomere. When the muscle fiber is contracted, as
ecules in the body. Also, because it is filamentous, it is
shown at the bottom of Figure 6-4, the length of
very springy. These springy titin molecules act as a
the sarcomere is about 2 micrometers. At this length,
framework that holds the myosin and actin filaments
the actin filaments completely overlap the myosin
in place so that the contractile machinery of the sar-
filaments, and the tips of the actin filaments are just
comere will work. There is reason to believe that the
74
Unit II
Membrane Physiology, Nerve, and Muscle
Figure 6-2
Electron micrograph of muscle myofibrils showing the detailed
Figure 6-3
organization of actin and myosin filaments. Note the mitochondria
lying between the myofibrils.
(From Fawcett DW: The Cell.
Sarcoplasmic reticulum in the extracellular spaces between the
Philadelphia: WB Saunders, 1981.)
myofibrils, showing a longitudinal system paralleling the myofi-
brils. Also shown in cross section are T tubules (arrows) that lead
to the exterior of the fiber membrane and are important for con-
ducting the electrical signal into the center of the muscle fiber.
(From Fawcett DW: The Cell. Philadelphia: WB Saunders, 1981.)
titin molecule itself acts as template for initial forma-
4. Opening of the acetylcholine-gated channels allows
tion of portions of the contractile filaments of the
large quantities of sodium ions to diffuse to the
sarcomere, especially the myosin filaments.
interior of the muscle fiber membrane. This initiates
an action potential at the membrane.
Sarcoplasm. The many myofibrils of each muscle fiber
5. The action potential travels along the muscle fiber
are suspended side by side in the muscle fiber. The
membrane in the same way that action potentials
spaces between the myofibrils are filled with intracel-
travel along nerve fiber membranes.
lular fluid called sarcoplasm, containing large quanti-
6. The action potential depolarizes the muscle
ties of potassium, magnesium, and phosphate, plus
membrane, and much of the action potential
multiple protein enzymes. Also present are tremen-
electricity flows through the center of the muscle
dous numbers of mitochondria that lie parallel to the
fiber. Here it causes the sarcoplasmic reticulum to
release large quantities of calcium ions that have
myofibrils. These supply the contracting myofibrils
been stored within this reticulum.
with large amounts of energy in the form of adenosine
7. The calcium ions initiate attractive forces between
triphosphate (ATP) formed by the mitochondria.
the actin and myosin filaments, causing them to
slide alongside each other, which is the contractile
Sarcoplasmic Reticulum. Also in the sarcoplasm sur-
process.
rounding the myofibrils of each muscle fiber is an
8. After a fraction of a second, the calcium ions are
extensive reticulum (Figure 6-3), called the sarcoplas-
pumped back into the sarcoplasmic reticulum by a
mic reticulum. This reticulum has a special organiza-
Ca++ membrane pump, and they remain stored in
tion that is extremely important in controlling muscle
the reticulum until a new muscle action potential
contraction, as discussed in Chapter 7. The very rapidly
comes along; this removal of calcium ions from the
myofibrils causes the muscle contraction to cease.
contracting types of muscle fibers have especially
We now describe the molecular machinery of the
extensive sarcoplasmic reticula.
muscle contractile process.
General Mechanism of Muscle
Contraction
Molecular Mechanism
of Muscle Contraction
The initiation and execution of muscle contraction
occur in the following sequential steps.
Sliding Filament Mechanism of Muscle Contraction. Figure
1. An action potential travels along a motor nerve to
6-4 demonstrates the basic mechanism of muscle con-
its endings on muscle fibers.
traction. It shows the relaxed state of a sarcomere
2. At each ending, the nerve secretes a small amount
(top) and the contracted state (bottom). In the relaxed
of the neurotransmitter substance acetylcholine.
state, the ends of the actin filaments extending from
3. The acetylcholine acts on a local area of the muscle
two successive Z discs barely begin to overlap one
fiber membrane to open multiple “acetylcholine-
gated” channels through protein molecules floating
another. Conversely, in the contracted state, these actin
in the membrane.
filaments have been pulled inward among the myosin
Chapter 6
Contraction of Skeletal Muscle
75
I
A
I
Head
A
Z
Z
Tail
Relaxed
Two heavy chains
I
A
I
Light chains
Z
Z
B
Actin filaments
Contracted
Figure 6-4
Cross-bridges
Hinges
Body
Relaxed and contracted states of a myofibril showing (top) sliding
Myosin filament
of the actin filaments (pink) into the spaces between the myosin
filaments (red), and (bottom) pulling of the Z membranes toward
each other.
Figure 6-5
A, Myosin molecule. B, Combination of many myosin molecules
to form a myosin filament. Also shown are thousands of myosin
filaments, so that their ends overlap one another to
cross-bridges and interaction between the heads of the cross-
their maximum extent. Also, the Z discs have been
bridges with adjacent actin filaments.
pulled by the actin filaments up to the ends of the
myosin filaments. Thus, muscle contraction occurs by a
sliding filament mechanism.
The two heavy chains wrap spirally around each other
But what causes the actin filaments to slide inward
to form a double helix, which is called the tail of the
among the myosin filaments? This is caused by forces
myosin molecule. One end of each of these chains is
generated by interaction of the cross-bridges from
folded bilaterally into a globular polypeptide structure
the myosin filaments with the actin filaments. Under
called a myosin head. Thus, there are two free heads
resting conditions, these forces are inactive, but when
at one end of the double-helix myosin molecule. The
an action potential travels along the muscle fiber,
four light chains are also part of the myosin head, two
this causes the sarcoplasmic reticulum to release large
to each head. These light chains help control the func-
quantities of calcium ions that rapidly surround the
tion of the head during muscle contraction.
myofibrils. The calcium ions in turn activate the forces
The myosin filament is made up of 200 or more indi-
between the myosin and actin filaments, and contrac-
vidual myosin molecules. The central portion of one of
tion begins. But energy is needed for the contractile
these filaments is shown in Figure 6-5B, displaying the
process to proceed. This energy comes from high-
tails of the myosin molecules bundled together to form
energy bonds in the ATP molecule, which is degraded
the body of the filament, while many heads of the
to adenosine diphosphate
(ADP) to liberate the
molecules hang outward to the sides of the body. Also,
energy. In the next few sections, we describe what is
part of the body of each myosin molecule hangs to the
known about the details of these molecular processes
side along with the head, thus providing an arm that
of contraction.
extends the head outward from the body, as shown in
the figure. The protruding arms and heads together are
called cross-bridges. Each cross-bridge is flexible at
Molecular Characteristics of the
two points called hinges—one where the arm leaves
Contractile Filaments
the body of the myosin filament, and the other where
the head attaches to the arm. The hinged arms allow
Myosin Filament. The myosin filament is composed of
the heads either to be extended far outward from the
multiple myosin molecules, each having a molecular
body of the myosin filament or to be brought close to
weight of about 480,000. Figure 6-5A shows an indi-
the body. The hinged heads in turn participate in the
vidual molecule; Figure 6-5B shows the organization
actual contraction process, as discussed in the follow-
of many molecules to form a myosin filament, as well
ing sections.
as interaction of this filament on one side with the ends
The total length of each myosin filament is uniform,
of two actin filaments.
almost exactly 1.6 micrometers. Note, however, that
The myosin molecule (see Figure 6-5A) is composed
there are no cross-bridge heads in the very center of
of six polypeptide chains—two heavy chains, each with
the myosin filament for a distance of about
0.2
a molecular weight of about 200,000, and four light
micrometer because the hinged arms extend away
chains with molecular weights of about 20,000 each.
from the center.
76
Unit II
Membrane Physiology, Nerve, and Muscle
Now, to complete the picture, the myosin filament
cannot occur between the actin and myosin filaments
itself is twisted so that each successive pair of cross-
to cause contraction.
bridges is axially displaced from the previous pair by
120 degrees. This ensures that the cross-bridges extend
Troponin and Its Role in Muscle Contraction. Attached
in all directions around the filament.
intermittently along the sides of the tropomyosin mol-
ecules are still other protein molecules called troponin.
ATPase Activity of the Myosin Head. Another feature
These are actually complexes of three loosely bound
of the myosin head that is essential for muscle con-
protein subunits, each of which plays a specific role in
traction is that it functions as an ATPase enzyme. As
controlling muscle contraction. One of the subunits
explained later, this property allows the head to cleave
(troponin I) has a strong affinity for actin, another
ATP and to use the energy derived from the ATP’s
(troponin T) for tropomyosin, and a third (troponin C)
high-energy phosphate bond to energize the contrac-
for calcium ions. This complex is believed to attach
tion process.
the tropomyosin to the actin. The strong affinity of the
troponin for calcium ions is believed to initiate the
Actin Filament. The actin filament is also complex.
contraction process, as explained in the next section.
It is composed of three protein components: actin,
tropomyosin, and troponin.
Interaction of One Myosin Filament,
The backbone of the actin filament is a double-
Two Actin Filaments, and Calcium Ions
stranded F-actin protein molecule, represented by the
to Cause Contraction
two lighter-colored strands in Figure 6-6. The two
strands are wound in a helix in the same manner as the
Inhibition of the Actin Filament by the Troponin-Tropomyosin
myosin molecule.
Complex; Activation by Calcium Ions. A pure actin filament
Each strand of the double F-actin helix is composed
without the presence of the troponin-tropomyosin
of polymerized G-actin molecules, each having a
complex (but in the presence of magnesium ions and
molecular weight of about 42,000. Attached to each
ATP) binds instantly and strongly with the heads
one of the G-actin molecules is one molecule of
of the myosin molecules. Then, if the troponin-
ADP. It is believed that these ADP molecules are the
tropomyosin complex is added to the actin filament,
active sites on the actin filaments with which the cross-
the binding between myosin and actin does not take
bridges of the myosin filaments interact to cause
place. Therefore, it is believed that the active sites on
muscle contraction. The active sites on the two F-actin
the normal actin filament of the relaxed muscle are
strands of the double helix are staggered, giving one
inhibited or physically covered by the troponin-
active site on the overall actin filament about every 2.7
tropomyosin complex. Consequently, the sites cannot
nanometers.
attach to the heads of the myosin filaments to cause
Each actin filament is about 1 micrometer long. The
contraction. Before contraction can take place, the
bases of the actin filaments are inserted strongly into
inhibitory effect of the troponin-tropomyosin complex
the Z discs; the ends of the filaments protrude in both
must itself be inhibited.
directions to lie in the spaces between the myosin
This brings us to the role of the calcium ions. In
molecules, as shown in Figure 6-4.
the presence of large amounts of calcium ions, the
inhibitory effect of the troponin-tropomyosin on
Tropomyosin Molecules. The actin filament also con-
the actin filaments is itself inhibited. The mechanism
tains another protein, tropomyosin. Each molecule of
of this is not known, but one suggestion is the follow-
tropomyosin has a molecular weight of 70,000 and a
ing: When calcium ions combine with troponin C, each
length of 40 nanometers. These molecules are wrapped
molecule of which can bind strongly with up to four
spirally around the sides of the F-actin helix. In the
calcium ions, the troponin complex supposedly under-
resting state, the tropomyosin molecules lie on top of
goes a conformational change that in some way tugs
the active sites of the actin strands, so that attraction
on the tropomyosin molecule and moves it deeper into
the groove between the two actin strands. This “uncov-
ers” the active sites of the actin, thus allowing these to
attract the myosin cross-bridge heads and cause con-
Active sites
Troponin complex
traction to proceed. Although this is a hypothetical
mechanism, it does emphasize that the normal relation
between the troponin-tropomyosin complex and actin
is altered by calcium ions, producing a new condition
that leads to contraction.
F-actin
Tropomyosin
Interaction Between the “Activated” Actin Filament and the
Figure 6-6
Myosin Cross-Bridges—The “Walk-Along” Theory of Contrac-
tion. As soon as the actin filament becomes activated
Actin filament, composed of two helical strands of F-actin mole-
by the calcium ions, the heads of the cross-bridges
cules and two strands of tropomyosin molecules that fit in the
from the myosin filaments become attracted to the
grooves between the actin strands. Attached to one end of each
tropomyosin molecule is a troponin complex that initiates
active sites of the actin filament, and this, in some way,
contraction.
causes contraction to occur. Although the precise
Chapter 6
Contraction of Skeletal Muscle
77
Movement
Active sites
Actin filament
ion, bound to the head. In this state, the
conformation of the head is such that it extends
perpendicularly toward the actin filament but is
Power
not yet attached to the actin.
Hinges
stroke
2.
When the troponin-tropomyosin complex binds
with calcium ions, active sites on the actin filament
are uncovered, and the myosin heads then bind
with these, as shown in Figure 6-7.
3.
The bond between the head of the cross-bridge
Myosin filament
and the active site of the actin filament causes a
conformational change in the head, prompting the
Figure 6-7
head to tilt toward the arm of the cross-bridge.
This provides the power stroke for pulling the
“Walk-along” mechanism for contraction of the muscle.
actin filament. The energy that activates the
power stroke is the energy already stored, like a
“cocked” spring, by the conformational change
manner by which this interaction between the cross-
that occurred in the head when the ATP molecule
bridges and the actin causes contraction is still partly
was cleaved earlier.
theoretical, one hypothesis for which considerable evi-
4.
Once the head of the cross-bridge tilts, this allows
dence exists is the “walk-along” theory (or “ratchet”
release of the ADP and phosphate ion that were
theory) of contraction.
previously attached to the head. At the site of
Figure 6-7 demonstrates this postulated walk-along
release of the ADP, a new molecule of ATP binds.
mechanism for contraction. The figure shows the heads
This binding of new ATP causes detachment of
of two cross-bridges attaching to and disengaging from
the head from the actin.
active sites of an actin filament. It is postulated that
5.
After the head has detached from the actin, the
when a head attaches to an active site, this attach-
new molecule of ATP is cleaved to begin the next
ment simultaneously causes profound changes in the
cycle, leading to a new power stroke. That is,
intramolecular forces between the head and arm of its
the energy again “cocks” the head back to its
cross-bridge. The new alignment of forces causes the
perpendicular condition, ready to begin the new
head to tilt toward the arm and to drag the actin fila-
power stroke cycle.
ment along with it. This tilt of the head is called the
6.
When the cocked head (with its stored energy
power stroke. Then, immediately after tilting, the head
derived from the cleaved ATP) binds with a new
automatically breaks away from the active site. Next,
active site on the actin filament, it becomes
the head returns to its extended direction. In this
uncocked and once again provides a new power
position, it combines with a new active site farther
stroke.
down along the actin filament; then the head tilts again
Thus, the process proceeds again and again until the
to cause a new power stroke, and the actin filament
actin filaments pull the Z membrane up against
moves another step. Thus, the heads of the cross-
the ends of the myosin filaments or until the load on
bridges bend back and forth and step by step walk
the muscle becomes too great for further pulling to
along the actin filament, pulling the ends of two suc-
occur.
cessive actin filaments toward the center of the myosin
filament.
Effect of Amount of Actin and Myosin
Each one of the cross-bridges is believed to operate
Filament Overlap on Tension
independently of all others, each attaching and pulling
in a continuous repeated cycle. Therefore, the greater
Developed by the Contracting Muscle
the number of cross-bridges in contact with the actin
filament at any given time, the greater, theoretically,
Figure 6-8 shows the effect of sarcomere length and
the force of contraction.
amount of myosin-actin filament overlap on the active
tension developed by a contracting muscle fiber. To the
ATP as the Source of Energy for Contraction—Chemical Events
right, shown in black, are different degrees of overlap
in the Motion of the Myosin Heads. When a muscle con-
of the myosin and actin filaments at different sarco-
tracts, work is performed and energy is required. Large
mere lengths. At point D on the diagram, the actin
amounts of ATP are cleaved to form ADP during the
filament has pulled all the way out to the end of the
contraction process; the greater the amount of work
myosin filament, with no actin-myosin overlap. At this
performed by the muscle, the greater the amount of
point, the tension developed by the activated muscle
ATP that is cleaved, which is called the Fenn effect. The
is zero. Then, as the sarcomere shortens and the actin
following sequence of events is believed to be the
filament begins to overlap the myosin filament, the
means by which this occurs:
tension increases progressively until the sarcomere
1. Before contraction begins, the heads of the cross-
length decreases to about 2.2 micrometers. At this
bridges bind with ATP. The ATPase activity of the
point, the actin filament has already overlapped all
myosin head immediately cleaves the ATP but
the cross-bridges of the myosin filament but has not
leaves the cleavage products, ADP plus phosphate
yet reached the center of the myosin filament. With
78
Unit II
Membrane Physiology, Nerve, and Muscle
Normal range of contraction
D
B C
C
100
B
Tension during
A
contraction
A
Increase in tension
during contraction
50
Tension
before contraction
D
D
0
0
0
1
2
3
4
1/2
Normal
2 x
Length of sarcomere (micrometers)
normal
normal
Length
Figure 6-8
Figure 6-9
Length-tension diagram for a single fully contracted sarcomere,
Relation of muscle length to tension in the muscle both before and
showing maximum strength of contraction when the sarcomere is
during muscle contraction.
2.0 to 2.2 micrometers in length. At the upper right are the rela-
tive positions of the actin and myosin filaments at different sar-
muscle is stretched beyond its normal length—that is,
comere lengths from point A to point D. (Modified from Gordon
AM, Huxley AF, Julian FJ: The length-tension diagram of single
to a sarcomere length greater than about 2.2 microm-
vertebrate striated muscle fibers. J Physiol 171:28P, 1964.)
eters. This is demonstrated by the decreased length of
the arrow in the figure at greater than normal muscle
length.
further shortening, the sarcomere maintains full
tension until point B is reached, at a sarcomere length
of about 2 micrometers. At this point, the ends of the
Relation of Velocity of Contraction
two actin filaments begin to overlap each other in
to Load
addition to overlapping the myosin filaments. As the
A skeletal muscle contracts extremely rapidly when it
sarcomere length falls from 2 micrometers down to
contracts against no load—to a state of full contraction
about 1.65 micrometers, at point A, the strength of
in about 0.1 second for the average muscle. When loads
contraction decreases rapidly. At this point, the two Z
are applied, the velocity of contraction becomes pro-
discs of the sarcomere abut the ends of the myosin fil-
gressively less as the load increases, as shown in Figure
aments. Then, as contraction proceeds to still shorter
6-10. That is, when the load has been increased to equal
sarcomere lengths, the ends of the myosin filaments
the maximum force that the muscle can exert, the veloc-
are crumpled and, as shown in the figure, the strength
ity of contraction becomes zero and no contraction
of contraction approaches zero, but the entire muscle
results, despite activation of the muscle fiber.
has now contracted to its shortest length.
This decreasing velocity of contraction with load is
caused by the fact that a load on a contracting muscle
is a reverse force that opposes the contractile force
Effect of Muscle Length on Force of Contraction in the Whole
caused by muscle contraction. Therefore, the net force
Intact Muscle. The top curve of Figure 6-9 is similar to
that is available to cause velocity of shortening is
that in Figure 6-8, but the curve in Figure 6-9 depicts
correspondingly reduced.
tension of the intact, whole muscle rather than of a
single muscle fiber. The whole muscle has a large
amount of connective tissue in it; also, the sarcomeres
Energetics of Muscle
in different parts of the muscle do not always contract
Contraction
the same amount. Therefore, the curve has somewhat
different dimensions from those shown for the
individual muscle fiber, but it exhibits the same general
Work Output During Muscle
form for the slope in the normal range of contraction,
Contraction
as noted in Figure 6-9.
Note in Figure 6-9 that when the muscle is at its
When a muscle contracts against a load, it performs
normal resting length, which is at a sarcomere length
work. This means that energy is transferred from the
of about 2 micrometers, it contracts upon activation
muscle to the external load to lift an object to a greater
with the approximate maximum force of contraction.
height or to overcome resistance to movement.
However, the increase in tension that occurs during
In mathematical terms, work is defined by the
contraction, called active tension, decreases as the
following equation:
Chapter 6
Contraction of Skeletal Muscle
79
energy than that of each ATP bond, as is discussed
more fully in Chapters 67 and 72. Therefore, phospho-
creatine is instantly cleaved, and its released energy
30
causes bonding of a new phosphate ion to ADP to
reconstitute the ATP. However, the total amount of
phosphocreatine in the muscle fiber is also very little—
only about five times as great as the ATP. Therefore,
20
the combined energy of both the stored ATP and
the phosphocreatine in the muscle is capable of
causing maximal muscle contraction for only 5 to 8
10
seconds.
The second important source of energy, which is
used to reconstitute both ATP and phosphocreatine,
is
“glycolysis” of glycogen previously stored in the
0
muscle cells. Rapid enzymatic breakdown of the glyco-
0
1
2
3
4
gen to pyruvic acid and lactic acid liberates energy
Load-opposing contraction (kg)
that is used to convert ADP to ATP; the ATP can
then be used directly to energize additional muscle
contraction and also to re-form the stores of
phosphocreatine.
Figure 6-10
The importance of this glycolysis mechanism is
Relation of load to velocity of contraction in a skeletal muscle
twofold. First, the glycolytic reactions can occur even
with a cross section of 1 square centimeter and a length of 8
in the absence of oxygen, so that muscle contraction
centimeters.
can be sustained for many seconds and sometimes up
to more than a minute, even when oxygen delivery
from the blood is not available. Second, the rate of for-
W=L¥D
mation of ATP by the glycolytic process is about 2.5
in which W is the work output, L is the load, and D is
times as rapid as ATP formation in response to cellu-
the distance of movement against the load. The energy
lar foodstuffs reacting with oxygen. However, so
required to perform the work is derived from the
many end products of glycolysis accumulate in the
chemical reactions in the muscle cells during contrac-
muscle cells that glycolysis also loses its capability to
tion, as described in the following sections.
sustain maximum muscle contraction after about 1
minute.
The third and final source of energy is oxidative
Sources of Energy for Muscle
metabolism. This means combining oxygen with the
Contraction
end products of glycolysis and with various other cel-
lular foodstuffs to liberate ATP. More than 95 per cent
We have already seen that muscle contraction depends
of all energy used by the muscles for sustained, long-
on energy supplied by ATP. Most of this energy is
term contraction is derived from this source. The
required to actuate the walk-along mechanism by
foodstuffs that are consumed are carbohydrates, fats,
which the cross-bridges pull the actin filaments, but
and protein. For extremely long-term maximal muscle
small amounts are required for (1) pumping calcium
activity—over a period of many hours—by far the
ions from the sarcoplasm into the sarcoplasmic retic-
greatest proportion of energy comes from fats, but for
ulum after the contraction is over, and (2) pumping
periods of 2 to 4 hours, as much as one half of the
sodium and potassium ions through the muscle fiber
energy can come from stored carbohydrates.
membrane to maintain appropriate ionic environment
The detailed mechanisms of these energetic
for propagation of muscle fiber action potentials.
processes are discussed in Chapters 67 through 72.
The concentration of ATP in the muscle fiber, about
In addition, the importance of the different mecha-
4 millimolar, is sufficient to maintain full contraction
nisms of energy release during performance of
for only 1 to 2 seconds at most. The ATP is split to form
different sports is discussed in Chapter 84 on sports
ADP, which transfers energy from the ATP molecule
physiology.
to the contracting machinery of the muscle fiber. Then,
as described in Chapter 2, the ADP is rephosphory-
Efficiency of Muscle Contraction. The efficiency of an
engine or a motor is calculated as the percentage of
lated to form new ATP within another fraction of a
energy input that is converted into work instead of heat.
second, which allows the muscle to continue its con-
The percentage of the input energy to muscle (the
traction. There are several sources of the energy for
chemical energy in nutrients) that can be converted into
this rephosphorylation.
work, even under the best conditions, is less than 25 per
The first source of energy that is used to reconsti-
cent, with the remainder becoming heat. The reason for
tute the ATP is the substance phosphocreatine, which
this low efficiency is that about one half of the energy
carries a high-energy phosphate bond similar to the
in foodstuffs is lost during the formation of ATP, and
bonds of ATP. The high-energy phosphate bond of
even then, only 40 to 45 per cent of the energy in the
phosphocreatine has a slightly higher amount of free
ATP itself can later be converted into work.
80
Unit II
Membrane Physiology, Nerve, and Muscle
Maximum efficiency can be realized only when the
quadriceps muscle, a half million times as large as the
muscle contracts at a moderate velocity. If the muscle
stapedius. Further, the fibers may be as small as 10
contracts slowly or without any movement, small
micrometers in diameter or as large as 80 micrometers.
amounts of maintenance heat are released during con-
Finally, the energetics of muscle contraction vary con-
traction, even though little or no work is performed,
siderably from one muscle to another. Therefore, it is no
thereby decreasing the conversion efficiency to as little
wonder that the mechanical characteristics of muscle
as zero. Conversely, if contraction is too rapid, large pro-
contraction differ among muscles.
portions of the energy are used to overcome viscous
Figure 6-12 shows records of isometric contractions
friction within the muscle itself, and this, too, reduces the
of three types of skeletal muscle: an ocular muscle,
efficiency of contraction. Ordinarily, maximum effi-
which has a duration of isometric contraction of less
ciency is developed when the velocity of contraction is
than 1/40 second; the gastrocnemius muscle, which has
about 30 per cent of maximum.
a duration of contraction of about 1/15 second; and the
soleus muscle, which has a duration of contrac-
tion of about 1/3 second. It is interesting that these dura-
Characteristics of Whole
tions of contraction are adapted to the functions of the
respective muscles. Ocular movements must be
Muscle Contraction
extremely rapid to maintain fixation of the eyes on
Many features of muscle contraction can be demon-
specific objects to provide accuracy of vision. The gas-
strated by eliciting single muscle twitches. This can be
trocnemius muscle must contract moderately rapidly
accomplished by instantaneous electrical excitation of
to provide sufficient velocity of limb movement for
the nerve to a muscle or by passing a short electrical
running and jumping, and the soleus muscle is con-
stimulus through the muscle itself, giving rise to a single,
cerned principally with slow contraction for continual,
sudden contraction lasting for a fraction of a second.
long-term support of the body against gravity.
Isometric Versus Isotonic Contraction. Muscle contraction is
Fast Versus Slow Muscle Fibers. As we discuss more fully in
said to be isometric when the muscle does not shorten
Chapter 84 on sports physiology, every muscle of the
during contraction and isotonic when it does shorten but
body is composed of a mixture of so-called fast and slow
the tension on the muscle remains constant throughout
muscle fibers, with still other fibers gradated between
the contraction. Systems for recording the two types of
these two extremes. The muscles that react rapidly are
muscle contraction are shown in Figure 6-11.
composed mainly of “fast” fibers with only small
In the isometric system, the muscle contracts against
numbers of the slow variety. Conversely, the muscles
a force transducer without decreasing the muscle
that respond slowly but with prolonged contraction are
length, as shown on the right in Figure 6-11. In the iso-
composed mainly of “slow” fibers. The differences
tonic system, the muscle shortens against a fixed load;
between these two types of fibers are as follows.
this is illustrated on the left in the figure, showing a
muscle lifting a pan of weights. The characteristics of
Fast Fibers. (1) Large fibers for great strength of con-
isotonic contraction depend on the load against which
traction. (2) Extensive sarcoplasmic reticulum for rapid
the muscle contracts, as well as the inertia of the load.
release of calcium ions to initiate contraction. (3) Large
However, the isometric system records strictly changes
amounts of glycolytic enzymes for rapid release of
in force of muscle contraction itself. Therefore, the iso-
energy by the glycolytic process. (4) Less extensive
metric system is most often used when comparing the
blood supply because oxidative metabolism is of
functional characteristics of different muscle types.
Characteristics of Isometric Twitches Recorded from Different
Muscles. The human body has many sizes of skeletal
muscles—from the very small stapedius muscle in the
middle ear, measuring only a few millimeters long and
a millimeter or so in diameter, up to the very large
Duration of
depolarization
Soleus
Stimulating
Stimulating
electrodes
electrodes
Gastrocnemius
Kymograph
Muscle
Ocular
muscle
Electronic force
0
40
80
120
160
200
Weights
transducer
Milliseconds
To electronic
recorder
ISOTONIC SYSTEM
ISOMETRIC SYSTEM
Figure 6-12
Figure 6-11
Duration of isometric contractions for different types of mammalian
skeletal muscles, showing a latent period between the action
Isotonic and isometric systems for recording muscle contractions.
potential (depolarization) and muscle contraction.
Chapter 6
Contraction of Skeletal Muscle
81
secondary importance. (5) Fewer mitochondria, also
because oxidative metabolism is secondary.
Slow Fibers. (1) Smaller fibers. (2) Also innervated by
smaller nerve fibers. (3) More extensive blood vessel
system and capillaries to supply extra amounts of
Tetanization
oxygen. (4) Greatly increased numbers of mitochondria,
also to support high levels of oxidative metabolism. (5)
Fibers contain large amounts of myoglobin, an iron-
containing protein similar to hemoglobin in red blood
cells. Myoglobin combines with oxygen and stores it
until needed; this also greatly speeds oxygen transport
to the mitochondria. The myoglobin gives the slow
muscle a reddish appearance and the name red muscle,
whereas a deficit of red myoglobin in fast muscle gives
5
10
15
20
25
30
35
40
45
50
55
it the name white muscle.
Rate of stimulation (times per second)
Mechanics of Skeletal Muscle
Contraction
Figure 6-13
Motor Unit. Each motoneuron that leaves the spinal cord
Frequency summation and tetanization.
innervates multiple muscle fibers, the number depend-
ing on the type of muscle. All the muscle fibers inner-
vated by a single nerve fiber are called a motor unit. In
Another important feature of multiple fiber summa-
general, small muscles that react rapidly and whose
tion is that the different motor units are driven asyn-
control must be exact have more nerve fibers for fewer
chronously by the spinal cord, so that contraction
muscle fibers (for instance, as few as two or three muscle
alternates among motor units one after the other, thus
fibers per motor unit in some of the laryngeal muscles).
providing smooth contraction even at low frequencies
Conversely, large muscles that do not require fine
of nerve signals.
control, such as the soleus muscle, may have several
hundred muscle fibers in a motor unit. An average
Frequency Summation and Tetanization. Figure 6-13
figure for all the muscles of the body is questionable, but
shows the principles of frequency summation and
a good guess would be about 80 to 100 muscle fibers to
tetanization. To the left are displayed individual twitch
a motor unit.
contractions occurring one after another at low fre-
The muscle fibers in each motor unit are not all
quency of stimulation. Then, as the frequency increases,
bunched together in the muscle but overlap other motor
there comes a point where each new contraction occurs
units in microbundles of 3 to 15 fibers. This interdigita-
before the preceding one is over. As a result, the second
tion allows the separate motor units to contract in
contraction is added partially to the first, so that the
support of one another rather than entirely as individ-
total strength of contraction rises progressively with
ual segments.
increasing frequency. When the frequency reaches a
critical level, the successive contractions eventually
Muscle Contractions of Different Force—Force Summation.
become so rapid that they fuse together, and the whole
Summation means the adding together of individual
muscle contraction appears to be completely smooth
twitch contractions to increase the intensity of overall
and continuous, as shown in the figure. This is called
muscle contraction. Summation occurs in two ways: (1)
tetanization. At a slightly higher frequency, the strength
by increasing the number of motor units contracting
of contraction reaches its maximum, so that any addi-
simultaneously, which is called multiple fiber summa-
tional increase in frequency beyond that point has no
tion, and (2) by increasing the frequency of contraction,
further effect in increasing contractile force. This occurs
which is called frequency summation and can lead to
because enough calcium ions are maintained in the
tetanization.
muscle sarcoplasm, even between action potentials, so
that full contractile state is sustained without allowing
Multiple Fiber Summation. When the central nervous
any relaxation between the action potentials.
system sends a weak signal to contract a muscle, the
smaller motor units of the muscle may be stimulated in
Maximum Strength of Contraction. The maximum strength
preference to the larger motor units. Then, as the
of tetanic contraction of a muscle operating at a normal
strength of the signal increases, larger and larger motor
muscle length averages between 3 and 4 kilograms per
units begin to be excited as well, with the largest motor
square centimeter of muscle, or 50 pounds per square
units often having as much as 50 times the contractile
inch. Because a quadriceps muscle can have as much as
force of the smallest units. This is called the size princi-
16 square inches of muscle belly, as much as 800 pounds
ple. It is important, because it allows the gradations of
of tension may be applied to the patellar tendon. Thus,
muscle force during weak contraction to occur in small
one can readily understand how it is possible for
steps, whereas the steps become progressively greater
muscles to pull their tendons out of their insertions in
when large amounts of force are required. The cause of
bone.
this size principle is that the smaller motor units are
driven by small motor nerve fibers, and the small
Changes in Muscle Strength at the Onset of Contraction—The
motoneurons in the spinal cord are more excitable than
Staircase Effect (Treppe). When a muscle begins to con-
the larger ones, so they naturally are excited first.
tract after a long period of rest, its initial strength of
82
Unit II
Membrane Physiology, Nerve, and Muscle
contraction may be as little as one half its strength 10
biceps muscle has a cross-sectional area of 6 square
to 50 muscle twitches later. That is, the strength of con-
inches, the maximum force of contraction would be
traction increases to a plateau, a phenomenon called the
about 300 pounds. When the forearm is at right angles
staircase effect, or treppe.
with the upper arm, the tendon attachment of the biceps
Although all the possible causes of the staircase effect
is about 2 inches anterior to the fulcrum at the elbow,
are not known, it is believed to be caused primarily by
and the total length of the forearm lever is about 14
increasing calcium ions in the cytosol because of the
inches. Therefore, the amount of lifting power of the
release of more and more ions from the sarcoplasmic
biceps at the hand would be only one seventh of the 300
reticulum with each successive muscle action potential
pounds of muscle force, or about 43 pounds. When the
and failure of the sarcoplasm to recapture the ions
arm is fully extended, the attachment of the biceps is
immediately.
much less than 2 inches anterior to the fulcrum, and the
force with which the hand can be brought forward is
Skeletal Muscle Tone. Even when muscles are at rest, a
also much less than 43 pounds.
certain amount of tautness usually remains. This is
In short, an analysis of the lever systems of the body
called muscle tone. Because normal skeletal muscle
depends on knowledge of (1) the point of muscle inser-
fibers do not contract without an action potential to
tion, (2) its distance from the fulcrum of the lever, (3)
stimulate the fibers, skeletal muscle tone results entirely
the length of the lever arm, and (4) the position of the
from a low rate of nerve impulses coming from the
lever. Many types of movement are required in the
spinal cord. These, in turn, are controlled partly by
body, some of which need great strength and others
signals transmitted from the brain to the appropriate
of which need large distances of movement. For this
spinal cord anterior motoneurons and partly by signals
reason, there are many different types of muscle; some
that originate in muscle spindles located in the muscle
are long and contract a long distance, and some are
itself. Both of these are discussed in relation to muscle
short but have large cross-sectional areas and can
spindle and spinal cord function in Chapter 54.
provide extreme strength of contraction over short dis-
tances. The study of different types of muscles, lever
Muscle Fatigue. Prolonged and strong contraction of a
systems, and their movements is called kinesiology
muscle leads to the well-known state of muscle fatigue.
and is an important scientific component of human
Studies in athletes have shown that muscle fatigue
physioanatomy.
increases in almost direct proportion to the rate of
“Positioning” of a Body Part by Contraction of Agonist and Antag-
depletion of muscle glycogen. Therefore, fatigue results
onist Muscles on Opposite Sides of a Joint—“Coactivation”
mainly from inability of the contractile and metabolic
of Antagonist Muscles. Virtually all body movements are
processes of the muscle fibers to continue supplying the
caused by simultaneous contraction of agonist and
same work output. However, experiments have also
antagonist muscles on opposite sides of joints. This
shown that transmission of the nerve signal through the
is called coactivation of the agonist and antagonist
neuromuscular junction, which is discussed in Chapter
muscles, and it is controlled by the motor control centers
7, can diminish at least a small amount after intense pro-
of the brain and spinal cord.
longed muscle activity, thus further diminishing muscle
The position of each separate part of the body, such
contraction. Interruption of blood flow through a con-
as an arm or a leg, is determined by the relative degrees
tracting muscle leads to almost complete muscle fatigue
of contraction of the agonist and antagonist sets of
within 1 or 2 minutes because of the loss of nutrient
muscles. For instance, let us assume that an arm or a leg
supply, especially loss of oxygen.
is to be placed in a midrange position. To achieve
this, agonist and antagonist muscles are excited about
Lever Systems of the Body. Muscles operate by applying
equally. Remember that an elongated muscle contracts
tension to their points of insertion into bones, and the
with more force than a shortened muscle, which was
bones in turn form various types of lever systems. Figure
demonstrated in Figure 6-9, showing maximum strength
6-14 shows the lever system activated by the biceps
of contraction at full functional muscle length and
muscle to lift the forearm. If we assume that a large
almost no strength of contraction at half normal length.
Therefore, the elongated muscle on one side of a joint
can contract with far greater force than the shorter
muscle on the opposite side. As an arm or leg moves
toward its midposition, the strength of the longer
muscle decreases, whereas the strength of the shorter
muscle increases until the two strengths equal each
other. At this point, movement of the arm or leg stops.
Thus, by varying the ratios of the degree of activation
of the agonist and antagonist muscles, the nervous
system directs the positioning of the arm or leg.
We learn in Chapter 54 that the motor nervous
system has additional important mechanisms to com-
pensate for different muscle loads when directing this
positioning process.
Remodeling of Muscle
to Match Function
Figure 6-14
All the muscles of the body are continually being
Lever system activated by the biceps muscle.
remodeled to match the functions that are required of
Chapter 6
Contraction of Skeletal Muscle
83
them. Their diameters are altered, their lengths are
about 2 months, degenerative changes also begin to
altered, their strengths are altered, their vascular sup-
appear in the muscle fibers themselves. If the nerve
plies are altered, and even the types of muscle fibers are
supply to the muscle grows back rapidly, full return
altered at least slightly. This remodeling process is often
of function can occur in as little as 3 months, but from
quite rapid, within a few weeks. Indeed, experiments in
that time onward, the capability of functional return
animals have shown that muscle contractile proteins in
becomes less and less, with no further return of function
some smaller, more active muscles can be replaced in as
after 1 to 2 years.
little as 2 weeks.
In the final stage of denervation atrophy, most of the
muscle fibers are destroyed and replaced by fibrous and
Muscle Hypertrophy and Muscle Atrophy. When the total
fatty tissue. The fibers that do remain are composed of
mass of a muscle increases, this is called muscle hyper-
a long cell membrane with a lineup of muscle cell nuclei
trophy. When it decreases, the process is called muscle
but with few or no contractile properties and little or no
atrophy.
capability of regenerating myofibrils if a nerve does
Virtually all muscle hypertrophy results from an
regrow.
increase in the number of actin and myosin filaments in
The fibrous tissue that replaces the muscle fibers
each muscle fiber, causing enlargement of the individ-
during denervation atrophy also has a tendency to con-
ual muscle fibers; this is called simply fiber hypertrophy.
tinue shortening for many months, which is called con-
Hypertrophy occurs to a much greater extent when the
tracture. Therefore, one of the most important problems
muscle is loaded during the contractile process. Only a
in the practice of physical therapy is to keep atrophying
few strong contractions each day are required to cause
muscles from developing debilitating and disfiguring
significant hypertrophy within 6 to 10 weeks.
contractures. This is achieved by daily stretching of the
The manner in which forceful contraction leads to
muscles or use of appliances that keep the muscles
hypertrophy is not known. It is known, however, that the
stretched during the atrophying process.
rate of synthesis of muscle contractile proteins is far
Recovery of Muscle Contraction in Poliomyelitis: Devel-
greater when hypertrophy is developing, leading also to
opment of Macromotor Units. When some but not all
progressively greater numbers of both actin and myosin
nerve fibers to a muscle are destroyed, as commonly
filaments in the myofibrils, often increasing as much as
occurs in poliomyelitis, the remaining nerve fibers
50 per cent. In turn, some of the myofibrils themselves
branch off to form new axons that then innervate many
have been observed to split within hypertrophying
of the paralyzed muscle fibers. This causes large motor
muscle to form new myofibrils, but how important this
units called macromotor units, which can contain as
is in usual muscle hypertrophy is still unknown.
many as five times the normal number of muscle fibers
Along with the increasing size of myofibrils, the
for each motoneuron coming from the spinal cord. This
enzyme systems that provide energy also increase. This
decreases the fineness of control one has over the
is especially true of the enzymes for glycolysis, allowing
muscles but does allow the muscles to regain varying
rapid supply of energy during short-term forceful
degrees of strength.
muscle contraction.
When a muscle remains unused for many weeks, the
rate of decay of the contractile proteins is more rapid
Rigor Mortis
than the rate of replacement. Therefore, muscle atrophy
occurs.
Several hours after death, all the muscles of the body go
into a state of contracture called “rigor mortis”; that is,
Adjustment of Muscle Length. Another type of hyper-
the muscles contract and become rigid, even without
trophy occurs when muscles are stretched to greater
action potentials. This rigidity results from loss of all the
than normal length. This causes new sarcomeres to be
ATP, which is required to cause separation of the cross-
added at the ends of the muscle fibers, where they attach
bridges from the actin filaments during the relaxation
to the tendons. In fact, new sarcomeres can be added
process. The muscles remain in rigor until the muscle
as rapidly as several per minute in newly developing
proteins deteriorate about 15 to 25 hours later, which
muscle, illustrating the rapidity of this type of
presumably results from autolysis caused by enzymes
hypertrophy.
released from lysosomes. All these events occur more
Conversely, when a muscle continually remains short-
rapidly at higher temperatures.
ened to less than its normal length, sarcomeres at the
ends of the muscle fibers can actually disappear. It is by
these processes that muscles are continually remodeled
to have the appropriate length for proper muscle
References
contraction.
Berchtold MW, Brinkmeier H, Muntener M: Calcium ion
Hyperplasia of Muscle Fibers. Under rare conditions of
in skeletal muscle: its crucial role for muscle function,
extreme muscle force generation, the actual number of
plasticity, and disease. Physiol Rev 80:1215, 2000.
muscle fibers has been observed to increase (but only
Brooks SV: Current topics for teaching skeletal muscle phys-
by a few percentage points), in addition to the fiber
iology. Adv Physiol Educ 27:171, 2003.
hypertrophy process. This increase in fiber number is
Clausen T: Na+-K+ pump regulation and skeletal muscle con-
called fiber hyperplasia. When it does occur, the mech-
tractility. Physiol Rev 83:1269, 2003.
anism is linear splitting of previously enlarged fibers.
Glass DJ: Molecular mechanisms modulating muscle mass.
Trends Mol Med 8:344, 2003.
Effects of Muscle Denervation. When a muscle loses its
Glass DJ: Signalling pathways that mediate skeletal muscle
nerve supply, it no longer receives the contractile signals
hypertrophy and atrophy. Nat Cell Biol 5:87, 2003.
that are required to maintain normal muscle size.
Gordon AM, Homsher E, Regnier M: Regulation of con-
Therefore, atrophy begins almost immediately. After
traction in striated muscle. Physiol Rev 80:853, 2000.
84
Unit II
Membrane Physiology, Nerve, and Muscle
Gordon AM, Regnier M, Homsher E: Skeletal and cardiac
MacIntosh BR: Role of calcium sensitivity modulation in
muscle contractile activation: tropomyosin “rocks and
skeletal muscle performance. News Physiol Sci 18:222,
rolls.” News Physiol Sci 16:49, 2001.
2003.
Huxley AF, Gordon AM: Striation patterns in active and
Matthews GG: Cellular Physiology of Nerve and Muscle.
passive shortening of muscle. Nature
(Lond) 193:280,
Malden, MA: Blackwell Science, 1998.
1962.
Sieck GC, Regnier M: Plasticity and energetic demands of
Huxley HE: A personal view of muscle and motility mecha-
contraction in skeletal and cardiac muscle. J Appl Physiol
nisms. Annu Rev Physiol 58:1, 1996.
90:1158, 2001.
Jurkat-Rott K, Lerche H, Lehmann-Horn F: Skeletal muscle
Stamler JS, Meissner G: Physiology of nitric oxide in skele-
channelopathies. J Neurol 249:1493, 2002.
tal muscle. Physiol Rev 81:209, 2001.
Kjær M: Role of extracellular matrix in adaptation of tendon
Szent-Gyorgyi AG: Regulation of contraction by calcium
and skeletal muscle to mechanical loading. Physiol Rev
binding myosins. Biophys Chem 59:357, 1996.
84:649, 2004.
C H A P T E R
7
Excitation of Skeletal Muscle:
Neuromuscular Transmission and
Excitation-Contraction Coupling
Transmission of Impulses
from Nerve Endings to
Skeletal Muscle Fibers:
The Neuromuscular
Junction
The skeletal muscle fibers are innervated by large,
myelinated nerve fibers that originate from large motoneurons in the anterior
horns of the spinal cord. As pointed out in Chapter 6, each nerve fiber, after
entering the muscle belly, normally branches and stimulates from three to
several hundred skeletal muscle fibers. Each nerve ending makes a junction,
called the neuromuscular junction, with the muscle fiber near its midpoint. The
action potential initiated in the muscle fiber by the nerve signal travels in both
directions toward the muscle fiber ends. With the exception of about 2 per cent
of the muscle fibers, there is only one such junction per muscle fiber.
Physiologic Anatomy of the Neuromuscular Junction—The Motor End Plate. Figure 7-1A
and B shows the neuromuscular junction from a large, myelinated nerve fiber
to a skeletal muscle fiber. The nerve fiber forms a complex of branching nerve
terminals that invaginate into the surface of the muscle fiber but lie outside the
muscle fiber plasma membrane. The entire structure is called the motor end
plate. It is covered by one or more Schwann cells that insulate it from the
surrounding fluids.
Figure 7-1C shows an electron micrographic sketch of the junction between
a single axon terminal and the muscle fiber membrane. The invaginated mem-
brane is called the synaptic gutter or synaptic trough, and the space between the
terminal and the fiber membrane is called the synaptic space or synaptic cleft.
This space is 20 to 30 nanometers wide. At the bottom of the gutter are numer-
ous smaller folds of the muscle membrane called subneural clefts, which greatly
increase the surface area at which the synaptic transmitter can act.
In the axon terminal are many mitochondria that supply adenosine triphos-
phate (ATP), the energy source that is used for synthesis of an excitatory
transmitter acetylcholine. The acetylcholine in turn excites the muscle
fiber membrane. Acetylcholine is synthesized in the cytoplasm of the terminal,
but it is absorbed rapidly into many small synaptic vesicles, about 300,000
of which are normally in the terminals of a single end plate. In the synaptic
space are large quantities of the enzyme acetylcholinesterase, which destroys
acetylcholine a few milliseconds after it has been released from the synaptic
vesicles.
Secretion of Acetylcholine by the Nerve Terminals
When a nerve impulse reaches the neuromuscular junction, about 125 vesicles
of acetylcholine are released from the terminals into the synaptic space. Some
of the details of this mechanism can be seen in Figure 7-2, which shows an
expanded view of a synaptic space with the neural membrane above and the
muscle membrane and its subneural clefts below.
85
86
Unit II
Membrane Physiology, Nerve, and Muscle
Axon
Myelin
sheath
Terminal nerve
branches
Teloglial cell
Myofibrils
Muscle
nuclei
A
B
Axon terminal in
Synaptic vesicles
synaptic trough
Figure 7-1
Different views of the motor end
plate. A, Longitudinal section
through the end plate. B, Surface
view of the end plate. C, Electron
micrographic appearance of the
contact point between a single
axon terminal and the muscle
fiber membrane. (Redrawn from
Fawcett DW, as modified from
Couteaux R, in Bloom W, Fawcett
DW: A Textbook of Histology.
Philadelphia: WB Saunders,
C
Subneural clefts
1986.)
On the inside surface of the neural membrane are
Release
Neural
Vesicles
linear dense bars, shown in cross section in Figure 7-2.
sites
membrane
To each side of each dense bar are protein particles
that penetrate the neural membrane; these are voltage-
Dense bar
gated calcium channels. When an action potential
spreads over the terminal, these channels open and
Calcium
channels
allow calcium ions to diffuse from the synaptic space
to the interior of the nerve terminal. The calcium ions,
Basal lamina
and
in turn, are believed to exert an attractive influence on
acetylcholinesterase
the acetylcholine vesicles, drawing them to the neural
membrane adjacent to the dense bars. The vesicles
Acetylcholine
then fuse with the neural membrane and empty their
receptors
acetylcholine into the synaptic space by the process of
exocytosis.
Subneural cleft
Although some of the aforementioned details are
speculative, it is known that the effective stimulus for
causing acetylcholine release from the vesicles is entry
of calcium ions and that acetylcholine from the vesi-
cles is then emptied through the neural membrane
Muscle
adjacent to the dense bars.
membrane
Figure 7-2
Effect of Acetylcholine on the Postsynaptic Muscle Fiber Mem-
brane to Open Ion Channels. Figure 7-2 also shows many
Release of acetylcholine from synaptic vesicles at the neural
very small acetylcholine receptors in the muscle fiber
membrane of the neuromuscular junction. Note the proximity of
membrane; these are acetylcholine-gated ion channels,
the release sites in the neural membrane to the acetylcholine
receptors in the muscle membrane, at the mouths of the subneural
and they are located almost entirely near the mouths
clefts.
of the subneural clefts lying immediately below the
dense bar areas, where the acetylcholine is emptied
into the synaptic space.
Each receptor is a protein complex that has a total
molecular weight of 275,000. The complex is composed
Chapter 7
Excitation of Skeletal Muscle: Neuromuscular Transmission and Excitation-Contraction Coupling
87
of five subunit proteins, two alpha proteins and one
In practice, far more sodium ions flow through the
each of beta, delta, and gamma proteins. These protein
acetylcholine channels than any other ions, for two
molecules penetrate all the way through the mem-
reasons. First, there are only two positive ions in large
brane, lying side by side in a circle to form a tubular
concentration: sodium ions in the extracellular fluid,
channel, illustrated in Figure 7-3. The channel remains
and potassium ions in the intracellular fluid. Second,
constricted, as shown in section A of the figure, until
the very negative potential on the inside of the muscle
two acetylcholine molecules attach respectively to the
membrane, -80 to -90 millivolts, pulls the positively
two alpha subunit proteins. This causes a conforma-
charged sodium ions to the inside of the fiber, while
tional change that opens the channel, as shown in
simultaneously preventing efflux of the positively
section B of the figure.
charged potassium ions when they attempt to pass
The opened acetylcholine channel has a diameter of
outward.
about 0.65 nanometer, which is large enough to allow
As shown in Figure 7-3B, the principal effect of
the important positive ions—sodium (Na+), potassium
opening the acetylcholine-gated channels is to allow
(K+), and calcium (Ca++)—to move easily through the
large numbers of sodium ions to pour to the inside of
opening. Conversely, negative ions, such as chloride
the fiber, carrying with them large numbers of positive
ions, do not pass through because of strong negative
charges. This creates a local positive potential change
charges in the mouth of the channel that repel these
inside the muscle fiber membrane, called the end plate
negative ions.
potential. In turn, this end plate potential initiates an
action potential that spreads along the muscle mem-
brane and thus causes muscle contraction.
Destruction of the Released Acetylcholine by Acetyl-
cholinesterase. The acetylcholine, once released into
-
-
the synaptic space, continues to activate the acetyl-
-
-
choline receptors as long as the acetylcholine persists
–
-
in the space. However, it is removed rapidly by two
means: (1) Most of the acetylcholine is destroyed
by the enzyme acetylcholinesterase, which is attached
mainly to the spongy layer of fine connective tissue
that fills the synaptic space between the presynaptic
nerve terminal and the postsynaptic muscle mem-
brane. (2) A small amount of acetylcholine diffuses out
of the synaptic space and is then no longer available
to act on the muscle fiber membrane.
The short time that the acetylcholine remains in the
synaptic space—a few milliseconds at most—normally
A
is sufficient to excite the muscle fiber. Then the rapid
removal of the acetylcholine prevents continued
Na+
Ach
muscle re-excitation after the muscle fiber has re-
covered from its initial action potential.
-
-
End Plate Potential and Excitation of the Skeletal Muscle Fiber.
-
-
The sudden insurgence of sodium ions into the muscle
-
-
fiber when the acetylcholine channels open causes the
electrical potential inside the fiber at the local area of
the end plate to increase in the positive direction as
much as 50 to 75 millivolts, creating a local potential
called the end plate potential. Recall from Chapter 5
that a sudden increase in nerve membrane potential
of more than 20 to 30 millivolts is normally sufficient
to initiate more and more sodium channel opening,
thus initiating an action potential at the muscle fiber
membrane.
Figure 7-4 shows the principle of an end plate
B
potential initiating the action potential. This figure
shows three separate end plate potentials. End plate
Figure 7-3
potentials A and C are too weak to elicit an action
Acetylcholine channel.
A,
Closed state. B,
After acetylcholine
potential, but they do produce weak local end plate
(Ach) has become attached and a conformational change has
voltage changes, as recorded in the figure. By contrast,
opened the channel, allowing sodium ions to enter the muscle
end plate potential B is much stronger and causes
fiber and excite contraction. Note the negative charges at the
enough sodium channels to open so that the self-
channel mouth that prevent passage of negative ions such as
chloride ions.
regenerative effect of more and more sodium ions
88
Unit II
Membrane Physiology, Nerve, and Muscle
nervous system for which most of the details of chemi-
cal transmission have been worked out. The formation
+ 60
and release of acetylcholine at this junction occur in the
+ 40
following stages:
+ 20
1.
Small vesicles, about 40 nanometers in size, are
formed by the Golgi apparatus in the cell body of
0
the motoneuron in the spinal cord. These vesicles
- 20
are then transported by axoplasm that “streams”
- 40
through the core of the axon from the central
- 60
cell body in the spinal cord all the way to the
neuromuscular junction at the tips of the peripheral
- 80
A
B
C
nerve fibers. About 300,000 of these small vesicles
-100
collect in the nerve terminals of a single skeletal
0
15
30
45
60
75
muscle end plate.
2.
Acetylcholine is synthesized in the cytosol of the
Milliseconds
nerve fiber terminal but is immediately transported
through the membranes of the vesicles to their
interior, where it is stored in highly concentrated
Figure 7-4
form, about 10,000 molecules of acetylcholine in
each vesicle.
End plate potentials (in millivolts). A, Weakened end plate poten-
3.
When an action potential arrives at the nerve
tial recorded in a curarized muscle, too weak to elicit an action
terminal, it opens many calcium channels in the
potential. B, Normal end plate potential eliciting a muscle action
membrane of the nerve terminal because this
potential. C, Weakened end plate potential caused by botulinum
terminal has an abundance of voltage-gated
toxin that decreases end plate release of acetylcholine, again too
weak to elicit a muscle action potential.
calcium channels. As a result, the calcium ion
concentration inside the terminal membrane
increases about 100-fold, which in turn increases
the rate of fusion of the acetylcholine vesicles with
flowing to the interior of the fiber initiates an action
the terminal membrane about 10,000-fold. This
potential. The weakness of the end plate potential at
fusion makes many of the vesicles rupture, allowing
exocytosis of acetylcholine into the synaptic space.
point A was caused by poisoning of the muscle fiber
About 125 vesicles usually rupture with each
with curare, a drug that blocks the gating action of
action potential. Then, after a few milliseconds, the
acetylcholine on the acetylcholine channels by com-
acetylcholine is split by acetylcholinesterase
peting for the acetylcholine receptor sites. The weak-
into acetate ion and choline, and the choline is
ness of the end plate potential at point C resulted from
reabsorbed actively into the neural terminal to be
the effect of botulinum toxin, a bacterial poison that
reused to form new acetylcholine. This sequence of
decreases the quantity of acetylcholine release by the
events occurs within a period of 5 to 10
nerve terminals.
milliseconds.
4.
The number of vesicles available in the nerve
Safety Factor for Transmission at the Neuromuscular Junction;
ending is sufficient to allow transmission of only a
few thousand nerve-to-muscle impulses. Therefore,
Fatigue of the Junction. Ordinarily, each impulse that
for continued function of the neuromuscular
arrives at the neuromuscular junction causes about
junction, new vesicles need to be re-formed rapidly.
three times as much end plate potential as that
Within a few seconds after each action potential is
required to stimulate the muscle fiber. Therefore, the
over, “coated pits” appear in the terminal nerve
normal neuromuscular junction is said to have a high
membrane, caused by contractile proteins in the
safety factor. However, stimulation of the nerve fiber
nerve ending, especially the protein clathrin, which
at rates greater than 100 times per second for several
is attached to the membrane in the areas of the
minutes often diminishes the number of acetylcholine
original vesicles. Within about 20 seconds, the
vesicles so much that impulses fail to pass into the
proteins contract and cause the pits to break away
muscle fiber. This is called fatigue of the neuromuscu-
to the interior of the membrane, thus forming new
vesicles. Within another few seconds, acetylcholine
lar junction, and it is the same effect that causes fatigue
is transported to the interior of these vesicles, and
of synapses in the central nervous system when the
they are then ready for a new cycle of acetylcholine
synapses are overexcited. Under normal functioning
release.
conditions, measurable fatigue of the neuromuscular
junction occurs rarely, and even then only at the most
exhausting levels of muscle activity.
Drugs That Enhance or
Block Transmission at the
Molecular Biology of
Neuromuscular Junction
Acetylcholine Formation
Drugs That Stimulate the Muscle Fiber by Acetylcholine-Like
and Release
Action. Many compounds, including methacholine, car-
Because the neuromuscular junction is large enough to
bachol, and nicotine, have the same effect on the muscle
be studied easily, it is one of the few synapses of the
fiber as does acetylcholine. The difference between
Chapter 7
Excitation of Skeletal Muscle: Neuromuscular Transmission and Excitation-Contraction Coupling
89
these drugs and acetylcholine is that the drugs are not
Muscle Action Potential
destroyed by cholinesterase or are destroyed so slowly
that their action often persists for many minutes to
Almost everything discussed in Chapter 5 regarding
several hours. The drugs work by causing localized areas
initiation and conduction of action potentials in nerve
of depolarization of the muscle fiber membrane at the
fibers applies equally to skeletal muscle fibers, except
motor end plate where the acetylcholine receptors
for quantitative differences. Some of the quantitative
are located. Then, every time the muscle fiber recovers
from a previous contraction, these depolarized areas, by
aspects of muscle potentials are the following:
virtue of leaking ions, initiate a new action potential,
1. Resting membrane potential: about -80 to -90
thereby causing a state of muscle spasm.
millivolts in skeletal fibers—the same as in large
myelinated nerve fibers.
Drugs That Stimulate the Neuromuscular Junction by
2. Duration of action potential: 1 to 5 milliseconds in
Inactivating Acetylcholinesterase. Three particularly well-
skeletal muscle—about five times as long as in
known drugs, neostigmine, physostigmine, and
large myelinated nerves.
diisopropyl fluorophosphate, inactivate the acetyl-
3. Velocity of conduction: 3 to 5 m/sec—about 1/13
cholinesterase in the synapses so that it no longer
the velocity of conduction in the large myelinated
hydrolyzes acetylcholine. Therefore, with each succes-
nerve fibers that excite skeletal muscle.
sive nerve impulse, additional acetylcholine accumu-
lates and stimulates the muscle fiber repetitively. This
causes muscle spasm when even a few nerve impulses
reach the muscle. Unfortunately, it also can cause death
Spread of the Action Potential to the
due to laryngeal spasm, which smothers the person.
Interior of the Muscle Fiber by Way of
Neostigmine and physostigmine combine with acetyl-
“Transverse Tubules”
cholinesterase to inactivate the acetylcholinesterase
for up to several hours, after which these drugs are
The skeletal muscle fiber is so large that action poten-
displaced from the acetylcholinesterase so that the
esterase once again becomes active. Conversely, diiso-
tials spreading along its surface membrane cause
propyl fluorophosphate, which has military potential
almost no current flow deep within the fiber. Yet, to
as a powerful “nerve” gas poison, inactivates acetyl-
cause maximum muscle contraction, current must pen-
cholinesterase for weeks, which makes this a particu-
etrate deeply into the muscle fiber to the vicinity of the
larly lethal poison.
separate myofibrils. This is achieved by transmission of
action potentials along transverse tubules (T tubules)
Drugs That Block Transmission at the Neuromuscular Junction. A
that penetrate all the way through the muscle fiber
group of drugs known as curariform drugs can prevent
from one side of the fiber to the other, as illustrated in
passage of impulses from the nerve ending into the
Figure
7-5. The T tubule action potentials cause
muscle. For instance, D-tubocurarine blocks the action
release of calcium ions inside the muscle fiber in the
of acetylcholine on the muscle fiber acetylcholine recep-
tors, thus preventing sufficient increase in permeability
immediate vicinity of the myofibrils, and these calcium
of the muscle membrane channels to initiate an action
ions then cause contraction. This overall process is
potential.
called excitation-contraction coupling.
Myasthenia Gravis
Excitation-Contraction
Coupling
Myasthenia gravis, which occurs in about 1 in every
20,000 persons, causes muscle paralysis because of
inability of the neuromuscular junctions to transmit
Transverse Tubule-Sarcoplasmic
enough signals from the nerve fibers to the muscle
Reticulum System
fibers. Pathologically, antibodies that attack the acetyl-
choline-gated sodium ion transport proteins have been
Figure 7-5 shows myofibrils surrounded by the T
demonstrated in the blood of most patients with
tubule-sarcoplasmic reticulum system. The T tubules
myasthenia gravis. Therefore, it is believed that myas-
thenia gravis is an autoimmune disease in which the
are very small and run transverse to the myofibrils.
patients have developed immunity against their own
They begin at the cell membrane and penetrate all the
acetylcholine-activated ion channels.
way from one side of the muscle fiber to the opposite
Regardless of the cause, the end plate potentials that
side. Not shown in the figure is the fact that these
occur in the muscle fibers are mostly too weak to stimu-
tubules branch among themselves so that they form
late the muscle fibers. If the disease is intense enough,
entire planes of T tubules interlacing among all the
the patient dies of paralysis—in particular, paralysis of
separate myofibrils. Also, where the T tubules originate
the respiratory muscles. The disease usually can be ame-
from the cell membrane, they are open to the exterior
liorated for several hours by administering neostigmine
of the muscle fiber. Therefore, they communicate with
or some other anticholinesterase drug, which allows
the extracellular fluid surrounding the muscle fiber,
larger than normal amounts of acetylcholine to accu-
mulate in the synaptic space. Within minutes, some of
and they themselves contain extracellular fluid in their
these paralyzed people can begin to function almost
lumens. In other words, the T tubules are actually
normally, until a new dose of neostigmine is required a
internal extensions of the cell membrane. Therefore,
few hours later.
when an action potential spreads over a muscle fiber
90
Unit II
Membrane Physiology, Nerve, and Muscle
Myofibrils
Sarcolemma
Terminal
Triad of the
cisternae
reticulum
Z line
Transverse
tubule
Mitochondrion
Figure 7-5
Transverse
(T) tubule-sarcoplas-
A band
mic reticulum system. Note that
the T tubules communicate with the
outside of the cell membrane, and
deep in the muscle fiber, each T
Sarcoplasmic
tubule lies adjacent to the ends of
reticulum
longitudinal sarcoplasmic reticulum
tubules that surround all sides of
the actual myofibrils that contract.
This illustration was drawn from frog
muscle, which has one T tubule per
Transverse
sarcomere, located at the Z line. A
I band
tubule
similar arrangement is found in
mammalian heart muscle, but
mammalian skeletal muscle has
two T tubules per sarcomere,
located at the A-I band junctions.
(Redrawn from Bloom W, Fawcett
DW: A Textbook of Histology.
Philadelphia: WB Saunders, 1986.
Modified after Peachey LD: J Cell
Biol 25:209, 1965. Drawn by Sylvia
Sarcotubules
Colard Keene.)
membrane, a potential change also spreads along the
sarcoplasm surrounding the myofibrils to cause con-
T tubules to the deep interior of the muscle fiber. The
traction, as discussed in Chapter 6.
electrical currents surrounding these T tubules then
elicit the muscle contraction.
Calcium Pump for Removing Calcium Ions from the Myofibrillar
Figure 7-5 also shows a sarcoplasmic reticulum, in
Fluid After Contraction Occurs. Once the calcium ions
yellow. This is composed of two major parts: (1) large
have been released from the sarcoplasmic tubules and
chambers called terminal cisternae that abut the T
have diffused among the myofibrils, muscle contrac-
tubules, and (2) long longitudinal tubules that sur-
tion continues as long as the calcium ions remain in
round all surfaces of the actual contracting myofibrils.
high concentration. However, a continually active
calcium pump located in the walls of the sarcoplasmic
reticulum pumps calcium ions away from the myofi-
Release of Calcium Ions by the
brils back into the sarcoplasmic tubules. This pump can
Sarcoplasmic Reticulum
concentrate the calcium ions about 10,000-fold inside
the tubules. In addition, inside the reticulum is a
One of the special features of the sarcoplasmic reticu-
protein called calsequestrin that can bind up to 40
lum is that within its vesicular tubules is an excess of
times more calcium.
calcium ions in high concentration, and many of these
ions are released from each vesicle when an action
Excitatory “Pulse” of Calcium Ions. The normal resting
potential occurs in the adjacent T tubule.
state concentration (less than 10-7 molar) of calcium
Figure 7-6 shows that the action potential of the T
ions in the cytosol that bathes the myofibrils is too
tubule causes current flow into the sarcoplasmic re-
little to elicit contraction. Therefore, the troponin-
ticular cisternae where they abut the T tubule. This in
tropomyosin complex keeps the actin filaments inhib-
turn causes rapid opening of large numbers of calcium
ited and maintains a relaxed state of the muscle.
channels through the membranes of the cisternae as
Conversely, full excitation of the T tubule and
well as their attached longitudinal tubules. These
sarcoplasmic reticulum system causes enough re-
channels remain open for a few milliseconds; during
lease of calcium ions to increase the concentration
this time, enough calcium ions are released into the
in the myofibrillar fluid to as high as 2
¥ 10-4 molar
Chapter 7
Excitation of Skeletal Muscle: Neuromuscular Transmission and Excitation-Contraction Coupling
91
Action potential
Sarcolemma
Calcium pump
Ca
Ca
ATP
required
Figure 7-6
Ca++
Ca++
Excitation-contraction coupling in
the muscle, showing (1) an action
potential that causes release of
calcium ions from the sarcoplas-
mic reticulum and then
(2) re-
uptake of the calcium ions by a
calcium pump.
Actin filaments
Myosin filaments
concentration, a 500-fold increase, which is about 10
neurophysiological basis and implication for respiratory
times the level required to cause maximum muscle
control. J Appl Physiol 96:407, 2004.
contraction. Immediately thereafter, the calcium pump
Hoch W: Molecular dissection of neuromuscular junction
formation. Trends Neurosci 26:335, 2003.
depletes the calcium ions again. The total duration of
Keesey JC: Clinical evaluation and management of myas-
this calcium “pulse” in the usual skeletal muscle fiber
thenia gravis. Muscle Nerve 29:484, 2004.
lasts about
1/20 of a second, although it may last
Lee C: Conformation, action, and mechanism of action of
several times as long in some fibers and several times
neuromuscular blocking muscle relaxants. Pharmacol Ther
less in others. (In heart muscle, the calcium pulse lasts
98:143, 2003.
about 1/3 of a second because of the long duration of
Leite JF, Rodrigues-Pinguet N, Lester HA: Insights into
the cardiac action potential.)
channel function via channel dysfunction. J Clin Invest
During this calcium pulse, muscle contraction
111:436, 2003.
occurs. If the contraction is to continue without inter-
Payne AM, Delbono O: Neurogenesis of excitation-
ruption for long intervals, a series of calcium pulses
contraction uncoupling in aging skeletal muscle. Exerc
Sport Sci Rev 32:36, 2004.
must be initiated by a continuous series of repetitive
Pette D: Historical perspectives: plasticity of mammalian
action potentials, as discussed in Chapter 6.
skeletal muscle. J Appl Physiol 90:1119, 2001.
Rekling JC, Funk GD, Bayliss DA, et al: Synaptic control of
motoneuronal excitability. Physiol Rev 80:767, 2000.
References
Schiaffino S, Serrano A: Calcineurin signaling and neural
control of skeletal muscle fiber type and size. Trends
Also see references for Chapters 5 and 6.
Pharmacol Sci 23:569, 2002.
Allman BL, Rice CL: Neuromuscular fatigue and aging:
Tang W, Sencer S, Hamilton SL: Calmodulin modulation of
central and peripheral factors. Muscle Nerve 25:785, 2002.
proteins involved in excitation-contraction coupling. Front
Amonof MJ: Electromyography in Clinical Practice. New
Biosci 7:583, 2002.
York: Churchill Livingstone, 1998.
Toyoshima C, Nomura H, Sugita Y: Structural basis of ion
Brown RH Jr: Dystrophin-associated proteins and the mus-
pumping by Ca2+-ATPase of sarcoplasmic reticulum.
cular dystrophies. Annu Rev Med 48:457, 1997.
FEBS Lett 555:106, 2003.
Chaudhuri A, Behan PO: Fatigue in neurological disorders.
Van der Kloot W, Molgo J: Quantal acetylcholine release at
Lancet 363:978, 2004.
the vertebrate neuromuscular junction. Physiol Rev
Engel AG, Ohno K, Shen XM, Sine SM: Congenital myas-
74:899, 1994.
thenic syndromes: multiple molecular targets at the
Vincent A: Unraveling the pathogenesis of myasthenia
neuromuscular junction. Ann N Y Acad Sci 998:138,
gravis. Nat Rev Immunol 10:797, 2002.
2003.
Vincent A, McConville J, Farrugia ME, et al: Antibodies in
Haouzi P, Chenuel B, Huszczuk A: Sensing vascular disten-
myasthenia gravis and related disorders. Ann N Y Acad
sion in skeletal muscle by slow conducting afferent fibers:
Sci 998:324, 2003.
C H A P T E R
8
Contraction and Excitation
of Smooth Muscle
Contraction of Smooth
Muscle
In Chapters 6 and 7, the discussion was concerned
with skeletal muscle. We now turn to smooth
muscle, which is composed of far smaller fibers—
usually 1 to 5 micrometers in diameter and only 20
to 500 micrometers in length. In contrast, skeletal
muscle fibers are as much as 30 times greater in diameter and hundreds of times
as long. Many of the same principles of contraction apply to smooth muscle as
to skeletal muscle. Most important, essentially the same attractive forces
between myosin and actin filaments cause contraction in smooth muscle as in
skeletal muscle, but the internal physical arrangement of smooth muscle fibers
is very different.
Types of Smooth Muscle
The smooth muscle of each organ is distinctive from that of most other organs
in several ways: (1) physical dimensions, (2) organization into bundles or sheets,
(3) response to different types of stimuli, (4) characteristics of innervation, and
(5) function. Yet, for the sake of simplicity, smooth muscle can generally be
divided into two major types, which are shown in Figure 8-1: multi-unit smooth
muscle and unitary (or single-unit) smooth muscle.
Multi-Unit Smooth Muscle. This type of smooth muscle is composed of discrete,
separate smooth muscle fibers. Each fiber operates independently of the others
and often is innervated by a single nerve ending, as occurs for skeletal muscle
fibers. Further, the outer surfaces of these fibers, like those of skeletal muscle
fibers, are covered by a thin layer of basement membrane-like substance, a
mixture of fine collagen and glycoprotein that helps insulate the separate fibers
from one another.
The most important characteristic of multi-unit smooth muscle fibers is that
each fiber can contract independently of the others, and their control is exerted
mainly by nerve signals. In contrast, a major share of control of unitary smooth
muscle is exerted by non-nervous stimuli. Some examples of multi-unit smooth
muscle are the ciliary muscle of the eye, the iris muscle of the eye, and the pilo-
erector muscles that cause erection of the hairs when stimulated by the sym-
pathetic nervous system.
Unitary Smooth Muscle. The term “unitary” is confusing because it does not mean
single muscle fibers. Instead, it means a mass of hundreds to thousands of
smooth muscle fibers that contract together as a single unit. The fibers usually
are arranged in sheets or bundles, and their cell membranes are adherent to one
another at multiple points so that force generated in one muscle fiber can be
transmitted to the next. In addition, the cell membranes are joined by many gap
junctions through which ions can flow freely from one muscle cell to the next
so that action potentials or simple ion flow without action potentials can travel
92
Chapter 8
Contraction and Excitation of Smooth Muscle
93
Adventitia
Actin
filaments
Dense bodies
Medial
muscle fibers
Endothelium
Small artery
Multi-unit smooth muscle
Unitary smooth muscle
Myosin filaments
A
B
Figure 8-1
Multi-unit (A) and unitary (B) smooth muscle.
from one fiber to the next and cause the muscle fibers
Cell membrane
to contract together. This type of smooth muscle is also
known as syncytial smooth muscle because of its syn-
cytial interconnections among fibers. It is also called
visceral smooth muscle because it is found in the walls
of most viscera of the body, including the gut, bile
ducts, ureters, uterus, and many blood vessels.
Contractile Mechanism in
Smooth Muscle
Chemical Basis for Smooth
Muscle Contraction
Smooth muscle contains both actin and myosin fila-
Figure 8-2
ments, having chemical characteristics similar to those
Physical structure of smooth muscle. The upper left-hand fiber
of the actin and myosin filaments in skeletal muscle.
shows actin filaments radiating from dense bodies. The lower left-
It does not contain the normal troponin complex
hand fiber and the right-hand diagram demonstrate the relation of
that is required in the control of skeletal muscle con-
myosin filaments to actin filaments.
traction, so the mechanism for control of contraction
is different. This is discussed in detail later in this
chapter.
Chemical studies have shown that actin and myosin
Physical Basis for Smooth Muscle Contraction
filaments derived from smooth muscle interact with
Smooth muscle does not have the same striated
each other in much the same way that they do in skele-
arrangement of actin and myosin filaments as is found
tal muscle. Further, the contractile process is activated
in skeletal muscle. Instead, electron micrographic tech-
by calcium ions, and adenosine triphosphate (ATP) is
niques suggest the physical organization exhibited in
degraded to adenosine diphosphate (ADP) to provide
Figure 8-2. This figure shows large numbers of actin
the energy for contraction.
filaments attached to so-called dense bodies. Some of
There are, however, major differences between the
these bodies are attached to the cell membrane.
physical organization of smooth muscle and that of
Others are dispersed inside the cell. Some of the mem-
skeletal muscle, as well as differences in excitation-
brane dense bodies of adjacent cells are bonded
contraction coupling, control of the contractile process
together by intercellular protein bridges. It is mainly
by calcium ions, duration of contraction, and amount
through these bonds that the force of contraction is
of energy required for contraction.
transmitted from one cell to the next.
94
Unit II
Membrane Physiology, Nerve, and Muscle
Interspersed among the actin filaments in the
This sparsity of energy utilization by smooth muscle
muscle fiber are myosin filaments. These have a diam-
is exceedingly important to the overall energy
eter more than twice that of the actin filaments. In
economy of the body, because organs such as the intes-
electron micrographs, one usually finds 5 to 10 times
tines, urinary bladder, gallbladder, and other viscera
as many actin filaments as myosin filaments.
often maintain tonic muscle contraction almost
To the right in Figure 8-2 is a postulated structure
indefinitely.
of an individual contractile unit within a smooth
muscle cell, showing large numbers of actin filaments
Slowness of Onset of Contraction and Relaxation of the Total
radiating from two dense bodies; the ends of these
Smooth Muscle Tissue. A typical smooth muscle tissue
filaments overlap a myosin filament located midway
begins to contract 50 to 100 milliseconds after it is
between the dense bodies. This contractile unit is
excited, reaches full contraction about 0.5 second later,
similar to the contractile unit of skeletal muscle, but
and then declines in contractile force in another 1 to
without the regularity of the skeletal muscle structure;
2 seconds, giving a total contraction time of 1 to 3
in fact, the dense bodies of smooth muscle serve the
seconds. This is about 30 times as long as a single
same role as the Z discs in skeletal muscle.
contraction of an average skeletal muscle fiber. But
There is another difference: Most of the myosin fila-
because there are so many types of smooth muscle,
ments have what are called “sidepolar” cross-bridges
contraction of some types can be as short as 0.2 second
arranged so that the bridges on one side hinge in one
or as long as 30 seconds.
direction and those on the other side hinge in the
The slow onset of contraction of smooth muscle,
opposite direction. This allows the myosin to pull an
as well as its prolonged contraction, is caused by
actin filament in one direction on one side while simul-
the slowness of attachment and detachment of the
taneously pulling another actin filament in the oppo-
cross-bridges with the actin filaments. In addition, the
site direction on the other side. The value of this
initiation of contraction in response to calcium ions is
organization is that it allows smooth muscle cells to
much slower than in skeletal muscle, as discussed later.
contract as much as 80 per cent of their length instead
of being limited to less than 30 per cent, as occurs in
Force of Muscle Contraction. Despite the relatively few
skeletal muscle.
myosin filaments in smooth muscle, and despite the
slow cycling time of the cross-bridges, the maximum
Comparison of Smooth Muscle Contraction
force of contraction of smooth muscle is often greater
and Skeletal Muscle Contraction
than that of skeletal muscle—as great as 4 to 6 kg/cm2
Although most skeletal muscles contract and relax
cross-sectional area for smooth muscle, in comparison
rapidly, most smooth muscle contraction is prolonged
with 3 to 4 kilograms for skeletal muscle. This great
tonic contraction, sometimes lasting hours or even
force of smooth muscle contraction results from the
days. Therefore, it is to be expected that both the physi-
prolonged period of attachment of the myosin cross-
cal and the chemical characteristics of smooth muscle
bridges to the actin filaments.
versus skeletal muscle contraction would differ.
Following are some of the differences.
“Latch” Mechanism for Prolonged Holding of Contractions of
Smooth Muscle. Once smooth muscle has developed full
Slow Cycling of the Myosin Cross-Bridges. The rapidity
contraction, the amount of continuing excitation
of cycling of the myosin cross-bridges in smooth
usually can be reduced to far less than the initial
muscle—that is, their attachment to actin, then release
level, yet the muscle maintains its full force of con-
from the actin, and reattachment for the next cycle—
traction. Further, the energy consumed to maintain
is much, much slower in smooth muscle than in skele-
contraction is often minuscule, sometimes as little as
tal muscle; in fact, the frequency is as little as 1/10 to
1/300 the energy required for comparable sustained
1/300 that in skeletal muscle. Yet the fraction of time
skeletal muscle contraction. This is called the “latch”
that the cross-bridges remain attached to the actin fila-
mechanism.
ments, which is a major factor that determines the
The importance of the latch mechanism is that it
force of contraction, is believed to be greatly increased
can maintain prolonged tonic contraction in smooth
in smooth muscle. A possible reason for the slow
muscle for hours with little use of energy. Little con-
cycling is that the cross-bridge heads have far less
tinued excitatory signal is required from nerve fibers
ATPase activity than in skeletal muscle, so that degra-
or hormonal sources.
dation of the ATP that energizes the movements
of the cross-bridge heads is greatly reduced, with
Stress-Relaxation of Smooth Muscle. Another impor-
corresponding slowing of the rate of cycling.
tant characteristic of smooth muscle, especially the vis-
ceral unitary type of smooth muscle of many hollow
Energy Required to Sustain Smooth Muscle Contraction. Only
organs, is its ability to return to nearly its original
1/10 to 1/300 as much energy is required to sustain the
force of contraction seconds or minutes after it has
same tension of contraction in smooth muscle as in
been elongated or shortened. For example, a sudden
skeletal muscle. This, too, is believed to result from the
increase in fluid volume in the urinary bladder, thus
slow attachment and detachment cycling of the cross-
stretching the smooth muscle in the bladder wall,
bridges and because only one molecule of ATP is
causes an immediate large increase in pressure in the
required for each cycle, regardless of its duration.
bladder. However, during the next 15 seconds to a
Chapter 8
Contraction and Excitation of Smooth Muscle
95
minute or so, despite continued stretch of the bladder
myosin phosphatase, located in the fluids of the
wall, the pressure returns almost exactly back to the
smooth muscle cell, which splits the phosphate from
original level. Then, when the volume is increased by
the regulatory light chain. Then the cycling stops and
another step, the same effect occurs again.
contraction ceases. The time required for relaxation of
Conversely, when the volume is suddenly decreased,
muscle contraction, therefore, is determined to a great
the pressure falls very low at first but then rises back
extent by the amount of active myosin phosphatase in
in another few seconds or minutes to or near to the
the cell.
original level. These phenomena are called stress-
relaxation and reverse stress-relaxation. Their impor-
Possible Mechanism for Regulation of the
tance is that, except for short periods of time, they
Latch Phenomenon
allow a hollow organ to maintain about the same
Because of the importance of the latch phenomenon
amount of pressure inside its lumen despite long-term,
in smooth muscle, and because this phenomenon
large changes in volume.
allows long-term maintenance of tone in many smooth
muscle organs without much expenditure of energy,
many attempts have been made to explain it. Among
Regulation of Contraction
the many mechanisms that have been postulated, one
by Calcium Ions
of the simplest is the following.
When the myosin kinase and myosin phosphatase
As is true for skeletal muscle, the initiating stimulus
enzymes are both strongly activated, the cycling fre-
for most smooth muscle contraction is an increase in
quency of the myosin heads and the velocity of
intracellular calcium ions. This increase can be caused
contraction are great. Then, as the activation of the
in different types of smooth muscle by nerve stimula-
enzymes decreases, the cycling frequency decreases,
tion of the smooth muscle fiber, hormonal stimulation,
but at the same time, the deactivation of these enzymes
stretch of the fiber, or even change in the chemical
allows the myosin heads to remain attached to the
environment of the fiber.
actin filament for a longer and longer proportion of
Yet smooth muscle does not contain troponin, the
the cycling period. Therefore, the number of heads
regulatory protein that is activated by calcium ions
attached to the actin filament at any given time
to cause skeletal muscle contraction. Instead, smooth
remains large. Because the number of heads attached
muscle contraction is activated by an entirely different
to the actin determines the static force of contraction,
mechanism, as follows.
tension is maintained, or “latched”; yet little energy
is used by the muscle, because ATP is not degraded
Combination of Calcium Ions with Calmodulin—Activation of
to ADP except on the rare occasion when a head
Myosin Kinase and Phosphorylation of the Myosin Head. In
detaches.
place of troponin, smooth muscle cells contain a large
amount of another regulatory protein called calmod-
ulin. Although this protein is similar to troponin, it is
Nervous and Hormonal Control
different in the manner in which it initiates contrac-
tion. Calmodulin does this by activating the myosin
of Smooth Muscle Contraction
cross-bridges. This activation and subsequent contrac-
tion occur in the following sequence:
Although skeletal muscle fibers are stimulated exclu-
1. The calcium ions bind with calmodulin.
sively by the nervous system, smooth muscle can be
2. The calmodulin-calcium combination joins with
stimulated to contract by multiple types of signals: by
and activates myosin kinase, a phosphorylating
nervous signals, by hormonal stimulation, by stretch of
enzyme.
the muscle, and in several other ways. The principal
3. One of the light chains of each myosin
reason for the difference is that the smooth muscle
head, called the regulatory chain, becomes
membrane contains many types of receptor proteins
phosphorylated in response to this myosin kinase.
that can initiate the contractile process. Still other
When this chain is not phosphorylated, the
receptor proteins inhibit smooth muscle contraction,
attachment-detachment cycling of the myosin
which is another difference from skeletal muscle.
head with the actin filament does not occur. But
Therefore, in this section, we discuss nervous control
when the regulatory chain is phosphorylated, the
of smooth muscle contraction, followed by hormonal
head has the capability of binding repetitively
control and other means of control.
with the actin filament and proceeding through
the entire cycling process of intermittent “pulls,”
the same as occurs for skeletal muscle, thus
Neuromuscular Junctions
causing muscle contraction.
of Smooth Muscle
Cessation of Contraction—Role of Myosin Phosphatase.
Physiologic Anatomy of Smooth Muscle Neuromuscular Junc-
When the calcium ion concentration falls below a crit-
tions. Neuromuscular junctions of the highly struc-
ical level, the aforementioned processes automatically
tured type found on skeletal muscle fibers do not occur
reverse, except for the phosphorylation of the myosin
in smooth muscle. Instead, the autonomic nerve fibers
head. Reversal of this requires another enzyme,
that innervate smooth muscle generally branch
96
Unit II
Membrane Physiology, Nerve, and Muscle
autonomic nerves innervating smooth muscle are
acetylcholine and norepinephrine, but they are never
secreted by the same nerve fibers. Acetylcholine is an
excitatory transmitter substance for smooth muscle
fibers in some organs but an inhibitory transmitter for
smooth muscle in other organs. When acetylcholine
excites a muscle fiber, norepinephrine ordinarily
inhibits it. Conversely, when acetylcholine inhibits a
fiber, norepinephrine usually excites it.
Varicosities
But why these different responses? The answer is
that both acetylcholine and norepinephrine excite or
inhibit smooth muscle by first binding with a receptor
protein on the surface of the muscle cell membrane.
Visceral
Multi-unit
Some of the receptor proteins are excitatory receptors,
whereas others are inhibitory receptors. Thus, the type
Figure 8-3
of receptor determines whether the smooth muscle is
inhibited or excited and also determines which of the
Innervation of smooth muscle.
two transmitters, acetylcholine or norepinephrine, is
effective in causing the excitation or inhibition. These
diffusely on top of a sheet of muscle fibers, as shown
receptors are discussed in more detail in Chapter 60 in
in Figure 8-3. In most instances, these fibers do not
relation to function of the autonomic nervous system.
make direct contact with the smooth muscle fiber cell
membranes but instead form so-called diffuse junc-
Membrane Potentials and Action
tions that secrete their transmitter substance into the
matrix coating of the smooth muscle often a few
Potentials in Smooth Muscle
nanometers to a few micrometers away from the
muscle cells; the transmitter substance then diffuses to
Membrane Potentials in Smooth Muscle. The quantitative
the cells. Furthermore, where there are many layers of
voltage of the membrane potential of smooth muscle
muscle cells, the nerve fibers often innervate only the
depends on the momentary condition of the muscle. In
outer layer, and muscle excitation travels from this
the normal resting state, the intracellular potential is
outer layer to the inner layers by action potential con-
usually about -50 to -60 millivolts, which is about 30
duction in the muscle mass or by additional diffusion
millivolts less negative than in skeletal muscle.
of the transmitter substance.
The axons that innervate smooth muscle fibers do
Action Potentials in Unitary Smooth Muscle. Action poten-
not have typical branching end feet of the type in the
tials occur in unitary smooth muscle (such as visceral
motor end plate on skeletal muscle fibers. Instead,
muscle) in the same way that they occur in skeletal
most of the fine terminal axons have multiple vari-
muscle. They do not normally occur in many, if not
cosities distributed along their axes. At these points the
most, multi-unit types of smooth muscle, as discussed
Schwann cells that envelop the axons are interrupted
in a subsequent section.
so that transmitter substance can be secreted through
The action potentials of visceral smooth muscle
the walls of the varicosities. In the varicosities are vesi-
occur in one of two forms: (1) spike potentials or
cles similar to those in the skeletal muscle end plate
(2) action potentials with plateaus.
that contain transmitter substance. But, in contrast to
the vesicles of skeletal muscle junctions, which always
Spike Potentials. Typical spike action potentials, such
contain acetylcholine, the vesicles of the autonomic
as those seen in skeletal muscle, occur in most types of
nerve fiber endings contain acetylcholine in some
unitary smooth muscle. The duration of this type of
fibers and norepinephrine in others—and occasionally
action potential is 10 to 50 milliseconds, as shown in
other substances as well.
Figure 8-4A. Such action potentials can be elicited in
In a few instances, particularly in the multi-unit type
many ways, for example, by electrical stimulation, by
of smooth muscle, the varicosities are separated from
the action of hormones on the smooth muscle, by the
the muscle cell membrane by as little as 20 to 30
action of transmitter substances from nerve fibers, by
nanometers—the same width as the synaptic cleft that
stretch, or as a result of spontaneous generation in the
occurs in the skeletal muscle junction. These are called
muscle fiber itself, as discussed subsequently.
contact junctions, and they function in much the same
way as the skeletal muscle neuromuscular junction; the
Action Potentials with Plateaus. Figure 8-4C shows a
rapidity of contraction of these smooth muscle fibers
smooth muscle action potential with a plateau. The
is considerably faster than that of fibers stimulated by
onset of this action potential is similar to that of the
the diffuse junctions.
typical spike potential. However, instead of rapid
repolarization of the muscle fiber membrane, the repo-
Excitatory and Inhibitory Transmitter Substances Secreted at
larization is delayed for several hundred to as much
the Smooth Muscle Neuromuscular Junction. The most
as 1000 milliseconds (1 second). The importance of
important transmitter substances secreted by the
the plateau is that it can account for the prolonged
Chapter 8
Contraction and Excitation of Smooth Muscle
97
ions act directly on the smooth muscle contractile
mechanism to cause contraction. Thus, the calcium
performs two tasks at once.
0
Slow Wave Potentials in Unitary Smooth Muscle, and Sponta-
neous Generation of Action Potentials. Some smooth
muscle is self-excitatory. That is, action potentials arise
+ 20
within the smooth muscle cells themselves without
an extrinsic stimulus. This often is associated with a
- 40
basic slow wave rhythm of the membrane potential.
A typical slow wave in a visceral smooth muscle of
the gut is shown in Figure 8-4B. The slow wave itself
- 60
Slow waves
is not the action potential. That is, it is not a self-
regenerative process that spreads progressively over
the membranes of the muscle fibers. Instead, it is a
0
50
100
0
10
20
30
local property of the smooth muscle fibers that make
A
Milliseconds
B
Seconds
up the muscle mass.
The cause of the slow wave rhythm is unknown. One
suggestion is that the slow waves are caused by waxing
0
and waning of the pumping of positive ions (presum-
ably sodium ions) outward through the muscle fiber
- 25
membrane; that is, the membrane potential becomes
more negative when sodium is pumped rapidly and
- 50
less negative when the sodium pump becomes less
active. Another suggestion is that the conductances of
0
0.1
0.2
0.3
0.4
the ion channels increase and decrease rhythmically.
C
Seconds
The importance of the slow waves is that, when they
are strong enough, they can initiate action potentials.
The slow waves themselves cannot cause muscle con-
Figure 8-4
traction, but when the peak of the negative slow wave
potential inside the cell membrane rises in the positive
A, Typical smooth muscle action potential (spike potential) elicited
direction from
-60 to about
-35 millivolts
(the
by an external stimulus. B, Repetitive spike potentials, elicited by
approximate threshold for eliciting action potentials in
slow rhythmical electrical waves that occur spontaneously in the
smooth muscle of the intestinal wall. C, Action potential with a
most visceral smooth muscle), an action potential
plateau, recorded from a smooth muscle fiber of the uterus.
develops and spreads over the muscle mass. Then con-
traction does occur. Figure 8-4B demonstrates this
effect, showing that at each peak of the slow wave, one
contraction that occurs in some types of smooth
or more action potentials occur. These repetitive
muscle, such as the ureter, the uterus under some con-
sequences of action potentials elicit rhythmical con-
ditions, and certain types of vascular smooth muscle.
traction of the smooth muscle mass. Therefore, the
(Also, this is the type of action potential seen in
slow waves are called pacemaker waves. In Chapter 62,
cardiac muscle fibers that have a prolonged period of
we see that this type of pacemaker activity controls the
contraction, as discussed in Chapters 9 and 10.)
rhythmical contractions of the gut.
Importance of Calcium Channels in Generating the
Excitation of Visceral Smooth Muscle by Muscle Stretch.
Smooth Muscle Action Potential. The smooth muscle
When visceral (unitary) smooth muscle is stretched
cell membrane has far more voltage-gated calcium
sufficiently, spontaneous action potentials usually
channels than does skeletal muscle but few voltage-
are generated. They result from a combination of (1)
gated sodium channels. Therefore, sodium participates
the normal slow wave potentials and (2) decrease in
little in the generation of the action potential in most
overall negativity of the membrane potential caused
smooth muscle. Instead, flow of calcium ions to the
by the stretch itself. This response to stretch allows the
interior of the fiber is mainly responsible for the action
gut wall, when excessively stretched, to contract auto-
potential. This occurs in the same self-regenerative
matically and rhythmically. For instance, when the gut
way as occurs for the sodium channels in nerve fibers
is overfilled by intestinal contents, local automatic con-
and in skeletal muscle fibers. However, the calcium
tractions often set up peristaltic waves that move the
channels open many times more slowly than do
contents away from the overfilled intestine, usually in
sodium channels, and they also remain open much
the direction of the anus.
longer. This accounts in large measure for the pro-
longed plateau action potentials of some smooth
Depolarization of Multi-Unit Smooth Muscle
muscle fibers.
Without Action Potentials
Another important feature of calcium ion entry into
The smooth muscle fibers of multi-unit smooth muscle
the cells during the action potential is that the calcium
(such as the muscle of the iris of the eye or the
98
Unit II
Membrane Physiology, Nerve, and Muscle
piloerector muscle of each hair) normally contract
angiotensin, endothelin, vasopressin, oxytocin, sero-
mainly in response to nerve stimuli. The nerve endings
tonin, and histamine.
secrete acetylcholine in the case of some multi-unit
A hormone causes contraction of a smooth muscle
smooth muscles and norepinephrine in the case of
when the muscle cell membrane contains hormone-
others. In both instances, the transmitter substances
gated excitatory receptors for the respective hormone.
cause depolarization of the smooth muscle membrane,
Conversely, the hormone causes inhibition if the mem-
and this in turn elicits contraction. Action potentials
brane contains inhibitory receptors for the hormone
usually do not develop; the reason is that the fibers are
rather than excitatory receptors.
too small to generate an action potential. (When
action potentials are elicited in visceral unitary smooth
Mechanisms of Smooth Muscle Excitation or Inhibition by Hor-
muscle, 30 to 40 smooth muscle fibers must depolarize
mones or Local Tissue Factors. Some hormone receptors
simultaneously before a self-propagating action poten-
in the smooth muscle membrane open sodium or
tial ensues.) Yet, in small smooth muscle cells, even
calcium ion channels and depolarize the membrane,
without an action potential, the local depolarization
the same as after nerve stimulation. Sometimes action
(called the junctional potential) caused by the nerve
potentials result, or action potentials that are already
transmitter substance itself spreads “electrotonically”
occurring may be enhanced. In other cases, depolari-
over the entire fiber and is all that is needed to cause
zation occurs without action potentials, and this depo-
muscle contraction.
larization allows calcium ion entry into the cell, which
promotes the contraction.
Inhibition, in contrast, occurs when the hormone (or
Effect of Local Tissue Factors and
other tissue factor) closes the sodium and calcium
Hormones to Cause Smooth Muscle
channels to prevent entry of these positive ions; inhi-
Contraction Without Action Potentials
bition also occurs if the normally closed potassium
channels are opened, allowing positive potassium ions
Probably half of all smooth muscle contraction is
to diffuse out of the cell. Both of these actions increase
initiated by stimulatory factors acting directly on
the degree of negativity inside the muscle cell, a
the smooth muscle contractile machinery and without
state called hyperpolarization, which strongly inhibits
action potentials. Two types of non-nervous and
muscle contraction.
non-action potential stimulating factors often
Sometimes smooth muscle contraction or inhibition
involved are (1) local tissue chemical factors and (2)
is initiated by hormones without directly causing any
various hormones.
change in the membrane potential. In these instances,
the hormone may activate a membrane receptor that
Smooth Muscle Contraction in Response to Local Tissue
does not open any ion channels but instead causes an
Chemical Factors. In Chapter 17, we discuss control of
internal change in the muscle fiber, such as release of
contraction of the arterioles, meta-arterioles, and pre-
calcium ions from the intracellular sarcoplasmic re-
capillary sphincters. The smallest of these vessels have
ticulum; the calcium then induces contraction. To
little or no nervous supply. Yet the smooth muscle is
inhibit contraction, other receptor mechanisms are
highly contractile, responding rapidly to changes in
known to activate the enzyme adenylate cyclase or
local chemical conditions in the surrounding intersti-
guanylate cyclase in the cell membrane; the portions of
tial fluid.
the receptors that protrude to the interior of the cells
In the normal resting state, many of these small
are coupled to these enzymes, causing the formation
blood vessels remain contracted. But when extra blood
of cyclic adenosine monophosphate (cAMP) or cyclic
flow to the tissue is needed, multiple factors can relax
guanosine monophosphate (cGMP), so-called second
the vessel wall, thus allowing for increased flow. In this
messengers. The cAMP or cGMP has many effects, one
way, a powerful local feedback control system controls
of which is to change the degree of phosphorylation of
the blood flow to the local tissue area. Some of the
several enzymes that indirectly inhibit contraction.
specific control factors are as follows:
The pump that moves calcium ions from the sar-
1. Lack of oxygen in the local tissues causes smooth
coplasm into the sarcoplasmic reticulum is activated,
muscle relaxation and, therefore, vasodilatation.
as well as the cell membrane pump that moves calcium
2. Excess carbon dioxide causes vasodilatation.
ions out of the cell itself; these effects reduce the
3. Increased hydrogen ion concentration causes
calcium ion concentration in the sarcoplasm, thereby
vasodilatation.
inhibiting contraction.
Adenosine, lactic acid, increased potassium ions,
Smooth muscles have considerable diversity in how
diminished calcium ion concentration, and increased
they initiate contraction or relaxation in response to
body temperature can all cause local vasodilatation.
different hormones, neurotransmitters, and other
substances. In some instances, the same substance
Effects of Hormones on Smooth Muscle Contraction. Most
may cause either relaxation or contraction of smooth
circulating hormones in the blood affect smooth
muscles in different locations. For example, norepi-
muscle contraction to some degree, and some have
nephrine inhibits contraction of smooth muscle in the
profound effects. Among the more important of
intestine but stimulates contraction of smooth muscle
these are norepinephrine, epinephrine, acetylcholine,
in blood vessels.
Chapter 8
Contraction and Excitation of Smooth Muscle
99
Source of Calcium Ions That Cause
Contraction (1) Through the Cell
Membrane and (2) from the
Sarcoplasmic Reticulum
Although the contractile process in smooth muscle,
as in skeletal muscle, is activated by calcium ions,
the source of the calcium ions differs; the difference
is that the sarcoplasmic reticulum, which provides
virtually all the calcium ions for skeletal muscle con-
traction, is only slightly developed in most smooth
Caveolae
muscle. Instead, almost all the calcium ions that
cause contraction enter the muscle cell from the extra-
cellular fluid at the time of the action potential or
other stimulus. That is, the concentration of calcium
ions in the extracellular fluid is greater than 10-3 molar,
Sarcoplasmic
in comparison with less than 10-7 molar inside the
reticulum
smooth muscle cell; this causes rapid diffusion of
the calcium ions into the cell from the extracellular
fluid when the calcium pores open. The time required
Figure 8-5
for this diffusion to occur averages 200 to 300 mil-
liseconds and is called the latent period before con-
Sarcoplasmic tubules in a large smooth muscle fiber showing their
traction begins. This latent period is about 50 times
relation to invaginations in the cell membrane called caveolae.
as great for smooth muscle as for skeletal muscle
contraction.
of the smooth muscle fiber back into the extra-
Role of the Smooth Muscle Sarcoplasmic Reticulum. Figure
cellular fluid, or into a sarcoplasmic reticulum, if it
8-5 shows a few slightly developed sarcoplasmic
is present. This pump is slow-acting in comparison
tubules that lie near the cell membrane in some larger
with the fast-acting sarcoplasmic reticulum pump in
smooth muscle cells. Small invaginations of the
skeletal muscle. Therefore, a single smooth muscle
cell membrane, called caveolae, abut the surfaces of
contraction often lasts for seconds rather than hun-
these tubules. The caveolae suggest a rudimentary
dredths to tenths of a second, as occurs for skeletal
analog of the transverse tubule system of skeletal
muscle.
muscle. When an action potential is transmitted into
the caveolae, this is believed to excite calcium ion
release from the abutting sarcoplasmic tubules in the
References
same way that action potentials in skeletal muscle
transverse tubules cause release of calcium ions from
Also see references for Chapters 5 and 6.
the skeletal muscle longitudinal sarcoplasmic tubules.
Blaustein MP, Lederer WJ: Sodium/calcium exchange:
In general, the more extensive the sarcoplasmic re-
its physiological implications. Physiol Rev 79:763, 1999.
ticulum in the smooth muscle fiber, the more rapidly
Davis MJ, Hill MA: Signaling mechanisms underlying
it contracts.
the vascular myogenic response. Physiol Rev
79:387,
1999.
Effect on Smooth Muscle Contraction Caused by Changing of
Harnett KM, Biancani P: Calcium-dependent and calcium-
independent contractions in smooth muscles. Am J Med
Extracellular Calcium Ion Concentration. Although chang-
115(Suppl 3A):24S, 2003.
ing the extracellular fluid calcium ion concentration
Horowitz A, Menice CB, Laporte R, Morgan KG: Mecha-
from normal has little effect on the force of contrac-
nisms of smooth muscle contraction. Physiol Rev 76:967,
tion of skeletal muscle, this is not true for most smooth
1996.
muscle. When the extracellular fluid calcium ion con-
Kamm KE, Stull JT: Regulation of smooth muscle contrac-
centration falls to about 1/3 to 1/10 normal, smooth
tile elements by second messengers. Annu Rev Physiol
muscle contraction usually ceases. Therefore, the
51:299, 1989.
force of contraction of smooth muscle usually is
Kuriyama H, Kitamura K, Itoh T, Inoue R: Physiological fea-
highly dependent on extracellular fluid calcium ion
tures of visceral smooth muscle cells, with special refer-
concentration.
ence to receptors and ion channels. Physiol Rev 78:811,
1998.
Lee CH, Poburko D, Kuo KH, et al: Ca2+ oscillations, gradi-
A Calcium Pump Is Required to Cause Smooth Muscle
ents, and homeostasis in vascular smooth muscle. Am J
Relaxation. To cause relaxation of smooth muscle after
Physiol Heart Circ Physiol 282:H1571, 2002.
it has contracted, the calcium ions must be removed
Rybalkin SD, Yan C, Bornfeldt KE, Beavo JA: Cyclic GMP
from the intracellular fluids. This removal is achieved
phosphodiesterases and regulation of smooth muscle
by a calcium pump that pumps calcium ions out
function. Circ Res 93:280, 2003.
100
Unit II
Membrane Physiology, Nerve, and Muscle
Somlyo AP, Somlyo AV: Ca2+ sensitivity of smooth muscle
Walker JS, Wingard CJ, Murphy RA: Energetics of cross-
and nonmuscle myosin II: modulated by G proteins,
bridge phosphorylation and contraction in vascular
kinases, and myosin phosphatase. Physiol Rev 83:1325,
smooth muscle. Hypertension 23:1106, 1994.
2003.
Webb RC: Smooth muscle contraction and relaxation. Adv
Stephens NL: Airway smooth muscle. Lung 179:333, 2001.
Physiol Educ 27:201, 2003.
U N I T III
The Heart
9. Heart Muscle; The Heart as a Pump and Function
of the Heart Valves
10. Rhythmical Excitation of the Heart
11. The Normal Electrocardiogram
12. Electrocardiographic Interpretation of Cardiac
Muscle and Coronary Blood Flow Abnormalities:
Vectorial Analysis
13. Cardiac Arrhythmias and Their
Electrocardiographic Interpretation
C H A P T E R
9
Heart Muscle; The Heart as
a Pump and Function of the
Heart Valves
With this chapter we begin discussion of the heart
and circulatory system. The heart, shown in Figure
9-1, is actually two separate pumps: a right heart
that pumps blood through the lungs, and a left heart
that pumps blood through the peripheral organs. In
turn, each of these hearts is a pulsatile two-chamber
pump composed of an atrium and a ventricle. Each
atrium is a weak primer pump for the ventricle,
helping to move blood into the ventricle. The ventricles then supply the main
pumping force that propels the blood either (1) through the pulmonary circu-
lation by the right ventricle or (2) through the peripheral circulation by the left
ventricle.
Special mechanisms in the heart cause a continuing succession of heart con-
tractions called cardiac rhythmicity, transmitting action potentials throughout
the heart muscle to cause the heart’s rhythmical beat. This rhythmical control
system is explained in Chapter 10. In this chapter, we explain how the heart
operates as a pump, beginning with the special features of heart muscle itself.
Physiology of Cardiac Muscle
The heart is composed of three major types of cardiac muscle: atrial muscle, ven-
tricular muscle, and specialized excitatory and conductive muscle fibers. The
atrial and ventricular types of muscle contract in much the same way as skele-
tal muscle, except that the duration of contraction is much longer. Conversely,
the specialized excitatory and conductive fibers contract only feebly because
they contain few contractile fibrils; instead, they exhibit either automatic rhyth-
mical electrical discharge in the form of action potentials or conduction of the
action potentials through the heart, providing an excitatory system that controls
the rhythmical beating of the heart.
Physiologic Anatomy of Cardiac Muscle
Figure 9-2 shows a typical histological picture of cardiac muscle, demonstrat-
ing cardiac muscle fibers arranged in a latticework, with the fibers dividing,
recombining, and then spreading again. One also notes immediately from this
figure that cardiac muscle is striated in the same manner as in typical skeletal
muscle. Further, cardiac muscle has typical myofibrils that contain actin and
myosin filaments almost identical to those found in skeletal muscle; these fila-
ments lie side by side and slide along one another during contraction in the
same manner as occurs in skeletal muscle (see Chapter 6). But in other ways,
cardiac muscle is quite different from skeletal muscle, as we shall see.
Cardiac Muscle as a Syncytium. The dark areas crossing the cardiac muscle fibers
in Figure 9-2 are called intercalated discs; they are actually cell membranes that
separate individual cardiac muscle cells from one another. That is, cardiac
muscle fibers are made up of many individual cells connected in series and in
parallel with one another.
103
104
Unit III
The Heart
HEAD AND UPPER EXTREMITY
Plateau
Aorta
+20
Pulmonary artery
0
- 20
Superior
vena cava
- 40
- 60
Lungs
- 80
Right atrium
-100
Purkinje fiber
Pulmonary
Plateau
Pulmonary
vein
valve
+20
Left atrium
0
Tricuspid
- 20
Mitral valve
valve
- 40
Aortic valve
- 60
Right ventricle
- 80
Inferior
Left
Ventricular muscle
-100
vena cava
ventricle
0
1
2
3
4
Seconds
TRUNK AND LOWER EXTREMITY
Figure 9-1
Figure 9-3
Structure of the heart, and course of blood flow through the heart
Rhythmical action potentials (in millivolts) from a Purkinje fiber
chambers and heart valves.
and from a ventricular muscle fiber, recorded by means of
microelectrodes.
The heart actually is composed of two syncytiums:
the atrial syncytium that constitutes the walls of the
two atria, and the ventricular syncytium that consti-
tutes the walls of the two ventricles. The atria are sepa-
rated from the ventricles by fibrous tissue that
surrounds the atrioventricular (A-V) valvular open-
ings between the atria and ventricles. Normally, poten-
tials are not conducted from the atrial syncytium into
the ventricular syncytium directly through this fibrous
tissue. Instead, they are conducted only by way of a
specialized conductive system called the A-V bundle,
a bundle of conductive fibers several millimeters in
diameter that is discussed in detail in Chapter 10.
This division of the muscle of the heart into two
Figure 9-2
functional syncytiums allows the atria to contract a
short time ahead of ventricular contraction, which is
“Syncytial,” interconnecting nature of cardiac muscle fibers.
important for effectiveness of heart pumping.
At each intercalated disc the cell membranes fuse
Action Potentials in Cardiac Muscle
with one another in such a way that they form perme-
able “communicating” junctions (gap junctions) that
The action potential recorded in a ventricular muscle
allow almost totally free diffusion of ions. Therefore,
fiber, shown in Figure 9-3, averages about 105 milli-
from a functional point of view, ions move with ease
volts, which means that the intracellular potential rises
in the intracellular fluid along the longitudinal axes of
from a very negative value, about
-85 millivolts,
the cardiac muscle fibers, so that action potentials
between beats to a slightly positive value, about +20
travel easily from one cardiac muscle cell to the next,
millivolts, during each beat. After the initial spike, the
past the intercalated discs. Thus, cardiac muscle is a
membrane remains depolarized for about 0.2 second,
syncytium of many heart muscle cells in which the
exhibiting a plateau as shown in the figure, followed at
cardiac cells are so interconnected that when one
the end of the plateau by abrupt repolarization. The
of these cells becomes excited, the action potential
presence of this plateau in the action potential causes
spreads to all of them, spreading from cell to cell
ventricular contraction to last as much as 15 times as
throughout the latticework interconnections.
long in cardiac muscle as in skeletal muscle.
Chapter 9
Heart Muscle; The Heart as a Pump and Function of the Heart Valves
105
What Causes the Long Action Potential and the Plateau? At
is this: Immediately after the onset of the action poten-
this point, we must ask the questions: Why is the action
tial, the permeability of the cardiac muscle membrane
potential of cardiac muscle so long, and why does it
for potassium ions decreases about fivefold, an effect
have a plateau, whereas that of skeletal muscle does
that does not occur in skeletal muscle. This decreased
not? The basic biophysical answers to these questions
potassium permeability may result from the excess
were presented in Chapter 5, but they merit summa-
calcium influx through the calcium channels just
rizing here as well.
noted. Regardless of the cause, the decreased potas-
At least two major differences between the mem-
sium permeability greatly decreases the outflux of
brane properties of cardiac and skeletal muscle
positively charged potassium ions during the action
account for the prolonged action potential and the
potential plateau and thereby prevents early return of
plateau in cardiac muscle. First, the action potential of
the action potential voltage to its resting level. When
skeletal muscle is caused almost entirely by sudden
the slow calcium-sodium channels do close at the end
opening of large numbers of so-called fast sodium
of 0.2 to 0.3 second and the influx of calcium and
channels that allow tremendous numbers of sodium
sodium ions ceases, the membrane permeability for
ions to enter the skeletal muscle fiber from the extra-
potassium ions also increases rapidly; this rapid loss of
cellular fluid. These channels are called “fast” channels
potassium from the fiber immediately returns the
because they remain open for only a few thousandths
membrane potential to its resting level, thus ending
of a second and then abruptly close. At the end of this
the action potential.
closure, repolarization occurs, and the action potential
is over within another thousandth of a second or so.
Velocity of Signal Conduction in Cardiac Muscle. The veloci-
In cardiac muscle, the action potential is caused by
ty of conduction of the excitatory action potential
opening of two types of channels: (1) the same fast
signal along both atrial and ventricular muscle fibers is
sodium channels as those in skeletal muscle and (2)
about 0.3 to 0.5 m/sec, or about 1/250 the velocity in very
another entirely different population of slow calcium
large nerve fibers and about 1/10 the velocity in skele-
channels, which are also called calcium-sodium
tal muscle fibers. The velocity of conduction in the
channels. This second population of channels differs
specialized heart conductive system—in the Purkinje
from the fast sodium channels in that they are slower
fibers—is as great as 4 m/sec in most parts of the
to open and, even more important, remain open
system, which allows reasonably rapid conduction of
for several tenths of a second. During this time, a large
the excitatory signal to the different parts of the heart,
quantity of both calcium and sodium ions flows
as explained in Chapter 10.
through these channels to the interior of the cardiac
muscle fiber, and this maintains a prolonged period of
Refractory Period of Cardiac Muscle. Cardiac muscle, like
depolarization, causing the plateau in the action poten-
all excitable tissue, is refractory to restimulation
tial. Further, the calcium ions that enter during this
during the action potential. Therefore, the refractory
plateau phase activate the muscle contractile process,
period of the heart is the interval of time, as shown to
while the calcium ions that cause skeletal muscle con-
the left in Figure 9-4, during which a normal cardiac
traction are derived from the intracellular sarcoplas-
impulse cannot re-excite an already excited area of
mic reticulum.
cardiac muscle. The normal refractory period of the
The second major functional difference between
ventricle is 0.25 to 0.30 second, which is about the
cardiac muscle and skeletal muscle that helps account
duration of the prolonged plateau action potential.
for both the prolonged action potential and its plateau
There is an additional relative refractory period of
Refractory period
Relative refractory
period
Later premature
contraction
Early premature
contraction
Figure 9-4
Force of ventricular heart muscle
contraction, showing also dura-
tion of the refractory period and
relative refractory period, plus the
0
1
2
3
effect of premature contraction.
Seconds
Note that premature contractions
do not cause wave summation, as
occurs in skeletal muscle.
106
Unit III
The Heart
about 0.05 second during which the muscle is more dif-
tubule system—that is, the availability of calcium ions
ficult than normal to excite but nevertheless can be
to cause cardiac muscle contraction—depends to a
excited by a very strong excitatory signal, as demon-
great extent on the extracellular fluid calcium ion
strated by the early “premature” contraction in the
concentration.
second example of Figure 9-4. The refractory period
(By way of contrast, the strength of skeletal muscle
of atrial muscle is much shorter than that for the ven-
contraction is hardly affected by moderate changes in
tricles (about 0.15 second for the atria compared with
extracellular fluid calcium concentration because
0.25 to 0.30 second for the ventricles).
skeletal muscle contraction is caused almost entirely
by calcium ions released from the sarcoplasmic retic-
Excitation-Contraction Coupling—Function
ulum inside the skeletal muscle fiber itself.)
of Calcium Ions and the Transverse Tubules
At the end of the plateau of the cardiac action
The term “excitation-contraction coupling” refers to
potential, the influx of calcium ions to the interior of
the mechanism by which the action potential causes
the muscle fiber is suddenly cut off, and the calcium
the myofibrils of muscle to contract. This was discussed
ions in the sarcoplasm are rapidly pumped back out of
for skeletal muscle in Chapter 7. Once again, there are
the muscle fibers into both the sarcoplasmic reticulum
differences in this mechanism in cardiac muscle that
and the T tubule-extracellular fluid space. As a result,
have important effects on the characteristics of cardiac
the contraction ceases until a new action potential
muscle contraction.
comes along.
As is true for skeletal muscle, when an action poten-
tial passes over the cardiac muscle membrane, the
Duration of Contraction. Cardiac muscle begins to contract
a few milliseconds after the action potential begins and
action potential spreads to the interior of the cardiac
continues to contract until a few milliseconds after the
muscle fiber along the membranes of the transverse
action potential ends. Therefore, the duration of con-
(T) tubules. The T tubule action potentials in turn act
traction of cardiac muscle is mainly a function of the
on the membranes of the longitudinal sarcoplasmic
duration of the action potential, including the plateau—
tubules to cause release of calcium ions into the
about 0.2 second in atrial muscle and 0.3 second in ven-
muscle sarcoplasm from the sarcoplasmic reticulum. In
tricular muscle.
another few thousandths of a second, these calcium
ions diffuse into the myofibrils and catalyze the chemi-
cal reactions that promote sliding of the actin and
The Cardiac Cycle
myosin filaments along one another; this produces the
muscle contraction.
The cardiac events that occur from the beginning of
Thus far, this mechanism of excitation-contraction
one heartbeat to the beginning of the next are called
coupling is the same as that for skeletal muscle, but
the cardiac cycle. Each cycle is initiated by sponta-
there is a second effect that is quite different. In addi-
neous generation of an action potential in the sinus
tion to the calcium ions that are released into the
node, as explained in Chapter 10. This node is located
sarcoplasm from the cisternae of the sarcoplasmic
in the superior lateral wall of the right atrium near the
reticulum, a large quantity of extra calcium ions also
opening of the superior vena cava, and the action
diffuses into the sarcoplasm from the T tubules them-
potential travels from here rapidly through both atria
selves at the time of the action potential. Indeed,
and then through the A-V bundle into the ventricles.
without this extra calcium from the T tubules, the
Because of this special arrangement of the conducting
strength of cardiac muscle contraction would be
system from the atria into the ventricles, there is a
reduced considerably because the sarcoplasmic re-
delay of more than 0.1 second during passage of the
ticulum of cardiac muscle is less well developed than
cardiac impulse from the atria into the ventricles. This
that of skeletal muscle and does not store enough
allows the atria to contract ahead of ventricular con-
calcium to provide full contraction. Conversely, the T
traction, thereby pumping blood into the ventricles
tubules of cardiac muscle have a diameter 5 times as
before the strong ventricular contraction begins. Thus,
great as that of the skeletal muscle tubules, which
the atria act as primer pumps for the ventricles, and
means a volume 25 times as great. Also, inside the T
the ventricles in turn provide the major source of
tubules is a large quantity of mucopolysaccharides that
power for moving blood through the body’s vascular
are electronegatively charged and bind an abundant
system.
store of calcium ions, keeping these always available
for diffusion to the interior of the cardiac muscle fiber
when a T tubule action potential appears.
Diastole and Systole
The strength of contraction of cardiac muscle
depends to a great extent on the concentration of
The cardiac cycle consists of a period of relaxation
calcium ions in the extracellular fluids. The reason for
called diastole, during which the heart fills with blood,
this is that the openings of the T tubules pass directly
followed by a period of contraction called systole.
through the cardiac muscle cell membrane into the
Figure 9-5 shows the different events during the
extracellular spaces surrounding the cells, allowing the
cardiac cycle for the left side of the heart. The top three
same extracellular fluid that is in the cardiac muscle
curves show the pressure changes in the aorta, left ven-
interstitium to percolate through the T tubules as well.
tricle, and left atrium, respectively. The fourth curve
Consequently, the quantity of calcium ions in the T
depicts the changes in left ventricular volume, the fifth
Chapter 9
Heart Muscle; The Heart as a Pump and Function of the Heart Valves
107
Isovolumic
relaxation
Rapid inflow
Ejection
Atrial systole
Isovolumic
Diastasis
contraction
Aortic valve
120
Aortic
closes
valve
100
opens
Aortic pressure
80
60
A-V valve
A-V valve
40
opens
closes
20
Atrial pressure
a
c
v
0
Ventricular pressure
130
Ventricular volume
90
R
50
P
T
Electrocardiogram
Q
S
1st
2nd
3rd
Phonocardiogram
Systole
Diastole
Systole
Figure 9-5
Events of the cardiac cycle for left ventricular function, showing changes in left atrial pressure, left ventricular pressure, aortic pressure,
ventricular volume, the electrocardiogram, and the phonocardiogram.
the electrocardiogram, and the sixth a phonocardio-
About 0.16 second after the onset of the P wave, the
gram, which is a recording of the sounds produced by
QRS waves appear as a result of electrical depolariza-
the heart—mainly by the heart valves—as it pumps. It
tion of the ventricles, which initiates contraction of the
is especially important that the reader study in detail
ventricles and causes the ventricular pressure to begin
this figure and understand the causes of all the events
rising, as also shown in the figure. Therefore, the QRS
shown.
complex begins slightly before the onset of ventricu-
lar systole.
Finally, one observes the ventricular T wave in the
electrocardiogram. This represents the stage of repo-
Relationship of the Electrocardiogram
larization of the ventricles when the ventricular muscle
to the Cardiac Cycle
fibers begin to relax. Therefore, the T wave occurs
slightly before the end of ventricular contraction.
The electrocardiogram in Figure 9-5 shows the P, Q,
R, S, and T waves, which are discussed in Chapters 11,
12, and 13. They are electrical voltages generated by
Function of the Atria as Primer Pumps
the heart and recorded by the electrocardiograph from
the surface of the body.
Blood normally flows continually from the great veins
The P wave is caused by spread of depolarization
into the atria; about 80 per cent of the blood flows
through the atria, and this is followed by atrial con-
directly through the atria into the ventricles even
traction, which causes a slight rise in the atrial pres-
before the atria contract. Then, atrial contraction
sure curve immediately after the electrocardiographic
usually causes an additional 20 per cent filling of the
P wave.
ventricles. Therefore, the atria simply function as
108
Unit III
The Heart
primer pumps that increase the ventricular pumping
causing the A-V valves to close. Then an additional
effectiveness as much as 20 per cent. However, the
0.02 to 0.03 second is required for the ventricle to build
heart can continue to operate under most conditions
up sufficient pressure to push the semilunar (aortic
even without this extra
20 per cent effectiveness
and pulmonary) valves open against the pressures in
because it normally has the capability of pumping 300
the aorta and pulmonary artery. Therefore, during this
to 400 per cent more blood than is required by the
period, contraction is occurring in the ventricles, but
resting body. Therefore, when the atria fail to function,
there is no emptying. This is called the period of iso-
the difference is unlikely to be noticed unless a person
volumic or isometric contraction, meaning that tension
exercises; then acute signs of heart failure occasionally
is increasing in the muscle but little or no shortening
develop, especially shortness of breath.
of the muscle fibers is occurring.
Pressure Changes in the Atria—The a, c, and v Waves. In the
atrial pressure curve of Figure 9-5, three minor pressure
Period of Ejection. When the left ventricular pressure
elevations, called the a, c, and v atrial pressure waves, are
rises slightly above 80 mm Hg (and the right ventricu-
noted.
lar pressure slightly above 8 mm Hg), the ventricular
The a wave is caused by atrial contraction. Ordinarily,
pressures push the semilunar valves open. Immedi-
the right atrial pressure increases 4 to 6 mm Hg during
ately, blood begins to pour out of the ventricles, with
atrial contraction, and the left atrial pressure increases
about 70 per cent of the blood emptying occurring
about 7 to 8 mm Hg.
during the first third of the period of ejection and the
The c wave occurs when the ventricles begin to
remaining 30 per cent emptying during the next two
contract; it is caused partly by slight backflow of blood
into the atria at the onset of ventricular contraction
thirds. Therefore, the first third is called the period of
but mainly by bulging of the A-V valves backward
rapid ejection, and the last two thirds, the period of
toward the atria because of increasing pressure in the
slow ejection.
ventricles.
The v wave occurs toward the end of ventricular
contraction; it results from slow flow of blood into the
Period of Isovolumic (Isometric) Relaxation. At the end of
atria from the veins while the A-V valves are closed
systole, ventricular relaxation begins suddenly, allow-
during ventricular contraction. Then, when ventricular
ing both the right and left intraventricular pressures
contraction is over, the A-V valves open, allowing this
to decrease rapidly. The elevated pressures in the dis-
stored atrial blood to flow rapidly into the ventricles and
tended large arteries that have just been filled with
causing the v wave to disappear.
blood from the contracted ventricles immediately
push blood back toward the ventricles, which snaps the
aortic and pulmonary valves closed. For another 0.03
Function of the Ventricles as Pumps
to 0.06 second, the ventricular muscle continues to
relax, even though the ventricular volume does not
Filling of the Ventricles. During ventricular systole, large
change, giving rise to the period of isovolumic or iso-
amounts of blood accumulate in the right and left atria
metric relaxation. During this period, the intraven-
because of the closed A-V valves. Therefore, as soon
tricular pressures decrease rapidly back to their low
as systole is over and the ventricular pressures fall
diastolic levels. Then the A-V valves open to begin a
again to their low diastolic values, the moderately
new cycle of ventricular pumping.
increased pressures that have developed in the atria
during ventricular systole immediately push the A-V
valves open and allow blood to flow rapidly into the
End-Diastolic Volume, End-Systolic Volume, and Stroke Volume
ventricles, as shown by the rise of the left ventricular
Output. During diastole, normal filling of the ventricles
volume curve in Figure 9-5. This is called the period of
increases the volume of each ventricle to about 110 to
rapid filling of the ventricles.
120 milliliters. This volume is called the end-diastolic
The period of rapid filling lasts for about the first
volume. Then, as the ventricles empty during systole,
third of diastole. During the middle third of diastole,
the volume decreases about 70 milliliters, which is
only a small amount of blood normally flows into the
called the stroke volume output. The remaining volume
ventricles; this is blood that continues to empty into
in each ventricle, about 40 to 50 milliliters, is called the
the atria from the veins and passes through the atria
end-systolic volume. The fraction of the end-diastolic
directly into the ventricles.
volume that is ejected is called the ejection fraction—
During the last third of diastole, the atria contract
usually equal to about 60 per cent.
and give an additional thrust to the inflow of blood
When the heart contracts strongly, the end-systolic
into the ventricles; this accounts for about 20 per cent
volume can be decreased to as little as 10 to 20 milli-
of the filling of the ventricles during each heart cycle.
liters. Conversely, when large amounts of blood flow
into the ventricles during diastole, the ventricular end-
Emptying of the Ventricles During Systole
diastolic volumes can become as great as 150 to 180
milliliters in the healthy heart. By both increasing the
Period of Isovolumic (Isometric) Contraction. Immedi-
end-diastolic volume and decreasing the end-systolic
ately after ventricular contraction begins, the ventric-
volume, the stroke volume output can be increased to
ular pressure rises abruptly, as shown in Figure 9-5,
more than double normal.
Chapter 9
Heart Muscle; The Heart as a Pump and Function of the Heart Valves
109
Function of the Valves
Aortic and Pulmonary Artery Valves. The aortic and pul-
monary artery semilunar valves function quite differ-
Atrioventricular Valves. The A-V valves
(the tricuspid
ently from the A-V valves. First, the high pressures in
and mitral valves) prevent backflow of blood from the
the arteries at the end of systole cause the semilunar
ventricles to the atria during systole, and the semilu-
valves to snap to the closed position, in contrast to the
nar valves (the aortic and pulmonary artery valves)
much softer closure of the A-V valves. Second, because
prevent backflow from the aorta and pulmonary ar-
of smaller openings, the velocity of blood ejection
teries into the ventricles during diastole. These valves,
through the aortic and pulmonary valves is far greater
shown in Figure 9-6 for the left ventricle, close and
than that through the much larger A-V valves. Also,
open passively. That is, they close when a backward
because of the rapid closure and rapid ejection, the
pressure gradient pushes blood backward, and they
edges of the aortic and pulmonary valves are subjected
open when a forward pressure gradient forces blood
to much greater mechanical abrasion than are the
in the forward direction. For anatomical reasons, the
A-V valves. Finally, the A-V valves are supported by
thin, filmy A-V valves require almost no backflow to
the chordae tendineae, which is not true for the semi-
cause closure, whereas the much heavier semilunar
lunar valves. It is obvious from the anatomy of the
valves require rather rapid backflow for a few
aortic and pulmonary valves (as shown for the aortic
milliseconds.
valve at the bottom of Figure 9-6) that they must be
constructed with an especially strong yet very pliable
Function of the Papillary Muscles. Figure 9-6 also
fibrous tissue base to withstand the extra physical
shows papillary muscles that attach to the vanes of the
stresses.
A-V valves by the chordae tendineae. The papillary
muscles contract when the ventricular walls contract,
Aortic Pressure Curve
but contrary to what might be expected, they do not
help the valves to close. Instead, they pull the vanes of
When the left ventricle contracts, the ventricular pres-
the valves inward toward the ventricles to prevent
sure increases rapidly until the aortic valve opens.
their bulging too far backward toward the atria during
Then, after the valve opens, the pressure in the ventri-
ventricular contraction. If a chorda tendinea becomes
cle rises much less rapidly, as shown in Figure 9-5,
ruptured or if one of the papillary muscles becomes
because blood immediately flows out of the ventricle
paralyzed, the valve bulges far backward during ven-
into the aorta and then into the systemic distribution
tricular contraction, sometimes so far that it leaks
arteries.
severely and results in severe or even lethal cardiac
The entry of blood into the arteries causes the walls
incapacity.
of these arteries to stretch and the pressure to increase
to about 120 mm Hg.
Next, at the end of systole, after the left ventricle
stops ejecting blood and the aortic valve closes, the
elastic walls of the arteries maintain a high pressure in
the arteries, even during diastole.
MITRAL VALVE
A so-called incisura occurs in the aortic pressure
Cusp
curve when the aortic valve closes. This is caused by a
short period of backward flow of blood immediately
before closure of the valve, followed by sudden cessa-
Chordae tendineae
tion of the backflow.
After the aortic valve has closed, the pressure in the
aorta decreases slowly throughout diastole because
Papillary muscles
the blood stored in the distended elastic arteries flows
continually through the peripheral vessels back to the
veins. Before the ventricle contracts again, the aortic
pressure usually has fallen to about 80 mm Hg (dias-
tolic pressure), which is two thirds the maximal pres-
sure of 120 mm Hg (systolic pressure) that occurs in
the aorta during ventricular contraction.
Cusp
The pressure curves in the right ventricle and pul-
monary artery are similar to those in the aorta, except
AORTIC VALVE
that the pressures are only about one sixth as great, as
discussed in Chapter 14.
Relationship of the Heart
Sounds to Heart Pumping
Figure 9-6
When listening to the heart with a stethoscope, one does
Mitral and aortic valves (the left ventricular valves).
not hear the opening of the valves because this is a
110
Unit III
The Heart
relatively slow process that normally makes no noise.
However, when the valves close, the vanes of the valves
and the surrounding fluids vibrate under the influence
300
Systolic pressure
of sudden pressure changes, giving off sound that travels
in all directions through the chest.
250
When the ventricles contract, one first hears a sound
caused by closure of the A-V valves. The vibration is low
200
Isovolumic
in pitch and relatively long-lasting and is known as the
relaxation
first heart sound. When the aortic and pulmonary valves
Period of ejection
close at the end of systole, one hears a rapid snap
150
because these valves close rapidly, and the surroundings
Isovolumic
vibrate for a short period. This sound is called the
III
100
contraction
second heart sound. The precise causes of the heart
EW
sounds are discussed more fully in Chapter 23, in rela-
50
IV
tion to listening to the sounds with the stethoscope.
II
Diastolic
I
pressure
0
0
50
100
150
200
250
Work Output of the Heart
Left ventricular volume (ml)
Period of filling
The stroke work output of the heart is the amount of
energy that the heart converts to work during each
heartbeat while pumping blood into the arteries. Minute
work output is the total amount of energy converted to
Figure 9-7
work in 1 minute; this is equal to the stroke work output
Relationship between left ventricular volume and intraventricular
times the heart rate per minute.
pressure during diastole and systole. Also shown by the heavy
Work output of the heart is in two forms. First, by far
red lines is the
“volume-pressure diagram,” demonstrating
the major proportion is used to move the blood from
changes in intraventricular volume and pressure during the
the low-pressure veins to the high-pressure arteries.
normal cardiac cycle. EW, net external work.
This is called volume-pressure work or external work.
Second, a minor proportion of the energy is used to
accelerate the blood to its velocity of ejection through
the aortic and pulmonary valves. This is the kinetic
Until the volume of the noncontracting ventricle rises
energy of blood flow component of the work output.
above about 150 milliliters, the “diastolic” pressure
Right ventricular external work output is normally
does not increase greatly. Therefore, up to this volume,
about one sixth the work output of the left ventricle
blood can flow easily into the ventricle from the atrium.
because of the sixfold difference in systolic pressures
Above 150 milliliters, the ventricular diastolic pressure
that the two ventricles pump. The additional work
increases rapidly, partly because of fibrous tissue in the
output of each ventricle required to create kinetic
heart that will stretch no more and partly because the
energy of blood flow is proportional to the mass of
pericardium that surrounds the heart becomes filled
blood ejected times the square of velocity of ejection.
nearly to its limit.
Ordinarily, the work output of the left ventricle
During ventricular contraction, the “systolic” pres-
required to create kinetic energy of blood flow is only
sure increases even at low ventricular volumes and
about 1 per cent of the total work output of the ventri-
reaches a maximum at a ventricular volume of 150 to
cle and therefore is ignored in the calculation of the
170 milliliters. Then, as the volume increases still
total stroke work output. But in certain abnormal con-
further, the systolic pressure actually decreases under
ditions, such as aortic stenosis, in which blood flows with
some conditions, as demonstrated by the falling systolic
great velocity through the stenosed valve, more than
pressure curve in Figure 9-7, because at these great
50 per cent of the total work output may be required to
volumes, the actin and myosin filaments of the cardiac
create kinetic energy of blood flow.
muscle fibers are pulled apart far enough that the
strength of each cardiac fiber contraction becomes less
than optimal.
Graphical Analysis of Ventricular
Note especially in the figure that the maximum sys-
Pumping
tolic pressure for the normal left ventricle is between
250 and 300 mm Hg, but this varies widely with each
Figure 9-7 shows a diagram that is especially useful in
person’s heart strength and degree of heart stimulation
explaining the pumping mechanics of the left ventricle.
by cardiac nerves. For the normal right ventricle,
The most important components of the diagram are
the maximum systolic pressure is between
60 and
the two curves labeled
“diastolic pressure” and
80 mm Hg.
“systolic pressure.” These curves are volume-pressure
curves.
“Volume-Pressure Diagram” During the Cardiac Cycle; Cardiac
The diastolic pressure curve is determined by filling
Work Output. The red lines in Figure 9-7 form a loop
the heart with progressively greater volumes of blood
called the volume-pressure diagram of the cardiac cycle
and then measuring the diastolic pressure immediately
for normal function of the left ventricle. It is divided into
before ventricular contraction occurs, which is the end-
four phases.
diastolic pressure of the ventricle.
Phase I: Period of filling. This phase in the volume-
The systolic pressure curve is determined by record-
pressure diagram begins at a ventricular volume of
ing the systolic pressure achieved during ventricular
about 45 milliliters and a diastolic pressure near
contraction at each volume of filling.
0 mm Hg. Forty-five milliliters is the amount of
Chapter 9
Heart Muscle; The Heart as a Pump and Function of the Heart Valves
111
blood that remains in the ventricle after the
The importance of the concepts of preload and after-
previous heartbeat and is called the end-systolic
load is that in many abnormal functional states of the
volume. As venous blood flows into the ventricle
heart or circulation, the pressure during filling of the
from the left atrium, the ventricular volume
ventricle
(the preload), the arterial pressure against
normally increases to about 115 milliliters, called
which the ventricle must contract (the afterload), or
the end-diastolic volume, an increase of 70
both are severely altered from normal.
milliliters. Therefore, the volume-pressure diagram
during phase I extends along the line labeled “I,”
with the volume increasing to 115 milliliters and the
Chemical Energy Required
diastolic pressure rising to about 5 mm Hg.
for Cardiac Contraction:
Phase II: Period of isovolumic contraction. During
Oxygen Utilization by
isovolumic contraction, the volume of the ventricle
does not change because all valves are closed.
the Heart
However, the pressure inside the ventricle increases
Heart muscle, like skeletal muscle, uses chemical energy
to equal the pressure in the aorta, at a pressure
to provide the work of contraction. This energy is
value of about 80 mm Hg, as depicted by the arrow
derived mainly from oxidative metabolism of fatty acids
end of the line labeled “II.”
and, to a lesser extent, of other nutrients, especially
Phase III: Period of ejection. During ejection, the
lactate and glucose. Therefore, the rate of oxygen con-
systolic pressure rises even higher because of still
sumption by the heart is an excellent measure of the
more contraction of the ventricle. At the same time,
chemical energy liberated while the heart performs its
the volume of the ventricle decreases because the
work. The different chemical reactions that liberate this
aortic valve has now opened and blood flows out of
energy are discussed in Chapters 67 and 68.
the ventricle into the aorta. Therefore, the curve
labeled “III” traces the changes in volume and
Efficiency of Cardiac Contraction. During heart muscle con-
systolic pressure during this period of ejection.
traction, most of the expended chemical energy is con-
Phase IV: Period of isovolumic relaxation. At the end
verted into heat and a much smaller portion into work
of the period of ejection, the aortic valve closes, and
output. The ratio of work output to total chemical
the ventricular pressure falls back to the diastolic
energy expenditure is called the efficiency of cardiac
pressure level. The line labeled “IV” traces this
contraction, or simply efficiency of the heart. Maximum
decrease in intraventricular pressure without any
efficiency of the normal heart is between 20 and 25 per
change in volume. Thus, the ventricle returns to its
cent. In heart failure, this can decrease to as low as 5 to
starting point, with about 45 milliliters of blood
10 per cent.
left in the ventricle and at an atrial pressure near
0 mm Hg.
Readers well trained in the basic principles of physics
should recognize that the area subtended by this func-
Regulation of Heart Pumping
tional volume-pressure diagram (the tan shaded area,
labeled EW) represents the net external work output of
When a person is at rest, the heart pumps only 4 to 6
the ventricle during its contraction cycle. In experimen-
liters of blood each minute. During severe exercise,
tal studies of cardiac contraction, this diagram is used
the heart may be required to pump four to seven times
for calculating cardiac work output.
this amount. The basic means by which the volume
When the heart pumps large quantities of blood, the
pumped by the heart is regulated are (1) intrinsic
area of the work diagram becomes much larger. That is,
cardiac regulation of pumping in response to changes
it extends far to the right because the ventricle fills with
in volume of blood flowing into the heart and (2)
more blood during diastole, it rises much higher because
control of heart rate and strength of heart pumping by
the ventricle contracts with greater pressure, and it
usually extends farther to the left because the ventricle
the autonomic nervous system.
contracts to a smaller volume—especially if the ventri-
cle is stimulated to increased activity by the sympathetic
nervous system.
Intrinsic Regulation of Heart
Pumping—The Frank-Starling
Mechanism
Concepts of Preload and Afterload. In assessing the contrac-
tile properties of muscle, it is important to specify the
In Chapter 20, we will learn that under most condi-
degree of tension on the muscle when it begins to con-
tions, the amount of blood pumped by the heart each
tract, which is called the preload, and to specify the load
minute is determined almost entirely by the rate of
against which the muscle exerts its contractile force,
blood flow into the heart from the veins, which is called
which is called the afterload.
venous return. That is, each peripheral tissue of the
For cardiac contraction, the preload is usually con-
body controls its own local blood flow, and all the local
sidered to be the end-diastolic pressure when the ven-
tissue flows combine and return by way of the veins to
tricle has become filled.
the right atrium. The heart, in turn, automatically
The afterload of the ventricle is the pressure in the
pumps this incoming blood into the arteries, so that it
artery leading from the ventricle. In Figure 9-7, this cor-
can flow around the circuit again.
responds to the systolic pressure described by the phase
III curve of the volume-pressure diagram. (Sometimes
This intrinsic ability of the heart to adapt to increas-
the afterload is loosely considered to be the resistance
ing volumes of inflowing blood is called the Frank-
in the circulation rather than the pressure.)
Starling mechanism of the heart, in honor of Frank and
112
Unit III
The Heart
Starling, two great physiologists of a century ago. Basi-
work output for that side increases until it reaches the
cally, the Frank-Starling mechanism means that the
limit of the ventricle’s pumping ability.
greater the heart muscle is stretched during filling,
Figure 9-9 shows another type of ventricular func-
the greater is the force of contraction and the greater
tion curve called the ventricular volume output curve.
the quantity of blood pumped into the aorta. Or, stated
The two curves of this figure represent function of the
another way: Within physiologic limits, the heart pumps
two ventricles of the human heart based on data
all the blood that returns to it by the way of the veins.
extrapolated from lower animals. As the right and left
atrial pressures increase, the respective ventricular
What Is the Explanation of the Frank-Starling Mechanism?
volume outputs per minute also increase.
When an extra amount of blood flows into the ventri-
Thus, ventricular function curves are another way of
cles, the cardiac muscle itself is stretched to greater
expressing the Frank-Starling mechanism of the heart.
length. This in turn causes the muscle to contract with
That is, as the ventricles fill in response to higher atrial
increased force because the actin and myosin filaments
pressures, each ventricular volume and strength of
are brought to a more nearly optimal degree of
cardiac muscle contraction increase, causing the
overlap for force generation. Therefore, the ventricle,
heart to pump increased quantities of blood into the
because of its increased pumping, automatically
arteries.
pumps the extra blood into the arteries.
This ability of stretched muscle, up to an optimal
Control of the Heart by the Sympathetic and
length, to contract with increased work output is char-
Parasympathetic Nerves
acteristic of all striated muscle, as explained in Chapter
The pumping effectiveness of the heart also is con-
6, and is not simply a characteristic of cardiac muscle.
trolled by the sympathetic and parasympathetic
In addition to the important effect of lengthening
(vagus) nerves, which abundantly supply the heart, as
the heart muscle, still another factor increases heart
shown in Figure 9-10. For given levels of input atrial
pumping when its volume is increased. Stretch of the
pressure, the amount of blood pumped each minute
right atrial wall directly increases the heart rate by
(cardiac output) often can be increased more than 100
10 to 20 per cent; this, too, helps increase the amount
per cent by sympathetic stimulation. By contrast, the
of blood pumped each minute, although its contribu-
output can be decreased to as low as zero or almost
tion is much less than that of the Frank-Starling
zero by vagal (parasympathetic) stimulation.
mechanism.
Mechanisms of Excitation of the Heart by the Sympathetic
Ventricular Function Curves
Nerves. Strong sympathetic stimulation can increase
One of the best ways to express the functional ability
the heart rate in young adult humans from the normal
of the ventricles to pump blood is by ventricular func-
rate of 70 beats per minute up to 180 to 200 and, rarely,
tion curves, as shown in Figures 9-8 and 9-9. Figure
even 250 beats per minute. Also, sympathetic stimula-
9-8 shows a type of ventricular function curve called
tion increases the force of heart contraction to as much
the stroke work output curve. Note that as the atrial
as double normal, thereby increasing the volume of
pressure for each side of the heart increases, the stroke
blood pumped and increasing the ejection pressure.
Left ventricular
Right ventricular
stroke work
stroke work
(gram meters)
(gram meters)
15
Right ventricle
40
4
30
3
10
Left ventricle
20
2
10
1
5
0
0
0
10
20
0
10
20
Left mean atrial
Right mean atrial
pressure
pressure
0
(mm Hg)
(mm Hg)
-4
0
+4
+8
+12
+16
Atrial pressure (mm Hg)
Figure 9-8
Left and right ventricular function curves recorded from dogs,
Figure 9-9
depicting ventricular stroke work output as a function of left and
right mean atrial pressures. (Curves reconstructed from data in
Approximate normal right and left ventricular volume output
Sarnoff SJ: Myocardial contractility as described by ventricular
curves for the normal resting human heart as extrapolated from
function curves. Physiol Rev 35:107, 1955.)
data obtained in dogs and data from human beings.
Chapter 9
Heart Muscle; The Heart as a Pump and Function of the Heart Valves
113
Sympathetic chains
Maximum sympathetic
Vagi
25
stimulation
20
S-A
A-V
node
node
Normal sympathetic
15
stimulation
10
Zero sympathetic
stimulation
(Parasympathetic
Sympathetic
Sympathetic
stimulation)
nerves
5
nerves
Figure 9-10
0
Cardiac sympathetic and parasympathetic nerves. (The vagus
-4
0
+4
+8
nerves to the heart are parasympathetic nerves.)
Right atrial pressure (mm Hg)
Thus, sympathetic stimulation often can increase the
Figure 9-11
maximum cardiac output as much as twofold to three-
Effect on the cardiac output curve of different degrees of sympa-
fold, in addition to the increased output caused by the
thetic or parasympathetic stimulation.
Frank-Starling mechanism already discussed.
Conversely, inhibition of the sympathetic nerves to
the heart can decrease cardiac pumping to a moderate
extent in the following way: Under normal conditions,
function curves. They are similar to the ventricular
the sympathetic nerve fibers to the heart discharge
function curves of Figure 9-9. However, they represent
continuously at a slow rate that maintains pumping at
function of the entire heart rather than of a single ven-
about 30 per cent above that with no sympathetic stimu-
tricle; they show the relation between right atrial pres-
lation. Therefore, when the activity of the sympathetic
sure at the input of the right heart and cardiac output
nervous system is depressed below normal, this
from the left ventricle into the aorta.
decreases both heart rate and strength of ventricular
The curves of Figure 9-11 demonstrate that at any
muscle contraction, thereby decreasing the level of
given right atrial pressure, the cardiac output increases
cardiac pumping as much as 30 per cent below normal.
during increased sympathetic stimulation and
decreases during increased parasympathetic stimula-
Parasympathetic
(Vagal) Stimulation of the Heart. Strong
tion. These changes in output caused by nerve stimu-
stimulation of the parasympathetic nerve fibers in the
lation result both from changes in heart rate and from
vagus nerves to the heart can stop the heartbeat for a
changes in contractile strength of the heart because
few seconds, but then the heart usually “escapes” and
both change in response to the nerve stimulation.
beats at a rate of 20 to 40 beats per minute as long
as the parasympathetic stimulation continues. In
addition, strong vagal stimulation can decrease the
Effect of Potassium and Calcium Ions
strength of heart muscle contraction by
20 to
30
on Heart Function
per cent.
The vagal fibers are distributed mainly to the atria
In the discussion of membrane potentials in Chapter
and not much to the ventricles, where the power con-
5, it was pointed out that potassium ions have a
traction of the heart occurs. This explains the effect of
marked effect on membrane potentials, and in Chapter
vagal stimulation mainly to decrease heart rate rather
6 it was noted that calcium ions play an especially
than to decrease greatly the strength of heart contrac-
important role in activating the muscle contractile
tion. Nevertheless, the great decrease in heart rate
process. Therefore, it is to be expected that the con-
combined with a slight decrease in heart contraction
centration of each of these two ions in the extracellu-
strength can decrease ventricular pumping 50 per cent
lar fluids should also have important effects on cardiac
or more.
pumping.
Effect of Sympathetic or Parasympathetic Stimulation on the
Effect of Potassium Ions. Excess potassium in the extra-
Cardiac Function Curve. Figure 9-11 shows four cardiac
cellular fluids causes the heart to become dilated and
114
Unit III
The Heart
flaccid and also slows the heart rate. Large quantities
also can block conduction of the cardiac impulse from
the atria to the ventricles through the A-V bundle.
Normal range
Elevation of potassium concentration to only 8 to
5
12 mEq/L—two to three times the normal value—can
cause such weakness of the heart and abnormal
4
rhythm that this can cause death.
These effects result partially from the fact that a
3
high potassium concentration in the extracellular
2
fluids decreases the resting membrane potential in
the cardiac muscle fibers, as explained in Chapter 5.
1
As the membrane potential decreases, the intensity of
0
the action potential also decreases, which makes con-
0
50
100
150
200
250
traction of the heart progressively weaker.
Arterial pressure (mm Hg)
Effect of Calcium Ions. An excess of calcium ions causes
effects almost exactly opposite to those of potassium
Figure 9-12
ions, causing the heart to go toward spastic contrac-
tion. This is caused by a direct effect of calcium ions to
Constancy of cardiac output up to a pressure level of 160 mm Hg.
initiate the cardiac contractile process, as explained
Only when the arterial pressure rises above this normal limit does
earlier in the chapter.
the increasing pressure load cause the cardiac output to fall
Conversely, deficiency of calcium ions causes cardiac
significantly.
flaccidity, similar to the effect of high potassium.
Fortunately, however, calcium ion levels in the blood
normally are regulated within a very narrow range.
the heart at normal systolic arterial pressures (80 to
Therefore, cardiac effects of abnormal calcium con-
140 mm Hg), the cardiac output is determined almost
centrations are seldom of clinical concern.
entirely by the ease of blood flow through the body’s
tissues, which in turn controls venous return of
blood to the heart. This is the principal subject of
Effect of Temperature on
Chapter 20.
Heart Function
Increased body temperature, as occurs when one has
References
fever, causes a greatly increased heart rate, sometimes
to as fast as double normal. Decreased temperature
Bers DM: Cardiac excitation-contraction coupling. Nature
causes a greatly decreased heart rate, falling to as low
415:198, 2002.
as a few beats per minute when a person is near death
Brette F, Orchard C: T-tubule function in mammalian cardiac
from hypothermia in the body temperature range of
myocytes. Circ Res 92:1182, 2003.
60° to
70°F. These effects presumably result from
Brutsaert DL: Cardiac endothelial-myocardial signaling: its
role in cardiac growth, contractile performance, and rhyth-
the fact that heat increases the permeability of the
micity. Physiol Rev 83:59, 2003.
cardiac muscle membrane to ions that control heart
Clancy CE, Kass RS: Defective cardiac ion channels: from
rate, resulting in acceleration of the self-excitation
mutations to clinical syndromes. J Clin Invest 110:1075,
process.
2002.
Contractile strength of the heart often is enhanced
Fozzard HA: Cardiac sodium and calcium channels: a history
temporarily by a moderate increase in temperature, as
of excitatory currents. Cardiovasc Res 55:1, 2002.
occurs during body exercise, but prolonged elevation
Fuchs F, Smith SH: Calcium, cross-bridges, and the Frank-
of temperature exhausts the metabolic systems of the
Starling relationship. News Physiol Sci 16:5, 2001.
heart and eventually causes weakness. Therefore,
Guyton AC: Determination of cardiac output by equating
optimal function of the heart depends greatly on
venous return curves with cardiac response curves. Physiol
Rev 35:123, 1955.
proper control of body temperature by the tempera-
Guyton AC, Jones CE, Coleman TG: Circulatory Physiology:
ture control mechanisms explained in Chapter 73.
Cardiac Output and Its Regulation, 2nd ed. Philadelphia:
WB Saunders, 1973.
Herring N, Danson EJ, Paterson DJ: Cholinergic control of
Increasing the Arterial Pressure Load
heart rate by nitric oxide is site specific. News Physiol Sci
(up to a Limit) Does Not Decrease the
17:202, 2002.
Korzick DH: Regulation of cardiac excitation-contraction
Cardiac Output
coupling: a cellular update. Adv Physiol Educ 27:192, 2003.
Olson EN: A decade of discoveries in cardiac biology. Nat
Note in Figure 9-12 that increasing the arterial pres-
Med 10:467, 2004.
sure in the aorta does not decrease the cardiac output
Page E, Fozzard HA, Solaro JR: Handbook of Physiology,
until the mean arterial pressure rises above about
sec 2: The Cardiovascular System, vol 1: The Heart. New
160 mm Hg. In other words, during normal function of
York: Oxford University Press, 2002.
Chapter 9
Heart Muscle; The Heart as a Pump and Function of the Heart Valves
115
Rudy Y: From genome to physiome: integrative models of
Sussman MA, Anversa P: Myocardial aging and senescence:
cardiac excitation. Ann Biomed Eng 28:945, 2000.
where have the stem cells gone? Annu Rev Physiol 66:29,
Sarnoff SJ: Myocardial contractility as described by ven-
2004.
tricular function curves. Physiol Rev 35:107, 1955.
Zucker IH, Schultz HD, Li YF, et al: The origin of sympa-
Starling EH: The Linacre Lecture on the Law of the Heart.
thetic outflow in heart failure: the roles of angiotensin II
London: Longmans Green, 1918.
and nitric oxide. Prog Biophys Mol Biol 84:217, 2004.
C H A P T E R
1
0
Rhythmical Excitation
of the Heart
The heart is endowed with a special system for (1)
generating rhythmical electrical impulses to cause
rhythmical contraction of the heart muscle and (2)
conducting these impulses rapidly through the
heart. When this system functions normally, the atria
contract about one sixth of a second ahead of ven-
tricular contraction, which allows filling of the ven-
tricles before they pump the blood through the
lungs and peripheral circulation. Another special importance of the system is
that it allows all portions of the ventricles to contract almost simultaneously,
which is essential for most effective pressure generation in the ventricular
chambers.
This rhythmical and conductive system of the heart is susceptible to damage
by heart disease, especially by ischemia of the heart tissues resulting from poor
coronary blood flow. The result is often a bizarre heart rhythm or abnormal
sequence of contraction of the heart chambers, and the pumping effectiveness
of the heart often is affected severely, even to the extent of causing death.
Specialized Excitatory and Conductive System
of the Heart
Figure 10-1 shows the specialized excitatory and conductive system of the heart
that controls cardiac contractions. The figure shows the sinus node (also called
sinoatrial or S-A node), in which the normal rhythmical impulse is generated;
the internodal pathways that conduct the impulse from the sinus node to the
atrioventricular (A-V) node; the A-V node, in which the impulse from the atria
is delayed before passing into the ventricles; the A-V bundle, which conducts
the impulse from the atria into the ventricles; and the left and right bundle
branches of Purkinje fibers, which conduct the cardiac impulse to all parts of
the ventricles.
Sinus (Sinoatrial) Node
The sinus node (also called sinoatrial node) is a small, flattened, ellipsoid strip
of specialized cardiac muscle about 3 millimeters wide, 15 millimeters long, and
1 millimeter thick. It is located in the superior posterolateral wall of the right
atrium immediately below and slightly lateral to the opening of the superior
vena cava. The fibers of this node have almost no contractile muscle filaments
and are each only 3 to 5 micrometers in diameter, in contrast to a diameter of
10 to 15 micrometers for the surrounding atrial muscle fibers. However, the
sinus nodal fibers connect directly with the atrial muscle fibers, so that any
action potential that begins in the sinus node spreads immediately into the atrial
muscle wall.
Automatic Electrical Rhythmicity of the Sinus Fibers
Some cardiac fibers have the capability of self-excitation, a process that can
cause automatic rhythmical discharge and contraction. This is especially true of
116
Chapter 10
Rhythmical Excitation of the Heart
117
fiber for three heartbeats and, by comparison, a single
ventricular muscle fiber action potential. Note that the
“resting membrane potential” of the sinus nodal fiber
between discharges has a negativity of about -55 to
-60 millivolts, in comparison with -85 to -90 millivolts
for the ventricular muscle fiber. The cause of this lesser
negativity is that the cell membranes of the sinus fibers
A-V node
Sinus
are naturally leaky to sodium and calcium ions, and
node
positive charges of the entering sodium and calcium
A-V bundle
ions neutralize much of the intracellular negativity.
Before attempting to explain the rhythmicity of the
Left
sinus nodal fibers, first recall from the discussions of
Internodal
bundle
Chapters 5 and 9 that cardiac muscle has three types
pathways
branch
of membrane ion channels that play important roles in
Right
causing the voltage changes of the action potential.
bundle
They are (1) fast sodium channels, (2) slow sodium-
branch
calcium channels, and (3) potassium channels. Opening
of the fast sodium channels for a few 10,000ths of a
second is responsible for the rapid upstroke spike of
the action potential observed in ventricular muscle,
Figure 10-1
because of rapid influx of positive sodium ions to the
interior of the fiber. Then the “plateau” of the ven-
Sinus node, and the Purkinje system of the heart, showing also
tricular action potential is caused primarily by slower
the A-V node, atrial internodal pathways, and ventricular bundle
opening of the slow sodium-calcium channels, which
branches.
lasts for about 0.3 second. Finally, opening of potas-
sium channels allows diffusion of large amounts of
positive potassium ions in the outward direction
through the fiber membrane and returns the mem-
brane potential to its resting level.
Sinus
But there is a difference in the function of these
nodal fiber
channels in the sinus nodal fiber because the “resting”
Ventricular
potential is much less negative—only -55 millivolts in
muscle fiber
Threshold for
the nodal fiber instead of the -90 millivolts in the ven-
+20
discharge
tricular muscle fiber. At this level of -55 millivolts,
0
the fast sodium channels mainly have already become
“inactivated,” which means that they have become
blocked. The cause of this is that any time the mem-
- 40
brane potential remains less negative than about -55
millivolts for more than a few milliseconds, the inacti-
vation gates on the inside of the cell membrane that
“Resting
potential”
close the fast sodium channels become closed and
- 80
remain so. Therefore, only the slow sodium-calcium
channels can open (i.e., can become “activated”) and
0
1
2
3
thereby cause the action potential. As a result, the
Seconds
atrial nodal action potential is slower to develop than
the action potential of the ventricular muscle. Also,
after the action potential does occur, return of the
potential to its negative state occurs slowly as well,
Figure 10-2
rather than the abrupt return that occurs for the
Rhythmical discharge of a sinus nodal fiber. Also, the sinus nodal
ventricular fiber.
action potential is compared with that of a ventricular muscle fiber.
Self-Excitation of Sinus Nodal Fibers. Because of the
high sodium ion concentration in the extracellular
the fibers of the heart’s specialized conducting system,
fluid outside the nodal fiber, as well as a moderate
including the fibers of the sinus node. For this reason,
number of already open sodium channels, positive
the sinus node ordinarily controls the rate of beat
sodium ions from outside the fibers normally tend to
of the entire heart, as discussed in detail later in
leak to the inside. Therefore, between heartbeats,
this chapter. First, let us describe this automatic
influx of positively charged sodium ions causes a slow
rhythmicity.
rise in the resting membrane potential in the positive
direction. Thus, as shown in Figure 10-2, the “resting”
Mechanism of Sinus Nodal Rhythmicity. Figure 10-2 shows
potential gradually rises between each two heartbeats.
action potentials recorded from inside a sinus nodal
When the potential reaches a threshold voltage of
118
Unit III
The Heart
about
-40 millivolts, the sodium-calcium channels
Internodal
become “activated,” thus causing the action potential.
pathways
Transitional fibers
Therefore, basically, the inherent leakiness of the sinus
nodal fibers to sodium and calcium ions causes their
self-excitation.
A-V node
Why does this leakiness to sodium and calcium ions
not cause the sinus nodal fibers to remain depolarized
all the time? The answer is that two events occur
(0.03)
during the course of the action potential to prevent
this. First, the sodium-calcium channels become inac-
Atrioventricular
tivated (i.e., they close) within about 100 to 150 mil-
fibrous tissue
liseconds after opening, and second, at about the same
(0.12)
Penetrating portion
time, greatly increased numbers of potassium channels
of A-V bundle
open. Therefore, influx of positive calcium and sodium
Distal portion of
ions through the sodium-calcium channels ceases,
A-V bundle
while at the same time large quantities of positive
potassium ions diffuse out of the fiber. Both of these
Left bundle branch
Right bundle branch
effects reduce the intracellular potential back to its
negative resting level and therefore terminate the
(0.16)
action potential. Furthermore, the potassium channels
remain open for another few tenths of a second, tem-
porarily continuing movement of positive charges out
of the cell, with resultant excess negativity inside the
Ventricular
fiber; this is called hyperpolarization. The hyperpolar-
septum
ization state initially carries the “resting” membrane
potential down to about -55 to -60 millivolts at the
Figure 10-3
termination of the action potential.
Organization of the A-V node. The numbers represent the interval
Last, we must explain why this new state of hyper-
of time from the origin of the impulse in the sinus node. The values
polarization is not maintained forever. The reason is
have been extrapolated to human beings.
that during the next few tenths of a second after the
action potential is over, progressively more and more
potassium channels close. The inward-leaking sodium
other small bands curve through the anterior, lateral,
and calcium ions once again overbalance the outward
and posterior atrial walls and terminate in the A-V
flux of potassium ions, and this causes the “resting”
node; shown in Figures 10-1 and 10-3, these are called,
potential to drift upward once more, finally reaching
respectively, the anterior, middle, and posterior inter-
the threshold level for discharge at a potential of about
nodal pathways. The cause of more rapid velocity of
-40 millivolts. Then the entire process begins again:
conduction in these bands is the presence of special-
self-excitation to cause the action potential, recovery
ized conduction fibers. These fibers are similar to even
from the action potential, hyperpolarization after the
more rapidly conducting “Purkinje fibers” of the ven-
action potential is over, drift of the “resting” potential
tricles, which will be discussed.
to threshold, and finally re-excitation to elicit another
cycle. This process continues indefinitely throughout a
person’s life.
Atrioventricular Node, and Delay
of Impulse Conduction from the Atria
to the Ventricles
Internodal Pathways and
Transmission of the Cardiac Impulse
The atrial conductive system is organized so that the
Through the Atria
cardiac impulse does not travel from the atria into
the ventricles too rapidly; this delay allows time for the
The ends of the sinus nodal fibers connect directly with
atria to empty their blood into the ventricles before
surrounding atrial muscle fibers. Therefore, action
ventricular contraction begins. It is primarily the A-V
potentials originating in the sinus node travel outward
node and its adjacent conductive fibers that delay this
into these atrial muscle fibers. In this way, the action
transmission into the ventricles.
potential spreads through the entire atrial muscle mass
The A-V node is located in the posterior wall of the
and, eventually, to the A-V node. The velocity of con-
right atrium immediately behind the tricuspid valve,
duction in most atrial muscle is about 0.3 m/sec, but
as shown in Figure 10-1. And Figure 10-3 shows dia-
conduction is more rapid, about 1 m/sec, in several
grammatically the different parts of this node, plus
small bands of atrial fibers. One of these, called the
its connections with the entering atrial internodal
anterior interatrial band, passes through the anterior
pathway fibers and the exiting A-V bundle. The figure
walls of the atria to the left atrium. In addition, three
also shows the approximate intervals of time in
Chapter 10
Rhythmical Excitation of the Heart
119
fractions of a second between initial onset of the
re-entry of cardiac impulses by this route from the
cardiac impulse in the sinus node and its subsequent
ventricles to the atria, allowing only forward conduc-
appearance in the A-V nodal system. Note that the
tion from the atria to the ventricles.
impulse, after traveling through the internodal path-
Furthermore, it should be recalled that everywhere,
ways, reaches the A-V node about 0.03 second after its
except at the A-V bundle, the atrial muscle is sepa-
origin in the sinus node. Then there is a delay of
rated from the ventricular muscle by a continuous
another 0.09 second in the A-V node itself before the
fibrous barrier, a portion of which is shown in Figure
impulse enters the penetrating portion of the A-V
10-3. This barrier normally acts as an insulator to
bundle, where it passes into the ventricles. A final delay
prevent passage of the cardiac impulse between atrial
of another 0.04 second occurs mainly in this penetrat-
and ventricular muscle through any other route
ing A-V bundle, which is composed of multiple small
besides forward conduction through the A-V bundle
fascicles passing through the fibrous tissue separating
itself. (In rare instances, an abnormal muscle bridge
the atria from the ventricles.
does penetrate the fibrous barrier elsewhere besides
Thus, the total delay in the A-V nodal and A-V
at the A-V bundle. Under such conditions, the cardiac
bundle system is about 0.13 second. This, in addition to
impulse can re-enter the atria from the ventricles and
the initial conduction delay of 0.03 second from the
cause a serious cardiac arrhythmia.)
sinus node to the A-V node, makes a total delay of 0.16
second before the excitatory signal finally reaches the
Distribution of the Purkinje Fibers in the Ventricles—The Left
contracting muscle of the ventricles.
and Right Bundle Branches. After penetrating the fibrous
tissue between the atrial and ventricular muscle, the
Cause of the Slow Conduction. The slow conduction in the
distal portion of the A-V bundle passes downward in
transitional, nodal, and penetrating A-V bundle fibers
the ventricular septum for 5 to 15 millimeters toward
is caused mainly by diminished numbers of gap junc-
the apex of the heart, as shown in Figures 10-1 and
tions between successive cells in the conducting path-
10-3. Then the bundle divides into left and right bundle
ways, so that there is great resistance to conduction of
branches that lie beneath the endocardium on the two
excitatory ions from one conducting fiber to the next.
respective sides of the ventricular septum. Each
Therefore, it is easy to see why each succeeding cell is
branch spreads downward toward the apex of the ven-
slow to be excited.
tricle, progressively dividing into smaller branches.
These branches in turn course sidewise around each
ventricular chamber and back toward the base of the
heart. The ends of the Purkinje fibers penetrate about
Rapid Transmission in the Ventricular
one third the way into the muscle mass and finally
Purkinje System
become continuous with the cardiac muscle fibers.
From the time the cardiac impulse enters the bundle
Special Purkinje fibers lead from the A-V node
branches in the ventricular septum until it reaches the
through the A-V bundle into the ventricles. Except for
terminations of the Purkinje fibers, the total elapsed
the initial portion of these fibers where they penetrate
time averages only 0.03 second. Therefore, once the
the A-V fibrous barrier, they have functional charac-
cardiac impulse enters the ventricular Purkinje con-
teristics that are quite the opposite of those of the
ductive system, it spreads almost immediately to the
A-V nodal fibers. They are very large fibers, even
entire ventricular muscle mass.
larger than the normal ventricular muscle fibers, and
they transmit action potentials at a velocity of 1.5 to
4.0 m/sec, a velocity about 6 times that in the usual
ventricular muscle and 150 times that in some of the
Transmission of the Cardiac Impulse
A-V nodal fibers. This allows almost instantaneous
in the Ventricular Muscle
transmission of the cardiac impulse throughout the
entire remainder of the ventricular muscle.
Once the impulse reaches the ends of the Purkinje
The rapid transmission of action potentials by Purk-
fibers, it is transmitted through the ventricular muscle
inje fibers is believed to be caused by a very high level
mass by the ventricular muscle fibers themselves. The
of permeability of the gap junctions at the intercalated
velocity of transmission is now only 0.3 to 0.5 m/sec,
discs between the successive cells that make up the
one sixth that in the Purkinje fibers.
Purkinje fibers. Therefore, ions are transmitted easily
The cardiac muscle wraps around the heart in a
from one cell to the next, thus enhancing the velocity
double spiral, with fibrous septa between the spiraling
of transmission. The Purkinje fibers also have very few
layers; therefore, the cardiac impulse does not neces-
myofibrils, which means that they contract little or not
sarily travel directly outward toward the surface of the
at all during the course of impulse transmission.
heart but instead angulates toward the surface along
the directions of the spirals. Because of this, transmis-
One-Way Conduction Through the A-V Bundle. A special
sion from the endocardial surface to the epicardial
characteristic of the A-V bundle is the inability, except
surface of the ventricle requires as much as another
in abnormal states, of action potentials to travel back-
0.03 second, approximately equal to the time required
ward from the ventricles to the atria. This prevents
for transmission through the entire ventricular portion
120
Unit III
The Heart
of the Purkinje system. Thus, the total time for trans-
Control of Excitation and
mission of the cardiac impulse from the initial bundle
Conduction in the Heart
branches to the last of the ventricular muscle fibers in
the normal heart is about 0.06 second.
The Sinus Node as the Pacemaker
of the Heart
Summary of the Spread of the Cardiac
In the discussion thus far of the genesis and transmis-
Impulse Through the Heart
sion of the cardiac impulse through the heart, we have
noted that the impulse normally arises in the sinus
Figure 10-4 shows in summary form the transmission
node. In some abnormal conditions, this is not the case.
of the cardiac impulse through the human heart. The
A few other parts of the heart can exhibit intrinsic
numbers on the figure represent the intervals of time,
rhythmical excitation in the same way that the sinus
in fractions of a second, that lapse between the origin
nodal fibers do; this is particularly true of the A-V
of the cardiac impulse in the sinus node and its appear-
nodal and Purkinje fibers.
ance at each respective point in the heart. Note that
The A-V nodal fibers, when not stimulated from
the impulse spreads at moderate velocity through the
some outside source, discharge at an intrinsic rhyth-
atria but is delayed more than 0.1 second in the A-V
mical rate of 40 to 60 times per minute, and the Purk-
nodal region before appearing in the ventricular septal
inje fibers discharge at a rate somewhere between
A-V bundle. Once it has entered this bundle, it spreads
15 and 40 times per minute. These rates are in contrast
very rapidly through the Purkinje fibers to the entire
to the normal rate of the sinus node of 70 to 80 times
endocardial surfaces of the ventricles. Then the impulse
per minute.
once again spreads slightly less rapidly through the
The question we must ask is: Why does the sinus
ventricular muscle to the epicardial surfaces.
node rather than the A-V node or the Purkinje fibers
It is extremely important that the student learn in
control the heart’s rhythmicity? The answer derives
detail the course of the cardiac impulse through the
from the fact that the discharge rate of the sinus node
heart and the precise times of its appearance in each
is considerably faster than the natural self-excitatory
separate part of the heart, because a thorough quanti-
discharge rate of either the A-V node or the Purkinje
tative knowledge of this process is essential to the
fibers. Each time the sinus node discharges, its impulse
understanding of electrocardiography, to be discussed
is conducted into both the A-V node and the Purkinje
in Chapters 11 through 13.
fibers, also discharging their excitable membranes. But
the sinus node discharges again before either the A-V
node or the Purkinje fibers can reach their own thresh-
olds for self-excitation. Therefore, the new impulse
from the sinus node discharges both the A-V node and
the Purkinje fibers before self-excitation can occur in
either of these.
Thus, the sinus node controls the beat of the heart
.07
because its rate of rhythmical discharge is faster than
that of any other part of the heart. Therefore, the sinus
.04
.09
.06
node is virtually always the pacemaker of the normal
S-A
.22
heart.
.03
.07
.19
Abnormal Pacemakers—“Ectopic” Pacemaker. Occasionally
some other part of the heart develops a rhythmical dis-
.00
.16
A-V
charge rate that is more rapid than that of the sinus
node. For instance, this sometimes occurs in the A-V
.05
.03
.18
.21
node or in the Purkinje fibers when one of these
.17
becomes abnormal. In either case, the pacemaker of
.07
.17
the heart shifts from the sinus node to the A-V node
.19
or to the excited Purkinje fibers. Under rarer condi-
tions, a place in the atrial or ventricular muscle devel-
.18
ops excessive excitability and becomes the pacemaker.
A pacemaker elsewhere than the sinus node is
.21
called an “ectopic” pacemaker. An ectopic pacemaker
causes an abnormal sequence of contraction of the
.20
different parts of the heart and can cause significant
debility of heart pumping.
Figure 10-4
Another cause of shift of the pacemaker is blockage
of transmission of the cardiac impulse from the sinus
Transmission of the cardiac impulse through the heart, showing
the time of appearance (in fractions of a second after initial
node to the other parts of the heart. The new pace-
appearance at the sinoatrial node) in different parts of the heart.
maker then occurs most frequently at the A-V node or
Chapter 10
Rhythmical Excitation of the Heart
121
in the penetrating portion of the A-V bundle on the
Parasympathetic (Vagal) Stimulation Can Slow or Even Block
way to the ventricles.
Cardiac Rhythm and Conduction—“Ventricular Escape.”
When A-V block occurs—that is, when the cardiac
Stimulation of the parasympathetic nerves to the heart
impulse fails to pass from the atria into the ventricles
(the vagi) causes the hormone acetylcholine to be
through the A-V nodal and bundle system—the atria
released at the vagal endings. This hormone has two
continue to beat at the normal rate of rhythm of the
major effects on the heart. First, it decreases the rate
sinus node, while a new pacemaker usually develops
of rhythm of the sinus node, and second, it decreases
in the Purkinje system of the ventricles and drives the
the excitability of the A-V junctional fibers between
ventricular muscle at a new rate somewhere between
the atrial musculature and the A-V node, thereby
15 and 40 beats per minute. After sudden A-V bundle
slowing transmission of the cardiac impulse into the
block, the Purkinje system does not begin to emit its
ventricles.
intrinsic rhythmical impulses until 5 to 20 seconds later
Weak to moderate vagal stimulation slows the rate
because, before the blockage, the Purkinje fibers had
of heart pumping, often to as little as one half normal.
been “overdriven” by the rapid sinus impulses and,
And strong stimulation of the vagi can stop completely
consequently, are in a suppressed state. During these
the rhythmical excitation by the sinus node or block
5 to 20 seconds, the ventricles fail to pump blood, and
completely transmission of the cardiac impulse from
the person faints after the first 4 to 5 seconds because
the atria into the ventricles through the A-V mode. In
of lack of blood flow to the brain. This delayed pickup
either case, rhythmical excitatory signals are no longer
of the heartbeat is called Stokes-Adams syndrome. If
transmitted into the ventricles. The ventricles stop
the delay period is too long, it can lead to death.
beating for 5 to 20 seconds, but then some point in the
Purkinje fibers, usually in the ventricular septal
portion of the A-V bundle, develops a rhythm of its
Role of the Purkinje System
own and causes ventricular contraction at a rate of
15 to 40 beats per minute. This phenomenon is called
in Causing Synchronous Contraction
ventricular escape.
of the Ventricular Muscle
Mechanism of the Vagal Effects. The acetylcholine
It is clear from our description of the Purkinje system
released at the vagal nerve endings greatly increases
that normally the cardiac impulse arrives at almost all
the permeability of the fiber membranes to potassium
portions of the ventricles within a narrow span of time,
ions, which allows rapid leakage of potassium out of
exciting the first ventricular muscle fiber only 0.03 to
the conductive fibers. This causes increased negativity
0.06 second ahead of excitation of the last ventricular
inside the fibers, an effect called hyperpolarization,
muscle fiber. This causes all portions of the ventricular
which makes this excitable tissue much less excitable,
muscle in both ventricles to begin contracting at
as explained in Chapter 5.
almost the same time and then to continue contract-
In the sinus node, the state of hyperpolarization
ing for about another 0.3 second.
decreases the “resting” membrane potential of the
Effective pumping by the two ventricular chambers
sinus nodal fibers to a level considerably more nega-
requires this synchronous type of contraction. If the
tive than usual, to -65 to -75 millivolts rather than
cardiac impulse should travel through the ventricles
the normal level of -55 to -60 millivolts. Therefore,
slowly, much of the ventricular mass would contract
the initial rise of the sinus nodal membrane potential
before contraction of the remainder, in which case the
caused by inward sodium and calcium leakage
overall pumping effect would be greatly depressed.
requires much longer to reach the threshold potential
Indeed, in some types of cardiac debilities, several of
for excitation. This greatly slows the rate of rhythmic-
which are discussed in Chapters 12 and 13, slow trans-
ity of these nodal fibers. If the vagal stimulation is
mission does occur, and the pumping effectiveness of
strong enough, it is possible to stop entirely the rhyth-
the ventricles is decreased as much as 20 to 30 per cent.
mical self-excitation of this node.
In the A-V node, a state of hyperpolarization caused
by vagal stimulation makes it difficult for the small
Control of Heart Rhythmicity and
atrial fibers entering the node to generate enough elec-
Impulse Conduction by the Cardiac
tricity to excite the nodal fibers. Therefore, the safety
Nerves: The Sympathetic and
factor for transmission of the cardiac impulse through
Parasympathetic Nerves
the transitional fibers into the A-V nodal fibers
decreases. A moderate decrease simply delays con-
The heart is supplied with both sympathetic and
duction of the impulse, but a large decrease blocks
parasympathetic nerves, as shown in Figure 9-10 of
conduction entirely.
Chapter 9. The parasympathetic nerves (the vagi) are
distributed mainly to the S-A and A-V nodes, to a
Effect of Sympathetic Stimulation on Cardiac Rhythm and Con-
lesser extent to the muscle of the two atria, and very
duction. Sympathetic stimulation causes essentially the
little directly to the ventricular muscle. The sympa-
opposite effects on the heart to those caused by vagal
thetic nerves, conversely, are distributed to all parts of
stimulation, as follows: First, it increases the rate of
the heart, with strong representation to the ventricu-
sinus nodal discharge. Second, it increases the rate
lar muscle as well as to all the other areas.
of conduction as well as the level of excitability in all
122
Unit III
The Heart
portions of the heart. Third, it increases greatly the
Gentlesk PJ, Markwood TT, Atwood JE: Chronotropic
force of contraction of all the cardiac musculature,
incompetence in a young adult: case report and literature
both atrial and ventricular, as discussed in Chapter 9.
review. Chest 125:297, 2004.
Huikuri HV, Castellanos A, Myerburg RJ: Sudden death due
In short, sympathetic stimulation increases the
to cardiac arrhythmias. N Engl J Med 345:1473, 2001.
overall activity of the heart. Maximal stimulation
Hume JR, Duan D, Collier ML, et al: Anion transport in
can almost triple the frequency of heartbeat and can
heart. Physiol Rev 80:31, 2000.
increase the strength of heart contraction as much as
James TN: Structure and function of the sinus node, AV node
twofold.
and His bundle of the human heart: part I—structure. Prog
Cardiovasc Dis 45:235, 2002.
Mechanism of the Sympathetic Effect. Stimulation of
James TN: Structure and function of the sinus node, AV node
the sympathetic nerves releases the hormone norepi-
and His bundle of the human heart: part II—function.
nephrine at the sympathetic nerve endings. The precise
Prog Cardiovasc Dis 45:327, 2003.
mechanism by which this hormone acts on cardiac
Kaupp UB, Seifert R: Molecular diversity of pacemaker ion
channels. Annu Rev Physiol 63:235, 2001.
muscle fibers is somewhat unclear, but the belief is
Kléber AG, Rudy Y: Basic mechanisms of cardiac impulse
that it increases the permeability of the fiber mem-
propagation and associated arrhythmias. Physiol Rev
brane to sodium and calcium ions. In the sinus node,
84:431, 2004.
an increase of sodium-calcium permeability causes
Leclercq C, Hare JM: Ventricular resynchronization: current
a more positive resting potential and also causes
state of the art. Circulation 109:296, 2004.
increased rate of upward drift of the diastolic mem-
Mazgalev TN, Ho SY, Anderson RH: Anatomic-
brane potential toward the threshold level for self-
electrophysiological correlations concerning the pathways
excitation, thus accelerating self-excitation and,
for atrioventricular conduction. Circulation
103:2660,
therefore, increasing the heart rate.
2001.
In the A-V node and A-V bundles, increased
Page E, Fozzard HA, Solaro JR: Handbook of Physiology,
sec 2: The Cardiovascular System, vol 1: The Heart. New
sodium-calcium permeability makes it easier for the
York: Oxford University Press, 2002.
action potential to excite each succeeding portion of
Petrashevskaya NN, Koch SE, Bodi I, Schwartz A: Calcium
the conducting fiber bundles, thereby decreasing the
cycling, historic overview and perspectives: role for auto-
conduction time from the atria to the ventricles.
nomic nervous system regulation. J Mol Cell Cardiol
The increase in permeability to calcium ions is at
34:885, 2002.
least partially responsible for the increase in contrac-
Priori SG: Inherited arrhythmogenic diseases: the com-
tile strength of the cardiac muscle under the influence
plexity beyond monogenic disorders. Circ Res 94:140,
of sympathetic stimulation, because calcium ions play
2004.
a powerful role in exciting the contractile process of
Roden DM, Balser JR, George AL Jr, Anderson ME:
the myofibrils.
Cardiac ion channels. Annu Rev Physiol 64:431, 2002.
Schram G, Pourrier M, Melnyk P, Nattel S: Differential dis-
tribution of cardiac ion channel expression as a basis for
regional specialization in electrical function. Circ Res
References
90:939, 2002.
Surawicz B: Electrophysiologic Basis of ECG and Cardiac
Blatter LA, Kockskamper J, Sheehan KA, et al: Local
Arrhythmias. Baltimore: Williams & Wilkins, 1995.
calcium gradients during excitation-contraction coupling
Waldo AL: Mechanisms of atrial fibrillation. J Cardiovasc
and alternans in atrial myocytes. J Physiol 546:19, 2003.
Electrophysiol 14(12 Suppl):S267, 2003.
Ferrier GR, Howlett SE: Cardiac excitation-contraction cou-
Yasuma F, Hayano J: Respiratory sinus arrhythmia: why does
pling: role of membrane potential in regulation of con-
the heartbeat synchronize with respiratory rhythm? Chest
traction. Am J Physiol Heart Circ Physiol 280:H1928, 2001.
125:683, 2004.
C H A P T E R
1
1
The Normal Electrocardiogram
When the cardiac impulse passes through the heart,
electrical current also spreads from the heart into
the adjacent tissues surrounding the heart. A small
portion of the current spreads all the way to the
surface of the body. If electrodes are placed on the
skin on opposite sides of the heart, electrical
potentials generated by the current can be recorded;
the recording is known as an electrocardiogram.
A normal electrocardiogram for two beats of the heart is shown in Figure 11-1.
Characteristics of the Normal
Electrocardiogram
The normal electrocardiogram (see Figure 11-1) is composed of a P wave, a
QRS complex, and a T wave. The QRS complex is often, but not always, three
separate waves: the Q wave, the R wave, and the S wave.
The P wave is caused by electrical potentials generated when the atria depo-
larize before atrial contraction begins. The QRS complex is caused by poten-
tials generated when the ventricles depolarize before contraction, that is, as the
depolarization wave spreads through the ventricles. Therefore, both the P wave
and the components of the QRS complex are depolarization waves.
The T wave is caused by potentials generated as the ventricles recover from
the state of depolarization. This process normally occurs in ventricular muscle
0.25 to 0.35 second after depolarization, and the T wave is known as a repolar-
ization wave.
Thus, the electrocardiogram is composed of both depolarization and repo-
larization waves. The principles of depolarization and repolarization are
discussed in Chapter 5. The distinction between depolarization waves and repo-
larization waves is so important in electrocardiography that further clarification
is needed.
Depolarization Waves Versus Repolarization Waves
Figure 11-2 shows a single cardiac muscle fiber in four stages of depolarization
and repolarization, the color red designating depolarization. During depolar-
ization, the normal negative potential inside the fiber reverses and becomes
slightly positive inside and negative outside.
In Figure 11-2A, depolarization, demonstrated by red positive charges inside
and red negative charges outside, is traveling from left to right. The first half of
the fiber has already depolarized, while the remaining half is still polarized.
Therefore, the left electrode on the outside of the fiber is in an area of nega-
tivity, and the right electrode is in an area of positivity; this causes the meter to
record positively. To the right of the muscle fiber is shown a record of changes
in potential between the two electrodes, as recorded by a high-speed recording
meter. Note that when depolarization has reached the halfway mark in Figure
11-2A, the record has risen to a maximum positive value.
123
124
Unit III
The Heart
In Figure 11-2B, depolarization has extended over
Figure 11-2C shows halfway repolarization of the
the entire muscle fiber, and the recording to the right
same muscle fiber, with positivity returning to the
has returned to the zero baseline because both elec-
outside of the fiber. At this point, the left electrode is
trodes are now in areas of equal negativity. The com-
in an area of positivity, and the right electrode is in an
pleted wave is a depolarization wave because it results
area of negativity. This is opposite to the polarity in
from spread of depolarization along the muscle fiber
Figure 11-2A. Consequently, the recording, as shown
membrane.
to the right, becomes negative.
In Figure 11-2D, the muscle fiber has completely
repolarized, and both electrodes are now in areas of
positivity, so that no potential difference is recorded
between them. Thus, in the recording to the right, the
potential returns once more to zero. This completed
Atria Ventricles
negative wave is a repolarization wave because it
+2
results from spread of repolarization along the muscle
RR interval
fiber membrane.
R
S-T
+1
segment
Relation of the Monophasic Action Potential of Ventricular
Muscle to the QRS and T Waves in the Standard Electrocardio-
T
P
gram. The monophasic action potential of ventricular
0
muscle, discussed in Chapter 10, normally lasts between
Q
S
0.25 and 0.35 second. The top part of Figure 11-3 shows
P-R
interval
Q-T interval
a monophasic action potential recorded from a micro-
= 0.16 sec
–1
electrode inserted to the inside of a single ventricular
0
0.2
0.4
0.6
0.8
1.0
1.2
1.4
1.6
muscle fiber. The upsweep of this action potential is
Time (sec)
caused by depolarization, and the return of the poten-
tial to the baseline is caused by repolarization.
Note in the lower half of the figure a simultaneous
Figure 11-1
recording of the electrocardiogram from this same
Normal electrocardiogram.
ventricle, which shows the QRS waves appearing at
0
-
+
-
+
- - - - - - -+ + + + + + + + +
+
- - - - - - - - -
+ + + + + + +
- - - - - - - - -
+ + + + + + +
A
- - - - - - -+ + + + + + + + +
–
Depolarization
wave
+
- - - - - - - - - - - - - - - -
+ + + + + + + + + + + + + + + +
+ + + + + + + + + + + + + + + +
B
– - - - - - - - - - - - - - - -
-
+
+ + + + + + + + + - - - - - - -
- - - - - - - - - + + + + + + +
- - - - - - - - - + + + + + + +
C
+ + + + + + + + + - - - - - - -
-
Repolarization
+ + + + + + + + + + + + + + + +
+
wave
- - - - - - - - - - - - - - - -
- - - - - - - - - - - - - - - -
D
Figure 11-2
-
+ + + + + + + + + + + + + + + +
0.30 second
Recording the depolarization wave (A and B) and
the repolarization wave (C and D) from a cardiac
muscle fiber.
Chapter 11
The Normal Electrocardiogram
125
(the QRS complex), but in many other fibers, it takes
as long as 0.35 second. Thus, the process of ventricular
repolarization extends over a long period, about 0.15
second. For this reason, the T wave in the normal elec-
trocardiogram is a prolonged wave, but the voltage of
the T wave is considerably less than the voltage of the
QRS complex, partly because of its prolonged length.
Voltage and Time Calibration of the
Electrocardiogram
R
T
All recordings of electrocardiograms are made with
Q
appropriate calibration lines on the recording paper.
S
Either these calibration lines are already ruled on the
paper, as is the case when a pen recorder is used, or
they are recorded on the paper at the same time that
the electrocardiogram is recorded, which is the case
with the photographic types of electrocardiographs.
Figure 11-3
As shown in Figure 11-1, the horizontal calibration
lines are arranged so that 10 of the small line divisions
Above, Monophasic action potential from a ventricular muscle
upward or downward in the standard electrocardio-
fiber during normal cardiac function, showing rapid depolarization
gram represent 1 millivolt, with positivity in the upward
and then repolarization occurring slowly during the plateau stage
but rapidly toward the end. Below, Electrocardiogram recorded
direction and negativity in the downward direction.
simultaneously.
The vertical lines on the electrocardiogram are time
calibration lines. Each inch in the horizontal direction
is 1 second, and each inch is usually broken into five
the beginning of the monophasic action potential and
segments by dark vertical lines; the intervals between
the T wave appearing at the end. Note especially that
these dark lines represent 0.20 second. The 0.20 second
no potential is recorded in the electrocardiogram when
intervals are then broken into five smaller intervals by
the ventricular muscle is either completely polarized
thin lines, each of which represents 0.04 second.
or completely depolarized. Only when the muscle is
Normal Voltages in the Electrocardiogram. The recorded
partly polarized and partly depolarized does current
voltages of the waves in the normal electrocardiogram
flow from one part of the ventricles to another part,
depend on the manner in which the electrodes are
and therefore current also flows to the surface of the
applied to the surface of the body and how close the
body to produce the electrocardiogram.
electrodes are to the heart. When one electrode is
placed directly over the ventricles and a second elec-
Relationship of Atrial and Ventricular
trode is placed elsewhere on the body remote from the
heart, the voltage of the QRS complex may be as great
Contraction to the Waves of the
as 3 to 4 millivolts. Even this voltage is small in com-
Electrocardiogram
parison with the monophasic action potential of 110
millivolts recorded directly at the heart muscle mem-
Before contraction of muscle can occur, depolarization
brane. When electrocardiograms are recorded from
must spread through the muscle to initiate the chemi-
electrodes on the two arms or on one arm and one leg,
cal processes of contraction. Refer again to Figure
the voltage of the QRS complex usually is 1.0 to 1.5
11-1; the P wave occurs at the beginning of contrac-
millivolt from the top of the R wave to the bottom of
tion of the atria, and the QRS complex of waves occurs
the S wave; the voltage of the P wave is between 0.1
at the beginning of contraction of the ventricles. The
and 0.3 millivolt; and that of the T wave is between 0.2
ventricles remain contracted until after repolarization
and 0.3 millivolt.
has occurred, that is, until after the end of the T wave.
The atria repolarize about 0.15 to 0.20 second after
P-Q or P-R Interval. The time between the beginning of
termination of the P wave. This is also approxima-
the P wave and the beginning of the QRS complex is
tely when the QRS complex is being recorded in
the interval between the beginning of electrical exci-
the electrocardiogram. Therefore, the atrial repo-
tation of the atria and the beginning of excitation of
larization wave, known as the atrial T wave, is usually
the ventricles. This period is called the P-Q interval.
obscured by the much larger QRS complex. For
The normal P-Q interval is about 0.16 second. (Often
this reason, an atrial T wave seldom is observed in the
this interval is called the P-R interval because the Q
electrocardiogram.
wave is likely to be absent.)
The ventricular repolarization wave is the T wave of
the normal electrocardiogram. Ordinarily, ventricular
Q-T Interval. Contraction of the ventricle lasts almost
muscle begins to repolarize in some fibers about 0.20
from the beginning of the Q wave (or R wave, if
second after the beginning of the depolarization wave
the Q wave is absent) to the end of the T wave. This
126
Unit III
The Heart
interval is called the Q-T interval and ordinarily is
about 0.35 second.
0
0
0
Rate of Heartbeat as Determined from the Electrocardiogram.
The rate of heartbeat can be determined easily from
–
+
–
+
-
+
an electrocardiogram because the heart rate is the
reciprocal of the time interval between two successive
–
+
-
+
-
+
heartbeats. If the interval between two beats as deter-
mined from the time calibration lines is 1 second, the
+++++
heart rate is 60 beats per minute. The normal interval
+
+++++++++
+
+
++
+
+
++++
++
++++++
between two successive QRS complexes in the adult
- - - - -
++++++
++++++
– - - - - -
-
+
- - - - - - - - -
++++
++
++++++
- - - - - -
- - -
person is about 0.83 second. This is a heart rate of
- - - - - -
- - -
++++++
- - - -
- - - - -
++++++
- - - - - - - - -
+++++
60/0.83 times per minute, or 72 beats per minute.
+++++
- - - - - - - - -
++++++-
- - - - - - - -
++++++
- - - - - - - -
++++++
- - - - - - -
++++++
- - - -
++++++++
- -
++++++
+
++++
+++++++++++
+
Methods for Recording
++++++++++
Electrocardiograms
Sometimes the electrical currents generated by the
cardiac muscle during each beat of the heart change
Figure 11-4
electrical potentials and polarities on the respective
Instantaneous potentials develop on the surface of a cardiac
sides of the heart in less than 0.01 second. Therefore,
muscle mass that has been depolarized in its center.
it is essential that any apparatus for recording electro-
cardiograms be capable of responding rapidly to these
changes in potentials.
Before stimulation, all the exteriors of the muscle cells
had been positive and the interiors negative. For
Pen Recorder
reasons presented in Chapter 5 in the discussion of
membrane potentials, as soon as an area of cardiac
Many modern clinical electrocardiographs use com-
syncytium becomes depolarized, negative charges leak
puter-based systems and electronic display, while
to the outsides of the depolarized muscle fibers,
others use a direct pen recorder that writes the elec-
making this part of the surface electronegative, as rep-
trocardiogram with a pen directly on a moving sheet
resented by the negative signs in Figure 11-4. The
of paper. Sometimes the pen is a thin tube connected
remaining surface of the heart, which is still polarized,
at one end to an inkwell, and its recording end is con-
is represented by the positive signs. Therefore, a meter
nected to a powerful electromagnet system that is
connected with its negative terminal on the area of
capable of moving the pen back and forth at high
depolarization and its positive terminal on one of the
speed. As the paper moves forward, the pen records
still-polarized areas, as shown to the right in the figure,
the electrocardiogram. The movement of the pen is con-
records positively.
trolled by appropriate electronic amplifiers connected
Two other electrode placements and meter readings
to electrocardiographic electrodes on the patient.
are also demonstrated in Figure 11-4. These should be
Other pen recording systems use special paper that
studied carefully, and the reader should be able to
does not require ink in the recording stylus. One such
explain the causes of the respective meter readings.
paper turns black when it is exposed to heat; the stylus
Because the depolarization spreads in all directions
itself is made very hot by electrical current flowing
through the heart, the potential differences shown
through its tip. Another type turns black when electri-
in the figure persist for only a few thousandths of
cal current flows from the tip of the stylus through the
a second, and the actual voltage measurements can
paper to an electrode at its back. This leaves a black
be accomplished only with a high-speed recording
line on the paper where the stylus touches.
apparatus.
Flow of Current Around
Flow of Electrical Currents in the
the Heart During the
Chest Around the Heart
Cardiac Cycle
Figure 11-5 shows the ventricular muscle lying within
the chest. Even the lungs, although mostly filled
Recording Electrical Potentials from a
with air, conduct electricity to a surprising extent, and
Partially Depolarized Mass of
fluids in other tissues surrounding the heart conduct
Syncytial Cardiac Muscle
electricity even more easily. Therefore, the heart
is actually suspended in a conductive medium.
Figure 11-4 shows a syncytial mass of cardiac muscle
When one portion of the ventricles depolarizes and
that has been stimulated at its centralmost point.
therefore becomes electronegative with respect to the
Chapter 11
The Normal Electrocardiogram
127
+0.5 mV
0
-
+
-
+
Lead I
0
-
+
-
+
-
-
-
+
+
+
B
A
-0.2 mV
+++
+0.3 mV
+
--
+
+
-
-
+
+
-
-
-+
++
-
-
+
+
+
-
+
+
+
-
++
+
+
++
+
++
+
++
++
+1.2 mV
+0.7 mV
0
0
-
+
-
+
-
+
-
+
Figure 11-5
Lead II
Lead III
Flow of current
in
the chest around
partially
depolarized
ventricles.
+1.0 mV
remainder, electrical current flows from the depolar-
Figure 11-6
ized area to the polarized area in large circuitous
routes, as noted in the figure.
Conventional arrangement of electrodes for recording the stan-
It should be recalled from the discussion of the
dard electrocardiographic leads. Einthoven’s triangle is superim-
Purkinje system in Chapter 10 that the cardiac impulse
posed on the chest.
first arrives in the ventricles in the septum and shortly
thereafter spreads to the inside surfaces of the remain-
der of the ventricles, as shown by the red areas and the
negative signs in Figure
11-5. This provides elec-
Thus, in normal heart ventricles, current flows from
tronegativity on the insides of the ventricles and elec-
negative to positive primarily in the direction from
tropositivity on the outer walls of the ventricles, with
the base of the heart toward the apex during almost
electrical current flowing through the fluids surround-
the entire cycle of depolarization, except at the very
ing the ventricles along elliptical paths, as demon-
end. And if a meter is connected to electrodes on
strated by the curving arrows in the figure. If one
the surface of the body as shown in Figure 11-5, the
algebraically averages all the lines of current flow (the
electrode nearer the base will be negative, whereas
elliptical lines), one finds that the average current flow
the electrode nearer the apex will be positive, and
occurs with negativity toward the base of the heart and
the recording meter will show positive recording in the
with positivity toward the apex.
electrocardiogram.
During most of the remainder of the depolarization
process, current also continues to flow in this same
direction, while depolarization spreads from the endo-
Electrocardiographic Leads
cardial surface outward through the ventricular
muscle mass. Then, immediately before depolarization
Three Bipolar Limb Leads
has completed its course through the ventricles, the
average direction of current flow reverses for about
Figure 11-6 shows electrical connections between the
0.01 second, flowing from the ventricular apex toward
patient’s limbs and the electrocardiograph for record-
the base, because the last part of the heart to become
ing electrocardiograms from the so-called standard
depolarized is the outer walls of the ventricles near the
bipolar limb leads. The term “bipolar” means that the
base of the heart.
electrocardiogram is recorded from two electrodes
128
Unit III
The Heart
located on different sides of the heart, in this case, on
Now, note that the sum of the voltages in leads I and
the limbs. Thus, a “lead” is not a single wire connect-
III equals the voltage in lead II; that is,
0.5 plus
ing from the body but a combination of two wires and
0.7 equals 1.2. Mathematically, this principle, called
their electrodes to make a complete circuit between
Einthoven’s law, holds true at any given instant while
the body and the electrocardiograph. The electrocar-
the three “standard” bipolar electrocardiograms are
diograph in each instance is represented by an elec-
being recorded.
trical meter in the diagram, although the actual
electrocardiograph is a high-speed recording meter
Normal Electrocardiograms Recorded from the Three Standard
with a moving paper.
Bipolar Limb Leads. Figure 11-7 shows recordings of the
electrocardiograms in leads I, II, and III. It is obvious
Lead I. In recording limb lead I, the negative terminal
that the electrocardiograms in these three leads are
of the electrocardiograph is connected to the right arm
similar to one another because they all record positive
and the positive terminal to the left arm. Therefore,
P waves and positive T waves, and the major portion
when the point where the right arm connects to the
of the QRS complex is also positive in each
chest is electronegative with respect to the point where
electrocardiogram.
the left arm connects, the electrocardiograph records
On analysis of the three electrocardiograms, it can
positively, that is, above the zero voltage line in the
be shown, with careful measurements and proper
electrocardiogram. When the opposite is true, the elec-
observance of polarities, that at any given instant the
trocardiograph records below the line.
sum of the potentials in leads I and III equals the
potential in lead II, thus illustrating the validity of
Lead II. To record limb lead II, the negative terminal of
Einthoven’s law.
the electrocardiograph is connected to the right arm and
Because the recordings from all the bipolar limb
the positive terminal to the left leg. Therefore, when the
leads are similar to one another, it does not matter
right arm is negative with respect to the left leg, the
greatly which lead is recorded when one wants to
electrocardiograph records positively.
diagnose different cardiac arrhythmias, because diag-
nosis of arrhythmias depends mainly on the time
Lead III. To record limb lead III, the negative terminal
relations between the different waves of the cardiac
of the electrocardiograph is connected to the left arm
cycle. But when one wants to diagnose damage in
and the positive terminal to the left leg. This means that
the ventricular or atrial muscle or in the Purkinje
the electrocardiograph records positively when the left
conducting system, it does matter greatly which leads
arm is negative with respect to the left leg.
are recorded, because abnormalities of cardiac
muscle contraction or cardiac impulse conduction do
Einthoven’s Triangle. In Figure 11-6, the triangle, called
Einthoven’s triangle, is drawn around the area of the
heart. This illustrates that the two arms and the left leg
form apices of a triangle surrounding the heart. The
two apices at the upper part of the triangle represent
the points at which the two arms connect electrically
with the fluids around the heart, and the lower apex is
the point at which the left leg connects with the fluids.
I
Einthoven’s Law. Einthoven’s law states that if the
electrical potentials of any two of the three bipolar
limb electrocardiographic leads are known at any
given instant, the third one can be determined
mathematically by simply summing the first two (but
II
note that the positive and negative signs of the
different leads must be observed when making this
summation).
For instance, let us assume that momentarily, as
noted in Figure 11-6, the right arm is -0.2 millivolt
(negative) with respect to the average potential in
the body, the left arm is + 0.3 millivolt (positive), and
III
the left leg is +1.0 millivolt (positive). Observing the
meters in the figure, it can be seen that lead I records
a positive potential of +0.5 millivolt, because this is the
difference between the -0.2 millivolt on the right arm
and the +0.3 millivolt on the left arm. Similarly, lead
III records a positive potential of +0.7 millivolt, and
Figure 11-7
lead II records a positive potential of +1.2 millivolts
because these are the instantaneous potential differ-
Normal electrocardiograms
recorded
from
the
three
standard
ences between the respective pairs of limbs.
electrocardiographic leads.
Chapter 11
The Normal Electrocardiogram
129
1
2
3456
RA
LA
V1
V2
V3
V4
V5
V6
5000
ohms
Figure 11-9
5000
ohms
Normal electrocardiograms recorded from the six standard chest
leads.
0
-
+
-
+
5000
ohms
aVR
aVL
aVF
Figure 11-8
Connections of the body with the electrocardiograph for record-
ing chest leads. LA, left arm; RA, right arm.
Figure 11-10
Normal electrocardiograms recorded from the three augmented
change the patterns of the electrocardiograms
unipolar limb leads.
markedly in some leads yet may not affect other leads.
Electrocardiographic interpretation of these two
types of conditions—cardiac myopathies and cardiac
electrical potential of the cardiac musculature imme-
arrhythmias—is discussed separately in Chapters 12
diately beneath the electrode. Therefore, relatively
and 13.
minute abnormalities in the ventricles, particularly in
the anterior ventricular wall, can cause marked
changes in the electrocardiograms recorded from indi-
Chest Leads (Precordial Leads)
vidual chest leads.
In leads V1 and V2, the QRS recordings of the
Often electrocardiograms are recorded with one elec-
normal heart are mainly negative because, as shown in
trode placed on the anterior surface of the chest
Figure 11-8, the chest electrode in these leads is nearer
directly over the heart at one of the points shown in
to the base of the heart than to the apex, and the base
Figure 11-8. This electrode is connected to the positive
of the heart is the direction of electronegativity during
terminal of the electrocardiograph, and the negative
most of the ventricular depolarization process. Con-
electrode, called the indifferent electrode, is connected
versely, the QRS complexes in leads V4, V5, and V6 are
through equal electrical resistances to the right arm,
mainly positive because the chest electrode in these
left arm, and left leg all at the same time, as also shown
leads is nearer the heart apex, which is the direction
in the figure. Usually six standard chest leads are
of electropositivity during most of depolarization.
recorded, one at a time, from the anterior chest wall,
the chest electrode being placed sequentially at the six
points shown in the diagram. The different recordings
Augmented Unipolar Limb Leads
are known as leads V1, V2, V3, V4, V5, and V6.
Figure 11-9 illustrates the electrocardiograms of the
Another system of leads in wide use is the augmented
healthy heart as recorded from these six standard
unipolar limb lead. In this type of recording, two of
chest leads. Because the heart surfaces are close
the limbs are connected through electrical resistances
to the chest wall, each chest lead records mainly the
to the negative terminal of the electrocardiograph,
130
Unit III
The Heart
and the third limb is connected to the positive termi-
recording from the aVR lead is inverted. (Why does
nal. When the positive terminal is on the right arm,
this inversion occur? Study the polarity connections to
the lead is known as the aVR lead; when on the left
the electrocardiograph to determine this.)
arm, the aVL lead; and when on the left leg, the aVF
lead.
Normal recordings of the augmented unipolar limb
References
leads are shown in Figure 11-10. They are all similar
to the standard limb lead recordings, except that the
See references for Chapter 13.
C H A P T E R
1
2
Electrocardiographic
Interpretation of Cardiac Muscle
and Coronary Blood Flow
Abnormalities: Vectorial Analysis
From the discussion in Chapter 10 of impulse trans-
mission through the heart, it is obvious that any
change in the pattern of this transmission can cause
abnormal electrical potentials around the heart and,
consequently, alter the shapes of the waves in the
electrocardiogram. For this reason, almost all
serious abnormalities of the heart muscle can be
diagnosed by analyzing the contours of the different
waves in the different electrocardiographic leads.
Principles of Vectorial Analysis
of Electrocardiograms
Use of Vectors to Represent Electrical Potentials
Before it is possible to understand how cardiac abnormalities affect the con-
tours of the electrocardiogram, one must first become thoroughly familiar with
the concept of vectors and vectorial analysis as applied to electrical potentials
in and around the heart.
Several times in Chapter 11 it was pointed out that heart current flows in a
particular direction in the heart at a given instant during the cardiac cycle. A
vector is an arrow that points in the direction of the electrical potential gener-
ated by the current flow, with the arrowhead in the positive direction. Also, by
convention, the length of the arrow is drawn proportional to the voltage of the
potential.
“Resultant” Vector in the Heart at Any Given Instant. Figure 12-1 shows, by the shaded
area and the negative signs, depolarization of the ventricular septum and parts
of the apical endocardial walls of the two ventricles. At this instant of heart exci-
tation, electrical current flows between the depolarized areas inside the heart
and the nondepolarized areas on the outside of the heart, as indicated by the
long elliptical arrows. Some current also flows inside the heart chambers directly
from the depolarized areas toward the still polarized areas. Overall, consider-
ably more current flows downward from the base of the ventricles toward the
apex than in the upward direction. Therefore, the summated vector of the gen-
erated potential at this particular instant, called the instantaneous mean vector,
is represented by the long black arrow drawn through the center of the ventri-
cles in a direction from base toward apex. Furthermore, because the summated
current is considerable in quantity, the potential is large, and the vector is long.
Direction of a Vector Is Denoted in Terms of Degrees
When a vector is exactly horizontal and directed toward the person’s left side,
the vector is said to extend in the direction of 0 degrees, as shown in Figure
131
132
Unit III
The Heart
+
+
+
-
+
+
+
-
aVF
-
- --
+
-
+
+
III
-
+
+
+
---
+
-
+
+
+
+
-
+
+
aVR
aVL
+
-
---
+
+
+
-
+
+
+
-
+
-
+
-
+
210∞
+
+
-
-30∞
+
-----
+
+
-
+
+
+++
+
- -
+
I
0∞
I
+
+
+
-
+
++
+
++
++
+ + + +
aVL
aVR
-
60∞
-
III
II
120∞
90∞
Figure 12-1
+
+
+
Mean vector through the partially depolarized ventricles.
Figure 12-3
Axes of the three bipolar and three unipolar leads.
-90∞
+270∞
through the center of Figure 12-2 in the +59-degree
direction. This means that during most of the depolar-
ization wave, the apex of the heart remains positive
-100∞
with respect to the base of the heart, as discussed later
in the chapter.
180∞
0∞
Axis for Each Standard Bipolar Lead
A
and Each Unipolar Limb Lead
In Chapter 11, the three standard bipolar and the three
120∞
59∞
unipolar limb leads are described. Each lead is actu-
ally a pair of electrodes connected to the body on
opposite sides of the heart, and the direction from neg-
-90∞
ative electrode to positive electrode is called the “axis”
of the lead. Lead I is recorded from two electrodes
placed respectively on the two arms. Because the elec-
trodes lie exactly in the horizontal direction, with
Figure 12-2
the positive electrode to the left, the axis of lead I is
Vectors drawn to represent potentials for several different hearts,
0 degrees.
and the “axis” of the potential (expressed in degrees) for each
In recording lead II, electrodes are placed on the
heart.
right arm and left leg. The right arm connects to the
torso in the upper right-hand corner and the left leg
connects in the lower left-hand corner. Therefore, the
12-2. From this zero reference point, the scale of
direction of this lead is about +60 degrees.
vectors rotates clockwise: when the vector extends
By similar analysis, it can be seen that lead III has
from above and straight downward, it has a direction
an axis of about +120 degrees; lead aVR, +210 degrees;
of +90 degrees; when it extends from the person’s left
aVF, +90 degrees; and aVL -30 degrees. The directions
to right, it has a direction of +180 degrees; and when
of the axes of all these leads are shown in Figure 12-3,
it extends straight upward, it has a direction of -90 (or
which is known as the hexagonal reference system. The
+270) degrees.
polarities of the electrodes are shown by the plus and
In a normal heart, the average direction of the
minus signs in the figure. The reader must learn these
vector during spread of the depolarization wave
axes and their polarities, particularly for the bipolar
through the ventricles, called the mean QRS vector, is
limb leads I, II, and III, to understand the remainder of
about +59 degrees, which is shown by vector A drawn
this chapter.
Chapter 12
Electrocardiographic Interpretation of Cardiac Muscle and Coronary Blood Flow Abnormalities
133
Vectorial Analysis of Potentials
in the electrocardiogram), and the voltage recorded
will be slight, about -0.3 millivolts. This figure demon-
Recorded in Different Leads
strates that when the vector in the heart is in a direc-
tion almost perpendicular to the axis of the lead, the
Now that we have discussed, first, the conventions for
voltage recorded in the electrocardiogram of this lead
representing potentials across the heart by means of
is very low. Conversely, when the heart vector has
vectors and, second, the axes of the leads, it is possible
almost exactly the same axis as the lead axis, essentially
to use these together to determine the instantaneous
the entire voltage of the vector will be recorded.
potential that will be recorded in the electrocardio-
gram of each lead for a given vector in the heart, as
Vectorial Analysis of Potentials in the Three Standard
follows.
Bipolar Limb Leads. In Figure 12-6, vector A depicts the
Figure 12-4 shows a partially depolarized heart;
vector A represents the instantaneous mean direction
of current flow in the ventricles. In this instance, the
direction of the vector is +55 degrees, and the voltage
of the potential, represented by the length of vector A,
is 2 millivolts. In the diagram below the heart, vector
A is shown again, and a line is drawn to represent the
axis of lead I in the 0-degree direction. To determine
how much of the voltage in vector A will be recorded
in lead I, a line perpendicular to the axis of lead I is
drawn from the tip of vector A to the lead I axis, and
a so-called projected vector (B) is drawn along the lead
I axis. The arrow of this projected vector points toward
B
I
- I
+
the positive end of the lead I axis, which means that
the record momentarily being recorded in the electro-
A
cardiogram of lead I is positive. And the instantaneous
recorded voltage will be equal to the length of B
divided by the length of A times 2 millivolts, or about
1 millivolt.
Figure 12-5 shows another example of vectorial
analysis. In this example, vector A represents the elec-
Figure 12-5
trical potential and its axis at a given instant during
Determination of the projected vector B along the axis of lead I
ventricular depolarization in a heart in which the left
when vector A represents the instantaneous potential in the
side of the heart depolarizes more rapidly than the
ventricles.
right. In this instance, the instantaneous vector has a
direction of 100 degrees, and its voltage is again 2 mil-
livolts. To determine the potential actually recorded in
lead I, we draw a perpendicular line from the tip of
vector A to the lead I axis and find projected vector B.
Vector B is very short and this time in the negative
-
-
direction, indicating that at this particular instant, the
II
III
recording in lead I will be negative (below the zero line
I
B
I
A
-
+
D
A
C
B
I
- I
+
III
II
+
+
A
Figure 12-4
Figure 12-6
Determination of a projected vector B along the axis of lead I when
Determination of projected vectors in leads I, II, and III when
vector A represents the instantaneous potential in the ventricles.
vector A represents the instantaneous potential in the ventricles.
134
Unit III
The Heart
instantaneous electrical potential of a partially depo-
understood. Each of these analyses should be studied
larized heart. To determine the potential recorded at
in detail by the procedure given here. A short
this instant in the electrocardiogram for each one of
summary of this sequence follows.
the three standard bipolar limb leads, perpendicular
In Figure 12-7A, the ventricular muscle has just
lines
(the dashed lines) are drawn from the tip of
begun to be depolarized, representing an instant about
vector A to the three lines representing the axes of the
0.01 second after the onset of depolarization. At this
three different standard leads, as shown in the figure.
time, the vector is short because only a small portion
The projected vector B depicts the potential recorded
of the ventricles—the septum—is depolarized. There-
at that instant in lead I, projected vector C depicts the
fore, all electrocardiographic voltages are low, as
potential in lead II, and projected vector D depicts the
recorded to the right of the ventricular muscle for each
potential in lead III. In each of these, the record in
of the leads. The voltage in lead II is greater than the
the electrocardiogram is positive—that is, above the
voltages in leads I and III because the heart vector
zero line—because the projected vectors point in the
extends mainly in the same direction as the axis of lead
positive directions along the axes of all the leads.
II.
The potential in lead I (vector B) is about one half that
In Figure 12-7B, which represents about 0.02 second
of the actual potential in the heart (vector A); in lead
after onset of depolarization, the heart vector is long
II (vector C), it is almost equal to that in the heart; and
because much of the ventricular muscle mass has
in lead III (vector D), it is about one third that in the
become depolarized. Therefore, the voltages in all
heart.
electrocardiographic leads have increased.
An identical analysis can be used to determine
In Figure 12-7C, about 0.035 second after onset of
potentials recorded in augmented limb leads, except
depolarization, the heart vector is becoming shorter
that the respective axes of the augmented leads (see
and the recorded electrocardiographic voltages are
Figure 12-3) are used in place of the standard bipolar
lower because the outside of the heart apex is now
limb lead axes used for Figure 12-6.
electronegative, neutralizing much of the positivity on
the other epicardial surfaces of the heart. Also, the axis
of the vector is beginning to shift toward the left side
Vectorial Analysis of the
of the chest because the left ventricle is slightly slower
to depolarize than the right. Therefore, the ratio of the
Normal Electrocardiogram
voltage in lead I to that in lead III is increasing.
In Figure 12-7D, about 0.05 second after onset of
Vectors That Occur at Successive
depolarization, the heart vector points toward the base
Intervals During Depolarization of the
of the left ventricle, and it is short because only a
Ventricles—The QRS Complex
minute portion of the ventricular muscle is still polar-
ized positive. Because of the direction of the vector at
When the cardiac impulse enters the ventricles
this time, the voltages recorded in leads II and III are
through the atrioventricular bundle, the first part of
both negative—that is, below the line—whereas the
the ventricles to become depolarized is the left endo-
voltage of lead I is still positive.
cardial surface of the septum. Then depolarization
In Figure 12-7E, about 0.06 second after onset of
spreads rapidly to involve both endocardial surfaces of
depolarization, the entire ventricular muscle mass is
the septum, as demonstrated by the shaded portion
depolarized, so that no current flows around the heart
of the ventricle in Figure 12-7A. Next, depolarization
and no electrical potential is generated. The vector
spreads along the endocardial surfaces of the remain-
becomes zero, and the voltages in all leads become
der of the two ventricles, as shown in Figure 12-7B and
zero.
C. Finally, it spreads through the ventricular muscle to
Thus, the QRS complexes are completed in the
the outside of the heart, as shown progressively in
three standard bipolar limb leads.
Figure 12-7C, D, and E.
Sometimes the QRS complex has a slight negative
At each stage in Figure 12-7, parts A to E, the
depression at its beginning in one or more of the leads,
instantaneous mean electrical potential of the ventri-
which is not shown in Figure 12-7; this depression is
cles is represented by a red vector superimposed on
the Q wave. When it occurs, it is caused by initial depo-
the ventricle in each figure. Each of these vectors is
larization of the left side of the septum before the right
then analyzed by the method described in the preced-
side, which creates a weak vector from left to right for
ing section to determine the voltages that will be
a fraction of a second before the usual base-to-apex
recorded at each instant in each of the three standard
vector occurs. The major positive deflection shown in
electrocardiographic leads. To the right in each figure
Figure 12-7 is the R wave, and the final negative
is shown progressive development of the electrocar-
deflection is the S wave.
diographic QRS complex. Keep in mind that a positive
vector in a lead will cause recording in the electrocar-
diogram above the zero line, whereas a negative vector
Electrocardiogram During
will cause recording below the zero line.
Repolarization—The T Wave
Before proceeding with further consideration of
vectorial analysis, it is essential that this analysis of the
After the ventricular muscle has become depolarized,
successive normal vectors presented in Figure 12-7 be
about 0.15 second later, repolarization begins and
Chapter 12
Electrocardiographic Interpretation of Cardiac Muscle and Coronary Blood Flow Abnormalities
135
−
−
−
−
II
III
II
III
I
I
I
I
I
I
−
+
−
+
II
II
III
II
III
II
+
+
+
+
III
III
A
B
−
−
−
−
II
III
II
III
I
I
I
I
I
I
−
+
−
+
II
II
III
II
III
II
+
+
+
+
III
III
C
D
−
−
II
III
I
I
I
−
+
II
III
II
+
+
III
E
Figure 12-7
Shaded areas of the ventricles are depolarized (-); nonshaded areas are still polarized (+). The ventricular vectors and QRS complexes
0.01 second after onset of ventricular depolarization (A); 0.02 second after onset of depolarization (B); 0.035 second after onset of
depolarization (C); 0.05 second after onset of depolarization (D); and after depolarization of the ventricles is complete, 0.06 second after
onset (E).
proceeds until complete at about 0.35 second.This repo-
flow to the endocardium, thereby slowing repolariza-
larization causes the T wave in the electrocardiogram.
tion in the endocardial areas.
Because the septum and endocardial areas of the
Because the outer apical surfaces of the ventricles
ventricular muscle depolarize first, it seems logical that
repolarize before the inner surfaces, the positive end
these areas should repolarize first as well. However,
of the overall ventricular vector during repolarization
this is not the usual case because the septum and other
is toward the apex of the heart. As a result, the normal
endocardial areas have a longer period of contraction
T wave in all three bipolar limb leads is positive, which
than most of the external surfaces of the heart. There-
is also the polarity of most of the normal QRS complex.
fore, the greatest portion of ventricular muscle mass to
In Figure 12-8, five stages of repolarization of the
repolarize first is the entire outer surface of the ventri-
ventricles are denoted by progressive increase of the
cles, especially near the apex of the heart. The endocar-
white areas—the repolarized areas. At each stage,
dial areas, conversely, normally repolarize last. This
the vector extends from the base of the heart toward
sequence of repolarization is postulated to be caused
the apex until it disappears in the last stage. At
by the high blood pressure inside the ventricles during
first, the vector is relatively small because the area of
contraction, which greatly reduces coronary blood
repolarization is small. Later, the vector becomes
136
Unit III
The Heart
+
+
+
P T
+
+
+
---
+
+
+
-
-
I
+
++
-
-
+
++
-
-
+
+
-
-
+
+
+
II
III
+
+
SA
+ +
-
-
II
II
III
I
I
-
+
I
I
III
III
II
-
+
+
+
I
III
II
+
+
II
Figure 12-9
III
Depolarization of the atria and generation of the P wave, showing
the maximum vector through the atria and the resultant vectors in
Figure 12-8
the three standard leads. At the right are the atrial P and T waves.
SA, sinoatrial node.
Generation of the T wave during repolarization of the ventricles,
showing also vectorial analysis of the first stage of repolarization.
The total time from the beginning of the T wave to its end is
approximately 0.15 second.
Repolarization of the Atria—The Atrial T Wave. Spread of
depolarization through the atrial muscle is much
slower than in the ventricles because the atria have no
Purkinje system for fast conduction of the depolariza-
stronger because of greater degrees of repolarization.
tion signal. Therefore, the musculature around the
Finally, the vector becomes weaker again because the
sinus node becomes depolarized a long time before the
areas of depolarization still persisting become so slight
musculature in distal parts of the atria. Because of this,
that the total quantity of current flow decreases. These
the area in the atria that also becomes repolarized first
changes also demonstrate that the vector is greatest
is the sinus nodal region, the area that had originally
when about half the heart is in the polarized state and
become depolarized first. Thus, when repolarization
about half is depolarized.
begins, the region around the sinus node becomes pos-
The changes in the electrocardiograms of the three
itive with respect to the rest of the atria. Therefore, the
standard limb leads during repolarization are noted
atrial repolarization vector is backward to the vector of
under each of the ventricles, depicting the progressive
depolarization. (Note that this is opposite to the effect
stages of repolarization. Thus, over about 0.15 second,
that occurs in the ventricles.) Therefore, as shown to
the period of time required for the whole process to
the right in Figure 12-9, the so-called atrial T wave
take place, the T wave of the electrocardiogram is
follows about 0.15 second after the atrial P wave, but
generated.
this T wave is on the opposite side of the zero refer-
ence line from the P wave; that is, it is normally nega-
tive rather than positive in the three standard bipolar
Depolarization of the Atria—
limb leads.
The P Wave
In the normal electrocardiogram, the atrial T wave
appears at about the same time that the QRS complex
Depolarization of the atria begins in the sinus node
of the ventricles appears. Therefore, it is almost always
and spreads in all directions over the atria. Therefore,
totally obscured by the large ventricular QRS
the point of original electronegativity in the atria is
complex, although in some very abnormal states, it
about at the point of entry of the superior vena cava
does appear in the recorded electrocardiogram.
where the sinus node lies, and the direction of initial
depolarization is denoted by the black vector in Figure
12-9. Furthermore, the vector remains generally in this
Vectorcardiogram
direction throughout the process of normal atrial
depolarization. Because this direction is generally
It has been noted in the discussion up to this point that
in the positive directions of the axes of the three
the vector of current flow through the heart changes
standard bipolar limb leads I, II, and III, the electro-
rapidly as the impulse spreads through the
cardiograms recorded from the atria during depolar-
myocardium. It changes in two aspects: First, the
ization are also usually positive in all three of these
vector increases and decreases in length because of
leads, as shown in Figure 12-9. This record of atrial
increasing and decreasing voltage of the vector.
depolarization is known as the atrial P wave.
Second, the vector changes direction because of
Chapter 12
Electrocardiographic Interpretation of Cardiac Muscle and Coronary Blood Flow Abnormalities
137
III
-
-60∞
1
2
3
4
5
I
4
5
-
+
I
I
II
1
180∞
0∞
3
III
120∞
59∞
Depolarization
2
Repolarization
+
QRS
T
III
Figure 12-10
QRS and T vectorcardiograms.
Figure 12-11
Plotting the mean electrical axis of the ventricles from two elec-
trocardiographic leads (leads I and III).
changes in the average direction of the electrical
potential from the heart. The so-called vectorcardio-
Mean Electrical Axis
gram depicts these changes at different times during
of the Ventricular QRS—
the cardiac cycle, as shown in Figure 12-10.
In the large vectorcardiogram of Figure 12-10, point
And Its Significance
5 is the zero reference point, and this point is the neg-
ative end of all the successive vectors. While the heart
The vectorcardiogram during ventricular depolariza-
muscle is polarized between heartbeats, the positive
tion
(the QRS vectorcardiogram) shown in Figure
end of the vector remains at the zero point because
12-10 is that of a normal heart. Note from this vec-
there is no vectorial electrical potential. However,
torcardiogram that the preponderant direction of the
as soon as current begins to flow through the ventri-
vectors of the ventricles during depolarization is
cles at the beginning of ventricular depolarization, the
mainly toward the apex of the heart. That is, during
positive end of the vector leaves the zero reference
most of the cycle of ventricular depolarization, the
point.
direction of the electrical potential (negative to posi-
When the septum first becomes depolarized, the
tive) is from the base of the ventricles toward the apex.
vector extends downward toward the apex of the ven-
This preponderant direction of the potential during
tricles, but it is relatively weak, thus generating the first
depolarization is called the mean electrical axis of the
portion of the ventricular vectorcardiogram, as shown
ventricles. The mean electrical axis of the normal
by the positive end of vector
1. As more of the
ventricles is 59 degrees. In many pathological condi-
ventricular muscle becomes depolarized, the vector
tions of the heart, this direction changes markedly—
becomes stronger and stronger, usually swinging
sometimes even to opposite poles of the heart.
slightly to one side. Thus, vector 2 of Figure 12-10 rep-
resents the state of depolarization of the ventricles
about 0.02 second after vector 1. After another 0.02
Determining the Electrical Axis from
second, vector 3 represents the potential, and vector 4
Standard Lead Electrocardiograms
occurs in another 0.01 second. Finally, the ventricles
become totally depolarized, and the vector becomes
Clinically, the electrical axis of the heart usually is esti-
zero once again, as shown at point 5.
mated from the standard bipolar limb lead electrocar-
The elliptical figure generated by the positive ends
diograms rather than from the vectorcardiogram.
of the vectors is called the QRS vectorcardiogram.
Figure 12-11 shows a method for doing this. After
Vectorcardiograms can be recorded on an oscilloscope
recording the standard leads, one determines the net
by connecting body surface electrodes from the neck
potential and polarity of the recordings in leads I and
and lower abdomen to the vertical plates of the oscil-
III. In lead I of Figure 12-11, the recording is positive,
loscope and connecting chest surface electrodes from
and in lead III, the recording is mainly positive but
each side of the heart to the horizontal plates. When
negative during part of the cycle. If any part of a
the vector changes, the spot of light on the oscilloscope
recording is negative, this negative potential is sub-
follows the course of the positive end of the changing
tracted from the positive part of the potential to deter-
vector, thus inscribing the vectorcardiogram on the
mine the net potential for that lead, as shown by the
oscilloscopic screen.
arrow to the right of the QRS complex for lead III.
138
Unit III
The Heart
Then each net potential for leads I and III is plotted
side of the heart than on the other side, and this allows
on the axes of the respective leads, with the base of the
excess generation of electrical potential on that side.
potential at the point of intersection of the axes, as
Second, more time is required for the depolarization
shown in Figure 12-11.
wave to travel through the hypertrophied ventricle
If the net potential of lead I is positive, it is plotted
than through the normal ventricle. Consequently, the
in a positive direction along the line depicting lead I.
normal ventricle becomes depolarized considerably in
Conversely, if this potential is negative, it is plotted in
advance of the hypertrophied ventricle, and this causes
a negative direction. Also, for lead III, the net poten-
a strong vector from the normal side of the heart
tial is placed with its base at the point of intersection,
toward the hypertrophied side, which remains strongly
and, if positive, it is plotted in the positive direction
positively charged. Thus, the axis deviates toward the
along the line depicting lead III. If it is negative, it is
hypertrophied ventricle.
plotted in the negative direction.
To determine the vector of the total QRS ventricu-
Vectorial Analysis of Left Axis Deviation Resulting from
lar mean electrical potential, one draws perpendicular
Hypertrophy of the Left Ventricle. Figure 12-12 shows
lines (the dashed lines in the figure) from the apices of
the three standard bipolar limb lead electrocardio-
leads I and III, respectively. The point of intersection
grams. Vectorial analysis demonstrates left axis devia-
of these two perpendicular lines represents, by vecto-
tion with mean electrical axis pointing in the
rial analysis, the apex of the mean QRS vector in the
-15-degree direction. This is a typical electrocardio-
ventricles, and the point of intersection of the lead I
gram caused by increased muscle mass of the left ven-
and lead III axes represents the negative end of the
tricle. In this instance, the axis deviation was caused by
mean vector. Therefore, the mean QRS vector is drawn
hypertension
(high arterial blood pressure), which
between these two points. The approximate average
caused the left ventricle to hypertrophy so that it could
potential generated by the ventricles during depolar-
pump blood against elevated systemic arterial pres-
ization is represented by the length of this mean QRS
sure. A similar picture of left axis deviation occurs
vector, and the mean electrical axis is represented by
when the left ventricle hypertrophies as a result of
the direction of the mean vector. Thus, the orientation
aortic valvular stenosis, aortic valvular regurgitation, or
of the mean electrical axis of the normal ventricles,
any number of congenital heart conditions in which the
as determined in Figure 12-11, is 59 degrees positive
left ventricle enlarges while the right ventricle remains
(+59 degrees).
relatively normal in size.
Vectorial Analysis of Right Axis Deviation Resulting
Abnormal Ventricular Conditions That
from Hypertrophy of the Right Ventricle. The electro-
Cause Axis Deviation
cardiogram of Figure 12-13 shows intense right axis
deviation, to an electrical axis of 170 degrees, which is
Although the mean electrical axis of the ventricles
averages about 59 degrees, this axis can swing even
in the normal heart from about 20 degrees to about
100 degrees. The causes of the normal variations are
mainly anatomical differences in the Purkinje distri-
bution system or in the musculature itself of different
hearts. However, a number of abnormal conditions of
the heart can cause axis deviation beyond the normal
limits, as follows.
Change in the Position of the Heart in the Chest. If the heart
I
II
III
itself is angulated to the left, the mean electrical axis
III
of the heart also shifts to the left. Such shift occurs (1)
-
at the end of deep expiration, (2) when a person lies
down, because the abdominal contents press upward
against the diaphragm, and (3) quite frequently in
stocky, fat people whose diaphragms normally press
upward against the heart all the time.
I
-
+
I
Likewise, angulation of the heart to the right causes
the mean electrical axis of the ventricles to shift to the
right. This occurs (1) at the end of deep inspiration,
+
(2) when a person stands up, and (3) normally in tall,
III
lanky people whose hearts hang downward.
Hypertrophy of One Ventricle. When one ventricle greatly
Figure 12-12
hypertrophies, the axis of the heart shifts toward the
hypertrophied ventricle for two reasons. First, a far
Left axis deviation in a hypertensive heart (hypertrophic left ven-
greater quantity of muscle exists on the hypertrophied
tricle). Note the slightly prolonged QRS complex as well.
Chapter 12
Electrocardiographic Interpretation of Cardiac Muscle and Coronary Blood Flow Abnormalities
139
I
II
III
III
I
II
III
-
III
-
I
-
+
I
I
-
+
I
+
III
+
III
Figure 12-14
Left axis deviation caused by left bundle branch block. Note also
Figure 12-13
the greatly prolonged QRS complex.
High-voltage electrocardiogram in congenital pulmonary valve
stenosis with right ventricular hypertrophy. Intense right axis devi-
blocked, cardiac depolarization spreads through the
ation and a slightly prolonged QRS complex also are seen.
right ventricle two to three times as rapidly as through
the left ventricle. Consequently, much of the left ven-
111 degrees to the right of the normal 59-degree mean
tricle remains polarized for as long as 0.1 second after
ventricular QRS axis. The right axis deviation demon-
the right ventricle has become totally depolarized.
strated in this figure was caused by hypertrophy of the
Thus, the right ventricle becomes electronegative,
right ventricle as a result of congenital pulmonary
whereas the left ventricle remains electropositive
valve stenosis. Right axis deviation also can occur in
during most of the depolarization process, and a strong
other congenital heart conditions that cause hypertro-
vector projects from the right ventricle toward the left
phy of the right ventricle, such as tetralogy of Fallot and
ventricle. In other words, there is intense left axis devi-
interventricular septal defect.
ation of about -50 degrees because the positive end of
the vector points toward the left ventricle. This is
Bundle Branch Block Causes Axis Deviation. Ordinarily, the
demonstrated in Figure 12-14, which shows typical left
lateral walls of the two ventricles depolarize at almost
axis deviation resulting from left bundle branch block.
the same instant because both the left and the right
Because of slowness of impulse conduction when
bundle branches of the Purkinje system transmit the
the Purkinje system is blocked, in addition to axis devi-
cardiac impulse to the two ventricular walls at almost
ation, the duration of the QRS complex is greatly pro-
the same instant. As a result, the potentials generated
longed because of extreme slowness of depolarization
by the two ventricles (on the two opposite sides of the
in the affected side of the heart. One can see this by
heart) almost neutralize each other. But if only one of
observing the excessive widths of the QRS waves in
the major bundle branches is blocked, the cardiac
Figure 12-14. This is discussed in greater detail later in
impulse spreads through the normal ventricle long
the chapter. This extremely prolonged QRS complex
before it spreads through the other. Therefore, depo-
differentiates bundle branch block from axis deviation
larization of the two ventricles does not occur even
caused by hypertrophy.
nearly simultaneously, and the depolarization poten-
tials do not neutralize each other. As a result, axis devi-
Vectorial Analysis of Right Axis Deviation in Right
ation occurs as follows.
Bundle Branch Block. When the right bundle branch
is blocked, the left ventricle depolarizes far more
Vectorial Analysis of Left Axis Deviation in Left Bundle
rapidly than the right ventricle, so that the left side of
Branch Block. When the left bundle branch is
the ventricles becomes electronegative as long as
140
Unit III
The Heart
0.1 second before the right. Therefore, a strong vector
the electrical potentials recorded in the electrocardio-
develops, with its negative end toward the left ventri-
graphic leads are considerably greater than normal, as
cle and its positive end toward the right ventricle. In
shown in Figures 12-12 and 12-13.
other words, intense right axis deviation occurs. Right
axis deviation caused by right bundle branch block is
demonstrated, and its vector is analyzed, in Figure
Decreased Voltage of the
12-15, which shows an axis of about 105 degrees
Electrocardiogram
instead of the normal 59 degrees and a prolonged QRS
complex because of slow conduction.
Decreased Voltage Caused by Cardiac Myopathies. One of
the most common causes of decreased voltage of the
QRS complex is a series of old myocardial artery
Conditions That Cause
infarctions with resultant diminished muscle mass. This
Abnormal Voltages of the QRS
also causes the depolarization wave to move through
the ventricles slowly and prevents major portions of
Complex
the heart from becoming massively depolarized all at
once. Consequently, this condition causes some pro-
Increased Voltage in the Standard
longation of the QRS complex along with the
Bipolar Limb Leads
decreased voltage. Figure 12-16 shows a typical low-
voltage electrocardiogram with prolongation of the
Normally, the voltages in the three standard bipolar
QRS complex, which is common after multiple small
limb leads, as measured from the peak of the R wave
infarctions of the heart have caused local delays of
to the bottom of the S wave, vary between 0.5 and 2.0
impulse conduction and reduced voltages due to loss
millivolts, with lead III usually recording the lowest
of muscle mass throughout the ventricles.
voltage and lead II the highest. However, these rela-
tions are not invariable, even for the normal heart. In
Decreased Voltage Caused by Conditions Surrounding the
general, when the sum of the voltages of all the QRS
Heart. One of the most important causes of decreased
complexes of the three standard leads is greater than
voltage in electrocardiographic leads is fluid in the
4 millivolts, the patient is considered to have a high-
pericardium. Because extracellular fluid conducts elec-
voltage electrocardiogram.
trical currents with great ease, a large portion of the
The cause of high-voltage QRS complexes most
electricity flowing out of the heart is conducted from
often is increased muscle mass of the heart, which
one part of the heart to another through the pericar-
ordinarily results from hypertrophy of the muscle in
dial fluid. Thus, this effusion effectively “short-circuits”
response to excessive load on one part of the heart or
the electrical potentials generated by the heart,
the other. For example, the right ventricle hypertro-
decreasing the electrocardiographic voltages that
phies when it must pump blood through a stenotic pul-
reach the outside surfaces of the body. Pleural effu-
monary valve, and the left ventricle hypertrophies
sion, to a lesser extent, also can “short-circuit” the
when a person has high blood pressure. The increased
electricity around the heart, so that the voltages at the
quantity of muscle causes generation of increased
quantities of electricity around the heart. As a result,
III
I
-
I
II
I-
+I
II
III
+
III
III
Figure 12-15
Figure 12-16
Right axis deviation caused by right bundle branch block. Note
Low-voltage electrocardiogram following local damage through-
also the greatly prolonged QRS complex.
out the ventricles caused by previous myocardial infarction.
Chapter 12
Electrocardiographic Interpretation of Cardiac Muscle and Coronary Blood Flow Abnormalities
141
surface of the body and in the electrocardiograms are
ventricular system, with replacement of this muscle by
decreased.
scar tissue, and (2) multiple small local blocks in the
Pulmonary emphysema can decrease the electrocar-
conduction of impulses at many points in the Purkinje
diographic potentials, but by a different method from
system. As a result, cardiac impulse conduction
that of pericardial effusion. In pulmonary emphysema,
becomes irregular, causing rapid shifts in voltages and
conduction of electrical current through the lungs is
axis deviations. This often causes double or even triple
depressed considerably because of excessive quantity
peaks in some of the electrocardiographic leads, such
of air in the lungs. Also, the chest cavity enlarges, and
as those shown in Figure 12-14.
the lungs tend to envelop the heart to a greater extent
than normally. Therefore, the lungs act as an insulator
to prevent spread of electrical voltage from the heart
Current of Injury
to the surface of the body, and this results in decreased
electrocardiographic potentials in the various leads.
Many different cardiac abnormalities, especially those
that damage the heart muscle itself, often cause part
of the heart to remain partially or totally depolarized
all the time. When this occurs, current flows between
Prolonged and Bizarre
the pathologically depolarized and the normally polar-
Patterns of the QRS Complex
ized areas even between heartbeats. This is called a
current of injury. Note especially that the injured part
Prolonged QRS Complex as a Result
of the heart is negative, because this is the part that is
of Cardiac Hypertrophy or Dilatation
depolarized and emits negative charges into the sur-
rounding fluids, whereas the remainder of the heart is
The QRS complex lasts as long as depolarization con-
neutral or positive polarity.
tinues to spread through the ventricles—that is, as long
Some abnormalities that can cause current of injury
as part of the ventricles is depolarized and part is
are (1) mechanical trauma, which sometimes makes
still polarized. Therefore, prolonged conduction of the
the membranes remain so permeable that full repo-
impulse through the ventricles always causes a pro-
larization cannot take place; (2) infectious processes
longed QRS complex. Such prolongation often occurs
that damage the muscle membranes; and (3) ischemia
when one or both ventricles are hypertrophied or
of local areas of heart muscle caused by local coronary
dilated, owing to the longer pathway that the impulse
occlusions, which is by far the most common cause of
must then travel. The normal QRS complex lasts 0.06
current of injury in the heart. During ischemia, not
to 0.08 second, whereas in hypertrophy or dilatation of
enough nutrients from the coronary blood supply are
the left or right ventricle, the QRS complex may be
available to the heart muscle to maintain normal mem-
prolonged to 0.09 to 0.12 second.
brane polarization.
Prolonged QRS Complex Resulting
Effect of Current of Injury on the
from Purkinje System Blocks
QRS Complex
When the Purkinje fibers are blocked, the cardiac
In Figure 12-17, a small area in the base of the left ven-
impulse must then be conducted by the ventricular
tricle is newly infarcted (loss of coronary blood flow).
muscle instead of by way of the Purkinje system. This
Therefore, during the T-P interval—that is, when the
decreases the velocity of impulse conduction to about
normal ventricular muscle is totally polarized—abnor-
one third of normal. Therefore, if complete block of
mal negative current still flows from the infarcted area
one of the bundle branches occurs, the duration of the
at the base of the left ventricle and spreads toward the
QRS complex usually is increased to 0.14 second or
rest of the ventricles. The vector of this “current of
greater.
injury,” as shown in the first heart in the figure, is in a
In general, a QRS complex is considered to be
direction of about 125 degrees, with the base of the
abnormally long when it lasts more than 0.09 second;
vector, the negative end, toward the injured muscle. As
when it lasts more than 0.12 second, the prolongation
shown in the lower portions of the figure, even before
is almost certainly caused by pathological block some-
the QRS complex begins, this vector causes an initial
where in the ventricular conduction system, as shown
record in lead I below the zero potential line, because
by the electrocardiograms for bundle branch block in
the projected vector of the current of injury in lead I
Figures 12-14 and 12-15.
points toward the negative end of the lead I axis. In
lead II, the record is above the line because the pro-
jected vector points more toward the positive terminal
Conditions That Cause Bizarre QRS
of the lead. In lead III, the projected vector points in
Complexes
the same direction as the positive terminal of lead III,
so that the record is positive. Furthermore, because the
Bizarre patterns of the QRS complex most frequently
vector lies almost exactly in the direction of the axis
are caused by two conditions:
(1) destruction of
of lead III, the voltage of the current of injury in lead
cardiac muscle in various areas throughout the
III is much greater than in either lead I or lead II.
142
Unit III
The Heart
Injured area
-
-
II
III
I
I
-
+
III
II
+
+
I
J
II
Current
Current
of injury
of injury
J
III
Figure 12-17
Effect of a current of injury on
J
the electrocardiogram.
As the heart then proceeds through its normal
heart. However, many stray currents exist in the body,
process of depolarization, the septum first becomes
such as currents resulting from “skin potentials” and
depolarized; then the depolarization spreads down to
from differences in ionic concentrations in different
the apex and back toward the bases of the ventricles.
fluids of the body. Therefore, when two electrodes are
The last portion of the ventricles to become totally
connected between the arms or between an arm and a
depolarized is the base of the right ventricle, because
leg, these stray currents make it impossible for one to
the base of the left ventricle is already totally and per-
predetermine the exact zero reference level in the
manently depolarized. By vectorial analysis, the suc-
electrocardiogram. For these reasons, the following
cessive stages of electrocardiogram generation by the
procedure must be used to determine the zero poten-
depolarization wave traveling through the ventricles
tial level: First, one notes the exact point at which the
can be constructed graphically, as demonstrated in the
wave of depolarization just completes its passage
lower part of Figure 12-17.
through the heart, which occurs at the end of the QRS
When the heart becomes totally depolarized, at the
complex. At exactly this point, all parts of the ventri-
end of the depolarization process (as noted by the
cles have become depolarized, including both the
next-to-last stage in Figure 12-17), all the ventricular
damaged parts and the normal parts, so that no current
muscle is in a negative state. Therefore, at this instant
is flowing around the heart. Even the current of injury
in the electrocardiogram, no current flows from the
disappears at this point. Therefore, the potential of the
ventricles to the electrocardiographic electrodes
electrocardiogram at this instant is at zero voltage. This
because now both the injured heart muscle and the
point is known as the “J point” in the electrocardio-
contracting muscle are depolarized.
gram, as shown in Figure 12-18.
Next, as repolarization takes place, all of the heart
Then, for analysis of the electrical axis of the injury
finally repolarizes, except the area of permanent depo-
potential caused by a current of injury, a horizontal
larization in the injured base of the left ventricle. Thus,
line is drawn in the electrocardiogram for each lead at
repolarization causes a return of the current of injury
the level of the J point. This horizontal line is then the
in each lead, as noted at the far right in Figure 12-17.
zero potential level in the electrocardiogram from
which all potentials caused by currents of injury must
be measured.
The J Point—The Zero Reference
Use of the J Point in Plotting Axis of Injury Potential. Figure
Potential for Analyzing Current
12-18 shows electrocardiograms (leads I and III) from
of Injury
an injured heart. Both records show injury potentials.
In other words, the J point of each of these two elec-
One would think that the electrocardiograph
trocardiograms is not on the same line as the T-P
machines for recording electrocardiograms could
segment. In the figure, a horizontal line has been
determine when no current is flowing around the
drawn through the J point to represent the zero
Chapter 12
Electrocardiographic Interpretation of Cardiac Muscle and Coronary Blood Flow Abnormalities
143
voltage level in each of the two recordings. The injury
Coronary Ischemia as a Cause
potential in each lead is the difference between the
of Injury Potential
voltage of the electrocardiogram immediately before
onset of the P wave and the zero voltage level deter-
Insufficient blood flow to the cardiac muscle depresses
mined from the J point. In lead I, the recorded voltage
the metabolism of the muscle for three reasons: (1)
of the injury potential is above the zero potential level
lack of oxygen, (2) excess accumulation of carbon
and is, therefore, positive. Conversely, in lead III, the
dioxide, and
(3) lack of sufficient food nutrients.
injury potential is below the zero voltage level and,
Consequently, repolarization of the muscle membrane
therefore, is negative.
cannot occur in areas of severe myocardial ischemia.
At the bottom in Figure 12-18, the respective injury
Often the heart muscle does not die because the blood
potentials in leads I and III are plotted on the coordi-
flow is sufficient to maintain life of the muscle even
nates of these leads, and the resultant vector of the
though it is not sufficient to cause repolarization of the
injury potential for the whole ventricular muscle mass
membranes. As long as this state exists, an injury
is determined by vectorial analysis as described. In this
potential continues to flow during the diastolic portion
instance, the resultant vector extends from the right
(the T-P portion) of each heart cycle.
side of the ventricles toward the left and slightly
Extreme ischemia of the cardiac muscle occurs after
upward, with an axis of about -30 degrees. If one
coronary occlusion, and a strong current of injury
places this vector for the injury potential directly over
flows from the infarcted area of the ventricles during
the ventricles, the negative end of the vector points
the T-P interval between heartbeats, as shown in
toward the permanently depolarized, “injured” area of
Figures 12-19 and 12-20. Therefore, one of the most
the ventricles. In the example shown in Figure 12-18,
important diagnostic features of electrocardiograms
the injured area would be in the lateral wall of the
recorded after acute coronary thrombosis is the
right ventricle.
current of injury.
This analysis is obviously complex. However, it is
essential that the student go over it again and
Acute Anterior Wall Infarction. Figure
12-19 shows the
again until he or she understands it thoroughly. No
electrocardiogram in the three standard bipolar limb
other aspect of electrocardiographic analysis is more
important.
I
±
0
0
“J” point
“J” point
III
0
0
±
I
II
III
III
-
III
-
I -
+
I
I
-
+
I
+
+
III
III
V2
Figure 12-18
Figure 12-19
J point as the zero reference potential of the electrocardiograms
for leads I and II. Also, the method for plotting the axis of the injury
Current of injury in acute anterior wall infarction. Note the intense
potential is shown by the lowermost panel.
injury potential in lead V2.
144
Unit III
The Heart
leads and in one chest lead (lead V2 ) recorded from a
also in the chest lead. If a zero potential reference line
patient with acute anterior wall cardiac infarction. The
is drawn through the J point of this lead, it is readily
most important diagnostic feature of this electrocar-
apparent that during the T-P interval, the potential of
diogram is the intense injury potential in chest lead V2.
the current of injury is positive. This means that the
If one draws a zero horizontal potential line through
positive end of the vector is in the direction of the
the J point of this electrocardiogram, a strong negative
anterior chest wall, and the negative end (injured end
injury potential during the T-P interval is found, which
of the vector) points away from the chest wall. In other
means that the chest electrode over the front of the
words, the current of injury is coming from the back
heart is in an area of strongly negative potential. In
of the heart opposite to the anterior chest wall, which
other words, the negative end of the injury potential
is the reason this type of electrocardiogram is the basis
vector in this heart is against the anterior chest wall.
for diagnosing posterior wall infarction.
This means that the current of injury is emanating
If one analyzes the injury potentials from leads II
from the anterior wall of the ventricles, which diag-
and III of Figure 12-20, it is readily apparent that the
noses this condition as anterior wall infarction.
injury potential is negative in both leads. By vectorial
Analyzing the injury potentials in leads I and III,
analysis, as shown in the figure, one finds that the
one finds a negative potential in lead I and a positive
resultant vector of the injury potential is about -95
potential in lead III. This means that the resultant
degrees, with the negative end pointing downward and
vector of the injury potential in the heart is about +150
the positive end pointing upward. Thus, because the
degrees, with the negative end pointing toward the left
infarct, as indicated by the chest lead, is on the poste-
ventricle and the positive end pointing toward the
rior wall of the heart and, as indicated by the injury
right ventricle. Thus, in this particular electrocardio-
potentials in leads II and III, is in the apical portion of
gram, the current of injury is coming mainly from the
the heart, one would suspect that this infarct is near
left ventricle as well as from the anterior wall of the
the apex on the posterior wall of the left ventricle.
heart. Therefore, one would conclude that this anterior
wall infarction almost certainly is caused by thrombo-
Infarction in Other Parts of the Heart. By the same proce-
sis of the anterior descending branch of the left coro-
dures demonstrated in the preceding discussions of
nary artery.
anterior and posterior wall infarctions, it is possible to
determine the locus of any infarcted area emitting a
Posterior Wall Infarction. Figure 12-20 shows the three
current of injury, regardless of which part of the heart
standard bipolar limb leads and one chest lead (lead
is involved. In making such vectorial analyses, it must
V2) from a patient with posterior wall infarction. The
be remembered that the positive end of the injury
major diagnostic feature of this electrocardiogram is
potential vector points toward the normal cardiac
muscle, and the negative end points toward the injured
portion of the heart that is emitting the current of injury.
Recovery from Acute Coronary Thrombosis. Figure
12-21
shows a V3 chest lead from a patient with acute pos-
terior wall infarction, demonstrating changes in the
electrocardiogram from the day of the attack to 1
I
II
III
V2
II
III
-
-
Same day
1 week
2 weeks
1 year
+
+
III
II
Figure 12-21
Recovery of the myocardium after moderate posterior wall infarc-
Figure 12-20
tion, demonstrating disappearance of the injury potential that is
present on the first day after the infarction and still slightly present
Injury potential in acute posterior wall, apical infarction.
at 1 week.
Chapter 12
Electrocardiographic Interpretation of Cardiac Muscle and Coronary Blood Flow Abnormalities
145
week later, 3 weeks later, and finally 1 year later.
Current of Injury in Angina Pectoris. “Angina pectoris”
From this electrocardiogram, one can see that the
means pain from the heart felt in the pectoral regions
injury potential is strong immediately after the
of the upper chest. This pain usually also radiates into
acute attack (T-P segment displaced positively from
the left neck area and down the left arm. The pain typ-
the S-T segment). However, after about 1 week, the
ically is caused by moderate ischemia of the heart.
injury potential has diminished considerably, and after
Usually, no pain is felt as long as the person is quiet,
3 weeks, it is gone. After that, the electrocardiogram
but as soon as he or she overworks the heart, the pain
does not change greatly during the next year. This
appears.
is the usual recovery pattern after acute cardiac
An injury potential sometimes appears in the elec-
infarction of moderate degree, showing that the new
trocardiogram during an attack of severe angina pec-
collateral coronary blood flow develops enough
toris, because the coronary insufficiency becomes great
to re-establish appropriate nutrition to most of the
enough to prevent adequate repolarization of some
infarcted area.
areas of the heart during diastole.
Conversely, in some patients with coronary infarc-
tion, the infarcted area never redevelops adequate
coronary blood supply. Often, some of the heart
Abnormalities in the T Wave
muscle dies, but if the muscle does not die, it will con-
tinue to show an injury potential as long as the
Earlier in the chapter, it was pointed out that the T
ischemia exists, particularly during bouts of exercise
wave is normally positive in all the standard bipolar
when the heart is overloaded.
limb leads and that this is caused by repolarization of
the apex and outer surfaces of the ventricles ahead
Old Recovered Myocardial Infarction. Figure 12-22 shows
of the intraventricular surfaces. That is, the T wave
leads I and III after anterior infarction and leads I and
becomes abnormal when the normal sequence of
III after posterior infarction about 1 year after the
repolarization does not occur. Several factors can
acute heart attack. The records show what might be
change this sequence of repolarization.
called the “ideal” configurations of the QRS complex
in these types of recovered myocardial infarction.
Usually a Q wave has developed at the beginning of
Effect of Slow Conduction of the
the QRS complex in lead I in anterior infarction
Depolarization Wave on the
because of loss of muscle mass in the anterior wall of
Characteristics of the T Wave
the left ventricle, but in posterior infarction, a Q wave
has developed at the beginning of the QRS complex
Referring back to Figure 12-14, note that the QRS
in lead III because of loss of muscle in the posterior
complex is considerably prolonged. The reason for this
apical part of the ventricle.
prolongation is delayed conduction in the left ventricle
These configurations are certainly not found in all
resulting from left bundle branch block. This causes
cases of old cardiac infarction. Local loss of muscle and
the left ventricle to become depolarized about 0.08
local points of cardiac signal conduction block can
second after depolarization of the right ventricle,
cause very bizarre QRS patterns (especially promi-
which gives a strong mean QRS vector to the left.
nent Q waves, for instance), decreased voltage, and
However, the refractory periods of the right and left
QRS prolongation.
ventricular muscle masses are not greatly different
from each other. Therefore, the right ventricle begins
to repolarize long before the left ventricle; this causes
strong positivity in the right ventricle and negativity in
the left ventricle at the time that the T wave is devel-
oping. In other words, the mean axis of the T wave is
now deviated to the right, which is opposite the mean
Anterior
Posterior
electrical axis of the QRS complex in the same elec-
trocardiogram. Thus, when conduction of the depolar-
ization impulse through the ventricles is greatly
delayed, the T wave is almost always of opposite polar-
ity to that of the QRS complex.
Q
Q
I
III
I
III
Shortened Depolarization in Portions
of the Ventricular Muscle as a Cause
of T Wave Abnormalities
Figure 12-22
If the base of the ventricles should exhibit an abnor-
Electrocardiograms of anterior and posterior wall infarctions that
mally short period of depolarization, that is, a short-
occurred about 1 year previously, showing a Q wave in lead I in
ened action potential, repolarization of the ventricles
anterior wall infarction and a Q wave in lead III in posterior wall
infarction.
would not begin at the apex as it normally does.
146
Unit III
The Heart
T
T
T
T
T
T
Figure 12-23
Inverted T wave resulting from mild ischemia at the apex of the
ventricles.
Figure 12-24
Instead, the base of the ventricles would repolarize
Biphasic T wave caused by digitalis toxicity.
ahead of the apex, and the vector of repolarization
would point from the apex toward the base of the
heart, opposite to the standard vector of repolariza-
inversion, for instance, or a biphasic wave—is often
tion. Consequently, the T wave in all three standard
evidence enough that some portion of the ventricular
leads would be negative rather than the usual positive.
muscle has a period of depolarization out of propor-
Thus, the simple fact that the base of the ventricles has
tion to the rest of the heart, caused by mild to moder-
a shortened period of depolarization is sufficient to
ate coronary insufficiency.
cause marked changes in the T wave, even to the
extent of changing the entire T wave polarity, as shown
Effect of Digitalis on the T Wave. As discussed in Chapter
in Figure 12-23.
22, digitalis is a drug that can be used during coronary
Mild ischemia is by far the most common cause of
insufficiency to increase the strength of cardiac muscle
shortening of depolarization of cardiac muscle,
contraction. But when overdosages of digitalis are
because this increases current flow through the potas-
given, depolarization duration in one part of the ven-
sium channels. When the ischemia occurs in only one
tricles may be increased out of proportion to that of
area of the heart, the depolarization period of this area
other parts. As a result, nonspecific changes, such as T
decreases out of proportion to that in other portions.
wave inversion or biphasic T waves, may occur in one
As a result, definite changes in the T wave can take
or more of the electrocardiographic leads. A biphasic
place. The ischemia might result from chronic, pro-
T wave caused by excessive administration of digitalis
gressive coronary occlusion; acute coronary occlusion;
is shown in Figure 12-24. Therefore, changes in the T
or relative coronary insufficiency that occurs during
wave during digitalis administration are often the ear-
exercise.
liest signs of digitalis toxicity.
One means for detecting mild coronary insufficiency
is to have the patient exercise and to record the elec-
trocardiogram, noting whether changes occur in the T
References
waves. The changes in the T waves need not be spe-
cific, because any change in the T wave in any lead—
See references for Chapter 13.
C H A P T E R
1
3
Cardiac Arrhythmias and
Their Electrocardiographic
Interpretation
Some of the most distressing types of heart malfunction
occur not as a result of abnormal heart muscle but
because of abnormal rhythm of the heart. For instance,
sometimes the beat of the atria is not coordinated with
the beat of the ventricles, so that the atria no longer
function as primer pumps for the ventricles.
The purpose of this chapter is to discuss the
physiology of common cardiac arrhythmias and their
effects on heart pumping, as well as their diagnosis by
electrocardiography. The causes of the cardiac arrhythmias are usually one or a com-
bination of the following abnormalities in the rhythmicity-conduction system of the
heart:
1. Abnormal rhythmicity of the pacemaker
2. Shift of the pacemaker from the sinus node to another place in the heart
3. Blocks at different points in the spread of the impulse through the heart
4. Abnormal pathways of impulse transmission through the heart
5. Spontaneous generation of spurious impulses in almost any part of the heart
Abnormal Sinus Rhythms
Tachycardia
The term “tachycardia” means fast heart rate, usually defined in an adult person as
faster than 100 beats per minute. An electrocardiogram recorded from a patient
with tachycardia is shown in Figure 13-1. This electrocardiogram is normal except
that the heart rate, as determined from the time intervals between QRS complexes,
is about 150 per minute instead of the normal 72 per minute.
The general causes of tachycardia include increased body temperature, stimulation
of the heart by the sympathetic nerves, or toxic conditions of the heart.
The heart rate increases about 10 beats per minute for each degree Fahrenheit
(18 beats per degree Celsius) increase in body temperature, up to a body tempera-
ture of about 105°F (40.5°C); beyond this, the heart rate may decrease because of
progressive debility of the heart muscle as a result of the fever. Fever causes tachy-
cardia because increased temperature increases the rate of metabolism of the sinus
node, which in turn directly increases its excitability and rate of rhythm.
Many factors can cause the sympathetic nervous system to excite the heart, as we
discuss at multiple points in this text. For instance, when a patient loses blood and
passes into a state of shock or semishock, sympathetic reflex stimulation of the heart
often increases the heart rate to 150 to 180 beats per minute.
Simple weakening of the myocardium usually increases the heart rate because
the weakened heart does not pump blood into the arterial tree to a normal extent,
and this elicits sympathetic reflexes to increase the heart rate.
Bradycardia
The term “bradycardia” means a slow heart rate, usually defined as fewer than 60
beats per minute. Bradycardia is shown by the electrocardiogram in Figure 13-2.
Bradycardia in Athletes. The athlete’s heart is larger and considerably stronger than
that of a normal person, which allows the athlete’s heart to pump a large stroke
volume output per beat even during periods of rest. When the athlete is at rest,
excessive quantities of blood pumped into the arterial tree with each beat initiate
feedback circulatory reflexes or other effects to cause bradycardia.
147
148
Unit III
The Heart
SA block
Figure 13-4
Figure 13-1
Sinoatrial nodal block, with A-V nodal rhythm during the block
Sinus tachycardia (lead I).
period (lead III).
quiet respiration (left half of the record). Then, during
deep respiration, the heart rate increased and decreased
with each respiratory cycle by as much as 30 per cent.
Sinus arrhythmia can result from any one of many cir-
culatory conditions that alter the strengths of the sym-
pathetic and parasympathetic nerve signals to the heart
sinus node. In the “respiratory” type of sinus arrhyth-
mia, as shown in Figure 13-3, this results mainly from
“spillover” of signals from the medullary respiratory
Figure 13-2
center into the adjacent vasomotor center during inspi-
ratory and expiratory cycles of respiration. The spillover
Sinus bradycardia (lead III).
signals cause alternate increase and decrease in the
number of impulses transmitted through the sympa-
thetic and vagus nerves to the heart.
60
70
Abnormal Rhythms That
80
100
Result from Block of Heart
120
Signals Within the
Intracardiac Conduction
Pathways
Figure 13-3
Sinoatrial Block
Sinus arrhythmia as recorded by a cardiotachometer. To the left is
the record when the subject was breathing normally; to the right,
In rare instances, the impulse from the sinus node is
when breathing deeply.
blocked before it enters the atrial muscle. This phe-
nomenon is demonstrated in Figure 13-4, which shows
sudden cessation of P waves, with resultant standstill of
Vagal Stimulation as a Cause of Bradycardia. Any circulatory
the atria. However, the ventricles pick up a new rhythm,
reflex that stimulates the vagus nerves causes release of
the impulse usually originating spontaneously in the
acetylcholine at the vagal endings in the heart, thus
atrioventricular (A-V) node, so that the rate of the ven-
giving a parasympathetic effect. Perhaps the most strik-
tricular QRS-T complex is slowed but not otherwise
ing example of this occurs in patients with carotid sinus
altered.
syndrome. In these patients, the pressure receptors
(baroreceptors) in the carotid sinus region of the carotid
artery walls are excessively sensitive. Therefore, even
Atrioventricular Block
mild external pressure on the neck elicits a strong
The only means by which impulses ordinarily can pass
baroreceptor reflex, causing intense vagal-acetylcholine
from the atria into the ventricles is through the A-V
effects on the heart, including extreme bradycardia.
bundle, also known as the bundle of His. Conditions that
Indeed, sometimes this reflex is so powerful that it actu-
can either decrease the rate of impulse conduction in
ally stops the heart for 5 to 10 seconds.
this bundle or block the impulse entirely are as follows:
1. Ischemia of the A-V node or A-V bundle fibers
Sinus Arrhythmia
often delays or blocks conduction from the atria to
the ventricles. Coronary insufficiency can cause
Figure 13-3 shows a cardiotachometer recording of the
ischemia of the A-V node and bundle in the same
heart rate, at first during normal and then (in the second
way that it can cause ischemia of the myocardium.
half of the record) during deep respiration. A cardiota-
2. Compression of the A-V bundle by scar tissue or by
chometer is an instrument that records by the height of
calcified portions of the heart can depress or block
successive spikes the duration of the interval between
conduction from the atria to the ventricles.
the successive QRS complexes in the electrocar-
3. Inflammation of the A-V node or A-V bundle
diogram. Note from this record that the heart rate
can depress conductivity from the atria to the
increased and decreased no more than 5 per cent during
ventricles. Inflammation results frequently from
Chapter 13
Cardiac Arrhythmias and Their Electrocardiographic Interpretation
149
Dropped beat
P
P
P
P
P
P
P
P
P
P
P
Figure 13-5
Prolonged P-R interval caused by first degree A-V heart block
(lead II).
Figure 13-6
different types of myocarditis, caused, for example,
Second degree A-V block, showing occasional failure of the ven-
by diphtheria or rheumatic fever.
tricles to receive the excitatory signals (lead V3).
4. Extreme stimulation of the heart by the vagus nerves
in rare instances blocks impulse conduction
through the A-V node. Such vagal excitation
occasionally results from strong stimulation of the
baroreceptors in people with carotid sinus
syndrome, discussed earlier in relation to
P
P
P
P
P
P
P
P
P
P
bradycardia.
Incomplete Atrioventricular
Heart Block
Figure 13-7
Prolonged P-R (or P-Q) Interval—First Degree Block. The usual
lapse of time between beginning of the P wave and
Complete A-V block (lead II).
beginning of the QRS complex is about 0.16 second
when the heart is beating at a normal rate. This so-
called P-R interval usually decreases in length with
faster heartbeat and increases with slower heartbeat. In
from the atria into the ventricles occurs. In this instance,
general, when the P-R interval increases to greater than
the ventricles spontaneously establish their own signal,
0.20 second, the P-R interval is said to be prolonged, and
usually originating in the A-V node or A-V bundle.
the patient is said to have first degree incomplete heart
Therefore, the P waves become dissociated from the
block.
QRS-T complexes, as shown in Figure 13-7. Note that
Figure 13-5 shows an electrocardiogram with pro-
the rate of rhythm of the atria in this electrocardiogram
longed P-R interval; the interval in this instance is about
is about 100 beats per minute, whereas the rate of ven-
0.30 second instead of the normal 0.20 or less. Thus, first
tricular beat is less than 40 per minute. Furthermore,
degree block is defined as a delay of conduction from
there is no relation between the rhythm of the P waves
the atria to the ventricles but not actual blockage of con-
and that of the QRS-T complexes because the ventri-
duction. The P-R interval seldom increases above 0.35
cles have “escaped” from control by the atria, and they
to 0.45 second because, by that time, conduction through
are beating at their own natural rate, controlled most
the A-V bundle is depressed so much that conduction
often by rhythmical signals generated in the A-V node
stops entirely. One means for determining the severity
or A-V bundle.
of some heart diseases—acute rheumatic heart disease,
for instance—is to measure the P-R interval.
Stokes-Adams Syndrome—Ventricular Escape. In some
patients with A-V block, the total block comes and goes;
Second Degree Block. When conduction through the A-V
that is, impulses are conducted from the atria into the
bundle is slowed enough to increase the P-R interval to
ventricles for a period of time and then suddenly
0.25 to 0.45 second, the action potential sometimes is
impulses are not conducted. The duration of block may
strong enough to pass through the bundle into the ven-
be a few seconds, a few minutes, a few hours, or even
tricles and sometimes is not strong enough. In this
weeks or longer before conduction returns. This condi-
instance, there will be an atrial P wave but no QRS-T
tion occurs in hearts with borderline ischemia of the
wave, and it is said that there are “dropped beats” of the
conductive system.
ventricles. This condition is called second degree heart
Each time A-V conduction ceases, the ventricles often
block.
do not start their own beating until after a delay of 5 to
Figure 13-6 shows P-R intervals of 0.30 second, as
30 seconds. This results from the phenomenon called
well as one dropped ventricular beat as a result of
overdrive suppression. This means that ventricular
failure of conduction from the atria to the ventricles.
excitability is at first in a suppressed state because the
At times, every other beat of the ventricles is
ventricles have been driven by the atria at a rate greater
dropped, so that a “2:1 rhythm” develops, with the atria
than their natural rate of rhythm. However, after a few
beating twice for every single beat of the ventricles. At
seconds, some part of the Purkinje system beyond the
other times, rhythms of 3:2 or 3:1 also develop.
block, usually in the distal part of the A-V node beyond
the blocked point in the node, or in the A-V bundle,
Complete A-V Block (Third Degree Block). When the condi-
begins discharging rhythmically at a rate of 15 to 40
tion causing poor conduction in the A-V node or A-V
times per minute and acting as the pacemaker of the
bundle becomes severe, complete block of the impulse
ventricles. This is called ventricular escape.
150
Unit III
The Heart
Because the brain cannot remain active for more than
areas of ischemia; (2) small calcified plaques at differ-
4 to 7 seconds without blood supply, most patients faint
ent points in the heart, which press against the adjacent
a few seconds after complete block occurs because the
cardiac muscle so that some of the fibers are irritated;
heart does not pump any blood for 5 to 30 seconds,
and (3) toxic irritation of the A-V node, Purkinje
until the ventricles “escape.” After escape, however, the
system, or myocardium caused by drugs, nicotine, or caf-
slowly beating ventricles usually pump enough blood to
feine. Mechanical initiation of premature contractions
allow rapid recovery from the faint and then to sustain
is also frequent during cardiac catheterization; large
the person. These periodic fainting spells are known as
numbers of premature contractions often occur when
the Stokes-Adams syndrome.
the catheter enters the right ventricle and presses
Occasionally the interval of ventricular standstill at
against the endocardium.
the onset of complete block is so long that it becomes
detrimental to the patient’s health or even causes death.
Consequently, most of these patients are provided
Premature Atrial Contractions
with an artificial pacemaker, a small battery-operated
Figure 13-9 shows a single premature atrial contraction.
electrical stimulator planted beneath the skin, with
The P wave of this beat occurred too soon in the heart
electrodes usually connected to the right ventricle. The
cycle; the P-R interval is shortened, indicating that the
pacemaker provides continued rhythmical impulses
ectopic origin of the beat is in the atria near the A-V
that take control of the ventricles.
node. Also, the interval between the premature con-
traction and the next succeeding contraction is slightly
Incomplete Intraventricular Block—
prolonged, which is called a compensatory pause. One
of the reasons for this is that the premature contraction
Electrical Alternans
originated in the atrium some distance from the sinus
Most of the same factors that can cause A-V block can
node, and the impulse had to travel through a consid-
also block impulse conduction in the peripheral ven-
erable amount of atrial muscle before it discharged the
tricular Purkinje system. Figure 13-8 shows the condi-
sinus node. Consequently, the sinus node discharged late
tion known as electrical alternans, which results from
in the premature cycle, and this made the succeeding
partial intraventricular block every other heartbeat.
sinus node discharge also late in appearing.
This electrocardiogram also shows tachycardia (rapid
Premature atrial contractions occur frequently in
heart rate), which is probably the reason the block has
otherwise healthy people. Indeed, they often occur in
occurred, because when the rate of the heart is rapid, it
athletes whose hearts are in very healthy condition.
may be impossible for some portions of the Purkinje
Mild toxic conditions resulting from such factors as
system to recover from the previous refractory period
smoking, lack of sleep, ingestion of too much coffee,
quickly enough to respond during every succeeding
alcoholism, and use of various drugs can also initiate
heartbeat. Also, many conditions that depress the heart,
such contractions.
such as ischemia, myocarditis, or digitalis toxicity, can
cause incomplete intraventricular block, resulting in
Pulse Deficit. When the heart contracts ahead of sched-
electrical alternans.
ule, the ventricles will not have filled with blood nor-
mally, and the stroke volume output during that
contraction is depressed or almost absent. Therefore,
Premature Contractions
the pulse wave passing to the peripheral arteries after a
premature contraction may be so weak that it cannot be
A premature contraction is a contraction of the heart
felt in the radial artery. Thus, a deficit in the number of
before the time that normal contraction would have
radial pulses occurs when compared with the actual
been expected. This condition is also called extrasystole,
number of contractions of the heart.
premature beat, or ectopic beat.
Causes of Premature Contractions. Most premature con-
A-V Nodal or A-V Bundle Premature
tractions result from ectopic foci in the heart, which emit
Contractions
abnormal impulses at odd times during the cardiac
rhythm. Possible causes of ectopic foci are (1) local
Figure
13-10 shows a premature contraction that
originated in the A-V node or in the A-V bundle. The
P wave is missing from the electrocardiographic record
Premature beat
Figure 13-8
Figure 13-9
Partial intraventricular block—“electrical alternans” (lead III).
Atrial premature beat (lead I).
Chapter 13
Cardiac Arrhythmias and Their Electrocardiographic Interpretation
151
ing with normal contractions. PVCs cause specific
effects in the electrocardiogram, as follows:
Premature beat
1. The QRS complex is usually considerably
prolonged. The reason is that the impulse is
conducted mainly through slowly conducting
P
T
P
T
P
T
P
T
P
T
muscle of the ventricles rather than through the
Purkinje system.
2. The QRS complex has a high voltage for the
following reasons: when the normal impulse passes
through the heart, it passes through both ventricles
nearly simultaneously; consequently, in the normal
heart, the depolarization waves of the two sides
of the heart—mainly of opposite polarity to each
Figure 13-10
other—partially neutralize each other in the
A-V nodal premature contraction (lead III).
electrocardiogram. When a PVC occurs, the
impulse almost always travels in only one direction,
so that there is no such neutralization effect,
and one entire side or end of the ventricles is
depolarized ahead of the other; this causes large
electrical potentials, as shown for the PVCs in
Figure 13-11.
3. After almost all PVCs, the T wave has an electrical
II
potential polarity exactly opposite to that of the
QRS complex, because the slow conduction of the
impulse through the cardiac muscle causes the
muscle fibers that depolarize first also to repolarize
first.
III
Some PVCs are relatively benign in their effects on
overall pumping by the heart; they can result from such
factors as cigarettes, coffee, lack of sleep, various mild
II
III
toxic states, and even emotional irritability. Conversely,
-
-
many other PVCs result from stray impulses or re-
entrant signals that originate around the borders of
infarcted or ischemic areas of the heart. The presence
of such PVCs is not to be taken lightly. Statistics show
that people with significant numbers of PVCs have a
much higher than normal chance of developing sponta-
neous lethal ventricular fibrillation, presumably initi-
ated by one of the PVCs. This is especially true when
the PVCs occur during the vulnerable period for
causing fibrillation, just at the end of the T wave when
+
+
III
II
the ventricles are coming out of refractoriness, as
explained later in the chapter.
Vector Analysis of the Origin of an Ectopic Premature Ventricular
Figure 13-11
Contraction. In Chapter 12, the principles of vectorial
analysis are explained. Applying these principles, one
Premature ventricular contractions (PVCs) demonstrated by the
can determine from the electrocardiogram in Figure
large abnormal QRS-T complexes (leads II and III). Axis of the
13-11 the point of origin of the PVC as follows: Note
premature contractions is plotted in accordance with the princi-
that the potentials of the premature contractions in
ples of vectorial analysis explained in Chapter 12; this shows the
leads II and III are both strongly positive. Plotting these
origin of the PVC to be near the base of the ventricles.
potentials on the axes of leads II and III and solving by
vectorial analysis for the mean QRS vector in the heart,
one finds that the vector of this premature contraction
of the premature contraction. Instead, the P wave is
has its negative end (origin) at the base of the heart and
superimposed onto the QRS-T complex because the
its positive end toward the apex. Thus, the first portion
cardiac impulse traveled backward into the atria at
of the heart to become depolarized during this prema-
the same time that it traveled forward into the ventri-
ture contraction is near the base of the ventricles, which
cles; this P wave slightly distorts the QRS-T complex,
therefore is the locus of the ectopic focus.
but the P wave itself cannot be discerned as such. In
general, A-V nodal premature contractions have
the same significance and causes as atrial premature
contractions.
Paroxysmal Tachycardia
Some abnormalities in different portions of the heart,
Premature Ventricular Contractions
including the atria, the Purkinje system, or the ventri-
cles, can occasionally cause rapid rhythmical discharge
The electrocardiogram of Figure 13-11 shows a series
of impulses that spread in all directions throughout the
of premature ventricular contractions (PVCs) alternat-
heart. This is believed to be caused most frequently by
152
Unit III
The Heart
Figure 13-12
Atrial paroxysmal tachycardia—onset in middle of record (lead I).
Figure 13-13
Ventricular paroxysmal tachycardia (lead III).
re-entrant circus movement feedback pathways that set
up local repeated self-re-excitation. Because of the
rapid rhythm in the irritable focus, this focus becomes
the pacemaker of the heart.
lar paroxysmal tachycardia has the appearance of a
The term “paroxysmal” means that the heart rate
series of ventricular premature beats occurring one
becomes rapid in paroxysms, with the paroxysm
after another without any normal beats interspersed.
beginning suddenly and lasting for a few seconds, a
Ventricular paroxysmal tachycardia is usually a
few minutes, a few hours, or much longer. Then the
serious condition for two reasons. First, this type of
paroxysm usually ends as suddenly as it began, with
tachycardia usually does not occur unless considerable
the pacemaker of the heart instantly shifting back to
ischemic damage is present in the ventricles. Second,
the sinus node.
ventricular tachycardia frequently initiates the lethal
Paroxysmal tachycardia often can be stopped by elic-
condition of ventricular fibrillation because of rapid
iting a vagal reflex. A type of vagal reflex sometimes
repeated stimulation of the ventricular muscle, as we
elicited for this purpose is to press on the neck in the
discuss in the next section.
regions of the carotid sinuses, which may cause enough
Sometimes intoxication from the heart treatment
of a vagal reflex to stop the paroxysm. Various drugs
drug digitalis causes irritable foci that lead to ventricu-
may also be used. Two drugs frequently used are quini-
lar tachycardia. Conversely, quinidine, which increases
dine and lidocaine, either of which depresses the normal
the refractory period and threshold for excitation of
increase in sodium permeability of the cardiac muscle
cardiac muscle, may be used to block irritable foci
membrane during generation of the action potential,
causing ventricular tachycardia.
thereby often blocking the rhythmical discharge of the
focal point that is causing the paroxysmal attack.
Ventricular Fibrillation
Atrial Paroxysmal Tachycardia
The most serious of all cardiac arrhythmias is ventricu-
lar fibrillation, which, if not stopped within
1 to 3
Figure 13-12 demonstrates in the middle of the record
minutes, is almost invariably fatal. Ventricular fibrilla-
a sudden increase in the heart rate from about 95 to
tion results from cardiac impulses that have gone
about 150 beats per minute. On close study of the elec-
berserk within the ventricular muscle mass, stimulating
trocardiogram during the rapid heartbeat, an inverted P
first one portion of the ventricular muscle, then another
wave is seen before each QRS-T complex, and this P
portion, then another, and eventually feeding back onto
wave is partially superimposed onto the normal T wave
itself to re-excite the same ventricular muscle over and
of the preceding beat. This indicates that the origin
over—never stopping. When this happens, many small
of this paroxysmal tachycardia is in the atrium, but
portions of the ventricular muscle will be contracting at
because the P wave is abnormal in shape, the origin is
the same time, while equally as many other portions will
not near the sinus node.
be relaxing. Thus, there is never a coordinate contrac-
tion of all the ventricular muscle at once, which is
A-V Nodal Paroxysmal Tachycardia. Paroxysmal tachycardia
required for a pumping cycle of the heart. Despite
often results from an aberrant rhythm that involves the
massive movement of stimulatory signals throughout
A-V node. This usually causes almost normal QRS-T
the ventricles, the ventricular chambers neither enlarge
complexes but totally missing or obscured P waves.
nor contract but remain in an indeterminate stage of
Atrial or A-V nodal paroxysmal tachycardia, both of
partial contraction, pumping either no blood or negligi-
which are called supraventricular tachycardias, usually
ble amounts. Therefore, after fibrillation begins, uncon-
occurs in young, otherwise healthy people, and they gen-
sciousness occurs within 4 to 5 seconds for lack of blood
erally grow out of the predisposition to tachycardia
flow to the brain, and irretrievable death of tissues
after adolescence. In general, supraventricular tachy-
begins to occur throughout the body within a few
cardia frightens a person tremendously and may cause
minutes.
weakness during the paroxysm, but only seldom does
Multiple factors can spark the beginning of ventricu-
permanent harm come from the attack.
lar fibrillation—a person may have a normal heartbeat
one moment, but 1 second later, the ventricles are in fib-
Ventricular Paroxysmal Tachycardia
rillation. Especially likely to initiate fibrillation are (1)
sudden electrical shock of the heart, or (2) ischemia of
Figure 13-13 shows a typical short paroxysm of ven-
the heart muscle, of its specialized conducting system,
tricular tachycardia. The electrocardiogram of ventricu-
or both.
Chapter 13
Cardiac Arrhythmias and Their Electrocardiographic Interpretation
153
Phenomenon of Re-entry—“Circus
the refractory state, and the impulse can continue
Movements” as the Basis for
around the circle again and again.
Third, the refractory period of the muscle might
Ventricular Fibrillation
become greatly shortened. In this case, the impulse could
When the normal cardiac impulse in the normal heart
also continue around and around the circle.
has traveled through the extent of the ventricles, it has
All these conditions occur in different pathological
no place to go because all the ventricular muscle is
states of the human heart, as follows: (1) A long pathway
refractory and cannot conduct the impulse farther.
typically occurs in dilated hearts. (2) Decreased rate of
Therefore, that impulse dies, and the heart awaits a new
conduction frequently results from (a) blockage of the
action potential to begin in the atrial sinus node.
Purkinje system, (b) ischemia of the muscle, (c) high
Under some circumstances, however, this normal
blood potassium levels, or (d) many other factors. (3)
sequence of events does not occur. Therefore, let us
A shortened refractory period commonly occurs in
explain more fully the background conditions that can
response to various drugs, such as epinephrine, or after
initiate re-entry and lead to “circus movements,” which
repetitive electrical stimulation. Thus, in many cardiac
in turn cause ventricular fibrillation.
disturbances, re-entry can cause abnormal patterns of
Figure
13-14 shows several small cardiac muscle
cardiac contraction or abnormal cardiac rhythms that
strips cut in the form of circles. If such a strip is stimu-
ignore the pace-setting effects of the sinus node.
lated at the 12 o’clock position so that the impulse travels
in only one direction, the impulse spreads progressively
Chain Reaction Mechanism
around the circle until it returns to the 12 o’clock posi-
tion. If the originally stimulated muscle fibers are still in
of Fibrillation
a refractory state, the impulse then dies out because
In ventricular fibrillation, one sees many separate and
refractory muscle cannot transmit a second impulse. But
small contractile waves spreading at the same time in
there are three different conditions that can cause this
different directions over the cardiac muscle. The re-
impulse to continue to travel around the circle, that is,
entrant impulses in fibrillation are not simply a single
to cause “re-entry” of the impulse into muscle that
impulse moving in a circle, as shown in Figure 13-14.
has already been excited. This is called a
“circus
Instead, they have degenerated into a series of multiple
movement.”
wave fronts that have the appearance of a “chain reac-
First, if the pathway around the circle is too long, by
tion.” One of the best ways to explain this process in
the time the impulse returns to the 12 o’clock position,
fibrillation is to describe the initiation of fibrillation by
the originally stimulated muscle will no longer be
electric shock caused by 60-cycle alternating electric
refractory and the impulse will continue around the
current.
circle again and again.
Second, if the length of the pathway remains constant
Fibrillation Caused by
60-Cycle Alternating Current. At a
but the velocity of conduction becomes decreased
central point in the ventricles of heart A in Figure 13-15,
enough, an increased interval of time will elapse before
a
60-cycle electrical stimulus is applied through a
the impulse returns to the 12 o’clock position. By this
stimulating electrode. The first cycle of the electrical
time, the originally stimulated muscle might be out of
stimulus causes a depolarization wave to spread in all
directions, leaving all the muscle beneath the electrode
in a refractory state. After about 0.25 second, part of this
muscle begins to come out of the refractory state. Some
portions come out of refractoriness before other
NORMAL PATHWAY
Stimulus
point
Dividing
impulses
Absolutely
refractory
Absolutely
refractory
Relatively
refractory
LONG PATHWAY
Blocked
impulse
A
B
Figure 13-14
Figure 13-15
Circus movement, showing annihilation of the impulse in the short
A, Initiation of fibrillation in a heart when patches of refractory mus-
pathway and continued propagation of the impulse in the long
culature are present. B, Continued propagation of fibrillatory
pathway.
impulses in the fibrillating ventricle.
154
Unit III
The Heart
portions. This state of events is depicted in heart A by
many lighter patches, which represent excitable cardiac
muscle, and dark patches, which represent still refrac-
tory muscle. Now, continuing 60-cycle stimuli from the
electrode can cause impulses to travel only in certain
directions through the heart but not in all directions.
Thus, in heart A, certain impulses travel for short dis-
tances, until they reach refractory areas of the heart, and
then are blocked. But other impulses pass between the
refractory areas and continue to travel in the excitable
Figure 13-16
areas. Then, several events transpire in rapid succession,
all occurring simultaneously and eventuating in a state
Ventricular fibrillation (lead II).
of fibrillation.
First, block of the impulses in some directions but suc-
cessful transmission in other directions creates one of
dency toward a regular rhythm of any type. During the
the necessary conditions for a re-entrant signal to
first few seconds of ventricular fibrillation, relatively
develop—that is, transmission of some of the depolar-
large masses of muscle contract simultaneously, and this
ization waves around the heart in only some directions
causes coarse, irregular waves in the electrocardiogram.
but not other directions.
After another few seconds, the coarse contractions of
Second, the rapid stimulation of the heart causes two
the ventricles disappear, and the electrocardiogram
changes in the cardiac muscle itself, both of which pre-
changes into a new pattern of low-voltage, very irregu-
dispose to circus movement: (1) The velocity of conduc-
lar waves. Thus, no repetitive electrocardiographic pat-
tion through the heart muscle decreases, which allows a
tern can be ascribed to ventricular fibrillation. Instead,
longer time interval for the impulses to travel around
the ventricular muscle contracts at as many as 30 to 50
the heart. (2) The refractory period of the muscle is
small patches of muscle at a time, and electrocardio-
shortened, allowing re-entry of the impulse into previ-
graphic potentials change constantly and spasmodically
ously excited heart muscle within a much shorter time
because the electrical currents in the heart flow first in
than normally.
one direction and then in another and seldom repeat
Third, one of the most important features of fibrilla-
any specific cycle.
tion is the division of impulses, as demonstrated in heart
The voltages of the waves in the electrocardiogram
A. When a depolarization wave reaches a refractory
in ventricular fibrillation are usually about 0.5 millivolt
area in the heart, it travels to both sides around the
when ventricular fibrillation first begins, but they
refractory area. Thus, a single impulse becomes two
decay rapidly so that after
20 to
30 seconds, they
impulses. Then, when each of these reaches another
are usually only 0.2 to 0.3 millivolt. Minute voltages of
refractory area, it, too, divides to form two more
0.1 millivolt or less may be recorded for 10 minutes or
impulses. In this way, many new wave fronts are contin-
longer after ventricular fibrillation begins. As already
ually being formed in the heart by progressive chain
pointed out, because no pumping of blood occurs
reactions until, finally, there are many small depolariza-
during ventricular fibrillation, this state is lethal unless
tion waves traveling in many directions at the same
stopped by some heroic therapy, such as immediate
time. Furthermore, this irregular pattern of impulse
electroshock through the heart, as explained in the next
travel causes many circuitous routes for the impulses to
section.
travel, greatly lengthening the conductive pathway, which
is one of the conditions that sustains the fibrillation. It
also results in a continual irregular pattern of patchy
Electroshock Defibrillation
refractory areas in the heart.
of the Ventricles
One can readily see when a vicious circle has been
initiated: More and more impulses are formed; these
Although a moderate alternating-current voltage
cause more and more patches of refractory muscle, and
applied directly to the ventricles almost invariably
the refractory patches cause more and more division of
throws the ventricles into fibrillation, a strong high-
the impulses. Therefore, any time a single area of cardiac
voltage alternating electrical current passed through the
muscle comes out of refractoriness, an impulse is close
ventricles for a fraction of a second can stop fibrillation
at hand to re-enter the area.
by throwing all the ventricular muscle into refractori-
Heart B in Figure 13-15 demonstrates the final state
ness simultaneously. This is accomplished by passing
that develops in fibrillation. Here one can see many
intense current through large electrodes placed on two
impulses traveling in all directions, some dividing and
sides of the heart. The current penetrates most of the
increasing the number of impulses, whereas others are
fibers of the ventricles at the same time, thus stimulat-
blocked by refractory areas. In fact, a single electric
ing essentially all parts of the ventricles simultaneously
shock during this vulnerable period frequently can lead
and causing them all to become refractory. All action
to an odd pattern of impulses spreading multidirection-
potentials stop, and the heart remains quiescent for 3 to
ally around refractory areas of muscle, which will lead
5 seconds, after which it begins to beat again, usually
to fibrillation.
with the sinus node or some other part of the heart
becoming the pacemaker. However, the same re-entrant
focus that had originally thrown the ventricles into fib-
Electrocardiogram in Ventricular
rillation often is still present, in which case fibrillation
Fibrillation
may begin again immediately.
When electrodes are applied directly to the two sides
In ventricular fibrillation, the electrocardiogram is
of the heart, fibrillation can usually be stopped using 110
bizarre (Figure 13-16) and ordinarily shows no ten-
volts of
60-cycle alternating current applied for
0.1
Chapter 13
Cardiac Arrhythmias and Their Electrocardiographic Interpretation
155
second or 1000 volts of direct current applied for a few
Atrial Fibrillation
thousandths of a second. When applied through two
electrodes on the chest wall, as shown in Figure 13-17,
Remember that except for the conducting pathway
the usual procedure is to charge a large electrical
through the A-V bundle, the atrial muscle mass is
capacitor up to several thousand volts and then to cause
separated from the ventricular muscle mass by fibrous
the capacitor to discharge for a few thousandths of a
tissue. Therefore, ventricular fibrillation often occurs
second through the electrodes and through the heart.
without atrial fibrillation. Likewise, fibrillation often
In our laboratory, the heart of a single anesthetized
occurs in the atria without ventricular fibrillation
dog was defibrillated 130 times through the chest wall,
(shown to the right in Figure 13-19).
and the animal lived thereafter in perfectly normal
The mechanism of atrial fibrillation is identical to that
condition.
of ventricular fibrillation, except that the process occurs
only in the atrial muscle mass instead of the ventricular
mass. A frequent cause of atrial fibrillation is atrial
Hand Pumping of the Heart
enlargement resulting from heart valve lesions that
(Cardiopulmonary Resuscitation) as
prevent the atria from emptying adequately into the
ventricles, or from ventricular failure with excess
an Aid to Defibrillation
damming of blood in the atria. The dilated atrial walls
Unless defibrillated within 1 minute after fibrillation
provide ideal conditions of a long conductive pathway
begins, the heart is usually too weak to be revived by
as well as slow conduction, both of which predispose to
defibrillation because of the lack of nutrition from coro-
atrial fibrillation.
nary blood flow. However, it is still possible to revive the
heart by preliminarily pumping the heart by hand
Pumping Characteristics of the Atria During Atrial Fibrillation.
(intermittent hand squeezing) and then defibrillating
For the same reasons that the ventricles will not pump
the heart later. In this way, small quantities of blood are
blood during ventricular fibrillation, neither do the atria
delivered into the aorta and a renewed coronary blood
pump blood in atrial fibrillation. Therefore, the atria
supply develops. Then, after a few minutes of hand
become useless as primer pumps for the ventricles. Even
pumping, electrical defibrillation often becomes possi-
so, blood flows passively through the atria into the
ble. Indeed, fibrillating hearts have been pumped by
ventricles, and the efficiency of ventricular pumping is
hand for as long as 90 minutes followed by successful
decreased only 20 to 30 per cent. Therefore, in contrast
defibrillation.
to the lethality of ventricular fibrillation, a person can
A technique for pumping the heart without opening
live for months or even years with atrial fibrillation,
the chest consists of intermittent thrusts of pressure on
although at reduced efficiency of overall heart pumping.
the chest wall along with artificial respiration. This, plus
defibrillation, is called cardiopulmonary resuscitation,
Electrocardiogram in Atrial Fibrillation. Figure 13-18 shows
or CPR.
the electrocardiogram during atrial fibrillation. Numer-
Lack of blood flow to the brain for more than 5 to
ous small depolarization waves spread in all directions
8 minutes usually causes permanent mental impair-
through the atria during atrial fibrillation. Because the
ment or even destruction of brain tissue. Even if the
waves are weak and many of them are of opposite
heart is revived, the person may die from the effects of
polarity at any given time, they usually almost com-
brain damage or may live with permanent mental
pletely electrically neutralize one another. Therefore, in
impairment.
the electrocardiogram, one can see either no P waves
from the atria or only a fine, high-frequency, very low
voltage wavy record. Conversely, the QRS-T complexes
are normal unless there is some pathology of the ven-
tricles, but their timing is irregular, as explained next.
Several thousand volts
for a few milliseconds
Irregularity of Ventricular Rhythm During Atrial Fibrillation.
When the atria are fibrillating, impulses arrive from the
atrial muscle at the A-V node rapidly but also irregu-
Handle for
larly. Because the A-V node will not pass a second
application
impulse for about 0.35 second after a previous one, at
of pressure
least 0.35 second must elapse between one ventricular
contraction and the next. Then an additional but
Electrode
Figure 13-17
Figure 13-18
Application of electrical current to the chest to stop ventricular
Atrial fibrillation (lead I). The waves that can be seen are ventric-
fibrillation.
ular QRS and T waves.
156
Unit III
The Heart
Figure 13-20
Atrial flutter—2:1 and 3:1 atrial to ventricle rhythm (lead I).
Atrial flutter
Atrial fibrillation
Figure 13-19
Cardiac Arrest
Pathways of impulses in atrial flutter and atrial fibrillation.
A final serious abnormality of the cardiac rhythmicity-
conduction system is cardiac arrest. This results from
cessation of all electrical control signals in the heart.
That is, no spontaneous rhythm remains.
variable interval of 0 to 0.6 second occurs before one of
Cardiac arrest is especially likely to occur during deep
the irregular atrial fibrillatory impulses happens to
anesthesia, when many patients develop severe hypoxia
arrive at the A-V node. Thus, the interval between suc-
because of inadequate respiration. The hypoxia pre-
cessive ventricular contractions varies from a minimum
vents the muscle fibers and conductive fibers from main-
of about 0.35 second to a maximum of about 0.95
taining normal electrolyte concentration differentials
second, causing a very irregular heartbeat. In fact, this
across their membranes, and their excitability may be so
irregularity, demonstrated by the variable spacing of the
affected that the automatic rhythmicity disappears.
heartbeats in the electrocardiogram of Figure 13-18, is
In most instances of cardiac arrest from anesthesia,
one of the clinical findings used to diagnose the condi-
prolonged cardiopulmonary resuscitation
(many
tion. Also, because of the rapid rate of the fibrillatory
minutes or even hours) is quite successful in re-
impulses in the atria, the ventricle is driven at a fast
establishing a normal heart rhythm. In some patients,
heart rate, usually between
125 and 150 beats per
severe myocardial disease can cause permanent or
minute.
semipermanent cardiac arrest, which can cause death.
To treat the condition, rhythmical electrical impulses
Electroshock Treatment of Atrial Fibrillation. In the same
from an implanted electronic cardiac pacemaker have
manner that ventricular fibrillation can be converted
been used successfully to keep patients alive for months
back to a normal rhythm by electroshock, so too can
to years.
atrial fibrillation be converted by electroshock. The pro-
cedure is essentially the same as for ventricular fibrilla-
tion conversion—passage of a single strong electric
References
shock through the heart, which throws the entire heart
into refractoriness for a few seconds; a normal rhythm
Al-Khatib SM, LaPointe NM, Kramer JM, Califf RM: What
often follows if the heart is capable of this.
clinicians should know about the QT interval. JAMA
289:2120, 2003.
Armoundas AA, Tomaselli GF, Esperer HD: Pathophysio-
Atrial Flutter
logical basis and clinical application of T-wave alternans.
Atrial flutter is another condition caused by a circus
J Am Coll Cardiol 40:207, 2002.
movement in the atria. It is different from atrial fibril-
Bigi R, Cortigiani L, Desideri A: Exercise electrocardiogra-
lation, in that the electrical signal travels as a single
phy after acute coronary syndromes: still the first testing
large wave always in one direction around and around
modality? Clin Cardiol 8:390, 2003.
the atrial muscle mass, as shown to the left in Figure
Cheitlin MD, Armstrong WF, Aurigemma GP, et al:
13-19. Atrial flutter causes a rapid rate of contraction of
ACC/AHA/ASE 2003 Guideline update for the clinical
the atria, usually between 200 and 350 beats per minute.
application of echocardiography: summary article. A
However, because one side of the atria is contracting
report of the American College of Cardiology/American
while the other side is relaxing, the amount of blood
Heart Association Task Force on Practice Guidelines
pumped by the atria is slight. Furthermore, the signals
(ACC/AHA/ASE Committee to Update the 1997 Guide-
reach the A-V node too rapidly for all of them to be
lines for the Clinical Application of Echocardiography).
passed into the ventricles, because the refractory
J Am Soc Echocardiogr 16:1091, 2003.
periods of the A-V node and A-V bundle are too long
Cohn PF, Fox KM, Daly C: Silent myocardial ischemia.
to pass more than a fraction of the atrial signals. There-
Circulation 108:1263, 2003.
fore, there are usually two to three beats of the atria for
Falk RH: Atrial fibrillation. N Engl J Med 344:1067, 2001.
every single beat of the ventricles.
Frenneaux MP: Assessing the risk of sudden cardiac death
Figure 13-20 shows a typical electrocardiogram in
in a patient with hypertrophic cardiomyopathy. Heart
atrial flutter. The P waves are strong because of con-
90:570, 2004.
traction of semicoordinate masses of muscle. However,
Greenland P, Gaziano JM: Clinical practice: selecting asymp-
note in the record that a QRS-T complex follows an
tomatic patients for coronary computed tomography or
atrial P wave only once for every two to three beats of
electrocardiographic exercise testing. N Engl J Med
the atria, giving a 2:1 or 3:1 rhythm.
349:465, 2003.
Chapter 13
Cardiac Arrhythmias and Their Electrocardiographic Interpretation
157
Hurst JW: Current status of clinical electrocardiography with
Roden DM: Drug-induced prolongation of the QT interval.
suggestions for the improvement of the interpretive
N Engl J Med 350:1013, 2004.
process. Am J Cardiol 92:1072, 2003.
Swynghedauw B, Baillard C, Milliez P: The long QT interval
Jalife J: Ventricular fibrillation: mechanisms of initiation and
is not only inherited but is also linked to cardiac hyper-
maintenance. Annu Rev Physiol 62:25, 2000.
trophy. J Mol Med 81:336, 2003.
Lee TH, Boucher CA: Clinical practice: noninvasive tests in
Topol EJ: A guide to therapeutic decision-making in patients
patients with stable coronary artery disease. N Engl J Med
with non-ST-segment elevation acute coronary syn-
344:1840, 2001.
dromes. J Am Coll Cardiol 41(4 Suppl S):S123, 2003.
Lehmann MH, Morady F: QT interval: metric for cardiac
Wang K, Asinger RW, Marriott HJ: ST-segment elevation in
prognosis? Am J Med 115:732, 2003.
conditions other than acute myocardial infarction. N Engl
Levy S: Pharmacologic management of atrial fibrillation: cur-
J Med 349:2128, 2003.
rent therapeutic strategies. Am Heart J 141(2 Suppl):S15,
Yan GX, Lankipalli RS, Burke JF, et al: Ventricular repolar-
2001.
ization components on the electrocardiogram: cellular
Marban E: The surprising role of vascular K(ATP)
basis and clinical significance. J Am Coll Cardiol 42:401,
channels in vasospastic angina. J Clin Invest
110:153,
2003.
2002.
Zimetbaum PJ, Josephson ME: Use of the electrocardiogram
Maron BJ: Sudden death in young athletes. N Engl J Med
in acute myocardial infarction. N Engl J Med 348:933,
349:1064, 2003.
2003.
Nattel S: New ideas about atrial fibrillation 50 years on.
Zipes DP, Jalife J: Cardiac Electrophysiology,
3rd ed.
Nature 415:219, 2002.
Philadelphia: WB Saunders, 1999.
U N I T IV
The Circulation
14. Overview of the Circulation; Medical Physics of
Pressure, Flow, and Resistance
15. Vascular Distensibility and Functions of the
Arterial and Venous Systems
16. The Microcirculation and the Lymphatic System:
Capillary Fluid Exchange, Interstitial Fluid,
and Lymph Flow
17. Local and Humoral Control of Blood Flow
by the Tissues
18. Nervous Regulation of the Circulation, and
Rapid Control of Arterial Pressure
19. Dominant Role of the Kidney in Long-Term
Regulation of Arterial Pressure and in Hypertension:
The Integrated System for Pressure Control
20. Cardiac Output, Venous Return, and
Their Regulation
21. Muscle Blood Flow and Cardiac Output During
Exercise; the Coronary Circulation and Ischemic
Heart Disease
22. Cardiac Failure
23. Heart Valves and Heart Sounds; Dynamics of
Valvular and Congenital Heart Defects
24. Circulatory Shock and Physiology of Its Treatment
C H A P T E R
1
4
Overview of the Circulation;
Medical Physics of Pressure,
Flow, and Resistance
The function of the circulation is to service the
needs of the body tissues—to transport nutrients to
the body tissues, to transport waste products away,
to conduct hormones from one part of the body to
another, and, in general, to maintain an appropriate
environment in all the tissue fluids of the body for
optimal survival and function of the cells.
The rate of blood flow through most tissues is
controlled in response to tissue need for nutrients. The heart and circulation in
turn are controlled to provide the necessary cardiac output and arterial pres-
sure to cause the needed tissue blood flow. What are the mechanisms for con-
trolling blood volume and blood flow, and how does this relate to all the other
functions of the circulation? These are some of the topics and questions that we
discuss in this section on the circulation.
Physical Characteristics of the Circulation
The circulation, shown in Figure 14-1, is divided into the systemic circulation
and the pulmonary circulation. Because the systemic circulation supplies blood
flow to all the tissues of the body except the lungs, it is also called the greater
circulation or peripheral circulation.
Functional Parts of the Circulation. Before discussing the details of circulatory func-
tion, it is important to understand the role of each part of the circulation.
The function of the arteries is to transport blood under high pressure to the
tissues. For this reason, the arteries have strong vascular walls, and blood flows
at a high velocity in the arteries.
The arterioles are the last small branches of the arterial system; they act as
control conduits through which blood is released into the capillaries. The arte-
riole has a strong muscular wall that can close the arteriole completely or can,
by relaxing, dilate it severalfold, thus having the capability of vastly altering
blood flow in each tissue bed in response to the need of the tissue.
The function of the capillaries is to exchange fluid, nutrients, electrolytes,
hormones, and other substances between the blood and the interstitial fluid.
To serve this role, the capillary walls are very thin and have numerous minute
capillary pores permeable to water and other small molecular substances.
The venules collect blood from the capillaries, and they gradually coalesce
into progressively larger veins.
The veins function as conduits for transport of blood from the venules back
to the heart; equally important, they serve as a major reservoir of extra blood.
Because the pressure in the venous system is very low, the venous walls are thin.
Even so, they are muscular enough to contract or expand and thereby act as a
controllable reservoir for the extra blood, either a small or a large amount,
depending on the needs of the circulation.
Volumes of Blood in the Different Parts of the Circulation. Figure
14-1 gives an
overview of the circulation and lists the percentage of the total blood volume
161
162
Unit IV The Circulation
Pulmonary circulation-9%
Vessel
Cross-Sectional Area (cm2)
Aorta
2.5
Small arteries
20
Arterioles
40
Capillaries
2500
Venules
250
Small veins
80
Aorta
Venae cavae
8
Superior
vena cava
Note particularly the much larger cross-sectional
Heart-7%
areas of the veins than of the arteries, averaging about
four times those of the corresponding arteries. This
explains the large storage of blood in the venous
system in comparison with the arterial system.
Because the same volume of blood must flow
Inferior
Systemic
through each segment of the circulation each minute,
vena cava
vessels
Arteries-13%
the velocity of blood flow is inversely proportional to
vascular cross-sectional area. Thus, under resting con-
ditions, the velocity averages about 33 cm/sec in the
Arterioles
aorta but only 1/1000 as rapidly in the capillaries, about
and
0.3 mm/sec. However, because the capillaries have a
capillaries-7%
typical length of only 0.3 to 1 millimeter, the blood
remains in the capillaries for only 1 to 3 seconds. This
short time is surprising because all diffusion of
nutrient food substances and electrolytes that occurs
through the capillary walls must do so in this exceed-
ingly short time.
Veins, venules,
and venous
Pressures in the Various Portions of the Circulation. Because
sinuses-64%
the heart pumps blood continually into the aorta,
the mean pressure in the aorta is high, averaging about
Figure 14-1
100 mm Hg. Also, because heart pumping is pulsatile,
Distribution of blood (in percentage of total blood) in the different
the arterial pressure alternates between a systolic pres-
parts of the circulatory system.
sure level of 120 mm Hg and a diastolic pressure level
of 80 mm Hg, as shown on the left side of Figure 14-2.
As the blood flows through the systemic circulation,
its mean pressure falls progressively to about 0 mm Hg
by the time it reaches the termination of the venae
cavae where they empty into the right atrium of the
heart.
The pressure in the systemic capillaries varies from
as high as 35 mm Hg near the arteriolar ends to as low
in major segments of the circulation. For instance,
as 10 mm Hg near the venous ends, but their average
about 84 per cent of the entire blood volume of the
“functional” pressure in most vascular beds is about
body is in the systemic circulation, and 16 per cent in
17 mm Hg, a pressure low enough that little of the
heart and lungs. Of the 84 per cent in the systemic cir-
plasma leaks through the minute pores of the capillary
culation, 64 per cent is in the veins, 13 per cent in the
walls, even though nutrients can diffuse easily through
arteries, and 7 per cent in the systemic arterioles and
these same pores to the outlying tissue cells.
capillaries. The heart contains 7 per cent of the blood,
Note at the far right side of Figure 14-2 the respec-
and the pulmonary vessels, 9 per cent.
tive pressures in the different parts of the pulmonary
Most surprising is the low blood volume in the cap-
circulation. In the pulmonary arteries, the pressure is
illaries. It is here, however, that the most important
pulsatile, just as in the aorta, but the pressure level is
function of the circulation occurs, diffusion of sub-
far less: pulmonary artery systolic pressure averages
stances back and forth between the blood and the
about 25 mm Hg and diastolic pressure 8 mm Hg, with
tissues. This function is discussed in detail in Chapter
a mean pulmonary arterial pressure of only 16 mm Hg.
16.
The mean pulmonary capillary pressure averages only
7 mm Hg. Yet the total blood flow through the lungs
Cross-Sectional Areas and Velocities of Blood Flow. If all the
each minute is the same as through the systemic cir-
systemic vessels of each type were put side by side,
culation. The low pressures of the pulmonary system
their approximate total cross-sectional areas for the
are in accord with the needs of the lungs, because
average human being would be as follows:
all that is required is to expose the blood in the
Chapter 14
Overview of the Circulation; Medical Physics of Pressure, Flow, and Resistance
163
120
100
80
60
40
20
0
0
Systemic
Pulmonary
Figure 14-2
Normal blood pressures in the different portions of the circulatory system when a person is lying in the horizontal position.
pulmonary capillaries to oxygen and other gases in the
through a tissue, it immediately returns by way of
pulmonary alveoli.
the veins to the heart. The heart responds
automatically to this increased inflow of blood by
pumping it immediately into the arteries from
Basic Theory of Circulatory
whence it had originally come. Thus, the heart acts
Function
as an automaton, responding to the demands of
the tissues. The heart, however, often needs help
Although the details of circulatory function are
in the form of special nerve signals to make it
complex, there are three basic principles that underlie
pump the required amounts of blood flow.
all functions of the system.
3.
In general the arterial pressure is controlled
1. The rate of blood flow to each tissue of the body
independently of either local blood flow control
is almost always precisely controlled in relation to
or cardiac output control. The circulatory
the tissue need. When tissues are active, they need
system is provided with an extensive system for
greatly increased supply of nutrients and
controlling the arterial blood pressure.
therefore much more blood flow than when at
For instance, if at any time the pressure falls
rest—occasionally as much as 20 to 30 times
significantly below the normal level of about
the resting level. Yet the heart normally cannot
100 mm Hg, within seconds a barrage of nervous
increase its cardiac output more than four to
reflexes elicits a series of circulatory changes to
seven times greater than resting levels. Therefore,
raise the pressure back toward normal. The
it is not possible simply to increase blood flow
nervous signals especially (a) increase the force of
everywhere in the body when a particular tissue
heart pumping, (b) cause contraction of the large
demands increased flow. Instead, the microvessels
venous reservoirs to provide more blood to the
of each tissue continuously monitor tissue needs,
heart, and (c) cause generalized constriction of
such as the availability of oxygen and other
most of the arterioles throughout the body so that
nutrients and the accumulation of carbon dioxide
more blood accumulates in the large arteries to
and other tissue waste products, and these in turn
increase the arterial pressure. Then, over more
act directly on the local blood vessels, dilating or
prolonged periods, hours and days, the kidneys
constricting them, to control local blood flow
play an additional major role in pressure control
precisely to that level required for the tissue
both by secreting pressure-controlling hormones
activity. Also, nervous control of the circulation
and by regulating the blood volume.
from the central nervous system provides
Thus, in summary, the needs of the individual tissues
additional help in controlling tissue blood
are served specifically by the circulation. In the
flow.
remainder of this chapter, we begin to discuss the basic
2. The cardiac output is controlled mainly by the
details of the management of tissue blood flow and
sum of all the local tissue flows. When blood flows
control of cardiac output and arterial pressure.
164
Unit IV The Circulation
Interrelationships Among
of time. Ordinarily, blood flow is expressed in milliliters
per minute or liters per minute, but it can be expressed
Pressure, Flow, and
in milliliters per second or in any other unit of flow.
Resistance
The overall blood flow in the total circulation of an
adult person at rest is about 5000 ml/min. This is called
Blood flow through a blood vessel is determined by
the cardiac output because it is the amount of blood
two factors: (1) pressure difference of the blood be-
pumped into the aorta by the heart each minute.
tween the two ends of the vessel, also sometimes called
“pressure gradient” along the vessel, which is the force
Methods for Measuring Blood Flow. Many mechanical and
that pushes the blood through the vessel, and (2) the
mechanoelectrical devices can be inserted in series with
impediment to blood flow through the vessel, which is
a blood vessel or, in some instances, applied to the
called vascular resistance. Figure 14-3 demonstrates
outside of the vessel to measure flow. They are called
these relationships, showing a blood vessel segment
flowmeters.
located anywhere in the circulatory system.
P1 represents the pressure at the origin of the vessel;
Electromagnetic Flowmeter. One of the most important
at the other end, the pressure is P2. Resistance occurs
devices for measuring blood flow without opening the
vessel is the electromagnetic flowmeter, the principles
as a result of friction between the flowing blood and
of which are illustrated in Figure 14-4. Figure 14-4A
the intravascular endothelium all along the inside of
shows the generation of electromotive force (electrical
the vessel. The flow through the vessel can be cal-
voltage) in a wire that is moved rapidly in a cross-wise
culated by the following formula, which is called
direction through a magnetic field. This is the well-
Ohm’s law:
known principle for production of electricity by the
P
electric generator. Figure 14-4B shows that the same
F
= D
principle applies for generation of electromotive force
R
in blood that is moving through a magnetic field. In
in which F is blood flow, DP is the pressure difference
this case, a blood vessel is placed between the poles
of a strong magnet, and electrodes are placed on the
(P1 - P2) between the two ends of the vessel, and R is
two sides of the vessel perpendicular to the magnetic
the resistance. This formula states, in effect, that the
lines of force. When blood flows through the vessel,
blood flow is directly proportional to the pressure dif-
an electrical voltage proportional to the rate of blood
ference but inversely proportional to the resistance.
flow is generated between the two electrodes, and this
Note that it is the difference in pressure between the
is recorded using an appropriate voltmeter or electronic
two ends of the vessel, not the absolute pressure in the
recording apparatus. Figure
14-4C shows an actual
vessel, that determines rate of flow. For example, if
“probe” that is placed on a large blood vessel to record
the pressure at both ends of a vessel is 100 mm Hg and
its blood flow. The probe contains both the strong
yet no difference exists between the two ends, there
magnet and the electrodes.
will be no flow despite the presence of 100 mm Hg
A special advantage of the electromagnetic flowme-
ter is that it can record changes in flow in less than 1/100
pressure.
of a second, allowing accurate recording of pulsatile
Ohm’s law, illustrated in Equation 1, expresses the
changes in flow as well as steady flow.
most important of all the relations that the reader
needs to understand to comprehend the hemodynam-
Ultrasonic Doppler Flowmeter. Another type of flowmeter
ics of the circulation. Because of the extreme impor-
that can be applied to the outside of the vessel and that
tance of this formula, the reader should also become
has many of the same advantages as the electromagnetic
familiar with its other algebraic forms:
flowmeter is the ultrasonic Doppler flowmeter, shown in
Figure 14-5. A minute piezoelectric crystal is mounted
DP = F ¥ R
at one end in the wall of the device. This crystal, when
P
energized with an appropriate electronic apparatus,
R
= D
transmits ultrasound at a frequency of several hun-
F
dred thousand cycles per second downstream along the
flowing blood. A portion of the sound is reflected by the
red blood cells in the flowing blood. The reflected ultra-
Blood Flow
sound waves then travel backward from the blood cells
toward the crystal. These reflected waves have a lower
Blood flow means simply the quantity of blood that
frequency than the transmitted wave because the red
passes a given point in the circulation in a given period
cells are moving away from the transmitter crystal. This
is called the Doppler effect. (It is the same effect that
one experiences when a train approaches and passes by
Pressure gradient
P1
P2
while blowing its whistle. Once the whistle has passed
Blood flow
by the person, the pitch of the sound from the whistle
suddenly becomes much lower than when the train is
approaching.)
Resistance
For the flowmeter shown in Figure 14-5, the high-
frequency ultrasound wave is intermittently cut off, and
the reflected wave is received back onto the crystal and
Figure 14-3
amplified greatly by the electronic apparatus. Another
portion of the electronic apparatus determines the
Interrelationships among pressure, resistance, and blood flow.
Chapter 14
Overview of the Circulation; Medical Physics of Pressure, Flow, and Resistance
165
+
+
0
0
N
S
N
S
-
+
-
+
-
-
A
B
Figure 14-4
Flowmeter of the electromagnetic
type, showing generation of an
electrical voltage in a wire as it
passes through an electromag-
netic field
(A); generation of an
electrical voltage in electrodes on
a blood vessel when the vessel
is placed in a strong magnetic
field and blood flows through
the vessel
(B); and a modern
C
electromagnetic flowmeter probe
for chronic implantation around
blood vessels (C).
Crystal
A
B
Transmitted
Reflected
C
wave
wave
Figure 14-6
Figure 14-5
A, Two fluids (one dyed red, and the other clear) before flow
Ultrasonic Doppler flowmeter.
begins; B, the same fluids 1 second after flow begins; C, turbu-
lent flow, with elements of the fluid moving in a disorderly pattern.
frequency difference between the transmitted wave and
the reflected wave, thus determining the velocity of
Parabolic Velocity Profile During Laminar Flow. When laminar
blood flow.
flow occurs, the velocity of flow in the center of the
Like the electromagnetic flowmeter, the ultrasonic
vessel is far greater than that toward the outer edges.
Doppler flowmeter is capable of recording rapid, pul-
This is demonstrated in Figure 14-6. In Figure 14-6A, a
satile changes in flow as well as steady flow.
vessel contains two fluids, the one at the left colored by
a dye and the one at the right a clear fluid, but there is
Laminar Flow of Blood in Vessels. When blood flows at a
no flow in the vessel. When the fluids are made to flow,
steady rate through a long, smooth blood vessel, it flows
a parabolic interface develops between them, as shown
in streamlines, with each layer of blood remaining the
1 second later in Figure 14-6B; the portion of fluid adja-
same distance from the vessel wall. Also, the central
cent to the vessel wall has hardly moved, the portion
most portion of the blood stays in the center of the
slightly away from the wall has moved a small distance,
vessel. This type of flow is called laminar flow or stream-
and the portion in the center of the vessel has moved a
line flow, and it is the opposite of turbulent flow, which
long distance. This effect is called the “parabolic profile
is blood flowing in all directions in the vessel and
for velocity of blood flow.”
continually mixing within the vessel, as discussed
The cause of the parabolic profile is the following: The
subsequently.
fluid molecules touching the wall barely move because
166
Unit IV
The Circulation
of adherence to the vessel wall. The next layer of mol-
100 mm Hg pressure
ecules slips over these, the third layer over the second,
the fourth layer over the third, and so forth. Therefore,
the fluid in the middle of the vessel can move rapidly
because many layers of slipping molecules exist
between the middle of the vessel and the vessel wall;
thus, each layer toward the center flows progressively
0 pressure
more rapidly than the outer layers.
Moving sooted
Turbulent Flow of Blood Under Some Conditions. When the rate
paper
of blood flow becomes too great, when it passes by an
Float
obstruction in a vessel, when it makes a sharp turn, or
Anticoagulant
when it passes over a rough surface, the flow may then
solution
become turbulent, or disorderly, rather than streamline
Mercury
(see Figure 14-6C). Turbulent flow means that the blood
flows crosswise in the vessel as well as along the vessel,
usually forming whorls in the blood called eddy cur-
Mercury
rents. These are similar to the whirlpools that one
manometer
frequently sees in a rapidly flowing river at a point of
obstruction.
When eddy currents are present, the blood flows with
much greater resistance than when the flow is stream-
Figure 14-7
line because eddies add tremendously to the overall
friction of flow in the vessel.
Recording arterial pressure with a mercury manometer, a method
The tendency for turbulent flow increases in direct
that has been used in the manner shown for recording pressure
proportion to the velocity of blood flow, the diameter of
throughout the history of physiology.
the blood vessel, and the density of the blood, and is
inversely proportional to the viscosity of the blood, in
accordance with the following equation:
reference for measuring pressure. Actually, blood pres-
n◊d ◊r
Re =
sure means the force exerted by the blood against any
h
unit area of the vessel wall. When one says that the
pressure in a vessel is 50 mm Hg, one means that the
where Re is Reynolds’ number and is the measure of the
force exerted is sufficient to push a column of mercury
tendency for turbulence to occur, n is the mean velocity
of blood flow (in centimeters/second), d is the vessel
against gravity up to a level 50 mm high. If the pres-
diameter (in centimeters), r is density, and h is the vis-
sure is 100 mm Hg, it will push the column of mercury
cosity
(in poise). The viscosity of blood is normally
up to 100 millimeters.
about 1/30 poise, and the density is only slightly greater
Occasionally, pressure is measured in centimeters of
than 1. When Reynolds’ number rises above 200 to 400,
water (cm H2O). A pressure of 10 cm H2O means a
turbulent flow will occur at some branches of vessels but
pressure sufficient to raise a column of water against
will die out along the smooth portions of the vessels.
gravity to a height of 10 centimeters. One millimeter of
However, when Reynolds’ number rises above approx-
mercury pressure equals
1.36 cm water pressure
imately 2000, turbulence will usually occur even in a
because the specific gravity of mercury is 13.6 times
straight, smooth vessel.
that of water, and 1 centimeter is 10 times as great as
Reynolds’ number for flow in the vascular system
even normally rises to 200 to 400 in large arteries; as a
1 millimeter.
result there is almost always some turbulence of flow at
the branches of these vessels. In the proximal portions
High-Fidelity Methods for Measuring Blood Pressure. The
of the aorta and pulmonary artery, Reynolds’ number
mercury in the mercury manometer has so much inertia
can rise to several thousand during the rapid phase of
that it cannot rise and fall rapidly. For this reason, the
ejection by the ventricles; this causes considerable tur-
mercury manometer, although excellent for recording
bulence in the proximal aorta and pulmonary artery
steady pressures, cannot respond to pressure changes
where many conditions are appropriate for turbulence:
that occur more rapidly than about one cycle every 2 to
(1) high velocity of blood flow, (2) pulsatile nature of
3 seconds. Whenever it is desired to record rapidly
the flow, (3) sudden change in vessel diameter, and
changing pressures, some other type of pressure
(4) large vessel diameter. However, in small vessels,
recorder is needed. Figure 14-8 demonstrates the basic
Reynolds’ number is almost never high enough to cause
principles of three electronic pressure transducers com-
turbulence.
monly used for converting blood pressure and/or rapid
changes in pressure into electrical signals and then
recording the electrical signals on a high-speed electri-
cal recorder. Each of these transducers uses a very thin,
Blood Pressure
highly stretched metal membrane that forms one wall
of the fluid chamber. The fluid chamber in turn is con-
Standard Units of Pressure. Blood pressure almost always
nected through a needle or catheter to the blood vessel
is measured in millimeters of mercury
(mm Hg)
in which the pressure is to be measured. When the pres-
because the mercury manometer (shown in Figure
sure is high, the membrane bulges slightly, and when it
14-7) has been used since antiquity as the standard
is low, it returns toward its resting position.
Chapter 14
Overview of the Circulation; Medical Physics of Pressure, Flow, and Resistance
167
any direct means. Instead, resistance must be calcu-
lated from measurements of blood flow and pressure
difference between two points in the vessel. If the
pressure difference between two points is 1 mm Hg
and the flow is 1 ml/sec, the resistance is said to be 1
peripheral resistance unit, usually abbreviated PRU.
A
Expression of Resistance in CGS Units. Occasionally, a
basic physical unit called the CGS (centimeters, grams,
seconds) unit is used to express resistance. This unit is
dyne seconds/centimeters5. Resistance in these units can
be calculated by the following formula:
B
dyne
sec
1333
¥
mm Hg
Ê
ˆ
R in
Ë
5
¯ =
cm
ml sec
Total Peripheral Vascular Resistance and Total Pulmonary Vas-
cular Resistance. The rate of blood flow through the
entire circulatory system is equal to the rate of blood
pumping by the heart—that is, it is equal to the cardiac
output. In the adult human being, this is approximately
100 ml/sec. The pressure difference from the systemic
arteries to the systemic veins is about 100 mm Hg.
Therefore, the resistance of the entire systemic circu-
lation, called the total peripheral resistance, is about
100/100, or 1 PRU.
In conditions in which all the blood vessels through-
out the body become strongly constricted, the total
peripheral resistance occasionally rises to as high as 4
C
PRU. Conversely, when the vessels become greatly
dilated, the resistance can fall to as little as 0.2 PRU.
In the pulmonary system, the mean pulmonary arte-
rial pressure averages 16 mm Hg and the mean left
Figure 14-8
atrial pressure averages 2 mm Hg, giving a net pres-
sure difference of 14 mm. Therefore, when the cardiac
Principles of three types of electronic transducers for recording
output is normal at about
100 ml/sec, the total
rapidly changing blood pressures (explained in the text).
pulmonary vascular resistance calculates to be about
0.14 PRU (about one seventh that in the systemic
In Figure 14-8A, a simple metal plate is placed a few
circulation).
hundredths of a centimeter above the membrane. When
the membrane bulges, the membrane comes closer to
the plate, which increases the electrical capacitance
“Conductance” of Blood in a Vessel and Its Relation to Resis-
between these two, and this change in capacitance can
tance. Conductance is a measure of the blood flow
be recorded using an appropriate electronic system.
through a vessel for a given pressure difference. This
In Figure 14-8B, a small iron slug rests on the mem-
is generally expressed in terms of milliliters per second
brane, and this can be displaced upward into a center
per millimeter of mercury pressure, but it can also be
space inside an electrical wire coil. Movement of the
expressed in terms of liters per second per millimeter
iron into the coil increases the inductance of the coil,
of mercury or in any other units of blood flow and
and this, too, can be recorded electronically.
pressure.
Finally, in Figure 14-8C, a very thin, stretched resist-
It is evident that conductance is the exact reciprocal
ance wire is connected to the membrane. When this wire
of resistance in accord with the following equation:
is stretched greatly, its resistance increases; when it is
stretched less, its resistance decreases. These changes,
1
too, can be recorded by an electronic system.
Conductance
=
With some of these high-fidelity types of recording
Resistance
systems, pressure cycles up to 500 cycles per second
have been recorded accurately. In common use are
Very Slight Changes in Diameter of a Vessel Can Change Its
recorders capable of registering pressure changes that
Conductance Tremendously! Slight changes in the diame-
occur as rapidly as 20 to 100 cycles per second, in the
ter of a vessel cause tremendous changes in the vessel’s
manner shown on the recording paper in Figure 14-8C.
ability to conduct blood when the blood flow is stream-
lined. This is demonstrated by the experiment illus-
Resistance to Blood Flow
trated in Figure 14-9A, which shows three vessels with
relative diameters of 1, 2, and 4 but with the same pres-
Units of Resistance. Resistance is the impediment to
sure difference of 100 mm Hg between the two ends
blood flow in a vessel, but it cannot be measured by
of the vessels. Although the diameters of these vessels
168
Unit IV The Circulation
Note particularly in this equation that the rate of
blood flow is directly proportional to the fourth power
d = 1
of the radius of the vessel, which demonstrates once
A
1 ml/min
again that the diameter of a blood vessel (which is equal
P =
d = 2
to twice the radius) plays by far the greatest role of all
16 ml/min
100 mm
factors in determining the rate of blood flow through a
Hg
d = 4
vessel.
256 ml/min
Importance of the Vessel Diameter “Fourth Power Law” in
Determining Arteriolar Resistance. In the systemic circula-
B
tion, about two thirds of the total systemic resistance
to blood flow is arteriolar resistance in the small
arterioles. The internal diameters of the arterioles
Small vessel
range from as little as 4 micrometers to as great as
25 micrometers. However, their strong vascular walls
allow the internal diameters to change tremendously,
Large vessel
often as much as fourfold. From the fourth power law
discussed above that relates blood flow to diameter of
the vessel, one can see that a fourfold increase in vessel
diameter can increase the flow as much as 256-fold.
Figure 14-9
Thus, this fourth power law makes it possible for
the arterioles, responding with only small changes in
A, Demonstration of the effect of vessel diameter on blood flow.
B, Concentric rings of blood flowing at different velocities; the
diameter to nervous signals or local tissue chemical
farther away from the vessel wall, the faster the flow.
signals, either to turn off almost completely the blood
flow to the tissue or at the other extreme to cause a
vast increase in flow. Indeed, ranges of blood flow of
more than 100-fold in separate tissue areas have been
increase only fourfold, the respective flows are 1, 16,
recorded between the limits of maximum arteriolar
and 256 ml/mm, which is a 256-fold increase in flow.
constriction and maximum arteriolar dilatation.
Thus, the conductance of the vessel increases in pro-
portion to the fourth power of the diameter, in accor-
Resistance to Blood Flow in Series and Parallel Vascular Cir-
dance with the following formula:
cuits. Blood pumped by the heart flows from the high
Conductance µ Diameter4
pressure part of the systemic circulation (i.e., aorta) to
the low pressure side (i.e., vena cava) through many
Poiseuille’s Law. The cause of this great increase in con-
miles of blood vessels arranged in series and in paral-
ductance when the diameter increases can be explained
lel. The arteries, arterioles, capillaries, venules, and
by referring to Figure 14-9B, which shows cross sections
veins are collectively arranged in series. When blood
of a large and a small vessel. The concentric rings inside
vessels are arranged in series, flow through each blood
the vessels indicate that the velocity of flow in each ring
vessel is the same and the total resistance to blood flow
is different from that in the adjacent rings because of
(Rtotal) is equal to the sum of the resistances of each
laminar flow, which was discussed earlier in the chapter.
vessel:
That is, the blood in the ring touching the wall of the
vessel is barely flowing because of its adherence to
R =R +R +R +R ...
total
1
2
3
4
the vascular endothelium. The next ring of blood toward
the center of the vessel slips past the first ring and,
The total peripheral vascular resistance is therefore
therefore, flows more rapidly. The third, fourth, fifth, and
equal to the sum of resistances of the arteries, arteri-
sixth rings likewise flow at progressively increasing
oles, capillaries, venules, and veins. In the example
velocities. Thus, the blood that is near the wall of the
shown in Figure 14-10A, the total vascular resistance
vessel flows extremely slowly, whereas that in the
is equal to the sum of R1 and R2.
middle of the vessel flows extremely rapidly.
Blood vessels branch extensively to form parallel
In the small vessel, essentially all the blood is near the
circuits that supply blood to the many organs and
wall, so that the extremely rapidly flowing central
tissues of the body. This parallel arrangement permits
stream of blood simply does not exist. By integrating the
velocities of all the concentric rings of flowing blood and
each tissue to regulate its own blood flow, to a great
multiplying them by the areas of the rings, one can
extent, independently of flow to other tissues.
derive the following formula, known as Poiseuille’s law:
For blood vessels arranged in parallel
(Figure
14-10B), the total resistance to blood flow is expressed
p
Pr4
as:
F
= D
8
hl
1
1
1
1
1
=
+
+
+ ...
in which F is the rate of blood flow, DP is the pressure
total
R R
1
R
2
R
3
R
4
difference between the ends of the vessel, r is the radius
of the vessel, l is length of the vessel, and h is viscosity
It is obvious that for a given pressure gradient,
of the blood.
far greater amounts of blood will flow through this
Chapter 14
Overview of the Circulation; Medical Physics of Pressure, Flow, and Resistance
169
R1
R2
100
100
100
A
90
90
90
R1
R2
80
80
80
70
70
70
B
60
60
60
R3
R4
50
50
50
40
40
40
Figure 14-10
30
30
30
Vascular resistances: A, in series and B, in parallel.
20
20
20
10
10
10
parallel system than through any of the individual
blood vessels. Therefore, the total resistance is far less
0
0
0
than the resistance of any single blood vessel. Flow
through each of the parallel vessels in Figure 14-10B
Normal
Anemia
Polycythemia
is determined by the pressure gradient and its own
Figure 14-11
resistance, not the resistance of the other parallel
blood vessels. However, increasing the resistance of
Hematocrits in a healthy (normal) person and in patients with
any of the blood vessels increases the total vascular
anemia and polycythemia.
resistance.
It may seem paradoxical that adding more blood
vessels to a circuit reduces the total vascular resist-
ance. Many parallel blood vessels, however, make it
each of which exerts frictional drag against adjacent
easier for blood to flow through the circuit because
cells and against the wall of the blood vessel.
each parallel vessel provides another pathway, or con-
ductance, for blood flow. The total conductance (Ctotal)
Hematocrit. The percentage of the blood that is cells is
for blood flow is the sum of the conductance of each
called the hematocrit. Thus, if a person has a hemat-
parallel pathway:
ocrit of 40, this means that 40 per cent of the blood
volume is cells and the remainder is plasma. The hema-
total
1
C =C +C +C +C ...2
3
4
tocrit of men averages about 42, while that of women
For example, brain, kidney, muscle, gastrointestinal,
averages about 38. These values vary tremendously,
skin, and coronary circulations are arranged in paral-
depending on whether the person has anemia, on the
lel, and each tissue contributes to the overall conduc-
degree of bodily activity, and on the altitude at which
tance of the systemic circulation. Blood flow through
the person resides. These changes in hematocrit are
each tissue is a fraction of the total blood flow (cardiac
discussed in relation to the red blood cells and their
output) and is determined by the resistance (the recip-
oxygen transport function in Chapter 32.
rocal of conductance) for blood flow in the tissue, as
Hematocrit is determined by centrifuging blood in
well as the pressure gradient. Therefore, amputation of
a calibrated tube, as shown in Figure 14-11. The cali-
a limb or surgical removal of a kidney also removes a
bration allows direct reading of the percentage of cells.
parallel circuit and reduces the total vascular conduc-
tance and total blood flow (i.e., cardiac output) while
Effect of Hematocrit on Blood Viscosity. The viscosity of
increasing total peripheral vascular resistance.
blood increases drastically as the hematocrit increases,
as shown in Figure 14-12. The viscosity of whole blood
Effect of Blood Hematocrit and Blood
at normal hematocrit is about 3; this means that three
Viscosity on Vascular Resistance and
times as much pressure is required to force whole
Blood Flow
blood as to force water through the same blood vessel.
Note especially that another of the important factors
When the hematocrit rises to 60 or 70, which it often
in Poiseuille’s equation is the viscosity of the blood.
does in polycythemia, the blood viscosity can become
The greater the viscosity, the less the flow in a vessel
as great as 10 times that of water, and its flow through
if all other factors are constant. Furthermore, the vis-
blood vessels is greatly retarded.
cosity of normal blood is about three times as great as
Other factors that affect blood viscosity are the
the viscosity of water.
plasma protein concentration and types of proteins in
But what makes the blood so viscous? It is mainly
the plasma, but these effects are so much less than
the large numbers of suspended red cells in the blood,
the effect of hematocrit that they are not significant
170
Unit IV The Circulation
Sympathetic
Viscosity of whole blood
7
inhibition
Normal
Viscosity of plasma
6
10
Sympathetic
Viscosity of water
9
stimulation
5
8
4
7
Critical
6
3
closing
5
pressure
2
4
Normal blood
1
3
2
0
1
0
20
40
60
80
100
120 140 160 180 200
0
Arterial pressure (mm Hg)
0
10
20
30
40
50
60
70
Hematocrit
Figure 14-13
Effect of arterial pressure on blood flow through a blood vessel at
Figure 14-12
different degrees of vascular tone caused by increased or
decreased sympathetic stimulation of the vessel.
Effect of hematocrit on blood viscosity. (Water viscosity = 1.)
considerations in most hemodynamic studies. The vis-
arterial pressure is usually four to six times as great as
cosity of blood plasma is about 1.5 times that of water.
blood flow at 50 mm Hg instead of two times as would
be true if there were no effect of increasing pressure
to increase vascular diameter.
Effects of Pressure on Vascular
Note also in Figure 14-13 the large changes in
Resistance and Tissue Blood Flow
blood flow that can be caused by either increased
or decreased sympathetic nerve stimulation of the
From the discussion thus far, one might expect an
peripheral blood vessels. Thus, as shown in the figure,
increase in arterial pressure to cause a proportionate
inhibition of sympathetic activity greatly dilates the
increase in blood flow through the various tissues of
vessels and can increase the blood flow twofold or
the body. However, the effect of pressure on blood
more. Conversely, very strong sympathetic stimulation
flow is greater than one would expect, as shown by the
can constrict the vessels so much that blood flow occa-
upward curving lines in Figure 14-13. The reason for
sionally decreases to as low as zero for a few seconds
this is that an increase in arterial pressure not only
despite high arterial pressure.
increases the force that pushes blood through the
vessels but also distends the vessels at the same time,
which decreases vascular resistance. Thus, greater
References
pressure increases the flow in both of these ways.
Therefore, for most tissues, blood flow at 100 mm Hg
See references for Chapter 15.
C H A P T E R
1
5
Vascular Distensibility and
Functions of the Arterial and
Venous Systems
Vascular Distensibility
A valuable characteristic of the vascular system is
that all blood vessels are distensible. We have seen
one example of this in Chapter 14: When the pres-
sure in blood vessels is increased, this dilates the
blood vessels and therefore decreases their resist-
ance. The result is increased blood flow not only
because of increased pressure but also because of decreased resistance, usually
giving at least twice as much flow increase for each increase in pressure as one
might expect.
Vascular distensibility also plays other important roles in circulatory function.
For example, the distensible nature of the arteries allows them to accommodate
the pulsatile output of the heart and to average out the pressure pulsations. This
provides smooth, continuous flow of blood through the very small blood vessels
of the tissues.
The most distensible by far of all the vessels are the veins. Even slight
increases in venous pressure cause the veins to store 0.5 to 1.0 liter of extra
blood. Therefore, the veins provide a reservoir function for storing large quan-
tities of extra blood that can be called into use whenever required elsewhere in
the circulation.
Units of Vascular Distensibility. Vascular distensibility normally is expressed as the
fractional increase in volume for each millimeter of mercury rise in pressure, in
accordance with the following formula:
Increase in volume
Vascular distensibility
=
Increase in pressure¥Original volume
That is, if 1 mm Hg causes a vessel that originally contained 10 millimeters of
blood to increase its volume by 1 milliliter, the distensibility would be 0.1 per
mm Hg, or 10 per cent per mm Hg.
Difference in Distensibility of the Arteries and the Veins. Anatomically, the walls
of the arteries are far stronger than those of the veins. Consequently, the arter-
ies, on average, are about eight times less distensible than the veins. That is, a
given increase in pressure causes about eight times as much increase in blood
in a vein as in an artery of comparable size.
In the pulmonary circulation, the pulmonary vein distensibilities are similar
to those of the systemic circulation. But, the pulmonary arteries normally
operate under pressures about one sixth of those in the systemic arterial system,
and their distensibilities are correspondingly greater, about six times the dis-
tensibility of systemic arteries.
Vascular Compliance (or Vascular Capacitance)
In hemodynamic studies, it usually is much more important to know the total
quantity of blood that can be stored in a given portion of the circulation for
each millimeter of mercury pressure rise than to know the distensibilities of the
171
172
Unit IV The Circulation
individual vessels. This value is called the compliance
In the entire systemic venous system, the volume
or capacitance of the respective vascular bed; that is,
normally ranges from 2000 to 3500 milliliters, and a
change of several hundred millimeters in this volume
Increase in volume
is required to change the venous pressure only 3 to
Vascular compliance
=
Increase in pressure
5 mm Hg. This mainly explains why as much as one
half liter of blood can be transfused into a healthy
Compliance and distensibility are quite different. A
person in only a few minutes without greatly altering
highly distensible vessel that has a slight volume may
function of the circulation.
have far less compliance than a much less distensible
vessel that has a large volume because compliance is
equal to distensibility times volume.
Effect of Sympathetic Stimulation or Sympathetic Inhibition on
The compliance of a systemic vein is about 24 times
the Volume-Pressure Relations of the Arterial and Venous
that of its corresponding artery because it is about 8
Systems. Also shown in Figure 15-1 are the effects that
times as distensible and it has a volume about 3 times
exciting or inhibiting the vascular sympathetic nerves
as great (8
¥ 3 = 24).
has on the volume-pressure curves. It is evident that
increase in vascular smooth muscle tone caused by
sympathetic stimulation increases the pressure at each
Volume-Pressure Curves of the
volume of the arteries or veins, whereas sympathetic
Arterial and Venous Circulations
inhibition decreases the pressure at each volume.
Control of the vessels in this manner by the sympa-
A convenient method for expressing the relation of
thetics is a valuable means for diminishing the
pressure to volume in a vessel or in any portion of the
dimensions of one segment of the circulation, thus
circulation is to use the so-called volume-pressure
transferring blood to other segments. For instance, an
curve. The red and blue solid curves in Figure 15-1
increase in vascular tone throughout the systemic cir-
represent, respectively, the volume-pressure curves
culation often causes large volumes of blood to shift
of the normal systemic arterial system and venous
into the heart, which is one of the principal methods
system, showing that when the arterial system of the
that the body uses to increase heart pumping.
average adult person (including all the large arteries,
Sympathetic control of vascular capacitance is also
small arteries, and arterioles) is filled with about 700
highly important during hemorrhage. Enhancement of
milliliters of blood, the mean arterial pressure is
sympathetic tone, especially to the veins, reduces the
100 mm Hg, but when it is filled with only 400 milli-
vessel sizes enough that the circulation continues to
liters of blood, the pressure falls to zero.
operate almost normally even when as much as 25 per
cent of the total blood volume has been lost.
Delayed Compliance
(Stress-Relaxation) of Vessels
The term “delayed compliance” means that a vessel
exposed to increased volume at first exhibits a large
140
increase in pressure, but progressive delayed stretching
of smooth muscle in the vessel wall allows the pressure
120
to return back toward normal over a period of minutes
Sympathetic stimulation
to hours. This effect is shown in Figure 15-2. In this
100
figure, the pressure is recorded in a small segment of a
Sympathetic inhibition
vein that is occluded at both ends. An extra volume of
80
blood is suddenly injected until the pressure rises from
5 to 12 mm Hg. Even though none of the blood is
60
Normal volume
removed after it is injected, the pressure begins to
40
decrease immediately and approaches about 9 mm Hg
Arterial system
after several minutes. In other words, the volume of
20
Venous system
blood injected causes immediate elastic distention of the
vein, but then the smooth muscle fibers of the vein begin
0
to “creep” to longer lengths, and their tensions corre-
0
500
1000
1500
2000
2500
3000
3500
spondingly decrease. This effect is a characteristic of all
Volume (ml)
smooth muscle tissue and is called stress-relaxation,
which was explained in Chapter 8.
Delayed compliance is a valuable mechanism by
which the circulation can accommodate much extra
blood when necessary, such as after too large a transfu-
Figure 15-1
sion. Delayed compliance in the reverse direction is one
“Volume-pressure curves” of the systemic arterial and venous
of the ways in which the circulation automatically
systems, showing the effects of stimulation or inhibition of the sym-
adjusts itself over a period of minutes or hours to dimin-
pathetic nerves to the circulatory system.
ished blood volume after serious hemorrhage.
Chapter 15
Vascular Distensibility and Functions of the Arterial and Venous Systems
173
Exponential diastolic decline
14
(may be distorted by
Slow rise
Sharp
reflected wave)
12
to peak
incisura
10
+20
8
6
4
- 80
2
Sharp
upstroke
0
0
20
40
60
80
- 80
Minutes
- 80
0
0.2
0.4
0.6
0.8
1.0
1.2
1.4
1.6
1.8
2.0
Figure 15-2
Seconds
Effect on the intravascular pressure of injecting a volume of blood
into a venous segment and later removing the excess blood,
demonstrating the principle of delayed compliance.
Figure 15-3
Pressure pulse contour recorded from the ascending
aorta.
(Redrawn from Opdyke DF: Fed Proc 11:734, 1952.)
Arterial Pressure Pulsations
With each beat of the heart a new surge of blood fills
the arteries. Were it not for distensibility of the arte-
rial system, all of this new blood would have to flow
160
through the peripheral blood vessels almost instanta-
neously, only during cardiac systole, and no flow would
120
occur during diastole. However, normally the compli-
ance of the arterial tree reduces the pressure pulsa-
80
Normal
Arteriosclerosis Aortic stenosis
tions to almost no pulsations by the time the blood
160
reaches the capillaries; therefore, tissue blood flow is
mainly continuous with very little pulsation.
120
A typical record of the pressure pulsations at the
root of the aorta is shown in Figure 15-3. In the
80
healthy young adult, the pressure at the top of each
Normal
pulse, called the systolic pressure, is about 120 mm Hg.
40
At the lowest point of each pulse, called the diastolic
Patent ductus
pressure, it is about 80 mm Hg. The difference between
0
arteriosus
Aortic
these two pressures, about 40 mm Hg, is called the
regurgitation
pulse pressure.
Two major factors affect the pulse pressure: (1) the
stroke volume output of the heart and (2) the compli-
ance (total distensibility) of the arterial tree. A third,
Figure 15-4
less important factor is the character of ejection from
Aortic pressure pulse contours in arteriosclerosis, aortic stenosis,
the heart during systole.
patent ductus arteriosus, and aortic regurgitation.
In general, the greater the stroke volume output, the
greater the amount of blood that must be accommo-
dated in the arterial tree with each heartbeat, and,
therefore, the greater the pressure rise and fall during
systole and diastole, thus causing a greater pulse pres-
have become hardened with arteriosclerosis and there-
sure. Conversely, the less the compliance of the arte-
fore are relatively noncompliant.
rial system, the greater the rise in pressure for a given
In effect, then, pulse pressure is determined ap-
stroke volume of blood pumped into the arteries. For
proximately by the ratio of stroke volume output to
instance, as demonstrated by the middle top curves in
compliance of the arterial tree. Any condition of the cir-
Figure 15-4, the pulse pressure in old age sometimes
culation that affects either of these two factors also
rises to as much as twice normal, because the arteries
affects the pulse pressure.
174
Unit IV The Circulation
Abnormal Pressure Pulse Contours
Wave fronts
Some conditions of the circulation also cause abnor-
mal contours of the pressure pulse wave in addition
to altering the pulse pressure. Especially distinctive
among these are aortic stenosis, patent ductus arterio-
sus, and aortic regurgitation, each of which is shown in
Figure 15-4.
In aortic stenosis, the diameter of the aortic valve
opening is reduced significantly, and the aortic pres-
sure pulse is decreased significantly because of dimin-
ished blood flow outward through the stenotic valve.
In patent ductus arteriosus, one half or more of the
blood pumped into the aorta by the left ventricle flows
immediately backward through the wide-open ductus
into the pulmonary artery and lung blood vessels, thus
allowing the diastolic pressure to fall very low before
the next heartbeat.
In aortic regurgitation, the aortic valve is absent or
will not close completely. Therefore, after each heart-
beat, the blood that has just been pumped into the
aorta flows immediately backward into the left ventri-
cle. As a result, the aortic pressure can fall all the way
Figure 15-5
to zero between heartbeats. Also, there is no incisura
in the aortic pulse contour because there is no aortic
Progressive stages in transmission of the pressure pulse along the
valve to close.
aorta.
Transmission of Pressure Pulses
to the Peripheral Arteries
When the heart ejects blood into the aorta during
systole, at first only the proximal portion of the aorta
becomes distended because the inertia of the blood
prevents sudden blood movement all the way to the
Incisura
periphery. However, the rising pressure in the proxi-
Systole
Diastole
mal aorta rapidly overcomes this inertia, and the wave
front of distention spreads farther and farther along
the aorta, as shown in Figure 15-5. This is called trans-
mission of the pressure pulse in the arteries.
Proximal aorta
The velocity of pressure pulse transmission in the
normal aorta is 3 to 5 m/sec; in the large arterial
branches, 7 to 10 m/sec; and in the small arteries, 15 to
35 m/sec. In general, the greater the compliance of
each vascular segment, the slower the velocity, which
Femoral artery
explains the slow transmission in the aorta and the
much faster transmission in the much less compliant
Radial artery
small distal arteries. In the aorta, the velocity of trans-
mission of the pressure pulse is 15 or more times the
velocity of blood flow because the pressure pulse is
simply a moving wave of pressure that involves little
Arteriole
forward total movement of blood volume.
Capillary
Damping of the Pressure Pulses in the Smaller Arteries, Arteri-
oles, and Capillaries. Figure 15-6 shows typical changes
in the contours of the pressure pulse as the pulse
0
1
2
travels into the peripheral vessels. Note especially in
Time (seconds)
the three lower curves that the intensity of pulsation
becomes progressively less in the smaller arteries,
the arterioles, and, especially, the capillaries. In fact,
Figure 15-6
only when the aortic pulsations are extremely large or
the arterioles are greatly dilated can pulsations be
Changes in the pulse pressure contour as the pulse wave travels
observed in the capillaries.
toward the smaller vessels.
Chapter 15
Vascular Distensibility and Functions of the Arterial and Venous Systems
175
This progressive diminution of the pulsations in the
arterial pressure cycle, a sound then is heard with each
periphery is called damping of the pressure pulses. The
pulsation. These sounds are called Korotkoff sounds.
cause of this is twofold: (1) resistance to blood move-
The exact cause of Korotkoff sounds is still
ment in the vessels and (2) compliance of the vessels.
debated, but they are believed to be caused mainly
The resistance damps the pulsations because a small
by blood jetting through the partly occluded vessel.
amount of blood must flow forward at the pulse wave
The jet causes turbulence in the vessel beyond the cuff,
front to distend the next segment of the vessel; the
and this sets up the vibrations heard through the
greater the resistance, the more difficult it is for this to
stethoscope.
occur. The compliance damps the pulsations because
In determining blood pressure by the auscultatory
the more compliant a vessel, the greater the quantity
method, the pressure in the cuff is first elevated well
of blood required at the pulse wave front to cause an
above arterial systolic pressure. As long as this cuff
increase in pressure. Therefore, in effect, the degree of
pressure is higher than systolic pressure, the brachial
damping is almost directly proportional to the product
artery remains collapsed so that no blood jets into the
of resistance times compliance.
lower artery during any part of the pressure cycle.
Therefore, no Korotkoff sounds are heard in the lower
artery. But then the cuff pressure gradually is reduced.
Clinical Methods for Measuring
Just as soon as the pressure in the cuff falls below sys-
Systolic and Diastolic Pressures
tolic pressure, blood begins to slip through the artery
beneath the cuff during the peak of systolic pressure,
It is not reasonable to use pressure recorders that
and one begins to hear tapping sounds from the ante-
require needle insertion into an artery for making
cubital artery in synchrony with the heartbeat. As soon
routine pressure measurements in human patients,
as these sounds begin to be heard, the pressure level
although these are used on occasion when special
indicated by the manometer connected to the cuff is
studies are necessary. Instead, the clinician determines
about equal to the systolic pressure.
systolic and diastolic pressures by indirect means,
As the pressure in the cuff is lowered still more, the
usually by the auscultatory method.
Korotkoff sounds change in quality, having less of the
tapping quality and more of a rhythmical and harsher
Auscultatory Method. Figure 15-7 shows the auscultatory
quality. Then, finally, when the pressure in the cuff falls
method for determining systolic and diastolic arterial
to equal diastolic pressure, the artery no longer closes
pressures. A stethoscope is placed over the antecubital
during diastole, which means that the basic factor
artery and a blood pressure cuff is inflated around the
causing the sounds (the jetting of blood through a
upper arm. As long as the cuff continues to compress
squeezed artery) is no longer present. Therefore, the
the arm with too little pressure to close the brachial
sounds suddenly change to a muffled quality, then dis-
artery, no sounds are heard from the antecubital artery
appear entirely after another 5- to 10-millimeter drop
with the stethoscope. However, when the cuff pressure
in cuff pressure. One notes the manometer pressure
is great enough to close the artery during part of the
when the Korotkoff sounds change to the muffled
quality; this pressure is about equal to the diastolic
pressure. The auscultatory method for determining
systolic and diastolic pressures is not entirely accurate,
Sounds
but it usually gives values within 10 per cent of those
determined by direct catheter measurement from
inside the arteries.
120
100
80
Normal Arterial Pressures as Measured by the Auscultatory
Method. Figure 15-8 shows the approximate normal
systolic and diastolic arterial pressures at different
ages. The progressive increase in pressure with age
150
results from the effects of aging on the blood pressure
control mechanisms. We shall see in Chapter 19 that
100
the kidneys are primarily responsible for this long-
term regulation of arterial pressure; and it is well
50
known that the kidneys exhibit definitive changes with
age, especially after the age of 50 years.
0
A slight extra increase in systolic pressure usually
occurs beyond the age of 60 years. This results from
hardening of the arteries, which itself is an end-stage
result of atherosclerosis. The final effect is a bounding
systolic pressure with considerable increase in pulse
pressure, as previously explained.
Figure 15-7
Auscultatory method for measuring systolic and diastolic arterial
Mean Arterial Pressure. The mean arterial pressure is the
pressures.
average of the arterial pressures measured millisecond
176
Unit IV The Circulation
Right atrial pressure is regulated by a balance
between (1) the ability of the heart to pump blood out
of the right atrium and ventricle into the lungs and (2)
200
the tendency for blood to flow from the peripheral veins
into the right atrium. If the right heart is pumping
150
strongly, the right atrial pressure decreases. Con-
versely, weakness of the heart elevates the right atrial
100
pressure. Also, any effect that causes rapid inflow of
blood into the right atrium from the peripheral veins
elevates the right atrial pressure. Some of the factors
50
that can increase this venous return (and thereby
increase the right atrial pressure) are (1) increased
0
blood volume, (2) increased large vessel tone through-
0
20
40
60
80
out the body with resultant increased peripheral
Age (years)
venous pressures, and (3) dilatation of the arterioles,
which decreases the peripheral resistance and allows
rapid flow of blood from the arteries into the veins.
The same factors that regulate right atrial pressure
Figure 15-8
also enter into the regulation of cardiac output
because the amount of blood pumped by the heart
Changes in systolic, diastolic, and mean arterial pressures with
depends on both the ability of the heart to pump and
age. The shaded areas show the approximate normal ranges.
the tendency for blood to flow into the heart from the
peripheral vessels. Therefore, we will discuss regula-
by millisecond over a period of time. It is not equal to
tion of right atrial pressure in much more depth in
the average of systolic and diastolic pressure because
Chapter 20 in connection with regulation of cardiac
the arterial pressure remains nearer to diastolic pres-
output.
sure than to systolic pressure during the greater part
The normal right atrial pressure is about 0 mm Hg,
of the cardiac cycle. Therefore, the mean arterial pres-
which is equal to the atmospheric pressure around the
sure is determined about 60 per cent by the diastolic
body. It can increase to 20 to 30 mm Hg under very
pressure and 40 per cent by the systolic pressure. Note
abnormal conditions, such as (1) serious heart failure
in Figure 15-8 that the mean pressure (solid green
or (2) after massive transfusion of blood, which greatly
line) at all ages is nearer to the diastolic pressure than
increases the total blood volume and causes excessive
to the systolic pressure.
quantities of blood to attempt to flow into the heart
from the peripheral vessels.
The lower limit to the right atrial pressure is usually
Veins and Their Functions
about -3 to -5 mm Hg below atmospheric pressure.
This is also the pressure in the chest cavity that sur-
For years, the veins were considered to be nothing
rounds the heart. The right atrial pressure approaches
more than passageways for flow of blood to the heart,
these low values when the heart pumps with excep-
but it has become apparent that they perform other
tional vigor or when blood flow into the heart from the
special functions that are necessary for operation of
peripheral vessels is greatly depressed, such as after
the circulation. Especially important, they are capable
severe hemorrhage.
of constricting and enlarging and thereby storing
either small or large quantities of blood and making
Venous Resistance and Peripheral
this blood available when it is required by the remain-
Venous Pressure
der of the circulation. The peripheral veins can also
Large veins have so little resistance to blood flow when
propel blood forward by means of a so-called venous
they are distended that the resistance then is almost
pump, and they even help to regulate cardiac output,
zero and is of almost no importance. However, as
an exceedingly important function that is described in
shown in Figure 15-9, most of the large veins that enter
detail in Chapter 20.
the thorax are compressed at many points by the sur-
rounding tissues, so that blood flow is impeded at these
points. For instance, the veins from the arms are com-
Venous Pressures—Right Atrial
pressed by their sharp angulations over the first rib.
Pressure (Central Venous Pressure)
Also, the pressure in the neck veins often falls so low
and Peripheral Venous Pressures
that the atmospheric pressure on the outside of the
neck causes these veins to collapse. Finally, veins
To understand the various functions of the veins, it is
coursing through the abdomen are often compressed
first necessary to know something about pressure in
by different organs and by the intra-abdominal pres-
the veins and what determines the pressure.
sure, so that they usually are at least partially collapsed
Blood from all the systemic veins flows into the right
to an ovoid or slitlike state. For these reasons, the
atrium of the heart; therefore, the pressure in the right
large veins do usually offer some resistance to blood
atrium is called the central venous pressure.
flow, and because of this, the pressure in the more
Chapter 15
Vascular Distensibility and Functions of the Arterial and Venous Systems
177
Sagittal sinus
-10 mm
0 mm
Atmospheric
0 mm
pressure
collapse in neck
+ 6 mm
+ 8 mm
Rib collapse
Axillary collapse
Intrathoracic
+ 22 mm
pressure = - 4 mm Hg
+ 35 mm
Abdominal
pressure
collapse
+ 40 mm
Figure 15-9
Compression points that tend to collapse the veins entering the
thorax.
peripheral small veins in a person lying down is usually
+4 to +6 mm Hg greater than the right atrial pressure.
Effect of High Right Atrial Pressure on Peripheral Venous Pres-
+ 90 mm
sure. When the right atrial pressure rises above its
normal value of 0 mm Hg, blood begins to back up in
the large veins. This enlarges the veins, and even the
collapse points in the veins open up when the right
Figure 15-10
atrial pressure rises above +4 to +6 mm Hg. Then, as
Effect of gravitational pressure on the venous pressures through-
the right atrial pressure rises still further, the addi-
out the body in the standing person.
tional increase causes a corresponding rise in periph-
eral venous pressure in the limbs and elsewhere.
Because the heart must be weakened greatly to cause
pressure, but the pressure rises 1 mm Hg for each 13.6
a rise in right atrial pressure as high as +4 to +6 mm
millimeters of distance below the surface. This pres-
Hg, one often finds that the peripheral venous pres-
sure results from the weight of the water and there-
sure is not noticeably elevated even in the early stages
fore is called gravitational pressure or hydrostatic
of heart failure.
pressure.
Gravitational pressure also occurs in the vascular
Effect of Intra-abdominal Pressure on Venous Pressures of the
system of the human being because of weight of the
Leg. The pressure in the abdominal cavity of a recum-
blood in the vessels, as shown in Figure 15-10. When
bent person normally averages about
+6 mm Hg,
a person is standing, the pressure in the right atrium
but it can rise to +15 to +30 mm Hg as a result of
remains about 0 mm Hg because the heart pumps into
pregnancy, large tumors, or excessive fluid
(called
the arteries any excess blood that attempts to accu-
“ascites”) in the abdominal cavity. When the intra-
mulate at this point. However, in an adult who is stand-
abdominal pressure does rise, the pressure in the veins
ing absolutely still, the pressure in the veins of the feet
of the legs must rise above the abdominal pressure
is about +90 mm Hg simply because of the gravita-
before the abdominal veins will open and allow
tional weight of the blood in the veins between
the blood to flow from the legs to the heart. Thus,
the heart and the feet. The venous pressures at other
if the intra-abdominal pressure is
+20 mm Hg, the
levels of the body are proportionately between 0 and
lowest possible pressure in the femoral veins is also
90 mm Hg.
+20 mm Hg.
In the arm veins, the pressure at the level of the top
rib is usually about +6 mm Hg because of compression
Effect of Gravitational Pressure on Venous
of the subclavian vein as it passes over this rib. The
Pressure
gravitational pressure down the length of the arm then
In any body of water that is exposed to air, the pres-
is determined by the distance below the level of this
sure at the surface of the water is equal to atmospheric
rib. Thus, if the gravitational difference between the
178
Unit IV The Circulation
level of the rib and the hand is +29 mm Hg, this grav-
Deep vein
itational pressure is added to the +6 mm Hg pressure
caused by compression of the vein as it crosses the rib,
making a total of +35 mm Hg pressure in the veins of
Perforating
the hand.
vein
The neck veins of a person standing upright collapse
almost completely all the way to the skull because of
Superficial
vein
atmospheric pressure on the outside of the neck. This
collapse causes the pressure in these veins to remain
at zero along their entire extent. The reason for this is
that any tendency for the pressure to rise above this
level opens the veins and allows the pressure to fall
back to zero because of flow of the blood. Conversely,
any tendency for the neck vein pressure to fall below
Valve
zero collapses the veins still more, which further
increases their resistance and again returns the pres-
sure back to zero.
The veins inside the skull, on the other hand, are in
a noncollapsible chamber (the skull cavity) so that
they cannot collapse. Consequently, negative pressure
can exist in the dural sinuses of the head; in the stand-
ing position, the venous pressure in the sagittal sinus
Figure 15-11
at the top of the brain is about -10 mm Hg because of
Venous valves of the leg.
the hydrostatic “suction” between the top of the skull
and the base of the skull. Therefore, if the sagittal sinus
is opened during surgery, air can be sucked immedi-
ately into the venous system; the air may even pass
legs increase to the full gravitational value of 90 mm
downward to cause air embolism in the heart, and
Hg in about 30 seconds. The pressures in the capillar-
death can ensue.
ies also increase greatly, causing fluid to leak from the
circulatory system into the tissue spaces. As a result,
Effect of the Gravitational Factor on Arterial and Other Pres-
the legs swell, and the blood volume diminishes.
sures. The gravitational factor also affects pressures in
Indeed, 10 to 20 per cent of the blood volume can be
the peripheral arteries and capillaries, in addition to its
lost from the circulatory system within the 15 to 30
effects in the veins. For instance, a standing person who
minutes of standing absolutely still, as often occurs
has a mean arterial pressure of 100 mm Hg at the level
when a soldier is made to stand at rigid attention.
of the heart has an arterial pressure in the feet of about
190 mm Hg. Therefore, when one states that the arte-
Venous Valve Incompetence Causes “Varicose” Veins. The
rial pressure is 100 mm Hg, this generally means that
valves of the venous system frequently become
this is the pressure at the gravitational level of the
“incompetent” or sometimes even are destroyed. This
heart but not necessarily elsewhere in the arterial
is especially true when the veins have been over-
vessels.
stretched by excess venous pressure lasting weeks or
months, as occurs in pregnancy or when one stands
Venous Valves and the “Venous Pump”:
most of the time. Stretching the veins increases their
Their Effects on Venous Pressure
cross-sectional areas, but the leaflets of the valves do
Were it not for valves in the veins, the gravitational
not increase in size. Therefore, the leaflets of the valves
pressure effect would cause the venous pressure in the
no longer close completely. When this develops, the
feet always to be about +90 mm Hg in a standing adult.
pressure in the veins of the legs increases greatly
However, every time one moves the legs, one tightens
because of failure of the venous pump; this further
the muscles and compresses the veins in or adjacent to
increases the sizes of the veins and finally destroys the
the muscles, and this squeezes the blood out of the
function of the valves entirely. Thus, the person devel-
veins. But the valves in the veins, shown in Figure
ops “varicose veins,” which are characterized by large,
15-11, are arranged so that the direction of venous
bulbous protrusions of the veins beneath the skin of
blood flow can be only toward the heart. Conse-
the entire leg, particularly the lower leg.
quently, every time a person moves the legs or even
Whenever people with varicose veins stand for more
tenses the leg muscles, a certain amount of venous
than a few minutes, the venous and capillary pressures
blood is propelled toward the heart. This pumping
become very high, and leakage of fluid from the cap-
system is known as the “venous pump” or “muscle
illaries causes constant edema in the legs. The edema
pump,” and it is efficient enough that under ordinary
in turn prevents adequate diffusion of nutritional
circumstances, the venous pressure in the feet of a
materials from the capillaries to the muscle and skin
walking adult remains less than +20 mm Hg.
cells, so that the muscles become painful and weak,
If a person stands perfectly still, the venous pump
and the skin frequently becomes gangrenous and
does not work, and the venous pressures in the lower
ulcerates. The best treatment for such a condition is
Chapter 15
Vascular Distensibility and Functions of the Arterial and Venous Systems
179
continual elevation of the legs to a level at least as high
significant gravitational changes in pressure at this point
as the heart. Tight binders on the legs also can be of
in the following way:
considerable assistance in preventing the edema and
If the pressure at the tricuspid valve rises slightly
above normal, the right ventricle fills to a greater extent
its sequelae.
than usual, causing the heart to pump blood more
rapidly and therefore to decrease the pressure at the tri-
Clinical Estimation of Venous Pressure. The venous pressure
cuspid valve back toward the normal mean value. Con-
often can be estimated by simply observing the degree
versely, if the pressure falls, the right ventricle fails to fill
of distention of the peripheral veins—especially of the
adequately, its pumping decreases, and blood dams up
neck veins. For instance, in the sitting position, the neck
in the venous system until the pressure at the tricuspid
veins are never distended in the normal quietly resting
level again rises to the normal value. In other words,
person. However, when the right atrial pressure be-
the heart acts as a feedback regulator of pressure at the
comes increased to as much as +10 mm Hg, the lower
tricuspid valve.
veins of the neck begin to protrude; and at +15 mm Hg
When a person is lying on his or her back, the tricus-
atrial pressure essentially all the veins in the neck
pid valve is located at almost exactly 60 per cent of the
become distended.
chest thickness in front of the back. This is the zero pres-
sure reference level for a person lying down.
Direct Measurement of Venous Pressure and
Right Atrial Pressure
Venous pressure can also be measured with ease by
inserting a needle directly into a vein and connecting it
Blood Reservoir Function of the Veins
to a pressure recorder. The only means by which right
atrial pressure can be measured accurately is by insert-
As pointed out in Chapter 14, more than 60 per cent
ing a catheter through the peripheral veins and into the
of all the blood in the circulatory system is usually in
right atrium. Pressures measured through such central
the veins. For this reason and also because the veins
venous catheters are used almost routinely in some types
are so compliant, it is said that the venous system
of hospitalized cardiac patients to provide constant
serves as a blood reservoir for the circulation.
assessment of heart pumping ability.
When blood is lost from the body and the arterial
pressure begins to fall, nervous signals are elicited
Pressure Reference Level for Measuring Venous and Other
Circulatory Pressures
from the carotid sinuses and other pressure-sensitive
In discussions up to this point, we often have spoken of
areas of the circulation, as discussed in Chapter 18.
right atrial pressure as being 0 mm Hg and arterial pres-
These in turn elicit nerve signals from the brain and
sure as being 100 mm Hg, but we have not stated the
spinal cord mainly through sympathetic nerves to the
gravitational level in the circulatory system to which this
veins, causing them to constrict. This takes up much of
pressure is referred. There is one point in the circula-
the slack in the circulatory system caused by the lost
tory system at which gravitational pressure factors
blood. Indeed, even after as much as 20 per cent of the
caused by changes in body position of a healthy person
total blood volume has been lost, the circulatory
usually do not affect the pressure measurement by more
system often functions almost normally because of this
than 1 to 2 mm Hg. This is at or near the level of the tri-
variable reservoir function of the veins.
cuspid valve, as shown by the crossed axes in Figure
15-12. Therefore, all circulatory pressure measurements
Specific Blood Reservoirs. Certain portions of the circu-
discussed in this text are referred to this level, which is
called the reference level for pressure measurement.
latory system are so extensive and/or so compliant that
The reason for lack of gravitational effects at the
they are called
“specific blood reservoirs.” These
tricuspid valve is that the heart automatically prevents
include (1) the spleen, which sometimes can decrease
in size sufficiently to release as much as 100 milliliters
of blood into other areas of the circulation; (2) the
Right ventricle
liver, the sinuses of which can release several hundred
milliliters of blood into the remainder of the circula-
tion; (3) the large abdominal veins, which can con-
tribute as much as 300 milliliters; and (4) the venous
plexus beneath the skin, which also can contribute
several hundred milliliters. The heart and the lungs,
Right atrium
although not parts of the systemic venous reservoir
system, must also be considered blood reservoirs. The
heart, for instance, shrinks during sympathetic stimu-
lation and in this way can contribute some 50 to 100
milliliters of blood; the lungs can contribute another
100 to 200 milliliters when the pulmonary pressures
decrease to low values.
Natural reference
point
The Spleen as a Reservoir for Storing Red Blood Cells.
Figure 15-13 shows that the spleen has two separate
Figure 15-12
areas for storing blood: the venous sinuses and the
Reference point for circulatory pressure measurement (located
pulp. The sinuses can swell the same as any other part
near the tricuspid valve).
of the venous system and store whole blood.
180
Unit IV The Circulation
lined with similar cells. These cells function as part of a
cleansing system for the blood, acting in concert with a
similar system of reticuloendothelial cells in the venous
sinuses of the liver. When the blood is invaded by infec-
tious agents, the reticuloendothelial cells of the spleen
rapidly remove debris, bacteria, parasites, and so forth.
Pulp
Also, in many chronic infectious processes, the spleen
enlarges in the same manner that lymph nodes enlarge
Capillaries
and then performs its cleansing function even more
avidly.
Venous sinuses
References
Vein
Artery
Badeer HS: Hemodynamics for medical students. Am J
Physiol (Adv Physiol Educ) 25:44, 2001.
Benetos A, Waeber B, Izzo J, Mitchell G, et al: Influence of
age, risk factors, and cardiovascular and renal disease on
arterial stiffness: clinical applications. Am J Hypertens
Figure 15-13
15:1101, 2002.
Gimbrone MA Jr, Topper JN, Nagel T, et al: Endothelial dys-
Functional structures of the spleen.
(Courtesy of Dr. Don W.
function, hemodynamic forces, and atherogenesis. Ann
Fawcett, Montana.)
N Y Acad Sci 902:230, 2000.
Guyton AC: Arterial Pressure and Hypertension.
Philadelphia: WB Saunders Co, 1980.
In the splenic pulp, the capillaries are so permeable
Guyton AC, Jones CE: Central venous pressure: physiologi-
cal significance and clinical implications. Am Heart J 86:
that whole blood, including the red blood cells, oozes
431, 1973.
through the capillary walls into a trabecular mesh,
Guyton AC, Jones CE, Coleman TG: Circulatory Physiology:
forming the red pulp. The red cells are trapped by the
Cardiac Output and Its Regulation. Philadelphia: WB
trabeculae, while the plasma flows on into the venous
Saunders Co, 1973.
sinuses and then into the general circulation. As a con-
Hall J: Integration and regulation of cardiovascular function.
sequence, the red pulp of the spleen is a special reser-
Am J Physiol (Adv Physiol Educ) 22:s174, 1999.
voir that contains large quantities of concentrated red
Hicks JW, Badeer HS: Gravity and the circulation: “open”
blood cells. These can then be expelled into the general
vs. “closed” systems. Am J Physiol 262:R725-R732, 1992.
circulation whenever the sympathetic nervous system
Jones DW, Appel LJ, Sheps SG, et al: Measuring blood pres-
becomes excited and causes the spleen and its vessels
sure accurately: New and persistent challenges. JAMA
289:1027, 2003.
to contract. As much as 50 milliliters of concentrated
Lakatta EG, Levy D: Arterial and cardiac aging: major
red blood cells can be released into the circulation,
shareholders in cardiovascular disease enterprises. Part I:
raising the hematocrit 1 to 2 per cent.
Aging arteries: a “set up” for vascular disease. Circulation
In other areas of the splenic pulp are islands of white
107:139, 2003.
blood cells, which collectively are called the white pulp.
McAlister FA, Straus SE: Evidence based treatment of
Here lymphoid cells are manufactured similar to those
hypertension. Measurement of blood pressure: an evi-
manufactured in the lymph nodes. They are part of the
dence based review. BMJ 322:908, 2001.
body’s immune system, described in Chapter 34.
O’Brien E, Asmar R, Beilin L, et al: European Society
of Hypertension recommendations for conventional,
Blood-Cleansing Function of the Spleen—Removal of Old Cells
ambulatory and home blood pressure measurement. J
Blood cells passing through the splenic pulp before
Hypertens 21:821, 2003.
entering the sinuses undergo thorough squeezing.
Perloff D, Grim C, Flack J, et al: Human blood pressure
Therefore, it is to be expected that fragile red blood cells
determination by sphygmomanometry. Circulation
would not withstand the trauma. For this reason, many
88:2460, 1993.
of the red blood cells destroyed in the body have their
Rothe CF, Gersting JM: Cardiovascular interactions: an
final demise in the spleen. After the cells rupture, the
interactive tutorial and mathematical model. Am J Physiol
released hemoglobin and the cell stroma are digested
(Adv Physiol Educ) 26:98, 2002.
by the reticuloendothelial cells of the spleen, and the
Safar ME, Levy BI, Struijker-Boudier H: Current perspec-
products of digestion are mainly reused by the body as
tives on arterial stiffness and pulse pressure in hyperten-
nutrients, often for making new blood cells.
sion and cardiovascular diseases. Circulation
107:2864,
2003.
Reticuloendothelial Cells of the Spleen
Verdecchia P, Angeli F, Gattobigio R: Clinical usefulness of
The pulp of the spleen contains many large phagocytic
ambulatory blood pressure monitoring. J Am Soc Nephrol
reticuloendothelial cells, and the venous sinuses are
15(Suppl 1):S30, 2004.
C H A P T E R
1
6
The Microcirculation and the
Lymphatic System: Capillary
Fluid Exchange, Interstitial Fluid,
and Lymph Flow
The most purposeful function of the circulation
occurs in the microcirculation: this is transport of
nutrients to the tissues and removal of cell excreta.
The small arterioles control blood flow to each
tissue area, and local conditions in the tissues in turn
control the diameters of the arterioles. Thus, each
tissue, in most instances, controls its own blood flow
in relation to its individual needs, a subject that is
discussed in detail in Chapter 17.
The walls of the capillaries are extremely thin, constructed of single-layer,
highly permeable endothelial cells. Therefore, water, cell nutrients, and cell
excreta can all interchange quickly and easily between the tissues and the cir-
culating blood.
The peripheral circulation of the whole body has about 10 billion capillaries
with a total surface area estimated to be 500 to 700 square meters (about one-
eighth the surface area of a football field). Indeed, it is rare that any single func-
tional cell of the body is more than 20 to 30 micrometers away from a capillary.
Structure of the Microcirculation and
Capillary System
The microcirculation of each organ is organized specifically to serve that organ’s
needs. In general, each nutrient artery entering an organ branches six to eight times
before the arteries become small enough to be called arterioles, which generally
have internal diameters of only 10 to 15 micrometers. Then the arterioles themselves
branch two to five times, reaching diameters of 5 to 9 micrometers at their ends
where they supply blood to the capillaries.
The arterioles are highly muscular, and their diameters can change manyfold. The
metarterioles (the terminal arterioles) do not have a continuous muscular coat, but
smooth muscle fibers encircle the vessel at intermittent points, as shown in Figure
16-1 by the black dots on the sides of the metarteriole.
At the point where each true capillary originates from a metarteriole, a smooth
muscle fiber usually encircles the capillary. This is called the precapillary sphincter.
This sphincter can open and close the entrance to the capillary.
The venules are larger than the arterioles and have a much weaker muscular coat.
Yet it must be remembered that the pressure in the venules is much less than that
in the arterioles, so that the venules still can contract considerably despite the weak
muscle.
This typical arrangement of the capillary bed is not found in all parts of the body;
however, some similar arrangement serves the same purposes. Most important, the
metarterioles and the precapillary sphincters are in close contact with the tissues
they serve. Therefore, the local conditions of the tissues—the concentrations of
nutrients, end products of metabolism, hydrogen ions, and so forth—can cause direct
effects on the vessels in controlling local blood flow in each small tissue area.
Structure of the Capillary Wall. Figure 16-2 shows the ultramicroscopic structure of
typical endothelial cells in the capillary wall as found in most organs of the body,
especially in muscles and connective tissue. Note that the wall is composed of
181
182
Unit IV The Circulation
Metarteriole
Preferential channel
Because the intercellular clefts are located only at
the edges of the endothelial cells, they usually repre-
Arteriole
sent no more than 1/1000 of the total surface area of
Precapillary
sphincter
the capillary wall. Nevertheless, the rate of thermal
motion of water molecules as well as most water-
True capillaries
soluble ions and small solutes is so rapid that all of
these diffuse with ease between the interior and exte-
rior of the capillaries through these “slit-pores,” the
Venule
intercellular clefts.
Also present in the endothelial cells are many
minute plasmalemmal vesicles. These form at one
surface of the cell by imbibing small packets of plasma
Figure 16-1
or extracellular fluid. They can then move slowly
through the endothelial cell. It also has been postu-
Structure of the mesenteric capillary bed. (Redrawn from Zweifach
BW: Factors Regulating Blood Pressure. New York: Josiah Macy,
lated that some of these vesicles coalesce to form
Jr., Foundation, 1950.)
vesicular channels all the way through the endothelial
cell, which is demonstrated to the right in Figure 16-2.
However, careful measurements in laboratory animals
Endothelial
probably have proved that these vesicular forms of
cell
transport are quantitatively of little importance.
Special Types of “Pores” Occur in the Capillaries of Certain
Vesicular
Organs. The “pores” in the capillaries of some organs
channel??
have special characteristics to meet the peculiar needs
of the organs. Some of these characteristics are as
Plasmalemmal
vesicles
follows:
1. In the brain, the junctions between the capillary
endothelial cells are mainly “tight” junctions that
allow only extremely small molecules such as
water, oxygen, and carbon dioxide to pass into or
out of the brain tissues.
Basement
2. In the liver, the opposite is true. The clefts between
Intercellular
membrane
the capillary endothelial cells are wide open, so
cleft
that almost all dissolved substances of the plasma,
including the plasma proteins, can pass from the
Figure 16-2
blood into the liver tissues.
Structure of the capillary wall. Note especially the intercellular cleft
3. The pores of the gastrointestinal capillary
at the junction between adjacent endothelial cells; it is believed
membranes are midway between those of the
that most water-soluble substances diffuse through the capillary
muscles and those of the liver.
membrane along the clefts.
4. In the glomerular tufts of the kidney, numerous
small oval windows called fenestrae penetrate all
the way through the middle of the endothelial
cells, so that tremendous amounts of very small
a unicellular layer of endothelial cells and is sur-
molecular and ionic substances (but not the large
molecules of the plasma proteins) can filter through
rounded by a very thin basement membrane on the
the glomeruli without having to pass through the
outside of the capillary. The total thickness of the cap-
clefts between the endothelial cells.
illary wall is only about 0.5 micrometer. The internal
diameter of the capillary is 4 to 9 micrometers, barely
large enough for red blood cells and other blood cells
Flow of Blood in the
to squeeze through.
Capillaries—Vasomotion
“Pores” in the Capillary Membrane. Studying Figure 16-2,
one sees two very small passageways connecting the
Blood usually does not flow continuously through the
interior of the capillary with the exterior. One of these
capillaries. Instead, it flows intermittently, turning on
is an intercellular cleft, which is the thin-slit, curving
and off every few seconds or minutes. The cause of this
channel that lies at the bottom of the figure between
intermittency is the phenomenon called vasomotion,
adjacent endothelial cells. Each cleft is interrupted
which means intermittent contraction of the metarte-
periodically by short ridges of protein attachments
rioles and precapillary sphincters
(and sometimes
that hold the endothelial cells together, but between
even the very small arterioles as well).
these ridges fluid can percolate freely through the
cleft. The cleft normally has a uniform spacing with a
Regulation of Vasomotion. The most important factor
width of about 6 to 7 nanometers (60 to 70 angstroms),
found thus far to affect the degree of opening and
slightly smaller than the diameter of an albumin
closing of the metarterioles and precapillary sphinc-
protein molecule.
ters is the concentration of oxygen in the tissues. When
Chapter 16
The Microcirculation and the Lymphatic System
183
the rate of oxygen usage by the tissue is great so that
Arterial end
Blood capillary
Venous end
tissue oxygen concentration decreases below normal,
the intermittent periods of capillary blood flow occur
more often, and the duration of each period of flow
lasts longer, thereby allowing the capillary blood to
carry increased quantities of oxygen (as well as other
nutrients) to the tissues. This effect, along with multi-
ple other factors that control local tissue blood flow, is
discussed in Chapter 17.
Average Function of the
Capillary System
Lymphatic
Despite the fact that blood flow through each capillary
capillary
is intermittent, so many capillaries are present in the
tissues that their overall function becomes averaged.
That is, there is an average rate of blood flow through
each tissue capillary bed, an average capillary pressure
within the capillaries, and an average rate of transfer of
Figure 16-3
substances between the blood of the capillaries and the
surrounding interstitial fluid. In the remainder of this
Diffusion of fluid molecules and dissolved substances between
chapter, we will be concerned with these averages,
the capillary and interstitial fluid spaces.
although one must remember that the average func-
tions are, in reality, the functions of literally billions of
individual capillaries, each operating intermittently in
response to local conditions in the tissues.
substances, such as sodium ions and glucose that can
go only through the pores.
Exchange of Water, Nutrients,
Water-Soluble, Non-Lipid-Soluble Substances Diffuse Only
and Other Substances
Through Intercellular
“Pores” in the Capillary Membrane.
Between the Blood and
Many substances needed by the tissues are soluble in
water but cannot pass through the lipid membranes of
Interstitial Fluid
the endothelial cells; such substances include water
molecules themselves, sodium ions, chloride ions, and
Diffusion Through the
glucose. Despite the fact that not more than 1/1000 of
Capillary Membrane
the surface area of the capillaries is represented by the
intercellular clefts between the endothelial cells, the
By far the most important means by which substances
velocity of thermal molecular motion in the clefts is so
are transferred between the plasma and the interstitial
great that even this small area is sufficient to allow
fluid is diffusion. Figure
16-3 demonstrates this
tremendous diffusion of water and water-soluble sub-
process, showing that as the blood flows along the
stances through these cleft-pores. To give one an idea
lumen of the capillary, tremendous numbers of water
of the rapidity with which these substances diffuse, the
molecules and dissolved particles diffuse back and
rate at which water molecules diffuse through the cap-
forth through the capillary wall, providing continual
illary membrane is about 80 times as great as the rate at
mixing between the interstitial fluid and the plasma.
which plasma itself flows linearly along the capillary.
Diffusion results from thermal motion of the water
That is, the water of the plasma is exchanged with
molecules and dissolved substances in the fluid, the
the water of the interstitial fluid 80 times before the
different molecules and ions moving first in one direc-
plasma can flow the entire distance through the
tion and then another, bouncing randomly in every
capillary.
direction.
Effect of Molecular Size on Passage Through the
Lipid-Soluble Substances Can Diffuse Directly Through the Cell
Pores. The width of the capillary intercellular cleft-
Membranes of the Capillary Endothelium. If a substance is
pores, 6 to 7 nanometers, is about 20 times the diame-
lipid soluble, it can diffuse directly through the cell
ter of the water molecule, which is the smallest
membranes of the capillary without having to go
molecule that normally passes through the capillary
through the pores. Such substances include oxygen and
pores. Conversely, the diameters of plasma protein
carbon dioxide. Because these substances can per-
molecules are slightly greater than the width of the
meate all areas of the capillary membrane, their rates
pores. Other substances, such as sodium ions, chloride
of transport through the capillary membrane are
ions, glucose, and urea, have intermediate diameters.
many times faster than the rates for lipid-insoluble
Therefore, the permeability of the capillary pores for
184
Unit IV The Circulation
Table 16-1
Relative Permeability of Skeletal Muscle Capillary Pores to
Different-Sized Molecules
Free fluid
vesicles
Substance
Molecular Weight
Permeability
Water
18
1.00
Rivulets
NaCl
58.5
0.96
of free
Urea
60
0.8
fluid
Glucose
180
0.6
Sucrose
342
0.4
Inulin
5,000
0.2
Myoglobin
17,600
0.03
Hemoglobin
68,000
0.01
Albumin
69,000
0.001
Data from Pappenheimer JR: Passage of molecules through capillary walls.
Physiol Rev 33:387, 1953.
Collagen fiber
Proteoglycan
Capillary
bundles
filaments
different substances varies according to their molecu-
Figure 16-4
lar diameters.
Structure of the interstitium. Proteoglycan filaments are every-
Table 16-1 gives the relative permeabilities of the
where in the spaces between the collagen fiber bundles. Free fluid
capillary pores in skeletal muscle for substances com-
vesicles and small amounts of free fluid in the form of rivulets
monly encountered, demonstrating, for instance, that
occasionally also occur.
the permeability for glucose molecules is 0.6 times that
for water molecules, whereas the permeability for
albumin molecules is very, very slight, only 1/1000 that
for water molecules.
to move into the blood and to be carried away from
A word of caution must be issued at this point.
the tissues.
The capillaries in different tissues have extreme dif-
The rates of diffusion through the capillary mem-
ferences in their permeabilities. For instance, the mem-
branes of most nutritionally important substances are
brane of the liver capillary sinusoids is so permeable
so great that only slight concentration differences
that even plasma proteins pass freely through these
suffice to cause more than adequate transport between
walls, almost as easily as water and other substances.
the plasma and interstitial fluid. For instance, the con-
Also, the permeability of the renal glomerular mem-
centration of oxygen in the interstitial fluid immedi-
brane for water and electrolytes is about 500 times the
ately outside the capillary is no more than a few per
permeability of the muscle capillaries, but this is not
cent less than its concentration in the plasma of the
true for the plasma proteins; for these, the capillary
blood, yet this slight difference causes enough oxygen
permeabilities are very slight, as in other tissues and
to move from the blood into the interstitial spaces to
organs. When we study these different organs later in
provide all the oxygen required for tissue metabolism,
this text, it should become clear why some tissues—the
often as much as several liters of oxygen per minute
liver, for instance—require greater degrees of capillary
during very active states of the body.
permeability than others to transfer tremendous
amounts of nutrients between the blood and liver
The Interstitium and
parenchymal cells, and the kidneys to allow filtration
of large quantities of fluid for formation of urine.
Interstitial Fluid
Effect of Concentration Difference on Net Rate of Diffusion
About one sixth of the total volume of the body con-
Through the Capillary Membrane. The “net” rate of diffu-
sists of spaces between cells, which collectively are
sion of a substance through any membrane is propor-
called the interstitium. The fluid in these spaces is the
tional to the concentration difference of the substance
interstitial fluid.
between the two sides of the membrane. That is, the
The structure of the interstitium is shown in Figure
greater the difference between the concentrations of
16-4. It contains two major types of solid structures:
any given substance on the two sides of the capillary
(1) collagen fiber bundles and (2) proteoglycan fila-
membrane, the greater the net movement of the sub-
ments. The collagen fiber bundles extend long dis-
stance in one direction through the membrane. For
tances in the interstitium. They are extremely strong
instance, the concentration of oxygen in capillary
and therefore provide most of the tensional strength
blood is normally greater than in the interstitial fluid.
of the tissues. The proteoglycan filaments, however, are
Therefore, large quantities of oxygen normally move
extremely thin coiled or twisted molecules composed
from the blood toward the tissues. Conversely, the con-
of about 98 per cent hyaluronic acid and 2 per cent
centration of carbon dioxide is greater in the tissues
protein. These molecules are so thin that they can
than in the blood, which causes excess carbon dioxide
never be seen with a light microscope and are difficult
Chapter 16
The Microcirculation and the Lymphatic System
185
to demonstrate even with the electron microscope.
Capillary
Plasma colloid
Nevertheless, they form a mat of very fine reticular fil-
pressure
osmotic pressure
aments aptly described as a “brush pile.”
(Pc)
(Pp)
“Gel” in the Interstitium. The fluid in the interstitium is
derived by filtration and diffusion from the capillaries.
It contains almost the same constituents as plasma
except for much lower concentrations of proteins
Interstitial
Interstitial fluid
because proteins do not pass outward through the
fluid pressure
colloid osmotic pressure
pores of the capillaries with ease. The interstitial fluid
(Pif)
(Pif)
is entrapped mainly in the minute spaces among the
proteoglycan filaments. This combination of proteo-
Figure 16-5
glycan filaments and fluid entrapped within them has
the characteristics of a gel and therefore is called tissue
Fluid pressure and colloid osmotic pressure forces operate at the
capillary membrane, tending to move fluid either outward or
gel.
inward through the membrane pores.
Because of the large number of proteoglycan fila-
ments, it is difficult for fluid to flow easily through the
tissue gel. Instead, it mainly diffuses through the gel;
that is, it moves molecule by molecule from one place
of fluid volume from the blood into the interstitial
to another by kinetic, thermal motion rather than by
spaces.
large numbers of molecules moving together.
Also important is the lymphatic system, which
Diffusion through the gel occurs about 95 to 99 per
returns to the circulation the small amounts of excess
cent as rapidly as it does through free fluid. For the
protein and fluid that leak from the blood into the
short distances between the capillaries and the tissue
interstitial spaces. In the remainder of this chapter, we
cells, this diffusion allows rapid transport through the
discuss the mechanisms that control capillary filtration
interstitium not only of water molecules but also of
and lymph flow function together to regulate the
electrolytes, small molecular weight nutrients, cellular
respective volumes of the plasma and the interstitial
excreta, oxygen, carbon dioxide, and so forth.
fluid.
“Free” Fluid in the Interstitium. Although almost all the
Four Primary Hydrostatic and Colloid Osmotic Forces Determine
fluid in the interstitium normally is entrapped within
Fluid Movement Through the Capillary Membrane. Figure
the tissue gel, occasionally small rivulets of “free” fluid
16-5 shows the four primary forces that determine
and small free fluid vesicles are also present, which
whether fluid will move out of the blood into the inter-
means fluid that is free of the proteoglycan molecules
stitial fluid or in the opposite direction. These forces,
and therefore can flow freely. When a dye is injected
called “Starling forces” in honor of the physiologist
into the circulating blood, it often can be seen to flow
who first demonstrated their importance, are:
through the interstitium in the small rivulets, usually
1. The capillary pressure (Pc), which tends to force
coursing along the surfaces of collagen fibers or sur-
fluid outward through the capillary membrane.
faces of cells.
2. The interstitial fluid pressure (Pif), which tends
The amount of “free” fluid present in normal tissues
to force fluid inward through the capillary
is slight, usually much less than 1 per cent. Conversely,
membrane when Pif is positive but outward when
when the tissues develop edema, these small pockets
Pif is negative.
and rivulets of free fluid expand tremendously until one
3. The capillary plasma colloid osmotic pressure
half or more of the edema fluid becomes freely flowing
(Pp), which tends to cause osmosis of fluid inward
fluid independent of the proteoglycan filaments.
through the capillary membrane.
4. The interstitial fluid colloid osmotic pressure (Pif),
which tends to cause osmosis of fluid outward
Fluid Filtration Across
through the capillary membrane.
Capillaries Is Determined
If the sum of these forces, the net filtration pressure,
is positive, there will be a net fluid filtration across the
by Hydrostatic and Colloid
capillaries. If the sum of the Starling forces is negative,
Osmotic Pressures, and
there will be a net fluid absorption from the intersti-
Capillary Filtration Coefficient
tial spaces into the capillaries. The net filtration pres-
sure (NFP) is calculated as:
The hydrostatic pressure in the capillaries tends to
NFP = Pc - Pif - Pp + Pif
force fluid and its dissolved substances through the
capillary pores into the interstitial spaces. Conversely,
As discussed later, the NFP is slightly positive under
osmotic pressure caused by the plasma proteins
normal conditions, resulting in a net filtration of fluid
(called colloid osmotic pressure) tends to cause fluid
across the capillaries into the interstitial space in most
movement by osmosis from the interstitial spaces
organs. The rate of fluid filtration in a tissue is also
into the blood. This osmotic pressure exerted by
determined by the number and size of the pores in
the plasma proteins normally prevents significant loss
each capillary as well as the number of capillaries in
186
Unit IV The Circulation
which blood is flowing. These factors are usually
expressed together as the capillary filtration coefficient
(Kf). The Kf is therefore a measure of the capacity of
the capillary membranes to filter water for a given
NFP and is usually expressed as ml/min per mm Hg
net filtration pressure.
The rate of capillary fluid filtration is therefore
determined as:
Filtration=K ¥NFP
f
In the following sections we discuss in detail each
of the forces that determine the rate of capillary fluid
filtration.
Gut
Arterial pressure
Capillary Hydrostatic Pressure
Venous pressure
Two experimental methods have been used to esti-
mate the capillary hydrostatic pressure:
(1) direct
micropipette cannulation of the capillaries, which has
100
given an average mean capillary pressure of about 25
mm Hg, and (2) indirect functional measurement of the
capillary pressure, which has given a capillary pressure
80
averaging about 17 mm Hg.
Micropipette Method for Measuring Capillary Pressure. To
60
measure pressure in a capillary by cannulation, a
microscopic glass pipette is thrust directly into the cap-
40
illary, and the pressure is measured by an appropriate
micromanometer system. Using this method, capillary
Capillary pressure
pressures have been measured in capillaries of
= 17 mm Hg
20
exposed tissues of animals and in large capillary loops
of the eponychium at the base of the fingernail in
humans. These measurements have given pressures of
0
30 to 40 mm Hg in the arterial ends of the capillaries,
100
50
0
Arterial pressure
- venous pressure
10 to 15 mm Hg in the venous ends, and about 25 mm
Hg in the middle.
Figure 16-6
Isogravimetric Method for Indirectly Measuring “Functional”
Isogravimetric method for measuring capillary pressure.
Capillary Pressure. Figure
16-6 demonstrates an iso-
gravimetric method for indirectly estimating capillary
pressure. This figure shows a section of gut held up by
one arm of a gravimetric balance. Blood is perfused
fluid through the capillary walls would have occurred.
through the blood vessels of the gut wall. When the
Thus, in a roundabout way, the “functional” capillary
arterial pressure is decreased, the resulting decrease in
pressure is measured to be about 17 mm Hg.
capillary pressure allows the osmotic pressure of the
plasma proteins to cause absorption of fluid out of the
Why Is the Functional Capillary Pressure Lower than Capillary
gut wall and makes the weight of the gut decrease. This
Pressure Measured by the Micropipette Method? It is clear
immediately causes displacement of the balance arm.
that the aforementioned two methods do not give the
To prevent this weight decrease, the venous pressure
same capillary pressure. However, the isogravimetric
is increased an amount sufficient to overcome the
method determines the capillary pressure that exactly
effect of decreasing the arterial pressure. In other
balances all the forces tending to move fluid into or
words, the capillary pressure is kept constant while
out of the capillaries. Because such a balance of forces
simultaneously (1) decreasing the arterial pressure
is the normal state, the average functional capillary
and (2) increasing the venous pressure.
pressure must be close to the pressure measured by
In the graph in the lower part of the figure, the
the isogravimetric method. Therefore, one is justified
changes in arterial and venous pressures that exactly
in believing that the true functional capillary pressure
nullify all weight changes are shown. The arterial and
averages about 17 mm Hg.
venous lines meet each other at a value of 17 mm Hg.
It is easy to explain why the cannulation methods
Therefore, the capillary pressure must have remained
give higher pressure values. The most important
at this same level of
17 mm Hg throughout these
reason is that these measurements usually are made
maneuvers; otherwise, either filtration or absorption of
in capillaries whose arterial ends are open and when
Chapter 16
The Microcirculation and the Lymphatic System
187
blood is actively flowing into the capillary. However, it
protruding from its end. The cotton fibers form a “wick”
should be recalled from the earlier discussion of cap-
that makes excellent contact with the tissue fluids and
illary vasomotion that the metarterioles and precapil-
transmits interstitial fluid pressure into the Teflon tube:
the pressure can then be measured from the tube by
lary sphincters normally are closed during a large part
usual manometric means. Pressures measured by this
of the vasomotion cycle. When closed, the pressure in
technique in loose subcutaneous tissue also have been
the capillaries beyond the closures should be almost
negative, usually measuring -1 to -3 mm Hg.
equal to the pressure at the venous ends of the capil-
laries, about
10 mm Hg. Therefore, when averaged
Interstitial Fluid Pressures in Tightly
over a period of time, one would expect the functional
Encased Tissues
mean capillary pressure to be much nearer to the pres-
Some tissues of the body are surrounded by tight
sure in the venous ends of the capillaries than to the
encasements, such as the cranial vault around the
pressure in the arterial ends.
brain, the strong fibrous capsule around the kidney, the
There are two other reasons why the functional cap-
fibrous sheaths around the muscles, and the sclera
illary pressure is less than the values measured by can-
around the eye. In most of these, regardless of the
nulation. First, there are far more capillaries nearer to
method used for measurement, the interstitial fluid
the venules than to the arterioles. Second, the venous
pressures are usually positive. However, these intersti-
capillaries are several times as permeable as the arte-
tial fluid pressures almost invariably are still less than
rial capillaries. Both of these effects further decrease
the pressures exerted on the outsides of the tissues by
the functional capillary pressure to a lower value.
their encasements. For instance, the cerebrospinal fluid
pressure surrounding the brain of an animal lying on
its side averages about +10 mm Hg, whereas the brain
Interstitial Fluid Hydrostatic Pressure
interstitial fluid pressure averages about +4 to +6 mm
Hg. In the kidneys, the capsular pressure surrounding
As is true for the measurement of capillary pressure,
the kidney averages about +13 mm Hg, whereas the
there are several methods for measuring interstitial
reported renal interstitial fluid pressures have averaged
fluid pressure, and each of these gives slightly differ-
about +6 mm Hg. Thus, if one remembers that the
ent values but usually values that are a few millime-
pressure exerted on the skin is atmospheric pressure,
ters of mercury less than atmospheric pressure, that is,
which is considered to be zero pressure, one might for-
values called negative interstitial fluid pressure. The
mulate a general rule that the normal interstitial fluid
methods most widely used have been (1) direct can-
pressure is usually several millimeters of mercury neg-
nulation of the tissues with a micropipette, (2) meas-
ative with respect to the pressure that surrounds each
urement of the pressure from implanted perforated
tissue.
capsules, and (3) measurement of the pressure from a
cotton wick inserted into the tissue.
Is the True Interstitial Fluid Pressure in Loose
Subcutaneous Tissue Subatmospheric?
Measurement of Interstitial Fluid Pressure Using the
The concept that the interstitial fluid pressure is sub-
Micropipette. The same type of micropipette used for
atmospheric in many if not most tissues of the body
measuring capillary pressure can also be used in some
began with clinical observations that could not be
tissues for measuring interstitial fluid pressure. The tip
explained by the previously held concept that intersti-
of the micropipette is about 1 micrometer in diameter,
tial fluid pressure was always positive. Some of the per-
but even this is 20 or more times larger than the sizes
tinent observations are the following:
of the spaces between the proteoglycan filaments of the
1. When a skin graft is placed on a concave surface
interstitium. Therefore, the pressure that is measured is
probably the pressure in a free fluid pocket.
of the body, such as in an eye socket after
The first pressures measured using the micropipette
removal of the eye, before the skin becomes
method ranged from -1 to +2 mm Hg but were usually
attached to the sublying socket, fluid tends to
slightly positive. With experience and improved equip-
collect underneath the graft. Also, the skin
ment for making such measurements, more recent pres-
attempts to shorten, with the result that it tends to
sures have averaged about -2 mm Hg, giving average
pull it away from the concavity. Nevertheless,
pressure values in loose tissues, such as skin, that are
some negative force underneath the skin causes
slightly less than atmospheric pressure.
absorption of the fluid and usually literally pulls
the skin back into the concavity.
Measurement of Interstitial Free Fluid Pressure in Implanted Per-
2. Less than 1 mm Hg of positive pressure is
forated Hollow Capsules. Interstitial free fluid pressure
required to inject tremendous volumes of fluid
measured by this method when using 2-centimeter
into loose subcutaneous tissues, such as beneath
diameter capsules in normal loose subcutaneous tissue
the lower eyelid, in the axillary space, and in the
averages about -6 mm Hg, but with smaller capsules,
the values are not greatly different from the -2 mm Hg
scrotum. Amounts of fluid calculated to be more
measured by the micropipette in Figure 16-7.
than 100 times the amount of fluid normally in the
interstitial space, when injected into these areas,
Measurement of Interstitial Free Fluid Pressure by Means of a
cause no more than about 2 mm Hg of positive
Cotton Wick. Still another method is to insert into a
pressure. The importance of these observations is
tissue a small Teflon tube with about eight cotton fibers
that they show that such tissues do not have
188
Unit IV The Circulation
strong fibers that can prevent the accumulation of
through the capillary pores, it is the proteins of the
fluid. Therefore, some other mechanism, such as a
plasma and interstitial fluids that are responsible for
negative fluid pressure system, must be available
the osmotic pressures on the two sides of the capillary
to prevent such fluid accumulation.
membrane. To distinguish this osmotic pressure from
3. In most natural cavities of the body where there
that which occurs at the cell membrane, it is called
is free fluid in dynamic equilibrium with the
either colloid osmotic pressure or oncotic pressure. The
surrounding interstitial fluids, the pressures that
term “colloid” osmotic pressure is derived from the
have been measured have been negative. Some of
fact that a protein solution resembles a colloidal solu-
these are the following:
tion despite the fact that it is actually a true molecu-
lar solution.
Intrapleural space: -8 mm Hg
Joint synovial spaces: -4 to -6 mm Hg
Normal Values for Plasma Colloid Osmotic Pressure. The
Epidural space: -4 to -6 mm Hg
colloid osmotic pressure of normal human plasma
4. The implanted capsule for measuring the
averages about 28 mm Hg; 19 mm of this is caused by
interstitial fluid pressure can be used to record
molecular effects of the dissolved protein and 9 mm by
dynamic changes in this pressure. The changes are
the Donnan effect—that is, extra osmotic pressure
approximately those that one would calculate to
caused by sodium, potassium, and the other cations
occur (1) when the arterial pressure is increased
held in the plasma by the proteins.
or decreased, (2) when fluid is injected into the
surrounding tissue space, or (3) when a highly
Effect of the Different Plasma Proteins on Colloid Osmotic
Pressure. The plasma proteins are a mixture that con-
concentrated colloid osmotic agent is injected into
tains albumin, with an average molecular weight of
the blood to absorb fluid from the tissue spaces. It
69,000; globulins, 140,000; and fibrinogen, 400,000. Thus,
is not likely that these dynamic changes could be
1 gram of globulin contains only half as many molecules
recorded this accurately unless the capsule
as 1 gram of albumin, and 1 gram of fibrinogen contains
pressure closely approximated the true interstitial
only one sixth as many molecules as 1 gram of albumin.
pressure.
It should be recalled from the discussion of osmotic
pressure in Chapter 4 that osmotic pressure is deter-
Summary—An Average Value for Negative Interstitial Fluid
mined by the number of molecules dissolved in a fluid
Pressure in Loose Subcutaneous Tissue. Although the
rather than by the mass of these molecules. Therefore,
aforementioned different methods give slightly differ-
when corrected for number of molecules rather than
mass, the following chart gives both the relative mass
ent values for interstitial fluid pressure, there currently
concentrations (g/dl) of the different types of proteins
is a general belief among most physiologists that the
in normal plasma and their respective contributions to
true interstitial fluid pressure in loose subcutaneous
the total plasma colloid osmotic pressure (Pp).
tissue is slightly less subatmospheric, averaging about
-3 mm Hg.
Pumping by the Lymphatic System Is the
g/dl
Pp (mm Hg)
Basic Cause of the Negative Interstitial
Albumin
4.5
21.8
Fluid Pressure
Globulins
2.5
6.0
The lymphatic system is discussed later in the chapter,
Fibrinogen
0.3
0.2
but we need to understand here the basic role that this
Total
7.3
28.0
system plays in determining interstitial fluid pressure.
The lymphatic system is a “scavenger” system that
removes excess fluid, excess protein molecules, debris,
Thus, about 80 per cent of the total colloid osmotic
and other matter from the tissue spaces. Normally,
pressure of the plasma results from the albumin frac-
when fluid enters the terminal lymphatic capillaries,
tion, 20 per cent from the globulins, and almost none
the lymph vessel walls automatically contract for a few
from the fibrinogen. Therefore, from the point of view
seconds and pump the fluid into the blood circulation.
of capillary and tissue fluid dynamics, it is mainly
This overall process creates the slight negative pres-
albumin that is important.
sure that has been measured for fluid in the interstitial
spaces.
Interstitial Fluid Colloid
Osmotic Pressure
Plasma Colloid Osmotic Pressure
Although the size of the usual capillary pore is smaller
Proteins in the Plasma Cause Colloid Osmotic Pressure. In the
than the molecular sizes of the plasma proteins, this is
basic discussion of osmotic pressure in Chapter 4, it
not true of all the pores. Therefore, small amounts of
was pointed out that only those molecules or ions that
plasma proteins do leak through the pores into the
fail to pass through the pores of a semiperme-
interstitial spaces.
able membrane exert osmotic pressure. Because the
The total quantity of protein in the entire 12 liters
proteins are the only dissolved constituents in the
of interstitial fluid of the body is slightly greater than
plasma and interstitial fluids that do not readily pass
the total quantity of protein in the plasma itself, but
Chapter 16
The Microcirculation and the Lymphatic System
189
because this volume is four times the volume of
mm Hg
plasma, the average protein concentration of the inter-
stitial fluid is usually only 40 per cent of that in plasma,
Forces tending to move fluid inward:
or about 3 g/dl. Quantitatively, one finds that the
Plasma colloid osmotic pressure
28
average interstitial fluid colloid osmotic pressure for
total inward force
28
this concentration of proteins is about 8 mm Hg.
Forces tending to move fluid outward:
Capillary pressure (venous end of capillary)
10
Negative interstitial free fluid pressure
3
Interstitial fluid colloid osmotic pressure
8
Exchange of Fluid Volume Through
total outward force
21
the Capillary Membrane
Summation of forces:
Inward
28
Now that the different factors affecting fluid move-
Outward
21
ment through the capillary membrane have been
net inward force
7
discussed, it is possible to put all these together to
see how the capillary system maintains normal fluid
Thus, the force that causes fluid to move into the
volume distribution between the plasma and the inter-
capillary,
28 mm Hg, is greater than that opposing
stitial fluid.
reabsorption, 21 mm Hg. The difference, 7 mm Hg, is
The average capillary pressure at the arterial ends
the net reabsorption pressure at the venous ends of the
of the capillaries is 15 to 25 mm Hg greater than at the
capillaries. This reabsorption pressure is considerably
venous ends. Because of this difference, fluid “filters”
less than the filtration pressure at the capillary arterial
out of the capillaries at their arterial ends, but at their
ends, but remember that the venous capillaries are
venous ends fluid is reabsorbed back into the capillar-
more numerous and more permeable than the arterial
ies. Thus, a small amount of fluid actually “flows”
capillaries, so that less reabsorption pressure is
through the tissues from the arterial ends of the cap-
required to cause inward movement of fluid.
illaries to the venous ends. The dynamics of this flow
The reabsorption pressure causes about nine tenths
are as follows.
of the fluid that has filtered out of the arterial ends of
the capillaries to be reabsorbed at the venous ends.
Analysis of the Forces Causing Filtration at the Arterial End of
The remaining one tenth flows into the lymph vessels
the Capillary. The approximate average forces operative
and returns to the circulating blood.
at the arterial end of the capillary that cause movement
through the capillary membrane are shown as follows:
Starling Equilibrium for
Capillary Exchange
mm Hg
E. H. Starling pointed out over a century ago that
Forces tending to move fluid outward:
Capillary pressure (arterial end of capillary)
30
under normal conditions, a state of near-equilibrium
Negative interstitial free fluid pressure
3
exists at the capillary membrane. That is, the amount
Interstitial fluid colloid osmotic pressure
8
of fluid filtering outward from the arterial ends of cap-
total outward force
41
illaries equals almost exactly the fluid returned to the
Forces tending to move fluid inward:
circulation by absorption. The slight disequilibrium
Plasma colloid osmotic pressure
28
that does occur accounts for the small amount of fluid
total inward force
28
that is eventually returned by way of the lymphatics.
Summation of forces:
The following chart shows the principles of the Star-
Outward
41
ling equilibrium. For this chart, the pressures in the arte-
Inward
28
rial and venous capillaries are averaged to calculate
net outward force (at arterial end)
13
mean functional capillary pressure for the entire length
of the capillary. This calculates to be 17.3 mm Hg.
Thus, the summation of forces at the arterial end of
mm Hg
the capillary shows a net filtration pressure of 13 mm
Mean forces tending to move fluid outward:
Hg, tending to move fluid outward through the capil-
Mean capillary pressure
17.3
lary pores.
Negative interstitial free fluid pressure
3.0
This 13 mm Hg filtration pressure causes, on the
Interstitial fluid colloid osmotic pressure
8.0
average, about 1/200 of the plasma in the flowing blood
total outward force
28.3
to filter out of the arterial ends of the capillaries into
Mean force tending to move fluid inward:
the interstitial spaces each time the blood passes
Plasma colloid osmotic pressure
28.0
through the capillaries.
total inward force
28.0
Summation of mean forces:
Analysis of Reabsorption at the Venous End of the Capillary.
Outward
28.3
The low blood pressure at the venous end of the cap-
Inward
28.0
illary changes the balance of forces in favor of absorp-
net outward force
0.3
tion as follows:
190
Unit IV The Circulation
Thus, for the total capillary circulation, we find a
Effect of Abnormal Imbalance of Forces
near-equilibrium between the total outward forces,
at the Capillary Membrane
28.3 mm Hg, and the total inward force, 28.0 mm Hg.
If the mean capillary pressure rises above 17 mm Hg,
This slight imbalance of forces, 0.3 mm Hg, causes
the net force tending to cause filtration of fluid into the
slightly more filtration of fluid into the interstitial
tissue spaces rises. Thus, a 20 mm Hg rise in mean cap-
spaces than reabsorption. This slight excess of filtration
illary pressure causes an increase in net filtration pres-
is called net filtration, and it is the fluid that must be
sure from 0.3 mm Hg to 20.3 mm Hg, which results in
returned to the circulation through the lymphatics. The
68 times as much net filtration of fluid into the inter-
normal rate of net filtration in the entire body is only
stitial spaces as normally occurs. To prevent accumu-
about 2 milliliters per minute.
lation of excess fluid in these spaces would require 68
times the normal flow of fluid into the lymphatic
system, an amount that is 2 to 5 times too much for the
Filtration Coefficient. In the above example, an average
lymphatics to carry away. As a result, fluid will begin
net imbalance of forces at the capillary membranes
to accumulate in the interstitial spaces, and edema will
of 0.3 mm Hg causes net fluid filtration in the entire
result.
body of 2 ml/min. Expressing this for each millimeter
Conversely, if the capillary pressure falls very low,
of mercury imbalance, one finds a net filtration rate
net reabsorption of fluid into the capillaries will occur
of 6.67 milliliters of fluid per minute per millimeter of
instead of net filtration, and the blood volume will
mercury for the entire body. This is called the whole
increase at the expense of the interstitial fluid volume.
body capillary filtration coefficient.
These effects of imbalance at the capillary membrane
The filtration coefficient can also be expressed for
in relation to the development of different kinds of
separate parts of the body in terms of rate of filtration
edema are discussed in Chapter 25.
per minute per millimeter of mercury per 100 grams
of tissue. On this basis, the filtration coefficient of the
average tissue is about 0.01 ml/min/mm Hg/100 g of
Lymphatic System
tissue. But, because of extreme differences in perme-
abilities of the capillary systems in different tissues,
The lymphatic system represents an accessory route
this coefficient varies more than 100-fold among the
through which fluid can flow from the interstitial
different tissues. It is very small in both brain and
spaces into the blood. Most important, the lymphatics
muscle, moderately large in subcutaneous tissue, large
can carry proteins and large particulate matter away
in the intestine, and extreme in the liver and glomeru-
from the tissue spaces, neither of which can be
lus of the kidney where the pores are either numerous
removed by absorption directly into the blood capil-
or wide open. By the same token, the permeation
laries. This return of proteins to the blood from the
of proteins through the capillary membranes varies
interstitial spaces is an essential function without
greatly as well. The concentration of protein in the
which we would die within about 24 hours.
interstitial fluid of muscles is about 1.5 g/dl; in subcu-
taneous tissue, 2 g/dl; in intestine, 4 g/dl; and in liver,
6 g/dl.
Lymph Channels of the Body
Almost all tissues of the body have special lymph chan-
nels that drain excess fluid directly from the interstitial
spaces. The exceptions include the superficial portions
of the skin, the central nervous system, the endomysium
Implanted
Blood
of muscles, and the bones. But, even these tissues have
Skin
capsule
vessels
minute interstitial channels called prelymphatics
through which interstitial fluid can flow; this fluid even-
tually empties either into lymphatic vessels or, in the
case of the brain, into the cerebrospinal fluid and then
directly back into the blood.
Essentially all the lymph vessels from the lower part
of the body eventually empty into the thoracic duct,
which in turn empties into the blood venous system at
the juncture of the left internal jugular vein and left sub-
clavian vein, as shown in Figure 16-8.
Lymph from the left side of the head, the left arm, and
parts of the chest region also enters the thoracic duct
before it empties into the veins.
Lymph from the right side of the neck and head, the
To
right arm, and parts of the right thorax enters the right
measure
lymph duct (much smaller than the thoracic duct), which
pressure
Fluid filled cavity
empties into the blood venous system at the juncture of
the right subclavian vein and internal jugular vein.
FIGURE 16-7
Perforated capsule method for measuring interstitial fluid
Terminal Lymphatic Capillaries and Their Permeability. Most
pressure.
of the fluid filtering from the arterial ends of blood
Chapter 16
The Microcirculation and the Lymphatic System
191
Cervical nodes
Sentinel node
Subclavian vein
R. lymphatic duct
Thoracic duct
Axillary nodes
Cisterna chyli
Abdominal nodes
Inguinal nodes
Peripheral lymphatics
FIGURE 16-8
Lymphatic system.
capillaries flows among the cells and finally is reab-
in such a way that the overlapping edge is free to flap
sorbed back into the venous ends of the blood capil-
inward, thus forming a minute valve that opens to the
laries; but on the average, about 1/10 of the fluid
interior of the lymphatic capillary. Interstitial fluid,
instead enters the lymphatic capillaries and returns to
along with its suspended particles, can push the valve
the blood through the lymphatic system rather than
open and flow directly into the lymphatic capillary. But
through the venous capillaries. The total quantity of all
this fluid has difficulty leaving the capillary once it has
this lymph is normally only 2 to 3 liters each day.
entered because any backflow closes the flap valve.
The fluid that returns to the circulation by way of
Thus, the lymphatics have valves at the very tips of the
the lymphatics is extremely important because sub-
terminal lymphatic capillaries as well as valves along
stances of high molecular weight, such as proteins,
their larger vessels up to the point where they empty
cannot be absorbed from the tissues in any other way,
into the blood circulation.
although they can enter the lymphatic capillaries
almost unimpeded. The reason for this is a special
structure of the lymphatic capillaries, demonstrated in
Formation of Lymph
Figure 16-9. This figure shows the endothelial cells of
the lymphatic capillary attached by anchoring fila-
Lymph is derived from interstitial fluid that flows into
ments to the surrounding connective tissue. At the
the lymphatics. Therefore, lymph as it first enters the
junctions of adjacent endothelial cells, the edge of one
terminal lymphatics has almost the same composition
endothelial cell overlaps the edge of the adjacent cell
as the interstitial fluid.
192
Unit IV The Circulation
4
Endothelial cells
Valves
2
2 times/
7 times/
Anchoring filaments
mm Hg
mm Hg
0
FIGURE 16-9
-6
-4
-2
0
2
4
Special structure of the lymphatic capillaries that permits passage
PT (mm Hg)
of substances of high molecular weight into the lymph.
The protein concentration in the interstitial fluid of
FIGURE 16-10
most tissues averages about 2 g/dl, and the protein con-
Relation between interstitial fluid pressure and lymph flow in the
centration of lymph flowing from these tissues is near
leg of a dog. Note that lymph flow reaches a maximum when the
this value. Conversely, lymph formed in the liver has a
interstitial pressure, PT, rises slightly above atmospheric pressure
protein concentration as high as 6 g/dl, and lymph
(0 mm Hg). (Courtesy Drs. Harry Gibson and Aubrey Taylor.)
formed in the intestines has a protein concentration as
high as 3 to 4 g/dl. Because about two thirds of all
lymph normally is derived from the liver and intes-
tines, the thoracic duct lymph, which is a mixture of
value of -6 mm Hg. Then, as the pressure rises to 0 mm
lymph from all areas of the body, usually has a protein
Hg (atmospheric pressure), flow increases more than
concentration of 3 to 5 g/dl.
20-fold. Therefore, any factor that increases interstitial
The lymphatic system is also one of the major routes
fluid pressure also increases lymph flow if the lymph
for absorption of nutrients from the gastrointestinal
vessels are functioning normally. Such factors include
tract, especially for absorption of virtually all fats in
the following:
food, as discussed in Chapter 65. Indeed, after a fatty
∑ Elevated capillary pressure
meal, thoracic duct lymph sometimes contains as much
∑ Decreased plasma colloid osmotic pressure
as 1 to 2 per cent fat.
∑ Increased interstitial fluid colloid osmotic pressure
Finally, even large particles, such as bacteria, can
∑ Increased permeability of the capillaries
push their way between the endothelial cells of the
All of these cause a balance of fluid exchange at the
lymphatic capillaries and in this way enter the lymph.
blood capillary membrane to favor fluid movement
As the lymph passes through the lymph nodes, these
into the interstitium, thus increasing interstitial fluid
particles are almost entirely removed and destroyed,
volume, interstitial fluid pressure, and lymph flow all
as discussed in Chapter 33.
at the same time.
However, note in Figure 16-10 that when the inter-
stitial fluid pressure becomes 1 or 2 millimeters greater
Rate of Lymph Flow
than atmospheric pressure (greater than 0 mm Hg),
lymph flow fails to rise any further at still higher pres-
About 100 milliliters per hour of lymph flows through
sures. This results from the fact that the increasing
the thoracic duct of a resting human, and approxi-
tissue pressure not only increases entry of fluid into
mately another 20 milliliters flows into the circulation
the lymphatic capillaries but also compresses the
each hour through other channels, making a total esti-
outside surfaces of the larger lymphatics, thus imped-
mated lymph flow of about 120 ml/hr or 2 to 3 liters
ing lymph flow. At the higher pressures, these two
per day.
factors balance each other almost exactly, so that
lymph flow reaches what is called the “maximum
Effect of Interstitial Fluid Pressure on Lymph Flow. Figure
lymph flow rate.” This is illustrated by the upper level
16-10 shows the effect of different levels of interstitial
plateau in Figure 16-10.
fluid pressure on lymph flow as measured in dog legs.
Note that normal lymph flow is very little at intersti-
Lymphatic Pump Increases Lymph Flow. Valves exist in all
tial fluid pressures more negative than the normal
lymph channels; typical valves are shown in Figure
Chapter 16
The Microcirculation and the Lymphatic System
193
Pores
Valves
Lymphatic
capillaries
Collecting
FIGURE 16-11
lymphatic
Structure of lymphatic capillaries
and a collecting lymphatic,
showing also the lymphatic
valves.
16-11 in collecting lymphatics into which the lym-
through the junctions between the endothelial cells.
phatic capillaries empty.
Then, when the tissue is compressed, the pressure
Motion pictures of exposed lymph vessels, both
inside the capillary increases and causes the overlap-
in animals and in human beings, show that when a
ping edges of the endothelial cells to close like valves.
collecting lymphatic or larger lymph vessel becomes
Therefore, the pressure pushes the lymph forward into
stretched with fluid, the smooth muscle in the wall of
the collecting lymphatic instead of backward through
the vessel automatically contracts. Furthermore, each
the cell junctions.
segment of the lymph vessel between successive valves
The lymphatic capillary endothelial cells also
functions as a separate automatic pump. That is, even
contain a few contractile actomyosin filaments. In
slight filling of a segment causes it to contract, and the
some animal tissues (e.g., the bat’s wing) these fila-
fluid is pumped through the next valve into the next
ments have been observed to cause rhythmical con-
lymphatic segment. This fills the subsequent segment,
traction of the lymphatic capillaries in the same way
and a few seconds later it, too, contracts, the process
that many of the small blood and larger lymphatic
continuing all along the lymph vessel until the fluid is
vessels also contract rhythmically. Therefore, it is
finally emptied into the blood circulation. In a very
probable that at least part of lymph pumping results
large lymph vessel such as the thoracic duct, this lym-
from lymph capillary endothelial cell contraction
phatic pump can generate pressures as great as 50 to
in addition to contraction of the larger muscular
100 mm Hg.
lymphatics.
Pumping Caused by External Intermittent Compression
Summary of Factors That Determine Lymph Flow. From the
of the Lymphatics. In addition to the pumping caused
above discussion, one can see that the two primary
by intrinsic intermittent contraction of the lymph
factors that determine lymph flow are (1) the intersti-
vessel walls, any external factor that intermittently
tial fluid pressure and (2) the activity of the lymphatic
compresses the lymph vessel also can cause pumping.
pump. Therefore, one can state that, roughly, the rate
In order of their importance, such factors are:
of lymph flow is determined by the product of intersti-
∑ Contraction of surrounding skeletal muscles
tial fluid pressure times the activity of the lymphatic
∑ Movement of the parts of the body
pump.
∑ Pulsations of arteries adjacent to the lymphatics
∑ Compression of the tissues by objects outside the
body
Role of the Lymphatic System in
The lymphatic pump becomes very active during exer-
Controlling Interstitial Fluid Protein
cise, often increasing lymph flow 10- to 30-fold. Con-
versely, during periods of rest, lymph flow is sluggish,
Concentration, Interstitial Fluid
almost zero.
Volume, and Interstitial Fluid Pressure
Lymphatic Capillary Pump. The terminal lymphatic capil-
It is already clear that the lymphatic system functions
lary is also capable of pumping lymph, in addition to
as an “overflow mechanism” to return to the circula-
the lymph pumping by the larger lymph vessels.
tion excess proteins and excess fluid volume from the
As explained earlier in the chapter, the walls of the
tissue spaces. Therefore, the lymphatic system also
lymphatic capillaries are tightly adherent to the sur-
plays a central role in controlling (1) the concentration
rounding tissue cells by means of their anchoring
of proteins in the interstitial fluids, (2) the volume of
filaments. Therefore, each time excess fluid enters
interstitial fluid, and (3) the interstitial fluid pressure.
the tissue and causes the tissue to swell, the anchoring
Let us explain how these factors interact.
filaments pull on the wall of the lymphatic capillary,
First, remember that small amounts of proteins
and fluid flows into the terminal lymphatic capillary
leak continuously out of the blood capillaries into the
194
Unit IV The Circulation
interstitium. Only minute amounts, if any, of the leaked
condition known as edema occurs, which is discussed
proteins return to the circulation by way of the venous
in Chapter 25.
ends of the blood capillaries. Therefore, these proteins
tend to accumulate in the interstitial fluid, and this in
turn increases the colloid osmotic pressure of the
References
interstitial fluids.
Second, the increasing colloid osmotic pressure in
Aukland K, Reed RK: Interstitial-lymphatic mechanisms in
the control of extracellular fluid volume. Physiol Rev 73:1,
the interstitial fluid shifts the balance of forces at the
1993.
blood capillary membranes in favor of fluid filtration
D’Amico G, Bazzi C: Pathophysiology of proteinuria.
into the interstitium. Therefore, in effect, fluid is
Kidney Int 63:809, 2003.
translocated osmotically outward through the capil-
Dejana E: Endothelial cell-cell junctions: happy together.
lary wall by the proteins and into the interstitium, thus
Nat Rev Mol Cell Biol 5:261, 2004.
increasing both interstitial fluid volume and interstitial
Frank PG, Woodman SE, Park DS, Lisanti MP: Caveolin,
fluid pressure.
caveolae, and endothelial cell function. Arterioscler
Third, the increasing interstitial fluid pressure
Thromb Vasc Biol 23:1161, 2003.
greatly increases the rate of lymph flow, as explained
Gashev AA: Physiologic aspects of lymphatic contractile
previously. This in turn carries away the excess inter-
function: current perspectives. Ann N Y Acad Sci 979:178,
2002.
stitial fluid volume and excess protein that has accu-
Guyton AC: Concept of negative interstitial pressure based
mulated in the spaces.
on pressures in implanted perforated capsules. Circ Res
Thus, once the interstitial fluid protein concentra-
12:399, 1963.
tion reaches a certain level and causes a comparable
Guyton AC: Interstitial fluid pressure: II. Pressure-volume
increase in interstitial fluid volume and interstitial
curves of interstitial space. Circ Res 16:452, 1965.
fluid pressure, the return of protein and fluid by way
Guyton AC, Granger HJ, Taylor AE: Interstitial fluid pres-
of the lymphatic system becomes great enough to
sure. Physiol Rev 51:527, 1971.
balance exactly the rate of leakage of these into the
Guyton AC, Prather J, Scheel K, McGehee J: Interstitial fluid
interstitium from the blood capillaries. Therefore, the
pressure: IV. Its effect on fluid movement through the
quantitative values of all these factors reach a steady
capillary wall. Circ Res 19:1022, 1966.
Guyton AC, Scheel K, Murphree D: Interstitial fluid pres-
state; they will remain balanced at these steady state
sure: III. Its effect on resistance to tissue fluid mobility.
levels until something changes the rate of leakage of
Circ Res 19:412, 1966.
proteins and fluid from the blood capillaries.
Guyton AC, Taylor AE, Granger HJ: Circulatory Physiology
II. Dynamics and Control of the Body Fluids. Philadel-
Significance of Negative Interstitial Fluid
phia: WB Saunders Co, 1975.
Pressure as a Means for Holding the
Michel CC, Curry FE: Microvascular permeability. Physiol
Body Tissues Together
Rev 79:703, 1999.
Traditionally, it has been assumed that the different
Miyasaka M, Tanaka T: Lymphocyte trafficking across high
tissues of the body are held together entirely by
endothelial venules: dogmas and enigmas. Nat Rev
connective tissue fibers. However, at many places in
Immunol 4:360, 2004.
Oliver G: Lymphatic vasculature development. Nat Rev
the body, connective tissue fibers are very weak or
Immunol 4:35, 2004.
even absent. This occurs particularly at points where
Rippe B, Rosengren BI, Carlsson O, Venturoli D:
tissues slide over one another, such as the skin sliding
Transendothelial transport: the vesicle controversy. J Vasc
over the back of the hand or over the face. Yet even
Res 39:375, 2002.
at these places, the tissues are held together by the
Taylor AE, Granger DN: Exchange of macromolecules
negative interstitial fluid pressure, which is actually a
across the microcirculation. In: Renkin EM, Michel CC
partial vacuum. When the tissues lose their negative
(eds): Handbook of Physiology. Sec. 2, Vol. IV. Bethesda:
pressure, fluid accumulates in the spaces and the
American Physiological Society, 1984, p 467.
C H A P T E R
1
7
Local and Humoral Control of
Blood Flow by the Tissues
Local Control of Blood
Flow in Response to
Tissue Needs
One of the most fundamental principles of circula-
tory function is the ability of each tissue to control
its own local blood flow in proportion to its meta-
bolic needs.
What are some of the specific needs of the tissues for blood flow? The answer
to this is manyfold, including the following:
1. Delivery of oxygen to the tissues
2. Delivery of other nutrients, such as glucose, amino acids, and fatty acids
3. Removal of carbon dioxide from the tissues
4. Removal of hydrogen ions from the tissues
5. Maintenance of proper concentrations of other ions in the tissues
6. Transport of various hormones and other substances to the different tissues
Certain organs have special requirements. For instance, blood flow to the skin
determines heat loss from the body and in this way helps to control body tem-
perature. Also, delivery of adequate quantities of blood plasma to the kidneys
allows the kidneys to excrete the waste products of the body.
We shall see that most of these factors exert extreme degrees of local blood
flow control.
Variations in Blood Flow in Different Tissues and Organs. Note in Table 17-1 the very
large blood flows in some organs—for example, several hundred milliliters per
minute per 100 grams of thyroid or adrenal gland tissue and a total blood flow
of 1350 ml/min in the liver, which is 95 ml/min/100 g of liver tissue.
Also note the extremely large blood flow through the kidneys—1100 ml/min.
This extreme amount of flow is required for the kidneys to perform their func-
tion of cleansing the blood of waste products.
Conversely, most surprising is the low blood flow to all the inactive muscles
of the body, only a total of 750 ml/min, even though the muscles constitute
between 30 and 40 per cent of the total body mass. In the resting state, the meta-
bolic activity of the muscles is very low, and so also is the blood flow, only
4 ml/min/100 g. Yet, during heavy exercise, muscle metabolic activity can
increase more than 60-fold and the blood flow as much as 20-fold, increasing to
as high as 16,000 ml/min in the body’s total muscle vascular bed (or 80 ml/
min/100 g of muscle).
Importance of Blood Flow Control by the Local Tissues. One might ask the simple ques-
tion: Why not simply allow a very large blood flow all the time through every
tissue of the body, always enough to supply the tissue’s needs whether the activ-
ity of the tissue is little or great? The answer is equally simple: To do this would
require many times more blood flow than the heart can pump.
Experiments have shown that the blood flow to each tissue usually is regu-
lated at the minimal level that will supply the tissue’s requirements— no more,
no less. For instance, in tissues for which the most important requirement is
delivery of oxygen, the blood flow is always controlled at a level only slightly
more than required to maintain full tissue oxygenation but no more than this.
195
196
Unit IV The Circulation
Table 17-1
4
Blood Flow to Different Organs and Tissues Under Basal
Conditions
3
Per cent
ml/min
ml/min/100 g
Brain
14
700
50
Heart
4
200
70
2
Bronchi
2
100
25
Kidneys
22
1100
360
Liver
27
1350
95
Portal
(21)
1050
1
Arterial
(6)
300
Normal level
Muscle (inactive state)
15
750
4
Bone
5
250
3
Skin (cool weather)
6
300
3
0
Thyroid gland
1
50
160
0
1
2
3
4
5
6
7
8
Adrenal glands
0.5
25
300
Rate of metabolism (x normal)
Other tissues
3.5
175
1.3
Total
100.0
5000
Based mainly on data compiled by Dr. L. A. Sapirstein.
Figure 17-1
Effect of increasing rate of metabolism on tissue blood flow.
By controlling local blood flow in such an exact way,
the tissues almost never suffer from oxygen nutritional
deficiency, and yet the workload on the heart is kept
(3) in carbon monoxide poisoning (which poisons the
at a minimum.
ability of hemoglobin to transport oxygen), or (4) in
cyanide poisoning (which poisons the ability of the
tissues to use oxygen), the blood flow through the
Mechanisms of Blood
tissues increases markedly. Figure 17-2 shows that as
Flow Control
the arterial oxygen saturation decreases to about 25
per cent of normal, the blood flow through an isolated
Local blood flow control can be divided into two
leg increases about threefold; that is, the blood flow
phases: (1) acute control and (2) long-term control.
increases almost enough, but not quite enough, to
Acute control is achieved by rapid changes in local
make up for the decreased amount of oxygen in the
vasodilation or vasoconstriction of the arterioles,
blood, thus almost maintaining an exact constant
metarterioles, and precapillary sphincters, occurring
supply of oxygen to the tissues.
within seconds to minutes to provide very rapid main-
Total cyanide poisoning of oxygen usage by a local
tenance of appropriate local tissue blood flow.
tissue area can cause local blood flow to increase as
Long-term control, however, means slow, controlled
much as sevenfold, thus demonstrating the extreme
changes in flow over a period of days, weeks, or even
effect of oxygen deficiency to increase blood flow.
months. In general, these long-term changes provide
There are two basic theories for the regulation of
even better control of the flow in proportion to the
local blood flow when either the rate of tissue metab-
needs of the tissues. These changes come about as a
olism changes or the availability of oxygen changes.
result of an increase or decrease in the physical sizes
They are (1) the vasodilator theory and (2) the oxygen
and numbers of actual blood vessels supplying the
lack theory.
tissues.
Vasodilator Theory for Acute Local Blood Flow Regu-
lation—Possible Special Role of Adenosine. Accord-
Acute Control of Local Blood Flow
ing to this theory, the greater the rate of metabolism
or the less the availability of oxygen or some other
Effect of Tissue Metabolism on Local Blood Flow. Figure
nutrients to a tissue, the greater the rate of formation
17-1 shows the approximate quantitative acute effect
of vasodilator substances in the tissue cells. The
on blood flow of increasing the rate of metabolism in
vasodilator substances then are believed to diffuse
a local tissue, such as in a skeletal muscle. Note that an
through the tissues to the precapillary sphincters,
increase in metabolism up to eight times normal
metarterioles, and arterioles to cause dilation. Some of
increases the blood flow acutely about fourfold.
the different vasodilator substances that have been
suggested are adenosine, carbon dioxide, adenosine
Acute Local Blood Flow Regulation When Oxygen Availability
phosphate compounds, histamine, potassium ions, and
Changes. One of the most necessary of the metabolic
hydrogen tons.
nutrients is oxygen. Whenever the availability of
Most of the vasodilator theories assume that the
oxygen to the tissues decreases, such as (1) at high alti-
vasodilator substance is released from the tissue
tude at the top of a high mountain, (2) in pneumonia,
mainly in response to oxygen deficiency. For instance,
Chapter 17
Local and Humoral Control of Blood Flow by the Tissues
197
Precapillary sphincter
Metarteriole
3
2
1
Sidearm capillary
0
100
75
50
25
Arterial oxygen saturation (per cent)
Figure 17-2
Effect of decreasing arterial oxygen saturation on blood flow
Figure 17-3
through an isolated dog leg.
Diagram of a tissue unit area for explanation of acute local feed-
back control of blood flow, showing a metarteriole passing through
experiments have shown that decreased availability of
the tissue and a sidearm capillary with its precapillary sphincter
oxygen can cause both adenosine and lactic acid (con-
for controlling capillary blood flow.
taining hydrogen ions) to be released into the spaces
between the tissue cells; these substances then cause
intense acute vasodilation and therefore are respon-
sible, or partially responsible, for the local blood flow
several critical facts have made other physiologists
regulation.
favor still another theory, which can be called either
Many physiologists have suggested that the sub-
the oxygen lack theory or, more accurately, the
stance adenosine is the most important of the local
nutrient lack theory (because other nutrients besides
vasodilators for controlling local blood flow. For
oxygen are involved). Oxygen (and other nutrients
example, minute quantities of adenosine are released
as well) is required as one of the metabolic nutrients
from heart muscle cells when coronary blood flow
to cause vascular muscle contraction. Therefore, in the
becomes too little, and this causes enough local vasodi-
absence of adequate oxygen, it is reasonable to
lation in the heart to return coronary blood flow
believe that the blood vessels simply would relax
back to normal. Also, whenever the heart becomes
and therefore naturally dilate. Also, increased utiliza-
more active than normal and the heart’s metabolism
tion of oxygen in the tissues as a result of increased
increases an extra amount, this, too, causes increased
metabolism theoretically could decrease the availabil-
utilization of oxygen, followed by
(1) decreased
ity of oxygen to the smooth muscle fibers in the
oxygen concentration in the heart muscle cells with (2)
local blood vessels, and this, too, would cause local
consequent degradation of adenosine triphosphate
vasodilation.
(ATP), which (3) increases the release of adenosine. It
A mechanism by which the oxygen lack theory
is believed that much of this adenosine leaks out of the
could operate is shown in Figure 17-3. This figure
heart muscle cells to cause coronary vasodilation, pro-
shows a tissue unit, consisting of a metarteriole with a
viding increased coronary blood flow to supply the
single sidearm capillary and its surrounding tissue. At
increased nutrient demands of the active heart.
the origin of the capillary is a precapillary sphincter,
Although research evidence is less clear, many phys-
and around the metarteriole are several other smooth
iologists also have suggested that the same adenosine
muscle fibers. Observing such a tissue under a micro-
mechanism is the most important controller of blood
scope—for example, in a bat’s wing—one sees that the
flow in skeletal muscle and many other tissues as
precapillary sphincters are normally either completely
well as in the heart. The problem with the different
open or completely closed. The number of precapillary
vasodilator theories of local blood flow regulation
sphincters that are open at any given time is roughly
has been the following: It has been difficult to prove
proportional to the requirements of the tissue for
that sufficient quantities of any single vasodilator sub-
nutrition. The precapillary sphincters and metarteri-
stance (including adenosine) are indeed formed in the
oles open and close cyclically several times per minute,
tissues to cause all the measured increase in blood
with the duration of the open phases being propor-
flow. But a combination of several different vasodila-
tional to the metabolic needs of the tissues for oxygen.
tors could increase the blood flow sufficiently.
The cyclical opening and closing is called vasomotion.
Let us explain how oxygen concentration in the
Oxygen Lack Theory for Local Blood Flow Control.
local tissue could regulate blood flow through the area.
Although the vasodilator theory is widely accepted,
Because smooth muscle requires oxygen to remain
198
Unit IV The Circulation
contracted, one might assume that the strength of con-
emphasizes the close connection between local blood
traction of the sphincters would increase with an
flow regulation and delivery of oxygen and other nutri-
increase in oxygen concentration. Consequently, when
ents to the tissues.
the oxygen concentration in the tissue rises above
a certain level, the precapillary and metarteriole
Active Hyperemia. When any tissue becomes highly
sphincters presumably would close until the tissue
active, such as an exercising muscle, a gastrointestinal
cells consume the excess oxygen. But when the excess
gland during a hypersecretory period, or even the
oxygen is gone and the oxygen concentration falls low
brain during rapid mental activity, the rate of blood
enough, the sphincters would open once more to begin
flow through the tissue increases. Here again, by
the cycle again.
simply applying the basic principles of local blood flow
Thus, on the basis of available data, either a
control, one can easily understand this active hyper-
vasodilator substance theory or an oxygen lack theory
emia. The increase in local metabolism causes the cells
could explain acute local blood flow regulation in
to devour tissue fluid nutrients extremely rapidly and
response to the metabolic needs of the tissues.
also to release large quantities of vasodilator sub-
Probably the truth lies in a combination of the two
stances. The result is to dilate the local blood vessels
mechanisms.
and, therefore, to increase local blood flow. In this way,
the active tissue receives the additional nutrients
Possible Role of Other Nutrients Besides Oxygen in Control of
required to sustain its new level of function. As
Local Blood Flow. Under special conditions, it has been
pointed out earlier, active hyperemia in skeletal
shown that lack of glucose in the perfusing blood can
muscle can increase local muscle blood flow as much
cause local tissue vasodilation. Also, it is possible that
as 20-fold during intense exercise.
this same effect occurs when other nutrients, such as
amino acids or fatty acids, are deficient, although this
“Autoregulation” of Blood Flow When the
has not been studied adequately. In addition, vasodi-
Arterial Pressure Changes from Normal—
lation occurs in the vitamin deficiency disease beriberi,
“Metabolic” and “Myogenic” Mechanisms
in which the patient has deficiencies of the vitamin B
In any tissue of the body, an acute increase in arterial
substances thiamine, niacin, and riboflavin. In this
pressure causes immediate rise in blood flow. But,
disease, the peripheral vascular blood flow everywhere
within less than a minute, the blood flow in most
in the body often increases twofold to threefold.
tissues returns almost to the normal level, even though
Because these vitamins all are needed for oxygen-
the arterial pressure is kept elevated. This return of
induced phosphorylation that is required to produce
flow toward normal is called “autoregulation of blood
ATP in the tissue cells, one can well understand how
flow.” After autoregulation has occurred, the local
deficiency of these vitamins might lead to diminished
blood flow in most body tissues will be related to arte-
smooth muscle contractile ability and therefore also
rial pressure approximately in accord with the solid
local vasodilation.
“acute” curve in Figure 17-4. Note that between an
Special Examples of Acute “Metabolic”
Control of Local Blood Flow
The mechanisms that we have described thus far for
local blood flow control are called “metabolic mecha-
2.5
nisms” because all of them function in response to the
Acute
metabolic needs of the tissues. Two additional special
examples of metabolic control of local blood flow are
2.0
reactive hyperemia and active hyperemia.
1.5
Reactive Hyperemia. When the blood supply to a tissue
is blocked for a few seconds to as long an hour or more
1.0
Long-term
and then is unblocked, blood flow through the tissue
usually increases immediately to four to seven times
0.5
normal; this increased flow will continue for a few
seconds if the block has lasted only a few seconds but
sometimes continues for as long as many hours if the
0
0
50
100
150
200
250
blood flow has been stopped for an hour or more. This
Arterial pressure (mm Hg)
phenomenon is called reactive hyperemia.
Reactive hyperemia is another manifestation of the
local “metabolic” blood flow regulation mechanism;
that is, lack of flow sets into motion all of those factors
Figure 17-4
that cause vasodilation. After short periods of vascu-
lar occlusion, the extra blood flow during the reactive
Effect of different levels of arterial pressure on blood flow through
a muscle. The solid red curve shows the effect if the arterial pres-
hyperemia phase lasts long enough to repay almost
sure is raised over a period of a few minutes. The dashed green
exactly the tissue oxygen deficit that has accrued
curve shows the effect if the arterial pressure is raised extremely
during the period of occlusion. This mechanism
slowly over a period of many weeks.
Chapter 17
Local and Humoral Control of Blood Flow by the Tissues
199
arterial pressure of about 70 mm Hg and 175 mm Hg,
operate in a few special areas. They all are discussed
the blood flow increases only 30 per cent even though
throughout this text in relation to specific organs, but
the arterial pressure increases 150 per cent.
two notable ones are as follows:
For almost a century, two views have been proposed
1. In the kidneys, blood flow control is vested mainly
to explain this acute autoregulation mechanism. They
in a mechanism called tubuloglomerular feedback,
in which the composition of the fluid in the early
have been called (1) the metabolic theory and (2) the
distal tubule is detected by an epithelial structure
myogenic theory.
of the distal tubule itself called the macula densa.
The metabolic theory can be understood easily by
This is located where the distal tubule lies adjacent
applying the basic principles of local blood flow regu-
to the afferent and efferent arterioles at the
lation discussed in previous sections. Thus, when the
nephron juxtaglomerular apparatus. When too
arterial pressure becomes too great, the excess flow
much fluid filters from the blood through the
provides too much oxygen and too many other nutri-
glomerulus into the tubular system, appropriate
ents to the tissues. These nutrients (especially oxygen)
feedback signals from the macula densa cause
then cause the blood vessels to constrict and the flow
constriction of the afferent arterioles, in this way
to return nearly to normal despite the increased
reducing both renal blood flow and glomerular
filtration rate back to or near to normal. The
pressure.
details of this mechanism are discussed in
The myogenic theory, however, suggests that still
Chapter 26.
another mechanism not related to tissue metabolism
2. In the brain, in addition to control of blood flow by
explains the phenomenon of autoregulation. This
tissue oxygen concentration, the concentrations of
theory is based on the observation that sudden stretch
carbon dioxide and hydrogen ions play very
of small blood vessels causes the smooth muscle of the
prominent roles. An increase of either or both of
vessel wall to contract for a few seconds. Therefore, it
these dilates the cerebral vessels and allows rapid
has been proposed that when high arterial pressure
washout of the excess carbon dioxide or hydrogen
stretches the vessel, this in turn causes reactive vascu-
ions from the brain tissues. This is important
lar constriction that reduces blood flow nearly back to
because the level of excitability of the brain itself is
highly dependent on exact control of both carbon
normal. Conversely, at low pressures, the degree of
dioxide concentration and hydrogen ion
stretch of the vessel is less, so that the smooth muscle
concentration. This special mechanism for cerebral
relaxes and allows increased flow.
blood flow control is presented in Chapter 61.
The myogenic response is inherent to vascular
smooth muscle and can occur in the absence of neural
or hormonal influences. It is most pronounced in arte-
Mechanism for Dilating Upstream Arteries
rioles but can also be observed in arteries, venules,
When Microvascular Blood Flow Increases—
veins, and even lymphatic vessels. Myogenic contrac-
The Endothelium-Derived Relaxing Factor
tion is initiated by stretch-induced vascular depolar-
(Nitric Oxide)
ization, which then rapidly increases calcium ion entry
The local mechanisms for controlling tissue blood flow
from the extracellular fluid into the cells, causing them
can dilate only the very small arteries and arterioles in
to contract. Changes in vascular pressure may also
each tissue because tissue cell vasodilator substances
open or close other ion channels that influence vascu-
or tissue cell oxygen deficiency can reach only these
lar contraction. The precise mechanisms by which
vessels, not the intermediate and larger arteries back
changes in pressure cause opening or closing of vas-
upstream. Yet, when blood flow through a micro-
cular ion channels are still uncertain, but likely involve
vascular portion of the circulation increases, this sec-
mechanical effects of pressure on extracellular pro-
ondarily entrains another mechanism that does dilate
teins that are tethered to cytoskeleton elements of the
the larger arteries as well. This mechanism is the
vascular wall or to the ion channels themselves.
following:
The myogenic mechanism may be important in pre-
The endothelial cells lining the arterioles and small
venting excessive stretch of blood vessel when blood
arteries synthesize several substances that, when
pressure is increased. However, the importance of
released, can affect the degree of relaxation or con-
the myogenic mechanism in blood flow regulation
traction of the arterial wall. The most important of
is unclear because this pressure sensing mechanism
these is a vasodilator substance called endothelium-
cannot directly detect changes in blood flow in the
derived relaxing factor (EDRF), which is composed
tissue. Indeed metabolic factors appear to override
principally of nitric oxide, which has a half-life in the
the myogenic mechanism in circumstances where the
blood of only 6 seconds. Rapid flow of blood through
metabolic demands of the tissues are significantly
the arteries and arterioles causes shear stress on the
increased, such as during vigorous muscle exercise,
endothelial cells because of viscous drag of the blood
which can cause dramatic increases in skeletal muscle
against the vascular walls. This stress contorts the
blood flow.
endothelial cells in the direction of flow and causes sig-
nificant increase in the release of nitric oxide. The
Special Mechanisms for Acute Blood Flow
nitric oxide then relaxes the blood vessels. This is for-
Control in Specific Tissues
tunate because it increases the diameters of the
Although the general mechanisms for local blood flow
upstream arterial blood vessels whenever microvascu-
control discussed thus far are present in almost all
lar blood flow increases downstream. Without such a
tissues of the body, distinctly different mechanisms
response, the effectiveness of local blood flow control
200
Unit IV The Circulation
would be significantly decreased because a significant
well-established tissues. Therefore, the time required
part of the resistance to blood flow is in the upstream
for long-term regulation to take place may be only
small arteries.
a few days in the neonate or as long as months in
the elderly person. Furthermore, the final degree of
response is much better in younger tissues than in
Long-Term Blood Flow Regulation
older, so that in the neonate, the vascularity will adjust
to match almost exactly the needs of the tissue for
Thus far, most of the mechanisms for local blood flow
blood flow, whereas in older tissues, vascularity fre-
regulation that we have discussed act within a few
quently lags far behind the needs of the tissues.
seconds to a few minutes after the local tissue condi-
Role of Oxygen in Long-Term Regulation. Oxygen is impor-
tions have changed. Yet, even after full activation
tant not only for acute control of local blood flow but
of these acute mechanisms, the blood flow usually
also for long-term control. One example of this is
is adjusted only about three quarters of the way to
increased vascularity in tissues of animals that live at
the exact additional requirements of the tissues.
high altitudes, where the atmospheric oxygen is low. A
For instance, when the arterial pressure suddenly is
second example is that fetal chicks hatched in low
increased from 100 to 150 mm Hg, the blood flow
oxygen have up to twice as much tissue blood vessel
increases almost instantaneously about 100 per cent.
conductivity as is normally true. This same effect is also
Then, within
30 seconds to
2 minutes, the flow
dramatically demonstrated in premature human
decreases back to about 15 per cent above the origi-
babies put into oxygen tents for therapeutic purposes.
nal control value. This illustrates the rapidity of the
The excess oxygen causes almost immediate cessation
acute mechanisms for local blood flow regulation, but
of new vascular growth in the retina of the premature
at the same time, it demonstrates that the regulation is
baby’s eyes and even causes degeneration of some of
still incomplete because there remains an excess 15 per
the small vessels that already have formed. Then when
cent increase in blood flow.
the infant is taken out of the oxygen tent, there is
However, over a period of hours, days, and weeks, a
explosive overgrowth of new vessels to make up for
long-term type of local blood flow regulation develops
the sudden decrease in available oxygen; indeed, there
in addition to the acute regulation. This long-term
is often so much overgrowth that the retinal vessels
regulation gives far more complete regulation. For
grow out from the retina into the eye’s vitreous humor;
instance, in the aforementioned example, if the arte-
and this eventually causes blindness. (This condition is
rial pressure remains at 150 mm Hg indefinitely, within
called retrolental fibroplasia.)
a few weeks the blood flow through the tissues gradu-
ally reapproaches almost exactly the normal flow level.
Importance of Vascular Endothelial Growth
Figure 17-4 shows by the dashed green curve the
Factor in Formation of New Blood Vessels
extreme effectiveness of this long-term local blood
A dozen or more factors that increase growth of new
flow regulation. Note that once the long-term regula-
blood vessels have been found, almost all of which are
tion has had time to occur, long-term changes in arte-
small peptides. Three of those that have been best
rial pressure between 50 and 250 mm Hg have little
characterized are vascular endothelial growth factor
effect on the rate of local blood flow.
(VEGF), fibroblast growth factor, and angiogenin, each
Long-term regulation of blood flow is especially
of which has been isolated from tissues that have inad-
important when the long-term metabolic demands
equate blood supply. Presumably, it is deficiency of
of a tissue change. Thus, if a tissue becomes chroni-
tissue oxygen or other nutrients, or both, that leads to
cally overactive and therefore requires chronically
formation of the vascular growth factors (also called
increased quantities of oxygen and other nutrients, the
“angiogenic factors”).
arterioles and capillary vessels usually increase both in
Essentially all the angiogenic factors promote new
number and size within a few weeks to match the
vessel growth in the same way. They cause new vessels
needs of the tissue—unless the circulatory system has
to sprout from other small vessels. The first step is dis-
become pathological or too old to respond.
solution of the basement membrane of the endothelial
cells at the point of sprouting. This is followed by rapid
Mechanism of Long-Term Regulation—
reproduction of new endothelial cells that stream
Change in “Tissue Vascularity”
outward through the vessel wall in extended cords
The mechanism of long-term local blood flow regula-
directed toward the source of the angiogenic factor.
tion is principally to change the amount of vascularity
The cells in each cord continue to divide and rapidly
of the tissues. For instance, if the metabolism in a
fold over into a tube. Next, the tube connects with
given tissue is increased for a prolonged period,
another tube budding from another donor vessel
vascularity increases; if the metabolism is decreased,
(another arteriole or venule) and forms a capillary
vascularity decreases.
loop through which blood begins to flow. If the flow is
Thus, there is actual physical reconstruction of
great enough, smooth muscle cells eventually invade
the tissue vasculature to meet the needs of the tissues.
the wall, so that some of the new vessels eventually
This reconstruction occurs rapidly (within days) in
grow to be new arterioles or venules or perhaps even
extremely young animals. It also occurs rapidly in new
larger vessels. Thus, angiogenesis explains the manner
growth tissue, such as in scar tissue and cancerous
in which metabolic factors in local tissues can cause
tissue; however, it occurs much more slowly in old,
growth of new vessels.
Chapter 17
Local and Humoral Control of Blood Flow by the Tissues
201
Certain other substances, such as some steroid hor-
metabolic dilation, followed chronically by manifold
mones, have exactly the opposite effect on small blood
growth and enlargement of new vessels over a period
vessels, occasionally even causing dissolution of vas-
of weeks and months.
cular cells and disappearance of vessels. Therefore,
The most important example of the development of
blood vessels can also be made to disappear when not
collateral blood vessels occurs after thrombosis of one
needed.
of the coronary arteries. Almost all people by the age
of 60 years have had at least one of the smaller branch
coronary vessels close. Yet most people do not know
Vascularity Is Determined by Maximum Blood Flow Need, Not
that this has happened because collaterals have devel-
by Average Need. An especially valuable characteristic
oped rapidly enough to prevent myocardial damage. It
of long-term vascular control is that vascularity is
is in those other instances in which coronary insuffi-
determined mainly by the maximum level of blood
ciency occurs too rapidly or too severely for collater-
flow need rather than by average need. For instance,
als to develop that serious heart attacks occur.
during heavy exercise the need for whole body blood
flow often increases to six to eight times the resting
blood flow. This great excess of flow may not be
Humoral Control of the
required for more than a few minutes each day. Nev-
ertheless, even this short need can cause enough
Circulation
VEGF to be formed by the muscles to increase their
vascularity as required. Were it not for this capability,
Humoral control of the circulation means control by
every time that a person attempted heavy exercise, the
substances secreted or absorbed into the body fluids—
muscles would fail to receive the required nutrients,
such as hormones and ions. Some of these substances
especially the required oxygen, so that the muscles
are formed by special glands and transported in
simply would fail to contract.
the blood throughout the entire body. Others are
However, after extra vascularity does develop, the
formed in local tissue areas and cause only local cir-
extra blood vessels normally remain mainly vasocon-
culatory effects. Among the most important of the
stricted, opening to allow extra flow only when ap-
humoral factors that affect circulatory function are the
propriate local stimuli such as oxygen lack, nerve
following.
vasodilatory stimuli, or other stimuli call forth the
required extra flow.
Vasoconstrictor Agents
Norepinephrine and Epinephrine. Norepinephrine is an
Development of Collateral
especially powerful vasoconstrictor hormone; epi-
Circulation—A Phenomenon of Long-
nephrine is less so and in some tissues even causes mild
Term Local Blood Flow Regulation
vasodilation.
(A special example of vasodilation
caused by epinephrine occurs to dilate the coronary
When an artery or a vein is blocked in virtually any
arteries during increased heart activity.)
tissue of the body, a new vascular channel usually
When the sympathetic nervous system is stimulated
develops around the blockage and allows at least
in most or all parts of the body during stress or exer-
partial resupply of blood to the affected tissue. The
cise, the sympathetic nerve endings in the individual
first stage in this process is dilation of small vascular
tissues release norepinephrine, which excites the heart
loops that already connect the vessel above the block-
and contracts the veins and arterioles. In addition,
age to the vessel below. This dilation occurs within the
the sympathetic nerves to the adrenal medullae cause
first minute or two, indicating that the dilation is
these glands to secrete both norepinephrine and epi-
simply a neurogenic or metabolic relaxation of the
nephrine into the blood. These hormones then circu-
muscle fibers of the small vessels involved. After this
late to all areas of the body and cause almost the same
initial opening of collateral vessels, the blood flow
effects on the circulation as direct sympathetic stimu-
often is still less than one quarter that needed to
lation, thus providing a dual system of control: (1)
supply all the tissue needs. However, further opening
direct nerve stimulation and (2) indirect effects of
occurs within the ensuing hours, so that within 1 day
norepinephrine and/or epinephrine in the circulating
as much as half the tissue needs may be met, and
blood.
within a few days often all the tissue needs.
The collateral vessels continue to grow for many
Angiotensin II. Angiotensin II is another powerful vaso-
months thereafter, almost always forming multiple
constrictor substance. As little as one millionth of a
small collateral channels rather than one single large
gram can increase the arterial pressure of a human
vessel. Under resting conditions, the blood flow usually
being 50 mm Hg or more.
returns very near to normal, but the new channels
The effect of angiotensin II is to constrict powerfully
seldom become large enough to supply the blood flow
the small arterioles. If this occurs in an isolated
needed during strenuous tissue activity. Thus, the
tissue area, the blood flow to that area can be
development of collateral vessels follows the usual
severely depressed. However, the real importance of
principles of both acute and long-term local blood flow
angiotensin II is that it normally acts on many of the
control, the acute control being rapid neurogenic and
arterioles of the body at the same time to increase the
202
Unit IV The Circulation
total peripheral resistance, thereby increasing the arte-
kallidin that then is converted by tissue enzymes into
rial pressure. Thus, this hormone plays an integral role
bradykinin. Once formed, bradykinin persists for only
in the regulation of arterial pressure, as is discussed in
a few minutes because it is inactivated by the enzyme
detail in Chapter 19.
carboxypeptidase or by converting enzyme, the same
enzyme that also plays an essential role in activating
Vasopressin. Vasopressin, also called antidiuretic
angiotensin, as discussed in Chapter 19. The activated
hormone, is even more powerful than angiotensin II as
kallikrein enzyme is destroyed by a kallikrein inhibitor
a vasoconstrictor, thus making it one of the body’s
also present in the body fluids.
most potent vascular constrictor substances. It is
Bradykinin causes both powerful arteriolar
formed in nerve cells in the hypothalamus of the brain
dilation and increased capillary permeability. For
(see Chapter 75) but is then transported downward by
instance, injection of 1 microgram of bradykinin into
nerve axons to the posterior pituitary gland, where it
the brachial artery of a person increases blood flow
is finally secreted into the blood.
through the arm as much as sixfold, and even smaller
It is clear that vasopressin could have enormous
amounts injected locally into tissues can cause marked
effects on circulatory function. Yet, normally, only
local edema resulting from increase in capillary pore
minute amounts of vasopressin are secreted, so that
size.
most physiologists have thought that vasopressin plays
There is reason to believe that kinins play special
little role in vascular control. However, experiments
roles in regulating blood flow and capillary leakage of
have shown that the concentration of circulating blood
fluids in inflamed tissues. It also is believed that
vasopressin after severe hemorrhage can rise high
bradykinin plays a normal role to help regulate blood
enough to increase the arterial pressure as much as
flow in the skin as well as in the salivary and gastroin-
60 mm Hg. In many instances, this can, by itself, bring
testinal glands.
the arterial pressure almost back up to normal.
Vasopressin has a major function to increase greatly
Histamine. Histamine is released in essentially every
water reabsorption from the renal tubules back into
tissue of the body if the tissue becomes damaged or
the blood (discussed in Chapter 28), and therefore
inflamed or is the subject of an allergic reaction. Most
to help control body fluid volume. That is why this
of the histamine is derived from mast cells in the
hormone is also called antidiuretic hormone.
damaged tissues and from basophils in the blood.
Histamine has a powerful vasodilator effect on
Endothelin—A Powerful Vasoconstrictor in Damaged Blood
the arterioles and, like bradykinin, has the ability to
Vessels. Still another vasoconstrictor substance that
increase greatly capillary porosity, allowing leakage
ranks along with angiotensin and vasopressin in its
of both fluid and plasma protein into the tissues. In
vasoconstrictor capability is a large 21 amino acid
many pathological conditions, the intense arteriolar
peptide called endothelin, which requires only
dilation and increased capillary porosity produced
nanogram quantities to cause powerful vasoconstric-
by histamine cause tremendous quantities of fluid
tion. This substance is present in the endothelial cells
to leak out of the circulation into the tissues, inducing
of all or most blood vessels. The usual stimulus for
edema. The local vasodilatory and edema-producing
release is damage to the endothelium, such as that
effects of histamine are especially prominent
caused by crushing the tissues or injecting a trauma-
during allergic reactions and are discussed in
tizing chemical into the blood vessel. After severe
Chapter 34.
blood vessel damage, release of local endothelin and
subsequent vasoconstriction helps to prevent exten-
sive bleeding from arteries as large as 5 millimeters in
Vascular Control by Ions and Other
diameter that might have been torn open by crushing
Chemical Factors
injury.
Many different ions and other chemical factors can
either dilate or constrict local blood vessels. Most of
Vasodilator Agents
them have little function in overall regulation of the
circulation, but some specific effects are:
Bradykinin. Several substances called kinins cause pow-
1. An increase in calcium ion concentration causes
erful vasodilation when formed in the blood and tissue
vasoconstriction. This results from the general
fluids of some organs.
effect of calcium to stimulate smooth muscle
The kinins are small polypeptides that are split away
contraction, as discussed in Chapter 8.
by proteolytic enzymes from alpha2-globulins in the
2. An increase in potassium ion concentration causes
plasma or tissue fluids. A proteolytic enzyme of par-
vasodilation. This results from the ability of
ticular importance for this purpose is kallikrein, which
potassium ions to inhibit smooth muscle
is present in the blood and tissue fluids in an inactive
contraction.
form. This inactive kallikrein is activated by macera-
3. An increase in magnesium ion concentration
tion of the blood, by tissue inflammation, or by other
causes powerful vasodilation because magnesium
similar chemical or physical effects on the blood or
ions inhibit smooth muscle contraction.
tissues. As kallikrein becomes activated, it acts imme-
4. An increase in hydrogen ion concentration
diately on alpha2-globulin to release a kinin called
(decrease in pH) causes dilation of the arterioles.
Chapter 17
Local and Humoral Control of Blood Flow by the Tissues
203
Conversely, slight decrease in hydrogen ion
Ferrara N, Gerber HP, LeCouter J: The biology of VEGF and
concentration causes arteriolar constriction.
its receptors. Nat Med 9:669, 2003.
5. Anions that have significant effects on blood
Granger HJ, Guyton AC: Autoregulation of the total sys-
temic circulation following destruction of the central
vessels are acetate and citrate, both of which cause
nervous system in the dog. Circ Res 25:379, 1969.
mild degrees of vasodilation.
Guyton AC, Coleman TG, Granger HJ: Circulation: overall
6. An increase in carbon dioxide concentration
regulation. Annu Rev Physiol 34:13, 1972.
causes moderate vasodilation in most tissues, but
Hall JE, Brands MW, Henegar JR: Angiotensin II and long-
marked vasodilation in the brain. Also, carbon
term arterial pressure regulation: the overriding domi-
dioxide in the blood, acting on the brain
nance of the kidney. J Am Soc Nephrol 10(Suppl 12):S258,
vasomotor center, has an extremely powerful
1999.
indirect effect, transmitted through the
Harder DR, Zhang C, Gebremedhin D: Astrocytes function
sympathetic nervous vasoconstrictor system, to
in matching blood flow to metabolic activity. News Physiol
cause widespread vasoconstriction throughout the
Sci 17:27, 2002.
Hester RL, Hammer LW: Venular-arteriolar communication
body.
in the regulation of blood flow. Am J Physiol Regul Integr
Comp Physiol 282:R1280, 2002.
Iglarz M, Schiffrin EL: Role of endothelin-1 in hypertension.
References
Curr Hypertens Rep 5:144, 2003.
Kerbel R, Folkman J: Clinical translation of angiogenesis
Adair TH, Gay WJ, Montani JP: Growth regulation of the
inhibitors. Nat Rev Cancer 2:727, 2002.
vascular system: evidence for a metabolic hypothesis. Am
Losordo DW, Dimmeler S: Therapeutic angiogenesis and
J Physiol 259:R393, 1990.
vasculogenesis for ischemic disease: Part I: angiogenic
Campbell WB, Gauthier KM: What is new in endothelium-
cytokines. Circulation 109:2487, 2004.
derived hyperpolarizing factors? Curr Opin Nephrol
Renkin EM: Control of microcirculation and blood-tissue
Hypertens 11:177, 2002.
exchange. In: Renkin EM, Michel CC (eds): Handbook of
Chang L, Kaipainen A, Folkman J: Lymphangiogenesis new
Physiology, Sec. 2, Vol. IV. Bethesda: American Physiolog-
mechanisms. Ann N Y Acad Sci 979:111, 2002.
ical Society, 1984, p 627.
Cowley AW Jr, Mori T, Mattson D, Zou AP: Role of renal
Rich S, McLaughlin VV: Endothelin receptor blockers in car-
NO production in the regulation of medullary blood flow.
diovascular disease. Circulation 108:2184, 2003.
Am J Physiol Regul Integr Comp Physiol 284:R1355, 2003.
Roman RJ: P-450 metabolites of arachidonic acid in the
Davis MJ, Hill MA: Signaling mechanisms underlying the
control of cardiovascular function. Physiol Rev 82:131,
vascular myogenic response. Physiol Rev 79:387, 1999.
2002.
Erdös EG, Marcic BM: Kinins, receptors, kininases and
Schnermann J, Levine DZ: Paracrine factors in tubu-
inhibitors—where did they lead us? Biol Chem 382:43,
loglomerular feedback: adenosine, ATP, and nitric oxide.
2001.
Annu Rev Physiol 65:501, 2003.
C H A P T E R
1
8
Nervous Regulation of the
Circulation, and Rapid Control
of Arterial Pressure
Nervous Regulation of the
Circulation
As discussed in Chapter 17, adjustment of blood
flow tissue by tissue is mainly the function of local
tissue blood flow control mechanisms. We shall see
in this chapter that nervous control of the circula-
tion has more global functions, such as redistribut-
ing blood flow to different areas of the body, increasing or decreasing pumping
activity by the heart, and, especially, providing very rapid control of systemic
arterial pressure.
The nervous system controls the circulation almost entirely through the auto-
nomic nervous system. The total function of this system is presented in Chapter
60, and this subject was also introduced in Chapter 17. For our present discus-
sion, we need to present still other specific anatomical and functional charac-
teristics, as follows.
Autonomic Nervous System
By far the most important part of the autonomic nervous system for regulating
the circulation is the sympathetic nervous system. The parasympathetic nervous
system also contributes specifically to regulation of heart function, as we shall
see later in the chapter.
Sympathetic Nervous System. Figure
18-1 shows the anatomy of sympathetic
nervous control of the circulation. Sympathetic vasomotor nerve fibers leave
the spinal cord through all the thoracic spinal nerves and through the first one
or two lumbar spinal nerves. They then pass immediately into a sympathetic
chain, one of which lies on each side of the vertebral column. Next, they pass
by two routes to the circulation: (1) through specific sympathetic nerves that
innervate mainly the vasculature of the internal viscera and the heart, as shown
on the right side of Figure 18-1, and (2) almost immediately into peripheral por-
tions of the spinal nerves distributed to the vasculature of the peripheral areas.
The precise pathways of these fibers in the spinal cord and in the sympathetic
chains are discussed more fully in Chapter 60.
Sympathetic Innervation of the Blood Vessels. Figure 18-2 shows distribution
of sympathetic nerve fibers to the blood vessels, demonstrating that in most
tissues all the vessels except the capillaries, precapillary sphincters, and metar-
terioles are innervated.
The innervation of the small arteries and arterioles allows sympathetic stim-
ulation to increase resistance to blood flow and thereby to decrease rate of blood
flow through the tissues.
The innervation of the large vessels, particularly of the veins, makes it possi-
ble for sympathetic stimulation to decrease the volume of these vessels. This can
push blood into the heart and thereby play a major role in regulation of heart
pumping, as we shall see later in this and subsequent chapters.
204
Chapter 18
Nervous Regulation of the Circulation, and Rapid Control of Arterial Pressure
205
Vasomotor center
Blood
vessels
Sympathetic chain
Vagus
Heart
Vasoconstrictor
Cardioinhibitor
Vasodilator
Blood
vessels
Figure 18-1
Anatomy of sympathetic nervous
control of the circulation. Also
shown by the red dashed line is a
vagus nerve that carries parasym-
pathetic signals to the heart.
Sympathetic Nerve Fibers to the Heart. In addition to
Arteries
sympathetic nerve fibers supplying the blood vessels,
sympathetic fibers also go directly to the heart, as
Arterioles
shown in Figure 18-1 and also discussed in Chapter 9.
It should be recalled that sympathetic stimulation
markedly increases the activity of the heart, both
increasing the heart rate and enhancing its strength
Sympathetic
and volume of pumping.
vasoconstriction
Capillaries
Parasympathetic Control of Heart Function, Especially Heart
Rate. Although the parasympathetic nervous system is
exceedingly important for many other autonomic
functions of the body, such as control of multiple gas-
trointestinal actions, it plays only a minor role in
Veins
Venules
regulation of the circulation. Its most important circu-
latory effect is to control heart rate by way of parasym-
Figure 18-2
pathetic nerve fibers to the heart in the vagus
Sympathetic innervation of the systemic circulation.
nerves, shown in Figure 18-1 by the dashed red line
from the brain medulla directly to the heart.
The effects of parasympathetic stimulation on
heart function were discussed in detail in Chapter 9.
206
Unit IV The Circulation
Principally, parasympathetic stimulation causes a
possible to identify certain important areas in this
marked decrease in heart rate and a slight decrease in
center, as follows:
heart muscle contractility.
1. A vasoconstrictor area located bilaterally in the
anterolateral portions of the upper medulla. The
neurons originating in this area distribute their
Sympathetic Vasoconstrictor System and
fibers to all levels of the spinal cord, where they
Its Control by the Central Nervous System
excite preganglionic vasoconstrictor neurons of the
The sympathetic nerves carry tremendous numbers of
sympathetic nervous system.
vasoconstrictor nerve fibers and only a few vasodilator
2. A vasodilator area located bilaterally in the
fibers. The vasoconstrictor fibers are distributed to
anterolateral portions of the lower half of the
essentially all segments of the circulation, but more to
medulla. The fibers from these neurons project
some tissues than others. This sympathetic vasocon-
upward to the vasoconstrictor area just described;
strictor effect is especially powerful in the kidneys,
they inhibit the vasoconstrictor activity of this area,
intestines, spleen, and skin but much less potent in
thus causing vasodilation.
skeletal muscle and the brain.
3. A sensory area located bilaterally in the tractus
solitarius in the posterolateral portions of the
medulla and lower pons. The neurons of this area
Vasomotor Center in the Brain and Its Control of the Vasocon-
receive sensory nerve signals from the circulatory
strictor System. Located bilaterally mainly in the retic-
system mainly through the vagus and
ular substance of the medulla and of the lower third
glossopharyngeal nerves, and output signals from
of the pons, shown in Figures 18-1 and 18-3, is an area
this sensory area then help to control activities of
called the vasomotor center. This center transmits
both the vasoconstrictor and vasodilator areas of
parasympathetic impulses through the vagus nerves to
the vasomotor center, thus providing “reflex”
the heart and transmits sympathetic impulses through
control of many circulatory functions. An example
the spinal cord and peripheral sympathetic nerves
is the baroreceptor reflex for controlling arterial
to virtually all arteries, arterioles, and veins of the
pressure, which we describe later in this chapter.
body.
Although the total organization of the vasomotor
Continuous Partial Constriction of the Blood Vessels Is Nor-
center is still unclear, experiments have made it
mally Caused by Sympathetic Vasoconstrictor Tone. Under
normal conditions, the vasoconstrictor area of the
vasomotor center transmits signals continuously to
the sympathetic vasoconstrictor nerve fibers over the
Reticular
entire body, causing continuous slow firing of these
Motor
substance
Cingulate
fibers at a rate of about one half to two impulses per
second. This continual firing is called sympathetic vaso-
Orbital
Mesencephalon
constrictor tone. These impulses normally maintain a
partial state of contraction in the blood vessels, called
vasomotor tone.
Figure 18-4 demonstrates the significance of vaso-
constrictor tone. In the experiment of this figure,
total spinal anesthesia was administered to an animal.
This blocked all transmission of sympathetic nerve
impulses from the spinal cord to the periphery. As a
result, the arterial pressure fell from 100 to 50 mm Hg,
demonstrating the effect of losing vasoconstrictor tone
throughout the body. A few minutes later, a small
Temporal
amount of the hormone norepinephrine was injected
into the blood
(norepinephrine is the principal
vasoconstrictor hormonal substance secreted at the
Pons
endings of the sympathetic vasoconstrictor nerve
fibers throughout the body). As this injected hormone
Medulla
VASOMOTOR
was transported in the blood to all blood vessels, the
CENTER
vessels once again became constricted, and the arterial
pressure rose to a level even greater than normal for
VASODILATOR
1 to 3 minutes, until the norepinephrine was destroyed.
VASOCONSTRICTOR
Control of Heart Activity by the Vasomotor Center. At the
same time that the vasomotor center is controlling the
amount of vascular constriction, it also controls heart
activity. The lateral portions of the vasomotor center
Figure 18-3
transmit excitatory impulses through the sympathetic
nerve fibers to the heart when there is need to increase
Areas of the brain that play important roles in the nervous regula-
tion of the circulation. The dashed lines represent inhibitory
heart rate and contractility. Conversely, when there is
pathways.
need to decrease heart pumping, the medial portion of
Chapter 18
Nervous Regulation of the Circulation, and Rapid Control of Arterial Pressure
207
150
125
Total spinal
anesthesia
100
75
50
Injection of norepinephrine
25
Figure 18-4
0
Effect of total spinal anesthesia on
0
5
10
15
20
25
the arterial pressure, showing
Seconds
marked decrease in pressure
resulting from loss of “vasomotor
tone.”
the vasomotor center sends signals to the adjacent
the cingulate gyrus, the amygdala, the septum, and the
dorsal motor nuclei of the vagus nerves, which then
hippocampus can all either excite or inhibit the vaso-
transmit parasympathetic impulses through the vagus
motor center, depending on the precise portions of
nerves to the heart to decrease heart rate and heart
these areas that are stimulated and on the intensity of
contractility. Therefore, the vasomotor center can
stimulus. Thus, widespread basal areas of the brain can
either increase or decrease heart activity. Heart rate
have profound effects on cardiovascular function.
and strength of heart contraction ordinarily increase
when vasoconstriction occurs and ordinarily decrease
Norepinephrine—The Sympathetic Vasoconstrictor Transmitter
when vasoconstriction is inhibited.
Substance. The substance secreted at the endings of the
vasoconstrictor nerves is almost entirely norepineph-
Control of the Vasomotor Center by Higher Nervous Centers.
rine. Norepinephrine acts directly on the alpha adren-
Large numbers of small neurons located throughout
ergic receptors of the vascular smooth muscle to cause
the reticular substance of the pons, mesencephalon,
vasoconstriction, as discussed in Chapter 60.
and diencephalon can either excite or inhibit the
vasomotor center. This reticular substance is shown
Adrenal Medullae and Their Relation to the Sympathetic Vaso-
in Figure 18-3 by the rose-colored area. In general, the
constrictor System. Sympathetic impulses are transmit-
neurons in the more lateral and superior portions
ted to the adrenal medullae at the same time that they
of the reticular substance cause excitation, whereas
are transmitted to the blood vessels. They cause the
the more medial and inferior portions cause inhibition.
medullae to secrete both epinephrine and norepineph-
The hypothalamus plays a special role in controlling
rine into the circulating blood. These two hormones are
the vasoconstrictor system because it can exert either
carried in the blood stream to all parts of the body,
powerful excitatory or inhibitory effects on the vaso-
where they act directly on all blood vessels, usually
motor center. The posterolateral portions of the hypo-
to cause vasoconstriction, but in an occasional tissue
thalamus cause mainly excitation, whereas the anterior
epinephrine causes vasodilation because it also has
portion can cause either mild excitation or inhibition,
a “beta” adrenergic receptor stimulatory effect, which
depending on the precise part of the anterior hypo-
dilates rather than constricts certain vessels, as dis-
thalamus stimulated.
cussed in Chapter 60.
Many parts of the cerebral cortex can also excite or
inhibit the vasomotor center. Stimulation of the motor
Sympathetic Vasodilator System and its Control by the Central
cortex, for instance, excites the vasomotor center
Nervous System. The sympathetic nerves to skeletal
because of impulses transmitted downward into the
muscles carry sympathetic vasodilator fibers as well as
hypothalamus and thence to the vasomotor center.
constrictor fibers. In lower animals such as the cat, these
Also, stimulation of the anterior temporal lobe, the
dilator fibers release acetylcholine, not norepinephrine,
orbital areas of the frontal cortex, the anterior part of
at their endings, although in primates, the vasodilator
208
Unit IV
The Circulation
effect is believed to be caused by epinephrine exciting
3. Finally, the heart itself is directly stimulated by the
specific beta adrenergic receptors in the muscle
autonomic nervous system, further enhancing
vasculature.
cardiac pumping. Much of this is caused by an
The pathway for central nervous system control of
increase in the heart rate, the rate sometimes
the vasodilator system is shown by the dashed lines in
increasing to as great as three times normal. In
Figure 18-3. The principal area of the brain controlling
addition, sympathetic nervous signals have a
this system is the anterior hypothalamus.
significant direct effect to increase contractile
force of the heart muscle, this, too, increasing the
capability of the heart to pump larger volumes of
Possible Unimportance of the Sympathetic Vasodilator System.
blood. During strong sympathetic stimulation, the
It is doubtful that the sympathetic vasodilator system
heart can pump about two times as much blood as
plays an important role in the control of the circulation
under normal conditions. This contributes still more
in the human being because complete block of the sym-
to the acute rise in arterial pressure.
pathetic nerves to the muscles hardly affects the ability
of these muscles to control their own blood flow in
response to their needs. Yet some experiments suggest
Rapidity of Nervous Control of Arterial Pressure. An espe-
that at the onset of exercise, the sympathetic vasodila-
cially important characteristic of nervous control of
tor system might cause initial vasodilation in skeletal
arterial pressure is its rapidity of response, beginning
muscles to allow anticipatory increase in blood flow even
within seconds and often increasing the pressure to
before the muscles require increased nutrients.
two times normal within 5 to 10 seconds. Conversely,
sudden inhibition of nervous cardiovascular stimula-
Emotional Fainting—Vasovagal Syncope. A particularly
tion can decrease the arterial pressure to as little as
interesting vasodilatory reaction occurs in people who
experience intense emotional disturbances that cause
one half normal within 10 to 40 seconds. Therefore,
fainting. In this case, the muscle vasodilator system
nervous control of arterial pressure is by far the most
becomes activated, and at the same time, the vagal car-
rapid of all our mechanisms for pressure control.
dioinhibitory center transmits strong signals to the heart
to slow the heart rate markedly. The arterial pressure
falls rapidly, which reduces blood flow to the brain and
Increase in Arterial Pressure
causes the person to lose consciousness. This overall
effect is called vasovagal syncope. Emotional fainting
During Muscle Exercise and Other
begins with disturbing thoughts in the cerebral cortex.
Types of Stress
The pathway probably then goes to the vasodilatory
center of the anterior hypothalamus next to the vagal
An important example of the ability of the nervous
centers of the medulla, to the heart through the vagus
system to increase the arterial pressure is the increase
nerves, and also through the spinal cord to the sympa-
in pressure that occurs during muscle exercise. During
thetic vasodilator nerves of the muscles.
heavy exercise, the muscles require greatly increased
blood flow. Part of this increase results from local
vasodilation of the muscle vasculature caused by
Role of the Nervous System
increased metabolism of the muscle cells, as explained
in Rapid Control of
in Chapter 17. Additional increase results from simul-
Arterial Pressure
taneous elevation of arterial pressure caused by sym-
pathetic stimulation of the overall circulation during
One of the most important functions of nervous
exercise. In most heavy exercise, the arterial pressure
control of the circulation is its capability to cause rapid
rises about 30 to 40 per cent, which increases blood
increases in arterial pressure. For this purpose, the
flow almost an additional twofold.
entire vasoconstrictor and cardioaccelerator functions
The increase in arterial pressure during exercise
of the sympathetic nervous system are stimulated
results mainly from the following effect: At the same
together. At the same time, there is reciprocal inhibi-
time that the motor areas of the brain become acti-
tion of parasympathetic vagal inhibitory signals to the
vated to cause exercise, most of the reticular activat-
heart. Thus, three major changes occur simultaneously,
ing system of the brain stem is also activated, which
each of which helps to increase arterial pressure. They
includes greatly increased stimulation of the vasocon-
are as follows:
strictor and cardioacceleratory areas of the vasomotor
1. Almost all arterioles of the systemic circulation
center. These increase the arterial pressure instanta-
are constricted. This greatly increases the total
neously to keep pace with the increase in muscle
peripheral resistance, thereby increasing the arterial
activity.
pressure.
In many other types of stress besides muscle exer-
2. The veins especially (but the other large vessels of
cise, a similar rise in pressure can also occur. For
the circulation as well) are strongly constricted.
instance, during extreme fright, the arterial pressure
This displaces blood out of the large peripheral
sometimes rises to as high as double normal within a
blood vessels toward the heart, thus increasing the
few seconds. This is called the alarm reaction, and it
volume of blood in the heart chambers. The stretch
provides an excess of arterial pressure that can imme-
of the heart then causes the heart to beat with far
greater force and therefore to pump increased
diately supply blood to any or all muscles of the body
quantities of blood. This, too, increases the arterial
that might need to respond instantly to cause flight
pressure.
from danger.
Chapter 18
Nervous Regulation of the Circulation, and Rapid Control of Arterial Pressure
209
Reflex Mechanisms for Maintaining
Normal Arterial Pressure
Aside from the exercise and stress functions of the
autonomic nervous system to increase arterial pres-
sure, there are multiple subconscious special nervous
Glossopharyngeal nerve
control mechanisms that operate all the time to main-
tain the arterial pressure at or near normal. Almost
all of these are negative feedback reflex mechanisms,
which we explain in the following sections.
Hering’s nerve
The Baroreceptor Arterial Pressure Control
Carotid body
System—Baroreceptor Reflexes
Carotid sinus
By far the best known of the nervous mechanisms for
arterial pressure control is the baroreceptor reflex.
Basically, this reflex is initiated by stretch receptors,
Vagus nerve
called either baroreceptors or pressoreceptors, located
at specific points in the walls of several large systemic
arteries. A rise in arterial pressure stretches the
baroreceptors and causes them to transmit signals into
the central nervous system. “Feedback” signals are
Aortic baroreceptors
then sent back through the autonomic nervous system
to the circulation to reduce arterial pressure down-
ward toward the normal level.
Physiologic Anatomy of the Baroreceptors and Their Innerva-
tion. Baroreceptors are spray-type nerve endings that
lie in the walls of the arteries; they are stimulated when
Figure 18-5
stretched. A few baroreceptors are located in the wall
of almost every large artery of the thoracic and neck
The baroreceptor system for controlling arterial pressure.
regions; but, as shown in Figure 18-5, baroreceptors
are extremely abundant in (1) the wall of each inter-
nal carotid artery slightly above the carotid bifurca-
tion, an area known as the carotid sinus, and (2) the
wall of the aortic arch.
Figure 18-5 shows that signals from the “carotid
baroreceptors” are transmitted through very small
Hering’s nerves to the glossopharyngeal nerves in
the high neck, and then to the tractus solitarius in the
medullary area of the brain stem. Signals from the
“aortic baroreceptors” in the arch of the aorta are
transmitted through the vagus nerves also to the same
tractus solitarius of the medulla.
DI
= maximum
DP
Response of the Baroreceptors to Pressure. Figure
18-6
shows the effect of different arterial pressure levels on
the rate of impulse transmission in a Hering’s carotid
sinus nerve. Note that the carotid sinus baroreceptors
are not stimulated at all by pressures between 0 and
50 to 60 mm Hg, but above these levels, they respond
0
80
160
244
progressively more rapidly and reach a maximum at
Arterial blood pressure (mm Hg)
about 180 mm Hg. The responses of the aortic barore-
ceptors are similar to those of the carotid receptors
except that they operate, in general, at pressure levels
about 30 mm Hg higher.
Figure 18-6
Note especially that in the normal operating range
of arterial pressure, around 100 mm Hg, even a slight
Activation of the baroreceptors at different levels of arterial
change in pressure causes a strong change in the
pressure. DI, change in carotid sinus nerve impulses per second;
baroreflex signal to readjust arterial pressure back
DP, change in arterial blood pressure in mm Hg.
toward normal. Thus, the baroreceptor feedback
mechanism functions most effectively in the pressure
range where it is most needed.
210
Unit IV The Circulation
The baroreceptors respond extremely rapidly to
carotids are occluded. Removal of the occlusion allows
changes in arterial pressure; in fact, the rate of impulse
the pressure in the carotid sinuses to rise, and the
firing increases in the fraction of a second during each
carotid sinus reflex now causes the aortic pressure to
systole and decreases again during diastole. Further-
fall immediately to slightly below normal as a momen-
more, the baroreceptors respond much more to a
tary overcompensation and then return to normal in
rapidly changing pressure than to a stationary pres-
another minute.
sure. That is, if the mean arterial pressure is 150 mm
Hg but at that moment is rising rapidly, the rate of
Function of the Baroreceptors During Changes in Body
impulse transmission may be as much as twice that
Posture. The ability of the baroreceptors to maintain
when the pressure is stationary at 150 mm Hg.
relatively constant arterial pressure in the upper body
is important when a person stands up after having
Circulatory Reflex Initiated by the Baroreceptors. After the
been lying down. Immediately on standing, the arterial
baroreceptor signals have entered the tractus solitar-
pressure in the head and upper part of the body tends
ius of the medulla, secondary signals inhibit the vaso-
to fall, and marked reduction of this pressure could
constrictor center of the medulla and excite the vagal
cause loss of consciousness. However, the falling pres-
parasympathetic center. The net effects are (1) vasodi-
sure at the baroreceptors elicits an immediate reflex,
lation of the veins and arterioles throughout the
resulting in strong sympathetic discharge throughout
peripheral circulatory system and (2) decreased heart
the body. This minimizes the decrease in pressure in
rate and strength of heart contraction. Therefore, exci-
the head and upper body.
tation of the baroreceptors by high pressure in the
arteries reflexly causes the arterial pressure to decrease
Pressure
“Buffer” Function of the Baroreceptor
because of both a decrease in peripheral resistance
Control System. Because the baroreceptor system
and a decrease in cardiac output. Conversely, low pres-
opposes either increases or decreases in arterial pres-
sure has opposite effects, reflexly causing the pressure
sure, it is called a pressure buffer system, and the nerves
to rise back toward normal.
from the baroreceptors are called buffer nerves.
Figure 18-7 shows a typical reflex change in arterial
Figure 18-8 shows the importance of this buffer
pressure caused by occluding the two common carotid
function of the baroreceptors. The upper record in this
arteries. This reduces the carotid sinus pressure; as a
figure shows an arterial pressure recording for 2 hours
result, the baroreceptors become inactive and lose
from a normal dog, and the lower record shows an
their inhibitory effect on the vasomotor center. The
arterial pressure recording from a dog whose barore-
vasomotor center then becomes much more active
ceptor nerves from both the carotid sinuses and the
than usual, causing the aortic arterial pressure to rise
aorta had been removed. Note the extreme variability
and remain elevated during the 10 minutes that the
of pressure in the denervated dog caused by simple
events of the day, such as lying down, standing, excite-
ment, eating, defecation, and noises.
Figure 18-9 shows the frequency distributions of the
mean arterial pressures recorded for a 24-hour day in
both the normal dog and the denervated dog. Note
that when the baroreceptors were functioning nor-
mally the mean arterial pressure remained throughout
the day within a narrow range between
85 and
115 mm Hg—indeed, during most of the day at almost
150
exactly 100 mm Hg. Conversely, after denervation of
the baroreceptors, the frequency distribution curve
became the broad, low curve of the figure, showing
100
that the pressure range increased 2.5-fold, frequently
falling to as low as 50 mm Hg or rising to over 160 mm
Both common
Carotids released
Hg. Thus, one can see the extreme variability of pres-
carotids clamped
sure in the absence of the arterial baroreceptor
50
system.
In summary, a primary purpose of the arterial
baroreceptor system is to reduce the minute by minute
0
variation in arterial pressure to about one third that
0
2
4
6
8
10
12
14
which would occur were the baroreceptor system not
Minutes
present.
Are the Baroreceptors Important in Long-Term Regulation of
Arterial Pressure? Although the arterial baroreceptors
Figure 18-7
provide powerful moment-to-moment control of
arterial pressure, their importance in long-term
Typical carotid sinus reflex effect on aortic arterial pressure
caused by clamping both common carotids (after the two vagus
blood pressure regulation has been controversial. One
nerves have been cut).
reason that the baroreceptors have been considered
Chapter 18
Nervous Regulation of the Circulation, and Rapid Control of Arterial Pressure
211
NORMAL
200
6
5
Normal
100
4
3
0
2
24
1
Denervated
DENERVATED
200
0
0
50
100
150
200
250
Mean arterial pressure (mm Hg)
100
Figure 18-9
Frequency distribution curves of the arterial pressure for a 24-hour
period in a normal dog and in the same dog several weeks after
0
the baroreceptors had been denervated. (Redrawn from Cowley
Time (min)
AW Jr, Liard JP, Guyton AC: Role of baroreceptor reflex in daily
24
control of arterial blood pressure and other variables in dogs. Circ
Res 32:564, 1973. By permission of the American Heart Associa-
tion, Inc.)
Figure 18-8
Two-hour records of arterial pressure in a normal dog (above) and
in the same dog (below) several weeks after the baroreceptors
long-term blood pressure regulation, especially by
had been denervated. (Redrawn from Cowley AW Jr, Liard JF,
influencing sympathetic nerve activity of the kidneys.
Guyton AC: Role of baroreceptor reflex in daily control of arterial
For example, with prolonged increases in arterial pres-
blood pressure and other variables in dogs. Circ Res 32:564,
sure, the baroreceptor reflexes may mediate decreases
1973. By permission of the American Heart Association, Inc.)
in renal sympathetic nerve activity that promote
increased excretion of sodium and water by the
kidneys. This, in turn, causes a gradual decrease in
by some physiologists to be relatively unimportant in
blood volume, which helps to restore arterial pressure
chronic regulation of arterial pressure chronically is
toward normal. Thus, long-term regulation of mean
that they tend to reset in 1 to 2 days to the pressure
arterial pressure by the baroreceptors requires
level to which they are exposed. That is, if the arterial
interaction with additional systems, principally the
pressure rises from the normal value of 100 mm Hg to
renal-body fluid-pressure control system (along with
160 mm Hg, a very high rate of baroreceptor impulses
its associated nervous and hormonal mechanisms), dis-
are at first transmitted. During the next few minutes,
cussed in Chapters 19 and 29.
the rate of firing diminishes considerably; then it
diminishes much more slowly during the next 1 to 2
Control of Arterial Pressure by the Carotid and Aortic
days, at the end of which time the rate of firing will
Chemoreceptors—Effect of Oxygen Lack on Arterial Pressure.
have returned to nearly normal despite the fact that
Closely associated with the baroreceptor pressure
the mean arterial pressure still remains at 160 mm Hg.
control system is a chemoreceptor reflex that operates in
Conversely, when the arterial pressure falls to a very
much the same way as the baroreceptor reflex except
low level, the baroreceptors at first transmit no
that chemoreceptors, instead of stretch receptors, initi-
ate the response.
impulses, but gradually, over 1 to 2 days, the rate of
The chemoreceptors are chemosensitive cells sensi-
baroreceptor firing returns toward the control level.
tive to oxygen lack, carbon dioxide excess, and hydro-
This “resetting” of the baroreceptors may attenuate
gen ion excess. They are located in several small
their potency as a control system for correcting dis-
chemoreceptor organs about 2 millimeters in size (two
turbances that tend to change arterial pressure for
carotid bodies, one of which lies in the bifurcation of
longer than a few days at a time. Experimental studies,
each common carotid artery, and usually one to three
however, have suggested that the baroreceptors do
aortic bodies adjacent to the aorta). The chemo-
not completely reset and may therefore contribute to
receptors excite nerve fibers that, along with the
212
Unit IV
The Circulation
baroreceptor fibers, pass through Hering’s nerves and
All these mechanisms that tend to return the blood
the vagus nerves into the vasomotor center of the brain
volume back toward normal after a volume overload act
stem.
indirectly as pressure controllers as well as blood
Each carotid or aortic body is supplied with an
volume controllers because excess volume drives the
abundant blood flow through a small nutrient artery, so
heart to greater cardiac output and leads, therefore, to
that the chemoreceptors are always in close contact
greater arterial pressure. This volume reflex mechanism
with arterial blood. Whenever the arterial pressure falls
is discussed again in Chapter 29, along with other mech-
below a critical level, the chemoreceptors become stim-
anisms of blood volume control.
ulated because diminished blood flow causes decreased
oxygen as well as excess buildup of carbon dioxide and
Atrial Reflex Control of Heart Rate (the Bainbridge Reflex). An
hydrogen ions that are not removed by the slowly
increase in atrial pressure also causes an increase in
flowing blood.
heart rate, sometimes increasing the heart rate as much
The signals transmitted from the chemoreceptors
as 75 per cent. A small part of this increase is caused by
excite the vasomotor center, and this elevates the arterial
a direct effect of the increased atrial volume to stretch
pressure back toward normal. However, this chemo-
the sinus node: it was pointed out in Chapter 10 that
receptor reflex is not a powerful arterial pressure con-
such direct stretch can increase the heart rate as much
troller until the arterial pressure falls below 80 mm Hg.
as 15 per cent. An additional 40 to 60 per cent increase
Therefore, it is at the lower pressures that this reflex
in rate is caused by a nervous reflex called the Bain-
becomes important to help prevent still further fall in
bridge reflex. The stretch receptors of the atria that elicit
pressure.
the Bainbridge reflex transmit their afferent signals
The chemoreceptors are discussed in much more
through the vagus nerves to the medulla of the brain.
detail in Chapter 41 in relation to respiratory control,
Then efferent signals are transmitted back through
in which they play a far more important role than in
vagal and sympathetic nerves to increase heart rate and
pressure control.
strength of heart contraction. Thus, this reflex helps
prevent damming of blood in the veins, atria, and pul-
Atrial and Pulmonary Artery Reflexes That Help Regulate Arterial
monary circulation.
Pressure and Other Circulatory Factors. Both the atria and
the pulmonary arteries have in their walls stretch recep-
Central Nervous System Ischemic
tors called low-pressure receptors. They are similar to
Response—Control of Arterial
the baroreceptor stretch receptors of the large systemic
arteries. These low-pressure receptors play an important
Pressure by the Brain’s Vasomotor
role, especially in minimizing arterial pressure changes
Center in Response to Diminished
in response to changes in blood volume. To give an
Brain Blood Flow
example, if 300 milliliters of blood suddenly are infused
into a dog with allreceptors intact, the arterial pressure
Most nervous control of blood pressure is achieved
rises only about 15 mm Hg. With the arterial barorecep-
by reflexes that originate in the baroreceptors, the
tors denervated, the pressure rises about 40 mm Hg. If
chemoreceptors, and the low-pressure receptors, all of
the low-pressure receptors also are denervated, the pres-
which are located in the peripheral circulation outside
sure rises about 100 mm Hg.
the brain. However, when blood flow to the vasomotor
Thus, one can see that even though the low-pressure
center in the lower brain stem becomes decreased
receptors in the pulmonary artery and in the atria
severely enough to cause nutritional deficiency—that is,
cannot detect the systemic arterial pressure, they do
to cause cerebral ischemia—the vasoconstrictor and
detect simultaneous increases in pressure in the low-
cardioaccelerator neurons in the vasomotor center
pressure areas of the circulation caused by increase in
respond directly to the ischemia and become strongly
volume, and they elicit reflexes parallel to the barore-
excited. When this occurs, the systemic arterial pressure
ceptor reflexes to make the total reflex system more
often rises to a level as high as the heart can possibly
potent for control of arterial pressure.
pump. This effect is believed to be caused by failure of
the slowly flowing blood to carry carbon dioxide away
Atrial Reflexes That Activate the Kidneys—The “Volume Reflex.”
from the brain stem vasomotor center: at low levels of
Stretch of the atria also causes significant reflex dilation
blood flow to the vasomotor center, the local con-
of the afferent arterioles in the kidneys. And still
centration of carbon dioxide increases greatly and
other signals are transmitted simultaneously from the
has an extremely potent effect in stimulating the sym-
atria to the hypothalamus to decrease secretion of
pathetic vasomotor nervous control areas in the brain’s
antidiuretic hormone. The decreased afferent arteriolar
medulla.
resistance in the kidneys causes the glomerular capillary
It is possible that other factors, such as buildup of
pressure to rise, with resultant increase in filtration of
lactic acid and other acidic substances in the vasomotor
fluid into the kidney tubules. The diminution of antidi-
center, also contribute to the marked stimulation and
uretic hormone diminishes the reabsorption of water
elevation in arterial pressure. This arterial pressure ele-
from the tubules. Combination of these two effects—
vation in response to cerebral ischemia is known as the
increase in glomerular filtration and decrease in reab-
central nervous system ischemic response, or simply CNS
sorption of the fluid—increases fluid loss by the kidneys
ischemic response.
and reduces an increased blood volume back toward
The magnitude of the ischemic effect on vasomotor
normal. (We will also see in Chapter 19 that atrial
activity is tremendous: it can elevate the mean arterial
stretch caused by increased blood volume also elicits a
pressure for as long as 10 minutes sometimes to as high
hormonal effect on the kidneys—release of atrial natri-
as 250 mm Hg. The degree of sympathetic vasoconstric-
uretic peptide that adds still further to the excretion of
tion caused by intense cerebral ischemia is often so great
fluid in the urine and return of blood volume toward
that some of the peripheral vessels become totally or
normal.)
almost totally occluded. The kidneys, for instance, often
Chapter 18
Nervous Regulation of the Circulation, and Rapid Control of Arterial Pressure
213
Pen
Arterial
recorder
pressure
Zero
CSF pressure CSF pressure
pressure
raised
reduced
Moving paper
Figure 18-10
“Cushing reaction,” showing a
rapid rise in arterial pressure
Connector to
Arterial
Pressure
subarachnoid
pressure
resulting from increased cere-
bottle
space
brospinal fluid (CSF) pressure.
transducer
entirely cease their production of urine because of renal
Special Features of Nervous
arteriolar constriction in response to the sympathetic
Control of Arterial Pressure
discharge. Therefore, the CNS ischemic response is one
of the most powerful of all the activators of the sympa-
Role of the Skeletal Nerves and
thetic vasoconstrictor system.
Skeletal Muscles in Increasing
Importance of the CNS Ischemic Response as a Regulator of Arte-
Cardiac Output and
rial Pressure. Despite the powerful nature of the CNS
Arterial Pressure
ischemic response, it does not become significant until
the arterial pressure falls far below normal, down to
Although most rapidly acting nervous control of the cir-
60 mm Hg and below, reaching its greatest degree of
culation is effected through the autonomic nervous
stimulation at a pressure of 15 to 20 mm Hg. Therefore,
system, at least two conditions in which the skeletal
it is not one of the normal mechanisms for regulating
nerves and muscles also play major roles in circulatory
arterial pressure. Instead, it operates principally as an
responses are the following.
emergency pressure control system that acts rapidly and
very powerfully to prevent further decrease in arterial
Abdominal Compression Reflex. When a baroreceptor or
pressure whenever blood flow to the brain decreases dan-
chemoreceptor reflex is elicited, nerve signals are trans-
gerously close to the lethal level. It is sometimes called
mitted simultaneously through skeletal nerves to skele-
the “last ditch stand” pressure control mechanism.
tal muscles of the body, particularly to the abdominal
muscles. This compresses all the venous reservoirs of
Cushing Reaction. The so-called Cushing reaction is a
the abdomen, helping to translocate blood out of the
special type of CNS ischemic response that results from
abdominal vascular reservoirs toward the heart. As a
increased pressure of the cerebrospinal fluid around
result, increased quantities of blood are made available
the brain in the cranial vault. For instance, when the
for the heart to pump. This overall response is called the
cerebrospinal fluid pressure rises to equal the arterial
abdominal compression reflex. The resulting effect on
pressure, it compresses the whole brain as well as the
the circulation is the same as that caused by sympathetic
arteries in the brain and cuts off the blood supply to the
vasoconstrictor impulses when they constrict the veins:
brain. This initiates a CNS ischemic response that causes
an increase in both cardiac output and arterial pressure.
the arterial pressure to rise. When the arterial pressure
The abdominal compression reflex is probably much
has risen to a level higher than the cerebrospinal fluid
more important than has been realized in the past
pressure, blood will flow once again into the vessels of
because it is well known that people whose skeletal
the brain to relieve the brain ischemia. Ordinarily, the
muscles have been paralyzed are considerably more
blood pressure comes to a new equilibrium level slightly
prone to hypotensive episodes than are people with
higher than the cerebrospinal fluid pressure, thus allow-
normal skeletal muscles.
ing blood to begin again to flow through the brain. A
typical Cushing reaction is shown in Figure
18-10,
Increased Cardiac Output and Arterial Pressure Caused by
caused in this instance by pumping fluid under pressure
Skeletal Muscle Contraction During Exercise. When the
into the cranial vault around the brain. The Cushing
skeletal muscles contract during exercise, they compress
reaction helps protect the vital centers of the brain from
blood vessels throughout the body. Even anticipation of
loss of nutrition if ever the cerebrospinal fluid pressure
exercise tightens the muscles, thereby compressing
rises high enough to compress the cerebral arteries.
the vessels in the muscles and in the abdomen. The
214
Unit IV
The Circulation
resulting effect is to translocate blood from the periph-
eral vessels into the heart and lungs and, therefore, to
increase the cardiac output. This is an essential effect in
helping to cause the fivefold to sevenfold increase in
200
cardiac output that sometimes occurs in heavy exercise.
100
160
The increase in cardiac output in turn is an essential
120
ingredient in increasing the arterial pressure during
60
80
exercise, an increase usually from a normal mean of
100 mm Hg up to 130 to 160 mm Hg.
40
0
A
B
Respiratory Waves in the
Arterial Pressure
With each cycle of respiration, the arterial pressure
usually rises and falls
4 to 6 mm Hg in a wavelike
Figure 18-11
manner, causing respiratory waves in the arterial pres-
sure. The waves result from several different effects,
A, Vasomotor waves caused by oscillation of the CNS ischemic
some of which are reflex in nature, as follows:
response. B, Vasomotor waves caused by baroreceptor reflex
1. Many of the “breathing signals” that arise in the
oscillation.
respiratory center of the medulla “spill over” into
the vasomotor center with each respiratory cycle.
2. Every time a person inspires, the pressure in the
thoracic cavity becomes more negative than usual,
causing the blood vessels in the chest to expand.
pressure in turn reduces the baroreceptor stimulation
This reduces the quantity of blood returning to the
and allows the vasomotor center to become active once
left side of the heart and thereby momentarily
again, elevating the pressure to a high value. The
decreases the cardiac output and arterial pressure.
response is not instantaneous, and it is delayed until a
3. The pressure changes caused in the thoracic vessels
few seconds later. This high pressure then initiates
by respiration can excite vascular and atrial stretch
another cycle, and the oscillation continues on and on.
receptors.
The chemoreceptor reflex can also oscillate to give the
Although it is difficult to analyze the exact relations
same type of waves. This reflex usually oscillates simul-
of all these factors in causing the respiratory pressure
taneously with the baroreceptor reflex. It probably plays
waves, the net result during normal respiration is usually
the major role in causing vasomotor waves when the
an increase in arterial pressure during the early part
arterial pressure is in the range of 40 to 80 mm Hg
of expiration and a decrease in pressure during the
because in this low range, chemoreceptor control of the
remainder of the respiratory cycle. During deep respi-
circulation becomes powerful, whereas baroreceptor
ration, the blood pressure can rise and fall as much as
control becomes weaker.
20 mm Hg with each respiratory cycle.
Oscillation of the CNS Ischemic Response. The record in
Figure 18-11A resulted from oscillation of the CNS
Arterial Pressure “Vasomotor”
ischemic pressure control mechanism. In this experi-
Waves—Oscillation of Pressure
ment, the cerebrospinal fluid pressure was raised to
Reflex Control Systems
160 mm Hg, which compressed the cerebral vessels and
initiated a CNS ischemic pressure response up to
Often while recording arterial pressure from an animal,
200 mm Hg. When the arterial pressure rose to such a
in addition to the small pressure waves caused by res-
high value, the brain ischemia was relieved and the sym-
piration, some much larger waves are also noted—as
pathetic nervous system became inactive. As a result,
great as 10 to 40 mm Hg at times—that rise and fall
the arterial pressure fell rapidly back to a much lower
more slowly than the respiratory waves. The duration of
value, causing brain ischemia once again. The ischemia
each cycle varies from 26 seconds in the anesthetized
then initiated another rise in pressure. Again the
dog to 7 to 10 seconds in the unanesthetized human.
ischemia was relieved and again the pressure fell. This
These waves are called vasomotor waves or “Mayer
repeated itself cyclically as long as the cerebrospinal
waves.” Such records are demonstrated in Figure 18-11,
fluid pressure remained elevated.
showing the cyclical rise and fall in arterial pressure.
Thus, any reflex pressure control mechanism can
The cause of vasomotor waves is “reflex oscillation”
oscillate if the intensity of “feedback” is strong enough
of one or more nervous pressure control mechanisms,
and if there is a delay between excitation of the pres-
some of which are the following.
sure receptor and the subsequent pressure response.
The vasomotor waves are of considerable theoretical
Oscillation of the Baroreceptor and Chemoreceptor Reflexes.
importance because they show that the nervous reflexes
The vasomotor waves of Figure 18-11B are often seen
that control arterial pressure obey the same principles
in experimental pressure recordings, although usually
as those applicable to mechanical and electrical control
much less intense than shown in the figure. They are
systems. For instance, if the feedback “gain” is too great
caused mainly by oscillation of the baroreceptor reflex.
in the guiding mechanism of an automatic pilot for an
That is, a high pressure excites the baroreceptors;
airplane and there is also delay in response time of the
this then inhibits the sympathetic nervous system and
guiding mechanism, the plane will oscillate from side to
lowers the pressure a few seconds later. The decreased
side instead of following a straight course.
Chapter 18
Nervous Regulation of the Circulation, and Rapid Control of Arterial Pressure
215
References
Hall JE, Hildebrandt DA, Kuo J: Obesity hypertension: role
of leptin and sympathetic nervous system. Am J Hyper-
tens 14:103S, 2001.
Antunes-Rodrigues J, De Castro M, Elias LLK, et al: Neu-
Ketch T, Biaggioni I, Robertson R, Robertson D: Four faces
roendocrine control of body fluid metabolism. Physiol Rev
of baroreflex failure: hypertensive crisis, volatile hyper-
84: 169, 2004.
tension, orthostatic tachycardia, and malignant vagotonia.
Cao WH, Fan W, Morrison SF: Medullary pathways mediat-
Circulation 105:2518, 2002.
ing specific sympathetic responses to activation of dorso-
Krieger EM, Da Silva GJ, Negrao CE: Effects of exercise
medial hypothalamus. Neuroscience 126:229, 2004.
training on baroreflex control of the cardiovascular
Cowley AW Jr, Guyton AC: Baroreceptor reflex contribu-
system. Ann N Y Acad Sci 940:338, 2001.
tion in angiotensin II-induced hypertension. Circulation
Lohmeier TE, Lohmeier JR, Warren S, et al: Sustained acti-
50:61, 1974.
vation of the central baroreceptor pathway in angiotensin
DiBona GF: Peripheral and central interactions between
hypertension. Hypertension 39:550, 2002.
the renin-angiotensin system and the renal sympathetic
Lohmeier TE: The sympathetic nervous system and long-
nerves in control of renal function. Ann N Y Acad Sci
term blood pressure regulation. Am J Hypertens 14:147S,
940:395, 2001.
2001.
DiCarlo SE, Bishop VS: Central baroreflex resetting as a
Malpas SC: What sets the long-term level of sympathetic
means of increasing and decreasing sympathetic outflow
nerve activity: is there a role for arterial baroreceptors?
and arterial pressure. Ann N Y Acad Sci 940:324, 2001.
Am J Physiol Regul Integr Comp Physiol 286:R1, 2004.
Esler M, Lambert G, Brunner-La Rocca HP, et al: Sympa-
Mifflin SW: What does the brain know about blood pressure?
thetic nerve activity and neurotransmitter release in
News Physiol Sci 16:266, 2001.
humans: translation from pathophysiology into clinical
Morrison SF: Differential control of sympathetic outflow.
practice. Acta Physiol Scand 177:275, 2003.
Am J Physiol Regul Integr Comp Physiol 281:R683, 2001.
Floras JS: Arterial baroreceptor and cardiopulmonary reflex
Sved AF, Ito S, Sved JC: Brainstem mechanisms of hyper-
control of sympathetic outflow in human heart failure.
tension: role of the rostral ventrolateral medulla. Curr
Ann N Y Acad Sci 940:500, 2001.
Hypertens Rep 5:262, 2003 .
Felder RB, Francis J, Zhang ZH, et al: Heart failure and the
Thrasher TN: Unloading arterial baroreceptors causes neu-
brain: new perspectives. Am J Physiol Regul Integr Comp
rogenic hypertension. Am J Physiol Regul Integr Comp
Physiol 284:R259, 2003.
Physiol 282:R1044, 2002.
Goldstein DS, Robertson D, Esler M, et al: Dysautonomias:
Zucker IH, Wang W, Pliquett RU, et al: The regulation
clinical disorders of the autonomic nervous system. Ann
of sympathetic outflow in heart failure. The roles of
Intern Med 137:753, 2002.
angiotensin II, nitric oxide, and exercise training. Ann
Guyton AC: Arterial Pressure and Hypertension. Philadel-
N Y Acad Sci 940:431, 2001.
phia: WB Saunders Co, 1980.
C H A P T E R
1
9
Dominant Role of the Kidney
in Long-Term Regulation of
Arterial Pressure and in
Hypertension: The Integrated
System for Pressure Control
Short-term control of arterial pressure by the sym-
pathetic nervous system, as discussed in Chapter 18,
occurs primarily through the effects of the nervous
system on total peripheral vascular resistance and
capacitance, and on cardiac pumping ability.
The body, however, also has powerful mecha-
nisms for regulating arterial pressure week
after week and month after month. This long-term
control of arterial pressure is closely intertwined with homeostasis of body fluid
volume, which is determined by the balance between the fluid intake and
output. For long-term survival, fluid intake and output must be precisely bal-
anced, a task that is performed by multiple nervous and hormonal controls, and
by local control systems within the kidneys that regulate their excretion of salt
and water. In this chapter we discuss these renal-body fluid systems that play
a dominant role in long-term blood pressure regulation.
Renal-Body Fluid System for Arterial
Pressure Control
The renal-body fluid system for arterial pressure control is a simple one: When
the body contains too much extracellular fluid, the blood volume and arterial
pressure rise. The rising pressure in turn has a direct effect to cause the kidneys
to excrete the excess extracellular fluid, thus returning the pressure back toward
normal.
In the phylogenetic history of animal development, this renal-body fluid
system for pressure control is a primitive one. It is fully operative in one of the
lowest of vertebrates, the hagfish. This animal has a low arterial pressure, only
8 to 14 mm Hg, and this pressure increases almost directly in proportion to its
blood volume. The hagfish continually drinks sea water, which is absorbed into
its blood, increasing the blood volume as well as the pressure. However, when
the pressure rises too high, the kidney simply excretes the excess volume into
the urine and relieves the pressure. At low pressure, the kidney excretes far less
fluid than is ingested. Therefore, because the hagfish continues to drink, extra-
cellular fluid volume, blood volume, and pressure all build up again to the higher
levels.
Throughout the ages, this primitive mechanism of pressure control has sur-
vived almost exactly as it functions in the hagfish; in the human being, kidney
output of water and salt is just as sensitive to pressure changes as in the hagfish,
if not more so. Indeed, an increase in arterial pressure in the human of only a
few millimeters of mercury can double renal output of water, which is called
pressure diuresis, as well as double the output of salt, which is called pressure
natriuresis.
216
Chapter 19
The Integrated System for Pressure Control
217
4000
8
7
3000
6
2000
5
4
1000
3
4
3
2
2
1
1
0
0
0
20
40
60
80 100 120 140 160 180 200
225
Arterial pressure (mm Hg)
200
175
150
125
Figure 19-1
100
75
Typical renal urinary output curve measured in a perfused isolated
kidney, showing pressure diuresis when the arterial pressure rises
50
Infusion period
above normal.
0 10 20 30 40 50 60
120
Time (minutes)
In the human being, the renal-body fluid system for
arterial pressure control, just as in the hagfish, is the
fundamental basis for long-term arterial pressure
Figure 19-2
control. However, through the stages of evolution,
Increases in cardiac output, urinary output, and arterial pressure
multiple refinements have been added to make this
caused by increased blood volume in dogs whose nervous pres-
system much more exact in its control in the human
sure control mechanisms had been blocked. This figure shows
being. An especially important refinement, as we shall
return of arterial pressure to normal after about an hour of fluid
see, has been addition of the renin-angiotensin
loss into the urine. (Courtesy Dr. William Dobbs.)
mechanism.
in cardiac output to about double normal and increase
Quantitation of Pressure Diuresis as
in mean arterial pressure to 205 mm Hg, 115 mm Hg
a Basis for Arterial Pressure Control
above its resting level. Shown by the middle curve is
the effect of this increased arterial pressure on urine
Figure 19-1 shows the approximate average effect of
output, which increased
12-fold. Along with this
different arterial pressure levels on urinary volume
tremendous loss of fluid in the urine, both the cardiac
output by an isolated kidney, demonstrating markedly
output and the arterial pressure returned to normal
increased output of volume as the pressure rises. This
during the subsequent hour. Thus, one sees an extreme
increased urinary output is the phenomenon of pres-
capability of the kidneys to eliminate fluid volume
sure diuresis. The curve in this figure is called a renal
from the body in response to high arterial pressure and
urinary output curve or a renal function curve. In the
in so doing to return the arterial pressure back to
human being, at an arterial pressure of 50 mm Hg, the
normal.
urine output is essentially zero. At 100 mm Hg it is
normal, and at 200 mm Hg it is about six to eight times
Graphical Analysis of Pressure Control by the Renal-Body Fluid
normal. Furthermore, not only does increasing the
Mechanism, Demonstrating an
“Infinite Feedback Gain”
arterial pressure increase urine volume output, but it
Feature. Figure 19-3 shows a graphical method that
causes approximately equal increase in sodium output,
can be used for analyzing arterial pressure control by
which is the phenomenon of pressure natriuresis.
the renal-body fluid system. This analysis is based on
two separate curves that intersect each other: (1) the
An Experiment Demonstrating the Renal-Body Fluid System for
renal output curve for water and salt in response to
Arterial Pressure Control. Figure 19-2 shows the results
rising arterial pressure, which is the same renal output
of a research experiment in dogs in which all the
curve as that shown in Figure 19-1, and (2) the curve
nervous reflex mechanisms for blood pressure control
(or line) that represents the net water and salt intake.
were first blocked. Then the arterial pressure was sud-
Over a long period, the water and salt output must
denly elevated by infusing about 400 milliliters of
equal the intake. Furthermore, the only place on the
blood intravenously. Note the instantaneous increase
graph in Figure 19-3 at which output equals intake is
218
Unit IV The Circulation
Renal output of water and salt
8
A
Water and salt intake
6
8
4
6
Normal
Elevated
2
pressure
4
0
0
50
100
150
200
250
Equilibrium point
2
8
0
B
0
50
100
150
250
Elevated
Arterial pressure (mm Hg)
6
pressure
4
Normal
Figure 19-3
2
Analysis of arterial pressure regulation by equating the “renal
output curve” with the “salt and water intake curve.” The equilib-
0
0
50
100
150
200
250
rium point describes the level to which the arterial pressure will
be regulated. (That small portion of the salt and water intake that
Arterial pressure (mm Hg)
is lost from the body through nonrenal routes is ignored in this and
similar figures in this chapter.)
Figure 19-4
Two ways in which the arterial pressure can be increased: A, by
where the two curves intersect, which is called the
shifting the renal output curve in the right-hand direction toward
equilibrium point. Now, let us see what happens if the
a higher pressure level or B, by increasing the intake level of salt
arterial pressure becomes some value that is different
and water.
from that at the equilibrium point.
First, assume that the arterial pressure rises to
150 mm Hg. At this level, the graph shows that renal
output of water and salt is about three times as great
As long as the two curves representing (1) renal
as the intake. Therefore, the body loses fluid, the blood
output of salt and water and (2) intake of salt and
volume decreases, and the arterial pressure decreases.
water remain exactly as they are shown in Figure 19-3,
Furthermore, this “negative balance” of fluid will not
the long-term mean arterial pressure level will always
cease until the pressure falls all the way back exactly
readjust exactly to 100 mm Hg, which is the pressure
to the equilibrium level. Indeed, even when the arte-
level depicted by the equilibrium point of this figure.
rial pressure is only 1 mm Hg greater than the equi-
Furthermore, there are only two ways in which the
librium level, there still is slightly more loss of water
pressure of this equilibrium point can be changed from
and salt than intake, so that the pressure continues to
the 100 mm Hg level. One of these is by shifting the
fall that last
1 mm Hg until the pressure eventually
pressure level of the renal output curve for salt and
returns exactly to the equilibrium point.
water; and the other is by changing the level of the
If the arterial pressure falls below the equilibrium
water and salt intake line. Therefore, expressed simply,
point, the intake of water and salt is greater than the
the two primary determinants of the long-term arte-
output. Therefore, body fluid volume increases, blood
rial pressure level are as follows:
volume increases, and the arterial pressure rises until
1. The degree of pressure shift of the renal output
once again it returns exactly to the equilibrium point.
curve for water and salt
This return of the arterial pressure always exactly
2. The level of the water and salt intake line
back to the equilibrium point is the infinite feedback
Operation of these two determinants in the control
gain principle for control of arterial pressure by the
of arterial pressure is demonstrated in Figure 19-4. In
renal-body fluid mechanism.
Figure 19-4A, some abnormality of the kidneys has
caused the renal output curve to shift 50 mm Hg in the
Two Determinants of the Long-Term Arterial Pressure Level. In
high-pressure direction (to the right). Note that the
Figure 19-3, one can also see that two basic long-term
equilibrium point has also shifted to 50 mm Hg higher
factors determine the long-term arterial pressure level.
than normal. Therefore, one can state that if the renal
This can be explained as follows.
output curve shifts to a new pressure level, so will the
Chapter 19
The Integrated System for Pressure Control
219
arterial pressure follow to this new pressure level
within a few days.
Figure 19-4B shows how a change in the level of salt
and water intake also can change the arterial pressure.
In this case, the intake level has increased fourfold and
the equilibrium point has shifted to a pressure level of
200
160 mm Hg, 60 mm Hg above the normal level. Con-
versely, a decrease in the intake level would reduce the
arterial pressure.
150
Thus, it is impossible to change the long-term mean
arterial pressure level to a new value without changing
one or both of the two basic determinants of long-term
100
arterial pressure—either (1) the level of salt and water
Arterial pressure
intake or (2) the degree of shift of the renal function
curve along the pressure axis. However, if either of
50
these is changed, one finds the arterial pressure there-
after to be regulated at a new pressure level, at the
pressure level at which the two new curves intersect.
0
40
60
80
100
120
140
160
Failure of Increased Total Peripheral
Total peripheral resistance
(per cent of normal)
Resistance to Elevate the Long-Term Level
of Arterial Pressure if Fluid Intake and Renal
Function Do Not Change
Now is the chance for the reader to see whether he or
Figure 19-5
she really understands the renal-body fluid mecha-
nism for arterial pressure control. Recalling the basic
Relations of total peripheral resistance to the long-term levels of
arterial pressure and cardiac output in different clinical abnor-
equation for arterial pressure—arterial pressure equals
malities. In these conditions, the kidneys were functioning nor-
cardiac output times total peripheral resistance—it is
mally. Note that changing the whole-body total peripheral
clear that an increase in total peripheral resistance
resistance caused equal and opposite changes in cardiac output
should elevate the arterial pressure. Indeed, when the
but in all cases had no effect on arterial pressure. (Redrawn from
Guyton AC: Arterial Pressure and Hypertension. Philadelphia: WB
total peripheral resistance is acutely increased, the arte-
Saunders Co, 1980.)
rial pressure does rise immediately. Yet if the kidneys
continue to function normally, the acute rise in arterial
pressure usually is not maintained. Instead, the arte-
rial pressure returns all the way to normal within a day
hypertension by shifting the renal function curve to a
or so. Why?
higher pressure level, in the manner shown in Figure
The answer to this is the following: Increasing resist-
19-4A. We see an example of this later in this chapter
ance in the blood vessels everywhere else in the body
when we discuss hypertension caused by vasoconstric-
besides in the kidneys does not change the equilibrium
tor mechanisms. But it is the increase in renal resistance
point for blood pressure control as dictated by the
that is the culprit, not the increased total peripheral
kidneys (see again Figures 19-3 and 19-4). Instead, the
resistance—an important distinction!)
kidneys immediately begin to respond to the high arte-
rial pressure, causing pressure diuresis and pressure
Increased Fluid Volume Can Elevate Arterial
natriuresis. Within hours, large amounts of salt and
Pressure by Increasing Cardiac Output or
water are lost from the body, and this continues until
Total Peripheral Resistance
the arterial pressure returns exactly to the pressure
The overall mechanism by which increased extracellu-
level of the equilibrium point.
lar fluid volume elevates arterial pressure is given in
As proof of this principle that changes in total
the schema of Figure 19-6. The sequential events are
peripheral resistance do not affect the long-term level
(1) increased extracellular fluid volume (2) increases
of arterial pressure if function of the kidneys is still
the blood volume, which (3) increases the mean cir-
normal, carefully study Figure 19-5. This figure shows
culatory filling pressure, which (4) increases venous
the approximate cardiac outputs and the arterial pres-
return of blood to the heart, which
(5) increases
sures in different clinical conditions in which the long-
cardiac output, which (6) increases arterial pressure.
term total peripheral resistance is either much less than
Note especially in this schema the two ways in which
or much greater than normal, but kidney excretion of
an increase in cardiac output can increase the arterial
salt and water is normal. Note in all these different
pressure. One of these is the direct effect of increased
clinical conditions that the arterial pressure is also
cardiac output to increase the pressure, and the other
exactly normal.
is an indirect effect to raise total peripheral vascular
(A word of caution! Many times when the total
resistance through autoregulation of blood flow. The
peripheral resistance increases, this increases the
second effect can be explained as follows.
intrarenal vascular resistance at the same time, which
Referring back to Chapter 17, let us recall that
alters the function of the kidney and can cause
whenever an excess amount of blood flows through a
220
Unit IV
The Circulation
pressure than is an increase in water intake. The reason
for this is that pure water is normally excreted by the
kidneys almost as rapidly as it is ingested, but salt
Increased extracellular fluid volume
is not excreted so easily. As salt accumulates in the
body, it also indirectly increases the extracellular fluid
volume for two basic reasons:
Increased blood volume
1. When there is excess salt in the extracellular fluid,
the osmolality of the fluid increases, and this in
turn stimulates the thirst center in the brain,
Increased mean circulatory filling pressure
making the person drink extra amounts of
water to return the extracellular salt
concentration to normal. This increases the
Increased venous return of blood to the heart
extracellular fluid volume.
2. The increase in osmolality caused by the excess
salt in the extracellular fluid also stimulates the
Increased cardiac output
hypothalamic-posterior pituitary gland secretory
mechanism to secrete increased quantities of
antidiuretic hormone. (This is discussed in Chapter
Autoregulation
28.) The antidiuretic hormone then causes the
kidneys to reabsorb greatly increased quantities
of water from the renal tubular fluid, thereby
Increased total
diminishing the excreted volume of urine but
peripheral resistance
increasing the extracellular fluid volume.
Thus, for these important reasons, the amount of salt
Increased arterial pressure
that accumulates in the body is the main determinant
of the extracellular fluid volume. Because only small
increases in extracellular fluid and blood volume can
often increase the arterial pressure greatly, accumula-
tion of even a small amount of extra salt in the body
Figure 19-6
can lead to considerable elevation of arterial pressure.
Sequential steps by which increased extracellular fluid volume
increases the arterial pressure. Note especially that increased
cardiac output has both a direct effect to increase arterial pres-
sure and an indirect effect by first increasing the total peripheral
Chronic Hypertension (High Blood
resistance.
Pressure) Is Caused by Impaired
Renal Fluid Excretion
tissue, the local tissue vasculature constricts and
When a person is said to have chronic hypertension (or
decreases the blood flow back toward normal. This
“high blood pressure”), it is meant that his or her mean
phenomenon is called “autoregulation,” which means
arterial pressure is greater than the upper range of the
simply regulation of blood flow by the tissue itself.
accepted normal measure. A mean arterial pressure
When increased blood volume increases the cardiac
greater than 110 mm Hg (normal is about 90 mm Hg)
output, the blood flow increases in all tissues of the
is considered to be hypertensive. (This level of mean
body, so that this autoregulation mechanism constricts
pressure occurs when the diastolic blood pressure is
blood vessels all over the body. This in turn increases
greater than about 90 mm Hg and the systolic pressure
the total peripheral resistance.
is greater than about 135 mm Hg.) In severe hyper-
Finally, because arterial pressure is equal to cardiac
tension, the mean arterial pressure can rise to 150
output times total peripheral resistance, the secondary
to
170 mm Hg, with diastolic pressure as high as
increase in total peripheral resistance that results from
130 mm Hg and systolic pressure occasionally as high
the autoregulation mechanism helps greatly in increas-
as 250 mm Hg.
ing the arterial pressure. For instance, only a 5 to 10
Even moderate elevation of arterial pressure leads
per cent increase in cardiac output can increase the
to shortened life expectancy. At severely high pres-
arterial pressure from the normal mean arterial pres-
sures—mean arterial pressures 50 per cent or more
sure of 100 mm Hg up to 150 mm Hg. In fact, the slight
above normal—a person can expect to live no more
increase in cardiac output is often unmeasurable.
than a few more years unless appropriately treated.
The lethal effects of hypertension are caused mainly
Importance of Salt (NaCl) in the Renal-Body
in three ways:
Fluid Schema for Arterial Pressure Regulation
1. Excess workload on the heart leads to early heart
Although the discussions thus far have emphasized the
failure and coronary heart disease, often causing
importance of volume in regulation of arterial pres-
death as a result of a heart attack.
sure, experimental studies have shown that an increase
2. The high pressure frequently damages a major
in salt intake is far more likely to elevate the arterial
blood vessel in the brain, followed by death of
Chapter 19
The Integrated System for Pressure Control
221
0.9% NaCl
Tap water
0.9% NaCl
150
140
130
120
35-45% of left
Entire right
kidney removed
kidney removed
110
100
0
0
20
40
60
80
100
Days
Figure 19-7
Average effect on arterial pressure of drinking 0.9 per cent saline solution instead of water in four dogs with 70 per cent of their
renal tissue removed. (Redrawn from Langston JB, Guyton AC, Douglas BH, Dorsett PE: Circ Res 12:508, 1963. By permission of the
American Heart Association, Inc.)
major portions of the brain; this is a cerebral
normal amounts of volume, and within a few days,
infarct. Clinically it is called a “stroke.” Depending
their average arterial pressure rose to about 40 mm Hg
on which part of the brain is involved, a stroke
above normal. After 2 weeks, the dogs were given tap
can cause paralysis, dementia, blindness, or
water again instead of salt solution; the pressure
multiple other serious brain disorders.
returned to normal within 2 days. Finally, at the end of
3. High pressure almost always causes injury in
the experiment, the dogs were given salt solution
the kidneys, producing many areas of renal
again, and this time the pressure rose much more
destruction and, eventually, kidney failure, uremia,
rapidly to an even higher level because the dogs had
and death.
already learned to tolerate the salt solution and there-
Lessons learned from the type of hypertension
fore drank much more. Thus, this experiment demon-
called
“volume-loading hypertension” have been
strates volume-loading hypertension.
crucial in understanding the role of the renal-body
If the reader considers again the basic determinants
fluid volume mechanism for arterial pressure regula-
of long-term arterial pressure regulation, he or she can
tion. Volume-loading hypertension means hyperten-
immediately understand why hypertension occurred in
sion caused by excess accumulation of extracellular
the volume-loading experiment of Figure 19-7. First,
fluid in the body, some examples of which follow.
reduction of the kidney mass to 30 per cent of normal
greatly reduced the ability of the kidneys to excrete
Experimental Volume-Loading Hypertension Caused by Reduced
salt and water. Therefore, salt and water accumulated
Renal Mass Along with Simultaneous Increase in Salt Intake.
in the body and in a few days raised the arterial pres-
Figure 19-7 shows a typical experiment demonstrating
sure high enough to excrete the excess salt and water
volume-loading hypertension in a group of dogs with
intake.
70 per cent of their kidney mass removed. At the first
circled point on the curve, the two poles of one of the
kidneys were removed, and at the second circled point,
Sequential Changes in Circulatory Function During the Devel-
the entire opposite kidney was removed, leaving the
opment of Volume-Loading Hypertension. It is especially
animals with only 30 per cent of normal renal mass.
instructive to study the sequential changes in circula-
Note that removal of this amount of kidney mass
tory function during progressive development of
increased the arterial pressure an average of only 6
volume-loading hypertension. Figure 19-8 shows these
mm Hg. Then, the dogs were given salt solution to
sequential changes. A week or so before the point
drink instead of water. Because salt solution fails to
labeled “0” days, the kidney mass had already been
quench the thirst, the dogs drank two to four times the
decreased to only 30 per cent of normal. Then, at this
222
Unit IV The Circulation
20
19
33%
18
17
4%
16
15
6.0
20%
5%
5.5
5.0
40%
7.0
6.5
6.0
5.5
5%
5.0
28
26
33%
24
22
20
18
-13%
Figure 19-8
150
40%
30%
Progressive changes in important circulatory
140
system variables during the first few weeks of
130
volume-loading hypertension. Note especially the
120
initial increase in cardiac output as the basic
110
cause of the hypertension. Subsequently, the auto-
0
regulation mechanism returns the cardiac output
0
2
4
6
8
10
12
14
almost to normal while simultaneously causing a
Days
secondary increase in total peripheral resistance.
(Modified from Guyton AC: Arterial Pressure and
Hypertension. Philadelphia: WB Saunders Co,
1980.)
point, the intake of salt and water was increased to
Chapter 17 and earlier in this chapter. That is, after the
about six times normal and kept at this high intake
cardiac output had risen to a high level and had initi-
thereafter. The acute effect was to increase extracellu-
ated the hypertension, the excess blood flow through
lar fluid volume, blood volume, and cardiac output to
the tissues then caused progressive constriction of the
20 to 40 per cent above normal. Simultaneously, the
local arterioles, thus returning the local blood flows
arterial pressure began to rise but not nearly so much
in all the body tissues and also the cardiac output
at first as did the fluid volumes and cardiac output. The
almost all the way back to normal, while simultane-
reason for this slower rise in pressure can be discerned
ously causing a secondary increase in total peripheral
by studying the total peripheral resistance curve, which
resistance.
shows an initial decrease in total peripheral resistance.
Note, too, that the extracellular fluid volume and
This decrease was caused by the baroreceptor mecha-
blood volume returned almost all the way back to
nism discussed in Chapter 18, which tried to prevent
normal along with the decrease in cardiac output. This
the rise in pressure. However, after 2 to 4 days, the
resulted from two factors: First, the increase in arteri-
baroreceptors adapted (reset) and were no longer able
olar resistance decreased the capillary pressure, which
to prevent the rise in pressure. At this time, the arte-
allowed the fluid in the tissue spaces to be absorbed
rial pressure had risen almost to its full height because
back into the blood. Second, the elevated arterial
of the increase in cardiac output, even though the total
pressure now caused the kidneys to excrete the excess
peripheral resistance was still almost at the normal
volume of fluid that had initially accumulated in the
level.
body.
After these early acute changes in the circulatory
Last, let us take stock of the final state of the circu-
variables had occurred, more prolonged secondary
lation several weeks after the initial onset of volume
changes occurred during the next few weeks. Espe-
loading. We find the following effects:
cially important was a progressive increase in total
1. Hypertension
peripheral resistance, while at the same time the cardiac
2. Marked increase in total peripheral resistance
3. Almost complete return of the extracellular fluid
output decreased almost all the way back to normal,
volume, blood volume, and cardiac output back to
mainly as a result of the long-term blood flow autoreg-
normal
ulation mechanism that is discussed in detail in
Chapter 19
The Integrated System for Pressure Control
223
Therefore, we can divide volume-loading hyperten-
The Renin-Angiotensin
sion into two separate sequential stages: The first stage
System: Its Role in Pressure
results from increased fluid volume causing increased
Control and in Hypertension
cardiac output. This increase in cardiac output causes
the hypertension. The second stage in volume-loading
Aside from the capability of the kidneys to control
hypertension is characterized by high blood pressure
arterial pressure through changes in extracellular
and high total peripheral resistance but return of the
fluid volume, the kidneys also have another powerful
cardiac output so near to normal that the usual meas-
mechanism for controlling pressure. It is the renin-
uring techniques frequently cannot detect an abnor-
angiotensin system.
mally elevated cardiac output.
Renin is a protein enzyme released by the kidneys
Thus, the increased total peripheral resistance in
when the arterial pressure falls too low. In turn, it
volume-loading hypertension occurs after the hyper-
raises the arterial pressure in several ways, thus
tension has developed and, therefore, is secondary to
helping to correct the initial fall in pressure.
the hypertension rather than being the cause of the
hypertension.
Components of the Renin-Angiotensin
Volume-Loading Hypertension in Patients
Who Have No Kidneys but Are Being
System
Maintained on an Artificial Kidney
When a patient is maintained on an artificial kidney, it
Figure 19-9 shows the functional steps by which the
is especially important to keep the patient’s body fluid
renin-angiotensin system helps to regulate arterial
volume at a normal level—that is, it is important
pressure.
to remove an appropriate amount of water and salt
Renin is synthesized and stored in an inactive form
each time the patient is dialyzed. If this is not done and
called prorenin in the juxtaglomerular cells (JG cells)
extracellular fluid volume is allowed to increase,
of the kidneys. The JG cells are modified smooth
hypertension almost invariably develops in exactly the
muscle cells located in the walls of the afferent
same way as shown in Figure 19-8. That is, the cardiac
output increases at first and causes hypertension. Then
the autoregulation mechanism returns the cardiac
output back toward normal while causing a secondary
increase in total peripheral resistance. Therefore, in
Decreased
the end, the hypertension is a high peripheral resist-
arterial pressure
ance type of hypertension.
Hypertension Caused by
Renin (kidney)
Primary Aldosteronism
Another type of volume-loading hypertension is
Renin substrate
caused by excess aldosterone in the body or, occa-
(angiotensinogen)
sionally, by excesses of other types of steroids. A small
tumor in one of the adrenal glands occasionally
Angiotensin I
secretes large quantities of aldosterone, which is
the condition called “primary aldosteronism.” As dis-
Converting
cussed in Chapter 29, aldosterone increases the rate of
enzyme
(lung)
reabsorption of salt and water by the tubules of the
kidneys, thereby reducing the loss of these in the
urine while at the same time causing an increase in
Angiotensin II
blood volume and extracellular fluid volume. Conse-
Angiotensinase
quently, hypertension occurs. And, if salt intake is
increased at the same time, the hypertension becomes
(Inactivated)
even greater. Furthermore, if the condition persists for
Renal retention
Vasoconstriction
months or years, the excess arterial pressure often
of salt and water
causes pathological changes in the kidneys that make
the kidneys retain even more salt and water in addi-
tion to that caused directly by the aldosterone. There-
fore, the hypertension often finally becomes lethally
Increased arterial pressure
severe.
Here again, in the early stages of this type of hyper-
tension, the cardiac output is increased, but in later
stages, the cardiac output generally returns almost to
Figure 19-9
normal while the total peripheral resistance becomes
secondarily elevated, as explained earlier in the
Renin-angiotensin vasoconstrictor mechanism for arterial pres-
chapter for primary volume-loading hypertension.
sure control.
224
Unit IV The Circulation
arterioles immediately proximal to the glomeruli. When
the arterial pressure falls, intrinsic reactions in the
kidneys themselves cause many of the prorenin mole-
100
With
cules in the JG cells to split and release renin. Most of
renin-angiotensin system
the renin enters the renal blood and then passes out
75
of the kidneys to circulate throughout the entire body.
However, small amounts of the renin do remain in the
50
Without
local fluids of the kidney and initiate several intrarenal
renin-angiotensin system
functions.
Renin itself is an enzyme, not a vasoactive sub-
25
Hemorrhage
stance. As shown in the schema of Figure
19-9,
renin acts enzymatically on another plasma protein,
0
a globulin called renin substrate (or angiotensinogen),
0
10
20
30
40
to release a
10-amino acid peptide, angiotensin I.
Minutes
Angiotensin I has mild vasoconstrictor properties but
not enough to cause significant changes in circulatory
function. The renin persists in the blood for 30 minutes
to 1 hour and continues to cause formation of still
Figure 19-10
more angiotensin I during this entire time.
Pressure-compensating effect of the renin-angiotensin vasocon-
Within a few seconds to minutes after formation of
strictor system after severe hemorrhage. (Drawn from experiments
angiotensin I, two additional amino acids are split
by Dr. Royce Brough.)
from the angiotensin I to form the
8-amino acid
peptide angiotensin II. This conversion occurs almost
entirely in the lungs while the blood flows through the
small vessels of the lungs, catalyzed by an enzyme
called converting enzyme that is present in the
endothelium of the lung vessels.
the system functioning (the system was interrupted
Angiotensin II is an extremely powerful vasocon-
by a renin-blocking antibody). Note that after hemor-
strictor, and it also affects circulatory function in other
rhage—enough to cause acute decrease of the arterial
ways as well. However, it persists in the blood only
pressure to
50 mm Hg—the arterial pressure rose
for 1 or 2 minutes because it is rapidly inactivated by
back to
83 mm Hg when the renin-angiotensin
multiple blood and tissue enzymes collectively called
system was functional. Conversely, it rose to only
angiotensinases.
60 mm Hg when the renin-angiotensin system was
During its persistence in the blood, angiotensin
blocked. This shows that the renin-angiotensin system
II has two principal effects that can elevate arterial
is powerful enough to return the arterial pressure at
pressure. The first of these, vasoconstriction in many
least halfway back to normal within a few minutes
areas of the body, occurs rapidly. Vasoconstriction
after severe hemorrhage. Therefore, sometimes it
occurs intensely in the arterioles and much less so in
can be of lifesaving service to the body, especially in
the veins. Constriction of the arterioles increases the
circulatory shock.
total peripheral resistance, thereby raising the arterial
Note also that the renin-angiotensin vasocon-
pressure, as demonstrated at the bottom of the schema
strictor system requires about 20 minutes to become
in Figure 19-9. Also, the mild constriction of the veins
fully active. Therefore, it is somewhat slower to act
promotes increased venous return of blood to the
for pressure control than are the nervous reflexes
heart, thereby helping the heart pump against the
and the sympathetic norepinephrine-epinephrine
increasing pressure.
system.
The second principal means by which angiotensin
increases the arterial pressure is to decrease excretion
Effect of Angiotensin in the Kidneys to
of both salt and water by the kidneys. This slowly
Cause Renal Retention of Salt and Water—
increases the extracellular fluid volume, which then
An Especially Important Means for
increases the arterial pressure during subsequent
Long-Term Control of Arterial Pressure
hours and days. This long-term effect, acting through
Angiotensin causes the kidneys to retain both salt and
the extracellular fluid volume mechanism, is even
water in two major ways:
more powerful than the acute vasoconstrictor mecha-
1. Angiotensin acts directly on the kidneys to cause
nism in eventually raising the arterial pressure.
salt and water retention.
2. Angiotensin causes the adrenal glands to secrete
Rapidity and Intensity of the
aldosterone, and the aldosterone in turn increases
Vasoconstrictor Pressure Response to
salt and water reabsorption by the kidney tubules.
the Renin-Angiotensin System
Thus, whenever excess amounts of angiotensin cir-
Figure
19-10 shows a typical experiment demon-
culate in the blood, the entire long-term renal-body
strating the effect of hemorrhage on the arterial
fluid mechanism for arterial pressure control auto-
pressure under two separate conditions: (1) with the
matically becomes set to a higher arterial pressure
renin-angiotensin system functioning and (2) without
level than normal.
Chapter 19
The Integrated System for Pressure Control
225
Mechanisms of the Direct Renal Effects of Angiotensin to Cause
Renal Retention of Salt and Water. Angiotensin has several
Angiotensin levels in the blood
direct renal effects that make the kidneys retain salt
(times normal)
and water. One major effect is to constrict the renal
arterioles, thereby diminishing blood flow through
0
2.5
10
the kidneys. As a result, less fluid filters through the
glomeruli into the tubules. Also, the slow flow of blood
reduces the pressure in the peritubular capillaries,
8
which causes rapid reabsorption of fluid from the
tubules. And still a third effect is that angiotensin has
important direct actions on the tubular cells them-
6
selves to increase tubular reabsorption of sodium and
water. The total result of all these effects is significant,
sometimes decreasing urine output less than one fifth
4
Equilibrium
of normal.
points
Stimulation of Aldosterone Secretion by Angiotensin, and the
2
Normal Intake
Effect of Aldosterone in Increasing Salt and Water Retention
by the Kidneys. Angiotensin is also one of the most
0
powerful stimulators of aldosterone secretion by the
0
60
80
100
120
140
160
adrenal glands, as we shall discuss in relation to body
Arterial pressure (mm Hg)
fluid regulation in Chapter
29 and in relation to
adrenal gland function in Chapter 77. Therefore, when
the renin-angiotensin system becomes activated, the
rate of aldosterone secretion usually also increases;
Figure 19-11
and an important subsequent function of aldosterone
Effect of two angiotensin II levels in the blood on the renal output
is to cause marked increase in sodium reabsorption by
curve, showing regulation of the arterial pressure at an equilibrium
the kidney tubules, thus increasing the total body
point of 75 mm Hg when the angiotensin II level is low and at
extracellular fluid sodium. This increased sodium then
115 mm Hg when the angiotensin II level is high.
causes water retention, as already explained, increas-
ing the extracellular fluid volume and leading second-
arily to still more long-term elevation of the arterial
pressure.
angiotensin to cause renal retention of salt and water
Thus both the direct effect of angiotensin on the
can have a powerful effect in promoting chronic eleva-
kidney and its effect acting through aldosterone are
tion of the arterial pressure.
important in long-term arterial pressure control.
However, research in our own laboratory has sug-
Role of the Renin-Angiotensin System in
gested that the direct effect of angiotensin on the
Maintaining a Normal Arterial Pressure
kidneys is perhaps three or more times as potent as the
Despite Wide Variations in Salt Intake
indirect effect acting through aldosterone—even
One of the most important functions of the renin-
though the indirect effect is the one most widely
angiotensin system is to allow a person to eat either
known.
very small or very large amounts of salt without
causing great changes in either extracellular fluid
Quantitative Analysis of Arterial Pressure Changes Caused by
volume or arterial pressure. This function is explained
Angiotensin. Figure 19-11 shows a quantitative analysis
by the schema in Figure 19-12, which shows that the
of the effect of angiotensin in arterial pressure control.
initial effect of increased salt intake is to elevate the
This figure shows two renal output curves as well as a
extracellular fluid volume and this in turn to elevate
line depicting normal level of sodium intake. The left-
the arterial pressure. Then, the increased arterial pres-
hand renal output curve is that measured in dogs whose
sure causes increased blood flow through the kidneys,
renin-angiotensin system had been blocked by the drug
captopril (which blocks the conversion of angiotensin I
which reduces the rate of secretion of renin to a much
to angiotensin II, the active form of angiotensin). The
lower level and leads sequentially to decreased renal
right-hand curve was measured in dogs infused contin-
retention of salt and water, return of the extracellular
uously with angiotensin II at a level about 2.5 times the
fluid volume almost to normal, and, finally, return of
normal rate of angiotensin formation in the blood. Note
the arterial pressure also almost to normal. Thus, the
the shift of the renal output curve toward higher pres-
renin-angiotensin system is an automatic feedback
sure levels under the influence of angiotensin II. This
mechanism that helps maintain the arterial pressure at
shift is caused by both the direct effects of angiotensin
or near the normal level even when salt intake is
on the kidney and the indirect effect acting through
increased. Or, when salt intake is decreased below
aldosterone secretion, as explained above.
normal, exactly opposite effects take place.
Finally, note the two equilibrium points, one for zero
angiotensin showing an arterial pressure level of 75 mm
To emphasize the efficacy of the renin-angiotensin
Hg, and one for elevated angiotensin showing a
system in controlling arterial pressure, when the
pressure level of 115 mm Hg. Therefore, the effect of
system functions normally, the pressure rises no more
226
Unit IV The Circulation
Increased salt intake
Increased extracellular volume
Increased arterial pressure
Decreased renin and angiotensin
Renal artery constricted
Constriction released
Decreased renal retention of salt and water
Systemic arterial
pressure
200
Return of extracellular volume almost to normal
150
Return of arterial pressure almost to normal
Distal renal arterial
100
pressure
Figure 19-12
50
Sequential events by which increased salt intake increases the
arterial pressure, but feedback decrease in activity of the renin
angiotensin system returns the arterial pressure almost to the
normal level.
7
Renin secretion
than 4 to 6 mm Hg in response to as much as a 50-fold
1
0
increase in salt intake. Conversely, when the renin-
0
4
8
12
angiotensin system is blocked, the same increase in salt
Days
intake sometimes causes the pressure to rise 10 times
the normal increase, often as much as 50 to 60 mm Hg.
Figure 19-13
Types of Hypertension in Which
Angiotensin Is Involved: Hypertension
Effect of placing a constricting clamp on the renal artery of one
Caused by a Renin-Secreting Tumor
kidney after the other kidney has been removed. Note the
changes in systemic arterial pressure, renal artery pressure distal
or by Infusion of Angiotensin II
to the clamp, and rate of renin secretion. The resulting hyperten-
sion is called “one-kidney” Goldblatt hypertension.
Occasionally a tumor of the renin-secreting juxta-
glomerular cells (the JG cells) occurs and secretes
tremendous quantities of renin; in turn, equally large
2. By causing the kidneys to retain salt and water;
over a period of days, this, too, causes hypertension
quantities of angiotensin II are formed. In all patients
and is the principal cause of the long-term
in whom this has occurred, severe hypertension has
continuation of the elevated pressure.
developed. Also, when large amounts of angiotensin
are infused continuously for days or weeks into
“One-Kidney” Goldblatt Hypertension. When one kidney is
animals, similar severe long-term hypertension
removed and a constrictor is placed on the renal artery
develops.
of the remaining kidney, as shown in Figure 19-13, the
We have already noted that angiotensin can increase
immediate effect is greatly reduced pressure in the
the arterial pressure in two ways:
renal artery beyond the constrictor, as demonstrated
1. By constricting the arterioles throughout the entire
by the dashed curve in the figure. Then, within seconds
body, thereby increasing the total peripheral
resistance and arterial pressure; this effect occurs
or minutes, the systemic arterial pressure begins to rise
within seconds after one begins to infuse
and continues to rise for several days. The pressure
angiotensin.
usually rises rapidly for the first hour or so, and this is
Chapter 19
The Integrated System for Pressure Control
227
followed by a slower additional rise during the next
Other Types of Hypertension
several days. When the systemic arterial pressure
Caused by Combinations of Volume
reaches its new stable pressure level, the renal arterial
Loading and Vasoconstriction
pressure (the dashed curve in the figure) will have
returned almost all the way back to normal. The hyper-
Hypertension in the Upper Part of the Body Caused by Coarctation
of the Aorta. One out of every few thousand babies is
tension produced in this way is called “one-kidney”
born with pathological constriction or blockage of the
Goldblatt hypertension in honor of Dr. Goldblatt, who
aorta at a point beyond the aortic arterial branches to
first studied the important quantitative features of
the head and arms but proximal to the renal arteries,
hypertension caused by renal artery constriction.
a condition called coarctation of the aorta. When this
The early rise in arterial pressure in Goldblatt
occurs, blood flow to the lower body is carried by mul-
hypertension is caused by the renin-angiotensin vaso-
tiple, small collateral arteries in the body wall, with
constrictor mechanism. That is, because of poor blood
much vascular resistance between the upper aorta and
flow through the kidney after acute constriction of the
the lower aorta. As a consequence, the arterial pressure
renal artery, large quantities of renin are secreted by
in the upper part of the may be 40-50 per cent higher
the kidney, as demonstrated by the lowermost curve in
than that in the lower body.
The mechanism of this upper-body hypertension is
Figure 19-13, and this causes increased angiotensin
almost identical to that of one-kidney Goldblatt hyper-
II and aldosterone in the blood. The angiotensin in
tension. That is, when a constrictor is placed on the aorta
turn raises the arterial pressure acutely. The secretion
above the renal arteries, the blood pressure in both
of renin rises to a peak in an hour or so but returns
kidneys at first falls, renin is secreted, angiotensin and
nearly to normal in 5 to 7 days because the renal arte-
aldosterone are formed, and hypertension occurs in the
rial pressure by that time has also risen back to
upper body. The arterial pressure in the lower body at
normal, so that the kidney is no longer ischemic.
the level of the kidneys rises approximately to normal,
The second rise in arterial pressure is caused by
but high pressure persists in the upper body. The
retention of salt and water by the constricted kidney
kidneys are no longer ischemic, so that secretion of
(that is also stimulated by angiotensin II and aldos-
renin and formation of angiotensin and aldosterone
return to normal. Likewise, in coarctation of the aorta,
terone). In 5 to 7 days, the body fluid volume will have
the arterial pressure in the lower body is usually almost
increased enough to raise the arterial pressure to its
normal, whereas the pressure in the upper body is far
new sustained level. The quantitative value of this sus-
higher than normal.
tained pressure level is determined by the degree of
constriction of the renal artery. That is, the aortic pres-
Role of Autoregulation in the Hypertension Caused by Aortic
sure must rise high enough so that renal arterial pres-
Coarctation. A significant feature of hypertension caused
sure distal to the constrictor is enough to cause normal
by aortic coarctation is that blood flow in the arms,
urine output.
where the pressure may be 40 to 60 per cent above
normal, is almost exactly normal. Also, blood flow in the
“Two-Kidney” Goldblatt Hypertension. Hypertension also
legs, where the pressure is not elevated, is almost exactly
can result when the artery to only one kidney is con-
normal. How could this be, with the pressure in the
stricted while the artery to the other kidney is normal.
upper body 40 to 60 per cent greater than in the lower
This hypertension results from the following mecha-
body? The answer is not that there are differences in
nism: The constricted kidney secretes renin and also
vasoconstrictor substances in the blood of the upper and
retains salt and water because of decreased renal arte-
lower body, because the same blood flows to both areas.
Likewise, the nervous system innervates both areas of
rial pressure in this kidney. Then the “normal” oppo-
the circulation similarly, so that there is no reason to
site kidney retains salt and water because of the renin
believe that there is a difference in nervous control of
produced by the ischemic kidney. This renin causes
the blood vessels. The only reasonable answer is that
formation of angiotension II and aldosterone both of
long-term autoregulation develops so nearly completely
which circulate to the opposite kidney and cause it also
that the local blood flow control mechanisms have com-
to retain salt and water. Thus, both kidneys, but for dif-
pensated almost 100 per cent for the differences in pres-
ferent reasons, become salt and water retainers. Con-
sure. The result is that, in both the high-pressure area
sequently, hypertension develops.
and the low-pressure area, the local blood flow is con-
trolled almost exactly in accord with the needs of the
Hypertension Caused by Diseased Kidneys That Secrete Renin
tissue and not in accord with the level of the pressure.
One of the reasons these observations are so important
Chronically. Often, patchy areas of one or both kidneys
is that they demonstrate how nearly complete the long-
are diseased and become ischemic because of local
term autoregulation process can be.
vascular constrictions, whereas other areas of the
kidneys are normal. When this occurs, almost identical
Hypertension in Preeclampsia (Toxemia of Pregnancy). Approx-
effects occur as in the two-kidney type of Goldblatt
imately 5 to 10 per cent of expectant mothers develop
hypertension. That is, the patchy ischemic kidney
a syndrome called preeclampsia (also called toxemia of
tissue secretes renin, and this in turn, acting through
pregnancy). One of the manifestations of preeclampsia
the formation of angiotensin II, causes the remaining
is hypertension that usually subsides after delivery of
kidney mass also to retain salt and water. Indeed, one
the baby. Although the precise causes of preeclampsia
of the most common causes of renal hypertension,
are not completely understood, ischemia of the placenta
especially in older persons, is such patchy ischemic
and subsequent release by the placenta of toxic factors
kidney disease.
are believed to play a role in causing many of the man-
228
Unit IV
The Circulation
ifestations of this disorder, including hypertension in the
hypertension. In other strains of hypertensive rats,
mother. Substances released by the ischemic placenta,
impaired renal function also has been observed.
in turn, cause dysfunction of vascular endothelial cells
throughout the body, including the blood vessels of the
kidneys. This endothelial dysfunction decreases release of
“Primary (Essential) Hypertension”
nitric oxide and other vasodilator substances, causing
vasoconstriction, decreased rate of fluid filtration from
About 90 to 95 per cent of all people who have hyper-
the glomeruli into the renal tubules, impaired renal-
pressure natriuresis, and development of hypertension.
tension are said to have “primary hypertension,” also
Another pathological abnormality that may con-
widely known as “essential hypertension” by many cli-
tribute to hypertension in preeclampsia is thickening of
nicians. These terms mean simply that the hypertension
the kidney glomerular membranes (perhaps caused by
is of unknown origin, in contrast to those forms of
an autoimmune process), which also reduces the rate of
hypertension that are secondary to known causes, such
glomerular fluid filtration. For obvious reasons, the arte-
as renal artery stenosis. In some patients with primary
rial pressure level required to cause normal formation
hypertension, there is a strong hereditary tendency, the
of urine becomes elevated, and the long-term level of
same as occurs in animal strains of genetic hyperten-
arterial pressure becomes correspondingly elevated.
sion discussed above.
These patients are especially prone to extra degrees of
In most patients, excess weight gain and sedentary
hypertension when they have excess salt intake.
lifestyle appear to play a major role in causing hyper-
Neurogenic Hypertension. Acute neurogenic hypertension
tension. The majority of patients with hypertension
can be caused by strong stimulation of the sympathetic
are overweight, and studies of different populations
nervous system. For instance, when a person becomes
suggest that excess weight gain and obesity may
excited for any reason or at times during states of
account for as much as 65 to 70 percent of the risk
anxiety, the sympathetic system becomes excessively
for developing primary hypertension. Clinical studies
stimulated, peripheral vasoconstriction occurs every-
have clearly shown the value of weight loss for reduc-
where in the body, and acute hypertension ensues.
ing blood pressure in most patients with hypertension.
In fact, new clinical guidelines for treating hyper-
Acute Neurogenic Hypertension Caused by Sectioning the Barore-
tension recommend increased physical activity and
ceptor Nerves. Another type of acute neurogenic hyper-
weight loss as a first step in treating most patients with
tension occurs when the nerves leading from the
hypertension.
baroreceptors are cut or when the tractus solitarius
is destroyed in each side of the medulla oblongata
Some of the characteristics of primary hypertension
(these are the areas where the nerves from the carotid
caused by excess weight gain and obesity include:
and aortic baroreceptors connect in the brain stem).
1.
Cardiac output is increased due, in part, to the
The sudden cessation of normal nerve signals from
additional blood flow required for the extra
the baroreceptors has the same effect on the nervous
adipose tissue. However, blood flow in the heart,
pressure control mechanisms as a sudden reduction
kidneys, gastrointestinal tract, and skeletal muscle
of the arterial pressure in the aorta and carotid arteries.
also increases with weight gain due to increased
That is, loss of the normal inhibitory effect on the
metabolic rate and growth of the organs and tissues
vasomotor center caused by normal baroreceptor
in response to their increased metabolic demands.
nervous signals allows the vasomotor center suddenly
As the hypertension is sustained for many months
to become extremely active and the mean arterial pres-
and years, total peripheral vascular resistance may
sure to increase from 100 mm Hg to as high as 160 mm
be increased.
Hg. The pressure returns to nearly normal within about
2.
Sympathetic nerve activity, especially in the kidneys,
2 days because the response of the vasomotor center to
is increased in overweight patients. The causes of
the absent baroreceptor signal fades away, which is
increased sympathetic activity in obesity are not
called central “resetting” of the baroreceptor pressure
fully understood, but recent studies suggest that
control mechanism. Therefore, the neurogenic hyper-
hormones, such as leptin, released from fat cells
tension caused by sectioning the baroreceptor nerves is
may directly stimulate multiple regions of the
mainly an acute type of hypertension, not a chronic
hypothalamus, which, in turn, have an excitatory
type.
influence on the vasomotor centers of the brain
medulla.
Spontaneous Hereditary Hypertension in Lower Animals. Spon-
3.
Angiotensin II and aldosterone levels are increased
taneous hereditary hypertension has been observed in
two- to threefold in many obese patients. This may
a number of strains of lower animals, including several
be caused partly by increased sympathetic nerve
different strains of rats, at least one strain of rabbits, and
stimulation, which increases renin release by the
at least one strain of dogs. In the strain of rats that has
kidneys and therefore formation of angiotensin II,
been studied to the greatest extent, the Okamoto strain,
which, in turn, stimulates the adrenal gland to
there is evidence that in early development of the
secrete aldosterone.
hypertension, the sympathetic nervous system is con-
4.
The renal-pressure natriuresis mechanism is
siderably more active than in normal rats. However, in
impaired, and the kidneys will not excrete adequate
the late stages of this type of hypertension, two struc-
amounts of salt and water unless the arterial
tural changes have been observed in the nephrons of the
pressure is high or unless kidney function is
kidneys:
(1) increased preglomerular renal arterial
somehow improved. In other words, if the mean
resistance and
(2) decreased permeability of the
arterial pressure in the essential hypertensive
glomerular membranes. These structural changes could
person is 150 mm Hg, acute reduction of the mean
easily be the basis for the long-term continuance of the
arterial pressure artificially to the normal value of
Chapter 19
The Integrated System for Pressure Control
229
100 mm Hg (but without otherwise altering renal
increasing the level of sodium intake. The sodium-
function except for the decreased pressure) will
loading type of curve can be determined by increasing
cause almost total anuria, and the person will retain
the level of sodium intake to a new level every few
salt and water until the pressure rises back to the
days, then waiting for the renal output of sodium to
elevated value of 150 mm Hg. Chronic reductions
come into balance with the intake, and at the same
in arterial pressure with effective antihypertensive
time recording the changes in arterial pressure.
therapies, however, usually do not cause marked
When this procedure is used in essential hyperten-
salt and water retention by the kidneys because
sive patients, two types of curves, shown to the right in
these therapies also improve renal-pressure
natriuresis, as discussed below.
Figure 19-14, can be recorded in essential hyperten-
Experimental studies in obese animals and obese
sive patients, one called (1) nonsalt-sensitive hyperten-
patients suggest that impaired renal-pressure natri-
sion and the other (2) salt-sensitive hypertension. Note
uresis in obesity hypertension is caused mainly by
in both instances that the curves are shifted to the
increased renal tubular reabsorption of salt and
right, to a much higher pressure level than for normal
water due to increased sympathetic nerve activity and
people. Now, let us plot on this same graph (1) a
increased levels of angiotensin II and aldosterone.
normal level of salt intake and (2) a high level of salt
However, if hypertension is not effectively treated,
intake representing 3.5 times the normal intake. In
there may also be vascular damage in the kidneys that
the case of the person with nonsalt-sensitive essential
can reduce the glomerular filtration rate and increase
hypertension, the arterial pressure does not increase
the severity of the hypertension. Eventually uncon-
significantly when changing from normal salt intake
trolled hypertension associated with obesity can lead
to high salt intake. Conversely, in those patients who
to severe vascular injury and complete loss of kidney
have salt-sensitive essential hypertension, the high salt
function.
intake significantly exacerbates the hypertension.
Two additional points should be emphasized: (1)
Graphical Analysis of Arterial Pressure Control in Essential
Salt-sensitivity of blood pressure is not an all-or-none
Hypertension. Figure 19-14 is a graphical analysis of
characteristic—it is a quantitative characteristic, with
essential hypertension. The curves of this figure are
some individuals being more salt-sensitive than others.
called sodium-loading renal function curves because
(2) Salt-sensitivity of blood pressure is not a fixed
the arterial pressure in each instance is increased
characteristic; instead, blood pressure usually becomes
very slowly, over many days or weeks, by gradually
more salt-sensitive as a person ages, especially after 50
or 60 years of age.
The reason for the difference between nonsalt-
sensitive essential hypertension and salt-sensitive
hypertension is presumably related to structural or
functional differences in the kidneys of these two types
of hypertensive patients. For example, salt-sensitive
Normal
hypertension may occur with different types of chronic
Nonsalt-sensitive
renal disease due to gradual loss of the functional units
Salt-sensitive
of the kidneys (the nephrons) or to normal aging as
discussed in Chapter 31. Abnormal function of the
6
renin-angiotensin system can also cause blood pres-
5
sure to become salt-sensitive, as discussed previously
in this chapter.
4
High intake
E
B
B1
Treatment of Essential Hypertension. Current guidelines
3
for treating hypertension recommend, as a first step,
Essential
Normal
lifestyle modifications that are aimed at increasing
2
hypertension
physical activity and weight loss in most patients.
Normal intake
D
A
Unfortunately, many patients are unable to lose
1
weight, and pharmacological treatment with antihy-
C
pertensive drugs must be initiated.
0
0
50
100
150
Two general classes of drugs are used to treat hyper-
tension: (1) vasodilator drugs that increase renal blood
Arterial pressure (mm Hg)
flow and (2) natriuretic or diuretic drugs that decrease
tubular reabsorption of salt and water.
Vasodilator drugs usually cause vasodilation in
many other tissues of the body as well as in the
Figure 19-14
kidneys. Different ones act in one of the following
ways: (1) by inhibiting sympathetic nervous signals to
Analysis of arterial pressure regulation in (1) nonsalt-sensitive
essential hypertension and (2) salt-sensitive essential hyperten-
the kidneys or by blocking the action of the sympa-
sion. (Redrawn from Guyton AC, Coleman TG, Young DB, et al:
thetic transmitter substance on the renal vasculature,
Salt balance and long-term blood pressure control. Annu Rev Med
31:15, 1980. With permission, from the Annual Review of Medi-
(2) by directly relaxing the smooth muscle of the renal
vasculature, or
(3) by blocking the action of the
230
Unit IV The Circulation
renin-angiotensin system on the renal vasculature or
renal tubules.
Renin-angiotensin-vasoconstriction
Those drugs that reduce reabsorption of salt and
water by the renal tubules include especially drugs that
•
• !!
block active transport of sodium through the tubular
wall; this blockage in turn also prevents the reabsorp-
11
tion of water, as explained earlier in the chapter. These
10
natriuretic or diuretic drugs are discussed in greater
9
detail in Chapter 31.
8
Baroreceptors
7
6
Summary of the Integrated,
5
Multifaceted System for
4
n
3
Arterial Pressure Regulation
2
1
By now, it is clear that arterial pressure is regulated
0
not by a single pressure controlling system but instead
0 1530 1 2 4 816321 2 4 816 1 2 4
8 16
•
by several interrelated systems, each of which per-
forms a specific function. For instance, when a person
Seconds Minutes
Hours
Days
bleeds severely so that the pressure falls suddenly, two
Time after sudden change in pressure
problems confront the pressure control system. The
first is survival, that is, to return the arterial pressure
immediately to a high enough level that the person can
live through the acute episode. The second is to return
Figure 19-15
the blood volume eventually to its normal level so that
Approximate potency of various arterial pressure control mecha-
the circulatory system can re-establish full normality,
nisms at different time intervals after onset of a disturbance to the
including return of the arterial pressure all the way
arterial pressure. Note especially the infinite gain (∞) of the renal
back to its normal value, not merely back to a pressure
body fluid pressure control mechanism that occurs after a few
weeks’ time. (Redrawn from Guyton AC: Arterial Pressure and
level required for survival.
Hypertension. Philadelphia: WB Saunders Co, 1980.)
In Chapter 18, we saw that the first line of de-
fense against acute changes in arterial pressure is
the nervous control system. In this chapter, we have
emphasized a second line of defense achieved mainly
(2) to cause increased heart rate and contractility of
by kidney mechanisms for long-term control of arte-
the heart to provide greater pumping capacity by the
rial pressure. However, there are other pieces to the
heart, and (3) to cause constriction of most peripheral
puzzle. Figure 19-15 helps to put these together.
arterioles to impede flow of blood out of the arteries;
Figure
19-15 shows the approximate immediate
all these effects occur almost instantly to raise the
(seconds and minutes) and long-term (hours and days)
arterial pressure back into a survival range.
control responses, expressed as feedback gain, of eight
When the pressure suddenly rises too high, as might
arterial pressure control mechanism. These mecha-
occur in response to rapid overadministration of a
nisms can be divided into three groups: (1) those that
blood transfusion, the same control mechanisms
react rapidly, within seconds or minutes; (2) those that
operate in the reverse direction, again returning the
respond over an intermediate time period, minutes or
pressure back toward normal.
hours; and (3) those that provide long-term arterial
pressure regulation, days, months, and years. Let us see
Pressure Control Mechanisms That Act After Many Minutes.
how they fit together as a total, integrated system for
Several pressure control mechanisms exhibit signifi-
pressure control.
cant responses only after a few minutes following
acute arterial pressure change. Three of these, shown
Rapidly Acting Pressure Control Mechanisms, Acting Within
in Figure 19-15, are (1) the renin-angiotensin vaso-
Seconds or Minutes. The rapidly acting pressure control
constrictor mechanism, (2) stress-relaxation of the
mechanisms are almost entirely acute nervous reflexes
vasculature, and (3) shift of fluid through the tissue
or other nervous responses. Note in Figure 19-15 the
capillary walls in and out of the circulation to readjust
three mechanisms that show responses within seconds.
the blood volume as needed.
They are (1) the baroreceptor feedback mechanism,
We have already described at length the role of the
(2) the central nervous system ischemic mechanism,
renin-angiotensin vasoconstrictor system to provide a
and (3) the chemoreceptor mechanism. Not only do
semi-acute means for increasing the arterial pressure
these mechanisms begin to react within seconds, but
when this is needed. The stress-relaxation mechanism
they are also powerful. After any acute fall in pressure,
is demonstrated by the following example: When the
as might be caused by severe hemorrhage, the nervous
pressure in the blood vessels becomes too high, they
mechanisms combine (1) to cause constriction of the
become stretched and keep on stretching more and
veins and provide transfer of blood into the heart,
more for minutes or hours; as a result, the pressure in
Chapter 19
The Integrated System for Pressure Control
231
the vessels falls toward normal. This continuing stretch
pressure controls, then continues with the sustaining
of the vessels, called stress-relaxation, can serve as an
characteristics of the intermediate pressure controls,
intermediate-term pressure “buffer.”
and, finally, is stabilized at the long-term pressure level
The capillary fluid shift mechanism means simply
by the renal-body fluid mechanism. This long-term
that any time capillary pressure falls too low, fluid is
mechanism in turn has multiple interactions with the
absorbed through the capillary membranes from the
renin-angiotensin-aldosterone system, the nervous
tissues into the circulation, thus building up the blood
system, and several other factors that provide special
volume and increasing the pressure in the circulation.
blood pressure control capabilities for special pur-
Conversely, when the capillary pressure rises too high,
poses.
fluid is lost out of the circulation into the tissues, thus
reducing the blood volume as well as virtually all the
pressures throughout the circulation.
References
These three intermediate mechanisms become
mostly activated within 30 minutes to several hours.
Chobanian AV, Bakris GL, Black HR, et al: Joint National
During this time, the nervous mechanisms usually
Committee on Prevention, Detection, Evaluation, and
Treatment of High Blood Pressure. National High Blood
become less and less effective, which explains the
Pressure Education Program Coordinating Committee.
importance of these non-nervous, intermediate time
Seventh Report of the Joint National Committee on
pressure control measures.
prevention, detection, evaluation, and treatment of high
blood pressure. Hypertension 42:1206, 2003.
Long-Term Mechanisms for Arterial Pressure Regulation. The
Cowley AW J: Long-term control of arterial blood pressure.
goal of this chapter has been to explain the role of the
Physiol Rev 72:231, 1992.
kidneys in long-term control of arterial pressure. To
Granger JP, Alexander BT, Bennett WA, Khalil RA: Patho-
the far right in Figure 19-15 is shown the renal-blood
physiology of pregnancy-induced hypertension. Am J
volume pressure control mechanism (which is the
Hypertens 14:178S, 2001.
same as the renal-body fluid pressure control mecha-
Granger JP, Alexander BT: Abnormal pressure-natriuresis in
hypertension: role of nitric oxide. Acta Physiol Scand
nism), demonstrating that it takes a few hours to begin
168:161, 2000.
showing significant response. Yet it eventually devel-
Guyton AC: Arterial Pressure and Hypertension. Philadel-
ops a feedback gain for control of arterial pressure
phia: WB Saunders Co, 1980.
equal to infinity. This means that this mechanism can
Guyton AC: Blood pressure control—special role of the
eventually return the arterial pressure all the way back,
kidneys and body fluids. Science 252:1813, 1991.
not merely partway back, to that pressure level that
Guyton AC, Coleman TG, Cowley AW Jr, et al: Arterial pres-
provides normal output of salt and water by the
sure regulation: overriding dominance of the kidneys in
kidneys. By now, the reader should be familiar with
long-term regulation and in hypertension. Am J Med
this concept, which has been the major point of this
52:584, 1972.
chapter.
Hall JE: The kidney, hypertension, and obesity. Hypertension
41:625, 2003.
It must also be remembered that many factors can
Hall JE, Brands MW, Henegar JR: Angiotensin II and long-
affect the pressure-regulating level of the renal-body
term arterial pressure regulation: the overriding domi-
fluid mechanism. One of these, shown in Figure 19-15,
nance of the kidney. J Am Soc Nephrol 10(Suppl 12):S258,
is aldosterone. A decrease in arterial pressure leads
1999.
within minutes to an increase in aldosterone secretion,
Hall JE, Guyton AC, Brands MW: Pressure-volume regula-
and over the next hour or days, this plays an important
tion in hypertension. Kidney Int Suppl 55:S35, 1996.
role in modifying the pressure control characteristics
Hall JE, Jones DW, Kuo JJ, et al: Impact of the obesity epi-
of the renal-body fluid mechanism. Especially impor-
demic on hypertension and renal disease. Curr Hypertens
tant is interaction of the renin-angiotensin system with
Rep 5:386, 2003.
the aldosterone and renal fluid mechanisms. For
Lifton RP, Gharavi AG, Geller DS: Molecular mechanisms
of human hypertension. Cell 104:545, 2001.
instance, a person’s salt intake varies tremendously
Manning RD Jr, Hu L, Tan DY, Meng S: Role of abnormal
from one day to another. We have seen in this chapter
nitric oxide systems in salt-sensitive hypertension. Am J
that the salt intake can decrease to as little as 1/10
Hypertens 14:68S, 2001.
normal or can increase to 10 to 15 times normal and
Oparil S, Zaman MA, Calhoun DA: Pathogenesis of hyper-
yet the regulated level of the mean arterial pressure
tension. Ann Intern Med 139:761, 2003.
will change only a few millimeters of mercury if the
O’Shaughnessy KM, Karet FE: Salt handling and hyperten-
renin-angiotensin-aldosterone system is fully opera-
sion. J Clin Invest 113:1075, 2004.
tive. But, without a functional renin-angiotensin-aldos-
Reckelhoff JF: Gender differences in the regulation of blood
terone system, blood pressure becomes very sensitive
pressure. Hypertension 37:1199, 2001.
to changes in salt intake. Thus, arterial pressure control
Rossier BC: Negative regulators of sodium transport in the
kidney: key factors in understanding salt-sensitive hyper-
begins with the lifesaving measures of the nervous
tension? J Clin Invest 111:947, 2003.
C H A P T E R
2
0
Cardiac Output, Venous Return,
and Their Regulation
Cardiac output is the quantity of blood pumped into
the aorta each minute by the heart. This is
also the quantity of blood that flows through the
circulation. Cardiac output is perhaps the most
important factor that we have to consider in rela-
tion to the circulation.
Venous return is the quantity of blood flowing
from the veins into the right atrium each minute.
The venous return and the cardiac output must equal each other except for a
few heartbeats at a time when blood is temporarily stored in or removed from
the heart and lungs.
Normal Values for Cardiac Output at Rest and
During Activity
Cardiac output varies widely with the level of activity of the body. The follow-
ing factors, among others, directly affect cardiac output: (1) the basic level of
body metabolism, (2) whether the person is exercising, (3) the person’s age, and
(4) size of the body.
For young, healthy men, resting cardiac output averages about 5.6 L/min.
For women, this value is about 4.9 L/min. When one considers the factor of age
as well—because with increasing age, body activity diminishes—the average
cardiac output for the resting adult, in round numbers, is often stated to be
almost exactly 5 L/min.
Cardiac Index
Experiments have shown that the cardiac output increases approximately in pro-
portion to the surface area of the body. Therefore, cardiac output is frequently stated
in terms of the cardiac index, which is the cardiac output per square meter of body
surface area. The normal human being weighing 70 kilograms has a body surface
area of about 1.7 square meters, which means that the normal average cardiac index
for adults is about 3 L/min/m2 of body surface area.
Effect of Age on Cardiac Output. Figure 20-1 shows the cardiac output, expressed
as cardiac index, at different ages. Rising rapidly to a level greater than
4 L/min/m2 at age 10 years, the cardiac index declines to about 2.4 L/min/m2 at
age 80 years. We will see later in the chapter that the cardiac output is regu-
lated throughout life almost directly in proportion to the overall bodily meta-
bolic activity. Therefore, the declining cardiac index is indicative of declining
activity with age.
Control of Cardiac Output by Venous
Return—Role of the Frank-Starling
Mechanism of the Heart
When one states that cardiac output is controlled by venous return, this means
that it is not the heart itself that is the primary controller of cardiac output.
232
Chapter 20
Cardiac Output, Venous Return, and Their Regulation
233
Cardiac output
35
and cardiac index
4
4
Oxygen
30
15
consumption
4
25
20
3
3
3
10
15
2
Dexter
1951
5
10
Douglas
1922
2
2
1
5
Christensen
1931
Donald
1055
0
0
0
0
200 400 600 800 1000 1200 14001600
1
1
Work output during exercise (kg-m/min)
0
0
Figure 20-2
0
10
20
30
40
50
60
70
80
Age in years
Effect of increasing levels of exercise to increase cardiac output
(red solid line) and oxygen consumption
(blue dashed line).
(Redrawn from Guyton AC, Jones CE, Coleman TB: Circulatory
Physiology: Cardiac Output and Its Regulation. 2nd ed. Philadel-
Figure 20-1
phia: WB Saunders Co, 1973.)
Cardiac index for the human being (cardiac output per square
meter of surface area) at different ages. (Redrawn from Guyton
Under most normal unstressful conditions, the
AC, Jones CE, Coleman TB: Circulatory Physiology: Cardiac
cardiac output is controlled almost entirely by periph-
Output and Its Regulation. 2nd ed. Philadelphia: WB Saunders Co,
1973.)
eral factors that determine venous return. However,
we shall see later in the chapter that if the returning
blood does become more than the heart can pump,
then the heart becomes the limiting factor that deter-
mines cardiac output.
Instead, it is the various factors of the peripheral cir-
culation that affect flow of blood into the heart from
the veins, called venous return, that are the primary
Cardiac Output Regulation Is the Sum
controllers.
of Blood Flow Regulation in All the
The main reason peripheral factors are usually more
Local Tissues of the Body—Tissue
important than the heart itself in controlling cardiac
output is that the heart has a built-in mechanism that
Metabolism Regulates Most Local
normally allows it to pump automatically whatever
Blood Flow
amount of blood that flows into the right atrium from
the veins. This mechanism, called the Frank-Starling
The venous return to the heart is the sum of all the
law of the heart, was discussed in Chapter 9. Basically,
local blood flows through all the individual tissue
this law states that when increased quantities of blood
segments of the peripheral circulation. Therefore, it
flow into the heart, the increased blood stretches the
follows that cardiac output regulation is the sum of all
walls of the heart chambers. As a result of the stretch,
the local blood flow regulations.
the cardiac muscle contracts with increased force, and
The mechanisms of local blood flow regulation were
this empties the extra blood that has entered from the
discussed in Chapter 17. In most tissues, blood flow
systemic circulation. Therefore, the blood that flows
increases mainly in proportion to each tissue’s metab-
into the heart is automatically pumped without delay
olism. For instance, local blood flow almost always
into the aorta and flows again through the circulation.
increases when tissue oxygen consumption increases;
Another important factor, discussed in Chapter 10,
this effect is demonstrated in Figure 20-2 for different
is that stretching the heart causes the heart to pump
levels of exercise. Note that at each increasing level of
faster—at an increased heart rate. That is, stretch of the
work output during exercise, the oxygen consumption
sinus node in the wall of the right atrium has a direct
and the cardiac output increase in parallel to each
effect on the rhythmicity of the node itself to increase
other.
heart rate as much as 10 to 15 per cent. In addition,
To summarize, cardiac output is determined by the
the stretched right atrium initiates a nervous reflex
sum of all the various factors throughout the body that
called the Bainbridge reflex, passing first to the vaso-
control local blood flow. All the local blood flows
motor center of the brain and then back to the heart
summate to form the venous return, and the heart
by way of the sympathetic nerves and vagi, also to
automatically pumps this returning blood back into
increase the heart rate.
the arteries to flow around the system again.
234
Unit IV The Circulation
25
200
20
Hypereffective
150
15
Normal
100
10
Hypoeffective
50
5
0
40
60
80
100
120
140
160
0
Total peripheral resistance
-4
0
+4
+8
(percentage of normal)
Right atrial pressure (mm Hg)
Figure 20-3
Figure 20-4
Chronic effect of different levels of total peripheral resistance on
cardiac output, showing a reciprocal relationship between total
Cardiac output curves for the normal heart and for hypoeffective
peripheral resistance and cardiac output. (Redrawn from Guyton
and hypereffective hearts. (Redrawn from Guyton AC, Jones CE,
AC: Arterial Pressure and Hypertension. Philadelphia: WB
Coleman TB: Circulatory Physiology: Cardiac Output and Its
Saunders Co, 1980.)
Regulation. 2nd ed. Philadelphia: WB Saunders Co, 1973.)
Effect of Total Peripheral Resistance on the Long-Term Cardiac
Output Level. Figure 20-3 is the same as Figure 19-5.
Figure 20-4 demonstrates the normal cardiac output
It is repeated here to illustrate an extremely important
curve, showing the cardiac output per minute at each
principle in cardiac output control: Under most
level of right atrial pressure. This is one type of cardiac
normal conditions, the long-term cardiac output level
function curve, which was discussed in Chapter 9. Note
varies reciprocally with changes in total peripheral
that the plateau level of this normal cardiac output
resistance. Note in Figure 20-3 that when the total
curve is about 13 L/min, 2.5 times the normal cardiac
peripheral resistance is exactly normal (at the 100 per
output of about 5 L/min. This means that the normal
cent mark in the figure), the cardiac output is also
human heart, functioning without any special stimula-
normal. Then, when the total peripheral resistance
tion, can pump an amount of venous return up to
increases above normal, the cardiac output falls; con-
about 2.5 times the normal venous return before the
versely, when the total peripheral resistance decreases,
heart becomes a limiting factor in the control of
the cardiac output increases. One can easily under-
cardiac output.
stand this by reconsidering one of the forms of Ohm’s
Shown in Figure 20-4 are several other cardiac
law, as expressed in Chapter 14:
output curves for hearts that are not pumping nor-
Arterial Pressure
mally. The uppermost curves are for hypereffective
Cardiac Output
=
Total Peripheral Resistance
hearts that are pumping better than normal. The low-
ermost curves are for hypoeffective hearts that are
The meaning of this formula, and of Figure 20-3, is
pumping at levels below normal.
simply the following: Any time the long-term level of
total peripheral resistance changes (but no other func-
Factors That Can Cause Hypereffective Heart
tions of the circulation change), the cardiac output
Only two types of factors usually can make the heart
changes quantitatively in exactly the opposite direction.
a better pump than normal. They are (1) nervous stim-
ulation and (2) hypertrophy of the heart muscle.
The Heart Has Limits for the Cardiac
Effect of Nervous Excitation to Increase Heart Pumping. In
Output That It Can Achieve
Chapter 9, we saw that a combination of (1) sympa-
thetic stimulation and (2) parasympathetic inhibition
There are definite limits to the amount of blood that
does two things to increase the pumping effectiveness
the heart can pump, which can be expressed quantita-
of the heart: (1) it greatly increases the heart rate—
tively in the form of cardiac output curves.
sometimes, in young people, from the normal level of
Chapter 20
Cardiac Output, Venous Return, and Their Regulation
235
72 beats/min up to 180 to 200 beats/min—and (2) it
increases the strength of heart contraction (which is
called increased “contractility”) to twice its normal
6
Dinitrophenol
strength. Combining these two effects, maximal
5
nervous excitation of the heart can raise the plateau
4
level of the cardiac output curve to almost twice the
3
plateau of the normal curve, as shown by the 25-liter
2
level of the uppermost curve in Figure 20-4.
0
Increased Pumping Effectiveness Caused by Heart Hypertrophy.
With pressure control
A long-term increased workload, but not so much
Without pressure control
excess load that it damages the heart, causes the heart
muscle to increase in mass and contractile strength in
100
the same way that heavy exercise causes skeletal
75
muscles to hypertrophy. For instance, it is common for
50
the hearts of marathon runners to be increased in mass
by 50 to 75 per cent. This increases the plateau level of
0
0
10
20
30
the cardiac output curve, sometimes 60 to 100 per cent,
and therefore allows the heart to pump much greater
Minutes
than usual amounts of cardiac output.
When one combines nervous excitation of the heart
and hypertrophy, as occurs in marathon runners, the
total effect can allow the heart to pump as much 30 to
Figure 20-5
40 L/min, about 21/2 times normal; this increased level
Experiment in a dog to demonstrate the importance of nervous
of pumping is one of the most important factors in
maintenance of the arterial pressure as a prerequisite for cardiac
determining the runner’s running time.
output control. Note that with pressure control, the metabolic stim-
ulant dinitrophenol increases cardiac output greatly; without pres-
sure control, the arterial pressure falls and the cardiac output rises
Factors That Cause a Hypoeffective Heart
very little. (Drawn from experiments by Dr. M. Banet.)
Any factor that decreases the heart’s ability to pump
blood causes hypoeffectivity. Some of the factors that
can do this are the following:
However, after autonomic control of the nervous
Coronary artery blockage, causing a “heart
system had been blocked, none of the normal circula-
attack”
tory reflexes for maintaining the arterial pressure
Inhibition of nervous excitation of the heart
could function, and vasodilation of the vessels with
Pathological factors that cause abnormal heart
dinitrophenol (dashed curves) then caused a profound
rhythm or rate of heartbeat
fall in arterial pressure to about one half normal, and
Valvular heart disease
the cardiac output rose only 1.6-fold instead of 4-fold.
Increased arterial pressure against which the
Thus, maintenance of a normal arterial pressure by
heart must pump, such as in hypertension
the nervous reflexes, by mechanisms explained in
Congenital heart disease
Chapter 18, is essential to achieve high cardiac outputs
Myocarditis
when the peripheral tissues dilate their vessels to
Cardiac hypoxia
increase the venous return.
Effect of the Nervous System to Increase the Arterial Pressure
What Is the Role of the
During Exercise. During exercise, intense increase in
Nervous System in Controlling
metabolism in active skeletal muscles acts directly on
Cardiac Output?
the muscle arterioles to relax them and to allow ade-
quate oxygen and other nutrients needed to sustain
Importance of the Nervous System in
muscle contraction. Obviously, this greatly decreases
Maintaining the Arterial Pressure When the
the total peripheral resistance, which normally would
Venous Return and Cardiac Output Increase
decrease the arterial pressure also. However, the
Figure 20-5 shows an important difference in cardiac
nervous system immediately compensates. The same
output control with and without a functioning auto-
brain activity that sends motor signals to the muscles
nomic nervous system. The solid curves demonstrate
sends simultaneous signals into the autonomic nervous
the effect in the normal dog of intense dilation of
centers of the brain to excite circulatory activity,
the peripheral blood vessels caused by administering
causing large vein constriction, increased heart rate,
the drug dinitrophenol, which increased the metabo-
and increased contractility of the heart. All these
lism of virtually all tissues of the body about fourfold.
changes acting together increase the arterial pressure
Note that with nervous control to keep the arterial
above normal, which in turn forces still more blood
pressure from falling, dilating all the peripheral blood
flow through the active muscles.
vessels caused almost no change in arterial pressure
In summary, when local tissue vessels dilate and
but increased the cardiac output almost fourfold.
thereby increase venous return and cardiac output
236
Unit IV The Circulation
above normal, the nervous system plays an exceed-
and at the same time increase the cardiac output to
ingly important role in preventing the arterial pressure
above normal.
from falling to disastrously low levels. In fact, during
1.
Beriberi. This disease is caused by insufficient
quantity of the vitamin thiamine (vitamin B1) in the
exercise, the nervous system goes even further, pro-
diet. Lack of this vitamin causes diminished ability
viding additional signals to raise the arterial pressure
of the tissues to use some cellular nutrients, and the
even above normal, which serves to increase the
local tissue blood flow mechanisms in turn cause
cardiac output an extra 30 to 100 per cent.
marked compensatory peripheral vasodilation.
Sometimes the total peripheral resistance decreases
to as little as one-half normal. Consequently, the
Pathologically High and
long-term levels of venous return and cardiac
Pathologically Low
output also often increase to twice normal.
2.
Arteriovenous fistula (shunt). Earlier, we pointed
Cardiac Outputs
out that whenever a fistula (also called an AV
In healthy human beings, the cardiac outputs are
shunt) occurs between a major artery and a major
surprisingly constant from one person to another.
vein, tremendous amounts of blood flow directly
However, multiple clinical abnormalities can cause
from the artery into the vein. This, too, greatly
either high or low cardiac outputs. Some of the more
decreases the total peripheral resistance and,
important of these are shown in Figure 20-6.
likewise, increases the venous return and cardiac
output.
3.
Hyperthyroidism. In hyperthyroidism, the
High Cardiac Output Caused
metabolism of most tissues of the body becomes
by Reduced Total Peripheral
greatly increased. Oxygen usage increases, and
Resistance
vasodilator products are released from the tissues.
Therefore, the total peripheral resistance decreases
The left side of Figure 20-6 identifies conditions that
markedly because of the local tissue blood
commonly cause cardiac outputs higher than normal.
flow control reactions throughout the body;
One of the distinguishing features of these conditions is
consequently, the venous return and cardiac output
that they all result from chronically reduced total periph-
often increase to 40 to 80 per cent above normal.
eral resistance. None of them result from excessive
4.
Anemia. In anemia, two peripheral effects greatly
excitation of the heart itself, which we will explain sub-
decrease the total peripheral resistance. One of
sequently. For the present, let us look at some of the
these is reduced viscosity of the blood, resulting
conditions that can decrease the peripheral resistance
from the decreased concentration of red blood
7
200
175
6
150
5
125
4
Control (young adults)
100
Average 45-year-old adult
3
75
2
50
1
25
0
0
Figure 20-6
Cardiac output in different pathological conditions. The numbers in parentheses indicate number of patients studied in each condition.
(Redrawn from Guyton AC, Jones CE, Coleman TB: Circulatory Physiology: Cardiac Output and Its Regulation. 2nd ed. Philadelphia:
WB Saunders Co, 1973.)
Chapter 20
Cardiac Output, Venous Return, and Their Regulation
237
cells. The other is diminished delivery of oxygen to
blood flow needs of the muscles, resulting in
the tissues, which causes local vasodilation. As a
decreases in skeletal muscle blood flow and cardiac
consequence, the cardiac output increases greatly.
output.
Any other factor that decreases the total peripheral
Regardless of the cause of low cardiac output,
resistance chronically also increases the cardiac output.
whether it be a peripheral factor or a cardiac factor, if
ever the cardiac output falls below that level required
for adequate nutrition of the tissues, the person is said
Low Cardiac Output
to suffer circulatory shock. This condition can be lethal
within a few minutes to a few hours. Circulatory shock
Figure 20-6 shows at the far right several conditions that
is such an important clinical problem that it is discussed
cause abnormally low cardiac output. These conditions
in detail in Chapter 24.
fall into two categories: (1) those abnormalities that
cause the pumping effectiveness of the heart to fall too
low and (2) those that cause venous return to fall too
A More Quantitative Analysis
low.
of Cardiac Output Regulation
Decreased Cardiac Output Caused by Cardiac Factors. When-
Our discussion of cardiac output regulation thus far is
ever the heart becomes severely damaged, regardless
adequate for understanding the factors that control
of the cause, its limited level of pumping may fall below
cardiac output in most simple conditions. However, to
that needed for adequate blood flow to the tissues. Some
understand cardiac output regulation in especially
examples of this include (1) severe coronary blood vessel
stressful situations, such as the extremes of exercise,
blockage and consequent myocardial infarction,
(2)
cardiac failure, and circulatory shock, a more complex
severe valvular heart disease, (3) myocarditis, (4) cardiac
quantitative analysis is presented in the following
tamponade, and (5) cardiac metabolic derangements.
sections.
The effects of several of these are shown on the right in
To perform the more quantitative analysis, it is nec-
Figure 20-6, demonstrating the low cardiac outputs that
essary to distinguish separately the two primary factors
result.
concerned with cardiac output regulation:
(1) the
When the cardiac output falls so low that the tissues
pumping ability of the heart, as represented by cardiac
throughout the body begin to suffer nutritional defi-
output curves, and (2) the peripheral factors that affect
ciency, the condition is called cardiac shock. This is dis-
flow of blood from the veins into the heart, as repre-
cussed fully in Chapter 22 in relation to cardiac failure.
sented by venous return curves. Then one can put these
Decrease in Cardiac Output Caused by Non-cardiac Peripheral
curves together in a quantitative way to show how they
Factors—Decreased Venous Return. Anything that inter-
interact with each other to determine cardiac output,
feres with venous return also can lead to decreased
venous return, and right atrial pressure at the same time.
cardiac output. Some of these factors are the following:
1.
Decreased blood volume. By far, the most
common non-cardiac peripheral factor that leads
Cardiac Output Curves Used in the
to decreased cardiac output is decreased blood
Quantitative Analysis
volume, resulting most often from hemorrhage.
It is clear why this condition decreases the cardiac
Some of the cardiac output curves used to depict quan-
output: Loss of blood decreases the filling of the
titative heart pumping effectiveness have already been
vascular system to such a low level that there is not
shown in Figure 20-4. However, an additional set of
enough blood in the peripheral vessels to create
curves is required to show the effect on cardiac output
peripheral vascular pressures high enough to push
caused by changing external pressures on the outside of
the blood back to the heart.
the heart, as explained in the next section.
2.
Acute venous dilation. On some occasions, the
peripheral veins become acutely vasodilated. This
Effect of External Pressure Outside the Heart on Cardiac Output
results most often when the sympathetic nervous
Curves. Figure 20-7 shows the effect of changes in exter-
system suddenly becomes inactive. For instance,
nal cardiac pressure on the cardiac output curve. The
fainting often results from sudden loss of
normal external pressure is equal to the normal
sympathetic nervous system activity, which causes
intrapleural pressure (the pressure in the chest cavity),
the peripheral capacitative vessels, especially the
which is -4 mm Hg. Note in the figure that a rise in
veins, to dilate markedly. This decreases the filling
intrapleural pressure, to -2 mm Hg, shifts the entire
pressure of the vascular system because the blood
cardiac output curve to the right by the same amount.
volume can no longer create adequate pressure
This shift occurs because to fill the cardiac chambers
in the now flaccid peripheral blood vessels. As a
with blood requires an extra 2 mm Hg right atrial pres-
result, the blood “pools” in the vessels and does not
sure to overcome the increased pressure on the outside
return to the heart.
of the heart. Likewise, an increase in intrapleural pres-
3.
Obstruction of the large veins. On rare occasions,
sure to +2 mm Hg requires a 6 mm Hg increase in right
the large veins leading into the heart become
atrial pressure from the normal -4 mm Hg, which shifts
obstructed, so that the blood in the peripheral
the entire cardiac output curve 6 mm Hg to the right.
vessels cannot flow back into the heart.
Some of the factors that can alter the intrapleural
Consequently, the cardiac output falls markedly.
pressure and thereby shift the cardiac output curve are
4.
Decreased tissue mass, especially decreased skeletal
the following:
muscle mass. With normal aging or with prolonged
1. Cyclical changes of intrapleural pressure during
periods of physical inactivity, there is usually a
respiration, which are about ±2 mm Hg during
reduction in the size of the skeletal muscles. This, in
normal breathing but can be as much as
turn, decreases the total oxygen consumption and
±50 mm Hg during strenuous breathing.
238
Unit IV The Circulation
Hypereffective-increased
15
intrapleural pressure
15
Normal
10
10
5
5
Hypoeffective-reduced
intrapleural pressure
0
-4
0
+4
+8
+12
0
Right atrial pressure (mm Hg)
-4
0
+4
+8
+12
Right atrial pressure (mm Hg)
Figure 20-7
Figure 20-8
Cardiac output curves at different levels of intrapleural pressure
and at different degrees of cardiac tamponade. (Redrawn from
Combinations of two major patterns of cardiac output curves
Guyton AC, Jones CE, Coleman TB: Circulatory Physiology:
showing the effect of alterations in both extracardiac pressure and
Cardiac Output and Its Regulation. 2nd ed. Philadelphia: WB
effectiveness of the heart as a pump. (Redrawn from Guyton AC,
Saunders Co, 1973.)
Jones CE, Coleman TB: Circulatory Physiology: Cardiac Output
and Its Regulation. 2nd ed. Philadelphia: WB Saunders Co, 1973.)
2. Breathing against a negative pressure, which shifts
the curve to a more negative right atrial pressure
three principal factors that affect venous return to the
(to the left).
heart from the systemic circulation. They are as follows:
3. Positive pressure breathing, which shifts the curve to
1. Right atrial pressure, which exerts a backward force
the right.
on the veins to impede flow of blood from the
4. Opening the thoracic cage, which increases the
veins into the right atrium.
intrapleural pressure to 0 mm Hg and shifts the
2. Degree of filling of the systemic circulation
cardiac output curve to the right 4 mm Hg.
(measured by the mean systemic filling pressure),
5. Cardiac tamponade, which means accumulation of
which forces the systemic blood toward the heart
a large quantity of fluid in the pericardial cavity
(this is the pressure measured everywhere in the
around the heart with resultant increase in external
systemic circulation when all flow of blood is
cardiac pressure and shifting of the curve to the
stopped—we discuss this in detail later).
right. Note in Figure 20-7 that cardiac tamponade
3. Resistance to blood flow between the peripheral
shifts the upper parts of the curves farther to the
vessels and the right atrium.
right than the lower parts because the external
These factors can all be expressed quantitatively
“tamponade” pressure rises to higher values as the
by the venous return curve, as we explain in the next
chambers of the heart fill to increased volumes
sections.
during high cardiac output.
Normal Venous Return Curve
Combinations of Different Patterns of Cardiac Output Curves.
In the same way that the cardiac output curve relates
Figure 20-8 shows that the final cardiac output curve can
pumping of blood by the heart to right atrial pressure,
change as a result of simultaneous changes in (a) exter-
the venous return curve relates venous return also to
nal cardiac pressure and (b) effectiveness of the heart as
right atrial pressure—that is, the venous flow of blood
a pump. Thus, by knowing what is happening to the
into the heart from the systemic circulation at different
external pressure as well as to the capability of the heart
levels of right atrial pressure.
as a pump, one can express the momentary ability of the
The curve in Figure 20-9 is the normal venous return
heart to pump blood by a single cardiac output curve.
curve. This curve shows that when heart pumping capa-
bility becomes diminished and causes the right atrial
Venous Return Curves
pressure to rise, the backward force of the rising atrial
pressure on the veins of the systemic circulation
There remains the entire systemic circulation that must
decreases venous return of blood to the heart. If all
be considered before total analysis of cardiac regulation
nervous circulatory reflexes are prevented from acting,
can be achieved. To analyze the function of the systemic
venous return decreases to zero when the right atrial
circulation, we first remove the heart and lungs from the
pressure rises to about +7 mm Hg. Such a slight rise in
circulation of an animal and replace them with a pump
right atrial pressure causes a drastic decrease in venous
and artificial oxygenator system. Then, different factors,
return because the systemic circulation is a distensible
such blood volume, vascular resistances, and central
bag, so that any increase in back pressure causes blood
venous pressure in the right atrium, are altered to
to dam up in this bag instead of returning to the heart.
determine how the systemic circulation operates in
At the same time that the right atrial pressure is rising
different circulatory states. In these studies, one finds
and causing venous stasis, pumping by the heart also
Chapter 20
Cardiac Output, Venous Return, and Their Regulation
239
Plateau
Transitional zone
5
Mean
systemic
filling
Figure 20-9
pressure
Normal venous return curve. The
plateau is caused by collapse of
the large veins entering the chest
0
when the right atrial pressure falls
-8
-4
0
+4
+8
below atmospheric pressure.
Note also that venous return
Right atrial pressure (mm Hg)
becomes zero when the right
atrial pressure rises to equal the
mean systemic filling pressure.
approaches zero because of decreasing venous return.
Both the arterial and the venous pressures come to
equilibrium when all flow in the systemic circulation
ceases at a pressure of 7 mm Hg, which, by definition, is
Strong sympathetic
the mean systemic filling pressure (Psf).
stimulation
14
Normal circulatory
Plateau in the Venous Return Curve at Negative Atrial
system
12
Pressures—Caused by Collapse of the Large Veins. When the
Complete sympathetic
right atrial pressure falls below zero—that is, below
inhibition
10
atmospheric pressure—further increase in venous
Normal volume
return almost ceases. And by the time the right atrial
pressure has fallen to about -2 mm Hg, the venous
8
return will have reached a plateau. It remains at this
plateau level even though the right atrial pressure falls
6
to -20 mm Hg, -50 mm Hg, or even further. This plateau
is caused by collapse of the veins entering the chest. Neg-
4
ative pressure in the right atrium sucks the walls of the
veins together where they enter the chest, which pre-
2
vents any additional flow of blood from the peripheral
veins. Consequently, even very negative pressures in the
0
right atrium cannot increase venous return significantly
-0
1000 2000 3000 4000 5000 6000 7000
above that which exists at a normal atrial pressure of
Volume (ml)
0 mm Hg.
Mean Circulatory Filling Pressure and Mean Systemic Filling
Pressure, and Their Effect on Venous Return
Figure 20-10
When heart pumping is stopped by shocking the heart
with electricity to cause ventricular fibrillation or is
Effect of changes in total blood volume on the mean circulatory
stopped in any other way, flow of blood everywhere in
filling pressure (i.e., “volume-pressure curves” for the entire cir-
the circulation ceases a few seconds later. Without blood
culatory system). These curves also show the effects of strong
flow, the pressures everywhere in the circulation
sympathetic stimulation and complete sympathetic inhibition.
become equal. This equilibrated pressure level is called
the mean circulatory filling pressure.
Effect of Sympathetic Nervous Stimulation of the Circulation on
Effect of Blood Volume on Mean Circulatory Filling Pressure.
Mean Circulatory Filling Pressure. The green curve and blue
The greater the volume of blood in the circulation, the
curve in Figure 20-10 show the effects, respectively,
greater is the mean circulatory filling pressure because
of high and low levels of sympathetic nervous activity
extra blood volume stretches the walls of the vascula-
on the mean circulatory filling pressure. Strong sympa-
ture. The red curve in Figure 20-10 shows the approxi-
thetic stimulation constricts all the systemic blood
mate normal effect of different levels of blood volume
vessels as well as the larger pulmonary blood vessels
on the mean circulatory filling pressure. Note that at a
and even the chambers of the heart. Therefore, the
blood volume of about 4000 milliliters, the mean circu-
capacity of the system decreases, so that at each level of
latory filling pressure is close to zero because this is the
blood volume, the mean circulatory filling pressure is
“unstressed volume” of the circulation, but at a volume
increased. At normal blood volume, maximal sympa-
of 5000 milliliters, the filling pressure is the normal value
thetic stimulation increases the mean circulatory filling
of 7 mm Hg. Similarly, at still higher volumes, the mean
pressure from 7 mm Hg to about 2.5 times that value, or
circulatory filling pressure increases almost linearly.
about 17 mm Hg.
240
Unit IV
The Circulation
Conversely, complete inhibition of the sympathetic
Conversely, the lower the mean systemic filling pressure,
nervous system relaxes both the blood vessels and the
the more the curve shifts downward and to the left.
heart, decreasing the mean circulatory filling pressure
To express this another way, the greater the system is
from the normal value of 7 mm Hg down to about 4 mm
filled, the easier it is for blood to flow into the heart.
Hg. Before leaving Figure 20-10, note specifically how
The less the filling, the more difficult it is for blood to
steep the curves are. This means that even slight changes
flow into the heart.
in blood volume or slight changes in the capacity of the
system caused by various levels of sympathetic activity
can have large effects on the mean circulatory filling
“Pressure Gradient for Venous Return”—When This Is Zero, There
pressure.
Is No Venous Return. When the right atrial pressure rises
to equal the mean systemic filling pressure, there is no
Mean Systemic Filling Pressure and Its Relation to Mean Circula-
longer any pressure difference between the peripheral
tory Filling Pressure. The mean systemic filling pressure,
vessels and the right atrium. Consequently, there can no
Psf, is slightly different from the mean circulatory filling
longer be any blood flow from any peripheral vessels
pressure. It is the pressure measured everywhere in the
back to the right atrium. However, when the right atrial
systemic circulation after blood flow has been stopped
pressure falls progressively lower than the mean sys-
by clamping the large blood vessels at the heart, so that
temic filling pressure, the flow to the heart increases
the pressures in the systemic circulation can be meas-
proportionately, as one can see by studying any of the
ured independently from those in the pulmonary circu-
venous return curves in Figure 20-11. That is, the greater
lation. The mean systemic pressure, although almost
the difference between the mean systemic filling pressure
impossible to measure in the living animal, is the impor-
and the right atrial pressure, the greater becomes the
tant pressure for determining venous return. The mean
venous return. Therefore, the difference between these
systemic filling pressure, however, is almost always nearly
two pressures is called the pressure gradient for venous
equal to the mean circulatory filling pressure because the
return.
pulmonary circulation has less than one eighth as much
capacitance as the systemic circulation and only about
one tenth as much blood volume.
Resistance to Venous Return
In the same way that mean systemic filling pressure rep-
Effect on the Venous Return Curve of Changes in Mean Systemic
resents a pressure pushing venous blood from the
Filling Pressure. Figure 20-11 shows the effects on the
periphery toward the heart, there is also resistance to
venous return curve caused by increasing or decreasing
this venous flow of blood. It is called the resistance to
the mean systemic filling pressure (Psf). Note in Figure
venous return. Most of the resistance to venous return
20-11 that the normal mean systemic filling pressure is
occurs in the veins, although some occurs in the arteri-
7 mm Hg. Then, for the uppermost curve in the figure,
oles and small arteries as well.
the mean systemic filling pressure has been increased to
Why is venous resistance so important in determin-
14 mm Hg, and for the lowermost curve, has been
ing the resistance to venous return? The answer is that
decreased to 3.5 mm Hg. These curves demonstrate that
when the resistance in the veins increases, blood begins
the greater the mean systemic filling pressure (which
to be dammed up, mainly in the veins themselves. But
also means the greater the “tightness” with which the
the venous pressure rises very little because the veins
circulatory system is filled with blood) the more the
are highly distensible. Therefore, this rise in venous
venous return curve shifts upward and to the right.
pressure is not very effective in overcoming the resist-
ance, and blood flow into the right atrium decreases
drastically. Conversely, when arteriolar and small artery
resistances increase, blood accumulates in the arteries,
which have a capacitance only 1/30 as great as that of
the veins. Therefore, even slight accumulation of blood
in the arteries raises the pressure greatly—30 times as
10
much as in the veins—and this high pressure does over-
Psf = 3.5
come much of the increased resistance. Mathematically,
Psf = 7
it turns out that about two thirds of the so-called “resist-
Psf = 14
ance to venous return” is determined by venous resist-
5
ance, and about one third by the arteriolar and small
artery resistance.
Venous return can be calculated by the following
0
formula:
-4
0
+4
+8
+12
Right atrial pressure (mm Hg)
Psf PRA
VR =
RVR
in which VR is venous return, Psf is mean systemic
Figure 20-11
filling pressure, PRA is right atrial pressure, and RVR
is resistance to venous return. In the healthy human
Venous return curves showing the normal curve when the mean
adult, the values for these are as follows: venous return
systemic filling pressure (Psf) is 7 mm Hg, and showing the effect
equals 5 L/min, mean systemic filling pressure equals
of altering the Psf to either
3.5 or 14 mm Hg. (Redrawn from
Guyton AC, Jones CE, Coleman TB: Circulatory Physiology:
7 mm Hg, right atrial pressure equals 0 mm Hg, and
Cardiac Output and Its Regulation. 2nd ed. Philadelphia: WB
resistance to venous return equals 1.4 mm Hg per liter
Saunders Co, 1973.)
of blood flow.
Chapter 20
Cardiac Output, Venous Return, and Their Regulation
241
20
Normal resistance
2
¥ resistance
15
1/2 resistance
1/3 resistance
15
10
10
5
Psf = 10.5
Psf = 10
5
Psf = 7
Psf = 2.3
Psf = 7
0
-4
0
+4
+8
+12
Right atrial pressure (mm Hg)
0
-4
0
+4
+8
Right atrial pressure (mm Hg)
Figure 20-13
Combinations of the major patterns of venous return curves,
showing the effects of simultaneous changes in mean systemic
Figure 20-12
filling pressure
(Psf) and in
“resistance to venous return.”
(Redrawn from Guyton AC, Jones CE, Coleman TB: Circulatory
Venous return curves depicting the effect of altering the “resist-
Physiology: Cardiac Output and Its Regulation. 2nd ed. Philadel-
ance to venous return.” Psf, mean systemic filling pressure.
phia: WB Saunders Co, 1973.)
(Redrawn from Guyton AC, Jones CE, Coleman TB: Circulatory
Physiology: Cardiac Output and Its Regulation. 2nd ed. Philadel-
phia: WB Saunders Co, 1973.)
equal the cardiac output from the heart and (2) the right
atrial pressure is the same for both the heart and the
Effect of Resistance to Venous Return on the Venous Return
systemic circulation.
Curve. Figure 20-12 demonstrates the effect of different
Therefore, one can predict the cardiac output and
levels of resistance to venous return on the venous
right atrial pressure in the following way: (1) Determine
return curve, showing that a decrease in this resistance
the momentary pumping ability of the heart and depict
to one-half normal allows twice as much flow of blood
this in the form of a cardiac output curve; (2) determine
and, therefore, rotates the curve upward to twice as great
the momentary state of flow from the systemic circula-
a slope. Conversely, an increase in resistance to twice
tion into the heart and depict this in the form of a
normal rotates the curve downward to one-half as great
venous return curve; and (3) “equate” these curves
a slope.
against each other, as shown in Figure 20-14.
Note also that when the right atrial pressure rises to
Two curves in the figure depict the normal cardiac
equal the mean systemic filling pressure, venous return
output curve (red line) and the normal venous return
becomes zero at all levels of resistance to venous return
curve (blue line). There is only one point on the graph,
because when there is no pressure gradient to cause
point A, at which the venous return equals the cardiac
flow of blood, it makes no difference what the resistance
output and at which the right atrial pressure is the same
is in the circulation; the flow is still zero. Therefore, the
for both the heart and the systemic circulation. There-
highest level to which the right atrial pressure can rise,
fore, in the normal circulation, the right atrial pressure,
regardless of how much the heart might fail, is equal to
cardiac output, and venous return are all depicted by
the mean systemic filling pressure.
point A, called the equilibrium point, giving a normal
value for cardiac output of 5 liters per minute and a
Combinations of Venous Return Curve Patterns. Figure 20-13
right atrial pressure of 0 mm Hg .
shows effects on the venous return curve caused by
simultaneous changes in mean systemic pressure (Psf)
Effect of Increased Blood Volume on Cardiac Output. A sudden
and resistance to venous return, demonstrating that
increase in blood volume of about 20 per cent increases
both these factors can operate simultaneously.
the cardiac output to about 2.5 to 3 times normal. An
analysis of this effect is shown in Figure 20-14. Imme-
diately on infusing the large quantity of extra blood, the
Analysis of Cardiac Output and
increased filling of the system causes the mean systemic
Right Atrial Pressure, Using
filling pressure (Psf) to increase to 16 mm Hg, which
Simultaneous Cardiac Output and
shifts the venous return curve to the right. At the same
time, the increased blood volume distends the blood
Venous Return Curves
vessels, thus reducing their resistance and thereby
In the complete circulation, the heart and the systemic
reducing the resistance to venous return, which rotates
circulation must operate together. This means that (1)
the curve upward. As a result of these two effects, the
the venous return from the systemic circulation must
venous return curve of Figure 20-14 is shifted to the
242
Unit IV The Circulation
25
20
Maximal sympathetic
stimulation
Moderate sympathetic
20
stimulation
15
B
Normal
Spinal anesthesia
15
10
D
10
5
A
Psf = 7
Psf = 16
C
A
5
0
-4
0
+4
+8
+12
+16
B
Right atrial pressure (mm Hg)
0
-4
0
+4
+8
+12
+16
Right atrial pressure (mm Hg)
Figure 20-14
The two solid curves demonstrate an analysis of cardiac output
Figure 20-15
and right atrial pressure when the cardiac output (red line) and
venous return (blue line) curves are normal. Transfusion of blood
Analysis of the effect on cardiac output of (1) moderate sympa-
equal to 20 per cent of the blood volume causes the venous return
thetic stimulation (from point A to point C), (2) maximal sympa-
curve to become the dashed curve; as a result, the cardiac output
thetic stimulation (point D), and (3) sympathetic inhibition caused
and right atrial pressure shift from point A to point B. Psf, mean
by total spinal anesthesia (point B). (Redrawn from Guyton AC,
systemic filling pressure.
Jones CE, Coleman TB: Circulatory Physiology: Cardiac Output
and Its Regulation. 2nd ed. Philadelphia: WB Saunders Co, 1973.)
right. This new curve equates with the cardiac output
each other at point A, which represents a normal venous
curve at point B, showing that the cardiac output and
return and cardiac output of 5 L/min and a right atrial
venous return increase 2.5 to 3 times, and that the right
pressure of 0 min Hg. Note in the figure that maximal
atrial pressure rises to about +8 mm Hg.
sympathetic stimulation (green curves) increases the
mean systemic filling pressure to 17 mm Hg (depicted
Further Compensatory Effects Initiated in Response to Increased
by the point at which the venous return curve reaches
Blood Volume. The greatly increased cardiac output
the zero venous return level). And the sympathetic stim-
caused by increased blood volume lasts for only a few
ulation also increases pumping effectiveness of the
minutes because several compensatory effects immedi-
heart by nearly 100 per cent. As a result, the cardiac
ately begin to occur: (1) The increased cardiac output
output rises from the normal value at equilibrium point
increases the capillary pressure so that fluid begins to
A to about double normal at equilibrium point D—and
transude out of the capillaries into the tissues, thereby
yet the right atrial pressure hardly changes. Thus, differ-
returning the blood volume toward normal. (2) The
ent degrees of sympathetic stimulation can increase the
increased pressure in the veins causes the veins to
cardiac output progressively to about twice normal for
continue distending gradually by the mechanism called
short periods of time, until other compensatory effects
stress-relaxation, especially causing the venous blood
occur within seconds or minutes.
reservoirs, such as the liver and spleen, to distend, thus
Effect of Sympathetic Inhibition on Cardiac Output. The sym-
reducing the mean systemic pressure. (3) The excess
pathetic nervous system can be blocked by inducing
blood flow through the peripheral tissues causes
total spinal anesthesia or by using some drug, such as
autoregulatory increase in the peripheral resistance,
hexamethonium, that blocks transmission of nerve
thus increasing the resistance to venous return. These
signals through the autonomic ganglia. The lowermost
factors cause the mean systemic filling pressure to
curves in Figure 20-15 show the effect of sympathetic
return back toward normal and the resistance vessels of
inhibition caused by total spinal anesthesia, demon-
the systemic circulation to constrict. Therefore, gradu-
strating that (1) the mean systemic filling pressure falls
ally, over a period of 10 to 40 minutes, the cardiac output
to about 4 mm Hg and (2) the effectiveness of the heart
returns almost to normal.
as a pump decreases to about 80 per cent of normal. The
cardiac output falls from point A to point B, which is a
Effect of Sympathetic Stimulation on Cardiac Output. Sympa-
decrease to about 60 per cent of normal.
thetic stimulation affects both the heart and the sys-
temic circulation: (1) It makes the heart a stronger pump.
Effect of Opening a Large Arteriovenous Fistula. Figure 20-16
(2) In the systemic circulation, it increases the mean
shows various stages of circulatory changes that occur
systemic filling pressure because of contraction of
after opening a large arteriovenous fistula, that is, after
the peripheral vessels—especially the veins—and it
making an opening directly between a large artery and
increases the resistance to venous return.
a large vein.
In Figure
20-15, the normal cardiac output and
1. The two red curves crossing at point A show the
venous return curves are depicted; these equate with
normal condition.
Chapter 20
Cardiac Output, Venous Return, and Their Regulation
243
D
20
20
15
C
10
5
15
B
0
0
1
2
10
Seconds
A
5
Figure 20-17
Pulsatile blood flow in the root of the aorta recorded using an elec-
tromagnetic flowmeter.
0
-4
0
+4
+8
+12
Right atrial pressure (mm Hg)
4. Point D shows the effect after several more weeks.
By this time, the blood volume has increased
because the slight reduction in arterial pressure and
the sympathetic stimulation have both reduced
Figure 20-16
kidney output of urine. The mean systemic filling
pressure has now risen to +12 mm Hg, shifting the
Analysis of successive changes in cardiac output and right atrial
venous return curve another 3 mm Hg to the right.
pressure in a human being after a large arteriovenous (AV) fistula
Also, the prolonged increased workload on the
is suddenly opened. The stages of the analysis, as shown by the
heart has caused the heart muscle to hypertrophy
equilibrium points, are A, normal conditions; B, immediately after
slightly, raising the level of the cardiac output curve
opening the AV fistula; C, 1 minute or so after the sympathetic
reflexes have become active; and D, several weeks after the blood
still further. Therefore, point D shows a cardiac
volume has increased and the heart has begun to hypertrophy.
output now of almost 20 L/min and a right atrial
(Redrawn from Guyton AC, Jones CE, Coleman TB: Circulatory
pressure of about 6 mm Hg.
Physiology: Cardiac Output and Its Regulation. 2nd ed. Philadel-
phia: WB Saunders Co, 1973.)
Other Analyses of Cardiac Output Regulation. In Chapter 21,
analysis of cardiac output regulation during exercise is
presented, and in Chapter 22, analyses of cardiac output
regulation at various stages of congestive heart failure
2.
The curves crossing at point B show the circulatory
are shown.
condition immediately after opening the large
fistula. The principal effects are (1) a sudden and
precipitous rotation of the venous return curve
Methods for Measuring
upward caused by the large decrease in resistance to
Cardiac Output
venous return when blood is allowed to flow with
almost no impediment directly from the large
In animal experiments, one can cannulate the aorta, pul-
arteries into the venous system, bypassing most
monary artery, or great veins entering the heart and
of the resistance elements of the peripheral
measure the cardiac output using any type of flowme-
circulation, and (2) a slight increase in the level of
ter. An electromagnetic or ultrasonic flowmeter can also
the cardiac output curve because opening the fistula
be placed on the aorta or pulmonary artery to measure
decreases the peripheral resistance and allows
cardiac output.
an acute fall in arterial pressure against which
In the human, except in rare instances, cardiac output
the heart can pump more easily. The net result,
is measured by indirect methods that do not require
depicted by point B, is an increase in cardiac output
surgery. Two of the methods commonly used are the
from 5 L/min up to 13 L/min and an increase in
oxygen Fick method and the indicator dilution method.
right atrial pressure to about +3 mm Hg.
3.
Point C represents the effects about 1 minute later,
after the sympathetic nerve reflexes have restored
Pulsatile Output of the Heart as
the arterial pressure almost to normal and caused
Measured by an Electromagnetic or
two other effects: (1) an increase in the mean
Ultrasonic Flowmeter
systemic filling pressure (because of constriction of
all veins and arteries) from 7 to 9 mm Hg, thus
Figure 20-17 shows a recording in a dog of blood flow
shifting the venous return curve 2 mm Hg to the
in the root of the aorta made using an electromagnetic
right, and (2) further elevation of the cardiac
flowmeter. It demonstrates that the blood flow rises
output curve because of sympathetic nervous
rapidly to a peak during systole, and then at the end of
excitation of the heart. The cardiac output now
systole reverses for a fraction of a second. This reverse
rises to almost 16 L/min, and the right atrial
flow causes the aortic valve to close and the flow to
pressure to about 4 mm Hg.
return to zero.
244
Unit IV The Circulation
Measurement of Cardiac Output Using
Indicator Dilution Method for
the Oxygen Fick Principle
Measuring Cardiac Output
To measure cardiac output by the so-called “indicator
The Fick principle is explained by Figure 20-18. This
dilution method,” a small amount of indicator, such as
figure shows that 200 milliliters of oxygen are being
a dye, is injected into a large systemic vein or, prefer-
absorbed from the lungs into the pulmonary blood
ably, into the right atrium. This passes rapidly through
each minute. It also shows that the blood entering the
the right side of the heart, then through the blood
right heart has an oxygen concentration of 160 milli-
vessels of the lungs, through the left side of the heart
liters per liter of blood, whereas that leaving the left
and, finally, into the systemic arterial system. The con-
heart has an oxygen concentration of 200 milliliters
centration of the dye is recorded as the dye passes
through one of the peripheral arteries, giving a curve as
per liter of blood. From these data, one can calculate
shown in Figure 20-19. In each of these instances, 5 mil-
that each liter of blood passing through the lungs
ligrams of Cardio-Green dye was injected at zero time.
absorbs 40 milliliters of oxygen.
In the top recording, none of the dye passed into the
Because the total quantity of oxygen absorbed into
arterial tree until about 3 seconds after the injection, but
the blood from the lungs each minute is 200 milliliters,
then the arterial concentration of the dye rose rapidly
dividing 200 by 40 calculates a total of five 1-liter por-
to a maximum in about 6 to 7 seconds. After that, the
tions of blood that must pass through the pulmonary
concentration fell rapidly, but before the concentration
circulation each minute to absorb this amount of
reached zero, some of the dye had already circulated all
oxygen. Therefore, the quantity of blood flowing
the way through some of the peripheral systemic vessels
through the lungs each minute is 5 liters, which is also
and returned through the heart for a second time. Con-
sequently, the dye concentration in the artery began to
a measure of the cardiac output. Thus, the cardiac
rise again. For the purpose of calculation, it is necessary
output can be calculated by the following formula:
to extrapolate the early down-slope of the curve to the
zero point, as shown by the dashed portion of each
Cardiac output(L min)
curve. In this way, the extrapolated time-concentration
O absorbed per minute by the lungs(ml min
2
)
curve of the dye in the systemic artery without recircu-
=
Arteriovenous O2 difference
(ml L of blood)
lation can be measured in its first portion and estimated
reasonably accurately in its latter portion.
In applying this Fick procedure for measuring
Once the extrapolated time-concentration curve has
cardiac output in the human being, mixed venous
been determined, one then calculates the mean con-
blood is usually obtained through a catheter inserted
centration of dye in the arterial blood for the duration
of the curve. For instance, in the top example of Figure
up the brachial vein of the forearm, through the sub-
20-19, this was done by measuring the area under the
clavian vein, down to the right atrium, and, finally, into
the right ventricle or pulmonary artery. And systemic
arterial blood can then be obtained from any systemic
artery in the body. The rate of oxygen absorption by
the lungs is measured by the rate of disappearance of
oxygen from the respired air, using any type of oxygen
5 mg
meter.
injected
0.5
0.4
0.3
0.2
0.1
0
0
10
20
30
LUNGS
5 mg
0.5
injected
0.4
Oxygen used = 200 ml/min
0.3
0.2
0.1
0
Cardiac output =
0
10
20
30
O2 =
5000 ml/min
O2 =
Seconds
160 ml/L
200 ml/L
right heart
left heart
Figure 20-19
Extrapolated dye concentration curves used to calculate two sep-
Figure 20-18
arate cardiac outputs by the dilution method. (The rectangular
areas are the calculated average concentrations of dye in the arte-
Fick principle for determining cardiac output.
rial blood for the durations of the respective extrapolated curves.)
Chapter 20
Cardiac Output, Venous Return, and Their Regulation
245
entire initial and extrapolated curve and then averaging
Guyton AC: Venous return. In: Hamilton WF (ed): Hand-
the concentration of dye for the duration of the curve;
book of Physiology. Sec 2, Vol. 2. Baltimore, Williams &
one can see from the shaded rectangle straddling the
Wilkins, 1963, p 1099.
curve in the upper figure that the average concentration
Guyton AC: Determination of cardiac output by equating
of dye was 0.25 mg/dl of blood and that the duration
venous return curves with cardiac response curves. Physiol
of this average value was 12 seconds. A total of 5 mil-
Rev 35:123, 1955.
ligrams of dye had been injected at the beginning of the
Guyton AC, Coleman TG, Granger HJ: Circulation: overall
experiment. For blood carrying only 0.25 milligram of
regulation. Annu Rev Physiol 34:13, 1972.
dye in each deciliter to carry the entire 5 milligrams of
Guyton AC, Jones CE, Coleman TG: Circulatory Physiology:
dye through the heart and lungs in 12 seconds, a total
Cardiac Output and Its Regulation. Philadelphia: WB
of 20 1-deciliter portions of blood would have passed
Saunders Co, 1973.
through the heart during the 12 seconds, which would
Guyton AC, Lindsey AW, Kaufmann BN: Effect of mean cir-
be the same as a cardiac output of 2 L/12 sec, or
culatory filling pressure and other peripheral circulatory
10 L/min. We leave it to the reader to calculate the
factors on cardiac output. Am J Physiol 180:463-468, 1955.
cardiac output from the bottom extrapolated curve of
Koch WJ, Lefkowitz RJ, Rockman HA: Functional conse-
Figure 20-19. To summarize, the cardiac output can be
quences of altering myocardial adrenergic receptor sig-
determined using the following formula:
naling. Annu Rev Physiol 62:237, 2000.
Rockman HA, Koch WJ, Lefkowitz RJ: Seven-transmem-
brane-spanning receptors and heart function. Nature
Cardiac output(ml min)=
415:206, 2002.
Milligrams of dye injected
¥
60
Rothe CF: Mean circulatory filling pressure: its meaning and
Ê Average concentration of dyeˆ
ÊDuration ofˆ
measurement. J Appl Physiol 74:499, 1993.
Á
in each milliliter of blood
˜
¥
Á
the curve
˜
Rothe CF: Reflex control of veins and vascular capacitance.
Ë
for the duration of the curve
¯
Ë
in seconds
¯
Physiol Rev 63:1281, 1983.
Sarnoff SJ, Berglund E: Ventricular function. 1. Starling’s law
of the heart, studied by means of simultaneous right and
left ventricular function curves in the dog. Circulation
References
9:706-718, 1953.
Uemura K, Sugimachi M, Kawada T, et al: A novel frame-
Brengelmann GL: A critical analysis of the view that right
work of circulatory equilibrium. Am J Physiol Heart Circ
atrial pressure determines venous return. J Appl Physiol
Physiol 286:H2376, 2004.
94:849, 2003.
Vatner SF, Braunwald E: Cardiovascular control mecha-
Gaasch WH, Zile MR: Left ventricular diastolic dysfunction
nisms in the conscious state. N Engl J Med 293:970, 1975.
and diastolic heart failure. Annu Rev Med. 55:373, 2004.
C H A P T E R
2
1
Muscle Blood Flow and Cardiac
Output During Exercise; the
Coronary Circulation and Ischemic
Heart Disease
In this chapter we consider (1) blood flow to the
skeletal muscles and
(2) coronary blood flow
to the heart. Regulation of each of these is
achieved mainly by local control of vascular
resistance in response to muscle tissue metabolic
needs.
In addition, related subjects are discussed, such as
(1) cardiac output control during exercise, (2) char-
acteristics of heart attacks, and (3) the pain of angina pectoris.
Blood Flow in Skeletal Muscle and Blood Flow
Regulation During Exercise
Very strenuous exercise is one of the most stressful conditions that the normal
circulatory system faces. This is true because there is such a large mass of skele-
tal muscle in the body, all of it requiring large amounts of blood flow. Also, the
cardiac output often must increase in the non-athlete to four to five times
normal, or in the well-trained athlete to six to seven times normal.
Rate of Blood Flow Through the Muscles
During rest, blood flow through skeletal muscle averages 3 to 4 ml/min/100 g of
muscle. During extreme exercise in the well-conditioned athlete, this can increase
15- to 25-fold, rising to 50 to 80 ml/min/100 g of muscle.
Blood Flow During Muscle Contractions. Figure 21-1 shows a record of blood flow
changes in a calf muscle of a human leg during strong rhythmical muscular exer-
cise. Note that the flow increases and decreases with each muscle contraction.
At the end of the contractions, the blood flow remains very high for a few
seconds but then fades toward normal during the next few minutes.
The cause of the lower flow during the muscle contraction phase of exercise
is compression of the blood vessels by the contracted muscle. During strong
tetanic contraction, which causes sustained compression of the blood vessels, the
blood flow can be almost stopped, but this also causes rapid weakening of the
contraction.
Increased Blood Flow in Muscle Capillaries During Exercise. During rest, some muscle
capillaries have little or no flowing blood. But during strenuous exercise, all
the capillaries open. This opening of dormant capillaries diminishes the dis-
tance that oxygen and other nutrients must diffuse from the capillaries to the
contracting muscle fibers and sometimes contributes a twofold to threefold
increased capillary surface area through which oxygen and nutrients can diffuse
from the blood.
246
Chapter 21
Muscle Blood Flow and Cardiac Output During Exercise
247
(in some species of animals) sympathetic vasodilator
nerves as well.
Rhythmic exercise
Sympathetic Vasoconstrictor Nerves. The sympa-
40
thetic vasoconstrictor nerve fibers secrete norepineph-
rine at their nerve endings. When maximally activated,
this can decrease blood flow through resting muscles
to as little as one half to one third normal. This vaso-
20
constriction is of physiologic importance in circulatory
shock and during other periods of stress when it is
necessary to maintain a normal or even high arterial
pressure.
Calf
flow
In addition to the norepinephrine secreted at the
0
sympathetic vasoconstrictor nerve endings, the medul-
0
10
16
18
lae of the two adrenal glands also secrete large
Minutes
amounts of norepinephrine plus even more epineph-
rine into the circulating blood during strenuous exer-
cise. The circulating norepinephrine acts on the muscle
vessels to cause a vasoconstrictor effect similar to that
Figure 21-1
caused by direct sympathetic nerve stimulation. The
Effects of muscle exercise on blood flow in the calf of a leg during
epinephrine, however, often has a slight vasodilator
strong rhythmical contraction. The blood flow was much less
effect because epinephrine excites more of the beta
during contractions than between contractions. (Adapted from
adrenergic receptors of the vessels, which are vasodila-
Barcroft and Dornhorst: J Physiol 109:402, 1949.)
tor receptors, in contrast to the alpha vasoconstrictor
receptors excited especially by norepinephrine. These
receptors are discussed in Chapter 60.
Control of Blood Flow Through the
Skeletal Muscles
Total Body Circulatory Readjustments
Local Regulation—Decreased Oxygen in Muscle Greatly
During Exercise
Enhances Flow. The tremendous increase in muscle
blood flow that occurs during skeletal muscle activity
Three major effects occur during exercise that are
is caused primarily by chemical effects acting directly
essential for the circulatory system to supply the
on the muscle arterioles to cause dilation. One of the
tremendous blood flow required by the muscles. They
most important chemical effects is reduction of oxygen
are (1) mass discharge of the sympathetic nervous
in the muscle tissues. That is, during muscle activity,
system throughout the body with consequent stimula-
the muscle uses oxygen rapidly, thereby decreasing the
tory effects on the entire circulation, (2) increase in
oxygen concentration in the tissue fluids. This in turn
arterial pressure, and (3) increase in cardiac output.
causes local arteriolar vasodilation both because the
arteriolar walls cannot maintain contraction in the
Effects of Mass Sympathetic Discharge
absence of oxygen and because oxygen deficiency
At the onset of exercise, signals are transmitted not
causes release of vasodilator substances. The most
only from the brain to the muscles to cause muscle
important vasodilator substance is probably adeno-
contraction but also into the vasomotor center to ini-
sine, but experiments have shown that even large
tiate mass sympathetic discharge throughout the body.
amounts of adenosine infused directly into a muscle
Simultaneously, the parasympathetic signals to the
artery cannot sustain vasodilation in skeletal muscle
heart are attenuated. Therefore, three major circula-
for more than about 2 hours.
tory effects result.
Fortunately, even after the muscle blood vessels
First, the heart is stimulated to greatly increased
have become insensitive to the vasodilator effects of
heart rate and increased pumping strength as a result
adenosine, still other vasodilator factors continue to
of the sympathetic drive to the heart plus release of
maintain increased capillary blood flow as long as the
the heart from normal parasympathetic inhibition.
exercise continues. These factors include (1) potassium
Second, most of the arterioles of the peripheral
ions, (2) adenosine triphosphate (ATP), (3) lactic acid,
circulation are strongly contracted, except for the
and (4) carbon dioxide. We still do not know quanti-
arterioles in the active muscles, which are strongly
tatively how great a role each of these plays in in-
vasodilated by the local vasodilator effects in the
creasing muscle blood flow during muscle activity;
muscles as noted above. Thus, the heart is stimulated
this subject was discussed in additional detail in
to supply the increased blood flow required by the
Chapter 17.
muscles, while at the same time blood flow through
most nonmuscular areas of the body is temporarily
Nervous Control of Muscle Blood Flow. In addition to local
reduced, thereby temporarily “lending” their blood
tissue vasodilator mechanisms, skeletal muscles are
supply to the muscles. This accounts for as much as
provided with sympathetic vasoconstrictor nerves and
2 L/min of extra blood flow to the muscles, which is
248
Unit IV The Circulation
exceedingly important when one thinks of a person
running for his life—even a fractional increase in
running speed may make the difference between life
25
and death. Two of the peripheral circulatory systems,
B
the coronary and cerebral systems, are spared this vaso-
20
constrictor effect because both these circulatory areas
have poor vasoconstrictor innervation—fortunately so
15
because both the heart and the brain are as essential
to exercise as are the skeletal muscles.
10
Third, the muscle walls of the veins and other capac-
A
itative areas of the circulation are contracted power-
5
fully, which greatly increases the mean systemic filling
pressure. As we learned in Chapter 20, this is one of
0
-4
0
+4
+8
+12
+16
+20
+24
the most important factors in promoting increase in
venous return of blood to the heart and, therefore, in
Right atrial pressure (mm Hg)
increasing the cardiac output.
Increase in Arterial Pressure During Exercise—
Figure 21-2
An Important Result of Increased
Sympathetic Stimulation
Graphical analysis of change in cardiac output and right atrial
pressure with onset of strenuous exercise. Black curves, normal
One of the most important effects of increased sym-
circulation. Red curves, heavy exercise.
pathetic stimulation in exercise is to increase the arte-
rial pressure. This results from multiple stimulatory
effects, including (1) vasoconstriction of the arterioles
and small arteries in most tissues of the body except
assume, for instance, that the arterial pressure rises
the active muscles, (2) increased pumping activity
30 per cent, a common increase during heavy exercise.
by the heart, and (3) a great increase in mean systemic
This 30 per cent increase causes 30 per cent more force
filling pressure caused mainly by venous contraction.
to push blood through the muscle tissue vessels. But
These effects, working together, virtually always
this is not the only important effect; the extra pressure
increase the arterial pressure during exercise. This
also stretches the walls of the vessels so much that
increase can be as little as 20 mm Hg or as great as
muscle total flow often rises to more than 20 times
80 mm Hg, depending on the conditions under which
normal.
the exercise is performed. When a person performs
exercise under tense conditions but uses only a few
Importance of the Increase in Cardiac Output
muscles, the sympathetic nervous response still occurs
During Exercise
everywhere in the body. In the few active muscles,
Many different physiologic effects occur at the same
vasodilation occurs, but everywhere else in the body
time during exercise to increase cardiac output
the effect is mainly vasoconstriction, often increasing
approximately in proportion to the degree of exercise.
the mean arterial pressure to as high as 170 mm Hg.
In fact, the ability of the circulatory system to provide
Such a condition might occur in a person standing
increased cardiac output for delivery of oxygen and
on a ladder and nailing with a hammer on the ceiling
other nutrients to the muscles during exercise is
above. The tenseness of the situation is obvious.
equally as important as the strength of the muscles
Conversely, when a person performs massive
themselves in setting the limit for continued muscle
whole-body exercise, such as running or swimming,
work. For instance, marathon runners who can
the increase in arterial pressure is often only 20 to
increase their cardiac outputs the most are generally
40 mm Hg. This lack of a large increase in pressure
the same persons who have record-breaking running
results from the extreme vasodilation that occurs
times.
simultaneously in large masses of active muscle.
Graphical Analysis of the Changes in Cardiac Output During
Why Is the Arterial Pressure Increase During Exercise Impor-
Heavy Exercise. Figure 21-2 shows a graphical analysis
tant? When muscles are stimulated maximally in a lab-
of the large increase in cardiac output that occurs
oratory experiment but without allowing the arterial
during heavy exercise. The cardiac output and venous
pressure to rise, muscle blood flow seldom rises more
return curves crossing at point A give the analysis for
than about eightfold. Yet, we know from studies of
the normal circulation; and the curves crossing at point
marathon runners that muscle blood flow can increase
B analyze heavy exercise. Note that the great increase
from as little as 1 L/min for the whole body during rest
in cardiac output requires significant changes in both
to at least 20 L/min during maximal activity. Therefore,
the cardiac output curve and the venous return curve,
it is clear that muscle blood flow can increase much
as follow.
more than occurs in the aforementioned simple labo-
The increased level of the cardiac output curve is
ratory experiment. What is the difference? Mainly, the
easy to understand. It results almost entirely from
arterial pressure rises during normal exercise. Let us
sympathetic stimulation of the heart that causes (1)
Chapter 21
Muscle Blood Flow and Cardiac Output During Exercise
249
increased heart rate, often up to rates as high as 170 to
Aortic valve
190 beats/min, and (2) increased strength of contrac-
tion of the heart, often to as much as twice normal.
Without this increased level of the output curve, the
increase in cardiac output would be limited to the
plateau level of the normal heart, which would be a
Left coronary
maximum increase of cardiac output of only about
artery
2.5-fold rather than the 4-fold that can commonly be
achieved by the untrained runner and the 7-fold that
Right coronary
can be achieved in some marathon runners.
artery
Now study the venous return curves. If no change
occurred from the normal venous return curve, the
cardiac output could hardly rise at all in exercise
because the upper plateau level of the normal venous
return curve is only
6 L/min. Yet two important
changes do occur:
1. The mean systemic filling pressure rises
Figure 21-3
tremendously at the onset of heavy exercise. This
results partly from the sympathetic stimulation
The coronary arteries.
that contracts the veins and other capacitative parts
of the circulation. In addition, tensing of the
abdominal and other skeletal muscles of the body
compresses many of the internal vessels, thus
Physiologic Anatomy of the Coronary
providing more compression of the entire
Blood Supply
capacitative vascular system, causing a still greater
increase in mean systemic filling pressure.
Figure 21-3 shows the heart and its coronary blood
During maximal exercise, these two effects
supply. Note that the main coronary arteries lie on the
together can increase the mean systemic filling
surface of the heart and smaller arteries then pene-
pressure from a normal level of 7 mm Hg to as
trate from the surface into the cardiac muscle mass. It
high as 30 mm Hg.
is almost entirely through these arteries that the heart
2. The slope of the venous return curve rotates
receives its nutritive blood supply. Only the inner 1/10
upward. This is caused by decreased resistance in
millimeter of the endocardial surface can obtain sig-
virtually all the blood vessels in active muscle
nificant nutrition directly from the blood inside the
tissue, which also causes resistance to venous
cardiac chambers, so that this source of muscle nutri-
return to decrease, thus increasing the upward
tion is minuscule.
slope of the venous return curve.
The left coronary artery supplies mainly the anterior
Therefore, the combination of increased mean sys-
and left lateral portions of the left ventricle, whereas
temic filling pressure and decreased resistance to
the right coronary artery supplies most of the right
venous return raises the entire level of the venous
ventricle as well as the posterior part of the left ven-
return curve.
tricle in 80 to 90 per cent of people.
In response to the changes in both the venous return
Most of the coronary venous blood flow from the
curve and the cardiac output curve, the new equilib-
left ventricular muscle returns to the right atrium of
rium point in Figure 21-2 for cardiac output and
the heart by way of the coronary sinus—which is about
right atrial pressure is now point B, in contrast to the
75 per cent of the total coronary blood flow. And most
normal level at point A. Note especially that the right
of the coronary venous blood from the right ventricu-
atrial pressure has hardly changed, having risen only
lar muscle returns through small anterior cardiac veins
1.5 mm Hg. In fact, in a person with a strong heart,
that flow directly into the right atrium, not by way of
the right atrial pressure often falls below normal in
the coronary sinus. A very small amount of coronary
very heavy exercise because of the greatly increased
venous blood also flows back into the heart through
sympathetic stimulation of the heart during exercise.
very minute thebesian veins, which empty directly into
all chambers of the heart.
Coronary Circulation
Normal Coronary Blood Flow
About one third of all deaths in the affluent society of
The resting coronary blood flow in the human being
the Western world result from coronary artery disease,
averages about 225 ml/min, which is about 4 to 5 per
and almost all elderly people have at least some
cent of the total cardiac output.
impairment of the coronary artery circulation. For this
During strenuous exercise, the heart in the young
reason, understanding normal and pathological physi-
adult increases its cardiac output fourfold to seven-
ology of the coronary circulation is one of the most
fold, and it pumps this blood against a higher than
important subjects in medicine.
normal arterial pressure. Consequently, the work
250
Unit IV The Circulation
Epicardial coronary arteries
Cardiac
300
muscle
Subendocardial arterial plexus
200
Figure 21-5
Diagram of the epicardial, intramuscular, and subendocardial
100
coronary vasculature.
the special arrangement of the coronary vessels at dif-
0
Systole
Diastole
ferent depths in the heart muscle, showing on the outer
surface epicardial coronary arteries that supply most of
the muscle. Smaller, intramuscular arteries derived
from the epicardial arteries penetrate the muscle,
Figure 21-4
supplying the needed nutrients. Lying immediately
beneath the endocardium is a plexus of subendocar-
dial arteries. During systole, blood flow through the
Phasic flow of blood through the coronary capillaries of the human
left ventricle during cardiac systole and diastole (as extrapolated
subendocardial plexus of the left ventricle, where the
from measured flows in dogs).
intramuscular coronary vessels are compressed greatly
by ventricular muscle contraction, tends to be reduced.
output of the heart under severe conditions may
But the extra vessels of the subendocardial plexus nor-
increase sixfold to ninefold. At the same time, the
mally compensate for this. Later in the chapter, we will
coronary blood flow increases threefold to fourfold to
see that this peculiar difference between blood flow in
supply the extra nutrients needed by the heart. This
the epicardial and subendocardial arteries plays an
increase is not as much as the increase in workload,
important role in certain types of coronary ischemia.
which means that the ratio of energy expenditure by
the heart to coronary blood flow increases. Thus, the
“efficiency” of cardiac utilization of energy increases
Control of Coronary Blood Flow
to make up for the relative deficiency of coronary
blood supply.
Local Muscle Metabolism Is the Primary
Controller of Coronary Flow
Phasic Changes in Coronary Blood Flow During Systole and
Blood flow through the coronary system is regulated
Diastole—Effect of Cardiac Muscle Compression. Figure
mostly by local arteriolar vasodilation in response to
21-4 shows the changes in blood flow through the
cardiac muscle need for nutrition. That is, whenever
nutrient capillaries of the left ventricular coronary
the vigor of cardiac contraction is increased, regard-
system in milliliters per minute in the human heart
less of cause, the rate of coronary blood flow also
during systole and diastole, as extrapolated from
increases. Conversely, decreased heart activity is
experiments in lower animals. Note from this diagram
accompanied by decreased coronary flow. This local
that the coronary capillary blood flow in the left ven-
regulation of coronary blood flow is almost identical
tricle muscle falls to a low value during systole, which
to that occurring in many other tissues of the body,
is opposite to flow in vascular beds elsewhere in the
especially in the skeletal muscles all over the body.
body. The reason for this is strong compression of the
left ventricular muscle around the intramuscular
Oxygen Demand as a Major Factor in Local Coronary Blood Flow
vessels during systolic contraction.
Regulation. Blood flow in the coronaries usually is reg-
During diastole, the cardiac muscle relaxes and no
ulated almost exactly in proportion to the need of the
longer obstructs blood flow through the left ventricu-
cardiac musculature for oxygen. Normally, about 70
lar muscle capillaries, so that blood flows rapidly
per cent of the oxygen in the coronary arterial blood
during all of diastole.
is removed as the blood flows through the heart
Blood flow through the coronary capillaries of the
muscle. Because not much oxygen is left, very little
right ventricle also undergoes phasic changes during
additional oxygen can be supplied to the heart mus-
the cardiac cycle, but because the force of contraction
culature unless the coronary blood flow increases. For-
of the right ventricular muscle is far less than that of
tunately, the coronary blood flow does increase almost
the left ventricular muscle, the inverse phasic changes
in direct proportion to any additional metabolic con-
are only partial in contrast to those in the left ventric-
sumption of oxygen by the heart.
ular muscle.
However, the exact means by which increased
oxygen consumption causes coronary dilation has
Epicardial Versus Subendocardial Coronary Blood Flow—Effect
not been determined. It is speculated by many
of Intramyocardial Pressure. Figure
21-5 demonstrates
research workers that a decrease in the oxygen
Chapter 21
Muscle Blood Flow and Cardiac Output During Exercise
251
concentration in the heart causes vasodilator sub-
There is much more extensive sympathetic innerva-
stances to be released from the muscle cells and that
tion of the coronary vessels. In Chapter 60, we see that
these dilate the arterioles. A substance with great
the sympathetic transmitter substances norepineph-
vasodilator propensity is adenosine. In the presence of
rine and epinephrine can have either vascular con-
very low concentrations of oxygen in the muscle cells,
strictor or vascular dilator effects, depending on the
a large proportion of the cell’s ATP degrades to adeno-
presence or absence of constrictor or dilator receptors
sine monophosphate; then small portions of this are
in the blood vessel walls. The constrictor receptors are
further degraded and release adenosine into the tissue
called alpha receptors and the dilator receptors are
fluids of the heart muscle, with resultant increase in
called beta receptors. Both alpha and beta receptors
local coronary blood flow. After the adenosine causes
exist in the coronary vessels. In general, the epicardial
vasodilation, much of it is reabsorbed into the cardiac
coronary vessels have a preponderance of alpha recep-
cells to be reused.
tors, whereas the intramuscular arteries may have a
Adenosine is not the only vasodilator product
preponderance of beta receptors. Therefore, sympa-
that has been identified. Others include adenosine
thetic stimulation can, at least theoretically, cause
phosphate compounds, potassium ions, hydrogen
slight overall coronary constriction or dilation, but
ions, carbon dioxide, bradykinin, and, possibly,
usually constriction. In some people, the alpha vaso-
prostaglandins and nitric oxide.
constrictor effects seem to be disproportionately
Yet, difficulties with the vasodilator hypothesis exist.
severe, and these people can have vasospastic myocar-
First, pharmacologic agents that block or partially
dial ischemia during periods of excess sympathetic
block the vasodilator effect of adenosine do not
drive, often with resultant anginal pain.
prevent coronary vasodilation caused by increased
Metabolic factors—especially myocardial oxygen
heart muscle activity. Second, studies in skeletal
consumption—are the major controllers of myocardial
muscle have shown that continued infusion of adeno-
blood flow. Whenever the direct effects of nervous
sine maintains vascular dilation for only 1 to 3 hours,
stimulation alter the coronary blood flow in the wrong
and yet muscle activity still dilates the local blood
direction, the metabolic control of coronary flow
vessels even when the adenosine can no longer dilate
usually overrides the direct coronary nervous effects
them. Therefore, the other vasodilator mechanisms
within seconds.
listed above must be remembered.
Nervous Control of Coronary Blood Flow
Special Features of Cardiac
Stimulation of the autonomic nerves to the heart can
Muscle Metabolism
affect coronary blood flow both directly and indirectly.
The direct effects result from action of the nervous
The basic principles of cellular metabolism, discussed
transmitter substances acetylcholine from the vagus
in Chapters 67 through 72, apply to cardiac muscle the
nerves and norepinephrine and epinephrine from the
same as for other tissues, but there are some quantita-
sympathetic nerves on the coronary vessels them-
tive differences. Most important, under resting condi-
selves. The indirect effects result from secondary
tions, cardiac muscle normally consumes fatty acids to
changes in coronary blood flow caused by increased or
supply most of its energy instead of carbohydrates
decreased activity of the heart.
(about 70 per cent of the energy is derived from fatty
The indirect effects, which are mostly opposite to
acids). However, as is also true of other tissues, under
the direct effects, play a far more important role in
anaerobic or ischemic conditions, cardiac metabolism
normal control of coronary blood flow. Thus, sympa-
must call on anaerobic glycolysis mechanisms for
thetic stimulation, which releases norepinephrine and
energy. Unfortunately, glycolysis consumes tremen-
epinephrine, increases both heart rate and heart con-
dous quantities of the blood glucose and at the same
tractility as well as increases the rate of metabolism of
time forms large amounts of lactic acid in the cardiac
the heart. In turn, the increased metabolism of the
tissue, which is probably one of the causes of cardiac
heart sets off local blood flow regulatory mechanisms
pain in cardiac ischemic conditions, as discussed later
for dilating the coronary vessels, and the blood flow
in this chapter.
increases approximately in proportion to the meta-
As is true in other tissues, more than 95 per cent of
bolic needs of the heart muscle. In contrast, vagal stim-
the metabolic energy liberated from foods is used to
ulation, with its release of acetylcholine, slows the
form ATP in the mitochondria. This ATP in turn acts
heart and has a slight depressive effect on heart con-
as the conveyer of energy for cardiac muscular con-
tractility. These effects in turn decrease cardiac oxygen
traction and other cellular functions. In severe coro-
consumption and, therefore, indirectly constrict the
nary ischemia, the ATP degrades first to adenosine
coronary arteries.
diphosphate, then to adenosine monophosphate and
adenosine. Because the cardiac muscle cell membrane
Direct Effects of Nervous Stimuli on the Coronary Vasculature.
is slightly permeable to adenosine, much of this
The distribution of parasympathetic
(vagal) nerve
can diffuse from the muscle cells into the circulating
fibers to the ventricular coronary system is not very
blood.
great. However, the acetylcholine released by parasym-
The released adenosine is believed to be one of the
pathetic stimulation has a direct effect to dilate the coro-
substances that causes dilation of the coronary arteri-
nary arteries.
oles during coronary hypoxia, as discussed earlier.
252
Unit IV The Circulation
However, loss of adenosine also has a serious cellular
the flowing blood. Because the plaque presents an
consequence. Within as little as 30 minutes of severe
unsmooth surface, blood platelets adhere to it,
coronary ischemia, as occurs after a myocardial infarct,
fibrin is deposited, and red blood cells become
about one half of the adenine base can be lost from
entrapped to form a blood clot that grows until
the affected cardiac muscle cells. Furthermore, this loss
it occludes the vessel. Or, occasionally, the
can be replaced by new synthesis of adenine at a rate
clot breaks away from its attachment on the
of only 2 per cent per hour. Therefore, once a serious
atherosclerotic plaque and flows to a more
bout of coronary ischemia has persisted for 30 or more
peripheral branch of the coronary arterial tree,
minutes, relief of the ischemia may be too late to save
where it blocks the artery at that point. A
the lives of the cardiac cells. This almost certainly is
thrombus that flows along the artery in this way
one of the major causes of cardiac cellular death
and occludes the vessel more distally is called a
during myocardial ischemia.
coronary embolus.
2. Many clinicians believe that local muscular spasm
of a coronary artery also can occur. The spasm
Ischemic Heart Disease
might result from direct irritation of the smooth
muscle of the arterial wall by the edges of an
The most common cause of death in Western culture
arteriosclerotic plaque, or it might result from
is ischemic heart disease, which results from insuffi-
local nervous reflexes that cause excess coronary
cient coronary blood flow. About 35 per cent of people
vascular wall contraction. The spasm may then
in the United States die of this cause. Some deaths
lead to secondary thrombosis of the vessel.
occur suddenly as a result of acute coronary occlusion
or fibrillation of the heart, whereas other deaths occur
Lifesaving Value of Collateral Circulation in the Heart. The
slowly over a period of weeks to years as a result of
degree of damage to the heart muscle caused either by
progressive weakening of the heart pumping process.
slowly developing atherosclerotic constriction of the
In this chapter, we discuss acute coronary ischemia
coronary arteries or by sudden coronary occlusion is
caused by acute coronary occlusion and myocardial
determined to a great extent by the degree of collat-
infarction. In Chapter 22, we discuss congestive heart
eral circulation that has already developed or that can
failure, the most frequent cause of which is slowly
open within minutes after the occlusion.
increasing coronary ischemia and weakening of the
In a normal heart, almost no large communications
cardiac muscle.
exist among the larger coronary arteries. But many
anastomoses do exist among the smaller arteries
Atherosclerosis as a Cause of Ischemic Heart Disease. The
sized 20 to 250 micrometers in diameter, as shown in
most frequent cause of diminished coronary blood
Figure 21-6.
flow is atherosclerosis. The atherosclerotic process is
When a sudden occlusion occurs in one of the larger
discussed in connection with lipid metabolism in
coronary arteries, the small anastomoses begin to
Chapter 68. Briefly, this process is the following.
In people who have genetic predisposition to ath-
erosclerosis, or in people who eat excessive quantities
of cholesterol and have a sedentary lifestyle, large
Artery
quantities of cholesterol gradually become deposited
beneath the endothelium at many points in arteries
throughout the body. Gradually, these areas of deposit
are invaded by fibrous tissue and frequently become
Vein
calcified. The net result is the development of athero-
sclerotic plaques that actually protrude into the vessel
lumens and either block or partially block blood flow.
A common site for development of atherosclerotic
plaques is the first few centimeters of the major coro-
nary arteries.
Acute Coronary Occlusion
Acute occlusion of a coronary artery most frequently
occurs in a person who already has underlying ath-
erosclerotic coronary heart disease but almost never
in a person with a normal coronary circulation. Acute
Artery
occlusion can result from any one of several effects,
two of which are the following:
1. The atherosclerotic plaque can cause a local blood
clot called a thrombus, which in turn occludes
Vein
the artery. The thrombus usually occurs where the
Figure 21-6
arteriosclerotic plaque has broken through the
endothelium, thus coming in direct contact with
Minute anastomoses in the normal coronary arterial system.
Chapter 21
Muscle Blood Flow and Cardiac Output During Exercise
253
dilate within seconds. But the blood flow through these
Subendocardial Infarction. The subendocardial muscle
minute collaterals is usually less than one half that
frequently becomes infarcted even when there is no
needed to keep alive most of the cardiac muscle that
evidence of infarction in the outer surface portions of
they now supply; the diameters of the collateral vessels
the heart. The reason for this is that the subendocar-
do not enlarge much more for the next 8 to 24 hours.
dial muscle has extra difficulty obtaining adequate
But then collateral flow does begin to increase, dou-
blood flow because the blood vessels in the subendo-
bling by the second or third day and often reaching
cardium are intensely compressed by systolic contrac-
normal or almost normal coronary flow within about
tion of the heart, as explained earlier. Therefore, any
1 month. Because of these developing collateral chan-
condition that compromises blood flow to any area of
nels, many patients recover almost completely from
the heart usually causes damage first in the subendo-
various degrees of coronary occlusion when the area
cardial regions, and the damage then spreads outward
of muscle involved is not too great.
toward the epicardium.
When atherosclerosis constricts the coronary arter-
ies slowly over a period of many years rather than
Causes of Death After Acute
suddenly, collateral vessels can develop at the same
Coronary Occlusion
time while the atherosclerosis becomes more and
more severe. Therefore, the person may never experi-
The most common causes of death after acute
ence an acute episode of cardiac dysfunction. But,
myocardial infarction are (1) decreased cardiac output;
eventually, the sclerotic process develops beyond the
(2) damming of blood in the pulmonary blood vessels
limits of even the collateral blood supply to provide
and then death resulting from pulmonary edema; (3)
the needed blood flow, and sometimes the collateral
fibrillation of the heart; and, occasionally, (4) rupture of
blood vessels themselves develop atherosclerosis.
the heart.
When this occurs, the heart muscle becomes severely
limited in its work output, often so much so that the
Decreased Cardiac Output—Systolic Stretch and Cardiac
heart cannot pump even normally required amounts
Shock. When some of the cardiac muscle fibers are not
of blood flow. This is one of the most common causes
functioning and others are too weak to contract with
of the cardiac failure that occurs in vast numbers of
great force, the overall pumping ability of the affected
older people.
ventricle is proportionately depressed. Indeed, the
overall pumping strength of the infarcted heart is often
Myocardial Infarction
decreased more than one might expect because of a
Immediately after an acute coronary occlusion, blood
phenomenon called systolic stretch, shown in Figure
flow ceases in the coronary vessels beyond the occlu-
21-7. That is, when the normal portions of the ventric-
sion except for small amounts of collateral flow from
ular muscle contract, the ischemic portion of the
surrounding vessels. The area of muscle that has either
muscle, whether this be dead or simply nonfunctional,
zero flow or so little flow that it cannot sustain cardiac
instead of contracting is forced outward by the pres-
muscle function is said to be infarcted. The overall
sure that develops inside the ventricle. Therefore,
process is called a myocardial infarction.
Soon after the onset of the infarction, small amounts
of collateral blood begin to seep into the infarcted area,
and this, combined with progressive dilation of local
blood vessels, causes the area to become overfilled with
stagnant blood. Simultaneously the muscle fibers use
the last vestiges of the oxygen in the blood, causing
the hemoglobin to become totally de-oxygenated.
Therefore, the infarcted area takes on a bluish-brown
Normal contraction
hue, and the blood vessels of the area appear to be
engorged despite lack of blood flow. In later stages, the
vessel walls become highly permeable and leak fluid;
Non-functional
muscle
the local muscle tissue becomes edematous, and the
cardiac muscle cells begin to swell because of dimin-
ished cellular metabolism. Within a few hours of almost
no blood supply, the cardiac muscle cells die.
Cardiac muscle requires about
1.3 milliliters of
oxygen per 100 grams of muscle tissue per minute just
to remain alive. This is in comparison with about 8 mil-
liliters of oxygen per
100 grams delivered to the
Systolic stretch
normal resting left ventricle each minute. Therefore, if
there is even 15 to 30 per cent of normal resting
coronary blood flow, the muscle will not die. In the
central portion of a large infarct, however, where
Figure 21-7
there is almost no collateral blood flow, the muscle
does die.
Systolic stretch in an area of ischemic cardiac muscle.
254
Unit IV The Circulation
much of the pumping force of the ventricle is dissi-
increases the irritability of the cardiac
pated by bulging of the area of nonfunctional cardiac
musculature and, therefore, its likelihood of
muscle.
fibrillating.
When the heart becomes incapable of contracting
2.
Ischemia of the muscle causes an “injury current,”
with sufficient force to pump enough blood into the
which is described in Chapter 12 in relation to
peripheral arterial tree, cardiac failure and death of
electrocardiograms in patients with acute
peripheral tissues ensue as a result of peripheral
myocardial infarction. That is, the ischemic
ischemia. This condition is called coronary shock, car-
musculature often cannot completely repolarize
diogenic shock, cardiac shock, or low cardiac output
its membranes after a heart beat, so that the
failure. It is discussed more fully in the next chapter.
external surface of this muscle remains negative
Cardiac shock almost always occurs when more than
with respect to normal cardiac muscle membrane
40 per cent of the left ventricle is infarcted. And death
potential elsewhere in the heart. Therefore,
occurs in about 85 per cent of patients once they
electric current flows from this ischemic area of
develop cardiac shock.
the heart to the normal area and can elicit
abnormal impulses that can cause fibrillation.
Damming of Blood in the Body’s Venous System. When the
3.
Powerful sympathetic reflexes often develop after
heart is not pumping blood forward, it must be
massive infarction, principally because the heart
damming blood in the atria and in the blood vessels
does not pump an adequate volume of blood into
of the lungs or in the systemic circulation. This leads
the arterial tree. The sympathetic stimulation also
to increased capillary pressures, particularly in the
increases irritability of the cardiac muscle and
lungs.
thereby predisposes to fibrillation.
This damming of blood in the veins often causes
4.
Cardiac muscle weakness caused by the
little difficulty during the first few hours after myocar-
myocardial infarction often causes the ventricle to
dial infarction. Instead, symptoms develop a few days
dilate excessively. This increases the pathway length
later for the following reason: The acutely diminished
for impulse conduction in the heart and frequently
cardiac output leads to diminished blood flow to the
causes abnormal conduction pathways all the way
kidneys. Then, for reasons that are discussed in
around the infarcted area of the cardiac muscle.
Chapter 22. the kidneys fail to excrete enough urine.
Both of these effects predispose to development
This adds progressively to the total blood volume and,
of circus movements because, as discussed in
therefore, leads to congestive symptoms. Consequently,
Chapter 13, excess prolongation of conduction
many patients who seemingly are getting along well
pathways in the ventricles allows impulses to re-
during the first few days after onset of heart failure will
enter muscle that is already recovering from
suddenly develop acute pulmonary edema and often
refractoriness, thereby initiating a “circus
will die within a few hours after appearance of the
movement” cycle of new excitation and causing
initial pulmonary symptoms.
the process to continue on and on.
Fibrillation of the Ventricles After Myocardial Infarction.
Rupture of the Infarcted Area. During the first day or so
Many people who die of coronary occlusion die
after an acute infarct, there is little danger of rupture
because of sudden ventricular fibrillation. The ten-
of the ischemic portion of the heart, but a few days
dency to develop fibrillation is especially great after a
later, the dead muscle fibers begin to degenerate, and
large infarction, but fibrillation can sometimes occur
the heart wall becomes stretched very thin. When this
after small occlusions as well. Indeed, some patients
happens, the dead muscle bulges outward severely
with chronic coronary insufficiency die suddenly from
with each heart contraction, and this systolic stretch
fibrillation without any acute infarction.
becomes greater and greater until finally the heart rup-
There are two especially dangerous periods after
tures. In fact, one of the means used in assessing
coronary infarction during which fibrillation is most
progress of severe myocardial infarction is to record
likely to occur. The first is during the first 10 minutes
by cardiac imaging (i.e., x-rays) whether the degree of
after the infarction occurs. Then there is a short period
systolic stretch is worsening.
of relative safety, followed by a secondary period of
When a ventricle does rupture, loss of blood into the
cardiac irritability beginning 1 hour or so later and
pericardial space causes rapid development of cardiac
lasting for another few hours. Fibrillation can also
tamponade—that is, compression of the heart from the
occur many days after the infarct but less likely so.
outside by blood collecting in the pericardial cavity.
At least four factors enter into the tendency for the
Because of this compression of the heart, blood cannot
heart to fibrillate:
flow into the right atrium, and the patient dies of sud-
1. Acute loss of blood supply to the cardiac muscle
denly decreased cardiac output.
causes rapid depletion of potassium from the
ischemic musculature. This also increases the
potassium concentration in the extracellular
Stages of Recovery from Acute
fluids surrounding the cardiac muscle fibers.
Myocardial Infarction
Experiments in which potassium has been injected
into the coronary system have demonstrated that
The upper left part of Figure 21-8 shows the effects of
an elevated extracellular potassium concentration
acute coronary occlusion in a patient with a small area
Chapter 21
Muscle Blood Flow and Cardiac Output During Exercise
255
Mild
heart recovers either partially or almost completely
Mild
ischemia
ischemia
within a few months.
Non-
Non-
Value of Rest in Treating Myocardial Infarction. The degree
functional
functional
of cardiac cellular death is determined by the degree
of ischemia and the workload on the heart muscle.
When the workload is greatly increased, such as during
exercise, in severe emotional strain, or as a result of
Dead fibers
fatigue, the heart needs increased oxygen and other
Nonfunctional
nutrients for sustaining its life. Furthermore, anasto-
motic blood vessels that supply blood to ischemic
areas of the heart must also still supply the areas of
the heart that they normally supply. When the heart
becomes excessively active, the vessels of the normal
musculature become greatly dilated. This allows most
Dead fibers
Fibrous tissue
of the blood flowing into the coronary vessels to flow
Figure 21-8
through the normal muscle tissue, thus leaving little
blood to flow through the small anastomotic channels
Top, Small and large areas of coronary ischemia. Bottom, Stages
into the ischemic area, so that the ischemic condition
of recovery from myocardial infarction.
worsens. This condition is called the “coronary steal”
syndrome. Consequently, one of the most important
factors in the treatment of a patient with myocardial
of muscle ischemia; to the right is shown a heart with
infarction is observance of absolute body rest during
a large area of ischemia. When the area of ischemia is
the recovery process.
small, little or no death of the muscle cells may occur,
but part of the muscle often does become temporarily
nonfunctional because of inadequate nutrition to
Function of the Heart After Recovery
support muscle contraction.
from Myocardial Infarction
When the area of ischemia is large, some of the
muscle fibers in the center of the area die rapidly,
Occasionally, a heart that has recovered from a large
within 1 to 3 hours where there is total cessation of
myocardial infarction returns almost to full functional
coronary blood supply. Immediately around the dead
capability, but more frequently its pumping capability
area is a nonfunctional area, with failure of contrac-
is permanently decreased below that of a healthy
tion and usually failure of impulse conduction. Then,
heart. This does not mean that the person is necessar-
extending circumferentially around the nonfunctional
ily a cardiac invalid or that the resting cardiac output
area is an area that is still contracting but weakly so
is depressed below normal, because the normal heart
because of mild ischemia.
is capable of pumping 300 to 400 per cent more blood
per minute than the body requires during rest—that is,
Replacement of Dead Muscle by Scar Tissue. In the lower
a normal person has a “cardiac reserve” of 300 to 400
part of Figure 21-8, the various stages of recovery after
per cent. Even when the cardiac reserve is reduced to
a large myocardial infarction are shown. Shortly after
as little as 100 per cent, the person can still perform
the occlusion, the muscle fibers in the center of the
normal activity of a quiet, restful type but not strenu-
ischemic area die. Then, during the ensuing days, this
ous exercise that would overload the heart.
area of dead fibers becomes bigger because many of
the marginal fibers finally succumb to the prolonged
Pain in Coronary Heart Disease
ischemia. At the same time, because of enlargement of
collateral arterial channels supplying the outer rim of
Normally, a person cannot “feel” his or her heart, but
the infarcted area, much of the nonfunctional muscle
ischemic cardiac muscle often does cause pain sensa-
recovers. After a few days to three weeks, most of
tion—sometimes severe pain. Exactly what causes this
the nonfunctional muscle becomes functional again or
pain is not known, but it is believed that ischemia
dies—one or the other. In the meantime, fibrous tissue
causes the muscle to release acidic substances, such as
begins developing among the dead fibers because
lactic acid, or other pain-promoting products, such as
ischemia can stimulate growth of fibroblasts and
histamine, kinins, or cellular proteolytic enzymes, that
promote development of greater than normal quanti-
are not removed rapidly enough by the slowly moving
ties of fibrous tissue. Therefore, the dead muscle tissue
coronary blood flow. The high concentrations of these
is gradually replaced by fibrous tissue. Then, because
abnormal products then stimulate pain nerve endings
it is a general property of fibrous tissue to undergo
in the cardiac muscle, sending pain impulses through
progressive contraction and dissolution, the fibrous
sensory afferent nerve fibers into the central nervous
scar may grow smaller over a period of several months
system.
to a year.
Finally, the normal areas of the heart gradually
Angina Pectoris
hypertrophy to compensate at least partially for the
In most people who develop progressive constriction
lost dead cardiac musculature. By these means, the
of their coronary arteries, cardiac pain, called angina
256
Unit IV The Circulation
pectoris, begins to appear whenever the load on the
procedure may provide the patient with normal sur-
heart becomes too great in relation to the available
vival expectation. Conversely, if the heart has already
coronary blood flow. This pain is usually felt beneath
been severely damaged, the bypass procedure is likely
the upper sternum over the heart, and in addition it is
to be of little value.
often referred to distant surface areas of the body,
most commonly to the left arm and left shoulder but
Coronary Angioplasty. Since the 1980s, a procedure has
also frequently to the neck and even to the side of the
been used to open partially blocked coronary vessels
face. The reason for this distribution of pain is that the
before they become totally occluded. This procedure,
heart originates during embryonic life in the neck, as
called coronary artery angioplasty, is the following:
do the arms. Therefore, both the heart and these
A small balloon-tipped catheter, about 1 millimeter in
surface areas of the body receive pain nerve fibers
diameter, is passed under radiographic guidance into
from the same spinal cord segments.
the coronary system and pushed through the partially
Most people who have chronic angina pectoris feel
occluded artery until the balloon portion of the
pain when they exercise or when they experience
catheter straddles the partially occluded point. Then
emotions that increase metabolism of the heart or
the balloon is inflated with high pressure, which
temporarily constrict the coronary vessels because of
markedly stretches the diseased artery. After this pro-
sympathetic vasoconstrictor nerve signals. The pain
cedure is performed, the blood flow through the vessel
usually lasts for only a few minutes. However, some
often increases threefold to fourfold, and more than
patients have such severe and lasting ischemia that the
three quarters of the patients who undergo the proce-
pain is present all the time. The pain is frequently
dure are relieved of the coronary ischemic symptoms
described as hot, pressing, and constricting; it is of such
for at least several years, although many of the patients
quality that it usually makes the patient stop all unnec-
still eventually require coronary bypass surgery.
essary body activity and come to a complete state of
Still newer procedures for opening atherosclerotic
rest.
coronary arteries are constantly in experimental
development. One of these employs a laser beam from
Treatment with Drugs. Several vasodilator drugs, when
the tip of a coronary artery catheter aimed at the
administered during an acute anginal attack, can
atherosclerotic lesion. The laser literally dissolves the
often give immediate relief from the pain. Commonly
lesion without substantially damaging the rest of
used vasodilators are nitroglycerin and other nitrate
the arterial wall.
drugs.
Another development has been a minute metal
A second class of drugs that are used for prolonged
“sleeve” placed inside a coronary artery dilated by
treatment of angina pectoris is the beta blockers,
angioplasty to hold the artery open, thus preventing its
such as propranolol. These drugs block sympathetic
restenosis.
beta adrenergic receptors, which prevents sympathetic
enhancement of heart rate and cardiac metabolism
during exercise or emotional episodes. Therefore,
References
therapy with a beta blocker decreases the need
of the heart for extra metabolic oxygen during
Casscells W, Nahai M, Willerson JT: Vulnerable atheroscle-
stressful conditions. For obvious reasons, this can also
rotic plaque: a multifocal disease. Circulation 107:2072,
reduce the number of anginal attacks as well as their
2003.
severity.
Cohn PF, Fox KM, Daly C: Silent myocardial ischemia.
Circulation 108:1263, 2003.
Dalal H, Evans PH, Campbell JL: Recent developments in
Surgical Treatment of
secondary prevention and cardiac rehabilitation after
acute myocardial infarction. BMJ 328:693, 2004.
Coronary Disease
Freedman SB, Isner JM: Therapeutic angiogenesis for coro-
nary artery disease. Ann Intern Med 136:54, 2002.
Aortic-Coronary Bypass Surgery. In many patients with
Gehlbach BK, Geppert E: The pulmonary manifestations of
coronary ischemia, the constricted areas of the coro-
left heart failure. Chest 125:669, 2004.
nary arteries are located at only a few discrete points
Guyton AC, Jones CE, Coleman TG: Circulatory Pathology:
blocked by atherosclerotic disease, and the coronary
Cardiac Output and Its Regulation. Philadelphia: WB
vessels elsewhere are normal or almost normal. A sur-
Saunders Co, 1973.
gical procedure was developed in the 1960s, called
Hao H, Gabbiani J, Bochaton-Piallat M: Arterial smooth
aortic-coronary bypass, for removing a section of a
muscle cell heterogeneity: implications for atherosclerosis
subcutaneous vein from an arm or leg and then graft-
and restenosis development. Arterioscler Thromb Vasc
Biol 23:1510, 2003.
ing this vein from the root of the aorta to the side of
Hester RL, Hammer LW: Venular-arteriolar communication
a peripheral coronary artery beyond the atheroscle-
in the regulation of blood flow. Am J Physiol 282:R1280,
rotic blockage point. One to five such grafts are usually
2002.
performed, each of which supplies a peripheral coro-
Hochman JS: Cardiogenic shock complicating acute myocar-
nary artery beyond a block.
dial infarction: expanding the paradigm. Circulation 107:
Anginal pain is relieved in most patients. Also, in
2998, 2003.
patients whose hearts have not become too severely
Joyner MJ, Dietz NM: Sympathetic vasodilation in human
damaged before the operation, the coronary bypass
muscle. Acta Physiol Scand 177:329, 2003.
Chapter 21
Muscle Blood Flow and Cardiac Output During Exercise
257
Koerselman J, van der Graaf Y, de Jaegere PP, Grobbee DE:
Ridker PM: Clinical application of C-reactive protein for
Coronary collaterals: an important and underexposed
cardiovascular disease detection and prevention. Circula-
aspect of coronary artery disease. Circulation 107:2507,
tion 107:363, 2003.
2003.
Tsai AG, Johnson PC, Intaglietta M: Oxygen gradients in the
Libby P: Inflammation in atherosclerosis. Nature 420:868,
microcirculation. Physiol Rev 83:933, 2003.
2002.
Yellon DM, Downey JM: Preconditioning the myocardium:
Richardson RS: Oxygen transport and utilization: an inte-
from cellular physiology to clinical cardiology. Physiol Rev
gration of the muscle systems. Adv Physiol Educ 27:183,
83:1113, 2003.
2003.
C H A P T E R
2
2
Cardiac Failure
One of the most important ailments that must be
treated by the physician is cardiac failure (“heart
failure”). This can result from any heart condition
that reduces the ability of the heart to pump blood.
The cause usually is decreased contractility of the
myocardium resulting from diminished coronary
blood flow. However, failure can also be caused by
damaged heart valves, external pressure around the
heart, vitamin B deficiency, primary cardiac muscle disease, or any other abnor-
mality that makes the heart a hypoeffective pump. In this chapter, we discuss
mainly cardiac failure caused by ischemic heart disease resulting from partial
blockage of the coronary blood vessels. In Chapter 23, we discuss valvular and
congenital heart disease.
Definition of Cardiac Failure. The term “cardiac failure” means simply failure of
the heart to pump enough blood to satisfy the needs of the body.
Dynamics of the Circulation in Cardiac Failure
Acute Effects of Moderate Cardiac Failure
If a heart suddenly becomes severely damaged, such as by myocardial infarc-
tion, the pumping ability of the heart is immediately depressed. As a result, two
main effects occur: (1) reduced cardiac output and (2) damming of blood in the
veins, resulting in increased venous pressure.
The progressive changes in heart pumping effectiveness at different times
after an acute myocardial infarction are shown graphically in Figure 22-1. The
top curve of this figure shows a normal cardiac output curve. Point A on this
curve is the normal operating point, showing a normal cardiac output under
resting conditions of 5 L/min and a right atrial pressure of 0 mm Hg.
Immediately after the heart becomes damaged, the cardiac output curve
becomes greatly lowered, falling to the lowest curve at the bottom of the graph.
Within a few seconds, a new circulatory state is established at point B rather
than point A, illustrating that the cardiac output has fallen to 2 L/min, about
two-fifths normal, whereas the right atrial pressure has risen to +4 mm Hg
because venous blood returning to the heart from the body is dammed up in
the right atrium. This low cardiac output is still sufficient to sustain life for
perhaps a few hours, but it is likely to be associated with fainting. Fortunately,
this acute stage usually lasts for only a few seconds because sympathetic nerve
reflexes occur immediately and compensate, to a great extent, for the damaged
heart, as follows.
Compensation for Acute Cardiac Failure by Sympathetic Nervous Reflexes. When the
cardiac output falls precariously low, many of the circulatory reflexes discussed
in Chapter 18 are immediately activated. The best known of these is the barore-
ceptor reflex, which is activated by diminished arterial pressure. It is probable
that the chemoreceptor reflex, the central nervous system ischemic response,
and even reflexes that originate in the damaged heart also contribute to activat-
ing the sympathetic nervous system. But whatever the reflexes might be, the
sympathetics become strongly stimulated within a few seconds, and the
258
Chapter 22
Cardiac Failure
259
The sympathetic reflexes become maximally devel-
oped in about 30 seconds. Therefore, a person who has
Normal heart
a sudden, moderate heart attack might experience
Partially recovered heart
nothing more than cardiac pain and a few seconds of
fainting. Shortly thereafter, with the aid of the sympa-
Damaged heart + sympathetic stimulation
15
thetic reflex compensations, the cardiac output may
Acutely damaged heart
return to a level adequate to sustain the person if he
or she remains quiet, although the pain might persist.
10
Chronic Stage of Failure—Fluid
Retention Helps to Compensate
Cardiac Output
A
D
5
After the first few minutes of an acute heart attack, a
C
prolonged semi-chronic state begins, characterized
B
mainly by two events: (1) retention of fluid by the
kidneys and (2) varying degrees of recovery of the
0
-4
-2
0
+2
+4
+6
+8
+10 +12 +14
heart itself over a period of weeks to months, as illus-
Right atrial pressure (mm Hg)
trated by the light green curve in Figure 22-1; this was
also discussed in Chapter 21.
Renal Retention of Fluid and Increase in Blood
Figure 22-1
Volume Occur for Hours to Days
Progressive changes in the cardiac output curve after acute
A low cardiac output has a profound effect on renal
myocardial infarction. Both the cardiac output and right atrial pres-
function, sometimes causing anuria when the cardiac
sure change progressively from point A to point D (illustrated by
output falls to one-half to two-thirds normal. In
the black line) over a period of seconds, minutes, days, and
general, the urine output remains reduced below
weeks.
normal as long as the cardiac output and arterial pres-
sure remain significantly less than normal, and urine
output usually does not return all the way to normal
after an acute heart attack until the cardiac output and
parasympathetic nervous signals to the heart become
arterial pressure rise either all the way back to normal
reciprocally inhibited at the same time.
or almost to normal.
Strong sympathetic stimulation has two major
effects on the circulation: first on the heart itself,
and second on the peripheral vasculature. If all the
Moderate Fluid Retention in Cardiac Failure Can Be Beneficial.
ventricular musculature is diffusely damaged but is
Many cardiologists formerly considered fluid retention
still functional, sympathetic stimulation strengthens
always to have a detrimental effect in cardiac failure.
this damaged musculature. If part of the muscle is non-
But it is now known that a moderate increase in body
functional and part of it is still normal, the normal
fluid and blood volume is an important factor in
muscle is strongly stimulated by sympathetic stimula-
helping to compensate for the diminished pumping
tion, in this way partially compensating for the non-
ability of the heart by increasing the venous return.
functional muscle. Thus, the heart, one way or another,
The increased blood volume increases venous return
becomes a stronger pump. This effect is also demon-
in two ways: First, it increases the mean systemic filling
strated in Figure 22-1, showing after sympathetic com-
pressure, which increases the pressure gradient for
pensation about twofold elevation of the very low
causing venous flow of blood toward the heart. Second,
cardiac output curve.
it distends the veins, which reduces the venous resist-
Sympathetic stimulation also increases venous
ance and allows even more ease of flow of blood to the
return because it increases the tone of most of the
heart.
blood vessels of the circulation, especially the veins,
If the heart is not too greatly damaged, this
raising the mean systemic filling pressure to 12 to 14
increased venous return can often fully compensate
mm Hg, almost 100 per cent above normal. As dis-
for the heart’s diminished pumping ability—enough in
cussed in Chapter 20, this increased filling pressure
fact that even when the heart’s pumping ability is
greatly increases the tendency for blood to flow from
reduced to as low as 40 to 50 per cent of normal, the
the veins back into the heart. Therefore, the damaged
increased venous return can often cause an entirely
heart becomes primed with more inflowing blood than
normal cardiac output as long as the person remains
usual, and the right atrial pressure rises still further,
in a quiet resting state.
which helps the heart to pump still larger quantities of
When the heart’s pumping capability is reduced
blood. Thus, in Figure 22-1, the new circulatory state
still more, blood flow to the kidneys finally becomes
is depicted by point C, showing a cardiac output of
too low for the kidneys to excrete enough salt and
4.2 L/min and a right atrial pressure of 5 mm Hg.
water to equal salt and water intake. Therefore, fluid
260
Unit IV The Circulation
retention begins and continues indefinitely, unless
thetic stimulation gradually abates toward normal for
major therapeutic procedures are used to prevent this.
the following reasons: The partial recovery of the
Furthermore, because the heart is already pumping at
heart can elevate the cardiac output curve the same as
its maximum pumping capacity, this excess fluid no
sympathetic stimulation can. Therefore, as the heart
longer has a beneficial effect on the circulation. Instead,
recovers even slightly, the fast pulse rate, cold skin,
severe edema develops throughout the body, which
and pallor resulting from sympathetic stimulation in
can be very detrimental in itself and can lead to death.
the acute stage of cardiac failure gradually disappear.
Detrimental Effects of Excess Fluid Retention in Severe Cardiac
Summary of the Changes That Occur
Failure. In contrast to the beneficial effects of moder-
After Acute Cardiac Failure—
ate fluid retention in cardiac failure, in severe failure
extreme excesses of fluid can have serious physiologi-
“Compensated Heart Failure”
cal consequences. They include (1) overstretching of
the heart, thus weakening the heart still more; (2)
To summarize the events discussed in the past few sec-
filtration of fluid into the lungs, causing pulmonary
tions describing the dynamics of circulatory changes
edema and consequent deoxygenation of the blood;
after an acute, moderate heart attack, we can divide
and (3) development of extensive edema in most parts
the stages into (1) the instantaneous effect of the
of the body. These detrimental effects of excessive fluid
cardiac damage; (2) compensation by the sympathetic
are discussed in later sections of this chapter.
nervous system, which occurs mainly within the first 30
seconds to 1 minute; and (3) chronic compensations
Recovery of the Myocardium After
resulting from partial heart recovery and renal reten-
Myocardial Infarction
tion of fluid. All these changes are shown graphically
After a heart becomes suddenly damaged as a result
by the black curve in Figure 22-1. The progression of
of myocardial infarction, the natural reparative
this curve shows the normal state of the circulation
processes of the body begin immediately to help
(point A), the state a few seconds after the heart attack
restore normal cardiac function. For instance, a new
but before sympathetic reflexes have occurred (point
collateral blood supply begins to penetrate the periph-
B), the rise in cardiac output toward normal caused
eral portions of the infarcted area of the heart, often
by sympathetic stimulation (point C), and final return
causing much of the heart muscle in the fringe areas
of the cardiac output almost exactly to normal after
to become functional again. Also, the undamaged
several days to several weeks of partial cardiac recov-
portion of the heart musculature hypertrophies, in this
ery and fluid retention (point D). This final state is
way offsetting much of the cardiac damage.
called compensated heart failure.
The degree of recovery depends on the type of
Compensated Heart Failure. Note especially in Figure
cardiac damage, and it varies from no recovery to
22-1 that the maximum pumping ability of the partly
almost complete recovery. After acute myocardial
recovered heart, as depicted by the plateau level of the
infarction, the heart ordinarily recovers rapidly during
light green curve, is still depressed to less than one-half
the first few days and weeks and achieves most of its
normal. This demonstrates that an increase in right
final state of recovery within 5 to 7 weeks, although
atrial pressure can maintain the cardiac output at a
mild degrees of additional recovery can continue for
normal level despite continued weakness of the heart.
months.
Thus many people, especially older people, have
normal resting cardiac outputs but mildly to moder-
The Cardiac Output Curve After Partial Recovery. Figure 22-1
ately elevated right atrial pressures because of various
shows function of the partially recovered heart a week
degrees of “compensated heart failure.” These persons
or so after acute myocardial infarction. By this time,
may not know that they have cardiac damage because
considerable fluid has been retained in the body and
the damage often has occurred a little at a time, and
the tendency for venous return has increased
the compensation has occurred concurrently with the
markedly as well; therefore, the right atrial pressure
progressive stages of damage.
has risen even more. As a result, the state of the cir-
When a person is in compensated heart failure, any
culation is now changed from point C to point D, which
attempt to perform heavy exercise usually causes
shows a normal cardiac output of 5 L/min but right
immediate return of the symptoms of acute failure
atrial pressure increased to 6 mm Hg.
because the heart is not able to increase its pumping
Because the cardiac output has returned to normal,
capacity to the levels required for the exercise. There-
renal output of fluid also returns to normal, and no
fore, it is said that the cardiac reserve is reduced in
further fluid retention occurs, except that the retention
compensated heart failure. This concept of cardiac
of fluid that has already occurred continues to maintain
reserve is discussed more fully later in the chapter.
moderate excesses of fluid. Therefore, except for the
high right atrial pressure represented by point D in
this figure, the person now has essentially normal car-
Dynamics of Severe Cardiac Failure—
diovascular dynamics as long as he or she remains at
Decompensated Heart Failure
rest.
If the heart recovers to a significant extent and if
If the heart becomes severely damaged, no amount of
adequate fluid volume has been retained, the sympa-
compensation, either by sympathetic nervous reflexes
Chapter 22
Cardiac Failure
261
or by fluid retention, can make the excessively weak-
circulation continues to rise; this forces progressively
ened heart pump a normal cardiac output. As a con-
increasing quantities of blood from the person’s
sequence, the cardiac output cannot rise high enough
peripheral veins into the right atrium, thus increasing
to make the kidneys excrete normal quantities of fluid.
the right atrial pressure. After 1 day or so, the state of
Therefore, fluid continues to be retained, the person
the circulation changes in Figure 22-2 from point B to
develops more and more edema, and this state of
point C—the right atrial pressure rising to 7 mm Hg
events eventually leads to death. This is called decom-
and the cardiac output to 4.2 L/min. Note again that
pensated heart failure. Thus, the main cause of decom-
the cardiac output is still not high enough to cause
pensated heart failure is failure of the heart to pump
normal renal output of fluid; therefore, fluid continues
sufficient blood to make the kidneys excrete daily the
to be retained. After another day or so, the right atrial
necessary amounts of fluid.
pressure rises to 9 mm Hg, and the circulatory state
becomes that depicted by point D. Still, the cardiac
Graphical Analysis of Decompensated Heart Failure. Figure
output is not enough to establish normal fluid
22-2 shows greatly depressed cardiac output at differ-
balance.
ent times (points A to F) after the heart has become
After another few days of fluid retention, the right
severely weakened. Point A on this curve represents
atrial pressure has risen still further, but by now,
the approximate state of the circulation before any
cardiac function is beginning to decline toward a lower
compensation has occurred, and point B, the state a
level. This decline is caused by overstretch of the heart,
few minutes later after sympathetic stimulation has
edema of the heart muscle, and other factors that
compensated as much as it can but before fluid reten-
diminish the heart’s pumping performance. It is
tion has begun. At this time, the cardiac output has
now clear that further retention of fluid will be more
risen to 4 L/min and the right atrial pressure has risen
detrimental than beneficial to the circulation. Yet the
to 5 mm Hg. The person appears to be in reasonably
cardiac output still is not high enough to bring about
good condition, but this state will not remain stable
normal renal function, so that fluid retention not
because the cardiac output has not risen high enough
only continues but accelerates because of the falling
to cause adequate kidney excretion of fluid; therefore,
cardiac output (and falling arterial pressure that also
fluid retention continues and can eventually be the
occurs). Consequently, within a few days, the state
cause of death. These events can be explained quanti-
of the circulation has reached point F on the curve,
tatively in the following way.
with the cardiac output now less than 2.5 L/min and
Note the straight line in Figure 22-2, at a cardiac
the right atrial pressure 16 mm Hg. This state has
output level of 5 L/min. This is approximately the crit-
approached or reached incompatibility with life, and
ical cardiac output level that is required in the normal
the patient dies. This state of heart failure in which the
adult person to make the kidneys re-establish normal
failure continues to worsen is called decompensated
fluid balance—that is, for the output of salt and water
heart failure.
to be as great as the intake of these. At any cardiac
Thus, one can see from this analysis that failure of
output below this level, all the fluid-retaining mecha-
the cardiac output (and arterial pressure) to rise to the
nisms discussed in the earlier section remain in play
critical level required for normal renal function results
and the body fluid volume increases progressively.
in (1) progressive retention of more and more fluid,
And because of this progressive increase in fluid
which causes (2) progressive elevation of the mean
volume, the mean systemic filling pressure of the
systemic filling pressure, and (3) progressive elevation
of the right atrial pressure until finally the heart is so
overstretched or so edematous that it cannot pump
even moderate quantities of blood and, therefore, fails
completely. Clinically, one detects this serious condi-
Critical cardiac output level
tion of decompensation principally by the progressing
for normal fluid balance
edema, especially edema of the lungs, which leads
to bubbling rales in the lungs and to dyspnea (air
hunger). All clinicians know that lack of appropriate
5.0
therapy when this state of events occurs leads to rapid
2.5
B
C D E
death.
A
F
0
Treatment of Decompensation. The decompensation
-4
0
+4
+8
+12
+16
process can often be stopped by (1) strengthening the
Right atrial pressure (mm Hg)
heart in any one of several ways, especially by admin-
istration of a cardiotonic drug, such as digitalis, so that
the heart becomes strong enough to pump adequate
quantities of blood required to make the kidneys func-
Figure 22-2
tion normally again, or (2) administering diuretic drugs
to increase kidney excretion while at the same time
Greatly depressed cardiac output that indicates decompensated
reducing water and salt intake, which brings about a
heart disease. Progressive fluid retention raises the right atrial
pressure over a period of days, and the cardiac output progresses
balance between fluid intake and output despite low
from point A to point F, until death occurs.
cardiac output.
262
Unit IV The Circulation
Both methods stop the decompensation process by
it can cause death by suffocation in 20 to 30 minutes,
re-establishing normal fluid balance, so that at least as
which we discuss more fully later in the chapter.
much fluid leaves the body as enters it.
Mechanism of Action of the Cardiotonic Drugs Such as
Low-Output Cardiac Failure—
Digitalis. Cardiotonic drugs, such as digitalis, when
Cardiogenic Shock
administered to a person with a healthy heart, have
little effect on increasing the contractile strength of
In many instances after acute heart attacks and
the cardiac muscle. However, when administered to a
often after prolonged periods of slow progressive
person with a chronically failing heart, the same drugs
cardiac deterioration, the heart becomes incapable of
can sometimes increase the strength of the failing
pumping even the minimal amount of blood flow
myocardium as much as 50 to 100 per cent. Therefore,
required to keep the body alive. Consequently, all the
they are one of the mainstays of therapy in chronic
body tissues begin to suffer and even to deteriorate,
heart failure.
often leading to death within a few hours to a few days.
Digitalis and other cardiotonic glycosides are
The picture then is one of circulatory shock, as
believed to strengthen heart contraction by increasing
explained in Chapter
24. Even the cardiovascular
the quantity of calcium ions in muscle fibers. In the
system suffers from lack of nutrition, and it, too (along
failing heart muscle, the sarcoplasmic reticulum fails
with the remainder of the body), deteriorates, thus has-
to accumulate normal quantities of calcium and,
tening death. This circulatory shock syndrome caused
therefore, cannot release enough calcium ions into
by inadequate cardiac pumping is called cardiogenic
the free-fluid compartment of the muscle fibers to
shock or simply cardiac shock. Once a person devel-
cause full contraction of the muscle. One effect of dig-
ops cardiogenic shock, the survival rate is often less
italis is to depress the calcium pump of the cell mem-
than 15 per cent.
brane of the cardiac muscle fibers. This pump normally
pumps calcium ions out of the muscle. However, in the
Vicious Circle of Cardiac Deterioration in Cardiogenic Shock.
case of a failing heart, extra calcium is needed to
The discussion of circulatory shock in Chapter 24
increase the muscle contractile force. Therefore, it is
emphasizes the tendency for the heart to become pro-
usually beneficial to depress the calcium pumping
gressively more damaged when its coronary blood
mechanism a moderate amount using digitalis, allow-
supply is reduced during the course of the shock. That
ing the muscle fiber intracellular calcium level to rise
is, the low arterial pressure that occurs during shock
slightly.
reduces the coronary blood supply even more. This
makes the heart still weaker, which makes the arterial
pressure fall still more, which makes the shock still
Unilateral Left Heart Failure
worse, the process eventually becoming a vicious circle
of cardiac deterioration. In cardiogenic shock caused
In the discussions thus far in this chapter, we have con-
by myocardial infarction, this problem is greatly com-
sidered failure of the heart as a whole. Yet, in a large
pounded by already existing coronary vessel blockage.
number of patients, especially those with early acute
For instance, in a healthy heart, the arterial pres-
failure, left-sided failure predominates over right-
sure usually must be reduced below about 45 mm Hg
sided failure, and in rare instances, the right side fails
before cardiac deterioration sets in. However, in a
without significant failure of the left side. Therefore,
heart that already has a blocked major coronary
we need especially to discuss the special features of
vessel, deterioration sets in when the coronary arterial
unilateral heart failure.
pressure falls below 80 to 90 mm Hg. In other words,
When the left side of the heart fails without con-
even a small decrease in arterial pressure can now
comitant failure of the right side, blood continues to
set off a vicious circle of cardiac deterioration. For
be pumped into the lungs with usual right heart vigor,
this reason, in treating myocardial infarction, it is
whereas it is not pumped adequately out of the lungs
extremely important to prevent even short periods of
by the left heart into the systemic circulation. As
hypotension.
a result, the mean pulmonary filling pressure rises
because of shift of large volumes of blood from the sys-
temic circulation into the pulmonary circulation.
Physiology of Treatment. Often a patient dies of cardio-
As the volume of blood in the lungs increases, the
genic shock before the various compensatory
pulmonary capillary pressure increases, and if this rises
processes can return the cardiac output (and arterial
above a value approximately equal to the colloid
pressure) to a life-sustaining level. Therefore, treat-
osmotic pressure of the plasma, about 28 mm Hg, fluid
ment of this condition is one of the most important
begins to filter out of the capillaries into the lung inter-
problems in the management of acute heart attacks.
stitial spaces and alveoli, resulting in pulmonary
Immediate administration of digitalis is often used
edema.
for strengthening the heart if the ventricular muscle
Thus, among the most important problems of left
shows signs of deterioration. Also, infusion of whole
heart failure are pulmonary vascular congestion and
blood, plasma, or a blood pressure-raising drug is used
pulmonary edema. In severe, acute left heart failure,
to sustain the arterial pressure. If the arterial pressure
pulmonary edema occasionally occurs so rapidly that
can be elevated high enough, the coronary blood flow
Chapter 22
Cardiac Failure
263
often will increase enough to prevent the vicious circle
of 13 mm Hg. Thus, severe acute cardiac failure often
of deterioration. And this allows enough time for
causes a fall in peripheral capillary pressure rather than
appropriate compensatory mechanisms in circulatory
a rise. Therefore, animal experiments, as well as expe-
system to correct the shock.
rience in humans, show that acute cardiac failure
Some success has also been achieved in saving the
almost never causes immediate development of peri-
lives of patients in cardiogenic shock by using one of
pheral edema.
the following procedures: (1) surgically removing the
clot in the coronary artery, often in combination with
Long-Term Fluid Retention by the Kidneys—
coronary bypass graft, or (2) catheterizing the blocked
The Cause of Peripheral Edema in Persisting
coronary artery and infusing either streptokinase or
Heart Failure
tissue-type plasminogen activator enzymes that cause
After the first day or so of overall heart failure or of
dissolution of the clot. The results occasionally are
right-ventricular heart failure, peripheral edema does
astounding when one of these procedures is instituted
begin to occur principally because of fluid retention by
within the first hour of cardiogenic shock but of little,
the kidneys. The retention of fluid increases the mean
if any, benefit after 3 hours.
systemic filling pressure, resulting in increased ten-
dency for blood to return to the heart. This elevates
the right atrial pressure to a still higher value and
Edema in Patients with
returns the arterial pressure back toward normal.
Cardiac Failure
Therefore, the capillary pressure now also rises
markedly, thus causing loss of fluid into the tissues and
Inability of Acute Cardiac Failure to Cause Peripheral Edema.
development of severe edema.
Acute left heart failure can cause terrific and rapid
There are three known causes of the reduced renal
congestion of the lungs, with development of pul-
output of urine during cardiac failure, all of which are
monary edema and even death within minutes to
equally important but in different ways.
hours.
1.
Decreased glomerular filtration. A decrease in
However, either left or right heart failure is very
cardiac output has a tendency to reduce the
slow to cause peripheral edema. This can best be
glomerular pressure in the kidneys because
explained by referring to Figure 22-3. When a previ-
of (1) reduced arterial pressure and (2) intense
ously healthy heart acutely fails as a pump, the aortic
sympathetic constriction of the afferent arterioles
pressure falls and the right atrial pressure rises. As the
of the kidney. As a consequence, except in the
cardiac output approaches zero, these two pressures
mildest degrees of heart failure, the glomerular
approach each other at an equilibrium value of about
filtration rate becomes less than normal. It is
13 mm Hg. Capillary pressure also falls from its normal
clear from the discussion of kidney function in
value of 17 mm Hg to the new equilibrium pressure
Chapters 26 through 29 that even a slight decrease
in glomerular filtration often markedly decreases
urine output. When the cardiac output falls to
about one-half normal, this can result in almost
complete anuria.
2.
Activation of the renin-angiotensin system and
Mean aortic pressure
increased reabsorption of water and salt by the
renal tubules. The reduced blood flow to the
Capillary pressure
kidneys causes marked increase in renin secretion
Right atrial pressure
100
by the kidneys, and this in turn causes the
formation of angiotensin, as described in Chapter
80
19. The angiotensin in turn has a direct effect on
the arterioles of the kidneys to decrease further
60
the blood flow through the kidneys, which
especially reduces the pressure in the capillaries
40
13 mm Hg
surrounding the renal tubules, promoting greatly
increased reabsorption of both water and salt
20
from the tubules. Therefore, loss of water and salt
into the urine decreases greatly, and large
quantities of salt and water accumulate in the
0
Normal
1/2 Normal
Zero
blood and interstitial fluids everywhere in the
body.
Cardiac output
3.
Increased aldosterone secretion. In the chronic
stage of heart failure, large quantities of
aldosterone are secreted by the adrenal cortex.
Figure 22-3
This results mainly from the effect of angiotensin
to stimulate aldosterone secretion by the adrenal
Progressive changes in mean aortic pressure, peripheral tissue
cortex. But some of the increase in aldosterone
capillary pressure, and right atrial pressure as the cardiac output
falls from normal to zero.
secretion often results from increased plasma
264
Unit IV The Circulation
potassium. Excess potassium is one of the most
5. The peripheral vasodilation increases venous
powerful stimuli known for aldosterone secretion,
return of blood from the peripheral circulation
and the potassium concentration rises in response
still more.
to reduced renal function in cardiac failure.
6. The increased venous return further increases the
The elevated aldosterone level further increases
damming of the blood in the lungs, leading to still
the reabsorption of sodium from the renal
more transudation of fluid, more arterial oxygen
tubules. This in turn leads to a secondary increase
desaturation, more venous return, and so forth.
in water reabsorption for two reasons: First, as the
Thus, a vicious circle has been established.
sodium is reabsorbed, it reduces the osmotic
Once this vicious circle has proceeded beyond a
pressure in the tubules but increases the osmotic
certain critical point, it will continue until death of the
pressure in the renal interstitial fluids; these
patient unless heroic therapeutic measures are used
changes promote osmosis of water into the blood.
within minutes. The types of heroic therapeutic meas-
Second, the absorbed sodium and anions that go
ures that can reverse the process and save the patient’s
with the sodium, mainly chloride ions, increase
life include the following:
the osmotic concentration of the extracellular
1. Putting tourniquets on both arms and legs to
fluid everywhere in the body. This elicits
sequester much of the blood in the veins and,
antidiuretic hormone secretion by the
therefore, decrease the workload on the left side
hypothalamic-posterior pituitary gland system
of the heart
(discussed in Chapter 29). The antidiuretic
2. Bleeding the patient
hormone in turn promotes still greater increase in
3. Giving a rapidly acting diuretic, such as
tubular reabsorption of water.
furosemide, to cause rapid loss of fluid from the
body
4. Giving the patient pure oxygen to breathe to
Role of Atrial Natriuretic Factor to Delay Onset of Cardiac
reverse the blood oxygen desaturation, the heart
Decompensation. Atrial natriuretic factor
(ANF) is a
deterioration, and the peripheral vasodilation
hormone released by the atrial walls of the heart when
5. Giving the patient a rapidly acting cardiotonic
they become stretched. Because heart failure almost
drug, such as digitalis, to strengthen the heart
always causes excessive increase in both the right and
This vicious circle of acute pulmonary edema can
left atrial pressures that stretch the atrial walls, the cir-
proceed so rapidly that death can occur in 20 minutes
culating levels of ANF in the blood increase fivefold
to 1 hour. Therefore, any procedure that is to be suc-
to tenfold in severe heart failure. The ANF in turn has
cessful must be instituted immediately.
a direct effect on the kidneys to increase greatly their
excretion of salt and water. Therefore, ANF plays a
natural role to help prevent extreme congestive symp-
Cardiac Reserve
toms during cardiac failure. The renal effects of ANF
are discussed in Chapter 29.
The maximum percentage that the cardiac output can
increase above normal is called the cardiac reserve.
Acute Pulmonary Edema in Late-Stage Heart
Thus, in the healthy young adult, the cardiac reserve is
Failure—Another Lethal Vicious Circle
300 to 400 per cent. In athletically trained persons, it
A frequent cause of death in heart failure is acute pul-
is occasionally 500 to 600 per cent. But in heart failure,
monary edema occurring in patients who have already
there is no cardiac reserve. As an example of normal
had chronic heart failure for a long time. When this
reserve, during severe exercise the cardiac output of a
occurs in a person without new cardiac damage, it
healthy young adult can rise to about five times
usually is set off by some temporary overload of the
normal; this is an increase above normal of 400 per
heart, such as might result from a bout of heavy exer-
cent—that is, a cardiac reserve of 400 per cent.
cise, some emotional experience, or even a severe cold.
Any factor that prevents the heart from pumping
The acute pulmonary edema is believed to result from
blood satisfactorily will decrease the cardiac reserve.
the following vicious circle:
This can result from ischemic heart disease, primary
1. A temporarily increased load on the already weak
myocardial disease, vitamin deficiency that affects
left ventricle initiates the vicious circle. Because of
cardiac muscle, physical damage to the myocardium,
limited pumping capacity of the left heart, blood
valvular heart disease, and many other factors, some of
begins to dam up in the lungs.
which are shown in Figure 22-4.
2. The increased blood in the lungs elevates the
pulmonary capillary pressure, and a small amount
Diagnosis of Low Cardiac Reserve—Exercise Test. As long as
of fluid begins to transude into the lung tissues
persons with low cardiac reserve remain in a state of
and alveoli.
rest, they usually will not know that they have heart
3. The increased fluid in the lungs diminishes the
disease. However, a diagnosis of low cardiac reserve
degree of oxygenation of the blood.
usually can be easily made by requiring the person to
4. The decreased oxygen in the blood further
exercise either on a treadmill or by walking up and
weakens the heart and also weakens the arterioles
down steps, either of which requires greatly increased
everywhere in the body, thus causing peripheral
cardiac output. The increased load on the heart rapidly
vasodilation.
uses up the small amount of reserve that is available,
Chapter 22
Cardiac Failure
265
Athlete
15
Normal
600
10
500
Normal
Mild
400
D
valvular
A
5
C
disease
300
Moderate
coronary
B
200
disease
Diphtheria
0
Severe
Severe
-4
-2
0
2
4
6
8
10
12
14
100
coronary
valvular
Right atrial pressure (mm Hg)
thrombosis
disease
0
Normal
operation
Figure 22-5
Progressive changes in cardiac output and right atrial pressure
during different stages of cardiac failure.
Figure 22-4
Cardiac reserve in different conditions, showing less than zero
reserve for two of the conditions.
and the cardiac output soon fails to rise high enough
circulation. The two curves passing through Point A
to sustain the body’s new level of activity. The acute
are (1) the normal cardiac output curve and (2) the
effects are as follows:
normal venous return curve. As pointed out in Chapter
1. Immediate and sometimes extreme shortness of
20, there is only one point on each of these two curves
breath (dyspnea) resulting from failure of the
at which the circulatory system can operate. This point
heart to pump sufficient blood to the tissues,
is where the two curves cross at point A. Therefore, the
thereby causing tissue ischemia and creating a
normal state of the circulation is a cardiac output and
sensation of air hunger
venous return of 5 L/min and a right atrial pressure of
2. Extreme muscle fatigue resulting from muscle
0 mm Hg.
ischemia, thus limiting the person’s ability to
continue with the exercise
Effect of Acute Heart Attack. During the first few seconds
3. Excessive increase in heart rate because the
after a moderately severe heart attack, the cardiac
nervous reflexes to the heart overreact in an
output curve falls to the lowermost curve. During these
attempt to overcome the inadequate cardiac
few seconds, the venous return curve still has not
output
changed because the peripheral circulatory system is
Exercise tests are part of the armamentarium of the
still operating normally. Therefore, the new state of the
cardiologist. These tests take the place of cardiac
circulation is depicted by point B, where the new
output measurements that cannot be made with ease
cardiac output curve crosses the normal venous return
in most clinical settings.
curve. Thus, the right atrial pressure rises immedi-
ately to 4 mm Hg, whereas the cardiac output falls to
2 L/min.
Quantitative Graphical Method for
Analysis of Cardiac Failure
Effect of Sympathetic Reflexes. Within the next
30
Although it is possible to understand most general
seconds, the sympathetic reflexes become very active.
principles of cardiac failure using mainly qualitative
They affect both the cardiac output and the venous
logic, as we have done thus far in this chapter, one can
return curves, raising both of them. Sympathetic stim-
grasp the importance of the different factors in cardiac
ulation can increase the plateau level of the cardiac
failure with far greater depth by using more quantita-
output curve as much as 30 to 100 per cent. It can also
tive approaches. One such approach is the graphical
increase the mean systemic filling pressure (depicted
method for analysis of cardiac output regulation intro-
by the point where the venous return curve crosses the
duced in Chapter 20. In the remaining sections of this
zero venous return axis) by several millimeters of
chapter, we analyze several aspects of cardiac failure,
mercury—in this figure, from a normal value of 7 mm
using this graphical technique.
Hg up to 10 mm Hg. This increase in mean systemic
filling pressure shifts the entire venous return curve to
Graphical Analysis of Acute Heart Failure and
the right and upward. The new cardiac output and
Chronic Compensation
venous return curves now equilibrate at point C, that
Figure 22-5 shows cardiac output and venous return
is, at a right atrial pressure of +5 mm Hg and a cardiac
curves for different states of the heart and peripheral
output of 4 L/min.
266
Unit IV The Circulation
Compensation During the Next Few Days. During the
curve to this low level. At point A, the curve at time
ensuing week, the cardiac output and venous return
zero equates with the normal venous return curve to
curves rise further because of (1) some recovery of the
give a cardiac output of about 3 L/min. However, stim-
heart and (2) renal retention of salt and water, which
ulation of the sympathetic nervous system, caused by
raises the mean systemic filling pressure still further—
this low cardiac output, increases the mean systemic
this time up to +12 mm Hg. The two new curves now
filling pressure within 30 seconds from 7 to 10.5 mm
equilibrate at point D. Thus, the cardiac output has
Hg. This shifts the venous return curve upward and to
now returned to normal. The right atrial pressure,
the right to produce the curve labeled “autonomic
however, has risen still further to +6 mm Hg. Because
compensation.” Thus, the new venous return curve
the cardiac output is now normal, renal output is also
equates with the cardiac output curve at point B. The
normal, so that a new state of equilibrated fluid
cardiac output has been improved to a level of 4 L/min
balance has been achieved. The circulatory system will
but at the expense of an additional rise in right atrial
continue to function at point D and remain stable, with
pressure to 5 mm Hg.
a normal cardiac output and an elevated right atrial
The cardiac output of 4 L/min is still too low to cause
pressure, until some additional extrinsic factor changes
the kidneys to function normally. Therefore, fluid con-
either the cardiac output curve or the venous return
tinues to be retained, and the mean systemic filling
curve.
pressure rises from 10.5 to almost 13 mm Hg. Now the
Using this technique for analysis, one can see espe-
venous return curve becomes that labeled “2nd day”
cially the importance of moderate fluid retention and
and equilibrates with the cardiac output curve at point
how it eventually leads to a new stable state of the cir-
C. The cardiac output rises to 4.2 L/min and the right
culation in mild to moderate heart failure. And one
atrial pressure to 7 mm Hg.
can also see the interrelation between mean systemic
During the succeeding days, the cardiac output
filling pressure and cardiac pumping at various
never rises quite high enough to re-establish normal
degrees of heart failure.
renal function. Fluid continues to be retained, the
Note that the events described in Figure 22-5 are
mean systemic filling pressure continues to rise, the
the same as those presented in Figure 22-1, but in
venous return curve continues to shift to the right, and
Figure 22-5, they are presented in a more quantitative
the equilibrium point between the venous return curve
manner.
and the cardiac output curve also shifts progressively
to point D, to point E, and, finally, to point F. The equi-
Graphical Analysis of “Decompensated”
libration process is now on the down slope of the
Cardiac Failure
cardiac output curve, so that further retention of fluid
The black cardiac output curve in Figure 22-6 is the
causes only more severe cardiac edema and a detri-
same as the curve shown in Figure 22-2, a very low
mental effect on cardiac output. The condition accel-
curve that has already reached a degree of recovery as
erates downhill until death occurs.
great as this heart can achieve. In this figure, we have
Thus, “decompensation” results from the fact that
added venous return curves that occur during succes-
the cardiac output curve never rises to the critical level
sive days after the acute fall of the cardiac output
of 5 L/min needed to re-establish normal kidney excre-
tion of fluid that would be required to cause balance
between fluid input and output.
Treatment of Decompensated Heart Disease with Digitalis. Let
us assume that the stage of decompensation has
already reached point E in Figure 22-6, and let us
proceed to the same point E in Figure 22-7. At this
time, digitalis is given to strengthen the heart. This
15
raises the cardiac output curve to the level shown in
Critical cardiac
Figure 22-7, but there is not an immediate change in
output level
10
the venous return curve. Therefore, the new cardiac
Au
for normal
fluid balance
output curve equates with the venous return curve at
point G. The cardiac output is now 5.7 L/min, a value
5
C D
greater than the critical level of 5 liters required to
E
F
A B
make the kidneys excrete normal amounts of urine.
0
Therefore, the kidneys eliminate much more fluid than
-4
-2
0
2
4
6
8
10
12
14
16
normally, causing diuresis, a well-known therapeutic
Right atrial pressure (mm Hg)
effect of digitalis.
The progressive loss of fluid over a period of several
days reduces the mean systemic filling pressure back
down to 11.5 mm Hg, and the new venous return curve
Figure 22-6
becomes the curve labeled “Several days later.” This
curve equates with the cardiac output curve of the
Graphical analysis of decompensated heart disease showing
digitalized heart at point H, at an output of 5 L/min
progressive shift of the venous return curve to the right as a result
of continued fluid retention.
and a right atrial pressure of 4.6 mm Hg. This cardiac
Chapter 22
Cardiac Failure
267
15
25
Critical cardiac output
level for normal
10
fluid balance
20
G
H
Normal
Normal cardiac
5
15
venous
output curve
E
return
B
curve
0
10
C
-4
-2
0
2
4
6
8
10
12
14
16
Right atrial pressure (mm Hg)
5
Beriberi
A
heart
disease
0
Figure 22-7
-4 -2
0
2
4
6
8
10
12
14
16
Right atrial pressure (mm Hg)
Treatment of decompensated heart disease showing the effect of
digitalis in elevating the cardiac output curve, this in turn causing
increased urine output and progressive shift of the venous return
curve to the left.
Figure 22-8
Graphical analysis of two types of conditions that can cause high-
output cardiac failure:
(1) arteriovenous
(AV) fistula and
(2)
output is precisely that required for normal fluid
beriberi heart disease.
balance. Therefore, no additional fluid will be lost and
none will be gained. Consequently, the circulatory
system has now stabilized, or in other words, the
decompensation of the heart failure has been “com-
pensated.” And to state this another way, the final
steady-state condition of the circulation is defined by
is slightly elevated, and there are mild signs of periph-
the crossing point of three curves: the cardiac output
eral congestion. If the person attempts to exercise, he
curve, the venous return curve, and the critical level
or she will have little cardiac reserve because the heart
for normal fluid balance. The compensatory mecha-
is already being used almost to maximum capacity to
nisms automatically stabilize the circulation when all
pump the extra blood through the arteriovenous
three curves cross at the same point.
fistula. This condition resembles a failure condition
and is called “high-output failure,” but in reality, the
Graphical Analysis of High-Output
heart is overloaded by excess venous return.
Cardiac Failure
Figure 22-8 gives an analysis of two types of high-
Beriberi. Figure 22-8 shows the approximate changes
output cardiac failure. One of these is caused by an
in the cardiac output and venous return curves caused
arteriovenous fistula that overloads the heart because
by beriberi. The decreased level of the cardiac output
of excessive venous return, even though the pumping
curve is caused by weakening of the heart because of
capability of the heart is not depressed. The other is
the avitaminosis (mainly lack of thiamine) that causes
caused by beriberi, in which the venous return is
the beriberi syndrome. The weakening of the heart has
greatly increased because of diminished systemic vas-
decreased the blood flow to the kidneys. Therefore, the
cular resistance, but at the same time, the pumping
kidneys have retained a large amount of extra body
capability of the heart is depressed.
fluid, which in turn has increased the mean systemic
filling pressure (represented by the point where the
Arteriovenous Fistula. The
“normal” curves of Figure
venous return curve now intersects the zero cardiac
22-8 depict the normal cardiac output and normal
output level) from the normal value of 7 mm Hg up to
venous return curves. These equate with each other at
11 mm Hg. This has shifted the venous return curve to
point A, which depicts a normal cardiac output of 5 L/
the right. Finally, the venous return curve has rotated
min and a normal right atrial pressure of 0 mm Hg.
upward from the normal curve because the avita-
Now let us assume that the systemic resistance (the
minosis has dilated the peripheral blood vessels, as
total peripheral resistance) becomes greatly decreased
explained in Chapter 17.
because of opening a large arteriovenous fistula (a
The two blue curves (cardiac output curve and
direct opening between a large artery and a large
venous return curve) intersect with each other at
vein). The venous return curve rotates upward to give
point C, which describes the circulatory condition in
the curve labeled “AV fistula.” This venous return
beriberi, with a right atrial pressure in this instance of
curve equates with the normal cardiac output curve at
9 mm Hg and a cardiac output about 65 per cent above
point B, with a cardiac output of 12.5 L/min and a right
normal; this high cardiac output occurs despite the
atrial pressure of 3 mm Hg. Thus, the cardiac output
weak heart, as demonstrated by the depressed plateau
has become greatly elevated, the right atrial pressure
level of the cardiac output curve.
268
Unit IV The Circulation
References
Lohmeier TE: Neurohumoral regulation of arterial pressure
in hemorrhage and heart failure. Am J Physiol Regul
Integr Comp Physiol 283:R810, 2002.
Andrew P: Diastolic heart failure demystified. Chest 124:744,
Mehra MR, Gheorghiade M, Bonow RO: Mitral regurgita-
2003.
tion in chronic heart failure: more questions than answers?
Braunwald E, Zipes DP, Liby P: A Textbook for Cardiovas-
Curr Cardiol Rep 6:96, 2004.
cular Medicine. 6th Ed. Philadelphia: WB Saunders Co,
McMurray J, Pfeffer MA: New therapeutic options in con-
2001.
gestive heart failure: Part I. Circulation 105:2099, 2002.
Dorn GW 2nd, Molkentin JD: Manipulating cardiac con-
McMurray J, Pfeffer MA: New therapeutic options in con-
tractility in heart failure: data from mice and men. Circu-
gestive heart failure: Part II. Circulation 105:2223, 2002
lation 109:150, 2004.
Pfisterer M: Right ventricular involvement in myocardial
Ducas J, Grech ED: ABC of interventional cardiology. Per-
infarction and cardiogenic shock. Lancet 362:392, 2003.
cutaneous coronary intervention: cardiogenic shock. BMJ
Ruskoaho H: Cardiac hormones as diagnostic tools in heart
326:1450, 2003.
failure. Endocr Rev 24:341, 2003.
Eikelboom J, White H, Yusuf S: The evolving role of direct
Shlipak MG: Pharmacotherapy for heart failure in patients
thrombin inhibitors in acute coronary syndromes. J Am
with renal insufficiency. Ann Intern Med 138:917, 2003.
Coll Cardiol 41(4 Suppl S):70S, 2003.
Sjaastad I, Wasserstrom JA, Sejersted OM: Heart failure—
Floras JS: Sympathetic activation in human heart failure:
a challenge to our current concepts of excitation-
diverse mechanisms, therapeutic opportunities. Acta
contraction coupling. J Physiol 546:33, 2003.
Physiol Scand 177:391, 2003.
Spodick DH: Acute cardiac tamponade. N Engl J Med
Gavras H, Brunner HR: Role of angiotensin and its inhibi-
349:684, 2003.
tion in hypertension, ischemic heart disease, and heart
Zile MR, Brutsaert DL: New concepts in diastolic dysfunc-
failure. Hypertension 37:342, 2001.
tion and diastolic heart failure: Part I: diagnosis, progno-
Gehlbach BK, Geppert E: The pulmonary manifestations of
sis, and measurements of diastolic function. Circulation
left heart failure. Chest 125:669, 2004.
105:1387, 2002.
Guyton AC, Jones CE, Coleman TG: Circulatory Physiology:
Zile MR, Brutsaert DL: New concepts in diastolic dysfunc-
Cardiac Output and Its Regulation. Philadelphia: WB
tion and diastolic heart failure: Part II: causal mechanisms
Saunders Co, 1973.
and treatment. Circulation 105:1503, 2002.
Hochman JS: Cardiogenic shock complicating acute myocar-
dial infarction: expanding the paradigm. Circulation 107:
2998, 2003.
C H A P T E R
2
3
Heart Valves and Heart Sounds;
Dynamics of Valvular and
Congenital Heart Defects
Function of the heart valves was discussed in
Chapter 9, where it was pointed out that closing of
the valves causes audible sounds. Ordinarily, no
audible sounds occur when the valves open. In this
chapter, we first discuss the factors that cause the
sounds in the heart under normal and abnormal
conditions. Then we discuss what happens in the
overall circulatory system when valvular or congen-
ital heart defects are present.
Heart Sounds
Normal Heart Sounds
Listening with a stethoscope to a normal heart, one hears a sound usually
described as “lub, dub, lub, dub.” The “lub” is associated with closure of the atrio-
ventricular (A-V) valves at the beginning of systole, and the “dub” is associated
with closure of the semilunar (aortic and pulmonary) valves at the end of
systole. The “lub” sound is called the first heart sound, and the “dub” is called
the second heart sound, because the normal pumping cycle of the heart is con-
sidered to start when the A-V valves close at the onset of ventricular systole.
Causes of the First and Second Heart Sounds. The earliest explanation for the cause
of the heart sounds was that the “slapping” together of the valve leaflets sets
up vibrations. However, this has been shown to cause little, if any, of the sound,
because the blood between the leaflets cushions the slapping effect and pre-
vents significant sound. Instead, the cause is vibration of the taut valves imme-
diately after closure, along with vibration of the adjacent walls of the heart and
major vessels around the heart. That is, in generating the first heart sound, con-
traction of the ventricles first causes sudden backflow of blood against the
A-V valves (the tricuspid and mitral valves), causing them to close and bulge
toward the atria until the chordae tendineae abruptly stop the back bulging.
The elastic tautness of the chordae tendineae and of the valves then causes the
back surging blood to bounce forward again into each respective ventricle. This
causes the blood and the ventricular walls, as well as the taut valves, to vibrate
and causes vibrating turbulence in the blood. The vibrations travel through the
adjacent tissues to the chest wall, where they can be heard as sound by using
the stethoscope.
The second heart sound results from sudden closure of the semilunar valves
at the end of systole. When the semilunar valves close, they bulge backward
toward the ventricles, and their elastic stretch recoils the blood back into the
arteries, which causes a short period of reverberation of blood back and forth
between the walls of the arteries and the semilunar valves, as well as between
these valves and the ventricular walls. The vibrations occurring in the arterial
walls are then transmitted mainly along the arteries. When the vibrations of the
vessels or ventricles come into contact with a “sounding board,” such as the
chest wall, they create sound that can be heard.
269
270
Unit IV The Circulation
Duration and Pitch of the First and Second Heart Sounds. The
third of diastole. A logical but unproved explanation
duration of each of the heart sounds is slightly more
of this sound is oscillation of blood back and forth
than 0.10 second—the first sound about 0.14 second,
between the walls of the ventricles initiated by inrush-
and the second about 0.11 second. The reason for the
ing blood from the atria. This is analogous to running
shorter second sound is that the semilunar valves are
water from a faucet into a paper sack, the inrushing
more taut than the A-V valves, so that they vibrate for
water reverberating back and forth between the walls
a shorter time than do the A-V valves.
of the sack to cause vibrations in its walls. The reason
The audible range of frequency (pitch) in the first
the third heart sound does not occur until the middle
and second heart sounds, as shown in Figure 23-1,
third of diastole is believed to be that in the early part
begins at the lowest frequency the ear can detect,
of diastole, the ventricles are not filled sufficiently to
about 40 cycles/sec, and goes up above 500 cycles/sec.
create even the small amount of elastic tension neces-
When special electronic apparatus is used to record
sary for reverberation. The frequency of this sound is
these sounds, by far a larger proportion of the re-
usually so low that the ear cannot hear it, yet it can
corded sound is at frequencies and sound levels below
often be recorded in the phonocardiogram.
the audible range, going down to 3 to 4 cycles/sec and
peaking at about 20 cycles/sec, as illustrated by the
Atrial Heart Sound (Fourth Heart Sound). An atrial heart
lower shaded area in Figure 23-1. For this reason,
sound can sometimes be recorded in the phonocar-
major portions of the heart sounds can be recorded
diogram, but it can almost never be heard with a
electronically in phonocardiograms even though they
stethoscope because of its weakness and very low
cannot be heard with a stethoscope.
frequency—usually 20 cycles/sec or less. This sound
The second heart sound normally has a higher fre-
occurs when the atria contract, and presumably, it is
quency than the first heart sound for two reasons: (1)
caused by the inrush of blood into the ventricles, which
the tautness of the semilunar valves in comparison
initiates vibrations similar to those of the third heart
with the much less taut A-V valves, and (2) the greater
sound.
elastic coefficient of the taut arterial walls that provide
the principal vibrating chambers for the second sound,
Chest Surface Areas for Auscultation
in comparison with the much looser, less elastic ven-
of Normal Heart Sounds
tricular chambers that provide the vibrating system for
Listening to the sounds of the body, usually with the
the first heart sound. The clinician uses these differ-
aid of a stethoscope, is called auscultation. Figure 23-2
ences to distinguish special characteristics of the two
shows the areas of the chest wall from which the dif-
respective sounds.
ferent heart valvular sounds can best be distinguished.
Although the sounds from all the valves can be heard
Third Heart Sound. Occasionally a weak, rumbling third
from all these areas, the cardiologist distinguishes
heart sound is heard at the beginning of the middle
the sounds from the different valves by a process of
Heart sounds
Inaudible and murmurs
Aortic area
Pulmonic area
100
10
Speech
1
area
0.1
Heart sounds
0.01
and murmurs
0.001
0.0001
0
8
32
64 128 256 5121024 2048 4096
Frequency in cycles per second
Figure 23-1
Amplitude of different-frequency vibrations in the heart sounds
and heart murmurs in relation to the threshold of audibility,
Tricuspid area
Mitral area
showing that the range of sounds that can be heard is between
40 and 520 cycles/sec. (Modified from Butterworth JS, Chassin
Figure 23-2
JL, McGrath JJ: Cardiac Auscultation, 2nd ed. New York: Grune
& Stratton, 1960.)
Chest areas from which sound from each valve is best heard.
Chapter 23
Heart Valves and Heart Sounds; Dynamics of Valvular and Congenital Heart Defects
271
Valvular Lesions
1st
2nd
3rd
Atrial
Rheumatic Valvular Lesions
By far the greatest number of valvular lesions results
A
Normal
from rheumatic fever. Rheumatic fever is an autoim-
mune disease in which the heart valves are likely to be
B
damaged or destroyed. It is usually initiated by strep-
Aortic stenosis
tococcal toxin in the following manner.
The sequence of events almost always begins with a
C
preliminary streptococcal infection caused specifically
Mitral regurgitation
by group A hemolytic streptococci. These bacteria ini-
tially cause a sore throat, scarlet fever, or middle ear
D
infection. But the streptococci also release several dif-
Aortic regurgitation
ferent proteins against which the person’s reticuloen-
dothelial system produces antibodies. The antibodies
E
Mitral stenosis
react not only with the streptococcal protein but also
with other protein tissues of the body, often causing
severe immunologic damage. These reactions continue
F
to take place as long as the antibodies persist in the
Patent ductus
blood—1 year or more.
arteriosus
Rheumatic fever causes damage especially in
Diastole
Systole
Diastole
Systole
certain susceptible areas, such as the heart valves. The
degree of heart valve damage is directly correlated
with the concentration and persistence of the anti-
bodies. The principles of immunity that relate to this
Figure 23-3
type of reaction are discussed in Chapter 34, and it is
noted in Chapter 31 that acute glomerular nephritis of
Phonocardiograms from normal and abnormal hearts.
the kidneys has a similar immunologic basis.
In rheumatic fever, large hemorrhagic, fibrinous,
bulbous lesions grow along the inflamed edges of
the heart valves. Because the mitral valve receives
more trauma during valvular action than any of the
elimination. That is, he or she moves the stethoscope
other valves, it is the one most often seriously
from one area to another, noting the loudness of the
damaged, and the aortic valve is second most fre-
sounds in different areas and gradually picking out the
quently damaged. The right heart valves, the tricuspid
sound components from each valve.
and pulmonary valves, are usually affected much less
The areas for listening to the different heart sounds
severely, probably because the low-pressure stresses
are not directly over the valves themselves. The aortic
that act on these valves are slight compared with
area is upward along the aorta because of sound trans-
the high-pressure stresses that act on the left heart
mission up the aorta, and the pulmonic area is upward
valves.
along the pulmonary artery. The tricuspid area is over
the right ventricle, and the mitral area is over the apex
Scarring of the Valves. The lesions of acute rheumatic
of the left ventricle, which is the portion of the heart
fever frequently occur on adjacent valve leaflets simul-
nearest the surface of the chest; the heart is rotated
taneously, so that the edges of the leaflets become
so that the remainder of the left ventricle lies more
stuck together. Then, weeks, months, or years later, the
posteriorly.
lesions become scar tissue, permanently fusing por-
tions of adjacent valve leaflets. Also, the free edges of
Phonocardiogram
the leaflets, which are normally filmy and free-flapping,
If a microphone specially designed to detect low-fre-
often become solid, scarred masses.
quency sound is placed on the chest, the heart sounds
A valve in which the leaflets adhere to one another
can be amplified and recorded by a high-speed record-
so extensively that blood cannot flow through it nor-
ing apparatus. The recording is called a phonocardio-
mally is said to be stenosed. Conversely, when the valve
gram, and the heart sounds appear as waves, as shown
edges are so destroyed by scar tissue that they cannot
schematically in Figure
23-3. Recording A is an
close as the ventricles contract, regurgitation (back-
example of normal heart sounds, showing the vibra-
flow) of blood occurs when the valve should be closed.
tions of the first, second, and third heart sounds and
Stenosis usually does not occur without the coexis-
even the very weak atrial sound. Note specifically that
tence of at least some degree of regurgitation, and vice
the third and atrial heart sounds are each a very low
versa.
rumble. The third heart sound can be recorded in only
one third to one half of all people, and the atrial heart
Other Causes of Valvular Lesions. Stenosis or lack of one
sound can be recorded in perhaps one fourth of all
or more leaflets of a valve also occurs occasionally as
people.
a congenital defect. Complete lack of leaflets is rare;
272
Unit IV The Circulation
congenital stenosis is more common, as is discussed
frequency, so that most of the sound spectrum is below
later in this chapter.
the low-frequency end of human hearing.
During the early part of diastole, a left ventricle with
Heart Murmurs Caused by Valvular Lesions
a stenotic mitral valve has so little blood in it and its
As shown by the phonocardiograms in Figure 23-3,
walls are so flabby that blood does not reverberate
many abnormal heart sounds, known as
“heart
back and forth between the walls of the ventricle. For
murmurs,” occur when there are abnormalities of the
this reason, even in severe mitral stenosis, no murmur
valves, as follows.
may be heard during the first third of diastole. Then,
after partial filling, the ventricle has stretched enough
Systolic Murmur of Aortic Stenosis. In aortic stenosis,
for blood to reverberate, and a low rumbling murmur
blood is ejected from the left ventricle through only a
begins.
small fibrous opening of the aortic valve. Because of
the resistance to ejection, sometimes the blood pres-
Phonocardiograms of Valvular Murmurs. Phonocardio-
sure in the left ventricle rises as high as 300 mm Hg,
grams B, C, D, and E of Figure 23-3 show, respectively,
while the pressure in the aorta is still normal. Thus,
idealized records obtained from patients with aortic
a nozzle effect is created during systole, with blood
stenosis, mitral regurgitation, aortic regurgitation, and
jetting at tremendous velocity through the small
mitral stenosis. It is obvious from these phonocardio-
opening of the valve. This causes severe turbulence of
grams that the aortic stenotic lesion causes the loudest
the blood in the root of the aorta. The turbulent blood
murmur, and the mitral stenotic lesion causes the
impinging against the aortic walls causes intense vibra-
weakest. The phonocardiograms show how the inten-
tion, and a loud murmur (see recording B, Figure 23-3)
sity of the murmurs varies during different portions of
occurs during systole and is transmitted throughout
systole and diastole, and the relative timing of each
the superior thoracic aorta and even into the large
murmur is also evident. Note especially that the
arteries of the neck. This sound is harsh and in severe
murmurs of aortic stenosis and mitral regurgitation
stenosis may be so loud that it can be heard several
occur only during systole, whereas the murmurs of
feet away from the patient. Also, the sound vibrations
aortic regurgitation and mitral stenosis occur only
can often be felt with the hand on the upper chest and
during diastole. If the reader does not understand this
lower neck, a phenomenon known as a “thrill.”
timing, extra review should be undertaken until it is
understood.
Diastolic Murmur of Aortic Regurgitation. In aortic regurgi-
tation, no abnormal sound is heard during systole,
but during diastole, blood flows backward from the
Abnormal Circulatory
high-pressure aorta into the left ventricle, causing
Dynamics in Valvular
a “blowing” murmur of relatively high pitch with a
Heart Disease
swishing quality heard maximally over the left ventri-
cle (see recording D, Figure 23-3). This murmur results
from turbulence of blood jetting backward into the
Dynamics of the Circulation in Aortic
blood already in the low-pressure diastolic left
Stenosis and Aortic Regurgitation
ventricle.
In aortic stenosis, the contracting left ventricle fails to
Systolic Murmur of Mitral Regurgitation. In mitral regurgi-
empty adequately, whereas in aortic regurgitation,
tation, blood flows backward through the mitral valve
blood flows backward into the ventricle from the aorta
into the left atrium during systole. This also causes a
after the ventricle has just pumped the blood into the
high-frequency “blowing,” swishing sound (see record-
aorta. Therefore, in either case, the net stroke volume
ing C, Figure 23-3) similar to that of aortic regurgita-
output of the heart is reduced.
tion but occurring during systole rather than diastole.
Several important compensations take place that
It is transmitted most strongly into the left atrium.
can ameliorate the severity of the circulatory defects.
However, the left atrium is so deep within the chest
Some of these compensations are the following.
that it is difficult to hear this sound directly over the
atrium. As a result, the sound of mitral regurgitation
Hypertrophy of the Left Ventricle. In both aortic stenosis
is transmitted to the chest wall mainly through the left
and aortic regurgitation, the left ventricular muscula-
ventricle to the apex of the heart.
ture hypertrophies because of the increased ventricu-
lar workload.
Diastolic Murmur of Mitral Stenosis. In mitral stenosis,
In regurgitation, the left ventricular chamber also
blood passes with difficulty through the stenosed
enlarges to hold all the regurgitant blood from the
mitral valve from the left atrium into the left ventri-
aorta. Sometimes the left ventricular muscle mass
cle, and because the pressure in the left atrium seldom
increases fourfold to fivefold, creating a tremendously
rises above 30 mm Hg, a large pressure differential
large left side of the heart.
forcing blood from the left atrium into the left ventri-
When the aortic valve is seriously stenosed, the
cle does not develop. Consequently, the abnormal
hypertrophied muscle allows the left ventricle to
sounds heard in mitral stenosis
(see recording E,
develop as much as 400 mm Hg intraventricular pres-
Figure
23-3) are usually weak and of very low
sure at systolic peak.
Chapter 23
Heart Valves and Heart Sounds; Dynamics of Valvular and Congenital Heart Defects
273
In severe aortic regurgitation, sometimes the hyper-
development of serious pulmonary edema. Ordinarily,
trophied muscle allows the left ventricle to pump a
lethal edema does not occur until the mean left atrial
stroke volume output as great as
250 milliliters,
pressure rises above 25 mm Hg and sometimes as high
although as much as three fourths of this blood returns
as 40 mm Hg, because the lung lymphatic vasculature
to the ventricle during diastole, and only one fourth
enlarges manyfold and can carry fluid away from the
flows through the aorta to the body.
lung tissues extremely rapidly.
Increase in Blood Volume. Another effect that helps
Enlarged Left Atrium and Atrial Fibrillation. The high left
compensate for the diminished net pumping by the left
atrial pressure in mitral valvular disease also causes
ventricle is increased blood volume. This results from
progressive enlargement of the left atrium, which
(1) an initial slight decrease in arterial pressure, plus
increases the distance that the cardiac electrical exci-
(2) peripheral circulatory reflexes that the decrease in
tatory impulse must travel in the atrial wall. This
pressure induces. These together diminish renal output
pathway may eventually become so long that it pre-
of urine, causing the blood volume to increase and
disposes to development of excitatory signal circus
the mean arterial pressure to return to normal. Also,
movements, as discussed in Chapter 13. Therefore,
red cell mass eventually increases because of a slight
in late stages of mitral valvular disease, especially in
degree of tissue hypoxia.
mitral stenosis, atrial fibrillation usually occurs. This
The increase in blood volume tends to increase
further reduces the pumping effectiveness of the heart
venous return to the heart. This, in turn, causes the left
and causes further cardiac debility.
ventricle to pump with the extra power required to
overcome the abnormal pumping dynamics.
Compensation in Early Mitral Valvular Disease. As also
occurs in aortic valvular disease and in many types of
Eventual Failure of the Left Ventricle, and
congenital heart disease, the blood volume increases
Development of Pulmonary Edema
in mitral valvular disease principally because of dimin-
In the early stages of aortic stenosis or aortic regurgi-
ished excretion of water and salt by the kidneys. This
tation, the intrinsic ability of the left ventricle to adapt
increased blood volume increases venous return to the
to increasing loads prevents significant abnormalities
heart, thereby helping to overcome the effect of the
in circulatory function in the person during rest, other
cardiac debility. Therefore, after compensation, cardiac
than increased work output required of the left ven-
output may fall only minimally until the late stages of
tricle. Therefore, considerable degrees of aortic steno-
mitral valvular disease, even though the left atrial pres-
sis or aortic regurgitation often occur before the
sure is rising.
person knows that he or she has serious heart disease
As the left atrial pressure rises, blood begins to dam
(such as a resting left ventricular systolic pressure as
up in the lungs, eventually all the way back to the pul-
high as 200 mm Hg in aortic stenosis or a left ventric-
monary artery. In addition, incipient edema of the
ular stroke volume output as high as double normal in
lungs causes pulmonary arteriolar constriction. These
aortic regurgitation).
two effects together increase systolic pulmonary arte-
Beyond a critical stage in these aortic valve lesions,
rial pressure and also right ventricular pressure, some-
the left ventricle finally cannot keep up with the work
times to as high as 60 mm Hg, which is more than
demand. As a consequence, the left ventricle dilates
double normal. This, in turn, causes hypertrophy of the
and cardiac output begins to fall; blood simultaneously
right side of the heart, which partially compensates for
dams up in the left atrium and in the lungs behind the
its increased workload.
failing left ventricle. The left atrial pressure rises pro-
gressively, and at mean left atrial pressures above 25
to 40 mm Hg, serious edema appears in the lungs, as
Circulatory Dynamics During Exercise
discussed in detail in Chapter 38.
in Patients with Valvular Lesions
During exercise, large quantities of venous blood are
Dynamics of Mitral Stenosis and
returned to the heart from the peripheral circulation.
Mitral Regurgitation
Therefore, all the dynamic abnormalities that occur in
the different types of valvular heart disease become
In mitral stenosis, blood flow from the left atrium into
tremendously exacerbated. Even in mild valvular
the left ventricle is impeded, and in mitral regurgita-
heart disease, in which the symptoms may be unrec-
tion, much of the blood that has flowed into the left
ognizable at rest, severe symptoms often develop
ventricle during diastole leaks back into the left atrium
during heavy exercise. For instance, in patients with
during systole rather than being pumped into the
aortic valvular lesions, exercise can cause acute left
aorta. Therefore, either of these conditions reduces net
ventricular failure followed by acute pulmonary
movement of blood from the left atrium into the left
edema. Also, in patients with mitral disease, exercise
ventricle.
can cause so much damming of blood in the lungs that
serious or even lethal pulmonary edema may ensue in
Pulmonary Edema in Mitral Valvular Disease. The buildup
as little as 10 minutes.
of blood in the left atrium causes progressive increase
Even in mild to moderate cases of valvular disease,
in left atrial pressure, and this eventually results in
the patient’s cardiac reserve diminishes in proportion
274
Unit IV The Circulation
to the severity of the valvular dysfunction. That is, the
Head and upper
cardiac output does not increase as much as it should
extremities
Ductus
during exercise. Therefore, the muscles of the body
arteriosus
Aorta
fatigue rapidly because of too little increase in muscle
Right
blood flow.
lung
Left lung
Abnormal Circulatory
Dynamics in Congenital
Heart Defects
Occasionally, the heart or its associated blood vessels
are malformed during fetal life; the defect is called a
congenital anomaly. There are three major types of
Trunk and lower
Pulmonary
Left
congenital anomalies of the heart and its associated
extremities
artery
pulmonary
vessels: (1) stenosis of the channel of blood flow at
artery
some point in the heart or in a closely allied major
Figure 23-4
blood vessel; (2) an anomaly that allows blood to flow
backward from the left side of the heart or aorta to the
Patent ductus arteriosus, showing by the intensity of the pink color
right side of the heart or pulmonary artery, thus failing
that dark venous blood changes into oxygenated blood at differ-
to flow through the systemic circulation—called a left-
ent points in the circulation. The right-hand diagram shows back-
flow of blood from the aorta into the pulmonary artery and then
to-right shunt; and (3) an anomaly that allows blood to
through the lungs for a second time.
flow directly from the right side of the heart into the
left side of the heart, thus failing to flow through the
lungs—called a right-to-left shunt.
The effects of the different stenotic lesions are easily
lungs. This lack of blood flow through the lungs is not
understood. For instance, congenital aortic valve steno-
detrimental to the fetus because the blood is oxy-
sis results in the same dynamic effects as aortic valve
genated by the placenta.
stenosis caused by other valvular lesions, namely, a
tendency to develop serious pulmonary edema and a
Closure of the Ductus Arteriosus After Birth. As soon as a
reduced cardiac output.
baby is born and begins to breathe, the lungs inflate;
Another type of congenital stenosis is coarctation
not only do the alveoli fill with air, but also the resist-
of the aorta, often occurring near the level of the
ance to blood flow through the pulmonary vascular
diaphragm. This causes the arterial pressure in the
tree decreases tremendously, allowing the pulmonary
upper part of the body (above the level of the coarc-
arterial pressure to fall. Simultaneously, the aortic
tation) to be much greater than the pressure in the
pressure rises because of sudden cessation of blood
lower body because of the great resistance to blood
flow from the aorta through the placenta. Thus, the
flow through the coarctation to the lower body; part of
pressure in the pulmonary artery falls, while that in
the blood must go around the coarctation through
the aorta rises. As a result, forward blood flow through
small collateral arteries, as discussed in Chapter 19.
the ductus arteriosus ceases suddenly at birth, and in
fact, blood begins to flow backward through the ductus
from the aorta into the pulmonary artery. This new
Patent Ductus Arteriosus—
state of backward blood flow causes the ductus arte-
A Left-to-Right Shunt
riosus to become occluded within a few hours to a few
days in most babies, so that blood flow through the
During fetal life, the lungs are collapsed, and the
ductus does not persist. The ductus is believed to close
elastic compression of the lungs that keeps the alveoli
because the oxygen concentration of the aortic blood
collapsed keeps most of the lung blood vessels col-
now flowing through it is about twice as high as that
lapsed as well. Therefore, resistance to blood flow
of the blood flowing from the pulmonary artery into
through the lungs is so great that the pulmonary arte-
the ductus during fetal life. The oxygen presumably
rial pressure is high in the fetus. Also, because of low
constricts the muscle in the ductus wall. This is dis-
resistance to blood flow from the aorta through the
cussed further in Chapter 83.
large vessels of the placenta, the pressure in the aorta
Unfortunately, in about 1 of every 5500 babies, the
of the fetus is lower than normal—in fact, lower than
ductus does not close, causing the condition known as
in the pulmonary artery. This causes almost all the pul-
patent ductus arteriosus, which is shown in Figure 23-4.
monary arterial blood to flow through a special artery
present in the fetus that connects the pulmonary artery
Dynamics of the Circulation with a Persistent Patent Ductus.
with the aorta (Figure 23-4), called the ductus arterio-
During the early months of an infant’s life, a patent
sus, thus bypassing the lungs. This allows immediate
ductus usually does not cause severely abnormal func-
recirculation of the blood through the systemic arter-
tion. But as the child grows older, the differential
ies of the fetus without the blood going through the
between the high pressure in the aorta and the lower
Chapter 23
Heart Valves and Heart Sounds; Dynamics of Valvular and Congenital Heart Defects
275
pressure in the pulmonary artery progressively in-
Head and upper
creases, with corresponding increase in backward flow
extremities
of blood from the aorta into the pulmonary artery.
Also, the high aortic blood pressure usually causes the
Right
diameter of the partially open ductus to increase with
lung
time, making the condition even worse.
Left lung
Recirculation Through the Lungs. In an older child
with a patent ductus, one half to two thirds of the aortic
blood flows backward through the ductus into the pul-
monary artery, then through the lungs, and finally back
into the left ventricle and aorta, passing through the
lungs and left side of the heart two or more times for
every one time that it passes through the systemic cir-
Trunk and lower
culation. These people do not show cyanosis until later
extremities
in life, when the heart fails or the lungs become con-
gested. Indeed, early in life, the arterial blood is often
better oxygenated than normal because of the extra
times it passes through the lungs.
Diminished Cardiac and Respiratory Reserve. The
major effects of patent ductus arteriosus on the patient
are decreased cardiac and respiratory reserve. The left
ventricle is pumping about two or more times the
Figure 23-5
normal cardiac output, and the maximum that it can
Tetralogy of Fallot, showing by the intensity of the pink color that
pump after hypertrophy of the heart has occurred is
most of the dark venous blood is shunted from the right ventricle
about four to seven times normal. Therefore, during
into the aorta without passing through the lungs.
exercise, the net blood flow through the remainder of
the body can never increase to the levels required for
strenuous activity. With even moderately strenuous
exercise, the person is likely to become weak and may
Tetralogy of Fallot—
even faint from momentary heart failure.
A Right-to-Left Shunt
The high pressures in the pulmonary vessels caused
by excess flow through the lungs often lead to pul-
Tetralogy of Fallot is shown in Figure 23-5; it is the
monary congestion and pulmonary edema. As a result
most common cause of “blue baby.” Most of the blood
of the excessive load on the heart, and especially
bypasses the lungs, so the aortic blood is mainly unoxy-
because the pulmonary congestion becomes progres-
genated venous blood. In this condition, four abnor-
sively more severe with age, most patients with uncor-
malities of the heart occur simultaneously:
rected patent ductus die from heart disease between
1. The aorta originates from the right ventricle
ages 20 and 40 years.
rather than the left, or it overrides a hole in the
septum, as shown in Figure 23-5, receiving blood
from both ventricles.
Heart Sounds: Machinery Murmur. In a newborn infant
2. The pulmonary artery is stenosed, so that much
with patent ductus arteriosus, occasionally no abnor-
lower than normal amounts of blood pass from
mal heart sounds are heard because the quantity of
the right ventricle into the lungs; instead, most of
reverse blood flow through the ductus may be insuffi-
the blood passes directly into the aorta, thus
cient to cause a heart murmur. But as the baby grows
bypassing the lungs.
older, reaching age 1 to 3 years, a harsh, blowing
3. Blood from the left ventricle flows either through
murmur begins to be heard in the pulmonary artery
a ventricular septal hole into the right ventricle
area of the chest, as shown in recording F, Figure 23-3.
and then into the aorta or directly into the aorta
This sound is much more intense during systole when
that overrides this hole.
the aortic pressure is high and much less intense
4. Because the right side of the heart must pump
during diastole when the aortic pressure falls low, so
large quantities of blood against the high pressure
that the murmur waxes and wanes with each beat of
in the aorta, its musculature is highly developed,
the heart, creating the so-called machinery murmur.
causing an enlarged right ventricle.
Surgical Treatment. Surgical treatment of patent ductus
Abnormal Circulatory Dynamics. It is readily apparent that
arteriosus is extremely simple; one need only ligate the
the major physiological difficulty caused by tetralogy
patent ductus or divide it and then close the two ends.
of Fallot is the shunting of blood past the lungs without
In fact, this was one of the first successful heart sur-
its becoming oxygenated. As much as 75 per cent of
geries ever performed.
the venous blood returning to the heart passes directly
276
Unit IV The Circulation
from the right ventricle into the aorta without becom-
passing the blood between thin membranes or through
ing oxygenated.
thin tubes that are permeable to oxygen and carbon
A diagnosis of tetralogy of Fallot is usually based on
dioxide.
(1) the fact that the baby’s skin is cyanotic (blue); (2)
The different systems have all been fraught with
measurement of high systolic pressure in the right
many difficulties, including hemolysis of the blood,
ventricle, recorded through a catheter; (3) charac-
development of small clots in the blood, likelihood of
teristic changes in the radiological silhouette of the
small bubbles of oxygen or small emboli of antifoam
heart, showing an enlarged right ventricle; and (4)
agent passing into the arteries of the patient, necessity
angiograms (x-ray pictures) showing abnormal blood
for large quantities of blood to prime the entire
flow through the interventricular septal hole and into
system, failure to exchange adequate quantities of
the overriding aorta, but much less flow through the
oxygen, and necessity to use heparin to prevent blood
stenosed pulmonary artery.
coagulation in the extracorporeal system. Heparin also
interferes with adequate hemostasis during the surgi-
cal procedure. Yet despite these difficulties, in the
Surgical Treatment. Tetralogy of Fallot can usually be
hands of experts, patients can be kept alive on artifi-
treated successfully by surgery. The usual operation is
cial heart-lung machines for many hours while opera-
to open the pulmonary stenosis, close the septal defect,
tions are performed on the inside of the heart.
and reconstruct the flow pathway into the aorta. When
surgery is successful, the average life expectancy
increases from only 3 to 4 years to 50 or more years.
Hypertrophy of the Heart
in Valvular and Congenital
Causes of Congenital Anomalies
Heart Disease
One of the most common causes of congenital heart
Hypertrophy of cardiac muscle is one of the most
defects is a viral infection in the mother during the first
important mechanisms by which the heart adapts to
trimester of pregnancy when the fetal heart is being
increased workloads, whether these loads are caused
formed. Defects are particularly prone to develop
by increased pressure against which the heart muscle
when the expectant mother contracts German
must contract or by increased cardiac output that
measles; thus, obstetricians often advise termination of
must be pumped. Some physicians believe that the
pregnancy if German measles occurs in the first
increased strength of contraction of the heart muscle
trimester.
causes the hypertrophy; others believe that the
Some congenital defects of the heart are hereditary,
increased metabolic rate of the muscle is the primary
because the same defect has been known to occur in
stimulus. Regardless of which of these is correct, one
identical twins as well as in succeeding generations.
can calculate approximately how much hypertrophy
Children of patients surgically treated for congenital
will occur in each chamber of the heart by multiplying
heart disease have about a 10 times greater chance of
ventricular output by the pressure against which the
having congenital heart disease than other children do.
ventricle must work, with emphasis on pressure. Thus,
Congenital defects of the heart are also frequently
hypertrophy occurs in most types of valvular and con-
associated with other congenital defects of the baby’s
genital disease, sometimes causing heart weights as
body.
great as 800 grams instead of the normal 300 grams.
Use of Extracorporeal
References
Circulation During
Arad M, Seidman JG, Seidman CE: Phenotypic diversity in
Cardiac Surgery
hypertrophic cardiomyopathy. Hum Mol Genet 11:2499,
2002.
It is almost impossible to repair intracardiac defects
Braunwald E, Seidman CE, Sigwart U: Contemporary eval-
surgically while the heart is still pumping. Therefore,
uation and management of hypertrophic cardiomyopathy.
many types of artificial heart-lung machines have been
Circulation 106:1312, 2002.
developed to take the place of the heart and lungs
Brickner ME, Hillis LD, Lange RA: Congenital heart disease
during the course of operation. Such a system is called
in adults: second of two parts. N Engl J Med 342:334, 2000.
extracorporeal circulation. The system consists princi-
Gottdiener JS: Overview of stress echocardiography: uses,
pally of a pump and an oxygenating device. Almost
advantages, and limitations. Curr Probl Cardiol 28:485,
2003.
any type of pump that does not cause hemolysis of the
Grech ED: Non-coronary percutaneous intervention. BMJ
blood seems to be suitable.
327:97, 2003.
Methods used for oxygenating blood include (1)
Guidelines for the management of patients with valvular
bubbling oxygen through the blood and removing the
heart disease. A report of the American College of Cardi-
bubbles from the blood before passing it back into the
ology/American Heart Association Task Force on Practice
patient, (2) dripping the blood downward over the sur-
Guidelines. Circulation 98:1949, 1998.
faces of plastic sheets in the presence of oxygen, (3)
Hoffman JI, Kaplan S: The incidence of congenital heart
passing the blood over surfaces of rotating discs, or (4)
disease. J Am Coll Cardiol 39:1890, 2002.
Chapter 23
Heart Valves and Heart Sounds; Dynamics of Valvular and Congenital Heart Defects
277
Levi DS, Alejos JC, Moore JW: Future of interventional car-
Rosenhek R, Burder T, Pozenta G, et al: Predictors of
diology in pediatrics. Curr Opin Cardiol 18:79, 2003.
outcome in severe, asymptomatic aortic stenosis. N Engl J
Maron BJ: Hypertrophic cardiomyopathy: a systematic
Med 343:611, 2000.
review. JAMA 287:1308, 2002.
Turgeman Y, Atar S, Rosenfeld T: The subvalvular apparatus
McDonald M, Currie BJ, Carapetis JR: Acute rheumatic
in rheumatic mitral stenosis: methods of assessment and
fever: a chink in the chain that links the heart to the
therapeutic implications. Chest 124:1929, 2003.
throat? Lancet Infect Dis 4:240, 2004.
Yacoub MH, Cohn LH: Novel approaches to cardiac valve
Nishimura RA, Holmes DR Jr: Clinical practice: hyper-
repair: from structure to function. Part II. Circulation
trophic obstructive cardiomyopathy. N Engl J Med 350:
109:942, 2004.
1320, 2004.
Yoerger DM, Weyman AE: Hypertrophic obstructive car-
Reimold SC, Rutherford JD: Clinical practice: valvular heart
diomyopathy: mechanism of obstruction and response to
disease in pregnancy. N Engl J Med 349:52, 2003.
therapy. Rev Cardiovasc Med 4:199, 2003.
C H A P T E R
2
4
Circulatory Shock and
Physiology of Its Treatment
Circulatory shock means generalized inadequate
blood flow through the body, to the extent that the
body tissues are damaged because of too little flow,
especially because of too little oxygen and other
nutrients delivered to the tissue cells. Even the car-
diovascular system itself—the heart musculature,
walls of the blood vessels, vasomotor system, and
other circulatory parts—begins to deteriorate, so
that the shock, once begun, is prone to become progressively worse.
Physiologic Causes of Shock
Circulatory Shock Caused by Decreased Cardiac Output
Shock usually results from inadequate cardiac output. Therefore, any condition
that reduces the cardiac output far below normal will likely lead to circulatory
shock. Two types of factors can severely reduce cardiac output:
1. Cardiac abnormalities that decrease the ability of the heart to pump blood.
These include especially myocardial infarction but also toxic states of
the heart, severe heart valve dysfunction, heart arrhythmias, and other
conditions. The circulatory shock that results from diminished cardiac
pumping ability is called cardiogenic shock. This is discussed in detail in
Chapter 22, where it is pointed out that as many as 85 per cent of people
who develop cardiogenic shock do not survive.
2. Factors that decrease venous return also decrease cardiac output because
the heart cannot pump blood that does not flow into it. The most common
cause of decreased venous return is diminished blood volume, but venous
return can also be reduced as a result of decreased vascular tone, especially
of the venous blood reservoirs, or obstruction to blood flow at some point
in the circulation, especially in the venous return pathway to the heart.
Circulatory Shock That Occurs Without Diminished
Cardiac Output
Occasionally, the cardiac output is normal or even greater than normal, yet the
person is in circulatory shock. This can result from (1) excessive metabolism of
the body, so that even a normal cardiac output is inadequate, or (2) abnormal
tissue perfusion patterns, so that most of the cardiac output is passing through
blood vessels besides those that supply the local tissues with nutrition.
The specific causes of shock are discussed later in the chapter. For the present,
it is important to note that all of them lead to inadequate delivery of nutrients
to critical tissues and critical organs and also cause inadequate removal of cellu-
lar waste products from the tissues.
278
Chapter 24
Circulatory Shock and Physiology of Its Treatment
279
What Happens to the Arterial Pressure
in Circulatory Shock?
Arterial
100
pressure
In the minds of many physicians, the arterial pressure
level is the principal measure of adequacy of circula-
tory function. However, the arterial pressure can often
be seriously misleading. At times, a person may be in
50
Cardiac
severe shock and still have an almost normal arterial
output
pressure because of powerful nervous reflexes that
keep the pressure from falling. At other times, the
arterial pressure can fall to half of normal, but the
0
person still has normal tissue perfusion and is not in
0
10
20
30
40
50
shock.
In most types of shock, especially shock caused by
Percentage of total blood removed
severe blood loss, the arterial blood pressure decreases
at the same time as the cardiac output decreases,
although usually not as much.
Figure 24-1
Effect of hemorrhage on cardiac output and arterial pressure.
Tissue Deterioration Is the End Result
Shock Caused by
of Circulatory Shock, Whatever
Hypovolemia—Hemorrhagic
the Cause
Shock
Once circulatory shock reaches a critical state of sever-
Hypovolemia means diminished blood volume. Hem-
ity, regardless of its initiating cause, the shock itself
orrhage is the most common cause of hypovolemic
breeds more shock. That is, the inadequate blood flow
shock. Hemorrhage decreases the filling pressure of the
causes the body tissues to begin deteriorating, includ-
circulation and, as a consequence, decreases venous
ing the heart and circulatory system itself. This causes
return. As a result, the cardiac output falls below
an even greater decrease in cardiac output, and a
normal, and shock may ensue.
vicious circle ensues, with progressively increasing cir-
culatory shock, less adequate tissue perfusion, more
shock, and so forth until death. It is with this late stage
Relationship of Bleeding Volume to
of circulatory shock that we are especially concerned,
Cardiac Output and Arterial Pressure
because appropriate physiologic treatment can often
reverse the rapid slide to death.
Figure 24-1 shows the approximate effects on both
cardiac output and arterial pressure of removing blood
from the circulatory system over a period of about 30
minutes. About 10 per cent of the total blood volume
Stages of Shock
can be removed with almost no effect on either arte-
rial pressure or cardiac output, but greater blood loss
Because the characteristics of circulatory shock
usually diminishes the cardiac output first and later the
change with different degrees of severity, shock is
arterial pressure, both of which fall to zero when about
divided into the following three major stages:
35 to 45 per cent of the total blood volume has been
1. A nonprogressive stage (sometimes called
removed.
the compensated stage), in which the normal
circulatory compensatory mechanisms eventually
Sympathetic Reflex Compensations in Shock—Their Special
cause full recovery without help from outside
Value to Maintain Arterial Pressure. The decrease in arte-
therapy.
rial pressure after hemorrhage—as well as decreases
2. A progressive stage, in which, without therapy,
in pressures in the pulmonary arteries and veins in the
the shock becomes steadily worse until death.
thorax—causes powerful sympathetic reflexes (initi-
3. An irreversible stage, in which the shock has
ated mainly by the arterial baroreceptors and other
progressed to such an extent that all forms of
vascular stretch receptors, as explained in Chapter 18).
known therapy are inadequate to save the
These reflexes stimulate the sympathetic vasoconstric-
person’s life, even though, for the moment, the
tor system throughout the body, resulting in three
person is still alive.
important effects: (1) The arterioles constrict in most
Now, let us discuss the stages of circulatory shock
parts of the systemic circulation, thereby increasing
caused by decreased blood volume, which illustrate the
the total peripheral resistance.
(2) The veins and
basic principles. Then we can consider special charac-
venous reservoirs constrict, thereby helping to main-
teristics of shock initiated by other causes.
tain adequate venous return despite diminished blood
280
Unit IV The Circulation
I
100
90
II
80
III
70
IV
60
50
V
40
30
20
VI
10
0
Figure 24-2
0
60
120
180
240
300
360
Time in minutes
Time course of arterial pressure in dogs after dif-
ferent degrees of acute hemorrhage. Each curve
represents average results from six dogs.
volume. (3) Heart activity increases markedly, some-
The sympathetic stimulation does not cause significant
times increasing the heart rate from the normal value
constriction of either the cerebral or the cardiac
of 72 beats/min to as high as 160 to 180 beats/min.
vessels. In addition, in both these vascular beds, local
blood flow autoregulation is excellent, which prevents
Value of the Sympathetic Nervous Reflexes. In the
moderate decreases in arterial pressure from signifi-
absence of the sympathetic reflexes, only 15 to 20 per
cantly decreasing their blood flows. Therefore, blood
cent of the blood volume can be removed over a
flow through the heart and brain is maintained essen-
period of 30 minutes before a person dies; this is in
tially at normal levels as long as the arterial pressure
contrast to a 30 to 40 per cent loss of blood volume
does not fall below about 70 mm Hg, despite the fact
that a person can sustain when the reflexes are intact.
that blood flow in some other areas of the body might
Therefore, the reflexes extend the amount of blood
be decreased to as little as one third to one quarter
loss that can occur without causing death to about
normal by this time because of vasoconstriction.
twice that which is possible in their absence.
Greater Effect of the Sympathetic Nervous Reflexes in
Progressive and Nonprogressive
Maintaining Arterial Pressure than in Maintaining
Hemorrhagic Shock
Cardiac Output. Referring again to Figure 24-1, note
that the arterial pressure is maintained at or near
Figure 24-2 shows an experiment that we performed
normal levels in the hemorrhaging person longer than
in dogs to demonstrate the effects of different degrees
is the cardiac output. The reason for this is that the
of sudden acute hemorrhage on the subsequent course
sympathetic reflexes are geared more for maintaining
of arterial pressure. The dogs were bled rapidly until
arterial pressure than for maintaining output. They
their arterial pressures fell to different levels. Those
increase the arterial pressure mainly by increasing the
dogs whose pressures fell immediately to no lower
total peripheral resistance, which has no beneficial
than 45 mm Hg (groups I, II, and III) all eventually
effect on cardiac output; however, the sympathetic con-
recovered; the recovery occurred rapidly if the pres-
striction of the veins is important to keep venous return
sure fell only slightly (group I) but occurred slowly if
and cardiac output from falling too much, in addition
it fell almost to the 45 mm Hg level (group III). When
to their role in maintaining arterial pressure.
the arterial pressure fell below 45 mm Hg (groups IV,
Especially interesting is the second plateau occur-
V, and VI), all the dogs died, although many of them
ring at about 50 mm Hg in the arterial pressure curve
hovered between life and death for hours before the
of Figure 24-1. This results from activation of the
circulatory system deteriorated to the stage of death.
central nervous system ischemic response, which
This experiment demonstrates that the circulatory
causes extreme stimulation of the sympathetic nervous
system can recover as long as the degree of hemor-
system when the brain begins to suffer from lack of
rhage is no greater than a certain critical amount.
oxygen or from excess buildup of carbon dioxide, as
Crossing this critical threshold by even a few milliliters
discussed in Chapter 18. This effect of the central
of blood loss makes the eventual difference between
nervous system ischemic response can be called the
life and death. Thus, hemorrhage beyond a certain crit-
“last-ditch stand” of the sympathetic reflexes in their
ical level causes shock to become progressive. That is,
attempt to keep the arterial pressure from falling too
the shock itself causes still more shock, and the condi-
low.
tion becomes a vicious circle that eventually leads to
deterioration of the circulation and to death.
Protection of Coronary and Cerebral Blood Flow by
the Reflexes. A special value of the maintenance
Nonprogressive Shock—Compensated Shock
of normal arterial pressure even in the presence of
If shock is not severe enough to cause its own pro-
decreasing cardiac output is protection of blood flow
gression, the person eventually recovers. Therefore,
through the coronary and cerebral circulatory systems.
shock of this lesser degree is called nonprogressive
Chapter 24
Circulatory Shock and Physiology of Its Treatment
281
shock. It is also called compensated shock, meaning
the shock to become progressive. Some of the more
that the sympathetic reflexes and other factors com-
important feedbacks are the following.
pensate enough to prevent further deterioration of the
circulation.
Cardiac Depression. When the arterial pressure falls low
The factors that cause a person to recover from
enough, coronary blood flow decreases below that
moderate degrees of shock are all the negative feed-
required for adequate nutrition of the myocardium.
back control mechanisms of the circulation that
This weakens the heart muscle and thereby decreases
attempt to return cardiac output and arterial pressure
the cardiac output more. Thus, a positive feedback
back to normal levels. They include the following:
cycle has developed, whereby the shock becomes more
1.
Baroreceptor reflexes, which elicit powerful
and more severe.
sympathetic stimulation of the circulation.
Figure 24-4 shows cardiac output curves extrapo-
2.
Central nervous system ischemic response,
lated to the human heart from experiments in dogs,
which elicits even more powerful sympathetic
demonstrating progressive deterioration of the heart
stimulation throughout the body but is not
at different times after the onset of shock. A dog was
activated significantly until the arterial pressure
bled until the arterial pressure fell to 30 mm Hg, and
falls below 50 mm Hg.
the pressure was held at this level by further bleeding
3.
Reverse stress-relaxation of the circulatory system,
or retransfusion of blood as required. Note from the
which causes the blood vessels to contract around
second curve in the figure that there was little deteri-
the diminished blood volume, so that the blood
oration of the heart during the first 2 hours, but by 4
volume that is available more adequately fills the
hours, the heart had deteriorated about 40 per cent;
circulation.
then, rapidly, during the last hour of the experiment
4.
Formation of angiotensin by the kidneys, which
(after 4 hours of low coronary blood pressure), the
constricts the peripheral arteries and also causes
heart deteriorated completely.
decreased output of water and salt by the
Thus, one of the important features of progressive
kidneys, both of which help prevent progression
shock, whether it is hemorrhagic in origin or caused in
of shock.
another way, is eventual progressive deterioration of
5.
Formation of vasopressin (antidiuretic hormone)
the heart. In the early stages of shock, this plays very
by the posterior pituitary gland, which constricts
little role in the condition of the person, partly because
the peripheral arteries and veins and greatly
deterioration of the heart is not severe during the first
increases water retention by the kidneys.
hour or so of shock, but mainly because the heart has
6.
Compensatory mechanisms that return the blood
tremendous reserve capability that normally allows
volume back toward normal, including absorption
it to pump 300 to 400 per cent more blood than is
of large quantities of fluid from the intestinal
required by the body for adequate bodywide tissue
tract, absorption of fluid into the blood capillaries
nutrition. In the latest stages of shock, however, dete-
from the interstitial spaces of the body,
rioration of the heart is probably the most important
conservation of water and salt by the kidneys, and
factor in the final lethal progression of the shock.
increased thirst and increased appetite for salt,
which make the person drink water and eat salty
Vasomotor Failure. In the early stages of shock, various
foods if able.
circulatory reflexes cause intense activity of the
The sympathetic reflexes provide immediate help
sympathetic nervous system. This, as discussed earlier,
toward bringing about recovery because they become
helps delay depression of the cardiac output and
maximally activated within 30 seconds to a minute
especially helps prevent decreased arterial pressure.
after hemorrhage.
However, there comes a point when diminished blood
The angiotensin and vasopressin mechanisms, as
flow to the brain’s vasomotor center depresses the
well as the reverse stress-relaxation that causes
center so much that it, too, becomes progressively less
contraction of the blood vessels and venous reservoirs,
active and finally totally inactive. For instance, com-
all require 10 minutes to 1 hour to respond completely,
plete circulatory arrest to the brain causes, during the
but they aid greatly in increasing the arterial pressure
first 4 to 8 minutes, the most intense of all sympathetic
or increasing the circulatory filling pressure and
discharges, but by the end of 10 to 15 minutes, the vaso-
thereby increasing the return of blood to the heart.
motor center becomes so depressed that no further
Finally, readjustment of blood volume by absorption
evidence of sympathetic discharge can be demon-
of fluid from the interstitial spaces and intestinal tract,
strated. Fortunately, the vasomotor center usually does
as well as oral ingestion and absorption of additional
not fail in the early stages of shock if the arterial
quantities of water and salt, may require from 1 to 48
pressure remains above 30 mm Hg.
hours, but recovery eventually takes place, provided
the shock does not become severe enough to enter the
Blockage of Very Small Vessels—“Sludged Blood.” In time,
progressive stage.
blockage occurs in many of the very small blood
vessels in the circulatory system, and this also causes
“Progressive Shock” Is Caused by a Vicious
the shock to progress. The initiating cause of this
Circle of Cardiovascular Deterioration
blockage is sluggish blood flow in the microvessels.
Figure 24-3 shows some of the positive feedbacks that
Because tissue metabolism continues despite the low
further depress cardiac output in shock, thus causing
flow, large amounts of acid, both carbonic acid and
282
Unit IV The Circulation
Decreased cardiac output
Decreased arterial pressure
Decreased systemic blood flow
Decreased cardiac nutrition
Decreased nutrition of tissues
Intravascular clotting
Decreased nutrition of brain
Decreased nutrition
Tissue ischemia
of vascular system
Decreased vasomotor
activity
Increased
Release of
capillary
toxins
permeability
Vascular dilation
Decreased
Venous pooling
blood volume
of blood
Cardiac depression
Decreased venous return
Figure 24-3
Different types of “positive feedback” that can lead to progression of shock.
lactic acid, continue to empty into the local blood
Release of Toxins by Ischemic Tissue. Throughout the
vessels and greatly increase the local acidity of the
history of research in the field of shock, it has been
blood. This acid, plus other deterioration products
suggested that shock causes tissues to release toxic
from the ischemic tissues, causes local blood aggluti-
substances, such as histamine, serotonin, and tissue
nation, resulting in minute blood clots, leading to very
enzymes, that cause further deterioration of the circu-
small plugs in the small vessels. Even if the vessels do
latory system. Quantitative studies have proved the
not become plugged, an increased tendency for the
significance of at least one toxin, endotoxin, in some
blood cells to stick to one another makes it more dif-
types of shock.
ficult for blood to flow through the microvasculature,
giving rise to the term sludged blood.
Cardiac Depression Caused by Endotoxin. Endotoxin
is released from the bodies of dead gram-negative
Increased Capillary Permeability. After many hours of
bacteria in the intestines. Diminished blood flow to
capillary hypoxia and lack of other nutrients, the
the intestines often causes enhanced formation and
permeability of the capillaries gradually increases,
absorption of this toxic substance. The circulating
and large quantities of fluid begin to transude into the
toxin then causes increased cellular metabolism
tissues. This decreases the blood volume even more,
despite inadequate nutrition of the cells; this has a spe-
with a resultant further decrease in cardiac output,
cific effect on the heart muscle, causing cardiac depres-
making the shock still more severe. Capillary hypoxia
sion. Endotoxin can play a major role in some types
does not cause increased capillary permeability until
of shock, especially “septic shock,” discussed later in
the late stages of prolonged shock.
the chapter.
Chapter 24
Circulatory Shock and Physiology of Its Treatment
283
15
0 time
2 hours
10
4 hours
41/2 hours
5
43/4 hours
5 hours
0
–4
0
4
8
12
Right atrial pressure (mm Hg)
Figure 24-4
Cardiac output curves of the heart at different times after hemor-
rhagic shock begins. (These curves are extrapolated to the human
heart from data obtained in dog experiments by Dr. J. W. Crowell.)
Figure 24-5
Necrosis of the central portion of a liver lobule in severe circula-
tory shock. (Courtesy Dr. J. W. Crowell.)
Generalized Cellular Deterioration. As shock becomes
severe, many signs of generalized cellular deteriora-
tion occur throughout the body. One organ especially
Tissue Necrosis in Severe Shock—Patchy Areas of
affected is the liver, as illustrated in Figure 24-5. This
Necrosis Occur Because of Patchy Blood Flows in
occurs mainly because of lack of enough nutrients to
Different Organs. Not all cells of the body are equally
support the normally high rate of metabolism in liver
damaged by shock, because some tissues have better
cells, but also partly because of the extreme exposure
blood supplies than others. For instance, the cells adja-
of the liver cells to any vascular toxin or other abnor-
cent to the arterial ends of capillaries receive better
mal metabolic factor occurring in shock.
nutrition than cells adjacent to the venous ends of the
Among the damaging cellular effects that are
same capillaries. Therefore, one would expect more
known to occur in most body tissues are the following:
nutritive deficiency around the venous ends of capil-
1. Active transport of sodium and potassium
laries than elsewhere. This is precisely the effect that
through the cell membrane is greatly diminished.
Crowell discovered in studying tissue areas in many
As a result, sodium and chloride accumulate in
parts of the body. For instance, Figure 24-5 shows
the cells, and potassium is lost from the cells. In
necrosis in the center of a liver lobule, the portion of
addition, the cells begin to swell.
the lobule that is last to be exposed to the blood as it
2. Mitochondrial activity in the liver cells, as well as
passes through the liver sinusoids.
in many other tissues of the body, becomes
Similar punctate lesions occur in heart muscle,
severely depressed.
although here a definite repetitive pattern, such as
3. Lysosomes in the cells in widespread tissue areas
occurs in the liver, cannot be demonstrated. Never-
begin to break open, with intracellular release of
theless, the cardiac lesions play an important role in
hydrolases that cause further intracellular
leading to the final irreversible stage of shock. Deteri-
deterioration.
orative lesions also occur in the kidneys, especially in
4. Cellular metabolism of nutrients, such as glucose,
the epithelium of the kidney tubules, leading to kidney
eventually becomes greatly depressed in the last
failure and occasionally uremic death several days
stages of shock. The actions of some hormones
later. Deterioration of the lungs also often leads to res-
are depressed as well, including almost 100 per
piratory distress and death several days later—called
cent depression of the action of insulin.
the shock lung syndrome.
All these effects contribute to further deterioration
of many organs of the body, including especially (1)
Acidosis in Shock. Most metabolic derangements that
the liver, with depression of its many metabolic and
occur in shocked tissue can lead to blood acidosis all
detoxification functions; (2) the lungs, with eventual
through the body. This results from poor delivery of
development of pulmonary edema and poor ability to
oxygen to the tissues, which greatly diminishes oxida-
oxygenate the blood; and (3) the heart, thereby further
tive metabolism of the foodstuffs. When this occurs,
depressing its contractility.
the cells obtain most of their energy by the anaerobic
284
Unit IV The Circulation
process of glycolysis, which leads to tremendous quan-
tities of excess lactic acid in the blood. In addition, poor
blood flow through tissues prevents normal removal
100
of carbon dioxide. The carbon dioxide reacts locally
in the cells with water to form high concentrations of
75
intracellular carbonic acid; this, in turn, reacts with
Progressive
Transfusion
stage
various tissue chemicals to form still other intracellu-
50
lar acidic substances. Thus, another deteriorative effect
25
of shock is both generalized and local tissue acidosis,
leading to further progression of the shock itself.
Irreversible shock
0
0
30
60
90
120
150
Positive Feedback Deterioration of Tissues
Minutes
in Shock and the Vicious Circle of
Progressive Shock
All the factors just discussed that can lead to further
Figure 24-6
progression of shock are types of positive feedback.
That is, each increase in the degree of shock causes a
Failure of transfusion to prevent death in irreversible shock.
further increase in the shock.
However, positive feedback does not necessarily
lead to a vicious circle. Whether a vicious circle devel-
the heart’s immediate ability to pump blood but, over
ops depends on the intensity of the positive feedback.
a long period, depress heart pumping enough to cause
In mild degrees of shock, the negative feedback mech-
death. Beyond a certain point, so much tissue damage
anisms of the circulation—sympathetic reflexes,
has occurred, so many destructive enzymes have been
reverse stress-relaxation mechanism of the blood
released into the body fluids, so much acidosis has
reservoirs, absorption of fluid into the blood from the
developed, and so many other destructive factors are
interstitial spaces, and others—can easily overcome
now in progress that even a normal cardiac output for
the positive feedback influences and, therefore, cause
a few minutes cannot reverse the continuing deterio-
recovery. But in severe degrees of shock, the deterio-
ration. Therefore, in severe shock, a stage is eventually
rative feedback mechanisms become more and more
reached at which the person will die even though vig-
powerful, leading to such rapid deterioration of the
orous therapy might still return the cardiac output to
circulation that all the normal negative feedback
normal for short periods.
systems of circulatory control acting together cannot
return the cardiac output to normal.
Depletion of Cellular High-Energy Phosphate Reserves in
Considering once again the principles of positive
Irreversible Shock. The high-energy phosphate reserves
feedback and vicious circle discussed in Chapter 1, one
in the tissues of the body, especially in the liver and
can readily understand why there is a critical cardiac
the heart, are greatly diminished in severe degrees of
output level above which a person in shock recovers
shock. Essentially all the creatine phosphate has been
and below which a person enters a vicious circle of cir-
degraded, and almost all the adenosine triphosphate
culatory deterioration that proceeds until death.
has downgraded to adenosine diphosphate, adenosine
monophosphate, and, eventually, adenosine. Then
much of this adenosine diffuses out of the cells into the
Irreversible Shock
circulating blood and is converted into uric acid, a
substance that cannot re-enter the cells to reconstitute
After shock has progressed to a certain stage, transfu-
the adenosine phosphate system. New adenosine can
sion or any other type of therapy becomes incapable
be synthesized at a rate of only about 2 per cent of
of saving the person’s life. The person is then said to
the normal cellular amount an hour, meaning that
be in the irreversible stage of shock. Ironically, even in
once the high-energy phosphate stores of the cells are
this irreversible stage, therapy can, on rare occasions,
depleted, they are difficult to replenish.
return the arterial pressure and even the cardiac
Thus, one of the most devastating end results of
output to normal or near normal for short periods, but
deterioration in shock, and the one that is perhaps
the circulatory system nevertheless continues to dete-
most significant for development of the final state of
riorate, and death ensues in another few minutes to
irreversibility, is this cellular depletion of these high-
few hours.
energy compounds.
Figure 24-6 demonstrates this effect, showing that
transfusion during the irreversible stage can some-
times cause the cardiac output (as well as the arterial
Hypovolemic Shock Caused
pressure) to return to normal. However, the cardiac
by Plasma Loss
output soon begins to fall again, and subsequent trans-
fusions have less and less effect. By this time, multiple
Loss of plasma from the circulatory system, even
deteriorative changes have occurred in the muscle
without loss of red blood cells, can sometimes be
cells of the heart that may not necessarily affect
severe enough to reduce the total blood volume
Chapter 24
Circulatory Shock and Physiology of Its Treatment
285
markedly, causing typical hypovolemic shock similar in
Neurogenic Shock—Increased
almost all details to that caused by hemorrhage. Severe
Vascular Capacity
plasma loss occurs in the following conditions:
1. Intestinal obstruction is often a cause of severely
Shock occasionally results without any loss of blood
reduced plasma volume. Distention of the
volume. Instead, the vascular capacity increases so
intestine in intestinal obstruction partly blocks
much that even the normal amount of blood becomes
venous blood flow in the intestinal walls, which
incapable of filling the circulatory system adequately.
increases intestinal capillary pressure. This in turn
One of the major causes of this is sudden loss of vaso-
causes fluid to leak from the capillaries into the
motor tone throughout the body, resulting especially in
intestinal walls and also into the intestinal lumen.
massive dilation of the veins. The resulting condition
Because the lost fluid has a high protein content,
is known as neurogenic shock.
the result is reduced total blood plasma protein as
The role of vascular capacity in helping to regulate
well as reduced plasma volume.
circulatory function was discussed in Chapter
15,
2. In almost all patients who have severe burns or
where it was pointed out that either an increase in vas-
other denuding conditions of the skin, so much
cular capacity or a decrease in blood volume reduces
plasma is lost through the denuded skin areas
the mean systemic filling pressure, which reduces
that the plasma volume becomes markedly
venous return to the heart. Diminished venous return
reduced.
caused by vascular dilation is called venous pooling of
The hypovolemic shock that results from plasma
blood.
loss has almost the same characteristics as the shock
caused by hemorrhage, except for one additional com-
Causes of Neurogenic Shock. Some neurogenic factors
plicating factor: the blood viscosity increases greatly as
that can cause loss of vasomotor tone include the
a result of increased red blood cell concentration in
following:
the remaining blood, and this exacerbates the slug-
1. Deep general anesthesia often depresses the
gishness of blood flow.
vasomotor center enough to cause vasomotor
Loss of fluid from all fluid compartments of the
paralysis, with resulting neurogenic shock.
body is called dehydration; this, too, can reduce the
2. Spinal anesthesia, especially when this extends all
blood volume and cause hypovolemic shock similar to
the way up the spinal cord, blocks the sympathetic
that resulting from hemorrhage. Some of the causes of
nervous outflow from the nervous system and can
this type of shock are (1) excessive sweating, (2) fluid
be a potent cause of neurogenic shock.
loss in severe diarrhea or vomiting, (3) excess loss of
3. Brain damage is often a cause of vasomotor
fluid by nephrotic kidneys, (4) inadequate intake of
paralysis. Many patients who have had brain
fluid and electrolytes, or (5) destruction of the adrenal
concussion or contusion of the basal regions of
cortices, with loss of aldosterone secretion and conse-
the brain develop profound neurogenic shock.
quent failure of the kidneys to reabsorb sodium,
Also, even though brain ischemia for a few
chloride, and water, which occurs in the absence of
minutes almost always causes extreme vasomotor
the adrenocortical hormone aldosterone.
stimulation, prolonged ischemia (lasting longer
than 5 to 10 minutes) can cause the opposite
effect—total inactivation of the vasomotor
neurons in the brain stem, with consequent
Hypovolemic Shock Caused
development of severe neurogenic shock.
by Trauma
One of the most common causes of circulatory shock
Anaphylactic Shock and
is trauma to the body. Often the shock results simply
from hemorrhage caused by the trauma, but it can also
Histamine Shock
occur even without hemorrhage, because extensive
contusion of the body can damage the capillaries
Anaphylaxis is an allergic condition in which the
sufficiently to allow excessive loss of plasma into the
cardiac output and arterial pressure often decrease
tissues. This results in greatly reduced plasma volume,
drastically. This is discussed in Chapter 34. It results
with resultant hypovolemic shock.
primarily from an antigen-antibody reaction that takes
Various attempts have been made to implicate toxic
place immediately after an antigen to which the person
factors released by the traumatized tissues as one of
is sensitive enters the circulation. One of the principal
the causes of shock after trauma. However, cross-
effects is to cause the basophils in the blood and mast
transfusion experiments into normal animals have
cells in the pericapillary tissues to release histamine or
failed to show significant toxic elements.
a histamine-like substance. The histamine causes (1) an
In summary, traumatic shock seems to result
increase in vascular capacity because of venous dila-
mainly from hypovolemia, although there might also
tion, thus causing a marked decrease in venous return;
be a moderate degree of concomitant neurogenic
(2) dilation of the arterioles, resulting in greatly
shock caused by loss of vasomotor tone, as discussed
reduced arterial pressure; and (3) greatly increased
next.
capillary permeability, with rapid loss of fluid and
286
Unit IV The Circulation
protein into the tissue spaces. The net effect is a great
blood clotting factors to be used up, so that
reduction in venous return and sometimes such
hemorrhaging occurs in many tissues, especially in
serious shock that the person dies within minutes.
the gut wall of the intestinal tract.
Intravenous injection of large amounts of histamine
In early stages of septic shock, the patient usually
causes “histamine shock,” which has characteristics
does not have signs of circulatory collapse but only
almost identical to those of anaphylactic shock.
signs of the bacterial infection. As the infection
becomes more severe, the circulatory system usually
becomes involved either because of direct extension
Septic Shock
of the infection or secondarily as a result of toxins
from the bacteria, with resultant loss of plasma into the
A condition that was formerly known by the popular
infected tissues through deteriorating blood capillary
name “blood poisoning” is now called septic shock
walls. There finally comes a point at which deteriora-
by most clinicians. This refers to a bacterial infection
tion of the circulation becomes progressive in the same
widely disseminated to many areas of the body, with
way that progression occurs in all other types of shock.
the infection being borne through the blood from one
The end stages of septic shock are not greatly differ-
tissue to another and causing extensive damage. There
ent from the end stages of hemorrhagic shock, even
are many varieties of septic shock because of the many
though the initiating factors are markedly different in
types of bacterial infections that can cause it and
the two conditions.
because infection in different parts of the body
produces different effects.
Septic shock is extremely important to the clinician,
Physiology of Treatment
because other than cardiogenic shock, septic shock is
in Shock
the most frequent cause of shock-related death in the
modern hospital.
Replacement Therapy
Some of the typical causes of septic shock include
the following:
Blood and Plasma Transfusion. If a person is in shock
1. Peritonitis caused by spread of infection from the
caused by hemorrhage, the best possible therapy is
uterus and fallopian tubes, sometimes resulting
usually transfusion of whole blood. If the shock is
from instrumental abortion performed under
caused by plasma loss, the best therapy is administra-
unsterile conditions.
tion of plasma; when dehydration is the cause, admin-
2. Peritonitis resulting from rupture of the
istration of an appropriate electrolyte solution can
gastrointestinal system, sometimes caused by
correct the shock.
intestinal disease and sometimes by wounds.
Whole blood is not always available, such as under
3. Generalized bodily infection resulting from
battlefield conditions. Plasma can usually substitute
spread of a skin infection such as streptococcal
adequately for whole blood because it increases the
or staphylococcal infection.
blood volume and restores normal hemodynamics.
4. Generalized gangrenous infection resulting
Plasma cannot restore a normal hematocrit, but the
specifically from gas gangrene bacilli, spreading
human body can usually stand a decrease in hemat-
first through peripheral tissues and finally by way
ocrit to about half of normal before serious con-
of the blood to the internal organs, especially the
sequences result, if cardiac output is adequate.
liver.
Therefore, in emergency conditions, it is reasonable to
5. Infection spreading into the blood from the
use plasma in place of whole blood for treatment of
kidney or urinary tract, often caused by colon
hemorrhagic or most other types of hypovolemic
bacilli.
shock.
Sometimes plasma is unavailable. In these instances,
Special Features of Septic Shock. Because of the multiple
various plasma substitutes have been developed that
types of septic shock, it is difficult to categorize this
perform almost exactly the same hemodynamic func-
condition. Some features often observed are:
tions as plasma. One of these is dextran solution.
1. High fever.
2. Often marked vasodilation throughout the body,
especially in the infected tissues.
Dextran Solution as a Plasma Substitute. The principal
3. High cardiac output in perhaps half of patients,
requirement of a truly effective plasma substitute is
caused by arteriolar dilation in the infected tissues
that it remain in the circulatory system—that is, not
and by high metabolic rate and vasodilation
filter through the capillary pores into the tissue spaces.
elsewhere in the body, resulting from bacterial
In addition, the solution must be nontoxic and must
toxin stimulation of cellular metabolism and from
contain appropriate electrolytes to prevent derange-
high body temperature.
ment of the body’s extracellular fluid electrolytes on
4. Sludging of the blood, caused by red cell
administration.
agglutination in response to degenerating tissues.
To remain in the circulation, the plasma substitute
5. Development of micro-blood clots in widespread
must contain some substance that has a large enough
areas of the body, a condition called disseminated
molecular size to exert colloid osmotic pressure. One
intravascular coagulation. Also, this causes the
of the most satisfactory substances developed for this
Chapter 24
Circulatory Shock and Physiology of Its Treatment
287
purpose is dextran, a large polysaccharide polymer of
the tissues, giving the patient oxygen to breathe can be
glucose. Certain bacteria secrete dextran as a by-
of benefit in many instances. However, this frequently
product of their growth, and commercial dextran can
is far less beneficial than one might expect, because the
be manufactured using a bacterial culture procedure.
problem in most types of shock is not inadequate oxy-
By varying the growth conditions of the bacteria, the
genation of the blood by the lungs but inadequate
molecular weight of the dextran can be controlled to
transport of the blood after it is oxygenated.
the desired value. Dextrans of appropriate molecular
size do not pass through the capillary pores and, there-
Treatment with Glucocorticoids (Adrenal Cortex Hormones That
fore, can replace plasma proteins as colloid osmotic
Control Glucose Metabolism). Glucocorticoids are fre-
agents.
quently given to patients in severe shock for several
Few toxic reactions have been observed when using
reasons: (1) experiments have shown empirically that
purified dextran to provide colloid osmotic pressure;
glucocorticoids frequently increase the strength of the
therefore, solutions containing this substance have
heart in the late stages of shock; (2) glucocorticoids
proved to be a satisfactory substitute for plasma in
stabilize lysosomes in tissue cells and thereby prevent
most fluid replacement therapy.
release of lysosomal enzymes into the cytoplasm of the
cells, thus preventing deterioration from this source;
and (3) glucocorticoids might aid in the metabolism of
Treatment of Shock with
glucose by the severely damaged cells.
Sympathomimetic Drugs—Sometimes
Useful, Sometimes Not
Circulatory Arrest
A sympathomimetic drug is a drug that mimics sym-
pathetic stimulation. These drugs include norepineph-
A condition closely allied to circulatory shock is cir-
rine, epinephrine, and a large number of long-acting
culatory arrest, in which all blood flow stops. This
drugs that have the same effect as epinephrine and
occurs frequently on the surgical operating table as a
norepinephrine.
result of cardiac arrest or ventricular fibrillation.
In two types of shock, sympathomimetic drugs have
Ventricular fibrillation can usually be stopped by
proved to be especially beneficial. The first of these is
strong electroshock of the heart, the basic principles
neurogenic shock, in which the sympathetic nervous
of which are described in Chapter 13.
system is severely depressed. Administering a sympa-
Cardiac arrest often results from too little oxygen in
thomimetic drug takes the place of the diminished
the anesthetic gaseous mixture or from a depressant
sympathetic actions and can often restore full circula-
effect of the anesthesia itself. A normal cardiac rhythm
tory function.
can usually be restored by removing the anesthetic and
The second type of shock in which sympatho-
immediately applying cardiopulmonary resuscitation
mimetic drugs are valuable is anaphylactic shock, in
procedures, while at the same time supplying the
which excess histamine plays a prominent role. The
patient’s lungs with adequate quantities of ventilatory
sympathomimetic drugs have a vasoconstrictor effect
oxygen.
that opposes the vasodilating effect of histamine.
Therefore, either norepinephrine or another sympa-
thomimetic drug is often lifesaving.
Effect of Circulatory Arrest
Sympathomimetic drugs have not proved to be very
on the Brain
valuable in hemorrhagic shock. The reason is that in
this type of shock, the sympathetic nervous system is
A special problem in circulatory arrest is to prevent
almost always maximally activated by the circulatory
detrimental effects in the brain as a result of the arrest.
reflexes already; so much norepinephrine and epi-
In general, more than 5 to 8 minutes of total circula-
nephrine are already circulating in the blood that sym-
tory arrest can cause at least some degree of perma-
pathomimetic drugs have essentially no additional
nent brain damage in more than half of patients.
beneficial effect.
Circulatory arrest for as long as 10 to 15 minutes
almost always permanently destroys significant
amounts of mental power.
Other Therapy
For many years, it was taught that this detrimental
effect on the brain was caused by the acute cerebral
Treatment by the Head-Down Position. When the pressure
hypoxia that occurs during circulatory arrest.
falls too low in most types of shock, especially in
However, experiments have shown that if blood clots
hemorrhagic and neurogenic shock, placing the patient
are prevented from occurring in the blood vessels of
with the head at least 12 inches lower than the feet
the brain, this will also prevent most of the early dete-
helps tremendously in promoting venous return,
rioration of the brain during circulatory arrest. For
thereby also increasing cardiac output. This head-
instance, in animal experiments performed by Crowell,
down position is the first essential step in the treat-
all the blood was removed from the animal’s blood
ment of many types of shock.
vessels at the beginning of circulatory arrest and
Oxygen Therapy. Because the major deleterious effect of
then replaced at the end of circulatory arrest so that
most types of shock is too little delivery of oxygen to
no intravascular blood clotting could occur. In this
288
Unit IV The Circulation
experiment, the brain was usually able to withstand up
Goodnough LT, Shander A: Evolution in alternatives to
to 30 minutes of circulatory arrest without permanent
blood transfusion. Hematol J 4:87, 2003.
brain damage. Also, administration of heparin or strep-
Granger DN, Stokes KY, Shigematsu T, et al: Splanchnic
ischaemia-reperfusion injury: mechanistic insights pro-
tokinase (to prevent blood coagulation) before cardiac
vided by mutant mice. Acta Physiol Scand 173:83, 2001.
arrest was shown to increase the survivability of the
Guyton AC, Jones CE, Coleman TG: Circulatory Physiology:
brain up to two to four times longer than usual.
Cardiac Output and Its Regulation. Philadelphia: WB
It is likely that the severe brain damage that occurs
Saunders, 1973.
from circulatory arrest is caused mainly by permanent
Ledgerwood AM, Lucas CE: A review of studies on the
blockage of many small blood vessels by blood clots,
effects of hemorrhagic shock and resuscitation on the
thus leading to prolonged ischemia and eventual death
coagulation profile. J Trauma 54(5 Suppl):S68, 2003.
of the neurons.
Martin GS, Mannino DM, Eaton S, Moss M: The epidemiol-
ogy of sepsis in the United States from 1979 through 2000.
N Engl J Med 348:1546, 2003.
McLean-Tooke AP, Bethune CA, Fay AC, Spickett GP:
References
Adrenaline in the treatment of anaphylaxis: what is the
evidence? BMJ 327:1332, 2003.
Annane D, Sebille V, Charpentier C, et al: Effect of treat-
Proctor KG: Blood substitutes and experimental models of
ment with low doses of hydrocortisone and fludrocorti-
trauma. J Trauma 54(5 Suppl):S106, 2003.
sone on mortality in patients with septic shock. JAMA
Rivers E, Nguyen B, Havstad S, et al: The Early Goal-
288:862, 2002.
Directed Therapy Collaborative Group. Early goal-
Burry LD, Wax RS: Role of corticosteroids in septic shock.
directed therapy in the treatment of severe sepsis and
Ann Pharmacother 38:464, 2004.
septic shock. N Engl J Med 345:1368, 2001.
Creteur J, Vincent JL: Hemoglobin solutions. Crit Care Med
Toh CH, Dennis M: Disseminated intravascular coagulation:
31(12 Suppl):S698, 2003.
old disease, new hope. BMJ 327:974, 2003.
Crowell JW, Guyton AC: Evidence favoring a cardiac mech-
Wernly JA: Ischemia, reperfusion, and the role of surgery in
anism in irreversible hemorrhagic shock. Am J Physiol
the treatment of cardiogenic shock secondary to acute
201:893, 1961.
myocardial infarction: an interpretative review. J Surg Res
Crowell JW, Smith EE: Oxygen deficit and irreversible
117:6, 2004.
hemorrhagic shock. Am J Physiol 206:313, 1964.
Wilson M, Davis DP, Coimbra R: Diagnosis and monitoring
de Jonge E, Levi M: Effects of different plasma substitutes
of hemorrhagic shock during the initial resuscitation of
on blood coagulation: a comparative review. Crit Care
multiple trauma patients: a review. J Emerg Med 24:413,
Med 29:1261, 2001.
2003.
U N I T V
The Body Fluids
and Kidneys
25. The Body Fluid Compartments: Extracellular and
Intracellular Fluids; Interstitial Fluid and Edema
26. Urine Formation by the Kidneys: I. Glomerular
Filtration, Renal Blood Flow, and Their Control
27. Urine Formation by the Kidneys: II. Tubular
Processing of the Glomerular Filtrate
28. Regulation of Extracellular Fluid Osmolarity
and Sodium Concentration
29. Renal Regulation of Potassium, Calcium,
Phosphate, and Magnesium; Integration of Renal
Mechanisms for Control of Blood Volume and
Extracellular Fluid Volume
30. Regulation of Acid-Base Balance
31. Kidney Diseases and Diuretics
C H A P T E R
2
5
The Body Fluid Compartments:
Extracellular and
Intracellular Fluids;
Interstitial Fluid and Edema
The maintenance of a relatively constant volume
and a stable composition of the body fluids is essen-
tial for homeostasis, as discussed in Chapter 1. Some
of the most common and important problems in
clinical medicine arise because of abnormalities in
the control systems that maintain this constancy of
the body fluids. In this chapter and in the following
chapters on the kidneys, we discuss the overall reg-
ulation of body fluid volume, constituents of the extracellular fluid, acid-base
balance, and control of fluid exchange between extracellular and intracellular
compartments.
Fluid Intake and Output Are Balanced During
Steady-State Conditions
The relative constancy of the body fluids is remarkable because there is con-
tinuous exchange of fluid and solutes with the external environment as well as
within the different compartments of the body. For example, there is a highly
variable fluid intake that must be carefully matched by equal output from the
body to prevent body fluid volumes from increasing or decreasing.
Daily Intake of Water
Water is added to the body by two major sources: (1) it is ingested in the form
of liquids or water in the food, which together normally add about 2100 ml/day
to the body fluids, and (2) it is synthesized in the body as a result of oxidation
of carbohydrates, adding about 200 ml/day. This provides a total water intake
of about 2300 ml/day (Table 25-1). Intake of water, however, is highly variable
among different people and even within the same person on different days,
depending on climate, habits, and level of physical activity.
Daily Loss of Body Water
Insensible Water Loss. Some of the water losses cannot be precisely regulated. For
example, there is a continuous loss of water by evaporation from the respira-
tory tract and diffusion through the skin, which together account for about
700 ml/day of water loss under normal conditions. This is termed insensible
water loss because we are not consciously aware of it, even though it occurs con-
tinually in all living humans.
The insensible water loss through the skin occurs independently of sweating
and is present even in people who are born without sweat glands; the average
water loss by diffusion through the skin is about 300 to 400 ml/day. This loss is
minimized by the cholesterol-filled cornified layer of the skin, which provides a
barrier against excessive loss by diffusion. When the cornified layer becomes
291
292
Unit V The Body Fluids and Kidneys
Table 25-1
OUTPUT
INTAKE
•Kidneys
Daily Intake and Output of Water (ml/day)
•Lungs
•Feces
•Sweat
Plasma
Prolonged,
•Skin
3.0 L
Normal
Heavy Exercise
Capillary membrane
Intake
Fluids ingested
2100
?
Interstitial
From metabolism
200
200
fluid
Total intake
2300
?
11.0 L
Output
Insensible—skin
350
350
Insensible—lungs
350
650
Cell membrane
Sweat
100
5000
Feces
100
100
Urine
1400
500
Total output
2300
6600
Intracellular
fluid
denuded, as occurs with extensive burns, the rate of
28.0 L
evaporation can increase as much as 10-fold, to 3 to
5 L/day. For this reason, burn victims must be given
large amounts of fluid, usually intravenously, to
balance fluid loss.
Insensible water loss through the respiratory tract
averages about 300 to 400 ml/day. As air enters the
Figure 25-1
respiratory tract, it becomes saturated with moisture,
to a vapor pressure of about 47 mm Hg, before it is
Summary of body fluid regulation, including the major body fluid
expelled. Because the vapor pressure of the inspired
compartments and the membranes that separate these compart-
ments. The values shown are for an average 70-kilogram person.
air is usually less than 47 mm Hg, water is continuously
lost through the lungs with respiration. In cold
weather, the atmospheric vapor pressure decreases to
nearly 0, causing an even greater loss of water from
20 L/day in a person who has been drinking tremen-
the lungs as the temperature decreases. This explains
dous amounts of water.
the dry feeling in the respiratory passages in cold
This variability of intake is also true for most of the
weather.
electrolytes of the body, such as sodium, chloride, and
potassium. In some people, sodium intake may be as
Fluid Loss in Sweat. The amount of water lost by sweat-
low as 20 mEq/day, whereas in others, sodium intake
ing is highly variable, depending on physical activity
may be as high as 300 to 500 mEq/day. The kidneys are
and environmental temperature. The volume of sweat
faced with the task of adjusting the excretion rate of
normally is about 100 ml/day, but in very hot weather
water and electrolytes to match precisely the intake of
or during heavy exercise, water loss in sweat occa-
these substances, as well as compensating for excessive
sionally increases to 1 to 2 L/hour. This would rapidly
losses of fluids and electrolytes that occur in certain
deplete the body fluids if intake were not also
disease states. In Chapters 26 through 30, we discuss
increased by activating the thirst mechanism discussed
the mechanisms that allow the kidneys to perform
in Chapter 29.
these remarkable tasks.
Water Loss in Feces. Only a small amount of water
(100 ml/day) normally is lost in the feces. This can
Body Fluid Compartments
increase to several liters a day in people with severe
diarrhea. For this reason, severe diarrhea can be life
The total body fluid is distributed mainly between two
threatening if not corrected within a few days.
compartments: the extracellular fluid and the intracel-
lular fluid
(Figure 25-1). The extracellular fluid is
Water Loss by the Kidneys. The remaining water loss from
divided into the interstitial fluid and the blood plasma.
the body occurs in the urine excreted by the kidneys.
There is another small compartment of fluid that is
There are multiple mechanisms that control the rate
referred to as transcellular fluid. This compartment
of urine excretion. In fact, the most important means
includes fluid in the synovial, peritoneal, pericardial,
by which the body maintains a balance between water
and intraocular spaces, as well as the cerebrospinal
intake and output, as well as a balance between intake
fluid; it is usually considered to be a specialized type
and output of most electrolytes in the body, is by con-
of extracellular fluid, although in some cases, its com-
trolling the rates at which the kidneys excrete these
position may differ markedly from that of the plasma
substances. For example, urine volume can be as low
or interstitial fluid. All the transcellular fluids together
as 0.5 L/day in a dehydrated person or as high as
constitute about 1 to 2 liters.
Chapter 25
The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema
293
In the average 70-kilogram adult human, the total
chamber of its own, the circulatory system. The blood
body water is about 60 per cent of the body weight, or
volume is especially important in the control of car-
about 42 liters. This percentage can change, depending
diovascular dynamics.
on age, gender, and degree of obesity. As a person
The average blood volume of adults is about 7 per
grows older, the percentage of total body weight that
cent of body weight, or about 5 liters. About 60 per
is fluid gradually decreases. This is due in part to the
cent of the blood is plasma and 40 per cent is red blood
fact that aging is usually associated with an increased
cells, but these percentages can vary considerably in
percentage of the body weight being fat, which
different people, depending on gender, weight, and
decreases the percentage of water in the body. Because
other factors.
women normally have more body fat than men, they
contain slightly less water than men in proportion to
Hematocrit (Packed Red Cell Volume). The hematocrit is
their body weight. Therefore, when discussing the
the fraction of the blood composed of red blood cells,
“average” body fluid compartments, we should realize
as determined by centrifuging blood in a “hematocrit
that variations exist, depending on age, gender, and
tube” until the cells become tightly packed in the
percentage of body fat.
bottom of the tube. It is impossible to completely pack
the red cells together; therefore, about 3 to 4 per cent
of the plasma remains entrapped among the cells, and
Intracellular Fluid Compartment
the true hematocrit is only about 96 per cent of the
measured hematocrit.
About 28 of the 42 liters of fluid in the body are inside
In men, the measured hematocrit is normally about
the 75 trillion cells and are collectively called the intra-
0.40, and in women, it is about 0.36. In severe anemia,
cellular fluid. Thus, the intracellular fluid constitutes
the hematocrit may fall as low as 0.10, a value that is
about 40 per cent of the total body weight in an
barely sufficient to sustain life. Conversely, there are
“average” person.
some conditions in which there is excessive production
The fluid of each cell contains its individual mixture
of red blood cells, resulting in polycythemia. In these
of different constituents, but the concentrations of
conditions, the hematocrit can rise to 0.65.
these substances are similar from one cell to another.
In fact, the composition of cell fluids is remarkably
similar even in different animals, ranging from the
Constituents of Extracellular
most primitive microorganisms to humans. For this
reason, the intracellular fluid of all the different
and Intracellular Fluids
cells together is considered to be one large fluid
compartment.
Comparisons of the composition of the extracellular
fluid, including the plasma and interstitial fluid, and the
intracellular fluid are shown in Figures 25-2 and 25-3
Extracellular Fluid
and in Table 25-2.
Compartment
All the fluids outside the cells are collectively called
Ionic Composition of Plasma and
the extracellular fluid. Together these fluids account for
Interstitial Fluid Is Similar
about 20 per cent of the body weight, or about 14 liters
in a normal 70-kilogram adult. The two largest com-
Because the plasma and interstitial fluid are separated
partments of the extracellular fluid are the interstitial
only by highly permeable capillary membranes, their
fluid, which makes up more than three fourths of the
ionic composition is similar. The most important dif-
extracellular fluid, and the plasma, which makes up
ference between these two compartments is the higher
almost one fourth of the extracellular fluid, or about 3
concentration of protein in the plasma; because the
liters. The plasma is the noncellular part of the blood;
capillaries have a low permeability to the plasma pro-
it exchanges substances continuously with the intersti-
teins, only small amounts of proteins are leaked into
tial fluid through the pores of the capillary mem-
the interstitial spaces in most tissues.
branes. These pores are highly permeable to almost all
Because of the Donnan effect, the concentration of
solutes in the extracellular fluid except the proteins.
positively charged ions (cations) is slightly greater
Therefore, the extracellular fluids are constantly
(about 2 per cent) in the plasma than in the intersti-
mixing, so that the plasma and interstitial fluids have
tial fluid. The plasma proteins have a net negative
about the same composition except for proteins, which
charge and, therefore, tend to bind cations, such as
have a higher concentration in the plasma.
sodium and potassium ions, thus holding extra
amounts of these cations in the plasma along with the
Blood Volume
plasma proteins. Conversely, negatively charged ions
(anions) tend to have a slightly higher concentration
Blood contains both extracellular fluid (the fluid in
in the interstitial fluid compared with the plasma,
plasma) and intracellular fluid (the fluid in the red
because the negative charges of the plasma proteins
blood cells). However, blood is considered to be a sep-
repel the negatively charged anions. For practical
arate fluid compartment because it is contained in a
purposes, however, the concentration of ions in the
294
Unit V The Body Fluids and Kidneys
Cations
Anions
150
Phospholipids - 280 mg/dl
100
50
Na
+
Ca++
Cl-
Cholesterol - 150 mg/dl
0
K+
Mg++
50
Neutral fat - 125 mg/dl
Glucose - 100 mg/dl
100
Urea - 15 mg/dl
Lactic acid - 10 mg/dl
150
Uric acid - 3 mg/dl
Creatinine - 1.5 mg/dl
Bilirubin - 0.5 mg/dl
Figure 25-2
Bile salts - trace
Figure 25-3
Major cations and anions of the intracellular and extracellular
fluids. The concentrations of Ca++ and Mg++ represent the sum of
Nonelectrolytes of the plasma.
these two ions. The concentrations shown represent the total of
free ions and complexed ions.
Table 25-2
Osmolar Substances in Extracellular and Intracellular Fluids
Plasma (mOsm/L H2O)
Interstitial (mOsm/L H2O)
Intracellular (mOsm/L H2O)
Na+
142
139
14
K+
4.2
4.0
140
Ca++
1.3
1.2
0
Mg+
0.8
0.7
20
Cl-
108
108
4
HCO3-
24
28.3
10
HPO4-, H2PO4-
2
2
11
SO4-
0.5
0.5
1
Phosphocreatine
45
Carnosine
14
Amino acids
2
2
8
Creatine
0.2
0.2
9
Lactate
1.2
1.2
1.5
Adenosine triphosphate
5
Hexose monophosphate
3.7
Glucose
5.6
5.6
Protein
1.2
0.2
4
Urea
4
4
4
Others
4.8
3.9
10
Total mOsm/L
301.8
300.8
301.2
Corrected osmolar activity (mOsm/L)
282.0
281.0
281.0
Total osmotic pressure at 37∞C (mm Hg)
5443
5423
5423
Chapter 25
The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema
295
interstitial fluid and in the plasma is considered to be
about equal.
Referring again to Figure 25-2, one can see that
the extracellular fluid, including the plasma and the
Indicator Mass A = Volume A x Concentration A
interstitial fluid, contains large amounts of sodium
and chloride ions, reasonably large amounts of bicar-
bonate ions, but only small quantities of potassium,
Indicator Mass A = Indicator Mass B
calcium, magnesium, phosphate, and organic acid ions.
The composition of extracellular fluid is carefully
regulated by various mechanisms, but especially by the
kidneys, as discussed later. This allows the cells to
remain continually bathed in a fluid that contains the
proper concentration of electrolytes and nutrients for
optimal cell function.
Important Constituents of the
Intracellular Fluid
Indicator Mass B = Volume B x Concentration B
Volume B = Indicator Mass B / Concentration B
The intracellular fluid is separated from the extracel-
Figure 25-4
lular fluid by a cell membrane that is highly permeable
to water but not to most of the electrolytes in the body.
Indicator-dilution method for measuring fluid volumes.
In contrast to the extracellular fluid, the intracellu-
lar fluid contains only small quantities of sodium and
chloride ions and almost no calcium ions. Instead, it
tion, one can calculate the unknown volume
of
contains large amounts of potassium and phosphate
chamber B as
ions plus moderate quantities of magnesium and
sulfate ions, all of which have low concentrations in the
Volume A ¥Concentration A
extracellular fluid. Also, cells contain large amounts of
Volume B=
Concentration B
protein, almost four times as much as in the plasma.
Note that all one needs to know for this calculation
is
(1) the total amount of substance injected into
Measurement of Fluid
the chamber (the numerator of the equation) and (2)
Volumes in the Different Body
the concentration of the fluid in the chamber after the
substance has been dispersed
(the denominator).
Fluid Compartments—The
For example, if 1 milliliter of a solution containing
Indicator-Dilution Principle
10 mg/ml of dye is dispersed into chamber B and
the final concentration in the chamber is 0.01 mil-
The volume of a fluid compartment in the body can be
ligram for each milliliter of fluid, the unknown volume
measured by placing an indicator substance in the
of the chamber can be calculated as follows:
compartment, allowing it to disperse evenly through-
out the compartment’s fluid, and then analyzing the
1
ml
¥
10 mg ml
Volume B=
=
1000
ml
extent to which the substance becomes diluted. Figure
0.01 mg ml
25-4 shows this “indicator-dilution” method of meas-
uring the volume of a fluid compartment, which is
This method can be used to measure the volume of
based on the principle of conservation of mass. This
virtually any compartment in the body as long as (1)
means that the total mass of a substance after disper-
the indicator disperses evenly throughout the com-
sion in the fluid compartment will be the same as the
partment, (2) the indicator disperses only in the com-
total mass injected into the compartment.
partment that is being measured, and (3) the indicator
In the example shown in Figure
25-4, a small
is not metabolized or excreted. Several substances can
amount of dye or other substance contained in the
be used to measure the volume of each of the differ-
syringe is injected into a chamber, and the substance
ent body fluids.
is allowed to disperse throughout the chamber until it
becomes mixed in equal concentrations in all areas.
Determination of Volumes
Then a sample of fluid containing the dispersed sub-
of Specific Body Fluid
stance is removed and the concentration is analyzed
chemically, photoelectrically, or by other means. If
Compartments
none of the substance leaks out of the compartment,
Measurement of Total Body Water. Radioactive water
the total mass of substance in the compartment
(tritium,3H2O) or heavy water (deuterium,2H2O) can
(Volume B ¥ Concentration B) will equal the total
be used to measure total body water. These forms of
mass of the substance injected (Volume A ¥ Con-
water mix with the total body water within a few hours
centration A). By simple rearrangement of the equa-
after being injected into the blood, and the dilution
296
Unit V The Body Fluids and Kidneys
volume can also be calculated if one knows the
Table 25-3
hematocrit (the fraction of the total blood volume com-
Measurement of Body Fluid Volumes
posed of cells), using the following equation:
Plasma volume
Volume
Indicators
Total blood volume
=
1-Hematocrit
Total body water
3H
2O,2H2O, antipyrine
For example, if plasma volume is 3 liters and hemat-
Extracellular fluid
22Na,125I-iothalamate, thiosulfate, inulin
ocrit is 0.40, total blood volume would be calculated as
Intracellular fluid
(Calculated as Total body water -
Extracellular fluid volume)
3 liters
=
5
liters
Plasma volume
125I-albumin, Evans blue dye (T-1824)
1-
04
Blood volume
51Cr-labeled red blood cells, or calculated
Another way to measure blood volume is to inject
as Blood volume = Plasma volume/
into the circulation red blood cells that have been
(1 - Hematocrit)
labeled with radioactive material. After these mix in the
Interstitial fluid
(Calculated as Extracellular fluid
circulation, the radioactivity of a mixed blood sample
volume - Plasma volume)
can be measured, and the total blood volume can be cal-
culated using the dilution principle. A substance fre-
quently used to label the red blood cells is radioactive
From Guyton AC, Hall JE: Human Physiology and Mechanisms of Disease,
6th ed. Philadelphia: WB Saunders, 1997.
chromium (51Cr), which binds tightly with the red blood
cells.
principle can be used to calculate total body water
(Table 25-3). Another substance that has been used to
Regulation of Fluid Exchange
measure total body water is antipyrine, which is very
lipid soluble and can rapidly penetrate cell membranes
and Osmotic Equilibrium
and distribute itself uniformly throughout the intracel-
Between Intracellular and
lular and extracellular compartments.
Extracellular Fluid
Measurement of Extracellular Fluid Volume. The volume of
extracellular fluid can be estimated using any of several
A frequent problem in treating seriously ill patients is
substances that disperse in the plasma and interstitial
maintaining adequate fluids in one or both of the intra-
fluid but do not readily permeate the cell membrane.
cellular and extracellular compartments. As discussed
They include radioactive sodium, radioactive chloride,
in Chapter 16 and later in this chapter, the relative
radioactive iothalamate, thiosulfate ion, and inulin.
amounts of extracellular fluid distributed between the
When any one of these substances is injected into the
plasma and interstitial spaces are determined mainly
blood, it usually disperses almost completely through-
out the extracellular fluid within 30 to 60 minutes. Some
by the balance of hydrostatic and colloid osmotic
of these substances, however, such as radioactive
forces across the capillary membranes.
sodium, may diffuse into the cells in small amounts.
The distribution of fluid between intracellular and
Therefore, one frequently speaks of the sodium space or
extracellular compartments, in contrast, is determined
the inulin space, instead of calling the measurement the
mainly by the osmotic effect of the smaller solutes—
true extracellular fluid volume.
especially sodium, chloride, and other electrolytes—
acting across the cell membrane. The reason for this is
Calculation of Intracellular Volume. The intracellular
that the cell membranes are highly permeable to water
volume cannot be measured directly. However, it can be
calculated as
but relatively impermeable to even small ions such as
sodium and chloride. Therefore, water moves across
Intracellular volume = Total body water
the cell membrane rapidly, so that the intracellular
- Extracellular volume
fluid remains isotonic with the extracellular fluid.
In the next section, we discuss the interrelations
Measurement of Plasma Volume. To measure plasma
volume, a substance must be used that does not readily
between intracellular and extracellular fluid volumes
penetrate capillary membranes but remains in the vas-
and the osmotic factors that can cause shifts of fluid
cular system after injection. One of the most commonly
between these two compartments.
used substances for measuring plasma volume is serum
albumin labeled with radioactive iodine (125I-albumin).
Also, dyes that avidly bind to the plasma proteins, such
Basic Principles of Osmosis
as Evans blue dye (also called T-1824), can be used to
and Osmotic Pressure
measure plasma volume.
The basic principles of osmosis and osmotic pressure
Calculation of Interstitial Fluid Volume. Interstitial fluid
were presented in Chapter 4. Therefore, we review
volume cannot be measured directly, but it can be cal-
culated as
here only the most important aspects of these princi-
ples as they apply to volume regulation.
Interstitial fluid volume = Extracellular fluid volume
Osmosis is the net diffusion of water across a selec-
- Plasma volume
tively permeable membrane from a region of high water
Measurement of Blood Volume. If one measures plasma
concentration to one that has a lower water concentra-
volume using the methods described earlier, blood
tion. When a solute is added to pure water, this reduces
Chapter 25
The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema
297
the concentration of water in the mixture. Thus, the
the osmosis. The precise amount of pressure required
higher the solute concentration in a solution, the lower
to prevent the osmosis is called the osmotic pressure.
the water concentration. Further, water diffuses from
Osmotic pressure, therefore, is an indirect measure-
a region of low solute concentration (high water con-
ment of the water and solute concentrations of a solu-
centration) to one with a high solute concentration
tion. The higher the osmotic pressure of a solution,
(low water concentration).
the lower the water concentration and the higher the
Because cell membranes are relatively impermeable
solute concentration of the solution.
to most solutes but highly permeable to water (i.e.,
selectively permeable), whenever there is a higher
Relation Between Osmotic Pressure and Osmolarity. The
concentration of solute on one side of the cell mem-
osmotic pressure of a solution is directly proportional
brane, water diffuses across the membrane toward the
to the concentration of osmotically active particles in
region of higher solute concentration. Thus, if a solute
that solution. This is true regardless of whether the
such as sodium chloride is added to the extracellular
solute is a large molecule or a small molecule. For
fluid, water rapidly diffuses from the cells through the
example, one molecule of albumin with a molecular
cell membranes into the extracellular fluid until the
weight of 70,000 has the same osmotic effect as one
water concentration on both sides of the membrane
molecule of glucose with a molecular weight of 180.
becomes equal. Conversely, if a solute such as sodium
One molecule of sodium chloride, however, has two
chloride is removed from the extracellular fluid, water
osmotically active particles, Na+ and Cl-, and therefore
diffuses from the extracellular fluid through the cell
has twice the osmotic effect of either an albumin mol-
membranes and into the cells. The rate of diffusion of
ecule or a glucose molecule. Thus, the osmotic pressure
water is called the rate of osmosis.
of a solution is proportional to its osmolarity, a
measure of the concentration of solute particles.
Relation Between Moles and Osmoles. Because the water
Expressed mathematically, according to van’t Hoff ’s
concentration of a solution depends on the number of
law, osmotic pressure (p) can be calculated as
solute particles in the solution, a concentration term is
p = CRT
needed to describe the total concentration of solute
particles, regardless of their exact composition. The
where C is the concentration of solutes in osmoles per
total number of particles in a solution is measured in
liter, R is the ideal gas constant, and T is the absolute
osmoles. One osmole (osm) is equal to 1 mole (mol)
temperature in degrees kelvin (273° + centigrade°). If
(6.02
¥ 1023) of solute particles. Therefore, a solution
p is expressed in millimeters of mercury (mm Hg), the
containing 1 mole of glucose in each liter has a con-
unit of pressure commonly used for biological fluids,
centration of 1 osm/L. If a molecule dissociates into
and T is normal body temperature (273° + 37° =
two ions (giving two particles), such as sodium chlo-
310° kelvin), the value of p calculates to be about
ride ionizing to give chloride and sodium ions, then a
19,300 mm Hg for a solution having a concentration
solution containing 1 mol/L will have an osmolar con-
of 1 osm/L. This means that for a concentration of
centration of 2 osm/L. Likewise, a solution that con-
1 mOsm/L, p is equal to 19.3 mm Hg. Thus, for each
tains 1 mole of a molecule that dissociates into three
milliosmole concentration gradient across the cell
ions, such as sodium sulfate (Na2SO4), will contain
membrane, 19.3 mm Hg osmotic pressure is exerted.
3 osm/L. Thus, the term osmole refers to the number
of osmotically active particles in a solution rather than
Calculation of the Osmolarity and Osmotic Pressure of a
to the molar concentration.
Solution. Using van’t Hoff ’s law, one can calculate the
In general, the osmole is too large a unit for express-
potential osmotic pressure of a solution, assuming that
ing osmotic activity of solutes in the body fluids.
the cell membrane is impermeable to the solute.
The term milliosmole (mOsm), which equals 1/1000
For example, the osmotic pressure of a 0.9 per cent
osmole, is commonly used.
sodium chloride solution is calculated as follows: A
0.9 per cent solution means that there is 0.9 gram of
Osmolality and Osmolarity. The osmolal concentration of
sodium chloride per 100 milliliters of solution, or
a solution is called osmolality when the concentration
9 g/L. Because the molecular weight of sodium chlo-
is expressed as osmoles per kilogram of water; it is
ride is 58.5 g/mol, the molarity of the solution is 9 g/L
called osmolarity when it is expressed as osmoles per
divided by 58.5 g/mol, or about 0.154 mol/L. Because
liter of solution. In dilute solutions such as the body
each molecule of sodium chloride is equal to
fluids, these two terms can be used almost synony-
2 osmoles, the osmolarity of the solution is 0.154
¥ 2,
mously because the differences are small. In most
or 0.308 osm/L. Therefore, the osmolarity of this solu-
cases, it is easier to express body fluid quantities in
tion is 308 mOsm/L. The potential osmotic pressure
liters of fluid rather than in kilograms of water. There-
of this solution would therefore be
308 mOsm/L
fore, most of the calculations used clinically and the
¥ 19.3 mm Hg/mOsm/L, or 5944 mm Hg.
calculations expressed in the next several chapters are
This calculation is only an approximation, because
based on osmolarities rather than osmolalities.
sodium and chloride ions do not behave entirely inde-
pendently in solution because of interionic attraction
Osmotic Pressure. Osmosis of water molecules across a
between them. One can correct for these deviations
selectively permeable membrane can be opposed by
from the predictions of van’t Hoff ’s law by using a
applying a pressure in the direction opposite that of
correction factor called the osmotic coefficient. For
298
Unit V The Body Fluids and Kidneys
sodium chloride, the osmotic coefficient is about 0.93.
Therefore, the actual osmolarity of a 0.9 per cent
sodium chloride solution is
308
¥ 0.93, or about
286 mOsm/L. For practical reasons, the osmotic coef-
A
ficients of different solutes are sometimes neglected in
determining the osmolarity and osmotic pressures of
physiologic solutions.
280 mOsm/L
Osmolarity of the Body Fluids. Turning back to Table 25-2,
B
ISOTONIC
note the approximate osmolarity of the various osmot-
C
No change
ically active substances in plasma, interstitial fluid, and
intracellular fluid. Note that about 80 per cent of the
total osmolarity of the interstitial fluid and plasma is
due to sodium and chloride ions, whereas for intracel-
lular fluid, almost half the osmolarity is due to potas-
sium ions, and the remainder is divided among many
other intracellular substances.
As shown in Table 25-2, the total osmolarity of each
200 mOsm/L
360 mOsm/L
of the three compartments is about 300 mOsm/L, with
the plasma being about 1 mOsm/L greater than that
HYPOTONIC
HYPERTONIC
of the interstitial and intracellular fluids. The slight dif-
Cell swells
Cell shrinks
ference between plasma and interstitial fluid is caused
Figure 25-5
by the osmotic effects of the plasma proteins, which
maintain about 20 mm Hg greater pressure in the cap-
Effects of isotonic (A), hypertonic (B), and hypotonic (C) solutions
illaries than in the surrounding interstitial spaces, as
on cell volume.
discussed in Chapter 16.
Corrected Osmolar Activity of the Body Fluids. At the
bottom of Table 25-2 are shown corrected osmolar
potential osmotic pressure that can develop across the
activities of plasma, interstitial fluid, and intracellular
cell membrane is more than 5400 mm Hg. This demon-
fluid. The reason for these corrections is that molecules
strates the large force that can move water across the
and ions in solution exert interionic and intermolecu-
cell membrane when the intracellular and extracellu-
lar attraction or repulsion from one solute molecule to
lar fluids are not in osmotic equilibrium. As a result of
the next, and these two effects can cause, respectively,
these forces, relatively small changes in the concen-
a slight decrease or an increase in the osmotic “activ-
tration of impermeant solutes in the extracellular fluid
ity” of the dissolved substance.
can cause large changes in cell volume.
Total Osmotic Pressure Exerted by the Body Fluids. Table
Isotonic, Hypotonic, and Hypertonic Fluids. The effects of
25-2 also shows the total osmotic pressure in millime-
different concentrations of impermeant solutes in the
ters of mercury that would be exerted by each of the
extracellular fluid on cell volume are shown in Figure
different fluids if it were placed on one side of the cell
25-5. If a cell is placed in a solution of impermeant
membrane with pure water on the other side. Note
solutes having an osmolarity of 282 mOsm/L, the cells
that this total pressure averages about 5443 mm Hg for
will not shrink or swell because the water concentra-
plasma, which is 19.3 times the corrected osmolarity of
tion in the intracellular and extracellular fluids is equal
282 mOsm/L for plasma.
and the solutes cannot enter or leave the cell. Such a
solution is said to be isotonic because it neither shrinks
nor swells the cells. Examples of isotonic solutions
Osmotic Equilibrium Is
include a 0.9 per cent solution of sodium chloride or a
5 per cent glucose solution. These solutions are impor-
Maintained Between
tant in clinical medicine because they can be infused
Intracellular and
into the blood without the danger of upsetting osmotic
Extracellular Fluids
equilibrium between the intracellular and extracellu-
lar fluids.
Large osmotic pressures can develop across the cell
If a cell is placed into a hypotonic solution that has
membrane with relatively small changes in the con-
a lower concentration of impermeant solutes (less than
centrations of solutes in the extracellular fluid. As dis-
282 mOsm/L), water will diffuse into the cell, causing
cussed earlier, for each milliosmole concentration
it to swell; water will continue to diffuse into the cell,
gradient of an impermeant solute (one that will not
diluting the intracellular fluid while also concentrating
permeate the cell membrane), about
19.3 mm Hg
the extracellular fluid until both solutions have about
osmotic pressure is exerted across the cell membrane.
the same osmolarity. Solutions of sodium chloride with
If the cell membrane is exposed to pure water and the
a concentration of less than 0.9 per cent are hypotonic
osmolarity of intracellular fluid is 282 mOsm/L, the
and cause cells to swell.
Chapter 25
The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema
299
If a cell is placed in a hypertonic solution having a
1. Water moves rapidly across cell membranes;
higher concentration of impermeant solutes, water will
therefore, the osmolarities of intracellular and
flow out of the cell into the extracellular fluid, con-
extracellular fluids remain almost exactly equal
centrating the intracellular fluid and diluting the extra-
to each other except for a few minutes after a
cellular fluid. In this case, the cell will shrink until the
change in one of the compartments.
two concentrations become equal. Sodium chloride
2. Cell membranes are almost completely
solutions of greater than 0.9 per cent are hypertonic.
impermeable to many solutes; therefore, the
number of osmoles in the extracellular or
Isosmotic, Hyperosmotic, and Hypo-osmotic Fluids. The
intracellular fluid generally remains constant
terms isotonic, hypotonic, and hypertonic refer to
unless solutes are added to or lost from the
whether solutions will cause a change in cell volume.
extracellular compartment.
The tonicity of solutions depends on the concentration
With these basic principles in mind, we can analyze
of impermeant solutes. Some solutes, however, can
the effects of different abnormal fluid conditions on
permeate the cell membrane. Solutions with an osmo-
extracellular and intracellular fluid volumes and
larity the same as the cell are called isosmotic, regard-
osmolarities.
less of whether the solute can penetrate the cell
membrane.
The terms hyperosmotic and hypo-osmotic refer to
Effect of Adding Saline Solution
solutions that have a higher or lower osmolarity,
to the Extracellular Fluid
respectively, compared with the normal extracellular
fluid, without regard for whether the solute permeates
If an isotonic saline solution is added to the extracel-
the cell membrane. Highly permeating substances,
lular fluid compartment, the osmolarity of the extra-
such as urea, can cause transient shifts in fluid volume
cellular fluid does not change; therefore, no osmosis
between the intracellular and extracellular fluids, but
occurs through the cell membranes. The only effect is
given enough time, the concentrations of these sub-
an increase in extracellular fluid volume
(Figure
stances eventually become equal in the two compart-
25-6A). The sodium and chloride largely remain in the
ments and have little effect on intracellular volume
extracellular fluid because the cell membrane behaves
under steady-state conditions.
as though it were virtually impermeable to the sodium
chloride.
Osmotic Equilibrium Between Intracellular and Extracellular
If a hypertonic solution is added to the extracellular
Fluids Is Rapidly Attained. The transfer of fluid across the
fluid, the extracellular osmolarity increases and causes
cell membrane occurs so rapidly that any differences
osmosis of water out of the cells into the extracellular
in osmolarities between these two compartments are
compartment (see Figure 25-6B). Again, almost all the
usually corrected within seconds or, at the most,
added sodium chloride remains in the extracellular
minutes. This rapid movement of water across the cell
compartment, and fluid diffuses from the cells into the
membrane does not mean that complete equilibrium
extracellular space to achieve osmotic equilibrium.
occurs between the intracellular and extracellular
The net effect is an increase in extracellular volume
compartments throughout the whole body within the
(greater than the volume of fluid added), a decrease in
same short period. The reason for this is that fluid
intracellular volume, and a rise in osmolarity in both
usually enters the body through the gut and must be
compartments.
transported by the blood to all tissues before complete
If a hypotonic solution is added to the extracellular
osmotic equilibrium can occur. It usually takes about
fluid, the osmolarity of the extracellular fluid decreases
30 minutes to achieve osmotic equilibrium everywhere
and some of the extracellular water diffuses into the
in the body after drinking water.
cells until the intracellular and extracellular compart-
ments have the same osmolarity (see Figure 25-6C).
Both the intracellular and the extracellular volumes
are increased by the addition of hypotonic fluid,
Volume and Osmolality of
although the intracellular volume increases to a
Extracellular and Intracellular
greater extent.
Fluids in Abnormal States
Calculation of Fluid Shifts and Osmolarities After Infusion
Some of the different factors that can cause extracel-
of Hypertonic Saline. We can calculate the sequential
lular and intracellular volumes to change markedly are
effects of infusing different solutions on extracellular
ingestion of water, dehydration, intravenous infusion
and intracellular fluid volumes and osmolarities. For
of different types of solutions, loss of large amounts of
example, if 2 liters of a hypertonic 3.0 per cent sodium
fluid from the gastrointestinal tract, and loss of abnor-
chloride solution are infused into the extracellular
mal amounts of fluid by sweating or through the
fluid compartment of a 70-kilogram patient whose
kidneys.
initial plasma osmolarity is 280 mOsm/L, what would
One can calculate both the changes in intracellular
be the intracellular and extracellular fluid volumes and
and extracellular fluid volumes and the types of
osmolarities after osmotic equilibrium?
therapy that should be instituted if the following basic
The first step is to calculate the initial conditions,
principles are kept in mind:
including the volume, concentration, and total
300
Unit V The Body Fluids and Kidneys
Intracellular fluid
Extracellular fluid
Normal State
A. Add Isotonic NaCl
300
200
100
0
10
20
30
40
Volume (liters)
Figure 25-6
C. Add Hypotonic NaCl
B. Add Hypertonic NaCl
Effect of adding isotonic, hyper-
tonic, and hypotonic solutions
to the extracellular fluid after
osmotic equilibrium. The normal
state is indicated by the solid
lines, and the shifts from normal
are shown by the shaded areas.
The volumes of intracellular and
extracellular fluid compartments
are shown in the abscissa of each
diagram, and the osmolarities of
these compartments are shown
on the ordinates.
milliosmoles in each compartment. Assuming that
would be an additional 2051 milliosmoles of total
extracellular fluid volume is 20 per cent of body weight
solute, yielding a total of 5791 milliosmoles. Because
and intracellular fluid volume is 40 per cent of body
the extracellular compartment now has 16 liters of
weight, the following volumes and concentrations
volume, the concentration can be calculated by divid-
can be calculated.
ing
5791 milliosmoles by 16 liters to yield a con-
centration of 373 mOsm/L. Thus, the following values
Step 1. Initial Conditions
would occur instantly after adding the solution.
Volume
Concentration
Total
Step 2. Instantaneous Effect of Adding 2 Liters of
(Liters)
(mOsm/L)
(mOsm)
3.0 Per Cent Sodium Chloride
Extracellular fluid
14
280
3,920
Intracellular fluid
28
280
7,840
Volume
Concentration
Total
Total body fluid
42
280
11,760
(Liters)
(mOsm/L)
(mOsm)
Extracellular fluid
16
373
5,971
Intracellular fluid
28
280
7,840
Next, we calculate the total milliosmoles added to
Total body fluid
44
No equilibrium
13,811
the extracellular fluid in 2 liters of 3.0 per cent sodium
chloride. A 3.0 per cent solution means that there are
3.0 g/100 ml, or 30 grams of sodium chloride per liter.
In the third step, we calculate the volumes and con-
Because the molecular weight of sodium chloride is
centrations that would occur within a few minutes
about 58.5 g/mol, this means that there is about 0.513
after osmotic equilibrium develops. In this case, the
mole of sodium chloride per liter of solution. For 2
concentrations in the intracellular and extracellular
liters of solution, this would be 1.026 mole of sodium
fluid compartments would be equal and can be calcu-
chloride. Because 1 mole of sodium chloride is about
lated by dividing the total milliosmoles in the body,
equal to 2 osmoles (sodium chloride has two osmoti-
13,811, by the total volume, which is now 44 liters. This
cally active particles per mole), the net effect of adding
yields a concentration of 313.9 mOsm/L. Therefore, all
2 liters of this solution is to add 2051 milliosmoles of
the body fluid compartments will have this same con-
sodium chloride to the extracellular fluid.
centration after osmotic equilibrium. Assuming that
In Step 2, we calculate the instantaneous effect of
no solute or water has been lost from the body and
adding 2051 milliosmoles of sodium chloride to the
that there is no movement of sodium chloride into or
extracellular fluid along with 2 liters of volume. There
out of the cells, we then calculate the volumes of
would be no change in the intracellular fluid concen-
the intracellular and extracellular compartments. The
tration or volume, and there would be no osmotic
intracellular fluid volume is calculated by dividing the
equilibrium. In the extracellular fluid, however, there
total milliosmoles in the intracellular fluid (7840) by
Chapter 25
The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema
301
the concentration (313.9 mOsm/L), to yield a volume
After the glucose or other nutrients are metabolized, an
of 24.98 liters. Extracellular fluid volume is calculated
excess of water often remains, especially if additional
by dividing the total milliosmoles in extracellular fluid
fluid is ingested. Ordinarily, the kidneys excrete this in
the form of a very dilute urine. The net result, therefore,
(5971) by the concentration (313.9 mOsm/L), to yield
is the addition of only nutrients to the body.
a volume of 19.02 liters. Again, these calculations are
based on the assumption that the sodium chloride
added to the extracellular fluid remains there and does
not move into the cells.
Clinical Abnormalities of
Fluid Volume Regulation:
Step 3. Effect of Adding 2 Liters of 3.0 Per Cent Sodium
Hyponatremia and
Chloride After Osmotic Equilibrium
Hypernatremia
The primary measurement that is readily available to
Volume
Concentration
Total
the clinician for evaluating a patient’s fluid status is the
(Liters)
(mOsm/L)
(mOsm)
plasma sodium concentration. Plasma osmolarity is not
Extracellular fluid
19.02
313.9
5,971
routinely measured, but because sodium and its associ-
Intracellular fluid
24.98
313.9
7,840
ated anions (mainly chloride) account for more than
Total body fluid
44.0
313.9
13,811
90 per cent of the solute in the extracellular fluid,
plasma sodium concentration is a reasonable indicator
of plasma osmolarity under many conditions. When
Thus, one can see from this example that adding 2
plasma sodium concentration is reduced more than a
liters of a hypertonic sodium chloride solution causes
few milliequivalents below normal (about 142 mEq/L),
more than a 5-liter increase in extracellular fluid
a person is said to have hyponatremia. When plasma
volume while decreasing intracellular fluid volume by
sodium concentration is elevated above normal, a
almost 3 liters.
person is said to have hypernatremia.
This method of calculating changes in intracellular
and extracellular fluid volumes and osmolarities can
be applied to virtually any clinical problem of fluid
Causes of Hyponatremia: Excess
volume regulation. The reader should be familiar with
Water or Loss of Sodium
such calculations because an understanding of the
mathematical aspects of osmotic equilibrium between
Decreased plasma sodium concentration can result
intracellular and extracellular fluid compartments is
from loss of sodium chloride from the extracellular fluid
essential for understanding almost all fluid abnormal-
or addition of excess water to the extracellular fluid
ities of the body and their treatment.
(Table 25-4). A primary loss of sodium chloride usually
results in hypo-osmotic dehydration and is associated
with decreased extracellular fluid volume. Conditions
that can cause hyponatremia owing to loss of sodium
Glucose and Other
chloride include diarrhea and vomiting. Overuse of
Solutions Administered
diuretics that inhibit the ability of the kidneys to con-
for Nutritive Purposes
serve sodium and certain types of sodium-wasting
kidney diseases can also cause modest degrees of
Many types of solutions are administered intravenously
hyponatremia. Finally, Addison’s disease, which results
to provide nutrition to people who cannot otherwise
from decreased secretion of the hormone aldosterone,
take adequate amounts of nutrition. Glucose solutions
impairs the ability of the kidneys to reabsorb sodium
are widely used, and amino acid and homogenized fat
and can cause a modest degree of hyponatremia.
solutions are used to a lesser extent. When these solu-
Hyponatremia can also be associated with excess
tions are administered, their concentrations of osmoti-
water retention, which dilutes the sodium in the extra-
cally active substances are usually adjusted nearly to
cellular fluid, a condition that is referred to as hypo-
isotonicity, or they are given slowly enough that they do
osmotic overhydration. For example, excessive secretion
not upset the osmotic equilibrium of the body fluids.
of antidiuretic hormone, which causes the kidney
Table 25-4
Abnormalities of Body Fluid Volume Regulation: Hyponatremia and Hypernatremia
Plasma Na+
Extracellular
Intracellular
Abnormality
Cause
Concentration
Fluid Volume
Fluid Volume
Hypo-osmotic dehydration
Adrenal insufficiency; overuse of diuretics
Ø
Ø
≠
Hypo-osmotic overhydration
Excess ADH; bronchogenic tumor
Ø
≠
≠
Hyper-osmotic dehydration
Diabetes insipidus; excessive sweating
≠
Ø
Ø
Hyper-osmotic overhydration
Cushing’s disease; primary aldosteronism
≠
≠
Ø
ADH, antidiuretic hormone.
302
Unit V The Body Fluids and Kidneys
tubules to reabsorb more water, can lead to hypona-
cell can no longer be pumped out of the cells, and the
tremia and overhydration.
excess sodium ions inside the cells cause osmosis of
water into the cells. Sometimes this can increase intra-
cellular volume of a tissue area—even of an entire
Causes of Hypernatremia: Water
ischemic leg, for example—to two to three times
Loss or Excess Sodium
normal. When this occurs, it is usually a prelude to
death of the tissue.
Increased plasma sodium concentration, which also
Intracellular edema can also occur in inflamed
causes increased osmolarity, can be due to either loss of
tissues. Inflammation usually has a direct effect on the
water from the extracellular fluid, which concentrates
the sodium ions, or excess sodium in the extracellular
cell membranes to increase their permeability, allow-
fluid. When there is primary loss of water from the
ing sodium and other ions to diffuse into the interior
extracellular fluid, this results in hyperosmotic dehydra-
of the cell, with subsequent osmosis of water into the
tion. This condition can occur from an inability to
cells.
secrete antidiuretic hormone, which is needed for the
kidneys to conserve water. As a result of lack of anti-
diuretic hormone, the kidneys excrete large amounts
Extracellular Edema
of dilute urine
(a disorder referred to as diabetes
insipidus), causing dehydration and increased concen-
Extracellular fluid edema occurs when there is excess
tration of sodium chloride in the extracellular fluid. In
fluid accumulation in the extracellular spaces. There
certain types of renal diseases, the kidneys cannot
are two general causes of extracellular edema: (1)
respond to antidiuretic hormone, also causing a type of
nephrogenic diabetes insipidus. A more common cause
abnormal leakage of fluid from the plasma to the inter-
of hypernatremia associated with decreased extracellu-
stitial spaces across the capillaries, and (2) failure of
lar fluid volume is dehydration caused by water intake
the lymphatics to return fluid from the interstitium
that is less than water loss, as can occur with sweating
back into the blood. The most common clinical cause
during prolonged, heavy exercise.
of interstitial fluid accumulation is excessive capillary
Hypernatremia can also occur as a result of excessive
fluid filtration.
sodium chloride added to the extracellular fluid. This
often results in hyperosmotic overhydration because
Factors That Can Increase Capillary Filtration
excess extracellular sodium chloride is usually associ-
To understand the causes of excessive capillary filtra-
ated with at least some degree of water retention by the
tion, it is useful to review the determinants of capillary
kidneys as well. For example, excessive secretion of the
filtration discussed in Chapter 16. Mathematically, cap-
sodium-retaining hormone aldosterone can cause a mild
illary filtration rate can be expressed as
degree of hypernatremia and overhydration. The reason
that the hypernatremia is not more severe is that
Filtration = Kf ¥ (Pc - Pif - pc + pif),
increased aldosterone secretion causes the kidneys to
reabsorb greater amounts of water as well as sodium.
where Kf is the capillary filtration coefficient
(the
Thus, in analyzing abnormalities of plasma sodium
product of the permeability and surface area of the
concentration and deciding on proper therapy, one
capillaries), Pc is the capillary hydrostatic pressure, Pif
should first determine whether the abnormality is
is the interstitial fluid hydrostatic pressure, pc is the
caused by a primary loss or gain of sodium or a primary
capillary plasma colloid osmotic pressure, and pif is the
loss or gain of water.
interstitial fluid colloid osmotic pressure. From this
equation, one can see that any one of the following
changes can increase the capillary filtration rate:
Edema: Excess Fluid
∑ Increased capillary filtration coefficient.
in the Tissues
∑ Increased capillary hydrostatic pressure.
∑ Decreased plasma colloid osmotic pressure.
Edema refers to the presence of excess fluid in the
body tissues. In most instances, edema occurs mainly
Lymphatic Blockage Causes Edema
in the extracellular fluid compartment, but it can
When lymphatic blockage occurs, edema can become
involve intracellular fluid as well.
especially severe because plasma proteins that leak
into the interstitium have no other way to be removed.
The rise in protein concentration raises the colloid
Intracellular Edema
osmotic pressure of the interstitial fluid, which draws
even more fluid out of the capillaries.
Two conditions are especially prone to cause intracel-
Blockage of lymph flow can be especially severe
lular swelling: (1) depression of the metabolic systems
with infections of the lymph nodes, such as occurs with
of the tissues, and (2) lack of adequate nutrition to
infection by filaria nematodes. Blockage of the lymph
the cells. For example, when blood flow to a tissue
vessels can occur in certain types of cancer or after
is decreased, the delivery of oxygen and nutrients is
surgery in which lymph vessels are removed or
reduced. If the blood flow becomes too low to main-
obstructed. For example, large numbers of lymph
tain normal tissue metabolism, the cell membrane
vessels are removed during radical mastectomy,
ionic pumps become depressed. When this occurs,
impairing removal of fluid from the breast and arm
sodium ions that normally leak into the interior of the
areas and causing edema and swelling of the tissue
Chapter 25
The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema
303
spaces. A few lymph vessels eventually regrow after
factors acting together cause serious generalized extra-
this type of surgery, so that the interstitial edema is
cellular edema.
usually temporary.
In patients with left-sided heart failure but without
significant failure of the right side of the heart, blood is
pumped into the lungs normally by the right side of the
Summary of Causes of
heart but cannot escape easily from the pulmonary
veins to the left side of the heart because this part of the
Extracellular Edema
heart has been greatly weakened. Consequently, all
A large number of conditions can cause fluid accumu-
the pulmonary vascular pressures, including pulmon-
lation in the interstitial spaces by the abnormal leaking
ary capillary pressure, rise far above normal, causing
of fluid from the capillaries or by preventing the lym-
serious and life-threatening pulmonary edema. When
phatics from returning fluid from the interstitium back
untreated, fluid accumulation in the lungs can rapidly
to the circulation. The following is a partial list of con-
progress, causing death within a few hours.
ditions that can cause extracellular edema by these two
types of abnormalities:
Edema Caused by Decreased Kidney Excretion of Salt and Water.
I.
Increased capillary pressure
As discussed earlier, most sodium chloride added to the
A. Excessive kidney retention of salt and water
blood remains in the extracellular compartment, and
1. Acute or chronic kidney failure
only small amounts enter the cells. Therefore, in kidney
2. Mineralocorticoid excess
diseases that compromise urinary excretion of salt and
B. High venous pressure and venous constriction
water, large amounts of sodium chloride and water are
1. Heart failure
added to the extracellular fluid. Most of this salt and
2. Venous obstruction
water leaks from the blood into the interstitial spaces,
3. Failure of venous pumps
but some remains in the blood. The main effects of this
(a) Paralysis of muscles
are to cause (1) widespread increases in interstitial fluid
(b) Immobilization of parts of the body
volume (extracellular edema) and
(2) hypertension
(c) Failure of venous valves
because of the increase in blood volume, as explained
C. Decreased arteriolar resistance
in Chapter 19. As an example, children who develop
1. Excessive body heat
acute glomerulonephritis, in which the renal glomeruli
2. Insufficiency of sympathetic nervous system
are injured by inflammation and therefore fail to filter
3. Vasodilator drugs
adequate amounts of fluid, also develop serious
II.
Decreased plasma proteins
extracellular fluid edema in the entire body; along with
A. Loss of proteins in urine (nephrotic
the edema, these children usually develop severe
syndrome)
hypertension.
B. Loss of protein from denuded skin areas
1. Burns
Edema Caused by Decreased Plasma Proteins. A reduction in
2. Wounds
plasma concentration of proteins because of either
C. Failure to produce proteins
failure to produce normal amounts of proteins or
1. Liver disease (e.g., cirrhosis)
leakage of proteins from the plasma causes the plasma
2. Serious protein or caloric malnutrition
colloid osmotic pressure to fall. This leads to increased
III.
Increased capillary permeability
capillary filtration throughout the body as well as extra-
A. Immune reactions that cause release of
cellular edema.
histamine and other immune products
One of the most important causes of decreased
B. Toxins
plasma protein concentration is loss of proteins in the
C. Bacterial infections
urine in certain kidney diseases, a condition referred to
D. Vitamin deficiency, especially vitamin C
as nephrotic syndrome. Multiple types of renal diseases
E. Prolonged ischemia
can damage the membranes of the renal glomeruli,
F. Burns
causing the membranes to become leaky to the plasma
IV.
Blockage of lymph return
proteins and often allowing large quantities of these
A. Cancer
proteins to pass into the urine. When this loss exceeds
B. Infections (e.g., filaria nematodes)
the ability of the body to synthesize proteins, a reduc-
C. Surgery
tion in plasma protein concentration occurs. Serious
D. Congenital absence or abnormality of
generalized edema occurs when the plasma protein con-
lymphatic vessels
centration falls below 2.5 g/100 ml.
Cirrhosis of the liver is another condition that causes
Edema Caused by Heart Failure. One of the most serious and
a reduction in plasma protein concentration. Cirrhosis
most common causes of edema is heart failure. In heart
means development of large amounts of fibrous tissue
failure, the heart fails to pump blood normally from the
among the liver parenchymal cells. One result is failure
veins into the arteries; this raises venous pressure and
of these cells to produce sufficient plasma proteins,
capillary pressure, causing increased capillary filtration.
leading to decreased plasma colloid osmotic pressure
In addition, the arterial pressure tends to fall, causing
and the generalized edema that goes with this condition.
decreased excretion of salt and water by the kidneys,
Another way that liver cirrhosis causes edema is that
which increases blood volume and further raises capil-
the liver fibrosis sometimes compresses the abdominal
lary hydrostatic pressure to cause still more edema.
portal venous drainage vessels as they pass through the
Also, diminished blood flow to the kidneys stimulates
liver before emptying back into the general circulation.
secretion of renin, causing increased formation of
Blockage of this portal venous outflow raises capillary
angiotensin II and increased secretion of aldosterone,
hydrostatic pressure throughout the gastrointestinal
both of which cause additional salt and water retention
area and further increases filtration of fluid out of
by the kidneys. Thus, in untreated heart failure, all these
the plasma into the intra-abdominal areas. When this
304
Unit V The Body Fluids and Kidneys
occurs, the combined effects of decreased plasma
tissues with relatively small additional increases in
protein concentration and high portal capillary pres-
interstitial fluid hydrostatic pressure. Thus, in the pos-
sures cause transudation of large amounts of fluid and
itive tissue pressure range, this safety factor against
protein into the abdominal cavity, a condition referred
edema is lost because of the large increase in compli-
to as ascites.
ance of the tissues.
Importance of Interstitial Gel in Preventing Fluid Accumulation in
Safety Factors That Normally
the Interstitium. Note in Figure
25-7 that in normal
tissues with negative interstitial fluid pressure, virtually
Prevent Edema
all the fluid in the interstitium is in gel form. That is, the
fluid is bound in a proteoglycan meshwork so that there
Even though many disturbances can cause edema,
are virtually no “free” fluid spaces larger than a few
usually the abnormality must be severe before serious
hundredths of a micrometer in diameter. The impor-
edema develops. The reason for this is that three major
tance of the gel is that it prevents fluid from flowing
safety factors prevent excessive fluid accumulation in
easily through the tissues because of impediment from
the interstitial spaces: (1) low compliance of the inter-
the “brush pile” of trillions of proteoglycan filaments.
stitium when interstitial fluid pressure is in the nega-
Also, when the interstitial fluid pressure falls to very
tive pressure range, (2) the ability of lymph flow
negative values, the gel does not contract greatly
to increase 10- to 50-fold, and (3) washdown of inter-
because the meshwork of proteoglycan filaments offers
an elastic resistance to compression. In the negative
stitial fluid protein concentration, which reduces
fluid pressure range, the interstitial fluid volume does
interstitial fluid colloid osmotic pressure as capillary
not change greatly, regardless of whether the degree of
filtration increases.
suction is only a few millimeters of mercury negative
pressure or 10 to 20 mm Hg negative pressure. In other
Safety Factor Caused by Low Compliance
words, the compliance of the tissues is very low in the
of the Interstitium in the Negative
negative pressure range.
Pressure Range
In Chapter 16, we noted that interstitial fluid hydro-
static pressure in most loose subcutaneous tissues of
the body is slightly less than atmospheric pressure,
60
averaging about -3 mm Hg. This slight suction in the
tissues helps hold the tissues together. Figure 25-7
56
Free fluid
Gel fluid
shows the approximate relations between different
52
levels of interstitial fluid pressure and interstitial fluid
volume, as extrapolated to the human being from
48
animal studies. Note in Figure 25-7 that as long as the
44
interstitial fluid pressure is in the negative range, small
changes in interstitial fluid volume are associated with
40
relatively large changes in interstitial fluid hydrostatic
36
pressure. Therefore, in the negative pressure range, the
32
compliance of the tissues, defined as the change in
volume per millimeter of mercury pressure change, is
28
(High
low.
compliance)
24
How does the low compliance of the tissues in the
negative pressure range act as a safety factor against
20
Normal
edema? To answer this question, recall the determi-
16
nants of capillary filtration discussed previously. When
interstitial fluid hydrostatic pressure increases, this
12
increased pressure tends to oppose further capillary
8
(Low compliance)
filtration. Therefore, as long as the interstitial fluid
4
hydrostatic pressure is in the negative pressure range,
small increases in interstitial fluid volume cause rela-
0
tively large increases in interstitial fluid hydrostatic
-10
-8
-6
-4
-2
0
2
4
6
pressure, opposing further filtration of fluid into the
Interstitial free fluid pressure
tissues.
(mm Hg)
Because the normal interstitial fluid hydrostatic
pressure is -3 mm Hg, the interstitial fluid hydrostatic
pressure must increase by about 3 mm Hg before large
Figure 25-7
amounts of fluid will begin to accumulate in the tissues.
Therefore, the safety factor against edema is a change
Relation between interstitial fluid hydrostatic pressure and inter-
stitial fluid volumes, including total volume, free fluid volume, and
of interstitial fluid pressure of about 3 mm Hg.
gel fluid volume, for loose tissues such as skin. Note that signifi-
Once interstitial fluid pressure rises above 0 mm Hg,
cant amounts of free fluid occur only when the interstitial fluid pres-
the compliance of the tissues increases markedly,
sure becomes positive. (Modified from Guyton AC, Granger HJ,
allowing large amounts of fluid to accumulate in the
Taylor AE: Interstitial fluid pressure. Physiol Rev 51:527, 1971.)
Chapter 25
The Body Fluid Compartments: Extracellular and Intracellular Fluids; Interstitial Fluid and Edema
305
By contrast, when interstitial fluid pressure rises to
caused by increased lymph flow has been calculated to
the positive pressure range, there is a tremendous accu-
be about 7 mm Hg.
mulation of free fluid in the tissues. In this pressure
range, the tissues are compliant, allowing large amounts
“Washdown” of the Interstitial Fluid Protein as
of fluid to accumulate with relatively small additional
a Safety Factor Against Edema
increases in interstitial fluid hydrostatic pressure. Most
As increased amounts of fluid are filtered into the
of the extra fluid that accumulates is “free fluid” because
it pushes the brush pile of proteoglycan filaments apart.
interstitium, the interstitial fluid pressure increases,
Therefore, the fluid can flow freely through the tissue
causing increased lymph flow. In most tissues, the
spaces because it is not in gel form. When this occurs,
protein concentration of the interstitium decreases as
the edema is said to be pitting edema because one can
lymph flow is increased, because larger amounts of
press the thumb against the tissue area and push the
protein are carried away than can be filtered out of the
fluid out of the area. When the thumb is removed, a pit
capillaries; the reason for this is that the capillaries
is left in the skin for a few seconds until the fluid flows
are relatively impermeable to proteins, compared
back from the surrounding tissues. This type of edema
with the lymph vessels. Therefore, the proteins are
is distinguished from nonpitting edema, which occurs
“washed out” of the interstitial fluid as lymph flow
when the tissue cells swell instead of the interstitium or
increases.
when the fluid in the interstitium becomes clotted with
fibrinogen so that it cannot move freely within the tissue
Because the interstitial fluid colloid osmotic pres-
spaces.
sure caused by the proteins tends to draw fluid out of
the capillaries, decreasing the interstitial fluid proteins
Importance of the Proteoglycan Filaments as a “Spacer” for the
lowers the net filtration force across the capillaries and
Cells and in Preventing Rapid Flow of Fluid in the Tissues. The
tends to prevent further accumulation of fluid. The
proteoglycan filaments, along with much larger collagen
safety factor from this effect has been calculated to be
fibrils in the interstitial spaces, act as a “spacer” between
about 7 mm Hg.
the cells. Nutrients and ions do not diffuse readily
through cell membranes; therefore, without adequate
spacing between the cells, these nutrients, electrolytes,
Summary of Safety Factors That
and cell waste products could not be rapidly exchanged
Prevent Edema
between the blood capillaries and cells located at a dis-
tance from one another.
Putting together all the safety factors against edema, we
The proteoglycan filaments also prevent fluid from
find the following:
flowing too easily through the tissue spaces. If it were
1. The safety factor caused by low tissue compliance
not for the proteoglycan filaments, the simple act of a
in the negative pressure range is about 3 mm Hg.
person standing up would cause large amounts of inter-
2. The safety factor caused by increased lymph flow is
stitial fluid to flow from the upper body to the lower
about 7 mm Hg.
body. When too much fluid accumulates in the intersti-
3. The safety factor caused by washdown of proteins
tium, as occurs in edema, this extra fluid creates large
from the interstitial spaces is about 7 mm Hg.
channels that allow the fluid to flow readily through the
Therefore, the total safety factor against edema is
interstitium. Therefore, when severe edema occurs in
about 17 mm Hg. This means that the capillary pres-
the legs, the edema fluid often can be decreased by
sure in a peripheral tissue could theoretically rise by
simply elevating the legs.
17 mm Hg, or approximately double the normal value,
Even though fluid does not flow easily through the
before marked edema would occur.
tissues in the presence of the compacted proteoglycan
filaments, different substances within the fluid can
diffuse through the tissues at least 95 per cent as easily
Fluids in the “Potential
as they normally diffuse. Therefore, the usual diffusion
of nutrients to the cells and the removal of waste prod-
Spaces” of the Body
ucts from the cells are not compromised by the proteo-
glycan filaments of the interstitium.
Perhaps the best way to describe a “potential space”
is to list some examples: pleural cavity, pericardial
Increased Lymph Flow as a Safety Factor
cavity, peritoneal cavity, and synovial cavities, includ-
Against Edema
ing both the joint cavities and the bursae. Virtually all
A major function of the lymphatic system is to return
these potential spaces have surfaces that almost touch
to the circulation the fluid and proteins filtered from
each other, with only a thin layer of fluid in between,
the capillaries into the interstitium. Without this con-
and the surfaces slide over each other. To facilitate the
tinuous return of the filtered proteins and fluid to the
sliding, a viscous proteinaceous fluid lubricates the
blood, the plasma volume would be rapidly depleted,
surfaces.
and interstitial edema would occur.
The lymphatics act as a safety factor against edema
Fluid Is Exchanged Between the Capillaries and the Potential
because lymph flow can increase 10- to 50-fold when
Spaces. The surface membrane of a potential space
fluid begins to accumulate in the tissues. This allows
usually does not offer significant resistance to the
the lymphatics to carry away large amounts of fluid
passage of fluids, electrolytes, or even proteins, which
and proteins in response to increased capillary filtra-
all move back and forth between the space and the
tion, preventing the interstitial pressure from rising
interstitial fluid in the surrounding tissue with relative
into the positive pressure range. The safety factor
ease. Therefore, each potential space is in reality a
306
Unit V The Body Fluids and Kidneys
large tissue space. Consequently, fluid in the capillar-
References
ies adjacent to the potential space diffuses not only
into the interstitial fluid but also into the potential
Amiry-Moghaddam M, Ottersen OP: The molecular basis of
space.
water transport in the brain. Nat Rev Neurosci 4:991, 2003.
Aukland K: Why don’t our feet swell in the upright position?
Lymphatic Vessels Drain Protein from the Potential Spaces.
News Physiol Sci 9:214, 1994.
Basnyat B, Murdoch DR: High-altitude illness. Lancet
Proteins collect in the potential spaces because of
361:1967, 2003.
leakage out of the capillaries, similar to the collection
Cho S, Atwood JE: Peripheral edema. Am J Med 113:580,
of protein in the interstitial spaces throughout the
2002.
body. The protein must be removed through lymphat-
Decaux G, Soupart A: Treatment of symptomatic hypona-
ics or other channels and returned to the circulation.
tremia. Am J Med Sci 326:25, 2003.
Each potential space is either directly or indirectly
Drake RE, Doursout MF: Pulmonary edema and elevated
connected with lymph vessels. In some cases, such as
left atrial pressure: four hours and beyond. News Physiol
the pleural cavity and peritoneal cavity, large lymph
Sci 17:223, 2002.
vessels arise directly from the cavity itself.
Gashev AA: Physiologic aspects of lymphatic contractile
function: current perspectives. Ann N Y Acad Sci 979:178,
2002.
Edema Fluid in the Potential Spaces Is Called “Effusion.”
Guyton AC, Granger HJ, Taylor AE: Interstitial fluid pres-
When edema occurs in the subcutaneous tissues adja-
sure. Physiol Rev 51:527, 1971.
cent to the potential space, edema fluid usually collects
Guyton AC, Taylor AE, Granger HJ: Circulatory Physiology
in the potential space as well, and this fluid is called
II: Dynamics and Control of the Body Fluids. Philadel-
effusion. Thus, lymph blockage or any of the multiple
phia: WB Saunders, 1975.
abnormalities that can cause excessive capillary filtra-
Halperin ML, Bohn D: Clinical approach to disorders of salt
tion can cause effusion in the same way that intersti-
and water balance: emphasis on integrative physiology.
tial edema is caused. The abdominal cavity is especially
Crit Care Clin 18:249, 2002.
prone to collect effusion fluid, and in this instance, the
Jussila L, Alitalo K: Vascular growth factors and lymphan-
effusion is called ascites. In serious cases, 20 liters or
giogenesis. Physiol Rev 82:673, 2002.
Matthay MA, Folkesson HG, Clerici C: Lung epithelial fluid
more of ascitic fluid can accumulate.
transport and the resolution of pulmonary edema. Physiol
The other potential spaces, such as the pleural
Rev 82:569, 2002.
cavity, pericardial cavity, and joint spaces, can become
Michel CC: Exchange of fluid and solutes across microvas-
seriously swollen when there is generalized edema.
cular walls. In Seldin DW, Giebisch G (eds): The Kidney—
Also, injury or local infection in any one of the cavi-
Physiology & Pathophysiology,
3rd ed. Philadelphia:
ties often blocks the lymph drainage, causing isolated
Lippincott Williams & Wilkins, 2000.
swelling in the cavity.
Moritz ML, Ayus JC: Disorders of water metabolism in
The dynamics of fluid exchange in the pleural cavity
children: hyponatremia and hypernatremia. Pediatr Rev
are discussed in detail in Chapter 38. These dynamics
23:371, 2002.
are mainly representative of all the other potential
Saaristo A, Karkkainen MJ, Alitalo K: Insights into the
molecular pathogenesis and targeted treatment of lym-
spaces as well. It is especially interesting that the
phedema. Ann N Y Acad Sci 979:94, 2002.
normal fluid pressure in most or all of the potential
Winaver J, Abassi Z, Green J, Skorecki KL: Control of extra-
spaces in the nonedematous state is negative in the
cellular fluid volume and the pathophysiology of edema
same way that this pressure is negative (subatmos-
formation. In Brenner BM, Rector FC (eds): The Kidney,
pheric) in loose subcutaneous tissue. For instance, the
6th ed. Philadelphia: WB Saunders, 2000.
interstitial fluid hydrostatic pressure is normally about
-7 to -8 mm Hg in the pleural cavity, -3 to -5 mm Hg
in the joint spaces, and -5 to -6 mm Hg in the peri-
cardial cavity.
C H A P T E R
2
6
Urine Formation by the Kidneys:
I. Glomerular Filtration, Renal
Blood Flow, and Their Control
Multiple Functions of the
Kidneys in Homeostasis
Most people are familiar with one important func-
tion of the kidneys—to rid the body of waste mate-
rials that are either ingested or produced by
metabolism. A second function that is especially
critical is to control the volume and composition of
the body fluids. For water and virtually all electrolytes in the body, the balance
between intake (due to ingestion or metabolic production) and output (due to
excretion or metabolic consumption) is maintained in large part by the kidneys.
This regulatory function of the kidneys maintains the stable environment of the
cells necessary for them to perform their various activities.
The kidneys perform their most important functions by filtering the plasma
and removing substances from the filtrate at variable rates, depending on the
needs of the body. Ultimately, the kidneys “clear” unwanted substances from
the filtrate (and therefore from the blood) by excreting them in the urine while
returning substances that are needed back to the blood.
Although this chapter and the next few chapters focus mainly on the control
of renal excretion, it is important to recognize that the kidneys serve multiple
functions, including the following:
∑ Excretion of metabolic waste products and foreign chemicals
∑ Regulation of water and electrolyte balances
∑ Regulation of body fluid osmolality and electrolyte concentrations
∑ Regulation of arterial pressure
∑ Regulation of acid-base balance
∑ Secretion, metabolism, and excretion of hormones
∑ Gluconeogenesis
Excretion of Metabolic Waste Products, Foreign Chemicals, Drugs, and Hormone Metabo-
lites. The kidneys are the primary means for eliminating waste products of
metabolism that are no longer needed by the body. These products include urea
(from the metabolism of amino acids), creatinine (from muscle creatine), uric
acid (from nucleic acids), end products of hemoglobin breakdown (such as
bilirubin), and metabolites of various hormones. These waste products must be
eliminated from the body as rapidly as they are produced. The kidneys also
eliminate most toxins and other foreign substances that are either produced by
the body or ingested, such as pesticides, drugs, and food additives.
Regulation of Water and Electrolyte Balances. For maintenance of homeostasis, excre-
tion of water and electrolytes must precisely match intake. If intake exceeds
excretion, the amount of that substance in the body will increase. If intake is
less than excretion, the amount of that substance in the body will decrease.
Intake of water and many electrolytes is governed mainly by a person’s eating
and drinking habits, requiring the kidneys to adjust their excretion rates to
match the intake of various substances. Figure 26-1 shows the response of the
kidneys to a sudden 10-fold increase in sodium intake from a low level of
30 mEq/day to a high level of 300 mEq/day. Within 2 to 3 days after raising the
307
308
Unit V The Body Fluids and Kidneys
Regulation of Acid-Base Balance. The kidneys contribute
to acid-base regulation, along with the lungs and body
Sodium
fluid buffers, by excreting acids and by regulating the
retention
body fluid buffer stores. The kidneys are the only
300
Intake
means of eliminating from the body certain types of
acids, such as sulfuric acid and phosphoric acid, gen-
200
erated by the metabolism of proteins.
Excretion
Sodium
100
loss
Regulation of Erythrocyte Production. The kidneys secrete
erythropoietin, which stimulates the production of red
0
blood cells, as discussed in Chapter 32. One important
stimulus for erythropoietin secretion by the kidneys is
15
hypoxia. The kidneys normally account for almost all
the erythropoietin secreted into the circulation. In
people with severe kidney disease or who have had
10
their kidneys removed and have been placed on
hemodialysis, severe anemia develops as a result of
5
decreased erythropoietin production.
-4
-2
0
2
4
6
8
10
12
14
Time (days)
Regulation of
1,25-Dihydroxyvitamin D3
Production. The
kidneys produce the active form of vitamin D, 1,25-
dihydroxyvitamin D3 (calcitriol), by hydroxylating this
Figure 26-1
vitamin at the “number 1” position. Calcitriol is essen-
tial for normal calcium deposition in bone and calcium
Effect of increasing sodium intake 10-fold (from 30 to 300 mEq/
day) on urinary sodium excretion and extracellular fluid volume.
reabsorption by the gastrointestinal tract. As discussed
The shaded areas represent the net sodium retention or the net
in Chapter 79, calcitriol plays an important role in
sodium loss, determined from the difference between sodium
calcium and phosphate regulation.
intake and sodium excretion.
Glucose Synthesis. The kidneys synthesize glucose from
amino acids and other precursors during prolonged
fasting, a process referred to as gluconeogenesis. The
sodium intake, renal excretion also increases to about
kidneys’ capacity to add glucose to the blood during
300 mEq/day, so that a balance between intake and
prolonged periods of fasting rivals that of the liver.
output is re-established. However, during the 2 to 3
With chronic kidney disease or acute failure of the
days of renal adaptation to the high sodium intake,
kidneys, these homeostatic functions are disrupted,
there is a modest accumulation of sodium that raises
and severe abnormalities of body fluid volumes and
extracellular fluid volume slightly and triggers hor-
composition rapidly occur. With complete renal
monal changes and other compensatory responses
failure, enough potassium, acids, fluid, and other sub-
that signal the kidneys to increase their sodium
stances accumulate in the body to cause death within
excretion.
a few days, unless clinical interventions such as
The capacity of the kidneys to alter sodium excre-
hemodialysis are initiated to restore, at least partially,
tion in response to changes in sodium intake is enor-
the body fluid and electrolyte balances.
mous. Experimental studies have shown that in many
people, sodium intake can be increased to 1500 mEq/
day (more than 10 times normal) or decreased to
Physiologic Anatomy
10 mEq/day (less than one tenth normal) with rela-
of the Kidneys
tively small changes in extracellular fluid volume or
plasma sodium concentration. This is also true for
General Organization of the Kidneys
water and for most other electrolytes, such as chloride,
potassium, calcium, hydrogen, magnesium, and phos-
and Urinary Tract
phate ions. In the next few chapters, we discuss the spe-
cific mechanisms that permit the kidneys to perform
The two kidneys lie on the posterior wall of the
these amazing feats of homeostasis.
abdomen, outside the peritoneal cavity (Figure 26-2).
Each kidney of the adult human weighs about 150
Regulation of Arterial Pressure. As discussed in Chapter
grams and is about the size of a clenched fist. The
19, the kidneys play a dominant role in long-term reg-
medial side of each kidney contains an indented region
ulation of arterial pressure by excreting variable
called the hilum through which pass the renal artery
amounts of sodium and water. The kidneys also con-
and vein, lymphatics, nerve supply, and ureter, which
tribute to short-term arterial pressure regulation by
carries the final urine from the kidney to the bladder,
secreting vasoactive factors or substances, such as
where it is stored until emptied. The kidney is sur-
renin, that lead to the formation of vasoactive prod-
rounded by a tough, fibrous capsule that protects its
ucts (e.g., angiotensin II).
delicate inner structures.
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
309
Minor calyx
Major calyx
Nephron (enlarged)
Papilla
Renal cortex
Renal medulla
Renal pelvis
Renal
Kidney
artery
Renal pyramid
Capsule
Ureter
Ureter
Kidney
Figure 26-2
Bladder
Urethra
General organization of the
kidneys and the urinary system.
If the kidney is bisected from top to bottom, the two
much lower hydrostatic pressure in the peritubular
major regions that can be visualized are the outer
capillaries (about 13 mm Hg) permits rapid fluid reab-
cortex and the inner region referred to as the medulla.
sorption. By adjusting the resistance of the afferent
The medulla is divided into multiple cone-shaped
and efferent arterioles, the kidneys can regulate the
masses of tissue called renal pyramids. The base of
hydrostatic pressure in both the glomerular and the
each pyramid originates at the border between the
peritubular capillaries, thereby changing the rate of
cortex and medulla and terminates in the papilla,
which projects into the space of the renal pelvis, a
funnel-shaped continuation of the upper end of the
ureter. The outer border of the pelvis is divided into
open-ended pouches called major calyces that extend
downward and divide into minor calyces, which collect
Interlobar
arteries
urine from the tubules of each papilla. The walls of the
Renal artery
calyces, pelvis, and ureter contain contractile elements
that propel the urine toward the bladder, where urine
Arcuate arteries
Segmental
is stored until it is emptied by micturition, discussed in
arteries
later in this chapter.
Interlobular
arterioles
Renal Blood Supply
Blood flow to the two kidneys is normally about 22 per
Efferent
Bowman's
cent of the cardiac output, or 1100 ml/min. The renal
Glomerulus
arteriole
capsule
Proximal tubule
artery enters the kidney through the hilum and then
branches progressively to form the interlobar arteries,
Juxtaglomerular
Cortical
apparatus
arcuate arteries, interlobular arteries (also called radial
collecting tubule
arteries) and afferent arterioles, which lead to the
Afferent
arteriole
Distal tubule
glomerular capillaries, where large amounts of fluid
and solutes (except the plasma proteins) are filtered to
Arcuate
artery
begin urine formation (Figure 26-3). The distal ends of
the capillaries of each glomerulus coalesce to form the
Arcuate
Loop of
efferent arteriole, which leads to a second capillary
vein
Henle
network, the peritubular capillaries, that surrounds the
renal tubules.
Peritubular
The renal circulation is unique in that it has two cap-
capillaries
Collecting duct
illary beds, the glomerular and peritubular capillaries,
which are arranged in series and separated by the
Figure 26-3
efferent arterioles, which help regulate the hydrostatic
pressure in both sets of capillaries. High hydro-
Section of the human kidney showing the
major vessels that
static pressure in the glomerular capillaries (about
supply the blood flow to the kidney and schematic of the micro-
60 mm Hg) causes rapid fluid filtration, whereas a
circulation of each nephron.
310
Unit V The Body Fluids and Kidneys
glomerular filtration, tubular reabsorption, or both in
Proximal tubule
Cortex
response to body homeostatic demands.
Connecting tubule
The peritubular capillaries empty into the vessels of
Distal tubule
the venous system, which run parallel to the arteriolar
Bowman's capsule
vessels and progressively form the interlobular vein,
arcuate vein, interlobar vein, and renal vein, which
Macula densa
Cortical
leaves the kidney beside the renal artery and ureter.
collecting tubule
The Nephron Is the Functional Unit
Loop of Henle:
Medulla
Thick segment of
of the Kidney
ascending limb
Thin segment of
Medullary
Each kidney in the human contains about 1 million
ascending limb
collecting tubule
nephrons, each capable of forming urine. The kidney
Descending limb
cannot regenerate new nephrons. Therefore, with renal
injury, disease, or normal aging, there is a gradual
decrease in nephron number. After age 40, the number
of functioning nephrons usually decreases about 10
Collecting duct
per cent every 10 years; thus, at age 80, many people
have 40 per cent fewer functioning nephrons than they
did at age 40. This loss is not life threatening because
Figure 26-4
adaptive changes in the remaining nephrons allow
Basic tubular segments of the nephron. The relative lengths of the
them to excrete the proper amounts of water,
different tubular segments are not drawn to scale.
electrolytes, and waste products, as discussed in
Chapter 31.
Each nephron contains (1) a tuft of glomerular cap-
illaries called the glomerulus, through which large
amounts of fluid are filtered from the blood, and (2) a
eventually empty into the renal pelvis through the tips
long tubule in which the filtered fluid is converted into
of the renal papillae. In each kidney, there are about
urine on its way to the pelvis of the kidney (see Figure
250 of the very large collecting ducts, each of which
26-3).
collects urine from about 4000 nephrons.
The glomerulus contains a network of branching
and anastomosing glomerular capillaries that, com-
Regional Differences in Nephron Structure: Cortical and
pared with other capillaries, have high hydrostatic
Juxtamedullary Nephrons. Although each nephron has all
pressure (about 60 mm Hg). The glomerular capillar-
the components described earlier, there are some dif-
ies are covered by epithelial cells, and the total
ferences, depending on how deep the nephron lies
glomerulus is encased in Bowman’s capsule. Fluid
within the kidney mass. Those nephrons that have
filtered from the glomerular capillaries flows into
glomeruli located in the outer cortex are called corti-
Bowman’s capsule and then into the proximal tubule,
cal nephrons; they have short loops of Henle that
which lies in the cortex of the kidney (Figure 26-4).
penetrate only a short distance into the medulla
From the proximal tubule, fluid flows into the loop
(Figure 26-5).
of Henle, which dips into the renal medulla. Each loop
About 20 to 30 per cent of the nephrons have
consists of a descending and an ascending limb. The
glomeruli that lie deep in the renal cortex near the
walls of the descending limb and the lower end of the
medulla and are called juxtamedullary nephrons.
ascending limb are very thin and therefore are called
These nephrons have long loops of Henle that dip
the thin segment of the loop of Henle. After the ascend-
deeply into the medulla, in some cases all the way to
ing limb of the loop has returned partway back to the
the tips of the renal papillae.
cortex, its wall becomes much thicker, and it is referred
The vascular structures supplying the jux-
to as the thick segment of the ascending limb.
tamedullary nephrons also differ from those supplying
At the end of the thick ascending limb is a short
the cortical nephrons. For the cortical nephrons, the
segment, which is actually a plaque in its wall, known
entire tubular system is surrounded by an extensive
as the macula densa. As we discuss later, the macula
network of peritubular capillaries. For the jux-
densa plays an important role in controlling nephron
tamedullary nephrons, long efferent arterioles extend
function. Beyond the macula densa, fluid enters the
from the glomeruli down into the outer medulla and
distal tubule, which, like the proximal tubule, lies in the
then divide into specialized peritubular capillaries
renal cortex. This is followed by the connecting tubule
called vasa recta that extend downward into the
and the cortical collecting tubule, which lead to the cor-
medulla, lying side by side with the loops of Henle.
tical collecting duct. The initial parts of 8 to 10 cortical
Like the loops of Henle, the vasa recta return toward
collecting ducts join to form a single larger collecting
the cortex and empty into the cortical veins. This
duct that runs downward into the medulla and
specialized network of capillaries in the medulla plays
becomes the medullary collecting duct. The collecting
an essential role in the formation of a concentrated
ducts merge to form progressively larger ducts that
urine.
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
311
Micturition
Although the micturition reflex is an autonomic spinal
cord reflex, it can also be inhibited or facilitated by
Micturition is the process by which the urinary bladder
centers in the cerebral cortex or brain stem.
empties when it becomes filled. This involves two main
steps: First, the bladder fills progressively until the
Physiologic Anatomy and
tension in its walls rises above a threshold level; this
Nervous Connections
elicits the second step, which is a nervous reflex called
the micturition reflex that empties the bladder or, if this
of the Bladder
fails, at least causes a conscious desire to urinate.
The urinary bladder, shown in Figure 26-6, is a smooth
muscle chamber composed of two main parts: (1) the
Cortical
body, which is the major part of the bladder in which
Efferent
nephron
urine collects, and (2) the neck, which is a funnel-shaped
arteriole
extension of the body, passing inferiorly and anteriorly
Afferent
into the urogenital triangle and connecting with the
arteriole
urethra. The lower part of the bladder neck is also called
Juxtamedullary
the posterior urethra because of its relation to the
Interlobar
nephron
urethra.
artery
vein
The smooth muscle of the bladder is called the detru-
sor muscle. Its muscle fibers extend in all directions and,
when contracted, can increase the pressure in the
bladder to 40 to 60 mm Hg. Thus, contraction of the
Thick loop
of Henle
detrusor muscle is a major step in emptying the bladder.
Smooth muscle cells of the detrusor muscle fuse with
Interlobar
Vasa
one another so that low-resistance electrical pathways
artery
recta
exist from one muscle cell to the other. Therefore, an
vein
action potential can spread throughout the detrusor
muscle, from one muscle cell to the next, to cause con-
traction of the entire bladder at once.
On the posterior wall of the bladder, lying immedi-
Collecting
Thin loop
duct
ately above the bladder neck, is a small triangular area
of Henle
called the trigone. At the lowermost apex of the trigone,
the bladder neck opens into the posterior urethra, and
the two ureters enter the bladder at the uppermost
Duct of
angles of the trigone. The trigone can be identified by
Bellini
the fact that its mucosa, the inner lining of the bladder,
is smooth, in contrast to the remaining bladder mucosa,
which is folded to form rugae. Each ureter, as it enters
Figure 26-5
the bladder, courses obliquely through the detrusor
muscle and then passes another 1 to 2 centimeters
Schematic of relations between blood vessels and tubular struc-
tures and differences between cortical and juxtamedullary
beneath the bladder mucosa before emptying into the
nephrons.
bladder.
Ureter
L1
L2
L3
Body
Sympathetics
L4
L5
S1
S2
Trigone
Parasympathetics
S3
S4
Bladder neck
(posterior urethra)
Pudendal
External sphincter
Figure 26-6
Urinary bladder and its innervation.
312
Unit V The Body Fluids and Kidneys
The bladder neck (posterior urethra) is 2 to 3 cen-
thereby forcing urine from the renal pelvis toward the
timeters long, and its wall is composed of detrusor
bladder. The walls of the ureters contain smooth
muscle interlaced with a large amount of elastic tissue.
muscle and are innervated by both sympathetic and
The muscle in this area is called the internal sphincter.
parasympathetic nerves as well as by an intramural
Its natural tone normally keeps the bladder neck and
plexus of neurons and nerve fibers that extends along
posterior urethra empty of urine and, therefore, pre-
the entire length of the ureters. As with other visceral
vents emptying of the bladder until the pressure in the
smooth muscle, peristaltic contractions in the ureter are
main part of the bladder rises above a critical threshold.
enhanced by parasympathetic stimulation and inhibited
Beyond the posterior urethra, the urethra passes
through the urogenital diaphragm, which contains a
by sympathetic stimulation.
layer of muscle called the external sphincter of the
The ureters enter the bladder through the detrusor
bladder. This muscle is a voluntary skeletal muscle, in
muscle in the trigone region of the bladder, as shown
contrast to the muscle of the bladder body and bladder
in Figure 26-6. Normally, the ureters course obliquely
neck, which is entirely smooth muscle. The external
for several centimeters through the bladder wall. The
sphincter muscle is under voluntary control of the
normal tone of the detrusor muscle in the bladder wall
nervous system and can be used to consciously prevent
tends to compress the ureter, thereby preventing back-
urination even when involuntary controls are attempt-
flow of urine from the bladder when pressure builds
ing to empty the bladder.
up in the bladder during micturition or bladder
compression. Each peristaltic wave along the ureter
increases the pressure within the ureter so that the
Innervation of the Bladder
region passing through the bladder wall opens and
The principal nerve supply of the bladder is by way of
allows urine to flow into the bladder.
the pelvic nerves, which connect with the spinal cord
In some people, the distance that the ureter courses
through the sacral plexus, mainly connecting with cord
through the bladder wall is less than normal, so that
segments S-2 and S-3. Coursing through the pelvic
contraction of the bladder during micturition does not
nerves are both sensory nerve fibers and motor nerve
always lead to complete occlusion of the ureter. As a
fibers. The sensory fibers detect the degree of stretch in
result, some of the urine in the bladder is propelled
the bladder wall. Stretch signals from the posterior
backward into the ureter, a condition called vesi-
urethra are especially strong and are mainly re-
coureteral reflux. Such reflux can lead to enlargement
sponsible for initiating the reflexes that cause bladder
of the ureters and, if severe, can increase the pressure
emptying.
The motor nerves transmitted in the pelvic nerves are
in the renal calyces and structures of the renal
parasympathetic fibers. These terminate on ganglion
medulla, causing damage to these regions.
cells located in the wall of the bladder. Short postgan-
glionic nerves then innervate the detrusor muscle.
Pain Sensation in the Ureters, and the Ureterorenal Reflex.
In addition to the pelvic nerves, two other types of
The ureters are well supplied with pain nerve fibers.
innervation are important in bladder function. Most
When a ureter becomes blocked (e.g., by a ureteral
important are the skeletal motor fibers transmitted
stone), intense reflex constriction occurs, associated
through the pudendal nerve to the external bladder
with severe pain. Also, the pain impulses cause a sym-
sphincter. These are somatic nerve fibers that innervate
pathetic reflex back to the kidney to constrict the renal
and control the voluntary skeletal muscle of the sphinc-
arterioles, thereby decreasing urine output from the
ter. Also, the bladder receives sympathetic innervation
from the sympathetic chain through the hypogastric
kidney. This effect is called the ureterorenal reflex and
nerves, connecting mainly with the L-2 segment of the
is important for preventing excessive flow of fluid into
spinal cord. These sympathetic fibers stimulate mainly
the pelvis of a kidney with a blocked ureter.
the blood vessels and have little to do with bladder con-
traction. Some sensory nerve fibers also pass by way of
Filling of the Bladder
the sympathetic nerves and may be important in the
sensation of fullness and, in some instances, pain.
and Bladder Wall Tone;
the Cystometrogram
Transport of Urine from the
Figure 26-7 shows the approximate changes in intra-
vesicular pressure as the bladder fills with urine. When
Kidney Through the Ureters
there is no urine in the bladder, the intravesicular pres-
and into the Bladder
sure is about 0, but by the time 30 to 50 milliliters of
urine has collected, the pressure rises to 5 to 10 cen-
Urine that is expelled from the bladder has essentially
timeters of water. Additional urine—200 to 300 milli-
liters—can collect with only a small additional rise in
the same composition as fluid flowing out of the col-
pressure; this constant level of pressure is caused by
lecting ducts; there are no significant changes in the
intrinsic tone of the bladder wall itself. Beyond 300 to
composition of urine as it flows through the renal
400 milliliters, collection of more urine in the bladder
calyces and ureters to the bladder.
causes the pressure to rise rapidly.
Urine flowing from the collecting ducts into the
Superimposed on the tonic pressure changes during
renal calyces stretches the calyces and increases their
filling of the bladder are periodic acute increases in
inherent pacemaker activity, which in turn initiates
pressure that last from a few seconds to more than a
peristaltic contractions that spread to the renal pelvis
minute. The pressure peaks may rise only a few cen-
and then downward along the length of the ureter,
timeters of water or may rise to more than
100
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
313
Thus, the micturition reflex is a single complete cycle
of (1) progressive and rapid increase of pressure, (2) a
period of sustained pressure, and (3) return of the
pressure to the basal tone of the bladder. Once a mic-
turition reflex has occurred but has not succeeded in
40
Micturition
emptying the bladder, the nervous elements of this
contractions
reflex usually remain in an inhibited state for a few
minutes to 1 hour or more before another micturition
30
reflex occurs. As the bladder becomes more and more
filled, micturition reflexes occur more and more often
20
and more and more powerfully.
Once the micturition reflex becomes powerful
enough, it causes another reflex, which passes through
10
the pudendal nerves to the external sphincter to inhibit
it. If this inhibition is more potent in the brain than the
voluntary constrictor signals to the external sphincter,
0
urination will occur. If not, urination will not occur
0
100
200
300
400
until the bladder fills still further and the micturition
Volume (milliliters)
reflex becomes more powerful.
Figure 26-7
Facilitation or Inhibition of Micturition
by the Brain
Normal cystometrogram, showing also acute pressure waves
(dashed spikes) caused by micturition reflexes.
The micturition reflex is a completely autonomic
spinal cord reflex, but it can be inhibited or facilitated
centimeters of water. These pressure peaks are called
by centers in the brain. These centers include (1)
micturition waves in the cystometrogram and are caused
by the micturition reflex.
strong facilitative and inhibitory centers in the brain
stem, located mainly in the pons, and (2) several centers
located in the cerebral cortex that are mainly inhibitory
Micturition Reflex
but can become excitatory.
The micturition reflex is the basic cause of micturi-
Referring again to Figure 26-7, one can see that as the
tion, but the higher centers normally exert final control
bladder fills, many superimposed micturition contrac-
of micturition as follows:
tions begin to appear, as shown by the dashed spikes.
1. The higher centers keep the micturition reflex
They are the result of a stretch reflex initiated by
partially inhibited, except when micturition is
sensory stretch receptors in the bladder wall, especially
desired.
by the receptors in the posterior urethra when this
2. The higher centers can prevent micturition, even
area begins to fill with urine at the higher bladder pres-
if the micturition reflex occurs, by continual tonic
sures. Sensory signals from the bladder stretch recep-
contraction of the external bladder sphincter until
tors are conducted to the sacral segments of the cord
a convenient time presents itself.
through the pelvic nerves and then reflexively back
3. When it is time to urinate, the cortical centers can
again to the bladder through the parasympathetic
facilitate the sacral micturition centers to help
nerve fibers by way of these same nerves.
initiate a micturition reflex and at the same time
When the bladder is only partially filled, these mic-
inhibit the external urinary sphincter so that
turition contractions usually relax spontaneously after
urination can occur.
a fraction of a minute, the detrusor muscles stop con-
Voluntary urination is usually initiated in the fol-
tracting, and pressure falls back to the baseline. As the
lowing way: First, a person voluntarily contracts his or
bladder continues to fill, the micturition reflexes
her abdominal muscles, which increases the pressure
become more frequent and cause greater contractions
in the bladder and allows extra urine to enter the
of the detrusor muscle.
bladder neck and posterior urethra under pressure,
Once a micturition reflex begins, it is “self-regener-
thus stretching their walls. This stimulates the stretch
ative.” That is, initial contraction of the bladder acti-
receptors, which excites the micturition reflex and
vates the stretch receptors to cause a greater increase
simultaneously inhibits the external urethral sphincter.
in sensory impulses to the bladder and posterior
Ordinarily, all the urine will be emptied, with rarely
urethra, which causes a further increase in reflex con-
more than 5 to 10 milliliters left in the bladder.
traction of the bladder; thus, the cycle is repeated again
and again until the bladder has reached a strong
Abnormalities of Micturition
degree of contraction. Then, after a few seconds to
more than a minute, the self-regenerative reflex begins
Atonic Bladder Caused by Destruction of Sensory Nerve Fibers.
to fatigue and the regenerative cycle of the micturition
Micturition reflex contraction cannot occur if the
reflex ceases, permitting the bladder to relax.
sensory nerve fibers from the bladder to the spinal cord
314
Unit V The Body Fluids and Kidneys
are destroyed, thereby preventing transmission of
Afferent
Efferent
stretch signals from the bladder. When this happens, a
arteriole
arteriole
person loses bladder control, despite intact efferent
1. Filtration
fibers from the cord to the bladder and despite intact
2. Reabsorption
neurogenic connections within the brain. Instead of
3. Secretion
emptying periodically, the bladder fills to capacity and
Glomerular
4. Excretion
capillaries
overflows a few drops at a time through the urethra. This
is called overflow incontinence.
A common cause of atonic bladder is crush injury to
the sacral region of the spinal cord. Certain diseases can
also cause damage to the dorsal root nerve fibers that
Bowman's
capsule
1
enter the spinal cord. For example, syphilis can cause
constrictive fibrosis around the dorsal root nerve fibers,
destroying them. This condition is called tabes dorsalis,
2
and the resulting bladder condition is called tabetic
Peritubular
bladder.
capillaries
3
Automatic Bladder Caused by Spinal Cord Damage Above the
Sacral Region. If the spinal cord is damaged above the
sacral region but the sacral cord segments are still intact,
Renal
4
typical micturition reflexes can still occur. However,
vein
they are no longer controlled by the brain. During the
first few days to several weeks after the damage to the
cord has occurred, the micturition reflexes are sup-
pressed because of the state of “spinal shock” caused by
Urinary excretion
the sudden loss of facilitative impulses from the brain
Excretion = Filtration - Reabsorption + Secretion
stem and cerebrum. However, if the bladder is emptied
periodically by catheterization to prevent bladder injury
Figure 26-8
caused by overstretching of the bladder, the excitability
Basic kidney processes that determine the composition of the
of the micturition reflex gradually increases until typical
urine. Urinary excretion rate of a substance is equal to the rate at
micturition reflexes return; then, periodic (but unan-
which the substance is filtered minus its reabsorption rate plus the
nounced) bladder emptying occurs.
rate at which it is secreted from the peritubular capillary blood into
Some patients can still control urination in this con-
the tubules.
dition by stimulating the skin (scratching or tickling) in
the genital region, which sometimes elicits a micturition
reflex.
Urine formation begins when a large amount of
Uninhibited Neurogenic Bladder Caused by Lack of Inhibitory
fluid that is virtually free of protein is filtered from the
Signals from the Brain. Another abnormality of micturi-
tion is the so-called uninhibited neurogenic bladder,
glomerular capillaries into Bowman’s capsule. Most
which results in frequent and relatively uncontrolled
substances in the plasma, except for proteins, are freely
micturition. This condition derives from partial damage
filtered, so that their concentration in the glomerular
in the spinal cord or the brain stem that interrupts most
filtrate in Bowman’s capsule is almost the same as in
of the inhibitory signals. Therefore, facilitative impulses
the plasma. As filtered fluid leaves Bowman’s capsule
passing continually down the cord keep the sacral
and passes through the tubules, it is modified by reab-
centers so excitable that even a small quantity of urine
sorption of water and specific solutes back into the
elicits an uncontrollable micturition reflex, thereby pro-
blood or by secretion of other substances from the
moting frequent urination.
peritubular capillaries into the tubules.
Figure 26-9 shows the renal handling of four hypo-
thetical substances. The substance shown in panel A is
Urine Formation Results
freely filtered by the glomerular capillaries but is
from Glomerular Filtration,
neither reabsorbed nor secreted. Therefore, its excre-
tion rate is equal to the rate at which it was filtered.
Tubular Reabsorption, and
Certain waste products in the body, such as creatinine,
Tubular Secretion
are handled by the kidneys in this manner, allowing
excretion of essentially all that is filtered.
The rates at which different substances are excreted
In panel B, the substance is freely filtered but is also
in the urine represent the sum of three renal pro-
partly reabsorbed from the tubules back into the
cesses, shown in Figure 26-8: (1) glomerular filtration,
blood. Therefore, the rate of urinary excretion is less
(2) reabsorption of substances from the renal tubules
than the rate of filtration at the glomerular capillaries.
into the blood, and
(3) secretion of substances
In this case, the excretion rate is calculated as the fil-
from the blood into the renal tubules. Expressed
tration rate minus the reabsorption rate. This is typical
mathematically,
for many of the electrolytes of the body.
Urinary excretion rate = Filtration rate
In panel C, the substance is freely filtered at the
- Reabsorption rate + Secretion rate
glomerular capillaries but is not excreted into the
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
315
A. Filtration only
B. Filtration, partial
Filtration, Reabsorption, and
reabsorption
Secretion of Different Substances
Substance A
Substance B
In general, tubular reabsorption is quantitatively more
important than tubular secretion in the formation of
urine, but secretion plays an important role in deter-
mining the amounts of potassium and hydrogen ions
and a few other substances that are excreted in the
urine. Most substances that must be cleared from the
blood, especially the end products of metabolism such
as urea, creatinine, uric acid, and urates, are poorly
reabsorbed and are therefore excreted in large
amounts in the urine. Certain foreign substances and
drugs are also poorly reabsorbed but, in addition, are
Urine
Urine
secreted from the blood into the tubules, so that their
excretion rates are high. Conversely, electrolytes, such
C. Filtration, complete
D. Filtration, secretion
reabsorption
as sodium ions, chloride ions, and bicarbonate ions, are
highly reabsorbed, so that only small amounts appear
in the urine. Certain nutritional substances, such as
Substance C
Substance D
amino acids and glucose, are completely reabsorbed
from the tubules and do not appear in the urine even
though large amounts are filtered by the glomerular
capillaries.
Each of the processes—glomerular filtration,
tubular reabsorption, and tubular secretion—is regu-
lated according to the needs of the body. For example,
when there is excess sodium in the body, the rate at
which sodium is filtered increases and a smaller frac-
tion of the filtered sodium is reabsorbed, resulting in
Urine
Urine
increased urinary excretion of sodium.
Figure 26-9
For most substances, the rates of filtration and reab-
sorption are extremely large relative to the rates of
Renal handling of four hypothetical substances. A, The substance
excretion. Therefore, subtle adjustments of filtration or
is freely filtered but not reabsorbed. B, The substance is freely fil-
reabsorption can lead to relatively large changes in
tered, but part of the filtered load is reabsorbed back in the blood.
renal excretion. For example, an increase in glomeru-
C, The substance is freely filtered but is not excreted in the urine
because all the filtered substance is reabsorbed from the tubules
lar filtration rate (GFR) of only 10 per cent (from 180
into the blood. D, The substance is freely filtered and is not reab-
to 198 L/day) would raise urine volume 13-fold (from
sorbed but is secreted from the peritubular capillary blood into the
1.5 to 19.5 L/day) if tubular reabsorption remained
renal tubules.
constant. In reality, changes in glomerular filtration
and tubular reabsorption usually act in a coordinated
manner to produce the necessary changes in renal
excretion.
urine because all the filtered substance is reabsorbed
from the tubules back into the blood. This pattern
Why Are Large Amounts of Solutes Filtered and Then Reab-
occurs for some of the nutritional substances in the
sorbed by the Kidneys? One might question the wisdom
blood, such as amino acids and glucose, allowing them
of filtering such large amounts of water and solutes
to be conserved in the body fluids.
and then reabsorbing most of these substances. One
The substance in panel D is freely filtered at the
advantage of a high GFR is that it allows the kidneys
glomerular capillaries and is not reabsorbed, but addi-
to rapidly remove waste products from the body that
tional quantities of this substance are secreted from
depend primarily on glomerular filtration for their
the peritubular capillary blood into the renal tubules.
excretion. Most waste products are poorly reabsorbed
This pattern often occurs for organic acids and bases,
by the tubules and, therefore, depend on a high GFR
permitting them to be rapidly cleared from the blood
for effective removal from the body.
and excreted in large amounts in the urine. The excre-
A second advantage of a high GFR is that it allows
tion rate in this case is calculated as filtration rate plus
all the body fluids to be filtered and processed by the
tubular secretion rate.
kidney many times each day. Because the entire
For each substance in the plasma, a particular com-
plasma volume is only about 3 liters, whereas the GFR
bination of filtration, reabsorption, and secretion
is about 180 L/day, the entire plasma can be filtered
occurs. The rate at which the substance is excreted in
and processed about 60 times each day. This high GFR
the urine depends on the relative rates of these three
allows the kidneys to precisely and rapidly control the
basic renal processes.
volume and composition of the body fluids.
316
Unit V The Body Fluids and Kidneys
Glomerular Filtration—The
Proximal tubule
First Step in Urine Formation
Podocytes
Composition of the Glomerular
Filtrate
Capillary loops
Urine formation begins with filtration of large
amounts of fluid through the glomerular capillaries
Bowman's space
Afferent arteriole
into Bowman’s capsule. Like most capillaries, the
glomerular capillaries are relatively impermeable to
Bowman's capsule
Efferent arteriole
proteins, so that the filtered fluid (called the glomeru-
lar filtrate) is essentially protein-free and devoid of
A
Slit pores
cellular elements, including red blood cells.
The concentrations of other constituents of the
glomerular filtrate, including most salts and organic
molecules, are similar to the concentrations in the
plasma. Exceptions to this generalization include a few
Epithelium
low-molecular-weight substances, such as calcium and
fatty acids, that are not freely filtered because they are
Basement
partially bound to the plasma proteins. Almost one
membrane
half of the plasma calcium and most of the plasma fatty
acids are bound to proteins, and these bound portions
Endothelium
are not filtered through the glomerular capillaries.
B
Fenestrations
GFR Is About 20 Per Cent of the
Figure 26-10
Renal Plasma Flow
A, Basic ultrastructure of the glomerular capillaries. B, Cross
section of the glomerular capillary membrane and its major com-
As in other capillaries, the GFR is determined by (1)
ponents: capillary endothelium, basement membrane, and epithe-
the balance of hydrostatic and colloid osmotic forces
lium (podocytes).
acting across the capillary membrane and (2) the cap-
illary filtration coefficient (Kf), the product of the per-
meability and filtering surface area of the capillaries.
The high filtration rate across the glomerular capil-
The glomerular capillaries have a much higher rate of
lary membrane is due partly to its special charac-
filtration than most other capillaries because of a high
teristics. The capillary endothelium is perforated by
glomerular hydrostatic pressure and a large Kf. In the
thousands of small holes called fenestrae, similar to the
average adult human, the GFR is about 125 ml/min, or
fenestrated capillaries found in the liver. Although the
180 L/day. The fraction of the renal plasma flow that is
fenestrations are relatively large, endothelial cells are
filtered (the filtration fraction) averages about 0.2; this
richly endowed with fixed negative charges that hinder
means that about 20 per cent of the plasma flowing
the passage of plasma proteins.
through the kidney is filtered through the glomerular
Surrounding the endothelium is the basement mem-
capillaries. The filtration fraction is calculated as
brane, which consists of a meshwork of collagen and
follows:
proteoglycan fibrillae that have large spaces through
Filtration fraction = GFR/Renal plasma flow
which large amounts of water and small solutes can
filter. The basement membrane effectively prevents
filtration of plasma proteins, in part because of
Glomerular Capillary Membrane
strong negative electrical charges associated with the
proteoglycans.
The glomerular capillary membrane is similar to that
The final part of the glomerular membrane is a layer
of other capillaries, except that it has three (instead
of epithelial cells that line the outer surface of the
of the usual two) major layers: (1) the endothelium of
glomerulus. These cells are not continuous but have
the capillary, (2) a basement membrane, and (3) a layer
long footlike processes (podocytes) that encircle the
of epithelial cells (podocytes) surrounding the outer
outer surface of the capillaries (see Figure 26-10).
surface of the capillary basement membrane (Figure
The foot processes are separated by gaps called
26-10). Together, these layers make up the filtration
slit pores through which the glomerular filtrate moves.
barrier, which, despite the three layers, filters several
The epithelial cells, which also have negative charges,
hundred times as much water and solutes as the usual
provide additional restriction to filtration of plasma
capillary membrane. Even with this high rate of filtra-
proteins. Thus, all layers of the glomerular capillary
tion, the glomerular capillary membrane normally pre-
wall provide a barrier to filtration of plasma
vents filtration of plasma proteins.
proteins.
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
317
Table 26-1
Filterability of Substances by Glomerular Capillaries Based
1.0
Polycationic dextran
on Molecular Weight
Neutral dextran
Polyanionic dextran
0.8
Substance
Molecular Weight
Filterability
Water
18
1.0
0.6
Sodium
23
1.0
Glucose
180
1.0
Inulin
5,500
1.0
0.4
Myoglobin
17,000
0.75
Albumin
69,000
0.005
0.2
Filterability of Solutes Is Inversely Related to Their
0
Size. The glomerular capillary membrane is thicker
18
22
26
30
34
38
42
than most other capillaries, but it is also much more
Effective molecular radius (A)
porous and therefore filters fluid at a high rate. Despite
the high filtration rate, the glomerular filtration barrier
is selective in determining which molecules will filter,
Figure 26-11
based on their size and electrical charge.
Table 26-1 lists the effect of molecular size on fil-
Effect of size and electrical charge of dextran on its filterability by
the glomerular capillaries. A value of 1.0 indicates that the sub-
terability of different molecules. A filterability of 1.0
stance is filtered as freely as water, whereas a value of 0 indicates
means that the substance is filtered as freely as water;
that it is not filtered. Dextrans are polysaccharides that can be
a filterability of 0.75 means that the substance is fil-
manufactured as neutral molecules or with negative or positive
tered only 75 per cent as rapidly as water. Note that
charges and with varying molecular weights.
electrolytes such as sodium and small organic com-
pounds such as glucose are freely filtered. As the
molecular weight of the molecule approaches that of
albumin, the filterability rapidly decreases, approach-
in the urine, a condition known as proteinuria or
ing zero.
albuminuria.
Negatively Charged Large Molecules Are Filtered Less Easily
Than Positively Charged Molecules of Equal Molecular Size.
Determinants of the GFR
The molecular diameter of the plasma protein albumin
is only about 6 nanometers, whereas the pores of
The GFR is determined by (1) the sum of the hydro-
the glomerular membrane are thought to be about
static and colloid osmotic forces across the glomerular
8 nanometers (80 angstroms). Albumin is restricted
membrane, which gives the net filtration pressure, and
from filtration, however, because of its negative charge
(2) the glomerular capillary filtration coefficient,
and the electrostatic repulsion exerted by negative
Kf. Expressed mathematically, the GFR equals the
charges of the glomerular capillary wall proteoglycans.
product of Kf and the net filtration pressure:
Figure 26-11 shows how electrical charge affects the
filtration of different molecular weight dextrans by the
GFR = Kf ¥ Net filtration pressure
glomerulus. Dextrans are polysaccharides that can be
The net filtration pressure represents the sum of the
manufactured as neutral molecules or with negative or
hydrostatic and colloid osmotic forces that either favor
positive charges. Note that for any given molecular
or oppose filtration across the glomerular capillaries
radius, positively charged molecules are filtered much
(Figure 26-12). These forces include (1) hydrostatic
more readily than negatively charged molecules.
pressure inside the glomerular capillaries (glomerular
Neutral dextrans are also filtered more readily than
hydrostatic pressure, PG), which promotes filtration;
negatively charged dextrans of equal molecular
(2) the hydrostatic pressure in Bowman’s capsule (PB)
weight. The reason for these differences in filterability
outside the capillaries, which opposes filtration; (3) the
is that the negative charges of the basement mem-
colloid osmotic pressure of the glomerular capillary
brane and the podocytes provide an important means
plasma proteins (pG), which opposes filtration; and
for restricting large negatively charged molecules,
(4) the colloid osmotic pressure of the proteins in
including the plasma proteins.
Bowman’s capsule (pB), which promotes filtration.
In certain kidney diseases, the negative charges on
(Under normal conditions, the concentration of
the basement membrane are lost even before there are
protein in the glomerular filtrate is so low that the
noticeable changes in kidney histology, a condition
colloid osmotic pressure of the Bowman’s capsule fluid
referred to as minimal change nephropathy. As a result
is considered to be zero.)
of this loss of negative charges on the basement
The GFR can therefore be expressed as
membranes, some of the lower-molecular-weight
proteins, especially albumin, are filtered and appear
GFR = Kf ¥ (PG - PB - pG + pB)
318
Unit V The Body Fluids and Kidneys
4.2 ml/min/mm Hg, a value about 400 times as high as
Afferent
Efferent
the Kf of most other capillary systems of the body; the
arteriole
Glomerular
Glomerular
arteriole
average Kf of many other tissues in the body is only
hydrostatic
colloid osmotic
about 0.01 ml/min/mm Hg per 100 grams. This high Kf
pressure
pressure
for the glomerular capillaries contributes tremen-
(60 mm Hg)
(32 mm Hg)
dously to their rapid rate of fluid filtration.
Although increased Kf raises GFR and decreased Kf
reduces GFR, changes in Kf probably do not provide
a primary mechanism for the normal day-to-day regu-
lation of GFR. Some diseases, however, lower Kf by
Bowman's
reducing the number of functional glomerular capil-
capsule
laries (thereby reducing the surface area for filtration)
pressure
or by increasing the thickness of the glomerular
(18 mm Hg)
capillary membrane and reducing its hydraulic con-
ductivity. For example, chronic, uncontrolled hyper-
tension and diabetes mellitus gradually reduce Kf by
Glomerular
Bowman's
Glomerular
Net filtration
increasing the thickness of the glomerular capillary
hydrostatic
capsule
pressure
=
-
- oncotic
basement membrane and, eventually, by damaging the
pressure
pressure
pressure
(10 mm Hg)
(60 mm Hg)
(18 mm Hg)
(32 mm Hg)
capillaries so severely that there is loss of capillary
function.
Figure 26-12
Summary of forces causing filtration by the glomerular capillaries.
The values shown are estimates for healthy humans.
Increased Bowman’s Capsule
Hydrostatic Pressure Decreases GFR
Although the normal values for the determinants of
Direct measurements, using micropipettes, of hydro-
GFR have not been measured directly in humans, they
static pressure in Bowman’s capsule and at different
have been estimated in animals such as dogs and rats.
points in the proximal tubule suggest that a reasonable
Based on the results in animals, the approximate
estimate for Bowman’s capsule pressure in humans is
normal forces favoring and opposing glomerular fil-
about 18 mm Hg under normal conditions. Increasing
tration in humans are believed to be as follows (see
the hydrostatic pressure in Bowman’s capsule reduces
Figure 26-12):
GFR, whereas decreasing this pressure raises GFR.
However, changes in Bowman’s capsule pressure nor-
Forces Favoring Filtration (mm Hg)
mally do not serve as a primary means for regulating
Glomerular hydrostatic pressure
60
GFR.
Bowman’s capsule colloid osmotic pressure
0
In certain pathological states associated with
obstruction of the urinary tract, Bowman’s capsule
Forces Opposing Filtration (mm Hg)
pressure can increase markedly, causing serious reduc-
Bowman’s capsule hydrostatic pressure
18
tion of GFR. For example, precipitation of calcium or
Glomerular capillary colloid osmotic pressure
32
of uric acid may lead to “stones” that lodge in the
Net filtration pressure = 60 - 18 - 32 = +10 mm Hg
urinary tract, often in the ureter, thereby obstructing
outflow of the urinary tract and raising Bowman’s
Some of these values can change markedly under
capsule pressure. This reduces GFR and eventually
different physiologic conditions, whereas others are
can damage or even destroy the kidney unless the
altered mainly in disease states, as discussed later.
obstruction is relieved.
Increased Glomerular Capillary
Increased Glomerular Capillary
Filtration Coefficient Increases GFR
Colloid Osmotic Pressure
The Kf is a measure of the product of the hydraulic
Decreases GFR
conductivity and surface area of the glomerular capil-
laries. The Kf cannot be measured directly, but it is
As blood passes from the afferent arteriole through
estimated experimentally by dividing the rate of
the glomerular capillaries to the efferent arterioles,
glomerular filtration by net filtration pressure:
the plasma protein concentration increases about
20 per cent (Figure 26-13). The reason for this is that
Kf = GFR/Net filtration pressure
about one fifth of the fluid in the capillaries filters into
Because total GFR for both kidneys is about 125 ml/
Bowman’s capsule, thereby concentrating the
min and the net filtration pressure is 10 mm Hg, the
glomerular plasma proteins that are not filtered.
normal Kf is calculated to be about 12.5 ml/min/mm
Assuming that the normal colloid osmotic pressure
Hg of filtration pressure. When Kf is expressed per
of plasma entering the glomerular capillaries is
100 grams of kidney weight, it averages about
28 mm Hg, this value usually rises to about 36 mm Hg
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
319
Glomerular
40
150
2000
Filtration
filtration
38
fraction
rate
Normal
36
100
1400
Normal
34
32
60
800
30
Filtration
fraction
Renal blood
28
flow
0
200
0
1
2
3
4
Afferent
Efferent
Efferent arteriolar resistance
end
Distance along
end
(X normal)
glomerular capillary
250
2000
Figure 26-13
100
Increase in colloid osmotic pressure in plasma flowing through the
1400
glomerular capillary. Normally, about one fifth of the fluid in the
150
Normal
glomerular capillaries filters into Bowman’s capsule, thereby con-
centrating the plasma proteins that are not filtered. Increases in
100
Renal blood
800
the filtration fraction (glomerular filtration rate/renal plasma flow)
Glomerular
flow
increase the rate at which the plasma colloid osmotic pressure
50
filtration
rises along the glomerular capillary; decreases in the filtration
rate
fraction have the opposite effect.
0
200
0
1
2
3
4
Afferent arteriolar resistance
(X normal)
by the time the blood reaches the efferent end of the
capillaries. Therefore, the average colloid osmotic
pressure of the glomerular capillary plasma proteins
is midway between 28 and 36 mm Hg, or about
Figure 26-14
32 mm Hg.
Effect of change in afferent arteriolar resistance or efferent arteri-
Thus, two factors that influence the glomerular cap-
olar resistance on glomerular filtration rate and renal blood flow.
illary colloid osmotic pressure are
(1) the arterial
plasma colloid osmotic pressure and (2) the fraction
of plasma filtered by the glomerular capillaries (filtra-
tion fraction). Increasing the arterial plasma colloid
osmotic pressure raises the glomerular capillary
Increased Glomerular Capillary
colloid osmotic pressure, which in turn decreases
Hydrostatic Pressure Increases GFR
GFR.
Increasing the filtration fraction also concentrates the
The glomerular capillary hydrostatic pressure has
plasma proteins and raises the glomerular colloid
been estimated to be about 60 mm Hg under normal
osmotic pressure (see Figure 26-13). Because the fil-
conditions. Changes in glomerular hydrostatic pres-
tration fraction is defined as GFR/renal plasma flow,
sure serve as the primary means for physiologic regu-
the filtration fraction can be increased either by raising
lation of GFR. Increases in glomerular hydrostatic
GFR or by reducing renal plasma flow. For example, a
pressure raise GFR, whereas decreases in glomerular
reduction in renal plasma flow with no initial change
hydrostatic pressure reduce GFR.
in GFR would tend to increase the filtration fraction,
Glomerular hydrostatic pressure is determined by
which would raise the glomerular capillary colloid
three variables, each of which is under physiologic
osmotic pressure and tend to reduce GFR. For this
control: (1) arterial pressure,
(2) afferent arteriolar
reason, changes in renal blood flow can influence GFR
resistance, and (3) efferent arteriolar resistance.
independently of changes in glomerular hydrostatic
Increased arterial pressure tends to raise glomeru-
pressure.
lar hydrostatic pressure and, therefore, to increase
With increasing renal blood flow, a lower fraction of
GFR. (However, as discussed later, this effect is
the plasma is initially filtered out of the glomerular
buffered by autoregulatory mechanisms that maintain
capillaries, causing a slower rise in the glomerular
a relatively constant glomerular pressure as blood
capillary colloid osmotic pressure and less inhibitory
pressure fluctuates.)
effect on GFR. Consequently, even with a constant
Increased resistance of afferent arterioles reduces
glomerular hydrostatic pressure, a greater rate of blood
glomerular hydrostatic pressure and decreases GFR.
flow into the glomerulus tends to increase GFR, and a
Conversely, dilation of the afferent arterioles increases
lower rate of blood flow into the glomerulus tends to
both glomerular hydrostatic pressure and GFR
decrease GFR.
(Figure 26-14).
320
Unit V The Body Fluids and Kidneys
Constriction of the efferent arterioles increases the
Renal Blood Flow
resistance to outflow from the glomerular capillaries.
This raises the glomerular hydrostatic pressure, and as
In an average 70-kilogram man, the combined blood
long as the increase in efferent resistance does not
flow through both kidneys is about 1100 ml/min, or
reduce renal blood flow too much, GFR increases
about 22 per cent of the cardiac output. Considering
slightly (see Figure 26-14). However, because efferent
the fact that the two kidneys constitute only about 0.4
arteriolar constriction also reduces renal blood flow,
per cent of the total body weight, one can readily see
the filtration fraction and glomerular colloid osmotic
that they receive an extremely high blood flow com-
pressure increase as efferent arteriolar resistance
pared with other organs.
increases. Therefore, if the constriction of efferent
As with other tissues, blood flow supplies the
arterioles is severe
(more than about a threefold
kidneys with nutrients and removes waste products.
increase in efferent arteriolar resistance), the rise in
However, the high flow to the kidneys greatly exceeds
colloid osmotic pressure exceeds the increase in
this need. The purpose of this additional flow is to
glomerular capillary hydrostatic pressure caused by
supply enough plasma for the high rates of glomeru-
efferent arteriolar constriction. When this occurs, the
lar filtration that are necessary for precise regulation
net force for filtration actually decreases, causing a
of body fluid volumes and solute concentrations. As
reduction in GFR.
might be expected, the mechanisms that regulate renal
Thus, efferent arteriolar constriction has a biphasic
blood flow are closely linked to the control of GFR
effect on GFR. At moderate levels of constriction,
and the excretory functions of the kidneys.
there is a slight increase in GFR, but with severe con-
striction, there is a decrease in GFR. The primary
cause of the eventual decrease in GFR is as follows:
Renal Blood Flow and Oxygen
As efferent constriction becomes severe and as plasma
Consumption
protein concentration increases, there is a rapid, non-
linear increase in colloid osmotic pressure caused by
On a per gram weight basis, the kidneys normally
the Donnan effect; the higher the protein concentra-
consume oxygen at twice the rate of the brain but have
tion, the more rapidly the colloid osmotic pressure
almost seven times the blood flow of the brain. Thus,
rises because of the interaction of ions bound to the
the oxygen delivered to the kidneys far exceeds their
plasma proteins, which also exert an osmotic effect, as
metabolic needs, and the arterial-venous extraction of
discussed in Chapter 16.
oxygen is relatively low compared with that of most
To summarize, constriction of afferent arterioles
other tissues.
reduces GFR. However, the effect of efferent arterio-
A large fraction of the oxygen consumed by the
lar constriction depends on the severity of the con-
kidneys is related to the high rate of active sodium
striction; modest efferent constriction raises GFR, but
reabsorption by the renal tubules. If renal blood flow
severe efferent constriction (more than a threefold
and GFR are reduced and less sodium is filtered, less
increase in resistance) tends to reduce GFR.
sodium is reabsorbed and less oxygen is consumed.
Table 26-2 summarizes the factors that can decrease
Therefore, renal oxygen consumption varies in pro-
GFR.
portion to renal tubular sodium reabsorption, which in
turn is closely related to GFR and the rate of sodium
filtered (Figure 26-15). If glomerular filtration com-
Table 26-2
pletely ceases, renal sodium reabsorption also ceases,
Factors That Can Decrease the Glomerular Filtration Rate
and oxygen consumption decreases to about one
(GFR)
fourth normal. This residual oxygen consumption
reflects the basic metabolic needs of the renal cells.
Physical Determinants* Physiologic/Pathophysiologic Causes
ØKf
Æ Ø GFR
Renal disease, diabetes mellitus,
Determinants of Renal Blood Flow
hypertension
≠ PB Æ Ø GFR
Urinary tract obstruction (e.g., kidney
stones)
Renal blood flow is determined by the pressure gradi-
≠ pG Æ Ø GFR
Ø Renal blood flow, increased plasma
ent across the renal vasculature
(the difference
proteins
between renal artery and renal vein hydrostatic pres-
Ø PG Æ Ø GFR
sures), divided by the total renal vascular resistance:
ØAP ÆØPG
Ø Arterial pressure (has only small effect
due to autoregulation)
(Renal artery pressure Renal vein pressure)
ØRE ÆØPG
Ø Angiotensin II (drugs that block
angiotensin II formation)
Total renal vascular resistance
≠RA ÆØPG
≠ Sympathetic activity, vasoconstrictor
Renal artery pressure is about equal to systemic
hormones (e.g., norepinephrine,
endothelin)
arterial pressure, and renal vein pressure averages
about 3 to 4 mm Hg under most conditions. As in other
* Opposite changes in the determinants usually increase GFR.
vascular beds, the total vascular resistance through the
Kf, glomerular filtration coefficient; PB, Bowman’s capsule hydrostatic pres-
kidneys is determined by the sum of the resistances in
sure; pG, glomerular capillary colloid osmotic pressure; PG, glomerular capil-
the individual vasculature segments, including the
lary hydrostatic pressure; AP, systemic arterial pressure; RE, efferent arteriolar
resistance; RA, afferent arteriolar resistance.
arteries, arterioles, capillaries, and veins (Table 26-3).
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
321
Table 26-3
Approximate Pressures and Vascular Resistances in the Circulation of a Normal Kidney
Pressure in Vessel (mm Hg)
Vessel
Beginning
End
Per Cent of Total Renal Vascular Resistance
Renal artery
100
100
~0
Interlobar, arcuate, and interlobular arteries
~100
85
~16
Afferent arteriole
85
60
~26
Glomerular capillaries
60
59
~1
Efferent arteriole
59
18
~43
Peritubular capillaries
18
8
~10
Interlobar, interlobular, and arcuate veins
8
4
~4
Renal vein
4
~4
~0
range between 80 and 170 mm Hg, a process called
autoregulation. This capacity for autoregulation occurs
3.0
through mechanisms that are completely intrinsic to
the kidneys, as discussed later in this chapter.
2.5
Blood Flow in the Vasa Recta of the
Renal Medulla Is Very Low Compared
2.0
with Flow in the Renal Cortex
1.5
The outer part of the kidney, the renal cortex, receives
most of the kidney’s blood flow. Blood flow in the renal
medulla accounts for only 1 to 2 per cent of the total
1.0
renal blood flow. Flow to the renal medulla is supplied
by a specialized portion of the peritubular capillary
0.5
system called the vasa recta. These vessels descend into
Basal oxygen consumption
the medulla in parallel with the loops of Henle and
then loop back along with the loops of Henle and
0
0
5
10
15
20
return to the cortex before emptying into the venous
Sodium reabsorption
system. As discussed in Chapter 28, the vasa recta play
(mEq/min per 100 g kidney weight)
an important role in allowing the kidneys to form a
concentrated urine.
Figure 26-15
Physiologic Control of
Glomerular Filtration and
Relationship between oxygen consumption and sodium reab-
Renal Blood Flow
sorption in dog kidneys. (Kramer K, Deetjen P: Relation of renal
oxygen consumption to blood supply and glomerular filtration
during variations of blood pressure. Pflugers Arch Physiol
The determinants of GFR that are most variable and
271:782, 1960.)
subject to physiologic control include the glomerular
hydrostatic pressure and the glomerular capillary
Most of the renal vascular resistance resides in three
colloid osmotic pressure. These variables, in turn, are
major segments: interlobular arteries, afferent arteri-
influenced by the sympathetic nervous system, hor-
oles, and efferent arterioles. Resistance of these vessels
mones and autacoids (vasoactive substances that are
is controlled by the sympathetic nervous system,
released in the kidneys and act locally), and other
various hormones, and local internal renal control
feedback controls that are intrinsic to the kidneys.
mechanisms, as discussed later. An increase in the
resistance of any of the vascular segments of the
kidneys tends to reduce the renal blood flow, whereas
Sympathetic Nervous System
a decrease in vascular resistance increases renal blood
Activation Decreases GFR
flow if renal artery and renal vein pressures remain
constant.
Essentially all the blood vessels of the kidneys, includ-
Although changes in arterial pressure have some
ing the afferent and the efferent arterioles, are richly
influence on renal blood flow, the kidneys have effec-
innervated by sympathetic nerve fibers. Strong activa-
tive mechanisms for maintaining renal blood flow and
tion of the renal sympathetic nerves can constrict the
GFR relatively constant over an arterial pressure
renal arterioles and decrease renal blood flow and
322
Unit V The Body Fluids and Kidneys
GFR. Moderate or mild sympathetic stimulation has
Angiotensin II Constricts Efferent Arterioles. A powerful
little influence on renal blood flow and GFR. For
renal vasoconstrictor, angiotensin II, can be consid-
example, reflex activation of the sympathetic nervous
ered a circulating hormone as well as a locally pro-
system resulting from moderate decreases in pressure
duced autacoid because it is formed in the kidneys
at the carotid sinus baroreceptors or cardiopulmonary
as well as in the systemic circulation. Because
receptors has little influence on renal blood flow or
angiotensin II preferentially constricts efferent arteri-
GFR.
oles, increased angiotensin II levels raise glomerular
The renal sympathetic nerves seem to be most
hydrostatic pressure while reducing renal blood flow.
important in reducing GFR during severe, acute dis-
It should be kept in mind that increased angiotensin
turbances lasting for a few minutes to a few hours,
II formation usually occurs in circumstances associ-
such as those elicited by the defense reaction, brain
ated with decreased arterial pressure or volume
ischemia, or severe hemorrhage. In the healthy resting
depletion, which tend to decrease GFR. In these cir-
person, sympathetic tone appears to have little influ-
cumstances, the increased level of angiotensin II, by
ence on renal blood flow.
constricting efferent arterioles, helps prevent decreases
in glomerular hydrostatic pressure and GFR; at the
same time, though, the reduction in renal blood flow
Hormonal and Autacoid Control
caused by efferent arteriolar constriction contributes
of Renal Circulation
to decreased flow through the peritubular capillaries,
which in turn increases reabsorption of sodium and
There are several hormones and autacoids that can
water, as discussed in Chapter 27.
influence GFR and renal blood flow, as summarized in
Thus, increased angiotensin II levels that occur with
Table 26-4.
a low-sodium diet or volume depletion help preserve
GFR and maintain normal excretion of metabolic
Norepinephrine, Epinephrine, and Endothelin Constrict Renal
waste products such as urea and creatinine that
Blood Vessels and Decrease GFR. Hormones that constrict
depend on glomerular filtration for their excretion; at
afferent and efferent arterioles, causing reductions in
the same time, the angiotensin II-induced constriction
GFR and renal blood flow, include norepinephrine and
of efferent arterioles increases tubular reabsorption of
epinephrine released from the adrenal medulla. In
sodium and water, which helps restore blood volume
general, blood levels of these hormones parallel the
and blood pressure. This effect of angiotensin II in
activity of the sympathetic nervous system; thus, nor-
helping to “autoregulate” GFR is discussed in more
epinephrine and epinephrine have little influence on
detail later in this chapter.
renal hemodynamics except under extreme conditions,
such as severe hemorrhage.
Endothelial-Derived Nitric Oxide Decreases Renal Vascular
Another vasoconstrictor, endothelin, is a peptide
Resistance and Increases GFR. An autacoid that de-
that can be released by damaged vascular endothelial
creases renal vascular resistance and is released by the
cells of the kidneys as well as by other tissues. The
vascular endothelium throughout the body is endothe-
physiologic role of this autacoid is not completely
lial-derived nitric oxide. A basal level of nitric oxide
understood. However, endothelin may contribute to
production appears to be important for maintaining
hemostasis
(minimizing blood loss) when a blood
vasodilation of the kidneys. This allows the kidneys to
vessel is severed, which damages the endothelium
excrete normal amounts of sodium and water. There-
and releases this powerful vasoconstrictor. Plasma
fore, administration of drugs that inhibit this normal
endothelin levels also are increased in certain disease
formation of nitric oxide increases renal vascular
states associated with vascular injury, such as toxemia
resistance and decreases GFR and urinary sodium
of pregnancy, acute renal failure, and chronic uremia,
excretion, eventually causing high blood pressure. In
and may contribute to renal vasoconstriction and
some hypertensive patients, impaired nitric oxide pro-
decreased GFR in some of these pathophysiologic
duction could be the cause of increased renal vaso-
conditions.
constriction and increased blood pressure.
Prostaglandins and Bradykinin Tend to Increase GFR. Hor-
mones and autacoids that cause vasodilation and
Table 26-4
increased renal blood flow and GFR include the
prostaglandins
(PGE2 and PGI2) and bradykinin.
Hormones and Autacoids That Influence Glomerular
These substances are discussed in Chapter
17.
Filtration Rate (GFR)
Although these vasodilators do not appear to be of
major importance in regulating renal blood flow or
Hormone or Autacoid
Effect on GFR
GFR in normal conditions, they may dampen the renal
Norepinephrine
Ø
vasoconstrictor effects of the sympathetic nerves or
Epinephrine
Ø
angiotensin II, especially their effects to constrict the
Endothelin
Ø
afferent arterioles.
Angiotensin II
¨Æ (prevents Ø)
By opposing vasoconstriction of afferent arterioles,
Endothelial-derived nitric oxide
≠
Prostaglandins
≠
the prostaglandins may help prevent excessive reduc-
tions in GFR and renal blood flow. Under stressful
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
323
conditions, such as volume depletion or after surgery,
arterial pressure to as low as 75 mm Hg or an increase
the administration of nonsteroidal anti-inflammatory
to as high as 160 mm Hg changes GFR only a few
agents, such as aspirin, that inhibit prostaglandin syn-
percentage points. In general, renal blood flow is auto-
thesis may cause significant reductions in GFR.
regulated in parallel with GFR, but GFR is more effi-
ciently autoregulated under certain conditions.
Autoregulation of GFR and
Renal Blood Flow
Importance of GFR Autoregulation
in Preventing Extreme Changes in
Feedback mechanisms intrinsic to the kidneys nor-
Renal Excretion
mally keep the renal blood flow and GFR relatively
constant, despite marked changes in arterial blood
The autoregulatory mechanisms of the kidney are not
pressure. These mechanisms still function in blood-
100 per cent perfect, but they do prevent potentially
perfused kidneys that have been removed from the
large changes in GFR and renal excretion of water and
body, independent of systemic influences. This relative
solutes that would otherwise occur with changes in
constancy of GFR and renal blood flow is referred to
blood pressure. One can understand the quantitative
as autoregulation (Figure 26-16).
importance of autoregulation by considering the rela-
The primary function of blood flow autoregulation
tive magnitudes of glomerular filtration, tubular reab-
in most tissues other than the kidneys is to maintain
sorption, and renal excretion and the changes in renal
the delivery of oxygen and nutrients at a normal level
excretion that would occur without autoregulatory
and to remove the waste products of metabolism,
mechanisms.
despite changes in the arterial pressure. In the kidneys,
Normally, GFR is about 180 L/day and tubular reab-
the normal blood flow is much higher than that
sorption is 178.5 L/day, leaving 1.5 L/day of fluid to be
required for these functions. The major function of
excreted in the urine. In the absence of autoregulation,
autoregulation in the kidneys is to maintain a rela-
a relatively small increase in blood pressure (from 100
tively constant GFR and to allow precise control of
to 125 mm Hg) would cause a similar 25 per cent
renal excretion of water and solutes.
increase in GFR (from about 180 to 225 L/day). If
The GFR normally remains autoregulated (that is,
tubular reabsorption remained constant at 178.5 L/
remains relatively constant), despite considerable
day, this would increase the urine flow to 46.5 L/day
arterial pressure fluctuations that occur during a
(the difference between GFR and tubular reabsorp-
person’s usual activities. For instance, a decrease in
tion)—a total increase in urine of more than 30-fold.
Because the total plasma volume is only about 3 liters,
such a change would quickly deplete the blood
volume.
But in reality, such a change in arterial pressure
1600
160
exerts much less of an effect on urine volume for
two reasons: (1) renal autoregulation prevents large
1200
120
changes in GFR that would otherwise occur, and (2)
there are additional adaptive mechanisms in the renal
800
80
tubules that allow them to increase their reabsorption
Renal blood flow
rate when GFR rises, a phenomenon referred to as
400
40
Glomerular filtration
glomerulotubular balance (discussed in Chapter 27).
rate
Even with these special control mechanisms, changes
0
0
in arterial pressure still have significant effects on
8
renal excretion of water and sodium; this is referred to
as pressure diuresis or pressure natriuresis, and it is
6
crucial in the regulation of body fluid volumes and
arterial pressure, as discussed in Chapters 19 and 29.
4
2
Role of Tubuloglomerular Feedback
0
in Autoregulation of GFR
50
100
150
200
Arterial pressure
To perform the function of autoregulation, the kidneys
(mm Hg)
have a feedback mechanism that links changes in
sodium chloride concentration at the macula densa
with the control of renal arteriolar resistance. This
Figure 26-16
feedback helps ensure a relatively constant delivery of
sodium chloride to the distal tubule and helps prevent
Autoregulation of renal blood flow and glomerular filtration rate but
lack of autoregulation of urine flow during changes in renal arte-
spurious fluctuations in renal excretion that would
rial pressure.
otherwise occur. In many circumstances, this feedback
324
Unit V The Body Fluids and Kidneys
autoregulates renal blood flow and GFR in parallel.
decreased GFR slows the flow rate in the loop of
However, because this mechanism is specifically
Henle, causing increased reabsorption of sodium and
directed toward stabilizing sodium chloride delivery to
chloride ions in the ascending loop of Henle, thereby
the distal tubule, there are instances when GFR is
reducing the concentration of sodium chloride at the
autoregulated at the expense of changes in renal blood
macula densa cells. This decrease in sodium chloride
flow, as discussed later.
concentration initiates a signal from the macula densa
The tubuloglomerular feedback mechanism has
that has two effects (Figure 26-18): (1) it decreases
two components that act together to control GFR:
resistance to blood flow in the afferent arterioles,
(1) an afferent arteriolar feedback mechanism and (2)
which raises glomerular hydrostatic pressure and helps
an efferent arteriolar feedback mechanism. These
return GFR toward normal, and (2) it increases renin
feedback mechanisms depend on special anatomical
release from the juxtaglomerular cells of the afferent
arrangements of the juxtaglomerular complex (Figure
and efferent arterioles, which are the major storage
26-17).
sites for renin. Renin released from these cells then
The juxtaglomerular complex consists of macula
functions as an enzyme to increase the formation of
densa cells in the initial portion of the distal tubule and
angiotensin I, which is converted to angiotensin II.
juxtaglomerular cells in the walls of the afferent and
Finally, the angiotensin II constricts the efferent arte-
efferent arterioles. The macula densa is a specialized
rioles, thereby increasing glomerular hydrostatic pres-
group of epithelial cells in the distal tubules that comes
sure and returning GFR toward normal.
in close contact with the afferent and efferent arteri-
These two components of the tubuloglomerular
oles. The macula densa cells contain Golgi apparatus,
feedback mechanism, operating together by way of the
which are intracellular secretory organelles directed
special anatomical structure of the juxtaglomerular
toward the arterioles, suggesting that these cells may
apparatus, provide feedback signals to both the affer-
be secreting a substance toward the arterioles.
ent and the efferent arterioles for efficient autoregu-
lation of GFR during changes in arterial pressure.
Decreased Macula Densa Sodium Chloride Causes Dilation of
When both of these mechanisms are functioning
Afferent Arterioles and Increased Renin Release. The
together, the GFR changes only a few percentage
macula densa cells sense changes in volume delivery
points, even with large fluctuations in arterial pressure
to the distal tubule by way of signals that are not com-
between the limits of 75 and 160 mm Hg.
pletely understood. Experimental studies suggest that
Arterial pressure
-
-
Glomerular hydrostatic
pressure
Glomerular
epithelium
GFR
Proximal
NaCl
Macula densa
reabsorption
NaCl
Juxtaglomerular
cells
Afferent
Renin
Efferent
arteriole
arteriole
Angiotensin II
Internal
Macula densa
elastic
lamina
Efferent
Afferent
arteriolar
arteriolar
Smooth
Basement
resistance
resistance
muscle
membrane
Distal
fiber
tubule
Figure 26-18
Figure 26-17
Macula densa feedback mechanism for autoregulation
of
Structure of the juxtaglomerular apparatus, demonstrating its pos-
glomerular hydrostatic pressure and glomerular filtration rate
sible feedback role in the control of nephron function.
(GFR) during decreased renal arterial pressure.
Chapter 26
Urine Formation by the Kidneys: I. Glomerular Filtration, Renal Blood Flow, and Their Control
325
Blockade of Angiotensin II Formation Further Reduces GFR During
The exact mechanisms by which this occurs are still
Renal Hypoperfusion. As discussed earlier, a preferential
not completely understood, but one possible explana-
constrictor action of angiotensin II on efferent arteri-
tion is the following: A high-protein meal increases the
oles helps prevent serious reductions in glomerular
release of amino acids into the blood, which are reab-
hydrostatic pressure and GFR when renal perfusion
sorbed in the proximal tubule. Because amino acids
pressure falls below normal. The administration of
and sodium are reabsorbed together by the proximal
drugs that block the formation of angiotensin II
tubules, increased amino acid reabsorption also stimu-
(angiotensin-converting enzyme inhibitors) or that
lates sodium reabsorption in the proximal tubules. This
block the action of angiotensin II (angiotensin II antag-
decreases sodium delivery to the macula densa, which
onists) causes greater reductions in GFR than usual
elicits a tubuloglomerular feedback-mediated decrease
when the renal arterial pressure falls below normal.
in resistance of the afferent arterioles, as discussed
Therefore, an important complication of using these
earlier. The decreased afferent arteriolar resistance then
drugs to treat patients who have hypertension because
raises renal blood flow and GFR. This increased GFR
of renal artery stenosis (partial blockage of the renal
allows sodium excretion to be maintained at a nearly
artery) is a severe decrease in GFR that can, in
normal level while increasing the excretion of the waste
some cases, cause acute renal failure. Nevertheless,
products of protein metabolism, such as urea.
angiotensin II-blocking drugs can be useful therapeutic
A similar mechanism may also explain the marked
agents in many patients with hypertension, congestive
increases in renal blood flow and GFR that occur with
heart failure, and other conditions, as long as they are
large increases in blood glucose levels in uncontrolled
monitored to ensure that severe decreases in GFR do
diabetes mellitus. Because glucose, like some of the
not occur.
amino acids, is also reabsorbed along with sodium in
the proximal tubule, increased glucose delivery to the
tubules causes them to reabsorb excess sodium along
with glucose. This, in turn, decreases delivery of sodium
Myogenic Autoregulation of Renal
chloride to the macula densa, activating a tubu-
Blood Flow and GFR
loglomerular feedback-mediated dilation of the affer-
ent arterioles and subsequent increases in renal blood
Another mechanism that contributes to the main-
flow and GFR.
tenance of a relatively constant renal blood flow
These examples demonstrate that renal blood flow
and GFR is the ability of individual blood vessels to
and GFR per se are not the primary variables controlled
resist stretching during increased arterial pressure, a
by the tubuloglomerular feedback mechanism. The
phenomenon referred to as the myogenic mechanism.
main purpose of this feedback is to ensure a constant
delivery of sodium chloride to the distal tubule, where
Studies of individual blood vessels (especially small
final processing of the urine takes place. Thus, distur-
arterioles) throughout the body have shown that they
bances that tend to increase reabsorption of sodium
respond to increased wall tension or wall stretch by
chloride at tubular sites before the macula densa tend
contraction of the vascular smooth muscle. Stretch
to elicit increased renal blood flow and GFR, which
of the vascular wall allows increased movement of
helps return distal sodium chloride delivery toward
calcium ions from the extracellular fluid into the cells,
normal so that normal rates of sodium and water excre-
causing them to contract through the mechanisms dis-
tion can be maintained (see Figure 26-18).
cussed in Chapter 8. This contraction prevents overdis-
An opposite sequence of events occurs when proxi-
tention of the vessel and at the same time, by raising
mal tubular reabsorption is reduced. For example, when
vascular resistance, helps prevent excessive increases
the proximal tubules are damaged (which can occur as
a result of poisoning by heavy metals, such as mercury,
in renal blood flow and GFR when arterial pressure
or large doses of drugs, such as tetracyclines), their
increases.
ability to reabsorb sodium chloride is decreased. As a
Although the myogenic mechanism probably oper-
consequence, large amounts of sodium chloride are
ates in most arterioles throughout the body, its impor-
delivered to the distal tubule and, without appropriate
tance in renal blood flow and GFR autoregulation has
compensations, would quickly cause excessive volume
been questioned by some physiologists because this
depletion. One of the important compensatory
pressure-sensitive mechanism has no means of directly
responses appears to be a tubuloglomerular feed-
detecting changes in renal blood flow or GFR per se.
back-mediated renal vasoconstriction that occurs in
response to the increased sodium chloride delivery to
the macula densa in these circumstances. These exam-
Other Factors That Increase Renal
ples again demonstrate the importance of this feedback
Blood Flow and GFR: High Protein
mechanism in ensuring that the distal tubule receives
the proper rate of delivery of sodium chloride, other
Intake and Increased Blood Glucose
tubular fluid solutes, and tubular fluid volume so that
Although renal blood flow and GFR are relatively
appropriate amounts of these substances are excreted
stable under most conditions, there are circumstances in
in the urine.
which these variables change significantly. For example,
a high protein intake is known to increase both renal
blood flow and GFR. With a chronic high-protein diet,
References
such as one that contains large amounts of meat, the
increases in GFR and renal blood flow are due partly to
Beeuwkes R III: The vascular organization of the kidney.
growth of the kidneys. However, GFR and renal blood
Annu Rev Physiol 42:531, 1980.
flow increase 20 to 30 per cent within 1 or 2 hours after
Bell PD, Lapointe JY, Peti-Peterdi J: Macula densa cell sig-
a person eats a high-protein meal.
naling. Annu Rev Physiol 65:481, 2003.
326
Unit V The Body Fluids and Kidneys
Blantz RC, Deng A, Lortie M, et al: The complex role of
Haraldsson B, Sörensson J: Why do we not all have protein-
nitric oxide in the regulation of glomerular ultrafiltration.
uria? An update of our current understanding of the
Kidney Int 61:782, 2002.
glomerular barrier. News Physiol Sci 19:7, 2004.
Cowley AW Jr, Mori T, Mattson D, Zou AP: Role of renal
Kriz W, Kaissling B: Structural organization of the mam-
NO production in the regulation of medullary blood flow.
malian kidney. In Seldin DW, Giebisch G (eds): The
Am J Physiol Regul Integr Comp Physiol 284:R1355,
Kidney—Physiology and Pathophysiology, 3rd ed. New
2003.
York: Raven Press, 2000, pp 587-654.
Davis MJ, Hill MA: Signaling mechanisms underlying
Navar LG, Kobori H, Prieto-Carrasquero M: Intrarenal
the vascular myogenic response. Physiol Rev
79:387,
angiotensin II and hypertension. Curr Hypertens Rep
1999.
5:135, 2003.
Deen WM, Lazzara MJ, Myers BD: Structural determinants
Pallone TL, Zhang Z, Rhinehart K: Physiology of the renal
of glomerular permeability. Am J Physiol Renal Physiol
medullary microcirculation. Am J Physiol Renal Physiol
281:F579, 2001.
284:F253, 2003.
DiBona GF: Neural control of the kidney: past, present, and
Roman RJ: P-450 metabolites of arachidonic acid in the
future. Hypertension 41:621, 2003.
control of cardiovascular function. Physiol Rev 82:131,
Hall JE: Angiotensin II and long-term arterial pressure reg-
2002.
ulation: the overriding dominance of the kidney. J Am Soc
Schnermann J, Levine DZ: Paracrine factors in tubu-
Nephrol 10 (Suppl 12):s258, 1999.
loglomerular feedback: adenosine, ATP, and nitric oxide.
Hall JE, Brands MW: The renin-angiotensin-aldosterone
Annu Rev Physiol 65:501, 2003.
system: renal mechanisms and circulatory homeostasis. In
Whelton A: Renal aspects of treatment with conventional
Seldin DW, Giebisch G (eds): The Kidney—Physiology
nonsteroidal anti-inflammatory drugs versus cyclooxyge-
and Pathophysiology, 3rd ed. New York: Raven Press,
nase-2-specific inhibitors. Am J Med 110 (Suppl 3A):33S,
2000, pp 1009-1046.
2001.
C H A P T E R
2
7
Urine Formation by the Kidneys:
II. Tubular Processing of the
Glomerular Filtrate
Reabsorption and Secretion
by the Renal Tubules
As the glomerular filtrate enters the renal tubules,
it flows sequentially through the successive parts of
the tubule—the proximal tubule, the loop of Henle,
the distal tubule, the collecting tubule, and, finally,
the collecting duct—before it is excreted as urine.
Along this course, some substances are selectively reabsorbed from the tubules
back into the blood, whereas others are secreted from the blood into the tubular
lumen. Eventually, the urine that is formed and all the substances in the urine
represent the sum of three basic renal processes—glomerular filtration, tubular
reabsorption, and tubular secretion—as follows:
Urinary excretion = Glomerular filtration - Tubular reabsorption
+ Tubular secretion
For many substances, reabsorption plays a much more important role than
does secretion in determining the final urinary excretion rate. However, secre-
tion accounts for significant amounts of potassium ions, hydrogen ions, and a
few other substances that appear in the urine.
Tubular Reabsorption Is Selective and Quantitatively Large
Table 27-1 shows the renal handling of several substances that are all freely fil-
tered in the kidneys and reabsorbed at variable rates.
The rate at which each of these substances is filtered is calculated as
Filtration = Glomerular filtration rate ¥ Plasma concentration
This calculation assumes that the substance is freely filtered and not bound
to plasma proteins. For example, if plasma glucose concentration is 1 g/L, the
amount of glucose filtered each day is about 180 L/day ¥ 1 g/L, or 180 g/day.
Because virtually none of the filtered glucose is normally excreted, the rate of
glucose reabsorption is also 180 g/day.
From Table 27-1, two things are immediately apparent. First, the processes of
glomerular filtration and tubular reabsorption are quantitatively very large rel-
ative to urinary excretion for many substances. This means that a small change
in glomerular filtration or tubular reabsorption can potentially cause a relatively
large change in urinary excretion. For example, a 10 per cent decrease in tubular
reabsorption, from 178.5 to 160.7 L/day, would increase urine volume from 1.5
to 19.3 L/day (almost a 13-fold increase) if the glomerular filtration rate (GFR)
remained constant. In reality, however, changes in tubular reabsorption and
glomerular filtration are closely coordinated, so that large fluctuations in urinary
excretion are avoided.
Second, unlike glomerular filtration, which is relatively nonselective (that is,
essentially all solutes in the plasma are filtered except the plasma proteins or
substances bound to them), tubular reabsorption is highly selective. Some sub-
stances, such as glucose and amino acids, are almost completely reabsorbed
327
328
Unit V The Body Fluids and Kidneys
Table 27-1
Filtration, Reabsorption, and Excretion Rates of Different Substances by the Kidneys
Amount Filtered
Amount Reabsorbed
Amount Excreted
% of Filtered Load Reabsorbed
Glucose (g/day)
180
180
0
100
Bicarbonate (mEq/day)
4,320
4,318
2
>99.9
Sodium (mEq/day)
25,560
25,410
150
99.4
Chloride (mEq/day)
19,440
19,260
180
99.1
Potassium (mEq/day)
756
664
92
87.8
Urea (g/day)
46.8
23.4
23.4
50
Creatinine (g/day)
1.8
0
1.8
0
from the tubules, so that the urinary excretion rate is
FILTRATION
essentially zero. Many of the ions in the plasma, such
Peritubular
Tubular
capillary
cells
as sodium, chloride, and bicarbonate, are also highly
Lumen
reabsorbed, but their rates of reabsorption and urinary
excretion are variable, depending on the needs of the
body. Certain waste products, such as urea and creati-
Paracellular
nine, conversely, are poorly reabsorbed from the
Bulk
path
tubules and excreted in relatively large amounts.
flow
Transcellular
Therefore, by controlling the rate at which they
path
Active
reabsorb different substances, the kidneys regulate the
ATP
excretion of solutes independently of one another, a
Blood
Passive
Solutes
capability that is essential for precise control of the
(diffusion)
composition of body fluids. In this chapter, we discuss
the mechanisms that allow the kidneys to selectively
Osmosis
H2O
reabsorb or secrete different substances at variable
rates.
REABSORPTION
EXCRETION
Tubular Reabsorption
Figure 27-1
Includes Passive and
Active Mechanisms
Reabsorption of filtered water and solutes from the tubular lumen
across the tubular epithelial cells, through the renal interstitium, and
back into the blood. Solutes are transported through the cells (trans-
For a substance to be reabsorbed, it must first be trans-
cellular route) by passive diffusion or active transport, or between
ported (1) across the tubular epithelial membranes
the cells
(paracellular route) by diffusion. Water is transported
into the renal interstitial fluid and then (2) through the
through the cells and between the tubular cells by osmosis. Trans-
port of water and solutes from the interstitial fluid into the peri-
peritubular capillary membrane back into the blood
tubular capillaries occurs by ultrafiltration (bulk flow).
(Figure 27-1). Thus, reabsorption of water and solutes
includes a series of transport steps. Reabsorption
across the tubular epithelium into the interstitial fluid
includes active or passive transport by way of the same
Active Transport
basic mechanisms discussed in Chapter 4 for transport
across other membranes of the body. For instance,
Active transport can move a solute against an elec-
water and solutes can be transported either through
trochemical gradient and requires energy derived
the cell membranes themselves (transcellular route) or
from metabolism. Transport that is coupled directly to
through the junctional spaces between the cells (para-
an energy source, such as the hydrolysis of adenosine
cellular route). Then, after absorption across the
triphosphate (ATP), is termed primary active trans-
tubular epithelial cells into the interstitial fluid, water
port. A good example of this is the sodium-potassium
and solutes are transported the rest of the way through
ATPase pump that functions throughout most parts of
the peritubular capillary walls into the blood by ultra-
the renal tubule. Transport that is coupled indirectly to
filtration (bulk flow) that is mediated by hydrostatic
an energy source, such as that due to an ion gradient,
and colloid osmotic forces. The peritubular capillaries
is referred to as secondary active transport. Reabsorp-
behave very much like the venous ends of most other
tion of glucose by the renal tubule is an example of
capillaries because there is a net reabsorptive force
secondary active transport. Although solutes can be
that moves the fluid and solutes from the interstitium
reabsorbed by active and/or passive mechanisms by
into the blood.
the tubule, water is always reabsorbed by a passive
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
329
(nonactive) physical mechanism called osmosis, which
Peritubular
Tubular
Tubular
means water diffusion from a region of low solute con-
capillary
epithelial cells
lumen
centration (high water concentration) to one of high
solute concentration (low water concentration).
+
Na
Solutes Can Be Transported Through Epithelial Cells or Between
Cells. Renal tubular cells, like other epithelial cells, are
ATP
Na+
held together by tight junctions. Lateral intercellular
Na+
spaces lie behind the tight junctions and separate the
ATP
K+
(-3 mv)
epithelial cells of the tubule. Solutes can be reabsorbed
K+
or secreted across the cells by way of the transcellular
(-70 mV)
Tight junction
pathway or between the cells by moving across the
tight junctions and intercellular spaces by way of
Brush border
Basal
(luminal
the paracellular pathway. Sodium is a substance that
channels
membrane)
moves through both routes, although most of the
sodium is transported through the transcellular
Interstitial
Basement
Intercellular space
fluid
membrane
pathway. In some nephron segments, especially the
proximal tubule, water is also reabsorbed across the
Figure 27-2
paracellular pathway, and substances dissolved in
the water, especially potassium, magnesium, and chlo-
Basic mechanism for active transport of sodium through the tubular
ride ions, are carried with the reabsorbed fluid
epithelial cell. The sodium-potassium pump transports sodium from
the interior of the cell across the basolateral membrane, creating a
between the cells.
low intracellular sodium concentration and a negative intracellular
electrical potential. The low intracellular sodium concentration and
Primary Active Transport Through the Tubular Membrane Is
the negative electrical potential cause sodium ions to diffuse from
Linked to Hydrolysis of ATP. The special importance of
the tubular lumen into the cell through the brush border.
primary active transport is that it can move solutes
against an electrochemical gradient. The energy for this
active transport comes from the hydrolysis of ATP by
way of membrane-bound ATPase; the ATPase is also
brush border on the luminal side of the membrane
a component of the carrier mechanism that binds and
(the side that faces the tubular lumen) that multiplies
moves solutes across the cell membranes. The primary
the surface area about 20-fold. There are also sodium
active transporters that are known include sodium-
carrier proteins that bind sodium ions on the luminal
potassium ATPase, hydrogen ATPase, hydrogen-
surface of the membrane and release them inside the
potassium ATPase, and calcium ATPase.
cell, providing facilitated diffusion of sodium through
A good example of a primary active transport
the membrane into the cell. These sodium carrier pro-
system is the reabsorption of sodium ions across the
teins are also important for secondary active transport
proximal tubular membrane, as shown in Figure 27-2.
of other substances, such as glucose and amino acids,
On the basolateral sides of the tubular epithelial cell,
as discussed later.
the cell membrane has an extensive sodium-potassium
Thus, the net reabsorption of sodium ions from the
ATPase system that hydrolyzes ATP and uses the
tubular lumen back into the blood involves at least
released energy to transport sodium ions out of the
three steps:
cell into the interstitium. At the same time, potassium
1. Sodium diffuses across the luminal membrane
is transported from the interstitium to the inside of the
(also called the apical membrane) into the cell
cell. The operation of this ion pump maintains low
down an electrochemical gradient established
intracellular sodium and high intracellular potassium
by the sodium-potassium ATPase pump on the
concentrations and creates a net negative charge of
basolateral side of the membrane.
about -70 millivolts within the cell. This pumping of
2. Sodium is transported across the basolateral
sodium out of the cell across the basolateral membrane
membrane against an electrochemical gradient
of the cell favors passive diffusion of sodium across the
by the sodium-potassium ATPase pump.
luminal membrane of the cell, from the tubular lumen
3. Sodium, water, and other substances are
into the cell, for two reasons: (1) There is a concen-
reabsorbed from the interstitial fluid into the
tration gradient favoring sodium diffusion into the cell
peritubular capillaries by ultrafiltration, a passive
because intracellular sodium concentration is low
process driven by the hydrostatic and colloid
(12 mEq/L) and tubular fluid sodium concentration is
osmotic pressure gradients.
high (140 mEq/L). (2) The negative, -70-millivolt,
intracellular potential attracts the positive sodium ions
Secondary Active Reabsorption Through the Tubular Membrane.
from the tubular lumen into the cell.
In secondary active transport, two or more substances
Active reabsorption of sodium by sodium-
interact with a specific membrane protein (a carrier
potassium ATPase occurs in most parts of the tubule.
molecule) and are transported together across the
In certain parts of the nephron, there are additional
membrane. As one of the substances (for instance,
provisions for moving large amounts of sodium into
sodium) diffuses down its electrochemical gradient,
the cell. In the proximal tubule, there is an extensive
the energy released is used to drive another substance
330
Unit V The Body Fluids and Kidneys
Interstitial
Tubular
Tubular
the simultaneous uphill transport of glucose across the
fluid
cells
lumen
luminal membrane. Thus, this reabsorption of glucose
Co-transport
is referred to as “secondary active transport” because
glucose itself is reabsorbed uphill against a chemical
Glucose
Glucose
gradient, but it is “secondary” to primary active trans-
port of sodium.
Na+
Na+
Another important point is that a substance is said
ATP
-70 mV
to undergo “active” transport when at least one of the
K+
steps in the reabsorption involves primary or second-
Na+
ary active transport, even though other steps in
Amino acids
Amino acids
the reabsorption process may be passive. For glucose
reabsorption, secondary active transport occurs at the
luminal membrane, but passive facilitated diffusion
occurs at the basolateral membrane, and passive
uptake by bulk flow occurs at the peritubular
capillaries.
Na+
Na+
ATP
-70 mV
Secondary Active Secretion into the Tubules. Some sub-
K+
H+
stances are secreted into the tubules by secondary
active transport. This often involves counter-transport
of the substance with sodium ions. In counter-
transport, the energy liberated from the downhill
Counter-transport
movement of one of the substances (for example,
sodium ions) enables uphill movement of a second
Figure 27-3
substance in the opposite direction.
Mechanisms of secondary active transport. The upper cell shows the
One example of counter-transport, shown in Figure
co-transport of glucose and amino acids along with sodium ions
27-3, is the active secretion of hydrogen ions coupled
through the apical side of the tubular epithelial cells, followed by
to sodium reabsorption in the luminal membrane of
facilitated diffusion through the basolateral membranes. The lower
the proximal tubule. In this case, sodium entry into the
cell shows the counter-transport of hydrogen ions from the interior
of the cell across the apical membrane and into the tubular lumen;
cell is coupled with hydrogen extrusion from the cell
movement of sodium ions into the cell, down an electrochemical gra-
by sodium-hydrogen counter-transport. This transport
dient established by the sodium-potassium pump on the basolateral
is mediated by a specific protein in the brush border
membrane, provides the energy for transport of the hydrogen ions
of the luminal membrane. As sodium is carried to the
from inside the cell into the tubular lumen.
interior of the cell, hydrogen ions are forced outward
in the opposite direction into the tubular lumen. The
(for instance, glucose) against its electrochemical
basic principles of primary and secondary active trans-
gradient. Thus, secondary active transport does not
port are discussed in additional detail in Chapter 4.
require energy directly from ATP or from other high-
energy phosphate sources. Rather, the direct source of
Pinocytosis—An Active Transport Mechanism for Reabsorption
the energy is that liberated by the simultaneous facil-
of Proteins. Some parts of the tubule, especially the
itated diffusion of another transported substance
proximal tubule, reabsorb large molecules such as
down its own electrochemical gradient.
proteins by pinocytosis. In this process, the protein
Figure 27-3 shows secondary active transport of
attaches to the brush border of the luminal membrane,
glucose and amino acids in the proximal tubule. In
and this portion of the membrane then invaginates to
both instances, a specific carrier protein in the brush
the interior of the cell until it is completely pinched off
border combines with a sodium ion and an amino acid
and a vesicle is formed containing the protein. Once
or a glucose molecule at the same time. These trans-
inside the cell, the protein is digested into its con-
port mechanisms are so efficient that they remove vir-
stituent amino acids, which are reabsorbed through the
tually all the glucose and amino acids from the tubular
basolateral membrane into the interstitial fluid.
lumen. After entry into the cell, glucose and amino
Because pinocytosis requires energy, it is considered a
acids exit across the basolateral membranes by facili-
form of active transport.
tated diffusion, driven by the high glucose and amino
acid concentrations in the cell.
Transport Maximum for Substances That Are Actively Reab-
Although transport of glucose against a chemical
sorbed. For most substances that are actively reab-
gradient does not directly use ATP, the reabsorption
sorbed or secreted, there is a limit to the rate at which
of glucose depends on energy expended by the
the solute can be transported, often referred to as the
primary active sodium-potassium ATPase pump in the
transport maximum. This limit is due to saturation
basolateral membrane. Because of the activity of
of the specific transport systems involved when the
this pump, an electrochemical gradient for facilitated
amount of solute delivered to the tubule (referred to
diffusion of sodium across the luminal membrane is
as tubular load) exceeds the capacity of the carrier
maintained, and it is this downhill diffusion of sodium
proteins and specific enzymes involved in the trans-
to the interior of the cell that provides the energy for
port process.
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
331
nephrons have the same transport maximum for
glucose, and some of the nephrons excrete glucose
900
before others have reached their transport maximum.
The overall transport maximum for the kidneys, which
800
is normally about 375 mg/min, is reached when all
nephrons have reached their maximal capacity to reab-
700
Filtered
sorb glucose.
load
The plasma glucose of a healthy person almost
600
never becomes high enough to cause excretion of
Excretion
500
glucose in the urine, even after eating a meal.
However, in uncontrolled diabetes mellitus, plasma
400
glucose may rise to high levels, causing the filtered load
of glucose to exceed the transport maximum and
Transport
300
resulting in urinary glucose excretion. Some of the
maximum Reabsorption
Normal
important transport maximums for substances actively
200
reabsorbed by the tubules are as follows:
100
Threshold
Substance
Transport Maximum
0
Glucose
375 mg/min
0
100
200
300
400
500
600
700
800
Phosphate
0.10 mM/min
Plasma glucose concentration
Sulfate
0.06 mM/min
(mg/100 ml)
Amino acids
1.5 mM/min
Urate
15 mg/min
Lactate
75 mg/min
Plasma protein
30 mg/min
Figure 27-4
Relations among the filtered load of glucose, the rate of glucose
Transport Maximums for Substances That Are Actively
reabsorption by the renal tubules, and the rate of glucose excretion
in the urine. The transport maximum is the maximum rate at which
Secreted. Substances that are actively secreted also
glucose can be reabsorbed from the tubules. The threshold for
exhibit transport maximums as follows:
glucose refers to the filtered load of glucose at which glucose first
begins to be excreted in the urine.
Substance
Transport Maximum
The glucose transport system in the proximal tubule
Creatinine
16 mg/min
is a good example. Normally, measurable glucose does
Para-aminohippuric acid
80 mg/min
not appear in the urine because essentially all the fil-
tered glucose is reabsorbed in the proximal tubule.
However, when the filtered load exceeds the capabil-
Substances That Are Actively Transported but Do Not Exhibit a
ity of the tubules to reabsorb glucose, urinary excre-
Transport Maximum. The reason that actively trans-
tion of glucose does occur.
ported solutes often exhibit a transport maximum is
In the adult human, the transport maximum for
that the transport carrier system becomes saturated as
glucose averages about 375 mg/min, whereas the fil-
the tubular load increases. Substances that are passively
tered load of glucose is only about 125 mg/min (GFR
reabsorbed do not demonstrate a transport maximum
¥ plasma glucose = 125 ml/min ¥ 1 mg/ml). With large
because their rate of transport is determined by other
increases in GFR and/or plasma glucose concentration
factors, such as (1) the electrochemical gradient for
that increase the filtered load of glucose above 375 mg/
diffusion of the substance across the membrane, (2)
min, the excess glucose filtered is not reabsorbed and
the permeability of the membrane for the substance,
passes into the urine.
and (3) the time that the fluid containing the substance
Figure 27-4 shows the relation between plasma con-
remains within the tubule. Transport of this type is
centration of glucose, filtered load of glucose, tubular
referred to as gradient-time transport because the rate
transport maximum for glucose, and rate of glucose
of transport depends on the electrochemical gradient
loss in the urine. Note that when the plasma glucose
and the time that the substance is in the tubule, which
concentration is 100 mg/100 mL and the filtered load
in turn depends on the tubular flow rate.
is at its normal level, 125 mg/min, there is no loss of
Some actively transported substances also have char-
glucose in the urine. However, when the plasma con-
acteristics of gradient-time transport. An example is
centration of glucose rises above about 200 mg/100 ml,
sodium reabsorption in the proximal tubule. The
increasing the filtered load to about 250 mg/min, a
main reason that sodium transport in the proximal
small amount of glucose begins to appear in the urine.
tubule does not exhibit a transport maximum is that
This point is termed the threshold for glucose. Note
other factors limit the reabsorption rate besides the
that this appearance of glucose in the urine (at the
maximum rate of active transport. For example, in the
threshold) occurs before the transport maximum is
proximal tubules, the maximum transport capacity of
reached. One reason for the difference between
the basolateral sodium-potassium ATPase pump is
threshold and transport maximum is that not all
usually far greater than the actual rate of net sodium
332
Unit V The Body Fluids and Kidneys
reabsorption. One of the reasons for this is that a sig-
lecting tubule, the tight junctions become far less per-
nificant amount of sodium transported out of the
meable to water and solutes, and the epithelial cells
cell leaks back into the tubular lumen through the
also have a greatly decreased membrane surface area.
epithelial tight junctions. The rate at which this back-
Therefore, water cannot move easily across the tubular
leak occurs depends on several factors, including (1)
membrane by osmosis. However, antidiuretic hormone
the permeability of the tight junctions and (2) the
(ADH) greatly increases the water permeability in the
interstitial physical forces, which determine the rate of
distal and collecting tubules, as discussed later.
bulk flow reabsorption from the interstitial fluid into
Thus, water movement across the tubular epithe-
the peritubular capillaries. Therefore, sodium trans-
lium can occur only if the membrane is permeable to
port in the proximal tubules obeys mainly gradient-
water, no matter how large the osmotic gradient. In the
time transport principles rather than tubular
proximal tubule, the water permeability is always high,
maximum transport characteristics. This means that
and water is reabsorbed as rapidly as the solutes. In
the greater the concentration of sodium in the proxi-
the ascending loop of Henle, water permeability is
mal tubules, the greater its reabsorption rate. Also, the
always low, so that almost no water is reabsorbed,
slower the flow rate of tubular fluid, the greater the
despite a large osmotic gradient. Water permeability in
percentage of sodium that can be reabsorbed from
the last parts of the tubules—the distal tubules, col-
the proximal tubules.
lecting tubules, and collecting ducts—can be high or
In the more distal parts of the nephron, the epithe-
low, depending on the presence or absence of ADH.
lial cells have much tighter junctions and transport
much smaller amounts of sodium. In these segments,
sodium reabsorption exhibits a transport maximum
Reabsorption of Chloride, Urea, and
similar to that for other actively transported substances.
Other Solutes by Passive Diffusion
Furthermore, this transport maximum can be increased
in response to certain hormones, such as aldosterone.
When sodium is reabsorbed through the tubular
epithelial cell, negative ions such as chloride are trans-
ported along with sodium because of electrical poten-
Passive Water Reabsorption
tials. That is, transport of positively charged sodium
by Osmosis Is Coupled Mainly
ions out of the lumen leaves the inside of the lumen
to Sodium Reabsorption
negatively charged, compared with the interstitial
fluid. This causes chloride ions to diffuse passively
When solutes are transported out of the tubule by
through the paracellular pathway. Additional reab-
either primary or secondary active transport, their
sorption of chloride ions occurs because of a chloride
concentrations tend to decrease inside the tubule
concentration gradient that develops when water is
while increasing in the renal interstitium. This creates
reabsorbed from the tubule by osmosis, thereby con-
a concentration difference that causes osmosis of
centrating the chloride ions in the tubular lumen
water in the same direction that the solutes are trans-
(Figure 27-5). Thus, the active reabsorption of sodium
ported, from the tubular lumen to the renal intersti-
is closely coupled to the passive reabsorption of chlo-
tium. Some parts of the renal tubule, especially the
ride by way of an electrical potential and a chloride
proximal tubule, are highly permeable to water, and
concentration gradient.
water reabsorption occurs so rapidly that there is only
a small concentration gradient for solutes across the
tubular membrane.
A large part of the osmotic flow of water occurs
Na+ reabsorption
through the so-called tight junctions between the
epithelial cells as well as through the cells themselves.
The reason for this, as already discussed, is that the
junctions between the cells are not as tight as their
H2O reabsorption
name would imply, and they allow significant diffusion
of water and small ions. This is especially true in the
proximal tubules, which have a high permeability for
water and a smaller but significant permeability to
Lumen
Luminal
Luminal Cl-
most ions, such as sodium, chloride, potassium,
negative
urea
concentration
potential
concentration
calcium, and magnesium.
As water moves across the tight junctions by
osmosis, it can also carry with it some of the solutes, a
process referred to as solvent drag. And because the
Passive Cl-
Passive urea
reabsorption of water, organic solutes, and ions is
reabsorption
reabsorption
coupled to sodium reabsorption, changes in sodium
reabsorption significantly influence the reabsorption
Figure 27-5
of water and many other solutes.
In the more distal parts of the nephron, beginning
Mechanisms by which water, chloride, and urea reabsorption are
in the loop of Henle and extending through the col-
coupled with sodium reabsorption.
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
333
Chloride ions can also be reabsorbed by secondary
65%
active transport. The most important of the secondary
active transport processes for chloride reabsorption
involves co-transport of chloride with sodium across
Proximal tubule
the luminal membrane.
Urea is also passively reabsorbed from the tubule,
but to a much lesser extent than chloride ions. As
Na+, Cl-, HCO3-, K+,
water is reabsorbed from the tubules (by osmosis
H2O, glucose, amino acids
coupled to sodium reabsorption), urea concentration
in the tubular lumen increases (see Figure 27-5). This
Isosmotic
creates a concentration gradient favoring the reab-
sorption of urea. However, urea does not permeate
H+, organic acids, bases
the tubule as readily as water. In some parts of the
nephron, especially the inner medullary collecting
duct, passive urea reabsorption is facilitated by specific
urea transporters. Yet only about one half of the urea
that is filtered by the glomerular capillaries is reab-
Figure 27-6
sorbed from the tubules. The remainder of the urea
passes into the urine, allowing the kidneys to excrete
Cellular ultrastructure and primary transport characteristics of the
proximal tubule. The proximal tubules reabsorb about 65 per cent
large amounts of this waste product of metabolism.
of the filtered sodium, chloride, bicarbonate, and potassium and
Another waste product of metabolism, creatinine, is
essentially all the filtered glucose and amino acids. The proximal
an even larger molecule than urea and is essentially
tubules also secrete organic acids, bases, and hydrogen ions into the
impermeant to the tubular membrane. Therefore,
tubular lumen.
almost none of the creatinine that is filtered is reab-
sorbed, so that virtually all the creatinine filtered by
the glomerulus is excreted in the urine.
tubular cells have an extensive brush border on the
luminal (apical) side of the membrane as well as an
extensive labyrinth of intercellular and basal channels,
Reabsorption and Secretion
all of which together provide an extensive membrane
Along Different Parts of
surface area on the luminal and basolateral sides of the
the Nephron
epithelium for rapid transport of sodium ions and
other substances.
In the previous sections, we discussed the basic princi-
The extensive membrane surface of the epithelial
ples by which water and solutes are transported across
brush border is also loaded with protein carrier mole-
the tubular membrane. With these generalizations in
cules that transport a large fraction of the sodium ions
mind, we can now discuss the different characteristics
across the luminal membrane linked by way of the co-
of the individual tubular segments that enable them to
transport mechanism with multiple organic nutrients
perform their specific excretory functions. Only the
such as amino acids and glucose. The remainder of the
tubular transport functions that are quantitatively
sodium is transported from the tubular lumen into the
most important are discussed, especially as they relate
cell by counter-transport mechanisms, which reabsorb
to the reabsorption of sodium, chloride, and water. In
sodium while secreting other substances into the
subsequent chapters, we discuss the reabsorption and
tubular lumen, especially hydrogen ions. As discussed
secretion of other specific substances in different parts
in Chapter 30, the secretion of hydrogen ions into the
of the tubular system.
tubular lumen is an important step in the removal of
bicarbonate ions from the tubule (by combining H+
with the HCO3_ to form H2CO3, which then dissociates
Proximal Tubular Reabsorption
into H2O and CO2).
Although the sodium-potassium ATPase pump pro-
Normally, about 65 per cent of the filtered load of
vides the major force for reabsorption of sodium,
sodium and water and a slightly lower percentage of
chloride, and water throughout the proximal tubule,
filtered chloride are reabsorbed by the proximal
there are some differences in the mechanisms by
tubule before the filtrate reaches the loops of Henle.
which sodium and chloride are transported through
These percentages can be increased or decreased in
the luminal side of the early and late portions of the
different physiologic conditions, as discussed later.
proximal tubular membrane.
In the first half of the proximal tubule, sodium is
Proximal Tubules Have a High Capacity for Active and Passive
reabsorbed by co-transport along with glucose, amino
Reabsorption. The high capacity of the proximal tubule
acids, and other solutes. But in the second half of the
for reabsorption results from its special cellular char-
proximal tubule, little glucose and amino acids remain
acteristics, as shown in Figure
27-6. The proximal
to be reabsorbed. Instead, sodium is now reabsorbed
tubule epithelial cells are highly metabolic and have
mainly with chloride ions. The second half of the prox-
large numbers of mitochondria to support potent
imal tubule has a relatively high concentration of
active transport processes. In addition, the proximal
chloride (around 140 mEq/L) compared with the early
334
Unit V The Body Fluids and Kidneys
Secretion of Organic Acids and Bases by the Proximal Tubule.
The proximal tubule is also an important site for secre-
5.0
tion of organic acids and bases such as bile salts,
Creatinine
oxalate, urate, and catecholamines. Many of these sub-
2.0
stances are the end products of metabolism and must
Urea
Cl-
be rapidly removed from the body. The secretion of
1.0
these substances into the proximal tubule plus filtra-
Na+
Osmolality
tion into the proximal tubule by the glomerular capil-
0.5
laries and the almost total lack of reabsorption by the
tubules, all combined, contribute to rapid excretion in
HCO3
-
0.2
the urine.
In addition to the waste products of metabolism, the
0.1
kidneys secrete many potentially harmful drugs or
toxins directly through the tubular cells into the
tubules and rapidly clear these substances from the
Glucose
0.05
blood. In the case of certain drugs, such as penicillin
Amino acids
and salicylates, the rapid clearance by the kidneys
creates a problem in maintaining a therapeutically
0.01
effective drug concentration.
0
20
40
60
80
100
Another compound that is rapidly secreted by the
% Total proximal tubule length
proximal tubule is para-aminohippuric acid (PAH).
PAH is secreted so rapidly that the average person
can clear about 90 per cent of the PAH from the
plasma flowing through the kidneys and excrete it in
Figure 27-7
the urine. For this reason, the rate of PAH clearance
can be used to estimate the renal plasma flow, as dis-
Changes in concentrations of different substances in tubular fluid
cussed later.
along the proximal convoluted tubule relative to the concentrations
of these substances in the plasma and in the glomerular filtrate. A
value of 1.0 indicates that the concentration of the substance in the
Solute and Water Transport in the
tubular fluid is the same as the concentration in the plasma. Values
below 1.0 indicate that the substance is reabsorbed more avidly than
Loop of Henle
water, whereas values above 1.0 indicate that the substance is reab-
sorbed to a lesser extent than water or is secreted into the tubules.
The loop of Henle consists of three functionally dis-
tinct segments: the thin descending segment, the thin
proximal tubule (about 105 mEq/L) because when
ascending segment, and the thick ascending segment.
sodium is reabsorbed, it preferentially carries with it
The thin descending and thin ascending segments, as
glucose, bicarbonate, and organic ions in the early
their names imply, have thin epithelial membranes
proximal tubule, leaving behind a solution that has a
with no brush borders, few mitochondria, and minimal
higher concentration of chloride. In the second half of
levels of metabolic activity (Figure 27-8).
the proximal tubule, the higher chloride concentration
The descending part of the thin segment is highly
favors the diffusion of this ion from the tubule lumen
permeable to water and moderately permeable to
through the intercellular junctions into the renal inter-
most solutes, including urea and sodium. The function
stitial fluid.
of this nephron segment is mainly to allow simple dif-
Concentrations of Solutes Along the Proximal Tubule. Figure
fusion of substances through its walls. About 20 per
27-7 summarizes the changes in concentrations of
cent of the filtered water is reabsorbed in the loop of
various solutes along the proximal tubule. Although
Henle, and almost all of this occurs in the thin descend-
the amount of sodium in the tubular fluid decreases
ing limb. The ascending limb, including both the thin
markedly along the proximal tubule, the concentration
and the thick portions, is virtually impermeable to
of sodium (and the total osmolarity) remains relatively
water, a characteristic that is important for concen-
constant because water permeability of the proximal
trating the urine.
tubules is so great that water reabsorption keeps pace
The thick segment of the loop of Henle, which
with sodium reabsorption. Certain organic solutes,
begins about halfway up the ascending limb, has thick
such as glucose, amino acids, and bicarbonate, are
epithelial cells that have high metabolic activity and
much more avidly reabsorbed than water, so that their
are capable of active reabsorption of sodium, chloride,
concentrations decrease markedly along the length of
and potassium (see Figure 27-8). About 25 per cent of
the proximal tubule. Other organic solutes that are less
the filtered loads of sodium, chloride, and potassium
permeant and not actively reabsorbed, such as creati-
are reabsorbed in the loop of Henle, mostly in the
nine, increase their concentration along the proximal
thick ascending limb. Considerable amounts of other
tubule. The total solute concentration, as reflected by
ions, such as calcium, bicarbonate, and magnesium, are
osmolarity, remains essentially the same all along the
also reabsorbed in the thick ascending loop of Henle.
proximal tubule because of the extremely high per-
The thin segment of the ascending limb has a much
meability of this part of the nephron to water.
lower reabsorptive capacity than the thick segment,
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
335
Thin descending
Renal
Tubular
loop of Henle
interstitial
Tubular
lumen
fluid
cells
(+8 mV)
Paracellular
Na+, K+
Mg++, Ca++
diffusion
H2O
Na+
Na+
ATP
H+
K+
Na+
Cl-
K+
2Cl-
-
K+
Thick ascending
loop of Henle
25%
Loop diuretics
Na+, Cl-, K+,
• Furosemide
-, Mg++
Ca++, HCO3
• Ethacrynic acid
• Bumetanide
Hypo-
osmotic
Figure 27-9
H+
Mechanisms of sodium, chloride, and potassium transport in the
thick ascending loop of Henle. The sodium-potassium ATPase pump
in the basolateral cell membrane maintains a low intracellular
sodium concentration and a negative electrical potential in the cell.
The 1-sodium, 2-chloride, 1-potassium co-transporter in the luminal
Figure 27-8
membrane transports these three ions from the tubular lumen into
the cells, using the potential energy released by diffusion of sodium
Cellular ultrastructure
and transport characteristics of the thin
down an electrochemical gradient into the cells. Sodium is also
descending loop of Henle (top) and the thick ascending segment of
transported into the tubular cell by sodium-hydrogen counter-trans-
the loop of Henle (bottom). The descending part of the thin segment
port. The positive charge (+8 mV) of the tubular lumen relative to
of the loop of Henle is highly permeable to water and moderately
the interstitial fluid forces cations such as Mg++ and Ca++ to diffuse
permeable to most solutes but has few mitochondria and little or no
from the lumen to the interstitial fluid via the paracellular pathway.
active reabsorption. The thick ascending limb of the loop of Henle
reabsorbs about 25 per cent of the filtered loads of sodium, chloride,
and potassium, as well as large amounts of calcium, bicarbonate,
and magnesium. This segment also secretes hydrogen ions into the
The thick ascending limb of the loop of Henle is
tubular lumen.
the site of action of the powerful “loop” diuretics
furosemide, ethacrynic acid, and bumetanide, all of
which inhibit the action of the sodium 2-chloride,
and the thin descending limb does not reabsorb sig-
potassium co-transporter. These diuretics are dis-
nificant amounts of any of these solutes.
cussed in Chapter 31.
An important component of solute reabsorption in
There is also significant paracellular reabsorption of
the thick ascending limb is the sodium-potassium
cations, such as Mg++, Ca++, Na+, and K+, in the thick
ATPase pump in the epithelial cell basolateral mem-
ascending limb owing to the slight positive charge of
branes. As in the proximal tubule, the reabsorption of
the tubular lumen relative to the interstitial fluid.
other solutes in the thick segment of the ascending
Although the 1-sodium, 2-chloride, 1-potassium co-
loop of Henle is closely linked to the reabsorptive
transporter moves equal amounts of cations and
capability of the sodium-potassium ATPase pump,
anions into the cell, there is a slight backleak of potas-
which maintains a low intracellular sodium concentra-
sium ions into the lumen, creating a positive charge of
tion. The low intracellular sodium concentration in
about +8 millivolts in the tubular lumen. This positive
turn provides a favorable gradient for movement
charge forces cations such as Mg++ and Ca++ to diffuse
of sodium from the tubular fluid into the cell. In the
from the tubular lumen through the paracellular space
thick ascending loop, movement of sodium across
and into the interstitial fluid.
the luminal membrane is mediated primarily by a
The thick ascending limb also has a sodium-
1-sodium,
2-chloride,
1-potassium co-transporter
hydrogen counter-transport mechanism in its luminal
(Figure 27-9). This co-transport protein carrier in the
cell membrane that mediates sodium reabsorption and
luminal membrane uses the potential energy released
hydrogen secretion in this segment.
by downhill diffusion of sodium into the cell to drive
The thick segment of the ascending loop of Henle is
the reabsorption of potassium into the cell against a
virtually impermeable to water. Therefore, most of the
concentration gradient.
water delivered to this segment remains in the tubule,
336
Unit V The Body Fluids and Kidneys
despite reabsorption of large amounts of solute. The
Late Distal Tubule and Cortical
tubular fluid in the ascending limb becomes very dilute
Collecting Tubule
as it flows toward the distal tubule, a feature that is
important in allowing the kidneys to dilute or concen-
The second half of the distal tubule and the subse-
trate the urine under different conditions, as we
quent cortical collecting tubule have similar functional
discuss much more fully in Chapter 28.
characteristics. Anatomically, they are composed of
two distinct cell types, the principal cells and the inter-
calated cells (Figure 27-11). The principal cells reab-
Distal Tubule
sorb sodium and water from the lumen and secrete
potassium ions into the lumen. The intercalated cells
The thick segment of the ascending limb of the loop
reabsorb potassium ions and secrete hydrogen ions
of Henle empties into the distal tubule. The very first
into the tubular lumen.
portion of the distal tubule forms part of the juxta-
glomerular complex that provides feedback control of
Principal Cells Reabsorb Sodium and Secrete Potassium.
GFR and blood flow in this same nephron. The next
Sodium reabsorption and potassium secretion by the
part of the distal tubule is highly convoluted and has
principal cells depend on the activity of a sodium-
many of the same reabsorptive characteristics of the
potassium ATPase pump in each cell’s basolateral
thick segment of the ascending limb of the loop of
membrane (Figure 27-12). This pump maintains a low
Henle. That is, it avidly reabsorbs most of the ions,
sodium concentration inside the cell and, therefore,
including sodium, potassium, and chloride, but is vir-
favors sodium diffusion into the cell through special
tually impermeable to water and urea. For this reason,
it is referred to as the diluting segment because it also
dilutes the tubular fluid.
Approximately 5 percent of the filtered load of
Early distal tubule
sodium chloride is reabsorbed in the early distal
tubule. The sodium-chloride co-transporter moves
sodium chloride from the tubular lumen into the cell,
and the sodium-potassium ATPase pump transports
Na+, Cl-, Ca++, Mg++
sodium out of the cell across the basolateral mem-
brane (Figure 27-10). Chloride diffuses out of the cell
into the renal interstitial fluid through chloride chan-
nels in the basolateral membrane. The thiazide diuret-
ics, which are widely used to treat disorders such as
hypertension and heart failure, inhibit the sodium-
chloride co-transporter.
Late distal tubule
and collecting tubule
Principal
Na+, Cl-
cells
Renal
Tubular
interstitial
Tubular
lumen
fluid
cells
(-10mV)
(+ADH) H2O
K+
H+
Na+
ATP
Na+
-
K+
Intercalated
HCO3
-
K+
cells
Cl-
Cl-
Figure 27-11
Cellular ultrastructure and transport characteristics of the early
distal tubule and the late distal tubule and collecting tubule. The
Thiazide diuretics:
early distal tubule has many of the same characteristics as the thick
ascending loop of Henle and reabsorbs sodium, chloride, calcium,
and magnesium but is virtually impermeable to water and urea. The
Figure 27-10
late distal tubules and cortical collecting tubules are composed of
two distinct cell types, the principal cells and the intercalated cells.
Mechanism of sodium chloride transport in the early distal tubule.
The principal cells reabsorb sodium from the lumen and secrete
Sodium and chloride are transported from the tubular lumen into
potassium ions into the lumen. The intercalated cells reabsorb potas-
the cell by a co-transporter that is inhibited by thiazide diuretics.
sium and bicarbonate ions from the lumen and secrete hydrogen
Sodium is pumped out of the cell by sodium-potassium ATPase and
ions into the lumen. The reabsorption of water from this tubular
chloride diffuses into the interstitial fluid via chloride channels.
segment is controlled by the concentration of antidiuretic hormone.
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
337
Renal
Tubular
then dissociates into hydrogen ions and bicarbonate
interstitial
Tubular
lumen
ions. The hydrogen ions are then secreted into the
fluid
cells
(-50 mV)
tubular lumen, and for each hydrogen ion secreted, a
bicarbonate ion becomes available for reabsorption
across the basolateral membrane. A more detailed dis-
cussion of this mechanism is presented in Chapter 30.
The intercalated cells can also reabsorb potassium
K+
ions.
Na+
The functional characteristics of the late distal tubule
+
ATP
Na
and cortical collecting tubule can be summarized as
K+
-
-
follows:
1.
The tubular membranes of both segments are
Cl-
almost completely impermeable to urea, similar
to the diluting segment of the early distal tubule;
thus, almost all the urea that enters these
Aldosterone antagonists
Na+ channel blockers
segments passes on through and into the
• Spironolactone
• Amiloride
collecting duct to be excreted in the urine,
• Eplerenone
• Triamterene
although some reabsorption of urea occurs
in the medullary collecting ducts.
Figure 27-12
2.
Both the late distal tubule and the cortical
collecting tubule segments reabsorb sodium ions,
Mechanism of sodium chloride reabsorption and potassium secre-
and the rate of reabsorption is controlled by
tion in the late distal tubules and cortical collecting tubules. Sodium
hormones, especially aldosterone. At the same
enters the cell through special channels and is transported out of the
cell by the sodium-potassium ATPase pump. Aldosterone antago-
time, these segments secrete potassium ions
nists compete with aldosterone for binding sites in the cell and
from the peritubular capillary blood into the
therefore inhibit the effects of aldosterone to stimulate sodium reab-
tubular lumen, a process that is also controlled
sorption and potassium secretion. Sodium channel blockers directly
by aldosterone and by other factors such as
inhibit the entry of sodium into the sodium channels.
the concentration of potassium ions in the body
fluids.
channels. The secretion of potassium by these cells
3.
The intercalated cells of these nephron segments
from the blood into the tubular lumen involves two
avidly secrete hydrogen ions by an active
steps: (1) Potassium enters the cell because of the
hydrogen-ATPase mechanism. This process is
sodium-potassium ATPase pump, which maintains a
different from the secondary active secretion of
high intracellular potassium concentration, and then
hydrogen ions by the proximal tubule because it is
(2) once in the cell, potassium diffuses down its con-
capable of secreting hydrogen ions against a large
centration gradient across the luminal membrane into
concentration gradient, as much as 1000 to 1. This
the tubular fluid.
is in contrast to the relatively small gradient (4- to
The principal cells are the primary sites of action
10-fold) for hydrogen ions that can be achieved
of the potassium-sparing diuretics, including spirono-
by secondary active secretion in the proximal
lactone, eplerenone, amiloride, and triamterene.
tubule. Thus, the intercalated cells play a key role
Aldosterone antagonists compete with aldosterone for
in acid-base regulation of the body fluids.
receptor sites in the principal cells and therefore
4.
The permeability of the late distal tubule and
inhibit the stimulatory effects of aldosterone on
cortical collecting duct to water is controlled by
sodium reabsorption and potassium secretion. Sodium
the concentration of ADH, which is also called
channel blockers directly inhibit the entry of sodium
vasopressin. With high levels of ADH, these
into the sodium channels of the luminal membranes
tubular segments are permeable to water, but
and therefore reduce the amount of sodium that can
in the absence of ADH, they are virtually
be transported across the basolateral membranes by
impermeable to water. This special characteristic
the sodium-potassium ATPase pump. This, in turn,
provides an important mechanism for controlling
decreases transport of potassium into the cells and
the degree of dilution or concentration of the
ultimately reduces potassium secretion into the
urine.
tubular fluid. For this reason the sodium channel
blockers as well as the aldosterone antagonists
decrease urinary excretion of potassium and act as
Medullary Collecting Duct
potassium-sparing diuretics.
Although the medullary collecting ducts reabsorb less
Intercalated Cells Avidly Secrete Hydrogen and Reabsorb Bicar-
than 10 per cent of the filtered water and sodium, they
bonate and Potassium Ions. Hydrogen ion secretion by
are the final site for processing the urine and, there-
the intercalated cells is mediated by a hydrogen-
fore, play an extremely important role in determining
ATPase transport mechanism. Hydrogen is generated
the final urine output of water and solutes.
in this cell by the action of carbonic anhydrase on
The epithelial cells of the collecting ducts are nearly
water and carbon dioxide to form carbonic acid, which
cuboidal in shape with smooth surfaces and relatively
338
Unit V The Body Fluids and Kidneys
Medullary
collecting duct
100.0
50.0
PAH
Urea
20.0
10.0
H+
5.0
Cl
2.0
K
1.0
K
Cl
and Na
Na
0.50
Figure 27-13
Cellular ultrastructure and transport characteristics of the
0.20
medullary collecting duct. The medullary collecting ducts actively
reabsorb sodium and secrete hydrogen ions and are permeable to
0.10
urea, which is reabsorbed in these tubular segments. The reabsorp-
HCO3
3
tion of water in medullary collecting ducts is controlled by the con-
0.05
centration of antidiuretic hormone.
0.02
Proximal
Loop of
Distal
Collecting
few mitochondria (Figure 27-13). Special characteris-
tubule
Henle
tubule
tubule
tics of this tubular segment are as follows:
1. The permeability of the medullary collecting duct
to water is controlled by the level of ADH. With
Figure 27-14
high levels of ADH, water is avidly reabsorbed
into the medullary interstitium, thereby reducing
Changes in average concentrations of different substances at differ-
the urine volume and concentrating most of the
ent points in the tubular system relative to the concentration of that
solutes in the urine.
substance in the plasma and in the glomerular filtrate. A value of 1.0
indicates that the concentration of the substance in the tubular fluid
2. Unlike the cortical collecting tubule, the
is the same as the concentration of that substance in the plasma.
medullary collecting duct is permeable to urea.
Values below 1.0 indicate that the substance is reabsorbed more
Therefore, some of the tubular urea is reabsorbed
avidly than water, whereas values above 1.0 indicate that the sub-
into the medullary interstitium, helping to raise
stance is reabsorbed to a lesser extent than water or is secreted into
the tubules.
the osmolality in this region of the kidneys and
contributing to the kidneys’ overall ability to form
a concentrated urine.
3. The medullary collecting duct is capable of
a substance. If plasma concentration of the substance
secreting hydrogen ions against a large
is assumed to be constant, any change in the ratio
concentration gradient, as also occurs in the
of tubular fluid/plasma concentration rate reflects
cortical collecting tubule. Thus, the medullary
changes in tubular fluid concentration.
collecting duct also plays a key role in regulating
As the filtrate moves along the tubular system, the
acid-base balance.
concentration rises to progressively greater than 1.0 if
more water is reabsorbed than solute, or if there has
been a net secretion of the solute into the tubular fluid.
Summary of Concentrations of
If the concentration ratio becomes progressively less
Different Solutes in the Different
than 1.0, this means that relatively more solute has
Tubular Segments
been reabsorbed than water.
The substances represented at the top of Figure
Whether a solute will become concentrated in the
27-14, such as creatinine, become highly concentrated
tubular fluid is determined by the relative degree of
in the urine. In general, these substances are not
reabsorption of that solute versus the reabsorption of
needed by the body, and the kidneys have become
water. If a greater percentage of water is reabsorbed,
adapted to reabsorb them only slightly or not at all, or
the substance becomes more concentrated. If a greater
even to secrete them into the tubules, thereby excret-
percentage of the solute is reabsorbed, the substance
ing especially great quantities into the urine. Con-
becomes more diluted.
versely, the substances represented toward the bottom
Figure 27-14 shows the degree of concentration of
of the figure, such as glucose and amino acids, are all
several substances in the different tubular segments.
strongly reabsorbed; these are all substances that the
All the values in this figure represent the tubular fluid
body needs to conserve, and almost none of them are
concentration divided by the plasma concentration of
lost in the urine.
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
339
Tubular Fluid /Plasma Inulin Concentration Ratio Can Be Used
are not fully understood but may be due partly to
to Measure Water Reabsorption by the Renal Tubules. Inulin,
changes in physical forces in the tubule and surround-
a polysaccharide used to measure GFR, is not reab-
ing renal interstitium, as discussed later. It is clear that
sorbed or secreted by the renal tubules. Changes in
the mechanisms for glomerulotubular balance can
inulin concentration at different points along the renal
occur independently of hormones and can be demon-
tubule, therefore, reflect changes in the amount of
strated in completely isolated kidneys or even in com-
water present in the tubular fluid. For example, the
pletely isolated proximal tubular segments.
tubular fluid/plasma concentration ratio for inulin
The importance of glomerulotubular balance is that
rises to about 3.0 at the end of the proximal tubules,
it helps to prevent overloading of the distal tubular
indicating that inulin concentration in the tubular fluid
segments when GFR increases. Glomerulotubular
is
3 times greater than in the plasma and in the
balance acts as a second line of defense to buffer the
glomerular filtrate. Since inulin is not secreted or reab-
effects of spontaneous changes in GFR on urine
sorbed from the tubules, a tubular fluid/plasma con-
output. (The first line of defense, discussed earlier,
centration ratio of 3.0 means that only one third of the
includes the renal autoregulatory mechanisms, espe-
water that was filtered remains in the renal tubule and
cially tubuloglomerular feedback, which help prevent
that two thirds of the filtered water has been reab-
changes in GFR.) Working together, the autoregula-
sorbed as the fluid passes through the proximal tubule.
tory and glomerulotubular balance mechanisms
At the end of the collecting ducts, the tubular
prevent large changes in fluid flow in the distal tubules
fluid/plasma inulin concentration ratio rises to about
when the arterial pressure changes or when there
125 (see Figure 27-14), indicating that only 1/125 of
are other disturbances that would otherwise wreak
the filtered water remains in the tubule and that more
havoc with the maintenance of sodium and volume
than 99% has been reabsorbed.
homeostasis.
Peritubular Capillary and Renal
Regulation of Tubular
Interstitial Fluid Physical Forces
Reabsorption
Hydrostatic and colloid osmotic forces govern the rate
Because it is essential to maintain a precise balance
of reabsorption across the peritubular capillaries,
between tubular reabsorption and glomerular filtra-
just as these physical forces control filtration in the
tion, there are multiple nervous, hormonal, and local
glomerular capillaries. Changes in peritubular capil-
control mechanisms that regulate tubular reabsorp-
lary reabsorption can in turn influence the hydrostatic
tion, just as there are for control of glomerular filtra-
and colloid osmotic pressures of the renal interstitium
tion. An important feature of tubular reabsorption is
and, ultimately, reabsorption of water and solutes from
that reabsorption of some solutes can be regulated
the renal tubules.
independently of others, especially through hormonal
control mechanisms.
Normal Values for Physical Forces and Reabsorption Rate. As
the glomerular filtrate passes through the renal
tubules, more than 99 per cent of the water and most
Glomerulotubular Balance—The
of the solutes are normally reabsorbed. Fluid and elec-
Ability of the Tubules to Increase
trolytes are reabsorbed from the tubules into the renal
Reabsorption Rate in Response
interstitium and from there into the peritubular capil-
to Increased Tubular Load
laries. The normal rate of peritubular capillary reab-
sorption is about 124 ml/min.
One of the most basic mechanisms for controlling
Reabsorption across the peritubular capillaries can
tubular reabsorption is the intrinsic ability of the
be calculated as
tubules to increase their reabsorption rate in response
Reabsorption = Kf ¥ Net reabsorptive force
to increased tubular load (increased tubular inflow).
This phenomenon is referred to as glomerulotubular
The net reabsorptive force represents the sum of the
balance. For example, if GFR is increased from 125 ml/
hydrostatic and colloid osmotic forces that either favor
min to 150 ml/min, the absolute rate of proximal
or oppose reabsorption across the peritubular capil-
tubular reabsorption also increases from about 81 ml/
laries. These forces include (1) hydrostatic pressure
min (65 per cent of GFR) to about 97.5 ml/min (65 per
inside the peritubular capillaries (peritubular hydro-
cent of GFR). Thus, glomerulotubular balance refers
static pressure
[Pc]), which opposes reabsorption;
to the fact that the total rate of reabsorption increases
(2) hydrostatic pressure in the renal interstitium (Pif)
as the filtered load increases, even though the
outside the capillaries, which favors reabsorption; (3)
percentage of GFR reabsorbed in the proximal
colloid osmotic pressure of the peritubular capillary
tubule remains relatively constant at about 65 per
plasma proteins (pc), which favors reabsorption; and
cent.
(4) colloid osmotic pressure of the proteins in the renal
Some degree of glomerulotubular balance also
interstitium (pif), which opposes reabsorption.
occurs in other tubular segments, especially the loop
Figure 27-15 shows the approximate normal forces
of Henle. The precise mechanisms responsible for this
that favor and oppose peritubular reabsorption.
340
Unit V The Body Fluids and Kidneys
Peritubular
Interstitial
Tubular
Tubular
raise peritubular capillary hydrostatic pressure and
capillary
fluid
cells
lumen
decrease reabsorption rate. This effect is buffered to
some extent by autoregulatory mechanisms that main-
tain relatively constant renal blood flow as well as
Pif
if
6 mm Hg
relatively constant hydrostatic pressures in the renal
Pc
blood vessels. (2) Increase in resistance of either the
13 mm Hg
πif
if
afferent or the efferent arterioles reduces peritubular
15 mm Hg
πcc
capillary hydrostatic pressure and tends to increase
32 mm Hg
Bulk
reabsorption rate. Although constriction of the
H2O
H2
flow
O efferent arterioles increases glomerular capillary
hydrostatic pressure, it lowers peritubular capillary
Na+
Na+
10 mm Hg
ATP
hydrostatic pressure.
Net reabsorption
The second major determinant of peritubular capil-
pressure
lary reabsorption is the colloid osmotic pressure of the
plasma in these capillaries; raising the colloid osmotic
pressure increases peritubular capillary reabsorption.
Figure 27-15
The colloid osmotic pressure of peritubular capillaries
Summary of the hydrostatic and colloid osmotic forces that deter-
is determined by
(1) the systemic plasma colloid
mine fluid reabsorption by the peritubular capillaries. The numeri-
osmotic pressure; increasing the plasma protein con-
cal values shown are estimates of the normal values for humans. The
centration of systemic blood tends to raise peritubular
net reabsorptive pressure is normally about 10 mm Hg, causing fluid
capillary colloid osmotic pressure, thereby increasing
and solutes to be reabsorbed into the peritubular capillaries as they
are transported across the renal tubular cells. ATP, adenosine
reabsorption; and (2) the filtration fraction; the higher
triphosphate; Pc, peritubular capillary hydrostatic pressure; Pif, inter-
the filtration fraction, the greater the fraction of
, peritubular capillary colloid
stitial fluid hydrostatic pressure; pc
plasma filtered through the glomerulus and, conse-
, interstitial fluid colloid osmotic pressure.
osmotic pressure; pif
quently, the more concentrated the protein becomes in
the plasma that remains behind. Thus, increasing the
filtration fraction also tends to increase the peritubu-
Because the normal peritubular capillary pressure
lar capillary reabsorption rate. Because filtration frac-
averages about 13 mm Hg and renal interstitial fluid
tion is defined as the ratio of GFR/renal plasma flow,
hydrostatic pressure averages 6 mm Hg, there is a pos-
increased filtration fraction can occur as a result of
itive hydrostatic pressure gradient from the peritubu-
increased GFR or decreased renal plasma flow. Some
lar capillary to the interstitial fluid of about 7 mm Hg,
renal vasoconstrictors, such as angiotensin II, increase
which opposes fluid reabsorption. This is more than
peritubular capillary reabsorption by decreasing renal
counterbalanced by the colloid osmotic pressures that
plasma flow and increasing filtration fraction, as dis-
favor reabsorption. The plasma colloid osmotic pres-
cussed later.
sure, which favors reabsorption, is about 32 mm Hg,
Changes in the peritubular capillary Kf can also
and the colloid osmotic pressure of the interstitium,
influence the reabsorption rate because Kf is a
which opposes reabsorption, is 15 mm Hg, causing a
measure of the permeability and surface area of the
net colloid osmotic force of about 17 mm Hg, favoring
capillaries. Increases in Kf raise reabsorption, whereas
reabsorption. Therefore, subtracting the net hydro-
decreases in Kf lower peritubular capillary reabsorp-
static forces that oppose reabsorption (7 mm Hg) from
tion. Kf remains relatively constant in most physiologic
the net colloid osmotic forces that favor reabsorption
conditions. Table 27-2 summarizes the factors that
(17 mm Hg) gives a net reabsorptive force of about
can influence the peritubular capillary reabsorption
10 mm Hg. This is a high value, similar to that found
rate.
in the glomerular capillaries, but in the opposite
direction.
Renal Interstitial Hydrostatic and Colloid Osmotic Pressures.
The other factor that contributes to the high rate of
Ultimately, changes in peritubular capillary physical
fluid reabsorption in the peritubular capillaries is a
forces influence tubular reabsorption by changing the
large filtration coefficient
(Kf) because of the high
physical forces in the renal interstitium surrounding
hydraulic conductivity and large surface area of the
the tubules. For example, a decrease in the reabsorp-
capillaries. Because the reabsorption rate is normally
tive force across the peritubular capillary membranes,
about 124 ml/min and net reabsorption pressure is
caused by either increased peritubular capillary hydro-
10 mm Hg, Kf normally is about 12.4 ml/min/mm Hg.
static pressure or decreased peritubular capillary
colloid osmotic pressure, reduces the uptake of fluid
Regulation of Peritubular Capillary Physical Forces. The two
and solutes from the interstitium into the peritubular
determinants of peritubular capillary reabsorption
capillaries. This in turn raises renal interstitial fluid
that are directly influenced by renal hemodynamic
hydrostatic pressure and decreases interstitial fluid
changes are the hydrostatic and colloid osmotic pres-
colloid osmotic pressure because of dilution of the
sures of the peritubular capillaries. The peritubular
proteins in the renal interstitium. These changes then
capillary hydrostatic pressure is influenced by the arte-
decrease the net reabsorption of fluid from the renal
rial pressure and resistances of the afferent and efferent
tubules into the interstitium, especially in the proximal
arterioles. (1) Increases in arterial pressure tend to
tubules.
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
341
Table 27-2
Normal
Peritubular
Interstitial
Tubular
Lumen
Factors That Can Influence Peritubular Capillary
capillary
fluid
cells
Reabsorption
≠ Pc Æ Ø Reabsorption
Pc
c
• ØRA Æ≠Pc
πc
• ØRE Æ≠Pc
c
• ≠ Arterial Pressure Æ ≠ Pc
≠ pc Æ ≠ Reabsorption
• ≠pA Æ≠pc
ATP
• ≠ FF Æ ≠ pc
Net
≠ Kf Æ ≠ Reabsorption
reabsorption
Pc, peritubular capillary hydrostatic pressure; RA and RE, afferent and effer-
ent arteriolar resistances, respectively; pc, peritubular capillary colloid osmotic
ATP
pressure; pA, arterial plasma colloid osmotic pressure; FF, filtration fraction;
Backleak
Kf, peritubular capillary filtration coefficient.
The mechanisms by which changes in interstitial
Decreased reabsorption
fluid hydrostatic and colloid osmotic pressures influ-
ence tubular reabsorption can be understood by exam-
Pc
ining the pathways through which solute and water are
c
πc
reabsorbed (Figure 27-16). Once the solutes enter the
c
intercellular channels or renal interstitium by active
transport or passive diffusion, water is drawn from the
ATP
tubular lumen into the interstitium by osmosis. And
Decreased net
once the water and solutes are in the interstitial spaces,
reabsorption
they can either be swept up into the peritubular cap-
illaries or diffuse back through the epithelial junctions
ATP
into the tubular lumen. The so-called tight junctions
Increased
between the epithelial cells of the proximal tubule are
backleak
actually leaky, so that considerable amounts of sodium
can diffuse in both directions through these junctions.
With the normal high rate of peritubular capillary
Figure 27-16
reabsorption, the net movement of water and solutes
Proximal tubular and peritubular capillary
reabsorption under
is into the peritubular capillaries with little backleak
normal conditions (top) and during decreased peritubular capillary
into the lumen of the tubule. However, when peri-
reabsorption (bottom) caused by either increasing peritubular cap-
tubular capillary reabsorption is reduced, there is
illary hydrostatic pressure (Pc) or decreasing peritubular capillary
increased interstitial fluid hydrostatic pressure and a
colloid osmotic pressure (pc). Reduced peritubular capillary reab-
sorption, in turn, decreases the net reabsorption of solutes and water
tendency for greater amounts of solute and water to
by increasing the amounts of solutes and water that leak back into
backleak into the tubular lumen, thereby reducing the
the tubular lumen through the tight junctions of the tubular epithe-
rate of net reabsorption (refer again to Figure 27-16).
lial cells, especially in the proximal tubule.
The opposite is true when there is increased peri-
tubular capillary reabsorption above the normal level.
An initial increase in reabsorption by the peritubular
Effect of Arterial Pressure on Urine
capillaries tends to reduce interstitial fluid hydrostatic
pressure and raise interstitial fluid colloid osmotic
Output—The Pressure-Natriuresis and
pressure. Both of these forces favor movement of fluid
Pressure-Diuresis Mechanisms
and solutes out of the tubular lumen and into the inter-
stitium; therefore, backleak of water and solutes into
Even small increases in arterial pressure often cause
the tubular lumen is reduced, and net tubular reab-
marked increases in urinary excretion of sodium and
sorption increases.
water, phenomena that are referred to as pressure
Thus, through changes in the hydrostatic and colloid
natriuresis and pressure diuresis. Because of the
osmotic pressures of the renal interstitium, the uptake
autoregulatory mechanisms described in Chapter 26,
of water and solutes by the peritubular capillaries is
increasing the arterial pressure between the limits of
closely matched to the net reabsorption of water and
75 and 160 mm Hg usually has only a small effect on
solutes from the tubular lumen into the interstitium.
renal blood flow and GFR. The slight increase in GFR
Therefore, in general, forces that increase peritubular
that does occur contributes in part to the effect of
capillary reabsorption also increase reabsorption from
increased arterial pressure on urine output. When
the renal tubules. Conversely, hemodynamic changes
GFR autoregulation is impaired, as often occurs in
that inhibit peritubular capillary reabsorption also
kidney disease, increases in arterial pressure cause
inhibit tubular reabsorption of water and solutes.
much larger increases in GFR.
342
Unit V The Body Fluids and Kidneys
A second effect of increased renal arterial pressure
Aldosterone Increases Sodium Reabsorption and Increases
that raises urine output is that it decreases the per-
Potassium Secretion. Aldosterone, secreted by the zona
centage of the filtered load of sodium and water that
glomerulosa cells of the adrenal cortex, is an impor-
is reabsorbed by the tubules. The mechanisms respon-
tant regulator of sodium reabsorption and potassium
sible for this effect include a slight increase in peri-
secretion by the renal tubules. The primary site
tubular capillary hydrostatic pressure, especially in the
of aldosterone action is on the principal cells of the
vasa recta of the renal medulla, and a subsequent
cortical collecting tubule. The mechanism by which
increase in the renal interstitial fluid hydrostatic pres-
aldosterone increases sodium reabsorption while at
sure. As discussed earlier, an increase in the renal
the same time increasing potassium secretion is by
interstitial fluid hydrostatic pressure enhances back-
stimulating the sodium-potassium ATPase pump on
leak of sodium into the tubular lumen, thereby reduc-
the basolateral side of the cortical collecting tubule
ing the net reabsorption of sodium and water and
membrane. Aldosterone also increases the sodium
further increasing the rate of urine output when renal
permeability of the luminal side of the membrane.
arterial pressure rises.
The cellular mechanisms of aldosterone action are
A third factor that contributes to the pressure-
discussed in Chapter 77.
natriuresis and pressure-diuresis mechanisms is
In the absence of aldosterone, as occurs with adrenal
reduced angiotensin II formation. Angiotensin II itself
destruction or malfunction (Addison’s disease), there
increases sodium reabsorption by the tubules; it also
is marked loss of sodium from the body and accumu-
stimulates aldosterone secretion, which further
lation of potassium. Conversely, excess aldosterone
increases sodium reabsorption. Therefore, decreased
secretion, as occurs in patients with adrenal tumors
angiotensin II formation contributes to the decreased
(Conn’s syndrome) is associated with sodium retention
tubular sodium reabsorption that occurs when arterial
and potassium depletion. Although day-to-day regula-
pressure is increased.
tion of sodium balance can be maintained as long as
minimal levels of aldosterone are present, the inabil-
ity to appropriately adjust aldosterone secretion
Hormonal Control of Tubular
greatly impairs the regulation of renal potassium
Reabsorption
excretion and potassium concentration of the body
fluids. Thus, aldosterone is even more important as a
Precise regulation of body fluid volumes and solute
regulator of potassium concentration than it is for
concentrations requires the kidneys to excrete differ-
sodium concentration.
ent solutes and water at variable rates, sometimes
independently of one another. For example, when
Angiotensin II Increases Sodium and Water Reabsorption.
potassium intake is increased, the kidneys must
Angiotensin II is perhaps the body’s most powerful
excrete more potassium while maintaining normal
sodium-retaining hormone. As discussed in Chapter
excretion of sodium and other electrolytes. Likewise,
19, angiotensin II formation increases in circumstances
when sodium intake is changed, the kidneys must
associated with low blood pressure and/or low extra-
appropriately adjust urinary sodium excretion without
cellular fluid volume, such as during hemorrhage or
major changes in excretion of other electrolytes.
loss of salt and water from the body fluids. The
Several hormones in the body provide this specificity
increased formation of angiotensin II helps to return
of tubular reabsorption for different electrolytes and
blood pressure and extracellular volume toward
water. Table 27-3 summarizes some of the most impor-
normal by increasing sodium and water reabsorption
tant hormones for regulating tubular reabsorption,
from the renal tubules through three main effects:
their principal sites of action on the renal tubule, and
1. Angiotensin II stimulates aldosterone secretion,
their effects on solute and water excretion. Some
which in turn increases sodium reabsorption.
of these hormones are discussed in more detail in
2. Angiotensin II constricts the efferent arterioles,
Chapters 28 and 29, but we briefly review their renal
which has two effects on peritubular capillary
tubular actions in the next few paragraphs.
dynamics that raise sodium and water
Table 27-3
Hormones That Regulate Tubular Reabsorption
Hormone
Site of Action
Effects
Aldosterone
Collecting tubule and duct
≠ NaCl, H2O reabsorption, ≠ K+ secretion
Angiotensin II
Proximal tubule, thick ascending loop of Henle/distal
≠ NaCl, H2O reabsorption, ≠ H+ secretion
tubule, collecting tubule
Antidiuretic hormone
Distal tubule/collecting tubule and duct
≠ H2O reabsorption
Atrial natriuretic peptide
Distal tubule/collecting tubule and duct
Ø NaCl reabsorption
Parathyroid hormone
Proximal tubule, thick ascending loop of
Ø PO4- - - reabsorption, ≠ Ca++ reabsorption
Henle/distal tubule
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
343
reabsorption. First, efferent arteriolar constriction
distended because of plasma volume expansion,
reduces peritubular capillary hydrostatic pressure,
secrete a peptide called atrial natriuretic peptide.
which increases net tubular reabsorption,
Increased levels of this peptide in turn inhibit the reab-
especially from the proximal tubules. Second,
sorption of sodium and water by the renal tubules,
efferent arteriolar constriction, by reducing renal
especially in the collecting ducts. This decreased
blood flow, raises filtration fraction in the
sodium and water reabsorption increases urinary
glomerulus and increases the concentration of
excretion, which helps to return blood volume back
proteins and the colloid osmotic pressure in
toward normal.
the peritubular capillaries; this increases the
reabsorptive force at the peritubular capillaries
Parathyroid Hormone Increases Calcium Reabsorption.
and raises tubular reabsorption of sodium and
Parathyroid hormone is one of the most important
water.
calcium-regulating hormones in the body. Its principal
3. Angiotensin II directly stimulates sodium
action in the kidneys is to increase tubular reabsorp-
reabsorption in the proximal tubules, the loops of
tion of calcium, especially in the distal tubules and
Henle, the distal tubules, and the collecting tubules.
perhaps also in the loops of Henle. Parathyroid
One of the direct effects of angiotensin II is to
hormone also has other actions, including inhibition of
stimulate the sodium-potassium ATPase pump on
phosphate reabsorption by the proximal tubule and
the tubular epithelial cell basolateral membrane.
stimulation of magnesium reabsorption by the loop of
A second effect is to stimulate sodium-hydrogen
Henle, as discussed in Chapter 29.
exchange in the luminal membrane, especially
in the proximal tubule. Thus, angiotensin II
Sympathetic Nervous System
stimulates sodium transport across both the
luminal and the basolateral surfaces of the
Activation Increases Sodium
epithelial cell membrane in the tubules.
Reabsorption
These multiple actions of angiotensin II cause
marked sodium retention by the kidneys when
Activation of the sympathetic nervous system can
angiotensin II levels are increased.
decrease sodium and water excretion by constricting
the renal arterioles, thereby reducing GFR. Sympa-
ADH Increases Water Reabsorption. The most important
thetic activation also increases sodium reabsorption in
renal action of ADH is to increase the water perme-
the proximal tubule, the thick ascending limb of the
ability of the distal tubule, collecting tubule, and col-
loop of Henle, and perhaps in more distal parts of the
lecting duct epithelia. This effect helps the body to
renal tubule. And finally, sympathetic nervous system
conserve water in circumstances such as dehydration.
stimulation increases renin release and angiotensin II
In the absence of ADH, the permeability of the distal
formation, which adds to the overall effect to increase
tubules and collecting ducts to water is low, causing the
tubular reabsorption and decrease renal excretion of
kidneys to excrete large amounts of dilute urine. Thus,
sodium.
the actions of ADH play a key role in controlling the
degree of dilution or concentration of the urine, as dis-
Use of Clearance Methods
cussed further in Chapters 28 and 75.
ADH binds to specific V2 receptors in the late distal
to Quantify Kidney Function
tubules, collecting tubules, and collecting ducts,
increasing the formation of cyclic AMP and activating
The rates at which different substances are “cleared”
protein kinases. This, in turn, stimulates the movement
from the plasma provide a useful way of quantitating
of an intracellular protein, called aquaporin-2 (AQP-
the effectiveness with which the kidneys excrete
2), to the luminal side of the cell membranes. The mol-
various substances (Table 27-4). By definition, the
ecules of AQP-2 cluster together and fuse with the cell
renal clearance of a substance is the volume of plasma
membrane by exocytosis to form water channels that
that is completely cleared of the substance by the
permit rapid diffusion of water through the cells. There
kidneys per unit time. This concept is somewhat
are other aquaporins, AQP-3 and AQP-4, in the baso-
abstract because there is no single volume of plasma
lateral side of the cell membrane that provide a path
that is completely cleared of a substance. However,
for water to rapidly exit the cells, although these
renal clearance provides a useful way of quantifying
are not believed to be regulated by ADH. Chronic
the excretory function of the kidneys and, as discussed
increases in ADH levels also increase the formation of
later, can be used to quantify the rate at which blood
AQP-2 protein in the renal tubular cells by stimulat-
flows through the kidneys as well as the basic functions
ing AQP-2 gene transcription. When the concentration
of the kidneys: glomerular filtration, tubular reabsorp-
of ADH decreases, the molecules of AQP-2 are shut-
tion, and tubular secretion.
tled back to the cell cytoplasm, thereby removing the
To illustrate the clearance principle, consider the fol-
water channels from the luminal membrane and
lowing example: If the plasma passing through the
reducing water permeability.
kidneys contains 1 milligram of a substance in each mil-
liliter and if
1 milligram of this substance is also
Atrial Natriuretic Peptide Decreases Sodium and Water
excreted into the urine each minute, then 1 ml/min
Reabsorption. Specific cells of the cardiac atria, when
of the plasma is “cleared” of the substance. Thus,
344
Unit V The Body Fluids and Kidneys
Table 27-4
Use of Clearance to Quantify Kidney Function
Term
Equation
Units
s
¥
V
Clearance rate (Cs)
s
C =U
ml/min
P
s
U
inulin
¥
V
Glomerular filtration rate (GFR)
GFR
=
P
inulin
Cs
Clearance ratio
Clearance ratio
=
None
Cinulin
UPAH¥V
Effective renal plasma flow (ERPF)
ERPF =C
PAH
=
ml/min
P
PAH
C
PAH
(UPAH¥V
P
PAH
)
Renal plasma flow (RPF)
RPF
=
=
ml/min
E
PAH
(P
PAH
-
V
PAH
)
P
PAH
UPAH¥V
PAH
=
P
PAH
-
V
PAH
RPF
Renal blood flow (RBF)
RBF
=
ml/min
1-Hematocrit
Excretion rate
Excretion rate = Us ¥ V
mg/min, mmol/min, or mEq/min
Reabsorption rate
Reabsorption rate = Filtered load - Excretion rate
mg/min, mmol/min, or mEq/min
= (GFR ¥ Ps) - (Us ¥ V)
Secretion rate
Secretion rate = Excretion rate - Filtered load
mg/min, mmol/min, or mEq/min
S, a substance; U, urine concentration; V, urine flow rate; P, plasma concentration; PAH, para-aminohippuric acid; PPAH, renal arterial PAH concentration; EPAH, PAH
extraction ratio; VPAH, renal venous PAH concentration.
clearance refers to the volume of plasma that would be
5200. Inulin, which is not produced in the body, is found
necessary to supply the amount of substance excreted
in the roots of certain plants and must be administered
intravenously to a patient to measure GFR.
in the urine per unit time. Stated mathematically,
Figure 27-17 shows the renal handling of inulin. In
Cs ¥ Ps = Us ¥ V,
this example, the plasma concentration is 1 mg/ml, urine
concentration is 125 mg/ml, and urine flow rate is 1 ml/
where Cs is the clearance rate of a substance s, Ps is the
min. Therefore, 125 mg/min of inulin passes into the
plasma concentration of the substance, Us is the urine
urine. Then, inulin clearance is calculated as the urine
concentration of that substance, and V is the urine flow
excretion rate of inulin divided by the plasma concen-
rate. Rearranging this equation, clearance can be
tration, which yields a value of 125 ml/min. Thus, 125
expressed as
milliliters of plasma flowing through the kidneys must
s
¥
V
be filtered to deliver the inulin that appears in the urine.
C =U
s
P
Inulin is not the only substance that can be used for
s
determining GFR. Other substances that have been
Thus, renal clearance of a substance is calculated from
used clinically to estimate GFR include radioactive
the urinary excretion rate (Us ¥ V) of that substance
iothalamate and creatinine.
divided by its plasma concentration.
Inulin Clearance Can Be Used
Creatinine Clearance and Plasma
to Estimate GFR
Creatinine Concentration Can Be
Used to Estimate GFR
If a substance is freely filtered (filtered as freely as
water) and is not reabsorbed or secreted by the renal
Creatinine is a by-product of muscle metabolism and is
tubules, then the rate at which that substance is excreted
cleared from the body fluids almost entirely by glomeru-
in the urine (Us ¥ V) is equal to the filtration rate of the
lar filtration. Therefore, the clearance of creatinine can
substance by the kidneys (GFR ¥ Ps). Thus,
also be used to assess GFR. Because measurement of
creatinine clearance does not require intravenous infu-
GFR ¥ Ps = Us ¥ V
sion into the patient, this method is much more widely
The GFR, therefore, can be calculated as the clearance
used than inulin clearance for estimating GFR clinically.
of the substance as follows:
However, creatinine clearance is not a perfect marker
Us¥V
of GFR because a small amount of it is secreted by the
GFR
=
=
C
s
tubules, so that the amount of creatinine excreted
P
s
slightly exceeds the amount filtered. There is normally
A substance that fits these criteria is inulin, a polysac-
a slight error in measuring plasma creatinine that
charide molecule with a molecular weight of about
leads to an overestimate of the plasma creatinine
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
345
Pinulin = 1 mg/ml
100
Amount filtered = Amount excreted
50
GFR x Pinulin = Uinulin x V
0
Uinulin x V
GFR =
Pinulin
GFR = 125 ml/min
2
1
Uinulin = 125 mg/ml
0
V = 1 ml/min
Positive balance
Production
Figure 27-17
2
Measurement of glomerular filtration rate (GFR) from the renal
clearance of inulin. Inulin is freely filtered by the glomerular cap-
Excretion GFR x PCreatinine
illaries but is not reabsorbed by the renal tubules. Pinulin, plasma
1
inulin concentration; Uinulin, urine inulin concentration; V, urine flow
rate.
concentration, and fortuitously, these two errors tend to
0
cancel each other. Therefore, creatinine clearance pro-
0
1
2
3
4
vides a reasonable estimate of GFR.
Days
In some cases, it may not be practical to collect urine
in a patient for measuring creatinine clearance (CCr). An
approximation of changes in GFR, however, can be
obtained by simply measuring plasma creatinine con-
Figure 27-18
centration (PCr), which is inversely proportional to GFR:
UCr¥V
Effect of reducing glomerular filtration rate (GRF) by 50 per cent
GFRªC
Cr
=
on serum creatinine concentration and on creatinine excretion
P
Cr
rate when the production rate of creatinine remains constant.
PCreatinine, plasma creatinine concentration.
If GFR suddenly decreases by 50%, the kidneys will
transiently filter and excrete only half as much creati-
nine, causing accumulation of creatinine in the body
fluids and raising plasma concentration. Plasma con-
to the total renal plasma flow. In other words, the
centration of creatinine will continue to rise until the fil-
amount of the substance delivered to the kidneys in
tered load of creatinine (PCr ¥ GFR) and creatinine
the blood (renal plasma flow ¥ Ps) would be equal to
excretion (UCr ¥ V) return to normal and a balance
the amount excreted in the urine (Us ¥ V). Thus, renal
between creatinine production and creatinine excretion
plasma flow (RPF) could be calculated as
is reestablished. This will occur when plasma creatinine
increases to approximately twice normal, as shown in
Us¥V
RPF
=
=
Cs
Figure 27-18. If GFR falls to one-fourth normal, plasma
Ps
creatinine would increase to about 4 times normal, and
a decrease of GFR to one-eighth normal would raise
Because the GFR is only about 20 per cent of the
plasma creatinine to 8 times normal. Thus, under steady-
total plasma flow, a substance that is completely cleared
state conditions, the creatinine excretion rate equals the
from the plasma must be excreted by tubular secretion
rate of creatinine production, despite reductions in
as well as glomerular filtration (Figure 27-20). There is
GFR. However, this normal rate of creatinine excretion
no known substance that is completely cleared by the
occurs at the expense of elevated plasma creatinine con-
kidneys. One substance, however, PAH, is about 90 per
centration, as shown in Figure 27-19.
cent cleared from the plasma. Therefore, the clearance
of PAH can be used as an approximation of renal
plasma flow. To be more accurate, one can correct for
PAH Clearance Can Be Used to
the percentage of PAH that is still in the blood when it
Estimate Renal Plasma Flow
leaves the kidneys. The percentage of PAH removed
from the blood is known as the extraction ratio of PAH
Theoretically, if a substance is completely cleared from
and averages about 90 per cent in normal kidneys. In
the plasma, the clearance rate of that substance is equal
diseased kidneys, this extraction ratio may be reduced
346
Unit V The Body Fluids and Kidneys
PPAH = 0.01 mg/ml
14
12
10
Renal plasma flow
8
UPAH x V
=
P
PAH
6
4
Renal venous
PAH =
2
Normal
0.001 mg/ml
UPAH = 5.85 mg/ml
25
50
75
100
125
150
V = 1 ml/min
Glomerular filtration rate
(ml/min)
Figure 27-20
Measurement of renal plasma flow from the renal clearance of para-
aminohippuric acid (PAH). PAH is freely filtered by the glomerular
Figure 27-19
capillaries and is also secreted from the peritubular capillary blood
into the tubular lumen. The amount of PAH in the plasma of the
Approximate relationship between glomerular filtration rate (GFR)
renal artery is about equal to the amount of PAH excreted in
and plasma creatinine concentration under steady-state conditions.
the urine. Therefore, the renal plasma flow can be calculated from
Decreasing GFR by 50 per cent will increase plasma creatinine to
the clearance of PAH (CPAH). To be more accurate, one can correct
twice normal if creatinine production by the body remains constant.
for the percentage of PAH that is still in the blood when it leaves
the kidneys. PPAH, arterial plasma PAH concentration; UPAH, urine
PAH concentration; V, urine flow rate.
because of inability of damaged tubules to secrete PAH
Filtration Fraction Is Calculated
into the tubular fluid.
from GFR Divided by Renal
The calculation of RPF can be demonstrated by the
Plasma Flow
following example: Assume that the plasma concentra-
tion of PAH is 0.01 mg/ml, urine concentration is 5.85
To calculate the filtration fraction, which is the fraction
mg/ml, and urine flow rate is 1 ml/min. PAH clearance
of plasma that filters through the glomerular mem-
can be calculated from the rate of urinary PAH excre-
brane, one must first know the renal plasma flow (PAH
tion (5.85 mg/ml ¥ 1 ml/min) divided by the plasma PAH
clearance) and the GFR (inulin clearance). If renal
concentration (0.01 mg/ml). Thus, clearance of PAH cal-
plasma flow is 650 ml/min and GFR is 125 ml/min, the
culates to be 585 ml/min.
filtration fraction (FF) is calculated as
If the extraction ratio for PAH is 90 per cent, the
actual renal plasma flow can be calculated by dividing
FF = GFR/RPF = 125/650 = 0.19
585 ml/min by 0.9, yielding a value of 650 ml/min. Thus,
total renal plasma flow can be calculated as
Calculation of Tubular Reabsorption
Total renal plasma flow =
or Secretion from Renal Clearances
Clearance of PAH/Extraction ratio of PAH
If the rates of glomerular filtration and renal excretion
The extraction ratio (EPAH) is calculated as the dif-
of a substance are known, one can calculate whether
ference between the renal arterial PAH (PPAH) and renal
there is a net reabsorption or a net secretion of that sub-
venous PAH (VPAH) concentrations, divided by the renal
stance by the renal tubules. For example, if the rate of
arterial PAH concentration:
excretion of the substance (Us ¥ V) is less than the fil-
P
PAH
-V
PAH
tered load of the substance (GFR ¥ Ps), then some of
E
PAH
=
the substance must have been reabsorbed from the
P
PAH
renal tubules. Conversely, if the excretion rate of the
One can calculate the total blood flow through the
substance is greater than its filtered load, then the rate
kidneys from the total renal plasma flow and hematocrit
at which it appears in the urine represents the sum of
(the percentage of red blood cells in the blood). If the
the rate of glomerular filtration plus tubular secretion.
hematocrit is 0.45 and the total renal plasma flow is
The following example demonstrates the calculation
650 ml/min, the total blood flow through both kidneys
of tubular reabsorption. Assume the following labora-
is 650/(1 - 0.45), or 1182 ml/min.
tory values for a patient were obtained:
Chapter 27
Urine Formation by the Kidneys: II. Tubular Processing of the Glomerular Filtrate
347
Urine flow rate = 1 ml/min
Féraille E, Doucet A: Sodium-potassium-adenosine-
Urine concentration of sodium (UNa) = 70 mEq/L
triphosphatase-dependent sodium transport in the
= 70 mEq/ml
kidney: Hormonal control. Physiol Rev 81:345, 2001.
Plasma sodium concentration = 140 mEq/L
Granger JP, Alexander BT, Llinas M: Mechanisms of pres-
= 140 mEq/ml
sure natriuresis. Curr Hypertens Rep 4:152, 2002.
GFR (inulin clearance) = 100 ml/min
Hall JE, Brands MW: The renin-angiotensin-aldosterone
system: renal mechanisms and circulatory homeostasis.
In this example, the filtered sodium load is GFR ¥ PNa,
In Seldin DW, Giebisch G (eds): The Kidney—Physiology
or 100 ml/min ¥ 140 mEq/ml = 14,000 mEq/min. Urinary
and Pathophysiology, 3rd ed. New York: Raven Press,
sodium excretion (UNa ¥ urine flow rate) is 70 mEq/min.
2000.
Therefore, tubular reabsorption of sodium is the differ-
Humphreys MH, Valentin J-P: Natriuretic hormonal agents.
ence between the filtered load and urinary excretion, or
In Seldin DW, Giebisch G (eds): The Kidney—Physiology
14,000 mEq/min - 70 mEq/min = 13,930 mEq/min.
and Pathophysiology, 3rd ed. New York: Raven Press,
2000.
Comparisons of Inulin Clearance with Clearances of Different
Kellenberger S, Schild L: Epithelial sodium channel/
Solutes. The following generalizations can be made by
degenerin family of ion channels: A variety of functions
comparing the clearance of a substance with the clear-
for a shared structure. Physiol Rev 82:735, 2002.
ance of inulin, a measure of GFR: (1) if the clearance
Nielsen S, Frøkiær J, Marples D, et al: Aquaporins in the
rate of the substance equals that of inulin, the substance
kidney: from molecules to medicine. Physiol Rev 82:205,
is only filtered and not reabsorbed or secreted; (2) if the
2002.
clearance rate of a substance is less than inulin clear-
Palmer LG, Frindt G: Aldosterone and potassium secretion
ance, the substance must have been reabsorbed by the
by the cortical collecting duct. Kidney Int 57:1324, 2000.
nephron tubules; and (3) if the clearance rate of a sub-
Rahn KH, Heidenreich S, Bruckner D: How to assess
stance is greater than that of inulin, the substance must
glomerular function and damage in humans. J Hypertens
be secreted by the nephron tubules. Listed below are
17:309, 1999.
the approximate clearance rates for some of the sub-
Reeves WB, Andreoli TE: Sodium chloride transport in the
stances normally handled by the kidneys:
loop of Henle, distal convoluted tubule and collecting
duct. In Seldin DW, Giebisch G (eds): The Kidney—Phys-
Substance
Clearance Rate (ml/min)
iology and Pathophysiology, 3rd ed. New York: Raven
Press, 2000.
Glucose
0
Reilly RF, Ellison DH: Mammalian distal tubule: physiology,
Sodium
0.9
pathophysiology, and molecular anatomy. Physiol Rev
Chloride
1.3
80:277, 2000.
Potassium
12.0
Rossier BC, Praderv S, Schild L, Hummler E: Epithelial
Phosphate
25.0
sodium channel and the control of sodium balance: inter-
Inulin
125.0
Creatinine
140.0
action between genetic and environmental factors. Annu
Rev Physiol 64:877, 2002.
Russell JM: Sodium-potassium-chloride cotransport. Physiol
Rev 80:211, 2000.
Schafer JA: Abnormal regulation of ENaC: syndromes of
References
salt retention and salt wasting by the collecting duct. Am
J Physiol Renal Physiol 283:F221, 2002.
Aronson PS: Ion exchangers mediating NaCl transport in the
Weinstein AM: Mathematical models of renal fluid and elec-
renal proximal tubule. Cell Biochem Biophys 36:147, 2002.
trolyte transport: acknowledging our uncertainty. Am J
Benos DJ, Fuller CM, Shlyonsky VG, et al: Amiloride-
Physiol Renal Physiol 284:F871, 2003.
sensitive Na+ channels: insights and outlooks. News
Wright EM: Renal Na(+)-glucose cotransporters. Am J
Physiol Sci 12:55, 1997.
Physiol Renal Physiol 280:F10, 2001.
C H A P T E R
2
8
Regulation of Extracellular Fluid
Osmolarity and Sodium
Concentration
For the cells of the body to function properly, they
must be bathed in extracellular fluid with a rela-
tively constant concentration of electrolytes and
other solutes. The total concentration of solutes in
the extracellular fluid—and therefore the osmolar-
ity—is determined by the amount of solute divided
by the volume of the extracellular fluid. Thus, to a
large extent, extracellular fluid sodium concentra-
tion and osmolarity are regulated by the amount of extracellular water. The
body water in turn is controlled by (1) fluid intake, which is regulated by factors
that determine thirst, and (2) renal excretion of water, which is controlled by
multiple factors that influence glomerular filtration and tubular reabsorption.
In this chapter, we discuss specifically (1) the mechanisms that cause the
kidneys to eliminate excess water by excreting a dilute urine; (2) the mecha-
nisms that cause the kidneys to conserve water by excreting a concentrated
urine; (3) the renal feedback mechanisms that control the extracellular fluid
sodium concentration and osmolarity; and (4) the thirst and salt appetite mech-
anisms that determine the intakes of water and salt, which also help to control
extracellular fluid volume, osmolarity, and sodium concentration.
The Kidneys Excrete Excess Water by Forming
a Dilute Urine
The normal kidney has tremendous capability to vary the relative proportions
of solutes and water in the urine in response to various challenges. When there
is excess water in the body and body fluid osmolarity is reduced, the kidney can
excrete urine with an osmolarity as low as 50 mOsm/L, a concentration that is
only about one sixth the osmolarity of normal extracellular fluid. Conversely,
when there is a deficit of water and extracellular fluid osmolarity is high, the
kidney can excrete urine with a concentration of 1200 to 1400 mOsm/L. Equally
important, the kidney can excrete a large volume of dilute urine or a small
volume of concentrated urine without major changes in rates of excretion of
solutes such as sodium and potassium. This ability to regulate water excretion
independently of solute excretion is necessary for survival, especially when fluid
intake is limited.
Antidiuretic Hormone Controls Urine Concentration
There is a powerful feedback system for regulating plasma osmolarity and
sodium concentration that operates by altering renal excretion of water inde-
pendently of the rate of solute excretion. A primary effector of this feedback is
antidiuretic hormone (ADH), also called vasopressin.
When osmolarity of the body fluids increases above normal (that is, the
solutes in the body fluids become too concentrated), the posterior pituitary
gland secretes more ADH, which increases the permeability of the distal tubules
and collecting ducts to water, as discussed in Chapter 27. This allows large
amounts of water to be reabsorbed and decreases urine volume but does not
348
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
349
markedly alter the rate of renal excretion of the
However, the total amount of solute excreted remains
solutes.
relatively constant because the urine formed becomes
When there is excess water in the body and extra-
very dilute and urine osmolarity decreases from 600 to
cellular fluid osmolarity is reduced, the secretion of
about 100 mOsm/L. Thus, after ingestion of excess
ADH by the posterior pituitary decreases, thereby
water, the kidney rids the body of the excess water but
reducing the permeability of the distal tubule and col-
does not excrete excess amounts of solutes.
lecting ducts to water, which causes large amounts of
When the glomerular filtrate is initially formed,
dilute urine to be excreted. Thus, the rate of ADH
its osmolarity is about the same as that of plasma
secretion determines, to a large extent, whether the
(300 mOsm/L). To excrete excess water, it is necessary
kidney excretes a dilute or a concentrated urine.
to dilute the filtrate as it passes along the tubule. This
is achieved by reabsorbing solutes to a greater extent
than water, as shown in Figure 28-2, but this occurs
Renal Mechanisms for Excreting a
only in certain segments of the tubular system as
Dilute Urine
follows.
When there is a large excess of water in the body, the
Tubular Fluid Remains Isosmotic in the Proximal Tubule. As
kidney can excrete as much as 20 L/day of dilute urine,
fluid flows through the proximal tubule, solutes and
with a concentration as low as 50 mOsm/L. The kidney
water are reabsorbed in equal proportions, so that
performs this impressive feat by continuing to reab-
little change in osmolarity occurs; that is, the proximal
sorb solutes while failing to reabsorb large amounts of
tubule fluid remains isosmotic to the plasma, with an
water in the distal parts of the nephron, including the
osmolarity of about 300 mOsm/L. As fluid passes down
late distal tubule and the collecting ducts.
the descending loop of Henle, water is reabsorbed by
Figure 28-1 shows the approximate renal responses
osmosis and the tubular fluid reaches equilibrium with
in a human after ingestion of 1 liter of water. Note that
the surrounding interstitial fluid of the renal medulla,
urine volume increases to about six times normal
which is very hypertonic—about two to four times the
within 45 minutes after the water has been drunk.
osmolarity of the original glomerular filtrate. There-
fore, the tubular fluid becomes more concentrated as
it flows into the inner medulla.
Tubular Fluid Becomes Dilute in the Ascending Loop of Henle.
In the ascending limb of the loop of Henle, especially
Drink 1.0 L H2O
in the thick segment, sodium, potassium, and chloride
800
are avidly reabsorbed. However, this portion of the
Urine
osmolarity
tubular segment is impermeable to water, even in
400
Plasma
osmolarity
NaCl
H2O
NaCl
0
300
300
6
100
100
300
4
NaCl
2
0
1.2
400
70
400
400
NaCl
0.6
O
H2
NaCl
0
0
60
120
180
600
600
600
50
Time (minutes)
Figure 28-2
Figure 28-1
Formation of a dilute urine when antidiuretic hormone (ADH) levels
Water diuresis in a human after ingestion of 1 liter of water. Note
are very low. Note that in the ascending loop of Henle, the tubular
that after water ingestion, urine volume increases and urine osmo-
fluid becomes very dilute. In the distal tubules and collecting
larity decreases, causing the excretion of a large volume of dilute
tubules, the tubular fluid is further diluted by the reabsorption of
urine; however, the total amount of solute excreted by the kidneys
sodium chloride and the failure to reabsorb water when ADH
remains relatively constant. These responses of the kidneys
levels are very low. The failure to reabsorb water and continued
prevent plasma osmolarity from decreasing markedly during
reabsorption of solutes lead to a large volume of dilute urine.
excess water ingestion.
(Numerical values are in milliosmoles per liter.)
350
Unit V The Body Fluids and Kidneys
the presence of large amounts of ADH. Therefore, the
water produced in the body by metabolism of the food.
tubular fluid becomes more dilute as it flows up the
Animals adapted to aquatic environments, such as the
ascending loop of Henle into the early distal tubule,
beaver, have minimal urine concentrating ability; they
with the osmolarity decreasing progressively to about
can concentrate the urine to only about 500 mOsm/L.
100 mOsm/L by the time the fluid enters the early
distal tubular segment. Thus, regardless of whether
ADH is present or absent, fluid leaving the early distal
Obligatory Urine Volume
tubular segment is hypo-osmotic, with an osmolarity of
The maximal concentrating ability of the kidney dic-
only about one third the osmolarity of plasma.
tates how much urine volume must be excreted each
day to rid the body of waste products of metabolism and
Tubular Fluid in Distal and Collecting Tubules Is Further Diluted
ions that are ingested. A normal 70-kilogram human
in the Absence of ADH. As the dilute fluid in the early
must excrete about 600 milliosmoles of solute each day.
distal tubule passes into the late distal convoluted
If maximal urine concentrating ability is 1200 mOsm/L,
tubule, cortical collecting duct, and collecting duct,
the minimal volume of urine that must be excreted,
called the obligatory urine volume, can be calculated as
there is additional reabsorption of sodium chloride. In
the absence of ADH, this portion of the tubule is also
600 mOsm day
impermeable to water, and the additional reabsorption
= 0.5 L day
1200 mOsm L
of solutes causes the tubular fluid to become even
more dilute, decreasing its osmolarity to as low as
This minimal loss of volume in the urine contributes to
50 mOsm/L. The failure to reabsorb water and the con-
dehydration, along with water loss from the skin, respi-
ratory tract, and gastrointestinal tract, when water is not
tinued reabsorption of solutes lead to a large volume
available to drink.
of dilute urine.
The limited ability of the human kidney to con-
To summarize, the mechanism for forming a dilute
centrate the urine to a maximal concentration of
urine is to continue reabsorbing solutes from the distal
1200 mOsm/L explains why severe dehydration occurs
segments of the tubular system while failing to reab-
if one attempts to drink seawater. Sodium chloride
sorb water. In healthy kidneys, fluid leaving the
concentration in the oceans averages about 3.0 to 3.5
ascending loop of Henle and early distal tubule is
per cent, with an osmolarity between about 1000 and
always dilute, regardless of the level of ADH. In the
1200 mOsm/L. Drinking 1 liter of seawater with a con-
absence of ADH, the urine is further diluted in the late
centration of
1200 mOsm/L would provide a total
distal tubule and collecting ducts, and a large volume
sodium chloride intake of 1200 milliosmoles. If maximal
urine concentrating ability is 1200 mOsm/L, the amount
of dilute urine is excreted.
of urine volume needed to excrete 1200 milliosmoles
would be 1200 milliosmoles divided by 1200 mOsm/L,
or 1.0 liter. Why then does drinking seawater cause
The Kidneys Conserve Water
dehydration? The answer is that the kidney must also
by Excreting a Concentrated
excrete other solutes, especially urea, which contribute
about 600 mOsm/L when the urine is maximally con-
Urine
centrated. Therefore, the maximum concentration of
sodium chloride that can be excreted by the kidneys is
The ability of the kidney to form a urine that is more
about 600 mOsm/L. Thus, for every liter of seawater
concentrated than plasma is essential for survival of
drunk, 2 liters of urine volume would be required to rid
mammals that live on land, including humans. Water is
the body of
1200 milliosmoles of sodium chloride
continuously lost from the body through various
ingested in addition to other solutes such as urea. This
routes, including the lungs by evaporation into the
would result in a net fluid loss of 1 liter for every liter
of seawater drunk, explaining the rapid dehydration
expired air, the gastrointestinal tract by way of the
that occurs in shipwreck victims who drink seawater.
feces, the skin through evaporation and perspiration,
However, a shipwreck victim’s pet Australian hopping
and the kidneys through the excretion of urine. Fluid
mouse could drink with impunity all the seawater it
intake is required to match this loss, but the ability of
wanted.
the kidney to form a small volume of concentrated
urine minimizes the intake of fluid required to main-
tain homeostasis, a function that is especially impor-
Requirements for Excreting a
tant when water is in short supply.
When there is a water deficit in the body, the kidney
Concentrated Urine—High ADH Levels
forms a concentrated urine by continuing to excrete
and Hyperosmotic Renal Medulla
solutes while increasing water reabsorption and
decreasing the volume of urine formed. The human
The basic requirements for forming a concentrated
kidney can produce a maximal urine concentration of
urine are (1) a high level of ADH, which increases the
1200 to 1400 mOsm/L, four to five times the osmolar-
permeability of the distal tubules and collecting ducts
ity of plasma. Some desert animals, such as the Aus-
to water, thereby allowing these tubular segments to
tralian hopping mouse, can concentrate urine to as
avidly reabsorb water, and (2) a high osmolarity of the
high as 10,000 mOsm/L. This allows the mouse to
renal medullary interstitial fluid, which provides the
survive in the desert without drinking water; sufficient
osmotic gradient necessary for water reabsorption to
water can be obtained through the food ingested and
occur in the presence of high levels of ADH.
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
351
The renal medullary interstitium surrounding the
Table 28-1
collecting ducts normally is very hyperosmotic, so that
Summary of Tubule Characteristics—Urine Concentration
when ADH levels are high, water moves through the
tubular membrane by osmosis into the renal intersti-
tium; from there it is carried away by the vasa recta
Permeability
Active NaCl
back into the blood. Thus, the urine concentrating
Transport
H2O
NaCl
Urea
ability is limited by the level of ADH and by the
Proximal tubule
++
++
+
+
degree of hyperosmolarity of the renal medulla. We
Thin descending limb
0
++
+
+
discuss the factors that control ADH secretion later,
Thin ascending limb
0
0
+
+
but for now, what is the process by which renal
Thick ascending limb
++
0
0
0
Distal tubule
+
+ADH
0
0
medullary interstitial fluid becomes hyperosmotic?
Cortical collecting
+
+ADH
0
0
This process involves the operation of the countercur-
tubule
rent mechanism.
Inner medullary
+
+ADH
0
++ADH
The countercurrent mechanism depends on the
collecting duct
special anatomical arrangement of the loops of Henle
and the vasa recta, the specialized peritubular capillar-
0, minimal level of active transport or permeability; +, moderate level of active
transport or permeability; ++, high level of active transport or permeability;
ies of the renal medulla. In the human, about 25 per
+ADH, permeability to water or urea is increased by ADH.
cent of the nephrons are juxtamedullary nephrons,
with loops of Henle and vasa recta that go deeply into
the medulla before returning to the cortex. Some of
far less than the reabsorption of solutes into the
the loops of Henle dip all the way to the tips of the
medullary interstitium
renal papillae that project from the medulla into the
renal pelvis. Paralleling the long loops of Henle are
Special Characteristics of Loop of Henle That Cause Solutes to
the vasa recta, which also loop down into the medulla
Be Trapped in the Renal Medulla. The transport character-
before returning to the renal cortex. And finally, the
istics of the loops of Henle are summarized in Table
collecting ducts, which carry urine through the hyper-
28-1, along with the characteristics of the proximal
osmotic renal medulla before it is excreted, also play
tubules, distal tubules, cortical collecting tubules, and
a critical role in the countercurrent mechanism.
inner medullary collecting ducts.
The most important cause of the high medullary
osmolarity is active transport of sodium and co-
Countercurrent Mechanism
transport of potassium, chloride, and other ions from
Produces a Hyperosmotic Renal
the thick ascending loop of Henle into the interstitium.
Medullary Interstitium
This pump is capable of establishing about a 200-
milliosmole concentration gradient between the
The osmolarity of interstitial fluid in almost all parts
tubular lumen and the interstitial fluid. Because the
of the body is about 300 mOsm/L, which is similar to
thick ascending limb is virtually impermeable to water,
the plasma osmolarity. (As discussed in Chapter 25,
the solutes pumped out are not followed by osmotic
the corrected osmolar activity, which accounts for
flow of water into the interstitium. Thus, the active
intermolecular attraction and repulsion, is about
transport of sodium and other ions out of the thick
282 mOsm/L.) The osmolarity of the interstitial fluid
ascending loop adds solutes in excess of water to the
in the medulla of the kidney is much higher, increas-
renal medullary interstitium. There is some passive
ing progressively to about 1200 to 1400 mOsm/L in the
reabsorption of sodium chloride from the thin ascend-
pelvic tip of the medulla. This means that the renal
ing limb of Henle’s loop, which is also impermeable to
medullary interstitium has accumulated solutes in
water, adding further to the high solute concentration
great excess of water. Once the high solute concentra-
of the renal medullary interstitium.
tion in the medulla is achieved, it is maintained by a
The descending limb of Henle’s loop, in contrast to
balanced inflow and outflow of solutes and water in
the ascending limb, is very permeable to water, and the
the medulla.
tubular fluid osmolarity quickly becomes equal to the
The major factors that contribute to the buildup of
renal medullary osmolarity. Therefore, water diffuses
solute concentration into the renal medulla are as
out of the descending limb of Henle’s loop into the
follows:
interstitium, and the tubular fluid osmolarity gradually
1. Active transport of sodium ions and co-transport
rises as it flows toward the tip of the loop of Henle.
of potassium, chloride, and other ions out of the
thick portion of the ascending limb of the loop
Steps Involved in Causing Hyperosmotic Renal Medullary Inter-
of Henle into the medullary interstitium
stitium. With these characteristics of the loop of Henle
2. Active transport of ions from the collecting ducts
in mind, let us now discuss how the renal medulla
into the medullary interstitium
becomes hyperosmotic. First, assume that the loop of
3. Facilitated diffusion of large amounts of urea
Henle is filled with fluid with a concentration of
from the inner medullary collecting ducts into the
300 mOsm/L, the same as that leaving the proximal
medullary interstitium
tubule (Figure 28-3, step 1). Next, the active pump of
4. Diffusion of only small amounts of water from the
the thick ascending limb on the loop of Henle is turned
medullary tubules into the medullary interstitium,
on, reducing the concentration inside the tubule and
352
Unit V The Body Fluids and Kidneys
300
300
300
300
300
200
300
200
300
200
1
300
300
300
2
300
400
200
3
400
400
200
4
300
400
200
300
300
300
300
400
200
400
400
200
400
400
400
300
300
300
300
400
200
400
400
200
400
400
400
300
300
150
300
150
300
100
5
300
350
150
6
350
350
150
7
700
700
500
Repeat Steps 4-6
400
500
300
500
500
300
1000
1000
800
400
500
300
500
500
300
1200
1200
1000
Figure 28-3
Countercurrent multiplier system in the loop of Henle for producing a hyperosmotic renal medulla. (Numerical values are in milliosmoles
per liter.)
raising the interstitial concentration; this pump estab-
hyperosmotic tubular fluid from the descending limb
lishes a 200-mOsm/L concentration gradient between
of the loop of Henle flows into the ascending limb, still
the tubular fluid and the interstitial fluid (step 2). The
more solute is continuously pumped out of the tubules
limit to the gradient is about 200 mOsm/L because
and deposited into the medullary interstitium.
paracellular diffusion of ions back into the tubule
These steps are repeated over and over, with the net
eventually counterbalances transport of ions out of the
effect of adding more and more solute to the medulla
lumen when the 200-mOsm/L concentration gradient
in excess of water; with sufficient time, this process
is achieved.
gradually traps solutes in the medulla and multiplies
Step 3 is that the tubular fluid in the descending limb
the concentration gradient established by the active
of the loop of Henle and the interstitial fluid quickly
pumping of ions out of the thick ascending loop of
reach osmotic equilibrium because of osmosis of water
Henle, eventually raising the interstitial fluid osmolar-
out of the descending limb. The interstitial osmolarity
ity to 1200 to 1400 mOsm/L as shown in step 7.
is maintained at 400 mOsm/L because of continued
Thus, the repetitive reabsorption of sodium chloride
transport of ions out of the thick ascending loop of
by the thick ascending loop of Henle and continued
Henle. Thus, by itself, the active transport of sodium
inflow of new sodium chloride from the proximal
chloride out of the thick ascending limb is capable of
tubule into the loop of Henle is called the countercur-
establishing only a 200-mOsm/L concentration gradi-
rent multiplier. The sodium chloride reabsorbed from
ent, much less than that achieved by the countercur-
the ascending loop of Henle keeps adding to the newly
rent system.
arrived sodium chloride, thus “multiplying” its con-
Step 4 is additional flow of fluid into the loop of
centration in the medullary interstitium.
Henle from the proximal tubule, which causes the
hyperosmotic fluid previously formed in the descend-
Role of Distal Tubule and
ing limb to flow into the ascending limb. Once this fluid
is in the ascending limb, additional ions are pumped
Collecting Ducts in Excreting a
into the interstitium, with water remaining behind,
Concentrated Urine
until a 200-mOsm/L osmotic gradient is established,
with the interstitial fluid osmolarity rising to
When the tubular fluid leaves the loop of Henle and
500 mOsm/L (step 5). Then, once again, the fluid in the
flows into the distal convoluted tubule in the renal
descending limb reaches equilibrium with the hyper-
cortex, the fluid is dilute, with an osmolarity of only
osmotic medullary interstitial fluid (step 6), and as the
about 100 mOsm/L (Figure 28-4). The early distal
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
353
H2O
NaCl
Urea Contributes to Hyperosmotic
NaCl
H2O
Urea
300
Renal Medullary Interstitium and to a
100
300
Concentrated Urine
300
NaCl
Thus far, we have considered only the contribution of
sodium chloride to the hyperosmotic renal medullary
interstitium. However, urea contributes about
40
to 50 per cent of the osmolarity (500-600 mOsm/L) of
the renal medullary interstitium when the kidney
600
600
600
600
is forming a maximally concentrated urine. Unlike
sodium chloride, urea is passively reabsorbed from the
NaCl
tubule. When there is water deficit and blood concen-
H2O
H2O
NaCl
trations of ADH are high, large amounts of urea are
Urea
passively reabsorbed from the inner medullary col-
1200
1200
1200
1200
lecting ducts into the interstitium.
The mechanism for reabsorption of urea into the
renal medulla is as follows: As water flows up the
ascending loop of Henle and into the distal and corti-
Figure 28-4
cal collecting tubules, little urea is reabsorbed because
Formation of a concentrated urine when antidiuretic hormone
these segments are impermeable to urea (see Table
(ADH) levels are high. Note that the fluid leaving the loop of Henle
28-1). In the presence of high concentrations of ADH,
is dilute but becomes concentrated as water is absorbed from the
water is reabsorbed rapidly from the cortical col-
distal tubules and collecting tubules. With high ADH levels, the
osmolarity of the urine is about the same as the osmolarity of
lecting tubule and the urea concentration increases
the renal medullary interstitial fluid in the papilla, which is about
rapidly because urea is not very permeant in this part
1200 mOsm/L. (Numerical values are in milliosmoles per liter.)
of the tubule. Then, as the tubular fluid flows into the
inner medullary collecting ducts, still more water reab-
sorption takes place, causing an even higher concen-
tubule further dilutes the tubular fluid because this
tration of urea in the fluid. This high concentration of
segment, like the ascending loop of Henle, actively
urea in the tubular fluid of the inner medullary col-
transports sodium chloride out of the tubule but is rel-
lecting duct causes urea to diffuse out of the tubule
atively impermeable to water.
into the renal interstitium. This diffusion is greatly
As fluid flows into the cortical collecting tubule, the
facilitated by specific urea transporters. One of these
amount of water reabsorbed is critically dependent on
urea transporters, UT-AI, is activated by ADH,
the plasma concentration of ADH. In the absence of
increasing transport of urea out of the inner medullary
ADH, this segment is almost impermeable to water
collecting duct even more when ADH levels are ele-
and fails to reabsorb water but continues to reabsorb
vated. The simultaneous movement of water and urea
solutes and further dilutes the urine. When there is a
out of the inner medullary collecting ducts maintains
high concentration of ADH, the cortical collecting
a high concentration of urea in the tubular fluid and,
tubule becomes highly permeable to water, so that
eventually, in the urine, even though urea is being
large amounts of water are now reabsorbed from the
reabsorbed.
tubule into the cortex interstitium, where it is swept
The fundamental role of urea in contributing to
away by the rapidly flowing peritubular capillaries.
urine concentrating ability is evidenced by the fact that
The fact that these large amounts of water are reab-
people who ingest a high-protein diet, yielding large
sorbed into the cortex, rather than into the renal
amounts of urea as a nitrogenous “waste” product,
medulla, helps to preserve the high medullary intersti-
can concentrate their urine much better than people
tial fluid osmolarity.
whose protein intake and urea production are low.
As the tubular fluid flows along the medullary col-
Malnutrition is associated with a low urea concentra-
lecting ducts, there is further water reabsorption from
tion in the medullary interstitium and considerable
the tubular fluid into the interstitium, but the total
impairment of urine concentrating ability.
amount of water is relatively small compared with that
added to the cortex interstitium. The reabsorbed water
Recirculation of Urea from Collecting Duct to Loop of Henle
is quickly carried away by the vasa recta into the
Contributes to Hyperosmotic Renal Medulla. A person
venous blood. When high levels of ADH are present,
usually excretes about 20 to 50 per cent of the filtered
the collecting ducts become permeable to water, so
load of urea. In general, the rate of urea excretion is
that the fluid at the end of the collecting ducts has
determined mainly by two factors: (1) the concentra-
essentially the same osmolarity as the interstitial fluid
tion of urea in the plasma and (2) the glomerular fil-
of the renal medulla—about 1200 mOsm/L (see Figure
tration rate (GFR). In patients with renal disease who
28-3). Thus, by reabsorbing as much water as possible,
have large reductions of GFR, the plasma urea con-
the kidneys form a highly concentrated urine, excret-
centration increases markedly, returning the filtered
ing normal amounts of solutes in the urine while
urea load and urea excretion rate to the normal level
adding water back to the extracellular fluid and com-
(equal to the rate of urea production), despite the
pensating for deficits of body water.
reduced GFR.
354
Unit V The Body Fluids and Kidneys
back down into the medullary collecting duct again. In
100% remaining
this way, urea can recirculate through these terminal
parts of the tubular system several times before it is
Urea
4.5
Urea
4.5
excreted. Each time around the circuit contributes to
a higher concentration of urea.
100%
remaining
This urea recirculation provides an additional mech-
Cortex
7
anism for forming a hyperosmotic renal medulla.
Because urea is one of the most abundant waste prod-
50% remaining
30
30
ucts that must be excreted by the kidneys, this mech-
Outer
H2O
anism for concentrating urea before it is excreted is
medulla
essential to the economy of the body fluid when water
15
is in short supply.
Urea
When there is excess water in the body and low
Inner
levels of ADH, the inner medullary collecting ducts
300
300
medulla
have a much lower permeability to both water and
urea, and more urea is excreted in the urine.
500
550
Urea
20% remaining
Countercurrent Exchange in the Vasa
Recta Preserves Hyperosmolarity
Figure 28-5
of the Renal Medulla
Recirculation of urea absorbed from the medullary collecting duct
Blood flow must be provided to the renal medulla to
into the interstitial fluid. This urea diffuses into the thin loop of
supply the metabolic needs of the cells in this part of
Henle, and then passes through the distal tubules, and finally
passes back into the collecting duct. The recirculation of urea
the kidney. Without a special medullary blood flow
helps to trap urea in the renal medulla and contributes to the
system, the solutes pumped into the renal medulla by
hyperosmolarity of the renal medulla. The heavy dark lines, from
the countercurrent multiplier system would be rapidly
the thick ascending loop of Henle to the medullary collecting
dissipated.
ducts, indicate that these segments are not very permeable to
There are two special features of the renal
urea. (Numerical values are in milliosmoles per liter of urea during
antidiuresis, when large amounts of antidiuretic hormone are
medullary blood flow that contribute to the preserva-
present. Percentages of the filtered load of urea that remain in the
tion of the high solute concentrations:
tubules are indicated in the boxes.)
1. The medullary blood flow is low, accounting for
less than 5 per cent of the total renal blood flow.
This sluggish blood flow is sufficient to supply
the metabolic needs of the tissues but helps to
In the proximal tubule, 40 to 50 per cent of the fil-
minimize solute loss from the medullary
tered urea is reabsorbed, but even so, the tubular fluid
interstitium.
urea concentration increases because urea is not
2. The vasa recta serve as countercurrent exchangers,
nearly as permeant as water. The concentration of urea
minimizing washout of solutes from the medullary
continues to rise as the tubular fluid flows into the thin
interstitium.
segments of the loop of Henle, partly because of water
The countercurrent exchange mechanism operates as
reabsorption out of the descending loop of Henle but
follows (Figure 28-6): Blood enters and leaves the
also because of some secretion of urea into the thin
medulla by way of the vasa recta at the boundary
loop of Henle from the medullary interstitium (Figure
of the cortex and renal medulla. The vasa recta, like
28-5).
other capillaries, are highly permeable to solutes in
The thick limb of the loop of Henle, the distal
the blood, except for the plasma proteins. As blood
tubule, and the cortical collecting tubule are all rela-
descends into the medulla toward the papillae, it
tively impermeable to urea, and very little urea reab-
becomes progressively more concentrated, partly
sorption occurs in these tubular segments. When the
by solute entry from the interstitium and partly by
kidney is forming a concentrated urine and high levels
loss of water into the interstitium. By the time the
of ADH are present, the reabsorption of water from
blood reaches the tips of the vasa recta, it has a con-
the distal tubule and cortical collecting tubule further
centration of about 1200 mOsm/L, the same as that of
raises the tubular fluid concentration of urea. And as
the medullary interstitium. As blood ascends back
this urea flows into the inner medullary collecting
toward the cortex, it becomes progressively less
duct, the high tubular fluid concentration of urea
concentrated as solutes diffuse back out into the
and specific urea transporters cause urea to diffuse
medullary interstitium and as water moves into the
into the medullary interstitium. A moderate share of
vasa recta.
the urea that moves into the medullary interstitium
Thus, although there is a large amount of fluid and
eventually diffuses into the thin loop of Henle, so that
solute exchange across the vasa recta, there is little net
it passes upward through the ascending loop of Henle,
dilution of the concentration of the interstitial fluid at
the distal tubule, the cortical collecting tubule, and
each level of the renal medulla because of the U shape
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
355
Vasa recta
Interstitium
Summary of Urine Concentrating
mOsm/L
mOsm/L
Mechanism and Changes in
300
350
300
Osmolarity in Different Segments
Solute
of the Tubules
H2O
600
Solute
600
600
The changes in osmolarity and volume of the tubular
600
fluid as it passes through the different parts of the
Solute
nephron are shown in Figure 28-7.
H2O
800
Solute
800
800
Proximal Tubule. About
65 per cent of the filtered
900
Solute
electrolytes are reabsorbed in the proximal tubule.
H2O
1000
Solute
1000
However, the tubular membranes are highly perme-
1000
able to water, so that whenever solutes are reabsorbed,
water also diffuses through the tubular membrane by
1200
1200
osmosis. Therefore, the osmolarity of the fluid remains
about the same as the glomerular filtrate, 300 mOsm/L.
Figure 28-6
Descending Loop of Henle. As fluid flows down the
Countercurrent exchange in the vasa recta. Plasma flowing down
the descending limb of the vasa recta becomes more hyperos-
descending loop of Henle, water is absorbed into the
motic because of diffusion of water out of the blood and diffusion
medulla. The descending limb is highly permeable to
of solutes from the renal interstitial fluid into the blood. In the
water but much less permeable to sodium chloride and
ascending limb of the vasa recta, solutes diffuse back into the
urea. Therefore, the osmolarity of the fluid flowing
interstitial fluid and water diffuses back into the vasa recta. Large
amounts of solutes would be lost from the renal medulla without
through the descending loop gradually increases until
the U shape of the vasa recta capillaries. (Numerical values are
it is equal to that of the surrounding interstitial fluid,
in milliosmoles per liter.)
which is about 1200 mOsm/L when the blood concen-
tration of ADH is high. When a dilute urine is being
formed, owing to low ADH concentrations, the med-
ullary interstitial osmolarity is less than 1200 mOsm/L;
consequently, the descending loop tubular fluid osmo-
of the vasa recta capillaries, which act as countercur-
larity also becomes less concentrated. This is due
rent exchangers. Thus, the vasa recta do not create the
partly to the fact that less urea is absorbed into the
medullary hyperosmolarity, but they do prevent it from
medullary interstitium from the collecting ducts when
being dissipated.
ADH levels are low and the kidney is forming a large
The U-shaped structure of the vessels minimizes
volume of dilute urine.
loss of solute from the interstitium but does not
prevent the bulk flow of fluid and solutes into the
Thin Ascending Loop of Henle. The thin ascending limb is
blood through the usual colloid osmotic and hydro-
essentially impermeable to water but reabsorbs some
static pressures that favor reabsorption in these capil-
sodium chloride. Because of the high concentration of
laries. Thus, under steady-state conditions, the vasa
sodium chloride in the tubular fluid, owing to water
recta carry away only as much solute and water as is
removal from the descending loop of Henle, there is
absorbed from the medullary tubules, and the high
some passive diffusion of sodium chloride from the
concentration of solutes established by the counter-
thin ascending limb into the medullary interstitium.
current mechanism is maintained.
Thus, the tubular fluid becomes more dilute as the
sodium chloride diffuses out of the tubule and water
Increased Medullary Blood Flow Can Reduce Urine Concentrat-
remains in the tubule. Some of the urea absorbed into
ing Ability. Certain vasodilators can markedly increase
the medullary interstitium from the collecting ducts
renal medullary blood flow, thereby “washing out”
also diffuses into the ascending limb, thereby return-
some of the solutes from the renal medulla and reduc-
ing the urea to the tubular system and helping to
ing maximum urine concentrating ability. Large
prevent its washout from the renal medulla. This urea
increases in arterial pressure can also increase the
recycling is an additional mechanism that contributes
blood flow of the renal medulla to a greater extent
to the hyperosmotic renal medulla.
than in other regions of the kidney and tend to wash
out the hyperosmotic interstitium, thereby reducing
urine concentrating ability. As discussed earlier,
Thick Ascending Loop of Henle. The thick part of the
maximum concentrating ability of the kidney is deter-
ascending loop of Henle is also virtually impermeable
mined not only by the level of ADH but also by the
to water, but large amounts of sodium, chloride, potas-
osmolarity of the renal medulla interstitial fluid. Even
sium, and other ions are actively transported from
with maximal levels of ADH, urine concentrating
the tubule into the medullary interstitium. Therefore,
ability will be reduced if medullary blood flow
fluid in the thick ascending limb of the loop of Henle
increases enough to reduce the hyperosmolarity in the
becomes very dilute, falling to a concentration of
renal medulla.
about 100 mOsm/L.
356
Unit V The Body Fluids and Kidneys
0.2 ml
25 ml
1200
900
600
Figure 28-7
8 ml
Changes in osmolarity of the
300
tubular fluid as it passes through
125 ml
44 ml
the different tubular segments in
200
the presence of high levels of
100
antidiuretic hormone (ADH) and
25 ml
20 ml
in the absence of ADH. (Numeri-
0
Proximal
Loop of Henle
Distal
Collecting
Urine
cal values indicate the approxi-
tubule
tubule
tubule
mate volumes in milliliters per
and duct
minute or in osmolarities in mil-
liosmoles per liter of fluid flow-
ing along the different tubular
segments.)
Early Distal Tubule. The early distal tubule has proper-
fluid, and because the inner medullary collecting ducts
ties similar to those of the thick ascending loop of
have specific urea transporters that greatly facilitate
Henle, so that further dilution of the tubular fluid
diffusion, much of the highly concentrated urea in
occurs as solutes are reabsorbed while water remains
the ducts diffuses out of the tubular lumen into the
in the tubule.
medullary interstitium. This absorption of the urea
into the renal medulla contributes to the high osmo-
Late Distal Tubule and Cortical Collecting Tubules. In the late
larity of the medullary interstitium and the high con-
distal tubule and cortical collecting tubules, the osmo-
centrating ability of the kidney.
larity of the fluid depends on the level of ADH. With
There are several important points to consider that
high levels of ADH, these tubules are highly perme-
may not be obvious from this discussion. First,
able to water, and significant amounts of water are
although sodium chloride is one of the principal
reabsorbed. Urea, however, is not very permeant in
solutes that contributes to the hyperosmolarity of the
this part of the nephron, resulting in increased urea
medullary interstitium, the kidney can, when needed,
concentration as water is reabsorbed. This allows most
excrete a highly concentrated urine that contains little
of the urea delivered to the distal tubule and collect-
sodium chloride. The hyperosmolarity of the urine in
ing tubule to pass into the inner medullary collecting
these circumstances is due to high concentrations of
ducts, from which it is eventually reabsorbed or
other solutes, especially of waste products such as urea
excreted in the urine. In the absence of ADH, little
and creatinine. One condition in which this occurs is
water is reabsorbed in the late distal tubule and corti-
dehydration accompanied by low sodium intake. As
cal collecting tubule; therefore, osmolarity decreases
discussed in Chapter 29, low sodium intake stimulates
even further because of continued active reabsorption
formation of the hormones angiotensin II and aldos-
of ions from these segments.
terone, which together cause avid sodium reabsorption
from the tubules while leaving the urea and other
Inner Medullary Collecting Ducts. The concentration of
solutes to maintain the highly concentrated urine.
fluid in the inner medullary collecting ducts also
Second, large quantities of dilute urine can be
depends on (1) ADH and (2) the osmolarity of the
excreted without increasing the excretion of sodium.
medullary interstitium established by the countercur-
This is accomplished by decreasing ADH secretion,
rent mechanism. In the presence of large amounts of
which reduces water reabsorption in the more distal
ADH, these ducts are highly permeable to water, and
tubular segments without significantly altering sodium
water diffuses from the tubule into the interstitium
reabsorption.
until osmotic equilibrium is reached, with the tubular
And finally, we should keep in mind that there is
fluid having about the same concentration as the renal
an obligatory urine volume, which is dictated by the
medullary interstitium (1200 to 1400 mOsm/L). Thus,
maximum concentrating ability of the kidney and the
a very concentrated but small volume of urine is pro-
amount of solute that must be excreted. Therefore, if
duced when ADH levels are high. Because water reab-
large amounts of solute must be excreted, they must
sorption increases urea concentration in the tubular
be accompanied by the minimal amount of water
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
357
necessary to excrete them. For example, if 1200 mil-
Disorders of Urinary
liosmoles of solute must be excreted each day, this
Concentrating Ability
requires at least 1 liter of urine if maximal urine con-
centrating ability is 1200 mOsm/L.
An impairment in the ability of the kidneys to concen-
trate or dilute the urine appropriately can occur with
one or more of the following abnormalities:
1. Inappropriate secretion of ADH. Either too much
Quantifying Renal Urine
or too little ADH secretion results in abnormal
Concentration and Dilution:
fluid handling by the kidneys.
“Free Water” and
2. Impairment of the countercurrent mechanism. A
hyperosmotic medullary interstitium is required for
Osmolar Clearances
maximal urine concentrating ability. No matter how
The process of concentrating or diluting the urine
much ADH is present, maximal urine concentration
requires the kidneys to excrete water and solutes some-
is limited by the degree of hyperosmolarity of the
what independently. When the urine is dilute, water is
medullary interstitium.
excreted in excess of solutes. Conversely, when the urine
3. Inability of the distal tubule, collecting tubule, and
is concentrated, solutes are excreted in excess of water.
collecting ducts to respond to ADH.
The total clearance of solutes from the blood can be
expressed as the osmolar clearance (Cosm); this is the
Failure to Produce ADH:
“Central” Diabetes Insipidus. An
volume of plasma cleared of solutes each minute, in
inability to produce or release ADH from the posterior
the same way that clearance of a single substance is cal-
pituitary can be caused by head injuries or infections, or
culated:
it can be congenital. Because the distal tubular segments
cannot reabsorb water in the absence of ADH, this con-
osm
¥
V
dition, called “central” diabetes insipidus, results in the
C =U
osm
P
osm
formation of a large volume of dilute urine, with urine
volumes that can exceed 15 L/day. The thirst mecha-
where Uosm is the urine osmolarity, V is the urine flow
nisms, discussed later in this chapter, are activated when
rate, and Posm is the plasma osmolarity. For example, if
excessive water is lost from the body; therefore, as long
plasma osmolarity is 300 mOsm/L, urine osmolarity is
as the person drinks enough water, large decreases in
600 mOsm/L, and urine flow rate is 1 ml/min (0.001 L/
body fluid water do not occur. The primary abnormal-
min), the rate of osmolar excretion is 0.6 mOsm/min
ity observed clinically in people with this condition is
(600 mOsm/L ¥ 0.001 L/min) and osmolar clearance is
the large volume of dilute urine. However, if water
0.6 mOsm/min divided by 300 mOsm/L, or 0.002 L/min
intake is restricted, as can occur in a hospital setting
(2.0 ml/min). This means that 2 milliliters of plasma are
when fluid intake is restricted or the patient is uncon-
being cleared of solute each minute.
scious (for example, because of a head injury), severe
dehydration can rapidly occur.
Relative Rates at Which Solutes and Water Are Excreted Can Be
The treatment for central diabetes insipidus is admin-
Assessed Using the Concept of “Free-Water Clearance.” Free-
istration of a synthetic analog of ADH, desmopressin,
water clearance (CH
2O) is calculated as the difference
which acts selectively on V2 receptors to increase water
between water excretion (urine flow rate) and osmolar
permeability in the late distal and collecting tubules.
clearance:
Desmopressin can be given by injection, as a nasal spray,
or orally, and rapidly restores urine output toward
(Uosm¥V)
2
CH O=V Cosm=V
-
normal.
(P
osm
)
Thus, the rate of free-water clearance represents the
Inability of the Kidneys to Respond to ADH: “Nephrogenic”
rate at which solute-free water is excreted by the
Diabetes Insipidus. There are circumstances in which
kidneys. When free-water clearance is positive, excess
normal or elevated levels of ADH are present but the
water is being excreted by the kidneys; when free-water
renal tubular segments cannot respond appropriately.
clearance is negative, excess solutes are being removed
This condition is referred to as “nephrogenic” diabetes
from the blood by the kidneys and water is being
insipidus because the abnormality resides in the
conserved.
kidneys. This abnormality can be due to either failure of
Using the example discussed earlier, if urine flow
the countercurrent mechanism to form a hyperosmotic
rate is
1 ml/min and osmolar clearance is 2 ml/min,
renal medullary interstitium or failure of the distal and
free-water clearance would be -1 ml/min. This means
collecting tubules and collecting ducts to respond to
that instead of water being cleared from the kidneys in
ADH. In either case, large volumes of dilute urine are
excess of solutes, the kidneys are actually returning
formed, which tends to cause dehydration unless fluid
water back to the systemic circulation, as occurs during
intake is increased by the same amount as urine volume
water deficits. Thus, whenever urine osmolarity is greater
is increased.
than plasma osmolarity, free-water clearance will be neg-
Many types of renal diseases can impair the concen-
ative, indicating water conservation.
trating mechanism, especially those that damage the
When the kidneys are forming a dilute urine (that is,
renal medulla. Also, impairment of the function of the
urine osmolarity is less than plasma osmolarity), free-
loop of Henle, as occurs with diuretics that inhibit elec-
water clearance will be a positive value, denoting that
trolyte reabsorption by this segment, can compromise
water is being removed from the plasma by the kidneys
urine concentrating ability. And certain drugs, such as
in excess of solutes. Thus, water free of solutes, called
lithium
(used to treat manic-depressive disorders)
“free water,” is being lost from the body and the plasma
and tetracyclines (used as antibiotics), can impair the
is being concentrated when free-water clearance is
ability of the distal nephron segments to respond to
positive.
ADH.
358
Unit V The Body Fluids and Kidneys
Nephrogenic diabetes insipidus can be distinguished
Although multiple mechanisms control the amount
from central diabetes insipidus by administration of
of sodium and water excretion by the kidneys, two
desmopressin, the synthetic analog of ADH. Lack of a
primary systems are especially involved in regulating
prompt decrease in urine volume and an increase in
the concentration of sodium and osmolarity of extra-
urine osmolarity within
2 hours after injection of
cellular fluid: (1) the osmoreceptor-ADH system and
desmopressin is strongly suggestive of nephrogenic dia-
(2) the thirst mechanism.
betes insipidus. The treatment for nephrogenic diabetes
insipidus is to correct, if possible, the underlying renal
disorder. The hypernatremia can also be attenuated by
Osmoreceptor-ADH
a low-sodium diet and administration of a diuretic that
enhances renal sodium excretion, such as a thiazide
Feedback System
diuretic.
Figure
28-8 shows the basic components of the
osmoreceptor-ADH feedback system for control of
Control of Extracellular Fluid
extracellular fluid sodium concentration and osmolar-
Osmolarity and Sodium
ity. When osmolarity (plasma sodium concentration)
Concentration
increases above normal because of water deficit, for
example, this feedback system operates as follows:
Regulation of extracellular fluid osmolarity and
1.
An increase in extracellular fluid osmolarity
sodium concentration are closely linked because
(which in practical terms means an increase in
sodium is the most abundant ion in the extracellular
plasma sodium concentration) causes the special
compartment. Plasma sodium concentration is nor-
nerve cells called osmoreceptor cells, located in
mally regulated within close limits of 140 to 145 mEq/
the anterior hypothalamus near the supraoptic
L, with an average concentration of about 142 mEq/L.
nuclei, to shrink.
Osmolarity averages about
300 mOsm/L
(about
2.
Shrinkage of the osmoreceptor cells causes them
282 mOsm/L when corrected for interionic attraction)
to fire, sending nerve signals to additional nerve
and seldom changes more than ±2 to 3 per cent. As
cells in the supraoptic nuclei, which then relay
discussed in Chapter 25, these variables must be pre-
cisely controlled because they determine the distribu-
tion of fluid between the intracellular and extracellular
compartments.
-
Water deficit
Estimating Plasma Osmolarity from
Plasma Sodium Concentration
Extracellular osmolarity
In most clinical laboratories, plasma osmolarity is not
routinely measured. However, because sodium and its
Osmoreceptors
associated anions account for about 94 per cent of the
solute in the extracellular compartment, plasma osmo-
ADH secretion
(posterior pituitary)
larity (Posm) can be roughly approximated as
Posm = 2.1
¥ Plasma sodium concentration
For instance, with a plasma sodium concentration of
Plasma ADH
142 mEq/L, the plasma osmolarity would be estimated
from the formula above to be about 298 mOsm/L. To
be more exact, especially in conditions associated with
renal disease, the contribution of two other solutes,
H2O permeability in
glucose and urea, should be included. Such estimates
distal tubules,
collecting ducts
of plasma osmolarity are usually accurate within a few
percentage points of those measured directly.
Normally, sodium ions and associated anions (pri-
marily bicarbonate and chloride) represent about 94
H2O reabsorption
per cent of the extracellular osmoles, with glucose and
urea contributing about 3 to 5 per cent of the total
osmoles. However, because urea easily permeates
most cell membranes, it exerts little effective osmotic
H2O excreted
pressure under steady-state conditions. Therefore, the
sodium ions in the extracellular fluid and associated
anions are the principal determinants of fluid move-
Figure 28-8
ment across the cell membrane. Consequently, we can
Osmoreceptor-antidiuretic hormone (ADH) feedback mechanism
discuss the control of osmolarity and control of sodium
for regulating extracellular fluid osmolarity in response to a water
ion concentration at the same time.
deficit.
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
359
these signals down the stalk of the pituitary gland
to the posterior pituitary.
3. These action potentials conducted to the posterior
pituitary stimulate the release of ADH, which is
stored in secretory granules (or vesicles) in the
Pituitary
nerve endings.
4. ADH enters the blood stream and is transported
to the kidneys, where it increases the water
Osmoreceptors
permeability of the late distal tubules, cortical
collecting tubules, and medullary collecting ducts.
Baroreceptors
5. The increased water permeability in the distal
Cardiopulmonary
nephron segments causes increased water
Supraoptic
receptors
reabsorption and excretion of a small volume of
neuron
concentrated urine.
Paraventricular
neuron
Thus, water is conserved in the body while sodium and
Anterior
other solutes continue to be excreted in the urine. This
lobe
causes dilution of the solutes in the extracellular fluid,
Posterior
lobe
thereby correcting the initial excessively concentrated
extracellular fluid.
The opposite sequence of events occurs when the
extracellular fluid becomes too dilute (hypo-osmotic).
ADH
For example, with excess water ingestion and a
decrease in extracellular fluid osmolarity, less ADH is
formed, the renal tubules decrease their permeability
for water, less water is reabsorbed, and a large volume
of dilute urine is formed. This in turn concentrates the
body fluids and returns plasma osmolarity toward
normal.
ADH Synthesis in Supraoptic and
Urine:
decreased flow
Paraventricular Nuclei of the
and concentrated
Hypothalamus and ADH Release
Figure 28-9
from the Posterior Pituitary
Neuroanatomy of the hypothalamus, where antidiuretic hormone
Figure 28-9 shows the neuroanatomy of the hypothal-
(ADH) is synthesized, and the posterior pituitary gland, where
amus and the pituitary gland, where ADH is synthe-
ADH is released.
sized and released. The hypothalamus contains two
types of magnocellular (large) neurons that synthesize
ADH in the supraoptic and paraventricular nuclei of
anteroventral region of the third ventricle, called the
the hypothalamus, about five sixths in the supraoptic
AV3V region. At the upper part of this region is a
nuclei and about one sixth in the paraventricular
structure called the subfornical organ, and at the infe-
nuclei. Both of these nuclei have axonal extensions to
rior part is another structure called the organum vas-
the posterior pituitary. Once ADH is synthesized, it is
culosum of the lamina terminalis. Between these two
transported down the axons of the neurons to their
organs is the median preoptic nucleus, which has mul-
tips, terminating in the posterior pituitary gland. When
tiple nerve connections with the two organs as well as
the supraoptic and paraventricular nuclei are stimu-
with the supraoptic nuclei and the blood pressure
lated by increased osmolarity or other factors, nerve
control centers in the medulla of the brain. Lesions of
impulses pass down these nerve endings, changing
the AV3V region cause multiple deficits in the control
their membrane permeability and increasing calcium
of ADH secretion, thirst, sodium appetite, and blood
entry. ADH stored in the secretory granules (also
pressure. Electrical stimulation of this region or stim-
called vesicles) of the nerve endings is released in
ulation by angiotensin II can alter ADH secretion,
response to increased calcium entry. The released
thirst, and sodium appetite.
ADH is then carried away in the capillary blood of the
In the vicinity of the AV3V region and the supraop-
posterior pituitary into the systemic circulation.
tic nuclei are neuronal cells that are excited by small
Secretion of ADH in response to an osmotic stimu-
increases in extracellular fluid osmolarity; hence, the
lus is rapid, so that plasma ADH levels can increase
term osmoreceptors has been used to describe these
severalfold within minutes, thereby providing a rapid
neurons. These cells send nerve signals to the supraop-
means for altering renal excretion of water.
tic nuclei to control their firing and secretion of ADH.
A second neuronal area important in controlling
It is also likely that they induce thirst in response to
osmolarity and ADH secretion is located along the
increased extracellular fluid osmolarity.
360
Unit V The Body Fluids and Kidneys
Both the subfornical organ and the organum vascu-
losum of the lamina terminalis have vascular supplies
that lack the typical blood-brain barrier that impedes
Isotonic volume depletion
the diffusion of most ions from the blood into the brain
Isovolemic osmotic increase
tissue. This makes it possible for ions and other solutes
to cross between the blood and the local interstitial
PAVP = 1.3 e-0.17 vol.
fluid in this region. As a result, the osmoreceptors
50
rapidly respond to changes in osmolarity of the extra-
45
cellular fluid, exerting powerful control over the secre-
40
tion of ADH and over thirst, as discussed later.
35
30
Cardiovascular Reflex Stimulation
25
of ADH Release by Decreased
PAVP = 2.5 D Osm + 2.0
Arterial Pressure and/or Decreased
20
Blood Volume
15
10
ADH release is also controlled by cardiovascular
reflexes that respond to decreases in blood pressure
5
and/or blood volume, including (1) the arterial barore-
0
ceptor reflexes and (2) the cardiopulmonary reflexes,
both of which are discussed in Chapter 18. These reflex
0
5
10
15
20
pathways originate in high-pressure regions of the cir-
Per cent change
culation, such as the aortic arch and carotid sinus, and
in the low-pressure regions, especially in the cardiac
atria. Afferent stimuli are carried by the vagus and
glossopharyngeal nerves with synapses in the nuclei of
Figure 28-10
the tractus solitarius. Projections from these nuclei
relay signals to the hypothalamic nuclei that control
The effect of increased plasma osmolarity or decreased blood
ADH synthesis and secretion.
volume on the level of plasma (P) antidiuretic hormone (ADH), also
called arginine vasopressin
(AVP).
(Redrawn from Dunn FL,
Thus, in addition to increased osmolarity, two other
Brennan TJ, Nelson AE, Robertson GL: The role of blood osmo-
stimuli increase ADH secretion: (1) decreased arterial
lality and volume in regulating vasopressin secretion in the rat. J
pressure and (2) decreased blood volume. Whenever
Clin Invest 52(12):3212, 1973. By copyright permission of the
blood pressure and blood volume are reduced, such as
American Society of Clinical Investigation.)
occurs during hemorrhage, increased ADH secretion
causes increased fluid reabsorption by the kidneys,
helping to restore blood pressure and blood volume
Table 28-2
toward normal.
Regulation of ADH Secretion
Increase ADH
Decrease ADH
Quantitative Importance of
Cardiovascular Reflexes and
≠ Plasma osmolarity
Ø Plasma osmolarity
Ø Blood volume
≠ Blood volume
Osmolarity in Stimulating
Ø Blood pressure
≠ Blood pressure
ADH Secretion
Nausea
Hypoxia
As shown in Figure 28-10, either a decrease in effec-
Drugs:
Drugs:
tive blood volume or an increase in extracellular fluid
Morphine
Alcohol
osmolarity stimulates ADH secretion. However, ADH
Nicotine
Clonidine (antihypertensive drug)
is considerably more sensitive to small changes in
Cyclophosphamide
Haloperidol (dopamine blocker)
osmolarity than to similar changes in blood volume.
For example, a change in plasma osmolarity of only
1 per cent is sufficient to increase ADH levels. By
greatly enhances the ADH response to increased
contrast, after blood loss, plasma ADH levels do not
osmolarity.
change appreciably until blood volume is reduced by
about 10 per cent. With further decreases in blood
volume, ADH levels rapidly increase. Thus, with severe
Other Stimuli for ADH Secretion
decreases in blood volume, the cardiovascular reflexes
play a major role in stimulating ADH secretion. The
ADH secretion can also be increased or decreased by
usual day-to-day regulation of ADH secretion during
other stimuli to the central nervous system as well as
simple dehydration is effected mainly by changes in
by various drugs and hormones, as shown in Table
plasma osmolarity. Decreased blood volume, however,
28-2. For example, nausea is a potent stimulus for
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
361
ADH release, which may increase to as much as 100
Table 28-3
times normal after vomiting. Also, drugs such as nico-
Control of Thirst
tine and morphine stimulate ADH release, whereas
some drugs, such as alcohol, inhibit ADH release. The
marked diuresis that occurs after ingestion of alcohol
Increase Thirst
Decrease Thirst
is due in part to inhibition of ADH release.
≠ Osmolarity
Ø Osmolarity
Ø Blood volume
≠ Blood volume
Ø Blood pressure
≠ Blood pressure
≠ Angiotensin
Ø Angiotensin II
Role of Thirst in Controlling
Dryness of mouth
Gastric distention
Extracellular Fluid Osmolarity
and Sodium Concentration
The kidneys minimize fluid loss during water deficits
through the osmoreceptor-ADH feedback system.
Adequate fluid intake, however, is necessary to coun-
Decreases in extracellular fluid volume and arterial
terbalance whatever fluid loss does occur through
pressure also stimulate thirst by a pathway that is inde-
sweating and breathing and through the gastrointesti-
pendent of the one stimulated by increased plasma
nal tract. Fluid intake is regulated by the thirst mech-
osmolarity. Thus, blood volume loss by hemorrhage
anism, which, together with the osmoreceptor-ADH
stimulates thirst even though there might be no change
mechanism, maintains precise control of extracellular
in plasma osmolarity. This probably occurs because of
fluid osmolarity and sodium concentration.
neutral input from cardiopulmonary and systemic
Many of the same factors that stimulate ADH secre-
arterial baroreceptors in the circulation.
tion also increase thirst, which is defined as the con-
A third important stimulus for thirst is angiotensin II.
scious desire for water.
Studies in animals have shown that angiotensin II acts
on the subfornical organ and on the organum vascu-
losum of the lamina terminalis. These regions are
Central Nervous System Centers
outside the blood-brain barrier, and peptides such as
for Thirst
angiotensin II diffuse into the tissues. Because
angiotensin II is also stimulated by factors associated
Referring again to Figure 28-9, the same area along
with hypovolemia and low blood pressure, its effect on
the anteroventral wall of the third ventricle that pro-
thirst helps to restore blood volume and blood pres-
motes ADH release also stimulates thirst. Located
sure toward normal, along with the other actions
anterolaterally in the preoptic nucleus is another small
of angiotensin II on the kidneys to decrease fluid
area that, when stimulated electrically, causes immedi-
excretion.
ate drinking that continues as long as the stimulation
Dryness of the mouth and mucous membranes of the
lasts. All these areas together are called the thirst
esophagus can elicit the sensation of thirst. As a result,
center.
a thirsty person may receive relief from thirst almost
The neurons of the thirst center respond to injec-
immediately after drinking water, even though the
tions of hypertonic salt solutions by stimulating drink-
water has not been absorbed from the gastrointestinal
ing behavior. These cells almost certainly function as
tract and has not yet had an effect on extracellular
osmoreceptors to activate the thirst mechanism, in
fluid osmolarity.
the same way that the osmoreceptors stimulate ADH
Gastrointestinal and pharyngeal stimuli influence
release.
thirst. For example, in animals that have an esophageal
Increased osmolarity of the cerebrospinal fluid in
opening to the exterior so that water is never absorbed
the third ventricle has essentially the same effect to
into the blood, partial relief of thirst occurs after
promote drinking. It is likely that the organum vascu-
drinking, although the relief is only temporary. Also,
losum of the lamina terminalis, which lies immediately
gastrointestinal distention may partially alleviate
beneath the ventricular surface at the inferior end of
thirst; for instance, simple inflation of a balloon in the
the AV3V region, is intimately involved in mediating
stomach can relieve thirst. However, relief of thirst
this response.
sensations through gastrointestinal or pharyngeal
mechanisms is short-lived; the desire to drink is com-
pletely satisfied only when plasma osmolarity and/or
Stimuli for Thirst
blood volume returns to normal.
The ability of animals and humans to “meter” fluid
Table 28-3 summarizes some of the known stimuli for
intake is important because it prevents overhydration.
thirst. One of the most important is increased extra-
After a person drinks water, 30 to 60 minutes may be
cellular fluid osmolarity, which causes intracellular
required for the water to be reabsorbed and dis-
dehydration in the thirst centers, thereby stimulating
tributed throughout the body. If the thirst sensation
the sensation of thirst. The value of this response is
were not temporarily relieved after drinking water, the
obvious: it helps to dilute extracellular fluids and
person would continue to drink more and more, even-
returns osmolarity toward normal.
tually leading to overhydration and excess dilution of
362
Unit V The Body Fluids and Kidneys
the body fluids. Experimental studies have repeatedly
shown that animals drink almost exactly the amount
necessary to return plasma osmolarity and volume to
152
normal.
Threshold for Osmolar Stimulus
ADH and
148
thirst
of Drinking
systems
blocked
The kidneys must continually excrete at least some
fluid, even in a dehydrated person, to rid the body of
excess solutes that are ingested or produced by metab-
144
olism. Water is also lost by evaporation from the lungs
and the gastrointestinal tract and by evaporation and
Normal
sweating from the skin. Therefore, there is always a
tendency for dehydration, with resultant increased
140
extracellular fluid sodium concentration and osmolar-
ity. When the sodium concentration increases only
about 2 mEq/L above normal, the thirst mechanism is
activated, causing a desire to drink water. This is called
the threshold for drinking. Thus, even small increases
136
in plasma osmolarity are normally followed by water
0
30
60
90
120
150
180
intake, which restores extracellular fluid osmolarity
Sodium intake (mEq/day)
and volume toward normal. In this way, the extracel-
lular fluid osmolarity and sodium concentration are
precisely controlled.
Figure 28-11
Effect of large changes in sodium intake on extracellular fluid
Integrated Responses of
sodium concentration in dogs under normal conditions (red line)
and after the antidiuretic hormone (ADH) and thirst feedback
Osmoreceptor-ADH and Thirst
systems had been blocked (blue line). Note that control of extra-
Mechanisms in Controlling
cellular fluid sodium concentration is poor in the absence of these
feedback systems. (Courtesy Dr. David B. Young.)
Extracellular Fluid Osmolarity and
Sodium Concentration
Role of Angiotensin II and
In a healthy person, the osmoreceptor-ADH and thirst
Aldosterone in Controlling
mechanisms work in parallel to precisely regulate
Extracellular Fluid Osmolarity and
extracellular fluid osmolarity and sodium concentra-
Sodium Concentration
tion, despite the constant challenges of dehydration.
Even with additional challenges, such as high salt
As discussed in Chapter 27, both angiotensin II and
intake, these feedback systems are able to keep plasma
aldosterone play an important role in regulating sodium
osmolarity reasonably constant. Figure 28-11 shows
reabsorption by the renal tubules. When sodium intake
that an increase in sodium intake to as high as six times
is low, increased levels of these hormones stimulate
normal has only a small effect on plasma sodium con-
sodium reabsorption by the kidneys and, therefore,
prevent large sodium losses, even though sodium intake
centration as long as the ADH and thirst mechanisms
may be reduced to as low as 10 per cent of normal. Con-
are both functioning normally.
versely, with high sodium intake, decreased formation of
When either the ADH or the thirst mechanism fails,
these hormones permits the kidneys to excrete large
the other ordinarily can still control extracellular
amounts of sodium.
osmolarity and sodium concentration with reasonable
Because of the importance of angiotensin II and
effectiveness, as long as there is enough fluid intake
aldosterone in regulating sodium excretion by the
to balance the daily obligatory urine volume and
kidneys, one might mistakenly infer that they also
water losses caused by respiration, sweating, or
play an important role in regulating extracellular fluid
gastrointestinal losses. However, if both the ADH
sodium concentration. Although these hormones
and thirst mechanisms fail simultaneously, plasma
increase the amount of sodium in the extracellular fluid,
they also increase the extracellular fluid volume by
sodium concentration and osmolarity are very poorly
increasing reabsorption of water along with the sodium.
controlled; thus, when sodium intake is increased
Therefore, angiotensin II and aldosterone have little
after blocking the total ADH-thirst system, relatively
effect on sodium concentration, except under extreme
large changes in plasma sodium concentration do
conditions.
occur. In the absence of the ADH-thirst mechanisms,
This relative unimportance of aldosterone in regulat-
no other feedback mechanism is capable of ad-
ing extracellular fluid sodium concentration is shown by
equately regulating plasma sodium concentration and
the experiment of Figure 28-12. This figure shows the
osmolarity.
effect on plasma sodium concentration of changing
Chapter 28
Regulation of Extracellular Fluid Osmolarity and Sodium Concentration
363
reasons for this is that large losses of sodium eventually
cause severe volume depletion and decreased blood
pressure, which can activate the thirst mechanism
through the cardiovascular reflexes. This leads to a
150
further dilution of the plasma sodium concentration,
Normal
even though the increased water intake helps to mini-
140
mize the decrease in body fluid volumes under these
Aldosterone system blocked
conditions.
130
Thus, there are extreme situations in which plasma
sodium concentration may change significantly, even
120
with a functional ADH-thirst mechanism. Even so, the
ADH-thirst mechanism is by far the most powerful
feedback system in the body for controlling extracellu-
110
lar fluid osmolarity and sodium concentration.
100
0
30
60
90
120
150
180
210
Salt-Appetite Mechanism
Sodium intake (mEq/L)
for Controlling Extracellular
Fluid Sodium Concentration
and Volume
Figure 28-12
Maintenance of normal extracellular fluid volume and
Effect of large changes in sodium intake on extracellular fluid
sodium concentration requires a balance between
sodium concentration in dogs under normal conditions (red line)
sodium excretion and sodium intake. In modern civi-
and after the aldosterone feedback system had been blocked
lizations, sodium intake is almost always greater than
(blue line). Note that sodium concentration is maintained relatively
necessary for homeostasis. In fact, the average sodium
constant over this wide range of sodium intakes, with or without
intake for individuals in industrialized cultures eating
aldosterone feedback control. (Courtesy Dr. David B. Young.)
processed foods usually ranges between
100 and
200 mEq/day, even though humans can survive and
function normally on 10 to 20 mEq/day. Thus, most
sodium intake more than sixfold under two conditions:
people eat far more sodium than is necessary for home-
(1) under normal conditions and (2) after the aldos-
ostasis, and there is evidence that our usual high sodium
terone feedback system has been blocked by removing
intake may contribute to cardiovascular disorders such
the adrenal glands and infusing the animals with aldos-
as hypertension.
terone at a constant rate so that plasma levels could not
Salt appetite is due in part to the fact that animals and
change upward or downward. Note that when sodium
humans like salt and eat it regardless of whether they
intake was increased sixfold, plasma concentration
are salt-deficient. There is also a regulatory component
changed only about 1 to 2 per cent in either case. This
to salt appetite in which there is a behavioral drive to
indicates that even without a functional aldosterone
obtain salt when there is sodium deficiency in the body.
feedback system, plasma sodium concentration can be
This is particularly important in herbivores, which nat-
well regulated. The same type of experiment has been
urally eat a low-sodium diet, but salt craving may also
conducted after blocking angiotensin II formation, with
be important in humans who have extreme deficiency
the same result.
of sodium, such as occurs in Addison’s disease. In this
There are two primary reasons why changes in
instance, there is deficiency of aldosterone secretion,
angiotensin II and aldosterone do not have a major
which causes excessive loss of sodium in the urine and
effect on plasma sodium concentration. First, as dis-
leads to decreased extracellular fluid volume and
cussed earlier, angiotensin II and aldosterone increase
decreased sodium concentration; both of these changes
both sodium and water reabsorption by the renal
elicit the desire for salt.
tubules, leading to increases in extracellular fluid
In general, the two primary stimuli that are believed to
volume and sodium quantity but little change in sodium
increase salt appetite are (1) decreased extracellular fluid
concentrations. Second, as long as the ADH-thirst mech-
sodium concentration and (2) decreased blood volume
anism is functional, any tendency toward increased
or blood pressure, associated with circulatory insuffi-
plasma sodium concentration is compensated for by
ciency. These are the same major stimuli that elicit thirst.
increased water intake or increased plasma ADH secre-
The neuronal mechanism for salt appetite is analo-
tion, which tends to dilute the extracellular fluid back
gous to that of the thirst mechanism. Some of the same
toward normal. The ADH-thirst system far overshad-
neuronal centers in the AV3V region of the brain seem
ows the angiotensin II and aldosterone systems for reg-
to be involved, because lesions in this region frequently
ulating sodium concentration under normal conditions.
affect both thirst and salt appetite simultaneously in
Even in patients with primary aldosteronism, who have
animals. Also, circulatory reflexes elicited by low blood
extremely high levels of aldosterone, the plasma sodium
pressure or decreased blood volume affect both thirst
concentration usually increases only about 3 to 5 mEq/L
and salt appetite at the same time.
above normal.
Under extreme conditions, caused by complete loss of
aldosterone secretion because of adrenalectomy or in
References
patients with Addison’s disease
(severely impaired
secretion or total lack of aldosterone), there is tremen-
Cowley AW Jr, Mori T, Mattson D, Zou AP: Role of renal
dous loss of sodium by the kidneys, which can lead to
NO production in the regulation of medullary blood flow.
reductions in plasma sodium concentration. One of the
Am J Physiol Regul Integr Comp Physiol 284:R1355, 2003.
364
Unit V The Body Fluids and Kidneys
Dwyer TM, Schmidt-Nielsen B: The renal pelvis: machinery
Pallone TL, Turner MR, Edwards A, Jamison RL: Counter-
that concentrates urine in the papilla. News Physiol Sci
current exchange in the renal medulla. Am J Physiol
18:1, 2003.
Regul Integr Comp Physiol 284:R1153, 2003.
Edwards A, Delong MJ, Pallone TL: Interstitial water and
Robertson GL: Vasopressin. In Seldin DW, Giebisch G (eds):
solute recovery by inner medullary vasa recta. Am J
The Kidney—Physiology and Pathophysiology, 3rd ed.
Physiol Renal Physiol 278:F257, 2000.
New York: Raven Press, 2000.
Fitzsimons JT: Angiotensin, thirst, and sodium appetite.
Sands JM, Layton HE: Urine concentrating mechanism and
Physiol Rev 78:583, 1998.
its regulation. In Seldin DW, Giebisch G
(eds): The
Knepper MA, Saidel GM, Hascall VC, Dwyer T: Concentra-
Kidney—Physiology and Pathophysiology, 3rd ed. New
tion of solutes in the renal inner medulla: interstitial
York: Raven Press, 2000.
hyaluronan as a mechano-osmotic transducer. Am J
Sands JM: Molecular mechanisms of urea transport. J Mem-
Physiol Renal Physiol 284:F433, 2003.
brane Biol 191:149, 2003.
Kozono D, Yasui M, King LS, Agre P: Aquaporin water chan-
Stricker EM, Sved AF: Controls of vasopressin secretion and
nels: atomic structure molecular dynamics meet clinical
thirst: similarities and dissimilarities in signals. Physiol
medicine. J Clin Invest 109:1395, 2002.
Behav 77:731, 2002.
McKinley MJ, Johnson AK: The physiological regulation of
Verbalis JG: Diabetes insipidus. Rev Endocr Metab Disord
thirst and fluid intake. News Physiol Sci 19:1, 2004.
4:177, 2003.
Morello JP, Bichet DG: Nephrogenic diabetes insipidus.
Annu Rev Physiol 63:607, 2001.
Nielsen S, Frokiaer J, Marples D, et al: Aquaporins in the
kidney: from molecules to medicine. Physiol Rev 82:205,
2002.
C H A P T E R
2
9
Renal Regulation of Potassium,
Calcium, Phosphate, and
Magnesium; Integration
of Renal Mechanisms for
Control of Blood Volume and
Extracellular Fluid Volume
Regulation of Potassium
Excretion and Potassium
Concentration in
Extracellular Fluid
Extracellular fluid potassium concentration nor-
mally is regulated precisely at about 4.2 mEq/L,
seldom rising or falling more than ± 0.3 mEq/L. This precise control is neces-
sary because many cell functions are very sensitive to changes in extracellular
fluid potassium concentration. For instance, an increase in plasma potassium
concentration of only 3 to 4 mEq/L can cause cardiac arrhythmias, and higher
concentrations can lead to cardiac arrest or fibrillation.
A special difficulty in regulating extracellular potassium concentration is the
fact that over 98 per cent of the total body potassium is contained in the cells
and only 2 per cent in the extracellular fluid (Figure 29-1). For a 70-kilogram
adult, who has about 28 liters of intracellular fluid (40 per cent of body weight)
and 14 liters of extracellular fluid (20 per cent of body weight), about 3920 mil-
liequivalents of potassium are inside the cells and only about 59 milliequiva-
lents are in the extracellular fluid. Also, the potassium contained in a single meal
is often as high as 50 milliequivalents, and the daily intake usually ranges
between 50 and 200 mEq/day; therefore, failure to rapidly rid the extracellular
fluid of the ingested potassium could cause life-threatening hyperkalemia
(increased plasma potassium concentration). Likewise, a small loss of potassium
from the extracellular fluid could cause severe hypokalemia (low plasma potas-
sium concentration) in the absence of rapid and appropriate compensatory
responses.
Maintenance of potassium balance depends primarily on excretion by the
kidneys because the amount excreted in the feces is only about 5 to 10 per cent
of the potassium intake. Thus, the maintenance of normal potassium balance
requires the kidneys to adjust their potassium excretion rapidly and precisely
to wide variations in intake, as is also true for most other electrolytes.
Control of potassium distribution between the extracellular and intracellular
compartments also plays an important role in potassium homeostasis. Because
over 98 per cent of the total body potassium is contained in the cells, they can
serve as an overflow site for excess extracellular fluid potassium during hyper-
kalemia or as a source of potassium during hypokalemia. Thus, redistribution
of potassium between the intra- and extracellular fluid compartments pro-
vides a first line of defense against changes in extracellular fluid potassium
concentration.
365
366
Unit V The Body Fluids and Kidneys
plasma potassium concentration after eating a meal is
K+ intake
100 mEq/day
much greater than normal. Injections of insulin,
however, can help to correct the hyperkalemia.
Extracellular
Intracellular
fluid K+
fluid K+
Aldosterone Increases Potassium Uptake into Cells. Increased
potassium intake also stimulates secretion of aldos-
terone, which increases cell potassium uptake. Excess
4.2 mEq/L
140 mEq/L
x 14 L
x 28 L
aldosterone secretion
(Conn’s syndrome) is almost
invariably associated with hypokalemia, due in part to
movement of extracellular potassium into the cells.
59 m Eq
3920 mEq
Conversely, patients with deficient aldosterone produc-
tion (Addison’s disease) often have clinically significant
hyperkalemia due to accumulation of potassium in
K+ output
the extracellular space as well as to renal retention of
Urine 92 mEq/day
potassium.
Feces 8 mEq/day
100 mEq/day
b-Adrenergic Stimulation Increases Cellular Uptake of Potassium.
Figure 29-1
Increased secretion of catecholamines, especially epi-
nephrine, can cause movement of potassium from the
Normal potassium intake, distribution of potassium in the body
extracellular to the intracellular fluid, mainly by activa-
fluids, and potassium output from the body.
tion of b2-adrenergic receptors. Treatment of hyperten-
sion with b-adrenergic receptor blockers, such as
propranolol, causes potassium to move out of the cells
and creates a tendency toward hyperkalemia.
Table 29-1
Factors That Can Alter Potassium Distribution Between the
Acid-Base Abnormalities Can Cause Changes in Potassium Distri-
Intra- and Extracellular Fluid
bution. Metabolic acidosis increases extracellular potas-
sium concentration, in part by causing loss of potassium
from the cells, whereas metabolic alkalosis decreases
Factors That Shift K+ into Cells
Factors That Shift K+ Out of Cells
extracellular fluid potassium concentration. Although
(Decrease Extracellular [K+])
(Increase Extracellular [K+])
the mechanisms responsible for the effect of hydrogen
• Insulin
• Insulin deficiency (diabetes
ion concentration on potassium internal distribution are
• Aldosterone
mellitus)
not completely understood, one effect of increased
• b-adrenergic stimulation
• Aldosterone deficiency
hydrogen ion concentration is to reduce the activity
• Alkalosis
(Addison’s disease)
of the sodium-potassium adenosine triphosphatase
• b-adrenergic blockade
(ATPase) pump. This in turn decreases cellular uptake
• Acidosis
of potassium and raises extracellular potassium
• Cell lysis
concentration.
• Strenuous exercise
• Increased extracellular fluid
osmolarity
Cell Lysis Causes Increased Extracellular Potassium Concentra-
tion. As cells are destroyed, the large amounts of potas-
sium contained in the cells are released into the
extracellular compartment. This can cause significant
hyperkalemia if large amounts of tissue are destroyed,
Regulation of Internal Potassium
as occurs with severe muscle injury or with red blood
Distribution
cell lysis.
After ingestion of a normal meal, extracellular fluid
Strenuous Exercise Can Cause Hyperkalemia by Releasing Potas-
potassium concentration would rise to a lethal level if
sium from Skeletal Muscle. During prolonged exercise,
the ingested potassium did not rapidly move into the
potassium is released from skeletal muscle into the
cells. For example, absorption of 40 milliequivalents of
extracellular fluid. Usually the hyperkalemia is mild, but
it may be clinically significant after heavy exercise in
potassium (the amount contained in a meal rich in veg-
patients treated with b-adrenergic blockers or in indi-
etables and fruit) into an extracellular fluid volume of
viduals with insulin deficiency. In rare instances, hyper-
14 liters would raise plasma potassium concentration
kalemia after exercise may be severe enough to cause
by about 2.9 mEq/L if all the potassium remained in
cardiac arrhythmias and sudden death.
the extracellular compartment. Fortunately, most of
the ingested potassium rapidly moves into the cells
Increased Extracellular Fluid Osmolarity Causes Redistribution of
until the kidneys can eliminate the excess. Table 29-1
Potassium from the Cells to Extracellular Fluid. Increased
summarizes some of the factors that can influence the
extracellular fluid osmolarity causes osmotic flow of
distribution of potassium between the intra- and extra-
water out of the cells. The cellular dehydration increases
cellular compartments.
intracellular potassium concentration, thereby pro-
moting diffusion of potassium out of the cells and
Insulin Stimulates Potassium Uptake into Cells. One of the
increasing extracellular fluid potassium concentration.
most important factors that increase cell potassium
Decreased extracellular fluid osmolarity has the oppo-
uptake after a meal is insulin. In people who have
site effect. In diabetes mellitus, large increases in plasma
insulin deficiency owing to diabetes mellitus, the rise in
glucose raise extracellular osmolarity, causing cell
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
367
dehydration and movement of potassium from the cells
segments, potassium can at times be reabsorbed or at
into the extracellular fluid.
other times be secreted, depending on the needs of the
body. With a normal potassium intake of 100 mEq/day,
the kidneys must excrete about 92 mEq/day (the
Overview of Renal Potassium
remaining 8 milliequivalents are lost in the feces).
Excretion
About one third (31 mEq/day) of this amount of
potassium is secreted into the distal and collecting
Potassium excretion is determined by the sum of three
tubules.
renal processes: (1) the rate of potassium filtration
With high potassium intakes, the required extra
(GFR multiplied by the plasma potassium concentra-
excretion of potassium is achieved almost entirely by
tion), (2) the rate of potassium reabsorption by the
increasing the secretion of potassium into the distal
tubules, and (3) the rate of potassium secretion by
and collecting tubules. In fact, with extremely high
the tubules. The normal rate of potassium filtration is
potassium diets, the rate of potassium excretion can
about 756 mEq/day (GFR, 180 L/day multiplied by
exceed the amount of potassium in the glomerular fil-
plasma potassium, 4.2 mEq/L); this rate of filtration is
trate, indicating a powerful mechanism for secreting
usually relatively constant because of the autoregula-
potassium.
tory mechanisms for GFR discussed previously and
When potassium intake is reduced below normal,
the precision with which plasma potassium concentra-
the secretion rate of potassium in the distal and
tion is regulated. Severe decreases in GFR in certain
collecting tubules decreases, causing a reduction in
renal diseases, however, can cause serious potassium
urinary potassium secretion. With extreme reductions
accumulation and hyperkalemia.
in potassium intake, there is net reabsorption of potas-
Figure 29-2 summarizes the tubular handling of
sium in the distal segments of the nephron, and potas-
potassium under normal conditions. About 65 per cent
sium excretion can fall to 1 per cent of the potassium
of the filtered potassium is reabsorbed in the proximal
in the glomerular filtrate (to less than 10 mEq/day).
tubule. Another 25 to 30 per cent of the filtered potas-
With potassium intakes below this level, severe
sium is reabsorbed in the loop of Henle, especially in
hypokalemia can develop.
the thick ascending part where potassium is actively
Thus, most of the day-to-day regulation of potas-
co-transported along with sodium and chloride. In
sium excretion occurs in the late distal and cortical
both the proximal tubule and the loop of Henle, a rel-
collecting tubules, where potassium can be either
atively constant fraction of the filtered potassium load
reabsorbed or secreted, depending on the needs of the
is reabsorbed. Changes in potassium reabsorption in
body. In the next section, we consider the basic mech-
these segments can influence potassium excretion, but
anisms of potassium secretion and the factors that reg-
most of the day-to-day variation of potassium excre-
ulate this process.
tion is not due to changes in reabsorption in the prox-
imal tubule or loop of Henle.
Potassium Secretion by Principal
Most Daily Variation in Potassium Excretion Is Caused by
Cells of Late Distal and Cortical
Changes in Potassium Secretion in Distal and Collecting
Collecting Tubules
Tubules. The most important sites for regulating potas-
sium excretion are the principal cells of the late distal
The cells in the late distal and cortical collecting
tubules and cortical collecting tubules. In these tubular
tubules that secrete potassium are called principal cells
65%
(491 mEq/day)
4%
(31 mEq/day)
756 mEq/day
(180 L/day x 4.2 mEq/L)
27%
Figure 29-2
(204 mEq/day)
Renal tubular sites of potassium reabsorp-
tion and secretion. Potassium is reab-
sorbed in the proximal tubule and in the
ascending loop of Henle, so that only about
8 per cent of the filtered load is delivered to
the distal tubule. Secretion of potassium
into the late distal tubules and collecting
ducts adds to the amount delivered, so that
the daily excretion is about 12 per cent of
the potassium filtered at the glomerular
capillaries. The percentages indicate how
much of the filtered load is reabsorbed or
secreted into the different tubular seg-
12%
ments.
(92 mEq/day)
368
Unit V The Body Fluids and Kidneys
Renal
Principal
Tubular
reabsorption occurs through the intercalated cells;
interstitial
cells
lumen
although this reabsorptive process is not completely
fluid
understood, one mechanism believed to contribute
is a hydrogen-potassium ATPase transport mechanism
located in the luminal membrane. This transporter reab-
Na+
sorbs potassium in exchange for hydrogen ions secreted
into the tubular lumen, and the potassium then diffuses
Na+
Na+
through the basolateral membrane of the cell into the
ATP
blood. This transporter is necessary to allow potassium
K+
K+
reabsorption during extracellular fluid potassium deple-
tion, but under normal conditions it plays a small role
in controlling the excretion of potassium.
K+
Summary of Factors That Regulate
Potassium Secretion: Plasma
0 mV
-70 mV
-50 mV
Potassium Concentration,
Aldosterone, Tubular Flow Rate, and
Figure 29-3
Hydrogen Ion Concentration
Mechanisms of potassium secretion and sodium reabsorption by
Because normal regulation of potassium excretion
the principal cells of the late distal and collecting tubules.
occurs mainly as a result of changes in potassium
secretion by the principal cells of the late distal and
collecting tubules, we will discuss the primary factors
and make up about 90 per cent of the epithelial cells
that influence secretion by these cells. The most impor-
in these regions. Figure 29-3 shows the basic cellular
tant factors that stimulate potassium secretion by the
mechanisms of potassium secretion by the principal
principal cells include (1) increased extracellular fluid
cells.
potassium concentration, (2) increased aldosterone,
Secretion of potassium from the blood into the
and (3) increased tubular flow rate.
tubular lumen is a two-step process, beginning with
One factor that decreases potassium secretion is
uptake from the interstitium into the cell by the
increased hydrogen ion concentration (acidosis).
sodium-potassium ATPase pump in the basolateral
membrane of the cell; this pump moves sodium out of
Increased Extracellular Fluid Potassium Concentration Stimu-
the cell into the interstitium and at the same time
lates Potassium Secretion. The rate of potassium secre-
moves potassium to the interior of the cell. The second
tion in the late distal and cortical collecting tubules is
step of the process is passive diffusion of potassium
directly stimulated by increased extracellular fluid
from the interior of the cell into the tubular fluid. The
potassium concentration, leading to increases in potas-
sodium-potassium ATPase pump creates a high intra-
sium excretion, as shown in Figure 29-4. This effect
cellular potassium concentration, which provides the
is especially pronounced when extracellular fluid
driving force for passive diffusion of potassium from
potassium concentration rises above about 4.1 mEq/L,
the cell into the tubular lumen. The luminal membrane
slightly less than the normal concentration. Increased
of the principal cells is highly permeable to potassium.
plasma potassium concentration, therefore, serves as
One reason for this high permeability is that there are
one of the most important mechanisms for increasing
special channels that are specifically permeable to
potassium secretion and regulating extracellular fluid
potassium ions, thus allowing these ions to diffuse
potassium ion concentration.
across the membrane.
There are three mechanisms by which increased
extracellular fluid potassium concentration raises
Control of Potassium Secretion by Principal Cells. The
potassium secretion: (1) Increased extracellular fluid
primary factors that control potassium secretion by the
potassium concentration stimulates the sodium-
principal cells of the late distal and cortical collecting
potassium ATPase pump, thereby increasing potas-
tubules are (1) the activity of the sodium-potassium
sium uptake across the basolateral membrane. This in
ATPase pump, (2) the electrochemical gradient for
turn increases intracellular potassium ion concentra-
potassium secretion from the blood to the tubular
tion, causing potassium to diffuse across the luminal
lumen, and (3) the permeability of the luminal mem-
membrane into the tubule. (2) Increased extracellular
brane for potassium. These three determinants of
potassium concentration increases the potassium gra-
potassium secretion are in turn regulated by the
dient from the renal interstitial fluid to the interior of
factors discussed later.
the epithelial cell; this reduces backleakage of potas-
sium ions from inside the cells through the basolateral
Intercalated Cells Can Reabsorb Potassium During Potassium
membrane. (3) Increased potassium concentration
Depletion. In circumstances associated with severe
potassium depletion, there is a cessation of potassium
stimulates aldosterone secretion by the adrenal cortex,
secretion and actually a net reabsorption of potas-
which further stimulates potassium secretion, as dis-
sium in the late distal and collecting tubules. This
cussed next.
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
369
4
70
Effect of aldosterone
Effect of extracellular
60
K+ concentration
3
50
40
2
30
20
1
10
0
0
3.0
3.5
4.0
4.5
5.0
5.5
6.0
6.5
0
1
2
3
4
5
Serum potassium concentration (mEq/L)
Plasma aldosterone (times normal)
1
2
3
4
5
Extracellular potassium concentration
(mEq/L)
Figure 29-5
Effect of extracellular fluid potassium ion concentration on plasma
aldosterone concentration. Note that small changes in potassium
Figure 29-4
concentration cause large changes in aldosterone concentration.
Effect of plasma aldosterone concentration (red line) and extra-
cellular potassium ion concentration (black line) on the rate of
urinary potassium excretion. These factors stimulate potassium
secretion by the principal cells of the cortical collecting tubules.
1
(Drawn from data in Young DB, Paulsen AW: Interrelated effects
Ald.
of aldosterone and plasma potassium on potassium excretion. Am
J Physiol 244:F28, 1983.)
K+
concentration
K+
4
Aldosterone Stimulates Potassium Secretion. In Chapter 27,
Aldosterone
we discuss the fact that aldosterone stimulates active
concentration
reabsorption of sodium ions by the principal cells of
K+ excretion
the late distal tubules and collecting ducts. This effect
3
is mediated through a sodium-potassium ATPase
K+
excretion
pump that transports sodium outward through the
+
-
Ald.
basolateral membrane of the cell and into the blood at
2
the same time that it pumps potassium into the cell.
K+ intake
Thus, aldosterone also has a powerful effect to control
the rate at which the principal cells secrete potassium.
A second effect of aldosterone is to increase the per-
Figure 29-6
meability of the luminal membrane for potassium,
further adding to the effectiveness of aldosterone in
Basic feedback mechanism for control of extracellular fluid potas-
stimulating potassium secretion. Therefore, aldos-
sium concentration by aldosterone (Ald.).
terone has a powerful effect to increase potassium
excretion, as shown in Figure 29-4.
The effect of potassium ion concentration to stimu-
Increased Extracellular Potassium Ion Concentration Stimu-
late aldosterone secretion is part of a powerful feed-
lates Aldosterone Secretion. In negative feedback control
back system for regulating potassium excretion, as
systems, the factor that is controlled usually has a
shown in Figure 29-6. In this feedback system, an
feedback effect on the controller. In the case of the
increase in plasma potassium concentration stimulates
aldosterone-potassium control system, the rate of
aldosterone secretion and, therefore, increases the
aldosterone secretion by the adrenal gland is con-
blood level of aldosterone (block 1). The increase in
trolled strongly by extracellular fluid potassium ion
blood aldosterone then causes a marked increase in
concentration. Figure 29-5 shows that an increase in
potassium excretion by the kidneys (block 2). The
plasma potassium concentration of about 3 mEq/L can
increased potassium excretion then reduces the extra-
increase plasma aldosterone concentration from
cellular fluid potassium concentration back toward
nearly 0 to as high as 60 ng/100 ml, a concentration
normal (blocks 3 and 4). Thus, this feedback mecha-
almost 10 times normal.
nism acts synergistically with the direct effect of
370
Unit V The Body Fluids and Kidneys
K+ intake
4.8
Plasma K+
4.6
concentration
Aldosterone
4.4
K+ secretion
Normal
Cortical collecting
tubules
4.2
Aldosterone
4.0
system
blocked
K+ excretion
3.8
Figure 29-7
0
30
60
90
120
150
180
210
Primary mechanisms by which high potassium intake raises
Potassium intake (mEq/day)
potassium excretion. Note that increased plasma potassium con-
centration directly raises potassium secretion by the cortical col-
lecting tubules and indirectly increases potassium secretion by
raising plasma aldosterone concentration.
Figure 29-8
Effect of large changes in potassium intake on extracellular fluid
potassium concentration under normal conditions (red line) and
increased extracellular potassium concentration to
after the aldosterone feedback had been blocked (blue line). Note
elevate potassium excretion when potassium intake is
that after blockade of the aldosterone system, regulation of potas-
raised (Figure 29-7).
sium concentration was greatly impaired. (Courtesy Dr. David B.
Young.)
Blockade of Aldosterone Feedback System Greatly Impairs
Control of Potassium Concentration. In the absence of
aldosterone secretion, as occurs in patients with
impairment of potassium regulation is observed in
Addison’s disease, renal secretion of potassium is
humans with poorly functioning aldosterone feedback
impaired, thus causing extracellular fluid potassium
systems, such as occurs in patients with either primary
concentration to rise to dangerously high levels. Con-
aldosteronism (too much aldosterone) or Addison’s
versely, with excess aldosterone secretion (primary
disease (too little aldosterone).
aldosteronism), potassium secretion becomes greatly
increased, causing potassium loss by the kidneys and
Increased Distal Tubular Flow Rate Stimulates Potassium
thus leading to hypokalemia.
Secretion. A rise in distal tubular flow rate, as occurs
The special quantitative importance of the aldos-
with volume expansion, high sodium intake, or diuretic
terone feedback system in controlling potassium con-
drug treatment, stimulates potassium secretion. Con-
centration is shown in Figure 29-8. In this experiment,
versely, a decrease in distal tubular flow rate, as caused
potassium intake was increased almost sevenfold in
by sodium depletion, reduces potassium secretion.
dogs under two conditions: (1) under normal condi-
The mechanism for the effect of high-volume flow
tions and (2) after the aldosterone feedback system
rate is as follows: When potassium is secreted into the
had been blocked by removing the adrenal glands and
tubular fluid, the luminal concentration of potassium
placing the animals on a fixed rate of aldosterone infu-
increases, thereby reducing the driving force for potas-
sion so that plasma aldosterone concentration could
sium diffusion across the luminal membrane. With
neither increase nor decrease.
increased tubular flow rate, the secreted potassium is
Note that in the normal animals, a sevenfold
continuously flushed down the tubule, so that the rise
increase in potassium intake caused only a slight in-
in tubular potassium concentration becomes mini-
crease in potassium concentration, from 4.2 to 4.3 mEq/
mized. Therefore, net potassium secretion is stimu-
L. Thus, when the aldosterone feedback system is
lated by increased tubular flow rate.
functioning normally, potassium concentration is pre-
The effect of increased tubular flow rate is especially
cisely controlled, despite large changes in potassium
important in helping to preserve normal potassium
intake.
excretion during changes in sodium intake. For
When the aldosterone feedback system was
example, with a high sodium intake, there is decreased
blocked, the same increases in potassium intake
aldosterone secretion, which by itself would tend to
caused a much larger increase in potassium concen-
decrease the rate of potassium secretion and, there-
tration, from 3.8 to almost 4.7 mEq/L. Thus, control of
fore, reduce urinary excretion of potassium. However,
potassium concentration is greatly impaired when the
the high distal tubular flow rate that occurs with a high
aldosterone feedback system is blocked. A similar
sodium intake tends to increase potassium secretion
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
371
to a loss of potassium, whereas acute acidosis leads to
Na+ intake
decreased potassium excretion.
Proximal
Control of Renal Calcium
Aldosterone
GFR
tubular Na+
reabsorption
Excretion and Extracellular
Calcium Ion Concentration
Distal tubular
The mechanisms for regulating calcium ion concen-
flow rate
tration are discussed in detail in Chapter 79, along with
the endocrinology of the calcium-regulating hormones
parathyroid hormone (PTH) and calcitonin. There-
-
K+ secretion
+
fore, calcium ion regulation is discussed only briefly in
Cortical collecting
this chapter.
ducts
Extracellular fluid calcium ion concentration
normally remains tightly controlled within a few
percentage points of its normal level,
2.4 mEq/L.
When calcium ion concentration falls to low levels
Unchanged K+
excretion
(hypocalcemia), the excitability of nerve and muscle
cells increases markedly and can in extreme cases
result in hypocalcemic tetany. This is characterized by
Figure 29-9
spastic skeletal muscle contractions. Hypercalcemia
Effect of high sodium intake on renal excretion of potassium. Note
(increased calcium concentration) depresses neuromus-
that a high-sodium diet decreases plasma aldosterone, which
cular excitability and can lead to cardiac arrhythmias.
tends to decrease potassium secretion by the cortical collecting
About 50 per cent of the total calcium in the plasma
tubules. However, the high-sodium diet simultaneously increases
(5 mEq/L) exists in the ionized form, which is the form
fluid delivery to the cortical collecting duct, which tends to
that has biological activity at cell membranes. The
increase potassium secretion. The opposing effects of a high-
sodium diet counterbalance each other, so that there is little
remainder is either bound to the plasma proteins
change in potassium excretion.
(about 40 per cent) or complexed in the non-ionized
form with anions such as phosphate and citrate (about
10 per cent).
(Figure 29-9), as discussed in the previous paragraph.
Changes in plasma hydrogen ion concentration can
Therefore, the two effects of high sodium intake,
influence the degree of calcium binding to plasma pro-
decreased aldosterone secretion and the high tubular
teins. With acidosis, less calcium is bound to the plasma
flow rate, counterbalance each other, so that there is
proteins. Conversely, in alkalosis, a greater amount of
little change in potassium excretion. Likewise, with a
calcium is bound to the plasma proteins. Therefore,
low sodium intake, there is little change in potassium
patients with alkalosis are more susceptible to hypocal-
excretion because of the counterbalancing effects of
cemic tetany.
increased aldosterone secretion and decreased tubular
As with other substances in the body, the intake of
flow rate on potassium secretion.
calcium must be balanced with the net loss of calcium
over the long term. Unlike ions such as sodium and
Acute Acidosis Decreases Potassium Secretion. Acute
chloride, however, a large share of calcium excretion
increases in hydrogen ion concentration of the extra-
occurs in the feces. The usual rate of dietary calcium
cellular fluid
(acidosis) reduce potassium secretion,
intake is about 1000 mg/day, with about 900 mg/day of
whereas decreased hydrogen ion concentration (alka-
calcium excreted in the feces. Under certain condi-
losis) increases potassium secretion. The primary
tions, fecal calcium excretion can exceed calcium
mechanism by which increased hydrogen ion concen-
ingestion because calcium can also be secreted into
tration inhibits potassium secretion is by reducing the
the intestinal lumen. Therefore, the gastrointestinal
activity of the sodium-potassium ATPase pump. This
tract and the regulatory mechanisms that influence
in turn decreases intracellular potassium concentra-
intestinal calcium absorption and secretion play a
tion and subsequent passive diffusion of potassium
major role in calcium homeostasis, as discussed in
across the luminal membrane into the tubule.
Chapter 79.
Almost all the calcium in the body (99 per cent) is
With more prolonged acidosis, lasting over a period of
stored in the bone, with only about 1 per cent in the
several days, there is an increase in urinary potassium
extracellular fluid and 0.1 per cent in the intracellular
excretion. The mechanism for this effect is due in part
fluid. The bone, therefore, acts as a large reservoir
to an effect of chronic acidosis to inhibit proximal
for storing calcium and as a source of calcium when
tubular sodium chloride and water reabsorption, which
extracellular fluid calcium concentration tends to
increases distal volume delivery, thereby stimulating
the secretion of potassium. This effect overrides
decrease.
the inhibitory effect of hydrogen ions on the sodium-
One of the most important regulators of bone uptake
potassium ATPase pump. Thus, chronic acidosis leads
and release of calcium is PTH. When extracellular fluid
372
Unit V The Body Fluids and Kidneys
proteins or complexed with anions such as phosphate.
[Ca++]
Therefore, only about
50 per cent of the plasma
calcium can be filtered at the glomerulus. Normally,
about 99 per cent of the filtered calcium is reabsorbed
by the tubules, with only about 1 per cent of the fil-
Vitamin D3
PTH
tered calcium being excreted. About 65 per cent of the
activation
filtered calcium is reabsorbed in the proximal tubule,
25 to 30 per cent is reabsorbed in the loop of Henle,
and 4 to 9 per cent is reabsorbed in the distal and col-
lecting tubules. This pattern of reabsorption is similar
Intestinal Ca++
Renal Ca++
Ca++ release
reabsorption
reabsorption
from bones
to that for sodium.
As is true with the other ions, calcium excretion is
adjusted to meet the body’s needs. With an increase in
Figure 29-10
calcium intake, there is also increased renal calcium
Compensatory responses to decreased plasma ionized calcium
excretion, although much of the increase of calcium
concentration mediated by parathyroid hormone
(PTH) and
intake is eliminated in the feces. With calcium deple-
vitamin D.
tion, calcium excretion by the kidneys decreases as a
result of enhanced tubular reabsorption.
One of the primary controllers of renal tubular
calcium concentration falls below normal, the parathy-
calcium reabsorption is PTH. With increased levels of
roid glands are directly stimulated by the low calcium
PTH, there is increased calcium reabsorption in the
levels to promote increased secretion of PTH. This
thick ascending loops of Henle and distal tubules,
hormone then acts directly on the bones to increase
which reduces urinary excretion of calcium. Con-
the resorption of bone salts (release of salts from the
versely, reduction of PTH promotes calcium excretion
bones) and, therefore, to release large amounts of
by decreasing reabsorption in the loops of Henle and
calcium into the extracellular fluid, thereby returning
distal tubules.
calcium levels back toward normal. When calcium ion
In the proximal tubule, calcium reabsorption usually
concentration is elevated, PTH secretion decreases, so
parallels sodium and water reabsorption. Therefore,
that almost no bone resorption now occurs; instead,
in instances of extracellular volume expansion or
excess calcium is deposited in the bones because of
increased arterial pressure—both of which decrease
new bone formation. Thus, the day-to-day regulation
proximal sodium and water reabsorption—there is
of calcium ion concentration is mediated in large part
also reduction in calcium reabsorption and, con-
by the effect of PTH on bone resorption.
sequently, increased urinary excretion of calcium.
The bones, however, do not have an inexhaustible
Conversely, with extracellular volume contraction or
supply of calcium. Therefore, over the long term, the
decreased blood pressure, calcium excretion decreases
intake of calcium must be balanced with calcium
primarily because of increased proximal tubular
excretion by the gastrointestinal tract and the kidneys.
reabsorption.
The most important regulator of calcium reabsorption
Another factor that influences calcium reabsorption
at both of these sites is PTH. Thus, PTH regulates
is the plasma concentration of phosphate. An increase
plasma calcium concentration through three main
in plasma phosphate stimulates PTH, which increases
effects: (1) by stimulating bone resorption; (2) by stim-
calcium reabsorption by the renal tubules, thereby
ulating activation of vitamin D, which then increases
reducing calcium excretion. The opposite occurs with
intestinal reabsorption of calcium; and (3) by directly
reduction in plasma phosphate concentration.
increasing renal tubular calcium reabsorption (Figure
Calcium reabsorption is also stimulated by meta-
29-10). The control of gastrointestinal calcium reab-
bolic acidosis and inhibited by metabolic alkalosis.
sorption and calcium exchange in the bones is dis-
Most of the effect of hydrogen ion concentration on
cussed elsewhere, and the remainder of this section
calcium excretion results from changes in calcium
focuses on the mechanisms that control renal calcium
reabsorption in the distal tubule.
excretion.
A summary of the factors that are known to influ-
ence calcium excretion by the renal tubules is shown
in Table 29-2.
Control of Calcium Excretion
by the Kidneys
Regulation of Renal Phosphate
Because calcium is both filtered and reabsorbed in the
Excretion
kidneys but not secreted, the rate of renal calcium
excretion is calculated as
Phosphate excretion by the kidneys is controlled
primarily by an overflow mechanism that can be
Renal calcium excretion = Calcium filtered
explained as follows: The renal tubules have a normal
- Calcium reabsorbed
transport maximum for reabsorbing phosphate of
Only about 50 per cent of the plasma calcium is
about
0.1 mM/min. When less than this amount
ionized, with the remainder being bound to the plasma
of phosphate is present in the glomerular filtrate,
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
373
normally excrete about 10 to 15 per cent of the mag-
Table 29-2
nesium in the glomerular filtrate.
Factors That Alter Renal Calcium Excretion
Renal excretion of magnesium can increase
markedly during magnesium excess or can decrease to
Ø Calcium Excretion
≠ Calcium Excretion
almost nil during magnesium depletion. Because mag-
nesium is involved in many biochemical processes in
≠ Parathyroid hormone (PTH)
Ø PTH
Ø Extracellular fluid volume
≠ Extracellular fluid volume
the body, including activation of many enzymes, its
Ø Blood pressure
≠ Blood pressure
concentration must be closely regulated.
≠ Plasma phosphate
Ø Plasma phosphate
Regulation of magnesium excretion is achieved
Metabolic acidosis
Metabolic alkalosis
mainly by changing tubular reabsorption. The proxi-
3
Vitamin D
mal tubule usually reabsorbs only about 25 per cent of
the filtered magnesium. The primary site of reabsorp-
tion is the loop of Henle, where about 65 per cent of
the filtered load of magnesium is reabsorbed. Only a
essentially all the filtered phosphate is reabsorbed.
small amount (usually less than 5 per cent) of the
When more than this is present, the excess is excreted.
filtered magnesium is reabsorbed in the distal and
Therefore, phosphate normally begins to spill into the
collecting tubules.
urine when its concentration in the extracellular fluid
The mechanisms that regulate magnesium excretion
rises above a threshold of about 0.8 mM/L, which gives
are not well understood, but the following distur-
a tubular load of phosphate of about 0.1 mM/min,
bances lead to increased magnesium excretion: (1)
assuming a GFR of 125 ml/min. Because most people
increased extracellular fluid magnesium concentra-
ingest large quantities of phosphate in milk products
tion,
(2) extracellular volume expansion, and
(3)
and meat, the concentration of phosphate is usually
increased extracellular fluid calcium concentration.
maintained above 1 mM/L, a level at which there is
continual excretion of phosphate into the urine.
Changes in tubular phosphate reabsorption can also
Integration of Renal
influence phosphate excretion. For instance, a diet low
in phosphate can, over time, increase the reabsorptive
Mechanisms for Control
transport maximum for phosphate, thereby reducing
of Extracellular Fluid
the tendency for phosphate to spill over into the urine.
PTH can play a significant role in regulating phos-
Extracellular fluid volume is determined mainly by the
phate concentration through two effects: (1) PTH
balance between intake and output of water and salt.
promotes bone resorption, thereby dumping large
In most cases, salt and fluid intakes are dictated by a
amounts of phosphate ions into the extracellular fluid
person’s habits rather than by physiologic control
from the bone salts, and (2) PTH decreases the trans-
mechanisms. Therefore, the burden of extracellular
port maximum for phosphate by the renal tubules,
volume regulation is usually placed on the kidneys,
so that a greater proportion of the tubular phosphate
which must adapt their excretion of salt and water to
is lost in the urine. Thus, whenever plasma PTH is
match intake of salt and water under steady-state
increased, tubular phosphate reabsorption is decreased
conditions.
and more phosphate is excreted. These interrelations
In discussing the regulation of extracellular fluid
among phosphate, PTH, and calcium are discussed in
volume, we also consider the factors that regulate the
more detail in Chapter 79.
amount of sodium chloride in the extracellular fluid,
because changes in extracellular fluid sodium chloride
content usually cause parallel changes in extracellular
Control of Renal Magnesium
fluid volume, provided the antidiuretic hormone
Excretion and Extracellular
(ADH)-thirst mechanisms are also operative. When
the ADH-thirst mechanisms are functioning normally,
Magnesium Ion Concentration
a change in the amount of sodium chloride in the
extracellular fluid is matched by a similar change in
More than one half of the body’s magnesium is stored
the amount of extracellular water, so that osmolality
in the bones. Most of the rest resides within the cells,
and sodium concentration are maintained relatively
with less than 1 per cent located in the extracellular
constant.
fluid. Although the total plasma magnesium concen-
tration is about 1.8 mEq/L, more than one half of this
is bound to plasma proteins. Therefore, the free
Sodium Excretion Is Precisely
ionized concentration of magnesium is only about
0.8 mEq/L.
Matched to Intake Under Steady-State
The normal daily intake of magnesium is about 250
Conditions
to 300 mg/day, but only about one half of this intake is
absorbed by the gastrointestinal tract. To maintain
An important consideration in overall control of
magnesium balance, the kidneys must excrete this
sodium excretion—or excretion of any electrolyte, for
absorbed magnesium, about one half the daily intake
that matter—is that under steady-state conditions,
of magnesium, or 125 to 150 mg/day. The kidneys
excretion by the kidneys is determined by intake. To
374
Unit V The Body Fluids and Kidneys
maintain life, a person must, over the long term,
compensations: (1) increased tubular reabsorption of
excrete almost precisely the amount of sodium
much of the extra sodium chloride filtered, called
ingested. Therefore, even with disturbances that cause
glomerulotubular balance, and (2) macula densa feed-
major changes in kidney function, balance between
back, in which increased sodium chloride delivery to
intake and output of sodium usually is restored within
the distal tubule causes afferent arteriolar constriction
a few days.
and return of GFR toward normal. Likewise, abnor-
If disturbances of kidney function are not too
malities of tubular reabsorption in the proximal tubule
severe, sodium balance may be achieved mainly by
or loop of Henle are partially compensated for by
intrarenal adjustments with minimal changes in extra-
these same intrarenal feedbacks.
cellular fluid volume or other systemic adjustments.
Because neither of these two mechanisms operates
But when perturbations to the kidneys are severe and
perfectly to restore distal sodium chloride delivery all
intrarenal compensations are exhausted, systemic
the way back to normal, changes in either GFR or
adjustments must be invoked, such as changes in blood
tubular reabsorption can lead to significant changes in
pressure, changes in circulating hormones, and alter-
urine sodium and water excretion. When this happens,
ations of sympathetic nervous system activity. These
other feedback mechanisms come into play, such as
adjustments are costly in terms of overall homeostasis
changes in blood pressure and changes in various
because they cause other changes throughout the
hormones, that eventually return sodium excretion to
body that may, in the long run, be damaging. These
equal sodium intake. In the next few sections, we
compensations, however, are necessary because a
review how these mechanisms operate together to
sustained imbalance between fluid and electrolyte
control sodium and water balance and in so doing act
intake and excretion would quickly lead to accumula-
also to control extracellular fluid volume. We should
tion or loss of electrolytes and fluid, causing cardio-
keep in mind, however, that all these feedback mech-
vascular collapse within a few days. Thus, one can view
anisms control renal excretion of sodium and water by
the systemic adjustments that occur in response to
altering either GFR or tubular reabsorption.
abnormalities of kidney function as a necessary trade-
off that brings electrolyte and fluid excretion back in
balance with intake.
Importance of Pressure
Natriuresis and Pressure
Diuresis in Maintaining Body
Sodium Excretion Is Controlled by
Sodium and Fluid Balance
Altering Glomerular Filtration or
Tubular Sodium Reabsorption Rates
One of the most basic and powerful mechanisms for
control of blood volume and extracellular fluid
The two variables that influence sodium and water
volume, as well as for the maintenance of sodium and
excretion are the rates of filtration and the rates of
fluid balance, is the effect of blood pressure on sodium
reabsorption:
and water excretion—called the pressure natriuresis
and pressure diuresis mechanisms, respectively. As
Excretion = Glomerular filtration - Tubular
discussed in Chapter 19, this feedback between the
reabsorption
kidneys and the circulatory system also plays a domi-
GFR normally is about 180 L/day, tubular reab-
nant role in long-term blood pressure regulation.
sorption is
178.5 L/day, and urine excretion is 1.5
Pressure diuresis refers to the effect of increased
L/day. Thus, small changes in GFR or tubular reab-
blood pressure to raise urinary volume excretion,
sorption potentially can cause large changes in renal
whereas pressure natriuresis refers to the rise in
excretion. For example, a 5 per cent increase in GFR
sodium excretion that occurs with elevated blood pres-
(to 189 L/day) would cause a 9 L/day increase in urine
sure. Because pressure diuresis and natriuresis usually
volume, if tubular compensations did not occur; this
occur in parallel, we refer to these mechanisms simply
would quickly cause catastrophic changes in body fluid
as “pressure natriuresis” in the following discussion.
volumes. Similarly, small changes in tubular reabsorp-
Figure 29-11 shows the effect of arterial pressure on
tion, in the absence of compensatory adjustments of
urinary sodium output. Note that acute increases in
GFR, would also lead to dramatic changes in urine
blood pressure of 30 to 50 mm Hg cause a twofold to
volume and sodium excretion. Tubular reabsorption
threefold increase in urinary sodium output. This
and GFR usually are regulated precisely, so that excre-
effect is independent of changes in activity of the sym-
tion by the kidneys can be exactly matched to intake
pathetic nervous system or of various hormones, such
of water and electrolytes.
as angiotensin II, ADH, or aldosterone, because pres-
Even with disturbances that alter GFR or tubular
sure natriuresis can be demonstrated in an isolated
reabsorption, changes in urinary excretion are mini-
kidney that has been removed from the influence of
mized by various buffering mechanisms. For example,
these factors. With chronic increases in blood pressure,
if the kidneys become greatly vasodilated and GFR
the effectiveness of pressure natriuresis is greatly
increases (as can occur with certain drugs or high
enhanced because the increased blood pressure also,
fever), this raises sodium chloride delivery to the
after a short time delay, suppresses renin release and,
tubules, which in turn leads to at least two intrarenal
therefore, decreases formation of angiotensin II and
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
375
aldosterone. As discussed previously, decreased levels
ates to maintain balance between fluid intake and
of angiotensin II and aldosterone inhibit renal tubular
output, as shown in Figure 29-12. This is the same
reabsorption of sodium, thereby amplifying the direct
mechanism that is discussed in Chapter 19 for arterial
effects of increased blood pressure to raise sodium and
pressure control. The extracellular fluid volume, blood
water excretion.
volume, cardiac output, arterial pressure, and urine
output are all controlled at the same time as separate
parts of this basic feedback mechanism.
Pressure Natriuresis and Diuresis Are
During changes in sodium and fluid intake, this feed-
Key Components of a Renal-Body
back mechanism helps to maintain fluid balance and
to minimize changes in blood volume, extracellular
Fluid Feedback for Regulating Body
fluid volume, and arterial pressure as follows:
Fluid Volumes and Arterial Pressure
1.
An increase in fluid intake (assuming that sodium
accompanies the fluid intake) above the level of
The effect of increased blood pressure to raise urine
urine output causes a temporary accumulation of
output is part of a powerful feedback system that oper-
fluid in the body.
2.
As long as fluid intake exceeds urine output, fluid
accumulates in the blood and interstitial spaces,
causing parallel increases in blood volume and
extracellular fluid volume. As discussed later, the
8
actual increases in these variables are usually
Chronic
small because of the effectiveness of this
Acute
6
feedback.
3.
An increase in blood volume raises mean
circulatory filling pressure.
4
4.
An increase in mean circulatory filling pressure
raises the pressure gradient for venous return.
2
5.
An increased pressure gradient for venous return
elevates cardiac output.
0
6.
An increased cardiac output raises arterial
0
20
40
60
80 100 120 140 160 180 200
pressure.
Arterial pressure (mm Hg)
7.
An increased arterial pressure increases urine
output by way of pressure diuresis. The steepness
of the normal pressure natriuresis relation
Figure 29-11
indicates that only a slight increase in blood
pressure is required to raise urinary excretion
Acute and chronic effects of arterial pressure on sodium output
severalfold.
by the kidneys (pressure natriuresis). Note that chronic increases
8.
The increased fluid excretion balances the
in arterial pressure cause much greater increases in sodium
output than those measured during acute increases in arterial
increased intake, and further accumulation of fluid
pressure.
is prevented.
Nonrenal
Fluid
fluid loss
intake
Arterial
Rate of change of
Extracellular
pressure
extracellular
fluid volume
fluid volume
Total peripheral
Arterial pressure
Blood
resistance
volume
Figure 29-12
Basic renal-body fluid feedback
Cardiac
Venous
Mean circulatory
mechanism for control of
output
return
filling pressure
blood volume, extracellular fluid
volume, and arterial pressure.
Solid lines indicate positive
effects, and dashed lines indicate
Heart strength
Vascular
negative effects.
capacity
376
Unit V The Body Fluids and Kidneys
pressure causes a large change in urine output. These
factors work together to provide effective feedback
6
control of blood volume.
Blood volume
The same control mechanisms operate whenever
5
there is a loss of whole blood because of hemorrhage.
Normal range
4
In this case, fluid is retained by the kidneys, and other
Death
parallel processes occur to reconstitute the red blood
3
cells and plasma proteins in the blood. If abnormali-
ties of red blood cell volume remain, such as occurs
2
when there is deficiency of erythropoietin or other
factors needed to stimulate red blood cell production,
1
the plasma volume will simply make up the difference,
0
and the overall blood volume will return essentially to
0
1
2
3
4
5
6
7
8
normal despite the low red blood cell mass.
Daily fluid intake
(water and electrolytes) (L/day)
Distribution of Extracellular
Fluid Between the Interstitial
Figure 29-13
Spaces and Vascular System
Approximate effect of changes in daily fluid intake on blood
volume. Note that blood volume remains relatively constant in the
From Figure 29-12 it is apparent that blood volume
normal range of daily fluid intakes.
and extracellular fluid volume are usually controlled
in parallel with each other. Ingested fluid initially goes
into the blood, but it rapidly becomes distributed
Thus, the renal-body fluid feedback mechanism
between the interstitial spaces and the plasma. There-
operates to prevent continuous accumulation of salt
fore, blood volume and extracellular fluid volume
and water in the body during increased salt and water
usually are controlled simultaneously.
intake. As long as kidney function is normal and the
There are circumstances, however, in which the dis-
pressure diuresis mechanism is operating effectively,
tribution of extracellular fluid between the interstitial
large changes in salt and water intake can be accom-
spaces and blood can vary greatly. As discussed in
modated with only slight changes in blood volume,
Chapter 25, the principal factors that can cause accu-
extracellular fluid volume, cardiac output, and arterial
mulation of fluid in the interstitial spaces include (1)
pressure.
increased capillary hydrostatic pressure, (2) decreased
The opposite sequence of events occurs when fluid
plasma colloid osmotic pressure, (3) increased perme-
intake falls below normal. In this case, there is a ten-
ability of the capillaries, and (4) obstruction of lym-
dency toward decreased blood volume and extracellu-
phatic vessels. In all these conditions, an unusually high
lar fluid volume, as well as reduced arterial pressure.
proportion of the extracellular fluid becomes distrib-
Even a small decrease in blood pressure causes a
uted to the interstitial spaces.
large decrease in urine output, thereby allowing fluid
Figure 29-14 shows the normal distribution of fluid
balance to be maintained with minimal changes in
between the interstitial spaces and the vascular system
blood pressure, blood volume, or extracellular fluid
and the distribution that occurs in edema states. When
volume. The effectiveness of this mechanism in pre-
small amounts of fluid accumulate in the blood as a
venting large changes in blood volume is demon-
result of either too much fluid intake or a decrease in
strated in Figure 29-13, which shows that changes in
renal output of fluid, about 20 to 30 per cent of it stays
blood volume are almost imperceptible despite large
in the blood and increases the blood volume. The
variations in daily intake of water and electrolytes,
remainder is distributed to the interstitial spaces.
except when intake becomes so low that it is not suf-
When the extracellular fluid volume rises more than
ficient to make up for fluid losses caused by evapora-
30 to 50 per cent above normal, almost all the addi-
tion or other inescapable losses.
tional fluid goes into the interstitial spaces and little
remains in the blood. This occurs because once the
interstitial fluid pressure rises from its normally nega-
Precision of Blood Volume and
tive value to become positive, the tissue interstitial
Extracellular Fluid Volume Regulation
spaces become compliant, and large amounts of fluid
then pour into the tissues without interstitial fluid
By studying Figure 29-12, one can see why the blood
pressure rising much more. In other words, the safety
volume remains almost exactly constant despite
factor against edema, owing to a rising interstitial
extreme changes in daily fluid intake. The reason for
fluid pressure that counteracts fluid accumulation in
this is the following: (1) a slight change in blood
the tissues, is lost once the tissues become highly
volume causes a marked change in cardiac output, (2)
compliant.
a slight change in cardiac output causes a large change
Thus, under normal conditions, the interstitial
in blood pressure, and (3) a slight change in blood
spaces act as an “overflow” reservoir for excess fluid,
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
377
Sympathetic Nervous System
Control of Renal Excretion: Arterial
8
Edema
Baroreceptor and Low-Pressure
7
Stretch Receptor Reflexes
6
5
Normal value
Because the kidneys receive extensive sympathetic
innervation, changes in sympathetic activity can alter
4
renal sodium and water excretion as well as regulation
3
Death
of extracellular fluid volume under some conditions.
2
For example, when blood volume is reduced by hem-
orrhage, the pressures in the pulmonary blood vessels
1
and other low-pressure regions of the thorax decrease,
0
causing reflex activation of the sympathetic nervous
0
5
10
15
20
25
30
35
40
system. This in turn increases renal sympathetic nerve
Extracellular fluid volume (liters)
activity, which has several effects to decrease sodium
and water excretion: (1) constriction of the renal arte-
rioles, with resultant decreased GFR; (2) increased
Figure 29-14
tubular reabsorption of salt and water; and (3) stimu-
lation of renin release and increased angiotensin II
Approximate relation between extracellular fluid volume and blood
and aldosterone formation, both of which further
volume, showing a nearly linear relation in the normal range but
increase tubular reabsorption. And if the reduction in
also showing the failure of blood volume to continue rising when
the extracellular fluid volume becomes excessive. When this
blood volume is great enough to lower systemic arte-
occurs, the additional extracellular fluid volume resides in the
rial pressure, further activation of the sympathetic
interstitial spaces, and edema results.
nervous system occurs because of decreased stretch of
the arterial baroreceptors located in the carotid sinus
and aortic arch. All these reflexes together play an
important role in the rapid restitution of blood volume
that occurs in acute conditions such as hemorrhage.
sometimes increasing in volume 10 to 30 liters. This
Also, reflex inhibition of renal sympathetic activity
causes edema, as explained in Chapter 25, but it also
may contribute to the rapid elimination of excess fluid
acts as an important overflow release valve for the cir-
in the circulation that occurs after eating a meal that
culation, protecting the cardiovascular system against
contains large amounts of salt and water.
dangerous overload that could lead to pulmonary
edema and cardiac failure.
To summarize, extracellular fluid volume and blood
Role of Angiotensin II In Controlling
volume are controlled simultaneously, but the quanti-
Renal Excretion
tative amounts of fluid distribution between the
interstitium and the blood depend on the physical
One of the body’s most powerful controllers of sodium
properties of the circulation and the interstitial spaces
excretion is angiotensin II. Changes in sodium and
as well as on the dynamics of fluid exchange through
fluid intake are associated with reciprocal changes in
the capillary membranes.
angiotensin II formation, and this in turn contributes
greatly to the maintenance of body sodium and fluid
Nervous and Hormonal
balances. That is, when sodium intake is elevated
above normal, renin secretion is decreased, causing
Factors Increase the
decreased angiotensin II formation. Because
Effectiveness of Renal-Body
angiotensin II has several important effects in increas-
Fluid Feedback Control
ing tubular reabsorption of sodium, as explained in
Chapter 27, a reduced level of angiotensin II decreases
In Chapter 27, we discuss the nervous and hormonal
tubular reabsorption of sodium and water, thus
factors that influence GFR and tubular reabsorption
increasing the kidneys’ excretion of sodium and water.
and, therefore, renal excretion of salt and water. These
The net result is to minimize the rise in extracellular
nervous and hormonal mechanisms usually act in
fluid volume and arterial pressure that would other-
concert with the pressure natriuresis and pressure
wise occur when sodium intake increases.
diuresis mechanisms, making them more effective in
Conversely, when sodium intake is reduced below
minimizing the changes in blood volume, extracellular
normal, increased levels of angiotensin II cause
fluid volume, and arterial pressure that occur in
sodium and water retention and oppose reductions in
response to day-to-day challenges. However, abnor-
arterial blood pressure that would otherwise occur.
malities of kidney function or of the various nervous
Thus, changes in activity of the renin-angiotensin
and hormonal factors that influence the kidneys can
system act as a powerful amplifier of the pressure
lead to serious changes in blood pressure and body
natriuresis mechanism for maintaining stable blood
fluid volumes, as discussed later.
pressures and body fluid volumes.
378
Unit V The Body Fluids and Kidneys
12
Angiotensin blockade
Normal
10
High angiotensin II
8
6
Figure 29-15
4
Effect of excessive angiotensin II
formation and effect of blocking
angiotensin II formation on the
2
renal-pressure natriuresis curve. Note
that high levels of angiotensin II for-
0
mation decrease the slope of pressure
60
80
100
120
140
160
natriuresis, making blood pressure
Arterial pressure (mm Hg)
very sensitive to changes in sodium
intake. Blockade of angiotensin II for-
mation shifts pressure natriuresis to
lower blood pressures.
Importance of Angiotensin II in Increasing Effectiveness of
lowering effects in hypertensive patients of the
Pressure Natriuresis. The importance of angiotensin II
angiotensin-converting enzyme inhibitors and
in making the pressure natriuresis mechanism more
angiotensin II receptor antagonists.
effective is shown in Figure 29-15. Note that when the
angiotensin control of natriuresis is fully functional,
Excessive Angiotensin II Does Not Cause Large Increases in
the pressure natriuresis curve is steep (normal curve),
Extracellular Fluid Volume Because Increased Arterial Pressure
indicating that only minor changes in blood pressure
Counterbalances Angiotensin-Mediated Sodium Retention.
are needed to increase sodium excretion when sodium
Although angiotensin II is one of the most powerful
intake is raised.
sodium- and water-retaining hormones in the body,
In contrast, when angiotensin levels cannot be
neither a decrease nor an increase in circulating
decreased in response to increased sodium intake
angiotensin II has a large effect on extracellular fluid
(high angiotensin II curve), as occurs in some hyper-
volume or blood volume. The reason for this is
tensive patients who have impaired ability to decrease
that with large increases in angiotensin II levels, as
renin secretion, the pressure natriuresis curve is not
occurs with a renin-secreting tumor of the kidney,
nearly as steep. Therefore, when sodium intake is
the high angiotensin II levels initially cause sodium
raised, much greater increases in arterial pressure are
and water retention by the kidneys and a small
needed to increase sodium excretion and maintain
increase in extracellular fluid volume. This also initi-
sodium balance. For example, in most people, a 10-fold
ates a rise in arterial pressure that quickly increases
increase in sodium intake causes an increase of only a
kidney output of sodium and water, thereby overcom-
few millimeters of mercury in arterial pressure,
ing the sodium- and water-retaining effects of the
whereas in subjects who cannot suppress angiotensin
angiotensin II and re-establishing a balance between
II formation appropriately in response to excess
intake and output of sodium at a higher blood pres-
sodium, the same rise in sodium intake causes blood
sure. Conversely, after blockade of angiotensin II for-
pressure to rise as much as 50 mm Hg. Thus, the inabil-
mation, as occurs when an angiotensin-converting
ity to suppress angiotensin II formation when there is
enzyme inhibitor is administered, there is initial loss of
excess sodium reduces the slope of pressure natriure-
sodium and water, but the fall in blood pressure offsets
sis and makes arterial pressure very salt sensitive, as
this effect, and sodium excretion is once again restored
discussed in Chapter 19.
to normal.
The use of drugs to block the effects of angiotensin
II has proved to be important clinically for improv-
ing the kidneys’ ability to excrete salt and water.
Role of Aldosterone in Controlling
When angiotensin II formation is blocked with an
Renal Excretion
angiotensin-converting enzyme inhibitor (see Figure
29-15) or an angiotensin II receptor antagonist, the
Aldosterone increases sodium reabsorption, especially
renal-pressure natriuresis curve is shifted to lower
in the cortical collecting tubules. The increased sodium
pressures; this indicates an enhanced ability of the
reabsorption is also associated with increased water
kidneys to excrete sodium because normal levels of
reabsorption and potassium secretion. Therefore, the
sodium excretion can now be maintained at reduced
net effect of aldosterone is to make the kidneys retain
arterial pressures. This shift of pressure natriuresis
sodium and water but to increase potassium excretion
provides the basis for the chronic blood pressure-
in the urine.
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
379
The function of aldosterone in regulating sodium
Conversely, when there is excess extracellular volume,
balance is closely related to that described for
decreased ADH levels reduce reabsorption of water by
angiotensin II. That is, with reduction in sodium intake,
the kidneys, thus helping to rid the body of the excess
the increased angiotensin II levels that occur stimulate
volume.
aldosterone secretion, which in turn contributes to the
reduction in urinary sodium excretion and, therefore,
Excess ADH Secretion Usually Causes Only Small Increases in
to the maintenance of sodium balance. Conversely,
Extracellular Fluid Volume but Large Decreases in Sodium Con-
with high sodium intake, suppression of aldosterone
centration. Although ADH is important in regulating
formation decreases tubular reabsorption, allowing
extracellular fluid volume, excessive levels of ADH
the kidneys to excrete larger amounts of sodium. Thus,
seldom cause large increases in arterial pressure or
changes in aldosterone formation also aid the pressure
extracellular fluid volume. Infusion of large amounts
natriuresis mechanism in maintaining sodium balance
of ADH into animals initially causes renal retention of
during variations in salt intake.
water and a 10 to 15 per cent increase in extracellular
fluid volume. As the arterial pressure rises in response
During Chronic Oversecretion of Aldosterone, Kidneys
to this increased volume, much of the excess volume
“Escape” from Sodium Retention as Arterial Pressure Rises.
is excreted because of the pressure diuresis mecha-
Although aldosterone has powerful effects on sodium
nism. After several days of ADH infusion, the blood
reabsorption, when there is excessive infusion of
volume and extracellular fluid volume are elevated no
aldosterone or excessive formation of aldosterone, as
more than 5 to 10 per cent, and the arterial pressure is
occurs in patients with tumors of the adrenal gland
also elevated by less than 10 mm Hg. The same is true
(Conn’s syndrome), the increased sodium reabsorp-
for patients with inappropriate ADH syndrome, in
tion and decreased sodium excretion by the kidneys
which ADH levels may be elevated severalfold.
are transient. After 1 to 3 days of sodium and water
Thus, high levels of ADH do not cause major
retention, the extracellular fluid volume rises by about
increases of either body fluid volume or arterial pres-
10 to 15 per cent and there is a simultaneous increase
sure, although high ADH levels can cause severe reduc-
in arterial blood pressure. When the arterial pressure
tions in extracellular sodium ion concentration. The
rises sufficiently, the kidneys “escape” from the sodium
reason for this is that increased water reabsorption by
and water retention and thereafter excrete amounts of
the kidneys dilutes the extracellular sodium, and at the
sodium equal to the daily intake, despite continued
same time, the small increase in blood pressure that
presence of high levels of aldosterone. The primary
does occur causes loss of sodium from the extracellu-
reason for the escape is the pressure natriuresis and
lar fluid in the urine through pressure natriuresis.
diuresis that occur when the arterial pressure rises.
In patients who have lost their ability to secrete
In patients with adrenal insufficiency who do not
ADH because of destruction of the supraoptic nuclei,
secrete enough aldosterone (Addison’s disease), there
the urine volume may become 5 to 10 times normal.
is increased excretion of sodium and water, reduction
This is almost always compensated for by ingestion of
in extracellular fluid volume, and a tendency toward
enough water to maintain fluid balance. If free access
low blood pressure. In the complete absence of aldos-
to water is prevented, the inability to secrete ADH
terone, the volume depletion may be severe unless the
may lead to marked reductions in blood volume and
person is allowed to eat large amounts of salt and
arterial pressure.
drink large amounts of water to balance the increased
urine output of salt and water.
Role of Atrial Natriuretic Peptide
in Controlling Renal Excretion
Role of ADH in Controlling Renal
Water Excretion
Thus far, we have discussed mainly the role of sodium-
and water-retaining hormones in controlling extracel-
As discussed in Chapter 28, ADH plays an important
lular fluid volume. However, several different natri-
role in allowing the kidneys to form a small volume of
uretic hormones may also contribute to volume
concentrated urine while excreting normal amounts of
regulation. One of the most important of the natri-
salt. This effect is especially important during water
uretic hormones is a peptide referred to as atrial natri-
deprivation, which strongly elevates plasma levels of
uretic peptide (ANP), released by the cardiac atrial
ADH that in turn increase water reabsorption by the
muscle fibers. The stimulus for release of this peptide
kidneys and help to minimize the decreases in extra-
appears to be overstretch of the atria, which can result
cellular fluid volume and arterial pressure that would
from excess blood volume. Once released by the
otherwise occur. Water deprivation for 24 to 48 hours
cardiac atria, ANP enters the circulation and acts on
normally causes only a small decrease in extracellular
the kidneys to cause small increases in GFR and
fluid volume and arterial pressure. However, if the
decreases in sodium reabsorption by the collecting
effects of ADH are blocked with a drug that antago-
ducts. These combined actions of ANP lead to
nizes the action of ADH to promote water reabsorp-
increased excretion of salt and water, which helps to
tion in the distal and collecting tubules, the same
compensate for the excess blood volume.
period of water deprivation causes a substantial fall in
Changes in ANP levels probably help to minimize
both extracellular fluid volume and arterial pressure.
changes in blood volume during various disturbances,
380
Unit V The Body Fluids and Kidneys
such as increased salt and water intake. However,
systems leads to an increase in sodium excretion when
excessive production of ANP or even complete lack of
sodium intake is increased. The opposite changes take
ANP does not cause major changes in blood volume
place when sodium intake is reduced below normal
because these effects can easily be overcome by small
levels.
changes in blood pressure, acting through pressure
natriuresis. For example, infusions of large amounts of
ANP initially raise urine output of salt and water and
Conditions That Cause
cause slight decreases in blood volume. In less than 24
Large Increases in Blood
hours, this effect is overcome by a slight decrease in
Volume and Extracellular
blood pressure that returns urine output toward
Fluid Volume
normal, despite continued excess of ANP.
Despite the powerful regulatory mechanisms that main-
tain blood volume and extracellular fluid volume rea-
sonably constant, there are abnormal conditions that
Integrated Responses to
can cause large increases in both of these variables.
Changes in Sodium Intake
Almost all of these conditions result from circulatory
abnormalities.
The integration of the different control systems that
regulate sodium and fluid excretion under normal
Increased Blood Volume and
conditions can be summarized by examining the
Extracellular Fluid Volume Caused
homeostatic responses to progressive increases in
by Heart Diseases
dietary sodium intake. As discussed previously, the
kidneys have an amazing capability to match their
In congestive heart failure, blood volume may increase
excretion of salt and water to intakes that can range
15 to 20 per cent, and extracellular fluid volume some-
from as low as one tenth of normal to as high as 10
times increases by 200 per cent or more. The reason for
times normal.
this can be understood by re-examination of Figure
29-12. Initially, heart failure reduces cardiac output and,
consequently, decreases arterial pressure. This in turn
High Sodium Intake Suppresses Antinatriuretic Systems and
activates multiple sodium-retaining systems, especially
Activates Natriuretic Systems. As sodium intake is
the renin-angiotensin, aldosterone, and sympathetic
increased, sodium output initially lags slightly behind
nervous systems. In addition, the low blood pressure
intake. The time delay results in a small increase in the
itself causes the kidneys to retain salt and water. There-
cumulative sodium balance, which causes a slight
fore, the kidneys retain volume in an attempt to return
increase in extracellular fluid volume. It is mainly this
the arterial pressure and cardiac output toward normal.
small increase in extracellular fluid volume that trig-
Indeed, if the heart failure is not too severe, the rise in
gers various mechanisms in the body to increase
blood volume can often return cardiac output and arte-
rial pressure virtually all the way to normal, and sodium
sodium excretion. These mechanisms include the
excretion will eventually increase back to normal,
following:
although there will remain excess extracellular fluid
1. Activation of low pressure receptor reflexes that
volume and blood volume to keep the weakened heart
originate from the stretch receptors of the right
pumping adequately. However, if the heart is greatly
atrium and the pulmonary blood vessels. Signals
weakened, arterial pressure will not be able to increase
from the stretch receptors go to the brain stem
enough to restore urine output to normal. When this
and there inhibit sympathetic nerve activity to the
occurs, the kidneys continue to retain volume until the
kidneys to decrease tubular sodium reabsorption.
person develops severe circulatory congestion and
This mechanism is most important in the first few
eventually dies of pulmonary edema.
hours—or perhaps the first day—after a large
In myocardial failure, heart valvular disease, and
congenital abnormalities of the heart, an important
increase in salt and water intake.
circulatory compensation is an increase in blood
2. Small increases in arterial pressure, caused by
volume, which helps to return cardiac output and blood
volume expansion, raise sodium excretion through
pressure to normal. This allows even the weakened
pressure natriuresis.
heart to maintain a life-sustaining level of cardiac
3. Suppression of angiotensin II formation, caused by
output.
increased arterial pressure and extracellular fluid
volume expansion, decreases tubular sodium
reabsorption by eliminating the normal effect of
Increased Blood Volume Caused by
angiotensin II to increase sodium reabsorption.
Increased Capacity of Circulation
Also, reduced angiotensin II decreases
Any condition that increases vascular capacity will also
aldosterone secretion, thus further reducing
cause the blood volume to increase to fill this extra
tubular sodium reabsorption.
capacity. An increase in vascular capacity initially
4. Stimulation of natriuretic systems, especially ANP,
reduces mean circulatory filling pressure (see Figure
contributes further to increased sodium excretion.
29-12), which leads to decreased cardiac output and
Thus, the combined activation of natriuretic systems
decreased arterial pressure. The fall in pressure causes
and suppression of sodium- and water-retaining
salt and water retention by the kidneys until the blood
Chapter 29
Renal Regulation; Integration of Renal Mechanisms
381
volume increases sufficiently to fill the extra capacity.
into the tissues of the body. The net result is massive
For example, in pregnancy the increased vascular capac-
fluid retention by the kidneys until tremendous extra-
ity of the uterus, placenta, and other enlarged organs
cellular edema occurs unless treatment is instituted to
of the woman’s body regularly increases the blood
restore the plasma proteins.
volume 15 to 25 per cent. Similarly, in patients who
have large varicose veins of the legs, which in rare
instances may hold up to an extra liter of blood,
Liver Cirrhosis—Decreased
the blood volume simply increases to fill the extra
Synthesis of Plasma Proteins
vascular capacity. In these cases, salt and water are
by the Liver and Sodium Retention
retained by the kidneys until the total vascular bed is
filled enough to raise blood pressure to the level
by the Kidneys
required to balance renal output of fluid with daily
A similar sequence of events occurs in cirrhosis of the
intake of fluid.
liver as in nephrotic syndrome, except that in liver cir-
rhosis, the reduction in plasma protein concentration
results from destruction of the liver cells, thus reducing
the ability of the liver to synthesize enough plasma
Conditions That Cause
proteins. Cirrhosis is also associated with large amounts
Large Increases in
of fibrous tissue in the liver structure, which greatly
Extracellular Fluid
impedes the flow of portal blood through the liver.
This in turn raises capillary pressure throughout the
Volume but with Normal
portal vascular bed, which also contributes to the
Blood Volume
leakage of fluid and proteins into the peritoneal cavity,
a condition called ascites. Once fluid and protein are lost
There are several conditions in which extracellular fluid
from the circulation, the renal responses are similar to
volume becomes markedly increased but blood volume
those observed in other conditions associated with
remains normal or even slightly reduced. These condi-
decreased plasma volume. That is, the kidneys continue
tions are usually initiated by leakage of fluid and protein
to retain salt and water until plasma volume and arte-
into the interstitium, which tends to decrease the blood
rial pressure are restored to normal. In some cases,
volume. The kidneys’ response to these conditions is
plasma volume may actually increase above normal
similar to the response after hemorrhage. That is, the
because of increased vascular capacity in cirrhosis;
kidneys retain salt and water in an attempt to restore
the high pressures in the portal circulation can
blood volume toward normal. Much of the extra fluid,
greatly distend veins and therefore increase vascular
however, leaks into the interstitium, causing further
capacity.
edema.
References
Nephrotic Syndrome—Loss of
Plasma Proteins in Urine and
Antunes-Rodrigues J, de Castro M, Elias LL, et al: Neu-
Sodium Retention by the Kidneys
roendocrine control of body fluid metabolism. Physiol Rev
84:169, 2004.
The general mechanisms that lead to extracellular
Cowley AW Jr: Long-term control of arterial pressure.
edema are reviewed in Chapter 25. One of the most
Physiol Rev 72:231, 1992.
important clinical causes of edema is the so-called
Fitzsimmons JT: Physiology and pathophysiology of thirst
nephrotic syndrome. In nephrotic syndrome, the
and salt appetite. In Seldin DW, Giebisch G (eds): The
glomerular capillaries leak large amounts of protein
Kidney—Physiology and Pathophysiology, 3rd ed. New
into the filtrate and the urine because of an increased
York: Raven Press, 2000, pp 1153-1174.
permeability of the glomerulus. Thirty to 50 grams of
Giebisch GH: A trail of research on potassium. Kidney Int
plasma protein can be lost in the urine each day, some-
62:1498, 2002.
times causing the plasma protein concentration to fall
Giebisch G, Hebert SC, Wang WH: New aspects of renal
to less than one-third normal. As a consequence of the
potassium transport. Pflugers Arch 446:289, 2003.
decreased plasma protein concentration, the plasma
Granger JP, Alexander BT, Llinas M: Mechanisms of pres-
colloid osmotic pressure falls to low levels. This causes
sure natriuresis. Curr Hypertens Rep 4:15, 2002.
the capillaries all over the body to filter large amounts
Guise TA, Mundy GR: Disorders of calcium metabolism. In
of fluid into the various tissues, which in turn causes
Seldin DW, Giebisch G (eds): The Kidney—Physiology
edema and decreases the plasma volume.
and Pathophysiology, 3rd ed. New York: Raven Press,
Renal sodium retention in nephrotic syndrome
2000, pp 1811-1840.
occurs through multiple mechanisms activated by
Guyton AC: Blood pressure control—special role of the
leakage of protein and fluid from the plasma into the
kidneys and body fluids. Science 252:1813, 1991.
interstitial fluid, including activation of various sodium-
Hall JE, Brands MW: The renin-angiotensin-aldosterone
retaining systems such as the renin-angiotensin system,
system: renal mechanisms and circulatory homeostasis. In
aldosterone, and possibly the sympathetic nervous
Seldin DW, Giebisch G (eds): The Kidney—Physiology
system. The kidneys continue to retain sodium and
and Pathophysiology, 3rd ed. New York: Raven Press,
water until plasma volume is restored nearly to normal.
2000, pp 1009-1046.
However, because of the large amount of sodium and
Hall JE: Angiotensin II and long-term arterial pressure reg-
water retention, the plasma protein concentration
ulation: the overriding dominance of the kidney. J Am Soc
becomes further diluted, causing still more fluid to leak
Nephrol 10(Suppl 12):s258, 1999.
382
Unit V The Body Fluids and Kidneys
Murer H, Hernando N, Forster I, Biber J: Regulation of
Kidney, 6th ed. Philadelphia: WB Saunders, 2000, pp 520-
Na/Pi transporter in the proximal tubule. Annu Rev
574.
Physiol 65:531, 2003.
Warnock DG. Renal genetic disorders related to K+ and
Rosa R, Epstein FH: Extrarenal potassium metabolism. In
Mg2+. Annu Rev Physiol 64:845, 2002.
Seldin DW, Giebisch G (eds): The Kidney—Physiology
Young DB: Quantitative analysis of aldosterone’s role in
and Pathophysiology, 3rd ed. New York: Raven Press,
potassium regulation. Am J Physiol 255: F811, 1988.
2000, pp 1551-1574.
Young DB: Analysis of long-term potassium regulation.
Suki WN, Lederer ED, Rouse D: Renal transport of calcium
Endocr Rev 6:24, 1985.
magnesium and phosphate. In: Brenner BM (ed): The
C H A P T E R
3
0
Regulation of Acid-Base Balance
Regulation of hydrogen ion (H+) balance is similar
in some ways to the regulation of other ions in the
body. For instance, to achieve homeostasis, there
must be a balance between the intake or production
of H+ and the net removal of H+ from the body. And,
as is true for other ions, the kidneys play a key role
in regulating H+ removal. However, precise control
of extracellular fluid H+ concentration involves
much more than simple elimination of H+ by the kidneys. There are also multi-
ple acid-base buffering mechanisms involving the blood, cells, and lungs that are
essential in maintaining normal H+ concentrations in both the extracellular and
the intracellular fluid.
In this chapter, the various mechanisms that contribute to the regulation of
H+ concentration are discussed, with special emphasis on the control of renal
H+ secretion and renal reabsorption, production, and excretion of bicarbonate
ions (HCO3-), one of the key components of acid-base control systems in the
body fluids.
Hydrogen Ion Concentration Is
Precisely Regulated
Precise H+ regulation is essential because the activities of almost all enzyme
systems in the body are influenced by H+ concentration. Therefore, changes in
hydrogen concentration alter virtually all cell and body functions.
Compared with other ions, the H+ concentration of the body fluids normally
is kept at a low level. For example, the concentration of sodium in extracellu-
lar fluid (142 mEq/L) is about 3.5 million times as great as the normal concen-
tration of H+, which averages only 0.00004 mEq/L. Equally important, the
normal variation in H+ concentration in extracellular fluid is only about one mil-
lionth as great as the normal variation in sodium ion (Na+) concentration. Thus,
the precision with which H+ is regulated emphasizes its importance to the
various cell functions.
Acids and Bases—Their Definitions
and Meanings
A hydrogen ion is a single free proton released from a hydrogen atom. Molecules
containing hydrogen atoms that can release hydrogen ions in solutions are referred
to as acids. An example is hydrochloric acid (HCl), which ionizes in water to form
hydrogen ions (H+) and chloride ions (Cl-). Likewise, carbonic acid (H2CO3) ionizes
in water to form H+ and bicarbonate ions (HCO3-).
A base is an ion or a molecule that can accept an H+. For example, HCO3- is a
base because it can combine with H+ to form H2CO3. Likewise, HPO4= is a base
because it can accept an H+ to form H2PO4-. The proteins in the body also function
as bases, because some of the amino acids that make up proteins have net negative
charges that readily accept H+. The protein hemoglobin in the red blood cells and
proteins in the other cells of the body are among the most important of the body’s
bases.
The terms base and alkali are often used synonymously. An alkali is a molecule
formed by the combination of one or more of the alkaline metals—sodium, potas-
sium, lithium, and so forth—with a highly basic ion such as a hydroxyl ion (OH-).
383
384
Unit V The Body Fluids and Kidneys
The base portion of these molecules reacts quickly with
Table 30-1
H+ to remove it from solution; they are, therefore,
typical bases. For similar reasons, the term alkalosis
pH and H+ Concentration of Body Fluids
refers to excess removal of H+ from the body fluids, in
contrast to the excess addition of H+, which is referred
H+ Concentration (mEq/L)
pH
to as acidosis.
Extracellular fluid
Strong and Weak Acids and Bases. A strong acid is one that
Arterial blood
4.0
¥ 10-5
7.40
Venous blood
4.5
¥ 10-5
7.35
rapidly dissociates and releases especially large
Interstitial fluid
4.5
¥ 10-5
7.35
amounts of H+ in solution. An example is HCl. Weak
acids have less tendency to dissociate their ions and,
Intracellular fluid
1
¥ 10-3 to 4
¥ 10-5
6.0 to 7.4
therefore, release H+ with less vigor. An example is
Urine
3
¥ 10-2 to 1
¥ 10-5
4.5 to 8.0
H2CO3. A strong base is one that reacts rapidly and
Gastric HCl
160
0.8
strongly with H+ and, therefore, quickly removes these
from a solution. A typical example is OH-, which reacts
with H+ to form water (H2O). A typical weak base is
HCO3- because it binds with H+ much more weakly than
does OH-. Most of the acids and bases in the extracel-
The pH of urine can range from 4.5 to 8.0, depending
lular fluid that are involved in normal acid-base regula-
on the acid-base status of the extracellular fluid. As dis-
tion are weak acids and bases. The most important ones
cussed later, the kidneys play a major role in correcting
that we discuss in detail are H2CO3 and bicarbonate
abnormalities of extracellular fluid H+ concentration by
base.
excreting acids or bases at variable rates.
An extreme example of an acidic body fluid is the
Normal Hydrogen Ion Concentration and pH of Body Fluids
HCl secreted into the stomach by the oxyntic (parietal)
and Changes That Occur in Acidosis and Alkalosis. As dis-
cells of the stomach mucosa, as discussed in Chapter 64.
cussed earlier, the blood H+ concentration is normally
The H+ concentration in these cells is about 4 million
maintained within tight limits around a normal value
times greater than the hydrogen concentration in blood,
of about 0.00004 mEq/L (40 nEq/L). Normal variations
with a pH of 0.8.
are only about
3 to
5 nEq/L, but under extreme
In the remainder of this chapter, we discuss the reg-
conditions, the H+ concentration can vary from as low
ulation of extracellular fluid H+ concentration.
as 10 nEq/L to as high as 160 nEq/L without causing
death.
Because H+ concentration normally is low, and
Defenses Against Changes in
because these small numbers are cumbersome, it is cus-
tomary to express H+ concentration on a logarithm
Hydrogen Ion Concentration:
scale, using pH units. pH is related to the actual H+ con-
Buffers, Lungs, and Kidneys
centration by the following formula (H+ concentration
[H+] is expressed in equivalents per liter):
There are three primary systems that regulate the H+
1
concentration in the body fluids to prevent acidosis or
pH=log
[
]
alkalosis: (1) the chemical acid-base buffer systems of
[
H+
]=-lo
the body fluids, which immediately combine with acid
For example, the normal
[H+] is
40 nEq/L
or base to prevent excessive changes in H+ concentra-
(0.00000004 Eq/L). Therefore, the normal pH is
tion; (2) the respiratory center, which regulates the
removal of CO2 (and, therefore, H2CO3) from the
pH = -log [0.00000004]
extracellular fluid; and
(3) the kidneys, which can
pH = 7.4
excrete either acid or alkaline urine, thereby readjust-
From this formula, one can see that pH is inversely
ing the extracellular fluid H+ concentration toward
related to the H+ concentration; therefore, a low pH
normal during acidosis or alkalosis.
corresponds to a high H+ concentration, and a high pH
When there is a change in H+ concentration, the
corresponds to a low H+ concentration.
buffer systems of the body fluids react within a fraction
The normal pH of arterial blood is 7.4, whereas the
of a second to minimize these changes. Buffer systems
pH of venous blood and interstitial fluids is about 7.35
do not eliminate H+ from or add them to the body
because of the extra amounts of carbon dioxide (CO2)
released from the tissues to form H2CO3 in these fluids
but only keep them tied up until balance can be re-
(Table 30-1). Because the normal pH of arterial blood
established.
is 7.4, a person is considered to have acidosis when the
The second line of defense, the respiratory system,
pH falls below this value and to have alkalosis when the
also acts within a few minutes to eliminate CO2 and,
pH rises above 7.4. The lower limit of pH at which a
therefore, H2CO3 from the body.
person can live more than a few hours is about 6.8, and
These first two lines of defense keep the H+ con-
the upper limit is about 8.0.
centration from changing too much until the more
Intracellular pH usually is slightly lower than plasma
slowly responding third line of defense, the kidneys,
pH because the metabolism of the cells produces acid,
can eliminate the excess acid or base from the body.
especially H2CO3. Depending on the type of cells, the
Although the kidneys are relatively slow to respond
pH of intracellular fluid has been estimated to range
between 6.0 and 7.4. Hypoxia of the tissues and poor
compared with the other defenses, over a period of
blood flow to the tissues can cause acid accumulation
hours to several days, they are by far the most power-
and decreased intracellular pH.
ful of the acid-base regulatory systems.
Chapter 30
Regulation of Acid-Base Balance
385
Buffering of Hydrogen Ions
Now, putting the entire system together, we have the
following:
in the Body Fluids
ææ
Æ
-
2
CO +H O
2
¨
2
H CO
3
¨
H++HCO
3
A buffer is any substance that can reversibly bind H+.
+
+
The general form of the buffering reaction is
Na
+
æ
æ
Because of the weak dissociation of H2CO3, the H+
Buffer+H
¨
H Buffer
concentration is extremely small.
In this example, a free H+ combines with the buffer to
When a strong acid such as HCl is added to the
form a weak acid (H buffer) that can either remain as
bicarbonate buffer solution, the increased H+ released
an unassociated molecule or dissociate back to buffer
from the acid (HCl Æ H+ + Cl-) is buffered by HCO3-.
and H+. When the H+ concentration increases, the
reaction is forced to the right, and more H+ binds to
≠H+ + HCO3- Æ H2CO3 Æ CO2 + H2O
the buffer, as long as buffer is available. Conversely,
As a result, more H2CO3 is formed, causing increased
when the H+ concentration decreases, the reaction
CO2 and H2O production. From these reactions, one
shifts toward the left, and H+ is released from the
can see that H+ from the strong acid HCl reacts with
buffer. In this way, changes in H+ concentration are
HCO3- to form the very weak acid H2CO3, which in
minimized.
turn forms CO2 and H2O. The excess CO2 greatly stim-
The importance of the body fluid buffers can be
ulates respiration, which eliminates the CO2 from the
quickly realized if one considers the low concentration
extracellular fluid.
of H+ in the body fluids and the relatively large
The opposite reactions take place when a strong
amounts of acids produced by the body each day. For
base, such as sodium hydroxide (NaOH), is added to
example, about 80 milliequivalents of hydrogen is
the bicarbonate buffer solution.
either ingested or produced each day by metabolism,
whereas the H+ concentration of the body fluids nor-
NaOH + H2CO3 Æ NaHCO3 + H2O
mally is only about 0.00004 mEq/L. Without buffering,
In this case, the OH- from the NaOH combines with
the daily production and ingestion of acids would
H2CO3 to form additional HCO3-.Thus, the weak base
cause huge changes in body fluid H+ concentration.
NaHCO3 replaces the strong base NaOH. At the same
The action of acid-base buffers can perhaps best be
time, the concentration of H2CO3 decreases (because
explained by considering the buffer system that is
it reacts with NaOH), causing more CO2 to combine
quantitatively the most important in the extracellular
with H2O to replace the H2CO3.
fluid—the bicarbonate buffer system.
+
2
CO +H O
2
æ
æ
2
H CO
3
æ
æ
≠HCO +H
3
+
+
Bicarbonate Buffer System
NaOH
Na
The net result, therefore, is a tendency for the CO2
The bicarbonate buffer system consists of a water solu-
levels in the blood to decrease, but the decreased CO2
tion that contains two ingredients: (1) a weak acid,
in the blood inhibits respiration and decreases the rate
H2CO3, and (2) a bicarbonate salt, such as NaHCO3.
of CO2 expiration. The rise in blood HCO3- that occurs
H2CO3 is formed in the body by the reaction of CO2
is compensated for by increased renal excretion of
with H2O.
HCO3-.
carbonic
anhydrase
CO +H O
H CO
2
2
2
3
¨
ææ
ææ
Quantitative Dynamics of the
This reaction is slow, and exceedingly small amounts
Bicarbonate Buffer System
of H2CO3 are formed unless the enzyme carbonic
All acids, including H2CO3, are ionized to some extent.
anhydrase is present. This enzyme is especially abun-
From mass balance considerations, the concentrations
dant in the walls of the lung alveoli, where CO2 is
of H+ and HCO3- are proportional to the concentration
released; carbonic anhydrase is also present in the
of H2CO3.
epithelial cells of the renal tubules, where CO2 reacts
Æ
+
-
with H2O to form H2CO3.
H2CO
3
¨
H
+
HCO
3
H2CO3 ionizes weakly to form small amounts of H+
For any acid, the concentration of the acid relative
and HCO3-.
to its dissociated ions is defined by the dissociation
constant K¢.
Æ
-
H C2
O
3
¨
H++HCO
3
+
-
H
¥
HCO
3
The second component of the system, bicarbonate
K¢=
(1)
H2CO
3
salt, occurs predominantly as sodium bicarbonate
(NaHCO3) in the extracellular fluid. NaHCO3
This equation indicates that in an H2CO3 solution, the
ionizes almost completely to form HCO3- and Na+, as
amount of free H+ is equal to
follows:
H2CO
3
H+=K¢¥
(2)
æ
æ
æ
Æ
-
-
NaHCO
3
¨
Na++HCO
3
HCO
3
386
Unit V The Body Fluids and Kidneys
The concentration of undissociated H2CO3 cannot be
toward alkalosis. An increase in PCO2 causes the pH to
measured in solution because it rapidly dissociates into
decrease, shifting the acid-base balance toward acidosis.
CO2 and H2O or to H+ and HCO3-. However, the CO2
The Henderson-Hasselbalch equation, in addition to
dissolved in the blood is directly proportional to the
defining the determinants of normal pH regulation and
amount of undissociated H2CO3. Therefore, equation 2
acid-base balance in the extracellular fluid, provides
can be rewritten as
insight into the physiologic control of acid and base
composition of the extracellular fluid. As discussed
CO
2
H+=K
¥
(3)
later, the bicarbonate concentration is regulated mainly
-
HCO3
by the kidneys, whereas the PCO2 in extracellular fluid is
controlled by the rate of respiration. By increasing the
The dissociation constant (K) for equation 3 is only
rate of respiration, the lungs remove CO2 from the
about 1/400 of the dissociation constant (K¢) of equation
plasma, and by decreasing respiration, the lungs elevate
2 because the proportionality ratio between H2CO3 and
PCO2. Normal physiologic acid-base homeostasis results
CO2 is 1:400.
from the coordinated efforts of both of these organs, the
Equation 3 is written in terms of the total amount of
lungs and the kidneys, and acid-base disorders occur
CO2 dissolved in solution. However, most clinical labo-
when one or both of these control mechanisms are
ratories measure the blood CO2 tension (PCO2) rather
impaired, thus altering either the bicarbonate concen-
than the actual amount of CO2. Fortunately, the amount
tration or the PCO2 of extracellular fluid.
of CO2 in the blood is a linear function of PCO2 times
When disturbances of acid-base balance result from
the solubility coefficient for CO2; under physiologic
a primary change in extracellular fluid bicarbonate con-
conditions, the solubility coefficient for CO2
is
centration, they are referred to as metabolic acid-base
0.03 mmol/mm Hg at body temperature. This means that
disorders. Therefore, acidosis caused by a primary
0.03 millimole of H2CO3 is present in the blood for each
decrease in bicarbonate concentration is termed meta-
millimeter of mercury PCO2 measured. Therefore, equa-
bolic acidosis, whereas alkalosis caused by a primary
tion 3 can be rewritten as
increase in bicarbonate concentration is called meta-
(0.03
¥
Pco
2
)
bolic alkalosis. Acidosis caused by an increase in PCO2
H+=K
¥
(4)
-
is called respiratory acidosis, whereas alkalosis caused
HCO
3
by a decrease in PCO2 is termed respiratory alkalosis.
Henderson-Hasselbalch Equation. As discussed earlier, it is
Bicarbonate Buffer System Titration Curve. Figure 30-1 shows
customary to express H+ concentration in pH units
the changes in pH of the extracellular fluid when the
rather than in actual concentrations. Recall that pH is
ratio of HCO3- to CO2 in extracellular fluid is altered.
defined as pH = -log H+.
When the concentrations of these two components are
The dissociation constant can be expressed in a
equal, the right-hand portion of equation 8 becomes the
similar manner.
log of 1, which is equal to 0. Therefore, when the two
components of the buffer system are equal, the pH of
pK = -log K
the solution is the same as the pK (6.1) of the bicar-
Therefore, we can express the H+ concentration in
bonate buffer system. When base is added to the system,
equation 4 in pH units by taking the negative logarithm
part of the dissolved CO2 is converted into HCO3-,
of that equation, which yields
causing an increase in the ratio of HCO3- to CO2 and
increasing the pH, as is evident from the Henderson-
(0 03
¥
Pco
)
+
2
Hasselbalch equation. When acid is added, it is buffered
-log H
=-log
pK
-log
(5)
-
HCO3
Therefore,
(0 03
¥
Pco
2
)
pH
=
pK
-log
(6)
-
HCO3
0
100
Normal
Rather than work with a negative logarithm, we can
operating
change the sign of the logarithm and invert the numer-
point in body
25
75
ator and denominator in the last term, using the law of
logarithms to yield
-
HCO
3
50
50
pH
=
pK+
log
(7)
pK
(0 03
¥
Pco
2
)
For the bicarbonate buffer system, the pK is 6.1, and
75
25
equation 7 can be written as
-
HCO
3
pH
=
6
1+
log
(8)
100
0
0 03
¥
Pco
2
4
5
6
7
8
pH
Equation 8 is the Henderson-Hasselbalch equation,
and with it, one can calculate the pH of a solution if
the molar concentration of HCO3- and the PCO2 are
Figure 30-1
known.
From the Henderson-Hasselbalch equation, it is
Titration curve for bicarbonate buffer system showing the pH of
apparent that an increase in HCO3- concentration
extracellular fluid when the percentages of buffer in the form of
causes the pH to rise, shifting the acid-base balance
HCO3- and CO2 (or H2CO3) are altered.
Chapter 30
Regulation of Acid-Base Balance
387
by HCO3-, which is then converted into dissolved CO2,
HCl + Na2HPO4 Æ NaH2PO4 + NaCl
decreasing the ratio of HCO3- to CO2 and decreasing
The result of this reaction is that the strong acid, HCl,
the pH of the extracellular fluid.
is replaced by an additional amount of a weak acid,
“Buffer Power” Is Determined by the Amount and Relative Con-
NaH2PO4, and the decrease in pH is minimized.
centrations of the Buffer Components. From the titration
When a strong base, such as NaOH, is added to the
curve in Figure 30-1, several points are apparent. First,
buffer system, the OH- is buffered by the H2PO4- to
the pH of the system is the same as the pK when each
form additional amounts of HPO4= + H2O.
of the components (HCO3- and CO2) constitutes 50 per
cent of the total concentration of the buffer system.
NaOH + NaH2PO4 Æ Na2HPO4 + H2O
Second, the buffer system is most effective in the central
In this case, a strong base, NaOH, is traded for a weak
part of the curve, where the pH is near the pK of the
base, NaH2PO4, causing only a slight increase in
system. This means that the change in pH for any given
pH.
amount of acid or base added to the system is least when
The phosphate buffer system has a pK of 6.8, which
the pH is near the pK of the system. The buffer system
is still reasonably effective for 1.0 pH unit on either side
is not far from the normal pH of 7.4 in the body fluids;
of the pK, which for the bicarbonate buffer system
this allows the system to operate near its maximum
extends from a pH of about 5.1 to 7.1 units. Beyond
buffering power. However, its concentration in the
these limits, the buffering power rapidly diminishes.
extracellular fluid is low, only about 8 per cent of the
-
And when all the CO2 has been converted into HCO3
concentration of the bicarbonate buffer. Therefore,
or when all the HCO3- has been converted into CO2, the
the total buffering power of the phosphate system in
system has no more buffering power.
the extracellular fluid is much less than that of the
The absolute concentration of the buffers is also an
bicarbonate buffering system.
important factor in determining the buffer power of a
In contrast to its rather insignificant role as an extra-
system. With low concentrations of the buffers, only a
cellular buffer, the phosphate buffer is especially
small amount of acid or base added to the solution
changes the pH considerably.
important in the tubular fluids of the kidneys, for two
reasons: (1) phosphate usually becomes greatly con-
Bicarbonate Buffer System Is the Most Important Extracellular
centrated in the tubules, thereby increasing the buffer-
Buffer. From the titration curve shown in Figure 30-1,
ing power of the phosphate system, and (2) the tubular
one would not expect the bicarbonate buffer system to
fluid usually has a considerably lower pH than
be powerful, for two reasons: First, the pH of the extra-
the extracellular fluid does, bringing the operating
cellular fluid is about 7.4, whereas the pK of the bicar-
range of the buffer closer to the pK (6.8) of the
bonate buffer system is 6.1. This means that there is
system.
about 20 times as much of the bicarbonate buffer
The phosphate buffer system is also important in
system in the form of HCO3- as in the form of dis-
buffering intracellular fluid because the concentration
solved CO2. For this reason, this system operates on
of phosphate in this fluid is many times that in the
the portion of the buffering curve where the slope is
extracellular fluid. Also, the pH of intracellular fluid is
low and the buffering power is poor. Second, the con-
lower than that of extracellular fluid and therefore is
centrations of the two elements of the bicarbonate
usually closer to the pK of the phosphate buffer
system, CO2 and HCO3-, are not great.
system compared with the extracellular fluid.
Despite these characteristics, the bicarbonate buffer
system is the most powerful extracellular buffer in the
body. This apparent paradox is due mainly to the fact
Proteins: Important
that the two elements of the buffer system, HCO3- and
Intracellular Buffers
CO2, are regulated, respectively, by the kidneys and the
lungs, as discussed later. As a result of this regulation,
Proteins are among the most plentiful buffers in the
the pH of the extracellular fluid can be precisely con-
body because of their high concentrations, especially
trolled by the relative rate of removal and addition of
within the cells.
HCO3- by the kidneys and the rate of removal of CO2
The pH of the cells, although slightly lower than in
by the lungs.
the extracellular fluid, nevertheless changes approxi-
mately in proportion to extracellular fluid pH changes.
-
There is a slight amount of diffusion of H+
and HCO3
Phosphate Buffer System
through the cell membrane, although these ions
require several hours to come to equilibrium with the
Although the phosphate buffer system is not impor-
extracellular fluid, except for rapid equilibrium that
tant as an extracellular fluid buffer, it plays a major
occurs in the red blood cells. CO2, however, can rapidly
role in buffering renal tubular fluid and intracellular
diffuse through all the cell membranes. This diffusion
fluids.
of the elements of the bicarbonate buffer system causes
The main elements of the phosphate buffer system
the pH in intracellular fluid to change when there are
are H2PO4- and HPO4=. When a strong acid such as
changes in extracellular pH. For this reason, the buffer
HCl is added to a mixture of these two substances, the
systems within the cells help prevent changes in the
hydrogen is accepted by the base HPO4= and con-
pH of extracellular fluid but may take several hours to
verted to H2PO4-.
become maximally effective.
388
Unit V The Body Fluids and Kidneys
In the red blood cell, hemoglobin (Hb) is an impor-
flowing blood transports it to the lungs, where it dif-
tant buffer, as follows:
fuses into the alveoli and then is transferred to the
atmosphere by pulmonary ventilation. About 1.2 mol/
æ
æ
H+ + Hb
¨
HHb
L of dissolved CO2 normally is in the extracellular
Approximately 60 to 70 per cent of the total chemi-
fluid, corresponding to a Pco2 of 40 mm Hg.
cal buffering of the body fluids is inside the cells, and
If the rate of metabolic formation of CO2 increases,
most of this results from the intracellular proteins.
the Pco2 of the extracellular fluid is likewise increased.
However, except for the red blood cells, the slowness
Conversely, a decreased metabolic rate lowers the
with which H+ and HCO3- move through the cell mem-
Pco2. If the rate of pulmonary ventilation is increased,
branes often delays for several hours the maximum
CO2 is blown off from the lungs, and the Pco2 in the
ability of the intracellular proteins to buffer extracel-
extracellular fluid decreases. Therefore, changes in
lular acid-base abnormalities.
either pulmonary ventilation or the rate of CO2 for-
In addition to the high concentration of proteins in
mation by the tissues can change the extracellular fluid
the cells, another factor that contributes to their
Pco2.
buffering power is the fact that the pKs of many of
these protein systems are fairly close to 7.4.
Increasing Alveolar Ventilation
Decreases Extracellular Fluid
Isohydric Principle: All Buffers
Hydrogen Ion Concentration and
in a Common Solution Are
Raises pH
in Equilibrium with the Same
Hydrogen Ion Concentration
If the metabolic formation of CO2 remains constant,
We have been discussing buffer systems as though they
the only other factor that affects Pco2 in extracellular
operated individually in the body fluids. However, they
fluid is the rate of alveolar ventilation. The higher the
all work together, because H+ is common to the reac-
alveolar ventilation, the lower the Pco2; conversely, the
tions of all the systems. Therefore, whenever there is a
lower the alveolar ventilation rate, the higher the Pco2.
change in H+ concentration in the extracellular fluid, the
As discussed previously, when CO2
concentration
balance of all the buffer systems changes at the same
increases, the H2CO3 concentration and H+ concentra-
time. This phenomenon is called the isohydric principle
tion also increase, thereby lowering extracellular
and is illustrated by the following formula:
fluid pH.
HA
1
2
3
Figure
30-2 shows the approximate changes in
H+=K
1
¥
=
2
K ¥HA
=
3
K ¥HA
blood pH that are caused by increasing or decreasing
A
1
A
2
A
3
the rate of alveolar ventilation. Note that increasing
K1, K2, K3 are the dissociation constants of three
alveolar ventilation to about twice normal raises the
respective acids, HA1, HA2, HA3, and A1, A2, A3 are the
pH of the extracellular fluid by about 0.23. If the pH
concentrations of the free negative ions that constitute
of the body fluids is 7.40 with normal alveolar ventila-
the bases of the three buffer systems.
tion, doubling the ventilation rate raises the pH to
The implication of this principle is that any condition
that changes the balance of one of the buffer systems
also changes the balance of all the others because the
buffer systems actually buffer one another by shifting
H+ back and forth between them.
+0.3
+0.2
Respiratory Regulation
+0.1
of Acid-Base Balance
0
Normal
The second line of defense against acid-base distur-
-0.1
bances is control of extracellular fluid CO2 concentra-
-0.2
tion by the lungs. An increase in ventilation eliminates
-0.3
CO2 from extracellular fluid, which, by mass action,
reduces the H+ concentration. Conversely, decreased
-0.4
ventilation increases CO2, thus also increasing H+ con-
-0.5
centration in the extracellular fluid.
0.5
1.0
1.5
2.0
2.5
Rate of alveolar ventilation
(normal = 1)
Pulmonary Expiration of CO2
Balances Metabolic Formation of CO2
Figure 30-2
CO2 is formed continually in the body by intracellular
metabolic processes. After it is formed, it diffuses from
Change in extracellular fluid pH caused by increased or
the cells into the interstitial fluids and blood, and the
decreased rate of alveolar ventilation, expressed as times normal.
Chapter 30
Regulation of Acid-Base Balance
389
about 7.63. Conversely, a decrease in alveolar ventila-
Feedback Control of Hydrogen Ion Concentration by the Respi-
tion to one fourth normal reduces the pH by 0.45. That
ratory System. Because increased H+ concentration
is, if the pH is 7.4 at a normal alveolar ventilation,
stimulates respiration, and because increased alveolar
reducing the ventilation to one fourth normal reduces
ventilation decreases the H+ concentration, the respi-
the pH to 6.95. Because the alveolar ventilation rate
ratory system acts as a typical negative feedback con-
can change markedly, from as low as 0 to as high as 15
troller of H+ concentration.
times normal, one can easily understand how much the
≠[H+] Æ ≠Alveolar ventilation
pH of the body fluids can be changed by the respira-
tory system.
-
Ø
ØPCO2
Increased Hydrogen Ion
That is, whenever the H+ concentration increases
above normal, the respiratory system is stimulated,
Concentration Stimulates
and alveolar ventilation increases. This decreases the
Alveolar Ventilation
PCO2 in extracellular fluid and reduces H+ concentra-
tion back toward normal. Conversely, if H+ concen-
Not only does the alveolar ventilation rate influence
tration falls below normal, the respiratory center
H+ concentration by changing the Pco2 of the body
becomes depressed, alveolar ventilation decreases, and
fluids, but the H+ concentration affects the rate of
H+ concentration increases back toward normal.
alveolar ventilation. Thus, Figure 30-3 shows that the
alveolar ventilation rate increases four to five times
Efficiency of Respiratory Control of Hydrogen Ion Concen-
normal as the pH decreases from the normal value of
tration. Respiratory control cannot return the H+
7.4 to the strongly acidic value of 7.0. Conversely, when
concentration all the way back to normal when a dis-
plasma pH rises above 7.4, this causes a decrease in the
turbance outside the respiratory system has altered
ventilation rate. As one can see from the graph, the
pH. Ordinarily, the respiratory mechanism for con-
change in ventilation rate per unit pH change is much
trolling H+ concentration has an effectiveness between
greater at reduced levels of pH (corresponding to ele-
50 and 75 per cent, corresponding to a feedback gain
vated H+ concentration) compared with increased
of 1 to 3. That is, if the H+ concentration is suddenly
levels of pH. The reason for this is that as the alveolar
increased by adding acid to the extracellular fluid and
ventilation rate decreases, owing to an increase in pH
pH falls from 7.4 to 7.0, the respiratory system can
(decreased H+ concentration), the amount of oxygen
return the pH to a value of about 7.2 to 7.3. This
added to the blood decreases and the partial pressure
response occurs within 3 to 12 minutes.
of oxygen (PO2) in the blood also decreases, which
stimulates the ventilation rate. Therefore, the respira-
Buffering Power of the Respiratory System. Respiratory reg-
tory compensation for an increase in pH is not nearly
ulation of acid-base balance is a physiologic type of
as effective as the response to a marked reduction in
+
buffer system because it acts rapidly and keeps the H
pH.
concentration from changing too much until the slowly
responding kidneys can eliminate the imbalance. In
general, the overall buffering power of the respiratory
system is one to two times as great as the buffering
power of all other chemical buffers in the extracellu-
lar fluid combined. That is, one to two times as much
acid or base can normally be buffered by this mecha-
nism as by the chemical buffers.
4
Impairment of Lung Function Can Cause Respiratory Acidosis.
We have discussed thus far the role of the normal res-
3
piratory mechanism as a means of buffering changes
in H+ concentration. However, abnormalities of respi-
ration can also cause changes in H+ concentration. For
2
example, an impairment of lung function, such as
severe emphysema, decreases the ability of the lungs
1
to eliminate CO2; this causes a buildup of CO2 in the
extracellular fluid and a tendency toward respiratory
acidosis. Also, the ability to respond to metabolic aci-
0
7.0
7.1
7.2
7.3
7.4
7.5
7.6
dosis is impaired because the compensatory reduc-
pH of arterial blood
tions in PCO2 that would normally occur by means of
increased ventilation are blunted. In these circum-
stances, the kidneys represent the sole remaining phys-
iologic mechanism for returning pH toward normal
Figure 30-3
after the initial chemical buffering in the extracellular
Effect of blood pH on the rate of alveolar ventilation.
fluid has occurred.
390
Unit V The Body Fluids and Kidneys
Renal Control of
body of the nonvolatile acids produced each day, for a
total of 4400 milliequivalents of H+ secreted into the
Acid-Base Balance
tubular fluid each day.
When there is a reduction in the extracellular fluid
The kidneys control acid-base balance by excreting
H+ concentration (alkalosis), the kidneys fail to reab-
either an acidic or a basic urine. Excreting an acidic
sorb all the filtered bicarbonate, thereby increasing the
urine reduces the amount of acid in extracellular fluid,
excretion of bicarbonate. Because HCO3- normally
whereas excreting a basic urine removes base from the
buffers hydrogen in the extracellular fluid, this loss of
extracellular fluid.
bicarbonate is the same as adding an H+ to the extra-
The overall mechanism by which the kidneys
cellular fluid. Therefore, in alkalosis, the removal of
excrete acidic or basic urine is as follows: Large
HCO3- raises the extracellular fluid H+ concentration
numbers of HCO3- are filtered continuously into the
back toward normal.
tubules, and if they are excreted into the urine, this
In acidosis, the kidneys do not excrete bicarbonate
removes base from the blood. Large numbers of H+ are
into the urine but reabsorb all the filtered bicarbonate
also secreted into the tubular lumen by the tubular
and produce new bicarbonate, which is added back to
epithelial cells, thus removing acid from the blood. If
the extracellular fluid. This reduces the extracellular
more H+ is secreted than HCO3- is filtered, there will
fluid H+ concentration back toward normal.
be a net loss of acid from the extracellular fluid. Con-
Thus, the kidneys regulate extracellular fluid H+ con-
versely, if more HCO3- is filtered than H+ is secreted,
centration through three fundamental mechanisms: (1)
there will be a net loss of base.
secretion of H+, (2) reabsorption of filtered HCO3-, and
As discussed previously, each day the body produces
(3) production of new HCO3-. All these processes are
about 80 milliequivalents of nonvolatile acids, mainly
accomplished through the same basic mechanism, as
from the metabolism of proteins. These acids are called
discussed in the next few sections.
nonvolatile because they are not H2CO3 and, there-
fore, cannot be excreted by the lungs. The primary
mechanism for removal of these acids from the body
is renal excretion. The kidneys must also prevent the
Secretion of Hydrogen Ions
loss of bicarbonate in the urine, a task that is quanti-
and Reabsorption of
tatively more important than the excretion of non-
Bicarbonate Ions by the
volatile acids. Each day the kidneys filter about 4320
milliequivalents of bicarbonate (180 L/day ¥ 24 mEq/
Renal Tubules
L); under normal conditions, almost all this is reab-
sorbed from the tubules, thereby conserving the
Hydrogen ion secretion and bicarbonate reabsorption
primary buffer system of the extracellular fluid.
occur in virtually all parts of the tubules except the
As discussed later, both the reabsorption of bicar-
descending and ascending thin limbs of the loop of
bonate and the excretion of H+ are accomplished
Henle. Figure 30-4 summarizes bicarbonate reabsorp-
through the process of H+ secretion by the tubules.
tion along the tubule. Keep in mind that for each bicar-
Because the HCO3- must react with a secreted H+ to
bonate reabsorbed, an H+ must be secreted.
form H2CO3 before it can be reabsorbed, 4320 mil-
About 80 to 90 per cent of the bicarbonate reab-
liequivalents of H+ must be secreted each day just to
sorption (and H+ secretion) occurs in the proximal
reabsorb the filtered bicarbonate. Then an additional
tubule, so that only a small amount of bicarbonate
80 milliequivalents of H+ must be secreted to rid the
flows into the distal tubules and collecting ducts. In the
85%
(3672 mEq/day)
4320 mEq/day
10%
(432 mEq/day)
>4.9%
(215 mEq/day)
Figure 30-4
Reabsorption of bicarbonate in differ-
ent segments of the renal tubule. The
percentages of the filtered load of
bicarbonate absorbed by the various
tubular segments are shown, as well
as the number of milliequivalents
reabsorbed per day under normal
(1 mEq/day)
conditions.
Chapter 30
Regulation of Acid-Base Balance
391
thick ascending loop of Henle, another 10 per cent of
process begins when CO2 either diffuses into the
the filtered bicarbonate is reabsorbed, and the remain-
tubular cells or is formed by metabolism in the tubular
der of the reabsorption takes place in the distal tubule
epithelial cells. CO2, under the influence of the enzyme
and collecting duct. As discussed previously, the mech-
carbonic anhydrase, combines with H2O to form
anism by which bicarbonate is reabsorbed also
H2CO3, which dissociates into HCO3- and H+. The H+
involves tubular secretion of H+, but different tubular
is secreted from the cell into the tubular lumen by
segments accomplish this task differently.
sodium-hydrogen counter-transport. That is, when an
Na+ moves from the lumen of the tubule to the inte-
rior of the cell, it first combines with a carrier protein
Hydrogen Ions Are Secreted
in the luminal border of the cell membrane; at the
by Secondary Active Transport
same time, an H+ in the interior of the cells combines
with the carrier protein. The Na+ moves into the cell
in the Early Tubular Segments
down a concentration gradient that has been estab-
lished by the sodium-potassium ATPase pump in the
The epithelial cells of the proximal tubule, the thick
basolateral membrane. The gradient for Na+ move-
segment of the ascending loop of Henle, and the early
ment into the cell then provides the energy for moving
distal tubule all secrete H+ into the tubular fluid
H+ in the opposite direction from the interior of the
by sodium-hydrogen counter-transport, as shown in
cell to the tubular lumen.
Figure 30-5. This secondary active secretion of H+
The HCO3- generated in the cell (when H+ dissoci-
is coupled with the transport of Na+ into the cell at
ates from H2CO3) then moves downhill across the
the luminal membrane by the sodium-hydrogen
basolateral membrane into the renal interstitial fluid
exchanger protein, and the energy for H+ secretion
and the peritubular capillary blood. The net result is
against a concentration gradient is derived from the
that for every H+ secreted into the tubular lumen, an
sodium gradient favoring Na+ movement into the cell.
HCO3- enters the blood.
This gradient is established by the sodium-potassium
adenosine triphosphatase (ATPase) pump in the baso-
lateral membrane. More than 90 per cent of the bicar-
bonate is reabsorbed in this manner, requiring about
Filtered Bicarbonate Ions Are
3900 milliequivalents of H+ to be secreted each day by
Reabsorbed by Interaction with
the tubules. This mechanism, however, does not estab-
Hydrogen Ions in the Tubules
lish a very high H+ concentration in the tubular fluid;
the tubular fluid becomes very acidic only in the col-
Bicarbonate ions do not readily permeate the luminal
lecting tubules and collecting ducts.
membranes of the renal tubular cells; therefore,
Figure 30-5 shows how the process of H+ secretion
HCO3- that is filtered by the glomerulus cannot be
achieves bicarbonate reabsorption. The secretory
directly reabsorbed. Instead, HCO3- is reabsorbed by
a special process in which it first combines with H+ to
form H2CO3, which eventually becomes CO2 and H2O,
as shown in Figure 30-5.
Renal
Tubular
This reabsorption of HCO3- is initiated by a reaction
interstitial
Tubular cells
lumen
in the tubules between HCO3- filtered at the glomeru-
fluid
Na+ + HCO3-
3
lus and H+ secreted by the tubular cells. The H2CO3
formed then dissociates into CO2 and H2O. The CO2
can move easily across the tubular membrane; there-
Na+
Na+
fore, it instantly diffuses into the tubular cell, where it
ATP
K+
H+
recombines with H2O, under the influence of carbonic
3
HCO3- + H+
anhydrase, to generate a new H2CO3 molecule. This
H2CO
3
H2CO3 in turn dissociates to form HCO3- and H+; the
H2CO3
HCO3- then diffuses through the basolateral mem-
3
Carbonic
brane into the interstitial fluid and is taken up into the
anhydrase
peritubular capillary blood. The transport of HCO3
H2O
CO
+
across the basolateral membrane is facilitated by
2
CO
2
CO2 + H2O
two mechanisms: (1) Na+-HCO3- co-transport and (2)
Cl--HCO3- exchange.
Thus, each time an H+ is formed in the tubular epithe-
Figure 30-5
lial cells, an HCO
3
- is also formed and released back
into the blood. The net effect of these reactions is
Cellular mechanisms for (1) active secretion of hydrogen ions into
the renal tubule; (2) tubular reabsorption of bicarbonate ions by
“reabsorption” of HCO3- from the tubules, although
combination with hydrogen ions to form carbonic acid, which dis-
the HCO3- that actually enters the extracellular fluid
sociates to form carbon dioxide and water; and (3) sodium ion
is not the same as that filtered into the tubules. The
reabsorption in exchange for hydrogen ions secreted. This pattern
reabsorption of filtered HCO3- does not result in net
of hydrogen ion secretion occurs in the proximal tubule, the thick
secretion of H+ because the secreted H+ combines with
ascending segment of the loop of Henle, and the early distal
tubule.
the filtered HCO3- and is therefore not excreted.
392
Unit V The Body Fluids and Kidneys
Bicarbonate Ions Are “Titrated” Against Hydrogen Ions in the
Renal
Tubular
Tubules. Under normal conditions, the rate of tubular
interstitial
Tubular cells
lumen
H+ secretion is about 4400 mEq/day, and the rate of fil-
fluid
tration by HCO3- is about 4320 mEq/day. Thus, the
quantities of these two ions entering the tubules are
Cl-
Cl-
Cl-
almost equal, and they combine with each other to
-
form CO2 and H2O. Therefore, it is said that HCO3
HCO3- + H+
3
H+
and H+ normally “titrate” each other in the tubules.
ATP
The titration process is not quite exact because there
H2CO3
3
is usually a slight excess of H+ in the tubules to be
Carbonic
excreted in the urine. This excess H+ (about 80 mEq/
anhydrase
day) rids the body of nonvolatile acids produced by
H2O
+
metabolism. As discussed later, most of this H+ is
CO2
CO2
2
2
not excreted as free H+ but rather in combination with
other urinary buffers, especially phosphate and
ammonia.
When there is an excess of HCO3- over H+ in the
Figure 30-6
urine, as occurs in metabolic alkalosis, the excess
Primary active secretion of hydrogen ions through the luminal
HCO3- cannot be reabsorbed; therefore, the excess
membrane of the intercalated epithelial cells of the late distal and
HCO3- is left in the tubules and eventually excreted
collecting tubules. Note that one bicarbonate ion is absorbed for
into the urine, which helps correct the metabolic
each hydrogen ion secreted, and a chloride ion is passively
alkalosis.
secreted along with the hydrogen ion.
In acidosis, there is excess H+ relative to HCO3-,
causing complete reabsorption of the bicarbonate;
the excess H+ passes into the urine. The excess H+ is
buffered in the tubules by phosphate and ammonia
instead of by counter-transport, as occurs in the early
and eventually excreted as salts. Thus, the basic mech-
parts of the nephron.
anism by which the kidneys correct either acidosis or
Although the secretion of H+ in the late distal tubule
alkalosis is incomplete titration of H+ against HCO3-,
and collecting tubules accounts for only about 5 per
leaving one or the other to pass into the urine and be
cent of the total H+ secreted, this mechanism is impor-
removed from the extracellular fluid.
tant in forming a maximally acidic urine. In the prox-
imal tubules, H+ concentration can be increased only
Primary Active Secretion of Hydrogen
about threefold to fourfold, and the tubular fluid pH
Ions in the Intercalated Cells of Late
can be reduced to only about 6.7, although large
amounts of H+ are secreted by this nephron segment.
Distal and Collecting Tubules
However, H+ concentration can be increased as much
as 900-fold in the collecting tubules. This decreases the
Beginning in the late distal tubules and continuing
pH of the tubular fluid to about 4.5, which is the lower
through the remainder of the tubular system, the
limit of pH that can be achieved in normal kidneys.
tubular epithelium secretes H+ by primary active
transport. The characteristics of this transport are dif-
ferent from those discussed for the proximal tubule,
Combination of Excess
loop of Henle, and early distal tubule.
The mechanism for primary active H+ secretion is
Hydrogen Ions with Phosphate
shown in Figure 30-6. It occurs at the luminal mem-
and Ammonia Buffers in
brane of the tubular cell, where H+ is transported
the Tubule—A Mechanism
directly by a specific protein, a hydrogen-transporting
for Generating “New”
ATPase. The energy required for pumping the H+ is
derived from the breakdown of ATP to adenosine
Bicarbonate Ions
diphosphate.
Primary active secretion of H+ occurs in a special
When H+ is secreted in excess of the bicarbonate fil-
type of cell called the intercalated cells of the late distal
tered into the tubular fluid, only a small part of the
tubule and in the collecting tubules. Hydrogen ion
excess H+ can be excreted in the ionic form (H+) in the
secretion in these cells is accomplished in two steps:
urine. The reason for this is that the minimal urine pH
(1) the dissolved CO2 in this cell combines with H2O
is about 4.5, corresponding to an H+ concentration
to form H2CO3, and (2) the H2CO3 then dissociates
of 10-4.5 mEq/L, or 0.03 mEq/L. Thus, for each liter of
into HCO3-, which is reabsorbed into the blood, plus
urine formed, a maximum of only about 0.03 mil-
H+, which is secreted into the tubule by means of the
liequivalent of free H+ can be excreted. To excrete the
hydrogen-ATPase mechanism. For each H+ secreted,
80 milliequivalents of nonvolatile acid formed by
an HCO3- is reabsorbed, similar to the process in the
metabolism each day, about 2667 liters of urine would
proximal tubules. The main difference is that H+ moves
have to be excreted if the H+ remained free in
across the luminal membrane by an active H+ pump
solution.
Chapter 30
Regulation of Acid-Base Balance
393
The excretion of large amounts of H+ (on occasion
Renal
Tubular
interstitial
lumen
as much as 500 mEq/day) in the urine is accomplished
Tubular cells
fluid
Na+ + NaHPO4-
primarily by combining the H+ with buffers in the
4
tubular fluid. The most important buffers are phos-
phate buffer and ammonia buffer. There are other
Na+
Na+
Na+
weak buffer systems, such as urate and citrate, that are
ATP
much less important.
K+
H+ + NaHPO4-
4
HCO3-
HCO3- + H+
When H+ is titrated in the tubular fluid with HCO3-,
3
3
this results in the reabsorption of one HCO3- for each
H+ secreted, as discussed earlier. But when there are
H2CO3
NaH2PO4
4
excess H+ in the urine, they combine with buffers other
3 Carbonic
anhydrase
than HCO3-, and this results in the generation of new
H2O
HCO3- that can also enter the blood. Thus, when there
CO2
+
2
is excess H+ in the extracellular fluid, the kidneys not
CO2
2
only reabsorb all the filtered HCO3- but also generate
-
new HCO3-, thereby helping to replenish the HCO3
Figure 30-7
lost from the extracellular fluid in acidosis. In the next
two sections, we discuss the mechanisms by which
Buffering of secreted hydrogen ions by filtered phosphate
phosphate and ammonia buffers contribute to the gen-
(NaHPO4-). Note that a new bicarbonate ion is returned to the
eration of new HCO3-.
blood for each NaHPO4- that reacts with a secreted hydrogen ion.
Phosphate Buffer System Carries
HCO3- to the blood. This demonstrates one of the
Excess Hydrogen Ions into the Urine
mechanisms by which the kidneys are able to replen-
and Generates New Bicarbonate
ish the extracellular fluid stores of HCO3-.
Under normal conditions, much of the filtered phos-
=
phate is reabsorbed, and only about 30 to 40 mEq/day
The phosphate buffer system is composed of HPO4
and H2PO4-. Both become concentrated in the tubular
is available for buffering H+. Therefore, much of the
fluid because of their relatively poor reabsorption and
buffering of excess H+ in the tubular fluid in acidosis
because of the reabsorption of water from the tubular
occurs through the ammonia buffer system.
fluid. Therefore, although phosphate is not an impor-
tant extracellular fluid buffer, it is much more effective
Excretion of Excess Hydrogen Ions
as a buffer in the tubular fluid.
Another factor that makes phosphate important as
and Generation of New Bicarbonate
a tubular buffer is the fact that the pK of this system
by the Ammonia Buffer System
is about 6.8. Under normal conditions, the urine is
slightly acidic, and the urine pH is near the pK of the
A second buffer system in the tubular fluid that is even
phosphate buffer system. Therefore, in the tubules, the
more important quantitatively than the phosphate
phosphate buffer system normally functions near its
buffer system is composed of ammonia (NH3) and the
most effective range of pH.
ammonium ion (NH4+). Ammonium ion is synthesized
Figure 30-7 shows the sequence of events by which
from glutamine, which comes mainly from the metab-
H+ is excreted in combination with phosphate buffer
olism of amino acids in the liver. The glutamine deliv-
and the mechanism by which new bicarbonate is added
ered to the kidneys is transported into the epithelial
to the blood. The process of H+ secretion into the
cells of the proximal tubules, thick ascending limb of
tubules is the same as described earlier. As long as
the loop of Henle, and distal tubules (Figure 30-8).
there is excess HCO3- in the tubular fluid, most of the
Once inside the cell, each molecule of glutamine is
secreted H+ combines with HCO3-. However, once all
metabolized in a series of reactions to ultimately form
the HCO3- has been reabsorbed and is no longer avail-
two NH4+ and two HCO3-. The NH4+ is secreted into
able to combine with H+, any excess H+ can combine
the tubular lumen by a counter-transport mechanism
with HPO4= and other tubular buffers. After the H+
in exchange for sodium, which is reabsorbed. The
combines with HPO4= to form H2PO4-, it can be
HCO3- is transported across the basolateral mem-
excreted as a sodium salt (NaH2PO4), carrying with it
brane, along with the reabsorbed Na+, into the inter-
the excess hydrogen.
stitial fluid and is taken up by the peritubular
There is one important difference in this sequence
capillaries. Thus, for each molecule of glutamine
of H+ excretion from that discussed previously. In this
metabolized in the proximal tubules, two NH4+ are
case, the HCO3- that is generated in the tubular cell
secreted into the urine and two HCO3- are reabsorbed
and enters the peritubular blood represents a net gain
into the blood. The HCO
3
- generated by this process
of HCO3- by the blood, rather than merely a replace-
constitutes new bicarbonate.
ment of filtered HCO3-. Therefore, whenever an H+
In the collecting tubules, the addition of NH4+ to the
secreted into the tubular lumen combines with a buffer
tubular fluids occurs through a different mechanism
other than HCO
3
-, the net effect is addition of a new
(Figure 30-9). Here, H+ is secreted by the tubular
394
Unit V The Body Fluids and Kidneys
Renal
Tubular
An increase in extracellular fluid H+ concentration
Proximal
interstitial
lumen
tubular cells
stimulates renal glutamine metabolism and, therefore,
fluid
increases the formation of NH4+ and new HCO3- to be
used in H+ buffering; a decrease in H+ concentration
has the opposite effect.
Under normal conditions, the amount of H+ elimi-
Glutamine
Glutamine
Glutamine
nated by the ammonia buffer system accounts for
about 50 per cent of the acid excreted and 50 per cent
Cl-
of the new HCO3- generated by the kidneys. However,
2HCO3-
3
2NH4+
4
with chronic acidosis, the rate of NH4+ excretion can
increase to as much as 500 mEq/day. Therefore, with
+
chronic acidosis, the dominant mechanism by which
NH4+
NH
+ Cl-
4
4
acid is eliminated is excretion of NH4+. This also pro-
Na+
Na+
vides the most important mechanism for generating
new bicarbonate during chronic acidosis.
Figure 30-8
Quantifying Renal
Production and secretion of ammonium ion (NH4+) by proximal
+
Acid-Base Excretion
tubular cells. Glutamine is metabolized in the cell, yielding NH4
and bicarbonate. The NH4+ is secreted into the lumen by a
sodium-NH4+ pump. For each glutamine molecule metabolized,
Based on the principles discussed earlier, we can quan-
two NH4+ are produced and secreted and two HCO3- are returned
to the blood.
titate the kidneys’ net excretion of acid or net addition
or elimination of bicarbonate from the blood as
follows.
Renal
Tubular
Bicarbonate excretion is calculated as the urine flow
Collecting
interstitial
lumen
rate multiplied by urinary bicarbonate concentration.
tubular cells
fluid
This number indicates how rapidly the kidneys are
removing HCO3- from the blood (which is the same as
adding an H+ to the blood). In alkalosis, the loss of
Na+
NH3
NH3
3
3
HCO3- helps return the plasma pH toward normal.
ATP
K+
Cl-
The amount of new bicarbonate contributed to
HCO3- + H+
ATP
3
the blood at any given time is equal to the amount of
H+
H+ secreted that ends up in the tubular lumen with
H2CO
nonbicarbonate urinary buffers. As discussed previ-
3
Carbonic
NH4+ + Cl-
ously, the primary sources of nonbicarbonate urinary
4
anhydrase
buffers are NH4+ and phosphate. Therefore, the
H2O
CO2
+
amount of HCO3- added to the blood (and H+ excreted
2
CO
by NH4+) is calculated by measuring NH4+
2
excretion (urine flow rate multiplied by urinary NH4+
concentration).
Figure 30-9
The rest of the nonbicarbonate, non-NH4+ buffer
excreted in the urine is measured by determining a
Buffering of hydrogen ion secretion by ammonia (NH3) in the col-
value known as titratable acid. The amount of titrat-
lecting tubules. Ammonia diffuses into the tubular lumen, where it
reacts with secreted hydrogen ions to form NH4+, which is then
able acid in the urine is measured by titrating the urine
excreted. For each NH4+ excreted, a new HCO3- is formed in the
with a strong base, such as NaOH, to a pH of 7.4, the
tubular cells and returned to the blood.
pH of normal plasma, and the pH of the glomerular
filtrate. This titration reverses the events that occurred
in the tubular lumen when the tubular fluid was
membrane into the lumen, where it combines with
titrated by excreted H+. Therefore, the number of mil-
NH3 to form NH4+, which is then excreted. The col-
liequivalents of NaOH required to return the urinary
lecting ducts are permeable to NH3, which can easily
pH to 7.4 equals the number of milliequivalents of H+
diffuse into the tubular lumen. However, the luminal
added to the tubular fluid that combined with phos-
membrane of this part of the tubules is much less per-
phate and other organic buffers. The titratable acid
meable to NH4+; therefore, once the H+ has reacted
measurement does not include H+ in association with
with NH3 to form NH4+, the NH4+ is trapped in the
NH4+, because the pK of the ammonia-ammonium
tubular lumen and eliminated in the urine. For each
reaction is 9.2, and titration with NaOH to a pH of 7.4
NH4+ excreted, a new HCO3- is generated and added to
does not remove the H+ from NH4+.
the blood.
Thus, the net acid excretion by the kidneys can be
assessed as
Chronic Acidosis Increases NH4+ Excretion. One of the most
important features of the renal ammonium-ammonia
Net acid excretion = NH4+ excretion + Urinary
buffer system is that it is subject to physiologic control.
titratable acid - Bicarbonate excretion
Chapter 30
Regulation of Acid-Base Balance
395
The reason we subtract bicarbonate excretion is that
Table 30-2
the loss of HCO3- is the same as the addition of H+ to
-
Factors That Increase or Decrease H+
Secretion and HCO3
the blood. To maintain acid-base balance, the net acid
Reabsorption by the Renal Tubules
excretion must equal the nonvolatile acid production
in the body. In acidosis, the net acid excretion increases
markedly, especially because of increased NH4+ excre-
Increase H+ Secretion and
Decrease H+ Secretion and
HCO3- Reabsorption
HCO3- Reabsorption
tion, thereby removing acid from the blood. The net
acid excretion also equals the rate of net HCO3- addi-
≠ PCO2
Ø PCO2
tion to the blood. Therefore, in acidosis, there is a net
-
≠ H+, Ø HCO3-
ØH+, ≠ HCO3
addition of HCO3- back to the blood as more NH4+ and
Ø Extracellular fluid volume
≠ Extracellular fluid volume
urinary titratable acid are excreted.
In alkalosis, titratable acid and NH4+ excretion drop
≠ Angiotensin II
Ø Angiotensin II
to 0, whereas HCO3- excretion increases. Therefore, in
≠ Aldosterone
Ø Aldosterone
alkalosis, there is a negative net acid secretion. This
Hypokalemia
Hyperkalemia
means that there is a net loss of HCO3- from the blood
(which is the same as adding H+ to the blood) and that
no new HCO3- is generated by the kidneys.
A special factor that can increase H+ secretion under
some pathophysiologic conditions is excessive aldos-
terone secretion. Aldosterone stimulates the secretion
Regulation of Renal Tubular Hydrogen
of H+ by the intercalated cells of the collecting duct.
Ion Secretion
Therefore, oversecretion of aldosterone, as occurs in
Conn’s syndrome, can cause excessive secretion of H+
As discussed earlier, H+ secretion by the tubular
into the tubular fluid and, consequently, increased
epithelium is necessary for both HCO3- reabsorption
amounts of bicarbonate added back to the blood. This
and generation of new HCO3- associated with titrat-
usually causes alkalosis in patients with excessive
able acid formation. Therefore, the rate of H+ secre-
aldosterone secretion.
tion must be carefully regulated if the kidneys are to
The tubular cells usually respond to a decrease in H+
effectively perform their functions in acid-base home-
concentration (alkalosis) by reducing H+ secretion.
ostasis. Under normal conditions, the kidney tubules
The decreased H+ secretion results from decreased
must secrete at least enough H+ to reabsorb almost all
extracellular PCO2, as occurs in respiratory alkalosis, or
the HCO3- that is filtered, and there must be enough
from a decrease in H+ concentration per se, as occurs
+
H+
left over to be excreted as titratable acid or NH4
in both respiratory and metabolic alkalosis.
to rid the body of the nonvolatile acids produced each
Table 30-2 summarizes the major factors that influ-
day from metabolism.
ence H+ secretion and HCO3- reabsorption. Some of
In alkalosis, tubular secretion of H+ must be reduced
these are not directly related to the regulation of acid-
-
to a level that is too low to achieve complete HCO3
base balance. For example, H+ secretion is coupled
–
reabsorption, enabling the kidneys to increase HCO3
to Na+ reabsorption by the Na+-H+ exchanger in the
excretion. In this condition, titratable acid and
proximal tubule and thick ascending loop of Henle.
ammonia are not excreted because there is no excess
Therefore, factors that stimulate Na+ reabsorption,
H+ available to combine with nonbicarbonate buffers;
such as decreased extracellular fluid volume, may also
therefore, there is no new HCO3- added to the
secondarily increase H+ secretion.
urine in alkalosis. During acidosis, the tubular H+
Extracellular fluid volume depletion stimulates
secretion must be increased sufficiently to reabsorb
sodium reabsorption by the renal tubules and
all the filtered HCO3- and still have enough H+ left
increases H+ secretion and HCO3- reabsorption
over to excrete large amounts of NH4+ and titratable
through multiple mechanisms, including (1) increased
acid, thereby contributing large amounts of new
angiotensin II levels, which directly stimulate the activ-
HCO3- to the total body extracellular fluid. The
ity of the Na+-H+ exchanger in the renal tubules, and
most important stimuli for increasing H+ secretion by
(2) increased aldosterone levels, which stimulate H+
the tubules in acidosis are (1) an increase in PCO2
secretion by the intercalated cells of the cortical col-
of the extracellular fluid and (2) an increase in H+
lecting tubules. Therefore, extracellular fluid volume
concentration of the extracellular fluid
(decreased
depletion tends to cause alkalosis due to excess H+
pH).
secretion and HCO3- reabsorption.
The tubular cells respond directly to an increase in
Changes in plasma potassium concentration can
PCO2 of the blood, as occurs in respiratory acidosis,
also influence H+ secretion, with hypokalemia stimu-
with an increase in the rate of H+ secretion as follows:
lating and hyperkalemia inhibiting H+ secretion in the
The increased PCO2 raises the PCO2 of the tubular cells,
proximal tubule. A decreased plasma potassium con-
causing increased formation of H+ in the tubular
centration tends to increase the H+ concentration in
cells, which in turn stimulates the secretion of H+. The
the renal tubular cells. This, in turn, stimulates H+
second factor that stimulates H+ secretion is an
secretion and HCO3- reabsorption and leads to alka-
-
increase in extracellular fluid H+ concentration
losis. Hyperkalemia decreases H+
secretion and HCO3
(decreased pH).
reabsorption and tends to cause acidosis.
396
Unit V The Body Fluids and Kidneys
Renal Correction of Acidosis—
Table 30-3
Increased Excretion of
Characteristics of Primary Acid-Base Disturbances
Hydrogen Ions and Addition
-
of Bicarbonate Ions to the
pH H+
PCO2
HCO3
Extracellular Fluid
Normal
7.4
40 mEq/L
40 mm Hg
24 mEq/L
Respiratory acidosis
Ø
≠
≠≠
≠
Now that we have described the mechanisms by which
Respiratory alkalosis
≠ Ø
ØØ
Ø
the kidneys secrete H+ and reabsorb HCO3-, we can
explain how the kidneys readjust the pH of the extra-
Metabolic acidosis
Ø
≠
Ø
ØØ
cellular fluid when it becomes abnormal.
Metabolic alkalosis
≠ Ø
≠
≠≠
Referring to equation
8, the Henderson-
Hasselbalch equation, we can see that acidosis occurs
The primary event is indicated by the double arrows (≠≠ or ØØ). Note that
respiratory acid-base disorders are initiated by an increase or decrease in
when the ratio of HCO3- to CO2 in the extracellular
PCO2, whereas metabolic disorders are initiated by an increase or decrease in
fluid decreases, thereby decreasing pH. If this ratio
HCO3-.
decreases because of a fall in HCO3-, the acidosis is
referred to as metabolic acidosis. If the pH falls
respiratory and metabolic alkalosis, which are dis-
because of an increase in PCO2, the acidosis is referred
cussed in the next section. Note that in respiratory
to as respiratory acidosis.
acidosis, there is a reduction in pH, an increase in
extracellular fluid H+ concentration, and an increase in
Acidosis Decreases the Ratio of
PCO2, which is the initial cause of the acidosis. The
HCO3-/H+ in Renal Tubular Fluid
compensatory response is an increase in plasma
HCO3-, caused by the addition of new bicarbonate to
-
Both respiratory and metabolic acidosis cause a
the extracellular fluid by the kidneys. The rise in HCO3
decrease in the ratio of HCO3- to H+ in the renal
helps offset the increase in PCO2, thereby returning the
tubular fluid. As a result, there is an excess of H+ in the
plasma pH toward normal.
-
renal tubules, causing complete reabsorption of HCO3
In metabolic acidosis, there is also a decrease in pH
and still leaving additional H+ available to combine
and a rise in extracellular fluid H+ concentration.
with the urinary buffers NH4+ and HPO4=. Thus, in aci-
However, in this case, the primary abnormality is a
dosis, the kidneys reabsorb all the filtered HCO3- and
decrease in plasma HCO3-. The primary compensa-
+
contribute new HCO3-
through the formation of NH4
tions include increased ventilation rate, which reduces
and titratable acid.
PCO2, and renal compensation, which, by adding new
-
In metabolic acidosis, an excess of H+
over HCO
3
bicarbonate to the extracellular fluid, helps minimize
occurs in the tubular fluid primarily because of
the initial fall in extracellular HCO3- concentration.
decreased filtration of HCO
3
-. This decreased filtration
of HCO3- is caused mainly by a decrease in the extra-
cellular fluid concentration of HCO3-.
Renal Correction of
In respiratory acidosis, the excess H+ in the tubular
Alkalosis—Decreased Tubular
fluid is due mainly to the rise in extracellular fluid
Secretion of Hydrogen Ions
PCO2, which stimulates H+ secretion.
and Increased Excretion
As discussed previously, with chronic acidosis,
regardless of whether it is respiratory or metabolic,
of Bicarbonate Ions
there is an increase in the production of NH4+, which
further contributes to the excretion of H+ and the addi-
The compensatory responses to alkalosis are basically
tion of new HCO3- to the extracellular fluid. With
opposite to those that occur in acidosis. In alkalosis,
severe chronic acidosis, as much as 500 mEq/day of H+
the ratio of HCO3- to CO2 in the extracellular fluid
can be excreted in the urine, mainly in the form of
increases, causing a rise in pH
(a decrease in H+
NH4+; this, in turn, contributes up to 500 mEq/day of
concentration), as is evident from the Henderson-
new HCO3- that is added to the blood.
Hasselbalch equation.
Thus, with chronic acidosis, the increased secretion
of H+ by the tubules helps eliminate excess H+ from
the body and increases the quantity of HCO3- in the
Alkalosis Increases the Ratio of
extracellular fluid. This increases the bicarbonate part
HCO3-/H+ in Renal Tubular Fluid
of the bicarbonate buffer system, which, in accordance
with the Henderson-Hasselbalch equation, helps raise
Regardless of whether the alkalosis is caused by meta-
the extracellular pH and corrects the acidosis. If the
bolic or respiratory abnormalities, there is still an
acidosis is metabolically mediated, additional com-
increase in the ratio of HCO3- to H+ in the renal
pensation by the lungs causes a reduction in PCO2, also
tubular fluid. The net effect of this is an excess of
helping to correct the acidosis.
HCO3- that cannot be reabsorbed from the tubules
Table 30-3 summarizes the characteristics associ-
and is, therefore, excreted in the urine. Thus, in alka-
ated with respiratory and metabolic acidosis as well as
losis, HCO3- is removed from the extracellular fluid by
Chapter 30
Regulation of Acid-Base Balance
397
renal excretion, which has the same effect as adding an
In respiratory acidosis, the compensatory responses
H+ to the extracellular fluid. This helps return the H+
available are (1) the buffers of the body fluids and (2)
concentration and pH back toward normal.
the kidneys, which require several days to compensate
for the disorder.
Table 30-3 shows the overall characteristics of res-
piratory and metabolic alkalosis. In respiratory alkalo-
sis, there is an increase in extracellular fluid pH and a
Respiratory Alkalosis Results
decrease in H+ concentration. The cause of the alkalo-
from Increased Ventilation and
sis is a decrease in plasma PCO2 , caused by hyperventi-
Decreased PCO2
lation. The reduction in PCO2 then leads to a decrease
in the rate of H+ secretion by the renal tubules. The
Respiratory alkalosis is caused by overventilation by
decrease in H+ secretion reduces the amount of H+ in
the lungs. Rarely does this occur because of physical
the renal tubular fluid. Consequently, there is not
pathological conditions. However, a psychoneurosis can
enough H+ to react with all the HCO3- that is filtered.
occasionally cause overbreathing to the extent that a
Therefore, the HCO3- that cannot react with H+ is not
person becomes alkalotic.
A physiologic type of respiratory alkalosis occurs
reabsorbed and is excreted in the urine. This results in
when a person ascends to high altitude. The low oxygen
a decrease in plasma HCO3- concentration and cor-
content of the air stimulates respiration, which causes
rection of the alkalosis. Therefore, the compensatory
excess loss of CO2 and development of mild respiratory
response to a primary reduction in PCO2 in respiratory
alkalosis. Again, the major means for compensation are
alkalosis is a reduction in plasma HCO3- concentration,
the chemical buffers of the body fluids and the ability
caused by increased renal excretion of HCO3-.
of the kidneys to increase HCO3- excretion.
In metabolic alkalosis, there is also an increase in
plasma pH and a decrease in H+ concentration.
Metabolic Acidosis Results from
The cause of metabolic alkalosis, however, is a rise in
the extracellular fluid HCO3- concentration. This is
Decreased Extracellular Fluid
partly compensated for by a reduction in the respira-
Bicarbonate Concentration
tion rate, which increases PCO2 and helps return the
The term metabolic acidosis refers to all other types of
extracellular fluid pH toward normal. In addition, the
acidosis besides those caused by excess CO2 in the body
increase in HCO3- concentration in the extracellular
fluids. Metabolic acidosis can result from several general
fluid leads to an increase in the filtered load of HCO3-,
causes: (1) failure of the kidneys to excrete metabolic
which in turn causes an excess of HCO3- over H+
acids normally formed in the body, (2) formation of
-
secreted in the renal tubular fluid. The excess HCO3
excess quantities of metabolic acids in the body, (3)
in the tubular fluid fails to be reabsorbed because
addition of metabolic acids to the body by ingestion or
there is no H+ to react with, and it is excreted in the
infusion of acids, and (4) loss of base from the body
urine. In metabolic alkalosis, the primary compensa-
fluids, which has the same effect as adding an acid to the
body fluids. Some specific conditions that cause meta-
tions are decreased ventilation, which raises PCO2 , and
bolic acidosis are the following.
increased renal HCO3- excretion, which helps compen-
-
sate for the initial rise in extracellular fluid HCO3
Renal Tubular Acidosis. This type of acidosis results from
concentration.
a defect in renal secretion of H+ or in reabsorption of
HCO3-, or both. These disorders are generally of two
types: (1) impairment of renal tubular HCO3- reab-
Clinical Causes of
sorption, causing loss of HCO3- in the urine, or (2)
Acid-Base Disorders
inability of the renal tubular H+ secretory mechanism to
establish a normal acidic urine, causing the excretion of
Respiratory Acidosis Is Caused
an alkaline urine. In these cases, inadequate amounts of
by Decreased Ventilation and
titratable acid and NH4+ are excreted, so that there is
Increased PCO2
net accumulation of acid in the body fluids. Some causes
of renal tubular acidosis include chronic renal failure,
From the previous discussion, it is obvious that any
insufficient aldosterone secretion (Addison’s disease),
factor that decreases the rate of pulmonary ventilation
and several hereditary and acquired disorders that
also increases the PCO2 of extracellular fluid. This causes
impair tubular function, such as Fanconi’s syndrome.
an increase in H2CO3 and H+ concentration, thus re-
sulting in acidosis. Because the acidosis is caused by
Diarrhea. Severe diarrhea is probably the most frequent
an abnormality in respiration, it is called respiratory
cause of metabolic acidosis. The cause of this acidosis is
acidosis.
the loss of large amounts of sodium bicarbonate into the
Respiratory acidosis can occur from pathological
feces. The gastrointestinal secretions normally contain
conditions that damage the respiratory centers or
large amounts of bicarbonate, and diarrhea results in
that decrease the ability of the lungs to eliminate CO2.
the loss of HCO3- from the body, which has the same
For example, damage to the respiratory center in the
effect as losing large amounts of bicarbonate in the
medulla oblongata can lead to respiratory acidosis. Also,
urine. This form of metabolic acidosis is particularly
obstruction of the passageways of the respiratory tract,
serious and can cause death, especially in young
pneumonia, emphysema, or decreased pulmonary mem-
children.
brane surface area, as well as any factor that interferes
with the exchange of gases between the blood and the
Vomiting of Intestinal Contents. Vomiting of gastric con-
alveolar air, can cause respiratory acidosis.
tents alone would cause loss of acid and a tendency
398
Unit V The Body Fluids and Kidneys
toward alkalosis because the stomach secretions are
Vomiting of Gastric Contents. Vomiting of the gastric con-
highly acidic. However, vomiting large amounts from
tents alone, without vomiting of the lower gastrointesti-
deeper in the gastrointestinal tract, which sometimes
nal contents, causes loss of the HCl secreted by the
occurs, causes loss of bicarbonate and results in meta-
stomach mucosa. The net result is a loss of acid from the
bolic acidosis in the same way that diarrhea causes
extracellular fluid and development of metabolic alka-
acidosis.
losis. This type of alkalosis occurs especially in neonates
who have pyloric obstruction caused by hypertrophied
Diabetes Mellitus. Diabetes mellitus is caused by lack of
pyloric sphincter muscles.
insulin secretion by the pancreas (type I diabetes) or
by insufficient insulin secretion to compensate for
Ingestion of Alkaline Drugs. A common cause of metabolic
decreased sensitivity to the effects of insulin (type II
alkalosis is ingestion of alkaline drugs, such as sodium
diabetes). In the absence of sufficient insulin, the normal
bicarbonate, for the treatment of gastritis or peptic
use of glucose for metabolism is prevented. Instead,
ulcer.
some of the fats are split into acetoacetic acid, and this
is metabolized by the tissues for energy in place of
glucose. With severe diabetes mellitus, blood acetoacetic
Treatment of Acidosis
acid levels can rise very high, causing severe metabolic
acidosis. In an attempt to compensate for this acidosis,
or Alkalosis
large amounts of acid are excreted in the urine, some-
The best treatment for acidosis or alkalosis is to correct
times as much as 500 mmol/day.
the condition that caused the abnormality. This is often
Ingestion of Acids. Rarely are large amounts of acids
difficult, especially in chronic diseases that cause
ingested in normal foods. However, severe metabolic
impaired lung function or kidney failure. In these cir-
acidosis occasionally results from the ingestion of
cumstances, various agents can be used to neutralize the
certain acidic poisons. Some of these include acetylsali-
excess acid or base in the extracellular fluid.
cylics (aspirin) and methyl alcohol (which forms formic
To neutralize excess acid, large amounts of sodium
acid when it is metabolized).
bicarbonate can be ingested by mouth. The sodium
bicarbonate is absorbed from the gastrointestinal tract
Chronic Renal Failure. When kidney function declines
into the blood and increases the bicarbonate portion of
markedly, there is a buildup of the anions of weak acids
the bicarbonate buffer system, thereby increasing pH
in the body fluids that are not being excreted by the
toward normal. Sodium bicarbonate can also be infused
kidneys. In addition, the decreased glomerular filtration
intravenously, but because of the potentially dangerous
rate reduces the excretion of phosphates and NH4+,
physiologic effects of such treatment, other substances
which reduces the amount of bicarbonate added back
are often used instead, such as sodium lactate and
to the body fluids. Thus, chronic renal failure can be
sodium gluconate. The lactate and gluconate portions of
associated with severe metabolic acidosis.
the molecules are metabolized in the body, leaving the
sodium in the extracellular fluid in the form of sodium
Metabolic Alkalosis Is Caused
bicarbonate and thereby increasing the pH of the fluid
by Increased Extracellular Fluid
toward normal.
For the treatment of alkalosis, ammonium chloride
Bicarbonate Concentration
can be administered by mouth. When the ammonium
When there is excess retention of HCO3- or loss of H+
chloride is absorbed into the blood, the ammonia
from the body, this results in metabolic alkalosis. Meta-
portion is converted by the liver into urea. This
bolic alkalosis is not nearly as common as metabolic aci-
reaction liberates HCl, which immediately reacts with
dosis, but some of the causes of metabolic alkalosis are
the buffers of the body fluids to shift the H+ concentra-
as follows.
tion in the acidic direction. Ammonium chloride
occasionally is infused intravenously, but NH4+ is
Administration of Diuretics
(Except the Carbonic Anhydrase
highly toxic, and this procedure can be dan-
Inhibitors). All diuretics cause increased flow of fluid
gerous. Another substance used occasionally is lysine
along the tubules, usually causing increased flow in the
monohydrochloride.
distal and collecting tubules. This leads to increased
reabsorption of Na+ from these parts of the nephrons.
Because the sodium reabsorption here is coupled with
Clinical Measurements and
H+ secretion, the enhanced sodium reabsorption also
Analysis of Acid-Base
leads to an increase in H+ secretion and an increase in
bicarbonate reabsorption. These changes lead to the
Disorders
development of alkalosis, characterized by increased
Appropriate therapy of acid-base disorders requires
extracellular fluid bicarbonate concentration.
proper diagnosis. The simple acid-base disorders
Excess Aldosterone. When large amounts of aldosterone
described previously can be diagnosed by analyzing
are secreted by the adrenal glands, a mild metabolic
three measurements from an arterial blood sample: pH,
alkalosis develops. As discussed previously, aldosterone
plasma bicarbonate concentration, and PCO2.
promotes extensive reabsorption of Na+ from the distal
The diagnosis of simple acid-base disorders involves
and collecting tubules and at the same time stimulates
several steps, as shown in Figure
30-10. By exam-
the secretion of H+ by the intercalated cells of the col-
ining the pH, one can determine whether the disorder
lecting tubules. This increased secretion of H+ leads to
is acidosis or alkalosis. A pH less than 7.4 indicates
its increased excretion by the kidneys and, therefore,
acidosis, whereas a pH greater than
7.4 indicates
metabolic alkalosis.
alkalosis.
Chapter 30
Regulation of Acid-Base Balance
399
Complex Acid-Base Disorders and
Arterial blood sample
Use of the Acid-Base Nomogram
for Diagnosis
<7.4
>7.4
In some instances, acid-base disorders are not accom-
pH?
panied by appropriate compensatory responses. When
this occurs, the abnormality is referred to as a mixed
acid-base disorder. This means that there are two or
Acidosis
Alkalosis
more underlying causes for the acid-base disturbance.
-
-
For example, a patient with low pH would be catego-
HCO3
Pco2
HCO3
Pco2
rized as acidotic. If the disorder was metabolically medi-
<24 mEq/L
>40 mm Hg
>24 mEq/L
<40 mm Hg
ated, this would also be accompanied by a low plasma
Metabolic
Respiratory
Metabolic
Respiratory
HCO3- concentration and, after appropriate respiratory
compensation, a low PCO2. However, if the low plasma
pH and low HCO3- concentration are associated with
elevated PCO2, one would suspect a respiratory compo-
Respiratory
Renal
Respiratory
Renal
nent to the acidosis as well as a metabolic component.
compensation
compensation
compensation
compensation
Therefore, this disorder would be categorized as a
Pco2
HCO3
-
Pco2
HCO3
-
mixed acidosis. This could occur, for example, in a
<40 mm Hg
>24 mEq/L
>40 mm Hg
<24 mEq/L
patient with acute HCO3- loss from the gastrointestinal
tract because of diarrhea (metabolic acidosis) who also
Figure 30-10
has emphysema (respiratory acidosis).
A convenient way to diagnose acid-base disorders is
Analysis of simple acid-base disorders. If the compensatory
to use an acid-base nomogram, as shown in Figure
responses are markedly different from those shown at the bottom
30-11. This diagram can be used to determine the type
of the figure, one should suspect a mixed acid-base disorder.
of acidosis or alkalosis, as well as its severity. In this
acid-base diagram, pH, HCO3- concentration, and PCO2
values intersect according to the Henderson-Hassel-
balch equation. The central open circle shows normal
values and the deviations that can still be considered
within the normal range. The shaded areas of the
The second step is to examine the plasma PCO2 and
diagram show the 95 per cent confidence limits for the
HCO3- concentration. The normal value for PCO2 is
normal compensations to simple metabolic and respira-
about 40 mm Hg, and for HCO3-, it is 24 mEq/L. If the
tory disorders.
disorder has been characterized as acidosis and the
When using this diagram, one must assume that suf-
plasma PCO2 is increased, there must be a respiratory
ficient time has elapsed for a full compensatory
component to the acidosis. After renal compensation,
response, which is 6 to 12 hours for the ventilatory com-
the plasma HCO3- concentration in respiratory acidosis
pensations in primary metabolic disorders and 3 to 5
would tend to increase above normal. Therefore, the
days for the metabolic compensations in primary respi-
expected values for a simple respiratory acidosis would
ratory disorders. If a value is within the shaded area, this
be reduced plasma pH, increased PCO2 , and increased
suggests that there is a simple acid-base disturbance.
plasma HCO3- concentration after partial renal
Conversely, if the values for pH, bicarbonate, or PCO2
compensation.
lie outside the shaded area, this suggests that there may
For metabolic acidosis, there would also be a decrease
be a mixed acid-base disorder.
in plasma pH. However, with metabolic acidosis, the
It is important to recognize that an acid-base value
-
primary abnormality is a decrease in plasma HCO3
within the shaded area does not always mean that there
concentration. Therefore, if a low pH is associated with
is a simple acid-base disorder. With this reservation in
a low HCO3- concentration, there must be a metabolic
mind, the acid-base diagrams can be used as a quick
component to the acidosis. In simple metabolic acidosis,
means of determining the specific type and severity of
the PCO2 is reduced because of partial respiratory com-
an acid-base disorder.
pensation, in contrast to respiratory acidosis, in which
For example, assume that the arterial plasma from a
PCO2 is increased. Therefore, in simple metabolic acido-
patient yields the following values: pH 7.30, plasma
sis, one would expect to find a low pH, a low plasma
HCO3- concentration 12.0 mEq/L, and plasma PCO2
HCO3- concentration, and a reduction in PCO2 after
25 mm Hg. With these values, one can look at the
partial respiratory compensation.
diagram and find that this represents a simple metabolic
The procedures for categorizing the types of alkalo-
acidosis, with appropriate respiratory compensation
sis involve the same basic steps. First, alkalosis implies
that reduces the PCO2 from its normal value of 40 mm
that there is an increase in plasma pH. If the increase in
Hg to 25 mm Hg.
pH is associated with decreased PCO2, there must be a
A second example would be a patient with the fol-
respiratory component to the alkalosis. If the rise in pH
lowing values: pH 7.15, plasma HCO3- concentration
is associated with increased HCO3-, there must be a
7 mEq/L, and plasma PCO2 50 mm Hg. In this example,
metabolic component to the alkalosis. Therefore, in
the patient is acidotic, and there appears to be a meta-
simple respiratory alkalosis, one would expect to find
bolic component because the plasma HCO3- concentra-
-
increased pH, decreased PCO2 , and decreased HCO3
tion is lower than the normal value of 24 mEq/L.
concentration in the plasma. In simple metabolic alkalo-
However, the respiratory compensation that would
sis, one would expect to find increased pH, increased
normally reduce PCO2 is absent, and PCO2 is slightly
plasma HCO3-, and increased PCO2.
increased above the normal value of 40 mm Hg. This is
400
Unit V The Body Fluids and Kidneys
60
120
100 90
80
70
60
50
40
56
110
52
35
48
Pco2 (mm Hg)
Metabolic
44
Chronic
alkalosis
30
respiratory
40
acidosis
Acute
36
25
respiratory
Figure 30-11
32
acidosis
Acid-base nomogram showing
28
20
arterial blood pH, arterial plasma
HCO3-, and PCO2
values. The
24
Normal
central open circle shows the
Acute
20
15
approximate limits for acid-base
respiratory
status in normal people. The
alkalosis
16
shaded areas in the nomogram
10
show the approximate limits
12
Metabolic
Chronic
for the normal compensations
8
acidosis
respiratory
caused by simple metabolic and
alkalosis
Pco2 (mm Hg)
respiratory disorders. For values
4
lying outside the shaded areas,
0
one should suspect a mixed acid-
7.00
7.10
7.20
7.30
7.40
7.50
7.60
7.70
7.80
base disorder.
(Adapted from
Cogan MG, Rector FC Jr: Acid-
Arterial blood pH
Base Disorders in the Kidney, 3rd
ed. Philadelphia: WB Saunders,
1986.)
consistent with a mixed acid-base disturbance con-
Table 30-4
sisting of metabolic acidosis as well as a respiratory
component.
Metabolic Acidosis Associated with Normal or Increased
The acid-base diagram serves as a quick way to assess
Plasma Anion Gap
the type and severity of disorders that may be con-
tributing to abnormal pH, PCO2 , and plasma bicarbon-
Increased Anion Gap
Normal Anion Gap
ate concentrations. In a clinical setting, the patient’s
(Normochloremia)
(Hyperchloremia)
history and other physical findings also provide impor-
tant clues concerning causes and treatment of the acid-
Diabetes mellitus (ketoacidosis)
Diarrhea
Lactic acidosis
Renal tubular acidosis
base disorders.
Chronic renal failure
Carbonic anhydrase inhibitors
Aspirin (acetylsalicylic acid)
Addison’s disease
poisoning
Use of Anion Gap to Diagnose
Methanol poisoning
Acid-Base Disorders
Ethylene glycol poisoning
Starvation
The concentrations of anions and cations in plasma
must be equal to maintain electrical neutrality. There-
fore, there is no real
“anion gap” in the plasma.
However, only certain cations and anions are routinely
dosis, the plasma HCO3- is reduced. If the plasma
measured in the clinical laboratory. The cation normally
sodium concentration is unchanged, the concentration
measured is Na+, and the anions are usually Cl- and
of anions (either Cl- or an unmeasured anion) must
HCO3-. The “anion gap” (which is only a diagnostic
increase to maintain electroneutrality. If plasma Cl-
concept) is the difference between unmeasured anions
increases in proportion to the fall in plasma HCO3-, the
and unmeasured cations, and is estimated as
anion gap will remain normal, and this is often referred
to as hyperchloremic metabolic acidosis.
Plasma anion gap = [Na+] - [HCO3-] - [Cl-]
If the decrease in plasma HCO3- is not accompanied
= 144 - 24 - 108 = 10 mEq/L
by increased Cl-, there must be increased levels of
The anion gap will increase if unmeasured anions rise
unmeasured anions and therefore an increase in the cal-
or if unmeasured cations fall. The most important
culated anion gap. Metabolic acidosis caused by excess
unmeasured cations include calcium, magnesium, and
nonvolatile acids (besides HCl), such as lactic acid or
potassium, and the major unmeasured anions are
ketoacids, is associated with an increased plasma anion
albumin, phosphate, sulfate, and other organic anions.
gap because the fall in HCO3- is not matched by an
Usually the unmeasured anions exceed the unmeasured
equal increase in Cl-. Some examples of metabolic aci-
cations, and the anion gap ranges between
8 and
dosis associated with a normal or increased anion gap
16 mEq/L.
are shown in Table 30-4. By calculating the anion gap,
The plasma anion gap is used mainly in diagnosing
one can narrow some of the potential causes of meta-
different causes of metabolic acidosis. In metabolic aci-
bolic acidosis.
Chapter 30
Regulation of Acid-Base Balance
401
References
Karet FE: Inherited distal renal tubular acidosis. J Am Soc
Nephrol 13:2178, 2002.
Laffey JG, Kavanagh BP: Hypocapnia. N Engl J Med 347:43,
Alpern RJ: Renal acidification mechanisms. In Brenner BM
2002.
(ed): The Kidney, 6th ed. Philadelphia: WB Saunders, 2000,
Lemann J Jr, Bushinsky DA, Hamm LL: Bone buffering of
pp 455-519.
acid and base in humans. Am J Physiol Renal Physiol
Capasso G, Unwin R, Rizzo M, et al: Bicarbonate transport
285:F811, 2003.
along the loop of Henle: molecular mechanisms and reg-
Madias NE, Adrogue HJ: Cross-talk between two organs:
ulation. J Nephrol 15(Suppl 5):S88, 2002.
how the kidney responds to disruption of acid-base
Decoursey TE: Voltage-gated proton channels and other
balance by the lung. Nephron Physiol 93:61, 2003.
proton transfer pathways. Physiol Rev 83:475, 2003.
Wagner CA, Geibel JP: Acid-base transport in the collecting
Gennari FJ, Maddox DA: Renal regulation of acid-base
duct. J Nephrol 15(Suppl 5):S112, 2002.
homeostasis. In Seldin DW, Giebisch G
(eds): The
Wesson DE, Alpern RJ, Seldin DW: Clinical syndromes of
Kidney—Physiology and Pathophysiology, 3rd ed. New
metabolic alkalosis. In Seldin DW, Giebisch G (eds): The
York: Raven Press, 2000, pp 2015-2054.
Kidney—Physiology and Pathophysiology, 3rd ed. New
Good DW: Ammonium transport by the thick ascending
York: Raven Press, 2000, pp 2055-2072.
limb of Henle’s loop. Ann Rev Physiol 56:623, 1994.
White NH: Management of diabetic ketoacidosis. Rev
Igarashi I, Sekine T, Inatomi J, Seki G: Unraveling the molec-
Endocr Metab Disord 4:343, 2003.
ular pathogenesis of isolated proximal renal tubular aci-
dosis. J Am Soc Nephrol 13:2171, 2002.
C H A P T E R
3
1
Kidney Diseases and Diuretics
Diuretics and Their
Mechanisms of Action
A diuretic is a substance that increases the rate of urine
volume output, as the name implies. Most diuretics also
increase urinary excretion of solutes, especially sodium
and chloride. In fact, most diuretics that are used clin-
ically act by decreasing the rate of sodium reabsorption
from the tubules, which causes natriuresis (increased
sodium output), which in turn causes diuresis (increased water output). That is, in
most cases, increased water output occurs secondary to inhibition of tubular
sodium reabsorption, because sodium remaining in the tubules acts osmotically to
decrease water reabsorption. Because the renal tubular reabsorption of many
solutes, such as potassium, chloride, magnesium, and calcium, is also influenced sec-
ondarily by sodium reabsorption, many diuretics raise renal output of these solutes
as well.
The most common clinical use of diuretics is to reduce extracellular fluid volume,
especially in diseases associated with edema and hypertension. As discussed in
Chapter 25, loss of sodium from the body mainly decreases extracellular fluid
volume; therefore, diuretics are most often administered in clinical conditions in
which extracellular fluid volume is expanded.
Some diuretics can increase urine output more than 20-fold within a few minutes
after they are administered. However, the effect of most diuretics on renal output
of salt and water subsides within a few days (Figure 31-1). This is due to activation
of other compensatory mechanisms initiated by decreased extracellular fluid
volume. For example, a decrease in extracellular fluid volume often reduces arterial
pressure and glomerular filtration rate (GFR) and increases renin secretion and
angiotensin II formation; all these responses, together, eventually override the
chronic effects of the diuretic on urine output. Thus, in the steady state, urine output
becomes equal to intake, but only after reductions in arterial pressure and extra-
cellular fluid volume have occurred, relieving the hypertension or edema that
prompted the use of diuretics in the first place.
The many diuretics available for clinical use have different mechanisms of action
and, therefore, inhibit tubular reabsorption at different sites along the renal
nephron. The general classes of diuretics and their mechanisms of action are shown
in Table 31-1.
Osmotic Diuretics Decrease Water Reabsorption
by Increasing Osmotic Pressure of Tubular Fluid
Injection into the blood stream of substances that are not easily reabsorbed by the
renal tubules, such as urea, mannitol, and sucrose, causes a marked increase in the
concentration of osmotically active molecules in the tubules. The osmotic pressure
of these solutes then greatly reduces water reabsorption, flushing large amounts of
tubular fluid into the urine.
Large volumes of urine are also formed in certain diseases associated with excess
solutes that fail to be reabsorbed from the tubular fluid. For example, when the
blood glucose concentration rises to high levels in diabetes mellitus, the increased
filtered load of glucose into the tubules exceeds their capacity to reabsorb glucose
(i.e., exceeds their transport maximum for glucose). Above a plasma glucose con-
centration of about 250 mg/dl, little of the extra glucose is reabsorbed by the tubules;
instead, the excess glucose remains in the tubules, acts as an osmotic diuretic, and
causes rapid loss of fluid into the urine. In patients with diabetes mellitus, the high
402
Chapter 31
Kidney Diseases and Diuretics
403
urine output is balanced by a high level of fluid intake
owing to activation of the thirst mechanism.
Diuretic therapy
“Loop” Diuretics Decrease Active
Sodium-Chloride-Potassium
200
Reabsorption in the Thick
Excretion
Ascending Loop of Henle
Furosemide, ethacrynic acid, and bumetanide are power-
100
ful diuretics that decrease active reabsorption in the
thick ascending limb of the loop of Henle by blocking
Intake
the
1-sodium, 2-chloride, 1-potassium co-transporter
located in the luminal membrane of the epithelial cells.
These diuretics are among the most powerful of the clin-
ically used diuretics.
By blocking active sodium-chloride-potassium co-
15.0
transport in the luminal membrane of the loop of Henle,
the loop diuretics raise urine output of sodium, chloride,
14.0
potassium, and other electrolytes, as well as water, for
two reasons: (1) they greatly increase the quantities of
solutes delivered to the distal parts of the nephrons, and
13.0
these act as osmotic agents to prevent water reabsorp-
tion as well; and (2) they disrupt the countercurrent
multiplier system by decreasing absorption of ions from
the loop of Henle into the medullary interstitium,
-4
-2
0
2
4
6
8
thereby decreasing the osmolarity of the medullary
Time (days)
interstitial fluid. Because of this effect, loop diuretics
impair the ability of the kidneys to either concentrate
or dilute the urine. Urinary dilution is impaired because
the inhibition of sodium and chloride reabsorption in
Figure 31-1
the loop of Henle causes more of these ions to be
excreted along with increased water excretion. Urinary
Sodium excretion and extracellular fluid volume during diuretic
concentration is impaired because the renal medullary
administration. The immediate increase in sodium excretion is
interstitial fluid concentration of these ions, and there-
accompanied by a decrease in extracellular fluid volume. If
fore renal medullary osmolarity, is reduced. Conse-
sodium intake is held constant, compensatory mechanisms will
quently, reabsorption of fluid from the collecting ducts
eventually return sodium excretion to equal sodium intake, thus
is decreased, so that the maximal concentrating ability
re-establishing sodium balance.
of the kidneys is also greatly reduced. In addition,
decreased renal medullary interstitial fluid osmolarity
reduces absorption of water from the descending loop
of Henle. Because of these multiple effects, 20 to 30 per
cent of the glomerular filtrate may be delivered into
the urine, causing, under acute conditions, urine output
to be as great as 25 times normal for at least a few
minutes.
Table 31-1
Classes of Diuretics, Their Mechanisms of Action, and Tubular Sites of Action
Class of Diuretic
Mechanism of Action
Tubular Site of Action
Osmotic diuretics (mannitol)
Inhibit water and solute reabsorption by increasing
Mainly proximal tubules
osmolarity of tubular fluid
Loop diuretics (furosemide, bumetanide)
Inhibit Na+-K+-Cl- co-transport in luminal membrane
Thick ascending loop of Henle
Thiazide diuretics (hydrochlorothiazide,
Inhibit Na+-Cl- co-transport in luminal membrane
Early distal tubules
chlorthalidone)
Carbonic anhydrase inhibitors
Inhibit H+ secretion and HCO3- reabsorption, which
Proximal tubules
(acetazolamide)
reduces Na+ reabsorption
Aldosterone antagonists
Inhibit action of aldosterone on tubular receptor,
Collecting tubules
(spironolactone, eplerenone)
decrease Na+ reabsorption, and decrease
K+ secretion
Sodium channel blockers
Block entry of Na+ into Na+ channels of luminal
Collecting tubules
(triamterene, amiloride)
membrane, decrease Na+ reabsorption, and
decrease K+ secretion
404
Unit V The Body Fluids and Kidneys
Thiazide Diuretics Inhibit Sodium-
Diuretics That Block Sodium
Chloride Reabsorption in the Early
Channels in the Collecting Tubules
Distal Tubule
Decrease Sodium Reabsorption
The thiazide derivatives, such as chlorothiazide, act
Amiloride and triamterene also inhibit sodium reab-
mainly on the early distal tubules to block the sodium-
sorption and potassium secretion in the collecting
chloride co-transporter in the luminal membrane of the
tubules, similar to the effects of spironolactone.
tubular cells. Under favorable conditions, these agents
However, at the cellular level, these drugs act directly
cause 5 to 10 per cent of the glomerular filtrate to pass
to block the entry of sodium into the sodium channels
into the urine. This is about the same amount of sodium
of the luminal membrane of the collecting tubule
normally reabsorbed by the distal tubules.
epithelial cells. Because of this decreased sodium entry
into the epithelial cells, there is also decreased sodium
transport across the cells’ basolateral membranes and,
therefore, decreased activity of the sodium-potassium-
Carbonic Anhydrase Inhibitors
adenosine triphosphatase pump. This decreased activity
Block Sodium-Bicarbonate
reduces the transport of potassium into the cells and
ultimately decreases the secretion of potassium into the
Reabsorption in the Proximal
tubular fluid. For this reason, the sodium channel block-
Tubules
ers are also potassium-sparing diuretics and decrease
the urinary excretion rate of potassium.
Acetazolamide inhibits the enzyme carbonic anhydrase,
which is critical for the reabsorption of bicarbonate in
the proximal tubule, as discussed in Chapter 30. Car-
Kidney Diseases
bonic anhydrase is abundant in the proximal tubule, the
primary site of action of carbonic anhydrase inhibitors.
Some carbonic anhydrase is also present in other
Diseases of the kidneys are among the most important
tubular cells, such as in the intercalated cells of the col-
causes of death and disability in many countries
lecting tubule.
throughout the world. For example, in 2004, more than
Because hydrogen ion secretion and bicarbonate
20 million adults in the United States were estimated
reabsorption in the proximal tubules are coupled to
to have chronic kidney disease.
sodium reabsorption through the sodium-hydrogen ion
Severe kidney diseases can be divided into two main
counter-transport mechanism in the luminal membrane,
categories: (1) acute renal failure, in which the kidneys
decreasing bicarbonate reabsorption also reduces
abruptly stop working entirely or almost entirely but
sodium reabsorption. The blockage of sodium and
may eventually recover nearly normal function, and
bicarbonate reabsorption from the tubular fluid causes
these ions to remain in the tubules and act as an osmotic
(2) chronic renal failure, in which there is progressive
diuretic. Predictably, a disadvantage of the carbonic
loss of function of more and more nephrons that grad-
anhydrase inhibitors is that they cause some degree of
ually decreases overall kidney function. Within these
acidosis because of the excessive loss of bicarbonate
two general categories, there are many specific kidney
ions in the urine.
diseases that can affect the kidney blood vessels,
glomeruli, tubules, renal interstitium, and parts of the
urinary tract outside the kidney, including the ureters
and bladder. In this chapter, we discuss specific physi-
Competitive Inhibitors of
ologic abnormalities that occur in a few of the more
Aldosterone Decrease Sodium
important types of kidney diseases.
Reabsorption from and Potassium
Secretion into the Cortical
Collecting Tubule
Acute Renal Failure
Spironolactone and eplerenone are aldosterone antago-
The causes of acute renal failure can be divided into
nists that compete with aldosterone for receptor sites
three main categories:
in the cortical collecting tubule epithelial cells and,
1. Acute renal failure resulting from decreased
therefore, can decrease the reabsorption of sodium and
secretion of potassium in this tubular segment. As a con-
blood supply to the kidneys; this condition is
sequence, sodium remains in the tubules and acts as an
often referred to as prerenal acute renal failure
osmotic diuretic, causing increased excretion of water as
to reflect the fact that the abnormality occurs
well as sodium. Because these drugs also block the
in a system before the kidneys. This can be a
effect of aldosterone to promote potassium secretion in
consequence of heart failure with reduced cardiac
the tubules, they decrease the excretion of potassium.
output and low blood pressure or conditions
Aldosterone antagonists also cause movement of potas-
associated with diminished blood volume and low
sium from the cells to the extracellular fluid. In some
blood pressure, such as severe hemorrhage.
instances, this causes extracellular fluid potassium con-
2. Intrarenal acute renal failure resulting from
centration to increase excessively. For this reason,
abnormalities within the kidney itself, including
spironolactone and other aldosterone inhibitors are
referred to as potassium-sparing diuretics. Many of the
those that affect the blood vessels, glomeruli, or
other diuretics cause loss of potassium in the urine, in
tubules.
contrast to the aldosterone antagonists, which “spare”
3. Postrenal acute renal failure, resulting from
the loss of potassium.
obstruction of the urinary collecting system
Chapter 31
Kidney Diseases and Diuretics
405
anywhere from the calyces to the outflow from
Table 31-2
the bladder. The most common causes of
Some Causes of Prerenal Acute Renal Failure
obstruction of the urinary tract outside the kidney
are kidney stones, caused by precipitation of
calcium, urate, or cystine.
Intravascular volume depletion
Hemorrhage (trauma, surgery, postpartum, gastrointestinal)
Diarrhea or vomiting
Prerenal Acute Renal Failure
Burns
Cardiac failure
Caused by Decreased Blood Flow
Myocardial infarction
to the Kidney
Valvular damage
Peripheral vasodilation and resultant hypotension
The kidneys normally receive an abundant blood supply
Anaphylactic shock
of about 1100 ml/min, or about 20 to 25 per cent of the
Anesthesia
cardiac output. The main purpose of this high blood flow
Sepsis, severe infections
to the kidneys is to provide enough plasma for the high
Primary renal hemodynamic abnormalities
rates of glomerular filtration needed for effective regu-
Renal artery stenosis, embolism, or thrombosis of renal artery
or vein
lation of body fluid volumes and solute concentrations.
Therefore, decreased renal blood flow is usually accom-
panied by decreased GFR and decreased urine output
of water and solutes. Consequently, conditions that
Table 31-3
acutely diminish blood flow to the kidneys usually cause
oliguria, which refers to diminished urine output below
Some Causes of Intrarenal Acute Renal Failure
the level of intake of water and solutes. This causes accu-
mulation of water and solutes in the body fluids. If renal
Small vessel and/or glomerular injury
blood flow is markedly reduced, total cessation of urine
Vasculitis (polyarteritis nodosa)
output can occur, a condition referred to as anuria.
Cholesterol emboli
As long as renal blood flow does not fall below about
Malignant hypertension
20 to 25 per cent of normal, acute renal failure can
Acute glomerulonephritis
usually be reversed if the cause of the ischemia is cor-
Tubular epithelial injury (tubular necrosis)
rected before damage to the renal cells has occurred.
Acute tubular necrosis due to ischemia
Acute tubular necrosis due to toxins (heavy metals, ethylene
Unlike some tissues, the kidney can endure a relatively
glycol, insecticides, poison mushrooms, carbon tetrachloride)
large reduction in blood flow before actual damage to
Renal interstitial injury
the renal cells occurs. The reason for this is that as renal
Acute pyelonephritis
blood flow is reduced, the GFR and the amount of
Acute allergic interstitial nephritis
sodium chloride filtered by the glomeruli (as well as
the filtration rate of water and other electrolytes) are
reduced. This decreases the amount of sodium chloride
that must be reabsorbed by the tubules, which uses most
acute renal failure can be further divided into
(1)
of the energy and oxygen consumed by the normal
conditions that injure the glomerular capillaries or
kidney. Therefore, as renal blood flow and GFR fall,
other small renal vessels, (2) conditions that damage the
the requirement for renal oxygen consumption is also
renal tubular epithelium, and (3) conditions that cause
reduced. As the GFR approaches zero, oxygen con-
damage to the renal interstitium. This type of classifica-
sumption of the kidney approaches the rate that is
tion refers to the primary site of injury, but because the
required to keep the renal tubular cells alive even when
renal vasculature and tubular system are functionally
they are not reabsorbing sodium. When blood flow is
interdependent, damage to the renal blood vessels can
reduced below this basal requirement, which is usually
lead to tubular damage, and primary tubular damage
less than 20 to 25 per cent of the normal renal blood
can lead to damage of the renal blood vessels. Causes
flow, the renal cells start to become hypoxic, and further
of intrarenal acute renal failure are listed in Table 31-3.
decreases in renal blood flow, if prolonged, will cause
damage or even death of the renal cells, especially the
Acute Renal Failure Caused by Glomerulonephritis. Acute
tubular epithelial cells. If the cause of prerenal acute
glomerulonephritis is a type of intrarenal acute renal
renal failure is not corrected and ischemia of the kidney
failure usually caused by an abnormal immune reaction
persists longer than a few hours, this type of renal failure
that damages the glomeruli. In about 95 per cent of the
can evolve into intrarenal acute renal failure, as dis-
patients with this disease, damage to the glomeruli
cussed later. Acute reduction of renal blood flow is a
occurs 1 to 3 weeks after an infection elsewhere in the
common cause of acute renal failure in hospitalized
body, usually caused by certain types of group A beta
patients. Table 31-2 shows some of the common causes
streptococci. The infection may have been a streptococ-
of decreased renal blood flow and prerenal acute renal
cal sore throat, streptococcal tonsillitis, or even strepto-
failure.
coccal infection of the skin. It is not the infection itself
that damages the kidneys. Instead, over a few weeks, as
antibodies develop against the streptococcal antigen,
Intrarenal Acute Renal Failure
the antibodies and antigen react with each other to form
Caused by Abnormalities
an insoluble immune complex that becomes entrapped
Within the Kidney
in the glomeruli, especially in the basement membrane
portion of the glomeruli.
Abnormalities that originate within the kidney and that
Once the immune complex has deposited in the
abruptly diminish urine output fall into the general cat-
glomeruli, many of the cells of the glomeruli begin to
egory of intrarenal acute renal failure. This category of
proliferate, but mainly the mesangial cells that lie
406
Unit V The Body Fluids and Kidneys
between the endothelium and the epithelium. In addi-
acute renal failure even when the kidneys’ blood supply
tion, large numbers of white blood cells become
and other functions are initially normal. If the urine
entrapped in the glomeruli. Many of the glomeruli
output of only one kidney is diminished, no major
become blocked by this inflammatory reaction, and
change in body fluid composition will occur because the
those that are not blocked usually become excessively
contralateral kidney can increase its urine output suffi-
permeable, allowing both protein and red blood cells to
ciently to maintain relatively normal levels of extracel-
leak from the blood of the glomerular capillaries into
lular electrolytes and solutes as well as normal
the glomerular filtrate. In severe cases, either total or
extracellular fluid volume. With this type of renal
almost complete renal shutdown occurs.
failure, normal kidney function can be restored if the
The acute inflammation of the glomeruli usually sub-
basic cause of the problem is corrected within a few
sides in about 2 weeks, and in most patients, the kidneys
hours. But chronic obstruction of the urinary tract,
return to almost normal function within the next few
lasting for several days or weeks, can lead to irreversible
weeks to few months. Sometimes, however, many of the
kidney damage. Some of the causes of postrenal acute
glomeruli are destroyed beyond repair, and in a small
failure include (1) bilateral obstruction of the ureters or
percentage of patients, progressive renal deterioration
renal pelvises caused by large stones or blood clots, (2)
continues indefinitely, leading to chronic renal failure, as
bladder obstruction, and (3) obstruction of the urethra.
described in a subsequent section of this chapter.
Tubular Necrosis as a Cause of Acute Renal Failure. Another
cause of intrarenal acute renal failure is tubular necro-
Physiologic Effects of Acute
sis, which means destruction of epithelial cells in the
tubules. Some common causes of tubular necrosis are
Renal Failure
(1) severe ischemia and inadequate supply of oxygen
and nutrients to the tubular epithelial cells and (2)
A major physiologic effect of acute renal failure is
poisons, toxins, or medications that destroy the tubular
retention in the blood and extracellular fluid of water,
epithelial cells.
waste products of metabolism, and electrolytes. This
Acute Tubular Necrosis Caused by Severe Renal
can lead to water and salt overload, which in turn can
Ischemia. Severe ischemia of the kidney can result from
lead to edema and hypertension. Excessive retention
circulatory shock or any other disturbance that severely
of potassium, however, is often a more serious threat
impairs the blood supply to the kidney. If the ischemia
to patients with acute renal failure, because increases
is severe enough to seriously impair the delivery of
in plasma potassium concentration (hyperkalemia) to
nutrients and oxygen to the renal tubular epithelial
more than about 8 mEq/L (only twice normal) can be
cells, and if the insult is prolonged, damage or eventual
fatal. Because the kidneys are also unable to excrete
destruction of the epithelial cells can occur. When this
sufficient hydrogen ions, patients with acute renal
happens, tubular cells “slough off ” and plug many of the
nephrons, so that there is no urine output from the
failure develop metabolic acidosis, which in itself can
blocked nephrons; the affected nephrons often fail to
be lethal or can aggravate the hyperkalemia.
excrete urine even when renal blood flow is restored to
In the most severe cases of acute renal failure, com-
normal, as long as the tubules remain plugged. The most
plete anuria occurs. The patient will die in 8 to 14 days
common causes of ischemic damage to the tubular
unless kidney function is restored or unless an artifi-
epithelium are the prerenal causes of acute renal failure
cial kidney is used to rid the body of the excessive
associated with circulatory shock, as discussed earlier in
retained water, electrolytes, and waste products of
this chapter.
metabolism. Other effects of diminished urine output,
Acute Tubular Necrosis Caused by Toxins or Medications.
as well as treatment with an artificial kidney, are dis-
There is a long list of renal poisons and medications that
cussed in the next section in relation to chronic renal
can damage the tubular epithelium and cause acute renal
failure.
failure. Some of these are carbon tetrachloride, heavy
metals (such as mercury and lead), ethylene glycol (which
is a major component in antifreeze), various insecticides,
various medications
(such as tetracyclines) used as
Chronic Renal Failure:
antibiotics, and cis-platinum, which is used in treating
An Irreversible Decrease
certain cancers. Each of these substances has a specific
toxic action on the renal tubular epithelial cells, causing
in the Number of Functional
death of many of them. As a result, the epithelial cells
Nephrons
slough away from the basement membrane and plug the
tubules. In some instances, the basement membrane also
Chronic renal failure results from progressive and irre-
is destroyed. If the basement membrane remains intact,
new tubular epithelial cells can grow along the surface
versible loss of large numbers of functioning nephrons.
of the membrane, so that the tubule repairs itself within
Serious clinical symptoms often do not occur until the
10 to 20 days.
number of functional nephrons falls to at least 70 to
75 per cent below normal. In fact, relatively normal
blood concentrations of most electrolytes and normal
Postrenal Acute Renal Failure
body fluid volumes can still be maintained until the
Caused by Abnormalities of the
number of functioning nephrons decreases below 20
Lower Urinary Tract
to 25 per cent of normal.
Multiple abnormalities in the lower urinary tract can
Table 31-4 gives some of the most important causes
block or partially block urine flow and therefore lead to
of chronic renal failure. In general, chronic renal
Chapter 31
Kidney Diseases and Diuretics
407
Table 31-4
Primary
kidney disease
Some Causes of Chronic Renal Failure
Metabolic disorders
+
Nephron
Diabetes mellitus
number
Obesity
Amyloidosis
Hypertension
Renal vascular disorders
Atherosclerosis
Nephrosclerosis-hypertension
Immunologic disorders
Glomerulonephritis
Hypertrophy
Polyarteritis nodosa
Glomerular
and vasodilation
Lupus erythematosus
sclerosis
of surviving
Infections
nephrons
Pyelonephritis
Tuberculosis
Arterial
Primary tubular disorders
pressure
Nephrotoxins (analgesics, heavy metals)
Urinary tract obstruction
Renal calculi
Glomerular
Hypertrophy of prostate
pressure
Urethral constriction
and/or
Congenital disorders
filtration
Polycystic disease
Congenital absence of kidney tissue (renal hypoplasia)
Figure 31-2
Vicious circle that can occur with primary kidney disease. Loss of
nephrons because of disease may increase pressure and flow in
the surviving glomerular capillaries, which in turn may eventually
failure, like acute renal failure, can occur because of
injure these “normal” capillaries as well, thus causing progressive
disorders of the blood vessels, glomeruli, tubules, renal
sclerosis and eventual loss of these glomeruli.
interstitium, and lower urinary tract. Despite the wide
variety of diseases that can lead to chronic renal
failure, the end result is essentially the same—a
The cause of this additional injury is not known, but
decrease in the number of functional nephrons.
some investigators believe that it may be related in
part to increased pressure or stretch of the remaining
glomeruli, which occurs as a result of functional
Vicious Circle of Chronic Renal
vasodilation or increased blood pressure; the chronic
Failure Leading to End-Stage
increase in pressure and stretch of the small arterioles
Renal Disease
and glomeruli are believed to cause sclerosis of these
vessels (replacement of normal tissue with connective
In many cases, an initial insult to the kidney leads to
tissue). These sclerotic lesions can eventually obliter-
progressive deterioration of kidney function and
ate the glomerulus, leading to further reduction in
further loss of nephrons to the point where the person
kidney function, further adaptive changes in the
must be placed on dialysis treatment or transplanted
remaining nephrons, and a slowly progressing vicious
with a functional kidney to survive. This condition is
circle that eventually terminates in end-stage renal
referred to as end-stage renal disease.
disease
(Figure
31-2). The only proven method
Studies in laboratory animals have shown that sur-
of slowing down this progressive loss of kidney func-
gical removal of large portions of the kidney initially
tion is to lower arterial pressure and glomerular
causes adaptive changes in the remaining nephrons
hydrostatic pressure, especially by using drugs such
that lead to increased blood flow, increased GFR, and
as angiotensin-converting enzyme inhibitors or
increased urine output in the surviving nephrons. The
angiotensin II antagonists.
exact mechanisms responsible for these changes are
Table 31-5 gives the most common causes of end-
not well understood but involve hypertrophy (growth
stage renal disease. In the early
1980s, glomeru-
of the various structures of the surviving nephrons) as
lonephritis in all its various forms was believed to be
well as functional changes that decrease vascular
the most common initiating cause of end-stage renal
resistance and tubular reabsorption in the surviving
disease. In recent years, diabetes mellitus and hyper-
nephrons. These adaptive changes permit a person to
tension have become recognized as the leading causes
excrete normal amounts of water and solutes even
of end-stage renal disease, together accounting for
when kidney mass is reduced to 20 to 25 per cent of
approximately 70 per cent of all chronic renal failure.
normal. Over a period of several years, however, the
Excessive weight gain (obesity) appears to be the
renal functional changes may lead to further injury of
most important risk factor for the two main causes of
the remaining nephrons, particularly to the glomeruli
end-stage renal disease—diabetes and hypertension.
of these nephrons.
As discussed in Chapter 78, type II diabetes, which is
408
Unit V The Body Fluids and Kidneys
Table 31-5
Most Common Causes of End-Stage Renal Disease (ESRD)
2.5
Cause
Percentage of Total ESRD Patients
2.0
Diabetes mellitus
44
Hypertension
26
1.5
Glomerulonephritis
8
Polycystic kidney disease
2
Other/unknown
20
1.0
0.5
closely linked to obesity, accounts for approximately
0.0
90 per cent of all diabetes mellitus. Excess weight gain
0
20
40
60
80
is also a major cause of essential hypertension,
Age (years)
accounting for as much as 65 to 75 per cent of the risk
for developing hypertension in adults. In addition to
causing renal injury through diabetes and hyperten-
sion, obesity may have additive or synergistic effects
Figure 31-3
to worsen renal function in patients with pre-existing
kidney disease.
Effect of aging on the number of functional glomeruli.
Injury to the Renal Vasculature as a
in both renal blood flow and GFR. Even in “normal”
Cause of Chronic Renal Failure
people, kidney plasma flow and GFR decrease by 40 to
50 per cent by age 80.
Many types of vascular lesions can lead to renal
The frequency and severity of nephrosclerosis and
ischemia and death of kidney tissue. The most common
glomerulosclerosis are greatly increased by concurrent
of these are (1) atherosclerosis of the larger renal arter-
hypertension or diabetes mellitus. In fact, diabetes mel-
ies, with progressive sclerotic constriction of the vessels;
litus and hypertension are the two most important
(2) fibromuscular hyperplasia of one or more of the
causes of end-stage renal disease, as discussed previ-
large arteries, which also causes occlusion of the vessels;
ously. Thus, benign nephrosclerosis in association with
and (3) nephrosclerosis, caused by sclerotic lesions of
severe hypertension can lead to a rapidly progressing
the smaller arteries, arterioles, and glomeruli.
malignant nephrosclerosis. The characteristic histologi-
Atherosclerotic or hyperplastic lesions of the large
cal features of malignant nephrosclerosis include large
arteries frequently affect one kidney more than the
amounts of fibrinoid deposits in the arterioles and pro-
other and, therefore, cause unilaterally diminished
gressive thickening of the vessels, with severe ischemia
kidney function. As discussed in Chapter 19, hyperten-
occurring in the affected nephrons. For unknown
sion often occurs when the artery of one kidney is
reasons, the incidence of malignant nephrosclerosis and
constricted while the artery of the other kidney is
severe glomerulosclerosis is significantly higher in
still normal, a condition analogous to “two-kidney”
blacks than in whites of similar ages who have similar
Goldblatt hypertension.
degrees of severity of hypertension or diabetes.
Benign nephrosclerosis, the most common form of
kidney disease, is seen to at least some extent in about
70 per cent of postmortem examinations in people who
Injury to the Glomeruli as a Cause
die after the age of 60. This type of vascular lesion
of Chronic Renal Failure—
occurs in the smaller interlobular arteries and in the
Glomerulonephritis
afferent arterioles of the kidney. It is believed to begin
with leakage of plasma through the intimal membrane
Chronic glomerulonephritis can be caused by several
of these vessels. This causes fibrinoid deposits to
diseases that cause inflammation and damage to the
develop in the medial layers of these vessels, followed
capillary loops in the glomeruli of the kidneys. In con-
by progressive thickening of the vessel wall that even-
trast to the acute form of this disease, chronic glomeru-
tually constricts the vessels and, in some cases, occludes
lonephritis is a slowly progressive disease that often
them. Because there is essentially no collateral circula-
leads to irreversible renal failure. It may be a primary
tion among the smaller renal arteries, occlusion of one
kidney disease, following acute glomerulonephritis, or it
or more of them causes destruction of a comparable
may be secondary to systemic diseases, such as lupus
number of nephrons. Therefore, much of the kidney
erythematosus.
tissue becomes replaced by small amounts of fibrous
In most cases, chronic glomerulonephritis begins with
tissue. When sclerosis occurs in the glomeruli, the injury
accumulation of precipitated antigen-antibody com-
is referred to as glomerulosclerosis.
plexes in the glomerular membrane. In contrast to acute
Nephrosclerosis and glomerulosclerosis occur to
glomerulonephritis, streptococcal infections account for
some extent in most people after the fourth decade of
only a small percentage of patients with the chronic
life, causing about a 10 per cent decrease in the number
form of glomerulonephritis. The results of the accumu-
of functional nephrons each
10 years after age
40
lation of antigen-antibody complex in the glomerular
(Figure
31-3). This loss of glomeruli and overall
membranes are inflammation, progressive thickening of
nephron function is reflected by a progressive decrease
the membranes, and eventual invasion of the glomeruli
Chapter 31
Kidney Diseases and Diuretics
409
by fibrous tissue. In the later stages of the disease, the
large quantities of plasma proteins into the urine. In
glomerular capillary filtration coefficient becomes
some instances, this occurs without evidence of other
greatly reduced because of decreased numbers of filter-
major abnormalities of kidney function, but more often
ing capillaries in the glomerular tufts and because of
it is associated with some degree of renal failure.
thickened glomerular membranes. In the final stages of
The cause of the protein loss in the urine is increased
the disease, many glomeruli are replaced by fibrous
permeability of the glomerular membrane. Therefore,
tissue and are, therefore, unable to filter fluid.
any disease that increases the permeability of this mem-
brane can cause the nephrotic syndrome. Such diseases
include (1) chronic glomerulonephritis, which affects
Injury to the Renal Interstitium as a
primarily the glomeruli and often causes greatly
Cause of Chronic Renal Failure—
increased permeability of the glomerular membrane;
Pyelonephritis
(2) amyloidosis, which results from deposition of an
abnormal proteinoid substance in the walls of the blood
Primary or secondary disease of the renal interstitium
vessels and seriously damages the basement membrane
is referred to as interstitial nephritis. In general, this can
of the glomeruli; and (3) minimal change nephrotic syn-
result from vascular, glomerular, or tubular damage that
drome, which is associated with no major abnormality
destroys individual nephrons, or it can involve primary
in the glomerular capillary membrane that can be
damage to the renal interstitium by poisons, drugs, and
detected with light microscopy. As discussed in Chapter
bacterial infections.
26, minimal change nephropathy has been found to
Renal interstitial injury caused by bacterial infec-
be associated with loss of the negative charges that are
tion is called pyelonephritis. The infection can result
normally present in the glomerular capillary basement
from different types of bacteria but especially from
membrane. Immunologic studies have also shown
Escherichia coli that originate from fecal contamination
abnormal immune reactions in some cases, suggesting
of the urinary tract. These bacteria reach the kidneys
that the loss of the negative charges may have resulted
either by way of the blood stream or, more commonly,
from antibody attack on the membrane. Loss of normal
by ascension from the lower urinary tract by way of the
negative charges in the basement membrane of the
ureters to the kidneys.
glomerular capillaries allows proteins, especially
Although the normal bladder is able to clear bacteria
albumin, to pass through the glomerular membrane
readily, there are two general clinical conditions that
with ease because the negative charges in the basement
may interfere with the normal flushing of bacteria from
membrane normally repel the negatively charged
the bladder: (1) the inability of the bladder to empty
plasma proteins.
completely, leaving residual urine in the bladder, and
Minimal change nephropathy can occur in adults, but
(2) the existence of obstruction of urine outflow. With
more frequently it occurs in children between the ages
impaired ability to flush bacteria from the bladder, the
of 2 and 6 years. Increased permeability of the glomeru-
bacteria multiply and the bladder becomes inflamed, a
lar capillary membrane occasionally allows as much as
condition termed cystitis. Once cystitis has occurred, it
40 grams of plasma protein loss into the urine each day,
may remain localized without ascending to the kidney,
which is an extreme amount for a young child. There-
or in some people, bacteria may reach the renal pelvis
fore, the child’s plasma protein concentration often falls
because of a pathological condition in which urine is
below 2 g/dl, and the colloid osmotic pressure falls from
propelled up one or both of the ureters during micturi-
a normal value of 28 to less than 10 mm Hg. As a con-
tion. This condition is called vesicoureteral reflux and is
sequence of this low colloid osmotic pressure in the
due to the failure of the bladder wall to occlude the
plasma, large amounts of fluid leak from the capillaries
ureter during micturition; as a result, some of the urine
all over the body into most of the tissues, causing severe
is propelled upward toward the kidney, carrying with it
edema, as discussed in Chapter 25.
bacteria that can reach the renal pelvis and renal
medulla, where they can initiate the infection and
inflammation associated with pyelonephritis.
Nephron Function in Chronic
Pyelonephritis begins in the renal medulla and there-
Renal Failure
fore usually affects the function of the medulla more
than it affects the cortex, at least in the initial stages.
Because one of the primary functions of the medulla is
Loss of Functional Nephrons Requires the Surviving Nephrons
to provide the countercurrent mechanism for concen-
to Excrete More Water and Solutes. It would be reasonable
trating urine, patients with pyelonephritis frequently
to suspect that decreasing the number of functional
have markedly impaired ability to concentrate the urine.
nephrons, which reduces the GFR, would also cause
With long-standing pyelonephritis, invasion of the
major decreases in renal excretion of water and
kidneys by bacteria not only causes damage to the renal
solutes. Yet patients who have lost as much as 75 per
medulla interstitium but also results in progressive
cent of their nephrons are able to excrete normal
damage of renal tubules, glomeruli, and other structures
amounts of water and electrolytes without serious
throughout the kidney. Consequently, large parts of
accumulation of any of these in the body fluids.
functional renal tissue are lost, and chronic renal failure
can develop.
Further reduction in the number of nephrons,
however, leads to electrolyte and fluid retention, and
death usually ensues when the number of nephrons
Nephrotic Syndrome—Excretion
falls below 5 to 10 per cent of normal.
of Protein in the Urine Because of
In contrast to the electrolytes, many of the waste
Increased Glomerular Permeability
products of metabolism, such as urea and creatinine,
Many patients with kidney disease develop the
accumulate almost in proportion to the number of
nephrotic syndrome, which is characterized by loss of
nephrons that have been destroyed. The reason for this
410
Unit V The Body Fluids and Kidneys
100
50
A
0
2
B
1
C
0
0
25
50
75
100
Glomerular filtration rate
(percentage of normal)
Positive balance
Production
2
Figure 31-5
Excretion GFR x PCreatinine
1
Representative patterns of adaptation for different types of solutes
in chronic renal failure. Curve A shows the approximate changes
in the plasma concentrations of solutes such as creatinine and
urea that are filtered and poorly reabsorbed. Curve B shows the
0
approximate concentrations for solutes such as phosphate and
0
1
2
3
4
urate. Curve C shows the approximate concentrations for solutes
Days
such as sodium and chloride.
Some solutes, such as phosphate, urate, and hydro-
Figure 31-4
gen ions, are often maintained near the normal range
Effect of reducing glomerular filtration rate (GFR) by 50 per cent
until GFR falls below 20 to 30 per cent of normal.
on serum creatinine concentration and on creatinine excretion rate
Thereafter, the plasma concentrations of these sub-
when the production rate of creatinine remains constant.
stances rise, but not in proportion to the fall in GFR,
as shown in curve B of Figure 31-5. Maintenance of
is that substances such as creatinine and urea depend
relatively constant plasma concentrations of these
largely on glomerular filtration for their excretion, and
solutes as GFR declines is accomplished by excreting
they are not reabsorbed as avidly as the electrolytes.
progressively larger fractions of the amounts of these
Creatinine, for example, is not reabsorbed at all, and
solutes that are filtered at the glomerular capillaries;
the excretion rate is equal to the rate at which it is
this occurs by decreasing the rate of tubular reab-
filtered.
sorption or, in some instances, by increasing tubular
secretion rates.
Creatinine filtration rate
In the case of sodium and chloride ions, their plasma
= GFR ¥ Plasma creatinine concentration
concentrations are maintained virtually constant
= Creatinine excretion rate
even with severe decreases in GFR (see curve C of
Therefore, if GFR decreases, the creatinine excre-
Figure
31-5). This is accomplished by greatly de-
tion rate also transiently decreases, causing accumula-
creasing tubular reabsorption of these electrolytes.
tion of creatinine in the body fluids and raising plasma
For example, with a 75 per cent loss of functional
concentration until the excretion rate of creatinine
nephrons, each surviving nephron must excrete four
returns to normal—the same rate at which creatinine
times as much sodium and four times as much volume
is produced in the body (Figure 31-4). Thus, under
as under normal conditions (Table 31-6).
steady-state conditions, the creatinine excretion rate
Part of this adaptation occurs because of increased
equals the rate of creatinine production, despite reduc-
blood flow and increased GFR in each of the surviv-
tions in GFR; however, this normal rate of creatinine
ing nephrons, owing to hypertrophy of the blood
excretion occurs at the expense of elevated plasma
vessels and glomeruli, as well as functional changes
creatinine concentration, as shown in curve A of
that cause the blood vessels to vasodilate. Even with
Figure 31-5.
large decreases in the total GFR, normal rates of renal
Chapter 31
Kidney Diseases and Diuretics
411
Table 31-6
Total Kidney Excretion and Excretion per Nephron in
1.050
Renal Failure
1.040
75% Loss of
Normal
Nephrons
1.030
Isosthenuria
Number of nephrons
2,000,000
500,000
Total GFR (ml/min)
125
40
Single nephron GFR (nl/min)
62.5
80
1.020
Volume excreted for all
1.5
1.5
nephrons (ml/min)
Glomerular filtrate specific gravity
1.010
Volume excreted per nephron
0.75
3.0
(nl/min)
1.000
2,000,000 1,500,000 1,000,000
500,000
0
GFR, glomerular filtration rate.
Number of nephrons in both kidneys
excretion can still be maintained by decreasing the
rate at which the tubules reabsorb water and solutes.
Figure 31-6
Isosthenuria—Inability of the Kidney to Concentrate or Dilute
Development of isosthenuria in a patient with decreased numbers
the Urine. One important effect of the rapid rate of
of functional nephrons.
tubular flow that occurs in the remaining nephrons of
diseased kidneys is that the renal tubules lose their
ability to concentrate or dilute the urine. The concen-
trating ability of the kidney is impaired mainly because
(1) the rapid flow of tubular fluid through the collect-
ing ducts prevents adequate water reabsorption, and
(2) the rapid flow through both the loop of Henle and
the collecting ducts prevents the countercurrent mech-
anism from operating effectively to concentrate the
medullary interstitial fluid solutes. Therefore, as pro-
gressively more nephrons are destroyed, the maximum
concentrating ability of the kidney declines, and urine
Normal
osmolarity and specific gravity (a measure of the total
solute concentration) approach the osmolarity and
specific gravity of the glomerular filtrate, as shown in
Figure 31-6.
The diluting mechanism in the kidney is also
Kidney shutdown
impaired when the number of nephrons decreases
0
3
6
9
12
because the rapid flushing of fluid through the loops
Days
of Henle and the high load of solutes such as urea
cause a relatively high solute concentration in the
tubular fluid of this part of the nephron. As a conse-
quence, the diluting capacity of the kidney is impaired,
Figure 31-7
and the minimal urine osmolality and specific gravity
approach those of the glomerular filtrate. Because the
Effect of kidney failure on extracellular fluid constituents. NPN,
nonprotein nitrogens.
concentrating mechanism becomes impaired to a
greater extent than does the diluting mechanism in
chronic renal failure, an important clinical test of renal
function is to determine how well the kidneys can
of different substances in the extracellular fluid are
concentrate urine when a person’s water intake is
approximately those shown in Figure 31-7. Important
restricted for 12 or more hours.
effects include (1) generalized edema resulting from
water and salt retention, (2) acidosis resulting from
failure of the kidneys to rid the body of normal acidic
Effects of Renal Failure on the Body
products, (3) high concentration of the nonprotein nitro-
Fluids—Uremia
gens—especially urea, creatinine, and uric acid—result-
ing from failure of the body to excrete the metabolic
The effect of complete renal failure on the body fluids
end products of proteins, and (4) high concentrations of
depends on (1) water and food intake and (2) the
other substances excreted by the kidney, including
degree of impairment of renal function. Assuming that
phenols, sulfates, phosphates, potassium, and guanidine
a person with complete renal failure continues to ingest
bases. This total condition is called uremia because of
the same amounts of water and food, the concentrations
the high concentration of urea in the body fluids.
412
Unit V The Body Fluids and Kidneys
Water Retention and Development of Edema in Renal Failure. If
Osteomalacia in Chronic Renal Failure Caused by Decreased
water intake is restricted immediately after acute renal
Production of Active Vitamin D and by Phosphate Retention by
failure begins, the total body fluid content may become
the Kidneys. Prolonged renal failure also causes osteo-
only slightly increased. If fluid intake is not limited and
malacia, a condition in which the bones are partially
the patient drinks in response to the normal thirst mech-
absorbed and, therefore, become greatly weakened. An
anisms, the body fluids begin to increase immediately
important cause of this condition is the following:
and rapidly.
Vitamin D must be converted by a two-stage process,
With chronic partial kidney failure, accumulation of
first in the liver and then in the kidneys, into 1,25-
fluid may not be severe, as long as salt and fluid intake
dihydroxycholecalciferol before it is able to promote
are not excessive, until kidney function falls to 25 per
calcium absorption from the intestine. Therefore,
cent of normal or lower. The reason for this, as discussed
serious damage to the kidney greatly reduces the blood
previously, is that the surviving nephrons excrete larger
concentration of active vitamin D, which in turn
amounts of salt and water. Even the small fluid reten-
decreases intestinal absorption of calcium and the avail-
tion that does occur, along with increased secretion of
ability of calcium to the bones.
renin and angiotensin II that usually occurs in ischemic
Another important cause of demineralization of the
kidney disease, often causes severe hypertension in
skeleton in chronic renal failure is the rise in serum
chronic renal failure. Almost all patients with kidney
phosphate concentration that occurs as a result of
function so reduced as to require dialysis to preserve life
decreased GFR. This rise in serum phosphate causes
develop hypertension. In most of these patients, severe
increased binding of phosphate with calcium in the
reduction of salt intake or removal of extracellular fluid
plasma, thus decreasing the plasma serum ionized
by dialysis can control the hypertension. The remaining
calcium concentration, which in turn stimulates parathy-
patients continue to have hypertension even after
roid hormone secretion. This secondary hyperparathy-
excess sodium has been removed by dialysis. In this
roidism then stimulates the release of calcium from
group, removal of the ischemic kidneys usually corrects
bones, causing further demineralization of the bones.
the hypertension (as long as fluid retention is prevented
by dialysis) because it removes the source of excessive
renin secretion and subsequent increased angiotensin II
Hypertension and Kidney Disease
formation.
As discussed earlier in this chapter, hypertension can
exacerbate injury to the glomeruli and blood vessels of
Uremia—Increase in Urea and Other Nonprotein Nitrogens
the kidneys and is a major cause of end-stage renal
(Azotemia). The nonprotein nitrogens include urea, uric
disease. Conversely, abnormalities of kidney function
acid, creatinine, and a few less important compounds.
can cause hypertension, as discussed in detail in Chapter
These, in general, are the end products of protein
19. Thus, the relation between hypertension and kidney
metabolism and must be removed from the body to
disease can, in some instances, propagate a vicious
ensure continued normal protein metabolism in the
circle: primary kidney damage leads to increased blood
cells. The concentrations of these, particularly of urea,
pressure, which in turn causes further damage to the
can rise to as high as 10 times normal during 1 to 2
kidneys, further increases in blood pressure, and so
weeks of total renal failure. With chronic renal failure,
forth, until end-stage renal disease develops.
the concentrations rise approximately in proportion to
Not all types of kidney disease cause hypertension,
the degree of reduction in functional nephrons. For this
because damage to certain portions of the kidney cause
reason, measuring the concentrations of these sub-
uremia without hypertension. Nevertheless, some types
stances, especially of urea and creatinine, provides an
of renal damage are particularly prone to cause hyper-
important means for assessing the degree of renal
tension. A classification of kidney disease relative
failure.
to hypertensive or nonhypertensive effects is the
following.
Acidosis in Renal Failure. Each day the body normally pro-
duces about 50 to 80 millimoles more metabolic acid
Renal Lesions That Reduce the Ability of the Kidneys to Excrete
than metabolic alkali. Therefore, when the kidneys fail
Sodium and Water Promote Hypertension. Renal lesions that
to function, acid accumulates in the body fluids. The
decrease the ability of the kidneys to excrete sodium
buffers of the body fluids normally can buffer 500 to
and water almost invariably cause hypertension. There-
1000 millimoles of acid without lethal increases in extra-
fore, lesions that either decrease GFR or increase
cellular fluid hydrogen ion concentration, and the phos-
tubular reabsorption usually lead to hypertension of
phate compounds in the bones can buffer an additional
varying degrees. Some specific types of renal abnormal-
few thousand millimoles of hydrogen ion. However,
ities that can cause hypertension are as follows:
when this buffering power is used up, the blood pH falls
1. Increased renal vascular resistance, which reduces
drastically, and the patient will become comatose and
renal blood flow and GFR. An example is
die if the pH falls below about 6.8.
hypertension caused by renal artery stenosis.
2. Decreased glomerular capillary filtration coefficient,
Anemia in Chronic Renal Failure Caused by Decreased Erythro-
which reduces GFR. An example of this is chronic
poietin Secretion. Patients with severe chronic renal
glomerulonephritis, which causes inflammation and
failure almost always develop anemia. The most im-
thickening of the glomerular capillary membranes,
portant cause of this is decreased renal secretion of
thereby reducing the glomerular capillary filtration
erythropoietin, which stimulates the bone marrow to
coefficient.
produce red blood cells. If the kidneys are seriously
3. Excessive tubular sodium reabsorption. An example
damaged, they are unable to form adequate quantities
is hypertension caused by excessive aldosterone
of erythropoietin, which leads to diminished red blood
secretion, which increases sodium reabsorption
cell production and consequent anemia.
mainly in the cortical collecting tubules.
Chapter 31
Kidney Diseases and Diuretics
413
Once hypertension has developed, renal excretion of
Specific Tubular Disorders
sodium and water returns to normal because the high
arterial pressure causes pressure natriuresis and pres-
In Chapter 27, we point out that several mechanisms are
sure diuresis, so that intake and output of sodium
responsible for transporting different individual sub-
and water become balanced once again. Even when
stances across the tubular epithelial membranes. In
there are large increases in renal vascular resistance or
Chapter 3, we also point out that each cellular enzyme
decreases in the glomerular capillary coefficient, the
and each carrier protein is formed in response to a
GFR may still return to nearly normal levels after the
respective gene in the nucleus. If any required gene
arterial blood pressure rises. Likewise, when tubular
happens to be absent or abnormal, the tubules may be
reabsorption is increased, as occurs with excessive
deficient in one of the appropriate carrier proteins or
aldosterone secretion, the urinary excretion rate is ini-
one of the enzymes needed for solute transport by the
tially reduced but then returns to normal as arterial
renal tubular epithelial cells. For this reason, many
pressure rises. Thus, after hypertension develops, there
hereditary tubular disorders occur because of the trans-
may be no sign of impaired excretion of sodium and
port of individual substances or groups of substances
water other than the hypertension. As explained in
through the tubular membrane. In addition, damage to
Chapter 19, normal excretion of sodium and water at an
the tubular epithelial membrane by toxins or ischemia
elevated arterial pressure means that pressure natriure-
can cause important renal tubular disorders.
sis and pressure diuresis have been reset to a higher
arterial pressure.
Renal Glycosuria—Failure of the Kidneys to Reabsorb Glucose.
In this condition, the blood glucose concentration may
Hypertension Caused by Patchy Renal Damage and Increased
be normal, but the transport mechanism for tubular
Renal Secretion of Renin. If one part of the kidney is
reabsorption of glucose is greatly limited or absent.
ischemic and the remainder is not ischemic, such as
Consequently, despite a normal blood glucose level,
occurs when one renal artery is severely constricted, the
large amounts of glucose pass into the urine each day.
ischemic renal tissue secretes large quantities of renin.
Because diabetes mellitus is also associated with the
This secretion leads to the formation of angiotensin II,
presence of glucose in the urine, renal glycosuria, which
which can cause hypertension. The most likely sequence
is a relatively benign condition, must be ruled out before
of events in causing this hypertension, as discussed in
making a diagnosis of diabetes mellitus.
Chapter 19, is
(1) the ischemic kidney tissue itself
excretes less than normal amounts of water and salt; (2)
Aminoaciduria—Failure of the Kidneys to Reabsorb Amino Acids.
the renin secreted by the ischemic kidney, and subse-
Some amino acids share mutual transport systems for
quent increased angiotensin II formation, affects the
reabsorption, whereas other amino acids have their own
nonischemic kidney tissue, causing it also to retain salt
distinct transport systems. Rarely, a condition called
and water; and (3) excess salt and water cause hyper-
generalized aminoaciduria results from deficient reab-
tension in the usual manner.
sorption of all amino acids; more frequently, deficiencies
A similar type of hypertension can result when patchy
of specific carrier systems may result in (1) essential
areas of one or both kidneys become ischemic as a
cystinuria, in which large amounts of cystine fail to be
result of arteriosclerosis or vascular injury in specific
reabsorbed and often crystallize in the urine to form
portions of the kidneys. When this occurs, the ischemic
renal stones; (2) simple glycinuria, in which glycine fails
nephrons excrete less salt and water but secrete greater
to be reabsorbed; or (3) beta-aminoisobutyricaciduria,
amounts of renin, which causes increased angiotensin
which occurs in about 5 per cent of all people but appar-
II formation. The high levels of angiotensin II then
ently has no major clinical significance.
impair the ability of the surrounding otherwise normal
nephrons to excrete sodium and water. As a result,
Renal Hypophosphatemia—Failure of the Kidneys to Reabsorb
hypertension develops, which restores the overall
Phosphate. In renal hypophosphatemia, the renal
excretion of sodium and water by the kidney, so that
tubules fail to reabsorb large enough quantities of phos-
balance between intake and output of salt and water
phate ions when the phosphate concentration of the
is maintained, but at the expense of high blood
body fluids falls very low. This condition usually does
pressure.
not cause serious immediate abnormalities, because
the phosphate concentration of the extracellular fluid
Kidney Diseases That Cause Loss of Entire Nephrons Lead to Renal
can vary widely without causing major cellular dys-
Failure But May Not Cause Hypertension. Loss of large
function. Over a long period, a low phosphate level
numbers of whole nephrons, such as occurs with the loss
causes diminished calcification of the bones, causing the
of one kidney and part of another kidney, almost always
person to develop rickets. This type of rickets is refrac-
leads to renal failure if the amount of kidney tissue lost
tory to vitamin D therapy, in contrast to the rapid
is great enough. If the remaining nephrons are normal
response of the usual type of rickets, as discussed in
and the salt intake is not excessive, this condition might
Chapter 79.
not cause clinically significant hypertension, because
even a slight rise in blood pressure will raise the GFR
and decrease tubular sodium reabsorption sufficiently
Renal Tubular Acidosis—Failure of the Tubules to Secrete Hydro-
to promote enough water and salt excretion in the urine,
gen Ions. In this condition, the renal tubules are unable
even with the few nephrons that remain intact.
to secrete adequate amounts of hydrogen ions. As a
However, a patient with this type of abnormality may
result, large amounts of sodium bicarbonate are contin-
become severely hypertensive if additional stresses are
ually lost in the urine. This causes a continued state of
imposed, such as eating a large amount of salt. In this
metabolic acidosis, as discussed in Chapter 30. This type
case, the kidneys simply cannot clear adequate quanti-
of renal abnormality can be caused by hereditary dis-
ties of salt with the small number of functioning
orders, or it can occur as a result of widespread injury
nephrons that remain.
to the renal tubules.
414
Unit V The Body Fluids and Kidneys
Nephrogenic Diabetes Insipidus—Failure of the Kidneys to
Respond to Antidiuretic Hormone. Occasionally, the renal
tubules do not respond to antidiuretic hormone, causing
large quantities of dilute urine to be excreted. As long
as the person is supplied with plenty of water, this con-
Semipermeable
Flowing
dition seldom causes severe difficulty. However, when
membrane
blood
adequate quantities of water are not available, the
person rapidly becomes dehydrated.
Fanconi’s Syndrome—A Generalized Reabsorptive Defect of the
Waste Water
Flowing
Renal Tubules. Fanconi’s syndrome is usually associated
products
dialysate
with increased urinary excretion of virtually all amino
acids, glucose, and phosphate. In severe cases, other
manifestations are also observed, such as (1) failure
to reabsorb sodium bicarbonate, which results in
metabolic acidosis; (2) increased excretion of potassium
Bubble
Blood out
and sometimes calcium; and (3) nephrogenic diabetes
trap
insipidus.
There are multiple causes of Fanconi’s syndrome,
Dialyzer
which results from a generalized inability of the renal
tubular cells to transport various substances. Some of
these causes include (1) hereditary defects in cell trans-
Blood in
port mechanisms, (2) toxins or drugs that injure the
Dialysate
Dialysate
renal tubular epithelial cells, and (3) injury to the renal
in
out
tubular cells as a result of ischemia. The proximal
tubular cells are especially affected in Fanconi’s syn-
drome caused by tubular injury, because these cells
reabsorb and secrete many of the drugs and toxins that
can cause damage.
Treatment of Renal Failure
by Dialysis with an
Artificial Kidney
Fresh dialyzing
Constant
Used dialyzing
solution
temperature
solution
Severe loss of kidney function, either acutely or chron-
bath
ically, is a threat to life and requires removal of toxic
waste products and restoration of body fluid volume
Figure 31-8
and composition toward normal. This can be accom-
Principles of dialysis with an artificial kidney.
plished by dialysis with an artificial kidney. In certain
types of acute renal failure, an artificial kidney may be
used to tide the patient over until the kidneys resume
their function. If the loss of kidney function is irre-
fluid back into the plasma. If the concentration of a sub-
versible, it is necessary to perform dialysis chronically
stance is greater in the plasma than in the dialyzing
to maintain life. In the United States alone, nearly
fluid, there will be a net transfer of the substance from
300,000 people with irreversible renal failure or even
the plasma into the dialyzing fluid.
total kidney removal are being maintained by dialysis
The rate of movement of solute across the dialyzing
with artificial kidneys. Because dialysis cannot maintain
membrane depends on (1) the concentration gradient
completely normal body fluid composition and cannot
of the solute between the two solutions, (2) the per-
replace all the multiple functions performed by the
meability of the membrane to the solute, (3) the surface
kidneys, the health of patients maintained on artificial
area of the membrane, and (4) the length of time
kidneys usually remains significantly impaired. A better
that the blood and fluid remain in contact with the
treatment for permanent loss of kidney function is to
membrane.
restore functional kidney tissue by means of a kidney
Thus, the maximum rate of solute transfer occurs ini-
transplant.
tially when the concentration gradient is greatest (when
dialysis is begun) and slows down as the concentration
Basic Principles of Dialysis. The basic principle of the arti-
gradient is dissipated. In a flowing system, as is the case
ficial kidney is to pass blood through minute blood
with “hemodialysis,” in which blood and dialysate fluid
channels bounded by a thin membrane. On the other
flow through the artificial kidney, the dissipation of the
side of the membrane is a dialyzing fluid into which
concentration gradient can be reduced and diffusion of
unwanted substances in the blood pass by diffusion.
solute across the membrane can be optimized by
Figure 31-8 shows the components of one type of arti-
increasing the flow rate of the blood, the dialyzing fluid,
ficial kidney in which blood flows continually between
or both.
two thin membranes of cellophane; outside the mem-
In normal operation of the artificial kidney, blood
brane is a dialyzing fluid. The cellophane is porous
flows continually or intermittently back into the vein.
enough to allow the constituents of the plasma, except
The total amount of blood in the artificial kidney at any
the plasma proteins, to diffuse in both directions—from
one time is usually less than 500 milliliters, the rate of
plasma into the dialyzing fluid or from the dialyzing
flow may be several hundred milliliters per minute, and
Chapter 31
Kidney Diseases and Diuretics
415
at a rate of 100 to 225 ml/min, which shows that at least
Table 31-7
for the excretion of urea, the artificial kidney can func-
Comparison of Dialyzing Fluid with Normal and Uremic
tion about twice as rapidly as two normal kidneys
Plasma
together, whose urea clearance is only 70 ml/min. Yet
the artificial kidney is used for only 4 to 6 hours per day,
three times a week. Therefore, the overall plasma clear-
Normal
Dialyzing
Uremic
Constituent
Plasma
Fluid
Plasma
ance is still considerably limited when the artificial
kidney replaces the normal kidneys. Also, it is important
Electrolytes (mEq/L)
to keep in mind that the artificial kidney cannot replace
Na+
142
133
142
some of the other functions of the kidneys, such as
K+
5
1.0
7
secretion of erythropoietin, which is necessary for red
Ca++
3
3.0
2
blood cell production.
Mg++
1.5
1.5
1.5
Cl-
107
105
107
HCO3-
24
35.7
14
Lactate-
1.2
1.2
1.2
References
HPO4=
3
0
9
Urate-
0.3
0
2
Andreoli TE (ed): Cecil’s Essentials of Medicine, 6th ed.
Sulfate=
0.5
0
3
Philadelphia: WB Saunders, 2004.
Nonelectrolytes
Glucose
100
125
100
Fishbane SA, Scribner BH: Blood pressure control in dialy-
Urea
26
0
200
sis patients. Semin Dial 15:144, 2002.
Creatinine
1
0
6
Hall JE: The kidney, hypertension, and obesity. Hypertension
41:625, 2003.
Hall JE, Henegar JR, Dwyer TM, et al: Is obesity a major
cause of chronic renal disease? Adv Ren Replace Ther
the total diffusion surface area is between 0.6 and 2.5
11:41, 2004.
square meters. To prevent coagulation of the blood in
Hostetter TH: Prevention of the development and progres-
the artificial kidney, a small amount of heparin is infused
sion of renal disease. J Am Soc Nephrol 14(Suppl 2):S144,
into the blood as it enters the artificial kidney. In addi-
2003.
tion to diffusion of solutes, mass transfer of solutes and
Levey AS, Beto JA, Coronado BE, et al: Controlling the epi-
water can be produced by applying a hydrostatic pres-
demic of cardiovascular disease in chronic renal disease.
sure to force the fluid and solutes across the membranes
What do we know? What do we need to learn? Where do
of the dialyzer; such filtration is called bulk flow.
we go from here? National Kidney Foundation Task Force
on Cardiovascular Disease. Am J Kidney Dis 32:853, 1998.
Dialyzing Fluid. Table 31-7 compares the constituents in
Luke RG: Chronic renal failure. In: Goldman F, Bennett JC
a typical dialyzing fluid with those in normal plasma and
(eds): Cecil Textbook of Medicine, 21st ed. Philadelphia:
uremic plasma. Note that the concentrations of ions and
WB Saunders, 2000, pp 571-578.
other substances in dialyzing fluid are not the same as
Mitch WE: Acute renal failure. In: Goldman F, Bennett JC
the concentrations in normal plasma or in uremic
(eds): Cecil Textbook of Medicine, 21st ed. Philadelphia:
plasma. Instead, they are adjusted to levels that are
WB Saunders, 2000, pp 567-570.
needed to cause appropriate movement of water and
Molitoris BA: Transitioning to therapy in ischemic acute
solutes through the membrane during dialysis.
renal failure. J Am Soc Nephrol 14:265, 2003.
Note that there is no phosphate, urea, urate, sulfate,
Sarnak MJ, Levey AS, Schoolwerth AC, et al: Kidney disease
or creatinine in the dialyzing fluid; however, these are
as a risk factor for development of cardiovascular disease.
present in high concentrations in the uremic blood.
Hypertension 42:1050, 2003.
Therefore, when a uremic patient is dialyzed, these sub-
Schrier RW: Atlas of Diseases of the Kidney. http://
stances are lost in large quantities into the dialyzing
fluid.
Shankar SS, Brater DC: Loop diuretics: from the Na-K-2Cl
The effectiveness of the artificial kidney can be
transporter to clinical use. Am J Physiol Renal Physiol
expressed in terms of the amount of plasma that is
284:F11, 2003.
cleared of different substances each minute, which, as
Singri N, Ahya SN, Levin ML: Acute renal failure. JAMA
discussed in Chapter
27, is the primary means for
289:747, 2003.
expressing the functional effectiveness of the kidneys
themselves to rid the body of unwanted substances.
Wilcox CS: New insights into diuretic use in patients with
Most artificial kidneys can clear urea from the plasma
chronic renal disease. J Am Soc Nephrol 13:798, 2002.
U N I T VI
Blood Cells,
Immunity, and
Blood Clotting
32. Red Blood Cells, Anemia, and Polycythemia
33. Resistance of the Body to Infection: I. Leukocytes,
Granulocytes, the Monocyte-Macrophage System,
and Inflammation
34. Resistance of the Body to Infection: II. Immunity
and Allergy
35. Blood Types; Transfusion; Tissue and Organ
Transplantation
36. Hemostasis and Blood Coagulation
C H A P T E R
3
2
Red Blood Cells, Anemia,
and Polycythemia
With this chapter we begin discussing the blood cells
and cells of the macrophage system and lymphatic
system. We first present the functions of red blood
cells, which are the most abundant cells of the blood
and are necessary for the delivery of oxygen to the
tissues.
Red Blood Cells (Erythrocytes)
The major function of red blood cells, also known as erythrocytes, is to trans-
port hemoglobin, which in turn carries oxygen from the lungs to the tissues. In
some lower animals, hemoglobin circulates as free protein in the plasma, not
enclosed in red blood cells. When it is free in the plasma of the human being,
about 3 per cent of it leaks through the capillary membrane into the tissue
spaces or through the glomerular membrane of the kidney into the glomerular
filtrate each time the blood passes through the capillaries. Therefore, for hemo-
globin to remain in the human blood stream, it must exist inside red blood cells.
The red blood cells have other functions besides transport of hemoglobin.
For instance, they contain a large quantity of carbonic anhydrase, an enzyme
that catalyzes the reversible reaction between carbon dioxide (CO2) and water
to form carbonic acid (H2CO3), increasing the rate of this reaction several thou-
sandfold. The rapidity of this reaction makes it possible for the water of the
blood to transport enormous quantities of CO2 in the form of bicarbonate ion
(HCO3-) from the tissues to the lungs, where it is reconverted to CO2 and
expelled into the atmosphere as a body waste product. The hemoglobin in the
cells is an excellent acid-base buffer (as is true of most proteins), so that the red
blood cells are responsible for most of the acid-base buffering power of whole
blood.
Shape and Size of Red Blood Cells. Normal red blood cells, shown in Figure 32-3,
are biconcave discs having a mean diameter of about 7.8 micrometers and
a thickness of 2.5 micrometers at the thickest point and 1 micrometer or less
in the center. The average volume of the red blood cell is 90 to 95 cubic
micrometers.
The shapes of red blood cells can change remarkably as the cells squeeze
through capillaries. Actually, the red blood cell is a “bag” that can be deformed
into almost any shape. Furthermore, because the normal cell has a great excess
of cell membrane for the quantity of material inside, deformation does not
stretch the membrane greatly and, consequently, does not rupture the cell, as
would be the case with many other cells.
Concentration of Red Blood Cells in the Blood. In normal men, the average number
of red blood cells per cubic millimeter is 5,200,000 (±300,000); in normal women,
it is 4,700,000 (±300,000). Persons living at high altitudes have greater numbers
of red blood cells. This is discussed later.
Quantity of Hemoglobin in the Cells. Red blood cells have the ability to concentrate
hemoglobin in the cell fluid up to about 34 grams in each 100 milliliters of cells.
419
420
Unit VI Blood Cells, Immunity, and Blood Clotting
The concentration does not rise above this value,
most red cells continue to be produced in the marrow
because this is the metabolic limit of the cell’s hemo-
of the membranous bones, such as the vertebrae,
globin-forming mechanism. Furthermore, in normal
sternum, ribs, and ilia. Even in these bones, the marrow
people, the percentage of hemoglobin is almost always
becomes less productive as age increases.
near the maximum in each cell. However, when hemo-
globin formation is deficient, the percentage of hemo-
Genesis of Blood Cells
globin in the cells may fall considerably below this
Pluripotential Hematopoietic Stem Cells, Growth Inducers, and
value, and the volume of the red cell may also decrease
Differentiation Inducers. The blood cells begin their lives
because of diminished hemoglobin to fill the cell.
in the bone marrow from a single type of cell called
When the hematocrit (the percentage of blood that
the pluripotential hematopoietic stem cell, from which
is cells—normally, 40 to 45 per cent) and the quantity
all the cells of the circulating blood are eventually
of hemoglobin in each respective cell are normal, the
derived. Figure 32-2 shows the successive divisions of
whole blood of men contains an average of 15 grams
the pluripotential cells to form the different circulat-
of hemoglobin per 100 milliliters of cells; for women,
ing blood cells. As these cells reproduce, a small
it contains an average of 14 grams per 100 milliliters.
portion of them remains exactly like the original
As discussed in connection with blood transport of
pluripotential cells and is retained in the bone marrow
oxygen in Chapter 40, each gram of pure hemoglobin
to maintain a supply of these, although their numbers
is capable of combining with 1.34 milliliters of oxygen.
diminish with age. Most of the reproduced cells,
Therefore, in a normal man, a maximum of about 20
however, differentiate to form the other cell types
milliliters of oxygen can be carried in combination
shown to the right in Figure 32-2. The intermediate-
with hemoglobin in each 100 milliliters of blood, and
stage cells are very much like the pluripotential
in a normal woman, 19 milliliters of oxygen can be
stem cells, even though they have already become
carried.
committed to a particular line of cells and are called
committed stem cells.
The different committed stem cells, when grown in
Production of Red Blood Cells
culture, will produce colonies of specific types of blood
cells. A committed stem cell that produces erythro-
Areas of the Body That Produce Red Blood Cells. In the early
cytes is called a colony-forming unit-erythrocyte, and
weeks of embryonic life, primitive, nucleated red blood
the abbreviation CFU-E is used to designate this type
cells are produced in the yolk sac. During the middle
of stem cell. Likewise, colony-forming units that form
trimester of gestation, the liver is the main organ for
granulocytes and monocytes have the designation
production of red blood cells, but reasonable numbers
CFU-GM, and so forth.
are also produced in the spleen and lymph nodes. Then,
Growth and reproduction of the different stem
during the last month or so of gestation and after birth,
cells are controlled by multiple proteins called growth
red blood cells are produced exclusively in the bone
inducers. Four major growth inducers have been
marrow.
described, each having different characteristics. One of
As demonstrated in Figure 32-1, the bone marrow
these, interleukin-3, promotes growth and reproduc-
of essentially all bones produces red blood cells until
tion of virtually all the different types of committed
a person is 5 years old. The marrow of the long bones,
stem cells, whereas the others induce growth of only
except for the proximal portions of the humeri and
specific types of cells.
tibiae, becomes quite fatty and produces no more red
The growth inducers promote growth but not dif-
blood cells after about age 20 years. Beyond this age,
ferentiation of the cells. This is the function of another
set of proteins called differentiation inducers. Each of
these causes one type of committed stem cell to dif-
ferentiate one or more steps toward a final adult blood
cell.
100
Formation of the growth inducers and differentia-
tion inducers is itself controlled by factors outside the
75
bone marrow. For instance, in the case of erythrocytes
(red blood cells), exposure of the blood to low oxygen
50
for a long time results in growth induction, differenti-
ation, and production of greatly increased numbers of
25
Rib
erythrocytes, as discussed later in the chapter. In the
case of some of the white blood cells, infectious
0
0
5 10 15 20
30
40
50
60
70
diseases cause growth, differentiation, and eventual
Age (years)
formation of specific types of white blood cells that are
needed to combat each infection.
Stages of Differentiation of Red Blood Cells
Figure 32-1
The first cell that can be identified as belonging to the
red blood cell series is the proerythroblast, shown at
Relative rates of red blood cell production in the bone marrow of
different bones at different ages.
the starting point in Figure 32-3. Under appropriate
Chapter 32
Red Blood Cells, Anemia, and Polycythemia
421
Erythrocytes
CFU-B
CFU-E
(Colony-forming
(Colony-forming
unit-blast)
unit-erythrocytes)
Granulocytes
(Neutrophils)
(Eosinophils)
(Basophils)
Monocytes
PHSC
CFU-S
CFU-GM
(Pluripotent
(Colony-forming
(Colony-forming unit-
hematopoietic
unit-spleen)
granulocytes, monocytes)
Macrocytes
stem cell)
Megakaryocytes
CFU-M
Platelets
(Colony-forming unit-
megakaryocytes)
T lymphocytes
Figure 32-2
PHSC
LSC
Formation of the multiple different
B lymphocytes
blood cells from the original pluripo-
(Lymphoid stem cell)
tent hematopoietic stem cell (PHSC)
in the bone marrow.
GENESIS OF RBC
Proerythroblast
Basophil
erythroblast
Microcytic,
hypochromic anemia
Sickle cell anemia
Polychromatophil
erythroblast
Orthochromatic
erythroblast
Reticulocyte
Erythrocytes
Megaloblastic anemia
Erythroblastosis fetalis
Figure 32-3
Genesis of normal red blood cells (RBCs) and characteristics of RBCs in different types of anemias.
422
Unit VI Blood Cells, Immunity, and Blood Clotting
stimulation, large numbers of these cells are formed
from the CFU-E stem cells.
Once the proerythroblast has been formed, it
Hematopoietic Stem Cells
divides multiple times, eventually forming many
Kidney
mature red blood cells. The first-generation cells are
called basophil erythroblasts because they stain with
Proerythroblasts
basic dyes; the cell at this time has accumulated very
Erythropoietin
little hemoglobin. In the succeeding generations, as
shown in Figure 32-3, the cells become filled with
Red Blood Cells
hemoglobin to a concentration of about 34 per cent,
Decreases
the nucleus condenses to a small size, and its final
remnant is absorbed or extruded from the cell. At the
Tissue Oxygenation
same time, the endoplasmic reticulum is also reab-
sorbed. The cell at this stage is called a reticulocyte
because it still contains a small amount of basophilic
Decreases
material, consisting of remnants of the Golgi appara-
tus, mitochondria, and a few other cytoplasmic
organelles. During this reticulocyte stage, the cells pass
Factors that decrease
oxygenation
from the bone marrow into the blood capillaries by
1. Low blood volume
diapedesis (squeezing through the pores of the capil-
2. Anemia
lary membrane).
3. Low hemoglobin
The remaining basophilic material in the reticulo-
4. Poor blood flow
cyte normally disappears within 1 to 2 days, and the
5. Pulmonary disease
cell is then a mature erythrocyte. Because of the short
life of the reticulocytes, their concentration among all
the red cells of the blood is normally slightly less than
1 per cent.
Figure 32-4
Function of the erythropoietin mechanism to increase production
Regulation of Red Blood Cell Production—Role
of red blood cells when tissue oxygenation decreases.
of Erythropoietin
The total mass of red blood cells in the circulatory
system is regulated within narrow limits, so that (1) an
adequate number of red cells is always available to
can also increase the rate of red cell production. This
provide sufficient transport of oxygen from the lungs
is especially apparent in prolonged cardiac failure and
to the tissues, yet (2) the cells do not become so numer-
in many lung diseases, because the tissue hypoxia
ous that they impede blood flow. What we know about
resulting from these conditions increases red cell pro-
this control mechanism is diagrammed in Figure 32-4
duction, with a resultant increase in hematocrit and
and is as follows.
usually total blood volume as well.
Tissue Oxygenation Is the Most Essential Regulator of Red
Erythropoietin Stimulates Red Cell Production, and Its Forma-
Blood Cell Production. Any condition that causes the
tion Increases in Response to Hypoxia. The principal stim-
quantity of oxygen transported to the tissues to
ulus for red blood cell production in low oxygen states
decrease ordinarily increases the rate of red blood cell
is a circulating hormone called erythropoietin, a gly-
production. Thus, when a person becomes extremely
coprotein with a molecular weight of about 34,000. In
anemic as a result of hemorrhage or any other condi-
the absence of erythropoietin, hypoxia has little or no
tion, the bone marrow immediately begins to produce
effect in stimulating red blood cell production. But
large quantities of red blood cells. Also, destruction of
when the erythropoietin system is functional, hypoxia
major portions of the bone marrow by any means,
causes a marked increase in erythropoietin produc-
especially by x-ray therapy, causes hyperplasia of the
tion, and the erythropoietin in turn enhances red
remaining bone marrow, thereby attempting to supply
blood cell production until the hypoxia is relieved.
the demand for red blood cells in the body.
At very high altitudes, where the quantity of oxygen
Role of the Kidneys in Formation of Erythropoietin. In
in the air is greatly decreased, insufficient oxygen is
the normal person, about 90 per cent of all erythro-
transported to the tissues, and red cell production is
poietin is formed in the kidneys; the remainder is
greatly increased. In this case, it is not the concentra-
formed mainly in the liver. It is not known exactly
tion of red blood cells in the blood that controls red
where in the kidneys the erythropoietin is formed.
cell production but the amount of oxygen transported
One likely possibility is that the renal tubular epithe-
to the tissues in relation to tissue demand for oxygen.
lial cells secrete the erythropoietin, because anemic
Various diseases of the circulation that cause
blood is unable to deliver enough oxygen from the
decreased blood flow through the peripheral vessels,
peritubular capillaries to the highly oxygen-consuming
and particularly those that cause failure of oxygen
tubular cells, thus stimulating erythropoietin pro-
absorption by the blood as it passes through the lungs,
duction.
Chapter 32
Red Blood Cells, Anemia, and Polycythemia
423
At times, hypoxia in other parts of the body, but
building blocks of DNA. Therefore, lack of either
not in the kidneys, stimulates kidney erythropoietin
vitamin B12 or folic acid causes abnormal and dimin-
secretion, which suggests that there might be some
ished DNA and, consequently, failure of nuclear
nonrenal sensor that sends an additional signal to the
maturation and cell division. Furthermore, the ery-
kidneys to produce this hormone. In particular, both
throblastic cells of the bone marrow, in addition to
norepinephrine and epinephrine and several of the
failing to proliferate rapidly, produce mainly larger
prostaglandins stimulate erythropoietin production.
than normal red cells called macrocytes, and the cell
When both kidneys are removed from a person or
itself has a flimsy membrane and is often irregular,
when the kidneys are destroyed by renal disease, the
large, and oval instead of the usual biconcave disc.
person invariably becomes very anemic because the 10
These poorly formed cells, after entering the circulat-
per cent of the normal erythropoietin formed in other
ing blood, are capable of carrying oxygen normally, but
tissues (mainly in the liver) is sufficient to cause only
their fragility causes them to have a short life, one half
one third to one half the red blood cell formation
to one third normal. Therefore, it is said that deficiency
needed by the body.
of either vitamin B12 or folic acid causes maturation
failure in the process of erythropoiesis.
Effect of Erythropoietin in Erythrogenesis. When an
animal or a person is placed in an atmosphere of low
Maturation Failure Caused by Poor Absorption of Vitamin
oxygen, erythropoietin begins to be formed within
B12
from the Gastrointestinal Tract—Pernicious Anemia. A
minutes to hours, and it reaches maximum production
common cause of red blood cell maturation failure is
within 24 hours. Yet almost no new red blood cells
failure to absorb vitamin B12 from the gastrointestinal
appear in the circulating blood until about 5 days later.
tract. This often occurs in the disease pernicious
From this fact, as well as other studies, it has been
anemia, in which the basic abnormality is an atrophic
determined that the important effect of erythropoietin
gastric mucosa that fails to produce normal gastric
is to stimulate the production of proerythroblasts from
secretions. The parietal cells of the gastric glands
hematopoietic stem cells in the bone marrow. In ad-
secrete a glycoprotein called intrinsic factor, which
dition, once the proerythroblasts are formed, the
combines with vitamin B12 in food and makes the B12
erythropoietin causes these cells to pass more rapidly
available for absorption by the gut. It does this in the
through the different erythroblastic stages than they
following way: (1) Intrinsic factor binds tightly with
normally do, further speeding up the production of
the vitamin B12. In this bound state, the B12 is protected
new red blood cells. The rapid production of cells con-
from digestion by the gastrointestinal secretions. (2)
tinues as long as the person remains in a low oxygen
Still in the bound state, intrinsic factor binds to specific
state or until enough red blood cells have been pro-
receptor sites on the brush border membranes of the
duced to carry adequate amounts of oxygen to the
mucosal cells in the ileum. (3) Then, vitamin B12 is
tissues despite the low oxygen; at this time, the rate of
transported into the blood during the next few hours
erythropoietin production decreases to a level that will
by the process of pinocytosis, carrying intrinsic factor
maintain the required number of red cells but not an
and the vitamin together through the membrane. Lack
excess.
of intrinsic factor, therefore, causes diminished avail-
In the absence of erythropoietin, few red blood
ability of vitamin B12 because of faulty absorption of
cells are formed by the bone marrow. At the other
the vitamin.
extreme, when large quantities of erythropoietin
Once vitamin B12 has been absorbed from the gas-
are formed available, and if there is plenty of iron and
trointestinal tract, it is first stored in large quantities in
other required nutrients available, the rate of red
the liver, then released slowly as needed by the bone
blood cell production can rise to perhaps 10 or more
marrow. The minimum amount of vitamin B12 required
times normal. Therefore, the erythropoietin mecha-
each day to maintain normal red cell maturation is
nism for controlling red blood cell production is a
only 1 to 3 micrograms, and the normal storage in the
powerful one.
liver and other body tissues is about 1000 times this
amount. Therefore, 3 to 4 years of defective B12 absorp-
tion are usually required to cause maturation failure
Maturation of Red Blood Cells—Requirement
anemia.
for Vitamin B12 (Cyanocobalamin) and
Folic Acid
Because of the continuing need to replenish red blood
Failure of Maturation Caused by Deficiency of Folic Acid
cells, the erythropoietic cells of the bone marrow are
(Pteroylglutamic Acid). Folic acid is a normal constituent
among the most rapidly growing and reproducing cells
of green vegetables, some fruits, and meats (especially
in the entire body. Therefore, as would be expected,
liver). However, it is easily destroyed during cooking.
their maturation and rate of production are affected
Also, people with gastrointestinal absorption abnor-
greatly by a person’s nutritional status.
malities, such as the frequently occurring small intes-
Especially important for final maturation of the red
tinal disease called sprue, often have serious difficulty
blood cells are two vitamins, vitamin B
12 and folic acid.
absorbing both folic acid and vitamin B12. Therefore,
Both of these are essential for the synthesis of DNA,
in many instances of maturation failure, the cause is
because each in a different way is required for the for-
deficiency of intestinal absorption of both folic acid
mation of thymidine triphosphate, one of the essential
and vitamin B12.
424
Unit VI Blood Cells, Immunity, and Blood Clotting
A
P
C
C
I.
2 succinyl-CoA + 2 glycine
HC
CH
N
II.
4 pyrrole
protoporphyrin IX
H
(pyrrole)
III.
protoporphyrin IX + Fe++
heme
IV.
heme + polypeptide
hemoglobin chain (a or b)
V.
2 a chains + 2 b chains
hemoglobin A
Figure 32-5
Formation of hemoglobin.
Formation of Hemoglobin
Synthesis of hemoglobin begins in the proerythro-
CH2
blasts and continues even into the reticulocyte stage of
H
the red blood cells. Therefore, when reticulocytes leave
C
CH
CH3
the bone marrow and pass into the blood stream, they
A
B
CH
CH2
continue to form minute quantities of hemoglobin for
H3C
O2
another day or so until they become mature erythro-
N
(-)N
cytes.
Figure 32-5 shows the basic chemical steps in the
HC
Fe
CH
formation of hemoglobin. First, succinyl-CoA, formed
N(-)
N
in the Krebs metabolic cycle (as explained in Chapter
67), binds with glycine to form a pyrrole molecule. In
H3C
D
C
CH3
turn, four pyrroles combine to form protoporphyrin
IX, which then combines with iron to form the heme
C
molecule. Finally, each heme molecule combines with
CH2
H
CH2
a long polypeptide chain, a globin synthesized by
ribosomes, forming a subunit of hemoglobin called a
CH2
CH2
hemoglobin chain (Figure 32-6). Each chain has a
molecular weight of about 16,000; four of these in turn
COOH
COOH
bind together loosely to form the whole hemoglobin
Polypepitide
molecule.
(hemoglobin chain-a or b)
There are several slight variations in the different
subunit hemoglobin chains, depending on the amino
acid composition of the polypeptide portion. The dif-
ferent types of chains are designated alpha chains, beta
Figure 32-6
chains, gamma chains, and delta chains. The most
common form of hemoglobin in the adult human
Basic structure of the hemoglobin molecule, showing one of the
being, hemoglobin A, is a combination of two alpha
four heme chains that bind together to form the hemoglobin
molecule.
chains and two beta chains. Hemoglobin A has a
molecular weight of 64,458.
Because each hemoglobin chain has a heme pros-
thetic group containing an atom of iron, and because
one point in each of the two beta chains. When this
there are four hemoglobin chains in each hemoglobin
type of hemoglobin is exposed to low oxygen, it forms
molecule, one finds four iron atoms in each hemoglo-
elongated crystals inside the red blood cells that are
bin molecule; each of these can bind loosely with one
sometimes 15 micrometers in length. These make it
molecule of oxygen, making a total of four molecules
almost impossible for the cells to pass through many
of oxygen (or eight oxygen atoms) that can be trans-
small capillaries, and the spiked ends of the crystals are
ported by each hemoglobin molecule.
likely to rupture the cell membranes, leading to sickle
The types of hemoglobin chains in the hemoglobin
cell anemia.
molecule determine the binding affinity of the hemo-
globin for oxygen. Abnormalities of the chains can
Combination of Hemoglobin with Oxygen. The most impor-
alter the physical characteristics of the hemoglobin
tant feature of the hemoglobin molecule is its ability
molecule as well. For instance, in sickle cell anemia, the
to combine loosely and reversibly with oxygen. This
amino acid valine is substituted for glutamic acid at
ability is discussed in detail in Chapter 40 in relation
Chapter 32
Red Blood Cells, Anemia, and Polycythemia
425
to respiration, because the primary function of hemo-
globulin, apotransferrin, to form transferrin, which is
globin in the body is to combine with oxygen in the
then transported in the plasma. The iron is loosely
lungs and then to release this oxygen readily in the
bound in the transferrin and, consequently, can be
peripheral tissue capillaries, where the gaseous tension
released to any tissue cell at any point in the body.
of oxygen is much lower than in the lungs.
Excess iron in the blood is deposited especially in the
Oxygen does not combine with the two positive
liver hepatocytes and less in the reticuloendothelial
bonds of the iron in the hemoglobin molecule. Instead,
cells of the bone marrow.
it binds loosely with one of the so-called coordination
In the cell cytoplasm, iron combines mainly with
bonds of the iron atom. This is an extremely loose
a protein, apoferritin, to form ferritin. Apoferritin
bond, so that the combination is easily reversible. Fur-
has a molecular weight of about
460,000, and
thermore, the oxygen does not become ionic oxygen
varying quantities of iron can combine in clusters of
but is carried as molecular oxygen (composed of two
iron radicals with this large molecule; therefore,
oxygen atoms) to the tissues, where, because of the
ferritin may contain only a small amount of iron or a
loose, readily reversible combination, it is released into
large amount. This iron stored as ferritin is called
the tissue fluids still in the form of molecular oxygen
storage iron.
rather than ionic oxygen.
Smaller quantities of the iron in the storage pool are
in an extremely insoluble form called hemosiderin.
This is especially true when the total quantity of iron
Iron Metabolism
in the body is more than the apoferritin storage pool
can accommodate. Hemosiderin collects in cells in the
Because iron is important for the formation not only
form of large clusters that can be observed micro-
of hemoglobin but also of other essential elements in
scopically as large particles. In contrast, ferritin parti-
the body (e.g., myoglobin, cytochromes, cytochrome
cles are so small and dispersed that they usually can
oxidase, peroxidase, catalase), it is important to under-
be seen in the cell cytoplasm only with the electron
stand the means by which iron is utilized in the body.
microscope.
The total quantity of iron in the body averages 4 to 5
When the quantity of iron in the plasma falls low,
grams, about 65 per cent of which is in the form of
some of the iron in the ferritin storage pool is removed
hemoglobin. About 4 per cent is in the form of myo-
easily and transported in the form of transferrin in the
globin, 1 per cent is in the form of the various heme
plasma to the areas of the body where it is needed. A
compounds that promote intracellular oxidation, 0.1
unique characteristic of the transferrin molecule is that
per cent is combined with the protein transferrin in the
it binds strongly with receptors in the cell membranes
blood plasma, and 15 to 30 per cent is stored for later
of erythroblasts in the bone marrow. Then, along with
use, mainly in the reticuloendothelial system and liver
its bound iron, it is ingested into the erythroblasts by
parenchymal cells, principally in the form of ferritin.
endocytosis. There the transferrin delivers the iron
directly to the mitochondria, where heme is synthe-
Transport and Storage of Iron. Transport, storage, and
sized. In people who do not have adequate quantities
metabolism of iron in the body are diagrammed in
of transferrin in their blood, failure to transport iron
Figure 32-7 and can be explained as follows: When
to the erythroblasts in this manner can cause severe
iron is absorbed from the small intestine, it immedi-
hypochromic anemia—that is, red cells that contain
ately combines in the blood plasma with a beta
much less hemoglobin than normal.
Bilirubin (excreted)
Tissues
Ferritin
Hemosiderin
Macrophages
Heme
Free
Degrading hemoglobin
Enzymes
iron
Free iron
Hemoglobin
Transferrin-Fe
Red Cells
Plasma
Blood loss - 0.7 mg Fe Fe++ absorbed Fe excreted-0.6 mg daily
daily in menses
(small intestine)
Figure 32-7
Iron transport and metabolism.
426
Unit VI Blood Cells, Immunity, and Blood Clotting
When red blood cells have lived their life span and
progressively less active, and the cells become more
are destroyed, the hemoglobin released from the cells
and more fragile, presumably because their life
is ingested by monocyte-macrophage cells. There,
processes wear out.
iron is liberated and is stored mainly in the ferritin
Once the red cell membrane becomes fragile, the
pool to be used as needed for the formation of new
cell ruptures during passage through some tight spot
hemoglobin.
of the circulation. Many of the red cells self-destruct
in the spleen, where they squeeze through the red pulp
Daily Loss of Iron. A man excretes about 0.6 milligram
of the spleen. There, the spaces between the structural
of iron each day, mainly into the feces. Additional
trabeculae of the red pulp, through which most of the
quantities of iron are lost when bleeding occurs. For a
cells must pass, are only 3 micrometers wide, in com-
woman, additional menstrual loss of blood brings long-
parison with the 8-micrometer diameter of the red
term iron loss to an average of about 1.3 mg/day.
cell. When the spleen is removed, the number of old
abnormal red cells circulating in the blood increases
Absorption of Iron from the Intestinal Tract
considerably.
Iron is absorbed from all parts of the small intestine,
mostly by the following mechanism. The liver secretes
Destruction of Hemoglobin. When red blood cells burst
moderate amounts of apotransferrin into the bile,
and release their hemoglobin, the hemoglobin is
which flows through the bile duct into the duodenum.
phagocytized almost immediately by macrophages in
Here, the apotransferrin binds with free iron and also
many parts of the body, but especially by the Kupffer
with certain iron compounds, such as hemoglobin and
cells of the liver and macrophages of the spleen and
myoglobin from meat, two of the most important
bone marrow. During the next few hours to days, the
sources of iron in the diet. This combination is called
macrophages release iron from the hemoglobin and
transferrin. It, in turn, is attracted to and binds with
pass it back into the blood, to be carried by transfer-
receptors in the membranes of the intestinal epithelial
rin either to the bone marrow for the production of
cells. Then, by pinocytosis, the transferrin molecule,
new red blood cells or to the liver and other tissues for
carrying its iron store, is absorbed into the epithelial
storage in the form of ferritin. The porphyrin portion
cells and later released into the blood capillaries
of the hemoglobin molecule is converted by the
beneath these cells in the form of plasma transferrin.
macrophages, through a series of stages, into the bile
Iron absorption from the intestines is extremely
pigment bilirubin, which is released into the blood and
slow, at a maximum rate of only a few milligrams per
later removed from the body by secretion through the
day. This means that even when tremendous quantities
liver into the bile; this is discussed in relation to liver
of iron are present in the food, only small proportions
function in Chapter 70.
can be absorbed.
Regulation of Total Body Iron by Controlling Rate of Absorption.
Anemias
When the body has become saturated with iron so that
essentially all apoferritin in the iron storage areas is
Anemia means deficiency of hemoglobin in the blood,
already combined with iron, the rate of additional iron
which can be caused by either too few red blood cells
absorption from the intestinal tract becomes greatly
or too little hemoglobin in the cells. Some types of
decreased. Conversely, when the iron stores have
anemia and their physiologic causes are the following.
become depleted, the rate of absorption can acceler-
ate probably five or more times normal. Thus, total
Blood Loss Anemia. After rapid hemorrhage, the body
body iron is regulated mainly by altering the rate of
replaces the fluid portion of the plasma in 1 to 3 days,
absorption.
but this leaves a low concentration of red blood cells.
If a second hemorrhage does not occur, the red blood
cell concentration usually returns to normal within 3
Life Span and Destruction of Red
to 6 weeks.
Blood Cells
In chronic blood loss, a person frequently cannot
absorb enough iron from the intestines to form hemo-
When red blood cells are delivered from the bone
globin as rapidly as it is lost. Red cells are then pro-
marrow into the circulatory system, they normally cir-
duced that are much smaller than normal and have too
culate an average of 120 days before being destroyed.
little hemoglobin inside them, giving rise to microcytic,
Even though mature red cells do not have a nucleus,
hypochromic anemia, which is shown in Figure 32-3.
mitochondria, or endoplasmic reticulum, they do have
cytoplasmic enzymes that are capable of metabolizing
Aplastic Anemia. Bone marrow aplasia means lack of
glucose and forming small amounts of adenosine
functioning bone marrow. For instance, a person
triphosphate. These enzymes also (1) maintain pliabil-
exposed to gamma ray radiation from a nuclear bomb
ity of the cell membrane, (2) maintain membrane
blast can sustain complete destruction of bone
transport of ions, (3) keep the iron of the cells’ hemo-
marrow, followed in a few weeks by lethal anemia.
globin in the ferrous form rather than ferric form, and
Likewise, excessive x-ray treatment, certain industrial
(4) prevent oxidation of the proteins in the red cells.
chemicals, and even drugs to which the person might
Even so, the metabolic systems of old red cells become
be sensitive can cause the same effect.
Chapter 32
Red Blood Cells, Anemia, and Polycythemia
427
Megaloblastic Anemia. Based on the earlier discussions
Rh-positive cells fragile, leading to rapid rupture and
of vitamin B12, folic acid, and intrinsic factor from the
causing the child to be born with serious anemia. This
stomach mucosa, one can readily understand that loss
is discussed in Chapter 35 in relation to the Rh factor
of any one of these can lead to slow reproduction of
of blood. The extremely rapid formation of new red
erythroblasts in the bone marrow. As a result, the red
cells to make up for the destroyed cells in erythro-
cells grow too large, with odd shapes, and are called
blastosis fetalis causes a large number of early blast
megaloblasts. Thus, atrophy of the stomach mucosa,
forms of red cells to be released from the bone marrow
as occurs in pernicious anemia, or loss of the entire
into the blood.
stomach after surgical total gastrectomy can lead to
megaloblastic anemia. Also, patients who have intes-
tinal sprue, in which folic acid, vitamin B12, and other
Effects of Anemia on Function of the
vitamin B compounds are poorly absorbed, often
Circulatory System
develop megaloblastic anemia. Because in these states
the erythroblasts cannot proliferate rapidly enough to
The viscosity of the blood, which was discussed in
form normal numbers of red blood cells, those red cells
Chapter 14, depends almost entirely on the blood con-
that are formed are mostly oversized, have bizarre
centration of red blood cells. In severe anemia, the
shapes, and have fragile membranes. These cells
blood viscosity may fall to as low as 1.5 times that of
rupture easily, leaving the person in dire need of an
water rather than the normal value of about 3. This
adequate number of red cells.
decreases the resistance to blood flow in the periph-
eral blood vessels, so that far greater than normal
Hemolytic Anemia. Different abnormalities of the red
quantities of blood flow through the tissues and return
blood cells, many of which are hereditarily acquired,
to the heart, thereby greatly increasing cardiac output.
make the cells fragile, so that they rupture easily as
Moreover, hypoxia resulting from diminished trans-
they go through the capillaries, especially through the
port of oxygen by the blood causes the peripheral
spleen. Even though the number of red blood cells
tissue blood vessels to dilate, allowing a further
formed may be normal, or even much greater than
increase in the return of blood to the heart and
normal in some hemolytic diseases, the life span of the
increasing the cardiac output to a still higher level—
fragile red cell is so short that the cells are destroyed
sometimes three to four times normal. Thus, one of the
faster than they can be formed, and serious anemia
major effects of anemia is greatly increased cardiac
results. Some of these types of anemia are the
output, as well as increased pumping workload on the
following.
heart.
In hereditary spherocytosis, the red cells are very
The increased cardiac output in anemia partially
small and spherical rather than being biconcave discs.
offsets the reduced oxygen-carrying effect of the
These cells cannot withstand compression forces
anemia, because even though each unit quantity of
because they do not have the normal loose, baglike cell
blood carries only small quantities of oxygen, the rate
membrane structure of the biconcave discs. On passing
of blood flow may be increased enough so that almost
through the splenic pulp and some other tight vas-
normal quantities of oxygen are actually delivered to
cular beds, they are easily ruptured by even slight
the tissues. However, when a person with anemia
compression.
begins to exercise, the heart is not capable of pumping
In sickle cell anemia, which is present in 0.3 to 1.0
much greater quantities of blood than it is already
per cent of West African and American blacks, the cells
pumping. Consequently, during exercise, which
have an abnormal type of hemoglobin called hemo-
greatly increases tissue demand for oxygen, extreme
globin S, containing faulty beta chains in the hemo-
tissue hypoxia results, and acute cardiac failure
globin molecule, as explained earlier in the chapter.
ensues.
When this hemoglobin is exposed to low concentra-
tions of oxygen, it precipitates into long crystals inside
the red blood cell. These crystals elongate the cell and
Polycythemia
give it the appearance of a sickle rather than a bicon-
cave disc. The precipitated hemoglobin also damages
Secondary Polycythemia. Whenever the tissues become
the cell membrane, so that the cells become highly
hypoxic because of too little oxygen in the breathed
fragile, leading to serious anemia. Such patients fre-
air, such as at high altitudes, or because of failure of
quently experience a vicious circle of events called a
oxygen delivery to the tissues, such as in cardiac
sickle cell disease “crisis,” in which low oxygen tension
failure, the blood-forming organs automatically
in the tissues causes sickling, which leads to ruptured
produce large quantities of extra red blood cells. This
red cells, which causes a further decrease in oxygen
condition is called secondary polycythemia, and the
tension and still more sickling and red cell destruction.
red cell count commonly rises to 6 to 7 million/mm3,
Once the process starts, it progresses rapidly, eventu-
about 30 per cent above normal.
ating in a serious decrease in red blood cells within a
A common type of secondary polycythemia, called
few hours and, often, death.
physiologic polycythemia, occurs in natives who live at
In erythroblastosis fetalis, Rh-positive red blood
altitudes of 14,000 to 17,000 feet, where the atmos-
cells in the fetus are attacked by antibodies from
pheric oxygen is very low. The blood count is generally
an Rh-negative mother. These antibodies make the
6 to 7 million/mm3; this allows these people to perform
428
Unit VI Blood Cells, Immunity, and Blood Clotting
reasonably high levels of continuous work even in a
increase peripheral resistance and, thereby, increase
rarefied atmosphere.
arterial pressure. Beyond certain limits, however, these
regulations fail, and hypertension develops.
Polycythemia Vera
(Erythremia). In addition to those
The color of the skin depends to a great extent on
people who have physiologic polycythemia, others
the quantity of blood in the skin subpapillary venous
have a pathological condition known as polycythemia
plexus. In polycythemia vera, the quantity of blood in
vera, in which the red blood cell count may be 7 to 8
this plexus is greatly increased. Further, because the
million/mm3 and the hematocrit may be 60 to 70 per
blood passes sluggishly through the skin capillaries
cent instead of the normal 40 to 45 per cent. Poly-
before entering the venous plexus, a larger than
cythemia vera is caused by a genetic aberration in the
normal quantity of hemoglobin is deoxygenated. The
hemocytoblastic cells that produce the blood cells. The
blue color of all this deoxygenated hemoglobin masks
blast cells no longer stop producing red cells when too
the red color of the oxygenated hemoglobin. There-
many cells are already present. This causes excess pro-
fore, a person with polycythemia vera ordinarily has a
duction of red blood cells in the same manner that a
ruddy complexion with a bluish (cyanotic) tint to the
breast tumor causes excess production of a specific
skin.
type of breast cell. It usually causes excess production
of white blood cells and platelets as well.
In polycythemia vera, not only does the hematocrit
References
increase, but the total blood volume also increases, on
some occasions to almost twice normal. As a result, the
Alayash AI: Oxygen therapeutics: can we tame haemoglo-
entire vascular system becomes intensely engorged. In
bin? Nat Rev Drug Discov 3:152, 2004.
Brissot P, Troadec MB, Loreal O: The clinical relevance of
addition, many blood capillaries become plugged by
new insights in iron transport and metabolism. Curr
the viscous blood; the viscosity of the blood in poly-
Hematol Rep 3:107, 2004.
cythemia vera sometimes increases from the normal of
Claster S, Vichinsky EP: Managing sickle cell disease. BMJ
3 times the viscosity of water to 10 times that of water.
327:1151, 2003.
Fandrey J: Oxygen-dependent and tissue-specific regulation
of erythropoietin gene expression. Am J Physiol Regul
Effect of Polycythemia on Function of
Integr Comp Physiol 286:R977, 2004.
Hallberg L: Perspectives on nutritional iron deficiency. Annu
the Circulatory System
Rev Nutr 21:1, 2001.
Hentze MW, Muckenthaler MU, Andrews NC: Balancing
Because of the greatly increased viscosity of the blood
acts: molecular control of mammalian iron metabolism.
in polycythemia, blood flow through the peripheral
Cell 117:285, 2004.
blood vessels is often very sluggish. In accordance with
Lappin T: The cellular biology of erythropoietin receptors.
the factors that regulate return of blood to the heart,
Oncologist 8(Suppl 1):15, 2003.
as discussed in Chapter 20, increasing blood viscosity
Maxwell P: HIF-1: an oxygen response system with special
decreases the rate of venous return to the heart. Con-
relevance to the kidney. J Am Soc Nephrol 14:2712, 2003.
versely, the blood volume is greatly increased in poly-
Persons DA: Update on gene therapy for hemoglobin disor-
cythemia, which tends to increase venous return.
ders. Curr Opin Mol Ther 5:508, 2003.
Pietrangelo A: Hereditary hemochromatosis—a new look at
Actually, the cardiac output in polycythemia is not far
an old disease. N Engl J Med 350:2383, 2004.
from normal, because these two factors more or less
Shah S, Vega R: Hereditary spherocytosis. Pediatr Rev
neutralize each other.
25:168, 2004.
The arterial pressure is also normal in most people
Tefferi A: A contemporary approach to the diagnosis and
with polycythemia, although in about one third of
management of polycythemia vera. Curr Hematol Rep
them, the arterial pressure is elevated. This means that
2:237, 2003.
the blood pressure-regulating mechanisms can usually
Trigg ME: Hematopoietic stem cells. Pediatrics
113(4
offset the tendency for increased blood viscosity to
Suppl):1051, 2004.
C H A P T E R
3
3
Resistance of the Body
to Infection: I. Leukocytes,
Granulocytes, the
Monocyte-Macrophage System,
and Inflammation
Our bodies are exposed continually to bacteria,
viruses, fungi, and parasites, all of which occur
normally and to varying degrees in the skin, the
mouth, the respiratory passageways, the intestinal
tract, the lining membranes of the eyes, and even the
urinary tract. Many of these infectious agents are
capable of causing serious abnormal physiologic
function or even death if they invade the deeper
tissues. In addition, we are exposed intermittently to other highly infectious bac-
teria and viruses besides those that are normally present, and these can cause
acute lethal diseases such as pneumonia, streptococcal infection, and typhoid
fever.
Our bodies have a special system for combating the different infectious and
toxic agents. This is comprised of blood leukocytes (white blood cells) and tissue
cells derived from leukocytes. These cells work together in two ways to prevent
disease: (1) by actually destroying invading bacteria or viruses by phagocytosis,
and (2) by forming antibodies and sensitized lymphocytes, one or both of which
may destroy or inactivate the invader. This chapter is concerned with the first
of these methods, and Chapter 34 with the second.
Leukocytes (White Blood Cells)
The leukocytes, also called white blood cells, are the mobile units of the body’s
protective system. They are formed partially in the bone marrow (granulocytes
and monocytes and a few lymphocytes) and partially in the lymph tissue (lym-
phocytes and plasma cells). After formation, they are transported in the blood
to different parts of the body where they are needed.
The real value of the white blood cells is that most of them are specifically
transported to areas of serious infection and inflammation, thereby providing a
rapid and potent defense against infectious agents. As we see later, the granu-
locytes and monocytes have a special ability to “seek out and destroy” a foreign
invader.
General Characteristics of Leukocytes
Types of White Blood Cells. Six types of white blood cells are normally present in
the blood. They are polymorphonuclear neutrophils, polymorphonuclear
eosinophils, polymorphonuclear basophils, monocytes, lymphocytes, and, occa-
sionally, plasma cells. In addition, there are large numbers of platelets, which are
fragments of another type of cell similar to the white blood cells found in the
bone marrow, the megakaryocyte. The first three types of cells, the polymor-
phonuclear cells, all have a granular appearance, as shown in cell numbers 7,
429
430
Unit VI Blood Cells, Immunity, and Blood Clotting
Genesis of Myelocytes
Genesis of Lymphocytes
1
3
2
13
4
8
11
Figure 33-1
14
Genesis of white blood cells. The
5
different cells of the myelocyte
series are
1, myeloblast;
2,
9
promyelocyte; 3, megakaryocyte;
4, neutrophil myelocyte; 5, young
15
neutrophil metamyelocyte;
6,
6
“band” neutrophil metamyelocyte;
7, polymorphonuclear neutrophil;
8, eosinophil myelocyte;
9,
eosinophil metamyelocyte;
10,
10
12
16
polymorphonuclear
eosinophil;
7
11, basophil myelocyte; 12, poly-
morphonuclear basophil;
13-16,
stages of monocyte formation.
10, and 12 in Figure 33-1, for which reason they are
Diapedesis
called granulocytes, or, in clinical terminology, “polys,”
because of the multiple nuclei.
The granulocytes and monocytes protect the body
against invading organisms mainly by ingesting
them—that is, by phagocytosis. The lymphocytes and
plasma cells function mainly in connection with the
Chemotaxis
immune system; this is discussed in Chapter 34. Finally,
source
the function of platelets is specifically to activate the
blood clotting mechanism, which is discussed in
Chapter 36.
Concentrations of the Different White Blood Cells in the Blood.
The adult human being has about 7000 white blood
Increased
cells per microliter of blood (in comparison with 5
permeability
million red blood cells). Of the total white blood cells,
Chemotactic
Margination
substance
the normal percentages of the different types are
approximately the following:
Figure 33-2
Movement of neutrophils by diapedesis through capillary pores
Polymorphonuclear neutrophils
62.0%
and by chemotaxis toward an area of tissue damage.
Polymorphonuclear eosinophils
2.3%
Polymorphonuclear basophils
0.4%
Monocytes
5.3%
Lymphocytes
30.0%
chapter. Aside from those cells committed to form red
blood cells, two major lineages of white blood cells are
formed, the myelocytic and the lymphocytic lineages.
The number of platelets, which are only cell frag-
The left side of Figure 33-1 shows the myelocytic
ments, in each microliter of blood is normally about
lineage, beginning with the myeloblast; the right
300,000.
shows the lymphocytic lineage, beginning with the
lymphoblast.
Genesis of the White Blood Cells
The granulocytes and monocytes are formed only
in the bone marrow. Lymphocytes and plasma cells
Early differentiation of the pluripotential hematopoi-
are produced mainly in the various lymphogenous
etic stem cell into the different types of committed
tissues—especially the lymph glands, spleen, thymus,
stem cells is shown in Figure 32-2 in the previous
tonsils, and various pockets of lymphoid tissue
Chapter 33
Resistance of the Body to Infection: I. Leukocytes, Granulocytes, the Monocyte-Macrophage System
431
elsewhere in the body, such as in the bone marrow and
other injurious agents. The neutrophils are mature
in so-called Peyer’s patches underneath the epithelium
cells that can attack and destroy bacteria even in the
in the gut wall.
circulating blood. Conversely, the tissue macrophages
The white blood cells formed in the bone marrow
begin life as blood monocytes, which are immature
are stored within the marrow until they are needed in
cells while still in the blood and have little ability to
the circulatory system. Then, when the need arises,
fight infectious agents at that time. However, once they
various factors cause them to be released
(these
enter the tissues, they begin to swell—sometimes
factors are discussed later). Normally, about three
increasing their diameters as much as fivefold—to as
times as many white blood cells are stored in the
great as 60 to 80 micrometers, a size that can barely be
marrow as circulate in the entire blood. This represents
seen with the naked eye. These cells are now called
about a 6-day supply of these cells.
macrophages, and they are extremely capable of com-
The lymphocytes are mostly stored in the various
bating intratissue disease agents.
lymphoid tissues, except for a small number that are
temporarily being transported in the blood.
White Blood Cells Enter the Tissue Spaces by Diapedesis.
As shown in Figure 33-1, megakaryocytes (cell 3)
Neutrophils and monocytes can squeeze through the
are also formed in the bone marrow. These megakary-
pores of the blood capillaries by diapedesis. That is,
ocytes fragment in the bone marrow; the small frag-
even though a pore is much smaller than a cell, a small
ments, known as platelets (or thrombocytes), then pass
portion of the cell slides through the pore at a time;
into the blood. They are very important in the initia-
the portion sliding through is momentarily constricted
tion of blood clotting.
to the size of the pore, as shown in Figure 33-2.
White Blood Cells Move Through Tissue Spaces by Ameboid
Life Span of the White Blood Cells
Motion. Both neutrophils and macrophages can move
through the tissues by ameboid motion, described in
The life of the granulocytes after being released from
Chapter 2. Some cells move at velocities as great as
the bone marrow is normally 4 to 8 hours circulating
40 mm/min, a distance as great as their own length
in the blood and another 4 to 5 days in tissues where
each minute.
they are needed. In times of serious tissue infection,
this total life span is often shortened to only a few
White Blood Cells Are Attracted to Inflamed Tissue Areas by
hours because the granulocytes proceed even more
Chemotaxis. Many different chemical substances in the
rapidly to the infected area, perform their functions,
tissues cause both neutrophils and macrophages to
and, in the process, are themselves destroyed.
move toward the source of the chemical. This phe-
The monocytes also have a short transit time, 10 to
nomenon, shown in Figure 33-2, is known as chemo-
20 hours in the blood, before wandering through the
taxis. When a tissue becomes inflamed, at least a dozen
capillary membranes into the tissues. Once in the
different products are formed that can cause chemo-
tissues, they swell to much larger sizes to become tissue
taxis toward the inflamed area. They include (1) some
macrophages, and, in this form, can live for months
of the bacterial or viral toxins, (2) degenerative prod-
unless destroyed while performing phagocytic func-
ucts of the inflamed tissues themselves, (3) several
tions. These tissue macrophages are the basis of the
reaction products of the
“complement complex”
tissue macrophage system, discussed in greater detail
(discussed in Chapter 34) activated in inflamed tissues,
later, which provides continuing defense against
and (4) several reaction products caused by plasma
infection.
clotting in the inflamed area, as well as other
Lymphocytes enter the circulatory system continu-
substances.
ally, along with drainage of lymph from the lymph
As shown in Figure 33-2, chemotaxis depends on
nodes and other lymphoid tissue. After a few hours,
the concentration gradient of the chemotactic sub-
they pass out of the blood back into the tissues by dia-
stance. The concentration is greatest near the source,
pedesis. Then, still later, they re-enter the lymph and
which directs the unidirectional movement of the
return to the blood again and again; thus, there is con-
white cells. Chemotaxis is effective up to 100 microm-
tinual circulation of lymphocytes through the body.
eters away from an inflamed tissue. Therefore, because
The lymphocytes have life spans of weeks or months;
almost no tissue area is more than 50 micrometers
this life span depends on the body’s need for these
away from a capillary, the chemotactic signal can easily
cells.
move hordes of white cells from the capillaries into the
The platelets in the blood are replaced about once
inflamed area.
every 10 days; in other words, about 30,000 platelets
are formed each day for each microliter of blood.
Phagocytosis
Neutrophils and Macrophages
The most important function of the neutrophils and
Defend Against Infections
macrophages is phagocytosis, which means cellular
ingestion of the offending agent. Phagocytes must be
It is mainly the neutrophils and tissue macrophages
selective of the material that is phagocytized; other-
that attack and destroy invading bacteria, viruses, and
wise, normal cells and structures of the body might be
432
Unit VI Blood Cells, Immunity, and Blood Clotting
ingested. Whether phagocytosis will occur depends
and digestion of the phagocytized particle begins
especially on three selective procedures.
immediately.
First, most natural structures in the tissues have
Both neutrophils and macrophages contain an
smooth surfaces, which resist phagocytosis. But if the
abundance of lysosomes filled with proteolytic
surface is rough, the likelihood of phagocytosis is
enzymes especially geared for digesting bacteria
increased.
and other foreign protein matter. The lysosomes of
Second, most natural substances of the body have
macrophages (but not of neutrophils) also contain
protective protein coats that repel the phagocytes.
large amounts of lipases, which digest the thick lipid
Conversely, most dead tissues and foreign particles
membranes possessed by some bacteria such as the
have no protective coats, which makes them subject to
tuberculosis bacillus.
phagocytosis.
Third, the immune system of the body (described in
Both Neutrophils and Macrophages Can Kill Bacteria. In
detail in Chapter 34) develops antibodies against infec-
addition to the digestion of ingested bacteria in
tious agents such as bacteria. The antibodies then
phagosomes, neutrophils and macrophages contain
adhere to the bacterial membranes and thereby make
bactericidal agents that kill most bacteria even when
the bacteria especially susceptible to phagocytosis. To
the lysosomal enzymes fail to digest them. This is espe-
do this, the antibody molecule also combines with the
cially important, because some bacteria have protec-
C3 product of the complement cascade, which is an
tive coats or other factors that prevent their
additional part of the immune system discussed in the
destruction by digestive enzymes. Much of the killing
next chapter. The C3 molecules, in turn, attach to
effect results from several powerful oxidizing agents
receptors on the phagocyte membrane, thus initiating
formed by enzymes in the membrane of the phago-
phagocytosis. This selection and phagocytosis process
some or by a special organelle called the peroxisome.
is called opsonization.
These oxidizing agents include large quantities of
superoxide
(O2-), hydrogen peroxide
(H2O2), and
Phagocytosis by Neutrophils. The neutrophils entering
hydroxyl ions (-OH-), all of which are lethal to most
the tissues are already mature cells that can immedi-
bacteria, even in small quantities. Also, one of the
ately begin phagocytosis. On approaching a particle to
lysosomal enzymes, myeloperoxidase, catalyzes the
be phagocytized, the neutrophil first attaches itself to
reaction between H2O2 and chloride ions to form
the particle and then projects pseudopodia in all direc-
hypochlorite, which is exceedingly bactericidal.
tions around the particle. The pseudopodia meet one
Some bacteria, however, notably the tuberculosis
another on the opposite side and fuse. This creates an
bacillus, have coats that are resistant to lysosomal
enclosed chamber that contains the phagocytized par-
digestion and also secrete substances that partially
ticle. Then the chamber invaginates to the inside of the
resist the killing effects of the neutrophils and
cytoplasmic cavity and breaks away from the outer cell
macrophages. These bacteria are responsible for
membrane to form a free-floating phagocytic vesicle
many of the chronic diseases, an example of which is
(also called a phagosome) inside the cytoplasm. A
tuberculosis.
single neutrophil can usually phagocytize 3 to 20 bac-
teria before the neutrophil itself becomes inactivated
and dies.
Monocyte-Macrophage Cell
System (Reticuloendothelial
Phagocytosis by Macrophages. Macrophages are the end-
stage product of monocytes that enter the tissues from
System)
the blood. When activated by the immune system as
described in Chapter 34, they are much more power-
In the preceding paragraphs, we described the
ful phagocytes than neutrophils, often capable of
macrophages mainly as mobile cells that are capable
phagocytizing as many as 100 bacteria. They also have
of wandering through the tissues. However, after
the ability to engulf much larger particles, even whole
entering the tissues and becoming macrophages,
red blood cells or, occasionally, malarial parasites,
another large portion of monocytes becomes attached
whereas neutrophils are not capable of phagocytizing
to the tissues and remains attached for months or even
particles much larger than bacteria. Also, after digest-
years until they are called on to perform specific local
ing particles, macrophages can extrude the residual
protective functions. They have the same capabilities
products and often survive and function for many
as the mobile macrophages to phagocytize large
more months.
quantities of bacteria, viruses, necrotic tissue, or other
foreign particles in the tissue. And, when appropriately
Once Phagocytized, Most Particles Are Digested by Intracellu-
stimulated, they can break away from their attach-
lar Enzymes. Once a foreign particle has been phagocy-
ments and once again become mobile macrophages
tized, lysosomes and other cytoplasmic granules in
that respond to chemotaxis and all the other stimuli
the neutrophil or macrophage immediately come in
related to the inflammatory process. Thus, the body has
contact with the phagocytic vesicle, and their mem-
a widespread “monocyte-macrophage system” in vir-
branes fuse, thereby dumping many digestive enzymes
tually all tissue areas.
and bactericidal agents into the vesicle. Thus, the
The total combination of monocytes, mobile
phagocytic vesicle now becomes a digestive vesicle,
macrophages, fixed tissue macrophages, and a few
Chapter 33
Resistance of the Body to Infection: I. Leukocytes, Granulocytes, the Monocyte-Macrophage System
433
specialized endothelial cells in the bone marrow,
Large numbers of macrophages line the lymph
spleen, and lymph nodes is called the reticuloendothe-
sinuses, and if any particles enter the sinuses by way
lial system. However, all or almost all these cells orig-
of the lymph, the macrophages phagocytize them and
inate from monocytic stem cells; therefore, the
prevent general dissemination throughout the body.
reticuloendothelial system is almost synonymous with
the monocyte-macrophage system. Because the term
Alveolar Macrophages in the Lungs. Another route by
reticuloendothelial system is much better known
which invading organisms frequently enter the body
in medical literature than the term monocyte-
is through the lungs. Large numbers of tissue
macrophage system, it should be remembered as a
macrophages are present as integral components of
generalized phagocytic system located in all tissues,
the alveolar walls. They can phagocytize particles that
especially in those tissue areas where large quantities
become entrapped in the alveoli. If the particles are
of particles, toxins, and other unwanted substances
digestible, the macrophages can also digest them and
must be destroyed.
release the digestive products into the lymph. If the
particle is not digestible, the macrophages often form
Tissue Macrophages in the Skin and Subcutaneous Tissues (His-
a “giant cell” capsule around the particle until such
tiocytes). Although the skin is mainly impregnable to
time—if ever—that it can be slowly dissolved. Such
infectious agents, this is no longer true when the skin
capsules are frequently formed around tuberculosis
is broken. When infection begins in a subcutaneous
bacilli, silica dust particles, and even carbon particles.
tissue and local inflammation ensues, local tissue
macrophages can divide in situ and form still more
Macrophages
(Kupffer Cells) in the Liver Sinusoids. Still
macrophages. Then they perform the usual functions
another favorite route by which bacteria invade the
of attacking and destroying the infectious agents, as
body is through the gastrointestinal tract. Large
described earlier.
numbers of bacteria from ingested food constantly
pass through the gastrointestinal mucosa into the
Macrophages in the Lymph Nodes. Essentially no particu-
portal blood. Before this blood enters the general cir-
late matter that enters the tissues, such as bacteria, can
culation, it passes through the sinusoids of the liver;
be absorbed directly through the capillary membranes
these sinusoids are lined with tissue macrophages
into the blood. Instead, if the particles are not
called Kupffer cells, shown in Figure 33-4. These cells
destroyed locally in the tissues, they enter the lymph
form such an effective particulate filtration system that
and flow to the lymph nodes located intermittently
almost none of the bacteria from the gastrointestinal
along the course of the lymph flow. The foreign parti-
tract succeeds in passing from the portal blood into the
cles are then trapped in these nodes in a meshwork of
general systemic circulation. Indeed, motion pictures
sinuses lined by tissue macrophages.
of phagocytosis by Kupffer cells have demonstrated
Figure 33-3 illustrates the general organization of
phagocytosis of a single bacterium in less than 1/100 of a
the lymph node, showing lymph entering through the
second.
lymph node capsule by way of afferent lymphatics,
then flowing through the nodal medullary sinuses, and
finally passing out the hilus into efferent lymphatics
that eventually empty into the venous blood.
Afferent lymphatics
Primary
nodule
Capsule
Valve
Subcapsular
sinus
Lymph in
medullary
sinuses
Germinal
center
Hilus
Medullary cord
Efferent lymphatics
Kupffer cells
Figure 33-3
Figure 33-4
Functional diagram of a lymph node. (Redrawn from Ham AW:
Kupffer cells lining the liver sinusoids, showing phagocytosis of
Histology, 6th ed. Philadelphia: JB Lippincott, 1969.) (Modified
India ink particles into the cytoplasm of the Kupffer cells.
from Gartner LP, Hiatt JL: Color Textbook of Histology, 2nd ed.
(Redrawn from Copenhaver WM, et al: Bailey’s Textbook of His-
Philadelphia, WB Saunders, 2001.)
tology, 10th ed. Baltimore: Williams & Wilkins, 1971.)
434
Unit VI Blood Cells, Immunity, and Blood Clotting
Macrophages of the Spleen and Bone Marrow. If an invad-
and cause dramatic secondary changes in the sur-
ing organism succeeds in entering the general circula-
rounding uninjured tissues. This entire complex of
tion, there are other lines of defense by the tissue
tissue changes is called inflammation.
macrophage system, especially by macrophages of the
Inflammation is characterized by (1) vasodilation of
spleen and bone marrow. In both these tissues,
the local blood vessels, with consequent excess local
macrophages have become entrapped by the reticular
blood flow; (2) increased permeability of the capillar-
meshwork of the two organs, and when foreign parti-
ies, allowing leakage of large quantities of fluid into
cles come in contact with these macrophages, they are
the interstitial spaces; (3) often clotting of the fluid in
phagocytized.
the interstitial spaces because of excessive amounts of
The spleen is similar to the lymph nodes, except that
fibrinogen and other proteins leaking from the capil-
blood, instead of lymph, flows through the tissue
laries; (4) migration of large numbers of granulocytes
spaces of the spleen. Figure 33-5 shows a small periph-
and monocytes into the tissue; and (5) swelling of the
eral segment of spleen tissue. Note that a small artery
tissue cells. Some of the many tissue products that
penetrates from the splenic capsule into the splenic
cause these reactions are histamine, bradykinin, sero-
pulp and terminates in small capillaries. The capillar-
tonin, prostaglandins, several different reaction prod-
ies are highly porous, allowing whole blood to pass out
ucts of the complement system (described in Chapter
of the capillaries into cords of red pulp. The blood then
34), reaction products of the blood clotting system, and
gradually squeezes through the trabecular meshwork
multiple substances called lymphokines that are
of these cords and eventually returns to the circulation
released by sensitized T cells (part of the immune
through the endothelial walls of the venous sinuses.
system; also discussed in Chapter 34). Several of these
The trabeculae of the red pulp are lined with vast
substances strongly activate the macrophage system,
numbers of macrophages, and the venous sinuses are
and within a few hours, the macrophages begin to
also lined with macrophages. This peculiar passage of
devour the destroyed tissues. But at times, the
blood through the cords of the red pulp provides an
macrophages also further injure the still-living tissue
exceptional means of phagocytizing unwanted debris
cells.
in the blood, including especially old and abnormal red
blood cells.
“Walling-Off” Effect of Inflammation. One of the first
results of inflammation is to “wall off ” the area of
injury from the remaining tissues. The tissue spaces
Inflammation: Role of
and the lymphatics in the inflamed area are blocked
Neutrophils and Macrophages
by fibrinogen clots so that after a while, fluid barely
flows through the spaces. This walling-off process
Inflammation
delays the spread of bacteria or toxic products.
The intensity of the inflammatory process is usually
When tissue injury occurs, whether caused by bacteria,
proportional to the degree of tissue injury. For
trauma, chemicals, heat, or any other phenomenon,
instance, when staphylococci invade tissues, they
multiple substances are released by the injured tissues
release extremely lethal cellular toxins. As a result,
inflammation develops rapidly—indeed, much more
rapidly than the staphylococci themselves can multi-
ply and spread. Therefore, local staphylococcal in-
fection is characteristically walled off rapidly and
prevented from spreading through the body. Strepto-
cocci, in contrast, do not cause such intense local tissue
destruction. Therefore, the walling-off process devel-
ops slowly over many hours, while many streptococci
Pulp
reproduce and migrate. As a result, streptococci often
have a far greater tendency to spread through the
Capillaries
body and cause death than do staphylococci, even
though staphylococci are far more destructive to the
Venous sinuses
tissues.
Vein
Artery
Macrophage and Neutrophil
Responses During Inflammation
Tissue Macrophage Is a First Line of Defense Against Infection.
Within minutes after inflammation begins, the
Figure 33-5
macrophages already present in the tissues, whether
histiocytes in the subcutaneous tissues, alveolar
Functional structures of the spleen.
(Modified from Bloom W,
macrophages in the lungs, microglia in the brain, or
Fawcett DW: A Textbook of Histology, 10th ed. Philadelphia: WB
Saunders, 1975.)
others, immediately begin their phagocytic actions.
Chapter 33
Resistance of the Body to Infection: I. Leukocytes, Granulocytes, the Monocyte-Macrophage System
435
When activated by the products of infection and
come to dominate the phagocytic cells of the inflamed
inflammation, the first effect is rapid enlargement of
area because of greatly increased bone marrow pro-
each of these cells. Next, many of the previously sessile
duction of new monocytes, as explained later.
macrophages break loose from their attachments
As already pointed out, macrophages can phagocy-
and become mobile, forming the first line of defense
tize far more bacteria (about five times as many) and
against infection during the first hour or so. The
far larger particles, including even neutrophils them-
numbers of these early mobilized macrophages often
selves and large quantities of necrotic tissue, than can
are not great, but they are lifesaving.
neutrophils. Also, the macrophages play an important
role in initiating the development of antibodies, as we
Neutrophil Invasion of the Inflamed Area Is a Second Line of
discuss in Chapter 34.
Defense. Within the first hour or so after inflammation
begins, large numbers of neutrophils begin to invade
Increased Production of Granulocytes and Monocytes by the
the inflamed area from the blood. This is caused by
Bone Marrow Is a Fourth Line of Defense. The fourth line of
products from the inflamed tissues that initiate the fol-
defense is greatly increased production of both gran-
lowing reactions: (1) They alter the inside surface of
ulocytes and monocytes by the bone marrow. This
the capillary endothelium, causing neutrophils to stick
results from stimulation of the granulocytic and mono-
to the capillary walls in the inflamed area. This effect
cytic progenitor cells of the marrow. However, it
is called margination and is shown in Figure 33-2. (2)
takes 3 to 4 days before newly formed granulocytes
They cause the intercellular attachments between the
and monocytes reach the stage of leaving the bone
endothelial cells of the capillaries and small venules
marrow. If the stimulus from the inflamed tissue con-
to loosen, allowing openings large enough for neu-
tinues, the bone marrow can continue to produce these
trophils to pass by diapedesis directly from the blood
cells in tremendous quantities for months and even
into the tissue spaces. (3) Other products of inflam-
years, sometimes at a rate 20 to 50 times normal.
mation then cause chemotaxis of the neutrophils
toward the injured tissues, as explained earlier.
Feedback Control of the Macrophage and
Thus, within several hours after tissue damage
Neutrophil Responses
begins, the area becomes well supplied with neu-
Although more than two dozen factors have been
trophils. Because the blood neutrophils are already
implicated in control of the macrophage response to
mature cells, they are ready to immediately begin their
inflammation, five of these are believed to play domi-
scavenger functions for killing bacteria and removing
nant roles. They are shown in Figure 33-6 and consist
foreign matter.
of (1) tumor necrosis factor (TNF), (2) interleukin-1
(IL-1),
(3) granulocyte-monocyte colony-stimulating
Acute Increase in Number of Neutrophils in the Blood—“Neu-
factor (GM-CSF), (4) granulocyte colony-stimulating
trophilia.” Also within a few hours after the onset of
factor (G-CSF), and (5) monocyte colony-stimulating
acute, severe inflammation, the number of neutrophils
factor (M-CSF). These factors are formed by activated
in the blood sometimes increases fourfold to five-
macrophage cells in the inflamed tissues and in smaller
fold—from a normal of 4000 to 5000 to 15,000 to
quantities by other inflamed tissue cells.
25,000 neutrophils per microliter. This is called neu-
The cause of the increased production of granulo-
trophilia, which means an increase in the number of
cytes and monocytes by the bone marrow is mainly
neutrophils in the blood. Neutrophilia is caused by
the three colony-stimulating factors, one of which,
products of inflammation that enter the blood stream,
GM-CSF, stimulates both granulocyte and monocyte
are transported to the bone marrow, and there act on
production; the other two, G-CSF and M-CSF, stimu-
the stored neutrophils of the marrow to mobilize these
late granulocyte and monocyte production, respec-
into the circulating blood. This makes even more neu-
tively. This combination of TNF, IL-1, and colony-stim-
trophils available to the inflamed tissue area.
ulating factors provides a powerful feedback mecha-
nism that begins with tissue inflammation and
Second Macrophage Invasion into the Inflamed Tissue Is a Third
proceeds to formation of large numbers of defensive
Line of Defense. Along with the invasion of neutrophils,
white blood cells that help remove the cause of the
monocytes from the blood enter the inflamed tissue
inflammation.
and enlarge to become macrophages. However, the
number of monocytes in the circulating blood is low:
Formation of Pus
also, the storage pool of monocytes in the bone
When neutrophils and macrophages engulf large
marrow is much less than that of neutrophils. There-
numbers of bacteria and necrotic tissue, essentially
fore, the buildup of macrophages in the inflamed tissue
all the neutrophils and many, if not most, of the
area is much slower than that of neutrophils, requiring
macrophages eventually die. After several days, a
several days to become effective. Furthermore, even
cavity is often excavated in the inflamed tissues that
after invading the inflamed tissue, monocytes are still
contains varying portions of necrotic tissue, dead neu-
immature cells, requiring 8 hours or more to swell to
trophils, dead macrophages, and tissue fluid. This
much larger sizes and develop tremendous quantities
mixture is commonly known as pus. After the infec-
of lysosomes; only then do they acquire the full capac-
tion has been suppressed, the dead cells and necrotic
ity of tissue macrophages for phagocytosis. Yet, after
tissue in the pus gradually autolyze over a period of
several days to several weeks, the macrophages finally
days, and the end products are eventually absorbed
436
Unit VI Blood Cells, Immunity, and Blood Clotting
INFLAMMATION
and kill many of them. They do so in several ways: (1)
by releasing hydrolytic enzymes from their granules,
which are modified lysosomes; (2) probably by also
releasing highly reactive forms of oxygen that are
especially lethal to parasites; and (3) by releasing from
the granules a highly larvacidal polypeptide called
Activated
TNF
major basic protein.
macrophage
IL-1
In a few areas of the world, another parasitic disease
that causes eosinophilia is trichinosis. This results from
invasion of the body’s muscles by the Trichinella par-
Endothelial cells,
asite (“pork worm”) after a person eats undercooked
fibroblasts,
infested pork.
TNF
lymphocytes
Eosinophils also have a special propensity to collect
IL-1
in tissues in which allergic reactions occur, such as in
GM-CSF
the peribronchial tissues of the lungs in people with
G-CSF
GM-CSF
asthma and in the skin after allergic skin reactions.
M-CSF
G-CSF
This is caused at least partly by the fact that many mast
M-CSF
cells and basophils participate in allergic reactions, as
we discuss in the next paragraph. The mast cells and
basophils release an eosinophil chemotactic factor that
causes eosinophils to migrate toward the inflamed
allergic tissue. The eosinophils are believed to detox-
Bone marrow
ify some of the inflammation-inducing substances
released by the mast cells and basophils and probably
also to phagocytize and destroy allergen-antibody
complexes, thus preventing excess spread of the local
inflammatory process.
Granulocytes
Monocytes/macrophages
Basophils
Figure 33-6
Control of bone marrow production of granulocytes and mono-
The basophils in the circulating blood are similar to
cyte-macrophages in response to multiple growth factors released
the large tissue mast cells located immediately outside
from activated macrophages in an inflamed tissue. G-CSF, gran-
many of the capillaries in the body. Both mast cells and
ulocyte colony-stimulating factor; GM-CSF, granulocyte-monocyte
basophils liberate heparin into the blood, a substance
colony-stimulating factor; IL-1, interleukin-1; M-CSF, monocyte
colony-stimulating factor; TNF, tumor necrosis factor.
that can prevent blood coagulation.
The mast cells and basophils also release histamine,
as well as smaller quantities of bradykinin and
into the surrounding tissues and lymph until most of
serotonin. Indeed, it is mainly the mast cells in
the evidence of tissue damage is gone.
inflamed tissues that release these substances during
inflammation.
The mast cells and basophils play an exceedingly
Eosinophils
important role in some types of allergic reactions
because the type of antibody that causes allergic reac-
The eosinophils normally constitute about 2 per cent
tions, the immunoglobulin E (IgE) type (see Chapter
of all the blood leukocytes. Eosinophils are weak
34), has a special propensity to become attached to
phagocytes, and they exhibit chemotaxis, but in com-
mast cells and basophils. Then, when the specific
parison with the neutrophils, it is doubtful that the
antigen for the specific IgE antibody subsequently
eosinophils are significant in protecting against the
reacts with the antibody, the resulting attachment of
usual types of infection.
antigen to antibody causes the mast cell or basophil
Eosinophils, however, are often produced in large
to rupture and release exceedingly large quantities of
numbers in people with parasitic infections, and they
histamine, bradykinin, serotonin, heparin, slow-reacting
migrate in large numbers into tissues diseased by par-
substance of anaphylaxis, and a number of lysosomal
asites. Although most parasites are too large to be
enzymes. These cause local vascular and tissue reac-
phagocytized by eosinophils or any other phagocytic
tions that cause many, if not most, of the allergic man-
cells, eosinophils attach themselves to the parasites by
ifestations. These reactions are discussed in greater
way of special surface molecules and release sub-
detail in Chapter 34.
stances that kill many of the parasites. For instance,
one of the most widespread infections is schistosomi-
asis, a parasitic infection found in as many as one third
Leukopenia
of the population of some Third World countries; the
parasite can invade any part of the body. Eosinophils
A clinical condition known as leukopenia occasionally
attach themselves to the juvenile forms of the parasite
occurs in which the bone marrow produces very few
Chapter 33
Resistance of the Body to Infection: I. Leukocytes, Granulocytes, the Monocyte-Macrophage System
437
white blood cells, leaving the body unprotected against
extramedullary tissues—especially in the lymph nodes,
many bacteria and other agents that might invade the
spleen, and liver.
tissues.
In myelogenous leukemia, the cancerous process
Normally, the human body lives in symbiosis with
occasionally produces partially differentiated cells,
many bacteria, because all the mucous membranes of
resulting in what might be called neutrophilic
the body are constantly exposed to large numbers of
leukemia, eosinophilic leukemia, basophilic leukemia,
bacteria. The mouth almost always contains various
or monocytic leukemia. More frequently, however, the
spirochetal, pneumococcal, and streptococcal bacteria,
leukemia cells are bizarre and undifferentiated and
and these same bacteria are present to a lesser extent
not identical to any of the normal white blood cells.
in the entire respiratory tract. The distal gastrointesti-
Usually, the more undifferentiated the cell, the more
nal tract is especially loaded with colon bacilli. Fur-
acute is the leukemia, often leading to death within a
thermore, one can always find bacteria on the surfaces
few months if untreated. With some of the more dif-
of the eyes, urethra, and vagina. Any decrease in the
ferentiated cells, the process can be chronic, some-
number of white blood cells immediately allows inva-
times developing slowly over 10 to 20 years. Leukemic
sion of adjacent tissues by bacteria that are already
cells, especially the very undifferentiated cells, are
present.
usually nonfunctional for providing the normal pro-
Within 2 days after the bone marrow stops produc-
tection against infection.
ing white blood cells, ulcers may appear in the mouth
and colon, or the person might develop some form of
severe respiratory infection. Bacteria from the ulcers
Effects of Leukemia on the Body
rapidly invade surrounding tissues and the blood.
Without treatment, death often ensues in less than a
The first effect of leukemia is metastatic growth of
week after acute total leukopenia begins.
leukemic cells in abnormal areas of the body.
Irradiation of the body by x-rays or gamma rays, or
Leukemic cells from the bone marrow may reproduce
exposure to drugs and chemicals that contain benzene
so greatly that they invade the surrounding bone,
or anthracene nuclei, is likely to cause aplasia of the
causing pain and, eventually, a tendency for bones to
bone marrow. Indeed, some common drugs, such as
fracture easily.
chloramphenicol (an antibiotic), thiouracil (used to
Almost all leukemias eventually spread to the
treat thyrotoxicosis), and even various barbiturate
spleen, lymph nodes, liver, and other vascular regions,
hypnotics, on very rare occasions cause leukopenia,
regardless of whether the origin of the leukemia is in
thus setting off the entire infectious sequence of this
the bone marrow or the lymph nodes. Common effects
malady.
in leukemia are the development of infection, severe
After moderate irradiation injury to the bone
anemia, and a bleeding tendency caused by thrombo-
marrow, some stem cells, myeloblasts, and hemocyto-
cytopenia
(lack of platelets). These effects result
blasts may remain undestroyed in the marrow and are
mainly from displacement of the normal bone marrow
capable of regenerating the bone marrow, provided
and lymphoid cells by the nonfunctional leukemic
sufficient time is available. A patient properly treated
cells.
with transfusions, plus antibiotics and other drugs to
Finally, perhaps the most important effect of
ward off infection, usually develops enough new bone
leukemia on the body is excessive use of metabolic
marrow within weeks to months for blood cell con-
substrates by the growing cancerous cells. The
centrations to return to normal.
leukemic tissues reproduce new cells so rapidly that
tremendous demands are made on the body reserves
for foodstuffs, specific amino acids, and vitamins. Con-
The Leukemias
sequently, the energy of the patient is greatly depleted,
and excessive utilization of amino acids by the
Uncontrolled production of white blood cells can be
leukemic cells causes especially rapid deterioration of
caused by cancerous mutation of a myelogenous or
the normal protein tissues of the body. Thus, while the
lymphogenous cell. This causes leukemia, which is
leukemic tissues grow, other tissues become debili-
usually characterized by greatly increased numbers
tated. After metabolic starvation has continued long
of abnormal white blood cells in the circulating
enough, this alone is sufficient to cause death.
blood.
References
Types of Leukemia. Leukemias are divided into two
general types: lymphocytic leukemias and myeloge-
Alexander JS, Granger DN: Lymphocyte trafficking medi-
nous leukemias. The lymphocytic leukemias are caused
ated by vascular adhesion protein-1: implications for
by cancerous production of lymphoid cells, usually
immune targeting and cardiovascular disease. Circ Res
beginning in a lymph node or other lymphocytic tissue
86:1190, 2000.
and spreading to other areas of the body. The second
Blander JM, Medzhitov R: Regulation of phagosome matu-
type of leukemia, myelogenous leukemia, begins by
ration by signals from toll-like receptors. Science 304:1014,
cancerous production of young myelogenous cells in
2004.
the bone marrow and then spreads throughout the
Bleakley M, Riddell SR: Molecules and mechanisms of the
body so that white blood cells are produced in many
graft-versus-leukaemia effect. Nat Rev Cancer 4:371, 2004.
438
Unit VI Blood Cells, Immunity, and Blood Clotting
Ferrajoli A, O’Brien SM: Treatment of chronic lymphocytic
Ossovskaya VS, Bunnett NW: Protease-activated receptors:
leukemia. Semin Oncol 31(Suppl 4):60, 2004.
contribution to physiology and disease. Physiol Rev
Heasman SJ, Giles KM, Ward C, et al: Glucocorticoid-
84:579, 2004.
mediated regulation of granulocyte apoptosis and
Park JE, Barbul A: Understanding the role of immune reg-
macrophage phagocytosis of apoptotic cells: implications
ulation in wound healing. Am J Surg 187:11S, 2004.
for the resolution of inflammation. J Endocrinol 178:29,
Pui CH, Relling MV, Downing JR: Acute lymphoblastic
2003.
leukemia. N Engl J Med 350:1535, 2004.
Kunkel EJ, Butcher EC: Plasma-cell homing. Nat Rev
Roufosse F, Cogan E, Goldman M: The hypereosinophilic
Immunol 3:822, 2003.
syndrome revisited. Annu Rev Med 54:169, 2003.
Kvietys PR, Sandig M: Neutrophil diapedesis: paracellular or
von Herrath MG, Harrison LC: Regulatory lymphocytes:
transcellular? News Physiol Sci 16:15, 2001.
antigen-induced regulatory T cells in autoimmunity. Nat
Ley K, Kansas GS: Selectins in T-cell recruitment to non-
Rev Immunol 3:223, 2003.
lymphoid tissues and sites of inflammation. Nat Rev
Werner S, Grose R: Regulation of wound healing by growth
Immunol 4:325, 2004.
factors and cytokines. Physiol Rev 83:835, 2003.
Moser B, Wolf M, Walz A, Loetscher P: Chemokines: multi-
Zullig S, Hengartner MO: Cell biology: tickling macro-
ple levels of leukocyte migration control. Trends Immunol
phages, a serious business. Science 304:1123, 2004.
25:75, 2004.
Muller WA: Leukocyte-endothelial-cell interactions in
leukocyte transmigration and the inflammatory response.
Trends Immunol 24:327, 2003.
C H A P T E R
3
4
Resistance of the Body to
Infection: II. Immunity and Allergy
Innate Immunity
The human body has the ability to resist almost all
types of organisms or toxins that tend to damage the
tissues and organs. This capability is called immu-
nity. Much of immunity is acquired immunity that
does not develop until after the body is first
attacked by a bacterium, virus, or toxin, often
requiring weeks or months to develop the immunity. An additional portion of
immunity results from general processes, rather than from processes directed
at specific disease organisms. This is called innate immunity. It includes the
following:
1. Phagocytosis of bacteria and other invaders by white blood cells and cells
of the tissue macrophage system, as described in Chapter 33.
2. Destruction of swallowed organisms by the acid secretions of the stomach
and the digestive enzymes.
3. Resistance of the skin to invasion by organisms.
4. Presence in the blood of certain chemical compounds that attach to
foreign organisms or toxins and destroy them. Some of these compounds
are (1) lysozyme, a mucolytic polysaccharide that attacks bacteria and
causes them to dissolute; (2) basic polypeptides, which react with and
inactivate certain types of gram-positive bacteria; (3) the complement
complex that is described later, a system of about 20 proteins that can be
activated in various ways to destroy bacteria; and (4) natural killer
lymphocytes that can recognize and destroy foreign cells, tumor cells, and
even some infected cells.
This innate immunity makes the human body resistant to such diseases as
some paralytic viral infections of animals, hog cholera, cattle plague, and dis-
temper—a viral disease that kills a large percentage of dogs that become
afflicted with it. Conversely, many lower animals are resistant or even immune
to many human diseases, such as poliomyelitis, mumps, human cholera, measles,
and syphilis, which are very damaging or even lethal to human beings.
Acquired (Adaptive) Immunity
In addition to its generalized innate immunity, the human body has the ability
to develop extremely powerful specific immunity against individual invading
agents such as lethal bacteria, viruses, toxins, and even foreign tissues from other
animals. This is called acquired or adaptive immunity. Acquired immunity is
caused by a special immune system that forms antibodies and/or activated lym-
phocytes that attack and destroy the specific invading organism or toxin. It is
with this acquired immunity mechanism and some of its associated reactions—
especially the allergies—that this chapter is concerned.
Acquired immunity can often bestow extreme protection. For instance,
certain toxins, such as the paralytic botulinum toxin or the tetanizing toxin of
tetanus, can be protected against in doses as high as 100,000 times the amount
that would be lethal without immunity. This is the reason the treatment process
known as immunization is so important in protecting human beings against
439
440
Unit VI Blood Cells, Immunity, and Blood Clotting
disease and against toxins, as explained in the course
the gastrointestinal tract, thymus, and bone marrow.
of this chapter.
The lymphoid tissue is distributed advantageously in
the body to intercept invading organisms or toxins
before they can spread too widely.
Basic Types of Acquired Immunity
In most instances, the invading agent first enters the
tissue fluids and then is carried by way of lymph vessels
Two basic but closely allied types of acquired immu-
to the lymph node or other lymphoid tissue. For
nity occur in the body. In one of these the body
instance, the lymphoid tissue of the gastrointestinal
develops circulating antibodies, which are globulin
walls is exposed immediately to antigens invading
molecules in the blood plasma that are capable of
from the gut. The lymphoid tissue of the throat and
attacking the invading agent. This type of immunity is
pharynx (the tonsils and adenoids) is well located to
called humoral immunity or B-cell immunity (because
intercept antigens that enter by way of the upper res-
B lymphocytes produce the antibodies). The second
piratory tract. The lymphoid tissue in the lymph nodes
type of acquired immunity is achieved through the
is exposed to antigens that invade the peripheral
formation of large numbers of activated T lymphocytes
tissues of the body. And, finally, the lymphoid tissue of
that are specifically crafted in the lymph nodes to
the spleen, thymus, and bone marrow plays the specific
destroy the foreign agent. This type of immunity is
role of intercepting antigenic agents that have suc-
called cell-mediated immunity or T-cell immunity
ceeded in reaching the circulating blood.
(because the activated lymphocytes are T lympho-
cytes). We shall see shortly that both the antibodies
Two Types of Lymphocytes Promote “Cell-Mediated” Immunity
and the activated lymphocytes are formed in the lym-
or
“Humoral” Immunity—the T and the B Lymphocytes.
phoid tissues of the body. Let us discuss the initiation
Although most lymphocytes in normal lymphoid
of the immune process by antigens.
tissue look alike when studied under the microscope,
these cells are distinctly divided into two major
populations. One of the populations, the T lympho-
Both Types of Acquired Immunity Are
cytes, is responsible for forming the activated lympho-
Initiated by Antigens
cytes that provide “cell-mediated” immunity, and the
other population, the B lymphocytes, is responsible
Because acquired immunity does not develop until
for forming antibodies that provide
“humoral”
after invasion by a foreign organism or toxin, it is clear
immunity.
that the body must have some mechanism for recog-
Both types of lymphocytes are derived originally in
nizing this invasion. Each toxin or each type of organ-
the embryo from pluripotent hematopoietic stem cells
ism almost always contains one or more specific
that form lymphocytes as one of their most important
chemical compounds in its makeup that are different
offspring as they differentiate. Almost all of the lym-
from all other compounds. In general, these are pro-
phocytes that are formed eventually end up in the
teins or large polysaccharides, and it is they that initi-
lymphoid tissue, but before doing so, they are further
ate the acquired immunity. These substances are called
differentiated or “preprocessed” in the following ways.
antigens (antibody generations).
The lymphocytes that are destined to eventually
For a substance to be antigenic, it usually must have
form activated T lymphocytes first migrate to and are
a high molecular weight, 8000 or greater. Furthermore,
preprocessed in the thymus gland, and thus they are
the process of antigenicity usually depends on regu-
called
“T” lymphocytes to designate the role of
larly recurring molecular groups, called epitopes, on
the thymus. They are responsible for cell-mediated
the surface of the large molecule. This also explains
immunity.
why proteins and large polysaccharides are almost
The other population of lymphocytes—the B lym-
always antigenic, because both of these have this ster-
phocytes that are destined to form antibodies—are
eochemical characteristic.
preprocessed in the liver during midfetal life and in the
bone marrow in late fetal life and after birth. This pop-
ulation of cells was first discovered in birds, which have
Lymphocytes Are Responsible for
a special preprocessing organ called the bursa of Fabri-
Acquired Immunity
cius. For this reason, these lymphocytes are called “B”
lymphocytes to designate the role of the bursa, and
Acquired immunity is the product of the body’s lym-
they are responsible for humoral immunity. Figure
phocytes. In people who have a genetic lack of lym-
34-1 shows the two lymphocyte systems for the for-
phocytes or whose lymphocytes have been destroyed
mation, respectively, of (1) the activated T lympho-
by radiation or chemicals, no acquired immunity can
cytes and (2) the antibodies.
develop. And within days after birth, such a person
dies of fulminating bacterial infection unless treated
by heroic measures. Therefore, it is clear that the lym-
Preprocessing of the T and B
phocytes are essential to survival of the human being.
Lymphocytes
The lymphocytes are located most extensively in the
lymph nodes, but they are also found in special lym-
Although all lymphocytes in the body originate from
phoid tissues such as the spleen, submucosal areas of
lymphocyte-committed stem cells of the embryo, these
Chapter 34
Resistance of the Body to Infection: II. Immunity and Allergy
441
Cell-Mediated Immunity
Thymus
Activated T
Antigen
lymphocytes
T lymphocytes
Lymph node
B lymphocytes
Figure 34-1
Stem cell
Formation of antibodies and sen-
Antigen
Plasma
Antibodies
sitized lymphocytes by a lymph
cells
node in response to antigens.
This figure also shows the origin
Fetal liver,
of thymic (T) and bursal (B) lym-
bone marrow
phocytes that respectively are
responsible for the cell-mediated
Humoral Immunity
and humoral immune processes.
stem cells themselves are incapable of forming directly
Most of the preprocessing of T lymphocytes in the
either activated T lymphocytes or antibodies. Before
thymus occurs shortly before birth of a baby and for a
they can do so, they must be further differentiated in
few months after birth. Beyond this period, removal of
appropriate processing areas as follows.
the thymus gland diminishes (but does not eliminate)
the T-lymphocytic immune system. However, removal
Thymus Gland Preprocesses the T Lymphocytes. The T lym-
of the thymus several months before birth can prevent
phocytes, after origination in the bone marrow, first
development of all cell-mediated immunity. Because
migrate to the thymus gland. Here they divide rapidly
this cellular type of immunity is mainly responsible for
and at the same time develop extreme diversity for
rejection of transplanted organs, such as hearts and
reacting against different specific antigens. That is, one
kidneys, one can transplant organs with much less like-
thymic lymphocyte develops specific reactivity against
lihood of rejection if the thymus is removed from an
one antigen. Then the next lymphocyte develops speci-
animal a reasonable time before its birth.
ficity against another antigen. This continues until
there are thousands of different types of thymic lym-
Liver and Bone Marrow Preprocess the B Lymphocytes. Much
phocytes with specific reactivities against many thou-
less is known about the details for preprocessing B
sands of different antigens. These different types of
lymphocytes than for preprocessing T lymphocytes. In
preprocessed T lymphocytes now leave the thymus
the human being, B lymphocytes are known to be pre-
and spread by way of the blood throughout the body
processed in the liver during midfetal life and in the
to lodge in lymphoid tissue everywhere.
bone marrow during late fetal life and after birth.
The thymus also makes certain that any T lympho-
B lymphocytes are different from T lymphocytes in
cytes leaving the thymus will not react against proteins
two ways: First, instead of the whole cell developing
or other antigens that are present in the body’s
reactivity against the antigen, as occurs for the T lym-
own tissues; otherwise, the T lymphocytes would be
phocytes, the B lymphocytes actively secrete antibod-
lethal to the person’s own body in only a few days.
ies that are the reactive agents. These agents are large
The thymus selects which T lymphocytes will be
protein molecules that are capable of combining with
released by first mixing them with virtually all the spe-
and destroying the antigenic substance, which is
cific “self-antigens” from the body’s own tissues. If a T
explained elsewhere in this chapter and in Chapter
lymphocyte reacts, it is destroyed and phagocytized
33. Second, the B lymphocytes have even greater
instead of being released. This happens to as many as
diversity than the T lymphocytes, thus forming many
90 per cent of the cells. Thus, the only cells that are
millions of types of B-lymphocyte antibodies with dif-
finally released are those that are nonreactive against
ferent specific reactivities. After preprocessing, the B
the body’s own antigens—they react only against anti-
lymphocytes, like the T lymphocytes, migrate to lym-
gens from an outside source, such as from a bacterium,
phoid tissue throughout the body, where they lodge
a toxin, or even transplanted tissue from another
near but slightly removed from the T-lymphocyte
person.
areas.
442
Unit VI Blood Cells, Immunity, and Blood Clotting
T Lymphocytes and B-Lymphocyte
in random combinations, in this way finally forming
whole genes.
Antibodies React Highly Specifically
Because there are several hundred types of gene
Against Specific Antigens—Role of
segments, as well as millions of different combinations
Lymphocyte Clones
in which the segments can be arranged in single cells,
one can understand the millions of different cell gene
When specific antigens come in contact with T and B
types that can occur. For each functional T or B lym-
lymphocytes in the lymphoid tissue, certain of the T
phocyte that is finally formed, the gene structure codes
lymphocytes become activated to form activated T
for only a single antigen specificity. These mature cells
cells, and certain of the B lymphocytes become acti-
then become the highly specific T and B cells that
vated to form antibodies. The activated T cells and
spread to and populate the lymphoid tissue.
antibodies in turn react highly specifically against the
particular types of antigens that initiated their devel-
Mechanism for Activating a Clone
opment. The mechanism of this specificity is the
of Lymphocytes
following.
Each clone of lymphocytes is responsive to only a
single type of antigen (or to several similar antigens
Millions of Specific Types of Lymphocytes Are Stored in the
that have almost exactly the same stereochemical
Lymphoid Tissue. Millions of different types of pre-
characteristics). The reason for this is the following: In
formed B lymphocytes and preformed T lymphocytes
the case of the B lymphocytes, each of these has on the
that are capable of forming highly specific types of
surface of its cell membrane about 100,000 antibody
antibodies or T cells have been stored in the lymph
molecules that will react highly specifically with
tissue, as explained earlier. Each of these preformed
only one specific type of antigen. Therefore, when
lymphocytes is capable of forming only one type of
the appropriate antigen comes along, it immediately
antibody or one type of T cell with a single type of
attaches to the antibody in the cell membrane;
specificity. And only the specific type of antigen with
this leads to the activation process, which we describe
which it can react can activate it. Once the specific lym-
in more detail subsequently. In the case of the T
phocyte is activated by its antigen, it reproduces wildly,
lymphocytes, molecules similar to antibodies, called
forming tremendous numbers of duplicate lympho-
surface receptor proteins (or T-cell markers), are on
cytes. If it is a B lymphocyte, its progeny will eventu-
the surface of the T-cell membrane, and these, too,
ally secrete the specific type of antibody that then
are highly specific for one specified activating
circulates throughout the body. If it is a T lymphocyte,
antigen.
its progeny are specific sensitized T cells that are
released into the lymph and then carried to the blood
and circulated through all the tissue fluids and back
Role of Macrophages in the Activation Process. Aside from
into the lymph, sometimes circulating around and
the lymphocytes in lymphoid tissue, literally millions
around in this circuit for months or years.
of macrophages are also present in the same tissue.
All the different lymphocytes that are capable of
These line the sinusoids of the lymph nodes, spleen,
forming one specificity of antibody or T cell are called
and other lymphoid tissue, and they lie in apposition
a clone of lymphocytes. That is, the lymphocytes in
to many of the lymph node lymphocytes. Most invad-
each clone are alike and are derived originally from
ing organisms are first phagocytized and partially
one or a few early lymphocytes of its specific type.
digested by the macrophages, and the antigenic prod-
ucts are liberated into the macrophage cytosol. The
macrophages then pass these antigens by cell-to-cell
Origin of the Many Clones
contact directly to the lymphocytes, thus leading to
activation of the specified lymphocytic clones. The
of Lymphocytes
macrophages, in addition, secrete a special activating
substance that promotes still further growth and
Only several hundred to a few thousand genes code
reproduction of the specific lymphocytes. This sub-
for the millions of different types of antibodies and T
stance is called interleukin-1.
lymphocytes. At first, it was a mystery how it was pos-
sible for so few genes to code for the millions of dif-
ferent specificities of antibody molecules or T cells that
Role of the T Cells in Activation of the B Lymphocytes. Most
can be produced by the lymphoid tissue, especially
antigens activate both T lymphocytes and B lympho-
when one considers that a single gene is usually nec-
cytes at the same time. Some of the T cells that are
essary for the formation of each different type of
formed, called helper cells, secrete specific substances
protein. This mystery has now been solved.
(collectively called lymphokines) that activate the
The whole gene for forming each type of T cell or B
specific B lymphocytes. Indeed, without the aid of
cell is never present in the original stem cells from
these helper T cells, the quantity of antibodies formed
which the functional immune cells are formed. Instead,
by the B lymphocytes is usually slight. We will discuss
there are only “gene segments”—actually, hundreds of
this cooperative relationship between helper T cells
such segments—but not whole genes. During prepro-
and B cells again after we have an opportunity
cessing of the respective T- and B-cell lymphocytes,
to describe the mechanisms of the T-cell system of
these gene segments become mixed with one another
immunity.
Chapter 34
Resistance of the Body to Infection: II. Immunity and Allergy
443
Specific Attributes of the
B-Lymphocyte System—Humoral
Immunity and the Antibodies
128
Secondary
Primary
Formation of Antibodies by Plasma Cells. Before exposure
64
to a specific antigen, the clones of B lymphocytes
remain dormant in the lymphoid tissue. On entry of a
32
foreign antigen, macrophages in the lymphoid tissue
phagocytize the antigen and then present it to adjacent
16
B lymphocytes. In addition, the antigen is presented to
T cells at the same time, and activated helper T cells
8
are formed. These helper cells also contribute to
extreme activation of the B lymphocytes, as we discuss
more fully later.
0
Those B lymphocytes specific for the antigen imme-
0
2
4
6
8
diately enlarge and take on the appearance of
Weeks
lymphoblasts. Some of the lymphoblasts further dif-
ferentiate to form plasmablasts, which are precursors
of plasma cells. In the plasmablasts, the cytoplasm
Figure 34-2
expands and the rough endoplasmic reticulum vastly
proliferates. The plasmablasts then begin to divide at
Time course of the antibody response in the circulating blood to
a rate of about once every 10 hours for about nine divi-
a primary injection of antigen and to a secondary injection several
sions, giving in 4 days a total population of about 500
months later.
cells for each original plasmablast. The mature plasma
cell then produces gamma globulin antibodies at an
extremely rapid rate—about
2000 molecules per
second for each plasma cell. In turn, the antibodies are
increased potency and duration of the secondary
secreted into the lymph and carried to the circulating
response explain why immunization is usually accom-
blood. This process continues for several days or weeks
plished by injecting antigen in multiple doses with
until finally exhaustion and death of the plasma cells
periods of several weeks or several months between
occur.
injections.
Formation of “Memory” Cells—Difference Between Primary
Nature of the Antibodies
Response and Secondary Response. A few of the lym-
The antibodies are gamma globulins called immuno-
phoblasts formed by activation of a clone of B lym-
globulins (abbreviated as Ig), and they have molecu-
phocytes do not go on to form plasma cells but instead
lar weights between 160,000 and 970,000. They usually
form moderate numbers of new B lymphocytes similar
constitute about 20 per cent of all the plasma proteins.
to those of the original clone. In other words, the B-
All the immunoglobulins are composed of combi-
cell population of the specifically activated clone
nations of light and heavy polypeptide chains. Most are
becomes greatly enhanced, and the new B lympho-
a combination of two light and two heavy chains,
cytes are added to the original lymphocytes of the
as shown in Figure
34-3. However, some of the
same clone. They also circulate throughout the body to
immunoglobulins have combinations of as many as
populate all the lymphoid tissue; immunologically,
10 heavy and 10 light chains, which gives rise to
however, they remain dormant until activated once
high-molecular-weight immunoglobulins. Yet, in all
again by a new quantity of the same antigen. These
immunoglobulins, each heavy chain is paralleled by a
lymphocytes are called memory cells. Subsequent
light chain at one of its ends, thus forming a heavy-light
exposure to the same antigen will cause a much more
pair, and there are always at least 2 and as many as 10
rapid and much more potent antibody response this
such pairs in each immunoglobulin molecule.
second time around, because there are many more
Figure 34-3 shows by the circled area a designated
memory cells than there were original B lymphocytes
end of each light and heavy chain, called the variable
of the specific clone.
portion; the remainder of each chain is called the con-
Figure
34-2 shows the differences between the
stant portion. The variable portion is different for each
primary response for forming antibodies that occurs on
specificity of antibody, and it is this portion that
first exposure to a specific antigen and the secondary
attaches specifically to a particular type of antigen. The
response that occurs after second exposure to the same
constant portion of the antibody determines other
antigen. Note the 1-week delay in the appearance of
properties of the antibody, establishing such factors as
the primary response, its weak potency, and its short
diffusivity of the antibody in the tissues, adherence of
life. The secondary response, by contrast, begins
the antibody to specific structures within the tissues,
rapidly after exposure to the antigen (often within
attachment to the complement complex, the ease with
hours), is far more potent, and forms antibodies for
which the antibodies pass through membranes, and
many months rather than for only a few weeks. The
other biological properties of the antibody.
444
Unit VI Blood Cells, Immunity, and Blood Clotting
Antigen-binding sites
Variable portion
Antigen
Antigen
Antibodies
S • S
Light
S • S
chain
Constant portion
Heavy
chain
Figure 34-3
Structure of the typical IgG antibody, showing it to be composed
of two heavy polypeptide chains and two light polypeptide chains.
Figure 34-4
The antigen binds at two different sites on the variable portions of
the chains.
Binding of antigen molecules to one another by bivalent
antibodies.
Specificity of Antibodies. Each antibody is specific for a
in allergy. The IgM class is also interesting because
particular antigen; this is caused by its unique struc-
a large share of the antibodies formed during the
tural organization of amino acids in the variable por-
primary response are of this type. These antibodies
tions of both the light and heavy chains. The amino
have 10 binding sites that make them exceedingly
acid organization has a different steric shape for each
effective in protecting the body against invaders, even
antigen specificity, so that when an antigen comes in
though there are not many IgM antibodies.
contact with it, multiple prosthetic groups of the
antigen fit as a mirror image with those of the anti-
Mechanisms of Action of Antibodies
body, thus allowing rapid and tight bonding between
Antibodies act mainly in two ways to protect the body
the antibody and the antigen. When the antibody is
against invading agents: (1) by direct attack on the
highly specific, there are so many bonding sites that the
invader and (2) by activation of the “complement
antibody-antigen coupling is exceedingly strong, held
system” that then has multiple means of its own for
together by (1) hydrophobic bonding, (2) hydrogen
destroying the invader.
bonding, (3) ionic attractions, and (4) van der Waals
forces. It also obeys the thermodynamic mass action
Direct Action of Antibodies on Invading Agents. Figure 34-4
law.
shows antibodies (designated by the red Y-shaped
bars) reacting with antigens (designated by the shaded
K = Concentration of bound antibody-antigen
a
objects). Because of the bivalent nature of the anti-
Concentration of antibody
bodies and the multiple antigen sites on most invad-
¥
Concentration of antigen
ing agents, the antibodies can inactivate the invading
Ka is called the affinity constant and is a measure of
agent in one of several ways, as follows:
how tightly the antibody binds with the antigen.
1. Agglutination, in which multiple large particles
Note, especially, in Figure 34-3 that there are two
with antigens on their surfaces, such as bacteria or
variable sites on the illustrated antibody for attach-
red cells, are bound together into a clump
ment of antigens, making this type of antibody biva-
2. Precipitation, in which the molecular complex of
lent. A small proportion of the antibodies, which
soluble antigen (such as tetanus toxin) and
consist of combinations of up to 10 light and 10 heavy
antibody becomes so large that it is rendered
chains, have as many as 10 binding sites.
insoluble and precipitates
3. Neutralization, in which the antibodies cover the
Classes of Antibodies. There are five general classes of
toxic sites of the antigenic agent
antibodies, respectively named IgM, IgG, IgA, IgD,
4. Lysis, in which some potent antibodies are
and IgE. Ig stands for immunoglobulin, and the other
occasionally capable of directly attacking
five respective letters designate the respective classes.
membranes of cellular agents and thereby cause
For the purpose of our present limited discussion,
rupture of the agent
two of these classes of antibodies are of particular
These direct actions of antibodies attacking the anti-
importance: IgG, which is a bivalent antibody and con-
genic invaders often are not strong enough to play a
stitutes about 75 per cent of the antibodies of the
major role in protecting the body against the invader.
normal person, and IgE, which constitutes only a small
Most of the protection comes through the amplifying
percentage of the antibodies but is especially involved
effects of the complement system described next.
Chapter 34
Resistance of the Body to Infection: II. Immunity and Allergy
445
Antigen-antibody complex
Opsonization of bacteria
C1
C1
Activate mast
cells and basophils
C4 + C2
C42 + C4a
Chemotaxis of
C3
C3b + C3a
white blood cells
C5
C5b + C5a
Micro-organism +
B and D
C6 + C7
C5b67
Figure 34-5
Cascade of reactions during
C8 + C9
C5b6789
activation of the classic pathway
of complement.
(Modified from
Alexander JW, Good RA: Funda-
Lysis of cells
mentals of Clinical Immunology.
Philadelphia: WB Saunders,
1977.)
Complement System for Antibody Action
to engulf the bacteria to which the antigen-
“Complement” is a collective term that describes a
antibody complexes are attached. This process is
system of about 20 proteins, many of which are enzyme
called opsonization. It often enhances the number
precursors. The principal actors in this system are 11
of bacteria that can be destroyed by many
proteins designated C1 through C9, B, and D, shown
hundredfold.
in Figure 34-5. All these are present normally among
2.
Lysis. One of the most important of all the
the plasma proteins in the blood as well as among the
products of the complement cascade is the lytic
proteins that leak out of the capillaries into the tissue
complex, which is a combination of multiple
spaces. The enzyme precursors are normally inactive,
complement factors and designated C5b6789.
but they can be activated mainly by the so-called
This has a direct effect of rupturing the cell
classic pathway.
membranes of bacteria or other invading
organisms.
Classic Pathway. The classic pathway is initiated by an
3.
Agglutination. The complement products also
antigen-antibody reaction. That is, when an antibody
change the surfaces of the invading organisms,
binds with an antigen, a specific reactive site on the
causing them to adhere to one another, thus
“constant” portion of the antibody becomes uncov-
promoting agglutination.
ered, or “activated,” and this in turn binds directly with
4.
Neutralization of viruses. The complement
the C1 molecule of the complement system, setting
enzymes and other complement products can
into motion a “cascade” of sequential reactions, shown
attack the structures of some viruses and thereby
in Figure 34-5, beginning with activation of the proen-
render them nonvirulent.
zyme C1 itself. The C1 enzymes that are formed then
5.
Chemotaxis. Fragment C5a initiates chemotaxis of
activate successively increasing quantities of enzymes in
neutrophils and macrophages, thus causing large
the later stages of the system, so that from a small
numbers of these phagocytes to migrate into the
beginning, an extremely large “amplified” reaction
tissue area adjacent to the antigenic agent.
occurs. Multiple end products are formed, as shown to
6.
Activation of mast cells and basophils. Fragments
the right in the figure, and several of these cause
C3a, C4a, and C5a activate mast cells and
important effects that help to prevent damage to the
basophils, causing them to release histamine,
body’s tissues caused by the invading organism or
heparin, and several other substances into the
toxin. Among the more important effects are the
local fluids. These substances in turn cause
following:
increased local blood flow, increased leakage of
1. Opsonization and phagocytosis. One of the
fluid and plasma protein into the tissue, and other
products of the complement cascade, C3b,
local tissue reactions that help inactivate or
strongly activates phagocytosis by both
immobilize the antigenic agent. The same factors
neutrophils and macrophages, causing these cells
play a major role in inflammation (which was
446
Unit VI Blood Cells, Immunity, and Blood Clotting
discussed in Chapter 33) and in allergy, as we
discuss later.
T cell
7. Inflammatory effects. In addition to inflammatory
effects caused by activation of the mast cells and
basophils, several other complement products
T-cell receptor
Cell-cell
contribute to local inflammation. These products
adhesion
Foreign protein
cause (1) the already increased blood flow to
proteins
MHC protein
increase still further, (2) the capillary leakage of
proteins to be increased, and (3) the interstitial
fluid proteins to coagulate in the tissue spaces,
thus preventing movement of the invading
organism through the tissues.
Antigen-
Special Attributes of the
presenting
T-Lymphocyte System-Activated
cell
T Cells and Cell-Mediated Immunity
Figure 34-6
Release of Activated T Cells from Lymphoid Tissue and Forma-
Activation of T cells requires interaction of T-cell receptors with an
tion of Memory Cells. On exposure to the proper antigen,
antigen (foreign protein) that is transported to the surface of the
as presented by adjacent macrophages, the T lympho-
antigen-presenting cell by a major histocompatibility complex
cytes of a specific lymphocyte clone proliferate and
(MHC) protein. Cell-to-cell adhesion proteins enable the T cell
to bind to the antigen-presenting cell long enough to become
release large numbers of activated, specifically react-
activated.
ing T cells in ways that parallel antibody release by
activated B cells. The principal difference is that
instead of releasing antibodies, whole activated T cells
to present antigens to T cells. Interaction of cell adhe-
are formed and released into the lymph. These then
sion proteins is critical in permitting the T cells to bind
pass into the circulation and are distributed through-
to antigen-presenting cells long enough to become
out the body, passing through the capillary walls into
activated.
the tissue spaces, back into the lymph and blood once
The MHC proteins are encoded by a large group of
again, and circulating again and again throughout the
genes called the major histocompatibility complex
body, sometimes lasting for months or even years.
(MHC). The MHC proteins bind peptide fragments
Also, T-lymphocyte memory cells are formed in the
of antigen proteins that are degraded inside antigen-
same way that B memory cells are formed in the anti-
presenting cells and then transport them to the cell
body system. That is, when a clone of T lymphocytes is
surface. There are two types of MHC proteins: (1)
activated by an antigen, many of the newly formed
MHC I proteins, which present antigens to cytotoxic T
lymphocytes are preserved in the lymphoid tissue to
cells, and (2) MHC II proteins, which present antigens
become additional T lymphocytes of that specific
to T helper cells. The specific functions of cytotoxic and
clone; in fact, these memory cells even spread through-
helper T cells are discussed later.
out the lymphoid tissue of the entire body. Therefore,
The antigens on the surface of antigen-presenting
on subsequent exposure to the same antigen anywhere
cells bind with receptor molecules on the surfaces of T
in the body, release of activated T cells occurs far more
cells in the same way that they bind with plasma
rapidly and much more powerfully than had occurred
protein antibodies. These receptor molecules are com-
during first exposure.
posed of a variable unit similar to the variable portion
of the humoral antibody, but its stem section is firmly
Antigen-Presenting Cells, MHC Proteins, and Antigen Recep-
bound to the cell membrane of the T lymphocyte.
tors on the T Lymphocytes. T-cell responses are extremely
There are as many as 100,000 receptor sites on a single
antigen specific, like the antibody responses of B cells,
T cell.
and are at least as important as antibodies in de-
fending against infection. In fact, acquired immune
responses usually require assistance from T cells to
Several Types of T Cells and Their
begin the process, and T cells play a major role in actu-
Different Functions
ally helping to eliminate invading pathogens.
Although B lymphocytes recognize intact antigens,
It has become clear that there are multiple types of T
T lymphocytes respond to antigens only when they are
cells. They are classified into three major groups: (1)
bound to specific molecules called MHC proteins on
helper T cells, (2) cytotoxic T cells, and (3) suppressor
the surface of antigen-presenting cells in the lymphoid
T cells. The functions of each of these are distinct.
tissues
(Figure
34-6). The three major types of
antigen-presenting cells are macrophages, B lympho-
Helper T Cells—Their Role in Overall
cytes, and dendritic cells. The dendritic cells, the most
Regulation of Immunity
potent of the antigen-presenting cells, are located
The helper T cells are by far the most numerous of the
throughout the body, and their only known function is
T cells, usually constituting more than three quarters
Chapter 34
Resistance of the Body to Infection: II. Immunity and Allergy
447
lethal effects of AIDS. Some of the specific regulatory
functions are the following.
PrPreprocesr
sor
arareas
Antigen
Stimulation of Growth and Proliferation of Cytotoxic T
Cells and Suppressor T Cells. In the absence of helper
T cells, the clones for producing cytotoxic T cells and
Processed
MHC
suppressor T cells are activated only slightly by most
antigen
antigens. The lymphokine interleukin-2 has an espe-
Interleukin-1
cially strong stimulatory effect in causing growth and
Antigen-specific receptor
proliferation of both cytotoxic and suppressor T cells.
In addition, several of the other lymphokines have less
Cytotoxic
T cells
potent effects.
Helper
T cells
Stimulation of B-Cell Growth and Differentiation to
Form Plasma Cells and Antibodies. The direct actions
of antigen to cause B-cell growth, proliferation, for-
mation of plasma cells, and secretion of antibodies are
also slight without the “help” of the helper T cells.
Lymphokines!!
Suppressor
Almost all the interleukins participate in the B-cell
T cells
response, but especially interleukins 4, 5, and 6. In fact,
these three interleukins have such potent effects on
the B cells that they have been called B-cell stimulat-
Proliferation
Differentiation
ing factors or B-cell growth factors.
Plasma
cells
IgM
Activation of the Macrophage System. The lym-
phokines also affect the macrophages. First, they slow
IgG
or stop the migration of the macrophages after they
Antigen
have been chemotactically attracted into the inflamed
IgA
tissue area, thus causing great accumulation of
B cell
IgE
macrophages. Second, they activate the macrophages
to cause far more efficient phagocytosis, allowing them
Figure 34-7
to attack and destroy increasing numbers of invading
bacteria or other tissue-destroying agents.
Regulation of the immune system, emphasizing a pivotal role of
the helper T cells. MHC, major histocompatibility complex.
Feedback Stimulatory Effect on the Helper Cells Them-
selves. Some of the lymphokines, especially inter-
of all of them. As their name implies, they help in the
leukin-2, have a direct positive feedback effect in
functions of the immune system, and they do so in
stimulating activation of the helper T cells themselves.
many ways. In fact, they serve as the major regulator
This acts as an amplifier by further enhancing the
of virtually all immune functions, as shown in Figure
helper cell response as well as the entire immune
34-7. They do this by forming a series of protein medi-
response to an invading antigen.
ators, called lymphokines, that act on other cells of the
immune system as well as on bone marrow cells.
Cytotoxic T Cells
Among the important lymphokines secreted by the
The cytotoxic T cell is a direct-attack cell that is
helper T cells are the following:
capable of killing micro-organisms and, at times, even
some of the body’s own cells. For this reason, these
Interleukin-2
cells are called killer cells. The receptor proteins on the
Interleukin-3
surfaces of the cytotoxic cells cause them to bind
Interleukin-4
tightly to those organisms or cells that contain the
Interleukin-5
appropriate binding-specific antigen. Then, they kill
Interleukin-6
the attacked cell in the manner shown in Figure 34-8.
Granulocyte-monocyte colony-stimulating factor
After binding, the cytotoxic T cell secretes hole-
Interferon-g
forming proteins, called perforins, that literally punch
round holes in the membrane of the attacked cell.
Specific Regulatory Functions of the Lymphokines. In the
Then fluid flows rapidly into the cell from the intersti-
absence of the lymphokines from the helper T cells,
tial space. In addition, the cytotoxic T cell releases
the remainder of the immune system is almost para-
cytotoxic substances directly into the attacked cell.
lyzed. In fact, it is the helper T cells that are inactivated
Almost immediately, the attacked cell becomes greatly
or destroyed by the acquired immunodeficiency syn-
swollen, and it usually dissolves shortly thereafter.
drome (AIDS) virus, which leaves the body almost
Especially important, these cytotoxic killer cells can
totally unprotected against infectious disease, there-
pull away from the victim cells after they have
fore leading to the now well-known debilitating and
punched holes and delivered cytotoxic substances and
448
Unit VI Blood Cells, Immunity, and Blood Clotting
as being distinctive from bacteria or viruses, and the
Cytotoxic
person’s immunity system forms few antibodies or
T cells
activated T cells against his or her own antigens.
(killer cells)
Cytotoxic
Most Tolerance Results from Clone Selection During Prepro-
and
digestive
cessing. It is believed that most tolerance develops
enzymes
during preprocessing of T lymphocytes in the thymus
and of B lymphocytes in the bone marrow. The reason
for this belief is that injecting a strong antigen into a
fetus while the lymphocytes are being preprocessed in
these two areas prevents development of clones of
lymphocytes in the lymphoid tissue that are specific for
the injected antigen. Experiments have shown that
Attacked
Specific
specific immature lymphocytes in the thymus, when
Antigen
cell
binding
exposed to a strong antigen, become lymphoblastic,
receptors
proliferate considerably, and then combine with the
stimulating antigen—an effect that is believed to cause
Antigen
the cells themselves to be destroyed by the thymic
epithelial cells before they can migrate to and colonize
Figure 34-8
the total body lymphoid tissue.
It is believed that during the preprocessing of lym-
Direct destruction of an invading cell by sensitized lymphocytes
phocytes in the thymus and bone marrow, all or most
(cytotoxic T cells).
of those clones of lymphocytes that are specific to
damage the body’s own tissues are self-destroyed
because of their continual exposure to the body’s
antigens.
then move on to kill more cells. Indeed, some of these
cells persist for months in the tissues.
Failure of the Tolerance Mechanism Causes Autoimmune Dis-
Some of the cytotoxic T cells are especially lethal to
eases. Sometimes people lose their immune tolerance
tissue cells that have been invaded by viruses because
of their own tissues. This occurs to a greater extent the
many virus particles become entrapped in the mem-
older a person becomes. It usually occurs after destruc-
branes of the tissue cells and attract T cells in response
tion of some of the body’s own tissues, which releases
to the viral antigenicity. The cytotoxic cells also play
considerable quantities of
“self-antigens” that cir-
an important role in destroying cancer cells, heart
culate in the body and presumably cause acquired
transplant cells, or other types of cells that are foreign
immunity in the form of either activated T cells or
to the person’s own body.
antibodies.
Several specific diseases that result from autoim-
Suppressor T Cells
munity include (1) rheumatic fever, in which the body
Much less is known about the suppressor T cells than
becomes immunized against tissues in the joints and
about the others, but they are capable of suppressing
heart, especially the heart valves, after exposure to a
the functions of both cytotoxic and helper T cells. It is
specific type of streptococcal toxin that has an epitope
believed that these suppressor functions serve the
in its molecular structure similar to the structure of
purpose of preventing the cytotoxic cells from causing
some of the body’s own self-antigens; (2) one type of
excessive immune reactions that might be damaging to
glomerulonephritis, in which the person becomes
the body’s own tissues. For this reason, the suppressor
immunized against the basement membranes of
cells are classified, along with the helper T cells, as reg-
glomeruli; (3) myasthenia gravis, in which immunity
ulatory T cells. It is probable that the suppressor T-cell
develops against the acetylcholine receptor proteins of
system plays an important role in limiting the ability
the neuromuscular junction, causing paralysis; and (4)
of the immune system to attack a person’s own body
lupus erythematosus, in which the person becomes
tissues, called immune tolerance, as we discuss in the
immunized against many different body tissues at the
next section.
same time, a disease that causes extensive damage and
often rapid death.
Tolerance of the Acquired Immunity
System to One’s Own Tissues—Role
Immunization by Injection of Antigens
of Preprocessing in the Thymus and
Bone Marrow
Immunization has been used for many years to
produce acquired immunity against specific diseases. A
If a person should become immune to his or her own
person can be immunized by injecting dead organisms
tissues, the process of acquired immunity would
that are no longer capable of causing disease but that
destroy the individual’s own body. The immune mech-
still have some of their chemical antigens. This type of
anism normally “recognizes” a person’s own tissues
immunization is used to protect against typhoid fever,
Chapter 34
Resistance of the Body to Infection: II. Immunity and Allergy
449
whooping cough, diphtheria, and many other types of
diffuse from the circulating blood in large numbers
bacterial diseases.
into the skin to respond to the poison ivy toxin. And,
Immunity can be achieved against toxins that have
at the same time, these T cells elicit a cell-mediated
been treated with chemicals so that their toxic nature
type of immune reaction. Remembering that this type
has been destroyed even though their antigens for
of immunity can cause release of many toxic sub-
causing immunity are still intact. This procedure is
stances from the activated T cells as well as extensive
used in immunizing against tetanus, botulism, and
invasion of the tissues by macrophages along with
other similar toxic diseases.
their subsequent effects, one can well understand that
And, finally, a person can be immunized by being
the eventual result of some delayed-reaction allergies
infected with live organisms that have been “attenu-
can be serious tissue damage. The damage normally
ated.” That is, these organisms either have been grown
occurs in the tissue area where the instigating antigen
in special culture media or have been passed through
is present, such as in the skin in the case of poison ivy,
a series of animals until they have mutated enough
or in the lungs to cause lung edema or asthmatic
that they will not cause disease but do still carry spe-
attacks in the case of some airborne antigens.
cific antigens required for immunization. This proce-
dure is used to protect against poliomyelitis, yellow
fever, measles, smallpox, and many other viral diseases.
Allergies in the “Allergic” Person,
Who Has Excess IgE Antibodies
Passive Immunity
Some people have an “allergic” tendency. Their aller-
gies are called atopic allergies because they are caused
Thus far, all the acquired immunity we have discussed
by a nonordinary response of the immune system. The
has been active immunity. That is, the person’s own
allergic tendency is genetically passed from parent to
body develops either antibodies or activated T cells in
child and is characterized by the presence of large
response to invasion of the body by a foreign antigen.
quantities of IgE antibodies in the blood. These anti-
However, temporary immunity can be achieved in a
bodies are called reagins or sensitizing antibodies to
person without injecting any antigen. This is done by
distinguish them from the more common IgG anti-
infusing antibodies, activated T cells, or both obtained
bodies. When an allergen
(defined as an antigen
from the blood of someone else or from some other
that reacts specifically with a specific type of IgE
animal that has been actively immunized against the
reagin antibody) enters the body, an allergen-reagin
antigen.
reaction lakes place, and a subsequent allergic reaction
Antibodies last in the body of the recipient for 2 to
occurs.
3 weeks, and during that time, the person is protected
A special characteristic of the IgE antibodies (the
against the invading disease. Activated T cells last for
reagins) is a strong propensity to attach to mast cells
a few weeks if transfused from another person but
and basophils. Indeed, a single mast cell or basophil
only for a few hours to a few days if transfused
can bind as many as half a million molecules of IgE
from an animal. Such transfusion of antibodies or T
antibodies. Then, when an antigen (an allergen) that
lymphocytes to confer immunity is called passive
has multiple binding sites binds with several IgE anti-
immunity.
bodies that are already attached to a mast cell or
basophil, this causes immediate change in the mem-
brane of the mast cell or basophil, perhaps resulting
ALLERGY AND
from a physical effect of the antibody molecules to
HYPERSENSITIVITY
contort the cell membrane. At any rate, many of the
mast cells and basophils rupture; others release special
An important undesirable side effect of immunity is
agents immediately or shortly thereafter, including his-
the development, under some conditions, of allergy or
tamine, protease, slow-reacting substance of anaphy-
other types of immune hypersensitivity. There are
laxis
(which is a mixture of toxic leukotrienes),
several types of allergy and other hypersensitivities,
eosinophil chemotactic substance, neutrophil chemo-
some of which occur only in people who have a spe-
tactic substance, heparin, and platelet activating factors.
cific allergic tendency.
These substances cause such effects as dilation of the
local blood vessels; attraction of eosinophils and neu-
trophils to the reactive site; increased permeability of
Allergy Caused by Activated T Cells:
the capillaries with loss of fluid into the tissues; and
Delayed-Reaction Allergy
contraction of local smooth muscle cells. Therefore,
several different tissue responses can occur, depend-
Delayed-reaction allergy is caused by activated T cells
ing on the type of tissue in which the allergen-reagin
and not by antibodies. In the case of poison ivy, the
reaction occurs. Among the different types of allergic
toxin of poison ivy in itself does not cause much harm
reactions caused in this manner are the following.
to the tissues. However, on repeated exposure, it does
cause the formation of activated helper and cytotoxic
Anaphylaxis. When a specific allergen is injected
T cells. Then, after subsequent exposure to the poison
directly into the circulation, the allergen can react with
ivy toxin, within a day or so, the activated T cells
basophils of the blood and mast cells in the tissues
450
Unit VI Blood Cells, Immunity, and Blood Clotting
located immediately outside the small blood vessels
not appear to be the major factor eliciting the asth-
if the basophils and mast cells have been sensitized
matic reaction.
by attachment of IgE reagins. Therefore, a widespread
allergic reaction occurs throughout the vascular
system and closely associated tissues. This is called
References
anaphylaxis. Histamine is released into the circulation
and causes body-wide vasodilation as well as increased
Abreu MT, Arditi M: Innate immunity and toll-like recep-
permeability of the capillaries with resultant marked
tors: clinical implications of basic science research. J
loss of plasma from the circulation. An occasional
Pediatr 144(4):421, 2004.
person who experiences this reaction dies of circula-
Albert ML: Death-defying immunity: do apoptotic cells
tory shock within a few minutes unless treated with
influence antigen processing and presentation? Nat Rev
epinephrine to oppose the effects of the histamine.
Immunol 4:223, 2004.
Also released from the activated basophils and mast
Alberts B, Johnson A, Lewis J, et al: Molecular Biology of
cells is a mixture of leukotrienes called slow-reacting
the Cell. New York: Garland Science, 2002.
substance of anaphylaxis. These leukotrienes can cause
Cheroutre H, Madakamutil L: Acquired and natural
memory T cells join forces at the mucosal front line. Nat
spasm of the smooth muscle of the bronchioles, elicit-
Rev Immunol 4:290, 2004.
ing an asthma-like attack, sometimes causing death by
Cooper MA, Pommering TL, Koranyi K: Primary immuno-
suffocation.
deficiencies. Am Fam Physician 68:2001, 2003.
Cossart P, Sansonetti PJ: Bacterial invasion: the paradigms
Urticaria. Urticaria results from antigen entering spe-
of enteroinvasive pathogens. Science 304:242, 2004.
cific skin areas and causing localized anaphylactoid
Eisenbarth GS, Gottlieb PA: Autoimmune polyendocrine
reactions. Histamine released locally causes (1) vasodi-
syndromes. N Engl J Med. 350:2068, 2004.
lation that induces an immediate red flare and (2)
Figdor CG, de Vries IJ, Lesterhuis WJ, Melief CJ: Dendritic
increased local permeability of the capillaries that
cell immunotherapy: mapping the way. Nat Med 10:475,
leads to local circumscribed areas of swelling of the
2004.
Frossi B, De Carli M, Pucillo C: The mast cell: an antenna of
skin within another few minutes. The swellings are
the microenvironment that directs the immune response.
commonly called hives. Administration of antihista-
J Leukoc Biol 75:579, 2004.
mine drugs to a person before exposure will prevent
Grossman Z, Min B, Meier-Schellersheim M, Paul WE: Con-
the hives.
comitant regulation of T-cell activation and homeostasis.
Nat Rev Immunol 4:387, 2004.
Hay Fever. In hay fever, the allergen-reagin reaction
Kupper TS, Fuhlbrigge RC: Immune surveillance in the skin:
occurs in the nose. Histamine released in response to
mechanisms and clinical consequences. Nat Rev Immunol
the reaction causes local intranasal vascular dilation,
4:211, 2004.
with resultant increased capillary pressure as well as
La Cava A, Matarese G: The weight of leptin in immunity.
increased capillary permeability. Both these effects
Nat Rev Immunol 4:371, 2004.
Linton PJ, Dorshkind K: Age-related changes in lymphocyte
cause rapid fluid leakage into the nasal cavities and
development and function. Nat Immunol 5:133, 2004.
into associated deeper tissues of the nose; and the
MacGlashan D Jr: Histamine: a mediator of inflammation. J
nasal linings become swollen and secretory. Here
Allergy Clin Immunol 112(4 Suppl):S53, 2003.
again, use of antihistamine drugs can prevent this
McGeady SJ: Immunocompetence and allergy. Pediatrics
swelling reaction. But other products of the allergen-
113(4 Suppl):1107, 2004.
reagin reaction can still cause irritation of the nose,
Nikolich-Zugich J, Slifka MK, Messaoudi I: The many impor-
eliciting the typical sneezing syndrome.
tant facets of T-cell repertoire diversity. Nat Rev Immunol
4:123, 2004 .
Asthma. Asthma often occurs in the “allergic” type of
Petrie HT: Cell migration and the control of post-natal T-cell
person. In such a person, the allergen-reagin reaction
lymphopoiesis in the thymus. Nat Rev Immunol 3:859,
2003.
occurs in the bronchioles of the lungs. Here, an impor-
Scott CC, Botelho RJ, Grinstein S: Phagosome maturation:
tant product released from the mast cells is believed
a few bugs in the system. J Membr Biol 193:137, 2003.
to be the slow-reacting substance of anaphylaxis, which
Theoharides TC, Cochrane DE: Critical role of mast cells in
causes spasm of the bronchiolar smooth muscle. Con-
inflammatory diseases and the effect of acute stress. J Neu-
sequently, the person has difficulty breathing until the
roimmunol 146:1, 2004.
reactive products of the allergic reaction have been
Vivier E, Anfossi N: Inhibitory NK-cell receptors on T cells:
removed. Administration of antihistaminics has less
witness of the past, actors of the future. Nat Rev Immunol
effect on the course of asthma because histamine does
4:190, 2004.
C H A P T E R
3
5
Blood Types; Transfusion; Tissue
and Organ Transplantation
Antigenicity Causes Immune
Reactions of Blood
When blood transfusions from one person to
another were first attempted, immediate or delayed
agglutination and hemolysis of the red blood cells
often occurred, resulting in typical transfusion reac-
tions that frequently led to death. Soon it was dis-
covered that the bloods of different people have different antigenic and immune
properties, so that antibodies in the plasma of one blood will react with anti-
gens on the surfaces of the red cells of another blood type. If proper precau-
tions are taken, one can determine ahead of time whether the antibodies
and antigens present in the donor and recipient bloods will cause a transfusion
reaction.
Multiplicity of Antigens in the Blood Cells. At least 30 commonly occurring antigens
and hundreds of other rare antigens, each of which can at times cause antigen-
antibody reactions, have been found in human blood cells, especially on the sur-
faces of the cell membranes. Most of the antigens are weak and therefore are
of importance principally for studying the inheritance of genes to establish
parentage.
Two particular types of antigens are much more likely than the others to
cause blood transfusion reactions. They are the O-A-B system of antigens and
the Rh system.
O-A-B Blood Types
A and B Antigens—Agglutinogens
Two antigens—type A and type B—occur on the surfaces of the red blood cells
in a large proportion of human beings. It is these antigens (also called agglu-
tinogens because they often cause blood cell agglutination) that cause most
blood transfusion reactions. Because of the way these agglutinogens are inher-
ited, people may have neither of them on their cells, they may have one, or they
may have both simultaneously.
Major O-A-B Blood Types. In transfusing blood from one person to another, the
bloods of donors and recipients are normally classified into four major O-A-B
blood types, as shown in Table 35-1, depending on the presence or absence of
the two agglutinogens, the A and B agglutinogens. When neither A nor B agglu-
tinogen is present, the blood is type O. When only type A agglutinogen is
present, the blood is type A. When only type B agglutinogen is present, the blood
is type B. When both A and B agglutinogens are present, the blood is type AB.
Genetic Determination of the Agglutinogens. Two genes, one on each of two paired
chromosomes, determine the O-A-B blood type. These genes can be any one of
three types but only one type on each of the two chromosomes: type O, type A,
451
452
Unit VI Blood Cells, Immunity, and Blood Clotting
Table 35-1
Blood Types with Their Genotypes and Their Constituent
Agglutinogens and Agglutinins
Anti-A agglutinins in
groups B and O blood
Anti-B agglutinins in
Genotypes
Blood Types
Agglutinogens
Agglutinins
400
groups A and O blood
OO
O
—
Anti-A and
Anti-B
300
OA or AA
A
A
Anti-B
OB or BB
B
B
Anti-A
AB
AB
A and B
—
200
or type B. The type O gene is either functionless or
100
almost functionless, so that it causes no significant type
O agglutinogen on the cells. Conversely, the type A
and type B genes do cause strong agglutinogens on the
0
0
10 20 30 40 50 60 70 80 90 100
cells.
Age of person (years)
The six possible combinations of genes, as shown in
Table 35-1, are OO, OA, OB, AA, BB, and AB. These
combinations of genes are known as the genotypes,
and each person is one of the six genotypes.
One can also observe from Table 35-1 that a person
Figure 35-1
with genotype OO produces no agglutinogens, and
Average titers of anti-A and anti-B agglutinins in the plasmas of
therefore the blood type is O. A person with genotype
people with different blood types.
OA or AA produces type A agglutinogens and there-
fore has blood type A. Genotypes OB and BB give
type B blood, and genotype AB gives type AB blood.
when type A agglutinogens are not present in the cells,
and anti-B agglutinins when type B agglutinogens are
Relative Frequencies of the Different Blood Types.
not in the cells. Figure 35-1 shows the changing titers
The prevalence of the different blood types among one
of the anti-A and anti-B agglutinins at different ages.
group of persons studied was approximately:
A maximum titer is usually reached at 8 to 10 years
of age, and this gradually declines throughout the
O
47%
A
41%
remaining years of life.
B
9%
AB
3%
Origin of Agglutinins in the Plasma. The agglutinins are
gamma globulins, as are almost all antibodies, and they
It is obvious from these percentages that the O and
are produced by the same bone marrow and lymph
A genes occur frequently, whereas the B gene is
gland cells that produce antibodies to any other anti-
infrequent.
gens. Most of them are IgM and IgG immunoglobulin
molecules.
But why are these agglutinins produced in people
Agglutinins
who do not have the respective agglutinogens in their
red blood cells? The answer to this is that small
When type A agglutinogen is not present in a person’s
amounts of type A and B antigens enter the body in
red blood cells, antibodies known as anti-A agglutinins
food, in bacteria, and in other ways, and these sub-
develop in the plasma. Also, when type B agglutino-
stances initiate the development of the anti-A and
gen is not present in the red blood cells, antibodies
anti-B agglutinins.
known as anti-B agglutinins develop in the plasma.
For instance, infusion of group A antigen into a
Thus, referring once again to Table 35-1, note that
recipient having a non-A blood type causes a typical
type O blood, although containing no agglutinogens,
immune response with formation of greater quantities
does contain both anti-A and anti-B agglutinins; type
of anti-A agglutinins than ever. Also, the neonate has
A blood contains type A agglutinogens and anti-B
few, if any, agglutinins, showing that agglutinin forma-
agglutinins; type B blood contains type B agglutino-
tion occurs almost entirely after birth.
gens and anti-A agglutinins. Finally, type AB blood
contains both A and B agglutinogens but no
agglutinins.
Agglutination Process In Transfusion
Reactions
Titer of the Agglutinins at Different Ages. Immediately after
birth, the quantity of agglutinins in the plasma is
When bloods are mismatched so that anti-A or anti-B
almost zero. Two to 8 months after birth, an infant
plasma agglutinins are mixed with red blood cells that
begins to produce agglutinins—anti-A agglutinins
contain A or B agglutinogens, respectively, the red
Chapter 35
Blood Types; Transfusion; Tissue and Organ Transplantation
453
cells agglutinate as a result of the agglutinins’ attach-
O red blood cells have no agglutinogens and therefore
ing themselves to the red blood cells. Because the
do not react with either the anti-A or the anti-B agglu-
agglutinins have two binding sites (IgG type) or 10
tinins. Type A blood has A agglutinogens and there-
binding sites (IgM type), a single agglutinin can attach
fore agglutinates with anti-A agglutinins. Type B
to two or more red blood cells at the same time,
blood has B agglutinogens and agglutinates with
thereby causing the cells to be bound together by the
anti-B agglutinins. Type AB blood has both A and B
agglutinin. This causes the cells to clump, which is the
agglutinogens and agglutinates with both types of
process of “agglutination.” Then these clumps plug
agglutinins.
small blood vessels throughout the circulatory system.
During ensuing hours to days, either physical distor-
tion of the cells or attack by phagocytic white blood
Rh Blood Types
cells destroys the membranes of the agglutinated cells,
releasing hemoglobin into the plasma, which is called
Along with the O-A-B blood type system, the Rh
“hemolysis” of the red blood cells.
blood type system is also important when transfusing
blood. The major difference between the O-A-B
Acute Hemolysis Occurs in Some Transfusion Reactions.
system and the Rh system is the following: In the
Sometimes, when recipient and donor bloods are mis-
O-A-B system, the plasma agglutinins responsible for
matched, immediate hemolysis of red cells occurs in
causing transfusion reactions develop spontaneously,
the circulating blood. In this case, the antibodies cause
whereas in the Rh system, spontaneous agglutinins
lysis of the red blood cells by activating the comple-
almost never occur. Instead, the person must first be
ment system, which releases proteolytic enzymes (the
massively exposed to an Rh antigen, such as by trans-
lytic complex) that rupture the cell membranes, as
fusion of blood containing the Rh antigen, before
described in Chapter
34. Immediate intravascular
enough agglutinins to cause a significant transfusion
hemolysis is far less common than agglutination fol-
reaction will develop.
lowed by delayed hemolysis, because not only does
there have to be a high titer of antibodies for lysis to
Rh Antigens—“Rh-Positive” and
“Rh-Negative” People.
occur, but also a different type of antibody seems to
There are six common types of Rh antigens, each of
be required, mainly the IgM antibodies; these anti-
which is called an Rh factor. These types are desig-
bodies are called hemolysins.
nated C, D, E, c, d, and e. A person who has a C antigen
does not have the c antigen, but the person missing the
C antigen always has the c antigen. The same is true
Blood Typing
for the D-d and E-e antigens. Also, because of the
manner of inheritance of these factors, each person has
Before giving a transfusion to a person, it is necessary
one of each of the three pairs of antigens.
to determine the blood type of the recipient’s blood
The type D antigen is widely prevalent in the
and the blood type of the donor blood so that the
population and considerably more antigenic than
bloods can be appropriately matched. This is called
the other Rh antigens. Anyone who has this type of
blood typing and blood matching, and these are per-
antigen is said to be Rh positive, whereas a person who
formed in the following way: The red blood cells are
does not have type D antigen is said to be Rh negative.
first separated from the plasma and diluted with saline.
However, it must be noted that even in Rh-negative
One portion is then mixed with anti-A agglutinin and
people, some of the other Rh antigens can still cause
another portion with anti-B agglutinin. After several
transfusion reactions, although the reactions are
minutes, the mixtures are observed under a micro-
usually much milder.
scope. If the red blood cells have become clumped—
About 85 per cent of all white people are Rh posi-
that is, “agglutinated”—one knows that an antibody-
tive and 15 per cent, Rh negative. In American blacks,
antigen reaction has resulted.
the percentage of Rh-positives is about 95, whereas in
Table 35-2 lists the presence (+) or absence (-) of
African blacks, it is virtually 100 per cent.
agglutination of the four types of red blood cells. Type
Table 35-2
Rh Immune Response
Blood Typing, Showing Agglutination of Cells of the
Different Blood Types with Anti-A or Anti-B Agglutinins
Formation of Anti-Rh Agglutinins. When red blood cells
in the Sera
containing Rh factor are injected into a person whose
blood does not contain the Rh factor—that is, into
Red Blood Cell Types
Sera
an Rh-negative person—anti-Rh agglutinins develop
Anti-A
Anti-B
slowly, reaching maximum concentration of agglu-
tinins about 2 to 4 months later. This immune response
O
-
-
occurs to a much greater extent in some people than
A
+
-
in others. With multiple exposures to the Rh factor,
B
-
+
AB
+
+
an Rh-negative person eventually becomes strongly
“sensitized” to Rh factor.
454
Unit VI Blood Cells, Immunity, and Blood Clotting
Characteristics of Rh Transfusion Reactions. If an Rh-
passed from the baby’s bone marrow into the circula-
negative person has never before been exposed to Rh-
tory system, and it is because of the presence of these
positive blood, transfusion of Rh-positive blood into
nucleated blastic red blood cells that the disease is
that person will likely cause no immediate reaction.
called erythroblastosis fetalis.
However, anti-Rh antibodies can develop in sufficient
Although the severe anemia of erythroblastosis
quantities during the next 2 to 4 weeks to cause agglu-
fetalis is usually the cause of death, many children who
tination of those transfused cells that are still circulat-
barely survive the anemia exhibit permanent mental
ing in the blood. These cells are then hemolyzed by the
impairment or damage to motor areas of the brain
tissue macrophage system. Thus, a delayed transfusion
because of precipitation of bilirubin in the neuronal
reaction occurs, although it is usually mild. On subse-
cells, causing destruction of many, a condition called
quent transfusion of Rh-positive blood into the same
kernicterus.
person, who is now already immunized against the Rh
factor, the transfusion reaction is greatly enhanced and
Treatment of the Erythroblastotic Neonate. One treatment
can be immediate and as severe as a transfusion reac-
for erythroblastosis fetalis is to replace the neonate’s
tion caused by mismatched type A or B blood.
blood with Rh-negative blood. About 400 milliliters of
Rh-negative blood is infused over a period of 1.5 or
Erythroblastosis Fetalis (“Hemolytic Disease
more hours while the neonate’s own Rh-positive blood
of the Newborn”)
is being removed. This procedure may be repeated
Erythroblastosis fetalis is a disease of the fetus and
several times during the first few weeks of life, mainly
newborn child characterized by agglutination and
to keep the bilirubin level low and thereby prevent
phagocytosis of the fetus’s red blood cells. In most
kernicterus. By the time these transfused Rh-negative
instances of erythroblastosis fetalis, the mother is Rh
cells are replaced with the infant’s own Rh-positive
negative and the father Rh positive. The baby has
cells, a process that requires 6 or more weeks, the anti-
inherited the Rh-positive antigen from the father, and
Rh agglutinins that had come from the mother will
the mother develops anti-Rh agglutinins from expo-
have been destroyed.
sure to the fetus’s Rh antigen. In turn, the mother’s
agglutinins diffuse through the placenta into the fetus
Prevention of Erythroblastosis Fetalis. The D antigen of
and cause red blood cell agglutination.
the Rh blood group system is the primary culprit in
causing immunization of an Rh-negative mother to
Incidence of the Disease. An Rh-negative mother having
an Rh-positive fetus. In the 1970’s, a dramatic reduc-
her first Rh-positive child usually does not develop
tion in the incidence of erythroblastosis fetalis was
sufficient anti-Rh agglutinins to cause any harm.
achieved with the development of Rh immunoglobu-
However, about 3 per cent of second Rh-positive
lin globin, an anti-D antibody that is administered to
babies exhibit some signs of erythroblastosis fetalis;
the expectant mother starting at 28 to 30 weeks of
about 10 per cent of third babies exhibit the disease;
gestation. The anti-D antibody is also administered to
and the incidence rises progressively with subsequent
Rh-negative women who deliver Rh-positive babies to
pregnancies.
prevent sensitization of the mothers to the D antigen.
This greatly reduces the risk of developing large
Effect of the Mother’s Antibodies on the Fetus. After anti-
amounts of D antibodies during the second pregnancy.
Rh antibodies have formed in the mother, they diffuse
The mechanism by which Rh immunoglobulin
slowly through the placental membrane into the
globin prevents sensitization of the D antigen is not
fetus’s blood. There they cause agglutination of the
completely understood, but one effect of the anti-D
fetus’s blood. The agglutinated red blood cells subse-
antibody is to inhibit antigen-induced B lymphocyte
quently hemolyze, releasing hemoglobin into the
antibody production in the expectant mother. The
blood. The fetus’s macrophages then convert the
administered anti-D antibody also attaches to D-
hemoglobin into bilirubin, which causes the baby’s
antigen sites on Rh-positive fetal red blood cells that
skin to become yellow (jaundiced). The antibodies can
may cross the placenta and enter the circulation of
also attack and damage other cells of the body.
the expectant mother, thereby interfering with the
immune response to the D antigen.
Clinical Picture of Erythroblastosis. The jaundiced, ery-
throblastotic newborn baby is usually anemic at birth,
and the anti-Rh agglutinins from the mother usually
Transfusion Reactions Resulting
circulate in the infant’s blood for another
1 to 2
from Mismatched Blood Types
months after birth, destroying more and more red
blood cells.
If donor blood of one blood type is transfused into a
The hematopoietic tissues of the infant attempt to
recipient who has another blood type, a transfusion
replace the hemolyzed red blood cells. The liver and
reaction is likely to occur in which the red blood cells
spleen become greatly enlarged and produce red
of the donor blood are agglutinated. It is rare that the
blood cells in the same manner that they normally do
transfused blood causes agglutination of the recipient’s
during the middle of gestation. Because of the rapid
cells, for the following reason: The plasma portion of
production of red cells, many early forms of red blood
the donor blood immediately becomes diluted by all
cells, including many nucleated blastic forms, are
the plasma of the recipient, thereby decreasing the
Chapter 35
Blood Types; Transfusion; Tissue and Organ Transplantation
455
titer of the infused agglutinins to a level usually too
its own additional complement of antigens. Conse-
low to cause agglutination. Conversely, the small
quently, foreign cells transplanted anywhere into the
amount of infused blood does not significantly dilute
body of a recipient can produce immune reactions. In
the agglutinins in the recipient’s plasma. Therefore, the
other words, most recipients are just as able to resist
recipient’s agglutinins can still agglutinate the mis-
invasion by foreign tissue cells as to resist invasion by
matched donor cells.
foreign bacteria or red cells.
As explained earlier, all transfusion reactions even-
tually cause either immediate hemolysis resulting from
Autografts, Isografts, Allografts, and Xenografts. A trans-
hemolysins or later hemolysis resulting from phagocy-
plant of a tissue or whole organ from one part of the
tosis of agglutinated cells. The hemoglobin released
same animal to another part is called an autograft;
from the red cells is then converted by the phagocytes
from one identical twin to another, an isograft; from
into bilirubin and later excreted in the bile by the liver,
one human being to another or from any animal to
as discussed in Chapter 70. The concentration of biliru-
another animal of the same species, an allograft; and
bin in the body fluids often rises high enough to cause
from a lower animal to a human being or from an
jaundice—that is, the person’s internal tissues and skin
animal of one species to one of another species, a
become colored with yellow bile pigment. But if liver
xenograft.
function is normal, the bile pigment will be excreted
into the intestines by way of the liver bile, so that jaun-
Transplantation of Cellular Tissues. In the case of auto-
dice usually does not appear in an adult person unless
grafts and isografts, cells in the transplant contain vir-
more than 400 milliliters of blood is hemolyzed in less
tually the same types of antigens as in the tissues of
than a day.
the recipient and will almost always continue to live
normally and indefinitely if an adequate blood supply
Acute Kidney Shutdown After Transfusion Reactions. One of
is provided.
the most lethal effects of transfusion reactions is
At the other extreme, in the case of xenografts,
kidney failure, which can begin within a few minutes
immune reactions almost always occur, causing death
to few hours and continue until the person dies of
of the cells in the graft within 1 day to 5 weeks after
renal failure.
transplantation unless some specific therapy is used to
The kidney shutdown seems to result from three
prevent the immune reactions.
causes: First, the antigen-antibody reaction of the
Some of the different cellular tissues and organs that
transfusion reaction releases toxic substances from the
have been transplanted as allografts, either experi-
hemolyzing blood that cause powerful renal vasocon-
mentally or for therapeutic purposes, from one person
striction. Second, loss of circulating red cells in the
to another are skin, kidney, heart, liver, glandular
recipient, along with production of toxic substances
tissue, bone marrow, and lung. With proper “matching”
from the hemolyzed cells and from the immune reac-
of tissues between persons, many kidney allografts
tion, often causes circulatory shock. The arterial blood
have been successful for at least 5 to 15 years, and allo-
pressure falls very low, and renal blood flow and urine
graft liver and heart transplants for 1 to 15 years.
output decrease. Third, if the total amount of free
hemoglobin released into the circulating blood is
greater than the quantity that can bind with “hapto-
Attempts to Overcome Immune
globin” (a plasma protein that binds small amounts of
Reactions in Transplanted Tissue
hemoglobin), much of the excess leaks through the
glomerular membranes into the kidney tubules. If this
Because of the extreme potential importance of trans-
amount is still slight, it can be reabsorbed through the
planting certain tissues and organs, serious attempts
tubular epithelium into the blood and will cause no
have been made to prevent antigen-antibody reactions
harm; if it is great, then only a small percentage is reab-
associated with transplantation. The following specific
sorbed. Yet water continues to be reabsorbed, causing
procedures have met with some degrees of clinical or
the tubular hemoglobin concentration to rise so high
experimental success.
that the hemoglobin precipitates and blocks many of
the kidney tubules. Thus, renal vasoconstriction, circu-
Tissue Typing—The HLA Complex of Antigens
latory shock, and renal tubular blockage together
The most important antigens for causing graft rejec-
cause acute renal shutdown. If the shutdown is com-
tion are a complex called the HLA antigens. Six of
plete and fails to resolve, the patient dies within a week
these antigens are present on the tissue cell mem-
to 12 days, as explained in Chapter 31, unless treated
branes of each person, but there are about 150 dif-
with an artificial kidney.
ferent HLA antigens to choose from. Therefore, this
represents more than a trillion possible combinations.
Consequently, it is virtually impossible for two
Transplantation of Tissues
persons, except in the case of identical twins, to have
and Organs
the same six HLA antigens. Development of signifi-
cant immunity against any one of these antigens can
Most of the different antigens of red blood cells that
cause graft rejection.
cause transfusion reactions are also widely present
The HLA antigens occur on the white blood cells as
in other cells of the body, and each bodily tissue has
well as on the tissue cells. Therefore, tissue typing for
456
Unit VI Blood Cells, Immunity, and Blood Clotting
these antigens is done on the membranes of lympho-
in an immunosuppressed person, presumably because
cytes that have been separated from the person’s
the immune system is important in destroying many
blood. The lymphocytes are mixed with appropriate
early cancer cells before they can begin to proliferate.
antisera and complement; after incubation, the cells
To summarize, transplantation of living tissues in
are tested for membrane damage, usually by testing
human beings has had very limited but important
the rate of trans-membrane uptake by the lymphocytic
success. When someone does finally succeed in block-
cells of a special dye.
ing the immune response of the recipient without at
Some of the HLA antigens are not severely anti-
the same time destroying the recipient’s specific immu-
genic, for which reason a precise match of some
nity for disease, the story will change overnight.
antigens between donor and recipient is not always
essential to allow allograft acceptance. Therefore, by
obtaining the best possible match between donor and
References
recipient, the grafting procedure has become far less
hazardous. The best success has been with tissue-type
Alayash AI: Oxygen therapeutics: can we tame haemoglo-
matches between siblings and between parent and
bin? Nat Rev Drug Discov 3:152, 2004.
child. The match in identical twins is exact, so that
Altomonte M, Fonsatti E, Visintin A, Maio M: Targeted
transplants between identical twins are almost never
therapy of solid malignancies via HLA class II antigens: a
new biotherapeutic approach? Oncogene 22:6564, 2003.
rejected because of immune reactions.
Avent ND, Reid ME: The Rh blood group system: a review.
Blood 95:375, 2000.
Prevention of Graft Rejection by Suppressing
Bowman J: Thirty-five years of Rh prophylaxis. Transfusion
the Immune System
43:1661, 2003.
If the immune system were completely suppressed,
Goodnough LT, Shander A: Evolution in alternatives to
graft rejection would not occur. In fact, in an occa-
blood transfusion. Hematol J 4:87, 2003.
sional person who has serious depression of the
Gottstein R, Cooke RW: Systematic review of intravenous
immune system, grafts can be successful without the
immunoglobulin in haemolytic disease of the newborn.
use of significant therapy to prevent rejection. But in
Arch Dis Child Fetal Neonatal Ed 88:F6, 2003.
the normal person, even with the best possible tissue
Heeger PS: T-cell allorecognition and transplant rejection: a
summary and update. Am J Transplant 3:525, 2003.
typing, allografts seldom resist rejection for more than
Horn KD: The classification, recognition and significance
a few days or weeks without use of specific therapy to
of polyagglutination in transfusion medicine. Blood Rev
suppress the immune system. Furthermore, because
13:36, 1999.
the T cells are mainly the portion of the immune
Miller J, Mathew JM, Esquenazi V: Toward tolerance to
system important for killing grafted cells, their sup-
human organ transplants: a few additional corollaries and
pression is much more important than suppression of
questions. Transplantation 77:940, 2004.
plasma antibodies. Some of the therapeutic agents that
Ricordi C, Strom TB: Clinical islet transplantation: advances
have been used for this purpose include the following:
and immunological challenges. Nat Rev Immunol 4:259,
1. Glucocorticoid hormones isolated from adrenal
2004.
cortex glands (or drugs with glucocorticoid-like
Schroeder RA, Marroquin CE, Kuo PC: Tolerance and
the “Holy Grail” of transplantation. J Surg Res 111:109,
activity), which suppress the growth of all
2003.
lymphoid tissue and, therefore, decrease
Schulak JA: Steroid immunosuppression in kidney trans-
formation of antibodies and T cells.
plantation: a passing era. J Surg Res 117:154, 2004.
2. Various drugs that have a toxic effect on the
Spahn DR, Pasch T: Physiological properties of blood sub-
lymphoid system and, therefore, block formation
stitutes. News Physiol Sci 16:38, 2001.
of antibodies and T cells, especially the drug
Strober S, Lowsky RJ, Shizuru JA, et al: Approaches to trans-
azathioprine.
plantation tolerance in humans. Transplantation 77:932,
3. Cyclosporine, which has a specific inhibitory effect
2004.
on the formation of helper T cells and, therefore,
Sumpter TL, Wilkes DS: Role of autoimmunity in organ allo-
is especially efficacious in blocking the T-cell
graft rejection: a focus on immunity to type V collagen
in the pathogenesis of lung transplant rejection. Am J
rejection reaction. This has proved to be one of
Physiol Lung Cell Mol Physiol 286:L1129, 2004.
the most valuable of all the drugs because it does
Telen MJ: Red blood cell surface adhesion molecules: their
not depress some other portions of the immune
possible roles in normal human physiology and disease.
system.
Semin Hematol 37:130, 2000.
Use of these agents often leaves the person unpro-
Trigg ME: Hematopoietic stem cells. Pediatrics
113(4
tected from infectious disease; therefore, sometimes
Suppl):1051, 2004.
bacterial and viral infections become rampant. In addi-
Triulzi DJ: Specialized transfusion support for solid organ
tion, the incidence of cancer is several times as great
transplantation. Curr Opin Hematol 9:527, 2002.
C H A P T E R
3
6
Hemostasis and Blood Coagulation
Events in Hemostasis
The term hemostasis means prevention of blood
loss. Whenever a vessel is severed or ruptured,
hemostasis is achieved by several mechanisms: (1)
vascular constriction, (2) formation of a platelet
plug, (3) formation of a blood clot as a result of
blood coagulation, and
(4) eventual growth of
fibrous tissue into the blood clot to close the hole in the vessel permanently.
Vascular Constriction
Immediately after a blood vessel has been cut or ruptured, the trauma to the
vessel wall itself causes the smooth muscle in the wall to contract; this instan-
taneously reduces the flow of blood from the ruptured vessel. The contraction
results from (1) local myogenic spasm, (2) local autacoid factors from the trau-
matized tissues and blood platelets, and (3) nervous reflexes. The nervous
reflexes are initiated by pain nerve impulses or other sensory impulses that orig-
inate from the traumatized vessel or nearby tissues. However, even more vaso-
constriction probably results from local myogenic contraction of the blood
vessels initiated by direct damage to the vascular wall. And, for the smaller
vessels, the platelets are responsible for much of the vasoconstriction by releas-
ing a vasoconstrictor substance, thromboxane A
2.
The more severely a vessel is traumatized, the greater the degree of vascular
spasm. The spasm can last for many minutes or even hours, during which time
the processes of platelet plugging and blood coagulation can take place.
Formation of the Platelet Plug
If the cut in the blood vessel is very small—indeed, many very small vascular
holes do develop throughout the body each day—the cut is often sealed by a
platelet plug, rather than by a blood clot. To understand this, it is important that
we first discuss the nature of platelets themselves.
Physical and Chemical Characteristics of Platelets
Platelets (also called thrombocytes) are minute discs 1 to 4 micrometers in diam-
eter. They are formed in the bone marrow from megakaryocytes, which are
extremely large cells of the hematopoietic series in the marrow; the megakary-
ocytes fragment into the minute platelets either in the bone marrow or soon
after entering the blood, especially as they squeeze through capillaries. The
normal concentration of platelets in the blood is between 150,000 and 300,000
per microliter.
Platelets have many functional characteristics of whole cells, even though
they do not have nuclei and cannot reproduce. In their cytoplasm are such active
factors as (1) actin and myosin molecules, which are contractile proteins similar
to those found in muscle cells, and still another contractile protein, throm-
bosthenin, that can cause the platelets to contract; (2) residuals of both the
endoplasmic reticulum and the Golgi apparatus that synthesize various enzymes
and especially store large quantities of calcium ions; (3) mitochondria and
457
458
Unit VI Blood Cells, Immunity, and Blood Clotting
enzyme systems that are capable of forming adenosine
important for closing minute ruptures in very small
triphosphate
(ATP) and adenosine diphosphate
blood vessels that occur many thousands of times
(ADP);
(4)
enzyme systems that synthesize
daily. Indeed, multiple small holes through the
prostaglandins, which are local hormones that cause
endothelial cells themselves are often closed by
many vascular and other local tissue reactions; (5) an
platelets actually fusing with the endothelial cells to
important protein called fibrin-stabilizing factor, which
form additional endothelial cell membrane. A person
we discuss later in relation to blood coagulation; and
who has few blood platelets develops each day liter-
(6) a growth factor that causes vascular endothelial
ally thousands of small hemorrhagic areas under the
cells, vascular smooth muscle cells, and fibroblasts to
skin and throughout the internal tissues, but this does
multiply and grow, thus causing cellular growth that
not occur in the normal person.
eventually helps repair damaged vascular walls.
The cell membrane of the platelets is also important.
On its surface is a coat of glycoproteins that repulses
Blood Coagulation in the
adherence to normal endothelium and yet causes
Ruptured Vessel
adherence to injured areas of the vessel wall, especially
to injured endothelial cells and even more so to any
The third mechanism for hemostasis is formation of
exposed collagen from deep within the vessel wall. In
the blood clot. The clot begins to develop in 15 to 20
addition, the platelet membrane contains large
seconds if the trauma to the vascular wall has been
amounts of phospholipids that activate multiple stages
severe, and in 1 to 2 minutes if the trauma has been
in the blood-clotting process, as we discuss later.
minor. Activator substances from the traumatized vas-
Thus, the platelet is an active structure. It has a half-
cular wall, from platelets, and from blood proteins
life in the blood of 8 to 12 days, so that over several
adhering to the traumatized vascular wall initiate the
weeks its functional processes run out. Then it is
clotting process. The physical events of this process are
eliminated from the circulation mainly by the tissue
shown in Figure 36-1, and Table 36-1 lists the most
macrophage system. More than one half of the
important of the clotting factors.
platelets are removed by macrophages in the spleen,
Within 3 to 6 minutes after rupture of a vessel, if the
where the blood passes through a latticework of tight
vessel opening is not too large, the entire opening or
trabeculae.
broken end of the vessel is filled with clot. After 20
minutes to an hour, the clot retracts; this closes the
Mechanism of the Platelet Plug
vessel still further. Platelets also play an important role
Platelet repair of vascular openings is based on several
in this clot retraction, as is discussed later.
important functions of the platelet itself. When
platelets come in contact with a damaged vascular
surface, especially with collagen fibers in the vascular
Fibrous Organization or Dissolution
wall, the platelets themselves immediately change
of the Blood Clot
their own characteristics drastically. They begin to
swell; they assume irregular forms with numerous irra-
Once a blood clot has formed, it can follow one of two
diating pseudopods protruding from their surfaces;
courses: (1) It can become invaded by fibroblasts,
their contractile proteins contract forcefully and cause
the release of granules that contain multiple active
factors; they become sticky so that they adhere to col-
lagen in the tissues and to a protein called von Wille-
brand factor that leaks into the traumatized tissue
from the plasma; they secrete large quantities of ADP;
1. Severed vessel
2. Platelets agglutinate
and their enzymes form thromboxane A
2. The ADP
and thromboxane in turn act on nearby platelets to
activate them as well, and the stickiness of these addi-
tional platelets causes them to adhere to the original
activated platelets.
Therefore, at the site of any opening in a blood
vessel wall, the damaged vascular wall activates suc-
3. Fibrin appears
4. Fibrin clot forms
cessively increasing numbers of platelets that them-
selves attract more and more additional platelets, thus
forming a platelet plug. This is at first a loose plug, but
it is usually successful in blocking blood loss if the vas-
cular opening is small. Then, during the subsequent
process of blood coagulation, fibrin threads form.
5. Clot retraction occurs
These attach tightly to the platelets, thus constructing
an unyielding plug.
Figure 36-1
Clotting process in a traumatized blood vessel. (Modified from
Importance of the Platelet Mechanism for Closing Vascular
Seegers WH: Hemostatic Agents, 1948. Courtesy of Charles C
Holes. The platelet-plugging mechanism is extremely
Thomas, Publisher, Ltd., Springfield, IL.)
Chapter 36
Hemostasis and Blood Coagulation
459
Table 36-1
Prothrombin
Clotting Factors in Blood and Their Synonyms
Prothrombin
Ca++
Clotting Factor
Synonyms
activator
Fibrinogen
Factor I
Thrombin
Prothrombin
Factor II
Tissue factor
Factor III; tissue thromboplastin
Calcium
Factor IV
Factor V
Proaccelerin; labile factor; Ac-globulin
Fibrinogen
Fibrinogen monomer
(Ac-G)
Ca++
Factor VII
Serum prothrombin conversion
accelerator (SPCA); proconvertin;
Fibrin fibers
stable factor
Factor VIII
Antihemophilic factor (AHF);
Thrombin activated
antihemophilic globulin (AHG);
fibrin-stabilizing
antihemophilic factor A
factor
Factor IX
Plasma thromboplastin component
(PTC); Christmas factor;
Cross-linked fibrin fibers
antihemophilic factor B
Factor X
Stuart factor; Stuart-Prower factor
Factor XI
Plasma thromboplastin antecedent
(PTA); antihemophilic factor C
Figure 36-2
Factor XII
Hageman factor
Factor XIII
Fibrin-stabilizing factor
Schema for conversion of prothrombin to thrombin and polymer-
Prekallikrein
Fletcher factor
ization of fibrinogen to form fibrin fibers.
High-molecular-weight
Fitzgerald factor; HMWK
kininogen
(high-molecular-weight) kininogen
Platelets
three essential steps: (1) In response to rupture of the
vessel or damage to the blood itself, a complex cascade
of chemical reactions occurs in the blood involving
which subsequently form connective tissue all through
more than a dozen blood coagulation factors. The net
the clot, or (2) it can dissolve. The usual course for a
result is formation of a complex of activated sub-
clot that forms in a small hole of a vessel wall is inva-
stances collectively called prothrombin activator. (2)
sion by fibroblasts, beginning within a few hours after
The prothrombin activator catalyzes conversion of
the clot is formed (which is promoted at least partially
prothrombin into thrombin. (3) The thrombin acts as
by growth factor secreted by platelets). This continues
an enzyme to convert fibrinogen into fibrin fibers that
to complete organization of the clot into fibrous tissue
enmesh platelets, blood cells, and plasma to form the
within about 1 to 2 weeks.
clot.
Conversely, when excess blood has leaked into the
Let us discuss first the mechanism by which the
tissues and tissue clots have occurred where they are
blood clot itself is formed, beginning with conversion
not needed, special substances within the clot itself
of prothrombin to thrombin; then we will come back
usually become activated. These function as enzymes
to the initiating stages in the clotting process by which
to dissolve the clot, as discussed later in the chapter.
prothrombin activator is formed.
Mechanism of Blood
Conversion of Prothrombin
Coagulation
to Thrombin
Basic Theory. More than 50 important substances that
First, prothrombin activator is formed as a result of
cause or affect blood coagulation have been found in
rupture of a blood vessel or as a result of damage
the blood and in the tissues—some that promote coag-
to special substances in the blood. Second, the pro-
ulation, called procoagulants, and others that inhibit
thrombin activator, in the presence of sufficient
coagulation, called anticoagulants. Whether blood will
amounts of ionic Ca++, causes conversion of pro-
coagulate depends on the balance between these two
thrombin to thrombin (Figure 36-2). Third, the throm-
groups of substances. In the blood stream, the anti-
bin causes polymerization of fibrinogen molecules into
coagulants normally predominate, so that the blood
fibrin fibers within another 10 to 15 seconds. Thus, the
does not coagulate while it is circulating in the blood
rate-limiting factor in causing blood coagulation is
vessels. But when a vessel is ruptured, procoagulants
usually the formation of prothrombin activator and
from the area of tissue damage become “activated”
not the subsequent reactions beyond that point,
and override the anticoagulants, and then a clot does
because these terminal steps normally occur rapidly to
develop.
form the clot itself.
Platelets also play an important role in the con-
General Mechanism. All research workers in the field of
version of prothrombin to thrombin because much
blood coagulation agree that clotting takes place in
of the prothrombin first attaches to prothrombin
460
Unit VI Blood Cells, Immunity, and Blood Clotting
receptors on the platelets already bound to the
This involves a substance called fibrin-stabilizing
damaged tissue.
factor that is present in small amounts in normal
plasma globulins but is also released from platelets
Prothrombin and Thrombin. Prothrombin is a plasma
entrapped in the clot. Before fibrin-stabilizing factor
protein, an alpha2-globulin, having a molecular weight
can have an effect on the fibrin fibers, it must itself be
of 68,700. It is present in normal plasma in a concen-
activated. The same thrombin that causes fibrin for-
tration of about 15 mg/dl. It is an unstable protein that
mation also activates the fibrin-stabilizing factor.
can split easily into smaller compounds, one of which
Then this activated substance operates as an enzyme
is thrombin, which has a molecular weight of 33,700,
to cause covalent bonds between more and more of the
almost exactly one half that of prothrombin.
fibrin monomer molecules, as well as multiple cross-
Prothrombin is formed continually by the liver, and
linkages between adjacent fibrin fibers, thus adding
it is continually being used throughout the body for
tremendously to the three-dimensional strength of the
blood clotting. If the liver fails to produce prothrom-
fibrin meshwork.
bin, in a day or so prothrombin concentration in
the plasma falls too low to provide normal blood
Blood Clot. The clot is composed of a meshwork of
coagulation.
fibrin fibers running in all directions and entrapping
Vitamin K is required by the liver for normal for-
blood cells, platelets, and plasma. The fibrin fibers also
mation of prothrombin as well as for formation of a
adhere to damaged surfaces of blood vessels; there-
few other clotting factors. Therefore, either lack of
fore, the blood clot becomes adherent to any vascular
vitamin K or the presence of liver disease that pre-
opening and thereby prevents further blood loss.
vents normal prothrombin formation can decrease the
prothrombin level so low that a bleeding tendency
Clot Retraction—Serum. Within a few minutes after a
results.
clot is formed, it begins to contract and usually
expresses most of the fluid from the clot within 20 to
60 minutes. The fluid expressed is called serum because
Conversion of Fibrinogen to Fibrin—
all its fibrinogen and most of the other clotting factors
Formation of the Clot
have been removed; in this way, serum differs from
plasma. Serum cannot clot because it lacks these
Fibrinogen. Fibrinogen is a high-molecular-weight
factors.
protein (MW = 340,000) that occurs in the plasma in
Platelets are necessary for clot retraction to occur.
quantities of 100 to 700 mg/dl. Fibrinogen is formed in
Therefore, failure of clot retraction is an indication
the liver, and liver disease can decrease the concen-
that the number of platelets in the circulating blood
tration of circulating fibrinogen, as it does the concen-
might be low. Electron micrographs of platelets in
tration of prothrombin, pointed out above.
blood clots show that they become attached to the
Because of its large molecular size, little fibrinogen
fibrin fibers in such a way that they actually bond
normally leaks from the blood vessels into the inter-
different fibers together. Furthermore, platelets
stitial fluids, and because fibrinogen is one of the essen-
entrapped in the clot continue to release procoagulant
tial factors in the coagulation process, interstitial fluids
substances, one of the most important of which is
ordinarily do not coagulate. Yet, when the permeabil-
fibrin-stabilizing factor, which causes more and more
ity of the capillaries becomes pathologically increased,
cross-linking bonds between adjacent fibrin fibers. In
fibrinogen does then leak into the tissue fluids in suf-
addition, the platelets themselves contribute directly
ficient quantities to allow clotting of these fluids in
to clot contraction by activating platelet throm-
much the same way that plasma and whole blood can
bosthenin, actin, and myosin molecules, which are all
clot.
contractile proteins in the platelets and cause strong
contraction of the platelet spicules attached to the
Action of Thrombin on Fibrinogen to Form Fibrin. Thrombin
fibrin. This also helps compress the fibrin meshwork
is a protein enzyme with weak proteolytic capabilities.
into a smaller mass. The contraction is activated and
It acts on fibrinogen to remove four low-molecular-
accelerated by thrombin as well as by calcium ions
weight peptides from each molecule of fibrinogen,
released from calcium stores in the mitochondria,
forming one molecule of fibrin monomer that has the
endoplasmic reticulum, and Golgi apparatus of the
automatic capability to polymerize with other fibrin
platelets.
monomer molecules to form fibrin fibers. Therefore,
As the clot retracts, the edges of the broken blood
many fibrin monomer molecules polymerize within
vessel are pulled together, thus contributing still
seconds into long fibrin fibers that constitute the retic-
further to the ultimate state of hemostasis.
ulum of the blood clot.
In the early stages of polymerization, the fibrin
monomer molecules are held together by weak non-
Vicious Circle of Clot Formation
covalent hydrogen bonding, and the newly forming
fibers are not cross-linked with one another; therefore,
Once a blood clot has started to develop, it normally
the resultant clot is weak and can be broken apart with
extends within minutes into the surrounding blood.
ease. But another process occurs during the next few
That is, the clot itself initiates a vicious circle (positive
minutes that greatly strengthens the fibrin reticulum.
feedback) to promote more clotting. One of the most
Chapter 36
Hemostasis and Blood Coagulation
461
important causes of this is the fact that the proteolytic
Prothrombin activator is generally considered to be
action of thrombin allows it to act on many of the
formed in two ways, although, in reality, the two ways
other blood-clotting factors in addition to fibrinogen.
interact constantly with each other: (1) by the extrin-
For instance, thrombin has a direct proteolytic effect
sic pathway that begins with trauma to the vascular
on prothrombin itself, tending to convert this into still
wall and surrounding tissues and (2) by the intrinsic
more thrombin, and it acts on some of the blood-clot-
pathway that begins in the blood itself.
ting factors responsible for formation of prothrombin
In both the extrinsic and the intrinsic pathways, a
activator. (These effects, discussed in subsequent para-
series of different plasma proteins called blood-
graphs, include acceleration of the actions of Factors
clotting factors play major roles. Most of these are inac-
VIII, IX, X, XI, and XII and aggregation of platelets.)
tive forms of proteolytic enzymes. When converted to
Once a critical amount of thrombin is formed, a vicious
the active forms, their enzymatic actions cause the suc-
circle develops that causes still more blood clotting
cessive, cascading reactions of the clotting process.
and more and more thrombin to be formed; thus,
Most of the clotting factors, which are listed in Table
the blood clot continues to grow until blood leakage
36-1, are designated by Roman numerals. To indicate
ceases.
the activated form of the factor, a small letter “a” is
added after the Roman numeral, such as Factor VIIIa
to indicate the activated state of Factor VIII.
Initiation of Coagulation: Formation
of Prothrombin Activator
Extrinsic Pathway for Initiating Clotting
The extrinsic pathway for initiating the formation of
Now that we have discussed the clotting process itself,
prothrombin activator begins with a traumatized vas-
we must turn to the more complex mechanisms that
cular wall or traumatized extravascular tissues that
initiate clotting in the first place. These mechanisms
come in contact with the blood. This leads to the fol-
are set into play by (1) trauma to the vascular wall and
lowing steps, as shown in Figure 36-3:
adjacent tissues, (2) trauma to the blood, or (3) contact
1. Release of tissue factor. Traumatized tissue
of the blood with damaged endothelial cells or with
releases a complex of several factors called tissue
collagen and other tissue elements outside the blood
factor or tissue thromboplastin. This factor is
vessel. In each instance, this leads to the formation of
composed especially of phospholipids from the
prothrombin activator, which then causes prothrombin
membranes of the tissue plus a lipoprotein
conversion to thrombin and all the subsequent clotting
complex that functions mainly as a proteolytic
steps.
enzyme.
(1)
Tissue trauma
Tissue factor
(2)
Vll
VIIa
X
Activated X (Xa)
Ca++
Ca++
V
Prothrombin
(3)
Activator
Platelet
phospholipids
Prothrombin
Thrombin
Ca++
Figure 36-3
Extrinsic pathway for initiating blood clotting.
462
Unit VI Blood Cells, Immunity, and Blood Clotting
2. Activation of Factor X—role of Factor VII and
accelerator of prothrombin activation. Thus, in the
tissue factor. The lipoprotein complex of tissue
final prothrombin activator complex, activated
factor further complexes with blood coagulation
Factor X is the actual protease that causes
Factor VII and, in the presence of calcium ions,
splitting of prothrombin to form thrombin;
acts enzymatically on Factor X to form activated
activated Factor V greatly accelerates this
Factor X (Xa).
protease activity, and platelet phospholipids act as
3. Effect of activated Factor X (Xa) to form
a vehicle that further accelerates the process. Note
prothrombin activator—role of Factor V. The
especially the positive feedback effect of thrombin,
activated Factor X combines immediately with
acting through Factor V, to accelerate the entire
tissue phospholipids that are part of tissue factor
process once it begins.
or with additional phospholipids released from
platelets as well as with Factor V to form the
Intrinsic Pathway for Initiating Clotting
complex called prothrombin activator. Within a
The second mechanism for initiating formation of pro-
few seconds, in the presence of calcium ions
thrombin activator, and therefore for initiating clot-
(Ca++), this splits prothrombin to form thrombin,
ting, begins with trauma to the blood itself or exposure
and the clotting process proceeds as already
of the blood to collagen from a traumatized blood
explained. At first, the Factor V in the
vessel wall. Then the process continues through the
prothrombin activator complex is inactive, but
series of cascading reactions shown in Figure 36-4.
once clotting begins and thrombin begins to form,
1. Blood trauma causes (1) activation of Factor XII
the proteolytic action of thrombin activates Factor
and (2) release of platelet phospholipids. Trauma
V. This then becomes an additional strong
to the blood or exposure of the blood to vascular
Blood trauma or
contact with collagen
(1)
XII
Activated XII (XIIa)
(HMW kininogen, prekallikrein)
(2)
XI
Activated XI (XIa)
Ca++
(3)
IX
Activated IX (IXa)
VIII
Thrombin
VIIIa
Ca++
(4)
X
Activated X (Xa)
Platelet
(5)
phospholipids
Thrombin
Ca++
V
Prothrombin
Activator
Platelet
phospholipids
Prothrombin
Thrombin
Ca++
Figure 36-4
Intrinsic pathway
for
initiating
blood clotting.
Chapter 36
Hemostasis and Blood Coagulation
463
wall collagen alters two important clotting factors
Interaction Between the Extrinsic
in the blood: Factor XII and the platelets. When
and Intrinsic Pathways—Summary
Factor XII is disturbed, such as by coming into
of Blood-Clotting Initiation
contact with collagen or with a wettable surface
It is clear from the schemas of the intrinsic and extrin-
such as glass, it takes on a new molecular
sic systems that after blood vessels rupture, clotting
configuration that converts it into a proteolytic
occurs by both pathways simultaneously. Tissue factor
enzyme called “activated Factor XII.”
initiates the extrinsic pathway, whereas contact of
Simultaneously, the blood trauma also damages
Factor XII and platelets with collagen in the vascular
the platelets because of adherence to either
wall initiates the intrinsic pathway.
collagen or a wettable surface (or by damage
An especially important difference between the
in other ways), and this releases platelet
extrinsic and intrinsic pathways is that the extrinsic
phospholipids that contain the lipoprotein called
pathway can be explosive; once initiated, its speed of
platelet factor 3, which also plays a role in
completion to the final clot is limited only by the
subsequent clotting reactions.
amount of tissue factor released from the traumatized
2.
Activation of Factor XI. The activated Factor XII
tissues and by the quantities of Factors X, VII, and V
acts enzymatically on Factor XI to activate this
in the blood. With severe tissue trauma, clotting can
factor as well, which is the second step in the
occur in as little as 15 seconds. The intrinsic pathway
intrinsic pathway. This reaction also requires
is much slower to proceed, usually requiring 1 to 6
HMW (high-molecular-weight) kininogen and is
minutes to cause clotting.
accelerated by prekallikrein.
3.
Activation of Factor IX by activated Factor XI.
Prevention of Blood Clotting
The activated Factor XI then acts enzymatically
on Factor IX to activate this factor also.
in the Normal Vascular System—
4.
Activation of Factor X—role of Factor VIII.
Intravascular Anticoagulants
The activated Factor IX, acting in concert with
activated Factor VIII and with the platelet
Endothelial Surface Factors. Probably the most important
phospholipids and factor 3 from the traumatized
factors for preventing clotting in the normal vascular
platelets, activates Factor X. It is clear that when
system are (1) the smoothness of the endothelial cell
either Factor VIII or platelets are in short supply,
surface, which prevents contact activation of the intrin-
this step is deficient. Factor VIII is the factor that
sic clotting system; (2) a layer of glycocalyx on the
is missing in a person who has classic hemophilia,
endothelium
(glycocalyx is a mucopolysaccharide
for which reason it is called antihemophilic factor.
adsorbed to the surfaces of the endothelial cells),
Platelets are the clotting factor that is lacking in
which repels clotting factors and platelets, thereby pre-
the bleeding disease called thrombocytopenia.
venting activation of clotting; and (3) a protein bound
5.
Action of activated Factor X to form prothrombin
with the endothelial membrane, thrombomodulin,
activator—role of Factor V. This step in the
which binds thrombin. Not only does the binding of
intrinsic pathway is the same as the last step in
thrombin with thrombomodulin slow the clotting
the extrinsic pathway. That is, activated Factor X
process by removing thrombin, but the thrombomod-
combines with Factor V and platelet or tissue
ulin-thrombin complex also activates a plasma protein,
phospholipids to form the complex called
protein C, that acts as an anticoagulant by inactivating
prothrombin activator. The prothrombin activator
activated Factors V and VIII.
in turn initiates within seconds the cleavage of
When the endothelial wall is damaged, its smooth-
prothrombin to form thrombin, thereby setting
ness and its glycocalyx-thrombomodulin layer are lost,
into motion the final clotting process, as described
which activates both Factor XII and the platelets,
earlier.
thus setting off the intrinsic pathway of clotting. If
Factor XII and platelets come in contact with the
Role of Calcium Ions in the Intrinsic and
subendothelial collagen, the activation is even more
Extrinsic Pathways
powerful.
Except for the first two steps in the intrinsic pathway,
calcium ions are required for promotion or accelera-
Antithrombin Action of Fibrin and Antithrombin III. Among
tion of all the blood-clotting reactions. Therefore, in
the most important anticoagulants in the blood itself
the absence of calcium ions, blood clotting by either
are those that remove thrombin from the blood. The
pathway does not occur.
most powerful of these are (1) the fibrin fibers that
In the living body, the calcium ion concentration
themselves are formed during the process of clotting
seldom falls low enough to significantly affect the
and (2) an alpha-globulin called antithrombin III or
kinetics of blood clotting. But, when blood is removed
antithrombin-heparin cofactor.
from a person, it can be prevented from clotting by
While a clot is forming, about 85 to 90 per cent of
reducing the calcium ion concentration below the
the thrombin formed from the prothrombin becomes
threshold level for clotting, either by deionizing the
adsorbed to the fibrin fibers as they develop. This helps
calcium by causing it to react with substances such as
prevent the spread of thrombin into the remaining
citrate ion or by precipitating the calcium with sub-
blood and, therefore, prevents excessive spread of the
stances such as oxalate ion.
clot.
464
Unit VI Blood Cells, Immunity, and Blood Clotting
The thrombin that does not adsorb to the fibrin
is trapped in the clot along with other plasma proteins.
fibers soon combines with antithrombin III, which
This will not become plasmin or cause lysis of the clot
further blocks the effect of the thrombin on the fi-
until it is activated. The injured tissues and vascular
brinogen and then also inactivates the thrombin itself
endothelium very slowly release a powerful activator
during the next 12 to 20 minutes.
called tissue plasminogen activator (t-PA) that a few
days later, after the clot has stopped the bleeding,
Heparin. Heparin is another powerful anticoagulant,
eventually converts plasminogen to plasmin, which in
but its concentration in the blood is normally low, so
turn removes the remaining unnecessary blood clot. In
that only under special physiologic conditions does
fact, many small blood vessels in which blood flow has
it have significant anticoagulant effects. However,
been blocked by clots are reopened by this mecha-
heparin is used widely as a pharmacological agent in
nism. Thus, an especially important function of the
medical practice in much higher concentrations to
plasmin system is to remove minute clots from millions
prevent intravascular clotting.
of tiny peripheral vessels that eventually would
The heparin molecule is a highly negatively charged
become occluded were there no way to clear them.
conjugated polysaccharide. By itself, it has little or no
anticoagulant properties, but when it combines with
antithrombin III, the effectiveness of antithrombin III
Conditions That Cause
for removing thrombin increases by a hundredfold to
Excessive Bleeding in
a thousandfold, and thus it acts as an anticoagulant.
Therefore, in the presence of excess heparin, removal
Human Beings
of free thrombin from the circulating blood by
antithrombin III is almost instantaneous.
Excessive bleeding can result from deficiency of any
The complex of heparin and antithrombin III
one of the many blood-clotting factors. Three particu-
removes several other activated coagulation factors in
lar types of bleeding tendencies that have been studied
addition to thrombin, further enhancing the effective-
to the greatest extent are discussed here: bleeding
ness of anticoagulation. The others include activated
caused by (1) vitamin K deficiency, (2) hemophilia, and
Factors XII, XI, X, and IX.
(3) thrombocytopenia (platelet deficiency).
Heparin is produced by many different cells of the
body, but especially large quantities are formed by the
basophilic mast cells located in the pericapillary con-
Decreased Prothrombin, Factor VII,
nective tissue throughout the body. These cells contin-
Factor IX, and Factor X Caused
ually secrete small quantities of heparin that diffuse
by Vitamin K Deficiency
into the circulatory system. The basophil cells of the
blood, which are functionally almost identical to the
With few exceptions, almost all the blood-clotting
mast cells, release small quantities of heparin into
factors are formed by the liver. Therefore, diseases of
the plasma.
the liver such as hepatitis, cirrhosis, and acute yellow
Mast cells are abundant in tissue surrounding the
atrophy can sometimes depress the clotting system so
capillaries of the lungs and to a lesser extent capillar-
greatly that the patient develops a severe tendency to
ies of the liver. It is easy to understand why large quan-
bleed.
tities of heparin might be needed in these areas
Another cause of depressed formation of clotting
because the capillaries of the lungs and liver receive
factors by the liver is vitamin K deficiency. Vitamin K
many embolic clots formed in slowly flowing venous
is necessary for liver formation of five of the impor-
blood; sufficient formation of heparin prevents further
tant clotting factors: prothrombin, Factor VII, Factor
growth of the clots.
IX, Factor X, and protein C. In the absence of vitamin
K, subsequent insufficiency of these coagulation
factors in the blood can lead to serious bleeding ten-
Lysis of Blood Clots—Plasmin
dencies.
Vitamin K is continually synthesized in the intes-
The plasma proteins contain a euglobulin called
tinal tract by bacteria, so that vitamin K deficiency
plasminogen (or profibrinolysin) that, when activated,
seldom occurs in the normal person as a result of
becomes a substance called plasmin (or fibrinolysin).
vitamin K absence from the diet (except in neonates
Plasmin is a proteolytic enzyme that resembles trypsin,
before they establish their intestinal bacterial flora).
the most important proteolytic digestive enzyme of
However, in gastrointestinal disease, vitamin K defi-
pancreatic secretion. Plasmin digests fibrin fibers and
ciency often occurs as a result of poor absorption of
some other protein coagulants such as fibrinogen,
fats from the gastrointestinal tract. The reason is that
Factor V, Factor VIII, prothrombin, and Factor XII.
vitamin K is fat-soluble and ordinarily is absorbed into
Therefore, whenever plasmin is formed, it can cause
the blood along with the fats.
lysis of a clot by destroying many of the clotting
One of the most prevalent causes of vitamin K defi-
factors, thereby sometimes even causing hypocoagula-
ciency is failure of the liver to secrete bile into the gas-
bility of the blood.
trointestinal tract (which occurs either as a result of
Activation of Plasminogen to Form Plasmin: Then Lysis of Clots.
obstruction of the bile ducts or as a result of liver
When a clot is formed, a large amount of plasminogen
disease). Lack of bile prevents adequate fat digestion
Chapter 36
Hemostasis and Blood Coagulation
465
and absorption and, therefore, depresses vitamin K
with thrombocytopenia have a tendency to bleed, as
absorption as well. Thus, liver disease often causes
do hemophiliacs, except that the bleeding is usually
decreased production of prothrombin and some other
from many small venules or capillaries, rather than
clotting factors both because of poor vitamin K
from larger vessels as in hemophilia. As a result, small
absorption and because of the diseased liver cells.
punctate hemorrhages occur throughout all the body
Because of this, vitamin K is injected into all surgical
tissues. The skin of such a person displays many small,
patients with liver disease or with obstructed bile ducts
purplish blotches, giving the disease the name throm-
before performing the surgical procedure. Ordinarily,
bocytopenic purpura. As stated earlier, platelets are
if vitamin K is given to a deficient patient 4 to 8 hours
especially important for repair of minute breaks in
before the operation and the liver parenchymal cells
capillaries and other small vessels.
are at least one-half normal in function, sufficient
Ordinarily, bleeding will not occur until the number
clotting factors will be produced to prevent excessive
of platelets in the blood falls below 50,000/ml, rather
bleeding during the operation.
than the normal 150,000 to 300,000. Levels as low as
10,000/ml are frequently lethal.
Even without making specific platelet counts in the
Hemophilia
blood, sometimes one can suspect the existence of
thrombocytopenia if the person’s blood fails to retract,
Hemophilia is a bleeding disease that occurs almost
because, as pointed out earlier, clot retraction is nor-
exclusively in males. In 85 per cent of cases, it is caused
mally dependent on release of multiple coagulation
by an abnormality or deficiency of Factor VIII; this
factors from the large numbers of platelets entrapped
type of hemophilia is called hemophilia A or classic
in the fibrin mesh of the clot.
hemophilia. About 1 of every 10,000 males in the
Most people with thrombocytopenia have the
United States has classic hemophilia. In the other 15
disease known as idiopathic thrombocytopenia, which
per cent of hemophilia patients, the bleeding tendency
means thrombocytopenia of unknown cause. In most
is caused by deficiency of Factor IX. Both of these
of these people, it has been discovered that for
factors are transmitted genetically by way of the
unknown reasons, specific antibodies have formed and
female chromosome. Therefore, almost never will a
react against the platelets themselves to destroy them.
woman have hemophilia because at least one of her
Relief from bleeding for 1 to 4 days can often be
two X chromosomes will have the appropriate genes.
effected in a patient with thrombocytopenia by
If one of her X chromosomes is deficient, she will be
giving fresh whole blood transfusions that contain
a hemophilia carrier, transmitting the disease to half of
large numbers of platelets. Also, splenectomy is often
her male offspring and transmitting the carrier state to
helpful, sometimes effecting almost complete cure
half of her female offspring.
because the spleen normally removes large numbers
The bleeding trait in hemophilia can have various
of platelets from the blood.
degrees of severity, depending on the character of the
genetic deficiency. Bleeding usually does not occur
except after trauma, but in some patients, the degree
of trauma required to cause severe and prolonged
Thromboembolic Conditions
bleeding may be so mild that it is hardly noticeable.
in the Human Being
For instance, bleeding can often last for days after
extraction of a tooth.
Thrombi and Emboli. An abnormal clot that develops in
Factor VIII has two active components, a large com-
a blood vessel is called a thrombus. Once a clot has
ponent with a molecular weight in the millions and a
developed, continued flow of blood past the clot is
smaller component with a molecular weight of about
likely to break it away from its attachment and cause
230,000. The smaller component is most important in
the clot to flow with the blood; such freely flowing clots
the intrinsic pathway for clotting, and it is deficiency
are known as emboli. Also, emboli that originate in
of this part of Factor VIII that causes classic hemo-
large arteries or in the left side of the heart can flow
philia. Another bleeding disease with somewhat dif-
peripherally and plug arteries or arterioles in the
ferent characteristics, called von Willebrand’s disease,
brain, kidneys, or elsewhere. Emboli that originate in
results from loss of the large component.
the venous system or in the right side of the heart gen-
When a person with classic hemophilia experiences
erally flow into the lungs to cause pulmonary arterial
severe prolonged bleeding, almost the only therapy
embolism.
that is truly effective is injection of purified Factor
VIII. The cost of Factor VIII is high, and its availabil-
ity is limited because it can be gathered only from
Cause of Thromboembolic Conditions. The causes of throm-
human blood and only in extremely small quantities.
boembolic conditions in the human being are usually
twofold: (1) Any roughened endothelial surface of a
vessel—as may be caused by arteriosclerosis, infection,
Thrombocytopenia
or trauma—is likely to initiate the clotting process. (2)
Blood often clots when it flows very slowly through
Thrombocytopenia means the presence of very low
blood vessels, where small quantities of thrombin and
numbers of platelets in the circulating blood. People
other procoagulants are always being formed.
466
Unit VI Blood Cells, Immunity, and Blood Clotting
Use of t-PA in Treating Intravascular Clots. Genetically
clotting factors are removed by the widespread clot-
engineered t-PA
(tissue plasminogen activator) is
ting that too few procoagulants remain to allow
available. When delivered directly to a thrombosed
normal hemostasis of the remaining blood.
area through a catheter, it is effective in activating
plasminogen to plasmin, which in turn can dissolve
some intravascular clots. For instance, if used within
Anticoagulants for
the first hour or so after thrombotic occlusion of a
Clinical Use
coronary artery, the heart is often spared serious
damage.
In some thromboembolic conditions, it is desirable to
delay the coagulation process. Various anticoagulants
have been developed for this purpose. The ones most
Femoral Venous Thrombosis and
useful clinically are heparin and the coumarins.
Massive Pulmonary Embolism
Because clotting almost always occurs when blood
Heparin as an Intravenous
flow is blocked for many hours in any vessel of the
Anticoagulant
body, the immobility of patients confined to bed plus
the practice of propping the knees with pillows often
Commercial heparin is extracted from several differ-
causes intravascular clotting because of blood stasis in
ent animal tissues and prepared in almost pure form.
one or more of the leg veins for hours at a time. Then
Injection of relatively small quantities, about 0.5 to
the clot grows, mainly in the direction of the slowly
1 mg/kg of body weight, causes the blood-clotting time
moving venous blood, sometimes growing the entire
to increase from a normal of about 6 minutes to 30 or
length of the leg veins and occasionally even up into
more minutes. Furthermore, this change in clotting
the common iliac vein and inferior vena cava. Then,
time occurs instantaneously, thereby immediately pre-
about 1 time out of every 10, a large part of the clot
venting or slowing further development of a throm-
disengages from its attachments to the vessel wall and
boembolic condition.
flows freely with the venous blood through the right
The action of heparin lasts about 1.5 to 4 hours. The
side of the heart and into the pulmonary arteries to
injected heparin is destroyed by an enzyme in the
cause massive blockage of the pulmonary arteries,
blood known as heparinase.
called massive pulmonary embolism. If the clot is large
enough to occlude both of the pulmonary arteries at
the same time, immediate death ensues. If only one
Coumarins as Anticoagulants
pulmonary artery is blocked, death may not occur, or
the embolism may lead to death a few hours to several
When a coumarin, such as warfarin, is given to a
days later because of further growth of the clot within
patient, the plasma levels of prothrombin and Factors
the pulmonary vessels. But, again, t-PA therapy can be
VII, IX, and X, all formed by the liver, begin to fall,
a lifesaver.
indicating that warfarin has a potent depressant effect
on liver formation of these compounds. Warfarin
causes this effect by competing with vitamin K for
Disseminated Intravascular
reactive sites in the enzymatic processes for formation
Coagulation
of prothrombin and the other three clotting factors,
thereby blocking the action of vitamin K.
Occasionally the clotting mechanism becomes acti-
After administration of an effective dose of war-
vated in widespread areas of the circulation, giving rise
farin, the coagulant activity of the blood decreases to
to the condition called disseminated intravascular
about 50 per cent of normal by the end of 12 hours and
coagulation. This often results from the presence of
to about 20 per cent of normal by the end of 24 hours.
large amounts of traumatized or dying tissue in the
In other words, the coagulation process is not blocked
body that releases great quantities of tissue factor into
immediately but must await the natural consumption
the blood. Frequently, the clots are small but numer-
of the prothrombin and the other affected coagulation
ous, and they plug a large share of the small periph-
factors already present in the plasma. Normal coagu-
eral blood vessels. This occurs especially in patients
lation usually returns 1 to 3 days after discontinuing
with widespread septicemia, in which either circulat-
coumarin therapy.
ing bacteria or bacterial toxins—especially endotox-
ins—activate the clotting mechanisms. Plugging of
small peripheral vessels greatly diminishes delivery of
Prevention of Blood Coagulation
oxygen and other nutrients to the tissues—a situation
Outside the Body
that leads to or exacerbates circulatory shock. It is
partly for this reason that septicemic shock is lethal in
Although blood removed from the body and held in a
85 per cent or more of patients.
glass test tube normally clots in about 6 minutes, blood
A peculiar effect of disseminated intravascular
collected in siliconized containers often does not clot
coagulation is that the patient on occasion begins
for 1 hour or more. The reason for this delay is that
to bleed. The reason for this is that so many of the
preparing the surfaces of the containers with silicone
Chapter 36
Hemostasis and Blood Coagulation
467
prevents contact activation of platelets and Factor XII,
collect blood in a chemically clean glass test tube and
the two principal factors that initiate the intrinsic clot-
then to tip the tube back and forth about every 30
ting mechanism. Conversely, untreated glass contain-
seconds until the blood has clotted. By this method,
ers allow contact activation of the platelets and Factor
the normal clotting time is 6 to 10 minutes. Procedures
XII, with rapid development of clots.
using multiple test tubes have also been devised for
Heparin can be used for preventing coagulation of
determining clotting time more accurately.
blood outside the body as well as in the body. Heparin
Unfortunately, the clotting time varies widely,
is especially used in surgical procedures in which the
depending on the method used for measuring it, so it
blood must be passed through a heart-lung machine
is no longer used in many clinics. Instead, measure-
or artificial kidney machine and then back into the
ments of the clotting factors themselves are made,
person.
using sophisticated chemical procedures.
Various substances that decrease the concentration
of calcium ions in the blood can also be used for pre-
venting blood coagulation outside the body. For
Prothrombin Time
instance, a soluble oxalate compound mixed in a very
small quantity with a sample of blood causes precipi-
Prothrombin time gives an indication of the concen-
tation of calcium oxalate from the plasma and thereby
tration of prothrombin in the blood. Figure 36-5 shows
decreases the ionic calcium level so much that blood
the relation of prothrombin concentration to pro-
coagulation is blocked.
thrombin time. The method for determining pro-
Any substance that deionizes the blood calcium will
thrombin time is the following.
prevent coagulation. The negatively charged citrate ion
Blood removed from the patient is immediately
is especially valuable for this purpose, mixed with
oxalated so that none of the prothrombin can change
blood usually in the form of sodium, ammonium, or
into thrombin. Then, a large excess of calcium ion and
potassium citrate. The citrate ion combines with
tissue factor is quickly mixed with the oxalated blood.
calcium in the blood to cause an un-ionized calcium
The excess calcium nullifies the effect of the oxalate,
compound, and the lack of ionic calcium prevents
and the tissue factor activates the prothrombin-to-
coagulation. Citrate anticoagulants have an important
thrombin reaction by means of the extrinsic clotting
advantage over the oxalate anticoagulants because
pathway. The time required for coagulation to take
oxalate is toxic to the body, whereas moderate quan-
place is known as the prothrombin time. The shortness
tities of citrate can be injected intravenously. After
of the time is determined mainly by prothrombin con-
injection, the citrate ion is removed from the blood
centration. The normal prothrombin time is about 12
within a few minutes by the liver and is polymerized
seconds. In each laboratory, a curve relating pro-
into glucose or metabolized directly for energy.
thrombin concentration to prothrombin time, such as
Consequently, 500 milliliters of blood that has been
rendered incoagulable by citrate can ordinarily be
transfused into a recipient within a few minutes
without dire consequences. But if the liver is damaged
or if large quantities of citrated blood or plasma are
100
given too rapidly (within fractions of a minute), the
citrate ion may not be removed quickly enough, and
the citrate can, under these conditions, greatly depress
the level of calcium ion in the blood, which can result
in tetany and convulsive death.
Blood Coagulation Tests
50.0
Bleeding Time
When a sharp-pointed knife is used to pierce the tip
25.0
of the finger or lobe of the ear, bleeding ordinarily lasts
for 1 to 6 minutes. The time depends largely on the
12.5
depth of the wound and the degree of hyperemia in
6.25
the finger or ear lobe at the time of the test. Lack of
0
0
10
20
30
40
50
60
any one of several of the clotting factors can prolong
the bleeding time, but it is especially prolonged by lack
Prothrombin time
(seconds)
of platelets.
Clotting Time
Figure 36-5
Many methods have been devised for determining
Relation of prothrombin concentration in the blood to “prothrom-
blood clotting times. The one most widely used is to
bin time.”
468
Unit VI Blood Cells, Immunity, and Blood Clotting
that shown in Figure 36-5, is drawn for the method
Levi M: Current understanding of disseminated intravascu-
used so that the prothrombin in the blood can be quan-
lar coagulation. Br J Haematol 124:567, 2004.
tified.
Lindsberg PJ, Kaste M: Thrombolysis for acute stroke. Curr
Opin Neurol 16:73, 2003.
Tests similar to that for prothrombin time have been
Moake JL: Thrombotic microangiopathies. N Engl J Med
devised to determine the quantities of other blood
347:589, 2002.
clotting factors. In each of these tests, excesses of
Roberts HR, Monroe DM, Escobar MA: Current concepts
calcium ions and all the other factors besides the one
of hemostasis: implications for therapy. Anesthesiology
being tested are added to oxalated blood all at once.
100:722, 2004.
Then the time required for coagulation is determined
Saenko EL, Ananyeva NM, Shima M, et al: The future of
in the same manner as for prothrombin time. If the
recombinant coagulation factors. J Thromb Haemost
factor being tested is deficient, the coagulation time is
1:922, 2003.
prolonged. The time itself can then be used to quanti-
Solum NO: Procoagulant expression in platelets and defects
tate the concentration of the factor.
leading to clinical disorders. Arterioscler Thromb Vasc
Biol 19:2841, 1999.
Toh CH, Dennis M: Disseminated intravascular coagulation:
old disease, new hope. BMJ 327:974, 2003.
References
Tsai HM: Advances in the pathogenesis, diagnosis, and treat-
ment of thrombotic thrombocytopenic purpura. J Am Soc
Brass LF: Thrombin and platelet activation. Chest 124(3
Nephrol 14:1072, 2003.
Suppl):18S, 2003.
Tsai HM: Platelet activation and the formation of the
Caprini JA, Glase CJ, Anderson CB, Hathaway K: Labora-
platelet plug: deficiency of ADAMTS13 causes thrombotic
tory markers in the diagnosis of venous thromboem-
thrombocytopenic purpura. Arterioscler Thromb Vasc
bolism. Circulation 109(12 Suppl 1):I4, 2004.
Biol 23:388, 2003.
Dorsam RT, Kunapuli SP: Central role of the P2Y12 recep-
VandenDriessche T, Collen D, Chuah MK: Gene therapy for
tor in platelet activation. J Clin Invest 113:340, 2004.
the hemophilias. J Thromb Haemost 1:1550, 2003.
Fisher M, Brott TG: Emerging therapies for acute ischemic
Vesely SK, Perdue JJ, Rizvi MA, et al: Management of adult
stroke: new therapies on trial. Stroke 34:359, 2003.
patients with persistent idiopathic thrombocytopenic
Geddis AE, Kaushansky K: Inherited thrombocytopenias:
purpura following splenectomy: a systematic review. Ann
toward a molecular understanding of disorders of platelet
Intern Med 140:112, 2004.
production. Curr Opin Pediatr 16:15, 2004.
White RH, Ginsberg JS: Low-molecular-weight heparins: are
Kahn SR, Ginsberg JS: Relationship between deep venous
they all the same? Br J Haematol 121:12, 2003.
thrombosis and the postthrombotic syndrome. Arch
Intern Med 164:17, 2004.
Koreth R, Weinert C, Weisdorf DJ, Key NS: Measurement of
bleeding severity: a critical review. Transfusion 44:605,
2004.
U N I T VII
Respiration
37. Pulmonary Ventilation
38. Pulmonary Circulation, Pulmonary Edema,
Pleural Fluid
39. Physical Principles of Gas Exchange; Diffusion
of Oxygen and Carbon Dioxide Through the
Respiratory Membrane
40. Transport of Oxygen and Carbon Dioxide in
Blood and Tissue Fluids
41. Regulation of Respiration
42. Respiratory Insufficiency—Pathophysiology,
Diagnosis, Oxygen Therapy
C H A P T E R
3
7
Pulmonary Ventilation
The goals of respiration are to provide oxygen to
the tissues and to remove carbon dioxide. To
achieve these goals, respiration can be divided into
four major functions: (1) pulmonary ventilation,
which means the inflow and outflow of air between
the atmosphere and the lung alveoli; (2) diffusion
of oxygen and carbon dioxide between the alveoli
and the blood; (3) transport of oxygen and carbon
dioxide in the blood and body fluids to and from the body’s tissue cells; and
(4) regulation of ventilation and other facets of respiration. This chapter is a
discussion of pulmonary ventilation, and the subsequent five chapters
cover other respiratory functions plus the physiology of special respiratory
abnormalities.
Mechanics of Pulmonary Ventilation
Muscles That Cause Lung Expansion and Contraction
The lungs can be expanded and contracted in two ways: (1) by downward and
upward movement of the diaphragm to lengthen or shorten the chest cavity,
and (2) by elevation and depression of the ribs to increase and decrease the
anteroposterior diameter of the chest cavity. Figure 37-1 shows these two
methods.
Normal quiet breathing is accomplished almost entirely by the first method,
that is, by movement of the diaphragm. During inspiration, contraction of the
diaphragm pulls the lower surfaces of the lungs downward. Then, during expi-
ration, the diaphragm simply relaxes, and the elastic recoil of the lungs, chest
wall, and abdominal structures compresses the lungs and expels the air. During
heavy breathing, however, the elastic forces are not powerful enough to cause
the necessary rapid expiration, so that extra force is achieved mainly by con-
traction of the abdominal muscles, which pushes the abdominal contents
upward against the bottom of the diaphragm, thereby compressing the lungs.
The second method for expanding the lungs is to raise the rib cage. This
expands the lungs because, in the natural resting position, the ribs slant down-
ward, as shown on the left side of Figure 37-1, thus allowing the sternum to fall
backward toward the vertebral column. But when the rib cage is elevated, the
ribs project almost directly forward, so that the sternum also moves forward,
away from the spine, making the anteroposterior thickness of the chest about
20 per cent greater during maximum inspiration than during expiration. There-
fore, all the muscles that elevate the chest cage are classified as muscles of inspi-
ration, and those muscles that depress the chest cage are classified as muscles
of expiration. The most important muscles that raise the rib cage are the exter-
nal intercostals, but others that help are the (1) sternocleidomastoid muscles,
which lift upward on the sternum; (2) anterior serrati, which lift many of the
ribs; and (3) scaleni, which lift the first two ribs.
The muscles that pull the rib cage downward during expiration are mainly
the (1) abdominal recti, which have the powerful effect of pulling downward on
the lower ribs at the same time that they and other abdominal muscles also com-
press the abdominal contents upward against the diaphragm, and (2) internal
intercostals.
471
472
Unit VII
Respiration
Increased
vertical diameter
Increased
Elevated
Lung volume
A-P diameter
rib cage
0.50
External
intercostals
contracted
0.25
Internal
intercostals
relaxed
0
Diaphragmatic
Abdominals
contraction
Alveolar pressure
contracted
+2
EXPIRATION
INSPIRATION
0
Figure 37-1
-2
Contraction and expansion of the thoracic cage during expiration
Transpulmonary pressure
and inspiration, demonstrating diaphragmatic contraction, func-
tion of the intercostal muscles, and elevation and depression of
-4
the rib cage.
-6
Figure 37-1 also shows the mechanism by which the
Pleural pressure
-8
external and internal intercostals act to cause inspira-
tion and expiration. To the left, the ribs during
Inspiration
Expiration
expiration are angled downward, and the external
intercostals are elongated forward and downward. As
they contract, they pull the upper ribs forward in rela-
tion to the lower ribs, and this causes leverage on the
ribs to raise them upward, thereby causing inspiration.
Figure 37-2
The internal intercostals function exactly in the oppo-
Changes in lung volume, alveolar pressure, pleural pressure, and
site manner, functioning as expiratory muscles because
transpulmonary pressure during normal breathing.
they angle between the ribs in the opposite direction
and cause opposite leverage.
is about -5 centimeters of water, which is the amount
Movement of Air In and Out of the
of suction required to hold the lungs open to their
Lungs and the Pressures
resting level. Then, during normal inspiration, expan-
That Cause the Movement
sion of the chest cage pulls outward on the lungs with
greater force and creates more negative pressure, to an
The lung is an elastic structure that collapses like a
average of about -7.5 centimeters of water.
balloon and expels all its air through the trachea when-
These relationships between pleural pressure and
ever there is no force to keep it inflated. Also, there
changing lung volume are demonstrated in Figure
are no attachments between the lung and the walls of
37-2, showing in the lower panel the increasing nega-
the chest cage, except where it is suspended at its hilum
tivity of the pleural pressure from -5 to -7.5 during
from the mediastinum. Instead, the lung “floats” in the
inspiration and in the upper panel an increase in lung
thoracic cavity, surrounded by a thin layer of pleural
volume of 0.5 liter. Then, during expiration, the events
fluid that lubricates movement of the lungs within the
are essentially reversed.
cavity. Further, continual suction of excess fluid into
lymphatic channels maintains a slight suction between
Alveolar Pressure
the visceral surface of the lung pleura and the parietal
Alveolar pressure is the pressure of the air inside the
pleural surface of the thoracic cavity. Therefore, the
lung alveoli. When the glottis is open and no air is
lungs are held to the thoracic wall as if glued there,
flowing into or out of the lungs, the pressures in all
except that they are well lubricated and can slide
parts of the respiratory tree, all the way to the alveoli,
freely as the chest expands and contracts.
are equal to atmospheric pressure, which is considered
to be zero reference pressure in the airways—that is,
Pleural Pressure and Its Changes
0 centimeters water pressure. To cause inward flow of
During Respiration
air into the alveoli during inspiration, the pressure in
Pleural pressure is the pressure of the fluid in the thin
the alveoli must fall to a value slightly below atmos-
space between the lung pleura and the chest wall
pheric pressure (below 0). The second curve (labeled
pleura. As noted earlier, this is normally a slight
“alveolar pressure”) of Figure 37-2 demonstrates that
suction, which means a slightly negative pressure. The
during normal inspiration, alveolar pressure decreases
normal pleural pressure at the beginning of inspiration
to about -1 centimeter of water. This slight negative
Chapter 37
Pulmonary Ventilation
473
pressure is enough to pull 0.5 liter of air into the lungs
between successive steps. The two curves are called,
in the 2 seconds required for normal quiet inspiration.
respectively, the inspiratory compliance curve and the
During expiration, opposite pressures occur: The
expiratory compliance curve, and the entire diagram is
alveolar pressure rises to about +1 centimeter of water,
called the compliance diagram of the lungs.
and this forces the 0.5 liter of inspired air out of the
The characteristics of the compliance diagram are
lungs during the 2 to 3 seconds of expiration.
determined by the elastic forces of the lungs. These can
be divided into two parts: (1) elastic forces of the lung
Transpulmonary Pressure. Finally, note in Figure 37-2 the
tissue itself and (2) elastic forces caused by surface
difference between the alveolar pressure and the
tension of the fluid that lines the inside walls of the
pleural pressure. This is called the transpulmonary
alveoli and other lung air spaces.
pressure. It is the pressure difference between that in
The elastic forces of the lung tissue are determined
the alveoli and that on the outer surfaces of the lungs,
mainly by elastin and collagen fibers interwoven
and it is a measure of the elastic forces in the lungs
among the lung parenchyma. In deflated lungs, these
that tend to collapse the lungs at each instant of res-
fibers are in an elastically contracted and kinked state;
piration, called the recoil pressure.
then, when the lungs expand, the fibers become
stretched and unkinked, thereby elongating and exert-
Compliance of the Lungs
ing even more elastic force.
The extent to which the lungs will expand for each unit
The elastic forces caused by surface tension are
increase in transpulmonary pressure (if enough time is
much more complex. The significance of surface
allowed to reach equilibrium) is called the lung com-
tension is shown in Figure 37-4, which compares the
pliance. The total compliance of both lungs together in
compliance diagram of the lungs when filled with
the normal adult human being averages about 200 mil-
saline solution and when filled with air. When the lungs
liliters of air per centimeter of water transpulmonary
are filled with air, there is an interface between the
pressure. That is, every time the transpulmonary pres-
alveolar fluid and the air in the alveoli. In the case of
sure increases 1 centimeter of water, the lung volume,
the saline solution-filled lungs, there is no air-fluid
after 10 to 20 seconds, will expand 200 milliliters.
interface; therefore, the surface tension effect is not
present—only tissue elastic forces are operative in the
Compliance Diagram of the Lungs. Figure 37-3 is a diagram
saline solution-filled lung.
relating lung volume changes to changes in transpul-
Note that transpleural pressures required to expand
monary pressure. Note that the relation is different for
air-filled lungs are about three times as great as those
inspiration and expiration. Each curve is recorded by
required to expand saline solution-filled lungs. Thus,
changing the transpulmonary pressure in small steps
one can conclude that the tissue elastic forces tending
and allowing the lung volume to come to a steady level
to cause collapse of the air-filled lung represent only
about one third of the total lung elasticity, whereas the
Saline-filled
Air-filled
0.50
0.50
Expiration
Expiration
0.25
0.25
Inspiration
Inspiration
0
0
0
-2
-4
-6
-8
-4
-5
-6
Pleural pressure (cm H2O)
Pleural pressure (cm H2O)
Figure 37-4
Figure 37-3
Comparison of the compliance diagrams of saline-filled and air-
Compliance diagram in a healthy person. This diagram shows
filled lungs when the alveolar pressure is maintained at atmos-
compliance of the lungs alone.
pheric pressure (0 cm H2O) and pleural pressure is changed.
474
Unit VII
Respiration
fluid-air surface tension forces in the alveoli represent
2
¥
Surface tension
Pressure
=
about two thirds.
Radius of alveolus
The fluid-air surface tension elastic forces of the
For the average-sized alveolus with a radius of about
lungs also increase tremendously when the substance
100 micrometers and lined with normal surfactant, this
called surfactant is not present in the alveolar fluid. Let
calculates to be about 4 centimeters of water pressure
us now discuss surfactant and its relation to the surface
(3 mm Hg). If the alveoli were lined with pure water
tension forces.
without any surfactant, the pressure would calculate to
be about 18 centimeters of water pressure, 4.5 times as
Surfactant, Surface Tension, and Collapse
great. Thus, one sees how important surfactant is in
of the Alveoli
reducing alveolar surface tension and therefore also
reducing the effort required by the respiratory muscles
to expand the lungs.
Principle of Surface Tension. When water forms a surface
with air, the water molecules on the surface of the
Effect of Alveolar Radius on the Pressure Caused by Surface
water have an especially strong attraction for one
Tension. Note from the preceding formula that the pres-
another. As a result, the water surface is always
sure generated as a result of surface tension in the
attempting to contract. This is what holds raindrops
alveoli is inversely affected by the radius of the alveo-
together: that is, there is a tight contractile membrane
lus, which means that the smaller the alveolus, the
of water molecules around the entire surface of the
greater the alveolar pressure caused by the surface
raindrop. Now let us reverse these principles and see
tension. Thus, when the alveoli have half the normal
what happens on the inner surfaces of the alveoli.
radius (50 instead of 100 micrometers), the pressures
noted earlier are doubled. This is especially significant
Here, the water surface is also attempting to contract.
in small premature babies, many of whom have alveoli
This results in an attempt to force the air out of the
with radii less than one quarter that of an adult person.
alveoli through the bronchi and, in doing so, causes the
Further, surfactant does not normally begin to be
alveoli to try to collapse. The net effect is to cause an
secreted into the alveoli until between the sixth and
elastic contractile force of the entire lungs, which is
seventh months of gestation, and in some cases, even
called the surface tension elastic force.
later than that. Therefore, many premature babies have
little or no surfactant in the alveoli when they are born,
and their lungs have an extreme tendency to collapse,
Surfactant and Its Effect on Surface Tension. Surfactant is a
sometimes as great as six to eight times that in a normal
surface active agent in water, which means that it
adult person. This causes the condition called respira-
greatly reduces the surface tension of water. It is
tory distress syndrome of the newborn. It is fatal if
secreted by special surfactant-secreting epithelial cells
not treated with strong measures, especially properly
called type II alveolar epithelial cells, which constitute
applied continuous positive pressure breathing.
about 10 per cent of the surface area of the alveoli.
These cells are granular, containing lipid inclusions
that are secreted in the surfactant into the alveoli.
Effect of the Thoracic Cage
Surfactant is a complex mixture of several
on Lung Expansibility
phospholipids, proteins, and ions. The most important
components are the phospholipid dipalmitoylphos-
Thus far, we have discussed the expansibility of the
phatidylcholine, surfactant apoproteins, and calcium
lungs alone, without considering the thoracic cage. The
ions. The dipalmitoylphosphatidylcholine, along with
thoracic cage has its own elastic and viscous charac-
several less important phospholipids, is responsible
teristics, similar to those of the lungs; even if the lungs
for reducing the surface tension. It does this by not
were not present in the thorax, muscular effort would
dissolving uniformly in the fluid lining the alveolar
still be required to expand the thoracic cage.
surface. Instead, part of the molecule dissolves,
while the remainder spreads over the surface of the
Compliance of the Thorax and the
water in the alveoli. This surface has from one
Lungs Together
twelfth to one half the surface tension of a pure water
The compliance of the entire pulmonary system (the
surface.
lungs and thoracic cage together) is measured while
In quantitative terms, the surface tension of differ-
expanding the lungs of a totally relaxed or paralyzed
ent water fluids is approximately the following: pure
person. To do this, air is forced into the lungs a little at
water, 72 dynes/cm; normal fluids lining the alveoli but
a time while recording lung pressures and volumes. To
without surfactant, 50 dynes/cm; normal fluids lining
inflate this total pulmonary system, almost twice as
the alveoli and with normal amounts of surfactant
much pressure is needed as to inflate the same lungs
included, between 5 and 30 dynes/cm.
after removal from the chest cage. Therefore, the com-
pliance of the combined lung-thorax system is almost
Pressure in Occluded Alveoli Caused by Surface Tension. If the
exactly one half that of the lungs alone—110 milliliters
air passages leading from the alveoli of the lungs are
blocked, the surface tension in the alveoli tends to col-
of volume per centimeter of water pressure for
lapse the alveoli. This creates positive pressure in the
the combined system, compared with 200 ml/cm for
alveoli, attempting to push the air out. The amount of
the lungs alone. Furthermore, when the lungs are
pressure generated in this way in an alveolus can be cal-
expanded to high volumes or compressed to low
culated from the following formula:
volumes, the limitations of the chest become extreme;
Chapter 37
Pulmonary Ventilation
475
when near these limits, the compliance of the com-
bined lung-thorax system can be less than one fifth
that of the lungs alone.
“Work” of Breathing
Floating
drum
We have already pointed out that during normal quiet
breathing, all respiratory muscle contraction occurs
Oxygen
Recording
chamber
drum
during inspiration; expiration is almost entirely a
passive process caused by elastic recoil of the lungs and
chest cage. Thus, under resting conditions, the respira-
Water
Mouthpiece
tory muscles normally perform “work” to cause inspi-
Counterbalancing
ration but not to cause expiration.
weight
The work of inspiration can be divided into three
fractions: (1) that required to expand the lungs against
the lung and chest elastic forces, called compliance work
or elastic work; (2) that required to overcome the vis-
cosity of the lung and chest wall structures, called tissue
Figure 37-5
resistance work; and (3) that required to overcome
airway resistance to movement of air into the lungs,
Spirometer.
called airway resistance work.
Energy Required for Respiration. During normal quiet respi-
1.
The tidal volume is the volume of air inspired or
ration, only 3 to 5 per cent of the total energy expended
expired with each normal breath; it amounts to
by the body is required for pulmonary ventilation. But
during heavy exercise, the amount of energy required
about 500 milliliters in the adult male.
can increase as much as 50-fold, especially if the person
2.
The inspiratory reserve volume is the extra volume
has any degree of increased airway resistance or de-
of air that can be inspired over and above the
creased pulmonary compliance. Therefore, one of the
normal tidal volume when the person inspires
major limitations on the intensity of exercise that can
with full force; it is usually equal to about
be performed is the person’s ability to provide enough
3000 milliliters.
muscle energy for the respiratory process alone.
3.
The expiratory reserve volume is the maximum
extra volume of air that can be expired by
forceful expiration after the end of a normal
Pulmonary Volumes
tidal expiration; this normally amounts to about
and Capacities
1100 milliliters.
4.
The residual volume is the volume of air
Recording Changes in Pulmonary
remaining in the lungs after the most forceful
Volume—Spirometry
expiration; this volume averages about
1200 milliliters.
A simple method for studying pulmonary ventilation
is to record the volume movement of air into and out
Pulmonary Capacities
of the lungs, a process called spirometry. A typical
In describing events in the pulmonary cycle, it is
basic spirometer is shown in Figure 37-5. It consists of
sometimes desirable to consider two or more of the
a drum inverted over a chamber of water, with the
volumes together. Such combinations are called pul-
drum counterbalanced by a weight. In the drum is a
monary capacities. To the right in Figure 37-6 are listed
breathing gas, usually air or oxygen; a tube connects
the important pulmonary capacities, which can be
the mouth with the gas chamber. When one breathes
described as follows:
into and out of the chamber, the drum rises and falls,
1. The inspiratory capacity equals the tidal volume
and an appropriate recording is made on a moving
plus the inspiratory reserve volume. This is the
sheet of paper.
amount of air (about 3500 milliliters) a person can
Figure 37-6 shows a spirogram indicating changes in
breathe in, beginning at the normal expiratory
lung volume under different conditions of breathing.
level and distending the lungs to the maximum
For ease in describing the events of pulmonary venti-
amount.
lation, the air in the lungs has been subdivided in this
2. The functional residual capacity equals the
diagram into four volumes and four capacities, which
expiratory reserve volume plus the residual
are the average for a young adult man.
volume. This is the amount of air that remains in
the lungs at the end of normal expiration (about
Pulmonary Volumes
2300 milliliters).
To the left in Figure 37-6 are listed four pulmonary
3. The vital capacity equals the inspiratory reserve
lung volumes that, when added together, equal the
volume plus the tidal volume plus the expiratory
maximum volume to which the lungs can be expanded.
reserve volume. This is the maximum amount of
The significance of each of these volumes is the
air a person can expel from the lungs after first
following:
filling the lungs to their maximum extent and then
476
Unit VII Respiration
6000
Inspiration
5000
Inspiratory
Inspiratory
Vital
Total lung
reserve
capacity
capacity
capacity
4000
volume
Tidal
3000
volume
2000
Expiratory
Functional
reserve volume
residual
capacity
1000
Residual
Expiration
volume
Figure 37-6
Time
Diagram showing respiratory excursions during
normal breathing and during maximal inspiration
and maximal expiration.
expiring to the maximum extent (about
Determination of Functional
4600 milliliters).
Residual Capacity, Residual
4. The total lung capacity is the maximum volume to
Volume, and Total Lung Capacity—
which the lungs can be expanded with the greatest
Helium Dilution Method
possible effort (about 5800 milliliters); it is equal
to the vital capacity plus the residual volume.
The functional residual capacity (FRC), which is the
All pulmonary volumes and capacities are about 20
volume of air that remains in the lungs at the end of
each normal expiration, is important to lung function.
to 25 per cent less in women than in men, and they are
Because its value changes markedly in some types of
greater in large and athletic people than in small and
pulmonary disease, it is often desirable to measure this
asthenic people.
capacity. The spirometer cannot be used in a direct way
to measure the functional residual capacity, because the
air in the residual volume of the lungs cannot be expired
Abbreviations and Symbols Used
into the spirometer, and this volume constitutes about
in Pulmonary Function Studies
one half of the functional residual capacity. To measure
functional residual capacity, the spirometer must be
Spirometry is only one of many measurement proce-
used in an indirect manner, usually by means of a
dures that the pulmonary physician uses daily. Many of
helium dilution method, as follows.
these measurement procedures depend heavily on
A spirometer of known volume is filled with air mixed
mathematical computations. To simplify these calcula-
with helium at a known concentration. Before breath-
tions as well as the presentation of pulmonary function
ing from the spirometer, the person expires normally. At
data, a number of abbreviations and symbols have
the end of this expiration, the remaining volume in the
become standardized. The more important of these are
lungs is equal to the functional residual capacity. At this
given in Table 37-1. Using these symbols, we present
point, the subject immediately begins to breathe from
here a few simple algebraic exercises showing some of
the spirometer, and the gases of the spirometer mix with
the interrelations among the pulmonary volumes and
the gases of the lungs. As a result, the helium becomes
capacities; the student should think through and verify
diluted by the functional residual capacity gases, and the
these interrelations.
volume of the functional residual capacity can be cal-
VC = IRV + VT + ERV
culated from the degree of dilution of the helium, using
VC = IC + ERV
the following formula:
TLC = VC + RV
ÊCi
He
ˆ
TLC = IC + FRC
FRC
=
-1
Vi
Spir
Ë
¯
FRC = ERV + RV
CfHe
Chapter 37
Pulmonary Ventilation
477
Table 37-1
Abbreviations and Symbols for Pulmonary Function
VT
tidal volume
PB
atmospheric pressure
FRC
functional residual capacity
Palv
alveolar pressure
ERV
expiratory reserve volume
Ppl
pleural pressure
RV
residual volume
PO2
partial pressure of oxygen
IC
inspiratory capacity
PCO2
partial pressure of carbon dioxide
IRV
inspiratory reserve volume
PN2
partial pressure of nitrogen
TLC
total lung capacity
PaO2
partial pressure of oxygen in arterial blood
VC
vital capacity
PaCO2
partial pressure of carbon dioxide in arterial blood
Raw
resistance of the airways to flow of air into
PAO2
partial pressure of oxygen in alveolar gas
the lung
C
compliance
PACO2
partial pressure of carbon dioxide in alveolar gas
VD
volume of dead space gas
PAH2O
partial pressure of water in alveolar gas
VA
volume of alveolar gas
R
respiratory exchange ratio
˙
VI.
inspired volume of ventilation per minute
Q
cardiac output
VE
expired volume of ventilation per minute
Vs
shunt flow
VA.
alveolar ventilation per minute
CaO2
concentration of oxygen in arterial blood
V
o2
rate of oxygen uptake per minute
C
¯o2
concentration of oxygen in mixed venous blood
V
co2
amount of carbon dioxide eliminated per
SO2
percentage saturation of hemoglobin with oxygen
minute
Vco
rate of carbon monoxide uptake per minute
SaO2
percentage saturation of hemoglobin with oxygen
in arterial blood
Dlo2
diffusing capacity of the lungs for oxygen
DlCO
diffusing capacity of the lungs for carbon
monoxide
where FRC is functional residual capacity, CiHe is initial
greater than 200 L/min, or more than 30 times normal.
concentration of helium in the spirometer, CfHe is final
Most people cannot sustain more than one half to two
concentration of helium in the spirometer, and ViSpir is
thirds these values for longer than 1 minute.
initial volume of the spirometer.
Once the FRC has been determined, the residual
volume (RV) can be determined by subtracting expira-
Alveolar Ventilation
tory reserve volume (ERV), as measured by normal
spirometry, from the FRC. Also, the total lung capacity
(TLC) can be determined by adding the inspiratory
The ultimate importance of pulmonary ventilation is
capacity (IC) to the FRC. That is,
to continually renew the air in the gas exchange areas
of the lungs, where air is in proximity to the pulmonary
RV = FRC - ERV
blood. These areas include the alveoli, alveolar sacs,
and
alveolar ducts, and respiratory bronchioles. The rate at
which new air reaches these areas is called alveolar
TLC = FRC + IC
ventilation.
Minute Respiratory Volume
“Dead Space” and Its Effect
Equals Respiratory Rate
on Alveolar Ventilation
Times Tidal Volume
Some of the air a person breathes never reaches the
The minute respiratory volume is the total amount of
gas exchange areas but simply fills respiratory pas-
new air moved into the respiratory passages each
sages where gas exchange does not occur, such as the
minute; this is equal to the tidal volume times the res-
nose, pharynx, and trachea. This air is called dead space
piratory rate per minute. The normal tidal volume is
air because it is not useful for gas exchange.
about 500 milliliters, and the normal respiratory rate
On expiration, the air in the dead space is expired
is about 12 breaths per minute. Therefore, the minute
first, before any of the air from the alveoli reaches the
respiratory volume averages about 6 L/min. A person
atmosphere. Therefore, the dead space is very disad-
can live for a short period with a minute respiratory
vantageous for removing the expiratory gases from the
volume as low as 1.5 L/min and a respiratory rate of
lungs.
only 2 to 4 breaths per minute.
The respiratory rate occasionally rises to 40 to 50
Measurement of the Dead Space Volume. A simple method
per minute, and the tidal volume can become as great
for measuring dead space volume is demonstrated by
as the vital capacity, about 4600 milliliters in a young
the graph in Figure 37-7. In making this measurement,
adult man. This can give a minute respiratory volume
the subject suddenly takes a deep breath of oxygen. This
478
Unit VII
Respiration
Anatomic Versus Physiologic Dead Space. The method just
described for measuring the dead space measures the
volume of all the space of the respiratory system other
80
than the alveoli and their other closely related gas
exchange areas; this space is called the anatomic dead
concentration
space. On occasion, some of the alveoli themselves are
60
nonfunctional or only partially functional because of
absent or poor blood flow through the adjacent pul-
monary capillaries. Therefore, from a functional point of
40
view, these alveoli must also be considered dead space.
When the alveolar dead space is included in the total
measurement of dead space, this is called the physio-
20
logic dead space, in contradistinction to the anatomic
dead space. In a normal person, the anatomic and phys-
iologic dead spaces are nearly equal because all alveoli
0
are functional in the normal lung, but in a person with
0
100
200
300
400
500
partially functional or nonfunctional alveoli in some
Air expired (ml)
parts of the lungs, the physiologic dead space may be as
much as 10 times the volume of the anatomic dead
space, or 1 to 2 liters. These problems are discussed
further in Chapter 39 in relation to pulmonary gaseous
exchange and in Chapter 42 in relation to certain pul-
Figure 37-7
monary diseases.
Record of the changes in nitrogen concentration in the expired air
after a single previous inspiration of pure oxygen. This record can
be used to calculate dead space, as discussed in the text.
Rate of Alveolar Ventilation
Alveolar ventilation per minute is the total volume of
new air entering the alveoli and adjacent gas exchange
fills the entire dead space with pure oxygen. Some
areas each minute. It is equal to the respiratory rate
oxygen also mixes with the alveolar air but does not
times the amount of new air that enters these areas
completely replace this air. Then the person expires
with each breath.
through a rapidly recording nitrogen meter, which
makes the record shown in the figure. The first portion
V
A = Freq • (VT - VD)
of the expired air comes from the dead space regions of
whereVA is the volume of alveolar ventilation per
the respiratory passageways, where the air has been
minute, Freq is the frequency of respiration per
completely replaced by oxygen. Therefore, in the early
minute, VT is the tidal volume, and VD is the physio-
part of the record, only oxygen appears, and the nitro-
logic dead space volume.
gen concentration is zero. Then, when alveolar air
begins to reach the nitrogen meter, the nitrogen con-
Thus, with a normal tidal volume of 500 milliliters, a
centration rises rapidly, because alveolar air containing
normal dead space of 150 milliliters, and a respiratory
large amounts of nitrogen begins to mix with the dead
rate of 12 breaths per minute, alveolar ventilation
space air. After still more air has been expired, all the
equals 12
¥ (500 - 150), or 4200 ml/min.
dead space air has been washed from the passages, and
Alveolar ventilation is one of the major factors
only alveolar air remains. Therefore, the recorded nitro-
determining the concentrations of oxygen and carbon
gen concentration reaches a plateau level equal to its
dioxide in the alveoli. Therefore, almost all discussions
concentration in the alveoli, as shown to the right in the
of gaseous exchange in the following chapters on the
figure. With a little thought, the student can see that the
respiratory system emphasize alveolar ventilation.
gray area represents the air that has no nitrogen in it;
this area is a measure of the volume of dead space air.
For exact quantification, the following equation is used:
Functions of the Respiratory
Gray area ¥ V
E
Passageways
V
D
=
Pink area+Gray area
Trachea, Bronchi, and Bronchioles
where VD is dead space air and VE is the total volume
of expired air.
Figure 37-8 shows the respiratory system, demonstrat-
Let us assume, for instance, that the gray area on the
ing especially the respiratory passageways. The air is dis-
graph is 30 square centimeters, the pink area is 70
tributed to the lungs by way of the trachea, bronchi, and
square centimeters, and the total volume expired is 500
bronchioles.
milliliters. The dead space would be
One of the most important problems in all the respi-
ratory passageways is to keep them open and allow easy
30
¥
500, or
150
ml
passage of air to and from the alveoli. To keep the
30+
70
trachea from collapsing, multiple cartilage rings extend
about five sixths of the way around the trachea. In the
Normal Dead Space Volume. The normal dead space air in
walls of the bronchi, less extensive curved cartilage
a young adult man is about 150 milliliters. This increases
plates also maintain a reasonable amount of rigidity yet
slightly with age.
allow sufficient motion for the lungs to expand and
Chapter 37
Pulmonary Ventilation
479
Conchae
CO2
O2
Alveolus
Epiglottis
O2
O2
Glottis
Pharynx
CO2
CO2
Larynx, vocal
Esophagus
cords
Pulmonary
Trachea
capillary
Pulmonary arteries
Pulmonary veins
Alveoli
Figure 37-8
Respiratory passages.
contract. These plates become progressively less exten-
are easily occluded by (1) muscle contraction in their
sive in the later generations of bronchi and are gone in
walls, (2) edema occurring in the walls, or (3) mucus col-
the bronchioles, which usually have diameters less than
lecting in the lumens of the bronchioles.
1.5 millimeters. The bronchioles are not prevented from
collapsing by the rigidity of their walls. Instead, they are
Nervous and Local Control of the Bronchiolar Musculature—
kept expanded mainly by the same transpulmonary
“Sympathetic” Dilation of the Bronchioles. Direct control of
pressures that expand the alveoli. That is, as the alveoli
the bronchioles by sympathetic nerve fibers is relatively
enlarge, the bronchioles also enlarge, but not as much.
weak because few of these fibers penetrate to the
central portions of the lung. However, the bronchial tree
Muscular Wall of the Bronchi and Bronchioles and Its Control. In
is very much exposed to norepinephrine and epineph-
all areas of the trachea and bronchi not occupied by car-
rine released into the blood by sympathetic stimulation
tilage plates, the walls are composed mainly of smooth
of the adrenal gland medullae. Both these hormones—
muscle. Also, the walls of the bronchioles are almost
especially epinephrine, because of its greater stimula-
entirely smooth muscle, with the exception of the most
tion of beta-adrenergic receptors—cause dilation of the
terminal bronchiole, called the respiratory bronchiole,
bronchial tree.
which is mainly pulmonary epithelium and underlying
fibrous tissue plus a few smooth muscle fibers. Many
Parasympathetic Constriction of the Bronchioles. A few
obstructive diseases of the lung result from narrowing
parasympathetic nerve fibers derived from the vagus
of the smaller bronchi and larger bronchioles, often
nerves penetrate the lung parenchyma. These nerves
because of excessive contraction of the smooth muscle
secrete acetylcholine and, when activated, cause mild
itself.
to moderate constriction of the bronchioles. When a
disease process such as asthma has already caused some
Resistance to Airflow in the Bronchial Tree. Under normal
bronchiolar constriction, superimposed parasympa-
respiratory conditions, air flows through the respiratory
thetic nervous stimulation often worsens the condition.
passageways so easily that less than 1 centimeter of
When this occurs, administration of drugs that block the
water pressure gradient from the alveoli to the atmos-
effects of acetylcholine, such as atropine, can sometimes
phere is sufficient to cause enough airflow for quiet
relax the respiratory passages enough to relieve the
breathing. The greatest amount of resistance to airflow
obstruction.
occurs not in the minute air passages of the terminal
Sometimes the parasympathetic nerves are also acti-
bronchioles but in some of the larger bronchioles and
vated by reflexes that originate in the lungs. Most of
bronchi near the trachea. The reason for this high resist-
these begin with irritation of the epithelial membrane
ance is that there are relatively few of these larger
of the respiratory passageways themselves, initiated by
bronchi in comparison with the approximately 65,000
noxious gases, dust, cigarette smoke, or bronchial infec-
parallel terminal bronchioles, through each of which
tion. Also, a bronchiolar constrictor reflex often occurs
only a minute amount of air must pass.
when microemboli occlude small pulmonary arteries.
Yet in disease conditions, the smaller bronchioles
often play a far greater role in determining airflow
Local Secretory Factors Often Cause Bronchiolar Constriction.
resistance because of their small size and because they
Several substances formed in the lungs themselves are
480
Unit VII
Respiration
often quite active in causing bronchiolar constriction.
invaginate inward, so that the exploding air actually
Two of the most important of these are histamine and
passes through bronchial and tracheal slits. The rapidly
slow reactive substance of anaphylaxis. Both of these are
moving air usually carries with it any foreign matter that
released in the lung tissues by mast cells during allergic
is present in the bronchi or trachea.
reactions, especially those caused by pollen in the air.
Therefore, they play key roles in causing the airway
Sneeze Reflex
obstruction that occurs in allergic asthma; this is espe-
The sneeze reflex is very much like the cough reflex,
cially true of the slow reactive substance of anaphylaxis.
except that it applies to the nasal passageways instead
The same irritants that cause parasympathetic con-
of the lower respiratory passages. The initiating stimu-
strictor reflexes of the airways—smoke, dust, sulfur
lus of the sneeze reflex is irritation in the nasal pas-
dioxide, and some of the acidic elements in smog—often
sageways; the afferent impulses pass in the fifth cranial
act directly on the lung tissues to initiate local, non-
nerve to the medulla, where the reflex is triggered. A
nervous reactions that cause obstructive constriction of
series of reactions similar to those for the cough reflex
the airways.
takes place; however, the uvula is depressed, so that
large amounts of air pass rapidly through the nose, thus
Mucus Lining the Respiratory Passageways, and Action of Cilia
helping to clear the nasal passages of foreign matter.
to Clear the Passageways
All the respiratory passages, from the nose to the ter-
minal bronchioles, are kept moist by a layer of mucus
Normal Respiratory Functions
that coats the entire surface. The mucus is secreted
of the Nose
partly by individual mucous goblet cells in the epithe-
lial lining of the passages and partly by small submu-
As air passes through the nose, three distinct normal
cosal glands. In addition to keeping the surfaces moist,
respiratory functions are performed by the nasal cavi-
the mucus traps small particles out of the inspired air
ties: (1) the air is warmed by the extensive surfaces of
and keeps most of these from ever reaching the alveoli.
the conchae and septum, a total area of about 160
The mucus itself is removed from the passages in the
square centimeters (see Figure 37-8); (2) the air is
following manner.
almost completely humidified even before it passes
The entire surface of the respiratory passages, both in
beyond the nose; and (3) the air is partially filtered.
the nose and in the lower passages down as far as the
These functions together are called the air conditioning
terminal bronchioles, is lined with ciliated epithelium,
function of the upper respiratory passageways. Ordi-
with about 200 cilia on each epithelial cell. These cilia
narily, the temperature of the inspired air rises to within
beat continually at a rate of 10 to 20 times per second
1°F of body temperature and to within 2 to 3 per cent
by the mechanism explained in Chapter 2, and the direc-
of full saturation with water vapor before it reaches the
tion of their
“power stroke” is always toward the
trachea. When a person breathes air through a tube
pharynx. That is, the cilia in the lungs beat upward,
directly into the trachea (as through a tracheostomy),
whereas those in the nose beat downward. This contin-
the cooling and especially the drying effect in the lower
ual beating causes the coat of mucus to flow slowly, at a
lung can lead to serious lung crusting and infection.
velocity of a few millimeters per minute, toward the
pharynx. Then the mucus and its entrapped particles are
Filtration Function of the Nose. The hairs at the entrance to
either swallowed or coughed to the exterior.
the nostrils are important for filtering out large par-
ticles. Much more important, though, is the removal
Cough Reflex
of particles by turbulent precipitation. That is, the air
The bronchi and trachea are so sensitive to light touch
passing through the nasal passageways hits many
that very slight amounts of foreign matter or other
obstructing vanes: the conchae (also called turbinates,
causes of irritation initiate the cough reflex. The larynx
because they cause turbulence of the air), the septum,
and carina (the point where the trachea divides into the
and the pharyngeal wall. Each time air hits one of these
bronchi) are especially sensitive, and the terminal bron-
obstructions, it must change its direction of movement.
chioles and even the alveoli are sensitive to corrosive
The particles suspended in the air, having far more mass
chemical stimuli such as sulfur dioxide gas or chlorine
and momentum than air, cannot change their direction
gas. Afferent nerve impulses pass from the respiratory
of travel as rapidly as the air can. Therefore, they con-
passages mainly through the vagus nerves to the
tinue forward, striking the surfaces of the obstructions,
medulla of the brain. There, an automatic sequence of
and are entrapped in the mucous coating and trans-
events is triggered by the neuronal circuits of the
ported by the cilia to the pharynx to be swallowed.
medulla, causing the following effect.
First, up to
2.5 liters of air are rapidly inspired.
Size of Particles Entrapped in the Respiratory Passages.
Second, the epiglottis closes, and the vocal cords shut
The nasal turbulence mechanism for removing particles
tightly to entrap the air within the lungs. Third, the
from air is so effective that almost no particles larger
abdominal muscles contract forcefully, pushing against
than 6 micrometers in diameter enter the lungs through
the diaphragm while other expiratory muscles, such as
the nose. This size is smaller than the size of red blood
the internal intercostals, also contract forcefully. Conse-
cells.
quently, the pressure in the lungs rises rapidly to as
Of the remaining particles, many that are between 1
much as 100 mm Hg or more. Fourth, the vocal cords
and 5 micrometers settle in the smaller bronchioles as a
and the epiglottis suddenly open widely, so that air
result of gravitational precipitation. For instance, termi-
under this high pressure in the lungs explodes outward.
nal bronchiolar disease is common in coal miners
Indeed, sometimes this air is expelled at velocities
because of settled dust particles. Some of the still
ranging from 75 to 100 miles per hour. Importantly, the
smaller particles (smaller than 1 micrometer in diame-
strong compression of the lungs collapses the bronchi
ter) diffuse against the walls of the alveoli and adhere
and trachea by causing their noncartilaginous parts to
to the alveolar fluid. But many particles smaller than 0.5
Chapter 37
Pulmonary Ventilation
481
Thyroid
cartilage
Thyroarytenoid
muscle
Vocal
ligament
Lateral
Full
Gentle
Intermediate position-
Figure 37-9
cricoarytenoid
abduction
abduction
loud whisper
muscle
A, Anatomy of the larynx.
B, Laryngeal function in phona-
tion, showing the positions of the
Arytenoid
vocal cords during different
cartilage
Posterior
types of phonation.
(Modified
cricoarytenoid
Stage
Phonation
Transverse
from Greene MC: The Voice and
muscle
whisper
arytenoid
Its Disorders,
4th ed. Philadel-
muscle
phia: JB Lippincott, 1980.)
A
B
micrometer in diameter remain suspended in the alve-
ligament is attached to the vocal processes of two ary-
olar air and are expelled by expiration. For instance, the
tenoid cartilages. The thyroid cartilage and the arytenoid
particles of cigarette smoke are about 0.3 micrometer.
cartilages articulate from below with another cartilage
Almost none of these particles are precipitated in the
not shown in Figure 37-9, the cricoid cartilage.
respiratory passageways before they reach the alveoli.
The vocal cords can be stretched by either forward
Unfortunately, up to one third of them do precipitate in
rotation of the thyroid cartilage or posterior rotation of
the alveoli by the diffusion process, with the balance
the arytenoid cartilages, activated by muscles stretching
remaining suspended and expelled in the expired air.
from the thyroid cartilage and arytenoid cartilages to
Many of the particles that become entrapped in
the cricoid cartilage. Muscles located within the vocal
the alveoli are removed by alveolar macrophages, as
cords lateral to the vocal ligaments, the thyroarytenoid
explained in Chapter 33, and others are carried away by
muscles, can pull the arytenoid cartilages toward the
the lung lymphatics. An excess of particles can cause
thyroid cartilage and, therefore, loosen the vocal cords.
growth of fibrous tissue in the alveolar septa, leading to
Also, slips of these muscles within the vocal cords can
permanent debility.
change the shapes and masses of the vocal cord edges,
sharpening them to emit high-pitched sounds and blunt-
ing them for the more bass sounds.
Vocalization
Finally, several other sets of small laryngeal muscles
lie between the arytenoid cartilages and the cricoid car-
Speech involves not only the respiratory system but
tilage and can rotate these cartilages inward or outward
also (1) specific speech nervous control centers in the
or pull their bases together or apart to give the various
cerebral cortex, which are discussed in Chapter 57; (2)
configurations of the vocal cords shown in Figure 37-9B.
respiratory control centers of the brain; and (3) the
articulation and resonance structures of the mouth and
Articulation and Resonance. The three major organs of
nasal cavities. Speech is composed of two mechanical
articulation are the lips, tongue, and soft palate. They
functions:
(1) phonation, which is achieved by the
need not be discussed in detail because we are all famil-
larynx, and (2) articulation, which is achieved by the
iar with their movements during speech and other
structures of the mouth.
vocalizations.
The resonators include the mouth, the nose and asso-
ciated nasal sinuses, the pharynx, and even the chest
Phonation. The larynx, shown in Figure 37-9A, is espe-
cavity. Again, we are all familiar with the resonating
cially adapted to act as a vibrator. The vibrating element
qualities of these structures. For instance, the function
is the vocal folds, commonly called the vocal cords. The
of the nasal resonators is demonstrated by the change
vocal cords protrude from the lateral walls of the larynx
in voice quality when a person has a severe cold that
toward the center of the glottis; they are stretched and
blocks the air passages to these resonators.
positioned by several specific muscles of the larynx
itself.
Figure 37-9B shows the vocal cords as they are seen
when looking into the glottis with a laryngoscope.
References
During normal breathing, the cords are wide open to
allow easy passage of air. During phonation, the cords
Carr MJ, Undem BJ: Bronchopulmonary afferent nerves.
move together so that passage of air between them will
Respirology 8:291, 2003.
cause vibration. The pitch of the vibration is determined
Coulson FR, Fryer AD: Muscarinic acetylcholine receptors
mainly by the degree of stretch of the cords, but also by
and airway diseases. Pharmacol Ther 98:59, 2003.
how tightly the cords are approximated to one another
Daniels CB, Orgeig S: Pulmonary surfactant: the key to the
and by the mass of their edges.
evolution of air breathing. News Physiol Sci 18:151, 2003.
Figure 37-9A shows a dissected view of the vocal
Foster WM: Mucociliary transport and cough in humans.
folds after removal of the mucous epithelial lining.
Pulm Pharmacol Ther 15:277, 2002.
Immediately inside each cord is a strong elastic ligament
Haitsma JJ, Papadakos PJ, Lachmann B: Surfactant therapy
called the vocal ligament. This is attached anteriorly to
for acute lung injury/acute respiratory distress syndrome.
the large thyroid cartilage, which is the cartilage that
Curr Opin Crit Care 10:18, 2004.
projects forward from the anterior surface of the neck
Hilaire G, Duron B: Maturation of the mammalian respira-
and is called the “Adam’s apple.” Posteriorly, the vocal
tory system. Physiol Rev 79:325, 1999.
482
Unit VII Respiration
Lai-Fook SJ: Pleural mechanics and fluid exchange. Physiol
Powell FL, Hopkins SR: Comparative physiology of lung
Rev 84:385, 2004.
complexity: implications for gas exchange. News Physiol
Mason RJ, Greene K, Voelker DR: Surfactant protein A and
Sci 19:55, 2004.
surfactant protein D in health and disease. Am J Physiol
Sant’Ambrogio G, Widdicombe J: Reflexes from airway
Lung Cell Mol Physiol 275:L1, 1998.
rapidly adapting receptors. Respir Physiol 125:33, 2001.
Massaro D, Massaro GD: Pulmonary alveoli: formation, the
Uhlig S, Taylor AE: Methods in Pulmonary Research. Basel:
“call for oxygen,” and other regulators. Am J Physiol Lung
Birkhauser Verlag, 1998.
Cell Mol Physiol 282:L345, 2002.
West JB: Respiratory Physiology. New York: Oxford Uni-
Maxfield RA: New and emerging minimally invasive
versity Press, 1996.
techniques for lung volume reduction. Chest
125:777,
West JB: Why doesn’t the elephant have a pleural space?
2004.
News Physiol Sci 17:47, 2002.
McConnell AK, Romer LM: Dyspnoea in health and
Widdicombe J: Neuroregulation of cough: implications for
obstructive pulmonary disease: the role of respiratory
drug therapy. Curr Opin Pharmacol 2:256, 2002.
muscle function and training. Sports Med 34:117, 2004.
Wright JR: Pulmonary surfactant: a front line of lung host
Paton JF, Dutschmann M: Central control of upper airway
defense. J Clin Invest 111:1453, 2003.
resistance regulating respiratory airflow in mammals.
Zeitels SM, Healy GB: Laryngology and phonosurgery.
J Anat 201:319, 2002.
N Engl J Med 349:882, 2003.
C H A P T E R
3
8
Pulmonary Circulation,
Pulmonary Edema, Pleural Fluid
Some aspects of blood flow distribution and other
hemodynamics are particular to the pulmonary cir-
culation and are especially important for gas
exchange in the lungs. The present discussion is con-
cerned with these special features of the pulmonary
circulation.
Physiologic Anatomy of the Pulmonary
Circulatory System
Pulmonary Vessels. The pulmonary artery extends only 5 centimeters beyond the apex
of the right ventricle and then divides into right and left main branches that supply
blood to the two respective lungs.
The pulmonary artery is thin, with a wall thickness one third that of the aorta.
The pulmonary arterial branches are very short, and all the pulmonary arteries, even
the smaller arteries and arterioles, have larger diameters than their counterpart sys-
temic arteries. This, combined with the fact that the vessels are thin and distensible,
gives the pulmonary arterial tree a large compliance, averaging almost 7 ml/mm Hg,
which is similar to that of the entire systemic arterial tree. This large compliance
allows the pulmonary arteries to accommodate the stroke volume output of the
right ventricle.
The pulmonary veins, like the pulmonary arteries, are also short. They immedi-
ately empty their effluent blood into the left atrium, to be pumped by the left heart
through the systemic circulation.
Bronchial Vessels. Blood also flows to the lungs through small bronchial arteries that
originate from the systemic circulation, amounting to about 1 to 2 per cent of the
total cardiac output. This bronchial arterial blood is oxygenated blood, in contrast
to the partially deoxygenated blood in the pulmonary arteries. It supplies the sup-
porting tissues of the lungs, including the connective tissue, septa, and large and
small bronchi. After this bronchial and arterial blood has passed through the sup-
porting tissues, it empties into the pulmonary veins and enters the left atrium, rather
than passing back to the right atrium. Therefore, the flow into the left atrium and
the left ventricular output are about 1 to 2 per cent greater than the right ventric-
ular output.
Lymphatics. Lymph vessels are present in all the supportive tissues of the lung, begin-
ning in the connective tissue spaces that surround the terminal bronchioles, cours-
ing to the hilum of the lung, and thence mainly into the right thoracic lymph duct.
Particulate matter entering the alveoli is partly removed by way of these channels,
and plasma protein leaking from the lung capillaries is also removed from the lung
tissues, thereby helping to prevent pulmonary edema.
Pressures in the Pulmonary System
Pressure Pulse Curve in the Right Ventricle. The pressure pulse curves of the right
ventricle and pulmonary artery are shown in the lower portion of Figure 38-1.
These curves are contrasted with the much higher aortic pressure curve shown
in the upper portion of the figure. The systolic pressure in the right ventricle of
the normal human being averages about 25 mm Hg, and the diastolic pressure
483
484
Unit VII
Respiration
normal human being, the diastolic pulmonary arterial
pressure is about 8 mm Hg, and the mean pulmonary
arterial pressure is 15 mm Hg.
Aortic pressure curve
120
Pulmonary Capillary Pressure. The mean pulmonary cap-
illary pressure, as diagrammed in Figure 38-2, is about
7 mm Hg. The importance of this low capillary pres-
sure is discussed in detail later in the chapter in rela-
75
tion to fluid exchange functions of the pulmonary
capillaries.
Right ventricular curve
Left Atrial and Pulmonary Venous Pressures. The mean
Pulmonary artery curve
pressure in the left atrium and the major pulmonary
25
veins averages about
2 mm Hg in the recumbent
human being, varying from as low as 1 mm Hg to as
8
0
high as 5 mm Hg. It usually is not feasible to measure
0
1
2
a human being’s left atrial pressure using a direct
Seconds
measuring device because it is difficult to pass a
catheter through the heart chambers into the left
atrium. However, the left atrial pressure can often be
estimated with moderate accuracy by measuring the
so-called pulmonary wedge pressure. This is achieved
Figure 38-1
by inserting a catheter first through a peripheral vein
to the right atrium, then through the right side of the
Pressure pulse contours in the right ventricle, pulmonary artery,
and aorta.
heart and through the pulmonary artery into one of
the small branches of the pulmonary artery, finally
pushing the catheter until it wedges tightly in the small
branch.
The pressure measured through the catheter, called
the “wedge pressure,” is about 5 mm Hg. Because all
blood flow has been stopped in the small wedged
25
S
artery, and because the blood vessels extending
beyond this artery make a direct connection with the
Pulmonary
pulmonary capillaries, this wedge pressure is usually
15
M
capillaries
Left
atrium
only 2 to 3 mm Hg greater than the left atrial pressure.
8
D
When the left atrial pressure rises to high values,
7
the pulmonary wedge pressure also rises. Therefore,
2
wedge pressure measurements can be used to clinically
0
study changes in pulmonary capillary pressure and
Pulmonary
Pulmonary
Left
artery
capillaries
atrium
left atrial pressure in patients with congestive heart
failure.
Figure 38-2
Blood Volume of the Lungs
Pressures in the different vessels of the lungs. D, diastolic; M,
The blood volume of the lungs is about 450 milliliters,
mean; S, systolic; red curve, arterial pulsations.
about 9 per cent of the total blood volume of the entire
circulatory system. Approximately 70 milliliters of this
pulmonary blood volume is in the pulmonary capil-
averages about 0 to 1 mm Hg, values that are only one
laries, and the remainder is divided about equally
fifth those for the left ventricle.
between the pulmonary arteries and the veins.
Pressures in the Pulmonary Artery. During systole, the
Lungs as a Blood Reservoir. Under various physiological
pressure in the pulmonary artery is essentially equal
and pathological conditions, the quantity of blood in
to the pressure in the right ventricle, as also shown
the lungs can vary from as little as one half normal up
in Figure 38-1. However, after the pulmonary valve
to twice normal. For instance, when a person blows out
closes at the end of systole, the ventricular pressure
air so hard that high pressure is built up in the lungs—
falls precipitously, whereas the pulmonary arterial
such as when blowing a trumpet—as much as 250 mil-
pressure falls more slowly as blood flows through the
liliters of blood can be expelled from the pulmonary
capillaries of the lungs.
circulatory system into the systemic circulation.
As shown in Figure 38-2, the systolic pulmonary
Also, loss of blood from the systemic circulation by
arterial pressure averages about
25 mm Hg in the
hemorrhage can be partly compensated for by the
Chapter 38
Pulmonary Circulation, Pulmonary Edema, Pleural Fluid
485
automatic shift of blood from the lungs into the sys-
Effect of Hydrostatic
temic vessels.
Pressure Gradients in the
Lungs on Regional
Shift of Blood Between the Pulmonary and Systemic
Pulmonary Blood Flow
Circulatory Systems as a Result of Cardiac Pathology.
Failure of the left side of the heart or increased resist-
In Chapter 15, it was pointed out that the blood pres-
ance to blood flow through the mitral valve as a result
sure in the foot of a standing person can be as much as
90 mm Hg greater than the pressure at the level of the
of mitral stenosis or mitral regurgitation causes blood
heart. This is caused by hydrostatic pressure—that is, by
to dam up in the pulmonary circulation, sometimes
the weight of the blood itself in the blood vessels. The
increasing the pulmonary blood volume as much as
same effect, but to a lesser degree, occurs in the lungs.
100 per cent and causing large increases in the pul-
In the normal, upright adult, the lowest point in the
monary vascular pressures. Because the volume of the
lungs is about 30 centimeters below the highest point.
systemic circulation is about nine times that of the pul-
This represents a 23 mm Hg pressure difference, about
monary system, a shift of blood from one system to the
15 mm Hg of which is above the heart and 8 below. That
other affects the pulmonary system greatly but usually
is, the pulmonary arterial pressure in the uppermost
has only mild systemic circulatory effects.
portion of the lung of a standing person is about 15 mm
Hg less than the pulmonary arterial pressure at the level
of the heart, and the pressure in the lowest portion of
the lungs is about 8 mm Hg greater. Such pressure dif-
Blood Flow Through the Lungs
ferences have profound effects on blood flow through
and Its Distribution
the different areas of the lungs. This is demonstrated by
the lower curve in Figure 38-3, which depicts blood flow
The blood flow through the lungs is essentially equal
per unit of lung tissue at different levels of the lung in
the upright person. Note that in the standing position at
to the cardiac output. Therefore, the factors that
rest, there is little flow in the top of the lung but about
control cardiac output—mainly peripheral factors,
five times as much flow in the bottom. To help explain
as discussed in Chapter 20—also control pulmonary
these differences, one often describes the lung as being
blood flow. Under most conditions, the pulmonary
divided into three zones, as shown in Figure 38-4.
vessels act as passive, distensible tubes that enlarge
In each zone, the patterns of blood flow are quite
with increasing pressure and narrow with decreasing
different.
pressure. For adequate aeration of the blood to
occur, it is important for the blood to be distributed to
Zones 1, 2, and 3 of Pulmonary
those segments of the lungs where the alveoli are
Blood Flow
best oxygenated. This is achieved by the following
mechanism.
The capillaries in the alveolar walls are distended by the
blood pressure inside them, but simultaneously, they are
Effect of Diminished Alveolar Oxygen on Local Alveolar Blood
Flow—Automatic Control of Pulmonary Blood Flow Distribution.
When the concentration of oxygen in the air of the
alveoli decreases below normal—especially when it
falls below 70 per cent of normal (below 73 mm Hg
Po2)—the adjacent blood vessels constrict, with the
vascular resistance increasing more than fivefold at
extremely low oxygen levels. This is opposite to the
effect observed in systemic vessels, which dilate rather
than constrict in response to low oxygen. It is believed
that the low oxygen concentration causes some yet
undiscovered vasoconstrictor substance to be released
from the lung tissue; this substance promotes con-
striction of the small arteries and arterioles. It has been
Exercise
suggested that this vasoconstrictor might be secreted
Standing at rest
by the alveolar epithelial cells when they become
hypoxic.
This effect of low oxygen on pulmonary vascular
Top
Middle
Bottom
resistance has an important function: to distribute
Lung level
blood flow where it is most effective. That is, if some
alveoli are poorly ventilated so that their oxygen con-
centration becomes low, the local vessels constrict. This
causes the blood to flow through other areas of the
Figure 38-3
lungs that are better aerated, thus providing an auto-
Blood flow at different levels in the lung of an upright person at
matic control system for distributing blood flow to the
rest and during exercise. Note that when the person is at rest, the
pulmonary areas in proportion to their alveolar
blood flow is very low at the top of the lungs; most of the flow is
oxygen pressures.
through the bottom of the lung.
486
Unit VII
Respiration
during cardiac systole. Conversely, during diastole, the
ZONE 1
8 mm Hg diastolic pressure at the level of the heart is
not sufficient to push the blood up the 15 mm Hg hydro-
Artery
PALV
Vein
static pressure gradient required to cause diastolic cap-
illary flow. Therefore, blood flow through the apical part
of the lung is intermittent, with flow during systole but
cessation of flow during diastole; this is called zone 2
Ppc
blood flow. Zone 2 blood flow begins in the normal
lungs about 10 centimeters above the midlevel of the
ZONE 2
heart and extends from there to the top of the lungs.
In the lower regions of the lungs, from about 10 cen-
timeters above the level of the heart all the way to the
Artery
PALV
Vein
bottom of the lungs, the pulmonary arterial pressure
during both systole and diastole remains greater than
the zero alveolar air pressure. Therefore, there is con-
Ppc
tinuous flow through the alveolar capillaries, or zone 3
blood flow. Also, when a person is lying down, no part
of the lung is more than a few centimeters above the
ZONE 3
level of the heart. In this case, blood flow in a normal
person is entirely zone 3 blood flow, including the lung
Artery
PALV
Vein
apices.
Zone 1 Blood Flow Occurs Only Under Abnormal Conditions.
Ppc
Zone 1 blood flow, which is blood flow at no time during
the cardiac cycle, occurs when either the pulmonary sys-
tolic arterial pressure is too low or the alveolar pressure
is too high to allow flow. For instance, if an upright
Figure 38-4
person is breathing against a positive air pressure so
Mechanics of blood flow in the three blood flow zones of the lung:
that the intra-alveolar air pressure is at least 10 mm Hg
zone 1, no flow—alveolar air pressure (PALV) is greater than arte-
greater than normal but the pulmonary systolic blood
rial pressure; zone 2, intermittent flow—systolic arterial pressure
pressure is normal, one would expect zone 1 blood
rises higher than alveolar air pressure, but diastolic arterial pres-
flow—no blood flow—in the lung apices. Another
sure falls below alveolar air pressure; and zone 3, continuous
instance in which zone 1 blood flow occurs is in an
flow—arterial pressure and pulmonary capillary pressure (Ppc)
upright person whose pulmonary systolic arterial pres-
remain greater than alveolar air pressure at all times.
sure is exceedingly low, as might occur after severe
blood loss.
compressed by the alveolar air pressure on their out-
Effect of Exercise on Blood Flow Through the Different Parts of
sides. Therefore, any time the lung alveolar air pressure
the Lungs. Referring again to Figure 38-3, one sees that
becomes greater than the capillary blood pressure,
the blood flow in all parts of the lung increases during
the capillaries close and there is no blood flow. Under
exercise. The increase in flow in the top of the lung may
different normal and pathological lung conditions, one
be 700 to 800 per cent, whereas the increase in the lower
may find any one of three possible zones of pulmonary
part of the lung may be no more than 200 to 300 per
blood flow, as follows:
cent. The reason for these differences is that the pul-
Zone 1: No blood flow during all portions of the
monary vascular pressures rise enough during exercise
cardiac cycle because the local alveolar capillary
to convert the lung apices from a zone 2 pattern into a
pressure in that area of the lung never rises higher
zone 3 pattern of flow.
than the alveolar air pressure during any part of the
cardiac cycle
Effect of Increased Cardiac Output
Zone 2: Intermittent blood flow only during the pul-
monary arterial pressure peaks because the systolic
on Pulmonary Blood Flow and
pressure is then greater than the alveolar air pres-
Pulmonary Arterial Pressure During
sure, but the diastolic pressure is less than the alve-
Heavy Exercise
olar air pressure
Zone 3: Continuous blood flow because the alveolar
capillary pressure remains greater than alveolar air
During heavy exercise, blood flow through the lungs
pressure during the entire cardiac cycle
increases fourfold to sevenfold. This extra flow is
Normally, the lungs have only zones 2 and 3 blood
accommodated in the lungs in three ways: (1) by
flow—zone 2 (intermittent flow) in the apices, and zone
increasing the number of open capillaries, sometimes
3 (continuous flow) in all the lower areas. For example,
as much as threefold; (2) by distending all the capil-
when a person is in the upright position, the pulmonary
laries and increasing the rate of flow through each
arterial pressure at the lung apex is about 15 mm Hg
capillary more than twofold; and (3) by increasing the
less than the pressure at the level of the heart. There-
pulmonary arterial pressure. In the normal person, the
fore, the apical systolic pressure is only
10 mm Hg
first two changes decrease pulmonary vascular resist-
(25 mm Hg at heart level minus 15 mm Hg hydrostatic
ance so much that the pulmonary arterial pressure
pressure difference). This 10 mm Hg apical blood pres-
sure is greater than the zero alveolar air pressure, so that
rises very little, even during maximum exercise; this
blood flows through the pulmonary apical capillaries
effect is shown in Figure 38-5.
Chapter 38
Pulmonary Circulation, Pulmonary Edema, Pleural Fluid
487
above 30 mm Hg, causing similar increases in capillary
pressure, pulmonary edema is likely to develop, as we
discuss later in the chapter.
30
Pulmonary Capillary Dynamics
Normal value
Exchange of gases between the alveolar air and the
20
pulmonary capillary blood is discussed in the next
chapter. However, it is important for us to note here
that the alveolar walls are lined with so many capil-
laries that, in most places, the capillaries almost touch
10
one another side by side. Therefore, it is often said that
the capillary blood flows in the alveolar walls as a
“sheet of flow,” rather than in individual capillaries.
0
0
4
8
12
16
20
24
Pulmonary Capillary Pressure. No direct measurements
Cardiac output (L/min)
of pulmonary capillary pressure have ever been
made. However, “isogravimetric” measurement of pul-
monary capillary pressure, using a technique described
in Chapter 16, has given a value of 7 mm Hg. This is
Figure 38-5
probably nearly correct, because the mean left atrial
pressure is about 2 mm Hg and the mean pulmonary
Effect on mean pulmonary arterial pressure caused by increasing
the cardiac output during exercise.
arterial pressure is only
15 mm Hg, so the mean
pulmonary capillary pressure must lie somewhere
between these two values.
The ability of the lungs to accommodate greatly
Length of Time Blood Stays in the Pulmonary Capillaries.
increased blood flow during exercise without increas-
From histological study of the total cross-sectional
ing the pulmonary arterial pressure conserves the
area of all the pulmonary capillaries, it can be calcu-
energy of the right side of the heart. This ability also
lated that when the cardiac output is normal, blood
prevents a significant rise in pulmonary capillary pres-
passes through the pulmonary capillaries in about 0.8
sure, thus also preventing the development of pul-
second. When the cardiac output increases, this can
monary edema.
shorten to as little as 0.3 second. The shortening would
be much greater were it not for the fact that additional
capillaries, which normally are collapsed, open up to
Function of the Pulmonary Circulation
accommodate the increased blood flow. Thus, in only
When the Left Atrial Pressure Rises
a fraction of a second, blood passing through the
as a Result of Left-Sided Heart Failure
alveolar capillaries becomes oxygenated and loses its
excess carbon dioxide.
The left atrial pressure in a healthy person almost
never rises above +6 mm Hg, even during the most
Capillary Exchange of Fluid in the
strenuous exercise. These small changes in left atrial
Lungs, and Pulmonary Interstitial
pressure have virtually no effect on pulmonary circu-
latory function because this merely expands the pul-
Fluid Dynamics
monary venules and opens up more capillaries so that
blood continues to flow with almost equal ease from
The dynamics of fluid exchange across the lung capil-
the pulmonary arteries.
lary membranes are qualitatively the same as for
When the left side of the heart fails, however, blood
peripheral tissues. However, quantitatively, there are
begins to dam up in the left atrium. As a result, the
important differences, as follows:
left atrial pressure can rise on occasion from its normal
1. The pulmonary capillary pressure is low, about
value of
1 to
5 mm Hg all the way up to 40 to
7 mm Hg, in comparison with a considerably
50 mm Hg. The initial rise in atrial pressure, up to
higher functional capillary pressure in the
about 7 mm Hg, has very little effect on pulmonary cir-
peripheral tissues of about 17 mm Hg.
culatory function. But when the left atrial pressure
2. The interstitial fluid pressure in the lung is
rises to greater than 7 or 8 mm Hg, further increases
slightly more negative than that in the peripheral
in left atrial pressure above these levels cause almost
subcutaneous tissue. (This has been measured in
equally great increases in pulmonary arterial pressure,
two ways: by a micropipette inserted into the
thus causing a concomitant increased load on the right
pulmonary interstitium, giving a value of about
heart. Any increase in left atrial pressure above 7 or
-5 mm Hg, and by measuring the absorption
8 mm Hg increases the capillary pressure almost
pressure of fluid from the alveoli, giving a value of
equally as much. When the left atrial pressure has risen
about -8 mm Hg.)
488
Unit VII
Respiration
3. The pulmonary capillaries are relatively leaky to
pressure at the pulmonary capillary membrane; this
protein molecules, so that the colloid osmotic
can be calculated as follows:
pressure of the pulmonary interstitial fluid is
about 14 mm Hg, in comparison with less than
mm Hg
half this value in the peripheral tissues.
4. The alveolar walls are extremely thin, and the
Total outward force
+29
Total inward force
-28
alveolar epithelium covering the alveolar surfaces
MEAN FILTRATION PRESSURE
+1
is so weak that it can be ruptured by any positive
pressure in the interstitial spaces greater than
alveolar air pressure (greater than 0 mm Hg),
This filtration pressure causes a slight continual flow
which allows dumping of fluid from the interstitial
of fluid from the pulmonary capillaries into the inter-
spaces into the alveoli.
stitial spaces, and except for a small amount that evap-
Now let us see how these quantitative differences
orates in the alveoli, this fluid is pumped back to the
affect pulmonary fluid dynamics.
circulation through the pulmonary lymphatic system.
Interrelations Between Interstitial Fluid Pressure and Other
Negative Pulmonary Interstitial Pressure and the Mechanism
Pressures in the Lung. Figure 38-6 shows a pulmonary
for Keeping the Alveoli “Dry.” One of the most important
capillary, a pulmonary alveolus, and a lymphatic capil-
problems in lung function is to understand why the
lary draining the interstitial space between the blood
alveoli do not normally fill with fluid. One’s first incli-
capillary and the alveolus. Note the balance of forces
nation is to think that the alveolar epithelium is strong
at the blood capillary membrane, as follows:
enough and continuous enough to keep fluid from
leaking out of the interstitial spaces into the alveoli.
This is not true, because experiments have shown
mm Hg
that there are always openings between the alveolar
Forces tending to cause movement of fluid outward from the
epithelial cells through which even large protein mol-
capillaries and into the pulmonary interstitium:
ecules, as well as water and electrolytes, can pass.
Capillary pressure
7
However, if one remembers that the pulmonary cap-
Interstitial fluid colloid osmotic pressure
14
Negative interstitial fluid pressure
8
illaries and the pulmonary lymphatic system normally
TOTAL OUTWARD FORCE
29
maintain a slight negative pressure in the interstitial
spaces, it is clear that whenever extra fluid appears
Forces tending to cause absorption of fluid into the capillaries:
Plasma colloid osmotic pressure
28
in the alveoli, it will simply be sucked mechanically
TOTAL INWARD FORCE
28
into the lung interstitium through the small openings
between the alveolar epithelial cells. Then the excess
fluid is either carried away through the pulmonary
Thus, the normal outward forces are slightly greater
lymphatics or absorbed into the pulmonary capillaries.
than the inward forces, providing a mean filtration
Thus, under normal conditions, the alveoli are kept
“dry,” except for a small amount of fluid that seeps
from the epithelium onto the lining surfaces of the
Pressures Causing Fluid Movement
alveoli to keep them moist.
CAPILLARY
ALVEOLUS
Pulmonary Edema
Pulmonary edema occurs in the same way that edema
Hydrostatic
occurs elsewhere in the body. Any factor that causes the
-8
pressure
+7
-8
-8
(Surface
pulmonary interstitial fluid pressure to rise from the
tension
negative range into the positive range will cause rapid
at pore)
Osmotic
filling of the pulmonary interstitial spaces and alveoli
pressure
- 28
- 14
with large amounts of free fluid.
The most common causes of pulmonary edema are as
Net
(0)
(Evaporation)
(+1)
follows:
pressure
-5
1. Left-sided heart failure or mitral valve disease, with
consequent great increases in pulmonary venous
-4
pressure and pulmonary capillary pressure and
flooding of the interstitial spaces and alveoli.
2. Damage to the pulmonary blood capillary
Lymphatic pump
membranes caused by infections such as
pneumonia or by breathing noxious substances
Figure 38-6
such as chlorine gas or sulfur dioxide gas. Each of
these causes rapid leakage of both plasma proteins
Hydrostatic and osmotic forces at the capillary (left) and alveolar
and fluid out of the capillaries and into both the
membrane (right) of the lungs. Also shown is the tip end of a lym-
lung interstitial spaces and the alveoli.
phatic vessel (center) that pumps fluid from the pulmonary inter-
stitial spaces. (Modified from Guyton AC, Taylor AE, Granger HJ:
Circulatory Physiology II: Dynamics and Control of the Body
“Pulmonary Edema Safety Factor.” Experiments in animals
Fluids. Philadelphia: WB Saunders, 1975.)
have shown that the pulmonary capillary pressure nor-
Chapter 38
Pulmonary Circulation, Pulmonary Edema, Pleural Fluid
489
10
9
x
x
8
7
x
6
x
5
4
x
x
x
x
3
x
x
x
2
x
x
x
1
x
x
x
x
Figure 38-7
x
0
x
x
x
x
x x
Rate of fluid loss into the lung tissues when the left
0
5
10
15
20
25
30
35
40
45
50
atrial pressure (and pulmonary capillary pressure)
Left atrial pressure (mm Hg)
is increased. (From Guyton AC, Lindsey AW: Effect
of elevated left atrial pressure and decreased
plasma protein concentration on the development
of pulmonary edema. Circ Res 7:649, 1959.)
mally must rise to a value at least equal to the colloid
which the pulmonary capillary pressure occasionally
osmotic pressure of the plasma inside the capillaries
does rise to 50 mm Hg, death frequently ensues in less
before significant pulmonary edema will occur. To give
than 30 minutes from acute pulmonary edema.
an example, Figure 38-7 shows how different levels of
left atrial pressure increase the rate of pulmonary
edema formation in dogs. Remember that every time
Fluid in the Pleural Cavity
the left atrial pressure rises to high values, the pul-
monary capillary pressure rises to a level 1 to 2 mm Hg
When the lungs expand and contract during normal
greater than the left atrial pressure. In these experi-
breathing, they slide back and forth within the pleural
ments, as soon as the left atrial pressure rose above
cavity. To facilitate this, a thin layer of mucoid fluid lies
23 mm Hg (causing the pulmonary capillary pressure to
between the parietal and visceral pleurae.
rise above 25 mm Hg), fluid began to accumulate in the
Figure 38-8 shows the dynamics of fluid exchange in
lungs. This fluid accumulation increased even more
the pleural space. The pleural membrane is a porous,
rapidly with further increases in capillary pressure. The
mesenchymal, serous membrane through which small
plasma colloid osmotic pressure during these experi-
amounts of interstitial fluid transude continually into
ments was equal to this 25 mm Hg critical pressure
the pleural space. These fluids carry with them tissue
level. Therefore, in the human being, whose normal
proteins, giving the pleural fluid a mucoid characteris-
plasma colloid osmotic pressure is 28 mm Hg, one can
tic, which is what allows extremely easy slippage of the
predict that the pulmonary capillary pressure must
moving lungs.
rise from the normal level of 7 mm Hg to more than
The total amount of fluid in each pleural cavity is nor-
28 mm Hg to cause pulmonary edema, giving an acute
mally slight, only a few milliliters. Whenever the quan-
safety factor against pulmonary edema of 21 mm Hg.
tity becomes more than barely enough to begin flowing
in the pleural cavity, the excess fluid is pumped away by
Safety Factor in Chronic Conditions. When the pul-
lymphatic vessels opening directly from the pleural
monary capillary pressure remains elevated chronically
cavity into (1) the mediastinum, (2) the superior surface
(for at least 2 weeks), the lungs become even more
of the diaphragm, and (3) the lateral surfaces of the
resistant to pulmonary edema because the lymph
parietal pleura. Therefore, the pleural space—the space
vessels expand greatly, increasing their capability of car-
between the parietal and visceral pleurae—is called a
rying fluid away from the interstitial spaces perhaps as
potential space because it normally is so narrow that it
much as 10-fold. Therefore, in patients with chronic
is not obviously a physical space.
mitral stenosis, pulmonary capillary pressures of 40 to
45 mm Hg have been measured without the develop-
“Negative Pressure” in Pleural Fluid. A negative force is
ment of lethal pulmonary edema.
always required on the outside of the lungs to keep the
lungs expanded. This is provided by negative pressure
Rapidity of Death in Acute Pulmonary Edema. When the pul-
in the normal pleural space. The basic cause of this neg-
monary capillary pressure rises even slightly above the
ative pressure is pumping of fluid from the space by the
safety factor level, lethal pulmonary edema can occur
lymphatics (which is also the basis of the negative pres-
within hours, or even within 20 to 30 minutes if the cap-
sure found in most tissue spaces of the body). Because
illary pressure rises 25 to 30 mm Hg above the safety
the normal collapse tendency of the lungs is about
factor level. Thus, in acute left-sided heart failure, in
-4 mm Hg, the pleural fluid pressure must always be at
490
Unit VII
Respiration
(3) greatly reduced plasma colloid osmotic pressure,
Venous system
thus allowing excessive transudation of fluid; and (4)
infection or any other cause of inflammation of the sur-
Lymphatics
faces of the pleural cavity, which breaks down the cap-
illary membranes and allows rapid dumping of both
plasma proteins and fluid into the cavity.
Artery
References
Gehlbach BK, Geppert E: The pulmonary manifestations of
left heart failure. Chest 125:669, 2004.
Guazzi M: Alveolar-capillary membrane dysfunction in
heart failure: evidence of a pathophysiologic role. Chest
124:1090, 2003.
Guyton AC, Lindsey AW: Effect of elevated left atrial pres-
sure and decreased plasma protein concentration on the
Vein
development of pulmonary edema. Circ Res 7:649, 1959.
Guyton AC, Parker JC, Taylor AE, et al: Forces governing
water movement in the lung. In: Fishman AP, Renkin EM
(eds): Pulmonary Edema. Baltimore: Waverly Press, 1979,
p 65.
Figure 38-8
Guyton AC, Taylor AE, Granger HJ: Circulatory Physiology.
II. Dynamics and Control of the Body Fluids. Philadel-
Dynamics of fluid exchange in the intrapleural space.
phia: WB Saunders, 1975.
Hoschele S, Mairbaurl H: Alveolar flooding at high altitude:
failure of reabsorption? News Physiol Sci 18:55, 2003.
least as negative as
-4 mm Hg to keep the lungs
Lai-Fook SJ: Pleural mechanics and fluid exchange. Physiol
expanded. Actual measurements have shown that the
Rev 84:385, 2004.
pressure is usually about -7 mm Hg, which is a few mil-
Matalon S, Hardiman KM, Jain L, et al: Regulation of ion
limeters of mercury more negative than the collapse
channel structure and function by reactive oxygen-
pressure of the lungs. Thus, the negativity of the pleural
nitrogen species. Am J Physiol Lung Cell Mol Physiol
fluid keeps the normal lungs pulled against the parietal
285:L1184, 2003.
pleura of the chest cavity, except for an extremely thin
Miserocchi G, Negrini D, Passi A, De Luca G: Development
layer of mucoid fluid that acts as a lubricant.
of lung edema: interstitial fluid dynamics and molecular
structure. News Physiol Sci 16:66, 2001.
Pleural Effusion. Pleural effusion means the collection of
Schoene RB: Limits of human lung function at high altitude.
large amounts of free fluid in the pleural space. The effu-
J Exp Biol 204:3121, 2001.
sion is analogous to edema fluid in the tissues and can
Taylor AE, Guyton AC, Bishop VS: Permeability of the alve-
be called “edema of the pleural cavity.” The causes of
olar membrane to solutes. Circ Res 16:353, 1965.
the effusion are the same as the causes of edema in
Wallace J: Update in pulmonary diseases. Ann Intern Med
other tissues (discussed in Chapter 25), including (1)
139:499, 2003.
blockage of lymphatic drainage from the pleural cavity;
West JB: Respiratory Physiology—The Essentials, 5th ed.
(2) cardiac failure, which causes excessively high periph-
Baltimore: Williams & Wilkins, 1994.
eral and pulmonary capillary pressures, leading to
West JB: Invited review: pulmonary capillary stress failure.
excessive transudation of fluid into the pleural cavity;
J Appl Physiol 89:2483, 2000.
C H A P T E R
3
9
Physical Principles of Gas
Exchange; Diffusion of Oxygen
and Carbon Dioxide Through
the Respiratory Membrane
After the alveoli are ventilated with fresh air, the
next step in the respiratory process is diffusion of
oxygen from the alveoli into the pulmonary blood
and diffusion of carbon dioxide in the opposite
direction, out of the blood. The process of diffusion
is simply the random motion of molecules inter-
twining their way in all directions through
the respiratory membrane and adjacent fluids.
However, in respiratory physiology, one is concerned not only with the basic
mechanism by which diffusion occurs but also with the rate at which it occurs;
this is a much more complex problem, requiring a deeper understanding of the
physics of diffusion and gas exchange.
Physics of Gas Diffusion and Gas
Partial Pressures
Molecular Basis of Gas Diffusion
All the gases of concern in respiratory physiology are simple molecules that are free
to move among one another, which is the process called “diffusion.” This is also true
of gases dissolved in the fluids and tissues of the body.
For diffusion to occur, there must be a source of energy. This is provided by the
kinetic motion of the molecules themselves. Except at absolute zero temperature,
all molecules of all matter are continually undergoing motion. For free molecules
that are not physically attached to others, this means linear movement at high veloc-
ity until they strike other molecules. Then they bounce away in new directions and
continue until striking other molecules again. In this way, the molecules move
rapidly and randomly among one another.
Net Diffusion of a Gas in One Direction—Effect of a Concentration Gradient. If a gas chamber
or a solution has a high concentration of a particular gas at one end of the chamber
and a low concentration at the other end, as shown in Figure 39-1, net diffusion of
the gas will occur from the high-concentration area toward the low-concentration
area. The reason is obvious: There are far more molecules at end A of the chamber
to diffuse toward end B than there are molecules to diffuse in the opposite direc-
tion. Therefore, the rates of diffusion in each of the two directions are proportion-
ately different, as demonstrated by the lengths of the arrows in the figure.
Gas Pressures in a Mixture of Gases—“Partial Pressures”
of Individual Gases
Pressure is caused by multiple impacts of moving molecules against a surface. There-
fore, the pressure of a gas acting on the surfaces of the respiratory passages and
alveoli is proportional to the summated force of impact of all the molecules of that
gas striking the surface at any given instant. This means that the pressure is directly
proportional to the concentration of the gas molecules.
491
492
Unit VII
Respiration
When partial pressure is expressed in atmospheres
Dissolved gas molecules
(1 atmosphere pressure equals 760 mm Hg) and con-
centration is expressed in volume of gas dissolved in
each volume of water, the solubility coefficients for
important respiratory gases at body temperature are the
following:
Oxygen
0.024
Carbon dioxide
0.57
A
B
Carbon monoxide
0.018
Nitrogen
0.012
Figure 39-1
Helium
0.008
Diffusion of oxygen from one end of a chamber (A) to the other
(B). The difference between the lengths of the arrows represents
From this table, one can see that carbon dioxide is
net diffusion.
more than 20 times as soluble as oxygen. Therefore, the
partial pressure of carbon dioxide (for a given concen-
In respiratory physiology, one deals with mixtures of
tration) is less than one twentieth that exerted by
gases, mainly of oxygen, nitrogen, and carbon dioxide.
oxygen.
The rate of diffusion of each of these gases is directly
proportional to the pressure caused by that gas alone,
Diffusion of Gases Between the Gas Phase in the Alveoli and the
which is called the partial pressure of that gas. The
Dissolved Phase in the Pulmonary Blood. The partial pressure
concept of partial pressure can be explained as follows.
of each gas in the alveolar respiratory gas mixture tends
Consider air, which has an approximate composition
to force molecules of that gas into solution in the blood
of 79 per cent nitrogen and 21 per cent oxygen. The
of the alveolar capillaries. Conversely, the molecules of
total pressure of this mixture at sea level averages
the same gas that are already dissolved in the blood
760 mm Hg. It is clear from the preceding description of
are bouncing randomly in the fluid of the blood, and
the molecular basis of pressure that each gas contributes
some of these bouncing molecules escape back into the
to the total pressure in direct proportion to its concen-
alveoli. The rate at which they escape is directly pro-
tration. Therefore, 79 per cent of the 760 mm Hg is
portional to their partial pressure in the blood.
caused by nitrogen (600 mm Hg) and 21 per cent by
But in which direction will net diffusion of the gas
oxygen (160 mm Hg). Thus, the “partial pressure” of
occur? The answer is that net diffusion is determined by
nitrogen in the mixture is 600 mm Hg, and the “partial
the difference between the two partial pressures. If the
pressure” of oxygen is 160 mm Hg; the total pressure is
partial pressure is greater in the gas phase in the alveoli,
760 mm Hg, the sum of the individual partial pressures.
as is normally true for oxygen, then more molecules will
The partial pressures of individual gases in a mixture are
diffuse into the blood than in the other direction. Alter-
designated by the symbols Po2, Pco2, Pn2, Ph2o, Phe, and
natively, if the partial pressure of the gas is greater in
so forth.
the dissolved state in the blood, which is normally true
for carbon dioxide, then net diffusion will occur toward
the gas phase in the alveoli.
Pressures of Gases Dissolved in
Water and Tissues
Vapor Pressure of Water
Gases dissolved in water or in body tissues also exert
pressure, because the dissolved gas molecules are
When nonhumidified air is breathed into the respiratory
moving randomly and have kinetic energy. Further,
passageways, water immediately evaporates from the
when the gas dissolved in fluid encounters a surface,
surfaces of these passages and humidifies the air. This
such as the membrane of a cell, it exerts its own partial
results from the fact that water molecules, like the dif-
pressure in the same way that a gas in the gas phase
ferent dissolved gas molecules, are continually escaping
does. The partial pressures of the separate dissolved
from the water surface into the gas phase. The partial
gases are designated the same as the partial pressures
pressure that the water molecules exert to escape
in the gas state, that is, Po2, Pco2, Pn2, Phe, and so forth.
through the surface is called the vapor pressure of the
water. At normal body temperature, 37°C, this vapor
Factors That Determine the Partial Pressure of a Gas Dissolved in
pressure is 47 mm Hg. Therefore, once the gas mixture
a Fluid. The partial pressure of a gas in a solution is
has become fully humidified—that is, once it is in “equi-
determined not only by its concentration but also by the
librium” with the water—the partial pressure of the
solubility coefficient of the gas. That is, some types of
water vapor in the gas mixture is 47 mm Hg. This partial
molecules, especially carbon dioxide, are physically or
pressure, like the other partial pressures, is designated
chemically attracted to water molecules, whereas others
Ph2o.
are repelled. When molecules are attracted, far more of
The vapor pressure of water depends entirely on the
them can be dissolved without building up excess partial
temperature of the water. The greater the temperature,
pressure within the solution. Conversely, in the case of
the greater the kinetic activity of the molecules and,
those that are repelled, high partial pressure will
therefore, the greater the likelihood that the water mol-
develop with fewer dissolved molecules. These relations
ecules will escape from the surface of the water into the
are expressed by the following formula, which is Henry’s
gas phase. For instance, the water vapor pressure at
law:
0°C is 5 mm Hg, and at 100°C it is 760 mm Hg. But the
most important value to remember is the vapor pres-
Concentration of dissolved gas
sure at body temperature, 47 mm Hg; this value appears
Partial pressure
=
Solubility coefficient
in many of our subsequent discussions.
Chapter 39
Physical Principles of Gas Exchange; Diffusion of Oxygen and Carbon Dioxide
493
Diffusion of Gases Through
pathway, A is the cross-sectional area of the pathway, S
Fluids—Pressure Difference Causes
is the solubility of the gas, d is the distance of diffusion,
and MW is the molecular weight of the gas.
Net Diffusion
It is obvious from this formula that the characteristics
Now, let us return to the problem of diffusion. From the
of the gas itself determine two factors of the formula:
preceding discussion, it is clear that when the partial
solubility and molecular weight. Together, these two
pressure of a gas is greater in one area than in another
factors determine the diffusion coefficient of the gas,
area, there will be net diffusion from the high-pressure
which is proportional to S/
MW
. That is, the relative
area toward the low-pressure area. For instance, re-
rates at which different gases at the same partial pres-
turning to Figure 39-1, one can readily see that the mol-
sure levels will diffuse are proportional to their diffu-
ecules in the area of high pressure, because of their
sion coefficients. Assuming that the diffusion coefficient
greater number, have a greater statistical chance of
for oxygen is 1, the relative diffusion coefficients for dif-
moving randomly into the area of low pressure than
ferent gases of respiratory importance in the body fluids
do molecules attempting to go in the other direction.
are as follows:
However, some molecules do bounce randomly from
the area of low pressure toward the area of high pres-
Oxygen
1.0
sure. Therefore, the net diffusion of gas from the area of
Carbon dioxide
20.3
high pressure to the area of low pressure is equal to the
Carbon monoxide
0.81
number of molecules bouncing in this forward direction
Nitrogen
0.53
minus the number bouncing in the opposite direction;
Helium
0.95
this is proportional to the gas partial pressure difference
between the two areas, called simply the pressure dif-
ference for causing diffusion.
Diffusion of Gases Through Tissues
The gases that are of respiratory importance are all
Quantifying the Net Rate of Diffusion in Fluids. In addition to
highly soluble in lipids and, consequently, are highly
the pressure difference, several other factors affect the
soluble in cell membranes. Because of this, the major
rate of gas diffusion in a fluid. They are (1) the solubil-
limitation to the movement of gases in tissues is the rate
ity of the gas in the fluid, (2) the cross-sectional area of
at which the gases can diffuse through the tissue water
the fluid, (3) the distance through which the gas must
instead of through the cell membranes. Therefore, dif-
diffuse, (4) the molecular weight of the gas, and (5) the
fusion of gases through the tissues, including through
temperature of the fluid. In the body, the last of these
the respiratory membrane, is almost equal to the diffu-
factors, the temperature, remains reasonably constant
sion of gases in water, as given in the preceding list.
and usually need not be considered.
The greater the solubility of the gas, the greater the
number of molecules available to diffuse for any given
partial pressure difference. The greater the cross-
Composition of Alveolar
sectional area of the diffusion pathway, the greater the
Air—Its Relation to
total number of molecules that diffuse. Conversely,
Atmospheric Air
the greater the distance the molecules must diffuse, the
longer it will take the molecules to diffuse the entire dis-
tance. Finally, the greater the velocity of kinetic move-
Alveolar air does not have the same concentrations of
ment of the molecules, which is inversely proportional
gases as atmospheric air by any means, which can
to the square root of the molecular weight, the greater
readily be seen by comparing the alveolar air compo-
the rate of diffusion of the gas. All these factors can be
sition in Table 39-1 with that of atmospheric air. There
expressed in a single formula, as follows:
are several reasons for the differences. First, the alve-
olar air is only partially replaced by atmospheric air
P¥A ¥S
D
Dµ
,
with each breath. Second, oxygen is constantly being
d
¥
MW
absorbed into the pulmonary blood from the alveolar
in which D is the diffusion rate, DP is the partial pres-
air. Third, carbon dioxide is constantly diffusing from
sure difference between the two ends of the diffusion
the pulmonary blood into the alveoli. And fourth, dry
Table 39-1
Partial Pressures of Respiratory Gases as They Enter and Leave the Lungs (at Sea Level)
Atmospheric Air*
Humidified Air
Alveolar Air
Expired Air
(mm Hg)
(mm Hg)
(mm Hg)
(mm Hg)
N2
597.0
(78.62%)
563.4
(74.09%)
569.0
(74.9%)
566.0
(74.5%)
O2
159.0
(20.84%)
149.3
(19.67%)
104.0
(13.6%)
120.0
(15.7%)
CO2
0.3
(0.04%)
0.3
(0.04%)
40.0
(5.3%)
27.0
(3.6%)
H2O
3.7
(0.50%)
47.0
(6.20%)
47.0
(6.2%)
47.0
(6.2%)
TOTAL
760.0
(100.0%)
760.0
(100.0%)
760.0
(100.0%)
760.0
(100.0%)
* On an average cool, clear day.
494
Unit VII
Respiration
atmospheric air that enters the respiratory passages is
gas still has not been completely removed from the
humidified even before it reaches the alveoli.
alveoli.
Figure 39-3 demonstrates graphically the rate at
Humidification of the Air in the Respiratory Passages. Table
which excess gas in the alveoli is normally removed,
39-1 shows that atmospheric air is composed almost
showing that with normal alveolar ventilation, about
entirely of nitrogen and oxygen; it normally contains
one half the gas is removed in 17 seconds. When a
almost no carbon dioxide and little water vapor.
person’s rate of alveolar ventilation is only one half
However, as soon as the atmospheric air enters the res-
normal, one half the gas is removed in 34 seconds, and
piratory passages, it is exposed to the fluids that cover
when the rate of ventilation is twice normal, one half
the respiratory surfaces. Even before the air enters the
is removed in about 8 seconds.
alveoli, it becomes (for all practical purposes) totally
humidified.
Importance of the Slow Replacement of Alveolar Air. The slow
The partial pressure of water vapor at a normal
replacement of alveolar air is of particular importance
body temperature of 37°C is 47 mm Hg, which is there-
in preventing sudden changes in gas concentrations in
fore the partial pressure of water vapor in the alveo-
the blood. This makes the respiratory control mecha-
lar air. Because the total pressure in the alveoli
nism much more stable than it would be otherwise, and
cannot rise to more than the atmospheric pressure
it helps prevent excessive increases and decreases in
(760 mm Hg at sea level), this water vapor simply
tissue oxygenation, tissue carbon dioxide concentra-
dilutes all the other gases in the inspired air. Table 39-1
tion, and tissue pH when respiration is temporarily
also shows that humidification of the air dilutes the
interrupted.
oxygen partial pressure at sea level from an average
of 159 mm Hg in atmospheric air to 149 mm Hg in the
humidified air, and it dilutes the nitrogen partial pres-
Oxygen Concentration and Partial
sure from 597 to 563 mm Hg.
Pressure in the Alveoli
Oxygen is continually being absorbed from the alveoli
Rate at Which Alveolar Air Is Renewed
into the blood of the lungs, and new oxygen is contin-
by Atmospheric Air
ually being breathed into the alveoli from the atmos-
phere. The more rapidly oxygen is absorbed, the lower
In Chapter 37, it was pointed out that the average male
its concentration in the alveoli becomes; conversely,
functional residual capacity of the lungs (the volume
the more rapidly new oxygen is breathed into the
of air remaining in the lungs at the end of normal expi-
alveoli from the atmosphere, the higher its concentra-
ration) measures about 2300 milliliters. Yet only 350
tion becomes. Therefore, oxygen concentration in the
milliliters of new air is brought into the alveoli with
alveoli, and its partial pressure as well, is controlled by
each normal inspiration, and this same amount of old
(1) the rate of absorption of oxygen into the blood and
alveolar air is expired. Therefore, the volume of alve-
(2) the rate of entry of new oxygen into the lungs by
olar air replaced by new atmospheric air with each
the ventilatory process.
breath is only one seventh of the total, so that multi-
ple breaths are required to exchange most of the alve-
olar air. Figure 39-2 shows this slow rate of renewal of
the alveolar air. In the first alveolus of the figure, an
excess amount of a gas is present in the alveoli, but
note that even at the end of 16 breaths, the excess
100
80
60
40
1st breath
2nd breath
3rd breath
20
0
0
10
20
30
40
50
60
Time (seconds)
4th breath
8th breath
12th breath
16th breath
Figure 39-2
Figure 39-3
Expiration of a gas from an alveolus with successive breaths.
Rate of removal of excess gas from alveoli.
Chapter 39
Physical Principles of Gas Exchange; Diffusion of Oxygen and Carbon Dioxide
495
Upper limit at maximum ventilation
150
250 ml O2/min
125
100
A
Normal alveolar PO
2
75
50
1000 ml O2/min
25
0
Figure 39-4
0
5
10
15
20
25
30
35
40
Alveolar ventilation (L/min)
Effect of alveolar ventilation on the alveolar PO
2 at
two rates of oxygen absorption from the alveoli—
250 ml/min and 1000 ml/min. Point A is the normal
operating point.
Figure 39-4 shows the effect of both alveolar venti-
lation and rate of oxygen absorption into the blood on
the alveolar partial pressure of oxygen (Po2). One
175
curve represents oxygen absorption at a rate of
150
250 ml/min, and the other curve represents a rate of
1000 ml/min. At a normal ventilatory rate of 4.2 L/min
125
and an oxygen consumption of 250 ml/min, the normal
800 ml CO2/min
operating point in Figure 39-4 is point A. The figure
100
also shows that when 1000 milliliters of oxygen is being
75
absorbed each minute, as occurs during moderate
exercise, the rate of alveolar ventilation must increase
50
Normal alveolar PCO2
fourfold to maintain the alveolar Po2 at the normal
A
value of 104 mm Hg.
25
200 ml CO2/min
Another effect shown in Figure 39-4 is that an
extremely marked increase in alveolar ventilation can
0
0
5
10
15
20
25
30
35
40
never increase the alveolar Po2 above 149 mm Hg as
Alveolar ventilation (L/min)
long as the person is breathing normal atmospheric
air at sea level pressure, because this is the maximum
Po2 in humidified air at this pressure. If the person
breathes gases that contain partial pressures of oxygen
Figure 39-5
higher than 149 mm Hg, the alveolar Po2 can approach
these higher pressures at high rates of ventilation.
Effect of alveolar ventilation on the alveolar PCO2 at two rates of
carbon dioxide excretion from the blood—800 ml/min and 200 ml/
min. Point A is the normal operating point.
CO2 Concentration and Partial
Pressure in the Alveoli
to the rate of carbon dioxide excretion, as represented
Carbon dioxide is continually being formed in the
by the fourfold elevation of the curve (when 800
body and then carried in the blood to the alveoli; it is
milliliters of CO2 are excreted per minute). Second,
continually being removed from the alveoli by venti-
the alveolar PCO2 decreases in inverse proportion to
lation. Figure 39-5 shows the effects on the alveolar
alveolar ventilation. Therefore, the concentrations and
partial pressure of carbon dioxide (Pco2) of both alve-
partial pressures of both oxygen and carbon dioxide in
olar ventilation and two rates of carbon dioxide excre-
the alveoli are determined by the rates of absorption
tion,
200 and 800 ml/min. One curve represents a
or excretion of the two gases and by the amount of
normal rate of carbon dioxide excretion of 200 ml/min.
alveolar ventilation.
At the normal rate of alveolar ventilation of 4.2 L/min,
the operating point for alveolar Pco2 is at point A in
Expired Air
Figure 39-5—that is, 40 mm Hg.
Two other facts are also evident from Figure 39-5:
Expired air is a combination of dead space air and alve-
First, the alveolar PCO2 increases directly in proportion
olar air; its overall composition is therefore determined
496
Unit VII Respiration
160
140
120
Oxygen (PO2)
100
Dead
Alveolar air
80
space
and dead
Alveolar air
air
space air
60
Carbon dioxide (PCO2)
40
20
0
0
100
200
300
400
500
Figure 39-6
Milliliters of air expired
Oxygen and carbon dioxide partial pressures in the
various portions of normal expired air.
by (1) the amount of the expired air that is dead space
air and (2) the amount that is alveolar air. Figure 39-6
shows the progressive changes in oxygen and carbon
dioxide partial pressures in the expired air during the
course of expiration. The first portion of this air, the
Terminal bronchiole
dead space air from the respiratory passageways, is
typical humidified air, as shown in Table 39-1. Then, pro-
gressively more and more alveolar air becomes mixed
with the dead space air until all the dead space air has
finally been washed out and nothing but alveolar air is
Respiratory bronchiole
expired at the end of expiration. Therefore, the method
Alveolar
of collecting alveolar air for study is simply to collect
duct
Alveoli
Atrium
a sample of the last portion of the expired air after
forceful expiration has removed all the dead space
air.
Normal expired air, containing both dead space air
and alveolar air, has gas concentrations and partial
pressures approximately as shown in Table 39-1—that
is, concentrations between those of alveolar air and
humidified atmospheric air.
Diffusion of Gases Through
the Respiratory Membrane
Alveolar sacs
Respiratory Unit. Figure 39-7 shows the respiratory unit
(also called “respiratory lobule”), which is composed
of a respiratory bronchiole, alveolar ducts, atria, and
alveoli. There are about 300 million alveoli in the two
lungs, and each alveolus has an average diameter of
Figure 39-7
about 0.2 millimeter. The alveolar walls are extremely
Respiratory unit. (Redrawn from Miller WS: The Lung. Springfield,
thin, and between the alveoli is an almost solid
Ill: Charles C Thomas, 1947.)
network of interconnecting capillaries, shown in
Figure 39-8. Indeed, because of the extensiveness of
the capillary plexus, the flow of blood in the alveolar
and the pulmonary blood occurs through the mem-
wall has been described as a “sheet” of flowing blood.
branes of all the terminal portions of the lungs, not
Thus, it is obvious that the alveolar gases are in very
merely in the alveoli themselves. All these membranes
close proximity to the blood of the pulmonary capil-
are collectively known as the respiratory membrane,
laries. Further, gas exchange between the alveolar air
also called the pulmonary membrane.
Chapter 39
Physical Principles of Gas Exchange; Diffusion of Oxygen and Carbon Dioxide
497
Epithelial
Alveolar
basement
epithelium
membrane
Fluid and
surfactant
layer
Alveolus
Capillary
Diffusion
Oxygen
Diffusion
Carbon dioxide
A
Alveolus
Alveolus
Red blood
Interstitial space
cell
Capillaries
Lymphatic
Alveolus
vessel
Capillary endothelium
Interstitial space
Capillary basement membrane
Figure 39-9
Perivascular
Ultrastructure of the alveolar respiratory membrane, shown in
Vein
Artery
interstitial space
cross section.
Alveolus
5. A capillary basement membrane that in many
B
places fuses with the alveolar epithelial basement
membrane
Figure 39-8
6. The capillary endothelial membrane
Despite the large number of layers, the overall
A, Surface view of capillaries in an alveolar wall. B, Cross-sec-
tional view of alveolar walls and their vascular supply. (A, From
thickness of the respiratory membrane in some areas
Maloney JE, Castle BL: Pressure-diameter relations of capillaries
is as little as 0.2 micrometer, and it averages about 0.6
and small blood vessels in frog lung. Respir Physiol 7:150, 1969.
Reproduced by permission of ASP Biological and Medical Press,
micrometer, except where there are cell nuclei. From
North-Holland Division.)
histological studies, it has been estimated that the total
surface area of the respiratory membrane is about 70
square meters in the normal adult human male. This is
Respiratory Membrane. Figure 39-9 shows the ultrastruc-
equivalent to the floor area of a 25-by-30-foot room.
ture of the respiratory membrane drawn in cross
The total quantity of blood in the capillaries of the
section on the left and a red blood cell on the right. It
lungs at any given instant is 60 to 140 milliliters. Now
also shows the diffusion of oxygen from the alveolus
imagine this small amount of blood spread over the
into the red blood cell and diffusion of carbon dioxide
entire surface of a 25-by-30-foot floor, and it is easy
in the opposite direction. Note the following different
to understand the rapidity of the respiratory exchange
layers of the respiratory membrane:
of oxygen and carbon dioxide.
1. A layer of fluid lining the alveolus and containing
The average diameter of the pulmonary capillaries
surfactant that reduces the surface tension of the
is only about 5 micrometers, which means that red
alveolar fluid
blood cells must squeeze through them. The red blood
2. The alveolar epithelium composed of thin
cell membrane usually touches the capillary wall, so
epithelial cells
that oxygen and carbon dioxide need not pass through
3. An epithelial basement membrane
significant amounts of plasma as they diffuse between
4. A thin interstitial space between the alveolar
the alveolus and the red cell. This, too, increases the
epithelium and the capillary membrane
rapidity of diffusion.
498
Unit VII
Respiration
Factors That Affect the Rate
pressure of the gas in the blood represents the number
of molecules that attempt to escape from the blood
of Gas Diffusion Through the
in the opposite direction. Therefore, the difference
Respiratory Membrane
between these two pressures is a measure of the net
tendency for the gas molecules to move through the
Referring to the earlier discussion of diffusion of gases
membrane. When the partial pressure of a gas in the
in water, one can apply the same principles and math-
alveoli is greater than the pressure of the gas in
ematical formulas to diffusion of gases through the
the blood, as is true for oxygen, net diffusion from the
respiratory membrane. Thus, the factors that deter-
alveoli into the blood occurs; when the pressure of the
mine how rapidly a gas will pass through the mem-
gas in the blood is greater than the partial pressure in
brane are (1) the thickness of the membrane, (2) the
the alveoli, as is true for carbon dioxide, net diffusion
surface area of the membrane, (3) the diffusion coeffi-
from the blood into the alveoli occurs.
cient of the gas in the substance of the membrane, and
(4) the partial pressure difference of the gas between
the two sides of the membrane.
Diffusing Capacity of the
The thickness of the respiratory membrane occa-
Respiratory Membrane
sionally increases—for instance, as a result of edema
fluid in the interstitial space of the membrane and in
The ability of the respiratory membrane to exchange
the alveoli—so that the respiratory gases must then
a gas between the alveoli and the pulmonary blood
diffuse not only through the membrane but also
is expressed in quantitative terms by the respiratory
through this fluid. Also, some pulmonary diseases
membrane’s diffusing capacity, which is defined as
cause fibrosis of the lungs, which can increase the
the volume of a gas that will diffuse through the mem-
thickness of some portions of the respiratory mem-
brane each minute for a partial pressure difference of
brane. Because the rate of diffusion through the mem-
1 mm Hg. All the factors discussed earlier that affect
brane is inversely proportional to the thickness of the
diffusion through the respiratory membrane can affect
membrane, any factor that increases the thickness to
this diffusing capacity.
more than two to three times normal can interfere sig-
nificantly with normal respiratory exchange of gases.
Diffusing Capacity for Oxygen. In the average young man,
The surface area of the respiratory membrane can be
the diffusing capacity for oxygen under resting condi-
greatly decreased by many conditions. For instance,
tions averages 21 ml/min/mm Hg. In functional terms,
removal of an entire lung decreases the total surface
what does this mean? The mean oxygen pressure
area to one half normal. Also, in emphysema, many of
difference across the respiratory membrane during
the alveoli coalesce, with dissolution of many alveolar
normal, quiet breathing is about 11 mm Hg. Multipli-
walls. Therefore, the new alveolar chambers are much
cation of this pressure by the diffusing capacity (11
¥
larger than the original alveoli, but the total surface
21) gives a total of about 230 milliliters of oxygen dif-
area of the respiratory membrane is often decreased
fusing through the respiratory membrane each minute;
as much as fivefold because of loss of the alveolar
this is equal to the rate at which the resting body uses
walls. When the total surface area is decreased to
oxygen.
about one third to one fourth normal, exchange of
gases through the membrane is impeded to a signifi-
Change in Oxygen Diffusing Capacity During Exercise.
cant degree, even under resting conditions, and during
During strenuous exercise or other conditions that
competitive sports and other strenuous exercise, even
greatly increase pulmonary blood flow and alveolar
the slightest decrease in surface area of the lungs can
ventilation, the diffusing capacity for oxygen increases
be a serious detriment to respiratory exchange of
in young men to a maximum of about 65 ml/min/
gases.
mm Hg, which is three times the diffusing capacity
The diffusion coefficient for transfer of each gas
under resting conditions. This increase is caused by
through the respiratory membrane depends on the
several factors, among which are (1) opening up of
gas’s solubility in the membrane and, inversely, on the
many previously dormant pulmonary capillaries or
square root of the gas’s molecular weight. The rate
extra dilation of already open capillaries, thereby
of diffusion in the respiratory membrane is almost
increasing the surface area of the blood into which the
exactly the same as that in water, for reasons explained
oxygen can diffuse; and (2) a better match between
earlier. Therefore, for a given pressure difference,
the ventilation of the alveoli and the perfusion of the
carbon dioxide diffuses about 20 times as rapidly as
alveolar capillaries with blood, called the ventilation-
oxygen. Oxygen diffuses about twice as rapidly as
perfusion ratio, which is explained in detail later in
nitrogen.
this chapter. Therefore, during exercise, oxygenation of
The pressure difference across the respiratory mem-
the blood is increased not only by increased alveolar
brane is the difference between the partial pressure of
ventilation but also by greater diffusing capacity of
the gas in the alveoli and the partial pressure of the
the respiratory membrane for transporting oxygen into
gas in the pulmonary capillary blood. The partial pres-
the blood.
sure represents a measure of the total number of mol-
ecules of a particular gas striking a unit area of the
Diffusing Capacity for Carbon Dioxide. The diffusing capac-
alveolar surface of the membrane in unit time, and the
ity for carbon dioxide has never been measured
Chapter 39
Physical Principles of Gas Exchange; Diffusion of Oxygen and Carbon Dioxide
499
because of the following technical difficulty: Carbon
capacity by such a direct procedure, except on an exper-
dioxide diffuses through the respiratory membrane so
imental basis.
rapidly that the average Pco2 in the pulmonary blood
To obviate the difficulties encountered in measuring
oxygen diffusing capacity directly, physiologists usually
is not far different from the Pco2 in the alveoli—the
measure carbon monoxide diffusing capacity instead
average difference is less than 1 mm Hg—and with the
and then calculate the oxygen diffusing capacity from
available techniques, this difference is too small to be
this. The principle of the carbon monoxide method is
measured.
the following: A small amount of carbon monoxide is
Nevertheless, measurements of diffusion of other
breathed into the alveoli, and the partial pressure of the
gases have shown that the diffusing capacity varies
carbon monoxide in the alveoli is measured from appro-
directly with the diffusion coefficient of the particular
priate alveolar air samples. The carbon monoxide pres-
gas. Because the diffusion coefficient of carbon dioxide
sure in the blood is essentially zero, because hemoglobin
is slightly more than 20 times that of oxygen, one
combines with this gas so rapidly that its pressure never
would expect a diffusing capacity for carbon dioxide
has time to build up. Therefore, the pressure difference
of carbon monoxide across the respiratory membrane is
under resting conditions of about 400 to 450 ml/min/
equal to its partial pressure in the alveolar air sample.
mm Hg and during exercise of about 1200 to 1300 ml/
Then, by measuring the volume of carbon monoxide
min/mm Hg. Figure 39-10 compares the measured or
absorbed in a short period and dividing this by the
calculated diffusing capacities of carbon monoxide,
alveolar carbon monoxide partial pressure, one can
oxygen, and carbon dioxide at rest and during exercise,
determine accurately the carbon monoxide diffusing
showing the extreme diffusing capacity of carbon
capacity.
dioxide and the effect of exercise on the diffusing
To convert carbon monoxide diffusing capacity to
capacity of each of these gases.
oxygen diffusing capacity, the value is multiplied by a
factor of
1.23 because the diffusion coefficient for
Measurement of Diffusing Capacity—The Carbon Monoxide
oxygen is 1.23 times that for carbon monoxide. Thus,
Method. The oxygen diffusing capacity can be calculated
the average diffusing capacity for carbon monoxide
from measurements of (1) alveolar Po2, (2) Po2 in the
in young men at rest is 17 ml/min/mm Hg, and the
pulmonary capillary blood, and (3) the rate of oxygen
diffusing capacity for oxygen is
1.23 times this, or
uptake by the blood. However, measuring the Po2 in the
21 ml/min/mm Hg.
pulmonary capillary blood is so difficult and so impre-
cise that it is not practical to measure oxygen diffusing
Effect of the Ventilation-
Perfusion Ratio on Alveolar
Gas Concentration
In the early part of this chapter, we learned that two
1300
Resting
factors determine the Po2 and the Pco2 in the alveoli: (1)
1200
the rate of alveolar ventilation and (2) the rate of trans-
Exercise
fer of oxygen and carbon dioxide through the respira-
1100
tory membrane. These earlier discussions made the
assumption that all the alveoli are ventilated equally
1000
and that blood flow through the alveolar capillaries is
the same for each alveolus. However, even normally to
900
some extent, and especially in many lung diseases, some
800
areas of the lungs are well ventilated but have almost
no blood flow, whereas other areas may have excellent
700
blood flow but little or no ventilation. In either of these
conditions, gas exchange through the respiratory mem-
600
brane is seriously impaired, and the person may suffer
severe respiratory distress despite both normal total
500
ventilation and normal total pulmonary blood flow, but
400
with the ventilation and blood flow going to different
parts of the lungs. Therefore, a highly quantitative
300
concept has been developed to help us understand res-
piratory exchange when there is imbalance between
200
alveolar ventilation and alveolar blood flow. This
concept is called the ventilation-perfusion ratio.
100
In quantitativ.e te.rms, the .entilation-perfusion ratio
0
is expressed as V
a/Q. When V
a (a.veolar ventilation) is
normal for a given alveolus and Q
(blood flow) is also
CO
O2
CO2
normal.for.the same alveolus, the ventilation-perfusion
ratio (V
a/Q.
) is also said to be normal. When t .e venti-
lation (V
a) is .ero., yet there is still perfusion (Q) of the
alveolus, the V
a/Q
is zero. Or, at t.e other extreme,
Figure 39-10
when t.here is adequ.ate.ventilation (V
a) but zero perfu-
Diffusing capacities
for carbon monoxide, oxygen, and carbon
sion (Q), the ratio V
a/Q
is infinity. At a ratio of either
dioxide in the normal lungs under resting conditions and during
zero or infinity, there is no exchange of gases through
exercise.
the respiratory membrane of the affected alveoli, which
500
Unit VII
Respiration
explains the importance of this concept. Therefore, let
us explain the respiratory consequences of these two
extremes.
50
v VA /Q = 0
VA /Q = Normal
Alveolar Oxygen and .arb.n Dioxide Partial Pressures When VA/Q
40
2
(PO = 40)
Equals Zero. When V
a/Q
is equal to zero—that is, without
(PCO = 45)
2
any alveolar ventilation—the air in the alveolus
Normal alveolar
comes to equilibrium with the blood oxygen and carbon
30
air
dioxide because these gases diffuse between the blood
(PO = 104)
2
and the alveolar air. Because the blood that perfuses
20
(PCO = 40)
2
the capillaries is venous blood returning to the lungs
from the systemic circulation, it is the gases in this
10
(PO2
= 149)
VA /Q = ∞
blood with which the alveolar gases equilibrate. In
I
(PCO = 0)
2
Chapter 40, we will learn that the normal venous blood
(v¯ ) has a Po2 of 40 mm Hg and a Pco2 of 45 mm Hg.
0
20
40
60
80
100
120
140
160
Therefore, these are also the normal partial pressures of
PO2 (mm Hg)
these two gases in alveoli that have blood flow but no
ventilation.
Alveolar Oxygen and Carbon Dioxide Partial Pressures When VA/Q
Figure 39-11
Equals Infinity. .Th . effect on the alveolar gas partial pres-
sures when V
a/Q
equa.ls i.nfinity is entirely different
Normal PO2-PCO2, VA/Q
diagram.
from the effect when V
a/Q
equals zero because now
there is no capillary blood flow to carry oxygen away or
to bring carbon dioxide to the alveoli. Therefore, instead
Concept of “Physiologic Shunt”
of the alveolar gases coming to equilibrium with the
(When VA/Q
Is Below Normal)
venous blood, the alveolar air becomes equal to the
humidified inspired air. That is, the air that is inspired
Whenever Va/Q
is below normal, there is inadequate
loses no oxygen to the blood and gains no carbon
ventilation to provide the oxygen needed to fully oxy-
dioxide from the blood. And because normal inspired
genate the blood flowing through the alveolar capil-
and humidified air has a Po2 of 149 mm Hg and a Pco2
laries. Therefore, a certain fraction of the venous
of 0 mm Hg, these will be the partial pressures of these
blood passing through the pulmonary capillaries does
two gases in the alveoli.
not become oxygenated. This fraction is called shunted
blood. Also, some additional blood flows through
Gas Exchange and Alveolar Partial Pressures When VA/Q
Is
bronchial vessels rather than through alveolar capillar-
Normal. When there is both normal alveolar ventilation
ies, normally about 2 per cent of the cardiac output; this,
and normal alveolar capillary blood flow (normal alve-
too, is unoxygenated, shunted blood.
olar perfusion), exchange of oxygen and carbon dioxide
The total quantitative amount of shunted blood per
through the respiratory membrane is nearly optimal,
minute is called the physiologic shunt. This physiologic
and alveolar Po2 is normally at a level of 104 mm Hg,
shunt is measured in clinical pulmonary function labo-
which lies between that of the inspired air (149 mm Hg)
ratories by analyzing the concentration of oxygen in
and that of venous blood
(40 mm Hg). Likewise,
both mixed venous blood and arterial blood, along with
alveolar Pco2 lies between two extremes; it is normally
simultaneous measurement of cardiac output. From
40 mm Hg, in contrast to 45 mm Hg in venous blood and
these values, the physiologic shunt can be calculated by
0 mm Hg in inspired air. Thus, under normal conditions,
the following equation:
the alveolar air Po2 averages 104 mm Hg and the Pco2
averages 40 mm Hg.
Qps
Ci
O
2
-Ca
O
2
=
Qt
Ci
-Cv
O
2
O
2
PO2-PCO2, VA/Q
Diagram
in which.Qps is the physiologic shunt blood flow per
minute, Qt is cardiac output per minute, Ci is the con-O
2
The concepts presented in the preceding sections can be
centration of oxygen in the arterial blood if there is an
shown in graphical form, .s .emonstrated in Figure
“ideal” ventilation-perfusion ratio, Ca is the measuredO
2
39-11, called the Po2-Pco2, V
a/Q
diagram. The curve in
concentration of oxygen in the arterial blood, and C
¯O
2
the diagram represents all possib.e .o2 and Pco2 com-
is the measured concentration of oxygen in the mixed
b.ina.tions between the limits of V
a/Q
equals zero and
venous blood.
V
a/Q
equals infinity when the gas pressures in the
The greater the physiologic shunt, the greater the
venous blood are normal and the person is breathing
amount of blood that fails to be oxygenated as it passes
air at sea-level p.res.ure. Thus, point v¯ is the plot of Po2
through the lungs.
and Pco2 when V
a/Q
equals zero. At this point, the Po2
is 40 mm Hg and the Pco2 is 45 mm Hg, which are the
values in normal venous blood.
Concept of the “Physiologic Dead
At the other end of the curve, when Va/ Qequals infin-
Space” (When VA/Q
Is Greater
ity, point I represents inspired air, showing Po2 to be 149
Than Normal)
mm Hg while Pco2 is zero. Also plotted on the curve
i. th.e point that represents normal alveolar air when
When ventilation of some of the alveoli is great but
V
a/Q
is normal. At this point, Po2 is 104 mm Hg and
alveolar blood flow is low, there is far more available
Pco2 is 40 mm Hg.
oxygen in the alveoli than can be transported away from
Chapter 39
Physical Principles of Gas Exchange; Diffusion of Oxygen and Carbon Dioxide
501
the alveoli by the flowing blood. Thus, the ventilation of
t.wo.abnormalities occur in smokers to cause abnormal
these alveoli is said to be wasted. The ventilation of the
V
a/Q. First, because many of the small bronchioles are
anatomical dead space areas of the respiratory pas-
obstructed, the alveoli b.eyo.nd the obstructions are
sageways is also wasted. The sum of these two types of
unventilated, causing a V
a/Q
that approaches zero.
wasted ventilation is called the physiologic dead space.
Second, in those areas of the lung where the alveolar
This is measured in the clinical pulmonary function lab-
walls have been mainly destroyed but there is still
oratory by making appropriate blood and expiratory
alveolar ventilation, most of the ventilation is wasted
gas measurements and using the following equation,
because of inadequate blood flow to transport the blood
called the Bohr equation:
gases.
Thus, in chronic obstructive lung disease, some areas
Vd
phys
Pa
CO
2
-Pe
CO
2
of the lung exhibit serious physiologic shunt, and other
=
,
Vt
PaCO
areas exhibit serious physiologic dead space. Both these
2
conditions tremendously decrease the effectiveness of
in which Vdphys is the physiologic dead space, Vt is the
the lungs as gas exchange organs, sometimes reducing
tidal volume, Pa
is the partial pressure of carbon
CO2
their effectiveness to as little as one tenth normal. In
dioxide in the arterial blood, and
is the average
PeCO2
fact, this is the most prevalent cause of pulmonary dis-
partial pressure of carbon dioxide in the entire expired
ability today.
air.
When the physiologic dead space is great, much of the
work of ventilation is wasted effort because so much of
the ventilating air never reaches the blood.
References
Abnormalities of Ventilation-
Albert R, Spiro S, Jett J: Comprehensive Respiratory Medi-
Perfusion Ratio
cine. Philadelphia: Mosby, 2002.
Cole RP: CO2 and lung mechanical or gas exchange func-
tion. Crit Care Med 32:1240, 2004.
Abnormal VA/Q
in the Upper and Lower Normal Lung. In a
Guazzi M: Alveolar-capillary membrane dysfunction in
normal person in the upright position, both pulmonary
heart failure: evidence of a pathophysiologic role. Chest
capillary blood flow and alveolar ventilation are con-
124:1090, 2003.
siderably less in the upper part of the lung than in the
Hsia CC: Recruitment of lung diffusing capacity: update of
lower part; however, blood flow is decreased consider-
concept and application. Chest 122:1774, 2002.
ably more.th.n ventilation is. Therefore, at the top of
Knight DA, Holgate ST: The airway epithelium: structural
the lung, V
a/Q
is as much as 2.5 times as great as the
and functional properties in health and disease. Respirol-
ideal value, which causes a moderate degree of physio-
ogy 8:432, 2003.
logic dead space in this area of the lung.
Otis AB: Quantitative relationships in steady-state gas
At the other extreme, in the bottom of the lung, there
exchange. In: Fenn WQ, Rahn H (eds): Handbook of Phys-
is slig.tly.too little ventilation in relation to blood flow,
iology. Sec 3, Vol 1. Baltimore: Williams & Wilkins, 1964,
with V
a/Q
as low as 0.6 times the ideal value. In this
p 681.
area, a small fraction of the blood fails to become
Parker JC, Townsley MI: Evaluation of lung injury in rats and
normally oxygenated, and this represents a physiologic
mice. Am J Physiol Lung Cell Mol Physiol 286:L231, 2004.
shunt.
Powell FL, Hopkins SR: Comparative physiology of lung
In both extremes, inequalities of ventilation and
complexity: implications for gas exchange. News Physiol
perfusion decrease slightly the lung’s effectiveness
Sci 19:55, 2004.
for exchanging oxygen and carbon dioxide. However,
Rahn H, Farhi EE: Ventilation, perfusion, and gas
during exercise, blood flow to the upper part of the lung
exchange—the Va/Q concept. In: Fenn WO, Rahn H (eds):
increases markedly, so that far less physiologic dead
Handbook of Physiology. Sec 3, Vol 1. Baltimore: Williams
space occurs, and the effectiveness of gas exchange now
& Wilkins, 1964, p 125.
approaches optimum.
Uhlig S, Taylor AE: Methods in Pulmonary Research. Basel:
Birkhauser Verlag, 1998.
Abnormal VA/Q
in Chronic Obstructive Lung Disease. Most
West JB: Pulmonary Physiology and Pathophysiology: An
people who smoke for many years develop various
Integrated, Case-Based Approach. Philadelphia: Lippin-
degrees of bronchial obstruction; in a large share of
cott Williams & Wilkins, 2001.
these persons, this condition eventually becomes so
West JB: Pulmonary Physiology—The Essentials. Baltimore:
severe that they develop serious alveolar air trapping
Lippincott Williams & Wilkins, 2003.
and resultant emphysema. The emphysema in turn
Williams MC: Alveolar type I cells: molecular phenotype and
causes many of the alveolar walls to be destroyed. Thus,
development. Annu Rev Physiol 65:669, 2003.
C H A P T E R
4
0
Transport of Oxygen and Carbon
Dioxide in Blood and Tissue Fluids
Once oxygen has diffused from the alveoli into
the pulmonary blood, it is transported to the
peripheral tissue capillaries almost entirely in com-
bination with hemoglobin. The presence of hemo-
globin in the red blood cells allows the blood to
transport 30 to 100 times as much oxygen as could
be transported in the form of dissolved oxygen in
the water of the blood.
In the body’s tissue cells, oxygen reacts with various foodstuffs to form large
quantities of carbon dioxide. This carbon dioxide enters the tissue capillaries
and is transported back to the lungs. Carbon dioxide, like oxygen, also combines
with chemical substances in the blood that increase carbon dioxide transport
15- to 20-fold.
The purpose of this chapter is to present both qualitatively and quantitatively
the physical and chemical principles of oxygen and carbon dioxide transport in
the blood and tissue fluids.
Transport of Oxygen from the Lungs to the
Body Tissues
In Chapter 39, we pointed out that gases can move from one point to another
by diffusion and that the cause of this movement is always a partial pressure
difference from the first point to the next. Thus, oxygen diffuses from the alveoli
into the pulmonary capillary blood because the oxygen partial pressure (Po2)
in the alveoli is greater than the Po2 in the pulmonary capillary blood. In the
other tissues of the body, a higher Po2 in the capillary blood than in the tissues
causes oxygen to diffuse into the surrounding cells.
Conversely, when oxygen is metabolized in the cells to form carbon dioxide,
the intracellular carbon dioxide pressure (Pco2) rises to a high value, which
causes carbon dioxide to diffuse into the tissue capillaries. After blood flows to
the lungs, the carbon dioxide diffuses out of the blood into the alveoli, because
the Pco2 in the pulmonary capillary blood is greater than that in the alveoli.
Thus, the transport of oxygen and carbon dioxide by the blood depends on both
diffusion and the flow of blood. We now consider quantitatively the factors
responsible for these effects.
Diffusion of Oxygen from the Alveoli to the Pulmonary
Capillary Blood
The top part of Figure 40-1 shows a pulmonary alveolus adjacent to a pul-
monary capillary, demonstrating diffusion of oxygen molecules between the
alveolar air and the pulmonary blood. The Po2 of the gaseous oxygen in the
alveolus averages 104 mm Hg, whereas the Po2 of the venous blood entering the
pulmonary capillary at its arterial end averages only 40 mm Hg because a large
amount of oxygen was removed from this blood as it passed through the periph-
eral tissues. Therefore, the initial pressure difference that causes oxygen to
diffuse into the pulmonary capillary is 104 - 40, or 64 mm Hg. In the graph at
502
Chapter 40
Transport of Oxygen and Carbon Dioxide in Blood and Tissue Fluids
503
Mixed with
Alveolus PO
2 = 104 mm Hg
pulmonary
shunt blood
100
Systemic
Pulmonary Capillary
venous
PO2 = 40 mm Hg
PO2 = 104 mm Hg
blood
80
Arterial End
Venous End
Pulmonary
Systemic
Systemic
Systemic
capillaries
arterial
capillaries
venous
110
60
Alveolar oxygen partial pressure
blood
blood
100
40
90
20
80
70
0
60
50
Figure 40-2
40
Changes in PO2 in the pulmonary capillary blood, systemic arte-
rial blood, and systemic capillary blood, demonstrating the effect
of “venous admixture.”
Figure 40-1
Therefore, during exercise, even with a shortened time
Uptake of oxygen by the pulmonary capillary blood. (The curve in
of exposure in the capillaries, the blood can still
this figure was constructed from data in Milhorn HT Jr, Pulley PE
become fully oxygenated, or nearly so.
Jr: A theoretical study of pulmonary capillary gas exchange and
venous admixture. Biophys J 8:337, 1968.)
Transport of Oxygen in the
Arterial Blood
the bottom of the figure, the curve shows the rapid rise
in blood Po2 as the blood passes through the capillary;
About 98 per cent of the blood that enters the left
the blood Po2 rises almost to that of the alveolar air by
atrium from the lungs has just passed through the alve-
the time the blood has moved a third of the distance
olar capillaries and has become oxygenated up to a Po2
through the capillary, becoming almost 104 mm Hg.
of about 104 mm Hg. Another 2 per cent of the blood
has passed from the aorta through the bronchial cir-
Uptake of Oxygen by the Pulmonary Blood During Exercise.
culation, which supplies mainly the deep tissues of the
During strenuous exercise, a person’s body may
lungs and is not exposed to lung air. This blood flow is
require as much as 20 times the normal amount of
called “shunt flow,” meaning that blood is shunted past
oxygen. Also, because of increased cardiac output
the gas exchange areas. On leaving the lungs, the Po2
during exercise, the time that the blood remains in the
of the shunt blood is about that of normal systemic
pulmonary capillary may be reduced to less than one
venous blood, about
40 mm Hg. When this blood
half normal. Yet, because of the great safety factor for
combines in the pulmonary veins with the oxygenated
diffusion of oxygen through the pulmonary mem-
blood from the alveolar capillaries, this so-called
brane, the blood still becomes almost saturated with
venous admixture of blood causes the Po2 of the blood
oxygen by the time it leaves the pulmonary capillaries.
entering the left heart and pumped into the aorta to
This can be explained as follows.
fall to about 95 mm Hg. These changes in blood Po2 at
First, it was pointed out in Chapter 39 that the dif-
different points in the circulatory system are shown in
fusing capacity for oxygen increases almost threefold
Figure 40-2.
during exercise; this results mainly from increased
surface area of capillaries participating in the diffusion
Diffusion of Oxygen from the
and also from a more nearly ideal ventilation-perfu-
sion ratio in the upper part of the lungs.
Peripheral Capillaries into the
Second, note in the curve of Figure 40-1 that under
Tissue Fluid
nonexercising conditions, the blood becomes almost
saturated with oxygen by the time it has passed
When the arterial blood reaches the peripheral tissues,
through one third of the pulmonary capillary, and little
its Po2 in the capillaries is still 95 mm Hg. Yet, as shown
additional oxygen normally enters the blood during
in Figure 40-3, the Po2 in the interstitial fluid that sur-
the latter two thirds of its transit. That is, the blood
rounds the tissue cells averages only 40 mm Hg. Thus,
normally stays in the lung capillaries about three
there is a tremendous initial pressure difference that
times as long as necessary to cause full oxygenation.
causes oxygen to diffuse rapidly from the capillary
504
Unit VII
Respiration
In summary, tissue Po2 is determined by a balance
Arterial end
Venous end
between (1) the rate of oxygen transport to the tissues
40 mm Hg
of capillary
of capillary
in the blood and (2) the rate at which the oxygen is used
by the tissues.
PO2 = 95 mm Hg
23 mm Hg
PO2
= 40 mm Hg
Diffusion of Oxygen from the
Peripheral Capillaries to the
Figure 40-3
Tissue Cells
Diffusion of oxygen from a tissue capillary to the cells. (PO2
in
interstitial fluid = 40 mm Hg, and in tissue cells = 23 mm Hg.)
Oxygen is always being used by the cells. Therefore,
the intracellular Po2
in the peripheral tissue cells
remains lower than the Po2 in the peripheral capillar-
ies. Also, in many instances, there is considerable phys-
ical distance between the capillaries and the cells.
100
Upper limit of infinite blood flow
Therefore, the normal intracellular Po2 ranges from
as low as 5 mm Hg to as high as 40 mm Hg, averaging
80
(by direct measurement in lower animals) 23 mm Hg.
Because only 1 to 3 mm Hg of oxygen pressure is
B
normally required for full support of the chemical
60
processes that use oxygen in the cell, one can see that
even this low intracellular Po2 of 23 mm Hg is more
40
than adequate and provides a large safety factor.
A
20
C
Diffusion of Carbon Dioxide from
the Peripheral Tissue Cells into the
0
0
100
200
300
400
500
600
700
Capillaries and from the Pulmonary
Blood flow (per cent of normal)
Capillaries into the Alveoli
When oxygen is used by the cells, virtually all of it
becomes carbon dioxide, and this increases the intra-
Figure 40-4
cellular Pco2; because of this high tissue cell Pco2,
Effect of blood flow and rate of oxygen consumption on tissue PO2.
carbon dioxide diffuses from the cells into the tissue
capillaries and is then carried by the blood to the lungs.
In the lungs, it diffuses from the pulmonary capillaries
blood into the tissues—so rapidly that the capillary Po2
into the alveoli and is expired.
falls almost to equal the 40 mm Hg pressure in the
Thus, at each point in the gas transport chain, carbon
interstitium. Therefore, the Po2 of the blood leaving
dioxide diffuses in the direction exactly opposite to the
the tissue capillaries and entering the systemic veins is
diffusion of oxygen. Yet there is one major difference
also about 40 mm Hg.
between diffusion of carbon dioxide and of oxygen:
carbon dioxide can diffuse about 20 times as rapidly as
Effect of Rate of Blood Flow on Interstitial Fluid PO
2. If the
oxygen. Therefore, the pressure differences required to
blood flow through a particular tissue is increased,
cause carbon dioxide diffusion are, in each instance, far
greater quantities of oxygen are transported into the
less than the pressure differences required to cause
tissue, and the tissue Po2
becomes correspondingly
oxygen diffusion. The CO2 pressures are approxi-
higher. This is shown in Figure 40-4. Note that an
mately the following:
increase in flow to 400 per cent of normal increases
1. Intracellular Pco2, 46 mm Hg; interstitial Pco2,
the Po2 from 40 mm Hg (at point A in the figure) to
66 mm Hg (at point B). However, the upper limit to
45 mm Hg. Thus, there is only a 1 mm Hg pressure
which the Po2 can rise, even with maximal blood flow, is
differential, as shown in Figure 40-5.
95 mm Hg, because this is the oxygen pressure in the
2. Pco2 of the arterial blood entering the tissues,
arterial blood. Conversely, if blood flow through the
40 mm Hg; Pco2 of the venous blood leaving the
tissue decreases, the tissue Po2 also decreases, as shown
tissues, 45 mm Hg. Thus, as shown in Figure 40-5,
at point C.
the tissue capillary blood comes almost exactly
of
to equilibrium with the interstitial Pco2
Effect of Rate of Tissue Metabolism on Interstitial Fluid PO
2. If
45 mm Hg.
the cells use more oxygen for metabolism than nor-
3. Pco2 of the blood entering the pulmonary
mally, this reduces the interstitial fluid Po2. Figure 40-4
capillaries at the arterial end, 45 mm Hg; Pco2 of
also demonstrates this effect, showing reduced intersti-
tial fluid Po2 when the cellular oxygen consumption is
the alveolar air, 40 mm Hg. Thus, only a 5 mm Hg
increased, and increased Po2
when consumption is
pressure difference causes all the required carbon
decreased.
dioxide diffusion out of the pulmonary capillaries
Chapter 40
Transport of Oxygen and Carbon Dioxide in Blood and Tissue Fluids
505
Arterial end
Venous end
of capillary
45 mm Hg
of capillary
120
PCO2 = 40 mm Hg
46 mm Hg
PCO2 = 45 mm Hg
100
80
Figure 40-5
10
normal metabolism
B
Uptake of carbon dioxide by the blood in the tissue capillaries.
60
(PCO2
in tissue cells
= 46 mm Hg, and in interstitial fluid
=
A
Normal metabolism
45 mm Hg.)
40
C
Lower limit of infinite blood flow
1/4 normal metabolism
20
Alveolus PCO
2 = 40 mm Hg
0
0
100
200
300
400
500
600
Blood flow (per cent of normal)
Pulmonary Capillary
PCO2 = 45 mm Hg
PCO
2 = 40 mm Hg
Arterial End
Venous End
45
Figure 40-7
44
Effect of blood flow and metabolic rate on peripheral tissue PCO2.
43
42
Pco2 from the normal value of 45 mm Hg to
41 mm Hg, down to a level almost equal to the
41
Pco2 in the arterial blood (40 mm Hg) entering
Pulmonary capillary blood
the tissue capillaries.
40
Alveolar carbon dioxide partial pressure
2. Note also that a 10-fold increase in tissue
metabolic rate greatly elevates the interstitial fluid
Pco2 at all rates of blood flow, whereas decreasing
the metabolism to one quarter normal causes the
Figure 40-6
interstitial fluid Pco2 to fall to about 41 mm Hg,
closely approaching that of the arterial blood,
Diffusion of carbon dioxide from the pulmonary blood into the alve-
40 mm Hg.
olus. (This curve was constructed from data in Milhorn HT Jr,
Pulley PE Jr: A theoretical study of pulmonary capillary gas
exchange and venous admixture. Biophys J 8:337, 1968.)
Role of Hemoglobin in Oxygen
Transport
into the alveoli. Furthermore, as shown in Figure
40-6, the Pco2 of the pulmonary capillary blood
Normally, about 97 per cent of the oxygen transported
falls to almost exactly equal the alveolar Pco2 of
from the lungs to the tissues is carried in chemical
40 mm Hg before it has passed more than about
combination with hemoglobin in the red blood cells.
one third the distance through the capillaries. This
The remaining 3 per cent is transported in the dis-
is the same effect that was observed earlier for
solved state in the water of the plasma and blood cells.
oxygen diffusion, except that it is in the opposite
Thus, under normal conditions, oxygen is carried to the
direction.
tissues almost entirely by hemoglobin.
Effect of Rate of Tissue Metabolism and Tissue Blood Flow on
Reversible Combination of Oxygen
Interstitial PCO2. Tissue capillary blood flow and tissue
metabolism affect the Pco2 in ways exactly opposite to
with Hemoglobin
their effect on tissue Po2. Figure 40-7 shows these
effects, as follows:
The chemistry of hemoglobin is presented in Chapter
1. A decrease in blood flow from normal (point A)
32, where it was pointed out that the oxygen molecule
to one quarter normal (point B) increases
combines loosely and reversibly with the heme portion
peripheral tissue Pco2 from the normal value of
of hemoglobin. When Po2 is high, as in the pulmonary
45 mm Hg to an elevated level of 60 mm Hg.
capillaries, oxygen binds with the hemoglobin, but
Conversely, increasing the blood flow to six
when Po2 is low, as in the tissue capillaries, oxygen is
times normal (point C) decreases the interstitial
released from the hemoglobin. This is the basis for
506
Unit VII Respiration
100
20
90
18
Oxygenated blood
80
leaving the lungs
16
70
14
60
12
50
10
Reduced blood returning
40
8
from tissues
30
6
20
4
10
2
0
0
0
10
20
30
40
50
60
70
80
90
100
110
120
130
140
Pressure of oxygen in blood (PO2) (mm Hg)
Figure 40-8
Oxygen-hemoglobin dissociation curve.
almost all oxygen transport from the lungs to the
tissues.
Oxygen-Hemoglobin Dissociation Curve. Figure 40-8 shows
20
in
the oxygen-hemoglobin dissociation curve, which
18
demonstrates a progressive increase in the percentage
16
of hemoglobin bound with oxygen as blood Po2
14
increases, which is called the per cent saturation of
12
hemoglobin. Because the blood leaving the lungs and
10
entering the systemic arteries usually has a Po2 of
8
about 95 mm Hg, one can see from the dissociation
6
curve that the usual oxygen saturation of systemic arte-
4
rial blood averages 97 per cent. Conversely, in normal
2
venous blood returning from the peripheral tissues, the
0
Po2 is about 40 mm Hg, and the saturation of hemo-
0
20
40
60
80
100
120
140
globin averages 75 per cent.
Pressure of oxygen in blood (PO2) (mm Hg)
Maximum Amount of Oxygen That Can Combine with the
Hemoglobin of the Blood. The blood of a normal person
contains about 15 grams of hemoglobin in each 100
Figure 40-9
milliliters of blood, and each gram of hemoglobin can
bind with a maximum of 1.34 milliliters of oxygen (1.39
Effect of blood PO2 on the quantity of oxygen bound with hemo-
globin in each 100 milliliters of blood.
milliliters when the hemoglobin is chemically pure, but
impurities such as methemoglobin reduce this). There-
fore, 15 times 1.34 equals 20.1, which means that, on
average, the 15 grams of hemoglobin in 100 milliliters
systemic arterial blood, which is 97 per cent saturated,
of blood can combine with a total of almost exactly 20
is about 19.4 milliliters per 100 milliliters of blood. This
milliliters of oxygen if the hemoglobin is 100 per cent
is shown in Figure 40-9. On passing through the tissue
saturated. This is usually expressed as 20 volumes per
capillaries, this amount is reduced, on average, to
cent. The oxygen-hemoglobin dissociation curve for
14.4 milliliters (Po2 of 40 mm Hg, 75 per cent saturated
the normal person can also be expressed in terms of
hemoglobin). Thus, under normal conditions, about 5
volume per cent of oxygen, as shown by the far right
milliliters of oxygen are transported from the lungs to
scale in Figure 40-8, instead of per cent saturation of
the tissues by each 100 milliliters of blood flow.
hemoglobin.
Transport of Oxygen During Strenuous Exercise. During
Amount of Oxygen Released from the Hemoglobin When Sys-
heavy exercise, the muscle cells use oxygen at a rapid
temic Arterial Blood Flows Through the Tissues. The total
rate, which, in extreme cases, can cause the muscle
quantity of oxygen bound with hemoglobin in normal
interstitial fluid Po2 to fall from the normal 40 mm Hg
Chapter 40
Transport of Oxygen and Carbon Dioxide in Blood and Tissue Fluids
507
to as low as 15 mm Hg. At this low pressure, only 4.4
because of (1) the steep slope of the dissociation curve
milliliters of oxygen remain bound with the hemoglo-
and (2) the increase in tissue blood flow caused by the
bin in each 100 milliliters of blood, as shown in Figure
decreased Po2; that is, a very small fall in Po2 causes
40-9. Thus, 19.4 - 4.4, or 15 milliliters, is the quantity
large amounts of extra oxygen to be released from the
of oxygen actually delivered to the tissues by each 100
hemoglobin. It can be seen, then, that the hemoglobin
milliliters of blood flow. Thus, three times as much
in the blood automatically delivers oxygen to the
oxygen as normal is delivered in each volume of blood
tissues at a pressure that is held rather tightly between
that passes through the tissues. And keep in mind that
about 15 and 40 mm Hg.
the cardiac output can increase to six to seven times
normal in well-trained marathon runners. Thus, multi-
When Atmospheric Oxygen Concentration Changes Markedly,
plying the increase in cardiac output (six- to sevenfold)
the Buffer Effect of Hemoglobin Still Maintains Almost Constant
by the increase in oxygen transport in each volume of
Tissue PO2. The normal Po2
in the alveoli is about
blood (threefold) gives a 20-fold increase in oxygen
104 mm Hg, but as one ascends a mountain or ascends
transport to the tissues. We see later in the chapter
in an airplane, the Po2 can easily fall to less than half
that several other factors facilitate delivery of oxygen
this amount. Alternatively, when one enters areas of
into muscles during exercise, so that muscle tissue Po2
compressed air, such as deep in the sea or in pressur-
often falls very little below normal even during very
ized chambers, the Po2 may rise to 10 times this level.
strenuous exercise.
Even so, the tissue Po2 changes little.
It can be seen from the oxygen-hemoglobin dissoci-
Utilization Coefficient. The percentage of the blood that
ation curve in Figure 40-8 that when the alveolar Po2
gives up its oxygen as it passes through the tissue cap-
is decreased to as low as 60 mm Hg, the arterial hemo-
illaries is called the utilization coefficient. The normal
globin is still 89 per cent saturated with oxygen—only
value for this is about 25 per cent, as is evident from
8 per cent below the normal saturation of 97 per cent.
the preceding discussion—that is, 25 per cent of the
Further, the tissues still remove about 5 milliliters of
oxygenated hemoglobin gives its oxygen to the tissues.
oxygen from each 100 milliliters of blood passing
During strenuous exercise, the utilization coefficient in
through the tissues; to remove this oxygen, the Po2
the entire body can increase to 75 to 85 per cent. And
of the venous blood falls to 35 mm Hg—only 5 mm Hg
in local tissue areas where blood flow is extremely
below the normal value of 40 mm Hg. Thus, the tissue
slow or the metabolic rate is very high, utilization
Po2 hardly changes, despite the marked fall in alveolar
coefficients approaching
100 per cent have been
Po2 from 104 to 60 mm Hg.
recorded—that is, essentially all the oxygen is given to
Conversely, when the alveolar Po2 rises as high as
the tissues.
500 mm Hg, the maximum oxygen saturation of
hemoglobin can never rise above 100 per cent, which
is only 3 per cent above the normal level of 97 per cent.
Effect of Hemoglobin to “Buffer” the
Only a small amount of additional oxygen dissolves in
Tissue PO2
the fluid of the blood, as will be discussed subse-
quently. Then, when the blood passes through the
Although hemoglobin is necessary for the transport of
tissue capillaries and loses several milliliters of oxygen
oxygen to the tissues, it performs another function
to the tissues, this reduces the Po2 of the capillary
essential to life. This is its function as a “tissue oxygen
blood to a value only a few millimeters greater than
buffer” system. That is, the hemoglobin in the blood is
the normal 40 mm Hg. Consequently, the level of alve-
mainly responsible for stabilizing the oxygen pressure
olar oxygen may vary greatly—from 60 to more than
in the tissues. This can be explained as follows.
500 mm Hg Po2—and still the Po2 in the peripheral
tissues does not vary more than a few millimeters from
Role of Hemoglobin in Maintaining Nearly Constant PO2 in the
normal, demonstrating beautifully the tissue “oxygen
Tissues. Under basal conditions, the tissues require
buffer” function of the blood hemoglobin system.
about 5 milliliters of oxygen from each 100 milliliters
of blood passing through the tissue capillaries. Refer-
ring back to the oxygen-hemoglobin dissociation curve
Factors That Shift the Oxygen-
in Figure 40-9, one can see that for the normal 5 mil-
Hemoglobin Dissociation Curve—
liliters of oxygen to be released per 100 milliliters of
blood flow, the Po2 must fall to about 40 mm Hg.
Their Importance for Oxygen
Therefore, the tissue Po2 normally cannot rise above
Transport
this 40 mm Hg level, because if it did, the amount of
oxygen needed by the tissues would not be released
The oxygen-hemoglobin dissociation curves of Figures
from the hemoglobin. In this way, the hemoglobin nor-
40-8 and
40-9 are for normal, average blood.
mally sets an upper limit on the oxygen pressure in the
However, a number of factors can displace the disso-
tissues at about 40 mm Hg.
ciation curve in one direction or the other in the
Conversely, during heavy exercise, extra amounts of
manner shown in Figure 40-10. This figure shows
oxygen (as much as 20 times normal) must be deliv-
that when the blood becomes slightly acidic, with the
ered from the hemoglobin to the tissues. But this can
pH decreasing from the normal value of 7.4 to 7.2, the
be achieved with little further decrease in tissue Po2
oxygen-hemoglobin dissociation curve shifts, on
508
Unit VII
Respiration
oxygen-hemoglobin dissociation curve to the left and
upward. Therefore, the quantity of oxygen that binds
with the hemoglobin at any given alveolar Po2
becomes considerably increased, thus allowing greater
100
oxygen transport to the tissues.
90
80
Effect of BPG to Shift the Oxygen-Hemoglobin Dissociation
70
pH
Curve. The normal BPG in the blood keeps the
oxygen-hemoglobin dissociation curve shifted slightly
60
Shift to right:
to the right all the time. In hypoxic conditions that
50
(1) Increased hydrogen ions
(2) Increased CO2
last longer than a few hours, the quantity of BPG in
40
(3) Increased temperature
the blood increases considerably, thus shifting the
30
(4) Increased BPG
oxygen-hemoglobin dissociation curve even farther to
20
the right. This causes oxygen to be released to the
10
tissues at as much as 10 mm Hg higher tissue oxygen
0
pressure than would be the case without this increased
0 10 20 30 40 50 60 70 80 90100110
120130
140
BPG. Therefore, under some conditions, the BPG
Pressure of oxygen in blood (PO2) (mm Hg)
mechanism can be important for adaptation to
hypoxia, especially to hypoxia caused by poor tissue
blood flow.
Shift of the Dissociation Curve During Exercise. During exer-
Figure 40-10
cise, several factors shift the dissociation curve con-
Shift of the oxygen-hemoglobin dissociation curve to the right
siderably to the right, thus delivering extra amounts
caused by an increase in hydrogen ion concentration (decrease
of oxygen to the active, exercising muscle fibers. The
in pH). BPG, 2,3-biphosphoglycerate.
exercising muscles, in turn, release large quantities of
carbon dioxide; this and several other acids released
by the muscles increase the hydrogen ion concentra-
average, about 15 per cent to the right. Conversely, an
tion in the muscle capillary blood. In addition, the tem-
increase in pH from the normal 7.4 to 7.6 shifts the
perature of the muscle often rises 2° to 3°C, which can
curve a similar amount to the left.
increase oxygen delivery to the muscle fibers even
In addition to pH changes, several other factors are
more. All these factors act together to shift the oxygen-
known to shift the curve. Three of these, all of which
hemoglobin dissociation curve of the muscle capillary
shift the curve to the right, are (1) increased carbon
blood considerably to the right. This right-hand shift
dioxide concentration, (2) increased blood tempera-
of the curve forces oxygen to be released from the
ture, and (3) increased 2,3-biphosphoglycerate (BPG),
blood hemoglobin to the muscle at Po2 levels as great
a metabolically important phosphate compound
as 40 mm Hg, even when 70 per cent of the oxygen has
present in the blood in different concentrations under
already been removed from the hemoglobin. Then, in
different metabolic conditions.
the lungs, the shift occurs in the opposite direction,
allowing the pickup of extra amounts of oxygen from
Increased Delivery of Oxygen to the Tissues When Carbon
the alveoli.
Dioxide and Hydrogen Ions Shift the Oxygen-Hemoglobin Disso-
ciation Curve—The Bohr Effect. A shift of the oxygen-
hemoglobin dissociation curve to the right in response
Metabolic Use of Oxygen by the Cells
to increases in blood carbon dioxide and hydrogen
ions has a significant effect by enhancing the release
Effect of Intracellular PO2 on Rate of Oxygen Usage. Only
of oxygen from the blood in the tissues and enhancing
a minute level of oxygen pressure is required in the
oxygenation of the blood in the lungs. This is called the
cells for normal intracellular chemical reactions to
Bohr effect, which can be explained as follows: As the
take place. The reason for this is that the respiratory
blood passes through the tissues, carbon dioxide dif-
enzyme systems of the cell, which are discussed in
fuses from the tissue cells into the blood. This increases
Chapter 67, are geared so that when the cellular Po2 is
the blood Po2, which in turn raises the blood H2CO3
more than 1 mm Hg, oxygen availability is no longer a
(carbonic acid) and the hydrogen ion concentration.
limiting factor in the rates of the chemical reactions.
These effects shift the oxygen-hemoglobin dissocia-
Instead, the main limiting factor is the concentration
tion curve to the right and downward, as shown in
of adenosine diphosphate (ADP) in the cells. This
Figure 40-10, forcing oxygen away from the hemoglo-
effect is demonstrated in Figure 40-11, which shows
bin and therefore delivering increased amounts of
the relation between intracellular Po2 and the rate of
oxygen to the tissues.
oxygen usage at different concentrations of ADP. Note
Exactly the opposite effects occur in the lungs,
that whenever the intracellular Po2 is above 1 mm Hg,
where carbon dioxide diffuses from the blood into
the rate of oxygen usage becomes constant for any
the alveoli. This reduces the blood Pco2 and decreases
given concentration of ADP in the cell. Conversely,
the hydrogen ion concentration, shifting the
when the ADP concentration is altered, the rate of
Chapter 40
Transport of Oxygen and Carbon Dioxide in Blood and Tissue Fluids
509
oxygen that can be transported to the tissue in each
100 milliliters of blood and (2) the rate of blood flow.
If the rate of blood flow falls to zero, the amount of
ADP = 11/2 normal
available oxygen also falls to zero. Thus, there are
1.5
times when the rate of blood flow through a tissue
can be so low that tissue Po2 falls below the critical
1 mm Hg required for intracellular metabolism. Under
ADP = Normal resting level
these conditions, the rate of tissue usage of oxygen
1.0
is blood flow limited. Neither diffusion-limited nor
blood flow-limited oxygen states can continue for
long, because the cells receive less oxygen than is
required to continue the life of the cells.
ADP =
1/2 normal
0.5
Transport of Oxygen in the
Dissolved State
At the normal arterial Po2 of 95 mm Hg, about 0.29 mil-
0
liliter of oxygen is dissolved in every 100 milliliters of
0
1
2
3
4
water in the blood, and when the Po2 of the blood falls
Intracellular PO2 (mm Hg)
to the normal 40 mm Hg in the tissue capillaries, only
0.12 milliliter of oxygen remains dissolved. In other
words, 0.17 milliliter of oxygen is normally transported
in the dissolved state to the tissues by each 100 milli-
liters of arterial blood flow. This compares with almost
Figure 40-11
5 milliliters of oxygen transported by the red cell hemo-
Effect of intracellular adenosine diphosphate (ADP) and PO2 on
globin. Therefore, the amount of oxygen transported to
rate of oxygen usage by the cells. Note that as long as the intra-
the tissues in the dissolved state is normally slight, only
cellular PO2
remains above 1 mm Hg, the controlling factor for the
about 3 per cent of the total, as compared with 97 per
rate of oxygen usage is the intracellular concentration of ADP.
cent transported by the hemoglobin.
During strenuous exercise, when hemoglobin release
of oxygen to the tissues increases another threefold, the
oxygen usage changes in proportion to the change in
relative quantity of oxygen transported in the dissolved
ADP concentration.
state falls to as little as 1.5 per cent. But if a person
As explained in Chapter 3, when adenosine triphos-
breathes oxygen at very high alveolar Po2 levels, the
amount transported in the dissolved state can become
phate (ATP) is used in the cells to provide energy, it is
much greater, sometimes so much so that a serious
converted into ADP. The increasing concentration
excess of oxygen occurs in the tissues, and “oxygen poi-
of ADP increases the metabolic usage of oxygen as
soning” ensues. This often leads to brain convulsions
it combines with the various cell nutrients, releasing
and even death, as discussed in detail in Chapter 44
energy that reconverts the ADP back to ATP. Under
in relation to the high-pressure breathing of oxygen
normal operating conditions, the rate of oxygen usage
among deep-sea divers.
by the cells is controlled ultimately by the rate of energy
expenditure within the cells—that is, by the rate at which
ADP is formed from ATP.
Combination of Hemoglobin with
Carbon Monoxide—Displacement
Effect of Diffusion Distance from the Capillary to the Cell on
of Oxygen
Oxygen Usage. Tissue cells are seldom more than 50
Carbon monoxide combines with hemoglobin at the
micrometers away from a capillary, and oxygen nor-
same point on the hemoglobin molecule as does oxygen;
mally can diffuse readily enough from the capillary to
it can therefore displace oxygen from the hemoglobin,
the cell to supply all the required amounts of oxygen
thereby decreasing the oxygen carrying capacity of
for metabolism. However, occasionally, cells are
blood. Further, it binds with about 250 times as much
located farther from the capillaries, and the rate of
tenacity as oxygen, which is demonstrated by the carbon
oxygen diffusion to these cells can become so low that
monoxide-hemoglobin dissociation curve in Figure
intracellular Po2 falls below the critical level required
40-12. This curve is almost identical to the oxygen-
to maintain maximal intracellular metabolism. Thus,
hemoglobin dissociation curve, except that the carbon
under these conditions, oxygen usage by the cells is
monoxide partial pressures, shown on the abscissa, are
at a level 1/250 of those for the oxygen-hemoglobin dis-
said to be diffusion limited and is no longer deter-
sociation curve of Figure 40-8. Therefore, a carbon
mined by the amount of ADP formed in the cells.
monoxide partial pressure of only 0.4 mm Hg in the
But this almost never occurs, except in pathological
alveoli,1/250 that of normal alveolar oxygen (100 mm Hg
states.
Po2), allows the carbon monoxide to compete equally
with the oxygen for combination with the hemoglobin
Effect of Blood Flow on Metabolic Use of Oxygen. The total
and causes half the hemoglobin in the blood to become
amount of oxygen available each minute for use in
bound with carbon monoxide instead of with oxygen.
any given tissue is determined by (1) the quantity of
Therefore, a carbon monoxide pressure of only
510
Unit VII
Respiration
Capillary
Interstitial
Red blood cell
100
fluid
90
Cell
2
Hgb • CO
80
Carbonic
Hgb
70
anhydrase
+
H2CO3
H2O +
CO2
CO2
CO2
60
HCO3- +
H+
50
+
H2O
Hgb
40
CO2
Cl
30
H2O
HHgb
CO2 transported as:
20
Cl
-
1. CO2
=
7%
HCO3
10
2. Hgb • CO2
= 23%
3. HCO3
= 70%
Plasma
0
0
0.1
0.2
0.3
0.4
Gas pressure of carbon monoxide (mm Hg)
Figure 40-13
Transport of carbon dioxide in the blood.
Figure 40-12
dioxide in the blood has a lot to do with the acid-base
Carbon monoxide-hemoglobin dissociation curve. Note the
balance of the body fluids, which is discussed in
extremely low carbon monoxide pressures at which carbon
Chapter
30. Under normal resting conditions, an
monoxide combines with hemoglobin.
average of 4 milliliters of carbon dioxide is transported
from the tissues to the lungs in each 100 milliliters of
0.6 mm Hg (a volume concentration of less than one
blood.
part per thousand in air) can be lethal.
Even though the oxygen content of blood is greatly
reduced in carbon monoxide poisoning, the Po2 of the
Chemical Forms in Which Carbon
blood may be normal. This makes exposure to carbon
Dioxide Is Transported
monoxide especially dangerous, because the blood is
bright red and there are no obvious signs of hypoxemia,
To begin the process of carbon dioxide transport,
such as a bluish color of the fingertips or lips (cyanosis).
carbon dioxide diffuses out of the tissue cells in the
Also, Po2 is not reduced, and the feedback mechanism
dissolved molecular carbon dioxide form. On entering
that usually stimulates increased respiration rate in
response to lack of oxygen (usually reflected by a low
the tissue capillaries, the carbon dioxide initiates a host
Po2) is absent. Because the brain is one of the first
of almost instantaneous physical and chemical reac-
organs affected by lack of oxygen, the person may
tions, shown in Figure 40-13, which are essential for
become disoriented and unconscious before becoming
carbon dioxide transport.
aware of the danger.
A patient severely poisoned with carbon monoxide
Transport of Carbon Dioxide in the Dissolved State. A small
can be treated by administering pure oxygen, because
portion of the carbon dioxide is transported in the dis-
oxygen at high alveolar pressure can displace carbon
solved state to the lungs. Recall that the Pco2 of venous
monoxide rapidly from its combination with hemoglo-
blood is
45 mm Hg and that of arterial blood is
bin. The patient can also benefit from simultaneous
40 mm Hg. The amount of carbon dioxide dissolved in
administration of 5 per cent carbon dioxide, because
this strongly stimulates the respiratory center, which
the fluid of the blood at 45 mm Hg is about 2.7 ml/dl
increases alveolar ventilation and reduces the alveolar
(2.7 volumes per cent). The amount dissolved at
carbon monoxide. With intensive oxygen and carbon
40 mm Hg is about 2.4 milliliters, or a difference of 0.3
dioxide therapy, carbon monoxide can be removed from
milliliter. Therefore, only about 0.3 milliliter of carbon
the blood as much as 10 times as rapidly as without
dioxide is transported in the dissolved form by each
therapy.
100 milliliters of blood flow. This is about 7 per cent of
all the carbon dioxide normally transported.
Transport of Carbon Dioxide
Transport of Carbon Dioxide in the Form of Bicarbonate Ion
in the Blood
Reaction of Carbon Dioxide with Water in the Red
Blood Cells—Effect of Carbonic Anhydrase. The dis-
Transport of carbon dioxide by the blood is not nearly
solved carbon dioxide in the blood reacts with water
as problematical as transport of oxygen is, because
to form carbonic acid. This reaction would occur
even in the most abnormal conditions, carbon dioxide
much too slowly to be of importance were it not for
can usually be transported in far greater quantities
the fact that inside the red blood cells is a protein
than oxygen can be. However, the amount of carbon
enzyme called carbonic anhydrase, which catalyzes the
Chapter 40
Transport of Oxygen and Carbon Dioxide in Blood and Tissue Fluids
511
reaction between carbon dioxide and water and accel-
dioxide with water inside the red blood cells, it is
erates its reaction rate about 5000-fold. Therefore,
doubtful that under normal conditions this carbamino
instead of requiring many seconds or minutes to occur,
mechanism transports more than 20 per cent of the
as is true in the plasma, the reaction occurs so rapidly
total carbon dioxide.
in the red blood cells that it reaches almost complete
equilibrium within a very small fraction of a second.
This allows tremendous amounts of carbon dioxide to
Carbon Dioxide Dissociation Curve
react with the red blood cell water even before the
blood leaves the tissue capillaries.
The curve shown in Figure 40-14—called the carbon
dioxide dissociation curve—depicts the dependence
Dissociation of Carbonic Acid into Bicarbonate and
of total blood carbon dioxide in all its forms on Pco2.
Hydrogen Ions. In another fraction of a second, the
Note that the normal blood Pco2 ranges between the
carbonic acid formed in the red cells (H2CO3) disso-
limits of 40 mm Hg in arterial blood and 45 mm Hg in
ciates into hydrogen and bicarbonate ions (H+ and
venous blood, which is a very narrow range. Note also
HCO3-). Most of the hydrogen ions then combine with
that the normal concentration of carbon dioxide in the
the hemoglobin in the red blood cells, because the
blood in all its different forms is about 50 volumes per
hemoglobin protein is a powerful acid-base buffer. In
cent, but only 4 volumes per cent of this is exchanged
turn, many of the bicarbonate ions diffuse from the red
during normal transport of carbon dioxide from the
cells into the plasma, while chloride ions diffuse into
tissues to the lungs. That is, the concentration rises to
the red cells to take their place. This is made possible
about 52 volumes per cent as the blood passes through
by the presence of a special bicarbonate-chloride
the tissues and falls to about 48 volumes per cent as it
carrier protein in the red cell membrane that shuttles
passes through the lungs.
these two ions in opposite directions at rapid veloci-
ties. Thus, the chloride content of venous red blood
cells is greater than that of arterial red cells, a phe-
When Oxygen Binds with
nomenon called the chloride shift.
Hemoglobin, Carbon Dioxide Is
The reversible combination of carbon dioxide with
Released (the Haldane Effect)
water in the red blood cells under the influence of car-
to Increase CO2 Transport
bonic anhydrase accounts for about 70 per cent of the
carbon dioxide transported from the tissues to the
Earlier in the chapter, it was pointed out that an
lungs. Thus, this means of transporting carbon dioxide
increase in carbon dioxide in the blood causes oxygen
is by far the most important. Indeed, when a carbonic
to be displaced from the hemoglobin (the Bohr effect),
anhydrase inhibitor (acetazolamide) is administered
which is an important factor in increasing oxygen
to an animal to block the action of carbonic anhydrase
transport. The reverse is also true: binding of oxygen
in the red blood cells, carbon dioxide transport from
with hemoglobin tends to displace carbon dioxide
the tissues becomes so poor that the tissue Pco2 can be
from the blood. Indeed, this effect, called the Haldane
made to rise to 80 mm Hg instead of the normal
effect, is quantitatively far more important in
45 mm Hg.
Transport of Carbon Dioxide in Combination with Hemoglobin
and Plasma Proteins—Carbaminohemoglobin. In addition to
reacting with water, carbon dioxide reacts directly with
amine radicals of the hemoglobin molecule to form
the compound carbaminohemoglobin (CO2Hgb). This
80
combination of carbon dioxide and hemoglobin is a
reversible reaction that occurs with a loose bond, so
70
that the carbon dioxide is easily released into the
60
alveoli, where the Pco2 is lower than in the pulmonary
50
capillaries.
40
A small amount of carbon dioxide also reacts in the
same way with the plasma proteins in the tissue capil-
30
laries. This is much less significant for the transport of
20
carbon dioxide because the quantity of these proteins
10
in the blood is only one fourth as great as the quantity
0
of hemoglobin.
0
10 20 30
40
50 60
70
80
90
100
110
120
The quantity of carbon dioxide that can be carried
Gas pressure of carbon dioxide (mm Hg)
from the peripheral tissues to the lungs by carbamino
combination with hemoglobin and plasma proteins is
about 30 per cent of the total quantity transported—
that is, normally about 1.5 milliliters of carbon dioxide
Figure 40-14
in each 100 milliliters of blood. However, because this
reaction is much slower than the reaction of carbon
Carbon dioxide dissociation curve.
512
Unit VII
Respiration
falls to 48 volumes per cent (point B). This represents
an additional
2 volumes per cent loss of carbon
55
dioxide. Thus, the Haldane effect approximately
doubles the amount of carbon dioxide released from
the blood in the lungs and approximately doubles the
A
pickup of carbon dioxide in the tissues.
PO2 = 40 mm Hg
Change in Blood Acidity During
50
Carbon Dioxide Transport
PO2 = 100 mm Hg
The carbonic acid formed when carbon dioxide enters
B
the blood in the peripheral tissues decreases the blood
pH. However, reaction of this acid with the acid-base
buffers of the blood prevents the hydrogen ion concen-
45
tration from rising greatly (and the pH from falling
35
40
45
50
greatly). Ordinarily, arterial blood has a pH of about
PCO2
7.41, and as the blood acquires carbon dioxide in the
tissue capillaries, the pH falls to a venous value of about
7.37. In other words, a pH change of 0.04 unit takes
place. The reverse occurs when carbon dioxide is
Figure 40-15
released from the blood in the lungs, with the pH rising
to the arterial value of 7.41 once again. In heavy exer-
Portions of the carbon dioxide dissociation curve when the PO2 is
cise or other conditions of high metabolic activity, or
100 mm Hg or 40 mm Hg. The arrow represents the Haldane
when blood flow through the tissues is sluggish, the
effect on the transport of carbon dioxide, as discussed in the text.
decrease in pH in the tissue blood (and in the tissues
themselves) can be as much as 0.50, about 12 times
normal, thus causing significant tissue acidosis.
promoting carbon dioxide transport than is the Bohr
effect in promoting oxygen transport.
Respiratory Exchange Ratio
The Haldane effect results from the simple fact that
The discerning student will have noted that normal
the combination of oxygen with hemoglobin in the
transport of oxygen from the lungs to the tissues by each
lungs causes the hemoglobin to become a stronger
100 milliliters of blood is about 5 milliliters, whereas
acid. This displaces carbon dioxide from the blood and
normal transport of carbon dioxide from the tissues
into the alveoli in two ways: (1) The more highly acidic
to the lungs is about 4 milliliters. Thus, under normal
hemoglobin has less tendency to combine with carbon
resting conditions, only about
82 per cent as much
dioxide to form carbaminohemoglobin, thus displacing
carbon dioxide is expired from the lungs as oxygen is
much of the carbon dioxide that is present in the car-
taken up by the lungs. The ratio of carbon dioxide
bamino form from the blood. (2) The increased acidity
output to oxygen uptake is called the respiratory
of the hemoglobin also causes it to release an excess
exchange ratio (R). That is,
of hydrogen ions, and these bind with bicarbonate
ions to form carbonic acid; this then dissociates into
R= Rate of carbon dioxide output
Rate of oxygen uptake
water and carbon dioxide, and the carbon dioxide is
released from the blood into the alveoli and, finally,
The value for R changes under different metabolic
into the air.
conditions. When a person is using exclusively carbohy-
drates for body metabolism, R rises to 1.00. Conversely,
Figure 40-15 demonstrates quantitatively the signif-
when a person is using exclusively fats for metabolic
icance of the Haldane effect on the transport of carbon
energy, the R level falls to as low as 0.7. The reason
dioxide from the tissues to the lungs. This figure shows
for this difference is that when oxygen is metabolized
small portions of two carbon dioxide dissociation
with carbohydrates, one molecule of carbon dioxide is
curves: (1) when the Po2 is 100 mm Hg, which is the
formed for each molecule of oxygen consumed; when
case in the blood capillaries of the lungs, and (2) when
oxygen reacts with fats, a large share of the oxygen
the Po2 is 40 mm Hg, which is the case in the tissue cap-
combines with hydrogen atoms from the fats to form
illaries. Point A shows that the normal Pco2
of
water instead of carbon dioxide. In other words, when
45 mm Hg in the tissues causes 52 volumes per cent of
fats are metabolized, the respiratory quotient of the
carbon dioxide to combine with the blood. On enter-
chemical reactions in the tissues is about 0.70 instead of
1.00. (The tissue respiratory quotient is discussed in
ing the lungs, the Pco2 falls to 40 mm Hg and the Po2
Chapter 71.) For a person on a normal diet consuming
rises to 100 mm Hg. If the carbon dioxide dissociation
average amounts of carbohydrates, fats, and proteins,
curve did not shift because of the Haldane effect, the
the average value for R is considered to be 0.825.
carbon dioxide content of the blood would fall only to
50 volumes per cent, which would be a loss of only 2
volumes per cent of carbon dioxide. However, the
References
increase in Po2 in the lungs lowers the carbon dioxide
dissociation curve from the top curve to the lower
Albert R, Spiro S, Jett J: Comprehensive Respiratory Medi-
curve of the figure, so that the carbon dioxide content
cine. Philadelphia: Mosby, 2002.
Chapter 40
Transport of Oxygen and Carbon Dioxide in Blood and Tissue Fluids
513
Dempsey JA, Wagner PD: Exercise-induced arterial hypox-
Richardson RS: Oxygen transport and utilization: an inte-
emia. J Appl Physiol 87:1997, 1999.
gration of the muscle systems. Adv Physiol Educ 27:183,
Geers C, Gros G: Carbon dioxide transport and carbonic
2003.
anhydrase in blood and muscle. Physiol Rev
80:681,
Roy TK, Popel AS: Theoretical predictions of end-capillary
2000.
Po2 in muscles of athletic and nonathletic animals at
Henry RP, Swenson ER: The distribution and physiological
Vo2max. Am J Physiol 271:H721, 1996.
significance of carbonic anhydrase in vertebrate gas
Spahn DR, Pasch T: Physiological properties of blood sub-
exchange organs. Respir Physiol 121:1, 2000.
stitutes. News Physiol Sci 16:38, 2001.
Jones AM, Koppo K, Burnley M: Effects of prior exercise on
Tsai AG, Johnson PC, Intaglietta M: Oxygen gradients in the
metabolic and gas exchange responses to exercise. Sports
microcirculation. Physiol Rev 83:933, 2003.
Med 33:949, 2003.
Wagner PD: Diffusive resistance to O2 transport in muscle.
Nikinmaa M: Membrane transport and control of
Acta Physiol Scand 168:609, 2000.
hemoglobin-oxygen affinity in nucleated erythrocytes.
West JB: Pulmonary Physiology and Pathophysiology: An
Physiol Rev 72:301, 1992.
Integrated, Case-Based Approach. Philadelphia: Lippin-
Piiper J: Perfusion, diffusion and their heterogeneities limit-
cott Williams & Wilkins, 2001.
ing blood-tissue O2 transfer in muscle. Acta Physiol Scand
West JB: Pulmonary Physiology—The Essentials. Baltimore:
168:603, 2000.
Lippincott Williams & Wilkins, 2003.
C H A P T E R
4
1
Regulation of Respiration
The nervous system normally adjusts the rate of
alveolar ventilation almost exactly to the demands
of the body so that the oxygen pressure (Po2) and
carbon dioxide pressure (Pco2) in the arterial blood
are hardly altered even during heavy exercise and
most other types of respiratory stress. This chapter
describes the function of this neurogenic system for
regulation of respiration.
Respiratory Center
The respiratory center is composed of several groups of neurons located bilat-
erally in the medulla oblongata and pons of the brain stem, as shown in Figure
41-1. It is divided into three major collections of neurons: (1) a dorsal respira-
tory group, located in the dorsal portion of the medulla, which mainly causes
inspiration; (2) a ventral respiratory group, located in the ventrolateral part of
the medulla, which mainly causes expiration; and (3) the pneumotaxic center,
located dorsally in the superior portion of the pons, which mainly controls rate
and depth of breathing. The dorsal respiratory group of neurons plays the most
fundamental role in the control of respiration. Therefore, let us discuss its func-
tion first.
Dorsal Respiratory Group of Neurons—Its Control
of Inspiration and of Respiratory Rhythm
The dorsal respiratory group of neurons extends most of the length of the
medulla. Most of its neurons are located within the nucleus of the tractus soli-
tarius, although additional neurons in the adjacent reticular substance of the
medulla also play important roles in respiratory control. The nucleus of the
tractus solitarius is the sensory termination of both the vagal and the glos-
sopharyngeal nerves, which transmit sensory signals into the respiratory center
from (1) peripheral chemoreceptors, (2) baroreceptors, and (3) several types of
receptors in the lungs.
Rhythmical Inspiratory Discharges from the Dorsal Respiratory Group. The basic rhythm
of respiration is generated mainly in the dorsal respiratory group of neurons.
Even when all the peripheral nerves entering the medulla have been sectioned
and the brain stem transected both above and below the medulla, this group of
neurons still emits repetitive bursts of inspiratory neuronal action potentials. The
basic cause of these repetitive discharges is unknown. In primitive animals,
neural networks have been found in which activity of one set of neurons excites
a second set, which in turn inhibits the first. Then, after a period of time, the
mechanism repeats itself, continuing throughout the life of the animal. There-
fore, most respiratory physiologists believe that some similar network of
neurons is present in the human being, located entirely within the medulla; it
probably involves not only the dorsal respiratory group but adjacent areas of
the medulla as well, and is responsible for the basic rhythm of respiration.
Inspiratory “Ramp” Signal. The nervous signal that is transmitted to the inspira-
tory muscles, mainly the diaphragm, is not an instantaneous burst of action
514
Chapter 41
Regulation of Respiration
515
or more seconds, thus filling the lungs with a great
excess of air.
The function of the pneumotaxic center is primarily
to limit inspiration. This has a secondary effect of
Pneumotaxic center
increasing the rate of breathing, because limitation
of inspiration also shortens expiration and the entire
Inhibits
Fourth ventricle
period of each respiration. A strong pneumotaxic
signal can increase the rate of breathing to 30 to 40
? Apneustic center
breaths per minute, whereas a weak pneumotaxic
Dorsal respiratory
signal may reduce the rate to only 3 to 5 breaths per
Ventral respiratory
group (inspiration)
group (expiration
minute.
and inspiration)
Ventral Respiratory Group of
Vagus and
glossopharyngeal
Respiratory motor
Neurons—Functions in Both
pathways
Inspiration and Expiration
Figure 41-1
Located in each side of the medulla, about 5 millime-
Organization of the respiratory center.
ters anterior and lateral to the dorsal respiratory group
of neurons, is the ventral respiratory group of neurons,
found in the nucleus ambiguus rostrally and the
nucleus retroambiguus caudally. The function of this
potentials. Instead, in normal respiration, it begins
neuronal group differs from that of the dorsal respi-
weakly and increases steadily in a ramp manner for
ratory group in several important ways:
about 2 seconds. Then it ceases abruptly for approxi-
1.
The neurons of the ventral respiratory group
mately the next 3 seconds, which turns off the excita-
remain almost totally inactive during normal quiet
tion of the diaphragm and allows elastic recoil of the
respiration. Therefore, normal quiet breathing is
lungs and the chest wall to cause expiration. Next, the
caused only by repetitive inspiratory signals from
inspiratory signal begins again for another cycle; this
the dorsal respiratory group transmitted mainly to
cycle repeats again and again, with expiration occur-
the diaphragm, and expiration results from elastic
ring in between. Thus, the inspiratory signal is a ramp
recoil of the lungs and thoracic cage.
signal. The obvious advantage of the ramp is that it
2.
There is no evidence that the ventral respiratory
causes a steady increase in the volume of the lungs
neurons participate in the basic rhythmical
during inspiration, rather than inspiratory gasps.
oscillation that controls respiration.
There are two qualities of the inspiratory ramp that
3.
When the respiratory drive for increased
are controlled, as follows:
pulmonary ventilation becomes greater than
1. Control of the rate of increase of the ramp signal,
normal, respiratory signals spill over into the
so that during heavy respiration, the ramp
ventral respiratory neurons from the basic
increases rapidly and therefore fills the lungs
oscillating mechanism of the dorsal respiratory
rapidly.
area. As a consequence, the ventral respiratory
2. Control of the limiting point at which the ramp
area contributes extra respiratory drive as well.
suddenly ceases. This is the usual method for
4.
Electrical stimulation of a few of the neurons in
controlling the rate of respiration; that is, the
the ventral group causes inspiration, whereas
earlier the ramp ceases, the shorter the duration
stimulation of others causes expiration. Therefore,
of inspiration. This also shortens the duration of
these neurons contribute to both inspiration and
expiration. Thus, the frequency of respiration is
expiration. They are especially important in
increased.
providing the powerful expiratory signals to the
abdominal muscles during very heavy expiration.
A Pneumotaxic Center Limits the
Thus, this area operates more or less as an
overdrive mechanism when high levels of
Duration of Inspiration and Increases
pulmonary ventilation are required, especially
the Respiratory Rate
during heavy exercise.
A pneumotaxic center, located dorsally in the nucleus
parabrachialis of the upper pons, transmits signals to
Lung Inflation Signals Limit
the inspiratory area. The primary effect of this center
Inspiration—The Hering-Breuer
is to control the “switch-off” point of the inspiratory
ramp, thus controlling the duration of the filling phase
Inflation Reflex
of the lung cycle. When the pneumotaxic signal is
strong, inspiration might last for as little as 0.5 second,
In addition to the central nervous system respiratory
thus filling the lungs only slightly; when the pneumo-
control mechanisms operating entirely within the
taxic signal is weak, inspiration might continue for 5
brain stem, sensory nerve signals from the lungs also
516
Unit VII
Respiration
help control respiration. Most important, located in
Direct Chemical Control of
the muscular portions of the walls of the bronchi and
Respiratory Center Activity by Carbon
bronchioles throughout the lungs are stretch receptors
Dioxide and Hydrogen Ions
that transmit signals through the vagi into the dorsal
respiratory group of neurons when the lungs become
Chemosensitive Area of the Respiratory Center. We have dis-
overstretched. These signals affect inspiration in much
cussed mainly three areas of the respiratory center:
the same way as signals from the pneumotaxic center;
the dorsal respiratory group of neurons, the ventral
that is, when the lungs become overly inflated, the
respiratory group, and the pneumotaxic center. It
stretch receptors activate an appropriate feedback
is believed that none of these is affected directly by
response that “switches off” the inspiratory ramp and
changes in blood carbon dioxide concentration or
thus stops further inspiration. This is called the Hering-
hydrogen ion concentration. Instead, an additional
Breuer inflation reflex. This reflex also increases the
neuronal area, a chemosensitive area, shown in Figure
rate of respiration, as is true for signals from the pneu-
41-2, is located bilaterally, lying only 0.2 millimeter
motaxic center.
beneath the ventral surface of the medulla. This area
In human beings, the Hering-Breuer reflex probably
is highly sensitive to changes in either blood Pco2 or
is not activated until the tidal volume increases to
hydrogen ion concentration, and it in turn excites the
more than three times normal (greater than about 1.5
other portions of the respiratory center.
liters per breath). Therefore, this reflex appears to be
mainly a protective mechanism for preventing excess
Excitation of the Chemosensitive Neurons by
lung inflation rather than an important ingredient in
Hydrogen Ions Is Likely the Primary Stimulus
normal control of ventilation.
The sensor neurons in the chemosensitive area are
especially excited by hydrogen ions; in fact, it is
believed that hydrogen ions may be the only impor-
Control of Overall Respiratory
tant direct stimulus for these neurons. However,
Center Activity
hydrogen ions do not easily cross the blood-brain
barrier. For this reason, changes in hydrogen ion con-
Up to this point, we have discussed the basic mecha-
centration in the blood have considerably less effect
nisms for causing inspiration and expiration, but it is
in stimulating the chemosensitive neurons than do
also important to know how the intensity of the respi-
changes in blood carbon dioxide, even though carbon
ratory control signals is increased or decreased to
dioxide is believed to stimulate these neurons second-
match the ventilatory needs of the body. For example,
arily by changing the hydrogen ion concentration, as
during heavy exercise, the rates of oxygen usage and
explained in the following section.
carbon dioxide formation are often increased to as
much as 20 times normal, requiring commensurate
Carbon Dioxide Stimulates the
increases in pulmonary ventilation. The major purpose
Chemosensitive Area
of the remainder of this chapter is to discuss this
Although carbon dioxide has little direct effect in
control of ventilation in accord with the respiratory
stimulating the neurons in the chemosensitive area, it
needs of the body.
Chemical Control
of Respiration
Chemosensitive
The ultimate goal of respiration is to maintain proper
area
concentrations of oxygen, carbon dioxide, and hydro-
gen ions in the tissues. It is fortunate, therefore, that
respiratory activity is highly responsive to changes in
each of these.
Inspiratory area
H+
+ HCO3
-
Excess carbon dioxide or excess hydrogen ions in
the blood mainly act directly on the respiratory center
itself, causing greatly increased strength of both the
H2CO3
inspiratory and the expiratory motor signals to the
respiratory muscles.
Oxygen, in contrast, does not have a significant
CO2 + H2O
direct effect on the respiratory center of the brain in
controlling respiration. Instead, it acts almost entirely
Figure 41-2
on peripheral chemoreceptors located in the carotid
and aortic bodies, and these in turn transmit appropri-
Stimulation of the brain stem inspiratory area by signals from the
chemosensitive area located bilaterally in the medulla, lying only
ate nervous signals to the respiratory center for
a fraction of a millimeter beneath the ventral medullary surface.
control of respiration.
Note also that hydrogen ions stimulate the chemosensitive area,
Let us discuss first the stimulation of the respiratory
but carbon dioxide in the fluid gives rise to most of the hydrogen
center itself by carbon dioxide and hydrogen ions.
ions.
Chapter 41
Regulation of Respiration
517
does have a potent indirect effect. It does this by react-
ing with the water of the tissues to form carbonic acid,
which dissociates into hydrogen and bicarbonate ions;
11
the hydrogen ions then have a potent direct stimula-
tory effect on respiration. These reactions are shown
10
in Figure 41-2.
9
Why does blood carbon dioxide have a more potent
effect in stimulating the chemosensitive neurons than
8
do blood hydrogen ions? The answer is that the blood-
brain barrier is not very permeable to hydrogen ions,
7
but carbon dioxide passes through this barrier almost
6
as if the barrier did not exist. Consequently, whenever
the blood Pco2 increases, so does the Pco2 of both the
5
interstitial fluid of the medulla and the cerebrospinal
fluid. In both these fluids, the carbon dioxide immedi-
4
ately reacts with the water to form new hydrogen ions.
3
Thus, paradoxically, more hydrogen ions are released
into the respiratory chemosensitive sensory area of the
2
medulla when the blood carbon dioxide concentration
PCO2
increases than when the blood hydrogen ion concen-
1
pH
tration increases. For this reason, respiratory center
0
activity is increased very strongly by changes in blood
20
30
40
50
60
70
80
90
100
carbon dioxide, a fact that we subsequently discuss
PCO2 (mm Hg)
quantitatively.
7.6
7.5
7.4
7.3
7.2
7.1
7.0
6.9
pH
Decreased Stimulatory Effect of Carbon Dioxide After the First
1 to 2 Days. Excitation of the respiratory center by
carbon dioxide is great the first few hours after the
blood carbon dioxide first increases, but then it grad-
Figure 41-3
ually declines over the next 1 to 2 days, decreasing to
about one fifth the initial effect. Part of this decline
Effects of increased arterial blood PCO2 and decreased arterial pH
results from renal readjustment of the hydrogen ion
(increased hydrogen ion concentration) on the rate of alveolar
concentration in the circulating blood back toward
ventilation.
normal after the carbon dioxide first increases the
hydrogen concentration. The kidneys achieve this by
Unimportance of Oxygen for Control of the
increasing the blood bicarbonate, which binds with the
Respiratory Center
hydrogen ions in the blood and cerebrospinal fluid to
Changes in oxygen concentration have virtually no
reduce their concentrations. But even more important,
direct effect on the respiratory center itself to alter
over a period of hours, the bicarbonate ions also
respiratory drive (although oxygen changes do have
slowly diffuse through the blood-brain and blood-
an indirect effect, acting through the peripheral
cerebrospinal fluid barriers and combine directly with
chemoreceptors, as explained in the next section).
the hydrogen ions adjacent to the respiratory neurons
We learned in Chapter 40 that the hemoglobin-
as well, thus reducing the hydrogen ions back to near
oxygen buffer system delivers almost exactly normal
normal. A change in blood carbon dioxide concentra-
amounts of oxygen to the tissues even when the pul-
tion therefore has a potent acute effect on controlling
monary Po2 changes from a value as low as 60 mm Hg
respiratory drive but only a weak chronic effect after
up to a value as high as 1000 mm Hg. Therefore, except
a few days’ adaptation.
under special conditions, adequate delivery of oxygen
can occur despite changes in lung ventilation ranging
Quantitative Effects of Blood PCO2
from slightly below one half normal to as high as 20 or
and Hydrogen Ion Concentration on
more times normal. This is not true for carbon dioxide,
Alveolar Ventilation
because both the blood and tissue Pco2
changes
Figure
41-3 shows quantitatively the approximate
inversely with the rate of pulmonary ventilation; thus,
effects of blood Pco2 and blood pH (which is an
the processes of animal evolution have made carbon
inverse logarithmic measure of hydrogen ion concen-
dioxide the major controller of respiration, not
tration) on alveolar ventilation. Note especially the
oxygen.
very marked increase in ventilation caused by an
Yet, for those special conditions in which the tissues
increase in Pco2 in the normal range between 35 and
get into trouble for lack of oxygen, the body has a
75 mm Hg. This demonstrates the tremendous effect
special mechanism for respiratory control located in
that carbon dioxide changes have in controlling respi-
the peripheral chemoreceptors, outside the brain res-
ration. By contrast, the change in respiration in the
piratory center; this mechanism responds when the
normal blood pH range between 7.3 and 7.5 is less than
blood oxygen falls too low, mainly below a Po2 of
one tenth as great.
70 mm Hg, as explained in the next section.
518
Unit VII
Respiration
Peripheral Chemoreceptor
are exposed at all times to arterial blood, not venous
blood, and their Po2s are arterial Po2s.
System for Control of
Respiratory Activity—Role of
Stimulation of the Chemoreceptors by Decreased Arterial
Oxygen in Respiratory Control
Oxygen. When the oxygen concentration in the arterial
blood falls below normal, the chemoreceptors become
In addition to control of respiratory activity by the res-
strongly stimulated. This is demonstrated in Figure
piratory center itself, still another mechanism is avail-
41-5, which shows the effect of different levels of arte-
able for controlling respiration. This is the peripheral
rial Po2 on the rate of nerve impulse transmission from
chemoreceptor system, shown in Figure 41-4. Special
a carotid body. Note that the impulse rate is particu-
nervous chemical receptors, called chemoreceptors, are
larly sensitive to changes in arterial Po2 in the range of
located in several areas outside the brain. They are
60 down to 30 mm Hg, a range in which hemoglobin
especially important for detecting changes in oxygen
saturation with oxygen decreases rapidly.
in the blood, although they also respond to a lesser
extent to changes in carbon dioxide and hydrogen ion
Effect of Carbon Dioxide and Hydrogen Ion Concentration on
concentrations. The chemoreceptors transmit nervous
Chemoreceptor Activity. An increase in either carbon
signals to the respiratory center in the brain to help
dioxide concentration or hydrogen ion concentration
regulate respiratory activity.
also excites the chemoreceptors and, in this way,
Most of the chemoreceptors are in the carotid
indirectly increases respiratory activity. However, the
bodies. However, a few are also in the aortic bodies,
direct effects of both these factors in the respiratory
shown in the lower part of Figure 41-4, and a very few
center itself are so much more powerful than their
are located elsewhere in association with other arter-
effects mediated through the chemoreceptors (about
ies of the thoracic and abdominal regions.
seven times as powerful) that, for practical purposes,
The carotid bodies are located bilaterally in the
the indirect effects of carbon dioxide and hydrogen
bifurcations of the common carotid arteries. Their
ions through the chemoreceptors do not need to be
afferent nerve fibers pass through Hering’s nerves to
considered. Yet there is one difference between the
the glossopharyngeal nerves and then to the dorsal
peripheral and central effects of carbon dioxide: the
respiratory area of the medulla. The aortic bodies are
stimulation by way of the peripheral chemoreceptors
located along the arch of the aorta; their afferent
occurs as much as five times as rapidly as central stim-
nerve fibers pass through the vagi, also to the dorsal
ulation, so that the peripheral chemoreceptors might
medullary respiratory area.
be especially important in increasing the rapidity of
Each of the chemoreceptor bodies receives its own
response to carbon dioxide at the onset of exercise.
special blood supply through a minute artery directly
from the adjacent arterial trunk. Further, blood flow
Basic Mechanism of Stimulation of the Chemoreceptors by
through these bodies is extreme, 20 times the weight
Oxygen Deficiency. The exact means by which low Po2
of the bodies themselves each minute. Therefore, the
excites the nerve endings in the carotid and aortic
percentage of oxygen removed from the flowing blood
bodies is still unknown. However, these bodies have
is virtually zero. This means that the chemoreceptors
Medulla
Glossopharyngeal nerve
800
Vagus nerve
600
Carotid body
400
200
0
0
100
200
300
400
500
Aortic bodies
Arterial PO2 (mm Hg)
Figure 41-4
Figure 41-5
Respiratory control by peripheral chemoreceptors in the carotid
Effect of arterial PO2 on impulse rate from the carotid body of a
and aortic bodies.
cat.
Chapter 41
Regulation of Respiration
519
multiple highly characteristic glandular-like cells,
period of hours, they breathe much more deeply and
called glomus cells, that synapse directly or indirectly
therefore can withstand far lower atmospheric oxygen
with the nerve endings. Some investigators have sug-
concentrations than when they ascend rapidly. This is
gested that these glomus cells might function as the
called acclimatization.
chemoreceptors and then stimulate the nerve endings.
The reason for acclimatization is that, within 2 to 3
But other studies suggest that the nerve endings them-
days, the respiratory center in the brain stem loses
selves are directly sensitive to the low Po2.
about four fifths of its sensitivity to changes in Pco2
and hydrogen ions. Therefore, the excess ventilatory
Effect of Low Arterial PO2 to Stimulate
blow-off of carbon dioxide that normally would inhibit
Alveolar Ventilation When Arterial Carbon
an increase in respiration fails to occur, and low
Dioxide and Hydrogen Ion Concentrations
oxygen can drive the respiratory system to a much
Remain Normal
higher level of alveolar ventilation than under acute
Figure 41-6 shows the effect of low arterial Po2 on
conditions. Instead of the 70 per cent increase in ven-
alveolar ventilation when the Pco2 and the hydrogen
tilation that might occur after acute exposure to low
ion concentration are kept constant at their normal
oxygen, the alveolar ventilation often increases 400
levels. In other words, in this figure, only the ventila-
to 500 per cent after 2 to 3 days of low oxygen; this
tory drive due to the effect of low oxygen on the
helps immensely in supplying additional oxygen to the
chemoreceptors is active. The figure shows almost no
mountain climber.
effect on ventilation as long as the arterial Po2 remains
greater than 100 mm Hg. But at pressures lower than
100 mm Hg, ventilation approximately doubles when
Composite Effects of PCO2, pH, and
the arterial Po2 falls to 60 mm Hg and can increase as
PO2 on Alveolar Ventilation
much as fivefold at very low Po2s. Under these condi-
tions, low arterial Po2 obviously drives the ventilatory
Figure 41-7 gives a quick overview of the manner in
process quite strongly.
which the chemical factors Po2, Pco2, and pH—
together—affect alveolar ventilation. To understand
Chronic Breathing of Low Oxygen Stimulates
this diagram, first observe the four red curves.
Respiration Even More—The Phenomenon
These curves were recorded at different levels of
of “Acclimatization”
arterial Po2—40 mm Hg, 50 mm Hg, 60 mm Hg, and
Mountain climbers have found that when they ascend
100 mm Hg. For each of these curves, the Pco2 was
a mountain slowly, over a period of days rather than a
changed from lower to higher levels. Thus, this
7
60
PO2 (mm Hg)
6
40
pH = 7.4
40
pH = 7.3
5040
50 60
100
5
50
4
30
40
60
3
30
100
2
20
20
1
PCO2
Ventilation
10
0
0
160 140 120 100
80
60
40
20
0
0
Arterial PO2 (mm Hg)
0
10
20
30
40
50
60
Alveolar PCO2 (mm Hg)
Figure 41-6
The lower curve demonstrates the effect of different levels of arte-
Figure 41-7
rial PO2 on alveolar ventilation, showing a sixfold increase in ven-
tilation as the PO2 decreases from the normal level of 100 mm Hg
Composite diagram showing the interrelated effects of PCO2, PO2,
to 20 mm Hg. The upper line shows that the arterial PCO2 was kept
and pH on alveolar ventilation. (Drawn from data in Cunningham
at a constant level during the measurements of this study; pH also
DJC, Lloyd BB: The Regulation of Human Respiration. Oxford:
was kept constant.
Blackwell Scientific Publications, 1963.)
520
Unit VII
Respiration
“family” of red curves represents the combined effects
to transmit at the same time collateral impulses into
of alveolar Pco2 and Po2 on ventilation.
the brain stem to excite the respiratory center. This is
Now observe the green curves. The red curves were
analogous to the stimulation of the vasomotor center
measured at a blood pH of 7.4; the green curves were
of the brain stem during exercise that causes a simul-
measured at a pH of 7.3. We now have two families of
taneous increase in arterial pressure.
curves representing the combined effects of Pco2 and
Actually, when a person begins to exercise, a large
Po2 on ventilation at two different pH values. Still
share of the total increase in ventilation begins imme-
other families of curves would be displaced to the right
diately on initiation of the exercise, before any blood
at higher pHs and displaced to the left at lower pHs.
chemicals have had time to change. It is likely that
Thus, using this diagram, one can predict the level of
most of the increase in respiration results from neuro-
alveolar ventilation for most combinations of alveolar
genic signals transmitted directly into the brain stem
Pco2, alveolar Po2, and arterial pH.
respiratory center at the same time that signals go to
the body muscles to cause muscle contraction.
Regulation of Respiration
Interrelation Between Chemical Factors and Nervous: Factors
During Exercise
in the Control of Respiration During Exercise. When a
person exercises, direct nervous signals presumably
In strenuous exercise, oxygen consumption and carbon
stimulate the respiratory center almost the proper
dioxide formation can increase as much as 20-fold. Yet,
amount to supply the extra oxygen required for exer-
as illustrated in Figure 41-8, in the healthy athlete,
cise and to blow off extra carbon dioxide. Occasion-
alveolar ventilation ordinarily increases almost exactly
ally, however, the nervous respiratory control signals
in step with the increased level of oxygen metabolism.
are either too strong or too weak. Then chemical
The arterial Po2, Pco2, and pH remain almost exactly
factors play a significant role in bringing about the
normal.
final adjustment of respiration required to keep the
In trying to analyze what causes the increased ven-
oxygen, carbon dioxide, and hydrogen ion concentra-
tilation during exercise, one is tempted to ascribe this
tions of the body fluids as nearly normal as possible.
to increases in blood carbon dioxide and hydrogen
This is demonstrated in Figure 41-9, which shows in
ions, plus a decrease in blood oxygen. However, this is
the lower curve changes in alveolar ventilation during
questionable, because measurements of arterial Pco2,
a 1-minute period of exercise and in the upper curve
pH, and Po2 show that none of these values changes
changes in arterial Pco2. Note that at the onset of exer-
significantly during exercise, so that none of them
cise, the alveolar ventilation increases instantaneously,
becomes abnormal enough to stimulate respiration.
without an initial increase in arterial Pco2. In fact, this
Therefore, the question must be asked: What causes
intense ventilation during exercise? At least one effect
seems to be predominant. The brain, on transmitting
motor impulses to the exercising muscles, is believed
44
42
40
38
120
36
110
Exercise
18
100
14
80
10
60
6
40
2
20
Moderate
Severe
0
1
2
exercise
exercise
Minutes
0
0
1.0
2.0
3.0
4.0
O2 consumption (L/min)
Figure 41-9
Changes in alveolar ventilation (bottom curve) and arterial PCO2
Figure 41-8
(top curve) during a 1-minute period of exercise and also after ter-
mination of exercise. (Extrapolated to the human being from data
Effect of exercise on oxygen consumption and ventilatory rate.
in dogs in Bainton CR: Effect of speed vs grade and shivering on
(From Gray JS: Pulmonary Ventilation and Its Physiological Regu-
ventilation in dogs during active exercise. J Appl Physiol 33:778,
lation. Springfield, Ill: Charles C Thomas, 1950.)
1972.)
Chapter 41
Regulation of Respiration
521
the rate of carbon dioxide release, thus keeping arte-
rial Pco2 near its normal value. The upper curve of
Figure 41-10 also shows that if, during exercise, the
arterial Pco2 does change from its normal value of
140
40 mm Hg, it has an extra stimulatory effect on venti-
lation at a Pco2 greater than 40 mm Hg and a depres-
120
sant effect at a Pco2 less than 40 mm Hg.
Possibility That the Neurogenic Factor for Control of Ventila-
100
tion During Exercise Is a Learned Response. Many experi-
ments suggest that the brain’s ability to shift the
80
ventilatory response curve during exercise, as shown
in Figure 41-10, is at least partly a learned response.
60
That is, with repeated periods of exercise, the brain
becomes progressively more able to provide the
40
proper signals required to keep the blood Pco2 at its
normal level. Also, there is reason to believe that even
Exercise
20
the cerebral cortex is involved in this learning, because
Resting
experiments that block only the cortex also block the
Normal
learned response.
0
20
30
40
50
60
80
100
Arterial PCO2 (mm Hg)
Other Factors That
Affect Respiration
Voluntary Control of Respiration. Thus far, we have dis-
Figure 41-10
cussed the involuntary system for the control of respi-
ration. However, we all know that for short periods of
Approximate effect of maximum exercise in an athlete to shift the
time, respiration can be controlled voluntarily and that
alveolar PCO2-ventilation response curve to a level much higher
than normal. The shift, believed to be caused by neurogenic
one can hyperventilate or hypoventilate to such an
factors, is almost exactly the right amount to maintain arterial PCO2
extent that serious derangements in Pco2, pH, and Po2
at the normal level of 40 mm Hg both in the resting state and
can occur in the blood.
during heavy exercise.
Effect of Irritant Receptors in the Airways. The epithelium of
the trachea, bronchi, and bronchioles is supplied with
increase in ventilation is usually great enough so that
sensory nerve endings called pulmonary irritant recep-
at first it actually decreases arterial Pco2 below normal,
tors that are stimulated by many incidents. These cause
as shown in the figure. The presumed reason that the
coughing and sneezing, as discussed in Chapter 39. They
ventilation forges ahead of the buildup of blood
may also cause bronchial constriction in such diseases
carbon dioxide is that the brain provides an “anticipa-
as asthma and emphysema.
tory” stimulation of respiration at the onset of exer-
cise, causing extra alveolar ventilation even before it
Function of Lung
“J Receptors.” A few sensory nerve
is needed. However, after about 30 to 40 seconds,
endings have been described in the alveolar walls in
juxtaposition to the pulmonary capillaries—hence the
the amount of carbon dioxide released into the blood
name “J receptors.” They are stimulated especially when
from the active muscles approximately matches the
the pulmonary capillaries become engorged with blood
increased rate of ventilation, and the arterial Pco2
or when pulmonary edema occurs in such conditions as
returns essentially to normal even as the exercise con-
congestive heart failure. Although the functional role of
tinues, as shown toward the end of the 1-minute period
the J receptors is not clear, their excitation may give the
of exercise in the figure.
person a feeling of dyspnea.
Figure 41-10 summarizes the control of respiration
during exercise in still another way, this time more
Effect of Brain Edema. The activity of the respiratory
quantitatively. The lower curve of this figure shows the
center may be depressed or even inactivated by acute
effect of different levels of arterial Pco2 on alveolar
brain edema resulting from brain concussion. For
ventilation when the body is at rest—that is, not exer-
instance, the head might be struck against some solid
cising. The upper curve shows the approximate shift of
object, after which the damaged brain tissues swell,
compressing the cerebral arteries against the cranial
this ventilatory curve caused by neurogenic drive from
vault and thus partially blocking cerebral blood supply.
the respiratory center that occurs during heavy exer-
Occasionally, respiratory depression resulting from
cise. The points indicated on the two curves show the
brain edema can be relieved temporarily by intravenous
arterial Pco2 first in the resting state and then in the
injection of hypertonic solutions such as highly concen-
exercising state. Note in both instances that the Pco2
trated mannitol solution. These solutions osmotically
is at the normal level of 40 mm Hg. In other words, the
remove some of the fluids of the brain, thus relieving
neurogenic factor shifts the curve about 20-fold in the
intracranial pressure and sometimes re-establishing res-
upward direction, so that ventilation almost matches
piration within a few minutes.
522
Unit VII
Respiration
Anesthesia. Perhaps the most prevalent cause of respi-
carbon dioxide and oxygen. Therefore, normally, the
ratory depression and respiratory arrest is overdosage
lungs cannot build up enough extra carbon dioxide or
with anesthetics or narcotics. For instance, sodium pen-
depress the oxygen sufficiently in a few seconds to cause
tobarbital depresses the respiratory center considerably
the next cycle of the periodic breathing. But under two
more than many other anesthetics, such as halothane. At
separate conditions, the damping factors can be over-
one time, morphine was used as an anesthetic, but this
ridden, and Cheyne-Stokes breathing does occur:
drug is now used only as an adjunct to anesthetics
1.
When a long delay occurs for transport of blood
because it greatly depresses the respiratory center while
from the lungs to the brain, changes in carbon
having less ability to anesthetize the cerebral cortex.
dioxide and oxygen in the alveoli can continue
for many more seconds than usual. Under these
conditions, the storage capacities of the alveoli and
Periodic Breathing
pulmonary blood for these gases are exceeded;
then, after a few more seconds, the periodic
An abnormality of respiration called periodic breathing
respiratory drive becomes extreme, and Cheyne-
occurs in a number of disease conditions. The person
Stokes breathing begins. This type of Cheyne-
breathes deeply for a short interval and then breathes
Stokes breathing often occurs in patients with
slightly or not at all for an additional interval, with the
severe cardiac failure because blood flow is slow,
cycle repeating itself over and over. One type of peri-
thus delaying the transport of blood gases from the
odic breathing, Cheyne-Stokes breathing, is character-
lungs to the brain. In fact, in patients with chronic
ized by slowly waxing and waning respiration occur-
heart failure, Cheyne-Stokes breathing can
ring about every 40 to 60 seconds, as illustrated in Figure
sometimes occur on and off for months.
41-11.
2.
A second cause of Cheyne-Stokes breathing is
increased negative feedback gain in the respiratory
Basic Mechanism of Cheyne-Stokes Breathing. The basic
control areas. This means that a change in blood
cause of Cheyne-Stokes breathing is the following:
carbon dioxide or oxygen causes a far greater
When a person overbreathes, thus blowing off too much
change in ventilation than normally. For instance,
carbon dioxide from the pulmonary blood while at the
instead of the normal 2- to 3-fold increase in
same time increasing blood oxygen, it takes several
ventilation that occurs when the Pco2 rises
seconds before the changed pulmonary blood can be
3 mm Hg, the same 3 mm Hg rise might increase
transported to the brain and inhibit the excess ventila-
ventilation 10- to 20-fold. The brain feedback
tion. By this time, the person has already overventilated
tendency for periodic breathing is now strong
for an extra few seconds. Therefore, when the overven-
enough to cause Cheyne-Stokes breathing without
tilated blood finally reaches the brain respiratory
extra blood flow delay between the lungs and
center, the center becomes depressed an excessive
brain. This type of Cheyne-Stokes breathing occurs
amount. Then the opposite cycle begins. That is, carbon
mainly in patients with brain damage. The brain
dioxide increases and oxygen decreases in the alveoli.
damage often turns off the respiratory drive
Again, it takes a few seconds before the brain can
entirely for a few seconds; then an extra intense
respond to these new changes. When the brain does
increase in blood carbon dioxide turns it back on
respond, the person breathes hard once again, and the
with great force. Cheyne-Stokes breathing of this
cycle repeats.
type is frequently a prelude to death from brain
The basic cause of Cheyne-Stokes breathing occurs
malfunction.
in everyone. However, under normal conditions, this
Typical records of changes in pulmonary and respira-
mechanism is highly “damped.” That is, the fluids of the
tory center Pco2 during Cheyne-Stokes breathing are
blood and the respiratory center control areas have
shown in Figure 41-11. Note that the Pco2 of the
large amounts of dissolved and chemically bound
pulmonary blood changes in advance of the Pco2 of
the respiratory neurons. But the depth of respiration
corresponds with the Pco2 in the brain, not with the
Pco2 in the pulmonary blood where the ventilation is
occurring.
Depth of
respiration
Sleep Apnea
The term apnea means absence of spontaneous breath-
PCO2 of
Respiratory
ing. Occasional apneas occur during normal sleep, but
respiratory
center excited
in persons with sleep apnea, the frequency and duration
neurons
are greatly increased, with episodes of apnea lasting for
10 seconds or longer and occurring 300 to 500 times
each night. Sleep apneas can be caused by obstruction
PCO2 of
of the upper airways, especially the pharynx, or by
lung blood
impaired central nervous system respiratory drive.
Obstructive Sleep Apnea Is Caused by Blockage of the Upper
Airway. The muscles of the pharynx normally keep this
Figure 41-11
passage open to allow air to flow into the lungs during
Cheyne-Stokes breathing, showing changing PCO2 in the pul-
inspiration. During sleep, these muscles usually relax,
monary blood (red line) and delayed changes in the PCO2 of the
but the airway passage remains open enough to permit
fluids of the respiratory center (blue line).
adequate airflow. Some individuals have an especially
Chapter 41
Regulation of Respiration
523
narrow passage, and relaxation of these muscles during
stimulatory effects of carbon dioxide and hydrogen ions.
sleep causes the pharynx to completely close so that air
Patients with this disease are extremely sensitive to
cannot flow into the lungs.
even small doses of sedatives or narcotics, which further
In persons with sleep apnea, loud snoring and labored
reduce the responsiveness of the respiratory centers to
breathing occur soon after falling asleep. The snoring
the stimulatory effects of carbon dioxide. Medications
proceeds, often becoming louder, and is then inter-
that stimulate the respiratory centers can sometimes be
rupted by a long silent period during which no breath-
helpful, but ventilation with CPAP at night is usually
ing
(apnea) occurs. These periods of apnea result
necessary.
in significant decreases in Po2 and increases in Pco2,
which greatly stimulate respiration. This, in turn, causes
sudden attempts to breathe, which result in loud snorts
References
and gasps followed by snoring and repeated episodes of
apnea. The periods of apnea and labored breathing are
Dean JB, Ballantyne D, Cardone DL, et al: Role of gap junc-
repeated several hundred times during the night, result-
tions in CO2 chemoreception and respiratory control. Am
ing in fragmented, restless sleep. Therefore, patients
J Physiol Lung Cell Mol Physiol 283:L665, 2002.
with sleep apnea usually have excessive daytime drowsi-
Dutschmann M, Paton JF: Inhibitory synaptic mechanisms
ness as well as other disorders, including increased sym-
regulating upper airway patency. Respir Physiol Neuro-
pathetic activity, high heart rates, pulmonary and
biol 131:57, 2002.
systemic hypertension, and a greatly elevated risk for
Feldman JL, Mitchell GS, Nattie EE: Breathing: rhythmicity,
cardiovascular disease.
plasticity, chemosensitivity. Annu Rev Neurosci 26:239,
Obstructive sleep apnea most commonly occurs in
2003.
older, obese persons in whom there is increased fat dep-
Forster HV: Plasticity in the control of breathing following
osition in the soft tissues of the pharynx or compression
sensory denervation. J Appl Physiol 94:784, 2003.
of the pharynx due to excessive fat masses in the neck.
Hilaire G, Pasaro R: Genesis and control of the respiratory
In a few individuals, sleep apnea may be associated with
rhythm in adult mammals. News Physiol Sci 18:23, 2003.
nasal obstruction, a very large tongue, enlarged tonsils,
Howard RS, Rudd AG, Wolfe CD, Williams AJ: Pathophysi-
or certain shapes of the palate that greatly increase
ological and clinical aspects of breathing after stroke. Post-
resistance to the flow of air to the lungs during inspira-
grad Med J 77:700, 2001.
tion. The most common treatments of obstructive sleep
Jordan D: Central nervous pathways and control of the
apnea include (1) surgery to remove excess fat tissue
airways. Respir Physiol 125:67, 2001.
at the back of the throat (a procedure called uvu-
Kara T, Narkiewicz K, Somers VK: Chemoreflexes—physi-
lopalatopharyngoplasty), to remove enlarged tonsils or
ology and clinical implications. Acta Physiol Scand 177:
adenoids, or to create an opening in the trachea (tra-
377, 2003.
cheostomy) to bypass the obstructed airway during
Morris KF, Baekey DM, Nuding SC, et al: Neural network
sleep, and (2) nasal ventilation with continuous positive
plasticity in respiratory control. J Appl Physiol 94:1242,
airway pressure (CPAP).
2003.
Mortola JP, Frappell PB: Ventilatory responses to changes in
“Central” Sleep Apnea Occurs When the Neural Drive to Respira-
temperature in mammals and other vertebrates. Annu Rev
tory Muscles Is Transiently Abolished. In a few persons
Physiol 62:847, 2000.
with sleep apnea, the central nervous system drive to
Richerson GB: Serotonergic neurons as carbon dioxide
the ventilatory muscles transiently ceases. Disorders
sensors that maintain pH homeostasis. Nat Rev Neurosci
that can cause cessation of the ventilatory drive during
5:449, 2004.
sleep include damage to the central respiratory centers
Semenza GL: O2-regulated gene expression: transcriptional
or abnormalities of the respiratory neuromuscular appa-
control of cardiorespiratory physiology by HIF-1. J Appl
ratus. Patients affected by central sleep apnea may have
Physiol 96:1173, 2004.
decreased ventilation when they are awake, although
Sharp FR, Bernaudin M: HIF1 and oxygen sensing in the
they are fully capable of normal voluntary breathing.
brain. Nat Rev Neurosci 5:437, 2004.
During sleep, their breathing disorders usually worsen,
Taylor EW, Jordan D, Coote JH: Central control of the car-
resulting in more frequent episodes of apnea that
diovascular and respiratory systems and their interactions
decrease Po2 and increase Pco2 until a critical level is
in vertebrates. Physiol Rev 79:855, 1999.
reached that eventually stimulates respiration. These
West JB: Pulmonary Physiology—The Essentials. Baltimore:
transient instabilities of respiration cause restless sleep
Lippincott Williams & Wilkins, 2003.
and clinical features similar to those observed in
Wolk R, Shamsuzzaman AS, Somers VK: Obesity, sleep
obstructive sleep apnea.
apnea, and hypertension. Hypertension 42:1067, 2003.
In most patients, the cause of central sleep apnea is
Young T, Skatrud J, Peppard PE: Risk factors for obstructive
unknown, although instability of the respiratory drive
sleep apnea in adults. JAMA 291:2013, 2004.
can result from strokes or other disorders that make the
Zuperku EJ, McCrimmon DR: Gain modulation of respira-
respiratory centers of the brain less responsive to the
tory neurons. Respir Physiol Neurobiol 131:121, 2002.
C H A P T E R
4
2
Respiratory Insufficiency—
Pathophysiology, Diagnosis,
Oxygen Therapy
Diagnosis and treatment of most respiratory disor-
ders depend heavily on understanding the basic
physiologic principles of respiration and gas
exchange. Some respiratory diseases result from
inadequate ventilation. Others result from abnor-
malities of diffusion through the pulmonary mem-
brane or abnormal blood transport of gases
between the lungs and tissues. Therapy is often
entirely different for these diseases, so it is no longer satisfactory simply to make
a diagnosis of “respiratory insufficiency.”
Useful Methods for Studying Respiratory Abnormalities
In the previous few chapters, we have discussed a number of methods for study-
ing respiratory abnormalities, including measuring vital capacity, tidal air, func-
tional residual capacity, dead space, physiologic shunt, and physiologic dead
space. This array of measurements is only part of the armamentarium of the
clinical pulmonary physiologist. Some other interesting tools are described here.
Study of Blood Gases and Blood pH
Among the most fundamental of all tests of pulmonary performance are deter-
minations of the blood Po2, CO2, and pH. It is often important to make these
measurements rapidly as an aid in determining appropriate therapy for acute
respiratory distress or acute abnormalities of acid-base balance. Several simple
and rapid methods have been developed to make these measurements within
minutes, using no more than a few drops of blood. They are the following.
Determination of Blood pH. Blood pH is measured using a glass pH electrode of
the type used in all chemical laboratories. However, the electrodes used for this
purpose are miniaturized. The voltage generated by the glass electrode is a
direct measure of pH, and this is generally read directly from a voltmeter scale,
or it is recorded on a chart.
Determination of Blood CO2. A glass electrode pH meter can also be used to deter-
mine blood CO2 in the following way: When a weak solution of sodium bicar-
bonate is exposed to carbon dioxide gas, the carbon dioxide dissolves in the
solution until an equilibrium state is established. In this equilibrium state, the
pH of the solution is a function of the carbon dioxide and bicarbonate ion con-
centrations in accordance with the Henderson-Hasselbalch equation that is
explained in Chapter 30; that is,
-
HCO
3
pH
= 6.1 + log
CO
2
When the glass electrode is used to measure CO2 in blood, a miniature glass
electrode is surrounded by a thin plastic membrane. In the space between the
electrode and plastic membrane is a solution of sodium bicarbonate of known
524
Chapter 42
Respiratory Insufficiency—Pathophysiology, Diagnosis, Oxygen Therapy
525
concentration. Blood is then superfused onto the outer
surface of the plastic membrane, allowing carbon
dioxide to diffuse from the blood into the bicarbonate
A
solution. Only a drop or so of blood is required. Next,
the pH is measured by the glass electrode, and the CO2
500
is calculated by use of the above formula.
400
Determination of Blood PO2. The concentration of oxygen
in a fluid can be measured by a technique called
polarography. Electric current is made to flow
300
between a small negative electrode and the solution.
If the voltage of the electrode is more than -0.6 volt
200
different from the voltage of the solution, oxygen will
deposit on the electrode. Furthermore, the rate of
100
Total lung
Residual
current flow through the electrode will be directly pro-
capacity
volume
portional to the concentration of oxygen (and there-
fore to PO2 as well). In practice, a negative platinum
0
6
5
4
3
2
1
0
electrode with a surface area of about 1 square mil-
Lung volume (liters)
limeter is used, and this is separated from the blood
B
by a thin plastic membrane that allows diffusion of
oxygen but not diffusion of proteins or other sub-
stances that will “poison” the electrode.
Figure 42-1
Often all three of the measuring devices for pH,
CO2, and Po2 are built into the same apparatus, and all
A, Collapse of the respiratory passageway during maximum expi-
these measurements can be made within a minute or
ratory effort, an effect that limits expiratory flow rate. B, Effect of
lung volume on the maximum expiratory air flow, showing
so using a single, droplet-size sample of blood. Thus,
decreasing maximum expiratory air flow as the lung volume
changes in the blood gases and pH can be followed
becomes smaller.
almost moment by moment at the bedside.
Therefore, beyond a critical degree of expiratory force,
Measurement of Maximum
a maximum expiratory flow has been reached.
Expiratory Flow
Figure 42-1B shows the effect of different degrees
of lung collapse (and therefore of bronchiolar collapse
In many respiratory diseases, particularly in asthma,
as well) on the maximum expiratory flow. The curve
the resistance to airflow becomes especially great
recorded in this section shows the maximum expira-
during expiration, sometimes causing tremendous dif-
tory flow at all levels of lung volume after a healthy
ficulty in breathing. This has led to the concept called
person first inhales as much air as possible and then
maximum expiratory flow, which can be defined as
expires with maximum expiratory effort until he or she
follows: When a person expires with great force, the
can expire at no greater rate. Note that the person
expiratory airflow reaches a maximum flow beyond
quickly reaches a maximum expiratory airflow of more
which the flow cannot be increased any more even
than 400 L/min. But regardless of how much addi-
with greatly increased additional force. This is the
tional expiratory effort the person exerts, this is still
maximum expiratory flow. The maximum expiratory
the maximum flow rate that he or she can achieve.
flow is much greater when the lungs are filled with a
Note also that as the lung volume becomes smaller,
large volume of air than when they are almost empty.
the maximum expiratory flow rate also becomes less.
These principles can be understood by referring to
The main reason for this is that in the enlarged lung
Figure 42-1.
the bronchi and bronchioles are held open partially by
Figure 42-1A shows the effect of increased pressure
way of elastic pull on their outsides by lung structural
applied to the outsides of the alveoli and air passage-
elements; however, as the lung becomes smaller, these
ways caused by compressing the chest cage. The arrows
structures are relaxed, so that the bronchi and bron-
indicate that the same pressure compresses the
chioles are collapsed more easily by external chest
outsides of both the alveoli and the bronchioles. There-
pressure, thus progressively reducing the maximum
fore, not only does this pressure force air from
expiratory flow rate as well.
the alveoli toward the bronchioles, but it also tends
to collapse the bronchioles at the same time, which
Abnormalities of the Maximum Expiratory Flow-Volume Curve.
will oppose movement of air to the exterior. Once
Figure 42-2 shows the normal maximum expiratory
the bronchioles have almost completely collapsed,
flow-volume curve, along with two additional flow-
further expiratory force can still greatly increase the
volume curves recorded in two types of lung diseases:
alveolar pressure, but it also increases the degree of
constricted lungs and partial airway obstruction. Note
bronchiolar collapse and airway resistance by an equal
that the constricted lungs have both reduced total lung
amount, thus preventing further increase in flow.
capacity (TLC) and reduced residual volume (RV).
526
Unit VII Respiration
500
Normal
NORMAL
Maximum
Airway obstruction
A
400
inspiration
Constricted lungs
4
300
3
FEV1
2
FVC
200
FEV1/FVC%
1
= 80%
100
0
0
1
2
3
4
5
6
7
TLC
RV
0
B
7
6
5
4
3
2
1
0
AIRWAY OBSTRUCTION
4
Lung volume (liters)
3
FEV1
2
FEV
1/FVC%
FVC
Figure 42-2
1
= 47%
0
Effect of two respiratory abnormalities—constricted lungs and
0
1
2
3
4
5
6
7
airway obstruction—-on the maximum expiratory flow-volume
Seconds
curve. TLC, total lung capacity; RV, residual volume.
Furthermore, because the lung cannot expand to a
Figure 42-3
normal maximum volume, even with the greatest pos-
sible expiratory effort, the maximal expiratory flow
Recordings during the forced vital capacity maneuver: A, in a
cannot rise to equal that of the normal curve. Con-
healthy person and B, in a person with partial airway obstruction.
stricted lung diseases include fibrotic diseases of the
(The “zero” on the volume scale is residual volume.)
lung itself, such as tuberculosis and silicosis, and dis-
eases that constrict the chest cage, such as kyphosis,
scoliosis, and fibrotic pleurisy.
spirometer with maximum expiratory effort as rapidly
In diseases with airway obstruction, it is usually
and as completely as possible. The total distance of the
much more difficult to expire than to inspire because
downslope of the lung volume record represents the
the closing tendency of the airways is greatly increased
FVC, as shown in the figure.
by the extra positive pressure required in the chest
Now, study the difference between the two records
to cause expiration. By contrast, the extra negative
(1) for normal lungs and (2) for partial airway obstruc-
pleural pressure that occurs during inspiration actually
tion. The total volume changes of the FVCs are not
“pulls” the airways open at the same time that it
greatly different, indicating only a moderate difference
expands the alveoli. Therefore, air tends to enter the
in basic lung volumes in the two persons. There is,
lung easily but then becomes trapped in the lungs.
however, a major difference in the amounts of air that
Over a period of months or years, this effect increases
these persons can expire each second, especially during
both the TLC and the RV, as shown by the green curve
the first second. Therefore, it is customary to compare
in Figure 42-2. Also, because of the obstruction of the
the recorded forced expiratory volume during the first
airways and because they collapse more easily than
second (FEV1) with the normal. In the normal person
normal airways, the maximum expiratory flow rate is
(see Figure 42-3A), the percentage of the FVC that is
greatly reduced.
expired in the first second divided by the total FVC
The classic disease that causes severe airway
(FEV1/FVC%) is 80 per cent. However, note in Figure
obstruction is asthma. Serious airway obstruction also
42-3B that, with airway obstruction, this value
occurs in some stages of emphysema.
decreased to only
47 per cent. In serious airway
obstruction, as often occurs in acute asthma, this can
decrease to less than 20 per cent.
Forced Expiratory Vital Capacity and
Forced Expiratory Volume
Physiologic Peculiarities
Another exceedingly useful clinical pulmonary test,
of Specific Pulmonary
and one that is also simple, is to make a record on a
spirometer of the forced expiratory vital capacity
Abnormalities
(FVC). Such a record is shown in Figure 42-3A for a
person with normal lungs and in Figure 42-3B for a
Chronic Pulmonary Emphysema
person with partial airway obstruction. In performing
the FVC maneuver, the person first inspires maximally
The term pulmonary emphysema literally means
to the total lung capacity, then exhales into the
excess air in the lungs. However, this term is usually
Chapter 42
Respiratory Insufficiency—Pathophysiology, Diagnosis, Oxygen Therapy
527
used to describe complex obstructive and destructive
process of the lungs caused by many years of smoking.
It results from the following major pathophysiologic
changes in the lungs:
1. Chronic infection, caused by inhaling smoke or
other substances that irritate the bronchi and
bronchioles. The chronic infection seriously
deranges the normal protective mechanisms of the
airways, including partial paralysis of the cilia of
the respiratory epithelium, an effect caused by
nicotine. As a result, mucus cannot be moved
easily out of the passageways. Also, stimulation
of excess mucus secretion occurs, which further
exacerbates the condition. Too, inhibition of the
alveolar macrophages occurs, so that they become
less effective in combating infection.
2. The infection, excess mucus, and inflammatory
edema of the bronchiolar epithelium together
cause chronic obstruction of many of the smaller
airways.
3. The obstruction of the airways makes it especially
difficult to expire, thus causing entrapment of
air in the alveoli and overstretching them. This,
combined with the lung infection, causes marked
destruction of as much as 50 to 80 per cent of the
alveolar walls. Therefore, the final picture of the
emphysematous lung is that shown in Figures
42-4 (top) and 42-5.
The physiologic effects of chronic emphysema are
extremely varied, depending on the severity of the
disease and the relative degrees of bronchiolar ob-
Figure 42-4
struction versus lung parenchymal destruction. Among
the different abnormalities are the following:
Contrast of the emphysematous lung (top figure) with the normal
1.
The bronchiolar obstruction increases airway
lung (bottom figure), showing extensive alveolar destruction in
resistance and results in greatly increased work of
emphysema. (Reproduced with permission of Patricia Delaney
and the Department of Anatomy, The Medical College of
breathing. It is especially difficult for the person
Wisconsin.)
to move air through the bronchioles during
expiration because the compressive force on the
outside of the lung not only compresses the
alveoli but also compresses the bronchioles,
which further increases their resistance during
turn overloads the right side of the heart and
expiration.
frequently causes right-sided heart failure.
2.
The marked loss of alveolar walls greatly
Chronic emphysema usually progresses slowly over
decreases the diffusing capacity of the lung, which
many years. The person develops both hypoxia and
reduces the ability of the lungs to oxygenate the
hypercapnia because of hypoventilation of many
blood and remove carbon dioxide from the blood.
alveoli plus loss of alveolar walls. The net result of
3.
The obstructive process is frequently much worse
all these effects is severe, prolonged, devastating air
in some parts of the lungs than in other parts,
hunger that can last for years until the hypoxia and
so that some portions of the lungs are well
hypercapnia cause death—a high penalty to pay for
ventilated, while other portions are poorly
smoking.
ventilated. This often causes extremely abnormal
v.ent.lation-perfusion ratios, with a very low
Va/Q
in some parts (physiologic shunt), resul.ing
Pneumonia
in poor aeration of the blood, and very high Va/Q
in other parts (physiologic dead space), resulting
The term pneumonia includes any inflammatory con-
in wasted ventilation, both effects occurring in the
dition of the lung in which some or all of the alveoli
same lungs.
are filled with fluid and blood cells, as shown in Figure
4.
Loss of large portions of the alveolar walls also
42-5. A common type of pneumonia is bacterial pneu-
decreases the number of pulmonary capillaries
monia, caused most frequently by pneumococci.
through which blood can pass. As a result, the
This disease begins with infection in the alveoli; the
pulmonary vascular resistance often increases
pulmonary membrane becomes inflamed and highly
markedly, causing pulmonary hypertension. This in
porous so that fluid and even red and white blood cells
528
Unit VII Respiration
Fluid and blood cells
Confluent alveoli
Edema
Normal
Pneumonia
Emphysema
Figure 42-5
Lung alveolar changes in pneumonia and emphysema.
leak out of the blood into the alveoli. Thus, the infected
Pulmonary arterial blood
alveoli become progressively filled with fluid and cells,
60% saturated with O2
and the infection spreads by extension of bacteria or
virus from alveolus to alveolus. Eventually, large areas
of the lungs, sometimes whole lobes or even a whole
lung, become “consolidated,” which means that they
Pneumonia
are filled with fluid and cellular debris.
In pneumonia, the gas exchange functions of the
lungs change in different stages of the disease. In early
stages, the pneumonia process might well be localized
to only one lung, with alveolar ventilation reduced
Left
Right
pulmonary
while blood flow through the lung continues normally.
pulmonary
vein
60%
This results in two major pulmonary abnormalities: (1)
vein
97%
saturated
saturated
reduction in the total available surface area of the res-
piratory membrane and (2) decreased ventilation-
Aorta:
perfusion ratio. Both these effects cause hypoxemia
Blood 1/2 = 97%
(low blood oxygen) and hypercapnia
(high blood
1/2 = 60%
carbon dioxide).
Mean
= 78%
Figure
42-6 shows the effect of the decreased
ventilation-perfusion ratio in pneumonia, showing that
Figure 42-6
the blood passing through the aerated lung becomes
Effect of pneumonia on percentage saturation of oxygen in the
97 per cent saturated with oxygen, whereas that
pulmonary artery, the right and left pulmonary veins, and the aorta.
passing through the unaerated lung is about 60 per
cent saturated. Therefore, the average saturation of the
blood pumped by the left heart into the aorta is only
about 78 per cent, which is far below normal.
is pliable enough, this will lead simply to collapse of
the alveoli. However, if the lung is rigid because of
Atelectasis
fibrotic tissue and cannot collapse, absorption of air
from the alveoli creates very negative pressures within
Atelectasis means collapse of the alveoli. It can occur
the alveoli, which pull fluid out of the pulmonary cap-
in localized areas of a lung or in an entire lung. Its most
illaries into the alveoli, thus causing the alveoli to fill
common causes are (1) total obstruction of the airway
completely with edema fluid. This almost always is the
or (2) lack of surfactant in the fluids lining the alveoli.
effect that occurs when an entire lung becomes
atelectatic, a condition called massive collapse of the
Airway Obstruction. The airway obstruction type of
lung.
atelectasis usually results from (1) blockage of many
The effects on overall pulmonary function caused by
small bronchi with mucus or (2) obstruction of a major
massive collapse (atelectasis) of an entire lung are
bronchus by either a large mucus plug or some solid
shown in Figure 42-7. Collapse of the lung tissue not
object such as a tumor. The air entrapped beyond the
only occludes the alveoli but also almost always
block is absorbed within minutes to hours by the blood
increases the resistance to blood flow through the pul-
flowing in the pulmonary capillaries. If the lung tissue
monary vessels of the collapsed lung. This resistance
Chapter 42
Respiratory Insufficiency—Pathophysiology, Diagnosis, Oxygen Therapy
529
Pulmonary arterial blood
Asthma
60% saturated with O2
Asthma is characterized by spastic contraction of the
smooth muscle in the bronchioles, which partially
obstructs the bronchioles and causes extremely diffi-
cult breathing. It occurs in 3 to 5 per cent of all people
Atelectasis
at some time in life.
The usual cause of asthma is contractile hypersensi-
tivity of the bronchioles in response to foreign sub-
stances in the air. In about 70 per cent of patients
Left
younger than age 30 years, the asthma is caused by
Right
pulmonary
pulmonary
allergic hypersensitivity, especially sensitivity to plant
vein 60%
vein
97%
pollens. In older people, the cause is almost always
saturated-
saturated
hypersensitivity to nonallergenic types of irritants in
flow 1/5 normal
the air, such as irritants in smog.
Aorta:
The allergic reaction that occurs in the allergic type
Blood 5/6 = 97%
of asthma is believed to occur in the following way: The
1/6 = 60%
Mean saturation
typical allergic person has a tendency to form abnor-
= 91%
mally large amounts of IgE antibodies, and these anti-
bodies cause allergic reactions when they react with the
Figure 42-7
specific antigens that have caused them to develop in
the first place, as explained in Chapter 34. In asthma,
Effect of atelectasis on aortic blood oxygen saturation.
these antibodies are mainly attached to mast cells that
are present in the lung interstitium in close association
with the bronchioles and small bronchi. When the asth-
matic person breathes in pollen to which he or she is
increase occurs partially because of the lung collapse
sensitive (that is, to which the person has developed
itself, which compresses and folds the vessels as the
IgE antibodies), the pollen reacts with the mast cell-
volume of the lung decreases. In addition, hypoxia in
attached antibodies and causes the mast cells to release
the collapsed alveoli causes additional vasoconstric-
several different substances. Among them are
(a)
tion, as explained in Chapter 38.
histamine, (b) slow-reacting substance of anaphylaxis
Because of the vascular constriction, blood flow
(which is a mixture of leukotrienes), (c) eosinophilic
through the atelectatic lung becomes slight. Fortu-
chemotactic factor, and (d) bradykinin. The combined
nately, most of the blood is routed through the venti-
effects of all these factors, especially the slow-reacting
lated lung and therefore becomes well aerated. In the
substance of anaphylaxis, are to produce (1) localized
situation shown in Figure 42-7, five sixths of the blood
edema in the walls of the small bronchioles, as well as
passes through the aerated lung and only one sixth
secretion of thick mucus into the bronchiolar lumens,
through the unaerated lung. As a result, the overall
and (2) spasm of the bronchiolar smooth muscle.
ventilation-perfusion ratio is only moderately com-
Therefore, the airway resistance increases greatly.
promised, so that the aortic blood has only mild
As discussed earlier in this chapter, the bronchiolar
oxygen desaturation despite total loss of ventilation in
diameter becomes more reduced during expiration
an entire lung.
than during inspiration in asthma, caused by bronchi-
olar collapse during expiratory effort that compresses
Lack of “Surfactant” as a Cause of Lung Collapse. The secre-
the outsides of the bronchioles. Because the bronchi-
tion and function of surfactant in the alveoli were
oles of the asthmatic lungs are already partially
discussed in Chapter 37. It was pointed out that the
occluded, further occlusion resulting from the external
surfactant is secreted by special alveolar epithelial
pressure creates especially severe obstruction during
cells into the fluids that coat the inside surface of the
expiration. That is, the asthmatic person often can
alveoli. The surfactant in turn decreases the surface
inspire quite adequately but has great difficulty expir-
tension in the alveoli 2- to 10-fold, which normally
ing. Clinical measurements show (1) greatly reduced
plays a major role in preventing alveolar collapse.
maximum expiratory rate and (2) reduced timed expi-
However, in a number of conditions, such as in hyaline
ratory volume. Also, all of this together results in
membrane disease (also called respiratory distress syn-
dyspnea, or “air hunger,” which is discussed later in
drome), which often occurs in newborn premature
this chapter.
babies, the quantity of surfactant secreted by the
The functional residual capacity and residual volume
alveoli is so greatly depressed that the surface tension
of the lung become especially increased during the
of the alveolar fluid becomes several times normal.
acute asthmatic attack because of the difficulty in
This causes a serious tendency for the lungs of these
expiring air from the lungs. Also, over a period of
babies to collapse or to become filled with fluid. As
years, the chest cage becomes permanently enlarged,
explained in Chapter 37, many of these infants die of
causing a “barrel chest,” and both the functional resid-
suffocation when large portions of the lungs become
ual capacity and lung residual volume become perma-
atelectatic.
nently increased.
530
Unit VII
Respiration
Tuberculosis
b. Diminished cellular metabolic capacity for using
oxygen, because of toxicity, vitamin deficiency, or
In tuberculosis, the tubercle bacilli cause a peculiar
other factors
tissue reaction in the lungs, including (1) invasion of
This classification of the types of hypoxia is mainly
the infected tissue by macrophages and (2) “walling
self-evident from the discussions earlier in the chapter.
off ” of the lesion by fibrous tissue to form the so-called
Only one of the types of hypoxia in the classification
tubercle. This walling-off process helps to limit further
needs further elaboration: this is the hypoxia caused
transmission of the tubercle bacilli in the lungs and
by inadequate capability of the body’s tissue cells to
therefore is part of the protective process against
use oxygen.
extension of the infection. However, in about 3 per
cent of all people who develop tuberculosis, if
Inadequate Tissue Capability to Use Oxygen. The classic
untreated, the walling-off process fails and tubercle
cause of inability of the tissues to use oxygen is cyanide
bacilli spread throughout the lungs, often causing
poisoning, in which the action of the enzyme
extreme destruction of lung tissue with formation of
cytochrome oxidase is completely blocked by the
large abscess cavities.
cyanide—to such an extent that the tissues simply
Thus, tuberculosis in its late stages is characterized
cannot use oxygen even when plenty is available. Also,
by many areas of fibrosis throughout the lungs, as well
deficiencies of some of the tissue cellular oxidative
as reduced total amount of functional lung tissue.
enzymes or of other elements in the tissue oxidative
These effects cause (1) increased “work” on the part
system can lead to this type of hypoxia. A special
of the respiratory muscles to cause pulmonary venti-
example occurs in the disease beriberi, in which several
lation and reduced vital capacity and breathing capac-
important steps in tissue utilization of oxygen and
ity; (2) reduced total respiratory membrane surface area
formation of carbon dioxide are compromised because
and increased thickness of the respiratory membrane,
of vitamin B deficiency.
causing progressively diminished pulmonary diffusing
capacity; and (3) abnormal ventilation-perfusion ratio
Effects of Hypoxia on the Body. Hypoxia, if severe enough,
in the lungs, further reducing overall pulmonary diffu-
can cause death of cells throughout the body, but in
sion of oxygen and carbon dioxide.
less severe degrees it causes principally (1) depressed
mental activity, sometimes culminating in coma, and
(2) reduced work capacity of the muscles. These effects
Hypoxia and Oxygen Therapy
are specifically discussed in Chapter 43 in relation to
high-altitude physiology.
Almost any of the conditions discussed in the past
few sections of this chapter can cause serious degrees
of bodywide cellular hypoxia. Sometimes, oxygen
Oxygen Therapy in Different Types
therapy is of great value; other times, it is of moderate
of Hypoxia
value; and, at still other times, it is of almost no value.
Therefore, it is important to understand the different
Oxygen can be administered by
(1) placing the
types of hypoxia; then we can discuss the physiologic
patient’s head in a “tent” that contains air fortified
principles of oxygen therapy. The following is a
with oxygen, (2) allowing the patient to breathe either
descriptive classification of the causes of hypoxia:
pure oxygen or high concentrations of oxygen from
1. Inadequate oxygenation of the blood in the lungs
a mask, or
(3) administering oxygen through an
because of extrinsic reasons
intranasal tube.
a. Deficiency of oxygen in the atmosphere
Recalling the basic physiologic principles of the
b. Hypoventilation (neuromuscular disorders)
different types of hypoxia, one can readily decide
2. Pulmonary disease
when oxygen therapy will be of value and, if so, how
a. Hypoventilation caused by increased airway
valuable.
resistance or decreased pulmonary compliance
In atmospheric hypoxia, oxygen therapy can com-
b. Abnormal alveolar ventilation-perfusion ratio
pletely correct the depressed oxygen level in the
(including either increased physiologic dead
inspired gases and, therefore, provide 100 per cent
space or increased physiologic shunt)
effective therapy.
c. Diminished respiratory membrane diffusion
In hypoventilation hypoxia, a person breathing 100
3. Venous-to-arterial shunts
(“right-to-left” cardiac
per cent oxygen can move five times as much oxygen
shunts)
into the alveoli with each breath as when breathing
4. Inadequate oxygen transport to the tissues by the
normal air. Therefore, here again oxygen therapy can
blood
be extremely beneficial. (However, this provides no
a. Anemia or abnormal hemoglobin
benefit for the excess blood carbon dioxide also caused
b. General circulatory deficiency
by the hypoventilation.)
c. Localized circulatory deficiency
(peripheral,
In hypoxia caused by impaired alveolar membrane
cerebral, coronary vessels)
diffusion, essentially the same result occurs as in
d. Tissue edema
hypoventilation hypoxia, because oxygen therapy can
5. Inadequate tissue capability of using oxygen
increase the Po2 in the lung alveoli from the normal
a. Poisoning of cellular oxidation enzymes
value of about 100 mm Hg to as high as 600 mm Hg.
Chapter 42
Respiratory Insufficiency—Pathophysiology, Diagnosis, Oxygen Therapy
531
dark blue-purple color that is transmitted through the
skin.
300
In general, definite cyanosis appears whenever the
arterial blood contains more than 5 grams of deoxy-
genated hemoglobin in each 100 milliliters of blood. A
Alveolar PO2 with tent therapy
person with anemia almost never becomes cyanotic
2
Normal alveolar PO
200
because there is not enough hemoglobin for 5 grams
Pulmonary edema + O2 therapy
to be deoxygenated in 100 milliliters of arterial blood.
Pulmonary edema with no therapy
Conversely, in a person with excess red blood cells, as
occurs in polycythemia vera, the great excess of avail-
100
able hemoglobin that can become deoxygenated leads
frequently to cyanosis, even under otherwise normal
Capillary blood
conditions.
0
Arterial end
Venous end
Blood in pulmonary capillary
Hypercapnia
Hypercapnia means excess carbon dioxide in the body
fluids.
Figure 42-8
One might suspect, on first thought, that any respi-
ratory condition that causes hypoxia would also cause
Absorption of oxygen into the pulmonary capillary blood in pul-
hypercapnia. However, hypercapnia usually occurs
monary edema with and without oxygen tent therapy.
in association with hypoxia only when the hypoxia
is caused by hypoventilation or circulatory deficiency.
The reasons for this are the following.
This raises the oxygen pressure gradient for diffusion
Hypoxia caused by too little oxygen in the air, too
of oxygen from the alveoli to the blood from the
little hemoglobin, or poisoning of the oxidative
normal value of 60 mm Hg to as high as 560 mm Hg,
enzymes has to do only with the availability of oxygen
an increase of more than 800 per cent. This highly ben-
or use of oxygen by the tissues. Therefore, it is readily
eficial effect of oxygen therapy in diffusion hypoxia is
understandable that hypercapnia is not a concomitant
demonstrated in Figure 42-8, which shows that the
of these types of hypoxia.
pulmonary blood in this patient with pulmonary
In hypoxia resulting from poor diffusion through
edema picks up oxygen three to four times as rapidly
the pulmonary membrane or through the tissues,
as would occur with no therapy.
serious hypercapnia usually does not occur at the same
In hypoxia caused by anemia, abnormal hemoglobin
time because carbon dioxide diffuses
20 times as
transport of oxygen, circulatory deficiency, or physio-
rapidly as oxygen. If hypercapnia does begin to occur,
logic shunt, oxygen therapy is of much less value
this immediately stimulates pulmonary ventilation,
because normal oxygen is already available in the
which corrects the hypercapnia but not necessarily the
alveoli. The problem instead is that one or more of the
hypoxia.
mechanisms for transporting oxygen from the lungs to
Conversely, in hypoxia caused by hypoventilation,
the tissues is deficient. Even so, a small amount of
carbon dioxide transfer between the alveoli and the
extra oxygen, between 7 and 30 per cent, can be trans-
atmosphere is affected as much as is oxygen transfer.
ported in the dissolved state in the blood when alveo-
Hypercapnia then occurs along with the hypoxia. And
lar oxygen is increased to maximum even though the
in circulatory deficiency, diminished flow of blood
amount transported by the hemoglobin is hardly
decreases carbon dioxide removal from the tissues,
altered. This small amount of extra oxygen may be the
resulting in tissue hypercapnia in addition to tissue
difference between life and death.
hypoxia. However, the transport capacity of the blood
In the different types of hypoxia caused by inade-
for carbon dioxide is more than three times that for
quate tissue use of oxygen, there is abnormality neither
oxygen, so that the resulting tissue hypercapnia is
of oxygen pickup by the lungs nor of transport to the
much less than the tissue hypoxia.
tissues. Instead, the tissue metabolic enzyme system is
When the alveolar Pco2 rises above about 60 to
simply incapable of using the oxygen that is delivered.
75 mm Hg, an otherwise normal person by then is
Therefore, oxygen therapy is of hardly any measura-
breathing about as rapidly and deeply as he or she
ble benefit.
can, and “air hunger,” also called dyspnea, becomes
severe.
If the Pco2 rises to 80 to 100 mm Hg, the person
Cyanosis
becomes lethargic and sometimes even semicomatose.
Anesthesia and death can result when the Pco2 rises
The term cyanosis means blueness of the skin, and its
to 120 to 150 mm Hg. At these higher levels of Pco2,
cause is excessive amounts of deoxygenated hemoglo-
the excess carbon dioxide now begins to depress res-
bin in the skin blood vessels, especially in the capil-
piration rather than stimulate it, thus causing a vicious
laries. This deoxygenated hemoglobin has an intense
circle: (1) more carbon dioxide, (2) further decrease in
532
Unit VII
Respiration
respiration,
(3) then more carbon dioxide, and so
A
forth—culminating rapidly in a respiratory death.
Dyspnea
Mechanism
for applying
positive and
Dyspnea means mental anguish associated with inabil-
negative
ity to ventilate enough to satisfy the demand for air. A
pressure
common synonym is air hunger.
At least three factors often enter into the develop-
ment of the sensation of dyspnea. They are (1) abnor-
B
Positive
Negative
mality of respiratory gases in the body fluids, especially
pressure
pressure
hypercapnia and, to a much less extent, hypoxia; (2)
valve
valve
the amount of work that must be performed by the res-
piratory muscles to provide adequate ventilation; and
(3) state of mind.
A person becomes very dyspneic especially from
excess buildup of carbon dioxide in the body fluids. At
times, however, the levels of both carbon dioxide and
oxygen in the body fluids are normal, but to attain this
normality of the respiratory gases, the person has to
breathe forcefully. In these instances, the forceful
activity of the respiratory muscles frequently gives the
person a sensation of dyspnea.
Leather diaphragm
Finally, the person’s respiratory functions may be
normal and still dyspnea may be experienced because
Figure 42-9
of an abnormal state of mind. This is called neurogenic
A, Resuscitator. B, Tank respirator.
dyspnea or emotional dyspnea. For instance, almost
anyone momentarily thinking about the act of breath-
ing may suddenly start taking breaths a little more
deeply than ordinarily because of a feeling of mild
dyspnea. This feeling is greatly enhanced in people
who have a psychological fear of not being able to
receive a sufficient quantity of air, such as on entering
and the head protruding through a flexible but airtight
small or crowded rooms.
collar. At the end of the tank opposite the patient’s
head a motor-driven leather diaphragm moves back
and forth with sufficient excursion to raise and lower
Artificial Respiration
the pressure inside the tank. As the leather diaphragm
moves inward, positive pressure develops around the
Resuscitator. Many types of respiratory resuscitators
body and causes expiration; as the diaphragm moves
are available, and each has its own characteristic prin-
outward, negative pressure causes inspiration. Check
ciples of operation. The resuscitator shown in Figure
valves on the respirator control the positive and
42-9A consists of a tank supply of oxygen or air; a
negative pressures. Ordinarily these pressures are
mechanism for applying intermittent positive pressure
adjusted so that the negative pressure that causes
and, with some machines, negative pressure as well;
inspiration falls to -10 to -20 cm H2O and the positive
and a mask that fits over the face of the patient or a
pressure rises to 0 to +5 cm H2O.
connector for joining the equipment to an endotra-
cheal tube. This apparatus forces air through the mask
Effect of the Resuscitator and the Tank Respirator on Venous
or endotracheal tube into the lungs of the patient
Return. When air is forced into the lungs under posi-
during the positive-pressure cycle of the resuscitator
tive pressure by a resuscitator, or when the pressure
and then usually allows the air to flow passively out of
around the patient’s body is reduced by the tank
the lungs during the remainder of the cycle.
respirator, the pressure inside the lungs becomes
Earlier resuscitators often caused damage to the
greater than pressure everywhere else in the body.
lungs because of excessive positive pressure. Their
Flow of blood into the chest and heart from the
usage was at one time greatly decried. However, resus-
peripheral veins becomes impeded. As a result, use
citators now have adjustable positive-pressure limits
of excessive pressures with either the resuscitator or
that are commonly set at 12 to 15 cm H2O pressure for
the tank respirator can reduce the cardiac output—
normal lungs (but sometimes much higher for non-
sometimes to lethal levels. For instance, continuous
compliant lungs).
exposure for more than a few minutes to greater than
30 mm Hg positive pressure in the lungs can cause
Tank Respirator (the “Iron-Lung”). Figure 42-9B shows the
death because of inadequate venous return to the
tank respirator with a patient’s body inside the tank
heart.
Chapter 42
Respiratory Insufficiency—Pathophysiology, Diagnosis, Oxygen Therapy
533
References
Page S, Ammit AJ, Black JL, Armour CL: Human mast cell
and airway smooth muscle cell interactions: implications
for asthma. Am J Physiol Lung Cell Mol Physiol 281:
Albert R, Spiro S, Jett J: Comprehensive Respiratory Medi-
L1313, 2001.
cine. Philadelphia: Mosby, 2002.
Rodrigo GJ, Rodrigo C, Hall JB: Acute asthma in adults: a
Barnes PJ, Adcock IM: How do corticosteroids work in
review. Chest 125:1081, 2004.
asthma? Ann Intern Med 139:359, 2003.
Schwiebert LM: Cystic fibrosis, gene therapy, and lung
Basu S, Fenton MJ: Toll-like receptors: function and roles in
inflammation: for better or worse? Am J Physiol Lung Cell
lung disease. Am J Physiol Lung Cell Mol Physiol 286:
Mol Physiol 286:L715, 2004.
L887, 2004.
Sin DD, McAlister FA, Man SF, Anthonisen NR: Contem-
Cardoso WV: Molecular regulation of lung development.
porary management of chronic obstructive pulmonary
Annu Rev Physiol 63:471, 2001.
disease: scientific review. JAMA 290:2301, 2003.
Carter EP, Garat C, Imamura M: Continual emerging roles
Wardlaw AJ, Brightling CE, Green R, et al: New insights
of HO-1: protection against airway inflammation. Am J
into the relationship between airway inflammation and
Physiol Lung Cell Mol Physiol 287:L24, 2004.
asthma. Clin Sci (Lond) 103:201, 2002.
Dwyer TM: Cigarette smoke-induced airway inflammation
West JB: Pulmonary Physiology and Pathophysiology: An
as sampled by the expired breath condensate. Am J Med
Integrated, Case-Based Approach. Philadelphia: Lippin-
Sci 326:174, 2003.
cott Williams & Wilkins, 2001.
Knight DA, Holgate ST: The airway epithelium: structural
Whitsett JA, Weaver TE: Hydrophobic surfactant proteins
and functional properties in health and disease. Respirol-
in lung function and disease. N Engl J Med 347:2141,
ogy 8:432, 2003.
2002.
McConnell AK, Romer LM: Dyspnoea in health and
Wills-Karp M, Ewart SL: Time to draw breath: asthma-
obstructive pulmonary disease: the role of respiratory
susceptibility genes are identified. Nat Rev Genet 5:376,
muscle function and training. Sports Med 34:117, 2004.
2004.
Naureckas ET, Solway J: Clinical practice. Mild asthma.
N Engl J Med 345:1257, 2001.
U N I T VIII
Aviation, Space, and
Deep-Sea Diving
Physiology
43. Aviation, High-Altitude, and Space Physiology
44. Physiology of Deep-Sea Diving and Other
Hyperbaric Conditions
C H A P T E R
4
3
Aviation, High-Altitude,
and Space Physiology
As we have ascended to higher and higher altitudes
in aviation, mountain climbing, and space vehicles,
it has become progressively more important to
understand the effects of altitude and low gas pres-
sures (as well as several other factors—acceleratory
forces, weightlessness, and so forth) on the human
body. This chapter deals with these problems.
Effects of Low Oxygen Pressure on the Body
Barometric Pressures at Different Altitudes. Table 43-1 gives the approximate baro-
metric and oxygen pressures at different altitudes, showing that at sea level, the
barometric pressure is 760 mm Hg; at 10,000 feet, only 523 mm Hg; and at
50,000 feet, 87 mm Hg. This decrease in barometric pressure is the basic cause
of all the hypoxia problems in high-altitude physiology because, as the baro-
metric pressure decreases, the atmospheric oxygen partial pressure decreases
proportionately, remaining at all times slightly less than 21 per cent of the total
barometric pressure—Po2 at sea level about 159 mm Hg, but at 50,000 feet only
18 mm Hg.
Alveolar PO2 at Different Elevations
Carbon Dioxide and Water Vapor Decrease the Alveolar Oxygen. Even at high altitudes,
carbon dioxide is continually excreted from the pulmonary blood into the
alveoli. Also, water vaporizes into the inspired air from the respiratory surfaces.
These two gases dilute the oxygen in the alveoli, thus reducing the oxygen con-
centration. Water vapor pressure in the alveoli remains 47 mm Hg as long as
the body temperature is normal, regardless of altitude.
In the case of carbon dioxide, during exposure to very high altitudes, the alve-
olar Pco2 falls from the sea-level value of 40 mm Hg to lower values. In the
acclimatized person, who increases his or her ventilation about fivefold, the Pco2
falls to about 7 mm Hg because of increased respiration.
Now let us see how the pressures of these two gases affect the alveolar
oxygen. For instance, assume that the barometric pressure falls from the normal
sea-level value of 760 mm Hg to 253 mm Hg, which is the usual measured value
at the top of 29,028-foot Mount Everest. Forty-seven millimeters of mercury of
this must be water vapor, leaving only 206 mm Hg for all the other gases. In the
acclimatized person, 7 mm of the 206 mm Hg must be carbon dioxide, leaving
only 199 mm Hg. If there were no use of oxygen by the body, one fifth of this
199 mm Hg would be oxygen and four fifths would be nitrogen; that is, the Po2
in the alveoli would be 40 mm Hg. However, some of this remaining alveolar
oxygen is continually being absorbed into the blood, leaving about 35 mm Hg
oxygen pressure in the alveoli. At the summit of Mount Everest, only the best
of acclimatized people can barely survive when breathing air. But the effect is
very different when the person is breathing pure oxygen, as we see in the fol-
lowing discussions.
537
538
Unit VIII
Aviation, Space, and Deep-Sea Diving Physiology
Table 43-1
Effects of Acute Exposure to Low Atmospheric Pressures on Alveolar Gas Concentrations and Arterial Oxygen Saturation*
Breathing Air
Breathing Pure Oxygen
Arterial
Arterial
Barometric
PO2 in
PCO2 in
PO2 in
Oxygen
PCO2 in
PO2 in
Oxygen
Pressure
Air
Alveoli
Alveoli
Saturation
Alveoli
Alveoli
Saturation
Altitude (ft)
(mm Hg)
(mm Hg)
(mm Hg)
(mm Hg)
(%)
(mm Hg)
(mm Hg)
(%)
0
760
159
40 (40)
104 (104)
97 (97)
40
673
100
10,000
523
110
36 (23)
67 (77)
90 (92)
40
436
100
20,000
349
73
24 (10)
40 (53)
73 (85)
40
262
100
30,000
226
47
24 (7)
18 (30)
24 (38)
40
139
99
40,000
141
29
36
58
84
50,000
87
18
24
16
15
* Numbers in parentheses are acclimatized values.
Alveolar PO2 at Different Altitudes. The fifth column of
Table 43-1 shows the approximate Po2s in the alveoli
at different altitudes when one is breathing air for both
Breathing pure oxygen
the unacclimatized and the acclimatized person. At sea
Breathing air
level, the alveolar Po2 is 104 mm Hg; at 20,000 feet alti-
100
tude, it falls to about 40 mm Hg in the unacclimatized
person but only to 53 mm Hg in the acclimatized. The
90
difference between these two is that alveolar ventila-
tion increases much more in the acclimatized person
80
than in the unacclimatized person, as we discuss later.
Saturation of Hemoglobin with Oxygen at Different Altitudes.
70
Figure 43-1 shows arterial blood oxygen saturation at
different altitudes while a person is breathing air and
60
while breathing oxygen. Up to an altitude of about
10,000 feet, even when air is breathed, the arterial
50
oxygen saturation remains at least as high as 90 per
0
10
20
30
40
50
cent. Above 10,000 feet, the arterial oxygen saturation
Altitude (thousands of feet)
falls rapidly, as shown by the blue curve of the figure,
until it is slightly less than 70 per cent at 20,000 feet
and much less at still higher altitudes.
Figure 43-1
Effect of high altitude on arterial oxygen saturation when breath-
Effect of Breathing Pure Oxygen on
ing air and when breathing pure oxygen.
Alveolar PO2 at Different Altitudes
breathing pure oxygen in an unpressurized airplane
When a person breathes pure oxygen instead of air,
can ascend to far higher altitudes than one breathing
most of the space in the alveoli formerly occupied by
air. For instance, the arterial saturation at 47,000 feet
nitrogen becomes occupied by oxygen. At 30,000 feet,
when one is breathing oxygen is about 50 per cent and
an aviator could have an alveolar Po2 as high as
is equivalent to the arterial oxygen saturation at 23,000
139 mm Hg instead of the 18 mm Hg when breathing
feet when one is breathing air. In addition, because an
air (see Table 43-1).
unacclimatized person usually can remain conscious
The red curve of Figure 43-1 shows arterial blood
until the arterial oxygen saturation falls to 50 per cent,
hemoglobin oxygen saturation at different altitudes
for short exposure times the ceiling for an aviator in
when one is breathing pure oxygen. Note that the sat-
an unpressurized airplane when breathing air is about
uration remains above 90 per cent until the aviator
23,000 feet and when breathing pure oxygen is about
ascends to about 39,000 feet; then it falls rapidly to
47,000 feet, provided the oxygen-supplying equipment
about 50 per cent at about 47,000 feet.
operates perfectly.
The “Ceiling” When Breathing Air
and When Breathing Oxygen in an
Acute Effects of Hypoxia
Unpressurized Airplane
Comparing the two arterial blood oxygen satura-
Some of the important acute effects of hypoxia in the
tion curves in Figure 43-1, one notes that an aviator
unacclimatized person breathing air, beginning at an
Chapter 43
Aviation, High-Altitude, and Space Physiology
539
altitude of about 12,000 feet, are drowsiness, lassitude,
An important mechanism for the gradual decrease
mental and muscle fatigue, sometimes headache, occa-
in bicarbonate concentration is compensation by the
sionally nausea, and sometimes euphoria. These effects
kidneys for the respiratory alkalosis, as discussed in
progress to a stage of twitchings or seizures above
Chapter 30. The kidneys respond to decreased Pco2
18,000 feet and end, above 23,000 feet in the unaccli-
by reducing hydrogen ion secretion and increasing
matized person, in coma, followed shortly thereafter
bicarbonate excretion. This metabolic compensation
by death.
for the respiratory alkalosis gradually reduces plasma
One of the most important effects of hypoxia is
and cerebrospinal fluid bicarbonate concentration and
decreased mental proficiency, which decreases judg-
pH toward normal and removes part of the inhibitory
ment, memory, and performance of discrete motor
effect on respiration of low hydrogen ion concentra-
movements. For instance, if an unacclimatized aviator
tion. Thus, the respiratory centers are much more
stays at 15,000 feet for 1 hour, mental proficiency ordi-
responsive to the peripheral chemoreceptor stimulus
narily falls to about 50 per cent of normal, and after
caused by the hypoxia after the kidneys compensate
18 hours at this level it falls to about 20 per cent of
for the alkalosis.
normal.
Increase in Red Blood Cells and Hemoglobin Concentration
During Acclimatization. As discussed in Chapter
32,
Acclimatization to Low PO2
hypoxia is the principal stimulus for causing an
increase in red blood cell production. Ordinarily, when
A person remaining at high altitudes for days, weeks,
a person remains exposed to low oxygen for weeks at
or years becomes more and more acclimatized to the
a time, the hematocrit rises slowly from a normal value
low Po2, so that it causes fewer deleterious effects on
of 40 to 45 to an average of about 60, with an average
the body. And it becomes possible for the person to
increase in whole blood hemoglobin concentration
work harder without hypoxic effects or to ascend to
from normal of 15 g/dl to about 20 g/dl.
still higher altitudes.
In addition, the blood volume also increases, often
The principal means by which acclimatization comes
by 20 to 30 per cent, and this increase times the
about are (1) a great increase in pulmonary ventila-
increased blood hemoglobin concentration gives an
tion, (2) increased numbers of red blood cells, (3)
increase in total body hemoglobin of 50 or more per
increased diffusing capacity of the lungs, (4) increased
cent.
vascularity of the peripheral tissues, and (5) increased
ability of the tissue cells to use oxygen despite low Po2.
Increased Diffusing Capacity After Acclimatization. It will
be recalled that the normal diffusing capacity for
Increased Pulmonary Ventilation—Role of Arterial Chemore-
oxygen through the pulmonary membrane is about
ceptors. Immediate exposure to low Po2 stimulates the
21 ml/mm Hg/min, and this diffusing capacity can
arterial chemoreceptors, and this increases alveolar
increase as much as threefold during exercise. A
ventilation to a maximum of about 1.65 times normal.
similar increase in diffusing capacity occurs at high
Therefore, compensation occurs within seconds for the
altitude.
high altitude, and it alone allows the person to rise
Part of the increase results from increased pul-
several thousand feet higher than would be possible
monary capillary blood volume, which expands the
without the increased ventilation. Then, if the person
capillaries and increases the surface area through
remains at very high altitude for several days, the
which oxygen can diffuse into the blood. Another part
chemoreceptors increase ventilation still more, up to
results from an increase in lung air volume, which
about five times normal.
expands the surface area of the alveolar-capillary
The immediate increase in pulmonary ventilation on
interface still more. A final part results from an
rising to a high altitude blows off large quantities of
increase in pulmonary arterial blood pressure; this
carbon dioxide, reducing the Pco2 and increasing the
forces blood into greater numbers of alveolar capil-
pH of the body fluids. These changes inhibit the brain
laries than normally—especially in the upper parts
stem respiratory center and thereby oppose the effect
of the lungs, which are poorly perfused under usual
of low PO2 to stimulate respiration by way of the
conditions.
peripheral arterial chemoreceptors in the carotid and
aortic bodies. But during the ensuing 2 to 5 days, this
Peripheral Circulatory System Changes During Acclimatiza-
inhibition fades away, allowing the respiratory center
tion—Increased Tissue Capillarity. The cardiac output
to respond with full force to the peripheral chemore-
often increases as much as 30 per cent immediately
ceptor stimulus from hypoxia, and ventilation in-
after a person ascends to high altitude but then
creases to about five times normal.
decreases back toward normal over a period of weeks
The cause of this fading inhibition is believed to be
as the blood hematocrit increases, so that the amount
mainly a reduction of bicarbonate ion concentration in
of oxygen transported to the peripheral body tissues
the cerebrospinal fluid as well as in the brain tissues.
remains about normal.
This in turn decreases the pH in the fluids surround-
Another circulatory adaptation is growth of
ing the chemosensitive neurons of the respiratory
increased numbers of systemic circulatory capillaries
center, thus increasing the respiratory stimulatory
in the nonpulmonary tissues, which is called in-
activity of the center.
creased tissue capillarity (or angiogenesis). This occurs
540
Unit VIII
Aviation, Space, and Deep-Sea Diving Physiology
especially in animals born and bred at high altitudes
but less so in animals that later in life become exposed
to high altitude.
Mountain dwellers
28
In active tissues exposed to chronic hypoxia, the
26
(15,000 ft)
increase in capillarity is especially marked. For
24
(Arterial values)
instance, capillary density in right ventricular muscle
22
X
increases markedly because of the combined effects
20
X
of hypoxia and excess workload on the right ventricle
18
X
16
X
Sea-level dwellers
caused by pulmonary hypertension at high altitude.
14
(Venous values)
Cellular Acclimatization. In animals native to altitudes of
12
10
13,000 to 17,000 feet, cell mitochondria and cellular
8
oxidative enzyme systems are slightly more plentiful
6
than in sea-level inhabitants. Therefore, it is presumed
4
that the tissue cells of high altitude-acclimatized
2
human beings also can use oxygen more effectively
0
than can their sea-level counterparts.
0
20
40
60
80
100
120
140
Pressure of oxygen in blood (PO2) (mm Hg)
Natural Acclimatization
of Native Human Beings Living
at High Altitudes
Figure 43-2
Many native human beings in the Andes and in the
Oxygen-hemoglobin dissociation curves for blood of high-altitude
Himalayas live at altitudes above 13,000 feet—one
residents
(red curve) and sea-level residents
(blue curve),
showing the respective arterial and venous PO2 levels and oxygen
group in the Peruvian Andes lives at an altitude of
contents as recorded in their native surroundings. (Data from
17,500 feet and works a mine at an altitude of 19,000
Oxygen-dissociation curves for bloods of high-altitude and sea-
feet. Many of these natives are born at these altitudes
level residents. PAHO Scientific Publication No. 140, Life at High
and live there all their lives. In all aspects of acclima-
Altitudes, 1966.)
tization, the natives are superior to even the best-
acclimatized lowlanders, even though the lowlanders
might also have lived at high altitudes for 10 or more
years. Acclimatization of the natives begins in infancy.
not only skeletal muscles but also cardiac muscles.
The chest size, especially, is greatly increased, whereas
In general, work capacity is reduced in direct pro-
the body size is somewhat decreased, giving a high
portion to the decrease in maximum rate of oxygen
ratio of ventilatory capacity to body mass. In addition,
uptake that the body can achieve.
their hearts, which from birth onward pump extra
To give an idea of the importance of acclimatization
amounts of cardiac output, are considerably larger
in increasing work capacity, consider this: The work
than the hearts of lowlanders.
capacities as per cent of normal for unacclimatized and
Delivery of oxygen by the blood to the tissues is also
acclimatized people at an altitude of 17,000 feet are as
highly facilitated in these natives. For instance, Figure
follows:
43-2 shows oxygen-hemoglobin dissociation curves for
natives who live at sea level and for their counterparts
who live at 15,000 feet. Note that the arterial oxygen
Work capacity
Po2 in the natives at high altitude is only 40 mm Hg,
(per cent of normal)
but because of the greater quantity of hemoglobin,
Unacclimatized
50
the quantity of oxygen in their arterial blood is greater
Acclimatized for 2 months
68
than that in the blood of the natives at the lower
Native living at 13,200 feet but working at
altitude. Note also that the venous Po2 in the high-
17,000 feet
87
altitude natives is only 15 mm Hg less than the venous
Po2 for the lowlanders, despite the very low arterial
Thus, naturally acclimatized native persons can
Po2, indicating that oxygen transport to the tissues is
achieve a daily work output even at high altitude
exceedingly effective in the naturally acclimatized
almost equal to that of a lowlander at sea level, but
high-altitude natives.
even well-acclimatized lowlanders can almost never
achieve this result.
Reduced Work Capacity at
High Altitudes and Positive Effect
of Acclimatization
Acute Mountain Sickness and
High-Altitude Pulmonary Edema
In addition to the mental depression caused by
hypoxia, as discussed earlier, the work capacity of all
A small percentage of people who ascend rapidly to
muscles is greatly decreased in hypoxia. This includes
high altitudes become acutely sick and can die if not
Chapter 43
Aviation, High-Altitude, and Space Physiology
541
given oxygen or removed to a low altitude. The sick-
Effects of Acceleratory Forces
ness begins from a few hours up to about 2 days after
on the Body in Aviation and
ascent. Two events frequently occur:
1. Acute cerebral edema. This is believed to result
Space Physiology
from local vasodilation of the cerebral blood
vessels, caused by the hypoxia. Dilation of the
Because of rapid changes in velocity and direction of
arterioles increases blood flow into the capillaries,
motion in airplanes or spacecraft, several types of
thus increasing capillary pressure, which in turn
acceleratory forces affect the body during flight. At the
causes fluid to leak into the cerebral tissues.
beginning of flight, simple linear acceleration occurs;
The cerebral edema can then lead to severe
at the end of flight, deceleration; and every time the
disorientation and other effects related to cerebral
vehicle turns, centrifugal acceleration.
dysfunction.
2. Acute pulmonary edema. The cause of this is still
unknown, but a suggested answer is the following:
The severe hypoxia causes the pulmonary arterioles
Centrifugal Acceleratory Forces
to constrict potently, but the constriction is much
greater in some parts of the lungs than in other
When an airplane makes a turn, the force of cen-
parts, so that more and more of the pulmonary
trifugal acceleration is determined by the following
blood flow is forced through fewer and fewer still
relation:
unconstricted pulmonary vessels. The postulated
2
result is that the capillary pressure in these areas of
mv
f =
the lungs becomes especially high and local edema
r
occurs. Extension of the process to progressively
more areas of the lungs leads to spreading
in which f is centrifugal acceleratory force, m is the
pulmonary edema and severe pulmonary
mass of the object, v is velocity of travel, and r is radius
dysfunction that can be lethal. Allowing the person
of curvature of the turn. From this formula, it is
to breathe oxygen usually reverses the process
obvious that as the velocity increases, the force of
within hours.
centrifugal acceleration increases in proportion to the
square of the velocity. It is also obvious that the force
of acceleration is directly proportional to the sharpness
of the turn (the less the radius).
Chronic Mountain Sickness
Measurement of Acceleratory Force—“G.” When an
Occasionally, a person who remains at high altitude
aviator is simply sitting in his seat, the force with which
too long develops chronic mountain sickness, in which
he is pressing against the seat results from the pull of
the following effects occur: (1) the red cell mass and
gravity and is equal to his weight. The intensity of this
hematocrit become exceptionally high, (2) the pul-
force is said to be +1 G because it is equal to the pull
monary arterial pressure becomes elevated even more
of gravity. If the force with which he presses against
than the normal elevation that occurs during acclima-
the seat becomes five times his normal weight during
tization, (3) the right side of the heart becomes greatly
pull-out from a dive, the force acting on the seat is
enlarged, (4) the peripheral arterial pressure begins to
+5 G.
fall, (5) congestive heart failure ensues, and (6) death
If the airplane goes through an outside loop so that
often follows unless the person is removed to a lower
the person is held down by his seat belt, negative G is
altitude.
applied to his body; if the force with which he is held
The causes of this sequence of events are probably
down by his belt is equal to the weight of his body, the
threefold: First, the red cell mass becomes so great that
negative force is -1 G.
the blood viscosity increases severalfold; this increased
viscosity tends to decrease tissue blood flow so that
Effects of Centrifugal Acceleratory Force on the Body—
oxygen delivery also begins to decrease. Second, the
(Positive G)
pulmonary arterioles become vasoconstricted because
Effects on the Circulatory System. The most impor-
of the lung hypoxia. This results from the hypoxic
tant effect of centrifugal acceleration is on the circu-
vascular constrictor effect that normally operates to
latory system, because blood is mobile and can be
divert blood flow from low-oxygen to high-oxygen
translocated by centrifugal forces.
alveoli, as explained in Chapter 38. But because all the
When an aviator is subjected to positive G, blood is
alveoli are now in the low-oxygen state, all the arteri-
centrifuged toward the lowermost part of the body.
oles become constricted, the pulmonary arterial pres-
Thus, if the centrifugal acceleratory force is +5 G and
sure rises excessively, and the right side of the heart
the person is in an immobilized standing position,
fails. Third, the alveolar arteriolar spasm diverts much
the pressure in the veins of the feet becomes greatly
of the blood flow through nonalveolar pulmonary
increased (to about 450 mm Hg). In the sitting posi-
vessels, thus causing an excess of pulmonary shunt
tion, the pressure becomes nearly 300 mm Hg. And, as
blood flow where the blood is poorly oxygenated; this
pressure in the vessels of the lower body increases,
further compounds the problem. Most of these people
these vessels passively dilate so that a major portion
recover within days or weeks when they are moved to
of the blood from the upper body is translocated into
a lower altitude.
the lower vessels. Because the heart cannot pump
542
Unit VIII
Aviation, Space, and Deep-Sea Diving Physiology
cranium show less tendency for rupture than would be
expected for the following reason: The cerebrospinal
fluid is centrifuged toward the head at the same time
that blood is centrifuged toward the cranial vessels,
100
and the greatly increased pressure of the cerebrospinal
fluid acts as a cushioning buffer on the outside of the
50
brain to prevent intracerebral vascular rupture.
Because the eyes are not protected by the cranium,
0
intense hyperemia occurs in them during strong nega-
0
5
10
15
20
25
30
tive G. As a result, the eyes often become temporarily
Time from start of G to symptoms
blinded with “red-out.”
(sec)
Protection of the Body Against Centrifugal Acceleratory
Forces. Specific procedures and apparatus have been
Figure 43-3
developed to protect aviators against the circulatory
collapse that might occur during positive G. First, if
Changes in systolic (top of curve) and diastolic (bottom of curve)
arterial pressures after abrupt and continuing exposure of a sitting
the aviator tightens his or her abdominal muscles to
person to an acceleratory force from top to bottom of 3.3. G. (Data
an extreme degree and leans forward to compress the
from Martin EE, Henry JP: Effects of time and temperature upon
abdomen, some of the pooling of blood in the large
tolerance to positive acceleration. J Aviation Med 22:382, 1951.)
vessels of the abdomen can be prevented, there-
by delaying the onset of blackout. Also, special “anti-
G” suits have been devised to prevent pooling of blood
unless blood returns to it, the greater the quantity of
in the lower abdomen and legs. The simplest of these
blood “pooled” in this way in the lower body, the less
applies positive pressure to the legs and abdomen by
that is available for the cardiac output.
inflating compression bags as the G increases. Theo-
Figure
43-3 shows the changes in systolic and
retically, a pilot submerged in a tank or suit of water
diastolic arterial pressures (top and bottom curves,
might experience little effect of G forces on the circu-
respectively) in the upper body when a centrifugal
lation because the pressures developed in the water
acceleratory force of +3.3 G is suddenly applied to a
pressing on the outside of the body during centrifugal
sitting person. Note that both these pressures fall
acceleration would almost exactly balance the forces
below 22 mm Hg for the first few seconds after the
acting in the body. However, the presence of air in
acceleration begins but then return to a systolic pres-
the lungs still allows displacement of the heart, lung
sure of about 55 mm Hg and a diastolic pressure of
tissues, and diaphragm into seriously abnormal posi-
20 mm Hg within another 10 to 15 seconds. This sec-
tions despite submersion in water. Therefore, even if
ondary recovery is caused mainly by activation of the
this procedure were used, the limit of safety almost
baroreceptor reflexes.
certainly would still be less than 10 G.
Acceleration greater than 4 to 6 G causes “black-
out” of vision within a few seconds and unconscious-
ness shortly thereafter. If this great degree of
Effects of Linear Acceleratory Forces
acceleration is continued, the person will die.
on the Body
Effects on the Vertebrae. Extremely high acceleratory
forces for even a fraction of a second can fracture the
Acceleratory Forces in Space Travel. Unlike an airplane, a
vertebrae. The degree of positive acceleration that the
spacecraft cannot make rapid turns; therefore, cen-
average person can withstand in the sitting position
trifugal acceleration is of little importance except
before vertebral fracture occurs is about 20 G.
when the spacecraft goes into abnormal gyrations.
However, blast-off acceleration and landing decelera-
Negative G. The effects of negative G on the body are
tion can be tremendous; both of these are types
less dramatic acutely but possibly more damaging per-
of linear acceleration, one positive and the other
manently than the effects of positive G. An aviator can
negative.
usually go through outside loops up to negative accel-
Figure 43-4 shows an approximate profile of accel-
eratory forces of -4 to -5 G without causing perma-
eration during blast-off in a three-stage spacecraft,
nent harm, although causing intense momentary
demonstrating that the first-stage booster causes accel-
hyperemia of the head. Occasionally, psychotic distur-
eration as high as 9 G, and the second-stage booster as
bances lasting for 15 to 20 minutes occur as a result of
high as 8 G. In the standing position, the human body
brain edema.
could not withstand this much acceleration, but in a
Occasionally, negative G forces can be so great
semireclining position transverse to the axis of acceler-
(-20 G, for instance) and centrifugation of the blood
ation, this amount of acceleration can be withstood
into the head is so great that the cerebral blood pres-
with ease despite the fact that the acceleratory forces
sure reaches 300 to 400 mm Hg, sometimes causing
continue for as long as several minutes at a time.
small vessels on the surface of the head and in the
Therefore, we see the reason for the reclining seats
brain to rupture. However, the vessels inside the
used by astronauts.
Chapter 43
Aviation, High-Altitude, and Space Physiology
543
other words, the speed of landing is about 20 feet per
second, and the force of impact against the earth is
10
1/81 the impact force without a parachute. Even so,
the force of impact is still great enough to cause con-
siderable damage to the body unless the parachutist
8
is properly trained in landing. Actually, the force of
impact with the earth is about the same as that which
6
would be experienced by jumping without a parachute
from a height of about 6 feet. Unless forewarned, the
parachutist will be tricked by his senses into striking
4
the earth with extended legs, and this will result in
tremendous deceleratory forces along the skeletal axis
of the body, resulting in fracture of his pelvis, verte-
2
brae, or leg. Consequently, the trained parachutist
First
Second
Space
strikes the earth with knees bent but muscles taut to
booster
booster
ship
0
cushion the shock of landing.
0
1
2
3
4
5
Minutes
“Artificial Climate” in the
Sealed Spacecraft
Figure 43-4
Because there is no atmosphere in outer space, an arti-
Acceleratory forces during takeoff of a spacecraft.
ficial atmosphere and climate must be produced in a
spacecraft. Most important, the oxygen concentration
must remain high enough and the carbon dioxide con-
Problems also occur during deceleration when the
centration low enough to prevent suffocation. In some
spacecraft re-enters the atmosphere. A person travel-
earlier space missions, a capsule atmosphere contain-
ing at Mach 1 (the speed of sound and of fast air-
ing pure oxygen at about 260 mm Hg pressure was
planes) can be safely decelerated in a distance of about
used, but in the modern space shuttle, gases about
0.12 mile, whereas a person traveling at a speed of
equal to those in normal air are used, with four times
Mach 100 (a speed possible in interplanetary space
as much nitrogen as oxygen and a total pressure of
travel) would require a distance of about 10,000 miles
760 mm Hg. The presence of nitrogen in the mixture
for safe deceleration. The principal reason for this dif-
greatly diminishes the likelihood of fire and explosion.
ference is that the total amount of energy that must
It also protects against development of local patches
be dispelled during deceleration is proportional to
of lung atelectasis that often occur when breathing
the square of the velocity, which alone increases the
pure oxygen because oxygen is absorbed rapidly when
required distance for decelerations between Mach 1
small bronchi are temporarily blocked by mucous
versus Mach 100 about 10,000-fold. But in addition to
plugs.
this, a human being can withstand far less deceleration
For space travel lasting more than several months,
if the period of deceleration lasts for a long time than
it is impractical to carry along an adequate oxygen
for a short time. Therefore, deceleration must be
supply. For this reason, recycling techniques have been
accomplished much more slowly from high velocities
proposed for use of the same oxygen over and over
than is necessary at lower velocities.
again. Some recycling processes depend on purely
physical procedures, such as electrolysis of water to
Deceleratory Forces Associated with Parachute Jumps.
release oxygen. Others depend on biological methods,
When the parachuting aviator leaves the airplane, his
such as use of algae with their large store of chloro-
velocity of fall is at first exactly 0 feet per second.
phyll to release oxygen from carbon dioxide by the
However, because of the acceleratory force of gravity,
process of photosynthesis. A completely satisfactory
within 1 second his velocity of fall is 32 feet per second
system for recycling has yet to be achieved.
(if there is no air resistance); in 2 seconds it is 64 feet
per second; and so on. As the velocity of fall increases,
the air resistance tending to slow the fall also increases.
Weightlessness in Space
Finally, the deceleratory force of the air resistance
exactly balances the acceleratory force of gravity, so
A person in an orbiting satellite or a nonpropelled
that after falling for about 12 seconds, the person will
spacecraft experiences weightlessness, or a state of
be falling at a “terminal velocity” of 109 to 119 miles
near-zero G force, which is sometimes called micro-
per hour (175 feet per second). If the parachutist has
gravity. That is, the person is not drawn toward the
already reached terminal velocity before opening his
bottom, sides, or top of the spacecraft but simply floats
parachute, an “opening shock load” of up to 1200
inside its chambers. The cause of this is not failure of
pounds can occur on the parachute shrouds.
gravity to pull on the body, because gravity from any
The usual-sized parachute slows the fall of the para-
nearby heavenly body is still active. However, the
chutist to about one ninth the terminal velocity. In
gravity acts on both the spacecraft and the person at
544
Unit VIII
Aviation, Space, and Deep-Sea Diving Physiology
the same time, so that both are pulled with exactly the
upright or perform normal daily activities after return-
same acceleratory forces and in the same direction.
ing to the full gravity of Earth. Astronauts returning
For this reason, the person simply is not attracted
from space flights lasting 4 to 6 months are also sus-
toward any specific wall of the spacecraft.
ceptible to bone fractures and may require several
weeks before they return to pre-flight cardiovascular,
Physiologic Problems of Weightlessness (Microgravity). The
bone, and muscle fitness. As space flights become
physiologic problems of weightlessness have not
longer in preparation for possible human exploration
proved to be of much significance, as long as the period
of other planets, such as Mars, the effects of prolonged
of weightlessness is not too long. Most of the problems
microgravity could pose a very serious threat to astro-
that do occur are related to three effects of the weight-
nauts after they land, especially in the event of an
lessness: (1) motion sickness during the first few days
emergency landing. Therefore, considerable research
of travel, (2) translocation of fluids within the body
effort has been directed toward developing counter-
because of failure of gravity to cause normal hydro-
measures, in addition to exercise, that can prevent or
static pressures, and (3) diminished physical activity
more effectively attenuate these changes. One such
because no strength of muscle contraction is required
countermeasure that is being tested is the application
to oppose the force of gravity.
of intermittent
“artificial gravity” caused by short
Almost 50 per cent of astronauts experience motion
periods (e.g., 1 hour each day) of centrifugal accelera-
sickness, with nausea and sometimes vomiting, during
tion of the astronauts while they sit in specially
the first 2 to 5 days of space travel. This probably
designed short-arm centrifuges that create forces of up
results from an unfamiliar pattern of motion signals
to 2 to 3 G.
arriving in the equilibrium centers of the brain, and at
the same time lack of gravitational signals.
The observed effects of prolonged stay in space
References
are the following: (1) decrease in blood volume, (2)
decrease in red blood cell mass, (3) decrease in muscle
Adams GR, Caiozzo VJ, Baldwin KM: Skeletal muscle
strength and work capacity, (4) decrease in maximum
unweighting: spaceflight and ground-based models. J Appl
cardiac output, and (5) loss of calcium and phosphate
Physiol 95:2185, 2003.
from the bones, as well as loss of bone mass. Most of
Alfrey CP, Udden MM, Leach-Huntoon C, et al: Control of
these same effects also occur in people who lie in bed
red blood cell mass in spaceflight. J Appl Physiol 81:98,
for an extended period of time. For this reason, exer-
1996.
cise programs are carried out by astronauts during
Basnyat B, Murdoch DR: High-altitude illness. Lancet
361:1967, 2003.
prolonged space missions.
Convertino VA: Mechanisms of microgravity induced ortho-
In previous space laboratory expeditions in which
static intolerance: implications for effective countermea-
the exercise program had been less vigorous, the astro-
sures. J Gravit Physiol 9:1, 2002.
nauts had severely decreased work capacities for the
Eckberg DL: Bursting into space: alterations of sympathetic
first few days after returning to earth. They also had a
control by space travel. Acta Physiol Scand 177:299, 2003.
tendency to faint (and still do, to some extent) when
Hackett PH, Roach RC: High-altitude illness. N Engl J Med
they stood up during the first day or so after return to
345:107, 2001.
gravity because of diminished blood volume and
Harm DL, Jennings RT, Meck JV, et al: Gender issues related
diminished responses of the arterial pressure control
to spaceflight: a NASA perspective. J Appl Physiol
mechanisms.
91:2374, 2001.
Hochachka PW, Beatty CL, Burelle Y, et al: The lactate
paradox in human high-altitude physiological perform-
Cardiovascular, Muscle, and Bone “Deconditioning” During
ance. News Physiol Sci 17:122, 2002.
Prolonged Exposure to Weightlessness. During very long
Hoschele S, Mairbaurl H: Alveolar flooding at high altitude:
space flights and prolonged exposure to microgravity,
failure of reabsorption? News Physiol Sci 18:55, 2003.
gradual “deconditioning” effects occur on the cardio-
Hultgren HN: High-altitude pulmonary edema: current con-
vascular system, skeletal muscles, and bone despite rig-
cepts. Annu Rev Med 47:267, 1996.
orous exercise during the flight. Studies of astronauts
Rupert JL, Hochachka PW: Genetic approaches to under-
on space flights lasting several months have shown that
standing human adaptation to altitude in the Andes. J Exp
they may lose as much 1.0 percent of their bone mass
Biol 204(Pt 18):3151, 2001.
each month even though they continue to exercise.
Smith SM, Heer M: Calcium and bone metabolism during
space flight. Nutrition 18:849, 2002.
Substantial atrophy of cardiac and skeletal muscles
West JB: Climbing Mount Everest without oxygen. News
also occurs during prolonged exposure to a micro-
Physiol Sci 1:25, 1986.
gravity environment.
West JB: Man in space. News Physiol Sci 1:198, 1986.
One of the most serious effects is cardiovascular
West JB: High Life—History of High-Altitude Physiology
“deconditioning”, which includes decreased work
and Medicine. Bethesda, MD: American Physiological
capacity, reduced blood volume, impaired barorecep-
Society, 1998.
tor reflexes, and reduced orthostatic tolerance. These
Zhang LF: Vascular adaptation to microgravity: what have
changes greatly limit the astronauts’ ability to stand
we learned? J Appl Physiol 91:2415, 2001.
C H A P T E R
4
4
Physiology of Deep-Sea Diving
and Other Hyperbaric Conditions
When human beings descend beneath the sea, the
pressure around them increases tremendously. To
keep the lungs from collapsing, air must be supplied
at very high pressure to keep them inflated. This
exposes the blood in the lungs also to extremely
high alveolar gas pressure, a condition called hyper-
barism. Beyond certain limits, these high pressures
can cause tremendous alterations in body physiol-
ogy and can be lethal.
Relationship of Pressure to Sea Depth. A column of seawater 33 feet deep exerts the
same pressure at its bottom as the pressure of the atmosphere above the sea.
Therefore, a person 33 feet beneath the ocean surface is exposed to 2 atmos-
pheres pressure, 1 atmosphere of pressure caused by the weight of the air above
the water and the second atmosphere by the weight of the water itself. At 66
feet the pressure is 3 atmospheres, and so forth, in accord with the table in
Figure 44-1.
Effect of Sea Depth on the Volume of Gases-Boyle’s Law. Another important effect of
depth is compression of gases to smaller and smaller volumes. The lower part
of Figure 44-1 shows a bell jar at sea level containing 1 liter of air. At 33 feet
beneath the sea, where the pressure is 2 atmospheres, the volume has been com-
pressed to only one-half liter, and at 8 atmospheres (233 feet) to one-eighth
liter. Thus, the volume to which a given quantity of gas is compressed is inversely
proportional to the pressure. This is a principle of physics called Boyle’s law,
which is extremely important in diving physiology because increased pressure
can collapse the air chambers of the diver’s body, especially the lungs, and often
causes serious damage.
Many times in this chapter it is necessary to refer to actual volume versus
sea-level volume. For instance, we might speak of an actual volume of 1 liter at
a depth of 300 feet; this is the same quantity of air as a sea-level volume of
10 liters.
Effect of High Partial Pressures of Individual
Gases on the Body
The individual gases to which a diver is exposed when breathing air are nitro-
gen, oxygen, and carbon dioxide; each of these at times can cause significant
physiologic effects at high pressures.
Nitrogen Narcosis at High Nitrogen Pressures
About four fifths of the air is nitrogen. At sea-level pressure, the nitrogen has
no significant effect on bodily function, but at high pressures it can cause varying
degrees of narcosis. When the diver remains beneath the sea for an hour or
more and is breathing compressed air, the depth at which the first symptoms of
mild narcosis appear is about 120 feet. At this level the diver begins to exhibit
545
546
Unit VIII
Aviation, Space, and Deep-Sea Diving Physiology
Depth (feet)
Atmosphere(s)
30
Sea level
1
33
2
A
66
3
100
4
25
133
5
166
6
B
200
7
300
10
20
400
13
Oxygen-hemoglobin dissociation curve
500
16
1 liter
Total O2 in blood
15
Sea level
Combined with
Oxygen
hemoglobin
poisoning
Dissolved in
1/2 liter
10
water of blood
33 ft
Normal alveolar
oxygen pressure
5
0
1/4 liter
100 ft
0
760
1560
2280
3040
Oxygen partial pressure in lungs (mm Hg)
Figure 44-2
Quantity of oxygen dissolved in the fluid of the blood and in com-
bination with hemoglobin at very high PO2s.
Oxygen Toxicity at High Pressures
Effect of Very High PO2 on Blood Oxygen Transport. When the
233 ft
1/8 liter
Po2 in the blood rises above 100 mm Hg, the amount
of oxygen dissolved in the water of the blood increases
markedly. This is shown in Figure 44-2, which depicts
the same oxygen-hemoglobin dissociation curve as
that shown in Chapter 40 but with the alveolar Po2
Figure 44-1
extended to more than 3000 mm Hg. Also depicted by
the lowest curve in the figure is the volume of oxygen
Effect of sea depth on pressure (top table) and on gas volume
(bottom).
dissolved in the fluid of the blood at each Po2 level.
Note that in the normal range of alveolar Po2 (below
120 mm Hg), almost none of the total oxygen in the
joviality and to lose many of his or her cares. At 150
blood is accounted for by dissolved oxygen, but as the
to 200 feet, the diver becomes drowsy. At 200 to 250
oxygen pressure rises into the thousands of millime-
feet, his or her strength wanes considerably, and the
ters of mercury, a large portion of the total oxygen is
diver often becomes too clumsy to perform the work
then dissolved in the water of the blood, in addition to
required. Beyond 250 feet (8.5 atmospheres pressure),
that bound with hemoglobin.
the diver usually becomes almost useless as a result of
nitrogen narcosis if he or she remains at these depths
Effect of High Alveolar PO2 on Tissue PO2. Let us assume that
too long.
the Po2 in the lungs is about 3000 mm Hg (4 atmos-
Nitrogen narcosis has characteristics similar to those
pheres pressure). Referring to Figure 44-2, one finds
of alcohol intoxication, and for this reason it has
that this represents a total oxygen content in each 100
frequently been called “raptures of the depths.” The
milliliters of blood of about 29 volumes per cent, as
mechanism of the narcotic effect is believed to be the
demonstrated by point A in the figure—this means
same as that of most other gas anesthetics. That is, it
20 volumes per cent bound with hemoglobin and 9
dissolves in the fatty substances in neuronal mem-
volumes per cent dissolved in the blood water. As this
branes and, because of its physical effect on altering
blood passes through the tissue capillaries and the
ionic conductance through the membranes, reduces
tissues use their normal amount of oxygen, about
neuronal excitability.
5 milliliters from each 100 milliliters of blood, the
Chapter 44
Physiology of Deep-Sea Diving and Other Hyperbaric Conditions
547
oxygen content on leaving the tissue capillaries is still
Therefore, most of the acute lethal effects of acute
24 volumes per cent (point B in the figure). At this
oxygen toxicity are caused by brain dysfunction.
point, the Po2 is approximately 1200 mm Hg, which
means that oxygen is delivered to the tissues at this
Chronic Oxygen Poisoning Causes Pulmonary Disabil-
extremely high pressure instead of at the normal value
ity. A person can be exposed to only 1 atmosphere
of 40 mm Hg. Thus, once the alveolar Po2 rises above
pressure of oxygen almost indefinitely without devel-
a critical level, the hemoglobin-oxygen buffer mecha-
oping the acute oxygen toxicity of the nervous system
nism (discussed in Chapter 40) is no longer capable
just described. However, after only about 12 hours of
of keeping the tissue Po2 in the normal, safe range
1 atmosphere oxygen exposure, lung passageway con-
between 20 and 60 mm Hg.
gestion, pulmonary edema, and atelectasis caused by
damage to the linings of the bronchi and alveoli begin
Acute Oxygen Poisoning. The extremely high tissue Po2
to develop. The reason for this effect in the lungs but
that occurs when oxygen is breathed at very high alve-
not in other tissues is that the air spaces of the lungs
olar oxygen pressure can be detrimental to many of
are directly exposed to the high oxygen pressure, but
the body’s tissues. For instance, breathing oxygen at
oxygen is delivered to the other body tissues at almost
4 atmospheres pressure of oxygen (Po2 = 3040 mm Hg)
normal Po2 because of the hemoglobin-oxygen buffer
will cause brain seizures followed by coma in most
system.
people within 30 to 60 minutes. The seizures often
occur without warning and, for obvious reasons, are
likely to be lethal to divers submerged beneath the
Carbon Dioxide Toxicity at Great
sea.
Depths in the Sea
Other symptoms encountered in acute oxygen poi-
soning include nausea, muscle twitchings, dizziness,
If the diving gear is properly designed and functions
disturbances of vision, irritability, and disorientation.
properly, the diver has no problem due to carbon
Exercise greatly increases the diver’s susceptibility to
dioxide toxicity because depth alone does not increase
oxygen toxicity, causing symptoms to appear much
the carbon dioxide partial pressure in the alveoli. This
earlier and with far greater severity than in the resting
is true because depth does not increase the rate of
person.
carbon dioxide production in the body, and as long as
the diver continues to breathe a normal tidal volume
Excessive Intracellular Oxidation as a Cause of
and expires the carbon dioxide as it is formed, alveo-
Nervous System Oxygen Toxicity—“Oxidizing Free
lar carbon dioxide pressure will be maintained at a
Radicals.” Molecular oxygen (O2) has little capability
normal value.
of oxidizing other chemical compounds. Instead, it
In certain types of diving gear, however, such as the
must first be converted into an “active” form of
diving helmet and some types of rebreathing appara-
oxygen. There are several forms of active oxygen
tuses, carbon dioxide can build up in the dead space
called oxygen free radicals. One of the most important
air of the apparatus and be rebreathed by the diver.
of these is the superoxide free radical O2_, and another
Up to an alveolar carbon dioxide pressure (Pco2) of
is the peroxide radical in the form of hydrogen perox-
about 80 mm Hg, twice that in normal alveoli, the
ide. Even when the tissue Po2 is normal at the level of
diver usually tolerates this buildup by increasing the
40 mm Hg, small amounts of free radicals are contin-
minute respiratory volume a maximum of 8- to 11-fold
ually being formed from the dissolved molecular
to compensate for the increased carbon dioxide.
oxygen. Fortunately, the tissues also contain multiple
Beyond
80-mm Hg alveolar Pco2, the situation
enzymes that rapidly remove these free radicals,
becomes intolerable, and eventually the respiratory
including peroxidases, catalases, and superoxide dis-
center begins to be depressed, rather than excited,
mutases. Therefore, so long as the hemoglobin-oxygen
because of the negative tissue metabolic effects of high
buffering mechanism maintains a normal tissue Po2,
Pco2. The diver’s respiration then begins to fail rather
the oxidizing free radicals are removed rapidly enough
than to compensate. In addition, the diver develops
that they have little or no effect in the tissues.
severe respiratory acidosis, and varying degrees of
Above a critical alveolar Po2
(above about
2
lethargy, narcosis, and finally even anesthesia, as dis-
atmospheres Po2), the hemoglobin-oxygen buffering
cussed in Chapter 42.
mechanism fails, and the tissue Po2 can then rise to
hundreds or thousands of millimeters of mercury. At
these high levels, the amounts of oxidizing free radi-
Decompression of the Diver After
cals literally swamp the enzyme systems designed to
Excess Exposure to High Pressure
remove them, and now they can have serious destruc-
tive and even lethal effects on the cells. One of the
When a person breathes air under high pressure for a
principal effects is to oxidize the polyunsaturated fatty
long time, the amount of nitrogen dissolved in the
acids that are essential components of many of the cell
body fluids increases. The reason for this is the fol-
membranes. Another effect is to oxidize some of the
lowing: Blood flowing through the pulmonary capil-
cellular enzymes, thus damaging severely the cellular
laries becomes saturated with nitrogen to the same
metabolic systems. The nervous tissues are especially
high pressure as that in the alveolar breathing mixture.
susceptible because of their high lipid content.
And over several more hours, enough nitrogen is
548
Unit VIII
Aviation, Space, and Deep-Sea Diving Physiology
carried to all the tissues of the body to raise their tissue
Pressure Outside Body
Pn2 also to equal the Pn2 in the breathing air.
Before
After sudden
Because nitrogen is not metabolized by the body, it
decompression
decompression
remains dissolved in all the body tissues until the nitro-
O2 = 1044 mm Hg
O2 = 159 mm Hg
gen pressure in the lungs is decreased back to some
N2 = 3956
N2 = 601
lower level, at which time the nitrogen can be removed
Total = 5000 mm Hg
Total = 760 mm Hg
by the reverse respiratory process; however, this
removal often takes hours to occur and is the source
of multiple problems collectively called decompression
sickness.
Volume of Nitrogen Dissolved in the Body Fluids at Different
Depths. At sea level, almost exactly 1 liter of nitrogen
is dissolved in the entire body. Slightly less than one
half of this is dissolved in the water of the body and a
Body
Body
little more than one half in the fat of the body. This is
Gaseous pressure
Gaseous pressure
in the body fluids
in the body fluids
true because nitrogen is five times as soluble in fat as
H2O = 47 mm Hg
H2O = 47 mm Hg
in water.
CO2 = 40
CO2 = 40
After the diver has become saturated with nitrogen,
O2
= 60
O2
= 60
the sea-level volume of nitrogen dissolved in the body
N2
= 3918
N2
= 3918
at different depths is as follows:
Total = 4065
Total = 4065
A
B
Feet
Liters
Figure 44-3
0
1
Gaseous pressures both inside and outside the body, showing (A)
33
2
saturation of the body to high gas pressures when breathing air
100
4
at a total pressure of 5000 mm Hg, and (B) the great excesses of
200
7
intra-body pressures that are responsible for bubble formation in
300
10
the tissues when the lung intra-alveolar pressure body is
suddenly returned from
5000 mm Hg to normal pressure of
760 mm Hg.
Several hours are required for the gas pressures of
nitrogen in all the body tissues to come nearly to equi-
librium with the gas pressure of nitrogen in the alveoli.
nitrogen pressure
(Pn2
= 3918 mm Hg), about 6.5
The reason for this is that the blood does not flow
times the normal amount of nitrogen in the tissues.
rapidly enough and the nitrogen does not diffuse
As long as the diver remains deep beneath the sea, the
rapidly enough to cause instantaneous equilibrium.
pressure against the outside of his or her body
The nitrogen dissolved in the water of the body comes
(5000 mm Hg) compresses all the body tissues suffi-
to almost complete equilibrium in less than 1 hour, but
ciently to keep the excess nitrogen gas dissolved. But
the fat tissue, requiring five times as much transport of
when the diver suddenly rises to sea level (Figure
nitrogen and having a relatively poor blood supply,
44-3B), the pressure on the outside of the body
reaches equilibrium only after several hours. For this
becomes only 1 atmosphere (760 mm Hg), while the
reason, if a person remains at deep levels for only a
gas pressure inside the body fluids is the sum of the
few minutes, not much nitrogen dissolves in the body
pressures of water vapor, carbon dioxide, oxygen, and
fluids and tissues, whereas if the person remains at a
nitrogen, or a total of 4065 mm Hg, 97 per cent of
deep level for several hours, both the body water and
which is caused by the nitrogen. Obviously, this total
body fat become saturated with nitrogen.
value of 4065 mm Hg is far greater than the 760 mm
Hg pressure on the outside of the body. Therefore, the
Decompression Sickness (Synonyms: Bends, Compressed Air
gases can escape from the dissolved state and form
Sickness, Caisson Disease, Diver’s Paralysis, Dysbarism).
If a
actual bubbles, composed almost entirely of nitrogen,
diver has been beneath the sea long enough that large
both in the tissues and in the blood where they plug
amounts of nitrogen have dissolved in his or her body
many small blood vessels. The bubbles may not appear
and the diver then suddenly comes back to the surface
for many minutes to hours, because sometimes the
of the sea, significant quantities of nitrogen bubbles
gases can remain dissolved in the “supersaturated”
can develop in the body fluids either intracellularly or
state for hours before bubbling.
extracellularly and can cause minor or serious damage
in almost any area of the body, depending on the
Symptoms of Decompression Sickness
(“Bends”).
number and sizes of bubbles formed; this is called
The symptoms of decompression sickness are caused
decompression sickness.
by gas bubbles blocking many blood vessels in
The principles underlying bubble formation are
different tissues. At first, only the smallest vessels
shown in Figure 44-3. In Figure 44-3A, the diver’s
are blocked by minute bubbles, but as the bubbles
tissues have become equilibrated to a high dissolved
coalesce, progressively larger vessels are affected.
Chapter 44
Physiology of Deep-Sea Diving and Other Hyperbaric Conditions
549
Tissue ischemia and sometimes tissue death are the
level near that at which they will be working. This
result.
keeps the tissues and fluids of the body saturated with
In most people with decompression sickness, the
the gases to which they will be exposed while diving.
symptoms are pain in the joints and muscles of the legs
Then, when they return to the same tank after
and arms, affecting 85 to 90 per cent of those persons
working, there are no significant changes in pressure,
who develop decompression sickness. The joint pain
so that decompression bubbles do not occur.
accounts for the term “bends” that is often applied to
In very deep dives, especially during saturation
this condition.
diving, helium is usually used in the gas mixture
In 5 to 10 per cent of people with decompression
instead of nitrogen for three reasons: (1) it has only
sickness, nervous system symptoms occur, ranging
about one fifth the narcotic effect of nitrogen; (2) only
from dizziness in about 5 per cent to paralysis or col-
about one half as much volume of helium dissolves in
lapse and unconsciousness in as many as 3 per cent.
the body tissues as nitrogen, and the volume that does
The paralysis may be temporary, but in some instances,
dissolve diffuses out of the tissues during decompres-
damage is permanent.
sion several times as rapidly as does nitrogen, thus
Finally, about 2 per cent of people with decompres-
reducing the problem of decompression sickness; and
sion sickness develop “the chokes,” caused by massive
(3) the low density of helium (one seventh the density
numbers of microbubbles plugging the capillaries of
of nitrogen) keeps the airway resistance for breathing
the lungs; this is characterized by serious shortness of
at a minimum, which is very important because highly
breath, often followed by severe pulmonary edema
compressed nitrogen is so dense that airway resistance
and, occasionally, death.
can become extreme, sometimes making the work of
breathing beyond endurance.
Nitrogen Elimination from the Body; Decompression Tables. If
Finally, in very deep dives it is important to reduce
a diver is brought to the surface slowly, enough of the
the oxygen concentration in the gaseous mixture
dissolved nitrogen can usually be eliminated by expi-
because otherwise oxygen toxicity would result. For
ration through the lungs to prevent decompression
instance, at a depth of 700 feet (22 atmospheres of
sickness. About two thirds of the total nitrogen is lib-
pressure), a 1 per cent oxygen mixture will provide all
erated in 1 hour and about 90 per cent in 6 hours.
the oxygen required by the diver, whereas a 21 per
Decompression tables have been prepared by the
cent mixture of oxygen (the percentage in air) deliv-
U.S. Navy that detail procedures for safe decompres-
ers a Po2 to the lungs of more than 4 atmospheres,
sion. To give the student an idea of the decompression
a level very likely to cause seizures in as little as
process, a diver who has been breathing air and has
30 minutes.
been on the sea bottom for 60 minutes at a depth of
190 feet is decompressed according to the following
Scuba (Self-Contained
schedule:
Underwater Breathing
10 minutes at 50 feet depth
17 minutes at 40 feet depth
Apparatus) Diving
19 minutes at 30 feet depth
50 minutes at 20 feet depth
Before the 1940s, almost all diving was done using a
84 minutes at 10 feet depth
diving helmet connected to a hose through which air
Thus, for a work period on the bottom of only
was pumped to the diver from the surface. Then, in
1 hour, the total time for decompression is about
1943, Jacques Cousteau popularized a self-contained
3 hours.
underwater breathing apparatus, known as the SCUBA
apparatus. The type of SCUBA apparatus used in
Tank Decompression and Treatment of Decompression Sick-
more than 99 per cent of all sports and commercial
ness. Another procedure widely used for decompres-
diving is the open-circuit demand system shown in
sion of professional divers is to put the diver into a
Figure 44-4. This system consists of the following com-
pressurized tank and then to lower the pressure grad-
ponents: (1) one or more tanks of compressed air or
ually back to normal atmospheric pressure, using
some other breathing mixture, (2) a first-stage “reduc-
essentially the same time schedule as noted above.
ing” valve for reducing the very high pressure from the
Tank decompression is even more important for
tanks to a low pressure level, (3) a combination inhala-
treating people in whom symptoms of decompression
tion “demand” valve and exhalation valve that allows
sickness develop minutes or even hours after they
air to be pulled into the lungs with slight negative pres-
have returned to the surface. In this case, the diver
sure of breathing and then to be exhaled into the sea
is recompressed immediately to a deep level. Then
at a pressure level slightly positive to the surrounding
decompression is carried out over a period several
water pressure, and (4) a mask and tube system with
times as long as the usual decompression period.
small “dead space.”
The demand system operates as follows: The first-
“Saturation Diving” and Use of Helium-Oxygen Mixtures in Deep
stage reducing valve reduces the pressure from the
Dives. When divers must work at very deep levels—
tanks so that the air delivered to the mask has a pres-
between 250 feet and nearly 1000 feet—they fre-
sure only a few mm Hg greater than the surrounding
quently live in a large compression tank for days or
water pressure. The breathing mixture does not
weeks at a time, remaining compressed at a pressure
flow continually into the mask. Instead, with each
550
Unit VIII
Aviation, Space, and Deep-Sea Diving Physiology
devices, especially when using helium, theoretically
can allow escape from as deep as 600 feet or perhaps
more.
One of the major problems of escape is prevention
Mask
of air embolism. As the person ascends, the gases in
the lungs expand and sometimes rupture a pulmonary
Hose
blood vessel, forcing the gases to enter the vessel and
cause air embolism of the circulation. Therefore, as the
person ascends, he or she must make a special effort
Demand valve
to exhale continually.
First stage
valve
Health Problems in the Submarine Internal Environment.
Except for escape, submarine medicine generally
centers around several engineering problems to keep
hazards out of the internal environment. First, in
atomic submarines, there exists the problem of ra-
diation hazards, but with appropriate shielding, the
amount of radiation received by the crew submerged
beneath the sea has been less than normal radiation
received above the surface of the sea from cosmic rays.
Second, poisonous gases on occasion escape into the
atmosphere of the submarine and must be controlled
rapidly. For instance, during several weeks’ submer-
gence, cigarette smoking by the crew can liberate
enough carbon monoxide, if not removed rapidly,
Air cylinders
to cause carbon monoxide poisoning. And, on occa-
sion, even freon gas has been found to diffuse out of
refrigeration systems in sufficient quantity to cause
toxicity.
Figure 44-4
Open-circuit demand type of SCUBA apparatus.
Hyperbaric Oxygen Therapy
The intense oxidizing properties of high-pressure
inspiration, slight extra negative pressure in the
oxygen
(hyperbaric oxygen) can have valuable
demand valve of the mask pulls the diaphragm of the
therapeutic effects in several important clinical
valve open, and this automatically releases air from
conditions. Therefore, large pressure tanks are now
the tank into the mask and lungs. In this way, only the
available in many medical centers into which patients
amount of air needed for inhalation enters the mask.
can be placed and treated with hyperbaric oxygen.
Then, on expiration, the air cannot go back into the
The oxygen is usually administered at Po2s of 2 to
tank but instead is expired into the sea.
3 atmospheres of pressure through a mask or intra-
The most important problem in use of the self-
tracheal tube, whereas the gas around the body is
contained underwater breathing apparatus is the
normal air compressed to the same high-pressure
limited amount of time one can remain beneath the
level.
sea surface; for instance, only a few minutes are possi-
It is believed that the same oxidizing free radicals
ble at a 200-foot depth. The reason for this is that
responsible for oxygen toxicity are also responsible for
tremendous airflow from the tanks is required to wash
at least some of the therapeutic benefits. Some of the
carbon dioxide out of the lungs—the greater the
conditions in which hyperbaric oxygen therapy has
depth, the greater the airflow in terms of quantity of
been especially beneficial follow.
air per minute that is required, because the volumes
Probably the most successful use of hyperbaric
have been compressed to small sizes.
oxygen has been for treatment of gas gangrene. The
bacteria that cause this condition, clostridial organ-
isms, grow best under anaerobic conditions and
Special Physiologic Problems
stop growing at oxygen pressures greater than about
in Submarines
70 mm Hg. Therefore, hyperbaric oxygenation of the
tissues can frequently stop the infectious process
Escape from Submarines. Essentially the same problems
entirely and thus convert a condition that formerly was
encountered in deep-sea diving are often met in rela-
almost 100 per cent fatal into one that is cured in most
tion to submarines, especially when it is necessary
instances by early treatment with hyperbaric therapy.
to escape from a submerged submarine. Escape is
Other conditions in which hyperbaric oxygen
possible from as deep as 300 feet without using any
therapy has been either valuable or possibly valuable
apparatus. However, proper use of rebreathing
include decompression sickness, arterial gas embolism,
Chapter 44
Physiology of Deep-Sea Diving and Other Hyperbaric Conditions
551
carbon monoxide poisoning, osteomyelitis, and
Nilsson GE: Surviving anoxia with the brain turned on. News
myocardial infarction.
Physiol Sci 16:217, 2001.
Russi EW: Diving and the risk of barotrauma. Thorax
53(Suppl 2):S20, 1998.
References
Wang C, Schwaitzberg S, Berliner E, et al: Hyperbaric
oxygen for treating wounds: a systematic review of the
Butler PJ: Diving beyond the limits. News Physiol Sci 16:222,
literature. Arch Surg 138:272, 2003.
2001.
Wang J, Li F, Calhoun JH, Mader JT: The role and effective-
Kooyman GL, Ponganis PJ: The physiological basis of diving
ness of adjunctive hyperbaric oxygen therapy in the man-
to depth: birds and mammals. Annu Rev Physiol 60:19,
agement of musculoskeletal disorders. J Postgrad Med
1998.
48:226, 2002
Leach RM, Rees PJ, Wilmshurst P: Hyperbaric oxygen
West JB, Fu Z, Gaeth AP, Short RV: Fetal lung development
therapy. BMJ 317:1140, 1998.
in the elephant reflects the adaptations required for snor-
Neuman TS: Arterial gas embolism and decompression sick-
keling in adult life. Respir Physiol Neurobiol 138:325,
ness. News Physiol Sci 17:77, 2002.
2003.
U N I T IX
The Nervous
System: A. General
Principles and
Sensory Physiology
45. Organization of the Nervous System, Basic
Functions of Synapses, “Transmitter Substances”
46. Sensory Receptors, Neuronal Circuits for
Processing Information
47. Somatic Sensations: I. General Organization,
the Tactile and Position Senses
48. Somatic Sensations: II. Pain, Headache,
and Thermal Sensations
C H A P T E R
4
5
Organization of the Nervous
System, Basic Functions of
Synapses, “Transmitter Substances”
The nervous system is unique in the vast complex-
ity of thought processes and control actions it can
perform. It receives each minute literally millions of
bits of information from the different sensory
nerves and sensory organs and then integrates all
these to determine responses to be made by the
body.
However, before beginning this discussion of the
nervous system, the reader should review Chapters 5 and 7, which present the
principles of membrane potentials and transmission of signals in nerves and
through neuromuscular junctions.
General Design of the Nervous System
Central Nervous System Neuron: The Basic
Functional Unit
The central nervous system contains more than 100 billion neurons. Figure 45-1
shows a typical neuron of a type found in the brain motor cortex. Incoming
signals enter this neuron through synapses located mostly on the neuronal den-
drites, but also on the cell body. For different types of neurons, there may be
only a few hundred or as many as 200,000 such synaptic connections from input
fibers. Conversely, the output signal travels by way of a single axon leaving the
neuron. Then, this axon has many separate branches to other parts of the
nervous system or peripheral body.
A special feature of most synapses is that the signal normally passes only in
the forward direction (from the axon of a preceding neuron to dendrites on cell
membranes of subsequent neurons). This forces the signal to travel in required
directions for performing specific nervous functions.
Sensory Part of the Nervous System—Sensory Receptors
Most activities of the nervous system are initiated by sensory experience
exciting sensory receptors, whether visual receptors in the eyes, auditory
receptors in the ears, tactile receptors on the surface of the body, or other
kinds of receptors. This sensory experience can either cause immediate reaction
from the brain, or memory of the experience can be stored in the brain
for minutes, weeks, or years and determine bodily reactions at some future
date.
Figure 45-2 shows the somatic portion of the sensory system, which transmits
sensory information from the receptors of the entire body surface and from
some deep structures. This information enters the central nervous system
through peripheral nerves and is conducted immediately to multiple sensory
areas in (1) the spinal cord at all levels; (2) the reticular substance of the
medulla, pons, and mesencephalon of the brain; (3) the cerebellum; (4) the thal-
amus; and (5) areas of the cerebral cortex.
555
556
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Somesthetic areas
Motor cortex
Dendrites
Thalamus
Brain
Bulboreticular
Pons
formation
Cell body
Cerebellum
Medulla
Skin
Spinal cord
Pain, cold,
warmth (Free
nerve ending)
Pressure
(Pacinian corpuscle)
(Expanded tip
receptor)
Touch
(Meissner's corpuscle)
Muscle spindle
Axon
Golgi tendon
apparatus
Muscle
Kinesthetic receptor
Synapses
Joint
Spinal cord
Second-order
neurons
Figure 45-2
Somatosensory axis of the nervous system.
Figure 45-1
Structure of a large neuron in the brain, showing its important func-
trolling smooth muscles, glands, and other internal
tional parts.
(Redrawn from Guyton AC: Basic Neuroscience:
bodily systems; this is discussed in Chapter 60.
Anatomy and Physiology. Philadelphia: WB Saunders Co, 1987.)
Note in Figure 45-3 that the skeletal muscles can be
controlled from many levels of the central nervous
system, including (1) the spinal cord; (2) the reticular
substance of the medulla, pons, and mesencephalon;
Motor Part of the Nervous System—
(3) the basal ganglia; (4) the cerebellum; and (5) the
Effectors
motor cortex. Each of these areas plays its own spe-
cific role, the lower regions concerned primarily with
The most important eventual role of the nervous
automatic, instantaneous muscle responses to sensory
system is to control the various bodily activities. This
stimuli, and the higher regions with deliberate
is achieved by controlling (1) contraction of appropri-
complex muscle movements controlled by the thought
ate skeletal muscles throughout the body, (2) contrac-
processes of the brain.
tion of smooth muscle in the internal organs, and
(3) secretion of active chemical substances by both
Processing of Information—
exocrine and endocrine glands in many parts of the
body. These activities are collectively called motor
“Integrative” Function of the
functions of the nervous system, and the muscles and
Nervous System
glands are called effectors because they are the actual
anatomical structures that perform the functions dic-
One of the most important functions of the nervous
tated by the nerve signals.
system is to process incoming information in such a
Figure 45-3 shows the “skeletal” motor nerve axis of
way that appropriate mental and motor responses will
the nervous system for controlling skeletal muscle con-
occur. More than 99 per cent of all sensory informa-
traction. Operating parallel to this axis is another
tion is discarded by the brain as irrelevant and unim-
system, called the autonomic nervous system, for con-
portant. For instance, one is ordinarily unaware of the
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
557
Motor nerve
Motor
system can control synaptic transmission, sometimes
to muscles
area
opening the synapses for transmission and at other
times closing them. In addition, some postsynaptic
Caudate
neurons respond with large numbers of output
nucleus
impulses, and others respond with only a few. Thus, the
synapses perform a selective action, often blocking
weak signals while allowing strong signals to pass, but
at other times selecting and amplifying certain weak
signals, and often channeling these signals in many
directions rather than only one direction.
Thalamus
Storage of Information—Memory
Putamen
Globus pallidus
Only a small fraction of even the most important
Subthalamic nucleus
sensory information usually causes immediate motor
response. But much of the information is stored for
Bulboreticular formation
Cerebellum
future control of motor activities and for use in
the thinking processes. Most storage occurs in the
cerebral cortex, but even the basal regions of the
brain and the spinal cord can store small amounts of
information.
The storage of information is the process we call
memory, and this, too, is a function of the synapses.
Gamma motor fiber
That is, each time certain types of sensory signals pass
Alpha motor fiber
through sequences of synapses, these synapses become
more capable of transmitting the same type of signal
the next time, a process called facilitation. After the
sensory signals have passed through the synapses a
Muscle spindle
large number of times, the synapses become so facili-
tated that signals generated within the brain itself can
Figure 45-3
also cause transmission of impulses through the same
sequences of synapses, even when the sensory input is
Skeletal motor nerve axis of the nervous system.
not excited. This gives the person a perception of expe-
riencing the original sensations, although the percep-
tions are only memories of the sensations.
parts of the body that are in contact with clothing, as
We know little about the precise mechanisms by
well as of the seat pressure when sitting. Likewise,
which long-term facilitation of synapses occurs in the
attention is drawn only to an occasional object in one’s
memory process, but what is known about this and
field of vision, and even the perpetual noise of our sur-
other details of the sensory memory process are dis-
roundings is usually relegated to the subconscious.
cussed in Chapter 57.
But, when important sensory information excites
Once memories have been stored in the nervous
the mind, it is immediately channeled into proper inte-
system, they become part of the brain processing
grative and motor regions of the brain to cause desired
mechanism for future “thinking.” That is, the thinking
responses. This channeling and processing of informa-
processes of the brain compare new sensory experi-
tion is called the integrative function of the nervous
ences with stored memories; the memories then help
system. Thus, if a person places a hand on a hot stove,
to select the important new sensory information and
the desired instantaneous response is to lift the hand.
to channel this into appropriate memory storage areas
And other associated responses follow, such as moving
for future use or into motor areas to cause immediate
the entire body away from the stove, and perhaps even
bodily responses.
shouting with pain.
Major Levels of Central
Role of Synapses in Processing Information. The synapse
is the junction point from one neuron to the next.
Nervous System Function
Later in this chapter, we will discuss the details of
synaptic function. However, it is important to point out
The human nervous system has inherited special func-
here that synapses determine the directions that the
tional capabilities from each stage of human evolu-
nervous signals will spread through the nervous
tionary development. From this heritage, three major
system. Some synapses transmit signals from one
levels of the central nervous system have specific func-
neuron to the next with ease, whereas others transmit
tional characteristics: (1) the spinal cord level, (2) the
signals only with difficulty. Also, facilitatory and
lower brain or subcortical level, and (3) the higher
inhibitory signals from other areas in the nervous
brain or cortical level.
558
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Spinal Cord Level
system performs specific functions. But it is the cortex
that opens a world of stored information for use by the
We often think of the spinal cord as being only a
mind.
conduit for signals from the periphery of the body to
the brain, or in the opposite direction from the brain
Comparison of the
back to the body. This is far from the truth. Even after
the spinal cord has been cut in the high neck region,
Nervous System with
many highly organized spinal cord functions still
a Computer
occur. For instance, neuronal circuits in the cord can
When computers were first developed, it soon became
cause (1) walking movements, (2) reflexes that with-
apparent that these machines have many features in
draw portions of the body from painful objects, (3)
common with the nervous system. First, all computers
reflexes that stiffen the legs to support the body
have input circuits that are comparable to the sensory
against gravity, and (4) reflexes that control local blood
portion of the nervous system, and output circuits that
vessels, gastrointestinal movements, or urinary excre-
are comparable to the motor portion of the nervous
tion. In fact, the upper levels of the nervous system
system.
often operate not by sending signals directly to the
In simple computers, the output signals are controlled
periphery of the body but by sending signals to
directly by the input signals, operating in a manner
similar to that of simple reflexes of the spinal cord. In
the control centers of the cord, simply “commanding”
more complex computers, the output is determined both
the cord centers to perform their functions.
by input signals and by information that has already
been stored in memory in the computer, which is anal-
ogous to the more complex reflex and processing mech-
Lower Brain or Subcortical Level
anisms of our higher nervous system. Furthermore,
as computers become even more complex, it is neces-
Many, if not most, of what we call subconscious activ-
sary to add still another unit, called the central process-
ities of the body are controlled in the lower areas
ing unit, that determines the sequence of all operations.
of the brain—in the medulla, pons, mesencephalon,
This unit is analogous to the control mechanisms in our
hypothalamus, thalamus, cerebellum, and basal
brain that direct our attention first to one thought or
sensation or motor activity, then to another, and so
ganglia. For instance, subconscious control of arterial
forth, until complex sequences of thought or action take
pressure and respiration is achieved mainly in the
place.
medulla and pons. Control of equilibrium is a com-
Figure 45-4 is a simple block diagram of a computer.
bined function of the older portions of the cerebellum
Even a rapid study of this diagram demonstrates its sim-
and the reticular substance of the medulla, pons, and
ilarity to the nervous system. The fact that the basic
mesencephalon. Feeding reflexes, such as salivation
components of the general-purpose computer are anal-
and licking of the lips in response to the taste of food,
ogous to those of the human nervous system demon-
are controlled by areas in the medulla, pons, mesen-
strates that the brain is basically a computer that
cephalon, amygdala, and hypothalamus. And many
continuously collects sensory information and uses this
emotional patterns, such as anger, excitement, sexual
along with stored information to compute the daily
course of bodily activity.
response, reaction to pain, and reaction to pleasure,
can still occur after destruction of much of the cere-
bral cortex.
Higher Brain or Cortical Level
Problem
After the preceding account of the many nervous
system functions that occur at the cord and lower brain
Input
Output
Answer
levels, one may ask, what is left for the cerebral cortex
to do? The answer to this is complex, but it begins with
the fact that the cerebral cortex is an extremely large
memory storehouse. The cortex never functions alone
Procedure
Initial
Result of
but always in association with lower centers of the
for solution
data
operations
Information
nervous system.
storage
Without the cerebral cortex, the functions of the
lower brain centers are often imprecise. The vast store-
Central
Computational
house of cortical information usually converts these
processing unit
unit
functions to determinative and precise operations.
Finally, the cerebral cortex is essential for most of
our thought processes, but it cannot function by itself.
In fact, it is the lower brain centers, not the cortex,
Figure 45-4
that initiate wakefulness in the cerebral cortex, thus
opening its bank of memories to the thinking machin-
Block diagram of a general-purpose computer, showing the basic
ery of the brain. Thus, each portion of the nervous
components and their interrelations.
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
559
Central Nervous System
Think for a moment about the extreme importance
of the one-way conduction mechanism. It allows
Synapses
signals to be directed toward specific goals. Indeed, it
is this specific transmission of signals to discrete and
Every medical student is aware that information is
highly focused areas both within the nervous system
transmitted in the central nervous system mainly in the
and at the terminals of the peripheral nerves that
form of nerve action potentials, called simply “nerve
allows the nervous system to perform its myriad func-
impulses,” through a succession of neurons, one after
tions of sensation, motor control, memory, and many
another. However, in addition, each impulse (1) may
others.
be blocked in its transmission from one neuron to the
next, (2) may be changed from a single impulse into
repetitive impulses, or
(3) may be integrated with
Physiologic Anatomy of the Synapse
impulses from other neurons to cause highly intricate
patterns of impulses in successive neurons. All these
Figure 45-5 shows a typical anterior motor neuron in
functions can be classified as synaptic functions of
the anterior horn of the spinal cord. It is composed of
neurons.
three major parts: the soma, which is the main body of
the neuron; a single axon, which extends from the
soma into a peripheral nerve that leaves the spinal
Types of Synapses—Chemical
cord; and the dendrites, which are great numbers of
and Electrical
branching projections of the soma that extend as
much as 1 millimeter into the surrounding areas of the
There are two major types of synapses: (1) the chemi-
cord.
cal synapse and (2) the electrical synapse.
As many as 10,000 to 200,000 minute synaptic knobs
Almost all the synapses used for signal transmission
called presynaptic terminals lie on the surfaces of the
in the central nervous system of the human being are
dendrites and soma of the motor neuron, about 80 to
chemical synapses. In these, the first neuron secretes at
95 per cent of them on the dendrites and only 5 to 20
its nerve ending synapse a chemical substance called a
per cent on the soma. These presynaptic terminals are
neurotransmitter (or often called simply transmitter
the ends of nerve fibrils that originate from many
substance), and this transmitter in turn acts on recep-
other neurons. Later, it will become evident that many
tor proteins in the membrane of the next neuron to
excite the neuron, inhibit it, or modify its sensitivity in
some other way. More than 40 important transmitter
substances have been discovered thus far. Some of
the best known are acetylcholine, norepinephrine,
epinephrine, histamine, gamma-aminobutyric acid
(GABA), glycine, serotonin, and glutamate.
Electrical synapses, in contrast, are characterized by
direct open fluid channels that conduct electricity from
one cell to the next. Most of these consist of small
protein tubular structures called gap junctions that
Dendrites
allow free movement of ions from the interior of one
cell to the interior of the next. Such junctions were dis-
cussed in Chapter 4. Only a few examples of gap junc-
tions have been found in the central nervous system.
Axon
However, it is by way of gap junctions and other
similar junctions that action potentials are transmitted
from one smooth muscle fiber to the next in visceral
smooth muscle (Chapter 8) and from one cardiac
Soma
muscle cell to the next in cardiac muscle (Chapter 10).
“One-Way” Conduction at Chemical Synapses. Chemical
synapses have one exceedingly important characteris-
tic that makes them highly desirable for transmitting
most nervous system signals: they always transmit
the signals in one direction: that is, from the neuron
that secretes the transmitter substance, called the
presynaptic neuron, to the neuron on which the trans-
mitter acts, called the postsynaptic neuron. This is the
principle of one-way conduction at chemical synapses,
Figure 45-5
and it is quite different from conduction through elec-
trical synapses, which often transmit signals in either
Typical anterior motor neuron, showing presynaptic terminals on
direction.
the neuronal soma and dendrites. Note also the single axon.
560
Unit IX The Nervous System: A. General Principles and Sensory Physiology
of these presynaptic terminals are excitatory—that is,
which in turn supplies the energy for synethesizing
they secrete a transmitter substance that excites the
new transmitter substance.
postsynaptic neuron. But other presynaptic terminals
When an action potential spreads over a presynap-
are inhibitory—they secrete a transmitter substance
tic terminal, depolarization of its membrane causes
that inhibits the postsynaptic neuron.
a small number of vesicles to empty into the cleft.
Neurons in other parts of the cord and brain differ
The released transmitter in turn causes an immediate
from the anterior motor neuron in (1) the size of the
change in permeability characteristics of the postsy-
cell body; (2) the length, size, and number of dendrites,
naptic neuronal membrane, and this leads to excitation
ranging in length from almost zero to many centime-
or inhibition of the postsynaptic neuron, depending on
ters; (3) the length and size of the axon; and (4) the
the neuronal receptor characteristics.
number of presynaptic terminals, which may range
from only a few to as many as 200,000. These differ-
Mechanism by Which an Action Potential
ences make neurons in different parts of the nervous
Causes Transmitter Release from the
system react differently to incoming synaptic signals
Presynaptic Terminals—Role of Calcium Ions
and, therefore, perform many different functions.
The membrane of the presynaptic terminal is called
the presynaptic membrane. It contains large numbers
Presynaptic Terminals. Electron microscopic studies of
of voltage-gated calcium channels. When an action
the presynaptic terminals show that they have varied
potential depolarizes the presynaptic membrane,
anatomical forms, but most resemble small round or
these calcium channels open and allow large numbers
oval knobs and, therefore, are sometimes called termi-
of calcium ions to flow into the terminal. The quantity
nal knobs, boutons, end-feet, or synaptic knobs.
of transmitter substance that is then released from
Figure
45-6 illustrates the basic structure of a
the terminal into the synaptic cleft is directly related
synapse, showing a single presynaptic terminal on the
to the number of calcium ions that enter. The precise
membrane surface of a postsomatic neuron. The presy-
mechanism by which the calcium ions cause this
naptic terminal is separated from the postsynaptic
release is not known, but it is believed to be the
neuronal soma by a synaptic cleft having a width
following.
usually of 200 to 300 angstroms. The terminal has
When the calcium ions enter the presynaptic termi-
two internal structures important to the excitatory or
nal, it is believed that they bind with special protein
inhibitory function of the synapse: the transmitter vesi-
molecules on the inside surface of the presynaptic
cles and the mitochondria. The transmitter vesicles
membrane, called release sites. This binding in turn
contain the transmitter substance that, when released
causes the release sites to open through the mem-
into the synaptic cleft, either excites or inhibits the
brane, allowing a few transmitter vesicles to release
postsynaptic neuron—excites if the neuronal mem-
their transmitter into the cleft after each single action
brane contains excitatory receptors, inhibits if the
potential. For those vesicles that store the neurotrans-
membrane contains inhibitory receptors. The mito-
mitter acetylcholine, between 2000 and 10,000 mole-
chondria provide adenosine triphosphate
(ATP),
cules of acetylcholine are present in each vesicle, and
there are enough vesicles in the presynaptic terminal
to transmit from a few hundred to more than 10,000
action potentials.
Action of the Transmitter Substance
on the Postsynaptic Neuron—Function of
“Receptor Proteins”
The membrane of the postsynaptic neuron contains
Transmitter vesicles
large numbers of receptor proteins, also shown in
Figure 45-6. The molecules of these receptors have
two important components: (1) a binding component
Mitochondria
that protrudes outward from the membrane into the
synaptic cleft—here it binds the neurotransmitter
coming from the presynaptic terminal—and (2) an
Presynaptic
terminal
ionophore component that passes all the way through
the postsynaptic membrane to the interior of the post-
synaptic neuron. The ionophore in turn is one of two
Receptor
types: (1) an ion channel that allows passage of speci-
proteins
fied types of ions through the membrane or (2) a
Synaptic cleft
(200-300
“second messenger” activator that is not an ion channel
angstroms)
Soma of neuron
but instead is a molecule that protrudes into the cell
cytoplasm and activates one or more substances inside
the postsynaptic neuron. These substances in turn
Figure 45-6
serve as “second messengers” to increase or decrease
Physiologic anatomy of the synapse.
specific cellular functions.
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
561
Ion Channels. The ion channels in the postsynaptic neu-
the process of memory—require prolonged changes in
ronal membrane are usually of two types: (1) cation
neurons for seconds to months after the initial trans-
channels that most often allow sodium ions to pass
mitter substance is gone. The ion channels are not suit-
when opened, but sometimes allow potassium and/or
able for causing prolonged postsynaptic neuronal
calcium ions as well, and (2) anion channels that allow
changes because these channels close within millisec-
mainly chloride ions to pass but also minute quantities
onds after the transmitter substance is no longer
of other anions.
present. However, in many instances, prolonged post-
The cation channels that conduct sodium ions are
synaptic neuronal excitation or inhibition is achieved
lined with negative charges. These charges attract the
by activating a “second messenger” chemical system
positively charged sodium ions into the channel when
inside the postsynaptic neuronal cell itself, and then it
the channel diameter increases to a size larger than
is the second messenger that causes the prolonged
that of the hydrated sodium ion. But those same neg-
effect.
ative charges repel chloride ions and other anions and
There are several types of second messenger
prevent their passage.
systems. One of the most common types uses a group
For the anion channels, when the channel diameters
of proteins called G-proteins. Figure 45-7 shows in the
become large enough, chloride ions pass into the
upper left corner a membrane receptor protein. A
channels and on through to the opposite side, whereas
G-protein is attached to the portion of the receptor
sodium, potassium, and calcium cations are blocked,
that protrudes into the interior of the cell. The
mainly because their hydrated ions are too large to
G-protein in turn consists of three components: an
pass.
alpha (a) component that is the activator portion of
We will learn later that when cation channels open
the G-protein, and beta (b) and gamma (g) compo-
and allow positively charged sodium ions to enter, the
nents that are attached to the alpha component and
positive electrical charges of the sodium ions will in
also to the inside of the cell membrane adjacent to the
turn excite this neuron. Therefore, a transmitter sub-
receptor protein. On activation by a nerve impulse, the
stance that opens cation channels is called an excita-
alpha portion of the G-protein separates from the beta
tory transmitter. Conversely, opening anion channels
and gamma portions and then is free to move within
allows negative electrical charges to enter, which
the cytoplasm of the cell.
inhibits the neuron. Therefore, transmitter substances
Inside the cytoplasm, the separated alpha compo-
that open these channels are called inhibitory
nent performs one or more of multiple functions,
transmitters.
depending on the specific characteristic of each type
When a transmitter substance activates an ion
of neuron. Shown in Figure 45-7 are four changes that
channel, the channel usually opens within a fraction of
can occur. They are as follows:
a millisecond; when the transmitter substance is no
1. Opening specific ion channels through the
longer present, the channel closes equally rapidly. The
postsynaptic cell membrane. Shown in the upper
opening and closing of ion channels provide a means
right of the figure is a potassium channel that is
for very rapid control of postsynaptic neurons.
opened in response to the G-protein; this channel
often stays open for a prolonged time, in contrast
“Second Messenger” System in the Postsynaptic Neuron.
to rapid closure of directly activated ion channels
Many functions of the nervous system—for instance,
that do not use the second messenger system.
Transmitter substance
Receptor
Potassium
protein
channel
1
Figure 45-7
g
b
Membrane
Opens
enzyme
“Second messenger” system by
G-protein
channel
K+
which a transmitter substance from
a
2
an initial neuron can activate a
a
second neuron by first releasing
a
“G-protein” into the second
neuron’s cytoplasm. Four subse-
quent possible effects of the
G-protein are shown, including 1,
3
Activates one or
4
ATP
GTP
opening an ion channel in the
Activates gene
Activates
more intracellular
membrane of the second neuron;
transcription
enzymes
or
enzymes
2, activating an enzyme system in
cAMP
cGMP
the neuron’s membrane; 3, activat-
ing an intracellular enzyme system;
and/or 4, causing gene transcrip-
Specific cellular
Proteins and
tion in the second neuron.
chemical activators
structural changes
562
Unit IX The Nervous System: A. General Principles and Sensory Physiology
2. Activation of cyclic adenosine monophosphate
excitatory membrane receptors or decrease the
(cAMP) or cyclic guanosine monophosphate
number of inhibitory membrane receptors.
(cGMP) in the neuronal cell. Recall that either
cyclic AMP or cyclic GMP can activate highly
Inhibition
specific metabolic machinery in the neuron and,
1. Opening of chloride ion channels through the
therefore, can initiate any one of many chemical
postsynaptic neuronal membrane. This allows
results, including long-term changes in cell
rapid diffusion of negatively charged chloride ions
structure itself, which in turn alters long-term
from outside the postsynaptic neuron to the
excitability of the neuron.
inside, thereby carrying negative charges inward
3. Activation of one or more intracellular enzymes.
and increasing the negativity inside, which is
The G-protein can directly activate one or more
inhibitory.
intracellular enzymes. In turn the enzymes can
2. Increase in conductance of potassium ions out of
cause any one of many specific chemical functions
the neuron. This allows positive ions to diffuse to
in the cell.
the exterior, which causes increased negativity
4. Activation of gene transcription. This is one of the
inside the neuron; this is inhibitory.
most important effects of activation of the second
3. Activation of receptor enzymes that inhibit cellular
messenger systems because gene transcription
metabolic functions that increase the number of
can cause formation of new proteins within the
inhibitory synaptic receptors or decrease the
neuron, thereby changing its metabolic machinery
number of excitatory receptors.
or its structure. Indeed, it is well known that
structural changes of appropriately activated
neurons do occur, especially in long-term memory
Chemical Substances That Function
processes.
as Synaptic Transmitters
It is clear that activation of second messenger
systems within the neuron, whether they be of the
More than 50 chemical substances have been proved
G-protein type or of other types, is extremely impor-
or postulated to function as synaptic transmitters.
tant for changing the long-term response characteris-
Many of them are listed in Tables 45-1 and 45-2, which
tics of different neuronal pathways. We will return to
give two groups of synaptic transmitters. One group
this subject in more detail in Chapter 57 when we
comprises small-molecule, rapidly acting transmitters.
discuss memory functions of the nervous system.
The other is made up of a large number of neuropep-
tides of much larger molecular size that are usually
Excitatory or Inhibitory Receptors in the
much more slowly acting.
Postsynaptic Membrane
The small-molecule, rapidly acting transmitters are
Some postsynaptic receptors, when activated, cause
the ones that cause most acute responses of the
excitation of the postsynaptic neuron, and others cause
nervous system, such as transmission of sensory signals
inhibition. The importance of having inhibitory as well
to the brain and of motor signals back to the muscles.
as excitatory types of receptors is that this gives an
The neuropeptides, in contrast, usually cause more
additional dimension to nervous function, allowing
prolonged actions, such as long-term changes in
restraint of nervous action as well as excitation.
numbers of neuronal receptors, long-term opening or
The different molecular and membrane mechanisms
closure of certain ion channels, and possibly even long-
used by the different receptors to cause excitation or
term changes in numbers of synapses or sizes of
inhibition include the following.
synapses.
Excitation
1. Opening of sodium channels to allow large
numbers of positive electrical charges to flow to the
Table 45-1
interior of the postsynaptic cell. This raises the
Small-Molecule, Rapidly Acting Transmitters
intracellular membrane potential in the positive
direction up toward the threshold level for
Class I
excitation. It is by far the most widely used means
Acetylcholine
for causing excitation.
Class II: The Amines
2. Depressed conduction through chloride or
Norepinephrine
potassium channels, or both. This decreases the
Epinephrine
diffusion of negatively charged chloride ions to
Dopamine
Serotonin
the inside of the postsynaptic neuron or decreases
Histamine
the diffusion of positively charged potassium ions
Class III: Amino Acids
to the outside. In either instance, the effect is to
Gamma-aminobutyric acid (GABA)
make the internal membrane potential more
Glycine
Glutamate
positive than normal, which is excitatory.
Aspartate
3. Various changes in the internal metabolism of the
Class IV
postsynaptic neuron to excite cell activity or, in
Nitric oxide (NO)
some instances, to increase the number of
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
563
membrane still contains appropriate enzyme proteins
Table 45-2
or transport proteins required for synthesizing and/or
Neuropeptide, Slowly Acting Transmitters or Growth
concentrating new transmitter substance inside the
Factors
vesicle.
Acetylcholine is a typical small-molecule transmit-
Hypothalamic-releasing hormones
ter that obeys the principles of synthesis and release
Thyrotropin-releasing hormone
stated earlier. This transmitter substance is synthesized
Luteinizing hormone-releasing hormone
in the presynaptic terminal from acetyl coenzyme A
Somatostatin (growth hormone inhibitory factor)
Pituitary peptides
and choline in the presence of the enzyme choline
Adrenocorticotropic hormone (ACTH)
acetyltransferase. Then it is transported into its specific
b-Endorphin
vesicles. When the vesicles later release the acetyl-
a-Melanocyte-stimulating hormone
choline into the synaptic cleft during synaptic neu-
Prolactin
Luteinizing hormone
ronal signal transmission, the acetylcholine is rapidly
Thyrotropin
split again to acetate and choline by the enzyme
Growth hormone
cholinesterase, which is present in the proteoglycan
Vasopressin
reticulum that fills the space of the synaptic cleft. And
Oxytocin
then again, inside the presynaptic terminal, the vesi-
Peptides that act on gut and brain
Leucine enkephalin
cles are recycled; choline is actively transported back
Methionine enkephalin
into the terminal to be used again for synthesis of new
Substance P
acetylcholine.
Gastrin
Cholecystokinin
Vasoactive intestinal polypeptide (VIP)
Characteristics of Some of the More Important Small-Molecule
Nerve growth factor
Transmitters. The most important of the small-molecule
Brain-derived neurotropic factor
transmitters are the following.
Neurotensin
Acetylcholine is secreted by neurons in many areas
Insulin
of the nervous system but specifically by (1) the ter-
Glucagon
From other tissues
minals of the large pyramidal cells from the motor
Angiotensin II
cortex, (2) several different types of neurons in the
Bradykinin
basal ganglia, (3) the motor neurons that innervate the
Carnosine
skeletal muscles,
(4) the preganglionic neurons of
Sleep peptides
Calcitonin
the autonomic nervous system, (5) the postganglionic
neurons of the parasympathetic nervous system, and
(6) some of the postganglionic neurons of the sympa-
thetic nervous system. In most instances, acetylcholine
Small-Molecule, Rapidly Acting Transmitters
has an excitatory effect; however, it is known to have
In most cases, the small-molecule types of transmitters
inhibitory effects at some peripheral parasympathetic
are synthesized in the cytosol of the presynaptic ter-
nerve endings, such as inhibition of the heart by the
minal and are absorbed by means of active transport
vagus nerves.
into the many transmitter vesicles in the terminal.
Norepinephrine is secreted by the terminals of many
Then, each time an action potential reaches the presy-
neurons whose cell bodies are located in the brain
naptic terminal, a few vesicles at a time release their
stem and hypothalamus. Specifically, norepinephrine-
transmitter into the synaptic cleft. This usually occurs
secreting neurons located in the locus ceruleus in the
within a millisecond or less by the mechanism
pons send nerve fibers to widespread areas of the brain
described earlier. The subsequent action of the small-
to help control overall activity and mood of the mind,
molecule type of transmitter on the membrane recep-
such as increasing the level of wakefulness. In most of
tors of the postsynaptic neuron usually also occurs
these areas, norepinephrine probably activates excita-
within another millisecond or less. Most often the
tory receptors, but in a few areas, it activates inhibitory
effect is to increase or decrease conductance through
receptors instead. Norepinephrine is also secreted by
ion channels; an example is to increase sodium con-
most postganglionic neurons of the sympathetic
ductance, which causes excitation, or to increase potas-
nervous system, where it excites some organs but
sium or chloride conductance, which causes inhibition.
inhibits others.
Dopamine is secreted by neurons that originate in
Recycling of the Small-Molecule Types of Vesicles. The vesi-
the substantia nigra. The termination of these neurons
cles that store and release small-molecule transmitters
is mainly in the striatal region of the basal ganglia. The
are continually recycled and used over and over again.
effect of dopamine is usually inhibition.
After they fuse with the synaptic membrane and open
Glycine is secreted mainly at synapses in the
to release their transmitter substance, the vesicle mem-
spinal cord. It is believed to always act as an inhibitory
brane at first simply becomes part of the synaptic
transmitter.
membrane. However, within seconds to minutes, the
GABA (gamma-aminobutyric acid) is secreted by
vesicle portion of the membrane invaginates back to
nerve terminals in the spinal cord, cerebellum, basal
the inside of the presynaptic terminal and pinches
ganglia, and many areas of the cortex. It is believed
off to form a new vesicle. And the new vesicular
always to cause inhibition.
564
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Glutamate is secreted by the presynaptic terminals
as potent as the small-molecule transmitters. Another
in many of the sensory pathways entering the central
important characteristic of the neuropeptides is that
nervous system, as well as in many areas of the cere-
they often cause much more prolonged actions. Some
bral cortex. It probably always causes excitation.
of these actions include prolonged closure of calcium
Serotonin is secreted by nuclei that originate in the
channels, prolonged changes in the metabolic machin-
median raphe of the brain stem and project to many
ery of cells, prolonged changes in activation or deacti-
brain and spinal cord areas, especially to the dorsal
vation of specific genes in the cell nucleus, and/or
horns of the spinal cord and to the hypothalamus.
prolonged alterations in numbers of excitatory or
Serotonin acts as an inhibitor of pain pathways in the
inhibitory receptors. Some of these effects last for
cord, and an inhibitor action in the higher regions of
days, but others perhaps for months or years. Our
the nervous system is believed to help control the
knowledge of the functions of the neuropeptides is
mood of the person, perhaps even to cause sleep.
only beginning to develop.
Nitric oxide is especially secreted by nerve terminals
in areas of the brain responsible for long-term behav-
ior and for memory. Therefore, this transmitter system
Electrical Events During
might in the future explain some behavior and
Neuronal Excitation
memory functions that thus far have defied under-
standing. Nitric oxide is different from other small-
The electrical events in neuronal excitation have been
molecule transmitters in its mechanism of formation
studied especially in the large motor neurons of the
in the presynaptic terminal and in its actions on the
anterior horns of the spinal cord. Therefore, the events
postsynaptic neuron. It is not preformed and stored in
described in the next few sections pertain essentially
vesicles in the presynaptic terminal as are other trans-
to these neurons. Except for quantitative differences,
mitters. Instead, it is synthesized almost instantly as
they apply to most other neurons of the nervous
needed, and it then diffuses out of the presynaptic
system as well.
terminals over a period of seconds rather than being
released in vesicular packets. Next, it diffuses into
Resting Membrane Potential of the Neuronal Soma. Figure
postsynaptic neurons nearby. In the postsynaptic
45-8 shows the soma of a spinal motor neuron, indi-
neuron, it usually does not greatly alter the membrane
cating a resting membrane potential of about -65 mil-
potential but instead changes intracellular metabolic
livolts. This is somewhat less negative than the -90
functions that modify neuronal excitability for
millivolts found in large peripheral nerve fibers and in
seconds, minutes, or perhaps even longer.
skeletal muscle fibers; the lower voltage is important
because it allows both positive and negative control
Neuropeptides
of the degree of excitability of the neuron. That is,
The neuropeptides are an entirely different class of
decreasing the voltage to a less negative value makes
transmitters that are synthesized differently and
the membrane of the neuron more excitable, whereas
whose actions are usually slow and in other ways quite
increasing this voltage to a more negative value makes
different from those of the small-molecule transmit-
the neuron less excitable. This is the basis for the two
ters. The neuropeptides are not synthesized in the
cytosol of the presynaptic terminals. Instead, they are
synthesized as integral parts of large-protein mole-
cules by ribosomes in the neuronal cell body.
The protein molecules then enter the spaces inside
Dendrite
the endoplasmic reticulum of the cell body and subse-
quently inside the Golgi apparatus, where two changes
occur: First, the neuropeptide-forming protein is enzy-
matically split into smaller fragments, some of which
are either the neuropeptide itself or a precursor of it.
Na+: 142 mEq/L
14 mEq/L
(Pumps)
Second, the Golgi apparatus packages the neuropep-
K+: 4.5 mEq/L
120 mEq/L
tide into minute transmitter vesicles that are released
-65
Axon
into the cytoplasm. Then the transmitter vesicles are
mV
8 mEq/L
transported all the way to the tips of the nerve fibers
Cl-: 107 mEq/L
?
by axonal streaming of the axon cytoplasm, traveling
Pump
at the slow rate of only a few centimeters per day.
Finally, these vesicles release their transmitter at the
Axon hillock
neuronal terminals in response to action potentials in
the same manner as for small-molecule transmitters.
However, the vesicle is autolyzed and is not reused.
Because of this laborious method of forming the
neuropeptides, much smaller quantities of them are
Figure 45-8
usually released than of the small-molecule transmit-
Distribution of sodium, potassium, and chloride ions across the
ters. This is partly compensated for by the fact that the
neuronal somal membrane; origin of the intrasomal membrane
neuropeptides are generally a thousand or more times
potential.
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
565
modes of function of the neuron—either excitation or
-65 that actually exists. Therefore, because of the high
inhibition—as explained in detail in the next sections.
intracellular potassium ion concentration, there is a
net tendency for potassium ions to diffuse to the
Concentration Differences of Ions Across the Neuronal
outside of the neuron, but this is opposed by continual
Somal Membrane. Figure 45-8 also shows the concen-
pumping of these potassium ions back to the
tration differences across the neuronal somal mem-
interior.
brane of the three ions that are most important for
Finally, the chloride ion gradient, 107 mEq/L outside
neuronal function: sodium ions, potassium ions, and
and 8 mEq/L inside, yields a Nernst potential of -70
chloride ions. At the top, the sodium ion concentra-
millivolts inside the neuron, which is only slightly more
tion is shown to be high in the extracellular fluid
negative than the actual measured value of -65 milli-
(142 mEq/L) but low inside the neuron (14 mEq/L).
volts. Therefore, chloride ions tend to leak very slightly
This sodium concentration gradient is caused by a
to the interior of the neuron, but those few that do leak
strong somal membrane sodium pump that continually
are moved back to the exterior, perhaps by an active
pumps sodium out of the neuron.
chloride pump.
The figure also shows that potassium ion concentra-
Keep these three Nernst potentials in mind and
tion is high inside the neuronal soma (120 mEq/L) but
remember the direction in which the different ions
low in the extracellular fluid (4.5 mEq/L). It shows that
tend to diffuse because this information is important
there is a potassium pump (the other half of the Na+-
in understanding both excitation and inhibition of the
K+ pump) that pumps potassium to the interior.
neuron by synapse activation or inactivation of ion
Figure 45-8 shows the chloride ion to be of high con-
channels.
centration in the extracellular fluid but low concentra-
tion inside the neuron. It also shows that the membrane
Uniform Distribution of Electrical Potential Inside the
is quite permeable to chloride ions and that there may
Soma. The interior of the neuronal soma contains a
be a weak chloride pump. Yet most of the reason
highly conductive electrolytic solution, the intracellu-
for the low concentration of chloride ions inside the
lar fluid of the neuron. Furthermore, the diameter of
neuron is the -65 millivolts in the neuron. That is, this
the neuronal soma is large (from 10 to 80 microme-
negative voltage repels the negatively charged chlo-
ters), causing almost no resistance to conduction of
ride ions, forcing them outward through the pores until
electric current from one part of the somal interior to
the concentration is much less inside the membrane
another part. Therefore, any change in potential in any
than outside.
part of the intrasomal fluid causes an almost exactly
Let us recall at this point what we learned in Chap-
equal change in potential at all other points inside the
ters 4 and 5 about the relation of ionic concentration
soma (that is, as long as the neuron is not transmitting
differences to membrane potentials. It will be recalled
an action potential). This is an important principle,
that an electrical potential across the cell membrane
because it plays a major role in “summation” of signals
can oppose movement of ions through a membrane if
entering the neuron from multiple sources, as we shall
the potential is of proper polarity and magnitude. A
see in subsequent sections of this chapter.
potential that exactly opposes movement of an ion is
called the Nernst potential for that ion; the equation
Effect of Synaptic Excitation on the Postsynaptic Membrane—
for this is the following:
Excitatory Postsynaptic Potential. Figure 45-9A shows the
resting neuron with an unexcited presynaptic terminal
Concentration inside
ˆ
EMF (mV)=±
61
¥logÊ
resting on its surface. The resting membrane potential
Ë Concentration outside¯
everywhere in the soma is -65 millivolts.
where EMF is the Nernst potential in millivolts on the
Figure 45-9B shows a presynaptic terminal that has
inside of the membrane. The potential will be negative
secreted an excitatory transmitter into the cleft
(-) for positive ions and positive (+) for negative ions.
between the terminal and the neuronal somal mem-
Now, let us calculate the Nernst potential that will
brane. This transmitter acts on the membrane excita-
exactly oppose the movement of each of the three sep-
tory receptor to increase the membrane’s permeability
arate ions: sodium, potassium, and chloride.
to Na+. Because of the large sodium concentration
For the sodium concentration difference shown in
gradient and large electrical negativity inside the
Figure 45-8, 142 mEq/L on the exterior and 14 mEq/L
neuron, sodium ions diffuse rapidly to the inside of the
on the interior, the membrane potential that will
membrane.
exactly oppose sodium ion movement through the
The rapid influx of positively charged sodium ions
sodium channels calculates to be
+61 millivolts.
to the interior neutralizes part of the negativity of the
However, the actual membrane potential is -65 milli-
resting membrane potential. Thus, in Figure 45-9B, the
volts, not +61 millivolts. Therefore, those sodium ions
resting membrane potential has increased in the posi-
that leak to the interior are immediately pumped back
tive direction from -65 to -45 millivolts. This positive
to the exterior by the sodium pump, thus maintaining
increase in voltage above the normal resting neuronal
the -65 millivolt negative potential inside the neuron.
potential—that is, to a less negative value—is called
For potassium ions, the concentration gradient is
the excitatory postsynaptic potential
(or EPSP),
120 mEq/L inside the neuron and 4.5 mEq/L outside.
because if this potential rises high enough in the pos-
This calculates to a Nernst potential of -86 millivolts
itive direction, it will elicit an action potential in the
inside the neuron, which is more negative than the
postsynaptic neuron, thus exciting it. (In this case, the
566
Unit IX The Nervous System: A. General Principles and Sensory Physiology
times as great a concentration of voltage-gated sodium
A
channels as does the soma and, therefore, can gener-
ate an action potential with much greater ease than
can the soma. The EPSP that will elicit an action
-65 mV
potential in the axon initial segment is between +10
and +20 millivolts. This is in contrast to the +30 or +40
millivolts or more required on the soma.
Resting Neuron
Once the action potential begins, it travels periph-
erally along the axon and usually also backward over
Initial segment
B
the soma. In some instances, it travels backward into
of axon
Excitatory
the dendrites, too, but not into all of them, because
they, like the neuronal soma, have very few voltage-
-45 mV
gated sodium channels and therefore frequently
cannot generate action potentials at all. Thus, in Figure
45-9B, the threshold for excitation of the neuron is
Na+
shown to be about -45 millivolts, which represents an
influx
Excited Neuron
Spread of
EPSP of +20 millivolts—that is, 20 millivolts more pos-
action potential
itive than the normal resting neuronal potential of
C
Cl- influx
-65 millivolts.
Inhibitory
-70 mV
Electrical Events During Neuronal
Inhibition
K+
efflux
Inhibited Neuron
Effect of Inhibitory Synapses on the Postsynaptic Membrane—
Inhibitory Postsynaptic Potential. The inhibitory synapses
Figure 45-9
open mainly chloride channels, allowing easy passage
of chloride ions. Now, to understand how the
Three states of a neuron. A, Resting neuron, with a normal intra-
inhibitory synapses inhibit the postsynaptic neuron,
neuronal potential of -65 millivolts. B, Neuron in an excited state,
with a less negative intraneuronal potential (-45 millivolts) caused
we must recall what we learned about the Nernst
by sodium influx. C, Neuron in an inhibited state, with a more neg-
potential for chloride ions. We calculated the Nernst
ative intraneuronal membrane potential (-70 millivolts) caused by
potential for chloride ions to be about -70 millivolts.
potassium ion efflux, chloride ion influx, or both.
This potential is more negative than the -65 millivolts
normally present inside the resting neuronal mem-
brane. Therefore, opening the chloride channels
EPSP is +20 millivolts—that is, 20 millivolts more pos-
will allow negatively charged chloride ions to move
itive than the resting value.)
from the extracellular fluid to the interior, which
However, we must issue a word of warning. Dis-
will make the interior membrane potential more
charge of a single presynaptic terminal can never
negative than normal, approaching the -70 millivolt
increase the neuronal potential from -65 millivolts
level.
all the way up to -45 millivolts. An increase of this
Opening potassium channels will allow positively
magnitude requires simultaneous discharge of many
charged potassium ions to move to the exterior, and
terminals—about 40 to 80 for the usual anterior motor
this will also make the interior membrane potential
neuron—at the same time or in rapid succession. This
more negative than usual. Thus, both chloride influx
occurs by a process called summation, which is dis-
and potassium efflux increase the degree of intracel-
cussed in detail in the next sections.
lular negativity, which is called hyperpolarization. This
inhibits the neuron because the membrane potential is
Generation of Action Potentials in the Initial Segment of the
even more negative than the normal intracellular
Axon Leaving the Neuron—Threshold for Excitation. When
potential. Therefore, an increase in negativity beyond
the EPSP rises high enough in the positive direction,
the normal resting membrane potential level is called
there comes a point at which this initiates an action
an inhibitory postsynaptic potential (IPSP).
potential in the neuron. However, the action potential
Figure 45-9C shows the effect on the membrane
does not begin adjacent to the excitatory synapses.
potential caused by activation of inhibitory synapses,
Instead, it begins in the initial segment of the axon
allowing chloride influx into the cell and/or potassium
where the axon leaves the neuronal soma. The main
efflux out of the cell, with the membrane potential
reason for this point of origin of the action potential
decreasing from its normal value of -65 millivolts to
is that the soma has relatively few voltage-gated
the more negative value of -70 millivolts. This mem-
sodium channels in its membrane, which makes it dif-
brane potential is
5 millivolts more negative than
ficult for the EPSP to open the required number of
normal and is therefore an IPSP of -5 millivolts, which
sodium channels to elicit an action potential. Con-
inhibits transmission of the nerve signal through the
versely, the membrane of the initial segment has seven
synapse.
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
567
Presynaptic Inhibition
required for the excess positive charges to leak out of
In addition to inhibition caused by inhibitory synapses
the excited neuron and to re-establish the normal
operating at the neuronal membrane, which is called
resting membrane potential.
postsynaptic inhibition, another type of inhibition
Precisely the opposite effect occurs for an IPSP; that
often occurs at the presynaptic terminals before the
is, the inhibitory synapse increases the permeability of
signal ever reaches the synapse. This type of inhibition,
the membrane to potassium or chloride ions, or both,
called presynaptic inhibition, occurs in the following
for 1 to 2 milliseconds, and this decreases the intra-
way.
neuronal potential to a more negative value than
Presynaptic inhibition is caused by release of an
normal, thereby creating the IPSP. This potential also
inhibitory substance onto the outsides of the presy-
dies away in about 15 milliseconds.
naptic nerve fibrils before their own endings terminate
Other types of transmitter substances can excite or
on the postsynaptic neuron. In most instances, the
inhibit the postsynaptic neuron for much longer
inhibitory transmitter substance is GABA (gamma-
periods—for hundreds of milliseconds or even for
aminobutyric acid). This has a specific effect of
seconds, minutes, or hours. This is especially true for
opening anion channels, allowing large numbers of
some of the neuropeptide types of transmitter
chloride ions to diffuse into the terminal fibril. The
substances.
negative charges of these ions inhibit synaptic trans-
mission because they cancel much of the excitatory
“Spatial Summation” in Neurons—
effect of the positively charged sodium ions that also
Threshold for Firing
enter the terminal fibrils when an action potential
Excitation of a single presynaptic terminal on the
arrives.
surface of a neuron almost never excites the neuron.
Presynaptic inhibition occurs in many of the sensory
The reason for this is that sufficient transmitter sub-
pathways in the nervous system. In fact, adjacent
stance is released by a single terminal to cause an
sensory nerve fibers often mutually inhibit one
EPSP usually no greater than 0.5 to 1 millivolt, instead
another, which minimizes sideways spread and mixing
of the 10 to 20 millivolts normally required to reach
of signals in sensory tracts. We discuss the importance
threshold for excitation. However, many presynaptic
of this phenomenon more fully in subsequent chapters.
terminals are usually stimulated at the same time.
Even though these terminals are spread over wide
Time Course of Postsynaptic Potentials
areas of the neuron, their effects can still summate;
When an excitatory synapse excites the anterior motor
that is, they can add to one another until neuronal exci-
neuron, the neuronal membrane becomes highly per-
tation does occur. The reason for this is the following:
meable to sodium ions for 1 to 2 milliseconds. During
It was pointed out earlier that a change in potential at
this very short time, enough sodium ions diffuse
any single point within the soma will cause the poten-
rapidly to the interior of the postsynaptic motor
tial to change everywhere inside the soma almost
neuron to increase its intraneuronal potential by a few
exactly equally. This is true because of the very high
millivolts, thus creating the excitatory postsynaptic
electrical conductivity inside the large neuronal cell
potential (EPSP) shown by the blue and green curves
body. Therefore, for each excitatory synapse that dis-
of Figure 45-10. This potential then slowly declines
charges simultaneously, the total intrasomal potential
over the next 15 milliseconds because this is the time
becomes more positive by 0.5 to 1.0 millivolt. When
16
16 synapses firing
+20
8
8 synapses firing
Action potential
0
4
4 synapses firing
–20
16
-40
Excitatory postsynaptic
potential
8
Figure 45-10
-60
4
Excitatory postsynaptic potentials, showing that
Resting membrane potential
simultaneous firing of only a few synapses will not
- 80
cause sufficient summated potential to elicit an
0
2
4
6
8
10
12
14
16
action potential, but that simultaneous firing of
Milliseconds
many synapses will raise the summated potential
to threshold for excitation and cause a superim-
posed action potential.
568
Unit IX The Nervous System: A. General Principles and Sensory Physiology
the EPSP becomes great enough, the threshold for
ronal soma. And these dendrites can receive signals
firing will be reached and an action potential will
from a large spatial area around the motor neuron.
develop spontaneously in the initial segment of the
This provides a vast opportunity for summation of
axon. This is demonstrated in Figure
45-10. The
signals from many separate presynaptic nerve fibers.
bottom postsynaptic potential in the figure was caused
It is also important that between 80 and 95 per cent
by simultaneous stimulation of 4 synapses; the next
of all the presynaptic terminals of the anterior motor
higher potential was caused by stimulation of
8
neuron terminate on dendrites, in contrast to only 5 to
synapses; finally, a still higher EPSP was caused by
20 per cent terminating on the neuronal soma. There-
stimulation of 16 synapses. In this last instance, the
fore, the preponderant share of the excitation is pro-
firing threshold had been reached, and an action
vided by signals transmitted by way of the dendrites.
potential was generated in the axon.
This effect of summing simultaneous postsynaptic
Most Dendrites Cannot Transmit Action Potentials—But They
potentials by activating multiple terminals on widely
Can Transmit Signals Within the Same Neuron by Electrotonic
spaced areas of the neuronal membrane is called
Conduction. Most dendrites fail to transmit action
spatial summation.
potentials because their membranes have relatively
few voltage-gated sodium channels, and their thresh-
“Temporal Summation”
olds for excitation are too high for action potentials to
Each time a presynaptic terminal fires, the released
occur. Yet they do transmit electrotonic current down
transmitter substance opens the membrane channels
the dendrites to the soma. Transmission of electrotonic
for at most a millisecond or so. But the changed post-
current means direct spread of electrical current by ion
synaptic potential lasts up to 15 milliseconds after the
conduction in the fluids of the dendrites but without
synaptic membrane channels have already closed.
generation of action potentials. Stimulation (or inhibi-
Therefore, a second opening of the same channels can
tion) of the neuron by this current has special charac-
increase the postsynaptic potential to a still greater
teristics, as follows.
level, and the more rapid the rate of stimulation, the
greater the postsynaptic potential becomes. Thus, suc-
Decrement of Electrotonic Conduction in the Den-
cessive discharges from a single presynaptic terminal,
drites—Greater Excitatory
(or Inhibitory) Effect by
if they occur rapidly enough, can add to one another;
Synapses Located Near the Soma. In Figure 45-11,
that is, they can “summate.” This type of summation is
multiple excitatory and inhibitory synapses are shown
called temporal summation.
stimulating the dendrites of a neuron. On the two den-
drites to the left, there are excitatory effects near the
Simultaneous Summation of Inhibitory and Excitatory Postsy-
tip ends; note the high levels of excitatory postsynap-
naptic Potentials. If an IPSP is tending to decrease the
tic potentials at these ends—that is, note the less neg-
membrane potential to a more negative value while an
ative membrane potentials at these points. However, a
EPSP is tending to increase the potential at the same
time, these two effects can either completely or par-
tially nullify each other. Thus, if a neuron is being
excited by an EPSP, an inhibitory signal from another
E
source can often reduce the postsynaptic potential to
E
less than threshold value for excitation, thus turning
E
off the activity of the neuron.
E
E
-40
E
E
“Facilitation” of Neurons
-50
I
I
-75
Often the summated postsynaptic potential is excita-
E
-70
tory but has not risen high enough to reach the thresh-
-60
old for firing by the postsynaptic neuron. When this
E
happens, the neuron is said to be facilitated. That is, its
membrane potential is nearer the threshold for firing
-40
-60 mV
I
I
-40
-50 -60
than normal, but not yet at the firing level. Conse-
-30
-70 -75
quently, another excitatory signal entering the neuron
E
I
I
from some other source can then excite the neuron
very easily. Diffuse signals in the nervous system often
do facilitate large groups of neurons so that they can
respond quickly and easily to signals arriving from
other sources.
Figure 45-11
Special Functions of Dendrites
for Exciting Neurons
Stimulation of a neuron by presynaptic terminals located on den-
drites, showing, especially, decremental conduction of excitatory
(E) electrotonic potentials in the two dendrites to the left and inhi-
Large Spatial Field of Excitation of the Dendrites. The den-
bition (I) of dendritic excitation in the dendrite that is uppermost.
drites of the anterior motor neurons often extend 500
A powerful effect of inhibitory synapses at the initial segment of
to 1000 micrometers in all directions from the neu-
the axon is also shown.
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
569
large share of the excitatory postsynaptic potential is
Relation of State of Excitation of the
lost before it reaches the soma. The reason is that the
Neuron to Rate of Firing
dendrites are long, and their membranes are thin and
at least partially permeable to potassium and chloride
ions, making them “leaky” to electric current. There-
“Excitatory State.” The “excitatory state” of a neuron is
fore, before the excitatory potentials can reach the
defined as the summated degree of excitatory drive to
soma, a large share of the potential is lost by leakage
the neuron. If there is a higher degree of excitation
through the membrane. This decrease in membrane
than inhibition of the neuron at any given instant, then
potential as it spreads electrotonically along dendrites
it is said that there is an excitatory state. Conversely, if
toward the soma is called decremental conduction.
there is more inhibition than excitation, then it is said
The farther the excitatory synapse is from the soma
that there is an inhibitory state.
of the neuron, the greater will be the decrement, and
When the excitatory state of a neuron rises above
the less will be excitatory signal reaching the soma.
the threshold for excitation, the neuron will fire repet-
Therefore, those synapses that lie near the soma have
itively as long as the excitatory state remains at that
far more effect in causing neuron excitation or inhibi-
level. Figure 45-12 shows responses of three types of
tion than those that lie far away from the soma.
neurons to varying levels of excitatory state. Note that
neuron 1 has a low threshold for excitation, whereas
Summation of Excitation and Inhibition in Dendrites. The
neuron 3 has a high threshold. But note also that
uppermost dendrite of Figure 45-11 is shown to be
neuron 2 has the lowest maximum frequency of dis-
stimulated by both excitatory and inhibitory synapses.
charge, whereas neuron 3 has the highest maximum
At the tip of the dendrite is a strong excitatory post-
frequency.
synaptic potential, but nearer the soma are two
Some neurons in the central nervous system fire
inhibitory synapses acting on the same dendrite. These
continuously because even the normal excitatory
inhibitory synapses provide a hyperpolarizing voltage
state is above the threshold level. Their frequency of
that completely nullifies the excitatory effect and
firing can usually be increased still more by further
indeed transmits a small amount of inhibition by elec-
increasing their excitatory state. The frequency can be
trotonic conduction toward the soma. Thus, dendrites
decreased, or firing can even be stopped, by superim-
can summate excitatory and inhibitory postsynaptic
posing an inhibitory state on the neuron. Thus, dif-
potentials in the same way that the soma can. Also
ferent neurons respond differently, have different
shown in the figure are several inhibitory synapses
thresholds for excitation, and have widely differing
located directly on the axon hillock and initial axon
maximum frequencies of discharge. With a little imag-
segment. This location provides especially powerful
ination, one can readily understand the importance of
inhibition because it has the direct effect of increasing
having different neurons with these many types of
the threshold for excitation at the very point where the
response characteristics to perform the widely varying
action potential is normally generated.
functions of the nervous system.
600
Neuron 1
Neuron 2
500
Neuron 3
400
300
200
Threshold
100
0
0
5
10
15
20
25
30
35
Figure 45-12
Excitatory state (arbitrary units)
Response characteristics of different types of
neurons to different levels of excitatory state.
570
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Some Special Characteristics
neuronal excitability, presumably by reducing the
threshold for excitation of neurons.
of Synaptic Transmission
Strychnine is one of the best known of all agents that
increase excitability of neurons. However, it does not
Fatigue of Synaptic Transmission. When excitatory
do this by reducing the threshold for excitation of
synapses are repetitively stimulated at a rapid rate, the
the neurons; instead, it inhibits the action of some nor-
number of discharges by the postsynaptic neuron is at
mally inhibitory transmitter substances, especially the
first very great, but the firing rate becomes progres-
inhibitory effect of glycine in the spinal cord. Therefore,
sively less in succeeding milliseconds or seconds. This
the effects of the excitatory transmitters become over-
whelming, and the neurons become so excited that they
is called fatigue of synaptic transmission.
go into rapidly repetitive discharge, resulting in severe
Fatigue is an exceedingly important characteristic of
tonic muscle spasms.
synaptic function because when areas of the nervous
Most anesthetics increase the neuronal membrane
system become overexcited, fatigue causes them to
threshold for excitation and thereby decrease synaptic
lose this excess excitability after awhile. For example,
transmission at many points in the nervous system.
fatigue is probably the most important means by which
Because many of the anesthetics are especially lipid-
the excess excitability of the brain during an epileptic
soluble, it has been reasoned that some of them might
seizure is finally subdued so that the seizure ceases.
change the physical characteristics of the neuronal
Thus, the development of fatigue is a protective mech-
membranes, making them less responsive to excitatory
anism against excess neuronal activity. This is dis-
agents.
cussed further in the description of reverberating
neuronal circuits in Chapter 46.
Synaptic Delay. During transmission of a neuronal signal
The mechanism of fatigue is mainly exhaustion or
from a presynaptic neuron to a postsynaptic neuron, a
partial exhaustion of the stores of transmitter sub-
certain amount of time is consumed in the process of
stance in the presynaptic terminals. The excitatory ter-
(1) discharge of the transmitter substance by the presy-
naptic terminal, (2) diffusion of the transmitter to the
minals on many neurons can store enough excitatory
postsynaptic neuronal membrane, (3) action of the
transmitter to cause only about 10,000 action poten-
transmitter on the membrane receptor, (4) action of
tials, and the transmitter can be exhausted in only a
the receptor to increase the membrane permeability,
few seconds to a few minutes of rapid stimulation. Part
and (5) inward diffusion of sodium to raise the excita-
of the fatigue process probably results from two other
tory postsynaptic potential to a high enough level to
factors as well: (1) progressive inactivation of many of
elicit an action potential. The minimal period of time
the postsynaptic membrane receptors and (2) slow
required for all these events to take place, even when
development of abnormal concentrations of ions
large numbers of excitatory synapses are stimulated
inside the postsynaptic neuronal cell.
simultaneously, is about 0.5 millisecond. This is called
the synaptic delay. Neurophysiologists can measure the
minimal delay time between an input volley of impulses
Effect of Acidosis or Alkalosis on Synaptic Transmission. Most
into a pool of neurons and the consequent output volley.
neurons are highly responsive to changes in pH of
From the measure of delay time, one can then estimate
the surrounding interstitial fluids. Normally, alkalosis
the number of series neurons in the circuit.
greatly increases neuronal excitability. For instance, a
rise in arterial blood pH from the 7.4 norm to 7.8 to
8.0 often causes cerebral epileptic seizures because of
increased excitability of some or all of the cerebral
References
neurons. This can be demonstrated especially well by
asking a person who is predisposed to epileptic seizures
Albright TD, Jessell TM, Kandell ER, Posner MI: Progress
to overbreathe. The overbreathing blows off carbon
in the neural sciences in the century after Cajal (and the
dioxide and therefore elevates the pH of the blood
mysteries that remain). Ann N Y Acad Sci 929:11, 2001.
momentarily, but even this short time can often precip-
Boehning D, Snyder SH: Novel neural modulators. Annu
itate an epileptic attack.
Rev Neurosci 26:105, 2003.
Conversely, acidosis greatly depresses neuronal activ-
Brasnjo G, Otis TS: Glycine transporters not only take out
ity; a fall in pH from 7.4 to below 7.0 usually causes a
the garbage, they recycle. Neuron 40:667, 2003.
comatose state. For instance, in very severe diabetic or
Cowley MA, Cone RD, Enriori P, et al: Electrophysiological
uremic acidosis, coma virtually always develops.
actions of peripheral hormones on melanocortin neurons.
Ann N Y Acad Sci 994:175, 2003.
Effect of Hypoxia on Synaptic Transmission. Neuronal
Engelman HS, MacDermott AB: Presynaptic inotropic
excitability is also highly dependent on an adequate
receptors and control of transmitter release. Nat Rev Neu-
supply of oxygen. Cessation of oxygen for only a few
rosci 5:135, 2004.
seconds can cause complete inexcitability of some
Girault JA, Greengard P: The neurobiology of dopamine
neurons. This is observed when the brain’s blood flow is
signaling. Arch Neurol 61:641, 2004.
temporarily interrupted, because within 3 to 7 seconds,
Haines DE: Fundamental Neuroscience. New York:
the person becomes unconscious.
Churchill Livingstone, 1997.
Haines DE, Lancon JA: Review of Neuroscience. New York:
Effect of Drugs on Synaptic Transmission. Many drugs are
Churchill Livingstone, 2003.
known to increase the excitability of neurons, and
Kandel ER: The molecular biology of memory storage: a dia-
others are known to decrease excitability. For instance,
logue between genes and synapses. Science 294:1030, 2001.
caffeine, theophylline, and theobromine, which are
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
found in coffee, tea, and cocoa, respectively, all increase
Science, 4th ed. New York: McGraw-Hill, 2000.
Chapter 45
Organization of the Nervous System, Basic Functions of Synapses, “Transmitter Substances”
571
Magee JC: Dendritic integration of excitatory synaptic input.
Schmolesky MT, Weber JT, De Zeeuw CI, Hansel C: The
Nat Rev Neurosci 1:181, 2000.
making of a complex spike: ionic composition and plastic-
Migliore M, Shepherd GM: Emerging rules for the distribu-
ity. Ann N Y Acad Sci 978:359, 2002.
tions of active dendritic conductances. Nat Rev Neurosci
Semyanov A, Walker MC, Kullmann DM, Silver RA:
3:362, 2002.
Tonically active GABA A receptors: modulating gain and
Muller D, Nikonenko I: Dynamic presynaptic varicosities: a
maintaining the tone. Trends Neurosci 27:262, 2004.
role in activity-dependent synaptogenesis. Trends Neu-
Stern JE: Nitric oxide and homeostatic control: an intercel-
rosci 26:573, 2003.
lular signaling molecule contributing to autonomic and
Nicholls DG, Budd SL: Mitochondria and neuronal survival.
neuroendocrine integration? Prog Biophys Mol Biol 84:
Physiol Rev 80:315, 2000.
197, 2004.
Nimchinsky EA, Sabatin BL, Svoboda K: Structure and
Timofeev I, Steriade M: Neocortical seizures: initiation,
function of dendritic spines. Annu Rev Physiol 64:313,
development and cessation. Neuroscience 123:299, 2004.
2002.
Tsay D, Yuste RL: On the electrical function of dendritic
Prast H, Philippu A: Nitric oxide as modulator of neuronal
spines. Trends Neurosci 27:77, 2004.
function. Prog Neurobiol 64:51, 2001.
West AR, Floresco SB, Charara A, et al: Electrophysiologi-
Reid CA, Bekkers JM, Clements JD: Presynaptic Ca2+ chan-
cal interactions between striatal glutamatergic and
nels: a functional patchwork. Trends Neurosci 26:683, 2003.
dopaminergic systems. Ann N Y Acad Sci 1003:53, 2003.
Robinson RB, Siegelbaum SA: Hyperpolarization-activated
Williams SR, Stuart GJ: Role of dendritic synapse location
cation currents: from molecules to physiological function.
in the control of action potential output. Trends Neurosci
Annu Rev Physiol 65:453, 2003.
26:147, 2003.
Ruff RL: Neurophysiology of the neuromuscular junction:
Zucker RS, Regehr WG: Short-term synaptic plasticity.
overview. Ann N Y Acad Sci 998:1, 2003.
Annu Rev Physiol 64:355, 2002.
C H A P T E R
4
6
Sensory Receptors, Neuronal
Circuits for Processing Information
Input to the nervous system is provided by sensory
receptors that detect such sensory stimuli as touch,
sound, light, pain, cold, and warmth. The purpose of
this chapter is to discuss the basic mechanisms by
which these receptors change sensory stimuli into
nerve signals that are then conveyed to and
processed in the central nervous system.
Types of Sensory Receptors and the Sensory
Stimuli They Detect
Table 46-1 lists and classifies most of the body’s sensory receptors. This table
shows that there are five basic types of sensory receptors: (1) mechanorecep-
tors, which detect mechanical compression or stretching of the receptor or of
tissues adjacent to the receptor; (2) thermoreceptors, which detect changes in
temperature, some receptors detecting cold and others warmth; (3) nociceptors
(pain receptors), which detect damage occurring in the tissues, whether physi-
cal damage or chemical damage; (4) electromagnetic receptors, which detect light
on the retina of the eye; and (5) chemoreceptors, which detect taste in the
mouth, smell in the nose, oxygen level in the arterial blood, osmolality of the
body fluids, carbon dioxide concentration, and perhaps other factors that make
up the chemistry of the body.
In this chapter, we discuss the function of a few specific types of receptors,
primarily peripheral mechanoreceptors, to illustrate some of the principles by
which receptors operate. Other receptors are discussed in other chapters in rela-
tion to the sensory systems that they subserve. Figure 46-1 shows some of the
types of mechanoreceptors found in the skin or in deep tissues of the body.
Differential Sensitivity of Receptors
The first question that must be answered is, how do two types of sensory recep-
tors detect different types of sensory stimuli? The answer is, by “differential sen-
sitivities.” That is, each type of receptor is highly sensitive to one type of stimulus
for which it is designed and yet is almost nonresponsive to other types of sensory
stimuli. Thus, the rods and cones of the eyes are highly responsive to light but
are almost completely nonresponsive to normal ranges of heat, cold, pressure
on the eyeballs, or chemical changes in the blood. The osmoreceptors of the
supraoptic nuclei in the hypothalamus detect minute changes in the osmolality
of the body fluids but have never been known to respond to sound. Finally, pain
receptors in the skin are almost never stimulated by usual touch or pressure
stimuli but do become highly active the moment tactile stimuli become severe
enough to damage the tissues.
Modality of Sensation—The “Labeled Line” Principle
Each of the principal types of sensation that we can experience—pain, touch,
sight, sound, and so forth—is called a modality of sensation. Yet despite the
fact that we experience these different modalities of sensation, nerve fibers
572
Chapter 46
Sensory Receptors, Neuronal Circuits for Processing Information
573
Table 46-1
Classification of Sensory Receptors
I.
Mechanoreceptors
Skin tactile sensibilities (epidermis and dermis)
Free nerve endings
Expanded tip endings
Free nerve
Expanded tip
Tactile hair
Merkel’s discs
endings
receptor
Plus several other variants
Spray endings
Ruffini’s endings
Encapsulated endings
Meissner’s corpuscles
Krause’s corpuscles
Hair end-organs
Deep tissue sensibilities
Free nerve endings
Expanded tip endings
Spray endings
Pacinian
Meissner’s
Krause’s
Ruffini’s endings
corpuscle
corpuscle
corpuscle
Encapsulated endings
Pacinian corpuscles
Plus a few other variants
Muscle endings
Muscle spindles
Golgi tendon receptors
Hearing
Sound receptors of cochlea
Equilibrium
Vestibular receptors
Arterial pressure
Ruffini’s
Golgi tendon
Muscle
Baroreceptors of carotid sinuses and aorta
end-organ
apparatus
spindle
II.
Thermoreceptors
Cold
Figure 46-1
Cold receptors
Warmth
Several types of somatic sensory nerve endings.
Warm receptors
III.
Nociceptors
Pain
Free nerve endings
IV.
Electromagnetic receptors
Vision
Rods
Cones
transmit only impulses. Therefore, how is it that
V. Chemoreceptors
different nerve fibers transmit different modalities of
Taste
sensation?
Receptors of taste buds
The answer is that each nerve tract terminates at a
Smell
Receptors of olfactory epithelium
specific point in the central nervous system, and
Arterial oxygen
the type of sensation felt when a nerve fiber is stimu-
Receptors of aortic and carotid bodies
lated is determined by the point in the nervous system
Osmolality
to which the fiber leads. For instance, if a pain fiber
Neurons in or near supraoptic nuclei
Blood CO2
is stimulated, the person perceives pain regardless
Receptors in or on surface of medulla and in aortic and
of what type of stimulus excites the fiber. The stimulus
carotid bodies
can be electricity, overheating of the fiber, crushing
Blood glucose, amino acids, fatty acids
of the fiber, or stimulation of the pain nerve ending
Receptors in hypothalamus
by damage to the tissue cells. In all these instances,
the person perceives pain. Likewise, if a touch fiber is
stimulated by electrical excitation of a touch receptor
or in any other way, the person perceives touch
Transduction of Sensory
because touch fibers lead to specific touch areas in
Stimuli into Nerve Impulses
the brain. Similarly, fibers from the retina of the eye
terminate in the vision areas of the brain, fibers from
Local Electrical Currents at Nerve
the ear terminate in the auditory areas of the brain,
and temperature fibers terminate in the temperature
Endings—Receptor Potentials
areas.
This specificity of nerve fibers for transmitting only
All sensory receptors have one feature in common.
one modality of sensation is called the labeled line
Whatever the type of stimulus that excites the recep-
principle.
tor, its immediate effect is to change the membrane
574
Unit IX The Nervous System: A. General Principles and Sensory Physiology
electrical potential of the receptor. This change in
receptor potential rises above the threshold level, the
potential is called a receptor potential.
greater becomes the action potential frequency.
Mechanisms of Receptor Potentials. Different receptors
Receptor Potential of the Pacinian Corpuscle—
can be excited in one of several ways to cause recep-
An Example of Receptor Function
tor potentials: (1) by mechanical deformation of the
The student should at this point restudy the anatomi-
receptor, which stretches the receptor membrane and
cal structure of the pacinian corpuscle shown in Figure
opens ion channels; (2) by application of a chemical to
46-1. Note that the corpuscle has a central nerve fiber
the membrane, which also opens ion channels; (3) by
extending through its core. Surrounding this are mul-
change of the temperature of the membrane, which
tiple concentric capsule layers, so that compression
alters the permeability of the membrane; or (4) by the
anywhere on the outside of the corpuscle will elongate,
effects of electromagnetic radiation, such as light on a
indent, or otherwise deform the central fiber.
retinal visual receptor, which either directly or indi-
Now study Figure 46-3, which shows only the
rectly changes the receptor membrane characteristics
central fiber of the pacinian corpuscle after all capsule
and allows ions to flow through membrane channels.
layers but one have been removed. The tip of the
It will be recognized that these four means of exciting
central fiber inside the capsule is unmyelinated, but
receptors correspond in general with the different
the fiber does become myelinated (the blue sheath
types of known sensory receptors. In all instances, the
shown in the figure) shortly before leaving the cor-
basic cause of the change in membrane potential is a
puscle to enter a peripheral sensory nerve.
change in membrane permeability of the receptor,
The figure also shows the mechanism by which a
which allows ions to diffuse more or less readily
receptor potential is produced in the pacinian corpus-
through the membrane and thereby to change the
cle. Observe the small area of the terminal fiber that
transmembrane potential.
has been deformed by compression of the corpuscle,
and note that ion channels have opened in the mem-
Maximum Receptor Potential Amplitude. The maximum
brane, allowing positively charged sodium ions to
amplitude of most sensory receptor potentials is about
diffuse to the interior of the fiber. This creates
100 millivolts, but this level occurs only at an extremely
increased positivity inside the fiber, which is the
high intensity of sensory stimulus. This is about the
“receptor potential.” The receptor potential in turn
same maximum voltage recorded in action potentials
induces a local circuit of current flow, shown by the
and is also the change in voltage when the membrane
arrows, that spreads along the nerve fiber. At the first
becomes maximally permeable to sodium ions.
node of Ranvier, which itself lies inside the capsule of
the pacinian corpuscle, the local current flow depolar-
Relation of the Receptor Potential to Action Potentials. When
izes the fiber membrane at this node, which then sets
the receptor potential rises above the threshold for
off typical action potentials that are transmitted along
eliciting action potentials in the nerve fiber attached
the nerve fiber toward the central nervous system.
to the receptor, then action potentials occur, as illus-
trated in Figure 46-2. Note also that the more the
Relation Between Stimulus Intensity and the Receptor Poten-
tial. Figure 46-4 shows the changing amplitude of the
receptor potential caused by progressively stronger
mechanical compression
(increasing
“stimulus
strength”) applied experimentally to the central core
of a pacinian corpuscle. Note that the amplitude
Action potentials
+30
Action
0
Receptor potential
potential
Deformed
-30
area
++++++++++
+++++
+
+ +
+
+
-- - -
+ + + +
+
Receptor potential
+
+ +
+
+
-60
Threshold
+
+ +
+
+
+
+
+ +
+
+
-- - -
+ + + +
++++++
++++++++++
-90
Resting membrane potential
Node of
Ranvier
0
10
20
30
40
60
80
100
120
140
Milliseconds
Figure 46-3
Figure 46-2
Excitation of a sensory nerve fiber by a receptor potential pro-
duced in a pacinian corpuscle. (Modified from Loëwenstein WR:
Typical relation between receptor potential and action potentials
Excitation and inactivation in a receptor membrane. Ann N Y Acad
when the receptor potential rises above threshold level.
Sci 94:510, 1961.)
Chapter 46
Sensory Receptors, Neuronal Circuits for Processing Information
575
Joint capsule receptors
100
Muscle spindle
250
90
Hair receptor
Pacinian corpuscle
80
200
70
150
60
100
50
40
50
30
0
20
0
1
2
3
4
5
6
7
8
Seconds
10
0
0
20
40
60
80
100
Figure 46-5
Stimulus strength
(per cent)
Adaptation of different types of receptors, showing rapid adapta-
tion of some receptors and slow adaptation of others.
Figure 46-4
within a second or so, whereas some joint capsule and
Relation of amplitude of receptor potential to strength of a
muscle spindle receptors adapt slowly.
mechanical stimulus applied to a pacinian corpuscle. (Data from
Furthermore, some sensory receptors adapt to a far
Loëwenstein WR: Excitation and inactivation in a receptor mem-
greater extent than others. For example, the pacinian
brane. Ann N Y Acad Sci 94:510, 1961.)
corpuscles adapt to “extinction” within a few hun-
dredths of a second, and the receptors at the bases of
the hairs adapt to extinction within a second or more.
increases rapidly at first but then progressively less
It is probable that all other mechanoreceptors eventu-
rapidly at high stimulus strength.
ally adapt almost completely, but some require hours
In turn, the frequency of repetitive action potentials
or days to do so, for which reason they are called “non-
transmitted from sensory receptors increases approx-
adapting” receptors. The longest measured time for
imately in proportion to the increase in receptor
complete adaptation of a mechanoreceptor is about
potential. Putting this principle together with the data
2 days, which is the adaptation time for many caro-
in Figure 46-4, one can see that very intense stimula-
tid and aortic baroreceptors. Conversely, some of
tion of the receptor causes progressively less and less
the nonmechanoreceptors—the chemoreceptors and
additional increase in numbers of action potentials.
pain receptors, for instance—probably never adapt
This is an exceedingly important principle that is appli-
completely.
cable to almost all sensory receptors. It allows the
receptor to be sensitive to very weak sensory experi-
Mechanisms by Which Receptors Adapt. The mechanism of
ence and yet not reach a maximum firing rate until the
receptor adaptation is different for each type of recep-
sensory experience is extreme. This allows the recep-
tor, in much the same way that development of a
tor to have an extreme range of response, from very
receptor potential is an individual property. For
weak to very intense.
instance, in the eye, the rods and cones adapt by chang-
ing the concentrations of their light-sensitive chemi-
cals (which is discussed in Chapter 50).
Adaptation of Receptors
In the case of the mechanoreceptors, the receptor
that has been studied in greatest detail is the pacinian
Another characteristic of all sensory receptors is that
corpuscle. Adaptation occurs in this receptor in two
they adapt either partially or completely to any con-
ways. First, the pacinian corpuscle is a viscoelastic
stant stimulus after a period of time. That is, when a
structure so that when a distorting force is suddenly
continuous sensory stimulus is applied, the receptor
applied to one side of the corpuscle, this force is
responds at a high impulse rate at first and then at a
instantly transmitted by the viscous component of the
progressively slower rate until finally the rate of action
corpuscle directly to the same side of the central nerve
potentials decreases to very few or often to none at all.
fiber, thus eliciting a receptor potential. However,
Figure
46-5 shows typical adaptation of certain
within a few hundredths of a second, the fluid within
types of receptors. Note that the pacinian corpuscle
the corpuscle redistributes, so that the receptor poten-
adapts extremely rapidly and hair receptors adapt
tial is no longer elicited. Thus, the receptor potential
576
Unit IX The Nervous System: A. General Principles and Sensory Physiology
appears at the onset of compression but disappears
status is taking place, one can predict in one’s mind the
within a small fraction of a second even though the
state of the body a few seconds or even a few minutes
compression continues.
later. For instance, the receptors of the semicircular
The second mechanism of adaptation of the pacin-
canals in the vestibular apparatus of the ear detect the
ian corpuscle, but a much slower one, results from a
rate at which the head begins to turn when one runs
process called accommodation, which occurs in the
around a curve. Using this information, a person can
nerve fiber itself. That is, even if by chance the central
predict how much he or she will turn within the next
core fiber should continue to be distorted, the tip of
2 seconds and can adjust the motion of the legs ahead
the nerve fiber itself gradually becomes “accommo-
of time to keep from losing balance. Likewise, recep-
dated” to the stimulus. This probably results from
tors located in or near the joints help detect the rates
progressive “inactivation” of the sodium channels in
of movement of the different parts of the body. For
the nerve fiber membrane, which means that sodium
instance, when one is running, information from the
current flow through the channels causes them gradu-
joint rate receptors allows the nervous system to
ally to close, an effect that seems to occur for all
predict where the feet will be during any precise frac-
or most cell membrane sodium channels, as was
tion of the next second. Therefore, appropriate motor
explained in Chapter 5.
signals can be transmitted to the muscles of the legs to
Presumably, these same two general mechanisms of
make any necessary anticipatory corrections in posi-
adaptation apply also to the other types of mechano-
tion so that the person will not fall. Loss of this pre-
receptors. That is, part of the adaptation results from
dictive function makes it impossible for the person to
readjustments in the structure of the receptor itself,
run.
and part from an electrical type of accommodation in
the terminal nerve fibril.
Nerve Fibers That Transmit
Slowly Adapting Receptors Detect Continuous Stimulus
Different Types of Signals,
Strength—The “Tonic” Receptors. Slowly adapting recep-
and Their Physiologic
tors continue to transmit impulses to the brain as long
Classification
as the stimulus is present (or at least for many minutes
or hours). Therefore, they keep the brain constantly
Some signals need to be transmitted to or from the
apprised of the status of the body and its relation to
central nervous system extremely rapidly; otherwise,
its surroundings. For instance, impulses from the
the information would be useless. An example of this is
the sensory signals that apprise the brain of the momen-
muscle spindles and Golgi tendon apparatuses allow
tary positions of the legs at each fraction of a second
the nervous system to know the status of muscle con-
during running. At the other extreme, some types of
traction and load on the muscle tendon at each instant.
sensory information, such as that depicting prolonged,
Other slowly adapting receptors include (1) recep-
aching pain, do not need to be transmitted rapidly, so
tors of the macula in the vestibular apparatus, (2) pain
that slowly conducting fibers will suffice. As shown in
receptors, (3) baroreceptors of the arterial tree, and (4)
Figure 46-6, nerve fibers come in all sizes between 0.5
chemoreceptors of the carotid and aortic bodies.
and 20 micrometers in diameter—the larger the diame-
Because the slowly adapting receptors can continue
ter, the greater the conducting velocity. The range of
to transmit information for many hours, they are called
conducting velocities is between 0.5 and 120 m/sec.
tonic receptors.
General Classification of Nerve Fibers. Shown in Figure 46-6
Rapidly Adapting Receptors Detect Change in Stimulus
is a “general classification” and a “sensory nerve classi-
fication” of the different types of nerve fibers. In the
Strength—The
“Rate Receptors,”
“Movement Receptors,”
general classification, the fibers are divided into types A
or
“Phasic Receptors.” Receptors that adapt rapidly
and C, and the type A fibers are further subdivided into
cannot be used to transmit a continuous signal because
a, b, g, and d fibers.
these receptors are stimulated only when the stimulus
Type A fibers are the typical large and medium-sized
strength changes. Yet they react strongly while a
myelinated fibers of spinal nerves. Type C fibers are the
change is actually taking place. Therefore, these recep-
small unmyelinated nerve fibers that conduct impulses
tors are called rate receptors, movement receptors, or
at low velocities. The C fibers constitute more than one
phasic receptors. Thus, in the case of the pacinian cor-
half of the sensory fibers in most peripheral nerves as
puscle, sudden pressure applied to the tissue excites
well as all the postganglionic autonomic fibers.
this receptor for a few milliseconds, and then its exci-
The sizes, velocities of conduction, and functions of
the different nerve fiber types are also given in Figure
tation is over even though the pressure continues. But
46-6. Note that a few large myelinated fibers can trans-
later, it transmits a signal again when the pressure is
mit impulses at velocities as great as
120 m/sec, a
released. In other words, the pacinian corpuscle is
distance in 1 second that is longer than a football field.
exceedingly important in apprising the nervous system
Conversely, the smallest fibers transmit impulses as
of rapid tissue deformations, but it is useless for trans-
slowly as 0.5 m/sec, requiring about 2 seconds to go from
mitting information about constant conditions in the
the big toe to the spinal cord.
body.
Alternative Classification Used by Sensory Physiologists.
Importance of the Rate Receptors—Their Predictive Function.
Certain recording techniques have made it possible to
If one knows the rate at which some change in bodily
separate the type Aa fibers into two subgroups; yet
Chapter 46
Sensory Receptors, Neuronal Circuits for Processing Information
577
Myelinated
Unmyelinated
Diameter (micrometers)
20
15
10
5
1
2.0
0.5
Conduction velocity (m/sec.)
120
80
60
30
6 2.0
0.5
General classification
C
A
a
b
g
d
Sensory nerve classification
I
II
III
IV
IA
IB
Sensory functions
Muscle spindle
Muscle spindle
(primary ending)
(secondary ending)
Muscle tendon
(Golgi tendon organ)
Hair receptors
Vibration
(pacinian corpuscle)
High discrimination touch
Crude touch
(Meissner's expanded tips)
and pressure
Deep pressure
Tickle
and touch
Pricking pain
Aching pain
Cold
Warmth
Motor function
Skeletal muscle
Muscle spindle
Sympathetic
(type Aa)
(type Ag)
(type C)
20
15
10
5
1 2.0
0.5
Figure 46-6
Nerve fiber diameter (micrometers)
Physiologic classifications and functions of nerve
fibers.
these same recording techniques cannot
distinguish
Group III
easily between Ab and Ag fibers. Therefore, the fol-
Fibers carrying temperature, crude touch, and pricking
lowing classification is frequently used by sensory
pain sensations (average about 3 micrometers in diame-
physiologists:
ter; they are d-type A fibers in the general classification).
Group IV
Group Ia
Unmyelinated fibers carrying pain, itch, temperature,
Fibers from the annulospiral endings of muscle spindles
and crude touch sensations (0.5 to 2 micrometers in
(average about 17 microns in diameter; these are a-type
diameter; they are type C fibers in the general classifi-
A fibers in the general classification).
cation).
Group Ib
Fibers from the Golgi tendon organs (average about 16
Transmission of Signals
micrometers in diameter; these also are a-type A
of Different Intensity in
fibers).
Nerve Tracts—Spatial and
Temporal Summation
Group II
Fibers from most discrete cutaneous tactile receptors
and from the flower-spray endings of the muscle spin-
One of the characteristics of each signal that always
dles (average about 8 micrometers in diameter; these
must be conveyed is signal intensity—for instance, the
are b- and g-type A fibers in the general classification).
intensity of pain. The different gradations of intensity
578
Unit IX The Nervous System: A. General Principles and Sensory Physiology
can be transmitted either by using increasing numbers
stimulus, with only a single nerve fiber in the middle
of parallel fibers or by sending more action potentials
of the bundle stimulated strongly (represented by the
along a single fiber. These two mechanisms are called,
red-colored fiber), whereas several adjacent fibers are
respectively, spatial summation and temporal
stimulated weakly (half-red fibers). The other two
summation.
views of the nerve cross section show the effect of a
moderate stimulus and a strong stimulus, with pro-
Spatial Summation. Figure 46-7 shows the phenomenon
gressively more fibers being stimulated. Thus, the
of spatial summation, whereby increasing signal
stronger signals spread to more and more fibers. This
strength is transmitted by using progressively greater
is the phenomenon of spatial summation.
numbers of fibers. This figure shows a section of skin
innervated by a large number of parallel pain fibers.
Temporal Summation. A second means for transmitting
Each of these arborizes into hundreds of minute free
signals of increasing strength is by increasing the fre-
nerve endings that serve as pain receptors. The entire
quency of nerve impulses in each fiber, which is called
cluster of fibers from one pain fiber frequently covers
temporal summation. Figure 46-8 demonstrates this,
an area of skin as large as 5 centimeters in diameter.
showing in the upper part a changing strength of signal
This area is called the receptor field of that fiber. The
and in the lower part the actual impulses transmitted
number of endings is large in the center of the field but
by the nerve fiber.
diminishes toward the periphery. One can also see
from the figure that the arborizing fibrils overlap those
from other pain fibers. Therefore, a pinprick of the skin
Transmission and Processing
usually stimulates endings from many different pain
of Signals in Neuronal Pools
fibers simultaneously. When the pinprick is in the
center of the receptive field of a particular pain fiber,
The central nervous system is composed of thousands
the degree of stimulation of that fiber is far greater
to millions of neuronal pools; some of these contain
than when it is in the periphery of the field, because
few neurons, while others have vast numbers. For
the number of free nerve endings in the middle of the
instance, the entire cerebral cortex could be consid-
field is much greater than at the periphery.
ered to be a single large neuronal pool. Other neuronal
Thus, the lower part of Figure 46-7 shows three
pools include the different basal ganglia and the
views of the cross section of the nerve bundle leading
specific nuclei in the thalamus, cerebellum, mesen-
from the skin area. To the left is the effect of a weak
cephalon, pons, and medulla. Also, the entire dorsal
gray matter of the spinal cord could be considered one
long pool of neurons.
Each neuronal pool has its own special organization
that causes it to process signals in its own unique way,
Pin
80
60
Nerve
40
Skin
20
0
Time
Weak
Moderate
Strong
stimulus
stimulus
stimulus
Figure 46-8
Figure 46-7
Translation of signal strength into a frequency-modulated series
of nerve impulses, showing the strength of signal (above) and the
Pattern of stimulation of pain fibers in a nerve leading from an area
separate nerve impulses (below). This is an example of temporal
of skin pricked by a pin. This is an example of spatial summation.
summation.
Chapter 46
Sensory Receptors, Neuronal Circuits for Processing Information
579
thus allowing the total consortium of pools to achieve
excitatory presynaptic terminal almost never causes an
the multitude of functions of the nervous system. Yet
action potential in a postsynaptic neuron. Instead,
despite their differences in function, the pools also
large numbers of input terminals must discharge on
have many similar principles of function, described in
the same neuron either simultaneously or in rapid suc-
the following pages.
cession to cause excitation. For instance, in Figure
46-9, let us assume that six terminals must discharge
almost simultaneously to excite any one of the
Relaying of Signals Through
neurons. If the student counts the number of terminals
Neuronal Pools
on each one of the neurons from each input fiber, he
or she will see that input fiber 1 has more than enough
Organization of Neurons for Relaying Signals. Figure 46-9 is
terminals to cause neuron a to discharge. The stimulus
a schematic diagram of several neurons in a neuronal
from input fiber 1 to this neuron is said to be an
pool, showing “input” fibers to the left and “output”
excitatory stimulus; it is also called a suprathreshold
fibers to the right. Each input fiber divides hundreds
stimulus because it is above the threshold required for
to thousands of times, providing a thousand or more
excitation.
terminal fibrils that spread into a large area in the pool
Input fiber 1 also contributes terminals to neurons
to synapse with dendrites or cell bodies of the neurons
b and c, but not enough to cause excitation. Never-
in the pool. The dendrites usually also arborize and
theless, discharge of these terminals makes both these
spread hundreds to thousands of micrometers in the
neurons more likely to be excited by signals arriving
pool.
through other incoming nerve fibers. Therefore, the
The neuronal area stimulated by each incoming
stimuli to these neurons are said to be subthreshold,
nerve fiber is called its stimulatory field. Note in Figure
and the neurons are said to be facilitated.
46-9 that large numbers of the terminals from each
Similarly, for input fiber 2, the stimulus to neuron d
input fiber lie on the nearest neuron in its “field,” but
is a suprathreshold stimulus, and the stimuli to neurons
progressively fewer terminals lie on the neurons
b and c are subthreshold, but facilitating, stimuli.
farther away.
Figure 46-9 represents a highly condensed version
of a neuronal pool because each input nerve fiber
Threshold and Subthreshold Stimuli—Excitation or Facilitation.
usually provides massive numbers of branching termi-
From the discussion of synaptic function in Chapter
nals to hundreds or thousands of neurons in its distri-
45,
it
will
be
recalled
that
discharge
of
a
single
bution “field,” as shown in Figure 46-10. In the central
portion of the field in this figure, designated by the
circled area, all the neurons are stimulated by the
incoming fiber. Therefore, this is said to be the dis-
charge zone of the incoming fiber, also called the
excited zone or liminal zone. To each side, the neurons
are facilitated but not excited, and these areas are
called the facilitated zone, also called the subthreshold
1
a
zone or subliminal zone.
Inhibition of a Neuronal Pool. We must also remember
b
that some incoming fibers inhibit neurons, rather than
exciting them. This is the opposite of facilitation, and
the entire field of the inhibitory branches is called the
inhibitory zone. The degree of inhibition in the center
c
of this zone is great because of large numbers of
endings in the center; it becomes progressively less
toward its edges.
d
2
Facilitated zone
Input nerve
Discharge zone
fiber
Facilitated zone
Figure 46-9
Figure 46-10
Basic organization of a neuronal pool.
“Discharge” and “facilitated” zones of a neuronal pool.
580
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Source
Source
#1
Source
Source
#2
#3
Convergence from
Convergence from
single source
multiple sources
A
B
Divergence in same tract
Divergence in multiple tracts
A
B
Figure 46-12
Figure 46-11
“Convergence” of multiple input fibers onto a single neuron.
A, Multiple input fibers from a single source. B, Input fibers from
“Divergence” in neuronal pathways. A, Divergence within a
multiple separate sources.
pathway to cause “amplification” of the signal. B, Divergence into
multiple tracts to transmit the signal to separate areas.
Convergence can also result from input signals (exci-
Divergence of Signals Passing Through
tatory or inhibitory) from multiple sources, as shown
Neuronal Pools
in Figure 46-12B. For instance, the interneurons of the
Often it is important for weak signals entering a neu-
spinal cord receive converging signals from (1) periph-
ronal pool to excite far greater numbers of nerve fibers
eral nerve fibers entering the cord, (2) propriospinal
leaving the pool. This phenomenon is called diver-
fibers passing from one segment of the cord to
gence. Two major types of divergence occur and have
another, (3) corticospinal fibers from the cerebral
entirely different purposes.
cortex, and (4) several other long pathways descend-
An amplifying type of divergence is shown in Figure
ing from the brain into the spinal cord. Then the signals
46-11A. This means simply that an input signal spreads
from the interneurons converge on the anterior motor
to an increasing number of neurons as it passes
neurons to control muscle function.
through successive orders of neurons in its path. This
Such convergence allows summation of information
type of divergence is characteristic of the corticospinal
from different sources, and the resulting response is a
pathway in its control of skeletal muscles, with a single
summated effect of all the different types of informa-
large pyramidal cell in the motor cortex capable, under
tion. Convergence is one of the important means by
highly facilitated conditions, of exciting as many as
which the central nervous system correlates, sum-
10,000 muscle fibers.
mates, and sorts different types of information.
The second type of divergence, shown in Figure
46-11B, is divergence into multiple tracts. In this case,
Neuronal Circuit with Both Excitatory and
the signal is transmitted in two directions from the
Inhibitory Output Signals
pool. For instance, information transmitted up the
Sometimes an incoming signal to a neuronal pool
dorsal columns of the spinal cord takes two courses in
causes an output excitatory signal going in one direc-
the lower part of the brain: (1) into the cerebellum and
tion and at the same time an inhibitory signal going
(2) on through the lower regions of the brain to the
elsewhere. For instance, at the same time that an exci-
thalamus and cerebral cortex. Likewise, in the thala-
tatory signal is transmitted by one set of neurons in the
mus, almost all sensory information is relayed both
spinal cord to cause forward movement of a leg, an
into still deeper structures of the thalamus and at the
inhibitory signal is transmitted through a separate set
same time to discrete regions of the cerebral cortex.
of neurons to inhibit the muscles on the back of the
leg so that they will not oppose the forward move-
Convergence of Signals
ment. This type of circuit is characteristic for control-
Convergence means signals from multiple inputs
ling all antagonistic pairs of muscles, and it is called the
uniting to excite a single neuron. Figure 46-12A shows
reciprocal inhibition circuit.
convergence from a single source. That is, multiple ter-
Figure 46-13 shows the means by which the inhibi-
minals from a single incoming fiber tract terminate on
tion is achieved. The input fiber directly excites the
the same neuron. The importance of this is that
excitatory output pathway, but it stimulates an inter-
neurons are almost never excited by an action poten-
mediate inhibitory neuron (neuron 2), which secretes
tial from a single input terminal. But action potentials
a different type of transmitter substance to inhibit the
converging on the neuron from multiple terminals
second output pathway from the pool. This type of
provide enough spatial summation to bring the neuron
circuit is also important in preventing overactivity in
to the threshold required for discharge.
many parts of the brain.
Chapter 46
Sensory Receptors, Neuronal Circuits for Processing Information
581
Excitatory synapse
Input
Output
#1
Input fiber
Excitation
A
#2
#3
Inhibition
Input
Output
Inhibitory synapse
Figure 46-13
B
Facilitation
Inhibitory circuit. Neuron 2 is an inhibitory neuron.
Input
Output
Prolongation of a Signal by a
Neuronal Pool—“Afterdischarge”
Thus far, we have considered signals that are merely
relayed through neuronal pools. However, in many
instances, a signal entering a pool causes a prolonged
output discharge, called afterdischarge, lasting a
few milliseconds to as long as many minutes after
the incoming signal is over. The most important
C
Inhibition
mechanisms by which afterdischarge occurs are the
following.
Input
Output
Synaptic Afterdischarge. When excitatory synapses dis-
charge on the surfaces of dendrites or soma of a
neuron, a postsynaptic electrical potential develops in
the neuron and lasts for many milliseconds, especially
when some of the long-acting synaptic transmitter sub-
stances are involved. As long as this potential lasts, it
can continue to excite the neuron, causing it to trans-
mit a continuous train of output impulses, as was
D
explained in Chapter 45. Thus, as a result of this synap-
tic “afterdischarge” mechanism alone, it is possible for
Figure 46-14
a single instantaneous input signal to cause a sustained
signal output (a series of repetitive discharges) lasting
Reverberatory circuits of increasing complexity.
for many milliseconds.
Reverberatory (Oscillatory) Circuit as a Cause of Signal Prolon-
intensity and frequency of reverberation, whereas an
gation. One of the most important of all circuits in the
inhibitory signal depresses or stops the reverberation.
entire nervous system is the reverberatory, or oscilla-
Figure 46-14D shows that most reverberating path-
tory, circuit. Such circuits are caused by positive feed-
ways are constituted of many parallel fibers. At each
back within the neuronal circuit that feeds back to
cell station, the terminal fibrils spread widely. In such
re-excite the input of the same circuit. Consequently,
a system, the total reverberating signal can be either
once stimulated, the circuit may discharge repetitively
weak or strong, depending on how many parallel nerve
for a long time.
fibers are momentarily involved in the reverberation.
Several possible varieties of reverberatory circuits
are shown in Figure 46-14. The simplest, shown in
Characteristics of Signal Prolongation from a Rever-
Figure 46-14A, involves only a single neuron. In this
beratory Circuit. Figure 46-15 shows output signals
case, the output neuron simply sends a collateral nerve
from a typical reverberatory circuit. The input stimu-
fiber back to its own dendrites or soma to restimulate
lus may last only 1 millisecond or so, and yet the output
itself. Although this type of circuit probably is not an
can last for many milliseconds or even minutes. The
important one, theoretically, once the neuron dis-
figure demonstrates that the intensity of the output
charges, the feedback stimuli could keep the neuron
signal usually increases to a high value early in rever-
discharging for a protracted time thereafter.
beration and then decreases to a critical point, at which
Figure 46-14B shows a few additional neurons in the
it suddenly ceases entirely. The cause of this sudden
feedback circuit, which causes a longer delay between
cessation of reverberation is fatigue of synaptic junc-
initial discharge and the feedback signal. Figure
tions in the circuit. Fatigue beyond a certain critical
46-14C shows a still more complex system in which
level lowers the stimulation of the next neuron in the
both facilitatory and inhibitory fibers impinge on the
circuit below threshold level so that the circuit feed-
reverberating circuit. A facilitatory signal enhances the
back is suddenly broken.
582
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Inhibited
Output
Normal
Excitation
Facilitated
Inhibition
Time
Time
Figure 46-15
Figure 46-16
Typical pattern of the output signal from a reverberatory circuit
after a single input stimulus, showing the effects of facilitation and
Continuous output from either a reverberating circuit or a pool of
inhibition.
intrinsically discharging neurons. This figure also shows the effect
of excitatory or inhibitory input signals.
The duration of the total signal before cessation can
also be controlled by signals from other parts of the
brain that inhibit or facilitate the circuit. Almost these
of reverberation. Note that an excitatory input signal
exact patterns of output signals are recorded from the
greatly increases the output signal, whereas an
motor nerves exciting a muscle involved in a flexor
inhibitory input signal greatly decreases the output.
reflex after pain stimulation of the foot (as shown later
Those students who are familiar with radio trans-
in Figure 46-18).
mitters will recognize this to be a carrier wave type of
information transmission. That is, the excitatory and
Continuous Signal Output from Some
inhibitory control signals are not the cause of the
Neuronal Circuits
output signal, but they do control its changing level of
Some neuronal circuits emit output signals continu-
intensity. Note that this carrier wave system allows a
ously, even without excitatory input signals. At least
decrease in signal intensity as well as an increase,
two mechanisms can cause this effect: (1) continuous
whereas up to this point, the types of information
intrinsic neuronal discharge and (2) continuous rever-
transmission we have discussed have been mainly pos-
beratory signals.
itive information rather than negative information.
This type of information transmission is used by the
Continuous Discharge Caused by Intrinsic Neuronal Excitabil-
autonomic nervous system to control such functions as
ity. Neurons, like other excitable tissues, discharge
vascular tone, gut tone, degree of constriction of the
repetitively if their level of excitatory membrane
iris in the eye, and heart rate. That is, the nerve exci-
potential rises above a certain threshold level. The
tatory signal to each of these can be either increased
membrane potentials of many neurons even normally
or decreased by accessory input signals into the rever-
are high enough to cause them to emit impulses con-
berating neuronal pathway.
tinually. This occurs especially in many of the neurons
of the cerebellum, as well as in most of the interneu-
Rhythmical Signal Output
rons of the spinal cord. The rates at which these cells
Many neuronal circuits emit rhythmical output
emit impulses can be increased by excitatory signals or
signals—for instance, a rhythmical respiratory signal
decreased by inhibitory signals; inhibitory signals often
originates in the respiratory centers of the medulla and
can decrease the rate of firing to zero.
pons. This respiratory rhythmical signal continues
throughout life. Other rhythmical signals, such as those
Continuous Signals Emitted from Reverberating Circuits as
that cause scratching movements by the hind leg of a
a Means for Transmitting Information. A reverberating
dog or the walking movements of any animal, require
circuit that does not fatigue enough to stop reverber-
input stimuli into the respective circuits to initiate the
ation is a source of continuous impulses. And excita-
rhythmical signals.
tory impulses entering the reverberating pool can
All or almost all rhythmical signals that have been
increase the output signal, whereas inhibition can
studied experimentally have been found to result from
decrease or even extinguish the signal.
reverberating circuits or a succession of sequential
Figure 46-16 shows a continuous output signal from
reverberating circuits that feed excitatory or inhibitory
a pool of neurons. The pool may be emitting impulses
signals in a circular pathway from one neuronal pool
because of intrinsic neuronal excitability or as a result
to the next.
Chapter 46
Sensory Receptors, Neuronal Circuits for Processing Information
583
Flexor reflexes-decremental responses
Stimulus
50
40
30
20
10
0
0
15
30
45
60
Seconds
Increasing carotid
body stimulation
Figure 46-18
Figure 46-17
Successive flexor reflexes showing fatigue of conduction through
the reflex pathway.
The rhythmical output of summated nerve impulses from the
respiratory center, showing that progressively increasing stimula-
tion of the carotid body increases both the intensity and the fre-
quency of the phrenic nerve signal to the diaphragm to increase
Inhibitory Circuits as a Mechanism for
respiration.
Stabilizing Nervous System Function
Two types of inhibitory circuits in widespread areas
of the brain help prevent excessive spread of signals:
Excitatory or inhibitory signals can also increase
(1) inhibitory feedback circuits that return from the
or decrease the amplitude of the rhythmical signal
termini of pathways back to the initial excitatory
output. Figure 46-17, for instance, shows changes in
neurons of the same pathways—these circuits occur in
the respiratory signal output in the phrenic nerve.
virtually all sensory nervous pathways and inhibit
When the carotid body is stimulated by arterial oxygen
either the input neurons or the intermediate neurons
deficiency, both the frequency and the amplitude of
in the sensory pathway when the termini become
the respiratory rhythmical output signal increase
overly excited; and (2) some neuronal pools that exert
progressively.
gross inhibitory control over widespread areas of the
brain—for instance, many of the basal ganglia exert
inhibitory influences throughout the muscle control
Instability and Stability
system.
of Neuronal Circuits
Almost every part of the brain connects either directly
Synaptic Fatigue as a Means for
or indirectly with every other part, and this creates a
Stabilizing the Nervous System
serious problem. If the first part excites the second, the
second the third, the third the fourth, and so on until
Synaptic fatigue means simply that synaptic transmis-
finally the signal re-excites the first part, it is clear that
sion becomes progressively weaker the more pro-
an excitatory signal entering any part of the brain
longed and more intense the period of excitation.
would set off a continuous cycle of re-excitation of all
Figure 46-18 shows three successive records of a flexor
parts. If this should occur, the brain would be inun-
reflex elicited in an animal caused by inflicting pain in
dated by a mass of uncontrolled reverberating
the footpad of the paw. Note in each record that the
signals—signals that would be transmitting no infor-
strength of contraction progressively “decrements”—
mation but, nevertheless, would be consuming the cir-
that is, its strength diminishes; much of this effect is
cuits of the brain so that none of the informational
caused by fatigue of synapses in the flexor reflex
signals could be transmitted. Such an effect occurs in
circuit. Furthermore, the shorter the interval between
widespread areas of the brain during epileptic seizures.
successive flexor reflexes, the less the intensity of the
How does the central nervous system prevent this
subsequent reflex response.
from happening all the time? The answer lies mainly
in two basic mechanisms that function throughout the
Automatic Short-Term Adjustment of Pathway Sensitivity by the
central nervous system: (1) inhibitory circuits and (2)
Fatigue Mechanism. Now let us apply this phenomenon
fatigue of synapses.
of fatigue to other pathways in the brain. Those that
584
Unit IX The Nervous System: A. General Principles and Sensory Physiology
are overused usually become fatigued, so that their
Baev KV: Biological Neural Networks. Boston: Birkhauser,
sensitivities decrease. Conversely, those that are
1998.
underused become rested, and their sensitivities
Basar E: Brain Function and Oscillations. Berlin: Springer,
1998.
increase. Thus, fatigue and recovery from fatigue con-
Fain GL, Matthews HR, Cornwall MC, Koutalos Y: Adapta-
stitute an important short-term means of moderating
tion in vertebrate photoreceptors. Physiol Rev 81:117,
the sensitivities of the different nervous system cir-
2001.
cuits. These help to keep the circuits operating in a
Gandevia SC: Spinal and supraspinal factors in human
range of sensitivity that allows effective function.
muscle fatigue. Physiol Rev 81:1725, 2001.
Gebhart GF: Descending modulation of pain. Neurosci
Long-Term Changes in Synaptic Sensitivity Caused by Automatic
Biobehav Rev 27:729, 2004.
Downregulation or Upregulation of Synaptic Receptors. The
Hamill OP, Martinac B: Molecular basis of mechanotrans-
long-term sensitivities of synapses can be changed
duction in living cells. Physiol Rev 81:685, 2001.
tremendously by upregulating the number of receptor
Ivry RB, Robertson LC: The Two Sides of Perception. Cam-
bridge, MA: MIT Press, 1998.
proteins at the synaptic sites when there is underac-
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
tivity, and downregulating the receptors when there is
Science, 4th ed. New York: McGraw-Hill, 2000.
overactivity. The mechanism for this is the following:
Krupa B, Liu G: Does the fusion pore contribute to synap-
Receptor proteins are being formed constantly by the
tic plasticity? Trends Neurosci 27:62, 2004.
endoplasmic reticular-Golgi apparatus system and are
McLachlan EM: Transmission of signals through sympa-
constantly being inserted into the receptor neuron
thetic ganglia—modulation, integration or simply distri-
synaptic membrane. However, when the synapses are
bution? Acta Physiol Scand 177:227, 2003.
overused so that excesses of transmitter substance
Mombaerts P: Genes and ligands for odorant, vomeronasal
combine with the receptor proteins, many of these
and taste receptors. Nat Rev Neurosci 5:263, 2004.
receptors are inactivated and removed from the synap-
Palmer MJ, von Gersdorff H: Phasic transmitter release from
tonic neurons. Neuron 35:600, 2002.
tic membrane.
Pearson KG: Neural adaptation in the generation of rhyth-
It is indeed fortunate that upregulation and down-
mic behavior. Annu Rev Physiol 62:723, 2000.
regulation of receptors, as well as other control mech-
Renteria RC, Johnson J, Copenhagen DR: Need rods? Get
anisms for adjusting synaptic sensitivity, continually
glycine receptors and taurine. Neuron 41:839, 2004.
adjust the sensitivity in each circuit to almost the exact
Richerson GB, Wu Y: Dynamic equilibrium of neurotrans-
level required for proper function. Think for a moment
mitter transporters: not just for reuptake anymore. J
how serious it would be if the sensitivities of only a few
Neurophysiol 90:1363, 2003.
of these circuits were abnormally high; one might then
Schwartz EA: Transport-mediated synapses in the retina.
expect almost continual muscle cramps, seizures, psy-
Physiol Rev 82:875, 2002.
chotic disturbances, hallucinations, mental tension,
Shen K: Molecular mechanisms of target specificity during
synapse formation. Curr Opin Neurobiol 14:83, 2004.
or other nervous disorders. But fortunately, the auto-
Williams JT, Christie MJ, Manzoni O: Cellular and synaptic
matic controls normally readjust the sensitivities of the
adaptations mediating opioid dependence. Physiol Rev
circuits back to controllable ranges of reactivity any
81:299, 2001.
time the circuits begin to be too active or too
depressed.
References
Buzsaki G: Large-scale recording of neuronal ensembles.
Nat Neurosci 7:446, 2004.
C H A P T E R
4
7
Somatic Sensations: I. General
Organization, the Tactile and
Position Senses
The somatic senses are the nervous mechanisms that
collect sensory information from all over the body.
These senses are in contradistinction to the special
senses, which mean specifically vision, hearing,
smell, taste, and equilibrium.
CLASSIFICATION OF SOMATIC SENSES
The somatic senses can be classified into three physiologic types: (1) the
mechanoreceptive somatic senses, which include both tactile and position sensa-
tions that are stimulated by mechanical displacement of some tissue of the body;
(2) the thermoreceptive senses, which detect heat and cold; and (3) the pain
sense, which is activated by any factor that damages the tissues.
This chapter deals with the mechanoreceptive tactile and position senses.
Chapter 48 discusses the thermoreceptive and pain senses. The tactile senses
include touch, pressure, vibration, and tickle senses, and the position senses
include static position and rate of movement senses.
Other Classifications of Somatic Sensations. Somatic sensations are also often
grouped together in other classes, as follows.
Exteroreceptive sensations are those from the surface of the body. Proprio-
ceptive sensations are those having to do with the physical state of the body,
including position sensations, tendon and muscle sensations, pressure sensations
from the bottom of the feet, and even the sensation of equilibrium (which is
often considered a “special” sensation rather than a somatic sensation).
Visceral sensations are those from the viscera of the body; in using this term,
one usually refers specifically to sensations from the internal organs.
Deep sensations are those that come from deep tissues, such as from fasciae,
muscles, and bone. These include mainly “deep” pressure, pain, and vibration.
Detection and Transmission of
Tactile Sensations
Interrelations Among the Tactile Sensations of Touch, Pressure, and Vibration. Although
touch, pressure, and vibration are frequently classified as separate sensations,
they are all detected by the same types of receptors. There are three
principal differences among them: (1) touch sensation generally results from
stimulation of tactile receptors in the skin or in tissues immediately beneath the
skin; (2) pressure sensation generally results from deformation of deeper
tissues; and
(3) vibration sensation results from rapidly repetitive sensory
signals, but some of the same types of receptors as those for touch and pressure
are used.
Tactile Receptors. There are at least six entirely different types of tactile recep-
tors, but many more similar to these also exist. Some were shown in Figure 46-1
of the previous chapter; their special characteristics are the following.
585
586
Unit IX The Nervous System: A. General Principles and Sensory Physiology
First, some free nerve endings, which are found
causes the epithelium at this point to protrude
everywhere in the skin and in many other tissues, can
outward, thus creating a dome and constituting an
detect touch and pressure. For instance, even light
extremely sensitive receptor. Also note that the entire
contact with the cornea of the eye, which contains no
group of Merkel’s discs is innervated by a single large
other type of nerve ending besides free nerve endings,
myelinated nerve fiber (type Ab). These receptors,
can nevertheless elicit touch and pressure sensations.
along with the Meissner’s corpuscles discussed earlier,
Second, a touch receptor with great sensitivity is the
play extremely important roles in localizing touch
Meissner’s corpuscle (illustrated in Figure 46-1), an
sensations to specific surface areas of the body and in
elongated encapsulated nerve ending of a large (type
determining the texture of what is felt.
Ab) myelinated sensory nerve fiber. Inside the capsu-
Fourth, slight movement of any hair on the body
lation are many branching terminal nerve filaments.
stimulates a nerve fiber entwining its base. Thus, each
These corpuscles are present in the nonhairy parts of
hair and its basal nerve fiber, called the hair end-organ,
the skin and are particularly abundant in the finger-
are also a touch receptor. This receptor adapts readily
tips, lips, and other areas of the skin where one’s ability
and, like Meissner’s corpuscles, detects mainly
(a)
to discern spatial locations of touch sensations is
movement of objects on the surface of the body or (b)
highly developed. Meissner’s corpuscles adapt in a
initial contact with the body.
fraction of a second after they are stimulated, which
Fifth, located in the deeper layers of the skin and
means that they are particularly sensitive to movement
also in still deeper internal tissues are many Ruffini’s
of objects over the surface of the skin as well as to low-
end-organs, which are multibranched, encapsulated
frequency vibration.
endings, as shown in Figure 46-1. These endings adapt
Third, the fingertips and other areas that contain
very slowly and, therefore, are important for signaling
large numbers of Meissner’s corpuscles usually also
continuous states of deformation of the tissues, such as
contain large numbers of expanded tip tactile receptors,
heavy prolonged touch and pressure signals. They are
one type of which is Merkel’s discs, shown in Figure
also found in joint capsules and help to signal the
47-1. The hairy parts of the skin also contain moder-
degree of joint rotation.
ate numbers of expanded tip receptors, even though
Sixth, pacinian corpuscles, which were discussed in
they have almost no Meissner’s corpuscles. These
detail in Chapter 46, lie both immediately beneath the
receptors differ from Meissner’s corpuscles in that
skin and deep in the fascial tissues of the body. They
they transmit an initially strong but partially adapting
are stimulated only by rapid local compression of the
signal and then a continuing weaker signal that adapts
tissues because they adapt in a few hundredths of a
only slowly. Therefore, they are responsible for giving
second. Therefore, they are particularly important for
steady-state signals that allow one to determine con-
detecting tissue vibration or other rapid changes in the
tinuous touch of objects against the skin.
mechanical state of the tissues.
Merkel’s discs are often grouped together in a
receptor organ called the Iggo dome receptor, which
Transmission of Tactile Signals in Peripheral Nerve Fibers.
projects upward against the underside of the epithe-
Almost all specialized sensory receptors, such as
lium of the skin, as also shown in Figure 47-1. This
Meissner’s corpuscles, Iggo dome receptors, hair
E
FF
C
Figure 47-1
Iggo dome receptor. Note the
CF
multiple numbers of Merkel’s
discs connecting to a single large
myelinated fiber and abutting
A
tightly the undersurface of the
AA
epithelium.
(From Iggo A, Muir
AR: The structure and function of
a slowly adapting touch corpus-
10 mm
cle in hairy skin. J Physiol 200:
763, 1969.)
Chapter 47
Somatic Sensations: I. General Organization, the Tactile and Position Senses
587
receptors, pacinian corpuscles, and Ruffini’s endings,
Sensory Pathways for
transmit their signals in type Ab nerve fibers that have
Transmitting Somatic
transmission velocities ranging from 30 to 70 m/sec.
Signals into the Central
Conversely, free nerve ending tactile receptors trans-
mit signals mainly by way of the small type Ad myeli-
Nervous System
nated fibers that conduct at velocities of only 5 to
30 m/sec.
Some tactile free nerve endings transmit by way of
Almost all sensory information from the somatic seg-
type C unmyelinated fibers at velocities from a frac-
ments of the body enters the spinal cord through the
tion of a meter up to 2 m/sec; these send signals into
dorsal roots of the spinal nerves. However, from
the spinal cord and lower brain stem, probably sub-
the entry point into the cord and then to the brain, the
serving mainly the sensation of tickle.
sensory signals are carried through one of two alter-
Thus, the more critical types of sensory signals—
native sensory pathways: (1) the dorsal column-medial
those that help to determine precise localization on
lemniscal system or (2) the anterolateral system. These
the skin, minute gradations of intensity, or rapid
two systems come back together partially at the level
changes in sensory signal intensity—are all transmit-
of the thalamus.
ted in more rapidly conducting types of sensory nerve
The dorsal column-medial lemniscal system, as its
fibers. Conversely, the cruder types of signals, such as
name implies, carries signals upward to the medulla of
crude pressure, poorly localized touch, and especially
the brain mainly in the dorsal columns of the cord.
tickle, are transmitted by way of much slower, very
Then, after the signals synapse and cross to the oppo-
small nerve fibers that require much less space in the
site side in the medulla, they continue upward through
nerve bundle than the fast fibers.
the brain stem to the thalamus by way of the medial
lemniscus.
Conversely, signals in the anterolateral system,
Detection of Vibration
immediately after entering the spinal cord from the
dorsal spinal nerve roots, synapse in the dorsal
All tactile receptors are involved in detection of vibra-
horns of the spinal gray matter, then cross to the oppo-
tion, although different receptors detect different fre-
site side of the cord and ascend through the anterior
quencies of vibration. Pacinian corpuscles can detect
and lateral white columns of the cord. They terminate
signal vibrations from 30 to 800 cycles per second
at all levels of the lower brain stem and in the
because they respond extremely rapidly to minute and
thalamus.
rapid deformations of the tissues, and they also trans-
The dorsal column-medial lemniscal system is com-
mit their signals over type Ab nerve fibers, which can
posed of large, myelinated nerve fibers that transmit
transmit as many as 1000 impulses per second. Low-
signals to the brain at velocities of 30 to 110 m/sec,
frequency vibrations from 2 up to 80 cycles per second,
whereas the anterolateral system is composed of
in contrast, stimulate other tactile receptors, especially
smaller myelinated fibers that transmit signals at
Meissner’s corpuscles, which are less rapidly adapting
velocities ranging from a few meters per second up to
than pacinian corpuscles.
40 m/sec.
Another difference between the two systems is
that the dorsal column-medial lemniscal system has a
TICKLE AND ITCH
high degree of spatial orientation of the nerve fibers
with respect to their origin, while the anterolateral
Neurophysiologic studies have demonstrated the exis-
system has much less spatial orientation. These
tence of very sensitive, rapidly adapting mechano-
differences immediately characterize the types of
receptive free nerve endings that elicit only the tickle
sensory information that can be transmitted by the
and itch sensations. Furthermore, these endings are
two systems. That is, sensory information that must
found almost exclusively in superficial layers of the
be transmitted rapidly and with temporal and
skin, which is also the only tissue from which the tickle
spatial fidelity is transmitted mainly in the dorsal
and itch sensations usually can be elicited. These sen-
column-medial lemniscal system; that which does not
sations are transmitted by very small type C, unmyeli-
need to be transmitted rapidly or with great spatial
nated fibers similar to those that transmit the aching,
fidelity is transmitted mainly in the anterolateral
slow type of pain.
system.
The purpose of the itch sensation is presumably to
The anterolateral system has a special capability
call attention to mild surface stimuli such as a flea
that the dorsal system does not have: the ability to
crawling on the skin or a fly about to bite, and the
transmit a broad spectrum of sensory modalities—
elicited signals then activate the scratch reflex or other
pain, warmth, cold, and crude tactile sensations;
maneuvers that rid the host of the irritant. Itch can
most of these are discussed in detail in Chapter 48.
be relieved by scratching if this removes the irritant
The dorsal system is limited to discrete types of
or if the scratch is strong enough to elicit pain. The
mechanoreceptive sensations.
pain signals are believed to suppress the itch signals
With this differentiation in mind, we can now
in the cord by lateral inhibition, as described in
list the types of sensations transmitted in the two
Chapter 48.
systems.
588
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Dorsal Column-Medial
then upward in the dorsal column, proceeding by way
Lemniscal System
of the dorsal column pathway all the way to the brain.
The lateral branch enters the dorsal horn of the cord
1. Touch sensations requiring a high degree of
gray matter, then divides many times to provide termi-
localization of the stimulus
nals that synapse with local neurons in the intermediate
2. Touch sensations requiring transmission of fine
and anterior portions of the cord gray matter. These
gradations of intensity
local neurons in turn serve three functions: (1) A major
3. Phasic sensations, such as vibratory sensations
share of them give off fibers that enter the dorsal
4. Sensations that signal movement against the skin
columns of the cord and then travel upward to the brain.
5. Position sensations from the joints
(2) Many of the fibers are very short and terminate
6. Pressure sensations having to do with fine degrees
locally in the spinal cord gray matter to elicit local spinal
of judgment of pressure intensity
cord reflexes, which are discussed in Chapter 54. (3)
Others give rise to the spinocerebellar tracts, which we
will discuss in Chapter 56 in relation to the function of
Anterolateral System
the cerebellum.
1. Pain
The Dorsal Column-Medial Lemniscal Pathway. Note in Figure
2. Thermal sensations, including both warmth and
47-3 that nerve fibers entering the dorsal columns
cold sensations
pass uninterrupted up to the dorsal medulla, where
3. Crude touch and pressure sensations capable only
they synapse in the dorsal column nuclei (the cuneate
of crude localizing ability on the surface of the
and gracile nuclei). From there, second-order neurons
body
4. Tickle and itch sensations
5. Sexual sensations
Cortex
Transmission in the
Dorsal Column-Medial
Lemniscal System
Anatomy of the Dorsal
Column-Medial Lemniscal System
On entering the spinal cord through the spinal nerve
dorsal roots, the large myelinated fibers from the spe-
cialized mechanoreceptors divide almost immediately
Internal
capsule
to form a medial branch and a lateral branch, shown by
the right-hand fiber entering through the spinal root in
Ventrobasal
Figure 47-2. The medial branch turns medially first and
complex
of thalamus
Spinal nerve
Lamina marginalis
Substantia gelatinosa
Dorsal
Medulla oblongata
Tract of
Lissauer
column
Medial lemniscus
Spinocervical
tract
I
Dorsal column nuclei
II
Dorsal
III
spinocerebellar
IV
Ascending branches of
tract
dorsal root fibers
V
VI
Spinal cord
VII
Ventral
Spinocervical tract
IX
VIII
spinocerebellar
tract
Dorsal root and spinal
Anterolateral
ganglion
spinothalamic
pathway
Figure 47-3
Figure 47-2
The dorsal column-medial lemniscal pathway for transmitting crit-
Cross section of the spinal cord, showing the anatomy of the cord
ical types of tactile signals. (Modified from Ranson SW, Clark SL:
gray matter and of ascending sensory tracts in the white columns
Anatomy of the Nervous System. Philadelphia: WB Saunders Co,
of the spinal cord.
1959.)
Chapter 47
Somatic Sensations: I. General Organization, the Tactile and Position Senses
589
decussate immediately to the opposite side of the brain
medial areas of the complex. Because of the crossing
stem and continue upward through the medial lemnisci
of the medial lemnisci in the medulla, the left side of
to the thalamus. In this pathway through the brain stem,
the body is represented in the right side of the thala-
each medial lemniscus is joined by additional fibers
mus, and the right side of the body in the left side of
from the sensory nuclei of the trigeminal nerve; these
the thalamus.
fibers subserve the same sensory functions for the head
that the dorsal column fibers subserve for the body.
In the thalamus, the medial lemniscal fibers terminate
Somatosensory Cortex
in the thalamic sensory relay area, called the ventrobasal
complex. From the ventrobasal complex, third-order
nerve fibers project, as shown in Figure 47-4, mainly to
Before discussing the role of the cerebral cortex in
the postcentral gyrus of the cerebral cortex, which is
somatic sensation, we need to give an orientation to
called somatic sensory area I (as shown in Figure 47-6,
the various areas of the cortex. Figure 47-5 is a map
these fibers also project to a smaller area in the lateral
of the human cerebral cortex, showing that it is divided
parietal cortex called somatic sensory area II).
into about 50 distinct areas called Brodmann’s areas
based on histological structural differences. This map
Spatial Orientation of the Nerve Fibers in the
is important because virtually all neurophysiologists
Dorsal Column-Medial Lemniscal System
and neurologists use it to refer by number to many of
One of the distinguishing features of the dorsal
the different functional areas of the human cortex.
column-medial lemniscal system is a distinct spatial
Note in the figure the large central fissure (also
orientation of nerve fibers from the individual parts of
called central sulcus) that extends horizontally across
the body that is maintained throughout. For instance,
the brain. In general, sensory signals from all modali-
in the dorsal columns of the spinal cord, the fibers from
ties of sensation terminate in the cerebral cortex
the lower parts of the body lie toward the center of the
immediately posterior to the central fissure. And,
cord, whereas those that enter the cord at progres-
generally, the anterior half of the parietal lobe is
sively higher segmental levels form successive layers
concerned almost entirely with reception and inter-
laterally.
pretation of somatosensory signals. But the posterior
In the thalamus, distinct spatial orientation is still
half of the parietal lobe provides still higher levels of
maintained, with the tail end of the body represented
interpretation.
by the most lateral portions of the ventrobasal
Visual signals terminate in the occipital lobe, and
complex and the head and face represented by the
auditory signals in the temporal lobe.
Conversely, that portion of the cerebral cortex ante-
rior to the central fissure and constituting the poste-
rior half of the frontal lobe is called the motor cortex
POSTCENTRAL GYRUS
and is devoted almost entirely to control of muscle
Lower extremity
Trunk
contractions and body movements. A major share of
this motor control is in response to somatosensory
Upper
extremity
Central fissure
3
2
Face
4
7a
8
5
6
9
7a
1
Ventrobasal complex of thalamus
46
40
39
10
44
43
19
45
MESENCEPHALON
41
42
47
22
11
37
Spinothalamic tract
18
38
21
Lateral fissure
17
Medial lemniscus
20
Figure 47-4
Figure 47-5
Projection of the dorsal column-medial lemniscal system through
the thalamus to the somatosensory cortex. (Modified from Brodal
Structurally distinct areas, called Brodmann’s areas, of the human
A: Neurological Anatomy in Relation to Clinical Medicine. New
cerebral cortex. Note specifically areas 1, 2, and 3, which consti-
York: Oxford University Press, 1969, by permission of Oxford
tute primary somatosensory area I, and areas 5 and 7, which con-
University Press.)
stitute the somatosensory association area.
590
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Somatosensory
area I
Thigh
Somatosensory
Thorax
area II
Neck
Shoulder
Hand
Fingers
Tongue
Leg
Abdomen
Arm
Face
Upper lip
Lips
Lower lip
Teeth, gums, and jaw
Tongue
Figure 47-6
Pharynx
Two somatosensory cortical areas, somatosensory areas I and II.
Intra-abdominal
Figure 47-7
signals received from the sensory portions of the
cortex, which keep the motor cortex informed at each
Representation of the different areas of the body in somatosen-
instant about the positions and motions of the differ-
sory area I of the cortex. (From Penfield W, Rasmussen T: Cere-
ent body parts.
bral Cortex of Man: A Clinical Study of Localization of Function.
New York: Hafner, 1968.)
Somatosensory Areas I and II. Figure 47-6 shows two sep-
arate sensory areas in the anterior parietal lobe called
somatosensory area I and somatosensory area II. The
postcentral gyrus of the human cerebral cortex (in
reason for this division into two areas is that a distinct
Brodmann’s areas 3, 1, and 2).
and separate spatial orientation of the different
Figure 47-7 shows a cross section through the brain
parts of the body is found in each of these two
at the level of the postcentral gyrus, demonstrating
areas. However, somatosensory area I is so much
representations of the different parts of the body in
more extensive and so much more important than
separate regions of somatosensory area I. Note,
somatosensory area II that in popular usage, the term
however, that each lateral side of the cortex receives
“somatosensory cortex” almost always means area I.
sensory information almost exclusively from the oppo-
Somatosensory area I has a high degree of localiza-
site side of the body.
tion of the different parts of the body, as shown by the
Some areas of the body are represented by large
names of virtually all parts of the body in Figure 47-6.
areas in the somatic cortex—the lips the greatest of all,
By contrast, localization is poor in somatosensory area
followed by the face and thumb—whereas the trunk
II, although roughly, the face is represented anteriorly,
and lower part of the body are represented by rela-
the arms centrally, and the legs posteriorly.
tively small areas. The sizes of these areas are directly
Little is known about the function of somatosensory
proportional to the number of specialized sensory
area II. It is known that signals enter this area from
receptors in each respective peripheral area of the
the brain stem, transmitted upward from both sides of
body. For instance, a great number of specialized nerve
the body. In addition, many signals come secondarily
endings are found in the lips and thumb, whereas only
from somatosensory area I as well as from other
a few are present in the skin of the body trunk.
sensory areas of the brain, even from the visual and
Note also that the head is represented in the most
auditory areas. Projections from somatosensory area I
lateral portion of somatosensory area I, and the lower
are required for function of somatosensory area II.
part of the body is represented medially.
However, removal of parts of somatosensory area II
has no apparent effect on the response of neurons in
Layers of the Somatosensory Cortex and
somatosensory area I. Thus, much of what we know
Their Function
about somatic sensation appears to be explained by
The cerebral cortex contains six layers of neurons,
the functions of somatosensory area I.
beginning with layer I next to the brain surface and
extending progressively deeper to layer VI, shown in
Spatial Orientation of Signals from Different Parts of the Body
Figure 47-8. As would be expected, the neurons in
in Somatosensory Area I. Somatosensory area I lies
each layer perform functions different from those
immediately behind the central fissure, located in the
in other layers. Some of these functions are:
Chapter 47
Somatic Sensations: I. General Organization, the Tactile and Position Senses
591
The Sensory Cortex Is Organized in Vertical
I
Columns of Neurons; Each Column Detects a
Different Sensory Spot on the Body with a
Specific Sensory Modality
II
Functionally, the neurons of the somatosensory cortex
are arranged in vertical columns extending all the way
through the six layers of the cortex, each column
having a diameter of 0.3 to 0.5 millimeter and con-
III
taining perhaps 10,000 neuronal cell bodies. Each of
these columns serves a single specific sensory modal-
ity, some columns responding to stretch receptors
around joints, some to stimulation of tactile hairs,
IV
others to discrete localized pressure points on the skin,
and so forth. At layer IV, where the input sensory
signals first enter the cortex, the columns of neurons
function almost entirely separately from one another.
V
At other levels of the columns, interactions occur that
initiate analysis of the meanings of the sensory signals.
In the most anterior 5 to 10 millimeters of the post-
central gyrus, located deep in the central fissure in
VIa
Brodmann’s area 3a, an especially large share of the
vertical columns respond to muscle, tendon, and joint
stretch receptors. Many of the signals from these
sensory columns then spread anteriorly, directly to
VIb
the motor cortex located immediately forward of the
central fissure. These signals play a major role in
controlling the effluent motor signals that activate
sequences of muscle contraction.
As one moves posteriorly in somatosensory area I,
Figure 47-8
more and more of the vertical columns respond to
Structure of the cerebral cortex, showing I, molecular layer; II,
slowly adapting cutaneous receptors, and then still
external granular layer; III, layer of small pyramidal cells; IV, inter-
farther posteriorly, greater numbers of the columns are
nal granular layer; V, large pyramidal cell layer; and VI, layer of
sensitive to deep pressure.
fusiform or polymorphic cells. (From Ranson SW, Clark SL [after
In the most posterior portion of somatosensory area
Brodmann]: Anatomy of the Nervous System. Philadelphia: WB
Saunders, 1959.)
I, about 6 per cent of the vertical columns respond only
when a stimulus moves across the skin in a particular
direction. Thus, this is a still higher order of interpre-
1. The incoming sensory signal excites neuronal
tation of sensory signals; the process becomes even
layer IV first; then the signal spreads toward the
more complex as the signals spread farther backward
surface of the cortex and also toward deeper
from somatosensory area I into the parietal cortex, an
layers.
area called the somatosensory association area, as we
2. Layers I and II receive diffuse, nonspecific input
discuss subsequently.
signals from lower brain centers that facilitate
specific regions of the cortex; this system is
Functions of Somatosensory Area I
described in Chapter 57. This input mainly
Widespread bilateral excision of somatosensory area I
controls the overall level of excitability of the
causes loss of the following types of sensory judgment:
respective regions stimulated.
1. The person is unable to localize discretely the
3. The neurons in layers II and III send axons to
different sensations in the different parts of the
related portions of the cerebral cortex on the
body. However, he or she can localize these
opposite side of the brain through the corpus
sensations crudely, such as to a particular hand, to
callosum.
a major level of the body trunk, or to one of the
4. The neurons in layers V and VI send axons to the
legs. Thus, it is clear that the brain stem, thalamus,
deeper parts of the nervous system. Those in layer
or parts of the cerebral cortex not normally
V are generally larger and project to more distant
considered to be concerned with somatic
areas, such as to the basal ganglia, brain stem,
sensations can perform some degree of
and spinal cord where they control signal
localization.
transmission. From layer VI, especially large
2. The person is unable to judge critical degrees of
numbers of axons extend to the thalamus,
pressure against the body.
providing signals from the cerebral cortex that
3. The person is unable to judge the weights of
interact with and help to control the excitatory
objects.
levels of incoming sensory signals entering the
4. The person is unable to judge shapes or forms of
thalamus.
objects. This is called astereognosis.
592
Unit IX The Nervous System: A. General Principles and Sensory Physiology
5. The person is unable to judge texture of materials
because this type of judgment depends on highly
critical sensations caused by movement of the
Strong stimulus
fingers over the surface to be judged.
Note that in the list nothing has been said about loss
of pain and temperature sense. In specific absence
of only somatosensory area I, appreciation of these
sensory modalities is still preserved both in quality and
Moderate
stimulus
intensity. But the sensations are poorly localized, indi-
cating that pain and temperature localization depend
greatly on the topographical map of the body in
somatosensory area I to localize the source.
Weak
stimulus
Somatosensory Association Areas
Brodmann’s areas 5 and 7 of the cerebral cortex,
Cortex
located in the parietal cortex behind somatosensory
area I (see Figure 47-5), play important roles in deci-
phering deeper meanings of the sensory information
in the somatosensory areas. Therefore, these areas are
called somatosensory association areas.
Electrical stimulation in a somatosensory associa-
Thalamus
tion area can occasionally cause an awake person to
experience a complex body sensation, sometimes even
the “feeling” of an object such as a knife or a ball.
Therefore, it seems clear that the somatosensory asso-
ciation area combines information arriving from mul-
Dorsal column nuclei
tiple points in the primary somatosensory area to
decipher its meaning. This also fits with the anatomi-
cal arrangement of the neuronal tracts that enter the
somatosensory association area because it receives
Single-point stimulus on skin
signals from (1) somatosensory area I, (2) the ven-
trobasal nuclei of the thalamus, (3) other areas of the
thalamus, (4) the visual cortex, and (5) the auditory
cortex.
Figure 47-9
Transmission of a pinpoint stimulus signal to the cerebral cortex.
Effect of Removing the Somatosensory Association Area—
Amorphosynthesis. When the somatosensory association
area is removed on one side of the brain, the person
organization of the neuronal circuit of the spinal cord
loses ability to recognize complex objects and complex
dorsal column pathway, demonstrating that at each
forms felt on the opposite side of the body. In addi-
synaptic stage, divergence occurs. The upper curves of
tion, he or she loses most of the sense of form of his
the figure show that the cortical neurons that discharge
or her own body or body parts on the opposite side.
to the greatest extent are those in a central part of the
In fact, the person is mainly oblivious to the opposite
cortical “field” for each respective receptor. Thus, a
side of the body—that is, forgets that it is there.
weak stimulus causes only the centralmost neurons to
Therefore, he or she also often forgets to use the other
fire. A stronger stimulus causes still more neurons to
side for motor functions as well. Likewise, when
fire, but those in the center discharge at a considerably
feeling objects, the person tends to recognize only
more rapid rate than do those farther away from the
one side of the object and forgets that the other side
center.
even exists. This complex sensory deficit is called
amorphosynthesis.
Two-Point Discrimination. A method frequently used to
test tactile discrimination is to determine a person’s
so-called “two-point” discriminatory ability. In this
Overall Characteristics of Signal
test, two needles are pressed lightly against the skin at
Transmission and Analysis
the same time, and the person determines whether two
in the Dorsal Column-Medial
points of stimulus are felt or one point. On the tips of
Lemniscal System
the fingers, a person can distinguish two separate
points even when the needles are as close together as
Basic Neuronal Circuit in the Dorsal Column-Medial Lemniscal
1 to 2 millimeters. However, on the person’s back,
System. The lower part of Figure 47-9 shows the basic
the needles must usually be as far apart as 30 to 70
Chapter 47
Somatic Sensations: I. General Organization, the Tactile and Position Senses
593
excitatory signal, short lateral pathways transmit
inhibitory signals to the surrounding neurons. That is,
these signals pass through additional interneurons that
secrete an inhibitory transmitter.
The importance of lateral inhibition is that it blocks
lateral spread of the excitatory signals and, therefore,
increases the degree of contrast in the sensory pattern
perceived in the cerebral cortex.
In the case of the dorsal column system, lateral
inhibitory signals occur at each synaptic level—for
instance, in
(1) the dorsal column nuclei of the
medulla, (2) the ventrobasal nuclei of the thalamus,
and (3) the cortex itself. At each of these levels, the
lateral inhibition helps to block lateral spread of the
excitatory signal. As a result, the peaks of excitation
stand out, and much of the surrounding diffuse stimu-
Cortex
lation is blocked. This effect is demonstrated by the
two red curves in Figure 47-10, showing complete
separation of the peaks when the intensity of lateral
inhibition is great.
Two adjacent points
Transmission of Rapidly Changing and Repetitive Sensations.
strongly stimulated
The dorsal column system also is of particular impor-
tance in apprising the sensorium of rapidly changing
peripheral conditions. Based on recorded action
potentials, this system can recognize changing stimuli
Figure 47-10
that occur in as little as 1/400 of a second.
Transmission of signals to the cortex from two adjacent pinpoint
stimuli. The blue curve represents the pattern of cortical stimula-
Vibratory Sensation. Vibratory signals are rapidly
tion without “surround” inhibition, and the two red curves repre-
repetitive and can be detected as vibration up to 700
sent the pattern when “surround” inhibition does occur.
cycles per second. The higher-frequency vibratory
signals originate from the pacinian corpuscles in the
millimeters before two separate points can be
skin and deeper tissues, but lower-frequency signals
detected. The reason for this difference is the different
(below about 200 per second) can originate from
numbers of specialized tactile receptors in the two
Meissner’s corpuscles as well. These signals are trans-
areas.
mitted only in the dorsal column pathway. For this
Figure 47-10 shows the mechanism by which the
reason, application of vibration (e.g., from a “tuning
dorsal column pathway (as well as all other sensory
fork”) to different peripheral parts of the body is an
pathways) transmits two-point discriminatory infor-
important tool used by neurologists for testing func-
mation. This figure shows two adjacent points on the
tional integrity of the dorsal columns.
skin that are strongly stimulated as well as the areas
of the somatosensory cortex (greatly enlarged) that
are excited by signals from the two stimulated points.
Interpretation of Sensory
The blue curve shows the spatial pattern of cortical
Stimulus Intensity
excitation when both skin points are stimulated simul-
The ultimate goal of most sensory stimulation is to
taneously. Note that the resultant zone of excitation
apprise the psyche of the state of the body and its sur-
has two separate peaks. These two peaks, separated by
roundings. Therefore, it is important that we discuss
a valley, allow the sensory cortex to detect the pres-
briefly some of the principles related to transmission of
ence of two stimulatory points, rather than a single
sensory stimulus intensity to the higher levels of the
point. The capability of the sensorium to distinguish
nervous system.
this presence of two points of stimulation is strongly
The first question that comes to mind is, how is it pos-
influenced by another mechanism, lateral inhibition, as
sible for the sensory system to transmit sensory experi-
explained in the next section.
ences of tremendously varying intensities? For instance,
the auditory system can detect the weakest possible
whisper but can also discern the meanings of an explo-
Effect of Lateral Inhibition (Also Called Surround Inhibition) to
sive sound, even though the sound intensities of these
Increase the Degree of Contrast in the Perceived Spatial
two experiences can vary more than 10 billion times; the
Pattern. As pointed out in Chapter 46, virtually every
eyes can see visual images with light intensities that vary
sensory pathway, when excited, gives rise simultane-
as much as a half million times; and the skin can detect
ously to lateral inhibitory signals; these spread to the
pressure differences of 10,000 to 100,000 times.
sides of the excitatory signal and inhibit adjacent
As a partial explanation of these effects, Figure 46-4
neurons. For instance, consider an excited neuron
in the previous chapter shows the relation of the recep-
in a dorsal column nucleus. Aside from the central
tor potential produced by the pacinian corpuscle to the
594
Unit IX The Nervous System: A. General Principles and Sensory Physiology
intensity of the sensory stimulus. At low stimulus inten-
Fechner principle is still a good one to remember,
sity, slight changes in intensity increase the potential
because it emphasizes that the greater the background
markedly, whereas at high levels of stimulus intensity,
sensory intensity, the greater an additional change must
further increases in receptor potential are slight. Thus,
be for the psyche to detect the change.
the pacinian corpuscle is capable of accurately measur-
ing extremely minute changes in stimulus at low-
Power Law. Another attempt by physiopsychologists to
intensity levels, but at high-intensity levels, the change
find a good mathematical relation is the following
in stimulus must be much greater to cause the same
formula, known as the power law.
amount of change in receptor potential.
Interpreted signal strength = K • (Stimulus - k)y
The transduction mechanism for detecting sound by
the cochlea of the ear demonstrates still another
In this formula, the exponent y and the constants K and
method for separating gradations of stimulus intensity.
k are different for each type of sensation.
When sound stimulates a specific point on the basilar
When this power law relation is plotted on a graph
membrane, weak sound stimulates only those hair cells
using double logarithmic coordinates, as shown in
at the point of maximum sound vibration. But as the
Figure 47-11, and when appropriate quantitative values
sound intensity increases, many more hair cells in each
for the constants y, K, and k are found, a linear relation
direction farther away from the maximum vibratory
can be attained between interpreted stimulus strength
point also become stimulated. Thus, signals are trans-
and actual stimulus strength over a large range for
mitted over progressively increasing numbers of nerve
almost any type of sensory perception.
fibers, which is another mechanism by which stimulus
intensity is transmitted to the central nervous system.
This mechanism, plus the direct effect of stimulus inten-
Position Senses
sity on impulse rate in each nerve fiber, as well as several
other mechanisms, makes it possible for some sensory
The position senses are frequently also called proprio-
systems to operate reasonably faithfully at stimulus
ceptive senses. They can be divided into two subtypes:
intensity levels changing as much as millions of times.
(1) static position sense, which means conscious per-
Importance of the Tremendous Intensity Range of Sensory
ception of the orientation of the different parts of
Reception. Were it not for the tremendous intensity
the body with respect to one another, and (2) rate of
range of sensory reception that we can experience, the
movement sense, also called kinesthesia or dynamic
various sensory systems would more often than not be
proprioception.
operating in the wrong range. This is demonstrated by
the attempts of most people, when taking photographs
Position Sensory Receptors. Knowledge of position, both
with a camera, to adjust the light exposure without using
static and dynamic, depends on knowing the degrees
a light meter. Left to intuitive judgment of light inten-
of angulation of all joints in all planes and their rates
sity, a person almost always overexposes the film on
of change. Therefore, multiple different types of recep-
bright days and greatly underexposes the film at twi-
tors help to determine joint angulation and are used
light. Yet, that person’s own eyes are capable of dis-
criminating with great detail visual objects in bright
sunlight or at twilight; the camera cannot do this without
very special manipulation because of the narrow criti-
cal range of light intensity required for proper exposure
of film.
500
200
Judgment of Stimulus Intensity
Weber-Fechner Principle—Detection of
“Ratio” of Stimulus
100
Strength. In the mid-1800s, Weber first and Fechner later
proposed the principle that gradations of stimulus
50
strength are discriminated approximately in proportion
to the logarithm of stimulus strength. That is, a person
already holding 30 grams weight in his or her hand can
20
barely detect an additional 1-gram increase in weight.
And, when already holding 300 grams, he or she can
10
barely detect a 10-gram increase in weight. Thus, in this
instance, the ratio of the change in stimulus strength
0
required for detection remains essentially constant,
0
10
100
1000
10,000
about 1 to 30, which is what the logarithmic principle
Stimulus strength (arbitrary units)
means. To express this mathematically.
Interpreted signal strength = Log (Stimulus)
+ Constant
Figure 47-11
More recently, it has become evident that the Weber-
Fechner principle is quantitatively accurate only for
Graphical demonstration of the “power law” relation between
higher intensities of visual, auditory, and cutaneous
actual stimulus strength and strength that the psyche interprets it
sensory experience and applies only poorly to most
to be. Note that the power law does not hold at either very weak
other types of sensory experience. Yet the Weber-
or very strong stimulus strengths.
Chapter 47
Somatic Sensations: I. General Organization, the Tactile and Position Senses
595
together for position sense. Both skin tactile receptors
maximally stimulated when the joint is at full rotation
and deep receptors near the joints are used. In the
and (2) those maximally stimulated when the joint is
case of the fingers, where skin receptors are in great
at minimal rotation. Thus, the signals from the indi-
abundance, as much as half of position recognition is
vidual joint receptors are used to tell the psyche how
believed to be detected through the skin receptors.
much each joint is rotated.
Conversely, for most of the larger joints of the body,
deep receptors are more important.
For determining joint angulation in mid ranges of
Transmission of Less Critical
motion, among the most important receptors are the
Sensory Signals in the
muscle spindles. They are also exceedingly important
in helping to control muscle movement, as we shall see
Anterolateral Pathway
in Chapter 54. When the angle of a joint is changing,
some muscles are being stretched while others are
The anterolateral pathway for transmitting sensory
loosened, and the net stretch information from the
signals up the spinal cord and into the brain, in con-
spindles is transmitted into the computational system
trast to the dorsal column pathway, transmits sensory
of the spinal cord and higher regions of the dorsal
signals that do not require highly discrete localization
column system for deciphering joint angulations.
of the signal source and do not require discrimination
At the extremes of joint angulation, stretch of the
of fine gradations of intensity. These types of signals
ligaments and deep tissues around the joints is an addi-
include pain, heat, cold, crude tactile, tickle, itch, and
tional important factor in determining position. Types
sexual sensations. In Chapter 48, pain and temperature
of sensory endings used for this are the pacinian cor-
sensations will be discussed specifically.
puscles, Ruffini’s endings, and receptors similar to the
Golgi tendon receptors found in muscle tendons.
Anatomy of the
The pacinian corpuscles and muscle spindles are
Anterolateral Pathway
especially adapted for detecting rapid rates of change.
It is likely that these are the receptors most responsi-
The spinal cord anterolateral fibers originate mainly in
ble for detecting rate of movement.
dorsal horn laminae I, IV, V, and VI (see Figure 47-2).
These laminae are where many of the dorsal root
Processing of Position Sense Information in the Dorsal
sensory nerve fibers terminate after entering the cord.
Column-Medial Lemniscal Pathway. Referring to Figure
As shown in Figure 47-13, the anterolateral fibers
47-12, one sees that thalamic neurons responding
cross immediately in the anterior commissure of the
to joint rotation are of two categories:
(1) those
cord to the opposite anterior and lateral white columns,
where they turn upward toward the brain by way of the
anterior spinothalamic and lateral spinothalamic tracts.
The upper terminus of the two spinothalamic tracts is
mainly twofold: (1) throughout the reticular nuclei of the
brain stem and (2) in two different nuclear complexes
of the thalamus, the ventrobasal complex and the
100
intralaminar nuclei. In general, the tactile signals are
transmitted mainly into the ventrobasal complex, ter-
minating in some of the same thalamic nuclei where the
80
dorsal column tactile signals terminate. From here, the
signals are transmitted to the somatosensory cortex
along with the signals from the dorsal columns.
60
#1
#4
Conversely, only a small fraction of the pain signals
project directly to the ventrobasal complex of the thal-
#2
#5
amus. Instead, most pain signals terminate in the retic-
40
#3
ular nuclei of the brain stem and from there are relayed
to the intralaminar nuclei of the thalamus where the
pain signals are further processed, as discussed in
20
greater detail in Chapter 48.
Characteristics of Transmission in the Anterolateral Pathway.
0
In general, the same principles apply to transmission
0
60
80
100
120
140
160
180
in the anterolateral pathway as in the dorsal
Degrees
column-medial lemniscal system, except for the fol-
lowing differences: (1) the velocities of transmission
are only one third to one half those in the dorsal
Figure 47-12
column-medial lemniscal system, ranging between 8
and 40 m/sec; (2) the degree of spatial localization of
Typical responses of five different thalamic neurons in the thala-
signals is poor; (3) the gradations of intensities are also
mic ventrobasal complex when the knee joint is moved through
far less accurate, most of the sensations being recog-
its range of motion. (Data from Mountcastle VB, Poggie GF, Werner
G: The relation of thalamic cell response to peripheral stimuli
nized in 10 to 20 gradations of strength, rather than as
varied over an intensive continuum. J Neurophysiol 26:807, 1963.)
many as 100 gradations for the dorsal column system;
596
Unit IX
The Nervous System: A. General Principles and Sensory Physiology
Cortex
C2
C2
C3
C3
C4
C5
C4
C5
T2
T2
T4
T3
T4
T5
T5
T4
T5
T6
Internal
T6
T7
T7
capsule
T8
T2
T8
T9
T9
Ventrobasal
T10
and intralaminar
T10
T12
nuclei of the
Mesencephalon
T11
T11
L3
thalamus
L5
T12
Medial lemniscus
S2
L1
L1
C6
S4&5
Medulla oblongata
L2
C8
L2
C7
S3
L3
L3
S2
Lateral
division of the
anterolateral
pathway
L4
Spinal cord
L4
Anterior
division of the
Dorsal root and spinal
anterolateral
ganglion
L5
pathway
L5
S1
L5
S1
L4
Figure 47-13
Figure 47-14
Anterior and lateral divisions of the anterolateral sensory pathway.
Dermatomes. (Modified from Grinker RR, Sahs AL: Neurology, 6th
ed. Springfield, IL: Charles C Thomas, 1966. Courtesy of Charles
C Thomas, Publisher, Ltd., Springfield, Illinois.)
and (4) the ability to transmit rapidly changing or
rapidly repetitive signals is poor.
Thus, it is evident that the anterolateral system is a
cruder type of transmission system than the dorsal
column-medial lemniscal system. Even so, certain
modalities of sensation are transmitted only in this
does return. Therefore, it must be assumed that the thal-
system and not at all in the dorsal column-medial lem-
amus (as well as other lower centers) has a slight ability
niscal system. They are pain, temperature, tickle, itch,
to discriminate tactile sensation, even though the thala-
and sexual sensations, in addition to crude touch and
mus normally functions mainly to relay this type of
pressure.
information to the cortex.
Conversely, loss of the somatosensory cortex has little
effect on one’s perception of pain sensation and only
Some Special Aspects of
a moderate effect on the perception of temperature.
Somatosensory Function
Therefore, there is much reason to believe that the
lower brain stem, the thalamus, and other associated
Function of the Thalamus
basal regions of the brain play dominant roles in dis-
crimination of these sensibilities. It is interesting that
in Somatic Sensation
these sensibilities appeared very early in the phyloge-
When the somatosensory cortex of a human being is
netic development of animals, whereas the critical
destroyed, that person loses most critical tactile sensi-
tactile sensibilities and the somatosensory cortex were
bilities, but a slight degree of crude tactile sensibility
late developments.
Chapter 47
Somatic Sensations: I. General Organization, the Tactile and Position Senses
597
Cortical Control of Sensory
Chen R, Cohen LG, Hallett M: Nervous system reorganiza-
Sensitivity—“Corticofugal” Signals
tion following injury. Neuroscience 111(4):761, 2002.
Cohen YE, Andersen RA: A common reference frame for
In addition to somatosensory signals transmitted from
movement plans in the posterior parietal cortex. Nat Rev
the periphery to the brain, corticofugal signals are trans-
Neurosci 3:553, 2002.
mitted in the backward direction from the cerebral
Craig AD: Pain mechanisms: labeled lines versus conver-
cortex to the lower sensory relay stations of the thala-
gence in central processing. Annu Rev Neurosci 26:1, 2003.
mus, medulla, and spinal cord; they control the intensity
Foeller E, Feldman DE: Synaptic basis for developmental
of sensitivity of the sensory input.
plasticity in somatosensory cortex. Curr Opin Neurobiol
Corticofugal signals are almost entirely inhibitory, so
14:89, 2004.
that when sensory input intensity becomes too great, the
Haines DE: Fundamental Neuroscience. New York:
corticofugal signals automatically decrease transmission
Churchill Livingstone, 1997.
in the relay nuclei. This does two things: First, it
Haines DE, Lancon JA: Review of Neuroscience. New York:
decreases lateral spread of the sensory signals into adja-
Churchill Livingstone, 2003.
cent neurons and, therefore, increases the degree of
Ivry RB, Spencer RM: The neural representation of time.
sharpness in the signal pattern. Second, it keeps the
Curr Opin Neurobiol 14:225, 2004.
sensory system operating in a range of sensitivity that is
Janig W, Baron R: Complex regional pain syndrome: mystery
not so low that the signals are ineffectual nor so high
explained? Lancet Neurol 2:687, 2003.
that the system is swamped beyond its capacity to dif-
Jeannerod M: The mechanism of self-recognition in humans.
ferentiate sensory patterns. This principle of corticofu-
Behav Brain Res 142:1, 2003.
gal sensory control is used by all sensory systems, not
Johnson KO: The roles and functions of cutaneous
only the somatic system, as explained in subsequent
mechanoreceptors. Curr Opin Neurobiol 11:455, 2001.
chapters.
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
Science, 4th ed. New York: McGraw-Hill, 2000.
Lumb BM: Hypothalamic and midbrain circuitry that dis-
Segmental Fields of Sensation—
tinguishes between escapable and inescapable pain. News
The Dermatomes
Physiol Sci 19:22, 2004.
Maravita A, Spence C, Driver J: Multisensory integration
Each spinal nerve innervates a “segmental field” of the
and the body schema: close to hand and within reach. Curr
skin called a dermatome. The different dermatomes are
Biol 13:R531, 2003.
shown in Figure 47-14. They are shown in the figure as
Pears S, Jackson SR: Cognitive neuroscience: vision and
if there were distinct borders between the adjacent der-
touch are constant companions. Curr Biol 14:R349, 2004.
matomes, which is far from true because much overlap
Petersen CC: The barrel cortex—integrating molecular, cel-
exists from segment to segment.
lular and systems physiology. Pflugers Arch 447:126, 2003.
The figure shows that the anal region of the body lies
Pouget A, Deneve S, Duhamel JR: A computational per-
in the dermatome of the most distal cord segment, der-
spective on the neural basis of multisensory spatial repre-
matome S5. In the embryo, this is the tail region and the
sentations. Nat Rev Neurosci 3:741, 2002.
most distal portion of the body. The legs originate
Sommer MA: The role of the thalamus in motor control.
embryologically from the lumbar and upper sacral seg-
Curr Opin Neurobiol 13:663, 2003.
ments (L2 to S3), rather than from the distal sacral seg-
Suga N, Ma X: Multiparametric corticofugal modulation and
ments, which is evident from the dermatomal map. One
plasticity in the auditory system. Nat Rev Neurosci 4:783,
can use a dermatomal map as shown in Figure 47-14 to
2003.
determine the level in the spinal cord at which a cord
Thaut MH: Neural basis of rhythmic timing networks in the
injury has occurred when the peripheral sensations are
human brain. Ann N Y Acad Sci 999:364, 2003.
disturbed by the injury.
References
Bosco G, Poppele RE: Proprioception from a spinocerebel-
lar perspective. Physiol Rev 81:539, 2001.
C H A P T E R
4
8
Somatic Sensations:
II. Pain, Headache, and
Thermal Sensations
Many, if not most, ailments of the body cause pain.
Furthermore, the ability to diagnose different dis-
eases depends to a great extent on a physician’s
knowledge of the different qualities of pain. For
these reasons, the first part of this chapter is devoted
mainly to pain and to the physiologic bases of some
associated clinical phenomena.
Pain Is a Protective Mechanism. Pain occurs whenever any tissues are being
damaged, and it causes the individual to react to remove the pain stimulus. Even
such simple activities as sitting for a long time on the ischia can cause tissue
destruction because of lack of blood flow to the skin where it is compressed by
the weight of the body. When the skin becomes painful as a result of the
ischemia, the person normally shifts weight subconsciously. But a person who
has lost the pain sense, as after spinal cord injury, fails to feel the pain and, there-
fore, fails to shift. This soon results in total breakdown and desquamation of the
skin at the areas of pressure.
Types of Pain and Their Qualities—Fast Pain
and Slow Pain
Pain has been classified into two major types: fast pain and slow pain. Fast pain
is felt within about 0.1 second after a pain stimulus is applied, whereas slow pain
begins only after 1 second or more and then increases slowly over many seconds
and sometimes even minutes. During the course of this chapter, we shall see
that the conduction pathways for these two types of pain are different and that
each of them has specific qualities.
Fast pain is also described by many alternative names, such as sharp pain,
pricking pain, acute pain, and electric pain. This type of pain is felt when a needle
is stuck into the skin, when the skin is cut with a knife, or when the skin is acutely
burned. It is also felt when the skin is subjected to electric shock. Fast-sharp
pain is not felt in most deeper tissues of the body.
Slow pain also goes by many names, such as slow burning pain, aching pain,
throbbing pain, nauseous pain, and chronic pain. This type of pain is usually
associated with tissue destruction. It can lead to prolonged, unbearable suffer-
ing. It can occur both in the skin and in almost any deep tissue or organ.
Pain Receptors and Their Stimulation
Pain Receptors Are Free Nerve Endings. The pain receptors in the skin and other
tissues are all free nerve endings. They are widespread in the superficial layers
of the skin as well as in certain internal tissues, such as the periosteum, the arte-
rial walls, the joint surfaces, and the falx and tentorium in the cranial vault. Most
other deep tissues are only sparsely supplied with pain endings; nevertheless,
any widespread tissue damage can summate to cause the slow-chronic-aching
type of pain in most of these areas.
598
Chapter 48
Somatic Sensations: II. Pain, Headache, and Thermal Sensations
599
Three Types of Stimuli Excite Pain Receptors—Mechanical,
Thermal, and Chemical. Pain can be elicited by multiple
types of stimuli. They are classified as mechanical,
thermal, and chemical pain stimuli. In general, fast pain
is elicited by the mechanical and thermal types of
stimuli, whereas slow pain can be elicited by all three
types.
Some of the chemicals that excite the chemical type
of pain are bradykinin, serotonin, histamine, potassium
ions, acids, acetylcholine, and proteolytic enzymes. In
addition, prostaglandins and substance P enhance the
sensitivity of pain endings but do not directly excite
them. The chemical substances are especially impor-
tant in stimulating the slow, suffering type of pain that
occurs after tissue injury.
Nonadapting Nature of Pain Receptors. In contrast to most
other sensory receptors of the body, pain receptors
adapt very little and sometimes not at all. In fact,
43
44
45
46
47
under some conditions, excitation of pain fibers
Temperature (∞C)
becomes progressively greater, especially so for
slow-aching-nauseous pain, as the pain stimulus
continues. This increase in sensitivity of the pain recep-
tors is called hyperalgesia. One can readily understand
Figure 48-1
the importance of this failure of pain receptors to
adapt, because it allows the pain to keep the person
Distribution curve obtained from a large number of persons
apprised of a tissue-damaging stimulus as long as it
showing the minimal skin temperature that will cause pain. (Mod-
ified from Hardy DJ: Nature of pain. J Clin Epidemiol 4:22, 1956.)
persists.
Rate of Tissue Damage as a Stimulus
for Pain
Tissue Ischemia as a Cause of Pain. When blood flow to a
tissue is blocked, the tissue often becomes very painful
The average person begins to perceive pain when the
within a few minutes. The greater the rate of metabo-
skin is heated above 45°C, as shown in Figure 48-1.
lism of the tissue, the more rapidly the pain appears.
This is also the temperature at which the tissues begin
For instance, if a blood pressure cuff is placed around
to be damaged by heat; indeed, the tissues are even-
the upper arm and inflated until the arterial blood flow
tually destroyed if the temperature remains above this
ceases, exercise of the forearm muscles sometimes
level indefinitely. Therefore, it is immediately apparent
can cause muscle pain within 15 to 20 seconds. In the
that pain resulting from heat is closely correlated
absence of muscle exercise, the pain may not appear
with the rate at which damage to the tissues is occur-
for 3 to 4 minutes even though the muscle blood flow
ring and not with the total damage that has already
remains zero.
occurred.
One of the suggested causes of pain during ischemia
The intensity of pain is also closely correlated with
is accumulation of large amounts of lactic acid in the
the rate of tissue damage from causes other than heat,
tissues, formed as a consequence of anaerobic metab-
such as bacterial infection, tissue ischemia, tissue con-
olism (metabolism without oxygen). It is also proba-
tusion, and so forth.
ble that other chemical agents, such as bradykinin and
proteolytic enzymes, are formed in the tissues because
Special Importance of Chemical Pain Stimuli During Tissue
of cell damage and that these, in addition to lactic acid,
Damage. Extracts from damaged tissue cause intense
stimulate the pain nerve endings.
pain when injected beneath the normal skin. Most of
the chemicals listed earlier that excite the chemical
Muscle Spasm as a Cause of Pain. Muscle spasm is also
pain receptors can be found in these extracts. One
a common cause of pain, and it is the basis of many
chemical that seems to be more painful than others is
clinical pain syndromes. This pain probably results
bradykinin. Many researchers have suggested that
partially from the direct effect of muscle spasm in
bradykinin might be the agent most responsible for
stimulating mechanosensitive pain receptors, but it
causing pain following tissue damage. Also, the inten-
might also result from the indirect effect of muscle
sity of the pain felt correlates with the local increase
spasm to compress the blood vessels and cause
in potassium ion concentration or the increase in pro-
ischemia. Also, the spasm increases the rate of metab-
teolytic enzymes that directly attack the nerve endings
olism in the muscle tissue, thus making the relative
and excite pain by making the nerve membranes more
ischemia even greater, creating ideal conditions for the
permeable to ions.
release of chemical pain-inducing substances.
600
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Dual Pathways for
C Ad
Transmission of Pain
Spinal
Fast-sharp
Signals into the Central
nerve
pain fibers
Nervous System
Tract of
I
II
Lissauer
III
Even though all pain receptors are free nerve endings,
IV
V
these endings use two separate pathways for transmit-
VI
ting pain signals into the central nervous system. The
two pathways mainly correspond to the two types of
VII
pain—a fast-sharp pain pathway and a slow-chronic
IX
VIII
pain pathway.
Lamina
marginalis
Peripheral Pain Fibers—“Fast” and “Slow” Fibers. The fast-
Substantia
Slow-chronic
sharp pain signals are elicited by either mechanical
gelatinosa
pain fibers
Anterolateral
or thermal pain stimuli; they are transmitted in the
pathway
peripheral nerves to the spinal cord by small type Ad
fibers at velocities between 6 and 30 m/sec. Conversely,
Figure 48-2
the slow-chronic type of pain is elicited mostly by
Transmission of both “fast-sharp” and “slow-chronic” pain signals
chemical types of pain stimuli but sometimes by
into and through the spinal cord on their way to the brain.
persisting mechanical or thermal stimuli. This slow-
chronic pain is transmitted to the spinal cord by type
C fibers at velocities between 0.5 and 2 m/sec.
Because of this double system of pain innervation,
To: Somatosensory areas
a sudden painful stimulus often gives a “double” pain
sensation: a fast-sharp pain that is transmitted to the
Thalamus
brain by the Ad fiber pathway, followed a second or so
later by a slow pain that is transmitted by the C fiber
Intralaminar
pathway. The sharp pain apprises the person rapidly of
nuclei
a damaging influence and, therefore, plays an impor-
Ventrobasal
complex and
tant role in making the person react immediately to
posterior
remove himself or herself from the stimulus. The slow
nuclear
pain tends to become greater over time. This sensation
group
"Slow" Pain
eventually produces the intolerable suffering of long-
Fibers
continued pain and makes the person keep trying to
"Fast" Pain
Fibers
relieve the cause of the pain.
Reticular
On entering the spinal cord from the dorsal spinal
formation
roots, the pain fibers terminate on relay neurons in the
dorsal horns. Here again, there are two systems for
processing the pain signals on their way to the brain,
as shown in Figures 48-2 and 48-3.
Pain tracts
Dual Pain Pathways in the
Cord and Brain Stem—The
Neospinothalamic Tract and the
Paleospinothalamic Tract
Figure 48-3
On entering the spinal cord, the pain signals take two
Transmission of pain signals into the brain stem, thalamus, and
pathways to the brain, through (1) the neospinothala-
cerebral cortex by way of the fast pricking pain pathway and the
mic tract and (2) the paleospinothalamic tract.
slow burning pain pathway.
Neospinothalamic Tract for Fast Pain. The fast type Ad
pain fibers transmit mainly mechanical and acute
thermal pain. They terminate mainly in lamina I
Termination of the Neospinothalamic Tract in the Brain
(lamina marginalis) of the dorsal horns, as shown in
Stem and Thalamus. A few fibers of the neospinothal-
Figure 48-2, and there excite second-order neurons of
amic tract terminate in the reticular areas of the brain
the neospinothalamic tract. These give rise to long
stem, but most pass all the way to the thalamus without
fibers that cross immediately to the opposite side of
interruption, terminating in the ventrobasal complex
the cord through the anterior commissure and then
along with the dorsal column-medial lemniscal tract
turn upward, passing to the brain in the anterolateral
for tactile sensations, as was discussed in Chapter 47.
columns.
A few fibers also terminate in the posterior nuclear
Chapter 48
Somatic Sensations: II. Pain, Headache, and Thermal Sensations
601
group of the thalamus. From these thalamic areas, the
Projection of the Paleospinothalamic Pathway (Slow-
signals are transmitted to other basal areas of the brain
Chronic Pain Signals) into the Brain Stem and Thala-
as well as to the somatosensory cortex.
mus. The slow-chronic paleospinothalamic pathway
terminates widely in the brain stem, in the large
shaded area shown in Figure 48-3. Only one tenth to
Capability of the Nervous System to Localize Fast Pain
one fourth of the fibers pass all the way to the thala-
in the Body. The fast-sharp type of pain can be local-
mus. Instead, most terminate in one of three areas: (1)
ized much more exactly in the different parts of the
the reticular nuclei of the medulla, pons, and mesen-
body than can slow-chronic pain. However, when only
cephalon; (2) the tectal area of the mesencephalon
pain receptors are stimulated, without the simultane-
deep to the superior and inferior colliculi; or (3) the
ous stimulation of tactile receptors, even fast pain may
periaqueductal gray region surrounding the aqueduct
be poorly localized, often only within 10 centimeters
of Sylvius. These lower regions of the brain appear to
or so of the stimulated area. Yet when tactile receptors
be important for feeling the suffering types of pain,
that excite the dorsal column-medial lemniscal system
because animals whose brains have been sectioned
are simultaneously stimulated, the localization can be
above the mesencephalon to block pain signals from
nearly exact.
reaching the cerebrum still evince undeniable evi-
dence of suffering when any part of the body is trau-
Glutamate, the Probable Neurotransmitter of the Type
matized. From the brain stem pain areas, multiple
Ad Fast Pain Fibers. It is believed that glutamate is the
short-fiber neurons relay the pain signals upward into
neurotransmitter substance secreted in the spinal cord
the intralaminar and ventrolateral nuclei of the thala-
at the type Ad pain nerve fiber endings. This is one of
mus and into certain portions of the hypothalamus and
the most widely used excitatory transmitters in the
other basal regions of the brain.
central nervous system, usually having a duration of
action lasting for only a few milliseconds.
Very Poor Capability of the Nervous System to Local-
ize Precisely the Source of Pain Transmitted in the
Paleospinothalamic Pathway for Transmitting Slow-Chronic
Slow-Chronic Pathway. Localization of pain transmit-
Pain. The paleospinothalamic pathway is a much older
ted by way of the paleospinothalamic pathway is poor.
system and transmits pain mainly from the peripheral
For instance, slow-chronic pain can usually be local-
slow-chronic type C pain fibers, although it does trans-
ized only to a major part of the body, such as to one
mit some signals from type Ad fibers as well. In this
arm or leg but not to a specific point on the arm or leg.
pathway, the peripheral fibers terminate in the spinal
This is in keeping with the multisynaptic, diffuse con-
cord almost entirely in laminae II and III of the dorsal
nectivity of this pathway. It explains why patients often
horns, which together are called the substantia gelati-
have serious difficulty in localizing the source of some
nosa, as shown by the lateral most dorsal root type C
chronic types of pain.
fiber in Figure 48-2. Most of the signals then pass
through one or more additional short fiber neurons
Function of the Reticular Formation, Thalamus, and Cerebral
within the dorsal horns themselves before entering
Cortex in the Appreciation of Pain. Complete removal of
mainly lamina V, also in the dorsal horn. Here the last
the somatic sensory areas of the cerebral cortex does
neurons in the series give rise to long axons that mostly
not destroy an animal’s ability to perceive pain. There-
join the fibers from the fast pain pathway, passing first
fore, it is likely that pain impulses entering the brain
through the anterior commissure to the opposite side
stem reticular formation, the thalamus, and other
of the cord, then upward to the brain in the anterolat-
lower brain centers cause conscious perception of
eral pathway.
pain. This does not mean that the cerebral cortex has
nothing to do with normal pain appreciation; electri-
Substance P, the Probable Slow-Chronic Neurotrans-
cal stimulation of cortical somatosensory areas does
mitter of Type C Nerve Endings. Research experi-
cause a human being to perceive mild pain from about
ments suggest that type C pain fiber terminals entering
3 per cent of the points stimulated. However, it is
the spinal cord secrete both glutamate transmitter and
believed that the cortex plays an especially important
substance P transmitter. The glutamate transmitter
role in interpreting pain quality, even though pain per-
acts instantaneously and lasts for only a few millisec-
ception might be principally the function of lower
onds. Substance P is released much more slowly, build-
centers.
ing up in concentration over a period of seconds or
even minutes. In fact, it has been suggested that the
Special Capability of Pain Signals to Arouse Overall Brain
“double” pain sensation one feels after a pinprick
Excitability. Electrical stimulation in the reticular areas
might result partly from the fact that the glutamate
of the brain stem and in the intralaminar nuclei of the
transmitter gives a faster pain sensation, whereas the
thalamus, the areas where the slow-suffering type of
substance P transmitter gives a more lagging sensa-
pain terminates, has a strong arousal effect on nervous
tion. Regardless of the yet unknown details, it seems
activity throughout the entire brain. In fact, these two
clear that glutamate is the neurotransmitter most
areas constitute part of the brain’s principal “arousal
involved in transmitting fast pain into the central
system,” which is discussed in Chapter 59. This explains
nervous system, and substance P is concerned with
why it is almost impossible for a person to sleep when
slow-chronic pain.
he or she is in severe pain.
602
Unit IX The Nervous System: A. General Principles and Sensory Physiology
Surgical Interruption of Pain Pathways. When a person has
severe and intractable pain (sometimes resulting from
Third
rapidly spreading cancer), it is necessary to relieve the
ventricle
pain. To do this, the pain nervous pathways can be cut
at any one of several points. If the pain is in the lower
Periventricular
Aqueduct
nuclei
part of the body, a cordotomy in the thoracic region of
the spinal cord often relieves the pain for a few weeks
Periaqueductal gray
Mesencephalon
to a few months. To do this, the spinal cord on the side
opposite to the pain is partially cut in its anterolateral
quadrant to interrupt the anterolateral sensory pathway.
Enkephalin neurons
A cordotomy, however, is not always successful in
relieving pain, for two reasons. First, many pain fibers
Fourth
Pons
from the upper part of the body do not cross to the
ventricle
Raphe magnus
opposite side of the spinal cord until they have reached
nucleus
the brain, so that the cordotomy does not transect these
fibers. Second, pain frequently returns several months
Medulla
later, partly as a result of sensitization of other pathways
that normally are too weak to be effectual (e.g., sparse
pathways in the dorsolateral cord). Another experi-
Serotonergic
mental operative procedure to relieve pain has been to
neurons
cauterize specific pain areas in the intralaminar nuclei
in the thalamus, which often relieves suffering types of
pain while leaving intact one’s appreciation of “acute”
pain, an important protective mechanism.
Pain fibers
Enkephalin
neurons
Presynaptic
Pain Suppression
pain inhibition
(“Analgesia”) System in the
Anterolateral
Brain and Spinal Cord
somatosensory tract
The degree to which a person reacts to pain varies
Figure 48-4
tremendously. This results partly from a capability of
the brain itself to suppress input of pain signals to the
Analgesia system of the brain and spinal cord, showing (1) inhi-
nervous system by activating a pain control system,
bition of incoming pain signals at the cord level and (2) presence
of enkephalin-secreting neurons that suppress pain signals in
called an analgesia system.
both the cord and the brain stem.
The analgesia system is shown in Figure 48-4. It
consists of three major components: (1) The periaque-
ductal gray and periventricular areas of the mesen-
cephalon and upper pons surround the aqueduct of
periventricular nuclei and from the periaqueductal
Sylvius and portions of the third and fourth ventricles.
gray area secrete enkephalin at their endings. Thus, as
Neurons from these areas send signals to (2) the raphe
shown in Figure 48-4, the endings of many fibers in
magnus nucleus, a thin midline nucleus located in
the raphe magnus nucleus release enkephalin when
the lower pons and upper medulla, and the nucleus
stimulated.
reticularis paragigantocellularis, located laterally in
Fibers originating in this area send signals to the
the medulla. From these nuclei, second-order signals
dorsal horns of the spinal cord to secrete serotonin at
are transmitted down the dorsolateral columns in the
their endings. The serotonin causes local cord neurons
spinal cord to (3) a pain inhibitory complex located in
to secrete enkephalin as well. The enkephalin is
the dorsal horns of the spinal cord. At this point, the
believed to cause both presynaptic and postsynaptic
analgesia signals can block the pain before it is relayed
inhibition of incoming type C and type Ad pain fibers
to the brain.
where they synapse in the dorsal horns.
Electrical stimulation either in the periaqueductal
Thus, the analgesia system can block pain signals at
gray area or in the raphe magnus nucleus can suppress
the initial entry point to the spinal cord. In fact, it can
many strong pain signals entering by way of the dorsal
also block many local cord reflexes that result from
spinal roots. Also, stimulation of areas at still higher
pain signals, especially withdrawal reflexes described
levels of the brain that excite the periaqueductal gray
in Chapter 54.
area can also suppress pain. Some of these areas are
(1) the periventricular nuclei in the hypothalamus, lying
adjacent to the third ventricle, and (2) to a lesser
Brain’s Opiate System—Endorphins
extent, the medial forebrain bundle, also in the hypo-
and Enkephalins
thalamus.
Several transmitter substances are involved in the
More than 35 years ago it was discovered that injec-
analgesia system; especially involved are enkephalin
tion of minute quantities of morphine either into the
and serotonin. Many nerve fibers derived from the
periventricular nucleus around the third ventricle or
Chapter 48
Somatic Sensations: II. Pain, Headache, and Thermal Sensations
603
into the periaqueductal gray area of the brain stem
been reported in some instances. Also, pain relief has
causes an extreme degree of analgesia. In subsequent
been reported to last for as long as 24 hours after only
studies, it has been found that morphine-like agents,
a few minutes of stimulation.
mainly the opiates, also act at many other points in the
analgesia system, including the dorsal horns of the
Referred Pain
spinal cord. Because most drugs that alter excitability
of neurons do so by acting on synaptic receptors, it was
Often a person feels pain in a part of the body that is
assumed that the “morphine receptors” of the analge-
fairly remote from the tissue causing the pain. This is
sia system must be receptors for some morphine-like
called referred pain. For instance, pain in one of the
neurotransmitter that is naturally secreted in the
visceral organs often is referred to an area on the body
brain. Therefore, an extensive search was undertaken
surface. Knowledge of the different types of referred
for the natural opiate of the brain. About a dozen such
pain is important in clinical diagnosis because in many
opiate-like substances have now been found at differ-
visceral ailments the only clinical sign is referred pain.
ent points of the nervous system; all are breakdown
products of three large protein molecules: pro-
Mechanism of Referred Pain. Figure 48-5 shows the prob-
opiomelanocortin, proenkephalin, and prodynorphin.
able mechanism by which most pain is referred. In the
Among the more important of these opiate-like
figure, branches of visceral pain fibers are shown to
substances are b-endorphin, met-enkephalin, leu-
synapse in the spinal cord on the same second-order
enkephalin, and dynorphin.
neurons (1 and 2) that receive pain signals from the
The two enkephalins are found in the brain stem
skin. When the visceral pain fibers are stimulated, pain
and spinal cord, in the portions of the analgesia system
signals from the viscera are conducted through at least
described earlier, and b-endorphin is present in both
some of the same neurons that conduct pain signals
the hypothalamus and the pituitary gland. Dynorphin
from the skin, and the person has the feeling that the
is found mainly in the same areas as the enkephalins,
sensations originate in the skin itself.
but in much lower quantities.
Thus, although the fine details of the brain’s opiate
system are not understood, activation of the analgesia
Visceral Pain
system by nervous signals entering the periaqueductal
gray and periventricular areas, or inactivation of pain
In clinical diagnosis, pain from the different viscera of
pathways by morphine-like drugs, can almost totally
the abdomen and chest is one of the few criteria that
suppress many pain signals entering through the
can be used for diagnosing visceral inflammation, vis-
ceral infectious disease, and other visceral ailments.
peripheral nerves.
Often, the viscera have sensory receptors for no other
modalities of sensation besides pain. Also, visceral pain
differs from surface pain in several important aspects.
Inhibition of Pain Transmission
One of the most important differences between
by Simultaneous Tactile
surface pain and visceral pain is that highly localized
Sensory Signals
types of damage to the viscera seldom cause severe
pain. For instance, a surgeon can cut the gut entirely in
Another important event in the saga of pain control was
two in a patient who is awake without causing signifi-
the discovery that stimulation of large type Ab sensory
cant pain. Conversely, any stimulus that causes diffuse
fibers from peripheral tactile receptors can depress
stimulation of pain nerve endings throughout a viscus
transmission of pain signals from the same body area.
causes pain that can be severe. For instance, ischemia
This presumably results from local lateral inhibition in
the spinal cord. It explains why such simple maneuvers
as rubbing the skin near painful areas is often effective
in relieving pain. And it probably also explains why lin-
iments are often useful for pain relief.
This mechanism and the simultaneous psychogenic
excitation of the central analgesia system are probably
also the basis of pain relief by acupuncture.
1
2
Treatment of Pain by Electrical
Stimulation
Several clinical procedures have been developed for
suppressing pain by electrical stimulation. Stimulating
electrodes are placed on selected areas of the skin or,
on occasion, implanted over the spinal cord, supposedly
Visceral
Skin nerve
to stimulate the dorsal sensory columns.
nerve fibers
fibers
In some patients, electrodes have been placed stereo-
taxically in appropriate intralaminar nuclei of the thal-
amus or in the periventricular or periaqueductal area of
Figure 48-5
the diencephalon. The patient can then personally
control the degree of stimulation. Dramatic relief has
Mechanism of referred pain and referred hyperalgesia.
604
Unit IX The Nervous System: A. General Principles and Sensory Physiology
caused by occluding the blood supply to a large area of
“Parietal Pain” Caused by
gut stimulates many diffuse pain fibers at the same time
Visceral Disease
and can result in extreme pain.
When a disease affects a viscus, the disease process
often spreads to the parietal peritoneum, pleura, or
Causes of True Visceral Pain
pericardium. These parietal surfaces, like the skin, are
supplied with extensive pain innervation from the
Any stimulus that excites pain nerve endings in diffuse
peripheral spinal nerves. Therefore, pain from the pari-
areas of the viscera can cause visceral pain. Such stimuli
etal wall overlying a viscus is frequently sharp. An
include ischemia of visceral tissue, chemical damage to
example can emphasize the difference between this
the surfaces of the viscera, spasm of the smooth muscle
pain and true visceral pain: a knife incision through the
of a hollow viscus, excess distention of a hollow viscus,
parietal peritoneum is very painful, whereas a similar
and stretching of the connective tissue surrounding or
cut through the visceral peritoneum or through a gut
within the viscus. Essentially all visceral pain that orig-
wall is not very painful, if painful at all.
inates in the thoracic and abdominal cavities is trans-
mitted through small type C pain fibers and, therefore,
can transmit only the chronic-aching-suffering type of
Localization of Visceral Pain—
pain.
“Visceral” and the “Parietal” Pain
Transmission Pathways
Ischemia. Ischemia causes visceral pain in the same way
that it does in other tissues, presumably because of the
Pain from the different viscera is frequently difficult to
formation of acidic metabolic end products or tissue-
localize, for a number of reasons. First, the patient’s
degenerative products such as bradykinin, proteolytic
brain does not know from firsthand experience that the
enzymes, or others that stimulate pain nerve endings.
different internal organs exist; therefore, any pain that
originates internally can be localized only generally.
Second, sensations from the abdomen and thorax are
Chemical Stimuli. On occasion, damaging substances leak
transmitted through two pathways to the central
from the gastrointestinal tract into the peritoneal cavity.
nervous system—the true visceral pathway and the pari-
For instance, proteolytic acidic gastric juice often leaks
etal pathway. True visceral pain is transmitted via pain
through a ruptured gastric or duodenal ulcer. This juice
sensory fibers within the autonomic nerve bundles, and
causes widespread digestion of the visceral peritoneum,
the sensations are referred to surface areas of the body
thus stimulating broad areas of pain fibers. The pain is
often far from the painful organ. Conversely, parietal
usually excruciatingly severe.
sensations are conducted directly into local spinal
nerves from the parietal peritoneum, pleura, or peri-
Spasm of a Hollow Viscus. Spasm of a portion of the gut,
cardium, and these sensations are usually localized
the gallbladder, a bile duct, a ureter, or any other hollow
directly over the painful area.
viscus can cause pain, possibly by mechanical stimula-
tion of the pain nerve endings. Or the spasm might cause
Localization of Referred Pain Transmitted via Visceral Pathways.
diminished blood flow to the muscle, combined with the
When visceral pain is referred to the surface of the body,
muscle’s increased metabolic need for nutrients, thus
the person generally localizes it in the dermatomal
causing severe pain.
segment from which the visceral organ originated in the
Often pain from a spastic viscus occurs in the form of
embryo, not necessarily where the visceral organ now
cramps, with the pain increasing to a high degree of
lies. For instance, the heart originated in the neck and
severity and then subsiding. This process continues
upper thorax, so that the heart’s visceral pain fibers pass
intermittently, once every few minutes. The intermittent
upward along the sympathetic sensory nerves and enter
cycles result from periods of contraction of smooth
the spinal cord between segments C-3 and T-5. There-
muscle. For instance, each time a peristaltic wave travels
fore, as shown in Figure 48-6, pain from the heart is
along an overly excitable spastic gut, a cramp occurs.
referred to the side of the neck, over the shoulder, over
The cramping type of pain frequently occurs in appen-
the pectoral muscles, down the arm, and into the sub-
dicitis, gastroenteritis, constipation, menstruation, par-
sternal area of the upper chest. These are the areas of
turition, gallbladder disease, or ureteral obstruction.
the body surface that send their own somatosensory
nerve fibers into the C-3 to T-5 cord segments. Most fre-
quently, the pain is on the left side rather than on the
Overdistention of a Hollow Viscus. Extreme overfilling of a
right because the left side of the heart is much more fre-
hollow viscus also can result in pain, presumably
quently involved in coronary disease than the right.
because of overstretch of the tissues themselves.
The stomach originated approximately from the
Overdistention can also collapse the blood vessels that
seventh to ninth thoracic segments of the embryo.
encircle the viscus or that pass into its wall, thus perhaps
Therefore, stomach pain is referred to the anterior epi-
promoting ischemic pain.
gastrium above the umbilicus, which is the surface area
of the body subserved by the seventh through ninth tho-
Insensitive Viscera. A few visceral areas are almost com-
racic segments. Figure 48-6 shows several other surface
pletely insensitive to pain of any type. These include the
areas to which visceral pain is referred from other
parenchyma of the liver and the alveoli of the lungs. Yet
organs, representing in general the areas in the embryo
the liver capsule is extremely sensitive to both direct
from which the respective organs originated.
trauma and stretch, and the bile ducts are also sensitive
to pain. In the lungs, even though the alveoli are insen-
Parietal Pathway for Transmission of Abdominal and Thoracic
sitive, both the bronchi and the parietal pleura are very
Pain. Pain from the viscera is frequently localized to two
sensitive to pain.
surface areas of the body at the same time because of
Chapter 48
Somatic Sensations: II. Pain, Headache, and Thermal Sensations
605
Heart
T-10
Esophagus
L-1
Stomach
Liver and
gallbladder
Pylorus
Umbilicus
Appendix and
small intestine
Right kidney
Visceral pain
Left kidney
Colon
Parietal pain
Ureter
Figure 48-6
Figure 48-7
Surface areas of referred pain from different visceral organs.
Visceral and parietal transmission of
pain
signals from the
appendix.
Herpes Zoster (Shingles)
the dual transmission of pain through the referred vis-
Occasionally herpesvirus infects a dorsal root ganglion.
ceral pathway and the direct parietal pathway. Thus,
This causes severe pain in the dermatomal segment sub-
Figure 48-7 shows dual transmission from an inflamed
served by the ganglion, thus eliciting a segmental type
appendix. Pain impulses pass first from the appendix
of pain that circles halfway around the body. The disease
through visceral pain fibers located within sympathetic
is called herpes zoster, or “shingles,” because of a skin
nerve bundles, and then into the spinal cord at about
eruption that often ensues.
T-10 or T-11; this pain is referred to an area around the
The cause of the pain is presumably infection of the
umbilicus and is of the aching, cramping type. Pain
pain neuronal cells in the dorsal root ganglion by the
impulses also often originate in the parietal peritoneum
virus. In addition to causing pain, the virus is carried by
where the inflamed appendix touches or is adherent to
neuronal cytoplasmic flow outward through the neu-
the abdominal wall. These cause pain of the sharp type
ronal peripheral axons to their cutaneous origins. Here
directly over the irritated peritoneum in the right lower
the virus causes a rash that vesiculates within a few days
quadrant of the abdomen.
and then crusts over within another few days, all of this
occurring within the dermatomal area served by the
infected dorsal root.
Some Clinical Abnormalities
of Pain and Other
Somatic Sensations
Tic Douloureux
Lancinating pain occasionally occurs in some people
Hyperalgesia
over one side of the face in the sensory distribution area
A pain nervous pathway sometimes becomes exces-
(or part of the area) of the fifth or ninth nerves; this phe-
sively excitable; this gives rise to hyperalgesia, which
nomenon is called tic douloureux (or trigeminal neural-
means hypersensitivity to pain. Possible causes of hyper-
gia or glossopharyngeal neuralgia). The pain feels like
algesia are (1) excessive sensitivity of the pain receptors
sudden electrical shocks, and it may appear for only a
themselves, which is called primary hyperalgesia, and
few seconds at a time or may be almost continuous.
(2) facilitation of sensory transmission, which is called
Often it is set off by exceedingly sensitive trigger areas
secondary hyperalgesia.
on the surface of the face, in the mouth, or inside the
An example of primary hyperalgesia is the extreme
throat—almost always by a mechanoreceptive stimulus
sensitivity of sunburned skin, which results from
rather than a pain stimulus. For instance, when the
sensitization of the skin pain endings by local tissue
patient swallows a bolus of food, as the food touches a
products from the burn—perhaps histamine, perhaps
tonsil, it might set off a severe lancinating pain in the
prostaglandins, perhaps others. Secondary hyperalgesia
mandibular portion of the fifth nerve.
frequently results from lesions in the spinal cord or the
The pain of tic douloureux can usually be blocked by
thalamus. Several of these lesions are discussed in sub-
surgically cutting the peripheral nerve from the hyper-
sequent sections.
sensitive area. The sensory portion of the fifth nerve is
606
Unit IX The Nervous System: A. General Principles and Sensory Physiology
often sectioned immediately inside the cranium, where
result from pain stimuli arising inside the cranium, but
the motor and sensory roots of the fifth nerve separate
others result from pain arising outside the cranium, such
from each other, so that the motor portions, which are
as from the nasal sinuses.
needed for many jaw movements, can be spared while
the sensory elements are destroyed. This operation
leaves the side of the face anesthetic, which in itself may
Headache of Intracranial Origin
be annoying. Furthermore, sometimes the operation is
Pain-Sensitive Areas in Cranial Vault. The brain tissues
unsuccessful, indicating that the lesion that causes the
themselves are almost totally insensitive to pain. Even
pain might be in the sensory nucleus in the brain stem
cutting or electrically stimulating the sensory areas
and not in the peripheral nerves.
of the cerebral cortex only occasionally causes pain;
instead, it causes prickly types of paresthesias on the
Brown-Séquard Syndrome
area of the body represented by the portion of the
sensory cortex stimulated. Therefore, it is likely that
If the spinal cord is transected entirely, all sensations
much or most of the pain of headache is not caused by
and motor functions distal to the segment of transection
damage within the brain itself.
are blocked, but if the spinal cord is transected on only
Conversely, tugging on the venous sinuses around the
one side, the Brown-Séquard syndrome occurs. The
brain, damaging the tentorium, or stretching the dura at
effects of such transection can be predicted from a
the base of the brain can cause intense pain that is rec-
knowledge of the cord fiber tracts shown in Figure 48-8.
ognized as headache. Also, almost any type of trauma-
All motor functions are blocked on the side of the tran-
tizing, crushing, or stretching stimulus to the blood
section in all segments below the level of the transec-
vessels of the meninges can cause headache. An espe-
tion. Yet only some of the modalities of sensation are
cially sensitive structure is the middle meningeal artery,
lost on the transected side, and others are lost on the
and neurosurgeons are careful to anesthetize this artery
opposite side. The sensations of pain, heat, and cold—
specifically when performing brain operations under
sensations served by the spinothalamic pathway—are
local anesthesia.
lost on the opposite side of the body in all dermatomes
two to six segments below the level of the transection.
Areas of the Head to Which Intracranial Headache Is Referred.
By contrast, the sensations that are transmitted only in
Stimulation of pain receptors in the cerebral vault
the dorsal and dorsolateral columns—kinesthetic and
above the tentorium, including the upper surface of the
position sensations, vibration sensation, discrete local-
tentorium itself, initiates pain impulses in the cerebral
ization, and two-point discrimination—are lost on the
portion of the fifth nerve and, therefore, causes referred
side of the transection in all dermatomes below the level
headache to the front half of the head in the surface
of the transection. Discrete “light touch” is impaired
areas supplied by this somatosensory portion of the fifth
on the side of the transection because the principal
cranial nerve, as shown in Figure 48-9.
pathway for the transmission of light touch, the dorsal
Conversely, pain impulses from beneath the
column, is transected. That is, the fibers in this column
tentorium enter the central nervous system mainly
do not cross to the opposite side until they reach the
through the glossopharyngeal, vagal, and second cervi-
medulla of the brain. “Crude touch,” which is poorly
cal nerves, which also supply the scalp above, behind,
localized, still persists because of partial transmission in
and slightly below the ear. Subtentorial pain stimuli
the opposite spinothalamic tract.
cause “occipital headache” referred to the posterior
part of the head.
Headache
Headaches are a type of pain referred to the surface of
the head from deep head structures. Some headaches
Cerebral vault
Brain stem and
headaches
cerebellar vault
headaches
Fasciculus gracilis
Fasciculus cuneatus
Lateral
Dorsal
corticospinal
spinocerebellar
Nasal sinus
Rubrospinal
and eye
Lateral
headaches
spinothalamic
Olivospinal
Ventral
spinocerebellar
Tectospinal
Spinotectal
Ventral
corticospinal
Vestibulospinal
Ventral spinothalamic
Descending
Ascending
Tracts
Tracts
Figure 48-8
Figure 48-9
Cross section of the spinal cord, showing principal ascending
tracts on the right and principal descending tracts on the left.
Areas of headache resulting from different causes.
Chapter 48
Somatic Sensations: II. Pain, Headache, and Thermal Sensations
607
Types of Intracranial Headache
absorbed toxic products or from changes in the circula-
Headache of Meningitis. One of the most severe
tory system resulting from loss of fluid into the gut.
headaches of all is that resulting from meningitis, which
causes inflammation of all the meninges, including the
sensitive areas of the dura and the sensitive areas
Extracranial Types of Headache
around the venous sinuses. Such intense damage can
Headache Resulting from Muscle Spasm. Emotional tension
cause extreme headache pain referred over the entire
often causes many of the muscles of the head, especially
head.
those muscles attached to the scalp and the neck
Headache Caused by Low Cerebrospinal Fluid Pressure.
muscles attached to the occiput, to become spastic, and
Removing as little as 20 milliliters of fluid from the
it is postulated that this is one of the common causes of
spinal canal, particularly if the person remains in an
headache. The pain of the spastic head muscles suppos-
upright position, often causes intense intracranial
edly is referred to the overlying areas of the head and
headache. Removing this quantity of fluid removes part
gives one the same type of headache as intracranial
of the flotation for the brain that is normally provided
lesions do.
by the cerebrospinal fluid. The weight of the brain
stretches and otherwise distorts the various dural
Headache Caused by Irritation of Nasal and Accessory Nasal
surfaces and thereby elicits the pain that causes the
Structures. The mucous membranes of the nose and
headache.
nasal sinuses are sensitive to pain, but not intensely so.
Nevertheless, infection or other irritative processes in
Migraine Headache. Migraine headache is a special type
widespread areas of the nasal structures often summate
of headache that is thought to result from abnormal vas-
and cause headache that is referred behind the eyes or,
cular phenomena, although the exact mechanism is
in the case of frontal sinus infection, to the frontal sur-
unknown. Migraine headaches often begin with various
faces of the forehead and scalp, as shown in Figure 48-9.
prodromal sensations, such as nausea, loss of vision in
Also, pain from the lower sinuses, such as from the max-
part of the field of vision, visual aura, and other types
illary sinuses, can be felt in the face.
of sensory hallucinations. Ordinarily, the prodromal
symptoms begin 30 minutes to
1 hour before the
Headache Caused by Eye Disorders. Difficulty in focusing
beginning of the headache. Any theory that explains
one’s eyes clearly may cause excessive contraction of
migraine headache must also explain the prodromal
the eye ciliary muscles in an attempt to gain clear vision.
symptoms.
Even though these muscles are extremely small, it is
One of the theories of the cause of migraine
believed that tonic contraction of them can cause retro-
headaches is that prolonged emotion or tension causes
orbital headache. Also, excessive attempts to focus
reflex vasospasm of some of the arteries of the head,
the eyes can result in reflex spasm in various facial
including arteries that supply the brain. The vasospasm
and extraocular muscles, which is a possible cause of
theoretically produces ischemia of portions of the brain,
headache.
and this is responsible for the prodromal symptoms.
A second type of headache that originates in the eyes
Then, as a result of the intense ischemia, something
occurs when the eyes are exposed to excessive irradia-
happens to the vascular walls, perhaps exhaustion of
tion by light rays, especially ultraviolet light. Looking at
smooth muscle contraction, to allow the blood vessels
the sun or the arc of an arc-welder for even a few
to become flaccid and incapable of maintaining vascu-
seconds may result in headache that lasts from 24 to 48
lar tone for 24 to 48 hours. The blood pressure in the
hours. The headache sometimes results from “actinic”
vessels causes them to dilate and pulsate intensely, and
irritation of the conjunctivae, and the pain is referred to
it is postulated that the excessive stretching of the walls
the surface of the head or retro-orbitally. However,
of the arteries—including some extracranial arteries,
focusing intense light from an arc or the sun on the
such as the temporal artery—causes the actual pain
retina can also burn the retina, and this could be the
of migraine headaches. Other theories of the cause
cause of the headache.
of migraine headaches include spreading cortical de-
pression, psychological abnormalities, and vasospasm
caused by excess local potassium in the cerebral extra-
Thermal Sensations
cellular fluid.
There may be a genetic predisposition to migraine
headaches, because a positive family history for
Thermal Receptors and
migraine has been reported in 65 to 90 per cent of cases.
Their Excitation
Migraine headaches also occur about twice as fre-
quently in women as in men.
The human being can perceive different gradations of
Alcoholic Headache. As many people have experi-
cold and heat, from freezing cold to cold to cool to
enced, a headache usually follows an alcoholic binge. It
indifferent to warm to hot to burning hot.
is most likely that alcohol, because it is toxic to tissues,
Thermal gradations are discriminated by at least
directly irritates the meninges and causes the intracra-
three types of sensory receptors: cold receptors,
nial pain.
warmth receptors, and pain receptors. The pain recep-
tors are stimulated only by extreme degrees of heat or
Headache Caused by Constipation. Constipation causes
cold and, therefore, are responsible, along with the
headache in many people. Because it has been shown
cold and warmth receptors, for “freezing cold” and
that constipation headache can occur in people whose
pain sensory tracts in the spinal cord have been cut, we
“burning hot” sensations.
know that this headache is not caused by nervous
The cold and warmth receptors are located imme-
impulses from the colon. Therefore, it may result from
diately under the skin at discrete separated spots. In
608
Unit IX The Nervous System: A. General Principles and Sensory Physiology
most areas of the body, there are 3 to 10 times as many
out slightly above
40°C. Above about
30°C, the
cold spots as warmth spots, and the number in differ-
warmth receptors begin to be stimulated, but these
ent areas of the body varies from 15 to 25 cold spots
also fade out at about 49°C. Finally, at around 45°C,
per square centimeter in the lips to 3 to 5 cold spots
the heat-pain fibers begin to be stimulated by heat and,
per square centimeter in the finger to less than 1 cold
paradoxically, some of the cold fibers begin to be stim-
spot per square centimeter in some broad surface
ulated again, possibly because of damage to the cold
areas of the trunk.
endings caused by the excessive heat.
Although the existence of distinctive warmth nerve
One can understand from Figure 48-10 that a
endings is quite certain, based on psychological tests,
person determines the different gradations of thermal
they have not been identified histologically. They are
sensations by the relative degrees of stimulation of the
presumed to be free nerve endings, because warmth
different types of endings. One can also understand
signals are transmitted mainly over type C nerve fibers
why extreme degrees of both cold and heat can be
at transmission velocities of only 0.4 to 2 m/sec.
painful and why both these sensations, when intense
Conversely, a definitive cold receptor has been iden-
enough, may give almost the same quality of sensa-
tified. It is a special, small type Ad myelinated nerve
tion—that is, freezing cold and burning hot sensations
ending that branches a number of times, the tips of
feel almost alike.
which protrude into the bottom surfaces of basal epi-
dermal cells. Signals are transmitted from these recep-
Stimulatory Effects of Rising and Falling Temperature—Adap-
tors via type Ad nerve fibers at velocities of about
tation of Thermal Receptors. When a cold receptor is sud-
20 m/sec. Some cold sensations are believed to be
denly subjected to an abrupt fall in temperature, it
transmitted in type C nerve fibers as well, which sug-
becomes strongly stimulated at first, but this stimula-
gests that some free nerve endings also might function
tion fades rapidly during the first few seconds and pro-
as cold receptors.
gressively more slowly during the next 30 minutes or
more. In other words, the receptor “adapts” to a great
Stimulation of Thermal Receptors—Sensations of Cold, Cool,
extent, but never 100 per cent.
Indifferent, Warm, and Hot. Figure
48-10 shows the
Thus, it is evident that the thermal senses respond
effects of different temperatures on the responses of
markedly to changes in temperature, in addition to
four types of nerve fibers: (1) a pain fiber stimulated
being able to respond to steady states of temperature.
by cold, (2) a cold fiber, (3) a warmth fiber, and (4) a
This means that when the temperature of the skin is
pain fiber stimulated by heat. Note especially that
actively falling, a person feels much colder than when
these fibers respond differently at different levels of
the temperature remains cold at the same level. Con-
temperature. For instance, in the very cold region, only
versely, if the temperature is actively rising, the person
the cold-pain fibers are stimulated (if the skin becomes
feels much warmer than he or she would at the same
even colder, so that it nearly freezes or actually does
temperature if it were constant. The response to
freeze, these fibers cannot be stimulated). As the tem-
changes in temperature explains the extreme degree
perature rises to +10° to 15°C, the cold-pain impulses
of heat one feels on first entering a tub of hot water
cease, but the cold receptors begin to be stimulated,
and the extreme degree of cold felt on going from a
reaching peak stimulation at about 24°C and fading
heated room to the out-of-doors on a cold day.
Freezing
Indiffer-
Burning
Cold
Cool
Warm
Hot
cold
ent
hot
Cold-pain
10
Cold-receptors
Warmth receptors
8
Heat-pain
6
4
2
5
10
15
20
25
30
35
40
45
50
55
60
Figure 48-10
Temperature (∞C)
Discharge frequencies at different skin tempera-
tures of a cold-pain fiber, a cold fiber, a warmth
fiber, and a heat-pain fiber.
Chapter 48
Somatic Sensations: II. Pain, Headache, and Thermal Sensations
609
Mechanism of Stimulation of
being reduces but does not abolish the ability to dis-
Thermal Receptors
tinguish gradations of temperature.
It is believed that the cold and warmth receptors are
stimulated by changes in their metabolic rates, and
that these changes result from the fact that tempera-
References
ture alters the rate of intracellular chemical reactions
more than twofold for each 10°C change. In other
Almeida TF, Roizenblatt S, Tufik S: Afferent pain pathways:
words, thermal detection probably results not from
a neuroanatomical review. Brain Res 1000:40, 2004.
direct physical effects of heat or cold on the nerve
Ballantyne JC, Mao J: Opioid therapy for chronic pain.
N Engl J Med 349:1943, 2003.
endings but from chemical stimulation of the endings
Borsook D, Becerra L: Pain imaging: future applications to
as modified by temperature.
integrative clinical and basic neurobiology. Adv Drug
Deliv Rev 55:967, 2003.
Spatial Summation of Thermal Sensations. Because the
Bromm B: Brain images of pain. News Physiol Sci 16:244,
number of cold or warm endings in any one surface
2001.
area of the body is slight, it is difficult to judge grada-
Bruera E, Kim HN: Cancer pain. JAMA 290:2476, 2003.
tions of temperature when small skin areas are stimu-
Cervero F, Laird JM: Role of ion channels in mechanisms
lated. However, when a large skin area is stimulated
controlling gastrointestinal pain pathways. Curr Opin
all at once, the thermal signals from the entire area
Pharmacol 3:608, 2003.
summate. For instance, rapid changes in temperature
Costigan M, Woolf CJ: No DREAM, no pain: closing the
spinal gate. Cell 108:297, 2002.
as little as 0.01°C can be detected if this change affects
Gebhart GF: Descending modulation of pain. Neurosci
the entire surface of the body simultaneously. Con-
Biobehav Rev 27:729, 2004.
versely, temperature changes 100 times as great often
Haines DE: Fundamental Neuroscience. New York:
will not be detected when the affected skin area is only
Churchill Livingstone, 1997.
1 square centimeter in size.
Haines DE, Lancon JA: Review of Neuroscience. New York:
Churchill Livingstone, 2003.
Jordt SE, McKemy DD, Julius D: Lessons from peppers and
Transmission of Thermal Signals
peppermint: the molecular logic of thermosensation. Curr
in the Nervous System
Opin Neurobiol 13:487, 2003.
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
Science, 4th ed. New York: McGraw-Hill, 2000.
In general, thermal signals are transmitted in pathways
Kors EE, Vanmolkot KR, Haan J, et al: Recent findings in
parallel to those for pain signals. On entering the
headache genetics. Curr Opin Neurol 17:283, 2004.
spinal cord, the signals travel for a few segments
Mendell JR, Sahenk Z: Clinical practice: painful sensory
upward or downward in the tract of Lissauer and then
neuropathy. N Engl J Med 348:1243, 2003.
terminate mainly in laminae I, II, and III of the dorsal
Montell C: Thermosensation: hot findings make TRPNs very
horns—the same as for pain. After a small amount of
cool. Curr Biol 13:R476, 2003.
processing by one or more cord neurons, the signals
Sanchez-del-Rio M, Reuter U: Migraine aura: new informa-
enter long, ascending thermal fibers that cross to the
tion on underlying mechanisms. Curr Opin Neurol 17:289,
opposite anterolateral sensory tract and terminate in
2004.
Schaible HG, Ebersberger A, Von Banchet GS: Mechanisms
both (1) the reticular areas of the brain stem and (2)
of pain in arthritis. Ann N Y Acad Sci 966:343, 2002.
the ventrobasal complex of the thalamus.
Silberstein SD: Migraine. Lancet 363:381, 2004.
A few thermal signals are also relayed to the cere-
Vogt BA: Knocking out the DREAM to study pain. N Engl
bral somatic sensory cortex from the ventrobasal
J Med 347:362, 2002.
complex. Occasionally a neuron in cortical somatic
Watkins LR, Maier SF: Beyond neurons: evidence that
sensory area I has been found by microelectrode
immune and glial cells contribute to pathological pain
studies to be directly responsive to either cold or warm
states. Physiol Rev 82:981, 2002.
stimuli on a specific area of the skin. However, removal
Zubrzycka M, Janecka A: Substance P: transmitter of noci-
of the entire cortical postcentral gyrus in the human
ception (minireview). Endocr Regul 34:195, 2000.
U N I T X
The Nervous
System: B. The
Special Senses
49. The Eye: I. Optics of Vision
50. The Eye: II. Receptor and Neural
Function of the Retina
51. The Eye: III. Central Neurophysiology of Vision
52. The Sense of Hearing
53. The Chemical Senses—Taste and Smell
C H A P T E R
4
9
The Eye: I. Optics of Vision
Physical Principles
of Optics
Before it is possible to understand the optical
system of the eye, the student must first be thor-
oughly familiar with the basic principles of optics,
including the physics of light refraction, focusing,
depth of focus, and so forth. A brief review of these
physical principles is presented; then the optics of the eye is discussed.
Refraction of Light
Refractive Index of a Transparent Substance. Light rays travel through air at a velocity of
about 300,000 km/sec, but they travel much slower through transparent solids
and liquids. The refractive index of a transparent substance is the ratio of the
velocity of light in air to the velocity in the substance. The refractive index of air
itself is 1.00. Thus, if light travels through a particular type of glass at a velocity of
200,000 km/sec, the refractive index of this glass is 300,000 divided by 200,000, or
1.50.
Refraction of Light Rays at an Interface Between Two Media with Different Refractive Indices.
When light rays traveling forward in a beam (as shown in Figure 49-1A) strike an
interface that is perpendicular to the beam, the rays enter the second medium
without deviating from their course. The only effect that occurs is decreased veloc-
ity of transmission and shorter wavelength, as shown in the figure by the shorter
distances between wave fronts.
If the light rays pass through an angulated interface as shown in Figure 49-1B,
the rays bend if the refractive indices of the two media are different from each other.
In this particular figure, the light rays are leaving air, which has a refractive index
of 1.00, and are entering a block of glass having a refractive index of 1.50. When the
beam first strikes the angulated interface, the lower edge of the beam enters the
glass ahead of the upper edge. The wave front in the upper portion of the beam con-
tinues to travel at a velocity of 300,000 km/sec, while that which entered the glass
travels at a velocity of 200,000 km/sec. This causes the upper portion of the wave
front to move ahead of the lower portion, so that the wave front is no longer ver-
tical but angulated to the right. Because the direction in which light travels is always
perpendicular to the plane of the wave front, the direction of travel of the light beam
bends downward.
This bending of light rays at an angulated interface is known as refraction. Note
particularly that the degree of refraction increases as a function of (1) the ratio of
the two refractive indices of the two transparent media and (2) the degree of angu-
lation between the interface and the entering wave front.
Application of Refractive Principles to Lenses
Convex Lens Focuses Light Rays. Figure 49-2 shows parallel light rays entering a convex
lens. The light rays passing through the center of the lens strike the lens exactly per-
pendicular to the lens surface and, therefore, pass through the lens without being
refracted. Toward either edge of the lens, however, the light rays strike a progres-
sively more angulated interface. The outer rays bend more and more toward the
center, which is called convergence of the rays. Half the bending occurs when
the rays enter the lens, and half as they exit from the opposite side. (At this time,
the student should pause and analyze why the rays bend toward the center on
leaving the lens.) Finally, if the lens has exactly the proper curvature, parallel light
613
614
Unit X The Nervous System: B. The Special Senses
A
Wave fronts
Glass
Light from
distant source
B
Figure 49-1
Figure 49-3
Light rays entering a glass surface perpendicular to the light rays
Bending of light rays at each surface of a concave spherical lens,
(A) and a glass surface angulated to the light rays (B). This figure
showing that parallel light rays are diverged.
demonstrates that the distance between waves after they enter
the glass is shortened to about two thirds that in air. It also shows
that light rays striking an angulated glass surface are bent.
Light from
Focal length
distant source
Figure 49-2
Bending of light rays at each surface of a convex spherical lens,
showing that parallel light rays are focused to a focal point.
A
rays passing through each part of the lens will be bent
exactly enough so that all the rays will pass through a
single point, which is called the focal point.
Concave Lens Diverges Light Rays. Figure 49-3 shows the
effect of a concave lens on parallel light rays. The rays
that enter the center of the lens strike an interface that
is perpendicular to the beam and, therefore, do not
refract. The rays at the edge of the lens enter the lens
ahead of the rays in the center. This is opposite to the
effect in the convex lens, and it causes the peripheral
light rays to diverge from the light rays that pass through
the center of the lens. Thus, the concave lens diverges
light rays, but the convex lens converges light rays.
Cylindrical Lens Bends Light Rays in Only One Plane—Comparison
with Spherical Lenses. Figure 49-4 shows both a convex
spherical lens and a convex cylindrical lens. Note that
B
the cylindrical lens bends light rays from the two sides
of the lens but not from the top or the bottom. That is,
Figure 49-4
bending occurs in one plane but not the other. Thus, par-
allel light rays are bent to a focal line. Conversely, light
A, Point focus of parallel light rays by a spherical convex lens.
rays that pass through the spherical lens are refracted
B, Line focus of parallel light rays by a cylindrical convex lens.
Chapter 49
The Eye: I. Optics of Vision
615
at all edges of the lens (in both planes) toward the
Focal Length of a Lens
central ray, and all the rays come to a focal point.
The cylindrical lens is well demonstrated by a test
The distance beyond a convex lens at which parallel rays
tube full of water. If the test tube is placed in a beam of
converge to a common focal point is called the focal
sunlight and a piece of paper is brought progressively
length of the lens. The diagram at the top of Figure 49-6
closer to the opposite side of the tube, a certain distance
demonstrates this focusing of parallel light rays.
will be found at which the light rays come to a focal line.
In the middle diagram, the light rays that enter the
The spherical lens is demonstrated by an ordinary mag-
convex lens are not parallel but are diverging because
nifying glass. If such a lens is placed in a beam of sun-
the origin of the light is a point source not far away from
light and a piece of paper is brought progressively closer
the lens itself. Because these rays are diverging outward
to the lens, the light rays will impinge on a common
from the point source, it can be seen from the diagram
focal point at an appropriate distance.
that they do not focus at the same distance away from
Concave cylindrical lenses diverge light rays in only
the lens as do parallel rays. In other words, when rays
one plane in the same manner that convex cylindrical
of light that are already diverging enter a convex lens,
lenses converge light rays in one plane.
the distance of focus on the other side of the lens is
farther from the lens than is the focal length of the lens
Combination of Two Cylindrical Lenses at Right Angles
for parallel rays.
Equals a Spherical Lens. Figure
49-5B shows two
The bottom diagram of Figure 49-6 shows light rays
convex cylindrical lenses at right angles to each other.
that are diverging toward a convex lens that has far
The vertical cylindrical lens converges the light rays that
greater curvature than that of the other two lenses in
pass through the two sides of the lens, and the horizon-
the figure. In this diagram, the distance from the lens at
tal lens converges the top and bottom rays. Thus, all the
which the light rays come to focus is exactly the same
light rays come to a single-point focus. In other words,
as that from the lens in the first diagram, in which the
two cylindrical lenses crossed at right angles to each other
lens is less convex but the rays entering it are parallel.
perform the same function as one spherical lens of the
This demonstrates that both parallel rays and diverging
same refractive power.
rays can be focused at the same distance beyond a lens,
provided the lens changes its convexity.
The relation of focal length of the lens, distance of the
point source of light, and distance of focus is expressed
by the following formula:
1
1
1
=
+
f
a b
Line focus
in which f is the focal length of the lens for parallel rays,
a is the distance of the point source of light from the
lens, and b is the distance of focus on the other side of
the lens.
Point source of light
Light from distant source
Focal
points
A
Point source
Point source of light
Point focus
B
Figure 49-6
Figure 49-5
The two upper lenses of this figure have the same focal length,
A, Focusing of light from a point source to a line focus by a cylin-
but the light rays entering the top lens are parallel, whereas those
drical lens. B, Two cylindrical convex lenses at right angles to
entering the middle lens are diverging; the effect of parallel versus
each other, demonstrating that one lens converges light rays in
diverging rays on the focal distance is shown. The bottom lens
one plane and the other lens converges light rays in the plane at
has far more refractive power than either of the other two lenses
a right angle. The two lenses combined give the same point focus
(i.e., has a much shorter focal length), demonstrating that the
as that obtained with a single spherical convex lens.
stronger the lens is, the nearer to the lens the point focus is.
616
Unit X The Nervous System: B. The Special Senses
Point sources
Focal points
A
Figure 49-7
A, Two point sources of light
focused at two separate points on
opposite sides of the lens. B, For-
mation of an image by a convex
B
spherical lens.
Formation of an Image by a
The refractive power of concave lenses cannot be
Convex Lens
stated in terms of the focal distance beyond the lens
because the light rays diverge, rather than focus to a
Figure
49-7A shows a convex lens with two point
point. However, if a concave lens diverges light rays at
sources of light to the left. Because light rays pass
the same rate that a 1-diopter convex lens converges
through the center of a convex lens without being
them, the concave lens is said to have a dioptric strength
refracted in either direction, the light rays from each
of -1. Likewise, if the concave lens diverges light rays as
point source of light are shown to come to a point focus
much as a +10-diopter lens converges them, this lens is
on the opposite side of the lens directly in line with the
said to have a strength of -10 diopters.
point source and the center of the lens.
Concave lenses “neutralize” the refractive power of
Any object in front of the lens is, in reality, a mosaic
convex lenses. Thus, placing a 1-diopter concave lens
of point sources of light. Some of these points are very
immediately in front of a 1-diopter convex lens results
bright, some are very weak, and they vary in color. Each
in a lens system with zero refractive power.
point source of light on the object comes to a separate
The strengths of cylindrical lenses are computed in
point focus on the opposite side of the lens in line with
the same manner as the strengths of spherical lenses,
the lens center. If a white sheet of paper is placed at the
except that the axis of the cylindrical lens must be stated
focus distance from the lens, one can see an image of
in addition to its strength. If a cylindrical lens focuses
the object, as demonstrated in Figure 49-7B. However,
this image is upside down with respect to the original
object, and the two lateral sides of the image are
reversed. This is the method by which the lens of a
camera focuses images on film.
1
diopter
Measurement of the Refractive
Power of a Lens—“Diopter”
The more a lens bends light rays, the greater is its
2
“refractive power.” This refractive power is measured in
diopters
terms of diopters. The refractive power in diopters of a
convex lens is equal to 1 meter divided by its focal
length. Thus, a spherical lens that converges parallel
light rays to a focal point 1 meter beyond the lens has
10
a refractive power of +1 diopter, as shown in Figure
diopters
49-8. If the lens is capable of bending parallel light rays
twice as much as a lens with a power of +1 diopter, it is
said to have a strength of +2 diopters, and the light rays
1 meter
come to a focal point 0.5 meter beyond the lens. A lens
capable of converging parallel light rays to a focal point
Figure 49-8
only 10 centimeters (0.10 meter) beyond the lens has a
refractive power of +10 diopters.
Effect of lens strength on the focal distance.
Chapter 49
The Eye: I. Optics of Vision
617
parallel light rays to a line focus 1 meter beyond the
The total refractive power of the internal lens of the
lens, it has a strength of +1 diopter. Conversely, if a cylin-
eye, as it normally lies in the eye surrounded by fluid
drical lens of a concave type diverges light rays as much
on each side, is only 20 diopters, about one third the
as a +1-diopter cylindrical lens converges them, it has a
total refractive power of the eye. But the importance
strength of -1 diopter. If the focused line is horizontal,
of the internal lens is that, in response to nervous
its axis is said to be 0 degrees. If it is vertical, its axis is
signals from the brain, its curvature can be increased
90 degrees.
markedly to provide “accommodation,” which is dis-
cussed later in the chapter.
Optics of the Eye
Formation of an Image on the Retina. In the same manner
that a glass lens can focus an image on a sheet of paper,
The Eye as a Camera
the lens system of the eye can focus an image on the
retina. The image is inverted and reversed with respect
The eye, shown in Figure 49-9, is optically equivalent
to the object. However, the mind perceives objects in
to the usual photographic camera. It has a lens system,
the upright position despite the upside-down orienta-
a variable aperture system (the pupil), and a retina
tion on the retina because the brain is trained to con-
that corresponds to the film. The lens system of the eye
sider an inverted image as the normal.
is composed of four refractive interfaces: (1) the inter-
face between air and the anterior surface of the
cornea, (2) the interface between the posterior surface
Mechanism of “Accommodation”
of the cornea and the aqueous humor, (3) the interface
between the aqueous humor and the anterior surface
In children, the refractive power of the lens of the eye
of the lens of the eye, and (4) the interface between
can be increased voluntarily from 20 diopters to about
the posterior surface of the lens and the vitreous
34 diopters; this in an “accommodation” of 14 diopters.
humor. The internal index of air is 1; the cornea, 1.38;
To do this, the shape of the lens is changed from that
the aqueous humor, 1.33; the crystalline lens
(on
of a moderately convex lens to that of a very convex
average), 1.40; and the vitreous humor, 1.34.
lens. The mechanism is as follows.
In a young person, the lens is composed of a strong
Reduced Eye. If all the refractive surfaces of the eye are
elastic capsule filled with viscous, proteinaceous, but
algebraically added together and then considered to
transparent fluid. When the lens is in a relaxed state
be one single lens, the optics of the normal eye may be
with no tension on its capsule, it assumes an almost
simplified and represented schematically as a “reduced
spherical shape, owing mainly to the elastic retraction
eye.” This is useful in simple calculations. In the
of the lens capsule. However, as shown in Figure
reduced eye, a single refractive surface is considered
49-10, about 70 suspensory ligaments attach radially
to exist, with its central point 17 millimeters in front of
around the lens, pulling the lens edges toward the
the retina and a total refractive power of 59 diopters
when the lens is accommodated for distant vision.
About two thirds of the 59 diopters of refractive
power of the eye is provided by the anterior surface of
Cornea
Meridional
the cornea (not by the eye lens). The principal reason
fibers
Sclera
for this is that the refractive index of the cornea is
markedly different from that of air, while the refrac-
tive index of the eye lens is not greatly different
from the indices of the aqueous humor and vitreous
humor.
Suspensory
ligaments
Choroid
Sclerocorneal
Circular
Total refractive power = 59 diopters
junction
fibers
Image
Object
Ciliary muscle
Suspensory
Lens
ligaments
Vitreous
Lens
Aqueous
Cornea
Air
humor
1.40
humor
1.38
1.00
1.34
1.33
Figure 49-9
Figure 49-10
The eye as a camera. The numbers are the refractive indices.
Mechanism of accommodation (focusing).
618
Unit X The Nervous System: B. The Special Senses
outer circle of the eyeball. These ligaments are con-
To see clearly both in the distance and nearby, an
stantly tensed by their attachments at the anterior
older person must wear bifocal glasses with the upper
border of the choroid and retina. The tension on the
segment focused for far-seeing and the lower segment
ligaments causes the lens to remain relatively flat
focused for near-seeing (e.g., for reading).
under normal conditions of the eye.
However, also located at the lateral attachments of
the lens ligaments to the eyeball is the ciliary muscle,
Pupillary Diameter
which itself has two separate sets of smooth muscle
fibers—meridional fibers and circular fibers. The
The major function of the iris is to increase the amount
meridional fibers extend from the peripheral ends of
of light that enters the eye during darkness and to
the suspensory ligaments to the corneoscleral junction.
decrease the amount of light that enters the eye in day-
When these muscle fibers contract, the peripheral
light. The reflexes for controlling this mechanism are
insertions of the lens ligaments are pulled medially
considered in the discussion of the neurology of the
toward the edges of the cornea, thereby releasing the
eye in Chapter 51.
ligaments’ tension on the lens. The circular fibers are
The amount of light that enters the eye through the
arranged circularly all the way around the ligament
pupil is proportional to the area of the pupil or to the
attachments so that when they contract, a sphincter-
square of the diameter of the pupil. The pupil of the
like action occurs, decreasing the diameter of the circle
human eye can become as small as about 1.5 millime-
of ligament attachments; this also allows the ligaments
ters and as large as 8 millimeters in diameter. The
to pull less on the lens capsule.
quantity of light entering the eye can change about 30-
Thus, contraction of either set of smooth muscle
fold as a result of changes in pupillary aperture.
fibers in the ciliary muscle relaxes the ligaments to the
lens capsule, and the lens assumes a more spherical
“Depth of Focus” of the Lens System Increases with Decreas-
shape, like that of a balloon, because of the natural
ing Pupillary Diameter. Figure 49-11 shows two eyes that
elasticity of the lens capsule.
are exactly alike except for the diameters of the pupil-
lary apertures. In the upper eye, the pupillary aperture
Accommodation Is Controlled by Parasympathetic Nerves.
is small, and in the lower eye, the aperture is large. In
The ciliary muscle is controlled almost entirely by
front of each of these two eyes are two small point
parasympathetic nerve signals transmitted to the eye
sources of light; light from each passes through the
through the third cranial nerve from the third nerve
pupillary aperture and focuses on the retina. Conse-
nucleus in the brain stem, as explained in Chapter 51.
quently, in both eyes, the retina sees two spots of light
Stimulation of the parasympathetic nerves contracts
in perfect focus. It is evident from the diagrams,
both sets of ciliary muscle fibers, which relaxes the lens
however, that if the retina is moved forward or back-
ligaments, thus allowing the lens to become thicker
ward to an out-of-focus position, the size of each spot
and increase its refractive power. With this increased
will not change much in the upper eye, but in the lower
refractive power, the eye focuses on objects nearer
eye the size of each spot will increase greatly, becom-
than when the eye has less refractive power. Conse-
ing a “blur circle.” In other words, the upper lens
quently, as a distant object moves toward the eye, the
system has far greater depth of focus than the bottom
number of parasympathetic impulses impinging on
lens system. When a lens system has great depth of
the ciliary muscle must be progressively increased for
focus, the retina can be displaced considerably from
the eye to keep the object constantly in focus. (Sym-
the focal plane or the lens strength can change con-
pathetic stimulation has an additional effect in relax-
siderably from normal and the image will still remain
ing the ciliary muscle, but this effect is so weak that it
plays almost no role in the normal accommodation
mechanism; the neurology of this is discussed in
Chapter 51.)
Point sources of light
Presbyopia. As a person grows older, the lens grows
larger and thicker and becomes far less elastic, partly
because of progressive denaturation of the lens pro-
Lens
teins. The ability of the lens to change shape decreases
Focal point
with age. The power of accommodation decreases
Lens
from about 14 diopters in a child to less than 2 diopters
by the time a person reaches 45 to 50 years; it then
decreases to essentially 0 diopters at age 70 years.
Point sources of light
Thereafter, the lens remains almost totally nonaccom-
modating, a condition known as “presbyopia.”
Once a person has reached the state of presbyopia,
each eye remains focused permanently at an almost
Figure 49-11
constant distance; this distance depends on the physi-
cal characteristics of each person’s eyes. The eyes can
Effect of small (top) and large (bottom) pupillary apertures on
no longer accommodate for both near and far vision.
“depth of focus.”
Chapter 49
The Eye: I. Optics of Vision
619
nearly in sharp focus, whereas when a lens system has
much accommodative power left, and objects closer
a “shallow” depth of focus, moving the retina only
and closer to the eye can also be focused sharply until
slightly away from the focal plane causes extreme blur-
the ciliary muscle has contracted to its limit. In old age,
when the lens becomes “presbyopic,” a farsighted
ring.
person is often unable to accommodate the lens suffi-
The greatest possible depth of focus occurs when the
ciently to focus even distant objects, much less near
pupil is extremely small. The reason for this is that,
objects.
with a very small aperture, almost all the rays pass
through the center of the lens, and the central most
Myopia
(Nearsightedness). In myopia, or
“nearsighted-
rays are always in focus, as explained earlier.
ness,” when the ciliary muscle is completely relaxed, the
light rays coming from distant objects are focused in
front of the retina, as shown in the bottom panel of
Errors of Refraction
Figure 49-12. This is usually due to too long an eyeball,
but it can result from too much refractive power in the
Emmetropia (Normal Vision). As shown in Figure 49-12, the
lens system of the eye.
eye is considered to be normal, or “emmetropic,” if par-
No mechanism exists by which the eye can decrease
allel light rays from distant objects are in sharp focus on
the strength of its lens to less than that which exists
the retina when the ciliary muscle is completely relaxed.
when the ciliary muscle is completely relaxed. A myopic
This means that the emmetropic eye can see all distant
person has no mechanism by which to focus distant
objects clearly with its ciliary muscle relaxed. However,
objects sharply on the retina. However, as an object
to focus objects at close range, the eye must contract its
moves nearer to the person’s eye, it finally gets close
ciliary muscle and thereby provide appropriate degrees
enough that its image can be focused. Then, when the
of accommodation.
object comes still closer to the eye, the person can use
the mechanism of accommodation to keep the image
Hyperopia
(Farsightedness). Hyperopia, which is also
focused clearly. A myopic person has a definite limiting
known as “farsightedness,” is usually due to either an
“far point” for clear vision.
eyeball that is too short or, occasionally, a lens system
that is too weak. In this condition, as seen in the middle
Correction of Myopia and Hyperopia by Use of Lenses.
panel of Figure 49-12, parallel light rays are not bent
It will be recalled that light rays passing through a
sufficiently by the relaxed lens system to come to focus
concave lens diverge. If the refractive surfaces of the eye
by the time they reach the retina. To overcome this
have too much refractive power, as in myopia, this
abnormality, the ciliary muscle must contract to increase
excessive refractive power can be neutralized by placing
the strength of the lens. By using the mechanism of
in front of the eye a concave spherical lens, which will
accommodation, a farsighted person is capable of focus-
diverge rays. Such correction is demonstrated in the
ing distant objects on the retina. If the person has used
upper diagram of Figure 49-13.
only a small amount of strength in the ciliary muscle to
Conversely, in a person who has hyperopia—that is,
accommodate for the distant objects, he or she still has
someone who has too weak a lens system—the abnor-
mal vision can be corrected by adding refractive power
using a convex lens in front of the eye. This correction
is demonstrated in the lower diagram of Figure 49-13.
One usually determines the strength of the concave
or convex lens needed for clear vision by “trial and
error”—that is, by trying first a strong lens and then a
Emmetropia
Hyperopia
Myopia
Figure 49-12
Figure 49-13
Parallel light rays focus on the retina in emmetropia, behind the
Correction of myopia with a concave lens, and correction of hyper-
retina in hyperopia, and in front of the retina in myopia.
opia with a convex lens.
620
Unit X The Nervous System: B. The Special Senses
stronger or weaker lens until the one that gives the best
astigmatism, the usual procedure is to find a spherical
visual acuity is found.
lens by trial and error that corrects the focus in one of
the two planes of the astigmatic lens. Then an additional
Astigmatism
cylindrical lens is used to correct the remaining error
Astigmatism is a refractive error of the eye that causes
in the remaining plane. To do this, both the axis and
the visual image in one plane to focus at a different dis-
the strength of the required cylindrical lens must be
tance from that of the plane at right angles. This most
determined.
often results from too great a curvature of the cornea
There are several methods for determining the axis
in one plane of the eye. An example of an astigmatic
of the abnormal cylindrical component of the lens
lens would be a lens surface like that of an egg lying
system of an eye. One of these methods is based on the
sidewise to the incoming light. The degree of curvature
use of parallel black bars of the type shown in Figure
in the plane through the long axis of the egg is not
49-15. Some of these parallel bars are vertical, some
nearly as great as the degree of curvature in the plane
horizontal, and some at various angles to the vertical
through the short axis.
and horizontal axes. After placing various spherical
Because the curvature of the astigmatic lens along
lenses in front of the astigmatic eye, a strength of lens
one plane is less than the curvature along the other
is usually found that causes sharp focus of one set of
plane, light rays striking the peripheral portions of the
parallel bars but does not correct the fuzziness of the
lens in one plane are not bent nearly as much as the rays
set of bars at right angles to the sharp bars. It can be
striking the peripheral portions of the other plane. This
shown from the physical principles of optics discussed
is demonstrated in Figure
49-14, which shows rays
earlier in this chapter that the axis of the out-of-focus
of light originating from a point source and passing
cylindrical component of the optical system is parallel
through an oblong, astigmatic lens. The light rays in the
to the bars that are fuzzy. Once this axis is found, the
vertical plane, indicated by plane BD, are refracted
examiner tries progressively stronger and weaker posi-
greatly by the astigmatic lens because of the greater cur-
tive or negative cylindrical lenses, the axes of which
vature in the vertical direction than in the horizontal
are placed in line with the out-of-focus bars, until the
direction. By contrast, the light rays in the horizontal
patient sees all the crossed bars with equal clarity. When
plane, indicated by plane AC, are not bent nearly as
this has been accomplished, the examiner directs the
much as the light rays in vertical plane BD. It is obvious
optician to grind a special lens combining both the
that light rays passing through an astigmatic lens do not
spherical correction and the cylindrical correction at
all come to a common focal point, because the light rays
the appropriate axis.
passing through one plane focus far in front of those
passing through the other plane.
Correction of Optical Abnormalities by Use of Contact Lenses
The accommodative power of the eye can never
Glass or plastic contact lenses can be inserted that fit
compensate for astigmatism because, during accommo-
snugly against the anterior surface of the cornea. These
dation, the curvature of the eye lens changes approxi-
lenses are held in place by a thin layer of tear fluid that
mately equally in both planes; therefore, in astigmatism,
fills the space between the contact lens and the anterior
each of the two planes requires a different degree of
eye surface.
accommodation. Thus, without the aid of glasses, a
A special feature of the contact lens is that it nullifies
person with astigmatism never sees in sharp focus.
almost entirely the refraction that normally occurs at
the anterior surface of the cornea. The reason for this is
Correction of Astigmatism with a Cylindrical Lens. One
may consider an astigmatic eye as having a lens system
made up of two cylindrical lenses of different strengths
and placed at right angles to each other. To correct for
12
Point source
of light
A B
C
D
Focal line
Plane AC
for plane BD
Plane BD
(less refractive
(more refractive
power)
power)
Focal line
for plane AC
Figure 49-14
Figure 49-15
Astigmatism, demonstrating that light rays focus at one focal dis-
tance in one focal plane (plane AC) and at another focal distance
Chart composed of parallel black bars at different angular orien-
in the plane at a right angle (plane BD).
tations for determining the axis of astigmatism.
Chapter 49
The Eye: I. Optics of Vision
621
that the tears between the contact lens and the cornea
have a refractive index almost equal to that of the
cornea, so that the anterior surface of the cornea no
longer plays a significant role in the eye’s optical system.
Instead, the outer surface of the contact lens plays the
major role. Thus, the refraction of this surface of the
contact lens substitutes for the cornea’s usual refraction.
2 µm
1 mm
This is especially important in people whose eye refrac-
tive errors are caused by an abnormally shaped cornea,
such as those who have an odd-shaped, bulging
cornea—a condition called keratoconus. Without the
contact lens, the bulging cornea causes such severe
17 mm
10 meters
abnormality of vision that almost no glasses can correct
the vision satisfactorily; when a contact lens is used,
however, the corneal refraction is neutralized, and
normal refraction by the outer surface of the contact
Figure 49-16
lens is substituted.
The contact lens has several other advantages as well,
Maximum visual acuity for two point sources of light.
including (1) the lens turns with the eye and gives a
broader field of clear vision than glasses do, and (2) the
contact lens has little effect on the size of the object the
The normal visual acuity of the human eye for dis-
person sees through the lens, whereas lenses placed 1
centimeter or so in front of the eye do affect the size of
criminating between point sources of light is about 25
the image, in addition to correcting the focus.
seconds of arc. That is, when light rays from two sepa-
rate points strike the eye with an angle of at least 25
Cataracts
seconds between them, they can usually be recognized
“Cataracts” are an especially common eye abnormality
as two points instead of one. This means that a person
that occurs mainly in older people. A cataract is a cloudy
with normal visual acuity looking at two bright pin-
or opaque area or areas in the lens. In the early stage of
point spots of light 10 meters away can barely distin-
cataract formation, the proteins in some of the lens
guish the spots as separate entities when they are 1.5
fibers become denatured. Later, these same proteins
to 2 millimeters apart.
coagulate to form opaque areas in place of the normal
The fovea is less than 0.5 millimeter (less than 500
transparent protein fibers.
When a cataract has obscured light transmission so
micrometers) in diameter, which means that maximum
greatly that it seriously impairs vision, the condition can
visual acuity occurs in less than 2 degrees of the visual
be corrected by surgical removal of the lens. When this
field. Outside this foveal area, the visual acuity
is done, the eye loses a large portion of its refractive
becomes progressively poorer, decreasing more than
power, which must be replaced by a powerful convex
10-fold as the periphery is approached. This is caused
lens in front of the eye; usually, however, an artificial
by the connection of more and more rods and cones
plastic lens is implanted in the eye in place of the
to each optic nerve fiber in the nonfoveal,
removed lens.
more peripheral parts of the retina, as discussed in
Chapter 51.
Visual Acuity
Clinical Method for Stating Visual Acuity. The chart for
testing eyes usually consists of letters of different sizes
Theoretically, light from a distant point source, when
placed 20 feet away from the person being tested. If
focused on the retina, should be infinitely small.
the person can see well the letters of a size that he or
However, because the lens system of the eye is never
she should be able to see at 20 feet, the person is said
perfect, such a retinal spot ordinarily has a total diam-
to have 20/20 vision—that is, normal vision. If the
eter of about 11 micrometers, even with maximal res-
person can see only letters that he or she should be
olution of the normal eye optical system. The spot is
able to see at 200 feet, the person is said to have 20/200
brightest in its center and shades off gradually toward
vision. In other words, the clinical method for express-
the edges, as shown by the two-point images in
ing visual acuity is to use a mathematical fraction that
Figure 49-16.
expresses the ratio of two distances, which is also the
The average diameter of the cones in the fovea of
ratio of one’s visual acuity to that of a person with
the retina—the central part of the retina, where vision
normal visual acuity.
is most highly developed—is about 1.5 micrometers,
which is one seventh the diameter of the spot of light.
Determination of Distance
Nevertheless, because the spot of light has a bright
center point and shaded edges, a person can normally
of an Object from the Eye—
distinguish two separate points if their centers lie as
“Depth Perception”
much as 2 micrometers apart on the retina, which is
slightly greater than the width of a foveal cone. This
A person normally perceives distance by three major
discrimination between points is also shown in Figure
means: (1) the sizes of the images of known objects on
49-16.
the retina, (2) the phenomenon of moving parallax,
622
Unit X The Nervous System: B. The Special Senses
and (3) the phenomenon of stereopsis. This ability to
determine distance is called depth perception.
Object of known
distance and size
Determination of Distance by Sizes of Retinal Images of Known
Objects. If one knows that a person being viewed is 6
1. Size of image
feet tall, one can determine how far away the person
is simply by the size of the person’s image on the
retina. One does not consciously think about the size,
Unknown
but the brain has learned to calculate automatically
object
from image sizes the distances of objects when the
dimensions are known.
Determination of Distance by Moving Parallax. Another
2. Stereopsis
important means by which the eyes determine distance
Figure 49-17
is that of moving parallax. If an individual looks off
into the distance with the eyes completely still, he or
Perception of distance (1) by the size of the image on the retina
she perceives no moving parallax, but when the person
and (2) as a result of stereopsis.
moves his or her head to one side or the other, the
images of close-by objects move rapidly across the
retinas, while the images of distant objects remain
the lens system of the eye. After passing through the
almost completely stationary. For instance, by moving
lens system, they are parallel with one another because
the head 1 inch to the side when the object is only 1
the retina is located one focal length distance behind
inch in front of the eye, the image moves almost all the
the lens system. Then, when these parallel rays pass into
way across the retinas, whereas the image of an object
an emmetropic eye of another person, they focus again
to a point focus on the retina of the second person,
200 feet away from the eyes does not move percepti-
because his or her retina is also one focal length dis-
bly. Thus, by using this mechanism of moving parallax,
tance behind the lens. Any spot of light on the retina of
one can tell the relative distances of different objects
the observed eye projects to a focal spot on the retina
even though only one eye is used.
of the observing eye. Thus, if the retina of one person is
made to emit light, the image of his or her retina will be
Determination of Distance by Stereopsis—Binocular Vision.
focused on the retina of the observer, provided the two
Another method by which one perceives parallax is
eyes are emmetropic and are simply looking into each
that of “binocular vision.” Because one eye is a little
other.
more than 2 inches to one side of the other eye, the
To make an ophthalmoscope, one need only devise a
images on the two retinas are different from each
means for illuminating the retina to be examined. Then,
the reflected light from that retina can be seen by the
other. For instance, an object 1 inch in front of the nose
observer simply by putting the two eyes close to each
forms an image on the left side of the retina of the left
other. To illuminate the retina of the observed eye, an
eye but on the right side of the retina of the right eye,
angulated mirror or a segment of a prism is placed in
whereas a small object 20 feet in front of the nose has
front of the observed eye in such a manner, as shown in
its image at closely corresponding points in the centers
Figure 49-18, that light from a bulb is reflected into the
of the two retinas. This type of parallax is demon-
observed eye. Thus, the retina is illuminated through the
strated in Figure 49-17, which shows the images of a
pupil, and the observer sees into the subject’s pupil by
red spot and a yellow square actually reversed on the
looking over the edge of the mirror or prism or through
two retinas because they are at different distances in
an appropriately designed prism.
front of the eyes. This gives a type of parallax that is
present all the time when both eyes are being used. It
is almost entirely this binocular parallax (or stereop-
Observed eye
Observer’s eye
sis) that gives a person with two eyes far greater ability
Mirror
to judge relative distances when objects are nearby
than a person who has only one eye. However, stere-
opsis is virtually useless for depth perception at dis-
tances beyond 50 to 200 feet.
Illuminate retina
Corrective lens in
Ophthalmoscope
showing blood
turret
(- 4 diopters
vessel
for normal eyes)
The ophthalmoscope is an instrument through which an
Collimating lens
observer can look into another person’s eye and see the
retina with clarity. Although the ophthalmoscope
appears to be a relatively complicated instrument, its
principles are simple. The basic components are shown
in Figure 49-18 and can be explained as follows.
Figure 49-18
If a bright spot of light is on the retina of an
emmetropic eye, light rays from this spot diverge toward
Optical system of the ophthalmoscope.
Chapter 49
The Eye: I. Optics of Vision
623
It is clear that these principles apply only to people
Formation of Aqueous Humor
with completely emmetropic eyes. If the refractive
by the Ciliary Body
power of either the observed eye or the observer’s eye
is abnormal, it is necessary to correct the refractive
Aqueous humor is formed in the eye at an average rate
power for the observer to see a sharp image of the
of 2 to 3 microliters each minute. Essentially all of it is
observed retina. The usual ophthalmoscope has a series
secreted by the ciliary processes, which are linear folds
of very small lenses mounted on a turret so that the
projecting from the ciliary body into the space behind
turret can be rotated from one lens to another until the
the iris where the lens ligaments and ciliary muscle
correction for abnormal refraction is made by selecting
attach to the eyeball. A cross section of these ciliary
a lens of appropriate strength. In normal young adults,
processes is shown in Figure 49-20, and their relation to
natural accommodative reflexes occur that cause an
the fluid chambers of the eye can be seen in Figure
approximate +2-diopter increase in strength of the lens
49-19. Because of their folded architecture, the total
of each eye. To correct for this, it is necessary that
surface area of the ciliary processes is about 6 square
the lens turret be rotated to approximately -4-diopter
centimeters in each eye—a large area, considering the
correction.
small size of the ciliary body. The surfaces of these
processes are covered by highly secretory epithelial
cells, and immediately beneath them is a highly vascu-
Fluid System of the Eye—
lar area.
Aqueous humor is formed almost entirely as an active
Intraocular Fluid
secretion by the epithelium of the ciliary processes.
The eye is filled with intraocular fluid, which maintains
Secretion begins with active transport of sodium ions
sufficient pressure in the eyeball to keep it distended.
into the spaces between the epithelial cells. The sodium
Figure 49-19 demonstrates that this fluid can be divided
ions pull chloride and bicarbonate ions along with them
into two portions—aqueous humor, which lies in front
to maintain electrical neutrality. Then all these ions
of the lens, and vitreous humor, which is between the
together cause osmosis of water from the blood capil-
posterior surface of the lens and the retina. The aqueous
laries lying below into the same epithelial intercellular
humor is a freely flowing fluid, whereas the vitreous
spaces, and the resulting solution washes from the
humor, sometimes called the vitreous body, is a gelati-
spaces of the ciliary processes into the anterior chamber
nous mass held together by a fine fibrillar network com-
of the eye. In addition, several nutrients are transported
posed primarily of greatly elongated proteoglycan
across the epithelium by active transport or facilitated
molecules. Both water and dissolved substances can
diffusion; they include amino acids, ascorbic acid, and
diffuse slowly in the vitreous humor, but there is little
glucose.
flow of fluid.
Aqueous humor is continually being formed and
reabsorbed. The balance between formation and reab-
Outflow of Aqueous Humor
sorption of aqueous humor regulates the total volume
from the Eye
and pressure of the intraocular fluid.
After aqueous humor is formed by the ciliary processes,
it first flows, as shown in Figure 49-19, through the pupil
into the anterior chamber of the eye. From here, the fluid
flows anterior to the lens and into the angle between the
Aqueous humor Iris
Flow of fluid
Spaces of Fontana
Ciliary processes
Formation
Formation
of aqueous
Canal of Schlemm
of aqueous
humor
humor
Ciliary body
Lens
Diffusion of
fluid and other
Vitreous
constituents
humor
Filtration and
diffusion at
retinal vessels
Ciliary muscle
Vascular layer
Optic nerve
Figure 49-20
Figure 49-19
Anatomy of the ciliary processes. Aqueous humor is formed on
Formation and flow of fluid in the eye.
surfaces.
624
Unit X The Nervous System: B. The Special Senses
Pressure applied
Cornea
Central plunger
Canal of Schlemm
Trabeculae
Aqueous veins
Iris
Footplate
Blood veins
Sclera
Figure 49-21
Intraocular pressure
Anatomy of the iridocorneal angle, showing the system for outflow
of aqueous humor from the eyeball into the conjunctival veins.
Figure 49-22
Principles of the tonometer.
cornea and the iris, then through a meshwork of trabec-
ulae, finally entering the canal of Schlemm, which
empties into extraocular veins. Figure 49-21 demon-
trabeculae through which the fluid must percolate on its
strates the anatomical structures at this iridocorneal
way from the lateral angles of the anterior chamber to
angle, showing that the spaces between the trabeculae
the wall of the canal of Schlemm. These trabeculae have
extend all the way from the anterior chamber to the
minute openings of only 2 to 3 micrometers. The rate of
canal of Schlemm. The canal of Schlemm is a thin-
fluid flow into the canal increases markedly as the pres-
walled vein that extends circumferentially all the way
sure rises. At about 15 mm Hg in the normal eye, the
around the eye. Its endothelial membrane is so porous
amount of fluid leaving the eye by way of the canal of
that even large protein molecules, as well as small par-
Schlemm usually averages 2.5 ml/min and equals the
ticulate matter up to the size of red blood cells, can pass
inflow of fluid from the ciliary body. The pressure nor-
from the anterior chamber into the canal of Schlemm.
mally remains at about this level of 15 mm Hg.
Even though the canal of Schlemm is actually a venous
blood vessel, so much aqueous humor normally flows
Mechanism for Cleansing the Trabecular Spaces and Intraocular
into it that it is filled only with aqueous humor rather
Fluid. When large amounts of debris are present in the
than with blood. The small veins that lead from the canal
aqueous humor, as occurs after hemorrhage into the eye
of Schlemm to the larger veins of the eye usually contain
or during intraocular infection, the debris is likely to
only aqueous humor, and they are called aqueous veins.
accumulate in the trabecular spaces leading from the
anterior chamber to the canal of Schlemm; this debris
Intraocular Pressure
can prevent adequate reabsorption of fluid from the
anterior chamber, sometimes causing “glaucoma,” as
The average normal intraocular pressure is about
explained subsequently. However, on the surfaces of the
15 mm Hg, with a range from 12 to 20 mm Hg.
trabecular plates are large numbers of phagocytic cells.
Immediately outside the canal of Schlemm is a layer of
Tonometry. Because it is impractical to pass a needle into
interstitial gel that contains large numbers of reticu-
a patient’s eye to measure intraocular pressure, this
loendothelial cells that have an extremely high capacity
pressure is measured clinically by using a “tonometer,”
for engulfing debris and digesting it into small molecu-
the principle of which is shown in Figure 49-22. The
lar substances that can then be absorbed. Thus, this
cornea of the eye is anesthetized with a local anesthetic,
phagocytic system keeps the trabecular spaces cleaned.
and the footplate of the tonometer is placed on the
The surface of the iris and other surfaces of the eye
cornea. A small force is then applied to a central
behind the iris are covered with an epithelium that is
plunger, causing the part of the cornea beneath the
capable of phagocytizing proteins and small particles
plunger to be displaced inward. The amount of dis-
from the aqueous humor, thereby helping to maintain a
placement is recorded on the scale of the tonometer,
clear fluid.
and this is calibrated in terms of intraocular pressure.
“Glaucoma,” a Principal Cause of Blindness. Glaucoma is one
Regulation of Intraocular Pressure. Intraocular pressure
of the most common causes of blindness. It is a disease
remains constant in the normal eye, usually within
of the eye in which the intraocular pressure becomes
±2 mm Hg of its normal level, which averages about
pathologically high, sometimes rising acutely to 60 to
15 mm Hg. The level of this pressure is determined
70 mm Hg. Pressures above 25 to 30 mm Hg can cause
mainly by the resistance to outflow of aqueous humor
loss of vision when maintained for long periods.
from the anterior chamber into the canal of Schlemm.
Extremely high pressures can cause blindness within
This outflow resistance results from the meshwork of
days or even hours. As the pressure rises, the axons of
Chapter 49
The Eye: I. Optics of Vision
625
the optic nerve are compressed where they leave the
Congdon NG, Friedman DS, Lietman T: Important causes of
eyeball at the optic disc. This compression is believed to
visual impairment in the world today. JAMA 290:2057,
block axonal flow of cytoplasm from the retinal neu-
2003.
ronal cell bodies into the optic nerve fibers leading to
Corbett JJ: The bedside and office neuro-ophthalmology
the brain. The result is lack of appropriate nutrition of
examination. Semin Neurol 23:63, 2003.
the fibers, which eventually causes death of the involved
Doane JF: Accommodating intraocular lenses. Curr Opin
fibers. It is possible that compression of the retinal
Ophthalmol 15:16, 2004.
artery, which enters the eyeball at the optic disc, also
Farr AK, Guyton DL: Strabismus after retinal detachment
adds to the neuronal damage by reducing nutrition to
surgery. Curr Opin Ophthalmol 11:207, 2000.
the retina.
Gilmartin B: Myopia: precedents for research in the twenty-
In most cases of glaucoma, the abnormally high pres-
first century. Clin Exp Ophthalmol 32:305, 2004.
sure results from increased resistance to fluid outflow
Guyton DL: Sights and Sounds in Ophthalmology: Ocular
through the trabecular spaces into the canal of Schlemm
Motility and Binocular Vision. St Louis: CV Mosby,
at the iridocorneal junction. For instance, in acute
1989.
eye inflammation, white blood cells and tissue debris
Khaw PT, Shah P, Elkington AR: Glaucoma—1: diagnosis.
can block these trabecular spaces and cause an acute
BMJ 328:97, 2004.
increase in intraocular pressure. In chronic conditions,
Krag S, Andreassen TT: Mechanical properties of the human
especially in older individuals, fibrous occlusion of the
lens capsule. Prog Retin Eye Res 22:749, 2003.
trabecular spaces appears to be the likely culprit.
Mathias RT, Rae JL, Baldo GJ: Physiological properties of
Glaucoma can sometimes be treated by placing drops
the normal lens. Physiol Rev 77:21, 1997.
in the eye that contain a drug that diffuses into the
McBrien NA, Gentle A: Role of the sclera in the develop-
eyeball and reduces the secretion or increases the
ment and pathological complications of myopia. Prog
absorption of aqueous humor. When drug therapy fails,
Retin Eye Res 22:307, 2003.
operative techniques to open the spaces of the trabec-
O’Shea J, Walsh V: Visual awareness: the eye fields have it?
ulae or to make channels to allow fluid to flow directly
Curr Biol 14:R279, 2004.
from the fluid space of the eyeball into the subconjunc-
Schaeffel F, Simon P, Feldkaemper M, et al: Molecular
tival space outside the eyeball can often effectively
biology of myopia. Clin Exp Optom 86:295, 2003.
reduce the pressure.
Schwartz K, Budenz D: Current management of glaucoma.
Curr Opin Ophthalmol 15:119, 2004.
Smith G: The optical properties of the crystalline lens and
their significance. Clin Exp Optom 86:3, 2003.
References
Smith G, Atchison DA: The Eye and Visual Optical Instru-
ments. Cambridge: Cambridge University Press, 1997.
Buisseret P: Influence of extraocular muscle proprioception
Weinreb RN, Khaw PT: Primary open-angle glaucoma.
on vision. Physiol Rev 75:323, 1995.
Lancet 363:1711, 2004.
C H A P T E R
5
0
The Eye: II. Receptor and
Neural Function of the Retina
The retina is the light-sensitive portion of the eye
that contains (1) the cones, which are responsible
for color vision, and (2) the rods, which are mainly
responsible for black and white vision and vision in
the dark. When either rods or cones are excited,
signals are transmitted first through successive
layers of neurons in the retina itself and, finally, into
optic nerve fibers and the cerebral cortex. The
purpose of this chapter is to explain the mechanisms by which the rods and
cones detect light and color and convert the visual image into optic nerve
signals.
Anatomy and Function of the Structural
Elements of the Retina
Layers of the Retina. Figure 50-1 shows the functional components of the retina which
are arranged in layers from the outside to the inside as follows: (1) pigmented layer,
(2) layer of rods and cones projecting to the pigment, (3) outer nuclear layer con-
taining the cell bodies of the rods and cones, (4) outer plexiform layer, (5) inner
nuclear layer, (6) inner plexiform layer, (7) ganglionic layer, (8) layer of optic nerve
fibers, and (9) inner limiting membrane.
After light passes through the lens system of the eye and then through the vitre-
ous humor, it enters the retina from the inside (see Figure 50-1); that is, it passes first
through the ganglion cells and then through the plexiform and nuclear layers before
it finally reaches the layer of rods and cones located all the way on the outer edge
of the retina. This distance is a thickness of several hundred micrometers; visual
acuity is decreased by this passage through such nonhomogeneous tissue. However,
in the central foveal region of the retina, as discussed subsequently, the inside layers
are pulled aside to decrease this loss of acuity.
Foveal Region of the Retina and Its Importance in Acute Vision. The fovea is a minute area
in the center of the retina, shown in Figure 50-2, occupying a total area a little more
than 1 square millimeter; it is especially capable of acute and detailed vision. The
central fovea, only 0.3 millimeter in diameter, is composed almost entirely of cones;
these cones have a special structure that aids their detection of detail in the visual
image. That is, the foveal cones have especially long and slender bodies, in con-
tradistinction to the much fatter cones located more peripherally in the retina. Also,
in the foveal region, the blood vessels, ganglion cells, inner nuclear layer of cells,
and plexiform layers are all displaced to one side rather than resting directly on top
of the cones. This allows light to pass unimpeded to the cones.
Rods and Cones. Figure 50-3 is a diagrammatic representation of the essential com-
ponents of a photoreceptor (either a rod or a cone). As shown in Figure 50-4, the
outer segment of the cone is conical in shape. In general, the rods are narrower and
longer than the cones, but this is not always the case. In the peripheral portions of
the retina, the rods are 2 to 5 micrometers in diameter, whereas the cones are 5 to
8 micrometers in diameter; in the central part of the retina, in the fovea, there are
rods, and the cones are slender and have a diameter of only 1.5 micrometers.
To the right in Figure 50-3 are labeled the major functional segments of either a
rod or a cone: (1) the outer segment, (2) the inner segment, (3) the nucleus, and (4)
the synaptic body. The light-sensitive photochemical is found in the outer segment.
In the case of the rods, this is rhodopsin; in the cones, it is one of three “color”
626
Chapter 50
The Eye: II. Receptor and Neural Function of the Retina
627
Pigmented layer
Rod
Cone
Cone
Outer nuclear layer
Outer plexiform
Distal
layer
Vertical
Horizontal cell
Bipolar
pathway
Bipolar
Lateral pathway
cell
Inner nuclear layer
Amacrine
cell
cell
Amacrine cell
Proximal
Inner plexiform
layer
Ganglion cell
Ganglion cell layer
To optic nerve
Stratum opticum
Figure 50-1
Inner limiting
Layers of retina.
DIRECTION OF LIGHT
membrane
Figure 50-2
Photomicrograph of the macula and of the fovea in its center. Note that the inner layers of the retina are pulled to the side to decrease
interference with light transmission. (From Fawcett DW: Bloom and Fawcett: A Textbook of Histology, 11th ed. Philadelphia: WB Saunders,
1986; courtesy H. Mizoguchi.)
photochemicals, usually called simply color pigments,
proteins. The concentrations of these photosensitive
that function almost exactly the same as rhodopsin
pigments in the discs are so great that the pigments
except for differences in spectral sensitivity.
themselves constitute about 40 per cent of the entire
Note in the outer segments of the rods and cones in
mass of the outer segment.
Figures 50-3 and 50-4 the large numbers of discs. Each
The inner segment of the rod or cone contains the
of the discs is actually an infolded shelf of cell mem-
usual cytoplasm with cytoplasmic organelles. Particu-
brane. There are as many as 1000 discs in each rod or
larly important are the mitochondria; as explained later,
cone.
these mitochondria play the important role of provid-
Both rhodopsin and the color pigments are conju-
ing energy for function of the photoreceptors.
gated proteins. They are incorporated into the mem-
The synaptic body is the portion of the rod or cone
branes of the discs in the form of transmembrane
that connects with subsequent neuronal cells, the
628
Unit X The Nervous System: B. The Special Senses
lighting of the retina rather than the normal contrast
between dark and light spots required for formation of
precise images.
The importance of melanin in the pigment layer is
Membrane shelves
Outer segment
well illustrated by its absence in albinos, people who are
lined with rhodopsin
hereditarily lacking in melanin pigment in all parts of
or color pigment
their bodies. When an albino enters a bright room, light
that impinges on the retina is reflected in all directions
inside the eyeball by the unpigmented surfaces of
the retina and by the underlying sclera, so that a
Mitochondria
single discrete spot of light that would normally
excite only a few rods or cones is reflected everywhere
Inner segment
and excites many receptors. Therefore, the visual acuity
of albinos, even with the best optical correction, is
seldom better than 20/100 to 20/200 rather than the
normal 20/20 values.
The pigment layer also stores large quantities of
Outer limiting
vitamin A. This vitamin A is exchanged back and forth
membrane
through the cell membranes of the outer segments of
Nucleus
the rods and cones, which themselves are embedded in
the pigment. We show later that vitamin A is an impor-
tant precursor of the photosensitive chemicals of the
rods and cones.
Synaptic body
Blood Supply of the Retina—The Central Retinal Artery and the
Choroid. The nutrient blood supply for the internal
Figure 50-3
layers of the retina is derived from the central retinal
artery, which enters the eyeball through the center of
the optic nerve and then divides to supply the entire
Schematic drawing of the functional parts of the rods and cones.
inside retinal surface. Thus, the inner layers of the retina
have their own blood supply independent of the other
structures of the eye.
However, the outermost layer of the retina is adher-
ent to the choroid, which is also a highly vascular tissue
lying between the retina and the sclera. The outer layers
of the retina, especially the outer segments of the rods
and cones, depend mainly on diffusion from the choroid
blood vessels for their nutrition, especially for their
oxygen.
Retinal Detachment. The neural retina occasionally
detaches from the pigment epithelium. In some instances,
the cause of such detachment is injury to the eyeball
that allows fluid or blood to collect between the neural
retina and the pigment epithelium. Detachment is occa-
sionally caused by contracture of fine collagenous fibrils
in the vitreous humor, which pull areas of the retina
toward the interior of the globe.
Partly because of diffusion across the detachment gap
and partly because of the independent blood supply
to the neural retina through the retinal artery, the
detached retina can resist degeneration for days and can
become functional again if it is surgically replaced in its
Figure 50-4
normal relation with the pigment epithelium. If it is not
Membranous structures of the outer segments of a rod (left) and
replaced soon, however, the retina will be destroyed and
a cone (right). (Courtesy Dr. Richard Young.)
will be unable to function even after surgical repair.
horizontal and bipolar cells, that represent the next
Photochemistry of Vision
stages in the vision chain.
Both rods and cones contain chemicals that decom-
Pigment Layer of the Retina. The black pigment melanin in
pose on exposure to light and, in the process, excite the
the pigment layer prevents light reflection throughout
nerve fibers leading from the eye. The light-sensitive
the globe of the eyeball; this is extremely important for
chemical in the rods is called rhodopsin; the light-
clear vision. This pigment performs the same function
in the eye as the black coloring inside the bellows of a
sensitive chemicals in the cones, called cone pigments
camera. Without it, light rays would be reflected in all
or color pigments, have compositions only slightly dif-
directions within the eyeball and would cause diffuse
ferent from that of rhodopsin.
Chapter 50
The Eye: II. Receptor and Neural Function of the Retina
629
In this section, we discuss principally the photo-
is extremely unstable and decays in nanoseconds to
chemistry of rhodopsin, but the same principles can be
lumirhodopsin. This then decays in microseconds
applied to the cone pigments.
to metarhodopsin I, then in about a millisecond to
metarhodopsin II, and finally, much more slowly (in
seconds), into the completely split products scotopsin
Rhodopsin-Retinal Visual Cycle, and
and all-trans retinal.
Excitation of the Rods
It is the metarhodopsin II, also called activated
rhodopsin, that excites electrical changes in the rods,
Rhodopsin and Its Decomposition by Light Energy. The outer
and the rods then transmit the visual image into the
segment of the rod that projects into the pigment layer
central nervous system in the form of optic nerve
of the retina has a concentration of about 40 per cent
action potential, as we discuss later.
of the light-sensitive pigment called rhodopsin, or
visual purple. This substance is a combination of the
Re-formation of Rhodopsin. The first stage in re-formation
protein scotopsin and the carotenoid pigment retinal
of rhodopsin, as shown in Figure 50-5, is to reconvert
(also called “retinene”). Furthermore, the retinal is a
the all-trans retinal into 11-cis retinal. This process
particular type called 11-cis retinal. This cis form of
requires metabolic energy and is catalyzed by the
retinal is important because only this form can bind
enzyme retinal isomerase. Once the 11-cis retinal is
with scotopsin to synthesize rhodopsin.
formed, it automatically recombines with the scotopsin
When light energy is absorbed by rhodopsin, the
to re-form rhodopsin, which then remains stable until
rhodopsin begins to decompose within a very small
its decomposition is again triggered by absorption of
fraction of a second, as shown at the top of Figure
light energy.
50-5. The cause of this is photoactivation of electrons
in the retinal portion of the rhodopsin, which leads to
Role of Vitamin A for Formation of Rhodopsin. Note in
instantaneous change of the cis form of retinal into an
Figure 50-5 that there is a second chemical route by
all-trans form that still has the same chemical structure
which all-trans retinal can be converted into 11-cis
as the cis form but has a different physical structure—
retinal. This is by conversion of the all-trans retinal first
a straight molecule rather than an angulated molecule.
into all-trans retinol, which is one form of vitamin A.
Because the three-dimensional orientation of the
Then the all-trans retinol is converted into
11-cis
reactive sites of the all-trans retinal no longer fits
retinol under the influence of the enzyme isomerase.
with the orientation of the reactive sites on the protein
Finally, the
11-cis retinol is converted into
11-cis
scotopsin, the all-trans retinal begins to pull
retinal, which combines with scotopsin to form new
away from the scotopsin. The immediate product is
rhodopsin.
bathorhodopsin, which is a partially split combination
Vitamin A is present both in the cytoplasm of the
of the all-trans retinal and scotopsin. Bathorhodopsin
rods and in the pigment layer of the retina. Therefore,
vitamin A is normally always available to form new
retinal when needed. Conversely, when there is excess
retinal in the retina, it is converted back into vitamin
Light energy
A, thus reducing the amount of light-sensitive pigment
Rhodopsin
Bathorhodopsin
in the retina. We shall see later that this interconver-
(p sec)
(nsec)
sion between retinal and vitamin A is especially impor-
tant in long-term adaptation of the retina to different
Lumirhodopsin
light intensities.
(µsec)
Night Blindness. Night blindness occurs in any person
Metarhodopsin I
with severe vitamin A deficiency. The simple reason for
(minutes)
(msec)
this is that without vitamin A, the amounts of retinal and
rhodopsin that can be formed are severely depressed.
This condition is called night blindness because the
Metarhodopsin II
(sec)
amount of light available at night is too little to permit
adequate vision in vitamin A-deficient persons.
For night blindness to occur, a person usually must
Scotopsin
remain on a vitamin A-deficient diet for months,
because large quantities of vitamin A are normally
Isomerase
11-cis retinal
all-trans retinal
stored in the liver and can be made available to the eyes.
Once night blindness develops, it can sometimes be
Isomerase
reversed in less than 1 hour by intravenous injection of
11-cis retinol
all-trans retinol
vitamin A.
(Vitamin A)
Excitation of the Rod When Rhodopsin
Figure 50-5
Is Activated by Light
Rhodopsin-retinal visual cycle in the rod, showing decomposition
of rhodopsin during exposure to light and subsequent slow re-
The Rod Receptor Potential Is Hyperpolarizing, Not Depolariz-
formation of rhodopsin by the chemical processes.
ing. When the rod is exposed to light, the resulting
630
Unit X The Nervous System: B. The Special Senses
receptor potential is different from the receptor poten-
there is reduced electronegativity inside the membrane
tials in almost all other sensory receptors. That is, exci-
of the rod, measuring about -40 millivolts rather than
tation of the rod causes increased negativity of the
the usual -70 to -80 millivolts found in most sensory
intrarod membrane potential, which is a state of hyper-
receptors.
polarization, meaning that there is more negativity
Then, when the rhodopsin in the outer segment
than normal inside the rod membrane. This is exactly
of the rod is exposed to light, the rhodopsin begins
opposite to the decreased negativity
(the process
to decompose, and this decreases the outer segment
of “depolarization”) that occurs in almost all other
membrane conductance of sodium to the interior of
sensory receptors.
the rod, even though sodium ions continue to be
But how does activation of rhodopsin cause hyper-
pumped outward through the membrane of the inner
polarization? The answer is that when rhodopsin
segment. Thus, more sodium ions now leave the rod
decomposes, it decreases the rod membrane conduc-
than leak back in. Because they are positive ions, their
tance for sodium ions in the outer segment of the rod.
loss from inside the rod creates increased negativity
This causes hyperpolarization of the entire rod mem-
inside the membrane, and the greater the amount of
brane in the following way.
light energy striking the rod, the greater the elec-
Figure 50-6 shows movement of sodium ions in a
tronegativity becomes—that is, the greater is the
complete electrical circuit through the inner and outer
degree of hyperpolarization. At maximum light inten-
segments of the rod. The inner segment continually
sity, the membrane potential approaches -70 to -80
pumps sodium from inside the rod to the outside,
millivolts, which is near the equilibrium potential for
thereby creating a negative potential on the inside of
potassium ions across the membrane.
the entire cell. However, the outer segment of the rod,
where the photoreceptor discs are located, is entirely
Duration of the Receptor Potential, and Logarithmic
different; here, the rod membrane, in the dark state,
Relation of the Receptor Potential to Light Intensity.
is very leaky to sodium ions. Therefore, positively
When a sudden pulse of light strikes the retina, the
charged sodium ions continually leak back to the
transient hyperpolarization that occurs in the rods—
inside of the rod and thereby neutralize much of the
that is, the receptor potential that occurs—reaches a
negativity on the inside of the entire cell. Thus, under
peak in about 0.3 second and lasts for more than a
normal dark conditions, when the rod is not excited,
second. In cones, the change occurs four times as fast
as in the rods. A visual image impinged on the rods
of the retina for only one millionth of a second can
sometimes cause the sensation of seeing the image for
longer than a second.
Another characteristic of the receptor potential is
Leakage current
that it is approximately proportional to the logarithm
decreased by
decomposing
of the light intensity. This is exceedingly important,
Na+
rhodopsin
because it allows the eye to discriminate light intensi-
-
-
ties through a range many thousand times as great as
would be possible otherwise.
-
Sodium
-
current
Mechanism by Which Rhodopsin Decomposition
Decreases Membrane Sodium Conductance—The
-
-
Excitation
“Cascade.” Under optimal conditions,
-
a single photon of light, the smallest possible quantal
-
-
unit of light energy, can cause a measurable receptor
-
Na+
potential in a rod of about 1 millivolt. Only 30 photons
Sodium
of light will cause half saturation of the rod. How
pump
can such a small amount of light cause such great
-
excitation? The answer is that the photoreceptors
-
have an extremely sensitive chemical cascade that
amplifies the stimulatory effects about a millionfold,
-
as follows:
-
1. The photon activates an electron in the 11-cis
retinal portion of the rhodopsin; this leads to the
-
formation of metarhodopsin II, which is the active
form of rhodopsin, as already discussed and
- -
-
-
shown in Figure 50-5.
2. The activated rhodopsin functions as an enzyme to
Figure 50-6
activate many molecules of transducin, a protein
present in an inactive form in the membranes of
Theoretical basis for generation of a “hyperpolarization receptor
the discs and cell membrane of the rod.
potential” caused by rhodopsin decomposition, which decreases
the flow of positively charged sodium ions into the outer segment
3. The activated transducin activates many more
of the rod.
molecules of phosphodiesterase.
Chapter 50
The Eye: II. Receptor and Neural Function of the Retina
631
4. Activated phosphodiesterase is another enzyme;
it immediately hydrolyzes many molecules of
Blue
Green Red
cyclic guanosine monophosphate (cGMP), thus
cone
Rods
cone
cone
destroying it. Before being destroyed, the cGMP
100
had been bound with the sodium channel protein
of the rod’s outer membrane in a way that
75
“splints” it in the open state. But in light, when
phosphodiesterase hydrolyzes the cGMP, this
50
removes the splinting and allows the sodium
channels to close. Several hundred channels
25
close for each originally activated molecule of
rhodopsin. Because the sodium flux through
0
each of these channels has been extremely rapid,
400
500
600
700
flow of more than a million sodium ions is
Wavelength (nanometers)
blocked by the channel closure before the
Violet
Blue
Green
Yellow
Orange
Red
channel opens again. This diminution of sodium
ion flow is what excites the rod, as already
discussed.
5. Within about a second, another enzyme,
Figure 50-7
rhodopsin kinase, which is always present in
the rod, inactivates the activated rhodopsin
Light absorption by the pigment of the rods and by the pigments
of the three color-receptive cones of the human retina. (Drawn
(the metarhodopsin II), and the entire cascade
from curves recorded by Marks WB, Dobelle WH, MacNichol EF
reverses back to the normal state with open
Jr: Visual pigments of single primate cones. Science 143:1181,
sodium channels.
1964, and by Brown PK, Wald G: Visual pigments in single rods
Thus, the rods have developed an important chemi-
and cones of the human retina: direct measurements reveal mech-
anisms of human night and color vision. Science 144:45, 1964.
cal cascade that amplifies the effect of a single photon
" 1964 by the American Association for the Advancement of
of light to cause movement of millions of sodium ions.
Science.)
This explains the extreme sensitivity of the rods under
dark conditions.
The cones are about 30 to 300 times less sensitive
Automatic Regulation of
than the rods, but even this allows color vision at
any intensity of light greater than extremely dim
Retinal Sensitivity—Light and
twilight.
Dark Adaptation
Photochemistry of Color Vision by the Cones
Light and Dark Adaptation. If a person has been in bright
It was pointed out at the outset of this discussion that
light for hours, large portions of the photochemicals in
the photochemicals in the cones have almost exactly
both the rods and the cones will have been reduced to
the same chemical composition as that of rhodopsin
retinal and opsins. Furthermore, much of the retinal of
in the rods. The only difference is that the protein por-
both the rods and the cones will have been converted
tions, or the opsins—called photopsins in the cones—
into vitamin A. Because of these two effects, the con-
are slightly different from the scotopsin of the rods.
centrations of the photosensitive chemicals remaining
The retinal portion of all the visual pigments is exactly
in the rods and cones are considerably reduced, and
the same in the cones as in the rods. The color-
the sensitivity of the eye to light is correspondingly
sensitive pigments of the cones, therefore, are combi-
reduced. This is called light adaptation.
nations of retinal and photopsins.
Conversely, if a person remains in darkness for a
In the discussion of color vision later in the chapter,
long time, the retinal and opsins in the rods and cones
it will become evident that only one of three types of
are converted back into the light-sensitive pigments.
color pigments is present in each of the different cones,
Furthermore, vitamin A is converted back into retinal
thus making the cones selectively sensitive to different
to give still more light-sensitive pigments, the final
colors: blue, green, or red. These color pigments
limit being determined by the amount of opsins in the
are called, respectively, blue-sensitive pigment, green-
rods and cones to combine with the retinal. This is
sensitive pigment, and red-sensitive pigment. The
called dark adaptation.
absorption characteristics of the pigments in the three
Figure 50-8 shows the course of dark adaptation
types of cones show peak absorbencies at light wave-
when a person is exposed to total darkness after
lengths of 445, 535, and 570 nanometers, respectively.
having been exposed to bright light for several hours.
These are also the wavelengths for peak light sensitiv-
Note that the sensitivity of the retina is very low on
ity for each type of cone, which begins to explain how
first entering the darkness, but within 1 minute, the
the retina differentiates the colors. The approximate
sensitivity has already increased 10-fold—that is, the
absorption curves for these three pigments are shown
retina can respond to light of one tenth the previously
in Figure 50-7. Also shown is the absorption curve
required intensity. At the end of 20 minutes, the sensi-
for the rhodopsin of the rods, with a peak at 505
tivity has increased about 6000-fold, and at the end of
nanometers.
40 minutes, about 25,000-fold.
632
Unit X The Nervous System: B. The Special Senses
contrast to the many minutes to hours required for full
adaptation by the photochemicals.
100,000
Value of Light and Dark Adaptation in Vision. Between the
limits of maximal dark adaptation and maximal light
10,000
adaptation, the eye can change its sensitivity to light
as much as 500,000 to 1 million times, the sensitivity
automatically adjusting to changes in illumination.
1000
Because registration of images by the retina
Rod adaptation
requires detection of both dark and light spots in the
image, it is essential that the sensitivity of the retina
100
always be adjusted so that the receptors respond to the
lighter areas but not to the darker areas. An example
10
of maladjustment of retinal adaptation occurs when a
Cone adaptation
person leaves a movie theater and enters the bright
sunlight. Then, even the dark spots in the images seem
1
exceedingly bright, and as a consequence, the entire
0
10
20
30
40
50
visual image is bleached, having little contrast among
Minutes in dark
its different parts. This is poor vision, and it remains
poor until the retina has adapted sufficiently so that
the darker areas of the image no longer stimulate the
Figure 50-8
receptors excessively.
Conversely, when a person first enters darkness, the
Dark adaptation, demonstrating the relation of cone adaptation to
sensitivity of the retina is usually so slight that even
rod adaptation.
the light spots in the image cannot excite the retina.
After dark adaptation, the light spots begin to regis-
ter. As an example of the extremes of light and dark
The resulting curve of Figure 50-8 is called the dark
adaptation, the intensity of sunlight is about 10 billion
adaptation curve. Note, however, the inflection in the
times that of starlight, yet the eye can function both in
curve. The early portion of the curve is caused by adap-
bright sunlight after light adaptation and in starlight
tation of the cones, because all the chemical events of
after dark adaptation.
vision, including adaptation, occur about four times as
rapidly in cones as in rods. However, the cones do not
achieve anywhere near the same degree of sensitivity
Color Vision
change in darkness as the rods do. Therefore, despite
rapid adaptation, the cones cease adapting after only
From the preceding sections, we have learned that dif-
a few minutes, while the slowly adapting rods continue
ferent cones are sensitive to different colors of light.
to adapt for many minutes and even hours, their sen-
This section is a discussion of the mechanisms by
sitivity increasing tremendously. In addition, still more
which the retina detects the different gradations of
sensitivity of the rods is caused by neuronal signal con-
color in the visual spectrum.
vergence of 100 or more rods onto a single ganglion
cell in the retina; these rods summate to increase their
sensitivity, as discussed later in the chapter.
Tricolor Mechanism of Color
Detection
Other Mechanisms of Light and Dark Adaptation. In addition
to adaptation caused by changes in concentrations of
All theories of color vision are based on the well-
rhodopsin or color photochemicals, the eye has two
known observation that the human eye can detect
other mechanisms for light and dark adaptation. The
almost all gradations of colors when only red, green,
first of these is a change in pupillary size, as discussed
and blue monochromatic lights are appropriately
in Chapter 49. This can cause adaptation of approxi-
mately 30-fold within a fraction of a second, because of
mixed in different combinations.
changes in the amount of light allowed through the
pupillary opening.
Spectral Sensitivities of the Three Types of Cones. On the
The other mechanism is neural adaptation, involving
basis of color vision tests, the spectral sensitivities of
the neurons in the successive stages of the visual chain
the three types of cones in humans have proved to be
in the retina itself and in the brain. That is, when light
essentially the same as the light absorption curves for
intensity first increases, the signals transmitted by the
the three types of pigment found in the cones. These
bipolar cells, horizontal cells, amacrine cells, and gan-
curves are shown in Figure 50-7 and slightly differ-
glion cells are all intense. However, most of these signals
ently in Figure 50-9. They can explain most of the phe-
decrease rapidly at different stages of transmission in
nomena of color vision.
the neural circuit. Although the degree of adaptation is
only a fewfold rather than the many thousandfold that
occurs during adaptation of the photochemical system,
Interpretation of Color in the Nervous System. Refer-
neural adaptation occurs in a fraction of a second, in
ring to Figure
50-9, one can see that an orange
Chapter 50
The Eye: II. Receptor and Neural Function of the Retina
633
from green and is therefore said to have red-green color
blindness.
Blue
Green Red
A person with loss of red cones is called a protanope;
cone
cone cone
100
97
99
the overall visual spectrum is noticeably shortened at
the long wavelength end because of a lack of the red
83
83
cones. A color-blind person who lacks green cones is
75
called a deuteranope; this person has a perfectly normal
67
visual spectral width because red cones are available to
50
42
detect the long wavelength red color.
36
Red-green color blindness is a genetic disorder that
31
occurs almost exclusively in males. That is, genes in the
25
female X chromosome code for the respective cones.
Yet color blindness almost never occurs in females
0
0
because at least one of the two X chromosomes almost
400
500
600
700
always has a normal gene for each type of cone. Because
Wavelength (nanometers)
the male has only one X chromosome, a missing gene
Violet
Blue
Green
Yellow
Orange
Red
can lead to color blindness.
Because the X chromosome in the male is always
inherited from the mother, never from the father, color
blindness is passed from mother to son, and the mother
Figure 50-9
is said to be a color blindness carrier; this is true in about
8 per cent of all women.
Demonstration of the degree of stimulation of the different color-
sensitive cones by monochromatic lights of four colors: blue,
Blue Weakness. Only rarely are blue cones missing,
green, yellow, and orange.
although sometimes they are underrepresented, which
is a genetically inherited state giving rise to the phe-
nomenon called blue weakness.
monochromatic light with a wavelength of
580
nanometers stimulates the red cones to a stimulus
Color Test Charts. A rapid method for determining color
blindness is based on the use of spot charts such as those
value of about 99 (99 per cent of the peak stimulation
shown in Figure 50-10. These charts are arranged with
at optimum wavelength); it stimulates the green cones
a confusion of spots of several different colors. In the
to a stimulus value of about 42, but the blue cones not
top chart, the person with normal color vision reads
at all. Thus, the ratios of stimulation of the three types
“74,” whereas the red-green color-blind person reads
of cones in this instance are
99:42:0. The nervous
“21.” In the bottom chart, the person with normal color
system interprets this set of ratios as the sensation of
vision reads “42,” whereas the red-blind person reads
orange. Conversely, a monochromatic blue light with a
“2,” and the green-blind person reads “4.”
wavelength of
450 nanometers stimulates the red
If one studies these charts while at the same time
cones to a stimulus value of 0, the green cones to a
observing the spectral sensitivity curves of the different
value of 0, and the blue cones to a value of 97. This set
cones depicted in Figure 50-9, it can be readily under-
stood how excessive emphasis can be placed on spots of
of ratios—0:0:97—is interpreted by the nervous
certain colors by color-blind people.
system as blue. Likewise, ratios of 83:83:0 are inter-
preted as yellow, and 31:67:36 as green.
Neural Function of the Retina
Perception of White Light. About equal stimulation of
all the red, green, and blue cones gives one the sensa-
Neural Circuitry of the Retina
tion of seeing white. Yet there is no single wavelength
of light corresponding to white; instead, white is a com-
Figure
50-1 shows the tremendous complexity of
bination of all the wavelengths of the spectrum. Fur-
neural organization in the retina. To simplify this,
thermore, the perception of white can be achieved by
Figure 50-11 presents the essentials of the retina’s
stimulating the retina with a proper combination of
neural connections, showing at the left the circuit in
only three chosen colors that stimulate the respective
the peripheral retina and at the right the circuit in the
types of cones about equally.
foveal retina. The different neuronal cell types are as
follows:
Color Blindness
1. The photoreceptors themselves—the rods and
cones—which transmit signals to the outer
Red-Green Color Blindness. When a single group of color-
plexiform layer, where they synapse with bipolar
receptive cones is missing from the eye, the person is
cells and horizontal cells
unable to distinguish some colors from others. For
2. The horizontal cells, which transmit signals
instance, one can see in Figure 50-9 that green, yellow,
horizontally in the outer plexiform layer from the
orange, and red colors, which are the colors between the
rods and cones to bipolar cells
wavelengths of 525 and 675 nanometers, are normally
distinguished from one another by the red and green
3. The bipolar cells, which transmit signals vertically
cones. If either of these two cones is missing, the person
from the rods, cones, and horizontal cells to the
cannot use this mechanism for distinguishing these four
inner plexiform layer, where they synapse with
colors; the person is especially unable to distinguish red
ganglion cells and amacrine cells
634
Unit X The Nervous System: B. The Special Senses
Figure 50-10
Two Ishihara charts. Upper: In this chart, the normal person
reads “74,” but the red-green color-blind person reads “21.”
Lower: In this chart, the red-blind person (protanope) reads
“2,” but the green-blind person (deuteranope) reads “4.” The
normal person reads “42.” (Reproduced from Ishihara’s Tests
for Colour Blindness. Tokyo: Kanehara & Co., but tests for
color blindness cannot be conducted with this material. For
accurate testing, the original plates should be used.)
4. The amacrine cells, which transmit signals in two
A sixth type of neuronal cell in the retina, not very
directions, either directly from bipolar cells to
prominent and not shown in the figure, is the inter-
ganglion cells or horizontally within the inner
plexiform cell. This cell transmits signals in the retro-
plexiform layer from axons of the bipolar cells to
grade direction from the inner plexiform layer to the
dendrites of the ganglion cells or to other
outer plexiform layer. These signals are inhibitory and
amacrine cells
are believed to control lateral spread of visual signals
5. The ganglion cells, which transmit output signals
by the horizontal cells in the outer plexiform layer.
from the retina through the optic nerve into the
Their role may be to help control the degree of con-
brain
trast in the visual image.
Chapter 50
The Eye: II. Receptor and Neural Function of the Retina
635
Neurotransmitters Released by Retinal Neurons. Not all the
neurotransmitter chemical substances used for synap-
Pigment layer
tic transmission in the retina have been entirely delin-
eated. However, both the rods and the cones release
Cones
glutamate at their synapses with the bipolar cells.
Histological and pharmacological studies have shown
Rods
there to be many types of amacrine cells secreting at
least eight types of transmitter substances, including
Rod nuclei
gamma-aminobutyric acid, glycine, dopamine, acetyl-
choline, and indolamine, all of which normally function
as inhibitory transmitters. The transmitters of the
bipolar, horizontal, and interplexiform cells are
Horizontal
Bipolar
unclear, but at least some of the horizontal cells
cells
cells
release inhibitory transmitters.
Transmission of Most Signals Occurs in the Retinal Neurons by
Amacrine
cells
Electrotonic Conduction, Not by Action Potentials. The only
retinal neurons that always transmit visual signals by
Ganglion
means of action potentials are the ganglion cells, and
cells
they send their signals all the way to the brain through
the optic nerve. Occasionally, action potentials have
also been recorded in amacrine cells, although the
Figure 50-11
importance of these action potentials is questionable.
Otherwise, all the retinal neurons conduct their visual
Neural organization of the retina: peripheral area to the left, foveal
area to the right.
signals by electrotonic conduction, which can be
explained as follows.
Electrotonic conduction means direct flow of elec-
The Visual Pathway from the Cones to the Ganglion Cells Func-
tric current, not action potentials, in the neuronal cyto-
tions Differently from the Rod Pathway. As is true for many
plasm and nerve axons from the point of excitation all
of our other sensory systems, the retina has both an
the way to the output synapses. Even in the rods and
old type of vision based on rod vision and a new type
cones, conduction from their outer segments, where
of vision based on cone vision. The neurons and nerve
the visual signals are generated, to the synaptic bodies
fibers that conduct the visual signals for cone vision
is by electrotonic conduction. That is, when hyperpo-
are considerably larger than those that conduct the
larization occurs in response to light in the outer
visual signals for rod vision, and the signals are con-
segment of a rod or a cone, almost the same degree of
ducted to the brain two to five times as rapidly. Also,
hyperpolarization is conducted by direct electric
the circuitry for the two systems is slightly different, as
current flow in the cytoplasm all the way to the synap-
follows.
tic body, and no action potential is required. Then,
To the right in Figure 50-11 is the visual pathway
when the transmitter from a rod or cone stimulates a
from the foveal portion of the retina, representing the
bipolar cell or horizontal cell, once again the signal is
new, fast cone system. This shows three neurons in
transmitted from the input to the output by direct elec-
the direct pathway: (1) cones, (2) bipolar cells, and (3)
tric current flow, not by action potentials.
ganglion cells. In addition, horizontal cells transmit
The importance of electrotonic conduction is that it
inhibitory signals laterally in the outer plexiform layer,
allows graded conduction of signal strength. Thus, for
and amacrine cells transmit signals laterally in the
the rods and cones, the strength of the hyperpolariz-
inner plexiform layer.
ing output signal is directly related to the intensity of
To the left in Figure 50-11 are the neural connec-
illumination; the signal is not all or none, as would be
tions for the peripheral retina, where both rods and
the case for each action potential.
cones are present. Three bipolar cells are shown; the
middle of these connects only to rods, representing the
Lateral Inhibition to Enhance Visual Contrast—
type of visual system present in many lower animals.
Function of the Horizontal Cells
The output from the bipolar cell passes only to
The horizontal cells, shown in Figure 50-11, connect
amacrine cells, which relay the signals to the ganglion
laterally between the synaptic bodies of the rods and
cells. Thus, for pure rod vision, there are four neurons
cones, as well as connecting with the dendrites of the
in the direct visual pathway: (1) rods, (2) bipolar
bipolar cells. The outputs of the horizontal cells are
cells,
(3) amacrine cells, and
(4) ganglion cells.
always inhibitory. Therefore, this lateral connection
Also, horizontal and amacrine cells provide lateral
provides the same phenomenon of lateral inhibition
connectivity.
that is important in all other sensory systems—that is,
The other two bipolar cells shown in the peripheral
helping to ensure transmission of visual patterns with
retinal circuitry of Figure 50-11 connect with both rods
proper visual contrast. This phenomenon is demon-
and cones; the outputs of these bipolar cells pass both
strated in Figure 50-12, which shows a minute spot of
directly to ganglion cells and by way of amacrine cells.
light focused on the retina. The visual pathway from
636
Unit X The Nervous System: B. The Special Senses
positive signals and the other half to transmit negative
signals. We shall see later that both positive and nega-
tive signals are used in transmitting visual information
to the brain.
Another important aspect of this reciprocal relation
Light beam
between depolarizing and hyperpolarizing bipolar
cells is that it provides a second mechanism for
lateral inhibition, in addition to the horizontal cell
mechanism. Because depolarizing and hyperpolarizing
Excited area
bipolar cells lie immediately against each other, this
provides a mechanism for separating contrast borders
in the visual image, even when the border lies exactly
Neither excited
between two adjacent photoreceptors. In contrast, the
nor inhibited
horizontal cell mechanism for lateral inhibition oper-
ates over a much greater distance.
Inhibited area
Amacrine Cells and Their Functions
About 30 types of amacrine cells have been identified
by morphological or histochemical means. The func-
tions of about half a dozen types of amacrine cells have
Figure 50-12
been characterized, and all of them are different. One
type of amacrine cell is part of the direct pathway
Excitation and inhibition of a retinal area caused by a small beam
of light, demonstrating the principle of lateral inhibition.
for rod vision—that is, from rod to bipolar cells to
amacrine cells to ganglion cells.
Another type of amacrine cell responds strongly at
the centralmost area where the light strikes is excited,
the onset of a continuing visual signal, but the response
whereas an area to the side is inhibited. In other words,
dies rapidly.
instead of the excitatory signal spreading widely in the
Other amacrine cells respond strongly at the offset
retina because of spreading dendritic and axonal trees
of visual signals, but again, the response dies quickly.
in the plexiform layers, transmission through the hor-
Still other amacrine cells respond when a light is
izontal cells puts a stop to this by providing lateral
turned either on or off, signaling simply a change in
inhibition in the surrounding areas. This is essential to
illumination, irrespective of direction.
allow high visual accuracy in transmitting contrast
Still another type of amacrine cell responds to
borders in the visual image.
movement of a spot across the retina in a specific
Some of the amacrine cells probably provide addi-
direction; therefore, these amacrine cells are said to be
tional lateral inhibition and further enhancement of
directional sensitive.
visual contrast in the inner plexiform layer of the
In a sense, then, many or most amacrine cells are
retina as well.
interneurons that help analyze visual signals before
they ever leave the retina.
Excitation of Some Bipolar Cells and
Inhibition of Others—The Depolarizing
and Hyperpolarizing Bipolar Cells
Ganglion Cells and Optic Nerve Fibers
Two types of bipolar cells provide opposing excitatory
and inhibitory signals in the visual pathway: (1) the
Each retina contains about 100 million rods and 3
depolarizing bipolar cell and (2) the hyperpolarizing
million cones; yet the number of ganglion cells is only
bipolar cell. That is, some bipolar cells depolarize
about 1.6 million. Thus, an average of 60 rods and 2
when the rods and cones are excited, and others
cones converge on each ganglion cell and the optic
hyperpolarize.
nerve fiber leading from the ganglion cell to the brain.
There are two possible explanations for this differ-
However, major differences exist between the
ence. One explanation is that the two bipolar cells are
peripheral retina and the central retina. As one
of entirely different types—one responding by depo-
approaches the fovea, fewer rods and cones converge
larizing in response to the glutamate neurotransmitter
on each optic fiber, and the rods and cones also
released by the rods and cones, and the other respond-
become more slender. These effects progressively
ing by hyperpolarizing. The other possibility is that one
increase the acuity of vision in the central retina. In
of the bipolar cells receives direct excitation from the
the center, in the central fovea, there are only slender
rods and cones, whereas the other receives its signal
cones—about 35,000 of them—and no rods. Also, the
indirectly through a horizontal cell. Because the hori-
number of optic nerve fibers leading from this part of
zontal cell is an inhibitory cell, this would reverse the
the retina is almost exactly equal to the number of
polarity of the electrical response.
cones, as shown to the right in Figure 50-11. This
Regardless of the mechanism for the two types of
explains the high degree of visual acuity in the central
bipolar responses, the importance of this phenomenon
retina in comparison with the much poorer acuity
is that it allows half the bipolar cells to transmit
peripherally.
Chapter 50
The Eye: II. Receptor and Neural Function of the Retina
637
Another difference between the peripheral and
of a second. These ganglion cells presumably apprise
central portions of the retina is the much greater sen-
the central nervous system almost instantaneously
sitivity of the peripheral retina to weak light. This
when a new visual event occurs anywhere in the visual
results partly from the fact that rods are 30 to 300
field, but without specifying with great accuracy the
times more sensitive to light than cones are, but it is
location of the event, other than to give appropriate
further magnified by the fact that as many as 200 rods
clues that make the eyes move toward the exciting
converge on a single optic nerve fiber in the more
vision.
peripheral portions of the retina, so that signals from
the rods summate to give even more intense stimula-
tion of the peripheral ganglion cells and their optic
Excitation of the Ganglion Cells
nerve fibers.
Spontaneous, Continuous Action Potentials in the Ganglion
Three Types of Retinal Ganglion Cells and
Cells. It is from the ganglion cells that the long fibers
Their Respective Fields
of the optic nerve lead into the brain. Because of the
There are three distinct types of ganglion cells, desig-
distance involved, the electrotonic method of conduc-
nated W, X, and Y cells. Each of these serves a differ-
tion employed in the rods, cones, and bipolar cells
ent function.
within the retina is no longer appropriate; therefore,
ganglion cells transmit their signals by means of repet-
Transmission of Rod Vision by the W Cells. The W cells, con-
itive action potentials instead. Furthermore, even
stituting about 40 per cent of all the ganglion cells, are
when unstimulated, they still transmit continuous im-
small, having a diameter less than 10 micrometers, and
pulses at rates varying between 5 and 40 per second.
they transmit signals in their optic nerve fibers at the
The visual signals, in turn, are superimposed onto this
slow velocity of only 8 m/sec. These ganglion cells
background ganglion cell firing.
receive most of their excitation from rods, transmitted
by way of small bipolar cells and amacrine cells. They
Transmission of Changes in Light Intensity—The On-Off
have broad fields in the peripheral retina because the
Response. As noted previously, many ganglion cells are
dendrites of the ganglion cells spread widely in the
specifically excited by changes in light intensity. This is
inner plexiform layer, receiving signals from broad
demonstrated by the records of nerve impulses in
areas.
Figure 50-13. The upper panel shows rapid impulses
On the basis of histology as well as physiologic
for a fraction of a second when a light is first turned
experiments, the W cells seem to be especially sensi-
on, but decreasing rapidity in the next fraction of a
tive for detecting directional movement in the field of
second. The lower tracing is from a ganglion cell
vision, and they are probably important for much of
located lateral to the spot of light; this cell is markedly
our crude rod vision under dark conditions.
inhibited when the light is turned on because of lateral
inhibition. Then, when the light is turned off, opposite
Transmission of the Visual Image and Color by the X Cells. The
effects occur. Thus, these records are called “on-off ”
most numerous of the ganglion cells are the X cells,
and “off-on” responses. The opposite directions of
representing 55 per cent of the total. They are of
these responses to light are caused, respectively, by the
medium diameter, between 10 and 15 micrometers,
depolarizing and hyperpolarizing bipolar cells, and the
and transmit signals in their optic nerve fibers at about
transient nature of the responses is probably at least
14 m/sec.
partly generated by the amacrine cells, many of which
The X cells have small fields because their dendrites
have similar transient responses themselves.
do not spread widely in the retina. Because of this,
This capability of the eyes to detect change in light
their signals represent discrete retinal locations. There-
intensity is strongly developed in both the peripheral
fore, it is mainly through the X cells that the fine
details of the visual image are transmitted. Also,
because every X cell receives input from at least one
on
off
cone, X cell transmission is probably responsible for
all color vision.
1
Excitation
Function of the Y Cells to Transmit Instantaneous Changes in
the Visual Image. The Y cells are the largest of all, up to
35 micrometers in diameter, and they transmit their
2
signals to the brain at 50 m/sec or faster. They are the
Lateral inhibition
least numerous of all the ganglion cells, representing
Figure 50-13
only 5 per cent of the total. Also, they have broad den-
dritic fields, so that signals are picked up by these cells
Responses of a ganglion cell to light in (1) an area excited by a
from widespread retinal areas.
spot of light and (2) an area adjacent to the excited spot; the gan-
The Y ganglion cells respond, like many of the
glion cell in this area is inhibited by the mechanism of lateral
inhibition.
(Modified from Granit R: Receptors and Sensory
amacrine cells, to rapid changes in the visual image—
Perception: A Discussion of Aims, Means, and Results of Electro-
either rapid movement or rapid change in light inten-
physiological Research into the Process of Reception. New
sity—sending bursts of signals for only small fractions
Haven, Conn: Yale University Press, 1955.)
638
Unit X The Nervous System: B. The Special Senses
retina and the central retina. For instance, a minute
gnat flying across the field of vision is instantaneously
detected. Conversely, the same gnat sitting quietly
remains below the threshold of visual detection.
Transmission of Signals Depicting Contrasts
in the Visual Scene—The Role of
Lateral Inhibition
Many ganglion cells respond mainly to contrast
borders in the scene. Because this seems to be the
Excitation
major means by which the pattern of a scene is trans-
mitted to the brain, let us explain how this process
occurs.
When flat light is applied to the entire retina—that
is, when all the photoreceptors are stimulated equally
by the incident light—the contrast type of ganglion cell
H
is neither stimulated nor inhibited. The reason for this
H
Inhibition
is that signals transmitted directly from the photore-
B
ceptors through depolarizing bipolar cells are excita-
tory, while the signals transmitted laterally through
hyperpolarizing bipolar cells as well as through
horizontal cells are mainly inhibitory. Thus, the direct
excitatory signal through one pathway is likely to be
neutralized by inhibitory signals through lateral path-
ways. One circuit for this is demonstrated in Figure
50-14, which shows at the top three photoreceptors.
The central receptor excites a depolarizing bipolar
G
cell. The two receptors on each side are connected
to the same bipolar cell through inhibitory horizontal
cells that neutralize the direct excitatory signal if
all three receptors are stimulated simultaneously by
Figure 50-14
light.
Now, let us examine what happens when a contrast
Typical arrangement of rods, horizontal cells (H), a bipolar cell (B),
and a ganglion cell (G) in the retina, showing excitation at the
border occurs in the visual scene. Referring again to
synapses between the rods and the bipolar cell and horizontal
Figure 50-14, assume that the central photoreceptor is
cells, but inhibition from the horizontal cells to the bipolar cell.
stimulated by a bright spot of light while one of the
two lateral receptors is in the dark. The bright spot of
light excites the direct pathway through the bipolar
cell. The fact that one of the lateral photoreceptors is
in the dark causes one of the horizontal cells to remain
unstimulated. Therefore, this cell does not inhibit
type. For instance, this frequently occurs for the red
the bipolar cell, and this allows extra excitation of the
and green cones, with red causing excitation and green
bipolar cell. Thus, where visual contrasts occur, the
causing inhibition, or vice versa.
signals through the direct and lateral pathways accen-
The same type of reciprocal effect occurs between
tuate one another.
blue cones on the one hand and a combination
In summary, the mechanism of lateral inhibition
of red and green cones (both of which are excited
functions in the eye in the same way that it functions
by yellow) on the other hand, giving a reciprocal
in most other sensory systems—to provide contrast
excitation-inhibition relation between the blue and
detection and enhancement.
yellow colors.
The mechanism of this opposing effect of colors is
Transmission of Color Signals by the
the following: One color type of cone excites the gan-
Ganglion Cells
glion cell by the direct excitatory route through a
A single ganglion cell may be stimulated by several
depolarizing bipolar cell, whereas the other color type
cones or by only a few. When all three types of cones—
inhibits the ganglion cell by the indirect inhibitory
the red, blue, and green types—stimulate the same
route through a hyperpolarizing bipolar cell.
ganglion cell, the signal transmitted through the gan-
The importance of these color-contrast mechanisms
glion cell is the same for any color of the spectrum.
is that they represent a means by which the retina itself
Therefore, the signal from the ganglion cell plays no
begins to differentiate colors. Thus, each color-contrast
role in the detection of different colors. Instead, it is a
type of ganglion cell is excited by one color but inhib-
“white” signal.
ited by the “opponent” color. Therefore, color analysis
Conversely, some of the ganglion cells are excited by
begins in the retina and is not entirely a function of
only one color type of cone but inhibited by a second
the brain.
Chapter 50
The Eye: II. Receptor and Neural Function of the Retina
639
References
Gegenfurtner KR, Kiper DC: Color vision. Annu Rev
Neurosci 26:181, 2003.
Hardie RC: Phototransduction: shedding light on transloca-
Arshavsky V: Like night and day: rods and cones have dif-
tion. Curr Biol 13:R775, 2003.
ferent pigment regeneration pathways. Neuron 36:1, 2002.
Hendee WA, Wells PNT: The Perception of Visual Informa-
Backharis W, Kliegl R, Werner JS: Color Vision. Berlin:
tion. New York: Springer, 1997.
Walter de Gruyter, 1998.
Kolb H, Nelson R, Ahnelt P, Cuenca N: Cellular organiza-
Berger JW, Fine SL, Maguire MG: Age-Related Macular
tion of the vertebrate retina. Prog Brain Res 131:3, 2001.
Degeneration. St Louis: Mosby, 1999.
Masland RH: The fundamental plan of the retina. Nat
Berson DM: Strange vision: ganglion cells as circadian pho-
Neurosci 4:877, 2001.
toreceptors. Trends Neurosci 26:314, 2003.
Michaelides M, Hunt DM, Moore AT: The cone dysfunction
Burr D, Ross J: Vision: the world through picket fences. Curr
syndromes. Br J Ophthalmol 88:291, 2004.
Biol 14:R381, 2004.
Neitz M, Neitz J: Molecular genetics of color vision and color
Calkins DJ: Seeing with S cones. Prog Retin Eye Res 20:255,
vision defects. Arch Ophthalmol 118:691, 2000.
2001.
Schwartz EA: Transport-mediated synapses in the retina.
Dacey DM, Packer OS: Colour coding in the primate retina:
Physiol Rev 82:875, 2002.
diverse cell types and cone-specific circuitry. Curr Opin
Taylor WR, Vaney DI: New directions in retinal research.
Neurobiol 13:421, 2003.
Trends Neurosci 26:379, 2003.
Fain GL, Matthews HR, Cornwall MC, Koutalos Y: Adapta-
Thompson DA, Gal A: Vitamin A metabolism in the retinal
tion in vertebrate photoreceptors. Physiol Rev 81:117,
pigment epithelium: genes, mutations, and diseases. Prog
2001.
Retin Eye Res 22:683, 2003.
Garriga P, Manyosa J: The eye photoreceptor protein
Zarbin MA: Current concepts in the pathogenesis of age-
rhodopsin: structural implications for retinal disease.
related macular degeneration. Arch Ophthalmol 122:598,
FEBS Lett 528:17, 2002.
2004.
Gegenfurtner KR: Cortical mechanisms of colour vision. Nat
Rev Neurosci 4:563, 2003.
C H A P T E R
5
1
The Eye: III. Central
Neurophysiology of Vision
Visual Pathways
Figure 51-1 shows the principal visual pathways
from the two retinas to the visual cortex. The visual
nerve signals leave the retinas through the optic
nerves. At the optic chiasm, the optic nerve fibers
from the nasal halves of the retinas cross to the
opposite sides, where they join the fibers from the
opposite temporal retinas to form the optic tracts. The fibers of each optic tract
then synapse in the dorsal lateral geniculate nucleus of the thalamus, and from
there, geniculocalcarine fibers pass by way of the optic radiation (also called the
geniculocalcarine tract) to the primary visual cortex in the calcarine fissure area
of the medial occipital lobe.
Visual fibers also pass to several older areas of the brain: (1) from the optic
tracts to the suprachiasmatic nucleus of the hypothalamus, presumably to
control circadian rhythms that synchronize various physiologic changes of the
body with night and day; (2) into the pretectal nuclei in the midbrain, to elicit
reflex movements of the eyes to focus on objects of importance and to activate
the pupillary light reflex; (3) into the superior colliculus, to control rapid direc-
tional movements of the two eyes; and (4) into the ventral lateral geniculate
nucleus of the thalamus and surrounding basal regions of the brain, presumably
to help control some of the body’s behavioral functions.
Thus, the visual pathways can be divided roughly into an old system
to the midbrain and base of the forebrain and a new system for direct trans-
mission of visual signals into the visual cortex located in the occipital lobes. In
human beings, the new system is responsible for perception of virtually all
aspects of visual form, colors, and other conscious vision. Conversely, in many
primitive animals, even visual form is detected by the older system, using the
superior colliculus in the same manner that the visual cortex is used in
mammals.
Function of the Dorsal Lateral Geniculate Nucleus
of the Thalamus
The optic nerve fibers of the new visual system terminate in the dorsal lateral
geniculate nucleus, located at the dorsal end of the thalamus and also called
simply the lateral geniculate body, as shown in Figure 51-1. The dorsal lateral
geniculate nucleus serves two principal functions: First, it relays visual infor-
mation from the optic tract to the visual cortex by way of the optic radiation
(also called the geniculocalcarine tract). This relay function is so accurate that
there is exact point-to-point transmission with a high degree of spatial fidelity
all the way from the retina to the visual cortex.
It will be recalled that half the fibers in each optic tract after passing the optic
chiasm are derived from one eye and half from the other eye, representing cor-
responding points on the two retinas. However, the signals from the two eyes
are kept apart in the dorsal lateral geniculate nucleus. This nucleus is composed
of six nuclear layers. Layers II, III, and V (from ventral to dorsal) receive signals
from the lateral half of the ipsilateral retina, whereas layers I, IV, and VI receive
signals from the medial half of the retina of the opposite eye. The respective
retinal areas of the two eyes connect with neurons that are superimposed over
640
Chapter 51
The Eye: III. Central Neurophysiology of Vision
641
Lateral geniculate body
Optic tract
Calcarine fissure
Optic radiation
Optic chiasm
Optic nerve
Secondary
visual areas
Left eye
Primary
visual cortex
Superior
Visual cortex
colliculus
Macula
20∞
60∞
90∞
Figure 51-2
Right eye
Visual cortex in the calcarine fissure area of the medial occipital
cortex.
Figure 51-1
Motor cortex Somatosensory area I
Form,
Principal visual pathways from the eyes to the visual cortex. (Mod-
3-D position,
ified from Polyak SL: The Retina. Chicago: University of Chicago,
motion
1941.)
one another in the paired layers, and similar parallel
transmission is preserved all the way to the visual
cortex.
18
The second major function of the dorsal lateral
geniculate nucleus is to “gate” the transmission of
17
signals to the visual cortex—that is, to control how
much of the signal is allowed to pass to the cortex. The
nucleus receives gating control signals from two major
Visual
Secondary
Primary
sources: (1) corticofugal fibers returning in a backward
detail,
visual
visual
direction from the primary visual cortex to the lateral
color
cortex
cortex
geniculate nucleus, and (2) reticular areas of the mes-
Figure 51-3
encephalon. Both of these are inhibitory and, when
stimulated, can turn off transmission through selected
Transmission of visual signals from the primary visual cortex into
portions of the dorsal lateral geniculate nucleus. It is
secondary visual areas on the lateral surfaces of the occipital and
assumed that both of these gating circuits help high-
parietal cortices. Note that the signals representing form, third-
light the visual information that is allowed to pass.
dimensional position, and motion are transmitted mainly into the
superior portions of the occipital lobe and posterior portions of the
Finally, the dorsal lateral geniculate nucleus is
parietal lobe. By contrast, the signals for visual detail and color
divided in another way: (1) Layers I and II are called
are transmitted mainly into the anteroventral portion of the occip-
magnocellular layers because they contain large
ital lobe and the ventral portion of the posterior temporal lobe.
neurons. These receive their input almost entirely from
the large type Y retinal ganglion cells. This magnocel-
lular system provides a rapidly conducting pathway to
Organization and Function
the visual cortex. However, this system is color blind,
transmitting only black-and-white information. Also,
of the Visual Cortex
its point-to-point transmission is poor because there
are not many Y ganglion cells, and their dendrites
Figures 51-2 and 51-3 show the visual cortex located
spread widely in the retina. (2) Layers III through VI
primarily on the medial aspect of the occipital lobes.
are called parvocellular layers because they contain
Like the cortical representations of the other sensory
large numbers of small to medium-sized neurons.
systems, the visual cortex is divided into a primary
These neurons receive their input almost entirely from
visual cortex and secondary visual areas.
the type X retinal ganglion cells that transmit color
and convey accurate point-to-point spatial informa-
Primary Visual Cortex. The primary visual cortex (see
tion, but at only a moderate velocity of conduction
Figure 51-2) lies in the calcarine fissure area, extend-
rather than at high velocity.
ing forward from the occipital pole on the medial
642
Unit X The Nervous System: B. The Special Senses
aspect of each occipital cortex. This area is the termi-
nus of direct visual signals from the eyes. Signals from
I
the macular area of the retina terminate near the
II
occipital pole, as shown in Figure 51-2, while signals
from the more peripheral retina terminate at or in con-
centric half circles anterior to the pole but still along
III
the calcarine fissure on the medial occipital lobe. The
upper portion of the retina is represented superiorly
(a)
and the lower portion inferiorly.
(b)
Color
Note in the figure the especially large area that rep-
IV
“blobs”
resents the macula. It is to this region that the retinal
(ca)
fovea transmits its signals. The fovea is responsible for
the highest degree of visual acuity. Based on retinal
(cb)
area, the fovea has several hundred times as much rep-
resentation in the primary visual cortex as do the most
V
peripheral portions of the retina.
The primary visual cortex is also called visual area
VI
I. Still another name is the striate cortex because this
area has a grossly striated appearance.
Secondary Visual Areas of the Cortex. The secondary visual
LGN
LGN
(magnocellular)
(parvocellular)
areas, also called visual association areas, lie lateral,
anterior, superior, and inferior to the primary visual
cortex. Most of these areas also fold outward over the
Retinal
Retinal
lateral surfaces of the occipital and parietal cortex, as
"Y"
"X"
shown in Figure 51-3. Secondary signals are transmit-
ganglion
ganglion
ted to these areas for analysis of visual meanings. For
instance, on all sides of the primary visual cortex is
Fast, Black and White
Very Accurate, Color
Brodmann’s area 18 (see Figure 51-3), which is where
virtually all signals from the primary visual cortex pass
Figure 51-4
next. Therefore, Brodmann’s area 18 is called visual
area II, or simply V-2. The other, more distant second-
Six layers of the primary visual cortex. The connections shown on
the left side of the figure originate in the magnocellular layers of
ary visual areas have specific designations—V-3, V-4,
the lateral geniculate nucleus (LGN) and transmit rapidly chang-
and so forth—up to more than a dozen areas. The
ing black-and-white visual signals. The pathways to the right orig-
importance of all these areas is that various aspects
inate in the parvocellular layers (layers III through VI) of the LGN;
of the visual image are progressively dissected and
they transmit signals that depict accurate spatial detail as well as
analyzed.
color. Note especially the areas of the visual cortex called “color
blobs,” which are necessary for detection of color.
Layered Structure of the Primary
Vertical Neuronal Columns in the Visual Cortex. The visual
Visual Cortex
cortex is organized structurally into several million
vertical columns of neuronal cells, each column having
Like almost all other portions of the cerebral cortex,
a diameter of 30 to 50 micrometers. The same vertical
the primary visual cortex has six distinct layers, as
columnar organization is found throughout the cere-
shown in Figure 51-4. Also, as is true for the other
bral cortex for the other senses as well (and also in the
sensory systems, the geniculocalcarine fibers terminate
motor and analytical cortical regions). Each column
mainly in layer IV. But this layer, too, is organized into
represents a functional unit. One can roughly calculate
subdivisions. The rapidly conducted signals from the Y
that each of the visual vertical columns has perhaps
retinal ganglion cells terminate in layer IVca, and
1000 or more neurons.
from there they are relayed vertically both outward
After the optic signals terminate in layer IV, they are
toward the cortical surface and inward toward deeper
further processed as they spread both outward and
levels.
inward along each vertical column unit. This process-
The visual signals from the medium-sized optic
ing is believed to decipher separate bits of visual infor-
nerve fibers, derived from the X ganglion cells in the
mation at successive stations along the pathway. The
retina, also terminate in layer IV, but at points differ-
signals that pass outward to layers I, II, and III even-
ent from the Y signals. They terminate in layers IVa
tually transmit signals for short distances laterally in
and IVcb, the shallowest and deepest portions of layer
the cortex. Conversely, the signals that pass inward to
IV, shown to the right in Figure 51-4. From there, these
layers V and VI excite neurons that transmit signals
signals are transmitted vertically both toward the
much greater distances.
surface of the cortex and to deeper layers. It is these
X ganglion pathways that transmit the accurate point-
“Color Blobs” in the Visual Cortex. Interspersed among the
to-point type of vision as well as color vision.
primary visual columns as well as among the columns
Chapter 51
The Eye: III. Central Neurophysiology of Vision
643
of some of the secondary visual areas are special
into secondary visual areas of the inferior, ventral, and
column-like areas called color blobs. They receive
medial regions of the occipital and temporal cortex,
lateral signals from adjacent visual columns and are
show the principal pathway for analysis of visual
activated specifically by color signals. Therefore, it is
detail. Separate portions of this pathway specifically
presumed that these blobs are the primary areas for
dissect out color as well. Therefore, this pathway is
deciphering color.
concerned with such visual feats as recognizing letters,
reading, determining the texture of surfaces, deter-
Interaction of Visual Signals from the Two Separate Eyes.
mining detailed colors of objects, and deciphering
Recall that the visual signals from the two separate
from all this information what the object is and what
eyes are relayed through separate neuronal layers in
it means.
the lateral geniculate nucleus. These signals still
remain separated from each other when they arrive in
layer IV of the primary visual cortex. In fact, layer IV
Neuronal Patterns of
is interlaced with stripes of neuronal columns, each
Stimulation During Analysis
stripe about 0.5 millimeter wide; the signals from one
of the Visual Image
eye enter the columns of every other stripe, alter-
nating with signals from the second eye. This cortical
Analysis of Contrasts in the Visual Image. If a person looks
area deciphers whether the respective areas of the
at a blank wall, only a few neurons in the primary
two visual images from the two separate eyes are “in
visual cortex will be stimulated, regardless of whether
register” with each other—that is, whether correspon-
the illumination of the wall is bright or weak. There-
ding points from the two retinas fit with each other. In
fore, the question must be asked, What does the
turn, the deciphered information is used to adjust the
primary visual cortex detect? To answer this, let us now
directional gaze of the separate eyes so that they will
place on the wall a large solid cross, as shown to the
fuse with each other (be brought into “register”). The
left in Figure 51-5. To the right is shown the spatial
information observed about degree of register of
pattern of the most excited neurons in the visual
images from the two eyes also allows a person to dis-
cortex. Note that the areas of maximum excitation
tinguish the distance of objects by the mechanism of
occur along the sharp borders of the visual pattern.
stereopsis.
Thus, the visual signal in the primary visual cortex is
concerned mainly with contrasts in the visual scene,
Two Major Pathways for Analysis of
rather than with noncontrasting areas. We saw in
Visual Information—(1) The Fast
Chapter 50 that this is true of most of the retinal gan-
glion cells as well, because equally stimulated adjacent
“Position” and “Motion” Pathway;
retinal receptors mutually inhibit one another. But at
(2) The Accurate Color Pathway
any border in the visual scene where there is a change
from dark to light or light to dark, mutual inhibition
Figure 51-3 shows that after leaving the primary visual
does not occur, and the intensity of stimulation of most
cortex, the visual information is analyzed in two major
neurons is proportional to the gradient of contrast—
pathways in the secondary visual areas.
that is, the greater the sharpness of contrast and the
greater the intensity difference between light and dark
1. Analysis of Third-Dimensional Position, Gross Form, and
areas, the greater the degree of stimulation.
Motion of Objects. One of the analytical pathways,
demonstrated in Figure 51-3 by the black arrows, ana-
Visual Cortex Also Detects Orientation of Lines and Borders—
lyzes the third-dimensional positions of visual objects
“Simple” Cells. The visual cortex detects not only the
in the space around the body. This pathway also ana-
existence of lines and borders in the different areas of
lyzes the gross physical form of the visual scene as well
the retinal image but also the direction of orientation
as motion in the scene. In other words, this pathway
tells where every object is during each instant and
whether it is moving. After leaving the primary visual
cortex, the signals flow generally into the posterior
midtemporal area and upward into the broad occipi-
toparietal cortex. At the anterior border of the parietal
cortex, the signals overlap with signals from the pos-
terior somatic association areas that analyze three-
dimensional aspects of somatosensory signals. The
signals transmitted in this position-form-motion
pathway are mainly from the large Y optic nerve fibers
of the retinal Y ganglion cells, transmitting rapid
signals but depicting only black and white with no
Retinal image
Cortical stimulation
color.
Figure 51-5
2. Analysis of Visual Detail and Color. The red arrows in
Pattern of excitation that occurs in the visual cortex in response
Figure 51-3, passing from the primary visual cortex
to a retinal image of a dark cross.
644
Unit X The Nervous System: B. The Special Senses
of each line or border—that is, whether it is vertical or
However, psychological studies demonstrate that such
horizontal or lies at some degree of inclination. This is
“blind” people can still, at times, react subconsciously
believed to result from linear organizations of mutu-
to changes in light intensity, to movement in the visual
ally inhibiting cells that excite second-order neurons
scene, or, rarely, even to some gross patterns of vision.
when inhibition occurs all along a line of cells where
These reactions include turning the eyes, turning the
there is a contrast edge. Thus, for each such orienta-
head, and avoidance. This vision is believed to be sub-
tion of a line, specific neuronal cells are stimulated. A
served by neuronal pathways that pass from the optic
line oriented in a different direction excites a different
tracts mainly into the superior colliculi and other por-
set of cells. These neuronal cells are called simple cells.
tions of the older visual system.
They are found mainly in layer IV of the primary
visual cortex.
Fields of Vision; Perimetry
Detection of Line Orientation When a Line Is Displaced Later-
The field of vision is the visual area seen by an eye at a
ally or Vertically in the Visual Field—“Complex” Cells. As
given instant. The area seen to the nasal side is called
the visual signal progresses farther away from layer IV,
the nasal field of vision, and the area seen to the lateral
some neurons respond to lines that are oriented in the
side is called the temporal field of vision.
same direction but are not position-specific. That is,
To diagnose blindness in specific portions of the
even if a line is displaced moderate distances laterally
retina, one charts the field of vision for each eye by a
process called perimetry. This is done by having the
or vertically in the field, the same few neurons will still
subject look with one eye closed and the other eye
be stimulated if the line has the same direction. These
looking toward a central spot directly in front of the eye.
cells are called complex cells.
Then a small dot of light or a small object is moved back
and forth in all areas of the field of vision, and the
Detection of Lines of Specific Lengths, Angles, or Other Shapes.
subject indicates when the spot of light or object can be
Some neurons in the outer layers of the primary visual
seen and when it cannot. Thus, the field of vision for the
columns, as well as neurons in some secondary visual
left eye is plotted as shown in Figure 51-6. In all perime-
areas, are stimulated only by lines or borders of spe-
try charts, a blind spot caused by lack of rods and cones
cific lengths, by specific angulated shapes, or by images
in the retina over the optic disc is found about 15
that have other characteristics. That is, these neurons
degrees lateral to the central point of vision, as shown
in the figure.
detect still higher orders of information from the
visual scene. Thus, as one goes farther into the analyt-
Abnormalities in the Fields of Vision. Occasionally, blind
ical pathway of the visual cortex, progressively more
spots are found in portions of the field of vision other
characteristics of each visual scene are deciphered.
than the optic disc area. Such blind spots are called sco-
tomata; they frequently are caused by damage to the
optic nerve resulting from glaucoma (too much fluid
Detection of Color
pressure in the eyeball), from allergic reactions in the
retina, or from toxic conditions such as lead poisoning
Color is detected in much the same way that lines are
or excessive use of tobacco.
detected: by means of color contrast. For instance, a
red area is often contrasted against a green area, a blue
area against a red area, or a green area against a yellow
area. All these colors can also be contrasted against a
105
90
75
Left
Right
white area within the visual scene. In fact, this con-
120
80
60
70
trasting against white is believed to be mainly respon-
135
45
60
sible for the phenomenon called “color constancy”;
50
that is, when the color of an illuminating light changes,
150
30
40
the color of the “white” changes with the light, and
30
appropriate computation in the brain allows red to be
165
20
15
interpreted as red even though the illuminating light
10
has changed the color entering the eyes.
180
80
70 6050 4030 20
102030 4050 6070
80
0
The mechanism of color contrast analysis depends
on the fact that contrasting colors, called “opponent
Optic
10
colors,” excite specific neuronal cells. It is presumed
disc
195
20
345
that the initial details of color contrast are detected by
30
simple cells, whereas more complex contrasts are
40
330
210
detected by complex and hypercomplex cells.
50
60
225
315
70
Effect of Removing the Primary
240
80
300
255
285
Visual Cortex
270
Figure 51-6
Removal of the primary visual cortex in the human
being causes loss of conscious vision, that is, blindness.
Perimetry chart, showing the field of vision for the left eye.
Chapter 51
The Eye: III. Central Neurophysiology of Vision
645
Still another condition that can be diagnosed by
superior and inferior recti, and (3) the superior and
perimetry is retinitis pigmentosa. In this disease, por-
inferior obliques. The medial and lateral recti contract
tions of the retina degenerate, and excessive melanin
to move the eyes from side to side. The superior and
pigment deposits in the degenerated areas. Retinitis pig-
inferior recti contract to move the eyes upward or
mentosa usually causes blindness in the peripheral field
downward. The oblique muscles function mainly to
of vision first and then gradually encroaches on the
rotate the eyeballs to keep the visual fields in the
central areas.
upright position.
Effect of Lesions in the Optic Pathway on the Fields of
Vision. Destruction of an entire optic nerve causes
Neural Pathways for Control of Eye Movements. Figure 51-7
blindness of the affected eye.
also shows brain stem nuclei for the third, fourth, and
Destruction of the optic chiasm prevents the crossing
sixth cranial nerves and their connections with the
of impulses from the nasal half of each retina to the
peripheral nerves to the ocular muscles. Shown, too,
opposite optic tract. Therefore, the nasal half of each
are interconnections among the brain stem nuclei by
retina is blinded, which means that the person is blind
way of the nerve tract called the medial longitudinal
in the temporal field of vision for each eye because the
fasciculus. Each of the three sets of muscles to each
image of the field of vision is inverted on the retina by
eye is reciprocally innervated so that one muscle of the
the optical system of the eye; this condition is called
bitemporal hemianopsia. Such lesions frequently result
pair relaxes while the other contracts.
from tumors of the pituitary gland pressing upward
Figure 51-8 demonstrates cortical control of the
from the sella turcica on the bottom of the optic chiasm.
oculomotor apparatus, showing spread of signals from
Interruption of an optic tract denervates the corre-
visual areas in the occipital cortex through occipito-
sponding half of each retina on the same side as the
tectal and occipitocollicular tracts to the pretectal and
lesion; as a result, neither eye can see objects to the
superior colliculus areas of the brain stem. From both
opposite side of the head. This condition is known as
the pretectal and the superior colliculus areas, the ocu-
homonymous hemianopsia.
lomotor control signals pass to the brain stem nuclei
of the oculomotor nerves. Strong signals are also trans-
mitted from the body’s equilibrium control centers in
Eye Movements and
the brain stem into the oculomotor system (from the
Their Control
vestibular nuclei by way of the medial longitudinal
fasciculus).
To make full use of the visual abilities of the eyes,
almost equally as important as interpretation of the
visual signals from the eyes is the cerebral control
Fixation Movements of the Eyes
system for directing the eyes toward the object to be
viewed.
Perhaps the most important movements of the eyes
are those that cause the eyes to “fix” on a discrete
Muscular Control of Eye Movements. The eye movements
portion of the field of vision. Fixation movements are
are controlled by three pairs of muscles, shown in
controlled by two neuronal mechanisms. The first of
Figure 51-7: (1) the medial and lateral recti, (2) the
these allows a person to move the eyes voluntarily to
find the object on which he or she wants to fix the
vision; this is called the voluntary fixation mechanism.
The second is an involuntary mechanism that holds the
Superior rectus
eyes firmly on the object once it has been found; this
Inferior oblique
is called the involuntary fixation mechanism.
The voluntary fixation movements are controlled by
a cortical field located bilaterally in the premotor cor-
Superior oblique
tical regions of the frontal lobes, as shown in Figure
Inferior rectus
51-8. Bilateral dysfunction or destruction of these
Medial rectus
areas makes it difficult or almost impossible for a
person to “unlock” the eyes from one point of fixation
and move them to another point. It is usually neces-
sary to blink the eyes or put a hand over the eyes for
a short time, which then allows the eyes to be moved.
N.III
Nuclei
Conversely, the fixation mechanism that causes the
eyes to “lock” on the object of attention once it is
N.IV
found is controlled by secondary visual areas in the
Lateral
occipital cortex, located mainly anterior to the primary
rectus
Medial
visual cortex. When this fixation area is destroyed
longitudinal
N.VI
bilaterally in an animal, the animal has difficulty
fasciculus
keeping its eyes directed toward a given fixation point
or may become totally unable to do so.
Figure 51-7
To summarize, posterior “involuntary” occipital cor-
Extraocular muscles of the eye and their innervation.
tical eye fields automatically “lock” the eyes on a given
646
Unit X The Nervous System: B. The Special Senses
Voluntary fixation
area
Involuntary
fixation
area
Visual
association
areas
Primary
visual cortex
Occipitotectal and
occipitocollicular tracts
Frontotectal tract
Pretectal nuclei
Visceral nucleus III nerve
Superior colliculus
Oculomotor nucleus
III nerve
Trochlear nucleus
Abducens nucleus
IV nerve
VI nerve
Vestibular nuclei
Figure 51-8
Medial longitudinal
Neural pathways for control of
conjugate movement of the eyes.
spot of the visual field and thereby prevent movement
These drifting and flicking motions are demon-
of the image across the retinas. To unlock this visual
strated in Figure 51-9, which shows by the dashed lines
fixation, voluntary signals must be transmitted from
the slow drifting across the fovea and by the solid lines
cortical “voluntary” eye fields located in the frontal
the flicks that keep the image from leaving the foveal
cortices.
region. This involuntary fixation capability is mostly
lost when the superior colliculi are destroyed.
Mechanism of Involuntary Locking Fixation—Role of the Supe-
rior Colliculi. The involuntary locking type of fixation
Saccadic Movement of the Eyes—A Mechanism of Successive
discussed in the previous section results from a nega-
Fixation Points. When a visual scene is moving contin-
tive feedback mechanism that prevents the object of
ually before the eyes, such as when a person is riding
attention from leaving the foveal portion of the retina.
in a car, the eyes fix on one highlight after another in
The eyes normally have three types of continuous but
the visual field, jumping from one to the next at a rate
almost imperceptible movements: (1) a continuous
of two to three jumps per second. The jumps are called
tremor at a rate of 30 to 80 cycles per second caused
saccades, and the movements are called opticokinetic
by successive contractions of the motor units in the
movements. The saccades occur so rapidly that no
ocular muscles, (2) a slow drift of the eyeballs in one
more than 10 per cent of the total time is spent in
direction or another, and (3) sudden flicking move-
moving the eyes, with 90 per cent of the time being
ments that are controlled by the involuntary fixation
allocated to the fixation sites. Also, the brain sup-
mechanism.
presses the visual image during saccades, so that the
When a spot of light has become fixed on the foveal
person is not conscious of the movements from point
region of the retina, the tremulous movements cause
to point.
the spot to move back and forth at a rapid rate across
the cones, and the drifting movements cause the spot
Saccadic Movements During Reading. During the
to drift slowly across the cones. Each time the spot
process of reading, a person usually makes several sac-
drifts as far as the edge of the fovea, a sudden reflex
cadic movements of the eyes for each line. In this case,
reaction occurs, producing a flicking movement that
the visual scene is not moving past the eyes, but the
moves the spot away from this edge back toward the
eyes are trained to move by means of several succes-
center of the fovea. Thus, an automatic response moves
sive saccades across the visual scene to extract the
the image back toward the central point of vision.
important information. Similar saccades occur when a
Chapter 51
The Eye: III. Central Neurophysiology of Vision
647
directional movement of the eyes, the superior colli-
culi also have topological maps of somatic sensations
from the body and acoustic signals from the ears.
The optic nerve fibers from the eyes to the colliculi
that are responsible for these rapid turning move-
ments are branches from the rapidly conducting Y
fibers, with one branch going to the visual cortex and
the other going to the superior colliculi. (The superior
colliculi and other regions of the brain stem are also
strongly supplied with visual signals transmitted in
Voluntary
type W optic nerve fibers. These represent the oldest
movement to
fixation site
visual pathway, but their function is unclear.)
In addition to causing the eyes to turn toward a
visual disturbance, signals are relayed from the supe-
rior colliculi through the medial longitudinal fascicu-
Figure 51-9
lus to other levels of the brain stem to cause turning
of the whole head and even of the whole body toward
Movements of a spot of light on the fovea, showing sudden “flick-
ing” eye movements that move the spot back toward the center
the direction of the disturbance. Other types of non-
of the fovea whenever it drifts to the foveal edge. (The dashed
visual disturbances, such as strong sounds or even
lines represent slow drifting movements, and the solid lines rep-
stroking of the side of the body, cause similar turning
resent sudden flicking movements.) (Modified from Whitteridge D:
of the eyes, head, and body, but only if the superior col-
Central control of the eye movements. In Field J, Magoun HW, Hall
VE (eds): Handbook of Physiology. vol. 2, sec. 1. Washington, DC,
liculi are intact. Therefore, the superior colliculi play a
American Physiological Society, 1960.)
global role in orienting the eyes, head, and body with
respect to external disturbances, whether they are
person observes a painting, except that the saccades
visual, auditory, or somatic.
occur in upward, sideways, downward, and angulated
directions one after another from one highlight of the
painting to another, and so forth.
“Fusion” of the Visual Images from
the Two Eyes
Fixation on Moving Objects—“Pursuit Movement.” The eyes
can also remain fixed on a moving object, which is
To make the visual perceptions more meaningful, the
called pursuit movement. A highly developed cortical
visual images in the two eyes normally fuse with each
mechanism automatically detects the course of move-
other on “corresponding points” of the two retinas.
ment of an object and then rapidly develops a similar
The visual cortex plays an important role in fusion. It
course of movement for the eyes. For instance, if an
was pointed out earlier in the chapter that correspon-
object is moving up and down in a wavelike form at a
ding points of the two retinas transmit visual signals to
rate of several times per second, the eyes at first may
different neuronal layers of the lateral geniculate
be unable to fixate on it. However, after a second or
body, and these signals in turn are relayed to parallel
so, the eyes begin to jump by means of saccades in
neurons in the visual cortex. Interactions occur
approximately the same wavelike pattern of move-
between these cortical neurons to cause interference
ment as that of the object. Then, after another few
excitation in specific neurons when the two visual
seconds, the eyes develop progressively smoother
images are not “in register”—that is, are not precisely
movements and finally follow the wave movement
“fused.” This excitation presumably provides the
almost exactly. This represents a high degree of auto-
signal that is transmitted to the oculomotor apparatus
matic subconscious computational ability by the
to cause convergence or divergence or rotation of the
pursuit system for controlling eye movements.
eyes so that fusion can be re-established. Once the cor-
responding points of the two retinas are in register,
Superior Colliculi Are Mainly Responsible
excitation of the specific “interference” neurons in the
for Turning the Eyes and Head Toward a
visual cortex disappears.
Visual Disturbance
Even after the visual cortex has been destroyed, a
Neural Mechanism of Stereopsis for Judging
sudden visual disturbance in a lateral area of the visual
Distances of Visual Objects
field often causes immediate turning of the eyes in that
In Chapter 49, it is pointed out that because the two
direction. This does not occur if the superior colliculi
eyes are more than 2 inches apart, the images on the
have also been destroyed. To support this function, the
two retinas are not exactly the same. That is, the right
various points of the retina are represented topo-
eye sees a little more of the right-hand side of the
graphically in the superior colliculi in the same way as
object, and the left eye a little more of the left-hand
in the primary visual cortex, although with less accu-
side, and the closer the object, the greater the dispar-
racy. Even so, the principal direction of a flash of light
ity. Therefore, even when the two eyes are fused with
in a peripheral retinal field is mapped by the colliculi,
each other, it is still impossible for all corresponding
and secondary signals are transmitted to the oculo-
points in the two visual images to be exactly in regis-
motor nuclei to turn the eyes. To help in this
ter at the same time. Furthermore, the nearer the
648
Unit X The Nervous System: B. The Special Senses
object is to the eyes, the less the degree of register. This
Autonomic Control of
degree of nonregister provides the neural mechanism
Accommodation and
for stereopsis, an important mechanism for judging
Pupillary Aperture
the distances of visual objects up to about 200 feet
(60 meters).
Autonomic Nerves to the Eyes. The eye is innervated by
The neuronal cellular mechanism for stereopsis is
both parasympathetic and sympathetic nerve fibers, as
based on the fact that some of the fiber pathways from
shown in Figure 51-11. The parasympathetic pregan-
the retinas to the visual cortex stray 1 to 2 degrees on
glionic fibers arise in the Edinger-Westphal nucleus
each side of the central pathway. Therefore, some optic
(the visceral nucleus portion of the third cranial nerve)
pathways from the two eyes are exactly in register for
and then pass in the third nerve to the ciliary ganglion,
objects 2 meters away; still another set of pathways is
which lies immediately behind the eye. There, the pre-
in register for objects 25 meters away. Thus, the dis-
ganglionic fibers synapse with postganglionic parasym-
tance is determined by which set or sets of pathways
pathetic neurons, which in turn send fibers through
are excited by nonregister or register. This phenome-
ciliary nerves into the eyeball. These nerves excite (1)
non is called depth perception, which is another name
the ciliary muscle that controls focusing of the eye lens
for stereopsis.
and (2) the sphincter of the iris that constricts the
Strabismus
pupil.
Strabismus, also called squint or cross-eye, means lack
The sympathetic innervation of the eye originates in
of fusion of the eyes in one or more of the visual coor-
the intermediolateral horn cells of the first thoracic
dinates: horizontal, vertical, or rotational. The basic
segment of the spinal cord. From there, sympathetic
types of strabismus are shown in Figure 51-10: (1) hor-
fibers enter the sympathetic chain and pass upward to
izontal strabismus, (2) torsional strabismus, and (3) ver-
the superior cervical ganglion, where they synapse
tical strabismus. Combinations of two or even all three
with postganglionic neurons. Postganglionic sympa-
of the different types of strabismus often occur.
thetic fibers from these then spread along the surfaces
Strabismus is often caused by abnormal “set” of the
of the carotid artery and successively smaller arteries
fusion mechanism of the visual system. That is, in a
young child’s early efforts to fixate the two eyes on the
until they reach the eye. There, the sympathetic fibers
same object, one of the eyes fixates satisfactorily while
the other fails do so, or they both fixate satisfactorily but
never simultaneously. Soon the patterns of conjugate
movements of the eyes become abnormally “set” in the
neuronal control pathways themselves, so that the eyes
never fuse.
Suppression of the Visual Image from a Repressed Eye. In a few
patients with strabismus, the eyes alternate in fixing on
Edinger-
the object of attention. In other patients, one eye alone
Pretectal
Westphal
Ciliary
is used all the time, and the other eye becomes repressed
region
nucleus
ganglion
and is never used for precise vision. The visual acuity of
the repressed eye develops only slightly, sometimes
N.III
N.II
remaining 20/400 or less. If the dominant eye then
becomes blinded, vision in the repressed eye can
develop only to a slight extent in adults but far more in
young children. This demonstrates that visual acuity is
highly dependent on proper development of central
nervous system synaptic connections from the eyes. In
Carotid plexus
Pons
fact, even anatomically, the numbers of neuronal con-
nections diminish in the visual cortex areas that would
Superior cervical
normally receive signals from the repressed eye.
sympathetic
ganglion
Cervical
sympathetic
trunk
Upper thoracic segments
of spinal cord
Horizontal
Torsional
Vertical
Figure 51-11
strabismus
strabismus
strabismus
Autonomic innervation of the eye, showing also the reflex arc of
Figure 51-10
the light reflex. (Modified from Ranson SW, Clark SL: Anatomy of
the Nervous System: Its Development and Function, 10th ed.
Basic types of strabismus.
Philadelphia: WB Saunders, 1959.)
Chapter 51
The Eye: III. Central Neurophysiology of Vision
649
innervate the radial fibers of the iris (which open the
The brain cortical areas that control accommoda-
pupil) as well as several extraocular muscles of the eye,
tion closely parallel those that control fixation move-
which are discussed subsequently in relation to
ments of the eyes, with analysis of the visual signals in
Horner’s syndrome.
Brodmann’s cortical areas 18 and 19 and transmission
of motor signals to the ciliary muscle through the
pretectal area in the brain stem, then through the
Control of Accommodation
Edinger-Westphal nucleus, and finally by way of
(Focusing the Eyes)
parasympathetic nerve fibers to the eyes.
The accommodation mechanism—that is, the mecha-
nism that focuses the lens system of the eye—is essen-
Control of Pupillary Diameter
tial for a high degree of visual acuity. Accommodation
results from contraction or relaxation of the eye ciliary
Stimulation of the parasympathetic nerves also excites
muscle. Contraction causes increased refractive power
the pupillary sphincter muscle, thereby decreasing the
of the lens, as explained in Chapter 49, and relaxation
pupillary aperture; this is called miosis. Conversely,
causes decreased power. How does a person adjust
stimulation of the sympathetic nerves excites the
accommodation to keep the eyes in focus all the time?
radial fibers of the iris and causes pupillary dilation,
Accommodation of the lens is regulated by a nega-
called mydriasis.
tive feedback mechanism that automatically adjusts
the refractive power of the lens to achieve the highest
Pupillary Light Reflex. When light is shone into the eyes,
degree of visual acuity. When the eyes have been
the pupils constrict, a reaction called the pupillary light
focused on some far object and must then suddenly
reflex. The neuronal pathway for this reflex is demon-
focus on a near object, the lens usually accommodates
strated by the upper two black traces in Figure 51-11.
for best acuity of vision within less than 1 second.
When light impinges on the retina, a few of the result-
Although the precise control mechanism that causes
ing impulses pass from the optic nerves to the pretec-
this rapid and accurate focusing of the eye is unclear,
tal nuclei. From here, secondary impulses pass to the
some of the known features are the following.
Edinger-Westphal nucleus and, finally, back through
First, when the eyes suddenly change distance of the
parasympathetic nerves to constrict the sphincter of the
fixation point, the lens changes its strength in the
iris. Conversely, in darkness, the reflex becomes inhib-
proper direction to achieve a new state of focus within
ited, which results in dilation of the pupil.
a fraction of a second. Second, different types of
The function of the light reflex is to help the eye
clues help to change the lens strength in the proper
adapt extremely rapidly to changing light conditions,
direction:
as explained in Chapter 50. The limits of pupillary
1.
Chromatic aberration appears to be important.
diameter are about 1.5 millimeters on the small side
That is, red light rays focus slightly posteriorly to
and 8 millimeters on the large side. Therefore, because
blue light rays because the lens bends blue rays
light brightness on the retina increases with the square
more than red rays. The eyes appear to be able to
of pupillary diameter, the range of light and dark adap-
detect which of these two types of rays is in better
tation that can be brought about by the pupillary reflex
focus, and this clue relays information to the
is about 30 to 1—that is, up to as much as 30 times
accommodation mechanism whether to make the
change in the amount of light entering the eye.
lens stronger or weaker.
2.
When the eyes fixate on a near object, the eyes
Pupillary Reflexes or Reactions in Central Nervous System
must converge. The neural mechanisms for
Disease. A few central nervous system diseases damage
convergence cause a simultaneous signal to
nerve transmission of visual signals from the retinas to
the Edinger-Westphal nucleus, thus sometimes blocking
strengthen the lens of the eye.
the pupillary reflexes. Such blocks frequently occur as a
3.
Because the fovea lies in a hollowed-out depression
result of central nervous system syphilis, alcoholism,
that is slightly deeper than the remainder of the
encephalitis, and so forth. The block usually occurs in the
retina, the clarity of focus in the depth of the fovea
pretectal region of the brain stem, although it can result
is different from the clarity of focus on the edges. It
from destruction of some small fibers in the optic
has been suggested that this also gives clues about
nerves.
which way the strength of the lens needs to be
The final nerve fibers in the pathway through the pre-
changed.
tectal area to the Edinger-Westphal nucleus are mostly
4.
It has been found that the degree of
of the inhibitory type. When their inhibitory effect is
accommodation of the lens oscillates slightly all
lost, the nucleus becomes chronically active, causing the
pupils to remain mostly constricted, in addition to their
the time at a frequency up to twice per second.
failure to respond to light.
The visual image becomes clearer when the
Yet the pupils can constrict a little more if the
oscillation of the lens strength is changing in
Edinger-Westphal nucleus is stimulated through some
the appropriate direction and becomes poorer
other pathway. For instance, when the eyes fixate on a
when the lens strength is changing in the wrong
near object, the signals that cause accommodation of
direction. This could give a rapid clue as to which
the lens and those that cause convergence of the two
way the strength of the lens needs to change to
eyes cause a mild degree of pupillary constriction at
provide appropriate focus.
the same time. This is called the pupillary reaction to
650
Unit X The Nervous System: B. The Special Senses
accommodation. A pupil that fails to respond to light
Crawford JD, Martinez-Trujillo JC, Klier EM: Neural control
but does respond to accommodation and is also very
of three-dimensional eye and head movements. Curr Opin
small (an Argyll Robertson pupil) is an important diag-
Neurobiol 13:655, 2003.
nostic sign of central nervous system disease—often
Dacey DM: Parallel pathways for spectral coding in primate
syphilis.
retina. Annu Rev Neurosci 23:743, 2000.
Demer JL: The orbital pulley system: a revolution in con-
Horner’s Syndrome. The sympathetic nerves to the eye are
cepts of orbital anatomy. Ann N Y Acad Sci 956:17, 2002.
occasionally interrupted. Interruption frequently occurs
Derrington AM, Webb BS: Visual system: how is the retina
in the cervical sympathetic chain. This causes the clini-
wired up to the cortex? Curr Biol 14:R14, 2004.
cal condition called Horner’s syndrome, which consists
Ferster D, Miller KD: Neural mechanisms of orientation
of the following effects: First, because of interruption of
selectivity in the visual cortex. Annu Rev Neurosci 23:441,
sympathetic nerve fibers to the pupillary dilator muscle,
2000.
the pupil remains persistently constricted to a smaller
Hendee WA, Wells PNT: The Perception of Visual Informa-
diameter than the pupil of the opposite eye. Second, the
tion. New York: Springer, 1997.
superior eyelid droops because it is normally main-
Hikosaka O, Takikawa Y, Kawagoe R: Role of the basal
tained in an open position during waking hours partly
ganglia in the control of purposive saccadic eye move-
by contraction of smooth muscle fibers embedded in the
ments. Physiol Rev 80:953, 2000.
superior eyelid and innervated by the sympathetics.
Kastner S, Ungerleider LG: Mechanisms of visual attention
Therefore, destruction of the sympathetic nerves makes
in the human cortex. Annu Rev Neurosci 23:315, 2000.
it impossible to open the superior eyelid as widely as
Krauzlis RJ: Recasting the smooth pursuit eye movement
normally. Third, the blood vessels on the corresponding
system. J Neurophysiol 91:591, 2004.
side of the face and head become persistently dilated.
Leigh RJ, Lee DS: The Neurology of Eye Movements. New
Fourth, sweating (which requires sympathetic nerve
York: Oxford University Press, 1999.
signals) cannot occur on the side of the face and head
Mante V, Carandini M: Visual cortex: seeing motion. Curr
affected by Horner’s syndrome.
Biol 13:R906, 2003.
Martinez-Conde S, Macknik SL, Hubel DH: The role of
fixational eye movements in visual perception. Nat Rev
References
Neurosci 5:229, 2004.
Munoz DP, Everling S: Look away: the anti-saccade task and
the voluntary control of eye movement. Nat Rev Neurosci
Backharis W, Kliegl R, Werner JS: Color Vision. Berlin:
5:218, 2004.
Walter de Gruyter, 1998.
Pierrot-Deseilligny C, Milea D, Muri RM: Eye movement
Becker W, Reubel H, Mergner T: Current Oculomotor
control by the cerebral cortex. Curr Opin Neurol 17:17,
Research. New York: Plenum Press, 1999.
2004.
Burr D: Eye movements: keeping vision stable. Curr Biol
Treue S: Visual attention: the where, what, how and why of
14:R195, 2004.
saliency. Curr Opin Neurobiol 13:428, 2003.
Buttner-Ennever JA, Eberhorn A, Horn AK: Motor and
van Wezel RJ, van der Smagt MJ: Motion processing: how
sensory innervation of extraocular eye muscles. Ann N Y
low can you go? Curr Biol 13:R840, 2003.
Acad Sci 1004:40, 2003.
Chalupa LM, Gunhan E: Development of on and off retinal
pathways and retinogeniculate projections. Prog Retin
Eye Res 23:31, 2004.
C H A P T E R
5
2
The Sense of Hearing
This chapter describes the mechanisms by which the
ear receives sound waves, discriminates their fre-
quencies, and transmits auditory information into
the central nervous system, where its meaning is
deciphered.
Tympanic Membrane and the Ossicular System
Conduction of Sound from the Tympanic Membrane
to the Cochlea
Figure 52-1 shows the tympanic membrane (commonly called the eardrum) and
the ossicles, which conduct sound from the tympanic membrane through the
middle ear to the cochlea (the inner ear). Attached to the tympanic membrane
is the handle of the malleus. The malleus is bound to the incus by minute liga-
ments, so that whenever the malleus moves, the incus moves with it. The oppo-
site end of the incus articulates with the stem of the stapes, and the faceplate of
the stapes lies against the membranous labyrinth of the cochlea in the opening
of the oval window.
The tip end of the handle of the malleus is attached to the center of the tym-
panic membrane, and this point of attachment is constantly pulled by the tensor
tympani muscle, which keeps the tympanic membrane tensed. This allows sound
vibrations on any portion of the tympanic membrane to be transmitted to the
ossicles, which would not be true if the membrane were lax.
The ossicles of the middle ear are suspended by ligaments in such a way that
the combined malleus and incus act as a single lever, having its fulcrum approx-
imately at the border of the tympanic membrane.
The articulation of the incus with the stapes causes the stapes to push forward
on the oval window and on the cochlear fluid on the other side of window every
time the tympanic membrane moves inward, and to pull backward on the fluid
every time the malleus moves outward.
“Impedance Matching” by the Ossicular System. The amplitude of movement of the
stapes faceplate with each sound vibration is only three fourths as much as the
amplitude of the handle of the malleus. Therefore, the ossicular lever system
does not increase the movement distance of the stapes, as is commonly believed.
Instead, the system actually reduces the distance but increases the force of
movement about 1.3 times. In addition, the surface area of the tympanic mem-
brane is about 55 square millimeters, whereas the surface area of the stapes
averages 3.2 square millimeters. This 17-fold difference times the 1.3-fold ratio
of the lever system causes about 22 times as much total force to be exerted on
the fluid of the cochlea as is exerted by the sound waves against the tympanic
membrane. Because fluid has far greater inertia than air does, it is easily under-
stood that increased amounts of force are needed to cause vibration in the fluid.
Therefore, the tympanic membrane and ossicular system provide impedance
matching between the sound waves in air and the sound vibrations in the fluid
of the cochlea. Indeed, the impedance matching is about 50 to 75 per cent of
perfect for sound frequencies between 300 and 3000 cycles per second, which
allows utilization of most of the energy in the incoming sound waves.
651
652
Unit X The Nervous System: B. The Special Senses
Malleus
Basilar
Spiral organ
membrane of Corti
Stapes
Incus
Spiral
Vestibular membrane
Scala tympani
ligament
Scala vestibuli
Scala vestibuli
Stria
Cochlear
vascularis
nerve
Auditory canal
Spiral
ganglion
Cochlea
Round
Tympanic membrane
window
Oval window
Scala media
Figure 52-1
Spiral ganglion
Cochlear nerve
Scala tympani
Tympanic membrane, ossicular system of the middle ear, and
inner ear.
Figure 52-2
Cochlea. (Redrawn from Gray H, Goss CM [eds]: Gray’s Anatomy
In the absence of the ossicular system and tympanic
of the Human Body. Philadelphia: Lea & Febiger, 1948.)
membrane, sound waves can still travel directly
through the air of the middle ear and enter the cochlea
at the oval window. However, the sensitivity for
hearing is then 15 to 20 decibels less than for ossicu-
Transmission of Sound Through Bone
lar transmission—equivalent to a decrease from a
medium to a barely perceptible voice level.
Because the inner ear, the cochlea, is embedded in a
bony cavity in the temporal bone, called the bony
Attenuation of Sound by Contraction of the Tensor Tympani and
labyrinth, vibrations of the entire skull can cause fluid
Stapedius Muscles. When loud sounds are transmitted
vibrations in the cochlea itself. Therefore, under appro-
through the ossicular system and from there into the
priate conditions, a tuning fork or an electronic vibra-
central nervous system, a reflex occurs after a latent
tor placed on any bony protuberance of the skull, but
period of only 40 to 80 milliseconds to cause contrac-
especially on the mastoid process near the ear, causes
tion of the stapedius muscle and, to a lesser extent, the
the person to hear the sound. However, the energy
tensor tympani muscle. The tensor tympani muscle
available even in loud sound in the air is not sufficient
pulls the handle of the malleus inward while the
to cause hearing via bone conduction unless a special
stapedius muscle pulls the stapes outward. These two
electromechanical sound-amplifying device is applied
forces oppose each other and thereby cause the entire
to the bone.
ossicular system to develop increased rigidity, thus
greatly reducing the ossicular conduction of low-
frequency sound, mainly frequencies below
1000
Cochlea
cycles per second.
This attenuation reflex can reduce the intensity of
Functional Anatomy of the Cochlea
lower-frequency sound transmission by 30 to 40 deci-
bels, which is about the same difference as that
The cochlea is a system of coiled tubes, shown in
between a loud voice and a whisper. The function of
Figure 52-1 and in cross section in Figures 52-2 and
this mechanism is believed to be twofold:
52-3. It consists of three tubes coiled side by side: (1)
1. To protect the cochlea from damaging vibrations
the scala vestibuli, (2) the scala media, and (3) the scala
caused by excessively loud sound.
tympani. The scala vestibuli and scala media are sepa-
2. To mask low-frequency sounds in loud
rated from each other by Reissner’s membrane (also
environments. This usually removes a major share
called the vestibular membrane), shown in Figure 52-3;
of the background noise and allows a person to
the scala tympani and scala media are separated from
concentrate on sounds above 1000 cycles per
each other by the basilar membrane. On the surface of
second, where most of the pertinent information
the basilar membrane lies the organ of Corti, which
in voice communication is transmitted.
contains a series of electromechanically sensitive
Another function of the tensor tympani and
cells, the hair cells. They are the receptive end organs
stapedius muscles is to decrease a person’s hearing
that generate nerve impulses in response to sound
sensitivity to his or her own speech. This effect is acti-
vibrations.
vated by collateral nerve signals transmitted to these
Figure 52-4 diagrams the functional parts of the
muscles at the same time that the brain activates the
uncoiled cochlea for conduction of sound vibrations.
voice mechanism.
First, note that Reissner’s membrane is missing from
Chapter 52
The Sense of Hearing
653
Tectorial membrane
Reissner's membrane
Scala vestibuli
Stria vascularis
Spiral limbus
Scala media
Spiral
prominence
Organ of Corti
Figure 52-3
Spiral ganglion
Section through one of the turns
Basilar membrane
of the cochlea. (Drawn by Sylvia
Colard Keene. From Fawcett DW:
Bloom & Fawcett: A Textbook of
Histology, 11th ed. Philadelphia:
Scala tympani
WB Saunders, 1986.)
Oval
Scala vestibuli
Basilar Membrane and Resonance in the Cochlea. The
window
and scala media
basilar membrane is a fibrous membrane that sepa-
Stapes
rates the scala media from the scala tympani. It
contains 20,000 to 30,000 basilar fibers that project
from the bony center of the cochlea, the modiolus,
toward the outer wall. These fibers are stiff, elastic,
reedlike structures that are fixed at their basal ends
Round
in the central bony structure of the cochlea (the modi-
Scala
Basilar
Helicotrema
window
tympani
membrane
olus) but are not fixed at their distal ends, except
that the distal ends are embedded in the loose basilar
Figure 52-4
membrane. Because the fibers are stiff and free at
one end, they can vibrate like the reeds of a
Movement of fluid in the cochlea after forward thrust of the stapes.
harmonica.
The lengths of the basilar fibers increase progres-
sively beginning at the oval window and going from
this figure. This membrane is so thin and so easily
the base of the cochlea to the apex, increasing from a
moved that it does not obstruct the passage of sound
length of about 0.04 millimeter near the oval and
vibrations from the scala vestibuli into the scala media.
round windows to 0.5 millimeter at the tip of the
Therefore, as far as fluid conduction of sound is con-
cochlea
(the
“helicotrema”), a
12-fold increase in
cerned, the scala vestibuli and scala media are con-
length.
sidered to be a single chamber. (The importance of
The diameters of the fibers, however, decrease from
Reissner’s membrane is to maintain a special kind of
the oval window to the helicotrema, so that their
fluid in the scala media that is required for normal
overall stiffness decreases more than 100-fold. As a
function of the sound-receptive hair cells, as discussed
result, the stiff, short fibers near the oval window of the
later in the chapter.)
cochlea vibrate best at a very high frequency, while the
Sound vibrations enter the scala vestibuli from the
long, limber fibers near the tip of the cochlea vibrate
faceplate of the stapes at the oval window. The face-
best at a low frequency.
plate covers this window and is connected with the
Thus, high-frequency resonance of the basilar mem-
window’s edges by a loose annular ligament so that it
brane occurs near the base, where the sound waves
can move inward and outward with the sound vibra-
enter the cochlea through the oval window. But low-
tions. Inward movement causes the fluid to move
frequency resonance occurs near the helicotrema,
forward in the scala vestibuli and scala media, and
mainly because of the less stiff fibers but also because
outward movement causes the fluid to move back-
of increased “loading” with extra masses of fluid that
ward.
must vibrate along the cochlear tubules.
654
Unit X The Nervous System: B. The Special Senses
Transmission of Sound Waves in the
before it reaches its resonant point and dies, a
medium-frequency sound wave travels about halfway
Cochlea—“Traveling Wave”
and then dies, and a very low frequency sound wave
travels the entire distance along the membrane.
When the foot of the stapes moves inward against the
Another feature of the traveling wave is that it
oval window, the round window must bulge outward
travels fast along the initial portion of the basilar
because the cochlea is bounded on all sides by bony
membrane but becomes progressively slower as it goes
walls. The initial effect of a sound wave entering at the
farther into the cochlea. The cause of this is the high
oval window is to cause the basilar membrane at
coefficient of elasticity of the basilar fibers near the
the base of the cochlea to bend in the direction of the
oval window and a progressively decreasing coefficient
round window. However, the elastic tension that is
farther along the membrane. This rapid initial trans-
built up in the basilar fibers as they bend toward the
mission of the wave allows the high-frequency sounds
round window initiates a fluid wave that “travels”
to travel far enough into the cochlea to spread out and
along the basilar membrane toward the helicotrema,
separate from one another on the basilar membrane.
as shown in Figure 52-5. Figure 52-5A shows move-
Without this, all the high-frequency waves would be
ment of a high-frequency wave down the basilar mem-
bunched together within the first millimeter or so of
brane; Figure 52-5B, a medium-frequency wave; and
the basilar membrane, and their frequencies could not
Figure 52-5C, a very low frequency wave. Movement
be discriminated from one another.
of the wave along the basilar membrane is compara-
ble to the movement of a pressure wave along the arte-
Amplitude Pattern of Vibration of the Basilar Mem-
rial walls, which is discussed in Chapter 15; it is also
brane. The dashed curves of Figure 52-6A show the
comparable to a wave that travels along the surface of
position of a sound wave on the basilar membrane
a pond.
when the stapes (a) is all the way inward, (b) has
moved back to the neutral point, (c) is all the way
Pattern of Vibration of the Basilar Membrane for Different
outward, and (d) has moved back again to the neutral
Sound Frequencies. Note in Figure 52-5 the different
point but is moving inward. The shaded area around
patterns of transmission for sound waves of different
these different waves shows the extent of vibration of
frequencies. Each wave is relatively weak at the outset
the basilar membrane during a complete vibratory
but becomes strong when it reaches that portion of the
cycle. This is the amplitude pattern of vibration of
basilar membrane that has a natural resonant fre-
the basilar membrane for this particular sound
quency equal to the respective sound frequency. At
frequency.
this point, the basilar membrane can vibrate back and
Figure 52-6B shows the amplitude patterns of vibra-
forth with such ease that the energy in the wave is dis-
tion for different frequencies, demonstrating that the
sipated. Consequently, the wave dies at this point and
fails to travel the remaining distance along the basilar
membrane. Thus, a high-frequency sound wave travels
only a short distance along the basilar membrane
b
a
c
High frequency
A
d
A
Frequency
8000
4000
2000
1000
600
400
200
Medium frequency
B
0
5
10
15
20
25
30
35
Distance from stapes (millimeters)
B
Figure 52-6
C
Low frequency
A, Amplitude pattern of vibration of the basilar membrane for
Figure 52-5
a medium-frequency sound. B, Amplitude patterns for sounds of
frequencies between 200 and 8000 cycles per second, showing
“Traveling waves” along the basilar membrane for high-,
the points of maximum amplitude on the basilar membrane for the
medium-, and low-frequency sounds.
different frequencies.
Chapter 52
The Sense of Hearing
655
maximum amplitude for sound at 8000 cycles per
of the organ of Corti to the spiral ganglion and to the
second occurs near the base of the cochlea, whereas
cochlear nerve is shown in Figure 52-2.
that for frequencies less than 200 cycles per second is
all the way at the tip of the basilar membrane near the
Excitation of the Hair Cells. Note in Figure
52-7 that
helicotrema, where the scala vestibuli opens into the
minute hairs, or stereocilia, project upward from the
scala tympani.
hair cells and either touch or are embedded in the
The principal method by which sound frequencies
surface gel coating of the tectorial membrane, which
are discriminated from one another is based on the
lies above the stereocilia in the scala media. These hair
“place” of maximum stimulation of the nerve fibers
cells are similar to the hair cells found in the macula
from the organ of Corti lying on the basilar membrane,
and cristae ampullaris of the vestibular apparatus,
as explained in the next section.
which are discussed in Chapter 55. Bending of the
hairs in one direction depolarizes the hair cells, and
bending in the opposite direction hyperpolarizes them.
Function of the Organ of Corti
This in turn excites the auditory nerve fibers synaps-
ing with their bases.
The organ of Corti, shown in Figures 52-2, 52-3, and
Figure 52-8 shows the mechanism by which vibra-
52-7, is the receptor organ that generates nerve
tion of the basilar membrane excites the hair endings.
impulses in response to vibration of the basilar mem-
The outer ends of the hair cells are fixed tightly in a
brane. Note that the organ of Corti lies on the surface
rigid structure composed of a flat plate, called the retic-
of the basilar fibers and basilar membrane. The actual
ular lamina, supported by triangular rods of Corti,
sensory receptors in the organ of Corti are two spe-
which are attached tightly to the basilar fibers. The
cialized types of nerve cells called hair cells—a single
basilar fibers, the rods of Corti, and the reticular
row of internal (or “inner”) hair cells, numbering about
lamina move as a rigid unit.
3500 and measuring about 12 micrometers in diame-
Upward movement of the basilar fiber rocks the
ter, and three or four rows of external (or “outer”) hair
reticular lamina upward and inward toward the modi-
cells, numbering about 12,000 and having diameters
olus. Then, when the basilar membrane moves down-
of only about 8 micrometers. The bases and sides of
ward, the reticular lamina rocks downward and
the hair cells synapse with a network of cochlea
outward. The inward and outward motion causes the
nerve endings. Between 90 and 95 per cent of these
hairs on the hair cells to shear back and forth against
endings terminate on the inner hair cells, which
the tectorial membrane. Thus, the hair cells are excited
emphasizes their special importance for the detection
whenever the basilar membrane vibrates.
of sound.
The nerve fibers stimulated by the hair cells lead to
Auditory Signals Are Transmitted Mainly by the Inner Hair Cells.
the spiral ganglion of Corti, which lies in the modiolus
Even though there are three to four times as many
(center) of the cochlea. The spiral ganglion neuronal
outer hair cells as inner hair cells, about 90 per cent of
cells send axons—a total of about 30,000—into the
the auditory nerve fibers are stimulated by the inner
cochlear nerve and then into the central nervous
cells rather than by the outer cells. Yet, despite this, if
system at the level of the upper medulla. The relation
the outer cells are damaged while the inner cells
remain fully functional, a large amount of hearing
loss occurs. Therefore, it has been proposed that the
outer hair cells in some way control the sensitivity
of the inner hair cells at different sound pitches, a
Tectorial membrane
Inner hair cells
Outer hair cells
Reticular lamina
Hairs Tectorial membrane
Basilar fiber
Basilar fiber
Rods of Corti
Spiral ganglion
Modiolus
Cochlear nerve
Figure 52-7
Figure 52-8
Organ of Corti, showing especially the hair cells and the tectorial
Stimulation of the hair cells by to-and-fro movement of the hairs
membrane pressing against the projecting hairs.
projecting into the gel coating of the tectorial membrane.
656
Unit X The Nervous System: B. The Special Senses
phenomenon called “tuning” of the receptor system.
the hair cells. Furthermore, the hair cells have a nega-
In support of this concept, a large number of retro-
tive intracellular potential of -70 millivolts with respect
grade nerve fibers pass from the brain stem to the
to the perilymph but -150 millivolts with respect to the
endolymph at their upper surfaces where the hairs
vicinity of the outer hair cells. Stimulating these nerve
project through the reticular lamina and into the
fibers can actually cause shortening of the outer hair
endolymph. It is believed that this high electrical poten-
cells and possibly also change their degree of stiffness.
tial at the tips of the stereocilia sensitizes the cell an
These effects suggest a retrograde nervous mechanism
extra amount, thereby increasing its ability to respond
for control of the ear’s sensitivity to different sound
to the slightest sound.
pitches, activated through the outer hair cells.
Hair Cell Receptor Potentials and Excitation of Auditory Nerve
Determination of Sound Frequency—
Fibers. The stereocilia (the “hairs” protruding from the
The “Place” Principle
ends of the hair cells) are stiff structures because each
has a rigid protein framework. Each hair cell has about
From earlier discussions in this chapter, it is apparent
100 stereocilia on its apical border. These become pro-
that low-frequency sounds cause maximal activation
gressively longer on the side of the hair cell away from
of the basilar membrane near the apex of the cochlea,
the modiolus, and the tops of the shorter stereocilia are
and high-frequency sounds activate the basilar mem-
attached by thin filaments to the back sides of their
brane near the base of the cochlea. Intermediate-
adjacent longer stereocilia. Therefore, whenever the
frequency sounds activate the membrane at inter-
cilia are bent in the direction of the longer ones, the
mediate distances between the two extremes.
tips of the smaller stereocilia are tugged outward from
Furthermore, there is spatial organization of the nerve
the surface of the hair cell. This causes a mechanical
fibers in the cochlear pathway, all the way from the
transduction that opens 200 to 300 cation-conducting
cochlea to the cerebral cortex. Recording of signals in
channels, allowing rapid movement of positively
the auditory tracts of the brain stem and in the audi-
charged potassium ions from the surrounding scala
tory receptive fields of the cerebral cortex shows that
media fluid into the stereocilia, which causes depolar-
specific brain neurons are activated by specific sound
ization of the hair cell membrane.
frequencies. Therefore, the major method used by
Thus, when the basilar fibers bend toward the scala
the nervous system to detect different sound frequen-
vestibuli, the hair cells depolarize, and in the opposite
cies is to determine the positions along the basilar
direction they hyperpolarize, thereby generating an
membrane that are most stimulated. This is called
alternating hair cell receptor potential. This, in turn,
the place principle for the determination of sound
stimulates the cochlear nerve endings that synapse
frequency.
with the bases of the hair cells. It is believed that a
Yet, referring again to Figure 52-6, one can see that
rapidly acting neurotransmitter is released by the hair
the distal end of the basilar membrane at the heli-
cells at these synapses during depolarization. It is pos-
cotrema is stimulated by all sound frequencies below
sible that the transmitter substance is glutamate, but
200 cycles per second. Therefore, it has been difficult
this is not certain.
to understand from the place principle how one can
differentiate between low sound frequencies in the
Endocochlear Potential. To explain even more fully the
range from 200 down to 20. It is postulated that these
electrical potentials generated by the hair cells, we need
low frequencies are discriminated mainly by the so-
to explain another electrical phenomenon called the
called volley or frequency principle. That is, low-
endocochlear potential: The scala media is filled with a
frequency sounds, from 20 to 1500 to 2000 cycles per
fluid called endolymph, in contradistinction to the peri-
second, can cause volleys of nerve impulses synchro-
lymph present in the scala vestibuli and scala tympani.
nized at the same frequencies, and these volleys are
The scala vestibuli and scala tympani communicate
transmitted by the cochlear nerve into the cochlear
directly with the subarachnoid space around the brain,
so that the perilymph is almost identical with cere-
nuclei of the brain. It is further suggested that the
brospinal fluid. Conversely, the endolymph that fills
cochlear nuclei can distinguish the different frequen-
the scala media is an entirely different fluid secreted by
cies of the volleys. In fact, destruction of the entire
the stria vascularis, a highly vascular area on the outer
apical half of the cochlea, which destroys the basilar
wall of the scala media. Endolymph contains a high
membrane where all lower-frequency sounds are nor-
concentration of potassium and a low concentration
mally detected, does not totally eliminate discrimina-
of sodium, which is exactly opposite to the contents of
tion of the lower-frequency sounds.
perilymph.
An electrical potential of about +80 millivolts exists
all the time between endolymph and perilymph, with
Determination of Loudness
positivity inside the scala media and negativity outside.
This is called the endocochlear potential, and it is gen-
Loudness is determined by the auditory system in at
erated by continual secretion of positive potassium ions
least three ways.
into the scala media by the stria vascularis.
First, as the sound becomes louder, the amplitude of
The importance of the endocochlear potential is that
the tops of the hair cells project through the reticular
vibration of the basilar membrane and hair cells also
lamina and are bathed by the endolymph of the scala
increases, so that the hair cells excite the nerve endings
media, whereas perilymph bathes the lower bodies of
at more rapid rates.
Chapter 52
The Sense of Hearing
657
Second, as the amplitude of vibration increases, it
causes more and more of the hair cells on the fringes
of the resonating portion of the basilar membrane to
Vibration
Sound
become stimulated, thus causing spatial summation of
100
Pricking
impulses—that is, transmission through many nerve
80
(in middle ear)
fibers rather than through only a few.
60
Third, the outer hair cells do not become stimulated
40
Tactual
significantly until vibration of the basilar membrane
threshold
20
reaches high intensity, and stimulation of these cells
0
presumably apprises the nervous system that the
Threshold
–20
of hearing
sound is loud.
-40
-60
Reference
Detection of Changes in Loudness—The Power Law. As
- 80
pressure = -73.8
pointed out in Chapter 46, a person interprets changes
in intensity of sensory stimuli approximately in pro-
1
2
5 10 20
100
500
2000
10,000
Frequency
portion to an inverse power function of the actual
intensity. In the case of sound, the interpreted sensa-
tion changes approximately in proportion to the cube
root of the actual sound intensity. To express this
Figure 52-9
another way, the ear can discriminate differences in
Relation of the threshold of hearing and of somesthetic percep-
sound intensity from the softest whisper to the loudest
tion (pricking and tactual threshold) to the sound energy level at
possible noise, representing an approximately 1 trillion
each sound frequency.
times increase in sound energy or 1 million times
increase in amplitude of movement of the basilar
membrane. Yet the ear interprets this much difference
in sound level as approximately a 10,000-fold change.
to 5000 cycles per second; only with intense sounds can
Thus, the scale of intensity is greatly “compressed” by
the complete range of 20 to 20,000 cycles be achieved.
the sound perception mechanisms of the auditory
In old age, this frequency range is usually shortened to
system. This allows a person to interpret differences in
50 to 8000 cycles per second or less, as discussed later in
sound intensities over an extremely wide range—a far
the chapter.
wider range than would be possible were it not for
compression of the intensity scale.
Central Auditory Mechanisms
Decibel Unit. Because of the extreme changes in sound
Auditory Nervous Pathways
intensities that the ear can detect and discriminate,
sound intensities are usually expressed in terms of the
Figure 52-10 shows the major auditory pathways. It
logarithm of their actual intensities. A 10-fold increase
shows that nerve fibers from the spiral ganglion of
in sound energy is called 1 bel, and 0.1 bel is called 1
Corti enter the dorsal and ventral cochlear nuclei
decibel. One decibel represents an actual increase in
located in the upper part of the medulla. At this point,
sound energy of 1.26 times.
all the fibers synapse, and second-order neurons pass
Another reason for using the decibel system to
mainly to the opposite side of the brain stem to ter-
express changes in loudness is that, in the usual sound
minate in the superior olivary nucleus. A few second-
intensity range for communication, the ears can barely
order fibers also pass to the superior olivary nucleus
distinguish an approximately
1-decibel change in
on the same side. From the superior olivary nucleus,
sound intensity.
the auditory pathway passes upward through the
lateral lemniscus. Some of the fibers terminate in the
Threshold for Hearing Sound at Different Frequencies. Figure
52-9 shows the pressure thresholds at which sounds of
nucleus of the lateral lemniscus, but many bypass this
different frequencies can barely be heard by the ear.
nucleus and travel on to the inferior colliculus, where
This figure demonstrates that a 3000-cycle-per-second
all or almost all the auditory fibers synapse. From
sound can be heard even when its intensity is as low as
there, the pathway passes to the medial geniculate
70 decibels below 1 dyne/cm2 sound pressure level,
nucleus, where all the fibers do synapse. Finally, the
which is one ten-millionth microwatt per square cen-
pathway proceeds by way of the auditory radiation to
timeter. Conversely, a 100-cycle-per-second sound can
the auditory cortex, located mainly in the superior
be detected only if its intensity is 10,000 times as great
gyrus of the temporal lobe.
as this.
Several important points should be noted. First,
signals from both ears are transmitted through the
Frequency Range of Hearing. The frequencies of sound that
pathways of both sides of the brain, with a prepon-
a young person can hear are between 20 and 20,000
cycles per second. However, referring again to Figure
derance of transmission in the contralateral pathway.
52-9, we see that the sound range depends to a great
In at least three places in the brain stem, crossing over
extent on loudness. If the loudness is 60 decibels below
occurs between the two pathways: (1) in the trapezoid
1 dyne/cm2 sound pressure level, the sound range is 500
body, (2) in the commissure between the two nuclei of
658
Unit X
The Nervous System: B. The Special Senses
vermis of the cerebellum, which is also activated instan-
taneously in the event of a sudden noise.
Third, a high degree of spatial orientation is main-
tained in the fiber tracts from the cochlea all the way
to the cortex. In fact, there are three spatial patterns for
termination of the different sound frequencies in the
cochlear nuclei, two patterns in the inferior colliculi,
one precise pattern for discrete sound frequencies in
the auditory cortex, and at least five other less precise
patterns in the auditory cortex and auditory associa-
tion areas.
Firing Rates at Different Levels of the Auditory Pathways. Single
Primary
nerve fibers entering the cochlear nuclei from the audi-
auditry
cortex
tory nerve can fire at rates up to at least 1000 per
second, the rate being determined mainly by the loud-
Midbrain
Medial
ness of the sound. At sound frequencies up to 2000 to
geniculate
4000 cycles per second, the auditory nerve impulses are
nucleus
often synchronized with the sound waves, but they do
not necessarily occur with every wave.
In the auditory tracts of the brain stem, the firing is
Inferior
usually no longer synchronized with the sound fre-
colliculus
quency, except at sound frequencies below 200 cycles
Midbrain
per second. Above the level of the inferior colliculi, even
this synchronization is mainly lost. These findings
demonstrate that the sound signals are not transmitted
unchanged directly from the ear to the higher levels of
the brain; instead, information from the sound signals
begins to be dissected from the impulse traffic at levels
Nucleus of
as low as the cochlear nuclei. We will have more to say
the lateral
about this later, especially in relation to perception of
Pons
lemniscus
direction from which sound comes.
Function of the Cerebral Cortex
in Hearing
Pons
The projection area of auditory signals to the cerebral
Superior
cortex is shown in Figure 52-11, which demonstrates
olivary
Dorsal
nuclei
that the auditory cortex lies principally on the
acoustic
supratemporal plane of the superior temporal gyrus but
stria
Intermediate
also extends onto the lateral side of the temporal lobe,
acoustic
Cochlear
over much of the insular cortex, and even onto the
site
nuclei
lateral portion of the parietal operculum.
Medulla
Two separate subdivisions are shown in Figure
N. VIlI
52-11: the primary auditory cortex and the auditory
Trapezoid
body
association cortex (also called the secondary auditory
cortex). The primary auditory cortex is directly excited
by projections from the medial geniculate body,
Figure 52-10
whereas the auditory association areas are excited
secondarily by impulses from the primary auditory
Auditory nervous pathways. (Modified from Brodal A: The auditory
cortex as well as by some projections from thalamic
system. In Neurological Anatomy in Relation to Clinical Medicine,
association areas adjacent to the medial geniculate
3rd ed. New York: Oxford University Press, 1981.)
body.
Sound Frequency Perception in the Primary Auditory Cortex.
the lateral lemnisci, and (3) in the commissure con-
At least six tonotopic maps have been found in the
necting the two inferior colliculi.
primary auditory cortex and auditory association
Second, many collateral fibers from the auditory
areas. In each of these maps, high-frequency sounds
tracts pass directly into the reticular activating system
excite neurons at one end of the map, whereas low-
of the brain stem. This system projects diffusely
frequency sounds excite neurons at the opposite end.
upward in the brain stem and downward into the
In most, the low-frequency sounds are located anteri-
spinal cord and activates the entire nervous system in
orly, as shown in Figure 52-11, and the high-frequency
response to loud sounds. Other collaterals go to the
sounds are located posteriorly. This is not true for all
Chapter 52
The Sense of Hearing
659
in Chapter 46 in relation to mechanisms for transmit-
Low
High
ting information in nerves. That is, stimulation of the
frequency
frequency
cochlea at one frequency inhibits sound frequencies
on both sides of this primary frequency; this is
caused by collateral fibers angling off the primary
signal pathway and exerting inhibitory influences on
adjacent pathways. The same effect has been demon-
strated to be important in sharpening patterns of
somesthetic images, visual images, and other types of
sensations.
Many of the neurons in the auditory cortex, espe-
Association
Primary
cially in the auditory association cortex, do not respond
only to specific sound frequencies in the ear. It is
believed that these neurons
“associate” different
sound frequencies with one another or associate sound
information with information from other sensory
areas of the cortex. Indeed, the parietal portion of the
auditory association cortex partly overlaps somatosen-
sory area II, which could provide an easy opportunity
Association
for the association of auditory information with
somatosensory information.
Primary
Discrimination of Sound “Patterns” by the Auditory Cortex.
Complete bilateral removal of the auditory cortex
does not prevent a cat or monkey from detecting
sounds or reacting in a crude manner to sounds.
However, it does greatly reduce or sometimes
even abolish the animal’s ability to discriminate dif-
Figure 52-11
ferent sound pitches and especially patterns of sound.
For instance, an animal that has been trained to rec-
Auditory cortex.
ognize a combination or sequence of tones, one fol-
lowing the other in a particular pattern, loses this
ability when the auditory cortex is destroyed; further-
more, the animal cannot relearn this type of response.
the maps. The question one must ask is, Why does the
Therefore, the auditory cortex is especially important
auditory cortex have so many different tonotopic
in the discrimination of tonal and sequential sound
maps? The answer, presumably, is that each of the sep-
patterns.
arate areas dissects out some specific feature of the
Destruction of both primary auditory cortices in
sounds. For instance, one of the large maps in the
the human being greatly reduces one’s sensitivity
primary auditory cortex almost certainly discriminates
for hearing. Destruction of one side only slightly
the sound frequencies themselves and gives the person
reduces hearing in the opposite ear; it does not cause
the psychic sensation of sound pitches. Another map
deafness in the ear because of many crossover con-
is probably used to detect the direction from which the
nections from side to side in the auditory neural
sound comes. Other auditory cortex areas detect
pathway. However, it does affect one’s ability to
special qualities, such as the sudden onset of sounds,
localize the source of a sound, because comparative
or perhaps special modulations, such as noise versus
signals in both cortices are required for the localiza-
pure frequency sounds.
tion function.
The frequency range to which each individual
Lesions that affect the auditory association areas
neuron in the auditory cortex responds is much
but not the primary auditory cortex do not decrease a
narrower than that in the cochlear and brain stem
person’s ability to hear and differentiate sound tones
relay nuclei. Referring back to Figure 52-6B, note that
or even to interpret at least simple patterns of sound.
the basilar membrane near the base of the cochlea is
However, he or she is often unable to interpret the
stimulated by sounds of all frequencies, and in the
meaning of the sound heard. For instance, lesions in
cochlear nuclei, this same breadth of sound represen-
the posterior portion of the superior temporal gyrus,
tation is found. Yet, by the time the excitation has
which is called Wernicke’s area and is part of the audi-
reached the cerebral cortex, most sound-responsive
tory association cortex, often make it impossible for a
neurons respond to only a narrow range of frequen-
person to interpret the meanings of words even though
cies rather than to a broad range. Therefore, some-
he or she hears them perfectly well and can even
where along the pathway, processing mechanisms
repeat them. These functions of the auditory associa-
“sharpen” the frequency response. It is believed that
tion areas and their relation to the overall intellectual
this sharpening effect is caused mainly by the phe-
functions of the brain are discussed in more detail in
nomenon of lateral inhibition, which is discussed
Chapter 57.
660
Unit X The Nervous System: B. The Special Senses
Determination of the Direction from
lag; those in between respond to intermediate time
lags. Thus, a spatial pattern of neuronal stimulation
Which Sound Comes
develops in the medial superior olivary nucleus, with
sound from directly in front of the head stimulating
A person determines the horizontal direction from
one set of olivary neurons maximally and sounds from
which sound comes by two principal means: (1) the
different side angles stimulating other sets of neurons
time lag between the entry of sound into one ear and
on opposite sides. This spatial orientation of signals is
its entry into the opposite ear, and (2) the difference
then transmitted to the auditory cortex, where sound
between the intensities of the sounds in the two ears.
direction is determined by the locus of the maximally
The first mechanism functions best at frequencies
stimulated neurons. It is believed that all these signals
below 3000 cycles per second, and the second mecha-
for determining sound direction are transmitted
nism operates best at higher frequencies because the
through a different pathway and excite a different
head is a greater sound barrier at these frequencies.
locus in the cerebral cortex from the transmission
The time lag mechanism discriminates direction much
pathway and termination locus for tonal patterns of
more exactly than the intensity mechanism because it
sound.
does not depend on extraneous factors but only on the
This mechanism for detection of sound direction
exact interval of time between two acoustical signals.
indicates again how specific information in sensory
If a person is looking straight toward the source of the
signals is dissected out as the signals pass through
sound, the sound reaches both ears at exactly the same
different levels of neuronal activity. In this case, the
instant, whereas if the right ear is closer to the sound
“quality” of sound direction is separated from the
than the left ear is, the sound signals from the right ear
“quality” of sound tones at the level of the superior
enter the brain ahead of those from the left ear.
olivary nuclei.
The two aforementioned mechanisms cannot tell
whether the sound is emanating from in front of or
behind the person or from above or below. This dis-
Centrifugal Signals from the
crimination is achieved mainly by the pinnae of the
Central Nervous System to Lower
two ears. The shape of the pinna changes the quality
Auditory Centers
of the sound entering the ear, depending on the direc-
tion from which the sound comes. It does this by
Retrograde pathways have been demonstrated at each
emphasizing specific sound frequencies from the dif-
level of the auditory nervous system from the cortex to
ferent directions.
the cochlea in the ear itself. The final pathway is mainly
from the superior olivary nucleus to the sound-receptor
Neural Mechanisms for Detecting Sound Direction. Destruc-
hair cells in the organ of Corti.
tion of the auditory cortex on both sides of the brain,
These retrograde fibers are inhibitory. Indeed, direct
whether in human beings or in lower mammals, causes
stimulation of discrete points in the olivary nucleus has
loss of almost all ability to detect the direction from
been shown to inhibit specific areas of the organ of
Corti, reducing their sound sensitivities 15 to 20 deci-
which sound comes. Yet the neural analyses for this
bels. One can readily understand how this could allow
detection process begin in the superior olivary nuclei
a person to direct his or her attention to sounds of par-
in the brain stem, even though the neural pathways all
ticular qualities while rejecting sounds of other quali-
the way from these nuclei to the cortex are required
ties. This is readily demonstrated when one listens to a
for interpretation of the signals. The mechanism is
single instrument in a symphony orchestra.
believed to be the following.
The superior olivary nucleus is divided into two sec-
tions: (1) the medial superior olivary nucleus and (2)
Hearing Abnormalities
the lateral superior olivary nucleus. The lateral nucleus
is concerned with detecting the direction from which
Types of Deafness
the sound is coming, presumably by simply comparing
Deafness is usually divided into two types: (1) that
the difference in intensities of the sound reaching the
caused by impairment of the cochlea or impairment of
two ears and sending an appropriate signal to the audi-
the auditory nerve, which is usually classified as “nerve
tory cortex to estimate the direction.
deafness,” and (2) that caused by impairment of the
The medial superior olivary nucleus, however, has a
physical structures of the ear that conduct sound
specific mechanism for detecting the time lag between
itself to the cochlea, which is usually called “conduction
acoustical signals entering the two ears. This nucleus
deafness.”
contains large numbers of neurons that have two
If either the cochlea or the auditory nerve is
destroyed, the person becomes permanently deaf.
major dendrites, one projecting to the right and the
However, if the cochlea and nerve are still intact but the
other to the left. The acoustical signal from the right
tympanum-ossicular system has been destroyed or
ear impinges on the right dendrite, and the signal from
ankylosed (“frozen” in place by fibrosis or calcification),
the left ear impinges on the left dendrite. The intensity
sound waves can still be conducted into the cochlea by
of excitation of each neuron is highly sensitive to a
means of bone conduction from a sound generator
specific time lag between the two acoustical signals
applied to the skull over the ear.
from the two ears. The neurons near one border of the
nucleus respond maximally to a short time lag, while
Audiometer. To determine the nature of hearing dis-
those near the opposite border respond to a long time
abilities, the “audiometer” is used. Simply an earphone
Chapter 52
The Sense of Hearing
661
-10
-10
Normal
Normal
10
X
10
*
*
*
*
*
*
20
20
* X
X
30
30
*
X
X
40
X
40
X
X
X
X
50
50
60
60
70
70
X Air conduction
X Air conduction
80
80
90
* Bone conduction
90
* Bone conduction
100
100
1
250
500
1000
2000
4000
8000
125
250
500
1000
2000
4000
8000
Frequency
Frequency
Figure 52-12
Figure 52-13
Audiogram of the old-age type of nerve deafness.
Audiogram of air conduction deafness resulting from middle ear
sclerosis.
connected to an electronic oscillator capable of emitting
pure tones ranging from low frequencies to high fre-
Audiogram for Middle Ear Conduction Deafness. A
quencies, the instrument is calibrated so that zero-
common type of deafness is caused by fibrosis in the
intensity-level sound at each frequency is the loudness
middle ear following repeated infection or by fibrosis
that can barely be heard by the normal ear. A calibrated
that occurs in the hereditary disease called otosclerosis.
volume control can increase the loudness above the
In either case, the sound waves cannot be transmitted
zero level. If the loudness must be increased to 30 deci-
easily through the ossicles from the tympanic mem-
bels above normal before it can be heard, the person is
brane to the oval window. Figure 52-13 shows an audio-
said to have a hearing loss of 30 decibels at that partic-
gram from a person with “middle ear air conduction
ular frequency.
deafness.” In this case, bone conduction is essentially
In performing a hearing test using an audiometer, one
normal, but conduction through the ossicular system is
tests about 8 to 10 frequencies covering the auditory
greatly depressed at all frequencies, but more so at low
spectrum, and the hearing loss is determined for each of
frequencies. In some instances of conduction deafness,
these frequencies. Then the so-called audiogram is
the faceplate of the stapes becomes “ankylosed” by
plotted, as shown in Figures 52-12 and 52-13, depicting
bone overgrowth to the edges of the oval window. In
hearing loss at each of the frequencies in the auditory
this case, the person becomes totally deaf for ossicular
spectrum. The audiometer, in addition to being
conduction but can regain almost normal hearing by the
equipped with an earphone for testing air conduction
surgical removal of the stapes and its replacement with
by the ear, is equipped with a mechanical vibrator for
a minute Teflon or metal prosthesis that transmits the
testing bone conduction from the mastoid process of the
sound from the incus to the oval window.
skull into the cochlea.
Audiogram in Nerve Deafness. In nerve deafness—
References
which includes damage to the cochlea, the auditory
nerve, or the central nervous system circuits from the
ear—the person has decreased or total loss of ability to
Brugge J: Central Auditory Processing and Neural Model-
hear sound as tested by both air conduction and bone
ing. New York: Plenum Press, 1998.
conduction. An audiogram depicting partial nerve deaf-
Eatock RA: Auditory physiology: listening with K+ channels.
ness is shown in Figure 52-12. In this figure, the deaf-
Curr Biol 13:R767, 2003.
ness is mainly for high-frequency sound. Such deafness
Fettiplace R, Ricci AJ: Adaptation in auditory hair cells. Curr
could be caused by damage to the base of the cochlea.
Opin Neurobiol 13:446, 2003.
This type of deafness occurs to some extent in almost
Frolenkov GI, Belyantseva IA, Friedman TB, Griffith AJ:
all older people.
Genetic insights into the morphogenesis of inner ear hair
Other patterns of nerve deafness frequently occur as
cells. Nat Rev Genet 5:489, 2004.
follows: (1) deafness for low-frequency sounds caused
Geisler CD: From Sound to Synapse: Physiology of the
by excessive and prolonged exposure to very loud
Mammalian Ear. New York: Oxford University Press,
sounds (a rock band or a jet airplane engine), because
1998.
low-frequency sounds are usually louder and more dam-
Gillespie PG, Cyr JL: Myosin-1c, the hair cell’s adaptation
aging to the organ of Corti, and (2) deafness for all
motor. Annu Rev Physiol 66:521, 2004.
frequencies caused by drug sensitivity of the organ of
Griffiths TD, Warren JD, Scott SK, et al: Cortical processing
Corti—in particular, sensitivity to some antibiotics such
of complex sound: a way forward? Trends Neurosci 27:181,
as streptomycin, kanamycin, and chloramphenicol.
2004.
662
Unit X The Nervous System: B. The Special Senses
Grothe B: New roles for synaptic inhibition in sound local-
Scott SK, Johnsrude IS: The neuroanatomical and functional
ization. Nat Rev Neurosci 4:540, 2003.
organization of speech perception. Trends Neurosci 26:
Joris PX, Schreiner CE, Rees A: Neural processing of ampli-
100, 2003.
tude-modulated sounds. Physiol Rev 84:541, 2004.
Semple MN, Scott BH: Cortical mechanisms in hearing. Curr
McDermott BM Jr, Lopez-Schier H: Inner ear: Ca2+ you feel
Opin Neurobiol 13:167, 2003.
the noise? Curr Biol 14:R231, 2004.
Suga N, Ma X: Multiparametric corticofugal modulation and
Minor LB, Schessel DA, Carey JP: Meniere’s disease. Curr
plasticity in the auditory system. Nat Rev Neurosci 4:783,
Opin Neurol 17:9, 2004.
2003.
Rauschecker JP, Shannon RV: Sending sound to the brain.
Syka J: Plastic changes in the central auditory system after
Science 295:1025, 2002.
hearing loss, restoration of function, and during learning.
Read HL, Winer JA, Schreiner CE: Functional architecture
Physiol Rev 82:601, 2002.
of auditory cortex. Curr Opin Neurobiol 12:433, 2002.
Trainor LJ, Shahin A, Roberts LE: Effects of musical train-
Robles L, Ruggero MA: Mechanics of the mammalian
ing on the auditory cortex in children. Ann N Y Acad Sci
cochlea. Physiol Rev 81:1305, 2001.
999:506, 2003.
Santos-Sacchi J: New tunes from Corti’s organ: the outer hair
Weinberger NM: Specific long-term memory traces in
cell boogie rules. Curr Opin Neurobiol 13:459, 2003.
primary auditory cortex. Nat Rev Neurosci 5:279, 2004.
Schreiner CE, Read HL, Sutter ML: Modular organization
of frequency integration in primary auditory cortex. Annu
Rev Neurosci 23:501, 2000.
C H A P T E R
5
3
The Chemical Senses—
Taste and Smell
The senses of taste and smell allow us to separate
undesirable or even lethal foods from those that are
pleasant to eat and nutritious. The sense of smell
also allows animals to recognize the proximity of
other animals or even individuals among animals.
Finally, both senses are strongly tied to primitive
emotional and behavioral functions of our nervous
systems.
Sense of Taste
Taste is mainly a function of the taste buds in the mouth, but it is common expe-
rience that one’s sense of smell also contributes strongly to taste perception. In
addition, the texture of food, as detected by tactual senses of the mouth, and
the presence of substances in the food that stimulate pain endings, such as
pepper, greatly alter the taste experience. The importance of taste lies in the
fact that it allows a person to select food in accord with desires and often in
accord with the body tissues’ metabolic need for specific substances.
Primary Sensations of Taste
The identities of the specific chemicals that excite different taste receptors are
not all known. Even so, psychophysiologic and neurophysiologic studies have
identified at least 13 possible or probable chemical receptors in the taste cells,
as follows: 2 sodium receptors, 2 potassium receptors, 1 chloride receptor,
1 adenosine receptor, 1 inosine receptor, 2 sweet receptors, 2 bitter receptors,
1 glutamate receptor, and 1 hydrogen ion receptor.
For practical analysis of taste, the aforementioned receptor capabilities have
also been grouped into five general categories called the primary sensations of
taste. They are sour, salty, sweet, bitter, and “umami.”
A person can perceive hundreds of different tastes. They are all supposed to
be combinations of the elementary taste sensations, just as all the colors we can
see are combinations of the three primary colors, as described in Chapter 50.
Sour Taste. The sour taste is caused by acids, that is, by the hydrogen ion con-
centration, and the intensity of this taste sensation is approximately propor-
tional to the logarithm of the hydrogen ion concentration. That is, the more acidic
the food, the stronger the sour sensation becomes.
Salty Taste. The salty taste is elicited by ionized salts, mainly by the sodium ion
concentration. The quality of the taste varies somewhat from one salt to
another, because some salts elicit other taste sensations in addition to saltiness.
The cations of the salts, especially sodium cations, are mainly responsible for
the salty taste, but the anions also contribute to a lesser extent.
Sweet Taste. The sweet taste is not caused by any single class of chemicals. Some
of the types of chemicals that cause this taste include sugars, glycols, alcohols,
aldehydes, ketones, amides, esters, some amino acids, some small proteins,
663
664
Unit X The Nervous System: B. The Special Senses
Table 53-1
Relative Taste Indices of Different Substances
Sour Substances
Index
Bitter Substances
Index
Sweet Substances
Index
Salty Substances
Index
Hydrochloric acid
1
Quinine
1
Sucrose
1
NaCl
1
Formic acid
1.1
Brucine
11
1-Propoxy-2-amino-
5000
NaF
2
Chloracetic acid
0.9
Strychnine
3.1
4-nitrobenzene
CaCl2
1
Acetylacetic acid
0.85
Nicotine
1.3
Saccharin
675
NaBr
0.4
Lactic acid
0.85
Phenylthiourea
0.9
Chloroform
40
NaI
0.35
Tartaric acid
0.7
Caffeine
0.4
Fructose
1.7
LiCl
0.4
Malic acid
0.6
Veratrine
0.2
Alanine
1.3
NH4Cl
2.5
Potassium H tartrate
0.58
Pilocarpine
0.16
Glucose
0.8
KCl
0.6
Acetic acid
0.55
Atropine
0.13
Maltose
0.45
Citric acid
0.46
Cocaine
0.02
Galactose
0.32
Carbonic acid
0.06
Morphine
0.02
Lactose
0.3
From Pfaffman C: Handbook of Physiology. Sec I, Vol 1. Baltimore: Williams & Wilkins, 1959, p. 507.
sulfonic acids, halogenated acids, and inorganic salts of
Threshold for Taste
lead and beryllium. Note specifically that most of the
The threshold for stimulation of the sour taste by
substances that cause a sweet taste are organic chem-
hydrochloric acid averages 0.0009 N; for stimulation of
icals. It is especially interesting that slight changes in
the salty taste by sodium chloride, 0.01 M; for the sweet
the chemical structure, such as addition of a simple
taste by sucrose, 0.01 M; and for the bitter taste by
radical, can often change the substance from sweet to
quinine, 0.000008 M. Note especially how much more
bitter.
sensitive is the bitter taste sense than all the others,
which would be expected, because this sensation pro-
Bitter Taste. The bitter taste, like the sweet taste, is not
vides an important protective function against many
caused by any single type of chemical agent. Here
dangerous toxins in food.
again, the substances that give the bitter taste are
Table 53-1 gives the relative taste indices (the recip-
almost entirely organic substances. Two particular
rocals of the taste thresholds) of different substances.
classes of substances are especially likely to cause
In this table, the intensities of four of the primary sen-
bitter taste sensations: (1) long-chain organic sub-
sations of taste are referred, respectively, to the inten-
stances that contain nitrogen, and (2) alkaloids. The
sities of the taste of hydrochloric acid, quinine, sucrose,
alkaloids include many of the drugs used in medicines,
and sodium chloride, each of which is arbitrarily
such as quinine, caffeine, strychnine, and nicotine.
chosen to have a taste index of 1.
Some substances that at first taste sweet have a
bitter aftertaste. This is true of saccharin, which makes
Taste Blindness. Some people are taste blind for certain
this substance objectionable to some people.
substances, especially for different types of thiourea
The bitter taste, when it occurs in high intensity,
compounds. A substance used frequently by psycholo-
usually causes the person or animal to reject the food.
gists for demonstrating taste blindness is phenylthio-
This is undoubtedly an important function of the bitter
carbamide, for which about 15 to 30 per cent of all
taste sensation, because many deadly toxins found in
people exhibit taste blindness; the exact percentage
poisonous plants are alkaloids, and virtually all of
depends on the method of testing and the concentra-
these cause intensely bitter taste, usually followed by
tion of the substance.
rejection of the food.
Umami Taste. Umami is a Japanese word (meaning
Taste Bud and Its Function
“delicious”) designating a pleasant taste sensation that
is qualitatively different from sour, salty, sweet, or
Figure 53-1 shows a taste bud, which has a diameter
bitter. Umami is the dominant taste of food contain-
of about 1/30 millimeter and a length of about 1/16 mil-
ing L-glutamate, such as meat extracts and aging
limeter. The taste bud is composed of about 50 modi-
cheese, and some physiologists consider it to be a sep-
fied epithelial cells, some of which are supporting cells
arate, fifth category of primary taste stimuli.
called sustentacular cells and others of which are taste
A taste receptor for L-glutamate may be related to
cells. The taste cells are continually being replaced by
one of the glutamate receptors expressed in neuronal
mitotic division of surrounding epithelial cells, so that
synapses of the brain. However, the precise molecular
some taste cells are young cells. Others are mature
mechanisms responsible for umami taste are still
cells that lie toward the center of the bud; these soon
unclear.
break up and dissolve. The life span of each taste cell
Chapter 53
The Chemical Senses—Taste and Smell
665
Stratified
stimuli, as well as by a few other taste stimuli that do
squamous
not fit into the “primary” categories.
epithelium
Mechanism of Stimulation of Taste Buds
Nerve fibers
Receptor Potential. The membrane of the taste cell,
Microvilli
like that of most other sensory receptor cells, is nega-
tively charged on the inside with respect to the outside.
Pore
Application of a taste substance to the taste hairs
causes partial loss of this negative potential—that is,
the taste cell becomes depolarized. In most instances,
the decrease in potential, within a wide range, is
Taste cells
approximately proportional to the logarithm of con-
centration of the stimulating substance. This change in
Subepithelial
electrical potential in the taste cell is called the recep-
connective
tor potential for taste.
tissue
The mechanism by which most stimulating sub-
stances react with the taste villi to initiate the recep-
Figure 53-1
tor potential is by binding of the taste chemical to a
Taste bud.
protein receptor molecule that lies on the outer
surface of the taste receptor cell near to or protruding
through a villus membrane. This, in turn, opens ion
is about 10 days in lower mammals but is unknown for
channels, which allows positively charged sodium ions
humans.
or hydrogen ions to enter and depolarize the normal
The outer tips of the taste cells are arranged around
negativity of the cell. Then the taste chemical itself is
a minute taste pore, shown in Figure 53-1. From the tip
gradually washed away from the taste villus by the
of each taste cell, several microvilli, or taste hairs, pro-
saliva, which removes the stimulus.
trude outward into the taste pore to approach the
The type of receptor protein in each taste villus
cavity of the mouth. These microvilli provide the
determines the type of taste that will be perceived.
receptor surface for taste.
For sodium ions and hydrogen ions, which elicit salty
Interwoven around the bodies of the taste cells is a
and sour taste sensations, respectively, the receptor
branching terminal network of taste nerve fibers that
proteins open specific ion channels in the apical mem-
are stimulated by the taste receptor cells. Some of
branes of the taste cells, thereby activating the recep-
these fibers invaginate into folds of the taste cell mem-
tors. However, for the sweet and bitter taste sensations,
branes. Many vesicles form beneath the cell membrane
the portions of the receptor protein molecules that
near the fibers. It is believed that these vesicles contain
protrude through the apical membranes activate
a neurotransmitter substance that is released through
second-messenger transmitter substances inside the
the cell membrane to excite the nerve fiber endings in
taste cells, and these second messengers cause intra-
response to taste stimulation.
cellular chemical changes that elicit the taste signals.
Location of the Taste Buds. The taste buds are found on
Generation of Nerve Impulses by the Taste Bud. On
three types of papillae of the tongue, as follows: (1)
first application of the taste stimulus, the rate of dis-
A large number of taste buds are on the walls of
charge of the nerve fibers from taste buds rises to a
the troughs that surround the circumvallate papillae,
peak in a small fraction of a second but then adapts
which form a V line on the surface of the posterior
within the next few seconds back to a lower, steady
tongue. (2) Moderate numbers of taste buds are on the
level as long as the taste stimulus remains. Thus, a
fungiform papillae over the flat anterior surface of the
strong immediate signal is transmitted by the taste
tongue. (3) Moderate numbers are on the foliate papil-
nerve, and a weaker continuous signal is transmitted
lae located in the folds along the lateral surfaces of
as long as the taste bud is exposed to the taste
the tongue. Additional taste buds are located on the
stimulus.
palate, and a few are found on the tonsillar pillars, on
the epiglottis, and even in the proximal esophagus.
Adults have 3000 to 10,000 taste buds, and children
Transmission of Taste Signals into the
have a few more. Beyond the age of 45 years, many
Central Nervous System
taste buds degenerate, causing the taste sensation to
become progressively less critical in old age.
Figure 53-2 shows the neuronal pathways for trans-
mission of taste signals from the tongue and pharyn-
Specificity of Taste Buds for a Primary Taste Stimulus. Micro-
geal region into the central nervous system. Taste
electrode studies from single taste buds show that each
impulses from the anterior two thirds of the tongue
taste bud usually responds mostly to one of the five
pass first into the lingual nerve, then through the
primary taste stimuli when the taste substance is in low
chorda tympani into the facial nerve, and finally into
concentration. But at high concentration, most buds
the tractus solitarius in the brain stem. Taste sensations
can be excited by two or more of the primary taste
from the circumvallate papillae on the back of the
666
Unit X
The Nervous System: B. The Special Senses
glands to help control the secretion of saliva during the
Gustatory cortex
ingestion and digestion of food.
(anterior insula-
frontal operculum)
Adaptation of Taste. Everyone is familiar with the fact
that taste sensations adapt rapidly, often almost com-
pletely within a minute or so of continuous stimula-
tion. Yet, from electrophysiologic studies of taste nerve
fibers, it is clear that adaptation of the taste buds them-
selves usually accounts for no more than about half of
this. Therefore, the final extreme degree of adaptation
that occurs in the sensation of taste almost certainly
Ventral posterior
occurs in the central nervous system itself, although
medial nucleus of
thalamus
the mechanism and site of this are not known. At any
Geniculate
rate, it is a mechanism different from that of most
Chorda
ganglion
other sensory systems, which adapt almost entirely at
tympani
Tongue
the receptors.
N. VII
Nucleus of
Taste Preference and Control
N. IX
solitary tract
Glossopharyngeal
Petrosal
of the Diet
ganglion
Gustatory
N. X
area
Taste preference simply means that an animal will
choose certain types of food in preference to others,
Nodose
and the animal automatically uses this to help control
ganglion
the type of diet it eats. Furthermore, its taste prefer-
ences often change in accord with the body’s need for
Pharynx
certain specific substances.
The following experiments demonstrate this ability
of animals to choose food in accord with the needs
Figure 53-2
of their bodies. First, adrenalectomized, salt-depleted
Transmission of taste signals into the central nervous system.
animals automatically select drinking water with a
high concentration of sodium chloride in preference to
pure water, and this is often sufficient to supply the
needs of the body and prevent salt-depletion death.
tongue and from other posterior regions of the mouth
Second, an animal given injections of excessive
and throat are transmitted through the glossopharyn-
amounts of insulin develops a depleted blood sugar,
geal nerve also into the tractus solitarius, but at a
and the animal automatically chooses the sweetest
slightly more posterior level. Finally, a few taste signals
food from among many samples. Third, calcium-
are transmitted into the tractus solitarius from the base
depleted parathyroidectomized animals automatically
of the tongue and other parts of the pharyngeal region
choose drinking water with a high concentration of
by way of the vagus nerve.
calcium chloride.
All taste fibers synapse in the posterior brain stem
The same phenomena are also observed in everyday
in the nuclei of the tractus solitarius. These nuclei send
life. For instance, the “salt licks” of desert regions are
second-order neurons to a small area of the ventral
known to attract animals from far and wide. Also,
posterior medial nucleus of the thalamus, located
human beings reject any food that has an unpleasant
slightly medial to the thalamic terminations of the
affective sensation, which in many instances protects
facial regions of the dorsal column-medial lemniscal
our bodies from undesirable substances.
system. From the thalamus, third-order neurons are
The phenomenon of taste preference almost cer-
transmitted to the lower tip of the postcentral gyrus in
tainly results from some mechanism located in the
the parietal cerebral cortex, where it curls deep into the
central nervous system and not from a mechanism in
sylvian fissure, and into the adjacent opercular insular
the taste receptors themselves, although it is true that
area. This lies slightly lateral, ventral, and rostral to the
the receptors often become sensitized in favor of a
area for tongue tactile signals in cerebral somatic area
needed nutrient. An important reason for believing
I. From this description of the taste pathways, it is
that taste preference is mainly a central nervous
evident that they closely parallel the somatosensory
system phenomenon is that previous experience with
pathways from the tongue.
unpleasant or pleasant tastes plays a major role in
determining one’s taste preferences. For instance, if a
Taste Reflexes Integrated in the Brain Stem. From the
person becomes sick soon after eating a particular type
tractus solitarius, many taste signals are transmitted
of food, the person generally develops a negative taste
within the brain stem itself directly into the superior
preference, or taste aversion, for that particular food
and inferior salivatory nuclei, and these areas transmit
thereafter; the same effect can be demonstrated in
signals to the submandibular, sublingual, and parotid
lower animals.
Chapter 53
The Chemical Senses—Taste and Smell
667
Olfactory tract
membrane are many small Bowman’s glands that
secrete mucus onto the surface of the olfactory
Olfactory bulb
membrane.
Mitral cell
Stimulation of the Olfactory Cells
Bowman’s
Glomerulus
gland
Mechanism of Excitation of the Olfactory Cells. The portion
of each olfactory cell that responds to the olfactory
chemical stimuli is the olfactory cilia. The odorant sub-
stance, on coming in contact with the olfactory mem-
Sustentacular
brane surface, first diffuses into the mucus that covers
cells
the cilia. Then it binds with receptor proteins in the
Olfactory cell
membrane of each cilium. Each receptor protein is
actually a long molecule that threads its way through
the membrane about seven times, folding inward and
Olfactory cilia
outward. The odorant binds with the portion of the
receptor protein that folds to the outside. The inside
Mucus layer
of the folding protein, however, is coupled to a so-
Figure 53-3
called G-protein, itself a combination of three sub-
units. On excitation of the receptor protein, an alpha
Organization of the olfactory membrane and olfactory bulb, and
subunit breaks away from the G-protein and immedi-
connections to the olfactory tract.
ately activates adenylyl cyclase, which is attached to
the inside of the ciliary membrane near the receptor
cell body. The activated cyclase, in turn, converts many
Sense of Smell
molecules of intracellular adenosine triphosphate into
cyclic adenosine monophosphate (cAMP). Finally, this
Smell is the least understood of our senses. This results
cAMP activates another nearby membrane protein, a
partly from the fact that the sense of smell is a sub-
gated sodium ion channel, that opens its “gate” and
jective phenomenon that cannot be studied with ease
allows large numbers of sodium ions to pour through
in lower animals. Another complicating problem is
the membrane into the receptor cell cytoplasm. The
that the sense of smell is poorly developed in human
sodium ions increase the electrical potential in the pos-
beings in comparison with the sense of smell in many
itive direction inside the cell membrane, thus exciting
lower animals.
the olfactory neuron and transmitting action poten-
tials into the central nervous system by way of the
olfactory nerve.
Olfactory Membrane
The importance of this mechanism for activating
olfactory nerves is that it greatly multiplies the excita-
The olfactory membrane, the histology of which is
tory effect of even the weakest odorant. To summarize:
shown in Figure 53-3, lies in the superior part of each
(1) Activation of the receptor protein by the odorant
nostril. Medially, the olfactory membrane folds down-
substance activates the G-protein complex. (2) This, in
ward along the surface of the superior septum; later-
turn, activates multiple molecules of adenylyl cyclase
ally, it folds over the superior turbinate and even over
inside the olfactory cell membrane. (3) This causes the
a small portion of the upper surface of the middle
formation of many times more molecules of cAMP. (4)
turbinate. In each nostril, the olfactory membrane has
Finally, the cAMP opens still many times more sodium
a surface area of about 2.4 square centimeters.
ion channels. Therefore, even the most minute con-
centration of a specific odorant initiates a cascading
Olfactory Cells. The receptor cells for the smell sensa-
effect that opens extremely large numbers of sodium
tion are the olfactory cells (see Figure 53-3), which are
channels. This accounts for the exquisite sensitivity of
actually bipolar nerve cells derived originally from the
the olfactory neurons to even the slightest amount of
central nervous system itself. There are about 100
odorant.
million of these cells in the olfactory epithelium inter-
In addition to the basic chemical mechanism by
spersed among sustentacular cells, as shown in Figure
which the olfactory cells are stimulated, several phys-
53-3. The mucosal end of the olfactory cell forms a
ical factors affect the degree of stimulation. First, only
knob from which 4 to 25 olfactory hairs (also called
volatile substances that can be sniffed into the nostrils
olfactory cilia), measuring 0.3 micrometer in diameter
can be smelled. Second, the stimulating substance
and up to 200 micrometers in length, project into the
must be at least slightly water soluble so that it can
mucus that coats the inner surface of the nasal cavity.
pass through the mucus to reach the olfactory cilia.
These projecting olfactory cilia form a dense mat in
Third, it is helpful for the substance to be at least
the mucus, and it is these cilia that react to odors in
slightly lipid soluble, presumably because lipid con-
the air and stimulate the olfactory cells, as discussed
stituents of the cilium itself are a weak barrier to
later. Spaced among the olfactory cells in the olfactory
non-lipid-soluble odorants.
668
Unit X The Nervous System: B. The Special Senses
Membrane Potentials and Action Potentials in Olfactory Cells.
of at least 100 primary sensations of smell—a marked
The membrane potential inside unstimulated olfactory
contrast to only three primary sensations of color
cells, as measured by microelectrodes, averages about
detected by the eyes and only four or five primary
-55 millivolts. At this potential, most of the cells gen-
sensations of taste detected by the tongue. Further
erate continuous action potentials at a very slow rate,
support for the many primary sensations of smell is
varying from once every 20 seconds up to two or three
that people have been found who have odor blindness
per second.
for single substances; such discrete odor blindness has
Most odorants cause depolarization of the olfactory
been identified for more than 50 different substances.
cell membrane, decreasing the negative potential in
It is presumed that odor blindness for each substance
the cell from the normal level of -55 millivolts to -30
represents lack of the appropriate receptor protein in
millivolts or less—that is, changing the voltage in the
olfactory cells for that particular substance.
positive direction. Along with this, the number of
action potentials increases to 20 to 30 per second,
“Affective Nature of Smell.” Smell, even more so than
which is a high rate for the minute olfactory nerve
taste, has the affective quality of either pleasantness or
fibers.
unpleasantness. Because of this, smell is probably even
Over a wide range, the rate of olfactory nerve
more important than taste for the selection of food.
impulses changes approximately in proportion to the
Indeed, a person who has previously eaten food that
logarithm of the stimulus strength, which demon-
disagreed with him or her is often nauseated by the
strates that the olfactory receptors obey principles
smell of that same food on a second occasion.
of transduction similar to those of other sensory
Conversely, perfume of the right quality can wreak
receptors.
havoc with human emotions. In addition, in some
lower animals, odors are the primary excitant of sexual
Adaptation. The olfactory receptors adapt about 50 per
drive.
cent in the first second or so after stimulation. There-
after, they adapt very little and very slowly. Yet we all
Threshold for Smell. One of the principal characteristics
know from our own experience that smell sensations
of smell is the minute quantity of stimulating agent in
adapt almost to extinction within a minute or so after
the air that can elicit a smell sensation. For instance,
entering a strongly odorous atmosphere. Because this
the substance methylmercaptan can be smelled when
psychological adaptation is far greater than the degree
only one 25 trillionth of a gram is present in each mil-
of adaptation of the receptors themselves, it is almost
liliter of air. Because of this very low threshold, this
certain that most of the additional adaptation occurs
substance is mixed with natural gas to give the gas an
within the central nervous system. This seems to be
odor that can be detected when even small amounts
true for the adaptation of taste sensations as well.
of gas leak from a pipeline.
A postulated neuronal mechanism for the adapta-
tion is the following: Large numbers of centrifugal
Gradations of Smell Intensities. Although the threshold
nerve fibers pass from the olfactory regions of the
concentrations of substances that evoke smell are
brain backward along the olfactory tract and terminate
extremely slight, for many (if not most) odorants, con-
on special inhibitory cells in the olfactory bulb, the
centrations only 10 to 50 times above the threshold
granule cells. It has been postulated that after the onset
evoke maximum intensity of smell. This is in contrast
of an olfactory stimulus, the central nervous system
to most other sensory systems of the body, in which the
quickly develops strong feedback inhibition to sup-
ranges of intensity discrimination are tremendous—
press relay of the smell signals through the olfactory
for example, 500,000 to 1 in the case of the eyes and
bulb.
1 trillion to 1 in the case of the ears. This difference
might be explained by the fact that smell is concerned
Search for the Primary Sensations of Smell
more with detecting the presence or absence of
In the past, most physiologists were convinced that the
odors rather than with quantitative detection of their
many smell sensations are subserved by a few rather
intensities.
discrete primary sensations, in the same way that
vision and taste are subserved by a few select primary
sensations. Based on psychological studies, one at-
tempt to classify these sensations is the following:
Transmission of Smell Signals into
1. Camphoraceous
the Central Nervous System
2. Musky
3. Floral
The olfactory portions of the brain were among the
4. Pepperminty
first brain structures developed in primitive animals,
5. Ethereal
and much of the remainder of the brain developed
6. Pungent
around these olfactory beginnings. In fact, part of
7. Putrid
the brain that originally subserved olfaction later
It is certain that this list does not represent the true
evolved into the basal brain structures that control
primary sensations of smell. In recent years, multiple
emotions and other aspects of human behavior; this is
clues, including specific studies of the genes that
the system we call the limbic system, discussed in
encode for the receptor proteins, suggest the existence
Chapter 58.
Chapter 53
The Chemical Senses—Taste and Smell
669
Transmission of Olfactory Signals into the Olfactory Bulb. The
Hypothalamus
Medial olfactory area
olfactory bulb is shown in Figure 53-4. The olfactory
nerve fibers leading backward from the bulb are called
Prefrontal
cranial nerve I, or the olfactory tract. However,
cortex
in reality, both the tract and the bulb are an anterior
Olfactory
outgrowth of brain tissue from the base of the brain;
tract
the bulbous enlargement at its end, the olfactory bulb,
lies over the cribriform plate, separating the brain
Mitral
cavity from the upper reaches of the nasal cavity.
cell
The cribriform plate has multiple small perforations
through which an equal number of small nerves
Olfactory
pass upward from the olfactory membrane in the
bulb
nasal cavity to enter the olfactory bulb in the cranial
Orbito-
Olfactory
cavity. Figure 53-3 demonstrates the close relation
frontal
nerve
between the olfactory cells in the olfactory membrane
Lateral
cortex
and the olfactory bulb, showing short axons from
olfactory
area
the olfactory cells terminating in multiple globular
Temporal
Brain stem Hippocampus
structures within the olfactory bulb called glomeruli.
cortex
Each bulb has several thousand such glomeruli,
each of which is the terminus for about 25,000 axons
Figure 53-4
from olfactory cells. Each glomerulus also is the ter-
minus for dendrites from about 25 large mitral cells
Neural connections of the olfactory system.
and about 60 smaller tufted cells, the cell bodies of
which lie in the olfactory bulb superior to the
glomeruli. These dendrites receive synapses from the
olfactory cell neurons, and the mitral and tufted cells
send axons through the olfactory tract to transmit
olfactory signals to higher levels in the central nervous
primitive responses to olfaction, such as licking the
system.
lips, salivation, and other feeding responses caused by
Some research has suggested that different
the smell of food or by primitive emotional drives
glomeruli respond to different odors. It is possible that
associated with smell. Conversely, removal of the
specific glomeruli are the real clue to the analysis of
lateral areas abolishes the more complicated olfactory
different odor signals transmitted into the central
conditioned reflexes.
nervous system.
The Less Old Olfactory System—The Lateral Olfactory Area.
The Very Old, the Less Old, and the
The lateral olfactory area is composed mainly of the
Newer Olfactory Pathways into the
prepyriform and pyriform cortex plus the cortical
Central Nervous System
portion of the amygdaloid nuclei. From these areas,
The olfactory tract enters the brain at the anterior
signal pathways pass into almost all portions of the
junction between the mesencephalon and cerebrum;
limbic system, especially into less primitive portions
there, the tract divides into two pathways, as shown in
such as the hippocampus, which seem to be most
Figure 53-4, one passing medially into the medial
important for learning to like or dislike certain foods
olfactory area of the brain stem, and the other passing
depending on one’s experiences with them. For
laterally into the lateral olfactory area. The medial
instance, it is believed that this lateral olfactory
olfactory area represents a very old olfactory system,
area and its many connections with the limbic behav-
whereas the lateral olfactory area is the input to (1) a
ioral system cause a person to develop an absolute
less old olfactory system and (2) a newer system.
aversion to foods that have caused nausea and
vomiting.
The Very Old Olfactory System—The Medial Olfactory Area.
An important feature of the lateral olfactory area is
The medial olfactory area consists of a group of nuclei
that many signal pathways from this area also feed
located in the midbasal portions of the brain immedi-
directly into an older part of the cerebral cortex called
ately anterior to the hypothalamus. Most conspicuous
the paleocortex in the anteromedial portion of the tem-
are the septal nuclei, which are midline nuclei that feed
poral lobe. This is the only area of the entire cerebral
into the hypothalamus and other primitive portions
cortex where sensory signals pass directly to the cortex
of the brain’s limbic system. This is the brain area
without passing first through the thalamus.
most concerned with basic behavior (described in
Chapter 58).
The Newer Pathway. A newer olfactory pathway has now
The importance of this medial olfactory area is best
been found that passes through the thalamus, passing
understood by considering what happens in animals
to the dorsomedial thalamic nucleus and then to the
when the lateral olfactory areas on both sides of the
lateroposterior quadrant of the orbitofrontale cortex.
brain are removed and only the medial system
Based on studies in monkeys, this newer system prob-
remains. The answer is that this hardly affects the more
ably helps in the conscious analysis of odor.
670
Unit X The Nervous System: B. The Special Senses
Summary. Thus, there appear to be a very old olfactory
Herness MS, Gilbertson TA: Cellular mechanisms of taste
system that subserves the basic olfactory reflexes, a
transduction. Annu Rev Physiol 61:873, 1999.
less old system that provides automatic but partially
Levine AS, Kotz CM, Gosnell BA: Sugars and fats: the neu-
robiology of preference. J Nutr 133:831S, 2003.
learned control of food intake and aversion to toxic
Lledo PM, Gheusi G: Olfactory processing in a changing
and unhealthy foods, and a newer system that is com-
brain. Neuroreport 14:1655, 2003.
parable to most of the other cortical sensory systems
Lowe G: Electrical signaling in the olfactory bulb. Curr Opin
and is used for conscious perception and analysis of
Neurobiol 13:476, 2003.
olfaction.
Margolskee RF: Molecular mechanisms of bitter and sweet
taste transduction. J Biol Chem 277:1, 2002.
Centrifugal Control of Activity in the Olfactory Bulb by the
Matthews HR, Reisert J: Calcium, the two-faced messenger
Central Nervous System. Many nerve fibers that originate
of olfactory transduction and adaptation. Curr Opin Neu-
in the olfactory portions of the brain pass from the
robiol 13:469, 2003.
brain in the outward direction into the olfactory tract
Menini A, Lagostena L, Boccaccio A: Olfaction: from
odorant molecules to the olfactory cortex. News Physiol
to the olfactory bulb (i.e., “centrifugally” from the
Sci 19:101, 2004.
brain to the periphery). These terminate on a large
Mombaerts P: Genes and ligands for odorant, vomeronasal
number of small granule cells located among the mitral
and taste receptors. Nat Rev Neurosci 5:263, 2004.
and tufted cells in the olfactory bulb. The granule cells
Mombaerts P: Odorant receptor gene choice in olfactory
send inhibitory signals to the mitral and tufted cells. It
sensory neurons: the one receptor-one neuron hypothesis
is believed that this inhibitory feedback might be a
revisited. Curr Opin Neurobiol 14:31, 2004.
means for sharpening one’s specific ability to distin-
Montmayeur JP, Matsunami H: Receptors for bitter and
guish one odor from another.
sweet taste. Curr Opin Neurobiol 12:366, 2002.
Reed RR: The contribution of signaling pathways to olfac-
tory organization and development. Curr Opin Neurobiol
References
13:482, 2003.
Ronnett GV, Moon C: G proteins and olfactory signal trans-
duction. Annu Rev Physiol 64:189, 2002.
Bermudez-Rattoni F: Molecular mechanisms of taste-recog-
Sewards TV: Dual separate pathways for sensory and
nition memory. Nat Rev Neurosci 5:209, 2004.
hedonic aspects of taste. Brain Res Bull 62:271, 2004.
Dohlman HG: G proteins and pheromone signaling. Annu
Smith DV, Margolskee RF: Making sense of taste. Sci Am
Rev Physiol 64:129, 2002.
284:32, 2001.
Gibson AD, Garbers DL: Guanylyl cyclases as a family of
putative odorant receptors. Annu Rev Neurosci 23:417,
2000.
U N I T XI
The Nervous
System: C. Motor
and Integrative
Neurophysiology
54. Motor Functions of the Spinal Cord; the
Cord Reflexes
55. Cortical and Brain Stem Control of
Motor Function
56. Contributions of the Cerebellum and Basal
Ganglia to Overall Motor Control
57. Cerebral Cortex, Intellectual Functions
of the Brain, Learning and Memory
58. Behavioral and Motivational Mechanisms
of the Brain—The Limbic System and
the Hypothalamus
59. States of Brain Activity—Sleep, Brain Waves,
Epilepsy, Psychoses
60. Autonomic Nervous System and the
Adrenal Medulla
61. Cerebral Blood Flow, Cerebrospinal Fluid,
and Brain Metabolism
C H A P T E R
5
4
Motor Functions of the Spinal
Cord; the Cord Reflexes
We have already noted that sensory information is
integrated at all levels of the nervous system and
causes appropriate motor responses that begin in
the spinal cord with relatively simple muscle
reflexes, extend into the brain stem with more com-
plicated responses, and finally extend to the cere-
brum, where the most complicated muscle skills are
controlled.
In this chapter, we discuss the control of muscle function by the spinal cord.
Without the special neuronal circuits of the cord, even the most complex motor
control systems in the brain could not cause any purposeful muscle movement.
To give an example, there is no neuronal circuit anywhere in the brain that
causes the specific to-and-fro movement of the legs that is required in walking.
Instead, the circuits for these movements are in the cord, and the brain simply
sends command signals to the spinal cord to set into motion the walking process.
Let us not belittle the role of the brain, however, because the brain gives
directions that control the sequential cord activities—to promote turning move-
ments when they are required, to lean the body forward during acceleration, to
change the movements from walking to jumping as needed, and to monitor con-
tinuously and control equilibrium. All this is done through “analytical” and
“command” signals generated in the brain. But it also requires the many neu-
ronal circuits of the spinal cord that are the objects of the commands. These
circuits provide all but a small fraction of the direct control of the muscles.
Spinal Animal and the Decerebrate Animal. Two types of experimental preparations have
been especially useful in studying spinal cord function: (1) the spinal animal, in
which the spinal cord is transected, frequently in the neck so that most of the cord
still remains functional, and (2) the decerebrate animal, in which the brain stem is
transected in the middle to lower part of the mesencephalon.
Immediately after preparation of a spinal animal, most spinal cord function below
the level of transection is severely depressed. After a few hours in rats and cats or
a few days to weeks in monkeys, most of the intrinsic spinal cord functions return
to nearly normal and provide a suitable experimental preparation for research
study.
In the decerebrate animal, the brain stem is transected at the middle to lower mes-
encephalic level, which blocks normal inhibitory signals from the higher control
centers of the brain to the pontile and vestibular muscle control nuclei. This allows
these nuclei to become tonically active, transmitting facilitatory signals to most of
the spinal cord motor control circuits. The result is that the spinal cord motor
reflexes become very excitable and, therefore, easy to activate by even the slightest
sensory input signals to the cord. Using this preparation, one can study the intrin-
sic excitatory motor functions of the cord itself.
Organization of the Spinal Cord
for Motor Functions
The cord gray matter is the integrative area for the cord reflexes. Figure 54-1
shows the typical organization of the cord gray matter in a single cord segment.
Sensory signals enter the cord almost entirely through the sensory (posterior)
roots. After entering the cord, every sensory signal travels to two separate
673
674
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Sensory root
Motor
Sensory Motor
Solitary cell
a
g
Ia
II
g
14 mm
5 mm
17 mm
8 mm
5 mm
External basal cells
Corticospinal tract
Alpha motor
Sheath Primary
Extrafusal
Interneurons
ending
ending
fibers
Gamma motor
Fluid
Secondary Intrafusal
Anterior motor
ending
cavity
ending
fibers
neurons
1 cm
Figure 54-2
Muscle spindle, showing its relation to the large extrafusal skele-
tal muscle fibers. Note also both motor and sensory innervation of
Motor root
the muscle spindle.
Figure 54-1
Connections of peripheral sensory fibers and corticospinal fibers
Gamma Motor Neurons. Along with the alpha motor
with the interneurons and anterior motor neurons of the spinal
cord.
neurons, which excite contraction of the skeletal
muscle fibers, about one half as many much smaller
gamma motor neurons are located in the spinal cord
anterior horns. These gamma motor neurons transmit
destinations: (1) One branch of the sensory nerve ter-
impulses through much smaller type A gamma (Ag)
minates almost immediately in the gray matter of the
motor nerve fibers, averaging 5 micrometers in diam-
cord and elicits local segmental cord reflexes and other
eter, which go to small, special skeletal muscle fibers
local effects. (2) Another branch transmits signals to
called intrafusal fibers, shown in Figure 54-2. These
higher levels of the nervous system—to higher levels
fibers constitute the middle of the muscle spindle,
in the cord itself, to the brain stem, or even to the cere-
which helps control basic muscle “tone,” as discussed
bral cortex, as described in earlier chapters.
later in this chapter.
Each segment of the spinal cord (at the level of
each spinal nerve) has several million neurons in its
Interneurons. Interneurons are present in all areas of
gray matter. Aside from the sensory relay neurons
the cord gray matter—in the dorsal horns, the anterior
discussed in Chapters 47 and 48, the other neurons
horns, and the intermediate areas between them, as
are of two types: (1) anterior motor neurons and (2)
shown in Figure 54-1. These cells are about 30 times
interneurons.
as numerous as the anterior motor neurons. They are
small and highly excitable, often exhibiting sponta-
Anterior Motor Neurons. Located in each segment of the
neous activity and capable of firing as rapidly as 1500
anterior horns of the cord gray matter are several
times per second. They have many interconnections
thousand neurons that are 50 to 100 per cent larger
with one another, and many of them also synapse
than most of the others and are called anterior motor
directly with the anterior motor neurons, as shown
neurons. They give rise to the nerve fibers that leave
in Figure
54-1. The interconnections among the
the cord by way of the anterior roots and directly
interneurons and anterior motor neurons are respon-
innervate the skeletal muscle fibers. The neurons are
sible for most of the integrative functions of the spinal
of two types, alpha motor neurons and gamma motor
cord that are discussed in the remainder of this
neurons.
chapter.
Essentially all the different types of neuronal cir-
Alpha Motor Neurons. The alpha motor neurons give
cuits described in Chapter
46 are found in the
rise to large type A alpha (Aa) motor nerve fibers,
interneuron pool of cells of the spinal cord, including
averaging 14 micrometers in diameter; these fibers
diverging, converging, repetitive-discharge, and other
branch many times after they enter the muscle and
types of circuits. In this chapter, we examine many
innervate the large skeletal muscle fibers. Stimulation
applications of these different circuits in the perform-
of a single alpha nerve fiber excites anywhere from
ance of specific reflex acts by the spinal cord.
three to several hundred skeletal muscle fibers, which
Only a few incoming sensory signals from the spinal
are collectively called the motor unit. Transmission of
nerves or signals from the brain terminate directly on
nerve impulses into skeletal muscles and their stimu-
the anterior motor neurons. Instead, almost all these
lation of the muscle motor units are discussed in
signals are transmitted first through interneurons,
Chapters 6 and 7.
where they are appropriately processed. Thus, in
Chapter 54
Motor Functions of the Spinal Cord; the Cord Reflexes
675
Figure 54-1, the corticospinal tract from the brain is
The signals from these two receptors are either
shown to terminate almost entirely on spinal interneu-
entirely or almost entirely for the purpose of intrinsic
rons, where the signals from this tract are combined
muscle control. They operate almost completely at a
with signals from other spinal tracts or spinal nerves
subconscious level. Even so, they transmit tremendous
before finally converging on the anterior motor
amounts of information not only to the spinal cord but
neurons to control muscle function.
also to the cerebellum and even to the cerebral cortex,
helping each of these portions of the nervous system
Renshaw Cell Inhibitory System. Also located in the anterior
function to control muscle contraction.
horns of the spinal cord, in close association with the
motor neurons, are a large number of small neurons
called Renshaw cells. Almost immediately after the
Receptor Function of the
anterior motor neuron axon leaves the body of the
Muscle Spindle
neuron, collateral branches from the axon pass to adja-
cent Renshaw cells. These are inhibitory cells that trans-
mit inhibitory signals to the surrounding motor neurons.
Structure and Motor Innervation of the Muscle Spindle. The
Thus, stimulation of each motor neuron tends to inhibit
organization of the muscle spindle is shown in Figure
adjacent motor neurons, an effect called lateral inhibi-
54-2. Each spindle is 3 to 10 millimeters long. It is built
tion. This effect is important for the following major
around 3 to 12 very small intrafusal muscle fibers that
reason: The motor system uses this lateral inhibition to
are pointed at their ends and attached to the glycoca-
focus, or sharpen, its signals in the same way that the
lyx of the surrounding large extrafusal skeletal muscle
sensory system uses the same principle—that is, to allow
fibers.
unabated transmission of the primary signal in the
Each intrafusal muscle fiber is a very small skeletal
desired direction while suppressing the tendency for
muscle fiber. However, the central region of each
signals to spread laterally.
of these fibers—that is, the area midway between
Multisegmental Connections from One Spinal Cord Level
its two ends—has few or no actin and myosin fila-
to Other Levels—Propriospinal Fibers
ments. Therefore, this central portion does not contract
More than half of all the nerve fibers that ascend and
when the ends do. Instead, it functions as a sensory
descend in the spinal cord are propriospinal fibers.
receptor, as described later. The end portions that
These fibers run from one segment of the cord to
do contract are excited by small gamma motor nerve
another. In addition, as the sensory fibers enter the cord
fibers that originate from small type A gamma motor
from the posterior cord roots, they bifurcate and branch
neurons in the anterior horns of the spinal cord,
both up and down the spinal cord; some of the branches
as described earlier. These gamma motor nerve fibers
transmit signals to only a segment or two, while others
are also called gamma efferent fibers, in contradistinc-
transmit signals to many segments. These ascending and
tion to the large alpha efferent fibers (type A alpha
descending propriospinal fibers of the cord provide
pathways for the multisegmental reflexes described
nerve fibers) that innervate the extrafusal skeletal
later in this chapter, including reflexes that coordinate
muscle.
simultaneous movements in the forelimbs and hind
limbs.
Sensory Innervation of the Muscle Spindle. The receptor
portion of the muscle spindle is its central portion. In
this area, the intrafusal muscle fibers do not have
Muscle Sensory Receptors—
myosin and actin contractile elements. As shown in
Figure 54-2 and in more detail in Figure 54-3, sensory
Muscle Spindles and Golgi
fibers originate in this area. They are stimulated by
Tendon Organs—And Their
stretching of this midportion of the spindle. One can
Roles in Muscle Control
readily see that the muscle spindle receptor can be
excited in two ways:
Proper control of muscle function requires not only
1. Lengthening the whole muscle stretches the
excitation of the muscle by spinal cord anterior motor
midportion of the spindle and, therefore, excites
neurons but also continuous feedback of sensory
the receptor.
information from each muscle to the spinal cord, indi-
2. Even if the length of the entire muscle does not
cating the functional status of each muscle at each
change, contraction of the end portions of the
instant. That is, what is the length of the muscle, what
spindle’s intrafusal fibers stretches the midportion
is its instantaneous tension, and how rapidly is its
of the spindle and therefore excites the receptor.
length or tension changing? To provide this informa-
Two types of sensory endings are found in this
tion, the muscles and their tendons are supplied abun-
central receptor area of the muscle spindle. They are
dantly with two special types of sensory receptors: (1)
the primary ending and the secondary ending.
muscle spindles (see Figure 54-2), which are distrib-
uted throughout the belly of the muscle and send
Primary Ending. In the center of the receptor area, a
information to the nervous system about muscle
large sensory nerve fiber encircles the central portion
length or rate of change of length, and (2) Golgi
of each intrafusal fiber, forming the so-called primary
tendon organs (see Figure 54-7), which are located in
ending or annulospiral ending. This nerve fiber is a
the muscle tendons and transmit information about
type Ia fiber averaging 17 micrometers in diameter,
tendon tension or rate of change of tension.
and it transmits sensory signals to the spinal cord at a
676
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Dynamic g fiber Static g fiber
Group Ia fiber
several minutes if the muscle spindle itself remains
(efferent)
(efferent)
(primary afferent)
stretched.
Plate
Group II fiber
ending
(secondary
Response of the Primary Ending (but Not the Sec-
efferent)
ondary Ending) to Rate of Change of Receptor
Nuclear bag fiber
Length—“Dynamic” Response. When the length of
(intrafusal muscle)
the spindle receptor increases suddenly, the primary
Nuclear chain fiber
ending (but not the secondary ending) is stimulated
(intrafusal muscle)
especially powerfully. This excess stimulus of the
Trail ending
primary ending is called the dynamic response, which
means that the primary ending responds extremely
Figure 54-3
actively to a rapid rate of change in spindle length.
Details of nerve connections from the nuclear bag and nuclear
Even when the length of a spindle receptor increases
chain muscle spindle fibers. (Modified from Stein RB: Peripheral
only a fraction of a micrometer, if this increase occurs
control of movement. Physiol Rev 54:225, 1974.)
in a fraction of a second, the primary receptor trans-
mits tremendous numbers of excess impulses to the
large
17-micrometer sensory nerve fiber, but only
while the length is actually increasing. As soon as the
length stops increasing, this extra rate of impulse dis-
velocity of 70 to 120 m/sec, as rapidly as any type of
charge returns to the level of the much smaller static
nerve fiber in the entire body.
response that is still present in the signal.
Conversely, when the spindle receptor shortens,
Secondary Ending. Usually one but sometimes two
exactly opposite sensory signals occur. Thus, the
smaller sensory nerve fibers—type II fibers with an
primary ending sends extremely strong, either positive
average diameter of 8 micrometers—innervate the
or negative, signals to the spinal cord to apprise it of
receptor region on one or both sides of the primary
any change in length of the spindle receptor.
ending, as shown in Figures
54-2 and 54-3. This
sensory ending is called the secondary ending; some-
Control of Intensity of the Static and Dynamic Responses by the
times it encircles the intrafusal fibers in the same way
Gamma Motor Nerves. The gamma motor nerves to the
that the type Ia fiber does, but often it spreads like
muscle spindle can be divided into two types: gamma-
branches on a bush.
dynamic (gamma-d) and gamma-static (gamma-s). The
first of these excites mainly the nuclear bag intrafusal
Division of the Intrafusal Fibers into Nuclear Bag and Nuclear
fibers, and the second excites mainly the nuclear chain
Chain Fibers—Dynamic and Static Responses of the Muscle
intrafusal fibers. When the gamma-d fibers excite the
Spindle. There are also two types of muscle spindle
nuclear bag fibers, the dynamic response of the muscle
intrafusal fibers: (1) nuclear bag muscle fibers (one to
spindle becomes tremendously enhanced, whereas the
three in each spindle), in which several muscle fiber
static response is hardly affected. Conversely, stimula-
nuclei are congregated in expanded “bags” in the
tion of the gamma-s fibers, which excite the nuclear
central portion of the receptor area, as shown by the
chain fibers, enhances the static response while having
top fiber in Figure 54-3, and (2) nuclear chain fibers
little influence on the dynamic response. Subsequent
(three to nine), which are about half as large in diam-
paragraphs illustrate that these two types of muscle
eter and half as long as the nuclear bag fibers and have
spindle responses are important in different types of
nuclei aligned in a chain throughout the receptor area,
muscle control.
as shown by the bottom fiber in the figure. The primary
sensory nerve ending
(the
17-micrometer sensory
Continuous Discharge of the Muscle Spindles Under Normal
fiber) is excited by both the nuclear bag intrafusal
Conditions. Normally, particularly when there is some
fibers and the nuclear chain fibers. Conversely, the sec-
degree of gamma nerve excitation, the muscle spindles
ondary ending (the 8-micrometer sensory fiber) is
emit sensory nerve impulses continuously. Stretching
usually excited only by nuclear chain fibers. These rela-
the muscle spindles increases the rate of firing,
tions are shown in Figure 54-3.
whereas shortening the spindle decreases the rate of
firing. Thus, the spindles can send to the spinal cord
Response of Both the Primary and the Secondary
either positive signals—that is, increased numbers of
Endings to the Length of the Receptor—“Static”
impulses to indicate stretch of a muscle—or negative
Response. When the receptor portion of the muscle
signals—below-normal numbers of impulses to indi-
spindle is stretched slowly, the number of impulses
cate that the muscle is unstretched.
transmitted from both the primary and the secondary
endings increases almost directly in proportion to the
degree of stretching, and the endings continue to trans-
Muscle Stretch Reflex
mit these impulses for several minutes. This effect is
called the static response of the spindle receptor,
The simplest manifestation of muscle spindle function
meaning simply that both the primary and secondary
is the muscle stretch reflex. Whenever a muscle is
endings continue to transmit their signals for at least
stretched suddenly, excitation of the spindles causes
Chapter 54
Motor Functions of the Spinal Cord; the Cord Reflexes
677
Stimulus
(8 per second)
Sensory nerve
Motor nerve
A
B
0
1
2
3
Muscle spindle
Seconds
Stretch reflex
Figure 54-5
Figure 54-4
Muscle contraction caused by a spinal cord signal under two con-
Neuronal circuit of the stretch reflex.
ditions: curve A, in a normal muscle, and curve B, in a muscle
whose muscle spindles were denervated by section of the poste-
rior roots of the cord 82 days previously. Note the smoothing effect
reflex contraction of the large skeletal muscle fibers of
of the muscle spindle reflex in curve A. (Modified from Creed RS,
the stretched muscle and also of closely allied syner-
et al: Reflex Activity of the Spinal Cord. New York: Oxford Univer-
gistic muscles.
sity Press, 1932.)
Neuronal Circuitry of the Stretch Reflex. Figure
54-4
demonstrates the basic circuit of the muscle spindle
stretch reflex, showing a type Ia proprioceptor nerve
fiber originating in a muscle spindle and entering a
secondary endings. The importance of the static stretch
dorsal root of the spinal cord. A branch of this fiber
reflex is that it causes the degree of muscle contraction
then goes directly to the anterior horn of the cord gray
to remain reasonably constant, except when the
matter and synapses with anterior motor neurons that
person’s nervous system specifically wills otherwise.
send motor nerve fibers back to the same muscle from
which the muscle spindle fiber originated. Thus, this is
“Damping” Function of the Dynamic and Static
a monosynaptic pathway that allows a reflex signal to
Stretch Reflexes
return with the shortest possible time delay back to the
An especially important function of the stretch reflex
muscle after excitation of the spindle. Most type II
is its ability to prevent oscillation or jerkiness of body
fibers from the muscle spindle terminate on multiple
movements. This is a damping, or smoothing, function,
interneurons in the cord gray matter, and these trans-
as explained in the following paragraph.
mit delayed signals to the anterior motor neurons or
serve other functions.
Damping Mechanism in Smoothing Muscle Contraction.
Signals from the spinal cord are often transmitted to a
Dynamic Stretch Reflex and Static Stretch Reflexes. The
muscle in an unsmooth form, increasing in intensity for
stretch reflex can be divided into two components: the
a few milliseconds, then decreasing in intensity, then
dynamic stretch reflex and the static stretch reflex.
changing to another intensity level, and so forth. When
The dynamic stretch reflex is elicited by the potent
the muscle spindle apparatus is not functioning satis-
dynamic signal transmitted from the primary sensory
factorily, the muscle contraction is jerky during the
endings of the muscle spindles, caused by rapid stretch
course of such a signal. This effect is demonstrated in
or unstretch. That is, when a muscle is suddenly
Figure 54-5. In curve A, the muscle spindle reflex of
stretched or unstretched, a strong signal is transmitted
the excited muscle is intact. Note that the contraction
to the spinal cord; this causes an instantaneous strong
is relatively smooth, even though the motor nerve to
reflex contraction (or decrease in contraction) of the
the muscle is excited at a slow frequency of only 8
same muscle from which the signal originated. Thus,
signals per second. Curve B illustrates the same exper-
the reflex functions to oppose sudden changes in muscle
iment in an animal whose muscle spindle sensory
length.
nerves had been sectioned 3 months earlier. Note the
The dynamic stretch reflex is over within a fraction
unsmooth muscle contraction. Thus, curve A graphi-
of a second after the muscle has been stretched (or
cally demonstrates the damping mechanism’s ability to
unstretched) to its new length, but then a weaker static
smooth muscle contractions, even though the primary
stretch reflex continues for a prolonged period there-
input signals to the muscle motor system may them-
after. This reflex is elicited by the continuous static
selves be jerky. This effect can also be called a signal
receptor signals transmitted by both primary and
averaging function of the muscle spindle reflex.
678
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Role of the Muscle Spindle
joint also increases, producing tight, tense muscles
opposing each other at the joint. The net effect is that
in Voluntary Motor Activity
the position of the joint becomes strongly stabilized,
and any force that tends to move the joint from its
To emphasize the importance of the gamma efferent
current position is opposed by highly sensitized stretch
system, one needs to recognize that 31 per cent of all
reflexes operating on both sides of the joint.
the motor nerve fibers to the muscle are the small type
Any time a person must perform a muscle function
A gamma efferent fibers rather than large type A
that requires a high degree of delicate and exact posi-
alpha motor fibers. Whenever signals are transmitted
tioning, excitation of the appropriate muscle spindles
from the motor cortex or from any other area of the
by signals from the bulboreticular facilitatory region
brain to the alpha motor neurons, in most instances the
of the brain stem stabilizes the positions of the major
gamma motor neurons are stimulated simultaneously,
joints. This aids tremendously in performing the
an effect called coactivation of the alpha and gamma
additional detailed voluntary movements (of fingers
motor neurons. This causes both the extrafusal skele-
or other body parts) required for intricate motor
tal muscle fibers and the muscle spindle intrafusal
procedures.
muscle fibers to contract at the same time.
The purpose of contracting the muscle spindle intra-
fusal fibers at the same time that the large skeletal
Clinical Applications of the
muscle fibers contract is twofold: First, it keeps the
Stretch Reflex
length of the receptor portion of the muscle spindle
from changing during the course of the whole muscle
Almost every time a clinician performs a physical exam-
contraction. Therefore, coactivation keeps the muscle
ination on a patient, he or she elicits multiple stretch
spindle reflex from opposing the muscle contraction.
reflexes. The purpose is to determine how much back-
ground excitation, or “tone,” the brain is sending to the
Second, it maintains the proper damping function of
spinal cord. This reflex is elicited as follows.
the muscle spindle, regardless of any change in muscle
length. For instance, if the muscle spindle did not con-
Knee Jerk and Other Muscle Jerks. Clinically, a method used
tract and relax along with the large muscle fibers,
to determine the sensitivity of the stretch reflexes is to
the receptor portion of the spindle would sometimes
elicit the knee jerk and other muscle jerks. The knee jerk
be flail and sometimes be overstretched, in neither
can be elicited by simply striking the patellar tendon
instance operating under optimal conditions for
with a reflex hammer; this instantaneously stretches the
spindle function.
quadriceps muscle and excites a dynamic stretch reflex
that causes the lower leg to “jerk” forward. The upper
part of Figure 54-6 shows a myogram from the quadri-
Brain Areas for Control of the Gamma
ceps muscle recorded during a knee jerk.
Motor System
Similar reflexes can be obtained from almost any
The gamma efferent system is excited specifically by
muscle of the body either by striking the tendon of the
signals from the bulboreticular facilitatory region of
muscle or by striking the belly of the muscle itself. In
the brain stem and, secondarily, by impulses transmit-
other words, sudden stretch of muscle spindles is all that
ted into the bulboreticular area from (1) the cerebel-
is required to elicit a dynamic stretch reflex.
lum, (2) the basal ganglia, and (3) the cerebral cortex.
The muscle jerks are used by neurologists to assess
the degree of facilitation of spinal cord centers. When
Little is known about the precise mechanisms of
control of the gamma efferent system. However,
because the bulboreticular facilitatory area is par-
ticularly concerned with antigravity contractions, and
because the antigravity muscles have an especially
Patellar tendon struck
high density of muscle spindles, emphasis is given to
the importance of the gamma efferent mechanism for
Knee jerk
damping the movements of the different body parts
during walking and running.
Muscle Spindle System Stabilizes Body
Position During Tense Action
One of the most important functions of the muscle
Ankle clonus
spindle system is to stabilize body position during
tense motor action. To do this, the bulboreticular facil-
0
200
400
600
800
itatory region and its allied areas of the brain stem
Milliseconds
transmit excitatory signals through the gamma nerve
fibers to the intrafusal muscle fibers of the muscle
spindles. This shortens the ends of the spindles and
stretches the central receptor regions, thus increasing
Figure 54-6
their signal output. However, if the spindles on both
Myograms recorded from the quadriceps muscle during elicita-
sides of each joint are activated at the same time, reflex
tion of the knee jerk (above) and from the gastrocnemius muscle
excitation of the skeletal muscles on both sides of the
during ankle clonus (below).
Chapter 54
Motor Functions of the Spinal Cord; the Cord Reflexes
679
large numbers of facilitatory impulses are being trans-
mitted from the upper regions of the central nervous
Nerve fiber (16 mm)
system into the cord, the muscle jerks are greatly exag-
gerated. Conversely, if the facilitatory impulses are
depressed or abrogated, the muscle jerks are consider-
ably weakened or absent. These reflexes are used most
frequently in determining the presence or absence of
muscle spasticity caused by lesions in the motor areas
of the brain or diseases that excite the bulboreticular
Tendon
facilitatory area of the brain stem. Ordinarily, large
lesions in the motor areas of the cerebral cortex but not
in the lower motor control areas (especially lesions
Muscle
caused by strokes or brain tumors) cause greatly exag-
gerated muscle jerks in the muscles on the opposite side
Figure 54-7
of the body.
Golgi tendon organ.
Clonus—Oscillation of Muscle Jerks. Under some condi-
tions, the muscle jerks can oscillate, a phenomenon
proportional to the muscle tension
(the static
called clonus (see lower myogram, Figure 54-6). Oscil-
response). Thus, Golgi tendon organs provide the
lation can be explained particularly well in relation to
ankle clonus, as follows.
nervous system with instantaneous information on the
If a person standing on the tip ends of the feet sud-
degree of tension in each small segment of each
denly drops his or her body downward and stretches
muscle.
the gastrocnemius muscles, stretch reflex impulses are
transmitted from the muscle spindles into the spinal
Transmission of Impulses from the Tendon Organ into the
cord. These impulses reflexively excite the stretched
Central Nervous System. Signals from the tendon organ
muscle, which lifts the body up again. After a fraction
are transmitted through large, rapidly conducting type
of a second, the reflex contraction of the muscle dies out
Ib nerve fibers that average 16 micrometers in diame-
and the body falls again, thus stretching the spindles a
ter, only slightly smaller than those from the primary
second time. Again, a dynamic stretch reflex lifts the
endings of the muscle spindle. These fibers, like those
body, but this too dies out after a fraction of a second,
and the body falls once more to begin a new cycle. In
from the primary spindle endings, transmit signals
this way, the stretch reflex of the gastrocnemius muscle
both into local areas of the cord and, after synapsing
continues to oscillate, often for long periods; this is
in a dorsal horn of the cord, through long fiber path-
clonus.
ways such as the spinocerebellar tracts into the cere-
Clonus ordinarily occurs only when the stretch reflex
bellum and through still other tracts to the cerebral
is highly sensitized by facilitatory impulses from the
cortex. The local cord signal excites a single inhibitory
brain. For instance, in a decerebrate animal, in which the
interneuron that inhibits the anterior motor neuron.
stretch reflexes are highly facilitated, clonus develops
This local circuit directly inhibits the individual muscle
readily. To determine the degree of facilitation of the
without affecting adjacent muscles. The relation
spinal cord, neurologists test patients for clonus by sud-
between signals to the brain and function of the cere-
denly stretching a muscle and applying a steady stretch-
ing force to it. If clonus occurs, the degree of facilitation
bellum and other parts of the brain for muscle control
is certain to be high.
is discussed in Chapter 56.
Inhibitory Nature of the Tendon Reflex and
Golgi Tendon Reflex
Its Importance
When the Golgi tendon organs of a muscle tendon are
Golgi Tendon Organ Helps Control Muscle Tension. The Golgi
stimulated by increased tension in the connecting
tendon organ, shown in Figure 54-7, is an encapsulated
muscle, signals are transmitted to the spinal cord to
sensory receptor through which muscle tendon fibers
cause reflex effects in the respective muscle. This reflex
pass. About 10 to 15 muscle fibers are usually con-
is entirely inhibitory. Thus, this reflex provides a nega-
nected to each Golgi tendon organ, and the organ is
tive feedback mechanism that prevents the develop-
stimulated when this small bundle of muscle fibers is
ment of too much tension on the muscle.
“tensed” by contracting or stretching the muscle. Thus,
When tension on the muscle and, therefore, on the
the major difference in excitation of the Golgi tendon
tendon becomes extreme, the inhibitory effect from
organ versus the muscle spindle is that the spindle
the tendon organ can be so great that it leads to a
detects muscle length and changes in muscle length,
sudden reaction in the spinal cord that causes instan-
whereas the tendon organ detects muscle tension as
taneous relaxation of the entire muscle. This effect is
reflected by the tension in itself.
called the lengthening reaction; it is probably a pro-
The tendon organ, like the primary receptor of the
tective mechanism to prevent tearing of the muscle or
muscle spindle, has both a dynamic response and a
avulsion of the tendon from its attachments to the
static response, responding intensely when the muscle
bone. We know, for instance, that direct electrical stim-
tension suddenly increases (the dynamic response) but
ulation of muscles in the laboratory, which cannot be
settling down within a fraction of a second to a lower
opposed by this negative reflex, can occasionally cause
level of steady-state firing that is almost directly
such destructive effects.
680
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Possible Role of the Tendon Reflex to Equalize Contractile
RECIPROCAL INHIBITION
Force Among the Muscle Fibers. Another likely function
of the Golgi tendon reflex is to equalize contractile
forces of the separate muscle fibers. That is, those fibers
that exert excess tension become inhibited by the
reflex, whereas those that exert too little tension
become more excited because of absence of reflex
Excited
inhibition. This spreads the muscle load over all the
Inhibited
Inhibited
fibers and prevents damage in isolated areas of
a muscle where small numbers of fibers might be
overloaded.
Excited
Polysynaptic
Function of the Muscle Spindles and
circuit
Golgi Tendon Organs in Conjunction
with Motor Control from Higher Levels
Painful
of the Brain
stimulus
from hand
Although we have emphasized the function of the
muscle spindles and Golgi tendon organs in spinal
cord control of motor function, these two sensory
organs also apprise the higher motor control centers
of instantaneous changes taking place in the muscles.
For instance, the dorsal spinocerebellar tracts carry
instantaneous information from both the muscle
FLEXOR
CROSSED EXTENSOR
spindles and the Golgi tendon organs directly to the
REFLEX
REFLEX
cerebellum at conduction velocities approaching
Figure 54-8
120 m/sec, the most rapid conduction anywhere in the
brain or spinal cord. Additional pathways transmit
Flexor reflex, crossed extensor reflex, and reciprocal inhibition.
similar information into the reticular regions of the
brain stem and, to a lesser extent, all the way to the
for the flexor reflex. In this instance, a painful stimu-
motor areas of the cerebral cortex. As discussed in
lus is applied to the hand; as a result, the flexor muscles
Chapters 55 and 56, the information from these recep-
of the upper arm become excited, thus withdrawing
tors is crucial for feedback control of motor signals
the hand from the painful stimulus.
that originate in all these areas.
The pathways for eliciting the flexor reflex do not
pass directly to the anterior motor neurons but instead
pass first into the spinal cord interneuron pool of
Flexor Reflex and the
neurons and only secondarily to the motor neurons.
Withdrawal Reflexes
The shortest possible circuit is a three- or four-neuron
pathway; however, most of the signals of the reflex tra-
In the spinal or decerebrate animal, almost any type
verse many more neurons and involve the following
of cutaneous sensory stimulus from a limb is likely to
basic types of circuits: (1) diverging circuits to spread
cause the flexor muscles of the limb to contract,
the reflex to the necessary muscles for withdrawal; (2)
thereby withdrawing the limb from the stimulating
circuits to inhibit the antagonist muscles, called recip-
object. This is called the flexor reflex.
rocal inhibition circuits; and (3) circuits to cause after-
In its classic form, the flexor reflex is elicited most
discharge lasting many fractions of a second after the
powerfully by stimulation of pain endings, such as by
stimulus is over.
a pinprick, heat, or a wound, for which reason it is also
Figure 54-9 shows a typical myogram from a flexor
called a nociceptive reflex, or simply a pain reflex. Stim-
muscle during a flexor reflex. Within a few milliseconds
ulation of touch receptors can also elicit a weaker and
after a pain nerve begins to be stimulated, the flexor
less prolonged flexor reflex.
response appears. Then, in the next few seconds, the
If some part of the body other than one of the limbs
reflex begins to fatigue, which is characteristic of essen-
is painfully stimulated, that part will similarly be with-
tially all complex integrative reflexes of the spinal
drawn from the stimulus, but the reflex may not be con-
cord. Finally, after the stimulus is over, the contraction
fined to flexor muscles, even though it is basically the
of the muscle returns toward the baseline, but because
same type of reflex. Therefore, the many patterns of
of afterdischarge, it takes many milliseconds for this
these reflexes in the different areas of the body are
to occur. The duration of afterdischarge depends on
called withdrawal reflexes.
the intensity of the sensory stimulus that elicited the
reflex; a weak tactile stimulus causes almost no after-
Neuronal Mechanism of the Flexor Reflex. The left-hand
discharge, but after a strong pain stimulus, the after-
portion of Figure 54-8 shows the neuronal pathways
discharge may last for a second or more.
Chapter 54
Motor Functions of the Spinal Cord; the Cord Reflexes
681
Fatigue
Afterdischarge
Duration of
stimulus
Afterdischarge
Duration of stimulus
0
1
2
3
0
1
2
3
4
Seconds
Seconds
Figure 54-9
Figure 54-10
Myogram of the flexor reflex showing rapid onset of the reflex,
Myogram of a crossed extensor reflex showing slow onset but pro-
an interval of fatigue, and, finally, afterdischarge after the input
longed afterdischarge.
stimulus is over.
This is called the crossed extensor reflex. Extension of
the opposite limb can push the entire body away from
The afterdischarge that occurs in the flexor reflex
the object causing the painful stimulus in the with-
almost certainly results from both types of repetitive
drawn limb.
discharge circuits discussed in Chapter 46. Electro-
physiologic studies indicate that immediate afterdis-
Neuronal Mechanism of the Crossed Extensor Reflex. The
charge, lasting for about 6 to 8 milliseconds, results
right-hand portion of Figure 54-8 shows the neuronal
from repetitive firing of the excited interneurons
circuit responsible for the crossed extensor reflex,
themselves. Also, prolonged afterdischarge occurs
demonstrating that signals from sensory nerves cross
after strong pain stimuli, almost certainly resulting
to the opposite side of the cord to excite extensor
from recurrent pathways that initiate oscillation in
muscles. Because the crossed extensor reflex usually
reverberating interneuron circuits. These, in turn,
does not begin until 200 to 500 milliseconds after onset
transmit impulses to the anterior motor neurons,
of the initial pain stimulus, it is certain that many
sometimes for several seconds after the incoming
interneurons are involved in the circuit between the
sensory signal is over.
incoming sensory neuron and the motor neurons of
Thus, the flexor reflex is appropriately organized to
the opposite side of the cord responsible for the
withdraw a pained or otherwise irritated part of the
crossed extension. After the painful stimulus is
body from a stimulus. Further, because of afterdis-
removed, the crossed extensor reflex has an even
charge, the reflex can hold the irritated part away from
longer period of afterdischarge than does the flexor
the stimulus for 0.1 to 3 seconds after the irritation is
reflex. Again, it is presumed that this prolonged after-
over. During this time, other reflexes and actions of the
discharge results from reverberating circuits among
central nervous system can move the entire body away
the interneuronal cells.
from the painful stimulus.
Figure 54-10 shows a typical myogram recorded
from a muscle involved in a crossed extensor reflex.
Pattern of Withdrawal. The pattern of withdrawal that
This demonstrates the relatively long latency before
results when the flexor reflex is elicited depends on
the reflex begins and the long afterdischarge at the end
which sensory nerve is stimulated. Thus, a pain stimu-
of the stimulus. The prolonged afterdischarge is of
lus on the inward side of the arm elicits not only
benefit in holding the pained area of the body away
contraction of the flexor muscles of the arm but also
from the painful object until other nervous reactions
contraction of abductor muscles to pull the arm
cause the entire body to move away.
outward. In other words, the integrative centers of
the cord cause those muscles to contract that can
most effectively remove the pained part of the body
Reciprocal Inhibition and
away from the object causing the pain. Although this
principle, called the principle of “local sign,” applies to
Reciprocal Innervation
any part of the body, it is especially applicable to the
limbs because of their highly developed flexor reflexes.
In the previous paragraphs, we pointed out several
times that excitation of one group of muscles is often
associated with inhibition of another group. For
Crossed Extensor Reflex
instance, when a stretch reflex excites one muscle, it
often simultaneously inhibits the antagonist muscles.
About 0.2 to 0.5 second after a stimulus elicits a flexor
This is the phenomenon of reciprocal inhibition, and
reflex in one limb, the opposite limb begins to extend.
the neuronal circuit that causes this reciprocal relation
682
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
raise itself to the standing position. This is called
the cord righting reflex. Such a reflex demonstrates
that some relatively complex reflexes associated with
Duration of inhibitory stimulus
posture are integrated in the spinal cord. Indeed, an
animal with a well-healed transected thoracic cord
between the levels for forelimb and hindlimb innerva-
tion can right itself from the lying position and even
walk using its hindlimbs in addition to its forelimbs.
Duration of flexor reflex stimulus
In the case of an opossum with a similar transection
of the thoracic cord, the walking movements of
0
1
2
3
the hindlimbs are hardly different from those in a
Seconds
normal opossum—except that the hindlimb walking
movements are not synchronized with those of the
forelimbs.
Figure 54-11
Stepping and Walking Movements
Myogram of a flexor reflex showing reciprocal inhibition caused
Rhythmical Stepping Movements of a Single Limb. Rhythmi-
by an inhibitory stimulus from a stronger flexor reflex on the oppo-
site side of the body.
cal stepping movements are frequently observed in the
limbs of spinal animals. Indeed, even when the lumbar
portion of the spinal cord is separated from the
remainder of the cord and a longitudinal section is
is called reciprocal innervation. Likewise, reciprocal
made down the center of the cord to block neuronal
relations often exist between the muscles on the two
connections between the two sides of the cord and
sides of the body, as exemplified by the flexor and
between the two limbs, each hindlimb can still perform
extensor muscle reflexes described earlier.
individual stepping functions. Forward flexion of the
Figure 54-11 shows a typical example of reciprocal
limb is followed a second or so later by backward
inhibition. In this instance, a moderate but prolonged
extension. Then flexion occurs again, and the cycle is
flexor reflex is elicited from one limb of the body;
repeated over and over.
while this reflex is still being elicited, a stronger flexor
This oscillation back and forth between flexor and
reflex is elicited in the limb on the opposite side of the
extensor muscles can occur even after the sensory
body. This stronger reflex sends reciprocal inhibitory
nerves have been cut, and it seems to result mainly
signals to the first limb and depresses its degree of
from mutually reciprocal inhibition circuits within the
flexion. Finally, removal of the stronger reflex allows
matrix of the cord itself, oscillating between the
the original reflex to reassume its previous intensity.
neurons controlling agonist and antagonist muscles.
The sensory signals from the footpads and from the
position sensors around the joints play a strong role in
Reflexes of Posture and
controlling foot pressure and frequency of stepping
Locomotion
when the foot is allowed to walk along a surface. In
fact, the cord mechanism for control of stepping can
Postural and Locomotive Reflexes
be even more complex. For instance, if the top of the
of the Cord
foot encounters an obstruction during forward thrust,
the forward thrust will stop temporarily; then, in rapid
Positive Supportive Reaction. Pressure on the footpad of
sequence, the foot will be lifted higher and proceed
a decerebrate animal causes the limb to extend against
forward to be placed over the obstruction. This is the
the pressure applied to the foot. Indeed, this reflex
stumble reflex. Thus, the cord is an intelligent walking
is so strong that if an animal whose spinal cord has
controller.
been transected for several months—that is, after the
reflexes have become exaggerated—is placed on its
Reciprocal Stepping of Opposite Limbs. If the lumbar spinal
feet, the reflex often stiffens the limbs sufficiently to
cord is not split down its center, every time stepping
support the weight of the body. This reflex is called the
occurs in the forward direction in one limb, the oppo-
positive supportive reaction.
site limb ordinarily moves backward. This effect results
The positive supportive reaction involves a complex
from reciprocal innervation between the two limbs.
circuit in the interneurons similar to the circuits
responsible for the flexor and cross extensor reflexes.
Diagonal Stepping of All Four Limbs—“Mark Time” Reflex. If
The locus of the pressure on the pad of the foot deter-
a well-healed spinal animal (with spinal transection in
mines the direction in which the limb will extend; pres-
the neck above the forelimb area of the cord) is held
sure on one side causes extension in that direction, an
up from the floor and its legs are allowed to dangle, as
effect called the magnet reaction. This helps keep an
shown in Figure 54-12, the stretch on the limbs occa-
animal from falling to that side.
sionally elicits stepping reflexes that involve all four
limbs. In general, stepping occurs diagonally between
Cord “Righting” Reflexes. When a spinal animal is laid on
the forelimbs and hindlimbs. This diagonal response is
its side, it will make incoordinate movements trying to
another manifestation of reciprocal innervation, this
Chapter 54
Motor Functions of the Spinal Cord; the Cord Reflexes
683
Spinal Cord Reflexes That
Cause Muscle Spasm
In human beings, local muscle spasm is often observed.
In many, if not most, instances, localized pain is the
cause of the local spasm.
Muscle Spasm Resulting from a Broken Bone. One type of
clinically important spasm occurs in muscles that sur-
round a broken bone. This results from pain impulses
initiated from the broken edges of the bone, which
cause the muscles that surround the area to contract
tonically. Pain relief obtained by injecting a local anes-
thetic at the broken edges of the bone relieves the
spasm; a deep general anesthetic of the entire body, such
as ether anesthesia, also relieves the spasm. One of
Figure 54-12
these two anesthetic procedures is often necessary
before the spasm can be overcome sufficiently for the
Diagonal stepping movements exhibited by a spinal animal.
two ends of the bone to be set back into their appro-
priate positions.
Abdominal Muscle Spasm in Peritonitis. Another type of
local spasm caused by cord reflexes is abdominal spasm
resulting from irritation of the parietal peritoneum by
time occurring the entire distance up and down the
peritonitis. Here again, relief of the pain caused by the
cord between the forelimbs and hindlimbs. Such a
peritonitis allows the spastic muscle to relax. The same
walking pattern is called a mark time reflex.
type of spasm often occurs during surgical operations;
for instance, during abdominal operations, pain im-
Galloping Reflex. Another type of reflex that occasion-
pulses from the parietal peritoneum often cause the
ally develops in a spinal animal is the galloping reflex,
abdominal muscles to contract extensively, sometimes
in which both forelimbs move backward in unison
extruding the intestines through the surgical wound. For
while both hindlimbs move forward. This often occurs
this reason, deep anesthesia is usually required for intra-
abdominal operations.
when almost equal stretch or pressure stimuli are
applied to the limbs on both sides of the body at the
Muscle Cramps. Still another type of local spasm is the
same time; unequal stimulation elicits the diagonal
typical muscle cramp. Electromyographic studies indi-
walking reflex. This is in keeping with the normal pat-
cate that the cause of at least some muscle cramps is as
terns of walking and galloping, because in walking,
follows: Any local irritating factor or metabolic abnor-
only one forelimb and one hindlimb at a time are stim-
mality of a muscle, such as severe cold, lack of blood
ulated, which would predispose the animal to continue
flow, or overexercise, can elicit pain or other sensory
walking. Conversely, when the animal strikes the
signals transmitted from the muscle to the spinal cord,
ground during galloping, both forelimbs and both
which in turn cause reflex feedback muscle contraction.
hindlimbs are stimulated about equally; this predis-
The contraction is believed to stimulate the same
sensory receptors even more, which causes the spinal
poses the animal to keep galloping and, therefore, con-
cord to increase the intensity of contraction. Thus, pos-
tinues this pattern of motion.
itive feedback develops, so that a small amount of initial
irritation causes more and more contraction until a full-
blown muscle cramp ensues.
Scratch Reflex
An especially important cord reflex in some animals is
the scratch reflex, which is initiated by itch or tickle sen-
Autonomic Reflexes in the
sation. It involves two functions: (1) a position sense that
Spinal Cord
allows the paw to find the exact point of irritation on
the surface of the body, and (2) a to-and-fro scratching
Many types of segmental autonomic reflexes are inte-
movement.
grated in the spinal cord, most of which are discussed in
The position sense of the scratch reflex is a highly
other chapters. Briefly, these include (1) changes in vas-
developed function. If a flea is crawling as far forward
cular tone resulting from changes in local skin heat (see
as the shoulder of a spinal animal, the hind paw can still
Chapter 73); (2) sweating, which results from localized
find its position, even though 19 muscles in the limb
heat on the surface of the body (see Chapter 73); (3)
must be contracted simultaneously in a precise pattern
intestinointestinal reflexes that control some motor
to bring the paw to the position of the crawling flea. To
functions of the gut (see Chapter 62); (4) peritoneoin-
make the reflex even more complicated, when the flea
testinal reflexes that inhibit gastrointestinal motility in
crosses the midline, the first paw stops scratching and
response to peritoneal irritation (see Chapter 66); and
the opposite paw begins the to-and-fro motion and
(5) evacuation reflexes for emptying the full bladder
eventually finds the flea.
(see Chapter 31) or the colon (see Chapter 63). In addi-
The to-and-fro movement, like the stepping move-
tion, all the segmental reflexes can at times be elicited
ments of locomotion, involves reciprocal innervation
simultaneously in the form of the so-called mass reflex,
circuits that cause oscillation.
described next.
684
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Mass Reflex. In a spinal animal or human being, some-
stretch reflexes, followed in order by the
times the spinal cord suddenly becomes excessively
progressively more complex reflexes: flexor reflexes,
active, causing massive discharge in large portions of the
postural antigravity reflexes, and remnants of
cord. The usual stimulus that causes this is a strong pain
stepping reflexes.
stimulus to the skin or excessive filling of a viscus, such
3. The sacral reflexes for control of bladder and colon
as overdistention of the bladder or the gut. Regardless
evacuation are suppressed in human beings for the
of the type of stimulus, the resulting reflex, called the
first few weeks after cord transection, but in most
mass reflex, involves large portions or even all of the
cases they eventually return. These effects are
cord. The effects are (1) a major portion of the body’s
discussed in Chapters 31 and 66.
skeletal muscles goes into strong flexor spasm; (2) the
colon and bladder are likely to evacuate; (3) the arte-
rial pressure often rises to maximal values, sometimes
References
to a systolic pressure well over 200 mm Hg; and (4) large
areas of the body break out into profuse sweating.
Buffelli M, Busetto G, Bidoia C, et al: Activity-dependent
Because the mass reflex can last for minutes, it pre-
synaptic competition at mammalian neuromuscular junc-
sumably results from activation of great numbers of
tions. News Physiol Sci 19:85, 2004.
reverberating circuits that excite large areas of the cord
Chen HH, Hippenmeyer S, Arber S, Frank E: Development
at once. This is similar to the mechanism of epileptic
of the monosynaptic stretch reflex circuit. Curr Opin
seizures, which involve reverberating circuits that occur
Neurobiol 13:96, 2003.
in the brain instead of in the cord.
Dietz V: Proprioception and locomotor disorders. Nat Rev
Neurosci 3:781, 2002.
Dietz V: Spinal cord pattern generators for locomotion. Clin
Spinal Cord Transection and
Neurophysiol 114:1379, 2003.
Spinal Shock
Dobkin BH, Havton LA: Basic advances and new avenues
in therapy of spinal cord injury. Annu Rev Med 55:255,
When the spinal cord is suddenly transected in the
2004.
upper neck, at first, essentially all cord functions, includ-
Duysens J, Clarac F, Cruse H: Load-regulating mechanisms
ing the cord reflexes, immediately become depressed to
in gait and posture: comparative aspects. Physiol Rev
the point of total silence, a reaction called spinal shock.
80:83, 2000.
The reason for this is that normal activity of the cord
Garwicz M: Spinal reflexes provide motor error signals to
neurons depends to a great extent on continual tonic
cerebellar modules—relevance for motor coordination.
excitation by the discharge of nerve fibers entering the
Brain Res Brain Res Rev 40:152, 2002.
cord from higher centers, particularly discharge trans-
Glover JC: Development of specific connectivity between
mitted through the reticulospinal tracts, vestibulospinal
premotor neurons and motoneurons in the brain stem and
tracts, and corticospinal tracts.
spinal cord. Physiol Rev 80:615, 2000.
After a few hours to a few weeks, the spinal neurons
Grillner S: The motor infrastructure: from ion channels to
gradually regain their excitability. This seems to be a
neuronal networks. Nat Rev Neurosci 4:573, 2003.
natural characteristic of neurons everywhere in the
Grillner S: Muscle twitches during sleep shape the precise
nervous system—that is, after they lose their source of
muscles of the withdrawal reflex. Trends Neurosci 27:169,
facilitatory impulses, they increase their own natural
2004.
degree of excitability to make up at least partially for
Guthrie S: Neuronal development: putting motor neurons in
the loss. In most nonprimates, excitability of the cord
their place. Curr Biol 14:R166, 2004.
centers returns essentially to normal within a few hours
Haines DE: Fundamental Neuroscience. New York:
to a day or so, but in human beings, the return is often
Churchill Livingstone, 1997.
delayed for several weeks and occasionally is never
Haines DE, Lancon JA: Review of Neuroscience. New York:
complete; conversely, sometimes recovery is excessive,
Churchill Livingstone, 2003.
with resultant hyperexcitability of some or all cord
Helms AW, Johnson JE: Specification of dorsal spinal cord
functions.
interneurons. Curr Opin Neurobiol 13:42, 2003.
Some of the spinal functions specifically affected
Jankowska E: Spinal interneuronal systems: identification,
during or after spinal shock are the following:
multifunctional character and reconfigurations in
1. At onset of spinal shock, the arterial blood
mammals. J Physiol 533:31, 2001.
pressure falls instantly and drastically—sometimes
Jankowska E, Hammar I: Spinal interneurons: how can
to as low as 40 mm Hg—thus demonstrating that
studies in animals contribute to the understanding of
sympathetic nervous system activity becomes
spinal interneuronal systems in man? Brain Res Brain Res
blocked almost to extinction. The pressure
Rev 40:19, 2002.
ordinarily returns to normal within a few days, even
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
in human beings.
Science, 4th ed. New York: McGraw-Hill, 2000.
2. All skeletal muscle reflexes integrated in the spinal
Marder E, Bucher D: Central pattern generators and the
cord are blocked during the initial stages of shock.
control of rhythmic movements. Curr Biol 11:R986, 2001.
In lower animals, a few hours to a few days are
Marder E, Prinz AA: Current compensation in neuronal
required for these reflexes to return to normal;
homeostasis. Neuron 37:2, 2003.
in human beings, 2 weeks to several months are
Pearson KG: Generating the walking gait: role of sensory
sometimes required. In both animals and
feedback. Prog Brain Res 143:123, 2004.
humans, some reflexes may eventually become
Rekling JC, Funk GD, Bayliss DA, et al: Synaptic control of
hyperexcitable, particularly if a few facilitatory
motoneuronal excitability. Physiol Rev 80:767, 2000.
pathways remain intact between the brain and the
Rossignol S, Bouyer L, Barthelemy D, et al: Recovery of
cord while the remainder of the spinal cord is
locomotion in the cat following spinal cord lesions. Brain
transected. The first reflexes to return are the
Res Brain Res Rev 40:257, 2002.
C H A P T E R
5
5
Cortical and Brain Stem Control
of Motor Function
In this chapter, we discuss control of body move-
ments by the cerebral cortex and brain stem.
Most “voluntary” movements initiated by the
cerebral cortex are achieved when the cortex acti-
vates “patterns” of function stored in lower brain
areas—the cord, brain stem, basal ganglia, and cere-
bellum. These lower centers, in turn, send specific
control signals to the muscles.
For a few types of movements, however, the cortex has almost a direct
pathway to the anterior motor neurons of the cord, bypassing some motor
centers on the way. This is especially true for control of the fine dexterous move-
ments of the fingers and hands. This chapter and Chapter 56 explain the inter-
play among the different motor areas of the brain and spinal cord to provide
overall synthesis of voluntary motor function.
MOTOR CORTEX AND CORTICOSPINAL TRACT
Figure 55-1 shows the functional areas of the cerebral cortex. Anterior to the
central cortical sulcus, occupying approximately the posterior one third of
the frontal lobes, is the motor cortex. Posterior to the central sulcus is the
somatosensory cortex (an area discussed in detail in earlier chapters), which
feeds the motor cortex many of the signals that initiate motor activities.
The motor cortex itself is divided into three subareas, each of which has its
own topographical representation of muscle groups and specific motor func-
tions: (1) the primary motor cortex, (2) the premotor area, and (3) the supple-
mentary motor area.
Primary Motor Cortex
The primary motor cortex, shown in Figure 55-1, lies in the first convolution of
the frontal lobes anterior to the central sulcus. It begins laterally in the sylvian
fissure, spreads superiorly to the uppermost portion of the brain, and then dips
deep into the longitudinal fissure. (This area is the same as area 4 in Brodmann’s
classification of the brain cortical areas, shown in Figure 47-5.)
Figure 55-1 lists the approximate topographical representations of the dif-
ferent muscle areas of the body in the primary motor cortex, beginning with the
face and mouth region near the sylvian fissure; the arm and hand area, in the
midportion of the primary motor cortex; the trunk, near the apex of the brain;
and the leg and foot areas, in the part of the primary motor cortex that dips into
the longitudinal fissure. This topographical organization is demonstrated even
more graphically in Figure 55-2, which shows the degrees of representation of
the different muscle areas as mapped by Penfield and Rasmussen. This mapping
was done by electrically stimulating the different areas of the motor cortex in
human beings who were undergoing neurosurgical operations. Note that more
than one half of the entire primary motor cortex is concerned with controlling
the muscles of the hands and the muscles of speech. Point stimulation in these
hand and speech motor areas on rare occasion causes contraction of a single
685
686
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Motor
Sensory
Premotor Area
Primary
motor
Somatic
The premotor area, also shown in Figure 55-1, lies 1
Supplementary
cortex
area 1
to 3 centimeters anterior to the primary motor cortex,
area
Somatic
extending inferiorly into the sylvian fissure and supe-
Legs
association
riorly into the longitudinal fissure, where it abuts
area
Feet
the supplementary motor area, which has functions
similar to those of the premotor area. The topograph-
Trunk
ical organization of the premotor cortex is roughly the
Arm
6
7
same as that of the primary motor cortex, with the
4
5
Hand
mouth and face areas located most laterally; as one
Face
moves upward, the hand, arm, trunk, and leg areas are
encountered.
Mouth
Nerve signals generated in the premotor area cause
much more complex “patterns” of movement than
the discrete patterns generated in the primary motor
cortex. For instance, the pattern may be to position
the shoulders and arms so that the hands are properly
Premotor
area
oriented to perform specific tasks. To achieve these
results, the most anterior part of the premotor area
Figure 55-1
first develops a “motor image” of the total muscle
movement that is to be performed. Then, in the poste-
Motor and somatosensory functional areas of the cerebral cortex.
rior premotor cortex, this image excites each succes-
The numbers 4, 5, 6, and 7 are Brodmann’s cortical areas, as
explained in Chapter 47.
sive pattern of muscle activity required to achieve the
image. This posterior part of the premotor cortex sends
its signals either directly to the primary motor cortex
to excite specific muscles or, often, by way of the basal
ganglia and thalamus back to the primary motor
cortex. Thus, the premotor cortex, basal ganglia, thal-
amus, and primary motor cortex constitute a complex
overall system for the control of complex patterns of
coordinated muscle activity.
Supplementary Motor Area
The supplementary motor area has yet another topo-
graphical organization for the control of motor func-
Lips
tion. It lies mainly in the longitudinal fissure but
extends a few centimeters onto the superior frontal
cortex. Contractions elicited by stimulating this area
are often bilateral rather than unilateral. For instance,
Mastication
stimulation frequently leads to bilateral grasping
Salivation
movements of both hands simultaneously; these
movements are perhaps rudiments of the hand func-
tions required for climbing. In general, this area func-
tions in concert with the premotor area to provide
body-wide attitudinal movements, fixation movements
Figure 55-2
of the different segments of the body, positional
movements of the head and eyes, and so forth, as back-
Degree of representation of the different muscles of the body in
ground for the finer motor control of the arms and
the motor cortex. (Redrawn from Penfield W, Rasmussen T: The
Cerebral Cortex of Man: A Clinical Study of Localization of Func-
hands by the premotor area and primary motor cortex.
tion. New York: Hafner, 1968.)
Some Specialized Areas of Motor
muscle; most often, stimulation contracts a group of
muscles instead. To express this in another way, exci-
Control Found in the Human
tation of a single motor cortex neuron usually excites
Motor Cortex
a specific movement rather than one specific muscle.
To do this, it excites a “pattern” of separate muscles,
Neurosurgeons have found a few highly specialized
each of which contributes its own direction and
motor regions of the human cerebral cortex (shown in
strength of muscle movement.
Figure 55-3) that control specific motor functions.
Chapter 55
Cortical and Brain Stem Control of Motor Function
687
Supplemental
Primary
Area for Hand Skills. In the premotor area immediately
and premotor
motor
anterior to the primary motor cortex for the hands and
areas
cortex
fingers is a region neurosurgeons have identified as
important for “hand skills.” That is, when tumors or
other lesions cause destruction in this area, hand
movements become uncoordinated and nonpurpose-
ful, a condition called motor apraxia.
Hand skills
Head rotation
Choice
Transmission of Signals from the
Contralateral
Lips
of words
Vocalization
Motor Cortex to the Muscles
eye movements
Jaw
Tongue
Swallowing
Eye
Motor signals are transmitted directly from the cortex
Chewing
fixation
to the spinal cord through the corticospinal tract and
indirectly through multiple accessory pathways that
Word formation
involve the basal ganglia, cerebellum, and various
(Broca’s area)
nuclei of the brain stem. In general, the direct pathways
are concerned more with discrete and detailed move-
ments, especially of the distal segments of the limbs,
Figure 55-3
particularly the hands and fingers.
Representation of the different muscles of the body in the motor
Corticospinal (Pyramidal) Tract
cortex and location of other cortical areas responsible for specific
The most important output pathway from the motor
types of motor movements.
cortex is the corticospinal tract, also called the pyram-
idal tract, shown in Figure 55-4. The corticospinal tract
originates about 30 per cent from the primary motor
cortex, 30 per cent from the premotor and supple-
These regions have been localized either by electrical
mentary motor areas, and
40 per cent from the
stimulation or by noting the loss of motor function
somatosensory areas posterior to the central sulcus.
when destructive lesions occur in specific cortical
After leaving the cortex, it passes through the pos-
areas. Some of the more important regions are the
terior limb of the internal capsule
(between the
following.
caudate nucleus and the putamen of the basal ganglia)
and then downward through the brain stem, forming
Broca’s Area and Speech. Figure 55-3 shows a premotor
the pyramids of the medulla. The majority of the
area labeled
“word formation” lying immediately
pyramidal fibers then cross in the lower medulla to the
anterior to the primary motor cortex and immediately
opposite side and descend into the lateral corticospinal
above the sylvian fissure. This region is called Broca’s
tracts of the cord, finally terminating principally on the
area. Damage to it does not prevent a person from
interneurons in the intermediate regions of the cord
vocalizing, but it does make it impossible for the
gray matter; a few terminate on sensory relay neurons
person to speak whole words rather than uncoordi-
in the dorsal horn, and a very few terminate directly
nated utterances or an occasional simple word such as
on the anterior motor neurons that cause muscle
“no” or “yes.” A closely associated cortical area also
contraction.
causes appropriate respiratory function, so that respi-
A few of the fibers do not cross to the opposite side
ratory activation of the vocal cords can occur simulta-
in the medulla but pass ipsilaterally down the cord in
neously with the movements of the mouth and tongue
the ventral corticospinal tracts. Many if not most of
during speech. Thus, the premotor neuronal activities
these fibers eventually cross to the opposite side of the
related to speech are highly complex.
cord either in the neck or in the upper thoracic region.
These fibers may be concerned with control of bilat-
“Voluntary” Eye Movement Field. In the premotor area
eral postural movements by the supplementary motor
immediately above Broca’s area is a locus for control-
cortex.
ling voluntary eye movements. Damage to this area
The most impressive fibers in the pyramidal tract are
prevents a person from voluntarily moving the eyes
a population of large myelinated fibers with a mean
toward different objects. Instead, the eyes tend to lock
diameter of 16 micrometers. These fibers originate
involuntarily onto specific objects, an effect controlled
from giant pyramidal cells, called Betz cells, that are
by signals from the occipital visual cortex, as explained
found only in the primary motor cortex. The Betz cells
in Chapter 51. This frontal area also controls eyelid
are about 60 micrometers in diameter, and their fibers
movements such as blinking.
transmit nerve impulses to the spinal cord at a veloc-
ity of about 70 m/sec, the most rapid rate of transmis-
Head Rotation Area. Slightly higher in the motor associ-
sion of any signals from the brain to the cord. There
ation area, electrical stimulation elicits head rotation.
are about 34,000 of these large Betz cell fibers in each
This area is closely associated with the eye movement
corticospinal tract. The total number of fibers in each
field; it directs the head toward different objects.
corticospinal tract is more than 1 million, so these large
688
Unit XI
The Nervous System: C. Motor and Integrative Neurophysiology
3. A moderate number of motor fibers pass to red
Motor cortex
nuclei of the midbrain. From these, additional fibers
pass down the cord through the rubrospinal tract.
4. A moderate number of motor fibers deviate into
the reticular substance and vestibular nuclei of the
brain stem; from there, signals go to the cord by
way of reticulospinal and vestibulospinal tracts,
and others go to the cerebellum by way of
reticulocerebellar and vestibulocerebellar tracts.
Posterior limb of internal
5. A tremendous number of motor fibers synapse
capsule
in the pontile nuclei, which give rise to the
pontocerebellar fibers, carrying signals into the
cerebellar hemispheres.
6. Collaterals also terminate in the inferior olivary
nuclei, and from there, secondary olivocerebellar
Genu of corpus callosum
fibers transmit signals to multiple areas of the
cerebellum.
Thus, the basal ganglia, brain stem, and cerebellum all
receive strong motor signals from the corticospinal
system every time a signal is transmitted down the
Basis pedunculi of
spinal cord to cause a motor activity.
mesencephalon
Incoming Fiber Pathways to the
Motor Cortex
Longitudinal fascicles
The functions of the motor cortex are controlled mainly
of pons
by nerve signals from the somatosensory system but
also, to some degree, from other sensory systems such
as hearing and vision. Once the sensory information is
received, the motor cortex operates in association with
the basal ganglia and cerebellum to excite an appropri-
ate course of motor action. The more important incom-
ing fiber pathways to the motor cortex are the following:
Pyramid of medulla
1. Subcortical fibers from adjacent regions of
oblongata
the cerebral cortex, especially from (a) the
somatosensory areas of the parietal cortex, (b) the
adjacent areas of the frontal cortex anterior to
Lateral corticospinal tract
the motor cortex, and (c) the visual and auditory
cortices.
Ventral corticospinal tract
2. Subcortical fibers that arrive through the corpus
callosum from the opposite cerebral hemisphere.
Figure 55-4
These fibers connect corresponding areas of the
cortices in the two sides of the brain.
Pyramidal tract. (Modified from Ranson SW, Clark SL: Anatomy of
3. Somatosensory fibers that arrive directly from the
the Nervous System. Philadelphia: WB Saunders, 1959.)
ventrobasal complex of the thalamus. These relay
mainly cutaneous tactile signals and joint and
fibers represent only 3 per cent of the total. The other
muscle signals from the peripheral body.
97 per cent are mainly fibers smaller than 4 microme-
4. Tracts from the ventrolateral and ventroanterior
ters in diameter that conduct background tonic signals
nuclei of the thalamus, which in turn receive signals
from the cerebellum and basal ganglia. These tracts
to the motor areas of the cord.
provide signals that are necessary for coordination
among the motor control functions of the motor
Other Fiber Pathways from the Motor Cortex
cortex, basal ganglia, and cerebellum.
The motor cortex gives rise to large numbers of addi-
5. Fibers from the intralaminar nuclei of the thalamus.
tional, mainly small fibers that go to deep regions in the
These fibers control the general level of excitability
cerebrum and brain stem, including the following:
of the motor cortex in the same way they control
1. The axons from the giant Betz cells send short
the general level of excitability of most other
collaterals back to the cortex itself. These
regions of the cerebral cortex.
collaterals are believed to inhibit adjacent regions
of the cortex when the Betz cells discharge, thereby
“sharpening” the boundaries of the excitatory
signal.
Red Nucleus Serves as an Alternative
2. A large number of fibers pass from the motor
Pathway for Transmitting Cortical
cortex into the caudate nucleus and putamen. From
Signals to the Spinal Cord
there, additional pathways extend into the brain
stem and spinal cord, as discussed in the next
chapter, mainly to control body postural muscle
The red nucleus, located in the mesencephalon, func-
contractions.
tions in close association with the corticospinal tract.
Chapter 55
Cortical and Brain Stem Control of Motor Function
689
the corticospinal fibers are destroyed but the corti-
Motor
corubrospinal pathway is intact, discrete movements
cortex
can still occur, except that the movements for fine
Corticorubral
control of the fingers and hands are considerably
tract
impaired. Wrist movements are still functional, which
is not the case when the corticorubrospinal pathway is
also blocked.
Therefore, the pathway through the red nucleus to
the spinal cord is associated with the corticospinal
system. Further, the rubrospinal tract lies in the lateral
columns of the spinal cord, along with the corti-
cospinal tract, and terminates on the interneurons and
motor neurons that control the more distal muscles of
Red nucleus
the limbs. Therefore, the corticospinal and rubrospinal
Interpositus
tracts together are called the lateral motor system
nucleus
Reticular formation
of the cord, in contradistinction to a vestibuloreticu-
Dentate
lospinal system, which lies mainly medially in the cord
nucleus
Rubrospinal tract
and is called the medial motor system of the cord, as
Cerebellum
discussed later in this chapter.
“Extrapyramidal” System
Figure 55-5
The term extrapyramidal motor system is widely used in
Corticorubrospinal pathway for motor control, showing also the
clinical circles to denote all those portions of the brain
relation of this pathway to the cerebellum.
and brain stem that contribute to motor control but are
not part of the direct corticospinal-pyramidal system.
These include pathways through the basal ganglia, the
reticular formation of the brain stem, the vestibular
As shown in Figure 55-5, it receives a large number of
nuclei, and often the red nuclei. This is such an all-
direct fibers from the primary motor cortex through
inclusive and diverse group of motor control areas that
it is difficult to ascribe specific neurophysiologic func-
the corticorubral tract, as well as branching fibers from
tions to the so-called extrapyramidal system as a whole.
the corticospinal tract as it passes through the mesen-
For this reason, the term “extrapyramidal” is being used
cephalon. These fibers synapse in the lower portion of
less often both clinically and physiologically.
the red nucleus, the magnocellular portion, which con-
tains large neurons similar in size to the Betz cells in
the motor cortex. These large neurons then give rise to
Excitation of the Spinal Cord Motor
the rubrospinal tract, which crosses to the opposite
side in the lower brain stem and follows a course
Control Areas by the Primary Motor
immediately adjacent and anterior to the corticospinal
Cortex and Red Nucleus
tract into the lateral columns of the spinal cord.
The rubrospinal fibers terminate mostly on the
Vertical Columnar Arrangement of the Neurons in the Motor
interneurons of the intermediate areas of the cord gray
Cortex. In Chapters 47 and 51, we pointed out that the
matter, along with the corticospinal fibers, but some of
cells in the somatosensory cortex and visual cortex are
the rubrospinal fibers terminate directly on anterior
organized in vertical columns of cells. In like manner,
motor neurons, along with some corticospinal fibers.
the cells of the motor cortex are organized in vertical
The red nucleus also has close connections with the
columns a fraction of a millimeter in diameter, with
cerebellum, similar to the connections between the
thousands of neurons in each column.
motor cortex and the cerebellum.
Each column of cells functions as a unit, usually
stimulating a group of synergistic muscles, but some-
Function of the Corticorubrospinal System. The magnocel-
times stimulating just a single muscle. Also, each
lular portion of the red nucleus has a somatographic
column has six distinct layers of cells, as is true
representation of all the muscles of the body, as is true
throughout nearly all the cerebral cortex. The pyram-
of the motor cortex. Therefore, stimulation of a single
idal cells that give rise to the corticospinal fibers all lie
point in this portion of the red nucleus causes con-
in the fifth layer of cells from the cortical surface. Con-
traction of either a single muscle or a small group of
versely, the input signals all enter by way of layers 2
muscles. However, the fineness of representation of
through 4. And the sixth layer gives rise mainly to
the different muscles is far less developed than in the
fibers that communicate with other regions of the cere-
motor cortex. This is especially true in human beings,
bral cortex itself.
who have relatively small red nuclei.
The corticorubrospinal pathway serves as an acces-
Function of Each Column of Neurons. The neurons of
sory route for transmission of relatively discrete
each column operate as an integrative processing
signals from the motor cortex to the spinal cord. When
system, using information from multiple input sources
690
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
to determine the output response from the column. In
Sensory neurons
addition, each column can function as an amplifying
system to stimulate large numbers of pyramidal fibers
Propriospinal tract
to the same muscle or to synergistic muscles simulta-
Interneurons
neously. This is important, because stimulation of a
single pyramidal cell can seldom excite a muscle.
Corticospinal tract
Usually, 50 to 100 pyramidal cells need to be excited
from pyramidal cells
of cortex
simultaneously or in rapid succession to achieve defin-
itive muscle contraction.
Rubrospinal tract
Reticulospinal tract
Dynamic and Static Signals Transmitted by the Py-
ramidal Neurons. If a strong signal is sent to a muscle
Anterior motor
neuron
to cause initial rapid contraction, then a much weaker
continuing signal can maintain the contraction for long
Motor nerve
periods thereafter. This is the usual manner in which
Tectospinal and
excitation is provided to cause muscle contractions. To
reticulospinal tracts
do this, each column of cells excites two populations
Vestibulospinal and
of pyramidal cell neurons, one called dynamic neurons
reticulospinal tracts
and the other static neurons. The dynamic neurons are
Figure 55-6
excessively excited for a short period at the beginning
of a contraction, causing the initial rapid development
Convergence of different motor control pathways on the anterior
of force. Then the static neurons fire at a much slower
motor neurons.
rate, but they continue firing at this slow rate to main-
tain the force of contraction as long as the contraction
is required.
excitation of the muscles and, therefore, increase the
The neurons of the red nucleus have similar
tightness of the hand grasp.
dynamic and static characteristics, except that a
greater percentage of dynamic neurons is in the red
Stimulation of the Spinal Motor Neurons
nucleus and a greater percentage of static neurons is
Figure 55-6 shows a cross section of a spinal cord
in the primary motor cortex. This may be related to the
segment demonstrating (1) multiple motor and senso-
fact that the red nucleus is closely allied with the cere-
rimotor control tracts entering the cord segment and
bellum, and the cerebellum plays an important role in
(2) a representative anterior motor neuron in the
rapid initiation of muscle contraction, as explained in
middle of the anterior horn gray matter. The corti-
the next chapter.
cospinal tract and the rubrospinal tract lie in the dorsal
portions of the lateral white columns. Their fibers ter-
Somatosensory Feedback to the Motor
minate mainly on interneurons in the intermediate
Cortex Helps Control the Precision of
area of the cord gray matter.
Muscle Contraction
In the cervical enlargement of the cord where the
When nerve signals from the motor cortex cause a
hands and fingers are represented, large numbers of
muscle to contract, somatosensory signals return all
both corticospinal and rubrospinal fibers also termi-
the way from the activated region of the body to the
nate directly on the anterior motor neurons, thus
neurons in the motor cortex that are initiating the
allowing a direct route from the brain to activate
action. Most of these somatosensory signals arise in (1)
muscle contraction. This is in keeping with the fact that
the muscle spindles, (2) the tendon organs of the
the primary motor cortex has an extremely high
muscle tendons, or (3) the tactile receptors of the skin
degree of representation for fine control of hand,
overlying the muscles. These somatic signals often
finger, and thumb actions.
cause positive feedback enhancement of the muscle
contraction in the following ways: In the case of the
Patterns of Movement Elicited by Spinal Cord Centers. From
muscle spindles, if the fusimotor muscle fibers in the
Chapter 54, recall that the spinal cord can provide
spindles contract more than the large skeletal muscle
certain specific reflex patterns of movement in
fibers contract, the central portions of the spindles
response to sensory nerve stimulation. Many of these
become stretched and, therefore, excited. Signals from
same patterns are also important when the cord’s ante-
these spindles then return rapidly to the pyramidal
rior motor neurons are excited by signals from the
cells in the motor cortex to signal them that the large
brain. For example, the stretch reflex is functional at
muscle fibers have not contracted enough. The pyram-
all times, helping to damp any oscillations of the motor
idal cells further excite the muscle, helping its con-
movements initiated from the brain, and probably also
traction to catch up with the contraction of the muscle
providing at least part of the motive power required
spindles. In the case of the tactile receptors, if the
to cause muscle contractions when the intrafusal fibers
muscle contraction causes compression of the skin
of the muscle spindles contract more than the large
against an object, such as compression of the fingers
skeletal muscle fibers do, thus eliciting reflex “servo-
around an object being grasped, the signals from
assist” stimulation of the muscle, in addition to the
the skin receptors can, if necessary, cause further
direct stimulation by the corticospinal fibers.
Chapter 55
Cortical and Brain Stem Control of Motor Function
691
Also, when a brain signal excites a muscle, it usually
Role of the Brain Stem in
is not necessary to transmit an inverse signal to relax
Controlling Motor Function
the antagonist muscle at the same time; this is achieved
by the reciprocal innervation circuit that is always
The brain stem consists of the medulla, pons, and mes-
present in the cord for coordinating the function of
encephalon. In one sense, it is an extension of the
antagonistic pairs of muscles.
spinal cord upward into the cranial cavity, because it
Finally, other cord reflex mechanisms, such as
contains motor and sensory nuclei that perform motor
withdrawal, stepping and walking, scratching, and
and sensory functions for the face and head regions in
postural mechanisms, can each be activated by
the same way that the spinal cord performs these func-
“command” signals from the brain. Thus, simple
tions from the neck down. But in another sense, the
command signals from the brain can initiate many
brain stem is its own master because it provides many
normal motor activities, particularly for such functions
special control functions, such as the following:
as walking and attaining different postural attitudes of
1. Control of respiration
the body.
2. Control of the cardiovascular system
3. Partial control of gastrointestinal function
Effect of Lesions in the Motor Cortex or in the Corticospinal
4. Control of many stereotyped movements of the
Pathway—The “Stroke”
body
The motor control system can be damaged by the
5. Control of equilibrium
common abnormality called a “stroke.” This is caused
6. Control of eye movements
either by a ruptured blood vessel that hemorrhages into
the brain or by thrombosis of one of the major arteries
Finally, the brain stem serves as a way station for
supplying the brain. In either case, the result is loss of
“command signals” from higher neural centers. In the
blood supply to the cortex or to the corticospinal tract
following sections, we discuss the role of the brain stem
where it passes through the internal capsule between
in controlling whole-body movement and equilibrium.
the caudate nucleus and the putamen. Also, experiments
Especially important for these purposes are the brain
have been performed in animals to selectively remove
stem’s reticular nuclei and vestibular nuclei.
different parts of the motor cortex.
Removal of the Primary Motor Cortex
(Area Pyramidalis).
Support of the Body Against Gravity—
Removal of a portion of the primary motor cortex—the
Roles of the Reticular and
area that contains the giant Betz pyramidal cells—
causes varying degrees of paralysis of the represented
Vestibular Nuclei
muscles. If the sublying caudate nucleus and adjacent
premotor and supplementary motor areas are not
Figure 55-7 shows the locations of the reticular and
damaged, gross postural and limb “fixation” movements
vestibular nuclei in the brain stem.
can still occur, but there is loss of voluntary control of
discrete movements of the distal segments of the limbs,
especially of the hands and fingers. This does not mean
that the hand and finger muscles themselves cannot con-
tract; rather, the ability to control the fine movements is
gone. From these observations, one can conclude that
the area pyramidalis is essential for voluntary initiation
of finely controlled movements, especially of the hands
and fingers.
Pontine reticular
nuclei
Muscle Spasticity Caused by Lesions That Damage Large Areas
Adjacent to the Motor Cortex. The primary motor cortex
normally exerts a continual tonic stimulatory effect on
the motor neurons of the spinal cord; when this stimu-
latory effect is removed, hypotonia results. Most lesions
Vestibular nuclei
of the motor cortex, especially those caused by a stroke,
involve not only the primary motor cortex but also adja-
cent parts of the brain such as the basal ganglia. In these
instances, muscle spasm almost invariably occurs in the
Medullary reticular
afflicted muscle areas on the opposite side of the body
nuclei
(because the motor pathways cross to the opposite
side). This spasm results mainly from damage to acces-
sory pathways from the nonpyramidal portions of the
motor cortex. These pathways normally inhibit the
vestibular and reticular brain stem motor nuclei. When
these nuclei cease their state of inhibition (i.e., are “dis-
inhibited”), they become spontaneously active and
cause excessive spastic tone in the involved muscles, as
we discuss more fully later in the chapter. This is the
Figure 55-7
spasticity that normally accompanies a “stroke” in a
human being.
Locations of the reticular and vestibular nuclei in the brain stem.
692
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Excitatory-Inhibitory Antagonism Between
signals from the pontine reticular system, so that under
Pontine and Medullary Reticular Nuclei
normal conditions, the body muscles are not abnor-
The reticular nuclei are divided into two major groups:
mally tense.
(1) pontine reticular nuclei, located slightly posteriorly
Yet some signals from higher areas of the brain can
and laterally in the pons and extending into the mes-
“disinhibit” the medullary system when the brain
encephalon, and (2) medullary reticular nuclei, which
wishes to excite the pontine system to cause standing.
extend through the entire medulla, lying ventrally and
At other times, excitation of the medullary reticular
medially near the midline. These two sets of nuclei
system can inhibit antigravity muscles in certain por-
function mainly antagonistically to each other, with
tions of the body to allow those portions to perform
the pontine exciting the antigravity muscles and the
special motor activities. The excitatory and inhibitory
medullary relaxing these same muscles.
reticular nuclei constitute a controllable system that is
manipulated by motor signals from the cerebral cortex
Pontine Reticular System. The pontine reticular nuclei
and elsewhere to provide necessary background
transmit excitatory signals downward into the cord
muscle contractions for standing against gravity and to
through the pontine reticulospinal tract in the anterior
inhibit appropriate groups of muscles as needed so
column of the cord, as shown in Figure 55-8. The fibers
that other functions can be performed.
of this pathway terminate on the medial anterior
motor neurons that excite the axial muscles of the
Role of the Vestibular Nuclei to Excite the
body, which support the body against gravity—that is,
Antigravity Muscles
the muscles of the vertebral column and the extensor
All the vestibular nuclei, shown in Figure 55-7, func-
muscles of the limbs.
tion in association with the pontine reticular nuclei to
The pontine reticular nuclei have a high degree of
control the antigravity muscles. The vestibular nuclei
natural excitability. In addition, they receive strong
transmit strong excitatory signals to the antigravity
excitatory signals from the vestibular nuclei, as well as
muscles by way of the lateral and medial vestibu-
from deep nuclei of the cerebellum. Therefore, when
lospinal tracts in the anterior columns of the spinal
the pontine reticular excitatory system is unopposed
cord, as shown in Figure 55-8. Without this support of
by the medullary reticular system, it causes powerful
the vestibular nuclei, the pontine reticular system
excitation of antigravity muscles throughout the body,
would lose much of its excitation of the axial anti-
so much so that four-legged animals can be placed in
gravity muscles.
a standing position, supporting the body against
The specific role of the vestibular nuclei, however,
gravity without any signals from higher levels of the
is to selectively control the excitatory signals to the dif-
brain.
ferent antigravity muscles to maintain equilibrium in
response to signals from the vestibular apparatus. We
Medullary Reticular System. The medullary reticular
discuss this more fully later in the chapter.
nuclei transmit inhibitory signals to the same anti-
gravity anterior motor neurons by way of a different
The Decerebrate Animal Develops Spastic Rigidity
tract, the medullary reticulospinal tract, located in the
When the brain stem of an animal is sectioned below
lateral column of the cord, as also shown in Figure
the midlevel of the mesencephalon, but the pontine and
55-8. The medullary reticular nuclei receive strong
medullary reticular systems as well as the vestibular
system are left intact, the animal develops a condition
input collaterals from (1) the corticospinal tract, (2)
called decerebrate rigidity. This rigidity does not occur
the rubrospinal tract, and (3) other motor pathways.
in all muscles of the body but does occur in the anti-
These normally activate the medullary reticular
gravity muscles—the muscles of the neck and trunk and
inhibitory system to counterbalance the excitatory
the extensors of the legs.
The cause of decerebrate rigidity is blockage of nor-
mally strong input to the medullary reticular nuclei
from the cerebral cortex, the red nuclei, and the basal
ganglia. Lacking this input, the medullary reticular
inhibitor system becomes nonfunctional; full overactiv-
ity of the pontine excitatory system occurs, and rigidity
Medullary reticulospinal
develops. We shall see later that other causes of rigidity
tract
occur in other neuromotor diseases, especially lesions of
the basal ganglia.
Vestibular Sensations and
Lateral vestibulospinal
tract
Maintenance of Equilibrium
Medial vestibulospinal tract
Pontine reticulospinal tract
Vestibular Apparatus
Figure 55-8
The vestibular apparatus, shown in Figure 55-9, is the
Vestibulospinal and reticulospinal tracts descending in the spinal
cord to excite (solid lines) or inhibit (dashed lines) the anterior
sensory organ for detecting sensations of equilibrium.
motor neurons that control the body’s axial musculature.
It is encased in a system of bony tubes and chambers
Chapter 55
Cortical and Brain Stem Control of Motor Function
693
Anterior
Ampullae
Kinocilium
Utricle
Maculae and
Stereocilia
statoconia
Semi-
Filamentous
circular
attachments
canals
Ductus
Saccule
Posterior
cochlearis
Crista ampullaris
Ductus endolymphaticus
MEMBRANOUS LABYRINTH
Gelatinous
Statoconia
mass of
cupula
Gelatinous
Hair tufts
layer
Hair tufts
Hair
Hair cells
cells
Nerve fibers
Nerve
fibers
Sustentacular cells
Sustentacular cells
Nerve fiber
CRISTA AMPULLARIS AND MACULA
Figure 55-9
Membranous labyrinth, and organization of the crista ampullaris
and the macula.
Figure 55-10
Hair cell of the equilibrium apparatus and its synapses with the
vestibular nerve.
located in the petrous portion of the temporal bone,
called the bony labyrinth. Within this system are mem-
branous tubes and chambers called the membranous
of the utricle and plays an important role in deter-
labyrinth. The membranous labyrinth is the functional
mining orientation of the head when the head is
part of the vestibular apparatus.
upright. Conversely, the macula of the saccule is
The top of Figure 55-9 shows the membranous
located mainly in a vertical plane and signals head
labyrinth. It is composed mainly of the cochlea (ductus
orientation when the person is lying down.
cochlearis); three semicircular canals; and two large
Each macula is covered by a gelatinous layer in
chambers, the utricle and saccule. The cochlea is the
which many small calcium carbonate crystals called
major sensory organ for hearing (see Chapter 52) and
statoconia are embedded. Also in the macula are thou-
has little to do with equilibrium. However, the semi-
sands of hair cells, one of which is shown in Figure
circular canals, the utricle, and the saccule are all inte-
55-10; these project cilia up into the gelatinous layer.
gral parts of the equilibrium mechanism.
The bases and sides of the hair cells synapse with
sensory endings of the vestibular nerve.
“Maculae”—Sensory Organs of the Utricle and Saccule for
The calcified statoconia have a specific gravity two
Detecting Orientation of the Head with Respect to Gravity.
to three times the specific gravity of the surrounding
Located on the inside surface of each utricle and
fluid and tissues. The weight of the statoconia bends
saccule, shown in the top diagram of Figure 55-9, is a
the cilia in the direction of gravitational pull.
small sensory area slightly over 2 millimeters in diam-
eter called a macula. The macula of the utricle lies
Directional Sensitivity of the Hair Cells—Kinocilium. Each
mainly in the horizontal plane on the inferior surface
hair cell has 50 to 70 small cilia called stereocilia, plus
694
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
one large cilium, the kinocilium, as shown in Figure
55-10. The kinocilium is always located to one side,
and the stereocilia become progressively shorter
toward the other side of the cell. Minute filamentous
attachments, almost invisible even to the electron
microscope, connect the tip of each stereocilium to the
Cupula
next longer stereocilium and, finally, to the kinocilium.
Cristae
Because of these attachments, when the stereocilia and
Ampulla
ampullaris
kinocilium bend in the direction of the kinocilium, the
filamentous attachments tug in sequence on the ster-
eocilia, pulling them outward from the cell body. This
opens several hundred fluid channels in the neuronal
cell membrane around the bases of the stereocilia,
and these channels are capable of conducting large
numbers of positive ions. Therefore, positive ions pour
into the cell from the surrounding endolymphatic
Hair cells
fluid, causing receptor membrane depolarization. Con-
Nerve
versely, bending the pile of stereocilia in the opposite
direction (backward to the kinocilium) reduces the
tension on the attachments; this closes the ion chan-
nels, thus causing receptor hyperpolarization.
Under normal resting conditions, the nerve fibers
Figure 55-11
leading from the hair cells transmit continuous nerve
impulses at a rate of about 100 per second. When the
Movement of the cupula and its embedded hairs at the onset of
rotation.
stereocilia are bent toward the kinocilium, the impulse
traffic increases, often to several hundred per second;
conversely, bending the cilia away from the kinocilium
decreases the impulse traffic, often turning it off com-
cupula. When a person’s head begins to rotate in any
pletely. Therefore, as the orientation of the head in
direction, the inertia of the fluid in one or more of the
space changes and the weight of the statoconia bends
semicircular ducts causes the fluid to remain station-
the cilia, appropriate signals are transmitted to the
ary while the semicircular duct rotates with the head.
brain to control equilibrium.
This causes fluid to flow from the duct and through the
In each macula, each of the hair cells is oriented in
ampulla, bending the cupula to one side, as demon-
a different direction so that some of the hair cells are
strated by the position of the colored cupula in Figure
stimulated when the head bends forward, some are
55-11. Rotation of the head in the opposite direction
stimulated when it bends backward, others are stimu-
causes the cupula to bend to the opposite side.
lated when it bends to one side, and so forth. There-
Into the cupula are projected hundreds of cilia from
fore, a different pattern of excitation occurs in the
hair cells located on the ampullary crest. The kinocilia
macular nerve fibers for each orientation of the head
of these hair cells are all oriented in the same direc-
in the gravitational field. It is this
“pattern” that
tion in the cupula, and bending the cupula in that
apprises the brain of the head’s orientation in space.
direction causes depolarization of the hair cells,
whereas bending it in the opposite direction hyperpo-
Semicircular Ducts. The three semicircular ducts in each
larizes the cells. Then, from the hair cells, appropriate
vestibular apparatus, known as the anterior, posterior,
signals are sent by way of the vestibular nerve to
and lateral
(horizontal) semicircular ducts, are
apprise the central nervous system of a change in rota-
arranged at right angles to one another so that they
tion of the head and the rate of change in each of the
represent all three planes in space. When the head is
three planes of space.
bent forward about 30 degrees, the lateral semicircu-
lar ducts are approximately horizontal with respect
to the surface of the earth; the anterior ducts are in
Function of the Utricle and Saccule in
vertical planes that project forward and 45 degrees
the Maintenance of Static Equilibrium
outward, whereas the posterior ducts are in
vertical planes that project backward and 45 degrees
It is especially important that the hair cells are all ori-
outward.
ented in different directions in the maculae of the utri-
Each semicircular duct has an enlargement at one
cles and saccules, so that with different positions of the
of its ends called the ampulla, and the ducts and
head, different hair cells become stimulated. The “pat-
ampulla are filled with a fluid called endolymph. Flow
terns” of stimulation of the different hair cells apprise
of this fluid through one of the ducts and through its
the brain of the position of the head with respect to
ampulla excites the sensory organ of the ampulla in
the pull of gravity. In turn, the vestibular, cerebellar,
the following manner: Figure 55-11 shows in each
and reticular motor nerve systems of the brain excite
ampulla a small crest called a crista ampullaris. On top
appropriate postural muscles to maintain proper
of this crista is a loose gelatinous tissue mass, the
equilibrium.
Chapter 55
Cortical and Brain Stem Control of Motor Function
695
This utricle and saccule system functions extremely
effectively for maintaining equilibrium when the head
is in the near-vertical position. Indeed, a person
400
Rotation
can determine as little as half a degree of dysequilib-
rium when the body leans from the precise upright
position.
300
Tonic
Detection of Linear Acceleration by the Utricle and Saccule
200
level of
Maculae. When the body is suddenly thrust forward—
discharge
Stop rotation
that is, when the body accelerates—the statoconia,
100
which have greater mass inertia than the surrounding
fluid, fall backward on the hair cell cilia, and informa-
Begin rotation
tion of dysequilibrium is sent into the nervous centers,
0
causing the person to feel as though he or she were
0
10
20
30
40
50
60
70
80
90
Seconds
falling backward. This automatically causes the person
to lean forward until the resulting anterior shift of the
statoconia exactly equals the tendency for the stato-
conia to fall backward because of the acceleration. At
Figure 55-12
this point, the nervous system senses a state of proper
Response of a hair cell when a semicircular canal is stimulated
equilibrium and leans the body forward no farther.
first by the onset of head rotation and then by stopping rotation.
Thus, the maculae operate to maintain equilibrium
during linear acceleration in exactly the same manner
as they operate during static equilibrium.
The maculae do not operate for the detection of
another 5 to 20 seconds, the cupula slowly returns to
linear velocity. When runners first begin to run, they
its resting position in the middle of the ampulla
must lean far forward to keep from falling backward
because of its own elastic recoil.
because of initial acceleration, but once they have
When the rotation suddenly stops, exactly opposite
achieved running speed, if they were running in a
effects take place: The endolymph continues to rotate
vacuum, they would not have to lean forward. When
while the semicircular duct stops. This time, the cupula
running in air, they lean forward to maintain equilib-
bends in the opposite direction, causing the hair cell to
rium only because of air resistance against their
stop discharging entirely. After another few seconds,
bodies; in this instance, it is not the maculae that make
the endolymph stops moving and the cupula gradually
them lean but air pressure acting on pressure end-
returns to its resting position, thus allowing hair cell
organs in the skin, which initiate appropriate equilib-
discharge to return to its normal tonic level, as shown
rium adjustments to prevent falling.
to the right in Figure 55-12. Thus, the semicircular
duct transmits a signal of one polarity when the head
begins to rotate and of opposite polarity when it stops
Detection of Head Rotation by the
rotating.
Semicircular Ducts
“Predictive” Function of the Semicircular Duct System in the
When the head suddenly begins to rotate in any direc-
Maintenance of Equilibrium. Because the semicircular
tion (called angular acceleration), the endolymph in
ducts do not detect that the body is off balance in the
the semicircular ducts, because of its inertia, tends to
forward direction, in the side direction, or in the back-
remain stationary while the semicircular ducts turn.
ward direction, one might ask: What is the semicircu-
This causes relative fluid flow in the ducts in the direc-
lar ducts’ function in the maintenance of equilibrium?
tion opposite to head rotation.
All they detect is that the person’s head is beginning
Figure 55-12 shows a typical discharge signal from
or stopping to rotate in one direction or another.
a single hair cell in the crista ampullaris when an
Therefore, the function of the semicircular ducts is not
animal is rotated for 40 seconds, demonstrating that
to maintain static equilibrium or to maintain equilib-
(1) even when the cupula is in its resting position, the
rium during steady directional or rotational move-
hair cell emits a tonic discharge of about 100 impulses
ments. Yet loss of function of the semicircular ducts
per second; (2) when the animal begins to rotate, the
does cause a person to have poor equilibrium when
hairs bend to one side and the rate of discharge
attempting to perform rapid, intricate changing body
increases greatly; and
(3) with continued rotation,
movements.
the excess discharge of the hair cell gradually
We can explain the function of the semicircular
subsides back to the resting level during the next few
ducts best by the following illustration: If a person is
seconds.
running forward rapidly and then suddenly begins to
The reason for this adaptation of the receptor is that
turn to one side, he or she will fall off balance a frac-
within the first few seconds of rotation, back resistance
tion of a second later unless appropriate corrections
to the flow of fluid in the semicircular duct and past
are made ahead of time. But the maculae of the utricle
the bent cupula causes the endolymph to begin rotat-
and saccule cannot detect that he or she is off balance
ing as rapidly as the semicircular canal itself; then, in
until after this has occurred. The semicircular ducts,
696
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
however, will have already detected that the person is
Proprioceptive and Exteroceptive Information from Other Parts
turning, and this information can easily apprise the
of the Body. Proprioceptive information from parts of
the body other than the neck is also important in the
central nervous system of the fact that the person will
maintenance of equilibrium. For instance, pressure
fall off balance within the next fraction of a second or
sensations from the footpads tell one
(1) whether
so unless some anticipatory correction is made.
weight is distributed equally between the two feet and
In other words, the semicircular duct mechanism
(2) whether weight on the feet is more forward or
predicts that dysequilibrium is going to occur and
backward.
thereby causes the equilibrium centers to make appro-
Exteroceptive information is especially necessary for
priate anticipatory preventive adjustments. In this way,
the maintenance of equilibrium when a person is
the person need not fall off balance at all before he or
running. The air pressure against the front of the body
she begins to correct the situation.
signals that a force is opposing the body in a direction
Removal of the flocculonodular lobes of the cere-
different from that caused by gravitational pull; as a
result, the person leans forward to oppose this.
bellum prevents normal detection of semicircular duct
signals but has less effect on detecting macular signals.
Importance of Visual Information in the Maintenance of Equilib-
It is especially interesting that the cerebellum serves
rium. After destruction of the vestibular apparatus, and
as a “predictive” organ for most rapid movements of
even after loss of most proprioceptive information from
the body, as well as for those having to do with equi-
the body, a person can still use the visual mechanisms
librium. These other functions of the cerebellum are
reasonably effectively for maintaining equilibrium.
discussed in the following chapter.
Even a slight linear or rotational movement of the body
instantaneously shifts the visual images on the retina,
and this information is relayed to the equilibrium
Vestibular Mechanisms for
centers. Some people with bilateral destruction of the
vestibular apparatus have almost normal equilibrium as
Stabilizing the Eyes
long as their eyes are open and all motions are per-
When a person changes his or her direction of move-
formed slowly. But when moving rapidly or when the
ment rapidly or even leans the head sideways, forward,
eyes are closed, equilibrium is immediately lost.
or backward, it would be impossible to maintain a stable
image on the retinas unless the person had some auto-
Neuronal Connections of the Vestibular Apparatus with the
matic control mechanism to stabilize the direction of the
Central Nervous System
eyes’ gaze. In addition, the eyes would be of little use in
Figure 55-13 shows the connections in the hindbrain of
detecting an image unless they remained “fixed” on
the vestibular nerve. Most of the vestibular nerve fibers
each object long enough to gain a clear image. Fortu-
terminate in the brain stem in the vestibular nuclei,
nately, each time the head is suddenly rotated, signals
which are located approximately at the junction of the
from the semicircular ducts cause the eyes to rotate in
medulla and the pons. Some fibers pass directly to the
a direction equal and opposite to the rotation of the
brain stem reticular nuclei without synapsing and also
head. This results from reflexes transmitted through the
to the cerebellar fastigial, uvular, and flocculonodular
vestibular nuclei and the medial longitudinal fasciculus
lobe nuclei. The fibers that end in the brain stem
to the oculomotor nuclei. These reflexes are described
vestibular nuclei synapse with second-order neurons
in Chapter 51.
that also send fibers into the cerebellum, the vestibu-
lospinal tracts, the medial longitudinal fasciculus, and
Other Factors Concerned
with Equilibrium
Fastigial
Medial longitudinal
Dentate nucleus
nucleus
fasciculus
Neck Proprioceptors. The vestibular apparatus detects the
orientation and movement only of the head. Therefore,
it is essential that the nervous centers also receive
Red
appropriate information about the orientation of the
nucleus
head with respect to the body. This information is trans-
Reticular
mitted from the proprioceptors of the neck and body
substance
directly to the vestibular and reticular nuclei in the
Fastigioreticular
brain stem and indirectly by way of the cerebellum.
tract
Among the most important proprioceptive informa-
tion needed for the maintenance of equilibrium is that
Vestibular nucleus
transmitted by joint receptors of the neck. When the
Vestibular nerve
head is leaned in one direction by bending the neck,
Flocculonodular
impulses from the neck proprioceptors keep the signals
lobe
originating in the vestibular apparatus from giving the
Vestibulospinal tract
person a sense of dysequilibrium. They do this by trans-
Rubrospinal tract
mitting signals that exactly oppose the signals transmit-
Recticulospinal
tract
ted from the vestibular apparatus. However, when the
entire body leans in one direction, the impulses from the
Figure 55-13
vestibular apparatus are not opposed by signals from
the neck proprioceptors; therefore, in this case, the
Connections of vestibular nerves through the vestibular nuclei (the
person does perceive a change in equilibrium status of
large oval white area) with other areas of the central nervous
the entire body.
system.
Chapter 55
Cortical and Brain Stem Control of Motor Function
697
other areas of the brain stem, particularly the reticular
objects with movements of the eyes and head. Also,
nuclei.
placing pressure on the upper anterior parts of their legs
The primary pathway for the equilibrium reflexes
causes them to pull to the sitting position. It is clear that
begins in the vestibular nerves, where the nerves are
many of the stereotyped motor functions of the human
excited by the vestibular apparatus. The pathway then
being are integrated in the brain stem.
passes to the vestibular nuclei and cerebellum. Next,
signals are sent into the reticular nuclei of the brain
stem, as well as down the spinal cord by way of the
References
vestibulospinal and reticulospinal tracts. The signals to
the cord control the interplay between facilitation and
Alitto HJ, Usrey WM: Corticothalamic feedback and sensory
inhibition of the many antigravity muscles, thus auto-
processing. Curr Opin Neurobiol 13:440, 2003.
matically controlling equilibrium.
Angelaki DE, Dickman JD: Gravity or translation: central
The flocculonodular lobes of the cerebellum are espe-
processing of vestibular signals to detect motion or tilt. J
cially concerned with dynamic equilibrium signals from
Vestib Res 13:245, 2003.
the semicircular ducts. In fact, destruction of these lobes
Barmack NH: Central vestibular system: vestibular nuclei
results in almost exactly the same clinical symptoms as
and posterior cerebellum. Brain Res Bull 60:511, 2003.
destruction of the semicircular ducts themselves. That is,
Blake DT, Byl NN, Merzenich MM: Representation of the
severe injury to either the lobes or the ducts causes loss
hand in the cerebral cortex. Behav Brain Res 135:179,
of dynamic equilibrium during rapid changes in direc-
2002.
tion of motion but does not seriously disturb equilib-
Boyle R: Vestibulospinal control of reflex and voluntary
rium under static conditions. It is believed that the uvula
head movement. Ann N Y Acad Sci 942:364, 2001.
of the cerebellum plays a similar important role in static
Cullen KE, Roy JE: Signal processing in the vestibular
equilibrium.
system during active versus passive head movements. J
Signals transmitted upward in the brain stem from
Neurophysiol 91:1919, 2004.
both the vestibular nuclei and the cerebellum by way of
Garwicz M: Spinal reflexes provide motor error signals to
the medial longitudinal fasciculus cause corrective
cerebellar modules—relevance for motor coordination.
movements of the eyes every time the head rotates, so
Brain Res Brain Res Rev 40:152, 2002.
that the eyes remain fixed on a specific visual object.
Johansen-Berg H: Motor physiology: a brain of two halves.
Signals also pass upward (either through this same tract
Curr Biol 13:R802, 2003.
or through reticular tracts) to the cerebral cortex, ter-
Raineteau O, Schwab ME: Plasticity of motor systems after
minating in a primary cortical center for equilibrium
incomplete spinal cord injury. Nat Rev Neurosci 2:263,
located in the parietal lobe deep in the sylvian fissure
2001.
on the opposite side of the fissure from the auditory
Raphael Y, Altschuler RA: Structure and innervation of the
area of the superior temporal gyrus. These signals
cochlea. Brain Res Bull 60:397, 2003.
apprise the psyche of the equilibrium status of the body.
Robles L, Ruggero MA: Mechanics of the mammalian
cochlea. Physiol Rev 81:1305, 2001.
Salenius S, Hari R: Synchronous cortical oscillatory activity
Functions of Brain Stem
during motor action. Curr Opin Neurobiol 13:678, 2003.
Nuclei in Controlling
Sanes JN: Neocortical mechanisms in motor learning. Curr
Subconscious, Stereotyped
Opin Neurobiol 13:225, 2003.
Schieber MH: Motor control: basic units of cortical output?
Movements
Curr Biol 14:R353, 2004.
Rarely, a baby is born without brain structures above
Scott SH: The role of primary motor cortex in goal-directed
the mesencephalic region, a condition called anen-
movements: insights from neurophysiological studies on
cephaly. Some of these babies have been kept alive for
non-human primates. Curr Opin Neurobiol 13:671, 2003.
many months. They are able to perform some stereo-
Umilta MA: Frontal cortex: goal-relatedness and the corti-
typed movements for feeding, such as suckling, extru-
cal motor system. Curr Biol 14:R204, 2004.
sion of unpleasant food from the mouth, and moving the
Yates BJ, Miller AD, Lucot JB: Physiological basis and phar-
hands to the mouth to suck the fingers. In addition, they
macology of motion sickness: an update. Brain Res Bull
can yawn and stretch. They can cry and can follow
47:395, 1998.
C H A P T E R
5
6
Contributions of the Cerebellum
and Basal Ganglia to Overall
Motor Control
Aside from the areas in the cerebral cortex that
stimulate muscle contraction, two other brain struc-
tures are also essential for normal motor function.
They are the cerebellum and the basal ganglia. Yet
neither of these two can control muscle function by
themselves. Instead, they always function in associa-
tion with other systems of motor control.
Basically, the cerebellum plays major roles in the
timing of motor activities and in rapid, smooth progression from one muscle
movement to the next. It also helps to control intensity of muscle contraction
when the muscle load changes, as well as controlling necessary instantaneous
interplay between agonist and antagonist muscle groups.
The basal ganglia help to plan and control complex patterns of muscle move-
ment, controlling relative intensities of the separate movements, directions of
movements, and sequencing of multiple successive and parallel movements for
achieving specific complicated motor goals. This chapter explains the basic
mechanisms of function of the cerebellum and basal ganglia and discusses the
overall brain mechanisms for achieving intricate coordination of total motor
activity.
Cerebellum and Its Motor Functions
The cerebellum, illustrated in Figures 56-1 and 56-2, has long been called a
silent area of the brain, principally because electrical excitation of the cerebel-
lum does not cause any conscious sensation and rarely causes any motor move-
ment. Removal of the cerebellum, however, does cause body movements to
become highly abnormal. The cerebellum is especially vital during rapid mus-
cular activities such as running, typing, playing the piano, and even talking. Loss
of this area of the brain can cause almost total incoordination of these activi-
ties even though its loss causes paralysis of no muscles.
But how is it that the cerebellum can be so important when it has no direct
ability to cause muscle contraction? The answer is that it helps to sequence
the motor activities and also monitors and makes corrective adjustments in the
body’s motor activities while they are being executed so that they will conform to
the motor signals directed by the cerebral motor cortex and other parts of the
brain.
The cerebellum receives continuously updated information about the desired
sequence of muscle contractions from the brain motor control areas; it also
receives continuous sensory information from the peripheral parts of the body,
giving sequential changes in the status of each part of the body—its position,
rate of movement, forces acting on it, and so forth. The cerebellum then com-
pares the actual movements as depicted by the peripheral sensory feedback
information with the movements intended by the motor system. If the two do
not compare favorably, then instantaneous subconscious corrective signals are
transmitted back into the motor system to increase or decrease the levels of
activation of specific muscles.
The cerebellum also aids the cerebral cortex in planning the next sequential
movement a fraction of a second in advance while the current movement is still
698
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
699
Posterior lobe
Anterior lobe
Pons
Medulla
Flocculonodular
lobe
Figure 56-1
Anatomical lobes of the cerebellum as seen from the lateral side.
Figure 56-3
Somatosensory projection areas in the cerebellar cortex.
Hemisphere Vermis
Anterior
lobe
controlling body equilibrium, as discussed in Chapter
55.
Longitudinal Functional Divisions of the Anterior and Posterior
Lobes. From a functional point of view, the anterior and
posterior lobes are organized not by lobes but along the
longitudinal axis, as demonstrated in Figure 56-2, which
Posterior
shows a posterior view of the human cerebellum after
lobe
the lower end of the posterior cerebellum has been
rolled downward from its normally hidden position.
Note down the center of the cerebellum a narrow band
called the vermis, separated from the remainder of the
cerebellum by shallow grooves. In this area, most cere-
Flocculonodular
Lateral zone
bellar control functions for muscle movements of the
Vermis
lobe
of hemisphere
axial body, neck, shoulders, and hips are located.
Intermediate zone
To each side of the vermis is a large, laterally pro-
of hemisphere
truding cerebellar hemisphere, and each of these hemi-
spheres is divided into an intermediate zone and a lateral
Figure 56-2
zone.
The intermediate zone of the hemisphere is con-
Functional parts of the cerebellum as seen from the posteroinfe-
cerned with controlling muscle contractions in the distal
rior view, with the inferiormost portion of the cerebellum rolled
outward to flatten the surface.
portions of the upper and lower limbs, especially the
hands and fingers and feet and toes.
The lateral zone of the hemisphere operates at a
much more remote level because this area joins with the
being executed, thus helping the person to progress
cerebral cortex in the overall planning of sequential
smoothly from one movement to the next. Also, it
motor movements. Without this lateral zone, most dis-
learns by its mistakes—that is, if a movement does not
crete motor activities of the body lose their appropriate
occur exactly as intended, the cerebellar circuit learns
timing and sequencing and therefore become incoordi-
to make a stronger or weaker movement the next time.
nate, as we discuss more fully later.
To do this, changes occur in the excitability of appro-
priate cerebellar neurons, thus bringing subsequent
Topographical Representation of the Body in the Vermis and Inter-
muscle contractions into better correspondence with the
mediate Zones. In the same manner that the cerebral
sensory cortex, motor cortex, basal ganglia, red nuclei,
intended movements.
and reticular formation all have topographical repre-
sentations of the different parts of the body, so also is
Anatomical Functional Areas
this true for the vermis and intermediate zones of the
cerebellum. Figure 56-3 shows two such representa-
of the Cerebellum
tions. Note that the axial portions of the body lie in the
Anatomically, the cerebellum is divided into three lobes
vermis part of the cerebellum, whereas the limbs and
by two deep fissures, as shown in Figures 56-1 and 56-2:
facial regions lie in the intermediate zones. These topo-
(1) the anterior lobe, (2) the posterior lobe, and (3) the
graphical representations receive afferent nerve signals
flocculonodular lobe. The flocculonodular lobe is the
from all the respective parts of the body as well as from
oldest of all portions of the cerebellum; it developed
corresponding topographical motor areas in the cere-
along with (and functions with) the vestibular system in
bral cortex and brain stem. In turn, they send motor
700
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
signals back to the same respective topographical areas
originate in the vestibular apparatus itself and others
of the cerebral motor cortex, as well as to topographi-
from the brain stem vestibular nuclei—almost all of
cal areas of the red nucleus and reticular formation in
these terminate in the flocculonodular lobe and fastigial
the brain stem.
nucleus of the cerebellum; and (3) reticulocerebellar
Note that the large lateral portions of the cerebellar
fibers, which originate in different portions of the brain
hemispheres do not have topographical representations
stem reticular formation and terminate in the midline
of the body. These areas of the cerebellum receive their
cerebellar areas (mainly in the vermis).
input signals almost exclusively from the cerebral
cortex, especially from the premotor areas of the frontal
Afferent Pathways from the Periphery. The cerebellum also
cortex and from the somatosensory and other sensory
receives important sensory signals directly from the
association areas of the parietal cortex. It is believed
peripheral parts of the body mainly through four tracts
that this connectivity with the cerebral cortex allows the
on each side, two of which are located dorsally in the
lateral portions of the cerebellar hemispheres to play
cord and two ventrally. The two most important of these
important roles in planning and coordinating the body’s
tracts are shown in Figure 56-5: the dorsal spinocere-
rapid sequential muscular activities that occur one after
bellar tract and the ventral spinocerebellar tract. The
another within fractions of a second.
dorsal tract enters the cerebellum through the inferior
cerebellar peduncle and terminates in the vermis and
intermediate zones of the cerebellum on the same side
Neuronal Circuit of the Cerebellum
as its origin. The ventral tract enters the cerebellum
through the superior cerebellar peduncle, but it termi-
The human cerebellar cortex is actually a large folded
nates in both sides of the cerebellum.
sheet, about 17 centimeters wide by 120 centimeters
The signals transmitted in the dorsal spinocerebellar
long, with the folds lying crosswise, as shown in Figures
tracts come mainly from the muscle spindles and to a
56-2 and 56-3. Each fold is called a folium. Lying deep
lesser extent from other somatic receptors throughout
beneath the folded mass of cerebellar cortex are deep
the body, such as Golgi tendon organs, large tactile
cerebellar nuclei.
receptors of the skin, and joint receptors. All these
signals apprise the cerebellum of the momentary status
Input Pathways to the Cerebellum
of (1) muscle contraction, (2) degree of tension on the
Afferent Pathways from Other Parts of the Brain. The basic
muscle tendons, (3) positions and rates of movement
input pathways to the cerebellum are shown in Figure
of the parts of the body, and (4) forces acting on the
56-4. An extensive and important afferent pathway is
surfaces of the body.
the corticopontocerebellar pathway, which originates in
Conversely, the ventral spinocerebellar tracts receive
the cerebral motor and premotor cortices and also in the
less information from the peripheral receptors. Instead,
cerebral somatosensory cortex. It passes by way of the
they are excited mainly by motor signals arriving in the
pontile nuclei and pontocerebellar tracts mainly to
anterior horns of the spinal cord from (1) the brain
the lateral divisions of the cerebellar hemispheres on
through the corticospinal and rubrospinal tracts and (2)
the opposite side of the brain from the cerebral areas.
the internal motor pattern generators in the cord itself.
In addition, important afferent tracts originate in
Thus, this ventral fiber pathway tells the cerebellum
each side of the brain stem; they include (1) an exten-
sive olivocerebellar tract, which passes from the inferior
olive to all parts of the cerebellum and is excited in the
olive by fibers from the cerebral motor cortex, basal
Superior cerebellar peduncle
Ventral spinocerebellar tract
ganglia, widespread areas of the reticular formation, and
spinal cord; (2) vestibulocerebellar fibers, some of which
Cerebellum
Anterior Superior cerebellar
lobe
peduncle
Inferior cerebellar peduncle
Ventral
spinocerebellar
Medulla oblongata
tract
Dorsal external arcuate fiber
Cerebropontile
tract
Ventral spinocerebellar tract
Pontocerebellar
tract
Middle cerebellar
Spinal cord
peduncle
Posterior
Vestibulocerebellar tract
lobe
Dorsal spinocerebellar tract
Olivocerebellar and
Flocculonodular
reticulocerebellar tract
lobe
Inferior cerebellar peduncle
Ventral spinocerebellar tract
Dorsal spinocerebellar tract
Clark’s cells
Figure 56-4
Figure 56-5
Principal afferent tracts to the cerebellum.
Spinocerebellar tracts.
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
701
which motor signals have arrived at the anterior horns;
Dentate
Cerebellothalamocortical
this feedback is called the efference copy of the anterior
tract
horn motor drive.
To thalamus
The spinocerebellar pathways can transmit impulses
Red nucleus
at velocities up to 120 m/sec, which is the most rapid
conduction in any pathway in the central nervous
system. This extremely rapid conduction is important
Reticulum of
for instantaneous apprisal of the cerebellum of changes
mesencephalon
in peripheral muscle actions.
In addition to signals from the spinocerebellar tracts,
Superior cerebellar
signals are transmitted into the cerebellum from the
peduncle
body periphery through the spinal dorsal columns to the
Fastigioreticular tract
dorsal column nuclei of the medulla and then relayed
Fastigial nucleus
to the cerebellum. Likewise, signals are transmitted up
the spinal cord through the spinoreticular pathway to
Fastigioreticular tract
the reticular formation of the brain stem and also
Paleocerebellum
through the spino-olivary pathway to the inferior
olivary nucleus. Then signals are relayed from both of
Figure 56-6
these areas to the cerebellum. Thus, the cerebellum con-
tinually collects information about the movements and
Principal efferent tracts from the cerebellum.
positions of all parts of the body even though it is oper-
ating at a subconscious level.
mainly the reciprocal contractions of agonist and
Output Signals from the Cerebellum
antagonist muscles in the peripheral portions of the
Deep Cerebellar Nuclei and the Efferent Pathways. Located
limbs, especially in the hands, fingers, and thumbs.
deep in the cerebellar mass on each side are three deep
3.
A pathway that begins in the cerebellar cortex of
cerebellar nuclei—the dentate, interposed, and fastigial.
the lateral zone of the cerebellar hemisphere and
(The vestibular nuclei in the medulla also function in
then passes to the dentate nucleus, next to the
some respects as if they were deep cerebellar nuclei
ventrolateral and ventroanterior nuclei of the
because of their direct connections with the cortex of
thalamus, and, finally, to the cerebral cortex. This
the flocculonodular lobe.) All the deep cerebellar nuclei
pathway plays an important role in helping
receive signals from two sources: (1) the cerebellar
coordinate sequential motor activities initiated by
cortex and (2) the deep sensory afferent tracts to the
the cerebral cortex.
cerebellum.
Each time an input signal arrives in the cerebellum,
Functional Unit of the Cerebellar Cortex—
it divides and goes in two directions: (1) directly to one
The Purkinje Cell and the Deep Nuclear Cell
of the cerebellar deep nuclei and (2) to a corresponding
The cerebellum has about 30 million nearly identical
area of the cerebellar cortex overlying the deep nucleus.
functional units, one of which is shown to the left in
Then, a fraction of a second later, the cerebellar cortex
Figure 56-7. This functional unit centers on a single,
relays an inhibitory output signal to the deep nucleus.
very large Purkinje cell (30 million of which are in
Thus, all input signals that enter the cerebellum even-
the cerebellar cortex) and on a corresponding deep
tually end in the deep nuclei in the form of initial exci-
tatory signals followed a fraction of a second later by
nuclear cell.
inhibitory signals. From the deep nuclei, output signals
To the top and right in Figure 56-7, the three major
leave the cerebellum and are distributed to other parts
layers of the cerebellar cortex are shown: the molecu-
of the brain.
lar layer, Purkinje cell layer, and granule cell layer.
The general plan of the major efferent pathways
Beneath these cortical layers, in the center of the cere-
leading out of the cerebellum is shown in Figure 56-6
bellar mass, are the deep cerebellar nuclei that send
and consists of the following:
output signals to other parts of the nervous system.
1. A pathway that originates in the midline structures
of the cerebellum (the vermis) and then passes
Neuronal Circuit of the Functional Unit. Also shown in the
through the fastigial nuclei into the medullary and
left half of Figure 56-7 is the neuronal circuit of the
pontile regions of the brain stem. This circuit
functions in close association with the equilibrium
functional unit, which is repeated with little variation
apparatus and brain stem vestibular nuclei to
30 million times in the cerebellum. The output from
control equilibrium, and also in association with the
the functional unit is from a deep nuclear cell. This cell
reticular formation of the brain stem to control the
is continually under both excitatory and inhibitory
postural attitudes of the body. It was discussed in
influences. The excitatory influences arise from direct
detail in Chapter 55 in relation to equilibrium.
connections with afferent fibers that enter the cere-
2. A pathway that originates in (1) the intermediate
bellum from the brain or the periphery. The inhibitory
zone of the cerebellar hemisphere and then passes
influence arises entirely from the Purkinje cell in the
through (2) the interposed nucleus to (3) the
cortex of the cerebellum.
ventrolateral and ventroanterior nuclei of the
The afferent inputs to the cerebellum are mainly of
thalamus and then to (4) the cerebral cortex, to (5)
several midline structures of the thalamus and then
two types, one called the climbing fiber type and the
to (6) the basal ganglia and (7) the red nucleus and
other called the mossy fiber type.
reticular formation of the upper portion of the
The climbing fibers all originate from the inferior
brain stem. This complex circuit helps to coordinate
olives of the medulla. There is one climbing fiber for
702
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Purkinje Cells and Deep Nuclear Cells Fire Continuously Under
Molecular
layer
Normal Resting Conditions. One characteristic of both
Purkinje
Purkinje cells and deep nuclear cells is that normally
Purkinje
cell
cell layer
both of them fire continuously; the Purkinje cell fires
at about 50 to 100 action potentials per second, and
Climbing
Granule
fiber
cell layer
the deep nuclear cells at much higher rates. Further-
Inhibition
Deep
Granule
more, the output activity of both these cells can be
nuclear
cells
Deep
modulated upward or downward.
cell
nuclei
Excitation
Mossy
fiber
Balance Between Excitation and Inhibition at the Deep Cere-
Input
bellar Nuclei. Referring again to the circuit of Figure
Input
(all other afferents)
56-7, one should note that direct stimulation of the
(inferior olive)
deep nuclear cells by both the climbing and the mossy
Output
fibers excites them. By contrast, signals arriving from
the Purkinje cells inhibit them. Normally, the balance
Figure 56-7
between these two effects is slightly in favor of exci-
tation, so that, under quiet conditions, output from the
The left side of this figure shows the basic neuronal circuit of the
deep nuclear cell remains relatively constant at a mod-
cerebellum, with excitatory neurons shown in red and the Purkinje
erate level of continuous stimulation.
cell (an inhibitory neuron) shown in black. To the right is shown
the physical relationship of the deep cerebellar nuclei to the cere-
In execution of a rapid motor movement, the
bellar cortex with its three layers.
initiating signal from the cerebral motor cortex or
brain stem at first greatly increases deep nuclear
cell excitation. Then, another few milliseconds later,
feedback inhibitory signals from the Purkinje cell
about 5 to 10 Purkinje cells. After sending branches to
circuit arrive. In this way, there is first a rapid excita-
several deep nuclear cells, the climbing fiber continues
tory signal sent by the deep nuclear cells into the
all the way to the outer layers of the cerebellar cortex,
motor output pathway to enhance the motor move-
where it makes about 300 synapses with the soma
ment, but this is followed within another small fraction
and dendrites of each Purkinje cell. This climbing
of a second by an inhibitory signal. This inhibitory
fiber is distinguished by the fact that a single impulse
signal resembles a
“delay-line” negative feedback
in it will always cause a single, prolonged (up to 1
signal of the type that is effective in providing
second), peculiar type of action potential in each
damping. That is, when the motor system is excited,
Purkinje cell with which it connects, beginning with
a negative feedback signal occurs after a short delay
a strong spike and followed by a trail of weakening
to stop the muscle movement from overshooting its
secondary spikes. This action potential is called the
mark. Otherwise, oscillation of the movement would
complex spike.
occur.
The mossy fibers are all the other fibers that enter
the cerebellum from multiple sources: from the higher
Other Inhibitory Cells in the Cerebellum. In addition to the
brain, brain stem, and spinal cord. These fibers also
deep nuclear cells, granule cells, and Purkinje cells, two
send collaterals to excite the deep nuclear cells. Then
other types of neurons are located in the cerebellum:
they proceed to the granule cell layer of the cortex,
basket cells and stellate cells. These are inhibitory cells
where they too synapse with hundreds to thousands of
with short axons. Both the basket cells and the stellate
granule cells. In turn, the granule cells send very, very
cells are located in the molecular layer of the cerebel-
small axons, less than 1 micrometer in diameter, up to
lar cortex, lying among and stimulated by the small
the molecular layer on the outer surface of the cere-
parallel fibers. These cells in turn send their axons at
bellar cortex. Here the axons divide into two branches
right angles across the parallel fibers and cause lateral
that extend 1 to 2 millimeters in each direction paral-
inhibition of adjacent Purkinje cells, thus sharpening
lel to the folia. There are many millions of these par-
the signal in the same manner that lateral inhibition
allel nerve fibers because there are some 500 to 1000
sharpens contrast of signals in many other neuronal
granule cells for every 1 Purkinje cell. It is into this
circuits of the nervous system.
molecular layer that the dendrites of the Purkinje cells
project and 80,000 to 200,000 of the parallel fibers
Turn-On/Turn-Off and Turn-Off/Turn-On Output
synapse with each Purkinje cell.
Signals from the Cerebellum
The mossy fiber input to the Purkinje cell is quite
The typical function of the cerebellum is to help
different from the climbing fiber input because their
provide rapid turn-on signals for the agonist muscles
synaptic connections are weak, so that large numbers
and simultaneous reciprocal turn-off signals for the
of mossy fibers must be stimulated simultaneously to
antagonist muscles at the onset of a movement. Then
excite the Purkinje cell. Furthermore, activation
on approaching termination of the movement, the
usually takes the form of a much weaker short-
cerebellum is mainly responsible for timing and exe-
duration Purkinje cell action potential called a simple
cuting the turn-off signals to the agonists and turn-on
spike, rather than the prolonged complex action
signals to the antagonists. Although the exact details
potential caused by climbing fiber input.
are not fully known, one can speculate from the basic
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
703
cerebellar circuit of Figure 56-7 how this might work,
are still to be determined; they, too, could play roles in
as follows.
the initial inhibition of the antagonist muscles at onset
Let us suppose that the turn-on/turn-off pattern of
of a movement and subsequent excitation at the end
agonist/antagonist contraction at the onset of move-
of a movement.
ment begins with signals from the cerebral cortex.
All these mechanisms are still partly speculation.
These signals pass through noncerebellar brain stem
They are presented here especially to illustrate ways
and cord pathways directly to the agonist muscle to
by which the cerebellum could cause exaggerated turn-
begin the initial contraction.
on and turn-off signals, controlling the agonist and
At the same time, parallel signals are sent by way of
antagonist muscles, and controlling the timing as well.
the pontile mossy fibers into the cerebellum. One
branch of each mossy fiber goes directly to deep
The Purkinje Cells “Learn” to Correct Motor
nuclear cells in the dentate or other deep cerebellar
Errors—Role of the Climbing Fibers
nuclei; this instantly sends an excitatory signal back
The degree to which the cerebellum supports onset
into the cerebral corticospinal motor system, either by
and offset of muscle contractions, as well as timing of
way of return signals through the thalamus to the cere-
contractions, must be learned by the cerebellum. Typ-
bral cortex or by way of neuronal circuitry in the brain
ically, when a person first performs a new motor act,
stem, to support the muscle contraction signal that had
the degree of motor enhancement by the cerebellum
already been begun by the cerebral cortex. As a con-
at the onset of contraction, the degree or inhibition at
sequence, the turn-on signal, after a few milliseconds,
the end of contraction, and the timing of these are
becomes even more powerful than it was at the start
almost always incorrect for precise performance of the
because it becomes the sum of both the cortical
movement. But after the act has been performed many
and the cerebellar signals. This is the normal effect
times, the individual events become progressively
when the cerebellum is intact, but in the absence of the
more precise, sometimes requiring only a few move-
cerebellum, the secondary extra supportive signal is
ments before the desired result is achieved, but at
missing. This cerebellar support makes the turn-on
other times requiring hundreds of movements.
muscle contraction much stronger than it would be if
How do these adjustments come about? The exact
the cerebellum did not exist.
answer is not known, although it is known that sensi-
Now, what causes the turn-off signal for the agonist
tivity levels of cerebellar circuits themselves progres-
muscles at the termination of the movement? Remem-
sively adapt during the training process. Especially,
ber that all mossy fibers have a second branch that
the sensitivity of the Purkinje cells to respond to the
transmits signals by way of the granule cells to the
granule cell excitation becomes altered. Furthermore,
cerebellar cortex and eventually, by way of “parallel”
this sensitivity change is brought about by signals from
fibers, to the Purkinje cells. The Purkinje cells in turn
the climbing fibers entering the cerebellum from the
inhibit the deep nuclear cells. This pathway passes
inferior olivary complex.
through some of the smallest, slowest-conducting
Under resting conditions, the climbing fibers fire
nerve fibers in the nervous system: that is, the parallel
about once per second. But each time they do fire, they
fibers of the cerebellar cortical molecular layer, which
cause extreme depolarization of the entire dendritic
have diameters of only a fraction of a millimeter. Also,
tree of the Purkinje cell, lasting for up to a second.
the signals from these fibers are weak so that they
During this time, the Purkinje cell fires with one initial
require a finite period of time to build up enough exci-
strong output spike followed by a series of diminish-
tation in the dendrites of the Purkinje cell to excite it.
ing spikes. When a person performs a new movement
But once the Purkinje cell is excited, it in turn sends a
for the first time, feedback signals from the muscle and
strong inhibitory signal to the same deep nuclear cell
joint proprioceptors will usually denote to the cere-
that had originally turned on the movement. There-
bellum how much the actual movement fails to match
fore, this helps to turn off the movement after a short
the intended movement. And the climbing fiber signals
time.
in some way alter long-term sensitivity of the Purkinje
Thus, one can see how the complete cerebellar
cells. Over a period of time, this change in sensitivity,
circuit could cause a rapid turn-on agonist muscle con-
along with other possible “learning” functions of the
traction at the beginning of a movement and yet cause
cerebellum, is believed to make the timing and other
also a precisely timed turn-off of the same agonist con-
aspects of cerebellar control of movements approach
traction after a given time period.
perfection. When this has been achieved, the climbing
Now let us speculate on the circuit for the antago-
fibers no longer need to send “error” signals to the
nist muscles. Most important, remember that through-
cerebellum to cause further change.
out the spinal cord there are reciprocal agonist/
antagonist circuits for virtually every movement that
the cord can initiate. Therefore, these circuits are part
Function of the Cerebellum in Overall
of the basis for antagonist turn-off at the onset of
Motor Control
movement and then turn-on at termination of move-
ment, mirroring whatever occurs in the agonist
The nervous system uses the cerebellum to coordinate
muscles. But we must remember, too, that the cere-
motor control functions at three levels, as follows:
bellum contains several other types of inhibitory cells
1. The vestibulocerebellum. This consists principally
besides Purkinje cells. The functions of some of these
of the small flocculonodular cerebellar lobes (that
704
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
lie under the posterior cerebellum) and adjacent
brain at the same time that the movements actually
portions of the vermis. It provides neural circuits
occur. How, then, is it possible for the brain to know
for most of the body’s equilibrium movements.
when to stop a movement and to perform the next
2. The spinocerebellum. This consists of most of the
sequential act, especially when the movements are
vermis of the posterior and anterior cerebellum
performed rapidly? The answer is that the signals from
plus the adjacent intermediate zones on both
the periphery tell the brain how rapidly and in which
sides of the vermis. It provides the circuitry for
directions the body parts are moving. It is then the
coordinating mainly movements of the distal
function of the vestibulocerebellum to calculate in
portions of the limbs, especially the hands and
advance from these rates and directions where the
fingers.
different parts will be during the next few milliseconds.
3. The cerebrocerebellum. This consists of the large
The results of these calculations are the key to the
lateral zones of the cerebellar hemispheres, lateral
brain’s progression to the next sequential movement.
to the intermediate zones. It receives virtually all
Thus, during control of equilibrium, it is presumed
its input from the cerebral motor cortex and
that information from both the body periphery and the
adjacent premotor and somatosensory cortices of
vestibular apparatus is used in a typical feedback
the cerebrum. It transmits its output information
control circuit to provide anticipatory correction of
in the upward direction back to the brain,
postural motor signals necessary for maintaining equi-
functioning in a feedback manner with the
librium even during extremely rapid motion, including
cerebral cortical sensorimotor system to plan
rapidly changing directions of motion.
sequential voluntary body and limb movements,
planning these as much as tenths of a second in
Spinocerebellum—Feedback Control of Distal
advance of the actual movements. This is called
Limb Movements by Way of the Intermediate
development of “motor imagery” of movements
Cerebellar Cortex and the Interposed Nucleus
to be performed.
As shown in Figure 56-8, the intermediate zone of
each cerebellar hemisphere receives two types of
Vestibulocerebellum—Its Function in
information when a movement is performed: (1) infor-
Association with the Brain Stem and
mation from the cerebral motor cortex and from
Spinal Cord to Control Equilibrium and
the midbrain red nucleus, telling the cerebellum the
Postural Movements
intended sequential plan of movement for the next few
The vestibulocerebellum originated phylogenetically
fractions of a second, and (2) feedback information
at about the same time that the vestibular apparatus
in the inner ear developed. Furthermore, as discussed
in Chapter 55, loss of the flocculonodular lobes and
adjacent portions of the vermis of the cerebellum,
Motor cortex
which constitute the vestibulocerebellum, causes
extreme disturbance of equilibrium and postural
movements.
We still must ask the question, what role does the
vestibulocerebellum play in equilibrium that cannot
be provided by other neuronal machinery of the brain
Red nucleus
Thalamus
stem? A clue is the fact that in people with vestibulo-
cerebellar dysfunction, equilibrium is far more dis-
turbed during performance of rapid motions than
during stasis, especially so when these movements
Intermediate
involve changes in direction of movement and stimu-
zone of
Mesencephalon,
late the semicircular ducts. This suggests that the
cerebellum
pons, and medulla
vestibulocerebellum is especially important in con-
trolling balance between agonist and antagonist
muscle contractions of the spine, hips, and shoulders
during rapid changes in body positions as required by
Corticospinal tract
the vestibular apparatus.
Spinocerebellar
One of the major problems in controlling balance is
tract
Reticulospinal
the amount of time required to transmit position
and rubrospinal
signals and velocity of movement signals from the dif-
tracts
ferent parts of the body to the brain. Even when the
most rapidly conducting sensory pathways are used, up
Muscles
to 120 m/sec in the spinocerebellar afferent tracts, the
delay for transmission from the feet to the brain is still
15 to 20 milliseconds. The feet of a person running
Figure 56-8
rapidly can move as much as 10 inches during that
time. Therefore, it is never possible for return signals
Cerebral and cerebellar control of voluntary movements, involving
from the peripheral parts of the body to reach the
especially the intermediate zone of the cerebellum.
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
705
from the peripheral parts of the body, especially from
pendular elements that have inertia must have
the distal proprioceptors of the limbs, telling the cere-
damping circuits built into the mechanisms. For motor
bellum what actual movements result.
control by the nervous system, the cerebellum pro-
After the intermediate zone of the cerebellum has
vides most of this damping function.
compared the intended movements with the actual
movements, the deep nuclear cells of the interposed
Cerebellar Control of Ballistic Movements. Most rapid
nucleus send corrective output signals (1) back to the
movements of the body, such as the movements of the
cerebral motor cortex through relay nuclei in the thal-
fingers in typing, occur so rapidly that it is not possi-
amus and (2) to the magnocellular portion (the lower
ble to receive feedback information either from the
portion) of the red nucleus that gives rise to the
periphery to the cerebellum or from the cerebellum
rubrospinal tract. The rubrospinal tract in turn joins
back to the motor cortex before the movements are
the corticospinal tract in innervating the lateral most
over. These movements are called ballistic movements,
motor neurons in the anterior horns of the spinal cord
meaning that the entire movement is preplanned and
gray matter, the neurons that control the distal parts
set into motion to go a specific distance and then to
of the limbs, particularly the hands and fingers.
stop. Another important example is the saccadic move-
This part of the cerebellar motor control system pro-
ments of the eyes, in which the eyes jump from one
vides smooth, coordinate movements of the agonist
position to the next when reading or when looking at
and antagonist muscles of the distal limbs for per-
successive points along a road as a person is moving
forming acute purposeful patterned movements. The
in a car.
cerebellum seems to compare the “intentions” of the
Much can be understood about the function of the
higher levels of the motor control system, as transmit-
cerebellum by studying the changes that occur in these
ted to the intermediate cerebellar zone through the
ballistic movements when the cerebellum is removed.
corticopontocerebellar tract, with the “performance”
Three major changes occur: (1) The movements are
by the respective parts of the body, as transmitted
slow to develop and do not have the extra onset surge
back to the cerebellum from the periphery. In fact,
that the cerebellum usually provides, (2) the force
the ventral spinocerebellar tract even transmits back
developed is weak, and (3) the movements are slow to
to the cerebellum an “efference” copy of the actual
turn off, usually allowing the movement to go well
motor control signals that reach the anterior motor
beyond the intended mark. Therefore, in the absence
neurons, and this, too, is integrated with the signals
of the cerebellar circuit, the motor cortex has to think
arriving from the muscle spindles and other proprio-
extra hard to turn ballistic movements on and again
ceptor sensory organs, transmitted principally in the
has to think hard and take extra time to turn the
dorsal spinocerebellar tract. We learned earlier that
movement off. Thus, the automatism of ballistic move-
similar comparator signals also go to the inferior
ments is lost.
olivary complex; if the signals do not compare favor-
If one considers once again the circuitry of the cere-
ably, the olivary-Purkinje cell system along with pos-
bellum as described earlier, one sees that it is beauti-
sibly other cerebellar learning mechanisms eventually
fully organized to perform this biphasic, first excitatory
corrects the motions until they perform the desired
and then delayed inhibitory function that is required
function.
for preplanned rapid ballistic movements. One also
sees that the built-in timing circuits of the cerebellar
Function of the Cerebellum to Prevent Overshoot of Movements
cortex are fundamental to this particular ability of the
and to “Damp” Movements. Almost all movements of the
cerebellum.
body are “pendular.” For instance, when an arm is
moved, momentum develops, and the momentum
Cerebrocerebellum—Function of the
must be overcome before the movement can be
Large Lateral Zone of the Cerebellar
stopped. Because of momentum, all pendular move-
Hemisphere to Plan, Sequence, and Time
ments have a tendency to overshoot. If overshooting
Complex Movements
does occur in a person whose cerebellum has been
In human beings, the lateral zones of the two cerebel-
destroyed, the conscious centers of the cerebrum even-
lar hemispheres are highly developed and greatly
tually recognize this and initiate a movement in the
enlarged. This goes along with human abilities to plan
reverse direction attempting to bring the arm to its
and perform intricate sequential patterns of move-
intended position. But the arm, by virtue of its momen-
ment, especially with the hands and fingers, and to
tum, overshoots once more in the opposite direction,
speak. Yet the large lateral zones of the cerebellar
and appropriate corrective signals must again be insti-
hemispheres have no direct input of information from
tuted. Thus, the arm oscillates back and forth past its
the peripheral parts of the body. Also, almost all com-
intended point for several cycles before it finally fixes
munication between these lateral cerebellar areas and
on its mark. This effect is called an action tremor, or
the cerebral cortex is not with the primary cerebral
intention tremor.
motor cortex itself but instead with the premotor area
But, if the cerebellum is intact, appropriate learned,
and primary and association somatosensory areas.
subconscious signals stop the movement precisely at
Even so, destruction of the lateral zones of the cere-
the intended point, thereby preventing the overshoot
bellar hemispheres along with their deep nuclei, the
as well as the tremor. This is the basic characteristic
dentate nuclei, can lead to extreme incoordination of
of a damping system. All control systems regulating
complex purposeful movements of the hands, fingers,
706
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
and feet and of the speech apparatus. This has been
body. For instance, the rates of progression of both
difficult to understand because of lack of direct com-
auditory and visual phenomena can be predicted by
munication between this part of the cerebellum and
the brain, but both of these require cerebellar partici-
the primary motor cortex. However, experimental
pation. As an example, a person can predict from
studies suggest that these portions of the cerebellum
the changing visual scene how rapidly he or she is
are concerned with two other important but indirect
approaching an object. A striking experiment that
aspects of motor control: (1) the planning of sequen-
demonstrates the importance of the cerebellum in this
tial movements and (2) the “timing” of the sequential
ability is the effects of removing the large lateral por-
movements.
tions of the cerebellum in monkeys. Such a monkey
occasionally charges the wall of a corridor and literally
Planning of Sequential Movements. The planning of
bashes its brains because it is unable to predict when
sequential movements requires that the lateral zones
it will reach the wall.
of the hemispheres communicate with both the pre-
We are only now beginning to learn about these
motor and the sensory portions of the cerebral cortex,
extramotor predictive functions of the cerebellum. It
and it requires two-way communication between these
is quite possible that the cerebellum provides a “time-
cerebral cortex areas with corresponding areas of the
base,” perhaps using time-delay circuits, against which
basal ganglia. It seems that the “plan” of sequential
signals from other parts of the central nervous system
movements actually begins in the sensory and premo-
can be compared; it is often stated that the cerebellum
tor areas of the cerebral cortex, and from there the
is particularly helpful in interpreting rapidly changing
plan is transmitted to the lateral zones of the cerebel-
spatiotemporal relations in sensory information.
lar hemispheres. Then, amid much two-way traffic
between cerebellum and cerebral cortex, appropriate
motor signals provide transition from one sequence of
Clinical Abnormalities of
movements to the next.
the Cerebellum
An exceedingly interesting observation that sup-
An important feature of clinical cerebellar abnormali-
ports this view is that many neurons in the cerebellar
ties is that destruction of small portions of the lateral
dentate nuclei display the activity pattern for the
cerebellar cortex seldom causes detectable abnormali-
sequential movement that is yet to come while the
ties in motor function. In fact, several months after as
present movement is still occurring. Thus, the lateral
much as one half of the lateral cerebellar cortex on one
cerebellar zones appear to be involved not with what
side of the brain has been removed, if the deep cere-
movement is happening at a given moment but with
bellar nuclei are not removed along with the cortex, the
what will be happening during the next sequential
motor functions of the animal appear to be almost
movement a fraction of a second or perhaps even
normal as long as the animal performs all movements
seconds later.
slowly. Thus, the remaining portions of the motor
control system are capable of compensating tremen-
To summarize, one of the most important features
dously for loss of parts of the cerebellum.
of normal motor function is one’s ability to progress
Therefore, to cause serious and continuing dysfunc-
smoothly from one movement to the next in orderly
tion of the cerebellum, the cerebellar lesion usually
succession. In the absence of the large lateral zones of
must involve one or more of the deep cerebellar
the cerebellar hemispheres, this capability is seriously
nuclei—the dentate, interposed, or fastigial nuclei.
disturbed for rapid movements.
Dysmetria and Ataxia. Two of the most important symp-
Timing Function. Another important function of the
toms of cerebellar disease are dysmetria and ataxia. As
lateral zones of the cerebellar hemispheres is to
pointed out previously, in the absence of the cerebel-
provide appropriate timing for each succeeding move-
lum, the subconscious motor control system cannot
predict how far movements will go. Therefore, the
ment. In the absence of these cerebellar zones, one
movements ordinarily overshoot their intended mark;
loses the subconscious ability to predict ahead of time
then the conscious portion of the brain overcompen-
how far the different parts of the body will move in a
sates in the opposite direction for the succeeding com-
given time. Without this timing capability, the person
pensatory movement. This effect is called dysmetria, and
becomes unable to determine when the next sequen-
it results in uncoordinated movements that are called
tial movement needs to begin. As a result, the suc-
ataxia. Dysmetria and ataxia can also result from lesions
ceeding movement may begin too early or, more likely,
in the spinocerebellar tracts because feedback informa-
too late. Therefore, lesions in the lateral zones of the
tion from the moving parts of the body to the cerebel-
cerebellum cause complex movements (such as those
lum is essential for cerebellar timing of movement
required for writing, running, or even talking) to
termination.
become incoordinate and lacking ability to progress in
Past Pointing. Past pointing means that in the absence
orderly sequence from one movement to the next.
of the cerebellum, a person ordinarily moves the hand
Such cerebellar lesions are said to cause failure of
or some other moving part of the body considerably
smooth progression of movements.
beyond the point of intention. This results from the
fact that normally the cerebellum initiates most of the
Extramotor Predictive Functions of the Cerebrocerebellum.
motor signal that turns off a movement after it is
The cerebrocerebellum (the large lateral lobes) also
begun; if the cerebellum is not available to do this, the
helps to “time” events other than movements of the
movement ordinarily goes beyond the intended mark.
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
707
Longitudinal fissure
Caudate nucleus
Tail of caudate
POSTERIOR
Figure 56-9
Thalamus
ANTERIOR
Anatomical relations of the basal
ganglia to the cerebral cortex and
thalamus, shown in three-dimen-
sional view. (Redrawn from Guyton
LATERAL
AC: Basic Neuroscience: Anatomy
Putamen and
Fibers to and from
and Physiology. Philadelphia: WB
globus pallidus
spinal cord in
Saunders Co, 1992.)
internal capsule
Therefore, past pointing is actually a manifestation of
Cerebellar Nystagmus. Cerebellar nystagmus is tremor of
dysmetria.
the eyeballs that occurs usually when one attempts to
fixate the eyes on a scene to one side of the head. This
Failure of Progression
off-center type of fixation results in rapid, tremulous
Dysdiadochokinesia. When the motor control system
movements of the eyes rather than steady fixation, and
fails to predict where the different parts of the body will
it is another manifestation of failure of damping by the
be at a given time, it “loses” perception of the parts
cerebellum. It occurs especially when the flocculonodu-
during rapid motor movements. As a result, the suc-
lar lobes of the cerebellum are damaged; in this instance
ceeding movement may begin much too early or much
it is also associated with loss of equilibrium because of
too late, so that no orderly “progression of movement”
dysfunction of the pathways through the flocculonodu-
can occur. One can demonstrate this readily by having
lar cerebellum from the semicircular ducts.
a patient with cerebellar damage turn one hand upward
and downward at a rapid rate. The patient rapidly
Hypotonia. Loss of the deep cerebellar nuclei, particu-
“loses” all perception of the instantaneous position of
larly of the dentate and interposed nuclei, causes
the hand during any portion of the movement. As a
decreased tone of the peripheral body musculature on
result, a series of stalled attempted but jumbled move-
the side of the cerebellar lesion. The hypotonia results
ments occurs instead of the normal coordinate upward
from loss of cerebellar facilitation of the motor cortex
and downward motions. This is called dysdiadochokine-
and brain stem motor nuclei by tonic signals from the
sia.
deep cerebellar nuclei.
Dysarthria. Another example in which failure of pro-
gression occurs is in talking because the formation of
Basal Ganglia—Their Motor
words depends on rapid and orderly succession of indi-
Functions
vidual muscle movements in the larynx, mouth, and res-
piratory system. Lack of coordination among these and
The basal ganglia, like the cerebellum, constitute
inability to adjust in advance either the intensity of
sound or duration of each successive sound causes
another accessory motor system that functions usually
jumbled vocalization, with some syllables loud, some
not by itself but in close association with the cerebral
weak, some held for long intervals, some held for short
cortex and corticospinal motor control system. In fact,
intervals, and resultant speech that is often unintelligi-
the basal ganglia receive most of their input signals
ble. This is called dysarthria.
from the cerebral cortex itself and also return almost
all their output signals back to the cortex.
Intention Tremor. When a person who has lost the cere-
Figure 56-9 shows the anatomical relations of the
bellum performs a voluntary act, the movements tend
basal ganglia to other structures of the brain. On each
to oscillate, especially when they approach the intended
side of the brain, these ganglia consist of the caudate
mark, first overshooting the mark and then vibrating
nucleus, putamen, globus pallidus, substantia nigra,
back and forth several times before settling on the
mark. This reaction is called an intention tremor or an
and subthalamic nucleus. They are located mainly
action tremor, and it results from cerebellar overshoot-
lateral to and surrounding the thalamus, occupying a
ing and failure of the cerebellar system to “damp” the
large portion of the interior regions of both cerebral
motor movements.
hemispheres. Note also that almost all motor and
708
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
sensory nerve fibers connecting the cerebral cortex
Function of the Basal Ganglia in
and spinal cord pass through the space that lies
Executing Patterns of Motor Activity—
between the major masses of the basal ganglia, the
The Putamen Circuit
caudate nucleus and the putamen. This space is called
the internal capsule of the brain. It is important for our
One of the principal roles of the basal ganglia in motor
current discussion because of the intimate association
control is to function in association with the corti-
between the basal ganglia and the corticospinal system
cospinal system to control complex patterns of motor
for motor control.
activity. An example is the writing of letters of the
alphabet. When there is serious damage to the basal
Neuronal Circuitry of the Basal Ganglia. The anatomical
ganglia, the cortical system of motor control can no
connections between the basal ganglia and the other
longer provide these patterns. Instead, one’s writing
brain elements that provide motor control are
becomes crude, as if one were learning for the first
complex, as shown in Figure 56-10. To the left is shown
time how to write.
the motor cortex, thalamus, and associated brain stem
Other patterns that require the basal ganglia are
and cerebellar circuitry. To the right is the major cir-
cutting paper with scissors, hammering nails, shooting
cuitry of the basal ganglia system, showing the tremen-
a basketball through a hoop, passing a football, throw-
dous interconnections among the basal ganglia
ing a baseball, the movements of shoveling dirt, most
themselves plus extensive input and output pathways
aspects of vocalization, controlled movements of the
between the other motor regions of the brain and the
eyes, and virtually any other of our skilled movements,
basal ganglia.
most of them performed subconsciously.
In the next few sections we will concentrate espe-
cially on two major circuits, the putamen circuit and the
Neural Pathways of the Putamen Circuit. Figure
56-11
caudate circuit.
shows the principal pathways through the basal
ganglia for executing learned patterns of movement.
They begin mainly in the premotor and supplementary
areas of the motor cortex and in the somatosensory
areas of the sensory cortex. Next they pass to the
Premotor and
putamen (mainly bypassing the caudate nucleus), then
supplemental motor
to the internal portion of the globus pallidus, next to
association areas
the ventroanterior and ventrolateral relay nuclei of
the thalamus, and finally return to the cerebral primary
Motor cortex
motor cortex and to portions of the premotor and sup-
plementary cerebral areas closely associated with the
Caudate
primary motor cortex. Thus, the putamen circuit has its
nucleus
Thalamus
Putamen
Premotor and
supplemental
Primary motor
Prefrontal
Somatosensory
Subthalamus
Globus
pallidus
Substantia nigra
Red nucleus
Ventroanterior and
Cerebellum
Inferior olive
ventrolateral
nuclei of thalamus
Caudate
Putamen
Reticular formation
Subthalamus
Globus
Muscles
pallidus
Substantia nigra
internal/external
Figure 56-10
Figure 56-11
Relation of the basal ganglial circuitry to the corticospinal-
Putamen circuit through the basal ganglia for subconscious exe-
cerebellar system for movement control.
cution of learned patterns of movement.
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
709
inputs mainly from those parts of the brain adjacent to
Premotor and
the primary motor cortex but not much from the
supplemental
Primary motor
Prefrontal
primary motor cortex itself. Then its outputs do go
Somatosensory
mainly back to the primary motor cortex or closely
associated premotor and supplementary cortex. Func-
tioning in close association with this primary putamen
circuit are ancillary circuits that pass from the
putamen through the external globus pallidus, the sub-
thalamus, and the substantia nigra—finally returning
to the motor cortex by way of the thalamus.
Abnormal Function in the Putamen Circuit: Athetosis, Hemibal-
Ventroanterior and
lismus, and Chorea. How does the putamen circuit func-
ventrolateral
tion to help execute patterns of movement? The
nuclei of thalamus
Caudate
answer is poorly known. However, when a portion of
the circuit is damaged or blocked, certain patterns of
Putamen
movement become severely abnormal. For instance,
Subthalamus
lesions in the globus pallidus frequently lead to spon-
Globus
taneous and often continuous writhing movements of
pallidus
a hand, an arm, the neck, or the face—movements
Substantia nigra
internal/external
called athetosis.
A lesion in the subthalamus often leads to sudden
Figure 56-12
flailing movements of an entire limb, a condition called
hemiballismus.
Caudate circuit through the basal ganglia for cognitive planning
Multiple small lesions in the putamen lead to flick-
of sequential and parallel motor patterns to achieve specific con-
ing movements in the hands, face, and other parts of
scious goals.
the body, called chorea.
Lesions of the substantia nigra lead to the common
and extremely severe disease of rigidity, akinesia, and
tremors known as Parkinson’s disease, which we
ventroanterior and ventrolateral thalamus, and finally
discuss in more detail later.
back to the prefrontal, premotor, and supplementary
motor areas of the cerebral cortex, but with almost
none of the returning signals passing directly to the
Role of the Basal Ganglia for
primary motor cortex. Instead, the returning signals go
Cognitive Control of Sequences of
to those accessory motor regions in the premotor and
supplementary motor areas that are concerned with
Motor Patterns—The Caudate Circuit
putting together sequential patterns of movement
lasting 5 or more seconds instead of exciting individ-
The term cognition means the thinking processes of
ual muscle movements.
the brain, using both sensory input to the brain plus
A good example of this would be a person seeing a
information already stored in memory. Most of our
lion approach and then responding instantaneously
motor actions occur as a consequence of thoughts gen-
and automatically by (1) turning away from the lion,
erated in the mind, a process called cognitive control
(2) beginning to run, and (3) even attempting to climb
of motor activity. The caudate nucleus plays a major
a tree. Without the cognitive functions, the person
role in this cognitive control of motor activity.
might not have the instinctive knowledge, without
The neural connections between the caudate
thinking for too long a time, to respond quickly and
nucleus and the corticospinal motor control system,
appropriately. Thus, cognitive control of motor activity
shown in Figure 56-12, are somewhat different from
determines subconsciously, and within seconds, which
those of the putamen circuit. Part of the reason for this
patterns of movement will be used together to achieve
is that the caudate nucleus, as shown in Figure 56-9,
a complex goal that might itself last for many seconds.
extends into all lobes of the cerebrum, beginning ante-
riorly in the frontal lobes, then passing posteriorly
through the parietal and occipital lobes, and finally
Function of the Basal Ganglia to
curving forward again like the letter “C” into the tem-
poral lobes. Furthermore, the caudate nucleus receives
Change the Timing and to Scale the
large amounts of its input from the association areas
Intensity of Movements
of the cerebral cortex overlying the caudate nucleus,
mainly areas that also integrate the different types of
Two important capabilities of the brain in controlling
sensory and motor information into usable thought
movement are (1) to determine how rapidly the move-
patterns.
ment is to be performed and (2) to control how large
After the signals pass from the cerebral cortex to
the movement will be. For instance, a person may write
the caudate nucleus, they are next transmitted to the
the letter “a” slowly or rapidly. Also, he or she may
internal globus pallidus, then to the relay nuclei of the
write a small “a” on a piece of paper or a large “a” on
710
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
a chalkboard. Regardless of the choice, the propor-
conjectured in the last few sections is analytical deduc-
tional characteristics of the letter remain nearly the
tion rather than proven fact.
same.
In patients with severe lesions of the basal ganglia,
these timing and scaling functions are poor; in fact,
Functions of Specific
sometimes they are nonexistent. Here again, the basal
Neurotransmitter Substances in the
ganglia do not function alone; they function in close
Basal Ganglial System
association with the cerebral cortex. One especially
important cortical area is the posterior parietal cortex,
Figure 56-14 demonstrates the interplay of several
which is the locus of the spatial coordinates for motor
specific neurotransmitters that are known to function
control of all parts of the body as well as for the rela-
within the basal ganglia, showing (1) dopamine path-
tion of the body and its parts to all its surroundings.
ways from the substantia nigra to the caudate nucleus
Figure 56-13 shows the way in which a person lacking
and putamen, (2) gamma-aminobutyric acid (GABA)
a left posterior parietal cortex might draw the face of
pathways from the caudate nucleus and putamen to
another human being, providing proper proportions
the globus pallidus and substantia nigra, (3) acetyl-
for the right side of the face but almost ignoring the
choline pathways from the cortex to the caudate
left side (which is in his or her right field of vision).
nucleus and putamen, and (4) multiple general path-
Also, such a person will try always to avoid using his
ways from the brain stem that secrete norepinephrine,
or her right arm, right hand, or other portions of his or
serotonin, enkephalin, and several other neurotrans-
her right body for the performance of tasks, almost not
mitters in the basal ganglia as well as in other parts of
knowing that these parts of his or her body exist.
the cerebrum. In addition to all these are multiple glu-
Because the caudate circuit of the basal ganglial
tamate pathways that provide most of the excitatory
system functions mainly with association areas of the
signals
(not shown in the figure) that balance out
cerebral cortex such as the posterior parietal cortex,
the large numbers of inhibitory signals transmitted
presumably the timing and scaling of movements are
especially by the dopamine, GABA, and serotonin
functions of this caudate cognitive motor control
inhibitory transmitters. We will have more to say about
circuit. However, our understanding of function in the
some of these neurotransmitter and hormonal systems
basal ganglia is still so imprecise that much of what is
in subsequent sections when we discuss diseases of the
basal ganglia, as well as in subsequent chapters when
we discuss behavior, sleep, wakefulness, and functions
of the autonomic nervous system.
For the present, it should be remembered that
the neurotransmitter GABA always functions as an
inhibitory agent. Therefore, GABA neurons in the
feedback loops from the cortex through the basal
From cortex
Caudate nucleus
Ach
Putamen
GABA
Globus
pallidus
Substantia
nigra
Dopamine
From brain stem
1. Norepinephrine
2. Serotonin
3. Enkephalin
Figure 56-13
Figure 56-14
Typical drawing that might be made by a person who has severe
Neuronal pathways that secrete different types of neurotransmit-
damage in his or her left parietal cortex where the spatial coordi-
ter substances in the basal ganglia. Ach, acetylcholine; GABA,
nates of the right field of vision are stored.
gamma-aminobutyric acid.
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
711
ganglia and then back to the cortex make virtually
and akinesia. The reason for this is believed to be that
all these loops negative feedback loops, rather than
L-dopa is converted in the brain into dopamine, and the
positive feedback loops, thus lending stability to the
dopamine then restores the normal balance between
inhibition and excitation in the caudate nucleus and
motor control systems. Dopamine also functions as an
putamen. Administration of dopamine itself does not
inhibitory neurotransmitter in most parts of the brain,
have the same effect because dopamine has a chemical
so that it, too, undoubtedly functions as a stabilizer
structure that will not allow it to pass through the blood-
under some conditions.
brain barrier, even though the slightly different struc-
ture of L-dopa does allow it to pass.
Clinical Syndromes Resulting from
Treatment with l-Deprenyl. Another treatment for
Damage to the Basal Ganglia
Parkinson’s disease is the drug L-deprenyl. This drug
Aside from athetosis and hemiballismus, which have
inhibits monoamine oxidase, which is responsible for
already been mentioned in relation to lesions in the
destruction of most of the dopamine after it has been
globus pallidus and subthalamus, two other major dis-
secreted. Therefore, any dopamine that is released
eases result from damage in the basal ganglia. These are
remains in the basal ganglial tissues for a longer time.
Parkinson’s disease and Huntington’s disease.
In addition, for reasons not understood, this treatment
helps to slow destruction of the dopamine-secreting
Parkinson’s Disease
neurons in the substantia nigra. Therefore, appropriate
Parkinson’s disease, known also as paralysis agitans,
combinations of L-dopa therapy along with L-deprenyl
results from widespread destruction of that portion of
therapy usually provide much better treatment than use
the substantia nigra (the pars compacta) that sends
of one of these drugs alone.
dopamine-secreting nerve fibers to the caudate nucleus
and putamen. The disease is characterized by (1) rigid-
Treatment with Transplanted Fetal Dopamine Cells.
ity of much of the musculature of the body, (2) invol-
Transplantation of dopamine-secreting cells
(cells
untary tremor of the involved areas even when the
obtained from the brains of aborted fetuses) into the
person is resting at a fixed rate of 3 to 6 cycles per
caudate nuclei and putamen has been used with some
second, and (3) serious difficulty in initiating movement,
short-term success to treat Parkinson’s disease.
called akinesia.
However, the cells do not live for more than a few
The causes of these abnormal motor effects are
months. If persistence could be achieved, perhaps this
unknown. However, the dopamine secreted in the
would become the treatment of the future.
caudate nucleus and putamen is an inhibitory transmit-
ter; therefore, destruction of the dopaminergic neurons
Treatment by Destroying Part of the Feedback Circuitry
in the substantia nigra of the parkinsonian patient the-
in the Basal Ganglia. Because abnormal signals from
oretically would allow the caudate nucleus and putamen
the basal ganglia to the motor cortex cause most of the
to become overly active and possibly cause continuous
abnormalities in Parkinson’s disease, multiple attempts
output of excitatory signals to the corticospinal motor
have been made to treat these patients by blocking
control system. These signals could overly excite
these signals surgically. For a number of years, surgical
many or all of the muscles of the body, thus leading to
lesions were made in the ventrolateral and ventroante-
rigidity.
rior nuclei of the thalamus, which blocked part of the
Some of the feedback circuits might easily oscillate
feedback circuit from the basal ganglia to the cortex;
because of high feedback gains after loss of their inhi-
variable degrees of success were achieved—as well as
bition, leading to the tremor of Parkinson’s disease. This
sometimes serious neurological damage. In monkeys
tremor is quite different from that of cerebellar disease
with Parkinson’s disease, lesions placed in the subthal-
because it occurs during all waking hours and therefore
amus have been used, sometimes with surprisingly good
is an involuntary tremor, in contradistinction to cere-
results.
bellar tremor, which occurs only when the person per-
forms intentionally initiated movements and therefore
Huntington’s Disease (Huntington’s Chorea)
is called intention tremor.
Huntington’s disease is a hereditary disorder that
The akinesia that occurs in Parkinson’s disease is often
usually begins causing symptoms at age 30 to 40 years.
much more distressing to the patient than are the symp-
It is characterized at first by flicking movements in indi-
toms of muscle rigidity and tremor, because to perform
vidual muscles and then progressive severe distortional
even the simplest movement in severe parkinsonism, the
movements of the entire body. In addition, severe
person must exert the highest degree of concentration.
dementia develops along with the motor dysfunctions.
The mental effort, even mental anguish, that is necessary
The abnormal movements of Huntington’s disease
to make the desired movements is often at the limit of
are believed to be caused by loss of most of the cell
the patient’s willpower. Then, when the movements do
bodies of the GABA-secreting neurons in the caudate
occur, they are usually stiff and staccato in character
nucleus and putamen and of acetylcholine-secreting
instead of smooth. The cause of this akinesia is still spec-
neurons in many parts of the brain. The axon terminals
ulative. However, dopamine secretion in the limbic
of the GABA neurons normally inhibit portions of the
system, especially in the nucleus accumbens, is often
globus pallidus and substantia nigra. This loss of inhibi-
decreased along with its decrease in the basal ganglia. It
tion is believed to allow spontaneous outbursts of
has been suggested that this might reduce the psychic
globus pallidus and substantia nigra activity that cause
drive for motor activity so greatly that akinesia results.
the distortional movements.
The dementia in Huntington’s disease probably does
Treatment with l-Dopa. Administration of the drug L-
not result from the loss of GABA neurons but from the
dopa to patients with Parkinson’s disease usually ame-
loss of acetylcholine-secreting neurons, perhaps espe-
liorates many of the symptoms, especially the rigidity
cially in the thinking areas of the cerebral cortex.
712
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
The abnormal gene that causes Huntington’s disease
Associated Functions of the Cerebellum. The cerebellum
has been found; it has a many-times-repeating codon,
functions with all levels of muscle control. It functions
CAG, that codes for multiple extra glutamine amino
with the spinal cord especially to enhance the stretch
acids in the molecular structure of an abnormal neu-
reflex, so that when a contracting muscle encounters
ronal cell protein called huntingtin that causes the symp-
an unexpectedly heavy load, a long stretch reflex signal
toms. How this protein causes the disease effects is now
transmitted all the way through the cerebellum and
the question for major research effort.
back again to the cord strongly enhances the load-
resisting effect of the basic stretch reflex.
At the brain stem level, the cerebellum functions to
Integration of the Many
make the postural movements of the body—especially
Parts of the Total Motor
the rapid movements required by the equilibrium
Control System
system—smooth and continuous and without abnor-
mal oscillations.
Finally, we need to summarize as best we can what is
At the cerebral cortex level, the cerebellum oper-
known about overall control of movement. To do this,
ates in association with the cortex to provide many
let us first give a synopsis of the different levels of
accessory motor functions, especially to provide extra
control.
motor force for turning on muscle contraction rapidly
at the start of a movement. Near the end of each move-
ment, the cerebellum turns on antagonist muscles at
Spinal Level
exactly the right time and with proper force to stop the
movement at the intended point. Furthermore, there
Programmed in the spinal cord are local patterns of
is good physiologic evidence that all aspects of this
movement for all muscle areas of the body—for
turn-on/turn-off patterning by the cerebellum can be
instance, programmed withdrawal reflexes that pull
learned with experience.
any part of the body away from a source of pain. The
The cerebellum functions with the cerebral cortex at
cord is the locus also of complex patterns of rhythmi-
still another level of motor control: it helps to program
cal motions such as to-and-fro movement of the limbs
in advance muscle contractions that are required for
for walking, plus reciprocal motions on opposite sides
smooth progression from a present rapid movement in
of the body or of the hindlimbs versus the forelimbs
one direction to the next rapid movement in another
in four-legged animals.
direction, all this occurring in a fraction of a second.
All these programs of the cord can be commanded
The neural circuit for this passes from the cerebral
into action by higher levels of motor control, or they
cortex to the large lateral zones of the cerebellar hemi-
can be inhibited while the higher levels take over
spheres and then back to the cerebral cortex.
control.
The cerebellum functions mainly when muscle
movements have to be rapid. Without the cerebellum,
slow and calculated movements can still occur, but it
Hindbrain Level
is difficult for the corticospinal system to achieve rapid
and changing intended movements to execute a par-
The hindbrain provides two major functions for
ticular goal or especially to progress smoothly from
general motor control of the body: (1) maintenance of
one rapid movement to the next.
axial tone of the body for the purpose of standing and
(2) continuous modification of the degrees of tone in
the different muscles in response to information from
Associated Functions of the Basal Ganglia. The basal
the vestibular apparatuses for the purpose of main-
ganglia are essential to motor control in ways entirely
taining body equilibrium.
different from those of the cerebellum. Their most
important functions are (1) to help the cortex execute
subconscious but learned patterns of movement and (2)
Motor Cortex Level
to help plan multiple parallel and sequential patterns
of movement that the mind must put together to
The motor cortex system provides most of the acti-
accomplish a purposeful task.
vating motor signals to the spinal cord. It functions
The types of motor patterns that require the basal
partly by issuing sequential and parallel commands
ganglia include those for writing all the different
that set into motion various cord patterns of motor
letters of the alphabet, for throwing a ball, and for
action. It can also change the intensities of the differ-
typing. Also, the basal ganglia are required to modify
ent patterns or modify their timing or other charac-
these patterns for writing small or writing very large,
teristics. When needed, the corticospinal system can
thus controlling dimensions of the patterns.
bypass the cord patterns, replacing them with higher-
At a still higher level of control is another combined
level patterns from the brain stem or cerebral cortex.
cerebral and basal ganglia circuit, beginning in the
The cortical patterns usually are complex; also, they
thinking processes of the cerebrum to provide overall
can be “learned,” whereas cord patterns are mainly
sequential steps of action for responding to each new
determined by heredity and are said to be “hard
situation, such as planning one’s immediate motor
wired.”
response to an assailant who hits the person in the face
Chapter 56
Contributions of the Cerebellum and Basal Ganglia to Overall Motor Control
713
or one’s sequential response to an unexpectedly fond
de Zeeuw CI, Strata P, Voogd J: The Cerebellum: From Struc-
embrace.
ture to Control. Amsterdam: Elsevier, 1997.
Elble RJ: Origins of tremor. Lancet 355:1113, 2000.
Garwicz M, Ekerot C-F, Jorntell H: Organizational principles
What Drives Us to Action?
of cerebellar neuronal circuitry. News Physiol Sci 13:26,
1998.
Gibson AR, Horn KM, Pong M: Inhibitory control of olivary
What is it that arouses us from inactivity and sets into
discharge. Ann N Y Acad Sci 978:219, 2002.
play our trains of movement? We are beginning to
Goodale MA, Westwood DA: An evolving view of duplex
learn about the motivational systems of the brain.
vision: separate but interacting cortical pathways for
Basically, the brain has an older core located beneath,
perception and action. Curr Opin Neurobiol
14:203,
anterior, and lateral to the thalamus—including the
2004.
hypothalamus, amygdala, hippocampus, septal region
Ito M: Cerebellar long-term depression: characterization,
anterior to the hypothalamus and thalamus, and even
signal transduction, and functional roles. Physiol Rev
old regions of the thalamus and cerebral cortex them-
81:1143, 2001.
selves—all of which function together to initiate most
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
Science, 4th ed. New York: McGraw-Hill, 2000.
motor and other functional activities of the brain.
Keifer J, Houk JC: Motor function of the cerebel-
These areas are collectively called the limbic system
lorubrospinal system. Physiol Rev 74:509, 1994.
of the brain. We discuss this system in detail in
Li JY, Plomann M, Brundin P: Huntington’s disease: a synap-
Chapter 58.
topathy? Trends Mol Med 9:414, 2003.
Obeso JA, Rodriguez-Oroz M, Marin C, et al: The origin
of motor fluctuations in Parkinson’s disease: importance
References
of dopaminergic innervation and basal ganglia circuits.
Neurology 62(1 Suppl 1):S17, 2004.
Albin RL: Dominant ataxias and Friedreich ataxia: an
Ohyama T, Medina JF, Nores WL, Mauk MD: Trying to
update. Curr Opin Neurol 16:507, 2003.
understand the cerebellum well enough to build one. Ann
Barnham KJ, Masters CL, Bush AI: Neurodegenerative dis-
N Y Acad Sci 978:425, 2002.
eases and oxidative stress. Nat Rev Drug Discov 3:205,
Olanow CW: The scientific basis for the current treatment of
2004.
Parkinson’s disease. Annu Rev Med 55:41, 2004.
Brooks VB, Thach WT: Cerebellar control of posture and
Samii A, Nutt JG, Ransom BR: Parkinson’s disease. Lancet
movement. In: Handbook of Physiology, Sec. 1, Vol. II.
363:1783, 2004.
Bethesda: American Physiological Society, 1981, p 877.
Schweighofer N, Doya K, Kuroda S: Cerebellar aminergic
DeKosky ST, Marek K: Looking backward to move forward:
neuromodulation: towards a functional understanding.
early detection of neurodegenerative disorders. Science
Brain Res Brain Res Rev 44:103, 2004.
302:830, 2003.
Sethi KD: Tremor. Curr Opin Neurol 16:481, 2003.
C H A P T E R
5
7
Cerebral Cortex, Intellectual
Functions of the Brain, Learning
and Memory
It is ironic that of all the parts of the brain, we know
least about the functions of the cerebral cortex,
even though it is by far the largest portion of the
nervous system. But we do know the effects of
damage or specific stimulation in various portions
of the cortex. In the first part of this chapter, the
facts known about cortical function are discussed;
then basic theories of neuronal mechanisms
involved in thought processes, memory, analysis of sensory information, and so
forth are presented briefly.
Physiologic Anatomy of the Cerebral Cortex
The functional part of the cerebral cortex is a thin layer of neurons covering the
surface of all the convolutions of the cerebrum. This layer is only 2 to 5 millimeters
thick, with a total area of about one quarter of a square meter. The total cerebral
cortex contains about 100 billion neurons.
Figure 57-1 shows the typical histological structure of the neuronal surface of the
cerebral cortex, with its successive layers of different types of neurons. Most of the
neurons are of three types: (1) granular (also called stellate), (2) fusiform, and (3)
pyramidal, the last named for their characteristic pyramidal shape.
The granular neurons generally have short axons and, therefore, function mainly
as interneurons that transmit neural signals only short distances within the cortex
itself. Some are excitatory, releasing mainly the excitatory neurotransmitter gluta-
mate; others are inhibitory and release mainly the inhibitory neurotransmitter
gamma-aminobutyric acid (GABA). The sensory areas of the cortex as well as the
association areas between sensory and motor areas have large concentrations of
these granule cells, suggesting a high degree of intracortical processing of incoming
sensory signals within the sensory areas and association areas.
The pyramidal and fusiform cells give rise to almost all the output fibers from the
cortex. The pyramidal cells are larger and more numerous than the fusiform cells.
They are the source of the long, large nerve fibers that go all the way to the spinal
cord. They also give rise to most of the large subcortical association fiber bundles
that pass from one major part of the brain to another.
To the right in Figure 57-1 is shown the typical organization of nerve fibers within
the different layers of the cerebral cortex. Note particularly the large number of
horizontal fibers that extend between adjacent areas of the cortex, but note also the
vertical fibers that extend to and from the cortex to lower areas of the brain and
some all the way to the spinal cord or to distant regions of the cerebral cortex
through long association bundles.
The functions of the specific layers of the cerebral cortex are discussed in Chap-
ters 47 and 51. By way of review, let us recall that most incoming specific sensory
signals from the body terminate in cortical layer IV. Most of the output signals leave
the cortex through neurons located in layers V and VI; the very large fibers to the
brain stem and cord arise generally in layer V; and the tremendous numbers of fibers
to the thalamus arise in layer VI. Layers I, II, and III perform most of the intra-
cortical association functions, with especially large numbers of neurons in layers II
and III making short horizontal connections with adjacent cortical areas.
Anatomical and Functional Relations of the Cerebral Cortex to the Thalamus and Other Lower
Centers. All areas of the cerebral cortex have extensive to-and-fro efferent
and afferent connections with deeper structures of the brain. It is especially
714
Chapter 57
Cerebral Cortex, Intellectual Functions of the Brain, Learning and Memory
715
I
II
N. dorsalis
N. lateralis
medialis
posterior
III
Med. geniculate
body indeterminate
Lat.
geniculate
IV
body
Figure 57-2
V
Areas of the cerebral cortex that connect with specific portions of
the thalamus.
VIa
Supplementary
motor synergies
VIb
Hand
skills
Elaboration
Figure 57-1
of thought
Speech
Structure of the cerebral cortex, showing: I, molecular layer; II,
external granular layer; III, layer of pyramidal cells; IV, internal
Speech
Bilateral
granular layer; V, large pyramidal cell layer; and VI, layer of fusiform
vision
or polymorphic cells. (Redrawn from Ranson SW, Clark SL [after
Brodmann]: Anatomy of the Nervous System. Philadelphia: WB
Saunders Co, 1959.)
Contralateral
vision
Figure 57-3
Functional areas of the human cerebral cortex as determined by
important to emphasize the relation between the cere-
electrical stimulation of the cortex during neurosurgical operations
bral cortex and the thalamus. When the thalamus is
and by neurological examinations of patients with destroyed corti-
cal regions. (Redrawn from Penfield W, Rasmussen T: The Cerebral
damaged along with the cortex, the loss of cerebral
Cortex of Man: A Clinical Study of Localization of Function. New
function is far greater than when the cortex alone is
York: Hafner Co, 1968.)
damaged because thalamic excitation of the cortex is
necessary for almost all cortical activity.
Figure 57-2 shows the areas of the cerebral cortex
that connect with specific parts of the thalamus. These
Functions of Specific
connections act in two directions, both from the thala-
Cortical Areas
mus to the cortex and then from the cortex back to
essentially the same area of the thalamus. Further-
Studies in human beings by neurosurgeons, neurolo-
more, when the thalamic connections are cut, the func-
gists, and neuropathologists have shown that different
tions of the corresponding cortical area become almost
cerebral cortical areas have separate functions. Figure
entirely lost. Therefore, the cortex operates in close
57-3 is a map of some of these functions as determined
association with the thalamus and can almost be con-
by Penfield and Rasmussen from electrical stimulation
sidered both anatomically and functionally a unit with
of the cortex in awake patients or during neurological
the thalamus: for this reason, the thalamus and the
examination of patients after portions of the cortex
cortex together are sometimes called the thalamocor-
had been removed. The electrically stimulated patients
tical system. Almost all pathways from the sensory
told their thoughts evoked by the stimulation, and
receptors and sensory organs to the cortex pass
sometimes they experienced movements. Occasionally
through the thalamus, with the principal exception of
they spontaneously emitted a sound or even a word or
some sensory pathways of olfaction.
gave some other evidence of the stimulation.
716
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Primary somatic
association areas are (1) the parieto-occipitotemporal
Supplemental
Primary motor
association area, (2) the prefrontal association area,
Secondary somatic
and premotor
and (3) the limbic association area. Following are
explanations of the functions of these areas.
Secondary
visual
Parieto-occipitotemporal Association Area. This association
Parieto-
area lies in the large parietal and occipital cortical
Prefrontal
occipito-
space bounded by the somatosensory cortex anteri-
Association
temporal
orly, the visual cortex posteriorly, and the auditory
Area
Association
cortex laterally. As would be expected, it provides a
Area
high level of interpretative meaning for signals from
all the surrounding sensory areas. However, even the
Limbic
parieto-occipitotemporal association area has its own
Association
Primary
functional subareas, which are shown in Figure 57-5.
Area
visual
Primary
Secondary
auditory
auditory
1. Analysis of the Spatial Coordinates of the Body. An
area beginning in the posterior parietal cortex and
Figure 57-4
extending into the superior occipital cortex provides
continuous analysis of the spatial coordinates of all
Locations of major association areas of the cerebral cortex, as well
parts of the body as well as of the surroundings
as primary and secondary motor and sensory areas.
of the body. This area receives visual sensory informa-
tion from the posterior occipital cortex and simulta-
neous somatosensory information from the anterior
parietal cortex. From all this information, it computes
Putting large amounts of information together from
the coordinates of the visual, auditory, and body
many different sources gives a more general map, as
surroundings.
shown in Figure 57-4. This figure shows the major
primary and secondary premotor and supplementary
2. Area for Language Comprehension. The major area
motor areas of the cortex as well as the major primary
for language comprehension, called Wernicke’s area,
and secondary sensory areas for somatic sensation,
lies behind the primary auditory cortex in the posterior
vision, and hearing, all of which are discussed in earlier
part of the superior gyrus of the temporal lobe. We
chapters. The primary motor areas have direct con-
discuss this area much more fully later; it is the most
nections with specific muscles for causing discrete
important region of the entire brain for higher intel-
muscle movements. The primary sensory areas detect
lectual function because almost all such intellectual
specific sensations—visual, auditory, or somatic—
functions are language based.
transmitted directly to the brain from peripheral
sensory organs.
3. Area for Initial Processing of Visual Language
The secondary areas make sense out of the signals
(Reading). Posterior to the language comprehension
in the primary areas. For instance, the supplementary
area, lying mainly in the anterolateral region of the
and premotor areas function along with the primary
occipital lobe, is a visual association area that feeds
motor cortex and basal ganglia to provide “patterns”
visual information conveyed by words read from a
of motor activity. On the sensory side, the secondary
book into Wernicke’s area, the language comprehen-
sensory areas, located within a few centimeters of the
sion area. This so-called angular gyrus area is needed
primary areas, begin to analyze the meanings of the
to make meaning out of the visually perceived words.
specific sensory signals, such as (1) interpretation of
In its absence, a person can still have excellent
the shape or texture of an object in one’s hand; (2)
language comprehension through hearing but not
interpretation of color, light intensity, directions of
through reading.
lines and angles, and other aspects of vision; and (3)
interpretations of the meanings of sound tones and
4. Area for Naming Objects. In the most lateral por-
sequence of tones in the auditory signals.
tions of the anterior occipital lobe and posterior tem-
poral lobe is an area for naming objects. The names are
learned mainly through auditory input, whereas the
Association Areas
physical natures of the objects are learned mainly
through visual input. In turn, the names are essential
Figure 57-4 also shows several large areas of the cere-
for both auditory and visual language comprehension
bral cortex that do not fit into the rigid categories of
(functions performed in Wernicke’s area located imme-
primary and secondary motor and sensory areas. These
diately superior to the auditory “names” region and
areas are called association areas because they receive
anterior to the visual word processing area).
and analyze signals simultaneously from multiple
regions of both the motor and sensory cortices as well
Prefrontal Association Area. In Chapter 56, we learned
as from subcortical structures. Yet even the association
that the prefrontal association area functions in close
areas have their specializations. The most important
association with the motor cortex to plan complex
Chapter 57
Cerebral Cortex, Intellectual Functions of the Brain, Learning and Memory
717
Motor
Somato-
Spatial
sensory
coordinates
Planning complex
of body and
movements and
surroundings
elaboration of
thoughts
Visual
Word
Language
processing
formation
comprehension
of words
Auditory
intelligence
Broca‘s
Area
Behavior,
Vision
Figure 57-5
emotions,
Naming of
motivation
objects
Map of specific functional areas in
the cerebral cortex, showing espe-
cially Wernicke’s and Broca’s
areas for language comprehension
Limbic
Wernicke‘s
and speech production, which in 95
Association
Area
per cent of all people are located
Area
in the left hemisphere.
patterns and sequences of motor movements. To aid in
association cortex, as we discuss more fully later in the
this function, it receives strong input through a
chapter.
massive subcortical bundle of nerve fibers connecting
An especially interesting discovery is the following:
the parieto-occipitotemporal association area with the
When a person has already learned one language and
prefrontal association area. Through this bundle, the
then learns a new language, the area in the brain where
prefrontal cortex receives much preanalyzed sensory
the new language is stored is slightly removed from the
information, especially information on the spatial
storage area for the first language. If both languages
coordinates of the body that is necessary for planning
are learned simultaneously, they are stored together in
effective movements. Much of the output from the
the same area of the brain.
prefrontal area into the motor control system passes
Limbic Association Area. Figures 57-4 and 57-5 show still
through the caudate portion of the basal ganglia-
another association area called the limbic association
thalamic feedback circuit for motor planning, which
area. This area is found in the anterior pole of the tem-
provides many of the sequential and parallel compo-
poral lobe, in the ventral portion of the frontal lobe,
nents of movement stimulation.
and in the cingulate gyrus lying deep in the longitu-
The prefrontal association area is also essential to
dinal fissure on the midsurface of each cerebral
carrying out “thought” processes in the mind. This pre-
hemisphere. It is concerned primarily with behavior,
sumably results from some of the same capabilities of
emotions, and motivation. We will learn in Chapter 58
the prefrontal cortex that allow it to plan motor activ-
that the limbic cortex is part of a much more extensive
ities. It seems to be capable of processing nonmotor as
system, the limbic system, that includes a complex set
well as motor information from widespread areas of
of neuronal structures in the midbasal regions of the
the brain and therefore to achieve nonmotor types of
brain. This limbic system provides most of the emo-
thinking as well as motor types. In fact, the prefrontal
tional drives for activating other areas of the brain and
association area is frequently described simply as
even provides motivational drive for the process of
important for elaboration of thoughts, and it is said to
learning itself.
store on a short-term basis “working memories” that
are used to combine new thoughts while they are
Area for Recognition of Faces
entering the brain.
An interesting type of brain abnormality called
prosophenosia is inability to recognize faces. This
Broca’s Area. A special region in the frontal cortex,
occurs in people who have extensive damage on the
called Broca’s area, provides the neural circuitry for
medial undersides of both occipital lobes and along
word formation. This area, shown in Figure 57-5, is
the medioventral surfaces of the temporal lobes, as
located partly in the posterior lateral prefrontal cortex
shown in Figure 57-6. Loss of these face recognition
and partly in the premotor area. It is here that plans
areas, strangely enough, results in little other abnor-
and motor patterns for expressing individual words or
mality of brain function.
even short phrases are initiated and executed. This
One wonders why so much of the cerebral cortex
area also works in close association with Wernicke’s
should be reserved for the simple task of face recog-
language comprehension center in the temporal
nition. Most of our daily tasks involve associations
718
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Facial
recognition area
Motor
Primary
Somatic
Interpretative
Prefrontal
areas
Somatic
area
Broca's
speech
area
Visual
interpretative
Auditory
areas
interpretative
Primary
areas
visual
Wernicke‘s area
Temporal
Frontal
lobe
lobe
Figure 57-7
Organization of the somatic auditory and visual association areas
Figure 57-6
into a general mechanism for interpretation of sensory experience.
All of these feed also into Wernicke’s area, located in the postero-
Facial recognition areas located on the underside of the brain in the
superior portion of the temporal lobe. Note also the prefrontal area
medial occipital and temporal lobes. (Redrawn from Geschwind N:
and Broca’s speech area in the frontal lobe.
Specializations of the human brain. Sci Am 241:180, 1979. " 1979 by
Scientific American, Inc. All rights reserved.)
to read words from the printed page but be unable to
recognize the thought that is conveyed.
with other people, and one can see the importance of
Electrical stimulation in Wernicke’s area of a con-
this intellectual function.
scious person occasionally causes a highly complex
The occipital portion of this facial recognition area
thought. This is particularly true when the stimulation
is contiguous with the visual cortex, and the temporal
electrode is passed deep enough into the brain to
portion is closely associated with the limbic system
approach the corresponding connecting areas of the
that has to do with emotions, brain activation, and
thalamus. The types of thoughts that might be experi-
control of one’s behavioral response to the environ-
enced include complicated visual scenes that one
ment, as we see in Chapter 58.
might remember from childhood, auditory hallucina-
tions such as a specific musical piece, or even a state-
ment made by a specific person. For this reason, it is
Comprehensive Interpretative
believed that activation of Wernicke’s area can call
Function of the Posterior Superior
forth complicated memory patterns that involve more
Temporal Lobe—“Wernicke’s Area”
than one sensory modality even though most of the
(a General Interpretative Area)
individual memories may be stored elsewhere. This
belief is in accord with the importance of Wernicke’s
The somatic, visual, and auditory association areas all
area in interpreting the complicated meanings of dif-
meet one another in the posterior part of the superior
ferent patterns of sensory experiences.
temporal lobe, shown in Figure 57-7, where the tem-
poral, parietal, and occipital lobes all come together.
Angular Gyrus—Interpretation of Visual Information. The
This area of confluence of the different sensory inter-
angular gyrus is the most inferior portion of the pos-
pretative areas is especially highly developed in the
terior parietal lobe, lying immediately behind Wer-
dominant side of the brain—the left side in almost all
nicke’s area and fusing posteriorly into the visual areas
right-handed people—and it plays the greatest single
of the occipital lobe as well. If this region is destroyed
role of any part of the cerebral cortex for the higher
while Wernicke’s area in the temporal lobe is still
comprehension levels of brain function that we call
intact, the person can still interpret auditory experi-
intelligence. Therefore, this region has been called by
ences as usual, but the stream of visual experiences
different names suggestive of an area that has almost
passing into Wernicke’s area from the visual cortex is
global importance: the general interpretative area, the
mainly blocked. Therefore, the person may be able to
gnostic area, the knowing area, the tertiary association
see words and even know that they are words but not
area, and so forth. It is best known as Wernicke’s area
be able to interpret their meanings. This is the condi-
in honor of the neurologist who first described its
tion called dyslexia, or word blindness.
special significance in intellectual processes.
Let us again emphasize the global importance of
After severe damage in Wernicke’s area, a person
Wernicke’s area for processing most intellectual
might hear perfectly well and even recognize different
functions of the brain. Loss of this area in an
words but still be unable to arrange these words into
adult usually leads thereafter to a lifetime of almost
a coherent thought. Likewise, the person may be able
demented existence.
Chapter 57
Cerebral Cortex, Intellectual Functions of the Brain, Learning and Memory
719
Concept of the Dominant Hemisphere
Role of Language in the Function of
The general interpretative functions of Wernicke’s
Wernicke’s Area and in Intellectual Functions
area and the angular gyrus, as well as the functions
A major share of our sensory experience is converted
of the speech and motor control areas, are usually
into its language equivalent before being stored in the
much more highly developed in one cerebral
memory areas of the brain and before being processed
hemisphere than in the other. Therefore, this hemi-
for other intellectual purposes. For instance, when we
sphere is called the dominant hemisphere. In about 95
read a book, we do not store the visual images of
per cent of all people, the left hemisphere is the dom-
the printed words but instead store the words them-
inant one.
selves or their conveyed thoughts often in language
Even at birth, the area of the cortex that will even-
form.
tually become Wernicke’s area is as much as 50 per
The sensory area of the dominant hemisphere for
cent larger in the left hemisphere than in the right in
interpretation of language is Wernicke’s area, and this
more than one half of neonates. Therefore, it is easy
is closely associated with both the primary and sec-
to understand why the left side of the brain might
ondary hearing areas of the temporal lobe. This close
become dominant over the right side. However, if for
relation probably results from the fact that the first
some reason this left side area is damaged or removed
introduction to language is by way of hearing. Later
in very early childhood, the opposite side of the brain
in life, when visual perception of language through
will usually develop dominant characteristics.
the medium of reading develops, the visual informa-
A theory that can explain the capability of one
tion conveyed by written words is then presumably
hemisphere to dominate the other hemisphere is the
channeled through the angular gyrus, a visual associa-
following.
tion area, into the already developed Wernicke’s lan-
The attention of the “mind” seems to be directed
guage interpretative area of the dominant temporal
to one principal thought at a time. Presumably,
lobe.
because the left posterior temporal lobe at birth is
usually slightly larger than the right, the left side
normally begins to be used to a greater extent
Functions of the Parieto-
than the right. Thereafter, because of the tendency to
occipitotemporal Cortex in the
direct one’s attention to the better developed region,
Nondominant Hemisphere
the rate of learning in the cerebral hemisphere that
gains the first start increases rapidly, whereas in the
When Wernicke’s area in the dominant hemisphere of
opposite, less-used side, learning remains slight.
an adult person is destroyed, the person normally loses
Therefore, the left side normally becomes dominant
almost all intellectual functions associated with lan-
over the right.
guage or verbal symbolism, such as the ability to read,
In about 95 per cent of all people, the left temporal
the ability to perform mathematical operations, and
lobe and angular gyrus become dominant, and in the
even the ability to think through logical problems.
remaining 5 per cent, either both sides develop simul-
Many other types of interpretative capabilities, some
taneously to have dual function, or, more rarely, the
of which use the temporal lobe and angular gyrus
right side alone becomes highly developed, with full
regions of the opposite hemisphere, are retained.
dominance.
Psychological studies in patients with damage to the
As discussed later in the chapter, the premotor
nondominant hemisphere have suggested that this
speech area (Broca’s area), located far laterally in the
hemisphere may be especially important for under-
intermediate frontal lobe, is also almost always domi-
standing and interpreting music, nonverbal visual
nant on the left side of the brain. This speech area is
experiences (especially visual patterns), spatial rela-
responsible for formation of words by exciting simul-
tions between the person and their surroundings, the
taneously the laryngeal muscles, respiratory muscles,
significance of “body language” and intonations of
and muscles of the mouth.
people’s voices, and probably many somatic experi-
The motor areas for controlling hands are also
ences related to use of the limbs and hands. Thus, even
dominant in the left side of the brain in about 9 of
though we speak of the “dominant” hemisphere, this
10 persons, thus causing right-handedness in most
is primarily for language-based intellectual functions;
people.
the so-called nondominant hemisphere might actually
Although the interpretative areas of the temporal
be dominant for some other types of intelligence.
lobe and angular gyrus, as well as many of the motor
areas, are usually highly developed in only the left
hemisphere, these areas receive sensory information
from both hemispheres and are capable also of con-
Higher Intellectual Functions of the
trolling motor activities in both hemispheres. For this
Prefrontal Association Areas
purpose, they use mainly fiber pathways in the corpus
callosum for communication between the two hemi-
For years, it has been taught that the prefrontal cortex
spheres. This unitary, cross-feeding organization pre-
is the locus of “higher intellect” in the human being,
vents interference between the two sides of the brain;
principally because the main difference between the
such interference could create havoc with both mental
brains of monkeys and of human beings is the great
thoughts and motor responses.
prominence of the human prefrontal areas. Yet, efforts
720
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
to show that the prefrontal cortex is more important
prefrontal association areas have the capability of
in higher intellectual functions than other portions of
calling forth information from widespread areas of the
the brain have not been successful. Indeed, destruction
brain and using this information to achieve deeper
of the language comprehension area in the posterior
thought patterns for attaining goals. If these goals
superior temporal lobe (Wernicke’s area) and the
include motor action, so be it. If they do not, then
adjacent angular gyrus region in the dominant hemi-
the thought processes attain intellectual analytical
sphere causes much more harm to the intellect than
goals.
does destruction of the prefrontal areas. The prefrontal
Although people without prefrontal cortices can
areas do, however, have less definable but nevertheless
still think, they show little concerted thinking in logical
important intellectual functions of their own. These
sequence for longer than a few seconds or a minute or
functions can best be explained by describing what
so at most. One of the results is that people without
happens to patients in whom the prefrontal areas have
prefrontal cortices are easily distracted from their
become nonfunctional, as follows.
central theme of thought, whereas people with func-
Several decades ago, before the advent of modern
tioning prefrontal cortices can drive themselves to
drugs for treating psychiatric conditions, it was found
completion of their thought goals irrespective of
that some patients could receive significant relief from
distractions.
severe psychotic depression by severing the neuronal
connections between the prefrontal areas of the brain
Elaboration of Thought, Prognostication, and Performance of
and the remainder of the brain, that is, by a procedure
Higher Intellectual Functions by the Prefrontal Areas—Concept
called prefrontal lobotomy. This was done by inserting
of a “Working Memory.” Another function that has been
a blunt, thin-bladed knife through a small opening in
ascribed to the prefrontal areas by psychologists and
the lateral frontal skull on each side of the head and
neurologists is elaboration of thought. This means
slicing the brain at the back edge of the prefrontal
simply an increase in depth and abstractness of the dif-
lobes from top to bottom. Subsequent studies in these
ferent thoughts put together from multiple sources of
patients showed the following mental changes:
information. Psychological tests have shown that pre-
1. The patients lost their ability to solve complex
frontal lobectomized lower animals presented with
problems.
successive bits of sensory information fail to keep
2. They became unable to string together sequential
track of these bits even in temporary memory, proba-
tasks to reach complex goals.
bly because they are distracted so easily that they
3. They became unable to learn to do several
cannot hold thoughts long enough for memory storage
parallel tasks at the same time.
to take place.
4. Their level of aggressiveness was decreased,
This ability of the prefrontal areas to keep track of
sometimes markedly, and, in general, they lost
many bits of information simultaneously and to cause
ambition.
recall of this information instantaneously as it is
5. Their social responses were often inappropriate
needed for subsequent thoughts is called the brain’s
for the occasion, often including loss of morals
“working memory.” This could well explain the many
and little reticence in relation to sex and
functions of the brain that we associate with higher
excretion.
intelligence. In fact, studies have shown that the pre-
6. The patients could still talk and comprehend
frontal areas are divided into separate segments for
language, but they were unable to carry through
storing different types of temporary memory, such as
any long trains of thought, and their moods
one area for storing shape and form of an object or a
changed rapidly from sweetness to rage to
part of the body and another for storing movement.
exhilaration to madness.
By combining all these temporary bits of working
7. The patients could also still perform most of the
memory, we have the abilities to (1) prognosticate; (2)
usual patterns of motor function that they had
plan for the future; (3) delay action in response to
performed throughout life, but often without
incoming sensory signals so that the sensory informa-
purpose.
tion can be weighed until the best course of response
From this information, let us try to piece together a
is decided; (4) consider the consequences of motor
coherent understanding of the function of the pre-
actions before they are performed; (5) solve compli-
frontal association areas.
cated mathematical, legal, or philosophical problems;
(6) correlate all avenues of information in diagnosing
Decreased Aggressiveness and Inappropriate Social Re-
rare diseases; and (7) control our activities in accord
sponses. These two characteristics probably result
with moral laws.
from loss of the ventral parts of the frontal lobes on
the underside of the brain. As explained earlier and
Function of the Brain in
shown in Figures 57-4 and 57-5, this area is part of the
Communication—Language
limbic association cortex, rather than of the prefrontal
association cortex. This limbic area helps to control
Input and Language Output
behavior, which is discussed in detail in Chapter 58.
One of the most important differences between human
beings and lower animals is the facility with which
Inability to Progress Toward Goals or to Carry Through Sequen-
human beings can communicate with one another. Fur-
tial Thoughts. We learned earlier in this chapter that the
thermore, because neurological tests can easily assess
Chapter 57
Cerebral Cortex, Intellectual Functions of the Brain, Learning and Memory
721
These effects are called, respectively, auditory receptive
SPEAKING A HEARD WORD
Motor cortex
Arcuate fasciculus
aphasia and visual receptive aphasia or, more commonly,
word deafness and word blindness (also called dyslexia).
Wernicke’s Aphasia and Global Aphasia. Some people
are capable of understanding either the spoken word or
the written word but are unable to interpret the thought
that is expressed. This results most frequently when
Wernicke’s area in the posterior superior temporal gyrus
in the dominant hemisphere is damaged or destroyed.
Therefore, this type of aphasia is called Wernicke’s
aphasia.
When the lesion in Wernicke’s area is widespread and
extends (1) backward into the angular gyrus region, (2)
inferiorly into the lower areas of the temporal lobe, and
(3) superiorly into the superior border of the sylvian
Broca’s area
fissure, the person is likely to be almost totally
demented for language understanding or communica-
Wernicke’s area
tion and therefore is said to have global aphasia.
Primary auditory area
Motor Aspects of Communication. The process of speech
SPEAKING A WRITTEN WORD
Motor cortex
involves two principal stages of mentation: (1) forma-
tion in the mind of thoughts to be expressed as well
as choice of words to be used and then (2) motor
control of vocalization and the actual act of vocalization
itself.
The formation of thoughts and even most choices of
words are the function of sensory association areas of
the brain. Again, it is Wernicke’s area in the posterior
part of the superior temporal gyrus that is most impor-
tant for this ability. Therefore, a person with either Wer-
nicke’s aphasia or global aphasia is unable to formulate
the thoughts that are to be communicated. Or, if the
lesion is less severe, the person may be able to formu-
late the thoughts but unable to put together appropri-
ate sequences of words to express the thought. The
person sometimes is even fluent with words but the
Broca’s area
Primary
words are jumbled.
visual area
Wernicke’s area Angular gyrus
Loss of Broca’s Area Causes Motor Aphasia. Sometimes
a person is capable of deciding what he or she wants to
Figure 57-8
say but cannot make the vocal system emit words
instead of noises. This effect, called motor aphasia,
Brain pathways for (top) perceiving a heard word and then speak-
results from damage to Broca’s speech area, which lies
ing the same word, and (bottom) perceiving a written word and then
in the prefrontal and premotor facial region of the cere-
speaking the same word. (Redrawn from Geschwind N: Specializa-
tions of the human brain. Sci Am 241:180, 1979. " 1979 by Scientific
bral cortex—about 95 per cent of the time in the left
American, Inc. All rights reserved.)
hemisphere, as shown in Figures 57-5 and 57-8. There-
fore, the skilled motor patterns for control of the larynx,
lips, mouth, respiratory system, and other accessory
muscles of speech are all initiated from this area.
the ability of a person to communicate with others, we
know more about the sensory and motor systems
Articulation. Finally, we have the act of articulation,
related to communication than about any other
which means the muscular movements of the mouth,
segment of brain cortex function. Therefore, we will
tongue, larynx, vocal cords, and so forth that are respon-
review, with the help of anatomical maps of neural path-
sible for the intonations, timing, and rapid changes in
ways in Figure 57-8, function of the cortex in commu-
intensities of the sequential sounds. The facial and laryn-
nication. From this, one will see immediately how the
geal regions of the motor cortex activate these muscles,
principles of sensory analysis and motor control apply
and the cerebellum, basal ganglia, and sensory cortex all
to this art.
help to control the sequences and intensities of muscle
There are two aspects to communication: first, the
contractions, making liberal use of basal ganglial and
sensory aspect (language input), involving the ears and
cerebellar feedback mechanisms described in Chapters
eyes, and, second, the motor aspect (language output),
55 and 56. Destruction of any of these regions can cause
involving vocalization and its control.
either total or partial inability to speak distinctly.
Sensory Aspects of Communication. We noted earlier in the
Summary. Figure 57-8 shows two principal pathways for
chapter that destruction of portions of the auditory or
communication. The upper half of the figure shows the
visual association areas of the cortex can result in inabil-
pathway involved in hearing and speaking. This
ity to understand the spoken word or the written word.
sequence is the following: (1) reception in the primary
722
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
auditory area of the sound signals that encode the
Thus, one of the functions of the corpus callosum
words; (2) interpretation of the words in Wernicke’s
and the anterior commissure is to make information
area; (3) determination, also in Wernicke’s area, of the
stored in the cortex of one hemisphere available to
thoughts and the words to be spoken; (4) transmission
corresponding cortical areas of the opposite hemi-
of signals from Wernicke’s area to Broca’s area by way
sphere. Important examples of such cooperation
of the arcuate fasciculus; (5) activation of the skilled
between the two hemispheres are the following.
motor programs in Broca’s area for control of word for-
1.
Cutting the corpus callosum blocks transfer
mation; and (6) transmission of appropriate signals into
of information from Wernicke’s area of the
the motor cortex to control the speech muscles.
The lower figure illustrates the comparable steps in
dominant hemisphere to the motor cortex on
reading and then speaking in response. The initial recep-
the opposite side of the brain. Therefore, the
tive area for the words is in the primary visual area
intellectual functions of Wernicke’s area, located
rather than in the primary auditory area. Then the infor-
in the left hemisphere, lose control over the right
mation passes through early stages of interpretation in
motor cortex that initiates voluntary motor
the angular gyrus region and finally reaches its full level
functions of the left hand and arm, even though
of recognition in Wernicke’s area. From here, the
the usual subconscious movements of the left
sequence is the same as for speaking in response to the
hand and arm are normal.
spoken word.
2.
Cutting the corpus callosum prevents transfer of
somatic and visual information from the right
hemisphere into Wernicke’s area in the left
Function of the Corpus
dominant hemisphere. Therefore, somatic and
Callosum and Anterior
visual information from the left side of the body
Commissure to Transfer
frequently fails to reach this general interpretative
Thoughts, Memories, Training,
area of the brain and therefore cannot be used for
decision making.
and Other Information
3.
Finally, people whose corpus callosum is
Between the Two Cerebral
completely sectioned have two entirely separate
Hemispheres
conscious portions of the brain. For example, in a
teenage boy with a sectioned corpus callosum,
Fibers in the corpus callosum provide abundant bi-
only the left half of his brain could understand
directional neural connections between most of the
both the written word and the spoken word
respective cortical areas of the two cerebral hemi-
because the left side was the dominant
spheres except for the anterior portions of the tempo-
hemisphere. Conversely, the right side of the brain
ral lobes; these temporal areas, including especially
could understand the written word but not the
the amygdala, are interconnected by fibers that pass
spoken word. Furthermore, the right cortex could
through the anterior commissure.
elicit a motor action response to the written word
Because of the tremendous number of fibers in the
without the left cortex ever knowing why the
corpus callosum, it was assumed from the beginning
response was performed.
that this massive structure must have some important
The effect was quite different when an emotional
function to correlate activities of the two cerebral
response was evoked in the right side of the brain:
hemispheres. However, when the corpus callosum was
In this case, a subconscious emotional response
destroyed in laboratory animals, it was at first difficult
occurred in the left side of the brain as well. This
to discern deficits in brain function. Therefore, for a
undoubtedly occurred because the areas of the
long time, the function of the corpus callosum was a
two sides of the brain for emotions, the anterior
mystery.
temporal cortices and adjacent areas, were still
Properly designed psychological experiments have
communicating with each other through the
now demonstrated extremely important functions for
anterior commissure that was not sectioned. For
the corpus callosum and anterior commissure. These
instance, when the command “kiss” was written
functions can best be explained by describing one of
for the right half of his brain to see, the boy
the experiments: A monkey is first prepared by cutting
immediately and with full emotion said, “No way!”
the corpus callosum and splitting the optic chiasm lon-
This response required function of Wernicke’s
gitudinally, so that signals from each eye can go only
area and the motor areas for speech in the left
to the cerebral hemisphere on the side of the eye. Then
hemisphere because these left-side areas were
the monkey is taught to recognize different objects
necessary to speak the words “No way!” But when
with its right eye while its left eye is covered. Next,
questioned why he said this, the boy could not
the right eye is covered and the monkey is tested
explain. Thus, the two halves of the brain have
to determine whether its left eye can recognize the
independent capabilities for consciousness, memory
same objects. The answer to this is that the left eye
storage, communication, and control of motor
cannot recognize the objects. However, on repeating
activities. The corpus callosum is required for the
the same experiment in another monkey with the optic
two sides to operate cooperatively at the superficial
chiasm split but the corpus callosum intact, it is found
subconscious level, and the anterior commissure
invariably that recognition in one hemisphere of the
plays an important additional role in unifying the
brain creates recognition in the opposite hemisphere.
emotional responses of the two sides of the brain.
Chapter 57
Cerebral Cortex, Intellectual Functions of the Brain, Learning and Memory
723
Thoughts, Consciousness,
Experiments in lower animals have demonstrated
that memory traces can occur at all levels of the
and Memory
nervous system. Even spinal cord reflexes can change
at least slightly in response to repetitive cord activa-
Our most difficult problem in discussing conscious-
tion, and these reflex changes are part of the memory
ness, thoughts, memory, and learning is that we do not
process. Also, long-term memories result from
know the neural mechanisms of a thought and we
changed synaptic conduction in lower brain centers.
know little about the mechanisms of memory. We
However, most memory that we associate with intel-
know that destruction of large portions of the cerebral
lectual processes is based on memory traces in the
cortex does not prevent a person from having
cerebral cortex.
thoughts, but it does reduce the depth of the thoughts
and also the degree of awareness of the surroundings.
Positive and Negative Memory—“Sensitization” or “Habitua-
Each thought certainly involves simultaneous
tion” of Synaptic Transmission. Although we often think
signals in many portions of the cerebral cortex, thala-
of memories as being positive recollections of previous
mus, limbic system, and reticular formation of the
thoughts or experiences, probably the greater share
brain stem. Some crude thoughts probably depend
of our memories are negative memories, not positive.
almost entirely on lower centers; the thought of pain
That is, our brain is inundated with sensory informa-
is probably a good example because electrical stimu-
tion from all our senses. If our minds attempted
lation of the human cortex seldom elicits anything
to remember all this information, the memory capacity
more than mild pain, whereas stimulation of certain
of the brain would be exceeded within minutes. Fortu-
areas of the hypothalamus, amygdala, and mesen-
nately, the brain has the capability to learn to ignore
cephalon can cause excruciating pain. Conversely, a
information that is of no consequence.This results from
type of thought pattern that does require large
inhibition of the synaptic pathways for this type of
involvement of the cerebral cortex is that of vision,
information; the resulting effect is called habituation.
because loss of the visual cortex causes complete
This is a type of negative memory.
inability to perceive visual form or color.
Conversely, for incoming information that causes
We might formulate a provisional definition of a
important consequences such as pain or pleasure, the
thought in terms of neural activity as follows: A
brain has a different automatic capability of enhanc-
thought results from a “pattern” of stimulation of
ing and storing the memory traces. This is positive
many parts of the nervous system at the same time,
memory. It results from facilitation of the synaptic
probably involving most importantly the cerebral
pathways, and the process is called memory sensitiza-
cortex, thalamus, limbic system, and upper reticular
tion. We will learn later that special areas in the basal
formation of the brain stem. This is called the holistic
limbic regions of the brain determine whether infor-
theory of thoughts. The stimulated areas of the limbic
mation is important or unimportant and make the sub-
system, thalamus, and reticular formation are believed
conscious decision whether to store the thought as a
to determine the general nature of the thought, giving
sensitized memory trace or to suppress it.
it such qualities as pleasure, displeasure, pain, comfort,
crude modalities of sensation, localization to gross
Classification of Memories. We know that some memo-
areas of the body, and other general characteristics.
ries last for only a few seconds, whereas others last for
However, specific stimulated areas of the cerebral
hours, days, months, or years. For the purpose of dis-
cortex determine discrete characteristics of the
cussing these, let us use a common classification of
thought, such as (1) specific localization of sensations
memories that divides memories into (1) short-term
on the surface of the body and of objects in the fields
memory, which includes memories that last for
of vision, (2) the feeling of the texture of silk, (3) visual
seconds or at most minutes unless they are converted
recognition of the rectangular pattern of a concrete
into longer-term memories; (2) intermediate long-term
block wall, and (4) other individual characteristics that
memories, which last for days to weeks but then fade
enter into one’s overall awareness of a particular
away; and (3) long-term memory, which, once stored,
instant. Consciousness can perhaps be described as our
can be recalled up to years or even a lifetime later.
continuing stream of awareness of either our sur-
In addition to this general classification of memo-
roundings or our sequential thoughts.
ries, we also discussed earlier (in connection with the
prefrontal lobes) another type of memory, called
“working memory,” that includes mainly short-term
Memory—Roles of Synaptic
memory that is used during the course of intellectual
Facilitation and Synaptic Inhibition
reasoning but is terminated as each stage of the
problem is resolved.
Physiologically, memories are stored in the brain by
Memories are frequently classified according to the
changing the basic sensitivity of synaptic transmission
type of information that is stored. One of these classi-
between neurons as a result of previous neural activ-
fications divides memory into declarative memory and
ity. The new or facilitated pathways are called memory
skill memory, as follows:
traces. They are important because once the traces are
1. Declarative memory basically means memory of
established, they can be selectively activated by the
the various details of an integrated thought, such
thinking mind to reproduce the memories.
as memory of an important experience that
724
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
includes (1) memory of the surroundings, (2)
Noxious
memory of time relationships, (3) memory of
stimulus
causes of the experience, (4) memory of the
Facilitator
meaning of the experience, and (5) memory of
terminal
one’s deductions that were left in the person’s
Serotonin
mind.
Sensory
2. Skill memory is frequently associated with motor
stimulus
activities of the person’s body, such as all the skills
developed for hitting a tennis ball, including
cAMP
Sensory
automatic memories to (1) sight the ball, (2)
terminal
calculate the relationship and speed of the ball to
the racquet, and (3) deduce rapidly the motions of
Calcium
Calcium
the body, the arms, and the racquet required to hit
channels
ions
the ball as desired—all of these activated instantly
based on previous learning of the game of
Figure 57-9
tennis—then moving on to the next stroke of the
Memory system that has been discovered in the snail Aplysia.
game while forgetting the details of the previous
stroke.
memories lasting from a few minutes up to 3 weeks in
the large snail Aplysia. In this figure, there are two
Short-Term Memory
synaptic terminals. One terminal is from a sensory
input neuron and terminates directly on the surface of
Short-term memory is typified by one’s memory of 7
the neuron that is to be stimulated; this is called the
to 10 numerals in a telephone number (or 7 to 10 other
sensory terminal. The other terminal is a presynaptic
discrete facts) for a few seconds to a few minutes at a
ending that lies on the surface of the sensory terminal,
time but lasting only as long as the person continues
and it is called the facilitator terminal. When the
to think about the numbers or facts.
sensory terminal is stimulated repeatedly but without
Many physiologists have suggested that this short-
stimulation of the facilitator terminal, signal transmis-
term memory is caused by continual neural activity
sion at first is great, but it becomes less and less intense
resulting from nerve signals that travel around and
with repeated stimulation until transmission almost
around a temporary memory trace in a circuit of rever-
ceases. This phenomenon is habituation, as was
berating neurons. It has not yet been possible to prove
explained previously. It is a type of negative memory
this theory. Another possible explanation of short-
that causes the neuronal circuit to lose its response to
term memory is presynaptic facilitation or inhibition.
repeated events that are insignificant.
This occurs at synapses that lie on terminal nerve
Conversely, if a noxious stimulus excites the facili-
fibrils immediately before these fibrils synapse with a
tator terminal at the same time that the sensory ter-
subsequent neuron. The neurotransmitter chemicals
minal is stimulated, then instead of the transmitted
secreted at such terminals frequently cause facilitation
signal into the postsynaptic neuron becoming pro-
or inhibition lasting for seconds up to several minutes.
gressively weaker, the ease of transmission becomes
Circuits of this type could lead to short-term memory.
stronger and stronger; and it will remain strong for
minutes, hours, days, or, with more intense training, up
to about 3 weeks even without further stimulation of
Intermediate Long-Term Memory
the facilitator terminal. Thus, the noxious stimulus
causes the memory pathway through the sensory
Intermediate long-term memories may last for many
terminal to become facilitated for days or weeks
minutes or even weeks. They will eventually be lost
thereafter. It is especially interesting that even after
unless the memory traces are activated enough to
habituation has occurred, this pathway can be con-
become more permanent; then they are classified as
verted back to a facilitated pathway with only a few
long-term memories. Experiments in primitive animals
noxious stimuli.
have demonstrated that memories of the intermediate
long-term type can result from temporary chemical or
Molecular Mechanism of Intermediate Memory
physical changes, or both, in either the synapse presy-
Mechanism for Habituation. At the molecular level,
naptic terminals or the synapse postsynaptic mem-
the habituation effect in the sensory terminal results
brane, changes that can persist for a few minutes up to
from progressive closure of calcium channels through
several weeks. These mechanisms are so important
the terminal membrane, though the cause of this
that they deserve special description.
calcium channel closure is not fully known. Neverthe-
less, much smaller than normal amounts of calcium
Memory Based on Chemical Changes in
ions can diffuse into the habituated terminal, and
the Presynaptic Terminal or Postsynaptic
much less sensory terminal transmitter is therefore
Neuronal Membrane
released because calcium entry is the principal stim-
Figure 57-9 shows a mechanism of memory studied
ulus for transmitter release
(as was discussed in
especially by Kandel and his colleagues that can cause
Chapter 45).
Chapter 57
Cerebral Cortex, Intellectual Functions of the Brain, Learning and Memory
725
Mechanism for Facilitation. In the case of facilitation,
Structural Changes Occur in Synapses During
at least part of the molecular mechanism is believed
the Development of Long-Term Memory
to be the following:
Electron microscopic pictures taken from invertebrate
1.
Stimulation of the facilitator presynaptic terminal
animals have demonstrated multiple physical struc-
at the same time that the sensory terminal is
tural changes in many synapses during development of
stimulated causes serotonin release at the
long-term memory traces. The structural changes will
facilitator synapse on the surface of the sensory
not occur if a drug is given that blocks DNA stimula-
terminal.
tion of protein replication in the presynaptic neuron;
2.
The serotonin acts on serotonin receptors in the
nor will the permanent memory trace develop. There-
sensory terminal membrane, and these receptors
fore, it appears that development of true long-term
activate the enzyme adenyl cyclase inside the
memory depends on physically restructuring the
membrane. And, finally, the adenyl cyclase causes
synapses themselves in a way that changes their sensi-
formation of cyclic adenosine monophosphate
tivity for transmitting nervous signals.
(cAMP) also inside the sensory presynaptic
The most important of the physical structural
terminal.
changes that occur are the following:
3.
The cyclic AMP activates a protein kinase that
1. Increase in vesicle release sites for secretion of
causes phosphorylation of a protein that itself is
transmitter substance.
part of the potassium channels in the sensory
2. Increase in number of transmitter vesicles
synaptic terminal membrane; this in turn blocks
released.
the channels for potassium conductance. The
3. Increase in number of presynaptic terminals.
blockage can last for minutes up to several
4. Changes in structures of the dendritic spines that
weeks.
permit transmission of stronger signals.
4.
Lack of potassium conductance causes a greatly
Thus, in several different ways, the structural capa-
prolonged action potential in the synaptic
bility of synapses to transmit signals appears to
terminal because flow of potassium ions out of the
increase during establishment of true long-term
terminal is necessary for rapid recovery from the
memory traces.
action potential.
Number of Neurons and Their Connectivities
5.
The prolonged action potential causes prolonged
Often Change Significantly During Learning
activation of the calcium channels, allowing
During the first few weeks, months, and perhaps even
tremendous quantities of calcium ions to enter the
year or so of life, many parts of the brain produce a
sensory synaptic terminal. These calcium ions
great excess of neurons, and the neurons send out
cause greatly increased transmitter release by the
numerous axon branches to make connections with
synapse, thereby markedly facilitating synaptic
other neurons. If the new axons fail to connect with
transmission to the subsequent neuron.
appropriate subsequent neurons, muscle cells, or gland
Thus, in a very indirect way, the associative effect of
cells, the new axons themselves will dissolute within a
stimulating the facilitator terminal at the same time
few weeks. Thus, the number of neuronal connections
that the sensory terminal is stimulated causes pro-
is determined by specific nerve growth factors released
longed increase in excitatory sensitivity of the sensory
retrogradely from the stimulated cells. Furthermore,
terminal, and this establishes the memory trace. Studies
when insufficient connectivity occurs, the entire
by Byrne and colleagues, also in the snail Aplysia, have
neuron that is sending out the axon branches might
suggested still another mechanism of synaptic memory.
eventually disappear.
Their studies have shown that stimuli from separate
Therefore, soon after birth, there is a principle of
sources acting on a single neuron, under appropriate
“use it or lose it” that governs the final number of
conditions, can cause long-term changes in membrane
neurons and their connectivities in respective parts of
properties of the post-synaptic neuron instead of in the
the human nervous system. This is a type of learning.
presynaptic neuronal membrane, but leading to essen-
For example, if one eye of a newborn animal is covered
tially the same memory effects.
for many weeks after birth, neurons in alternate stripes
of the cerebral visual cortex—neurons normally con-
nected to the covered eye—will degenerate, and the
Long-Term Memory
covered eye will remain either partially or totally blind
for the remainder of life. Until recently, it was believed
There is no obvious demarcation between the more
that very little “learning” is achieved in adult human
prolonged types of intermediate long-term memory
beings and animals by modification of numbers of
and true long-term memory. The distinction is one of
neurons in the memory circuits; however, recent
degree. However, long-term memory is generally
research suggests that even adults use this mechanism
believed to result from actual structural changes,
to at least some extent.
instead of only chemical changes, at the synapses, and
these enhance or suppress signal conduction. Again,
let us recall experiments in primitive animals (where
Consolidation of Memory
the nervous systems are much easier to study) that
have aided immensely in understanding possible
For short-term memory to be converted into long-term
mechanisms of long-term memory.
memory that can be recalled weeks or years later, it
726
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
must become “consolidated.” That is, the short-term
removed for the treatment of epilepsy in a few
memory if activated repeatedly will initiate chemical,
patients. This procedure does not seriously affect the
physical, and anatomical changes in the synapses that
person’s memory for information stored in the brain
are responsible for the long-term type of memory. This
before removal of the hippocampi. However, after
process requires 5 to 10 minutes for minimal consoli-
removal, these people have virtually no capability
dation and 1 hour or more for strong consolidation.
thereafter for storing verbal and symbolic types of
For instance, if a strong sensory impression is made on
memories (declarative types of memory) in long-term
the brain but is then followed within a minute or so by
memory, or even in intermediate memory lasting
an electrically induced brain convulsion, the sensory
longer than a few minutes. Therefore, these people are
experience will not be remembered. Likewise, brain
unable to establish new long-term memories of those
concussion, sudden application of deep general
types of information that are the basis of intelligence.
anesthesia, or any other effect that temporarily
This is called anterograde amnesia.
blocks the dynamic function of the brain can prevent
But why are the hippocampi so important in helping
consolidation.
the brain to store new memories? The probable
Consolidation and the time required for it to occur
answer is that the hippocampi are among the most
can probably be explained by the phenomenon of
important output pathways from the “reward” and
rehearsal of the short-term memory as follows.
“punishment” areas of the limbic system, as explained
in Chapter 58. Sensory stimuli or thoughts that cause
Rehearsal Enhances the Transference of Short-Term Memory
pain or aversion excite the limbic punishment centers,
into Long-Term Memory. Psychological studies have
and stimuli that cause pleasure, happiness, or sense of
shown that rehearsal of the same information again
reward excite the limbic reward centers. All these
and again in the mind accelerates and potentiates the
together provide the background mood and motiva-
degree of transfer of short-term memory into long-
tions of the person. Among these motivations is the
term memory and therefore accelerates and enhances
drive in the brain to remember those experiences and
consolidation. The brain has a natural tendency to
thoughts that are either pleasant or unpleasant. The
rehearse newfound information, especially newfound
hippocampi especially and to a lesser degree the dorsal
information that catches the mind’s attention. There-
medial nuclei of the thalamus, another limbic struc-
fore, over a period of time, the important features of
ture, have proved especially important in making the
sensory experiences become progressively more and
decision about which of our thoughts are important
more fixed in the memory stores. This explains why a
enough on a basis of reward or punishment to be
person can remember small amounts of information
worthy of memory.
studied in depth far better than large amounts of infor-
mation studied only superficially. It also explains why
Retrograde Amnesia—Inability to Recall Memories from the
a person who is wide awake can consolidate memories
Past. When retrograde amnesia occurs, the degree of
far better than a person who is in a state of mental
amnesia for recent events is likely to be much greater
fatigue.
than for events of the distant past. The reason for this
difference is probably that the distant memories have
New Memories Are Codified During Consolidation. One of the
been rehearsed so many times that the memory traces
most important features of consolidation is that new
are deeply engrained, and elements of these memories
memories are codified into different classes of infor-
are stored in widespread areas of the brain.
mation. During this process, similar types of informa-
In some people who have hippocampal lesions,
tion are pulled from the memory storage bins and used
some degree of retrograde amnesia occurs along with
to help process the new information. The new and old
anterograde amnesia, which suggests that these two
are compared for similarities and differences, and part
types of amnesia are at least partially related and
of the storage process is to store the information about
that hippocampal lesions can cause both. However,
these similarities and differences, rather than to store
damage in some thalamic areas may lead specifically
the new information unprocessed. Thus, during con-
to retrograde amnesia without causing significant
solidation, the new memories are not stored randomly
anterograde amnesia. A possible explanation of this is
in the brain but are stored in direct association with
that the thalamus may play a role in helping the person
other memories of the same type. This is necessary if
“search” the memory storehouses and thus “read out”
one is to be able to “search” the memory store at a
the memories. That is, the memory process not only
later date to find the required information.
requires the storing of memories but also an ability to
search and find the memory at a later date. The possi-
Role of Specific Parts of the Brain in the
ble function of the thalamus in this process is discussed
Memory Process
further in Chapter 58.
Hippocampus Promotes Storage of Memories—Anterograde
Hippocampi Are Not Important in Reflexive Learning. People
Amnesia After Hippocampal Lesions. The hippocampus is
with hippocampal lesions usually do not have difficulty
the most medial portion of the temporal lobe cortex,
in learning physical skills that do not involve verbal-
where it folds first medially underneath the brain and
ization or symbolic types of intelligence. For instance,
then upward into the lower, inside surface of the
these people can still learn the rapid hand and
lateral ventricle. The two hippocampi have been
physical skills required in many types of sports. This
Chapter 57
Cerebral Cortex, Intellectual Functions of the Brain, Learning and Memory
727
type of learning is called skill learning or reflexive
Hamann S, Canli T: Individual differences in emotion pro-
learning; it depends on physically repeating the
cessing. Curr Opin Neurobiol 14:233, 2004.
required tasks over and over again, rather than on
Kandel ER: The molecular biology of memory storage: a
dialogue between genes and synapses. Science 294:1030,
symbolical rehearsing in the mind.
2001.
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
Science, 4th ed. New York: McGraw-Hill, 2000.
References
LeDoux JE: Emotion, memory and the brain: the neural
routes underlying the formation of memories about prim-
Baddeley A: Working memory: looking back and looking
itive emotional experiences, such as fear, have been
forward. Nat Rev Neurosci 4:829, 2003.
traced. Sci Am June: 50, 1994.
Blank T, Nijholt I, Spiess J: Molecular determinants mediat-
Leuner B, Shors TJ: New spines, new memories. Mol Neuro-
ing effects of acute stress on hippocampus-dependent
biol 29:117, 2004.
synaptic plasticity and learning. Mol Neurobiol 29:131,
Lynch MA: Long-term potentiation and memory. Physiol
2004.
Rev 84:87, 2004.
Bookheimer S: Functional MRI of language: new
Miller BL, Cummings JL: The Human Frontal Lobes. New
approaches to understanding the cortical organization of
York: Guilford Press, 1998.
semantic processing. Annu Rev Neurosci 25:151, 2002.
Nakazawa K, McHugh TJ, Wilson MA, Tonegawa S: NMDA
Conlon R, Hobson JA: Understanding the Human Mind.
receptors, place cells and hippocampal spatial memory.
New York: John Wiley, 1999.
Nat Rev Neurosci 5:361, 2004.
Dash PK, Hebert AE, Runyan JD: A unified theory for
Phelps EA: Human emotion and memory: interactions of the
systems and cellular memory consolidation. Brain Res
amygdala and hippocampal complex. Curr Opin Neuro-
Brain Res Rev 45:30, 2004.
biol 14:198, 2004.
Dick P, Katsuyuki S: The prefrontal cortex and working
Sah P, Faber ES, Lopez De Armentia M, Power J: The amyg-
memory: physiology and brain imaging. Curr Opin Neu-
daloid complex: anatomy and physiology. Physiol Rev
robiol 14:163, 2004.
83:803, 2003.
Dudai Y: The neurobiology of consolidations, or, how stable
Shors TJ: Memory traces of trace memories: neurogenesis,
is the engram? Annu Rev Psychol 55:51, 2004.
synaptogenesis and awareness. Trends Neurosci 27:250,
Ehrlich YM: Molecular and Cellular Mechanisms of Neu-
2004.
ronal Plasticity. New York: Plenum Press, 1998.
Wixted JT: The psychology and neuroscience of forgetting.
Guillery RW: Branching thalamic afferents link action and
Annu Rev Psychol 55:235, 2004.
perception. J Neurophysiol 90:539, 2003.
Haines DE: Fundamental Neuroscience. New York:
Churchill Livingstone, 1997.
C H A P T E R
5
8
Behavioral and Motivational
Mechanisms of the Brain—The
Limbic System and the
Hypothalamus
Control of behavior is a function of the entire
nervous system. Even the wakefulness and sleep
cycle discussed in Chapter 59 is one of our most
important behavioral patterns.
In this chapter, we deal first with those mecha-
nisms that control levels of activity in the different
parts of the brain. Then we discuss the causes of
motivational drives, especially motivational control
of the learning process and feelings of pleasure and punishment. These func-
tions of the nervous system are performed mainly by the basal regions of the
brain, which together are loosely called the limbic system, meaning the “border”
system.
Activating-Driving Systems of the Brain
Without continuous transmission of nerve signals from the lower brain into the
cerebrum, the cerebrum becomes useless. In fact, severe compression of the
brain stem at the juncture between the mesencephalon and cerebrum, as some-
times results from a pineal tumor, often causes the person to go into unremit-
ting coma lasting for the remainder of his or her life.
Nerve signals in the brain stem activate the cerebral part of the brain in two
ways: (1) by directly stimulating a background level of neuronal activity in wide
areas of the brain and (2) by activating neurohormonal systems that release
specific facilitory or inhibitory hormone-like neurotransmitter substances into
selected areas of the brain.
Control of Cerebral Activity by Continuous Excitatory
Signals from the Brain Stem
Reticular Excitatory Area of the Brain Stem
Figure 58-1 shows a general system for controlling the level of activity of the
brain. The central driving component of this system is an excitatory area located
in the reticular substance of the pons and mesencephalon. This area is also known
by the name bulboreticular facilitory area. We also discuss this area in Chapter
55 because it is the same brain stem reticular area that transmits facilitory
signals downward to the spinal cord to maintain tone in the antigravity muscles
and to control levels of activity of the spinal cord reflexes. In addition to these
downward signals, this area also sends a profusion of signals in the upward direc-
tion. Most of these go first to the thalamus, where they excite a different set of
neurons that transmit nerve signals to all regions of the cerebral cortex as well
as to multiple subcortical areas.
The signals passing through the thalamus are of two types. One type is rapidly
transmitted action potentials that excite the cerebrum for only a few millisec-
onds. These originate from large neuronal cell bodies that lie throughout the
728
Chapter 58
Behavioral and Motivational Mechanisms of the Brain—The Limbic System and the Hypothalamus
729
Thalamus
nerves enter the pons. These nerves are the highest
nerves entering the brain that transmit significant
numbers of somatosensory signals into the brain.
When all these input sensory signals are gone, the level
of activity in the brain excitatory area diminishes
abruptly, and the brain proceeds instantly to a state
of greatly reduced activity, approaching a permanent
state of coma. But when the brain stem is transected
below the fifth nerves, which leaves much input of
sensory signals from the facial and oral regions, the
coma is averted.
Increased Activity of the Excitatory Area Caused by Feedback
Signals Returning from the Cerebral Cortex. Not only do
excitatory signals pass to the cerebral cortex from
Excitatory area
the bulboreticular excitatory area of the brain stem,
but feedback signals also return from the cerebral
5th Cranial nerve
cortex back to this same area. Therefore, any time
the cerebral cortex becomes activated by either
brain thought processes or motor processes, signals
Inhibitory area
are sent from the cortex to the brain stem excitatory
area, which in turn sends still more excitatory signals
to the cortex. This helps to maintain the level of
excitation of the cerebral cortex or even to enhance it.
This is a general mechanism of positive feedback that
allows any beginning activity in the cerebral cortex to
support still more activity, thus leading to an “awake”
Figure 58-1
mind.
Excitatory-activating
system
of the brain. Also shown is
an
Thalamus Is a Distribution Center That Controls Activity in Spe-
inhibitory area in the medulla that can inhibit or depress the acti-
vating system.
cific Regions of the Cortex. As pointed out in Chapter 57
and shown in Figure 57-2, almost every area of the
cerebral cortex connects with its own highly specific
brain stem reticular area. Their nerve endings release
area in the thalamus. Therefore, electrical stimulation
the neurotransmitter substance acetylcholine, which
of a specific point in the thalamus generally activates
serves as an excitatory agent, lasting for only a few mil-
its own specific small region of the cortex. Further-
liseconds before it is destroyed.
more, signals regularly reverberate back and forth
The second type of excitatory signal originates from
between the thalamus and the cerebral cortex, the
large numbers of small neurons spread throughout the
thalamus exciting the cortex and the cortex then re-
brain stem reticular excitatory area. Again, most of
exciting the thalamus by way of return fibers. It has
these pass to the thalamus, but this time through small,
been suggested that the thinking process establishes
slowly conducting fibers that synapse mainly in the
long-term memories by activating such back-and-forth
intralaminar nuclei of the thalamus and in the reticu-
reverberation of signals.
lar nuclei over the surface of the thalamus. From here,
Can the thalamus also function to call forth specific
additional small fibers are distributed everywhere in
memories from the cortex or to activate specific
the cerebral cortex. The excitatory effect caused by
thought processes? Proof of this is still lacking, but the
this system of fibers can build up progressively for
thalamus does have appropriate neuronal circuitry for
many seconds to a minute or more, which suggests that
these purposes.
its signals are especially important for controlling
longer-term background excitability level of the brain.
A Reticular Inhibitory Area Located in the
Lower Brain Stem
Excitation of the Excitatory Area by Peripheral Sensory Signals.
Figure 58-1 shows still another area that is important
The level of activity of the excitatory area in the brain
in controlling brain activity. This is the reticular
stem, and therefore the level of activity of the entire
inhibitory area, located medially and ventrally in the
brain, is determined to a great extent by the number
medulla. In Chapter 55, we learned that this area can
and type of sensory signals that enter the brain from
inhibit the reticular facilitory area of the upper brain
the periphery. Pain signals in particular increase activ-
stem and thereby decrease activity in the superior por-
ity in this excitatory area and therefore strongly excite
tions of the brain as well. One of the mechanisms for
the brain to attention.
this is to excite serotonergic neurons; these in turn
The importance of sensory signals in activating the
secrete the inhibitory neurohormone serotonin at
excitatory area is demonstrated by the effect of cutting
crucial points in the brain; we will discuss this in more
the brain stem above the point where the fifth cerebral
detail later.
730
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Neurohormonal Control of
as an excitatory hormone, whereas serotonin usually is
inhibitory, and dopamine is excitatory in some areas
Brain Activity
but inhibitory in others. As would be expected, these
three systems have different effects on levels of
Aside from direct control of brain activity by specific
excitability in different parts of the brain. The norepi-
transmission of nerve signals from the lower brain
nephrine system spreads to virtually every area of the
areas to the cortical regions of the brain, still another
brain, whereas the serotonin and dopamine systems
physiologic mechanism is very often used to control
are directed much more to specific brain regions—
brain activity. This is to secrete excitatory or inhibitory
the dopamine system mainly into the basal ganglial
neurotransmitter hormonal agents into the substance
regions and the serotonin system more into the
of the brain. These neurohormones often persist for
midline structures.
minutes or hours and thereby provide long periods of
control, rather than just instantaneous activation or
Neurohormonal Systems in the Human Brain. Figure 58-3
inhibition.
shows the brain stem areas in the human brain for acti-
Figure 58-2 shows three neurohormonal systems
vating four neurohormonal systems, the same three
that have been mapped in detail in the rat brain: (1) a
discussed for the rat and one other, the acetylcholine
norepinephrine system, (2) a dopamine system, and (3)
system. Some of the specific functions of these are as
a serotonin system. Norepinephrine usually functions
follows:
1. The locus ceruleus and the norepinephrine system.
The locus ceruleus is a small area located
Cerebellum
bilaterally and posteriorly at the juncture between
the pons and mesencephalon. Nerve fibers
from this area spread throughout the brain, the
same as shown for the rat in the top frame of
Figure 58-2, and they secrete norepinephrine. The
norepinephrine generally excites the brain to
increased activity. However, it has inhibitory
effects in a few brain areas because of inhibitory
Brain stem
receptors at certain neuronal synapses. In Chapter
Olfactory
59, we will see that this system probably plays an
region
Basal brain areas
important role in causing dreaming, thus leading
Locus cerulus
to a type of sleep called rapid eye movement
NOREPINEPHRINE
sleep (REM sleep).
2. The substantia nigra and the dopamine system.
The substantia nigra is discussed in Chapter 56 in
Frontal
cortex
Cingulate
cortex
Caudate nucleus
To diencephalon
and cerebrum
DOPAMINE
Mesencephalon
Substantia nigra
(dopamine)
To cerebellum
Gigantocellular
neurons of
reticular formation
Pons
(acetylcholine)
Locus ceruleus
(norepinephrine)
Medulla
Nuclei of the raphe
(serotonin)
Midline nuclei
SEROTONIN
To cord
Figure 58-2
Figure 58-3
Three neurohormonal systems that have been mapped in the rat
brain: a norepinephrine system, a dopamine system, and a sero-
Multiple centers in the brain stem, the neurons of which secrete
tonin system. (Adapted from Kelly, after Cooper, Bloom, and Roth,
different transmitter substances (specified in parentheses). These
in Kandel ER, Schwartz JH: Principles of Neural Science, 2nd ed.
neurons send control signals upward into the diencephalon and
New York: Elsevier, 1985.)
cerebrum and downward into the spinal cord.
Chapter 58
Behavioral and Motivational Mechanisms of the Brain—The Limbic System and the Hypothalamus
731
relation to the basal ganglia. It lies anteriorly in
limbic system, the term limbic system has been
the superior mesencephalon, and its neurons send
expanded to mean the entire neuronal circuitry that
nerve endings mainly to the caudate nucleus and
controls emotional behavior and motivational drives.
putamen of the cerebrum, where they secrete
A major part of the limbic system is the hypothala-
dopamine. Other neurons located in adjacent
mus, with its related structures. In addition to their
regions also secrete dopamine, but they send their
roles in behavioral control, these areas control many
endings into more ventral areas of the brain,
internal conditions of the body, such as body temper-
especially to the hypothalamus and the limbic
ature, osmolality of the body fluids, and the drives to
system. The dopamine is believed to act as an
eat and drink and to control body weight. These inter-
inhibitory transmitter in the basal ganglia, but
nal functions are collectively called vegetative func-
in some other areas of the brain it is possibly
tions of the brain, and their control is closely related
excitatory. Also, remember from Chapter 56 that
to behavior.
destruction of the dopaminergic neurons in the
substantia nigra is the basic cause of Parkinson’s
Functional Anatomy of the
disease.
Limbic System; Key Position
3.
The raphe nuclei and the serotonin system. In
the midline of the pons and medulla are several
of the Hypothalamus
thin nuclei called the raphe nuclei. Many of the
neurons in these nuclei secrete serotonin. They
Figure 58-4 shows the anatomical structures of the
send fibers into the diencephalon and a few fibers
limbic system, demonstrating that they are an inter-
to the cerebral cortex; still other fibers descend to
connected complex of basal brain elements. Located
the spinal cord. The serotonin secreted at the cord
in the middle of all these is the extremely small hypo-
fiber endings has the ability to suppress pain,
thalamus, which from a physiologic point of view is one
which was discussed in Chapter 48. The serotonin
of the central elements of the limbic system. Figure
released in the diencephalon and cerebrum almost
58-5 illustrates schematically this key position of the
certainly plays an essential inhibitory role to help
hypothalamus in the limbic system and shows sur-
cause normal sleep, as we discuss in Chapter 59.
rounding it other subcortical structures of the limbic
4.
The gigantocellular neurons of the reticular
system, including the septum, the paraolfactory area,
excitatory area and the acetylcholine system.
the anterior nucleus of the thalamus, portions of the
Earlier we discussed the gigantocellular neurons
basal ganglia, the hippocampus, and the amygdala.
(the giant cells) in the reticular excitatory area of
And surrounding the subcortical limbic areas is the
the pons and mesencephalon. The fibers from
limbic cortex, composed of a ring of cerebral cortex in
these large cells divide immediately into two
each side of the brain (1) beginning in the orbitofrontal
branches, one passing upward to the higher levels
area on the ventral surface of the frontal lobes, (2)
of the brain and the other passing downward
extending upward into the subcallosal gyrus, (3) then
through the reticulospinal tracts into the spinal
over the top of the corpus callosum onto the medial
cord. The neurohormone secreted at their
aspect of the cerebral hemisphere in the cingulate
terminals is acetylcholine. In most places,
gyrus, and finally (4) passing behind the corpus callo-
the acetylcholine functions as an excitatory
sum and downward onto the ventromedial surface of
neurotransmitter. Activation of these
the temporal lobe to the parahippocampal gyrus and
acetylcholine neurons leads to an acutely awake
uncus.
and excited nervous system.
Thus, on the medial and ventral surfaces of each
cerebral hemisphere is a ring of mostly paleocortex
Other Neurotransmitters and Neurohormonal Substances
that surrounds a group of deep structures intimately
Secreted in the Brain. Without describing their function,
associated with overall behavior and emotions. In turn,
the following is a list of still other neurohormonal
this ring of limbic cortex functions as a two-way com-
substances that function either at specific synapses or
munication and association linkage between the neo-
by release into the fluids of the brain: enkephalins,
cortex and the lower limbic structures.
gamma-aminobutyric acid, glutamate, vasopressin,
Many of the behavioral functions elicited from the
adrenocorticotropic hormone, epinephrine, histamine,
hypothalamus and other limbic structures are also
endorphins, angiotensin II, and neurotensin. Thus,
mediated through the reticular nuclei in the brain stem
there are multiple neurohormonal systems in the
and their associated nuclei. It was pointed out in
brain, the activation of each of which plays its own role
Chapter 55 as well as earlier in this chapter that stim-
in controlling a different quality of brain function.
ulation of the excitatory portion of this reticular for-
mation can cause high degrees of cerebral excitability
while also increasing the excitability of much of the
Limbic System
spinal cord synapses. In Chapter 60, we will see that
most of the hypothalamic signals for controlling the
The word “limbic” means “border.” Originally, the
autonomic nervous system are also transmitted
term “limbic” was used to describe the border struc-
through synaptic nuclei located in the brain stem.
tures around the basal regions of the cerebrum, but as
An important route of communication between
we have learned more about the functions of the
the limbic system and the brain stem is the medial
732
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Cingulate gyrus and cingulum
Stria medullaris
thalami
Indusium griseum
Body of fornix
and longitudinal striae
Dorsal fornix
Septum pellucidum
(supracommissural septum)
Anterior nuclear
Mamillothalamic
group of thalamus
tract
Anterior commissure
Mamillotegmental
tract
Subcallosal gyrus
Isthmus
Paraterminal gyrus
(precommissural
septum)
Gyrus
fasciolaris
Orbitofrontal cortex
Fimbria
Prehippocampal rudiment
of fornix
Paraolfactory area
Stria terminalis
Olfactory bulb
Connecting spinal cord
HYPOTHALAMUS
Column of fornix
Hippocampus
(postcommissural fornix)
Dentate gyrus
Uncus
Amygdaloid body
Parahippocampal gyrus
Mamillary body
Figure 58-4
Anatomy of the limbic system, shown in the dark pink area. (Redrawn from Warwick R, Williams PL: Gray’s Anatomy, 35th Br. ed. London:
Longman Group Ltd, 1973.)
short pathways among the reticular formation of the
brain stem, thalamus, hypothalamus, and most other
Cingulate gyrus
contiguous areas of the basal brain.
Portions
Anterior
Subcallosal
Hypothalamus, a Major
Septum
of basal
nuclei of
gyrus
area
Control Headquarters for the
ganglia
thalamus
Limbic System
Paraolfactory
Hypothalamus
Orbitofrontal
area
cortex
The hypothalamus, despite its very small size of only a
Hippocampus
few cubic centimeters, has two-way communicating
Uncus
Amygdala
pathways with all levels of the limbic system. In turn,
it and its closely allied structures send output signals
in three directions: (1) backward and downward to the
Parahippocampal gyrus
brain stem, mainly into the reticular areas of the mes-
encephalon, pons, and medulla and from these areas
into the peripheral nerves of the autonomic nervous
Figure 58-5
system; (2) upward toward many higher areas of the
diencephalon and cerebrum, especially to the anterior
Limbic system, showing the key position of the hypothalamus.
thalamus and limbic portions of the cerebral cortex;
and (3) into the hypothalamic infundibulum to control
or partially control most of the secretory functions of
forebrain bundle, which extends from the septal and
both the posterior and the anterior pituitary glands.
orbitofrontal regions of the cerebral cortex downward
Thus, the hypothalamus, which represents less than
through the middle of the hypothalamus to the brain
1 per cent of the brain mass, is one of the most impor-
stem reticular formation. This bundle carries fibers in
tant of the control pathways of the limbic system. It
both directions, forming a trunk line communication
controls most of the vegetative and endocrine func-
system. A second route of communication is through
tions of the body as well as many aspects of emotional
Chapter 58
Behavioral and Motivational Mechanisms of the Brain—The Limbic System and the Hypothalamus
733
POSTERIOR
ANTERIOR
Dorsomedial nucleus
Paraventricular nucleus
(GI stimulation)
(Oxytocin release)
(Water conservation)
Medial preoptic area
Posterior hypothalamus
(Bladder contraction)
(Increased blood pressure)
HYPOTHALAMUS
(Decreased heart rate)
(Pupillary dilation)
(Decreased blood pressure)
(Shivering)
Perifornical nucleus
Posterior preoptic and
(Hunger)
anterior hypothalamic areas
(Increased blood pressure)
(Body temperature regulation)
(Rage)
(Panting)
(Sweating)
Ventromedial nucleus
(Thyrotropin inhibition)
(Satiety)
(Neuroendocrine control)
Optic chiasm (Optic nerve)
Supraoptic nucleus
Mamillary body
(Vasopressin release)
(Feeding reflexes)
Arcuate nucleus and periventricular zone
Infundibulum
Figure 58-6
(Neuroendocrine control)
Control centers of the hypothala-
Lateral hypothalamic area (not shown)
mus (sagittal view).
(Thirst and hunger)
behavior. Let us discuss first the vegetative and
endocrine control functions and then return to the
Thalamus
behavioral functions of the hypothalamus to see how
Paraventricular
these operate together.
Periventricular
Dorsomedial
Anterior
Fornix
Vegetative and Endocrine Control
hypothalamic
Lateral
Functions of the Hypothalamus
hypothalamic
Optic
tract
Supraoptic
The different hypothalamic mechanisms for control-
ling multiple functions of the body are so important
that they are discussed in multiple chapters through-
Arcuate
Ventromedial
out this text. For instance, the role of the hypothala-
mus to help regulate arterial pressure is discussed in
Figure 58-7
Chapter 18, thirst and water conservation in Chapter
29, temperature regulation in Chapter
73, and
Coronal view of the hypothalamus, showing the mediolateral posi-
endocrine control in Chapter 75. To illustrate the
tions of the respective hypothalamic nuclei.
organization of the hypothalamus as a functional unit,
let us summarize the more important of its vegetative
activation of fiber tracts leading from or to control
and endocrine functions here as well.
nuclei located elsewhere. With this caution in mind, we
Figures 58-6 and 58-7 show enlarged sagittal and
can give the following general description of the veg-
coronal views of the hypothalamus, which represents
etative and control functions of the hypothalamus.
only a small area in Figure 58-4. Take a few minutes
to study these diagrams, especially to see in Figure
Cardiovascular Regulation. Stimulation of different areas
58-6 the multiple activities that are excited or inhib-
throughout the hypothalamus can cause every known
ited when respective hypothalamic nuclei are stimu-
type of neurogenic effect on the cardiovascular system,
lated. In addition to the centers shown in Figure 58-6,
including increased arterial pressure, decreased arterial
a large lateral hypothalamic area (shown in Figure
pressure, increased heart rate, and decreased heart rate.
58-7) is present on each side of the hypothalamus. The
In general, stimulation in the posterior and lateral hypo-
thalamus increases the arterial pressure and heart rate,
lateral areas are especially important in controlling
whereas stimulation in the preoptic area often has oppo-
thirst, hunger, and many of the emotional drives.
site effects, causing a decrease in both heart rate and
A word of caution must be issued for studying these
arterial pressure. These effects are transmitted mainly
diagrams because the areas that cause specific activi-
through specific cardiovascular control centers in the
ties are not nearly as accurately localized as suggested
reticular regions of the pons and medulla.
in the figures. Also, it is not known whether the effects
noted in the figures result from stimulation of specific
Regulation of Body Temperature. The anterior portion of the
control nuclei or whether they result merely from
hypothalamus, especially the preoptic area, is concerned
734
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
with regulation of body temperature. An increase in the
overactive, so that it has a voracious appetite, resulting
temperature of the blood flowing through this area
eventually in tremendous obesity. Another area of the
increases the activity of temperature-sensitive neurons,
hypothalamus that enters into overall control of gas-
while a decrease in temperature decreases their activ-
trointestinal activity is the mamillary bodies; these
ity. In turn, these neurons control mechanisms for
control at least partially the patterns of many feeding
increasing or decreasing body temperature, as discussed
reflexes, such as licking the lips and swallowing.
in Chapter 73.
Hypothalamic Control of Endocrine Hormone Secretion by the
Regulation of Body Water. The hypothalamus regulates
Anterior Pituitary Gland. Stimulation of certain areas of the
body water in two ways: (1) by creating the sensation of
hypothalamus also causes the anterior pituitary gland to
thirst, which makes the animal or person drink water,
secrete its endocrine hormones. This subject is discussed
and (2) by controlling the excretion of water into the
in detail in Chapter 74 in relation to neural control of
urine. An area called the thirst center is located in the
the endocrine glands. Briefly, the basic mechanisms are
lateral hypothalamus. When the fluid electrolytes in
the following.
either this center or closely allied areas become too
The anterior pituitary gland receives its blood supply
concentrated, the animal develops an intense desire to
mainly from blood that flows first through the lower
drink water; it will search out the nearest source of
part of the hypothalamus and then through the anterior
water and drink enough to return the electrolyte con-
pituitary vascular sinuses. As the blood courses through
centration of the thirst center to normal.
the hypothalamus before reaching the anterior pitu-
Control of renal excretion of water is vested mainly
itary, specific releasing and inhibitory hormones are
in the supraoptic nuclei. When the body fluids become
secreted into the blood by various hypothalamic nuclei.
too concentrated, the neurons of these areas become
These hormones are then transported via the blood
stimulated. Nerve fibers from these neurons project
to the anterior pituitary gland, where they act on the
downward through the infundibulum of the hypothala-
glandular cells to control release of specific anterior
mus into the posterior pituitary gland, where the nerve
pituitary hormones.
endings secrete the hormone antidiuretic hormone (also
called vasopressin). This hormone is then absorbed into
Summary. A number of areas of the hypothalamus
the blood and transported to the kidneys where it acts
control specific vegetative and endocrine functions.
on the collecting ducts of the kidneys to cause increased
These areas are still poorly delimited, so much so that
reabsorption of water. This decreases loss of water into
the specification given above of different areas for dif-
the urine but allows continuing excretion of electrolytes,
ferent hypothalamic functions is partially tentative.
thus decreasing the concentration of the body fluids
back toward normal. These functions are presented in
Chapter 28.
Behavioral Functions of the
Regulation of Uterine Contractility and of Milk Ejection from the
Hypothalamus and Associated
Breasts. Stimulation of the paraventricular nuclei causes
Limbic Structures
their neuronal cells to secrete the hormone oxytocin.
This in turn causes increased contractility of the uterus
Effects Caused by Stimulation. In addition to the vegeta-
as well as contraction of the myoepithelial cells sur-
tive and endocrine functions of the hypothalamus,
rounding the alveoli of the breasts, which then causes
stimulation of or lesions in the hypothalamus often
the alveoli to empty their milk through the nipples.
have profound effects on emotional behavior of
At the end of pregnancy, especially large quantities of
oxytocin are secreted, and this secretion helps to
animals and human beings.
promote labor contractions that expel the baby. Then,
In animals, some of the behavioral effects of stimu-
whenever the baby suckles the mother’s breast, a reflex
lation are the following:
signal from the nipple to the posterior hypothalamus
1. Stimulation in the lateral hypothalamus not only
also causes oxytocin release, and the oxytocin now per-
causes thirst and eating, as discussed above, but
forms the necessary function of contracting the ductules
also increases the general level of activity of the
of the breast, thereby expelling milk through the nipples
animal, sometimes leading to overt rage and
so that the baby can nourish itself. These functions are
fighting, as discussed subsequently.
discussed in Chapter 82.
2. Stimulation in the ventromedial nucleus
and surrounding areas mainly causes effects
Gastrointestinal and Feeding Regulation. Stimulation of
several areas of the hypothalamus causes an animal to
opposite to those caused by lateral hypothalamic
experience extreme hunger, a voracious appetite, and an
stimulation—that is, a sense of satiety, decreased
intense desire to search for food. One area associated
eating, and tranquility.
with hunger is the lateral hypothalamic area. Con-
3. Stimulation of a thin zone of periventricular
versely, damage to this area on both sides of the hypo-
nuclei, located immediately adjacent to the third
thalamus causes the animal to lose desire for food,
ventricle (or also stimulation of the central gray
sometimes causing lethal starvation.
area of the mesencephalon that is continuous with
A center that opposes the desire for food, called the
this portion of the hypothalamus), usually leads to
satiety center, is located in the ventromedial nuclei.
fear and punishment reactions.
When this center is stimulated electrically, an animal
4. Sexual drive can be stimulated from several
that is eating food suddenly stops eating and shows
complete indifference to food. However, if this area is
areas of the hypothalamus, especially the most
destroyed bilaterally, the animal cannot be satiated;
anterior and most posterior portions of the
instead, its hypothalamic hunger centers become
hypothalamus.
Chapter 58
Behavioral and Motivational Mechanisms of the Brain—The Limbic System and the Hypothalamus
735
Effects Caused by Hypothalamic Lesions. Lesions in the
hypothalamus, in general, cause effects opposite to
those caused by stimulation. For instance:
1. Bilateral lesions in the lateral hypothalamus will
decrease drinking and eating almost to zero, often
leading to lethal starvation. These lesions cause
extreme passivity of the animal as well, with loss
of most of its overt drives.
2. Bilateral lesions of the ventromedial areas of the
hypothalamus cause effects that are mainly
opposite to those caused by lesions of the lateral
hypothalamus: excessive drinking and eating as
well as hyperactivity and often continuous
savagery along with frequent bouts of extreme
rage on the slightest provocation.
Stimulation or lesions in other regions of the limbic
system, especially in the amygdala, the septal area,
and areas in the mesencephalon, often cause effects
similar to those elicited from the hypothalamus. We
will discuss some of these in more detail later.
Figure 58-8
“Reward” and “Punishment” Function
Technique for localizing reward and punishment centers in the
of the Limbic System
brain of a monkey.
From the discussion thus far, it is already clear that
several limbic structures are particularly concerned
stimuli giving a sense of reward and stronger ones a
with the affective nature of sensory sensations—that is,
sense of punishment. Less potent reward centers,
whether the sensations are pleasant or unpleasant.
which are perhaps secondary to the major ones in the
These affective qualities are also called reward or pun-
hypothalamus, are found in the septum, the amygdala,
ishment, or satisfaction or aversion. Electrical stimula-
certain areas of the thalamus and basal ganglia, and
tion of certain limbic areas pleases or satisfies the
extending downward into the basal tegmentum of the
animal, whereas electrical stimulation of other regions
mesencephalon.
causes terror, pain, fear, defense, escape reactions,
and all the other elements of punishment. The degrees
Punishment Centers
of stimulation of these two oppositely responding
The apparatus shown in Figure 58-8 can also be
systems greatly affect the behavior of the animal.
connected so that the stimulus to the brain continues
all the time except when the lever is pressed. In this
Reward Centers
case, the animal will not press the lever to turn the
Figure 58-8 shows a technique that has been used for
stimulus off when the electrode is in one of the reward
localizing specific reward and punishment areas of the
areas; but when it is in certain other areas, the animal
brain. In this figure, a lever is placed at the side of
immediately learns to turn it off. Stimulation in these
the cage and is arranged so that depressing the lever
areas causes the animal to show all the signs of
makes electrical contact with a stimulator. Electrodes
displeasure, fear, terror, pain, punishment, and even
are placed successively at different areas in the brain
sickness.
so that the animal can stimulate the area by pressing
By means of this technique, the most potent areas
the lever. If stimulating the particular area gives the
for punishment and escape tendencies have been
animal a sense of reward, then it will press the lever
found in the central gray area surrounding the aque-
again and again, sometimes as much as hundreds or
duct of Sylvius in the mesencephalon and extending
even thousands of times per hour. Furthermore, when
upward into the periventricular zones of the hypo-
offered the choice of eating some delectable food as
thalamus and thalamus. Less potent punishment areas
opposed to the opportunity to stimulate the reward
are found in some locations in the amygdala and hip-
center, the animal often chooses the electrical
pocampus. It is particularly interesting that stimulation
stimulation.
in the punishment centers can frequently inhibit the
By using this procedure, the major reward centers
reward and pleasure centers completely, demonstrat-
have been found to be located along the course of the
ing that punishment and fear can take precedence over
medial forebrain bundle, especially in the lateral and
pleasure and reward.
ventromedial nuclei of the hypothalamus. It is strange
that the lateral nucleus should be included among the
Rage—Its Association with
reward areas—indeed, it is one of the most potent of
Punishment Centers
all—because even stronger stimuli in this area can
An emotional pattern that involves the punish-
cause rage. But this is true in many areas, with weaker
ment centers of the hypothalamus and other limbic
736
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
structures, and has also been well characterized, is the
If the stimulus does cause either reward or punish-
rage pattern, described as follows.
ment rather than indifference, the cerebral cortical
Strong stimulation of the punishment centers of
response becomes progressively more and more
the brain, especially in the periventricular zone of the
intense during repeated stimulation instead of fading
hypothalamus and in the lateral hypothalamus, causes
away, and the response is said to be reinforced. An
the animal to (1) develop a defense posture, (2) extend
animal builds up strong memory traces for sensations
its claws, (3) lift its tail, (4) hiss, (5) spit, (6) growl, and
that are either rewarding or punishing but, conversely,
(7) develop piloerection, wide-open eyes, and dilated
develops complete habituation to indifferent sensory
pupils. Furthermore, even the slightest provocation
stimuli.
causes an immediate savage attack. This is approxi-
It is evident that the reward and punishment centers
mately the behavior that one would expect from an
of the limbic system have much to do with selecting
animal being severely punished, and it is a pattern of
the information that we learn, usually throwing away
behavior that is called rage.
more than 99 per cent of it and selecting less than 1
Fortunately, in the normal animal, the rage phe-
per cent for retention.
nomenon is held in check mainly by inhibitory signals
from the ventromedial nuclei of the hypothalamus. In
addition, portions of the hippocampi and anterior
Specific Functions of Other
limbic cortex, especially in the anterior cingulate
gyri and subcallosal gyri, help suppress the rage
Parts of the Limbic System
phenomenon.
Functions of the Hippocampus
Placidity and Tameness. Exactly the opposite emotional
The hippocampus is the elongated portion of the cere-
behavior patterns occur when the reward centers are
bral cortex that folds inward to form the ventral
stimulated: placidity and tameness.
surface of much of the inside of the lateral ventricle.
One end of the hippocampus abuts the amygdaloid
nuclei, and along its lateral border it fuses with the
Importance of Reward or Punishment
parahippocampal gyrus, which is the cerebral cortex
in Behavior
on the ventromedial outside surface of the temporal
lobe.
Almost everything that we do is related in some way
The hippocampus (and its adjacent temporal and
to reward and punishment. If we are doing something
parietal lobe structures, all together called the hip-
that is rewarding, we continue to do it; if it is punish-
pocampal formation) has numerous but mainly indi-
ing, we cease to do it. Therefore, the reward and pun-
rect connections with many portions of the cerebral
ishment centers undoubtedly constitute one of the
cortex as well as with the basal structures of the limbic
most important of all the controllers of our bodily
system—the amygdala, the hypothalamus, the septum,
activities, our drives, our aversions, our motivations.
and the mamillary bodies. Almost any type of sensory
experience causes activation of at least some part of
Effect of Tranquilizers on the Reward or Punishment Centers.
the hippocampus, and the hippocampus in turn dis-
Administration of a tranquilizer, such as chlorpro-
tributes many outgoing signals to the anterior thala-
mazine, usually inhibits both the reward and the pun-
mus, hypothalamus, and other parts of the limbic
ishment centers, thereby decreasing the affective
system, especially through the fornix, a major commu-
reactivity of the animal. Therefore, it is presumed that
nicating pathway. Thus, the hippocampus is an addi-
tranquilizers function in psychotic states by suppress-
tional channel through which incoming sensory signals
ing many of the important behavioral areas of the
can initiate behavioral reactions for different pur-
hypothalamus and its associated regions of the limbic
poses. As in other limbic structures, stimulation of dif-
brain.
ferent areas in the hippocampus can cause almost any
of the different behavioral patterns such as pleasure,
Importance of Reward or Punishment
rage, passivity, or excess sex drive.
in Learning and Memory—Habituation
Another feature of the hippocampus is that it can
Versus Reinforcement
become hyperexcitable. For instance, weak electrical
Animal experiments have shown that a sensory expe-
stimuli can cause focal epileptic seizures in small areas
rience that causes neither reward nor punishment is
of the hippocampi. These often persist for many
hardly remembered at all. Electrical recordings from
seconds after the stimulation is over, suggesting that
the brain show that a newly experienced sensory stim-
the hippocampi can perhaps give off prolonged output
ulus almost always excites multiple areas in the cere-
signals even under normal functioning conditions.
bral cortex. But, if the sensory experience does not
During hippocampal seizures, the person experiences
elicit a sense of either reward or punishment, repeti-
various psychomotor effects, including olfactory,
tion of the stimulus over and over leads to almost com-
visual, auditory, tactile, and other types of hallucina-
plete extinction of the cerebral cortical response. That
tions that cannot be suppressed as long as the seizure
is, the animal becomes habituated to that specific
persists even though the person has not lost con-
sensory stimulus and thereafter ignores it.
sciousness and knows these hallucinations to be
Chapter 58
Behavioral and Motivational Mechanisms of the Brain—The Limbic System and the Hypothalamus
737
unreal. Probably one of the reasons for this hyper-
In lower animals, the amygdala is concerned to a great
excitability of the hippocampi is that they have a dif-
extent with olfactory stimuli and their interrelations
ferent type of cortex from that elsewhere in the
with the limbic brain. Indeed, it is pointed out in
Chapter 53 that one of the major divisions of the olfac-
cerebrum, having only three nerve cell layers in some
tory tract terminates in a portion of the amygdala
of its areas instead of the six layers found elsewhere.
called the corticomedial nuclei, which lies immediately
beneath the cerebral cortex in the olfactory pyriform
Role of the Hippocampus in Learning
area of the temporal lobe. In the human being, another
Effect of Bilateral Removal of the Hippocampi—Inability to
portion of the amygdala, the basolateral nuclei, has
Learn. Portions of the hippocampi have been surgically
become much more highly developed than the olfactory
removed bilaterally in a few human beings for treat-
portion and plays important roles in many behavioral
ment of epilepsy. These people can recall most previ-
activities not generally associated with olfactory stimuli.
ously learned memories satisfactorily. However, they
The amygdala receives neuronal signals from all por-
often can learn essentially no new information that is
tions of the limbic cortex, as well as from the neocortex
of the temporal, parietal, and occipital lobes—especially
based on verbal symbolism. In fact, they often cannot
from the auditory and visual association areas. Because
even learn the names of people with whom they come
of these multiple connections, the amygdala has been
in contact every day. Yet they can remember for a
called the “window” through which the limbic system
moment or so what transpires during the course of
sees the place of the person in the world. In turn, the
their activities. Thus, they are capable of short-term
amygdala transmits signals (1) back into these same cor-
memory for seconds up to a minute or two, although
tical areas,
(2) into the hippocampus, (3) into the
their ability to establish memories lasting longer than
septum, (4) into the thalamus, and (5) especially into the
a few minutes is either completely or almost com-
hypothalamus.
pletely abolished. This is the phenomenon called
anterograde amnesia that was discussed in Chapter 57.
Effects of Stimulating the Amygdala. In general, stimulation
in the amygdala can cause almost all the same effects as
those elicited by direct stimulation of the hypothalamus,
Theoretical Function of the Hippocampus in Learning. The
plus other effects. Effects initiated from the amygdala
hippocampus originated as part of the olfactory
and then sent through the hypothalamus include (1)
cortex. In many lower animals, this cortex plays essen-
increases or decreases in arterial pressure, (2) increases
tial roles in determining whether the animal will eat
or decreases in heart rate, (3) increases or decreases in
a particular food, whether the smell of a particular
gastrointestinal motility and secretion, (4) defecation or
object suggests danger, or whether the odor is sexually
micturition, (5) pupillary dilation or, rarely, constriction,
inviting, thus making decisions that are of life-or-death
(6) piloerection, and (7) secretion of various anterior
importance. Very early in evolutionary development of
pituitary hormones, especially the gonadotropins and
the brain, the hippocampus presumably became a crit-
adrenocorticotropic hormone.
ical decision-making neuronal mechanism, determin-
Aside from these effects mediated through the hypo-
thalamus, amygdala stimulation can also cause several
ing the importance of the incoming sensory signals.
types of involuntary movement. These include (1) tonic
Once this critical decision-making capability had been
movements, such as raising the head or bending the
established, presumably the remainder of the brain
body; (2) circling movements; (3) occasionally clonic,
also began to call on the hippocampus for decision
rhythmical movements; and (4) different types of move-
making. Therefore, if the hippocampus signals that a
ments associated with olfaction and eating, such as
neuronal input is important, the information is likely
licking, chewing, and swallowing.
to be committed to memory.
In addition, stimulation of certain amygdaloid nuclei
Thus, a person rapidly becomes habituated to indif-
can cause a pattern of rage, escape, punishment, severe
ferent stimuli but learns assiduously any sensory expe-
pain, and fear similar to the rage pattern elicited from
rience that causes either pleasure or pain. But what is
the hypothalamus, as described earlier. Stimulation of
other amygdaloid nuclei can give reactions of reward
the mechanism by which this occurs? It has been sug-
and pleasure.
gested that the hippocampus provides the drive that
Finally, excitation of still other portions of the amyg-
causes translation of short-term memory into long-
dala can cause sexual activities that include erection,
term memory—that is, the hippocampus transmits
copulatory movements, ejaculation, ovulation, uterine
some signal or signals that seem to make the mind
activity, and premature labor.
rehearse over and over the new information until per-
manent storage takes place. Whatever the mechanism,
Effects of Bilateral Ablation of the Amygdala—The Klüver-Bucy
without the hippocampi, consolidation of long-term
Syndrome. When the anterior parts of both temporal
memories of the verbal or symbolic thinking type is
lobes are destroyed in a monkey, this removes not only
poor or does not take place.
portions of temporal cortex but also of the amygdalas
that lie inside these parts of the temporal lobes. This
causes changes in behavior called the Klüver-Bucy syn-
Functions of the Amygdala
drome, which is demonstrated by an animal that (1) is
not afraid of anything, (2) has extreme curiosity about
The amygdala is a complex of multiple small nuclei
everything, (3) forgets rapidly, (4) has a tendency to
located immediately beneath the cerebral cortex of the
place everything in its mouth and sometimes even tries
medial anterior pole of each temporal lobe. It has abun-
to eat solid objects, and (5) often has a sex drive so
dant bidirectional connections with the hypothalamus
strong that it attempts to copulate with immature
as well as with other areas of the limbic system.
animals, animals of the wrong sex, or even animals of a
738
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
different species. Although similar lesions in human
and olfactory behavioral associations. In the parahip-
beings are rare, afflicted people respond in a manner not
pocampal gyri, there is a tendency for complex auditory
too different from that of the monkey.
associations as well as complex thought associations
derived from Wernicke’s area of the posterior temporal
Overall Function of the Amygdalas. The amygdalas seem to
lobe. In the middle and posterior cingulate cortex, there
be behavioral awareness areas that operate at a semi-
is reason to believe that sensorimotor behavioral asso-
conscious level. They also seem to project into the
ciations occur.
limbic system one’s current status in relation to both
surroundings and thoughts. On the basis of this infor-
mation, the amygdala is believed to make the person’s
References
behavioral response appropriate for each occasion.
Adell A, Celada P, Abellan MT, Artigas F: Origin and func-
Function of the Limbic Cortex
tional role of the extracellular serotonin in the midbrain
raphe nuclei. Brain Res Brain Res Rev 39:154, 2002.
The most poorly understood portion of the limbic
Bechara A, Damasio H, Damasio AR: Role of the amygdala
system is the ring of cerebral cortex called the limbic
in decision-making. Ann N Y Acad Sci 985:356, 2003.
cortex that surrounds the subcortical limbic structures.
Blank T, Nijholt I, Spiess J: Molecular determinants mediat-
This cortex functions as a transitional zone through
ing effects of acute stress on hippocampus-dependent
which signals are transmitted from the remainder of the
synaptic plasticity and learning. Mol Neurobiol 29:131,
brain cortex into the limbic system and also in the oppo-
2004.
site direction. Therefore, the limbic cortex in effect
Bouret SG, Simerly RB: Leptin and development of hypo-
functions as a cerebral association area for control of
thalamic feeding circuits. Endocrinology 145:2621, 2004.
behavior.
Conlon R, Hobson JA: Understanding the Human Mind.
Stimulation of the different regions of the limbic
New York: John Wiley, 1999.
cortex has failed to give any real idea of their functions.
Denton DA, McKinley MJ, Weisinger RS: Hypothalamic
However, as is true of so many other portions of the
integration of body fluid regulation. Proc Natl Acad Sci
limbic system, essentially all behavioral patterns can be
U S A 93:7397, 1996.
elicited by stimulation of specific portions of the limbic
Drevets WC: Neuroimaging abnormalities in the amygdala
cortex. Likewise, ablation of some limbic cortical areas
in mood disorders. Ann N Y Acad Sci 985:420, 2003
can cause persistent changes in an animal’s behavior, as
Gerashchenko D, Shiromani PJ: Different neuronal pheno-
follows.
types in the lateral hypothalamus and their role in sleep
and wakefulness. Mol Neurobiol 29:41, 2004.
Ablation of the Anterior Temporal Cortex. When the anterior
Guillery RW: Branching thalamic afferents link action and
temporal cortex is ablated bilaterally, the amygdalas are
perception. J Neurophysiol 90:539, 2003.
almost invariably damaged as well. This was discussed
Haines DE: Fundamental Neuroscience. New York:
earlier in this chapter; it was pointed out that the
Churchill Livingstone, 1997.
Klüver-Bucy syndrome occurs. The animal especially
Holland PC, Gallagher M: Amygdala—frontal interactions
develops consummatory behavior: it investigates any
and reward expectancy. Curr Opin Neurobiol 14:148, 2004.
and all objects, has intense sex drives toward inappro-
Joels M, Verkuyl JM, Van Riel E: Hippocampal and hypo-
priate animals or even inanimate objects, and loses all
thalamic function after chronic stress. Ann N Y Acad Sci
fear—and thus develops tameness as well.
1007:367, 2003.
Kandel ER, Schwartz JH, Jessell TM: Principles of Neural
Ablation of the Posterior Orbital Frontal Cortex. Bilateral
Science, 4th ed. New York: McGraw-Hill, 2000.
removal of the posterior portion of the orbital frontal
Kelley AE: Ventral striatal control of appetitive motivation:
cortex often causes an animal to develop insomnia asso-
role in ingestive behavior and reward-related learning.
ciated with intense motor restlessness, becoming unable
Neurosci Biobehav Rev 27:765, 2004.
to sit still and moving about continuously.
LeDoux JE: Emotion circuits in the brain. Annu Rev Neu-
rosci 23:155, 2000.
Ablation of the Anterior Cingulate Gyri and Subcallosal Gyri. The
Lumb BM: Hypothalamic and midbrain circuitry that dis-
anterior cingulate gyri and the subcallosal gyri are the
tinguishes between escapable and inescapable pain. News
portions of the limbic cortex that communicate between
Physiol Sci 19:22, 2004.
the prefrontal cerebral cortex and the subcortical limbic
Morris JF, Ludwig M: Magnocellular dendrites: prototypic
structures. Destruction of these gyri bilaterally releases
receiver/transmitters. J Neuroendocrinol 16:403, 2004.
the rage centers of the septum and hypothalamus from
Phelps EA: Human emotion and memory: interactions of the
prefrontal inhibitory influence. Therefore, the animal
amygdala and hippocampal complex. Curr Opin Neuro-
can become vicious and much more subject to fits of
biol 14:198, 2004.
rage than normally.
Sah P, Faber ES, Lopez De Armentia M, Power J: The amyg-
daloid complex: anatomy and physiology. Physiol Rev
Summary. Until further information is available, it is
83:803, 2003.
perhaps best to state that the cortical regions of the
van den Pol AN: Weighing the role of hypothalamic feeding
limbic system occupy intermediate associative positions
neurotransmitters. Neuron 40:1059, 2003.
between the functions of the specific areas of the cere-
Vann SD, Aggleton JP: The mammillary bodies: two memory
bral cortex and functions of the subcortical limbic struc-
systems in one? Nat Rev Neurosci 5:35, 2004.
tures for control of behavioral patterns. Thus, in the
Wild B, Rodden FA, Grodd W, Ruch W: Neural correlates of
anterior temporal cortex, one especially finds gustatory
laughter and humour. Brain 126:2121, 2003.
C H A P T E R
5
9
States of Brain Activity—Sleep,
Brain Waves, Epilepsy, Psychoses
All of us are aware of the many different states
of brain activity, including sleep, wakefulness,
extreme excitement, and even different levels of
mood such as exhilaration, depression, and fear. All
these states result from different activating
or inhibiting forces generated usually within the
brain itself. In Chapter 58, we began a partial dis-
cussion of this subject when we described different
systems that are capable of activating large portions of the brain. In this chapter,
we present brief surveys of specific states of brain activity, beginning with
sleep.
Sleep
Sleep is defined as unconsciousness from which the person can be aroused by
sensory or other stimuli. It is to be distinguished from coma, which is uncon-
sciousness from which the person cannot be aroused. There are multiple stages
of sleep, from very light sleep to very deep sleep; sleep researchers also divide
sleep into two entirely different types of sleep that have different qualities, as
follows.
Two Types of Sleep. During each night, a person goes through stages of two types
of sleep that alternate with each other. They are called (1) slow-wave sleep,
because in this type of sleep the brain waves are very strong and very low fre-
quency, as we discuss later, and (2) rapid eye movement sleep (REM sleep),
because in this type of sleep the eyes undergo rapid movements despite the fact
that the person is still asleep.
Most sleep during each night is of the slow-wave variety; this is the deep,
restful sleep that the person experiences during the first hour of sleep after
having been awake for many hours. REM sleep, on the other hand, occurs in
episodes that occupy about 25 per cent of the sleep time in young adults; each
episode normally recurs about every 90 minutes. This type of sleep is not so
restful, and it is usually associated with vivid dreaming.
Slow-Wave Sleep
Most of us can understand the characteristics of deep slow-wave sleep by
remembering the last time we were kept awake for more than 24 hours and
then the deep sleep that occurred during the first hour after going to sleep. This
sleep is exceedingly restful and is associated with decrease in both peripheral
vascular tone and many other vegetative functions of the body. For instance,
there are 10 to 30 per cent decreases in blood pressure, respiratory rate, and
basal metabolic rate.
Although slow-wave sleep is frequently called “dreamless sleep,” dreams and
sometimes even nightmares do occur during slow-wave sleep. The difference
between the dreams that occur in slow-wave sleep and those that occur in REM
sleep is that those of REM sleep are associated with more bodily muscle
activity, and the dreams of slow-wave sleep usually are not remembered. That
739
740
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
is, during slow-wave sleep, consolidation of the dreams
Neuronal Centers, Neurohumoral Substances,
in memory does not occur.
and Mechanisms That Can Cause Sleep—
A Possible Specific Role for Serotonin
Stimulation of several specific areas of the brain
REM Sleep (Paradoxical Sleep,
can produce sleep with characteristics near those
Desynchronized Sleep)
of natural sleep. Some of these areas are the
following:
In a normal night of sleep, bouts of REM sleep lasting
1.
The most conspicuous stimulation area for causing
5 to 30 minutes usually appear on the average every
almost natural sleep is the raphe nuclei in the
90 minutes. When the person is extremely sleepy, each
lower half of the pons and in the medulla. These
bout of REM sleep is short, and it may even be absent.
nuclei are a thin sheet of special neurons located
Conversely, as the person becomes more rested
in the midline. Nerve fibers from these nuclei
through the night, the durations of the REM bouts
spread locally in the brain stem reticular
increase.
formation and also upward into the thalamus,
There are several important characteristics of REM
hypothalamus, most areas of the limbic system,
sleep:
and even the neocortex of the cerebrum. In
1. It is usually associated with active dreaming and
addition, fibers extend downward into the spinal
active bodily muscle movements.
cord, terminating in the posterior horns where
2. The person is even more difficult to arouse by
they can inhibit incoming sensory signals,
sensory stimuli than during deep slow-wave sleep,
including pain, as discussed in Chapter 48. It is
and yet people usually awaken spontaneously in
also known that many nerve endings of fibers
the morning during an episode of REM sleep.
from these raphe neurons secrete serotonin.
3. Muscle tone throughout the body is exceedingly
When a drug that blocks the formation of
depressed, indicating strong inhibition of the
serotonin is administered to an animal, the animal
spinal muscle control areas.
often cannot sleep for the next several days.
4. Heart rate and respiratory rate usually become
Therefore, it has been assumed that serotonin is a
irregular, which is characteristic of the dream
transmitter substance associated with production
state.
of sleep.
5. Despite the extreme inhibition of the peripheral
2.
Stimulation of some areas in the nucleus of the
muscles, irregular muscle movements do occur.
tractus solitarius can also cause sleep. This nucleus
These are in addition to the rapid movements of
is the termination in the medulla and pons for
the eyes.
visceral sensory signals entering by way of the
6. The brain is highly active in REM sleep, and
vagus and glossopharyngeal nerves.
overall brain metabolism may be increased as
3.
Stimulation of several regions in the diencephalon
much as 20 per cent. The electroencephalogram
can also promote sleep, including (1) the rostral
(EEG) shows a pattern of brain waves similar to
part of the hypothalamus, mainly in the
those that occur during wakefulness. This type of
suprachiasmal area, and (2) an occasional area in
sleep is also called paradoxical sleep because it is
the diffuse nuclei of the thalamus.
a paradox that a person can still be asleep despite
marked activity in the brain.
Lesions in Sleep-Promoting Centers Can Cause Intense Wake-
In summary, REM sleep is a type of sleep in which
fulness. Discrete lesions in the raphe nuclei lead to a
the brain is quite active. However, the brain activity is
high state of wakefulness. This is also true of bilateral
not channeled in the proper direction for the person
lesions in the medial rostral suprachiasmal area in the
to be fully aware of his or her surroundings, and there-
anterior hypothalamus. In both instances, the excita-
fore the person is truly asleep.
tory reticular nuclei of the mesencephalon and upper
pons seem to become released from inhibition, thus
causing the intense wakefulness. Indeed, sometimes
Basic Theories of Sleep
lesions of the anterior hypothalamus can cause such
intense wakefulness that the animal actually dies of
Sleep Is Believed to Be Caused by an Active Inhibitory Process.
exhaustion.
An earlier theory of sleep was that the excitatory areas
of the upper brain stem, the reticular activating system,
simply fatigued during the waking day and became
Other Possible Transmitter Substances Related to Sleep.
inactive as a result. This was called the passive theory
Experiments have shown that the cerebrospinal fluid
of sleep. An important experiment changed this view
as well as the blood or urine of animals that have been
to the current belief that sleep is caused by an active
kept awake for several days contains a substance or
inhibitory process: it was discovered that transecting
substances that will cause sleep when injected into the
the brain stem at the level of the midpons creates a
brain ventricular system of another animal. One likely
brain whose cortex never goes to sleep. In other words,
substance has been identified as muramyl peptide, a
there seems to be some center located below the mid-
low-molecular-weight substance that accumulates in
pontile level of the brain stem that is required to cause
the cerebrospinal fluid and urine in animals kept
sleep by inhibiting other parts of the brain.
awake for several days. When only micrograms of this
Chapter 59
States of Brain Activity—Sleep, Brain Waves, Epilepsy, Psychoses
741
sleep-producing substance are injected into the third
Physiologic Effects of Sleep
ventricle, almost natural sleep occurs within a few
minutes, and the animal may stay asleep for several
Sleep causes two major types of physiologic effects:
hours. Another substance that has similar effects in
first, effects on the nervous system itself, and second,
causing sleep is a nonapeptide isolated from the blood
effects on other functional systems of the body. The
of sleeping animals. And still a third sleep factor, not
nervous system effects seem to be by far the more
yet identified molecularly, has been isolated from the
important because any person who has a transected
neuronal tissues of the brain stem of animals kept
spinal cord in the neck (and therefore has no sleep-
awake for days. It is possible that prolonged wakeful-
wakefulness cycle below the transection) shows no
ness causes progressive accumulation of a sleep factor
harmful effects in the body beneath the level of
or factors in the brain stem or in the cerebrospinal
transection that can be attributed directly to a sleep-
fluid that lead to sleep.
wakefulness cycle.
Lack of sleep certainly does, however, affect the
functions of the central nervous system. Prolonged
Possible Cause of REM Sleep. Why slow-wave sleep is
wakefulness is often associated with progressive mal-
broken periodically by REM sleep is not understood.
function of the thought processes and sometimes even
However, drugs that mimic the action of acetylcholine
causes abnormal behavioral activities.
increase the occurrence of REM sleep. Therefore, it
We are all familiar with the increased sluggishness
has been postulated that the large acetylcholine-
of thought that occurs toward the end of a prolonged
secreting neurons in the upper brain stem reticular for-
wakeful period, but in addition, a person can become
mation might, through their extensive efferent fibers,
irritable or even psychotic after forced wakefulness.
activate many portions of the brain. This theoretically
Therefore, we can assume that sleep in multiple ways
could cause the excess activity that occurs in certain
restores both normal levels of brain activity and
brain regions in REM sleep, even though the signals
normal “balance” among the different functions of the
are not channeled appropriately in the brain to cause
central nervous system. This might be likened to the
normal conscious awareness that is characteristic of
“rezeroing” of electronic analog computers after pro-
wakefulness.
longed use, because computers of this type gradually
lose their “baseline” of operation; it is reasonable to
Cycle Between Sleep and Wakefulness
assume that the same effect occurs in the central
The preceding discussions have merely identified
nervous system because overuse of some brain areas
neuronal areas, transmitters, and mechanisms that are
during wakefulness could easily throw these areas out
related to sleep. They have not explained the cyclical,
of balance with the remainder of the nervous system.
reciprocal operation of the sleep-wakefulness cycle.
We might postulate that the principal value of sleep
There is as yet no explanation. Therefore, we can let
is to restore natural balances among the neuronal
our imaginations run wild and suggest the following
centers. The specific physiologic functions of sleep
possible mechanism for causing the sleep-wakefulness
remain a mystery, and they are the subject of much
cycle.
research.
When the sleep centers are not activated, the mes-
encephalic and upper pontile reticular activating
nuclei are released from inhibition, which allows the
Brain Waves
reticular activating nuclei to become spontaneously
active. This in turn excites both the cerebral cortex and
Electrical recordings from the surface of the brain or
the peripheral nervous system, both of which send
even from the outer surface of the head demonstrate
numerous positive feedback signals back to the same
that there is continuous electrical activity in the brain.
Both the intensity and the patterns of this electrical
reticular activating nuclei to activate them still further.
activity are determined by the level of excitation of dif-
Therefore, once wakefulness begins, it has a natural
ferent parts of the brain resulting from sleep, wakeful-
tendency to sustain itself because of all this positive
ness, or brain diseases such as epilepsy or even
feedback activity.
psychoses. The undulations in the recorded electrical
Then, after the brain remains activated for many
potentials, shown in Figure 59-1, are called brain waves,
hours, even the neurons themselves in the activating
and the entire record is called an EEG (electroen-
system presumably become fatigued. Consequently,
cephalogram).
the positive feedback cycle between the mesen-
The intensities of brain waves recorded from the
cephalic reticular nuclei and the cerebral cortex fades,
surface of the scalp range from 0 to 200 microvolts, and
and the sleep-promoting effects of the sleep centers
their frequencies range from once every few seconds to
50 or more per second. The character of the waves is
take over, leading to rapid transition from wakefulness
dependent on the degree of activity in respective parts
back to sleep.
of the cerebral cortex, and the waves change markedly
This overall theory could explain the rapid transi-
between the states of wakefulness and sleep and coma.
tions from sleep to wakefulness and from wakefulness
Much of the time, the brain waves are irregular, and
to sleep. It could also explain arousal, the insomnia
no specific pattern can be discerned in the EEG. At
that occurs when a person’s mind becomes preoccu-
other times, distinct patterns do appear, some of which
pied with a thought, and the wakefulness that is pro-
are characteristic of specific abnormalities of the brain
duced by bodily physical activity.
such as epilepsy, which is discussed later.
742
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Alpha
Eyes open
Eyes closed
Beta
Figure 59-2
Theta
Replacement of the alpha rhythm by an asynchronous, low-
voltage beta rhythm when the eyes are opened.
50 mV
Delta
1 sec
Origin of Brain Waves
The discharge of a single neuron or single nerve fiber
in the brain can never be recorded from the surface of
Figure 59-1
the head. Instead, many thousands or even millions of
neurons or fibers must fire synchronously; only then will
Different types of brain waves in the normal electro-
the potentials from the individual neurons or fibers
encephalogram.
summate enough to be recorded all the way through the
skull. Thus, the intensity of the brain waves from the
scalp is determined mainly by the numbers of neurons
In normal healthy people, most waves in the EEG can
and fibers that fire in synchrony with one another, not
be classified as alpha, beta, theta, and delta waves, which
by the total level of electrical activity in the brain. In
are shown in Figure 59-1.
fact, strong nonsynchronous nerve signals often nullify
Alpha waves are rhythmical waves that occur at fre-
one another in the recorded brain waves because of
quencies between 8 and 13 cycles per second and are
opposing polarities. This is demonstrated in Figure 59-2,
found in the EEGs of almost all normal adult people
which shows, when the eyes were closed, synchronous
when they are awake and in a quiet, resting state of
discharge of many neurons in the cerebral cortex at a
cerebration. These waves occur most intensely in the oc-
frequency of about 12 per second, thus causing alpha
cipital region but can also be recorded from the parietal
waves. Then, when the eyes were opened, the activity of
and frontal regions of the scalp. Their voltage usually is
the brain increased greatly, but synchronization of the
about 50 microvolts. During deep sleep, the alpha waves
signals became so little that the brain waves mainly nul-
disappear.
lified one another, and the resultant effect was very low
When the awake person’s attention is directed to
voltage waves of generally high but irregular frequency,
some specific type of mental activity, the alpha waves
the beta waves.
are replaced by asynchronous, higher-frequency but
lower-voltage beta waves. Figure 59-2 shows the effect
Origin of Alpha Waves. Alpha waves will not occur in the
on the alpha waves of simply opening the eyes in bright
cerebral cortex without cortical connections with the
light and then closing the eyes. Note that the visual sen-
thalamus. Conversely, stimulation in the nonspecific
sations cause immediate cessation of the alpha waves
layer of reticular nuclei that surround the thalamus or
and that these are replaced by low-voltage, asynchro-
in “diffuse” nuclei deep inside the thalamus often sets
nous beta waves.
up electrical waves in the thalamocortical system at a
Beta waves occur at frequencies greater than 14 cycles
frequency between 8 and 13 per second, which is the
per second and as high as 80 cycles per second. They are
natural frequency of the alpha waves. Therefore, it is
recorded mainly from the parietal and frontal regions
believed that the alpha waves result from spontaneous
during specific activation of these parts of the brain.
feedback oscillation in this diffuse thalamocortical
Theta waves have frequencies between 4 and 7 cycles
system, possibly including the reticular activating
per second. They occur normally in the parietal and
system in the brain stem as well. This oscillation pre-
temporal regions in children, but they also occur during
sumably causes both the periodicity of the alpha waves
emotional stress in some adults, particularly during dis-
and the synchronous activation of literally millions of
appointment and frustration. Theta waves also occur in
cortical neurons during each wave.
many brain disorders, often in degenerative brain states.
Delta waves include all the waves of the EEG with
Origin of Delta Waves. Transection of the fiber tracts from
frequencies less than 3.5 cycles per second, and they
the thalamus to the cerebral cortex, which blocks thal-
often have voltages two to four times greater than most
amic activation of the cortex and thereby eliminates the
other types of brain waves. They occur in very deep
alpha waves, nevertheless does not block delta waves in
sleep, in infancy, and in serious organic brain disease.
the cortex. This indicates that some synchronizing mech-
They also occur in the cortex of animals that have had
anism can occur in the cortical neuronal system by
subcortical transections separating the cerebral cortex
itself—mainly independent of lower structures in the
from the thalamus. Therefore, delta waves can occur
brain—to cause the delta waves.
strictly in the cortex independent of activities in lower
Delta waves also occur during deep slow-wave sleep;
regions of the brain.
this suggests that the cortex then is mainly released
Chapter 59
States of Brain Activity—Sleep, Brain Waves, Epilepsy, Psychoses
743
Figure 59-3
Effect of varying degrees of cere-
Stupor
Sleep Psychomotor
Infants Relaxation Attention
Grand mal
bral activity on the basic rhythm
of the electroencephalogram.
Surgical
Slow component
Deteriorated epileptics Fright Fast component
(Redrawn from Gibbs FA, Gibbs
anesthesia
of petit mal
of petit mal
EL: Atlas of Electroencephalogra-
Confusion
phy, 2nd ed, Vol I: Methodology
and Controls. " 1974. Reprinted
1 second
by permission of Prentice-Hall,
Inc., Upper Saddle River, NJ.)
from the activating influences of the thalamus and other
lower centers.
Alert wakefulness (beta waves)
Effect of Varying Levels of
Cerebral Activity on the Frequency
of the EEG
Quiet wakefulness (alpha waves)
There is a general correlation between level of cerebral
activity and average frequency of the EEG rhythm, the
Stage 1 sleep (low voltage and spindles)
average frequency increasing progressively with higher
degrees of activity. This is demonstrated in Figure 59-3,
50 mV
which shows the existence of delta waves in stupor,
Stages 2 and 3 sleep (theta waves)
surgical anesthesia, and deep sleep; theta waves in
psychomotor states and in infants; alpha waves during
relaxed states; and beta waves during periods of intense
mental activity. During periods of mental activity, the
Stage 4 slow wave sleep (delta waves)
waves usually become asynchronous rather than syn-
chronous, so that the voltage falls considerably, despite
markedly increased cortical activity, as shown in
REM sleep (beta waves)
Figure 59-2.
1 sec
Changes in the EEG at Different
Stages of Wakefulness and Sleep
Figure 59-4
Figure 59-4 shows EEG patterns from a typical person
in different stages of wakefulness and sleep. Alert wake-
Progressive change in the characteristics of the brain waves
fulness is characterized by high-frequency beta waves,
during different stages of wakefulness and sleep.
whereas quiet wakefulness is usually associated with
alpha waves, as demonstrated by the first two EEGs of
the figure.
Slow-wave sleep is divided into four stages. In the first
central nervous system. A person who is predisposed to
stage, a stage of very light sleep, the voltage of the EEG
epilepsy has attacks when the basal level of excitability
waves becomes very low; this is broken by “sleep spin-
of the nervous system (or of the part that is susceptible
dles,” that is, short spindle-shaped bursts of alpha waves
to the epileptic state) rises above a certain critical
that occur periodically. In stages 2, 3, and 4 of slow-wave
threshold. As long as the degree of excitability is held
sleep, the frequency of the EEG becomes progressively
below this threshold, no attack occurs.
slower until it reaches a frequency of only 1 to 3 waves
Epilepsy can be classified into three major types:
per second in stage 4; these are delta waves.
grand mal epilepsy, petit mal epilepsy, and focal epilepsy.
Finally, the bottom record in Figure 59-4 shows the
EEG during REM sleep. It is often difficult to tell the
difference between this brain wave pattern and that of
Grand Mal Epilepsy
an awake, active person. The waves are irregular and
high-frequency, which are normally suggestive of desyn-
Grand mal epilepsy is characterized by extreme neu-
chronized nervous activity as found in the awake state.
ronal discharges in all areas of the brain—in the cere-
Therefore, REM sleep is frequently called desynchro-
bral cortex, in the deeper parts of the cerebrum, and
nized sleep because there is lack of synchrony in the
even in the brain stem. Also, discharges transmitted all
firing of the neurons, despite significant brain activity.
the way into the spinal cord sometimes cause general-
ized tonic seizures of the entire body, followed toward
the end of the attack by alternating tonic and spasmodic
Epilepsy
muscle contractions called tonic-clonic seizures. Often
the person bites or “swallows” his or her tongue and
Epilepsy (also called “seizures”) is characterized by
may have difficulty breathing, sometimes to the extent
uncontrolled excessive activity of either part or all of the
that cyanosis occurs. Also, signals transmitted from the
744
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Even in people who are not genetically predisposed,
certain types of traumatic lesions in almost any part of
the brain can cause excess excitability of local brain
100 mV
areas, as we discuss shortly; these, too, sometimes trans-
mit signals into the activating systems of the brain to
elicit grand mal seizures.
Grand mal
What Stops the Grand Mal Attack? The cause of the extreme
neuronal overactivity during a grand mal attack is pre-
50 mV
sumed to be massive simultaneous activation of many
reverberating neuronal pathways throughout the brain.
Presumably, the major factor that stops the attack after
Petit mal
a few minutes is neuronal fatigue. A second factor is
probably active inhibition by inhibitory neurons that
50 mV
have been activated by the attack.
Psychomotor
Petit Mal Epilepsy
Petit mal epilepsy almost certainly involves the thala-
mocortical brain activating system. It is usually charac-
Figure 59-5
terized by
3 to
30 seconds of unconsciousness
(or
diminished consciousness) during which time the
Electroencephalograms in different types of epilepsy.
person has twitch-like contractions of muscles usually in
the head region, especially blinking of the eyes; this is
followed by return of consciousness and resumption of
previous activities. This total sequence is called the
brain to the viscera frequently cause urination and defe-
absence syndrome or absence epilepsy. The patient
cation.
may have one such attack in many months or, in rare
The usual grand mal seizure lasts from a few seconds
instances, may have a rapid series of attacks, one after
to 3 to 4 minutes. It is also characterized by postseizure
the other. The usual course is for the petit mal attacks
depression of the entire nervous system; the person
to appear first during late childhood and then to disap-
remains in stupor for 1 to many minutes after the
pear by the age of 30. On occasion, a petit mal epilep-
seizure attack is over, and then often remains severely
tic attack will initiate a grand mal attack.
fatigued and asleep for hours thereafter.
The brain wave pattern in petit mal epilepsy is
The top recording of Figure 59-5 shows a typical
demonstrated by the middle recording of Figure 59-5,
EEG from almost any region of the cortex during the
which is typified by a spike and dome pattern. The spike
tonic phase of a grand mal attack. This demonstrates
and dome can be recorded over most or all of the cere-
that high-voltage, high-frequency discharges occur over
bral cortex, showing that the seizure involves much
the entire cortex. Furthermore, the same type of dis-
or most of the thalamocortical activating system of
charge occurs on both sides of the brain at the same
the brain. In fact, animal studies suggest that it results
time, demonstrating that the abnormal neuronal cir-
from oscillation of
(1) inhibitory thalamic reticular
cuitry responsible for the attack strongly involves the
neurons
(which are inhibitory gamma-aminobutyric
basal regions of the brain that drive the two halves of
acid [GABA]-producing neurons) and (2) excitatory
the cerebrum simultaneously.
thalamocortical and corticothalamic neurons.
In laboratory animals and even in human beings,
grand mal attacks can be initiated by administering a
neuronal stimulant such as the drug pentylenetetrazol,
Focal Epilepsy
or they can be caused by insulin hypoglycemia, or by
passage of alternating electrical current directly through
Focal epilepsy can involve almost any local part of the
the brain. Electrical recordings from the thalamus as
brain, either localized regions of the cerebral cortex or
well as from the reticular formation of the brain stem
deeper structures of both the cerebrum and brain stem.
during the grand mal attack show typical high-voltage
Most often, focal epilepsy results from some localized
activity in both of these areas similar to that recorded
organic lesion or functional abnormality, such as (1) scar
from the cerebral cortex. Presumably, therefore, a grand
tissue in the brain that pulls on the adjacent neuronal
mal attack involves not only abnormal activation of the
tissue, (2) a tumor that compresses an area of the brain,
thalamus and cerebral cortex but also abnormal activa-
(3) a destroyed area of brain tissue, or (4) congenitally
tion in the subthalamic brain stem portions of the brain
deranged local circuitry.
activating system itself.
Lesions such as these can promote extremely rapid
discharges in the local neurons; when the discharge rate
What Initiates a Grand Mal Attack? Most people who have
rises above several hundred per second, synchronous
grand mal attacks have hereditary predisposition to
waves begin to spread over adjacent cortical regions.
epilepsy, a predisposition that occurs in about 1 of every
These waves presumably result from localized reverber-
50 to 100 persons. In such people, factors that can
ating circuits that gradually recruit adjacent areas of the
increase the excitability of the abnormal “epilepto-
cortex into the epileptic discharge zone. The process
genic” circuitry enough to precipitate attacks include
spreads to adjacent areas at a rate as slow as a few mil-
(1) strong emotional stimuli, (2) alkalosis caused by
limeters a minute to as fast as several centimeters per
overbreathing, (3) drugs, (4) fever, and (5) loud noises
second. When such a wave of excitation spreads over the
or flashing lights.
motor cortex, it causes progressive “march” of muscle
Chapter 59
States of Brain Activity—Sleep, Brain Waves, Epilepsy, Psychoses
745
contractions throughout the opposite side of the body,
neurotransmitters.) Depressed patients experience
beginning most characteristically in the mouth region
symptoms of grief, unhappiness, despair, and misery.
and marching progressively downward to the legs but at
In addition, they often lose their appetite and sex
other times marching in the opposite direction. This is
drive and have severe insomnia. Often associated with
called jacksonian epilepsy.
these is a state of psychomotor agitation despite the
A focal epileptic attack may remain confined to a
depression.
single area of the brain, but in many instances, the
Moderate numbers of norepinephrine-secreting
strong signals from the convulsing cortex excite the
neurons are located in the brain stem, especially in
mesencephalic portion of the brain activating system so
the locus ceruleus. These neurons send fibers upward
greatly that a grand mal epileptic attack ensues as well.
to most parts of the brain limbic system, thalamus,
Another type of focal epilepsy is the so-called psy-
and cerebral cortex. Also, many serotonin-producing
chomotor seizure, which may cause (1) a short period of
neurons located in the midline raphe nuclei of the lower
amnesia; (2) an attack of abnormal rage; (3) sudden
pons and medulla send fibers to many areas of the
anxiety, discomfort, or fear; and/or (4) a moment of
limbic system and to some other areas of the brain.
incoherent speech or mumbling of some trite phrase.
A principal reason for believing that depres-
Sometimes the person cannot remember his or her
sion might be caused by diminished activity of
activities during the attack, but at other times, he or she
norepinephrine- and serotonin-secreting neurons is that
is conscious of everything that he or she is doing but
drugs that block secretion of norepinephrine and sero-
unable to control it. Attacks of this type frequently
tonin, such as reserpine, frequently cause depression.
involve part of the limbic portion of the brain, such as
Conversely, about 70 per cent of depressive patients can
the hippocampus, the amygdala, the septum, and/or por-
be treated effectively with drugs that increase the exci-
tions of the temporal cortex.
tatory effects of norepinephrine and serotonin at the
The lowest tracing of Figure 59-5 demonstrates a
nerve endings—for instance, (1) monoamine oxidase
typical EEG during a psychomotor seizure, showing a
inhibitors, which block destruction of norepinephrine
low-frequency rectangular wave with a frequency
and serotonin once they are formed; and (2) tricyclic
between 2 and 4 per second and with occasional super-
antidepressants, such as imipramine and amitriptyline,
imposed 14-per-second waves.
which block reuptake of norepinephrine and serotonin
by nerve endings so that these transmitters remain
Surgical Excision of Epileptic Foci Can Often Prevent Seizures.
active for longer periods after secretion.
The EEG can be used to localize abnormal spiking
Mental depression can be treated by electroconvul-
waves originating in areas of organic brain disease that
sive therapy—commonly called “shock therapy.” In this
predispose to focal epileptic attacks. Once such a focal
therapy, electrical current is passed through the brain to
point is found, surgical excision of the focus frequently
cause a generalized seizure similar to that of an epilep-
prevents future attacks.
tic attack. This has been shown to enhance norepine-
phrine activity.
Some patients with mental depression alternate
between depression and mania, which is called either
Psychotic Behavior and
bipolar disorder or manic-depressive psychosis, and a
Dementia—Roles of Specific
few people exhibit only mania without the depressive
Neurotransmitter Systems
episodes. Drugs that diminish the formation or action of
norepinephrine and serotonin, such as lithium com-
Clinical studies of patients with different psychoses or
pounds, can be effective in treating the manic phase of
different types of dementia have suggested that many
the condition.
of these conditions result from diminished function of
It is presumed that the norepinephrine and serotonin
neurons that secrete a specific neurotransmitter. Use of
systems normally provide drive to the limbic areas of
appropriate drugs to counteract loss of the respective
the brain to increase a person’s sense of well-being, to
neurotransmitter has been successful in treating some
create happiness, contentment, good appetite, appropri-
patients.
ate sex drive, and psychomotor balance—although too
In Chapter 56, we discussed the cause of Parkinson’s
much of a good thing can cause mania. In support of this
disease. This disease results from loss of neurons in the
concept is the fact that pleasure and reward centers of
substantia nigra whose nerve endings secrete dopamine
the hypothalamus and surrounding areas receive large
in the caudate nucleus and putamen. Also in Chapter
numbers of nerve endings from the norepinephrine and
56, we pointed out that in Huntington’s disease, loss of
serotonin systems.
GABA-secreting neurons and acetylcholine-secreting
neurons is associated with specific abnormal motor pat-
terns plus dementia occurring in the same patient.
Schizophrenia—Possible
Exaggerated Function of Part of the
Dopamine System
Depression and Manic-Depressive
Psychoses—Decreased Activity of
Schizophrenia comes in many varieties. One of the most
the Norepinephrine and Serotonin
common types is seen in the person who hears voices
Neurotransmitter Systems
and has delusions of grandeur, intense fear, or other
types of feelings that are unreal. Many schizophrenics
Much evidence has accumulated suggesting that mental
(1) are highly paranoid, with a sense of persecution from
depression psychosis, which occurs in about 8 million
outside sources; (2) may develop incoherent speech, dis-
people in the United States, might be caused by dimin-
sociation of ideas, and abnormal sequences of thought;
ished formation in the brain of norepinephrine or sero-
and (3) are often withdrawn, sometimes with abnormal
tonin, or both. (New evidence has implicated still other
posture and even rigidity.
746
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
There are reasons to believe that schizophrenia
behavioral disturbances in the later stages of the
results from one or more of three possibilities: (1) mul-
disease. Patients with Alzheimer’s disease usually
tiple areas in the cerebral cortex prefrontal lobes in
require continuous care within a few years after the
which neural signals have become blocked or where
disease begins.
processing of the signals becomes dysfunctional because
Alzheimer’s disease is the most common form of
many synapses normally excited by the neurotransmit-
dementia in the elderly and about 5 million people in
ter glutamate lose their responsiveness to this transmit-
the United States are estimated to be afflicted by this
ter; (2) excessive excitement of a group of neurons that
disorder. The percentage of persons with Alzheimer’s
secrete dopamine in the behavioral centers of the brain,
disease approximately doubles with every five years of
including in the frontal lobes; and/or (3) abnormal func-
age, with about 1 percent of 60-year-olds and about 30
tion of a crucial part of the brain’s limbic behavioral
percent of 85-year-olds having the disease.
control system centered around the hippocampus.
The reason for believing that the prefrontal lobes are
Alzheimer’s Disease Is Associated with Accumulation of Brain
involved in schizophrenia is that a schizophrenic-like
Beta-Amyloid Peptide. Pathologically, one finds increased
pattern of mental activity can be induced in monkeys by
amounts of beta-amyloid peptide in the brains of
making multiple minute lesions in widespread areas of
patients with Alzheimer’s disease. The peptide accumu-
the prefrontal lobes.
lates in amyloid plaques, which range in diameter from
Dopamine has been implicated as a possible cause of
10 micrometers to several hundred micrometers and are
schizophrenia because many patients with Parkinson’s
found in widespread areas of the brain, including in the
disease develop schizophrenic-like symptoms when
cerebral cortex, hippocampus, basal ganglia, thalamus,
they are treated with the drug called L-dopa. This drug
and even the cerebellum. Thus, Alzheimer’s disease
releases dopamine in the brain, which is advantageous
appears to be a metabolic degenerative disease.
for treating Parkinson’s disease, but at the same time it
A key role for excess accumulation of beta-amyloid
depresses various portions of the prefrontal lobes and
peptide in the pathogenesis of Alzheimer’s disease is
other related areas.
suggested by the following observations: (1) all cur-
It has been suggested that in schizophrenia excess
rently known mutations associated with Alzheimer’s
dopamine is secreted by a group of dopamine-secreting
disease increase the production of beta-amyloid
neurons whose cell bodies lie in the ventral tegmentum
peptide; (2) patients with trisomy 21 (Down syndrome)
of the mesencephalon, medial and superior to the sub-
have three copies of the gene for amyloid precursor
stantia nigra. These neurons give rise to the so-called
protein and develop neurological characteristics of
mesolimbic dopaminergic system that projects nerve
Alzheimer’s disease by midlife; (3) patients who have
fibers and dopamine secretion into the medial and ante-
abnormality of a gene that controls apolipoprotein E, a
rior portions of the limbic system, especially into the
blood protein that transports cholesterol to the tissues,
hippocampus, amygdala, anterior caudate nucleus, and
have accelerated deposition of amyloid and greatly
portions of the prefrontal lobes. All of these are pow-
increased risk for Alzheimer’s disease; (4) transgenic
erful behavioral control centers.
mice that overproduce the human amyloid precursor
An even more compelling reason for believing that
protein have learning and memory deficits in associa-
schizophrenia might be caused by excess production of
tion with the accumulation of amyloid plaques; and (5)
dopamine is that many drugs that are effective in treat-
generation of anti-amyloid antibodies in humans with
ing schizophrenia—such as chlorpromazine, haloperi-
Alzheimer’s disease appears to attenuate the disease
dol, and thiothixene—-all either decrease secretion of
process.
dopamine at dopaminergic nerve endings or decrease
the effect of dopamine on subsequent neurons.
Vascular Disorders May Contribute to Progression of Alzheimer’s
Finally, possible involvement of the hippocampus in
Disease. There is also accumulating evidence that
schizophrenia was discovered recently when it was
cerebrovascular disease caused by hypertension and
learned that in schizophrenia, the hippocampus is often
atherosclerosis may play a role in Alzheimer’s disease.
reduced in size, especially in the dominant hemisphere.
Cerebrovascular disease is the second most common
cause of acquired cognitive impairment and dementia
and likely contributes to cognitive decline in
Alzheimer’s Disease—Amyloid
Alzheimer’s disease. In fact, many of the common risk
Plaques and Depressed Memory
factors for cerebrovascular disease, such as hyperten-
sion, diabetes, and hyperlipidemia, are also recognized
Alzheimer’s disease is defined as premature aging of the
to greatly increase the risk for developing Alzheimer’s
brain, usually beginning in mid-adult life and progress-
disease.
ing rapidly to extreme loss of mental powers—similar
to that seen in very, very old age. The clinical features
of Alzheimer’s disease include (1) an amnesic type of
memory impairment, (2) deterioration of language, and
References
(3) visuospatial deficits. Motor and sensory abnormali-
ties, gait disturbances, and seizures are uncommon until
Aldrich MS: Sleep Medicine: Normal Sleep and Its Disor-
the late phases of the disease. One consistent finding in
ders. New York: Oxford University Press, 1999.
Alzheimer’s disease is loss of neurons in that part of the
Beardsley T: Waking up. Sci Am Jul 18, 1996.
limbic pathway that drives the memory process. Loss of
Bryant PA, Trinder J, Curtis N: Sick and tired: does sleep
this memory function is devastating.
have a vital role in the immune system? Nat Rev Immunol
Alzheimer’s disease is a progressive and fatal neu-
4:457, 2004.
rodegenerative disorder that results in impairment of
Casserly I, Topol E: Convergence of atherosclerosis and
the person’s ability to perform activities of daily living
Alzheimer’s disease: inflammation, cholesterol, and mis-
as well as a variety of neuropsychiatric symptoms and
folded proteins. Lancet 363:1139, 2004.
Chapter 59
States of Brain Activity—Sleep, Brain Waves, Epilepsy, Psychoses
747
Cummings JL: Alzheimer’s disease. N Engl J Med 351:56,
Iadecola C: Neurovascular regulation in the normal brain
2004.
and in Alzheimer’s disease. Nat Rev Neurosci 5:347-360,
de la Torre JC: Is Alzheimer’s disease a neurodegenerative
2004.
or a vascular disorder? Data, dogma, and dialectics. Lancet
LaRoche SM, Helmers SL: The new antiepileptic drugs: sci-
Neurol 3:184, 2004.
entific review. JAMA 291:605, 2004.
George AL Jr: Molecular basis of inherited epilepsy. Arch
McCormick DA, Contreras D: On the cellular and network
Neurol 61:473, 2004.
bases of epileptic seizures. Annu Rev Physiol
63:815,
Gerashchenko D, Shiromani PJ: Different neuronal pheno-
2001.
types in the lateral hypothalamus and their role in sleep
Noebels JL: The biology of epilepsy genes. Annu Rev Neu-
and wakefulness. Mol Neurobiol 29:41, 2004.
rosci 26:599, 2003.
Golde TE: Alzheimer disease therapy: can the amyloid
Selkoe DJ: Alzheimer disease: mechanistic understanding
cascade be halted? J Clin Invest 111:11, 2003.
predicts novel therapies. Ann Intern Med 140:627, 2004.
Gourfinkel-An I, Baulac S, Nabbout R, et al: Monogenic
Selkoe DJ: Alzheimer’s disease: genes, proteins, and therapy.
idiopathic epilepsies. Lancet Neurol 3:209, 2004.
Physiol Rev 81:741, 2001.
Greene R, Siegel J: Sleep: a functional enigma. Neuromo-
Steinlein OK: Genetic mechanisms that underlie epilepsy.
lecular Med 5:59, 2004.
Nat Rev Neurosci 5:400-408, 2004.
Heller HC, Ruby NF: Sleep and circadian rhythms in mam-
malian torpor. Annu Rev Physiol 66:275, 2004.
C H A P T E R
6
0
The Autonomic Nervous System
and the Adrenal Medulla
The portion of the nervous system that controls
most visceral functions of the body is called the
autonomic nervous system. This system helps to
control arterial pressure, gastrointestinal motility,
gastrointestinal secretion, urinary bladder empty-
ing, sweating, body temperature, and many other
activities, some of which are controlled almost
entirely and some only partially by the autonomic
nervous system.
One of the most striking characteristics of the autonomic nervous system is
the rapidity and intensity with which it can change visceral functions. For
instance, within 3 to 5 seconds it can increase the heart rate to twice normal,
and within 10 to 15 seconds the arterial pressure can be doubled; or, at the other
extreme, the arterial pressure can be decreased low enough within 10 to 15
seconds to cause fainting. Sweating can begin within seconds, and the urinary
bladder may empty involuntarily, also within seconds.
General Organization of the Autonomic
Nervous System
The autonomic nervous system is activated mainly by centers located in the
spinal cord, brain stem, and hypothalamus. Also, portions of the cerebral cortex,
especially of the limbic cortex, can transmit signals to the lower centers and in
this way influence autonomic control.
The autonomic nervous system also often operates by means of visceral
reflexes. That is, subconscious sensory signals from a visceral organ can enter
the autonomic ganglia, the brain stem, or the hypothalamus and then return
subconscious reflex responses directly back to the visceral organ to control its
activities.
The efferent autonomic signals are transmitted to the various organs of the
body through two major subdivisions called the sympathetic nervous system and
the parasympathetic nervous system, the characteristics and functions of which
follow.
Physiologic Anatomy of the Sympathetic Nervous System
Figure 60-1 shows the general organization of the peripheral portions of the sym-
pathetic nervous system. Shown specifically in the figure are (1) one of the two
paravertebral sympathetic chains of ganglia that are interconnected with the spinal
nerves on the side of the vertebral column, (2) two prevertebral ganglia (the celiac
and hypogastric), and (3) nerves extending from the ganglia to the different inter-
nal organs.
The sympathetic nerve fibers originate in the spinal cord along with spinal nerves
between cord segments T-1 and L-2 and pass first into the sympathetic chain and
then to the tissues and organs that are stimulated by the sympathetic nerves.
Preganglionic and Postganglionic Sympathetic Neurons
The sympathetic nerves are different from skeletal motor nerves in the following
way: Each sympathetic pathway from the cord to the stimulated tissue is composed
748
Chapter 60
The Autonomic Nervous System and the Adrenal Medulla
749
Posterior root
Spinal nerve
Eye
Intermedio-
White ramus
lateral horn
Gray ramus
Piloerector
muscle
Sympathetic chain
8
Heart
T-1
Sweat
Bronchi
Anterior root
gland
Preganglionic nerve
fiber
12
Peripheral ganglion
Blood
Celiac
Postganglionic nerve
vessel
ganglion
fibers
Pylorus
Effector endings
L-1
Sensory endings
Adrenal
Gut
5
medulla
Kidney
Ureter
Figure 60-2
5
Nerve connections between the spinal cord, spinal nerves, sym-
Intestine
pathetic chain, and peripheral sympathetic nerves.
Ileocecal valve
Sympathetic Nerve Fibers in the Skeletal Nerves. Some of the
Hypogastric plexus
Anal sphincter
postganglionic fibers pass back from the sympathetic
Detrusor
chain into the spinal nerves through gray rami at all
levels of the cord, as shown in Figure 60-2. These sym-
Trigone
pathetic fibers are all very small type C fibers, and they
extend to all parts of the body by way of the skeletal
Figure 60-1
nerves. They control the blood vessels, sweat glands, and
Sympathetic nervous system. The black dashed lines represent
piloerector muscles of the hairs. About 8 per cent of the
postganglionic fibers in the gray rami leading from the sympa-
fibers in the average skeletal nerve are sympathetic
thetic chains into spinal nerves for distribution to blood vessels,
fibers, a fact that indicates their great importance.
sweat glands, and piloerector muscles.
Segmental Distribution of the Sympathetic Nerve Fibers. The
of two neurons, a preganglionic neuron and a postgan-
sympathetic pathways that originate in the different
glionic neuron, in contrast to only a single neuron in the
segments of the spinal cord are not necessarily distrib-
skeletal motor pathway. The cell body of each pregan-
uted to the same part of the body as the somatic spinal
glionic neuron lies in the intermediolateral horn of the
nerve fibers from the same segments. Instead, the sym-
spinal cord; its fiber passes, as shown in Figure 60-2,
pathetic fibers from cord segment T-1 generally pass up
through an anterior root of the cord into the corre-
the sympathetic chain to terminate in the head; from T-2
sponding spinal nerve.
to terminate in the neck; from T-3, T-4, T-5, and T-6
Immediately after the spinal nerve leaves the spinal
into the thorax; from T-7, T-8, T-9, T-10, and T-11 into
canal, the preganglionic sympathetic fibers leave the
the abdomen; and from T-12, L-1, and L-2 into the legs.
spinal nerve and pass through a white ramus into one of
This distribution is only approximate and overlaps
the ganglia of the sympathetic chain. Then the course
greatly.
of the fibers can be one of the following three: (1) It
The distribution of sympathetic nerves to each organ
can synapse with postganglionic sympathetic neurons
is determined partly by the locus in the embryo from
in the ganglion that it enters; (2) it can pass upward
which the organ originated. For instance, the heart
or downward in the chain and synapse in one of the
receives many sympathetic nerve fibers from the neck
other ganglia of the chain; or (3) it can pass for variable
portion of the sympathetic chain because the heart orig-
distances through the chain and then through one
inated in the neck of the embryo before translocating
of the sympathetic nerves radiating outward from the
into the thorax. Likewise, the abdominal organs receive
chain, finally synapsing in a peripheral sympathetic
most of their sympathetic innervation from the lower
ganglion.
thoracic spinal cord segments because most of the prim-
The postganglionic sympathetic neuron thus origi-
itive gut originated in this area.
nates either in one of the sympathetic chain ganglia
or in one of the peripheral sympathetic ganglia.
Special Nature of the Sympathetic Nerve Endings in the Adrenal
From either of these two sources, the postganglionic
Medullae. Preganglionic sympathetic nerve fibers pass,
fibers then travel to their destinations in the various
without synapsing, all the way from the intermedio-
organs.
lateral horn cells of the spinal cord, through the
750
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
sympathetic chains, then through the splanchnic nerves,
esophagus, stomach, entire small intestine, proximal half
and finally into the two adrenal medullae. There they
of the colon, liver, gallbladder, pancreas, kidneys, and
end directly on modified neuronal cells that secrete epi-
upper portions of the ureters.
nephrine and norepinephrine into the blood stream.
Parasympathetic fibers in the third cranial nerve go to
These secretory cells embryologically are derived from
the pupillary sphincter and ciliary muscle of the eye.
nervous tissue and are actually themselves postgan-
Fibers from the seventh cranial nerve pass to the
glionic neurons; indeed, they even have rudimentary
lacrimal, nasal, and submandibular glands. And fibers
nerve fibers, and it is the endings of these fibers that
from the ninth cranial nerve go to the parotid gland.
secrete the adrenal hormones epinephrine and
The sacral parasympathetic fibers are in the pelvic
norepinephrine.
nerves, which pass through the spinal nerve sacral
plexus on each side of the cord at the S-2 and S-3 levels.
These fibers then distribute to the descending colon,
Physiologic Anatomy of the
rectum, urinary bladder, and lower portions of the
Parasympathetic Nervous System
ureters. Also, this sacral group of parasympathetics sup-
plies nerve signals to the external genitalia to cause
The parasympathetic nervous system is shown in Figure
erection.
60-3, demonstrating that parasympathetic fibers leave
the central nervous system through cranial nerves III,
Preganglionic and Postganglionic Parasympathetic Neurons.
VII, IX, and X; additional parasympathetic fibers leave
The parasympathetic system, like the sympathetic, has
the lowermost part of the spinal cord through the
both preganglionic and postganglionic neurons.
second and third sacral spinal nerves and occasionally
However, except in the case of a few cranial parasym-
the first and fourth sacral nerves. About 75 per cent of
pathetic nerves, the preganglionic fibers pass uninter-
all parasympathetic nerve fibers are in the vagus nerves
rupted all the way to the organ that is to be controlled.
(cranial nerve X), passing to the entire thoracic and
In the wall of the organ are located the postganglionic
abdominal regions of the body. Therefore, a physiologist
neurons. The preganglionic fibers synapse with these,
speaking of the parasympathetic nervous system often
and very, very short postganglionic fibers, a fraction of
thinks mainly of the two vagus nerves. The vagus nerves
a millimeter to several centimeters in length, leave the
supply parasympathetic
nerves
to the heart, lungs,
neurons to innervate the tissues of the organ. This loca-
tion of the parasympathetic postganglionic neurons in
the visceral organ itself is quite different from the
arrangement of the sympathetic ganglia, because the
cell bodies of the sympathetic postganglionic neurons
Ciliary ganglion
are almost always located in the ganglia of the sympa-
Ciliary muscles of eye
thetic chain or in various other discrete ganglia in the
III
abdomen, rather than in the excited organ itself.
Pupillary sphincter
Sphenopalatine ganglion
VII
IX
Lacrimal glands
V
Nasal glands
Basic Characteristics
X
Submandibular ganglion
of Sympathetic and
Submandibular gland
Parasympathetic Function
Otic ganglion
Parotid gland
Cholinergic and Adrenergic Fibers—
Heart
Secretion of Acetylcholine or
Norepinephrine
The sympathetic and parasympathetic nerve fibers
Stomach
secrete mainly one or the other of two synaptic trans-
mitter substances, acetylcholine or norepinephrine.
Pylorus
Those fibers that secrete acetylcholine are said to be
cholinergic. Those that secrete norepinephrine are said
Colon
to be adrenergic, a term derived from adrenalin, which
is an alternate name for epinephrine.
Small intestine
All preganglionic neurons are cholinergic in both
Sacral
the sympathetic and the parasympathetic nervous
1
systems. Acetylcholine or acetylcholine-like sub-
2
Ileocecal valve
stances, when applied to the ganglia, will excite both
3
4
Anal sphincter
sympathetic and parasympathetic postganglionic
neurons. Either all or almost all of the postganglionic
Bladder
neurons of the parasympathetic system are also cholin-
Detrusor
ergic. Conversely, most of the postganglionic sym-
Trigone
pathetic neurons are adrenergic. However, the
postganglionic sympathetic nerve fibers to the sweat
Figure 60-3
glands, to the piloerector muscles of the hairs, and to
Parasympathetic nervous system.
a very few blood vessels are cholinergic.
Chapter 60
The Autonomic Nervous System and the Adrenal Medulla
751
Thus, the terminal nerve endings of the parasympa-
Synthesis of Acetylcholine, Its Destruction After Secretion, and
thetic system all or virtually all secrete acetylcholine.
Its Duration of Action. Acetylcholine is synthesized in the
Almost all of the sympathetic nerve endings secrete
terminal endings and varicosities of the cholinergic
norepinephrine, but a few secrete acetylcholine. These
nerve fibers where it is stored in vesicles in highly con-
hormones in turn act on the different organs to cause
centrated form until it is released. The basic chemical
respective parasympathetic or sympathetic effects.
reaction of this synthesis is the following:
Therefore, acetylcholine is called a parasympathetic
choline acetyl-
transferase
transmitter and norepinephrine is called a sympathetic
Acetyl-CoA+Choline
ææ
æææÆAcetylcholine
transmitter.
The molecular structures of acetylcholine and nor-
Once acetylcholine is secreted into a tissue by a
epinephrine are the following:
cholinergic nerve ending, it persists in the tissue for a
few seconds while it performs its nerve signal trans-
mitter function. Then it is split into an acetate ion and
+
choline, catalyzed by the enzyme acetylcholinesterase
CH3
that is bound with collagen and glycosaminoglycans in
the local connective tissue. This is the same mechanism
CH
3
C
O CH2
CH2
N
for acetylcholine signal transmission and subsequent
CH3
acetylcholine destruction that occurs at the neuro-
O
CH3
muscular junctions of skeletal nerve fibers. The choline
Acetylcholine
that is formed is then transported back into the ter-
minal nerve ending, where it is used again and again
for synthesis of new acetylcholine.
HO
Synthesis of Norepinephrine, Its Removal, and Its Duration of
Action. Synthesis of norepinephrine begins in the axo-
plasm of the terminal nerve endings of adrenergic
HO
CH
CH2
NH2
nerve fibers but is completed inside the secretory vesi-
cles. The basic steps are the following:
OH
hydroxylation
1.
Tyrosineæ
ææææÆDopa
Norepinephrine
decarboxylation
2.
Dopaæ
ææææÆDopamine
3. Transport of dopamine into the vesicles
Mechanisms of Transmitter Secretion and
hydroxylation
4.
Dopamine
æ
ææææÆNorepinephrine
Subsequent Removal of the Transmitter
at the Postganglionic Endings
In the adrenal medulla, this reaction goes still one
step further to transform about 80 per cent of the nor-
Secretion of Acetylcholine and Norepinephrine by Postgan-
epinephrine into epinephrine, as follows:
glionic Nerve Endings. A few of the postganglionic
methylation
5.
Norepinephrine
æ
æææÆEpinephrine
autonomic nerve endings, especially those of the
parasympathetic nerves, are similar to but much
After secretion of norepinephrine by the terminal
smaller than those of the skeletal neuromuscular junc-
nerve endings, it is removed from the secretory site in
tion. However, many of the parasympathetic nerve
three ways: (1) reuptake into the adrenergic nerve
fibers and almost all the sympathetic fibers merely
endings themselves by an active transport process—
touch the effector cells of the organs that they inner-
accounting for removal of 50 to 80 per cent of the
vate as they pass by; or in some instances, they termi-
secreted norepinephrine; (2) diffusion away from the
nate in connective tissue located adjacent to the cells
nerve endings into the surrounding body fluids and
that are to be stimulated. Where these filaments touch
then into the blood—accounting for removal of most
or pass over or near the cells to be stimulated, they
of the remaining norepinephrine; and (3) destruction
usually have bulbous enlargements called varicosities;
of small amounts by tissue enzymes (one of these
it is in these varicosities that the transmitter vesicles of
enzymes is monoamine oxidase, which is found in the
acetylcholine or norepinephrine are synthesized and
nerve endings, and another is catechol-O-methyl trans-
stored. Also in the varicosities are large numbers of
ferase, which is present diffusely in all tissues).
mitochondria that supply adenosine triphosphate,
Ordinarily, the norepinephrine secreted directly into
which is required to energize acetylcholine or norepi-
a tissue remains active for only a few seconds, demon-
nephrine synthesis.
strating that its reuptake and diffusion away from the
When an action potential spreads over the terminal
tissue are rapid. However, the norepinephrine and
fibers, the depolarization process increases the perme-
epinephrine secreted into the blood by the adrenal
ability of the fiber membrane to calcium ions, allowing
medullae remain active until they diffuse into some
these ions to diffuse into the nerve terminals or nerve
tissue, where they can be destroyed by catechol-O-
varicosities. The calcium ions in turn cause the termi-
methyl transferase; this occurs mainly in the liver.
nals or varicosities to empty their contents to the exte-
Therefore, when secreted into the blood, both norepi-
rior. Thus, the transmitter substance is secreted.
nephrine and epinephrine remain very active for 10 to
752
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
30 seconds; but their activity declines to extinction
are likely to be entirely different from those in other
over 1 to several minutes.
organs.
Two Principal Types of Acetylcholine
Receptors on the Effector Organs
Receptors—Muscarinic and Nicotinic
Receptors
Before an acetylcholine, norepinephrine, or epineph-
Acetylcholine activates mainly two types of receptors.
rine secreted at an autonomic nerve ending can stim-
They are called muscarinic and nicotinic receptors. The
ulate an effector organ, it must first bind with specific
reason for these names is that muscarine, a poison
receptors on the effector cells. The receptor is on the
from toadstools, activates only muscarinic receptors
outside of the cell membrane, bound as a prosthetic
and will not activate nicotinic receptors, whereas nico-
group to a protein molecule that penetrates all the way
tine activates only nicotinic receptors; acetylcholine
through the cell membrane. When the transmitter sub-
activates both of them.
stance binds with the receptor, this causes a confor-
Muscarinic receptors are found on all effector cells
mational change in the structure of the protein
that are stimulated by the postganglionic cholinergic
molecule. In turn, the altered protein molecule excites
neurons of either the parasympathetic nervous system
or inhibits the cell, most often by (1) causing a change
or the sympathetic system.
in cell membrane permeability to one or more ions or
Nicotinic receptors are found in the autonomic
(2) activating or inactivating an enzyme attached to
ganglia at the synapses between the preganglionic and
the other end of the receptor protein where it pro-
postganglionic neurons of both the sympathetic and
trudes into the interior of the cell.
parasympathetic systems. (Nicotinic receptors are also
present at many nonautonomic nerve endings—for
Excitation or Inhibition of the Effector Cell by Changing Its
instance, at the neuromuscular junctions in skeletal
Membrane Permeability. Because the receptor protein
muscle [discussed in Chapter 7].)
is an integral part of the cell membrane, a conforma-
An understanding of the two types of receptors is
tional change in structure of the receptor protein
especially important because specific drugs are fre-
often opens or closes an ion channel through the
quently used as medicine to stimulate or block one or
interstices of the protein molecule, thus altering the
the other of the two types of receptors.
permeability of the cell membrane to various ions.
For instance, sodium and/or calcium ion channels
Adrenergic Receptors—Alpha and
frequently become opened and allow rapid influx of
Beta Receptors
the respective ions into the cell, usually depolarizing
There are also two major types of adrenergic recep-
the cell membrane and exciting the cell. At other
tors, alpha receptors and beta receptors. (The beta
times, potassium channels are opened, allowing potas-
receptors in turn are divided into beta1 and beta2 recep-
sium ions to diffuse out of the cell, and this usually
tors because certain chemicals affect only certain beta
inhibits the cell because loss of electropositive
receptors. Also, there is a division of alpha receptors
potassium ions creates hypernegativity inside the cell.
into alpha1 and alpha2 receptors.)
In some cells, the changed intracellular ion environ-
Norepinephrine and epinephrine, both of which are
ment will cause an internal cell action, such as a direct
secreted into the blood by the adrenal medulla,
effect of calcium ions to promote smooth muscle
have slightly different effects in exciting the alpha and
contraction.
beta receptors. Norepinephrine excites mainly alpha
receptors but excites the beta receptors to a lesser
Receptor Action by Altering Intracellular “Second Messenger”
extent as well. Conversely, epinephrine excites both
Enzymes. Another way a receptor often functions is to
types of receptors approximately equally. Therefore,
activate or inactivate an enzyme (or other intracellu-
the relative effects of norepinephrine and epinephrine
lar chemical) inside the cell. The enzyme often is
on different effector organs are determined by the
attached to the receptor protein where the receptor
types of receptors in the organs. If they are all
protrudes into the interior of the cell. For instance,
beta receptors, epinephrine will be the more effective
binding of norepinephrine with its receptor on the
excitant.
outside of many cells increases the activity of the
Table 60-1 gives the distribution of alpha and beta
enzyme adenylyl cyclase on the inside of the cell, and
receptors in some of the organs and systems controlled
this causes formation of cyclic adenosine monophos-
by the sympathetics. Note that certain alpha functions
phate (cAMP). The cAMP in turn can initiate any one
are excitatory, whereas others are inhibitory. Likewise,
of many different intracellular actions, the exact effect
certain beta functions are excitatory and others are
depending on the chemical machinery of the effector
inhibitory. Therefore, alpha and beta receptors are not
cell.
necessarily associated with excitation or inhibition but
It is easy to understand how an autonomic trans-
simply with the affinity of the hormone for the recep-
mitter substance can cause inhibition in some organs
tors in the given effector organ.
or excitation in others. This is usually determined by
A synthetic hormone chemically similar to epi-
the nature of the receptor protein in the cell mem-
nephrine and norepinephrine, isopropyl norepineph-
brane and the effect of receptor binding on its con-
rine, has an extremely strong action on beta receptors
formational state. In each organ, the resulting effects
but essentially no action on alpha receptors.
Chapter 60
The Autonomic Nervous System and the Adrenal Medulla
753
Focusing of the lens is controlled almost entirely by
Table 60-1
the parasympathetic nervous system. The lens is nor-
Adrenergic Receptors and Function
mally held in a flattened state by intrinsic elastic tension
of its radial ligaments. Parasympathetic excitation con-
tracts the ciliary muscle, which is a ringlike body of
Alpha Receptor
Beta Receptor
smooth muscle fibers that encircles the outside ends
Vasoconstriction
Vasodilation (b2)
of the lens radial ligaments. This contraction releases
Iris dilation
Cardioacceleration (b1)
the tension on the ligaments and allows the lens to
Intestinal relaxation
Increased myocardial
become more convex, causing the eye to focus on
strength (b1)
objects near at hand. The detailed focusing mechanism
Intestinal sphincter contraction
Intestinal relaxation (b2)
is discussed in Chapters 49 and 51 in relation to func-
Uterus relaxation (b2)
tion of the eyes.
Pilomotor contraction
Bronchodilation (b2)
Bladder sphincter contraction
Calorigenesis (b2)
Glycogenolysis (b2)
Glands of the Body. The nasal, lacrimal, salivary, and many
Lipolysis (b1)
gastrointestinal glands are strongly stimulated by the
Bladder wall relaxation (b2)
parasympathetic nervous system, usually resulting in
copious quantities of watery secretion. The glands of
the alimentary tract most strongly stimulated by the
parasympathetics are those of the upper tract, especially
those of the mouth and stomach. On the other hand, the
glands of the small and large intestines are controlled
Excitatory and Inhibitory
principally by local factors in the intestinal tract itself
Actions of Sympathetic and
and by the intestinal enteric nervous system and much
Parasympathetic Stimulation
less by the autonomic nerves.
Sympathetic stimulation has a direct effect on most
Table 60-2 lists the effects on different visceral func-
alimentary gland cells to cause formation of a concen-
tions of the body caused by stimulating either the
trated secretion that contains high percentages of
enzymes and mucus. But it also causes vasoconstriction
parasympathetic nerves or the sympathetic nerves.
of the blood vessels that supply the glands and in this
From this table, it can be seen again that sympathetic
way sometimes reduces their rates of secretion.
stimulation causes excitatory effects in some organs but
The sweat glands secrete large quantities of sweat
inhibitory effects in others. Likewise, parasympathetic
when the sympathetic nerves are stimulated, but no
stimulation causes excitation in some but inhibition in
effect is caused by stimulating the parasympathetic
others. Also, when sympathetic stimulation excites a
nerves. However, the sympathetic fibers to most sweat
particular organ, parasympathetic stimulation some-
glands are cholinergic
(except for a few adrenergic
times inhibits it, demonstrating that the two systems
fibers to the palms and soles), in contrast to almost all
occasionally act reciprocally to each other. But most
other sympathetic fibers, which are adrenergic. Further-
organs are dominantly controlled by one or the other
more, the sweat glands are stimulated primarily by
centers in the hypothalamus that are usually considered
of the two systems.
to be parasympathetic centers. Therefore, sweating
There is no generalization one can use to explain
could be called a parasympathetic function, even though
whether sympathetic or parasympathetic stimulation
it is controlled by nerve fibers that anatomically are dis-
will cause excitation or inhibition of a particular organ.
tributed through the sympathetic nervous system.
Therefore, to understand sympathetic and parasympa-
The apocrine glands in the axillae secrete a thick,
thetic function, one must learn all the separate func-
odoriferous secretion as a result of sympathetic stimu-
tions of these two nervous systems on each organ, as
lation, but they do not respond to parasympathetic stim-
listed in Table 60-2. Some of these functions need to
ulation. This secretion actually functions as a lubricant
be clarified in still greater detail, as follows.
to allow easy sliding motion of the inside surfaces under
the shoulder joint. The apocrine glands, despite their
close embryological relation to sweat glands, are acti-
Effects of Sympathetic and
vated by adrenergic fibers rather than by cholinergic
Parasympathetic Stimulation
fibers and are also controlled by the sympathetic centers
of the central nervous system rather than by the
on Specific Organs
parasympathetic centers.
Eyes. Two functions of the eyes are controlled by the
autonomic nervous system. They are (1) the pupillary
Intramural Nerve Plexus of the Gastrointestinal System. The
opening and (2) the focus of the lens.
gastrointestinal system has its own intrinsic set of nerves
Sympathetic stimulation contracts the meridional
known as the intramural plexus or the intestinal enteric
fibers of the iris that dilate the pupil, whereas parasym-
nervous system, located in the walls of the gut. Also, both
pathetic stimulation contracts the circular muscle of the
parasympathetic and sympathetic stimulation originat-
iris to constrict the pupil.
ing in the brain can affect gastrointestinal activity
The parasympathetics that control the pupil are
mainly by increasing or decreasing specific actions in
reflexly stimulated when excess light enters the eyes,
the gastrointestinal intramural plexus. Parasympathetic
which is explained in Chapter 51; this reflex reduces the
stimulation, in general, increases overall degree of activ-
pupillary opening and decreases the amount of light
ity of the gastrointestinal tract by promoting peristalsis
that strikes the retina. Conversely, the sympathetics
and relaxing the sphincters, thus allowing rapid propul-
become stimulated during periods of excitement and
sion of contents along the tract. This propulsive effect
increase pupillary opening at these times.
is associated with simultaneous increases in rates of
754
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Table 60-2
Autonomic Effects on Various Organs of the Body
Organ
Effect of Sympathetic Stimulation
Effect of Parasympathetic Stimulation
Eye
Pupil
Dilated
Constricted
Ciliary muscle
Slight relaxation (far vision)
Constricted (near vision)
Glands
Vasoconstriction and slight secretion
Stimulation of copious secretion (containing many enzymes for
Nasal
enzyme-secreting glands)
Lacrimal
Parotid
Submandibular
Gastric
Pancreatic
Sweat glands
Copious sweating (cholinergic)
Sweating on palms of hands
Apocrine glands
Thick, odoriferous secretion
None
Blood vessels
Most often constricted
Most often little or no effect
Heart
Muscle
Increased rate
Slowed rate
Increased force of contraction
Decreased force of contraction (especially of atria)
Coronaries
Dilated (b2); constricted (a)
Dilated
Lungs
Bronchi
Dilated
Constricted
Blood vessels
Mildly constricted
? Dilated
Gut
Lumen
Decreased peristalsis and tone
Increased peristalsis and tone
Sphincter
Increased tone (most times)
Relaxed (most times)
Liver
Glucose released
Slight glycogen synthesis
Gallbladder and bile ducts
Relaxed
Contracted
Kidney
Decreased output and renin secretion
None
Bladder
Detrusor
Relaxed (slight)
Contracted
Trigone
Contracted
Relaxed
Penis
Ejaculation
Erection
Systemic arterioles
Abdominal viscera
Constricted
None
Muscle
Constricted (adrenergic a)
None
Dilated (adrenergic b2)
Dilated (cholinergic)
Skin
Constricted
None
Blood
Coagulation
Increased
None
Glucose
Increased
None
Lipids
Increased
None
Basal metabolism
Increased up to 100%
None
Adrenal medullary secretion
Increased
None
Mental activity
Increased
None
Piloerector muscles
Contracted
None
Skeletal muscle
Increased glycogenolysis
None
Increased strength
Fat cells
Lipolysis
None
secretion by many of the gastrointestinal glands,
thetic stimulation increases the effectiveness of the
described earlier.
heart as a pump, as required during heavy exercise,
Normal function of the gastrointestinal tract is not
whereas parasympathetic stimulation decreases heart
very dependent on sympathetic stimulation. However,
pumping, allowing the heart to rest between bouts of
strong sympathetic stimulation inhibits peristalsis and
strenuous activity.
increases the tone of the sphincters. The net result is
greatly slowed propulsion of food through the tract and
Systemic Blood Vessels. Most systemic blood vessels, espe-
sometimes decreased secretion as well—even to the
cially those of the abdominal viscera and skin of the
extent of sometimes causing constipation.
limbs, are constricted by sympathetic stimulation.
Parasympathetic stimulation has almost no effects on
Heart. In general, sympathetic stimulation increases the
most blood vessels except to dilate vessels in certain
overall activity of the heart. This is accomplished by
restricted areas, such as in the blush area of the face.
increasing both the rate and force of heart contraction.
Under some conditions, the beta function of the sym-
Parasympathetic stimulation causes mainly opposite
pathetics causes vascular dilation instead of the usual
effects—decreased heart rate and strength of contrac-
sympathetic vascular constriction, but this occurs rarely
tion. To express these effects in another way, sympa-
except after drugs have paralyzed the sympathetic alpha
Chapter 60
The Autonomic Nervous System and the Adrenal Medulla
755
vasoconstrictor effects, which, in blood vessels, are
greater effect in stimulating the beta receptors, has a
usually far dominant over the beta effects.
greater effect on cardiac stimulation than does nor-
epinephrine. Second, epinephrine causes only weak
Effect of Sympathetic and Parasympathetic Stimulation on Arter-
constriction of the blood vessels in the muscles, in com-
ial Pressure. The arterial pressure is determined by two
parison with much stronger constriction caused by
factors: propulsion of blood by the heart and resistance
norepinephrine. Because the muscle vessels represent
to flow of blood through the peripheral blood vessels.
a major segment of the vessels of the body, this differ-
Sympathetic stimulation increases both propulsion by
ence is of special importance because norepinephrine
the heart and resistance to flow, which usually causes a
marked acute increase in arterial pressure but often very
greatly increases the total peripheral resistance and
little change in long-term pressure unless the sympa-
elevates arterial pressure, whereas epinephrine raises
thetics stimulate the kidneys to retain salt and water at
the arterial pressure to a lesser extent but increases the
the same time.
cardiac output more.
Conversely, moderate parasympathetic stimulation
A third difference between the actions of epineph-
via the vagal nerves decreases pumping by the heart but
rine and norepinephrine relates to their effects on
has virtually no effect on vascular peripheral resistance.
tissue metabolism. Epinephrine has 5 to 10 times as
Therefore, the usual effect is a slight decrease in arte-
great a metabolic effect as norepinephrine. Indeed, the
rial pressure. But very strong vagal parasympathetic
epinephrine secreted by the adrenal medullae can
stimulation can almost stop or occasionally actually stop
increase the metabolic rate of the whole body often to
the heart entirely for a few seconds and cause tempo-
rary loss of all or most arterial pressure.
as much as 100 per cent above normal, in this way
increasing the activity and excitability of the body. It
Effects of Sympathetic and Parasympathetic Stimulation on Other
also increases the rates of other metabolic activities,
Functions of the Body. Because of the great importance of
such as glycogenolysis in the liver and muscle, and
the sympathetic and parasympathetic control systems,
glucose release into the blood.
they are discussed many times in this text in relation
In summary, stimulation of the adrenal medullae
to multiple body functions. In general, most of the
causes release of the hormones epinephrine and nor-
entodermal structures, such as the ducts of the liver,
epinephrine, which together have almost the same
gallbladder, ureter, urinary bladder, and bronchi, are
effects throughout the body as direct sympathetic
inhibited by sympathetic stimulation but excited by
stimulation, except that the effects are greatly pro-
parasympathetic stimulation. Sympathetic stimulation
also has multiple metabolic effects such as release of
longed, lasting 2 to 4 minutes after the stimulation is
glucose from the liver, increase in blood glucose con-
over.
centration, increase in glycogenolysis in both liver and
muscle, increase in skeletal muscle strength, increase in
Value of the Adrenal Medullae to the Function of the Sympa-
basal metabolic rate, and increase in mental activity.
thetic Nervous System. Epinephrine and norepinephrine
Finally, the sympathetics and parasympathetics are
are almost always released by the adrenal medullae at
involved in execution of the male and female sexual
the same time that the different organs are stimulated
acts, as explained in Chapters 80 and 81.
directly by generalized sympathetic activation. There-
fore, the organs are actually stimulated in two ways:
Function of the Adrenal Medullae
directly by the sympathetic nerves and indirectly by
the adrenal medullary hormones. The two means
Stimulation of the sympathetic nerves to the adrenal
of stimulation support each other, and either can, in
medullae causes large quantities of epinephrine and
most instances, substitute for the other. For instance,
norepinephrine to be released into the circulating
destruction of the direct sympathetic pathways to
blood, and these two hormones in turn are carried in
the different body organs does not abrogate sympa-
the blood to all tissues of the body. On the average,
thetic excitation of the organs because norepinephrine
about 80 per cent of the secretion is epinephrine and
and epinephrine are still released into the circulating
20 per cent is norepinephrine, although the relative
blood and indirectly cause stimulation. Likewise,
proportions can change considerably under different
loss of the two adrenal medullae usually has little
physiologic conditions.
effect on the operation of the sympathetic nervous
The circulating epinephrine and norepinephrine
system because the direct pathways can still perform
have almost the same effects on the different organs
almost all the necessary duties. Thus, the dual mecha-
as the effects caused by direct sympathetic stimulation,
nism of sympathetic stimulation provides a safety
except that the effects last 5 to 10 times as long because
factor, one mechanism substituting for the other if it is
both of these hormones are removed from the blood
missing.
slowly over a period of 2 to 4 minutes.
Another important value of the adrenal medullae
The circulating norepinephrine causes constriction
is the capability of epinephrine and norepinephrine
of essentially all the blood vessels of the body; it also
to stimulate structures of the body that are not
causes increased activity of the heart, inhibition of the
innervated by direct sympathetic fibers. For instance,
gastrointestinal tract, dilation of the pupils of the eyes,
the metabolic rate of every cell of the body is increased
and so forth.
by these hormones, especially by epinephrine,
Epinephrine causes almost the same effects as those
even though only a small proportion of all the cells
caused by norepinephrine, but the effects differ in the
in the body are innervated directly by sympathetic
following respects: First, epinephrine, because of its
fibers.
756
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Relation of Stimulus Rate
Effect of Loss of Sympathetic or Parasympathetic Tone After
Denervation. Immediately after a sympathetic or
to Degree of Sympathetic and
parasympathetic nerve is cut, the innervated organ
Parasympathetic Effect
loses its sympathetic or parasympathetic tone. In the
case of the blood vessels, for instance, cutting the sym-
A special difference between the autonomic nervous
pathetic nerves results within 5 to 30 seconds in almost
system and the skeletal nervous system is that only a
maximal vasodilation. However, over minutes, hours,
low frequency of stimulation is required for full acti-
days, or weeks, intrinsic tone in the smooth muscle of
vation of autonomic effectors. In general, only one
the vessels increases—that is, increased tone caused by
nerve impulse every few seconds suffices to maintain
increased smooth muscle contractile force that is not
normal sympathetic or parasympathetic effect, and full
the result of sympathetic stimulation but of chemical
activation occurs when the nerve fibers discharge 10 to
adaptations in the smooth muscle fibers themselves.
20 times per second. This compares with full activation
This intrinsic tone eventually restores almost normal
in the skeletal nervous system at 50 to 500 or more
vasoconstriction.
impulses per second.
Essentially the same effects occur in most other
effector organs whenever sympathetic or parasympa-
thetic tone is lost. That is, intrinsic compensation soon
Sympathetic and
develops to return the function of the organ almost to
Parasympathetic “Tone”
its normal basal level. However, in the parasympa-
thetic system, the compensation sometimes requires
Normally, the sympathetic and parasympathetic
many months. For instance, loss of parasympathetic
systems are continually active, and the basal rates of
tone to the heart after cardiac vagotomy increases the
activity are known, respectively, as sympathetic tone
heart rate to 160 beats per minute in a dog, and this
and parasympathetic tone.
will still be partially elevated 6 months later.
The value of tone is that it allows a single nervous
system both to increase and to decrease the activity of
Denervation Supersensitivity of
a stimulated organ. For instance, sympathetic tone nor-
Sympathetic and Parasympathetic
mally keeps almost all the systemic arterioles con-
Organs After Denervation
stricted to about one half their maximum diameter.
By increasing the degree of sympathetic stimulation
During the first week or so after a sympathetic or
above normal, these vessels can be constricted even
parasympathetic nerve is destroyed, the innervated
more; conversely, by decreasing the stimulation below
organ becomes more sensitive to injected norepineph-
normal, the arterioles can be dilated. If it were not for
rine or acetylcholine, respectively. This effect is demon-
the continual background sympathetic tone, the sym-
strated in Figure
60-4, showing blood flow in the
pathetic system could cause only vasoconstriction,
forearm before removal of the sympathetics to be about
never vasodilation.
200 ml/min; a test dose of norepinephrine causes only a
slight depression in flow lasting a minute or so. Then the
Another interesting example of tone is the back-
stellate ganglion is removed, and normal sympathetic
ground
“tone” of the parasympathetics in the
tone is lost. At first, the blood flow rises markedly
gastrointestinal tract. Surgical removal of the parasym-
because of the lost vascular tone, but over a period of
pathetic supply to most of the gut by cutting the vagus
days to weeks the blood flow returns much of the way
nerves can cause serious and prolonged gastric and
back toward normal because of progressive increase in
intestinal “atony” with resulting blockage of much of
the normal gastrointestinal propulsion and consequent
serious constipation, thus demonstrating that
parasympathetic tone to the gut is normally very much
required. This tone can be decreased by the brain,
400
thereby inhibiting gastrointestinal motility, or it can be
increased, thereby promoting increased gastrointesti-
nal activity.
Effect of same
Normal
200
test dose of
Stellate
norepinephrine
Tone Caused by Basal Secretion of Epinephrine and Norepi-
ganglionectomy
nephrine by the Adrenal Medullae. The normal resting
Effect of test dose
rate of secretion by the adrenal medullae is about
of norepinephrine
0.2 mg/kg/min of epinephrine and about 0.05 mg/kg/min
0
0
1
2
3
4
5
6
of norepinephrine. These quantities are considerable—
Weeks
indeed, enough to maintain the blood pressure almost
up to normal even if all direct sympathetic pathways to
the cardiovascular system are removed. Therefore, it is
obvious that much of the overall tone of the sympa-
Figure 60-4
thetic nervous system results from basal secretion of
Effect of sympathectomy on blood flow in the arm, and effect of a
epinephrine and norepinephrine in addition to the tone
test dose of norepinephrine before and after sympathectomy,
resulting from direct sympathetic stimulation.
showing supersensitization of the vasculature to norepinephrine.
Chapter 60
The Autonomic Nervous System and the Adrenal Medulla
757
intrinsic tone of the vascular musculature itself, thus
bladder and relaxation of the urinary sphincters,
partially compensating for the loss of sympathetic tone.
thereby promoting micturition.
Then, another test dose of norepinephrine is adminis-
Also important are the sexual reflexes, which are ini-
tered, and the blood flow decreases much more than
tiated both by psychic stimuli from the brain and by
before, demonstrating that the blood vessels have
stimuli from the sexual organs. Impulses from these
become about two to four times as responsive to nor-
sources converge on the sacral cord and, in the male,
epinephrine as previously. This phenomenon is called
result first in erection, mainly a parasympathetic func-
denervation supersensitivity. It occurs in both sympa-
tion, and then ejaculation, partially a sympathetic
thetic and parasympathetic organs but to far greater
function.
extent in some organs than in others, occasionally
Other autonomic control functions include reflex
increasing the response more than 10-fold.
contributions to the regulation of pancreatic secretion,
gallbladder emptying, kidney excretion of urine, sweat-
Mechanism of Denervation Supersensitivity. The cause of
ing, blood glucose concentration, and many other vis-
denervation supersensitivity is only partially known.
ceral functions, all of which are discussed in detail at
Part of the answer is that the number of receptors in
other points in this text.
the postsynaptic membranes of the effector cells
increases—sometimes manyfold—when norepineph-
rine or acetylcholine is no longer released at the
Stimulation of Discrete
synapses, a process called “up-regulation” of the recep-
tors. Therefore, when a dose of the hormone is now
Organs in Some Instances and
injected into the circulating blood, the effector reaction
Mass Stimulation in Other
is vastly enhanced.
Instances by the Sympathetic
and Parasympathetic Systems
Autonomic Reflexes
Sympathetic System Often Responds by Mass Discharge. In
Many visceral functions of the body are regulated by
many instances, almost all portions of the sympathetic
autonomic reflexes. Throughout this text, the functions
nervous system discharge simultaneously as a com-
of these reflexes are discussed in relation to individual
plete unit, a phenomenon called mass discharge. This
organ systems; to illustrate their importance, a few are
frequently occurs when the hypothalamus is activated
presented here briefly.
by fright or fear or severe pain. The result is a wide-
Cardiovascular Autonomic Reflexes. Several reflexes in the
spread reaction throughout the body called the alarm
cardiovascular system help to control especially the
or stress response, which we shall discuss shortly.
arterial blood pressure and the heart rate. One of these
At other times, activation occurs in isolated portions
is the baroreceptor reflex, which is described in Chapter
of the sympathetic nervous system. The most impor-
18 along with other cardiovascular reflexes. Briefly,
tant of these are the following: (1) During the process
stretch receptors called baroreceptors are located in the
of heat regulation, the sympathetics control sweating
walls of several major arteries, including especially the
and blood flow in the skin without affecting other
internal carotid arteries and the arch of the aorta. When
organs innervated by the sympathetics.
(2) Many
these become stretched by high pressure, signals are
“local reflexes” involving sensory afferent fibers travel
transmitted to the brain stem, where they inhibit the
centrally in the peripheral nerves to the sympathetic
sympathetic impulses to the heart and blood vessels and
excite the parasympathetics; this allows the arterial
ganglia and spinal cord and cause highly localized
pressure to fall back toward normal.
reflex responses. For instance, heating a local skin area
causes local vasodilation and enhanced local sweating,
Gastrointestinal Autonomic Reflexes. The uppermost part of
whereas cooling causes opposite effects. (3) Many of
the gastrointestinal tract and the rectum are controlled
the sympathetic reflexes that control gastrointestinal
principally by autonomic reflexes. For instance, the
functions operate by way of nerve pathways that do
smell of appetizing food or the presence of food in
not even enter the spinal cord, merely passing from the
the mouth initiates signals from the nose and mouth
gut mainly to the paravertebral ganglia, and then back
to the vagal, glossopharyngeal, and salivatory nuclei
to the gut through sympathetic nerves to control
of the brain stem. These in turn transmit signals through
motor or secretory activity.
the parasympathetic nerves to the secretory glands of
the mouth and stomach, causing secretion of digestive
juices sometimes even before food enters the mouth.
Parasympathetic System Usually Causes Specific Localized
When fecal matter fills the rectum at the other end
Responses. In contrast to the common mass discharge
of the alimentary canal, sensory impulses initiated by
response of the sympathetic system, control functions
stretching the rectum are sent to the sacral portion of
by the parasympathetic system are much more likely
the spinal cord, and a reflex signal is transmitted back
to be highly specific. For instance, parasympathetic
through the sacral parasympathetics to the distal parts
cardiovascular reflexes usually act only on the heart to
of the colon; these result in strong peristaltic contrac-
increase or decrease its rate of beating. Likewise, other
tions that cause defecation.
parasympathetic reflexes cause secretion mainly by
the mouth glands, or in other instances secretion is
Other Autonomic Reflexes. Emptying of the urinary bladder
is controlled in the same way as emptying the rectum;
mainly by the stomach glands. Finally, the rectal emp-
stretching of the bladder sends impulses to the sacral
tying reflex does not affect other parts of the bowel to
cord, and this in turn causes reflex contraction of the
a major extent.
758
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Yet there is often association between closely allied
parasympathetic functions. For instance, although sali-
vary secretion can occur independently of gastric
secretion, these two also often occur together, and
Heat control
pancreatic secretion frequently occurs at the same
Sympathetic
Parasympathetic
time. Also, the rectal emptying reflex often initiates a
urinary bladder emptying reflex, resulting in simulta-
Water
neous emptying of both the bladder and the rectum.
balance
Conversely, the bladder emptying reflex can help ini-
Feeding
tiate rectal emptying.
control
Urinary bladder control
Hypothalamus
“Alarm” or “Stress” Response of the
Pneumotaxic center
Adenohypophysis
Sympathetic Nervous System
Cardiac acceleration
Mamillary body
and vasoconstriction
When large portions of the sympathetic nervous
Pons
Cardiac slowing
system discharge at the same time—that is, a mass dis-
charge—this increases in many ways the ability of the
Respiratory center
Medulla
body to perform vigorous muscle activity. Let us sum-
marize these ways:
1. Increased arterial pressure
2. Increased blood flow to active muscles concurrent
Figure 60-5
with decreased blood flow to organs such as the
Autonomic control areas in the brain stem and hypothalamus.
gastrointestinal tract and the kidneys that are not
needed for rapid motor activity
3. Increased rates of cellular metabolism throughout
the body
4. Increased blood glucose concentration
many special nuclei (Figure 60-5), control different
5. Increased glycolysis in the liver and in muscle
autonomic functions such as arterial pressure, heart
6. Increased muscle strength
rate, glandular secretion in the gastrointestinal tract,
7. Increased mental activity
gastrointestinal peristalsis, and degree of contraction
8. Increased rate of blood coagulation
of the urinary bladder. Control of each of these is dis-
The sum of these effects permits a person to
cussed at appropriate points in this text. Suffice it to
perform far more strenuous physical activity than
point out here that the most important factors con-
would otherwise be possible. Because either mental or
trolled in the brain stem are arterial pressure, heart rate,
physical stress can excite the sympathetic system, it is
and respiratory rate. Indeed, transection of the brain
frequently said that the purpose of the sympathetic
stem above the midpontine level allows basal control
system is to provide extra activation of the body in
of arterial pressure to continue as before but prevents
states of stress: this is called the sympathetic stress
its modulation by higher nervous centers such as the
response.
hypothalamus. Conversely, transection immediately
The sympathetic system is especially strongly acti-
below the medulla causes the arterial pressure to fall
vated in many emotional states. For instance, in the
to less than one-half normal.
state of rage, which is elicited to a great extent by stim-
Closely associated with the cardiovascular regula-
ulating the hypothalamus, signals are transmitted
tory centers in the brain stem are the medullary and
downward through the reticular formation of the brain
pontine centers for regulation of respiration, which are
stem and into the spinal cord to cause massive sym-
discussed in Chapter 41. Although this is not consid-
pathetic discharge; most aforementioned sympathetic
ered to be an autonomic function, it is one of the invol-
events ensue immediately. This is called the sympa-
untary functions of the body.
thetic alarm reaction. It is also called the fight or flight
reaction because an animal in this state decides almost
Control of Brain Stem Autonomic Centers by Higher Areas.
instantly whether to stand and fight or to run. In either
Signals from the hypothalamus and even from the
event, the sympathetic alarm reaction makes the
cerebrum can affect the activities of almost all the
animal’s subsequent activities vigorous.
brain stem autonomic control centers. For instance,
stimulation in appropriate areas mainly of the poste-
rior hypothalamus can activate the medullary cardio-
Medullary, Pontine, and
vascular control centers strongly enough to increase
Mesencephalic Control of the
arterial pressure to more than twice normal. Likewise,
Autonomic Nervous System
other hypothalamic centers control body temperature,
increase or decrease salivation and gastrointes-
Many neuronal areas in the brain stem reticular sub-
tinal activity, and cause bladder emptying. To some
stance and along the course of the tractus solitarius of
extent, therefore, the autonomic centers in the brain
the medulla, pons, and mesencephalon, as well as in
stem act as relay stations for control activities
Chapter 60
The Autonomic Nervous System and the Adrenal Medulla
759
initiated at higher levels of the brain, especially in the
autonomic ganglia. They are discussed in a later
hypothalamus.
section, but an important drug for blockade of both
In Chapters 58 and 59, it is pointed out also that
sympathetic and parasympathetic transmission
through the ganglia is hexamethonium.
many of our behavioral responses are mediated
through (1) the hypothalamus, (2) the reticular areas
of the brain stem, and (3) the autonomic nervous
Drugs That Act on Cholinergic
system. Indeed, some higher areas of the brain can
Effector Organs
alter function of the whole autonomic nervous system
or of portions of it strongly enough to cause severe
Parasympathomimetic Drugs
(Cholinergic Drugs). Acetyl-
autonomic-induced disease such as peptic ulcer of the
choline injected intravenously usually does not cause
stomach or duodenum, constipation, heart palpitation,
exactly the same effects throughout the body as
or even heart attack.
parasympathetic stimulation because most of the acetyl-
choline is destroyed by cholinesterase in the blood and
body fluids before it can reach all the effector organs.
Yet a number of other drugs that are not so rapidly
Pharmacology of the
destroyed can produce typical widespread parasympa-
Autonomic Nervous System
thetic effects, and they are called parasympathomimetic
drugs.
Drugs That Act on Adrenergic
Two commonly used parasympathomimetic drugs are
Effector Organs—
pilocarpine and methacholine. They act directly on the
muscarinic type of cholinergic receptors.
Sympathomimetic Drugs
From the foregoing discussion, it is obvious that intra-
Drugs That Have a Parasympathetic Potentiating Effect—Anti-
venous injection of norepinephrine causes essentially
cholinesterase Drugs. Some drugs do not have a direct
the same effects throughout the body as sympathetic
effect on parasympathetic effector organs but do poten-
stimulation. Therefore, norepinephrine is called a
tiate the effects of the naturally secreted acetylcholine
sympathomimetic or adrenergic drug. Epinephrine and
at the parasympathetic endings. They are the same drugs
methoxamine are also sympathomimetic drugs, and
as those discussed in Chapter 7 that potentiate the effect
there are many others. They differ from one another in
of acetylcholine at the neuromuscular junction. They
the degree to which they stimulate different sympa-
include neostigmine, pyridostigmine, and ambenonium.
thetic effector organs and in their duration of action.
These drugs inhibit acetylcholinesterase, thus prevent-
Norepinephrine and epinephrine have actions as short
ing rapid destruction of the acetylcholine liberated at
as 1 to 2 minutes, whereas the actions of some other
parasympathetic nerve endings. As a consequence, the
commonly used sympathomimetic drugs last for 30
quantity of acetylcholine increases with successive
minutes to 2 hours.
stimuli, and the degree of action also increases.
Important drugs that stimulate specific adrenergic
receptors but not others are phenylephrine
(alpha
Drugs That Block Cholinergic Activity at Effector Organs—
receptors), isoproterenol (beta receptors), and albuterol
Antimuscarinic Drugs. Atropine and similar drugs, such as
(only beta
receptors).
homatropine and scopolamine, block the action of
2
acetylcholine on the muscarinic type of cholinergic effec-
Drugs That Cause Release of Norepinephrine from Nerve Endings.
tor organs. These drugs do not affect the nicotinic action
Certain drugs have an indirect sympathomimetic action
of acetylcholine on the postganglionic neurons or on
instead of directly exciting adrenergic effector organs.
skeletal muscle.
These drugs include ephedrine, tyramine, and ampheta-
mine. Their effect is to cause release of norepinephrine
from its storage vesicles in the sympathetic nerve
Drugs That Stimulate or Block
endings. The released norepinephrine in turn causes the
Sympathetic and Parasympathetic
sympathetic effects.
Postganglionic Neurons
Drugs That Block Adrenergic Activity. Adrenergic activity
Drugs That Stimulate Autonomic Postganglionic Neurons. The
can be blocked at several points in the stimulatory
preganglionic neurons of both the parasympathetic and
process, as follows:
the sympathetic nervous systems secrete acetylcholine
1. The synthesis and storage of norepinephrine in the
at their endings, and this acetylcholine in turn stimulates
sympathetic nerve endings can be prevented. The
the postganglionic neurons. Furthermore, injected
best known drug that causes this effect is reserpine.
acetylcholine can also stimulate the postganglionic
2. Release of norepinephrine from the sympathetic
neurons of both systems, thereby causing at the same
endings can be blocked. This is caused by
time both sympathetic and parasympathetic effects
guanethidine.
throughout the body.
3. The sympathetic alpha receptors can be blocked.
Nicotine is another drug that can stimulate postgan-
Two drugs that cause this effect are
glionic neurons in the same manner as acetylcholine
phenoxybenzamine and phentolamine.
because the membranes of these neurons all contain the
4. The sympathetic beta receptors can be blocked.
nicotinic type of acetylcholine receptor. Therefore, drugs
A drug that blocks beta1 and beta2 receptors is
that cause autonomic effects by stimulating postgan-
propranolol. One that blocks mainly beta1
glionic neurons are called nicotinic drugs. Some other
receptors is metoprolol.
drugs, such as methacholine, have both nicotinic and
5. Sympathetic activity can be blocked by drugs that
muscarinic actions, whereas pilocarpine has only mus-
block transmission of nerve impulses through the
carinic actions.
760
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Nicotine excites both the sympathetic and parasym-
DiBona GF, Kopp UC: Neural control of renal function.
pathetic postganglionic neurons at the same time,
Physiol Rev 77:75, 1997.
resulting in strong sympathetic vasoconstriction in the
Gibbins IL, Jobling P, Morris JL: Functional organization
abdominal organs and limbs but at the same time result-
of peripheral vasomotor pathways. Acta Physiol Scand
ing in parasympathetic effects such as increased gas-
177:237, 2003.
trointestinal activity and, sometimes, slowing of the
Goldstein DS: Catecholamines and stress. Endocr Regul
heart.
37:69, 2003.
Goldstein DS, Robertson D, Esler M, et al: Dysautonomias:
Ganglionic Blocking Drugs. Many important drugs block
clinical disorders of the autonomic nervous system. Ann
impulse transmission from the autonomic preganglionic
Intern Med 137:753, 2002.
neurons to the postganglionic neurons, including
Hall JE, Hildebrandt DA, Kuo J: Obesity hypertension: role
tetraethyl ammonium ion, hexamethonium ion, and pen-
of leptin and sympathetic nervous system. Am J Hyper-
tolinium. These drugs block acetylcholine stimulation of
tens 14:103S, 2001.
the postganglionic neurons in both the sympathetic and
Hirst GD, Ward SM: Interstitial cells: involvement in rhyth-
the parasympathetic systems simultaneously. They are
micity and neural control of gut smooth muscle. J Physiol
often used for blocking sympathetic activity but seldom
550:337, 2003.
for blocking parasympathetic activity because their
Koch WJ, Lefkowitz RJ, Rockman HA: Functional conse-
effects of sympathetic blockade usually far overshadow
quences of altering myocardial adrenergic receptor sig-
the effects of parasympathetic blockade. The ganglionic
naling. Annu Rev Physiol 62:237, 2000.
blocking drugs especially can reduce the arterial pres-
Lefkowitz RJ, Rockman HA, Koch WJ: Catecholamines,
sure in many patients with hypertension, but these drugs
cardiac beta-adrenergic receptors, and heart failure.
are not very useful clinically because their effects are
Circulation 101:1634, 2000.
difficult to control.
Lohmeier TE: The sympathetic nervous system and long-
term blood pressure regulation. Am J Hypertens 14:147S,
2001.
References
Saper CB: The central autonomic nervous system: conscious
visceral perception and autonomic pattern generation.
Annu Rev Neurosci 25:433, 2002.
Andresen MC, Doyle MW, Jin YH, Bailey TW: Cellular
Taylor EW, Jordan D, Coote JH: Central control of the car-
mechanisms of baroreceptor integration at the nucleus
diovascular and respiratory systems and their interactions
tractus solitarius. Ann N Y Acad Sci 940:132, 2001
in vertebrates. Physiol Rev 79:855, 1999.
Chang HY, Mashimo H, Goyal RK: Musings on the wan-
Wanamaker CP, Christianson JC, Green WN: Regulation of
derer: what’s new in our understanding of vago-vagal
nicotinic acetylcholine receptor assembly. Ann N Y Acad
reflex? IV. Current concepts of vagal efferent projections
Sci 998:66, 2003.
to the gut. Am J Physiol Gastrointest Liver Physiol
Wess J: Novel insights into muscarinic acetylcholine recep-
284:G357, 2003.
tor function using gene targeting technology. Trends Phar-
Dajas-Bailador F, Wonnacott S: Nicotinic acetylcholine
macol Sci 24:414, 2003.
receptors and the regulation of neuronal signalling. Trends
Pharmacol Sci 25:317, 2004.
Dampney RA, Horiuchi J, Tagawa T, et al: Medullary and
supramedullary mechanisms regulating sympathetic vaso-
motor tone. Acta Physiol Scand 177:209, 2003.
C H A P T E R
6
1
Cerebral Blood Flow,
Cerebrospinal Fluid, and Brain
Metabolism
Thus far, we have discussed the function of the brain as
if it were independent of its blood flow, its metabolism,
and its fluids. However, this is far from true because
abnormalities of any of these can profoundly affect
brain function. For instance, total cessation of blood
flow to the brain causes unconsciousness within 5 to 10
seconds. This occurs because lack of oxygen delivery to
the brain cells shuts down most metabolism in these
cells. Also, on a longer time scale, abnormalities of the
cerebrospinal fluid, either its composition or its fluid pressure, can have equally
severe effects on brain function.
Cerebral Blood Flow
Normal Rate of Cerebral Blood Flow
Normal blood flow through the brain of the adult person averages 50 to 65 milli-
liters per 100 grams of brain tissue per minute. For the entire brain, this amounts to
750 to 900 ml/min, or 15 per cent of the resting cardiac output.
Regulation of Cerebral Blood Flow
As in most other vascular areas of the body, cerebral blood flow is highly related to
metabolism of the tissue. At least three metabolic factors have potent effects in con-
trolling cerebral blood flow: (1) carbon dioxide concentration, (2) hydrogen ion con-
centration, and (3) oxygen concentration.
Increase of Cerebral Blood Flow in Response to Excess Carbon Dioxide or Excess Hydrogen Ion
Concentration. An increase in carbon dioxide concentration in the arterial blood per-
fusing the brain greatly increases cerebral blood flow. This is demonstrated in Figure
61-1, which shows that a 70 per cent increase in arterial PCO2 approximately doubles
cerebral blood flow.
Carbon dioxide is believed to increase cerebral blood flow by combining first with
water in the body fluids to form carbonic acid, with subsequent dissociation of this
acid to form hydrogen ions. The hydrogen ions then cause vasodilation of the cere-
bral vessels—the dilation being almost directly proportional to the increase in
hydrogen ion concentration up to a blood flow limit of about twice normal.
Any other substance that increases the acidity of the brain tissue, and therefore
increases hydrogen ion concentration, will likewise increase cerebral blood flow.
Such substances include lactic acid, pyruvic acid, and any other acidic material
formed during the course of tissue metabolism.
Importance of Cerebral Blood Flow Control by Carbon Dioxide and Hydrogen Ions. Increased
hydrogen ion concentration greatly depresses neuronal activity. Therefore, it is for-
tunate that an increase in hydrogen ion concentration also causes an increase in
blood flow, which in turn carries hydrogen ions, carbon dioxide, and other acid-
forming substances away from the brain tissues. Loss of carbon dioxide removes
carbonic acid from the tissues; this, along with removal of other acids, reduces the
hydrogen ion concentration back toward normal. Thus, this mechanism helps main-
tain a constant hydrogen ion concentration in the cerebral fluids and thereby helps
to maintain a normal, constant level of neuronal activity.
Oxygen Deficiency as a Regulator of Cerebral Blood Flow. Except during periods of intense
brain activity, the rate of utilization of oxygen by the brain tissue remains within
761
762
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
2.0
140
1.6
130
120
1.2
Light shining
110
in eyes
Normal
100
0.8
0
0.5
1.0
1.5
Minutes
0.4
0
20
40
60
80
100
Arterial Pco2
2
Figure 61-2
Increase in blood flow to the occipital regions of a cat’s brain when
Figure 61-1
light is shined into its eyes.
Relationship between arterial PCO2 and cerebral blood flow.
opposite side of the brain. Reading a book increases the
blood flow, especially in the visual areas of the occipital
narrow limits—almost exactly 3.5 (± 0.2) milliliters of
cortex and in the language perception areas of the tem-
oxygen per 100 grams of brain tissue per minute. If
poral cortex. This measuring procedure can also be used
blood flow to the brain ever becomes insufficient to
for localizing the origin of epileptic attacks because
supply this needed amount of oxygen, the oxygen defi-
local brain blood flow increases acutely and markedly
ciency mechanism for causing vasodilation immediately
at the focal point of each attack.
causes vasodilation, returning the brain blood flow and
Demonstrating the effect of local neuronal activity on
transport of oxygen to the cerebral tissues to near
cerebral blood flow, Figure 61-2 shows a typical increase
normal. Thus, this local blood flow regulatory mecha-
in occipital blood flow recorded in a cat’s brain when
nism is almost exactly the same in the brain as in coro-
intense light is shined into its eyes for one-half minute.
nary blood vessels, in skeletal muscle, and in most other
circulatory areas of the body.
Autoregulation of Cerebral Blood Flow When the Arterial Pressure
Experiments have shown that a decrease in cerebral
Changes. Cerebral blood flow is
“autoregulated”
tissue PO2 below about 30 mm Hg (normal value is 35 to
extremely well between arterial pressure limits of 60
40 mm Hg) immediately begins to increase cerebral
and 140 mm Hg. That is, mean arterial pressure can be
blood flow. This is fortuitous because brain function
decreased acutely to as low as 60 mm Hg or increased
becomes deranged at not much lower values of PO2,
to as high as 140 mm Hg without significant change in
especially so at PO2 levels below 20 mm Hg. Even coma
cerebral blood flow. And, in people who have hyper-
can result at these low levels. Thus, the oxygen mecha-
tension, autoregulation of cerebral blood flow occurs
nism for local regulation of cerebral blood flow is a very
even when the mean arterial pressure rises to as high as
important protective response against diminished cere-
160 to 180 mm Hg. This is demonstrated in Figure 61-3,
bral neuronal activity and, therefore, against derange-
which shows cerebral blood flow measured both in
ment of mental capability.
persons with normal blood pressure and in hypertensive
and hypotensive patients. Note the extreme constancy
Measurement of Cerebral Blood Flow, and Effect of Brain Activ-
of cerebral blood flow between the limits of 60 and
ity on the Flow. A method has been developed to record
180 mm Hg mean arterial pressure. But, if the arterial
blood flow in as many as 256 isolated segments of the
pressure falls below 60 mm Hg, cerebral blood flow then
human cerebral cortex simultaneously. To do this, a
does become severely decreased.
radioactive substance, such as radioactive xenon, is
injected into the carotid artery; then the radioactivity of
Role of the Sympathetic Nervous System in Controlling Cerebral
each segment of the cortex is recorded as the radioac-
Blood Flow. The cerebral circulatory system has strong
tive substance passes through the brain tissue. For this
sympathetic innervation that passes upward from the
purpose, 256 small radioactive scintillation detectors are
superior cervical sympathetic ganglia in the neck and
pressed against the surface of the cortex. The rapidity
then into the brain along with the cerebral arteries. This
of rise and decay of radioactivity in each tissue segment
innervation supplies both the large brain arteries and
is a direct measure of the rate of blood flow through that
the arteries that penetrate into the substance of the
segment.
brain. However, transection of the sympathetic nerves
Using this technique, it has become clear that blood
or mild to moderate stimulation of them usually causes
flow in each individual segment of the brain changes as
very little change in cerebral blood flow because the
much as 100 to 150 per cent within seconds in response
blood flow autoregulation mechanism can override the
to changes in local neuronal activity. For instance,
nervous effects.
simply making a fist of the hand causes an immedi-
When mean arterial pressure rises acutely to an
ate increase in blood flow in the motor cortex of the
exceptionally high level, such as during strenuous
Chapter 61
Cerebral Blood Flow, Cerebrospinal Fluid, and Brain Metabolism
763
Cerebral “Stroke” Occurs When
Cerebral Blood Vessels Are Blocked
Almost all elderly people have blockage of some small
60
arteries in the brain, and as many as 10 per cent even-
tually have enough blockage to cause serious distur-
bance of brain function, a condition called a “stroke.”
Most strokes are caused by arteriosclerotic plaques
40
that occur in one or more of the feeder arteries to the
brain. The plaques can activate the clotting mechanism
of the blood, causing a blood clot to occur and block
20
blood flow in the artery, thereby leading to acute loss of
brain function in a localized area.
In about one quarter of people who develop strokes,
high blood pressure makes one of the blood vessels
Hypotension
Hypertension
0
burst; hemorrhage then occurs, compressing the local
0
50
100
150
brain tissue and further compromising its functions. The
Mean arterial blood pressure (mm Hg)
neurological effects of a stroke are determined by the
brain area affected. One of the most common types of
stroke is blockage of the middle cerebral artery that sup-
plies the midportion of one brain hemisphere. For
instance, if the middle cerebral artery is blocked on the
Figure 61-3
left side of the brain, the person is likely to become
almost totally demented because of lost function in
Effect of differences in mean arterial pressure, from hypotensive
Wernicke’s speech comprehension area in the left cere-
to hypertensive level, on cerebral blood flow in different human
beings.
(Modified from Lassen NA: Cerebral blood flow and
bral hemisphere, and he or she also becomes unable to
oxygen consumption in man. Physiol Rev 39:183, 1959.)
speak words because of loss of Broca’s motor area for
word formation. In addition, loss of function of neural
motor control areas of the left hemisphere can create
spastic paralysis of most muscles on the opposite side of
the body.
In a similar manner, blockage of a posterior cerebral
exercise or during other states of excessive circulatory
artery will cause infarction of the occipital pole of the
activity, the sympathetic nervous system normally con-
hemisphere on the same side as the blockage, which
stricts the large- and intermediate-sized brain arteries
causes loss of vision in both eyes in the half of the retina
enough to prevent the high pressure from reaching the
on the same side as the stroke lesion. Especially devas-
smaller brain blood vessels. This is important in pre-
tating are strokes that involve the blood supply to the
venting vascular hemorrhages into the brain—that is,
midbrain because this can block nerve conduction in
for preventing the occurrence of “cerebral stroke.”
major pathways between the brain and spinal cord,
causing both sensory and motor abnormalities.
Cerebral Microcirculation
As is true for almost all other tissues of the body, the
Cerebrospinal Fluid System
number of blood capillaries in the brain is greatest
where the metabolic needs are greatest. The overall
The entire cerebral cavity enclosing the brain and spinal
metabolic rate of the brain gray matter where the neu-
cord has a capacity of about 1600 to 1700 milliliters;
ronal cell bodies lie is about four times as great as that
about 150 milliliters of this capacity is occupied by cere-
of white matter; correspondingly, the number of capil-
brospinal fluid and the remainder by the brain and cord.
laries and rate of blood flow are also about four times
This fluid, as shown in Figure 61-4, is present in the ven-
as great in the gray matter.
tricles of the brain, in the cisterns around the outside of
An important structural characteristic of the brain
the brain, and in the subarachnoid space around both the
capillaries is that they are much less “leaky” than the
brain and the spinal cord. All these chambers are con-
blood capillaries in almost any other tissue of the body.
nected with one another, and the pressure of the fluid
One reason for this is that the capillaries are supported
is maintained at a surprisingly constant level.
on all sides by “glial feet,” which are small projections
from the surrounding glial cells that abut against all sur-
faces of the capillaries and provide physical support to
prevent overstretching of the capillaries in case of high
Cushioning Function of the
capillary blood pressure.
Cerebrospinal Fluid
The walls of the small arterioles leading to the brain
capillaries become greatly thickened in people who
A major function of the cerebrospinal fluid is to cushion
develop high blood pressure, and these arterioles
the brain within its solid vault. The brain and the cere-
remain significantly constricted all the time to prevent
brospinal fluid have about the same specific gravity
transmission of the high pressure to the capillaries. We
(only about 4 per cent different), so that the brain
shall see later in the chapter that whenever these
simply floats in the fluid. Therefore, a blow to the head,
systems for protecting against transudation of fluid into
if it is not too intense, moves the entire brain simulta-
the brain break down, serious brain edema ensues,
neously with the skull, causing no one portion of the
which can lead rapidly to coma and death.
brain to be momentarily contorted by the blow.
764
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Lateral
Arachnoidal
ventricles
villi
Foramen
of Monro
Choroid
Artery
plexuses
Ependyma
Vein
Third
ventricle
Tentorium
cerebelli
Taenia
Aqueduct
fornicis
of Sylvius
Fourth
ventricle
Tela
Foramen of
Foramen of
choroides
Luschka
Magendie
Taenia
choroidea
Figure 61-4
Blood vessel
The arrows show the pathway of cerebrospinal fluid flow from the
choroid plexuses in the lateral ventricles to the arachnoidal villi
Ependyma
protruding into the dural sinuses.
Villus epithelium
Contrecoup. When a blow to the head is extremely
Villus connective tissue
severe, it may not damage the brain on the side of the
head where the blow is struck but on the opposite side.
This phenomenon is known as “contrecoup,” and the
Figure 61-5
reason for this effect is the following: When the blow is
struck, the fluid on the struck side is so incompressible
Choroid plexus in a lateral ventricle.
that as the skull moves, the fluid pushes the brain at the
same time in unison with the skull. On the side oppo-
site to the area that is struck, the sudden movement of
from the third ventricle, it flows downward along the
the whole skull causes the skull to pull away from the
aqueduct of Sylvius into the fourth ventricle, where still
brain momentarily because of the brain’s inertia, creat-
another minute amount of fluid is added. Finally, the
ing for a split second a vacuum space in the cranial vault
fluid passes out of the fourth ventricle through three
in the area opposite to the blow. Then, when the skull is
small openings, two lateral foramina of Luschka and a
no longer being accelerated by the blow, the vacuum
midline foramen of Magendie, entering the cisterna
suddenly collapses and the brain strikes the inner
magna, a fluid space that lies behind the medulla and
surface of the skull.
beneath the cerebellum.
The poles and the inferior surfaces of the frontal and
The cisterna magna is continuous with the subarach-
temporal lobes, where the brain comes into contact with
noid space that surrounds the entire brain and spinal
bony protuberances in the base of the skull, are often
cord. Almost all the cerebrospinal fluid then flows
the sites of injury and contusions (bruises) after a severe
upward from the cisterna magna through the subarach-
blow to the head, such as that experienced by a boxer.
noid spaces surrounding the cerebrum. From here, the
If the contusion occurs on the same side as the impact
fluid flows into and through multiple arachnoidal villi
injury, it is a coup injury; if it occurs on the opposite side,
that project into the large sagittal venous sinus and
the contusion is a contrecoup injury.
other venous sinuses of the cerebrum. Thus, any extra
fluid empties into the venous blood through pores of
these villi.
Formation, Flow, and Absorption
of Cerebrospinal Fluid
Secretion by the Choroid Plexus. The choroid plexus, a
section of which is shown in Figure 61-5, is a cauliflower-
Cerebrospinal fluid is formed at a rate of about 500 mil-
like growth of blood vessels covered by a thin layer of
liliters each day, which is three to four times as much as
epithelial cells. This plexus projects into (1 and 2) the
the total volume of fluid in the entire cerebrospinal fluid
temporal horn of each lateral ventricle, (3) the posterior
system. About two thirds or more of this fluid originates
portion of the third ventricle, and (4) the roof of the
as secretion from the choroid plexuses in the four ven-
fourth ventricle.
tricles, mainly in the two lateral ventricles. Additional
Secretion of fluid into the ventricles by the choroid
small amounts of fluid are secreted by the ependymal
plexus depends mainly on active transport of sodium
surfaces of all the ventricles and by the arachnoidal
ions through the epithelial cells lining the outside of the
membranes; and a small amount comes from the brain
plexus. The sodium ions in turn pull along large amounts
itself through the perivascular spaces that surround the
of chloride ions as well because the positive charge of
blood vessels passing through the brain.
the sodium ion attracts the chloride ion’s negative
The arrows in Figure 61-4 show that the main chan-
charge. The two of these together increase the quantity
nels of fluid flow from the choroid plexuses and then
of osmotically active sodium chloride in the cere-
through the cerebrospinal fluid system. The fluid
brospinal fluid, which then causes almost immediate
secreted in the lateral ventricles passes first into the third
osmosis of water through the membrane, thus providing
ventricle; then, after addition of minute amounts of fluid
the fluid of the secretion.
Chapter 61
Cerebral Blood Flow, Cerebrospinal Fluid, and Brain Metabolism
765
Less important transport processes move small
lar spaces, in effect, are a specialized lymphatic system
amounts of glucose into the cerebrospinal fluid and both
for the brain.
potassium and bicarbonate ions out of the cerebrospinal
In addition to transporting fluid and proteins, the
fluid into the capillaries. Therefore, the resulting char-
perivascular spaces transport extraneous particulate
acteristics of the cerebrospinal fluid become the follow-
matter out of the brain. For instance, whenever infec-
ing: osmotic pressure, approximately equal to that of
tion occurs in the brain, dead white blood cells and
plasma; sodium ion concentration, also approximately
other infectious debris are carried away through the
equal to that of plasma; chloride ion, about 15 per cent
perivascular spaces.
greater than in plasma; potassium ion, approximately 40
per cent less; and glucose, about 30 per cent less.
Cerebrospinal Fluid Pressure
Absorption of Cerebrospinal Fluid Through the Arachnoidal Villi.
The normal pressure in the cerebrospinal fluid system
The arachnoidal villi are microscopic fingerlike inward
when one is lying in a horizontal position averages 130
projections of the arachnoidal membrane through the
millimeters of water (10 mm Hg), although this may be
walls and into the venous sinuses. Conglomerates of
as low as 65 millimeters of water or as high as 195 mil-
these villi form macroscopic structures called arach-
limeters of water even in the normal healthy person.
noidal granulations that can be seen protruding into the
sinuses. The endothelial cells covering the villi have
been shown by electron microscopy to have vesicular
Regulation of Cerebrospinal Fluid Pressure by the Arachnoidal
passages directly through the bodies of the cells large
Villi. The normal rate of cerebrospinal fluid formation
enough to allow relatively free flow of (1) cerebrospinal
remains very nearly constant, so that changes in fluid
fluid, (2) dissolved protein molecules, and (3) even par-
formation are seldom a factor in pressure control. Con-
ticles as large as red and white blood cells into the
versely, the arachnoidal villi function like “valves” that
venous blood.
allow cerebrospinal fluid and its contents to flow readily
into the blood of the venous sinuses while not allowing
Perivascular Spaces and Cerebrospinal Fluid. The large arter-
blood to flow backward in the opposite direction. Nor-
ies and veins of the brain lie on the surface of the brain
mally, this valve action of the villi allows cerebrospinal
but their ends penetrate inward, carrying with them a
fluid to begin to flow into the blood when cerebrospinal
layer of pia mater, the membrane that covers the brain,
fluid pressure is about 1.5 mm Hg greater than the pres-
as shown in Figure 61-6. The pia is only loosely adher-
sure of the blood in the venous sinuses. Then, if the cere-
ent to the vessels, so that a space, the perivascular space,
brospinal fluid pressure rises still higher, the valves open
exists between it and each vessel. Therefore, perivascu-
more widely, so that under normal conditions, the cere-
lar spaces follow both the arteries and the veins into the
brospinal fluid pressure almost never rises more than a
brain as far as the arterioles and venules go.
few millimeters of mercury higher than the pressure in
the cerebral venous sinuses.
Lymphatic Function of the Perivascular Spaces. As is true
Conversely, in disease states, the villi sometimes
elsewhere in the body, a small amount of protein leaks
become blocked by large particulate matter, by fibrosis,
out of the brain capillaries into the interstitial spaces of
or by excesses of blood cells that have leaked into the
the brain. Because no true lymphatics are present in
cerebrospinal fluid in brain diseases. Such blockage can
brain tissue, excess protein in the brain tissue leaves the
cause high cerebrospinal fluid pressure, as follows.
tissue flowing with fluid through the perivascular spaces
into the subarachnoid spaces. On reaching the sub-
High Cerebrospinal Fluid Pressure in Pathological Conditions of
arachnoid spaces, the protein then flows with the cere-
the Brain. Often a large brain tumor elevates the cere-
brospinal fluid, to be absorbed through the arachnoidal
brospinal fluid pressure by decreasing reabsorption of
villi into the large cerebral veins. Therefore, perivascu-
the cerebrospinal fluid back into the blood. As a result,
the cerebrospinal fluid pressure can rise to as much as
500 millimeters of water (37 mm Hg) or about four
times normal.
The cerebrospinal fluid pressure also rises consider-
Arachnoid membrane
ably when hemorrhage or infection occurs in the cranial
Arachnoid trabecula
vault. In both these conditions, large numbers of red
and/or white blood cells suddenly appear in the cere-
Subarachnoid space
brospinal fluid, and they can cause serious blockage of
the small absorption channels through the arachnoidal
Pia mater
villi. This also sometimes elevates the cerebrospinal
fluid pressure to 400 to 600 millimeters of water (about
Perivascular space
four times normal).
Blood vessel
Some babies are born with high cerebrospinal fluid
pressure. This is often caused by abnormally high resist-
ance to fluid reabsorption through the arachnoidal villi,
Brain tissue
resulting either from too few arachnoidal villi or from
villi with abnormal absorptive properties. This is dis-
cussed later in connection with hydrocephalus.
Figure 61-6
Measurement of Cerebrospinal Fluid Pressure. The usual pro-
cedure for measuring cerebrospinal fluid pressure is
Drainage of a perivascular space into the subarachnoid space.
(Redrawn from Ranson SW, Clark SL: Anatomy of the Nervous
very simple and is the following: First, the person lies
System. Philadelphia: WB Saunders Co, 1959.)
exactly horizontally on his or her side so that the fluid
766
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
pressure in the spinal canal is equal to the pressure in
extent inside the ventricles. This will also cause the head
the cranial vault. A spinal needle is then inserted into
to swell tremendously if it occurs in infancy when the
the lumbar spinal canal below the lower end of the cord,
skull is still pliable and can be stretched, and it can
and the needle is connected to a vertical glass tube that
damage the brain at any age. A therapy for many types
is open to the air at its top. The spinal fluid is allowed
of hydrocephalus is surgical placement of a silicone tube
to rise in the tube as high as it will. If it rises to a level
shunt all the way from one of the brain ventricles to the
136 millimeters above the level of the needle, the pres-
peritoneal cavity where the excess fluid can be absorbed
sure is said to be 136 millimeters of water pressure or,
into the blood.
dividing this by 13.6, which is the specific gravity of
mercury, about 10 mm Hg pressure.
Blood-Cerebrospinal Fluid and
High Cerebrospinal Fluid Pressure Causes Edema of the Optic
Blood-Brain Barriers
Disc—Papilledema. Anatomically, the dura of the brain
It has already been pointed out that the concentrations
extends as a sheath around the optic nerve and then
of several important constituents of cerebrospinal fluid
connects with the sclera of the eye. When the pressure
are not the same as in extracellular fluid elsewhere
rises in the cerebrospinal fluid system, it also rises inside
in the body. Furthermore, many large molecular sub-
the optic nerve sheath. The retinal artery and vein
stances hardly pass at all from the blood into the cere-
pierce this sheath a few millimeters behind the eye and
brospinal fluid or into the interstitial fluids of the brain,
then pass along with the optic nerve fibers into the eye
even though these same substances pass readily into
itself. Therefore, (1) high cerebrospinal fluid pressure
the usual interstitial fluids of the body. Therefore, it is
pushes fluid first into the optic nerve sheath and then
said that barriers, called the blood-cerebrospinal fluid
along the spaces between the optic nerve fibers to the
barrier and the blood-brain barrier, exist between the
interior of the eyeball; (2) the high pressure decreases
blood and the cerebrospinal fluid and brain fluid,
outward fluid flow in the optic nerves, causing accumu-
respectively.
lation of excess fluid in the optic disc at the center of
Barriers exist both at the choroid plexus and at the
the retina; and
(3) the pressure in the sheath also
tissue capillary membranes in essentially all areas of the
impedes flow of blood in the retinal vein, thereby
brain parenchyma except in some areas of the hypothal-
increasing the retinal capillary pressure throughout the
amus, pineal gland, and area postrema, where substances
eye, which results in still more retinal edema.
diffuse with greater ease into the tissue spaces. The ease
The tissues of the optic disc are much more distensi-
of diffusion in these areas is important because they
ble than those of the remainder of the retina, so that the
have sensory receptors that respond to specific changes
disc becomes far more edematous than the remainder
in the body fluids, such as changes in osmolality and in
of the retina and swells into the cavity of the eye. The
glucose concentration, as well as receptors for peptide
swelling of the disc can be observed with an ophthal-
hormones that regulate thirst, such as angiotensin II.
moscope and is called papilledema. Neurologists can
The blood-brain barrier also has specific carrier mole-
estimate the cerebrospinal fluid pressure by assessing
cules that facilitate transport of hormones, such as
the extent to which the edematous optic disc protrudes
leptin, from the blood into the hypothalamus where
into the eyeball.
they bind to specific receptors that control other func-
tions such as appetite and sympathetic nervous system
Obstruction to Flow of
activity.
In general, the blood-cerebrospinal fluid and blood-
Cerebrospinal Fluid Can
brain barriers are highly permeable to water, carbon
Cause Hydrocephalus
dioxide, oxygen, and most lipid-soluble substances
such as alcohol and anesthetics; slightly permeable to
“Hydrocephalus” means excess water in the cranial
electrolytes such as sodium, chloride, and potassium;
vault. This condition is frequently divided into commu-
and almost totally impermeable to plasma proteins and
nicating hydrocephalus and noncommunicating hydro-
most non-lipid-soluble large organic molecules. There-
cephalus. In communicating hydrocephalus fluid flows
fore, the blood-cerebrospinal fluid and blood-brain bar-
readily from the ventricular system into the subarach-
riers often make it impossible to achieve effective
noid space, whereas in noncommunicating hydro-
concentrations of therapeutic drugs, such as protein
cephalus fluid flow out of one or more of the ventricles
antibodies and non-lipid-soluble drugs, in the cere-
is blocked.
brospinal fluid or parenchyma of the brain.
Usually the noncommunicating type of hydro-
The cause of the low permeability of the blood-
cephalus is caused by a block in the aqueduct of Sylvius,
cerebrospinal fluid and blood-brain barriers is the
resulting from atresia (closure) before birth in many
manner in which the endothelial cells of the brain tissue
babies or from blockage by a brain tumor at any age.
capillaries are joined to one another. They are joined by
As fluid is formed by the choroid plexuses in the two
so-called tight junctions. That is, the membranes of the
lateral and the third ventricles, the volumes of these
adjacent endothelial cells are tightly fused rather than
three ventricles increase greatly. This flattens the brain
having large slit-pores between them, as is the case for
into a thin shell against the skull. In neonates, the
most other capillaries of the body.
increased pressure also causes the whole head to swell
because the skull bones have not yet fused.
The communicating type of hydrocephalus is usually
Brain Edema
caused by blockage of fluid flow in the subarachnoid
spaces around the basal regions of the brain or by block-
One of the most serious complications of abnormal
age of the arachnoidal villi where the fluid is normally
cerebral fluid dynamics is the development of brain
absorbed into the venous sinuses. Fluid therefore col-
edema. Because the brain is encased in a solid cranial
lects both on the outside of the brain and to a lesser
vault, accumulation of extra edema fluid compresses the
Chapter 61
Cerebral Blood Flow, Cerebrospinal Fluid, and Brain Metabolism
767
blood vessels, often causing seriously decreased blood
breaking down glucose and glycogen but without com-
flow and destruction of brain tissue.
bining these with oxygen. This delivers energy only at
The usual cause of brain edema is either greatly
the expense of consuming tremendous amounts of
increased capillary pressure or damage to the capillary
glucose and glycogen. However, it does keep the tissues
wall that makes the wall leaky to fluid. A very common
alive.
cause is a serious blow to the head, leading to brain con-
The brain is not capable of much anaerobic metabo-
cussion, in which the brain tissues and capillaries are
lism. One of the reasons for this is the high metabolic
traumatized so that capillary fluid leaks into the trau-
rate of the neurons, so that most neuronal activity
matized tissues.
depends on second-by-second delivery of oxygen from
Once brain edema begins, it often initiates two vicious
the blood. Putting these factors together, one can under-
circles because of the following positive feedbacks:
stand why sudden cessation of blood flow to the brain
(1) Edema compresses the vasculature. This in turn
or sudden total lack of oxygen in the blood can cause
decreases blood flow and causes brain ischemia. The
unconsciousness within 5 to 10 seconds.
ischemia in turn causes arteriolar dilation with still
further increase in capillary pressure. The increased cap-
Under Normal Conditions Most Brain Energy Is Supplied by
illary pressure then causes more edema fluid, so that the
Glucose. Under normal conditions, almost all the energy
edema becomes progressively worse. (2) The decreased
used by the brain cells is supplied by glucose derived
cerebral blood flow also decreases oxygen delivery. This
from the blood. As is true for oxygen, most of this is
increases the permeability of the capillaries, allowing
derived minute by minute and second by second from
still more fluid leakage. It also turns off the sodium
the capillary blood, with a total of only about a 2-minute
pumps of the neuronal tissue cells, thus allowing these
supply of glucose normally stored as glycogen in the
cells to swell in addition.
neurons at any given time.
Once these two vicious circles have begun, heroic
A special feature of glucose delivery to the neurons
measures must be used to prevent total destruction of
is that its transport into the neurons through the cell
the brain. One such measure is to infuse intravenously
membrane is not dependent on insulin, even though
a concentrated osmotic substance, such as a very con-
insulin is required for glucose transport into most other
centrated mannitol solution. This pulls fluid by osmosis
body cells. Therefore, in patients who have serious dia-
from the brain tissue and breaks up the vicious circles.
betes with essentially zero secretion of insulin, glucose
Another procedure is to remove fluid quickly from the
still diffuses readily into the neurons—which is most
lateral ventricles of the brain by means of ventricular
fortunate in preventing loss of mental function in
needle puncture, thereby relieving the intracerebral
diabetic patients. Yet, when a diabetic patient is
pressure.
overtreated with insulin, the blood glucose concentra-
tion can fall extremely low because the excess insulin
causes almost all the glucose in the blood to be trans-
Brain Metabolism
ported rapidly into the vast numbers of insulin-sensitive
non-neural cells throughout the body, especially into
Like other tissues, the brain requires oxygen and food
muscle and liver cells. When this happens, not enough
nutrients to supply its metabolic needs. However, there
glucose is left in the blood to supply the neurons prop-
are special peculiarities of brain metabolism that
erly, and mental function does then become seriously
require mention.
deranged, leading sometimes to coma and even more
often to mental imbalances and psychotic distur-
Total Brain Metabolic Rate and Metabolic Rate of Neurons.
bances—all caused by overtreatment with insulin.
Under resting but awake conditions, the metabolism of
the brain accounts for about 15 per cent of the total
metabolism in the body, even though the mass of the
References
brain is only 2 per cent of the total body mass. There-
fore, under resting conditions, brain metabolism per unit
Anderson CM, Nedergaard M: Astrocyte-mediated control
mass of tissue is about 7.5 times the average metabo-
of cerebral microcirculation. Trends Neurosci
26:340,
lism in non-nervous system tissues.
2003.
Most of this excess metabolism of the brain occurs in
Antunes-Rodrigues J, de Castro M, Elias LL, et al: Neu-
the neurons, not in the glial supportive tissues. The
roendocrine control of body fluid metabolism. Physiol Rev
major need for metabolism in the neurons is to pump
84:169, 2004.
ions through their membranes, mainly to transport
Burmester T, Hankeln T: Neuroglobin: a respiratory protein
sodium and calcium ions to the outside of the neuronal
of the nervous system. News Physiol Sci 19:110, 2004.
membrane and potassium ions to the interior. Each time
Chesler M: Regulation and modulation of pH in the brain.
a neuron conducts an action potential, these ions move
Physiol Rev 83:1183, 2003.
through the membranes, increasing the need for addi-
Duelli R, Kuschinsky W: Brain glucose transporters: rela-
tional membrane transport to restore proper ionic con-
tionship to local energy demand. News Physiol Sci 16:71,
centration differences across the neuron membranes.
2001.
Therefore, during excessive brain activity, neuronal
Faraci FM: Vascular protection. Stroke 34:327, 2003.
metabolism can increase as much as 100 to 150 per cent.
Faraci FM, Heistad DD: Regulation of the cerebral circula-
tion: role of endothelium and potassium channels. Physiol
Special Requirement of the Brain for Oxygen—Lack of Significant
Rev 78:53, 1998.
Anaerobic Metabolism. Most tissues of the body can live
Ganong WF: Circumventricular organs: definition and role
without oxygen for several minutes and some for as long
in the regulation of endocrine and autonomic function.
as 30 minutes. During this time, the tissue cells obtain
Clin Exp Pharmacol Physiol 27:422, 2000.
their energy through processes of anaerobic metab-
Gore JC: Principles and practice of functional MRI of the
olism, which means release of energy by partially
human brain. J Clin Invest 112:4, 2003.
768
Unit XI The Nervous System: C. Motor and Integrative Neurophysiology
Harder DR, Zhang C, Gebremedhin D: Astrocytes function
Pardridge WM: An Introduction to the Blood-Brain Barrier.
in matching blood flow to metabolic activity. News Physiol
Cambridge: Cambridge University Press, 1998.
Sci 17:27, 2002.
Paulson OB: Blood-brain barrier, brain metabolism and
Johnston M, Papaiconomou C: Cerebrospinal fluid trans-
cerebral blood flow. Eur Neuropsychopharmacol 12:495,
port: a lymphatic perspective. News Physiol Sci 17:227,
2002.
2002.
Roman RJ: P-450 metabolites of arachidonic acid in the
Logothetis NK, Wandell BA: Interpreting the BOLD signal.
control of cardiovascular function. Physiol Rev 82:131,
Annu Rev Physiol 66:735, 2004.
2002.
Nadareishvili Z, Hallenbeck J: Neuronal regeneration after
Roth J, Harre EM, Rummel C, et al: Signaling the brain in
stroke. N Engl J Med 348:2355, 2003.
systemic inflammation: role of sensory circumventricular
Osborn JW, Collister JP, Carlson SH: Angiotensin and
organs. Front Biosci 9:290, 2004.
osmoreceptor inputs to the area postrema: role in long-
term control of fluid homeostasis and arterial pressure.
Clin Exp Pharmacol Physiol 27:443, 2000.
U N I T XII
Gastrointestinal
Physiology
62. General Principles of Gastrointestinal Function—
Motility, Nervous Control, and Blood Circulation
63. Propulsion and Mixing of Food in the
Alimentary Tract
64. Secretory Functions of the Alimentary Tract
65. Digestion and Absorption in the
Gastrointestinal Tract
66. Physiology of Gastrointestinal Disorders
C H A P T E R
6
2
General Principles of
Gastrointestinal Function—
Motility, Nervous Control, and
Blood Circulation
The alimentary tract provides the body with a con-
tinual supply of water, electrolytes, and nutrients. To
achieve this requires (1) movement of food through
the alimentary tract; (2) secretion of digestive juices
and digestion of the food; (3) absorption of water,
various electrolytes, and digestive products;
(4)
circulation of blood through the gastrointestinal
organs to carry away the absorbed substances; and
(5) control of all these functions by local, nervous, and hormonal systems.
Figure 62-1 shows the entire alimentary tract. Each part is adapted to its spe-
cific functions: some to simple passage of food, such as the esophagus; others to
temporary storage of food, such as the stomach; and others to digestion and
absorption, such as the small intestine. In this chapter, we discuss the basic prin-
ciples of function in the entire alimentary tract; in the following chapters, we
will discuss the specific functions of different segments of the tract.
General Principles of Gastrointestinal Motility
Physiologic Anatomy of the Gastrointestinal Wall
Figure 62-2 shows a typical cross section of the intestinal wall, including the fol-
lowing layers from outer surface inward: (1) the serosa, (2) a longitudinal muscle
layer, (3) a circular muscle layer, (4) the submucosa, and (5) the mucosa. In addi-
tion, sparse bundles of smooth muscle fibers, the mucosal muscle, lie in the
deeper layers of the mucosa. The motor functions of the gut are performed by
the different layers of smooth muscle.
The general characteristics of smooth muscle and its function are discussed
in Chapter 8, which should be reviewed as a background for the following sec-
tions of this chapter. The specific characteristics of smooth muscle in the gut are
the following.
Gastrointestinal Smooth Muscle Functions as a Syncytium. The individual smooth
muscle fibers in the gastrointestinal tract are 200 to 500 micrometers in length
and 2 to 10 micrometers in diameter, and they are arranged in bundles of as
many as 1000 parallel fibers. In the longitudinal muscle layer, the bundles extend
longitudinally down the intestinal tract; in the circular muscle layer, they extend
around the gut.
Within each bundle, the muscle fibers are electrically connected with one
another through large numbers of gap junctions that allow low-resistance move-
ment of ions from one muscle cell to the next. Therefore, electrical signals that
initiate muscle contractions can travel readily from one fiber to the next within
each bundle but more rapidly along the length of the bundle than sideways.
Each bundle of smooth muscle fibers is partly separated from the next by
loose connective tissue, but the muscle bundles fuse with one another at many
771
772
Unit XII Gastrointestinal Physiology
Spikes
0
Depolarization
Parotid gland
-10
Mouth
-20
Slow
Salivary glands
waves
-30
Stimulation by
-40
Esophagus
1. Norepinephrine
-50
2. Sympathetics
-60
Resting
Stimulation by
-70
1. Stretch
Hyperpolarization
2. Acetylcholine
3. Parasympathetics
0
6
12
18
24
30
36
42
48
54
Seconds
Liver
Stomach
Gallbladder
Pancreas
Duodenum
Figure 62-3
Transverse
Jejunum
colon
Membrane potentials in intestinal smooth muscle. Note the slow
Ascending
Descending
waves, the spike potentials, total depolarization, and hyperpolar-
colon
colon
ization, all of which occur under different physiologic conditions
of the intestine.
Ileum
Anus
on the excitability of the muscle; sometimes it stops
after only a few millimeters and at other times it
travels many centimeters or even the entire length and
Figure 62-1
breadth of the intestinal tract.
Also, a few connections exist between the longitu-
Alimentary tract.
dinal and circular muscle layers, so that excitation of
one of these layers often excites the other as well.
Serosa
Electrical Activity of Gastrointestinal
Circular muscle
Smooth Muscle
Longitudinal
The smooth muscle of the gastrointestinal tract is
muscle
excited by almost continual slow, intrinsic electrical
Submucosa
activity along the membranes of the muscle fibers.
Meissner's
This activity has two basic types of electrical waves: (1)
nerve plexus
slow waves and (2) spikes, both of which are shown
Mucosa
in Figure 62-3. In addition, the voltage of the resting
Epithelial
membrane potential of the gastrointestinal smooth
lining
muscle can be made to change to different levels, and
Mucosal
this too can have important effects in controlling
muscle
motor activity of the gastrointestinal tract.
Mucosal gland
Slow Waves. Most gastrointestinal contractions occur
Myenteric nerve
rhythmically, and this rhythm is determined mainly by
plexus
the frequency of so-called “slow waves” of smooth
Submucosal gland
muscle membrane potential. These waves, shown in
Figure 62-3, are not action potentials. Instead, they are
Mesentery
slow, undulating changes in the resting membrane
potential. Their intensity usually varies between 5 and
Figure 62-2
15 millivolts, and their frequency ranges in different
Typical cross section of the gut.
parts of the human gastrointestinal tract from 3 to 12
per minute: about 3 in the body of the stomach, as
much as 12 in the duodenum, and about 8 or 9 in the
points, so that in reality each muscle layer represents
terminal ileum. Therefore, the rhythm of contraction
a branching latticework of smooth muscle bundles.
of the body of the stomach usually is about 3 per
Therefore, each muscle layer functions as a syncytium;
minute, of the duodenum about 12 per minute, and of
that is, when an action potential is elicited anywhere
the ileum 8 to 9 per minute.
within the muscle mass, it generally travels in all direc-
The precise cause of the slow waves is not com-
tions in the muscle. The distance that it travels depends
pletely understood, although they appear to be caused
Chapter 62
General Principles of Gastrointestinal Function—Motility, Nervous Control, and Blood Circulation
773
by complex interactions among the smooth muscle
which is called depolarization of the membrane, the
cells and specialized cells, called the interstitial cells of
muscle fibers become more excitable. When the poten-
Cajal, that are believed to act as electrical pacemakers
tial becomes more negative, which is called hyperpo-
for smooth muscle cells. These interstitial cells form a
larization, the fibers become less excitable.
network with each other and are interposed between
Factors that depolarize the membrane—that is,
the smooth muscle layers, with synaptic-like contacts
make it more excitable—are (1) stretching of the
to smooth muscle cells. The interstitial cells of Cajal
muscle, (2) stimulation by acetylcholine, (3) stimula-
undergo cyclic changes in membrane potential due
tion by parasympathetic nerves that secrete acetyl-
to unique ion channels that periodically open and
choline at their endings, and (4) stimulation by several
produce inward (pacemaker) currents that may gen-
specific gastrointestinal hormones.
erate slow wave activity.
Important factors that make the membrane poten-
The slow waves usually do not by themselves cause
tial more negative—that is, hyperpolarize the mem-
muscle contraction in most parts of the gastrointesti-
brane and make the muscle fibers less excitable—are
nal tract, except perhaps in the stomach. Instead, they
(1) the effect of norepinephrine or epinephrine on the
mainly excite the appearance of intermittent spike
fiber membrane and (2) stimulation of the sympathetic
potentials, and the spike potentials in turn actually
nerves that secrete mainly norepinephrine at their
excite the muscle contraction.
endings.
Spike Potentials. The spike potentials are true action
Calcium Ions and Muscle Contraction. Smooth muscle con-
potentials. They occur automatically when the resting
traction occurs in response to entry of calcium ions
membrane potential of the gastrointestinal smooth
into the muscle fiber. As explained in Chapter 8,
muscle becomes more positive than about -40 milli-
calcium ions, acting through a calmodulin control
volts (the normal resting membrane potential in the
mechanism, activate the myosin filaments in the fiber,
smooth muscle fibers of the gut is between -50 and
causing attractive forces to develop between the
-60 millivolts). Thus, note in Figure 62-3 that each time
myosin filaments and the actin filaments, thereby
the peaks of the slow waves temporarily become more
causing the muscle to contract.
positive than -40 millivolts, spike potentials appear on
The slow waves do not cause calcium ions to enter
these peaks. The higher the slow wave potential rises,
the smooth muscle fiber (only sodium ions). Therefore,
the greater the frequency of the spike potentials,
the slow waves by themselves usually cause no muscle
usually ranging between 1 and 10 spikes per second.
contraction. Instead, it is during the spike potentials,
The spike potentials last 10 to 40 times as long in gas-
generated at the peaks of the slow waves, that signifi-
trointestinal muscle as the action potentials in large
cant quantities of calcium ions do enter the fibers and
nerve fibers, each gastrointestinal spike lasting as long
cause most of the contraction.
as 10 to 20 milliseconds.
Another important difference between the action
Tonic Contraction of Some Gastrointestinal Smooth Muscle.
potentials of the gastrointestinal smooth muscle and
Some smooth muscle of the gastrointestinal tract
those of nerve fibers is the manner in which they are
exhibits tonic contraction as well as or instead of rhyth-
generated. In nerve fibers, the action potentials are
mical contractions. Tonic contraction is continuous, not
caused almost entirely by rapid entry of sodium ions
associated with the basic electrical rhythm of the slow
through sodium channels to the interior of the fibers.
waves but often lasting several minutes or even hours.
In gastrointestinal smooth muscle fibers, the channels
The tonic contraction often increases or decreases in
responsible for the action potentials are somewhat dif-
intensity but continues.
ferent; they allow especially large numbers of calcium
Tonic contraction is sometimes caused by con-
ions to enter along with smaller numbers of sodium
tinuous repetitive spike potentials—the greater the
ions and therefore are called calcium-sodium channels.
frequency, the greater the degree of contraction. At
These channels are much slower to open and close
other times, tonic contraction is caused by hormones
than are the rapid sodium channels of large nerve
or other factors that bring about continuous partial
fibers. The slowness of opening and closing of the
depolarization of the smooth muscle membrane
calcium-sodium channels accounts for the long dura-
without causing action potentials. A third cause of
tion of the action potentials. Also, the movement of
tonic contraction is continuous entry of calcium ions
large amounts of calcium ions to the interior of the
into the interior of the cell brought about in ways not
muscle fiber during the action potential plays a special
associated with changes in membrane potential. The
role in causing the intestinal muscle fibers to contract,
details of these mechanisms are still unclear.
as we discuss shortly.
Changes in Voltage of the Resting Membrane Potential. In
Neural Control of
addition to the slow waves and spike potentials, the
Gastrointestinal Function—
baseline voltage level of the smooth muscle resting
membrane potential also can change. Under normal
Enteric Nervous System
conditions, the resting membrane potential averages
about -56 millivolts, but multiple factors can change
The gastrointestinal tract has a nervous system all its
this level. When the potential becomes less negative,
own called the enteric nervous system. It lies entirely
774
Unit XII Gastrointestinal Physiology
Sympathetic
Parasympathetic
To prevertebral
ganglia, spinal
(mainly postganglionic)
(preganglionic)
cord, and brain
stem
Myenteric
plexus
Figure 62-4
Neural control of the gut wall, showing (1)
Submucosal
the myenteric and submucosal plexuses
plexus
(black fibers); (2) extrinsic control of these
plexuses by the sympathetic and
parasympathetic nervous systems
(red
Sensory
fibers); and
(3) sensory fibers passing
neurons
from the luminal epithelium and gut wall
to the enteric plexuses, then to the pre-
Epithelium
vertebral ganglia of the spinal cord and
directly to the spinal cord and brain stem
(dashed fibers).
in the wall of the gut, beginning in the esophagus and
Differences Between the Myenteric
extending all the way to the anus. The number of
and Submucosal Plexuses
neurons in this enteric system is about 100 million,
almost exactly equal to the number in the entire spinal
cord. This highly developed enteric nervous system is
The myenteric plexus consists mostly of a linear chain
especially important in controlling gastrointestinal
of many interconnecting neurons that extends the
movements and secretion.
entire length of the gastrointestinal tract. A section of
The enteric nervous system is composed mainly of
this chain is shown in Figure 62-4.
two plexuses, shown in Figure 62-4: (1) an outer plexus
Because the myenteric plexus extends all the way
lying between the longitudinal and circular muscle
along the intestinal wall and because it lies between
layers, called the myenteric plexus or Auerbach’s
the longitudinal and circular layers of intestinal
plexus, and (2) an inner plexus, called the submucosal
smooth muscle, it is concerned mainly with controlling
plexus or Meissner’s plexus, that lies in the submucosa.
muscle activity along the length of the gut. When this
The nervous connections within and between these
plexus is stimulated, its principal effects are
(1)
two plexuses are also shown in Figure 62-4.
increased tonic contraction, or “tone,” of the gut wall,
The myenteric plexus controls mainly the gastroin-
(2) increased intensity of the rhythmical contractions,
testinal movements, and the submucosal plexus con-
(3) slightly increased rate of the rhythm of contraction,
trols mainly gastrointestinal secretion and local blood
and (4) increased velocity of conduction of excitatory
flow.
waves along the gut wall, causing more rapid move-
Note especially in Figure 62-4 the extrinsic sympa-
ment of the gut peristaltic waves.
thetic and parasympathetic fibers that connect to both
The myenteric plexus should not be considered
the myenteric and submucosal plexuses. Although the
entirely excitatory because some of its neurons are
enteric nervous system can function on its own, inde-
inhibitory; their fiber endings secrete an inhibitory
pendently of these extrinsic nerves, stimulation by the
transmitter, possibly vasoactive intestinal polypeptide
parasympathetic and sympathetic systems can greatly
or some other inhibitory peptide. The resulting
enhance or inhibit gastrointestinal functions, as we
inhibitory signals are especially useful for inhibiting
discuss later.
some of the intestinal sphincter muscles that impede
Also shown in Figure
62-4 are sensory nerve
movement of food along successive segments of the
endings that originate in the gastrointestinal epithe-
gastrointestinal tract, such as the pyloric sphincter,
lium or gut wall and send afferent fibers to both
which controls emptying of the stomach into the duo-
plexuses of the enteric system, as well as (1) to the pre-
denum, and the sphincter of the ileocecal valve, which
vertebral ganglia of the sympathetic nervous system,
controls emptying from the small intestine into the
(2) to the spinal cord, and (3) in the vagus nerves all
cecum.
the way to the brain stem. These sensory nerves can
The submucosal plexus, in contrast to the myenteric
elicit local reflexes within the gut wall itself and still
plexus, is mainly concerned with controlling function
other reflexes that are relayed to the gut from either
within the inner wall of each minute segment of the
the prevertebral ganglia or the basal regions of the
intestine. For instance, many sensory signals originate
brain.
from the gastrointestinal epithelium and are then
Chapter 62
General Principles of Gastrointestinal Function—Motility, Nervous Control, and Blood Circulation
775
integrated in the submucosal plexus to help control
Sympathetic Innervation. The sympathetic fibers to the
local intestinal secretion, local absorption, and local
gastrointestinal tract originate in the spinal cord
contraction of the submucosal muscle that causes
between segments T-5 and L-2. Most of the pregan-
various degrees of infolding of the gastrointestinal
glionic fibers that innervate the gut, after leaving the
mucosa.
cord, enter the sympathetic chains that lie lateral to the
spinal column, and many of these fibers then pass on
through the chains to outlying ganglia such as to the
Types of Neurotransmitters Secreted
celiac ganglion and various mesenteric ganglia. Most of
by Enteric Neurons
the postganglionic sympathetic neuron bodies are in
these ganglia, and postganglionic fibers then spread
In an attempt to understand better the multiple func-
through postganglionic sympathetic nerves to all parts
tions of the gastrointestinal enteric nervous system,
of the gut.. The sympathetics innervate essentially all
research workers the world over have identified a
of the gastrointestinal tract, rather than being more
dozen or more different neurotransmitter substances
extensive nearest the oral cavity and anus as is true of
that are released by the nerve endings of different
the parasympathetics. The sympathetic nerve endings
types of enteric neurons. Two of them with which we
secrete mainly norepinephrine but also small amounts
are already familiar are (1) acetylcholine and (2) nor-
of epinephrine.
epinephrine. Others are (3) adenosine triphosphate,
In general, stimulation of the sympathetic nervous
(4) serotonin, (5) dopamine, (6) cholecystokinin, (7)
system inhibits activity of the gastrointestinal tract,
substance P,
(8) vasoactive intestinal polypeptide,
causing many effects opposite to those of the
(9) somatostatin,
(10) leu-enkephalin,
(11) met-
parasympathetic system. It exerts its effects in two
enkephalin, and (12) bombesin. The specific functions
ways: (1) to a slight extent by direct effect of secreted
of many of these are not known well enough to justify
norepinephrine to inhibit intestinal tract smooth
discussion here, other than to point out the following.
muscle (except the mucosal muscle, which it excites)
Acetylcholine most often excites gastrointestinal
and (2) to a major extent by an inhibitory effect of
activity. Norepinephrine almost always inhibits gas-
norepinephrine on the neurons of the entire enteric
trointestinal activity. This is also true of epinephrine,
nervous system.
which reaches the gastrointestinal tract mainly by way
Strong stimulation of the sympathetic system can
of the blood after it is secreted by the adrenal medul-
inhibit motor movements of the gut so greatly that this
lae into the circulation. The other aforementioned
literally can block movement of food through the gas-
transmitter substances are a mixture of excitatory and
trointestinal tract.
inhibitory agents, some of which we will discuss in the
following chapter.
Afferent Sensory Nerve Fibers from the Gut
Many afferent sensory nerve fibers innervate the gut.
Autonomic Control of the
Some of them have their cell bodies in the enteric
Gastrointestinal Tract
nervous system itself and some in the dorsal root
Parasympathetic Innervation. The parasympathetic sup-
ganglia of the spinal cord. These sensory nerves can be
ply to the gut is divided into cranial and sacral divi-
stimulated by (1) irritation of the gut mucosa, (2)
sions, which were discussed in Chapter 60.
excessive distention of the gut, or (3) presence of spe-
Except for a few parasympathetic fibers to the
cific chemical substances in the gut. Signals trans-
mouth and pharyngeal regions of the alimentary tract,
mitted through the fibers can then cause excitation or,
the cranial parasympathetic nerve fibers are almost
under other conditions, inhibition of intestinal move-
entirely in the vagus nerves. These fibers provide
ments or intestinal secretion.
extensive innervation to the esophagus, stomach, and
In addition, other sensory signals from the gut go all
pancreas and somewhat less to the intestines down
the way to multiple areas of the spinal cord and
through the first half of the large intestine.
even the brain stem. For example, 80 per cent of the
The sacral parasympathetics originate in the second,
nerve fibers in the vagus nerves are afferent rather
third, and fourth sacral segments of the spinal cord and
than efferent. These afferent fibers transmit sensory
pass through the pelvic nerves to the distal half of the
signals from the gastrointestinal tract into the brain
large intestine and all the way to the anus. The sig-
medulla, which in turn initiates vagal reflex signals that
moidal, rectal, and anal regions are considerably better
return to the gastrointestinal tract to control many of
supplied with parasympathetic fibers than are the
its functions.
other intestinal areas. These fibers function especially
to execute the defecation reflexes, discussed in
Gastrointestinal Reflexes
Chapter 63.
The anatomical arrangement of the enteric nervous
The postganglionic neurons of the gastrointestinal
system and its connections with the sympathetic and
parasympathetic system are located mainly in the
parasympathetic systems support three types of gas-
myenteric and submucosal plexuses. Stimulation of
trointestinal reflexes that are essential to gastroin-
these parasympathetic nerves causes general increase
testinal control. They are the following:
in activity of the entire enteric nervous system. This
1. Reflexes that are integrated entirely within the gut
in turn enhances activity of most gastrointestinal
wall enteric nervous system. These include reflexes
functions.
that control much gastrointestinal secretion,
776
Unit XII Gastrointestinal Physiology
peristalsis, mixing contractions, local inhibitory
of the duodenum in response to acidic gastric juice
effects, and so forth.
emptying into the duodenum from the pylorus of the
2. Reflexes from the gut to the prevertebral
stomach. Secretin has a mild effect on motility of the
sympathetic ganglia and then back to the
gastrointestinal tract and acts to promote pancreatic
gastrointestinal tract. These reflexes transmit
secretion of bicarbonate which in turn helps to neu-
signals long distances to other areas of the
tralize the acid in the small intestine.
gastrointestinal tract, such as signals from the
Gastric inhibitory peptide is secreted by the mucosa
stomach to cause evacuation of the colon (the
of the upper small intestine, mainly in response to fatty
gastrocolic reflex), signals from the colon and
acids and amino acids but to a lesser extent in response
small intestine to inhibit stomach motility and
to carbohydrate. It has a mild effect in decreasing
stomach secretion (the enterogastric reflexes), and
motor activity of the stomach and therefore slows
reflexes from the colon to inhibit emptying of ileal
emptying of gastric contents into the duodenum when
contents into the colon (the colonoileal reflex).
the upper small intestine is already overloaded with
3. Reflexes from the gut to the spinal cord or brain
food products.
stem and then back to the gastrointestinal tract.
Motilin is secreted by the upper duodenum during
These include especially (1) reflexes from the
fasting, and the only known function of this hormone
stomach and duodenum to the brain stem and
is to increase gastrointestinal motility. Motilin is
back to the stomach—by way of the vagus
released cyclically and stimulates waves of gastroin-
nerves—to control gastric motor and secretory
testinal motility called interdigestive myoelectric com-
activity; (2) pain reflexes that cause general
plexes that move through the stomach and small
inhibition of the entire gastrointestinal tract; and
intestine every 90 minutes in a fasted person. Motilin
(3) defecation reflexes that travel from the colon
secretion is inhibited after ingestion by mechanisms
and rectum to the spinal cord and back again
that are not fully understood.
to produce the powerful colonic, rectal, and
abdominal contractions required for defecation
Functional Types of
(the defecation reflexes).
Movements in the
Gastrointestinal Tract
Hormonal Control of
Gastrointestinal Motility
Two types of movements occur in the gastrointestinal
tract: (1) propulsive movements, which cause food to
In Chapter 64, we discuss the extreme importance of
move forward along the tract at an appropriate rate
several hormones for controlling gastrointestinal
to accommodate digestion and absorption, and (2)
secretion. Most of these same hormones also affect
mixing movements, which keep the intestinal contents
motility in some parts of the gastrointestinal
thoroughly mixed at all times.
tract. Although the motility effects are usually less
important than the secretory effects of the hormones,
some of the more important of them are the
Propulsive Movements—Peristalsis
following.
Gastrin is secreted by the “G” cells of the antrum of
The basic propulsive movement of the gastrointestinal
the stomach in response to stimuli associated with
tract is peristalsis, which is illustrated in Figure 62-5.
ingestion of a meal, such as distention of the stomach,
A contractile ring appears around the gut and then
the products of proteins, and gastrin releasing peptide,
moves forward; this is analogous to putting one’s
which is released by the nerves of the gastric mucosa
fingers around a thin distended tube, then constricting
during vagal stimulation. The primary actions of
the fingers and sliding them forward along the tube.
gastrin are (1) stimulation of gastric acid secretion and
Any material in front of the contractile ring is moved
(2) stimulation of growth of the gastric mucosa.
forward.
Cholecystokinin is secreted by
“I” cells in the
Peristalsis is an inherent property of many syncytial
mucosa of the duodenum and jejunum mainly in
smooth muscle tubes; stimulation at any point in the
response to digestive products of fat, fatty acids,
gut can cause a contractile ring to appear in the circu-
and monoglycerides in the intestinal contents. This
lar muscle, and this ring then spreads along the gut
hormone strongly contracts the gallbladder, expelling
tube. (Peristalsis also occurs in the bile ducts, glandu-
bile into the small intestine where the bile in turn plays
lar ducts, ureters, and many other smooth muscle tubes
important roles in emulsifying fatty substances, allow-
of the body.)
ing them to be digested and absorbed. Cholecystokinin
The usual stimulus for intestinal peristalsis is disten-
also inhibits stomach contraction moderately. There-
tion of the gut. That is, if a large amount of food col-
fore, at the same time that this hormone causes emp-
lects at any point in the gut, the stretching of the gut
tying of the gallbladder, it also slows the emptying of
wall stimulates the enteric nervous system to contract
food from the stomach to give adequate time for diges-
the gut wall 2 to 3 centimeters behind this point, and
tion of the fats in the upper intestinal tract.
a contractile ring appears that initiates a peristaltic
Secretin was the first gastrointestinal hormone dis-
movement. Other stimuli that can initiate peristalsis
covered and is secreted by the “S” cells in the mucosa
include chemical or physical irritation of the epithelial
Chapter 62
General Principles of Gastrointestinal Function—Motility, Nervous Control, and Blood Circulation
777
Peristaltic contraction
Vena cava
Leading wave of distention
Hepatic artery
Hepatic vein
Hepatic
sinuses
Aorta
Zero time
5 seconds later
Figure 62-5
Splenic
Peristalsis.
vein
Portal
vein
lining in the gut. Also, strong parasympathetic nervous
signals to the gut will elicit strong peristalsis.
Intestinal vein
Intestinal artery
Function of the Myenteric Plexus in Peristalsis. Peristalsis
occurs only weakly or not at all in any portion of the
gastrointestinal tract that has congenital absence of
the myenteric plexus. Also, it is greatly depressed or
Capillary
completely blocked in the entire gut when a person is
treated with atropine to paralyze the cholinergic nerve
Figure 62-6
endings of the myenteric plexus. Therefore, effectual
peristalsis requires an active myenteric plexus.
Splanchnic circulation.
Directional Movement of Peristaltic Waves Toward the Anus.
Peristalsis, theoretically, can occur in either direction
especially true when forward progression of the intes-
from a stimulated point, but it normally dies out
tinal contents is blocked by a sphincter, so that a
rapidly in the orad direction while continuing for a
peristaltic wave can then only churn the intestinal con-
considerable distance toward the anus. The exact cause
tents, rather than propelling them forward. At other
of this directional transmission of peristalsis has never
times, local intermittent constrictive contractions occur
been ascertained, although it probably results mainly
every few centimeters in the gut wall. These constric-
from the fact that the myenteric plexus itself is “polar-
tions usually last only 5 to 30 seconds; then new con-
ized” in the anal direction, which can be explained as
strictions occur at other points in the gut, thus
follows.
“chopping” and “shearing” the contents first here and
then there. These peristaltic and constrictive move-
Peristaltic Reflex and the “Law of the Gut.” When a
ments are modified in different parts of the gastroin-
segment of the intestinal tract is excited by distention
testinal tract for proper propulsion and mixing, as
and thereby initiates peristalsis, the contractile ring
discussed for each portion of the tract in Chapter 63.
causing the peristalsis normally begins on the orad side
of the distended segment and moves toward the dis-
tended segment, pushing the intestinal contents in the
Gastrointestinal Blood Flow—
anal direction for 5 to 10 centimeters before dying out.
At the same time, the gut sometimes relaxes several
“Splanchnic Circulation”
centimeters downstream toward the anus, which is
called “receptive relaxation,” thus allowing the food to
The blood vessels of the gastrointestinal system are
be propelled more easily anally than orad.
part of a more extensive system called the splanchnic
This complex pattern does not occur in the absence
circulation, shown in Figure 62-6. It includes the blood
of the myenteric plexus. Therefore, the complex is
flow through the gut itself plus blood flows through the
called the myenteric reflex or the peristaltic reflex. The
spleen, pancreas, and liver. The design of this system is
peristaltic reflex plus the anal direction of movement
such that all the blood that courses through the gut,
of the peristalsis is called the “law of the gut.”
spleen, and pancreas then flows immediately into the
liver by way of the portal vein. In the liver, the blood
passes through millions of minute liver sinusoids and
Mixing Movements
finally leaves the liver by way of hepatic veins that
empty into the vena cava of the general circulation.
Mixing movements differ in different parts of the ali-
This flow of blood through the liver, before it empties
mentary tract. In some areas, the peristaltic contrac-
into the vena cava, allows the reticuloendothelial cells
tions themselves cause most of the mixing. This is
that line the liver sinusoids to remove bacteria and
778
Unit XII Gastrointestinal Physiology
other particulate matter that might enter the blood
On entering the wall of the gut, the arteries branch
from the gastrointestinal tract, thus preventing direct
and send smaller arteries circling in both directions
transport of potentially harmful agents into the
around the gut, with the tips of these arteries meeting
remainder of the body.
on the side of the gut wall opposite the mesenteric
The nonfat, water-soluble nutrients absorbed from
attachment. From the circling arteries, still much
the gut (such as carbohydrates and proteins) are trans-
smaller arteries penetrate into the intestinal wall and
ported in the portal venous blood to the same liver
spread (1) along the muscle bundles, (2) into the intes-
sinusoids. Here, both the reticuloendothelial cells and
tinal villi, and (3) into submucosal vessels beneath the
the principal parenchymal cells of the liver, the hepatic
epithelium to serve the secretory and absorptive func-
cells, absorb and store temporarily from one half to
tions of the gut.
three quarters of the nutrients. Also, much chemical
Figure 62-8 shows the special organization of the
intermediary processing of these nutrients occurs in
blood flow through an intestinal villus, including a
the liver cells. We discuss these nutritional functions of
small arteriole and venule that interconnect with a
the liver in Chapters 67 through 71. Almost all of the
system of multiple looping capillaries. The walls of the
fats absorbed from the intestinal tract are not carried
arterioles are highly muscular and are highly active in
in the portal blood but instead are absorbed into the
controlling villus blood flow.
intestinal lymphatics and then conducted to the sys-
temic circulating blood by way of the thoracic duct,
Effect of Gut Activity and Metabolic
bypassing the liver.
Factors on Gastrointestinal
Blood Flow
Anatomy of the Gastrointestinal
Blood Supply
Under normal conditions, the blood flow in each area
of the gastrointestinal tract, as well as in each layer of
Figure 62-7 shows the general plan of the arterial
the gut wall, is directly related to the level of local
blood supply to the gut, including the superior mesen-
activity. For instance, during active absorption of nutri-
teric and inferior mesenteric arteries supplying the
ents, blood flow in the villi and adjacent regions of the
walls of the small and large intestines by way of an
submucosa is increased as much as eightfold. Likewise,
arching arterial system. Not shown in the figure is the
blood flow in the muscle layers of the intestinal wall
celiac artery, which provides a similar blood supply to
increases with increased motor activity in the gut. For
the stomach.
instance, after a meal, the motor activity, secretory
Aorta
Transverse
colon
Branch of
Middle colic
inferior
mesenteric
Ascending
colon
Superior
Right colic
mesenteric
Descending
colon
Ileocolic
Jejunum
Jejunal
Ileal
Figure 62-7
Arterial blood supply to the intes-
tines through the mesenteric
Ileum
web.
Chapter 62
General Principles of Gastrointestinal Function—Motility, Nervous Control, and Blood Circulation
779
100 per cent; therefore, the increased mucosal and gut
wall metabolic rate during gut activity probably lowers
the oxygen concentration enough to cause much of the
vasodilation. The decrease in oxygen can also lead to
as much as a fourfold increase of adenosine, a well-
known vasodilator that could be responsible for much
of the increased flow.
Thus, the increased blood flow during increased gas-
Central lacteal
trointestinal activity is probably a combination of
many of the aforementioned factors plus still others
yet undiscovered.
Blood capillaries
“Countercurrent” Blood Flow in the Villi. Note in Figure
62-8 that the arterial flow into the villus and the
venous flow out of the villus are in directions opposite
to each other, and that the vessels lie in close apposi-
tion to each other. Because of this vascular arrange-
ment, much of the blood oxygen diffuses out of the
arterioles directly into the adjacent venules without
ever being carried in the blood to the tips of the villi.
As much as 80 per cent of the oxygen may take this
short-circuit route and therefore not be available for
Vein
local metabolic functions of the villi. The reader will
recognize that this type of countercurrent mechanism
in the villi is analogous to the countercurrent mecha-
nism in the vasa recta of the kidney medulla, discussed
in detail in Chapter 28.
Artery
Under normal conditions, this shunting of oxygen
from the arterioles to the venules is not harmful to the
villi, but in disease conditions in which blood flow to
the gut becomes greatly curtailed, such as in circula-
tory shock, the oxygen deficit in the tips of the villi can
become so great that the villus tip or even the whole
Figure 62-8
villus suffers ischemic death and can disintegrate.
Microvasculature of the villus, showing a countercurrent arrange-
Therefore, for this reason and others, in many gas-
ment of blood flow in the arterioles and venules.
trointestinal diseases the villi become seriously
blunted, leading to greatly diminished intestinal
absorptive capacity.
activity, and absorptive activity all increase; likewise,
the blood flow increases greatly but then decreases
back to the resting level over another 2 to 4 hours.
Nervous Control of Gastrointestinal
Possible Causes of the Increased Blood Flow During Gastroin-
Blood Flow
testinal Activity. Although the precise cause or causes
of the increased blood flow during increased gastroin-
Stimulation of the parasympathetic nerves going to the
testinal activity are still unclear, some facts are known.
stomach and lower colon increases local blood flow at
First, several vasodilator substances are released
the same time that it increases glandular secretion.
from the mucosa of the intestinal tract during the
This increased flow probably results secondarily from
digestive process. Most of these are peptide hormones,
the increased glandular activity and not as a direct
including cholecystokinin, vasoactive intestinal peptide,
effect of the nervous stimulation.
gastrin, and secretin. These same hormones control
Sympathetic stimulation, by contrast, has a direct
specific motor and secretory activities of the gut, as we
effect on essentially all the gastrointestinal tract to
will see in Chapters 63 and 64.
cause intense vasoconstriction of the arterioles with
Second, some of the gastrointestinal glands also
greatly decreased blood flow. After a few minutes of
release into the gut wall two kinins, kallidin and
this vasoconstriction, the flow often returns almost to
bradykinin, at the same time that they secrete their
normal by means of a mechanism called “autoregula-
secretions into the lumen. These kinins are powerful
tory escape.” That is, the local metabolic vasodilator
vasodilators that are believed to cause much of the
mechanisms that are elicited by ischemia become pre-
increased mucosal vasodilation that occurs along with
potent over the sympathetic vasoconstriction and,
secretion.
therefore, redilate the arterioles, thus causing return of
Third, decreased oxygen concentration in the gut
necessary nutrient blood flow to the gastrointestinal
wall can increase intestinal blood flow at least 50 to
glands and muscle.
780
Unit XII Gastrointestinal Physiology
Importance of Nervous Depression of Gastrointestinal Blood
Hocker M: Molecular mechanisms of gastrin-dependent
Flow When Other Parts of the Body Need Extra Blood Flow. A
gene regulation. Ann N Y Acad Sci 1014:97, 2004.
major value of sympathetic vasoconstriction in the gut
Huizinga JD: Physiology and pathophysiology of the inter-
stitial cell of Cajal: from bench to bedside. II. Gastric
is that it allows shut-off of gastrointestinal and other
motility: lessons from mutant mice on slow waves and
splanchnic blood flow for short periods of time during
innervation. Am J Physiol Gastrointest Liver Physiol
heavy exercise, when increased flow is needed by the
281:G1129, 2001.
skeletal muscle and heart. Also, in circulatory shock,
Inui A, Asakawa A, Bowers CY, et al: Ghrelin, appetite, and
when all the body’s vital tissues are in danger of cel-
gastric motility: the emerging role of the stomach as an
lular death for lack of blood flow—especially the brain
endocrine organ. FASEB J 18:439, 2004.
and the heart—sympathetic stimulation can decrease
Johnson LR: Gastrointestinal Physiology, 6th ed. St. Louis:
splanchnic blood flow to very little for many hours.
Mosby, 2001.
Sympathetic stimulation also causes strong vaso-
Lammers WJ, Slack JR: Of slow waves and spike patches.
constriction of the large-volume intestinal and mesen-
News Physiol Sci 16:138, 2001.
Moran TH, Kinzig KP: Gastrointestinal satiety signals II.
teric veins. This decreases the volume of these veins,
Cholecystokinin. Am J Physiol Gastrointest Liver Physiol
thereby displacing large amounts of blood into other
286:G183, 2004.
parts of the circulation. In hemorrhagic shock or other
Powley TL, Phillips RJ: Musings on the wanderer: what’s new
states of low blood volume, this mechanism can
in our understanding of vago-vagal reflexes? I. Morphol-
provide as much as 200 to 400 milliliters of extra blood
ogy and topography of vagal afferents innervating the GI
to sustain the general circulation.
tract. Am J Physiol Gastrointest Liver Physiol 283:G1217,
2002.
Rehfeld JF: The new biology of gastrointestinal hormones.
References
Physiol Rev 78:1087, 1998.
Sanders KM, Ordog T, Ward SM: Physiology and patho-
Adelson DW, Million M: Tracking the moveable feast:
physiology of the interstitial cells of Cajal: from bench to
sonomicrometry and gastrointestinal motility. News
bedside. IV. Genetic and animal models of GI motility dis-
Physiol Sci 19:27, 2004.
orders caused by loss of interstitial cells of Cajal. Am J
Daniel EE: Physiology and pathophysiology of the intersti-
Physiol Gastrointest Liver Physiol 282:G747, 2002.
tial cell of Cajal: from bench to bedside. III. Interaction of
Smith GP: Satiation: From Gut to Brain. New York: Oxford
interstitial cells of Cajal with neuromediators: an interim
University Press, 1998.
assessment. Am J Physiol Gastrointest Liver Physiol
Thomas RP, Hellmich MR, Townsend CM Jr, Evers
281:G1329, 2001.
BM: Role of gastrointestinal hormones in the proliferation
Furness JB, Jones C, Nurgali K, Clerc N: Intrinsic primary
of normal and neoplastic tissues. Endocr Rev 24:571,
afferent neurons and nerve circuits within the intestine.
2003.
Prog Neurobiol 72:143, 2004.
Woods SC: Gastrointestinal satiety signals I. An overview of
Hansen MB: The enteric nervous system II: gastrointestinal
gastrointestinal signals that influence food intake. Am J
functions. Pharmacol Toxicol 92:249-57, 2003.
Physiol Gastrointest Liver Physiol 286:G7, 2004
Hobson AR, Aziz Q: Central nervous system processing of
human visceral pain in health and disease. News Physiol
Sci 18:109, 2003.
C H A P T E R
6
3
Propulsion and Mixing of
Food in the Alimentary Tract
For food to be processed optimally in the alimen-
tary tract, the time that it remains in each part of the
tract is critical. Also, appropriate mixing must be
provided. Yet because the requirements for mixing
and propulsion are quite different at each stage of
processing, multiple automatic nervous and hor-
monal feedback mechanisms control the timing of
each of these so that they will occur optimally, not
too rapidly, not too slowly.
The purpose of this chapter is to discuss these movements, especially the auto-
matic mechanisms of this control.
Ingestion of Food
The amount of food that a person ingests is determined principally by intrinsic
desire for food called hunger. The type of food that a person preferentially seeks
is determined by appetite. These mechanisms in themselves are extremely
important automatic regulatory systems for maintaining an adequate nutri-
tional supply for the body; they are discussed in Chapter 71 in relation to nutri-
tion of the body. The current discussion of food ingestion is confined to the
mechanics of ingestion, especially mastication and swallowing.
Mastication (Chewing)
The teeth are admirably designed for chewing, the anterior teeth (incisors) pro-
viding a strong cutting action and the posterior teeth (molars), a grinding action.
All the jaw muscles working together can close the teeth with a force as great
as 55 pounds on the incisors and 200 pounds on the molars.
Most of the muscles of chewing are innervated by the motor branch of the
fifth cranial nerve, and the chewing process is controlled by nuclei in the brain
stem. Stimulation of specific reticular areas in the brain stem taste centers will
cause rhythmical chewing movements. Also, stimulation of areas in the hypo-
thalamus, amygdala, and even the cerebral cortex near the sensory areas for
taste and smell can often cause chewing.
Much of the chewing process is caused by a chewing reflex, which may be
explained as follows: The presence of a bolus of food in the mouth at first ini-
tiates reflex inhibition of the muscles of mastication, which allows the lower jaw
to drop. The drop in turn initiates a stretch reflex of the jaw muscles that leads
to rebound contraction. This automatically raises the jaw to cause closure of the
teeth, but it also compresses the bolus again against the linings of the mouth,
which inhibits the jaw muscles once again, allowing the jaw to drop and rebound
another time; this is repeated again and again.
Chewing is important for digestion of all foods, but especially important for
most fruits and raw vegetables because these have indigestible cellulose mem-
branes around their nutrient portions that must be broken before the food can
be digested. Also, chewing aids the digestion of food for still another simple
reason: Digestive enzymes act only on the surfaces of food particles; therefore,
781
782
Unit XII Gastrointestinal Physiology
the rate of digestion is absolutely dependent on the
epithelial swallowing receptor areas all around the
total surface area exposed to the digestive secretions.
opening of the pharynx, especially on the tonsillar
In addition, grinding the food to a very fine particulate
pillars, and impulses from these pass to the brain stem
consistency prevents excoriation of the gastrointesti-
to initiate a series of automatic pharyngeal muscle
nal tract and increases the ease with which food is
contractions as follows:
emptied from the stomach into the small intestine,
1.
The soft palate is pulled upward to close the
then into all succeeding segments of the gut.
posterior nares, to prevent reflux of food into the
nasal cavities.
2.
The palatopharyngeal folds on each side of the
Swallowing (Deglutition)
pharynx are pulled medially to approximate each
other. In this way, these folds form a sagittal
Swallowing is a complicated mechanism, principally
slit through which the food must pass into the
because the pharynx subserves respiration as well as
posterior pharynx. This slit performs a selective
swallowing. The pharynx is converted for only a few
action, allowing food that has been masticated
seconds at a time into a tract for propulsion of food.
sufficiently to pass with ease. Because this stage of
It is especially important that respiration not be com-
swallowing lasts less than 1 second, any large
promised because of swallowing.
object is usually impeded too much to pass into
In general, swallowing can be divided into (1) a vol-
the esophagus.
untary stage, which initiates the swallowing process; (2)
3.
The vocal cords of the larynx are strongly
a pharyngeal stage, which is involuntary and consti-
approximated, and the larynx is pulled upward
tutes passage of food through the pharynx into the
and anteriorly by the neck muscles. These actions,
esophagus; and (3) an esophageal stage, another invol-
combined with the presence of ligaments that
untary phase that transports food from the pharynx to
prevent upward movement of the epiglottis, cause
the stomach.
the epiglottis to swing backward over the opening
of the larynx. All these effects acting together
Voluntary Stage of Swallowing. When the food is ready for
prevent passage of food into the nose and
swallowing, it is “voluntarily” squeezed or rolled pos-
trachea. Most essential is the tight approximation
teriorly into the pharynx by pressure of the tongue
of the vocal cords, but the epiglottis helps to
upward and backward against the palate, as shown in
prevent food from ever getting as far as the vocal
Figure
63-1. From here on, swallowing becomes
cords. Destruction of the vocal cords or of the
entirely—or almost entirely—automatic and ordinar-
muscles that approximate them can cause
ily cannot be stopped.
strangulation.
4.
The upward movement of the larynx also pulls
Pharyngeal Stage of Swallowing. As the bolus of food
up and enlarges the opening to the esophagus.
enters the posterior mouth and pharynx, it stimulates
At the same time, the upper 3 to 4 centimeters
of the esophageal muscular wall, called the
upper esophageal sphincter (also called the
Vagus
Glossopharyngeal
pharyngoesophageal sphincter) relaxes, thus
nerve
allowing food to move easily and freely from the
posterior pharynx into the upper esophagus.
Trigeminal nerve
Between swallows, this sphincter remains strongly
contracted, thereby preventing air from going
Swallowing
into the esophagus during respiration. The
center
upward movement of the larynx also lifts the
Medulla
glottis out of the main stream of food flow, so
Bolus of food
that the food mainly passes on each side of the
Uvula
epiglottis rather than over its surface; this adds
Pharynx
still another protection against entry of food into
Epiglottis
the trachea.
5.
Once the larynx is raised and the
Vocal cords
pharyngoesophageal sphincter becomes relaxed,
Esophagus
the entire muscular wall of the pharynx contracts,
beginning in the superior part of the pharynx,
then spreading downward over the middle and
inferior pharyngeal areas, which propels the food
Peristalsis
by peristalsis into the esophagus.
To summarize the mechanics of the pharyngeal
stage of swallowing: The trachea is closed, the esoph-
agus is opened, and a fast peristaltic wave initiated by
the nervous system of the pharynx forces the bolus of
Figure 63-1
food into the upper esophagus, the entire process
Swallowing mechanism.
occurring in less than 2 seconds.
Chapter 63
Propulsion and Mixing of Food in the Alimentary Tract
783
Nervous Initiation of the Pharyngeal Stage of Swallow-
the esophagus itself by the retained food; these waves
ing. The most sensitive tactile areas of the posterior
continue until all the food has emptied into the
mouth and pharynx for initiating the pharyngeal stage
stomach. The secondary peristaltic waves are initiated
of swallowing lie in a ring around the pharyngeal
partly by intrinsic neural circuits in the myenteric
opening, with greatest sensitivity on the tonsillar
nervous system and partly by reflexes that begin in the
pillars. Impulses are transmitted from these areas
pharynx and are then transmitted upward through
through the sensory portions of the trigeminal and
vagal afferent fibers to the medulla and back again to
glossopharyngeal nerves into the medulla oblongata,
the esophagus through glossopharyngeal and vagal
either into or closely associated with the tractus soli-
efferent nerve fibers.
tarius, which receives essentially all sensory impulses
The musculature of the pharyngeal wall and upper
from the mouth.
third of the esophagus is striated muscle. Therefore, the
The successive stages of the swallowing process
peristaltic waves in these regions are controlled by
are then automatically initiated in orderly sequence
skeletal nerve impulses from the glossopharyngeal and
by neuronal areas of the reticular substance of the
vagus nerves. In the lower two thirds of the esophagus,
medulla and lower portion of the pons. The sequence
the musculature is smooth muscle, but this portion of
of the swallowing reflex is the same from one swallow
the esophagus is also strongly controlled by the vagus
to the next, and the timing of the entire cycle also
nerves acting through connections with the esophageal
remains constant from one swallow to the next. The
myenteric nervous system. When the vagus nerves to
areas in the medulla and lower pons that control
the esophagus are cut, the myenteric nerve plexus of
swallowing are collectively called the deglutition or
the esophagus becomes excitable enough after several
swallowing center.
days to cause strong secondary peristaltic waves even
The motor impulses from the swallowing center to
without support from the vagal reflexes. Therefore,
the pharynx and upper esophagus that cause swallow-
even after paralysis of the brain stem swallowing
ing are transmitted successively by the 5th, 9th, 10th,
reflex, food fed by tube or in some other way into the
and 12th cranial nerves and even a few of the superior
esophagus still passes readily into the stomach.
cervical nerves.
In summary, the pharyngeal stage of swallowing is
Receptive Relaxation of the Stomach. When the
principally a reflex act. It is almost always initiated by
esophageal peristaltic wave approaches toward the
voluntary movement of food into the back of the
stomach, a wave of relaxation, transmitted through
mouth, which in turn excites involuntary pharyngeal
myenteric inhibitory neurons, precedes the peristalsis.
sensory receptors to elicit the swallowing reflex.
Furthermore, the entire stomach and, to a lesser
extent, even the duodenum become relaxed as this
Effect of the Pharyngeal Stage of Swallowing on Res-
wave reaches the lower end of the esophagus and
piration. The entire pharyngeal stage of swallowing
thus are prepared ahead of time to receive the food
usually occurs in less than 6 seconds, thereby inter-
propelled into the esophagus during the swallowing
rupting respiration for only a fraction of a usual res-
act.
piratory cycle. The swallowing center specifically
inhibits the respiratory center of the medulla during
Function of the Lower Esophageal Sphincter (Gastro-
this time, halting respiration at any point in its cycle to
esophageal Sphincter). At the lower end of the esoph-
allow swallowing to proceed. Yet, even while a person
agus, extending upward about 3 centimeters above its
is talking, swallowing interrupts respiration for such a
juncture with the stomach, the esophageal circular
short time that it is hardly noticeable.
muscle functions as a broad lower esophageal sphinc-
ter, also called the gastroesophageal sphincter. This
Esophageal Stage of Swallowing. The esophagus functions
sphincter normally remains tonically constricted with
primarily to conduct food rapidly from the pharynx to
an intraluminal pressure at this point in the esophagus
the stomach, and its movements are organized specif-
of about 30 mm Hg, in contrast to the midportion of
ically for this function.
the esophagus, which normally remains relaxed. When
The esophagus normally exhibits two types of peri-
a peristaltic swallowing wave passes down the esoph-
staltic movements: primary peristalsis and secondary
agus, there is
“receptive relaxation” of the lower
peristalsis. Primary peristalsis is simply continuation of
esophageal sphincter ahead of the peristaltic wave,
the peristaltic wave that begins in the pharynx and
which allows easy propulsion of the swallowed food
spreads into the esophagus during the pharyngeal
into the stomach. Rarely, the sphincter does not relax
stage of swallowing. This wave passes all the way from
satisfactorily, resulting in a condition called achalasia,
the pharynx to the stomach in about 8 to 10 seconds.
which is discussed in Chapter 66.
Food swallowed by a person who is in the upright posi-
The stomach secretions are highly acidic and
tion is usually transmitted to the lower end of the
contain many proteolytic enzymes. The esophageal
esophagus even more rapidly than the peristaltic wave
mucosa, except in the lower one eighth of the esoph-
itself, in about 5 to 8 seconds, because of the additional
agus, is not capable of resisting for long the digestive
effect of gravity pulling the food downward.
action of gastric secretions. Fortunately, the tonic con-
If the primary peristaltic wave fails to move into the
striction of the lower esophageal sphincter helps to
stomach all the food that has entered the esophagus,
prevent significant reflux of stomach contents into the
secondary peristaltic waves result from distention of
esophagus except under very abnormal conditions.
784
Unit XII Gastrointestinal Physiology
Additional Prevention of Esophageal Reflux by Valve-
and the oldest food lying nearest the outer wall of the
like Closure of the Distal End of the Esophagus.
stomach. Normally, when food stretches the stomach,
Another factor that helps to prevent reflux is a valve-
a “vagovagal reflex” from the stomach to the brain
like mechanism of a short portion of the esophagus
stem and then back to the stomach reduces the tone
that extends slightly into the stomach. Increased intra-
in the muscular wall of the body of the stomach so that
abdominal pressure caves the esophagus inward at this
the wall bulges progressively outward, accommodating
point. Thus, this valvelike closure of the lower esoph-
greater and greater quantities of food up to a limit in
agus helps to prevent high intra-abdominal pressure
the completely relaxed stomach of 0.8 to 1.5 liters. The
from forcing stomach contents backward into the
pressure in the stomach remains low until this limit is
esophagus. Otherwise, every time we walked, coughed,
approached.
or breathed hard, we might expel stomach acid into the
esophagus.
Mixing and Propulsion of Food in the
Stomach—The Basic Electrical
Motor Functions of the
Rhythm of the Stomach Wall
Stomach
The digestive juices of the stomach are secreted by
The motor functions of the stomach are threefold: (1)
gastric glands, which are present in almost the entire
storage of large quantities of food until the food can
wall of the body of the stomach except along a narrow
be processed in the stomach, duodenum, and lower
strip on the lesser curvature of the stomach. These
intestinal tract; (2) mixing of this food with gastric
secretions come immediately into contact with that
secretions until it forms a semifluid mixture called
portion of the stored food lying against the mucosal
chyme; and (3) slow emptying of the chyme from the
surface of the stomach. As long as food is in the
stomach into the small intestine at a rate suitable for
stomach, weak peristaltic constrictor waves, called
proper digestion and absorption by the small intestine.
mixing waves, begin in the mid- to upper portions of
Figure
63-2 shows the basic anatomy of the
the stomach wall and move toward the antrum about
stomach. Anatomically, the stomach is usually divided
once every 15 to 20 seconds. These waves are initiated
into two major parts: (1) the body and (2) the antrum.
by the gut wall basic electrical rhythm, which was dis-
Physiologically, it is more appropriately divided into
cussed in Chapter 62, consisting of electrical “slow
(1) the “orad” portion, comprising about the first two
waves” that occur spontaneously in the stomach wall.
thirds of the body, and (2) the “caudad” portion, com-
As the constrictor waves progress from the body of the
prising the remainder of the body plus the antrum.
stomach into the antrum, they become more intense,
some becoming extremely intense and providing pow-
erful peristaltic action potential-driven constrictor
Storage Function of the Stomach
rings that force the antral contents under higher and
higher pressure toward the pylorus.
As food enters the stomach, it forms concentric circles
These constrictor rings also play an important role
of the food in the orad portion of the stomach, the
in mixing the stomach contents in the following way:
newest food lying closest to the esophageal opening
Each time a peristaltic wave passes down the antral
wall toward the pylorus, it digs deeply into the food
contents in the antrum. Yet the opening of the pylorus
Esophagus
is still small enough that only a few milliliters or less
Fundus
of antral contents are expelled into the duodenum
with each peristaltic wave. Also, as each peristaltic
wave approaches the pylorus, the pyloric muscle itself
Cardia
often contracts, which further impedes emptying
through the pylorus. Therefore, most of the antral con-
tents are squeezed upstream through the peristaltic
ring toward the body of the stomach, not through the
Angular
pylorus. Thus, the moving peristaltic constrictive ring,
notch
Pyloric
combined with this upstream squeezing action, called
Pylorus sphincter
Body
“retropulsion,” is an exceedingly important mixing
mechanism in the stomach.
Chyme. After food in the stomach has become thor-
oughly mixed with the stomach secretions, the result-
ing mixture that passes down the gut is called chyme.
The degree of fluidity of the chyme leaving the
Duodenum
Antrum
Rugae
stomach depends on the relative amounts of food,
water, and stomach secretions and on the degree of
Figure 63-2
digestion that has occurred. The appearance of chyme
Physiologic anatomy of the stomach.
is that of a murky semifluid or paste.
Chapter 63
Propulsion and Mixing of Food in the Alimentary Tract
785
Hunger Contractions. Besides the peristaltic contractions
and other fluids to empty from the stomach into the
that occur when food is present in the stomach,
duodenum with ease. Conversely, the constriction
another type of intense contractions, called hunger
usually prevents passage of food particles until they
contractions, often occurs when the stomach has been
have become mixed in the chyme to almost fluid con-
empty for several hours or more. They are rhythmical
sistency. The degree of constriction of the pylorus is
peristaltic contractions in the body of the stomach.
increased or decreased under the influence of nervous
When the successive contractions become extremely
and humoral reflex signals from both the stomach and
strong, they often fuse to cause a continuing tetanic
the duodenum, as discussed shortly.
contraction that sometimes lasts for 2 to 3 minutes.
Hunger contractions are most intense in young,
healthy people who have high degrees of gastroin-
Regulation of Stomach Emptying
testinal tonus; they are also greatly increased by the
person’s having lower than normal levels of blood
The rate at which the stomach empties is regulated by
sugar. When hunger contractions occur in the stomach,
signals from both the stomach and the duodenum.
the person sometimes experiences mild pain in the pit
However, the duodenum provides by far the more
of the stomach, called hunger pangs. Hunger pangs
potent of the signals, controlling the emptying of
usually do not begin until 12 to 24 hours after the last
chyme into the duodenum at a rate no greater than the
ingestion of food; in starvation, they reach their great-
rate at which the chyme can be digested and absorbed
est intensity in 3 to 4 days and gradually weaken in
in the small intestine.
succeeding days.
Gastric Factors That Promote Emptying
Effect of Gastric Food Volume on Rate of Emptying. Increased
Stomach Emptying
food volume in the stomach promotes increased emp-
tying from the stomach. But this increased emptying
Stomach emptying is promoted by intense peristaltic
does not occur for the reasons that one would expect.
contractions in the stomach antrum. At the same time,
It is not increased storage pressure of the food in the
emptying is opposed by varying degrees of resistance
stomach that causes the increased emptying because,
to passage of chyme at the pylorus.
in the usual normal range of volume, the increase
in volume does not increase the pressure much.
Intense Antral Peristaltic Contractions During Stomach Empty-
However, stretching of the stomach wall does elicit
ing—“Pyloric Pump.” Most of the time, the rhythmical
local myenteric reflexes in the wall that greatly accen-
stomach contractions are weak and function mainly to
tuate activity of the pyloric pump and at the same time
cause mixing of food and gastric secretions. However,
inhibit the pylorus.
for about 20 per cent of the time while food is in the
stomach, the contractions become intense, beginning
Effect of the Hormone Gastrin on Stomach Emptying. In
in midstomach and spreading through the caudad
Chapter 64, we shall see that stomach wall stretch and
stomach no longer as weak mixing contractions but as
the presence of certain types of foods in the stomach—
strong peristaltic, very tight ringlike constrictions that
particularly digestive products of meat—elicit release
can cause stomach emptying. As the stomach becomes
of a hormone called gastrin from the antral mucosa.
progressively more and more empty, these constric-
This has potent effects to cause secretion of highly
tions begin farther and farther up the body of the
acidic gastric juice by the stomach glands. Gastrin also
stomach, gradually pinching off the food in the body
has mild to moderate stimulatory effects on motor
of the stomach and adding this food to the chyme in
functions in the body of the stomach. Most important,
the antrum. These intense peristaltic contractions
it seems to enhance the activity of the pyloric pump.
often create 50 to 70 centimeters of water pressure,
Thus, it, too, probably promotes stomach emptying.
which is about six times as powerful as the usual
mixing type of peristaltic waves.
Powerful Duodenal Factors That Inhibit
When pyloric tone is normal, each strong peristaltic
Stomach Emptying
wave forces up to several milliliters of chyme into the
Inhibitory Effect of Enterogastric Nervous Reflexes from the
duodenum. Thus, the peristaltic waves, in addition to
Duodenum. When food enters the duodenum, multiple
causing mixing in the stomach, also provide a pumping
nervous reflexes are initiated from the duodenal wall
action called the “pyloric pump.”
that pass back to the stomach to slow or even stop
stomach emptying if the volume of chyme in the duo-
Role of the Pylorus in Controlling Stomach Emptying. The
denum becomes too much. These reflexes are medi-
distal opening of the stomach is the pylorus. Here the
ated by three routes: (1) directly from the duodenum
thickness of the circular wall muscle becomes 50 to 100
to the stomach through the enteric nervous system
per cent greater than in the earlier portions of the
in the gut wall, (2) through extrinsic nerves that go to
stomach antrum, and it remains slightly tonically con-
the prevertebral sympathetic ganglia and then back
tracted almost all the time. Therefore, the pyloric cir-
through inhibitory sympathetic nerve fibers to the
cular muscle is called the pyloric sphincter.
stomach, and (3) probably to a slight extent through
Despite normal tonic contraction of the pyloric
the vagus nerves all the way to the brain stem, where
sphincter, the pylorus usually is open enough for water
they inhibit the normal excitatory signals transmitted
786
Unit XII Gastrointestinal Physiology
to the stomach through the vagi. All these parallel
hormone acts as an inhibitor to block increased
reflexes have two effects on stomach emptying: first,
stomach motility caused by gastrin.
they strongly inhibit the “pyloric pump” propulsive
Other possible inhibitors of stomach emptying are
contractions, and second, they increase the tone of the
the hormones secretin and gastric inhibitory peptide
pyloric sphincter.
(GIP). Secretin is released mainly from the duodenal
The types of factors that are continually monitored
mucosa in response to gastric acid passed from the
in the duodenum and that can initiate enterogastric
stomach through the pylorus. GIP has a general but
inhibitory reflexes include the following:
weak effect of decreasing gastrointestinal motility.
1. The degree of distention of the duodenum
GIP is released from the upper small intestine in
2. The presence of any degree of irritation of the
response mainly to fat in the chyme, but to a lesser
duodenal mucosa
extent to carbohydrates as well. Although GIP does
3. The degree of acidity of the duodenal chyme
inhibit gastric motility under some conditions, its effect
4. The degree of osmolality of the chyme
at physiologic concentrations is probably mainly to
5. The presence of certain breakdown products in
stimulate secretion of insulin by the pancreas.
the chyme, especially breakdown products of
These hormones are discussed at greater length
proteins and perhaps to a lesser extent of fats
elsewhere in this text, especially in Chapter 64 in rela-
The enterogastric inhibitory reflexes are especially
tion to control of gallbladder emptying and control of
sensitive to the presence of irritants and acids in the
rate of pancreatic secretion.
duodenal chyme, and they often become strongly acti-
In summary, hormones, especially CCK, can inhibit
vated within as little as 30 seconds. For instance, when-
gastric emptying when excess quantities of chyme,
ever the pH of the chyme in the duodenum falls below
especially acidic or fatty chyme, enter the duodenum
about 3.5 to 4, the reflexes frequently block further
from the stomach.
release of acidic stomach contents into the duodenum
until the duodenal chyme can be neutralized by pan-
Summary of the Control of Stomach Emptying
creatic and other secretions.
Emptying of the stomach is controlled only to a mod-
Breakdown products of protein digestion also elicit
erate degree by stomach factors such as the degree
inhibitory enterogastric reflexes; by slowing the rate of
of filling in the stomach and the excitatory effect of
stomach emptying, sufficient time is ensured for ade-
gastrin on stomach peristalsis. Probably the more
quate protein digestion in the duodenum and small
important control of stomach emptying resides in
intestine.
inhibitory feedback signals from the duodenum,
Finally, either hypotonic or hypertonic fluids (espe-
including both enterogastric inhibitory nervous feed-
cially hypertonic) elicit the inhibitory reflexes. Thus,
back reflexes and hormonal feedback by CCK. These
too rapid flow of nonisotonic fluids into the small
feedback inhibitory mechanisms work together to
intestine is prevented, thereby also preventing rapid
slow the rate of emptying when (1) too much chyme
changes in electrolyte concentrations in the whole-
is already in the small intestine or (2) the chyme is
body extracellular fluid during absorption of the intes-
excessively acidic, contains too much unprocessed
tinal contents.
protein or fat, is hypotonic or hypertonic, or is irritat-
ing. In this way, the rate of stomach emptying is limited
Hormonal Feedback from the Duodenum Inhibits Gastric Emp-
to that amount of chyme that the small intestine can
tying—Role of Fats and the Hormone Cholecystokinin. Not
process.
only do nervous reflexes from the duodenum to the
stomach inhibit stomach emptying, but hormones
released from the upper intestine do so as well. The
Movements of the
stimulus for releasing these inhibitory hormones is
Small Intestine
mainly fats entering the duodenum, although other
types of foods can increase the hormones to a lesser
The movements of the small intestine, like those else-
degree.
where in the gastrointestinal tract, can be divided into
On entering the duodenum, the fats extract several
mixing contractions and propulsive contractions. To a
different hormones from the duodenal and jejunal
great extent, this separation is artificial because essen-
epithelium, either by binding with “receptors” on
tially all movements of the small intestine cause at
the epithelial cells or in some other way. In turn, the
least some degree of both mixing and propulsion. The
hormones are carried by way of the blood to the
usual classification of these processes is the following.
stomach, where they inhibit the pyloric pump and at
the same time increase the strength of contraction of
the pyloric sphincter. These effects are important
Mixing Contractions (Segmentation
because fats are much slower to be digested than most
Contractions)
other foods.
Precisely which hormones cause the hormonal
When a portion of the small intestine becomes dis-
feedback inhibition of the stomach is not fully clear.
tended with chyme, stretching of the intestinal wall
The most potent appears to be cholecystokinin (CCK),
elicits localized concentric contractions spaced at
which is released from the mucosa of the jejunum in
intervals along the intestine and lasting a fraction of a
response to fatty substances in the chyme. This
minute. The contractions cause “segmentation” of the
Chapter 63
Propulsion and Mixing of Food in the Alimentary Tract
787
averages only 1 cm/min. This means that 3 to 5 hours
are required for passage of chyme from the pylorus to
Regularly spaced
the ileocecal valve.
Control of Peristalsis by Nervous and Hormonal Signals. Peri-
Isolated
staltic activity of the small intestine is greatly increased
after a meal. This is caused partly by the beginning
entry of chyme into the duodenum causing stretch of
the duodenal wall, but also by a so-called gastroenteric
Irregularly spaced
reflex that is initiated by distention of the stomach and
conducted principally through the myenteric plexus
from the stomach down along the wall of the small
Weak regularly spaced
intestine.
In addition to the nervous signals that may affect
Figure 63-3
small intestinal peristalsis, several hormonal factors
also affect peristalsis. They include gastrin, CCK,
Segmentation movements of the small intestine.
insulin, motilin, and serotonin, all of which enhance
intestinal motility and are secreted during various
phases of food processing. Conversely, secretin and
small intestine, as shown in Figure 63-3. That is, they
glucagon inhibit small intestinal motility. The physio-
divide the intestine into spaced segments that have the
logic importance of each of these hormonal factors for
appearance of a chain of sausages. As one set of seg-
controlling motility is still questionable.
mentation contractions relaxes, a new set often begins,
The function of the peristaltic waves in the small
but the contractions this time occur mainly at new
intestine is not only to cause progression of chyme
points between the previous contractions. Therefore,
toward the ileocecal valve but also to spread out the
the segmentation contractions “chop” the chyme two
chyme along the intestinal mucosa. As the chyme
to three times per minute, in this way promoting pro-
enters the intestines from the stomach and elicits peri-
gressive mixing of the food with secretions of the small
stalsis, this immediately spreads the chyme along the
intestine.
intestine; and this process intensifies as additional
The maximum frequency of the segmentation con-
chyme enters the duodenum. On reaching the ileoce-
tractions in the small intestine is determined by the
cal valve, the chyme is sometimes blocked for several
frequency of electrical slow waves in the intestinal wall,
hours until the person eats another meal; at that time,
which is the basic electrical rhythm described in
a gastroileal reflex intensifies peristalsis in the ileum
Chapter 62. Because this frequency normally is not
and forces the remaining chyme through the ileocecal
over 12 per minute in the duodenum and proximal
valve into the cecum of the large intestine.
jejunum, the maximum frequency of the segmentation
contractions in these areas is also about 12 per minute,
Propulsive Effect of the Segmentation Movements. The seg-
but this occurs only under extreme conditions of stim-
mentation movements, although lasting for only a few
ulation. In the terminal ileum, the maximum frequency
seconds at a time, often also travel 1 centimeter or so
is usually 8 to 9 contractions per minute.
in the anal direction and during that time help propel
The segmentation contractions become exceedingly
the food down the intestine. The difference between
weak when the excitatory activity of the enteric
the segmentation and the peristaltic movements is not
nervous system is blocked by the drug atropine. There-
as great as might be implied by their separation into
fore, even though it is the slow waves in the smooth
these two classifications.
muscle itself that cause the segmentation contractions,
these contractions are not effective without back-
Peristaltic Rush. Although peristalsis in the small
ground excitation mainly from the myenteric nerve
intestine is normally weak, intense irritation of the
plexus.
intestinal mucosa, as occurs in some severe cases of
infectious diarrhea, can cause both powerful and rapid
peristalsis, called the peristaltic rush. This is initiated
Propulsive Movements
partly by nervous reflexes that involve the autonomic
nervous system and brain stem and partly by intrinsic
Peristalsis in the Small Intestine. Chyme is propelled
enhancement of the myenteric plexus reflexes within
through the small intestine by peristaltic waves. These
the gut wall itself. The powerful peristaltic contractions
can occur in any part of the small intestine, and they
travel long distances in the small intestine within
move toward the anus at a velocity of 0.5 to 2.0 cm/sec,
minutes, sweeping the contents of the intestine into the
faster in the proximal intestine and slower in the ter-
colon and thereby relieving the small intestine of irri-
minal intestine. They normally are very weak and
tative chyme and excessive distention.
usually die out after traveling only 3 to 5 centimeters,
very rarely farther than 10 centimeters, so that forward
Movements Caused by the Muscularis Mucosae and Muscle Fibers
movement of the chyme is very slow, so slow in fact
of the Villi. The muscularis mucosae can cause short
that net movement along the small intestine normally
folds to appear in the intestinal mucosa. In addition,
788
Unit XII Gastrointestinal Physiology
individual fibers from this muscle extend into the intes-
Feedback Control of the Ileocecal Sphincter. The degree of
tinal villi and cause them to contract intermittently. The
contraction of the ileocecal sphincter and the intensity
mucosal folds increase the surface area exposed to the
of peristalsis in the terminal ileum are controlled sig-
chyme, thereby increasing absorption. Also, contrac-
nificantly by reflexes from the cecum. When the cecum
tions of the villi—shortening, elongating, and shorten-
is distended, contraction of the ileocecal sphincter
ing again—“milk” the villi, so that lymph flows freely
becomes intensified and ileal peristalsis is inhibited,
from the central lacteals of the villi into the lymphatic
both of which greatly delay emptying of additional
system. These mucosal and villous contractions are ini-
chyme into the cecum from the ileum. Also, any irri-
tiated mainly by local nervous reflexes in the submu-
cosal nerve plexus that occur in response to chyme in
tant in the cecum delays emptying. For instance, when
the small intestine.
a person has an inflamed appendix, the irritation of
this vestigial remnant of the cecum can cause such
intense spasm of the ileocecal sphincter and partial
Function of the Ileocecal Valve
paralysis of the ileum that these effects together block
emptying of the ileum into the cecum. The reflexes
A principal function of the ileocecal valve is to prevent
from the cecum to the ileocecal sphincter and ileum
backflow of fecal contents from the colon into the
are mediated both by way of the myenteric plexus in
small intestine. As shown in Figure 63-4, the ileocecal
the gut wall itself and of the extrinsic autonomic
valve itself protrudes into the lumen of the cecum and
nerves, especially by way of the prevertebral sympa-
therefore is forcefully closed when excess pressure
thetic ganglia.
builds up in the cecum and tries to push cecal contents
backward against the valve lips. The valve usually can
resist reverse pressure of at least 50 to 60 centimeters
Movements of the Colon
of water.
In addition, the wall of the ileum for several cen-
The principal functions of the colon are (1) absorption
timeters immediately upstream from the ileocecal
of water and electrolytes from the chyme to form solid
valve has a thickened circular muscle called the ileo-
feces and (2) storage of fecal matter until it can be
cecal sphincter. This sphincter normally remains mildly
expelled. The proximal half of the colon, shown in
constricted and slows emptying of ileal contents into
Figure 63-5, is concerned principally with absorption,
the cecum. However, immediately after a meal, a gas-
and the distal half with storage. Because intense colon
troileal reflex (described earlier) intensifies peristalsis
wall movements are not required for these functions,
in the ileum, and emptying of ileal contents into the
the movements of the colon are normally very slug-
cecum proceeds.
gish. Yet in a sluggish manner, the movements still
Resistance to emptying at the ileocecal valve pro-
have characteristics similar to those of the small intes-
longs the stay of chyme in the ileum and thereby
tine and can be divided once again into mixing move-
facilitates absorption. Normally, only 1500 to 2000 mil-
ments and propulsive movements.
liliters of chyme empty into the cecum each day.
Mixing Movements—“Haustrations.” In the same manner
that segmentation movements occur in the small
intestine, large circular constrictions occur in the large
Pressure and chemical
Semi-
irritation relax sphincter
mush
and excite peristalsis
Mush
Semifluid
Fluidity of contents
Colon
promotes emptying
Poor motility causes
greater absorption, and
Valve
Fluid
hard feces in transverse
colon causes
Semi-
constipation
solid
Ileum
Ileocecal
valve
Ileocecal sphincter
Pressure or chemical irritation
in cecum inhibits peristalsis
Solid
Excess motility causes
of ileum and excites sphincter
less absorption and
diarrhea or loose feces
Figure 63-4
Figure 63-5
Emptying at the ileocecal valve.
Absorptive and storage functions of the large intestine.
Chapter 63
Propulsion and Mixing of Food in the Alimentary Tract
789
intestine. At each of these constrictions, about 2.5 cen-
to the colon have been removed; therefore, the reflexes
timeters of the circular muscle contracts, sometimes
almost certainly are transmitted by way of the auto-
constricting the lumen of the colon almost to occlu-
nomic nervous system.
sion. At the same time, the longitudinal muscle of the
Irritation in the colon can also initiate intense mass
colon, which is aggregated into three longitudinal
movements. For instance, a person who has an ulcer-
strips called the teniae coli, contracts. These combined
ated condition of the colon mucosa (ulcerative colitis)
contractions of the circular and longitudinal strips of
frequently has mass movements that persist almost all
muscle cause the unstimulated portion of the large
the time.
intestine to bulge outward into baglike sacs called
haustrations.
Each haustration usually reaches peak intensity in
Defecation
about 30 seconds and then disappears during the next
60 seconds. They also at times move slowly toward the
Most of the time, the rectum is empty of feces. This
anus during contraction, especially in the cecum and
results partly from the fact that a weak functional
ascending colon, and thereby provide a minor amount
sphincter exists about 20 centimeters from the anus
of forward propulsion of the colonic contents. After
at the juncture between the sigmoid colon and the
another few minutes, new haustral contractions occur
rectum. There is also a sharp angulation here that con-
in other areas nearby. Therefore, the fecal material in
tributes additional resistance to filling of the rectum.
the large intestine is slowly dug into and rolled over in
When a mass movement forces feces into the
much the same manner that one spades the earth. In
rectum, the desire for defecation occurs immediately,
this way, all the fecal material is gradually exposed to
including reflex contraction of the rectum and relax-
the mucosal surface of the large intestine, and fluid and
ation of the anal sphincters.
dissolved substances are progressively absorbed until
Continual dribble of fecal matter through the anus
only 80 to 200 milliliters of feces are expelled each day.
is prevented by tonic constriction of (1) an internal
anal sphincter, a several-centimeters-long thickening
Propulsive Movements—“Mass Movements.” Much of the
of the circular smooth muscle that lies immediately
propulsion in the cecum and ascending colon results
inside the anus, and (2) an external anal sphincter, com-
from the slow but persistent haustral contractions,
posed of striated voluntary muscle that both surrounds
requiring as many as 8 to 15 hours to move the chyme
the internal sphincter and extends distal to it. The
from the ileocecal valve through the colon, while the
external sphincter is controlled by nerve fibers in the
chyme itself becomes fecal in quality, a semisolid slush
pudendal nerve, which is part of the somatic nervous
instead of semifluid.
system and therefore is under voluntary, conscious
From the cecum to the sigmoid, mass movements
or at least subconscious control; subconsciously,
can, for many minutes at a time, take over the propul-
the external sphincter is usually kept continuously
sive role. These movements usually occur only one to
constricted unless conscious signals inhibit the
three times each day, in many people especially for
constriction.
about 15 minutes during the first hour after eating
breakfast.
Defecation Reflexes. Ordinarily, defecation is initiated
A mass movement is a modified type of peristalsis
by defecation reflexes. One of these reflexes is an intrin-
characterized by the following sequence of events:
sic reflex mediated by the local enteric nervous system
First, a constrictive ring occurs in response to a dis-
in the rectal wall. This can be described as follows:
tended or irritated point in the colon, usually in the
When feces enter the rectum, distention of the rectal
transverse colon. Then, rapidly, the 20 or more cen-
wall initiates afferent signals that spread through the
timeters of colon distal to the constrictive ring lose their
myenteric plexus to initiate peristaltic waves in the
haustrations and instead contract as a unit, propelling
descending colon, sigmoid, and rectum, forcing feces
the fecal material in this segment en masse further
toward the anus. As the peristaltic wave approaches
down the colon. The contraction develops progres-
the anus, the internal anal sphincter is relaxed by
sively more force for about 30 seconds, and relaxation
inhibitory signals from the myenteric plexus; if the
occurs during the next 2 to 3 minutes. Then, another
external anal sphincter is also consciously, voluntarily
mass movement occurs, this time perhaps farther along
relaxed at the same time, defecation occurs.
the colon.
The intrinsic myenteric defecation reflex function-
A series of mass movements usually persists for 10
ing by itself normally is relatively weak. To be effec-
to 30 minutes. Then they cease but return perhaps a
tive in causing defecation, it usually must be fortified
half day later. When they have forced a mass of feces
by another type of defecation reflex, a parasympathetic
into the rectum, the desire for defecation is felt.
defecation reflex that involves the sacral segments of
the spinal cord, shown in Figure 63-6. When the nerve
Initiation of Mass Movements by Gastrocolic and Duo-
endings in the rectum are stimulated, signals are trans-
denocolic Reflexes. Appearance of mass movements
mitted first into the spinal cord and then reflexly back
after meals is facilitated by gastrocolic and duodeno-
to the descending colon, sigmoid, rectum, and anus by
colic reflexes. These reflexes result from distention of
way of parasympathetic nerve fibers in the pelvic
the stomach and duodenum. They occur either not at
nerves. These parasympathetic signals greatly intensify
all or hardly at all when the extrinsic autonomic nerves
the peristaltic waves as well as relax the internal anal
790
Unit XII Gastrointestinal Physiology
Descending
discussed in this chapter, several other important
From
colon
nervous reflexes also can affect the overall degree of
conscious
bowel activity. They are the peritoneointestinal reflex,
cortex
Afferent
renointestinal reflex, and vesicointestinal reflex.
nerve
The peritoneointestinal reflex results from irritation
fibers
of the peritoneum; it strongly inhibits the excitatory
Parasympathetic
enteric nerves and thereby can cause intestinal paral-
nerve fibers
ysis, especially in patients with peritonitis. The reno-
(pelvic nerves)
intestinal and vesicointestinal reflexes inhibit intestinal
activity as a result of kidney or bladder irritation.
Sigmoid
References
Skeletal
colon
motor nerve
Rectum
Adelson DW, Million M: Tracking the moveable feast:
sonomicrometry and gastrointestinal motility. News
External anal sphincter
Physiol Sci 19:27, 2004.
Internal anal sphincter
Chelimsky G, Chelimsky TC: Evaluation and treatment of
autonomic disorders of the gastrointestinal tract. Semin
Figure 63-6
Neurol 23:453, 2003.
Cooke HJ, Wunderlich J, Christofi FL: “The force be with
Afferent and efferent pathways of the parasympathetic mecha-
you”: ATP in gut mechanosensory transduction. News
nism for enhancing the defecation reflex.
Physiol Sci 18:43, 2003.
De Giorgio R, Guerrini S, Barbara G, et al: Inflammatory
neuropathies of the enteric nervous system. Gastroen-
terology 126:1872, 2004.
Gonella J, Bouvier M, Blanquet F: Extrinsic nervous control
sphincter, thus converting the intrinsic myenteric defe-
of motility of small and large intestines and related sphinc-
cation reflex from a weak effort into a powerful
ters. Physiol Rev 67:902, 1987.
process of defecation that is sometimes effective in
Hall KE: Aging and neural control of the GI tract. II. Neural
emptying the large bowel all the way from the splenic
control of the aging gut: can an old dog learn new tricks?
flexure of the colon to the anus.
Am J Physiol Gastrointest Liver Physiol 283:G827, 2002.
Defecation signals entering the spinal cord initiate
Hansen MB: Neurohumoral control of gastrointestinal
other effects, such as taking a deep breath, closure of
motility. Physiol Res 52:1, 2003.
the glottis, and contraction of the abdominal wall
Hatoum OA, Miura H, Binion DG: The vascular contribu-
tion in the pathogenesis of inflammatory bowel disease.
muscles to force the fecal contents of the colon down-
Am J Physiol Heart Circ Physiol 285:H1791, 2003.
ward and at the same time cause the pelvic floor to
Jensen RT: Involvement of cholecystokinin/gastrin-related
relax downward and pull outward on the anal ring to
peptides and their receptors in clinical gastrointestinal dis-
evaginate the feces.
orders. Pharmacol Toxicol 91:333, 2002.
When it becomes convenient for the person to defe-
Johnson LR: Gastrointestinal Physiology, 6th ed. St. Louis:
cate, the defecation reflexes can purposely be activated
Mosby, 2001.
by taking a deep breath to move the diaphragm down-
Kirkup AJ, Brunsden AM, Grundy D: Receptors and trans-
ward and then contracting the abdominal muscles to
mission in the brain-gut axis: potential for novel therapies.
increase the pressure in the abdomen, thus forcing
I. Receptors on visceral afferents. Am J Physiol Gastroin-
fecal contents into the rectum to cause new reflexes.
test Liver Physiol 280:G787, 2001.
Laroux FS, Pavlick KP, Wolf RE, Grisham MB: Dysregula-
Reflexes initiated in this way are almost never as effec-
tion of intestinal mucosal immunity: implications in
tive as those that arise naturally, for which reason
inflammatory bowel disease. News Physiol Sci 16:272,
people who too often inhibit their natural reflexes are
2001.
likely to become severely constipated.
Orr WC, Chen CL: Aging and neural control of the GI tract:
In newborn babies and in some people with tran-
IV. Clinical and physiological aspects of gastrointestinal
sected spinal cords, the defecation reflexes cause auto-
motility and aging. Am J Physiol Gastrointest Liver
matic emptying of the lower bowel at inconvenient
Physiol 283:G1226, 2002.
times during the day because of lack of conscious
Rao SS: Pathophysiology of adult fecal incontinence. Gas-
control exercised through voluntary contraction or
troenterology 126(1 Suppl 1):S14, 2004.
relaxation of the external anal sphincter.
Sanders KM, Ordog T, Koh SD, Ward SM:A Novel Pace-
maker Mechanism Drives Gastrointestinal Rhythmicity.
News Physiol Sci 15:291, 2000.
Takahashi T: Pathophysiological significance of neuronal
Other Autonomic Reflexes
nitric oxide synthase in the gastrointestinal tract. J Gas-
That Affect Bowel Activity
troenterol 38:421, 2003.
Timmons S, Liston R, Moriarty KJ: Functional dyspepsia:
Aside from the duodenocolic, gastrocolic, gastroileal,
motor abnormalities, sensory dysfunction, and therapeutic
enterogastric, and defecation reflexes that have been
options. Am J Gastroenterol 99:739, 2004.
C H A P T E R
6
4
Secretory Functions of the
Alimentary Tract
Throughout the gastrointestinal tract, secretory
glands subserve two primary functions: First, diges-
tive enzymes are secreted in most areas of the ali-
mentary tract, from the mouth to the distal end of
the ileum. Second, mucous glands, from the mouth
to the anus, provide mucus for lubrication and pro-
tection of all parts of the alimentary tract.
Most digestive secretions are formed only in
response to the presence of food in the alimentary tract, and the quantity
secreted in each segment of the tract is almost exactly the amount needed for
proper digestion. Furthermore, in some portions of the gastrointestinal tract,
even the types of enzymes and other constituents of the secretions are varied in
accordance with the types of food present. The purpose of this chapter, there-
fore, is to describe the different alimentary secretions, their functions, and reg-
ulation of their production.
General Principles of Alimentary
Tract Secretion
Anatomical Types of Glands
Several types of glands provide the different types of alimentary tract secretions.
First, on the surface of the epithelium in most parts of the gastrointestinal tract are
billions of single-cell mucous glands called simply mucous cells or sometimes goblet
cells because they look like goblets. They function mainly in response to local irri-
tation of the epithelium: they extrude mucus directly onto the epithelial surface to
act as a lubricant that also protects the surfaces from excoriation and digestion.
Second, many surface areas of the gastrointestinal tract are lined by pits that rep-
resent invaginations of the epithelium into the submucosa. In the small intestine,
these pits, called crypts of Lieberkühn, are deep and contain specialized secretory
cells. One of these cells is shown in Figure 64-1.
Third, in the stomach and upper duodenum are large numbers of deep tubular
glands. A typical tubular gland can be seen in Figure 64-4, which shows an acid- and
pepsinogen-secreting gland of the stomach (oxyntic gland).
Fourth, also associated with the alimentary tract are several complex glands—the
salivary glands, pancreas, and liver—that provide secretions for digestion or emul-
sification of food. The liver has a highly specialized structure that is discussed in
Chapter 70. The salivary glands and the pancreas are compound acinous glands of
the type shown in Figure 64-2. These glands lie outside the walls of the alimentary
tract and, in this, differ from all other alimentary glands. They contain millions of
acini lined with secreting glandular cells; these acini feed into a system of ducts that
finally empty into the alimentary tract itself.
Basic Mechanisms of Stimulation of the Alimentary
Tract Glands
Effect of Contact of Food with the Epithelium—Function of Enteric Nervous Stimuli. The
mechanical presence of food in a particular segment of the gastrointestinal tract
usually causes the glands of that region and often of adjacent regions to secrete
791
792
Unit XII Gastrointestinal Physiology
Nerve
Endoplasmic Golgi
secretion. This is especially true of the glands in the
Capillary
fiber
reticulum apparatus
upper portion of the tract (innervated by the glos-
sopharyngeal and vagus parasympathetic nerves)
such as the salivary glands, esophageal glands, gastric
Secretion
glands, pancreas, and Brunner’s glands in the duode-
num. It is also true of some glands in the distal portion
of the large intestine, innervated by pelvic parasym-
pathetic nerves. Secretion in the remainder of the
small intestine and in the first two thirds of the large
intestine occurs mainly in response to local neural and
hormonal stimuli in each segment of the gut.
Sympathetic Stimulation. Stimulation of the sympa-
thetic nerves going to the gastrointestinal tract causes
a slight to moderate increase in secretion by some
Basement
Mitochondria Ribosomes
Zymogen
of the local glands. But sympathetic stimulation also
membrane
granules
results in constriction of the blood vessels that supply
Figure 64-1
the glands. Therefore, sympathetic stimulation can
have a dual effect: First, sympathetic stimulation alone
Typical function of a glandular cell for formation and secretion of
usually slightly increases secretion. But, second, if
enzymes and other secretory substances.
parasympathetic or hormonal stimulation is already
causing copious secretion by the glands, superimposed
sympathetic stimulation usually reduces the secretion,
sometimes significantly so, mainly because of vaso-
Primary secretion:
constrictive reduction of the blood supply.
1. Ptyalin
2. Mucus
3. Extracellular fluid
Regulation of Glandular Secretion by Hormones. In the
stomach and intestine, several different gastrointestinal
hormones help regulate the volume and character of
the secretions. These hormones are liberated from the
gastrointestinal mucosa in response to the presence of
Na+ active absorption
food in the lumen of the gut. The hormones then are
Cl- passive absorption
absorbed into the blood and carried to the glands,
K+ active secretion
where they stimulate secretion. This type of stimula-
HCO3- secretion
tion is particularly valuable to increase the output of
gastric juice and pancreatic juice when food enters the
stomach or duodenum.
Chemically, the gastrointestinal hormones are
polypeptides or polypeptide derivatives.
Saliva
Figure 64-2
Basic Mechanism of Secretion
Formation and secretion of saliva by a submandibular salivary
by Glandular Cells
gland.
Secretion of Organic Substances. Although all the
basic mechanisms by which glandular cells function
moderate to large quantities of juices. Part of this local
are not known, experimental evidence points to the
effect, especially the secretion of mucus by mucous
following principles of secretion, as shown in Figure
cells, results from direct contact stimulation of the
64-1.
surface glandular cells by the food.
1. The nutrient material needed for formation of
In addition, local epithelial stimulation also acti-
the secretion must first diffuse or be actively
vates the enteric nervous system of the gut wall. The
transported by the blood in the capillaries into the
types of stimuli that do this are (1) tactile stimulation,
base of the glandular cell.
(2) chemical irritation, and (3) distention of the gut
2. Many mitochondria located inside the glandular
wall. The resulting nervous reflexes stimulate both the
cell near its base use oxidative energy to form
mucous cells on the gut epithelial surface and the deep
adenosine triphosphate (ATP).
glands in the gut wall to increase their secretion.
3. Energy from the ATP, along with appropriate
substrates provided by the nutrients, is then used
Autonomic Stimulation of Secretion
to synthesize the organic secretory substances;
Parasympathetic Stimulation. Stimulation of the
this synthesis occurs almost entirely in the
parasympathetic nerves to the alimentary tract almost
endoplasmic reticulum and Golgi complex of the
invariably increases the rates of alimentary glandular
glandular cell. Ribosomes adherent to the
Chapter 64
Secretory Functions of the Alimentary Tract
793
reticulum are specifically responsible for
through the membrane to the interior of the cell, thus
formation of the proteins that are secreted.
leading to secretion.
4. The secretory materials are transported through
Although this mechanism for secretion is still partly
the tubules of the endoplasmic reticulum, passing
theoretical, it does explain how it would be possible
in about 20 minutes all the way to the vesicles of
for nerve impulses to regulate secretion. Hormones
the Golgi complex.
acting on the cell membrane are believed also to cause
5. In the Golgi complex, the materials are modified,
similar secretory results to those caused by nervous
added to, concentrated, and discharged into the
stimulation.
cytoplasm in the form of secretory vesicles, which
are stored in the apical ends of the secretory cells.
6. These vesicles remain stored until nervous or
Lubricating and Protective
hormonal control signals cause the cells to
Properties of Mucus, and
extrude the vesicular contents through the cells’
Importance of Mucus in the
surface. This probably occurs in the following
Gastrointestinal Tract
way: The control signal first increases the cell
Mucus is a thick secretion composed mainly of water,
membrane permeability to calcium ions, and
electrolytes, and a mixture of several glycoproteins,
calcium enters the cell. The calcium in turn causes
which themselves are composed of large polysaccha-
many of the vesicles to fuse with the apical cell
rides bound with much smaller quantities of protein.
membrane. Then the apical cell membrane breaks
Mucus is slightly different in different parts of the gas-
open, thus emptying the vesicles to the exterior;
trointestinal tract, but everywhere it has several im-
this process is called exocytosis.
portant characteristics that make it both an excellent
lubricant and a protectant for the wall of the gut. First,
Water and Electrolyte Secretion. A second necessity for
mucus has adherent qualities that make it adhere tightly
glandular secretion is secretion of sufficient water and
to the food or other particles and to spread as a thin film
electrolytes to go along with the organic substances.
over the surfaces. Second, it has sufficient body that
it coats the wall of the gut and prevents actual contact
The following is a postulated method by which
of most food particles with the mucosa. Third, mucus
nervous stimulation causes water and salts to pass
has a low resistance for slippage, so that the particles
through the glandular cells in great profusion, washing
can slide along the epithelium with great ease. Fourth,
the organic substances through the secretory border of
mucus causes fecal particles to adhere to one another
the cells at the same time:
to form the feces that are expelled during a bowel move-
1. Nerve stimulation has a specific effect on the
ment. Fifth, mucus is strongly resistant to digestion by
basal portion of the cell membrane to cause active
the gastrointestinal enzymes. And sixth, the glycopro-
transport of chloride ions to the cell interior.
teins of mucus have amphoteric properties, which
2. The resulting increase in electronegativity induced
means that they are capable of buffering small amounts
inside the cell by excess negatively charged
of either acids or alkalies; also, mucus often contains
moderate quantities of bicarbonate ions which specifi-
chloride ions then causes positive ions such as
cally neutralize acids.
sodium ions also to move through the cell
In summary, mucus has the ability to allow easy slip-
membrane to the interior of the cell.
page of food along the gastrointestinal tract and to
3. Now, the new excess of both negative and
prevent excoriative or chemical damage to the epithe-
positive ions inside the cell creates an osmotic
lium. A person becomes acutely aware of the lubricat-
force that causes osmosis of water to the interior,
ing qualities of mucus when the salivary glands fail to
thereby increasing cell volume and hydrostatic
secrete saliva, because then it is difficult to swallow solid
pressure inside the cell, causing the cell itself to
food even when it is eaten along with large amounts of
swell.
water.
4. The pressure in the cell then initiates minute
openings of the secretory border of the cell,
causing flushing of water, electrolytes, and organic
Secretion of Saliva
materials out of the secretory end of the glandular
cell.
Salivary Glands; Characteristics of Saliva. The principal
In support of these secretory processes have been
glands of salivation are the parotid, submandibular,
the following findings: First, the nerve endings on glan-
and sublingual glands; in addition, there are many very
dular cells are principally on the bases of the cells.
small buccal glands. Daily secretion of saliva normally
Second, microelectrode studies show that the normal
ranges between 800 and 1500 milliliters, as shown by
electrical potential across the membrane at the base of
the average value of 1000 milliliters in Table 64-1.
the cell is between 30 and 40 millivolts, with negativ-
Saliva contains two major types of protein secretion:
ity on the interior and positivity on the exterior.
(1) a serous secretion that contains ptyalin
(an
Parasympathetic stimulation increases this polariza-
a-amylase), which is an enzyme for digesting starches,
tion voltage to values some 10 and 20 millivolts more
and (2) mucus secretion that contains mucin for lubri-
negative than normal. This increase in polarization
cating and for surface protective purposes.
voltage lasts for 1 second or longer after the nerve
The parotid glands secrete almost entirely the
signal has arrived, indicating that it is caused by move-
serous type of secretion, while the submandibular and
ment of negative ions
(presumably chloride ions)
sublingual glands secrete both serous secretion and
794
Unit XII Gastrointestinal Physiology
as in plasma; and the concentration of bicarbonate
Table 64-1
ions is 50 to 70 mEq/L, about two to three times that
Daily Secretion of Intestinal Juices
of plasma.
During maximal salivation, the salivary ionic con-
Daily Volume (ml)
pH
centrations change considerably because the rate of
formation of primary secretion by the acini can
Saliva
1000
6.0-7.0
Gastric secretion
1500
1.0-3.5
increase as much as 20-fold. This acinar secretion
Pancreatic secretion
1000
8.0-8.3
then flows through the ducts so rapidly that the ductal
Bile
1000
7.8
reconditioning of the secretion is considerably
Small intestine secretion
1800
7.5-8.0
reduced. Therefore, when copious quantities of saliva
Brunner’s gland secretion
200
8.0-8.9
are being secreted, the sodium chloride concentration
Large intestinal secretion
200
7.5-8.0
rises only to one half or two thirds that of plasma, and
Total
6700
the potassium concentration rises to only four times
that of plasma.
Function of Saliva for Oral Hygiene. Under basal awake con-
ditions, about 0.5 milliliter of saliva, almost entirely of
mucus. The buccal glands secrete only mucus. Saliva
the mucous type, is secreted each minute; but during
has a pH between 6.0 and 7.0, a favorable range for
sleep, secretion becomes very little. This secretion plays
the digestive action of ptyalin.
an exceedingly important role for maintaining healthy
oral tissues. The mouth is loaded with pathogenic bac-
Secretion of Ions in Saliva. Saliva contains especially
teria that can easily destroy tissues and cause dental
large quantities of potassium and bicarbonate ions.
caries. Saliva helps prevent the deteriorative processes
Conversely, the concentrations of both sodium and
in several ways.
chloride ions are several times less in saliva than in
First, the flow of saliva itself helps wash away patho-
plasma. One can understand these special concentra-
genic bacteria as well as food particles that provide their
metabolic support.
tions of ions in the saliva from the following descrip-
Second, saliva contains several factors that destroy
tion of the mechanism for secretion of saliva.
bacteria. One of these is thiocyanate ions and another
Figure 64-2 shows secretion by the submandibular
is several proteolytic enzymes—most important,
gland, a typical compound gland that contains acini
lysozyme—that (a) attack the bacteria, (b) aid the thio-
and salivary ducts. Salivary secretion is a two-stage
cyanate ions in entering the bacteria where these ions
operation: the first stage involves the acini, and the
in turn become bactericidal, and (c) digest food parti-
second, the salivary ducts. The acini secrete a primary
cles, thus helping further to remove the bacterial meta-
secretion that contains ptyalin and/or mucin in a solu-
bolic support.
tion of ions in concentrations not greatly different
Third, saliva often contains significant amounts of
from those of typical extracellular fluid. As the
protein antibodies that can destroy oral bacteria, includ-
ing some that cause dental caries. In the absence of
primary secretion flows through the ducts, two major
salivation, oral tissues often become ulcerated and oth-
active transport processes take place that markedly
erwise infected, and caries of the teeth can become
modify the ionic composition of the fluid in the saliva.
rampant.
First, sodium ions are actively reabsorbed from all
the salivary ducts and potassium ions are actively
secreted in exchange for the sodium. Therefore, the
Nervous Regulation of Salivary
sodium ion concentration of the saliva becomes
greatly reduced, whereas the potassium ion concen-
Secretion
tration becomes increased. However, there is excess
sodium reabsorption over potassium secretion, and
Figure 64-3 shows the parasympathetic nervous path-
this creates electrical negativity of about -70 millivolts
ways for regulating salivation, demonstrating that the
in the salivary ducts; this in turn causes chloride ions
salivary glands are controlled mainly by parasympa-
to be reabsorbed passively. Therefore, the chloride ion
thetic nervous signals all the way from the superior and
concentration in the salivary fluid falls to a very low
inferior salivatory nuclei in the brain stem.
level, matching the ductal decrease in sodium ion
The salivatory nuclei are located approximately at
concentration.
the juncture of the medulla and pons and are excited
Second, bicarbonate ions are secreted by the ductal
by both taste and tactile stimuli from the tongue and
epithelium into the lumen of the duct. This is at least
other areas of the mouth and pharynx. Many taste
partly caused by passive exchange of bicarbonate for
stimuli, especially the sour taste (caused by acids),
chloride ions, but it may also result partly from an
elicit copious secretion of saliva—often 8 to 20 times
active secretory process.
the basal rate of secretion. Also, certain tactile stimuli,
The net result of these transport processes is that
such as the presence of smooth objects in the mouth
under resting conditions, the concentrations of sodium
(e.g., a pebble), cause marked salivation, whereas
and chloride ions in the saliva are only about 15 mEq/
rough objects cause less salivation and occasionally
L each, about one seventh to one tenth their concen-
even inhibit salivation.
trations in plasma. Conversely, the concentration of
Salivation can also be stimulated or inhibited by
potassium ions is about 30 mEq/L, seven times as great
nervous signals arriving in the salivatory nuclei from
Chapter 64
Secretory Functions of the Alimentary Tract
795
Surface
Tractus
Superior and inferior
epithelium
solitarius
salivatory nuclei
Submandibular gland
Mucous neck
Submandibular
cells
ganglion
Oxyntic
Facial
(or parietal)
nerve
cells
Chorda
Peptic
tympani
(or chief)
Sublingual gland
cells
Parotid
gland
Otic ganglion Taste and
tactile stimuli
Figure 64-4
Glossopharyngeal
nerve
Oxyntic gland from the body of the stomach.
Tongue
Figure 64-3
Esophageal Secretion
The esophageal secretions are entirely mucous in char-
Parasympathetic nervous regulation of salivary secretion.
acter and principally provide lubrication for swallowing.
The main body of the esophagus is lined with many
simple mucous glands. At the gastric end and to a lesser
extent in the initial portion of the esophagus, there
higher centers of the central nervous system. For
are also many compound mucous glands. The mucus
instance, when a person smells or eats favorite foods,
secreted by the compound glands in the upper esopha-
salivation is greater than when disliked food is smelled
gus prevents mucosal excoriation by newly entering
or eaten. The appetite area of the brain, which partially
food, whereas the compound glands located near the
esophagogastric junction protect the esophageal wall
regulates these effects, is located in proximity to the
from digestion by acidic gastric juices that often reflux
parasympathetic centers of the anterior hypothalamus,
from the stomach back into the lower esophagus.
and it functions to a great extent in response to signals
Despite this protection, a peptic ulcer at times can still
from the taste and smell areas of the cerebral cortex
occur at the gastric end of the esophagus.
or amygdala.
Salivation also occurs in response to reflexes origi-
nating in the stomach and upper small intestines—par-
Gastric Secretion
ticularly when irritating foods are swallowed or when
a person is nauseated because of some gastrointestinal
Characteristics of the Gastric
abnormality. The saliva, when swallowed, helps to
Secretions
remove the irritating factor in the gastrointestinal tract
by diluting or neutralizing the irritant substances.
In addition to mucus-secreting cells that line the entire
Sympathetic stimulation can also increase salivation
surface of the stomach, the stomach mucosa has two
a slight amount, much less so than does parasympa-
important types of tubular glands: oxyntic glands (also
thetic stimulation. The sympathetic nerves originate
called gastric glands) and pyloric glands. The oxyntic
from the superior cervical ganglia and travel along the
(acid-forming) glands secrete hydrochloric acid,
surfaces of the blood vessel walls to the salivary
pepsinogen, intrinsic factor, and mucus. The pyloric
glands.
glands secrete mainly mucus for protection of the
A secondary factor that also affects salivary secre-
pyloric mucosa from the stomach acid. They also
tion is the blood supply to the glands because secretion
secrete the hormone gastrin.
always requires adequate nutrients from the blood.
The oxyntic glands are located on the inside surfaces
The parasympathetic nerve signals that induce copious
of the body and fundus of the stomach, constituting the
salivation also moderately dilate the blood vessels. In
proximal 80 per cent of the stomach. The pyloric
addition, salivation itself directly dilates the blood
glands are located in the antral portion of the stomach,
vessels, thus providing increased salivatory gland
the distal 20 per cent of the stomach.
nutrition as needed by the secreting cells. Part of this
additional vasodilator effect is caused by kallikrein
Secretions from the Oxyntic (Gastric) Glands
secreted by the activated salivary cells, which in turn
A typical stomach oxyntic gland is shown in Figure
acts as an enzyme to split one of the blood proteins,
64-4. It is composed of three types of cells: (1) mucous
an alpha2-globulin, to form bradykinin, a strong
neck cells, which secrete mainly mucus; (2) peptic
vasodilator.
(or chief) cells, which secrete large quantities of
796
Unit XII Gastrointestinal Physiology
pepsinogen; and (3) parietal (or oxyntic) cells, which
demonstrating that it contains large branching intra-
secrete hydrochloric acid and intrinsic factor. Secretion
cellular canaliculi. The hydrochloric acid is formed at
of hydrochloric acid by the parietal cells involves
the villus-like projections inside these canaliculi and is
special mechanisms, as follows.
then conducted through the canaliculi to the secretory
end of the cell.
Basic Mechanism of Hydrochloric Acid Secretion. When
Different suggestions for the chemical mechanism
stimulated, the parietal cells secrete an acid solution
of hydrochloric acid formation have been offered.
that contains about 160 millimoles of hydrochloric acid
One of these, shown in Figure 64-6, consists of the
per liter, which is almost exactly isotonic with the body
following steps:
fluids. The pH of this acid is about 0.8, demonstrating
1.
Chloride ion is actively transported from the
its extreme acidity. At this pH, the hydrogen ion con-
cytoplasm of the parietal cell into the lumen of
centration is about 3 million times that of the arterial
the canaliculus, and sodium ions are actively
blood. To concentrate the hydrogen ions this tre-
transported out of the canaliculus into the
mendous amount requires more than 1500 calories of
cytoplasm of the parietal cell. These two effects
energy per liter of gastric juice.
together create a negative potential of -40 to -70
Figure
64-5 shows schematically the functional
millivolts in the canaliculus, which in turn causes
structure of a parietal cell (also called oxyntic cell),
diffusion of positively charged potassium ions
and a small number of sodium ions from the
cell cytoplasm into the canaliculus. Thus, in
effect, mainly potassium chloride and much
smaller amounts of sodium chloride enter the
Mucous
canaliculus.
neck cells
2.
Water becomes dissociated into hydrogen ions
and hydroxyl ions in the cell cytoplasm. The
hydrogen ions are then actively secreted into the
canaliculus in exchange for potassium ions: this
active exchange process is catalyzed by H+,K+-
Oxyntic
ATPase. In addition, the sodium ions are actively
(parietal)
reabsorbed by a separate sodium pump. Thus,
Secretion
cell
most of the potassium and sodium ions that had
diffused into the canaliculus are reabsorbed into
Canaliculi
the cell cytoplasm, and hydrogen ions take their
place in the canaliculus, giving a strong solution
of hydrochloric acid in the canaliculus. The
hydrochloric acid is then secreted outward
through the open end of the canaliculus into the
lumen of the gland.
3.
Water passes into the canaliculus by osmosis
because of extra ions secreted into the canaliculus.
Thus, the final secretion from the canaliculus
Figure 64-5
contains water, hydrochloric acid at a
concentration of about 150 to 160 mEq/L,
Schematic anatomy of the canaliculi in a parietal (oxyntic) cell.
Extracellular fluid
Parietal cell
Lumen of canaliculus
CO2
CO2
H2O
H+ (155 mEq/L)
HCO3-
HCO3-
CO2 + OH- + H+
P
K+
K+
K+
K+ (15 mEq/L)
P
Na+
Na+
Na+
Na+
(3 mEq/L)
P
Figure 64-6
Postulated mechanism for secre-
Cl- (173 mEq/L)
Cl-
Cl-
Cl-
tion of hydrochloric acid.
(The
P
points labeled “P” indicate active
(Osmosis)
pumps, and the dashed lines
H2O
H2O
represent free diffusion and
osmosis.)
Chapter 64
Secretory Functions of the Alimentary Tract
797
potassium chloride at a concentration of 15 mEq/
Surface Mucous Cells
L, and a small amount of sodium chloride.
4. Finally, carbon dioxide, either formed during
The entire surface of the stomach mucosa between
metabolism in the cell or entering the cell from
glands has a continuous layer of a special type of
the blood, combines under the influence of
mucous cells called simply “surface mucous cells.”
carbonic anhydrase with the hydroxyl ions (from
They secrete large quantities of a very viscid mucus
step 2) to form bicarbonate ions. These then
that coats the stomach mucosa with a gel layer of
diffuse out of the cell cytoplasm into the
mucus often more than
1 millimeter thick, thus
extracellular fluid in exchange for chloride ions
providing a major shell of protection for the stomach
that enter the cell from the extracellular fluid and
wall as well as contributing to lubrication of food
are later secreted into the canaliculus.
transport.
Another characteristic of this mucus is that it is alka-
line. Therefore, the normal underlying stomach wall is
Secretion and Activation of Pepsinogen. Several slightly
not directly exposed to the highly acidic, proteolytic
different types of pepsinogen are secreted by
stomach secretion. Even the slightest contact with
the peptic and mucous cells of the gastric glands.
food or any irritation of the mucosa directly stimulates
Even so, all the pepsinogens perform the same
the surface mucous cells to secrete additional quanti-
functions.
ties of this thick, alkaline, viscid mucus.
When pepsinogen is first secreted, it has no diges-
tive activity. However, as soon as it comes in contact
with hydrochloric acid, it is activated to form active
Stimulation of Gastric Acid Secretion
pepsin. In this process, the pepsinogen molecule,
having a molecular weight of about 42,500, is split to
Parietal Cells of the Oxyntic Glands Are the Only Cells That
form a pepsin molecule, having a molecular weight of
Secrete Hydrochloric Acid. The parietal cells, located deep
about 35,000.
in the oxyntic glands of the main body of the stomach,
Pepsin functions as an active proteolytic enzyme in
are the only cells that secrete hydrochloric acid. As
a highly acid medium (optimum pH 1.8 to 3.5), but
noted earlier in the chapter, the acidity of the fluid
above a pH of about 5 it has almost no proteolytic
secreted by these cells can be very great, with pH as
activity and becomes completely inactivated in a short
low as 0.8. However, secretion of this acid is under con-
time. Hydrochloric acid is as necessary as pepsin for
tinuous control by both endocrine and nervous signals.
protein digestion in the stomach; this is discussed
Furthermore, the parietal cells operate in close associ-
further in Chapter 65.
ation with another type of cell called enterochromaf-
fin-like cells (ECL cells), the primary function of which
is to secrete histamine.
Secretion of Intrinsic Factor. The substance intrinsic factor,
The ECL cells lie in the deep recesses of the oxyntic
essential for absorption of vitamin B12 in the ileum, is
glands and therefore release histamine in direct
secreted by the parietal cells along with the secretion of
contact with the parietal cells of the glands. The rate
hydrochloric acid. When the acid-producing parietal
of formation and secretion of hydrochloric acid by the
cells of the stomach are destroyed, which frequently
parietal cells is directly related to the amount of his-
occurs in chronic gastritis, the person develops not only
tamine secreted by the ECL cells. In turn, the ECL
achlorhydria (lack of stomach acid secretion) but often
also pernicious anemia because of failure of maturation
cells can be stimulated to secrete histamine in several
of the red blood cells in the absence of vitamin B12 stim-
different ways: (1) Probably the most potent mecha-
ulation of the bone marrow. This is discussed in detail
nism for stimulating histamine secretion is by the hor-
in Chapter 32.
monal substance gastrin, which is formed almost
entirely in the antral portion of the stomach mucosa
in response to proteins in the foods being digested.
(2) In addition, the ECL cells can be stimulated by
Pyloric Glands—Secretion of Mucus
(a) acetylcholine released from stomach vagal nerve
and Gastrin
endings and (b) probably also by hormonal substances
secreted by the enteric nervous system of the stomach
The pyloric glands are structurally similar to the
wall. Let us discuss first the gastrin mechanism for
oxyntic glands but contain few peptic cells and almost
control of the ECL cells and their subsequent control
no parietal cells. Instead, they contain mostly mucous
of parietal cell secretion of hydrochloric acid.
cells that are identical with the mucous neck cells of
the oxyntic glands. These cells secrete a small amount
Stimulation of Acid Secretion by Gastrin. Gastrin is itself a
of pepsinogen, as discussed earlier, and an especially
hormone secreted by gastrin cells, also called G cells.
large amount of thin mucus that helps to lubricate
These cells are located in the pyloric glands in the
food movement, as well as to protect the stomach wall
distal end of the stomach. Gastrin is a large polypep-
from digestion by the gastric enzymes. The pyloric
tide secreted in two forms: a large form called G-34,
glands also secrete the hormone gastrin, which plays a
which contains 34 amino acids, and a smaller form,
key role in controlling gastric secretion, as we discuss
G-17, which contains 17 amino acids. Although both of
shortly.
these are important, the smaller is more abundant.
798
Unit XII Gastrointestinal Physiology
When meats or other protein-containing foods
Cephalic Phase. The cephalic phase of gastric secretion
reach the antral end of the stomach, some of the pro-
occurs even before food enters the stomach, especially
teins from these foods have a special stimulatory effect
while it is being eaten. It results from the sight, smell,
thought, or taste of food, and the greater the appetite,
on the gastrin cells in the pyloric glands to cause
the more intense is the stimulation. Neurogenic signals
release of gastrin into the digestive juices of the
that cause the cephalic phase of gastric secretion origi-
stomach. The vigorous mixing of the gastric juices
nate in the cerebral cortex and in the appetite centers
transports the gastrin rapidly to the ECL cells in the
of the amygdala and hypothalamus. They are trans-
body of the stomach, causing release of histamine
mitted through the dorsal motor nuclei of the vagi and
directly into the deep oxyntic glands. The histamine
thence through the vagus nerves to the stomach. This
then acts quickly to stimulate gastric hydrochloric acid
phase of secretion normally accounts for about 20 per
secretion.
cent of the gastric secretion associated with eating a
meal.
Regulation of Pepsinogen Secretion
Gastric Phase. Once food enters the stomach, it excites
(1) long vagovagal reflexes from the stomach to the
brain and back to the stomach, (2) local enteric reflexes,
Regulation of pepsinogen secretion by the peptic cells
and (3) the gastrin mechanism, all of which in turn cause
in the oxyntic glands is much less complex than regu-
secretion of gastric juice during several hours while food
lation of acid secretion; it occurs in response to two
remains in the stomach. The gastric phase of secretion
types of signals: (1) stimulation of the peptic cells by
accounts for about 70 per cent of the total gastric secre-
acetylcholine released from the vagus nerves or from
tion associated with eating a meal and therefore
the gastric enteric nervous plexus, and (2) stimulation
accounts for most of the total daily gastric secretion of
of peptic cell secretion in response to acid in the
about 1500 milliliters.
stomach. The acid probably does not stimulate the
peptic cells directly but instead elicits additional
Intestinal Phase. The presence of food in the upper
portion of the small intestine, particularly in the duo-
enteric nervous reflexes that support the original
denum, will continue to cause stomach secretion of
nervous signals to the peptic cells. Therefore, the rate
small amounts of gastric juice, probably partly because
of secretion of pepsinogen, the precursor of the
of small amounts of gastrin released by the duodenal
enzyme pepsin that causes protein digestion, is
mucosa.
strongly influenced by the amount of acid in the
stomach. In people who have lost the ability to secrete
normal amounts of acid, secretion of pepsinogen is
Inhibition of Gastric Secretion
also decreased, even though the peptic cells may
by Other Post-Stomach
otherwise appear to be normal.
Intestinal Factors
Phases of Gastric Secretion
Although intestinal chyme slightly stimulates gastric
secretion during the early intestinal phase of stomach
Gastric secretion is said to occur in three “phases” (as
secretion, it paradoxically inhibits gastric secretion at
shown in Figure 64-7): a cephalic phase, a gastric phase,
other times. This inhibition results from at least two
and an intestinal phase.
influences.
Vagal center
of medulla
Cephalic phase via vagus
Parasympathetics excite
pepsin and acid production
Secretory
Food
fiber
Gastric phase:
1. Local nervous
Afferent
Vagus
Local nerve
secretory reflexes
fibers
trunk
plexus
2. Vagal reflexes
3. Gastrin-histamine
stimulation
Circulatory system
Gastrin
Intestinal phase:
1. Nervous mechanisms
Figure 64-7
2. Hormonal mechanisms
Small bowel
Phases of gastric secretion and their regulation.
Chapter 64
Secretory Functions of the Alimentary Tract
799
1. The presence of food in the small intestine initiates
Pancreatic Secretion
a reverse enterogastric reflex, transmitted through
the myenteric nervous system as well as through
The pancreas, which lies parallel to and beneath the
extrinsic sympathetic and vagus nerves, that inhibits
stomach (illustrated in Figure 64-10), is a large com-
stomach secretion. This reflex can be initiated by
pound gland with most of its internal structure similar
distending the small bowel, by the presence of acid
to that of the salivary glands shown in Figure 64-2. The
in the upper intestine, by the presence of protein
breakdown products, or by irritation of the mucosa.
pancreatic digestive enzymes are secreted by pancre-
This is part of the complex mechanism discussed in
atic acini, and large volumes of sodium bicarbonate
Chapter 63 for slowing stomach emptying when the
solution are secreted by the small ductules and larger
intestines are already filled.
ducts leading from the acini. The combined product of
2. The presence of acid, fat, protein breakdown
enzymes and sodium bicarbonate then flows through
products, hyperosmotic or hypo-osmotic fluids, or
a long pancreatic duct that normally joins the hepatic
any irritating factor in the upper small intestine
duct immediately before it empties into the duodenum
causes release of several intestinal hormones. One
through the papilla of Vater, surrounded by the sphinc-
of these is secretin, which is especially important
ter of Oddi.
for control of pancreatic secretion. However,
Pancreatic juice is secreted most abundantly in
secretin opposes stomach secretion. Three other
hormones—gastric inhibitory peptide, vasoactive
response to the presence of chyme in the upper por-
intestinal polypeptide, and somatostatin—also have
tions of the small intestine, and the characteristics of
slight to moderate effects in inhibiting gastric
the pancreatic juice are determined to some extent by
secretion.
the types of food in the chyme. (The pancreas also
The functional purpose of inhibitory gastric secretion
secretes insulin, but this is not secreted by the same
by intestinal factors is presumably to slow passage of
pancreatic tissue that secretes intestinal pancreatic
chyme from the stomach when the small intestine is
juice. Instead, insulin is secreted directly into the
already filled or already overactive. In fact, the entero-
blood—not into the intestine—by the islets of Langer-
gastric inhibitory reflexes plus inhibitory hormones
hans that occur in islet patches throughout the pan-
usually also reduce stomach motility at the same time
creas. These are discussed in detail in Chapter 78.)
that they reduce gastric secretion, as was discussed in
Chapter 63.
Pancreatic Digestive Enzymes
Gastric Secretion During the Interdigestive Period. The
stomach secretes a few milliliters of gastric juice each
Pancreatic secretion contains multiple enzymes for
hour during the “interdigestive period,” when little or
digesting all of the three major types of food: proteins,
no digestion is occurring anywhere in the gut. The secre-
carbohydrates, and fats. It also contains large quanti-
tion that does occur usually is almost entirely of the
ties of bicarbonate ions, which play an important role
nonoxyntic type, composed mainly of mucus but little
pepsin and almost no acid.
in neutralizing the acidity of the chyme emptied from
Unfortunately, emotional stimuli frequently increase
the stomach into the duodenum.
interdigestive gastric secretion
(highly peptic and
The most important of the pancreatic enzymes for
acidic) to 50 milliliters or more per hour, in very much
digesting proteins are trypsin, chymotrypsin, and car-
the same way that the cephalic phase of gastric secre-
boxypolypeptidase. By far the most abundant of these
tion excites secretion at the onset of a meal. This
is trypsin.
increase of secretion in response to emotional stimuli is
Trypsin and chymotrypsin split whole and partially
believed to be one of the causative factors in develop-
digested proteins into peptides of various sizes but do
ment of peptic ulcers, as discussed in Chapter 66.
not cause release of individual amino acids. However,
carboxypolypeptidase does split some peptides into
individual amino acids, thus completing digestion of
Chemical Composition of Gastrin and
some proteins all the way to the amino acid state.
Other Gastrointestinal Hormones
The pancreatic enzyme for digesting carbohydrates
is pancreatic amylase, which hydrolyzes starches, glyco-
Gastrin, cholecystokinin, and secretin are all large
gen, and most other carbohydrates (except cellulose)
polypeptides with approximate molecular weights,
to form mostly disaccharides and a few trisaccharides.
respectively, of 2000, 4200, and 3400. The terminal five
The main enzymes for fat digestion are (1) pancre-
amino acids in the gastrin and cholecystokinin molec-
atic lipase, which is capable of hydrolyzing neutral fat
ular chains are the same. The functional activity of
into fatty acids and monoglycerides; (2) cholesterol
gastrin resides in the terminal four amino acids, and
esterase, which causes hydrolysis of cholesterol esters;
the activity for cholecystokinin resides in the terminal
and (3) phospholipase, which splits fatty acids from
eight amino acids. All the amino acids in the secretin
phospholipids.
molecule are essential.
When first synthesized in the pancreatic cells, the
A synthetic gastrin, composed of the terminal four
proteolytic digestive enzymes are in the inactive
amino acids of natural gastrin plus the amino acid
forms trypsinogen, chymotrypsinogen, and procar-
alanine, has all the same physiologic properties as
boxypolypeptidase, which are all inactive enzymati-
the natural gastrin. This synthetic product is called
cally. They become activated only after they are
pentagastrin.
secreted into the intestinal tract. Trypsinogen is
800
Unit XII Gastrointestinal Physiology
activated by an enzyme called enterokinase, which is
secreted by the intestinal mucosa when chyme comes
Blood
Lumen
in contact with the mucosa. Also, trypsinogen can be
Ductule cells
autocatalytically activated by trypsin that has already
been formed from previously secreted trypsinogen.
Chymotrypsinogen is activated by trypsin to form chy-
motrypsin, and procarboxypolypeptidase is activated
Na+
Na+
Na+
in a similar manner.
H+
H+
HCO3
HCO3-
(Active
transport)
(Active
Secretion of Trypsin Inhibitor Prevents Digestion of the
H2CO3
transport)
Pancreas Itself. It is important that the proteolytic
(Carbonic
anhydrase)
enzymes of the pancreatic juice not become activated
H2O
until after they have been secreted into the intestine
+
CO2
CO2
because the trypsin and the other enzymes would
digest the pancreas itself. Fortunately, the same cells
H2O
H2O
that secrete proteolytic enzymes into the acini of the
pancreas secrete simultaneously another substance
called trypsin inhibitor. This substance is formed in the
cytoplasm of the glandular cells, and it prevents acti-
Figure 64-8
vation of trypsin both inside the secretory cells and in
the acini and ducts of the pancreas. And, because it is
Secretion of isosmotic sodium bicarbonate solution by the pan-
trypsin that activates the other pancreatic proteolytic
creatic ductules and ducts.
enzymes, trypsin inhibitor prevents activation of the
others as well.
When the pancreas becomes severely damaged or
when a duct becomes blocked, large quantities of pan-
ions (HCO3_ and H+). Then the bicarbonate ions
creatic secretion sometimes become pooled in the
are actively transported in association with
damaged areas of the pancreas. Under these condi-
sodium ions (Na+) through the luminal border of
tions, the effect of trypsin inhibitor is often over-
the cell into the lumen of the duct.
whelmed, in which case the pancreatic secretions
2. The hydrogen ions formed by dissociation of
rapidly become activated and can literally digest the
carbonic acid inside the cell are exchanged for
entire pancreas within a few hours, giving rise to the
sodium ions through the blood border of the
condition called acute pancreatitis. This sometimes is
cell by a secondary active transport process.
lethal because of accompanying circulatory shock;
This supplies the sodium ions (Na+) that are
even if not lethal, it usually leads to a subsequent life-
transported through the luminal border into the
time of pancreatic insufficiency.
pancreatic duct lumen to provide electrical
neutrality for the secreted bicarbonate ions.
3. The overall movement of sodium and bicarbonate
Secretion of Bicarbonate Ions
ions from the blood into the duct lumen creates
an osmotic pressure gradient that causes osmosis
Although the enzymes of the pancreatic juice are
of water also into the pancreatic duct, thus
secreted entirely by the acini of the pancreatic glands,
forming an almost completely isosmotic
the other two important components of pancreatic
bicarbonate solution.
juice, bicarbonate ions and water, are secreted mainly
by the epithelial cells of the ductules and ducts that
lead from the acini. When the pancreas is stimulated
Regulation of Pancreatic Secretion
to secrete copious quantities of pancreatic juice, the
bicarbonate ion concentration can rise to as high as
Basic Stimuli That Cause Pancreatic Secretion
145 mEq/L, a value about five times that of bicarbon-
Three basic stimuli are important in causing pancre-
ate ions in the plasma. This provides a large quantity
atic secretion:
of alkali in the pancreatic juice that serves to neutral-
1. Acetylcholine, which is released from the
ize the hydrochloric acid emptied into the duodenum
parasympathetic vagus nerve endings and from
from the stomach.
other cholinergic nerves in the enteric nervous
The basic steps in the cellular mechanism for secret-
system
ing sodium bicarbonate solution into the pancreatic
2. Cholecystokinin, which is secreted by the
ductules and ducts are shown in Figure 64-8. They are
duodenal and upper jejunal mucosa when food
the following:
enters the small intestine
1. Carbon dioxide diffuses to the interior of the
3. Secretin, which is also secreted by the duodenal
cell from the blood and, under the influence of
and jejunal mucosa when highly acid food enters
carbonic anhydrase, combines with water to form
the small intestine
carbonic acid (H2CO3). The carbonic acid in turn
The first two of these stimuli, acetylcholine and
dissociates into bicarbonate ions and hydrogen
cholecystokinin, stimulate the acinar cells of the
Chapter 64
Secretory Functions of the Alimentary Tract
801
pancreas, causing production of large quantities of
Secretin in turn causes the pancreas to secrete large
pancreatic digestive enzymes but relatively small
quantities of fluid containing a high concentration of
quantities of water and electrolytes to go with the
bicarbonate ion (up to 145 mEq/L) but a low concen-
enzymes. Without the water, most of the enzymes
tration of chloride ion. The secretin mechanism is
remain temporarily stored in the acini and ducts until
especially important for two reasons: First, secretin
more fluid secretion comes along to wash them into
begins to be released from the mucosa of the small
the duodenum. Secretin, in contrast to the first two
intestine when the pH of the duodenal contents falls
basic stimuli, stimulates secretion of large quantities of
below 4.5 to 5.0, and its release increases greatly as the
water solution of sodium bicarbonate by the pancre-
pH falls to 3.0. This immediately causes copious secre-
atic ductal epithelium.
tion of pancreatic juice containing abundant amounts
of sodium bicarbonate. The net result is then the fol-
Multiplicative Effects of Different Stimuli. When all the dif-
lowing reaction in the duodenum:
ferent stimuli of pancreatic secretion occur at once, the
total secretion is far greater than the sum of the secre-
3
HCl+NaHCO ÆNaCl+H CO
2
3
tions caused by each one separately. Therefore, the
Then the carbonic acid immediately dissociates
various stimuli are said to “multiply,” or “potentiate,”
into carbon dioxide and water. The carbon dioxide
one another. Thus, pancreatic secretion normally
is absorbed into the blood and expired through the
results from the combined effects of the multiple basic
lungs, thus leaving a neutral solution of sodium chlo-
stimuli, not from one alone.
ride in the duodenum. In this way, the acid contents
emptied into the duodenum from the stomach become
Phases of Pancreatic Secretion
neutralized, so that further peptic digestive activity by
Pancreatic secretion occurs in three phases, the same
the gastric juices in the duodenum is immediately
as for gastric secretion: the cephalic phase, the gastric
blocked. Because the mucosa of the small intestine
phase, and the intestinal phase. Their characteristics are
cannot withstand the digestive action of acid gastric
as follows.
juice, this is an essential protective mechanism to
prevent development of duodenal ulcers, as is dis-
Cephalic and Gastric Phases. During the cephalic phase
cussed in further detail in Chapter 66.
of pancreatic secretion, the same nervous signals from
Bicarbonate ion secretion by the pancreas provides
the brain that cause secretion in the stomach also
an appropriate pH for action of the pancreatic diges-
cause acetylcholine release by the vagal nerve endings
tive enzymes, which function optimally in a slightly
in the pancreas. This causes moderate amounts of
alkaline or neutral medium, at a pH of 7.0 to 8.0.
enzymes to be secreted into the pancreatic acini,
Fortunately, the pH of the sodium bicarbonate secre-
accounting for about 20 per cent of the total secretion
tion averages 8.0.
of pancreatic enzymes after a meal. But little of the
secretion flows immediately through the pancreatic
ducts into the intestine because only small amounts of
Cholecystokinin—Its Contribution to Control of Diges-
water and electrolytes are secreted along with the
tive Enzyme Secretion by the Pancreas. The presence
enzymes.
of food in the upper small intestine also causes a
During the gastric phase, the nervous stimulation of
second hormone, cholecystokinin, a polypeptide con-
enzyme secretion continues, accounting for another 5
taining 33 amino acids, to be released from yet another
to 10 per cent of pancreatic enzymes secreted after a
group of cells, the I cells, in the mucosa of the duode-
meal. But, again, only small amounts reach the duo-
num and upper jejunum. This release of cholecys-
denum because of continued lack of significant fluid
tokinin results especially from the presence of
secretion.
proteoses and peptones (products of partial protein
digestion) and long-chain fatty acids in the chyme
Intestinal Phase. After chyme leaves the stomach and
coming from the stomach.
enters the small intestine, pancreatic secretion
Cholecystokinin, like secretin, passes by way of the
becomes copious, mainly in response to the hormone
blood to the pancreas but instead of causing sodium
secretin.
bicarbonate secretion causes mainly secretion of still
much more pancreatic digestive enzymes by the acinar
Secretin Stimulates Secretion of Copious Quantities of
cells. This effect is similar to that caused by vagal stim-
Bicarbonate Ions—Neutralization of Acidic Stomach
ulation but even more pronounced, accounting for 70
Chyme. Secretin is a polypeptide, containing 27 amino
to 80 per cent of the total secretion of the pancreatic
acids (molecular weight about 3400), present in an
digestive enzymes after a meal.
inactive form, prosecretin, in so-called S cells in the
The differences between the pancreatic stimulatory
mucosa of the duodenum and jejunum. When acid
effects of secretin and cholecystokinin are shown in
chyme with pH less than 4.5 to 5.0 enters the duode-
Figure 64-9, which demonstrates (1) intense sodium
num from the stomach, it causes duodenal mucosal
bicarbonate secretion in response to acid in the duo-
release and activation of secretin, which is then
denum, stimulated by secretin, (2) a dual effect in
absorbed into the blood. The one truly potent con-
response to soap (a fat), and (3) intense digestive
stituent of chyme that causes this secretin release is the
enzyme secretion (when peptones enter the duode-
hydrochloric acid from the stomach.
num) stimulated by cholecystokinin.
802
Unit XII Gastrointestinal Physiology
Acid from stomach
releases secretin from
wall of duodenum;
Water and
fats and amino acids
NaHCO3
cause release of
Enzymes
cholecystokinin
Common
bile duct
Vagal
stimulation
releases
enzymes
into acini
Secretin causes
copious secretion
Secretin and
of pancreatic fluid
cholecystokinin
and bicarbonate;
absorbed into
cholecystokinin
blood stream
causes secretion
HCI
Soap
Peptone
of enzymes
Figure 64-10
Figure 64-9
Regulation of pancreatic secretion.
Sodium bicarbonate (NaHCO3), water, and enzyme secretion by
the pancreas, caused by the presence of acid (HCl), fat (soap),
or peptone solutions in the duodenum.
(2) Next, the bile flows in the canaliculi toward the
interlobular septa, where the canaliculi empty into ter-
Figure
64-10 summarizes the more important
minal bile ducts and then into progressively larger
factors in the regulation of pancreatic secretion. The
ducts, finally reaching the hepatic duct and common
total amount secreted each day is about 1 liter.
bile duct. From these the bile either empties directly
into the duodenum or is diverted for minutes up to
several hours through the cystic duct into the gall-
Secretion of Bile by the Liver;
bladder, shown in Figure 64-11.
Functions of the Biliary Tree
In its course through the bile ducts, a second portion
of liver secretion is added to the initial bile. This addi-
One of the many functions of the liver is to secrete bile,
tional secretion is a watery solution of sodium and
normally between 600 and 1000 ml/day. Bile serves two
bicarbonate ions secreted by secretory epithelial cells
important functions:
that line the ductules and ducts. This second secretion
First, bile plays an important role in fat digestion
sometimes increases the total quantity of bile by as
and absorption, not because of any enzymes in the bile
much as an additional 100 per cent. The second secre-
that cause fat digestion, but because bile acids in the
tion is stimulated especially by secretin, which causes
bile do two things: (1) they help to emulsify the large
release of additional quantities of bicarbonate ions to
fat particles of the food into many minute particles,
supplement the bicarbonate ions in pancreatic secre-
the surface of which can then be attacked by lipase
tion (for neutralizing acid that empties into the duo-
enzymes secreted in pancreatic juice, and (2) they aid
denum from the stomach).
in absorption of the digested fat end products through
the intestinal mucosal membrane.
Storing and Concentrating Bile in the Gallbladder. Bile is
Second, bile serves as a means for excretion of
secreted continually by the liver cells, but most of it is
several important waste products from the blood.
normally stored in the gallbladder until needed in the
These include especially bilirubin, an end product of
duodenum. The maximum volume that the gallbladder
hemoglobin destruction, and excesses of cholesterol.
can hold is only 30 to 60 milliliters. Nevertheless, as
much as 12 hours of bile secretion (usually about 450
milliliters) can be stored in the gallbladder because
Physiologic Anatomy of
water, sodium, chloride, and most other small elec-
Biliary Secretion
trolytes are continually absorbed through the gall-
bladder mucosa, concentrating the remaining bile
Bile is secreted in two stages by the liver: (1) The initial
constituents that contain the bile salts, cholesterol,
portion is secreted by the principal functional cells of
lecithin, and bilirubin.
the liver, the hepatocytes; this initial secretion contains
Most of this gallbladder absorption is caused by
large amounts of bile acids, cholesterol, and other
active transport of sodium through the gallbladder
organic constituents. It is secreted into minute bile
epithelium, and this is followed by secondary absorp-
canaliculi that originate between the hepatic cells.
tion of chloride ions, water, and most other diffusible
Chapter 64
Secretory Functions of the Alimentary Tract
803
Vagal stimulation
Secretin via
Bile acids via blood
causes weak
blood stream
stimulate parenchymal
contraction of
stimulates
secretion
gallbladder
liver ductal
Stomach
secretion
Liver
Acid
Bile stored and
concentrated up
to 15 times in
Pancreas
gallbladder
Sphincter of
Duodenum
Oddi
Cholecystokinin via blood stream causes:
Figure 64-11
1. Gallbladder contraction
2. Relaxation of sphincter of Oddi
Liver secretion and gallbladder emptying.
constituents. Bile is normally concentrated in this way
Table 64-2
about 5-fold, but it can be concentrated up to a
Composition of Bile
maximum of 20-fold.
Composition of Bile. Table 64-2 gives the composition of
Liver Bile
Gallbladder Bile
bile when it is first secreted by the liver and then after
Water
97.5 g/dl
92 g/dl
it has been concentrated in the gallbladder. This table
Bile salts
1.1 g/dl
6 g/dl
shows that by far the most abundant substances
Bilirubin
0.04 g/dl
0.3 g/dl
secreted in the bile are bile salts, which account for
Cholesterol
0.1 g/dl
0.3 to 0.9 g/dl
Fatty acids
0.12 g/dl
0.3 to 1.2 g/dl
about one half of the total solutes also in the bile. Also
Lecithin
0.04 g/dl
0.3 g/dl
secreted or excreted in large concentrations are biliru-
Na+
145.04 mEq/L
130 mEq/L
bin, cholesterol, lecithin, and the usual electrolytes of
K+
5 mEq/L
12 mEq/L
plasma.
Ca++
5 mEq/L
23 mEq/L
Cl-
100 mEq/L
25 mEq/L
In the concentrating process in the gallbladder,
HCO3-
28 mEq/L
10 mEq/L
water and large portions of the electrolytes (except
calcium ions) are reabsorbed by the gallbladder
mucosa; essentially all other constituents, especially
the bile salts and the lipid substances cholesterol and
lecithin, are not reabsorbed and, therefore, become
This is the same cholecystokinin discussed earlier that
highly concentrated in the gallbladder bile.
causes increased secretion of digestive enzymes by the
acinar cells of the pancreas. The stimulus for cholecys-
Emptying of the Gallbladder—Stimulatory Role of Cholecys-
tokinin entry into the blood from the duodenal
tokinin. When food begins to be digested in the upper
mucosa is mainly the presence of fatty foods in the
gastrointestinal tract, the gallbladder begins to empty,
duodenum.
especially when fatty foods reach the duodenum about
In addition to cholecystokinin, the gallbladder is
30 minutes after a meal. The mechanism of gallblad-
stimulated less strongly by acetylcholine-secreting
der emptying is rhythmical contractions of the wall of
nerve fibers from both the vagi and the intestinal
the gallbladder, but effective emptying also requires
enteric nervous system. They are the same nerves that
simultaneous relaxation of the sphincter of Oddi,
promote motility and secretion in other parts of the
which guards the exit of the common bile duct into the
upper gastrointestinal tract.
duodenum.
In summary, the gallbladder empties its store of con-
By far the most potent stimulus for causing the gall-
centrated bile into the duodenum mainly in response
bladder contractions is the hormone cholecystokinin.
to the cholecystokinin stimulus that itself is initiated
804
Unit XII Gastrointestinal Physiology
mainly by fatty foods. When fat is not in the food,
bile salts is called the enterohepatic circulation of bile
the gallbladder empties poorly, but when significant
salts.
quantities of fat are present, the gallbladder normally
The quantity of bile secreted by the liver each day is
highly dependent on the availability of bile salts—the
empties completely in about 1 hour. Figure 64-11 sum-
greater the quantity of bile salts in the enterohepatic cir-
marizes the secretion of bile, its storage in the gall-
culation (usually a total of only about 2.5 grams), the
bladder, and its ultimate release from the bladder to
greater the rate of bile secretion. Indeed, ingestion of
the duodenum.
supplemental bile salts can increase bile secretion by
several hundred milliliters per day.
If a bile fistula empties the bile salts to the exterior
Function of Bile Salts in Fat Digestion
for several days to several weeks so that they cannot be
reabsorbed from the ileum, the liver increases its pro-
and Absorption
duction of bile salts 6- to 10-fold, which increases the
rate of bile secretion most of the way back to normal.
The liver cells synthesize about 6 grams of bile salts
This demonstrates that the daily rate of liver bile salt
daily. The precursor of the bile salts is cholesterol,
secretion is actively controlled by the availability (or
which is either present in the diet or synthesized in the
lack of availability) of bile salts in the enterohepatic
liver cells during the course of fat metabolism. The
circulation.
cholesterol is first converted to cholic acid or che-
nodeoxycholic acid in about equal quantities. These
Role of Secretin in Helping to Control Bile Secretion. In addi-
acids in turn combine principally with glycine and to a
tion to the strong stimulating effect of bile acids to cause
bile secretion, the hormone secretin that also stimulates
lesser extent with taurine to form glyco- and tauro-
pancreatic secretion increases bile secretion, sometimes
conjugated bile acids. The salts of these acids, mainly
more than doubling its secretion for several hours after
sodium salts, are then secreted in the bile.
a meal. This increase in secretion is almost entirely
The bile salts have two important actions in the
secretion of a sodium bicarbonate-rich watery solution
intestinal tract:
by the epithelial cells of the bile ductules and ducts, and
First, they have a detergent action on the fat parti-
not increased secretion by the liver parenchymal cells
cles in the food. This decreases the surface tension of
themselves. The bicarbonate in turn passes into the
the particles and allows agitation in the intestinal tract
small intestine and joins the bicarbonate from the pan-
to break the fat globules into minute sizes. This is
creas in neutralizing the hydrochloric acid from the
called the emulsifying or detergent function of bile
stomach. Thus, the secretin feedback mechanism for
neutralizing duodenal acid operates not only through its
salts.
effects on pancreatic secretion but also to a lesser extent
Second, and even more important than the emulsi-
through its effect on secretion by the liver ductules and
fying function, bile salts help in the absorption of
ducts.
(1) fatty acids, (2) monoglycerides, (3) cholesterol, and
(4) other lipids from the intestinal tract. They do this
by forming very small physical complexes with these
Liver Secretion of Cholesterol and
lipids; the complexes are called micelles, and they are
Gallstone Formation
semi-soluble in the chyme because of the electrical
charges of the bile salts. The intestinal lipids are
Bile salts are formed in the hepatic cells from choles-
“ferried” in this form to the intestinal mucosa, where
terol in the blood plasma. In the process of secreting the
they are then absorbed into the blood, as will be
bile salts, about 1 to 2 grams of cholesterol are removed
from the blood plasma and secreted into the bile each
described in detail in Chapter 65. Without the presence
day.
of bile salts in the intestinal tract, up to 40 per cent of
Cholesterol is almost completely insoluble in pure
the ingested fats are lost into the feces, and the person
water, but the bile salts and lecithin in bile combine
often develops a metabolic deficit because of this
physically with the cholesterol to form ultramicroscopic
nutrient loss.
micelles in the form of a colloidal solution, as explained
in more detail in Chapter 65. When the bile becomes
Enterohepatic Circulation of Bile Salts. About 94 per cent of
concentrated in the gallbladder, the bile salts and
the bile salts are reabsorbed into the blood from the
lecithin become concentrated along with the choles-
small intestine, about one half of this by diffusion
terol, which keeps the cholesterol in solution.
through the mucosa in the early portions of the small
Under abnormal conditions, the cholesterol may pre-
intestine and the remainder by an active transport
cipitate in the gallbladder, resulting in the formation of
process through the intestinal mucosa in the distal
cholesterol gallstones, as shown in Figure 64-12. The
ileum. They then enter the portal blood and pass back
amount of cholesterol in the bile is determined partly
to the liver. On reaching the liver, on first passage
by the quantity of fat that the person eats, because liver
through the venous sinusoids these salts are absorbed
cells synthesize cholesterol as one of the products of fat
almost entirely back into the hepatic cells and then are
metabolism in the body. For this reason, people on a
resecreted into the bile.
high-fat diet over a period of years are prone to the
In this way, about 94 per cent of all the bile salts are
development of gallstones.
recirculated into the bile, so that on the average these
Inflammation of the gallbladder epithelium, often
salts make the entire circuit some 17 times before being
resulting from low-grade chronic infection, may also
carried out in the feces. The small quantities of bile salts
change the absorptive characteristics of the gallbladder
lost into the feces are replaced by new amounts formed
mucosa, sometimes allowing excessive absorption of
continually by the liver cells. This recirculation of the
water and bile salts but leaving behind the cholesterol
Chapter 64
Secretory Functions of the Alimentary Tract
805
Causes of gallstones:
1. Too much absorption of water
from bile
2. Too much absorption of bile
acids from bile
Mucous goblet
3. Too much cholesterol in bile
Stones
cell
4. Inflammation of epithelium
Liver
Epithelial cell
Hepatic duct
Gallbladder
Stones
Course followed by bile:
Cystic duct
1. During rest
2. During digestion
Paneth cell
Common bile duct
Sphincter of Oddi
Figure 64-13
Papilla of Vater
Pancreatic duct
A crypt of Lieberkühn, found in all parts of the small intestine
between the villi, which secretes almost pure extracellular fluid.
Duodenum
Figure 64-12
area of the gastrointestinal tract to be the site of peptic
Formation of gallstones.
ulcers in about 50 per cent of ulcer patients.
in the bladder in progressively greater concentrations.
Secretion of Intestinal Digestive
Then the cholesterol begins to precipitate, first forming
Juices by the Crypts of Lieberkühn
many small crystals of cholesterol on the surface of
the inflamed mucosa, but then progressing to large
Located over the entire surface of the small intestine
gallstones.
are small pits called crypts of Lieberkühn, one of which
is illustrated in Figure 64-13. These crypts lie between
the intestinal villi. The surfaces of both the crypts and
Secretions of the
the villi are covered by an epithelium composed of two
Small Intestine
types of cells: (1) a moderate number of goblet cells,
which secrete mucus that lubricates and protects the
Secretion of Mucus by Brunner’s
intestinal surfaces, and (2) a large number of entero-
Glands in the Duodenum
cytes, which, in the crypts, secrete large quantities of
water and electrolytes and, over the surfaces of adja-
An extensive array of compound mucous glands,
cent villi, reabsorb the water and electrolytes along
called Brunner’s glands, is located in the wall of the
with end products of digestion.
first few centimeters of the duodenum, mainly
The intestinal secretions are formed by the entero-
between the pylorus of the stomach and the papilla of
cytes of the crypts at a rate of about 1800 ml/day. These
Vater where pancreatic secretion and bile empty into
secretions are almost pure extracellular fluid and have
the duodenum. These glands secrete large amounts of
a slightly alkaline pH in the range of 7.5 to 8.0. The
alkaline mucus in response to (1) tactile or irritating
secretions also are rapidly reabsorbed by the villi. This
stimuli on the duodenal mucosa; (2) vagal stimulation,
flow of fluid from the crypts into the villi supplies a
which causes increased Brunner’s glands secretion
watery vehicle for absorption of substances from
concurrently with increase in stomach secretion; and
chyme when it comes in contact with the villi. Thus, the
(3) gastrointestinal hormones, especially secretin.
primary function of the small intestine is to absorb
The function of the mucus secreted by Brunner’s
nutrients and their digestive products into the blood.
glands is to protect the duodenal wall from digestion
by the highly acid gastric juice emptying from the
Mechanism of Secretion of the Watery Fluid. The exact
stomach. In addition, the mucus contains a large excess
mechanism that controls the marked secretion of
of bicarbonate ions, which add to the bicarbonate ions
watery fluid by the crypts of Lieberkühn is not known.
from pancreatic secretion and liver bile in neutralizing
It is believed to involve at least two active secretory
the hydrochloric acid entering the duodenum from the
processes: (1) active secretion of chloride ions into the
stomach.
crypts and (2) active secretion of bicarbonate ions. The
Brunner’s glands are inhibited by sympathetic stim-
secretion of both of these ions causes electrical drag
ulation; therefore, such stimulation in very excitable
as well of positively charged sodium ions through the
persons is likely to leave the duodenal bulb unpro-
membrane and into the secreted fluid. Finally, all these
tected and is perhaps one of the factors that cause this
ions together cause osmotic movement of water.
806
Unit XII Gastrointestinal Physiology
Digestive Enzymes in the Small Intestinal Secretion. When
Mucus in the large intestine protects the intestinal
secretions of the small intestine are collected without
wall against excoriation, but in addition, it provides an
cellular debris, they have almost no enzymes. The en-
adherent medium for holding fecal matter together.
terocytes of the mucosa, especially those that cover the
Furthermore, it protects the intestinal wall from the
villi, do contain digestive enzymes that digest specific
great amount of bacterial activity that takes place
food substances while they are being absorbed through
inside the feces, and, finally, the mucus plus the alka-
the epithelium. These enzymes are the following:
linity of the secretion
(pH of 8.0 caused by large
(1) several peptidases for splitting small peptides into
amounts of sodium bicarbonate) provides a barrier to
amino acids, (2) four enzymes—sucrase, maltase, iso-
keep acids formed in the feces from attacking the
maltase, and lactase—for splitting disaccharides into
intestinal wall.
monosaccharides, and (3) small amounts of intestinal
lipase for splitting neutral fats into glycerol and fatty
Diarrhea Caused by Excess Secretion of Water and Electrolytes
acids.
in Response to Irritation. Whenever a segment of the
The epithelial cells deep in the crypts of Lieberkühn
large intestine becomes intensely irritated, as occurs
continually undergo mitosis, and new cells migrate
when bacterial infection becomes rampant during
along the basement membrane upward out of the
enteritis, the mucosa secretes extra large quantities of
crypts toward the tips of the villi, thus continually
water and electrolytes in addition to the normal viscid
replacing the villus epithelium and also forming new
alkaline mucus. This acts to dilute the irritating factors
digestive enzymes. As the villus cells age, they are
and to cause rapid movement of the feces toward the
finally shed into the intestinal secretions. The life cycle
anus. The result is diarrhea, with loss of large quanti-
of an intestinal epithelial cell is about 5 days. This rapid
ties of water and electrolytes. But the diarrhea also
growth of new cells also allows rapid repair of excori-
washes away irritant factors, which promotes earlier
ations that occur in the mucosa.
recovery from the disease than might otherwise occur.
Regulation of Small Intestine
References
Secretion—Local Stimuli
Barrett KE, Keely SJ: Chloride secretion by the intestinal
By far the most important means for regulating small
epithelium: molecular basis and regulatory aspects. Annu
intestine secretion are local enteric nervous reflexes,
Rev Physiol 62:535, 2000.
especially reflexes initiated by tactile or irritative
Chiang JY: Bile acid regulation of hepatic physiology: III.
stimuli from the chyme in the intestines.
Bile acids and nuclear receptors. Am J Physiol Gastroin-
test Liver Physiol 284:G349, 2003.
Dockray GJ, Varro A, Dimaline R, Wang T: The gastrins: their
Secretions of the
production and biological activities. Annu Rev Physiol
63:119, 2001.
Large Intestine
Dockray GJ, Varro A, Dimaline R: Gastric endocrine cells:
gene expression, processing, and targeting of active prod-
Mucus Secretion. The mucosa of the large intestine, like
ucts. Physiol Rev 76:767,1996.
that of the small intestine, has many crypts of
Flemstrom G, Isenberg JI: Gastroduodenal mucosal alkaline
Lieberkühn; however, unlike the small intestine, there
secretion and mucosal protection. News Physiol Sci 16:23,
are no villi. The epithelial cells contain almost no
2001.
enzymes. Instead, they consist mainly of mucous cells
Fuchs M: Bile acid regulation of hepatic physiology: III. Reg-
that secrete only mucus. The great preponderance of
ulation of bile acid synthesis: past progress and future
secretion in the large intestine is mucus. This mucus
challenges. Am J Physiol Gastrointest Liver Physiol
284:G551, 2003.
contains moderate amounts of bicarbonate ions
Hocker M: Molecular mechanisms of gastrin-dependent
secreted by a few non-mucus-secreting epithelial cells.
gene regulation. Ann N Y Acad Sci 1014:97, 2004.
The rate of secretion of mucus is regulated principally
Jass JR, Walsh MD: Altered mucin expression in the gas-
by direct, tactile stimulation of the epithelial cells
trointestinal tract: a review. J Cell Mol Med 5:327, 2001.
lining the large intestine and by local nervous reflexes
Johnson LR: Gastrointestinal Physiology, 6th ed. St. Louis:
to the mucous cells in the crypts of Lieberkühn.
Mosby, 2001.
Stimulation of the pelvic nerves from the spinal
Kidd JF, Thorn P: Intracellular Ca2+ and Cl- channel activa-
cord, which carry parasympathetic innervation to the
tion in secretory cells. Annu Rev Physiol 62:493, 2000.
distal one half to two thirds of the large intestine, also
Lee MG, Ahn W, Lee JA, Kim JY, et al: Coordination of pan-
can cause marked increase in mucus secretion. This
creatic HCO_ secretion by protein-protein interaction
between membrane transporters. JOP 2(4 Suppl):203,
occurs along with increase in peristaltic motility of the
2001.
colon, which was discussed in Chapter 63.
Marve GM: Nerves and hormones interact to control gall-
During extreme parasympathetic stimulation, often
bladder function. News Physiol Sci 13:64, 1998.
caused by emotional disturbances, so much mucus can
Portincasa P, Di Ciaula A, vanBerge-Henegouwen GP:
occasionally be secreted into the large intestine that
Smooth muscle function and dysfunction in gallbladder
the person has a bowel movement of ropy mucus as
disease. Curr Gastroenterol Rep 6:151, 2004.
often as every 30 minutes; this mucus often contains
Russell DW: The enzymes, regulation, and genetics of bile
little or no fecal material.
acid synthesis. Annu Rev Biochem 72:137, 2003.
Chapter 64
Secretory Functions of the Alimentary Tract
807
Tandler B, Gresik EW, Nagato T, Phillips CJ: Secretion by
Williams JA: Intracellular signaling mechanisms activated
striated ducts of mammalian major salivary glands: review
by cholecystokinin-regulating synthesis and secretion of
from an ultrastructural, functional, and evolutionary per-
digestive enzymes in pancreatic acinar cells. Annu Rev
spective. Anat Rec 264:121, 2001.
Physiol 63:77, 2001.
Trauner M, Boyer JL: Bile salt transporters: molecular char-
Zanner R, Gratzl M, Prinz C: Circle of life of secretory vesi-
acterization, function, and regulation. Physiol Rev 83:633,
cles in gastric enterochromaffin-like cells. Ann N Y Acad
2003.
Sci 971:389, 2002.
C H A P T E R
6
5
Digestion and Absorption in the
Gastrointestinal Tract
The major foods on which the body lives (with the
exception of small quantities of substances such as
vitamins and minerals) can be classified as carbo-
hydrates, fats, and proteins. They generally cannot be
absorbed in their natural forms through the gas-
trointestinal mucosa and, for this reason, are useless
as nutrients without preliminary digestion. There-
fore, this chapter discusses, first, the processes by
which carbohydrates, fats, and proteins are digested into small enough com-
pounds for absorption and, second, the mechanisms by which the digestive end
products as well as water, electrolytes, and other substances are absorbed.
Digestion of the Various Foods by Hydrolysis
Hydrolysis of Carbohydrates. Almost all the carbohydrates of the diet are either
large polysaccharides or disaccharides, which are combinations of monosaccha-
rides bound to one another by condensation. This means that a hydrogen ion
(H+) has been removed from one of the monosaccharides, and a hydroxyl ion
(-OH) has been removed from the next one. The two monosaccharides then
combine with each other at these sites of removal, and the hydrogen and
hydroxyl ions combine to form water (H2O).
When carbohydrates are digested, the above process is reversed and the car-
bohydrates are converted into monosaccharides. Specific enzymes in the diges-
tive juices of the gastrointestinal tract return the hydrogen and hydroxyl ions
from water to the polysaccharides and thereby separate the monosaccharides
from each other. This process, called hydrolysis, is the following (in which
R≤-R¢ is a disaccharide):
R¢¢
¢+
ædigestiveÆR¢¢
+
-R
¢
H O
OH
R
H
2
enzyme
Hydrolysis of Fats. Almost the entire fat portion of the diet consists of triglyc-
erides (neutral fats), which are combinations of three fatty acid molecules con-
densed with a single glycerol molecule. During condensation, three molecules
of water are removed.
Digestion of the triglycerides consists of the reverse process: the fat-
digesting enzymes return three molecules of water to the triglyceride molecule
and thereby split the fatty acid molecules away from the glycerol. Here again,
the digestive process is one of hydrolysis.
Hydrolysis of Proteins. Proteins are formed from multiple amino acids that are
bound together by peptide linkages. At each linkage, a hydroxyl ion has been
removed from one amino acid and a hydrogen ion has been removed from the
succeeding one; thus, the successive amino acids in the protein chain are also
bound together by condensation, and digestion occurs by the reverse effect:
hydrolysis. That is, the proteolytic enzymes return hydrogen and hydroxyl ions
from water molecules to the protein molecules to split them into their con-
stituent amino acids.
808
Chapter 65
Digestion and Absorption in the Gastrointestinal Tract
809
Therefore, the chemistry of digestion is simple
pH of the medium falls below about 4.0. Nevertheless,
because, in the case of all three major types of food,
on the average, before food and its accompanying
the same basic process of hydrolysis is involved. The
saliva do become completely mixed with the gastric
only difference lies in the types of enzymes required
secretions, as much as 30 to 40 per cent of the starches
to promote the hydrolysis reactions for each type of
will have been hydrolyzed mainly to form maltose.
food.
All the digestive enzymes are proteins. Their secre-
Digestion of Carbohydrates in the Small Intestine
tion by the different gastrointestinal glands was dis-
Digestion by Pancreatic Amylase. Pancreatic secre-
cussed in Chapter 64.
tion, like saliva, contains a large quantity of a-amylase
that is almost identical in its function with the
a-amylase of saliva but is several times as powerful.
Digestion of Carbohydrates
Therefore, within 15 to 30 minutes after the chyme
empties from the stomach into the duodenum and
Carbohydrate Foods of the Diet. Only three major sources
mixes with pancreatic juice, virtually all the carbohy-
of carbohydrates exist in the normal human diet. They
drates will have become digested.
are sucrose, which is the disaccharide known popularly
In general, the carbohydrates are almost totally con-
as cane sugar; lactose, which is a disaccharide found
verted into maltose and/or other very small glucose
in milk; and starches, which are large polysaccharides
polymers before passing beyond the duodenum or
present in almost all nonanimal foods, particularly in
upper jejunum.
potatoes and the different types of grains. Other car-
bohydrates ingested to a slight extent are amylose,
glycogen, alcohol, lactic acid, pyruvic acid, pectins, dex-
Hydrolysis of Disaccharides and Small Glucose Polymers
trins, and minor quantities of carbohydrate derivatives
into Monosaccharides by Intestinal Epithelial Enzymes. The
in meats.
enterocytes lining the villi of the small intestine
The diet also contains a large amount of cellulose,
contain four enzymes (lactase, sucrase, maltase, and
which is a carbohydrate. However, no enzymes
a-dextrinase), which are capable of splitting the disac-
capable of hydrolyzing cellulose are secreted in the
charides lactose, sucrose, and maltose, plus other small
human digestive tract. Consequently, cellulose cannot
glucose polymers, into their constituent monosaccha-
be considered a food for humans.
rides. These enzymes are located in the enterocytes cov-
ering the intestinal microvilli brush border, so that the
Digestion of Carbohydrates in the Mouth and Stomach. When
disaccharides are digested as they come in contact with
food is chewed, it is mixed with saliva, which contains
these enterocytes.
the digestive enzyme ptyalin (an a-amylase) secreted
Lactose splits into a molecule of galactose and a
mainly by the parotid glands. This enzyme hydrolyzes
molecule of glucose. Sucrose splits into a molecule of
starch into the disaccharide maltose and other small
fructose and a molecule of glucose. Maltose and other
polymers of glucose that contain three to nine glucose
small glucose polymers all split into multiple molecules
molecules, as shown in Figure 65-1. However, the food
of glucose. Thus, the final products of carbohydrate
remains in the mouth only a short time, so that prob-
digestion are all monosaccharides. They are all water
ably not more than 5 per cent of all the starches will
soluble and are absorbed immediately into the portal
have become hydrolyzed by the time the food is
blood.
swallowed.
In the ordinary diet, which contains far more
However, starch digestion sometimes continues in
starches than all other carbohydrates combined,
the body and fundus of the stomach for as long as 1
glucose represents more than 80 per cent of the final
hour before the food becomes mixed with the stomach
products of carbohydrate digestion, and galactose and
secretions. Then activity of the salivary amylase is
fructose each seldom more than 10 per cent.
blocked by acid of the gastric secretions because the
The major steps in carbohydrate digestion are sum-
amylase is essentially nonactive as an enzyme once the
marized in Figure 65-1.
Starches
Ptyalin (saliva)-20-40%
Pancreatic amylase-50-80%
Maltose and 3 to 9 glucose polymers Lactose
Sucrose
Lactase
Sucrase
Maltase and a-dextrinase
(intestine)
(intestine)
(intestine)
Glucose
Galactose
Fructose
Figure 65-1
Digestion of carbohydrates.
810
Unit XII Gastrointestinal Physiology
Digestion of Proteins
Proteins of the Diet. The dietary proteins are chemically
Pepsin
Proteoses
Proteins
Peptones
long chains of amino acids bound together by peptide
Polypeptides
linkages. A typical linkage is the following:
Trypsin, chymotrypsin, carboxypolypeptidase,
proelastase
NH
H
2
Polypeptides
Peptidases
+
Amino acids
R CH C
OH + H
N
CH COOH
Amino acids
O
R
Figure 65-2
NH2
H
Digestion of proteins.
R CH
C N
CH COOH + H2O
Digestion of Proteins by Pancreatic Secretions. Most
protein digestion occurs in the upper small intestine,
O
R
in the duodenum and jejunum, under the influence
of proteolytic enzymes from pancreatic secretion.
Immediately on entering the small intestine from the
The characteristics of each protein are determined
stomach, the partial breakdown products of the
by the types of amino acids in the protein molecule
protein foods are attacked by major proteolytic
and by the sequential arrangements of these amino
pancreatic enzymes: trypsin, chymotrypsin, carboxy-
acids. The physical and chemical characteristics of dif-
polypeptidase, and proelastase, as shown in Figure
ferent proteins important in human tissues are dis-
65-2.
cussed in Chapter 69.
Both trypsin and chymotrypsin split protein mole-
cules into small polypeptides; carboxypolypeptidase
Digestion of Proteins in the Stomach.
Pepsin, the important
then cleaves individual amino acids from the carboxyl
peptic enzyme of the stomach, is most active at a pH
ends of the polypeptides. Proelastase, in turn, is con-
of 2.0 to 3.0 and is inactive at a pH above about 5.0.
verted into elastase, which then digests elastin fibers
Consequently, for this enzyme to cause digestive
that partially hold meats together.
action on protein, the stomach juices must be acidic.
Only a small percentage of the proteins are digested
As explained in Chapter 64, the gastric glands secrete
all the way to their constituent amino acids by the
a large quantity of hydrochloric acid. This hydrochlo-
pancreatic juices. Most remain as dipeptides and
ric acid is secreted by the parietal (oxyntic) cells in the
tripeptides.
glands at a pH of about 0.8, but by the time it is mixed
with the stomach contents and with secretions from
Digestion of Peptides by Peptidases in the Enterocytes That
the nonoxyntic glandular cells of the stomach, the pH
Line the Small Intestinal Villi. The last digestive stage of
then averages around 2.0 to 3.0, a highly favorable
the proteins in the intestinal lumen is achieved by the
range of acidity for pepsin activity.
enterocytes that line the villi of the small intestine,
One of the important features of pepsin digestion is
mainly in the duodenum and jejunum. These cells have
its ability to digest the protein collagen, an albuminoid
a brush border that consists of hundreds of microvilli
type of protein that is affected little by other digestive
projecting from the surface of each cell. In the mem-
enzymes. Collagen is a major constituent of the inter-
brane of each of these microvilli are multiple pepti-
cellular connective tissue of meats; therefore, for the
dases that protrude through the membranes to the
digestive enzymes of the digestive tract to penetrate
exterior, where they come in contact with the intestinal
meats and digest the other meat proteins, it is first nec-
fluids.
essary that the collagen fibers be digested. Conse-
Two types of peptidase enzymes are especially
quently, in persons who lack pepsin in the stomach
important, aminopolypeptidase and several dipepti-
juices, the ingested meats are less well penetrated by
dases. They succeed in splitting the remaining larger
the other digestive enzymes and, therefore, may be
polypeptides into tripeptides and dipeptides and a few
poorly digested.
into amino acids. Both the amino acids plus the dipep-
As shown in Figure 65-2, pepsin only initiates the
tides and tripeptides are easily transported through
process of protein digestion, usually providing only 10
the microvillar membrane to the interior of the
to 20 per cent of the total protein digestion to convert
enterocyte.
the protein to proteoses, peptones, and a few polypep-
Finally, inside the cytosol of the enterocyte are mul-
tides. This splitting of proteins occurs as a result of
tiple other peptidases that are specific for the remain-
hydrolysis at the peptide linkages between amino
ing types of linkages between amino acids. Within
acids.
minutes, virtually all the last dipeptides and tripeptides
Chapter 65
Digestion and Absorption in the Gastrointestinal Tract
811
Instead, essentially all fat digestion occurs in the small
intestine as follows.
O
CH3
(CH2)16
C O CH2
Emulsification of Fat by Bile Acids and Lecithin. The
first step in fat digestion is physically to break the fat
O
globules into very small sizes so that the water-soluble
Lipase
CH3
(CH2)16
C
O CH + 2H2O
digestive enzymes can act on the globule surfaces. This
process is called emulsification of the fat, and it begins
O
by agitation in the stomach to mix the fat with the
CH3
(CH2)16
C O CH2
products of stomach digestion.
(Tristearin)
Then, most of the emulsification occurs in the duo-
denum under the influence of bile, the secretion from
O
HO CH2
O
the liver that does not contain any digestive enzymes.
CH3
(CH2)16
C O
CH + 2CH3
(CH2)16
C OH
However, bile does contain a large quantity of bile salts
as well as the phospholipid lecithin. Both of these,
HO CH2
but especially the lecithin, are extremely important for
(2-Monoglyceride)
(Stearic acid)
emulsification of the fat. The polar parts (the points
where ionization occurs in water) of the bile salts and
lecithin molecules are highly soluble in water, whereas
Figure 65-3
most of the remaining portions of their molecules are
highly soluble in fat. Therefore, the fat-soluble por-
Hydrolysis of neutral fat catalyzed by lipase.
tions of these liver secretions dissolve in the surface
layer of the fat globules, with the polar portions pro-
jecting. The polar projections, in turn, are soluble in the
surrounding watery fluids, which greatly decreases the
are digested to the final stage to form single amino
interfacial tension of the fat and makes it soluble as
acids; these then pass on through to the other side of
well.
the enterocyte and thence into the blood.
When the interfacial tension of a globule of non-
More than 99 per cent of the final protein digestive
miscible fluid is low, this nonmiscible fluid, on agita-
products that are absorbed are individual amino acids,
tion, can be broken up into many very minute particles
with only rare absorption of peptides and very, very
far more easily than it can when the interfacial tension
rare absorption of whole protein molecules. Even
is great. Consequently, a major function of the bile
these very few absorbed molecules of whole protein
salts and lecithin, especially the lecithin, in the bile is
can sometimes cause serious allergic or immunologic
to make the fat globules readily fragmentable by agi-
disturbances, as discussed in Chapter 34.
tation with the water in the small bowel. This action is
the same as that of many detergents that are widely
used in household cleaners for removing grease.
Digestion of Fats
Each time the diameters of the fat globules are sig-
nificantly decreased as a result of agitation in the small
Fats of the Diet. By far the most abundant fats of the
intestine, the total surface area of the fat increases
diet are the neutral fats, also known as triglycerides,
manyfold. Because the average diameter of the fat
each molecule of which is composed of a glycerol
particles in the intestine after emulsification has
nucleus and three fatty acid side chains, as shown in
occurred is less than 1 micrometer, this represents an
Figure 65-3. Neutral fat is a major constituent in food
increase of as much as 1000-fold in total surface areas
of animal origin but much, much less so in food of
of the fats caused by the emulsification process.
plant origin.
The lipase enzymes are water-soluble compounds
In the usual diet are also small quantities of phos-
and can attack the fat globules only on their surfaces.
pholipids, cholesterol, and cholesterol esters. The phos-
Consequently, it can be readily understood how impor-
pholipids and cholesterol esters contain fatty acid and
tant this detergent function of bile salts and lecithin is
therefore can be considered fats themselves. Choles-
for digestion of fats.
terol, however, is a sterol compound that contains no
fatty acid, but it does exhibit some of the physical and
Digestion of Triglycerides by Pancreatic Lipase. By far the
chemical characteristics of fats; plus, it is derived from
most important enzyme for digestion of the triglyc-
fats and is metabolized similarly to fats. Therefore,
erides is pancreatic lipase, present in enormous quan-
cholesterol is considered, from a dietary point of view,
tities in pancreatic juice, enough to digest within 1
a fat.
minute all triglycerides that it can reach. In addition,
the enterocytes of the small intestine contain still more
Digestion of Fats in the Intestine. A small amount of
lipase, known as enteric lipase, but this is usually not
triglycerides is digested in the stomach by lingual lipase
needed.
that is secreted by lingual glands in the mouth and
swallowed with the saliva. This amount of digestion is
End Products of Fat Digestion. Most of the triglyc-
less than
10 per cent and generally unimportant.
erides of the diet are split by pancreatic lipase into
812
Unit XII Gastrointestinal Physiology
(Bile + Agitation)
Fat
Emulsified fat
Villi
Pancreatic lipase
Emulsified fat
Fatty acids and
Food
2-monoglycerides
movement
Figure 65-4
Digestion of fats.
Valvulae
conniventes
free fatty acids and
2-monoglycerides, as shown in
Figure 65-4.
Role of Bile Salts to Accelerate Fat Digestion—Formation of
Figure 65-5
Micelles. The hydrolysis of triglycerides is a highly
reversible process; therefore, accumulation of mono-
Longitudinal section of the small intestine, showing the valvulae
conniventes covered by villi.
glycerides and free fatty acids in the vicinity of digest-
ing fats quickly blocks further digestion. But the bile
salts play the additional important role of removing
the monoglycerides and free fatty acids from the vicin-
The bile salt micelles play the same role in “ferry-
ity of the digesting fat globules almost as rapidly as
ing” free cholesterol and phospholipid molecule diges-
these end products of digestion are formed. This
tates that they play in “ferrying” monoglycerides and
occurs in the following way.
free fatty acids. Indeed, essentially no cholesterol is
Bile salts, when in high enough concentration in
absorbed without this function of the micelles.
water, have the propensity to form micelles, which are
small spherical, cylindrical globules 3 to 6 nanometers
in diameter composed of 20 to 40 molecules of bile
Basic Principles of
salt. These develop because each bile salt molecule is
Gastrointestinal Absorption
composed of a sterol nucleus that is highly fat-soluble
and a polar group that is highly water-soluble. The
It is suggested that the reader review the basic princi-
sterol nucleus encompasses the fat digestate, forming
ples of transport of substances through cell mem-
a small fat globule in the middle of a resulting micelle,
branes discussed in detail in Chapter 4. The following
with polar groups of bile salts projecting outward to
paragraphs present specialized applications of these
cover the surface of the micelle. Because these polar
transport processes during gastrointestinal absorption.
groups are negatively charged, they allow the entire
micelle globule to dissolve in the water of the diges-
tive fluids and to remain in stable solution until the fat
Anatomical Basis of Absorption
is absorbed into the blood.
The bile salt micelles also act as a transport medium
The total quantity of fluid that must be absorbed each
to carry the monoglycerides and free fatty acids, both
day by the intestines is equal to the ingested fluid
of which would otherwise be relatively insoluble, to
(about 1.5 liters) plus that secreted in the various gas-
the brush borders of the intestinal epithelial cells.
trointestinal secretions (about 7 liters). This comes to
There the monoglycerides and free fatty acids are
a total of 8 to 9 liters. All but about 1.5 liters of this is
absorbed into the blood, as discussed later, but the bile
absorbed in the small intestine, leaving only 1.5 liters
salts themselves are released back into the chyme to
to pass through the ileocecal valve into the colon each
be used again and again for this “ferrying” process.
day.
The stomach is a poor absorptive area of the gas-
Digestion of Cholesterol Esters and Phospholipids. Most cho-
trointestinal tract because it lacks the typical villus
lesterol in the diet is in the form of cholesterol esters,
type of absorptive membrane, and also because the
which are combinations of free cholesterol and one
junctions between the epithelial cells are tight junc-
molecule of fatty acid. Phospholipids also contain fatty
tions. Only a few highly lipid-soluble substances,
acid within their molecules. Both the cholesterol esters
such as alcohol and some drugs like aspirin, can be
and the phospholipids are hydrolyzed by two other
absorbed in small quantities.
lipases in the pancreatic secretion that free the fatty
acids—the enzyme cholesterol ester hydrolase to
Absorptive Surface of the Small Intestinal Mucosa Villi.
hydrolyze the cholesterol ester, and phospholipase A2
Figure 65-5 demonstrates the absorptive surface of the
to hydrolyze the phospholipid.
small intestinal mucosa, showing many folds called
Chapter 65
Digestion and Absorption in the Gastrointestinal Tract
813
Central
Brush
Basement
lacteal
border
membrane
Blood
capillaries
Venules
Arteriole
Central
lacteal
Figure 65-6
Vein
Functional organization of the
Capillaries
villus. A, Longitudinal section.
Artery
B, Cross section showing a base-
ment membrane beneath the
epithelial cells and a brush
border at the other ends of these
cells.
A
B
valvulae conniventes (or folds of Kerckring), which
Figure 65-6B shows a cross section of the villus, and
increase the surface area of the absorptive mucosa
Figure 65-7 shows many small pinocytic vesicles, which
about threefold. These folds extend circularly most of
are pinched-off portions of infolded enterocyte mem-
the way around the intestine and are especially well
brane forming vesicles of absorbed fluids that have
developed in the duodenum and jejunum, where they
been entrapped. Small amounts of substances are
often protrude as much as 8 millimeters into the
absorbed by this physical process of pinocytosis.
lumen.
Extending from the epithelial cell body into each
Also located on the epithelial surface of the small
microvillus of the brush border are multiple actin fila-
intestine all the way down to the ileocecal valve are
ments that contract rhythmically to cause continual
literally millions of small villi. These project about 1
movement of the microvilli, keeping them constantly
millimeter from the surface of the mucosa, as shown
exposed to new quantities of intestinal fluid.
on the surfaces of the valvulae conniventes in Figure
65-5 and in individual detail in Figure 65-6. The villi
lie so close to one another in the upper small intestine
Absorption in the
that they touch in most areas, but their distribution is
Small Intestine
less profuse in the distal small intestine. The presence
of villi on the mucosal surface enhances the total
Absorption from the small intestine each day consists
absorptive area another 10-fold.
of several hundred grams of carbohydrates, 100 or
Finally, each intestinal epithelial cell on each villus
is characterized by a brush border, consisting of as
many as 1000 microvilli 1 micrometer in length and 0.1
micrometer in diameter protruding into the intestinal
chyme; these microvilli are shown in the electron
micrograph in Figure 65-7. This increases the surface
area exposed to the intestinal materials at least
another 20-fold.
Thus, the combination of the folds of Kerckring, the
villi, and the microvilli increases the total absorptive
area of the mucosa perhaps
1000-fold, making a
tremendous total area of 250 or more square meters
for the entire small intestine—about the surface area
of a tennis court.
Figure
65-6A shows in longitudinal section the
general organization of the villus, emphasizing (1) the
Figure 65-7
advantageous arrangement of the vascular system for
Brush border of a gastrointestinal epithelial cell, showing also
absorption of fluid and dissolved material into the
absorbed pinocytic vesicles, mitochondria, and endoplasmic
portal blood and (2) the arrangement of the “central
reticulum lying immediately beneath the brush border. (Courtesy
lacteal” lymph vessel for absorption into the lymph.
Dr. William Lockwood.)
814
Unit XII Gastrointestinal Physiology
more grams of fat, 50 to 100 grams of amino acids, 50
to 100 grams of ions, and 7 to 8 liters of water. The
absorptive capacity of the normal small intestine is far
H2O
greater than this: as much as several kilograms of car-
Na
H2O
bohydrates per day, 500 grams of fat per day, 500 to
700 grams of proteins per day, and 20 or more liters of
water per day. The large intestine can absorb still addi-
H2O
Active transport
tional water and ions, although very few nutrients.
Diffusion
Osmosis
Na
Na (142 mEq/L)
(50 mEq/L)
Absorption of Water
H2O
H2O
Na
H2O
Isosmotic Absorption. Water is transported through the
H2O
intestinal membrane entirely by diffusion. Further-
more, this diffusion obeys the usual laws of osmosis.
Therefore, when the chyme is dilute enough, water is
absorbed through the intestinal mucosa into the blood
Figure 65-8
of the villi almost entirely by osmosis.
Absorption of sodium through the intestinal epithelium. Note also
Conversely, water can also be transported in the
osmotic absorption of water—that is, water
“follows” sodium
opposite direction—from plasma into the chyme. This
through the epithelial membrane.
occurs especially when hyperosmotic solutions are dis-
charged from the stomach into the duodenum. Within
minutes, sufficient water usually will be transferred by
osmosis to make the chyme isosmotic with the plasma.
passively “dragged” by the positive electrical charges
of the sodium ions.
Active transport of sodium through the basolateral
Absorption of Ions
membranes of the cell reduces the sodium concentra-
tion inside the cell to a low value (about 50 mEq/L),
Active Transport of Sodium. Twenty to 30 grams of sodium
as also shown in Figure 65-8. Because the sodium con-
are secreted in the intestinal secretions each day.
centration in the chyme is normally about 142 mEq/L
In addition, the average person eats 5 to 8 grams of
(that is, about equal to that in plasma), sodium moves
sodium each day. Therefore, to prevent net loss of
down this steep electrochemical gradient from the
sodium into the feces, the intestines must absorb 25 to
chyme through the brush border of the epithelial cell
35 grams of sodium each day, which is equal to about
into the epithelial cell cytoplasm. This provides still
one seventh of all the sodium present in the body.
more sodium ions to be transported by the epithelial
Whenever significant amounts of intestinal secre-
cells into the paracellular spaces.
tions are lost to the exterior, as in extreme diarrhea,
the sodium reserves of the body can sometimes be
Osmosis of the Water. The next step in the transport
depleted to lethal levels within hours. Normally,
process is osmosis of water into the paracellular
however, less than 0.5 per cent of the intestinal sodium
spaces. This occurs because a large osmotic gradient
is lost in the feces each day because it is rapidly
has been created by the elevated concentration of ions
absorbed through the intestinal mucosa. Sodium also
in the paracellular space. Much of this osmosis occurs
plays an important role in helping to absorb sugars
through the tight junctions between the apical borders
and amino acids, as we shall see in subsequent
of the epithelial cells, but much also occurs through the
discussions.
cells themselves. And osmotic movement of water
The basic mechanism of sodium absorption from the
creates flow of fluid into and through the paracellular
intestine is shown in Figure 65-8. The principles of this
spaces and, finally, into the circulating blood of the
mechanism, discussed in Chapter 4, are also essentially
villus.
the same as for absorption of sodium from the
gallbladder and renal tubules as discussed in Chapter
Aldosterone Greatly Enhances Sodium Absorption. When a
27.
person becomes dehydrated, large amounts of aldos-
The motive power for sodium absorption is pro-
terone almost always are secreted by the cortices of
vided by active transport of sodium from inside the
the adrenal glands. Within 1 to 3 hours this aldosterone
epithelial cells through the basal and side walls of
causes increased activation of the enzyme and trans-
these cells into paracellular spaces. This is demon-
port mechanisms for all aspects of sodium absorption
strated by the heavy red arrows in Figure 65-8. This
by the intestinal epithelium. And the increased sodium
active transport obeys the usual laws of active trans-
absorption in turn causes secondary increases in
port: it requires energy, and the energy process is cat-
absorption of chloride ions, water, and some other
alyzed by appropriate adenosine triphosphatase
substances.
enzymes in the cell membrane (see Chapter 4). Part of
This effect of aldosterone is especially important in
the sodium is absorbed along with chloride ions; in
the colon because it allows virtually no loss of sodium
fact, the negatively charged chloride ions are mainly
chloride in the feces and also little water loss. Thus, the
Chapter 65
Digestion and Absorption in the Gastrointestinal Tract
815
function of aldosterone in the intestinal tract is the
Extreme diarrheal secretion is initiated by entry of
same as that achieved by aldosterone in the renal
a subunit of cholera toxin into the epithelial cells.
tubules, which also serves to conserve sodium chloride
This stimulates formation of excess cyclic adenosine
monophosphate, which opens tremendous numbers of
and water in the body when a person becomes
chloride channels, allowing chloride ions to flow rapidly
dehydrated.
from inside the cell into the intestinal crypts. In turn, this
is believed to activate a sodium pump that pumps
Absorption of Chloride Ions in the Duodenum and Jejunum. In
sodium ions into the crypts to go along with the chlo-
the upper part of the small intestine, chloride ion
ride ions. Finally, all this extra sodium chloride causes
absorption is rapid and occurs mainly by diffusion—
extreme osmosis of water from the blood, thus provid-
that is, absorption of sodium ions through the epithe-
ing rapid flow of fluid along with the salt. All this excess
lium creates electronegativity in the chyme and
fluid washes away most of the bacteria and is of value
in combating the disease, but too much of a good thing
electropositivity in the paracellular spaces between
can be lethal because of serious dehydration of the
the epithelial cells. Then chloride ions move along this
whole body that might ensue. In most instances, the life
electrical gradient to “follow” the sodium ions.
of a cholera victim can be saved by administration of
tremendous amounts of sodium chloride solution to
Absorption of Bicarbonate Ions in the Duodenum and Jejunum.
make up for the loss.
Often large quantities of bicarbonate ions must be
reabsorbed from the upper small intestine because
Absorption of Other Ions. Calcium ions are actively
large amounts of bicarbonate ions have been secreted
absorbed into the blood especially from the duode-
into the duodenum in both pancreatic secretion and
num, and the amount of calcium ion absorption is very
bile. The bicarbonate ion is absorbed in an indirect way
exactly controlled to supply exactly the daily need of
as follows: When sodium ions are absorbed, moderate
the body for calcium. One important factor controlling
amounts of hydrogen ions are secreted into the lumen
calcium absorption is parathyroid hormone secreted
of the gut in exchange for some of the sodium. These
by the parathyroid glands, and another is vitamin D.
hydrogen ions in turn combine with the bicarbonate
Parathyroid hormone activates vitamin D, and the
ions to form carbonic acid (H2CO3), which then disso-
activated vitamin D in turn greatly enhances
ciates to form water and carbon dioxide. The water
calcium absorption. These effects are discussed in
remains as part of the chyme in the intestines, but the
Chapter 79.
carbon dioxide is readily absorbed into the blood and
Iron ions are also actively absorbed from the small
subsequently expired through the lungs. Thus, this is
intestine. The principles of iron absorption and regu-
so-called “active absorption of bicarbonate ions.” It is
lation of its absorption in proportion to the body’s
the same mechanism that occurs in the tubules of the
need for iron, especially for the formation of hemo-
kidneys.
globin, are discussed in Chapter 32.
Potassium, magnesium, phosphate, and probably still
Secretion of Bicarbonate Ions in the Ileum
other ions can also be actively absorbed through the
and Large Intestine—Simultaneous Absorption
intestinal mucosa. In general, the monovalent ions are
of Chloride Ions
absorbed with ease and in great quantities. Conversely,
The epithelial cells on the surfaces of the villi in the
bivalent ions are normally absorbed in only small
ileum as well as on all surfaces of the large intestine
amounts; for example, maximum absorption of
have a special capability of secreting bicarbonate ions
calcium ions is only 1/50 as great as the normal absorp-
in exchange for absorption of chloride ions. This is
tion of sodium ions. Fortunately, only small quantities
important because it provides alkaline bicarbonate
of the bivalent ions are normally required daily by the
ions that neutralize acid products formed by bacteria
body.
in the large intestine.
Absorption of Nutrients
Extreme Secretion of Chloride Ions, Sodium Ions, and Water from
the Large Intestine Epithelium in Some Types of Diarrhea. Deep
Absorption of Carbohydrates
in the spaces between the intestinal epithelial folds are
Essentially all the carbohydrates in the food are
immature epithelial cells that continually divide to form
new epithelial cells. These in turn spread outward over
absorbed in the form of monosaccharides; only a small
the luminal surfaces of the intestines. While still in the
fraction are absorbed as disaccharides and almost
deep folds, the epithelial cells secrete sodium chloride
none as larger carbohydrate compounds. By far
and water into the intestinal lumen. This secretion in
the most abundant of the absorbed monosaccharides
turn is reabsorbed by the older epithelial cells outside
is glucose, usually accounting for more than 80 per cent
the folds, thus providing flow of water for absorbing
of carbohydrate calories absorbed. The reason for
intestinal digestates.
this is that glucose is the final digestion product of our
The toxins of cholera and of some other types of diar-
most abundant carbohydrate food, the starches. The
rheal bacteria can stimulate the fold secretion so greatly
remaining 20 per cent of absorbed monosaccharides
that this secretion often becomes much greater than can
are composed almost entirely of galactose and fruc-
be reabsorbed, thus sometimes causing loss of 5 to 10
liters of water and sodium chloride as diarrhea each day.
tose, the galactose derived from milk and the fructose
Within 1 to 5 days, many severely affected patients die
as one of the monosaccharides digested from cane
from this loss of fluid alone.
sugar.
816
Unit XII Gastrointestinal Physiology
Virtually all the monosaccharides are absorbed by
microvillus membrane with a specific transport protein
an active transport process. Let us first discuss the
that requires sodium binding before transport can
absorption of glucose.
occur. After binding, the sodium ion then moves down
its electrochemical gradient to the interior of the cell
Glucose Is Transported by a Sodium Co-Transport Mechanism.
and pulls the amino acid or peptide along with it. This
In the absence of sodium transport through the intes-
is called co-transport (or secondary active transport)
tinal membrane, virtually no glucose can be absorbed.
of the amino acids and peptides. A few amino acids do
The reason is that glucose absorption occurs in a co-
not require this sodium co-transport mechanism but
transport mode with active transport of sodium.
instead are transported by special membrane trans-
There are two stages in the transport of sodium
port proteins in the same way that fructose is trans-
through the intestinal membrane. First is active trans-
ported, by facilitated diffusion.
port of sodium ions through the basolateral mem-
At least five types of transport proteins for trans-
branes of the intestinal epithelial cells into the blood,
porting amino acids and peptides have been found in
thereby depleting sodium inside the epithelial cells.
the luminal membranes of intestinal epithelial cells.
Second, decrease of sodium inside the cells causes
This multiplicity of transport proteins is required
sodium from the intestinal lumen to move through the
because of the diverse binding properties of different
brush border of the epithelial cells to the cell interiors
amino acids and peptides.
by a process of facilitated diffusion. That is, a sodium
ion combines with a transport protein, but the trans-
Absorption of Fats
port protein will not transport the sodium to the inte-
Earlier in this chapter, it was pointed out that when
rior of the cell until the protein itself also combines
fats are digested to form monoglycerides and free fatty
with some other appropriate substance such as
acids, both of these digestive end products first become
glucose. Fortunately, intestinal glucose also combines
dissolved in the central lipid portions of bile micelles.
simultaneously with the same transport protein, and
Because the molecular dimensions of these micelles
then both the sodium ion and glucose molecule are
are only 3 to 6 nanometers in diameter, and because
transported together to the interior of the cell. Thus,
of their highly charged exterior, they are soluble in
the low concentration of sodium inside the cell liter-
chyme. In this form, the monoglycerides and free fatty
ally “drags” sodium to the interior of the cell and along
acids are carried to the surfaces of the microvilli of the
with it the glucose at the same time. Once inside the
intestinal cell brush border and then penetrate into the
epithelial cell, other transport proteins and enzymes
recesses among the moving, agitating microvilli. Here,
cause facilitated diffusion of the glucose through the
both the monoglycerides and fatty acids diffuse imme-
cell’s basolateral membrane into the paracellular
diately out of the micelles and into the interior of
space and from there into the blood.
the epithelial cells, which is possible because the lipids
To summarize, it is the initial active transport of
are also soluble in the epithelial cell membrane. This
sodium through the basolateral membranes of the
leaves the bile micelles still in the chyme, where they
intestinal epithelial cells that provides the eventual
function again and again to help absorb still more
motive force for moving glucose also through the
monoglycerides and fatty acids.
membranes.
Thus, the micelles perform a “ferrying” function that
is highly important for fat absorption. In the presence
Absorption of Other Monosaccharides. Galactose is trans-
of an abundance of bile micelles, about 97 per cent of
ported by almost exactly the same mechanism as
the fat is absorbed; in the absence of the bile micelles,
glucose. Conversely, fructose transport does not occur
only 40 to 50 per cent can be absorbed.
by the sodium co-transport mechanism. Instead, fruc-
After entering the epithelial cell, the fatty acids and
tose is transported by facilitated diffusion all the way
monoglycerides are taken up by the cell’s smooth
through the intestinal epithelium but not coupled with
endoplasmic reticulum; here, they are mainly used to
sodium transport.
form new triglycerides that are subsequently released
Much of the fructose, on entering the cell, becomes
in the form of chylomicrons through the base of the
phosphorylated, then converted to glucose, and finally
epithelial cell, to flow upward through the thoracic
transported in the form of glucose the rest of the way
lymph duct and empty into the circulating blood.
into the blood. Because fructose is not co-transported
with sodium, its overall rate of transport is only about
Direct Absorption of Fatty Acids into the Portal Blood. Small
one half that of glucose or galactose.
quantities of short- and medium-chain fatty acids, such
as those from butterfat, are absorbed directly into the
Absorption of Proteins
portal blood rather than being converted into triglyc-
As explained earlier in the chapter, most proteins,
erides and absorbed by way of the lymphatics. The
after digestion, are absorbed through the luminal
cause of this difference between short- and long-chain
membranes of the intestinal epithelial cells in the form
fatty acid absorption is that the short-chain fatty acids
of dipeptides, tripeptides, and a few free amino acids.
are more water-soluble and mostly are not recon-
The energy for most of this transport is supplied by a
verted into triglycerides by the endoplasmic reticulum.
sodium co-transport mechanism in the same way that
This allows direct diffusion of these short-chain fatty
sodium co-transport of glucose occurs. That is, most
acids from the intestinal epithelial cells directly into
peptide or amino acid molecules bind in the cell’s
the capillary blood of the intestinal villi.
Chapter 65
Digestion and Absorption in the Gastrointestinal Tract
817
Absorption in the Large
source of energy is significant, although it is of negligi-
ble importance in human beings.
Intestine: Formation of Feces
Other substances formed as a result of bacterial activ-
, thiamine, riboflavin, and
ity are vitamin K, vitamin B
12
About 1500 milliliters of chyme normally pass through
various gases that contribute to flatus in the colon, espe-
the ileocecal valve into the large intestine each day.
cially carbon dioxide, hydrogen gas, and methane.
Most of the water and electrolytes in this chyme are
The bacteria-formed vitamin K is especially important
absorbed in the colon, usually leaving less than 100
because the amount of this vitamin in the daily ingested
milliliters of fluid to be excreted in the feces. Also,
foods is normally insufficient to maintain adequate
blood coagulation.
essentially all the ions are absorbed, leaving only 1 to
5 milliequivalents each of sodium and chloride ions to
be lost in the feces.
Composition of the Feces. The feces normally are about
Most of the absorption in the large intestine occurs
three-fourths water and one-fourth solid matter that
itself is composed of about 30 per cent dead bacteria, 10
in the proximal one half of the colon, giving this
to 20 per cent fat, 10 to 20 per cent inorganic matter, 2
portion the name absorbing colon, whereas the distal
to
3 per cent protein, and
30 per cent undigested
colon functions principally for feces storage until a
roughage from the food and dried constituents of diges-
propitious time for feces excretion and is therefore
tive juices, such as bile pigment and sloughed epithelial
called the storage colon.
cells. The brown color of feces is caused by stercobilin
and urobilin, derivatives of bilirubin. The odor is caused
Absorption and Secretion of Electrolytes and Water. The
principally by products of bacterial action; these prod-
mucosa of the large intestine, like that of the small
ucts vary from one person to another, depending on
intestine, has a high capability for active absorption of
each person’s colonic bacterial flora and on the type of
food eaten. The actual odoriferous products include
sodium, and the electrical potential gradient created
indole, skatole, mercaptans, and hydrogen sulfide.
by absorption of the sodium causes chloride absorp-
tion as well. The tight junctions between the epithelial
cells of the large intestinal epithelium are much tighter
than those of the small intestine. This prevents signif-
References
icant amounts of back-diffusion of ions through these
junctions, thus allowing the large intestinal mucosa to
Adibi SA: Regulation of expression of the intestinal
oligopeptide transporter (Pept-1) in health and disease.
absorb sodium ions far more completely—that is,
Am J Physiol Gastrointest Liver Physiol 285:G779, 2003.
against a much higher concentration gradient—than
Barrett KE, Keely SJ: Chloride secretion by the intestinal
can occur in the small intestine. This is especially true
epithelium: molecular basis and regulatory aspects. Annu
when large quantities of aldosterone are available
Rev Physiol 62:535, 2000.
because aldosterone greatly enhances sodium trans-
Daniel H: Molecular and integrative physiology of intestinal
port capability.
peptide transport. Annu Rev Physiol 66:361, 2004.
In addition, as occurs in the distal portion of the
Farhadi A, Banan A, Fields J, Keshavarzian A: Intestinal
small intestine, the mucosa of the large intestine
barrier: an interface between health and disease. J Gas-
secretes bicarbonate ions while it simultaneously
troenterol Hepatol 18:479, 2003.
absorbs an equal number of chloride ions in an
Ferraris RP, Diamond J: Regulation of intestinal sugar trans-
port. Physiol Rev 77:257, 1997.
exchange transport process that has already been
Field M: Intestinal ion transport and the pathophysiology of
described. The bicarbonate helps neutralize the acidic
diarrhea. J Clin Invest 111:931, 2003.
end products of bacterial action in the large intestine.
Greig E, Sandle GI: Diarrhea in ulcerative colitis. The role
Absorption of sodium and chloride ions creates an
of altered colonic sodium transport. Ann N Y Acad Sci
osmotic gradient across the large intestinal mucosa,
915:327, 2000.
which in turn causes absorption of water.
Hershberg RM: The epithelial cell cytoskeleton and intra-
cellular trafficking. V. Polarized compartmentalization of
Maximum Absorption Capacity of the Large Intestine. The
antigen processing and Toll-like receptor signaling in
large intestine can absorb a maximum of 5 to 8 liters
intestinal epithelial cells. Am J Physiol Gastrointest Liver
of fluid and electrolytes each day. When the total quan-
Physiol 283:G833, 2002.
Johnson LR: Gastrointestinal Physiology, 6th ed. St. Louis:
tity entering the large intestine through the ileocecal
Mosby, 2001.
valve or by way of large intestine secretion exceeds
Kullak-Ublick GA, Stieger B, Meier PJ: Enterohepatic bile
this amount, the excess appears in the feces as diar-
salt transporters in normal physiology and liver disease.
rhea. As noted earlier in the chapter, toxins from
Gastroenterology 126:322, 2004.
cholera or certain other bacterial infections often
Kunzelmann K, Mall M: Electrolyte transport in the mam-
cause the crypts in the terminal ileum and in the large
malian colon: mechanisms and implications for disease.
intestine to secrete 10 or more liters of fluid each day,
Physiol Rev 82:245, 2002.
leading to severe and sometimes lethal diarrhea.
Meier PJ, Stieger B: Bile salt transporters. Annu Rev Physiol
64:635, 2002.
Bacterial Action in the Colon. Numerous bacteria, especially
Pacha J: Development of intestinal transport function in
colon bacilli, are present even normally in the absorbing
mammals. Physiol Rev 80:1633, 2000.
colon. They are capable of digesting small amounts of
Pappenheimer JR: Intestinal absorption of hexoses and
cellulose, in this way providing a few calories of extra
amino acids: from apical cytosol to villus capillaries. J
nutrition for the body. In herbivorous animals, this
Membr Biol 184:233, 2001.
818
Unit XII Gastrointestinal Physiology
Peng JB, Brown EM, Hediger MA: Apical entry channels in
Said HM: Recent advances in carrier-mediated intestinal
calcium-transporting epithelia. News Physiol Sci 18:158,
absorption of water-soluble vitamins. Annu Rev Physiol
2003.
2004;66:419-46.
Reuss L: One-hundred years of inquiry: the mechanism of
Stevens CE, Hume ID: Contributions of microbes in verte-
glucose absorption in the intestine. Annu Rev Physiol
brate gastrointestinal tract to production and conservation
62:939, 2000.
of nutrients. Physiol Rev 78:393, 1998.
Rossier BC, Pradervand S, Schild L, Hummler E: Epithelial
Topping DL, Clifton PM: Short-chain fatty acids and human
sodium channel and the control of sodium balance: inter-
colonic function: roles of resistant starch and nonstarch
action between genetic and environmental factors. Annu
polysaccharides. Physiol Rev 81:1031, 2001.
Rev Physiol 64:877, 2002.
Rothman S, Liebow C, Isenman L: Conservation of digestive
enzymes. Physiol Rev 82:1, 2002.
C H A P T E R
6
6
Physiology of Gastrointestinal
Disorders
Effective therapy for most gastrointestinal disorders
depends on a basic knowledge of gastrointestinal phys-
iology. The purpose of this chapter, therefore, is to
discuss a few representative types of gastrointestinal
malfunction that have special physiologic bases or
consequences.
Disorders of Swallowing and of the Esophagus
Paralysis of the Swallowing Mechanism. Damage to the 5th, 9th, or 10th cerebral nerve
can cause paralysis of significant portions of the swallowing mechanism. Also, a few
diseases, such as poliomyelitis or encephalitis, can prevent normal swallowing by
damaging the swallowing center in the brain stem. Finally, paralysis of the swal-
lowing muscles, as occurs in muscle dystrophy or in failure of neuromuscular trans-
mission in myasthenia gravis or botulism, can also prevent normal swallowing.
When the swallowing mechanism is partially or totally paralyzed, the abnormal-
ities that can occur include (1) complete abrogation of the swallowing act so
that swallowing cannot occur, (2) failure of the glottis to close so that food passes
into the lungs instead of the esophagus, and (3) failure of the soft palate and
uvula to close the posterior nares so that food refluxes into the nose during
swallowing.
One of the most serious instances of paralysis of the swallowing mechanism
occurs when patients are under deep anesthesia. Often, while on the operating table,
they vomit large quantities of materials from the stomach into the pharynx; then,
instead of swallowing the materials again, they simply suck them into the trachea
because the anesthetic has blocked the reflex mechanism of swallowing. As a result,
such patients occasionally choke to death on their own vomitus.
Achalasia and Megaesophagus. Achalasia is a condition in which the lower esophageal
sphincter fails to relax during swallowing. As a result, food swallowed into the
esophagus then fails to pass from the esophagus into the stomach. Pathological
studies have shown damage in the neural network of the myenteric plexus in the
lower two thirds of the esophagus. As a result, the musculature of the lower esoph-
agus remains spastically contracted, and the myenteric plexus has lost its ability to
transmit a signal to cause “receptive relaxation” of the gastroesophageal sphincter
as food approaches this sphincter during swallowing.
When achalasia becomes severe, the esophagus often cannot empty the swallowed
food into the stomach for many hours, instead of the few seconds that is the normal
time. Over months and years, the esophagus becomes tremendously enlarged until
it often can hold as much as 1 liter of food, which often becomes putridly infected
during the long periods of esophageal stasis. The infection may also cause ulcera-
tion of the esophageal mucosa, sometimes leading to severe substernal pain or even
rupture and death. Considerable benefit can be achieved by stretching the lower
end of the esophagus by means of a balloon inflated on the end of a swallowed
esophageal tube. Antispasmotic drugs (drugs that relax smooth muscle) can also be
helpful.
Disorders of the Stomach
Gastritis—Inflammation of the Gastric Mucosa. Mild to moderate chronic gastritis is
exceedingly common in the population as a whole, especially in the middle to later
years of adult life.
819
820
Unit XII Gastrointestinal Physiology
The inflammation of gastritis may be only superficial
bines with vitamin B12 in the stomach and protects it
and therefore not very harmful, or it can penetrate
from being digested and destroyed as it passes into the
deeply into the gastric mucosa, in many long-standing
small intestine. Then, when the intrinsic factor-vitamin
cases causing almost complete atrophy of the gastric
B12 complex reaches the terminal ileum, the intrinsic
mucosa. In a few cases, gastritis can be acute and severe,
factor binds with receptors on the ileal epithelial
with ulcerative excoriation of the stomach mucosa by
surface. This in turn makes it possible for the vitamin
the stomach’s own peptic secretions.
B12 to be absorbed.
Research suggests that much gastritis is caused
In the absence of intrinsic factor, only about 1/50 of
by chronic bacterial infection of the gastric mucosa.
the vitamin B12
is absorbed. And, without intrinsic
This often can be treated successfully by an intensive
factor, an adequate amount of vitamin B12 is not made
regimen of antibacterial therapy.
available from the foods to cause young, newly forming
In addition, certain ingested irritant substances can be
red blood cells to mature in the bone marrow. The result
especially damaging to the protective gastric mucosal
is pernicious anemia. This is discussed in more detail in
barrier—that is, to the mucous glands and to the tight
Chapter 32.
epithelial junctions between the gastric lining cells—
often leading to severe acute or chronic gastritis. Two of
the most common of these substances are excesses of
Peptic Ulcer
alcohol or aspirin.
A peptic ulcer is an excoriated area of stomach or intes-
tinal mucosa caused principally by the digestive action
Gastric Barrier and Its Penetration in Gastritis. Absorp-
of gastric juice or upper small intestinal secretions.
tion of food from the stomach directly into the blood is
Figure 66-1 shows the points in the gastrointestinal tract
normally slight. This low level of absorption is mainly
at which peptic ulcers most frequently occur, demon-
caused by two specific features of the gastric mucosa:
strating that the most frequent site is within a few
(1) it is lined with highly resistant mucous cells that
centimeters of the pylorus. In addition, peptic ulcers fre-
secrete a viscid and adherent mucus and (2) it has tight
quently occur along the lesser curvature of the antral
junctions between the adjacent epithelial cells. These
end of the stomach or, more rarely, in the lower end of
two together plus other impediments to gastric absorp-
the esophagus where stomach juices frequently reflux.
tion are called the “gastric barrier.”
A type of peptic ulcer called a marginal ulcer also often
The gastric barrier normally is resistant enough to dif-
occurs wherever a surgical opening such as a gastro-
fusion so that even the highly concentrated hydrogen
jejunostomy has been made between the stomach and
ions of the gastric juice, averaging about 100,000 times
the jejunum of the small intestine.
the concentration of hydrogen ions in plasma, seldom
diffuse even to the slightest extent through the lining
Basic Cause of Peptic Ulceration. The usual cause of peptic
mucus as far as the epithelial membrane itself. In gas-
ulceration is an imbalance between the rate of secretion
tritis, the permeability of the barrier is greatly increased.
of gastric juice and the degree of protection afforded by
The hydrogen ions do then diffuse into the stomach
(1) the gastroduodenal mucosal barrier and (2) the neu-
epithelium, creating additional havoc and leading to a
tralization of the gastric acid by duodenal juices. It will
vicious circle of progressive stomach mucosal damage
be recalled that all areas normally exposed to gastric
and atrophy. It also makes the mucosa susceptible to
juice are well supplied with mucous glands, beginning
digestion by the peptic digestive enzymes, thus fre-
with compound mucous glands in the lower esophagus
quently resulting in gastric ulcer.
plus the mucous cell coating of the stomach mucosa, the
mucous neck cells of the gastric glands, the deep pyloric
Gastric Atrophy. In many people who have chronic gas-
glands that secrete mainly mucus, and, finally, the glands
tritis, the mucosa gradually becomes more and more
of Brunner of the upper duodenum, which secrete a
atrophic until little or no gastric gland digestive secre-
highly alkaline mucus.
tion remains. It is also believed that some people
In addition to the mucus protection of the mucosa,
develop autoimmunity against the gastric mucosa, this
the duodenum is protected by the alkalinity of the small
also leading eventually to gastric atrophy. Loss of the
stomach secretions in gastric atrophy leads to achlorhy-
Cardia
dria and, occasionally, to pernicious anemia.
Causes:
Achlorhydria
(and Hypochlorhydria). Achlorhydria
1. High acid and peptic content
means simply that the stomach fails to secrete
2. Irritation
hydrochloric acid; it is diagnosed when the pH of the
3. Poor blood supply
gastric secretions fails to decrease below
6.5 after
4. Poor secretion of mucus
Ulcer
maximal stimulation. Hypochlorhydria means dimin-
5. Infection, H. pylori
sites
ished acid secretion. When acid is not secreted, pepsin
also usually is not secreted; even when it is, the lack of
Pylorus
acid prevents it from functioning because pepsin
requires an acid medium for activity.
Marginal
Pernicious Anemia in Gastric Atrophy. Pernicious
ulcer
anemia is a common accompaniment of gastric atrophy
and achlorhydria. Normal gastric secretions contain a
glycoprotein called intrinsic factor, secreted by the same
parietal cells that secrete hydrochloric acid. Intrinsic
Figure 66-1
factor must be present for adequate absorption of
vitamin B12 from the ileum. That is, intrinsic factor com-
Peptic ulcer. H. pylori, Helicobacter pylori.
Chapter 66
Physiology of Gastrointestinal Disorders
821
intestinal secretions. Especially important is pancreatic
changed immensely. Initial reports are that almost all
secretion, which contains large quantities of sodium
patients with peptic ulceration can be treated effectively
bicarbonate that neutralize the hydrochloric acid of the
by two measures: (1) use of antibiotics along with other
gastric juice, thus also inactivating pepsin and prevent-
agents to kill infectious bacteria and (2) administration
ing digestion of the mucosa. In addition, large amounts
of an acid-suppressant drug, especially ranitidine, an
of bicarbonate ions are provided in (1) the secretions of
antihistaminic that blocks the stimulatory effect of his-
the large Brunner’s glands in the first few centimeters
tamine on gastric gland histamine2
receptors, thus
of the duodenal wall and (2) in bile coming from the
reducing gastric acid secretion by 70 to 80 per cent.
liver.
In the past, before these approaches to peptic ulcer
Finally, two feedback control mechanisms normally
therapy were developed, it was often necessary to
ensure that this neutralization of gastric juices is com-
remove as much as four fifths of the stomach, thus
plete, as follows:
reducing stomach acid-peptic juices enough to cure
1. When excess acid enters the duodenum, it reflexly
most patients. Another therapy was to cut the two vagus
inhibits gastric secretion and peristalsis in the
nerves that supply parasympathetic stimulation to the
stomach, both by nervous reflexes and by hormonal
gastric glands. This blocked almost all secretion of acid
feedback from the duodenum, thereby decreasing
and pepsin and often cured the ulcer or ulcers within 1
the rate of gastric emptying.
week after the operation was performed. However,
2. The presence of acid in the small intestine liberates
much of the basal stomach secretion returned after a
secretin from the intestinal mucosa, which then
few months, and in many patients the ulcer also
passes by way of the blood to the pancreas to
returned.
promote rapid secretion of pancreatic juice. This
The newer physiologic approaches to therapy may
juice also contains a high concentration of sodium
prove to be miraculous. Even so, in a few instances, the
bicarbonate, thus making still more sodium
patient’s condition is so severe—including massive
bicarbonate available for neutralization of the acid.
bleeding from the ulcer—that heroic operative proce-
Therefore, a peptic ulcer can be caused in either of
dures often must still be used.
two ways: (1) excess secretion of acid and pepsin by the
gastric mucosa or (2) diminished ability of the gastro-
duodenal mucosal barrier to protect against the diges-
Disorders of the
tive properties of the stomach acid-pepsin secretion.
Small Intestine
Specific Causes of Peptic Ulcer in
Abnormal Digestion of Food in the
the Human Being
Small Intestine—Pancreatic Failure
Bacterial Infection by Helicobacter pylori Breaks Down the Gas-
A serious cause of abnormal digestion is failure of the
troduodenal Mucosal Barrier. Many peptic ulcer patients
pancreas to secrete pancreatic juice into the small intes-
have been found to have chronic infection of the ter-
tine. Lack of pancreatic secretion frequently occurs (1)
minal portions of the gastric mucosa and initial portions
in pancreatitis (which is discussed later), (2) when the
of the duodenal mucosa, infection most often caused by
pancreatic duct is blocked by a gallstone at the papilla
the bacterium Helicobacter pylori. Once this infection
of Vater, or (3) after the head of the pancreas has been
begins, it can last a lifetime unless it is eradicated by
removed because of malignancy.
antibacterial therapy. Furthermore, the bacterium is
Loss of pancreatic juice means loss of trypsin, chy-
capable of penetrating the mucosal barrier both by
motrypsin, carboxypolypeptidase, pancreatic amylase,
virtue of its physical capability to burrow through the
pancreatic lipase, and still a few other digestive
barrier and by releasing bacterial digestive enzymes
enzymes. Without these enzymes, as much as 60 per cent
that liquefy the barrier. As a result, the strong acidic
of the fat entering the small intestine may be unab-
digestive juices of the stomach secretions can then pen-
sorbed, as well as one third to one half of the proteins
etrate into the underlying epithelium and literally digest
and carbohydrates. As a result, large portions of the
the gastrointestinal wall, thus leading to peptic
ingested food cannot be used for nutrition, and copious,
ulceration.
fatty feces are excreted.
Other Causes of Ulceration. In many people who have
Pancreatitis. Pancreatitis means inflammation of the
peptic ulcer in the initial portion of the duodenum, the
pancreas, and this can occur in the form of either acute
rate of gastric acid secretion is greater than normal,
pancreatitis or chronic pancreatitis.
sometimes as much as twice normal. Although part of
The most common cause of pancreatitis is drinking
this increased secretion may be stimulated by bacterial
excess alcohol, and the second most common cause is
infection, studies in both animals and human beings
blockage of the papilla of Vater by a gallstone; the two
have shown that excess secretion of gastric juices for
together account for more than 90 per cent of all cases.
any reason (for instance, even in psychic disturbances)
When a gallstone blocks the papilla of Vater, this blocks
may cause peptic ulceration.
the main secretory duct from the pancreas as well as the
Other factors that predispose to ulcers include (1)
common bile duct. The pancreatic enzymes are then
smoking, presumably because of increased nervous
dammed up in the ducts and acini of the pancreas. Even-
stimulation of the stomach secretory glands; (2) alcohol,
tually, so much trypsinogen accumulates that it over-
because it tends to break down the mucosal barrier; and
comes the trypsin inhibitor in the secretions, and a small
(3) aspirin and other non-steroidal anti-inflammatory
quantity of trypsinogen becomes activated to form
drugs that also have a strong propensity for breaking
trypsin. Once this happens, the trypsin activates still
down this barrier.
more trypsinogen as well as chymotrypsinogen and car-
Physiology of Treatment. Since discovery of the bacterial
boxypolypeptidase, resulting in a vicious circle until
infectious basis for much peptic ulceration, therapy has
most of the proteolytic enzymes in the pancreatic ducts
822
Unit XII Gastrointestinal Physiology
and acini become activated. These enzymes rapidly
of dry, hard feces in the descending colon that accumu-
digest large portions of the pancreas itself, sometimes
late because of over-absorption of fluid. Any pathology
completely and permanently destroying the ability of
of the intestines that obstructs movement of intestinal
the pancreas to secrete digestive enzymes.
contents, such as tumors, adhesions that constrict the
intestines, or ulcers, can cause constipation. A frequent
functional cause of constipation is irregular bowel
Malabsorption by the Small
habits that have developed through a lifetime of inhibi-
Intestinal Mucosa—Sprue
tion of the normal defecation reflexes.
Infants are seldom constipated, but part of their train-
Occasionally, nutrients are not adequately absorbed
ing in the early years of life requires that they learn to
from the small intestine even though the food has
control defecation; this control is effected by inhibiting
become well digested. Several diseases can cause
the natural defecation reflexes. Clinical experience
decreased absorption by the mucosa; they are often
shows that if one does not allow defecation to occur
classified together under the general term “sprue.” Mal-
when the defecation reflexes are excited or if one over-
absorption also can occur when large portions of the
uses laxatives to take the place of natural bowel func-
small intestine have been removed.
tion, the reflexes themselves become progressively less
strong over months or years, and the colon becomes
Nontropical Sprue. One type of sprue, called variously
atonic. For this reason, if a person establishes regular
idiopathic sprue, celiac disease (in children), or gluten
bowel habits early in life, usually defecating in the
enteropathy, results from the toxic effects of gluten
morning after breakfast when the gastrocolic and duo-
present in certain types of grains, especially wheat and
denocolic reflexes cause mass movements in the large
rye. Only some people are susceptible to this effect, but
intestine, the development of constipation in later life is
in those who are susceptible, gluten has a direct destruc-
much less likely.
tive effect on intestinal enterocytes. In milder forms of
Constipation can also result from spasm of a small
the disease, only the microvilli of the absorbing entero-
segment of the sigmoid colon. It should be recalled that
cytes on the villi are destroyed, thus decreasing the
motility even normally is weak in the large intestine, so
absorptive surface area as much as twofold. In the more
that even a slight degree of spasm is often capable of
severe forms, the villi themselves become blunted or
causing serious constipation. After the constipation has
disappear altogether, thus still further reducing the
continued for several days and excess feces have accu-
absorptive area of the gut. Removal of wheat and rye
mulated above a spastic sigmoid colon, excessive
flour from the diet frequently results in cure within
colonic secretions often then lead to a day or so of diar-
weeks, especially in children with this disease.
rhea. After this, the cycle begins again, with repeated
bouts of alternating constipation and diarrhea.
Tropical Sprue. A different type of sprue called tropical
sprue frequently occurs in the tropics and can often be
Megacolon. Occasionally, constipation is so severe that
treated with antibacterial agents. Even though no spe-
bowel movements occur only once every several days
cific bacterium has been implicated as the cause, it is
or sometimes only once a week. This allows tremendous
believed that this variety of sprue is usually caused by
quantities of fecal matter to accumulate in the colon,
inflammation of the intestinal mucosa resulting from
causing the colon sometimes to distend to a diameter of
unidentified infectious agents.
3 to 4 inches. The condition is called megacolon, or
Hirschsprung’s disease.
Malabsorption in Sprue. In the early stages of sprue, intes-
A frequent cause of megacolon is lack of or deficiency
tinal absorption of fat is more impaired than absorption
of ganglion cells in the myenteric plexus in a segment
of other digestive products. The fat that appears in the
of the sigmoid colon. As a consequence, neither defeca-
stools is almost entirely in the form of salts of fatty acids
tion reflexes nor strong peristaltic motility can occur in
rather than undigested fat, demonstrating that the
this area of the large intestine. The sigmoid itself
problem is one of absorption, not of digestion. In fact,
becomes small and almost spastic while feces accumu-
the condition is frequently called steatorrhea, which
late proximal to this area, causing megacolon in the
means simply excess fats in the stools.
ascending, transverse, and descending colons.
In very severe cases of sprue, in addition to malab-
sorption of fats there is also impaired absorption of pro-
teins, carbohydrates, calcium, vitamin K, folic acid, and
Diarrhea
vitamin B
12. As a result, the person suffers (1) severe
nutritional deficiency, often developing wasting of the
Diarrhea results from rapid movement of fecal matter
body; (2) osteomalacia (demineralization of the bones
through the large intestine. Several causes of diarrhea
because of lack of calcium); (3) inadequate blood coag-
with important physiologic sequelae are the following.
ulation caused by lack of vitamin K; and (4) macrocytic
anemia of the pernicious anemia type, owing to dimin-
Enteritis. Enteritis means inflammation usually caused
ished vitamin B
12
and folic acid absorption.
either by a virus or by bacteria in the intestinal tract. In
usual infectious diarrhea, the infection is most extensive
in the large intestine and the distal end of the ileum.
Disorders of the
Everywhere the infection is present, the mucosa
becomes extensively irritated, and its rate of secretion
Large Intestine
becomes greatly enhanced. In addition, motility of the
Constipation
intestinal wall usually increases manyfold. As a result,
large quantities of fluid are made available for washing
Constipation means slow movement of feces through the
the infectious agent toward the anus, and at the same
large intestine; it is often associated with large quantities
time strong propulsive movements propel this fluid
Chapter 66
Physiology of Gastrointestinal Disorders
823
forward. This is an important mechanism for ridding the
injury. But, because the cord defecation reflex can still
intestinal tract of a debilitating infection.
occur, a small enema to excite action of this cord reflex,
Of special interest is diarrhea caused by cholera (and
usually given in the morning shortly after a meal, can
less often by other bacteria such as some pathogenic
often cause adequate defecation. In this way, people
colon bacilli). As explained in Chapter 65, cholera toxin
with spinal cord injuries that do not destroy the conus
directly stimulates excessive secretion of electrolytes
medullaris of the spinal cord can usually control their
and fluid from the crypts of Lieberkühn in the distal
bowel movements each day.
ileum and colon. The amount can be 10 to 12 liters
per day, although the colon can usually reabsorb a
maximum of only 6 to 8 liters per day. Therefore, loss of
General Disorders of the
fluid and electrolytes can be so debilitating within
Gastrointestinal Tract
several days that death can ensue.
The most important physiologic basis of therapy in
Vomiting
cholera is to replace the fluid and electrolytes as rapidly
as they are lost, mainly by giving the patient intravenous
Vomiting is the means by which the upper gastroin-
solutions. With proper therapy, along with the use of
testinal tract rids itself of its contents when almost any
antibiotics, almost no cholera patients die, but without
part of the upper tract becomes excessively irritated,
therapy, as many as 50 per cent do.
overdistended, or even overexcitable. Excessive disten-
tion or irritation of the duodenum provides an espe-
Psychogenic Diarrhea. Everyone is familiar with the diar-
cially strong stimulus for vomiting.
rhea that accompanies periods of nervous tension, such
The sensory signals that initiate vomiting originate
as during examination time or when a soldier is about
mainly from the pharynx, esophagus, stomach, and
to go into battle. This type of diarrhea, called psy-
upper portions of the small intestines. And the nerve
chogenic emotional diarrhea, is caused by excessive
impulses are transmitted, as shown in Figure 66-2, by
stimulation of the parasympathetic nervous system,
both vagal and sympathetic afferent nerve fibers to mul-
which greatly excites both (1) motility and (2) excess
secretion of mucus in the distal colon. These two effects
added together can cause marked diarrhea.
Apomorphine, morphine
Ulcerative Colitis. Ulcerative colitis is a disease in which
Chemoreceptor
extensive areas of the walls of the large intestine
trigger zone
become inflamed and ulcerated. The motility of the
ulcerated colon is often so great that mass movements
“Vomiting center”
occur much of the day rather than for the usual 10 to
30 minutes. Also, the colon’s secretions are greatly
enhanced. As a result, the patient has repeated diarrheal
bowel movements.
The cause of ulcerative colitis is unknown. Some cli-
nicians believe that it results from an allergic or immune
Vagal
Vagal
destructive effect, but it also could result from chronic
afferents
afferents
bacterial infection not yet understood. Whatever the
cause, there is a strong hereditary tendency for suscep-
tibility to ulcerative colitis. Once the condition has pro-
gressed very far, the ulcers seldom will heal until an
ileostomy is performed to allow the small intestinal con-
tents to drain to the exterior rather than to pass through
the colon. Even then the ulcers sometimes fail to heal,
Sympathetic afferents
and the only solution might be surgical removal of the
entire colon.
Paralysis of Defecation in Spinal
Cord Injuries
From Chapter 63 it will be recalled that defecation is
normally initiated by accumulating feces in the rectum,
which causes a spinal cord-mediated defecation reflex
passing from the rectum to the conus medullaris of the
spinal cord and then back to the descending colon,
sigmoid, rectum, and anus.
When the spinal cord is injured somewhere between
the conus medullaris and the brain, the voluntary
portion of the defecation act is blocked while the basic
Figure 66-2
cord reflex for defecation is still intact. Nevertheless,
loss of the voluntary aid to defecation—that is, loss of
Neutral connections of the “vomiting center.” This so-called vom-
the increased abdominal pressure and relaxation of the
iting center includes multiple sensory, motor, and control nuclei
voluntary anal sphincter—often makes defecation a dif-
mainly in the medullary and pontile reticular formation but also
ficult process in the person with this type of upper cord
extending into the spinal cord.
824
Unit XII Gastrointestinal Physiology
tiple distributed nuclei in the brain stem that all
Also, it is well known that rapidly changing direction
together are called the “vomiting center.” From here,
or rhythm of motion of the body can cause certain
motor impulses that cause the actual vomiting are trans-
people to vomit. The mechanism for this is the follow-
mitted from the vomiting center by way of the 5th, 7th,
ing: The motion stimulates receptors in the vestibular
9th, 10th, and 12th cranial nerves to the upper gastroin-
labyrinth of the inner ear, and from here impulses are
testinal tract, through vagal and sympathetic nerves to
transmitted mainly by way of the brain stem vestibular
the lower tract, and through spinal nerves to the
nuclei into the cerebellum, then to the chemoreceptor
diaphragm and abdominal muscles.
trigger zone, and finally to the vomiting center to cause
vomiting.
Antiperistalsis, the Prelude to Vomiting. In the early stages
of excessive gastrointestinal irritation or overdistention,
antiperistalsis begins to occur often many minutes
Nausea
before vomiting appears. Antiperistalsis means peristal-
Everyone has experienced the sensation of nausea and
sis up the digestive tract rather than downward. This
knows that it is often a prodrome of vomiting. Nausea
may begin as far down in the intestinal tract as the
is the conscious recognition of subconscious excitation
ileum, and the antiperistaltic wave travels backward up
in an area of the medulla closely associated with or part
the intestine at a rate of 2 to 3 cm/sec; this process
of the vomiting center, and it can be caused by (1) irri-
can actually push a large share of the lower small intes-
tative impulses coming from the gastrointestinal tract,
tine contents all the way back to the duodenum
(2) impulses that originate in the lower brain associated
and stomach within 3 to 5 minutes. Then, as these upper
with motion sickness, or (3) impulses from the cerebral
portions of the gastrointestinal tract, especially the duo-
cortex to initiate vomiting. Vomiting occasionally occurs
denum, become overly distended, this distention
without the prodromal sensation of nausea, indicating
becomes the exciting factor that initiates the actual
that only certain portions of the vomiting center are
vomiting act.
associated with the sensation of nausea.
At the onset of vomiting, strong intrinsic contractions
occur in both the duodenum and the stomach, along
with partial relaxation of the esophageal-stomach
Gastrointestinal Obstruction
sphincter, thus allowing vomitus to begin moving from
the stomach into the esophagus. From here, a specific
The gastrointestinal tract can become obstructed at
vomiting act involving the abdominal muscles takes
almost any point along its course, as shown in Figure
over and expels the vomitus to the exterior, as explained
66-3. Some common causes of obstruction are (1)
in the next paragraph.
cancer, (2) fibrotic constriction resulting from ulceration
or from peritoneal adhesions, (3) spasm of a segment of
Vomiting Act. Once the vomiting center has been suffi-
the gut, and (4) paralysis of a segment of the gut.
ciently stimulated and the vomiting act instituted, the
The abnormal consequences of obstruction de-
first effects are (1) a deep breath, (2) raising of the hyoid
pend on the point in the gastrointestinal tract that
bone and larynx to pull the upper esophageal sphincter
becomes obstructed. If the obstruction occurs at the
open, (3) closing of the glottis to prevent vomitus flow
pylorus, which results often from fibrotic constriction
into the lungs, and (4) lifting of the soft palate to close
after peptic ulceration, persistent vomiting of stomach
the posterior nares. Next comes a strong downward con-
contents occurs. This depresses bodily nutrition; it
traction of the diaphragm along with simultaneous con-
also causes excessive loss of hydrogen ions from the
traction of all the abdominal wall muscles. This squeezes
stomach and can result in various degrees of whole-
the stomach between the diaphragm and the abdominal
body alkalosis.
muscles, building the intragastric pressure to a high
If the obstruction is beyond the stomach, antiperi-
level. Finally, the lower esophageal sphincter relaxes
staltic reflux from the small intestine causes intestinal
completely, allowing expulsion of the gastric contents
juices to flow backward into the stomach, and these
upward through the esophagus.
Thus, the vomiting act results from a squeezing action
of the muscles of the abdomen associated with simulta-
Obstruction at pylorus
Causes
neous contraction of the stomach wall and opening of
causes acid vomitus
1. Cancer
the esophageal sphincters so that the gastric contents
2. Ulcer
can be expelled.
Obstruction below
3. Spasm
duodenum causes
4. Paralytic ileus
“Chemoreceptor Trigger Zone” in the Brain Medulla for Initiation
neutral or basic
5. Adhesions
vomitus
of Vomiting by Drugs or by Motion Sickness. Aside from the
vomiting initiated by irritative stimuli in the gastroin-
High obstruction
testinal tract itself, vomiting can also be caused by
causes extreme
nervous signals arising in areas of the brain. This is par-
vomiting
ticularly true for a small area located bilaterally on the
floor of the fourth ventricle called the chemoreceptor
trigger zone for vomiting. Electrical stimulation of this
Low obstruction
area can initiate vomiting; but, more important, admin-
causes extreme
istration of certain drugs, including apomorphine, mor-
constipation with
phine, and some digitalis derivatives, can directly
less vomiting
stimulate this chemoreceptor trigger zone and initiate
vomiting. Destruction of this area blocks this type of
Figure 66-3
vomiting but does not block vomiting resulting from
irritative stimuli in the gastrointestinal tract itself.
Obstruction in different parts of the gastrointestinal tract.
Chapter 66
Physiology of Gastrointestinal Disorders
825
juices are vomited along with the stomach secretions. In
References
this instance, the person loses large amounts of water
and electrolytes, so that he or she becomes severely
Berkes J, Viswanathan VK, Savkovic SD, Hecht G: Intestinal
dehydrated, but the loss of acid from the stomach and
epithelial responses to enteric pathogens: effects on the
base from the small intestine may be approximately
tight junction barrier, ion transport, and inflammation.
equal, so that little change in acid-base balance occurs.
Gut 52:439, 2003.
If the obstruction is near the distal end of the large
Blaser MJ, Atherton JC: Helicobacter pylori persistence:
intestine, feces can accumulate in the colon for a week
biology and disease. J Clin Invest 113:321, 2004.
or more. The patient develops an intense feeling of con-
Dierkes J, Ebert M, Malfertheiner P, Luley C: Helicobacter
stipation, but at first vomiting is not severe. After the
pylori infection, vitamin B12 and homocysteine. A review.
large intestine has become completely filled and it
Dig Dis 21:237, 2003.
finally becomes impossible for additional chyme to
Egan LJ, Sandborn WJ: Advances in the treatment of
move from the small intestine into the large intestine,
Crohn’s disease. Gastroenterology 126:1574, 2004.
severe vomiting does then occur. Prolonged obstruction
Elson CO: Genes, microbes, and T cells—new therapeutic
of the large intestine can finally causes rupture of the
targets in Crohn’s disease. N Engl J Med 346(8):614, 2002.
intestine itself or dehydration and circulatory shock
Itzkowitz SH, Yio X: Inflammation and cancer. IV. Colorec-
resulting from the severe vomiting.
tal cancer in inflammatory bowel disease: the role of
inflammation. Am J Physiol Gastrointest Liver Physiol
287:G7, 2004
Gases in the Gastrointestinal
Johnson LR: Gastrointestinal Physiology, 6th ed. St. Louis:
Tract; “Flatus”
Mosby, 2001.
Kapadia CR: Gastric atrophy, metaplasia, and dysplasia: a
Gases, called flatus, can enter the gastrointestinal tract
clinical perspective. J Clin Gastroenterol 36(5 Suppl):S29,
from three sources: (1) swallowed air, (2) gases formed
2003
in the gut as a result of bacterial action, or (3) gases that
Kunzelmann K, Mall M: Electrolyte transport in the mam-
diffuse from the blood into the gastrointestinal tract.
malian colon: mechanisms and implications for disease.
Most gases in the stomach are mixtures of nitrogen and
Physiol Rev 82:245, 2002.
oxygen derived from swallowed air. In the typical
Laroux FS, Pavlick KP, Wolf RE, Grisham MB: Dysregula-
person these gases are expelled by belching. Only small
tion of intestinal mucosal immunity: implications in
amounts of gas normally occur in the small intestine,
inflammatory bowel disease. News Physiol Sci 16:272,
and much of this gas is air that passes from the stomach
2001.
into the intestinal tract.
Larson DW, Pemberton JH: Current concepts and contro-
In the large intestine, most of the gases are derived
versies in surgery for IBD. Gastroenterology 126:1611,
from bacterial action, including especially carbon
2004.
dioxide, methane, and hydrogen. When methane and
Ming SC, Goldman H: Pathology of the Gastrointestinal
hydrogen become suitably mixed with oxygen, an actual
Tract. Baltimore: Williams and Wilkins, 1998.
explosive mixture is sometimes formed. Use of the elec-
Podolsky DK: Inflammatory bowel disease. N Engl J Med
tric cautery during sigmoidoscopy has been known to
347:417, 2002.
cause a mild explosion.
Puri P, Shinkai T: Pathogenesis of Hirschsprung’s disease and
Certain foods are known to cause greater expulsion
its variants: recent progress. Semin Pediatr Surg 13:18,
of flatus through the anus than others—beans, cabbage,
2004.
onion, cauliflower, corn, and certain irritant foods
Spirt MJ: Stress-related mucosal disease: risk factors and
such as vinegar. Some of these foods serve as a
prophylactic therapy. Clin Ther 26:197, 2004.
suitable medium for gas-forming bacteria, especially
Suerbaum S, Michetti P: Helicobacter pylori infection.
unabsorbed fermentable types of carbohydrates. For
N Engl J Med 347(15):1175, 2002.
instance, beans contain an indigestible carbohydrate
Wolfe MM, Lichtenstein DR, Singh G: Gastrointestinal tox-
that passes into the colon and becomes a superior food
icity of nonsteroidal antiinflammatory drugs. N Engl J
for colonic bacteria. But in other instances, excess expul-
Med 340(24):1888, 1999.
sion of gas results from irritation of the large intestine,
Wood JD: Neuropathophysiology of irritable bowel syn-
which promotes rapid peristaltic expulsion of gases
drome. J Clin Gastroenterol 35(1 Suppl):S11, 2002.
through the anus before they can be absorbed.
Wright EM, Martin MG, Turk E: Intestinal absorption in
The amount of gases entering or forming in the large
health and disease—sugars. Best Pract Res Clin Gas-
intestine each day averages 7 to 10 liters, whereas the
troenterol 17:943, 2003.
average amount expelled through the anus is usually
only about 0.6 liter. The remainder is normally absorbed
into the blood through the intestinal mucosa and
expelled through the lungs.
U N I T XIII
Metabolism and
Temperature
Regulation
67. Metabolism of Carbohydrates, and Formation
of Adenosine Triphosphate
68. Lipid Metabolism
69. Protein Metabolism
70. The Liver as an Organ
71. Dietary Balances; Regulation of Feeding;
Obesity and Starvation; Vitamins and Minerals
72. Energetics and Metabolic Rate
73. Body Temperature, Temperature Regulation,
and Fever
C H A P T E R
6
7
Metabolism of Carbohydrates,
and Formation of Adenosine
Triphosphate
The next few chapters deal with metabolism in the
body, which means the chemical processes that make it
possible for the cells to continue living. It is not the
purpose of this textbook to present the chemical details
of all the various cellular reactions, because this lies in
the discipline of biochemistry. Instead, these chapters
are devoted to (1) a review of the principal chemical
processes of the cell and (2) an analysis of their physio-
logic implications, especially the manner in which they
fit into the overall concept of homeostasis.
Release of Energy from Foods, and the Concept of
“Free Energy”
A great proportion of the chemical reactions in the cells is concerned with making
the energy in foods available to the various physiologic systems of the cell. For
instance, energy is required for muscle activity, secretion by the glands, maintenance
of membrane potentials by the nerve and muscle fibers, synthesis of substances
in the cells, absorption of foods from the gastrointestinal tract, and many other
functions.
Coupled Reactions. All the energy foods—carbohydrates, fats, and proteins—can be
oxidized in the cells, and during this process, large amounts of energy are released.
These same foods can also be burned with pure oxygen outside the body in an actual
fire, also releasing large amounts of energy; in this case, however, the energy is
released suddenly, all in the form of heat. The energy needed by the physiologic
processes of the cells is not heat but energy to cause mechanical movement in the
case of muscle function, to concentrate solutes in the case of glandular secretion,
and to effect other functions. To provide this energy, the chemical reactions must be
“coupled” with the systems responsible for these physiologic functions. This cou-
pling is accomplished by special cellular enzyme and energy transfer systems, some
of which are explained in this and subsequent chapters.
“Free Energy.” The amount of energy liberated by complete oxidation of a food is
called the free energy of oxidation of the food, and this is generally represented by
the symbol DG. Free energy is usually expressed in terms of calories per mole of
substance. For instance, the amount of free energy liberated by complete oxidation
of 1 mole (180 grams) of glucose is 686,000 calories.
Role of Adenosine Triphosphate in Metabolism
Adenosine triphosphate
(ATP) is an essential link between energy-utilizing
and energy-producing functions of the body (Figure 67-1). For this reason, ATP
has been called the energy currency of the body, and it can be gained and spent
repeatedly.
Energy derived from the oxidation of carbohydrates, proteins, and fats is used to
convert adenosine diphosphate (ADP) to ATP, which is then consumed by the
various reactions of the body that are necessary for (1) active transport of mole-
cules across cell membranes; (2) contraction of muscles and performance of
mechanical work; (3) various synthetic reactions that create hormones, cell mem-
branes, and many other essential molecules of the body; (4) conduction of nerve
impulses; (5) cell division and growth; and (6) many other physiologic functions that
are necessary to maintain and propagate life.
829
830
Unit XIII
Metabolism and Temperature Regulation
ATP is a labile chemical compound that is present in
The interconversions among ATP, ADP, and AMP are
all cells. It has the chemical structure shown in Figure
the following:
67-2. From this formula, it can be seen that ATP is a
combination of adenine, ribose, and three phosphate
-12,000 cal
ADP
-12,000 cal
ADP
radicals. The last two phosphate radicals are connected
ATP
+
+
with the remainder of the molecule by high-energy
bonds, which are indicated by the symbol ~.
+12,000 cal
PO3
+12,000 cal
2PO3
The amount of free energy in each of these high-
energy bonds per mole of ATP is about 7300 calories
ATP is present everywhere in the cytoplasm and
under standard conditions and about 12,000 calories
nucleoplasm of all cells, and essentially all the physio-
under the usual conditions of temperature and concen-
logic mechanisms that require energy for operation
trations of the reactants in the body. Therefore, in the
obtain it directly from ATP (or another similar high-
body, removal of each of the last two phosphate radicals
energy compound—guanosine triphosphate [GTP]). In
liberates about 12,000 calories of energy. After loss of
turn, the food in the cells is gradually oxidized, and the
one phosphate radical from ATP, the compound
released energy is used to form new ATP, thus always
becomes ADP, and after loss of the second phosphate
maintaining a supply of this substance. All these energy
radical, it becomes adenosine monophosphate (AMP).
transfers take place by means of coupled reactions.
The principal purpose of this chapter is to explain
how the energy from carbohydrates can be used to form
ATP in the cells. Normally, 90 per cent or more of all
the carbohydrates utilized by the body are used for this
purpose.
Energy production
•Proteins
Central Role of Glucose in
•Carbohydrates
Oxidation
•Fats
Carbohydrate Metabolism
As explained in Chapter 65, the final products of car-
bohydrate digestion in the alimentary tract are almost
entirely glucose, fructose, and galactose—with glucose
ADP + Pi
ATP
representing, on average, about 80 per cent of these.
After absorption from the intestinal tract, much of the
fructose and almost all the galactose are rapidly con-
verted into glucose in the liver. Therefore, little fructose
Energy utilization
and galactose are present in the circulating blood.
•Active ion transport
•Muscle contraction
Glucose thus becomes the final common pathway for the
•Synthesis of molecules
transport of almost all carbohydrates to the tissue cells.
•Cell division and growth
In liver cells, appropriate enzymes are available to
promote interconversions among the monosaccha-
rides—glucose, fructose, and galactose—as shown in
Figure 67-3. Furthermore, the dynamics of the reactions
are such that when the liver releases the monosaccha-
Figure 67-1
rides back into the blood, the final product is almost
Adenosine triphosphate (ATP) as the central link between energy-
entirely glucose. The reason for this is that the liver cells
producing and energy-utilizing systems of the body. ADP, adeno-
contain large amounts of glucose phosphatase. There-
sine diphosphate; Pi, inorganic phosphate.
fore, glucose-6-phosphate can be degraded to glucose
NH2
N
C
Adenine
C
N
HC
Triphosphate
C
CH
O
O
O
N
N
O
2
CH
O P
O ~
P
O
~ P
O-
C
C
O-
O-
O-
H
H
Ribose
H
H
C C
Figure 67-2
OH OH
Chemical structure of adenosine
triphosphate (ATP).
Chapter 67
Metabolism of Carbohydrates, and Formation of Adenosine Triphosphate
831
The transport of glucose through the membranes of
most tissue cells is quite different from that which
Cell membrane
occurs through the gastrointestinal membrane or
through the epithelium of the renal tubules. In both
these cases, the glucose is transported by the mechanism
ATP
Galactose
Galactose-1-phosphate
of active sodium-glucose co-transport, in which active
transport of sodium provides energy for absorbing
glucose against a concentration difference. This sodium
co-transport mechanism functions only in certain
Uridine diphosphate galactose
special epithelial cells that are specifically adapted for
active absorption of glucose. At other cell membranes,
glucose is transported only from higher concentration
Uridine diphosphate glucose
toward lower concentration by facilitated diffusion,
made possible by the special binding properties of mem-
Glycogen
brane glucose carrier protein. The details of facilitated
diffusion for cell membrane transport are presented in
Glucose-1-phosphate
Chapter 4.
ATP
Insulin Increases Facilitated
Glucose
Glucose-6-phosphate
Diffusion of Glucose
The rate of glucose transport as well as transport of
some other monosaccharides is greatly increased by
insulin. When large amounts of insulin are secreted by
ATP
Fructose
Fructose-6-phosphate
the pancreas, the rate of glucose transport into most
cells increases to 10 or more times the rate of transport
when no insulin is secreted. Conversely, the amounts of
Glycolysis
glucose that can diffuse to the insides of most cells of
the body in the absence of insulin, with the exception
of liver and brain cells, are far too little to supply the
amount of glucose normally required for energy metab-
Figure 67-3
olism.
In effect, the rate of carbohydrate utilization by most
Interconversions of the three major monosaccharides—glucose,
cells is controlled by the rate of insulin secretion from
fructose, and galactose—in liver cells.
the pancreas. The functions of insulin and its control of
carbohydrate metabolism are discussed in detail in
Chapter 78.
and phosphate, and the glucose can then be transported
through the liver cell membrane back into the blood.
Once again, it should be emphasized that usually
Phosphorylation of Glucose
more than 95 per cent of all the monosaccharides that
circulate in the blood are the final conversion product,
Immediately on entry into the cells, glucose combines
glucose.
with a phosphate radical in accordance with the follow-
ing reaction:
Transport of Glucose
glucokinase or hexokinase
Glucoseæ
ææææææææ ÆGlucose-6-phosphate
Through the Cell Membrane
+
ATP
Before glucose can be used by the body’s tissue cells, it
This phosphorylation is promoted mainly by the
must be transported through the tissue cell membrane
enzyme glucokinase in the liver and by hexokinase in
into the cellular cytoplasm. However, glucose cannot
most other cells. The phosphorylation of glucose is
easily diffuse through the pores of the cell membrane
almost completely irreversible except in the liver cells,
because the maximum molecular weight of particles
the renal tubular epithelial cells, and the intestinal
that can diffuse readily is about 100, and glucose has a
epithelial cells; in these cells, another enzyme, glucose
molecular weight of 180. Yet glucose does pass to the
phosphatase, is also available, and when this is activated,
interior of the cells with a reasonable degree of freedom
it can reverse the reaction. In most tissues of the body,
by the mechanism of facilitated diffusion. The principles
phosphorylation serves to capture the glucose in the
of this type of transport are discussed in Chapter 4. Basi-
cell. That is, because of its almost instantaneous binding
cally, they are the following. Penetrating through the
with phosphate, the glucose will not diffuse back out,
lipid matrix of the cell membrane are large numbers of
except from those special cells, especially liver cells, that
protein carrier molecules that can bind with glucose. In
have phosphatase.
this bound form, the glucose can be transported by the
carrier from one side of the membrane to the other side
and then released. Therefore, if the concentration of
Glycogen Is Stored in Liver
glucose is greater on one side of the membrane than on
and Muscle
the other side, more glucose will be transported from
the high-concentration area to the low-concentration
After absorption into a cell, glucose can be used imme-
area than in the opposite direction.
diately for release of energy to the cell, or it can be
832
Unit XIII
Metabolism and Temperature Regulation
stored in the form of glycogen, which is a large polymer
Removal of Stored Glycogen—
of glucose.
Glycogenolysis
All cells of the body are capable of storing at least
some glycogen, but certain cells can store large amounts,
Glycogenolysis means the breakdown of the cell’s
especially liver cells, which can store up to 5 to 8 per
stored glycogen to re-form glucose in the cells. The
cent of their weight as glycogen, and muscle cells, which
glucose can then be used to provide energy. Glycogenol-
can store up to 1 to 3 per cent glycogen. The glycogen
ysis does not occur by reversal of the same chemical
molecules can be polymerized to almost any molecular
reactions that form glycogen; instead, each succeeding
weight, with the average molecular weight being
5
glucose molecule on each branch of the glycogen
million or greater; most of the glycogen precipitates in
polymer is split away by phosphorylation, catalyzed by
the form of solid granules.
the enzyme phosphorylase.
This conversion of the monosaccharides into a high-
Under resting conditions, the phosphorylase is in an
molecular-weight precipitated compound
(glycogen)
inactive form, so that glycogen will remain stored. When
makes it possible to store large quantities of carbohy-
it is necessary to re-form glucose from glycogen, the
drates without significantly altering the osmotic pres-
phosphorylase must first be activated. This can be
sure of the intracellular fluids. High concentrations of
accomplished in several ways, including the following
low-molecular-weight soluble monosaccharides would
two.
play havoc with the osmotic relations between intracel-
lular and extracellular fluids.
Activation of Phosphorylase by Epinephrine or by Glucagon. Two
hormones, epinephrine and glucagon, can activate phos-
Glycogenesis—The Process
phorylase and thereby cause rapid glycogenolysis. The
of Glycogen Formation
initial effect of each of these hormones is to promote
the formation of cyclic AMP in the cells, which then
The chemical reactions for glycogenesis are shown in
initiates a cascade of chemical reactions that activates
Figure 67-4. From this figure, it can be seen that glucose-
the phosphorylase. This is discussed in detail in Chapter
6-phosphate can become glucose-1-phosphate; this is
78.
converted to uridine diphosphate glucose, which is
Epinephrine is released by the adrenal medullae
finally converted into glycogen. Several specific
when the sympathetic nervous system is stimulated.
enzymes are required to cause these conversions, and
Therefore, one of the functions of the sympathetic
any monosaccharide that can be converted into glucose
nervous system is to increase the availability of glucose
can enter into the reactions. Certain smaller com-
for rapid energy metabolism. This function of epineph-
pounds, including lactic acid, glycerol, pyruvic acid, and
rine occurs markedly in both liver cells and muscle,
some deaminated amino acids, can also be converted
thereby contributing, along with other effects of sym-
into glucose or closely allied compounds and then con-
pathetic stimulation, to preparing the body for action,
verted into glycogen.
as discussed fully in Chapter 60.
Glucagon is a hormone secreted by the alpha cells
of the pancreas when the blood glucose concentration
falls too low. It stimulates formation of cyclic AMP
mainly in the liver cells, and this in turn promotes
conversion of liver glycogen into glucose and its
release into the blood, thereby elevating the blood
Cell membrane
glucose concentration. The function of glucagon in
blood glucose regulation is discussed more fully in
Chapter 78.
Glycogen
Uridine diphosphate glucose
(phosphorylase)
Release of Energy from the
Glucose Molecule by the
Glucose-1-phosphate
Glycolytic Pathway
Because complete oxidation of 1 gram-molecule of
Glucose
(glucokinase)
glucose releases
686,000 calories of energy and
Blood
Glucose-6-phosphate
only 12,000 calories of energy are required to form 1
glucose
(phosphatase)
gram-molecule of ATP, energy would be wasted if
glucose were decomposed all at once into water and
carbon dioxide while forming only a single ATP mole-
Glycolysis
cule. Fortunately, all cells of the body contain special
protein enzymes that cause the glucose molecule to split
a little at a time in many successive steps, so that its
energy is released in small packets to form one mole-
cule of ATP at a time, forming a total of 38 moles of
Figure 67-4
ATP for each mole of glucose metabolized by the cells.
The next sections describe the basic principles of
Chemical reactions of glycogenesis and glycogenolysis, showing
the processes by which the glucose molecule is pro-
also interconversions between blood glucose and liver glycogen.
(The phosphatase required for the release of glucose from the cell
gressively dissected and its energy released to form
is present in liver cells but not in most other cells.)
ATP.
Chapter 67
Metabolism of Carbohydrates, and Formation of Adenosine Triphosphate
833
Glycolysis and the Formation
ATP molecules by the entire glycolytic process is only 2
of Pyruvic Acid
moles for each mole of glucose utilized. This amounts to
24,000 calories of energy that becomes transferred to
By far the most important means of releasing energy
ATP, but during glycolysis, a total of 56,000 calories of
from the glucose molecule is initiated by glycolysis. The
energy were lost from the original glucose, giving an
end products of glycolysis are then oxidized to provide
overall efficiency for ATP formation of only 43 per cent.
energy. Glycolysis means splitting of the glucose mole-
The remaining 57 per cent of the energy is lost in the
cule to form two molecules of pyruvic acid.
form of heat.
Glycolysis occurs by 10 successive chemical reactions,
shown in Figure 67-5. Each step is catalyzed by at least
one specific protein enzyme. Note that glucose is first
Conversion of Pyruvic Acid
converted into fructose-1,6-diphosphate and then split
to Acetyl Coenzyme A
into two three-carbon-atom molecules, glyceraldehyde-
3-phosphate, each of which is then converted through
The next stage in the degradation of glucose is a two-
five additional steps into pyruvic acid.
step conversion of the two pyruvic acid molecules from
Figure 67-5 into two molecules of acetyl coenzyme
Formation of ATP During Glycolysis. Despite the many chem-
A (acetyl-CoA), in accordance with the following
ical reactions in the glycolytic series, only a small
reaction:
portion of the free energy in the glucose molecule is
released at most steps. However, between the
1,3-
O
diphosphoglyceric acid and the 3-phosphoglyceric acid
stages, and again between the phosphoenolpyruvic acid
and the pyruvic acid stages, the packets of energy
2CH3
C COOH
+
2CoA SH
released are greater than 12,000 calories per mole, the
(Pyruvic acid)
(Coenzyme A)
amount required to form ATP, and the reactions are
coupled in such a way that ATP is formed. Thus, a total
O
of 4 moles of ATP were formed for each mole of fruc-
tose-1,6-diphosphate that is split into pyruvic acid.
Yet 2 moles of ATP were required to phosphorylate
2CH3
C S CoA + 2CO2 + 4H
the original glucose to form fructose-1,6-diphosphate
(Acetyl-CoA)
before glycolysis could begin. Therefore, the net gain in
From this reaction, it can be seen that two carbon
dioxide molecules and four hydrogen atoms are
released, while the remaining portions of the two
Glucose
pyruvic acid molecules combine with coenzyme A, a
ATP
ADP
derivative of the vitamin pantothenic acid, to form two
Glucose-6-phosphate
molecules of acetyl-CoA. In this conversion, no ATP is
formed, but up to six molecules of ATP are formed
when the four released hydrogen atoms are later oxi-
Fructose-6-phosphate
dized, as discussed later.
ATP
ADP
Fructose-1,6-diphosphate
Citric Acid Cycle (Krebs Cycle)
Dihydroxyacetone phosphate
The next stage in the degradation of the glucose mole-
cule is called the citric acid cycle (also called the tricar-
2 (Glyceraldehyde-3-phosphate)
boxylic acid cycle or Krebs cycle). This is a sequence of
chemical reactions in which the acetyl portion of acetyl-
4H
CoA is degraded to carbon dioxide and hydrogen
2 (1,3-Diphosphoglyceric acid)
atoms. These reactions all occur in the matrix of the
2ADP
+2ATP
mitochondrion. The released hydrogen atoms add to
the number of these atoms that will subsequently be
2 (3-Phosphoglyceric acid)
oxidized
(as discussed later), releasing tremendous
amounts of energy to form ATP.
2 (2-Phosphoglyceric acid)
Figure 67-6 shows the different stages of the chemi-
cal reactions in the citric acid cycle. The substances to
the left are added during the chemical reactions, and the
2 (Phosphoenolpyruvic acid)
products of the chemical reactions are shown to the
2ADP
2ATP
right. Note at the top of the column that the cycle begins
2 (Pyruvic acid)
with oxaloacetic acid, and at the bottom of the chain of
reactions, oxaloacetic acid is formed again. Thus, the
Net reaction per molecule of glucose:
Glucose + 2ADP + 2PO4-
2 Pyruvic acid + 2ATP + 4H
cycle can continue over and over.
In the initial stage of the citric acid cycle, acetyl-CoA
combines with oxaloacetic acid to form citric acid. The
coenzyme A portion of the acetyl-CoA is released and
Figure 67-5
can be used again and again for the formation of still
Sequence of chemical reactions responsible for glycolysis.
more quantities of acetyl-CoA from pyruvic acid. The
834
Unit XIII
Metabolism and Temperature Regulation
acetyl portion, however, becomes an integral part of the
citric acid molecule. During the successive stages of the
CH3
CO CoA
O
C
COOH
citric acid cycle, several molecules of water are added,
(Acetyl coenzyme A)
as shown on the left in the figure, and carbon dioxide
H2C COOH
and hydrogen atoms are released at other stages in the
(Oxaloacetic acid)
cycle, as shown on the right in the figure.
H2O
CoA
The net results of the entire citric acid cycle are given
H2C COOH
in the explanation at the bottom of Figure 67-6, demon-
HOC COOH
strating that for each molecule of glucose originally
metabolized, 2 acetyl-CoA molecules enter into the
H2C COOH
citric acid cycle, along with 6 molecules of water. These
(Citric acid)
are then degraded into 4 carbon dioxide molecules, 16
H2O
hydrogen atoms, and 2 molecules of coenzyme A. Two
H2C COOH
molecules of ATP are formed, as follows.
C
COOH
Formation of ATP in the Citric Acid Cycle. Not a great amount
of energy is released during the citric acid cycle itself; in
HC COOH
(cis-Aconitic acid)
only one of the chemical reactions—during the change
H
from a-ketoglutaric acid to succinic acid—is a molecule
2O
of ATP formed. Thus, for each molecule of glucose
H2C COOH
metabolized, two acetyl-CoA molecules pass through
HC COOH
the citric acid cycle, each forming a molecule of ATP, or
a total of two molecules of ATP formed.
HOC COOH
Function of Dehydrogenases and Nicotinamide Adenine Dinu-
H
(Isocitric acid)
cleotide in Causing Release of Hydrogen Atoms in the Citric Acid
2H
Cycle. As already noted at several points in this discus-
H2C COOH
sion, hydrogen atoms are released during different
chemical reactions of the citric acid cycle—4 hydrogen
HC COOH
atoms during glycolysis, 4 during formation of acetyl-
CoA from pyruvic acid, and 16 in the citric acid cycle;
O
C
COOH
this makes a total of 24 hydrogen atoms released for each
(Oxalosuccinic acid)
original molecule of glucose. However, the hydrogen
CO
2
atoms are not simply turned loose in the intracellular
H2C COOH
fluid. Instead, they are released in packets of two, and
H2C
in each instance, the release is catalyzed by a specific
protein enzyme called a dehydrogenase. Twenty of the
O
C
COOH
24 hydrogen atoms immediately combine with nicoti-
(a-Ketoglutaric acid)
namide adenine dinucleotide (NAD+), a derivative of
H2O
CO2
the vitamin niacin, in accordance with the following
ADP
2H
reaction:
H2C COOH
ATP
C COOH
H2
H
(Succinic acid)
2H
Substrate
+ NAD+ dehydrogenase
HC COOH
H
HOOC CH
(Fumaric acid)
NADH + H+ + Substrate
H
2O
H
This reaction will not occur without intermediation of
HO
C
COOH
the specific dehydrogenase or without the availability of
NAD+ to act as a hydrogen carrier. Both the free hydro-
H2C COOH
gen ion and the hydrogen bound with NAD+ subse-
(Malic acid)
quently enter into multiple oxidative chemical reactions
2H
that form tremendous quantities of ATP, as discussed
O
C
COOH
later.
The remaining four hydrogen atoms released during
C COOH
H2
the breakdown of glucose—the four released during the
(Oxaloacetic acid)
citric acid cycle between the succinic and fumaric acid
Net reaction per molecule of glucose:
stages—combine with a specific dehydrogenase but are
O + 2ADP
2 Acetyl-CoA + 6H2
not subsequently released to NAD+. Instead, they pass
4CO2 + 16H + 2CoA + 2ATP
directly from the dehydrogenase into the oxidative
process.
Figure 67-6
Function of Decarboxylases in Causing Release of Carbon Dioxide.
Referring again to the chemical reactions of the
Chemical reactions of the citric acid cycle, showing the release of
citric acid cycle, as well as to those for the formation of
carbon dioxide and a number of hydrogen atoms during the cycle.
Chapter 67
Metabolism of Carbohydrates, and Formation of Adenosine Triphosphate
835
acetyl-CoA from pyruvic acid, we find that there are
this sequence of oxidative reactions, tremendous
three stages in which carbon dioxide is released. To
quantities of energy are released to form ATP. Forma-
cause the release of carbon dioxide, other specific
tion of ATP in this manner is called oxidative phospho-
protein enzymes, called decarboxylases, split the carbon
rylation. This occurs entirely in the mitochondria by a
dioxide away from the substrate. The carbon dioxide is
highly specialized process called the chemiosmotic
then dissolved in the body fluids and transported to the
mechanism.
lungs, where it is expired from the body (see Chapter
40).
Chemiosmotic Mechanism of the
Mitochondria to Form ATP
Formation of Large Quantities
Ionization of Hydrogen, the Electron Transport Chain, and Forma-
of ATP by Oxidation of Hydrogen
tion of Water. The first step in oxidative phosphorylation
(the Process of Oxidative
in the mitochondria is to ionize the hydrogen atoms that
Phosphorylation)
have been removed from the food substrates. As
described earlier, these hydrogen atoms are removed in
Despite all the complexities of (1) glycolysis, (2) the
pairs: one immediately becomes a hydrogen ion, H+; the
citric acid cycle, (3) dehydrogenation, and (4) decar-
other combines with NAD+ to form NADH. The upper
boxylation, pitifully small amounts of ATP are formed
portion of Figure 67-7 shows the subsequent fate of the
during all these processes—only two ATP molecules in
NADH and H+. The initial effect is to release the other
the glycolysis scheme and another two in the citric acid
hydrogen atom from the NADH to form another hydro-
cycle for each molecule of glucose metabolized. Instead,
gen ion, H+; this process also reconstitutes NAD+ that
almost 90 per cent of the total ATP created through
will be reused again and again.
glucose metabolism is formed during subsequent oxi-
The electrons that are removed from the hydrogen
dation of the hydrogen atoms that were released at
atoms to cause the hydrogen ionization immediately
early stages of glucose degradation. Indeed, the princi-
enter an electron transport chain of electron acceptors
pal function of all these earlier stages is to make the
that are an integral part of the inner membrane (the
hydrogen of the glucose molecule available in forms
shelf membrane) of the mitochondrion. The electron
that can be oxidized.
acceptors can be reversibly reduced or oxidized by
Oxidation of hydrogen is accomplished, as illustrated
accepting or giving up electrons. The important
in Figure 67-7, by a series of enzymatically catalyzed
members of this electron transport chain include flavo-
reactions in the mitochondria. These reactions (1) split
protein, several iron sulfide proteins, ubiquinone, and
each hydrogen atom into a hydrogen ion and an elec-
cytochromes B, C1, C, A, and A3. Each electron is shut-
tron and (2) use the electrons eventually to combine dis-
tled from one of these acceptors to the next until it
solved oxygen of the fluids with water molecules to form
finally reaches cytochrome A3, which is called
hydroxyl ions. Then the hydrogen and hydroxyl ions
cytochrome oxidase because it is capable of giving up
combine with each other to form water. During
two electrons and thus reducing elemental oxygen to
form ionic oxygen, which then combines with hydrogen
Food substrate
ions to form water.
Thus, Figure 67-7 shows the transport of electrons
through the electron chain and then their ultimate
NADH +
H+
use by cytochrome oxidase to cause the formation
of water molecules. During the transport of these elec-
H+
FMN
-2e
trons through the electron transport chain, energy is
2H+
FeS
NAD+
released that is used to cause the synthesis of ATP, as
+
Q
follows.
2H
b
+
6H
FeS
2H+
C1
H2O
1
Pumping of Hydrogen Ions into the Outer Chamber of the Mito-
C1
chondrion, Caused by the Electron Transport Chain. As the
a
electrons pass through the electron transport chain,
a
3
large amounts of energy are released. This energy is
2e + 1/2 O2
2
used to pump hydrogen ions from the inner matrix of
6H+
the mitochondrion (to the right in Figure 67-7) into the
ATP
outer chamber between the inner and outer mitochon-
ATPase
drial membranes (to the left). This creates a high con-
centration of positively charged hydrogen ions in this
3 ADP
ADP
Facilitated
chamber; it also creates a strong negative electrical
Diffusion
diffusion
potential in the inner matrix.
3 ATP
Outer
Inner
Formation of ATP. The next step in oxidative phosphory-
membrane
membrane
lation is to convert ADP into ATP. This occurs in con-
junction with a large protein molecule that protrudes all
Figure 67-7
the way through the inner mitochondrial membrane
Mitochondrial chemiosmotic mechanism of oxidative phosphory-
and projects with a knoblike head into the inner mito-
lation for forming large quantities of ATP. This figure shows the
chondrial matrix. This molecule is an ATPase, the phys-
relationship of the oxidative and phosphorylation steps at the outer
ical nature of which is shown in Figure 67-7. It is called
and inner membranes of the mitochondrion.
ATP synthetase.
836
Unit XIII
Metabolism and Temperature Regulation
The high concentration of positively charged hydro-
Control of Energy Release from
gen ions in the outer chamber and the large electrical
Stored Glycogen When the Body
potential difference across the inner membrane cause
Needs Additional Energy: Effect
the hydrogen ions to flow into the inner mitochondrial
of ATP and ADP Cell Concentrations
matrix through the substance of the ATPase molecule. In
doing so, energy derived from this hydrogen ion flow is
in Controlling the Rate of Glycolysis
used by ATPase to convert ADP into ATP by combin-
Continual release of energy from glucose when energy
ing ADP with a free ionic phosphate radical (Pi), thus
is not needed by the cells would be an extremely waste-
adding another high-energy phosphate bond to the
ful process. Instead, glycolysis and the subsequent oxi-
molecule.
dation of hydrogen atoms are continually controlled in
The final step in the process is transfer of ATP from
accordance with the cells’ need for ATP. This control is
the inside of the mitochondrion back to the cell cyto-
accomplished by multiple feedback control mechanisms
plasm. This occurs by facilitated diffusion outward
within the chemical schemata. Among the more impor-
through the inner membrane and then by simple dif-
tant of these are the effects of cell concentrations of
fusion through the permeable outer mitochondrial
both ADP and ATP in controlling the rates of chemical
membrane. In turn, ADP is continually transferred in
reactions in the energy metabolism sequence.
the other direction for continual conversion into ATP.
One important way in which ATP helps control
For each two electrons that pass through the entire elec-
energy metabolism is to inhibit the enzyme phospho-
tron transport chain (representing the ionization of
fructokinase. Because this enzyme promotes the forma-
two hydrogen atoms), up to three ATP molecules are
tion of fructose-1,6-diphosphate, one of the initial steps
synthesized.
in the glycolytic series of reactions, the net effect of
excess cellular ATP is to slow or even stop glycolysis,
which in turn stops most carbohydrate metabolism.
Summary of ATP Formation During
Conversely, ADP (and AMP as well) causes the oppo-
the Breakdown of Glucose
site change in this enzyme, greatly increasing its activ-
ity. Whenever ATP is used by the tissues for energizing
We can now determine the total number of ATP mole-
a major fraction of almost all intracellular chemical
cules that, under optimal conditions, can be formed by
reactions, this reduces the ATP inhibition of the enzyme
the energy from one molecule of glucose.
phosphofructokinase and at the same time increases its
1. During glycolysis, four molecules of ATP are
activity as a result of the excess ADP formed. Thus, the
formed, and two are expended to cause the initial
glycolytic process is set in motion, and the total cellular
phosphorylation of glucose to get the process
store of ATP is replenished.
going. This gives a net gain of two molecules of
Another control linkage is the citrate ion formed in
ATP.
the citric acid cycle. An excess of this ion also strongly
2. During each revolution of the citric acid cycle,
inhibits phosphofructokinase, thus preventing the
one molecule of ATP is formed. However, because
glycolytic process from getting ahead of the citric
each glucose molecule splits into two pyruvic acid
acid cycle’s ability to use the pyruvic acid formed during
molecules, there are two revolutions of the cycle
glycolysis.
for each molecule of glucose metabolized, giving
A third way by which the ATP-ADP-AMP system
a net production of two more molecules of
controls carbohydrate metabolism, as well as control-
ATP.
ling energy release from fats and proteins, is the fol-
3. During the entire schema of glucose breakdown, a
lowing: Referring back to the various chemical reactions
total of 24 hydrogen atoms are released during
for energy release, we see that if all the ADP in the
glycolysis and during the citric acid cycle. Twenty of
cell has already been converted into ATP, additional
these atoms are oxidized in conjunction with the
ATP simply cannot be formed. As a result, the entire
chemiosmotic mechanism shown in Figure 67-7,
sequence involved in the use of foodstuffs—glucose,
with the release of three ATP molecules per two
fats, and proteins—to form ATP is stopped. Then, when
atoms of hydrogen metabolized. This gives an
ATP is used by the cell to energize the different phy-
additional 30 ATP molecules.
siologic functions in the cell, the newly formed ADP and
4. The remaining four hydrogen atoms are released
AMP turn on the energy processes again, and ADP and
by their dehydrogenase into the chemiosmotic
AMP are almost instantly returned to the ATP state. In
oxidative schema in the mitochondrion beyond the
this way, essentially a full store of ATP is automatically
first stage of Figure 67-7. Two ATP molecules are
maintained, except during extreme cellular activity, such
usually released for every two hydrogen atoms
as very strenuous exercise.
oxidized, thus giving a total of four more ATP
molecules.
Now, adding all the ATP molecules formed, we
find a maximum of 38 ATP molecules formed for each
Anaerobic Release of Energy—
molecule of glucose degraded to carbon dioxide
“Anaerobic Glycolysis”
and water. Thus, 456,000 calories of energy can be stored
in the form of ATP, whereas
686,000 calories are
Occasionally, oxygen becomes either unavailable or
released during the complete oxidation of each gram-
insufficient, so that oxidative phosphorylation cannot
molecule of glucose. This represents an overall
take place. Yet even under these conditions, a small
maximum efficiency of energy transfer of 66 per cent.
amount of energy can still be released to the cells by the
The remaining 34 per cent of the energy becomes heat
glycolysis stage of carbohydrate degradation, because
and, therefore, cannot be used by the cells to perform
the chemical reactions for the breakdown of glucose to
specific functions.
pyruvic acid do not require oxygen.
Chapter 67
Metabolism of Carbohydrates, and Formation of Adenosine Triphosphate
837
This process is extremely wasteful of glucose, because
occurs in the liver, but a small amount can also occur in
only 24,000 calories of energy are used to form ATP for
other tissues.
each molecule of glucose metabolized, which represents
only a little over 3 per cent of the total energy in the
Use of Lactic Acid by the Heart for Energy. Heart muscle
glucose molecule. Nevertheless, this release of glycolytic
is especially capable of converting lactic acid to pyruvic
energy to the cells, which is called anaerobic energy, can
acid and then using the pyruvic acid for energy. This
be a lifesaving measure for up to a few minutes when
occurs to a great extent during heavy exercise, when
oxygen becomes unavailable.
large amounts of lactic acid are released into the blood
from the skeletal muscles and consumed as an extra
Formation of Lactic Acid During Anaerobic Glycolysis Allows
energy source by the heart.
Release of Extra Anaerobic Energy. The law of mass action
states that as the end products of a chemical reaction
build up in a reacting medium, the rate of the reaction
Release of Energy from
decreases, approaching zero. The two end products of
Glucose by the Pentose
the glycolytic reactions (see Figure 67-5) are (1) pyruvic
Phosphate Pathway
acid and (2) hydrogen atoms combined with NAD+ to
form NADH and H+. The buildup of either or both of
In almost all the body’s muscles, essentially all the car-
these would stop the glycolytic process and prevent
bohydrates utilized for energy are degraded to pyruvic
further formation of ATP. When their quantities begin
acid by glycolysis and then oxidized. However, this gly-
to be excessive, these two end products react with each
colytic scheme is not the only means by which glucose
other to form lactic acid, in accordance with the fol-
can be degraded and used to provide energy. A second
lowing equation:
important mechanism for the breakdown and oxidation
of glucose is called the pentose phosphate pathway (or
phosphogluconate pathway), which is responsible for as
OH
much as 30 per cent of the glucose breakdown in the
lactic
liver and even more than this in fat cells.
dehydrogenase
This pathway is especially important because it can
CH
C COOH
+
NADH
+
H+
3
provide energy independently of all the enzymes of the
(Pyruvic acid)
citric acid cycle and therefore is an alternative pathway
for energy metabolism when certain enzymatic abnor-
malities occur in cells. It has a special capacity for pro-
OH
viding energy to multiple cellular synthetic processes.
CH3
C COOH
+
NAD+
Release of Carbon Dioxide and Hydrogen by Means of the Pentose
Phosphate Pathway. Figure 67-8 shows most of the basic
chemical reactions in the pentose phosphate pathway. It
H
demonstrates that glucose, during several stages of con-
(Lactic acid)
version, can release one molecule of carbon dioxide and
four atoms of hydrogen, with the resultant formation
Thus, under anaerobic conditions, the major portion
of a five-carbon sugar, D-ribulose. This substance can
of the pyruvic acid is converted into lactic acid, which
change progressively into several other five-, four-,
diffuses readily out of the cells into the extracellular
seven-, and three-carbon sugars. Finally, various combi-
fluids and even into the intracellular fluids of other
nations of these sugars can resynthesize glucose.
less active cells. Therefore, lactic acid represents a type
However, only five molecules of glucose are resynthe-
of “sinkhole” into which the glycolytic end products
sized for every six molecules of glucose that initially enter
can disappear, thus allowing glycolysis to proceed far
into the reactions. That is, the pentose phosphate
longer than would otherwise be possible. Indeed, gly-
pathway is a cyclical process in which one molecule of
colysis could proceed for only a few seconds without
glucose is metabolized for each revolution of the cycle.
this conversion. Instead, it can proceed for several
Thus, by repeating the cycle again and again, all the
minutes, supplying the body with considerable extra
glucose can eventually be converted into carbon dioxide
quantities of ATP, even in the absence of respiratory
and hydrogen, and the hydrogen can enter the oxidative
oxygen.
phosphorylation pathway to form ATP; more often,
however, it is used for the synthesis of fat or other sub-
Reconversion of Lactic Acid to Pyruvic Acid When Oxygen
stances, as follows.
Becomes Available Again. When a person begins to breathe
oxygen again after a period of anaerobic metabolism,
Use of Hydrogen to Synthesize Fat; the Function of Nicotinamide
the lactic acid is rapidly reconverted to pyruvic acid and
Adenine Dinucleotide Phosphate. The hydrogen released
NADH plus H+. Large portions of these are immedi-
during the pentose phosphate cycle does not combine
ately oxidized to form large quantities of ATP. This
with NAD+ as in the glycolytic pathway but combines
excess ATP then causes as much as three fourths of the
with nicotinamide adenine dinucleotide phosphate
remaining excess pyruvic acid to be converted back into
(NADP+), which is almost identical to NAD+ except
glucose.
for an extra phosphate radical, P. This difference is
Thus, the large amount of lactic acid that forms during
extremely significant, because only hydrogen bound
anaerobic glycolysis is not lost from the body because,
with NADP+ in the form of NADPH can be used for
when oxygen is available again, the lactic acid can be
the synthesis of fats from carbohydrates (as discussed
either reconverted to glucose or used directly for
in Chapter 68) and for the synthesis of some other
energy. By far the greatest portion of this reconversion
substances.
838
Unit XIII
Metabolism and Temperature Regulation
Formation of Carbohydrates
from Proteins and Fats—
Glucose-6-phosphate
“Gluconeogenesis”
2H
6-Phosphoglucono-d-lactone
When the body’s stores of carbohydrates decrease
below normal, moderate quantities of glucose can be
formed from amino acids and the glycerol portion of fat.
6-Phosphogluconic acid
This process is called gluconeogenesis.
2H
Gluconeogenesis is especially important in prevent-
3-Keto-6-phosphogluconic acid
ing an excessive reduction in the blood glucose concen-
CO2
tration during fasting. Glucose is the primary substrate
for energy in tissues such as the brain and the red blood
D-Ribulose-5-phosphate
H2O
cells, and adequate amounts of glucose must be present
in the blood for several hours between meals. The liver
D-Xylulose-5-phosphate
plays a key role in maintaining blood glucose levels
+
during fasting by converting its stored glycogen to
D-Ribose-5-phosphate
glucose (glycogenolysis) and by synthesizing glucose,
mainly from lactate and amino acids (gluconeogenesis).
Approximately 25 per cent of the liver’s glucose pro-
D-Sedoheptulose-7-phosphate
duction during fasting is from gluconeogenesis, helping
+
to provide a steady supply of glucose to the brain.
D-Glyceraldehyde-3-phosphate
During prolonged fasting, the kidneys also synthesize
considerable amounts of glucose from amino acids and
other precursors.
Fructose-6-phosphate
About 60 per cent of the amino acids in the body pro-
+
teins can be converted easily into carbohydrates; the
Erythrose-4-phosphate
remaining 40 per cent have chemical configurations that
make this difficult or impossible. Each amino acid is
Net reaction:
converted into glucose by a slightly different chemical
Glucose + 12NADP+ + 6H2O
6CO2 + 12H + 12NADPH
process. For instance, alanine can be converted directly
into pyruvic acid simply by deamination; the pyruvic
acid is then converted into glucose or stored glycogen.
Several of the more complicated amino acids can be
Figure 67-8
converted into different sugars that contain three-,
four-, five-, or seven-carbon atoms; they can then enter
Pentose phosphate pathway for glucose metabolism.
the phosphogluconate pathway and eventually form
glucose. Thus, by means of deamination plus several
simple interconversions, many of the amino acids can
become glucose. Similar interconversions can change
glycerol into glucose or glycogen.
When the glycolytic pathway for using glucose
becomes slowed because of cellular inactivity, the
Regulation of Gluconeogenesis. Diminished carbohydrates
pentose phosphate pathway remains operative (mainly
in the cells and decreased blood sugar are the basic
in the liver) to break down any excess glucose that con-
stimuli that increase the rate of gluconeogenesis. Dimin-
tinues to be transported into the cells, and NADPH
ished carbohydrates can directly reverse many of the
becomes abundant to help convert acetyl-CoA, also
glycolytic and phosphogluconate reactions, thus allow-
derived from glucose, into long fatty acid chains. This is
ing the conversion of deaminated amino acids and
another way in which energy in the glucose molecule is
glycerol into carbohydrates. In addition, the hormone
used other than for the formation of ATP—in this
cortisol is especially important in this regulation, as
instance, for the formation and storage of fat in the body.
follows.
Glucose Conversion to Glycogen
Effect of Corticotropin and Glucocorticoids on Gluco-
or Fat
neogenesis. When normal quantities of carbohydrates
are not available to the cells, the adenohypophysis,
When glucose is not immediately required for energy,
for reasons not completely understood, begins to
the extra glucose that continually enters the cells is
secrete increased quantities of the hormone corti-
either stored as glycogen or converted into fat. Glucose
cotropin. This stimulates the adrenal cortex to produce
is preferentially stored as glycogen until the cells have
large quantities of glucocorticoid hormones, especially
stored as much glycogen as they can—an amount suffi-
cortisol. In turn, cortisol mobilizes proteins from
cient to supply the energy needs of the body for only 12
essentially all cells of the body, making these available
to 24 hours.
in the form of amino acids in the body fluids. A high
When the glycogen-storing cells (primarily liver and
proportion of these immediately become deaminated
muscle cells) approach saturation with glycogen, the
in the liver and provide ideal substrates for conversion
additional glucose is converted into fat in liver and fat
into glucose. Thus, one of the most important means
cells and is stored as fat in the fat cells. Other steps
by which gluconeogenesis is promoted is through
in the chemistry of this conversion are discussed in
the release of glucocorticoids from the adrenal
Chapter 68.
cortex.
Chapter 67
Metabolism of Carbohydrates, and Formation of Adenosine Triphosphate
839
Blood Glucose
Jiang G, Zhang BB: Glucagon and regulation of glucose
metabolism. Am J Physiol Endocrinol Metab 284:E671,
The normal blood glucose concentration in a person
2003.
who has not eaten a meal within the past 3 to 4 hours
Jungas RL, Halperin ML, Brosnan JT: Quantitative analysis
is about 90 mg/dl. After a meal containing large amounts
of amino acid oxidation and related gluconeogenesis in
of carbohydrates, this level seldom rises above 140 mg/
humans. Physiol Rev 72:419, 1992.
dl unless the person has diabetes mellitus, which is dis-
Krebs HA: The tricarboxylic acid cycle. Harvey Lect 44:165,
cussed in Chapter 78.
1948-1949.
The regulation of blood glucose concentration is inti-
Kunji ER: The role and structure of mitochondrial carriers.
mately related to the pancreatic hormones insulin and
FEBS Lett 564:239, 2004.
glucagon; this subject is discussed in detail in Chapter
Lam TK, Carpentier A, Lewis GF, et al: Mechanisms of
78 in relation to the functions of these hormones.
the free fatty acid-induced increase in hepatic glucose
production. Am J Physiol Endocrinol Metab 284:E863,
2003.
References
Mills DA, Ferguson-Miller S: Understanding the mechanism
of proton movement linked to oxygen reduction in
Barrett EJ: Insulin’s effect on glucose production: direct or
cytochrome c oxidase: lessons from other proteins. FEBS
indirect? J Clin Invest 111:434, 2003.
Lett 545:47, 2003.
Barthel A, Schmoll D: Novel concepts in insulin regulation
Pilkis SJ, Granner DK: Molecular physiology of the regula-
of hepatic gluconeogenesis. Am J Physiol Endocrinol
tion of hepatic gluconeogenesis and glycolysis. Annu Rev
Metab 285:E685, 2003.
Physiol 54:885, 1992.
Ceulemans H, Bollen M: Functional diversity of protein
Roden M, Bernroider E: Hepatic glucose metabolism in
phosphatase-1, a cellular economizer and reset button.
humans—its role in health and disease. Best Pract Res
Physiol Rev 84:1, 2004.
Clin Endocrinol Metab 17:365, 2003.
Duchen MR: Roles of mitochondria in health and disease.
Ronquist G, Waldenstrom A: Imbalance of plasma mem-
Diabetes 53(Suppl 1):S96, 2004.
brane ion leak and pump relationship as a new aetiologi-
Ferrer JC, Favre C, Gomis RR, et al: Control of glycogen
cal basis of certain disease states. J Intern Med 254:517,
deposition. FEBS Lett 546:127, 2003.
2003.
Gleeson TT: Post-exercise lactate metabolism: a comparative
Spriet LL, Watt MJ: Regulatory mechanisms in the interac-
review of sites, pathways, and regulation. Annu Rev
tion between carbohydrate and lipid oxidation during
Physiol 58:565, 1996.
exercise. Acta Physiol Scand 178:443, 2003.
Gunter TE, Yule DI, Gunter KK, et al: Calcium and mito-
Wolfsdorf JI, Weinstein DA: Glycogen storage diseases. Rev
chondria. FEBS Lett 567:96, 2004.
Endocr Metab Disord 4:95, 2003.
Jackson JB: Proton translocation by transhydrogenase.
FEBS Lett 545:18, 2003.
C H A P T E R
6
8
Lipid Metabolism
Several chemical compounds in food and in the
body are classified as lipids. They include (1) neutral fat,
also known as triglycerides;
(2) phospholipids;
(3) cholesterol; and (4) a few others of less importance.
Chemically, the basic lipid moiety of the triglycerides
and the phospholipids is fatty acids, which are simply
long-chain hydrocarbon organic acids. A typical fatty
acid, palmitic acid, is the following: CH3(CH2)14COOH.
Although cholesterol does not contain fatty acid, its
sterol nucleus is synthesized from portions of fatty acid molecules, thus giving it
many of the physical and chemical properties of other lipid substances.
The triglycerides are used in the body mainly to provide energy for the different
metabolic processes, a function they share almost equally with the carbohydrates.
However, some lipids, especially cholesterol, the phospholipids, and small amounts
of triglycerides, are used to form the membranes of all cells of the body and to
perform other cellular functions.
Basic Chemical Structure of Triglycerides (Neutral Fat). Because most of this chapter deals
with the utilization of triglycerides for energy, the following typical structure of the
triglyceride molecule must be understood.
CH3—(CH2)16—COO—CH2
|
CH3—(CH2)16—COO—CH
|
CH3—(CH2)16—COO—CH2
Tristearin
Note that three long-chain fatty acid molecules are bound with one molecule of
glycerol. The three fatty acids most commonly present in the triglycerides of the
human body are (1) stearic acid (shown in the tristearin example above), which has
an 18-carbon chain and is fully saturated with hydrogen atoms; (2) oleic acid, which
also has an 18-carbon chain but has one double bond in the middle of the chain;
and (3) palmitic acid, which has 16 carbon atoms and is fully saturated.
Transport of Lipids in the Body Fluids
Transport of Triglycerides and Other Lipids from the
Gastrointestinal Tract by Lymph—The Chylomicrons
As explained in Chapter 65, almost all the fats in the diet, with the principal excep-
tion of a few short-chain fatty acids, are absorbed from the intestines into the intes-
tinal lymph. During digestion, most triglycerides are split into monoglycerides and
fatty acids. Then, while passing through the intestinal epithelial cells, the mono-
glycerides and fatty acids are resynthesized into new molecules of triglycerides that
enter the lymph as minute, dispersed droplets called chylomicrons, whose diame-
ters are between 0.08 and 0.6 micron. A small amount of apoprotein B is adsorbed
to the outer surfaces of the chylomicrons. This leaves the remainder of the protein
molecules projecting into the surrounding water and thereby increases the suspen-
sion stability of the chylomicrons in the lymph fluid and prevents their adherence
to the lymphatic vessel walls.
Most of the cholesterol and phospholipids absorbed from the gastrointestinal
tract enter the chylomicrons. Thus, although the chylomicrons are composed prin-
cipally of triglycerides, they also contain about 9 per cent phospholipids, 3 per cent
cholesterol, and 1 per cent apoprotein B. The chylomicrons are then transported
upward through the thoracic duct and emptied into the circulating venous blood at
the juncture of the jugular and subclavian veins.
840
Chapter 68
Lipid Metabolism
841
Removal of the Chylomicrons
1. Despite the minute amount of free fatty acid in
from the Blood
the blood, its rate of “turnover” is extremely
rapid: half the plasma fatty acid is replaced by
About 1 hour after a meal that contains large quantities
new fatty acid every 2 to 3 minutes. One can
of fat, the chylomicron concentration in the plasma may
calculate that at this rate, almost all the normal
rise to 1 to 2 per cent of the total plasma, and because
energy requirements of the body can be provided
of the large size of the chylomicrons, the plasma appears
by the oxidation of transported free fatty acids,
turbid and sometimes yellow. However, the chylomi-
without using any carbohydrates or proteins for
crons have a half-life of less than 1 hour, so the plasma
energy.
becomes clear again within a few hours. The fat of the
2. Conditions that increase the rate of utilization
chylomicrons is removed mainly in the following way.
of fat for cellular energy also increase the free
fatty acid concentration in the blood; in fact, the
Chylomicron Triglycerides Are Hydrolyzed by Lipoprotein Lipase,
concentration sometimes increases fivefold to
and Fat Is Stored in Adipose Tissue and Liver Cells. Most of the
eightfold. Such a large increase occurs especially in
chylomicrons are removed from the circulating blood as
cases of starvation and in diabetes; in both these
they pass through the capillaries of adipose tissue or the
conditions, the person derives little or no metabolic
liver. Both adipose tissue and the liver contain large
energy from carbohydrates.
quantities of the enzyme lipoprotein lipase. This enzyme
Under normal conditions, only about 3 molecules of
is especially active in the capillary endothelium, where
fatty acid combine with each molecule of albumin, but
it hydrolyzes the triglycerides of chylomicrons as they
as many as 30 fatty acid molecules can combine with a
come in contact with the endothelial wall, thus releas-
single albumin molecule when the need for fatty acid
ing fatty acids and glycerol.
transport is extreme. This shows how variable the rate
The fatty acids, being highly miscible with the mem-
of lipid transport can be under different physiologic
branes of the cells, immediately diffuse into the fat cells
conditions.
of the adipose tissue and into the liver cells. Once inside
these cells, the fatty acids are again synthesized into
triglycerides, with new glycerol being supplied by the
Lipoproteins—Their Special
metabolic processes of the storage cells, as discussed
Function in Transporting
later in the chapter. The lipase also causes hydrolysis of
phospholipids; this, too, releases fatty acids to be stored
Cholesterol and Phospholipids
in the cells in the same way.
In the postabsorptive state, after all the chylomicrons
have been removed from the blood, more than 95
“Free Fatty Acids” Are Transported
per cent of all the lipids in the plasma are in the
in the Blood in Combination
form of lipoprotein. These are small particles—much
with Albumin
smaller than chylomicrons, but qualitatively similar
in composition—containing triglycerides, cholesterol,
When fat that has been stored in the adipose tissue is to
phospholipids, and protein. The total concentration
be used elsewhere in the body to provide energy, it must
of lipoproteins in the plasma averages about 700 mg
first be transported from the adipose tissue to the other
per 100 ml of plasma—that is, 700 mg/dl. This can be
tissue. It is transported mainly in the form of free fatty
broken down into the following individual lipoprotein
acids. This is achieved by hydrolysis of the triglycerides
constituents:
back into fatty acids and glycerol.
At least two classes of stimuli play important roles
mg/dl of plasma
in promoting this hydrolysis. First, when the amount
of glucose available to the fat cell is inadequate, one of
Cholesterol
180
the glucose breakdown products, a-glycerophosphate, is
Phospholipids
160
Triglycerides
160
also available in insufficient quantities. Because this
Protein
200
substance is required to maintain the glycerol portion
of triglycerides, the result is hydrolysis of triglycerides.
Second, a hormone-sensitive cellular lipase can be acti-
Types of Lipoproteins. Aside from the chylomicrons, which
vated by several hormones from the endocrine glands,
are themselves very large lipoproteins, there are four
and this also promotes rapid hydrolysis of triglycerides.
major types of lipoproteins, classified by their densities
This is discussed later in the chapter.
as measured in the ultracentrifuge: (1) very low density
On leaving fat cells, fatty acids ionize strongly in the
lipoproteins, which contain high concentrations of
plasma, and the ionic portion combines immediately
triglycerides and moderate concentrations of both cho-
with albumin molecules of the plasma proteins. Fatty
lesterol and phospholipids;
(2) intermediate-density
acids bound in this manner are called free fatty acids
lipoproteins, which are very low density lipoproteins
or nonesterified fatty acids, to distinguish them from
from which a share of the triglycerides has been
other fatty acids in the plasma that exist in the form
removed, so that the concentrations of cholesterol and
of (1) esters of glycerol, (2) cholesterol, or (3) other
phospholipids are increased; (3) low-density lipopro-
substances.
teins, which are derived from intermediate-density
The concentration of free fatty acids in the plasma
lipoproteins by the removal of almost all the triglyc-
under resting conditions is about 15 mg/dl, which is a
erides, leaving an especially high concentration of
total of only 0.45 gram of fatty acids in the entire circu-
cholesterol and a moderately high concentration of
latory system. Strangely enough, even this small amount
phospholipids; and (4) high-density lipoproteins, which
accounts for almost all the transport of fatty acids from
contain a high concentration of protein (about 50 per
one part of the body to another for the following
cent) but much smaller concentrations of cholesterol
reasons:
and phospholipids.
842
Unit XIII
Metabolism and Temperature Regulation
Formation and Function of Lipoproteins. Almost all the
can be used for energy; (2) synthesize triglycerides,
lipoproteins are formed in the liver, which is also where
mainly from carbohydrates, but to a lesser extent from
most of the plasma cholesterol, phospholipids, and
proteins as well; and (3) synthesize other lipids from
triglycerides are synthesized. In addition, small quanti-
fatty acids, especially cholesterol and phospholipids.
ties of high-density lipoproteins are synthesized in the
Large quantities of triglycerides appear in the liver
intestinal epithelium during the absorption of fatty
(1) during the early stages of starvation, (2) in diabetes
acids from the intestines.
mellitus, and (3) in any other condition in which fat
The primary function of the lipoproteins is to trans-
instead of carbohydrates is being used for energy. In
port their lipid components in the blood. The very low
these conditions, large quantities of triglycerides are
density lipoproteins transport triglycerides synthesized
mobilized from the adipose tissue, transported as free
in the liver mainly to the adipose tissue, whereas the
fatty acids in the blood, and redeposited as triglycerides
other lipoproteins are especially important in the dif-
in the liver, where the initial stages of much of fat degra-
ferent stages of phospholipid and cholesterol transport
dation begin. Thus, under normal physiologic condi-
from the liver to the peripheral tissues or from the
tions, the total amount of triglycerides in the liver is
periphery back to the liver. Later in the chapter, we
determined to a great extent by the overall rate at which
discuss in more detail special problems of cholesterol
lipids are being used for energy.
transport in relation to the disease atherosclerosis, which
The liver cells, in addition to containing triglycerides,
is associated with the development of fatty lesions on
contain large quantities of phospholipids and choles-
the insides of arterial walls.
terol, which are continually synthesized by the liver.
Also, the liver cells are much more capable than other
tissues of desaturating fatty acids, so that liver triglyc-
Fat Deposits
erides normally are much more unsaturated than the
triglycerides of adipose tissue. This capability of the liver
Adipose Tissue
to desaturate fatty acids is functionally important to all
tissues of the body, because many structural elements of
Large quantities of fat are stored in two major tissues
all cells contain reasonable quantities of unsaturated
of the body, the adipose tissue and the liver. The adipose
fats, and their principal source is the liver. This desatu-
tissue is usually called fat deposits, or simply tissue fat.
ration is accomplished by a dehydrogenase in the liver
The major function of adipose tissue is storage of
cells.
triglycerides until they are needed to provide energy
elsewhere in the body. A subsidiary function is to
provide heat insulation for the body, as discussed in
Use of Triglycerides
Chapter 73.
for Energy: Formation
Fat Cells
(Adipocytes). The fat cells
(adipocytes) of
of Adenosine Triphosphate
adipose tissue are modified fibroblasts that store almost
About 40 per cent of the calories in a typical American
pure triglycerides in quantities as great as 80 to 95 per
diet are derived from fats, which is almost equal to the
cent of the entire cell volume. Triglycerides inside the
calories derived from carbohydrates. Therefore, the use
fat cells are generally in a liquid form. When the tissues
of fats by the body for energy is as important as the use
are exposed to prolonged cold, the fatty acid chains of
of carbohydrates is. In addition, many of the carbohy-
the cell triglycerides, over a period of weeks, become
drates ingested with each meal are converted into
either shorter or more unsaturated to decrease their
triglycerides, then stored, and used later in the form of
melting point, thereby always allowing the fat to remain
fatty acids released from the triglycerides for energy.
in a liquid state. This is particularly important, because
only liquid fat can be hydrolyzed and transported from
Hydrolysis of Triglycerides. The first stage in using triglyc-
the cells.
erides for energy is their hydrolysis into fatty acids and
Fat cells can synthesize very small amounts of fatty
glycerol. Then, both the fatty acids and the glycerol are
acids and triglycerides from carbohydrates; this function
transported in the blood to the active tissues, where they
supplements the synthesis of fat in the liver, as discussed
will be oxidized to give energy. Almost all cells—with
later in the chapter.
some exceptions, such as brain tissue and red blood
cells—can use fatty acids for energy.
Exchange of Fat Between the Adipose Tissue and the Blood—
Glycerol, on entering the active tissue, is immediately
Tissue Lipases. As discussed earlier, large quantities of
changed by intracellular enzymes into glycerol-3-
lipases are present in adipose tissue. Some of these
phosphate, which enters the glycolytic pathway for
enzymes catalyze the deposition of cell triglycerides
glucose breakdown and is thus used for energy. Before
from the chylomicrons and lipoproteins. Others, when
the fatty acids can be used for energy, they must be
activated by hormones, cause splitting of the triglyc-
processed further in the following way.
erides of the fat cells to release free fatty acids. Because
of the rapid exchange of fatty acids, the triglycerides in
Entry of Fatty Acids into Mitochondria. Degradation and oxi-
fat cells are renewed about once every 2 to 3 weeks,
dation of fatty acids occur only in the mitochondria.
which means that the fat stored in the tissues today is
Therefore, the first step for the use of fatty acids is
not the same fat that was stored last month, thus empha-
their transport into the mitochondria. This is a carrier-
sizing the dynamic state of storage fat.
mediated process that uses carnitine as the carrier sub-
stance. Once inside the mitochondria, fatty acids split
Liver Lipids
away from carnitine and are degraded and oxidized.
The principal functions of the liver in lipid metabolism
Degradation of Fatty Acids to Acetyl Coenzyme A by Beta-
are to (1) degrade fatty acids into small compounds that
Oxidation. The fatty acid molecule is degraded in the
Chapter 68
Lipid Metabolism
843
Thiokinase
(1) RCH2CH2CH2COOH + CoA + ATP
RCH2CH2CH2COCoA + AMP + Pyrophosphate
(Fatty acid)
(Fatty acyl-CoA)
Acyl dehydrogenase
(2) RCH2CH2CH2COCoA + FAD
RCH2CH=CHCOCoA + FADH2
(Fatty acyl-CoA)
Enoyl hydrase
(3) RCH2CH=CHCOCoA + H2O
RCH2CHOHCH2COCoA
b-Hydroxyacyl
(4) RCH2CHOHCH2COCoA + NAD+
RCH2COCH2COCoA + NADH + H+
dehydrogenase
Thiolase
(5) RCH
RCH2COCoA + CH3COCoA
2COCH2COCoA + CoA
(Fatty acyl-CoA) (Acetyl-CoA)
Figure 68-1
Beta-oxidation of fatty acids to yield acetyl coenzyme A.
mitochondria by progressive release of two-carbon seg-
Thus, after initial degradation of fatty acids to acetyl-
ments in the form of acetyl coenzyme A (acetyl-CoA).
CoA, their final breakdown is precisely the same as that
This process, which is shown in Figure 68-1, is called the
of the acetyl-CoA formed from pyruvic acid during the
beta-oxidation process for degradation of fatty acids.
metabolism of glucose. And the extra hydrogen atoms
To understand the essential steps in the beta-
are also oxidized by the same chemiosmotic oxidative
oxidation process, note that in equation 1 the first step
system of the mitochondria that is used in carbohydrate
is combination of the fatty acid molecule with coenzyme
oxidation, liberating large amounts of adenosine
A (CoA) to form fatty acyl-CoA. In equations 2, 3, and
triphosphate (ATP).
4, the beta carbon (the second carbon from the right) of
the fatty acyl-CoA binds with an oxygen molecule—that
Tremendous Amounts of ATP Are Formed by Oxidation of Fatty
is, the beta carbon becomes oxidized.
Acids. In Figure 68-1, note that the 4 separate hydrogen
Then, in equation
5, the right-hand two-carbon
atoms released each time a molecule of acetyl-CoA is
portion of the molecule is split off to release acetyl-CoA
split from the fatty acid chain are released in the forms
into the cell fluid. At the same time, another CoA mol-
FADH2, NADH, and H+. Therefore, for every stearic
ecule binds at the end of the remaining portion of the
fatty acid molecule that is split to form 9 acetyl-CoA
fatty acid molecule, and this forms a new fatty acyl-CoA
molecules, 32 extra hydrogen atoms are removed. In
molecule; this time, however, the molecule is two carbon
addition, for each of the 9 molecules of acetyl-CoA that
atoms shorter because of the loss of the first acetyl-CoA
are subsequently degraded by the citric acid cycle, 8
from its terminal end.
more hydrogen atoms are removed, making another 72
Next, this shorter fatty acyl-CoA enters into equation
hydrogens. This makes a total of 104 hydrogen atoms
2 and progresses through equations 3, 4, and 5 to release
eventually released by the degradation of each stearic
still another acetyl-CoA molecule, thus shortening the
acid molecule. Of this group, 34 are removed from
original fatty acid molecule by another two carbons. In
the degrading fatty acids by flavoproteins, and 70 are
addition to the released acetyl-CoA molecules, four
removed by nicotinamide adenine dinucleotide (NAD+)
atoms of hydrogen are released from the fatty acid mol-
as NADH and H+.
ecule at the same time, entirely separate from the
These two groups of hydrogen atoms are oxidized in
acetyl-CoA.
the mitochondria, as discussed in Chapter 67, but they
enter the oxidative system at different points, so that
Oxidation of Acetyl-CoA. The acetyl-CoA molecules
1 molecule of ATP is synthesized for each of the 34
formed by beta-oxidation of fatty acids in the mito-
flavoprotein hydrogens, and 1.5 molecules of ATP are
chondria enter immediately into the citric acid cycle (see
synthesized for each of the 70 NADH and H+ hydro-
Chapter 67), combining first with oxaloacetic acid to
gens. This makes 34 plus 105, or a total of 139 molecules
form citric acid, which then is degraded into carbon
of ATP formed by the oxidation of hydrogen derived
dioxide and hydrogen atoms. The hydrogen is subse-
from each molecule of stearic acid. Another 9 molecules
quently oxidized by the chemiosmotic oxidative system
of ATP are formed in the citric acid cycle itself (sepa-
of the mitochondria, which was also explained in
rate from the ATP released by the oxidation of hydro-
Chapter 67. The net reaction in the citric acid cycle for
gen), one for each of the
9 acetyl-CoA molecules
each molecule of acetyl-CoA is the following:
metabolized. Thus, a total of 148 molecules of ATP are
formed during the complete oxidation of 1 molecule of
CH3COCo-A + Oxaloacetic acid+ 3H2O + ADP
stearic acid. However, two high-energy bonds are con-
Citric acid cycle
sumed in the initial combination of CoA with the stearic
æææææææÆ
acid molecule, making a net gain of 146 molecules of
2CO2+ 8H + HCo-A + ATP + Oxaloacetic acid
ATP.
844
Unit XIII
Metabolism and Temperature Regulation
Formation of Acetoacetic Acid
carbohydrates are metabolized—in starvation and with
in the Liver and Its Transport
a high-fat diet because carbohydrates are not available,
and in diabetes because insulin is not available to cause
in the Blood
glucose transport into the cells.
A large share of the initial degradation of fatty acids
When carbohydrates are not used for energy, almost
occurs in the liver, especially when excessive amounts
all the energy of the body must come from metabolism
of lipids are being used for energy. However, the liver
of fats. We shall see later in the chapter that the unavail-
uses only a small proportion of the fatty acids for its own
ability of carbohydrates automatically increases the rate
intrinsic metabolic processes. Instead, when the fatty
of removal of fatty acids from adipose tissues; in addi-
acid chains have been split into acetyl-CoA, two mole-
tion, several hormonal factors—such as increased secre-
cules of acetyl-CoA condense to form one molecule of
tion of glucocorticoids by the adrenal cortex, increased
acetoacetic acid, which is then transported in the blood
secretion of glucagon by the pancreas, and decreased
to the other cells throughout the body, where it is used
secretion of insulin by the pancreas—further enhance
for energy. The chemical processes are the following:
the removal of fatty acids from the fat tissues. As a
result, tremendous quantities of fatty acids become
available (1) to the peripheral tissue cells to be used for
liver cells
2CH3COCo-A + H2O
æææææƨææææ æ
energy and (2) to the liver cells, where much of the fatty
other cells
acids is converted to ketone bodies.
-
Acetyl
CoA
The ketone bodies pour out of the liver to be carried
CH3COCH2COOH
+
2HCo-A
to the cells. For several reasons, the cells are limited in
the amount of ketone bodies that can be oxidized; the
Acetoacetic acid
most important reason is the following: One of the prod-
ucts of carbohydrate metabolism is the oxaloacetate that
Part of the acetoacetic acid is also converted into
is required to bind with acetyl-CoA before it can be
b-hydroxybutyric acid, and minute quantities are
processed in the citric acid cycle. Therefore, deficiency
converted into acetone in accord with the following
of oxaloacetate derived from carbohydrates limits the
reactions:
entry of acetyl-CoA into the citric acid cycle, and when
there is a simultaneous outpouring of large quantities
O
O
of acetoacetic acid and other ketone bodies from the
liver, the blood concentrations of acetoacetic acid and
CH3
C CH2
C OH
b-hydroxybutyric acid sometimes rise to as high as
20 times normal, thus leading to extreme acidosis, as
Acetoacetic acid
explained in Chapter 30.
+ 2H
The acetone that is formed during ketosis is a volatile
-CO2
substance, some of which is blown off in small quanti-
OH
O
O
ties in the expired air of the lungs. This gives the breath
an acetone smell that is frequently used as a diagnostic
criterion of ketosis.
CH3
CH CH2
C OH
CH3
C CH3
b-Hydroxybutyric acid
Acetone
Adaptation to a High-Fat Diet. When changing slowly from
a carbohydrate diet to an almost completely fat diet, a
person’s body adapts to use far more acetoacetic acid
The acetoacetic acid, b-hydroxybutyric acid, and
than usual, and in this instance, ketosis normally does
acetone diffuse freely through the liver cell membranes
not occur. For instance, the Inuit (Eskimos), who some-
and are transported by the blood to the peripheral
times live almost entirely on a fat diet, do not develop
tissues. Here they again diffuse into the cells, where
ketosis. Undoubtedly, several factors, none of which is
reverse reactions occur and acetyl-CoA molecules are
clear, enhance the rate of acetoacetic acid metabolism
formed. These in turn enter the citric acid cycle and are
by the cells. After a few weeks, even the brain cells,
oxidized for energy, as already explained.
which normally derive almost all their energy from
Normally, the acetoacetic acid and b-hydroxybutyric
glucose, can derive 50 to 75 per cent of their energy from
acid that enter the blood are transported so rapidly to
fats.
the tissues that their combined concentration in the
plasma seldom rises above 3 mg/dl. Yet despite this
small concentration in the blood, large quantities are
Synthesis of Triglycerides
actually transported, as is also true for free fatty acid
from Carbohydrates
transport. The rapid transport of both these substances
results from their high solubility in the membranes of
Whenever a greater quantity of carbohydrates enters
the target cells, which allows almost instantaneous dif-
the body than can be used immediately for energy or
fusion into the cells.
can be stored in the form of glycogen, the excess is
rapidly converted into triglycerides and stored in this
Ketosis in Starvation, Diabetes, and Other Diseases. The con-
form in the adipose tissue.
centrations of acetoacetic acid, b-hydroxybutyric acid,
In human beings, most triglyceride synthesis occurs in
and acetone occasionally rise to levels many times
the liver, but minute quantities are also synthesized in
normal in the blood and interstitial fluids; this condition
the adipose tissue itself. The triglycerides formed in the
is called ketosis, because acetoacetic acid is a keto acid.
liver are transported mainly in very low density lipopro-
The three compounds are called ketone bodies. Ketosis
teins to the adipose tissue, where they are stored.
occurs especially in starvation, in diabetes mellitus, and
sometimes even when a person’s diet is composed
Conversion of Acetyl-CoA into Fatty Acids. The first step
almost entirely of fat. In all these states, essentially no
in the synthesis of triglycerides is conversion of
Chapter 68
Lipid Metabolism
845
carbohydrates into acetyl-CoA. As explained in
Efficiency of Carbohydrate Conversion into Fat. During triglyc-
Chapter 67, this occurs during the normal degradation
eride synthesis, only about 15 per cent of the original
of glucose by the glycolytic system. Because fatty acids
energy in the glucose is lost in the form of heat; the
are actually large polymers of acetic acid, it is easy to
remaining 85 per cent is transferred to the stored
understand how acetyl-CoA can be converted into fatty
triglycerides.
acids. However, the synthesis of fatty acids from acetyl-
CoA is not achieved by simply reversing the oxidative
Importance of Fat Synthesis and Storage. Fat synthesis from
degradation described earlier. Instead, this occurs by the
carbohydrates is especially important for two reasons:
two-step process shown in Figure 68-2, using malonyl-
1. The ability of the different cells of the body to
CoA and NADPH as the principal intermediates in the
store carbohydrates in the form of glycogen is
polymerization process.
generally slight; a maximum of only a few hundred
grams of glycogen can be stored in the liver, the
Combination of Fatty Acids with a-Glycerophosphate to Form
skeletal muscles, and all other tissues of the body
Triglycerides. Once the synthesized fatty acid chains have
put together. In contrast, many kilograms of fat
grown to contain 14 to 18 carbon atoms, they bind with
can be stored. Therefore, fat synthesis provides a
glycerol to form triglycerides. The enzymes that cause
means by which the energy of excess ingested
this conversion are highly specific for fatty acids with
carbohydrates (and proteins) can be stored for
chain lengths of 14 carbon atoms or greater, a factor that
later use. Indeed, the average person has almost
controls the physical quality of the triglycerides stored
150 times as much energy stored in the form of fat
in the body.
as stored in the form of carbohydrate.
As shown in Figure 68-3, the glycerol portion of
2. Each gram of fat contains almost two and a half
triglycerides is furnished by a-glycerophosphate, which
times the calories of energy contained by each
is another product derived from the glycolytic scheme
gram of glycogen. Therefore, for a given weight
of glucose degradation. This mechanism is discussed in
gain, a person can store several times as much
Chapter 67.
energy in the form of fat as in the form of
carbohydrate, which is exceedingly important when
an animal must be highly motile to survive.
Failure to Synthesize Fats from Carbohydrates in the Absence of
Insulin. When no insulin is available, as occurs in serious
Step 1:
diabetes mellitus, fats are poorly synthesized, if at all,
+ ATP
CH3COCoA + CO2
for the following reasons: First, when insulin is not avail-
(Acetyl-CoA carboxylase)
able, glucose does not enter the fat and liver cells satis-
factorily, so that little of the acetyl-CoA and NADPH
COOH
needed for fat synthesis can be derived from glucose.
Second, lack of glucose in the fat cells greatly reduces
CH
2
+ ADP + PO4-3
the availability of a-glycerophosphate, which also
makes it difficult for the tissues to form triglycerides.
O
C
CoA
Malonyl-CoA
Synthesis of Triglycerides
Step 2:
from Proteins
1 Acetyl-CoA + Malonyl-CoA + 16NADPH + 16H+
O
1 Steric acid + 8CO2 + 9CoA + 16NADP+ + 7H2
Many amino acids can be converted into acetyl-CoA, as
discussed in Chapter 69. The acetyl-CoA can then be
synthesized into triglycerides. Therefore, when people
Figure 68-2
have more proteins in their diets than their tissues can
use as proteins, a large share of the excess is stored as
Synthesis of fatty acids.
fat.
Glucose
Pentose
Glycolytic
phosphate
pathway
pathway
a-Glycerophosphate + Acetyl-CoA + NADH + H+
NADPH + H+
Fatty acids
Figure 68-3
Triglycerides
Overall schema for synthesis of
triglycerides from glucose.
846
Unit XIII
Metabolism and Temperature Regulation
Regulation of Energy
Probably the most dramatic increase that occurs in fat
utilization is that observed during heavy exercise. This
Release from Triglycerides
results almost entirely from release of epinephrine and
Carbohydrates Are Preferred over Fats for Energy When Excess
norepinephrine by the adrenal medullae during exer-
Carbohydrates Are Available. When excess quantities of
cise, as a result of sympathetic stimulation. These two
carbohydrates are available in the body, carbohydrates
hormones directly activate hormone-sensitive triglyc-
are used preferentially over triglycerides for energy.
eride lipase, which is present in abundance in the fat
There are several reasons for this “fat-sparing” effect of
cells, and this causes rapid breakdown of triglycerides
carbohydrates. One of the most important is the fol-
and mobilization of fatty acids. Sometimes the free fatty
lowing: The fats in adipose tissue cells are present in two
acid concentration in the blood of an exercising person
forms: stored triglycerides and small quantities of free
rises as much as eightfold, and the use of these fatty
fatty acids. They are in constant equilibrium with each
acids by the muscles for energy is correspondingly
other. When excess quantities of a-glycerophosphate are
increased. Other types of stress that activate the sym-
present (which occurs when excess carbohydrates are
pathetic nervous system can also increase fatty acid
available), the excess a-glycerophosphate binds the
mobilization and utilization in a similar manner.
free fatty acids in the form of stored triglycerides. As a
Stress also causes large quantities of corticotropin to
result, the equilibrium between free fatty acids and
be released by the anterior pituitary gland, and this
triglycerides shifts toward the stored triglycerides;
causes the adrenal cortex to secrete extra quantities of
consequently, only minute quantities of fatty acids
glucocorticoids. Both corticotropin and glucocorticoids
are available to be used for energy. Because a-
activate either the same hormone-sensitive triglyceride
glycerophosphate is an important product of glucose
lipase as that activated by epinephrine and norepi-
metabolism, the availability of large amounts of glucose
nephrine or a similar lipase. When corticotropin and
automatically inhibits the use of fatty acids for energy.
glucocorticoids are secreted in excessive amounts for
Second, when carbohydrates are available in excess,
long periods, as occurs in the endocrine condition called
fatty acids are synthesized more rapidly than they are
Cushing’s disease, fats are frequently mobilized to such
degraded. This effect is caused partially by the large
a great extent that ketosis results. Corticotropin and
quantities of acetyl-CoA formed from the carbohy-
glucocorticoids are then said to have a ketogenic effect.
drates and by the low concentration of free fatty acids
Growth hormone has an effect similar to but weaker
in the adipose tissue, thus creating conditions appropri-
than that of corticotropin and glucocorticoids in
ate for the conversion of acetyl-CoA into fatty acids.
activating hormone-sensitive lipase. Therefore, growth
An even more important effect that promotes the
hormone can also have a mild ketogenic effect.
conversion of carbohydrates to fats is the following: The
Finally, thyroid hormone causes rapid mobilization
first step, which is the rate-limiting step, in the synthesis
of fat, which is believed to result indirectly from an
of fatty acids is carboxylation of acetyl-CoA to form
increased overall rate of energy metabolism in all cells
malonyl-CoA. The rate of this reaction is controlled
of the body under the influence of this hormone. The
primarily by the enzyme acetyl-CoA carboxylase, the
resulting reduction in acetyl-CoA and other intermedi-
activity of which is accelerated in the presence of inter-
ates of both fat and carbohydrate metabolism in the
mediates of the citric acid cycle. When excess carbohy-
cells is a stimulus to fat mobilization.
drates are being used, these intermediates increase,
The effects of the different hormones on metabolism
automatically causing increased synthesis of fatty acids.
are discussed further in the chapters dealing with each
Thus, an excess of carbohydrates in the diet not only
hormone.
acts as a fat-sparer but also increases fat stores. In fact,
all the excess carbohydrates not used for energy or
stored in the small glycogen deposits of the body are
Obesity
converted to fat for storage.
Obesity means deposition of excess fat in the body. This
subject is discussed in Chapter 71 in relation to dietary
Acceleration of Fat Utilization for Energy in the Absence of Car-
balances, but briefly, it is caused by the ingestion of
bohydrates. All the fat-sparing effects of carbohydrates
greater amounts of food than can be used by the body
are lost and actually reversed when carbohydrates are
for energy. The excess food, whether fats, carbohydrates,
not available. The equilibrium shifts in the opposite
or proteins, is then stored almost entirely as fat in the
direction, and fat is mobilized from the adipose cells and
adipose tissue, to be used later for energy.
used for energy in place of carbohydrates.
Strains of rats have been found in which hereditary
Also important are several hormonal changes that
obesity occurs. In at least one of these, the obesity is
take place to promote rapid fatty acid mobilization from
caused by ineffective mobilization of fat from the
adipose tissue. Among the most important of these is
adipose tissue by tissue lipase, while synthesis and
a marked decrease in pancreatic secretion of insulin
storage of fat continue normally. Such a one-way
caused by the absence of carbohydrates. This not only
process causes progressive enhancement of the fat
reduces the rate of glucose utilization by the tissues
stores, resulting in severe obesity.
but also decreases fat storage, which further shifts the
equilibrium in favor of fat metabolism in place of
carbohydrates.
Phospholipids and
Hormonal Regulation of Fat Utilization. At least seven of
Cholesterol
the hormones secreted by the endocrine glands have
significant effects on fat utilization. Some important
Phospholipids
hormonal effects on fat metabolism—in addition to
insulin lack, discussed in the previous paragraph—are
The major types of body phospholipids are lecithins,
noted here.
cephalins, and sphingomyelin; their typical chemical
Chapter 68
Lipid Metabolism
847
O
CH3
CH3
H
2C
O
C
(CH2)7
CH CH
(CH2)7
CH3
CH3
CH
(CH2)
3
CH
O
CH3
CH3
HC
O
C
(CH2)16
CH3
O
CH3
CH3
HO
H2C
O
P
O
CH2
CH2
N+
CH3
OH
A lecithin
O
Figure 68-5
H
O
C
(CH2)7
CH CH
(CH2)7
CH
Cholesterol.
2C
3
O
HC
O
C
(CH2)16
CH3
rate of fat metabolism because, when triglycerides are
deposited in the liver, the rate of phospholipid forma-
O
tion increases. Also, certain specific chemical substances
H2C
O
P
O
CH2
CH2
N+H3
are needed for the formation of some phospholipids.
For instance, choline, either obtained in the diet or syn-
OH
thesized in the body, is needed for the formation of
A cephalin
lecithin, because choline is the nitrogenous base of the
lecithin molecule. Also, inositol is needed for the for-
CH3
mation of some cephalins.
(CH2)12
Specific Uses of Phospholipids. Several functions of the
CH
phospholipids are the following:
(1) Phospholipids
are an important constituent of lipoproteins in the
CH
blood and are essential for the formation and function
HO
C
H
O
of most of these; in their absence, serious abnormalities
of transport of cholesterol and other lipids can occur.
HC
NH
C
(CH2)16
CH3
(2) Thromboplastin, which is needed to initiate the clot-
ting process, is composed mainly of one of the cephalins.
O
CH
3
CH
(3) Large quantities of sphingomyelin are present in the
HC
O
P
O CH2
CH2
N+
nervous system; this substance acts as an electrical insu-
H
CH3
lator in the myelin sheath around nerve fibers. (4) Phos-
OH
pholipids are donors of phosphate radicals when these
Sphingomyelin
radicals are needed for different chemical reactions in
the tissues. (5) Perhaps the most important of all the
functions of phospholipids is participation in the for-
mation of structural elements—mainly membranes—in
Figure 68-4
cells throughout the body, as discussed in the next
Typical phospholipids.
section of this chapter in connection with a similar func-
tion for cholesterol.
formulas are shown in Figure
68-4. Phospholipids
Cholesterol
always contain one or more fatty acid molecules and
one phosphoric acid radical, and they usually contain a
Cholesterol, the formula of which is shown in Figure
nitrogenous base. Although the chemical structures of
68-5, is present in the diets of all people, and it can be
phospholipids are somewhat variant, their physical
absorbed slowly from the gastrointestinal tract into the
properties are similar because they are all lipid soluble,
intestinal lymph. It is highly fat soluble but only slightly
transported in lipoproteins, and used throughout the
soluble in water. It is specifically capable of forming
body for various structural purposes, such as in cell
esters with fatty acids. Indeed, about 70 per cent of the
membranes and intracellular membranes.
cholesterol in the lipoproteins of the plasma is in the
form of cholesterol esters.
Formation of Phospholipids. Phospholipids are synthesized
in essentially all cells of the body, although certain cells
Formation of Cholesterol. Besides the cholesterol absorbed
have a special ability to form great quantities of them.
each day from the gastrointestinal tract, which is called
Probably 90 per cent are formed in the liver cells; sub-
exogenous cholesterol, an even greater quantity is
stantial quantities are also formed by the intestinal
formed in the cells of the body, called endogenous cho-
epithelial cells during lipid absorption from the gut.
lesterol. Essentially all the endogenous cholesterol that
The rate of phospholipid formation is governed to
circulates in the lipoproteins of the plasma is formed by
some extent by the usual factors that control the overall
the liver, but all other cells of the body form at least
848
Unit XIII
Metabolism and Temperature Regulation
some cholesterol, which is consistent with the fact that
A large amount of cholesterol is precipitated in the
many of the membranous structures of all cells are par-
corneum of the skin. This, along with other lipids, makes
tially composed of this substance.
the skin highly resistant to the absorption of water-
The basic structure of cholesterol is a sterol nucleus.
soluble substances and to the action of many chemical
This is synthesized entirely from multiple molecules of
agents, because cholesterol and the other skin lipids are
acetyl-CoA. In turn, the sterol nucleus can be modified
highly inert to acids and to many solvents that might
by means of various side chains to form (1) cholesterol;
otherwise easily penetrate the body. Also, these lipid
(2) cholic acid, which is the basis of the bile acids formed
substances help prevent water evaporation from the
in the liver; and (3) many important steroid hormones
skin; without this protection, the amount of evaporation
secreted by the adrenal cortex, the ovaries, and the
can be 5 to 10 liters per day (as occurs in burn patients
testes (these hormones are discussed in later chapters).
who have lost their skin) instead of the usual 300 to
400 milliliters.
Factors That Affect Plasma Cholesterol Concentration—Feed-
back Control of Body Cholesterol. Among the important
Cellular Structural Functions of
factors that affect plasma cholesterol concentration are
Phospholipids and Cholesterol—
the following:
1.
An increase in the amount of cholesterol ingested
Especially for Membranes
each day increases the plasma concentration
The previously mentioned uses of phospholipids and
slightly. However, when cholesterol is ingested,
cholesterol are of only minor importance in comparison
the rising concentration of cholesterol inhibits the
with their function of forming specialized structures,
most essential enzyme for endogenous synthesis
mainly membranes, in all cells of the body. In
of cholesterol, 3-hydroxy-3-methylglutaryl CoA
Chapter 2, it was pointed out that large quantities of
reductase, thus providing an intrinsic feedback
phospholipids and cholesterol are present in both the
control system to prevent an excessive increase
cell membrane and the membranes of the internal
in plasma cholesterol concentration. As a result,
organelles of all cells. It is also known that the ratio
plasma cholesterol concentration usually is not
of membrane cholesterol to phospholipids is especially
changed upward or downward more than ±15 per
important in determining the fluidity of the cell
cent by altering the amount of cholesterol in the
membranes.
diet, although the response of individuals differs
For membranes to be formed, substances that are not
markedly.
soluble in water must be available. In general, the only
2.
A highly saturated fat diet increases blood
substances in the body that are not soluble in water
cholesterol concentration 15 to 25 per cent. This
(besides the inorganic substances of bone) are the lipids
results from increased fat deposition in the liver,
and some proteins. Thus, the physical integrity of cells
which then provides increased quantities of
everywhere in the body is based mainly on phospho-
acetyl-CoA in the liver cells for the production
lipids, cholesterol, and certain insoluble proteins. The
of cholesterol. Therefore, to decrease the blood
polar charges on the phospholipids also reduce the
cholesterol concentration, it is usually just as
interfacial tension between the cell membranes and
important, if not more important, to maintain a diet
the surrounding fluids.
low in saturated fat as to maintain a diet low in
Another fact that indicates the importance of phos-
cholesterol.
pholipids and cholesterol for the formation of structural
3.
Ingestion of fat containing highly unsaturated fatty
elements of the cells is the slow turnover rates of
acids usually depresses the blood cholesterol
these substances in most nonhepatic tissues—turnover
concentration a slight to moderate amount. The
rates measured in months or years. For instance, their
mechanism of this effect is unknown, despite the
function in brain cells to provide memory processes
fact that this observation is the basis of much
is related mainly to their indestructible physical
present-day dietary strategy.
properties.
4.
Lack of insulin or thyroid hormone increases the
blood cholesterol concentration, whereas excess
thyroid hormone decreases the concentration.
Atherosclerosis
These effects are probably caused mainly by
changes in the degree of activation of specific
Atherosclerosis is a disease of the large and intermedi-
enzymes responsible for the metabolism of lipid
ate-sized arteries in which fatty lesions called athero-
substances.
matous plaques develop on the inside surfaces of the
arterial walls. Arteriosclerosis, in contrast, is a general
Specific Uses of Cholesterol in the Body. By far the most
term that refers to thickened and stiffened blood vessels
abundant nonmembranous use of cholesterol in the
of all sizes.
body is to form cholic acid in the liver. As much as 80
One abnormality that can be measured very early
per cent of cholesterol is converted into cholic acid. As
in blood vessels that later become atherosclerotic is
explained in Chapter 70, this is conjugated with other
damage to the vascular endothelium. This, in turn,
substances to form bile salts, which promote digestion
increases the expression of adhesion molecules on
and absorption of fats.
endothelial cells and decreases their ability to release
A small quantity of cholesterol is used by (1) the
nitric oxide and other substances that help prevent
adrenal glands to form adrenocortical hormones, (2)
adhesion of macromolecules, platelets, and monocytes
the ovaries to form progesterone and estrogen, and (3)
to the endothelium. After damage to the vascular
the testes to form testosterone. These glands can
endothelium occurs, circulating monocytes and lipids
also synthesize their own sterols and then form hor-
(mostly low-density lipoproteins) begin to accumulate
mones from them, as discussed in the chapters on
at the site of injury (Figure 68-6A). The monocytes cross
endocrinology.
the endothelium, enter the intima of the vessel wall, and
Chapter 68
Lipid Metabolism
849
Blood monocyte
Monocyte
adhered to
Arterial
epithelium
lumen
Monocyte
migrating
into intima
Macrophage
foam cell
Damaged
Adhesion
endothelium
molecule
Growth/
inflammatory
Arterial
factors
intima
Receptor
Lipid
droplets
Lipoprotein
particle
A
Endothelium
Intima Media
Normal
Smooth
artery
muscle
cells
Adventitia
Small
plaque
Thrombosis
of a ruptured
plaque
Large
plaque
B
Figure 68-6
Development of atherosclerotic plaque. A, Attachment of a monocyte to an adhesion molecule on a damaged endothelial cell of an artery.
The monocyte then migrates through the endothelium into the intimal layer of the arterial wall and is transformed into a macrophage. The
macrophage then ingests and oxidizes lipoprotein molecules, becoming a macrophage foam cell. The foam cells release substances that
cause inflammation and growth of the intimal layer. B, Additional accumulation of macrophages and growth of the intima cause the plaque
to grow larger and accumulate lipids. Eventually, the plaque could occlude the vessel or rupture, causing the blood in the artery to coag-
ulate and form a thrombus. (Modified from Libby P: Inflammation in atherosclerosis. Nature 420:868, 2002.)
differentiate to become macrophages, which then
arterial wall. The lipid deposits plus the cellular prolif-
ingest and oxidize the accumulated lipoproteins,
eration can become so large that the plaque bulges into
giving the macrophages a foamlike appearance. These
the lumen of the artery and greatly reduces blood
macrophage foam cells then aggregate on the blood
flow, sometimes completely occluding the vessel. Even
vessel and form a visible fatty streak.
without occlusion, the fibroblasts of the plaque eventu-
With time, the fatty streaks grow larger and coalesce,
ally deposit extensive amounts of dense connective
and the surrounding fibrous and smooth muscle tissues
tissue; sclerosis
(fibrosis) becomes so great that the
proliferate to form larger and larger plaques (see Figure
arteries become stiff and unyielding. Still later, calcium
68-6B). Also, the macrophages release substances that
salts often precipitate with the cholesterol and other
cause inflammation and further proliferation of smooth
lipids of the plaques, leading to bony-hard calcifications
muscle and fibrous tissue on the inside surfaces of the
that can make the arteries rigid tubes. Both of these
850
Unit XIII
Metabolism and Temperature Regulation
later stages of the disease are called “hardening of the
high-density to low-density lipoproteins, the likelihood
arteries.”
of developing atherosclerosis is greatly reduced.
Atherosclerotic arteries lose most of their distensi-
bility, and because of the degenerative areas in their
Other Major Risk Factors
walls, they are easily ruptured. Also, where the plaques
protrude into the flowing blood, their rough surfaces
for Atherosclerosis
can cause blood clots to develop, with resultant throm-
In some people with perfectly normal levels of choles-
bus or embolus formation (see Chapter 36), leading to
terol and lipoproteins, atherosclerosis still develops.
a sudden blockage of all blood flow in the artery.
Some of the factors that are known to predispose to ath-
Almost half of all deaths in the United States and
erosclerosis are (1) physical inactivity and obesity, (2)
Europe are due to vascular disease. About two thirds of
diabetes mellitus, (3) hypertension, (4) hyperlipidemia,
these deaths are caused by thrombosis of one or more
and (5) cigarette smoking.
coronary arteries. The remaining one third are caused
Hypertension, for example, increases the risk for
by thrombosis or hemorrhage of vessels in other organs
atherosclerotic coronary artery disease by at least
of the body, especially the brain (causing strokes), but
twofold. Likewise, a person with diabetes mellitus has,
also the kidneys, liver, gastrointestinal tract, limbs, and
on average, more than a twofold increased risk of devel-
so forth.
oping coronary artery disease. When hypertension and
diabetes mellitus occur together, the risk for coronary
artery disease is increased by more than eightfold. And
Basic Causes of Atherosclerosis—
when hypertension, diabetes mellitus, and hyperlipemia
The Roles of Cholesterol
are all present, the risk for atherosclerotic coronary
and Lipoproteins
artery disease is increased almost 20-fold, suggesting
that these factors interact in a synergistic manner to
Increased Low-Density Lipoproteins. An important factor
increase the risk of developing atherosclerosis. In many
in causing atherosclerosis is a high blood plasma con-
overweight and obese patients, these three risk factors
centration of cholesterol in the form of low-density
do occur together, greatly increasing their risk for
lipoproteins. The plasma concentration of these high-
atherosclerosis, which in turn may lead to heart attack,
cholesterol low-density lipoproteins is increased by
stroke, and kidney disease.
several factors, including eating highly saturated fat in
In early and middle adulthood, men are more likely
the daily diet, obesity, and physical inactivity. To a lesser
to develop atherosclerosis than are women of compa-
extent, eating excess cholesterol may also raise plasma
rable age, suggesting that male sex hormones might be
levels of low-density lipoproteins.
atherogenic or, conversely, that female sex hormones
An interesting example occurs in rabbits, which
might be protective.
normally have low plasma cholesterol concentrations
Some of these factors cause atherosclerosis by
because of their vegetarian diet. Simply feeding these
increasing the concentration of low-density lipoproteins
animals large quantities of cholesterol as part of their
in the plasma. Others, such as hypertension, lead to
daily diet leads to serious atherosclerotic plaques
atherosclerosis by causing damage to the vascular en-
throughout their arterial systems.
dothelium and other changes in the vascular tissues that
predispose to cholesterol deposition.
Familial Hypercholesterolemia. This is a disease in which
To add to the complexity of atherosclerosis, experi-
the person inherits defective genes for the formation of
mental studies suggest that excess blood levels of iron
low-density lipoprotein receptors on the membrane sur-
can lead to atherosclerosis, perhaps by forming free rad-
faces of the body’s cells. In the absence of these recep-
icals in the blood that damage the vessel walls. About
tors, the liver cannot absorb either intermediate-density
one quarter of all people have a special type of low-
or low-density lipoproteins. Without this absorption, the
density lipoprotein called lipoprotein(a), containing an
cholesterol machinery of the liver cells goes on a
additional protein, apoprotein(a), that almost doubles
rampage, producing new cholesterol; it is no longer
the incidence of atherosclerosis. The precise mecha-
responsive to the feedback inhibition of too much
nisms of these atherogenic effects have yet to be
plasma cholesterol. As a result, the number of very low
discovered.
density lipoproteins released by the liver into the
plasma increases immensely.
Prevention of Atherosclerosis
Patients with full-blown familial hypercholes-
terolemia have blood cholesterol concentrations of 600
The most important measures to protect against the
to 1000 mg/dl, levels that are four to six times normal.
development of atherosclerosis and its progression to
Many of these people die before age 20 because of
serious vascular disease are (1) maintaining a healthy
myocardial infarction or other sequelae of atheroscle-
weight, being physically active, and eating a diet that
rotic blockage of blood vessels throughout the body.
contains mainly unsaturated fat with a low cholesterol
content; (2) preventing hypertension by maintaining a
Role of High-Density Lipoproteins in Preventing Atherosclerosis.
healthy diet and being physically active, or effectively
Much less is known about the function of high-density
controlling blood pressure with antihypertensive drugs
lipoproteins compared with that of low-density lipopro-
if hypertension does develop; (3) effectively controlling
teins. It is believed that high-density lipoproteins can
blood glucose with insulin treatment or other drugs if
actually absorb cholesterol crystals that are beginning
diabetes develops; and (4) avoiding cigarette smoking.
to be deposited in arterial walls. Whether this mecha-
Several types of drugs that lower plasma lipids and
nism is true or not, high-density lipoproteins do help
cholesterol have proved to be valuable in preventing
protect against the development of atherosclerosis.
atherosclerosis. Most of the cholesterol formed in the
Consequently, when a person has a high ratio of
liver is converted into bile acids and secreted in this
Chapter 68
Lipid Metabolism
851
form into the duodenum; then, more than 90 per cent of
Fromenty B, Robin MA, Igoudjil A, et al: The ins and outs
these same bile acids is reabsorbed in the terminal ileum
of mitochondrial dysfunction in NASH. Diabetes Metab
and used over and over again in the bile. Therefore, any
30:121, 2004.
agent that combines with the bile acids in the gastroin-
Gotto AM Jr, Brinton EA: Assessing low levels of high-
testinal tract and prevents their reabsorption into the
density lipoprotein cholesterol as a risk factor in coronary
circulation can decrease the total bile acid pool in the
heart disease: a working group report and update. J Am
circulating blood. This causes far more of the liver cho-
Coll Cardiol 43:717, 2004.
lesterol to be converted into new bile acids. Thus, simply
Grundy SM, Brewer HB Jr, Cleeman JI, et al: Definition of
eating oat bran, which binds bile acids and is a con-
metabolic syndrome: report of the National Heart, Lung,
stituent of many breakfast cereals, increases the pro-
and Blood Institute/American Heart Association confer-
portion of liver cholesterol that forms new bile acids
ence on scientific issues related to definition. Circulation
rather than forming new low-density lipoproteins and
109:433, 2004.
atherogenic plaques. Resin agents can also be used to
Haemmerle G, Zimmermann R, Zechner R: Letting lipids
bind bile acids in the gut and increase their fecal excre-
go: hormone-sensitive lipase. Curr Opin Lipidol 14:289,
tion, thereby reducing cholesterol synthesis by the liver.
2003.
Another group of drugs called statins competitively
Havel RJ, Hamilton RL: Hepatic catabolism of remnant
inhibits hydroxymethylglutaryl-coenzyme A
(HMG-
lipoproteins: where the action is. Arterioscler Thromb Vasc
CoA) reductase, a rate-limiting enzyme in the synthesis
Biol 24:213, 2004.
of cholesterol. This inhibition decreases cholesterol syn-
Hilgemann DW: Getting ready for the decade of the lipids.
thesis and increases low-density lipoprotein receptors in
Annu Rev Physiol 65:697, 2003.
the liver, usually causing a 25 to 50 per cent reduction
Horton JD, Goldstein JL, Brown MS: SREBPs: activators of
in plasma levels of low-density lipoproteins. The statins
the complete program of cholesterol and fatty acid syn-
may also have other beneficial effects that help prevent
thesis in the liver. J Clin Invest 109:1125, 2002.
atherosclerosis, such as attenuating vascular inflamma-
LaRosa JC, Gotto AM Jr: Past, present, and future standards
tion. These drugs are now widely used to treat patients
for management of dyslipidemia. Am J Med 116(Suppl
who have increased plasma cholesterol levels.
6A):3S, 2004.
In general, studies show that for each
1 mg/dl
Libby P: Inflammation in atherosclerosis. Nature 420:868,
decrease in low-density lipoprotein cholesterol in the
2002.
plasma, there is about a 2 per cent decrease in mortal-
Osterud B, Bjorklid E: Role of monocytes in atherogenesis.
ity from atherosclerotic heart disease. Therefore, appro-
Physiol Rev 83:1069, 2003.
priate preventive measures are valuable in decreasing
Rinaldo P, Matern D, Bennett MJ: Fatty acid oxidation dis-
heart attacks.
orders. Annu Rev Physiol 64:477, 2002.
Roden M: How free fatty acids inhibit glucose utilization in
human skeletal muscle. News Physiol Sci 19:92, 2004.
References
Ruderman NB, Saha AK, Kraegen EW: Malonyl CoA, AMP-
activated protein kinase, and adiposity. Endocrinology
144:5166, 2003.
Assmann G, Nofer JR: Atheroprotective effects of high-
Schonbeck U, Libby P: Inflammation, immunity, and HMG-
density lipoproteins. Annu Rev Med 54:321, 2003.
CoA reductase inhibitors: statins as antiinflammatory
Brown MS, Goldstein JL: A proteolytic pathway that con-
agents? Circulation 109(21 Suppl 1):II18, 2004.
trols the cholesterol content of membranes, cells, and
Spriet LL, Watt MJ: Regulatory mechanisms in the interac-
blood. Proc Natl Acad Sci U S A 96:11041, 1999.
tion between carbohydrate and lipid oxidation during
Casserly I, Topol E: Convergence of atherosclerosis and
exercise. Acta Physiol Scand 178:443, 2003.
Alzheimer’s disease: inflammation, cholesterol, and mis-
Unger RH: The physiology of cellular liporegulation. Annu
folded proteins. Lancet 363:1139, 2004.
Rev Physiol 65:333, 2003.
Fielding BA, Frayn KN: Lipid metabolism. Curr Opin
Lipidol 14:389, 2003.
C H A P T E R
6
9
Protein Metabolism
About three quarters of the body solids are proteins.
These include structural proteins, enzymes, nucleopro-
teins, proteins that transport oxygen, proteins of the
muscle that cause muscle contraction, and many other
types that perform specific intracellular and extracellu-
lar functions throughout the body.
The basic chemical properties that explain proteins’
diverse functions are so extensive that they
constitute a major portion of the entire discipline of
biochemistry. For this reason, the current discussion is confined to a few specific
aspects of protein metabolism that are important as background for other discus-
sions in this text.
Basic Properties
Amino Acids
The principal constituents of proteins are amino acids, 20 of which are present in
the body proteins in significant quantities. Figure 69-1 shows the chemical formu-
las of these 20 amino acids, demonstrating that they all have two features in
common: each amino acid has an acidic group (—COOH) and a nitrogen atom
attached to the molecule, usually represented by the amino group (—NH2).
Peptide Linkages and Peptide Chains. In proteins, the amino acids are aggregated into
long chains by means of peptide linkages. The chemical nature of this linkage is
demonstrated by the following reaction:
NH2
H NH
R CH CO OH + R' CH COOH
NH2
R CH CO
NH + H2O
R'
CH COOH
Note in this reaction that the nitrogen of the amino radical of one amino acid
bonds with the carbon of the carboxyl radical of the other amino acid. A hydrogen
ion is released from the amino radical, and a hydroxyl ion is released from the car-
boxyl radical; these two combine to form a molecule of water. After the peptide
linkage has been formed, an amino radical and a carboxyl radical are still at oppo-
site ends of the new, longer molecule. Each of these radicals is capable of combin-
ing with additional amino acids to form a peptide chain. Some complicated protein
molecules have many thousand amino acids combined by peptide linkages, and even
the smallest protein molecule usually has more than 20 amino acids combined by
peptide linkages. The average is about 400 amino acids.
Other Linkages in Protein Molecules. Some protein molecules are composed of several
peptide chains rather than a single chain, and these chains are bound to one another
852
Chapter 69
Protein Metabolism
853
AMINO ACIDS
Glycine
Proline
H
H2C
CH2
H
C
COOH
H2C
C COOH
NH2
N
H
Alanine
H
H H
ESSENTIAL AMINO ACIDS
H
C
C
COOH
THREONINE
LYSINE
H NH2
H H
NH
H H
H
H
H
2
Serine
H H
H C C
C
COOH
H
C C
C
C C
COOH
H
C
C
COOH
H
OH H
NH2
H
H
H
NH2
OH NH2
METHIONINE
ARGININE
Cysteine
H H
H
NH H
H
H
H
H
H H
CH
S C C C
COOH
H
C
N
C
C C
C
COOH
H
C
C
COOH
3
2N
H
H
NH2
H
H H
NH2
SH NH2
Aspartic Acid
VALINE
PHENYLALANINE
COOH
H
H
H
H C NH2
H
C
H H
C C COOH
H C H
H
C C
COOH
H
NH2
H
COOH
NH2
Glutamic Acid
H
C
COOH
H
H C NH2
LEUCINE
TRYPTOPHAN
H C H
H
H
H
H C H
H
C
C
C
C COOH
H H
H
COOH
H
CH
H
NH2
Asparagine
C C
C COOH
H
N
O H NH2
H
NH2
H
C
H
NH2
C
C
C
COOH
H
H H
Glutamine
ISOLEUCINE
HISTIDINE
O H H
NH2
H H H
H
HC N
NH2
C
C C C
COOH
H C C C
C COOH
CH
H
H H
H
H
CH3
NH2
C N H
Tyrosine
H C H
H
H
H C
NH2
HO
C C COOH
COOH
H NH2
Figure 69-1
Amino acids. The 10 essential amino acids cannot be synthesized in sufficient quantities in the body; these essential amino acids must
be obtained, already formed, from food.
854
Unit XIII
Metabolism and Temperature Regulation
by other linkages, often by hydrogen bonding between
Renal Threshold for Amino Acids. In the kidneys, the
the CO and NH radicals of the peptides, as follows:
different amino acids can be actively reabsorbed
through the proximal tubular epithelium, which
removes them from the glomerular filtrate and returns
them to the blood if they should filter into the renal
C
O
H N
tubules through the glomerular membranes. However,
as is true of other active transport mechanisms in the
R
HC
CH R'
renal tubules, there is an upper limit to the rate at which
each type of amino acid can be transported. For this
reason, when the concentration of a particular type
N H
O C
of amino acid becomes too high in the plasma and
glomerular filtrate, the excess that cannot be actively
reabsorbed is lost into the urine.
Many peptide chains are coiled or folded, and the suc-
cessive coils or folds are held in a tight spiral or in other
shapes by similar hydrogen bonding and other forces.
Storage of Amino Acids as Proteins
in the Cells
Transport and Storage
Almost immediately after entry into tissue cells, amino
acids combine with one another by peptide linkages,
of Amino Acids
under the direction of the cell’s messenger RNA and
ribosomal system, to form cellular proteins. Therefore,
Blood Amino Acids
the concentration of free amino acids inside the cells
The normal concentration of amino acids in the blood
usually remains low. Thus, storage of large quantities
is between 35 and 65 mg/dl. This is an average of about
of free amino acids does not occur in the cells; instead,
2 mg/dl for each of the 20 amino acids, although some
they are stored mainly in the form of actual proteins.
are present in far greater amounts than others. Because
But many of these intracellular proteins can be rapidly
the amino acids are relatively strong acids, they exist
decomposed again into amino acids under the influence
in the blood principally in the ionized state, resulting
of intracellular lysosomal digestive enzymes; these
from the removal of one hydrogen atom from the NH2
amino acids can then be transported back out of the
radical. They actually account for 2 to 3 milliequivalents
cell into the blood. Special exceptions to this reversal
of the negative ions in the blood. The precise distribu-
process are the proteins in the chromosomes of the
tion of the different amino acids in the blood depends
nucleus and the structural proteins such as collagen
to some extent on the types of proteins eaten, but
and muscle contractile proteins; these proteins do not
the concentrations of at least some individual amino
participate significantly in this reverse digestion and
acids are regulated by selective synthesis in the differ-
transport back out of the cells.
ent cells.
Some tissues of the body participate in the storage
of amino acids to a greater extent than others. For
instance, the liver, which is a large organ and has special
Fate of Amino Acids Absorbed from the Gastrointestinal Tract.
systems for processing amino acids, can store large
The products of protein digestion and absorption in the
quantities of rapidly exchangeable proteins; this is also
gastrointestinal tract are almost entirely amino acids;
true to a lesser extent of the kidneys and the intestinal
only rarely are polypeptides or whole protein molecules
mucosa.
absorbed from the digestive tract into the blood. Imme-
diately after a meal, the amino acid concentration in a
Release of Amino Acids from the Cells as a Means of Regulating
person’s blood rises, but the increase is usually only a
Plasma Amino Acid Concentration. Whenever plasma amino
few milligrams per deciliter, for two reasons: First,
acid concentrations fall below normal levels, the
protein digestion and absorption are usually extended
required amino acids are transported out of the cells to
over 2 to 3 hours, which allows only small quantities of
replenish their supply in the plasma. In this way, the
amino acids to be absorbed at a time. Second, after
plasma concentration of each type of amino acid is main-
entering the blood, the excess amino acids are absorbed
tained at a reasonably constant value. Later, it is noted
within 5 to 10 minutes by cells throughout the body,
that some of the hormones secreted by the endocrine
especially by the liver. Therefore, almost never do large
glands are able to alter the balance between tissue pro-
concentrations of amino acids accumulate in the blood
teins and circulating amino acids. For instance, growth
and tissue fluids. Nevertheless, the turnover rate of the
hormone and insulin increase the formation of tissue
amino acids is so rapid that many grams of proteins can
proteins, whereas adrenocortical glucocorticoid hor-
be carried from one part of the body to another in the
mones increase the concentration of plasma amino acids.
form of amino acids each hour.
Reversible Equilibrium Between the Proteins in Different Parts of
Active Transport of Amino Acids into the Cells. The molecules
the Body. Because cellular proteins in the liver (and, to
of all the amino acids are much too large to diffuse
a much less extent, in other tissues) can be synthesized
readily through the pores of the cell membranes. There-
rapidly from plasma amino acids, and because many of
fore, significant quantities of amino acids can move
these proteins can be degraded and returned to the
either inward or outward through the membranes only
plasma almost as rapidly, there is constant interchange
by facilitated transport or active transport using carrier
and equilibrium between the plasma amino acids and
mechanisms. The nature of some of the carrier mecha-
labile proteins in virtually all cells of the body. For
nisms is still poorly understood, but a few are discussed
instance, if any particular tissue requires proteins, it can
in Chapter 4.
synthesize new proteins from the amino acids of the
Chapter 69
Protein Metabolism
855
blood; in turn, the blood amino acids are replenished by
throughout the body to build cellular proteins wherever
degradation of proteins from other cells of the body,
needed. In this way, the plasma proteins function as a
especially from the liver cells. These effects are particu-
labile protein storage medium and represent a readily
larly noticeable in relation to protein synthesis in cancer
available source of amino acids whenever a particular
cells. Cancer cells are often prolific users of amino acids;
tissue requires them.
therefore, the proteins of the other cells can become
markedly depleted.
Reversible Equilibrium Between the Plasma Proteins and the
Tissue Proteins. There is a constant state of equilibrium,
Upper Limit for the Storage of Proteins. Each particular type
as shown in Figure 69-2, among the plasma proteins, the
of cell has an upper limit with regard to the amount of
amino acids of the plasma, and the tissue proteins. It has
proteins it can store. After all the cells have reached
been estimated from radioactive tracer studies that
their limits, the excess amino acids still in the circulation
normally about 400 grams of body protein are synthe-
are degraded into other products and used for energy,
sized and degraded each day as part of the continual
as discussed subsequently, or they are converted to fat
state of flux of amino acids. This demonstrates the
or glycogen and stored in these forms.
general principle of reversible exchange of amino acids
among the different proteins of the body. Even during
starvation or severe debilitating diseases, the ratio of
Functional Roles of the
total tissue proteins to total plasma proteins in the body
remains relatively constant at about 33:1.
Plasma Proteins
Because of this reversible equilibrium between
The major types of protein present in the plasma are
plasma proteins and the other proteins of the body, one
albumin, globulin, and fibrinogen.
of the most effective therapies for severe, acute whole-
A major function of albumin is to provide colloid
body protein deficiency is intravenous transfusion of
osmotic pressure in the plasma, which prevents plasma
plasma protein. Within a few days, or sometimes within
loss from the capillaries, as discussed in Chapter 16.
hours, the amino acids of the administered protein are
The globulins perform a number of enzymatic func-
distributed throughout the cells of the body to form new
tions in the plasma, but equally important, they are prin-
proteins where they are needed.
cipally responsible for the body’s both natural and
acquired immunity against invading organisms, dis-
Essential and Nonessential
cussed in Chapter 34.
Amino Acids
Fibrinogen polymerizes into long fibrin threads
during blood coagulation, thereby forming blood clots
Ten of the amino acids normally present in animal pro-
that help repair leaks in the circulatory system, dis-
teins can be synthesized in the cells, whereas the other
cussed in Chapter 36.
10 either cannot be synthesized or are synthesized in
quantities too small to supply the body’s needs. This
Formation of the Plasma Proteins. Essentially all the
second group of amino acids that cannot be synthesized
albumin and fibrinogen of the plasma proteins, as well
is called the essential amino acids. Use of the word
as 50 to 80 per cent of the globulins, are formed in the
“essential” does not mean that the other 10 “nonessen-
liver. The remainder of the globulins are formed almost
tial” amino acids are not required for the formation of
entirely in the lymphoid tissues. They are mainly the
proteins, but only that the others are not essential in the
gamma globulins that constitute the antibodies used in
diet because they can be synthesized in the body.
the immune system.
Synthesis of the nonessential amino acids depends
The rate of plasma protein formation by the liver can
mainly on the formation of appropriate a-keto acids,
be extremely high, as much as 30 g/day. Certain disease
conditions cause rapid loss of plasma proteins; severe
burns that denude large surface areas of the skin can
Tissue cells
Liver cells
cause the loss of several liters of plasma through the
denuded areas each day. The rapid production of plasma
proteins by the liver is valuable in preventing death in
such states. Occasionally, a person with severe renal
Proteins
Amino
Proteins
disease loses as much as 20 grams of plasma protein in
Amino acids
acids
the urine each day for months, and it is continually
replaced mainly by liver production of the required
proteins.
Amino acids
Blood
Plasma proteins
In cirrhosis of the liver, large amounts of fibrous tissue
develop among the liver parenchymal cells, causing a
reduction in their ability to synthesize plasma proteins.
As discussed in Chapter 25, this leads to decreased
plasma colloid osmotic pressure, which causes general-
Imbibed plasma protein
ized edema.
Plasma Proteins as a Source of Amino Acids for the Tissues.
When the tissues become depleted of proteins, the
Reticuloendothelial cell
plasma proteins can act as a source of rapid replace-
ment. Indeed, whole plasma proteins can be imbibed in
Figure 69-2
toto by tissue macrophages through the process of
pinocytosis; once in these cells, they are split into amino
Reversible equilibrium among the tissue proteins, plasma pro-
acids that are transported back into the blood and used
teins, and plasma amino acids.
856
Unit XIII
Metabolism and Temperature Regulation
NH2
C
CH2
CH2
CH COOH
CH3
C COOH
Transaminase
O
NH
+
O
2
(Glutamine)
(Pyruvic acid)
NH2
C
CH2
CH2
C
COOH
CH3
C COOH
O
O
+
NH
Figure 69-3
(a-Ketoglutamic acid)
(Alanine)
Synthesis of alanine from pyruvic
acid by transamination.
which are the precursors of the respective amino acids.
Note from this schema that the amino group from the
For instance, pyruvic acid, which is formed in large
amino acid is transferred to a-ketoglutaric acid, which
quantities during the glycolytic breakdown of glucose,
then becomes glutamic acid. The glutamic acid can then
is the keto acid precursor of the amino acid alanine.
transfer the amino group to still other substances or
Then, by the process of transamination, an amino
release it in the form of ammonia (NH3). In the process
radical is transferred to the a-keto acid, and the keto
of losing the amino group, the glutamic acid once again
oxygen is transferred to the donor of the amino radical.
becomes a-ketoglutaric acid, so that the cycle can be
This reaction is shown in Figure 69-3. Note in this figure
repeated again and again. To initiate this process, the
that the amino radical is transferred to the pyruvic acid
excess amino acids in the cells, especially in the liver,
from another chemical that is closely allied to the amino
induce the activation of large quantities of aminotrans-
acids, glutamine. Glutamine is present in the tissues in
ferases, the enzymes responsible for initiating most
large quantities, and one of its principal functions is to
deamination.
serve as an amino radical storehouse. In addition, amino
radicals can be transferred from asparagine, glutamic
Urea Formation by the Liver. The ammonia released
acid, and aspartic acid.
during deamination of amino acids is removed from the
Transamination is promoted by several enzymes,
blood almost entirely by conversion into urea; two mol-
among which are the aminotransferases, which are deriv-
ecules of ammonia and one molecule of carbon dioxide
atives of pyridoxine, one of the B vitamins (B6). Without
combine in accordance with the following net reaction:
this vitamin, the amino acids are synthesized only poorly,
2 NH3 + CO2 Æ H2N—C—NH2 + H2O
and protein formation cannot proceed normally.
||
O
Use of Proteins for Energy
Essentially all urea formed in the human body is syn-
thesized in the liver. In the absence of the liver or in
Once the cells are filled to their limits with stored
serious liver disease, ammonia accumulates in the
protein, any additional amino acids in the body fluids are
blood. This is extremely toxic, especially to the brain,
degraded and used for energy or are stored mainly as fat
often leading to a state called hepatic coma.
or secondarily as glycogen. This degradation occurs
The stages in the formation of urea are essentially the
almost entirely in the liver, and it begins with deamina-
following:
tion, which is explained in the following section.
Ornithine + CO2 + NH3
Deamination. Deamination means removal of the amino
Citrulline
groups from the amino acids. This occurs mainly by
-H2O
transamination, which means transfer of the amino
NH3
group to some acceptor substance, which is the reverse
-H2O
of the transamination explained earlier in relation to the
synthesis of amino acids.
Arginine
The greatest amount of deamination occurs by the
following transamination schema:
(Arginase)
+ H2O
Urea
a-Ketoglutaric acid + Amino acid
After its formation, the urea diffuses from the liver cells
into the body fluids and is excreted by the kidneys.
Glutamic acid + a-Keto acid
Oxidation of Deaminated Amino Acids. Once amino acids
have been deaminated, the resulting keto acids can, in
most instances, be oxidized to release energy for meta-
bolic purposes. This usually involves two successive
+ NAD+ + H2O
processes: (1) the keto acid is changed into an appro-
priate chemical substance that can enter the citric acid
NADH + H+ + NH3
cycle, and (2) this substance is degraded by the cycle and
Chapter 69
Protein Metabolism
857
used for energy in the same manner that acetyl coen-
Hormonal Regulation
zyme A (acetyl-CoA) derived from carbohydrate and
of Protein Metabolism
lipid metabolism is used, as explained in Chapters 67
and 68. In general, the amount of adenosine triphos-
Growth Hormone Increases the Synthesis of Cellular Proteins.
phate (ATP) formed for each gram of protein that is
Growth hormone causes the tissue proteins to increase.
oxidized is slightly less than that formed for each gram
The precise mechanism by which this occurs is not
of glucose oxidized.
known, but it is believed to result mainly from increased
transport of amino acids through the cell membranes or
acceleration of the DNA and RNA transcription and
Gluconeogenesis and Ketogenesis. Certain deaminated
translation processes for protein synthesis.
amino acids are similar to the substrates normally used
by the cells, mainly the liver cells, to synthesize glucose
Insulin Is Necessary for Protein Synthesis. Total lack of
or fatty acids. For instance, deaminated alanine is
insulin reduces protein synthesis to almost zero. The
pyruvic acid. This can be converted into either glucose
mechanism by which this occurs is also unknown, but
or glycogen. Alternatively, it can be converted into
insulin does accelerate the transport of some amino
acetyl-CoA, which can then be polymerized into fatty
acids into cells, which could be the stimulus to protein
acids. Also, two molecules of acetyl-CoA can condense
synthesis. Also, insulin increases the availability of
to form acetoacetic acid, which is one of the ketone
glucose to the cells, so that the need for amino acids for
bodies, as explained in Chapter 68.
energy is correspondingly reduced.
The conversion of amino acids into glucose or glyco-
gen is called gluconeogenesis, and the conversion of
Glucocorticoids Increase Breakdown of Most Tissue Proteins.
amino acids into keto acids or fatty acids is called keto-
The glucocorticoids secreted by the adrenal cortex
genesis. Of the 20 deaminated amino acids, 18 have
decrease the quantity of protein in most tissues while
chemical structures that allow them to be converted into
increasing the amino acid concentration in the plasma,
glucose, and 19 of them can be converted into fatty
as well as increasing both liver proteins and plasma pro-
acids.
teins. It is believed that the glucocorticoids act by
increasing the rate of breakdown of extrahepatic pro-
teins, thereby making increased quantities of amino
acids available in the body fluids. This supposedly allows
Obligatory Degradation of Proteins
the liver to synthesize increased quantities of hepatic
When a person eats no proteins, a certain proportion of
cellular proteins and plasma proteins.
body proteins is degraded into amino acids and then
deaminated and oxidized. This involves 20 to 30 grams
Testosterone Increases Protein Deposition in Tissues. Testos-
of protein each day, which is called the obligatory
terone, the male sex hormone, causes increased deposi-
loss of proteins. Therefore, to prevent net loss of
tion of protein in tissues throughout the body, especially
protein from the body, one must ingest a minimum
the contractile proteins of the muscles (30 to 50 per cent
of 20 to 30 grams of protein each day; to be on the
increase). The mechanism of this effect is unknown, but
safe side, a minimum of 60 to 75 grams is usually
it is definitely different from the effect of growth
recommended.
hormone, in the following way: Growth hormone causes
The ratios of the different amino acids in the dietary
tissues to continue growing almost indefinitely, whereas
protein must be about the same as the ratios in the body
testosterone causes the muscles and, to a much lesser
tissues if the entire dietary protein is to be fully usable
extent, some other protein tissues to enlarge for only
to form new proteins in the tissues. If one particular type
several months. Once the muscles and other protein
of essential amino acid is low in concentration, the
tissues have reached a maximum, despite continued
others become unusable because cells synthesize either
administration of testosterone, further protein deposi-
whole proteins or none at all, as explained in Chapter 3
tion ceases.
in relation to protein synthesis. The unusable amino
acids are deaminated and oxidized. A protein that has
Estrogen. Estrogen, the principal female sex hormone,
a ratio of amino acids different from that of the average
also causes some deposition of protein, but its effect
body protein is called a partial protein or incomplete
is relatively insignificant in comparison with that of
protein, and such a protein is less valuable for nutrition
testosterone.
than is a complete protein.
Thyroxine. Thyroxine increases the rate of metabolism of
all cells and, as a result, indirectly affects protein metab-
Effect of Starvation on Protein Degradation. Except for the 20
olism. If insufficient carbohydrates and fats are avail-
to 30 grams of obligatory protein degradation each day,
able for energy, thyroxine causes rapid degradation of
the body uses almost entirely carbohydrates or fats
proteins and uses them for energy. Conversely, if ade-
for energy, as long as they are available. However,
quate quantities of carbohydrates and fats are available
after several weeks of starvation, when the quantities
and excess amino acids are also available in the extra-
of stored carbohydrates and fats begin to run out, the
cellular fluid, thyroxine can actually increase the rate of
amino acids of the blood are rapidly deaminated and
protein synthesis. In growing animals or human beings,
oxidized for energy. From this point on, the proteins
deficiency of thyroxine causes growth to be greatly
of the tissues degrade rapidly—as much as 125 grams
inhibited because of lack of protein synthesis. In
daily—and, as a result, cellular functions deteriorate
essence, it is believed that thyroxine has little specific
precipitously. Because carbohydrate and fat utilization
effect on protein metabolism but does have an
for energy normally occurs in preference to protein
important general effect by increasing the rates of
utilization, carbohydrates and fats are called protein
both normal anabolic and normal catabolic protein
sparers.
reactions.
858
Unit XIII
Metabolism and Temperature Regulation
References
Kuhn CM: Anabolic steroids. Recent Prog Horm Res 57:411,
2002.
Layman DK, Baum JI: Dietary protein impact on glycemic
Altenberg GA: The engine of ABC proteins. News Physiol
control during weight loss. J Nutr 134:968S, 2004.
Sci 18:191, 2003.
Mann GE, Yudilevich DL, Sobrevia L: Regulation of amino
Caldwell J: Pharmacogenetics and individual variation in
acid and glucose transporters in endothelial and smooth
the range of amino acid adequacy: the biological aspects.
muscle cells. Physiol Rev 83:183, 2003.
J Nutr 134(6 Suppl):1600S, 2004.
Meijer AJ: Amino acids as regulators and components of
Daniel H: Molecular and integrative physiology of intestinal
nonproteinogenic pathways. J Nutr 133(6 Suppl 1):2057S,
peptide transport. Annu Rev Physiol 66:361, 2004.
2003.
Deves R, Boyd CA: Transporters for cationic amino acids in
Moriwaki H, Miwa Y, Tajika M, et al: Branched-chain amino
animal cells: discovery, structure, and function. Physiol
acids as a protein- and energy-source in liver cirrhosis.
Rev 78:487, 1998.
Biochem Biophys Res Commun 313:405, 2004.
Fukagawa NK, Galbraith RA: Advancing age and other
Pencharz PB, Ball RO: Amino acid needs for early growth
factors influencing the balance between amino acid
and development. J Nutr 134(6 Suppl):1566S, 2004.
requirements and toxicity. J Nutr
134(6 Suppl):1569S,
Prod’homme M, Rieu I, Balage M, et al: Insulin and amino
2004.
acids both strongly participate to the regulation of protein
Jans DA, Hubner S: Regulation of protein transport to the
metabolism. Curr Opin Clin Nutr Metab Care 7:71, 2004.
nucleus: central role of phosphorylation. Physiol Rev
Tessari P: Protein metabolism in liver cirrhosis: from albumin
76:651, 1996.
to muscle myofibrils. Curr Opin Clin Nutr Metab Care
Kadowaki M, Kanazawa T: Amino acids as regulators of pro-
6:79, 2003.
teolysis. J Nutr 133(6 Suppl 1):2052S, 2003.
van de Poll MC, Soeters PB, Deutz NE, et al: Renal metab-
Kimball SR, Jefferson LS: Regulation of global and specific
olism of amino acids: its role in interorgan amino acid
mRNA translation by oral administration of branched-
exchange. Am J Clin Nutr 79:185, 2004.
chain amino acids. Biochem Biophys Res Commun
313:423, 2004.
C H A P T E R
7
0
The Liver as an Organ
The liver performs many different functions yet is also
a discrete organ, and many of its functions interrelate
with one another. This becomes especially evident in
abnormalities of the liver, because many of its functions
are disturbed simultaneously. The purpose of this
chapter is to summarize the liver’s different functions,
including
(1) filtration and storage of blood;
(2)
metabolism of carbohydrates, proteins, fats, hormones,
and foreign chemicals; (3) formation of bile; (4) storage
of vitamins and iron; and (5) formation of coagulation factors.
Physiologic Anatomy of the Liver
The liver is the largest organ in the body, contributing about 2 per cent of the total
body weight, or about 1.5 kg in the average adult human. The basic functional unit
of the liver is the liver lobule, which is a cylindrical structure several millimeters in
length and 0.8 to 2 millimeters in diameter. The human liver contains 50,000 to
100,000 individual lobules.
The liver lobule, shown in cut-away format in Figure 70-1, is constructed around
a central vein that empties into the hepatic veins and then into the vena cava. The
lobule itself is composed principally of many liver cellular plates (two of which are
shown in Figure 70-1) that radiate from the central vein like spokes in a wheel. Each
hepatic plate is usually two cells thick, and between the adjacent cells lie small bile
canaliculi that empty into bile ducts in the fibrous septa separating the adjacent liver
lobules.
In the septa are small portal venules that receive their blood mainly from the
venous outflow of the gastrointestinal tract by way of the portal vein. From these
venules blood flows into flat, branching hepatic sinusoids that lie between the
hepatic plates and then into the central vein. Thus, the hepatic cells are exposed con-
tinuously to portal venous blood.
Hepatic arterioles are also present in the interlobular septa. These arterioles
supply arterial blood to the septal tissues between the adjacent lobules, and many
of the small arterioles also empty directly into the hepatic sinusoids, most frequently
emptying into those located about one third the distance from the interlobular
septa, as shown in Figure 70-1.
In addition to the hepatic cells, the venous sinusoids are lined by two other types
of cell: (1) typical endothelial cells and (2) large Kupffer cells (also called reticu-
loendothelial cells), which are resident macrophages that line the sinusoids and are
capable of phagocytizing bacteria and other foreign matter in the hepatic sinus
blood.
The endothelial lining of the sinusoids has extremely large pores, some of which
are almost 1 micrometer in diameter. Beneath this lining, lying between the
endothelial cells and the hepatic cells, are narrow tissue spaces called the spaces of
Disse, also known as the perisinusoidal spaces. The millions of spaces of Disse
connect with lymphatic vessels in the interlobular septa. Therefore, excess fluid in
these spaces is removed through the lymphatics. Because of the large pores in the
endothelium, substances in the plasma move freely into the spaces of Disse. Even
large portions of the plasma proteins diffuse freely into these spaces.
Hepatic Vascular and Lymph Systems
The function of the hepatic vascular system is discussed in Chapter 15 in connec-
tion with the portal veins and can be summarized as follows.
859
860
Unit XIII
Metabolism and Temperature Regulation
Central
The Liver Functions as
vein
a Blood Reservoir
Because the liver is an expandable organ, large quanti-
Liver cell plate
ties of blood can be stored in its blood vessels. Its
Sinusoids
normal blood volume, including both that in the hepatic
Space of Disse
Kupffer cell
veins and that in the hepatic sinuses, is about 450 milli-
Terminal
Bile canaliculi
liters, or almost 10 per cent of the body’s total blood
lymphatics
volume. When high pressure in the right atrium causes
backpressure in the liver, the liver expands, and 0.5 to 1
Lymphatic
liter of extra blood is occasionally stored in the hepatic
duct
Portal
veins and sinuses. This occurs especially in cardiac
vein
failure with peripheral congestion, which is discussed in
Hepatic
Chapter 22. Thus, in effect, the liver is a large, expand-
artery
able, venous organ capable of acting as a valuable blood
reservoir in times of excess blood volume and capable
Bile duct
of supplying extra blood in times of diminished blood
volume.
Figure 70-1
Basic structure of a liver lobule, showing the liver cellular plates,
The Liver Has Very High
the blood vessels, the bile-collecting system, and the lymph flow
system composed of the spaces of Disse and the interlobular
Lymph Flow
lymphatics. (Modified from Guyton AC, Taylor AE, Granger HJ:
Circulatory Physiology. Vol 2: Dynamics and Control of the Body
Because the pores in the hepatic sinusoids are very
Fluids. Philadelphia: WB Saunders, 1975.)
permeable and allow ready passage of both fluid and
proteins into the spaces of Disse, the lymph draining
from the liver usually has a protein concentration of
about 6 g/dl, which is only slightly less than the protein
Blood Flows Through the Liver from
concentration of plasma. Also, the extreme permeabil-
ity of the liver sinusoid epithelium allows large quanti-
the Portal Vein and Hepatic Artery
ties of lymph to form. Therefore, about half of all the
The Liver Has High Blood Flow and Low Vascular Resistance.
lymph formed in the body under resting conditions
About 1050 milliliters of blood flows from the portal
arises in the liver.
vein into the liver sinusoids each minute, and an addi-
tional 300 milliliters flows into the sinusoids from the
High Hepatic Vascular Pressures Can Cause Fluid Transudation
hepatic artery, the total averaging about 1350 ml/min.
into the Abdominal Cavity from the Liver and Portal Capillaries—
This amounts to 27 per cent of the resting cardiac
Ascites. When the pressure in the hepatic veins rises
output.
only 3 to 7 mm Hg above normal, excessive amounts of
The pressure in the portal vein leading into the liver
fluid begin to transude into the lymph and leak through
averages about
9 mm Hg, and the pressure in the
the outer surface of the liver capsule directly into the
hepatic vein leading from the liver into the vena cava
abdominal cavity. This fluid is almost pure plasma, con-
normally averages almost exactly 0 mm Hg. This small
taining 80 to 90 per cent as much protein as normal
pressure difference, only 9 mm Hg, shows that the resist-
plasma. At vena caval pressures of 10 to 15 mm Hg,
ance to blood flow through the hepatic sinusoids is nor-
hepatic lymph flow increases to as much as 20 times
mally very low, especially when one considers that about
normal, and the “sweating” from the surface of the liver
1350 milliliters of blood flows by this route each minute.
can be so great that it causes large amounts of free fluid
in the abdominal cavity, which is called ascites. Block-
Cirrhosis of the Liver Greatly Increases Resistance to Blood Flow.
age of portal flow through the liver also causes high
When liver parenchymal cells are destroyed, they are
capillary pressures in the entire portal vascular system
replaced with fibrous tissue that eventually contracts
of the gastrointestinal tract, resulting in edema of
around the blood vessels, thereby greatly impeding the
the gut wall and transudation of fluid through the serosa
flow of portal blood through the liver. This disease
of the gut into the abdominal cavity. This, too, can cause
process is known as cirrhosis of the liver. It results most
ascites.
commonly from alcoholism, but it can also follow inges-
tion of poisons such as carbon tetrachloride, viral dis-
eases such as infectious hepatitis, obstruction of the bile
Regulation of Liver Mass—
ducts, and infectious processes in the bile ducts.
Regeneration
The portal system is also occasionally blocked by a
large clot that develops in the portal vein or its major
The liver possesses a remarkable ability to restore itself
branches. When the portal system is suddenly blocked,
after significant hepatic tissue loss from either partial
the return of blood from the intestines and spleen
hepatectomy or acute liver injury, as long as the injury
through the liver portal blood flow system to the sys-
is uncomplicated by viral infection or inflammation.
temic circulation is tremendously impeded, resulting in
Partial hepatectomy, in which up to 70 per cent of the
portal hypertension and increasing the capillary pres-
liver is removed, causes the remaining lobes to enlarge
sure in the intestinal wall to 15 to 20 mm Hg above
and restore the liver to its original size. This regenera-
normal. The patient often dies within a few hours
tion is remarkably rapid and requires only 5 to 7 days
because of excessive loss of fluid from the capillaries
in rats. During liver regeneration, hepatocytes are esti-
into the lumens and walls of the intestines.
mated to replicate once or twice, and after the original
Chapter 70
The Liver as an Organ
861
size and volume of the liver are achieved, the hepato-
Carbohydrate Metabolism
cytes revert to their usual quiescent state.
Control of this rapid regeneration of the liver is still
In carbohydrate metabolism, the liver performs the fol-
poorly understood, but hepatocyte growth factor (HGF)
lowing functions, as summarized from Chapter 67:
appears to be an important factor causing liver cell divi-
1. Storage of large amounts of glycogen
sion and growth. HGF is produced by mesenchymal
2. Conversion of galactose and fructose to glucose
cells in the liver and in other tissues, but not by hepa-
3. Gluconeogenesis
tocytes. Blood levels of HGF rise more than 20-fold
4. Formation of many chemical compounds from
after partial hepatectomy, but mitogenic responses are
intermediate products of carbohydrate metabolism
usually found only in the liver after these operations,
The liver is especially important for maintaining a
suggesting that HGF may be activated only in the
normal blood glucose concentration. Storage of glyco-
affected organ. Other growth factors, especially epider-
gen allows the liver to remove excess glucose from the
mal growth factor, and cytokines such as tumor necro-
blood, store it, and then return it to the blood when the
sis factor and interleukin-6 may also be involved in
blood glucose concentration begins to fall too low. This
stimulating regeneration of liver cells.
is called the glucose buffer function of the liver. In a
After the liver has returned to its original size, the
person with poor liver function, blood glucose concen-
process of hepatic cell division is terminated. Again,
tration after a meal rich in carbohydrates may rise two
the factors involved are not well understood, although
to three times as much as in a person with normal liver
transforming growth factor-b, a cytokine secreted by
function.
hepatic cells, is a potent inhibitor of liver cell prolifera-
Gluconeogenesis in the liver is also important in
tion and has been suggested as the main terminator of
maintaining a normal blood glucose concentration,
liver regeneration.
because gluconeogenesis occurs to a significant extent
Physiologic experiments indicate that liver growth is
only when the glucose concentration falls below normal.
closely regulated by some unknown signal related to
In such a case, large amounts of amino acids and glyc-
body size, so that an optimal liver-to-body weight ratio
erol from triglycerides are converted into glucose,
is maintained for optimal metabolic function. In liver
thereby helping to maintain a relatively normal blood
diseases associated with fibrosis, inflammation, or viral
glucose concentration.
infections, however, the regenerative process of the liver
is severely impaired, and liver function deteriorates.
Fat Metabolism
Although most cells of the body metabolize fat, certain
Hepatic Macrophage System Serves
aspects of fat metabolism occur mainly in the liver.
a Blood-Cleansing Function
Specific functions of the liver in fat metabolism, as
summarized from Chapter 68, are the following:
Blood flowing through the intestinal capillaries picks up
1. Oxidation of fatty acids to supply energy for other
many bacteria from the intestines. Indeed, a sample of
body functions
blood taken from the portal veins before it enters the
2. Synthesis of large quantities of cholesterol,
liver almost always grows colon bacilli when cultured,
phospholipids, and most lipoproteins
whereas growth of colon bacilli from blood in the sys-
3. Synthesis of fat from proteins and carbohydrates
temic circulation is extremely rare.
To derive energy from neutral fats, the fat is first split
Special high-speed motion pictures of the action of
into glycerol and fatty acids; then the fatty acids are split
Kupffer cells, the large phagocytic macrophages that
by beta-oxidation into two-carbon acetyl radicals that
line the hepatic venous sinuses, have demonstrated that
form acetyl coenzyme A (acetyl-CoA). This can enter
these cells efficiently cleanse blood as it passes through
the citric acid cycle and be oxidized to liberate tremen-
the sinuses; when a bacterium comes into momentary
dous amounts of energy. Beta-oxidation can take place
contact with a Kupffer cell, in less than 0.01 second the
in all cells of the body, but it occurs especially rapidly
bacterium passes inward through the wall of the Kupffer
in the hepatic cells. The liver itself cannot use all the
cell to become permanently lodged therein until it is
acetyl-CoA that is formed; instead, it is converted by the
digested. Probably less than 1 per cent of the bacteria
condensation of two molecules of acetyl-CoA into
entering the portal blood from the intestines succeeds
acetoacetic acid, a highly soluble acid that passes from
in passing through the liver into the systemic
the hepatic cells into the extracellular fluid and is then
circulation.
transported throughout the body to be absorbed by
other tissues. These tissues reconvert the acetoacetic
acid into acetyl-CoA and then oxidize it in the usual
Metabolic Functions
manner. Thus, the liver is responsible for a major part
of the Liver
of the metabolism of fats.
About 80 per cent of the cholesterol synthesized in
The liver is a large, chemically reactant pool of cells that
the liver is converted into bile salts, which are secreted
have a high rate of metabolism, sharing substrates and
into the bile; the remainder is transported in the
energy from one metabolic system to another, process-
lipoproteins and carried by the blood to the tissue cells
ing and synthesizing multiple substances that are trans-
everywhere in the body. Phospholipids are likewise syn-
ported to other areas of the body, and performing
thesized in the liver and transported principally in the
myriad other metabolic functions. For these reasons, a
lipoproteins. Both cholesterol and phospholipids are
major share of the entire discipline of biochemistry is
used by the cells to form membranes, intracellular
devoted to the metabolic reactions in the liver. But here,
structures, and multiple chemical substances that are
let us summarize those metabolic functions that are
important to cellular function.
especially important in understanding the integrated
Almost all the fat synthesis in the body from carbo-
physiology of the body.
hydrates and proteins also occurs in the liver. After fat
862
Unit XIII
Metabolism and Temperature Regulation
is synthesized in the liver, it is transported in the
known as an excellent source of certain vitamins in the
lipoproteins to the adipose tissue to be stored.
treatment of patients. The vitamin stored in greatest
quantity in the liver is vitamin A, but large quantities of
Protein Metabolism
vitamin D and vitamin B12 are normally stored as well.
Sufficient quantities of vitamin A can be stored to
The body cannot dispense with the liver’s contribution
prevent vitamin A deficiency for as long as 10 months.
to protein metabolism for more than a few days without
Sufficient vitamin D can be stored to prevent deficiency
death ensuing. The most important functions of the liver
for 3 to 4 months, and enough vitamin B12 can be stored
in protein metabolism, as summarized from Chapter 69,
to last for at least 1 year and maybe several years.
are the following:
1. Deamination of amino acids
The Liver Stores Iron as Ferritin. Except for the iron in the
2. Formation of urea for removal of ammonia from
hemoglobin of the blood, by far the greatest proportion
the body fluids
of iron in the body is stored in the liver in the form of
3. Formation of plasma proteins
ferritin. The hepatic cells contain large amounts of a
4. Interconversions of the various amino acids and
protein called apoferritin, which is capable of combin-
synthesis of other compounds from amino acids
ing reversibly with iron. Therefore, when iron is avail-
Deamination of amino acids is required before they
able in the body fluids in extra quantities, it combines
can be used for energy or converted into carbohydrates
with apoferritin to form ferritin and is stored in this
or fats. A small amount of deamination can occur in the
form in the hepatic cells until needed elsewhere. When
other tissues of the body, especially in the kidneys, but
the iron in the circulating body fluids reaches a low
this is much less important than the deamination of
level, the ferritin releases the iron. Thus, the apoferritin-
amino acids by the liver.
ferritin system of the liver acts as a blood iron buffer, as
Formation of urea by the liver removes ammonia
well as an iron storage medium. Other functions of the
from the body fluids. Large amounts of ammonia are
liver in relation to iron metabolism and red blood cell
formed by the deamination process, and additional
formation are considered in Chapter 32.
amounts are continually formed in the gut by bacteria
and then absorbed into the blood. Therefore, if the liver
The Liver Forms a Large Proportion of the Blood Substances Used
does not form urea, the plasma ammonia concentration
in Coagulation. Substances formed in the liver that are
rises rapidly and results in hepatic coma and death.
used in the coagulation process include fibrinogen, pro-
Indeed, even greatly decreased blood flow through the
thrombin, accelerator globulin, Factor VII, and several
liver—as occurs occasionally when a shunt develops
other important factors. Vitamin K is required by the
between the portal vein and the vena cava—can cause
metabolic processes of the liver for the formation of
excessive ammonia in the blood, an extremely toxic
several of these substances, especially prothrombin and
condition.
Factors VII, IX, and X. In the absence of vitamin K, the
Essentially all the plasma proteins, with the exception
concentrations of all these decrease markedly, and this
of part of the gamma globulins, are formed by the
almost prevents blood coagulation.
hepatic cells. This accounts for about 90 per cent of all
the plasma proteins. The remaining gamma globulins
The Liver Removes or Excretes Drugs, Hormones, and Other Sub-
are the antibodies formed mainly by plasma cells in the
stances. The active chemical medium of the liver is well
lymph tissue of the body. The liver can form plasma pro-
known for its ability to detoxify or excrete into the bile
teins at a maximum rate of 15 to 50 g/day. Therefore,
many drugs, including sulfonamides, penicillin, ampi-
even if as much as half the plasma proteins are lost from
cillin, and erythromycin.
the body, they can be replenished in 1 or 2 weeks.
In a similar manner, several of the hormones secreted
It is particularly interesting that plasma protein
by the endocrine glands are either chemically altered or
depletion causes rapid mitosis of the hepatic cells and
excreted by the liver, including thyroxine and essentially
growth of the liver to a larger size; these effects are
all the steroid hormones, such as estrogen, cortisol, and
coupled with rapid output of plasma proteins until the
aldosterone. Liver damage can lead to excess accumu-
plasma concentration returns to normal. With chronic
lation of one or more of these hormones in the body
liver disease (e.g., cirrhosis), plasma proteins, such as
fluids and therefore cause overactivity of the hormonal
albumin, may fall to very low levels, causing generalized
systems.
edema and ascites, as explained in Chapter 29.
Finally, one of the major routes for excreting calcium
Among the most important functions of the liver is
from the body is secretion by the liver into the bile,
its ability to synthesize certain amino acids and to
which then passes into the gut and is lost in the feces.
synthesize other important chemical compounds from
amino acids. For instance, the so-called nonessential
amino acids can all be synthesized in the liver. To do
Measurement of Bilirubin
this, a keto acid having the same chemical composition
in the Bile as a Clinical
(except at the keto oxygen) as that of the amino acid to
Diagnostic Tool
be formed is synthesized. Then an amino radical is trans-
ferred through several stages of transamination from an
The formation of bile by the liver and the function of
available amino acid to the keto acid to take the place
the bile salts in the digestive and absorptive processes
of the keto oxygen.
of the intestinal tract are discussed in Chapters 64 and
65. In addition, many substances are excreted in the bile
Other Metabolic Functions
and then eliminated in the feces. One of these is the
greenish yellow pigment bilirubin. This is a major end
of the Liver
product of hemoglobin degradation, as pointed out in
The Liver Is a Storage Site for Vitamins. The liver has a par-
Chapter 32. However, it also provides an exceedingly
ticular propensity for storing vitamins and has long been
valuable tool for diagnosing both hemolytic blood
Chapter 70
The Liver as an Organ
863
Plasma
Fragile red blood cells
Reticuloendothelial
system
Free bilirubin (protein-bound)
Liver
Urobilinogen
Liver
Kidneys
Absorbed
Conjugated bilirubin
Urobilinogen
Bacterial
Oxidation
action
Urobilin
Urobilinogen
Stercobilinogen
Oxidation
Stercobilin
Intestinal Contents
Urine
Figure 70-2
Bilirubin formation and excretion.
diseases and various types of liver diseases. Therefore,
forms, the bilirubin is excreted from the hepatocytes by
while referring to Figure 70-2, let us explain this.
an active transport process into the bile canaliculi and
Briefly, when the red blood cells have lived out their
then into the intestines.
life span (on average, 120 days) and have become too
fragile to exist in the circulatory system, their cell
Formation and Fate of Urobilinogen. Once in the intestine,
membranes rupture, and the released hemoglobin is
about half of the “conjugated” bilirubin is converted by
phagocytized by tissue macrophages (also called the
bacterial action into the substance urobilinogen, which
reticuloendothelial system) throughout the body. The
is highly soluble. Some of the urobilinogen is reab-
hemoglobin is first split into globin and heme, and
sorbed through the intestinal mucosa back into the
the heme ring is opened to give (1) free iron, which is
blood. Most of this is re-excreted by the liver back into
transported in the blood by transferrin, and
(2) a
the gut, but about 5 per cent is excreted by the kidneys
straight chain of four pyrrole nuclei, which is the sub-
into the urine. After exposure to air in the urine, the uro-
strate from which bilirubin will eventually be formed.
bilinogen becomes oxidized to urobilin; alternatively, in
The first substance formed is biliverdin, but this is
the feces, it becomes altered and oxidized to form ster-
rapidly reduced to free bilirubin, which is gradually
cobilin. These interrelations of bilirubin and the other
released from the macrophages into the plasma. The
bilirubin products are shown in Figure 70-2.
free bilirubin immediately combines strongly with
plasma albumin and is transported in this combination
throughout the blood and interstitial fluids. Even when
Jaundice—Excess Bilirubin in the
bound with plasma protein, this bilirubin is still called
Extracellular Fluid
“free bilirubin” to distinguish it from “conjugated biliru-
bin,” which is discussed later.
Jaundice refers to a yellowish tint to the body tissues,
Within hours, the free bilirubin is absorbed through
including a yellowness of the skin as well as the deep
the hepatic cell membrane. In passing to the inside of
tissues. The usual cause of jaundice is large quantities of
the liver cells, it is released from the plasma albumin and
bilirubin in the extracellular fluids, either free bilirubin
soon thereafter conjugated about 80 per cent with glu-
or conjugated bilirubin. The normal plasma concentra-
curonic acid to form bilirubin glucuronide, about 10 per
tion of bilirubin, which is almost entirely the free form,
cent with sulfate to form bilirubin sulfate, and about 10
averages 0.5 mg/dl of plasma. In certain abnormal con-
per cent with a multitude of other substances. In these
ditions, this can rise to as high as 40 mg/dl, and much of
864
Unit XIII
Metabolism and Temperature Regulation
it can become the conjugated type. The skin usually
often possible to differentiate among multiple types of
begins to appear jaundiced when the concentration rises
hemolytic diseases and liver diseases, as well as to deter-
to about three times normal—that is, above 1.5 mg/dl.
mine the severity of the disease.
The common causes of jaundice are (1) increased
destruction of red blood cells, with rapid release of
bilirubin into the blood, and (2) obstruction of the bile
References
ducts or damage to the liver cells so that even the usual
amounts of bilirubin cannot be excreted into the gas-
Alison MR, Vig P, Russo F, et al: Hepatic stem cells: from
trointestinal tract. These two types of jaundice are
inside and outside the liver? Cell Prolif 37:1, 2004.
called, respectively, hemolytic jaundice and obstructive
Angulo P: Nonalcoholic fatty liver disease. N Engl J Med
jaundice. They differ from each other in the following
346:1221, 2002.
ways.
Ankoma-Sey V: Hepatic regeneration—revisiting the myth
of Prometheus. News Physiol Sci 14:149, 1999.
Hemolytic Jaundice Is Caused by Hemolysis of Red Blood Cells.
Barthel A, Schmoll D: Novel concepts in insulin regulation
In hemolytic jaundice, the excretory function of the liver
of hepatic gluconeogenesis. Am J Physiol Endocrinol
is not impaired, but red blood cells are hemolyzed so
Metab 285:E685, 2003.
rapidly that the hepatic cells simply cannot excrete the
Bauer M: Heme oxygenase in liver transplantation: heme
bilirubin as quickly as it is formed. Therefore, the
catabolism and metabolites in the search of function.
plasma concentration of free bilirubin rises to above-
Hepatology 38:286, 2003.
normal levels. Likewise, the rate of formation of uro-
Black D, Lyman S, Heider TR, Behrns KE: Molecular and
bilinogen in the intestine is greatly increased, and much
cellular features of hepatic regeneration. J Surg Res
of this is absorbed into the blood and later excreted in
117:306, 2004.
the urine.
Bonder CS, Kubes P: The future of GI and liver research:
editorial perspectives. II. Modulating leukocyte recruit-
Obstructive Jaundice Is Caused by Obstruction of Bile Ducts or
ment to splanchnic organs to reduce inflammation. Am J
Liver Disease. In obstructive jaundice, caused either by
Physiol Gastrointest Liver Physiol 284:G729, 2003.
obstruction of the bile ducts (which most often occurs
Crispe IN: Hepatic T cells and liver tolerance. Nat Rev
when a gallstone or cancer blocks the common bile
Immunol 3:51, 2003.
duct) or by damage to the hepatic cells (which occurs in
Diehl AM: Nonalcoholic steatosis and steatohepatitis. IV.
hepatitis), the rate of bilirubin formation is normal, but
Nonalcoholic fatty liver disease abnormalities in
the bilirubin formed cannot pass from the blood into the
macrophage function and cytokines. Am J Physiol Gas-
intestines. The free bilirubin still enters the liver cells
trointest Liver Physiol 282:G1, 2002.
and becomes conjugated in the usual way. This conju-
Gines P, Cardenas A, Arroyo V, Rodes J: Management of
gated bilirubin is then returned to the blood, probably
cirrhosis and ascites. N Engl J Med 350:1646, 2004.
by rupture of the congested bile canaliculi and direct
Gines P, Guevara M, Arroyo V, Rodes J: Hepatorenal syn-
emptying of the bile into the lymph leaving the liver.
drome. Lancet 362:1819, 2003.
Thus, most of the bilirubin in the plasma becomes the
Iredale JP: Cirrhosis: new research provides a basis for
conjugated type rather than the free type.
rational and targeted treatments. BMJ 327:143, 2003.
Koniaris LG, McKillop IH, Schwartz SI, Zimmers TA: Liver
Diagnostic Differences Between Hemolytic and Obstructive
regeneration. J Am Coll Surg 197:634, 2003.
Jaundice. Chemical laboratory tests can be used to dif-
Li MK, Crawford JM: The pathology of cholestasis. Semin
ferentiate between free and conjugated bilirubin in the
Liver Dis 24:21, 2004.
plasma. In hemolytic jaundice, almost all the bilirubin is
Portincasa P, Moschetta A, Mazzone A, et al: Water handling
in the “free” form; in obstructive jaundice, it is mainly
and aquaporins in bile formation: recent advances and
in the “conjugated” form. A test called the van den
research trends. J Hepatol 39:864, 2003.
Bergh reaction can be used to differentiate between the
Ramadori G, Saile B: Mesenchymal cells in the liver—one
two.
cell type or two? Liver 22:283, 2002.
When there is total obstruction of bile flow, no biliru-
Reichen J: The role of the sinusoidal endothelium in liver
bin can reach the intestines to be converted into uro-
function. News Physiol Sci 14:117, 1999.
bilinogen by bacteria. Therefore, no urobilinogen is
Sands JM: Mammalian urea transporters. Annu Rev Physiol
reabsorbed into the blood, and none can be excreted by
65:543, 2003.
the kidneys into the urine. Consequently, in total
Schoemaker MH, Moshage H: Defying death: the hepato-
obstructive jaundice, tests for urobilinogen in the urine
cyte’s survival kit. Clin Sci (Lond) 107:13, 2004.
are completely negative. Also, the stools become clay
Schrier RW, Gurevich AK, Cadnapaphornchai MA: Patho-
colored owing to a lack of stercobilin and other bile
genesis and management of sodium and water retention
pigments.
in cardiac failure and cirrhosis. Semin Nephrol 21:157,
Another major difference between free and conju-
2001.
gated bilirubin is that the kidneys can excrete small
Su GL: Lipopolysaccharides in liver injury: molecular mech-
quantities of the highly soluble conjugated bilirubin
anisms of Kupffer cell activation. Am J Physiol Gastroin-
but not the albumin-bound free bilirubin. Therefore,
test Liver Physiol 283:G256, 2002.
in severe obstructive jaundice, significant quantities of
Trauner M, Boyer JL: Bile salt transporters: molecular char-
conjugated bilirubin appear in the urine. This can be
acterization, function, and regulation. Physiol Rev 83:633,
demonstrated simply by shaking the urine and observ-
2003.
ing the foam, which turns an intense yellow. Thus,
Wolkoff AW, Cohen DE: Bile acid regulation of hepatic
by understanding the physiology of bilirubin excretion
physiology. I. Hepatocyte transport of bile acids. Am J
by the liver and by the use of a few simple tests, it is
Physiol Gastrointest Liver Physiol 284:G175, 2003.
C H A P T E R
7
1
Dietary Balances; Regulation of
Feeding; Obesity and Starvation;
Vitamins and Minerals
Energy Intake and Output
Are Balanced Under Steady-
State Conditions
Intake of carbohydrates, fats, and proteins provides
energy that can be used to perform various body
functions or stored for later use. Stability of body
weight and composition over long periods requires that a person’s energy intake
and energy expenditure be balanced. When a person is overfed and energy
intake persistently exceeds expenditure, most of the excess energy is stored as
fat, and body weight increases; conversely, loss of body mass and starvation
occur when energy intake is insufficient to meet the body’s metabolic needs.
Because different foods contain different proportions of proteins, carbohy-
drates, fats, minerals, and vitamins, appropriate balances must also be main-
tained among these constituents so that all segments of the body’s metabolic
systems can be supplied with the requisite materials. This chapter discusses the
mechanisms by which food intake is regulated in accordance with the body’s
metabolic needs and some of the problems of maintaining balance among the
different types of foods.
Dietary Balances
Energy Available in Foods
The energy liberated from each gram of carbohydrate as it is oxidized to carbon
dioxide and water is 4.1 Calories (1 Calorie equals 1 kilocalorie), and that liberated
from fat is 9.3 Calories. The energy liberated from metabolism of the average dietary
protein as each gram is oxidized to carbon dioxide, water, and urea is 4.35 Calories.
Also, these substances vary in the average percentages that are absorbed from the
gastrointestinal tract: about 98 per cent of carbohydrate, 95 per cent of fat, and 92
per cent of protein. Therefore, the average physiologically available energy in each
gram of these three foodstuffs is as follows:
Calories
Carbohydrate
4
Fat
9
Protein
4
Average Americans receive about 15 per cent of their energy from protein, 40
per cent from fat, and 45 per cent from carbohydrate. In most non-Western coun-
tries, the quantity of energy derived from carbohydrates far exceeds that derived
from both proteins and fats. Indeed, in some parts of the world where meat is scarce,
the energy received from fats and proteins combined may be no greater than 15 to
20 per cent.
Table 71-1 gives the compositions of selected foods, demonstrating especially the
high proportions of fat and protein in meat products and the high proportion of car-
bohydrate in most vegetable and grain products. Fat is deceptive in the diet because
it usually exists as 100 per cent fat, whereas both proteins and carbohydrates are
mixed in watery media so that each of these normally represents less than 25 per
865
866
Unit XIII
Metabolism and Temperature Regulation
Table 71-1
Protein, Fat, and Carbohydrate Content of Different Foods
Food
% Protein
% Fat
% Carbohydrate
Fuel Value per 100 Grams (Calories)
Apples
0.3
0.4
14.9
64
Asparagus
2.2
0.2
3.9
26
Bacon, fat
6.2
76.0
0.7
712
broiled
25.0
55.0
1.0
599
Beef (average)
17.5
22.0
1.0
268
Beets, fresh
1.6
0.1
9.6
46
Bread, white
9.0
3.6
49.8
268
Butter
0.6
81.0
0.4
733
Cabbage
1.4
0.2
5.3
29
Carrots
1.2
0.3
9.3
45
Cashew nuts
19.6
47.2
26.4
609
Cheese, cheddar, American
23.9
32.3
1.7
393
Chicken, total edible
21.6
2.7
1.0
111
Chocolate
5.5
52.9
18.0
570
Corn (maize)
10.0
4.3
73.4
372
Haddock
17.2
0.3
0.5
72
Lamb, leg (average)
18.0
17.5
1.0
230
Milk, fresh whole
3.5
3.9
4.9
69
Molasses
0.0
0.0
60.0
240
Oatmeal, dry, uncooked
14.2
7.4
68.2
396
Oranges
0.9
0.2
11.2
50
Peanuts
26.9
44.2
23.6
600
Peas, fresh
6.7
0.4
17.7
101
Pork, ham
15.2
31.0
1.0
340
Potatoes
2.0
0.1
19.1
85
Spinach
2.3
0.3
3.2
25
Strawberries
0.8
0.6
8.1
41
Tomatoes
1.0
0.3
4.0
23
Tuna, canned
24.2
10.8
0.5
194
Walnuts, English
15.0
64.4
15.6
702
cent of the weight. Therefore, the fat of one pat of butter
two substances, and little is derived from proteins.
mixed with an entire helping of potato sometimes con-
Therefore, both carbohydrates and fats are said to
tains as much energy as the potato itself.
be protein sparers. Conversely, in starvation, after the
carbohydrates and fats have been depleted, the
Average Daily Requirement for Protein Is
30 to
50 Grams.
body’s protein stores are consumed rapidly for energy,
Twenty to 30 grams of the body proteins are degraded
sometimes at rates approaching several hundred
and used to produce other body chemicals daily. There-
grams per day rather than the normal daily rate of 30
fore, all cells must continue to form new proteins to take
to 50 grams.
the place of those that are being destroyed, and a supply
of protein is needed in the diet for this purpose. An
average person can maintain normal stores of protein,
Methods for Determining Metabolic
provided the daily intake is above 30 to 50 grams.
Utilization of Proteins,
Some proteins have inadequate quantities of certain
Carbohydrates, and Fats
essential amino acids and therefore cannot be used to
replace the degraded proteins. Such proteins are called
Nitrogen Excretion Can Be Used to Assess Protein Metabolism.
partial proteins, and when they are present in large
The average protein contains about 16 per cent nitro-
quantities in the diet, the daily protein requirement is
gen. During metabolism of the protein, about 90 per
much greater than normal. In general, proteins derived
cent of this nitrogen is excreted in the urine in the form
from animal foodstuffs are more complete than are pro-
of urea, uric acid, creatinine, and other less important
teins derived from vegetable and grain sources. For
nitrogen products. The remaining 10 per cent is excreted
example, the protein of corn has almost no tryptophan,
in the feces. Therefore, the rate of protein breakdown in
one of the essential amino acids. Therefore, individuals
the body can be estimated by measuring the amount of
in economically disadvantaged countries who consume
nitrogen in the urine, then adding 10 per cent for the
cornmeal as the principal source of protein sometimes
nitrogen excreted in the feces, and multiplying by 6.25
develop the protein-deficiency syndrome called kwashi-
(i.e., 100/16) to determine the total amount of protein
orkor, which consists of failure to grow, lethargy,
metabolism in grams per day. Thus, excretion of 8 grams
depressed mentality, and edema caused by low plasma
of nitrogen in the urine each day means that there has
protein concentration.
been about 55 grams of protein breakdown. If the daily
intake of protein is less than the daily breakdown of
Carbohydrates and Fats Act as “Protein Sparers.” When the
protein, the person is said to have a negative nitrogen
diet contains an abundance of carbohydrates and
balance, which means that his or her body stores of
fats, almost all the body’s energy is derived from these
protein are decreasing daily.
Chapter 71
Dietary Balances; Regulation of Feeding; Obesity and Starvation; Vitamins and Minerals
867
“Respiratory Quotient” Is the Ratio of CO2
Production to O2
functional systems of the cells, and much of this is
Utilization and Can Be Used to Estimate Fat and Carbohydrate
eventually converted to heat, which is generated as a
Utilization. When carbohydrates are metabolized with
result of protein metabolism, muscle activity, and
oxygen, exactly one carbon dioxide molecule is formed
activities of the various organs and tissues of the body.
for each molecule of oxygen consumed. This ratio of
Excess energy intake is stored mainly as fat, whereas
carbon dioxide output to oxygen usage is called the
a deficit of energy intake causes loss of total body
respiratory quotient, so the respiratory quotient for
mass until energy expenditure eventually equals
carbohydrates is 1.0.
When fat is oxidized in the body’s cells, an average of
energy intake or death occurs.
70 carbon dioxide molecules are formed for each 100
Although there is considerable variability in the
molecules of oxygen consumed. The respiratory quo-
amount of energy storage (i.e., fat mass) in different
tient for the metabolism of fat averages 0.70. When pro-
individuals, maintenance of an adequate energy supply
teins are oxidized by the cells, the average respiratory
is necessary for survival. Therefore, the body is
quotient is 0.80. The reason that the respiratory quo-
endowed with powerful physiologic control systems
tients for fats and proteins are lower than that for
that help maintain adequate energy intake. Deficits of
carbohydrates is that a large share of the oxygen metab-
energy stores, for example, rapidly activate multiple
olized with these foods is required to combine with the
mechanisms that cause hunger and drive a person to
excess hydrogen atoms present in their molecules, so
seek food. In athletes and laborers, energy expenditure
that less carbon dioxide is formed in relation to the
oxygen used.
for the high level of muscle activity may be as high as
Now let us see how one can make use of the respira-
6000 to 7000 Calories per day, compared with only
tory quotient to determine the relative utilization of dif-
about 2000 Calories per day for sedentary individuals.
ferent foods by the body. First, it will be recalled from
Thus, a large energy expenditure associated with phys-
Chapter 39 that the output of carbon dioxide by the
ical work usually stimulates equally large increases in
lungs divided by the uptake of oxygen during the same
caloric intake.
period is called the respiratory exchange ratio. Over a
What are the physiologic mechanisms that sense
period of 1 hour or more, the respiratory exchange ratio
changes in energy balance and influence the quest
exactly equals the average respiratory quotient of the
for food? Maintenance of adequate energy supply in
metabolic reactions throughout the body. If a person has
the body is so critical that there are multiple short-
a respiratory quotient of 1.0, he or she is metabolizing
almost entirely carbohydrates, because the respiratory
term and long-term control systems that regulate not
quotients for both fat and protein metabolism are con-
only food intake but also energy expenditure and
siderably less than 1.0. Likewise, when the respiratory
energy stores. In the next few sections we describe
quotient is about 0.70, the body is metabolizing almost
some of these control systems and their operation
entirely fats, to the exclusion of carbohydrates and
in physiologic conditions, as well as in obesity and
proteins. And, finally, if we ignore the normally small
starvation.
amount of protein metabolism, respiratory quotients
between 0.70 and 1.0 describe the approximate ratios of
carbohydrate to fat metabolism. To be more exact, one
Neural Centers Regulate Food Intake
can first determine the protein utilization by measuring
nitrogen excretion and then, using the appropriate
mathematical formula, calculate almost exactly the uti-
The sensation of hunger is associated with a craving
lization of the three foodstuffs.
for food and several other several physiologic effects,
Some of the important findings from studies of respi-
such as rhythmical contractions of the stomach and
ratory quotients are the following:
restlessness, which cause the person to search for an
1. Immediately after a meal, almost all the food
adequate food supply. A person’s appetite is a desire
that is metabolized is carbohydrates, so that the
for food, often of a particular type, and is useful in
respiratory quotient at that time approaches 1.0.
helping to choose the quality of the food to be eaten.
2. About 8 to 10 hours after a meal, the body has
If the quest for food is successful, the feeling of satiety
already used up most of its readily available
occurs. Each of these feelings is influenced by envi-
carbohydrates, and the respiratory quotient
ronmental and cultural factors, as well as by physio-
approaches that for fat metabolism, about 0.70.
3. In untreated diabetes mellitus, little carbohydrate
logic controls that influence specific centers of the
can be used by the body’s cells under any
brain, especially the hypothalamus.
conditions, because insulin is required for this.
Therefore, when diabetes is severe, most of the
The Hypothalamus Contains Hunger and Satiety Centers.
time the respiratory quotient remains near that for
Several neuronal centers of the hypothalamus partici-
fat metabolism, 0.70.
pate in the control of food intake. The lateral nuclei of
the hypothalamus serve as a feeding center, and stimu-
lation of this area causes an animal to eat voraciously
Regulation of Food Intake and
(hyperphagia). Conversely, destruction of the lateral
Energy Storage
hypothalamus causes lack of desire for food and
progressive inanition, a condition characterized by
Stability of the body’s total mass and composition over
marked weight loss, muscle weakness, and decreased
long periods requires that energy intake match energy
metabolism. The lateral hypothalamic feeding center
expenditure. As discussed in Chapter 72, only about 27
operates by exciting the motor drives to search for
per cent of the energy ingested normally reaches the
food.
868
Unit XIII
Metabolism and Temperature Regulation
The ventromedial nuclei of the hypothalamus serve
as the satiety center. This center is believed to give a
sense of nutritional satisfaction that inhibits the
Hypothalamus
feeding center. Electrical stimulation of this region can
cause complete satiety, and even in the presence
of highly appetizing food, the animal refuses to eat
+
-
(aphagia). Conversely, destruction of the ventromedial
-
nuclei causes voracious and continued eating until the
animal becomes extremely obese, sometimes as large
as four times normal.
The paraventricular, dorsomedial, and arcuate nuclei
of the hypothalamus also play a major role in regulat-
ing food intake. For example, lesions of the paraven-
tricular nuclei often cause excessive eating, whereas
Vagus nerve
lesions of the dorsomedial nuclei usually depress
eating behavior. As discussed later, the arcuate nuclei
are the sites in the hypothalamus where multiple hor-
mones released from the gastrointestinal tract and
adipose tissue converge to regulate food intake as well
as energy expenditure.
There is much chemical cross-talk among the
neurons on the hypothalamus, and together, these
centers coordinate the processes that control eating
behavior and the perception of satiety. These nuclei
Stomach
of the hypothalamus also influence the secretion of
several hormones that are important in regulating
Leptin
Ghrelin
energy balance and metabolism, including those from
the thyroid and adrenal glands, as well as the pancre-
Insulin
atic islet cells.
CCK
The hypothalamus receives neural signals from the
gastrointestinal tract that provide sensory information
PYY
about stomach filling, chemical signals from nutrients
Large intestine
Small intestine
in the blood (glucose, amino acids, and fatty acids) that
signify satiety, signals from gastrointestinal hormones,
Figure 71-1
signals from hormones released by adipose tissue,
and signals from the cerebral cortex (sight, smell,
Feedback mechanisms for control of food intake. Stretch recep-
and taste) that influence feeding behavior. Some of
tors in the stomach activate sensory afferent pathways in the
vagus nerve and inhibit food intake. Peptide YY (PYY), cholecys-
these inputs to the hypothalamus are shown in Figure
tokinin (CCK), and insulin are gastrointestinal hormones that are
71-1.
released by the ingestion of food and suppress further feeding.
The hypothalamic feeding and satiety centers have
Ghrelin is released by the stomach, especially during fasting, and
a high density of receptors for neurotransmitters and
stimulates appetite. Leptin is a hormone produced in increasing
amounts by fat cells as they increase in size; it inhibits food intake.
hormones that influence feeding behavior. A few of
the many substances that have been shown to alter
appetite and feeding behavior in experimental studies
are listed in Table 71-2 and are generally categorized
As discussed later, these neurons appear to be the
as (1) orexigenic substances that stimulate feeding, or
major targets for the actions of several hormones that
(2) anorexigenic substances that inhibit feeding.
regulate appetite, including leptin, insulin, cholecys-
tokinin (CCK), and ghrelin. In fact, the neurons of the
Neurons and Neurotransmitters in the Hypothalamus That Stim-
arcuate nuclei appear to be a site of convergence of
ulate or Inhibit Feeding. There are two distinct types of
many of the nervous and peripheral signals that regu-
neurons in the arcuate nuclei of the hypothalamus that
late energy stores.
are especially important as controllers of both appetite
The POMC neurons release a-MSH, which then
and energy expenditure
(Figure
71-2):
(1) pro-
acts on melanocortin receptors found especially in
opiomelanocortin (POMC) neurons that produce a-
neurons of the paraventricular nuclei. Although there
melanocyte-stimulating hormone (a-MSH) together
are at least five subtypes of melanocortin receptor
with cocaine- and amphetamine-related transcript
(MCR), MCR-3 and MCR-4 are especially important
(CART), and (2) neurons that produce the orexigenic
in regulating food intake and energy balance. Activa-
substances neuropeptide Y (NPY) and agouti-related
tion of these receptors reduces food intake while
protein (AGRP). Activation of the POMC neurons
increasing energy expenditure. Conversely, inhibition
decreases food intake and increases energy expendi-
of MCR-3 and MCR-4 greatly increases food intake
ture, whereas activation of the NPY-AGRP neurons
and decreases energy expenditure. The effect of MCR
increases food intake and reduces energy expenditure.
activation to increase energy expenditure appears to
Chapter 71
Dietary Balances; Regulation of Feeding; Obesity and Starvation; Vitamins and Minerals
869
Table 71-2
Neurotransmitters and Hormones That Influence Feeding and Satiety Centers in the Hypothalamus
Decrease Feeding (Anorexigenic)
Increase Feeding (Orexigenic)
a-Melanocyte-stimulating hormone (a-MSH)
Neuropeptide Y (NPY)
Leptin
Agouti-related protein (AGRP)
Serotonin
Melanin-concentrating hormone (MCH)
Norepinephrine
Orexins A and B
Corticotropin-releasing hormone
Endorphins
Insulin
Galanin (GAL)
Cholecystokinin (CCK)
Amino acids (glutamate and g-aminobutyric acid)
Glucagon-like peptide (GLP)
Cortisol
Cocaine- and amphetamine-regulated transcript (CART)
Ghrelin
Peptide YY (PYY)
Food intake
Neurons
Neuron
of PVN
Y1r
MCR-4
a-MSH
To nucleus
tractus solitarius
Food
(NTS)
intake
•Sympathetic activity
Food
•Energy expenditure
intake
AGRP/
Y1r
NPY
Arcuate
nucleus
MCR-3
POMC/
CART
Third
ventricle
LepR
MCR-3
a-MSH
LepR
+
-
+
Insulin,
Ghrelin
leptin,
CCK
Figure 71-2
Control of energy balance by two types of neurons of the arcuate nuclei: (1) pro-opiomelanocortin (POMC) neurons that release a-
melanocyte-stimulating hormone (a-MSH) and cocaine- and amphetamine-regulated transcript (CART), decreasing food intake and
increasing energy expenditure; and (2) neurons that produce agouti-related protein (AGRP) and neuropeptide Y (NPY), increasing food
intake and reducing energy expenditure. a-MSH released by POMC neurons stimulates melanocortin receptors (MCR-3 and MCR-4) in
the paraventricular nuclei (PVN), which then activate neuronal pathways that project to the nucleus tractus solitarius (NTS) and increase
sympathetic activity and energy expenditure. AGRP acts as an antagonist of MCR-4. Insulin, leptin, and cholecystokinin (CCK) are hor-
mones that inhibit AGRP-NPY neurons and stimulate adjacent POMC-CART neurons, thereby reducing food intake. Ghrelin, a hormone
secreted from the stomach, activates AGRP-NPY neurons and stimulates food intake. LepR, leptin receptor; Y1R, neuropeptide Y1
receptor. (Redrawn from Barsh GS, Schwartz MW: Nature Rev Genetics 3:589, 2002).
be mediated, at least in part, by activation of neuronal
associated with extreme obesity. In fact, mutations of
pathways that project from the paraventricular nuclei
MCR-4 represent the most common known mono-
to the nucleus tractus solitarius and stimulate sympa-
genic (single-gene) cause of human obesity, and some
thetic nervous system activity.
studies suggest that MCR-4 mutations may account for
The hypothalamic melanocortin system plays a pow-
as much as 5 to 6 percent of early-onset severe obesity
erful role in regulating energy stores of the body, and
in children. In contrast, excessive activation of the
defective signaling of the melanocortin pathway is
melanocortin system reduces appetite. Some studies
870
Unit XIII
Metabolism and Temperature Regulation
suggest that this activation may play a role in causing
Short-Term Regulation of Food Intake
the anorexia associated with severe infections or
When a person is driven by hunger to eat voraciously
cancer tumors.
and rapidly, what turns off the eating when he or she
AGRP released from the orexigenic neurons of the
has eaten enough? There has not been enough time for
hypothalamus is a natural antagonist of MCR-3 and
changes in the body’s energy stores to occur, and
MCR-4 and probably increases feeding by inhibiting
it takes hours for enough nutritional factors to be
the effects of a-MSH to stimulate melanocortin recep-
absorbed into the blood to cause the necessary inhibi-
tors (see Figure 71-2). Although the role of AGRP in
tion of eating. Yet it is important that the person not
normal physiologic control of food intake is unclear,
overeat and that he or she eat an amount of food that
excessive formation of AGRP in mice and humans,
approximates nutritional needs. The following are
due to gene mutations, is associated with excessive
several types of rapid feedback signals that are impor-
feeding and obesity.
tant for these purposes.
NPY is also released from orexigenic neurons of the
arcuate nuclei. When energy stores of the body are
Gastrointestinal Filling Inhibits Feeding. When the gas-
low, orexigenic neurons are activated to release NPY,
trointestinal tract becomes distended, especially the
which stimulates appetite. At the same time, firing of
stomach and the duodenum, stretch inhibitory signals
the POMC neurons is reduced, thereby decreasing the
are transmitted mainly by way of the vagi to suppress
activity of the melanocortin pathway and further stim-
the feeding center, thereby reducing the desire for
ulating appetite.
food (Fig. 71-1).
Neural Centers That Influence the Mechanical Process of
Feeding. Another aspect of feeding is the mechanical
Gastrointestinal Hormonal Factors Suppress Feeding. Chole-
act of the feeding process itself. If the brain is sec-
cystokinin is released mainly in response to fat enter-
tioned below the hypothalamus but above the mesen-
ing the duodenum and has a direct effect on the
cephalon, the animal can still perform the basic
feeding centers to reduce subsequent eating. Studies in
mechanical features of the feeding process. It can sali-
experimental animals suggest that CCK may decrease
vate, lick its lips, chew food, and swallow. Therefore,
feeding mainly by activation of the melanocortin
the actual mechanics of feeding are controlled by
pathway in the hypothalamus.
centers in the brain stem. The function of the other
Peptide YY (PYY) is secreted from the entire gas-
centers in feeding, then, is to control the quantity of
trointestinal tract, but especially from the ileum and
food intake and to excite these centers of feeding
colon. Food intake stimulates release of PYY, with
mechanics to activity.
blood concentrations rising to peak levels 1 to 2 hours
Neural centers higher than the hypothalamus also
after ingesting a meal. These peak levels of PYY are
play important roles in the control of feeding, partic-
influenced by the number of calories ingested and the
ularly in the control of appetite. These centers include
composition of the food, with higher levels of PYY
the amygdala and the prefrontal cortex, which are
observed after meals with a high fat content. Although
closely coupled with the hypothalamus. It will be
injections of PYY into mice have been shown to
recalled from the discussion of the sense of smell in
decrease food intake for 12 hours or more, the impor-
Chapter 53 that portions of the amygdala are a major
tance of this gastrointestinal hormone in regulating
part of the olfactory nervous system. Destructive
appetite in humans is still unclear.
lesions in the amygdala have demonstrated that some
For reasons that are not entirely understood, the
of its areas increase feeding, whereas others inhibit
presence of food in the intestines stimulates them to
feeding. In addition, stimulation of some areas of the
secrete glucagon-like peptide, which in turn enhances
amygdala elicits the mechanical act of feeding. An
glucose-dependent insulin production and secretion
important effect of destruction of the amygdala on
from the pancreas. Glucagon-like peptide and insulin
both sides of the brain is a “psychic blindness” in the
both tend to suppress appetite. Thus, eating a meal
choice of foods. In other words, the animal (and pre-
stimulates the release of several gastrointestinal hor-
sumably the human being as well) loses or at least par-
mones that may induce satiety and reduce further
tially loses the appetite control that determines the
intake of food (see Fig. 71-1).
type and quality of food it eats.
Ghrelin—a Gastrointestinal Hormone—Increases Feeding.
Ghrelin is a hormone released mainly by the oxyntic
Factors That Regulate Quantity
cells of the stomach but also, to a much less extent, by
of Food Intake
the intestine. Blood levels of ghrelin rise during
fasting, peak just before eating, and then fall rapidly
Regulation of the quantity of food intake can be
after a meal, suggesting a possible role in stimulating
divided into short-term regulation, which is concerned
feeding. Also, administration of ghrelin increases food
primarily with preventing overeating at each meal, and
intake in experimental animals, further supporting
long-term regulation, which is concerned primarily
the possibility that it may be an orexigenic hormone.
with maintenance of normal quantities of energy
However, its physiologic role in humans is still
stores in the body.
uncertain.
Chapter 71
Dietary Balances; Regulation of Feeding; Obesity and Starvation; Vitamins and Minerals
871
Oral Receptors Meter Food Intake. When an animal with
system. This is important, because increased food
an esophageal fistula is fed large quantities of food,
intake in a cold animal (1) increases its metabolic rate
even though this food is immediately lost again to the
and (2) provides increased fat for insulation, both of
exterior, the degree of hunger is decreased after a
which tend to correct the cold state.
reasonable quantity of food has passed through the
mouth. This effect occurs despite the fact that the gas-
Feedback Signals from Adipose Tissue Regulate Food Intake.
trointestinal tract does not become the least bit filled.
Most of the stored energy in the body consists of fat,
Therefore, it is postulated that various “oral factors”
the amount of which can vary considerably in differ-
related to feeding, such as chewing, salivation, swal-
ent individuals. What regulates this energy reserve, and
lowing, and tasting, “meter” the food as it passes
why is there so much variability among individuals?
through the mouth, and after a certain amount has
Recent studies suggest that the hypothalamus
passed, the hypothalamic feeding center becomes
senses energy storage through the actions of leptin, a
inhibited. However, the inhibition caused by this
peptide hormone released from adipocytes. When the
metering mechanism is considerably less intense and
amount of adipose tissue increases (signaling excess
of shorter duration, usually lasting for only 20 to 40
energy storage), the adipocytes produce increased
minutes, than is the inhibition caused by gastrointesti-
amounts of leptin, which is released into the blood.
nal filling.
Leptin then circulates to the brain, where it moves
across the blood-brain barrier by facilitated diffusion
Intermediate and Long-Term Regulation
and occupies leptin receptors at multiple sites in the
of Food Intake
hypothalamus, especially the POMC neurons of the
An animal that has been starved for a long time and
arcuate nuclei and neurons of the paraventricular
is then presented with unlimited food eats a far greater
nuclei.
quantity than does an animal that has been on a
Stimulation of leptin receptors in these hypothala-
regular diet. Conversely, an animal that has been force-
mic nuclei initiates multiple actions that decrease fat
fed for several weeks eats very little when allowed to
storage, including
(1) decreased production in the
eat according to its own desires. Thus, the feeding
hypothalamus of appetite stimulators, such as NPY
control mechanism of the body is geared to the nutri-
and AGRP; (2) activation of POMC neurons, causing
tional status of the body.
release of a-MSH and activation of melanocortin
receptors; (3) increased production in the hypothala-
Effect of Blood Concentrations of Glucose, Amino Acids, and
mus of substances, such as corticotropin-releasing
Lipids on Hunger and Feeding. It has long been known that
hormone, that decrease food intake; (4) increased
a decrease in blood glucose concentration causes
sympathetic nerve activity (through neural projections
hunger, which has led to the so-called glucostatic
from the hypothalamus to the vasomotor centers),
theory of hunger and feeding regulation. Similar
which increases metabolic rate and energy expendi-
studies have demonstrated the same effect for blood
ture; and (5) decreased insulin secretion by the pan-
amino acid concentration and blood concentration of
creatic beta cells, which decreases energy storage.
breakdown products of lipids such as the keto acids
Thus, leptin may be an important means by which
and some fatty acids, leading to the aminostatic and
the adipose tissue signals the brain that enough energy
lipostatic theories of regulation. That is, when the avail-
has been stored and that intake of food is no longer
ability of any of the three major types of food
necessary.
decreases, the desire for feeding is increased, eventu-
In mice or humans with mutations that render their
ally returning the blood metabolite concentrations
fat cells unable to produce leptin or mutations that
back toward normal.
cause defective leptin receptors in the hypothalamus,
Neurophysiologic studies of function in specific
marked hyperphagia and morbid obesity occur. In
areas of the brain also support the glucostatic, amino-
most obese humans, however, there does not appear
static, and lipostatic theories, by the following obser-
to be a deficiency of leptin production, because plasma
vations: (1) A rise in blood glucose level increases the
leptin levels increase in proportion with increasing
rate of firing of glucoreceptor neurons in the satiety
adiposity. Therefore, some physiologists believe that
center in the ventromedial and paraventricular nuclei of
obesity may be associated with leptin resistance; that
the hypothalamus. (2) The same increase in blood
is, leptin receptors or post-receptor signaling pathways
glucose level simultaneously decreases the firing of glu-
normally activated by leptin may be defective in obese
cosensitive neurons in the hunger center of the lateral
people, who continue to eat despite very high levels of
hypothalamus. In addition, some amino acids and lipid
leptin.
substances affect the rates of firing of these same
Another explanation for the failure of leptin to
neurons or other closely associated neurons.
prevent increasing adiposity in obese individuals is
that there are many redundant systems that control
Temperature Regulation and Food Intake. When an animal
feeding behavior, as well as social and cultural factors
is exposed to cold, it tends to increase feeding; when
that can cause continued excess food intake even in
it is exposed to heat, it tends to decrease its caloric
the presence of high levels of leptin.
intake. This is caused by interaction within the hypo-
thalamus between the temperature-regulating system
Summary of Long-Term Regulation. Even though our
(see Chapter
73) and the food intake-regulating
information on the different feedback factors in
872
Unit XIII
Metabolism and Temperature Regulation
long-term feeding regulation is imprecise, we can
liver and other tissues of the body often accumulate sig-
make the following general statement: When the
nificant amounts of lipids in obese persons. The meta-
energy stores of the body fall below normal, the
bolic processes involved in fat storage were discussed in
Chapter 68.
feeding centers of the hypothalamus and other areas
It was previously believed that the number of
of the brain become highly active, and the person
adipocytes could increase substantially only during
exhibits increased hunger as well as searching for food;
infancy and childhood and that excess energy intake
conversely, when the energy stores (mainly the fat
in children led to hyperplastic obesity, associated with
stores) are already abundant, the person usually loses
increased numbers of adipocytes and only small
the sensation of hunger and develops a state of satiety.
increases in adipocyte size. In contrast, obesity devel-
oping in adults was thought to increase only adipocyte
Importance of Having Both Long- and Short-
size, resulting in hypertrophic obesity. Recent studies,
Term Regulatory Systems for Feeding
however, have shown that new adipocytes can differen-
The long-term regulatory system for feeding, which
tiate from fibroblast-like preadipocytes at any period of
life and that the development of obesity in adults is
includes all the nutritional energy feedback mecha-
accompanied by increased numbers, as well as increased
nisms, helps maintain constant stores of nutrients in
size, of adipocytes. An extremely obese person may have
the tissues, preventing them from becoming too low or
as many as four times as many adipocytes, each con-
too high. The short-term regulatory stimuli serve two
taining twice as much lipid, as a lean person.
other purposes. First, they tend to make the person eat
Once a person has become obese and a stable weight
smaller quantities at each eating session, thus allowing
is obtained, energy intake once again equals energy
food to pass through the gastrointestinal tract at a
output. For a person to lose weight, energy intake must
steadier pace, so that its digestive and absorptive
be less than energy expenditure.
mechanisms can work at optimal rates rather than
becoming periodically overburdened. Second, they
help prevent the person from eating amounts at each
Decreased Physical Activity and
meal that would be too much for the metabolic storage
Abnormal Feeding Regulation as
systems once all the food has been absorbed.
Causes of Obesity
The causes of obesity are complex. Although genes
Obesity
play an important role in determining food intake and
energy metabolism, lifestyle and environmental factors
Obesity can be defined as an excess of body fat. A sur-
may play the dominant role in many obese people. The
rogate marker for body fat content is the body mass
rapid increase in the prevalence of obesity in the past
index (BMI), which is calculated as:
20 to 30 years emphasizes the important role of lifestyle
and environmental factors, because genetic changes
BMI = Weight in kg/Height m2
could not have occurred so rapidly.
In clinical terms, a BMI between 25 and 29.9 kg/m2 is
called overweight, and a BMI greater than 30 kg/m2 is
Sedentary Lifestyle Is a Major Cause of Obesity. Regular phys-
called obese. BMI is not a direct estimate of adiposity
ical activity and physical training are known to increase
and does not take into account the fact that some indi-
muscle mass and decrease body fat mass, whereas
viduals have a high BMI due to a large muscle mass. A
inadequate physical activity is typically associated
better way to define obesity is to actually measure the
with decreased muscle mass and increased adiposity.
percentage of total body fat. Obesity is usually defined
For example, studies have shown a close association
as 25 per cent or greater total body fat in men and 35
between sedentary behaviors, such as prolonged televi-
per cent or greater in women. Although percentage of
sion watching, and obesity.
body fat can be estimated with various methods, such as
About 25 to 30 per cent of the energy used each day
measuring skin-fold thickness, bioelectrical impedance,
by the average person goes into muscular activity, and
or underwater weighing, these methods are rarely used
in a laborer, as much as 60 to 70 per cent is used in this
in clinical practice, where BMI is commonly used to
way. In obese people, increased physical activity usually
assess obesity.
increases energy expenditure more than food intake,
The prevalence of obesity in children and adults in
resulting in significant weight loss. Even a single episode
the United States and in many other industrialized
of strenuous exercise may increase basal energy expen-
countries is rapidly increasing, rising by more than 30
diture for several hours after the physical activity is
per cent over the past decade. Approximately 64 per
stopped. Because muscular activity is by far the most
cent of adults in the United States are overweight, and
important means by which energy is expended in the
nearly 33 per cent of adults are obese.
body, increased physical activity is often an effective
means of reducing fat stores.
Obesity Results from Greater Energy Intake than Energy Expendi-
ture. When greater quantities of energy (in the form of
Abnormal Feeding Behavior Is an Important Cause of Obesity.
food) enter the body than are expended, the body
Although powerful physiologic mechanisms regulate
weight increases, and most of the excess energy is stored
food intake, there are also important environmental and
as fat. Therefore, excessive adiposity (obesity) is caused
psychological factors that can cause abnormal feeding
by energy intake in excess of energy output. For each
behavior, excessive energy intake, and obesity.
9.3 Calories of excess energy that enter the body,
approximately 1 gram of fat is stored.
Environmental, Social, and Psychological Factors Con-
Fat is stored mainly in adipocytes in subcutaneous
tribute to Abnormal Feeding. As discussed previously,
tissue and in the intraperitoneal cavity, although the
the importance of environmental factors is evident from
Chapter 71
Dietary Balances; Regulation of Feeding; Obesity and Starvation; Vitamins and Minerals
873
the rapid increase in the prevalence of obesity in most
Genes can contribute to obesity by causing abnor-
industrialized countries, which has coincided with an
malities of (1) one or more of the pathways that regu-
abundance of high-energy foods (especially fatty foods)
late the feeding centers and (2) energy expenditure and
and sedentary lifestyles.
fat storage. Three of the monogenic (single-gene) causes
Psychological factors may contribute to obesity in
of obesity are
(1) mutations of MCR-4, the most
some people. For example, people often gain large
common monogenic form of obesity discovered thus
amounts of weight during or after stressful situations,
far; (2) congenital leptin deficiency caused by mutations
such as the death of a parent, a severe illness, or even
of the leptin gene, which are very rare; and (3) muta-
mental depression. It seems that eating can be a means
tions of the leptin receptor, also very rare. All these
of releasing tension.
monogenic forms of obesity account for only a very
small percentage of obesity. It is likely that many gene
Childhood Overnutrition as a Possible Cause of Obesity.
variations interact with environmental factors to influ-
One factor that may contribute to obesity is the preva-
ence the amount and distribution of body fat.
lent idea that healthy eating habits require three meals
a day and that each meal must be filling. Many young
children are forced into this habit by overly solicitous
Treatment of Obesity
parents, and the children continue to practice it
throughout life.
Treatment of obesity depends on decreasing energy
The rate of formation of new fat cells is especially
input below energy expenditure and creating a sus-
rapid in the first few years of life, and the greater the
tained negative energy balance until the desired weight
rate of fat storage, the greater the number of fat cells.
loss is achieved. In other words, this means either reduc-
The number of fat cells in obese children is often as
ing energy intake or increasing energy expenditure. The
much as three times that in normal children. Therefore,
current National Institutes of Health (NIH) guidelines
it has been suggested that overnutrition of children—
recommend a decrease in caloric intake of 500 kilo-
especially in infancy and, to a lesser extent, during the
calories per day for overweight and moderately obese
later years of childhood—can lead to a lifetime of
persons (BMI greater than 25 but less than 35 kg/m2) to
obesity.
achieve a weight loss of approximately 1 pound each
week. A more aggressive energy deficit of 500 to 1000
kilocalories per day is recommended for persons with
Neurogenic Abnormalities as a Cause of Obesity. We pre-
BMIs greater than 35 kg/m2. Typically, such an energy
viously pointed out that lesions in the ventromedial
deficit, if it can be achieved and sustained, will cause a
nuclei of the hypothalamus cause an animal to eat
weight loss of about 1 to 2 pounds per week, or about a
excessively and become obese. People with hypophysial
10 per cent weight loss after 6 months. For most people
tumors that encroach on the hypothalamus often
attempting to lose weight, increasing physical activity is
develop progressive obesity, demonstrating that obesity
also an important component of successful long-term
in human beings, too, can result from damage to the
weight loss.
hypothalamus.
To decrease energy intake, most reducing diets are
Although hypothalamic damage is almost never
designed to contain large quantities of “bulk,” which is
found in obese people, it is possible that the functional
generally made up of non-nutritive cellulose substances.
organization of the hypothalamic or other neurogenic
This bulk distends the stomach and thereby partially
feeding centers in obese individuals is different from
appeases hunger. In most lower animals, such a proce-
that in nonobese persons. Also, there may be abnor-
dure simply makes the animal increase its food intake
malities of neurotransmitters or receptor mechanisms in
even more, but human beings can often fool themselves
the neural pathways of the hypothalamus that control
because their food intake is sometimes controlled as
feeding. In support of this theory, an obese person who
much by habit as by hunger. As pointed out later in con-
has reduced to normal weight by strict dietary measures
nection with starvation, it is important to prevent
usually develops intense hunger that is demonstrably
vitamin deficiencies during the dieting period.
far greater than that of a normal person. This indicates
Various drugs for decreasing the degree of hunger
that the “set-point” of an obese person’s feeding control
have been used in the treatment of obesity. The most
system is at a much higher level of nutrient storage than
widely used drugs are amphetamines (or amphetamine
that of a nonobese person.
derivatives), which directly inhibit the feeding centers
Studies in experimental animals also indicate that
in the brain. One drug for treating obesity is sibu-
when food intake is restricted in obese animals, there
tramine, a sympathomimetic that reduces food intake
are marked neurotransmitter changes in the hypothala-
and increases energy expenditure. The danger in using
mus that greatly increase hunger and oppose weight
these drugs is that they simultaneously overexcite the
loss. Some of these changes include increased formation
central nervous system, making the person nervous and
of orexigenic neurotransmitters such as NPY and
elevating the blood pressure. Also, a person soon adapts
decreased formation of anorexic substances such as
to the drug, so that weight reduction is usually no
leptin and a-MSH.
greater than 5 to 10 per cent.
Another group of drugs works by altering lipid
Genetic Factors as a Cause of Obesity. Obesity definitely
metabolism. For example, orlistat, a lipase inhibitor,
runs in families. Yet it has been difficult to determine
reduces the intestinal digestion of fat. This causes a
the precise role of genetics in contributing to obesity,
portion of the ingested fat to be lost in the feces and
because family members generally share many of the
therefore reduces energy absorption. However, fecal fat
same eating habits and physical activity patterns.
loss may cause unpleasant gastrointestinal side effects,
Current evidence, however, suggests that 20 to 25 per
as well as loss of fat-soluble vitamins in the feces.
cent of cases of obesity may be caused by genetic
Significant weight loss can be achieved in many obese
factors.
persons with increased physical activity. The more
874
Unit XIII
Metabolism and Temperature Regulation
exercise one gets, the greater the daily energy expendi-
Several inflammatory cytokines, including tumor
ture and the more rapidly the obesity disappears.
necrosis factor-a , interleukin-6, interleukin-1b, and a
Therefore, forced exercise is often an essential part of
proteolysis-inducing factor, have been shown to cause
treatment. The current clinical guidelines for the treat-
anorexia and cachexia. Most of these inflammatory
ment of obesity recommend that the first step be
cytokines appear to mediate anorexia by activation of
lifestyle modifications that include increased physical
the melanocortin system in the hypothalamus. The
activity combined with a reduction in caloric intake. For
precise mechanisms by which cytokines or tumor prod-
morbidly obese patients with BMIs greater than 40, or
ucts interact with the melanocortin pathway to decrease
for patients with BMIs greater than 35 and conditions
food intake are still unclear, but blockade of the hypo-
such as hypertension or type II diabetes that predispose
thalamic melanocortin receptors appears to almost
them to other serious diseases, various surgical proce-
completely prevent their anorexic and cachectic effects
dures can be used to decrease the fat mass of the body
in experimental animals. Additional research, however,
or to decrease the amount of food that can be eaten at
is needed to better understand the pathophysiologic
each meal.
mechanisms of anorexia and cachexia in cancer patients
Two of the most common surgical procedures used in
and to develop therapeutic agents to improve their
the United States to treat morbid obesity are gastric
nutritional status and survival.
bypass surgery and gastric banding surgery. Gastric
bypass surgery involves construction of a small pouch in
the proximal part of the stomach that is then connected
Starvation
to the jejunum with a section of small bowel of varying
Depletion of Food Stores in the Body Tissues During Starvation.
lengths; the pouch is separated from the remaining part
Even though the tissues preferentially use carbohydrate
of the stomach with staples. Gastric banding surgery
for energy over both fat and protein, the quantity of car-
involves placing an adjustable band around the stomach
bohydrate normally stored in the entire body is only a
near its upper end; this also creates a small stomach
few hundred grams (mainly glycogen in the liver and
pouch that restricts the amount of food that can be
muscles), and it can supply the energy required for body
eaten at each meal. Although these surgical procedures
functions for perhaps half a day. Therefore, except for
generally produce substantial weight loss in obese
the first few hours of starvation, the major effects are
patients, they are major operations, and their long-
progressive depletion of tissue fat and protein. Because
term effects on overall health and mortality are still
fat is the prime source of energy (100 times as much fat
uncertain.
energy is stored in the normal person as carbohydrate
energy), the rate of fat depletion continues unabated, as
shown in Figure 71-3, until most of the fat stores in the
Inanition, Anorexia, and
body are gone.
Cachexia
Protein undergoes three phases of depletion: rapid
depletion at first, then greatly slowed depletion, and,
Inanition is the opposite of obesity and is characterized
finally, rapid depletion again shortly before death. The
by extreme weight loss. It can be caused by inadequate
initial rapid depletion is caused by the use of easily
availability of food or by pathophysiologic conditions
mobilized protein for direct metabolism or for conver-
that greatly decrease the desire for food, including psy-
sion to glucose and then metabolism of glucose mainly
chogenic disturbances, hypothalamic abnormalities,
by the brain. After the readily mobilized protein stores
and factors released from peripheral tissues. In many
have been depleted during the early phase of starvation,
instances, especially in those with serious diseases such
as cancer, the reduced desire for food may be associated
with increased energy expenditure, causing serious
weight loss.
Anorexia can be defined as a reduction in food intake
caused primarily by diminished appetite, as opposed to
the literal definition of “not eating.” This definition
emphasizes the important role of central neural mech-
12
anisms in the pathophysiology of anorexia in diseases
Protein
such as cancer, when other common problems, such as
10
pain and nausea, may also cause a person to consume
less food. Anorexia nervosa is an abnormal psychic state
8
in which a person loses all desire for food and even
Fat
becomes nauseated by food; as a result, severe inanition
6
occurs.
Cachexia is a metabolic disorder of increased energy
4
expenditure leading to weight loss greater than that
caused by reduced food intake alone. Anorexia and
2
cachexia often occur together in many types of cancer
Carbohydrate
or in the
“wasting syndrome” observed in patients
0
with acquired immunodeficiency syndrome (AIDS) and
0
1
2
3
4
5
6
7
8
Weeks of starvation
chronic inflammatory disorders. Almost all types of
cancer cause both anorexia and cachexia, and more than
half of cancer patients develop anorexia-cachexia syn-
drome during the course of their disease.
Figure 71-3
Central neural and peripheral factors are believed to
contribute to cancer-induced anorexia and cachexia.
Effect of starvation on the food stores of the body.
Chapter 71
Dietary Balances; Regulation of Feeding; Obesity and Starvation; Vitamins and Minerals
875
the remaining protein is not so easily removed. At this
major extent in the liver. For instance, the quantity of
time, the rate of gluconeogenesis decreases to one third
vitamin A stored in the liver may be sufficient to main-
to one fifth its previous rate, and the rate of depletion
tain a person for 5 to 10 months without any intake of
of protein becomes greatly decreased. The lessened
vitamin A. The quantity of vitamin D stored in the liver
availability of glucose then initiates a series of events
is usually sufficient to maintain a person for 2 to 4
that leads to excessive fat utilization and conversion of
months without any additional intake of vitamin D.
some of the fat breakdown products to ketone bodies,
The storage of most water-soluble vitamins is rela-
producing the state of ketosis, which is discussed in
tively slight. This applies especially to most vitamin B
Chapter 68. The ketone bodies, like glucose, can cross
compounds. When a person’s diet is deficient in vitamin
the blood-brain barrier and can be used by the brain
B compounds, clinical symptoms of the deficiency can
cells for energy. Therefore, about two thirds of the
sometimes be recognized within a few days (except for
brain’s energy is now derived from these ketone bodies,
vitamin B12, which can last in the liver in a bound form
principally from beta-hydroxybutyrate. This sequence
for a year or longer). Absence of vitamin C, another
of events leads to at least partial preservation of the
water-soluble vitamin, can cause symptoms within a few
protein stores of the body.
weeks and can cause death from scurvy in 20 to 30
There finally comes a time when the fat stores are
weeks.
almost depleted, and the only remaining source of
energy is protein. At that time, the protein stores once
again enter a stage of rapid depletion. Because proteins
Vitamin A
are also essential for the maintenance of cellular func-
Vitamin A occurs in animal tissues as retinol. This
tion, death ordinarily ensues when the proteins of the
vitamin does not occur in foods of vegetable origin, but
body have been depleted to about half their normal
provitamins for the formation of vitamin A do occur in
level.
abundance in many vegetable foods. These are the
yellow and red carotenoid pigments, which, because
Vitamin Deficiencies in Starvation. The stores of some of the
their chemical structures are similar to that of vitamin
vitamins, especially the water-soluble vitamins—the
A, can be changed into vitamin A in the liver.
vitamin B group and vitamin C—do not last long during
starvation. Consequently, after a week or more of
Vitamin A Deficiency Causes “Night Blindness” and Abnormal
starvation, mild vitamin deficiencies usually begin to
Epithelial Cell Growth. One basic function of vitamin A is
appear, and after several weeks, severe vitamin defi-
its use in the formation of the retinal pigments of the
ciencies can occur. These deficiencies can add to the
eye, which is discussed in Chapter 50. Vitamin A is
debility that leads to death.
needed to form the visual pigments and, therefore, to
prevent night blindness.
Vitamin A is also necessary for normal growth of
Vitamins
most cells of the body and especially for normal growth
Daily Requirements of Vitamins. A vitamin is an organic
and proliferation of the different types of epithelial
compound needed in small quantities for normal
cells. When vitamin A is lacking, the epithelial structures
metabolism that cannot be manufactured in the cells of
of the body tend to become stratified and keratinized.
the body. Lack of vitamins in the diet can cause impor-
Vitamin A deficiency manifests itself by (1) scaliness of
tant metabolic deficits. Table 71-3 lists the amounts
the skin and sometimes acne; (2) failure of growth of
of important vitamins required daily by the average
young animals, including cessation of skeletal growth;
person. These requirements vary considerably, depend-
(3) failure of reproduction, associated especially with
ing on such factors as body size, rate of growth, amount
atrophy of the germinal epithelium of the testes and
of exercise, and pregnancy.
sometimes with interruption of the female sexual cycle;
and (4) keratinization of the cornea, with resultant
Storage of Vitamins in the Body. Vitamins are stored to a
corneal opacity and blindness.
slight extent in all cells. Some vitamins are stored to a
In vitamin A deficiency, the damaged epithelial struc-
tures often become infected, for example, the conjunc-
tivae of the eyes, the linings of the urinary tract, and the
respiratory passages. Vitamin A has been called an
Table 71-3
“anti-infection” vitamin.
Required Daily Amounts of Vitamins
Thiamine (Vitamin B1)
Vitamin
Amount
Thiamine operates in the metabolic systems of the body
A
5000 IU
principally as thiamine pyrophosphate; this compound
Thiamine
1.5 mg
functions as a cocarboxylase, operating mainly in con-
Riboflavin
1.8 mg
junction with a protein decarboxylase for decarboxyla-
Niacin
20 mg
tion of pyruvic acid and other a-keto acids, as discussed
Ascorbic acid
45 mg
in Chapter 67.
D
400 IU
Thiamine deficiency (beriberi) causes decreased uti-
E
15 IU
lization of pyruvic acid and some amino acids by the
K
70 mg
tissues, but increased utilization of fats. Thus, thiamine
Folic acid
0.4 mg
B12
3 mg
is specifically needed for the final metabolism of carbo-
Pyridoxine
2 mg
hydrates and many amino acids. The decreased utiliza-
Pantothenic acid
Unknown
tion of these nutrients is responsible for many debilities
associated with thiamine deficiency.
876
Unit XIII
Metabolism and Temperature Regulation
Thiamine Deficiency Causes Lesions of the Central and Peripheral
In the early stages of niacin deficiency, simple phys-
Nervous Systems. The central nervous system normally
iologic changes such as muscle weakness and poor glan-
depends almost entirely on the metabolism of carbohy-
dular secretion may occur, but in severe niacin
drates for its energy. In thiamine deficiency, the utiliza-
deficiency, actual tissue death ensues. Pathological
tion of glucose by nervous tissue may be decreased 50
lesions appear in many parts of the central nervous
to 60 per cent and is replaced by the utilization of
system, and permanent dementia or many types of psy-
ketone bodies derived from fat metabolism. The neu-
choses may result. Also, the skin develops a cracked,
ronal cells of the central nervous system frequently
pigmented scaliness in areas that are exposed to
show chromatolysis and swelling during thiamine defi-
mechanical irritation or sun irradiation; thus, it appears
ciency, changes that are characteristic of neuronal
that in persons with niacin deficiency, the skin is unable
cells with poor nutrition. These changes can disrupt
to repair irritative damage.
communication in many portions of the central nervous
Niacin deficiency causes intense irritation and
system.
inflammation of the mucous membranes of the mouth
Thiamine deficiency can cause degeneration of myelin
and other portions of the gastrointestinal tract, result-
sheaths of nerve fibers in both the peripheral nerves and
ing in many digestive abnormalities that can lead to
the central nervous system. Lesions in the peripheral
widespread gastrointestinal hemorrhage in severe cases.
nerves frequently cause them to become extremely irri-
It is possible that this results from generalized depres-
table, resulting in “polyneuritis,” characterized by pain
sion of metabolism in the gastrointestinal epithelium
radiating along the course of one or many peripheral
and failure of appropriate epithelial repair.
nerves. Also, fiber tracts in the cord can degenerate to
The clinical entity called pellagra and the canine
such an extent that paralysis occasionally results; even
disease called black tongue are caused mainly by niacin
in the absence of paralysis, the muscles atrophy, result-
deficiency. Pellagra is greatly exacerbated in people on
ing in severe weakness.
a corn diet, because corn is deficient in the amino acid
tryptophan, which can be converted in limited quanti-
Thiamine Deficiency Weakens the Heart and Causes Peripheral
ties to niacin in the body.
Vasodilation. A person with severe thiamine deficiency
eventually develops cardiac failure because of weak-
Riboflavin (Vitamin B2)
ened cardiac muscle. Further, the venous return of
blood to the heart may be increased to as much as two
Riboflavin normally combines in the tissues with phos-
times normal. This occurs because thiamine deficiency
phoric acid to form two coenzymes, flavin mononu-
causes peripheral vasodilation throughout the circula-
cleotide (FMN) and flavin adenine dinucleotide (FAD).
tory system, presumably as a result of decreased release
They operate as hydrogen carriers in important oxida-
of metabolic energy in the tissues, leading to local vas-
tive systems of the mitochondria. NAD, operating
cular dilation. The cardiac effects of thiamine deficiency
in association with specific dehydrogenases, usually
are due partly to high blood flow into the heart and
accepts hydrogen removed from various food substrates
partly to primary weakness of the cardiac muscle.
and then passes the hydrogen to FMN or FAD; finally,
Peripheral edema and ascites also occur to a major
the hydrogen is released as an ion into the mitochon-
extent in some people with thiamine deficiency, mainly
drial matrix to become oxidized by oxygen (described
because of cardiac failure.
in Chapter 67).
Deficiency of riboflavin in experimental animals
Thiamine Deficiency Causes Gastrointestinal Tract Disturbances.
causes severe dermatitis, vomiting, diarrhea, muscle
Among the gastrointestinal symptoms of thiamine defi-
spasticity that finally becomes muscle weakness, coma
ciency are indigestion, severe constipation, anorexia,
and decline in body temperature, and then death. Thus,
gastric atony, and hypochlorhydria. All these effects pre-
severe riboflavin deficiency can cause many of the same
sumably result from failure of the smooth muscle and
effects as a lack of niacin in the diet; presumably, the
glands of the gastrointestinal tract to derive sufficient
debilities that result in each instance are due to gener-
energy from carbohydrate metabolism.
ally depressed oxidative processes within the cells.
The overall picture of thiamine deficiency, including
In the human being, there are no known cases of
polyneuritis, cardiovascular symptoms, and gastro-
riboflavin deficiency severe enough to cause the marked
intestinal disorders, is frequently referred to as
debilities noted in experimental animals, but mild
beriberi—especially when the cardiovascular symptoms
riboflavin deficiency is probably common. Such defi-
predominate.
ciency causes digestive disturbances, burning sensations
of the skin and eyes, cracking at the corners of the
mouth, headaches, mental depression, forgetfulness, and
so on.
Niacin
Although the manifestations of riboflavin deficiency
Niacin, also called nicotinic acid, functions in the body
are usually relatively mild, this deficiency frequently
as coenzymes in the form of nicotinamide adenine
occurs in association with deficiency of thiamine, niacin,
dinucleotide (NAD) and nicotinamide adenine dinu-
or both. Many deficiency syndromes, including pellagra,
cleotide phosphate
(NADP). These coenzymes are
beriberi, sprue, and kwashiorkor, are probably due to a
hydrogen acceptors; they combine with hydrogen atoms
combined deficiency of a number of vitamins, as well as
as they are removed from food substrates by many types
other aspects of malnutrition.
of dehydrogenases. The typical operation of both these
coenzymes is presented in Chapter 67. When a defi-
Vitamin B12
ciency of niacin exists, the normal rate of dehydrogena-
tion cannot be maintained; therefore, oxidative delivery
Several cobalamin compounds that possess the common
of energy from the foodstuffs to the functioning ele-
prosthetic group shown next exhibit so-called vitamin
ments of all cells cannot occur at normal rates.
B12 activity.
Chapter 71
Dietary Balances; Regulation of Feeding; Obesity and Starvation; Vitamins and Minerals
877
Pyridoxine (Vitamin B6)
N
Pyridoxine exists in the form of pyridoxal phosphate in
N
the cells and functions as a coenzyme for many chemi-
N
Co
N
cal reactions related to amino acid and protein metab-
olism. Its most important role is that of coenzyme in the
N
transamination process for the synthesis of amino acids.
2
CH
As a result, pyridoxine plays many key roles in metab-
olism, especially protein metabolism. Also, it is believed
to act in the transport of some amino acids across cell
membranes.
Note that this prosthetic group contains cobalt, which
has bonds similar to those of iron in the hemoglobin
OH
molecule. It is likely that the cobalt atom functions in
much the same way that the iron atom functions to
combine reversibly with other substances.
CH2
OH
Vitamin B12 Deficiency Causes Pernicious Anemia. Vitamin B12
performs several metabolic functions, acting as a hydro-
H2C
OH
gen acceptor coenzyme. Its most important function is
to act as a coenzyme for reducing ribonucleotides to
CH3
deoxyribonucleotides, a step that is necessary in the
N
replication of genes. This could explain the major func-
Pyridoxine
tions of vitamin B12: (1) promotion of growth and (2)
promotion of red blood cell formation and maturation.
This red cell function is described in detail in Chapter
Dietary lack of pyridoxine in lower animals can cause
32 in relation to pernicious anemia, a type of anemia
dermatitis, decreased rate of growth, development of
caused by failure of red blood cell maturation when
fatty liver, anemia, and evidence of mental deteriora-
vitamin B12 is deficient.
tion. Rarely, in children, pyridoxine deficiency has been
known to cause seizures, dermatitis, and gastrointestinal
Vitamin B12 Deficiency Causes Demyelination of the Large Nerve
disturbances such as nausea and vomiting.
Fibers of the Spinal Cord. The demyelination of nerve
fibers in people with vitamin B12 deficiency occurs espe-
cially in the posterior columns, and occasionally the
Pantothenic Acid
lateral columns, of the spinal cord. As a result, many
people with pernicious anemia have loss of peripheral
Pantothenic acid mainly is incorporated in the body into
sensation and, in severe cases, even become paralyzed.
coenzyme A (CoA), which has many metabolic roles in
The usual cause of vitamin B12 deficiency is not lack
the cells. Two of these discussed at length in Chapters
of this vitamin in the food but deficiency of formation
67 and 68 are (1) conversion of decarboxylated pyruvic
of intrinsic factor, which is normally secreted by the
acid into acetyl-CoA before its entry into the citric acid
parietal cells of the gastric glands and is essential for
cycle, and (2) degradation of fatty acid molecules into
absorption of vitamin B12 by the ileal mucosa. This is dis-
multiple molecules of acetyl-CoA. Thus, lack of pan-
cussed in Chapters 32 and 66.
tothenic acid can lead to depressed metabolism of both
carbohydrates and fats.
Deficiency of pantothenic acid in lower animals can
Folic Acid (Pteroylglutamic Acid)
cause retarded growth, failure of reproduction, graying
of the hair, dermatitis, fatty liver, and hemorrhagic
Several pteroylglutamic acids exhibit the “folic acid
adrenocortical necrosis. In the human being, no definite
effect.” Folic acid functions as a carrier of hydroxy-
deficiency syndrome has been proved, presumably
methyl and formyl groups. Perhaps its most important
because of the wide occurrence of this vitamin in almost
use in the body is in the synthesis of purines and thymine,
all foods and because small amounts can probably be
which are required for formation of DNA. Therefore,
synthesized in the body. This does not mean that pan-
folic acid, like vitamin B12, is required for replication of
tothenic acid is not of value in the metabolic systems of
the cellular genes. This may explain one of the most
the body; indeed, it is perhaps as necessary as any other
important functions of folic acid—to promote growth.
vitamin.
Indeed, when it is absent from the diet, an animal grows
very little.
Folic acid is an even more potent growth promoter
Ascorbic Acid (Vitamin C)
than vitamin B12 and, like vitamin B12, is important
for the maturation of red blood cells, as discussed
Ascorbic Acid Deficiency Weakens Collagen Fibers Throughout
in Chapter 32. However, vitamin B12 and folic acid
the Body. Ascorbic acid is essential for activating the
each perform specific and different chemical functions
enzyme prolyl hydroxylase, which promotes the hydrox-
in promoting growth and maturation of red blood
ylation step in the formation of hydroxyproline, an inte-
cells. One of the significant effects of folic acid
gral constituent of collagen. Without ascorbic acid, the
deficiency is the development of macrocytic anemia,
collagen fibers that are formed in virtually all tissues of
almost identical to that which occurs in pernicious
the body are defective and weak. Therefore, this vitamin
anemia. This often can be treated effectively with folic
is essential for the growth and strength of the fibers in
acid alone.
subcutaneous tissue, cartilage, bone, and teeth.
878
Unit XIII
Metabolism and Temperature Regulation
Ascorbic Acid Deficiency Causes Scurvy. Deficiency of ascor-
absence of vitamin E, the quantity of unsaturated fats
bic acid for 20 to 30 weeks, which occurred frequently
in the cells becomes diminished, causing abnormal
during long ship voyages in the past, causes scurvy. One
structure and function of such cellular organelles as
of the most important effects of scurvy is failure of
the mitochondria, the lysosomes, and even the cell
wounds to heal. This is caused by failure of the cells to
membrane.
deposit collagen fibrils and intercellular cement sub-
stances. As a result, healing of a wound may require
several months instead of the several days ordinarily
Vitamin K
necessary.
Vitamin K is necessary for the formation by the liver of
Lack of ascorbic acid also causes cessation of bone
prothrombin, Factor VII (proconvertin), Factor IX, and
growth. The cells of the growing epiphyses continue to
Factor X, all of which are important in blood coagula-
proliferate, but no new collagen is laid down between
tion. Therefore, when vitamin K deficiency occurs, blood
the cells, and the bones fracture easily at the point of
clotting is retarded. The function of this vitamin and
growth because of failure to ossify. Also, when an
its relation to some of the anticoagulants, such as
already ossified bone fractures in a person with ascor-
dicumarol, are presented in greater detail in Chapter 35.
bic acid deficiency, the osteoblasts cannot form new
Several compounds, both natural and synthetic,
bone matrix. Consequently, the fractured bone does not
exhibit vitamin K activity. Because vitamin K is synthe-
heal.
sized by bacteria in the colon, it is rare for a person to
The blood vessel walls become extremely fragile in
have a bleeding tendency because of vitamin K defi-
scurvy because of (1) failure of the endothelial cells to
ciency in the diet. However, when the bacteria of the
be cemented together properly and (2) failure to form
colon are destroyed by the administration of large quan-
the collagen fibrils normally present in vessel walls.
tities of antibiotic drugs, vitamin K deficiency occurs
The capillaries are especially likely to rupture, and as
rapidly because of the paucity of this compound in the
a result, many small petechial hemorrhages occur
normal diet.
throughout the body. The hemorrhages beneath the
skin cause purpuric blotches, sometimes over the entire
body. To test for ascorbic acid deficiency, one can
Mineral Metabolism
produce such petechial hemorrhages by inflating a
blood pressure cuff over the upper arm; this occludes
The functions of many of the minerals, such as sodium,
the venous return of blood, the capillary pressure rises,
potassium, and chloride, are presented at appropriate
and red blotches occur on the forearm if the ascorbic
points in the text. Only specific functions of minerals
acid deficiency is sufficiently severe.
not covered elsewhere are mentioned here. The body
In extreme scurvy, the muscle cells sometimes frag-
content of the most important minerals is listed in Table
ment; lesions of the gums occur, with loosening of the
71-4, and the daily requirements of these are given in
teeth; infections of the mouth develop; and vomiting of
Table 71-5.
blood, bloody stools, and cerebral hemorrhage can all
occur. Finally, high fever often develops before death.
Magnesium. Magnesium is about one sixth as plentiful in
cells as potassium. Magnesium is required as a catalyst
for many intracellular enzymatic reactions, particularly
Vitamin D
those related to carbohydrate metabolism.
The extracellular fluid magnesium concentration is
Vitamin D increases calcium absorption from the gas-
slight, only 1.8 to 2.5 mEq/L. Increased extracellular
trointestinal tract and helps control calcium deposition in
concentration of magnesium depresses nervous system
the bone. The mechanism by which vitamin D increases
activity as well as skeletal muscle contraction. This latter
calcium absorption is mainly to promote active trans-
effect can be blocked by the administration of calcium.
port of calcium through the epithelium of the ileum. In
Low magnesium concentration causes increased irri-
particular, it increases the formation of a calcium-
tability of the nervous system, peripheral vasodilation,
binding protein in the intestinal epithelial cells that aids
in calcium absorption. The specific functions of vitamin
D in relation to overall body calcium metabolism and
bone formation are presented in Chapter 79.
Table 71-4
Average Content of a 70-Kilogram Man
Vitamin E
Constituent
Amount (grams)
Several related compounds exhibit so-called vitamin E
activity. Only rare instances of proved vitamin E defi-
Water
41,400
ciency have occurred in human beings. In experimental
Fat
12,600
animals, lack of vitamin E can cause degeneration of the
Protein
12,600
germinal epithelium in the testis and, therefore, can
Carbohydrate
300
cause male sterility. Lack of vitamin E can also cause
Sodium
63
resorption of a fetus after conception in the female.
Potassium
150
Calcium
1,160
Because of these effects of vitamin E deficiency, vitamin
Magnesium
21
E is sometimes called the “antisterility vitamin.” Defi-
Chloride
85
ciency of vitamin E prevents normal growth and some-
Phosphorus
670
times causes degeneration of the renal tubular cells and
Sulfur
112
the muscle cells.
Iron
3
Vitamin E is believed to play a protective role in the
Iodine
0.014
prevention of oxidation of unsaturated fats. In the
Chapter 71
Dietary Balances; Regulation of Feeding; Obesity and Starvation; Vitamins and Minerals
879
with the formation and function of thyroid hormone; as
Table 71-5
shown in Table 71-4, the entire body contains an
Average Required Daily Amounts of Minerals for Adults
average of only 14 milligrams. Iodine is essential for the
formation of thyroxine and triiodothyronine, the two
thyroid hormones that are essential for maintenance of
Mineral
Amount
normal metabolic rates in all cells of the body.
Sodium
3.0 g
Potassium
1.0 g
Zinc. Zinc is an integral part of many enzymes, one of
Chloride
3.5 g
the most important of which is carbonic anhydrase,
Calcium
1.2 g
present in especially high concentration in the red blood
Phosphorus
1.2 g
cells. This enzyme is responsible for rapid combination
Iron
18.0 mg
of carbon dioxide with water in the red blood cells of
Iodine
150.0 mg
the peripheral capillary blood and for rapid release of
Magnesium
0.4 g
carbon dioxide from the pulmonary capillary blood into
Cobalt
Unknown
Copper
Unknown
the alveoli. Carbonic anhydrase is also present to a
Manganese
Unknown
major extent in the gastrointestinal mucosa, the tubules
Zinc
15 mg
of the kidney, and the epithelial cells of many glands of
the body. Consequently, zinc in small quantities is essen-
tial for the performance of many reactions related to
carbon dioxide metabolism.
Zinc is also a component of lactic dehydrogenase and
and cardiac arrhythmias, especially after acute myocar-
is therefore important for the interconversions between
dial infarction.
pyruvic acid and lactic acid. Finally, zinc is a component
of some peptidases and is important for the digestion of
Calcium. Calcium is present in the body mainly in the
proteins in the gastrointestinal tract.
form of calcium phosphate in the bone. This subject is
discussed in detail in Chapter 79, as is the calcium
Fluorine. Fluorine does not seem to be a necessary
content of extracellular fluid. Excess quantities of
element for metabolism, but the presence of a small
calcium ions in extracellular fluid can cause the heart to
quantity of fluorine in the body during the period of life
stop in systole and can act as a mental depressant. At
when the teeth are being formed subsequently protects
the other extreme, low levels of calcium can cause spon-
against caries. Fluorine does not make the teeth them-
taneous discharge of nerve fibers, resulting in tetany, as
selves stronger but has a poorly understood effect in
discussed in Chapter 79.
suppressing the cariogenic process. It has been sug-
gested that fluorine is deposited in the hydroxyapatite
Phosphorus. Phosphate is the major anion of intracellular
crystals of the tooth enamel and combines with and
fluid. Phosphates have the ability to combine reversibly
therefore blocks the functions of various trace metals
with many coenzyme systems and with multiple other
that are necessary for activation of the bacterial
compounds that are necessary for the operation of
enzymes that cause caries. Therefore, when fluorine is
metabolic processes. Many important reactions of phos-
present, the enzymes remain inactive and cause no
phates have been catalogued at other points in this text,
caries.
especially in relation to the functions of adenosine
Excessive intake of fluorine causes fluorosis, which
triphosphate, adenosine diphosphate, phosphocreatine,
manifests in its mild state by mottled teeth and in its
and so forth. Also, bone contains a tremendous amount
more severe state by enlarged bones. It has been pos-
of calcium phosphate, which is discussed in Chapter 79.
tulated that in this condition, fluorine combines with
trace metals in some of the metabolic enzymes, includ-
Iron. The function of iron in the body, especially in rela-
ing the phosphatases, so that various metabolic systems
tion to the formation of hemoglobin, is discussed in
become partially inactivated. According to this theory,
Chapter 32. Two thirds of the iron in the body is in the
the mottled teeth and enlarged bones are due to ab-
form of hemoglobin, although smaller quantities are
normal enzyme systems in the odontoblasts and
present in other forms, especially in the liver and the
osteoblasts. Even though the mottled teeth are highly
bone marrow. Electron carriers containing iron (espe-
resistant to the development of caries, the structural
cially the cytochromes) are present in the mitochondria
strength of these teeth may be considerably lessened by
of all cells of the body and are essential for most of the
the mottling process.
oxidation that occurs in the cells. Therefore, iron is
absolutely essential for both the transport of oxygen to
the tissues and the operation of oxidative systems within
References
the tissue cells, without which life would cease within a
few seconds.
Barsh GS, Schwartz MW: Genetic approaches to studying
energy balance: perception and integration. Nat Rev
Important Trace Elements in the Body. A few elements are
Genet 3:589, 2002.
present in the body in such small quantities that they
Coll AP, Farooqi IS, Challis BG, et al: Proopiomelanocortin
are called trace elements. The amounts of these elements
and energy balance: insights from human and murine
in foods are also usually minute. Yet without any one of
genetics. J Clin Endocrinol Metab 89:2557, 2004.
them, a specific deficiency syndrome is likely to develop.
Cowley MA, Cone RD, Enriori P, et al: Electrophysiological
Three of the most important are iodine, zinc, and
actions of peripheral hormones on melanocortin neurons.
fluorine.
Ann N Y Acad Sci 994:175, 2003.
Iodine. The best known of the trace elements is iodine.
Cowley MA, Grove KL: Ghrelin—satisfying a hunger for the
This element is discussed in Chapter 76 in connection
mechanism. Endocrinology 145:2604, 2004.
880
Unit XIII
Metabolism and Temperature Regulation
da Silva AA, Kuo JJ, Hall JE: Role of hypothalamic
Lucock M: Is folic acid the ultimate functional food compo-
melanocortin 3/4-receptors in mediating chronic cardio-
nent for disease prevention? BMJ 328:211, 2004.
vascular, renal, and metabolic actions of leptin. Hyperten-
National Institutes of Health: Clinical Guidelines on the
sion 43:1312, 2004.
Identification, Evaluation, and Treatment of Overweight
Davy KP, Hall JE: Obesity and hypertension: two epidemics
and Obesity in Adults: The Evidence Report. Bethesda
or one? Am J Physiol Regul Integr Comp Physiol
MD: National Heart, Lung, and Blood Institute and
286:R803, 2004.
National Institute of Diabetes and Digestive and Kidney
Druce MR, Small CJ, Bloom SR: Gut peptides regulating
Diseases,
satiety. Endocrinology 145:2660, 2004.
guidelines/index.htm.
Dutta A, Dutta SK: Vitamin E and its role in the prevention
O’Rahilly S, Farooqi IS, Yeo GS, Challis BG: Human
of atherosclerosis and carcinogenesis: a review. J Am Coll
obesity—lessons from monogenic disorders. Endocrinol-
Nutr 22:258, 2003.
ogy 144:3757, 2003.
Flier JS: Obesity wars: molecular progress confronts an
Powers HJ: Riboflavin (vitamin B2) and health. Am J Clin
expanding epidemic. Cell 116:337, 2004.
Nutr 77:1352, 2003.
Fraser PD, Bramley PM: The biosynthesis and nutritional
Ravussin E: Cellular sensors of feast and famine. J Clin
uses of carotenoids. Prog Lipid Res 43:228, 2004.
Invest 109:1537, 2002.
Friedman JM, Halaas JL: Leptin and the regulation of body
Rindi G, Laforenza U: Thiamine intestinal transport and
weight in mammals. Nature 395:763, 1998.
related issues: recent aspects. Proc Soc Exp Biol Med
Grundy SM: Obesity, metabolic syndrome, and cardiovascu-
224:246, 2000.
lar disease. J Clin Endocrinol Metab 89:2595, 2004.
Ross SA, McCaffery PJ, Drager UC, De Luca LM: Retinoids
Hall JE, Henegar JR, Dwyer TM, et al: Is obesity a major
in embryonal development. Physiol Rev 80:1021, 2000.
cause of chronic kidney disease? Adv Ren Replace Ther
Said HM: Recent advances in carrier-mediated intestinal
11:41, 2004.
absorption of water-soluble vitamins. Annu Rev Physiol
Hall JE, Jones DW: What can we do about the “epidemic”
66:419, 2004.
of obesity. Am J Hypertens 15:657, 2002.
Seeley R, Woods S: Monitoring of stored and available fuel
Hall JE, Jones DW, Kuo JJ, et al: Impact of the obesity epi-
by the CNS: implications for obesity. Nat Rev Neurosci 4:
demic on hypertension and renal disease. Curr Hypertens
901, 2003.
Rep 5:386, 2003.
Stanley S, Wynne K, Bloom S: Gastrointestinal satiety
Jequier E, Tappy L: Regulation of body weight in humans.
signals. III. Glucagon-like peptide
1, oxyntomodulin,
Physiol Rev 79:451, 1999.
peptide YY, and pancreatic polypeptide. Am J Physiol
Jones G, Strugnell SA, DeLuca HF: Current understanding
Gastrointest Liver Physiol 286:G693, 2004.
of the molecular actions of vitamin D. Physiol Rev
Wisse BE, Schwartz MW, Cummings DE: Melanocortin sig-
78:1193, 1998.
naling and anorexia in chronic disease states. Ann N Y
Kershaw EE, Flier JS: Adipose tissue as an endocrine organ.
Acad Sci 994:275, 2003.
J Clin Endocrinol Metab 89:2548, 2004.
Woods SC: Gastrointestinal satiety signals. I. An overview of
Korner J, Aronne LJ: Pharmacological approaches to weight
gastrointestinal signals that influence food intake. Am J
reduction: therapeutic targets. J Clin Endocrinol Metab
Physiol Gastrointest Liver Physiol 286:G7, 2004.
89:2616, 2004.
Wynne K, Stanley S, Bloom S: The gut and regulation of body
Korner J, Leibel RL: To eat or not to eat—how the gut talks
weight. J Clin Endocrinol Metab 89:2576, 2004.
to the brain. N Engl J Med 349:926, 2003.
Kuo JJ, Silva AA, Hall JE: Hypothalamic melanocortin
receptors and chronic regulation of arterial pressure and
renal function. Hypertension 41:768, 2003.
C H A P T E R
7
2
Energetics and Metabolic Rate
Adenosine Triphosphate
(ATP) Functions as
an “Energy Currency”
in Metabolism
In the past few chapters, we have pointed out that car-
bohydrates, fats, and proteins can all be used by cells to
synthesize large quantities of adenosine triphosphate
(ATP), which can be used as an energy source for almost all other cellular func-
tions. For this reason, ATP has been called an energy “currency” in cell metabolism.
Indeed, the transfer of energy from foodstuffs to most functional systems of the cells
can be done only through this medium of ATP (or the similar nucleotide guanosine
triphosphate, GTP). Many of the attributes of ATP are presented in Chapter 2.
An attribute of ATP that makes it highly valuable as an energy currency is the
large quantity of free energy (about 7300 calories, or 7.3 Calories [kilocalories], per
mole under standard conditions, but as much as 12,000 calories under physiologic
conditions) vested in each of its two high-energy phosphate bonds. The amount of
energy in each bond, when liberated by decomposition of ATP, is enough to cause
almost any step of any chemical reaction in the body to take place if appropriate
energy transfer is achieved. Some chemical reactions that require ATP energy use
only a few hundred of the available 12,000 calories, and the remainder of this energy
is lost in the form of heat.
ATP Is Generated by Combustion of Carbohydrates, Fats, and Proteins. In previous chapters,
we discussed the transfer of energy from various foods to ATP. To summarize, ATP
is produced from
1. Combustion of carbohydrates—mainly glucose, but also smaller amounts of
other sugars such as fructose; this occurs in the cytoplasm of the cell through
the anaerobic process of glycolysis and in the cell mitochondria through the
aerobic citric acid (Krebs) cycle.
2. Combustion of fatty acids in the cell mitochondria by beta-oxidation.
3. Combustion of proteins, which requires hydrolysis to their component amino
acids and degradation of the amino acids to intermediate compounds of the
citric acid cycle and then to acetyl coenzyme A and carbon dioxide.
ATP Energizes the Synthesis of Most Important Cellular Components. Among the most impor-
tant intracellular processes that require ATP energy is the formation of peptide link-
ages between amino acids during the synthesis of proteins. The different peptide
linkages, depending on which types of amino acids are linked, require from 500 to
5000 calories of energy per mole. It will be recalled from the discussion of protein
synthesis in Chapter 3 that four high-energy phosphate bonds are expended during
the cascade of reactions required to form each peptide linkage. This provides a total
of 48,000 calories of energy, which is far more than the 500 to 5000 calories even-
tually stored in each of the peptide linkages.
ATP energy is also used in the synthesis of glucose from lactic acid and in the syn-
thesis of fatty acids from acetyl coenzyme A. In addition, ATP energy is used for the
synthesis of cholesterol, phospholipids, the hormones, and almost all other substances
of the body. Even the urea excreted by the kidneys requires ATP to cause its
formation from ammonia. One might wonder about the advisability of expending
energy to form urea, which is simply discarded by the body. However, remembering
the extreme toxicity of ammonia in the body fluids, one can see the value of this reac-
tion, which keeps the ammonia concentration of the body fluids at a low level.
ATP Energizes Muscle Contraction. Muscle contraction will not occur without energy
from ATP. Myosin, one of the important contractile proteins of the muscle fiber, acts
as an enzyme to cause breakdown of ATP into adenosine diphosphate (ADP), thus
881
882
Unit XIII
Metabolism and Temperature Regulation
releasing the energy required to cause contraction. Only
CH3
NH
H
O
a small amount of ATP is normally degraded in muscles
when muscle contraction is not occurring, but this rate
HOOC CH
N
C
N
~
P OH
2
of ATP usage can rise to at least 150 times the resting
O
level during short bursts of maximal contraction. The
H
postulated mechanism by which ATP energy is used to
cause muscle contraction is discussed in Chapter 6.
and the functional cellular systems, but it can transfer
ATP Energizes Active Transport Across Membranes. In
energy interchangeably with ATP. When extra amounts
Chapters 4, 27, and 65, active transport of electrolytes
of ATP are available in the cell, much of its energy is
and various nutrients across cell membranes and from
used to synthesize phosphocreatine, thus building up
the renal tubules and gastrointestinal tract into the
this storehouse of energy. Then, when the ATP begins
blood is discussed. In each instance, we noted that active
to be used up, the energy in the phosphocreatine is
transport of most electrolytes and substances such as
transferred rapidly back to ATP and then to the func-
glucose, amino acids, and acetoacetate can occur against
tional systems of the cells. This reversible interrelation
an electrochemical gradient, even though the natural
between ATP and phosphocreatine is demonstrated by
diffusion of the substances would be in the opposite
the following equation:
direction. To oppose the electrochemical gradient
Phosphocreatine + ADP
requires energy, which is provided by ATP.
Ø≠
ATP + Creatine
ATP Energizes Glandular Secretion. The same principles
Note particularly that the higher energy level of the
apply to glandular secretion as to the absorption of
high-energy phosphate bond in phosphocreatine (1000
substances against concentration gradients, because
to 1500 calories per mole greater than that in ATP)
energy is required to concentrate substances as they are
causes the reaction between phosphocreatine and ADP
secreted by the glandular cells. In addition, energy is
to proceed rapidly toward the formation of new ATP
required to synthesize the organic compounds to be
every time even the slightest amount of ATP expends
secreted.
its energy elsewhere. Therefore, the slightest usage of
ATP by the cells calls forth the energy from the phos-
ATP Energizes Nerve Conduction. The energy used during
phocreatine to synthesize new ATP. This effect keeps
propagation of a nerve impulse is derived from
the concentration of ATP at an almost constant high
the potential energy stored in the form of concentration
level as long as any phosphocreatine remains. For this
differences of ions across the membranes. That is, a
reason, we can call the ATP-phosphocreatine system an
high concentration of potassium inside the fiber and a
ATP “buffer” system. One can readily understand the
low concentration outside the fiber constitute a type
importance of keeping the concentration of ATP nearly
of energy storage. Likewise, a high concentration
constant, because the rates of almost all the metabolic
of sodium on the outside of the membrane and a low
reactions in the body depend on this constancy.
concentration on the inside represent another store
of energy. The energy needed to pass each action
Anaerobic Versus Aerobic Energy
potential along the fiber membrane is derived from
this energy storage, with small amounts of potassium
Anaerobic energy means energy that can be derived
transferring out of the cell and sodium into the cell
from foods without the simultaneous utilization of
during each of the action potentials. However, active
oxygen; aerobic energy means energy that can be
transport systems energized by ATP then retransport
derived from foods only by oxidative metabolism. In the
the ions back through the membrane to their former
discussions in Chapters 67 through 69, it is noted that
positions.
carbohydrates, fats, and proteins can all be oxidized to
cause synthesis of ATP. However, carbohydrates are the
only significant foods that can be used to provide energy
Phosphocreatine Functions as an
without the utilization of oxygen; this energy release
Accessory Storage Depot for
occurs during glycolytic breakdown of glucose or glyco-
gen to pyruvic acid. For each mole of glucose that is split
Energy and as an “ATP Buffer”
into pyruvic acid, 2 moles of ATP are formed. However,
Despite the paramount importance of ATP as a cou-
when stored glycogen in a cell is split to pyruvic acid,
pling agent for energy transfer, this substance is not the
each mole of glucose in the glycogen gives rise to 3
most abundant store of high-energy phosphate bonds in
moles of ATP. The reason for this difference is that free
the cells. Phosphocreatine, which also contains high-
glucose entering the cell must be phosphorylated by
energy phosphate bonds, is three to eight times as
using 1 mole of ATP before it can begin to be split; this
abundant. Also, the high-energy bond (~) of phospho-
is not true of glucose derived from glycogen because it
creatine contains about 8500 calories per mole under
comes from the glycogen already in the phosphorylated
standard conditions and as much as 13,000 calories per
state, without the additional expenditure of ATP. Thus,
mole under conditions in the body (37°C and low con-
the best source of energy under anaerobic conditions is
centrations of the reactants). This is slightly greater than
the stored glycogen of the cells.
the 12,000 calories per mole in each of the two high-
energy phosphate bonds of ATP. The formula for crea-
Anaerobic Energy Utilization During Hypoxia. One of the prime
tinine phosphate is the following:
examples of anaerobic energy utilization occurs in acute
Unlike ATP, phosphocreatine cannot act as a direct
hypoxia. When a person stops breathing, there is already
coupling agent for energy transfer between the foods
a small amount of oxygen stored in the lungs and an
Chapter 72
Energetics and Metabolic Rate
883
additional amount stored in the hemoglobin of the
Oxygen Debt Is the Extra Consumption of Oxygen After Completion
blood. This oxygen is sufficient to keep the metabolic
of Strenuous Exercise. After a period of strenuous exer-
processes functioning for only about 2 minutes. Contin-
cise, a person continues to breathe hard and to consume
ued life beyond this time requires an additional source
large amounts of oxygen for at least a few minutes and
of energy. This can be derived for another minute or so
sometimes for as long as 1 hour thereafter. This addi-
from glycolysis—that is, the glycogen of the cells split-
tional oxygen is used (1) to reconvert the lactic acid that
ting into pyruvic acid, and the pyruvic acid becoming
has accumulated during exercise back into glucose, (2)
lactic acid, which diffuses out of the cells, as described
to reconvert adenosine monophosphate and ADP to
in Chapter 67.
ATP, (3) to reconvert creatine and phosphate to phos-
phocreatine, (4) to re-establish normal concentrations
Anaerobic Energy Utilization During Strenuous Bursts of Activity
of oxygen bound with hemoglobin and myoglobin, and
Is Derived Mainly from Glycolysis. Skeletal muscles can
(5) to raise the concentration of oxygen in the lungs to
perform extreme feats of strength for a few seconds but
its normal level. This extra consumption of oxygen after
are much less capable during prolonged activity. Most
exercise is over is called repaying the oxygen debt.
of the extra energy required during these bursts of activ-
The principle of oxygen debt is discussed further in
ity cannot come from the oxidative processes because
Chapter 84 in relation to sports physiology; the ability
they are too slow to respond. Instead, the extra energy
of a person to build up an oxygen debt is especially
comes from anaerobic sources: (1) ATP already present
important in many types of athletics.
in the muscle cells, (2) phosphocreatine in the cells, and
(3) anaerobic energy released by glycolytic breakdown
of glycogen to lactic acid.
Summary of Energy Utilization
The maximum amount of ATP in muscle is only about
by the Cells
5 mmol/L of intracellular fluid, and this amount can
maintain maximum muscle contraction for no more
With the background of the past few chapters and of the
than a second or so. The amount of phosphocreatine in
preceding discussion, we can now synthesize a compos-
the cells is three to eight times this amount, but even by
ite picture of overall energy utilization by the cells, as
using all the phosphocreatine, maximum contraction
shown in Figure 72-1. This figure demonstrates the
can be maintained for only 5 to 10 seconds.
anaerobic utilization of glycogen and glucose to form
Release of energy by glycolysis can occur much more
ATP and the aerobic utilization of compounds derived
rapidly than can oxidative release of energy. Conse-
from carbohydrates, fats, proteins, and other substances
quently, most of the extra energy required during stren-
to form additional ATP. In turn, ATP is in reversible
uous activity that lasts for more than 5 to 10 seconds but
equilibrium with phosphocreatine in the cells, and
less than 1 to 2 minutes is derived from anaerobic gly-
because larger quantities of phosphocreatine are
colysis. As a result, the glycogen content of muscles
present in the cells than ATP, much of the cells’ stored
during strenuous bouts of exercise is reduced, whereas
energy is in this energy storehouse.
the lactic acid concentration of the blood rises. After the
Energy from ATP can be used by the different func-
exercise is over, oxidative metabolism is used to recon-
tioning systems of the cells to provide for synthesis
vert about four fifths of the lactic acid into glucose;
and growth, muscle contraction, glandular secretion,
the remainder becomes pyruvic acid and is degraded
nerve impulse conduction, active absorption, and other
and oxidized in the citric acid cycle. The reconversion
cellular activities. If greater amounts of energy are
to glucose occurs principally in the liver cells, and
demanded for cellular activities than can be provided
the glucose is then transported in the blood back to the
by oxidative metabolism, the phosphocreatine store-
muscles, where it is stored once more in the form of
house is used first, and then anaerobic breakdown of
glycogen.
glycogen follows rapidly. Thus, oxidative metabolism
Glycogen
Energy for
Glucose
1. Synthesis and growth
2. Muscular contraction
ATP
3. Glandular secretion
4. Nerve conduction
Lactic acid
Pyruvic acid
5. Active absorption
6. Etc.
Acetyl-CoA
Deaminated
Phosphocreatine
Figure 72-1
amino acids
Overall schema of energy trans-
AMP
fer from foods to the adenylic
Other substrates
acid system and then to the
functional elements of the cells.
(Modified from Soskin S, Levine
CO2 + H2O
Creatine + PO4
R: Carbohydrate Metabolism.
Chicago: University of Chicago
Press, 1946, 1952.)
884
Unit XIII
Metabolism and Temperature Regulation
cannot deliver bursts of extreme energy to the cells
reabsorption, because the transport enzymes are satu-
nearly as rapidly as the anaerobic processes can, but at
rated. Under these conditions, the rate of reabsorption
slower rates of usage, the oxidative processes can con-
of the glucose is limited by the concentration of the
tinue as long as energy stores (mainly fat) exist.
transport enzymes in the proximal tubular cells, not by
the concentration of the glucose itself.
Control of Energy Release
Role of Substrate Concentration in Regulation of Meta-
bolic Reactions. Note also in Figure 72-2 that when the
in the Cell
substrate concentration becomes low enough that only
Rate Control of Enzyme-Catalyzed Reactions. Before dis-
a small portion of the enzyme is required in the reac-
cussing the control of energy release in the cell, it is nec-
tion, the rate of the reaction becomes directly propor-
essary to consider the basic principles of rate control of
tional to the substrate concentration as well as the
enzymatically catalyzed chemical reactions, which are
enzyme concentration. This is the relationship seen in
the types of reactions that occur almost universally
the absorption of substances from the intestinal tract
throughout the body.
and renal tubules when their concentrations are low.
The mechanism by which an enzyme catalyzes a
chemical reaction is for the enzyme first to combine
Rate Limitation in a Series of Reactions. Almost all chemical
loosely with one of the substrates of the reaction. This
reactions of the body occur in series, with the product
alters the bonding forces on the substrate sufficiently so
of one reaction acting as a substrate for the next reac-
that it can react with other substances. Therefore, the
tion, and so on. Therefore, the overall rate of a complex
rate of the overall chemical reaction is determined by
series of chemical reactions is determined mainly by the
both the concentration of the enzyme and the concen-
rate of reaction of the slowest step in the series. This is
tration of the substrate that binds with the enzyme. The
called the rate-limiting step in the entire series.
basic equation expressing this concept is as follows:
K1¥[Enzyme]¥[Substrate]
ADP Concentration as a Rate-Controlling Factor in Energy
Rate of reaction
=
Release. Under resting conditions, the concentration of
K2+[Substrate]
ADP in the cells is extremely slight, so that the chemi-
This is called the Michaelis-Menten equation. Figure
cal reactions that depend on ADP as one of the sub-
72-2 shows the application of this equation.
strates are quite slow. They include all the oxidative
metabolic pathways that release energy from food, as
Role of Enzyme Concentration in Regulation of Metabolic
well as essentially all other pathways for the release of
Reactions. Figure 72-2 shows that when the substrate is
energy in the body. Thus, ADP is a major rate-limiting
present in high concentration, as shown in the right half
factor for almost all energy metabolism of the body.
of the figure, the rate of a chemical reaction is deter-
When the cells become active, regardless of the type
mined almost entirely by the concentration of the
of activity, ATP is converted into ADP, increasing the
enzyme. Thus, as the enzyme concentration increases
concentration of ADP in direct proportion to the degree
from an arbitrary value of 1 up to 2, 4, or 8, the rate of
of activity of the cell. This ADP then automatically
the reaction increases proportionately, as demonstrated
increases the rates of all the reactions for the metabolic
by the rising levels of the curves. As an example, when
release of energy from food. Thus, by this simple
large quantities of glucose enter the renal tubules in a
process, the amount of energy released in the cell is con-
person with diabetes mellitus—that is, the substrate
trolled by the degree of activity of the cell. In the
glucose is in great excess in the tubules—further
absence of cellular activity, the release of energy stops
increases in tubular glucose have little effect on glucose
because all the ADP soon becomes ATP.
Metabolic Rate
The metabolism of the body simply means all the chem-
8
ical reactions in all the cells of the body, and the meta-
bolic rate is normally expressed in terms of the rate of
heat liberation during chemical reactions.
Heat Is the End Product of Almost All the Energy Released in the
4
Body. In discussing many of the metabolic reactions in
the preceding chapters, we noted that not all the energy
in foods is transferred to ATP; instead, a large portion
2
of this energy becomes heat. On average, 35 per cent of
1
the energy in foods becomes heat during ATP forma-
tion. Then, still more energy becomes heat as it is trans-
ferred from ATP to the functional systems of the cells,
Substrate concentration
so that even under optimal conditions, no more than 27
per cent of all the energy from food is finally used by
the functional systems.
Even when 27 per cent of the energy reaches the func-
tional systems of the cells, most of this eventually
Figure 72-2
becomes heat. For example, when proteins are syn-
Effect of substrate and enzyme concentrations on the rate of
thesized, large portions of ATP are used to form the
enzyme-catalyzed reaction.
peptide linkages, and this stores energy in these link-
Chapter 72
Energetics and Metabolic Rate
885
ages. But there is also continuous turnover of proteins—
Indirect Calorimetry—The
“Energy Equivalent” of Oxygen.
some being degraded while others are being formed.
Because more than 95 per cent of the energy expended
When proteins are degraded, the energy stored in the
in the body is derived from reactions of oxygen with the
peptide linkages is released in the form of heat into the
different foods, the whole-body metabolic rate can also
body.
be calculated with a high degree of accuracy from the
Another example is the energy used for muscle activ-
rate of oxygen utilization. When 1 liter of oxygen is
ity. Much of this energy simply overcomes the viscosity
metabolized with glucose, 5.01 Calories of energy are
of the muscles themselves or of the tissues so that the
released; when metabolized with starches, 5.06 Calories
limbs can move. This viscous movement causes friction
are released; with fat, 4.70 Calories; and with protein,
within the tissues, which generates heat.
4.60 Calories.
Consider also the energy expended by the heart in
Using these figures, it is striking how nearly equiva-
pumping blood. The blood distends the arterial system,
lent are the quantities of energy liberated per liter of
and this distention itself represents a reservoir of poten-
oxygen, regardless of the type of food being metabo-
tial energy. As the blood flows through the peripheral
lized. For the average diet, the quantity of energy liber-
vessels, the friction of the different layers of blood
ated per liter of oxygen used in the body averages about
flowing over one another and the friction of the blood
4.825 Calories. This is called the energy equivalent of
against the walls of the vessels turn all this energy into
oxygen; using this energy equivalent, one can calculate
heat.
with a high degree of precision the rate of heat libera-
Essentially all the energy expended by the body is
tion in the body from the quantity of oxygen used in a
eventually converted into heat. The only significant
given period of time.
exception occurs when the muscles are used to perform
If a person metabolizes only carbohydrates during
some form of work outside the body. For instance, when
the period of the metabolic rate determination, the cal-
the muscles elevate an object to a height or propel the
culated quantity of energy liberated, based on the value
body up steps, a type of potential energy is created by
for the average energy equivalent of oxygen (4.825
raising a mass against gravity. But when external expen-
Calories/L), would be about 4 per cent too little. Con-
diture of energy is not taking place, all the energy
versely, if the person obtains most energy from fat, the
released by the metabolic processes eventually becomes
calculated value would be about 4 per cent too great.
body heat.
Energy Metabolism—
The Calorie. To discuss the metabolic rate of the body
Factors That Influence
and related subjects intelligently, it is necessary to use
some unit for expressing the quantity of energy released
Energy Output
from the different foods or expended by the different
As discussed in Chapter 71, energy intake is balanced
functional processes of the body. Most often, the Calorie
with energy output in healthy adults who maintain a
is the unit used for this purpose. It will be recalled that
stable body weight. About 45 per cent of daily energy
1 calorie—spelled with a small “c” and often called a
intake is derived from carbohydrates,
40 per cent
gram calorie—is the quantity of heat required to raise
from fats, and 15 per cent from proteins in the average
the temperature of 1 gram of water 1°C. The calorie is
American diet. Energy output can also be partitioned
much too small a unit when referring to energy in the
into several measurable components, including energy
body. Consequently, the Calorie—sometimes spelled
used for (1) performing essential metabolic functions of
with a capital “C” and often called a kilocalorie, which
the body (the “basal” metabolic rate); (2) performing
is equivalent to 1000 calories—is the unit ordinarily
various physical activities; (3) digesting, absorbing, and
used in discussing energy metabolism.
processing food; and (4) maintaining body temperature.
Measurement of the Whole-Body
Overall Energy Requirements
Metabolic Rate
for Daily Activities
Direct Calorimetry Measures Heat Liberated from the Body.
An average man who weighs 70 kilograms and lies in
Because a person ordinarily is not performing any
bed all day uses about 1650 Calories of energy. The
external work, the whole-body metabolic rate can be
process of eating and digesting food increases the
determined by simply measuring the total quantity of
amount of energy used each day by an additional 200
heat liberated from the body in a given time.
or more Calories, so that the same man lying in bed and
In determining the metabolic rate by direct calorime-
eating a reasonable diet requires a dietary intake of
try, one measures the quantity of heat liberated from the
about 1850 Calories per day. If he sits in a chair all day
body in a large, specially constructed calorimeter. The
without exercising, his total energy requirement reaches
subject is placed in an air chamber that is so well insu-
2000 to 2250 Calories. Therefore, the approximate daily
lated that no heat can leak through the walls of the
energy requirement for a very sedentary man perform-
chamber. Heat formed by the subject’s body warms the
ing only essential functions is 2000 Calories.
air of the chamber. However, the air temperature within
The amount of energy used to perform daily physical
the chamber is maintained at a constant level by forcing
activities is normally about 25 per cent of the total
the air through pipes in a cool water bath. The rate of
energy expenditure, but it can vary markedly in differ-
heat gain by the water bath, which can be measured with
ent individuals, depending on the type and amount of
an accurate thermometer, is equal to the rate at which
physical activity. For example, walking up stairs requires
heat is liberated by the subject’s body.
about 17 times as much energy as lying in bed asleep.
Direct calorimetry is physically difficult to perform
In general, over a 24-hour period, a person performing
and is used only for research purposes.
heavy labor can achieve a maximal rate of energy uti-
886
Unit XIII
Metabolism and Temperature Regulation
lization as great as 6000 to 7000 Calories, or as much as
reason, BMR is usually corrected for differences in
3.5 times the energy used under conditions of no phys-
body size by expressing it as Calories per hour per
ical activity.
square meter of body surface area, calculated from
height and weight. The average values for males and
females of different ages are shown in Figure 72-4.
Basal Metabolic Rate (BMR)—
Much of the decline in BMR with increasing age is
The Minimum Energy Expenditure
probably related to loss of muscle mass and replace-
for the Body to Exist
ment of muscle with adipose tissue, which has a lower
rate of metabolism. Likewise, slightly lower BMRs in
Even when a person is at complete rest, considerable
women, compared with men, are due partly to their
energy is required to perform all the chemical reactions
lower percentage of muscle mass and higher percentage
of the body. This minimum level of energy required
of adipose tissue. However, there are other factors that
to exist is called the basal metabolic rate (BMR) and
can influence the BMR, as discussed next.
accounts for about 50 to 70 per cent of the daily energy
expenditure in most sedentary individuals (Figure 72-3).
Thyroid Hormone Increases Metabolic Rate. When the thyroid
Because the level of physical activity is highly vari-
gland secretes maximal amounts of thyroxine, the meta-
able among different individuals, measurement of
bolic rate sometimes rises 50 to 100 per cent above
the BMR provides a useful means of comparing one
normal. Conversely, total loss of thyroid secretion
person’s metabolic rate with that of another. The usual
decreases the metabolic rate to 40 to 60 per cent of
method for determining BMR is to measure the rate of
normal. As discussed in Chapter 76, thyroxine increases
oxygen utilization over a given period of time under the
the rates of the chemical reactions of many cells in the
following conditions:
body and therefore increases metabolic rate. Adapta-
1. The person must not have eaten food for at least 12
tion of the thyroid gland—with increased secretion in
hours.
cold climates and decreased secretion in hot climates—
2. The BMR is determined after a night of restful
contributes to the differences in BMRs among people
sleep.
living in different geographical zones; for example,
3. No strenuous activity is performed for at least 1
people living in arctic regions have BMRs 10 to 20 per
hour before the test.
cent higher than those of persons living in tropical
4. All psychic and physical factors that cause
regions.
excitement must be eliminated.
5. The temperature of the air must be comfortable
Male Sex Hormone Increases Metabolic Rate. The male sex
and between 68° and 80°F.
hormone testosterone can increase the metabolic rate
6. No physical activity is permitted during the test.
about 10 to 15 per cent. The female sex hormones may
The BMR normally averages about 65 to 70 Calories
increase the BMR a small amount, but usually not
per hour in an average 70-kilogram man. Although
enough to be significant. Much of this effect of the male
much of the BMR is accounted for by essential activi-
sex hormone is related to its anabolic effect to increase
ties of the central nervous system, heart, kidneys, and
skeletal muscle mass.
other organs, the variations in BMR among different
individuals are related mainly to differences in the
amount of skeletal muscle and body size.
Growth Hormone Increases Metabolic Rate. Growth hormone
Skeletal muscle, even under resting conditions,
can increase the metabolic rate 15 to 20 per cent as a
accounts for 20 to 30 per cent of the BMR. For this
result of direct stimulation of cellular metabolism.
54
100
52
50
Purposeful physical activity (25%)
48
75
46
Nonexercise activity (7%)
44
Thermic effect of food (8%)
42
Arousal
40
50
38
Males
36
Basal
Sleeping
metabolic
34
Females
25
metabolic
rate (60%)
32
rate
30
0
10
20
30
40
50
60
70
80
0
Age (years)
Figure 72-3
Figure 72-4
Components of energy expenditure.
Normal basal metabolic rates at different ages for each sex.
Chapter 72
Energetics and Metabolic Rate
887
Fever Increases Metabolic Rate. Fever, regardless of its
Even in sedentary individuals who perform little or
cause, increases the chemical reactions of the body by
no daily exercise or physical work, significant energy
an average of about 120 per cent for every 10°C rise
is spent on spontaneous physical activity required
in temperature. This is discussed in more detail in
to maintain muscle tone and body posture and on other
Chapter 73.
nonexercise activities such as
“fidgeting.” Together,
these nonexercise activities account for about 7 per cent
Sleep Decreases Metabolic Rate. The metabolic rate
of a person’s daily energy usage.
decreases 10 to 15 per cent below normal during sleep.
This fall is due to two principal factors: (1) decreased
tone of the skeletal musculature during sleep and (2)
Energy Used for Processing Food—
decreased activity of the central nervous system.
Thermogenic Effect of Food
Malnutrition Decreases Metabolic Rate. Prolonged malnutri-
After a meal is ingested, the metabolic rate increases as
tion can decrease the metabolic rate 20 to 30 per cent,
a result of the different chemical reactions associated
presumably due to the paucity of food substances in the
with digestion, absorption, and storage of food in the
cells. In the final stages of many disease conditions, the
body. This is called the thermogenic effect of food,
inanition that accompanies the disease causes a marked
because these processes require energy and generate
decrease in metabolic rate, to the extent that the body
heat.
temperature may fall several degrees shortly before
After a meal that contains a large quantity of carbo-
death.
hydrates or fats, the metabolic rate usually increases
about 4 per cent. However, after a high-protein meal,
the metabolic rate usually begins rising within an hour,
Energy Used for Physical Activities
reaching a maximum of about 30 per cent above normal,
and this lasts for 3 to 12 hours. This effect of protein on
The factor that most dramatically increases metabolic
the metabolic rate is called the specific dynamic action
rate is strenuous exercise. Short bursts of maximal
of protein. The thermogenic effect of food accounts for
muscle contraction in a single muscle can liberate as
about 8 per cent of the total daily energy expenditure
much as 100 times its normal resting amount of heat for
in many persons.
a few seconds. For the entire body, maximal muscle
exercise can increase the overall heat production of the
body for a few seconds to about 50 times normal, or to
Energy Used for Nonshivering
about 20 times normal for more sustained exercise in a
Thermogenesis—Role of
well-trained individual.
Sympathetic Stimulation
Table 72-1 shows the energy expenditure during dif-
ferent types of physical activity for a 70-kilogram man.
Although physical work and the thermogenic effect of
Because of the great variation in the amount of physi-
food cause liberation of heat, these mechanisms are not
cal activity among individuals, this component of energy
aimed primarily at regulation of body temperature.
expenditure is the most important reason for the dif-
Shivering provides a regulated means of producing heat
ferences in caloric intake required to maintain energy
by increasing muscle activity in response to cold stress,
balance. However, in industrialized countries where
as discussed in Chapter
73. Another mechanism,
food supplies are plentiful, such as the United States,
nonshivering thermogenesis, can also produce heat in
caloric intake periodically exceeds energy expenditure,
response to cold stress. This type of thermogenesis is
and the excess energy is stored mainly as fat. This under-
stimulated by sympathetic nervous system activation,
scores the importance of maintaining a proper level of
which releases norepinephrine and epinephrine, which
physical activity to prevent excess fat stores and obesity.
in turn increase metabolic activity and heat generation.
In certain types of fat tissue, called brown fat, sym-
pathetic nervous stimulation causes liberation of large
amounts of heat. This type of fat contains large numbers
of mitochondria and many small globules of fat instead
Table 72-1
of one large fat globule. In these cells, the process of
Energy Expenditure During Different Types of Activity for a
oxidative phosphorylation in the mitochondria is mainly
70-Kilogram Man
“uncoupled.” That is, when the cells are stimulated by
the sympathetic nerves, the mitochondria produce a
large amount of heat but almost no ATP, so that almost
Form of Activity
Calories per Hour
all the released oxidative energy immediately becomes
Sleeping
65
heat.
Awake lying still
77
A neonate has a considerable number of brown fat
Sitting at rest
100
cells, and maximal sympathetic stimulation can increase
Standing relaxed
105
the child’s metabolism more than 100 per cent. The mag-
Dressing and undressing
118
nitude of this type of thermogenesis in an adult human,
Typewriting rapidly
140
who has virtually no brown fat, is probably less than 15
Walking slowly (2.6 miles per hour)
200
per cent, although this might increase significantly after
Carpentry, metalworking, industrial painting
240
Sawing wood
480
cold adaptation.
Swimming
500
Nonshivering thermogenesis may also serve as a
Running (5.3 miles per hour)
570
buffer against obesity. Recent studies indicate that
Walking up stairs rapidly
1100
sympathetic nervous system activity is increased in
obese persons who have a persistent excess caloric
Extracted from data compiled by Professor M. S. Rose.
intake. The mechanism responsible for sympathetic acti-
888
Unit XIII
Metabolism and Temperature Regulation
vation in obese persons is uncertain, but it may be medi-
MD: National Heart, Lung, and Blood Institute and
ated partly through the effects of increased leptin, which
National Institute of Diabetes and Digestive and Kidney
activates pro-opiomelanocortin neurons in the hypo-
Diseases.
thalamus. Sympathetic stimulation, by increasing ther-
guidelines/index.htm
mogenesis, helps to limit excess weight gain.
Robidoux J, Martin TL, Collins S: Beta-adrenergic receptors
and regulation of energy expenditure: a family affair.
Annu Rev Pharmacol Toxicol 44:297, 2004.
References
Rousset S, Alves-Guerra MC, Mozo J, et al: The biology of
mitochondrial uncoupling proteins. Diabetes
53(Suppl
Argyropoulos G, Harper ME: Uncoupling proteins and ther-
1):S130, 2004.
moregulation. J Appl Physiol 92:2187, 2002.
Seals DR, Bell C: Chronic sympathetic activation: conse-
Cannon B, Nedergaard J: Brown adipose tissue: function and
quence and cause of age-associated obesity? Diabetes
physiological significance. Physiol Rev 84:277, 2004.
53:276, 2004.
Chakravarthy MV, Booth FW: Eating, exercise, and “thrifty”
Silva JE: The thermogenic effect of thyroid hormone and its
genotypes: connecting the dots toward an evolutionary
clinical implications. Ann Intern Med 139:205, 2003.
understanding of modern chronic diseases. J Appl Physiol
van Marken Lichtenbelt WD, Daanen HA: Cold-induced
96:3, 2004.
metabolism. Curr Opin Clin Nutr Metab Care 6:469, 2003.
Evans RM, Barish GD, Wang YX: PPARs and the complex
Westerterp KR: Limits to sustainable human metabolic rate.
journey to obesity. Nat Med 10:355, 2004.
J Exp Biol 204:3183, 2001.
Levine JA: Nonexercise activity thermogenesis (NEAT):
Westerterp KR: Impacts of vigorous and non-vigorous activ-
environment and biology. Am J Physiol Endocrinol Metab
ity on daily energy expenditure. Proc Nutr Soc 62:645,
286:E675, 2004.
2003.
Livingstone MB, Black AE: Markers of the validity of
Wilson MM, Morley JE: Aging and energy balance. J Appl
reported energy intake. J Nutr 133(Suppl 3):895S, 2003.
Physiol 95:1728, 2003.
Lowell BB, Bachman ES: Beta-adrenergic receptors, diet-
Winder WW: Energy-sensing and signaling by AMP-acti-
induced thermogenesis, and obesity. J Biol Chem
vated protein kinase in skeletal muscle. J Appl Physiol
278:29385, 2003.
91:1017, 2001.
Morrison SF: Central pathways controlling brown adipose
Yen PM: Physiological and molecular basis of thyroid
tissue thermogenesis. News Physiol Sci 19:67, 2004.
hormone action. Physiol Rev 81:1097, 2001.
National Institutes of Health: Clinical Guidelines on the
Identification, Evaluation, and Treatment of Overweight
and Obesity in Adults: The Evidence Report. Bethesda,
C H A P T E R
7
3
Body Temperature, Temperature
Regulation, and Fever
Normal Body Temperatures
Core Temperature and Skin Temperature. The tempera-
ture of the deep tissues of the body—the “core” of
the body—remains very constant, within
±1°F
(±0.6°C), day in and day out, except when a person
develops a febrile illness. Indeed, a nude person can
be exposed to temperatures as low as 55°F or as
high as 130°F in dry air and still maintain an almost constant core temperature.
The mechanisms for regulating body temperature represent a beautifully
designed control system. The purpose of this chapter is to discuss this system as
it operates in health and in disease.
The skin temperature, in contrast to the core temperature, rises and falls
with the temperature of the surroundings. The skin temperature is the im-
portant temperature when we refer to the skin’s ability to lose heat to the
surroundings.
Normal Core Temperature. No single core temperature can be considered normal,
because measurements in many healthy people have shown a range of normal
temperatures measured orally, as shown in Figure 73-1, from less than 97°F
(36°C) to over 99.5°F (37.5°C). The average normal core temperature is gener-
ally considered to be between 98.0° and 98.6°F when measured orally and about
1°F higher when measured rectally.
The body temperature increases during exercise and varies with temperature
extremes of the surroundings, because the temperature regulatory mechanisms
are not perfect. When excessive heat is produced in the body by strenuous
exercise, the temperature can rise temporarily to as high as 101° to 104°F. Con-
versely, when the body is exposed to extreme cold, the temperature can often
fall to values below 96°F.
Body Temperature Is Controlled by Balancing
Heat Production Against Heat Loss
When the rate of heat production in the body is greater than the rate at which
heat is being lost, heat builds up in the body and the body temperature rises.
Conversely, when heat loss is greater, both body heat and body temperature
decrease. Most of the remainder of this chapter is concerned with this balance
between heat production and heat loss and the mechanisms by which the body
controls each of these.
Heat Production
Heat production is a principal by-product of metabolism. In Chapter 72, which
summarizes body energetics, we discuss the different factors that determine the
rate of heat production, called the metabolic rate of the body. The most impor-
tant of these factors are listed again here: (1) basal rate of metabolism of all
the cells of the body; (2) extra rate of metabolism caused by muscle activity,
889
890
Unit XIII
Metabolism and Temperature Regulation
including muscle contractions caused by shivering; (3)
tissues to the skin, where it is lost to the air and other
extra metabolism caused by the effect of thyroxine
surroundings. Therefore, the rate at which heat is lost
(and, to a less extent, other hormones, such as growth
is determined almost entirely by two factors: (1) how
hormone and testosterone) on the cells; (4) extra
rapidly heat can be conducted from where it is pro-
metabolism caused by the effect of epinephrine, nor-
duced in the body core to the skin and (2) how rapidly
epinephrine, and sympathetic stimulation on the cells;
heat can then be transferred from the skin to the sur-
(5) extra metabolism caused by increased chemical
roundings. Let us begin by discussing the system that
activity in the cells themselves, especially when the cell
insulates the core from the skin surface.
temperature increases; and
(6) extra metabolism
needed for digestion, absorption, and storage of food
Insulator System of the Body
(thermogenic effect of food).
The skin, the subcutaneous tissues, and especially the
fat of the subcutaneous tissues act together as a heat
insulator for the body. The fat is important because it
Heat Loss
conducts heat only one third as readily as other tissues.
When no blood is flowing from the heated internal
Most of the heat produced in the body is generated in
organs to the skin, the insulating properties of the
the deep organs, especially in the liver, brain, and
normal male body are about equal to three quarters
heart, and in the skeletal muscles during exercise. Then
the insulating properties of a usual suit of clothes. In
this heat is transferred from the deeper organs and
women, this insulation is even better.
The insulation beneath the skin is an effective
means of maintaining normal internal core tempera-
ture, even though it allows the temperature of the skin
Oral
-F
-C
Rectal
to approach the temperature of the surroundings.
104
40
Blood Flow to the Skin from the Body Core
Provides Heat Transfer
Hard exercise
39
Blood vessels are distributed profusely beneath the
102
skin. Especially important is a continuous venous
Emotion or
Hard work, emotion
moderate exercise
plexus that is supplied by inflow of blood from the skin
A few normal adults
38
100
A few normal adults
capillaries, shown in Figure 73-2. In the most exposed
Many active children
Many active children
areas of the body—the hands, feet, and ears—blood is
Usual range
37
Usual range
also supplied to the plexus directly from the small
of normal
98
of normal
arteries through highly muscular arteriovenous
anastomoses.
36
Early morning
Early morning
The rate of blood flow into the skin venous plexus
96
Cold weather, etc.
Cold weather, etc.
can vary tremendously—from barely above zero to as
great as 30 per cent of the total cardiac output. A high
rate of skin flow causes heat to be conducted from the
core of the body to the skin with great efficiency,
Figure 73-1
whereas reduction in the rate of skin flow can decrease
the heat conduction from the core to very little.
Estimated range of body “core” temperature in normal people.
(Redrawn from DuBois EF: Fever. Springfield, IL: Charles C
Figure 73-3 shows quantitatively the effect of envi-
Thomas, 1948.)
ronmental air temperature on conductance of heat
Epidermis
Capillaries
Dermis
Arteries
Veins
Venous plexus
Subcutaneous tissue
Arteriovenous
anastomosis
Figure 73-2
Artery
Skin circulation.
Chapter 73
Body Temperature, Temperature Regulation, and Fever
891
Walls
Evaporation (22%)
Vasodilated
8
Radiation (60%)
7
heat waves
6
5
Conduction to air (15%)
4
Air currents
3
(convection)
Conduction to
objects (3%)
2
Vasoconstricted
1
Figure 73-4
0
Mechanisms of heat loss from the body.
50
60
70
80
90
100
110
120
Environmental temperature (°F)
have wavelengths of 5 to 20 micrometers, 10 to 30
times the wavelengths of light rays. All objects that are
not at absolute zero temperature radiate such rays. The
Figure 73-3
human body radiates heat rays in all directions. Heat
rays are also being radiated from the walls of rooms
Effect of changes in the environmental temperature on heat con-
ductance from the body core to the skin surface. (Modified from
and other objects toward the body. If the temperature
Benzinger TH: Heat and Temperature Fundamentals of Medical
of the body is greater than the temperature of the sur-
Physiology. New York: Dowden, Hutchinson & Ross, 1980.)
roundings, a greater quantity of heat is radiated from
the body than is radiated to the body.
from the core to the skin surface and then conductance
Conduction. As shown in Figure
73-4, only minute
into the air, demonstrating an approximate eightfold
quantities of heat, about 3 per cent, are normally lost
increase in heat conductance between the fully vaso-
from the body by direct conduction from the surface
constricted state and the fully vasodilated state.
of the body to solid objects, such as a chair or a bed.
Therefore, the skin is an effective controlled “heat
Loss of heat by conduction to air, however, represents
radiator” system, and the flow of blood to the skin is a
a sizable proportion of the body’s heat loss (about 15
most effective mechanism for heat transfer from the
per cent) even under normal conditions.
body core to the skin.
It will be recalled that heat is actually the kinetic
energy of molecular motion, and the molecules of
Control of Heat Conduction to the Skin by the Sympathetic
the skin are continually undergoing vibratory motion.
Nervous System. Heat conduction to the skin by the
Much of the energy of this motion can be transferred
blood is controlled by the degree of vasoconstriction
to the air if the air is colder than the skin, thus increas-
of the arterioles and the arteriovenous anastomoses
ing the velocity of the air molecules’ motion. Once the
that supply blood to the venous plexus of the skin. This
temperature of the air adjacent to the skin equals the
vasoconstriction is controlled almost entirely by the
temperature of the skin, no further loss of heat occurs
sympathetic nervous system in response to changes in
in this way, because now an equal amount of heat is
body core temperature and changes in environmental
conducted from the air to the body. Therefore, con-
temperature. This is discussed later in the chapter in
duction of heat from the body to the air is self-limited
connection with control of body temperature by the
unless the heated air moves away from the skin, so that
hypothalamus.
new, unheated air is continually brought in contact
with the skin, a phenomenon called air convection.
Basic Physics of How Heat Is Lost from the
Skin Surface
Convection. The removal of heat from the body by con-
The various methods by which heat is lost from the
vection air currents is commonly called heat loss by
skin to the surroundings are shown in Figure 73-4.
convection. Actually, the heat must first be conducted
They include radiation, conduction, and evaporation,
to the air and then carried away by the convection air
which are explained next.
currents.
A small amount of convection almost always occurs
Radiation. As shown in Figure 73-4, in a nude person
around the body because of the tendency for air adja-
sitting inside at normal room temperature, about 60
cent to the skin to rise as it becomes heated. There-
per cent of total heat loss is by radiation.
fore, in a nude person seated in a comfortable room
Loss of heat by radiation means loss in the form of
without gross air movement, about 15 per cent of his
infrared heat rays, a type of electromagnetic wave.
or her total heat loss occurs by conduction to the air
Most infrared heat rays that radiate from the body
and then by air convection away from the body.
892
Unit XIII
Metabolism and Temperature Regulation
Cooling Effect of Wind. When the body is exposed to
suit of clothes decreases the rate of heat loss to about
wind, the layer of air immediately adjacent to the skin
half that from the nude body, but arctic-type clothing
is replaced by new air much more rapidly than normally,
can decrease this heat loss to as little as one sixth.
and heat loss by convection increases accordingly. The
About half the heat transmitted from the skin to the
cooling effect of wind at low velocities is about propor-
clothing is radiated to the clothing instead of being
tional to the square root of the wind velocity. For
conducted across the small intervening space. There-
instance, a wind of 4 miles per hour is about twice as
fore, coating the inside of clothing with a thin layer of
effective for cooling as a wind of 1 mile per hour.
gold, which reflects radiant heat back to the body,
Conduction and Convection of Heat from a Person Sus-
makes the insulating properties of clothing far more
pended in Water. Water has a specific heat several thou-
effective than otherwise. Using this technique, clothing
sand times as great as that of air, so that each unit
for use in the arctic can be decreased in weight by
portion of water adjacent to the skin can absorb far
about half.
greater quantities of heat than air can. Also, heat con-
The effectiveness of clothing in maintaining body
ductivity in water is very great in comparison with that
temperature is almost completely lost when the cloth-
in air. Consequently, it is impossible for the body to heat
ing becomes wet, because the high conductivity of
a thin layer of water next to the body to form an “insu-
water increases the rate of heat transmission through
lator zone” as occurs in air. Therefore, the rate of heat
loss to water is usually many times greater than the rate
cloth
20-fold or more. Therefore, one of the most
of heat loss to air.
important factors for protecting the body against cold
in arctic regions is extreme caution against allowing
Evaporation. When water evaporates from the body
the clothing to become wet. Indeed, one must be
surface, 0.58 Calorie (kilocalorie) of heat is lost for
careful not to become overheated even temporarily,
each gram of water that evaporates. Even when a
because sweating in one’s clothes makes them much
person is not sweating, water still evaporates insensi-
less effective thereafter as an insulator.
bly from the skin and lungs at a rate of about 600 to
700 ml/day. This causes continual heat loss at a rate of
Sweating and Its Regulation by the Autonomic
16 to 19 Calories per hour. This insensible evaporation
Nervous System
through the skin and lungs cannot be controlled for
Stimulation of the anterior hypothalamus-preoptic
purposes of temperature regulation because it results
area in the brain either electrically or by excess heat
from continual diffusion of water molecules through
causes sweating. The nerve impulses from this area
the skin and respiratory surfaces. However, loss of heat
that cause sweating are transmitted in the autonomic
by evaporation of sweat can be controlled by regulat-
pathways to the spinal cord and then through sympa-
ing the rate of sweating, which is discussed later in the
thetic outflow to the skin everywhere in the body.
chapter.
It should be recalled from the discussion of the
autonomic nervous system in Chapter 60 that the
Evaporation Is a Necessary Cooling Mechanism at Very High
sweat glands are innervated by cholinergic nerve
Air Temperatures. As long as skin temperature is greater
fibers (fibers that secrete acetylcholine but that run
than the temperature of the surroundings, heat can be
in the sympathetic nerves along with the adrenergic
lost by radiation and conduction. But when the tem-
fibers). These glands can also be stimulated to
perature of the surroundings becomes greater than that
some extent by epinephrine or norepinephrine circu-
of the skin, instead of losing heat, the body gains heat
by both radiation and conduction. Under these condi-
lating in the blood, even though the glands themselves
tions, the only means by which the body can rid itself of
do not have adrenergic innervation. This is important
heat is by evaporation.
during exercise, when these hormones are secreted by
Therefore, anything that prevents adequate evapo-
the adrenal medullae and the body needs to lose
ration when the surrounding temperature is higher
excessive amounts of heat produced by the active
than the skin temperature will cause the internal body
muscles.
temperature to rise. This occurs occasionally in human
beings who are born with congenital absence of sweat
Mechanism of Sweat Secretion. In Figure 73-5, the sweat
glands. These people can stand cold temperatures as
gland is shown to be a tubular structure consisting of
well as normal people can, but they are likely to die of
two parts: (1) a deep subdermal coiled portion that
heatstroke in tropical zones because without the evap-
secretes the sweat, and (2) a duct portion that passes
orative refrigeration system, they cannot prevent a rise
outward through the dermis and epidermis of the skin.
in body temperature when the air temperature is
As is true of so many other glands, the secretory
above that of the body.
portion of the sweat gland secretes a fluid called the
primary secretion or precursor secretion; the concen-
Effect of Clothing on Conductive Heat Loss. Clothing
trations of constituents in the fluid are then modified
entraps air next to the skin in the weave of the cloth,
as the fluid flows through the duct.
thereby increasing the thickness of the so-called
The precursor secretion is an active secretory
private zone of air adjacent to the skin and also
product of the epithelial cells lining the coiled portion
decreasing the flow of convection air currents. Conse-
of the sweat gland. Cholinergic sympathetic nerve
quently, the rate of heat loss from the body by con-
fibers ending on or near the glandular cells elicit the
duction and convection is greatly depressed. A usual
secretion.
Chapter 73
Body Temperature, Temperature Regulation, and Fever
893
Pore
duct may reabsorb only slightly more than half the
sodium chloride; the concentrations of sodium and
chloride ions are then (in an unacclimatized person) a
maximum of about 50 to 60 mEq/L, slightly less than
half the concentrations in plasma. Furthermore, the
sweat flows through the glandular tubules so rapidly
that little of the water is reabsorbed. Therefore, the
other dissolved constituents of sweat are only moder-
ately increased in concentration—urea is about twice
that in the plasma, lactic acid about 4 times, and potas-
Duct
sium about 1.2 times.
Absorption, mainly
There is a significant loss of sodium chloride in the
sodium and chloride ions
sweat when a person is unacclimatized to heat. There
is much less electrolyte loss, despite increased sweat-
ing capacity, once a person has become acclimatized,
as follows.
Acclimatization of the Sweating Mechanism to Heat—Role
of Aldosterone. Although a normal, unacclimatized
Gland
person seldom produces more than about 1 liter of
sweat per hour, when this person is exposed to hot
Primary
weather for 1 to 6 weeks, he or she begins to sweat
secretion,
mainly
more profusely, often increasing maximum sweat pro-
protein-
duction to as much as 2 to 3 L/hour. Evaporation of
free
this much sweat can remove heat from the body at a
filtrate
rate more than 10 times the normal basal rate of heat
production. This increased effectiveness of the sweat-
ing mechanism is caused by a change in the internal
Sympathetic
sweat gland cells themselves to increase their sweating
nerve
capability.
Also associated with acclimatization is a further
Figure 73-5
decrease in the concentration of sodium chloride in
Sweat gland innervated by an acetylcholine-secreting sympa-
the sweat, which allows progressively better conserva-
thetic nerve. A primary protein-free secretion is formed by the
tion of body salt. Most of this effect is caused by
glandular portion, but most of the electrolytes are reabsorbed in
increased secretion of aldosterone by the adrenocor-
the duct, leaving a dilute, watery secretion.
tical glands, which results from a slight decrease in
sodium chloride concentration in the extracellular
fluid and plasma. An unacclimatized person who
The composition of the precursor secretion is
sweats profusely often loses 15 to 30 grams of salt each
similar to that of plasma, except that it does not
day for the first few days. After 4 to 6 weeks of acclima-
contain plasma proteins. The concentration of sodium
tization, the loss is usually 3 to 5 g/day.
is about
142 mEq/L and that of chloride is about
104 mEq/L, with much smaller concentrations of the
Loss of Heat by Panting
Many lower animals have little ability to lose heat from
other solutes of plasma. As this precursor solution
the surfaces of their bodies, for two reasons: (1) the sur-
flows through the duct portion of the gland, it is mod-
faces are usually covered with fur, and (2) the skin of
ified by reabsorption of most of the sodium and chlo-
most lower animals is not supplied with sweat glands,
ride ions. The degree of this reabsorption depends on
which prevents most of the evaporative loss of heat
the rate of sweating, as follows.
from the skin. A substitute mechanism, the panting
When the sweat glands are stimulated only slightly,
mechanism, is used by many lower animals as a means
the precursor fluid passes through the duct slowly. In
of dissipating heat.
this instance, essentially all the sodium and chloride
The phenomenon of panting is “turned on” by the
ions are reabsorbed, and the concentration of each
thermoregulator centers of the brain. That is, when the
falls to as low as 5 mEq/L. This reduces the osmotic
blood becomes overheated, the hypothalamus initiates
neurogenic signals to decrease the body temperature.
pressure of the sweat fluid to such a low level that most
One of these signals initiates panting. The actual panting
of the water is also reabsorbed, which concentrates
process is controlled by a panting center that is associ-
most of the other constituents. Therefore, at low rates
ated with the pneumotaxic respiratory center located in
of sweating, such constituents as urea, lactic acid, and
the pons.
potassium ions are usually very concentrated.
When an animal pants, it breathes in and out rapidly,
Conversely, when the sweat glands are strongly
so that large quantities of new air from the exterior
stimulated by the sympathetic nervous system, large
come in contact with the upper portions of the respira-
amounts of precursor secretion are formed, and the
tory passages; this cools the blood in the respiratory
894
Unit XIII
Metabolism and Temperature Regulation
passage mucosa as a result of water evaporation from
Role of the Anterior Hypothalamic-Preoptic
the mucosal surfaces, especially evaporation of saliva
Area in Thermostatic Detection
from the tongue. Yet panting does not increase the alve-
of Temperature
olar ventilation more than is required for proper control
Experiments have been performed in which minute
of the blood gases, because each breath is extremely
areas in the brain of an animal have been either heated
shallow; therefore, most of the air that enters the alveoli
or cooled by use of a thermode. This small, needle-like
is dead-space air mainly from the trachea and not from
device is heated by electrical means or by passing hot
the atmosphere.
water through it, or it is cooled by cold water. The
principal areas in the brain where heat or cold from
a thermode affects body temperature control are the
Regulation of Body
preoptic and anterior hypothalamic nuclei of the
Temperature—Role of the
hypothalamus.
Hypothalamus
Using the thermode, the anterior hypothalamic-
preoptic area has been found to contain large numbers
Figure 73-6 shows what happens to the body “core”
of heat-sensitive neurons as well as about one third
temperature of a nude person after a few hours’ expo-
as many cold-sensitive neurons. These neurons are
sure to dry air ranging from 30° to 160°F. The precise
believed to function as temperature sensors for con-
dimensions of this curve depend on the wind move-
trolling body temperature. The heat-sensitive neurons
ment of the air, the amount of moisture in the air, and
increase their firing rate 2- to 10-fold in response to a
even the nature of the surroundings. In general, a nude
10°C increase in body temperature. The cold-sensitive
person in dry air between 55° and 130°F is capable of
neurons, by contrast, increase their firing rate when the
maintaining a normal body core temperature some-
body temperature falls.
where between 97° and 100°F.
When the preoptic area is heated, the skin all over
The temperature of the body is regulated almost
the body immediately breaks out in a profuse sweat,
entirely by nervous feedback mechanisms, and almost
while the skin blood vessels over the entire body
all these operate through temperature-regulating
become greatly dilated. This is an immediate reaction
centers located in the hypothalamus. For these feed-
to cause the body to lose heat, thereby helping to
back mechanisms to operate, there must also be
return the body temperature toward the normal level.
temperature detectors to determine when the body
In addition, any excess body heat production is inhib-
temperature becomes either too high or too low.
ited. Therefore, it is clear that the hypothalamic-
preoptic area has the capability to serve as a ther-
mostatic body temperature control center.
Detection of Temperature by Receptors in the
Skin and Deep Body Tissues
Although the signals generated by the temperature
receptors of the hypothalamus are extremely power-
ful in controlling body temperature, receptors in other
120
parts of the body play additional roles in temperature
regulation. This is especially true of temperature
110
receptors in the skin and in a few specific deep tissues
of the body.
100
It will be recalled from the discussion of sensory
receptors in Chapter 48 that the skin is endowed
with both cold and warmth receptors. There are far
90
more cold receptors than warmth receptors—in fact,
10 times as many in many parts of the skin. Therefore,
80
peripheral detection of temperature mainly concerns
detecting cool and cold instead of warm temperatures.
70
When the skin is chilled over the entire body, imme-
diate reflex effects are invoked and begin to increase
the temperature of the body in several ways: (1) by
60
providing a strong stimulus to cause shivering, with a
30
50
70
90
110
130
150
170
resultant increase in the rate of body heat production;
Atmospheric temperature (°F)
(2) by inhibiting the process of sweating, if this is
already occurring; and (3) by promoting skin vaso-
constriction to diminish loss of body heat from the
Figure 73-6
skin.
Deep body temperature receptors are found mainly
Effect of high and low atmospheric temperatures of several hours’
in the spinal cord, in the abdominal viscera, and in or
duration on the internal body “core” temperature. Note that the
around the great veins in the upper abdomen and
internal body temperature remains stable despite wide changes
in atmospheric temperature.
thorax. These deep receptors function differently from
Chapter 73
Body Temperature, Temperature Regulation, and Fever
895
the skin receptors because they are exposed to the
body core temperature rather than the body surface
90
temperature. Yet, like the skin temperature receptors,
they detect mainly cold rather than warmth. It is prob-
Heat production
80
able that both the skin and the deep body receptors
Evaporative heat loss
are concerned with preventing hypothermia—that is,
70
preventing low body temperature.
60
Posterior Hypothalamus Integrates the Central
and Peripheral Temperature Sensory Signals
50
Even though many temperature sensory signals arise
40
in peripheral receptors, these signals contribute to
body temperature control mainly through the hypo-
30
thalamus. The area of the hypothalamus that they
stimulate is located bilaterally in the posterior hypo-
20
thalamus approximately at the level of the mammil-
lary bodies. The temperature sensory signals from the
10
anterior hypothalamic-preoptic area are also trans-
mitted into this posterior hypothalamic area. Here the
0
signals from the preoptic area and the signals from
36.4
36.6
36.8
37.0
37.2
37.4
37.6
elsewhere in the body are combined and integrated to
Head temperature (°C)
control the heat-producing and heat-conserving reac-
tions of the body.
Figure 73-7
Neuronal Effector Mechanisms
Effect of hypothalamic temperature on evaporative heat loss from
the body and on heat production caused primarily by muscle
That Decrease or Increase Body
activity and shivering. This figure demonstrates the extremely crit-
Temperature
ical temperature level at which increased heat loss begins and
heat production reaches a minimum stable level.
When the hypothalamic temperature centers detect
that the body temperature is either too high or too
low, they institute appropriate temperature-decreasing
or temperature-increasing procedures. The reader is
probably familiar with most of these from personal
Temperature-Increasing Mechanisms When the
experience, but special features are the following.
Body Is Too Cold
When the body is too cold, the temperature control
Temperature-Decreasing Mechanisms
system institutes exactly opposite procedures. They
When the Body Is Too Hot
are:
The temperature control system uses three important
1. Skin vasoconstriction throughout the body.
mechanisms to reduce body heat when the body tem-
This is caused by stimulation of the posterior
perature becomes too great:
hypothalamic sympathetic centers.
1. Vasodilation of skin blood vessels. In almost all
2. Piloerection. Piloerection means hairs “standing
areas of the body, the skin blood vessels become
on end.” Sympathetic stimulation causes the
intensely dilated. This is caused by inhibition
arrector pili muscles attached to the hair follicles
of the sympathetic centers in the posterior
to contract, which brings the hairs to an upright
hypothalamus that cause vasoconstriction. Full
stance. This is not important in human beings, but
vasodilation can increase the rate of heat transfer
in lower animals, upright projection of the hairs
to the skin as much as eightfold.
allows them to entrap a thick layer of “insulator
2. Sweating. The effect of increased body
air” next to the skin, so that transfer of heat to
temperature to cause sweating is demonstrated by
the surroundings is greatly depressed.
the blue curve in Figure 73-7, which shows a
3. Increase in thermogenesis (heat production). Heat
sharp increase in the rate of evaporative heat
production by the metabolic systems is increased
loss resulting from sweating when the body core
by promoting shivering, sympathetic excitation of
temperature rises above the critical level of 37°C
heat production, and thyroxine secretion. These
(98.6°F). An additional 1°C increase in body
methods of increasing heat require additional
temperature causes enough sweating to remove 10
explanation, which follows.
times the basal rate of body heat production.
3. Decrease in heat production. The mechanisms that
Hypothalamic Stimulation of Shivering. Located in the
cause excess heat production, such as shivering
dorsomedial portion of the posterior hypothalamus
and chemical thermogenesis, are strongly
near the wall of the third ventricle is an area called
inhibited.
the primary motor center for shivering. This area is
896
Unit XIII
Metabolism and Temperature Regulation
normally inhibited by signals from the heat center in
of the hypothalamic portal veins to the anterior pitu-
the anterior hypothalamic-preoptic area but is excited
itary gland, where it stimulates secretion of thyroid-
by cold signals from the skin and spinal cord. There-
stimulating hormone.
fore, as shown by the sudden increase in “heat pro-
Thyroid-stimulating hormone in turn stimulates
duction” (see the red curve in Figure 73-7), this center
increased output of thyroxine by the thyroid gland, as
becomes activated when the body temperature falls
explained in Chapter
76. The increased thyroxine
even a fraction of a degree below a critical tempera-
increases the rate of cellular metabolism throughout
ture level. It then transmits signals that cause shiver-
the body, which is yet another mechanism of chemical
ing through bilateral tracts down the brain stem, into
thermogenesis. This increase in metabolism does not
the lateral columns of the spinal cord, and finally to
occur immediately but requires several weeks’ expo-
the anterior motor neurons. These signals are non-
sure to cold to make the thyroid gland hypertrophy
rhythmical and do not cause the actual muscle shaking.
and reach its new level of thyroxine secretion.
Instead, they increase the tone of the skeletal muscles
Exposure of animals to extreme cold for several
throughout the body by facilitating the activity of the
weeks can cause their thyroid glands to increase in size
anterior motor neurons. When the tone rises above a
20 to 40 per cent. However, human beings seldom
certain critical level, shivering begins. This probably
allow themselves to be exposed to the same degree of
results from feedback oscillation of the muscle spindle
cold as that to which animals are often subjected.
stretch reflex mechanism, which is discussed in
Therefore, we still do not know, quantitatively, how
Chapter 54. During maximum shivering, body heat
important the thyroid mechanism of adaptation to
production can rise to four to five times normal.
cold is in the human being.
Isolated measurements have shown that military
Sympathetic “Chemical” Excitation of Heat Production. As
personnel residing for several months in the arctic
pointed out in Chapter 72, an increase in either sym-
develop increased metabolic rates; some Inuit
pathetic stimulation or circulating norepinephrine and
(Eskimos) also have abnormally high basal metabolic
epinephrine in the blood can cause an immediate
rates. Further, the continuous stimulatory effect of
increase in the rate of cellular metabolism. This effect
cold on the thyroid gland may explain the much higher
is called chemical thermogenesis. It results at least par-
incidence of toxic thyroid goiters in people who live in
tially from the ability of norepinephrine and epineph-
cold climates than in those who live in warm climates.
rine to uncouple oxidative phosphorylation, which
means that excess foodstuffs are oxidized and thereby
release energy in the form of heat but do not cause
Concept of a “Set-Point”
adenosine triphosphate to be formed.
for Temperature Control
The degree of chemical thermogenesis that occurs
in an animal is almost directly proportional to the
In the example of Figure 73-7, it is clear that at a crit-
amount of brown fat in the animal’s tissues. This is a
ical body core temperature of about 37.1°C (98.8∞F),
type of fat that contains large numbers of special
drastic changes occur in the rates of both heat loss and
mitochondria where uncoupled oxidation occurs, as
heat production. At temperatures above this level, the
described in Chapter 72; these cells are supplied by
rate of heat loss is greater than that of heat produc-
strong sympathetic innervation.
tion, so the body temperature falls and approaches the
Acclimatization greatly affects the intensity of
37.1°C level. At temperatures below this level, the rate
chemical thermogenesis; some animals, such as rats,
of heat production is greater than that of heat loss, so
that have been exposed to a cold environment for
the body temperature rises and again approaches the
several weeks exhibit a 100 to 500 per cent increase in
37.1°C level. This crucial temperature level is called
heat production when acutely exposed to cold, in con-
the “set-point” of the temperature control mechanism.
trast to the unacclimatized animal, which responds
That is, all the temperature control mechanisms con-
with an increase of perhaps one third as much. This
tinually attempt to bring the body temperature back
increased thermogenesis also leads to a corresponding
to this set-point level.
increase in food intake.
In adult human beings, who have almost no brown
Feedback Gain for Body Temperature Control. Let us recall
fat, it is rare for chemical thermogenesis to increase
the discussion of feedback gain of control systems pre-
the rate of heat production more than 10 to 15 per
sented in Chapter 1. Feedback gain is a measure of the
cent. However, in infants, who do have a small amount
effectiveness of a control system. In the case of body
of brown fat in the interscapular space, chemical ther-
temperature control, it is important for the internal
mogenesis can increase the rate of heat production 100
core temperature to change as little as possible, even
per cent, which is probably an important factor in
though the environmental temperature might change
maintaining normal body temperature in neonates.
greatly from day to day or even hour to hour. The feed-
back gain of the temperature control system is equal
Increased Thyroxine Output as a Long-Term Cause of Increased
to the ratio of the change in environmental tempera-
Heat Production. Cooling the anterior hypothalamic-
ture to the change in body core temperature minus
preoptic area also increases production of the neu-
1.0
(see Chapter 1 for this formula). Experiments
rosecretory hormone thyrotropin-releasing hormone
have shown that the body temperature of humans
by the hypothalamus. This hormone is carried by way
changes about 1°C for each 25° to 30°C change in
Chapter 73
Body Temperature, Temperature Regulation, and Fever
897
environmental temperature. Therefore, the feedback
temperature was high, sweating began at a lower
gain of the total mechanism for body temperature
hypothalamic temperature than when the skin tem-
control averages about 27 (28/1.0 - 1.0 = 27), which is
perature was low. One can readily understand the
an extremely high gain for a biological control system
value of such a system, because it is important that
(the baroreceptor arterial pressure control system, for
sweating be inhibited when the skin temperature is
instance, has a feedback gain of less than 2).
low; otherwise, the combined effect of low skin
temperature and sweating could cause far too much
Skin Temperature Can Slightly Alter the
loss of body heat.
Set-Point for Core Temperature Control
A similar effect occurs in shivering, as shown in
The critical temperature set-point in the hypothalamus
Figure 73-9. That is, when the skin becomes cold,
above which sweating begins and below which shiver-
it drives the hypothalamic centers to the shivering
ing begins is determined mainly by the degree of activ-
threshold even when the hypothalamic temperature
ity of the heat temperature receptors in the anterior
itself is still on the hot side of normal. Here again, one
hypothalamic-preoptic area. However, temperature
can understand the value of the control system,
signals from the peripheral areas of the body, espe-
because a cold skin temperature would soon lead to a
cially from the skin and certain deep body tissues
deeply depressed body temperature unless heat pro-
(spinal cord and abdominal viscera), also contribute
duction were increased. Thus, a cold skin temperature
slightly to body temperature regulation. But how do
actually “anticipates” a fall in internal body tempera-
they contribute? The answer is that they alter the set-
ture and prevents this.
point of the hypothalamic temperature control center.
This effect is shown in Figures 73-8 and 73-9.
Figure 73-8 demonstrates the effect of different skin
Behavioral Control of Body
temperatures on the set-point for sweating, showing
Temperature
that the set-point increases as the skin temperature
decreases. Thus, for the person represented in this
Aside from the subconscious mechanisms for
figure, the hypothalamic set-point increased from
body temperature control, the body has another
36.7°C when the skin temperature was higher than
temperature-control mechanism that is even more
33°C to a set-point of 37.4°C when the skin tempera-
potent. This is behavioral control of temperature, which
ture had fallen to 29°C. Therefore, when the skin
Skin temperature (20°)
90
90
80
20°
33°C
80
22°
to
70
39°C
70
24°
Shivering
60
60
26°
Skin temperature
50
28°
Sweating
50
32°C
40
30°
40
31°C
Set-point
30
30
31°
30°C
20
Basal heat
Set-point
20
29°C
production
10
Insensible
10
evaporation
0
0
36.0 36.2 36.4
36.6
36.8
37.0 37.2 37.4
36.6
36.8
37.0
37.2
37.4
37.6
Internal head temperature (°C)
Internal head temperature (°C)
Figure 73-8
Figure 73-9
Effect of changes in the internal head temperature on the rate of
Effect of changes in the internal head temperature on the rate of
evaporative heat loss from the body. Note that the skin tempera-
heat production by the body. Note that the skin temperature
ture determines the set-point level at which sweating begins.
determines the set-point level at which shivering begins.
(Courtesy Dr. T. H. Benzinger.)
(Courtesy Dr. T. H. Benzinger.)
898
Unit XIII
Metabolism and Temperature Regulation
can be explained as follows: Whenever the internal
body temperature becomes too high, signals from the
-F
-C
temperature-controlling areas in the brain give the
Upper limit
person a psychic sensation of being overheated. Con-
Temperature
114
of survival?
regulation
versely, whenever the body becomes too cold, signals
44
seriously
110
Heatstroke
from the skin and probably also from some deep body
impaired
42
Brain lesions
receptors elicit the feeling of cold discomfort. There-
106
Fever therapy
Temperature
40
fore, the person makes appropriate environmental
regulation
102
Febrile disease
38
adjustments to re-establish comfort, such as moving
efficient in
and hard exercise
98
into a heated room or wearing well-insulated clothing
febrile disease,
36
Usual range
health, and work
94
in freezing weather. This is a much more powerful
34
of normal
system of body temperature control than most
Temperature
90
32
regulation
physiologists have acknowledged in the past. Indeed,
impaired
86
30
this is the only really effective mechanism to prevent
body heat control breakdown in severely cold
82
28
Temperature
environments.
26
regulation
78
Lower limit
lost
24
of survival?
74
Local Skin Temperature Reflexes
When a person places a foot under a hot lamp and
leaves it there for a short time, local vasodilation and
Figure 73-10
mild local sweating occur. Conversely, placing the foot
in cold water causes local vasoconstriction and local ces-
Body temperatures under different conditions. (Redrawn from
sation of sweating. These reactions are caused by local
DuBois EF: Fever. Springfield, IL: Charles C Thomas, 1948.)
effects of temperature directly on the blood vessels and
also by local cord reflexes conducted from skin recep-
tors to the spinal cord and back to the same skin area
brain tumors, and environmental conditions that may
and the sweat glands. The intensity of these local effects
terminate in heatstroke.
is, in addition, controlled by the central brain tempera-
ture controller, so that their overall effect is propor-
Resetting the Hypothalamic Temperature-
tional to the hypothalamic heat control signal times the
local signal. Such reflexes can help prevent excessive
Regulating Center in Febrile Diseases—
heat exchange from locally cooled or heated portions of
Effect of Pyrogens
the body.
Many proteins, breakdown products of proteins, and
certain other substances, especially lipopolysaccharide
Regulation of Internal Body Temperature Is Impaired by Cutting
toxins released from bacterial cell membranes, can
the Spinal Cord. After cutting the spinal cord in the neck
cause the set-point of the hypothalamic thermostat to
above the sympathetic outflow from the cord, regulation
rise. Substances that cause this effect are called pyro-
of body temperature becomes extremely poor because
gens. Pyrogens released from toxic bacteria or those
the hypothalamus can no longer control either skin
released from degenerating body tissues cause fever
blood flow or the degree of sweating anywhere in the
during disease conditions. When the set-point of the
body. This is true even though the local temperature
reflexes originating in the skin, spinal cord, and intra-
hypothalamic temperature-regulating center becomes
abdominal receptors still exist. These reflexes are
higher than normal, all the mechanisms for raising the
extremely weak in comparison with hypothalamic
body temperature are brought into play, including heat
control of body temperature.
conservation and increased heat production. Within a
In people with this condition, body temperature
few hours after the set-point has been increased, the
must be regulated principally by the patient’s psychic
body temperature also approaches this level, as shown
response to cold and hot sensations in the head region—
in Figure 73-11.
that is, by behavioral control of clothing and by moving
into an appropriate warm or cold environment.
Mechanism of Action of Pyrogens in Causing Fever—Role of
Interleukin-1. Experiments in animals have shown that
some pyrogens, when injected into the hypothalamus,
Abnormalities of Body
can act directly and immediately on the hypothalamic
Temperature Regulation
temperature-regulating center to increase its set-point.
Other pyrogens function indirectly and may require
Fever
several hours of latency before causing their effects.
This is true of many of the bacterial pyrogens, espe-
Fever, which means a body temperature above the
cially the endotoxins from gram-negative bacteria.
usual range of normal, can be caused by abnormalities
When bacteria or breakdown products of bacteria
in the brain itself or by toxic substances that affect the
are present in the tissues or in the blood, they are
temperature-regulating centers. Some causes of fever
phagocytized by the blood leukocytes, by tissue
(and also of subnormal body temperatures) are pre-
macrophages, and by large granular killer lymphocytes.
sented in Figure 73-10. They include bacterial diseases,
All these cells digest the bacterial products and then
Chapter 73
Body Temperature, Temperature Regulation, and Fever
899
the normal level to higher than normal (as a result of
tissue destruction, pyrogenic substances, or dehydra-
tion), the body temperature usually takes several hours
Set-point
suddenly
Setting of the thermostat
to reach the new temperature set-point.
105
raised to
Actual body temperature
Figure 73-11 demonstrates the effect of suddenly
high value
increasing the temperature set-point to a level of 103°F.
104
Because the blood temperature is now less than the set-
Crisis
103
point of the hypothalamic temperature controller, the
usual responses that cause elevation of body tempera-
Vasodilation
102
ture occur. During this period, the person experiences
Sweating
chills and feels extremely cold, even though his or her
101
Chills:
Set-point
body temperature may already be above normal. Also,
1. Vasoconstriction
suddenly
100
2. Piloerection
reduced to
the skin becomes cold because of vasoconstriction,
3. Epinephrine
low value
and the person shivers. Chills can continue until the
99
secretion
body temperature reaches the hypothalamic set-point
4. Shivering
of 103°F. Then the person no longer experiences chills
98
but instead feels neither cold nor hot. As long as the
0
1
2
3
4
5
factor that is causing the higher set-point of the hypo-
Time in hours
thalamic temperature controller is present, the body
temperature is regulated more or less in the normal
manner, but at the high temperature set-point level.
Figure 73-11
Crisis, or “Flush.” If the factor that is causing the high
temperature is removed, the set-point of the hypothal-
Effects of changing the set-point of the hypothalamic temperature
amic temperature controller will be reduced to a lower
controller.
value—perhaps even back to the normal level, as shown
in Figure 73-11. In this instance, the body temperature
release the substance interleukin-1—also called leuko-
is still
103°F, but the hypothalamus is attempting
cyte pyrogen or endogenous pyrogen—into the body
to regulate the temperature to 98.6°F. This situation
fluids. The interleukin-1, on reaching the hypothala-
is analogous to excessive heating of the anterior
mus, immediately activates the processes to produce
hypothalamic-preoptic area, which causes intense
sweating and the sudden development of hot skin
fever, sometimes increasing the body temperature a
because of vasodilation everywhere. This sudden change
noticeable amount in only 8 to 10 minutes. As little as
of events in a febrile state is known as the “crisis” or,
one ten-millionth of a gram of endotoxin lipopolysac-
more appropriately, the “flush.” In the days before the
charide from bacteria, acting in concert with the blood
advent of antibiotics, the crisis was always anxiously
leukocytes, tissue macrophages, and killer lympho-
awaited, because once this occurred, the doctor
cytes, can cause fever. The amount of interleukin-1 that
assumed that the patient’s temperature would soon
is formed in response to lipopolysaccharide to cause
begin falling.
fever is only a few nanograms.
Several experiments have suggested that inter-
Heatstroke
leukin-1 causes fever by first inducing the formation of
The upper limit of air temperature that one can stand
one of the prostaglandins, mainly prostaglandin E2, or
depends almost entirely on whether the air is dry or wet.
If the air is dry and sufficient convection air currents are
a similar substance, which acts in the hypothalamus to
flowing to promote rapid evaporation from the body, a
elicit the fever reaction. When prostaglandin forma-
person can withstand several hours of air temperature
tion is blocked by drugs, the fever is either completely
at 130°F. Conversely, if the air is 100 per cent humidi-
abrogated or at least reduced. In fact, this may be the
fied or if the body is in water, the body temperature
explanation for the manner in which aspirin reduces
begins to rise whenever the environmental temperature
fever, because aspirin impedes the formation of
rises above about 94°F. If the person is performing
prostaglandins from arachidonic acid. Drugs such as
heavy work, the critical environmental temperature
aspirin that reduce fever are called antipyretics.
above which heatstroke is likely to occur may be as low
as 85° to 90°F.
Fever Caused by Brain Lesions. When a brain surgeon
When the body temperature rises beyond a critical
temperature, into the range of 105° to 108°F, the person
operates in the region of the hypothalamus, severe
is likely to develop heatstroke. The symptoms include
fever almost always occurs; rarely, the opposite effect,
dizziness, abdominal distress sometimes accompanied
hypothermia, occurs, demonstrating both the potency
by vomiting, sometimes delirium, and eventually loss of
of the hypothalamic mechanisms for body tempera-
consciousness if the body temperature is not soon
ture control and the ease with which abnormalities of
decreased. These symptoms are often exacerbated by a
the hypothalamus can alter the set-point of tempera-
degree of circulatory shock brought on by excessive loss
ture control. Another condition that frequently causes
of fluid and electrolytes in the sweat.
prolonged high temperature is compression of the
The hyperpyrexia itself is also exceedingly damaging
hypothalamus by a brain tumor.
to the body tissues, especially the brain, and is respon-
sible for many of the effects. In fact, even a few minutes
Characteristics of Febrile Conditions
of very high body temperature can sometimes be
Chills. When the set-point of the hypothalamic
fatal. For this reason, many authorities recommend
temperature-control center is suddenly changed from
immediate treatment of heatstroke by placing the
900
Unit XIII
Metabolism and Temperature Regulation
person in a cold water bath. Because this often induces
Cold-Induced Vasodilation Is a Final Protection Against
uncontrollable shivering, with a considerable increase in
Frostbite at Almost Freezing Temperatures. When the
the rate of heat production, others have suggested that
temperature of tissues falls almost to freezing, the
sponge or spray cooling of the skin is likely to be
smooth muscle in the vascular wall becomes paralyzed
more effective for rapidly decreasing the body core
because of the cold itself, and sudden vasodilation
temperature.
occurs, often manifested by a flush of the skin. This
mechanism helps prevent frostbite by delivering warm
Harmful Effects of High Temperature. The pathological find-
blood to the skin. This mechanism is far less developed
ings in a person who dies of hyperpyrexia are local
in humans than in most lower animals that live in the
hemorrhages and parenchymatous degeneration of cells
cold all the time.
throughout the entire body, but especially in the brain.
Once neuronal cells are destroyed, they can never be
Artificial Hypothermia. It is easy to decrease the tempera-
replaced. Also, damage to the liver, kidneys, and other
ture of a person by first administering a strong sedative
organs can often be severe enough that failure of one
to depress the reactivity of the hypothalamic tempera-
or more of these organs eventually causes death, but
ture controller and then cooling the person with ice or
sometimes not until several days after the heatstroke.
cooling blankets until the temperature falls. The tem-
Acclimatization to Heat. It can be extremely important
perature can then be maintained below 90°F for several
to acclimatize people to extreme heat. Examples of
days to a week or more by continual sprinkling of cool
people requiring acclimatization are soldiers on duty
water or alcohol on the body. Such artificial cooling has
in the tropics and miners working in the 2-mile-deep
been used during heart surgery so that the heart can be
gold mines of South Africa, where the temperature
stopped artificially for many minutes at a time. Cooling
approaches body temperature and the humidity
to this extent does not cause tissue damage, but it does
approaches 100 per cent. A person exposed to heat for
slow the heart and greatly depresses cell metabolism,
several hours each day while performing a reasonably
so that the body’s cells can survive 30 minutes to more
heavy workload will develop increased tolerance to hot
than 1 hour without blood flow during the surgical
and humid conditions in 1 to 3 weeks.
procedure.
Among the most important physiological changes
that occur during this acclimatization process are an
approximately twofold increase in the maximum rate of
References
sweating, an increase in plasma volume, and diminished
loss of salt in the sweat and urine to almost none; the
Aronoff DM, Neilson EG: Antipyretics: mechanisms of
last two effects result from increased secretion of aldos-
action and clinical use in fever suppression. Am J Med
terone by the adrenal glands.
111:304, 2001.
Blatteis CM, Li S, Li Z, et al: Signaling the brain in systemic
inflammation: the role of complement. Front Biosci 9:915,
Exposure of the Body to
2004.
Extreme Cold
Boulant JA: Hypothalamic neurons. Mechanisms of sensi-
tivity to temperature. Ann N Y Acad Sci 856:108, 1998.
Unless treated immediately, a person exposed to ice
Conti B, Tabarean I, Andrei C, Bartfai T: Cytokines and
water for 20 to 30 minutes ordinarily dies because of
fever. Front Biosci 9:1433, 2004.
heart standstill or heart fibrillation. By that time, the
Florez-Duquet M, McDonald RB: Cold-induced thermoreg-
internal body temperature will have fallen to about
ulation and biological aging. Physiol Rev 78:339, 1998.
77°F. If warmed rapidly by the application of external
Gourine AV, Dale N, Gourine VN, Spyer KM: Fever in sys-
heat, the person’s life can often be saved.
temic inflammation: roles of purines. Front Biosci 9:1011,
Loss of Temperature Regulation at Low Temperatures. As noted
2004.
in Figure 73-10, once the body temperature has fallen
Hildebrand F, Giannoudis PV, van Griensven M, et al:
below about 85°F, the ability of the hypothalamus to
Pathophysiologic changes and effects of hypothermia on
regulate temperature is lost; it is greatly impaired even
outcome in elective surgery and trauma patients. Am J
when the body temperature falls below about 94°F. Part
Surg 187:363, 2004.
of the reason for this diminished temperature regula-
Ivanov AI, Romanovsky AA: Prostaglandin E2 as a media-
tion is that the rate of chemical heat production in each
tor of fever: synthesis and catabolism. Front Biosci 9:1977,
cell is depressed almost twofold for each 10°F decrease
2004.
in body temperature. Also, sleepiness develops (later
Katschinski DM: On heat and cells and proteins. News
followed by coma), which depresses the activity of the
Physiol Sci 19:11, 2004.
central nervous system heat control mechanisms and
Kenney WL, Munce TA: Aging and human temperature reg-
prevents shivering.
ulation. J Appl Physiol 95:2598, 2003.
Kozak W, Kluger MJ, Tesfaigzi J, et al: Molecular mechanisms
Frostbite. When the body is exposed to extremely low
of fever and endogenous antipyresis. Ann N Y Acad Sci
temperatures, surface areas can freeze; the freezing is
917:121, 2000.
called frostbite. This occurs especially in the lobes of the
Leon LR: Cytokine regulation of fever: studies using gene
ears and in the digits of the hands and feet. If the freeze
knockout mice. J Appl Physiol 92:2648, 2002.
has been sufficient to cause extensive formation of ice
McDermott MF: Genetic clues to understanding periodic
crystals in the cells, permanent damage usually results,
fevers, and possible therapies. Trends Mol Med 8:550,
such as permanent circulatory impairment as well as
2002.
local tissue damage. Often gangrene follows thawing,
Morrison SF: Central pathways controlling brown adipose
and the frostbitten areas must be removed surgically.
tissue thermogenesis. News Physiol Sci 19:67, 2004.
Chapter 73
Body Temperature, Temperature Regulation, and Fever
901
Olsen TS, Weber UJ, Kammersgaard LP: Therapeutic
Saper CB: Neurobiological basis of fever. Ann N Y Acad Sci
hypothermia for acute stroke. Lancet Neurol 2:410, 2003.
856:90, 1998.
Patapoutian A, Peier AM, Story GM, Viswanath V: Ther-
Tatro JB, Sinha PS: The central melanocortin system and
moTRP channels and beyond: mechanisms of tempera-
fever. Ann N Y Acad Sci 994:246, 2003.
ture sensation. Nat Rev Neurosci 4:529, 2003.
van Marken Lichtenbelt WD, Daanen HA: Cold-induced
Roth J, Zeisberger E, Vybiral S, Jansky L: Endogenous
metabolism. Curr Opin Clin Nutr Metab Care 6:469, 2003.
antipyretics: neuropeptides and glucocorticoids. Front
Biosci 9:816, 2004.
U N I T XIV
Endocrinology and
Reproduction
74. Introduction to Endocrinology
75. Pituitary Hormones and Their Control
by the Hypothalamus
76. Thyroid Metabolic Hormones
77. Adrenocortical Hormones
78. Insulin, Glucagon, and Diabetes Mellitus
79. Parathyroid Hormone, Calcitonin, Calcium and
Phosphate Metabolism, Vitamin D, Bone, and Teeth
80. Reproductive and Hormonal Functions of the Male
81. Female Physiology Before Pregnancy
and the Female Hormones
82. Pregnancy and Lactation
83. Fetal and Neonatal Physiology
C H A P T E R
7
4
Introduction to Endocrinology
Coordination of Body
Functions by Chemical
Messengers
The multiple activities of the cells, tissues, and
organs of the body are coordinated by the interplay
of several types of chemical messenger systems:
1. Neurotransmitters are released by axon terminals of neurons into the
synaptic junctions and act locally to control nerve cell functions.
2. Endocrine hormones are released by glands or specialized cells into the
circulating blood and influence the function of cells at another location in
the body.
3. Neuroendocrine hormones are secreted by neurons into the circulating
blood and influence the function of cells at another location in the body.
4. Paracrines are secreted by cells into the extracellular fluid and affect
neighboring cells of a different type.
5. Autocrines are secreted by cells into the extracellular fluid and affect the
function of the same cells that produced them by binding to cell surface
receptors.
6. Cytokines are peptides secreted by cells into the extracellular fluid and can
function as autocrines, paracrines, or endocrine hormones. Examples of
cytokines include the interleukins and other lymphokines that are secreted
by helper cells and act on other cells of the immune system (see Chapter
34). Cytokine hormones (e.g., leptin) produced by adipocytes are
sometimes called adipokines.
In the next few chapters, we discuss mainly the endocrine and neuroendocrine
hormone systems, keeping in mind that many of the body’s chemical messen-
ger systems interact with one another to maintain homeostasis. For example,
the adrenal medullae and the pituitary gland secrete their hormones primarily
in response to neural stimuli. The neuroendocrine cells, located in the hypo-
thalamus, have axons that terminate in the posterior pituitary gland and median
eminence and secrete several neurohormones, including antidiuretic hormone
(ADH), oxytocin, and hypophysiotropic hormones, which control the secretion
of anterior pituitary hormones.
The endocrine hormones are carried by the circulatory system to cells
throughout the body, including the nervous system in some cases, where they
bind with receptors and initiate many reactions. Some endocrine hormones
affect many different types of cells of the body; for example, growth hormone
(from the anterior pituitary gland) causes growth in most parts of the body, and
thyroxine (from the thyroid gland) increases the rate of many chemical reac-
tions in almost all the body’s cells.
Other hormones affect only specific target tissues, because only these tissues
have receptors for the hormone. For example, adrenocorticotropic hormone
(ACTH) from the anterior pituitary gland specifically stimulates the adrenal
cortex, causing it to secrete adrenocortical hormones, and the ovarian hormones
have specific effects on the female sex organs as well as on the secondary sexual
characteristics of the female body.
Figure 74-1 shows the anatomical loci of the major endocrine glands and
endocrine tissues of the body, except for the placenta, which is an additional
source of the sex hormones. Table 74-1 provides an overview of the different
hormone systems and their most important actions.
905
906
Unit XIV Endocrinology and Reproduction
Chemical Structure and
Hypothalamus
Synthesis of Hormones
There are three general classes of hormones:
Pineal gland
1. Proteins and polypeptides, including hormones
Pituitary gland
secreted by the anterior and posterior pituitary
gland, the pancreas (insulin and glucagon), the
parathyroid gland (parathyroid hormone), and
many others (see Table 74-1).
2. Steroids secreted by the adrenal cortex (cortisol
Parathyroid glands
and aldosterone), the ovaries (estrogen and
(behind thyroid gland)
Thyroid gland
progesterone), the testes (testosterone), and the
Thymus gland
placenta (estrogen and progesterone).
3. Derivatives of the amino acid tyrosine, secreted
by the thyroid (thyroxine and triiodothyronine)
and the adrenal medullae (epinephrine and
norepinephrine). There are no known
polysaccharides or nucleic acid hormones.
Polypeptide and Protein Hormones Are Stored in Secretory Vesi-
cles Until Needed. Most of the hormones in the body
Stomach
are polypeptides and proteins. These hormones range
in size from small peptides with as few as 3 amino
Adrenal
glands
acids
(thyrotropin-releasing hormone) to proteins
with almost 200 amino acids (growth hormone and
Pancreas
Adipose
prolactin). In general, polypeptides with 100 or more
Kidney
tissue
amino acids are called proteins, and those with fewer
than 100 amino acids are referred to as peptides.
Small
Protein and peptide hormones are synthesized on
intestine
the rough end of the endoplasmic reticulum of the dif-
ferent endocrine cells, in the same fashion as most
other proteins (Figure 74-2). They are usually synthe-
sized first as larger proteins that are not biologically
Ovaries
active
(preprohormones) and are cleaved to form
(female)
smaller prohormones in the endoplasmic reticulum.
These are then transferred to the Golgi apparatus for
packaging into secretory vesicles. In this process,
enzymes in the vesicles cleave the prohormones to
Testes (male)
produce smaller, biologically active hormones and
inactive fragments. The vesicles are stored within the
Figure 74-1
cytoplasm, and many are bound to the cell membrane
until their secretion is needed. Secretion of the hor-
Anatomical loci of the principal endocrine glands and tissues of
mones (as well as the inactive fragments) occurs when
the body.
the secretory vesicles fuse with the cell membrane and
the granular contents are extruded into the interstitial
fluid or directly into the blood stream by exocytosis.
In many cases, the stimulus for exocytosis is an
increase in cytosolic calcium concentration caused by
The multiple hormone systems play a key role in
depolarization of the plasma membrane. In other
regulating almost all body functions, including metab-
instances, stimulation of an endocrine cell surface
olism, growth and development, water and electrolyte
receptor causes increased cyclic adenosine monophos-
balance, reproduction, and behavior. For instance,
phate (cAMP) and subsequently activation of protein
without growth hormone, a person would be a dwarf.
kinases that initiate secretion of the hormone. The
Without thyroxine and triiodothyronine from the
peptide hormones are water soluble, allowing them to
thyroid gland, almost all the chemical reactions of the
enter the circulatory system easily, where they are
body would become sluggish, and the person would
carried to their target tissues.
become sluggish as well. Without insulin from the pan-
creas, the body’s cells could use little of the food car-
Steroid Hormones Are Usually Synthesized from Cholesterol and
bohydrates for energy. And without the sex hormones,
Are Not Stored. The chemical structure of steroid hor-
sexual development and sexual functions would be
mones is similar to that of cholesterol, and in most
absent.
instances they are synthesized from cholesterol itself.
Chapter 74
Introduction to Endocrinology
907
Table 74-1
Endocrine Glands, Hormones, and Their Functions and Structure
Chemical
Gland/Tissue
Hormones
Major Functions
Structure
Hypothalamus
Thyrotropin-releasing hormone (TRH)
Stimulates secretion of TSH and prolactin
Peptide
(Chapter 75)
Corticotropin-releasing hormone (CRH)
Causes release of ACTH
Peptide
Growth hormone-releasing hormone (GHRH)
Causes release of growth hormone
Peptide
Growth hormone inhibitory hormone (GHIH)
Inhibits release of growth hormone
Peptide
(somatostatin)
Gonadotropin-releasing hormone (GnRH)
Causes release of LH and FSH
Dopamine or prolactin-inhibiting factor (PIF)
Inhibits release of prolactin
Amine
Anterior pituitary
Growth hormone
Stimulates protein synthesis and overall growth
Peptide
(Chapter 75)
of most cells and tissues
Thyroid-stimulating hormone (TSH)
Stimulates synthesis and secretion of thyroid
Peptide
hormones (thyroxine and triiodothyronine)
Adrenocorticotropic hormone (ACTH)
Stimulates synthesis and secretion of adrenocortical
Peptide
hormones (cortisol, androgens, and aldosterone)
Prolactin
Promotes development of the female breasts and
Peptide
secretion of milk
Follicle-stimulating hormone (FSH)
Causes growth of follicles in the ovaries and sperm
Peptide
maturation in Sertoli cells of testes
Luteinizing hormone (LH)
Stimulates testosterone synthesis in Leydig cells of
Peptide
testes; stimulates ovulation, formation of corpus
luteum, and estrogen and progesterone synthesis
in ovaries
Posterior pituitary
Antidiuretic hormone (ADH) (also called
Increases water reabsorption by the kidneys and
Peptide
(Chapter 75)
vasopressin)
causes vasoconstriction and increased blood
pressure
Oxytocin
Stimulates milk ejection from breasts and uterine
Peptide
contractions
Thyroid
Thyroxine (T4) and triiodothyronine (T3)
Increases the rates of chemical reactions in most
Amine
(Chapter 76)
cells, thus increasing body metabolic rate
Calcitonin
Promotes deposition of calcium in the bones and
Peptide
decreases extracellular fluid calcium ion
concentration
Adrenal cortex
Cortisol
Has multiple metabolic functions for controlling
Steroid
(Chapter 77)
metabolism of proteins, carbohydrates, and fats;
also has anti-inflammatory effects
Aldosterone
Increases renal sodium reabsorption, potassium
Steroid
secretion, and hydrogen ion secretion
Adrenal medulla
Norepinephrine, epinephrine
Same effects as sympathetic stimulation
Amine
(Chapter 60)
Pancreas
Insulin (b cells)
Promotes glucose entry in many cells, and in
Peptide
(Chapter 78)
this way controls carbohydrate metabolism
Glucagon (a cells)
Increases synthesis and release of glucose from
Peptide
the liver into the body fluids
Parathyroid
Parathyroid hormone (PTH)
Controls serum calcium ion concentration by
Peptide
(Chapter 79)
increasing calcium absorption by the gut and
kidneys and releasing calcium from bones
Testes
Testosterone
Promotes development of male reproductive
Steroid
(Chapter 80)
system and male secondary sexual characteristics
Ovaries
Estrogens
Promotes growth and development of female
Steroid
(Chapter 81)
reproductive system, female breasts, and female
secondary sexual characteristics
Progesterone
Stimulates secretion of “uterine milk” by the uterine
Steroid
endometrial glands and promotes development of
secretory apparatus of breasts
Placenta
Human chorionic gonadotropin (HCG)
Promotes growth of corpus luteum and secretion of
Peptide
(Chapter 82)
estrogens and progesterone by corpus luteum
Human somatomammotropin
Probably helps promote development of some
Peptide
fetal tissues as well as the mother’s breasts
Estrogens
See actions of estrogens from ovaries
Steroid
Progesterone
See actions of progesterone from ovaries
Steroid
Kidney
Renin
Catalyzes conversion of angiotensinogen to
Peptide
(Chapter 26)
angiotensin I (acts as an enzyme)
1,25-Dihydroxycholecalciferol
Increases intestinal absorption of calcium and bone
Steroid
mineralization
Erythropoietin
Increases erythrocyte production
Peptide
Heart
Atrial natriuretic peptide (ANP)
Increases sodium excretion by kidneys, reduces
Peptide
(Chapter 22)
blood pressure
Stomach
Gastrin
Stimulates HCl secretion by parietal cells
Peptide
(Chapter 64)
Small intestine
Secretin
Stimulates pancreatic acinar cells to release
Peptide
(Chapter 64)
bicarbonate and water
Cholecystokinin (CCK)
Stimulates gallbladder contraction and release of
Peptide
pancreatic enzymes
Adipocytes
Leptin
Inhibits appetite, stimulates thermogenesis
Peptide
(Chapter 71)
908
Unit XIV
Endocrinology and Reproduction
Nucleus
DNA
CH2OH
O CH2OH
C O
C O
HC
HO
HO
OH
Transcription
Synthesis
mRNA
O
O
Cortisol
Aldosterone
Translation
OH
OH
Endoplasmic
reticulum
Packaging
O
HO
Testosterone
Estradiol
Golgi
apparatus
Figure 74-3
Chemical structures of several steroid hormones.
Secretory
Storage
vesicles
Ca++
hormones are then released into the blood stream.
cAMP
After entering the blood, most of the thyroid hor-
mones combine with plasma proteins, especially thy-
Secretion
roxine-binding globulin, which slowly releases the
Extracellular
Stimulus
hormones to the target tissues.
fluid
Epinephrine and norepinephrine are formed in the
Figure 74-2
adrenal medulla, which normally secretes about four
times more epinephrine than norepinephrine. Cate-
Synthesis and secretion of peptide hormones. The stimulus for
cholamines are taken up into preformed vesicles and
hormone secretion often involves changes in intracellular calcium
stored until secreted. Similar to the protein hormones
or changes in cyclic adenosine monophosphate (cAMP) in the
cell.
stored in secretory granules, catecholamines are also
released from adrenal medullary cells by exocytosis.
Once the catecholamines enter the circulation, they
They are lipid soluble and consist of three cyclohexyl
can exist in the plasma in free form or in conjugation
rings and one cyclopentyl ring combined into a single
with other substances.
structure (Figure 74-3).
Although there is usually very little hormone
Hormone Secretion, Transport,
storage in steroid-producing endocrine cells, large
stores of cholesterol esters in cytoplasm vacuoles can
and Clearance from the Blood
be rapidly mobilized for steroid synthesis after a stim-
ulus. Much of the cholesterol in steroid-producing cells
Onset of Hormone Secretion After a Stimulus, and Duration of
comes from the plasma, but there is also de novo
Action of Different Hormones. Some hormones, such as
synthesis of cholesterol in steroid-producing cells.
norepinephrine and epinephrine, are secreted within
Because the steroids are highly lipid soluble, once they
seconds after the gland is stimulated, and they may
are synthesized, they simply diffuse across the cell
develop full action within another few seconds to
membrane and enter the interstitial fluid and then the
minutes; the actions of other hormones, such as thy-
blood.
roxine or growth hormone, may require months for
full effect. Thus, each of the different hormones has its
Amine Hormones Are Derived from Tyrosine. The two groups
own characteristic onset and duration of action—each
of hormones derived from tyrosine, the thyroid and
tailored to perform its specific control function.
the adrenal medullary hormones, are formed by the
actions of enzymes in the cytoplasmic compartments
Concentrations of Hormones in the Circulating Blood, and Hor-
of the glandular cells. The thyroid hormones are
monal Secretion Rates. The concentrations of hormones
synthesized and stored in the thyroid gland and incor-
required to control most metabolic and endocrine
porated into macromolecules of the protein thy-
functions are incredibly small. Their concentrations in
roglobulin, which is stored in large follicles within
the blood range from as little as 1 picogram (which is
the thyroid gland. Hormone secretion occurs when
one millionth of one millionth of a gram) in each mil-
the amines are split from thyroglobulin, and the free
liliter of blood up to at most a few micrograms (a few
Chapter 74
Introduction to Endocrinology
909
millionths of a gram) per milliliter of blood. Similarly,
of neural pathways involved in controlling hormone
the rates of secretion of the various hormones are
release.
extremely small, usually measured in micrograms or
milligrams per day. We shall see later in this chapter
that highly specialized mechanisms are available in the
Transport of Hormones in the Blood
target tissues that allow even these minute quantities
of hormones to exert powerful control over the phys-
Water-soluble hormones
(peptides and cate-
iological systems.
cholamines) are dissolved in the plasma and trans-
ported from their sites of synthesis to target tissues,
where they diffuse out of the capillaries, into the inter-
Feedback Control of
stitial fluid, and ultimately to target cells.
Hormone Secretion
Steroid and thyroid hormones, in contrast, circulate
in the blood mainly bound to plasma proteins. Usually
Negative Feedback Prevents Overactivity of Hormone Systems.
less than 10 per cent of steroid or thyroid hormones in
Although the plasma concentrations of many hor-
the plasma exist free in solution. For example, more
mones fluctuate in response to various stimuli that
than 99 per cent of the thyroxine in the blood is bound
occur throughout the day, all hormones studied thus
to plasma proteins. However, protein-bound hor-
far appear to be closely controlled. In most instances,
mones cannot easily diffuse across the capillaries and
this control is exerted through negative feedback mech-
gain access to their target cells and are therefore bio-
anisms that ensure a proper level of hormone activity
logically inactive until they dissociate from plasma
at the target tissue. After a stimulus causes release of
proteins.
the hormone, conditions or products resulting from the
The relatively large amounts of hormones bound to
action of the hormone tend to suppress its further
proteins serve as reservoirs, replenishing the concen-
release. In other words, the hormone (or one of its
tration of free hormones when they are bound to
products) has a negative feedback effect to prevent
target receptors or lost from the circulation. Binding
oversecretion of the hormone or overactivity at the
of hormones to plasma proteins greatly slows their
target tissue.
clearance from the plasma.
The controlled variable is often not the secretory
rate of the hormone itself but the degree of activity of
the target tissue. Therefore, only when the target tissue
“Clearance” of Hormones
activity rises to an appropriate level will feedback
from the Blood
signals to the endocrine gland become powerful
enough to slow further secretion of the hormone.
Two factors can increase or decrease the concentration
Feedback regulation of hormones can occur at all
of a hormone in the blood. One of these is the rate of
levels, including gene transcription and translation
hormone secretion into the blood. The second is
steps involved in the synthesis of hormones and steps
the rate of removal of the hormone from the blood,
involved in processing hormones or releasing stored
which is called the metabolic clearance rate. This
hormones.
is usually expressed in terms of the number of milli-
liters of plasma cleared of the hormone per minute.
Surges of Hormones Can Occur with Positive Feedback. In a
To calculate this clearance rate, one measures (1)
few instances, positive feedback occurs when the bio-
the rate of disappearance of the hormone from the
logical action of the hormone causes additional secre-
plasma per minute and (2) the concentration of the
tion of the hormone. One example of this is the surge
hormone in each milliliter of plasma. Then, the meta-
of luteinizing hormone (LH) that occurs as a result of
bolic clearance rate is calculated by the following
the stimulatory effect of estrogen on the anterior pitu-
formula:
itary before ovulation. The secreted LH then acts on
Metabolic clearance rate = Rate of disappearance
the ovaries to stimulate additional secretion of estro-
of hormone from the plasma/Concentration of
gen, which in turn causes more secretion of LH. Even-
hormone in each milliliter of plasma
tually, LH reaches an appropriate concentration, and
typical negative feedback control of hormone secre-
The usual procedure for making this measurement
tion is then exerted.
is the following: A purified solution of the hormone to
be measured is tagged with a radioactive substance.
Cyclical Variations Occur in Hormone Release. Superim-
Then the radioactive hormone is infused at a constant
posed on the negative and positive feedback control
rate into the blood stream until the radioactive con-
of hormone secretion are periodic variations in
centration in the plasma becomes steady. At this time,
hormone release that are influenced by seasonal
the rate of disappearance of the radioactive hormone
changes, various stages of development and aging, the
from the plasma equals the rate at which it is infused,
diurnal (daily) cycle, and sleep. For example, the secre-
which gives one the rate of disappearance. At the same
tion of growth hormone is markedly increased during
time, the plasma concentration of the radioactive
the early period of sleep but is reduced during the later
hormone is measured using a standard radioactive
stages of sleep. In many cases, these cyclical variations
counting procedure. Then, using the formula just cited,
in hormone secretion are due to changes in activity
the metabolic clearance rate is calculated.
910
Unit XIV Endocrinology and Reproduction
Hormones are “cleared” from the plasma in several
2. In the cell cytoplasm. The primary receptors for
ways, including
(1) metabolic destruction by the
the different steroid hormones are found mainly
tissues, (2) binding with the tissues, (3) excretion by the
in the cytoplasm.
liver into the bile, and (4) excretion by the kidneys into
3. In the cell nucleus. The receptors for the thyroid
the urine. For certain hormones, a decreased metabolic
hormones are found in the nucleus and are
clearance rate may cause an excessively high concen-
believed to be located in direct association with
tration of the hormone in the circulating body fluids.
one or more of the chromosomes.
For instance, this occurs for several of the steroid hor-
mones when the liver is diseased, because these hor-
The Number and Sensitivity of Hormone Receptors Are Regu-
mones are conjugated mainly in the liver and then
lated. The number of receptors in a target cell usually
“cleared” into the bile.
does not remain constant from day to day, or even
Hormones are sometimes degraded at their target
from minute to minute. The receptor proteins them-
cells by enzymatic processes that cause endocytosis of
selves are often inactivated or destroyed during the
the cell membrane hormone-receptor complex; the
course of their function, and at other times they
hormone is then metabolized in the cell, and the recep-
are reactivated or new ones are manufactured by
tors are usually recycled back to the cell membrane.
the protein-manufacturing mechanism of the cell.
Most of the peptide hormones and catecholamines
For instance, increased hormone concentration and
are water soluble and circulate freely in the blood.
increased binding with its target cell receptors some-
They are usually degraded by enzymes in the blood
times cause the number of active receptors to
and tissues and rapidly excreted by the kidneys and
decrease. This down-regulation of the receptors can
liver, thus remaining in the blood for only a short time.
occur as a result of (1) inactivation of some of the
For example, the half-life of angiotensin II circulating
receptor molecules, (2) inactivation of some of the
in the blood is less than a minute.
intracellular protein signaling molecules, (3) tempo-
Hormones that are bound to plasma proteins are
rary sequestration of the receptor to the inside of the
cleared from the blood at much slower rates and may
cell, away from the site of action of hormones that
remain in the circulation for several hours or even
interact with cell membrane receptors, (4) destruction
days. The half-life of adrenal steroids in the circulation,
of the receptors by lysosomes after they are internal-
for example, ranges between 20 and 100 minutes,
ized, or (5) decreased production of the receptors. In
whereas the half-life of the protein-bound thyroid hor-
each case, receptor down-regulation decreases the
mones may be as long as 1 to 6 days.
target tissue’s responsiveness to the hormone.
Some hormones cause up-regulation of receptors
and intracellular signaling proteins; that is, the stimu-
lating hormone induces greater than normal formation
Mechanisms of Action
of receptor or intracellular signaling molecules by the
of Hormones
protein-manufacturing machinery of the target cell, or
greater availability of the receptor for interaction with
Hormone Receptors and Their
the hormone. When this occurs, the target tissue
Activation
becomes progressively more sensitive to the stimulat-
ing effects of the hormone.
The first step of a hormone’s action is to bind to spe-
cific receptors at the target cell. Cells that lack recep-
tors for the hormones do not respond. Receptors
Intracellular Signaling After Hormone
for some hormones are located on the target cell
Receptor Activation
membrane, whereas other hormone receptors are
located in the cytoplasm or the nucleus. When the
Almost without exception, a hormone affects its target
hormone combines with its receptor, this usually initi-
tissues by first forming a hormone-receptor complex.
ates a cascade of reactions in the cell, with each stage
This alters the function of the receptor itself, and the
becoming more powerfully activated so that even
activated receptor initiates the hormonal effects. To
small concentrations of the hormone can have a large
explain this, let us give a few examples of the different
effect.
types of interactions.
Hormonal receptors are large proteins, and each cell
that is to be stimulated usually has some 2000 to
Ion Channel-Linked Receptors. Virtually all the neuro-
100,000 receptors. Also, each receptor is usually highly
transmitter substances, such as acetylcholine and
specific for a single hormone; this determines the type
norepinephrine, combine with receptors in the postsy-
of hormone that will act on a particular tissue. The
naptic membrane. This almost always causes a change
target tissues that are affected by a hormone are those
in the structure of the receptor, usually opening or
that contain its specific receptors.
closing a channel for one or more ions. Some of these
The locations for the different types of hormone
ion channel-linked receptors open (or close) channels
receptors are generally the following:
for sodium ions, others for potassium ions, others for
1. In or on the surface of the cell membrane. The
calcium ions, and so forth. The altered movement of
membrane receptors are specific mostly for the
these ions through the channels causes the subsequent
protein, peptide, and catecholamine hormones.
effects on the postsynaptic cells. Although a few
Chapter 74
Introduction to Endocrinology
911
hormones may exert some of their actions through
such as adenylyl cyclase or phospholipase C, which
activation of ion channel receptors, most hormones
alters cell function.
that open or close ions channels do this indirectly by
The signaling event is rapidly terminated when the
coupling with G protein-linked or enzyme-linked
hormone is removed and the a subunit inactivates
receptors, as discussed next.
itself by converting its bound GTP to GDP; then the
a subunit once again combines with the b and g sub-
G Protein-Linked Hormone Receptors. Many hormones
units to form an inactive, membrane-bound trimeric G
activate receptors that indirectly regulate the activity
protein.
of target proteins (e.g., enzymes or ion channels) by
Some hormones are coupled to inhibitory G pro-
coupling with groups of cell membrane proteins called
teins
(denoted Gi proteins), whereas others are
heterotrimeric GTP-binding proteins
(G proteins)
coupled to stimulatory G proteins (denoted Gs pro-
(Figure 74-4). There are more than 1000 known G
teins). Thus, depending on the coupling of a hormone
protein-coupled receptors, all of which have seven
receptor to an inhibitory or stimulatory G protein, a
transmembrane segments that loop in and out of the
hormone can either increase or decrease the activity
cell membrane. Some parts of the receptor that pro-
of intracellular enzymes. This complex system of cell
trude into the cell cytoplasm (especially the cytoplas-
membrane G proteins provides a vast array of poten-
mic tail of the receptor) are coupled to G proteins that
tial cell responses to different hormones in the various
include three (i.e., trimeric) parts—the a, b, and g sub-
target tissues of the body.
units. When the ligand (hormone) binds to the extra-
cellular part of the receptor, a conformational change
Enzyme-Linked Hormone Receptors. Some receptors,
occurs in the receptor that activates the G proteins and
when activated, function directly as enzymes or are
induces intracellular signals that either (1) open or
closely associated with enzymes that they activate.
close cell membrane ion channels or (2) change the
These enzyme-linked receptors are proteins that pass
activity of an enzyme in the cytoplasm of the cell.
through the membrane only once, in contrast to the
The trimeric G proteins are named for their ability
seven-transmembrane G protein-coupled receptors.
to bind guanosine nucleotides. In their inactive state,
Enzyme-linked receptors have their hormone-binding
the a, b, and g subunits of G proteins form a complex
site on the outside of the cell membrane and their cat-
that binds guanosine diphosphate (GDP) on the a
alytic or enzyme-binding site on the inside. When the
subunit. When the receptor is activated, it undergoes
hormone binds to the extracellular part of the recep-
a conformational change that causes the GDP-bound
tor, an enzyme immediately inside the cell membrane
trimeric G protein to associate with the cytoplasmic
is activated
(or occasionally inactivated). Although
part of the receptor and to exchange GDP for guano-
many enzyme-linked receptors have intrinsic enzyme
sine triphosphate (GTP). Displacement of GDP by
activity, others rely on enzymes that are closely asso-
GTP causes the a subunit to dissociate from the
ciated with the receptor to produce changes in cell
trimeric complex and to associate with other intracel-
function.
lular signaling proteins; these proteins, in turn, alter
One example of an enzyme-linked receptor is the
the activity of ion channels or intracellular enzymes
leptin receptor
(Figure 74-5). Leptin is a hormone
Hormone
Receptor
Extracellular
fluid
g
Cytoplasm
g
a
g
b
b
a
a
b
GDP
G-protein
GTP activated
GTP
(inactive)
target protein
G-protein
(enzyme)
(active)
Figure 74-4
Mechanism of activation of a G protein-coupled receptor. When the hormone activates the receptor, the inactive a, b, and g G protein
complex associates with the receptor and is activated, with an exchange of guanosine triphosphate (GTP) for guanosine diphosphate
(GDP). This causes the a subunit (to which the GTP is bound) to dissociate from the b and g subunits of the G protein and to interact with
membrane-bound target proteins (enzymes) that initiate intracellular signals.
912
Unit XIV Endocrinology and Reproduction
whereas other actions occur more slowly and require
Leptin
synthesis of new proteins.
Another example, one widely used in hormonal
control of cell function, is for the hormone to bind
Leptin receptor
with a special transmembrane receptor, which then
P
P
becomes the activated enzyme adenylyl cyclase at the
Jak2
Jak2
Y
end that protrudes to the interior of the cell. This
Y
cyclase catalyzes the formation of cAMP, which has a
Y
Stat3
P
Y
P
Stat3
Activation
multitude of effects inside the cell to control cell activ-
of enzymes
P
Y
Y
P
ity, as discussed in greater detail later. cAMP is called
a second messenger because it is not the hormone itself
P
that directly institutes the intracellular changes;
instead, the cAMP serves as a second messenger to
Physiological
cause these effects.
effects
P
For a few peptide hormones, such as atrial natri-
Stat3
uretic peptide
(ANP), cyclic guanosine monophos-
phate (cGMP), which is only slightly different from
Stat3
Translation
cAMP, serves in a similar manner as a second
messenger.
Intracellular Hormone Receptors and Activation of Genes.
Target gene
mRNA
Several hormones, including adrenal and gonadal
steroid hormones, thyroid hormones, retinoid hor-
mones, and vitamin D, bind with protein receptors
Figure 74-5
inside the cell rather than in the cell membrane.
Because these hormones are lipid soluble, they readily
An enzyme-linked receptor—the leptin receptor. The receptor
cross the cell membrane and interact with receptors in
exists as a homodimer (two identical parts), and leptin binds to
the cytoplasm or nucleus. The activated hormone-
the extracellular part of the receptor, causing phosphorylation and
activation of the intracellular associated janus kinase 2 (JAK2).
receptor complex then binds with a specific regulatory
This causes phosphorylation of signal transducer and activator of
(promoter) sequence of the DNA called the hormone
transcription (STAT) proteins, which then activates the transcrip-
response element, and in this manner either activates
tion of target genes and the synthesis of proteins. JAK2 phos-
or represses transcription of specific genes and for-
phorylation also activates several other enzyme systems that
mation of messenger RNA (mRNA) (Figure 74-6).
mediate some of the more rapid effects of leptin.
Therefore, minutes, hours, or even days after the
hormone has entered the cell, newly formed proteins
appear in the cell and become the controllers of new
or altered cellular functions.
secreted by fat cells and has many physiological
Many different tissues have identical intracellular
effects, but it is especially important in regulating
hormone receptors, but the genes that the receptors
appetite and energy balance, as discussed in Chapter
regulate are different in the various tissues. An intra-
71. The leptin receptor is a member of a large family
cellular receptor can activate a gene response only if
of cytokine receptors that do not themselves contain
the appropriate combination of gene regulatory pro-
enzymatic activity but signal through associated
teins is present, and many of these regulatory proteins
enzymes. In the case of the leptin receptor, one of the
are tissue specific. Thus, the responses of different
signaling pathways occurs through a tyrosine kinase of
tissues to a hormone are determined not only by the
the janus kinase (JAK) family, JAK2. The leptin recep-
specificity of the receptors but also by the genes that
tor exists as a dimer (i.e., in two parts), and binding of
the receptor regulates.
leptin to the extracellular part of the receptor alters its
conformation, enabling phosphorylation and activa-
tion of the intracellular associated JAK2 molecules.
Second Messenger Mechanisms
The activated JAK2 molecules then phosphorylate
for Mediating Intracellular
other tyrosine residues within the leptin receptor-
JAK2 complex to mediate intracellular signaling. The
Hormonal Functions
intracellular signals include phosphorylation of signal
transducer and activator of transcription (STAT) pro-
We noted earlier that one of the means by which hor-
teins, which activates transcription by leptin target
mones exert intracellular actions is to stimulate for-
genes to initiate protein synthesis. Phosphorylation of
mation of the second messenger cAMP inside the cell
JAK2 also leads to activation of other intracellular
membrane. The cAMP then causes subsequent intra-
enzyme pathways such as mitogen-activated protein
cellular effects of the hormone. Thus, the only direct
kinases
(MAPK) and phosphatidylinositol 3-kinase
effect that the hormone has on the cell is to activate a
(PI3K). Some of the effects of leptin occur rapidly as
single type of membrane receptor. The second mes-
a result of activation of these intracellular enzymes,
senger does the rest.
Chapter 74
Introduction to Endocrinology
913
Lipophilic hormone
Extracellular fluid
Diffusion
Nucleus
Target cell
Proteins
DNA
Nuclear
Figure 74-6
receptor
Mechanisms of interaction of
Ribosome
lipophilic hormones, such as
steroids, with intracellular recep-
Cytoplasmic
mRNA
tors in target cells. After the
receptor
Hormone
hormone binds to the receptor in
receptor
mRNA
the cytoplasm or in the nucleus,
Hormone
complex
the hormone-receptor complex
response
binds to the hormone response
element
Nuclear envelope
element (promoter) on the DNA.
This either activates or inhibits
Nuclear pore
gene transcription, formation of
messenger RNA (mRNA), and
protein synthesis.
Table 74-2
Some Hormones That Use the Adenylyl Cyclase-cAMP
Extracellular
Hormone
Second Messenger System
fluid
Adrenocorticotropic hormone (ACTH)
Angiotensin II (epithelial cells)
Calcitonin
Catecholamines (b receptors)
Corticotropin-releasing hormone (CRH)
Cytoplasm
g
Follicle-stimulating hormone (FSH)
Glucagon
b
Human chorionic gonadotropin (HCG)
a
Luteinizing hormone (LH)
Adenylyl
Parathyroid hormone (PTH)
GTP
cyclase
Secretin
Somatostatin
Thyroid-stimulating hormone (TSH)
cAMP
ATP
Vasopressin (V2 receptor, epithelial cells)
Active
Inactive
cAMP-
cAMP-
dependent
dependent
protein
protein
cAMP is not the only second messenger used by the
kinase
kinase
different hormones. Two other especially important
ones are (1) calcium ions and associated calmodulin
Protein - PO4 + ADP
Protein + ATP
and
(2) products of membrane phospholipid
breakdown.
Cell’s response
Adenylyl Cyclase-cAMP Second
Figure 74-7
Messenger System
Table 74-2 shows a few of the many hormones that use
Cyclic adenosine monophosphate (cAMP) mechanism by which
many hormones exert their control of cell function. ADP, adeno-
the adenylyl cyclase-cAMP mechanism to stimulate
sine diphosphate; ATP, adenosine triphosphate.
their target tissues, and Figure 74-7 shows the adeny-
lyl
cyclase-cAMP second messenger system
itself. Binding of the hormones with the receptor
the conversion of a small amount of cytoplasmic
allows coupling of the receptor to a G protein. If the
adenosine triphosphate (ATP) into cAMP inside the
G protein stimulates the adenylyl cyclase-cAMP
cell. This then activates cAMP-dependent protein
system, it is called a Gs protein, denoting a stimulatory
kinase, which phosphorylates specific proteins in the
G protein. Stimulation of adenylyl cyclase, a mem-
cell, triggering biochemical reactions that ultimately
brane-bound enzyme, by the Gs protein then catalyzes
lead to the cell’s response to the hormone.
914
Unit XIV Endocrinology and Reproduction
Once cAMP is formed inside the cell, it usually acti-
reticulum, and the calcium ions then have their own
vates a cascade of enzymes. That is, first one enzyme is
second messenger effects, such as smooth muscle con-
activated, which activates a second enzyme, which acti-
traction and changes in cell secretion.
vates a third, and so forth. The importance of this
DAG, the other lipid second messenger, activates
mechanism is that only a few molecules of activated
the enzyme protein kinase C (PKC), which then phos-
adenylyl cyclase immediately inside the cell mem-
phorylates a large number of proteins, leading to the
brane can cause many more molecules of the next
cell’s response
(Figure 74-8). In addition to these
enzyme to be activated, which can cause still more
effects, the lipid portion of DAG is arachidonic acid,
molecules of the third enzyme to be activated, and
which is the precursor for the prostaglandins and other
so forth. In this way, even the slightest amount of
local hormones that cause multiple effects in tissues
hormone acting on the cell surface can initiate a pow-
throughout the body.
erful cascading activating force for the entire cell.
If binding of the hormone to its receptors is coupled
Calcium-Calmodulin Second
to an inhibitory G protein
(denoted Gi protein),
Messenger System
adenylyl cyclase will be inhibited, reducing the forma-
Another second messenger system operates in
tion of cAMP and ultimately leading to an inhibitory
response to the entry of calcium into the cells. Calcium
action in the cell. Thus, depending on the coupling of
entry may be initiated by (1) changes in membrane
the hormone receptor to an inhibitory or a stimulatory
potential that open calcium channels or (2) a hormone
G protein, a hormone can either increase or decrease
interacting with membrane receptors that open
the concentration of cAMP and phosphorylation of
calcium channels.
key proteins inside the cell.
On entering a cell, calcium ions bind with the
The specific action that occurs in response to
protein calmodulin. This protein has four calcium sites,
increases or decreases of cAMP in each type of target
and when three or four of these sites have bound with
cell depends on the nature of the intracellular machin-
calcium, the calmodulin changes its shape and initiates
ery—some cells have one set of enzymes, and other
multiple effects inside the cell, including activation
cells have other enzymes. Therefore, different func-
or inhibition of protein kinases. Activation
of
tions are elicited in different target cells, such as initi-
ating synthesis of specific intracellular chemicals,
causing muscle contraction or relaxation, initiating
secretion by the cells, and altering cell permeability.
Thus, a thyroid cell stimulated by cAMP forms the
Peptide
metabolic hormones thyroxine and triiodothyronine,
hormone
whereas the same cAMP in an adrenocortical cell
causes secretion of the adrenocortical steroid hor-
Extracellular fluid
mones. In epithelial cells of the renal tubules, cAMP
increases their permeability to water.
Receptor
The Cell Membrane Phospholipid Second
Messenger System
G protein
Some hormones activate transmembrane receptors
that activate the enzyme phospholipase C attached to
Cell
membrane
the inside projections of the receptors (Table 74-3).
This enzyme catalyzes the breakdown of some phos-
Phospholipase C
pholipids in the cell membrane, especially phos-
DAG + IP3
PIP2
phatidylinositol biphosphate (PIP
2), into two different
second messenger products: inositol triphosphate
(IP
3) and diacylglycerol
(DAG). The IP3 mobilizes
Cytoplasm
calcium ions from mitochondria and the endoplasmic
Active
Inactive
protein
protein
kinase C
kinase C
Table 74-3
Some Hormones That Use the Phospholipase C Second
Protein - PO4
Protein
Ca++
Messenger System
Endoplasmic reticulum
Angiotensin II (vascular smooth muscle)
Cell’s response
Cell’s response
Catecholamines (a receptors)
Gonadotropin-releasing hormone (GnRH)
Figure 74-8
Growth hormone-releasing hormone (GHRH)
Oxytocin
The cell membrane phospholipid second messenger system by
Thyroid-releasing hormone (TRH)
which some hormones exert their control of cell function. DAG,
Vasopressin (V1 receptor, vascular smooth muscle)
diacylglycerol; IP3, inositol triphosphate; PIP2, phosphatidylinosi-
tol biphosphate.
Chapter 74
Introduction to Endocrinology
915
calmodulin-dependent protein kinases causes, via
nucleus. To accomplish this, these hormones first bind
phosphorylation, activation or inhibition of proteins
directly with receptor proteins in the nucleus itself;
involved in the cell’s response to the hormone. For
these receptors are probably protein molecules
example, one specific function of calmodulin is to acti-
located within the chromosomal complex, and they
vate myosin kinase, which acts directly on the myosin
likely control the function of the genetic promoters or
of smooth muscle to cause smooth muscle contraction.
operators, as explained in Chapter 3.
The normal calcium ion concentration in most cells
Two important features of thyroid hormone func-
of the body is 10-8 to 10-7 mol/L, which is not enough
tion in the nucleus are the following:
to activate the calmodulin system. But when the
1. They activate the genetic mechanisms for the
calcium ion concentration rises to 10-6 to 10-5 mol/L,
formation of many types of intracellular
enough binding occurs to cause all the intracellular
proteins—probably 100 or more. Many of these
actions of calmodulin. This is almost exactly the same
are enzymes that promote enhanced intracellular
amount of calcium ion change that is required in skele-
metabolic activity in virtually all cells of the body.
tal muscle to activate troponin C, which causes skele-
2. Once bound to the intranuclear receptors, the
tal muscle contraction, as explained in Chapter 7. It is
thyroid hormones can continue to express their
interesting that troponin C is similar to calmodulin in
control functions for days or even weeks.
both function and protein structure.
Measurement of Hormone
Hormones That Act Mainly on the
Concentrations in the Blood
Genetic Machinery of the Cell
Most hormones are present in the blood in extremely
Steroid Hormones Increase Protein Synthesis
minute quantities; some concentrations are as low as
Another means by which hormones act—specifically,
one billionth of a milligram (1 picogram) per milliliter.
the steroid hormones secreted by the adrenal cortex,
Therefore, it was very difficult to measure these con-
ovaries, and testes—is to cause synthesis of proteins in
centrations by the usual chemical means. An extremely
the target cells. These proteins then function as
sensitive method, however, was developed about 40
enzymes, transport proteins, or structural proteins,
years ago that revolutionized the measurement of hor-
which in turn provide other functions of the cells.
mones, their precursors, and their metabolic end prod-
The sequence of events in steroid function is essen-
ucts. This method is called radioimmunoassay.
tially the following:
1. The steroid hormone diffuses across the cell
membrane and enters the cytoplasm of the cell,
Radioimmunoassay
where it binds with a specific receptor protein.
2. The combined receptor protein-hormone then
The method of performing radioimmunoassay is as
diffuses into or is transported into the nucleus.
follows. First, an antibody that is highly specific for the
3. The combination binds at specific points on the
hormone to be measured is produced.
DNA strands in the chromosomes, which activates
Second, a small quantity of this antibody is (1)
the transcription process of specific genes to form
mixed with a quantity of fluid from the animal con-
mRNA.
taining the hormone to be measured and (2) mixed
4. The mRNA diffuses into the cytoplasm, where it
simultaneously with an appropriate amount of purified
promotes the translation process at the ribosomes
standard hormone that has been tagged with a
to form new proteins.
radioactive isotope. However, one specific condition
To give an example, aldosterone, one of the hor-
must be met: There must be too little antibody to bind
mones secreted by the adrenal cortex, enters the cyto-
completely both the radioactively tagged hormone
plasm of renal tubular cells, which contain a specific
and the hormone in the fluid to be assayed. Therefore,
aldosterone receptor protein. Therefore, in these cells,
the natural hormone in the assay fluid and the radio-
the sequence of events cited earlier ensues. After
active standard hormone compete for the binding sites
about 45 minutes, proteins begin to appear in the renal
of the antibody. In the process of competing, the quan-
tubular cells and promote sodium reabsorption from
tity of each of the two hormones, the natural and the
the tubules and potassium secretion into the tubules.
radioactive, that binds is proportional to its concen-
Thus, the full action of the steroid hormone is charac-
tration in the assay fluid.
teristically delayed for at least
45 minutes—up to
Third, after binding has reached equilibrium, the
several hours or even days. This is in marked contrast
antibody-hormone complex is separated from the
to the almost instantaneous action of some of the
remainder of the solution, and the quantity of radio-
peptide and amino acid-derived hormones, such as
active hormone bound in this complex is measured by
vasopressin and norepinephrine.
radioactive counting techniques. If a large amount of
radioactive hormone has bound with the antibody, it is
Thyroid Hormones Increase Gene
clear that there was only a small amount of natural
Transcription in the Cell Nucleus
hormone to compete with the radioactive hormone,
The thyroid hormones thyroxine and triiodothyronine
and therefore the concentration of the natural
cause increased transcription by specific genes in the
hormone in the assayed fluid was small. Conversely, if
916
Unit XIV Endocrinology and Reproduction
100
S
S
P
90
S
S
P
P
P
S
80
S
P
P
70
E
60
AB
3
50
2
AB
40
H
30
20
AB1
10
0
Figure 74-10
2
4
8
16
32
64
128
Aldosterone concentration in
Basic principles of the enzyme-linked immunosorbent assay
test sample (ng/d)
1
(ELISA) for measuring the concentration of a hormone (H). AB
and AB2 are antibodies that recognize the hormone at different
binding sites, and AB3 is an antibody that recognizes AB2. E is an
enzyme linked to AB3 that catalyzes the formation of a colored
Figure 74-9
fluorescent product (P) from a substrate (S). The amount of the
product is measured using optical methods and is proportional to
“Standard curve” for radioimmunoassay of aldosterone. (Courtesy
the amount of hormone in the well if there are excess antibodies
Dr. Manis Smith.)
in the well.
detected by colorimetric or fluorescent optical
only a small amount of radioactive hormone has
methods.
bound, it is clear that there was a large amount of
Because each molecule of enzyme catalyzes the
natural hormone to compete for the binding sites.
formation of many thousands of product molecules,
Fourth, to make the assay highly quantitative, the
even very small amounts of hormone molecules can be
radioimmunoassay procedure is also performed for
detected. In contrast to competitive radioimmuno-
“standard” solutions of untagged hormone at several
assay methods, ELISA methods use excess antib-
concentration levels. Then a
“standard curve” is
odies so that all hormone molecules are captured in
plotted, as shown in Figure 74-9. By comparing the
antibody-hormone complexes. Therefore, the amount
radioactive counts recorded from the
“unknown”
of hormone present in the sample or in the standard
assay procedures with the standard curve, one can
is proportional to the amount of product formed.
determine within an error of 10 to 15 per cent the con-
The ELISA method has become widely used in clin-
centration of the hormone in the “unknown” assayed
ical laboratories because
(1) it does not employ
fluid. As little as billionths or even trillionths of a gram
radioactive isotopes, (2) much of the assay can be auto-
of hormone can often be assayed in this way.
mated using 96-well plates, and (3) it has proved to be
a cost-effective and accurate method for assessing
hormone levels.
Enzyme-Linked Immunosorbent
Assay (ELISA)
References
Enzyme-linked immunosorbent assays (ELISAs) can
be used to measure almost any protein, including hor-
Alberts B, Johnson A, Lewis J, et al: Molecular Biology of
mones. This test combines the specificity of antibodies
the Cell. New York: Garland Science, 2002.
with the sensitivity of simple enzyme assays. Figure
Aranda A, Pascual A: Nuclear hormone receptors and gene
74-10 shows the basic elements of this method, which
expression. Physiol Rev 81:1269, 2001.
Clark AJ, Baig AH, Noon L, et al: Expression, desensitiza-
is often performed on plastic plates that each have 96
tion, and internalization of the ACTH receptor (MC2R).
small wells. Each well is coated with an antibody (AB1)
Ann N Y Acad Sci 994:111, 2003.
that is specific for the hormone being assayed. Samples
Katzmann DJ, Odorizzi G, Emr SD: Receptor downregula-
or standards are added to each of the wells, followed
tion and multivesicular-body sorting. Nat Rev Mol Cell
by a second antibody (AB2) that is also specific for the
Biol 3:893, 2002.
hormone but binds to a different site of the hormone
Kelly MJ, Qiu J, Ronnekleiv OK: Estrogen modulation of G-
molecule. A third antibody (AB3) is added that recog-
protein-coupled receptor activation of potassium channels
nizes AB2 and is coupled to an enzyme that converts
in the central nervous system. Ann N Y Acad Sci 1007:6,
a suitable substrate to a product that can be easily
2003.
Chapter 74
Introduction to Endocrinology
917
Kuhn M: Structure, regulation, and function of mammalian
Pires-daSilva A, Sommer RJ: The evolution of signaling
membrane guanylyl cyclase receptors, with a focus on
pathways in animal development. Nat Rev Genet 4:39,
guanylyl cyclase-A. Circ Res 93:700, 2003.
2003.
Larsen PR, Kronenberg HM, Melmed S, Polonsky KS:
Privalsky ML: The role of corepressors in transcriptional
Williams Textbook of Endocrinolog, 10th ed. Philadelphia:
regulation by nuclear hormone receptors. Annu Rev
WB Saunders, 2003.
Physiol 66:315, 2004.
Lösel RM, Falkenstein E, Feuring M, et al: Nongenomic
Spat A, Hunyady L: Control of aldosterone secretion: a
steroid action: controversies, questions, and answers.
model for convergence in cellular signaling pathways.
Physiol Rev 83:965, 2003.
Physiol Rev 84:489, 2004.
Morris AJ, Malbon CC: Physiological regulation of G
Spiegel AM, Weinstein LS: Inherited diseases involving G
protein-linked signaling. Physiol Rev 79:1373, 1999.
proteins and G protein-coupled receptors. Annu Rev Med
Nadal A, Diaz M, Valverde MA: The estrogen trinity: mem-
55:27, 2004.
brane, cytosolic, and nuclear effects. News Physiol Sci
Tasken K, Aandahl EM: Localized effects of cAMP medi-
16:251, 2001.
ated by distinct routes of protein kinase A. Physiol Rev
Nagaich AK, Rayasam GV, Martinez ED, et al: Subnuclear
84:137, 2004.
trafficking and gene targeting by steroid receptors. Ann
Vasudevan N, Ogawa S, Pfaff D: Estrogen and thyroid
N Y Acad Sci 1024:213, 2004.
hormone receptor interactions: physiological flexibility by
Olson TS, Ley K: Chemokines and chemokine receptors in
molecular specificity. Physiol Rev 82:923, 2002.
leukocyte trafficking. Am J Physiol Regul Integr Comp
Yen PM: Physiological and molecular basis of thyroid
Physiol 283:R7, 2002.
hormone action. Physiol Rev 81:1097, 2001.
O’Shea JJ, Pesu M, Borie DC, Changelian PS: A new modal-
ity for immunosuppression: targeting the JAK/STAT
pathway. Nat Rev Drug Discov 3:555, 2004.
C H A P T E R
7
5
Pituitary Hormones and Their
Control by the Hypothalamus
Pituitary Gland and
Its Relation to the
Hypothalamus
Pituitary Gland: Two Distinct Parts-The Anterior and Poste-
rior Lobes. The pituitary gland (Figure 75-1), also
called the hypophysis, is a small gland—about 1 cen-
timeter in diameter and 0.5 to 1 gram in weight—
that lies in the sella turcica, a bony cavity at the base of the brain, and is
connected to the hypothalamus by the pituitary (or hypophysial) stalk. Physio-
logically, the pituitary gland is divisible into two distinct portions: the anterior
pituitary, also known as the adenohypophysis, and the posterior pituitary, also
known as the neurohypophysis. Between these is a small, relatively avascular
zone called the pars intermedia, which is almost absent in the human being but
is much larger and much more functional in some lower animals.
Embryologically, the two portions of the pituitary originate from different
sources—the anterior pituitary from Rathke’s pouch, which is an embryonic
invagination of the pharyngeal epithelium, and the posterior pituitary from a
neural tissue outgrowth from the hypothalamus. The origin of the anterior pitu-
itary from the pharyngeal epithelium explains the epithelioid nature of its cells,
and the origin of the posterior pituitary from neural tissue explains the pres-
ence of large numbers of glial-type cells in this gland.
Six important peptide hormones plus several less important ones are secreted
by the anterior pituitary, and two important peptide hormones are secreted
by the posterior pituitary. The hormones of the anterior pituitary play major
roles in the control of metabolic functions throughout the body, as shown in
Figure 75-2.
• Growth hormone promotes growth of the entire body by affecting protein
formation, cell multiplication, and cell differentiation.
• Adrenocorticotropin (corticotropin) controls the secretion of some of the
adrenocortical hormones, which affect the metabolism of glucose, proteins,
and fats.
• Thyroid-stimulating hormone (thyrotropin) controls the rate of secretion of
thyroxine and triiodothyronine by the thyroid gland, and these hormones
control the rates of most intracellular chemical reactions in the body.
• Prolactin promotes mammary gland development and milk production.
• Two separate gonadotropic hormones, follicle-stimulating hormone and
luteinizing hormone, control growth of the ovaries and testes, as well as
their hormonal and reproductive activities.
The two hormones secreted by the posterior pituitary play other roles.
• Antidiuretic hormone (also called vasopressin) controls the rate of water
excretion into the urine, thus helping to control the concentration of water
in the body fluids.
• Oxytocin helps express milk from the glands of the breast to the nipples
during suckling and possibly helps in the delivery of the baby at the end of
gestation.
Anterior Pituitary Gland Contains Several Different Cell Types That Synthesize and Secrete
Hormones. Usually, there is one cell type for each major hormone formed in the
918
Chapter 75
Pituitary Hormones and Their Control by the Hypothalamus
919
Hypothalamus
Gamma
Alpha
Epsilon (e)
Delta (d)
(g) cell
Sinusoid
(a) cell acidophil cell basophil cell
Hypophysial stalk
Posterior pituitary
Anterior pituitary
Pars intermedia
Beta (b) cell
Figure 75-1
Figure 75-3
Pituitary gland.
Cellular structure of the anterior pituitary gland. (Redrawn from
Guyton AC: Physiology of the Human Body, 6th ed. Philadelphia:
Saunders College Publishing, 1984.)
3. Thyrotropes—thyroid-stimulating hormone (TSH)
Thyroid
4. Gonadotropes—gonadotropic hormones, which
gland
include both luteinizing hormone (LH) and follicle-
stimulating hormone (FSH)
Growth
5. Lactotropes—prolactin (PRL)
About 30 to 40 per cent of the anterior pituitary cells
are somatotropes that secrete growth hormone, and
Increases blood
about 20 per cent are corticotropes that secrete ACTH.
glucose level
ACH
Each of the other cell types accounts for only 3 to 5 per
cent of the total; nevertheless, they secrete powerful
hormones for controlling thyroid function, sexual func-
Corticotropin
Promotes secretion
tions, and milk secretion by the breasts.
Anterior
of insulin
Somatotropes stain strongly with acid dyes and are
pituitary
Adrenal cortex
therefore called acidophils. Thus, pituitary tumors that
gland
Follicle
secrete large quantities of human growth hormone are
stimulating
called acidophilic tumors.
Pancreas
Luteinizing
Posterior Pituitary Hormones Are Synthesized by Cell Bodies in
Ovary
the Hypothalamus. The bodies of the cells that secrete
the posterior pituitary hormones are not located in the
Prolactin
pituitary gland itself but are large neurons, called mag-
Mammary
nocellular neurons, located in the supraoptic and par-
gland
aventricular nuclei of the hypothalamus. The hormones
are then transported in the axoplasm of the neurons’
nerve fibers passing from the hypothalamus to the pos-
Figure 75-2
terior pituitary gland. This is discussed more fully later
in the chapter.
Metabolic functions of the anterior pituitary hormones. ACH,
adrenal corticosteroid hormones.
Hypothalamus Controls
anterior pituitary gland. With special stains attached to
Pituitary Secretion
high-affinity antibodies that bind with the distinctive
hormones, at least five cell types can be differentiated
Almost all secretion by the pituitary is controlled by
(Figure 75-3). Table 75-1 provides a summary of these
either hormonal or nervous signals from the hypo-
cell types, the hormones they produce, and their physio-
logical actions. These five cell types are:
thalamus. Indeed, when the pituitary gland is removed
1. Somatotropes—human growth hormone (hGH)
from its normal position beneath the hypothalamus
2. Corticotropes—adrenocorticotropin (ACTH)
and transplanted to some other part of the body, its
920
Unit XIV Endocrinology and Reproduction
Table 75-1
Cells and Hormones of the Anterior Pituitary Gland and Their Physiological Functions
Cell
Hormone
Chemistry
Physiological Actions
Somatotropes
Growth hormone (GH;
Single chain of 191 amino acids
Stimulates body growth; stimulates
somatotropin)
secretion of IGF-1; stimulates lipolysis;
inhibits actions of insulin on
carbohydrate and lipid metabolism
Corticotropes
Adrenocorticotropic hormone
Single chain of 39 amino acids
Stimulates production of glucocorticoids
(ACTH; corticotropin)
and androgens by the adrenal cortex;
maintains size of zona fasciculata and
zona reticularis of cortex
Thyrotropes
Thyroid-stimulating hormone
Glycoprotein of two subunits,
Stimulates production of thyroid
(TSH; thyrotropin)
a (89 amino acids) and
hormones by thyroid follicular cells;
b (112 amino acids)
maintains size of follicular cells
Gonadotropes
Follicle-stimulating hormone
Glycoprotein of two subunits,
Stimulates development of ovarian
(FSH)
a (89 amino acids) and
follicles; regulates spermatogenesis in
b (112 amino acids)
the testis
Luteinizing hormone (LH)
Glycoprotein of two subunits,
Causes ovulation and formation of the
a (89 amino acids) and
corpus luteum in the ovary; stimulates
b (115 amino acids)
production of estrogen and progesterone
by the ovary; stimulates testosterone
production by the testis
Lactotropes,
Prolactin (PRL)
Single chain of 198 amino
Stimulates milk secretion and production
Mammotropes
acids
IGF, insulin-like
growth factor
rates of secretion of the different hormones (except
Hypothalamus
for prolactin) fall to very low levels.
Secretion from the posterior pituitary is controlled
by nerve signals that originate in the hypothalamus
and terminate in the posterior pituitary. In contrast,
secretion by the anterior pituitary is controlled by hor-
Optic chiasm
mones called hypothalamic releasing and hypothala-
Mamillary body
Artery
mic inhibitory hormones (or factors) secreted within
Primary capillary
the hypothalamus itself and then conducted, as shown
Median eminence
plexus
in Figure
75-4, to the anterior pituitary through
Hypothalamic-
minute blood vessels called hypothalamic-hypophysial
hypophysial
portal vessels. In the anterior pituitary, these releasing
portal vessels
and inhibitory hormones act on the glandular cells to
Posterior pituitary
Sinuses
control their secretion. This system of control is dis-
gland
cussed in the next section of this chapter.
Anterior pituitary
gland
The hypothalamus receives signals from many
Vein
sources in the nervous system. Thus, when a person is
exposed to pain, a portion of the pain signal is trans-
Figure 75-4
mitted into the hypothalamus. Likewise, when a
person experiences some powerful depressing or excit-
Hypothalamic-hypophysial portal system.
ing thought, a portion of the signal is transmitted into
the hypothalamus. Olfactory stimuli denoting pleasant
or unpleasant smells transmit strong signal compo-
Hypothalamic-Hypophysial Portal
nents directly and through the amygdaloid nuclei into
Blood Vessels of the Anterior
the hypothalamus. Even the concentrations of nutri-
ents, electrolytes, water, and various hormones in the
Pituitary Gland
blood excite or inhibit various portions of the hypo-
thalamus. Thus, the hypothalamus is a collecting center
The anterior pituitary is a highly vascular gland with
for information concerning the internal well-being
extensive capillary sinuses among the glandular cells.
of the body, and much of this information is used
Almost all the blood that enters these sinuses passes
to control secretions of the many globally important
first through another capillary bed in the lower
pituitary hormones.
hypothalamus. The blood then flows through small
Chapter 75
Pituitary Hormones and Their Control by the Hypothalamus
921
hypothalamic-hypophysial portal blood vessels into the
called somatostatin, which inhibits release of
anterior pituitary sinuses. Figure 75-4 shows the low-
growth hormone
ermost portion of the hypothalamus, called the median
4. Gonadotropin-releasing hormone (GnRH), which
eminence, which connects inferiorly with the pituitary
causes release of the two gonadotropic hormones,
stalk. Small arteries penetrate into the substance
luteinizing hormone and follicle-stimulating
of the median eminence and then additional small
hormone
vessels return to its surface, coalescing to form the
5. Prolactin inhibitory hormone (PIH), which causes
hypothalamic-hypophysial portal blood vessels. These
inhibition of prolactin secretion
pass downward along the pituitary stalk to supply
There are some additional hypothalamic hormones,
blood to the anterior pituitary sinuses.
including one that stimulates prolactin secretion and
perhaps others that inhibit release of the anterior pitu-
Hypothalamic Releasing and Inhibitory Hormones Are Secreted
itary hormones. Each of the more important hypo-
into the Median Eminence. Special neurons in the hypo-
thalamic hormones is discussed in detail as the specific
thalamus synthesize and secrete the hypothalamic
hormonal systems controlled by them are presented in
releasing and inhibitory hormones that control secre-
this and subsequent chapters.
tion of the anterior pituitary hormones. These neurons
originate in various parts of the hypothalamus and
Specific Areas in the Hypothalamus Control Secretion of Spe-
send their nerve fibers to the median eminence and
cific Hypothalamic Releasing and Inhibitory Hormones. All or
tuber cinereum, an extension of hypothalamic tissue
most of the hypothalamic hormones are secreted at
into the pituitary stalk.
nerve endings in the median eminence before being
The endings of these fibers are different from most
transported to the anterior pituitary gland. Electrical
endings in the central nervous system, in that their
stimulation of this region excites these nerve endings
function is not to transmit signals from one neuron to
and, therefore, causes release of essentially all the
another but rather to secrete the hypothalamic releas-
hypothalamic hormones. However, the neuronal cell
ing and inhibitory hormones into the tissue fluids.
bodies that give rise to these median eminence nerve
These hormones are immediately absorbed into the
endings are located in other discrete areas of the hypo-
hypothalamic-hypophysial portal system and carried
thalamus or in closely related areas of the basal brain.
directly to the sinuses of the anterior pituitary gland.
The specific loci of the neuronal cell bodies that form
the different hypothalamic releasing or inhibitory
Hypothalamic Releasing and Inhibitory Hormones Control Ante-
hormones are still poorly known, so that it would be
rior Pituitary Secretion. The function of the releasing and
misleading to attempt delineation here.
inhibitory hormones is to control secretion of the ante-
rior pituitary hormones. For most of the anterior pitu-
itary hormones, it is the releasing hormones that are
Physiological Functions
important, but for prolactin, a hypothalamic inhibitory
of Growth Hormone
hormone probably exerts more control. The major
hypothalamic releasing and inhibitory hormones are
All the major anterior pituitary hormones, except for
summarized in Table 75-2 and are the following:
growth hormone, exert their principal effects by stim-
1. Thyrotropin-releasing hormone (TRH), which
ulating target glands, including thyroid gland, adrenal
causes release of thyroid-stimulating hormone
cortex, ovaries, testicles, and mammary glands. The
2. Corticotropin-releasing hormone (CRH), which
functions of each of these pituitary hormones are so
causes release of adrenocorticotropin
intimately concerned with the functions of the respec-
3. Growth hormone-releasing hormone (GHRH),
tive target glands that, except for growth hormone,
which causes release of growth hormone, and
their functions are discussed in subsequent chapters
growth hormone inhibitory hormone (GHIH), also
along with the target glands. Growth hormone, in
Table 75-2
Hypothalamic Releasing and Inhibitory Hormones That Control Secretion of the Anterior Pituitary Gland
Hormone
Structure
Primary Action on Anterior Pituitary
Thyrotropin-releasing hormone (TRH)
Peptide of 3 amino acids
Stimulates secretion of TSH by thyrotropes
Gonadotropin-releasing hormone (GnRH)
Single chain of 10 amino acids
Stimulates secretion of FSH and LH by gonadotropes
Corticotropin-releasing hormone (CRH)
Single chain of 41 amino acids
Stimulates secretion of ACTH by corticotropes
Growth hormone-releasing hormone
Single chain of 44 amino acids
Stimulates secretion of growth hormone by somatotropes
(GHRH)
Growth hormone inhibitory hormone
Single chain of 14 amino acids
Inhibits secretion of growth hormone by somatotropes
(somatostatin)
Prolactin-inhibiting hormone (PIH)
Dopamine (a catecholamine)
Inhibits secretion of prolactin by lactotropes
ACTH, adrenocorticotropic hormone; FSH, follicle-stimulating hormone; LH, luteinizing hormone; TSH, thyroid-stimulating hormone.
922
Unit XIV Endocrinology and Reproduction
contrast to other hormones, does not function through
including (1) increased rate of protein synthesis in
a target gland but exerts its effects directly on all or
most cells of the body; (2) increased mobilization of
almost all tissues of the body.
fatty acids from adipose tissue, increased free fatty
acids in the blood, and increased use of fatty acids for
energy; and (3) decreased rate of glucose utilization
Growth Hormone Promotes Growth
throughout the body. Thus, in effect, growth hormone
of Many Body Tissues
enhances body protein, uses up fat stores, and con-
serves carbohydrates.
Growth hormone, also called somatotropic hormone
or somatotropin, is a small protein molecule that con-
Growth Hormone Promotes Protein Deposition
tains
191 amino acids in a single chain and has a
in Tissues
molecular weight of 22,005. It causes growth of almost
Although the precise mechanisms by which growth
all tissues of the body that are capable of growing. It
hormone increases protein deposition are not known,
promotes increased sizes of the cells and increased
a series of different effects are known, all of which
mitosis, with development of greater numbers of cells
could lead to enhanced protein deposition.
and specific differentiation of certain types of cells
such as bone growth cells and early muscle cells.
Enhancement of Amino Acid Transport Through the Cell Mem-
Figure 75-5 shows typical weight charts of two
branes. Growth hormone directly enhances transport
growing littermate rats, one of which received daily
of at least some and perhaps most amino acids through
injections of growth hormone and the other of which
the cell membranes to the interior of the cells. This
did not receive growth hormone. This figure shows
increases the amino acid concentrations in the cells
marked enhancement of growth in the rat given
and is presumed to be at least partly responsible for
growth hormone—in the early days of life and even
the increased protein synthesis. This control of amino
after the two rats reached adulthood. In the early
acid transport is similar to the effect of insulin in con-
stages of development, all organs of the treated rat
trolling glucose transport through the membrane, as
increased proportionately in size; after adulthood was
discussed in Chapters 67 and 78.
reached, most of the bones stopped lengthening, but
many of the soft tissues continued to grow. This results
Enhancement of RNA Translation to Cause Protein Synthesis by
from the fact that once the epiphyses of the long bones
the Ribosomes. Even when the amino acid concentra-
have united with the shafts, further lengthening of
tions are not increased in the cells, growth hormone
bone cannot occur, even though most other tissues of
still increases RNA translation, causing protein to be
the body can continue to grow throughout life.
synthesized in greater amounts by the ribosomes in the
cytoplasm.
Growth Hormone Has Several
Increased Nuclear Transcription of DNA to Form RNA. Over
Metabolic Effects
more prolonged periods (24 to 48 hours), growth
hormone also stimulates the transcription of DNA in
Aside from its general effect in causing growth, growth
the nucleus, causing the formation of increased quan-
hormone has multiple specific metabolic effects,
tities of RNA. This promotes more protein synthesis
and promotes growth if sufficient energy, amino acids,
vitamins, and other requisites for growth are available.
In the long run, this may be the most important func-
500
tion of growth hormone.
Injected daily with
growth hormone
Decreased Catabolism of Protein and Amino Acids. In addi-
400
tion to the increase in protein synthesis, there is a
decrease in the breakdown of cell protein. A probable
300
reason for this is that growth hormone also mobilizes
Control
large quantities of free fatty acids from the adipose
200
tissue, and these are used to supply most of the energy
for the body’s cells, thus acting as a potent “protein
200
sparer.”
Summary. Growth hormone enhances almost all facets
0
0
100
200
300
400
500
600
of amino acid uptake and protein synthesis by cells,
Days
while at the same time reducing the breakdown of
proteins.
Growth Hormone Enhances Fat Utilization
Figure 75-5
for Energy
Comparison of weight gain of a rat injected daily with growth
Growth hormone has a specific effect in causing
hormone with that of a normal littermate.
the release of fatty acids from adipose tissue and,
Chapter 75
Pituitary Hormones and Their Control by the Hypothalamus
923
therefore, increasing the concentration of fatty acids in
tive. Part of this requirement for carbohydrates and
the body fluids. In addition, in tissues throughout the
insulin is to provide the energy needed for the metab-
body, growth hormone enhances the conversion of
olism of growth, but there seem to be other effects as
fatty acids to acetyl coenzyme A (acetyl-CoA) and its
well. Especially important is insulin’s ability to
subsequent utilization for energy. Therefore, under
enhance the transport of some amino acids into cells,
the influence of growth hormone, fat is used for
in the same way that it stimulates glucose transport.
energy in preference to the use of carbohydrates and
proteins.
Growth hormone’s ability to promote fat utilization,
Growth Hormone Stimulates Cartilage
together with its protein anabolic effect, causes an
and Bone Growth
increase in lean body mass. However, mobilization of
fat by growth hormone requires several hours to occur,
Although growth hormone stimulates increased de-
whereas enhancement of protein synthesis can begin
position of protein and increased growth in almost
in minutes under the influence of growth hormone.
all tissues of the body, its most obvious effect is to
increase growth of the skeletal frame. This results from
“Ketogenic” Effect of Growth Hormone. Under the influ-
multiple effects of growth hormone on bone, including
ence of excessive amounts of growth hormone, fat
(1) increased deposition of protein by the chondro-
mobilization from adipose tissue sometimes becomes
cytic and osteogenic cells that cause bone growth,
so great that large quantities of acetoacetic acid are
(2) increased rate of reproduction of these cells,
formed by the liver and released into the body fluids,
and (3) a specific effect of converting chondrocytes
thus causing ketosis. This excessive mobilization of fat
into osteogenic cells, thus causing deposition of new
from the adipose tissue also frequently causes a fatty
bone.
liver.
There are two principal mechanisms of bone
growth. First, in response to growth hormone stimula-
Growth Hormone Decreases
tion, the long bones grow in length at the epiphyseal
Carbohydrate Utilization
cartilages, where the epiphyses at the ends of the bone
Growth hormone causes multiple effects that
are separated from the shaft. This growth first causes
influence carbohydrate metabolism, including
(1)
deposition of new cartilage, followed by its conversion
decreased glucose uptake in tissues such as skeletal
into new bone, thus elongating the shaft and pushing
muscle and fat, (2) increased glucose production by
the epiphyses farther and farther apart. At the same
the liver, and (3) increased insulin secretion.
time, the epiphyseal cartilage itself is progressively
Each of these changes results from growth
used up, so that by late adolescence, no additional epi-
hormone-induced “insulin resistance,” which attenu-
physeal cartilage remains to provide for further long
ates insulin’s actions to stimulate the uptake and uti-
bone growth. At this time, bony fusion occurs between
lization of glucose in skeletal muscle and fat and to
the shaft and the epiphysis at each end, so that no
inhibit gluconeogenesis (glucose production) by the
further lengthening of the long bone can occur.
liver; this leads to increased blood glucose concentra-
Second, osteoblasts in the bone periosteum and in
tion and a compensatory increase in insulin secretion.
some bone cavities deposit new bone on the surfaces
For these reasons, growth hormone’s effects are called
of older bone. Simultaneously, osteoclasts in the bone
diabetogenic, and excess secretion of growth hormone
(discussed in detail in Chapter 79) remove old bone.
can produce metabolic disturbances very similar to
When the rate of deposition is greater than that of
those found in patients with type II
(non-insulin-
resorption, the thickness of the bone increases. Growth
dependent) diabetes, who are also very resistant to the
hormone strongly stimulates osteoblasts. Therefore, the
metabolic effects of insulin.
bones can continue to become thicker throughout life
We do not know the precise mechanism by which
under the influence of growth hormone; this is espe-
growth hormone causes insulin resistance and
cially true for the membranous bones. For instance, the
decreased glucose utilization by the cells. However,
jaw bones can be stimulated to grow even after ado-
growth hormone-induced increases in blood concen-
lescence, causing forward protrusion of the chin and
trations of fatty acids may impair insulin’s actions on
lower teeth. Likewise, the bones of the skull can grow
tissue glucose utilization. Experimental studies indi-
in thickness and give rise to bony protrusions over the
cate that raising blood levels of fatty acids above
eyes.
normal rapidly decreases the sensitivity of the liver
and skeletal muscle to insulin’s effects on carbohy-
drate metabolism.
Growth Hormone Exerts Much
of Its Effect Through Intermediate
Necessity of Insulin and Carbohydrate for the Growth-
Substances Called “Somatomedins”
Promoting Action of Growth Hormone. Growth hormone
fails to cause growth in an animal that lacks a pan-
(Also Called “Insulin-Like
creas; it also fails to cause growth if carbohydrates are
Growth Factors”)
excluded from the diet. This shows that adequate
insulin activity and adequate availability of carbohy-
When growth hormone is supplied directly to cartilage
drates are necessary for growth hormone to be effec-
chondrocytes cultured outside the body, proliferation
924
Unit XIV Endocrinology and Reproduction
or enlargement of the chondrocytes usually fails to
occur. Yet growth hormone injected into the intact
animal does cause proliferation and growth of the
Sleep
30
same cells.
In brief, it has been found that growth hormone
Strenuous
causes the liver
(and, to a much less extent,
exercise
20
other tissues) to form several small proteins
called somatomedins that have the potent effect of
increasing all aspects of bone growth. Many of the
10
somatomedin effects on growth are similar to the
effects of insulin on growth. Therefore, the somato-
medins are also called insulin-like growth factors
0
(IGFs).
8 am
12
4 am
8 pm
12
4 am
8 am
At least four somatomedins have been isolated,
Noon
Midnight
but by far the most important of these is somatomedin
C (also called IGF-I). The molecular weight of
somatomedin C is about 7500, and its concentration in
the plasma closely follows the rate of growth hormone
Figure 75-6
secretion.
Typical variations in growth hormone secretion throughout the day,
The pygmies of Africa have a congenital inability to
demonstrating the especially powerful effect of strenuous exercise
synthesize significant amounts of somatomedin C.
and also the high rate of growth hormone secretion that occurs
Therefore, even though their plasma concentration of
during the first few hours of deep sleep.
growth hormone is either normal or high, they have
diminished amounts of somatomedin C in the plasma;
this apparently accounts for the small stature of these
Regulation of Growth
people. Some other dwarfs
(e.g., the Lévi-Lorain
Hormone Secretion
dwarf) also have this problem.
It has been postulated that most, if not all, of the
For many years it was believed that growth hormone
growth effects of growth hormone result from
was secreted primarily during the period of growth but
somatomedin C and other somatomedins, rather
then disappeared from the blood at adolescence. This
than from direct effects of growth hormone on the
has proved to be untrue. After adolescence, secretion
bones and other peripheral tissues. Even so, experi-
decreases slowly with aging, finally falling to about 25
ments have demonstrated that injection of growth
per cent of the adolescent level in very old age.
hormone directly into the epiphyseal cartilages of
Growth hormone is secreted in a pulsatile pattern,
bones of living animals causes the specific growth
increasing and decreasing. The precise mechanisms
of these cartilage areas, and the amount of growth
that control secretion of growth hormone are not fully
hormone required for this is minute. Some aspects
understood, but several factors related to a person’s
of the somatomedin hypothesis are still questionable.
state of nutrition or stress are known to stimulate
One possibility is that growth hormone can cause
secretion: (1) starvation, especially with severe protein
the formation of enough somatomedin C in the
deficiency; (2) hypoglycemia or low concentration of
local tissue to cause local growth. It is also possible
fatty acids in the blood; (3) exercise; (4) excitement; and
that growth hormone itself is directly responsible
(5) trauma. Growth hormone also characteristically
for increased growth in some tissues and that the
increases during the first 2 hours of deep sleep, as
somatomedin mechanism is an alternative means of
shown in Figure 75-6. Table 75-3 summarizes some of
increasing growth but not always a necessary
the factors that are known to influence growth
one.
hormone secretion.
The normal concentration of growth hormone in the
plasma of an adult is between 1.6 and 3 ng/ml; in a
Short Duration of Action of Growth Hormone but Prolonged
child or adolescent, it is about 6 ng/ml. These values
Action of Somatomedin C. Growth hormone attaches only
often increase to as high as 50 ng/ml after depletion of
weakly to the plasma proteins in the blood. Therefore,
the body stores of proteins or carbohydrates during
it is released from the blood into the tissues rapidly,
prolonged starvation.
having a half-time in the blood of less than 20 minutes.
Under acute conditions, hypoglycemia is a far more
By contrast, somatomedin C attaches strongly to a
potent stimulator of growth hormone secretion than is
carrier protein in the blood that, like somatomedin C,
an acute decrease in protein intake. Conversely, in
is produced in response to growth hormone. As a
chronic conditions, growth hormone secretion seems
result, somatomedin C is released only slowly from the
to correlate more with the degree of cellular protein
blood to the tissues, with a half-time of about 20 hours.
depletion than with the degree of glucose insufficiency.
This greatly prolongs the growth-promoting effects of
For instance, the extremely high levels of growth
the bursts of growth hormone secretion shown in
hormone that occur during starvation are closely
Figure 75-6.
related to the amount of protein depletion.
Chapter 75
Pituitary Hormones and Their Control by the Hypothalamus
925
hormone concentration. The third and fourth columns
Table 75-3
show the levels after treatment with protein supple-
Factors That Stimulate or Inhibit Secretion of
ments for 3 and 25 days, respectively, with a concomi-
Growth Hormone
tant decrease in the hormone.
These results demonstrate that under severe condi-
Stimulate Growth Hormone
Inhibit Growth Hormone
tions of protein malnutrition, adequate calories alone
Secretion
Secretion
are not sufficient to correct the excess production of
Decreased blood glucose
Increased blood glucose
growth hormone. The protein deficiency must also be
Decreased blood free fatty
Increased blood free fatty
corrected before the growth hormone concentration
acids
acids
will return to normal.
Starvation or fasting, protein
Aging
deficiency
Obesity
Trauma, stress, excitement
Growth hormone inhibitory
Role of the Hypothalamus, Growth
Exercise
hormone (somatostatin)
Hormone-Releasing Hormone, and
Testosterone, estrogen
Growth hormone (exogenous)
Somatostatin in the Control of Growth
Deep sleep ( stages II and IV)
Somatomedins (insulin-like
Hormone Secretion
Growth hormone-releasing
growth factors)
From the preceding description of the many factors
hormone
that can affect growth hormone secretion, one can
readily understand the perplexity of physiologists as
they attempt to unravel the mysteries of regulation of
growth hormone secretion. It is known that growth
hormone secretion is controlled by two factors
secreted in the hypothalamus and then transported to
40
the anterior pituitary gland through the hypothalamic-
hypophysial portal vessels. They are growth
hormone-releasing hormone and growth hormone
30
inhibitory hormone (also called somatostatin). Both of
these are polypeptides; GHRH is composed of 44
amino acids, and somatostatin is composed of
14
amino acids.
20
The part of the hypothalamus that causes secretion
of GHRH is the ventromedial nucleus; this is the
same area of the hypothalamus that is sensitive to
10
blood glucose concentration, causing satiety in hyper-
glycemic states and hunger in hypoglycemic states. The
secretion of somatostatin is controlled by other nearby
0
areas of the hypothalamus. Therefore, it is reasonable
Protein
Carbohydrate
Protein
Protein
to believe that some of the same signals that modify a
deficiency
treatment
treatment
treatment
(kwashiorkor)
(3 days)
(3 days)
(25 days)
person’s behavioral feeding instincts also alter the rate
of growth hormone secretion.
In a similar manner, hypothalamic signals depicting
emotions, stress, and trauma can all affect hypothala-
Figure 75-7
mic control of growth hormone secretion. In fact,
experiments have shown that catecholamines,
Effect of extreme protein deficiency on the plasma concentration
of growth hormone in the disease kwashiorkor. Also shown is the
dopamine, and serotonin, each of which is released by
failure of carbohydrate treatment but the effectiveness of protein
a different neuronal system in the hypothalamus, all
treatment in lowering growth hormone concentration. (Drawn from
increase the rate of growth hormone secretion.
data in Pimstone BL, Barbezat G, Hansen JD, Murray P: Studies
Most of the control of growth hormone secretion
on growth hormone secretion in protein-calorie malnutrition. Am J
Clin Nutr 21:482, 1968.)
is probably mediated through GHRH rather than
through the inhibitory hormone somatostatin. GHRH
stimulates growth hormone secretion by attaching to
specific cell membrane receptors on the outer surfaces
Figure 75-7 demonstrates the effect of protein defi-
of the growth hormone cells in the pituitary gland. The
ciency on plasma growth hormone and then the effect
receptors activate the adenylyl cyclase system inside
of adding protein to the diet. The first column shows
the cell membrane, increasing the intracellular level of
very high levels of growth hormone in children with
cyclic adenosine monophosphate (cAMP). This has
extreme protein deficiency during the protein malnu-
both a short-term and a long-term effect. The short-
trition condition called kwashiorkor; the second
term effect is to increase calcium ion transport into the
column shows the levels in the same children after 3
cell; within minutes, this causes fusion of the growth
days of treatment with more than adequate quantities
hormone secretory vesicles with the cell membrane
of carbohydrates in their diets, demonstrating that
and release of the hormone into the blood. The long-
the carbohydrates did not lower the plasma growth
term effect is to increase transcription in the nucleus
926
Unit XIV Endocrinology and Reproduction
by the genes to stimulate the synthesis of new growth
only in the one species or, at most, closely related
hormone.
species. For this reason, growth hormone prepared from
When growth hormone is administered directly into
lower animals (except, to some extent, from primates)
is not effective in human beings. Therefore, the growth
the blood of an animal over a period of hours, the rate
hormone of the human being is called human growth
of endogenous growth hormone secretion decreases.
hormone to distinguish it from the others.
This demonstrates that growth hormone secretion is
In the past, because growth hormone had to be pre-
subject to typical negative feedback control, as is true
pared from human pituitary glands, it was difficult to
for essentially all hormones. The nature of this feed-
obtain sufficient quantities to treat patients with growth
back mechanism and whether it is mediated mainly
hormone deficiency, except on an experimental basis.
through inhibition of GHRH or enhancement of
However, human growth hormone can now be synthe-
somatostatin, which inhibits growth hormone secre-
sized by Escherichia coli bacteria as a result of success-
tion, are uncertain.
ful application of recombinant DNA technology.
In summary, our knowledge of the regulation of
Therefore, this hormone is now available in sufficient
quantities for treatment purposes. Dwarfs who have
growth hormone secretion is not sufficient to describe
pure growth hormone deficiency can be completely
a composite picture. Yet, because of the extreme secre-
cured if treated early in life. Human growth hormone
tion of growth hormone during starvation and its
may prove to be beneficial in other metabolic disorders
important long-term effect to promote protein syn-
because of its widespread metabolic functions.
thesis and tissue growth, we can propose the follow-
ing: the major long-term controller of growth hormone
Panhypopituitarism in the Adult. Panhypopituitarism first
secretion is the long-term state of nutrition of the
occurring in adulthood frequently results from one of
tissues themselves, especially their level of protein
three common abnormalities. Two tumorous conditions,
nutrition. That is, nutritional deficiency or excess tissue
craniopharyngiomas or chromophobe tumors, may
need for cellular proteins—for instance, after a severe
compress the pituitary gland until the functioning
bout of exercise when the muscles’ nutritional status
anterior pituitary cells are totally or almost totally
destroyed. The third cause is thrombosis of the pituitary
has been taxed—in some way increases the rate of
blood vessels. This abnormality occasionally occurs
growth hormone secretion. Growth hormone, in turn,
when a new mother develops circulatory shock after the
promotes synthesis of new proteins while at the same
birth of her baby.
time conserving the proteins already present in the
The general effects of adult panhypopituitarism are
cells.
(1) hypothyroidism, (2) depressed production of gluco-
corticoids by the adrenal glands, and (3) suppressed
secretion of the gonadotropic hormones so that sexual
Abnormalities of Growth
functions are lost. Thus, the picture is that of a lethargic
Hormone Secretion
person (from lack of thyroid hormones) who is gaining
weight (because of lack of fat mobilization by growth,
Panhypopituitarism. This term means decreased secretion
adrenocorticotropic, adrenocortical, and thyroid hor-
of all the anterior pituitary hormones. The decrease in
mones) and has lost all sexual functions. Except for the
secretion may be congenital (present from birth), or it
abnormal sexual functions, the patient can usually be
may occur suddenly or slowly at any time during life,
treated satisfactorily by administering adrenocortical
most often resulting from a pituitary tumor that
and thyroid hormones.
destroys the pituitary gland.
Gigantism. Occasionally, the
acidophilic,
growth
Dwarfism. Most instances of dwarfism result from gener-
hormone-producing cells of the anterior pituitary gland
alized deficiency of anterior pituitary secretion (panhy-
become excessively active, and sometimes even aci-
popituitarism) during childhood. In general, all the
dophilic tumors occur in the gland. As a result, large
physical parts of the body develop in appropriate pro-
quantities of growth hormone are produced. All body
portion to one another, but the rate of development is
tissues grow rapidly, including the bones. If the condi-
greatly decreased. A child who has reached the age of
tion occurs before adolescence, before the epiphyses of
10 years may have the bodily development of a child
the long bones have become fused with the shafts,
aged 4 to 5 years, and the same person at age 20 years
height increases so that the person becomes a giant—
may have the bodily development of a child aged 7 to
up to 8 feet tall.
10 years.
The giant ordinarily has hyperglycemia, and the beta
A person with panhypopituitary dwarfism does not
cells of the islets of Langerhans in the pancreas are
pass through puberty and never secretes sufficient
prone to degenerate because they become overactive
quantities of gonadotropic hormones to develop adult
owing to the hyperglycemia. Consequently, in about 10
sexual functions. In one third of such dwarfs, however,
per cent of giants, full-blown diabetes mellitus eventu-
only growth hormone is deficient; these persons do
ally develops.
mature sexually and occasionally reproduce. In one type
In most giants, panhypopituitarism eventually devel-
of dwarfism (the African pygmy and the Lévi-Lorain
ops if they remain untreated, because the gigantism is
dwarf), the rate of growth hormone secretion is normal
usually caused by a tumor of the pituitary gland that
or high, but there is a hereditary inability to form
grows until the gland itself is destroyed. This eventual
somatomedin C, which is a key step for the promotion
general deficiency of pituitary hormones usually causes
of growth by growth hormone.
death in early adulthood. However, once gigantism is
Treatment with Human Growth Hormone. Growth hor-
diagnosed, further effects can often be blocked by
mones from different species of animals are sufficiently
microsurgical removal of the tumor or by irradiation of
different from one another that they will cause growth
the pituitary gland.
Chapter 75
Pituitary Hormones and Their Control by the Hypothalamus
927
Figure 75-8
Acromegalic patient.
Acromegaly. If an acidophilic tumor occurs after adoles-
ng/ml
cence—that is, after the epiphyses of the long bones
have fused with the shafts—the person cannot grow
5 to 20 years
6
taller, but the bones can become thicker and the soft
20 to 40 years
3
tissues can continue to grow. This condition, shown in
40 to 70 years
1.6
Figure 75-8, is known as acromegaly. Enlargement is
especially marked in the bones of the hands and feet
and in the membranous bones, including the cranium,
nose, bosses on the forehead, supraorbital ridges, lower
Thus, it is highly possible that some of the normal
jawbone, and portions of the vertebrae, because their
aging effects result from diminished growth hormone
growth does not cease at adolescence. Consequently,
secretion. In fact, multiple tests of growth hormone
the lower jaw protrudes forward, sometimes as much as
therapy in older people have demonstrated three
half an inch, the forehead slants forward because of
important effects that suggest antiaging actions: (1)
excess development of the supraorbital ridges, the
increased protein deposition in the body, especially in
nose increases to as much as twice normal size, the feet
the muscles; (2) decreased fat deposits; and (3) a feeling
require size 14 or larger shoes, and the fingers become
of increased energy.
extremely thickened so that the hands are almost twice
normal size. In addition to these effects, changes in the
vertebrae ordinarily cause a hunched back, which is
known clinically as kyphosis. Finally, many soft tissue
Posterior Pituitary Gland
organs, such as the tongue, the liver, and especially the
and Its Relation to the
kidneys, become greatly enlarged.
Hypothalamus
Possible Role of Decreased Growth Hormone Secretion
in Causing Changes Associated with Aging
The posterior pituitary gland, also called the neurohy-
In people who have lost the ability to secrete growth
pophysis, is composed mainly of glial-like cells called
hormone, some features of the aging process accelerate.
pituicytes. The pituicytes do not secrete hormones; they
For instance, a 50-year-old person who has been without
act simply as a supporting structure for large numbers
growth hormone for many years may have the appear-
of terminal nerve fibers and terminal nerve endings
ance of a person aged 65. The aged appearance seems
from nerve tracts that originate in the supraoptic and
to result mainly from decreased protein deposition in
paraventricular nuclei of the hypothalamus, as shown
most tissues of the body and increased fat deposition in
in Figure 75-9. These tracts pass to the neurohypoph-
its place. The physical and physiological effects are
ysis through the pituitary stalk (hypophysial stalk). The
increased wrinkling of the skin, diminished rates of
nerve endings are bulbous knobs that contain many
function of some of the organs, and diminished muscle
mass and strength.
secretory granules. These endings lie on the surfaces of
As one ages, the average plasma concentration of
capillaries, where they secrete two posterior pituitary
growth hormone in an otherwise normal person
hormones:
(1) antidiuretic hormone
(ADH), also
changes approximately as follows:
called vasopressin, and (2) oxytocin.
928
Unit XIV Endocrinology and Reproduction
except that in vasopressin, phenylalanine and arginine
Paraventricular
Supraoptic
nucleus
replace isoleucine and leucine of the oxytocin molecule.
nucleus
The similarity of the molecules explains their partial
functional similarities.
Optic chiasm
Physiological Functions of ADH
Mamillary body
The injection of extremely minute quantities of
Hypothalamic-
ADH—as small as 2 nanograms—can cause decreased
hypophysial
tract
excretion of water by the kidneys (antidiuresis). This
antidiuretic effect is discussed in detail in Chapter 28.
Briefly, in the absence of ADH, the collecting tubules
Posterior pituitary
and ducts become almost impermeable to water, which
prevents significant reabsorption of water and there-
Anterior pituitary
fore allows extreme loss of water into the urine, also
causing extreme dilution of the urine. Conversely, in
the presence of ADH, the permeability of the collect-
Figure 75-9
ing ducts and tubules to water increases greatly and
allows most of the water to be reabsorbed as the
Hypothalamic control of the posterior pituitary.
tubular fluid passes through these ducts, thereby con-
serving water in the body and producing very concen-
trated urine.
If the pituitary stalk is cut above the pituitary gland
The precise mechanism by which ADH acts on the
but the entire hypothalamus is left intact, the posterior
collecting ducts to increase their permeability is only
pituitary hormones continue to be secreted normally,
partially known. Without ADH, the luminal mem-
after a transient decrease for a few days; they are then
branes of the tubular epithelial cells of the collecting
secreted by the cut ends of the fibers within the hypo-
ducts are almost impermeable to water. However,
thalamus and not by the nerve endings in the poste-
immediately inside the cell membrane are a large
rior pituitary. The reason for this is that the hormones
number of special vesicles that have highly water-
are initially synthesized in the cell bodies of the
permeable pores called aquaporins. When ADH acts
supraoptic and paraventricular nuclei and are then
on the cell, it first combines with membrane receptors
transported in combination with “carrier” proteins
that activate adenylyl cyclase and cause the formation
called neurophysins down to the nerve endings in the
of cAMP inside the tubular cell cytoplasm. This causes
posterior pituitary gland, requiring several days to
phosphorylation of elements in the special vesicles,
reach the gland.
which then causes the vesicles to insert into the apical
ADH is formed primarily in the supraoptic nuclei,
cell membranes, thus providing many areas of high
whereas oxytocin is formed primarily in the paraven-
water permeability. All this occurs within
5 to 10
tricular nuclei. Each of these nuclei can synthesize
minutes. Then, in the absence of ADH, the entire
about one sixth as much of the second hormone as of
process reverses in another 5 to 10 minutes. Thus, this
its primary hormone.
process temporarily provides many new pores that
When nerve impulses are transmitted downward
allow free diffusion of water from the tubular fluid
along the fibers from the supraoptic or paraventri-
through the tubular epithelial cells and into the renal
cular nuclei, the hormone is immediately released
interstitial fluid. Water is then absorbed from the col-
from the secretory granules in the nerve endings by
lecting tubules and ducts by osmosis, as explained in
the usual secretory mechanism of exocytosis and is
Chapter 28 in relation to the urine-concentrating
absorbed into adjacent capillaries. Both the neuro-
mechanism of the kidneys.
physin and the hormone are secreted together, but
because they are only loosely bound to each other, the
Regulation of ADH Production
hormone separates almost immediately. The neuro-
Osmotic Regulation. When a concentrated electrolyte
physin has no known function after leaving the nerve
solution is injected into the artery that supplies the
terminals.
hypothalamus, the ADH neurons in the supraoptic and
paraventricular nuclei immediately transmit impulses
Chemical Structures of ADH
into the posterior pituitary to release large quantities
and Oxytocin
of ADH into the circulating blood, sometimes increas-
ing the ADH secretion to as high as 20 times normal.
Both oxytocin and ADH (vasopressin) are polypep-
Conversely, injection of a dilute solution into this
tides, each containing nine amino acids. Their amino
artery causes cessation of the impulses and therefore
acid sequences are the following:
almost total cessation of ADH secretion. Thus, the con-
Vasopressin: Cys-Tyr-Phe-Gln-Asn-Cys-Pro-Arg-
centration of ADH in the body fluids can change from
GlyNH
2
Oxytocin: Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-GlyNH2
small amounts to large amounts, or vice versa, in only
Note that these two hormones are almost identical
a few minutes.
Chapter 75
Pituitary Hormones and Their Control by the Hypothalamus
929
The precise way that the osmotic concentration of
indicating a possible effect of oxytocin during delivery.
the extracellular fluid controls ADH secretion is not
(2) The amount of oxytocin in the plasma increases
clear. Yet somewhere in or near the hypothalamus
during labor, especially during the last stage. (3) Stim-
are modified neuron receptors called osmoreceptors.
ulation of the cervix in a pregnant animal elicits
When the extracellular fluid becomes too concen-
nervous signals that pass to the hypothalamus and
trated, fluid is pulled by osmosis out of the osmore-
cause increased secretion of oxytocin. These effects
ceptor cell, decreasing its size and initiating
and this possible mechanism for aiding in the birth
appropriate nerve signals in the hypothalamus to
process are discussed in much more detail in Chapter
cause additional ADH secretion. Conversely, when the
82.
extracellular fluid becomes too dilute, water moves by
Oxytocin Aids in Milk Ejection by the Breasts. Oxytocin also
osmosis in the opposite direction, into the cell, and this
plays an especially important role in lactation—a role
decreases the signal for ADH secretion. Although
that is far better understood than its role in delivery.
some researchers place these osmoreceptors in the
In lactation, oxytocin causes milk to be expressed from
hypothalamus itself (possibly even in the supraoptic
the alveoli into the ducts of the breast so that the baby
nuclei), others believe that they are located in the
can obtain it by suckling.
organum vasculosum, a highly vascular structure in the
This mechanism works as follows: The suckling stim-
anteroventral wall of the third ventricle.
ulus on the nipple of the breast causes signals to be
Regardless of the mechanism, concentrated body
transmitted through sensory nerves to the oxytocin
fluids stimulate the supraoptic nuclei, whereas dilute
neurons in the paraventricular and supraoptic nuclei
body fluids inhibit them. A feedback control system is
in the hypothalamus, which causes release of oxytocin
available to control the total osmotic pressure of the
by the posterior pituitary gland. The oxytocin is then
body fluids.
carried by the blood to the breasts, where it causes
Further details on the role of ADH in controlling
contraction of myoepithelial cells that lie outside of
renal function and body fluid osmolality are presented
and form a latticework surrounding the alveoli of the
in Chapter 28.
mammary glands. In less than a minute after the begin-
ning of suckling, milk begins to flow. This mechanism
Vasoconstrictor and Pressor Effects of ADH,
is called milk letdown or milk ejection. It is discussed
and Increased ADH Secretion Caused by Low
further in Chapter 82 in relation to the physiology of
Blood Volume
lactation.
Whereas minute concentrations of ADH cause
increased water conservation by the kidneys, higher
References
concentrations of ADH have a potent effect of con-
stricting the arterioles throughout the body and there-
Antunes-Rodrigues J, de Castro M, Elias LL, et al: Neu-
fore increasing the arterial pressure. For this reason,
roendocrine control of body fluid metabolism. Physiol Rev
ADH has another name, vasopressin.
84:169, 2004.
One of the stimuli for causing intense ADH secre-
Besser GM, Thorner MO: Comprehensive Clinical
tion is decreased blood volume. This occurs especially
Endocrinology,
3rd ed. Philadelphia: Mosby, Elsevier
strongly when the blood volume decreases 15 to 25 per
Science Limited, 2002.
cent or more; the secretory rate then sometimes rises
Burbach JP, Luckman SM, Murphy D, Gainer H: Gene
to as high as 50 times normal. The cause of this is the
regulation in the magnocellular hypothalamo-
following.
neurohypophysial system. Physiol Rev 81:1197, 2001.
The atria have stretch receptors that are excited by
Butler AA, Le Roith D: Control of growth by the somatropic
overfilling. When excited, they send signals to the brain
axis: growth hormone and the insulin-like growth factors
have related and independent roles. Annu Rev Physiol
to inhibit ADH secretion. Conversely, when the recep-
63:141, 2001.
tors are unexcited as a result of underfilling, the oppo-
Cummings DE, Merriam GR: Growth hormone therapy in
site occurs, with greatly increased ADH secretion.
adults. Annu Rev Med 54:513, 2003.
Decreased stretch of the baroreceptors of the carotid,
Dattani M, Preece M: Growth hormone deficiency and
aortic, and pulmonary regions also stimulates ADH
related disorders: insights into causation, diagnosis, and
secretion. For further details about this blood volume-
treatment. Lancet 363:1977, 2004.
pressure feedback mechanism, refer to Chapter 28.
Dunn AJ, Swiergiel AH, Palamarchouk V: Brain circuits
involved in corticotropin-releasing factor-norepinephrine
interactions during stress. Ann N Y Acad Sci 1018:25, 2004.
Oxytocic Hormone
Edmondson SR, Thumiger SP, Werther GA, Wraight CJ: Epi-
dermal homeostasis: the role of the growth hormone and
insulin-like growth factor systems. Endocr Rev 24:737,
Oxytocin Causes Contraction of the Pregnant Uterus. The
2003.
hormone oxytocin, in accordance with its name, pow-
Eugster EA, Pescovitz OH: Gigantism. J Clin Endocrinol
erfully stimulates contraction of the pregnant uterus,
Metab 84:4379, 1999.
especially toward the end of gestation. Therefore,
Freeman ME, Kanyicska B, Lerant A, Nagy G: Prolactin:
many obstetricians believe that this hormone is at least
structure, function, and regulation of secretion. Physiol
partially responsible for causing birth of the baby. This
Rev 80:1523, 2000.
is supported by the following facts: (1) In a hypophy-
Gimpl G, Fahrenholz F: The oxytocin receptor system: struc-
sectomized animal, the duration of labor is prolonged,
ture, function, and regulation. Physiol Rev 81:629, 2001.
930
Unit XIV Endocrinology and Reproduction
Goldspink G: Age-related muscle loss and progressive dys-
Moenter SM, Defazio RA, Straume M, Nunemaker CS:
function in mechanosensitive growth factor signaling. Ann
Steroid regulation of GnRH neurons. Ann N Y Acad Sci
N Y Acad Sci 1019:294, 2004.
1007:143, 2003.
Isley WL: Growth hormone therapy for adults: not ready for
Murray RD: Adult growth hormone replacement: current
prime time? Ann Intern Med 137:190, 2002.
understanding. Curr Opin Pharmacol 3:642, 2003.
Larsen PR, Kronenberg HM, Melmed S, Polonsky KS:
Nielsen S, Frokiaer J, Marples D, et al: Aquaporins in the
Williams Textbook of Endocrinology, 10th ed. Philadel-
kidney: from molecules to medicine. Physiol Rev 82:205,
phia: WB Saunders, 2003.
2002.
Lohmeier TE: Neurohypophysial hormones. Am J Physiol
Paisley AN, Trainer PJ: Medical treatment in acromegaly.
Regul Integr Comp Physiol 285:R715, 2003.
Curr Opin Pharmacol 3:672, 2003.
Longo VD, Finch CE: Evolutionary medicine: from dwarf
Stricker EM, Sved AF: Controls of vasopressin secretion and
model systems to healthy centenarians? Science 299:1342,
thirst: similarities and dissimilarities in signals. Physiol
2003.
Behav 77:731, 2002.
C H A P T E R
7
6
Thyroid Metabolic Hormones
The thyroid gland, located immediately below the
larynx on each side of and anterior to the trachea,
is one of the largest of the endocrine glands,
normally weighing 15 to 20 grams in adults. The
thyroid secretes two major hormones, thyroxine and
triiodothyronine, commonly called T4 and T3, respec-
tively. Both of these hormones profoundly increase
the metabolic rate of the body. Complete lack of
thyroid secretion usually causes the basal metabolic rate to fall 40 to 50 per cent
below normal, and extreme excesses of thyroid secretion can increase the basal
metabolic rate to 60 to 100 per cent above normal. Thyroid secretion is con-
trolled primarily by thyroid-stimulating hormone (TSH) secreted by the ante-
rior pituitary gland.
The thyroid gland also secretes calcitonin, an important hormone for calcium
metabolism that is considered in detail in Chapter 79.
The purpose of this chapter is to discuss the formation and secretion of the
thyroid hormones, their metabolic functions, and regulation of their secretion.
Synthesis and Secretion of the Thyroid
Metabolic Hormones
About 93 per cent of the metabolically active hormones secreted by the thyroid
gland is thyroxine, and 7 per cent triiodothyronine. However, almost all the thy-
roxine is eventually converted to triiodothyronine in the tissues, so that both
are functionally important. The functions of these two hormones are qualita-
tively the same, but they differ in rapidity and intensity of action. Triiodothy-
ronine is about four times as potent as thyroxine, but it is present in the blood
in much smaller quantities and persists for a much shorter time than does
thyroxine.
Physiologic Anatomy of the Thyroid Gland. The thyroid gland is composed, as shown
in Figure 76-1, of large numbers of closed follicles (100 to 300 micrometers in
diameter) filled with a secretory substance called colloid and lined with cuboidal
epithelial cells that secrete into the interior of the follicles. The major constituent
of colloid is the large glycoprotein thyroglobulin, which contains the thyroid
hormones within its molecule. Once the secretion has entered the follicles, it
must be absorbed back through the follicular epithelium into the blood before
it can function in the body. The thyroid gland has a blood flow about five times
the weight of the gland each minute, which is a blood supply as great as that of
any other area of the body, with the possible exception of the adrenal cortex.
Iodine Is Required for Formation of Thyroxine
To form normal quantities of thyroxine, about 50 milligrams of ingested iodine
in the form of iodides are required each year, or about 1 mg/week. To prevent
iodine deficiency, common table salt is iodized with about 1 part sodium iodide
to every 100,000 parts sodium chloride.
Fate of Ingested Iodides. Iodides ingested orally are absorbed from the gastro-
intestinal tract into the blood in about the same manner as chlorides. Normally,
931
932
Unit XIV Endocrinology and Reproduction
most of the iodides are rapidly excreted by the
concentration of TSH; TSH stimulates and hypophy-
kidneys, but only after about one fifth are selectively
sectomy greatly diminishes the activity of the iodide
removed from the circulating blood by the cells of the
pump in thyroid cells.
thyroid gland and used for synthesis of the thyroid
hormones.
Thyroglobulin, and Chemistry
of Thyroxine and
Iodide Pump (Iodide Trapping)
Triiodothyronine Formation
The first stage in the formation of thyroid hormones,
Formation and Secretion of Thyroglobulin by the Thyroid Cells.
shown in Figure 76-2, is transport of iodides from the
The thyroid cells are typical protein-secreting glandu-
blood into the thyroid glandular cells and follicles. The
lar cells, as shown in Figure 76-2. The endoplasmic
basal membrane of the thyroid cell has the specific
reticulum and Golgi apparatus synthesize and secrete
ability to pump the iodide actively to the interior of
into the follicles a large glycoprotein molecule called
the cell. This is called iodide trapping. In a normal
thyroglobulin, with a molecular weight of about
gland, the iodide pump concentrates the iodide to
335,000.
about 30 times its concentration in the blood. When
Each molecule of thyroglobulin contains about 70
the thyroid gland becomes maximally active, this con-
tyrosine amino acids, and they are the major substrates
centration ratio can rise to as high as 250 times. The
that combine with iodine to form the thyroid hor-
rate of iodide trapping by the thyroid is influenced
mones. Thus, the thyroid hormones form within the
by several factors, the most important being the
thyroglobulin molecule. That is, the thyroxine and
triiodothyronine hormones formed from the tyrosine
amino acids remain part of the thyroglobulin molecule
during synthesis of the thyroid hormones and even
afterward as stored hormones in the follicular colloid.
Follicle
Oxidation of the Iodide Ion. The first essential step in the
formation of the thyroid hormones is conversion of
Cuboidal epithelial cells
the iodide ions to an oxidized form of iodine, either
nascent iodine (I0) or I3_, that is then capable of com-
Red blood cells
bining directly with the amino acid tyrosine. This oxi-
dation of iodine is promoted by the enzyme peroxidase
and its accompanying hydrogen peroxide, which
Colloid
provide a potent system capable of oxidizing iodides.
The peroxidase is either located in the apical mem-
brane of the cell or attached to it, thus providing the
oxidized iodine at exactly the point in the cell where
the thyroglobulin molecule issues forth from the Golgi
apparatus and through the cell membrane into the
stored thyroid gland colloid. When the peroxidase
Figure 76-1
system is blocked or when it is hereditarily absent
Microscopic appearance of the thyroid gland, showing secretion
from the cells, the rate of formation of thyroid hor-
of thyroglobulin into the follicles.
mones falls to zero.
I2
I-
I-
H2O2
Thyroglobulin
Peroxidase
precursor (TG)
+ Iodination
TG
and
Peroxidase
coupling
ER Golgi
MIT, DIT
Figure 76-2
MIT
DIT
TG
T
Secretion
T3
T3
Thyroid cellular mechanisms for iodine transport,
3
T
T4
Pinocytosis
T4
thyroxine and triiodothyronine formation, and thy-
4
Proteases
Colloid
roxine and triiodothyronine release into the blood.
droplet
MIT, monoiodotyrosine; DIT, diiodotyrosine; T3, tri-
iodothyronine; T4, thyroxine; TG, thyroglobulin.
Chapter 76
Thyroid Metabolic Hormones
933
Iodination of Tyrosine and Formation of the Thyroid Hormones—
monoiodotyrosine couples with one molecule of
“Organification” of Thyroglobulin. The binding of iodine
diiodotyrosine to form triiodothyronine, which repre-
with the thyroglobulin molecule is called organifica-
sents about one fifteenth of the final hormones.
tion of the thyroglobulin. Oxidized iodine even in the
molecular form will bind directly but very slowly with
Storage of Thyroglobulin. The thyroid gland is unusual
the amino acid tyrosine. In the thyroid cells, however,
among the endocrine glands in its ability to store large
the oxidized iodine is associated with an iodinase
amounts of hormone. After synthesis of the thyroid
enzyme (Figure 76-2) that causes the process to occur
hormones has run its course, each thyroglobulin mol-
within seconds or minutes. Therefore, almost as rapidly
ecule contains up to 30 thyroxine molecules and a few
as the thyroglobulin molecule is released from the
triiodothyronine molecules. In this form, the thyroid
Golgi apparatus or as it is secreted through the apical
hormones are stored in the follicles in an amount suf-
cell membrane into the follicle, iodine binds with
ficient to supply the body with its normal requirements
about one sixth of the tyrosine amino acids within the
of thyroid hormones for 2 to 3 months. Therefore,
thyroglobulin molecule.
when synthesis of thyroid hormone ceases, the physi-
Figure
76-3 shows the successive stages of
ologic effects of deficiency are not observed for several
iodination of tyrosine and final formation of the two
months.
important thyroid hormones, thyroxine and tri-
iodothyronine. Tyrosine is first iodized to monoiodoty-
rosine and then to diiodotyrosine. Then, during the
Release of Thyroxine and
next few minutes, hours, and even days, more and more
of the iodotyrosine residues become coupled with one
Triiodothyronine from the
another.
Thyroid Gland
The major hormonal product of the coupling reac-
tion is the molecule thyroxine that remains part of
Thyroglobulin itself is not released into the circulating
the thyroglobulin molecule. Or one molecule
of
blood in measurable amounts; instead, thyroxine and
triiodothyronine must first be cleaved from the thy-
roglobulin molecule, and then these free hormones are
released. This process occurs as follows: The apical
surface of the thyroid cells sends out pseudopod exten-
sions that close around small portions of the colloid to
Iodinase
form pinocytic vesicles that enter the apex of the
I2 + HO
CH2
CHNH2
COOH
thyroid cell. Then lysosomes in the cell cytoplasm
immediately fuse with these vesicles to form digestive
Tyrosine
vesicles containing digestive enzymes from the lyso-
somes mixed with the colloid. Multiple proteases
among the enzymes digest the thyroglobulin mole-
HO
CH2
CHNH2
COOH +
cules and release thyroxine and triiodothyronine in
free form. These then diffuse through the base of the
Monoiodotyrosine
thyroid cell into the surrounding capillaries. Thus, the
thyroid hormones are released into the blood.
HO
CH2
CHNH2
COOH
About three quarters of the iodinated tyrosine in
the thyroglobulin never becomes thyroid hormones
but remains monoiodotyrosine and diiodotyrosine.
Diiodotyrosine
During the digestion of the thyroglobulin molecule to
Monoiodotyrosine + Diiodotyrosine
cause release of thyroxine and triiodothyronine, these
iodinated tyrosines also are freed from the thyroglob-
ulin molecules. However, they are not secreted into the
HO
O
CH2
CHNH2
COOH
blood. Instead, their iodine is cleaved from them by a
deiodinase enzyme that makes virtually all this iodine
available again for recycling within the gland for
3,5,3'-Triiodothyronine
forming additional thyroid hormones. In the congeni-
Diiodotyrosine + Diiodotyrosine
tal absence of this deiodinase enzyme, many persons
become iodine-deficient because of failure of this recy-
cling process.
HO
O
CH2
CHNH2
COOH
Daily Rate of Secretion of Thyroxine and Triiodothyronine.
Thyroxine
About 93 per cent of the thyroid hormone released
from the thyroid gland is normally thyroxine and only
7 per cent is triiodothyronine. However, during the
ensuing few days, about one half of the thyroxine is
Figure 76-3
slowly deiodinated to form additional triiodothyro-
Chemistry of thyroxine and triiodothyronine formation.
nine. Therefore, the hormone finally delivered to and
934
Unit XIV Endocrinology and Reproduction
used by the tissues is mainly triiodothyronine, a total
period as short as 6 to 12 hours and maximal cellular
of about 35 micrograms of triiodothyronine per day.
activity occurring within 2 to 3 days.
Most of the latency and prolonged period of action
of these hormones are probably caused by their
Transport of Thyroxine and
binding with proteins both in the plasma and in the
Triiodothyronine to Tissues
tissue cells, followed by their slow release. However,
we shall see in subsequent discussions that part of the
Thyroxine and Triiodothyronine Are Bound to Plasma Proteins.
latent period also results from the manner in which
On entering the blood, over 99 per cent of the thy-
these hormones perform their functions in the cells
roxine and triiodothyronine combines immediately
themselves.
with several of the plasma proteins, all of which are
synthesized by the liver. They combine mainly with
thyroxine-binding globulin and much less so with
Physiologic Functions of the
thyroxine-binding prealbumin and albumin.
Thyroid Hormones
Thyroxine and Triiodothyronine Are Released Slowly to Tissue
Thyroid Hormones Increase the
Cells. Because of high affinity of the plasma-binding
Transcription of Large Numbers
proteins for the thyroid hormones, these substances—
of Genes
in particular, thyroxine—are released to the tissue
cells slowly. Half the thyroxine in the blood is released
The general effect of thyroid hormone is to activate
to the tissue cells about every 6 days, whereas half the
nuclear transcription of large numbers of genes
triiodothyronine—because of its lower affinity—is
(Figure 76-5). Therefore, in virtually all cells of the
released to the cells in about 1 day.
body, great numbers of protein enzymes, structural
On entering the tissue cells, both thyroxine and tri-
proteins, transport proteins, and other substances are
iodothyronine again bind with intracellular proteins,
synthesized. The net result is generalized increase in
the thyroxine binding more strongly than the tri-
functional activity throughout the body.
iodothyronine. Therefore, they are again stored, but
this time in the target cells themselves, and they are
Most of the Thyroxine Secreted by the Thyroid Is Converted to
used slowly over a period of days or weeks.
Triiodothyronine. Before acting on the genes to increase
genetic transcription, one iodide is removed from
Thyroid Hormones Have Slow Onset and Long Duration of Action.
almost all the thyroxine, thus forming triiodothyro-
After injection of a large quantity of thyroxine into a
nine. Intracellular thyroid hormone receptors have a
human being, essentially no effect on the metabolic
very high affinity for triiodothyronine. Consequently,
rate can be discerned for 2 to 3 days, thereby demon-
more than 90 per cent of the thyroid hormone mole-
strating that there is a long latent period before thy-
cules that bind with the receptors is triiodothyronine.
roxine activity begins. Once activity does begin, it
increases progressively and reaches a maximum in 10
Thyroid Hormones Activate Nuclear Receptors. The thyroid
to 12 days, as shown in Figure 76-4. Thereafter, it
hormone receptors are either attached to the DNA
decreases with a half-life of about 15 days. Some of the
genetic strands or located in proximity to them.
activity persists for as long as 6 weeks to 2 months.
The thyroid hormone receptor usually forms a het-
The actions of triiodothyronine occur about four
erodimer with retinoid X receptor (RXR) at specific
times as rapidly as those of thyroxine, with a latent
thyroid hormone response elements on the DNA. On
binding with thyroid hormone, the receptors become
activated and initiate the transcription process. Then
large numbers of different types of messenger RNA
+10
are formed, followed within another few minutes or
hours by RNA translation on the cytoplasmic ribo-
somes to form hundreds of new intracellular proteins.
However, not all the proteins are increased by similar
+5
percentages—some only slightly, and others at least as
much as sixfold. It is believed that most, if not all, of
the actions of thyroid hormone result from the subse-
Thyroxine injected
quent enzymatic and other functions of these new
0
proteins.
0
10
20
30
40
Days
Thyroid Hormones Increase Cellular
Metabolic Activity
Figure 76-4
Approximate prolonged effect on the basal metabolic rate caused
The thyroid hormones increase the metabolic activi-
by administering a single large dose of thyroxine.
ties of almost all the tissues of the body. The basal
Chapter 76
Thyroid Metabolic Hormones
935
T4
T3
Cell membrane
Iodinase
Cytoplasm
T
3
T3
Nuclear
Thyroid
Retinoid X
hormone
membrane
receptor
receptor
Gene
Thyroid
hormone
response
Nucleus
Gene
transcription
Figure 76-5
mRNA
Thyroid hormone activation of
target cells. Thyroxine
(T4) and
triiodothyronine
(T3)
readily
diffuse through the cell mem-
brane. Much of the T4 is deiodi-
Synthesis of
nated to form T3, which interacts
new proteins
with the thyroid hormone re-
ceptor, bound as a heterodimer
with a retinoid X receptor, of
Many other
CNS
the thyroid hormone response
Growth
Cardiovascular
Metabolism
systems
development
element of the gene. This causes
either increases or decreases in
Cardiac output
Mitochondria
transcription of genes that lead
Tissue blood flow
Na+-K+-ATPase
to formation of proteins, thus
Heart rate
O2 consumption
producing the thyroid hormone
Heart strength
Glucose absorption
response of the cell. The actions
Respiration
Gluconeogenesis
of thyroid hormone on cells
Glycogenolysis
of several different systems
Lipolysis
are shown. mRNA, messenger
Protein synthesis
ribonucleic acid.
BMR
metabolic rate can increase to 60 to 100 per cent above
formation of adenosine triphosphate (ATP) to ener-
normal when large quantities of the hormones are
gize cellular function. However, the increase in the
secreted. The rate of utilization of foods for energy is
number and activity of mitochondria could be the
greatly accelerated. Although the rate of protein syn-
result of increased activity of the cells as well as
thesis is increased, at the same time the rate of protein
the cause of the increase.
catabolism is also increased. The growth rate of young
people is greatly accelerated. The mental processes
Thyroid Hormones Increase Active Transport of Ions Through
are excited, and the activities of most of the other
Cell Membranes. One of the enzymes that increases its
endocrine glands are increased.
activity in response to thyroid hormone is Na+-K+-
ATPase. This in turn increases the rate of
Thyroid Hormones Increase the Number and Activity of Mito-
transport of both sodium and potassium ions through
chondria. When thyroxine or triiodothyronine is given
the cell membranes of some tissues. Because this
to an animal, the mitochondria in most cells of the
process uses energy and increases the amount of heat
animal’s body increase in size as well as number.
produced in the body, it has been suggested that this
Furthermore, the total membrane surface area of the
might be one of the mechanisms by which thyroid
mitochondria increases almost directly in proportion
hormone increases the body’s metabolic rate. In fact,
to the increased metabolic rate of the whole animal.
thyroid hormone also causes the cell membranes of
Therefore, one of the principal functions of thyroxine
most cells to become leaky to sodium ions, which
might be simply to increase the number and activity
further activates the sodium pump and further
of mitochondria, which in turn increases the rate of
increases heat production.
936
Unit XIV Endocrinology and Reproduction
Effect of Thyroid Hormone on Growth
hypothyroidism is often associated with severe ather-
osclerosis, discussed in Chapter 68.
Thyroid hormone has both general and specific effects
One of the mechanisms by which thyroid hormone
on growth. For instance, it has long been known that
decreases the plasma cholesterol concentration is to
thyroid hormone is essential for the metamorphic
increase significantly the rate of cholesterol secretion
change of the tadpole into the frog.
in the bile and consequent loss in the feces. A possible
In humans, the effect of thyroid hormone on growth
mechanism for the increased cholesterol secretion is
is manifest mainly in growing children. In those who
that thyroid hormone induces increased numbers of
are hypothyroid, the rate of growth is greatly retarded.
low-density lipoprotein receptors on the liver cells,
In those who are hyperthyroid, excessive skeletal
leading to rapid removal of low-density lipoproteins
growth often occurs, causing the child to become con-
from the plasma by the liver and subsequent secretion
siderably taller at an earlier age. However, the bones
of cholesterol in these lipoproteins by the liver cells.
also mature more rapidly and the epiphyses close
at an early age, so that the duration of growth and
Increased Requirement for Vitamins. Because thyroid
the eventual height of the adult may actually be
hormone increases the quantities of many bodily
shortened.
enzymes and because vitamins are essential parts of
An important effect of thyroid hormone is to
some of the enzymes or coenzymes, thyroid hormone
promote growth and development of the brain during
causes increased need for vitamins. Therefore, a rela-
fetal life and for the first few years of postnatal life.
tive vitamin deficiency can occur when excess thyroid
If the fetus does not secrete sufficient quantities of
hormone is secreted, unless at the same time increased
thyroid hormone, growth and maturation of the brain
quantities of vitamins are made available.
both before birth and afterward are greatly retarded,
and the brain remains smaller than normal. Without
Increased Basal Metabolic Rate. Because thyroid
specific thyroid therapy within days or weeks after
hormone increases metabolism in almost all cells of
birth, the child without a thyroid gland will remain
the body, excessive quantities of the hormone can
mentally deficient throughout life. This is discussed
occasionally increase the basal metabolic rate 60 to
more fully later in the chapter.
100 per cent above normal. Conversely, when no
thyroid hormone is produced, the basal metabolic
rate falls almost to one-half normal. Figure 76-6 shows
the approximate relation between the daily supply
Effects of Thyroid Hormone
of thyroid hormones and the basal metabolic rate.
on Specific Bodily Mechanisms
Extreme amounts of the hormones are required to
cause very high basal metabolic rates.
Stimulation of Carbohydrate Metabolism. Thyroid hormone
stimulates almost all aspects of carbohydrate meta-
Decreased Body Weight. Greatly increased thyroid
bolism, including rapid uptake of glucose by the
hormone almost always decreases the body weight,
cells, enhanced glycolysis, enhanced gluconeogenesis,
increased rate of absorption from the gastrointestinal
tract, and even increased insulin secretion with its
resultant secondary effects on carbohydrate metabo-
lism. All these effects probably result from the overall
+30
increase in cellular metabolic enzymes caused by
thyroid hormone.
+20
+10
Stimulation of Fat Metabolism. Essentially all aspects of
fat metabolism are also enhanced under the influence
0
of thyroid hormone. In particular, lipids are mobilized
rapidly from the fat tissue, which decreases the fat
-10
stores of the body to a greater extent than almost any
other tissue element. This also increases the free fatty
-20
acid concentration in the plasma and greatly acceler-
-30
ates the oxidation of free fatty acids by the cells.
-40
Effect on Plasma and Liver Fats. Increased thyroid
-45
hormone decreases the concentrations of cholesterol,
0
100
200
300
phospholipids, and triglycerides in the plasma, even
Thyroid hormones (mg/day)
though it increases the free fatty acids. Conversely,
decreased thyroid secretion greatly increases the
plasma concentrations of cholesterol, phospholipids,
Figure 76-6
and triglycerides and almost always causes excessive
deposition of fat in the liver as well. The large increase
Approximate relation of daily rate of thyroid hormone (T4 and T3)
in circulating plasma cholesterol in prolonged
secretion to the basal metabolic rate.
Chapter 76
Thyroid Metabolic Hormones
937
and greatly decreased hormone almost always
secretion of the digestive juices and the motility of the
increases the body weight; these effects do not always
gastrointestinal tract. Hyperthyroidism often results in
occur, because thyroid hormone also increases the
diarrhea. Lack of thyroid hormone can cause consti-
appetite, and this may counterbalance the change in
pation.
the metabolic rate.
Excitatory Effects on the Central Nervous System. In general,
Effect of Thyroid Hormones on the Cardiovascular System
thyroid hormone increases the rapidity of cerebration
Increased Blood Flow and Cardiac Output. Increased
but also often dissociates this; conversely, lack of
metabolism in the tissues causes more rapid utilization
thyroid hormone decreases this function. The hyper-
of oxygen than normal and release of greater than
thyroid individual is likely to have extreme nervous-
normal quantities of metabolic end products from the
ness and many psychoneurotic tendencies, such as
tissues. These effects cause vasodilation in most body
anxiety complexes, extreme worry, and paranoia.
tissues, thus increasing blood flow. The rate of blood
flow in the skin especially increases because of the
Effect on the Function of the Muscles. Slight increase in
increased need for heat elimination from the body. As
thyroid hormone usually makes the muscles react with
a consequence of the increased blood flow, cardiac
vigor, but when the quantity of hormone becomes
output also increases, sometimes rising to 60 per cent
excessive, the muscles become weakened because of
or more above normal when excessive thyroid
excess protein catabolism. Conversely, lack of thyroid
hormone is present and falling to only 50 per cent of
hormone causes the muscles to become sluggish, and
normal in very severe hypothyroidism.
they relax slowly after a contraction.
Increased Heart Rate. The heart rate increases con-
Muscle Tremor. One of the most characteristic signs of
siderably more under the influence of thyroid
hyperthyroidism is a fine muscle tremor. This is not the
hormone than would be expected from the increase in
coarse tremor that occurs in Parkinson’s disease or in
cardiac output. Therefore, thyroid hormone seems to
shivering, because it occurs at the rapid frequency of
have a direct effect on the excitability of the heart,
10 to 15 times per second. The tremor can be observed
which in turn increases the heart rate. This effect is of
easily by placing a sheet of paper on the extended
particular importance because the heart rate is one of
fingers and noting the degree of vibration of the paper.
the sensitive physical signs that the clinician uses in
This tremor is believed to be caused by increased reac-
determining whether a patient has excessive or dimin-
tivity of the neuronal synapses in the areas of the
ished thyroid hormone production.
spinal cord that control muscle tone. The tremor is an
important means for assessing the degree of thyroid
Increased Heart Strength. The increased enzymatic
hormone effect on the central nervous system.
activity caused by increased thyroid hormone produc-
tion apparently increases the strength of the heart
Effect on Sleep. Because of the exhausting effect of
when only a slight excess of thyroid hormone is
thyroid hormone on the musculature and on the
secreted. This is analogous to the increase in heart
central nervous system, the hyperthyroid subject often
strength that occurs in mild fevers and during exercise.
has a feeling of constant tiredness, but because of the
However, when thyroid hormone is increased
excitable effects of thyroid hormone on the synapses,
markedly, the heart muscle strength becomes
it is difficult to sleep. Conversely, extreme somnolence
depressed because of long-term excessive protein
is characteristic of hypothyroidism, with sleep some-
catabolism. Indeed, some severely thyrotoxic patients
times lasting 12 to 14 hours a day.
die of cardiac decompensation secondary to myocar-
dial failure and to increased cardiac load imposed by
Effect on Other Endocrine Glands. Increased thyroid
the increase in cardiac output.
hormone increases the rates of secretion of most other
endocrine glands, but it also increases the need of the
Normal Arterial Pressure. The mean arterial pressure
tissues for the hormones. For instance, increased thy-
usually remains about normal after administration of
roxine secretion increases the rate of glucose metabo-
thyroid hormone. Because of increased blood flow
lism everywhere in the body and therefore causes a
through the tissues between heartbeats, the pulse pres-
corresponding need for increased insulin secretion by
sure is often increased, with the systolic pressure ele-
the pancreas. Also, thyroid hormone increases many
vated in hyperthyroidism 10 to 15 mm Hg and the
metabolic activities related to bone formation and, as
diastolic pressure reduced a corresponding amount.
a consequence, increases the need for parathyroid
hormone. Thyroid hormone also increases the rate at
Increased Respiration. The increased rate of metabolism
which adrenal glucocorticoids are inactivated by the
increases the utilization of oxygen and formation of
liver. This leads to feedback increase in adrenocorti-
carbon dioxide; these effects activate all the mecha-
cotropic hormone production by the anterior pituitary
nisms that increase the rate and depth of respiration.
and, therefore, increased rate of glucocorticoid secre-
tion by the adrenal glands.
Increased Gastrointestinal Motility. In addition to
increased appetite and food intake, which has been
Effect of Thyroid Hormone on Sexual Function. For normal
discussed, thyroid hormone increases both the rates of
sexual function, thyroid secretion needs to be
938
Unit XIV Endocrinology and Reproduction
approximately normal. In men, lack of thyroid
The most important early effect after administration
hormone is likely to cause loss of libido; great excesses
of TSH is to initiate proteolysis of the thyroglobulin,
of the hormone, however, sometimes cause impotence.
which causes release of thyroxine and triiodothyro-
In women, lack of thyroid hormone often causes
nine into the blood within 30 minutes. The other
menorrhagia and polymenorrhea— that is, respec-
effects require hours or even days and weeks to
tively, excessive and frequent menstrual bleeding.
develop fully.
Yet, strangely enough, in other women thyroid lack
may cause irregular periods and occasionally even
Cyclic Adenosine Monophosphate Mediates the Stimulatory
amenorrhea.
Effect of TSH. In the past, it was difficult to explain the
A hypothyroid woman, like a man, is likely to have
many and varied effects of TSH on the thyroid cell. It
greatly decreased libido. To make the picture still more
is now clear that most, if not all, of these effects result
confusing, in the hyperthyroid woman, oligomenor-
from activation of the
“second messenger” cyclic
rhea, which means greatly reduced bleeding, is
adenosine monophosphate (cAMP) system of the cell.
common, and occasionally amenorrhea results.
The first event in this activation is binding of TSH
The action of thyroid hormone on the gonads
with specific TSH receptors on the basal membrane
cannot be pinpointed to a specific function but prob-
surfaces of the thyroid cell. This then activates adeny-
ably results from a combination of direct metabolic
lyl cyclase in the membrane, which increases the for-
effects on the gonads as well as excitatory and
mation of cAMP inside the cell. Finally, the cAMP acts
inhibitory feedback effects operating through the
as a second messenger to activate protein kinase, which
anterior pituitary hormones that control the sexual
causes multiple phosphorylations throughout the cell.
functions.
The result is both an immediate increase in secretion
of thyroid hormones and prolonged growth of the
thyroid glandular tissue itself.
Regulation of Thyroid
This method for control of thyroid cell activity is
similar to the function of cAMP as a “second messen-
Hormone Secretion
ger” in many other target tissues of the body, as
discussed in Chapter 74.
To maintain normal levels of metabolic activity in the
body, precisely the right amount of thyroid hormone
must be secreted at all times; to achieve this, specific
feedback mechanisms operate through the hypothala-
Anterior Pituitary Secretion of TSH Is
mus and anterior pituitary gland to control the rate of
Regulated by Thyrotropin-Releasing
thyroid secretion. These mechanisms are as follows.
Hormone from the Hypothalamus
TSH (from the Anterior Pituitary Gland) Increases Thyroid Secre-
Anterior pituitary secretion of TSH is controlled
tion. TSH, also known as thyrotropin, is an anterior
by a hypothalamic hormone, thyrotropin-releasing
pituitary hormone, a glycoprotein with a molecular
hormone (TRH), which is secreted by nerve endings
weight of about 28,000. This hormone, also discussed
in the median eminence of the hypothalamus. From
in Chapter 74, increases the secretion of thyroxine and
the median eminence, the TRH is then transported to
triiodothyronine by the thyroid gland. Its specific
the anterior pituitary by way of the hypothalamic-
effects on the thyroid gland are as follows:
hypophysial portal blood, as explained in Chapter 74.
1. Increased proteolysis of the thyroglobulin that
TRH has been obtained in pure form. It is a simple
has already been stored in the follicles, with
substance, a tripeptide amide—pyroglutamyl-histidyl-
resultant release of the thyroid hormones into
proline-amide. TRH directly affects the anterior pitu-
the circulating blood and diminishment of the
itary gland cells to increase their output of TSH. When
follicular substance itself
the blood portal system from the hypothalamus to the
2. Increased activity of the iodide pump, which
anterior pituitary gland becomes blocked, the rate of
increases the rate of “iodide trapping” in the
secretion of TSH by the anterior pituitary decreases
glandular cells, sometimes increasing the ratio of
greatly but is not reduced to zero.
intracellular to extracellular iodide concentration
The molecular mechanism by which TRH causes
in the glandular substance to as much as eight
the TSH-secreting cells of the anterior pituitary to
times normal
produce TSH is first to bind with TRH receptors in the
3. Increased iodination of tyrosine to form the
pituitary cell membrane. This in turn activates the phos-
thyroid hormones
pholipase second messenger system inside the pituitary
4. Increased size and increased secretory activity of
cells to produce large amounts of phospholipase C, fol-
the thyroid cells
lowed by a cascade of other second messengers,
5. Increased number of thyroid cells plus a change
including calcium ions and diacyl glycerol, which even-
from cuboidal to columnar cells and much
tually leads to TSH release.
infolding of the thyroid epithelium into the
follicles
Effects of Cold and Other Neurogenic Stimuli on TRH and TSH
In summary, TSH increases all the known secretory
Secretion. One of the best-known stimuli for increas-
activities of the thyroid glandular cells.
ing the rate of TRH secretion by the hypothalamus,
Chapter 76
Thyroid Metabolic Hormones
939
and therefore TSH secretion by the anterior pituitary
anterior pituitary gland itself. Regardless of the mech-
gland, is exposure of an animal to cold. This effect
anism of the feedback, its effect is to maintain an
almost certainly results from excitation of the hypo-
almost constant concentration of free thyroid hor-
thalamic centers for body temperature control.
mones in the circulating body fluids.
Exposure of rats for several weeks to severe cold
increases the output of thyroid hormones sometimes
Antithyroid Substances
to more than 100 per cent of normal and can increase
the basal metabolic rate as much as 50 per cent.
Drugs that suppress thyroid secretion are called antithy-
Indeed, persons moving to arctic regions have been
roid substances. The best known of these substances are
known to develop basal metabolic rates 15 to 20 per
thiocyanate, propylthiouracil, and high concentrations
cent above normal.
of inorganic iodides. The mechanism by which each of
Various emotional reactions can also affect the
these blocks thyroid secretion is different from the
output of TRH and TSH and therefore indirectly
others, and they can be explained as follows.
affect the secretion of thyroid hormones. Excitement
Thiocyanate Ions Decrease Iodide Trapping. The same active
and anxiety—conditions that greatly stimulate the
pump that transports iodide ions into the thyroid cells
sympathetic nervous system—cause an acute decrease
can also pump thiocyanate ions, perchlorate ions, and
in secretion of TSH, perhaps because these states
nitrate ions. Therefore, the administration of thio-
increase the metabolic rate and body heat and there-
cyanate (or one of the other ions as well) in high enough
fore exert an inverse effect on the heat control center.
concentration can cause competitive inhibition of
Neither these emotional effects nor the effect of
iodide transport into the cell— that is, inhibition of the
cold is observed after the hypophysial stalk has been
iodide-trapping mechanism.
cut, demonstrating that both of these effects are medi-
The decreased availability of iodide in the glandular
ated by way of the hypothalamus.
cells does not stop the formation of thyroglobulin; it
merely prevents the thyroglobulin that is formed from
becoming iodinated and therefore from forming the
thyroid hormones. This deficiency of the thyroid hor-
Feedback Effect of Thyroid Hormone
mones in turn leads to increased secretion of TSH by
to Decrease Anterior Pituitary
the anterior pituitary gland, which causes overgrowth of
Secretion of TSH
the thyroid gland even though the gland still does not
form adequate quantities of thyroid hormones. There-
Increased thyroid hormone in the body fluids
fore, the use of thiocyanates and some other ions to
block thyroid secretion can lead to development of a
decreases secretion of TSH by the anterior pituitary.
greatly enlarged thyroid gland, which is called a goiter.
When the rate of thyroid hormone secretion rises to
about 1.75 times normal, the rate of TSH secretion
Propylthiouracil Decreases Thyroid Hormone Formation. Propy-
falls essentially to zero. Almost all this feedback
lthiouracil
(and other, similar compounds, such as
depressant effect occurs even when the anterior pitu-
methimazole and carbimazole) prevents formation of
itary has been separated from the hypothalamus.
thyroid hormone from iodides and tyrosine. The mech-
Therefore, as shown in Figure 76-7, it is probable that
anism of this is partly to block the peroxidase enzyme
increased thyroid hormone inhibits anterior pituitary
that is required for iodination of tyrosine and partly to
secretion of TSH mainly by a direct effect on the
block the coupling of two iodinated tyrosines to form
thyroxine or triiodothyronine.
Propylthiouracil, like thiocyanate, does not prevent
formation of thyroglobulin. The absence of thyroxine
and triiodothyronine in the thyroglobulin can lead to
Hypothalamus
tremendous feedback enhancement of TSH secretion
(? increased temperature)
by the anterior pituitary gland, thus promoting growth
(Thyrotropin-releasing hormone)
of the glandular tissue and forming a goiter.
Anterior pituitary
Iodides in High Concentrations Decrease Thyroid Activity and
?
Thyroid Gland Size. When iodides are present in the blood
Thyroid-
stimulating
in high concentration (100 times the normal plasma
hormone
level), most activities of the thyroid gland are decreased,
Inhibits
but often they remain decreased for only a few weeks.
The effect is to reduce the rate of iodide trapping, so
Cells
that the rate of iodination of tyrosine to form thyroid
?
hormones is also decreased. Even more important, the
Hypertrophy
normal endocytosis of colloid from the follicles by the
Increased
Increased
Thyroxine
thyroid glandular cells is paralyzed by the high iodide
metabolism
secretion
concentrations. Because this is the first step in release
of the thyroid hormones from the storage colloid, there
Thyroid
Iodine
is almost immediate shutdown of thyroid hormone
secretion into the blood.
Because iodides in high concentrations decrease all
Figure 76-7
phases of thyroid activity, they slightly decrease the size
Regulation of thyroid secretion.
of the thyroid gland and especially decrease its blood
940
Unit XIV Endocrinology and Reproduction
supply, in contradistinction to the opposite effects
Symptoms of Hyperthyroidism
caused by most of the other antithyroid agents. For this
reason, iodides are frequently administered to patients
The symptoms of hyperthyroidism are obvious from the
for 2 to 3 weeks before surgical removal of the thyroid
preceding discussion of the physiology of the thyroid
gland to decrease the necessary amount of surgery,
hormones: (1) a high state of excitability, (2) intolerance
especially to decrease the amount of bleeding.
to heat, (3) increased sweating, (4) mild to extreme
weight loss (sometimes as much as 100 pounds), (5)
varying degrees of diarrhea, (6) muscle weakness, (7)
nervousness or other psychic disorders, (8) extreme
Diseases of the Thyroid
fatigue but inability to sleep, and (9) tremor of the
hands.
Hyperthyroidism
Exophthalmos. Most people with hyperthyroidism
Most effects of hyperthyroidism are obvious from the
develop some degree of protrusion of the eyeballs, as
preceding discussion of the various physiologic effects
shown in Figure 76-8. This condition is called exoph-
of thyroid hormone. However, some specific effects
thalmos. A major degree of exophthalmos occurs in
should be mentioned in connection especially with the
about one third of hyperthyroid patients, and the con-
development, diagnosis, and treatment of hyperthy-
dition sometimes becomes so severe that the eyeball
roidism.
protrusion stretches the optic nerve enough to damage
vision. Much more often, the eyes are damaged because
Causes of Hyperthyroidism (Toxic Goiter, Thyrotoxicosis, Graves’
the eyelids do not close completely when the person
Disease). In most patients with hyperthyroidism, the
blinks or is asleep. As a result, the epithelial surfaces of
thyroid gland is increased to two to three times
the eyes become dry and irritated and often infected,
normal size, with tremendous hyperplasia and infolding
resulting in ulceration of the cornea.
of the follicular cell lining into the follicles, so that the
The cause of the protruding eyes is edematous
number of cells is increased greatly. Also, each cell
swelling of the retro-orbital tissues and degenerative
increases its rate of secretion severalfold; radioactive
changes in the extraocular muscles. In most patients,
iodine uptake studies indicate that some of these hyper-
immunoglobulins can be found in the blood that react
plastic glands secrete thyroid hormone at rates 5 to 15
with the eye muscles. Furthermore, the concentration of
times normal.
these immunoglobulins is usually highest in patients
The changes in the thyroid gland in most instances
who have high concentrations of TSIs. Therefore, there
are similar to those caused by excessive TSH. However,
is much reason to believe that exophthalmos, like hyper-
plasma TSH concentrations are less than normal
thyroidism itself, is an autoimmune process. The exoph-
rather than enhanced in almost all patients and often
thalmos usually is greatly ameliorated with treatment of
are essentially zero. However, other substances that
the hyperthyroidism.
have actions similar to those of TSH are found in
the blood of almost all these patients. These substances
are immunoglobulin antibodies that bind with the
same membrane receptors that bind TSH. They induce
continual activation of the cAMP system of the cells,
with resultant development of hyperthyroidism. These
antibodies are called thyroid-stimulating immunoglobu-
lin and are designated TSI. They have a prolonged
stimulating effect on the thyroid gland, lasting for as
long as 12 hours, in contrast to a little over 1 hour for
TSH. The high level of thyroid hormone secretion
caused by TSI in turn suppresses anterior pituitary for-
mation of TSH.
The antibodies that cause hyperthyroidism almost
certainly occur as the result of autoimmunity that has
developed against thyroid tissue. Presumably, at some
time in the history of the person, an excess of thyroid
cell antigens was released from the thyroid cells, and this
has resulted in the formation of antibodies against the
thyroid gland itself.
Thyroid Adenoma. Hyperthyroidism occasionally results
from a localized adenoma (a tumor) that develops in the
thyroid tissue and secretes large quantities of thyroid
hormone. This is different from the more usual type of
hyperthyroidism, in that it usually is not associated with
evidence of any autoimmune disease. An interesting
effect of the adenoma is that as long as it continues to
secrete large quantities of thyroid hormone, secretory
Figure 76-8
function in the remainder of the thyroid gland is almost
totally inhibited because the thyroid hormone from the
Patient with exophthalmic hyperthyroidism. Note protrusion of the
adenoma depresses the production of TSH by the pitu-
eyes and retraction of the superior eyelids. The basal metabolic
itary gland.
rate was +40. (Courtesy Dr. Leonard Posey.)
Chapter 76
Thyroid Metabolic Hormones
941
Diagnostic Tests for Hyperthyroidism. For the usual case of
for the formation of adequate quantities of thyroid
hyperthyroidism, the most accurate diagnostic test is
hormone. In certain areas of the world, notably in the
direct measurement of the concentration of “free” thy-
Swiss Alps, the Andes, and the Great Lakes region of
roxine (and sometimes triiodothyronine) in the plasma,
the United States, insufficient iodine is present in the
using appropriate radioimmunoassay procedures.
soil for the foodstuffs to contain even this minute quan-
Other tests that are sometimes used are as follows:
tity. Therefore, in the days before iodized table salt,
1. The basal metabolic rate is usually increased to +30
many people who lived in these areas developed
to +60 in severe hyperthyroidism.
extremely large thyroid glands, called endemic goiters.
2. The concentration of TSH in the plasma is
The mechanism for development of large endemic
measured by radioimmunoassay. In the usual
goiters is the following: Lack of iodine prevents pro-
type of thyrotoxicosis, anterior pituitary secretion
duction of both thyroxine and triiodothyronine. As a
of TSH is so completely suppressed by the
result, no hormone is available to inhibit production of
large amounts of circulating thyroxine and
TSH by the anterior pituitary; this causes the pituitary
triiodothyronine that there is almost no plasma
to secrete excessively large quantities of TSH. The TSH
TSH.
then stimulates the thyroid cells to secrete tremendous
3. The concentration of TSI is measured by
amounts of thyroglobulin colloid into the follicles, and
radioimmunoassay. This is usually high in
the gland grows larger and larger. But because of lack
thyrotoxicosis but low in thyroid adenoma.
of iodine, thyroxine and triiodothyronine production
does not occur in the thyroglobulin molecule and there-
Physiology of Treatment in Hyperthyroidism. The most direct
fore does not cause the normal suppression of TSH pro-
treatment for hyperthyroidism is surgical removal of
duction by the anterior pituitary. The follicles become
most of the thyroid gland. In general, it is desirable to
tremendous in size, and the thyroid gland may increase
prepare the patient for surgical removal of the gland
to 10 to 20 times normal size.
before the operation. This is done by administering
propylthiouracil, usually for several weeks, until the
Idiopathic Nontoxic Colloid Goiter. Enlarged thyroid glands
basal metabolic rate of the patient has returned to
similar to those of endemic colloid goiter can also occur
normal. Then, administration of high concentrations of
in people who do not have iodine deficiency. These
iodides for 1 to 2 weeks immediately before operation
goitrous glands may secrete normal quantities of
causes the gland itself to recede in size and its blood
thyroid hormones, but more frequently, the secretion of
supply to diminish. By using these preoperative proce-
hormone is depressed, as in endemic colloid goiter.
dures, the operative mortality is less than 1 in 1000 in
The exact cause of the enlarged thyroid gland in
the better hospitals, whereas before development of
patients with idiopathic colloid goiter is not known, but
modern procedures, operative mortality was 1 in 25.
most of these patients show signs of mild thyroiditis;
therefore, it has been suggested that the thyroiditis
causes slight hypothyroidism, which then leads to
Treatment of the Hyperplastic Thyroid Gland with
increased TSH secretion and progressive growth of the
Radioactive Iodine
noninflamed portions of the gland. This could explain
Eighty to 90 per cent of an injected dose of iodide is
why these glands usually are nodular, with some por-
absorbed by the hyperplastic, toxic thyroid gland within
tions of the gland growing while other portions are
1 day after injection. If this injected iodine is radio-
being destroyed by thyroiditis.
active, it can destroy most of the secretory cells of the
In some persons with colloid goiter, the thyroid gland
thyroid gland. Usually 5 millicuries of radioactive iodine
has an abnormality of the enzyme system required for
is given to the patient, whose condition is reassessed
formation of the thyroid hormones. Among the abnor-
several weeks later. If the patient is still hyperthyroid,
malities often encountered are the following:
additional doses are administered until normal thyroid
1. Deficient iodide-trapping mechanism, in which
status is reached.
iodine is not pumped adequately into the thyroid
cells
Hypothyroidism
2. Deficient peroxidase system, in which the iodides
are not oxidized to the iodine state
The effects of hypothyroidism, in general, are opposite
3. Deficient coupling of iodinated tyrosines in the
to those of hyperthyroidism, but there are a few
thyroglobulin molecule, so that the final thyroid
physiologic mechanisms peculiar to hypothyroidism.
hormones cannot be formed
Hypothyroidism, like hyperthyroidism, probably is ini-
4. Deficiency of the deiodinase enzyme, which
tiated by autoimmunity against the thyroid gland, but
prevents recovery of iodine from the iodinated
immunity that destroys the gland rather than stimulates
tyrosines that are not coupled to form the thyroid
it. The thyroid glands of most of these patients first have
hormones (this is about two thirds of the iodine),
autoimmune “thyroiditis,” which means thyroid inflam-
thus leading to iodine deficiency
mation. This causes progressive deterioration and finally
Finally, some foods contain goitrogenic substances
fibrosis of the gland, with resultant diminished or absent
that have a propylthiouracil-type of antithyroid activity,
secretion of thyroid hormone. Several other types of
thus also leading to TSH-stimulated enlargement of the
hypothyroidism also occur, often associated with devel-
thyroid gland. Such goitrogenic substances are found
opment of enlarged thyroid glands, called thyroid goiter,
especially in some varieties of turnips and cabbages.
as follows.
Physiologic Characteristics of Hypothyroidism. Whether
Endemic Colloid Goiter Caused by Dietary Iodide Deficiency. The
hypothyroidism is due to thyroiditis, endemic colloid
term “goiter” means a greatly enlarged thyroid gland.
goiter, idiopathic colloid goiter, destruction of the
As pointed out in the discussion of iodine metabolism,
thyroid gland by irradiation, or surgical removal of the
about 50 milligrams of iodine are required each year
thyroid gland, the physiologic effects are the same. They
942
Unit XIV Endocrinology and Reproduction
include fatigue and extreme somnolence with sleeping
is usually associated with increased atherosclerosis.
up to 12 to 14 hours a day, extreme muscular sluggish-
Therefore, many hypothyroid patients, particularly
ness, slowed heart rate, decreased cardiac output,
those with myxedema, develop atherosclerosis, which
decreased blood volume, sometimes increased body
in turn results in peripheral vascular disease, deafness,
weight, constipation, mental sluggishness, failure of
and coronary artery disease with consequent early
many trophic functions in the body evidenced by
death.
depressed growth of hair and scaliness of the skin,
development of a froglike husky voice, and, in severe
Diagnostic Tests in Hypothyroidism. The tests already
cases, development of an edematous appearance
described for diagnosis of hyperthyroidism give oppo-
throughout the body called myxedema.
site results in hypothyroidism. The free thyroxine in
the blood is low. The basal metabolic rate in myxedema
Myxedema. Myxedema develops in the patient with
ranges between -30 and -50. And the secretion of
almost total lack of thyroid hormone function. Figure
TSH by the anterior pituitary when a test dose of
76-9 shows such a patient, demonstrating bagginess
TRH is administered is usually greatly increased
under the eyes and swelling of the face. In this
(except in those rare instances of hypothyroidism
condition, for reasons not explained, greatly increased
caused by depressed response of the pituitary gland to
quantities of hyaluronic acid and chondroitin sulfate
TRH).
bound with protein form excessive tissue gel in the
interstitial spaces, and this causes the total quantity of
Treatment of Hypothyroidism. Figure 76-4 shows the effect
interstitial fluid to increase. Because of the gel nature of
of thyroxine on the basal metabolic rate, demonstrating
the excess fluid, it is mainly immobile, and the edema is
that the hormone normally has a duration of action of
the nonpitting type.
more than 1 month. Consequently, it is easy to maintain
a steady level of thyroid hormone activity in the body
Atherosclerosis in Hypothyroidism. As pointed out earlier,
by daily oral ingestion of a tablet or more containing
lack of thyroid hormone increases the quantity of blood
thyroxine. Furthermore, proper treatment of the
cholesterol because of altered fat and cholesterol
hypothyroid patient results in such complete normality
metabolism and diminished liver excretion of
that formerly myxedematous patients have lived into
cholesterol in the bile. The increase in blood cholesterol
their 90s after treatment for more than 50 years.
Cretinism
Cretinism is caused by extreme hypothyroidism during
fetal life, infancy, or childhood. This condition is char-
acterized especially by failure of body growth and by
mental retardation. It results from congenital lack of a
thyroid gland (congenital cretinism), from failure of the
thyroid gland to produce thyroid hormone because of a
genetic defect of the gland, or from iodine lack in the
diet (endemic cretinism). The severity of endemic cre-
tinism varies greatly, depending on the amount of iodine
in the diet, and whole populaces of an endemic geo-
graphic iodine-deficient soil area have been known to
have cretinoid tendencies.
A neonate without a thyroid gland may have normal
appearance and function because it was supplied with
some (but usually not enough) thyroid hormone by the
mother while in utero, but a few weeks after birth, the
neonate’s movements become sluggish and both physi-
cal and mental growth begin to be greatly retarded.
Treatment of the neonate with cretinism at any time
with adequate iodine or thyroxine usually causes
normal return of physical growth, but unless the cre-
tinism is treated within a few weeks after birth, mental
growth remains permanently retarded. This results from
retardation of the growth, branching, and myelination
of the neuronal cells of the central nervous system at
this critical time in the normal development of the
mental powers.
Skeletal growth in the child with cretinism is charac-
teristically more inhibited than is soft tissue growth. As
a result of this disproportionate rate of growth, the soft
tissues are likely to enlarge excessively, giving the child
with cretinism an obese, stocky, and short appearance.
Occasionally the tongue becomes so large in relation to
the skeletal growth that it obstructs swallowing and
Figure 76-9
breathing, inducing a characteristic guttural breathing
Patient with myxedema. (Courtesy Dr. Herbert Langford.)
that sometimes chokes the child.
Chapter 76
Thyroid Metabolic Hormones
943
References
Roberts CG, Ladenson PW: Hypothyroidism. Lancet 363:
793, 2004.
Silva JE: The thermogenic effect of thyroid hormone and its
Besser GM, Thorner MO: Comprehensive Clinical
clinical implications. Ann Intern Med 139:205, 2003.
Endocrinology,
3rd ed. Philadelphia: Mosby, Elsevier
Stassi G, De Maria R: Autoimmune thyroid disease: new
Science, 2002.
models of cell death in autoimmunity. Nat Rev Immunol
Burger AG: Environment and thyroid function. J Clin
2:195, 2002.
Endocrinol Metab 89:1526, 2004.
Szkudlinski MW, Fremont V, Ronin C, Weintraub BD:
Cooper DS: Hyperthyroidism. Lancet 362:459, 2003.
Thyroid-stimulating hormone and thyroid-stimulating
De La Vieja A, Dohan O, et al: Molecular analysis of the
hormone receptor structure-function relationships.
sodium/iodide symporter: impact on thyroid and extrathy-
Physiol Rev 82:473, 2002.
roid pathophysiology. Physiol Rev 80:1083, 2000.
Tomer Y, Davies TF: Searching for the autoimmune thyroid
Dayan CM: Interpretation of thyroid function tests. Lancet
disease susceptibility genes: from gene mapping to gene
357:619, 2001.
function. Endocr Rev 24:694, 2003
Dohan O, De La Vieja A, Paroder V, et al: The sodium/iodide
Vaidya B, Kendall-Taylor P, Pearce SH: The genetics of
Symporter
(NIS): characterization, regulation, and
autoimmune thyroid disease. J Clin Endocrinol Metab
medical significance. Endocr Rev 24:48, 2003.
87:5385, 2002.
Larsen PR, Kronenberg HM, Melmed S, Polonsky KS:
Vasudevan N, Ogawa S, Pfaff D: Estrogen and thyroid
Williams Textbook of Endocrinology, 10th ed. Philadel-
hormone receptor interactions: physiological flexibility by
phia: WB Saunders Co, 2003.
molecular specificity. Physiol Rev 82:923, 2002.
Marino M, McCluskey RT: Role of thyroglobulin endocytic
Yen PM: Physiological and molecular basis of thyroid
pathways in the control of thyroid hormone release. Am J
hormone action. Physiol Rev 81:1097, 2001.
Physiol Cell Physiol 279:C1295, 2000
Zhang J, Lazar MA: The mechanism of action of thyroid hor-
O’Reilly DS: Thyroid function tests—time for a reassess-
mones. Annu Rev Physio 62:439, 2000.
ment. BMJ 320:1332, 2000.
Pearce EN, Farwell AP, Braverman LE: Thyroiditis. N Engl
J Med 348:2646, 2003.
C H A P T E R
7
7
Adrenocortical Hormones
The two adrenal glands, each of which weighs about
4 grams, lie at the superior poles of the two kidneys.
As shown in Figure 77-1, each gland is composed of
two distinct parts, the adrenal medulla and the
adrenal cortex. The adrenal medulla, the central 20
per cent of the gland, is functionally related to the
sympathetic nervous system; it secretes the hor-
mones epinephrine and norepinephrine in response
to sympathetic stimulation. In turn, these hormones cause almost the same
effects as direct stimulation of the sympathetic nerves in all parts of the body.
These hormones and their effects are discussed in detail in Chapter 60 in rela-
tion to the sympathetic nervous system.
The adrenal cortex secretes an entirely different group of hormones, called
corticosteroids. These hormones are all synthesized from the steroid cholesterol,
and they all have similar chemical formulas. However, slight differences in their
molecular structures give them several different but very important functions.
Corticosteroids Mineralocorticoids, Glucocorticoids, and Androgens. Two major types of
adrenocortical hormones, the mineralocorticoids and the glucocorticoids, are
secreted by the adrenal cortex. In addition to these, small amounts of sex hor-
mones are secreted, especially androgenic hormones, which exhibit about the
same effects in the body as the male sex hormone testosterone. They are nor-
mally of only slight importance, although in certain abnormalities of the adrenal
cortices, extreme quantities can be secreted (which is discussed later in the
chapter) and can result in masculinizing effects.
The mineralocorticoids have gained this name because they especially affect
the electrolytes (the “minerals”) of the extracellular fluids-sodium and potas-
sium, in particular. The glucocorticoids have gained their name because they
exhibit important effects that increase blood glucose concentration. They have
additional effects on both protein and fat metabolism that are equally as impor-
tant to body function as their effects on carbohydrate metabolism.
More than 30 steroids have been isolated from the adrenal cortex, but two
are of exceptional importance to the normal endocrine function of the human
body: aldosterone, which is the principal mineralocorticoid, and cortisol, which
is the principal glucocorticoid.
Synthesis and Secretion of
Adrenocortical Hormones
The Adrenal Cortex Has Three Distinct Layers. Figure 77-1 shows that the adrenal
cortex is composed of three relatively distinct layers:
1. The zona glomerulosa, a thin layer of cells that lies just underneath the
capsule, constitutes about 15 per cent of the adrenal cortex. These cells are
the only ones in the adrenal gland capable of secreting significant amounts
of aldosterone because they contain the enzyme aldosterone synthase,
which is necessary for synthesis of aldosterone. The secretion of these cells
is controlled mainly by the extracellular fluid concentrations of angiotensin
II and potassium, both of which stimulate aldosterone secretion.
2. The zona fasciculata, the middle and widest layer, constitutes about 75 per
cent of the adrenal cortex and secretes the glucocorticoids cortisol and
944
Chapter 77
Adrenocortical Hormones
945
(LDL) in the circulating plasma. The LDLs, which have
high concentrations of cholesterol, diffuse from the
plasma into the interstitial fluid and attach to specific
Zona glomerulosa
receptors contained in structures called coated pits on
aldosterone
the adrenocortical cell membranes. The coated pits are
then internalized by endocytosis, forming vesicles that
Zona fasciculata
eventually fuse with cell lysosomes and release choles-
Cortisol
terol that can be used to synthesize adrenal steroid
and
hormones.
androgens
Transport of cholesterol into the adrenal cells is reg-
Zona reticularis
ulated by feedback mechanisms that can markedly alter
the amount available for steroid synthesis. For example,
Medulla
(catecholamines)
Cortex
ACTH, which stimulates adrenal steroid synthesis,
increases the number of adrenocortical cell receptors
for LDL, as well as the activity of enzymes that liberate
cholesterol from LDL.
Once the cholesterol enters the cell, it is delivered to
the mitochondria, where it is cleaved by the enzyme
cholesterol desmolase to form pregnenolone; this is the
rate-limiting step in the eventual formation of adrenal
steroids (Figure 77-2). In all three zones of the adrenal
Magnified section
cortex, this initial step in steroid synthesis is stimulated
by the different factors that control secretion of the
major hormone products aldosterone and cortisol. For
example, both ACTH, which stimulates cortisol secre-
Figure 77-1
tion, and angiotensin II, which stimulates aldosterone
Secretion of adrenocortical hormones by the different zones of the
secretion, increase the conversion of cholesterol to
adrenal cortex and secretion of catecholamines by the adrenal
pregnenolone.
medulla.
Synthetic Pathways for Adrenal Steroids. Figure 77-2 gives
the principal steps in the formation of the important
steroid products of the adrenal cortex: aldosterone, cor-
corticosterone, as well as small amounts of
tisol, and the androgens. Essentially all these steps occur
adrenal androgens and estrogens. The secretion
in two of the organelles of the cell, the mitochondria and
of these cells is controlled in large part by
the endoplasmic reticulum, some steps occurring in one
the hypothalamic-pituitary axis via
of these organelles and some in the other. Each step is
adrenocorticotropic hormone (ACTH).
catalyzed by a specific enzyme system. A change in even
3. The zona reticularis, the deep layer of the
a single enzyme in the schema can cause vastly differ-
cortex, secretes the adrenal androgens
ent types and relative proportions of hormones to be
dehydroepiandrosterone (DHEA) and
formed. For example, very large quantities of masculin-
izing sex hormones or other steroid compounds not
androstenedione, as well as small amounts of
normally present in the blood can occur with altered
estrogens and some glucocorticoids. ACTH also
activity of only one of the enzymes in this pathway.
regulates secretion of these cells, although other
The chemical formulas of aldosterone and cortisol,
factors such as cortical androgen-stimulating
which are the most important mineralocorticoid and
hormone, released from the pituitary, may also be
glucocorticoid hormones, respectively, are shown in
involved. The mechanisms for controlling adrenal
Figure 77-2. Cortisol has a keto-oxygen on carbon
androgen production, however, are not nearly as
number 3 and is hydroxylated at carbon numbers 11 and
well understood as those for glucocorticoids and
21. The mineralocorticoid aldosterone has an oxygen
mineralocorticoids.
atom bound at the number 18 carbon.
Aldosterone and cortisol secretion are regulated by
In addition to aldosterone and cortisol, other steroids
having glucocorticoid or mineralocorticoid activities, or
independent mechanisms. Factors such as angiotensin
both, are normally secreted in small amounts by the
II that specifically increase the output of aldosterone
adrenal cortex. And several additional potent steroid
and cause hypertrophy of the zona glomerulosa have
hormones not normally formed in the adrenal glands
no effect on the other two zones. Similarly, factors such
have been synthesized and are used in various forms of
as ACTH that increase secretion of cortisol and
therapy. Some of the more important of the corticos-
adrenal androgens and cause hypertrophy of the zona
teroid hormones, including the synthetic ones, are the
fasciculata and zona reticularis have little or no effect
following as summarized in Table 77-1:
on the zona glomerulosa.
Mineralocorticoids
Adrenocortical Hormones Are Steroids Derived from Cholesterol.
• Aldosterone (very potent, accounts for about 90 per
All human steroid hormones, including those produced
cent of all mineralocorticoid activity)
by the adrenal cortex, are synthesized from cholesterol.
• Desoxycorticosterone (1/30 as potent as
Although the cells of the adrenal cortex can synthesize
aldosterone, but very small quantities secreted)
de novo small amounts of cholesterol from acetate,
• Corticosterone (slight mineralocorticoid activity)
approximately 80 per cent of the cholesterol used for
• 9a-Fluorocortisol (synthetic, slightly more potent
steroid synthesis is provided by low-density lipoproteins
than aldosterone)
946
Unit XIV Endocrinology and Reproduction
22
21 20
23
18
26
12
17
24
25
11
16
19
13
27
1
C
D
2
9
14
15
10
8
A
B
Cholesterol
7
3
5
HO
4
6
Cholesterol
CH3
CH3
desmolase
(P450 scc)
C O
C O
O
17a-Hydroxylase
17, 20 Lyase
(P450 c17)
OH
(P450 c17)
Pregnenolone
17-Hydroxypregnenolone
Dehydroepl-
HO
HO
HO
androsterone
3b-Hydroxysteroid
CH3
CH3
dehydrogenase
C O
C O
O
17a-Hydroxylase
17, 20 Lyase
(P450 c17)
OH
(P450 c17)
Progesterone
17-Hydroxyprogesterone
Androstenedione
O
O
O
CH2OH
CH2OH
21b-Hydroxylase
(P450 c21)
C O
C O
OH
11-Deoxycorticosterone
11-Deoxycortisol
O
O
11b-Hydroxylase
CH2OH
CH2OH
(P450 c11)
C O
C O
HO
HO
OH
Corticosterone
Cortisol
O
O
Aldosterone
synthase
CH2OH
O
(P450 c11AS)
C O
HC
HO
Aldosterone
O
Figure 77-2
Pathways for synthesis of steroid hormones by the adrenal cortex. The enzymes are shown in italics.
Chapter 77
Adrenocortical Hormones
947
Table 77-1
Adrenal Steroid Hormones in Adults; Synthetic Steroids and Their Relative Glucocorticoid and Mineralocorticoid Activities
Average Plasma
Concentration
(free and bound,
Average Amount
Glucocorticoid
Mineralocorticoid
Steroids
mg/100 ml)
Secreted (mg/24 hr)
Activity
Activity
Adrenal Steroids
Cortisol
12
15
1
1
Corticosterone
0.4
3
0.3
15.0
Aldosterone
0.006
0.15
0.3
3000
Deoxycorticosterone
0.006
0.2
0.2
100
Dehydroepiandrosterone
175
20
—
—
Synthetic Steroids
Cortisone
—
—
1.0
1.0
Prednisolone
—
—
4
0.8
Methylprednisone
—
—
5
—
Dexamethasone
—
—
30
—
9a-fluorocortisol
—
—
10
125
Glucocorticoid and mineralocorticoid activities of the steroids are relative to cortisol, with cortisol being 1.0.
• Cortisol (very slight mineralocorticoid activity, but
Binding of adrenal steroids to the plasma proteins
large quantity secreted)
may serve as a reservoir to lessen rapid fluctuations
• Cortisone (synthetic, slight mineralocorticoid
in free hormone concentrations, as would occur, for
activity)
example, with cortisol during brief periods of stress and
episodic secretion of ACTH. This reservoir function
Glucocorticoids
may also help to ensure a relatively uniform distribu-
• Cortisol (very potent, accounts for about 95 per
tion of the adrenal hormones to the tissues.
cent of all glucocorticoid activity)
• Corticosterone (provides about 4 per cent of total
Adrenocortical Hormones Are Metabolized in the Liver. The
glucocorticoid activity, but much less potent than
adrenal steroids are degraded mainly in the liver and
cortisol)
conjugated especially to glucuronic acid and, to a lesser
• Cortisone (synthetic, almost as potent as cortisol)
extent, sulfates. These substances are inactive and do
• Prednisone (synthetic, four times as potent as
not have mineralocorticoid or glucocorticoid activity.
cortisol)
About 25 per cent of these conjugates are excreted in
• Methylprednisone (synthetic, five times as potent as
the bile and then in the feces. The remaining conjugates
cortisol)
formed by the liver enter the circulation but are not
• Dexamethasone (synthetic, 30 times as potent as
bound to plasma proteins, are highly soluble in the
cortisol)
plasma, and are therefore filtered readily by the kidneys
It is clear from this list that some of these hormones
and excreted in the urine. Diseases of the liver markedly
have both glucocorticoid and mineralocorticoid activi-
depress the rate of inactivation of adrenocortical hor-
ties. It is especially significant that cortisol has a small
mones, and kidney diseases reduce the excretion of the
amount of mineralocorticoid activity, because some syn-
inactive conjugates.
dromes of excess cortisol secretion can cause significant
The normal concentration of aldosterone in blood
mineralocorticoid effects, along with its much more
is about
6 nanograms (6 billionths of a gram) per
potent glucocorticoid effects.
100 ml, and the average secretory rate is approximately
The intense glucocorticoid activity of the synthetic
150 µg/day (0.15 mg/day).
hormone dexamethasone, which has almost zero min-
The concentration of cortisol in the blood averages
eralocorticoid activity, makes this an especially impor-
12 µg/100 ml, and the secretory rate averages 15 to
tant drug for stimulating specific glucocorticoid activity.
20 mg/day.
Adrenocortical Hormones Are Bound to Plasma Proteins.
Approximately 90 to 95 per cent of the cortisol in the
Functions of the
plasma binds to plasma proteins, especially a globulin
called cortisol-binding globulin or transcortin and, to a
Mineralocorticoids-
lesser extent, to albumin. This high degree of binding to
Aldosterone
plasma proteins slows the elimination of cortisol from
the plasma; therefore, cortisol has a relatively long half-
Mineralocorticoid Deficiency Causes Severe Renal Sodium
life of 60 to 90 minutes. Only about 60 per cent of cir-
Chloride Wasting and Hyperkalemia. Total loss of adreno-
culating aldosterone combines with the plasma proteins,
cortical secretion usually causes death within 3 days to
so that about 40 per cent is in the free form; as a result,
2 weeks unless the person receives extensive salt
aldosterone has a relatively short half-life of about 20
minutes. In both the combined and free forms, the hor-
therapy or injection of mineralocorticoids.
mones are transported throughout the extracellular
Without mineralocorticoids, potassium ion concen-
fluid compartment.
tration of the extracellular fluid rises markedly, sodium
948
Unit XIV Endocrinology and Reproduction
and chloride are rapidly lost from the body, and the
concentration stimulate thirst and increased water
total extracellular fluid volume and blood volume
intake, if water is available. Therefore, the extracellu-
become greatly reduced. The person soon develops
lar fluid volume increases almost as much as the
diminished cardiac output, which progresses to a
retained sodium, but without much change in sodium
shocklike state, followed by death. This entire
concentration.
sequence can be prevented by the administration of
Even though aldosterone is one of the body’s most
aldosterone or some other mineralocorticoid. There-
powerful sodium-retaining hormones, only transient
fore, the mineralocorticoids are said to be the acute
sodium retention occurs when excess amounts are
“lifesaving” portion of the adrenocortical hormones.
secreted. An aldosterone-mediated increase in extra-
The glucocorticoids are equally necessary, however,
cellular fluid volume lasting more than 1 to 2 days
allowing the person to resist the destructive effects of
also leads to an increase in arterial pressure, as
life’s intermittent physical and mental “stresses,” as
explained in Chapter 19. The rise in arterial pressure
discussed later in the chapter.
then increases kidney excretion of both salt and
water, called pressure natriuresis and pressure diuresis,
Aldosterone Is the Major Mineralocorticoid Secreted by the
respectively. Thus, after the extracellular fluid volume
Adrenals. Aldosterone exerts nearly 90 per cent of the
increases 5 to 15 per cent above normal, arterial pres-
mineralocorticoid activity of the adrenocortical secre-
sure also increases 15 to 25 mm Hg, and this elevated
tions, but cortisol, the major glucocorticoid secreted by
blood pressure returns the renal output of salt and
the adrenal cortex, also provides a significant amount
water to normal despite the excess aldosterone (Figure
of mineralocorticoid activity. Aldosterone’s mineralo-
77-3).
corticoid activity is about 3000 times greater than that
This return to normal of salt and water excretion by
of cortisol, but the plasma concentration of cortisol is
the kidneys
as
a result of pressure natriuresis and
nearly 2000 times that of aldosterone.
Renal and Circulatory Effects
Aldosterone
of Aldosterone
120
Aldosterone Increases Renal Tubular Reabsorption of Sodium
and Secretion of Potassium. It will be recalled from
100
Chapter
27 that aldosterone increases absorption
of sodium and simultaneously increases secretion
of potassium by the renal tubular epithelial cells-
80
especially in the principal cells of the collecting tubules
and, to a lesser extent, in the distal tubules and col-
120
lecting ducts. Therefore, aldosterone causes sodium to
be conserved in the extracellular fluid while increasing
110
potassium excretion in the urine.
A high concentration of aldosterone in the plasma
100
can transiently decrease the sodium loss into the urine
to as little as a few milliequivalents a day. At the same
90
time, potassium loss into the urine increases several-
400
fold. Therefore, the net effect of excess aldosterone
in the plasma is to increase the total quantity of
300
sodium in the extracellular fluid while decreasing the
potassium.
200
Conversely, total lack of aldosterone secretion can
cause transient loss of 10 to 20 grams of sodium in the
100
urine a day, an amount equal to one tenth to one fifth
-4
-2
0
2
4
6
8
10
12
14
of all the sodium in the body. At the same time, potas-
Time (days)
sium is conserved tenaciously in the extracellular fluid.
Excess Aldosterone Increases Extracellular Fluid Volume and
Arterial Pressure but Has Only a Small Effect on Plasma Sodium
Figure 77-3
Concentration. Although aldosterone has a potent
Effect of aldosterone infusion on arterial pressure, extracellular
effect in decreasing the rate of sodium ion excretion
fluid volume, and sodium excretion in dogs. Although aldosterone
by the kidneys, the concentration of sodium in the
was infused at a rate that raised plasma concentrations to about
extracellular fluid often rises only a few milliequiva-
20 times normal, note the “escape” from sodium retention on the
second day of infusion as arterial pressure increased and urinary
lents. The reason for this is that when sodium is reab-
sodium excretion returned to normal. (Drawn from data in Hall JE,
sorbed by the tubules, there is simultaneous osmotic
Granger JP, Smith MJ Jr, Premen N: Role of hemodynamics and
absorption of almost equivalent amounts of water.
arterial pressure in aldosterone “escape.” Hypertension 6 (suppl
Also, small increases in extracellular fluid sodium
I):I-183-I-192, 1984.)
Chapter 77
Adrenocortical Hormones
949
diuresis is called aldosterone escape. Thereafter, the
tubules. Both these glands form a primary secretion
rate of gain of salt and water by the body is zero, and
that contains large quantities of sodium chloride, but
balance is maintained between salt and water intake
much of the sodium chloride, on passing through the
and output by the kidneys despite continued excess
excretory ducts, is reabsorbed, whereas potassium and
aldosterone. In the meantime, however, the person has
bicarbonate ions are secreted. Aldosterone greatly
developed hypertension, which lasts as long as the
increases the reabsorption of sodium chloride and the
person remains exposed to high levels of aldosterone.
secretion of potassium by the ducts. The effect on the
Conversely, when aldosterone secretion becomes
sweat glands is important to conserve body salt in hot
zero, large amounts of salt are lost in the urine, not
environments, and the effect on the salivary glands is
only diminishing the amount of sodium chloride in the
necessary to conserve salt when excessive quantities of
extracellular fluid but also decreasing the extracellular
saliva are lost.
fluid volume. The result is severe extracellular fluid
Aldosterone also greatly enhances sodium absorp-
dehydration and low blood volume, leading to circula-
tion by the intestines, especially in the colon, which
tory shock. Without therapy, this usually causes death
prevents loss of sodium in the stools. Conversely, in the
within a few days after the adrenal glands suddenly
absence of aldosterone, sodium absorption can be
stop secreting aldosterone.
poor, leading to failure to absorb chloride and other
anions and water as well. The unabsorbed sodium
chloride and water then lead to diarrhea, with further
Excess Aldosterone Causes Hypokalemia and Muscle Weak-
loss of salt from the body.
ness; Too Little Aldosterone Causes Hyperkalemia and Cardiac
Toxicity. Excess aldosterone not only causes loss of
potassium ions from the extracellular fluid into the
urine but also stimulates transport of potassium
Cellular Mechanism of
from the extracellular fluid into most cells of the
Aldosterone Action
body. Therefore, excessive secretion of aldosterone,
as occurs with some types of adrenal tumors, may
Although for many years we have known the overall
cause a serious decrease in the plasma potassium
effects of mineralocorticoids on the body, the basic
concentration, sometimes from the normal value of
action of aldosterone on the tubular cells to increase
4.5 mEq/L to as low as 2 mEq/L. This condition is
transport of sodium is still not fully understood.
called hypokalemia. When the potassium ion concen-
However, the cellular sequence of events that leads
tration falls below about one-half normal, severe
to increased sodium reabsorption seems to be the
muscle weakness often develops. This is caused by
following.
alteration of the electrical excitability of the nerve and
First, because of its lipid solubility in the cellular
muscle fiber membranes (see Chapter 5), which pre-
membranes, aldosterone diffuses readily to the interior
vents transmission of normal action potentials.
of the tubular epithelial cells.
Conversely, when aldosterone is deficient, the extra-
Second, in the cytoplasm of the tubular cells, aldos-
cellular fluid potassium ion concentration can rise far
terone combines with a highly specific cytoplasmic
above normal. When it rises to 60 to 100 per cent above
receptor protein, a protein that has a stereomolecular
normal, serious cardiac toxicity, including weakness of
configuration that allows only aldosterone or very
heart contraction and development of arrhythmia,
similar compounds to combine with it.
becomes evident; progressively higher concentrations
Third, the aldosterone-receptor complex or a
of potassium lead inevitably to heart failure.
product of this complex diffuses into the nucleus,
where it may undergo further alterations, finally induc-
Excess Aldosterone Increases Tubular Hydrogen Ion Secretion,
ing one or more specific portions of the DNA to form
and Causes Mild Alkalosis. Aldosterone not only causes
one or more types of messenger RNA related to the
potassium to be secreted into the tubules in exchange
process of sodium and potassium transport.
for sodium reabsorption in the principal cells of the
Fourth, the messenger RNA diffuses back into the
renal collecting tubules but also causes secretion of
cytoplasm, where, operating in conjunction with the
hydrogen ions in exchange for sodium in the interca-
ribosomes, it causes protein formation. The proteins
lated cells of the cortical collecting tubules. This
formed are a mixture of (1) one or more enzymes
decreases the hydrogen ion concentration in the extra-
and
(2) membrane transport proteins that, all
cellular fluid, causing a mild degree of alkalosis.
acting together, are required for sodium, potassium,
and hydrogen transport through the cell membrane.
One of the enzymes especially increased is sodium-
potassium adenosine triphosphatase, which serves as
Aldosterone Stimulates Sodium and
the principal part of the pump for sodium and potas-
Potassium Transport in Sweat Glands,
sium exchange at the basolateral membranes of the
Salivary Glands, and Intestinal
renal tubular cells. Additional proteins, perhaps
Epithelial Cells
equally important, are epithelial sodium channel pro-
teins inserted into the luminal membrane of the same
Aldosterone has almost the same effects on sweat
tubular cells that allows rapid diffusion of sodium ions
glands and salivary glands as it has on the renal
from the tubular lumen into the cell; then the sodium
950
Unit XIV Endocrinology and Reproduction
is pumped the rest of the way by the sodium-potassium
1. Increased potassium ion concentration in the
pump located in the basolateral membranes of the cell.
extracellular fluid greatly increases aldosterone
Thus, aldosterone does not have an immediate effect
secretion.
on sodium transport; rather, this effect must await the
2. Increased activity of the renin-angiotensin system
sequence of events that leads to the formation of the
(increased levels of angiotensin II) also greatly
specific intracellular substances required for sodium
increases aldosterone secretion.
transport. About 30 minutes is required before new
3. Increased sodium ion concentration in the
RNA appears in the cells, and about 45 minutes is
extracellular fluid very slightly decreases
required before the rate of sodium transport begins to
aldosterone secretion.
increase; the effect reaches maximum only after
4. ACTH from the anterior pituitary gland is
several hours.
necessary for aldosterone secretion but has little
effect in controlling the rate of secretion.
Of these factors, potassium ion concentration
and the renin-angiotensin system are by far the most
Possible Nongenomic Actions
potent in regulating aldosterone secretion. A small
of Aldosterone and Other
percentage increase in potassium concentration can
Steroid Hormones
cause a severalfold increase in aldosterone secretion.
Likewise, activation of the renin-angiotensin system,
Recent studies suggest that many steroids, including
usually in response to diminished blood flow to the
aldosterone, elicit not only slowly developing genomic
kidneys or to sodium loss, can cause a severalfold
effects that have a latency of 60 to 90 minutes and
increase in aldosterone secretion. In turn, the aldos-
require gene transcription and synthesis of new pro-
terone acts on the kidneys (1) to help them excrete the
teins, but also rapid nongenomic effects that take place
excess potassium ions and (2) to increase the blood
in a few seconds or minutes.
volume and arterial pressure, thus returning the renin-
These nongenomic actions are believed to be medi-
angiotensin system toward its normal level of activity.
ated by binding of steroids to cell membrane receptors
These feedback control mechanisms are essential for
that are coupled to second messenger systems, similar
maintaining life, and the reader is referred again to
to those used for peptide hormone signal transduction.
Chapters 27 and 29 for a full understanding of their
For example, aldosterone has been shown to increase
functions.
formation of cAMP in vascular smooth muscle cells
Figure 77-4 shows the effects on plasma aldosterone
and in epithelial cells of the renal collecting tubules in
concentration caused by blocking the formation of
less than two minutes, a time period that is far too
angiotensin II with an angiotensin-converting enzyme
short for gene transcription and synthesis of new pro-
inhibitor after several weeks of a low-sodium diet that
teins. In other cell types, aldosterone has been shown
increases plasma aldosterone concentration several-
to rapidly stimulate the phosphatidylinositol second
fold. Note that blocking angiotensin II formation
messenger system. However, the precise structure of
markedly decreases plasma aldosterone concentration
receptors responsible for the rapid effects of aldos-
without significantly changing cortisol concentration;
terone has not been determined, nor is the physiolog-
this indicates the important role of angiotensin II in
ical significance of these nongenomic actions of
stimulating aldosterone secretion when sodium intake
steroids well understood.
and extracellular fluid volume are reduced.
By contrast, the effects of sodium ion concentration
per se and of ACTH in controlling aldosterone secre-
Regulation of Aldosterone Secretion
tion are usually minor. Nevertheless, a 10 to 20 per cent
decrease in extracellular fluid sodium ion concentra-
The regulation of aldosterone secretion is so deeply
tion, which occurs on rare occasions, can perhaps
intertwined with the regulation of extracellular fluid
double aldosterone secretion. In the case of ACTH, if
electrolyte concentrations, extracellular fluid volume,
there is even a small amount of ACTH secreted by the
blood volume, arterial pressure, and many special
anterior pituitary gland, it is usually enough to permit
aspects of renal function that it is difficult to discuss
the adrenal glands to secrete whatever amount of
the regulation of aldosterone secretion independently
aldosterone is required, but total absence of ACTH
of all these other factors. This subject is presented in
can significantly reduce aldosterone secretion.
detail in Chapters 28 and 29, to which the reader is
referred. However, it is important to list here some of
the more important points of aldosterone secretion
Functions of the
control.
The regulation of aldosterone secretion by the zona
Glucocorticoids
glomerulosa cells is almost entirely independent of the
regulation of cortisol and androgens by the zona fas-
Even though mineralocorticoids can save the life of
ciculata and zona reticularis.
an acutely adrenalectomized animal, the animal still
Four factors are known to play essential roles in the
is far from normal. Instead, its metabolic systems
regulation of aldosterone. In the probable order of
for utilization of proteins, carbohydrates, and fats
their importance, they are as follows:
remain considerably deranged. Furthermore, the
Chapter 77
Adrenocortical Hormones
951
the rate of gluconeogenesis as much as 6- to 10-fold.
This results mainly from two effects of cortisol.
50
1. Cortisol increases the enzymes required to convert
amino acids into glucose in the liver cells. This
results from the effect of the glucocorticoids to
40
activate DNA transcription in the liver cell nuclei
in the same way that aldosterone functions in the
renal tubular cells, with formation of messenger
30
RNAs that in turn lead to the array of enzymes
required for gluconeogenesis.
2. Cortisol causes mobilization of amino acids from
20
the extrahepatic tissues mainly from muscle. As a
result, more amino acids become available in the
3.0
plasma to enter into the gluconeogenesis process
of the liver and thereby to promote the formation
of glucose.
2.0
One of the effects of increased gluconeogenesis is a
marked increase in glycogen storage in the liver cells.
This effect of cortisol allows other glycolytic hor-
1.0
mones, such as epinephrine and glucagon, to mobilize
glucose in times of need, such as between meals.
0.0
Control
ACE
ACE inhibitor
inhibitor
+
Decreased Glucose Utilization by Cells. Cortisol also causes
Ang II infusion
a moderate decrease in the rate of glucose utilization
by most cells in the body. Although the cause of this
decrease is unknown, most physiologists believe that
somewhere between the point of entry of glucose
Figure 77-4
into the cells and its final degradation, cortisol directly
delays the rate of glucose utilization. A suggested
Effects of treating
sodium-depleted dogs with an angiotensin-
converting enzyme (ACE) inhibitor for 7 days to block formation
mechanism is based on the observation that glucocor-
of angiotensin II (Ang II) and of infusing exogenous Ang II to
ticoids depress the oxidation of nicotinamide-adenine
restore plasma Ang II levels after ACE inhibition. Note that block-
dinucleotide
(NADH) to form NAD+. Because
ing Ang II formation reduced plasma aldosterone concentration
NADH must be oxidized to allow glycolysis, this effect
with little effect on cortisol, demonstrating the important role of
Ang II in stimulating aldosterone secretion during sodium deple-
could account for the diminished utilization of glucose
tion.
(Drawn from data in Hall JE, Guyton AC, Smith MJ Jr,
by the cells.
Coleman TG: Chronic blockade of angiotensin II formation during
sodium deprivation. Am J Physiol 237:F424, 1979.)
Elevated Blood Glucose Concentration and “Adrenal Diabetes.”
Both the increased rate of gluconeogenesis and the
moderate reduction in the rate of glucose utilization
by the cells cause the blood glucose concentrations to
animal cannot resist different types of physical or even
rise. The rise in blood glucose in turn stimulates
mental stress, and minor illnesses such as respiratory
secretion of insulin. The increased plasma levels of
tract infections can lead to death. Therefore, the glu-
insulin, however, are not as effective in maintaining
cocorticoids have functions just as important to the
plasma glucose as they are under normal conditions.
long-continued life of the animal as those of the min-
For reasons that are not entirely clear, high levels
eralocorticoids. They are explained in the following
of glucocorticoid reduce the sensitivity of many
sections.
tissues, especially skeletal muscle and adipose tissue,
At least 95 per cent of the glucocorticoid activity of
to the stimulatory effects of insulin on glucose uptake
the adrenocortical secretions results from the secre-
and utilization. One possible explanation is that high
tion of cortisol, known also as hydrocortisone. In
levels of fatty acids, caused by the effect of glucocor-
addition to this, a small but significant amount of glu-
ticoids to mobilize lipids from fat depots, may impair
cocorticoid activity is provided by corticosterone.
insulin’s actions on the tissues. In this way, excess
secretion of glucocorticoids may produce disturbances
of carbohydrate metabolism very similar to those
Effects of Cortisol on
found in patients with excess levels of growth
Carbohydrate Metabolism
hormone.
The increase in blood glucose concentration is occa-
Stimulation of Gluconeogenesis. By far the best-known
sionally great enough (50 per cent or more above
metabolic effect of cortisol and other glucocorticoids
normal) that the condition is called adrenal diabetes.
on metabolism is their ability to stimulate gluconeo-
Administration of insulin lowers the blood glucose
genesis (formation of carbohydrate from proteins and
concentration only a moderate amount in adrenal
some other substances) by the liver, often increasing
diabetes-not nearly as much as it does in pancreatic
952
Unit XIV Endocrinology and Reproduction
diabetes-because the tissues are resistant to the effects
gluconeogenesis. Thus, it is possible that many of the
of insulin.
effects of cortisol on the metabolic systems of the body
result mainly from this ability of cortisol to mobilize
amino acids from the peripheral tissues while at the
Effects of Cortisol on
same time increasing the liver enzymes required for
Protein Metabolism
the hepatic effects.
Reduction in Cellular Protein. One of the principal effects
of cortisol on the metabolic systems of the body is
Effects of Cortisol on Fat Metabolism
reduction of the protein stores in essentially all body
cells except those of the liver. This is caused by both
Mobilization of Fatty Acids. In much the same manner
decreased protein synthesis and increased catabolism
that cortisol promotes amino acid mobilization from
of protein already in the cells. Both these effects may
muscle, it promotes mobilization of fatty acids from
result from decreased amino acid transport into extra-
adipose tissue. This increases the concentration of free
hepatic tissues, as discussed later; this probably is not
fatty acids in the plasma, which also increases their uti-
the major cause, because cortisol also depresses the
lization for energy. Cortisol also seems to have a direct
formation of RNA and subsequent protein synthesis
effect to enhance the oxidation of fatty acids in the
in many extrahepatic tissues, especially in muscle and
cells.
lymphoid tissue.
The mechanism by which cortisol promotes fatty
In the presence of great excesses of cortisol, the
acid mobilization is not completely understood.
muscles can become so weak that the person cannot
However, part of the effect probably results from
rise from the squatting position. And the immunity
diminished transport of glucose into the fat cells.
functions of the lymphoid tissue can be decreased to a
Recall that a-glycerophosphate, which is derived from
small fraction of normal.
glucose, is required for both deposition and mainte-
nance of triglycerides in these cells, and in its absence
Cortisol Increases Liver and Plasma Proteins. Coinciden-
the fat cells begin to release fatty acids.
tally with the reduced proteins elsewhere in the body,
The increased mobilization of fats by cortisol, com-
the liver proteins become enhanced. Furthermore, the
bined with increased oxidation of fatty acids in the
plasma proteins (which are produced by the liver and
cells, helps shift the metabolic systems of the cells from
then released into the blood) are also increased. These
utilization of glucose for energy to utilization of fatty
increases are exceptions to the protein depletion that
acids in times of starvation or other stresses. This cor-
occurs elsewhere in the body. It is believed that this
tisol mechanism, however, requires several hours to
difference results from a possible effect of cortisol to
become fully developed-not nearly so rapid or so pow-
enhance amino acid transport into liver cells (but
erful an effect as a similar shift elicited by a decrease
not into most other cells) and to enhance the
in insulin, as we discuss in Chapter 78. Nevertheless,
liver enzymes required for protein synthesis.
the increased use of fatty acids for metabolic energy is
an important factor for long-term conservation of
Increased Blood Amino Acids, Diminished Transport of Amino
body glucose and glycogen.
Acids into Extrahepatic Cells, and Enhanced Transport into
Hepatic Cells. Studies in isolated tissues have demon-
strated that cortisol depresses amino acid transport
Obesity Caused by Excess Cortisol. Despite the fact that
into muscle cells and perhaps into other extrahepatic
cortisol can cause a moderate degree of fatty acid
cells.
mobilization from adipose tissue, many people with
The decreased transport of amino acids into extra-
excess cortisol secretion develop a peculiar type of
hepatic cells decreases their intracellular amino acid
obesity, with excess deposition of fat in the chest and
concentrations and consequently decreases the syn-
head regions of the body, giving a buffalo-like torso
thesis of protein. Yet, catabolism of proteins in the cells
and a rounded “moon face.” Although the cause is
continues to release amino acids from the already
unknown, it has been suggested that this obesity
existing proteins, and these diffuse out of the cells to
results from excess stimulation of food intake, with fat
increase the plasma amino acid concentration. There-
being generated in some tissues of the body more
fore, cortisol mobilizes amino acids from the nonhep-
rapidly than it is mobilized and oxidized.
atic tissues and in doing so diminishes the tissue stores
of protein.
The increased plasma concentration of amino acids
and enhanced transport of amino acids into the
Cortisol is Important in Resisting
hepatic cells by cortisol could also account for
Stress and Inflammation
enhanced utilization of amino acids by the liver to
cause such effects as (1) increased rate of deamination
Almost any type of stress, whether physical or neuro-
of amino acids by the liver, (2) increased protein
genic, causes an immediate and marked increase
synthesis in the liver,
(3) increased formation of
in ACTH secretion by the anterior pituitary gland,
plasma proteins by the liver, and (4) increased con-
followed within minutes by greatly increased adreno-
version of amino acids to glucose-that is, enhanced
cortical secretion of cortisol. This is demonstrated
Chapter 77
Adrenocortical Hormones
953
pyrimidines, and creatine phosphate, which are neces-
sary for maintenance of cellular life and reproduction
of new cells.
45
40
But all this is mainly supposition. It is supported
35
only by the fact that cortisol usually does not mobilize
30
the basic functional proteins of the cells, such as
25
the muscle contractile proteins and the proteins of
20
neurons, until almost all other proteins have been
15
released. This preferential effect of cortisol in
10
5
mobilizing labile proteins could make amino acids
55
available to needy cells to synthesize substances essen-
50
tial to life.
45
40
Anti-inflammatory Effects of High Levels
35
of Cortisol
30
25
When tissues are damaged by trauma, by infection
20
with bacteria, or in other ways, they almost always
15
become “inflamed.” In some conditions, such as in
10
rheumatoid arthritis, the inflammation is more dam-
5
aging than the trauma or disease itself. The adminis-
-0
15
30
45 60
90 2
3
4 56
8 101215202530
tration of large amounts of cortisol can usually block
Seconds
Minutes
this inflammation or even reverse many of its effects
once it has begun. Before attempting to explain the
way in which cortisol functions to block inflammation,
Figure 77-5
let us review the basic steps in the inflammation
process, discussed in more detail in Chapter 33.
Rapid reaction of the adrenal cortex of a rat to stress caused by
There are five main stages of inflammation: (1)
fracture of the tibia and fibula at time zero. (In the rat, corticos-
release from the damaged tissue cells of chemical
terone is secreted in place of cortisol.) (Courtesy Drs. Guillemin,
substances that activate the inflammation process-
Dear, and Lipscomb.)
chemicals such as histamine, bradykinin, proteolytic
enzymes, prostaglandins, and leukotrienes;
(2) an
increase in blood flow in the inflamed area caused by
dramatically by the experiment shown in Figure 77-5,
some of the released products from the tissues, an
in which corticosteroid formation and secretion
effect called erythema; (3) leakage of large quantities
increased sixfold in a rat within 4 to 20 minutes after
of almost pure plasma out of the capillaries into the
fracture of two leg bones.
damaged areas because of increased capillary perme-
Some of the different types of stress that increase
ability, followed by clotting of the tissue fluid, thus
cortisol release are the following:
causing a nonpitting type of edema; (4) infiltration of
1. Trauma of almost any type
the area by leukocytes; and (5) after days or weeks,
2. Infection
ingrowth of fibrous tissue that often helps in the
3. Intense heat or cold
healing process.
4. Injection of norepinephrine and other
When large amounts of cortisol are secreted or
sympathomimetic drugs
injected into a person, the cortisol has two basic anti-
5. Surgery
inflammatory effects: (1) it can block the early stages
6. Injection of necrotizing substances beneath the
of the inflammation process before inflammation
skin
even begins, or (2) if inflammation has already begun,
7. Restraining an animal so that it cannot move
it causes rapid resolution of the inflammation and
8. Almost any debilitating disease
increased rapidity of healing. These effects are
Even though we know that cortisol secretion often
explained further as follows.
increases greatly in stressful situations, we are not sure
why this is of significant benefit to the animal. One
Cortisol Prevents the Development of Inflammation by Stabiliz-
possibility is that the glucocorticoids cause rapid mobi-
ing Lysosomes and by Other Effects. Cortisol has the fol-
lization of amino acids and fats from their cellular
lowing effects in preventing inflammation:
stores, making them immediately available both for
1. Cortisol stabilizes the lysosomal membranes. This
energy and for synthesis of other compounds, includ-
is one of its most important anti-inflammatory
ing glucose, needed by the different tissues of the body.
effects, because it is much more difficult than
Indeed, it has been shown in a few instances that
normal for the membranes of the intracellular
damaged tissues that are momentarily depleted of pro-
lysosomes to rupture. Therefore, most of the
teins can use the newly available amino acids to form
proteolytic enzymes that are released by damaged
new proteins that are essential to the lives of the cells.
cells to cause inflammation, which are mainly
Also, the amino acids are perhaps used to synthesize
stored in the lysosomes, are released in greatly
other essential intracellular substances such as purines,
decreased quantity.
954
Unit XIV Endocrinology and Reproduction
2. Cortisol decreases the permeability of the
And even though the cortisol does not correct the
capillaries, probably as a secondary effect of the
basic disease condition, merely preventing the damag-
reduced release of proteolytic enzymes. This
ing effects of the inflammatory response, this alone can
prevents loss of plasma into the tissues.
often be a lifesaving measure.
3. Cortisol decreases both migration of white blood
cells into the inflamed area and phagocytosis of the
Other Effects of Cortisol
damaged cells. These effects probably result from
the fact that cortisol diminishes the formation
Cortisol Blocks the Inflammatory Response to Allergic Reactions.
of prostaglandins and leukotrienes that
The basic allergic reaction between antigen and anti-
otherwise would increase vasodilation, capillary
body is not affected by cortisol, and even some of the
permeability, and mobility of white blood cells.
secondary effects of the allergic reaction still occur.
4. Cortisol suppresses the immune system, causing
However, because the inflammatory response is respon-
lymphocyte reproduction to decrease markedly.
sible for many of the serious and sometimes lethal
The T lymphocytes are especially suppressed. In
effects of allergic reactions, administration of cortisol,
followed by its effect in reducing inflammation and the
turn, reduced amounts of T cells and antibodies
release of inflammatory products, can be lifesaving. For
in the inflamed area lessen the tissue reactions
instance, cortisol effectively prevents shock or death in
that would otherwise promote the inflammation
anaphylaxis, which otherwise kills many people, as
process.
explained in Chapter 34.
5. Cortisol attenuates fever mainly because it reduces
the release of interleukin-1 from the white blood
Effect on Blood Cells and on Immunity in Infectious Diseases.
cells, which is one of the principal excitants to the
Cortisol decreases the number of eosinophils and lym-
hypothalamic temperature control system. The
phocytes in the blood; this effect begins within a few
decreased temperature in turn reduces the degree
minutes after the injection of cortisol and becomes
of vasodilation.
marked within a few hours. Indeed, a finding of lym-
phocytopenia or eosinopenia is an important diagnostic
Thus, cortisol has an almost global effect in reduc-
criterion for overproduction of cortisol by the adrenal
ing all aspects of the inflammatory process. How much
gland.
of this results from the simple effect of cortisol in
Likewise, the administration of large doses of cortisol
stabilizing lysosomal and cell membranes versus its
causes significant atrophy of all the lymphoid tissue
effect to reduce the formation of prostaglandins and
throughout the body, which in turn decreases the output
leukotrienes from arachidonic acid in damaged cell
of both T cells and antibodies from the lymphoid tissue.
membranes and other effects of cortisol is unclear.
As a result, the level of immunity for almost all foreign
invaders of the body is decreased. This occasionally can
Cortisol Causes Resolution of Inflammation. Even after
lead to fulminating infection and death from diseases
inflammation has become well established, the admin-
that would otherwise not be lethal, such as fulminating
tuberculosis in a person whose disease had previously
istration of cortisol can often reduce inflammation
been arrested. Conversely, this ability of cortisol and
within hours to a few days. The immediate effect is to
other glucocorticoids to suppress immunity makes them
block most of the factors that are promoting the
useful drugs in preventing immunological rejection of
inflammation. But in addition, the rate of healing is
transplanted hearts, kidneys, and other tissues.
enhanced. This probably results from the same, mainly
Cortisol increases the production of red blood cells
undefined, factors that allow the body to resist many
by mechanisms that are unclear. When excess cortisol is
other types of physical stress when large quantities of
secreted by the adrenal glands, polycythemia often
cortisol are secreted. Perhaps this results from the
results, and conversely, when the adrenal glands secrete
mobilization of amino acids and use of these to repair
no cortisol, anemia often results.
the damaged tissues; perhaps it results from the
increased glucogenesis that makes extra glucose avail-
Cellular Mechanism of
able in critical metabolic systems; perhaps it results
Cortisol Action
from increased amounts of fatty acids available for cel-
lular energy; or perhaps it depends on some effect of
Cortisol, like other steroid hormones, exerts its effects
cortisol for inactivating or removing inflammatory
by first interacting with intracellular receptors in target
products.
cells. Because cortisol is lipid soluble, it can easily
Regardless of the precise mechanisms by which the
diffuse through the cell membrane. Once inside the cell,
anti-inflammatory effect occurs, this effect of cortisol
cortisol binds with its protein receptor in the cytoplasm,
plays a major role in combating certain types of dis-
and the hormone-receptor complex then interacts with
eases, such as rheumatoid arthritis, rheumatic fever,
specific regulatory DNA sequences, called glucocorti-
coid response elements, to induce or repress gene tran-
and acute glomerulonephritis. All these diseases are
scription. Other proteins in the cell, called transcription
characterized by severe local inflammation, and the
factors, are also necessary for the hormone-receptor
harmful effects on the body are caused mainly by
complex to interact appropriately with the glucocorti-
the inflammation itself and not by other aspects of the
coid response elements.
disease.
Glucocorticoids increase or decrease transcription of
When cortisol or other glucocorticoids are adminis-
many genes to alter synthesis of mRNA for the proteins
tered to patients with these diseases, almost invariably
that mediate their multiple physiologic effects. Thus,
the inflammation begins to subside within 24 hours.
most of the metabolic effects of cortisol are not
Chapter 77
Adrenocortical Hormones
955
immediate but require 45 to 60 minutes for proteins to
This is another example of cAMP as a second messen-
be synthesized, and up to several hours or days to fully
ger signal system.
develop. Recent evidence suggests that glucocorticoids,
The most important of all the ACTH-stimulated
especially at high concentrations, may also have some
steps for controlling adrenocortical secretion is acti-
rapid nongenomic effects on cell membrane ion trans-
vation of the enzyme protein kinase A, which causes
port that may contribute to their therapeutic benefits.
initial conversion of cholesterol to pregnenolone. This
initial conversion is the “rate-limiting” step for all the
adrenocortical hormones, which explains why ACTH
Regulation of Cortisol Secretion
normally is necessary for any adrenocortical hormones
by Adrenocorticotropic Hormone
to be formed. Long-term stimulation of the adrenal
from the Pituitary Gland
cortex by ACTH not only increases secretory activity
but also causes hypertrophy and proliferation of the
ACTH Stimulates Cortisol Secretion. Unlike aldosterone
adrenocortical cells, especially in the zona fasciculata
secretion by the zona glomerulosa, which is controlled
and zona reticularis, where cortisol and the androgens
mainly by potassium and angiotensin acting directly
are secreted.
on the adrenocortical cells, almost no stimuli have
direct control effects on the adrenal cells that secrete
Physiologic Stress Increases ACTH and
cortisol. Instead, secretion of cortisol is controlled
Adrenocortical Secretion
almost entirely by ACTH secreted by the anterior
As pointed out earlier in the chapter, almost any type
pituitary gland. This hormone, also called corticotropin
of physical or mental stress can lead within minutes
or adrenocorticotropin, also enhances the production
to greatly enhanced secretion of ACTH and conse-
of adrenal androgens.
quently cortisol as well, often increasing cortisol secre-
tion as much as 20-fold. This effect was demonstrated
by the rapid and strong adrenocortical secretory
Chemistry of ACTH. ACTH has been isolated in pure
responses after trauma shown in Figure 77-5.
form from the anterior pituitary. It is a large polypep-
Pain stimuli caused by physical stress or tissue
tide, having a chain length of 39 amino acids. A smaller
damage are transmitted first upward through the brain
polypeptide, a digested product of ACTH having a
stem and eventually to the median eminence of the
chain length of 24 amino acids, has all the effects of the
hypothalamus, as shown in Figure 77-6. Here CRF is
total molecule.
secreted into the hypophysial portal system. Within
minutes the entire control sequence leads to large
ACTH Secretion Is Controlled by Corticotropin-Releasing Factor
quantities of cortisol in the blood.
from the Hypothalamus. In the same way that other pitu-
Mental stress can cause an equally rapid increase in
itary hormones are controlled by releasing factors
ACTH secretion. This is believed to result from
from the hypothalamus, an important releasing factor
increased activity in the limbic system, especially in the
also controls ACTH secretion. This is called corti-
region of the amygdala and hippocampus, both of
cotropin-releasing factor (CRF). It is secreted into the
which then transmit signals to the posterior medial
primary capillary plexus of the hypophysial portal
hypothalamus.
system in the median eminence of the hypothalamus
and then carried to the anterior pituitary gland, where
Inhibitory Effect of Cortisol on the Hypothalamus and on the
it induces ACTH secretion. CRF is a peptide com-
Anterior Pituitary to Decrease ACTH Secretion. Cortisol has
posed of 41 amino acids. The cell bodies of the neurons
direct negative feedback effects on (1) the hypothala-
that secrete CRF are located mainly in the paraven-
mus to decrease the formation of CRF and (2) the
tricular nucleus of the hypothalamus. This nucleus in
anterior pituitary gland to decrease the formation
turn receives many nervous connections from the
of ACTH. Both of these feedbacks help regulate
limbic system and lower brain stem.
the plasma concentration of cortisol. That is, whenever
The anterior pituitary gland can secrete only minute
the cortisol concentration becomes too great, the
quantities of ACTH in the absence of CRF. Instead,
feedbacks automatically reduce the ACTH toward
most conditions that cause high ACTH secretory rates
a normal control level.
initiate this secretion by signals that begin in the basal
regions of the brain, including the hypothalamus, and
Summary of the Cortisol Control System
are then transmitted by CRF to the anterior pituitary
Figure 77-6 shows the overall system for control of
gland.
cortisol secretion. The key to this control is the exci-
tation of the hypothalamus by different types of stress.
ACTH Activates Adrenocortical Cells to Produce Steroids by
Stress stimuli activate the entire system to cause rapid
Increasing Cyclic Adenosine Monophosphate
(cAMP). The
release of cortisol, and the cortisol in turn initiates a
principal effect of ACTH on the adrenocortical cells is
series of metabolic effects directed toward relieving
to activate adenylyl cyclase in the cell membrane. This
the damaging nature of the stressful state.
then induces the formation of cAMP in the cell cyto-
There is also direct feedback of the cortisol to both
plasm, reaching its maximal effect in about 3 minutes.
the hypothalamus and the anterior pituitary gland to
The cAMP in turn activates the intracellular enzymes
decrease the concentration of cortisol in the plasma
that cause formation of the adrenocortical hormones.
at times when the body is not experiencing stress.
956
Unit XIV Endocrinology and Reproduction
Hypothalamus
Excites
Stress
20
Portal
Median
15
eminence
vessel
(CRF)
10
Relieves
Inhibits
5
ACTH
0
12:00
4:00
8:00
12:00
4:00
8:00
12:00
Adrenal
AM
PM
cortex
Cortisol
Noon
1 Gluconeogenesis
Figure 77-7
2 Protein mobilization
3 Fat mobilization
Typical pattern of cortisol concentration during the day. Note the
4 Stabilizes lysosomes
oscillations in secretion as well as a daily secretory surge an hour
or so after awaking in the morning.
Figure 77-6
Mechanism for regulation of glucocorticoid secretion. ACTH,
stimulating hormone (MSH), b-lipotropin, b-endor-
adrenocorticotropic hormone; CRF, corticotropin-releasing factor.
phin, and a few others (Figure 77-8). Under normal
conditions, none of these hormones is secreted in
enough quantity by the pituitary to have a significant
effect on the human body, but when the rate of secre-
However, the stress stimuli are the prepotent ones;
tion of ACTH is high, as may occur in Addison’s
they can always break through this direct inhibitory
disease, formation of some of the other POMC-
feedback of cortisol, causing either periodic exacerba-
derived hormones may also be increased.
tions of cortisol secretion at multiple times during the
The POMC gene is actively transcribed in several
day (Figure 77-7) or prolonged cortisol secretion in
tissues, including the corticotroph cells of the anterior
times of chronic stress.
pituitary, POMC neurons in the arcuate nucleus of
Circadian Rhythm of Glucocorticoid Secretion. The secretory
the hypothalamus, cells of the dermis, and lymphoid
rates of CRF, ACTH, and cortisol are high in the early
tissue. In all of these cell types, POMC is processed to
morning but low in the late evening, as shown in Figure
form a series of smaller peptides. The precise type
77-7; the plasma cortisol level ranges between a high of
of POMC-derived products from a particular tissue
about 20 mg/dl an hour before arising in the morning
depends on the type of processing enzymes present in
and a low of about 5 mg/dl around midnight. This effect
the tissue. Thus, pituitary corticotroph cells express
results from a 24-hour cyclical alteration in the signals
prohormone convertase 1 (PC1), but not PC2, result-
from the hypothalamus that cause cortisol secretion.
ing in the production of N-terminal peptide, joining
When a person changes daily sleeping habits, the cycle
peptide, ACTH, b-endorphin, and b-lipotropin. In the
changes correspondingly. Therefore, measurements of
blood cortisol levels are meaningful only when
hypothalamus, the expression of PC2 leads to the
expressed in terms of the time in the cycle at which the
production of a-, b-, and g-MSH, but not ACTH. As
measurements are made.
discussed in Chapter 71, a-MSH formed by neurons of
the hypothalamus plays a major role in appetite
Synthesis and Secretion of ACTH in
regulation.
Association with Melanocyte-Stimulating
In melanocytes located in abundance between the
Hormone, Lipotropin, and Endorphin
dermis and epidermis of the skin, MSH stimulates
When ACTH is secreted by the anterior pituitary
formation of the black pigment melanin and disperses
gland, several other hormones that have similar chem-
it to the epidermis. Injection of MSH into a person
ical structures are secreted simultaneously. The reason
over 8 to 10 days can greatly increase darkening of the
for this is that the gene that is transcribed to form the
skin. The effect is much greater in people who have
RNA molecule that causes ACTH synthesis initially
genetically dark skins than in light-skinned people.
causes the formation of a considerably larger protein,
In some lower animals, an intermediate “lobe” of
a preprohormone called proopiomelanocortin
the pituitary gland, called the pars intermedia, is highly
(POMC), which is the precursor of ACTH as well
developed, lying between the anterior and posterior
as several other peptides, including melanocyte-
pituitary lobes. This lobe secretes an especially large
Chapter 77
Adrenocortical Hormones
957
NH
2
COOH
Proopiomelanocortin
Figure 77-8
Proopiomelanocortin
(POMC)
processing by prohormone con-
vertase 1 (PC1, red arrows) and
N-Terminal protein
Joining
ACTH
b-Lipotropin
PC 2 (blue arrows). Tissue spe-
cific expression of these two
protein
enzymes results in different pep-
tides produced in various tissues.
PCI
The anterior pituitary expresses
PC1, resulting in formation of N-
g-MSH
a-MSH
CLIP
g-Lipotropin
b-Endorphin
PC2
terminal peptide, joining peptide,
ACTH, and b-lipotropin. Expres-
sion of PC2 within the hypothala-
mus leads to the production of a-,
b-, and g-melanocyte stimulating
hormone (MSH), but not ACTH.
b-MSH
CLIP, corticotropin-like intermedi-
ate peptide.
amount of MSH. Furthermore, this secretion is inde-
Abnormalities of
pendently controlled by the hypothalamus in response
Adrenocortical Secretion
to the amount of light to which the animal is exposed
or in response to other environmental factors. For
Hypoadrenalism-Addison’s Disease
instance, some arctic animals develop darkened fur in
Addison’s disease results from failure of the adrenal
the summer and yet have entirely white fur in the
cortices to produce adrenocortical hormones, and this
winter.
in turn is most frequently caused by primary atrophy of
ACTH, because it contains an MSH sequence, has
the adrenal cortices. In about 80 per cent of the cases,
about 1/30 as much melanocyte-stimulating effect as
the atrophy is caused by autoimmunity against the cor-
MSH. Furthermore, because the quantities of pure
tices. Adrenal gland hypofunction is also frequently
MSH secreted in the human being are extremely
caused by tuberculous destruction of the adrenal glands
small, whereas those of ACTH are large, it is likely that
or invasion of the adrenal cortices by cancer. The dis-
ACTH normally is more important than MSH in
turbances in Addison’s disease are as follows.
determining the amount of melanin in the skin.
Mineralocorticoid Deficiency. Lack of aldosterone secre-
tion greatly decreases renal tubular sodium reabsorp-
Adrenal Androgens
tion and consequently allows sodium ions, chloride ions,
and water to be lost into urine in great profusion. The
Several moderately active male sex hormones called
net result is a greatly decreased extracellular fluid
adrenal androgens (the most important of which is
volume. Furthermore, hyponatremia, hyperkalemia, and
dehydroepiandrosterone) are continually secreted by
mild acidosis develop because of failure of potassium
the adrenal cortex, especially during fetal life, as dis-
and hydrogen ions to be secreted in exchange for
cussed more fully in Chapter 83. Also, progesterone and
sodium reabsorption.
estrogens, which are female sex hormones, are secreted
As the extracellular fluid becomes depleted, plasma
in minute quantities.
volume falls, red blood cell concentration rises
Normally, the adrenal androgens have only weak
markedly, cardiac output decreases, and the patient dies
effects in humans. It is possible that part of the early
in shock, death usually occurring in the untreated
development of the male sex organs results from child-
patient 4 days to 2 weeks after cessation of mineralo-
hood secretion of adrenal androgens. The adrenal
corticoid secretion.
androgens also exert mild effects in the female, not only
before puberty but also throughout life. Much of the
growth of the pubic and axillary hair in the female
Glucocorticoid Deficiency. Loss of cortisol secretion makes
results from the action of these hormones.
it impossible for a person with Addison’s disease to
In extra-adrenal tissues, some of the adrenal andro-
maintain normal blood glucose concentration between
gens are converted to testosterone, the primary male sex
meals because he or she cannot synthesize significant
hormone, which probably accounts for much of their
quantities of glucose by gluconeogenesis. Furthermore,
androgenic activity. The physiologic effects of andro-
lack of cortisol reduces the mobilization of both
gens are discussed in Chapter 80 in relation to male
proteins and fats from the tissues, thereby depressing
sexual function.
many other metabolic functions of the body. This
958
Unit XIV Endocrinology and Reproduction
sluggishness of energy mobilization when cortisol is not
secretion” of ACTH by a tumor elsewhere in the body,
available is one of the major detrimental effects of glu-
such as an abdominal carcinoma; and (4) adenomas of
cocorticoid lack. Even when excess quantities of glucose
the adrenal cortex. When Cushing’s syndrome is sec-
and other nutrients are available, the person’s muscles
ondary to excess secretion of ACTH by the anterior
are weak, indicating that glucocorticoids are needed to
pituitary, this is referred to as Cushing’s disease.
maintain other metabolic functions of the tissues in
Excess ACTH secretion is the most common cause of
addition to energy metabolism.
Cushing’s syndrome and is characterized by high plasma
Lack of adequate glucocorticoid secretion also makes
levels of ACTH as well as cortisol. Primary overpro-
a person with Addison’s disease highly susceptible to
duction of cortisol by the adrenal glands accounts for
the deteriorating effects of different types of stress, and
about 20 to 25 per cent of clinical cases of Cushing’s syn-
even a mild respiratory infection can cause death.
drome and is usually associated with reduced ACTH
levels due to cortisol feedback inhibition of ACTH
Melanin Pigmentation. Another characteristic of most
secretion by the anterior pituitary gland.
people with Addison’s disease is melanin pigmentation
Administration of large doses of dexamethasone,
of the mucous membranes and skin. This melanin is not
a synthetic glucocorticoid, can be used to distinguish
always deposited evenly but occasionally is deposited in
between ACTH-dependent and ACTH-independent
blotches, and it is deposited especially in the thin skin
Cushing’s syndrome. In patients who have overproduc-
areas, such as the mucous membranes of the lips and the
tion of ACTH due to an ACTH-secreting pituitary
thin skin of the nipples.
adenoma or to hypothalamic-pituitary dysfunction,
The cause of the melanin deposition is believed to be
even large doses of dexamethasone usually do not
the following: When cortisol secretion is depressed, the
suppress ACTH secretion. In contrast, patients with
normal negative feedback to the hypothalamus and
primary adrenal overproduction of cortisol (ACTH-
anterior pituitary gland is also depressed, therefore
independent) usually have low or undetectable levels of
allowing tremendous rates of ACTH secretion as well
ACTH. The dexamethasone test, although widely used,
as simultaneous secretion of increased amounts of
can sometimes give an incorrect diagnosis, because
MSH. Probably the tremendous amounts of ACTH
some ACTH-secreting pituitary tumors respond to
cause most of the pigmenting effect because they can
dexamethasone with suppressed ACTH secretion.
stimulate formation of melanin by the melanocytes in
Therefore, it is usually considered to be a first step in
the same way that MSH does.
the differential diagnosis of Cushing’s syndrome.
Cushing’s syndrome can also occur when large
Treatment of People with Addison’s Disease. An untreated
amounts of glucocorticoids are administered over pro-
person with total adrenal destruction dies within a few
longed periods for therapeutic purposes. For example,
days to a few weeks because of weakness and usually
patients with chronic inflammation associated with dis-
circulatory shock. Yet such a person can live for years if
eases such as rheumatoid arthritis are often treated with
small quantities of mineralocorticoids and glucocorti-
glucocorticoids and may develop some of the clinical
coids are administered daily.
symptoms of Cushing’s syndrome.
A special characteristic of Cushing’s syndrome is
Addisonian Crisis. As noted earlier in the chapter, great
mobilization of fat from the lower part of the body, with
quantities of glucocorticoids are occasionally secreted
concomitant extra deposition of fat in the thoracic and
in response to different types of physical or mental
upper abdominal regions, giving rise to a buffalo torso.
stress. In a person with Addison’s disease, the output of
The excess secretion of steroids also leads to an ede-
glucocorticoids does not increase during stress. Yet
matous appearance of the face, and the androgenic
whenever different types of trauma, disease, or other
potency of some of the hormones sometimes causes
stresses, such as surgical operations, supervene, a person
acne and hirsutism (excess growth of facial hair). The
is likely to have an acute need for excessive amounts of
appearance of the face is frequently described as a
glucocorticoids and often must be given 10 or more
“moon face,” as demonstrated in the untreated patient
times the normal quantities of glucocorticoids to
with Cushing’s syndrome to the left in Figure 77-8.
prevent death.
About 80 per cent of patients have hypertension, pre-
This critical need for extra glucocorticoids and the
sumably because of the slight mineralocorticoid effects
associated severe debility in times of stress is called an
of cortisol.
addisonian crisis.
Effects on Carbohydrate and Protein Metabolism. The abun-
dance of cortisol secreted in Cushing’s syndrome can
Hyperadrenalism-Cushing’s
cause increased blood glucose concentration, some-
Syndrome
times to values as high as 200 mg/dl after meals-as much
as twice normal. This results mainly from enhanced glu-
Hypersecretion by the adrenal cortex causes a complex
coneogenesis and decreased glucose utilization by the
cascade of hormone effects called Cushing’s syndrome.
tissues.
Most of the abnormalities of Cushing’s syndrome are
The effects of glucocorticoids on protein catabolism
ascribable to abnormal amounts of cortisol, but excess
are often profound in Cushing’s syndrome, causing
secretion of androgens may also cause important
greatly decreased tissue proteins almost everywhere in
effects. Hypercortisolism can occur from multiple
the body with the exception of the liver; the plasma pro-
causes, including (1) adenomas of the anterior pituitary
teins also remain unaffected. The loss of protein from
that secrete large amounts of ACTH, which then causes
the muscles in particular causes severe weakness. The
adrenal hyperplasia and excess cortisol secretion; (2)
loss of protein synthesis in the lymphoid tissues leads to
abnormal function of the hypothalamus that causes high
a suppressed immune system, so that many of these
levels of corticotropin-releasing hormone
(CRH),
patients die of infections. Even the protein collagen
which stimulates excess ACTH release; (3) “ectopic
fibers in the subcutaneous tissue are diminished so that
Chapter 77
Adrenocortical Hormones
959
the subcutaneous tissues tear easily, resulting in devel-
paralysis caused by the hypokalemia. The paralysis is
opment of large purplish striae where they have torn
caused by a depressant effect of low extracellular potas-
apart. In addition, severely diminished protein deposi-
sium concentration on action potential transmission by
tion in the bones often causes severe osteoporosis with
the nerve fibers, as explained in Chapter 5.
consequent weakness of the bones.
One of the diagnostic criteria of primary aldostero-
nism is a decreased plasma renin concentration. This
Treatment of Cushing’s Syndrome. Treatment of Cushing’s
results from feedback suppression of renin secretion
syndrome consists of removing an adrenal tumor if this
caused by the excess aldosterone or by the excess extra-
is the cause or decreasing the secretion of ACTH, if
cellular fluid volume and arterial pressure resulting
this is possible. Hypertrophied pituitary glands or even
from the aldosteronism. Treatment of primary aldos-
small tumors in the pituitary that oversecrete ACTH
teronism is usually surgical removal of the tumor or of
can sometimes be surgically removed or destroyed
most of the adrenal tissue when hyperplasia is the cause.
by radiation. Drugs that block steroidogenesis, such as
metyrapone, ketoconazole, and aminoglutethimide, or
that inhibit ACTH secretion, such as serotonin antago-
Adrenogenital Syndrome
nists and GABA-transaminase inhibitors, can also be
used when surgery is not feasible. If ACTH secretion
An occasional adrenocortical tumor secretes excessive
cannot easily be decreased, the only satisfactory treat-
quantities of androgens that cause intense masculiniz-
ment is usually bilateral partial (or even total) adrena-
ing effects throughout the body. If this occurs in a
lectomy, followed by administration of adrenal steroids
female, she develops virile characteristics, including
to make up for any insufficiency that develops.
growth of a beard, a much deeper voice, occasionally
baldness if she also has the genetic trait for baldness,
masculine distribution of hair on the body and the pubis,
Primary Aldosteronism
growth of the clitoris to resemble a penis, and deposi-
(Conn’s Syndrome)
tion of proteins in the skin and especially in the muscles
to give typical masculine characteristics.
Occasionally a small tumor of the zona glomerulosa
In the prepubertal male, a virilizing adrenal tumor
cells occurs and secretes large amounts of aldosterone;
causes the same characteristics as in the female plus
the resulting condition is called “primary aldostero-
rapid development of the male sexual organs, as shown
nism” or “Conn’s syndrome.” Also, in a few instances,
in Figure 77-9, which depicts a 4-year-old boy with
hyperplastic adrenal cortices secrete aldosterone rather
adrenogenital syndrome. In the adult male, the viriliz-
than cortisol. The effects of the excess aldosterone are
ing characteristics of adrenogenital syndrome are
discussed in detail earlier in the chapter. The most
usually obscured by the normal virilizing characteristics
important effects are hypokalemia, slight increase
of the testosterone secreted by the testes. It is often dif-
in extracellular fluid volume and blood volume, very
ficult to make a diagnosis of adrenogenital syndrome in
slight increase in plasma sodium concentration (usually
the adult male. In adrenogenital syndrome, the excre-
not more than a
4 to
6 mEq/L increase), and,
tion of 17-ketosteroids (which are derived from andro-
almost always, hypertension. Especially interesting in
gens) in the urine may be 10 to 15 times normal. This
primary aldosteronism are occasional periods of muscle
finding can be used in diagnosing the disease.
Figure 77-9
A person with Cushing’s syn-
drome before
(left) and after
(right) subtotal adrenalectomy.
(Courtesy Dr. Leonard Posey.)
960
Unit XIV
Endocrinology and Reproduction
syndrome: a consensus statement. J Clin Endocrinol
Metab 88:5593, 2003.
Besser GM, Thorner MO: Comprehensive Clinical
Endocrinology,
3rd ed. Philadelphia: Mosby, Elsevier
Science Limited, 2002.
Boldyreff B, Wehling M: Aldosterone: refreshing a slow
hormone by swift action. News Physiol Sci 19:97, 2004.
Boscaro M, Barzon L, Fallo F, Sonino N: Cushing’s syn-
drome. Lancet 357:783, 2001.
Cooper MS, Stewart PM: Corticosteroid insufficiency in
acutely ill patients. N Engl J Med 348:727, 2003.
De Bosscher K, Vanden Berghe W, Haegeman G: The inter-
play between the glucocorticoid receptor and nuclear
factor-kappa B or activator protein-1: molecular mecha-
nisms for gene repression. Endocr Rev 24:488, 2003.
de Paula RB, da Silva AA, Hall JE: Aldosterone antagonism
attenuates obesity-induced hypertension and glomerular
hyperfiltration. Hypertension 43:41, 2004.
Farman N, Rafestin-Oblin ME: Multiple aspects of miner-
alocorticoid selectivity. Am J Physiol Renal Physiol 280:
F181, 2001.
Hall JE, Granger JP, Smith MJ Jr, Premen AJ: Role of renal
hemodynamics and arterial pressure in aldosterone
“escape.” Hypertension 6 :I183, 1984.
Larsen PR, Kronenberg HM, Melmed S, Polonsky KS:
Williams Textbook of Endocrinology, 10th ed. Philadel-
phia: WB Saunders Co, 2003.
Lösel RM, Falkenstein E, Feuring M, Schultz M, Tillmann
HC, Rossol-Haseroth K, Wehling M: Nongenomic steroid
action: Controversies, questions, and answers. Physiol Rev
83:965, 2003.
New MI: Inborn errors of adrenal steroidogenesis. Mol Cell
Endocrinol 211:75, 2003
Oberleithner H: Unorthodox sites and modes of aldosterone
action. News Physiol Sci 19:51, 2004.
O’Shaughnessy KM, Karet FE: Salt handling and hyperten-
sion. J Clin Invest 113:1075, 2004.
Raff H, Findling JW: A physiologic approach to diagnosis of
the Cushing syndrome. Ann Intern Med 138:980, 2003.
Sheppard KE: Nuclear receptors. II. Intestinal corticosteroid
receptors. Am J Physiol Gastrointest Liver Physiol
282:G742, 2002.
Spat A, Hunyady L: Control of aldosterone secretion: a
model for convergence in cellular signaling pathways
Figure 77-10
Physiol Rev 84:489, 2004.
Adrenogenital
syndrome
in
a
4-year-old
boy.
(Courtesy
Dr.
Speiser PW, White PC: Congenital adrenal hyperplasia.
Leonard Posey.)
N Engl J Med 349:776, 2003.
Stewart PM: Adrenal replacement therapy: time for an
inward look to the medulla? J Clin Endocrinol Metab
89:3677, 2004.
References
Stocco DM: StAR protein and the regulation of steroid
hormone biosynthesis. Annu Rev Physiol 63:193, 2001.
Arnaldi G, Angeli A, Atkinson AB, Bertagna X, Cavagnini
Stockand JD: New ideas about aldosterone signaling in
F, Chrousos GP, Fava GA, Findling JW, Gaillard RC,
epithelia. Am J Physiol Renal Physiol 282:F559, 2002.
Grossman AB, Kola B, Lacroix A, Mancini T, Mantero F,
Stowasser M, Gordon RD: Primary aldosteronism: from
Newell-Price J, Nieman LK, Sonino N, Vance ML, Giustina
genesis to genetics. Trends Endocrinol Metab
14:310,
A, Boscaro M: Diagnosis and complications of Cushing’s
2003.
C H A P T E R
7
8
Insulin, Glucagon, and
Diabetes Mellitus
The pancreas, in addition to its digestive functions,
secretes two important hormones, insulin and
glucagon, that are crucial for normal regulation of
glucose, lipid, and protein metabolism. Although the
pancreas secretes other hormones, such as amylin,
somatostatin, and pancreatic polypeptide, their func-
tions are not as well established. The main purpose
of this chapter is to discuss the physiologic roles of
insulin and glucagon and the pathophysiology of diseases, especially diabetes
mellitus, caused by abnormal secretion or activity of these hormones.
Physiologic Anatomy of the Pancreas. The pancreas is composed of two major types of
tissues, as shown in Figure 78-1: (1) the acini, which secrete digestive juices into the
duodenum, and (2) the islets of Langerhans, which secrete insulin and glucagon
directly into the blood. The digestive secretions of the pancreas are discussed in
Chapter 64.
The human pancreas has 1 to 2 million islets of Langerhans, each only about 0.3
millimeter in diameter and organized around small capillaries into which its cells
secrete their hormones. The islets contain three major types of cells, alpha, beta, and
delta cells, which are distinguished from one another by their morphological and
staining characteristics.
The beta cells, constituting about 60 per cent of all the cells of the islets, lie mainly
in the middle of each islet and secrete insulin and amylin, a hormone that is often
secreted in parallel with insulin, although its function is unclear. The alpha cells,
about 25 per cent of the total, secrete glucagon. And the delta cells, about 10 per
cent of the total, secrete somatostatin. In addition, at least one other type of cell,
the PP cell, is present in small numbers in the islets and secretes a hormone of uncer-
tain function called pancreatic polypeptide.
The close interrelations among these cell types in the islets of Langerhans allow
cell-to-cell communication and direct control of secretion of some of the hormones
by the other hormones. For instance, insulin inhibits glucagon secretion, amylin
inhibits insulin secretion, and somatostatin inhibits the secretion of both insulin and
glucagon.
Insulin and Its Metabolic Effects
Insulin was first isolated from the pancreas in 1922 by Banting and Best, and
almost overnight the outlook for the severely diabetic patient changed from one
of rapid decline and death to that of a nearly normal person. Historically, insulin
has been associated with “blood sugar,” and true enough, insulin has profound
effects on carbohydrate metabolism. Yet it is abnormalities of fat metabolism,
causing such conditions as acidosis and arteriosclerosis, that are the usual causes
of death in diabetic patients. Also, in patients with prolonged diabetes, dimin-
ished ability to synthesize proteins leads to wasting of the tissues as well as many
cellular functional disorders. Therefore, it is clear that insulin affects fat and
protein metabolism almost as much as it does carbohydrate metabolism.
Insulin Is a Hormone Associated with Energy Abundance
As we discuss insulin in the next few pages, it will become apparent that insulin
secretion is associated with energy abundance. That is, when there is great
961
962
Unit XIV Endocrinology and Reproduction
abundance of energy-giving foods in the diet, espe-
Chapter 3, beginning with translation of the insulin
cially excess amounts of carbohydrates, insulin is
RNA by ribosomes attached to the endoplasmic re-
secreted in great quantity. In turn, the insulin plays an
ticulum to form an insulin preprohormone. This
important role in storing the excess energy. In the case
initial preprohormone has a molecular weight of about
of excess carbohydrates, it causes them to be stored as
11,500, but it is then cleaved in the endoplasmic retic-
glycogen mainly in the liver and muscles. Also, all the
ulum to form a proinsulin with a molecular weight
excess carbohydrates that cannot be stored as glyco-
of about 9000; most of this is further cleaved in the
gen are converted under the stimulus of insulin into
Golgi apparatus to form insulin and peptide fragments
fats and stored in the adipose tissue. In the case of pro-
before being packaged in the secretory granules.
teins, insulin has a direct effect in promoting amino
However, about one sixth of the final secreted product
acid uptake by cells and conversion of these amino
is still in the form of proinsulin. The proinsulin has vir-
acids into protein. In addition, it inhibits the break-
tually no insulin activity.
down of the proteins that are already in the cells.
When insulin is secreted into the blood, it circulates
almost entirely in an unbound form; it has a plasma
Insulin Chemistry and Synthesis
half-life that averages only about 6 minutes, so that it
Insulin is a small protein; human insulin has a molec-
is mainly cleared from the circulation within 10 to 15
ular weight of 5808. It is composed of two amino acid
minutes. Except for that portion of the insulin that
chains, shown in Figure 78-2, connected to each other
combines with receptors in the target cells, the remain-
by disulfide linkages. When the two amino acid chains
der is degraded by the enzyme insulinase mainly in the
are split apart, the functional activity of the insulin
liver, to a lesser extent in the kidneys and muscles, and
molecule is lost.
slightly in most other tissues. This rapid removal from
Insulin is synthesized in the beta cells by the usual
the plasma is important, because, at times, it is as
cell machinery for protein synthesis, as explained in
important to turn off rapidly as to turn on the control
functions of insulin.
Activation of Target Cell Receptors by Insulin
Islet of
Pancreatic
and the Resulting Cellular Effects
Langerhans
acini
To initiate its effects on target cells, insulin first binds
with and activates a membrane receptor protein that
Delta cell
has a molecular weight of about 300,000 (Figure 78-3).
It is the activated receptor, not the insulin, that causes
the subsequent effects.
The insulin receptor is a combination of four sub-
units held together by disulfide linkages: two alpha
subunits that lie entirely outside the cell membrane
and two beta subunits that penetrate through the mem-
brane, protruding into the cell cytoplasm. The insulin
Alpha cell
binds with the alpha subunits on the outside of the cell,
but because of the linkages with the beta subunits, the
Red blood cells
portions of the beta subunits protruding into the cell
Beta cell
become autophosphorylated. Thus, the insulin recep-
tor is an example of an enzyme-linked receptor, dis-
Figure 78-1
cussed in Chapter 74. Autophosphorylation of the beta
subunits of the receptor activates a local tyrosine
Physiologic anatomy of an islet of Langerhans in the pancreas.
NH2
S
NH2
NH2
NH2
Gly•Ileu•Val•Glu•Glu•Cy•Cy•Thr•Ser•Ileu•Cy•Ser•Leu•Tyr•Glu•Leu•Glu•Asp•Tyr•Cy•Asp
S
S
NH2
NH2
S
S
Phe•Val•Asp•Glu•His•Leu•Cy•Gly•Ser•His•Leu•Val•Glu•Ala•Leu•Tyr•Leu•Val•Cy•Gly•Glu•Arg•Gly•Phe•Phe•Tyr•Thr•Pro•Lys•Thr
Figure 78-2
Human insulin molecule.
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
963
about 3 to 5 minutes and move back to the cell
Insulin
interior to be used again and again as needed.
2. The cell membrane becomes more permeable to
Insulin
many of the amino acids, potassium ions, and
a
S
S
a
receptor
phosphate ions, causing increased transport of
S S
S
S
these substances into the cell.
3. Slower effects occur during the next 10 to 15
minutes to change the activity levels of many
Glucose
more intracellular metabolic enzymes. These
effects result mainly from the changed states of
b
Cell membrane
b
phosphorylation of the enzymes.
4. Much slower effects continue to occur for hours
and even several days. They result from changed
Tyrosine
Tyrosine
rates of translation of messenger RNAs at the
kinase
kinase
ribosomes to form new proteins and still slower
Insulin receptor substrates (IRS)
effects from changed rates of transcription of
Phosphorylation of enzymes
DNA in the cell nucleus. In this way, insulin
remolds much of the cellular enzymatic machinery
Glucose
Fat
Growth
to achieve its metabolic goals.
transport
synthesis
and gene
expression
Protein
Glucose
Effect of Insulin on Carbohydrate
synthesis
synthesis
Metabolism
Figure 78-3
Immediately after a high-carbohydrate meal, the
Schematic of the insulin receptor. Insulin binds to the a-subunit of
glucose that is absorbed into the blood causes rapid
its receptor, which causes autophosphorylation of the b-subunit
secretion of insulin, which is discussed in detail later
receptor, which in turn induces tyrosine kinase activity. The recep-
in the chapter. The insulin in turn causes rapid uptake,
tor tyrosine kinase activity begins a cascade of cell phosphoryla-
storage, and use of glucose by almost all tissues of the
tion that increases or decreases the activity of enzymes, including
body, but especially by the muscles, adipose tissue, and
insulin receptor substrates, that mediate the effects of glucose on
liver.
glucose, fat, and protein metabolism. For example, glucose trans-
porters are moved to the cell membrane to facilitate glucose entry
into the cell.
Insulin Promotes Muscle Glucose Uptake
and Metabolism
During much of the day, muscle tissue depends not on
kinase, which in turn causes phosphorylation of multi-
glucose for its energy but on fatty acids. The principal
ple other intracellular enzymes including a group
reason for this is that the normal resting muscle mem-
called insulin-receptor substrates
(IRS). Different
brane is only slightly permeable to glucose, except
types of IRS (e.g. IRS-1, IRS-2, IRS-3) are expressed
when the muscle fiber is stimulated by insulin; between
in different tissues. The net effect is to activate some
meals, the amount of insulin that is secreted is too
of these enzymes while inactivating others. In this way,
small to promote significant amounts of glucose entry
insulin directs the intracellular metabolic machinery to
into the muscle cells.
produce the desired effects on carbohydrate, fat, and
However, under two conditions the muscles do use
protein metabolism. The end effects of insulin stimu-
large amounts of glucose. One of these is during mod-
lation are the following:
erate or heavy exercise. This usage of glucose does not
1. Within seconds after insulin binds with its
require large amounts of insulin, because exercising
membrane receptors, the membranes of about
muscle fibers become more permeable to glucose even
80 per cent of the body’s cells markedly increase
in the absence of insulin because of the contraction
their uptake of glucose. This is especially true of
process itself.
muscle cells and adipose cells but is not true of
The second condition for muscle usage of large
most neurons in the brain. The increased glucose
amounts of glucose is during the few hours after a
transported into the cells is immediately
meal. At this time the blood glucose concentration is
phosphorylated and becomes a substrate for all
high and the pancreas is secreting large quantities of
the usual carbohydrate metabolic functions. The
insulin. The extra insulin causes rapid transport of
increased glucose transport is believed to result
glucose into the muscle cells. This causes the muscle
from translocation of multiple intracellular
cell during this period to use glucose preferentially
vesicles to the cell membranes; these vesicles
over fatty acids, as we discuss later.
carry in their own membranes multiple molecules
of glucose transport proteins, which bind with the
Storage of Glycogen in Muscle. If the muscles are not
cell membrane and facilitate glucose uptake into
exercising after a meal and yet glucose is transported
the cells. When insulin is no longer available, these
into the muscle cells in abundance, then most of the
vesicles separate from the cell membrane within
glucose is stored in the form of muscle glycogen
964
Unit XIV Endocrinology and Reproduction
instead of being used for energy, up to a limit of 2 to
The mechanism by which insulin causes glucose
3 per cent concentration. The glycogen can later be
uptake and storage in the liver includes several almost
used for energy by the muscle. It is especially useful
simultaneous steps:
for short periods of extreme energy use by the muscles
1. Insulin inactivates liver phosphorylase, the
and even to provide spurts of anaerobic energy for a
principal enzyme that causes liver glycogen to
few minutes at a time by glycolytic breakdown of the
split into glucose. This prevents breakdown of the
glycogen to lactic acid, which can occur even in the
glycogen that has been stored in the liver cells.
absence of oxygen.
2. Insulin causes enhanced uptake of glucose from
the blood by the liver cells. It does this by
Quantitative Effect of Insulin to Facilitate Glucose Transport
increasing the activity of the enzyme glucokinase,
Through the Muscle Cell Membrane. The quantitative
which is one of the enzymes that causes the initial
effect of insulin to facilitate glucose transport through
phosphorylation of glucose after it diffuses into
the muscle cell membrane is demonstrated by the
the liver cells. Once phosphorylated, the glucose is
experimental results shown in Figure 78-4. The lower
temporarily trapped inside the liver cells because
curve labeled “control” shows the concentration of
phosphorylated glucose cannot diffuse back
free glucose measured inside the cell, demonstrating
through the cell membrane.
that the glucose concentration remained almost zero
3. Insulin also increases the activities of the enzymes
despite increased extracellular glucose concentration
that promote glycogen synthesis, including
up to as high as 750 mg/100 ml. In contrast, the curve
especially glycogen synthase, which is responsible
labeled “insulin” demonstrates that the intracellular
for polymerization of the monosaccharide units to
glucose concentration rose to as high as 400 mg/100 ml
form the glycogen molecules.
when insulin was added. Thus, it is clear that insulin
The net effect of all these actions is to increase the
can increase the rate of transport of glucose into the
amount of glycogen in the liver. The glycogen can
resting muscle cell by at least 15-fold.
increase to a total of about 5 to 6 per cent of the liver
mass, which is equivalent to almost 100 grams of stored
Insulin Promotes Liver Uptake, Storage, and
glycogen in the whole liver.
Use of Glucose
One of the most important of all the effects of insulin
Glucose Is Released from the Liver Between Meals. When the
is to cause most of the glucose absorbed after a meal
blood glucose level begins to fall to a low level
to be stored almost immediately in the liver in the
between meals, several events transpire that cause the
form of glycogen. Then, between meals, when food is
liver to release glucose back into the circulating blood:
not available and the blood glucose concentration
1. The decreasing blood glucose causes the pancreas
begins to fall, insulin secretion decreases rapidly and
to decrease its insulin secretion.
the liver glycogen is split back into glucose, which is
2. The lack of insulin then reverses all the effects
released back into the blood to keep the glucose con-
listed earlier for glycogen storage, essentially
centration from falling too low.
stopping further synthesis of glycogen in the liver
and preventing further uptake of glucose by the
liver from the blood.
3. The lack of insulin (along with increase of
glucagon, which is discussed later) activates the
enzyme phosphorylase, which causes the splitting
400
of glycogen into glucose phosphate.
300
Insulin
4. The enzyme glucose phosphatase, which had been
inhibited by insulin, now becomes activated by the
200
insulin lack and causes the phosphate radical to
split away from the glucose; this allows the free
100
Control
glucose to diffuse back into the blood.
0
Thus, the liver removes glucose from the blood
when it is present in excess after a meal and returns it
to the blood when the blood glucose concentration
0
300
600
900
falls between meals. Ordinarily, about 60 per cent of
Extracellular glucose
the glucose in the meal is stored in this way in the liver
(mg/100 ml)
and then returned later.
Insulin Promotes Conversion of Excess Glucose into Fatty Acids
Figure 78-4
and Inhibits Gluconeogenesis in the Liver. When the quan-
tity of glucose entering the liver cells is more than can
Effect of insulin in enhancing the concentration of glucose inside
muscle cells. Note that in the absence of insulin (control), the
be stored as glycogen or can be used for local hepato-
intracellular glucose concentration remains near zero, despite
cyte metabolism, insulin promotes the conversion
high extracellular glucose concentrations. (Data from Eisenstein
AB: The Biochemical Aspects of Hormone Action, Boston, Little,
of all this excess glucose into fatty acids. These fatty
Brown, 1964.)
acids are subsequently packaged as triglycerides in
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
965
very-low-density lipoproteins and transported in this
automatically decreases the utilization of fat, thus
form by way of the blood to the adipose tissue and
functioning as a fat sparer. However, insulin also
deposited as fat.
promotes fatty acid synthesis. This is especially true
Insulin also inhibits gluconeogenesis. It does this
when more carbohydrates are ingested than can be
mainly by decreasing the quantities and activities of
used for immediate energy, thus providing the sub-
the liver enzymes required for gluconeogenesis.
strate for fat synthesis. Almost all this synthesis occurs
However, part of the effect is caused by an action
in the liver cells, and the fatty acids are then trans-
of insulin that decreases the release of amino acids
ported from the liver by way of the blood lipoproteins
from muscle and other extrahepatic tissues and in
to the adipose cells to be stored. The different factors
turn the availability of these necessary precursors
that lead to increased fatty acid synthesis in the liver
required for gluconeogenesis. This is discussed
include the following:
further in relation to the effect of insulin on protein
1.
Insulin increases the transport of glucose into the
metabolism.
liver cells. After the liver glucogen concentration
reaches 5 to 6 per cent, this in itself inhibits
further glycogen synthesis. Then all the additional
Lack of Effect of Insulin on Glucose Uptake
glucose entering the liver cells becomes available
and Usage by the Brain
to form fat. The glucose is first split to pyruvate
The brain is quite different from most other tissues of
in the glycolytic pathway, and the pyruvate
the body in that insulin has little effect on uptake or
subsequently is converted to acetyl coenzyme A
use of glucose. Instead, the brain cells are permeable to
(acetyl-CoA), the substrate from which fatty acids
glucose and can use glucose without the intermediation
are synthesized.
of insulin.
2.
An excess of citrate and isocitrate ions is formed
The brain cells are also quite different from most
by the citric acid cycle when excess amounts of
other cells of the body in that they normally use only
glucose are being used for energy. These ions then
glucose for energy and can use other energy sub-
have a direct effect in activating acetyl-CoA
strates, such as fats, only with difficulty. Therefore, it is
carboxylase, the enzyme required to carboxylate
essential that the blood glucose level always be main-
acetyl-CoA to form malonyl-CoA, the first stage
tained above a critical level, which is one of the most
of fatty acid synthesis.
important functions of the blood glucose control
3.
Most of the fatty acids are then synthesized within
system. When the blood glucose falls too low, into
the liver itself and used to form triglycerides, the
the range of 20 to 50 mg/100 ml, symptoms of hypo-
usual form of storage fat. They are released from
glycemic shock develop, characterized by progressive
the liver cells to the blood in the lipoproteins.
nervous irritability that leads to fainting, seizures, and
Insulin activates lipoprotein lipase in the capillary
even coma.
walls of the adipose tissue, which splits the
triglycerides again into fatty acids, a requirement
Effect of Insulin on Carbohydrate Metabolism
for them to be absorbed into the adipose cells,
in Other Cells
where they are again converted to triglycerides
Insulin increases glucose transport into and glucose
and stored.
usage by most other cells of the body (with the excep-
tion of the brain cells, as noted) in the same way that
it affects glucose transport and usage in muscle cells.
Role of Insulin in Storage of Fat in the Adipose Cells. Insulin
The transport of glucose into adipose cells mainly pro-
has two other essential effects that are required for fat
vides substrate for the glycerol portion of the fat mol-
storage in adipose cells:
ecule. Therefore, in this indirect way, insulin promotes
1. Insulin inhibits the action of hormone-sensitive
deposition of fat in these cells.
lipase. This is the enzyme that causes hydrolysis of
the triglycerides already stored in the fat cells.
Therefore, the release of fatty acids from the
Effect of Insulin on Fat Metabolism
adipose tissue into the circulating blood is
inhibited.
Although not quite as visible as the acute effects of
2. Insulin promotes glucose transport through the cell
insulin on carbohydrate metabolism, insulin’s effects
membrane into the fat cells in exactly the same
on fat metabolism are, in the long run, equally impor-
ways that it promotes glucose transport into
tant. Especially dramatic is the long-term effect of
muscle cells. Some of this glucose is then used to
insulin lack in causing extreme atherosclerosis, often
synthesize minute amounts of fatty acids, but
leading to heart attacks, cerebral strokes, and other
more important, it also forms large quantities of
vascular accidents. But first, let us discuss the acute
a-glycerol phosphate. This substance supplies the
effects of insulin on fat metabolism.
glycerol that combines with fatty acids to form
the triglycerides that are the storage form of fat
Insulin Promotes Fat Synthesis and Storage
in adipose cells. Therefore, when insulin is not
Insulin has several effects that lead to fat storage in
available, even storage of the large amounts of
adipose tissue. First, insulin increases the utilization
fatty acids transported from the liver in the
of glucose by most of the body’s tissues, which
lipoproteins is almost blocked.
966
Unit XIV Endocrinology and Reproduction
Insulin Deficiency Increases Use of Fat
the liver, are then discharged into the blood in the
for Energy
lipoproteins. Occasionally the plasma lipoproteins
All aspects of fat breakdown and use for providing
increase as much as threefold in the absence of insulin,
energy are greatly enhanced in the absence of insulin.
giving a total concentration of plasma lipids of several
This occurs even normally between meals when secre-
per cent rather than the normal 0.6 per cent. This high
tion of insulin is minimal, but it becomes extreme in
lipid concentration—especially the high concentration
diabetes mellitus when secretion of insulin is almost
of cholesterol—promotes the development of athero-
zero. The resulting effects are as follows.
sclerosis in people with serious diabetes.
Insulin Deficiency Causes Lipolysis of Storage Fat and Release
Excess Usage of Fats During Insulin Lack Causes Ketosis and
of Free Fatty Acids. In the absence of insulin, all the
Acidosis. Insulin lack also causes excessive amounts of
effects of insulin noted earlier that cause storage of
acetoacetic acid to be formed in the liver cells. This
fat are reversed. The most important effect is that
results from the following effect: In the absence of
the enzyme hormone-sensitive lipase in the fat cells
insulin but in the presence of excess fatty acids in the
becomes strongly activated. This causes hydrolysis of
liver cells, the carnitine transport mechanism for trans-
the stored triglycerides, releasing large quantities of
porting fatty acids into the mitochondria becomes
fatty acids and glycerol into the circulating blood.
increasingly activated. In the mitochondria, beta oxi-
Consequently, the plasma concentration of free fatty
dation of the fatty acids then proceeds very rapidly,
acids begins to rise within minutes. This free fatty acid
releasing extreme amounts of acetyl-CoA. A large part
then becomes the main energy substrate used by
of this excess acetyl-CoA is then condensed to form
essentially all tissues of the body besides the brain.
acetoacetic acid, which in turn is released into the cir-
Figure 78-5 shows the effect of insulin lack on the
culating blood. Most of this passes to the peripheral
plasma concentrations of free fatty acids, glucose, and
cells, where it is again converted into acetyl-CoA and
acetoacetic acid. Note that almost immediately after
used for energy in the usual manner.
removal of the pancreas, the free fatty acid concentra-
At the same time, the absence of insulin also
tion in the plasma begins to rise, more rapidly even
depresses the utilization of acetoacetic acid in the
than the concentration of glucose.
peripheral tissues. Thus, so much acetoacetic acid is
released from the liver that it cannot all be metabo-
Insulin Deficiency Increases Plasma Cholesterol and Phospho-
lized by the tissues. Therefore, as shown in Figure 78-5,
lipid Concentrations. The excess of fatty acids in the
its concentration rises during the days after cessation
plasma associated with insulin deficiency also pro-
of insulin secretion, sometimes reaching concentra-
motes liver conversion of some of the fatty acids into
tions of 10 mEq/L or more, which is a severe state of
phospholipids and cholesterol, two of the major prod-
body fluid acidosis.
ucts of fat metabolism. These two substances, along
As explained in Chapter 68, some of the acetoacetic
with excess triglycerides formed at the same time in
acid is also converted into b-hydroxybutyric acid and
acetone. These two substances, along with the ace-
toacetic acid, are called ketone bodies, and their pres-
ence in large quantities in the body fluids is called
ketosis. We see later that in severe diabetes the ace-
Control
Depancreatized
toacetic acid and the b-hydroxybutyric acid can cause
severe acidosis and coma, which often leads to death.
Blood glucose
Effect of Insulin on Protein
Metabolism and on Growth
Free fatty acids
Insulin Promotes Protein Synthesis and Storage. During the
few hours after a meal when excess quantities of nutri-
ents are available in the circulating blood, not only car-
bohydrates and fats but proteins as well are stored in
the tissues; insulin is required for this to occur. The
manner in which insulin causes protein storage is not
Acetoacetic acid
as well understood as the mechanisms for both glucose
and fat storage. Some of the facts follow.
0
1
2
3
4
1. Insulin stimulates transport of many of the amino
Days
acids into the cells. Among the amino acids most
strongly transported are valine, leucine, isoleucine,
tyrosine, and phenylalanine. Thus, insulin shares
Figure 78-5
with growth hormone the capability of increasing
the uptake of amino acids into cells. However, the
Effect of removing the pancreas on the approximate concentra-
amino acids affected are not necessarily the same
tions of blood glucose, plasma free fatty acids, and acetoacetic
acid.
ones.
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
967
2.
Insulin increases the translation of messenger
RNA, thus forming new proteins. In some
Growth hormone
unexplained way, insulin “turns on” the ribosomal
and insulin
machinery. In the absence of insulin, the
250
ribosomes simply stop working, almost as if
Depancreatized and
insulin operates an “on-off” mechanism.
200
hypophysectomized
3.
Over a longer period of time, insulin also
increases the rate of transcription of selected DNA
150
Growth
Insulin
genetic sequences in the cell nuclei, thus forming
hormone
100
increased quantities of RNA and still more
protein synthesis—especially promoting a vast
50
array of enzymes for storage of carbohydrates,
fats, and proteins.
0
4.
Insulin inhibits the catabolism of proteins, thus
0
50
100
150
200
250
decreasing the rate of amino acid release from the
Days
cells, especially from the muscle cells. Presumably
this results from the ability of insulin to diminish
the normal degradation of proteins by the cellular
Figure 78-6
lysosomes.
5.
In the liver, insulin depresses the rate of
Effect of growth hormone, insulin, and growth hormone plus insulin
gluconeogenesis. It does this by decreasing
on growth in a depancreatized and hypophysectomized rat.
the activity of the enzymes that promote
gluconeogenesis. Because the substrates most used
for synthesis of glucose by gluconeogenesis are
Mechanisms of Insulin Secretion
the plasma amino acids, this suppression of
gluconeogenesis conserves the amino acids in the
Figure 78-7 shows the basic cellular mechanisms for
protein stores of the body.
insulin secretion by the pancreatic beta cells in
In summary, insulin promotes protein formation and
response to increased blood glucose concentration, the
prevents the degradation of proteins.
primary controller of insulin secretion. The beta cells
have a large number of glucose transporters (GLUT-
Insulin Lack Causes Protein Depletion and Increased Plasma
2) that permit a rate of glucose influx that is propor-
Amino Acids. Virtually all protein storage comes to a
tional to the blood concentration in the physiologic
halt when insulin is not available. The catabolism of
range. Once inside the cells, glucose is phosphorylated
proteins increases, protein synthesis stops, and large
to glucose-6-phosphate by glucokinase. This step
quantities of amino acids are dumped into the plasma.
appears to be the rate limiting for glucose metabolism
The plasma amino acid concentration rises consider-
in the beta cell and is considered the major mechanism
ably, and most of the excess amino acids are used
for glucose sensing and adjustment of the amount of
either directly for energy or as substrates for gluco-
secreted insulin to the blood glucose levels.
neogenesis. This degradation of the amino acids also
The glucose-6-phosphate is subsequently oxidized to
leads to enhanced urea excretion in the urine. The
form adenosine triphosphate (ATP), which inhibits the
resulting protein wasting is one of the most serious of
ATP-sensitive potassium channels of the cell. Closure
all the effects of severe diabetes mellitus. It can lead
of the potassium channels depolarizes the cell mem-
to extreme weakness as well as many deranged func-
brane, thereby opening voltage-gated calcium chan-
tions of the organs.
nels, which are sensitive to changes in membrane
voltage. This produces an influx of calcium that stimu-
Insulin and Growth Hormone Interact Synergistically to Promote
lates fusion of the docked insulin-containing vesicles
Growth. Because insulin is required for the synthesis of
with the cell membrane and secretion of insulin into
proteins, it is as essential for growth of an animal as
the extracellular fluid by exocytosis.
growth hormone is. This is demonstrated in Figure
Other nutrients, such as certain amino acids, can also
78-6, which shows that a depancreatized, hypophysec-
be metabolized by the beta cells to increase intracel-
tomized rat without therapy hardly grows at all.
lular ATP levels and stimulate insulin secretion. Some
Furthermore, the administration of either growth
hormones, such as glucagon and gastric inhibitory
hormone or insulin one at a time causes almost no
peptide, as well as acetylcholine increase intracellular
growth. Yet a combination of these hormones causes
calcium levels through other signaling pathways and
dramatic growth. Thus, it appears that the two hor-
enhance the effect of glucose, although they do not
mones function synergistically to promote growth,
have major effects on insulin secretion in the absence
each performing a specific function that is separate
of glucose. Other hormones, including somatostatin
from that of the other. Perhaps a small part of this
and norepinephrine (by activating a-adrenergic recep-
necessity for both hormones results from the fact that
tors), inhibit exocytosis of insulin.
each promotes cellular uptake of a different selection
Sulfonylurea drugs stimulate insulin secretion by
of amino acids, all of which are required if growth is
binding to the ATP-sensitive potassium channels and
to be achieved.
blocking their activity. This results in a depolarizing
968
Unit XIV Endocrinology and Reproduction
Glucose
Insulin
GLUT 2
250
80
Glucose
60
Glucokinase
40
20
Glucose-6-phosphate
0
Oxidation
-10
0
10
20
30
40
50
60
70
80
Minutes
ATP
Ca++
K+
Depolarization
Figure 78-8
Increase in plasma insulin concentration after a sudden increase
in blood glucose to two to three times the normal range. Note an
initial rapid surge in insulin concentration and then a delayed but
ATP + K+ channel
Ca++ channel
higher and continuing increase in concentration beginning 15 to
(closed)
(open)
20 minutes later.
Figure 78-7
Basic mechanisms of glucose stimulation of insulin secretion by
metabolism, it has become apparent that blood amino
beta cells of the pancreas. GLUT, glucose transporter.
acids and other factors also play important roles in
controlling insulin secretion (see Table 78-1).
Table 78-1
Increased Blood Glucose Stimulates Insulin Secretion. At
the normal fasting level of blood glucose of 80 to
Factors and Conditions That Increase or Decrease
90 mg/100 ml, the rate of insulin secretion is minimal—
Insulin Secretion
on the order of 25 ng/min/kg of body weight, a level
that has only slight physiologic activity. If the blood
Increase Insulin Secretion
Decrease Insulin Secretion
glucose concentration is suddenly increased to a level
two to three times normal and kept at this high level
• Increased blood glucose
• Decreased blood glucose
• Increased blood free fatty acids
• Fasting
thereafter, insulin secretion increases markedly in two
• Increased blood amino acids
• Somatostatin
stages, as shown by the changes in plasma insulin con-
• Gastrointestinal hormones
• a-Adrenergic activity
centration seen in Figure 78-8.
(gastrin, cholecystokinin, secretin,
• Leptin
1. Plasma insulin concentration increases almost
gastric inhibitory peptide)
• Glucagon, growth hormone,
10-fold within 3 to 5 minutes after the acute
cortisol
elevation of the blood glucose; this results from
• Parasympathetic stimulation;
immediate dumping of preformed insulin from the
acetylcholine
beta cells of the islets of Langerhans. However,
• b-Adrenergic stimulation
the initial high rate of secretion is not maintained;
• Insulin resistance; obesity
• Sulfonylurea drugs (glyburide,
instead, the insulin concentration decreases about
tolbutamide)
halfway back toward normal in another 5 to 10
minutes.
2. Beginning at about 15 minutes, insulin secretion
rises a second time and reaches a new plateau in
effect that triggers insulin secretion, making these
2 to 3 hours, this time usually at a rate of
drugs very useful in stimulating insulin secretion in
secretion even greater than that in the initial
patients with type II diabetes, as we will discuss later.
phase. This secretion results both from additional
Table 78-1 summarizes some of the factors that can
release of preformed insulin and from activation
increase or decrease insulin secretion.
of the enzyme system that synthesizes and
releases new insulin from the cells.
Control of Insulin Secretion
Feedback Relation Between Blood Glucose Concentration and
Insulin Secretion Rate. As the concentration of blood
Formerly, it was believed that insulin secretion was
glucose rises above 100 mg/100 ml of blood, the rate
controlled almost entirely by the blood glucose con-
of insulin secretion rises rapidly, reaching a peak some
centration. However, as more has been learned about
10 to 25 times the basal level at blood glucose con-
the metabolic functions of insulin for protein and fat
centrations between 400 and 600 mg/100 ml, as shown
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
969
cholecystokinin, and gastric inhibitory peptide (which
seems to be the most potent)—causes a moderate
20
increase in insulin secretion. These hormones are
released in the gastrointestinal tract after a person eats
a meal. They then cause an “anticipatory” increase in
15
blood insulin in preparation for the glucose and amino
acids to be absorbed from the meal. These gastroin-
10
testinal hormones generally act the same way as amino
acids to increase the sensitivity of insulin response to
increased blood glucose, almost doubling the rate of
5
insulin secretion as the blood glucose level rises.
0
Other Hormones and the Autonomic Nervous System. Other hor-
0
100
200
300
400
500
600
mones that either directly increase insulin secretion or
Plasma glucose concentration
potentiate the glucose stimulus for insulin secretion
(mg/100 ml)
include glucagon, growth hormone, cortisol, and, to a
lesser extent, progesterone and estrogen. The impor-
tance of the stimulatory effects of these hormones is
that prolonged secretion of any one of them in large
Figure 78-9
quantities can occasionally lead to exhaustion of the
beta cells of the islets of Langerhans and thereby
Approximate insulin secretion at different plasma glucose levels.
increase the risk for developing diabetes mellitus.
Indeed, diabetes often occurs in people who are main-
tained on high pharmacological doses of some of these
in Figure 78-9. Thus, the increase in insulin secretion
hormones. Diabetes is particularly common in giants or
under a glucose stimulus is dramatic both in its rapid-
acromegalic people with growth hormone-secreting
ity and in the tremendous level of secretion achieved.
tumors, or in people whose adrenal glands secrete
excess glucocorticoids.
Furthermore, the turn-off of insulin secretion is almost
Under some conditions, stimulation of the parasym-
equally as rapid, occurring within 3 to 5 minutes after
pathetic nerves to the pancreas can increase insulin
reduction in blood glucose concentration back to the
secretion. However, it is doubtful that this effect is of
fasting level.
physiologic significance for regulating insulin secretion.
This response of insulin secretion to an elevated
blood glucose concentration provides an extremely
important feedback mechanism for regulating blood
glucose concentration. That is, any rise in blood
Role of Insulin (and Other Hormones)
glucose increases insulin secretion, and the insulin in
in “Switching” Between Carbohydrate
turn increases transport of glucose into liver, muscle,
and Lipid Metabolism
and other cells, thereby reducing the blood glucose
concentration back toward the normal value.
From the preceding discussions, it should be clear that
insulin promotes the utilization of carbohydrates for
energy, whereas it depresses the utilization of fats.
Other Factors That Stimulate
Conversely, lack of insulin causes fat utilization mainly
Insulin Secretion
to the exclusion of glucose utilization, except by brain
Amino Acids. In addition to the stimulation of insulin
tissue. Furthermore, the signal that controls this
secretion by excess blood glucose, some of the amino
switching mechanism is principally the blood glucose
acids have a similar effect. The most potent of these are
concentration. When the glucose concentration is low,
arginine and lysine. This effect differs from glucose stim-
insulin secretion is suppressed and fat is used almost
ulation of insulin secretion in the following way: Amino
exclusively for energy everywhere except in the brain.
acids administered in the absence of a rise in blood
When the glucose concentration is high, insulin secre-
glucose cause only a small increase in insulin secretion.
tion is stimulated and carbohydrate is used instead of
However, when administered at the same time that the
fat, and the excess blood glucose is stored in the form
blood glucose concentration is elevated, the glucose-
of liver glycogen, liver fat, and muscle glycogen. There-
induced secretion of insulin may be as much as doubled
in the presence of the excess amino acids. Thus, the
fore, one of the most important functional roles of
amino acids strongly potentiate the glucose stimulus for
insulin in the body is to control which of these two
insulin secretion.
foods from moment to moment will be used by the
The stimulation of insulin secretion by amino acids
cells for energy.
is important, because the insulin in turn promotes
At least four other known hormones also play
transport of amino acids into the tissue cells as well as
important roles in this switching mechanism: growth
intracellular formation of protein. That is, insulin is
hormone from the anterior pituitary gland, cortisol
important for proper utilization of excess amino acids
from the adrenal cortex, epinephrine from the adrenal
in the same way that it is important for the utilization
medulla, and glucagon from the alpha cells of the islets
of carbohydrates.
of Langerhans in the pancreas. Glucagon is discussed
Gastrointestinal Hormones. A mixture of several impor-
in the next section of this chapter. Both growth
tant gastrointestinal hormones—gastrin, secretin,
hormone and cortisol are secreted in response to
970
Unit XIV Endocrinology and Reproduction
hypoglycemia, and both inhibit cellular utilization of
1. Glucagon activates adenylyl cyclase in the hepatic
glucose while promoting fat utilization. However,
cell membrane,
the effects of both of these hormones develop
2. Which causes the formation of cyclic adenosine
slowly, usually requiring many hours for maximal
monophosphate,
expression.
3. Which activates protein kinase regulator protein,
Epinephrine is especially important in increasing
4. Which activates protein kinase,
plasma glucose concentration during periods of stress
5. Which activates phosphorylase b kinase,
when the sympathetic nervous system is excited.
6. Which converts phosphorylase b into
However, epinephrine acts differently from the other
phosphorylase a,
hormones in that it increases the plasma fatty acid
7. Which promotes the degradation of glycogen into
concentration at the same time. The reasons for these
glucose-1-phosphate,
effects are as follows: (1) epinephrine has the potent
8. Which then is dephosphorylated; and the glucose
effect of causing glycogenolysis in the liver, thus
is released from the liver cells.
releasing within minutes large quantities of glucose
This sequence of events is exceedingly important for
into the blood; (2) it also has a direct lipolytic effect
several reasons. First, it is one of the most thoroughly
on the adipose cells because it activates adipose tissue
studied of all the second messenger functions of cyclic
hormone-sensitive lipase, thus greatly enhancing the
adenosine monophosphate. Second, it demonstrates
blood concentration of fatty acids as well. Quantita-
a cascade system in which each succeeding product
tively, the enhancement of fatty acids is far greater
is produced in greater quantity than the preceding
than the enhancement of blood glucose. Therefore,
product. Therefore, it represents a potent amplifying
epinephrine especially enhances the utilization of fat
mechanism; this type of amplifying mechanism is
in such stressful states as exercise, circulatory shock,
widely used throughout the body for controlling many,
and anxiety.
if not most, cellular metabolic systems, often causing
as much as a millionfold amplification in response. This
explains how only a few micrograms of glucagon can
cause the blood glucose level to double or increase even
Glucagon and Its Functions
more within a few minutes.
Infusion of glucagon for about 4 hours can cause such
Glucagon, a hormone secreted by the alpha cells of the
intensive liver glycogenolysis that all the liver stores of
islets of Langerhans when the blood glucose concen-
glycogen become depleted.
tration falls, has several functions that are diametri-
cally opposed to those of insulin. Most important of
Glucagon Increases Gluconeogenesis. Even after all the
these functions is to increase the blood glucose con-
glycogen in the liver has been exhausted under the
centration, an effect that is exactly the opposite that of
influence of glucagon, continued infusion of this
insulin.
hormone still causes continued hyperglycemia. This
Like insulin, glucagon is a large polypeptide. It has
results from the effect of glucagon to increase the rate
a molecular weight of 3485 and is composed of a chain
of amino acid uptake by the liver cells and then the
of 29 amino acids. On injection of purified glucagon
conversion of many of the amino acids to glucose by
into an animal, a profound hyperglycemic effect
gluconeogenesis. This is achieved by activating multi-
occurs. Only 1 mg/kg of glucagon can elevate the blood
ple enzymes that are required for amino acid transport
glucose concentration about 20 mg/100 ml of blood (a
and gluconeogenesis, especially activation of the
25 per cent increase) in about 20 minutes. For this
enzyme system for converting pyruvate to phospho-
reason, glucagon is also called the hyperglycemic
enolpyruvate, a rate-limiting step in gluconeogenesis.
hormone.
Other Effects of Glucagon
Most other effects of glucagon occur only when its
Effects on Glucose Metabolism
concentration rises well above the maximum normally
found in the blood. Perhaps the most important effect
The major effects of glucagon on glucose metabolism
is that glucagon activates adipose cell lipase, making
are (1) breakdown of liver glycogen (glycogenolysis)
increased quantities of fatty acids available to the
and (2) increased gluconeogenesis in the liver. Both of
energy systems of the body. Glucagon also inhibits
these effects greatly enhance the availability of glucose
the storage of triglycerides in the liver, which prevents
to the other organs of the body.
the liver from removing fatty acids from the blood; this
also helps make additional amounts of fatty acids
Glucagon Causes Glycogenolysis and Increased Blood Glucose
available for the other tissues of the body.
Concentration. The most dramatic effect of glucagon is
Glucagon in very high concentrations also
(1)
its ability to cause glycogenolysis in the liver, which in
enhances the strength of the heart; (2) increases blood
turn increases the blood glucose concentration within
flow in some tissues, especially the kidneys;
(3)
minutes.
enhances bile secretion; and (4) inhibits gastric acid
It does this by the following complex cascade of
secretion. All these effects are probably of minimal
events:
importance in the normal function of the body.
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
971
One of the factors that might increase glucagon
secretion in exercise is increased circulating amino
acids. Other factors, such as b-adrenergic stimulation
4
of the islets of Langerhans, may also play a role.
3
Somatostatin Inhibits
2
Glucagon and Insulin
Secretion
1
The delta cells of the islets of Langerhans secrete the
hormone somatostatin, a polypeptide containing only 14
0
amino acids that has an extremely short half-life of only
60
80
100
120
3 minutes in the circulating blood. Almost all factors
Blood glucose
related to the ingestion of food stimulate somatostatin
(mg/100 ml)
secretion. They include (1) increased blood glucose, (2)
increased amino acids, (3) increased fatty acids, and (4)
increased concentrations of several of the gastrointesti-
nal hormones released from the upper gastrointestinal
Figure 78-10
tract in response to food intake.
In turn, somatostatin has multiple inhibitory effects
Approximate plasma glucagon concentration at different blood
glucose levels.
as follows:
1. Somatostatin acts locally within the islets of
Langerhans themselves to depress the secretion of
Regulation of Glucagon Secretion
both insulin and glucagon.
2. Somatostatin decreases the motility of the stomach,
duodenum, and gallbladder.
Increased Blood Glucose Inhibits Glucagon Secretion. The
3. Somatostatin decreases both secretion and
blood glucose concentration is by far the most potent
absorption in the gastrointestinal tract.
factor that controls glucagon secretion. Note specifi-
Putting all this information together, it has been sug-
cally, however, that the effect of blood glucose concen-
gested that the principal role of somatostatin is to
tration on glucagon secretion is in exactly the opposite
extend the period of time over which the food nutrients
direction from the effect of glucose on insulin secretion.
are assimilated into the blood. At the same time, the
This is demonstrated in Figure 78-10, showing that
effect of somatostatin to depress insulin and glucagon
a decrease in the blood glucose concentration from its
secretion decreases the utilization of the absorbed
normal fasting level of about 90 mg/100 ml of blood
nutrients by the tissues, thus preventing rapid exhaus-
down to hypoglycemic levels can increase the plasma
tion of the food and therefore making it available over
a longer period of time.
concentration of glucagon severalfold. Conversely,
It should also be recalled that somatostatin is the
increasing the blood glucose to hyperglycemic levels
same chemical substance as growth hormone inhibitory
decreases plasma glucagon. Thus, in hypoglycemia,
hormone, which is secreted in the hypothalamus and
glucagon is secreted in large amounts; it then greatly
suppresses anterior pituitary gland growth hormone
increases the output of glucose from the liver and
secretion.
thereby serves the important function of correcting the
hypoglycemia.
Summary of Blood
Increased Blood Amino Acids Stimulate Glucagon Secretion.
High concentrations of amino acids, as occur in the
Glucose Regulation
blood after a protein meal (especially the amino acids
alanine and arginine), stimulate the secretion of
In a normal person, the blood glucose concentration is
glucagon. This is the same effect that amino acids have
narrowly controlled, usually between 80 and 90 mg/
in stimulating insulin secretion. Thus, in this instance,
100 ml of blood in the fasting person each morning
the glucagon and insulin responses are not opposites.
before breakfast. This concentration increases to 120
The importance of amino acid stimulation of glucagon
to 140 mg/100 ml during the first hour or so after a
secretion is that the glucagon then promotes rapid
meal, but the feedback systems for control of blood
conversion of the amino acids to glucose, thus making
glucose return the glucose concentration rapidly back
even more glucose available to the tissues.
to the control level, usually within 2 hours after the last
absorption of carbohydrates. Conversely, in starvation,
Exercise Stimulates Glucagon Secretion. In exhaustive
the gluconeogenesis function of the liver provides the
exercise, the blood concentration of glucagon often
glucose that is required to maintain the fasting blood
increases fourfold to fivefold. What causes this is not
glucose level.
understood, because the blood glucose concentration
The mechanisms for achieving this high degree of
does not necessarily fall. A beneficial effect of the
control have been presented in this chapter. Let us
glucagon is that it prevents a decrease in blood glucose.
summarize them.
972
Unit XIV Endocrinology and Reproduction
1.
The liver functions as an important blood glucose
not secrete any insulin during this time; otherwise, the
buffer system. That is, when the blood glucose
scant supplies of glucose that are available would all
rises to a high concentration after a meal and the
go into the muscles and other peripheral tissues,
rate of insulin secretion also increases, as much as
leaving the brain without a nutritive source.
two thirds of the glucose absorbed from the gut is
It is also important that the blood glucose concen-
almost immediately stored in the liver in the form
tration not rise too high for four reasons: (1) Glucose
of glycogen. Then, during the succeeding hours,
can exert a large amount of osmotic pressure in the
when both the blood glucose concentration and
extracellular fluid, and if the glucose concentration
the rate of insulin secretion fall, the liver releases
rises to excessive values, this can cause considerable
the glucose back into the blood. In this way, the
cellular dehydration.(2) An excessively high level of
liver decreases the fluctuations in blood glucose
blood glucose concentration causes loss of glucose in
concentration to about one third of what they
the urine. (3) Loss of glucose in the urine also causes
would otherwise be. In fact, in patients with
osmotic diuresis by the kidneys, which can deplete
severe liver disease, it becomes almost impossible
the body of its fluids and electrolytes.(4) Long-term
to maintain a narrow range of blood glucose
increases in blood glucose may cause damage to many
concentration.
tissues, especially to blood vessels. Vascular injury,
2.
Both insulin and glucagon function as important
associated with uncontrolled diabetes mellitus, leads to
feedback control systems for maintaining a normal
increased risk for heart attack, stroke, end-stage renal
blood glucose concentration. When the glucose
disease, and blindness.
concentration rises too high, insulin is secreted;
the insulin in turn causes the blood glucose
concentration to decrease toward normal.
Diabetes Mellitus
Conversely, a decrease in blood glucose stimulates
Diabetes mellitus is a syndrome of impaired carbohy-
glucagon secretion; the glucagon then functions in
drate, fat, and protein metabolism caused by either lack
the opposite direction to increase the glucose
of insulin secretion or decreased sensitivity of the
toward normal. Under most normal conditions,
tissues to insulin. There are two general types of dia-
the insulin feedback mechanism is much more
betes mellitus:
important than the glucagon mechanism, but in
1. Type I diabetes, also called insulin-dependent
instances of starvation or excessive utilization of
diabetes mellitus (IDDM), is caused by lack of
glucose during exercise and other stressful
insulin secretion.
situations, the glucagon mechanism also becomes
2. Type II diabetes, also called non-insulin-dependent
diabetes mellitus (NIDDM), is caused by decreased
valuable.
sensitivity of target tissues to the metabolic effect
3.
Also, in severe hypoglycemia, a direct effect of
of insulin. This reduced sensitivity to insulin is often
low blood glucose on the hypothalamus stimulates
called insulin resistance.
the sympathetic nervous system. In turn, the
In both types of diabetes mellitus, metabolism of all
epinephrine secreted by the adrenal glands causes
the main foodstuffs is altered. The basic effect of insulin
still further release of glucose from the liver. This,
lack or insulin resistance on glucose metabolism is to
too, helps protect against severe hypoglycemia.
prevent the efficient uptake and utilization of glucose
4.
And finally, over a period of hours and days, both
by most cells of the body, except those of the brain. As
growth hormone and cortisol are secreted in
a result, blood glucose concentration increases, cell
response to prolonged hypoglycemia, and they
utilization of glucose falls increasingly lower, and
utilization of fats and proteins increases.
both decrease the rate of glucose utilization by
most cells of the body, converting instead to
greater amounts of fat utilization. This, too, helps
Type I Diabetes—Lack of Insulin
return the blood glucose concentration toward
Production by Beta Cells
normal.
of the Pancreas
Importance of Blood Glucose Regulation. One might ask the
Injury to the beta cells of the pancreas or diseases that
question: Why is it so important to maintain a constant
impair insulin production can lead to type I diabetes.
blood glucose concentration, particularly because
Viral infections or autoimmune disorders may be
most tissues can shift to utilization of fats and proteins
involved in the destruction of beta cells in many patients
for energy in the absence of glucose? The answer is
with type I diabetes, although heredity also plays a
that glucose is the only nutrient that normally can be
major role in determining the susceptibility of the beta
used by the brain, retina, and germinal epithelium
cells to destruction by these insults. In some instances,
there may be a hereditary tendency for beta cell degen-
of the gonads in sufficient quantities to supply them
eration even without viral infections or autoimmune
optimally with their required energy. Therefore, it is
disorders.
important to maintain the blood glucose concentration
The usual onset of type I diabetes occurs at about 14
at a sufficiently high level to provide this necessary
years of age in the United States, and for this reason it
nutrition.
is often called juvenile diabetes mellitus. Type I diabetes
Most of the glucose formed by gluconeogenesis
may develop very abruptly, over a period of a few days
during the interdigestive period is used for metabolism
or weeks, with three principal sequelae: (1) increased
in the brain. Indeed, it is important that the pancreas
blood glucose, (2) increased utilization of fats for energy
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
973
and for formation of cholesterol by the liver, and (3)
amplify the tissue damage caused by the elevated
depletion of the body’s proteins.
glucose.
Blood Glucose Concentration Rises to Very High Levels in Diabetes
Diabetes Mellitus Causes Increased Utilization of Fats and Meta-
Mellitus. The lack of insulin decreases the efficiency
bolic Acidosis. The shift from carbohydrate to fat metab-
of peripheral glucose utilization and augments
olism in diabetes increases the release of keto acids,
glucose production, raising plasma glucose to 300 to
such as acetoacetic acid and b-hydroxybutyric acid, into
1200 mg/100 ml. The increased plasma glucose then has
the plasma more rapidly than they can be taken up and
multiple effects throughout the body.
oxidized by the tissue cells. As a result, the patient
develops severe metabolic acidosis from the excess keto
Increased Blood Glucose Causes Loss of Glucose in the Urine
acids, which, in association with dehydration due to the
The high blood glucose causes more glucose to filter
excessive urine formation, can cause severe acidosis.
into the renal tubules than can be reabsorbed, and the
This leads rapidly to diabetic coma and death unless the
excess glucose spills into the urine. This normally occurs
condition is treated immediately with large amounts of
when the blood glucose concentration rises above
insulin.
180 mg/100 ml, a level that is called the blood “thresh-
All the usual physiologic compensations that occur
old” for the appearance of glucose in the urine. When
in metabolic acidosis take place in diabetic acidosis.
the blood glucose level rises to 300 to 500 mg/100 ml—
They include rapid and deep breathing, which
common values in people with severe untreated dia-
causes increased expiration of carbon dioxide; this
betes—100 or more grams of glucose can be lost into
buffers the acidosis but also depletes extracellular fluid
the urine each day.
bicarbonate stores. The kidneys compensate by decreas-
ing bicarbonate excretion and generating new bicar-
Increased Blood Glucose Causes Dehydration The very high
bonate that is added back to the extracellular fluid.
levels of blood glucose (sometimes as high as 8 to 10
Although extreme acidosis occurs only in the most
times normal in severe untreated diabetes) can cause
severe instances of uncontrolled diabetes, when the pH
severe cell dehydration throughout the body. This
of the blood falls below about 7.0, acidotic coma and
occurs partly because glucose does not diffuse easily
death can occur within hours. The overall changes in the
through the pores of the cell membrane, and the
electrolytes of the blood as a result of severe diabetic
increased osmotic pressure in the extracellular fluids
acidosis are shown in Figure 78-11.
causes osmotic transfer of water out of the cells.
Excess fat utilization in the liver occurring over a long
In addition to the direct cellular dehydrating effect of
time causes large amounts of cholesterol in the circu-
excessive glucose, the loss of glucose in the urine causes
lating blood and increased deposition of cholesterol in
osmotic diuresis. That is, the osmotic effect of glucose in
the renal tubules greatly decreases tubular reabsorption
of fluid. The overall effect is massive loss of fluid in the
urine, causing dehydration of the extracellular fluid,
which in turn causes compensatory dehydration of the
intracellular fluid, for reasons discussed in Chapter 26.
Thus, polyuria (excessive urine excretion), intracellular
100 mg/dL
and extracellular dehydration, and increased thirst are
Glucose
classic symptoms of diabetes.
400+ mg/dL
Chronic High Glucose Concentration Causes Tissue Injury When
155 mEq
Keto acids
blood glucose is poorly controlled over long periods in
30 mEq
diabetes mellitus, blood vessels in multiple tissues
throughout the body begin to function abnormally and
155 mEq
Total cations
undergo structural changes that result in inadequate
130 mEq
blood supply to the tissues. This in turn leads to
increased risk for heart attack, stroke, end-stage kidney
27 mEq
disease, retinopathy and blindness, and ischemia and
HCO3
5 mEq
gangrene of the limbs.
Chronic high glucose concentration also causes
103 mEq
damage to many other tissues. For example, peripheral
Cl-
90 mEq
neuropathy, which is abnormal function of peripheral
nerves, and autonomic nervous system dysfunction are
7.4
frequent complications of chronic, uncontrolled dia-
pH
betes mellitus. These abnormalities can result in
6.9
impaired cardiovascular reflexes, impaired bladder
180 mg/dL
control, decreased sensation in the extremities, and
Cholesterol
other symptoms of peripheral nerve damage.
360 mg/dL
The precise mechanisms that cause tissue injury in
diabetes are not well understood but probably involve
multiple effects of high glucose concentrations and
other metabolic abnormalities on proteins of endothe-
lial and vascular smooth muscle cells, as well as other
Figure 78-11
tissues. In addition, hypertension, secondary to renal
injury, and atherosclerosis, secondary to abnormal lipid
Changes in blood constituents in diabetic coma, showing normal
metabolism, often develop in patients with diabetes and
values (lavender bars) and diabetic coma values (red bars).
974
Unit XIV Endocrinology and Reproduction
the arterial walls. This leads to severe arteriosclerosis
The role of insulin resistance in contributing to some
and other vascular lesions, as discussed earlier.
of the components of the metabolic syndrome is
unclear, although it is clear that insulin resistance is the
primary cause of increased blood glucose concentra-
Diabetes Causes Depletion of the Body’s Proteins. Failure to
tion. The major adverse consequence of the metabolic
use glucose for energy leads to increased utilization and
syndrome is cardiovascular disease, including athero-
decreased storage of proteins as well as fat. Therefore,
sclerosis and injury to various organs throughout the
a person with severe untreated diabetes mellitus suffers
body. Several of the metabolic abnormalities associated
rapid weight loss and asthenia (lack of energy) despite
with the syndrome are risk factors for cardiovascular
eating large amounts of food (polyphagia). Without
disease, and insulin resistance predisposes to the devel-
treatment, these metabolic abnormalities can cause
opment of type II diabetes mellitus, also a major cause
severe wasting of the body tissues and death within a
of cardiovascular disease.
few weeks.
Other Factors That Can Cause Insulin Resistance and Type II Dia-
betes. Although most patients with type II diabetes are
Type II Diabetes—Resistance to the
overweight or have substantial accumulation of visceral
Metabolic Effects of Insulin
fat, severe insulin resistance and type II diabetes can
also occur as a result of other acquired or genetic con-
Type II diabetes is far more common than type I,
ditions that impair insulin signaling in peripheral tissues
accounting for about 90 per cent of all cases of diabetes
(Table 78-2).
mellitus. In most cases, the onset of type II diabetes
Polycystic ovary syndrome (PCOS), for example, is
occurs after age 30, often between the ages of 50 and 60
associated with marked increases in ovarian androgen
years, and the disease develops gradually. Therefore, this
production and insulin resistance and is one of the most
syndrome is often referred to as adult-onset diabetes. In
common endocrine disorders in women, affecting ap-
recent years, however, there has been a steady increase
proximately 6 per cent of all women during their re-
in the number of younger individuals, some less than 20
productive life. Although the pathogenesis of PCOS
years old, with type II diabetes. This trend appears to be
remains uncertain, insulin resistance and hyperinsuline-
related mainly to the increasing prevalence of obesity,
mia are found in approximately 80 per cent of affected
the most important risk factor for type II diabetes in chil-
women. The long-term consequences include increased
dren as well as in adults.
risk for diabetes mellitus, increased blood lipids, and
cardiovascular disease.
Obesity, Insulin Resistance, and “Metabolic Syndrome” Usually
Excess formation of glucocorticoids (Cushing’s syn-
Precede Development of Type II Diabetes. Type II diabetes, in
drome) or growth hormone (acromegaly) also decreases
contrast to type I, is associated with increased plasma
the sensitivity of various tissues to the metabolic effects
insulin concentration (hyperinsulinemia). This occurs as
of insulin and can lead to development of diabetes mel-
a compensatory response by the pancreatic beta cells
litus. Genetic causes of obesity and insulin resistance, if
for diminished sensitivity of target tissues to the meta-
severe enough, also can lead to type II diabetes as well
bolic effects of insulin, a condition referred to as insulin
as many other features of the metabolic syndrome,
resistance. The decrease in insulin sensitivity impairs
including cardiovascular disease.
carbohydrate utilization and storage, raising blood
glucose and stimulating a compensatory increase in
Development of Type II Diabetes During Prolonged Insulin Resis-
insulin secretion.
tance. With prolonged and severe insulin resistance,
Development of insulin resistance and impaired
even the increased levels of insulin are not sufficient to
glucose metabolism is usually a gradual process, begin-
maintain normal glucose regulation. As a result, mod-
ning with excess weight gain and obesity. The mecha-
erate hyperglycemia occurs after ingestion of carbohy-
nisms that link obesity with insulin resistance, however,
drates in the early stages of the disease.
are still uncertain. Some studies suggest that there are
fewer insulin receptors, especially in the skeletal muscle,
liver, and adipose tissue, in obese than in lean subjects.
However, most of the insulin resistance appears to be
Table 78-2
caused by abnormalities of the signaling pathways that
link receptor activation with multiple cellular effects.
Some Causes of Insulin Resistance
Impaired insulin signaling appears to be closely related
to toxic effects of lipid accumulation in tissues such as
• Obesity/overweight (especially excess visceral adiposity)
skeletal muscle and liver secondary to excess weight
• Excess glucocorticoids (Cushing’s syndrome or steroid therapy)
gain.
• Excess growth hormone (acromegaly)
Insulin resistance is part of a cascade of disorders that
• Pregnancy, gestational diabetes
is often called the “metabolic syndrome.” Some of the
• Polycystic ovary disease
features of the metabolic syndrome include: (1) obesity,
• Lipodystrophy (acquired or genetic; associated with lipid
especially accumulation of abdominal fat; (2) insulin
accumulation in liver)
• Autoantibodies to the insulin receptor
resistance; (3) fasting hyperglycemia; (4) lipid abnor-
• Mutations of insulin receptor
malities such as increased blood triglycerides and
• Mutations of the peroxisome proliferators’ activator receptor g
decreased blood high-density lipoprotein-cholesterol;
(PPARg)
and (5) hypertension. All of the features of the meta-
• Mutations that cause genetic obesity (e.g., melanocortin
bolic syndrome are closely related to excess weight gain,
receptor mutations)
especially when it is associated with accumulation of
• Hemochromatosis (a hereditary disease that causes tissue iron
adipose tissue in the abdominal cavity around the vis-
accumulation)
ceral organs.
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
975
In the later stages of type II diabetes, the pancreatic
large amounts, in proportion to the severity of disease
beta cells become
“exhausted” and are unable to
and the intake of carbohydrates.
produce enough insulin to prevent more severe
hyperglycemia, especially after the person ingests a
Fasting Blood Glucose and Insulin Levels. The fasting blood
carbohydrate-rich meal.
glucose level in the early morning is normally 80 to
Some obese people, although having marked insulin
90 mg/100 ml, and 110 mg/100 ml is considered to be the
resistance and greater than normal increases in blood
upper limit of normal. A fasting blood glucose level
glucose after a meal, never develop clinically significant
above this value often indicates diabetes mellitus or a
diabetes mellitus; apparently, the pancreas in these
least marked insulin resistance.
people produces enough insulin to prevent severe
In type I diabetes, plasma insulin levels are very low
abnormalities of glucose metabolism. In others,
or undetectable during fasting and even after a meal. In
however, the pancreas gradually becomes exhausted
type II diabetes, plasma insulin concentration may be
from secreting large amounts of insulin, and full-blown
severalfold higher than normal and usually increases to
diabetes mellitus occurs. Some studies suggest that
a greater extent after ingestion of a standard glucose
genetic factors play an important role in determining
load during a glucose tolerance test
(see the next
whether an individual’s pancreas can sustain the high
paragraph).
output of insulin over many years that is necessary to
avoid the severe abnormalities of glucose metabolism in
Glucose Tolerance Test. As demonstrated by the bottom
type II diabetes.
curve in Figure
78-12, called a
“glucose tolerance
In many instances, type II diabetes can be effectively
curve,” when a normal, fasting person ingests 1 gram
treated, at least in the early stages, with exercise, caloric
of glucose per kilogram of body weight, the blood
restriction, and weight reduction, and no exogenous
glucose level rises from about 90 mg/100 ml to 120 to
insulin administration is required. Drugs that increase
140 mg/100 ml and falls back to below normal in
insulin sensitivity, such as thiazolidinediones and met-
about 2 hours.
formin, or drugs that cause additional release of insulin
In a person with diabetes, the fasting blood glucose
by the pancreas, such as sulfonylureas, may also be used.
concentration is almost always above 110 mg/100 ml
However, in the later stages of type II diabetes, insulin
and often above 140 mg/100 ml. Also, the glucose toler-
administration is usually required to control plasma
ance test is almost always abnormal. On ingestion of
glucose.
glucose, these people exhibit a much greater than
normal rise in blood glucose level, as demonstrated by
the upper curve in Figure 78-12, and the glucose level
Physiology of Diagnosis
falls back to the control value only after 4 to 6 hours;
of Diabetes Mellitus
furthermore, it fails to fall below the control level. The
slow fall of this curve and its failure to fall below the
Table 78-3 compares some of clinical features of type I
control level demonstrate that either (1) the normal
and type II diabetes mellitus. The usual methods for
increase in insulin secretion after glucose ingestion does
diagnosing diabetes are based on various chemical tests
not occur or (2) there is decreased sensitivity to insulin.
of the urine and the blood.
A diagnosis of diabetes mellitus can usually be estab-
lished on the basis of such a curve, and type I and type
Urinary Glucose. Simple office tests or more complicated
II diabetes can be distinguished from each other by
quantitative laboratory tests may be used to determine
measurements of plasma insulin, with plasma insulin
the quantity of glucose lost in the urine. In general, a
being low or undetectable in type I diabetes and
normal person loses undetectable amounts of glucose,
increased in type II diabetes.
whereas a person with diabetes loses glucose in small to
Table 78-3
Clinical Characteristics of Patients with Type I and Type II
200
Diabetes
Diabetes Mellitus
180
Feature
Type I
Type II
160
Age at onset
Usually <20 years
Usually >30 years
140
Body mass
Low (wasted) to
Obese
120
normal
Normal
Plasma insulin
Low or absent
Normal to high
100
initially
Plasma glucagon
High, can be
High, resistant to
80
suppressed
suppression
0
1
2
3
4
5
Plasma glucose
Increased
Increased
Hours
Insulin sensitivity
Normal
Reduced
Therapy
Insulin
Weight loss,
thiazolidinediones,
metformin,
Figure 78-12
sulfonylureas,
insulin
Glucose tolerance curve in a normal person and in a person with
diabetes.
976
Unit XIV Endocrinology and Reproduction
Acetone Breath. As pointed out in Chapter 68, small
Because the complications of diabetes—such as ath-
quantities of acetoacetic acid in the blood, which
erosclerosis, greatly increased susceptibility to infection,
increase greatly in severe diabetes, are converted to
diabetic retinopathy, cataracts, hypertension, and
acetone. This is volatile and vaporized into the expired
chronic renal disease—are closely associated with the
air. Consequently, one can frequently make a diagnosis
level of blood lipids as well as the level of blood glucose,
of type I diabetes mellitus simply by smelling acetone
most physicians also use lipid-lowering drugs to help
on the breath of a patient. Also, keto acids can be
prevent these disturbances.
detected by chemical means in the urine, and their
quantitation aids in determining the severity of the dia-
betes. In the early stages of type II diabetes, however,
Insulinoma—Hyperinsulinism
keto acids are usually not produced in excess amounts.
Although much rarer than diabetes, excessive insulin
However, when insulin resistance becomes very severe
production occasionally occurs from an adenoma of an
and there is greatly increased utilization of fats for
islet of Langerhans. About 10 to 15 per cent of these
energy, keto acids are then produced in persons with
adenomas are malignant, and occasionally metastases
type II diabetes.
from the islets of Langerhans spread throughout the
body, causing tremendous production of insulin by both
Treatment of Diabetes
the primary and the metastatic cancers. Indeed, more
than 1000 grams of glucose have had to be administered
The theory of treatment of type I diabetes mellitus is to
every 24 hours to prevent hypoglycemia in some of
administer enough insulin so that the patient will have
these patients.
carbohydrate, fat, and protein metabolism that is as
normal as possible. Insulin is available in several forms.
Insulin Shock and Hypoglycemia. As already emphasized,
“Regular” insulin has a duration of action that lasts
the central nervous system normally derives essentially
from 3 to 8 hours, whereas other forms of insulin (pre-
all its energy from glucose metabolism, and insulin is not
cipitated with zinc or with various protein derivatives)
necessary for this use of glucose. However, if high levels
are absorbed slowly from the injection site and there-
of insulin cause blood glucose to fall to low values, the
fore have effects that last as long as 10 to 48 hours. Ordi-
metabolism of the central nervous system becomes
narily, a patient with severe type I diabetes is given a
depressed. Consequently, in patients with insulin-secret-
single dose of one of the longer-acting insulins each day
ing tumors or in patients with diabetes who administer
to increase overall carbohydrate metabolism through-
too much insulin to themselves, the syndrome called
out the day. Then additional quantities of regular insulin
insulin shock may occur as follows.
are given during the day at those times when the blood
As the blood glucose level falls into the range
glucose level tends to rise too high, such as at mealtimes.
of
50 to
70 mg/100 ml, the central nervous system
Thus, each patient is provided with an individualized
usually becomes quite excitable, because this degree of
pattern of treatment.
hypoglycemia sensitizes neuronal activity. Sometimes
In persons with type II diabetes, dieting and exercise
various forms of hallucinations result, but more often
are usually recommended in an attempt to induce
the patient simply experiences extreme nervousness,
weight loss and to reverse the insulin resistance. If this
trembles all over, and breaks out in a sweat. As the
fails, drugs may be administered to increase insulin sen-
blood glucose level falls to 20 to 50 mg/100 ml, clonic
sitivity or to stimulate increased production of insulin
seizures and loss of consciousness are likely to occur. As
by the pancreas. In many persons, however, exogenous
the glucose level falls still lower, the seizures cease and
insulin must be used to regulate blood glucose.
only a state of coma remains. Indeed, at times it is dif-
In the past, the insulin used for treatment was derived
ficult by simple clinical observation to distinguish
from animal pancreata. However, human insulin pro-
between diabetic coma as a result of insulin-lack acido-
duced by the recombinant DNA process has become
sis and coma due to hypoglycemia caused by excess
more widely used because some patients develop immu-
insulin. The acetone breath and the rapid, deep breath-
nity and sensitization against animal insulin, thus limit-
ing of diabetic coma are not present in hypoglycemic
ing its effectiveness.
coma.
Proper treatment for a patient who has hypoglycemic
Relation of Treatment to Arteriosclerosis. Diabetic patients,
shock or coma is immediate intravenous administration
mainly because of their high levels of circulating cho-
of large quantities of glucose. This usually brings
lesterol and other lipids, develop atherosclerosis, arte-
the patient out of shock within a minute or more. Also,
riosclerosis, severe coronary heart disease, and multiple
the administration of glucagon
(or, less effectively,
microcirculatory lesions far more easily than do normal
epinephrine) can cause glycogenolysis in the liver and
people. Indeed, those who have poorly controlled dia-
thereby increase the blood glucose level extremely
betes throughout childhood are likely to die of heart
rapidly. If treatment is not effected immediately, per-
disease in early adulthood.
manent damage to the neuronal cells of the central
In the early days of treating diabetes, the tendency
nervous system often occurs.
was to severely reduce the carbohydrates in the diet so
that the insulin requirements would be minimized. This
procedure kept the blood glucose from increasing too
high and attenuated loss of glucose in the urine, but it
References
did not prevent many of the abnormalities of fat metab-
olism. Consequently, the current tendency is to allow the
Barrett EJ: Insulin’s effect on glucose production: direct or
patient an almost normal carbohydrate diet and to give
indirect? J Clin Invest 111:434, 2003.
large enough insulin to metabolize the carbohydrates.
Barthel A, Schmoll D: Novel concepts in insulin regulation
This decreases the rate of fat metabolism and depresses
of hepatic gluconeogenesis. Am J Physiol Endocrinol
the high level of blood cholesterol.
Metab 285:E685, 2003.
Chapter 78
Insulin, Glucagon, and Diabetes Mellitus
977
Besser GM, Thorner MO: Comprehensive Clinical Endo-
Hussain MA, Theise ND: Stem-cell therapy for diabetes mel-
crinology, 3rd ed. Philadelphia: Mosby, Elsevier Science
litus. Lancet 364:203, 2004.
Limited, 2002.
Kowluru A: Regulatory roles for small G proteins in the
Bryant NJ, Govers R, James DE: Regulated transport of the
pancreatic beta-cell: lessons from models of impaired
glucose transporter GLUT4. Nat Rev Mol Cell Biol 3:267,
insulin secretion. Am J Physiol Endocrinol Metab
2002.
285:E669, 2003.
Caumo A, Luzi L: First-phase insulin secretion: does it exist
Larsen PR, Kronenberg HM, Melmed S, Polonsky KS:
in real life? Considerations on shape and function. Am J
Williams Textbook of Endocrinology, 10th ed. Philadel-
Physiol Endocrinol Metab 287:E371, 2004.
phia: WB Saunders Co, 2003.
DeWitt DE, Hirsch IB: Outpatient insulin therapy in type 1
List JF, Habener JF: Glucagon-like peptide 1 agonists and
and type 2 diabetes mellitus: scientific review. JAMA
the development and growth of pancreatic beta-cells. Am
289:2254, 2003.
J Physiol Endocrinol Metab 286:E875, 2004.
Dunne MJ, Cosgrove KE, Shepherd RM, et al: Hyperin-
Mann GE, Yudilevich DL, Sobrevia L: Regulation of amino
sulinism in infancy: from basic science to clinical disease.
acid and glucose transporters in endothelial and smooth
Physiol Rev 84:239, 2004.
muscle cells. Physiol Rev 83:183, 2003.
Efrat S: Regulation of insulin secretion: insights from engi-
Perseghin G, Petersen K, Shulman GI: Cellular mechanism
neered beta-cell lines. Ann N Y Acad Sci 1014:88, 2004.
of insulin resistance: potential links with inflammation. Int
Gurnell M, Savage DB, Chatterjee VK, O’Rahilly S: The
J Obes Relat Metab Disord 27(Suppl 3):S6, 2003.
metabolic syndrome: peroxisome proliferator-activated
Pessin JE, Saltiel AR: Signaling pathways in insulin action:
receptor gamma and its therapeutic modulation. J Clin
molecular targets of insulin resistance. J Clin Invest
Endocrinol Metab 88:2412, 2003
106:165, 2000.
Grundy SM, Brewer HB Jr, Cleeman JI, et al: Definition of
Roden M: How free fatty acids inhibit glucose utilization in
metabolic syndrome: Report of the National Heart, Lung,
human skeletal muscle. News Physiol Sci 19:92, 2004.
and Blood Institute/American Heart Association confer-
Saltiel AR: Putting the brakes on insulin signaling. N Engl J
ence on scientific issues related to definition. Circulation
Med 349:2560, 2003.
109:433, 2004.
Shi Y, Taylor SI, Tan SL, Sonenberg N: When translation
Hall JE, Summers RL, Brands MW, et al: Resistance to the
meets metabolism: multiple links to diabetes. Endocr Rev
metabolic actions of insulin and its role in hypertension.
24:91, 2003.
Am J Hypertens 7:772, 1994.
Ten S, Maclaren N: Insulin resistance syndrome in children.
Hattersley AT: Unlocking the secrets of the pancreatic beta
J Clin Endocrinol Metab 89:2526, 2004.
cell: man and mouse provide the key. J Clin Invest 114:314,
Wilson PW, Grundy SM: The metabolic syndrome: practical
2004.
guide to origins and treatment: Part I. Circulation
Holst JJ, Gromada J: Role of incretin hormones in the reg-
108:1422, 2003.
ulation of insulin secretion in diabetic and nondiabetic
humans. Am J Physiol Endocrinol Metab 287:E199, 2004.
C H A P T E R
7
9
Parathyroid Hormone, Calcitonin,
Calcium and Phosphate
Metabolism, Vitamin D, Bone,
and Teeth
The physiology of calcium and phosphate metabo-
lism, formation of bone and teeth, and regulation of
vitamin D, parathyroid hormone (PTH), and calci-
tonin are all closely intertwined. Extracellular
calcium ion concentration, for example, is deter-
mined by the interplay of calcium absorption from
the intestine, renal excretion of calcium, and bone
uptake and release of calcium, each of which is reg-
ulated by the hormones just noted. Because phosphate homeostasis and calcium
homeostasis are closely associated, they are discussed together in this chapter.
Overview of Calcium and Phosphate
Regulation in the Extracellular Fluid
and Plasma
Extracellular fluid calcium concentration normally is regulated very precisely,
seldom rising or falling more than a few per cent from the normal value of about
9.4 mg/dl, which is equivalent to 2.4 mmol calcium per liter. This precise control
is essential, because calcium plays a key role in many physiologic processes,
including contraction of skeletal, cardiac, and smooth muscles; blood clotting;
and transmission of nerve impulses, to name just a few. Excitable cells, such as
neurons, are very sensitive to changes in calcium ion concentrations, and
increases in calcium ion concentration above normal (hypercalcemia) cause pro-
gressive depression of the nervous system; conversely, decreases in calcium con-
centration (hypocalcemia) cause the nervous system to become more excited.
An important feature of extracellular calcium regulation is that only about
0.1 per cent of the total body calcium is in the extracellular fluid, about 1 per
cent is in the cells, and the rest is stored in bones. Therefore, the bones can serve
as large reservoirs, releasing calcium when extracellular fluid concentration
decreases and storing excess calcium.
Approximately 85 per cent of the body’s phosphate is stored in bones, 14 to
15 per cent is in the cells, and less than 1 per cent is in the extracellular fluid.
Although extracellular fluid phosphate concentration is not nearly as well reg-
ulated as calcium concentration, phosphate serves several important functions
and is controlled by many of the same factors that regulate calcium.
Calcium in the Plasma and Interstitial Fluid
The calcium in the plasma is present in three forms, as shown in Figure 79-1.
(1) About 41 per cent (1 mmol/L) of the calcium is combined with the plasma
proteins and in this form is nondiffusible through the capillary membrane.
(2) About 9 per cent of the calcium (0.2 mmol/L) is diffusible through the cap-
illary membrane but is combined with anionic substances of the plasma and
interstitial fluids (citrate and phosphate, for instance) in such a manner that it
978
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
979
Calcium complexed to
anions 9% (0.2 mmol/L)
Ionized calcium
Protein-bound calcium
50%
41%
(1.2 mmol/L)
(1.0 mmol/L)
Figure 79-2
Figure 79-1
Hypocalcemic tetany in the hand, called carpopedal spasm.
Distribution of ionized calcium (Ca++), diffusible but un-ionized
calcium complexed to anions, and nondiffusible protein-bound
calcium in blood plasma.
Non-Bone Physiologic Effects
of Altered Calcium and Phosphate
Concentrations in the Body Fluids
is not ionized. (3) The remaining 50 per cent of the
calcium in the plasma is both diffusible through the
Changing the level of phosphate in the extracellular
capillary membrane and ionized.
fluid from far below normal to two to three times
Thus, the plasma and interstitial fluids have a normal
normal does not cause major immediate effects on the
calcium ion concentration of about 1.2 mmol/L (or
body. In contrast, even slight increases or decreases of
2.4 mEq/L, because it is a divalent ion), a level only
calcium ion in the extracellular fluid can cause extreme
one half the total plasma calcium concentration.
immediate physiologic effects. In addition, chronic
This ionic calcium is the form that is important for
hypocalcemia or hypophosphatemia greatly decreases
most functions of calcium in the body, including the
bone mineralization, as explained later in the chapter.
effect of calcium on the heart, the nervous system, and
bone formation.
Hypocalcemia Causes Nervous System Excitement and Tetany. When
the extracellular fluid concentration of calcium ions
falls below normal, the nervous system becomes
Inorganic Phosphate in the
progressively more excitable, because this causes
Extracellular Fluids
increased neuronal membrane permeability to sodium
ions, allowing easy initiation of action potentials. At
Inorganic phosphate in the plasma is mainly in two
plasma calcium ion concentrations about 50 per cent
forms: HPO4- and H2PO4-. The concentration of
below normal, the peripheral nerve fibers become so
HPO4- is about 1.05 mmol/L, and the concentration of
excitable that they begin to discharge spontaneously,
H2PO4- is about 0.26 mmol/L.When the total quantity
initiating trains of nerve impulses that pass to the
of phosphate in the extracellular fluid rises, so does the
peripheral skeletal muscles to elicit tetanic muscle
quantity of each of these two types of phosphate ions.
contraction. Consequently, hypocalcemia causes
Furthermore, when the pH of the extracellular fluid
tetany. It also occasionally causes seizures because of
becomes more acidic, there is a relative increase in
its action of increasing excitability in the brain.
H2PO4_ and a decrease in HPO4-, whereas the oppo-
Figure 79-2 shows tetany in the hand, which usually
site occurs when the extracellular fluid becomes alka-
occurs before tetany develops in most other parts of
line. These relations were presented in the discussion
the body. This is called “carpopedal spasm.”
of acid-base balance in Chapter 30.
Tetany ordinarily occurs when the blood concentra-
Because it is difficult to determine chemically the
tion of calcium falls from its normal level of 9.4 mg/dl
exact quantities of HPO4- and H2PO4- in the blood,
to about 6 mg/dl, which is only 35 per cent below the
ordinarily the total quantity of phosphate is expressed
normal calcium concentration, and it is usually lethal
in terms of milligrams of phosphorus per deciliter
at about 4 mg/dl.
(100 ml) of blood. The average total quantity of inor-
In laboratory animals, in which calcium can gradu-
ganic phosphorus represented by both phosphate ions
ally be reduced beyond the usual lethal levels, very
is about 4 mg/dl, varying between normal limits of 3 to
extreme hypocalcemia can cause other effects that are
4 mg/dl in adults and 4 to 5 mg/dl in children.
seldom evident in patients, such as marked dilatation
980
Unit XIV Endocrinology and Reproduction
of the heart, changes in cellular enzyme activities,
Calcium
increased membrane permeability in some cells (in
intake
Cells
(350 mg/day)
(13,000 mg)
addition to nerve cells), and impaired blood clotting.
Bone
(1,000,000 mg)
Hypercalcemia Depresses Nervous System and Muscle Activity.
When the level of calcium in the body fluids rises
Absorption
Deposition
(350 mg/day)
(500 mg/day)
above normal, the nervous system becomes depressed
Extracellular
and reflex activities of the central nervous system are
fluid
sluggish. Also, increased calcium ion concentration
(1300 mg)
Secretion
Absorption
decreases the QT interval of the heart and causes
(250 mg/day)
(500 mg/day)
lack of appetite and constipation, probably because of
Filtration
Reabsorption
depressed contractility of the muscle walls of the gas-
(9980 mg/day)
(9880 mg/day)
trointestinal tract.
Feces
(900 mg/day)
Kidneys
These depressive effects begin to appear when the
Urine
blood level of calcium rises above about 12 mg/dl, and
(100 mg/day)
they can become marked as the calcium level rises
above 15 mg/dl. When the level of calcium rises above
about 17 mg/dl in the blood, calcium phosphate crys-
Figure 79-3
tals are likely to precipitate throughout the body; this
Overview of calcium exchange between different tissue compart-
condition is discussed later in connection with
ments in a person ingesting 1000 mg of calcium per day. Note
parathyroid poisoning.
that most of the ingested calcium is normally eliminated in the
feces, although the kidneys have the capacity to excrete large
amounts by reducing tubular reabsorption of calcium.
Absorption and Excretion of Calcium
and Phosphate
selective, depending on the calcium ion concentration
in the blood.
Intestinal Absorption and Fecal Excretion of Calcium and Phos-
When calcium concentration is low, this reabsorp-
phate. The usual rates of intake are about 1000 mg/day
tion is great, so that almost no calcium is lost in the
each for calcium and phosphorus, about the amounts
urine. Conversely, even a minute increase in blood
in 1 liter of milk. Normally, divalent cations such as
calcium ion concentration above normal increases
calcium ions are poorly absorbed from the intestines.
calcium excretion markedly. We shall see later in the
However, as discussed later, vitamin D promotes
chapter that the most important factor controlling this
calcium absorption by the intestines, and about 35 per
reabsorption of calcium in the distal portions of the
cent (350 mg/day) of the ingested calcium is usually
nephron, and therefore controlling the rate of calcium
absorbed; the calcium remaining in the intestine is
excretion, is PTH.
excreted in the feces. An additional 250 mg/day of
Renal phosphate excretion is controlled by an over-
calcium enters the intestines via secreted gastroin-
flow mechanism, as explained in Chapter 29. That is,
testinal juices and sloughed mucosal cells. Thus, about
when phosphate concentration in the plasma is below
90 per cent (900 mg/day) of the daily intake of calcium
the critical value of about 1 mmol/L, all the phosphate
is excreted in the feces (Figure 79-3).
in the glomerular filtrate is reabsorbed and no phos-
Intestinal absorption of phosphate occurs very
phate is lost in the urine. But above this critical con-
easily. Except for the portion of phosphate that is
centration, the rate of phosphate loss is directly
excreted in the feces in combination with nonabsorbed
proportional to the additional increase. Thus, the
calcium, almost all the dietary phosphate is absorbed
kidneys regulate the phosphate concentration in the
into the blood from the gut and later excreted in the
extracellular fluid by altering the rate of phosphate
urine.
excretion in accordance with the plasma phosphate
concentration and the rate of phosphate filtration by
Renal Excretion of Calcium and Phosphate. Approximately
the kidneys.
10 per cent (100 mg/day) of the ingested calcium is
However, as discussed later in the chapter, PTH can
excreted in the urine. About 41 per cent of the plasma
greatly increase phosphate excretion by the kidneys,
calcium is bound to plasma proteins and is therefore
thereby playing an important role in the control of
not filtered by the glomerular capillaries. The rest is
plasma phosphate concentration as well as calcium
combined with anions such as phosphate (9 per cent)
concentration.
or ionized (50 per cent) and is filtered through the
glomeruli into the renal tubules.
Normally, the renal tubules reabsorb 99 per cent of
Bone and Its Relation
the filtered calcium, and about 100 mg/day is excreted
in the urine. Approximately 90 per cent of the calcium
to Extracellular Calcium
in the glomerular filtrate is reabsorbed in the proximal
and Phosphate
tubules, loops of Henle, and early distal tubules.
Then in the late distal tubules and early collecting
Bone is composed of a tough organic matrix that is
ducts, reabsorption of the remaining 10 per cent is very
greatly strengthened by deposits of calcium salts.
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
981
Average compact bone contains by weight about 30
properties plus the degree of bondage between the
per cent matrix and 70 per cent salts. Newly formed
collagen fibers and the crystals provide a bony struc-
bone may have a considerably higher percentage of
ture that has both extreme tensile strength and
matrix in relation to salts.
extreme compressional strength.
Organic Matrix of Bone. The organic matrix of bone is 90
Precipitation and Absorption
to 95 per cent collagen fibers, and the remainder is a
homogeneous gelatinous medium called ground sub-
of Calcium and Phosphate
stance. The collagen fibers extend primarily along the
in Bone—Equilibrium with the
lines of tensional force and give bone its powerful
Extracellular Fluids
tensile strength.
The ground substance is composed of extracellular
Hydroxyapatite Does Not Precipitate in Extracellular Fluid
fluid plus proteoglycans, especially chondroitin sulfate
Despite Supersaturation of Calcium and Phosphate Ions. The
and hyaluronic acid. The precise function of each of
concentrations of calcium and phosphate ions in extra-
these is not known, although they do help to control
cellular fluid are considerably greater than those
the deposition of calcium salts.
required to cause precipitation of hydroxyapatite.
However, inhibitors are present in almost all tissues of
Bone Salts. The crystalline salts deposited in the
the body as well as in plasma to prevent such precipi-
organic matrix of bone are composed principally of
tation; one such inhibitor is pyrophosphate. Therefore,
calcium and phosphate. The formula for the major
hydroxyapatite crystals fail to precipitate in normal
crystalline salt, known as hydroxyapatite, is the
tissues except in bone despite the state of supersatu-
following:
ration of the ions.
Ca10 (PO4)6(OH)2
Mechanism of Bone Calcification. The initial stage in bone
Each crystal—about 400 angstroms long, 10 to 30
production is the secretion of collagen molecules
angstroms thick, and 100 angstroms wide—is shaped
(called collagen monomers) and ground substance
like a long, flat plate. The relative ratio of calcium to
(mainly proteoglycans) by osteoblasts. The collagen
phosphorus can vary markedly under different nutri-
monomers polymerize rapidly to form collagen fibers;
tional conditions, the Ca/P ratio on a weight basis
the resultant tissue becomes osteoid, a cartilage-like
varying between 1.3 and 2.0.
material differing from cartilage in that calcium salts
Magnesium, sodium, potassium, and carbonate ions
readily precipitate in it. As the osteoid is formed, some
are also present among the bone salts, although x-ray
of the osteoblasts become entrapped in the osteoid
diffraction studies fail to show definite crystals formed
and become quiescent. At this stage they are called
by them. Therefore, they are believed to be conjugated
osteocytes.
to the hydroxyapatite crystals rather than organized
Within a few days after the osteoid is formed,
into distinct crystals of their own. This ability of many
calcium salts begin to precipitate on the surfaces of the
types of ions to conjugate to bone crystals extends to
collagen fibers. The precipitates first appear at inter-
many ions normally foreign to bone, such as strontium,
vals along each collagen fiber, forming minute nidi
uranium, plutonium, the other transuranic elements,
that rapidly multiply and grow over a period of days
lead, gold, other heavy metals, and at least 9 of 14 of
and weeks into the finished product, hydroxyapatite
the major radioactive products released by explosion of
crystals.
the hydrogen bomb. Deposition of radioactive sub-
The initial calcium salts to be deposited are not
stances in the bone can cause prolonged irradiation
hydroxyapatite crystals but amorphous compounds
of the bone tissues, and if a sufficient amount is
(noncrystalline), a mixture of salts such as CaHPO4 ·
deposited, an osteogenic sarcoma (bone cancer) even-
2H2O, Ca3(PO4)2 · 3H2O, and others. Then by a process
tually develops in most cases.
of substitution and addition of atoms, or reabsorption
and reprecipitation, these salts are converted into
Tensile and Compressional Strength of Bone. Each collagen
the hydroxyapatite crystals over a period of weeks or
fiber of compact bone is composed of repeating peri-
months. A few per cent may remain permanently in
odic segments every 640 angstroms along its length;
the amorphous form. This is important because
hydroxyapatite crystals lie adjacent to each segment of
these amorphous salts can be absorbed rapidly when
the fiber, bound tightly to it. This intimate bonding pre-
there is need for extra calcium in the extracellular
vents “shear” in the bone; that is, it prevents the crys-
fluid.
tals and collagen fibers from slipping out of place,
The mechanism that causes calcium salts to be
which is essential in providing strength to the bone. In
deposited in osteoid is not fully understood. One
addition, the segments of adjacent collagen fibers
theory holds that at the time of formation, the colla-
overlap one another, also causing hydroxyapatite crys-
gen fibers are specially constituted in advance for
tals to be overlapped like bricks keyed to one another
causing precipitation of calcium salts. The osteoblasts
in a brick wall.
supposedly also secrete a substance into the osteoid to
The collagen fibers of bone, like those of tendons,
neutralize an inhibitor (believed to be pyrophosphate)
have great tensile strength, whereas the calcium salts
that normally prevents hydroxyapatite crystallization.
have great compressional strength. These combined
Once the pyrophosphate has been neutralized, the
982
Unit XIV Endocrinology and Reproduction
natural affinity of the collagen fibers for calcium salts
Osteoblasts
Fibrous periosteum
causes the precipitation.
Precipitation of Calcium in Nonosseous Tissues Under
Abnormal Conditions. Although calcium salts almost
never precipitate in normal tissues besides bone, under
abnormal conditions, they do precipitate. For instance,
they precipitate in arterial walls in the condition called
arteriosclerosis and cause the arteries to become bone-
like tubes. Likewise, calcium salts frequently deposit in
degenerating tissues or in old blood clots. Presumably,
Osteoclasts
in these instances, the inhibitor factors that normally
Vein
prevent deposition of calcium salts disappear from the
tissues, thereby allowing precipitation.
Bone
Calcium Exchange Between Bone and
Extracellular Fluid
If soluble calcium salts are injected intravenously, the
Figure 79-4
calcium ion concentration may increase immediately
to high levels. However, within 30 minutes to 1 hour
Osteoblastic and osteoclastic activity in the same bone.
or more, the calcium ion concentration returns to
normal. Likewise, if large quantities of calcium ions are
removed from the circulating body fluids, the calcium
monocyte-like cells formed in the bone marrow. The
ion concentration again returns to normal within 30
osteoclasts are normally active on less than 1 per cent
minutes to about 1 hour. These effects result in great
of the bone surfaces of an adult. Later in the chapter we
part from the fact that the bone contains a type of
see that PTH controls the bone absorptive activity of
exchangeable calcium that is always in equilibrium
osteoclasts.
with the calcium ions in the extracellular fluids.
Histologically, bone absorption occurs immediately
A small portion of this exchangeable calcium is also
adjacent to the osteoclasts. The mechanism of this
the calcium found in all tissue cells, especially in highly
absorption is believed to be the following: The osteo-
permeable types of cells such as those of the liver
clasts send out villus-like projections toward the bone,
and the gastrointestinal tract. However, most of the
forming a so-called ruffled border adjacent to the bone.
The villi secrete two types of substances: (1) proteolytic
exchangeable calcium is in the bone. It normally
enzymes, released from the lysosomes of the osteoclasts,
amounts to about 0.4 to 1 per cent of the total bone
and (2) several acids, including citric acid and lactic acid,
calcium. This calcium is deposited in the bones in a
released from the mitochondria and secretory vesicles.
form of readily mobilizable salt such as CaHPO4 and
The enzymes digest or dissolve the organic matrix of the
other amorphous calcium salts.
bone, and the acids cause solution of the bone salts. The
The importance of exchangeable calcium is that it
osteoclastic cells also imbibe by phagocytosis minute
provides a rapid buffering mechanism to keep the
particles of bone matrix and crystals, eventually also dis-
calcium ion concentration in the extracellular fluids
soluting these and releasing the products into the blood.
from rising to excessive levels or falling to very low
levels under transient conditions of excess or
Bone Deposition and Absorption Are Normally in Equilibrium.
Normally, except in growing bones, the rates of bone
decreased availability of calcium.
deposition and absorption are equal to each other, so
that the total mass of bone remains constant. Osteo-
Deposition and Absorption of
clasts usually exist in small but concentrated masses, and
Bone—Remodeling of Bone
once a mass of osteoclasts begins to develop, it usually
eats away at the bone for about 3 weeks, creating a
Deposition of Bone by the Osteoblasts. Bone is continually
tunnel that ranges in diameter from 0.2 to 1 millimeter
being deposited by osteoblasts, and it is continually
and is several millimeters long. At the end of this time,
being absorbed where osteoclasts are active (Figure
the osteoclasts disappear and the tunnel is invaded by
79-4). Osteoblasts are found on the outer surfaces of
osteoblasts instead; then new bone begins to develop.
the bones and in the bone cavities. A small amount of
Bone deposition then continues for several months, the
osteoblastic activity occurs continually in all living
new bone being laid down in successive layers of con-
bones (on about 4 per cent of all surfaces at any given
centric circles (lamellae) on the inner surfaces of the
time in an adult), so that at least some new bone is being
cavity until the tunnel is filled. Deposition of new bone
formed constantly.
ceases when the bone begins to encroach on the blood
vessels supplying the area. The canal through which
Absorption of Bone—Function of the Osteoclasts. Bone is also
these vessels run, called the haversian canal, is all that
being continually absorbed in the presence of osteo-
remains of the original cavity. Each new area of bone
clasts, which are large phagocytic, multinucleated cells
deposited in this way is called an osteon, as shown in
(as many as 50 nuclei), derivatives of monocytes or
Figure 79-5.
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
983
straight, especially in children because of the rapid
remodeling of bone at younger ages.
Epiphyseal line
Repair of a Fracture Activates Osteoblasts. Fracture of a bone
in some way maximally activates all the periosteal and
intraosseous osteoblasts involved in the break. Also,
Magnified
immense numbers of new osteoblasts are formed almost
section
immediately from osteoprogenitor cells, which are bone
Osteon
stem cells in the surface tissue lining bone, called the
“bone membrane.” Therefore, within a short time, a
Haversian
large bulge of osteoblastic tissue and new organic bone
canal
matrix, followed shortly by the deposition of calcium
salts, develops between the two broken ends of the
bone. This is called a callus.
Many bone surgeons use the phenomenon of bone
stress to accelerate the rate of fracture healing. This is
done by use of special mechanical fixation apparatuses
for holding the ends of the broken bone together so that
Canaliculi
the patient can continue to use the bone immediately.
Lacunae
This causes stress on the opposed ends of the broken
bones, which accelerates osteoblastic activity at the
break and often shortens convalescence.
Epiphyseal line
Vitamin D
Figure 79-5
Vitamin D has a potent effect to increase calcium
absorption from the intestinal tract; it also has impor-
Structure of bone.
tant effects on both bone deposition and bone absorp-
tion, as discussed later. However, vitamin D itself is not
the active substance that actually causes these effects.
Value of Continual Bone Remodeling The continual deposi-
tion and absorption of bone have several physiologi-
Instead, vitamin D must first be converted through a
cally important functions. First, bone ordinarily adjusts
succession of reactions in the liver and the kidneys to
its strength in proportion to the degree of bone stress.
the final active product, 1,25-dihydroxycholecalciferol,
Consequently, bones thicken when subjected to heavy
also called 1,25(OH)2D3. Figure 79-6 shows the suc-
loads. Second, even the shape of the bone can be
cession of steps that lead to the formation of this sub-
rearranged for proper support of mechanical forces by
stance from vitamin D. Let us discuss these steps.
deposition and absorption of bone in accordance with
stress patterns. Third, because old bone becomes rela-
Cholecalciferol (Vitamin D3) Is Formed in the Skin. Several
tively brittle and weak, new organic matrix is needed as
compounds derived from sterols belong to the vitamin
the old organic matrix degenerates. In this manner, the
D family, and they all perform more or less the same
normal toughness of bone is maintained. Indeed, the
bones of children, in whom the rates of deposition and
functions. Vitamin D3 (also called cholecalciferol) is
absorption are rapid, show little brittleness in compari-
the most important of these and is formed in the skin
son with the bones of the elderly, in whom the rates of
as a result of irradiation of 7-dehydrocholesterol, a sub-
deposition and absorption are slow.
stance normally in the skin, by ultraviolet rays from
the sun. Consequently, appropriate exposure to the
Control of the Rate of Bone Deposition by Bone “Stress.” Bone
sun prevents vitamin D deficiency. The additional
is deposited in proportion to the compressional load
vitamin D compounds that we ingest in food are iden-
that the bone must carry. For instance, the bones of
tical to the cholecalciferol formed in the skin, except
athletes become considerably heavier than those of
for the substitution of one or more atoms that do not
nonathletes. Also, if a person has one leg in a cast but
affect their function.
continues to walk on the opposite leg, the bone of the
leg in the cast becomes thin and as much as 30 per cent
decalcified within a few weeks, whereas the opposite
Cholecalciferol Is Converted to 25-Hydroxycholecalciferol in
bone remains thick and normally calcified. Therefore,
the Liver. The first step in the activation of cholecal-
continual physical stress stimulates osteoblastic deposi-
ciferol is to convert it to 25-hydroxycholecalciferol;
tion and calcification of bone.
this occurs in the liver. The process is a limited one,
Bone stress also determines the shape of bones under
because the 25-hydroxycholecalciferol has a feedback
certain circumstances. For instance, if a long bone of the
inhibitory effect on the conversion reactions. This
leg breaks in its center and then heals at an angle, the
feedback effect is extremely important for two
compression stress on the inside of the angle causes
reasons.
increased deposition of bone, and increased absorption
First, the feedback mechanism precisely regulates
occurs on the outer side of the angle where the bone is
not compressed. After many years of increased deposi-
the concentration of 25-hydroxycholecalciferol in the
tion on the inner side of the angulated bone and absorp-
plasma, an effect that is shown in Figure 79-7. Note
tion on the outer side, the bone can become almost
that the intake of vitamin D3 can increase many times,
984
Unit XIV Endocrinology and Reproduction
Skin
6
Cholecalciferol (vitamin D3)
5
Liver
4
Inhibition
25-Hydroxycholecalciferol
3
Kidney
2
Normal
Activation
Parathyroid
1
hormone
0
1,25-Dihydroxycholecalciferol
0
2
4
6
8
10
12
14
16
Plasma calcium (mg/100 mL)
Intestinal
epithelium
Figure 79-8
Calcium-
Calcium-
Alkaline
Effect of plasma calcium concentration on the plasma concentra-
binding
stimulated
phosphatase
tion of
1,25-dihydroxycholecalciferol. This figure shows that a
protein
ATPase
slight decrease in calcium concentration below normal causes
increased formation of activated vitamin D, which in turn leads to
Inhibition
greatly increased absorption of calcium from the intestine.
Intestinal absorption of calcium
Plasma calcium ion concentration
and yet the concentration of 25-hydroxycholecalcif-
erol remains nearly normal. This high degree of feed-
back control prevents excessive action of vitamin D
when intake of vitamin D3 is altered over a wide range.
Figure 79-6
Second, this controlled conversion of vitamin D3 to
25-hydroxycholecalciferol conserves the vitamin D
Activation of vitamin D3
to form
1,25-dihydroxycholecalciferol
and the role of vitamin D in controlling the plasma calcium
stored in the liver for future use. Once it is converted,
concentration.
it persists in the body for only a few weeks, whereas in
the vitamin D form, it can be stored in the liver for
many months.
Formation of
1,25-Dihydroxycholecalciferol in the Kidneys
and Its Control by Parathyroid Hormone. Figure
79-6
1.2
Normal range
also shows the conversion in the proximal tubules
1.0
of the kidneys of 25-hydroxycholecalciferol to 1,25-
dihydroxycholecalciferol. This latter substance is by far
0.8
the most active form of vitamin D, because the previ-
ous products in the scheme of Figure 79-6 have less
0.6
than 1/1000 of the vitamin D effect. Therefore, in the
0.4
absence of the kidneys, vitamin D loses almost all its
effectiveness.
0.2
Note also in Figure 79-6 that the conversion of 25-
hydroxycholecalciferol to
1,25-dihydroxycholecalcif-
0
erol requires PTH. In the absence of PTH, almost
0
0.5
1.0
1.5
2.0
2.5
none of the 1,25-dihydroxycholecalciferol is formed.
Intake of vitamin D3 (times normal)
Therefore, PTH exerts a potent influence in determin-
ing the functional effects of vitamin D in the body.
Figure 79-7
Calcium Ion Concentration Controls the Formation of 1,25-
Dihydroxycholecalciferol. Figure 79-8 demonstrates that
intake on the plasma concentration
Effect of increasing vitamin D3
the plasma concentration of 1,25-dihydroxycholecal-
of 25-hydroxycholecalciferol. This figure shows that tremendous
changes in vitamin D intake have little effect on the final quantity
ciferol is inversely affected by the concentration of
of activated vitamin D that is formed.
calcium in the plasma. There are two reasons for this.
First, the calcium ion itself has a slight effect in pre-
venting the conversion of 25-hydroxycholecalciferol to
1,25-dihydroxycholecalciferol. Second, and even more
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
985
important, as we shall see later in the chapter, the rate
thereby tending to decrease excretion of these sub-
of secretion of PTH is greatly suppressed when the
stances in the urine. However, this is a weak effect
plasma calcium ion concentration rises above 9 to
and probably not of major importance in regulating
10 mg/100 ml. Therefore, at calcium concentrations
the extracellular fluid concentration of these
below this level, PTH promotes the conversion of 25-
substances.
hydroxycholecalciferol to
1,25-dihydroxycholecalcif-
erol in the kidneys. At higher calcium concentrations,
Effect of Vitamin D on Bone and Its Relation to Parathyroid
when PTH is suppressed, the 25-hydroxycholecalcif-
Hormone Activity. Vitamin D plays important roles in
erol is converted to a different compound—24,25-
both bone absorption and bone deposition. The
dihydroxycholecalciferol—that has almost no vitamin
administration of extreme quantities of vitamin D
D effect.
causes absorption of bone. In the absence of vitamin
When the plasma calcium concentration is already
D, the effect of PTH in causing bone absorption (dis-
too high, the formation of 1,25-dihydroxycholecalcif-
cussed in the next section) is greatly reduced or even
erol is greatly depressed. Lack of this in turn decreases
prevented. The mechanism of this action of vitamin D
the absorption of calcium from the intestines, the
is not known, but it is believed to result from the effect
bones, and the renal tubules, thus causing the calcium
of 1,25-dihydroxycholecalciferol to increase calcium
ion concentration to fall back toward its normal level.
transport through cellular membranes.
Vitamin D in smaller quantities promotes bone cal-
cification. One of the ways in which it does this is to
Actions of Vitamin D
increase calcium and phosphate absorption from the
intestines. However, even in the absence of such
The active form of vitamin D, 1,25-dihydroxycholecal-
increase, it enhances the mineralization of bone. Here
ciferol, has several effects on the intestines, kidneys,
again, the mechanism of the effect is unknown, but it
and bones that increase absorption of calcium and
probably also results from the ability of 1,25-dihy-
phosphate into the extracellular fluid and contribute
droxycholecalciferol to cause transport of calcium ions
to feedback regulation of these substances.
through cell membranes—but in this instance, perhaps
in the opposite direction through the osteoblastic or
“Hormonal” Effect of Vitamin D to Promote Intestinal Calcium
osteocytic cell membranes.
Absorption.
1,25-Dihydroxycholecalciferol itself func-
tions as a type of “hormone” to promote intestinal
absorption of calcium. It does this principally by
Parathyroid Hormone
increasing, over a period of about 2 days, formation of
a calcium-binding protein in the intestinal epithelial
Parathyroid hormone provides a powerful mechanism
cells. This protein functions in the brush border of
for controlling extracellular calcium and phosphate
these cells to transport calcium into the cell cytoplasm,
concentrations by regulating intestinal reabsorption,
and the calcium then moves through the basolateral
renal excretion, and exchange between the extracellu-
membrane of the cell by facilitated diffusion. The rate
lar fluid and bone of these ions. Excess activity of the
of calcium absorption is directly proportional to the
parathyroid gland causes rapid absorption of calcium
quantity of this calcium-binding protein. Furthermore,
salts from the bones, with resultant hypercalcemia in
this protein remains in the cells for several weeks after
the extracellular fluid; conversely, hypofunction of the
the 1,25-dihydroxycholecalciferol has been removed
parathyroid glands causes hypocalcemia, often with
from the body, thus causing a prolonged effect on
resultant tetany.
calcium absorption.
Other effects of 1,25-dihydroxycholecalciferol that
Physiologic Anatomy of the Parathyroid Glands. Normally
might play a role in promoting calcium absorption are
there are four parathyroid glands in humans; they are
the formation of (1) a calcium-stimulated ATPase in
located immediately behind the thyroid gland—one
the brush border of the epithelial cells and (2) an alka-
behind each of the upper and each of the lower poles
line phosphatase in the epithelial cells. The precise
of the thyroid. Each parathyroid gland is about 6 mil-
details of all these effects are unclear.
limeters long, 3 millimeters wide, and 2 millimeters
thick and has a macroscopic appearance of dark brown
Vitamin D Promotes Phosphate Absorption by the Intestines.
fat. The parathyroid glands are difficult to locate
Although phosphate is usually absorbed easily, phos-
during thyroid operations because they often look like
phate flux through the gastrointestinal epithelium is
just another lobule of the thyroid gland. For this
enhanced by vitamin D. It is believed that this results
reason, before the importance of these glands was gen-
from a direct effect of 1,25-dihydroxycholecalciferol,
erally recognized, total or subtotal thyroidectomy fre-
but it is possible that it results secondarily from this
quently resulted in removal of the parathyroid glands
hormone’s action on calcium absorption, the cal-
as well.
cium in turn acting as a transport mediator for the
Removal of half the parathyroid glands usually
phosphate.
causes no major physiologic abnormalities. However,
Vitamin D Decreases Renal Calcium and Phosphate Excretion.
removal of three of the four normal glands causes
Vitamin D also increases calcium and phosphate
transient hypoparathyroidism. But even a small quan-
absorption by the epithelial cells of the renal tubules,
tity of remaining parathyroid tissue is usually capable
986
Unit XIV Endocrinology and Reproduction
Begin parathyroid hormone
Parathyroid gland
(located on posterior
Calcium
2.40
Thyroid gland
side of the thyroid
2.35
gland)
2.30
1.2
Phosphate
1.0
0.8
0
1
2
3
4
5
6
Hours
Figure 79-10
Chief cell
Approximate changes in calcium and phosphate concentrations
during the first 5 hours of parathyroid hormone infusion at a mod-
erate rate.
Oxyphil cell
Red blood cell
Effect of Parathyroid Hormone
on Calcium and Phosphate
Concentrations in the
Figure 79-9
Extracellular Fluid
The four parathyroid glands lie immediately behind the thyroid
gland. Almost all of the parathyroid hormone (PTH) is synthesized
Figure 79-10 shows the approximate effects on the
and secreted by the chief cells. The function of the oxyphil cells
blood calcium and phosphate concentrations caused
is uncertain, but they may be modified or depleted chief cells that
by suddenly infusing PTH into an animal and contin-
no longer secrete PTH.
uing this for several hours. Note that at the onset of
infusion the calcium ion concentration begins to rise
and reaches a plateau in about 4 hours. The phosphate
concentration, however, falls more rapidly than the
of hypertrophying satisfactorily to perform the func-
calcium rises and reaches a depressed level within 1 or
tion of all the glands.
2 hours. The rise in calcium concentration is caused
The parathyroid gland of the adult human being,
principally by two effects: (1) an effect of PTH to
shown in Figure 79-9, contains mainly chief cells and
increase calcium and phosphate absorption from the
a small to moderate number of oxyphil cells, but
bone and (2) a rapid effect of PTH to decrease the
oxyphil cells are absent in many animals and in
excretion of calcium by the kidneys. The decline in
young humans. The chief cells are believed to secrete
phosphate concentration is caused by a strong effect
most, if not all, of the PTH. The function of the oxyphil
of PTH to increase renal phosphate excretion, an
cells is not certain, but they are believed to be modi-
effect that is usually great enough to override
fied or depleted chief cells that no longer secrete
increased phosphate absorption from the bone.
hormone.
Parathyroid Hormone Increases Calcium and
Chemistry of Parathyroid Hormone. PTH has been isolated
Phosphate Absorption from the Bone
in a pure form. It is first synthesized on the ribosomes
PTH has two effects on bone in causing absorption of
in the form of a preprohormone, a polypeptide chain
calcium and phosphate. One is a rapid phase that
of 110 amino acids. This is cleaved first to a prohor-
begins in minutes and increases progressively for
mone with 90 amino acids, then to the hormone itself
several hours. This phase results from activation of the
with 84 amino acids by the endoplasmic reticulum and
already existing bone cells (mainly the osteocytes) to
Golgi apparatus, and finally is packaged in secretory
promote calcium and phosphate absorption. The
granules in the cytoplasm of the cells. The final
second phase is a much slower one, requiring several
hormone has a molecular weight of about
9500.
days or even weeks to become fully developed; it
Smaller compounds with as few as 34 amino acids
results from proliferation of the osteoclasts, followed
adjacent to the N terminus of the molecule have also
by greatly increased osteoclastic reabsorption of the
been isolated from the parathyroid glands that exhibit
bone itself, not merely absorption of the calcium phos-
full PTH activity. In fact, because the kidneys rapidly
phate salts from the bone.
remove the whole 84-amino acid hormone within
minutes but fail to remove many of the fragments for
Rapid Phase of Calcium and Phosphate Absorption—Osteoly-
hours, a large share of the hormonal activity is caused
sis. When large quantities of PTH are injected, the
by the fragments.
calcium ion concentration in the blood begins to rise
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
987
within minutes, long before any new bone cells can be
Activation of the osteoclastic system occurs in two
developed. Histological and physiologic studies have
stages: (1) immediate activation of the osteoclasts that
shown that PTH causes removal of bone salts from
are already formed and (2) formation of new osteo-
two areas in the bone: (1) from the bone matrix in the
clasts. Several days of excess PTH usually cause the
vicinity of the osteocytes lying within the bone itself
osteoclastic system to become well developed, but it
and (2) in the vicinity of the osteoblasts along the bone
can continue to grow for months under the influence
surface.
of strong PTH stimulation.
One does not usually think of either osteoblasts or
After a few months of excess PTH, osteoclastic
osteocytes functioning to cause bone salt absorption,
resorption of bone can lead to weakened bones and
because both these types of cells are osteoblastic in
secondary stimulation of the osteoblasts that attempt
nature and normally associated with bone deposition
to correct the weakened state. Therefore, the late
and its calcification. However, studies have shown that
effect is actually to enhance both osteoblastic and
the osteoblasts and osteocytes form a system of inter-
osteoclastic activity. Still, even in the late stages, there
connected cells that spreads all through the bone and
is more bone absorption than bone deposition in the
over all the bone surfaces except the small surface
presence of continued excess PTH.
areas adjacent to the osteoclasts. In fact, long, filmy
Bone contains such great amounts of calcium in
processes extend from osteocyte to osteocyte through-
comparison with the total amount in all the extracel-
out the bone structure, and these processes also
lular fluids (about 1000 times as much) that even when
connect with the surface osteocytes and osteoblasts.
PTH causes a great rise in calcium concentration in the
This extensive system is called the osteocytic mem-
fluids, it is impossible to discern any immediate effect
brane system, and it is believed to provide a membrane
on the bones. Prolonged administration or secretion of
that separates the bone itself from the extracellular
PTH—over a period of many months or years—finally
fluid.
results in very evident absorption in all the bones and
Between the osteocytic membrane and the bone is
even development of large cavities filled with large,
a small amount of bone fluid. Experiments suggest that
multinucleated osteoclasts.
the osteocytic membrane pumps calcium ions from the
bone fluid into the extracellular fluid, creating a
Parathyroid Hormone Decreases Calcium
calcium ion concentration in the bone fluid only one
Excretion and Increases Phosphate Excretion
third that in the extracellular fluid. When the osteo-
by the Kidneys
cytic pump becomes excessively activated, the bone
Administration of PTH causes rapid loss of phosphate
fluid calcium concentration falls even lower, and
in the urine owing to the effect of the hormone to
calcium phosphate salts are then absorbed from the
diminish proximal tubular reabsorption of phosphate
bone. This effect is called osteolysis, and it occurs
ions.
without absorption of the bone’s fibrous and gel
PTH also increases renal tubular reabsorption of
matrix. When the pump is inactivated, the bone
calcium at the same time that it diminishes phosphate
fluid calcium concentration rises to a higher level,
reabsorption. Moreover, it increases the rate of reab-
and calcium phosphate salts are redeposited in the
sorption of magnesium ions and hydrogen ions while
matrix.
it decreases the reabsorption of sodium, potassium,
But where does PTH fit into this picture? First,
and amino acid ions in much the same way that it
the cell membranes of both the osteoblasts and the
affects phosphate. The increased calcium absorption
osteocytes have receptor proteins for binding PTH.
occurs mainly in the late distal tubules, the collecting
PTH can activate the calcium pump strongly, thereby
tubules, the early collecting ducts, and possibly the
causing rapid removal of calcium phosphate salts from
ascending loop of Henle to a lesser extent.
those amorphous bone crystals that lie near the cells.
Were it not for the effect of PTH on the kidneys
PTH is believed to stimulate this pump by increasing
to increase calcium reabsorption, continual loss of
the calcium permeability of the bone fluid side of the
calcium into the urine would eventually deplete both
osteocytic membrane, thus allowing calcium ions to
the extracellular fluid and the bones of this mineral.
diffuse into the membrane cells from the bone fluid.
Then the calcium pump on the other side of the cell
Parathyroid Hormone Increases Intestinal
membrane transfers the calcium ions the rest of the
Absorption of Calcium and Phosphate
way into the extracellular fluid.
At this point, we should be reminded again that PTH
greatly enhances both calcium and phosphate absorp-
Slow Phase of Bone Absorption and Calcium Phosphate
tion from the intestines by increasing the formation in
Release—Activation of the Osteoclasts. A much better
the kidneys of
1,25-dihydroxycholecalciferol from
known effect of PTH and one for which the evidence
vitamin D, as discussed earlier in the chapter.
is much clearer is its activation of the osteoclasts. Yet
the osteoclasts do not themselves have membrane
Cyclic Adenosine Monophosphate Mediates the Effects of
receptor proteins for PTH. Instead, it is believed that
Parathyroid Hormone. A large share of the effect of PTH
the activated osteoblasts and osteocytes send a sec-
on its target organs is mediated by the cyclic adeno-
ondary but unknown
“signal” to the osteoclasts,
sine monophosphate
(cAMP) second messenger
causing them to set about their usual task of gobbling
mechanism. Within a few minutes after PTH adminis-
up the bone over a period of weeks or months.
tration, the concentration of cAMP increases in the
988
Unit XIV Endocrinology and Reproduction
osteocytes, osteoclasts, and other target cells. This
PTH concentration. The solid red curve shows the
cAMP in turn is probably responsible for such func-
acute effect when the calcium concentration is
tions as osteoclastic secretion of enzymes and acids to
changed over a period of a few hours. This shows that
cause bone reabsorption and formation of
1,25-
even small decreases in calcium concentration from
dihydroxycholecalciferol in the kidneys. There are
the normal value can double or triple the plasma PTH.
probably other direct effects of PTH that function
The approximate chronic effect that one finds when
independently of the second messenger mechanism.
the calcium ion concentration changes over a period
of many weeks, thus allowing time for the glands to
hypertrophy greatly, is shown by the dashed red line;
Control of Parathyroid Secretion
this demonstrates that a decrease of only a fraction of
by Calcium Ion Concentration
a milligram per deciliter in plasma calcium concentra-
tion can double PTH secretion. This is the basis of the
Even the slightest decrease in calcium ion concentra-
body’s extremely potent feedback system for long-
tion in the extracellular fluid causes the parathyroid
term control of plasma calcium ion concentration.
glands to increase their rate of secretion within
minutes; if the decreased calcium concentration per-
sists, the glands will hypertrophy, sometimes fivefold or
Calcitonin
more. For instance, the parathyroid glands become
greatly enlarged in rickets, in which the level of
Calcitonin, a peptide hormone secreted by the thyroid
calcium is usually depressed only a small amount; also,
gland, tends to decrease plasma calcium concentra-
they become greatly enlarged in pregnancy, even
tion and, in general, has effects opposite to those of
though the decrease in calcium ion concentration in
PTH. However, the quantitative role of calcitonin is
the mother’s extracellular fluid is hardly measurable;
far less than that of PTH in regulating calcium ion
and they are greatly enlarged during lactation because
concentration.
calcium is used for milk formation.
Synthesis and secretion of calcitonin occur in the
Conversely, conditions that increase the calcium ion
parafollicular cells, or C cells, lying in the interstitial
concentration above normal cause decreased activity
fluid between the follicles of the thyroid gland. These
and reduced size of the parathyroid glands. Such con-
cells constitute only about 0.1 per cent of the human
ditions include (1) excess quantities of calcium in the
thyroid gland and are the remnants of the ultimo-
diet, (2) increased vitamin D in the diet, and (3) bone
brachial glands of lower animals such as fish, amphib-
absorption caused by factors other than PTH (for
ians, reptiles, and birds. Calcitonin is a 32-amino acid
example, bone absorption caused by disuse of the
peptide with a molecular weight of about 3400.
bones).
Figure
79-11 shows the approximate relation
Increased Plasma Calcium Concentration Stimulates Calcitonin
between plasma calcium concentration and plasma
Secretion. The primary stimulus for calcitonin secre-
tion is increased plasma calcium ion concentration.
This contrasts with PTH secretion, which is stimulated
by decreased calcium concentration.
In young animals, but much less so in older animals
Parathyroid hormone
and in humans, an increase in plasma calcium concen-
Chronic
Calcitonin
tration of about 10 per cent causes an immediate
Acute
effect
3
twofold or more increase in the rate of secretion of cal-
effect
1000
citonin, which is shown by the blue line in Figure
79-11. This provides a second hormonal feedback
800
mechanism for controlling the plasma calcium ion
2
concentration, but one that is relatively weak and
600
works in a way opposite that of the PTH system.
400
1
Calcitonin Decreases Plasma Calcium Concentration. In
some young animals, calcitonin decreases blood
Normal levels
200
calcium ion concentration rapidly, beginning within
0
0
minutes after injection of the calcitonin, in at least two
0
2
4
6
8
10
12
14
16
ways.
Plasma calcium (mg/dL)
1. The immediate effect is to decrease the absorptive
activities of the osteoclasts and possibly the
osteolytic effect of the osteocytic membrane
Figure 79-11
throughout the bone, thus shifting the balance
in favor of deposition of calcium in the
Approximate effect of plasma calcium concentration on the
exchangeable bone calcium salts. This effect is
plasma concentrations of parathyroid hormone and calcitonin.
especially significant in young animals because of
Note especially that long-term, chronic changes in calcium con-
centration of only a few percentage points can cause as much as
the rapid interchange of absorbed and deposited
100 per cent change in parathyroid hormone concentration.
calcium.
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
989
2. The second and more prolonged effect of
hypocalcemia. However, there is a first line of defense
calcitonin is to decrease the formation of new
to prevent this from occurring even before the
osteoclasts. Also, because osteoclastic resorption
parathyroid and calcitonin hormonal feedback
of bone leads secondarily to osteoblastic activity,
systems have a chance to act.
decreased numbers of osteoclasts are followed
by decreased numbers of osteoblasts. Therefore,
Buffer Function of the Exchangeable Calcium in Bones—the
over a long period, the net result is reduced
First Line of Defense. The exchangeable calcium salts
osteoclastic and osteoblastic activity and,
in the bones, discussed earlier in this chapter, are
consequently, very little prolonged effect on
amorphous calcium phosphate compounds, probably
plasma calcium ion concentration. That is, the
mainly CaHPO4 or some similar compound loosely
effect on plasma calcium is mainly a transient one,
bound in the bone and in reversible equilibrium with
lasting for a few hours to a few days at most.
the calcium and phosphate ions in the extracellular
Calcitonin also has minor effects on calcium han-
fluid.
dling in the kidney tubules and the intestines. Again,
The quantity of these salts that is available for
the effects are opposite those of PTH, but they appear
exchange is about 0.5 to 1 per cent of the total calcium
to be of such little import that they are seldom
salts of the bone, a total of 5 to 10 grams of calcium.
considered.
Because of the ease of deposition of these exchange-
able salts and their ease of resolubility, an increase in
the concentrations of extracellular fluid calcium and
Calcitonin Has a Weak Effect on Plasma Calcium Concentration
phosphate ions above normal causes immediate dep-
in the Adult Human. The reason for the weak effect of
osition of exchangeable salt. Conversely, a decrease in
calcitonin on plasma calcium is twofold. First, any
these concentrations causes immediate absorption of
initial reduction of the calcium ion concentration
exchangeable salt. This reaction is rapid because the
caused by calcitonin leads within hours to a powerful
amorphous bone crystals are extremely small and their
stimulation of PTH secretion, which almost overrides
total surface area exposed to the fluids of the bone is
the calcitonin effect. When the thyroid gland is
perhaps 1 acre or more.
removed and calcitonin is no longer secreted, the long-
Also, about 5 per cent of all the blood flows through
term blood calcium ion concentration is not measura-
the bones each minute—that is, about 1 per cent of all
bly altered, which again demonstrates the overriding
the extracellular fluid each minute. Therefore, about
effect of the PTH system of control.
one half of any excess calcium that appears in the
Second, in the adult, the daily rates of absorption
extracellular fluid is removed by this buffer function
and deposition of calcium are small, and even after the
of the bones in about 70 minutes.
rate of absorption is slowed by calcitonin, this still has
In addition to the buffer function of the bones, the
only a small effect on plasma calcium ion concentra-
mitochondria of many of the tissues of the body, espe-
tion. The effect of calcitonin in children is much
cially of the liver and intestine, contain a reasonable
greater because bone remodeling occurs rapidly in
amount of exchangeable calcium (a total of about 10
children, with absorption and deposition of calcium as
grams in the whole body) that provides an additional
great as 5 grams or more per day—equal to 5 to 10
buffer system for helping to maintain constancy of the
times the total calcium in all the extracellular fluid.
extracellular fluid calcium ion concentration.
Also, in certain bone diseases, such as Paget’s disease,
in which osteoclastic activity is greatly accelerated, cal-
Hormonal Control of Calcium Ion Concentration—the Second
citonin has a much more potent effect of reducing the
Line of Defense. At the same time that the exchange-
calcium absorption.
able calcium mechanism in the bones is “buffering” the
calcium in the extracellular fluid, both the parathyroid
and the calcitonin hormonal systems are beginning to
Summary of Control of
act. Within 3 to 5 minutes after an acute increase in the
Calcium Ion Concentration
calcium ion concentration, the rate of PTH secretion
decreases. As already explained, this sets into play
At times, the amount of calcium absorbed into or lost
multiple mechanisms for reducing the calcium ion con-
from the body fluids is as much as 0.3 gram in 1 hour.
centration back toward normal.
For instance, in cases of diarrhea, several grams of
At the same time that PTH decreases, calcitonin
calcium can be secreted in the intestinal juices, passed
increases. In young animals and possibly in young chil-
into the intestinal tract, and lost into the feces each
dren (but probably to a smaller extent in adults), the
day.
calcitonin causes rapid deposition of calcium in the
Conversely, after ingestion of large quantities of
bones, and perhaps in some cells of other tissues.
calcium, particularly when there is also an excess of
Therefore, in very young animals, excess calcitonin can
vitamin D activity, a person may absorb as much as 0.3
cause a high calcium ion concentration to return to
gram in 1 hour. This figure compares with a total quan-
normal perhaps considerably more rapidly than can
tity of calcium in all the extracellular fluid of about
be achieved by the exchangeable calcium-buffering
1 gram. The addition or subtraction of 0.3 gram to
mechanism alone.
or from such a small amount of calcium in the extra-
In prolonged calcium excess or prolonged calcium
cellular fluid would cause serious hypercalcemia or
deficiency, only the PTH mechanism seems to be really
990
Unit XIV Endocrinology and Reproduction
important in maintaining a normal plasma calcium ion
secretion. The cause of primary hyperparathyroidism
concentration. When a person has a continuing defi-
ordinarily is a tumor of one of the parathyroid glands;
ciency of calcium in the diet, PTH often can stimulate
such tumors occur much more frequently in women
than in men or children, mainly because pregnancy and
enough calcium absorption from the bones to main-
lactation stimulate the parathyroid glands and therefore
tain a normal plasma calcium ion concentration for 1
predispose to the development of such a tumor.
year or more, but eventually, even the bones will run
Hyperparathyroidism causes extreme osteoclastic
out of calcium. Thus, in effect, the bones are a large
activity in the bones. This elevates the calcium ion con-
buffer-reservoir of calcium that can be manipulated
centration in the extracellular fluid while usually
by PTH. Yet, when the bone reservoir either runs out
depressing the concentration of phosphate ions because
of calcium or, oppositely, becomes saturated with
of increased renal excretion of phosphate.
calcium, the long-term control of extracellular calcium
ion concentration resides almost entirely in the roles
Bone Disease in Hyperparathyroidism. Although in mild
of PTH and vitamin D in controlling calcium absorp-
hyperparathyroidism new bone can be deposited
rapidly enough to compensate for the increased osteo-
tion from the gut and calcium excretion in the urine.
clastic reabsorption of bone, in severe hyperparathy-
roidism the osteoclastic absorption soon far outstrips
Pathophysiology of
osteoblastic deposition, and the bone may be eaten
away almost entirely. Indeed, the reason a hyper-
Parathyroid Hormone,
parathyroid person seeks medical attention is often a
Vitamin D, and
broken bone. Radiographs of the bone show extensive
Bone Disease
decalcification and, occasionally, large punched-out
cystic areas of the bone that are filled with osteoclasts
Hypoparathyroidism
in the form of so-called giant cell osteoclast “tumors.”
Multiple fractures of the weakened bones can result
When the parathyroid glands do not secrete sufficient
from only slight trauma, especially where cysts develop.
PTH, the osteocytic reabsorption of exchangeable
The cystic bone disease of hyperparathyroidism is called
calcium decreases and the osteoclasts become almost
osteitis fibrosa cystica.
totally inactive. As a result, calcium reabsorption from
Osteoblastic activity in the bones also increases
the bones is so depressed that the level of calcium in the
greatly in a vain attempt to form enough new bone to
body fluids decreases. Yet, because calcium and phos-
make up for the old bone absorbed by the osteoclastic
phates are not being absorbed from the bone, the bone
activity. When the osteoblasts become active, they
usually remains strong.
secrete large quantities of alkaline phosphatase. There-
When the parathyroid glands are suddenly removed,
fore, one of the important diagnostic findings in hyper-
the calcium level in the blood falls from the normal of
parathyroidism is a high level of plasma alkaline
9.4 mg/dl to 6 to 7 mg/dl within 2 to 3 days, and the
phosphatase.
blood phosphate concentration may double. When this
low calcium level is reached, the usual signs of tetany
Effects of Hypercalcemia in Hyperparathyroidism. Hyper-
develop. Among the muscles of the body especially sen-
parathyroidism can at times cause the plasma calcium
sitive to tetanic spasm are the laryngeal muscles. Spasm
level to rise to 12 to 15 mg/dl and, rarely, even higher.
of these muscles obstructs respiration, which is the usual
The effects of such elevated calcium levels, as detailed
cause of death in tetany unless appropriate treatment is
earlier in the chapter, are depression of the central and
applied.
peripheral nervous systems, muscle weakness, constipa-
tion, abdominal pain, peptic ulcer, lack of appetite, and
Treatment of Hypoparathyroidism with PTH and Vitamin D. PTH
depressed relaxation of the heart during diastole.
is occasionally used for treating hypoparathyroidism.
However, because of the expense of this hormone,
Parathyroid Poisoning and Metastatic Calcification When, on
because its effect lasts for a few hours at most, and
rare occasions, extreme quantities of PTH are secreted,
because the tendency of the body to develop anti-
the level of calcium in the body fluids rises rapidly to
bodies against it makes it progressively less and less
high values. Even the extracellular fluid phosphate con-
effective, hypoparathyroidism is usually not treated
centration often rises markedly instead of falling, as is
with PTH administration.
usually the case, probably because the kidneys cannot
In most patients with hypothyroidism, the administra-
excrete rapidly enough all the phosphate being
tion of extremely large quantities of vitamin D, to as high
absorbed from the bone. Therefore, the calcium and
as 100,000 units per day, along with intake of 1 to 2 grams
phosphate in the body fluids become greatly supersatu-
of calcium, keeps the calcium ion concentration in a
rated, so that calcium phosphate (CaHPO4) crystals
normal range. At times, it might be necessary to admin-
begin to deposit in the alveoli of the lungs, the tubules
ister 1,25-dihydroxycholecalciferol instead of the nonac-
of the kidneys, the thyroid gland, the acid-producing
tivated form of vitamin D because of its much more
area of the stomach mucosa, and the walls of the arter-
potent and much more rapid action. This can also cause
ies throughout the body. This extensive metastatic dep-
unwanted effects, because it is sometimes difficult to
osition of calcium phosphate can develop within a few
prevent overactivity by this activated form of vitamin D.
days.
Ordinarily, the level of calcium in the blood must rise
Primary Hyperparathyroidism
above 17 mg/dl before there is danger of parathyroid
poisoning, but once such elevation develops along with
In primary hyperparathyroidism, an abnormality of the
concurrent elevation of phosphate, death can occur in
parathyroid glands causes inappropriate, excess PTH
only a few days.
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
991
Formation of Kidney Stones in Hyperparathyroidism Most
Rickets Weakens the Bones During prolonged rickets, the
patients with mild hyperparathyroidism show few signs
marked compensatory increase in PTH secretion causes
of bone disease and few general abnormalities as a
extreme osteoclastic absorption of the bone; this in turn
result of elevated calcium, but they do have an extreme
causes the bone to become progressively weaker and
tendency to form kidney stones. The reason is that the
imposes marked physical stress on the bone, resulting in
excess calcium and phosphate absorbed from the intes-
rapid osteoblastic activity as well. The osteoblasts lay
tines or mobilized from the bones in hyperparathy-
down large quantities of osteoid, which does not
roidism must eventually be excreted by the kidneys,
become calcified because of insufficient calcium and
causing a proportionate increase in the concentrations
phosphate ions. Consequently, the newly formed, uncal-
of these substances in the urine. As a result, crystals of
cified, and weak osteoid gradually takes the place of the
calcium phosphate tend to precipitate in the kidney,
older bone that is being reabsorbed.
forming calcium phosphate stones. Also, calcium oxalate
stones develop because even normal levels of oxalate
Tetany in Rickets In the early stages of rickets, tetany
cause calcium precipitation at high calcium levels.
almost never occurs because the parathyroid glands
Because the solubility of most renal stones is slight in
continually stimulate osteoclastic absorption of bone
alkaline media, the tendency for formation of renal
and, therefore, maintain an almost normal level of
calculi is considerably greater in alkaline urine than in
calcium in the extracellular fluid. However, when the
acid urine. For this reason, acidotic diets and acidic
bones finally become exhausted of calcium, the level of
drugs are frequently used for treating renal calculi.
calcium may fall rapidly. As the blood level of calcium
falls below 7 mg/dl, the usual signs of tetany develop,
and the child may die of tetanic respiratory spasm
Secondary Hyperparathyroidism
unless intravenous calcium is administered, which
relieves the tetany immediately.
In secondary hyperparathyroidism, high levels of PTH
occur as a compensation for hypocalcemia rather than
Treatment of Rickets. The treatment of rickets depends on
as a primary abnormality of the parathyroid glands. This
supplying adequate calcium and phosphate in the diet
contrasts with primary hyperparathyroidism, which is
and, equally important, on administering large amounts
associated with hypercalcemia.
of vitamin D. If vitamin D is not administered, little
Secondary hyperparathyroidism can be caused by
calcium and phosphate are absorbed from the gut.
vitamin D deficiency or chronic renal disease in which
the damaged kidneys are unable to produce sufficient
Osteomalacia—“Adult Rickets”. Adults seldom have a
amounts of the active form of vitamin D,
1,25-
serious dietary deficiency of vitamin D or calcium
dihydroxycholecalciferol. As discussed in more detail in
because large quantities of calcium are not needed for
the next section, the vitamin D deficiency leads to osteo-
bone growth as in children. However, serious deficien-
malacia (inadequate mineralization of the bones), and
cies of both vitamin D and calcium occasionally
high levels of PTH cause absorption of the bones.
occur as a result of steatorrhea (failure to absorb fat)
because vitamin D is fat-soluble and calcium tends to
form insoluble soaps with fat; consequently, in steator-
Rickets—Vitamin D Deficiency
rhea, both vitamin D and calcium tend to pass into the
feces. Under these conditions, an adult occasionally has
Rickets occurs mainly in children. It results from
such poor calcium and phosphate absorption that adult
calcium or phosphate deficiency in the extracellular
rickets can occur, although this almost never proceeds
fluid, usually caused by lack of vitamin D. If the child is
to the stage of tetany but often is a cause of severe bone
adequately exposed to sunlight, the 7-dehydrocholes-
disability.
terol in the skin becomes activated by the ultraviolet
rays and forms vitamin D3, which prevents rickets by
Osteomalacia and Rickets Caused by Renal Disease. “Renal
promoting calcium and phosphate absorption from the
rickets” is a type of osteomalacia that results from
intestines, as discussed earlier in the chapter.
prolonged kidney damage. The cause of this
Children who remain indoors through the winter in
condition is mainly failure of the damaged kidneys to
general do not receive adequate quantities of vitamin D
form 1,25-dihydroxycholecalciferol, the active form of
without some supplementation in the diet. Rickets tends
vitamin D. In patients whose kidneys have been
to occur especially in the spring months because vitamin
removed or destroyed and who are being treated by
D formed during the preceding summer is stored in the
hemodialysis, the problem of renal rickets is often a
liver and available for use during the early winter
severe one.
months. Also, calcium and phosphate absorption from
Another type of renal disease that leads to rickets
the bones can prevent clinical signs of rickets for the
and osteomalacia is congenital hypophosphatemia,
first few months of vitamin D deficiency.
resulting from congenitally reduced reabsorption of
phosphates by the renal tubules. This type of rickets
Plasma Concentrations of Calcium and Phosphate Decrease
must be treated with phosphate compounds instead
in Rickets The plasma calcium concentration in rickets
of calcium and vitamin D, and it is called vitamin
is only slightly depressed, but the level of phosphate is
D-resistant rickets.
greatly depressed. This is because the parathyroid
glands prevent the calcium level from falling by pro-
moting bone absorption every time the calcium level
Osteoporosis—Decreased
begins to fall. However, there is no good regulatory
Bone Matrix
system for preventing a falling level of phosphate, and
the increased parathyroid activity actually increases the
Osteoporosis is the most common of all bone diseases
excretion of phosphates in the urine.
in adults, especially in old age. It is different from osteo-
992
Unit XIV Endocrinology and Reproduction
malacia and rickets because it results from diminished
organic bone matrix rather than from poor bone calci-
fication. In osteoporosis the osteoblastic activity in the
Enamel
bone usually is less than normal, and consequently the
Crown
rate of bone osteoid deposition is depressed. But occa-
sionally, as in hyperparathyroidism, the cause of the
diminished bone is excess osteoclastic activity.
Neck
The many common causes of osteoporosis are (1) lack
of physical stress on the bones because of inactivity; (2)
Pulp chamber
malnutrition to the extent that sufficient protein matrix
cannot be formed; (3) lack of vitamin C, which is nec-
essary for the secretion of intercellular substances by all
cells, including formation of osteoid by the osteoblasts;
(4) postmenopausal lack of estrogen secretion because
Dentin
estrogens decrease the number and activity of osteo-
Root
clasts; (5) old age, in which growth hormone and other
growth factors diminish greatly, plus the fact that many
of the protein anabolic functions also deteriorate with
Cementum
age, so that bone matrix cannot be deposited satisfac-
torily; and
(6) Cushing’s syndrome, because massive
quantities of glucocorticoids secreted in this disease
cause decreased deposition of protein throughout the
body and increased catabolism of protein and have the
specific effect of depressing osteoblastic activity. Thus,
many diseases of deficiency of protein metabolism can
Figure 79-12
cause osteoporosis.
Functional parts of a tooth.
Physiology of the Teeth
The crystalline structure of the salts makes the
The teeth cut, grind, and mix the food eaten. To
enamel extremely hard-much harder than the dentin.
perform these functions, the jaws have powerful
Also, the special protein fiber meshwork, although
muscles capable of providing an occlusive force
constituting only about 1 per cent of the enamel mass,
between the front teeth of 50 to 100 pounds and for
makes enamel resistant to acids, enzymes, and other
the jaw teeth, 150 to 200 pounds. Also, the upper and
corrosive agents because this protein is one of the
lower teeth are provided with projections and facets
most insoluble and resistant proteins known.
that interdigitate, so that the upper set of teeth fits with
the lower. This fitting is called occlusion, and it allows
Dentin. The main body of the tooth is composed of
even small particles of food to be caught and ground
dentin, which has a strong, bony structure. Dentin is
between the tooth surfaces.
made up principally of hydroxyapatite crystals similar
to those in bone but much more dense. These crystals
Function of the Different Parts
are imbedded in a strong meshwork of collagen fibers.
of the Teeth
In other words, the principal constituents of dentin are
very much the same as those of bone. The major dif-
Figure
79-12 shows a sagittal section of a tooth,
ference is its histological organization, because dentin
demonstrating its major functional parts: the enamel,
does not contain any osteoblasts, osteocytes, osteo-
dentin, cementum, and pulp. The tooth can also be
clasts, or spaces for blood vessels or nerves. Instead, it
divided into the crown, which is the portion that pro-
is deposited and nourished by a layer of cells called
trudes out from the gum into the mouth, and the root,
odontoblasts, which line its inner surface along the wall
which is the portion within the bony socket of the jaw.
of the pulp cavity.
The collar between the crown and the root where the
The calcium salts in dentin make it extremely resist-
tooth is surrounded by the gum is called the neck.
ant to compressional forces, and the collagen fibers
make it tough and resistant to tensional forces that
Enamel. The outer surface of the tooth is covered by a
might result when the teeth are struck by solid objects.
layer of enamel that is formed before eruption of the
tooth by special epithelial cells called ameloblasts.
Cementum. Cementum is a bony substance secreted
Once the tooth has erupted, no more enamel is
by cells of the periodontal membrane, which lines
formed. Enamel is composed of very large and very
the tooth socket. Many collagen fibers pass directly
dense crystals of hydroxyapatite with adsorbed
from the bone of the jaw, through the periodontal
carbonate, magnesium, sodium, potassium, and other
membrane, and then into the cementum. These colla-
ions imbedded in a fine meshwork of strong and
gen fibers and the cementum hold the tooth in place.
almost insoluble protein fibers that are similar in phys-
When the teeth are exposed to excessive strain, the
ical characteristics (but not chemically identical) to the
layer of cementum becomes thicker and stronger.
keratin of hair.
Also, it increases in thickness and strength with age,
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
993
causing the teeth to become more firmly seated in the
jaws by adulthood and later.
Enamel organ
Pulp. The pulp cavity of each tooth is filled with pulp,
Primordium of
of milk tooth
enamel organ of
which is composed of connective tissue with an abun-
permanent tooth
dant supply of nerve fibers, blood vessels, and lym-
phatics. The cells lining the surface of the pulp cavity
are the odontoblasts, which, during the formative years
of the tooth, lay down the dentin but at the same time
encroach more and more on the pulp cavity, making
it smaller. In later life, the dentin stops growing and
Mesenchymal primordium of pulp
the pulp cavity remains essentially constant in size.
A
However, the odontoblasts are still viable and send
projections into small dentinal tubules that penetrate
Enamel
all the way through the dentin; they are of importance
Dentin
for exchange of calcium, phosphate, and other miner-
Oral epithelium
als with the dentin.
Dentition
Humans and most other mammals develop two sets of
teeth during a lifetime. The first teeth are called the
deciduous teeth, or milk teeth, and they number 20 in
humans. They erupt between the 7th month and the
2nd year of life, and they last until the 6th to the 13th
year. After each deciduous tooth is lost, a permanent
tooth replaces it, and an additional 8 to 12 molars
Alveolar bone
B
C
appear posteriorly in the jaws, making the total
number of permanent teeth 28 to 32, depending on
Figure 79-13
whether the four wisdom teeth finally appear, which
A, Primordial tooth organ. B, Developing tooth. C, Erupting tooth.
does not occur in everyone.
Formation of the Teeth. Figure 79-13 shows the forma-
like the deciduous tooth, pushes outward through the
tion and eruption of teeth. Figure
79-13A shows
bone. In so doing, it erodes the root of the deciduous
invagination of the oral epithelium into the dental
tooth and eventually causes it to loosen and fall out.
lamina; this is followed by the development of a tooth-
Soon thereafter, the permanent tooth erupts to take
producing organ. The epithelial cells above form
the place of the original one.
ameloblasts, which form the enamel on the outside of
the tooth. The epithelial cells below invaginate upward
Metabolic Factors Influence Development of the Teeth.
into the middle of the tooth to form the pulp cavity
The rate of development and the speed of eruption of
and the odontoblasts that secrete dentin. Thus, enamel
teeth can be accelerated by both thyroid and growth
is formed on the outside of the tooth, and dentin is
hormones. Also, the deposition of salts in the early-
formed on the inside, giving rise to an early tooth, as
forming teeth is affected considerably by various
shown in Figure 79-13B.
factors of metabolism, such as the availability of
calcium and phosphate in the diet, the amount of
Eruption of Teeth. During early childhood, the teeth
vitamin D present, and the rate of PTH secretion.When
begin to protrude outward from the bone through the
all these factors are normal, the dentin and enamel will
oral epithelium into the mouth. The cause of “erup-
be correspondingly healthy, but when they are defi-
tion” is unknown, although several theories have been
cient, the calcification of the teeth also may be defec-
offered in an attempt to explain this phenomenon. The
tive, so that the teeth will be abnormal throughout life.
most likely theory is that growth of the tooth root as
well as of the bone underneath the tooth progressively
shoves the tooth forward.
Mineral Exchange in Teeth
Development of the Permanent Teeth. During embry-
The salts of teeth, like those of bone, are composed of
onic life, a tooth-forming organ also develops in the
hydroxyapatite with adsorbed carbonates and various
deeper dental lamina for each permanent tooth that
cations bound together in a hard crystalline substance.
will be needed after the deciduous teeth are gone.
Also, new salts are constantly being deposited while
These tooth-producing organs slowly form the perma-
old salts are being reabsorbed from the teeth, as occurs
nent teeth throughout the first 6 to 20 years of life.
in bone. Deposition and reabsorption occur mainly in
When each permanent tooth becomes fully formed, it,
the dentin and cementum and to a very limited extent
994
Unit XIV Endocrinology and Reproduction
in the enamel. In the enamel, these processes occur
parcels throughout the day, such as in the form of
mostly by diffusional exchange of minerals with the
candy, the bacteria are supplied with their preferential
saliva instead of with the fluids of the pulp cavity.
metabolic substrate for many hours of the day, and the
The rate of absorption and deposition of minerals in
development of caries is greatly increased.
the cementum is about equal to that in the surround-
ing bone of the jaw, whereas the rate of deposition and
Role of Fluorine in Preventing Caries. Teeth formed in chil-
absorption of minerals in the dentin is only one third
dren who drink water that contains small amounts of
that of bone. The cementum has characteristics almost
fluorine develop enamel that is more resistant to caries
identical to those of usual bone, including the presence
than the enamel in children who drink water that does
of osteoblasts and osteoclasts, whereas dentin does not
not contain fluorine. Fluorine does not make the
have these characteristics, as explained earlier. This
enamel harder than usual, but fluorine ions replace
difference undoubtedly explains the different rates of
many of the hydroxyl ions in the hydroxyapatite crys-
mineral exchange.
tals, which in turn makes the enamel several times less
In summary, continual mineral exchange occurs in
soluble. Fluorine may also be toxic to the bacteria.
the dentin and cementum of teeth, although the mech-
Finally, when small pits do develop in the enamel, flu-
anism of this exchange in dentin is unclear. However,
orine is believed to promote deposition of calcium
enamel exhibits extremely slow mineral exchange, so
phosphate to “heal” the enamel surface. Regardless of
that it maintains most of its original mineral comple-
the precise means by which fluorine protects the teeth,
ment throughout life.
it is known that small amounts of fluorine deposited in
enamel make teeth about three times as resistant to
caries as teeth without fluorine.
Dental Abnormalities
Malocclusion. Malocclusion is usually caused by a
The two most common dental abnormalities are caries
hereditary abnormality that causes the teeth of one
and malocclusion. Caries refers to erosion of the teeth,
jaw to grow to abnormal positions. In malocclusion,
whereas malocclusion is failure of the projections of
the teeth do not interdigitate properly and therefore
the upper and lower teeth to interdigitate properly.
cannot perform their normal grinding or cutting action
adequately. Malocclusion occasionally also results in
Caries and the Role of Bacteria and Ingested Carbohydrates.
abnormal displacement of the lower jaw in relation to
It is generally agreed that caries result from the action
the upper jaw, causing such undesirable effects as pain
of bacteria on the teeth, the most common of which is
in the mandibular joint and deterioration of the teeth.
Streptococcus mutans. The first event in the develop-
The orthodontist can usually correct malocclusion
ment of caries is the deposit of plaque, a film of pre-
by applying prolonged gentle pressure against the
cipitated products of saliva and food, on the teeth.
teeth with appropriate braces. The gentle pressure
Large numbers of bacteria inhabit this plaque and
causes absorption of alveolar jaw bone on the com-
are readily available to cause caries. These bacteria
pressed side of the tooth and deposition of new bone
depend to a great extent on carbohydrates for their
on the tensional side of the tooth. In this way, the tooth
food. When carbohydrates are available, their meta-
gradually moves to a new position as directed by the
bolic systems are strongly activated and they multiply.
applied pressure.
In addition, they form acids (particularly lactic acid)
and proteolytic enzymes. The acids are the major
culprit in causing caries because the calcium salts of
References
teeth are slowly dissolved in a highly acidic medium.
And once the salts have become absorbed, the remain-
Altkorn D, Vokes T: Treatment of postmenopausal osteo-
ing organic matrix is rapidly digested by the prote-
porosis. JAMA 285:1415, 2001.
olytic enzymes.
Bilezikian JP, Silverberg SJ: Clinical practice. Asymptomatic
The enamel of the tooth is the primary barrier to the
primary hyperparathyroidism. N Engl J Med 350:1746,
development of caries. Enamel is far more resistant to
2004.
demineralization by acids than is dentin, primarily
Chen RA, Goodman WG: Role of the calcium-sensing
because the crystals of enamel are dense, but also
receptor in parathyroid gland physiology. Am J Physiol
because each enamel crystal is about 200 times as large
Renal Physiol 286:F1005, 2004.
in volume as each dentin crystal. Once the carious
Compston JE: Sex steroids and bone. Physiol Rev 81:419,
process has penetrated through the enamel to the
2001.
dentin, it proceeds many times as rapidly because of
Delmas PD: Treatment of postmenopausal osteoporosis.
Lancet 359:2018, 2002.
the high degree of solubility of the dentin salts.
Goodman WG, Juppner H, Salusky IB, Sherrard DJ:
Because of the dependence of the caries bacteria on
Parathyroid hormone (PTH), PTH-derived peptides, and
carbohydrates for their nutrition, it has frequently
new PTH assays in renal osteodystrophy. Kidney Int 63:1,
been taught that eating a diet high in carbohydrate
2003.
content will lead to excessive development of caries.
Gurlek A, Pittelkow MR, Kumar R: Modulation of growth
However, it is not the quantity of carbohydrate
factor/cytokine synthesis and signaling by
1alpha,25-
ingested but the frequency with which it is eaten that
dihydroxyvitamin D(3): implications in cell growth and
is important. If carbohydrates are eaten in many small
differentiation. Endocr Rev 23:763, 2002.
Chapter 79
Parathyroid Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth
995
Hofer AM, Brown EM: Extracellular calcium sensing and
NIH Consensus Development Panel on Osteoporosis Pre-
signalling. Nat Rev Mol Cell Biol 4:530, 2003.
vention, Diagnosis, and Therapy. Osteoporosis prevention,
Jones G, Strugnell SA, DeLuca HF: Current understanding
diagnosis, and therapy. JAMA 285:785, 2001.
of the molecular actions of vitamin D. Physiol Rev
Peng JB, Brown EM, Hediger MA: Apical entry channels in
78:1193, 1998.
calcium-transporting epithelia, News Physiol Sci 18:158,
Larsen PR, Kronenberg HM, Melmed S, Polonsky KS:
2003.
Williams Textbook of Endocrinology, 10th ed. Philadel-
Silver J, Kilav R, Naveh-Many T: Mechanisms of secondary
phia: WB Saunders Co, 2003.
hyperparathyroidism. Am J Physiol Renal Physiol
Lips P: Vitamin D deficiency and secondary hyperparathy-
283:F367, 2002.
roidism in the elderly: consequences for bone loss and
Tordoff MG: Calcium: taste, intake, and appetite. Physiol
fractures and therapeutic implications. Endocr Rev
Rev 81:1567, 2001.
22:477, 2001.
Wharton B, Bishop N: Rickets. Lancet 362:1389, 2003.
Marx SJ: Hyperparathyroid and hypoparathyroid disorders.
Woolf AD, Akesson K: Preventing fractures in elderly
N Engl J Med 343:1863, 2000.
people. BMJ 327:89, 2003.
C H A P T E R
8
0
Reproductive and Hormonal
Functions of the Male (and
Function of the Pineal Gland)
The reproductive functions of the male can be
divided into three major subdivisions: (1) sper-
matogenesis, which means simply the formation of
sperm; (2) performance of the male sexual act; and
(3) regulation of male reproductive functions
by the various hormones. Associated with these
reproductive functions are the effects of the male
sex hormones on the accessory sexual organs, cellu-
lar metabolism, growth, and other functions of the body.
Physiologic Anatomy of the
Male Sexual Organs
Figure 80-1A shows the various portions of the male reproductive system, and
Figure 80-1B gives a more detailed structure of the testis and epididymis. The testis
is composed of up to 900 coiled seminiferous tubules, each averaging more than one
half meter long, in which the sperm are formed. The sperm then empty into the
epididymis, another coiled tube about 6 meters long. The epididymis leads into the
vas deferens, which enlarges into the ampulla of the vas deferens immediately before
the vas enters the body of the prostate gland.
Two seminal vesicles, one located on each side of the prostate, empty into the pro-
static end of the ampulla, and the contents from both the ampulla and the seminal
vesicles pass into an ejaculatory duct leading through the body of the prostate gland
and then emptying into the internal urethra. Prostatic ducts, too, empty from the
prostate gland into the ejaculatory duct and from there into the prostatic urethra.
Finally, the urethra is the last connecting link from the testis to the exterior. The
urethra is supplied with mucus derived from a large number of minute urethral
glands located along its entire extent and even more so from bilateral bulbourethral
glands (Cowper’s glands) located near the origin of the urethra.
Spermatogenesis
During formation of the embryo, the primordial germ cells migrate into the
testes and become immature germ cells called spermatogonia which lie in two
or three layers of the inner surfaces of the seminiferous tubules (a cross section
of one is shown in Figure 80-2A). The spermatogonia begin to undergo mitotic
division, beginning at puberty, and continually proliferate and differentiate
through definite stages of development to form sperm, as shown in Figure
80-2B.
Steps of Spermatogenesis
Spermatogenesis occurs in the seminiferous tubules during active sexual life as
the result of stimulation by anterior pituitary gonadotropic hormones, begin-
ning at an average age of 13 years and continuing throughout most of the
remainder of life but decreasing markedly in old age.
In the first stage of spermatogenesis, the spermatogonia migrate among
Sertoli cells toward the central lumen of the seminiferous tubule. The Sertoli
996
Chapter 80
Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
997
Urinary
bladder
Ampulla
Seminal
Interstitial cells
vesicle
Prostate
Ejaculatory
Seminiferous
gland
duct
tubules
Bulbourethral
Erectile
gland
tissue
Vas deferens
Epididymis
A
Prepuce
Seminiferous
Glans
tubules
penis
Spermatids
Testis
Scrotum
A
Spermatozoa
Head of
epididymis
Secondary
Testicular artery
spermatocyte
Vas deferens
Primary
spermatocyte
Efferent
ductules
Sertoli cell
Body of
epididymis
Seminiferous
tubules
Rete testis
Spermatogonium
B
Tail of epididymis
Figure 80-2
B
A, Cross section of a seminiferous tubule. B, Stages in the devel-
Figure 80-1
opment of sperm from spermatogonia.
A, Male reproduction system. (Modified from Bloom V, Fawcett
DW: Textbook of Histology, 10th ed. Philadelphia: WB Saunders
Co, 1975.) B, Internal structure of the testis and relation of the
the father, while the other half are derived from the
testis to the epididymis. (Redrawn from Guyton AC: Anatomy and
oocyte provided by the mother.
Physiology. Philadelphia: Saunders College Publishing, 1985.)
The entire period of spermatogenesis, from sper-
matogonia to spermatozoa, takes about 74 days.
cells are very large, with overflowing cytoplasmic
Sex Chromosomes. In each spermatogonium, one of the
envelopes that surround the developing spermatogo-
23 pairs of chromosomes carries the genetic informa-
nia all the way to the central lumen of the tubule.
tion that determines the sex of each eventual offspring.
This pair is composed of one X chromosome, which is
Meiosis. Spermatogonia that cross the barrier into the
called the female chromosome, and one Y chromo-
Sertoli cell layer become progressively modified and
some, the male chromosome. During meiotic division,
enlarged to form large primary spermatocytes (Figure
the male Y chromosome goes to one spermatid that
80-3). Each of these, in turn, undergoes meiotic
then becomes a male sperm, and the female X chro-
division to form two secondary spermatocytes. After
mosome goes to another spermatid that becomes a
another few days, these too divide to form spermatids
female sperm. The sex of the eventual offspring is
that are eventually modified to become spermatozoa
determined by which of these two types of sperm
(sperm).
fertilizes the ovum. This is discussed further in
During the change from the spermatocyte stage to
Chapter 82.
the spermatid stage, the 46 chromosomes (23 pairs of
chromosomes) of the spermatocyte are divided, so that
Formation of Sperm. When the spermatids are first
23 chromosomes go to one spermatid and the other 23
formed, they still have the usual characteristics of
to the second spermatid. This also divides the chro-
epithelioid cells, but soon they begin to differentiate
mosomal genes so that only one half of the genetic
and elongate into spermatozoa. As shown in Figure
characteristics of the eventual fetus are provided by
80-4, each spermatozoon is composed of a head and a
998
Unit XIV Endocrinology and Reproduction
Acrosome
Birth
Primordial germ cell
Surface membrane
Vacuole
Enters
Anterior head cap
testis
12-14
Posterior head cap
years
Neck
Spermatogonia
Body
Puberty
Mitochondria
Spermatogonia
proliferate by mitotic
cell division inside
Chief piece of tail
testis
25 days
End piece of tail
Primary
spermatocyte
Figure 80-4
9 days
Meiotic division I
Structure of the human spermatozoon.
Secondary
spermatocytes
hyaluronidase
(which can digest proteoglycan
fila-
Meiotic division II
ments of tissues) and powerful proteolytic enzymes
19 days
(which can digest proteins). These enzymes play
important roles in allowing the sperm to enter the
ovum and fertilize it.
Spermatids
The tail of the sperm, called the flagellum, has three
major components: (1) a central skeleton constructed
of 11 microtubules, collectively called the axoneme—
21 days
Differentiation
the structure of this is similar to that of cilia found
Mature
on the surfaces of other types of cells described in
sperm
Chapter 2; (2) a thin cell membrane covering the
axoneme; and (3) a collection of mitochondria sur-
rounding the axoneme in the proximal portion of the
tail (called the body of the tail).
Back-and-forth movement of the tail
(flagellar
movement) provides motility for the sperm. This
Figure 80-3
movement results from a rhythmical longitudinal
sliding motion between the anterior and posterior
Cell divisions
during
spermatogenesis. During embryonic
development the primordial germ cells migrate to the testis where
tubules that make up the axoneme. The energy for this
they become spermatogonia. At puberty (usually 12 to14 years
process is supplied in the form of adenosine triphos-
after birth), the spermatogonia proliferate rapidly by mitosis. Some
phate that is synthesized by the mitochondria in the
begin meiosis to become primary spermatocytes and continue
body of the tail.
through meiotic division I to become secondary spermato-
cytes. After completion of meiotic division II, the secondary
Normal sperm move in a fluid medium at a velocity
spermatocytes produce spermatids, which differentiate to form
of 1 to 4 mm/min. This allows them to move through
spermatozoa.
the female genital tract in quest of the ovum.
Hormonal Factors That Stimulate
tail. The head comprises the condensed nucleus of the
Spermatogenesis
cell with only a thin cytoplasmic and cell membrane
We shall discuss the role of hormones in reproduction
layer around its surface. On the outside of the anterior
later, but at this point, let us note that several hor-
two thirds of the head is a thick cap called the acro-
mones play essential roles in spermatogenesis. Some
some that is formed mainly from the Golgi apparatus.
of these are as follows:
This contains a number of enzymes similar to those
1. Testosterone, secreted by the Leydig cells located
found in lysosomes of the typical cell, including
in the interstitium of the testis, is essential for
Chapter 80
Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
999
growth and division of the testicular germinal
The activity of sperm increases markedly with in-
cells, which is the first stage in forming sperm.
creasing temperature, but so does the rate of metabo-
2. Luteinizing hormone, secreted by the anterior
lism, causing the life of the sperm to be considerably
pituitary gland, stimulates the Leydig cells to
shortened. Although sperm can live for many weeks in
secrete testosterone.
the suppressed state in the genital ducts of the testes,
3. Follicle-stimulating hormone, also secreted by the
life expectancy of ejaculated sperm in the female
anterior pituitary gland, stimulates the Sertoli
genital tract is only 1 to 2 days.
cells; without this stimulation, the conversion
of the spermatids to sperm (the process of
spermiogenesis) will not occur.
Function of the Seminal Vesicles
4. Estrogens, formed from testosterone by the
Sertoli cells when they are stimulated by follicle-
Each seminal vesicle is a tortuous, loculated tube lined
stimulating hormone, are probably also essential
with a secretory epithelium that secretes a mucoid
for spermiogenesis.
material containing an abundance of fructose,
5. Growth hormone (as well as most of the other
citric acid, and other nutrient substances, as well
body hormones) is necessary for controlling
as large quantities of prostaglandins and fibrinogen.
background metabolic functions of the testes.
During the process of emission and ejaculation, each
Growth hormone specifically promotes early
seminal vesicle empties its contents into the ejacula-
division of the spermatogonia themselves; in its
tory duct shortly after the vas deferens empties the
absence, as in pituitary dwarfs, spermatogenesis
sperm. This adds greatly to the bulk of the ejaculated
is severely deficient or absent, thus causing
semen, and the fructose and other substances in the
infertility.
seminal fluid are of considerable nutrient value for
the ejaculated sperm until one of the sperm fertilizes
Maturation of Sperm in the Epididymis
the ovum.
After formation in the seminiferous tubules, the sperm
Prostaglandins are believed to aid fertilization in
require several days to pass through the 6-meter-long
two ways: (1) by reacting with the female cervical
tubule of the epididymis. Sperm removed from the
mucus to make it more receptive to sperm movement
seminiferous tubules and from the early portions of
and (2) by possibly causing backward, reverse peri-
the epididymis are nonmotile, and they cannot fertil-
staltic contractions in the uterus and fallopian tubes to
ize an ovum. However, after the sperm have been
move the ejaculated sperm toward the ovaries (a few
in the epididymis for some 18 to 24 hours, they deve-
sperm reach the upper ends of the fallopian tubes
lop the capability of motility, even though several
within 5 minutes).
inhibitory proteins in the epididymal fluid still prevent
final motility until after ejaculation.
Function of the Prostate Gland
Storage of Sperm. The two testes of the human adult
form up to 120 million sperm each day. A small quan-
The prostate gland secretes a thin, milky fluid that con-
tity of these can be stored in the epididymis, but most
tains calcium, citrate ion, phosphate ion, a clotting
are stored in the vas deferens. They can remain stored,
enzyme, and a profibrinolysin. During emission, the
maintaining their fertility, for at least a month. During
capsule of the prostate gland contracts simultaneously
this time, they are kept in a deeply suppressed inactive
with the contractions of the vas deferens so that the
state by multiple inhibitory substances in the secre-
thin, milky fluid of the prostate gland adds further to
tions of the ducts. Conversely, with a high level of
the bulk of the semen. A slightly alkaline characteris-
sexual activity and ejaculations, storage may be no
tic of the prostatic fluid may be quite important for
longer than a few days.
successful fertilization of the ovum, because the fluid
After ejaculation, the sperm become motile, and
of the vas deferens is relatively acidic owing to the
they also become capable of fertilizing the ovum, a
presence of citric acid and metabolic end products of
process called maturation. The Sertoli cells and the
the sperm and, consequently, helps to inhibit sperm
epithelium of the epididymis secrete a special nutrient
fertility. Also, the vaginal secretions of the female are
fluid that is ejaculated along with the sperm. This fluid
acidic (pH of 3.5 to 4.0). Sperm do not become opti-
contains hormones (including both testosterone and
mally motile until the pH of the surrounding fluids
estrogens), enzymes, and special nutrients that are
rises to about 6.0 to 6.5. Consequently, it is probable
essential for sperm maturation.
that the slightly alkaline prostatic fluid helps to neu-
tralize the acidity of the other seminal fluids during
Physiology of the Mature Sperm. The normal motile, fertile
ejaculation, and thus enhances the motility and fertil-
sperm are capable of flagellated movement though the
ity of the sperm.
fluid medium at velocities of
1 to 4 mm/min. The
activity of sperm is greatly enhanced in a neutral
and slightly alkaline medium, as exists in the ejacu-
Semen
lated semen, but it is greatly depressed in a mildly
acidic medium. A strong acidic medium can cause
Semen, which is ejaculated during the male sexual
rapid death of sperm.
act, is composed of the fluid and sperm from the vas
1000
Unit XIV Endocrinology and Reproduction
deferens (about 10 per cent of the total), fluid from the
the membrane at the head of the sperm (the
seminal vesicles (almost 60 per cent), fluid from the
acrosome) becomes much weaker.
prostate gland (about 30 per cent), and small amounts
3. The membrane of the sperm also becomes much
from the mucous glands, especially the bulbourethral
more permeable to calcium ions, so that calcium
glands. Thus, the bulk of the semen is seminal vesicle
now enters the sperm in abundance and changes
fluid, which is the last to be ejaculated and serves
the activity of the flagellum, giving it a powerful
to wash the sperm through the ejaculatory duct and
whiplash motion in contrast to its previously weak
urethra.
undulating motion. In addition, the calcium ions
The average pH of the combined semen is about 7.5,
cause changes in the cellular membrane that
the alkaline prostatic fluid having more than neutral-
covers the leading edge of the acrosome, making
ized the mild acidity of the other portions of the
it possible for the acrosome to release its enzymes
semen. The prostatic fluid gives the semen a milky
rapidly and easily as the sperm penetrates the
appearance, and fluid from the seminal vesicles and
granulosa cell mass surrounding the ovum, and
mucous glands gives the semen a mucoid consistency.
even more so as it attempts to penetrate the zona
Also, a clotting enzyme from the prostatic fluid causes
pellucida of the ovum itself.
the fibrinogen of the seminal vesicle fluid to form a
Thus, multiple changes occur during the process of
weak fibrin coagulum that holds the semen in the
capacitation. Without these, the sperm cannot make its
deeper regions of the vagina where the uterine cervix
way to the interior of the ovum to cause fertilization.
lies. The coagulum then dissolves during the next 15 to
30 minutes because of lysis by fibrinolysin formed
Acrosome Enzymes, the “Acrosome Reaction,”
from the prostatic profibrinolysin. In the early minutes
and Penetration of the Ovum
after ejaculation, the sperm remain relatively immo-
Stored in the acrosome of the sperm are large quanti-
bile, possibly because of the viscosity of the coagulum.
ties of hyaluronidase and proteolytic enzymes.
As the coagulum dissolves, the sperm simultaneously
Hyaluronidase depolymerizes the hyaluronic acid
become highly motile.
polymers in the intercellular cement that hold the
Although sperm can live for many weeks in the male
ovarian granulosa cells together. The proteolytic
genital ducts, once they are ejaculated in the semen,
enzymes digest proteins in the structural elements of
their maximal life span is only 24 to 48 hours at
tissue cells that still adhere to the ovum.
body temperature. At lowered temperatures, however,
When the ovum is expelled from the ovarian fol-
semen can be stored for several weeks, and when
licle into the fallopian tube, it still carries with it mul-
frozen at temperatures below -100°C, sperm have
tiple layers of granulosa cells. Before a sperm can
been preserved for years.
fertilize the ovum, it must dissolute these granulosa
cell layers, and then it must penetrate though the thick
“Capacitation” of the Spermatozoa—Making It
covering of the ovum itself, the zona pellucida. To
Possible for Them to Penetrate the Ovum
achieve this, the stored enzymes in the acrosome begin
Although spermatozoa are said to be “mature” when
to be released. It is believed that the hyaluronidase
they leave the epididymis, their activity is held in check
among these enzymes is especially important in open-
by multiple inhibitory factors secreted by the genital
ing pathways between the granulosa cells so that the
duct epithelia. Therefore, when they are first expelled
sperm can reach the ovum.
in the semen, they are unable to perform their duties
When the sperm reaches the zona pellucida of the
in fertilizing the ovum. However, on coming in contact
ovum, the anterior membrane of the sperm itself binds
with the fluids of the female genital tract, multiple
specifically with receptor proteins in the zona pellu-
changes occur that activate the sperm for the final
cida. Then, rapidly, the entire acrosome dissolves,
processes of fertilization. These collective changes are
and all the acrosomal enzymes are released. Within
called capacitation of the spermatozoa. This normally
minutes, these enzymes open a penetrating pathway
requires from 1 to 10 hours. Some changes that are
for passage of the sperm head through the zona pel-
believed to occur are the following:
lucida to the inside of the ovum. Within another 30
1. The uterine and fallopian tube fluids wash away
minutes, the cell membranes of the sperm head and of
the various inhibitory factors that suppress sperm
the oocyte fuse with each other to form a single cell.
activity in the male genital ducts.
At the same time, the genetic material of the sperm
2. While the spermatozoa remain in the fluid of the
and the oocyte combine to form a completely new cell
male genital ducts, they are continually exposed
genome, containing equal numbers of chromosomes
to many floating vesicles from the seminiferous
and genes from mother and father. This is the process
tubules containing large amounts of cholesterol.
of fertilization; then the embryo begins to develop, as
This cholesterol is continually added to the
discussed in Chapter 82.
cellular membrane covering the sperm acrosome,
toughening this membrane and preventing release
Why Does Only One Sperm Enter the Oocyte? With as many
of its enzymes. After ejaculation, the sperm
sperm as there are, why does only one enter the
deposited in the vagina swim away from the
oocyte? The reason is not entirely known, but within
cholesterol vesicles upward into the uterine cavity,
a few minutes after the first sperm penetrates the zona
and they gradually lose much of their other excess
pellucida of the ovum, calcium ions diffuse inward
cholesterol over the next few hours. In so doing,
through the oocyte membrane and cause multiple
Chapter 80
Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
1001
cortical granules to be released by exocytosis from
the oocyte into the perivitelline space. These granules
contain substances that permeate all portions of the
zona pellucida and prevent binding of additional
sperm, and they even cause any sperm that have
already begun to bind to fall off. Thus, almost never
does more than one sperm enter the oocyte during
fertilization.
Abnormal Spermatogenesis and
Male Fertility
The seminiferous tubular epithelium can be destroyed
by a number of diseases. For instance, bilateral orchitis
of the testes resulting from mumps causes sterility in
some affected males. Also, some male infants are born
Figure 80-5
with degenerate tubular epithelia as a result of strictures
in the genital ducts or other abnormalities. Finally,
Abnormal infertile sperm, compared with a normal sperm on the
another cause of sterility, usually temporary, is excessive
right.
temperature of the testes.
Effect of Temperature on Spermatogenesis. Increasing the
enough testosterone. The surgical operation for cryp-
temperature of the testes can prevent spermatogenesis
torchidism in these patients is unlikely to be successful.
by causing degeneration of most cells of the semini-
ferous tubules besides the spermatogonia. It has often
Effect of Sperm Count on Fertility. The usual quantity of
been stated that the reason the testes are located in
semen ejaculated during each coitus averages about 3.5
the dangling scrotum is to maintain the temperature
milliliters, and in each milliliter of semen is an average
of these glands below the internal temperature of the
of about 120 million sperm, although even in “normal”
body, although usually only about 2°C below the inter-
males this can vary from 35 million to 200 million. This
nal temperature. On cold days, scrotal reflexes cause
means an average total of 400 million sperm are usually
the musculature of the scrotum to contract, pulling the
present in the several milliliters of each ejaculate. When
testes close to the body to maintain this 2° differential.
the number of sperm in each milliliter falls below about
Thus, the scrotum theoretically acts as a cooling mech-
20 million, the person is likely to be infertile. Thus, even
anism for the testes (but a controlled cooling), without
though only a single sperm is necessary to fertilize the
which spermatogenesis might be deficient during hot
ovum, for reasons not understood, the ejaculate usually
weather.
must contain a tremendous number of sperm for only
one sperm to fertilize the ovum.
Cryptorchidism
Cryptorchidism means failure of a testis to descend
Effect of Sperm Morphology and Motility on Fertility. Occa-
from the abdomen into the scrotum at or near the time
sionally a man has a normal number of sperm but is still
of birth of a fetus. During development of the male
infertile. When this occurs, sometimes as many as one
fetus, the testes are derived from the genital ridges in
half the sperm are found to be abnormal physically,
the abdomen. However, at about 3 weeks to 1 month
having two heads, abnormally shaped heads, or abnor-
before birth of the baby, the testes normally descend
mal tails, as shown in Figure 80-5. At other times, the
through the inguinal canals into the scrotum. Occasion-
sperm appear to be structurally normal, but for reasons
ally this descent does not occur or occurs incompletely,
not understood, they are either entirely nonmotile
so that one or both testes remain in the abdomen, in the
or relatively nonmotile. Whenever the majority of the
inguinal canal, or elsewhere along the route of descent.
sperm are morphologically abnormal or are nonmotile,
A testis that remains throughout life in the abdomi-
the person is likely to be infertile, even though the
nal cavity is incapable of forming sperm. The tubular
remainder of the sperm appear to be normal.
epithelium becomes degenerate, leaving only the
interstitial structures of the testis. It has been claimed
that even the few degrees’ higher temperature in the
Male Sexual Act
abdomen than in the scrotum is sufficient to cause
this degeneration of the tubular epithelium and, conse-
quently, to cause sterility, although this is not certain.
Neuronal Stimulus for Performance
Nevertheless, for this reason, operations to relocate the
of the Male Sexual Act
cryptorchid testes from the abdominal cavity into the
scrotum before the beginning of adult sexual life are
The most important source of sensory nerve signals
frequently performed on boys who have undescended
for initiating the male sexual act is the glans penis.
testes.
The glans contains an especially sensitive sensory end-
Testosterone secretion by the fetal testes themselves
is the normal stimulus that causes the testes to descend
organ system that transmits into the central nervous
into the scrotum from the abdomen. Therefore, many, if
system that special modality of sensation called sexual
not most, instances of cryptorchidism are caused by
sensation. The slippery massaging action of intercourse
abnormally formed testes that are unable to secrete
on the glans stimulates the sensory end-organs, and the
1002
Unit XIV Endocrinology and Reproduction
sexual signals in turn pass through the pudendal nerve,
then through the sacral plexus into the sacral portion
of the spinal cord, and finally up the cord to undefined
Deep penile
areas of the brain.
fascia
Central artery
Impulses may also enter the spinal cord from
Corpus
areas adjacent to the penis to aid in stimulating the
cavernosum
sexual act. For instance, stimulation of the anal epithe-
lium, the scrotum, and perineal structures in general
Corpus
Urethra
spongiosum
can send signals into the cord that add to the sexual
sensation. Sexual sensations can even originate in
internal structures, such as in areas of the urethra,
Figure 80-6
bladder, prostate, seminal vesicles, testes, and vas
Erectile tissue of the penis.
deferens. Indeed, one of the causes of “sexual drive”
is filling of the sexual organs with secretions. Mild
infection and inflammation of these sexual organs
sometimes cause almost continual sexual desire,
This erectile tissue consists of large cavernous sinu-
and some “aphrodisiac” drugs, such as cantharidin,
soids, which are normally relatively empty of blood
increase sexual desire by irritating the bladder and
but become dilated tremendously when arterial
urethral mucosa, inducing inflammation and vascular
blood flows rapidly into them under pressure while the
congestion.
venous outflow is partially occluded. Also, the erectile
bodies, especially the two corpora cavernosa, are
Psychic Element of Male Sexual Stimulation. Appropriate
surrounded by strong fibrous coats; therefore, high
psychic stimuli can greatly enhance the ability of a
pressure within the sinusoids causes ballooning of the
person to perform the sexual act. Simply thinking
erectile tissue to such an extent that the penis becomes
sexual thoughts or even dreaming that the act of inter-
hard and elongated. This is the phenomenon of
course is being performed can initiate the male act,
erection.
culminating in ejaculation. Indeed, nocturnal emissions
during dreams occur in many males during some
Lubrication, a Parasympathetic Function. During sexual
stages of sexual life, especially during the teens.
stimulation, the parasympathetic impulses, in addition
to promoting erection, cause the urethral glands and
Integration of the Male Sexual Act in the Spinal Cord.
the bulbourethral glands to secrete mucus. This mucus
Although psychic factors usually play an important
flows through the urethra during intercourse to aid in
part in the male sexual act and can initiate or inhibit
the lubrication during coitus. However, most of the
it, brain function is probably not necessary for its per-
lubrication of coitus is provided by the female sexual
formance because appropriate genital stimulation can
organs rather than by the male. Without satisfactory
cause ejaculation in some animals and occasionally in
lubrication, the male sexual act is seldom successful
humans after their spinal cords have been cut above
because unlubricated intercourse causes grating,
the lumbar region. The male sexual act results from
painful sensations that inhibit rather than excite sexual
inherent reflex mechanisms integrated in the sacral
sensations.
and lumbar spinal cord, and these mechanisms can be
initiated by either psychic stimulation from the brain
Emission and Ejaculation—Function of the Sympathetic
or actual sexual stimulation from the sex organs, but
Nerves. Emission and ejaculation are the culmination
usually it is a combination of both.
of the male sexual act. When the sexual stimulus
becomes extremely intense, the reflex centers of the
spinal cord begin to emit sympathetic impulses that
Stages of the Male Sexual Act
leave the cord at T-12 to L-2 and pass to the genital
organs through the hypogastric and pelvic sympathetic
Penile Erection—Role of the Parasympathetic Nerves. Penile
nerve plexuses to initiate emission, the forerunner of
erection is the first effect of male sexual stimulation,
ejaculation.
and the degree of erection is proportional to the
Emission begins with contraction of the vas defer-
degree of stimulation, whether psychic or physical.
ens and the ampulla to cause expulsion of sperm into
Erection is caused by parasympathetic impulses that
the internal urethra. Then, contractions of the muscu-
pass from the sacral portion of the spinal cord through
lar coat of the prostate gland followed by contraction
the pelvic nerves to the penis. These parasympathetic
of the seminal vesicles expel prostatic and seminal
nerve fibers, in contrast to most other parasympathetic
fluid also into the urethra, forcing the sperm forward.
fibers, are believed to release nitric oxide and/or
All these fluids mix in the internal urethra with mucus
vasoactive intestinal peptide in addition to acetyl-
already secreted by the bulbourethral glands to form
choline. The nitric oxide especially relaxes the arteries
the semen. The process to this point is emission.
of the penis, as well as relaxes the trabecular mesh-
The filling of the internal urethra with semen
work of smooth muscle fibers in the erectile tissue of
elicits sensory signals that are transmitted through the
the corpora cavernosa and corpus spongiosum in the
pudendal nerves to the sacral regions of the cord,
shaft of the penis, shown in Figure 80-6.
giving the feeling of sudden fullness in the internal
Chapter 80
Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
1003
genital organs. Also, these sensory signals further
excite rhythmical contraction of the internal genital
organs and cause contraction of the ischiocavernosus
Interstitial cells
and bulbocavernosus muscles that compress the bases
of Leydig
of the penile erectile tissue. These effects together
cause rhythmical, wavelike increases in pressure in
Blood vessel
both the erectile tissue of the penis and the genital
ducts and urethra, which “ejaculate” the semen from
the urethra to the exterior. This final process is called
Fibroblasts
ejaculation. At the same time, rhythmical contractions
of the pelvic muscles and even of some of the muscles
of the body trunk cause thrusting movements of the
pelvis and penis, which also help propel the semen into
the deepest recesses of the vagina and perhaps even
Germinal
slightly into the cervix of the uterus.
epithelium
This entire period of emission and ejaculation is
called the male orgasm. At its termination, the male
sexual excitement disappears almost entirely within
1 to 2 minutes and erection ceases, a process called
Figure 80-7
resolution.
Interstitial cells of Leydig, the cells that secrete testosterone,
located in the interstices between the seminiferous tubules.
Testosterone and Other Male
Sex Hormones
than 5 per cent of the total in the adult male) that even
Secretion, Metabolism, and Chemistry
in women they do not cause significant masculine char-
of the Male Sex Hormone
acteristics, except for causing growth of pubic and axil-
lary hair. But when an adrenal tumor of the adrenal
androgen-producing cells occurs, the quantity of
Secretion of Testosterone by the Interstitial Cells of Leydig
androgenic hormones may then become great enough
in the Testes. The testes secrete several male sex
to cause all the usual male secondary sexual charac-
hormones, which are collectively called androgens,
teristics to occur even in the female. These effects
including testosterone, dihydrotestosterone, and
are described in connection with the adrenogenital
androstenedione. Testosterone is so much more abun-
syndrome in Chapter 77.
dant than the others that one can consider it to be the
Rarely, embryonic rest cells in the ovary can deve-
significant testicular hormone, although as we shall
lop into a tumor that produces excessive quantities
see, much, if not most, of the testosterone is eventually
of androgens in women; one such tumor is the
converted into the more active hormone dihy-
arrhenoblastoma. The normal ovary also produces
minute quantities of androgens, but they are not
drotestosterone in the target tissues.
significant.
Testosterone is formed by the interstitial cells of
Leydig, which lie in the interstices between the semi-
Chemistry of the Androgens. All androgens are steroid
niferous tubules and constitute about 20 per cent of
compounds, as shown by the formulas in Figure 80-8 for
the mass of the adult testes, as shown in Figure 80-7.
testosterone and dihydrotestosterone. Both in the testes
Leydig cells are almost nonexistent in the testes during
and in the adrenals, the androgens can be synthesized
childhood when the testes secrete almost no testos-
either from cholesterol or directly from acetyl coen-
terone, but they are numerous in the newborn male
zyme A.
infant for the first few months of life and in the adult
male any time after puberty; at both these times the
Metabolism of Testosterone. After secretion by the testes,
about 97 per cent of the testosterone becomes either
testes secrete large quantities of testosterone. Fur-
loosely bound with plasma albumin or more tightly
thermore, when tumors develop from the interstitial
bound with a beta globulin called sex hormone-binding
cells of Leydig, great quantities of testosterone are
globulin and circulates in the blood in these states
secreted. Finally, when the germinal epithelium of the
for
30 minutes to several hours. By that time, the
testes is destroyed by x-ray treatment or excessive
testosterone either is transferred to the tissues or is
heat, the Leydig cells, which are less easily destroyed,
degraded into inactive products that are subsequently
often continue to produce testosterone.
excreted.
Much of the testosterone that becomes fixed to
Secretion of Androgens Elsewhere in the Body. The term
the tissues is converted within the tissue cells to dihy-
“androgen” means any steroid hormone that has mas-
drotestosterone, especially in certain target organs such
culinizing effects, including testosterone itself; it also
as the prostate gland in the adult and the external gen-
includes male sex hormones produced elsewhere in the
italia of the male fetus. Some actions of testosterone are
body besides the testes. For instance, the adrenal glands
dependent on this conversion, whereas other actions are
secrete at least five androgens, although the total mas-
not. The intracellular functions are discussed later in the
culinizing activity of all these is normally so slight (less
chapter.
1004
Unit XIV Endocrinology and Reproduction
Functions of Testosterone During
Fetal Development
OH
Testosterone begins to be elaborated by the male fetal
CH3
testes at about the seventh week of embryonic life.
OH
Indeed, one of the major functional differences
CH3
CH
3
between the female and the male sex chromosome is
that the male chromosome causes the newly develop-
3
CH
ing genital ridge to secrete testosterone, whereas the
female chromosome causes this ridge to secrete estro-
O
gens. Injection of large quantities of male sex hormone
Testosterone
into pregnant animals causes development of male
O
sexual organs even though the fetus is female. Also,
H
H
removal of the testes in the early male fetus causes
Dihydrotestosterone
development of female sexual organs.
Thus, testosterone secreted first by the genital ridges
and later by the fetal testes is responsible for the devel-
opment of the male body characteristics, including
Figure 80-8
the formation of a penis and a scrotum rather than
formation of a clitoris and a vagina. Also, it causes
Testosterone and dihydrotestosterone.
formation of the prostate gland, seminal vesicles, and
male genital ducts, while at the same time suppressing
the formation of female genital organs.
Degradation and Excretion of Testosterone. The testosterone
that does not become fixed to the tissues is rapidly con-
Effect of Testosterone to Cause Descent of the Testes. The
verted, mainly by the liver, into androsterone and dehy-
testes usually descend into the scrotum during the last
droepiandrosterone and simultaneously conjugated as
2 to 3 months of gestation when the testes begin secret-
either glucuronides or sulfates (glucuronides, particu-
ing reasonable quantities of testosterone. If a male
larly). These are excreted either into the gut by way of
child is born with undescended but otherwise normal
the liver bile or into the urine through the kidneys.
testes, the administration of testosterone usually
causes the testes to descend in the usual manner if the
Production of Estrogen in the Male. In addition to testos-
inguinal canals are large enough to allow the testes to
terone, small amounts of estrogens are formed in the
pass.
male (about one fifth the amount in the nonpregnant
Administration of gonadotropic hormones, which
female), and a reasonable quantity of estrogens can be
recovered from a man’s urine. The exact source of estro-
stimulate the Leydig cells of the newborn child’s testes
gens in the male is unclear, but the following are known:
to produce testosterone, can also cause the testes to
(1) the concentration of estrogens in the fluid of the
descend. Thus, the stimulus for descent of the testes is
seminiferous tubules is quite high and probably plays an
testosterone, indicating again that testosterone is an
important role in spermiogenesis. This estrogen is
important hormone for male sexual development
believed to be formed by the Sertoli cells by converting
during fetal life.
testosterone to estradiol.
(2) Much larger amounts
of estrogens are formed from testosterone and
Effect of Testosterone on Development
androstanediol in other tissues of the body, especially
of Adult Primary and Secondary
the liver, probably accounting for as much as 80 per cent
Sexual Characteristics
of the total male estrogen production.
After puberty, the increasing amounts of testosterone
secretion cause the penis, scrotum, and testes to
enlarge about eightfold before the age of 20 years. In
Functions of Testosterone
addition, testosterone causes the secondary sexual
characteristics of the male to develop, beginning at
In general, testosterone is responsible for the distin-
puberty and ending at maturity. These secondary
guishing characteristics of the masculine body. Even
sexual characteristics, in addition to the sexual organs
during fetal life, the testes are stimulated by chorionic
themselves, distinguish the male from the female as
gonadotropin from the placenta to produce moderate
follows.
quantities of testosterone throughout the entire period
of fetal development and for 10 or more weeks after
Effect on the Distribution of Body Hair. Testosterone causes
birth; thereafter, essentially no testosterone is
growth of hair (1) over the pubis, (2) upward along the
produced during childhood until about the ages of 10
linea alba of the abdomen sometimes to the umbilicus
to 13 years. Then testosterone production increases
and above, (3) on the face, (4) usually on the chest, and
rapidly under the stimulus of anterior pituitary
(5) less often on other regions of the body, such as the
gonadotropic hormones at the onset of puberty and
back. It also causes the hair on most other portions of
lasts throughout most of the remainder of life, as
the body to become more prolific.
shown in Figure 80-9, dwindling rapidly beyond age 50
to become 20 to 50 per cent of the peak value by age
Baldness. Testosterone decreases the growth of hair on
80.
the top of the head; a man who does not have func-
Chapter 80
Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
1005
Fetal
Neonatal
Pubertal
Adult
Old age
100
5.0
Figure 80-9
2.5
50
The different stages of male
sexual function as reflected by
average plasma testosterone
concentrations
(red line) and
sperm production (blue line) at
different ages.
(Modified from
1st
2nd
3rd
1
10
17
40
60
80
Griffin JF, Wilson JD: The testis. In:
Trimester
Year
Bondy PK, Rosenberg LE [eds]:
gestation
Metabolic Control and Disease,
Birth
8th ed. Philadelphia: WB Saun-
ders Co, 1980.)
tional testes does not become bald. However, many
to deposition of proteins in the skin, and the changes
virile men never become bald because baldness is a
in the voice also result partly from this protein ana-
result of two factors: first, a genetic background for the
bolic function of testosterone.
development of baldness and, second, superimposed
Because of the great effect that testosterone and
on this genetic background, large quantities of andro-
other androgens have on the body musculature,
genic hormones. A woman who has the appropriate
synthetic androgens are widely used by athletes to
genetic background and who develops a long-
improve their muscular performance. This practice is to
sustained androgenic tumor becomes bald in the same
be severely deprecated because of prolonged harmful
manner as does a man.
effects of excess androgens, as we discuss in Chapter 84
in relation to sports physiology. Testosterone or syn-
Effect on the Voice. Testosterone secreted by the testes
thetic androgens are also occasionally used in old age
or injected into the body causes hypertrophy of the
as a “youth hormone” to improve muscle strength and
laryngeal mucosa and enlargement of the larynx. The
vigor, but with questionable results.
effects cause at first a relatively discordant, “cracking”
voice, but this gradually changes into the typical adult
Testosterone Increases Bone Matrix and Causes Calcium Reten-
masculine voice.
tion. After the great increase in circulating testos-
terone that occurs at puberty (or after prolonged
Testosterone Increases Thickness of the Skin and Can Con-
injection of testosterone), the bones grow considerably
tribute to Development of Acne. Testosterone increases
thicker and deposit considerable additional calcium
the thickness of the skin over the entire body and
salts. Thus, testosterone increases the total quantity
increases the ruggedness of the subcutaneous tissues.
of bone matrix and causes calcium retention. The
Testosterone also increases the rate of secretion by
increase in bone matrix is believed to result from the
some or perhaps all the body’s sebaceous glands.
general protein anabolic function of testosterone plus
Especially important is excessive secretion by the
deposition of calcium salts in response to the increased
sebaceous glands of the face, because this can result
protein.
in acne. Therefore, acne is one of the most common
Testosterone has a specific effect on the pelvis to (1)
features of male adolescence when the body is first
narrow the pelvic outlet, (2) lengthen it, (3) cause a
becoming introduced to increased testosterone. After
funnel-like shape instead of the broad ovoid shape of
several years of testosterone secretion, the skin nor-
the female pelvis, and (4) greatly increase the strength
mally adapts to the testosterone in a way that allows
of the entire pelvis for load-bearing. In the absence of
it to overcome the acne.
testosterone, the male pelvis develops into a pelvis that
is similar to that of the female.
Testosterone Increases Protein Formation and Muscle Devel-
Because of the ability of testosterone to increase the
opment. One of the most important male characteris-
size and strength of bones, it is often used in older men
tics is development of increasing musculature after
to treat osteoporosis.
puberty, averaging about a 50 per cent increase in
When great quantities of testosterone (or any other
muscle mass over that in the female. This is associated
androgen) are secreted abnormally in the still-growing
with increased protein in the nonmuscle parts of the
child, the rate of bone growth increases markedly,
body as well. Many of the changes in the skin are due
causing a spurt in total body height. However, the
1006
Unit XIV Endocrinology and Reproduction
testosterone also causes the epiphyses of the long
in the prostate gland has also increased and there
bones to unite with the shafts of the bones at an early
has been a simultaneous increase in the number of
age. Therefore, despite the rapidity of growth, this
prostatic cells.
early uniting of the epiphyses prevents the person
Testosterone stimulates production of proteins
from growing as tall as he would have grown had
virtually everywhere in the body, although more
testosterone not been secreted at all. Even in normal
specifically affecting those proteins in “target” organs
men, the final adult height is slightly less than that
or tissues responsible for the development of both
which occurs in males castrated before puberty.
primary and secondary male sexual characteristics.
Recent studies suggest that testosterone, like other
Testosterone Increases Basal Metabolism. Injection of
steroidal hormones, may also exert some rapid, non-
large quantities of testosterone can increase the basal
genomic effects that do not require synthesis of new
metabolic rate by as much as 15 per cent. Also, even
proteins. The physiological role of these nongenomic
the usual quantity of testosterone secreted by the
actions of testosterone, however, has yet to be
testes during adolescence and early adult life increases
determined.
the rate of metabolism some 5 to 10 per cent above
the value that it would be were the testes not active.
This increased rate of metabolism is possibly an
Control of Male Sexual Functions
indirect result of the effect of testosterone on protein
by Hormones from the Hypothalamus
anabolism, the increased quantity of proteins—the
and Anterior Pituitary Gland
enzymes especially—increasing the activities of all
cells.
A major share of the control of sexual functions in
both the male and the female begins with secretion
Effect on Red Blood Cells. When normal quantities of
of gonadotropin-releasing hormone (GnRH) by the
testosterone are injected into a castrated adult, the
hypothalamus (see Figure 80-10). This hormone in
number of red blood cells per cubic millimeter of
turn stimulates the anterior pituitary gland to secrete
blood increases 15 to 20 per cent. Also, the average
two other hormones called gonadotropic hormones:
man has about 700,000 more red blood cells per cubic
(1) luteinizing hormone (LH) and (2) follicle-stimulat-
millimeter than the average woman. This difference
ing hormone (FSH). In turn, LH is the primary stimu-
may be due partly to the increased metabolic rate that
lus for the secretion of testosterone by the testes, and
occurs after testosterone administration rather than
FSH mainly stimulates spermatogenesis.
to a direct effect of testosterone on red blood cell
production.
GnRH and Its Effect in Increasing the
Secretion of LH and FSH
Effect on Electrolyte and Water Balance. As pointed out in
GnRH is a 10-amino acid peptide secreted by neurons
Chapter 77, many steroid hormones can increase the
whose cell bodies are located in the arcuate nuclei of
reabsorption of sodium in the distal tubules of the
the hypothalamus. The endings of these neurons
kidneys. Testosterone also has such an effect, but only
terminate mainly in the median eminence of the
to a minor degree in comparison with the adrenal min-
hypothalamus, where they release GnRH into the
eralocorticoids. Nevertheless, after puberty, the blood
hypothalamic-hypophysial portal vascular system.
and extracellular fluid volumes of the male in relation
Then the GnRH is transported to the anterior
to body weight increase as much as 5 to 10 per cent.
pituitary gland in the hypophysial portal blood and
stimulates the release of the two gonadotropins, LH
and FSH.
Basic Intracellular Mechanism
GnRH is secreted intermittently a few minutes at a
of Action of Testosterone
time once every 1 to 3 hours. The intensity of this
hormone’s stimulus is determined in two ways: (1) by
Most of the effects of testosterone result basically
the frequency of these cycles of secretion and (2) by
from increased rate of protein formation in the target
the quantity of GnRH released with each cycle.
cells. This has been studied extensively in the prostate
The secretion of LH by the anterior pituitary gland
gland, one of the organs that is most affected by testos-
is also cyclical, with LH following fairly faithfully the
terone. In this gland, testosterone enters the prostatic
pulsatile release of GnRH. Conversely, FSH secretion
cells within a few minutes after secretion. Then it
increases and decreases only slightly with each
is most often converted, under the influence of the
fluctuation of GnRH secretion; instead, it changes
intracellular enzyme 5a-reductase, to dihydrotestos-
more slowly over a period of many hours in response
terone, and this in turn binds with a cytoplasmic
to longer-term changes in GnRH. Because of the
“receptor protein.” This combination migrates to the
much closer relation between GnRH secretion and
cell nucleus, where it binds with a nuclear protein and
LH secretion, GnRH is also widely known as LH-
induces DNA-RNA transcription. Within 30 minutes,
releasing hormone.
RNA polymerase has become activated and the con-
centration of RNA begins to increase in the prostatic
Gonadotropic Hormones: LH and FSH
cells; this is followed by progressive increase in cellu-
Both of the gonadotropic hormones, LH and FSH, are
lar protein. After several days, the quantity of DNA
secreted by the same cells, called gonadotropes, in the
Chapter 80
Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
1007
CNS
from the anterior pituitary gland. Furthermore, the
quantity of testosterone secreted increases approxi-
Behavorial
mately in direct proportion to the amount of LH
effects
available.
Mature Leydig cells are normally found in a child’s
testes for a few weeks after birth but then disappear
until after the age of about 10 years. However, either
injection of purified LH into a child at any age or
secretion of LH at puberty causes testicular interstitial
cells that look like fibroblasts to evolve into function-
ing Leydig cells.
Inhibition of Anterior Pituitary Secretion of LH and FSH by
Hypothalamus
Testosterone—Negative Feedback Control of Testosterone
-
-
Secretion. The testosterone secreted by the testes in
response to LH has the reciprocal effect of inhibiting
anterior pituitary secretion of LH (see Figure 80-10).
Most of this inhibition probably results from a direct
GnRH
effect of testosterone on the hypothalamus to decrease
+
the secretion of GnRH. This in turn causes a corre-
sponding decrease in secretion of both LH and FSH
by the anterior pituitary, and the decrease in LH
Anterior
pituitary
-
reduces the secretion of testosterone by the testes.
Thus, whenever secretion of testosterone becomes too
-
great, this automatic negative feedback effect, operat-
ing through the hypothalamus and anterior pituitary
LH
FSH
gland, reduces the testosterone secretion back toward
the desired operating level. Conversely, too little
testosterone allows the hypothalamus to secrete large
+
+
amounts of GnRH, with a corresponding increase in
Testis
anterior pituitary LH and FSH secretion and conse-
Inhibin
quent increase in testicular testosterone secretion.
+
Leydig cell
Sertoli
cell
Regulation of Spermatogenesis by FSH
Testosterone
and Testosterone
FSH binds with specific FSH receptors attached to the
Sertoli cells in the seminiferous tubules. This causes
+
+
these cells to grow and secrete various spermatogenic
substances. Simultaneously, testosterone
(and dihy-
Virilizing effects
Spermatogenesis
drotestosterone) diffusing into the seminiferous
tubules from the Leydig cells in the interstitial spaces
also has a strong tropic effect on spermatogenesis.
Figure 80-10
Thus, to initiate spermatogenesis, both FSH and testos-
terone are necessary.
Feedback regulation of the hypothalamic-pituitary-testicular
axis in males. Stimulatory effects are shown by and negative
feedbacks inhibitory effects are shown by
GnRH,
Negative Feedback Control of Seminiferous Tubule Activity—
gonadotropin-releasing hormone; LH, luteinizing hormone; FSH,
Role of the Hormone Inhibin. When the seminiferous
follicle-stimulating hormone.
tubules fail to produce sperm, secretion of FSH by
the anterior pituitary gland increases markedly. Con-
versely, when spermatogenesis proceeds too rapidly,
anterior pituitary gland. In the absence of GnRH
pituitary secretion of FSH diminishes. The cause of this
secretion from the hypothalamus, the gonadotropes in
negative feedback effect on the anterior pituitary is
the pituitary gland secrete almost no LH or FSH.
believed to be secretion by the Sertoli cells of still
LH and FSH are glycoproteins. They exert their
another hormone called inhibin (see Figure 80-10).
effects on their target tissues in the testes mainly by
This hormone has a strong direct effect on the ante-
activating the cyclic adenosine monophosphate second
rior pituitary gland to inhibit the secretion of FSH and
messenger system, which in turn activates specific
possibly a slight effect on the hypothalamus to inhibit
enzyme systems in the respective target cells.
secretion of GnRH.
Inhibin is a glycoprotein, like both LH and FSH,
Testosterone—Regulation of Its Production by LH. Testos-
having a molecular weight between 10,000 and 30,000.
terone is secreted by the interstitial cells of Leydig in
It has been isolated from cultured Sertoli cells. Its
the testes, but only when they are stimulated by LH
potent inhibitory feedback effect on the anterior pitu-
1008
Unit XIV Endocrinology and Reproduction
itary gland provides an important negative feedback
Abnormalities of Male
mechanism for control of spermatogenesis, operating
Sexual Function
simultaneously with and in parallel to the negative
feedback mechanism for control of testosterone
Prostate Gland and
secretion.
Its Abnormalities
The prostate gland remains relatively small throughout
Psychic Factors That Affect Gonadotropin
childhood and begins to grow at puberty under the
Secretion and Sexual Activity
stimulus of testosterone. This gland reaches an almost
Many psychic factors, feeding especially from the
stationary size by the age of 20 years and remains at
limbic system of the brain into the hypothalamus, can
this size up to the age of about 50 years. At that time, in
some men it begins to involute, along with decreased
affect the rate of secretion of GnRH by the hypothal-
production of testosterone by the testes.
amus and therefore can also affect most other aspects
A benign prostatic fibroadenoma frequently develops
of sexual and reproductive functions in both the male
in the prostate in many older men and can cause urinary
and the female. For instance, transporting a prize bull
obstruction. This hypertrophy is caused not by testos-
in a rough truck is said to inhibit the bull’s fertility—
terone but instead by abnormal overgrowth of prostate
and the human male is hardly different.
tissue itself.
Cancer of the prostate gland is a different problem
and is a common cause of death, accounting for about
Human Chorionic Gonadotropin Secreted by
2 to 3 per cent of all male deaths. Once cancer of the
the Placenta During Pregnancy Stimulates
prostate gland does occur, the cancerous cells are
Testosterone Secretion by the Fetal Testes
usually stimulated to more rapid growth by testosterone
During pregnancy, the hormone human chorionic
and are inhibited by removal of both testes so that
gonadotropin (hCG) is secreted by the placenta, and
testosterone cannot be formed. Prostatic cancer usually
it circulates both in the mother and in the fetus. This
can be inhibited by administration of estrogens.
hormone has almost the same effects on the sexual
Even some patients who have prostatic cancer that has
organs as LH.
already metastasized to almost all the bones of the body
can be successfully treated for a few months to years by
During pregnancy, if the fetus is a male, hCG from
removal of the testes, by estrogen therapy, or by both;
the placenta causes the testes of the fetus to secrete
after this therapy the metastases usually diminish in size
testosterone. This testosterone is critical for promoting
and the bones partially heal. This treatment does not
formation of the male sexual organs, as pointed out
stop the cancer but does slow it and sometimes greatly
earlier. We discuss hCG and its functions during preg-
diminishes the severe bone pain.
nancy in greater detail in Chapter 82.
Hypogonadism in the Male
Puberty and Regulation of Its Onset
Initiation of the onset of puberty has long been a
When the testes of a male fetus are nonfunctional
mystery. But it has now been determined that during
during fetal life, none of the male sexual characteristics
childhood the hypothalamus simply does not secrete
develop in the fetus. Instead, female organs are formed.
significant amounts of GnRH. One of the reasons
The reason for this is that the basic genetic characteris-
for this is that, during childhood, the slightest secretion
tic of the fetus, whether male or female, is to form
of any sex steroid hormones exerts a strong inhibitory
female sexual organs if there are no sex hormones. But
in the presence of testosterone, formation of female
effect on hypothalamic secretion of GnRH. Yet, for
sexual organs is suppressed, and instead, male organs
reasons still not understood, at the time of puberty, the
are induced.
secretion of hypothalamic GnRH breaks through the
When a boy loses his testes before puberty, a state of
childhood inhibition, and adult sexual life begins.
eunuchism ensues in which he continues to have infan-
tile sex organs and other infantile sexual characteristics
Male Adult Sexual Life and Male Climacteric. After puberty,
throughout life. The height of an adult eunuch is slightly
gonadotropic hormones are produced by the male pitu-
greater than that of a normal man because the bone
itary gland for the remainder of life, and at least some
epiphyses are slow to unite, although the bones are
spermatogenesis usually continues until death. Most
quite thin and the muscles are considerably weaker
men, however, begin to exhibit slowly decreasing sexual
than those of a normal man. The voice is childlike, there
functions in their late 40s or 50s, and one study showed
is no loss of hair on the head, and the normal adult
that the average age for terminating intersexual rela-
masculine hair distribution on the face and elsewhere
tions was 68, although the variation was great. This
does not occur.
decline in sexual function is related to decrease in
When a man is castrated after puberty, some of his
testosterone secretion, as shown in Figure 80-9. The
male secondary sexual characteristics revert to those of
decrease in male sexual function is called the male cli-
a child and others remain of adult masculine character.
macteric. Occasionally the male climacteric is associated
The sexual organs regress slightly in size but not to a
with symptoms of hot flashes, suffocation, and psychic
childlike state, and the voice regresses from the bass
disorders similar to the menopausal symptoms of the
quality only slightly. Conversely, there is loss of mascu-
female. These symptoms can be abrogated by adminis-
line hair production, loss of the thick masculine bones,
tration of testosterone, synthetic androgens, or even
and loss of the musculature of the virile male.
estrogens that are used for treatment of menopausal
Also in a castrated adult male, sexual desires are
symptoms in the female.
decreased but not lost, provided sexual activities have
Chapter 80
Reproductive and Hormonal Functions of the Male (and Function of the Pineal Gland)
1009
that the eventual adult height actually is considerably
less than that which would have been achieved other-
wise. Such interstitial cell tumors also cause excessive
development of the male sexual organs, all skeletal
muscles, and other male sexual characteristics. In the
adult male, small interstitial cell tumors are difficult
to diagnose because masculine features are already
present.
Much more common than the interstitial Leydig cell
tumors are tumors of the germinal epithelium. Because
germinal cells are capable of differentiating into almost
any type of cell, many of these tumors contain multiple
tissues, such as placental tissue, hair, teeth, bone, skin,
and so forth, all found together in the same tumorous
mass called a teratoma. These tumors often secrete few
hormones, but if a significant quantity of placental tissue
develops in the tumor, it may secrete large quantities
of hCG with functions similar to those of LH. Also,
estrogenic hormones are sometimes secreted by these
tumors and cause the condition called gynecomastia
(overgrowth of the breasts).
Pineal Gland—Its Function
in Controlling Seasonal
Fertility in Some Animals
For as long as the pineal gland has been known to exist,
myriad functions have been ascribed to it, including its
(1) being the seat of the soul, (2) enhancing sex, (3)
staving off infection, (4) promoting sleep, (5) enhancing
mood, and (6) increasing longevity (as much as 10 to 25
per cent). It is known from comparative anatomy that
the pineal gland is a vestigial remnant of what was a
third eye located high in the back of the head in some
lower animals. Many physiologists have been content
Figure 80-11
with the idea that this gland is a nonfunctional remnant,
but others have claimed for many years that it plays
Adiposogenital syndrome in an adolescent male. Note the obesity
important roles in the control of sexual activities
and childlike sexual organs. (Courtesy Dr. Leonard Posey.)
and reproduction, functions that still others said were
nothing more than the fanciful imaginings of physiolo-
gists preoccupied with sexual delusions.
But now, after years of dispute, it looks as though the
been practiced previously. Erection can still occur as
sex advocates have won and that the pineal gland does
before, although with less ease, but it is rare that ejacu-
indeed play a regulatory role in sexual and reproductive
lation can take place, primarily because the semen-
function. In lower animals that bear their young at
forming organs degenerate and there has been a loss of
certain seasons of the year and in which the pineal gland
the testosterone-driven psychic desire.
has been removed or the nervous circuits to the pineal
Some instances of hypogonadism are caused by a
gland have been sectioned, the normal periods of sea-
genetic inability of the hypothalamus to secrete normal
sonal fertility are lost. To these animals, such seasonal
amounts of GnRH. This often is associated with a
fertility is important because it allows birth of the off-
simultaneous abnormality of the feeding center of the
spring at the time of year, usually springtime or early
hypothalamus, causing the person to greatly overeat.
summer, when survival is most likely. The mechanism of
Consequently, obesity occurs along with eunuchism. A
this effect is not entirely clear, but it seems to be the
patient with this condition is shown in Figure 80-11; the
following.
condition is called adiposogenital syndrome, Fröhlich’s
First, the pineal gland is controlled by the amount of
syndrome, or hypothalamic eunuchism.
light or “time pattern” of light seen by the eyes each day.
For instance, in the hamster, greater than 13 hours of
darkness each day activates the pineal gland, whereas
Testicular Tumors and
less than that amount of darkness fails to activate it,
Hypergonadism in the Male
with a critical balance between activation and nonacti-
vation. The nervous pathway involves the passage of
Interstitial Leydig cell tumors develop in rare instances
light signals from the eyes to the suprachiasmal nucleus
in the testes, but when they do develop, they sometimes
of the hypothalamus and then to the pineal gland,
produce as much as 100 times the normal quantities of
activating pineal secretion.
testosterone. When such tumors develop in young chil-
Second, the pineal gland secretes melatonin and
dren, they cause rapid growth of the musculature and
several other, similar substances. Either melatonin or
bones but also cause early uniting of the epiphyses, so
one of the other substances is believed to pass either by
1010
Unit XIV Endocrinology and Reproduction
way of the blood or through the fluid of the third
DeMarzo AM, Nelson WG, Isaacs WB, Epstein JI: Patho-
ventricle to the anterior pituitary gland to decrease
logical and molecular aspects of prostate cancer. Lancet
gonadotropic hormone secretion.
361:955, 2003.
Thus, in the presence of pineal gland secretion,
Foresta C, Moro E, Ferlin A: Y chromosome microdeletions
gonadotropic hormone secretion is suppressed in some
and alterations of spermatogenesis. Endocr Rev 22:226,
species of animals, and the gonads become inhibited and
2001.
even partly involuted. This is what presumably occurs
Heinlein CA, Chang C: Androgen receptor (AR) coregula-
during the early winter months when there is increasing
tors: an overview. Endocr Rev 23:175, 2002.
darkness. But after about 4 months of dysfunction,
Jobling MA, Tyler-Smith C: The human Y chromosome: an
gonadotropic hormone secretion breaks through the
evolutionary marker comes of age. Nat Rev Genet 4:598,
inhibitory effect of the pineal gland and the gonads
2003.
become functional once more, ready for a full spring-
Kandeel FR, Koussa VK, Swerdloff RS: Male sexual func-
time of activity.
tion and its disorders: physiology, pathophysiology, clinical
But does the pineal gland have a similar function for
investigation, and treatment. Endocr Rev 22:342, 2001.
control of reproduction in humans? The answer to this
Lahn BT, Pearson NM, Jegalian K: The human Y chromo-
question is unknown. However, tumors often occur in
some, in the light of evolution. Nat Rev Genet 2:207, 2001.
the region of the pineal gland. Some of these secrete
Lanfranco F, Kamischke A, Zitzmann M, Nieschlag E:
excessive quantities of pineal hormones, whereas others
Klinefelter’s syndrome. Lancet 364:273, 2004.
are tumors of surrounding tissue and press on the pineal
Liu PY, Death AK, Handelsman DJ: Androgens and
gland to destroy it. Both types of tumors are often asso-
cardiovascular disease. Endocr Rev 24:313, 2003.
ciated with hypogonadal or hypergonadal function. So
Nelson WG, De Marzo AM, Isaacs WB: Prostate cancer.
perhaps the pineal gland does play at least some role in
N Engl J Med 349:366, 2003.
controlling sexual drive and reproduction in humans.
Nelson PS, Montgomery B: Unconventional therapy for
prostate cancer: good, bad or questionable? Nat Rev
Cancer 3:845, 2003.
References
O’Donnell L, Robertson KM, Jones ME, Simpson ER:
Estrogen and spermatogenesis. Endocr Rev 22:289, 2001.
Anderson RA, Baird DT: Male contraception. Endocr Rev
Plant TM, Marshall GR: The functional significance of FSH
23:735, 2002.
in spermatogenesis and the control of its secretion in male
Barry MJ, Roehrborn CG: Benign prostatic hyperplasia.
primates. Endocr Rev 22:764, 2001.
BMJ 323:1042, 2001.
Reckelhoff JF: Gender differences in the regulation of blood
Brennan J, Capel B: One tissue, two fates: molecular genetic
pressure. Hypertension 37:1199, 2001.
events that underlie testis versus ovary development. Nat
Rhoden EL, Morgentaler A: Risks of testosterone-
Rev Genet 5:509, 2004.
replacement therapy and recommendations for monitor-
Cajochen C, Krauchi K, Wirz-Justice A: Role of melatonin
ing. N Engl J Med 350:482, 2004.
in the regulation of human circadian rhythms and sleep.
Riggs BL, Khosla S, Melton LJ 3rd: Sex steroids and the con-
J Neuroendocrinol 15:432, 2003.
struction and conservation of the adult skeleton. Endocr
Cheng CY, Mruk DD: Cell junction dynamics in the testis:
Rev 23:279, 2002.
Sertoli-germ cell interactions and male contraceptive
Shabsigh R, Anastasiadis AG: Erectile dysfunction. Annu
development. Physiol Rev 82:825, 2002.
Rev Med 54:153, 2003.
Cooke HJ, Saunders PT: Mouse models of male infertility.
Simonneaux V, Ribelayga C: Generation of the melatonin
Nat Rev Genet 3:790, 2002.
endocrine message in mammals: a review of the complex
de Kretser DM: Is spermatogenic damage associated with
regulation of melatonin synthesis by norepinephrine,
Leydig cell dysfunction? J Clin Endocrinol Metab 89:3158,
peptides, and other pineal transmitters. Pharmacol Rev
2004.
55:325, 2003.
C H A P T E R
8
1
Female Physiology Before
Pregnancy and Female Hormones
Female reproductive functions can be divided into
two major phases: (1) preparation of the female
body for conception and pregnancy, and (2) the
period of pregnancy itself. This chapter is concerned
with preparation of the female body for pregnancy,
and Chapter 82 presents the physiology of preg-
nancy and childbirth.
Physiologic Anatomy of the Female
Sexual Organs
Figures 81-1 and 81-2 show the principal organs of the human female reproductive
tract, the most important of which are the ovaries, fallopian tubes, uterus, and vagina.
Reproduction begins with the development of ova in the ovaries. In the middle of
each monthly sexual cycle, a single ovum is expelled from an ovarian follicle into
the abdominal cavity near the open fimbriated ends of the two fallopian tubes. This
ovum then passes through one of the fallopian tubes into the uterus; if it has been
fertilized by a sperm, it implants in the uterus, where it develops into a fetus, a
placenta, and fetal membranes—and eventually into a baby.
During fetal life, the outer surface of the ovary is covered by a germinal epithe-
lium, which embryologically is derived from the epithelium of the germinal ridges.
As the female fetus develops, primordial ova differentiate from this germinal epithe-
lium and migrate into the substance of the ovarian cortex. Each ovum then collects
around it a layer of spindle cells from the ovarian stroma (the supporting tissue of
the ovary) and causes them to take on epithelioid characteristics; they are then
called granulosa cells. The ovum surrounded by a single layer of granulosa cells is
called a primordial follicle. The ovum itself at this stage is still immature, requiring
two more cell divisions before it can be fertilized by a sperm. At this time, the ovum
is called a primary oocyte.
During all the reproductive years of adult life, between about 13 and 46 years of
age, 400 to 500 of the primordial follicles develop enough to expel their ova—one
each month; the remainder degenerate (become atretic). At the end of reproductive
capability (at menopause), only a few primordial follicles remain in the ovaries, and
even these degenerate soon thereafter.
Female Hormonal System
The female hormonal system, like that of the male, consists of three hierarchies
of hormones, as follows:
1. A hypothalamic releasing hormone, gonadotropin-releasing hormone
(GnRH)
2. The anterior pituitary sex hormones, follicle-stimulating hormone (FSH)
and luteinizing hormone (LH), both of which are secreted in response to
the release of GnRH from the hypothalamus
3. The ovarian hormones, estrogen and progesterone, which are secreted by
the ovaries in response to the two female sex hormones from the anterior
pituitary gland
These various hormones are not secreted in constant amounts throughout the
female monthly sexual cycle; they are secreted at drastically differing rates
during different parts of the cycle. Figure
81-3 shows the approximate
1011
1012
Unit XIV Endocrinology and Reproduction
changing concentrations of the anterior pituitary
during the monthly sexual cycle. It is secreted in short
gonadotropic hormones FSH and LH (bottom two
pulses averaging once every 90 minutes, as occurs in
curves) and of the ovarian hormones estradiol (estro-
the male.
gen) and progesterone (top two curves).
The amount of GnRH released from the hypothal-
Monthly Ovarian Cycle;
amus increases and decreases much less drastically
Function of the Gonadotropic
Hormones
Uterine tube
Ovary
The normal reproductive years of the female are char-
acterized by monthly rhythmical changes in the rates
of secretion of the female hormones and correspon-
ding physical changes in the ovaries and other sexual
organs. This rhythmical pattern is called the female
monthly sexual cycle (or, less accurately, the menstrual
Uterus
cycle). The duration of the cycle averages 28 days. It
may be as short as 20 days or as long as 45 days in some
Cervix
women, although abnormal cycle length is frequently
Urinary
associated with decreased fertility.
bladder
There are two significant results of the female sexual
Vagina
cycle. First, only a single ovum is normally released
Urethra
from the ovaries each month, so that normally only a
Clitoris
single fetus will begin to grow at a time. Second, the
Labium
uterine endometrium is prepared in advance for
minora
implantation of the fertilized ovum at the required
Labium
time of the month.
majora
Anus
Rectum
Gonadotropic Hormones and
Uterine tube (sectioned)
Uterine cavity
Uterine tube
Their Effects on the Ovaries
The ovarian changes that occur during the sexual cycle
depend completely on the gonadotropic hormones
FSH and LH, secreted by the anterior pituitary gland.
Ovary
In the absence of these hormones, the ovaries remain
Entrance to
Ovary
Fimbriae
uterine tube
inactive, which is the case throughout childhood,
Uterus
when almost no pituitary gonadotropic hormones are
secreted. At age 9 to 12 years, the pituitary begins to
secrete progressively more FSH and LH, which leads
Cervix
to onset of normal monthly sexual cycles beginning
between the ages of 11 and 15 years. This period of
Vagina
change is called puberty, and the time of the first men-
strual cycle is called menarche. Both FSH and LH are
Figure 81-1
small glycoproteins having molecular weights of about
Female reproductive organs.
30,000.
Ovarian ligament Ovarian stroma
Perimetrium
Isthmus of uterine tube
Ampulla of
uterine tube
Mucosal folds
of uterine tube
Endometrium
Fimbriae
Uterine cavity
Ovarian vessels
Figure 81-2
Myometrium
Corpus albicans
Uterosacral
Internal structures of the uterus,
ligament
Ovarian follicles
ovary, and a uterine tube.
Isthmus of uterus
(Redrawn from Guyton AC:
Cervical canal
Corpus luteum
Physiology of the Human Body,
Vagina
Broad ligament of uterus
Cervix
6th ed. Philadelphia: Saunders
Vaginal rugae
College Publishing, 1984.)
Chapter 81
Female Physiology Before Pregnancy and Female Hormones
1013
Zona pellucida
Antral
Preovulatory
Preantral
Antrum
follicle
Ovum
(mature) follicle
Progesterone
follicle
800
8
Primordial
600
follicle
400
Theca
200
Granulosa cells
0
0
800
Corona radiata
LH
600
400
FSH
200
Degenerating
0
corpus luteum
0
2
4
6
8 10 1214 16 18 20 22 24 2628
Ovulation
Days of female sexual cycle
Corpus luteum
Figure 81-3
Ovum
Approximate plasma concentrations of the gonadotropins and
Corona radiata
ovarian hormones during the normal female sexual cycle. FSH,
follicle-stimulating hormone; LH, luteinizing hormone.
Figure 81-4
Stages of follicular growth in the ovary, also showing formation of
the corpus luteum.
During each month of the female sexual cycle, there
is a cyclical increase and decrease of both FSH and
LH, as shown in the bottom of Figure 81-3. These
ovaries, together with some of the follicles within
cyclical variations cause cyclical ovarian changes,
them, begin to grow.
which are explained in the following sections.
The first stage of follicular growth is moderate
Both FSH and LH stimulate their ovarian target
enlargement of the ovum itself, which increases in
cells by combining with highly specific FSH and LH
diameter twofold to threefold. Then follows growth of
receptors in the ovarian target cell membranes. In
additional layers of granulosa cells in some of the fol-
turn, the activated receptors increase the cells’ rates
licles; these follicles are known as primary follicles.
of secretion and usually the growth and proliferation
of the cells as well. Almost all these stimulatory
Development of Antral and Vesicular Follicles. During the
effects result from activation of the cyclic adenosine
first few days of each monthly female sexual cycle,
monophosphate second messenger system in the cell
the concentrations of both FSH and LH secreted by
cytoplasm, which causes the formation of protein
the anterior pituitary gland increase slightly to mod-
kinase and multiple phosphorylations of key enzymes
erately, with the increase in FSH slightly greater than
that stimulate sex hormone synthesis, as explained in
that of LH and preceding it by a few days. These hor-
Chapter 74.
mones, especially FSH, cause accelerated growth of 6
to 12 primary follicles each month. The initial effect is
rapid proliferation of the granulosa cells, giving rise to
Ovarian Follicle Growth—
many more layers of these cells. In addition, spindle
“Follicular” Phase of the
cells derived from the ovary interstitium collect in
Ovarian Cycle
several layers outside the granulosa cells, giving rise to
a second mass of cells called the theca. This is divided
Figure 81-4 shows the progressive stages of follicular
into two layers. In the theca interna, the cells take on
growth in the ovaries. When a female child is born,
epithelioid characteristics similar to those of the gran-
each ovum is surrounded by a single layer of granu-
ulosa cells and develop the ability to secrete additional
losa cells; the ovum, with this granulosa cell sheath, is
steroid sex hormones (estrogen and progesterone).
called a primordial follicle, as shown in the figure.
The outer layer, the theca externa, develops into a
Throughout childhood, the granulosa cells are
highly vascular connective tissue capsule that becomes
believed to provide nourishment for the ovum and to
the capsule of the developing follicle.
secrete an oocyte maturation-inhibiting factor that
After the early proliferative phase of growth, lasting
keeps the ovum suspended in its primordial state in
for a few days, the mass of granulosa cells secretes a
the prophase stage of meiotic division. Then, after
follicular fluid that contains a high concentration of
puberty, when FSH and LH from the anterior pituitary
estrogen, one of the important female sex hormones
gland begin to be secreted in significant quantities, the
(discussed later). Accumulation of this fluid causes an
1014
Unit XIV Endocrinology and Reproduction
antrum to appear within the mass of granulosa cells, as
occupied the central portion of the follicle, to evagi-
shown in Figure 81-4.
nate outward. This viscous fluid carries with it the
The early growth of the primary follicle up to
ovum surrounded by a mass of several thousand small
the antral stage is stimulated mainly by FSH alone.
granulosa cells, called the corona radiata.
Then greatly accelerated growth occurs, leading to still
larger follicles called vesicular follicles. This acceler-
Surge of LH Is Necessary for Ovulation. LH is necessary
ated growth is caused by the following: (1) Estrogen is
for final follicular growth and ovulation. Without this
secreted into the follicle and causes the granulosa cells
hormone, even when large quantities of FSH are
to form increasing numbers of FSH receptors; this
available, the follicle will not progress to the stage
causes a positive feedback effect, because it makes the
of ovulation.
granulosa cells even more sensitive to FSH. (2) The
About 2 days before ovulation (for reasons that are
pituitary FSH and the estrogens combine to promote
not completely understood but are discussed in more
LH receptors on the original granulosa cells, thus
detail later in the chapter), the rate of secretion of LH
allowing LH stimulation to occur in addition to FSH
by the anterior pituitary gland increases markedly,
stimulation and creating an even more rapid increase
rising 6- to 10-fold and peaking about 16 hours before
in follicular secretion. (3) The increasing estrogens
ovulation. FSH also increases about 2-fold to 3-fold at
from the follicle plus the increasing LH from the ante-
the same time, and the FSH and LH act synergistically
rior pituitary gland act together to cause proliferation
to cause rapid swelling of the follicle during the last
of the follicular thecal cells and increase their secre-
few days before ovulation. The LH also has a specific
tion as well.
effect on the granulosa and theca cells, converting
Once the antral follicles begin to grow, their
them mainly to progesterone-secreting cells. There-
growth occurs almost explosively. The ovum itself
fore, the rate of secretion of estrogen begins to
also enlarges in diameter another threefold to four-
fall about 1 day before ovulation, while increasing
fold, giving a total ovum diameter increase up to 10-
amounts of progesterone begin to be secreted.
fold, or a mass increase of 1000-fold. As the follicle
It is in this environment of (1) rapid growth of the
enlarges, the ovum itself remains embedded in a mass
follicle, (2) diminishing estrogen secretion after a pro-
of granulosa cells located at one pole of the follicle.
longed phase of excessive estrogen secretion, and (3)
initiation of secretion of progesterone that ovulation
Only One Follicle Fully Matures Each Month, and the Remain-
occurs. Without the initial preovulatory surge of LH,
der Undergo Atresia. After a week or more of growth—
ovulation will not take place.
but before ovulation occurs—one of the follicles
begins to outgrow all the others; the remaining 5 to 11
Initiation of Ovulation. Figure 81-5 gives a schema for the
developing follicles involute (a process called atresia),
initiation of ovulation, showing the role of the large
and these follicles are said to become atretic.
quantity of LH secreted by the anterior pituitary
The cause of the atresia is unknown, but it has been
gland. This LH causes rapid secretion of follicular
postulated to be the following: The large amounts of
steroid hormones that contain progesterone. Within a
estrogen from the most rapidly growing follicle act on
few hours, two events occur, both of which are neces-
the hypothalamus to depress further enhancement of
sary for ovulation: (1) The theca externa (the capsule
FSH secretion by the anterior pituitary gland, in this
of the follicle) begins to release proteolytic enzymes
way blocking further growth of the less well developed
from lysosomes, and these cause dissolution of the fol-
follicles. Therefore, the largest follicle continues to
licular capsular wall and consequent weakening of the
grow because of its intrinsic positive feedback effects,
wall, resulting in further swelling of the entire follicle
while all the other follicles stop growing and actually
and degeneration of the stigma. (2) Simultaneously,
involute.
there is rapid growth of new blood vessels into the fol-
This process of atresia is important, because it nor-
licle wall, and at the same time, prostaglandins (local
mally allows only one of the follicles to grow large
hormones that cause vasodilation) are secreted into
enough each month to ovulate; this usually prevents
the follicular tissues. These two effects cause plasma
more than one child from developing with each preg-
transudation into the follicle, which contributes to
nancy. The single follicle reaches a diameter of 1 to 1.5
follicle swelling. Finally, the combination of follicle
centimeters at the time of ovulation and is called the
swelling and simultaneous degeneration of the stigma
mature follicle.
causes follicle rupture, with discharge of the ovum.
Ovulation
Ovulation in a woman who has a normal 28-day
Corpus Luteum—“Luteal” Phase
female sexual cycle occurs 14 days after the onset of
of the Ovarian Cycle
menstruation. Shortly before ovulation, the protruding
outer wall of the follicle swells rapidly, and a small area
During the first few hours after expulsion of the ovum
in the center of the follicular capsule, called the stigma,
from the follicle, the remaining granulosa and theca
protrudes like a nipple. In another 30 minutes or
interna cells change rapidly into lutein cells. They
so, fluid begins to ooze from the follicle through the
enlarge in diameter two or more times and become
stigma, and about 2 minutes later, the stigma ruptures
filled with lipid inclusions that give them a yellowish
widely, allowing a more viscous fluid, which has
appearance. This process is called luteinization, and the
Chapter 81
Female Physiology Before Pregnancy and Female Hormones
1015
Secretion by the Corpus Luteum: An Additional Function of LH.
The corpus luteum is a highly secretory organ, secret-
Luteinizing hormone
ing large amounts of both progesterone and estrogen.
Once LH (mainly that secreted during the ovulatory
surge) has acted on the granulosa and theca cells
Follicular steroid hormones
to cause luteinization, the newly formed lutein cells
(progesterone)
seem to be programmed to go through a preordained
sequence of (1) proliferation, (2) enlargement, and (3)
secretion, followed by (4) degeneration. All this occurs
Proteolytic enzymes
Follicular hyperemia
in about 12 days. We shall see in the discussion of
(collagenase)
and
pregnancy in Chapter 82 that another hormone with
prostaglandin secretion
almost exactly the same properties as LH, chorionic
gonadotropin, which is secreted by the placenta, can
Weakened follicle wall
Plasma transudation
act on the corpus luteum to prolong its life—usually
into follicle
maintaining it for at least the first 2 to 4 months of
pregnancy.
Degeneration
Follicle swelling
Involution of the Corpus Luteum and Onset of the Next Ovarian
of stigma
Cycle. Estrogen in particular and progesterone to a
lesser extent, secreted by the corpus luteum during the
luteal phase of the ovarian cycle, have strong feedback
Follicle rupture
effects on the anterior pituitary gland to maintain low
secretory rates of both FSH and LH.
Evagination of ovum
In addition, the lutein cells secrete small amounts of
the hormone inhibin, the same as the inhibin secreted
by the Sertoli cells of the male testes. This hormone
inhibits secretion by the anterior pituitary gland, espe-
Figure 81-5
cially FSH secretion. Low blood concentrations of both
Postulated mechanism of ovulation.
FSH and LH result, and loss of these hormones finally
causes the corpus luteum to degenerate completely, a
process called involution of the corpus luteum.
Final involution normally occurs at the end of
total mass of cells together is called the corpus luteum,
almost exactly 12 days of corpus luteum life, which is
which is shown in Figure 81-4. A well-developed
around the 26th day of the normal female sexual cycle,
vascular supply also grows into the corpus luteum.
2 days before menstruation begins. At this time, the
The granulosa cells in the corpus luteum develop
sudden cessation of secretion of estrogen, proges-
extensive intracellular smooth endoplasmic reticula
terone, and inhibin by the corpus luteum removes the
that form large amounts of the female sex hormones
feedback inhibition of the anterior pituitary gland,
progesterone and estrogen (more progesterone than
allowing it to begin secreting increasing amounts of
estrogen). The theca cells form mainly the androgens
FSH and LH again. FSH and LH initiate the growth
androstenedione and testosterone rather than female
of new follicles, beginning a new ovarian cycle. The
sex hormones. However, most of these hormones are
paucity of secretion of progesterone and estrogen at
also converted by the granulosa cells into the female
this time also leads to menstruation by the uterus, as
hormones.
explained later.
In the normal female, the corpus luteum grows to
about 1.5 centimeters in diameter, reaching this stage
of development 7 to 8 days after ovulation. Then it
Summary
begins to involute and eventually loses its secretory
function as well as its yellowish, lipid characteristic
About every 28 days, gonadotropic hormones from the
about 12 days after ovulation, becoming the corpus
anterior pituitary gland cause about 8 to 12 new folli-
albicans; during the ensuing few weeks, this is replaced
cles to begin to grow in the ovaries. One of these fol-
by connective tissue and over months is absorbed.
licles finally becomes “mature” and ovulates on the
14th day of the cycle. During growth of the follicles,
Luteinizing Function of LH. The change of granulosa and
mainly estrogen is secreted.
theca interna cells into lutein cells is dependent mainly
After ovulation, the secretory cells of the ovulating
on LH secreted by the anterior pituitary gland. In fact,
follicle develop into a corpus luteum that secretes
this function gives LH its name—“luteinizing,” for
large quantities of both the major female hormones,
“yellowing.” Luteinization also depends on extrusion
progesterone and estrogen. After another 2 weeks, the
of the ovum from the follicle. A yet uncharacterized
corpus luteum degenerates, whereupon the ovarian
local hormone in the follicular fluid, called luteiniza-
hormones estrogen and progesterone decrease greatly,
tion-inhibiting factor, seems to hold the luteinization
and menstruation begins. A new ovarian cycle then
process in check until after ovulation.
follows.
1016
Unit XIV Endocrinology and Reproduction
Functions of the Ovarian
peripheral tissues from androgens secreted by the
adrenal cortices and by ovarian thecal cells. Estriol is
Hormones—Estradiol
a weak estrogen; it is an oxidative product derived
and Progesterone
from both estradiol and estrone, with the conversion
occurring mainly in the liver.
The two types of ovarian sex hormones are the estro-
The estrogenic potency of b-estradiol is 12 times
gens and the progestins. By far the most important of
that of estrone and 80 times that of estriol. Consider-
the estrogens is the hormone estradiol, and by far the
ing these relative potencies, one can see that the total
most important progestin is progesterone. The estro-
estrogenic effect of b-estradiol is usually many times
gens mainly promote proliferation and growth of spe-
that of the other two together. For this reason, b-
cific cells in the body that are responsible for the
estradiol is considered the major estrogen, although
development of most secondary sexual characteristics
the estrogenic effects of estrone are not negligible.
of the female. The progestins function mainly to
prepare the uterus for pregnancy and the breasts for
Progestins. By far the most important of the progestins
lactation.
is progesterone. However, small amounts of another
progestin,
17-a-hydroxyprogesterone, are secreted
along with progesterone and have essentially the same
Chemistry of the Sex Hormones
effects. Yet, for practical purposes, it is usually reason-
able to consider progesterone the only important
Estrogens. In the normal nonpregnant female, estro-
progestin.
gens are secreted in significant quantities only by the
In the normal nonpregnant female, progesterone is
ovaries, although minute amounts are also secreted by
secreted in significant amounts only during the latter
the adrenal cortices. During pregnancy, tremendous
half of each ovarian cycle, when it is secreted by the
quantities of estrogens are also secreted by the
corpus luteum.
placenta, as discussed in Chapter 82.
As we shall see in Chapter 82, large amounts of
Only three estrogens are present in significant quan-
progesterone are also secreted by the placenta during
tities in the plasma of the human female: b-estradiol,
pregnancy, especially after the fourth month of
estrone, and estriol, the formulas for which are shown
gestation.
in Figure 81-6. The principal estrogen secreted by
the ovaries is b-estradiol. Small amounts of estrone
Synthesis of the Estrogens and Progestins. Note from the
are also secreted, but most of this is formed in the
chemical formulas of the estrogens and progesterone
in Figure 81-6 that they are all steroids. They are
synthesized in the ovaries mainly from cholesterol
derived from the blood but also to a slight extent from
H
acetyl coenzyme A, multiple molecules of which can
CH
3
OH
combine to form the appropriate steroid nucleus.
O
During synthesis, mainly progesterone and the male
CH
3
sex hormone testosterone are synthesized first; then,
during the follicular phase of the ovarian cycle, before
these two initial hormones can leave the ovaries,
almost all the testosterone and much of the proges-
HO
terone are converted into estrogens by the granulosa
b-Estradiol
cells. During the luteal phase of the cycle, far too much
HO
Estrone
progesterone is formed for all of it to be converted,
which accounts for the large secretion of progesterone
into the circulating blood at this time. Also, about one
fifteenth as much testosterone is secreted into the
H
CH3
O
plasma of the female by the ovaries as is secreted into
OH
H
the plasma of the male by the testes.
C
CH3
OH
CH3
Estrogens and Progesterone Are Transported in the Blood Bound
to Plasma Proteins. Both estrogens and progesterone
CH3
are transported in the blood bound mainly with
HO
plasma albumin and with specific estrogen- and prog-
esterone-binding globulins. The binding between these
Estriol
hormones and the plasma proteins is loose enough
O
that they are rapidly released to the tissues over a
Progesterone
period of 30 minutes or so.
Functions of the Liver in Estrogen Degradation. The liver
Figure 81-6
conjugates the estrogens to form glucuronides and sul-
Chemical formulas of the principal female hormones.
fates, and about one fifth of these conjugated products
Chapter 81
Female Physiology Before Pregnancy and Female Hormones
1017
is excreted in the bile; most of the remainder is
the glandular tissues of this lining to proliferate;
excreted in the urine. Also, the liver converts the
especially important, they cause the number of ciliated
potent estrogens estradiol and estrone into the almost
epithelial cells that line the fallopian tubes to increase.
totally impotent estrogen estriol. Therefore, dimin-
Also, activity of the cilia is considerably enhanced.
ished liver function actually increases the
These cilia always beat toward the uterus, which helps
activity of estrogens in the body, sometimes causing
propel the fertilized ovum in that direction.
hyperestrinism.
Effect of Estrogens on the Breasts. The primordial breasts
Fate of Progesterone. Within a few minutes after secre-
of females and males are exactly alike. In fact, under
tion, almost all the progesterone is degraded to other
the influence of appropriate hormones, the masculine
steroids that have no progestational effect. As with the
breast during the first 2 decades of life can develop
estrogens, the liver is especially important for this
sufficiently to produce milk in the same manner as the
metabolic degradation.
female breast.
The major end product of progesterone degradation
Estrogens cause (1) development of the stromal
is pregnanediol. About 10 per cent of the original prog-
tissues of the breasts, (2) growth of an extensive ductile
esterone is excreted in the urine in this form. There-
system, and (3) deposition of fat in the breasts. The
fore, one can estimate the rate of progesterone
lobules and alveoli of the breast develop to a slight
formation in the body from the rate of this excretion.
extent under the influence of estrogens alone, but it
is progesterone and prolactin that cause the ultimate
determinative growth and function of these structures.
In summary, the estrogens initiate growth of the
Functions of the Estrogens—
breasts and of the milk-producing apparatus. They
Their Effects on the Primary and
are also responsible for the characteristic growth
Secondary Female Sex Characteristics
and external appearance of the mature female breast.
However, they do not complete the job of converting
A primary function of the estrogens is to cause cellu-
the breasts into milk-producing organs.
lar proliferation and growth of the tissues of the sex
organs and other tissues related to reproduction.
Effect of Estrogens on the Skeleton. Estrogens inhibit
osteoclastic activity in the bones and therefore stimu-
Effect of Estrogens on the Uterus and External Female Sex
late bone growth. At puberty, when the female enters
Organs. During childhood, estrogens are secreted only
her reproductive years, her growth in height becomes
in minute quantities, but at puberty, the quantity
rapid for several years. However, estrogens have
secreted in the female under the influence of the
another potent effect on skeletal growth: They cause
pituitary gonadotropic hormones increases 20-fold or
uniting of the epiphyses with the shafts of the long
more. At this time, the female sex organs change from
bones. This effect of estrogen in the female is much
those of a child to those of an adult. The ovaries, fal-
stronger than the similar effect of testosterone in the
lopian tubes, uterus, and vagina all increase several
male. As a result, growth of the female usually ceases
times in size. Also, the external genitalia enlarge, with
several years earlier than growth of the male. A female
deposition of fat in the mons pubis and labia majora
eunuch who is devoid of estrogen production usually
and enlargement of the labia minora.
grows several inches taller than a normal mature
In addition, estrogens change the vaginal epithelium
female because her epiphyses do not unite at the
from a cuboidal into a stratified type, which is consid-
normal time.
erably more resistant to trauma and infection than
is the prepubertal cuboidal cell epithelium. Vaginal
Osteoporosis of the Bones Caused by Estrogen
infections in children can often be cured by the admin-
Deficiency in Old Age. After menopause, almost no
istration of estrogens simply because of the resulting
estrogens are secreted by the ovaries. This estrogen
increased resistance of the vaginal epithelium.
deficiency leads to (1) increased osteoclastic activity in
During the first few years after puberty, the size of
the bones,
(2) decreased bone matrix, and
(3)
the uterus increases twofold to threefold, but more
decreased deposition of bone calcium and phosphate.
important than the increase in uterus size are the
In some women, this effect is extremely severe, and the
changes that take place in the uterine endometrium
resulting condition is osteoporosis, described in
under the influence of estrogens. Estrogens cause
Chapter 79. Because this can greatly weaken the bones
marked proliferation of the endometrial stroma
and lead to bone fracture, especially fracture of the
and greatly increased development of the endometrial
vertebrae, a large share of postmenopausal women are
glands, which will later aid in providing nutrition to
treated prophylactically with estrogen replacement to
the implanted ovum. These effects are discussed later
prevent the osteoporotic effects.
in the chapter in connection with the endometrial
cycle.
Effect of Estrogens on Protein Deposition. Estrogens cause
a slight increase in total body protein, which is evi-
Effect of Estrogens on the Fallopian Tubes. The estrogens’
denced by a slight positive nitrogen balance when
effect on the mucosal lining of the fallopian tubes is
estrogens are administered. This mainly results from
similar to that on the uterine endometrium. They cause
the growth-promoting effect of estrogen on the sexual
1018
Unit XIV Endocrinology and Reproduction
organs, the bones, and a few other tissues of the body.
necessary for nutrition of the fertilized, dividing ovum
The enhanced protein deposition caused by testos-
as it traverses the fallopian tube before implantation.
terone is much more general and many times as pow-
erful as that caused by estrogens.
Effect of Progesterone on the Breasts. Progesterone
promotes development of the lobules and alveoli of
Effect of Estrogens on Body Metabolism and Fat Deposition.
the breasts, causing the alveolar cells to proliferate,
Estrogens increase the whole-body metabolic rate
enlarge, and become secretory in nature. However,
slightly, but only about one third as much as the
progesterone does not cause the alveoli to secrete
increase caused by the male sex hormone testosterone.
milk; as discussed in Chapter 82, milk is secreted only
They also cause deposition of increased quantities of
after the prepared breast is further stimulated by pro-
fat in the subcutaneous tissues. As a result, the per-
lactin from the anterior pituitary gland.
centage of body fat in the female body is considerably
Progesterone also causes the breasts to swell. Part
greater than that in the male body, which contains
of this swelling is due to the secretory development in
more protein. In addition to deposition of fat in the
the lobules and alveoli, but part also results from
breasts and subcutaneous tissues, estrogens cause the
increased fluid in the subcutaneous tissue.
deposition of fat in the buttocks and thighs, which is
characteristic of the feminine figure.
Monthly Endometrial Cycle
Effect of Estrogens on Hair Distribution. Estrogens do not
and Menstruation
greatly affect hair distribution. However, hair does
develop in the pubic region and in the axillae after
Associated with the monthly cyclical production
puberty. Androgens formed in increased quantities by
of estrogens and progesterone by the ovaries is an
the female adrenal glands after puberty are mainly
endometrial cycle in the lining of the uterus that oper-
responsible for this.
ates through the following stages: (1) proliferation of
the uterine endometrium; (2) development of secre-
Effect of Estrogens on the Skin. Estrogens cause the skin
tory changes in the endometrium; and (3) desqua-
to develop a texture that is soft and usually smooth,
mation of the endometrium, which is known as
but even so, the skin of a woman is thicker than that
menstruation. The various phases of this endometrial
of a child or a castrated female. Also, estrogens cause
cycle are shown in Figure 81-7.
the skin to become more vascular; this is often associ-
ated with increased warmth of the skin and also pro-
Proliferative Phase (Estrogen Phase) of the Endometrial Cycle,
motes greater bleeding of cut surfaces than is observed
Occurring Before Ovulation. At the beginning of each
in men.
monthly cycle, most of the endometrium has been
desquamated by menstruation. After menstruation,
Effect of Estrogens on Electrolyte Balance. The chemical
only a thin layer of endometrial stroma remains, and
similarity of estrogenic hormones to adrenocortical
the only epithelial cells that are left are those located
hormones has been pointed out. Estrogens, like aldos-
in the remaining deeper portions of the glands and
terone and some other adrenocortical hormones, cause
crypts of the endometrium. Under the influence of
sodium and water retention by the kidney tubules. This
estrogens, secreted in increasing quantities by the
effect of estrogens is normally slight and rarely of sig-
ovary during the first part of the monthly ovarian
nificance, but during pregnancy, the tremendous for-
cycle, the stromal cells and the epithelial cells
mation of estrogens by the placenta may contribute to
body fluid retention, as discussed in Chapter 82.
Functions of Progesterone
Effect of Progesterone on the Uterus. By far the most
important function of progesterone is to promote
secretory changes in the uterine endometrium during
the latter half of the monthly female sexual cycle, thus
preparing the uterus for implantation of the fertilized
ovum. This function is discussed later in connection
with the endometrial cycle of the uterus.
Proliferative
Secretory
Menstrual
In addition to this effect on the endometrium,
phase
phase
phase
progesterone decreases the frequency and intensity
(11 days)
(12 days)
(5 days)
of uterine contractions, thereby helping to prevent
expulsion of the implanted ovum.
Figure 81-7
Effect of Progesterone on the Fallopian Tubes. Progesterone
also promotes increased secretion by the mucosal
Phases of endometrial growth and menstruation during each
lining of the fallopian tubes. These secretions are
monthly female sexual cycle.
Chapter 81
Female Physiology Before Pregnancy and Female Hormones
1019
proliferate rapidly. The endometrial surface is re-
decreased stimulation of the endometrial cells by these
epithelialized within 4 to 7 days after the beginning of
two hormones, followed rapidly by involution of the
menstruation.
endometrium itself to about 65 per cent of its previous
Then, during the next week and a half—that is,
thickness. Then, during the 24 hours preceding the
before ovulation occurs—the endometrium increases
onset of menstruation, the tortuous blood vessels
greatly in thickness, owing to increasing numbers of
leading to the mucosal layers of the endometrium
stromal cells and to progressive growth of the endome-
become vasospastic, presumably because of some
trial glands and new blood vessels into the endo-
effect of involution, such as release of a vasoconstric-
metrium. At the time of ovulation, the endometrium is
tor material—possibly one of the vasoconstrictor types
3 to 5 millimeters thick.
of prostaglandins that are present in abundance at this
The endometrial glands, especially those of the cer-
time.
vical region, secrete a thin, stringy mucus. The mucus
The vasospasm, the decrease in nutrients to the
strings actually align themselves along the length of
endometrium, and the loss of hormonal stimulation
the cervical canal, forming channels that help guide
initiate necrosis in the endometrium, especially of the
sperm in the proper direction from the vagina into the
blood vessels. As a result, blood at first seeps into the
uterus.
vascular layer of the endometrium, and the hemor-
rhagic areas grow rapidly over a period of 24 to 36
Secretory Phase (Progestational Phase) of the Endometrial
hours. Gradually, the necrotic outer layers of the
Cycle, Occurring After Ovulation. During most of the latter
endometrium separate from the uterus at the sites
half of the monthly cycle, after ovulation has occurred,
of the hemorrhages until, about 48 hours after the
progesterone and estrogen together are secreted in
onset of menstruation, all the superficial layers of
large quantities by the corpus luteum. The estrogens
the endometrium have desquamated. The mass of
cause slight additional cellular proliferation in the
desquamated tissue and blood in the uterine cavity,
endometrium during this phase of the cycle, whereas
plus contractile effects of prostaglandins or other
progesterone causes marked swelling and secretory
substances in the decaying desquamate, all acting
development of the endometrium. The glands increase
together, initiate uterine contractions that expel the
in tortuosity; an excess of secretory substances accu-
uterine contents.
mulates in the glandular epithelial cells. Also, the cyto-
During normal menstruation, approximately
40
plasm of the stromal cells increases; lipid and glycogen
milliliters of blood and an additional 35 milliliters of
deposits increase greatly in the stromal cells; and the
serous fluid are lost. The menstrual fluid is normally
blood supply to the endometrium further increases
nonclotting because a fibrinolysin is released along
in proportion to the developing secretory activity,
with the necrotic endometrial material. If excessive
with the blood vessels becoming highly tortuous. At
bleeding occurs from the uterine surface, the quantity
the peak of the secretory phase, about 1 week after
of fibrinolysin may not be sufficient to prevent clot-
ovulation, the endometrium has a thickness of 5 to
ting. The presence of clots during menstruation is often
6 millimeters.
clinical evidence of uterine pathology.
The whole purpose of all these endometrial changes
Within 4 to 7 days after menstruation starts, the loss
is to produce a highly secretory endometrium that con-
of blood ceases because, by this time, the endometrium
tains large amounts of stored nutrients to provide
has become re-epithelialized.
appropriate conditions for implantation of a fertilized
ovum during the latter half of the monthly cycle. From
Leukorrhea During Menstruation. During menstrua-
the time a fertilized ovum enters the uterine cavity
tion, tremendous numbers of leukocytes are released
from the fallopian tube (which occurs 3 to 4 days
along with the necrotic material and blood. It is prob-
after ovulation) until the time the ovum implants (7
able that some substance liberated by the endometrial
to
9 days after ovulation), the uterine secretions,
necrosis causes this outflow of leukocytes. As a result
called “uterine milk,” provide nutrition for the early
of these leukocytes and possibly other factors, the
dividing ovum. Then, once the ovum implants in the
uterus is highly resistant to infection during men-
endometrium, the trophoblastic cells on the surface of
struation, even though the endometrial surfaces are
the implanting ovum (in the blastocyst stage) begin to
denuded. This is of extreme protective value.
digest the endometrium and absorb the endometrial
stored substances, thus making great quantities of
nutrients available to the early implanting embryo.
Regulation of the Female
Monthly Rhythm—Interplay
Menstruation. If the ovum is not fertilized, about 2 days
Between the Ovarian and
before the end of the monthly cycle, the corpus luteum
Hypothalamic-Pituitary
in the ovary suddenly involutes, and the ovarian
hormones (estrogens and progesterone) decrease to
Hormones
low levels of secretion, as shown in Figure
81-3.
Menstruation follows.
Now that we have presented the major cyclical
Menstruation is caused by the reduction of estro-
changes that occur during the monthly female sexual
gens and progesterone, especially progesterone, at the
cycle, we can attempt to explain the basic rhythmical
end of the monthly ovarian cycle. The first effect is
mechanism that causes the cyclical variations.
1020
Unit XIV Endocrinology and Reproduction
The Hypothalamus Secretes GnRH, Which
believed that these arcuate nuclei control most
Causes the Anterior Pituitary Gland to Secrete
female sexual activity, although neurons located in the
LH and FSH
preoptic area of the anterior hypothalamus also
As pointed out in Chapter 74, secretion of most of the
secrete GnRH in moderate amounts. Multiple neu-
anterior pituitary hormones is controlled by “releasing
ronal centers in the higher brain’s “limbic” system (the
hormones” formed in the hypothalamus and then
system for psychic control) transmit signals into the
transported to the anterior pituitary gland by way of
arcuate nuclei to modify both the intensity of GnRH
the hypothalamic-hypophysial portal system. In the
release and the frequency of the pulses, thus providing
case of the gonadotropins, one releasing hormone,
a partial explanation of why psychic factors often
GnRH, is important. This hormone has been purified
modify female sexual function.
and has been found to be a decapeptide with the
following formula:
Negative Feedback Effects of Estrogen
and Progesterone in Decreasing Both LH
Glu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2
and FSH Secretion
Estrogen in small amounts has a strong effect to
Intermittent, Pulsatile Secretion of GnRH by the Hypothalamus
inhibit the production of both LH and FSH. Also,
Stimulates Pulsatile Release of LH from the Anterior Pituitary
when progesterone is available, the inhibitory effect of
Gland. Experiments have demonstrated that the hypo-
estrogen is multiplied, even though progesterone by
thalamus does not secrete GnRH continuously but
itself has little effect.
instead secretes it in pulses lasting 5 to 25 minutes that
These feedback effects seem to operate mainly on
occur every 1 to 2 hours. The lower curve in Figure
the anterior pituitary gland directly, but they also
81-8 shows the electrical pulsatile signals in the hypo-
operate to a lesser extent on the hypothalamus to
thalamus that cause the hypothalamic pulsatile output
decrease secretion of GnRH, especially by altering the
of GnRH.
frequency of the GnRH pulses.
It is intriguing that when GnRH is infused continu-
ously so that it is available all the time rather than in
Hormone Inhibin from the Corpus Luteum Inhibits FSH and LH
pulses, its ability to cause the release of LH and FSH
Secretion. In addition to the feedback effects of estro-
by the anterior pituitary gland is lost. Therefore, for
gen and progesterone, other hormones seem to be
reasons unknown, the pulsatile nature of GnRH
involved, especially inhibin, which is secreted along
release is essential to its function.
with the steroid sex hormones by the granulosa cells
The pulsatile release of GnRH also causes intermit-
of the ovarian corpus luteum in the same way that
tent output of LH secretion about every 90 minutes.
Sertoli cells secrete inhibin in the male testes. This
This is shown by the upper curve in Figure 81-8.
hormone has the same effect in the female as in the
male—inhibiting the secretion of FSH and, to a lesser
Hypothalamic Centers for Release of GnRH. The neuronal
extent, LH by the anterior pituitary gland. Therefore,
activity that causes pulsatile release of GnRH occurs
it is believed that inhibin might be especially impor-
primarily in the mediobasal hypothalamus, especially
tant in causing the decrease in secretion of FSH and
in the arcuate nuclei of this area. Therefore, it is
LH at the end of the monthly female sexual cycle.
100
LH
80
2000
60
Figure 81-8
40
Upper curve: Pulsatile change in
luteinizing hormone
(LH) in the
0
peripheral circulation of a
1000
pentobarbital-anesthetized
ovariectomized rhesus monkey.
Lower curve: Minute-by-minute
recording of multi-unit electrical
MUA
activity (MUA) in the mediobasal
hypothalamus. (Data from Wilson
RC, Kesner JS, Kaufman JM, et
0
120
240
360
480
al:
Central electrophysiologic
Minutes
correlates of pulsatile luteinizing
hormone secretion. Neuroen-
docrinology 39:256, 1984.)
Chapter 81
Female Physiology Before Pregnancy and Female Hormones
1021
Positive Feedback Effect of Estrogen Before
2. Follicular Growth Phase. Two to 3 days before men-
Ovulation—The Preovulatory LH Surge
struation, the corpus luteum has regressed to almost
For reasons not completely understood, the anterior
total involution, and the secretion of estrogen, proges-
pituitary gland secretes greatly increased amounts of
terone, and inhibin from the corpus luteum decreases
LH for 1 to 2 days beginning 24 to 48 hours before
to a low ebb. This releases the hypothalamus and
ovulation. This effect is demonstrated in Figure 81-3.
anterior pituitary from the negative feedback effect of
The figure shows a much smaller preovulatory surge
these hormones. Therefore, a day or so later, at about
of FSH as well.
the time that menstruation begins, pituitary secretion
Experiments have shown that infusion of estrogen
of FSH begins to increase again, as much as twofold;
into a female above a critical rate for 2 to 3 days during
then, several days after menstruation begins, LH secre-
the latter part of the first half of the ovarian cycle will
tion increases slightly as well. These hormones initiate
cause rapidly accelerating growth of the ovarian folli-
new ovarian follicle growth and a progressive increase
cles, as well as rapidly accelerating secretion of ovarian
in the secretion of estrogen, reaching a peak estrogen
estrogens. During this period, secretions of both FSH
secretion at about 12.5 to 13 days after the onset of the
and LH by the anterior pituitary gland are at first
new female monthly sexual cycle.
slightly suppressed. Then secretion of LH increases
During the first 11 to 12 days of this follicle growth,
abruptly sixfold to eightfold, and secretion of FSH
the rates of pituitary secretion of the gonadotropins
increases about twofold. The greatly increased secre-
FSH and LH decrease slightly because of the negative
tion of LH causes ovulation to occur.
feedback effect, mainly of estrogen, on the anterior
The cause of this abrupt surge in LH secretion is not
pituitary gland. Then there is a sudden, marked
known. However, several possible explanations are as
increase in the secretion of LH and, to a lesser extent,
follows: (1) It has been suggested that estrogen at this
FSH. This is the preovulatory surge of LH and FSH,
point in the cycle has a peculiar positive feedback effect
which is followed by ovulation.
of stimulating pituitary secretion of LH and, to a lesser
extent, FSH; this is in sharp contrast to its normal neg-
3. Preovulatory Surge of LH and FSH Causes Ovulation. At
ative feedback effect that occurs during the remainder
about 11.5 to 12 days after the onset of the monthly
of the female monthly cycle. (2) The granulosa cells of
cycle, the decline in secretion of FSH and LH comes
the follicles begin to secrete small but increasing quan-
to an abrupt halt. It is believed that the high level of
tities of progesterone a day or so before the preovu-
estrogens at this time (or the beginning of proges-
latory LH surge, and it has been suggested that this
terone secretion by the follicles) causes a positive
might be the factor that stimulates the excess LH
feedback stimulatory effect on the anterior pituitary,
secretion.
as explained earlier, which leads to a terrific surge in
Without this normal preovulatory surge of LH,
the secretion of LH and, to a lesser extent, FSH. What-
ovulation will not occur.
ever the cause of this preovulatory LH and FSH surge,
the great excess of LH leads to both ovulation and sub-
sequent development of and secretion by the corpus
luteum. Thus, the hormonal system begins its new
Feedback Oscillation of the
round of secretions until the next ovulation.
Hypothalamic-Pituitary-Ovarian
System
Anovulatory Cycles—Sexual Cycles at Puberty
If the preovulatory surge of LH is not of sufficient
Now, after discussing much of the known information
magnitude, ovulation will not occur, and the cycle is
about the interrelations of the different components
said to be “anovulatory.” The phases of the sexual cycle
of the female hormonal system, we can attempt to
continue, but they are altered in the following ways:
explain the feedback oscillation that controls the
First, lack of ovulation causes failure of development
rhythm of the female sexual cycle. It seems to operate
of the corpus luteum, so there is almost no secretion
in approximately the following sequence of three
of progesterone during the latter portion of the cycle.
events.
Second, the cycle is shortened by several days, but the
rhythm continues. Therefore, it is likely that proges-
1. Postovulatory Secretion of the Ovarian Hormones, and
terone is not required for maintenance of the cycle
Depression of the Pituitary Gonadotropins. The easiest part
itself, although it can alter its rhythm.
of the cycle to explain is the events that occur during
The first few cycles after the onset of puberty are
the postovulatory phase—between ovulation and
usually anovulatory, as are the cycles occurring several
the beginning of menstruation. During this time, the
months to years before menopause, presumably
corpus luteum secretes large quantities of both prog-
because the LH surge is not potent enough at these
esterone and estrogen, as well as the hormone inhibin.
times to cause ovulation.
All these hormones together have a combined nega-
tive feedback effect on the anterior pituitary gland and
hypothalamus, causing the suppression of both FSH
Puberty and Menarche
and LH secretion and decreasing them to their lowest
levels about 3 to 4 days before the onset of menstrua-
Puberty means the onset of adult sexual life, and
tion. These effects are shown in Figure 81-3.
menarche means the beginning of the cycle of
1022
Unit XIV Endocrinology and Reproduction
60
400
50
300
40
Female
300
30
100
20
Male
10
0
0
12
13
40
50
60
0
Age (yr)
0
10
20
30
40
50
60
70
80
Age (yr)
Figure 81-10
Figure 81-9
Estrogen secretion throughout the sexual life of the female human
being.
Total rates of secretion of gonadotropic hormones throughout the
sexual lives of female and male human beings, showing an espe-
cially abrupt increase in gonadotropic hormones at menopause in
the female.
months to a few years, the cycle ceases altogether, as
shown in Figure 81-10. The period during which the
cycle ceases and the female sex hormones diminish to
almost none is called menopause.
menstruation. The period of puberty is caused by a
The cause of menopause is “burning out” of the
gradual increase in gonadotropic hormone secretion
ovaries. Throughout a woman’s reproductive life,
by the pituitary, beginning in about the eighth year of
about 400 of the primordial follicles grow into mature
life, as shown in Figure 81-9, and usually culminating
follicles and ovulate, and hundreds of thousands of ova
in the onset of puberty and menstruation between ages
degenerate. At about age 45 years, only a few primor-
11 and 16 years in girls (average, 13 years).
dial follicles remain to be stimulated by FSH and LH,
In the female, as in the male, the infantile pituitary
and, as shown in Figure 81-10, the production of
gland and ovaries are capable of full function if appro-
estrogens by the ovaries decreases as the number of
priately stimulated. However, as is also true in the
primordial follicles approaches zero. When estrogen
male, and for reasons not understood, the hypothala-
production falls below a critical value, the estrogens
mus does not secrete significant quantities of GnRH
can no longer inhibit the production of the
during childhood. Experiments have shown that
gonadotropins FSH and LH. Instead, as shown in
the hypothalamus itself is capable of secreting this
Figure 81-9, the gonadotropins FSH and LH (mainly
hormone, but the appropriate signal from some other
FSH) are produced after menopause in large and con-
area of brain to cause the secretion is lacking. There-
tinuous quantities, but as the remaining primordial fol-
fore, it is now believed that the onset of puberty is
licles become atretic, the production of estrogens by
initiated by some maturation process that occurs else-
the ovaries falls virtually to zero.
where in the brain, perhaps somewhere in the limbic
At the time of menopause, a woman must readjust
system.
her life from one that has been physiologically stimu-
Figure 81-10 shows (1) the increasing levels of estro-
lated by estrogen and progesterone production to one
gen secretion at puberty, (2) the cyclical variation
devoid of these hormones. The loss of estrogens often
during the monthly sexual cycle,
(3) the further
causes marked physiological changes in the function
increase in estrogen secretion during the first few years
of the body, including (1) “hot flushes” characterized
of reproductive life, (4) the progressive decrease in
by extreme flushing of the skin, (2) psychic sensations
estrogen secretion toward the end of reproductive life,
of dyspnea, (3) irritability, (4) fatigue, (5) anxiety,
and, finally, (5) almost no estrogen or progesterone
(6) occasionally various psychotic states, and
(7)
secretion beyond menopause.
decreased strength and calcification of bones through-
out the body. These symptoms are of sufficient magni-
tude in about
15 per cent of women to warrant
Menopause
treatment. If counseling fails, daily administration of
estrogen in small quantities usually reverses the symp-
At age 40 to 50 years, the sexual cycle usually becomes
toms, and by gradually decreasing the dose, post-
irregular, and ovulation often fails to occur. After a few
menopausal women can likely avoid severe symptoms.
Chapter 81
Female Physiology Before Pregnancy and Female Hormones
1023
Abnormalities of Secretion
Thinking sexual thoughts can lead to female sexual
desire, and this aids greatly in the performance of the
by the Ovaries
female sexual act. Such desire is based largely on a
Hypogonadism. Less than normal secretion by the ovaries
woman’s background training as well as on her physi-
can result from poorly formed ovaries, lack of ovaries,
ological drive, although sexual desire does increase in
or genetically abnormal ovaries that secrete the wrong
proportion to the level of sex hormones secreted.
hormones because of missing enzymes in the secretory
Desire also changes during the monthly sexual cycle,
cells. When ovaries are absent from birth or when
reaching a peak near the time of ovulation, probably
they become nonfunctional before puberty, female
because of the high levels of estrogen secretion during
eunuchism occurs. In this condition, the usual secondary
sexual characteristics do not appear, and the sexual
the preovulatory period.
organs remain infantile. Especially characteristic of
Local sexual stimulation in women occurs in more
this condition is prolonged growth of the long bones
or less the same manner as in men because massage
because the epiphyses do not unite with the shafts as
and other types of stimulation of the vulva, vagina, and
early as they do in a normal woman. Consequently, the
other perineal regions can create sexual sensations.
female eunuch is essentially as tall as or perhaps even
The glans of the clitoris is especially sensitive for ini-
slightly taller than her male counterpart of similar
tiating sexual sensations.
genetic background.
As in the male, the sexual sensory signals are trans-
When the ovaries of a fully developed woman are
mitted to the sacral segments of the spinal cord
removed, the sexual organs regress to some extent so
through the pudendal nerve and sacral plexus. Once
that the uterus becomes almost infantile in size, the
vagina becomes smaller, and the vaginal epithelium
these signals have entered the spinal cord, they are
becomes thin and easily damaged. The breasts atrophy
transmitted to the cerebrum. Also, local reflexes inte-
and become pendulous, and the pubic hair becomes
grated in the sacral and lumbar spinal cord are at least
thinner. The same changes occur in women after
partly responsible for some of the reactions in the
menopause.
female sexual organs.
Irregularity of Menses, and Amenorrhea Caused by
Female Erection and Lubrication. Located around the
Hypogonadism. As pointed out in the preceding dis-
introitus and extending into the clitoris is erectile
cussion of menopause, the quantity of estrogens pro-
duced by the ovaries must rise above a critical value in
tissue almost identical to the erectile tissue of the
order to cause rhythmical sexual cycles. Consequently,
penis. This erectile tissue, like that of the penis, is
in hypogonadism or when the gonads are secreting
controlled by the parasympathetic nerves that pass
small quantities of estrogens as a result of other factors,
through the nervi erigentes from the sacral plexus to
such as hypothyroidism, the ovarian cycle often does
the external genitalia. In the early phases of sexual
not occur normally. Instead, several months may elapse
stimulation, parasympathetic signals dilate the arteries
between menstrual periods, or menstruation may cease
of the erectile tissue, probably resulting from release
altogether (amenorrhea). Prolonged ovarian cycles are
of acetylcholine, nitric oxide, and vasoactive intestinal
frequently associated with failure of ovulation, presum-
polypeptide (VIP) at the nerve endings. This allows
ably because of insufficient secretion of LH at the time
rapid accumulation of blood in the erectile tissue so
of the preovulatory surge of LH, which is necessary for
ovulation.
that the introitus tightens around the penis; this aids
the male greatly in his attainment of sufficient sexual
Hypersecretion by the Ovaries. Extreme hypersecretion of
stimulation for ejaculation to occur.
ovarian hormones by the ovaries is a rare clinical entity,
Parasympathetic signals also pass to the bilateral
because excessive secretion of estrogens automatically
Bartholin’s glands located beneath the labia minora
decreases the production of gonadotropins by the
and cause them to secrete mucus immediately inside
pituitary, and this limits the production of ovarian
the introitus. This mucus is responsible for much of the
hormones. Consequently, hypersecretion of feminizing
lubrication during sexual intercourse, although much
hormones is usually recognized clinically only when a
is also provided by mucus secreted by the vaginal
feminizing tumor develops.
epithelium and a small amount from the male urethral
A rare granulosa cell tumor can develop in an ovary,
occurring more often after menopause than before.
glands. This lubrication is necessary during intercourse
These tumors secrete large quantities of estrogens,
to establish a satisfactory massaging sensation rather
which exert the usual estrogenic effects, including
than an irritative sensation, which may be provoked
hypertrophy of the uterine endometrium and irregular
by a dry vagina. A massaging sensation constitutes
bleeding from this endometrium. In fact, bleeding is
the optimal stimulus for evoking the appropriate
often the first and only indication that such a tumor
reflexes that culminate in both the male and female
exists.
climaxes.
Female Orgasm. When local sexual stimulation reaches
Female Sexual Act
maximum intensity, and especially when the local
sensations are supported by appropriate psychic
Stimulation of the Female Sexual Act. As is true in the male
conditioning signals from the cerebrum, reflexes are
sexual act, successful performance of the female sexual
initiated that cause the female orgasm, also called
act depends on both psychic stimulation and local
the female climax. The female orgasm is analogous to
sexual stimulation.
emission and ejaculation in the male, and it may help
1024
Unit XIV Endocrinology and Reproduction
promote fertilization of the ovum. Indeed, the human
the 26th day of the cycle. Finally, if the periodicity of the
female is known to be somewhat more fertile when
cycle is 21 days, ovulation usually occurs within 1 day of
inseminated by normal sexual intercourse rather than
the 7th day of the cycle. Therefore, it is usually stated
that avoidance of intercourse for 4 days before the cal-
by artificial methods, thus indicating an important
culated day of ovulation and 3 days afterward prevents
function of the female orgasm. Possible reasons for
conception. But such a method of contraception can be
this are as follows.
used only when the periodicity of the menstrual cycle is
First, during the orgasm, the perineal muscles of the
regular.
female contract rhythmically, which results from spinal
cord reflexes similar to those that cause ejaculation in
Hormonal Suppression of Fertility—“The Pill.” It has long
the male. It is possible that these reflexes increase
been known that administration of either estrogen or
uterine and fallopian tube motility during the orgasm,
progesterone, if given in appropriate quantities during
thus helping to transport the sperm upward through
the first half of the monthly cycle, can inhibit ovulation.
the uterus toward the ovum; information on this
The reason for this is that appropriate administration of
either of these hormones can prevent the preovulatory
subject is scanty, however. Also, the orgasm seems to
surge of LH secretion by the pituitary gland, which is
cause dilation of the cervical canal for up to
30
essential in causing ovulation.
minutes, thus allowing easy transport of the sperm.
Why the administration of estrogen or progesterone
Second, in many lower animals, copulation causes
prevents the preovulatory surge of LH secretion is not
the posterior pituitary gland to secrete oxytocin; this
fully understood. However, experimental work has sug-
effect is probably mediated through the brain amyg-
gested that immediately before the surge occurs, there
daloid nuclei and then through the hypothalamus to
is probably a sudden depression of estrogen secretion
the pituitary. The oxytocin causes increased rhythmi-
by the ovarian follicles, and this might be the necessary
cal contractions of the uterus, which have been postu-
signal that causes the subsequent feedback effect on the
lated to cause increased transport of the sperm. A few
anterior pituitary that leads to the LH surge. The admin-
istration of sex hormones (estrogens or progesterone)
sperm have been shown to traverse the entire length
could prevent the initial ovarian hormonal depression
of the fallopian tube in the cow in about 5 minutes, a
that might be the initiating signal for ovulation.
rate at least 10 times as fast as that which the swim-
The problem in devising methods for the hormonal
ming motions of the sperm themselves could possibly
suppression of ovulation has been in developing appro-
achieve. Whether this occurs in the human female is
priate combinations of estrogens and progestins that
unknown.
suppress ovulation but do not cause other, unwanted
In addition to the possible effects of the orgasm on
effects. For instance, too much of either hormone can
fertilization, the intense sexual sensations that develop
cause abnormal menstrual bleeding patterns. However,
during the orgasm also pass to the cerebrum and cause
use of certain synthetic progestins in place of proges-
intense muscle tension throughout the body. But after
terone, especially the 19-norsteroids, along with small
amounts of estrogens usually prevents ovulation yet
culmination of the sexual act, this gives way during the
allows an almost normal pattern of menstruation.
succeeding minutes to a sense of satisfaction charac-
Therefore, almost all “pills” used for the control of fer-
terized by relaxed peacefulness, an effect called
tility consist of some combination of synthetic estrogens
resolution.
and synthetic progestins. The main reason for using syn-
thetic estrogens and progestins is that the natural hor-
mones are almost entirely destroyed by the liver within
Female Fertility
a short time after they are absorbed from the gastroin-
testinal tract into the portal circulation. However,
Fertile Period of Each Sexual Cycle. The ovum remains
many of the synthetic hormones can resist this
viable and capable of being fertilized after it is expelled
destructive propensity of the liver, thus allowing oral
from the ovary probably no longer than 24 hours. There-
administration.
fore, sperm must be available soon after ovulation if fer-
Two of the most commonly used synthetic estrogens
tilization is to take place. A few sperm can remain fertile
are ethinyl estradiol and mestranol. Among the most
in the female reproductive tract for up to 5 days. There-
commonly used progestins are norethindrone, norethyn-
fore, for fertilization to take place, intercourse must
odrel, ethynodiol, and norgestrel. The drug is usually
occur sometime between 4 and 5 days before ovulation
begun in the early stages of the monthly cycle and con-
up to a few hours after ovulation. Thus, the period of
tinued beyond the time that ovulation would normally
female fertility during each month is short, about 4 to 5
occur. Then the drug is stopped, allowing menstruation
days.
to occur and a new cycle to begin.
Rhythm Method of Contraception. One of the commonly
Abnormal Conditions That Cause Female Sterility. About 5 to
practiced methods of contraception is to avoid inter-
10 per cent of women are infertile. Occasionally, no
course near the time of ovulation. The difficulty with
abnormality can be discovered in the female genital
this method of contraception is predicting the exact
organs, in which case it must be assumed that the infer-
time of ovulation. Yet the interval from ovulation until
tility is due to either abnormal physiological function of
the next succeeding onset of menstruation is almost
the genital system or abnormal genetic development of
always between 13 and 15 days. Therefore, if the men-
the ova themselves.
strual cycle is regular, with an exact periodicity of 28
Probably by far the most common cause of female
days, ovulation usually occurs within 1 day of the 14th
sterility is failure to ovulate. This can result from
day of the cycle. If, in contrast, the periodicity of the
hyposecretion of gonadotropic hormones, in which
cycle is 40 days, ovulation usually occurs within 1 day of
case the intensity of the hormonal stimuli is simply
Chapter 81
Female Physiology Before Pregnancy and Female Hormones
1025
ovaries. Endometriosis causes fibrosis throughout the
pelvis, and this fibrosis sometimes so enshrouds
99°
the ovaries that an ovum cannot be released into the
Ovulation
abdominal cavity. Often, endometriosis occludes the fal-
lopian tubes, either at the fimbriated ends or elsewhere
along their extent.
98°
Another common cause of female infertility is salp-
ingitis, that is, inflammation of the fallopian tubes; this
causes fibrosis in the tubes, thereby occluding them. In
the past, such inflammation occurred mainly as a result
of gonococcal infection, but with modern therapy,
97°
this is becoming a less prevalent cause of female
infertility.
Still another cause of infertility is secretion of abnor-
mal mucus by the uterine cervix. Ordinarily, at the time
0
of ovulation, the hormonal environment of estrogen
0
2
4
6
8 10 12 14 16 18 20 22 24 26 28
Day of cycle
causes the secretion of mucus with special charac-
teristics that allow rapid mobility of sperm into the
uterus and actually guide the sperm up along mucous
“threads.” Abnormalities of the cervix itself, such as
Figure 81-11
low-grade infection or inflammation, or abnormal hor-
monal stimulation of the cervix can lead to a viscous
Elevation in body temperature shortly after ovulation.
mucous plug that prevents fertilization.
References
insufficient to cause ovulation, or it can result from
abnormal ovaries that do not allow ovulation. For
Altkorn D, Vokes T: Treatment of postmenopausal osteo-
instance, thick ovarian capsules occasionally exist on the
porosis. JAMA 285:1415, 2001.
outsides of the ovaries, making ovulation difficult.
Bastian LA, Smith CM, Nanda K: Is this woman peri-
Because of the high incidence of anovulation in
menopausal? JAMA 289:895, 2003.
sterile women, special methods are often used to
Beral V, Banks E, Reeves G: Evidence from randomised
determine whether ovulation occurs. These methods are
trials on the long-term effects of hormone replacement
based mainly on the effects of progesterone on the
therapy. Lancet 360:942, 2002.
body, because the normal increase in progesterone
Compston JE: Sex steroids and bone. Physiol Rev 81:419,
secretion usually does not occur during the latter half
2001.
of anovulatory cycles. In the absence of progestational
Gruber CJ, Tschugguel W, Schneeberger C, Huber JC: Pro-
effects, the cycle can be assumed to be anovulatory.
duction and actions of estrogens. N Engl J Med 346:340,
One of these tests is simply to analyze the urine for a
2002.
surge in pregnanediol, the end product of progesterone
Hamilton-Fairley D, Taylor A: Anovulation. BMJ 327:546,
metabolism, during the latter half of the sexual cycle;
2003.
the lack of this substance indicates failure of ovulation.
Lorenzo J: A new hypothesis for how sex steroid hormones
Another common test is for the woman to chart her
regulate bone mass. J Clin Invest 111:1641, 2003.
body temperature throughout the cycle. Secretion of
Manson JE, Martin KA: Postmenopausal hormone-
progesterone during the latter half of the cycle raises
replacement therapy. N Engl J Med 345:34, 2001.
the body temperature about 0.5°F, with the temperature
Nadal A, Diaz M, Valverde MA: The estrogen trinity:
rise coming abruptly at the time of ovulation. Such a
membrane, cytosolic, and nuclear effects. News Physiol Sci
temperature chart, showing the point of ovulation, is
16:251, 2001.
illustrated in Figure 81-11.
Nelson HD: Commonly used types of postmenopausal estro-
Lack of ovulation caused by hyposecretion of the
gen for treatment of hot flashes: scientific review. JAMA
pituitary gonadotropic hormones can sometimes be
291:1610, 2004.
treated by appropriately timed administration of hu-
Nilsson S, Makela S, Treuter E, et al: Mechanisms of estro-
man chorionic gonadotropin, a hormone (discussed in
gen action. Physiol Rev 81:1535, 2001.
Chapter 82) that is extracted from the human placenta.
Niswender GD, Juengel JL, Silva PJ, et al: Mechanisms con-
This hormone, although secreted by the placenta, has
trolling the function and life span of the corpus luteum.
almost the same effects as LH and is therefore a pow-
Physiol Rev 80:1, 2000.
erful stimulator of ovulation. However, excess use of
Petitti DB: Combination estrogen-progestin oral contracep-
this hormone can cause ovulation from many follicles
tives. N Engl J Med 349:1443, 2003.
simultaneously; this results in multiple births, an effect
Pettersson K, Gustafsson JA: Role of estrogen receptor beta
that has caused as many as eight babies
(mostly
in estrogen action. Annu Rev Physiol 63:165, 2001.
stillborn) to be born to mothers treated for infertility
Riggs BL: The mechanisms of estrogen regulation of bone
with this hormone.
resorption. J Clin Invest 106:1203, 2000.
One of the most common causes of female sterility is
Riggs BL, Hartmann LC: Selective estrogen-receptor mod-
endometriosis, a common condition in which endome-
ulators—mechanisms of action and application to clinical
trial tissue almost identical to that of the normal uterine
practice. N Engl J Med 348:618, 2003.
endometrium grows and even menstruates in the pelvic
Smith S, Pfeifer SM, Collins JA: Diagnosis and management
cavity surrounding the uterus, fallopian tubes, and
of female infertility. JAMA 290:1767, 2003.
1026
Unit XIV Endocrinology and Reproduction
Terasawa E, Fernandez DL: Neurobiological mechanisms
van der Eerden BC, Karperien M, Wit JM: Systemic and
of the onset of puberty in primates. Endocr Rev 22:111,
local regulation of the growth plate. Endocr Rev 24:782,
2001.
2003.
Toran-Allerand CD: A plethora of estrogen receptors in
Vasudevan N, Ogawa S, Pfaff D: Estrogen and thyroid
the brain: where will it end? Endocrinology 145:1069,
hormone receptor interactions: physiological flexibility by
2004.
molecular specificity. Physiol Rev 82:923, 2002.
C H A P T E R
8
2
Pregnancy and Lactation
In Chapters 80 and 81, the sexual functions of the
male and female are described to the point of
fertilization of the ovum. If the ovum becomes
fertilized, a new sequence of events called gestation,
or pregnancy, takes place, and the fertilized ovum
eventually develops into a full-term fetus. The
purpose of this chapter is to discuss the early stages
of ovum development after fertilization and then to
discuss the physiology of pregnancy. In Chapter 83, some special aspects of fetal
and early childhood physiology are discussed.
Maturation and Fertilization of the Ovum
While still in the ovary, the ovum is in the primary oocyte stage. Shortly before
it is released from the ovarian follicle, its nucleus divides by meiosis and a first
polar body is expelled from the nucleus of the oocyte. The primary oocyte then
becomes the secondary oocyte. In this process, each of the 23 pairs of chromo-
somes loses one of its partners, which becomes incorporated in a polar body
that is expelled. This leaves 23 unpaired chromosomes in the secondary oocyte.
It is at this time that the ovum, still in the secondary oocyte stage, is ovulated
into the abdominal cavity. Then, almost immediately, it enters the fimbriated
end of one of the fallopian tubes.
Entry of the Ovum into the Fallopian Tube (Oviduct). When ovulation occurs, the ovum,
along with a hundred or more attached granulosa cells that constitute the
corona radiata, is expelled directly into the peritoneal cavity and must then
enter one of the fallopian tubes to reach the cavity of the uterus. The fimbri-
ated ends of each fallopian tube fall naturally around the ovaries. The inner sur-
faces of the fimbriated tentacles are lined with ciliated epithelium, and the cilia
are activated by estrogen from the ovaries, which causes the cilia to beat toward
the opening, or ostium, of the involved fallopian tube. One can actually see a
slow fluid current flowing toward the ostium. By this means, the ovum enters
one of the fallopian tubes.
It seems likely that many ova might fail to enter the fallopian tubes. However,
on the basis of conception studies, it is probable that as many as 98 per cent
succeed in this task. Indeed, in some recorded cases, women with one ovary
removed and the opposite fallopian tube removed have had several children
with relative ease of conception, thus demonstrating that ova can even enter
the opposite fallopian tube.
Fertilization of the Ovum. After the male ejaculates semen into the vagina during
intercourse, a few sperm are transported within 5 to 10 minutes upward from
the vagina and through the uterus and fallopian tubes to the ampullae of the
fallopian tubes near the ovarian ends of the tubes. This transport of the sperm
is aided by contractions of the uterus and fallopian tubes stimulated by
prostaglandins in the male seminal fluid and also by oxytocin released from the
posterior pituitary gland of the female during her orgasm. Of the almost half a
billion sperm deposited in the vagina, a few thousand succeed in reaching each
ampulla.
Fertilization of the ovum normally takes place in the ampulla of one of the
fallopian tubes soon after both the sperm and the ovum enter the ampulla. But
1027
1028
Unit XIV Endocrinology and Reproduction
Dispersed corona radiata
Fertilization
Corona
Cell division
Blastocyst
(day 1)
radiata
Zygote
Fallopian
tube
Sperm
A
B
Blastocyst
reaches
Male
uterus
Sperm
pronucleus
(days 4-5)
Ovary
Blastocyst
Ovulation
Ovum
implants
(days 5-7)
A
Female
Centrosome
Amniotic
pronucleus
C
D
E
cavity
Uterus
Figure 82-1
Fertilization of the ovum. A, The mature ovum surrounded by the
corona radiata. B, Dispersal of the corona radiata. C, Entry of the
sperm. D, Formation of the male and female pronuclei. E, Reor-
ganization of a full complement of chromosomes and beginning
division of the ovum. (Modified from Arey LB: Developmental
Trophoblastic
Anatomy: A Textbook and Laboratory Manual of Embryology, 7th
cells invading
ed. Philadelphia: WB Saunders, 1974.)
endometrium
B
Figure 82-2
before a sperm can enter the ovum, it must first pene-
A, Ovulation, fertilization of the ovum in the fallopian tube, and
trate the multiple layers of granulosa cells attached to
implantation of the blastocyst in the uterus. B, Action of tro-
the outside of the ovum (the corona radiata) and then
phoblast cells in implantation of the blastocyst in the uterine
endometrium.
bind to and penetrate the zona pellucida surrounding
the ovum itself. The mechanisms used by the sperm for
these purposes are presented in Chapter 80.
Once a sperm has entered the ovum (which is still
Transport of the Fertilized Ovum
in the secondary oocyte stage of development), the
in the Fallopian Tube
oocyte divides again to form the mature ovum plus
a second polar body that is expelled. The mature
After fertilization has occurred, an additional 3 to 5
ovum still carries in its nucleus (now called the female
days is normally required for transport of the fertilized
pronucleus) 23 chromosomes. One of these chromo-
ovum through the remainder of the fallopian tube into
somes is the female chromosome, known as the X
the cavity of the uterus (Figure 82-2). This transport is
chromosome.
effected mainly by a feeble fluid current in the tube
In the meantime, the fertilizing sperm has also
resulting from epithelial secretion plus action of the
changed. On entering the ovum, its head swells to form
ciliated epithelium that lines the tube; the cilia always
a male pronucleus, shown in Figure 82-1D. Later, the
beat toward the uterus. Weak contractions of the
23 unpaired chromosomes of the male pronucleus and
fallopian tube may also aid the ovum passage.
the 23 unpaired chromosomes of the female pronu-
The fallopian tubes are lined with a rugged, cryptoid
cleus align themselves to re-form a complete comple-
surface that impedes passage of the ovum despite the
ment of 46 chromosomes (23 pairs) in the fertilized
fluid current. Also, the isthmus of the fallopian tube
ovum (see Figure 82-1E).
(the last
2 centimeters before the tube enters the
uterus) remains spastically contracted for about the
What Determines the Sex of the Fetus That Is Created? After
first 3 days after ovulation. After this time, the rapidly
formation of the mature sperm, half of these carry in
increasing progesterone secreted by the ovarian
their genome an X chromosome (the female chromo-
corpus luteum first promotes increasing progesterone
some) and half carry a Y chromosome (the male
receptors on the fallopian tube smooth muscle cells;
chromosome). Therefore, if an X chromosome from
then the progesterone activates the receptors, exerting
a sperm combines with an X chromosome from an
a tubular relaxing effect that allows entry of the ovum
ovum, giving an XX combination, a female child will
into the uterus.
be born, as explained in Chapter 80. But if a Y chro-
This delayed transport of the fertilized ovum
mosome from a sperm is paired with an X chromo-
through the fallopian tube allows several stages of cell
some from an ovum, giving an XY combination, a male
division to occur before the dividing ovum—now
child will be born.
called a blastocyst, with about 100 cells—enters the
Chapter 82
Pregnancy and Lactation
1029
uterus. During this time, the fallopian tube secretory
endometrial stromal cells into large swollen cells con-
cells produce large quantities of secretions used for the
taining extra quantities of glycogen, proteins, lipids,
nutrition of the developing blastocyst.
and even some minerals necessary for development of
the conceptus. Then, when the conceptus implants in
the endometrium, the continued secretion of proges-
Implantation of the Blastocyst
terone causes the endometrial cells to swell further
in the Uterus
and to store even more nutrients. These cells are now
called decidual cells, and the total mass of cells is called
After reaching the uterus, the developing blastocyst
the decidua.
usually remains in the uterine cavity an additional 1 to
As the trophoblast cells invade the decidua, digest-
3 days before it implants in the endometrium; thus,
ing and imbibing it, the stored nutrients in the decidua
implantation ordinarily occurs on about the fifth to
are used by the embryo for growth and development.
seventh day after ovulation. Before implantation,
During the first week after implantation, this is the
the blastocyst obtains its nutrition from the uterine
only means by which the embryo can obtain nutrients;
endometrial secretions, called “uterine milk.”
the embryo continues to obtain at least some of its
Implantation results from the action of trophoblast
nutrition in this way for up to 8 weeks, although the
cells that develop over the surface of the blastocyst.
placenta also begins to provide nutrition after about
These cells secrete proteolytic enzymes that digest and
the 16th day beyond fertilization (a little more than 1
liquefy the adjacent cells of the uterine endometrium.
week after implantation). Figure 82-4 shows this tro-
Some of the fluid and nutrients released are actively
phoblastic period of nutrition, which gradually gives
transported by the same trophoblast cells into the blas-
way to placental nutrition.
tocyst, adding more sustenance for growth. Figure
82-3 shows an early implanted human blastocyst, with
a small embryo. Once implantation has taken place,
Function of the Placenta
the trophoblast cells and other adjacent cells (from the
blastocyst and the uterine endometrium) proliferate
Developmental and Physiologic
rapidly, forming the placenta and the various mem-
Anatomy of the Placenta
branes of pregnancy.
While the trophoblastic cords from the blastocyst are
attaching to the uterus, blood capillaries grow into the
cords from the vascular system of the newly forming
Early Nutrition of the Embryo
embryo. By the 16th day after fertilization, blood also
begins to be pumped by the heart of the embryo itself.
In Chapter 81, we pointed out that the progesterone
Simultaneously, blood sinuses supplied with blood from
secreted by the ovarian corpus luteum during the
the mother develop around the outsides of the tro-
latter half of each monthly sexual cycle has an
phoblastic cords. The trophoblast cells send out more
effect on the uterine endometrium, converting the
and more projections, which become placental villi into
Trophoblasts
Endometrium
100
Placental
diffusion
75
Ovum
50
Trophoblastic
25
nutrition
0
0
4
8
12
16
20
24
28
32
36
40
Duration of pregnancy (weeks)
Figure 82-4
Figure 82-3
Nutrition of the fetus. Most of the early nutrition is due to tro-
Implantation of the early human embryo, showing trophoblastic
phoblastic digestion and absorption of nutrients from the endome-
digestion and invasion of the endometrium. (Courtesy Dr. Arthur
trial decidua, and essentially all the later nutrition results from
Hertig.)
diffusion through the placental membrane.
1030
Unit XIV Endocrinology and Reproduction
PLACENTA
Placental Permeability and Membrane
Placental septum
To the mother
Diffusion Conductance
Stratum spongiosum
From the mother
The major function of the placenta is to provide for
diffusion of foodstuffs and oxygen from the mother’s
Limiting layer
blood into the fetus’s blood and diffusion of excretory
Maternal
products from the fetus back into the mother.
vessels
In the early months of pregnancy, the placental
membrane is still thick because it is not fully devel-
Villus
oped. Therefore, its permeability is low. Further, the
surface area is small because the placenta has not
grown significantly. Therefore, the total diffusion con-
ductance is minuscule at first. Conversely, in later preg-
nancy, the permeability increases because of thinning
of the membrane diffusion layers and because the
Intravillus
surface area expands many times over, thus giving the
space
tremendous increase in placental diffusion shown in
Amnion
Trophoblast
Figure 82-4.
Chorion
Umbilical arteries
Rarely, “breaks” occur in the placental membrane,
Marginal
which allows fetal blood cells to pass into the mother
Umbilical vein
sinus
or, even less commonly, the mother’s cells to pass into
Umbilical cord
the fetus. Fortunately, it is rare for the fetus to bleed
severely into the mother’s circulation because of a rup-
VILLUS
tured placental membrane.
Diffusion of Oxygen Through the Placental Membrane.
Fetal capillaries
Almost the same principles for diffusion of oxygen
through the pulmonary membrane (discussed in detail
Intervillous space
in Chapter 39) are applicable for diffusion of oxy-
gen through the placental membrane. The dissolved
Chorionic epithelium
oxygen in the blood of the large maternal sinuses
passes into the fetal blood by simple diffusion, driven
by an oxygen pressure gradient from the mother’s
blood to the fetus’s blood. Near the end of pregnancy,
Figure 82-5
the mean PO2 of the mother’s blood in the placental
sinuses is about 50 mm Hg, and the mean PO2 in the
Above, Organization of the mature placenta. Below, Relation of the
fetal blood in the villus capillaries to the mother’s blood in the inter-
fetal blood after it becomes oxygenated in the placenta
villous spaces. (Modified from Gray H, Goss CM: Anatomy of the
is about 30 mm Hg. Therefore, the mean pressure gra-
Human Body, 25th ed. Philadelphia: Lea & Febiger, 1948; and
from Arey LB: Developmental Anatomy: A Textbook and Labora-
dient for diffusion of oxygen through the placental
tory Manual of Embryology, 7th ed. Philadelphia: WB Saunders,
membrane is about 20 mm Hg.
1974.)
One might wonder how it is possible for a fetus to
obtain sufficient oxygen when the fetal blood leaving
the placenta has a PO2 of only 30 mm Hg. There are
which fetal capillaries grow. Thus, the villi, carrying fetal
three reasons why even this low PO2 is capable of
blood, are surrounded by sinuses that contain maternal
allowing the fetal blood to transport almost as much
blood.
oxygen to the fetal tissues as is transported by the
The final structure of the placenta is shown in Figure
mother’s blood to her tissues.
82-5. Note that the fetus’s blood flows through two
First, the hemoglobin of the fetus is mainly fetal
umbilical arteries, then into the capillaries of the villi,
hemoglobin, a type of hemoglobin synthesized in the
and finally back through a single umbilical vein into the
fetus before birth. Figure 82-6 shows the comparative
fetus. At the same time, the mother’s blood flows from
oxygen dissociation curves for maternal hemoglobin
her uterine arteries into large maternal sinuses that sur-
round the villi and then back into the uterine veins of
and fetal hemoglobin, demonstrating that the curve for
the mother. The lower part of Figure 82-5 shows the
fetal hemoglobin is shifted to the left of that for mater-
relation between the fetal blood of each fetal placental
nal hemoglobin. This means that at the low PO2 levels
villus and the blood of the mother surrounding the out-
in fetal blood, the fetal hemoglobin can carry 20 to 50
sides of the villus in the fully developed placenta.
per cent more oxygen than maternal hemoglobin can.
The total surface area of all the villi of the mature pla-
Second, the hemoglobin concentration of fetal blood
centa is only a few square meters—many times less than
is about 50 per cent greater than that of the mother; this
the area of the pulmonary membrane in the lungs. Nev-
is an even more important factor in enhancing the
ertheless, nutrients and other substances pass through
amount of oxygen transported to the fetal tissues.
this placental membrane mainly by diffusion in much
Third, the Bohr effect, which is explained in relation
the same manner that diffusion occurs through the
alveolar membranes of the lungs and the capillary
to the exchange of carbon dioxide and oxygen in the
membranes elsewhere in the body.
lung in Chapter 40, provides another mechanism to
Chapter 82
Pregnancy and Lactation
1031
mother’s blood. The PCO2 of the fetal blood is 2 to 3
mm Hg higher than that of the maternal blood. This
100
small pressure gradient for carbon dioxide across the
membrane is more than sufficient to allow adequate
Fetal
diffusion of carbon dioxide, because the extreme sol-
80
ubility of carbon dioxide in the placental membrane
allows carbon dioxide to diffuse about 20 times as
60
rapidly as oxygen.
Maternal
40
Diffusion of Foodstuffs Through the Placental Membrane.
Other metabolic substrates needed by the fetus
diffuse into the fetal blood in the same manner as
20
oxygen does. For instance, in the late stages of
Human
pregnancy, the fetus often uses as much glucose as
0
the entire body of the mother uses. To provide this
0
20
40
60
80
100
much glucose, the trophoblast cells lining the placen-
PO2
(mm Hg)
tal villi provide for facilitated diffusion of glucose
through the placental membrane. That is, the glucose
is transported by carrier molecules in the trophoblast
cells of the membrane. Even so, the glucose level in
Figure 82-6
fetal blood is 20 to 30 per cent lower than that in
maternal blood.
Oxygen-hemoglobin dissociation curves for maternal and fetal
blood, showing that fetal blood can carry a greater quantity of
Because of the high solubility of fatty acids in cell
oxygen than can maternal blood for a given blood PO2. (Data from
membranes, these also diffuse from the maternal
Metcalfe J, Moll W, Bartels H: Gas exchange across the placenta.
blood into the fetal blood, but more slowly than
Fed Proc 23:775, 1964.)
glucose, so that glucose is used more easily by the fetus
for nutrition. Also, such substances as ketone bodies
and potassium, sodium, and chloride ions diffuse with
enhance the transport of oxygen by fetal blood. That
relative ease from the maternal blood into the fetal
is, hemoglobin can carry more oxygen at a low PCO2
blood.
than it can at a high PCO2. The fetal blood entering the
placenta carries large amounts of carbon dioxide, but
Excretion of Waste Products Through the Placental Membrane.
much of this carbon dioxide diffuses from the fetal
In the same manner that carbon dioxide diffuses
blood into the maternal blood. Loss of the carbon
from the fetal blood into the maternal blood, other
dioxide makes the fetal blood more alkaline, whereas
excretory products formed in the fetus also diffuse
the increased carbon dioxide in the maternal blood
through the placental membrane into the maternal
makes it more acidic.
blood and are then excreted along with the excretory
These changes cause the capacity of fetal blood to
products of the mother. These include especially the
combine with oxygen to increase and that of maternal
nonprotein nitrogens such as urea, uric acid, and crea-
blood to decrease. This forces still more oxygen from
tinine. The level of urea in fetal blood is only slightly
the maternal blood, while enhancing oxygen uptake by
greater than that in maternal blood, because urea dif-
the fetal blood. Thus, the Bohr shift operates in one
fuses through the placental membrane with great ease.
direction in the maternal blood and in the other direc-
However, creatinine, which does not diffuse as easily,
tion in the fetal blood. These two effects make the
has a fetal blood concentration considerably higher
Bohr shift twice as important here as it is for oxygen
than that in the mother’s blood. Therefore, excretion
exchange in the lungs; therefore, it is called the double
from the fetus occurs mainly, if not entirely, as a result
Bohr effect.
of diffusion gradients across the placental membrane,
By these three means, the fetus is capable of receiv-
because there are higher concentrations of the excre-
ing more than adequate oxygen through the placental
tory products in the fetal blood than in the maternal
membrane, despite the fact that the fetal blood leaving
blood.
the placenta has a PO2 of only 30 mm Hg.
The total diffusing capacity of the entire placenta for
oxygen at term is about 1.2 milliliters of oxygen per
Hormonal Factors
minute per millimeter of mercury oxygen pressure dif-
ference across the membrane. This compares favorably
in Pregnancy
with that of the lungs of the newborn baby.
In pregnancy, the placenta forms especially large
Diffusion of Carbon Dioxide Through the Placental Membrane.
quantities of human chorionic gonadotropin, estrogens,
Carbon dioxide is continually formed in the tissues of
progesterone, and human chorionic somatomam-
the fetus in the same way that it is formed in maternal
motropin, the first three of which, and probably
tissues, and the only means for excreting the carbon
the fourth as well, are all essential to a normal
dioxide from the fetus is through the placenta into the
pregnancy.
1032
Unit XIV Endocrinology and Reproduction
Human Chorionic Gonadotropin
amounts of nutrients rather than being shed in the
menstruum. As a result, the decidua-like cells that
and Its Effect to Cause Persistence
develop in the endometrium during the normal female
of the Corpus Luteum and
sexual cycle become actual decidual cells—greatly
to Prevent Menstruation
swollen and nutritious—at about the time that the
blastocyst implants.
Menstruation normally occurs in a nonpregnant
Under the influence of human chorionic
woman about 14 days after ovulation, at which time
gonadotropin, the corpus luteum in the mother’s ovary
most of the endometrium of the uterus sloughs away
grows to about twice its initial size by a month or so
from the uterine wall and is expelled to the exterior.
after pregnancy begins, and its continued secretion of
If this should happen after an ovum has implanted,
estrogens and progesterone maintains the decidual
the pregnancy would terminate. However, this is
nature of the uterine endometrium, which is necessary
prevented by the secretion of human chorionic
for the early development of the fetus.
gonadotropin by the newly developing embryonic
If the corpus luteum is removed before approxi-
tissues in the following manner.
mately the
7th week of pregnancy, spontaneous
Coincidental with the development of the tro-
abortion almost always occurs, sometimes even up to
phoblast cells from the early fertilized ovum, the
the 12th week. After that time, the placenta secretes
hormone human chorionic gonadotropin is secreted by
sufficient quantities of progesterone and estrogens to
the syncytial trophoblast cells into the fluids of the
maintain pregnancy for the remainder of the gestation
mother, as shown in Figure 82-7. The secretion of this
period. The corpus luteum involutes slowly after the
hormone can first be measured in the blood 8 to 9 days
13th to 17th week of gestation.
after ovulation, shortly after the blastocyst implants in
the endometrium. Then the rate of secretion rises
Effect of Human Chorionic Gonadotropin on the Fetal
rapidly to reach a maximum at about 10 to 12 weeks
Testes. Human chorionic gonadotropin also exerts an
of pregnancy and decreases back to a lower value by
interstitial cell-stimulating effect on the testes of the
16 to 20 weeks. It continues at this level for the remain-
male fetus, resulting in the production of testosterone
der of pregnancy.
in male fetuses until the time of birth. This small secre-
tion of testosterone during gestation is what causes the
Function of Human Chorionic Gonadotropin. Human
fetus to grow male sex organs instead of female
chorionic gonadotropin is a glycoprotein having a
organs. Near the end of pregnancy, the testosterone
molecular weight of about
39,000 and much the
secreted by the fetal testes also causes the testes to
same molecular structure and function as luteinizing
descend into the scrotum.
hormone secreted by the pituitary gland. By far, its
most important function is to prevent involution of the
corpus luteum at the end of the monthly female sexual
cycle. Instead, it causes the corpus luteum to secrete
Secretion of Estrogens
even larger quantities of its sex hormones—proges-
by the Placenta
terone and estrogens—for the next few months. These
sex hormones prevent menstruation and cause the
The placenta, like the corpus luteum, secretes both
endometrium to continue to grow and store large
estrogens and progesterone. Histochemical and
Human chorionic
120
gonadotropin
24
Progesterone
22
100
Estrogens
20
300
18
80
16
14
200
60
12
10
40
8
100
6
20
4
2
Figure 82-7
0
0
0
Rates of secretion of estrogens
0
4
8
12
16
20
24
28
32
36
40
and progesterone, and con-
Duration of pregnancy (weeks)
centration of human chorionic
gonadotropin at different stages
of pregnancy.
Chapter 82
Pregnancy and Lactation
1033
physiological studies show that these two hormones,
2. Progesterone decreases the contractility
like most other placental hormones, are secreted by
of the pregnant uterus, thus preventing uterine
the syncytial trophoblast cells of the placenta.
contractions from causing spontaneous
Figure 82-7 shows that toward the end of pregnancy,
abortion.
the daily production of placental estrogens in-
3. Progesterone contributes to the development of
creases to about 30 times the mother’s normal level
the conceptus even before implantation, because
of production. However, the secretion of estrogens
it specifically increases the secretions of the
by the placenta is quite different from secretion by
mother’s fallopian tubes and uterus to provide
the ovaries. Most important, the estrogens secreted
appropriate nutritive matter for the developing
by the placenta are not synthesized de novo from
morula and blastocyst. There is also reason to
basic substrates in the placenta. Instead, they are
believe that progesterone affects cell cleavage in
formed almost entirely from androgenic steroid
the early developing embryo.
compounds, dehydroepiandrosterone and
16-
4. The progesterone secreted during pregnancy helps
hydroxydehydroepiandrosterone, which are formed
the estrogen prepare the mother’s breasts for
both in the mother’s adrenal glands and in the adrenal
lactation, which is discussed later in this chapter.
glands of the fetus. These weak androgens are trans-
ported by the blood to the placenta and converted by
the trophoblast cells into estradiol, estrone, and estriol.
Human Chorionic
(The cortices of the fetal adrenal glands are extremely
Somatomammotropin
large, and about 80 per cent consists of a so-called
fetal zone, the primary function of which seems to
A more recently discovered placental hormone is
be to secrete dehydroepiandrosterone during
called human chorionic somatomammotropin. It is a
pregnancy.)
protein with a molecular weight of about 38,000, and
it begins to be secreted by the placenta at about the
fifth week of pregnancy. Secretion of this hormone
Function of Estrogen in Pregnancy. In the discussions of
increases progressively throughout the remainder
estrogens in Chapter 81, we pointed out that these hor-
of pregnancy in direct proportion to the weight of
mones exert mainly a proliferative function on most
the placenta. Although the functions of chorionic
reproductive and associated organs of the mother.
somatomammotropin are uncertain, it is secreted in
During pregnancy, the extreme quantities of estrogens
quantities several times greater than all the other preg-
cause (1) enlargement of the mother’s uterus, (2)
nancy hormones combined. It has several possible
enlargement of the mother’s breasts and growth of the
important effects.
breast ductal structure, and (3) enlargement of the
First, when administered to several types of lower
mother’s female external genitalia.
animals, human chorionic somatomammotropin
The estrogens also relax the pelvic ligaments
causes at least partial development of the animal’s
of the mother, so that the sacroiliac joints become
breasts and in some instances causes lactation.
relatively limber and the symphysis pubis becomes
Because this was the first function of the hormone dis-
elastic. These changes allow easier passage of the fetus
covered, it was first named human placental lactogen
through the birth canal. There is much reason to
and was believed to have functions similar to those of
believe that estrogens also affect many general aspects
prolactin. However, attempts to promote lactation in
of fetal development during pregnancy, for example,
humans with its use have not been successful.
by affecting the rate of cell reproduction in the early
Second, this hormone has weak actions similar to
embryo.
those of growth hormone, causing the formation of
protein tissues in the same way that growth hormone
does. It also has a chemical structure similar to that of
Secretion of Progesterone
growth hormone, but
100 times as much human
by the Placenta
chorionic somatomammotropin as growth hormone
is required to promote growth.
Progesterone is also essential for a successful
Third, human chorionic somatomammotropin
pregnancy—in fact, it is just as important as estrogen.
causes decreased insulin sensitivity and decreased uti-
In addition to being secreted in moderate quantities
lization of glucose in the mother, thereby making
by the corpus luteum at the beginning of pregnancy, it
larger quantities of glucose available to the fetus.
is secreted later in tremendous quantities by the
Because glucose is the major substrate used by the
placenta, averaging about a 10-fold increase during the
fetus to energize its growth, the possible importance
course of pregnancy, as shown in Figure 82-7.
of such a hormonal effect is obvious. Further, the
The special effects of progesterone that are essen-
hormone promotes the release of free fatty acids from
tial for the normal progression of pregnancy are as
the fat stores of the mother, thus providing this alter-
follows:
native source of energy for the mother’s metabolism
1. Progesterone causes decidual cells to develop
during pregnancy. Therefore, it appears that human
in the uterine endometrium, and these cells play
chorionic somatomammotropin is a general metabolic
an important role in the nutrition of the early
hormone that has specific nutritional implications for
embryo.
both the mother and the fetus.
1034
Unit XIV Endocrinology and Reproduction
Other Hormonal Factors
probably played mainly by the estrogens, which also
in Pregnancy
cause relaxation of the pelvic ligaments. It has also been
claimed that relaxin softens the cervix of the pregnant
Almost all the nonsexual endocrine glands of the
woman at the time of delivery.
mother also react markedly to pregnancy. This results
mainly from the increased metabolic load on the mother
but also, to some extent, from the effects of placental
Response of the Mother’s
hormones on the pituitary and other glands. Some of the
Body to Pregnancy
most notable effects are the following.
Most apparent among the many reactions of the mother
Pituitary Secretion. The anterior pituitary gland of the
to the fetus and to the excessive hormones of pregnancy
mother enlarges at least 50 per cent during pregnancy
is the increased size of the various sexual organs. For
and increases its production of corticotropin, thy-
instance, the uterus increases from about 50 grams to
rotropin, and prolactin. Conversely, pituitary secretion
1100 grams, and the breasts approximately double in
of follicle-stimulating hormone and luteinizing
size. At the same time, the vagina enlarges and the
hormone is almost totally suppressed as a result of the
introitus opens more widely. Also, the various hormones
inhibitory effects of estrogens and progesterone from
can cause marked changes in a pregnant woman’s
the placenta.
appearance, sometimes resulting in the development of
edema, acne, and masculine or acromegalic features.
Corticosteroid Secretion. The rate of adrenocortical secre-
tion of the glucocorticoids is moderately increased
Weight Gain in the Pregnant Woman
throughout pregnancy. It is possible that these gluco-
The average weight gain during pregnancy is about 24
corticoids help mobilize amino acids from the mother’s
pounds, with most of this gain occurring during the last
tissues so that these can be used for synthesis of tissues
two trimesters. Of this, about 7 pounds is fetus and 4
in the fetus.
pounds is amniotic fluid, placenta, and fetal membranes.
Pregnant women usually have about a twofold
The uterus increases about 2 pounds and the breasts
increase in the secretion of aldosterone, reaching a peak
another
2 pounds, still leaving an average weight
at the end of gestation. This, along with the actions of
increase of 9 pounds. About 6 pounds of this is extra
estrogens, causes a tendency for even a normal pregnant
fluid in the blood and extracellular fluid, and the
woman to reabsorb excess sodium from her renal
remaining 3 pounds is generally fat accumulation. The
tubules and, therefore, to retain fluid, occasionally
extra fluid is excreted in the urine during the first few
leading to pregnancy-induced hypertension.
days after birth, that is, after loss of the fluid-retaining
hormones from the placenta.
Secretion by the Thyroid Gland. The mother’s thyroid gland
During pregnancy, a woman often has a greatly
ordinarily enlarges up to 50 per cent during pregnancy
increased desire for food, partly as a result of removal
and increases its production of thyroxine a correspon-
of food substrates from the mother’s blood by the fetus
ding amount. The increased thyroxine production is
and partly because of hormonal factors. Without appro-
caused at least partly by a thyrotropic effect of human
priate prenatal control of diet, the mother’s weight gain
chorionic gonadotropin secreted by the placenta and
can be as great as 75 pounds instead of the usual 24
by small quantities of a specific thyroid-stimulating
pounds.
hormone, human chorionic thyrotropin, also secreted by
the placenta.
Metabolism During Pregnancy
As a consequence of the increased secretion of many
Secretion by the Parathyroid Glands. The mother’s parathy-
hormones during pregnancy, including thyroxine,
roid glands usually enlarge during pregnancy; this is
adrenocortical hormones, and the sex hormones, the
especially true if the mother is on a calcium-deficient
basal metabolic rate of the pregnant woman increases
diet. Enlargement of these glands causes calcium
about 15 per cent during the latter half of pregnancy. As
absorption from the mother’s bones, thereby maintain-
a result, she frequently has sensations of becoming over-
ing normal calcium ion concentration in the mother’s
heated. Also, owing to the extra load that she is carry-
extracellular fluid even while the fetus removes calcium
ing, greater amounts of energy than normal must be
to ossify its own bones. This secretion of parathyroid
expended for muscle activity.
hormone is even more intensified during lactation after
the baby’s birth, because the growing baby requires
Nutrition During Pregnancy. By far the greatest growth of
many times more calcium than the fetus does.
the fetus occurs during the last trimester of pregnancy;
its weight almost doubles during the last 2 months of
Secretion of “Relaxin” by the Ovaries and Placenta. Another
pregnancy. Ordinarily, the mother does not absorb suf-
substance besides the estrogens and progesterone, a
ficient protein, calcium, phosphates, and iron from her
hormone called relaxin, is secreted by the corpus luteum
diet during the last months of pregnancy to supply these
of the ovary and by placental tissues. Its secretion is
extra needs of the fetus. However, anticipating these
increased by a stimulating effect of human chorionic
extra needs, the mother’s body has already been storing
gonadotropin at the same time that the corpus luteum
these substances—some in the placenta, but most in the
and the placenta secrete large quantities of estrogens
normal storage depots of the mother.
and progesterone.
If appropriate nutritional elements are not present in
Relaxin is a polypeptide having a molecular weight of
a pregnant woman’s diet, a number of maternal defi-
about 9000. This hormone, when injected, causes relax-
ciencies can occur, especially in calcium, phosphates,
ation of the ligaments of the symphysis pubis in the
iron, and the vitamins. For example, about 375 mil-
estrous rat and guinea pig. This effect is weak or possi-
ligrams of iron is needed by the fetus to form its blood,
bly even absent in pregnant women. Instead, this role is
and an additional 600 milligrams is needed by the
Chapter 82
Pregnancy and Lactation
1035
mother to form her own extra blood. The normal store
birth of the baby is about 20 per cent above normal, and
of nonhemoglobin iron in the mother at the outset of
a commensurate amount of carbon dioxide is formed.
pregnancy is often only 100 milligrams and almost never
These effects cause the mother’s minute ventilation to
more than 700 milligrams. Therefore, without sufficient
increase. It is also believed that the high levels of prog-
iron in her food, a pregnant woman usually develops
esterone during pregnancy increase the minute ventila-
hypochromic anemia. Also, it is especially important
tion even more, because progesterone increases the
that she receive vitamin D, because although the total
respiratory center’s sensitivity to carbon dioxide. The
quantity of calcium used by the fetus is small, calcium
net result is an increase in minute ventilation of about
is normally poorly absorbed by the mother’s gastroin-
50 per cent and a decrease in arterial PCO2 to several
testinal tract without vitamin D. Finally, shortly before
millimeters of mercury below that in a nonpregnant
birth of the baby, vitamin K is often added to the
woman. Simultaneously, the growing uterus presses
mother’s diet so that the baby will have sufficient pro-
upward against the abdominal contents, and these press
thrombin to prevent hemorrhage, particularly brain
upward against the diaphragm, so that the total excur-
hemorrhage, caused by the birth process.
sion of the diaphragm is decreased. Consequently, the
respiratory rate is increased to maintain the extra
ventilation.
Changes in the Maternal Circulatory
System During Pregnancy
Function of the Maternal Urinary System During Pregnancy
The rate of urine formation by a pregnant woman is
Blood Flow Through the Placenta, and Cardiac Output During
usually slightly increased because of increased fluid
Pregnancy. About 625 milliliters of blood flows through
intake and increased load or excretory products. But in
the maternal circulation of the placenta each minute
addition, several special alterations of urinary function
during the last month of pregnancy. This, plus the
occur.
general increase in the mother’s metabolism, increases
First, the renal tubules’ reabsorptive capacity for
the mother’s cardiac output to 30 to 40 per cent above
sodium, chloride, and water is increased as much as
normal by the 27th week of pregnancy; then, for reasons
50 per cent as a consequence of increased production
unexplained, the cardiac output falls to only a little
of steroid hormones by the placenta and adrenal
above normal during the last 8 weeks of pregnancy,
cortex.
despite the high uterine blood flow.
Second, the glomerular filtration rate increases as
much as 50 per cent during pregnancy, which tends to
Blood Volume During Pregnancy. The maternal blood
increase the rate of water and electrolyte excretion in
volume shortly before term is about 30 per cent above
the urine. When all these effects are considered, the
normal. This increase occurs mainly during the latter
normal pregnant woman ordinarily accumulates only
half of pregnancy, as shown by the curve of Figure 82-8.
about 6 pounds of extra water and salt.
The cause of the increased volume is likely due, at least
in part, to aldosterone and estrogens, which are greatly
Amniotic Fluid and Its Formation
increased in pregnancy, and to increased fluid retention
Normally, the volume of amniotic fluid (the fluid inside
by the kidneys. Also, the bone marrow becomes increas-
the uterus in which the fetus floats) is between 500 mil-
ingly active and produces extra red blood cells to go
liliters and 1 liter, but it can be only a few milliliters or
with the excess fluid volume. Therefore, at the time of
as much as several liters. Isotope studies of the rate of
birth of the baby, the mother has about 1 to 2 liters of
formation of amniotic fluid show that, on average, the
extra blood in her circulatory system. Only about one
water in amniotic fluid is replaced once every 3 hours,
fourth of this amount is normally lost through bleeding
and the electrolytes sodium and potassium are replaced
during delivery of the baby, thereby allowing a consid-
an average of once every 15 hours. A large portion of
erable safety factor for the mother.
the fluid is derived from renal excretion by the fetus.
Likewise, a certain amount of absorption occurs by way
Maternal Respiration During Pregnancy
of the gastrointestinal tract and lungs of the fetus.
Because of the increased basal metabolic rate of a preg-
However, even after in utero death of a fetus, some
nant woman and because of her greater size, the total
turnover of the amniotic fluid is still present, which indi-
amount of oxygen used by the mother shortly before
cates that some of the fluid is formed and absorbed
directly through the amniotic membranes.
Preeclampsia and Eclampsia
6
About 5 per cent of all pregnant women experience a
rapid rise in arterial blood pressure to hypertensive
levels during the last few months of pregnancy. This is
5
also associated with leakage of large amounts of protein
into the urine. This condition is called preeclampsia or
toxemia of pregnancy. It is often characterized by excess
4
salt and water retention by the mother’s kidneys and by
0
weight gain and development of edema and hyperten-
0
4
8
12
16
20
24
28
32
36
40
44
sion in the mother. In addition, there is impaired func-
Duration of pregnancy (weeks)
tion of the vascular endothelium, and arterial spasm
occurs in many parts of the mother’s body, most sig-
nificantly in the kidneys, brain, and liver. Both the renal
blood flow and the glomerular filtration rate are
Figure 82-8
decreased, which is exactly opposite to the changes that
Effect of pregnancy to increase the mother’s blood volume.
occur in the normal pregnant woman. The renal effects
1036
Unit XIV Endocrinology and Reproduction
also include thickened glomerular tufts that contain a
helping to prevent expulsion of the fetus. Conversely,
protein deposit in the basement membranes.
estrogens have a definite tendency to increase the
Various attempts have been made to prove that
degree of uterine contractility, partly because
preeclampsia is caused by excessive secretion of pla-
estrogens increase the number of gap junctions
cental or adrenal hormones, but proof of a hormonal
between the adjacent uterine smooth muscle cells, but
basis is still lacking. Another theory is that preeclamp-
also because of other poorly understood effects. Both
sia results from some type of autoimmunity or allergy
progesterone and estrogen are secreted in progres-
in the mother caused by the presence of the fetus. In
sively greater quantities throughout most of preg-
support of this, the acute symptoms usually disappear
within a few days after birth of the baby. There is also
nancy, but from the seventh month onward, estrogen
evidence that preeclampsia is initiated by insufficient
secretion continues to increase while progesterone
blood supply to the placenta, resulting in the placenta’s
secretion remains constant or perhaps even decreases
release of substances that cause widespread dysfunction
slightly. Therefore, it has been postulated that the
of the maternal vascular endothelium. During normal
estrogen-to-progesterone ratio increases sufficiently
placental development, the trophoblasts invade the
toward the end of pregnancy to be at least partly
arterioles of the uterine endometrium and completely
responsible for the increased contractility of the
remodel the maternal arterioles into large blood vessels
uterus.
with low resistance to blood flow. In patients with
preeclampsia, the maternal arterioles fail to undergo
Effect of Oxytocin on the Uterus. Oxytocin is a hormone
these adaptive changes, for reasons that are still unclear,
and there is insufficient blood supply to the placenta.
secreted by the neurohypophysis that specifically
This, in turn, causes the placenta to release various sub-
causes uterine contraction (see Chapter 75). There are
stances that enter the mother’s circulation and cause
four reasons to believe that oxytocin might be impor-
impaired vascular endothelial function, decreased blood
tant in increasing the contractility of the uterus near
flow to the kidneys, excess salt and water retention, and
term: (1) The uterine muscle increases its oxytocin
increased blood pressure. Although the factors that link
receptors and, therefore, increases its responsiveness
reduced placental blood supply with maternal endothe-
to a given dose of oxytocin during the latter few
lial dysfunction are still uncertain, some experimental
months of pregnancy. (2) The rate of oxytocin secre-
studies suggest a role for increased levels of inflamma-
tion by the neurohypophysis is considerably increased
tory cytokines such as tumor necrosis factor-a and
at the time of labor. (3) Although hypophysectomized
interleukin-6.
Eclampsia is an extreme degree of preeclampsia,
animals can still deliver their young at term, labor is
characterized by vascular spasm throughout the body;
prolonged.
(4) Experiments in animals indicate
clonic seizures in the mother, sometimes followed by
that irritation or stretching of the uterine cervix, as
coma; greatly decreased kidney output; malfunction of
occurs during labor, can cause a neurogenic reflex
the liver; often extreme hypertension; and a generalized
through the paraventricular and supraoptic nuclei of
toxic condition of the body. It usually occurs shortly
the hypothalamus that causes the posterior pituitary
before birth of the baby. Without treatment, a high per-
gland (the neurohypophysis) to increase its secretion
centage of eclamptic mothers die. However, with optimal
of oxytocin.
and immediate use of rapidly acting vasodilating drugs
to reduce the arterial pressure to normal, followed by
Effect of Fetal Hormones on the Uterus. The fetus’s pitu-
immediate termination of pregnancy—by cesarean
section if necessary—the mortality even in eclamptic
itary gland secretes increasing quantities of oxytocin,
mothers has been reduced to 1 per cent or less.
which might play a role in exciting the uterus. Also, the
fetus’s adrenal glands secrete large quantities of corti-
sol, another possible uterine stimulant. In addition, the
Parturition
fetal membranes release prostaglandins in high con-
centration at the time of labor. These, too, can increase
Increased Uterine Excitability
the intensity of uterine contractions.
Near Term
Mechanical Factors That Increase
Parturition means birth of the baby. Toward the end of
Uterine Contractility
pregnancy, the uterus becomes progressively more
Stretch of the Uterine Musculature. Simply stretching
excitable, until finally it develops such strong rhythmi-
smooth muscle organs usually increases their contrac-
cal contractions that the baby is expelled. The exact
tility. Further, intermittent stretch, as occurs repeat-
cause of the increased activity of the uterus is not
edly in the uterus because of fetal movements, can also
known, but at least two major categories of effects lead
elicit smooth muscle contraction. Note especially that
up to the intense contractions responsible for parturi-
twins are born, on average, 19 days earlier than a single
tion:
(1) progressive hormonal changes that cause
child, which emphasizes the importance of mechanical
increased excitability of the uterine musculature, and
stretch in eliciting uterine contractions.
(2) progressive mechanical changes.
Stretch or Irritation of the Cervix. There is reason to
Hormonal Factors That Increase
believe that stretching or irritating the uterine cervix
Uterine Contractility
is particularly important in eliciting uterine contrac-
Increased Ratio of Estrogens to Progesterone. Progesterone
tions. For instance, the obstetrician frequently induces
inhibits uterine contractility during pregnancy, thereby
labor by rupturing the membranes so that the head of
Chapter 82
Pregnancy and Lactation
1037
the baby stretches the cervix more forcefully than
usual or irritates it in other ways.
The mechanism by which cervical irritation excites
the body of the uterus is not known. It has been
suggested that stretching or irritation of nerves in the
cervix initiates reflexes to the body of the uterus, but
the effect could also result simply from myogenic
transmission of signals from the cervix to the body of
the uterus.
Onset of Labor—A Positive Feedback
Mechanism for Its Initiation
During most of the months of pregnancy, the uterus
undergoes periodic episodes of weak and slow rhyth-
mical contractions called Braxton Hicks contractions.
1. Baby's head stretches cervix
These contractions become progressively stronger
2. Cervical stretch excites fundic contraction
3. Fundic contraction pushes baby down and stretches
toward the end of pregnancy; then they change sud-
cervix some more
denly, within hours, to become exceptionally strong
4. Cycle repeats over and over again
contractions that start stretching the cervix and later
force the baby through the birth canal, thereby causing
Figure 82-9
parturition. This process is called labor, and the strong
Theory for the onset of intensely strong contractions during labor.
contractions that result in final parturition are called
labor contractions.
We do not know what suddenly changes the slow,
weak rhythmicity of the uterus into strong labor con-
contractility still more because of positive feedback,
tractions. However, based on experience with other
resulting in a second uterine contraction stronger than
types of physiological control systems, a theory has
the first, a third stronger than the second, and so forth.
been proposed for explaining the onset of labor. The
Once these contractions become strong enough to
positive feedback theory suggests that stretching of
cause this type of feedback, with each succeeding con-
the cervix by the fetus’s head finally becomes great
traction greater than the preceding one, the process
enough to elicit a strong reflex increase in contractil-
proceeds to completion—all because positive feedback
ity of the uterine body. This pushes the baby forward,
initiates a vicious circle when the gain of the feedback
which stretches the cervix more and initiates more
is greater than a critical level.
positive feedback to the uterine body. Thus, the
One might ask about the many instances of false
process repeats until the baby is expelled. This theory
labor, in which the contractions become stronger and
is shown in Figure 82-9, and the observations sup-
stronger and then fade away. Remember that for a
porting it are the following.
vicious circle to continue, each new cycle of the posi-
First, labor contractions obey all the principles of
tive feedback must be stronger than the previous one.
positive feedback. That is, once the strength of uterine
If at any time after labor starts some contractions fail
contraction becomes greater than a critical value, each
to re-excite the uterus sufficiently, the positive feed-
contraction leads to subsequent contractions that
back could go into a retrograde decline, and the labor
become stronger and stronger until maximum effect is
contractions would fade away.
achieved. Referring to the discussion in Chapter 1 of
positive feedback in control systems, one can see that
this is the precise nature of all positive feedback mech-
Abdominal Muscle Contractions
anisms when the feedback gain becomes greater than
During Labor
a critical value.
Second, two known types of positive feedback
Once uterine contractions become strong during
increase uterine contractions during labor: (1) Stretch-
labor, pain signals originate both from the uterus itself
ing of the cervix causes the entire body of the uterus
and from the birth canal. These signals, in addition to
to contract, and this contraction stretches the cervix
causing suffering, elicit neurogenic reflexes in the
even more because of the downward thrust of the
spinal cord to the abdominal muscles, causing intense
baby’s head. (2) Cervical stretching also causes the
contractions of these muscles. The abdominal contrac-
pituitary gland to secrete oxytocin, which is another
tions add greatly to the force that causes expulsion of
means for increasing uterine contractility.
the baby.
To summarize, we can assume that multiple factors
increase the contractility of the uterus toward the
Mechanics of Parturition
end of pregnancy. Eventually a uterine contraction
becomes strong enough to irritate the uterus, espe-
The uterine contractions during labor begin mainly at
cially at the cervix, and this increases uterine
the top of the uterine fundus and spread downward over
1038
Unit XIV Endocrinology and Reproduction
the body of the uterus. Also, the intensity of contraction
Labor Pains
is great in the top and body of the uterus but weak in
the lower segment of the uterus adjacent to the cervix.
With each uterine contraction, the mother experiences
Therefore, each uterine contraction tends to force the
considerable pain. The cramping pain in early labor
baby downward toward the cervix.
is probably caused mainly by hypoxia of the uterine
In the early part of labor, the contractions might
muscle resulting from compression of the blood vessels
occur only once every 30 minutes. As labor progresses,
in the uterus. This pain is not felt when the visceral
the contractions finally appear as often as once every 1
sensory hypogastric nerves, which carry the visceral
to 3 minutes, and the intensity of contraction increases
sensory fibers leading from the uterus, have been
greatly, with only a short period of relaxation between
sectioned.
contractions. The combined contractions of the uterine
However, during the second stage of labor, when the
and abdominal musculature during delivery of the baby
fetus is being expelled through the birth canal, much
cause a downward force on the fetus of about 25 pounds
more severe pain is caused by cervical stretching, per-
during each strong contraction.
ineal stretching, and stretching or tearing of structures
It is fortunate that the contractions of labor occur
in the vaginal canal itself. This pain is conducted to the
intermittently, because strong contractions impede or
mother’s spinal cord and brain by somatic nerves
sometimes even stop blood flow through the placenta
instead of by the visceral sensory nerves.
and would cause death of the fetus if the contractions
were continuous. Indeed, overuse of various uterine
stimulants, such as oxytocin, can cause uterine spasm
Involution of the Uterus
rather than rhythmical contractions and can lead to
After Parturition
death of the fetus.
In about 95 per cent of births, the head is the first part
During the first 4 to 5 weeks after parturition, the uterus
of the baby to be expelled, and in most of the remain-
involutes. Its weight becomes less than half its immedi-
ing instances, the buttocks are presented first. The head
ate postpartum weight within 1 week, and in 4 weeks, if
acts as a wedge to open the structures of the birth canal
the mother lactates, the uterus may become as small as
as the fetus is forced downward.
it was before pregnancy. This effect of lactation results
The first major obstruction to expulsion of the fetus
from the suppression of pituitary gonadotropin and
is the uterine cervix. Toward the end of pregnancy, the
ovarian hormone secretion during the first few months
cervix becomes soft, which allows it to stretch when
of lactation, as discussed later. During early involution
labor contractions begin in the uterus. The so-called first
of the uterus, the placental site on the endometrial
stage of labor is a period of progressive cervical dilation,
surface autolyzes, causing a vaginal discharge known as
lasting until the cervical opening is as large as the head
“lochia,” which is first bloody and then serous in nature,
of the fetus. This stage usually lasts for 8 to 24 hours in
continuing for a total of about 10 days. After this time,
the first pregnancy but often only a few minutes after
the endometrial surface becomes re-epithelialized and
many pregnancies.
ready for normal, nongravid sex life again.
Once the cervix has dilated fully, the fetal membranes
usually rupture and the amniotic fluid is lost suddenly
through the vagina. Then the fetus’s head moves rapidly
Lactation
into the birth canal, and with additional force from
above, it continues to wedge its way through the canal
Development of the Breasts
until delivery is effected. This is called the second stage
of labor, and it may last from as little as 1 minute after
many pregnancies to 30 minutes or more in the first
The breasts, shown in Figure 82-10, begin to develop
pregnancy.
at puberty. This development is stimulated by the
estrogens of the monthly female sexual cycle; estro-
gens stimulate growth of the breasts’ mammary glands
plus the deposition of fat to give the breasts mass. In
Separation and Delivery
addition, far greater growth occurs during the high-
of the Placenta
estrogen state of pregnancy, and only then does the
glandular tissue become completely developed for the
For 10 to 45 minutes after birth of the baby, the
production of milk.
uterus continues to contract to a smaller and smaller
size, which causes a shearing effect between the walls of
the uterus and the placenta, thus separating the placenta
Growth of the Ductal System—Role of the Estrogens. All
from its implantation site. Separation of the placenta
through pregnancy, the large quantities of estrogens
opens the placental sinuses and causes bleeding. The
secreted by the placenta cause the ductal system of the
amount of bleeding is limited to an average of 350 mil-
breasts to grow and branch. Simultaneously, the
liliters by the following mechanism: The smooth muscle
stroma of the breasts increases in quantity, and large
fibers of the uterine musculature are arranged in figures
quantities of fat are laid down in the stroma.
of eight around the blood vessels as the vessels pass
Also important for growth of the ductal system are
through the uterine wall. Therefore, contraction of
at least four other hormones: growth hormone, pro-
the uterus after delivery of the baby constricts the
lactin, the adrenal glucocorticoids, and insulin. Each of
vessels that had previously supplied blood to the pla-
these is known to play at least some role in protein
centa. In addition, it is believed that vasoconstrictor
prostaglandins formed at the placental separation site
metabolism, which presumably explains their function
cause additional blood vessel spasm.
in the development of the breasts.
Chapter 82
Pregnancy and Lactation
1039
Initiation of Lactation—Function
Pectoralis major
of Prolactin
Adipose tissue
Although estrogen and progesterone are essential for
the physical development of the breasts during preg-
Lobules and
nancy, a specific effect of both these hormones is to
alveoli
inhibit the actual secretion of milk. Conversely, the
hormone prolactin has exactly the opposite effect
Lactiferous
on milk secretion—promoting it. This hormone is
sinus (ampulla)
secreted by the mother’s anterior pituitary gland, and
Lactiferous duct
its concentration in her blood rises steadily from the
Nipple
fifth week of pregnancy until birth of the baby, at
which time it has risen to 10 to 20 times the normal
Areola
nonpregnant level. This high level of prolactin at the
end of pregnancy is shown in Figure 82-11.
In addition, the placenta secretes large quantities of
A
human chorionic somatomammotropin, which proba-
bly has lactogenic properties, thus supporting the pro-
Lobule
lactin from the mother’s pituitary during pregnancy.
Even so, because of the suppressive effects of estrogen
and progesterone, no more than a few milliliters of
Alveoli
fluid are secreted each day until after the baby is born.
The fluid secreted during the last few days before and
the first few days after parturition is called colostrum;
B
it contains essentially the same concentrations of pro-
teins and lactose as milk, but it has almost no fat, and
Myoepithelial cells
its maximum rate of production is about 1/100 the
subsequent rate of milk production.
Immediately after the baby is born, the sudden loss
of both estrogen and progesterone secretion from the
placenta allows the lactogenic effect of prolactin from
Ductule
the mother’s pituitary gland to assume its natural milk-
Milk
promoting role, and over the next 1 to 7 days, the
breasts begin to secrete copious quantities of milk
instead of colostrum. This secretion of milk requires
Milk
secreting
an adequate background secretion of most of the
epithelial
mother’s other hormones as well, but most important
cells
are growth hormone, cortisol, parathyroid hormone,
C
and insulin. These hormones are necessary to provide
the amino acids, fatty acids, glucose, and calcium
Figure 82-10
required for milk formation.
After birth of the baby, the basal level of prolactin
The breast and its secretory lobules, alveoli, and lactiferous ducts
secretion returns to the nonpregnant level over the
(milk ducts) that constitute its mammary gland (A). The enlarge-
ments show a lobule (B) and milk-secreting cells of an alveolus
next few weeks, as shown in Figure 82-11. However,
(C).
each time the mother nurses her baby, nervous signals
from the nipples to the hypothalamus cause a 10- to
20-fold surge in prolactin secretion that lasts for about
1 hour, which is also shown in Figure 82-11. This
prolactin acts on the mother’s breasts to keep the
Development of the Lobule-Alveolar System—Role of Proges-
mammary glands secreting milk into the alveoli for the
terone. Final development of the breasts into milk-
subsequent nursing periods. If this prolactin surge is
secreting organs also requires progesterone. Once the
absent or blocked as a result of hypothalamic or pitu-
ductal system has developed, progesterone—acting
itary damage or if nursing does not continue, the
synergistically with estrogen, as well as with the other
breasts lose their ability to produce milk within 1 week
hormones just mentioned—causes additional growth
or so. However, milk production can continue for
of the breast lobules, with budding of alveoli and
several years if the child continues to suckle, although
development of secretory characteristics in the cells of
the rate of milk formation normally decreases consid-
the alveoli. These changes are analogous to the
erably after 7 to 9 months.
secretory effects of progesterone on the endometrium
of the uterus during the latter half of the female
Hypothalamic Control of Prolactin Secretion. The hypothal-
menstrual cycle.
amus plays an essential role in controlling prolactin
1040
Unit XIV Endocrinology and Reproduction
Estrogens
Progesterone
Prolactin
Intermittent secretion
of prolactin during
2.0
300
nursing
200
1.5
Figure 82-11
200
Changes in rates of secretion
1.0
100
of estrogens, progesterone, and
prolactin for 8 weeks before par-
100
turition and 36 weeks thereafter.
0.5
Note especially the decrease of
prolactin secretion back to basal
levels within a few weeks after
0
0
0
parturition, but also the intermit-
-8
-4
0
4
8
12
16
20
24
28
32
36
tent periods of marked prolactin
Weeks after parturition
secretion (for about 1 hour at a
time) during and after periods of
nursing.
secretion, as it does for almost all the other anterior
Ejection (or “Let-Down”) Process in
pituitary hormones. However, this control is different
Milk Secretion—Function of Oxytocin
in one aspect: The hypothalamus mainly stimulates
production of all the other hormones, but it mainly
Milk is secreted continuously into the alveoli of the
inhibits prolactin production. Consequently, damage
breasts, but milk does not flow easily from the alveoli
to the hypothalamus or blockage of the hypothalamic-
into the ductal system and, therefore, does not contin-
hypophysial portal system often increases prolactin
ually leak from the breast nipples. Instead, the milk
secretion while it depresses secretion of the other
must be ejected from the alveoli into the ducts before
anterior pituitary hormones.
the baby can obtain it. This is caused by a combined
Therefore, it is believed that anterior pituitary secre-
neurogenic and hormonal reflex that involves the
tion of prolactin is controlled either entirely or almost
posterior pituitary hormone oxytocin, as follows.
entirely by an inhibitory factor formed in the hypo-
When the baby suckles, it receives virtually no milk
thalamus and transported through the hypothalamic-
for the first half minute or so. Sensory impulses must
hypophysial portal system to the anterior pituitary
first be transmitted through somatic nerves from the
gland. This factor is called prolactin inhibitory
nipples to the mother’s spinal cord and then to her
hormone. It is almost certainly the same as the cate-
hypothalamus, where they cause nerve signals that
cholamine dopamine, which is known to be secreted
promote oxytocin secretion at the same time that they
by the arcuate nuclei of the hypothalamus and can
cause prolactin secretion. The oxytocin is carried in the
decrease prolactin secretion as much as 10-fold.
blood to the breasts, where it causes myoepithelial cells
(which surround the outer walls of the alveoli) to con-
Suppression of the Female Ovarian Cycles in Nursing Mothers
tract, thereby expressing the milk from the alveoli into
for Many Months After Delivery. In most nursing mothers,
the ducts at a pressure of +10 to 20 mm Hg. Then the
the ovarian cycle (and ovulation) does not resume
baby’s suckling becomes effective in removing the
until a few weeks after cessation of nursing. The reason
milk. Thus, within 30 seconds to 1 minute after a baby
seems to be that the same nervous signals from
begins to suckle, milk begins to flow. This process is
the breasts to the hypothalamus that cause prolactin
called milk ejection or milk let-down.
secretion during suckling—either because of the
Suckling on one breast causes milk flow not only in
nervous signals themselves or because of a subsequent
that breast but also in the opposite breast. It is espe-
effect of increased prolactin—inhibit secretion of
cially interesting that fondling of the baby by the
gonadotropin-releasing hormone by the hypothala-
mother or hearing the baby crying often gives enough
mus. This, in turn, suppresses formation of the pituitary
of an emotional signal to the hypothalamus to cause
gonadotropic hormones—luteinizing hormone and
milk ejection.
follicle-stimulating hormone. However, after several
months of lactation, in some mothers, especially in
those who nurse their babies only some of the time,
Inhibition of Milk Ejection. A particular problem in
the pituitary begins to secrete sufficient gonadotropic
nursing a baby comes from the fact that many psy-
hormones to reinstate the monthly sexual cycle, even
chogenic factors or even generalized sympathetic
though nursing continues.
nervous system stimulation throughout the mother’s
Chapter 82
Pregnancy and Lactation
1041
especially lethal to bacteria that could cause deadly
Table 82-1
infections in newborn babies. Particularly important
Composition of Milk
are antibodies and macrophages that destroy
Escherichia coli bacteria, which often cause lethal diar-
Constituent
Human Milk (%)
Cow’s Milk (%)
rhea in newborns.
When cow’s milk is used to supply nutrition for the
Water
88.5
87.0
Fat
3.3
3.5
baby in place of mother’s milk, the protective agents
Lactose
6.8
4.8
in it are usually of little value because they are
Casein
0.9
2.7
normally destroyed within minutes in the internal
Lactalbumin and other
0.4
0.7
environment of the human being.
proteins
Ash
0.2
0.7
References
Alexander BT, Bennett WA, Khalil RA, Granger JP:
Preeclampsia: linking placental ischemia with cardiovas-
body can inhibit oxytocin secretion and consequently
cular-renal dysfunction. News Physiol Sci 16:282, 2001.
depress milk ejection. For this reason, many mothers
Ben-Jonathan N, Hnasko R: Dopamine as a prolactin (PRL)
must have an undisturbed puerperium if they are to be
inhibitor. Endocr Rev 22:724, 2001.
successful in nursing their babies.
Casey ML, MacDonald PC: The endocrinology of human
parturition. Ann N Y Acad Sci 828:273, 1997.
Challis JR, Lye SJ, Gibb W, et al: Understanding preterm
Milk Composition and the
labor. Ann N Y Acad Sci 943:225, 2001.
Metabolic Drain on the Mother
Cross JC, Simmons DG, Watson ED: Chorioallantoic mor-
phogenesis and formation of the placental villous tree.
Caused by Lactation
Ann N Y Acad Sci 995:84, 2003.
Davison JM, Homuth V, Jeyabalan A, et al: New aspects in
Table 82-1 lists the contents of human milk and cow’s
the pathophysiology of preeclampsia. J Am Soc Nephrol
milk. The concentration of lactose in human milk is
15:2440, 2004.
about 50 per cent greater than in cow’s milk, but the
Dekker G, Sibai B: Primary, secondary, and tertiary preven-
concentration of protein in cow’s milk is ordinarily two
tion of pre-eclampsia. Lancet 357:209, 2001.
or more times greater than in human milk. Finally, only
Freeman ME, Kanyicska B, Lerant A, Nagy G: Prolactin:
one third as much ash, which contains calcium and
structure, function, and regulation of secretion. Physiol
Rev 80:1523, 2000.
other minerals, is found in human milk compared with
Gimpl G, Fahrenholz F: The oxytocin receptor system: struc-
cow’s milk.
ture, function, and regulation. Physiol Rev 81:629, 2001.
At the height of lactation in the human mother, 1.5
Goffin V, Binart N, Touraine P, Kelly PA: Prolactin: the new
liters of milk may be formed each day (and even more
biology of an old hormone. Annu Rev Physiol 64:47, 2002.
if the mother has twins). With this degree of lactation,
Hall JG: Twinning. Lancet 362:735, 2003.
great quantities of metabolic substrates are drained
Kanaka-Gantenbein C, Mastorakos G, Chrousos GP:
from the mother. For instance, about 50 grams of fat
Endocrine-related causes and consequences of intrauter-
enter the milk each day, and about 100 grams of
ine growth retardation. Ann N Y Acad Sci 997:150, 2003.
lactose, which must be derived by conversion from the
Khalaf Y: ABC of subfertility: tubal subfertility. BMJ
mother’s glucose. Also, 2 to 3 grams of calcium phos-
327:610, 2003.
Khalil RA, Granger JP: Vascular mechanisms of increased
phate may be lost each day; unless the mother is drink-
arterial pressure in preeclampsia: lessons from animal
ing large quantities of milk and has an adequate intake
models. Am J Physiol Regul Integr Comp Physiol 283:R29,
of vitamin D, the output of calcium and phosphate by
2002.
the lactating mammae will often be much greater than
Labbok MH, Clark D, Goldman AS: Breastfeeding: main-
the intake of these substances. To supply the needed
taining an irreplaceable immunological resource. Nat Rev
calcium and phosphate, the parathyroid glands enlarge
Immunol 4:565, 2004.
greatly, and the bones become progressively decalci-
MacLaughlin DT, Donahoe PK: Sex determination and
fied. The mother’s bone decalcification is usually not a
differentiation. N Engl J Med 350:367, 2004.
big problem during pregnancy, but it can become more
Mastorakos G, Ilias I: Maternal and fetal hypothalamic-
important during lactation.
pituitary-adrenal axes during pregnancy and postpartum.
Ann N Y Acad Sci 997:136, 2003.
Moffett-King A: Natural killer cells and pregnancy. Nat Rev
Antibodies and Other Anti-infectious Agents in Milk. Not only
Immunol 2:656, 2002.
does milk provide the newborn baby with needed
Roberts JM, Cooper DW: Pathogenesis and genetics of
nutrients, but it also provides important protection
pre-eclampsia. Lancet 357:53, 2001.
against infection. For instance, multiple types of anti-
Roberts JM, Pearson G, Cutler J, Lindheimer M: Summary
bodies and other anti-infectious agents are secreted in
of the NHLBI Working Group on Research on Hyper-
milk along with the nutrients. Also, several different
tension During Pregnancy. Hypertension 41:437, 2003.
types of white blood cells are secreted, including both
Wu G, Bazer FW, Cudd TA, et al: Maternal nutrition and
neutrophils and macrophages, some of which are
fetal development. J Nutr 134:2169, 2004.
C H A P T E R
8
3
Fetal and Neonatal Physiology
A complete discussion of fetal development, function-
ing of the child immediately after birth, and growth and
development through the early years of life lies within
the province of formal courses in obstetrics and pedi-
atrics. However, many physiologic principles are pecu-
liar to the infant itself. This chapter discusses the more
important of these.
Growth and Functional Development
of the Fetus
Initial development of the placenta and fetal membranes occurs far more rapidly
than development of the fetus itself. In fact, during the first 2 to 3 weeks after
implantation of the blastocyst, the fetus remains almost microscopic, but thereafter,
as shown in Figure 83-1, the length of the fetus increases almost in proportion to
age. At 12 weeks, the length is about 10 centimeters; at 20 weeks, 25 centimeters;
and at term (40 weeks), 53 centimeters (about 21 inches). Because the weight of the
fetus is approximately proportional to the cube of length, the weight increases
almost in proportion to the cube of the age of the fetus.
Note in Figure 83-1 that the weight remains minuscule during the first 12 weeks
and reaches 1 pound only at 23 weeks (5 1/2 months) of gestation. Then, during the
last trimester of pregnancy, the fetus gains tremendously, so that 2 months before
birth, the weight averages 3 pounds, 1 month before birth 4.5 pounds, and at birth
7 pounds—the final birth weight varying from as low as 4.5 pounds to as high as 11
pounds in normal infants with normal gestational periods.
Development of the Organ Systems
Within 1 month after fertilization of the ovum, the gross characteristics of all the
different organs of the fetus have already begun to develop, and during the next 2
to 3 months, most of the details of the different organs are established. Beyond
month 4, the organs of the fetus are grossly the same as those of the neonate.
However, cellular development in each organ is usually far from complete and
requires the full remaining 5 months of pregnancy for complete development. Even
at birth, certain structures, particularly in the nervous system, the kidneys, and the
liver, lack full development, as discussed in more detail later in the chapter.
Circulatory System. The human heart begins beating during the fourth week after fer-
tilization, contracting at a rate of about 65 beats/min. This increases steadily to about
140 beats/min immediately before birth.
Formation of Blood Cells. Nucleated red blood cells begin to be formed in the yolk sac
and mesothelial layers of the placenta at about the third week of fetal development.
This is followed 1 week later (at 4 to 5 weeks) by formation of non-nucleated red
blood cells by the fetal mesenchyme and also by the endothelium of the fetal blood
vessels. Then, at 6 weeks, the liver begins to form blood cells, and in the third month,
the spleen and other lymphoid tissues of the body begin forming blood cells. Finally,
from the third month on, the bone marrow gradually becomes the principal source
of the red blood cells as well as most of the white blood cells, except for continued
lymphocyte and plasma cell production in lymphoid tissue.
Respiratory System. Respiration cannot occur during fetal life because there is no air
to breathe in the amniotic cavity. However, attempted respiratory movements do
take place beginning at the end of the first trimester of pregnancy. Tactile stimuli
and fetal asphyxia especially cause these attempted respiratory movements.
1042
Chapter 83
Fetal and Neonatal Physiology
1043
Length
25
Iron
250
50
Weight
Calcium
3
Phosphorus
20
200
40
2
30
15
150
20
10
100
1
10
5
50
0
0
0
4
8
12
16
20
24
28
32
36
40
0
0
Age of fetus (weeks after last menstruation)
0
4
8
12
16
20
24
28
32
36
40
Age of fetus (weeks after last menstruation)
Figure 83-1
Figure 83-2
Growth of the fetus.
Iron, calcium, and phosphorus storage in the fetus at different
stages of gestation.
During the last 3 to 4 months of pregnancy, the res-
piratory movements of the fetus are mainly inhibited,
for reasons unknown, and the lungs remain almost com-
and do not reach full development until a few months
pletely deflated. The inhibition of respiration during the
after birth.
later months of fetal life prevents filling of the lungs
with fluid and debris from the meconium excreted by
Fetal Metabolism. The fetus uses mainly glucose for
the fetus’s gastrointestinal tract into the amniotic fluid.
energy, and it has a high capability to store fat and
Also, small amounts of fluid are secreted into the lungs
protein, much if not most of the fat being synthesized
by the alveolar epithelium up until the moment of birth,
from glucose rather than being absorbed directly from
thus keeping only clean fluid in the lungs.
the mother’s blood. In addition to these generalities,
there are special problems of fetal metabolism in rela-
Nervous System. Most of the reflexes of the fetus that
tion to calcium, phosphate, iron, and some vitamins.
involve the spinal cord and even the brain stem are
present by the third to fourth months of pregnancy.
Metabolism of Calcium and Phosphate. Figure 83-2 shows
However, those nervous system functions that involve
the rates of calcium and phosphate accumulation in the
the cerebral cortex are still only in the early stages of
fetus, demonstrating that about 22.5 grams of calcium
development even at birth. Indeed, myelinization of
and 13.5 grams of phosphorus are accumulated in the
some major tracts of the brain itself becomes complete
average fetus during gestation. About one half of these
only after about 1 year of postnatal life.
accumulate during the last 4 weeks of gestation, which
is coincident with the period of rapid ossification of the
Gastrointestinal. Tract By midpregnancy, the fetus begins
fetal bones and with the period of rapid weight gain of
to ingest and absorb large quantities of amniotic fluid,
the fetus.
and during the last
2 to
3 months, gastrointestinal
During the earlier part of fetal life, the bones are
function approaches that of the normal neonate. By
relatively unossified and have mainly a cartilaginous
that time, small quantities of meconium are continually
matrix. Indeed x-ray films ordinarily do not show any
formed in the gastrointestinal tract and excreted from
ossification until after the fourth month of pregnancy.
the anus into the amniotic fluid. Meconium is composed
Note especially that the total amounts of calcium and
partly of residue from swallowed amniotic fluid and
phosphate needed by the fetus during gestation repre-
partly of mucus and other residues of excretory prod-
sent only about 2 per cent of the quantities of these
ucts from the gastrointestinal mucosa and glands.
substances in the mother’s bones. Therefore, this is a
minimal drain from the mother. Much greater drain
Kidneys. The fetal kidneys begin to excrete urine during
occurs after birth during lactation.
the second trimester pregnancy, and fetal urine accounts
for about 70 to 80 per cent of the amniotic fluid. Abnor-
Accumulation of Iron. Figure 83-2 also shows that iron
mal kidney development or severe impairment of
accumulates in the fetus even more rapidly than calcium
kidney function in the fetus greatly reduce the forma-
and phosphate. Most of the iron is in the form of
tion of amniotic fluid (oligohydramnios) and can lead to
hemoglobin, which begins to be formed as early as the
fetal death.
third week after fertilization of the ovum.
Although the fetal kidneys form urine, the renal
Small amounts of iron are concentrated in the
control systems for regulating fetal extracellular fluid
mother’s uterine progestational endometrium even
volume and electrolyte balances, and especially acid-
before implantation of the ovum; this iron is ingested
base balance, are almost nonexistent until late fetal life
into the embryo by the trophoblastic cells and is used
1044
Unit XIV Endocrinology and Reproduction
to form the very early red blood cells. About one third
progressively more hypoxic and hypercapnic, which
of the iron in a fully developed fetus is normally stored
provides additional stimulus to the respiratory center
in the liver. This iron can then be used for several
and usually causes breathing within an additional
months after birth by the neonate for formation of addi-
minute after birth.
tional hemoglobin.
Delayed or Abnormal Breathing at Birth—Danger of Hypoxia. If
Utilization and Storage of Vitamins. The fetus needs vitamins
the mother has been depressed by a general anesthetic
equally as much as the adult and in some instances to a
during delivery, which at least partially anesthetizes
far greater extent. In general, the vitamins function the
the fetus as well, the onset of respiration is likely to be
same in the fetus as in the adult, as discussed in Chapter
delayed for several minutes, thus demonstrating the
71. Special functions of several vitamins should be
importance of using as little anesthesia as feasible. Also,
mentioned, however.
many infants who have had head trauma during deliv-
The B vitamins, especially vitamin B12 and folic acid,
ery or who undergo prolonged delivery are slow to
are necessary for formation of red blood cells and
breathe or sometimes do not breathe at all. This can
nervous tissue, as well as for overall growth of the fetus.
result from two possible effects: First, in a few infants,
Vitamin C is necessary for appropriate formation of
intracranial hemorrhage or brain contusion causes a
intercellular substances, especially the bone matrix and
concussion syndrome with a greatly depressed respira-
fibers of connective tissue.
tory center. Second, and probably much more impor-
Vitamin D is needed for normal bone growth in the
tant, prolonged fetal hypoxia during delivery can cause
fetus, but even more important, the mother needs it for
serious depression of the respiratory center.
adequate absorption of calcium from her gastrointesti-
Hypoxia frequently occurs during delivery because of
nal tract. If the mother has plenty of this vitamin in her
(1) compression of the umbilical cord; (2) premature
body fluids, large quantities of the vitamin will be stored
separation of the placenta; (3) excessive contraction of
by the fetal liver to be used by the neonate for several
the uterus, which can cut off the mother’s blood flow to
months after birth.
the placenta; or (4) excessive anesthesia of the mother,
Vitamin E, although the mechanisms of its functions
which depresses oxygenation even of her blood.
are not clear, is necessary for normal development of
the early embryo. In its absence in laboratory animals,
Degree of Hypoxia That an Infant Can Tolerate. In an adult,
spontaneous abortion usually occurs at an early stage of
failure to breathe for only 4 minutes often causes death,
pregnancy.
but a neonate often survives as long as 10 minutes of
Vitamin K is used by the fetal liver for formation of
failure to breathe after birth. Permanent and very
Factor VII, prothrombin, and several other blood coag-
serious brain impairment often ensues if breathing is
ulation factors. When vitamin K is insufficient in the
delayed more than 8 to 10 minutes. Indeed, actual
mother, Factor VII and prothrombin become deficient
lesions develop mainly in the thalamus, in the inferior
in the fetus as well as in the mother. Because most
colliculi, and in other brain stem areas, thus permanently
vitamin K is formed by bacterial action in the mother’s
affecting many of the motor functions of the body.
colon, the neonate has no adequate source of vitamin K
for the first week or so of life after birth until normal
Expansion of the Lungs at Birth. At birth, the walls of the
colonic bacterial flora become established in the
alveoli are at first collapsed because of the surface
newborn infant. Therefore, prenatal storage in the fetal
tension of the viscid fluid that fills them. More than
liver of at least small amounts of vitamin K derived from
25 mm Hg of negative inspiratory pressure in the lungs
the mother is helpful in preventing fetal hemorrhage,
is usually required to oppose the effects of this surface
particularly hemorrhage in the brain when the head is
tension and to open the alveoli for the first time. But
traumatized by squeezing through the birth canal.
once the alveoli do open, further respiration can be
effected with relatively weak respiratory movements.
Fortunately, the first inspirations of the normal neonate
Adjustments of the Infant
are extremely powerful, usually capable of creating as
much as 60 mm Hg negative pressure in the intrapleural
to Extrauterine Life
space.
Figure
83-3 shows the tremendous negative
Onset of Breathing
intrapleural pressures required to open the lungs at
The most obvious effect of birth on the baby is loss of
the onset of breathing. At the top is shown the pres-
the placental connection with the mother and, there-
sure-volume curve (“compliance” curve) for the first
fore, loss of this means of metabolic support. One of the
breath after birth. Observe, first, the lower part of
most important immediate adjustments required of the
the curve beginning at the zero pressure point and
infant is to begin breathing.
moving to the right. The curve shows that the volume of
air in the lungs remains almost exactly zero until the
Cause of Breathing at Birth. After normal delivery from a
negative pressure has reached -40 centimeters water
mother who has not been depressed by anesthetics, the
(-30 mm Hg). Then, as the negative pressure increases
child ordinarily begins to breathe within seconds and
to -60 centimeters of water, about 40 milliliters of air
has a normal respiratory rhythm within less than 1
enters the lungs. To deflate the lungs, considerable pos-
minute after birth. The promptness with which the fetus
itive pressure, about
+40 centimeters of water, is
begins to breathe indicates that breathing is initiated by
required because of viscous resistance offered by the
sudden exposure to the exterior world, probably result-
fluid in the bronchioles.
ing from (1) a slightly asphyxiated state incident to the
Note that the second breath is much easier, with far
birth process, but also from (2) sensory impulses that
less negative and positive pressures required. Breathing
originate in the suddenly cooled skin. In an infant who
does not become completely normal until about 40
does not breathe immediately, the body becomes
minutes after birth, as shown by the third compliance
Chapter 83
Fetal and Neonatal Physiology
1045
One of the most characteristic findings in respiratory
distress syndrome is failure of the respiratory epithe-
First Breath
lium to secrete adequate quantities of surfactant, a sub-
60
stance normally secreted into the alveoli that decreases
the surface tension of the alveolar fluid, therefore allow-
ing the alveoli to open easily during inspiration. The sur-
40
factant-secreting cells (type II alveolar epithelial cells)
do not begin to secrete surfactant until the last 1 to 3
20
months of gestation. Therefore, many premature babies
and a few full-term babies are born without the capa-
bility to secrete sufficient surfactant, which causes both
0
a collapse tendency of the alveoli and development of
+40
+20
0
-20
-40
-60
pulmonary edema. The role of surfactant in preventing
Pressure
these effects is discussed in Chapter 37.
Second Breath
60
Circulatory Readjustments at Birth
Equally as essential as the onset of breathing at birth
40
are immediate circulatory adjustments that allow ade-
quate blood flow through the lungs. Also, circulatory
20
adjustments during the first few hours of life cause more
and more blood flow through the baby’s liver, which up
to this point has had very little blood flow. To describe
0
these readjustments, we must first consider the anatom-
+40
+20
0
-20
-40
-60
Pressure
ical structure of the fetal circulation.
40 Minutes
Specific Anatomical Structure of the Fetal Circulation. Because
60
the lungs are mainly nonfunctional during fetal life and
because the liver is only partially functional, it is not
necessary for the fetal heart to pump much blood
40
through either the lungs or the liver. However, the fetal
heart must pump large quantities of blood through the
placenta. Therefore, special anatomical arrangements
20
cause the fetal circulatory system to operate much
differently from that of the newborn baby.
0
First, as shown in Figure 83-4, blood returning from
+40
+20
0
-20
-40
-60
the placenta through the umbilical vein passes through
Pressure
the ductus venosus, mainly bypassing the liver. Then
most of the blood entering the right atrium from the
inferior vena cava is directed in a straight pathway
across the posterior aspect of the right atrium and
Figure 83-3
through the foramen ovale directly into the left atrium.
Thus, the well-oxygenated blood from the placenta
Pressure-volume curves of the lungs (“compliance” curves) of a
enters mainly the left side of the heart, rather than the
neonate immediately after birth, showing the extreme forces
right side, and is pumped by the left ventricle mainly
required for breathing during the first two breaths of life and devel-
opment of a nearly normal compliance curve within 40 minutes
into the arteries of the head and forelimbs.
after birth.
(Redrawn from Smith CA: The first breath. Sci Am
The blood entering the right atrium from the supe-
209:32,
1963, © 1963 by Scientific American, Inc. All rights
rior vena cava is directed downward through the tri-
reserved.)
cuspid valve into the right ventricle. This blood is mainly
deoxygenated blood from the head region of the fetus,
and it is pumped by the right ventricle into the pul-
curve, the shape of which compares favorably with that
monary artery and then mainly through the ductus arte-
for the normal adult, as shown in Chapter 38.
riosus into the descending aorta, then through the two
umbilical arteries into the placenta, where the deoxy-
Respiratory Distress Syndrome Caused When Surfactant Secretion
genated blood becomes oxygenated.
Is Deficient. A small number of infants, especially pre-
Figure 83-5 gives the relative percentages of the total
mature infants and infants born of diabetic mothers,
blood pumped by the heart that pass through the dif-
develop severe respiratory distress in the early hours to
ferent vascular circuits of the fetus. This figure shows
the first several days after birth, and some die within the
that 55 per cent of all the blood goes through the pla-
next day or so. The alveoli of these infants at death
centa, leaving only 45 per cent to pass through all the
contain large quantities of proteinaceous fluid, almost
tissues of the fetus. Furthermore, during fetal life, only
as if pure plasma had leaked out of the capillaries into
12 per cent of the blood flows through the lungs; imme-
the alveoli. The fluid also contains desquamated alveo-
diately after birth, virtually all the blood flows through
lar epithelial cells. This condition is called hyaline mem-
the lungs.
brane disease because microscopic slides of the lung
show the material filling the alveoli to look like a
Changes in the Fetal Circulation at Birth. The basic changes
hyaline membrane.
in the fetal circulation at birth are discussed in
1046
Unit XIV Endocrinology and Reproduction
Superior
vena cava
Aorta
Ductus arteriosus
15
Forequarters
43
Ductus
arteriosus
15
Lung
42
Lung
30
Right
Right
atrium
ventricle
27
Lungs
58
Foramen ovale
73
Left
Left
Ductus arteriosus
atrium
ventricle
Liver
46
73
Foramen
12
ovale
Hindquarters
Gut
18
Umbilical
vein
Placenta
55
Umbilical
arteries
Figure 83-5
Figure 83-4
Diagram of the fetal circulatory system, showing relative distribu-
tion of blood flow to the different vascular areas. The numerals
Organization of
the
fetal
circulation.
(Modified from Arey LB:
represent the percentage of the total output from both sides of the
Developmental Anatomy: A Textbook and Laboratory Manual of
heart flowing through each particular area.
Embryology. 7th ed. Philadelphia: WB Saunders Co, 1974.)
Chapter 23 in relation to congenital anomalies of the
and systemic resistances at birth cause blood now to
ductus arteriosus and foramen ovale that persist
attempt to flow backward through the foramen ovale;
throughout life in a few persons. Briefly, these changes
that is, from the left atrium into the right atrium, rather
are the following.
than in the other direction, as occurred during fetal life.
Consequently, the small valve that lies over the foramen
Primary Changes in Pulmonary and Systemic Vascular
ovale on the left side of the atrial septum closes over
Resistances at Birth
this opening, thereby preventing further flow through
The primary changes in the circulation at birth are, first,
the foramen ovale.
loss of the tremendous blood flow through the placenta,
In two thirds of all people, the valve becomes adher-
which approximately doubles the systemic vascular
ent over the foramen ovale within a few months to a few
resistance at birth. This increases the aortic pressure as
years and forms a permanent closure. But even if per-
well as the pressures in the left ventricle and left atrium.
manent closure does not occur, the left atrial pressure
Second, the pulmonary vascular resistance greatly
throughout life normally remains 2 to 4 mm Hg greater
decreases as a result of expansion of the lungs. In the
than the right atrial pressure, and the backpressure
unexpanded fetal lungs, the blood vessels are com-
keeps the valve closed.
pressed because of the small volume of the lungs. Imme-
diately on expansion, these vessels are no longer
Closure of the Ductus Arteriosus
compressed and the resistance to blood flow decreases
The ductus arteriosus also closes, but for different
severalfold. Also, in fetal life, the hypoxia of the lungs
reasons. First, the increased systemic resistance elevates
causes considerable tonic vasoconstriction of the lung
the aortic pressure while the decreased pulmonary
blood vessels, but vasodilation takes place when aera-
resistance reduces the pulmonary arterial pressure. As a
tion of the lungs eliminates the hypoxia. All these
consequence, after birth, blood begins to flow backward
changes together reduce the resistance to blood flow
from the aorta into the pulmonary artery through the
through the lungs as much as fivefold, which reduces the
ductus arteriosus, rather than in the other direction as
pulmonary arterial pressure, right ventricular pressure,
in fetal life. However, after only a few hours, the muscle
and right atrial pressure.
wall of the ductus arteriosus constricts markedly, and
Closure of the Foramen Ovale
within 1 to 8 days, the constriction is usually sufficient
The low right atrial pressure and the high left atrial pres-
to stop all blood flow. This is called functional closure
sure that occur secondarily to the changes in pulmonary
of the ductus arteriosus. Then, during the next 1 to
Chapter 83
Fetal and Neonatal Physiology
1047
4 months, the ductus arteriosus ordinarily becomes
systems. This results partly from immature development
anatomically occluded by growth of fibrous tissue into
of the different organs of the body and partly from the
its lumen.
fact that the control systems simply have not become
The cause of ductus arteriosus closure relates to the
adjusted to the new way of life.
increased oxygenation of the blood flowing through the
ductus. In fetal life the PO2 of the ductus blood is only
15 to 20 mm Hg, but it increases to about 100 mm Hg
Respiratory System
within a few hours after birth. Furthermore, many
The normal rate of respiration in a neonate is about 40
experiments have shown that the degree of contraction
breaths per minute, and tidal air with each breath aver-
of the smooth muscle in the ductus wall is highly related
ages 16 milliliters. This gives a total minute respiratory
to this availability of oxygen.
volume of 640 ml/min, which is about twice as great in
In one of several thousand infants, the ductus fails to
relation to the body weight as that of an adult. The func-
close, resulting in a patent ductus arteriosus, the conse-
tional residual capacity of the infant’s lungs is only one
quences of which are discussed in Chapter 23. The
half that of an adult in relation to body weight. This
failure of closure has been postulated to result from
difference causes excessive cyclical increases and
excessive ductus dilation caused by vasodilating
decreases in the newborn baby’s blood gas concentra-
prostaglandins in the ductus wall. In fact, administration
tions if the respiratory rate becomes slowed because it
of the drug indomethacin, which blocks synthesis of
is the residual air in the lungs that smooths out the
prostaglandins, often leads to closure.
blood gas variations.
Closure of the Ductus Venosus In fetal life, the portal blood
from the fetus’s abdomen joins the blood from the
umbilical vein, and these together pass by way of the
Circulation
ductus venosus directly into the vena cava immediately
Blood Volume. The blood volume of a neonate immedi-
below the heart but above the liver, thus bypassing the
ately after birth averages about 300 milliliters, but if the
liver.
infant is left attached to the placenta for a few minutes
Immediately after birth, blood flow through the
after birth or if the umbilical cord is stripped to force
umbilical vein ceases, but most of the portal blood
blood out of its vessels into the baby, an additional 75
still flows through the ductus venosus, with only a small
milliliters of blood enters the infant, to make a total of
amount passing through the channels of the liver.
375 milliliters. Then, during the ensuing few hours, fluid
However, within 1 to 3 hours the muscle wall of the
is lost into the neonate’s tissue spaces from this blood,
ductus venosus contracts strongly and closes this avenue
which increases the hematocrit but returns the blood
of flow. As a consequence, the portal venous pressure
volume once again to the normal value of about 300 mil-
rises from near 0 to 6 to 10 mm Hg, which is enough to
liliters. Some pediatricians believe that this extra blood
force portal venous blood flow through the liver sinuses.
volume caused by stripping the umbilical cord can lead
Although the ductus venosus rarely fails to close, we
to mild pulmonary edema with some degree of respira-
know almost nothing about what causes the closure.
tory distress, but the extra red blood cells are often very
valuable to the infant.
Nutrition of the Neonate
Cardiac Output. The cardiac output of the neonate
Before birth, the fetus derives almost all its energy from
averages 500 ml/min, which, like respiration and body
glucose obtained from the mother’s blood. After birth,
metabolism, is about twice as much in relation to body
the amount of glucose stored in the infant’s body in the
weight as in the adult. Occasionally a child is born with
form of liver and muscle glycogen is sufficient to supply
an especially low cardiac output caused by hemorrhage
the infant’s needs for only a few hours. The liver of the
of much of its blood volume from the placenta at birth.
neonate is still far from functionally adequate at birth,
which prevents significant gluconeogenesis. Therefore,
Arterial Pressure. The arterial pressure during the first
the infant’s blood glucose concentration frequently falls
day after birth averages about 70 mm Hg systolic and
the first day to as low as 30 to 40 mg/dl of plasma, less
50 mm Hg diastolic; this increases slowly during the
than one half the normal value. Fortunately, however,
next several months to about 90/60. Then there is a
appropriate mechanisms are available for the infant to
much slower rise during the subsequent years until the
use its stored fats and proteins for metabolism until
adult pressure of 115/70 is attained at adolescence.
mother’s milk can be provided 2 to 3 days later.
Special problems are also frequently associated with
Blood Characteristics. The red blood cell count in the
getting an adequate fluid supply to the neonate because
neonate averages about 4 million per cubic millimeter.
the infant’s rate of body fluid turnover averages seven
If blood is stripped from the cord into the infant, the red
times that of an adult, and the mother’s milk supply
blood cell count rises an additional 0.5 to 0.75 million
requires several days to develop. Ordinarily, the infant’s
during the first few hours of life, giving a red blood cell
weight decreases 5 to 10 per cent and sometimes as
count of about 4.75 million per cubic millimeter, as
much as 20 per cent within the first 2 to 3 days of life.
shown in Figure 83-6. Subsequent to this, however, few
Most of this weight loss is loss of fluid rather than of
new red blood cells are formed in the infant during the
body solids.
first few weeks of life, presumably because the hypoxic
stimulus of fetal life is no longer present to stimulate
Special Functional Problems
red cell production. Thus, as shown in Figure 83-6, the
average red blood cell count falls to less than 4 million
in the Neonate
per cubic millimeter by about 6 to 8 weeks of age. From
An important characteristic of the neonate is instability
that time on, increasing activity by the baby provides
of the various hormonal and neurogenic control
the appropriate stimulus for returning the red blood cell
1048
Unit XIV Endocrinology and Reproduction
Fluid Balance, Acid-Base Balance,
and Renal Function
5
5
The rate of fluid intake and fluid excretion in the
newborn infant is seven times as great in relation to
4
4
Red blood count
weight as in the adult, which means that even a slight
percentage alteration of fluid intake or fluid output can
cause rapidly developing abnormalities.
3
3
The rate of metabolism in the infant is also twice as
great in relation to body mass as in the adult, which
2
2
means that twice as much acid is normally formed,
which gives a tendency toward acidosis in the infant.
1
1
Bilirubin
Functional development of the kidneys is not complete
until the end of about the first month of life. For
0
0
instance, the kidneys of the neonate can concentrate
0
2
4
6
8
10
12
14
16
urine to only 1.5 times the osmolality of the plasma
Age in weeks
instead of the adult three to four times. Therefore, con-
sidering the immaturity of the kidneys, together with the
marked fluid turnover in the infant and rapid formation
of acid, one can readily understand that among the most
Figure 83-6
important problems of infancy are acidosis, dehydra-
tion, and, more rarely, overhydration.
Changes in the red blood cell count and in serum bilirubin con-
centration during the first 16 weeks of life, showing physiologic
Liver Function
anemia at 6 to 12 weeks of life and physiologic hyperbilirubine-
mia during the first 2 weeks of life.
During the first few days of life, liver function in the
neonate may be quite deficient, as evidenced by the
following effects:
1. The liver of the neonate conjugates bilirubin with
glucuronic acid poorly and therefore excretes
bilirubin only slightly during the first few days of
life.
count to normal within another 2 to 3 months. Immedi-
2. The liver of the neonate is deficient in forming
ately after birth, the white blood cell count of the
plasma proteins, so that the plasma protein
neonate is about 45,000 per cubic millimeter, which is
concentration falls during the first weeks of life to
about five times as great as that of the normal adult.
15 to 20 per cent less than that for older children.
Occasionally the protein concentration falls so low
Neonatal Jaundice and Erythroblastosis Fetalis. Biliru-
that the infant develops hypoproteinemic edema.
bin formed in the fetus can cross the placenta into
3. The gluconeogenesis function of the liver is
the mother and be excreted through the liver of the
particularly deficient. As a result, the blood glucose
mother, but immediately after birth, the only means
level of the unfed neonate falls to about 30 to 40
for ridding the neonate of bilirubin is through the
mg/dl (about 40 per cent of normal), and the infant
neonate’s own liver, which for the first week or so of life
must depend mainly on its stored fats for energy
functions poorly and is incapable of conjugating signi-
until sufficient feeding can occur.
ficant quantities of bilirubin with glucuronic acid for
4. The liver of the neonate usually also forms too
excretion into the bile. Consequently, the plasma biliru-
little of the blood factors needed for normal blood
bin concentration rises from a normal value of less than
coagulation.
1 mg/dl to an average of 5 mg/dl during the first 3 days
of life and then gradually falls back to normal as the
liver becomes functional. This effect, called physiologic
Digestion, Absorption, and
hyperbilirubinemia, is shown in Figure
83-6, and it
Metabolism of Energy Foods;
is associated with mild jaundice (yellowness) of the
and Nutrition
infant’s skin and especially of the sclerae of its eyes for
a week or two.
In general, the ability of the neonate to digest, absorb,
However, by far the most important abnormal cause
and metabolize foods is no different from that of the
of serious neonatal jaundice is erythroblastosis fetalis,
older child, with the following three exceptions.
which is discussed in detail in Chapter 32 in relation
First, secretion of pancreatic amylase in the neonate
to Rh factor incompatibility between the fetus and
is deficient, so that the neonate uses starches less ade-
mother. Briefly, the erythroblastotic baby inherits Rh-
quately than do older children.
positive red cells from the father, while the mother is
Second, absorption of fats from the gastrointestinal
Rh negative. The mother then becomes immunized
tract is somewhat less than that in the older child. Con-
against the Rh-positive factor (a protein) in the fetus’s
sequently, milk with a high fat content, such as cow’s
blood cells, and her antibodies destroy fetal red cells,
milk, is frequently inadequately absorbed.
releasing extreme quantities of bilirubin into the fetus’s
Third, because the liver functions imperfectly during
plasma and often causing fetal death for lack of ade-
at least the first week of life, the glucose concentration
quate red cells. Before the advent of modern obstetri-
in the blood is unstable and low.
cal therapeutics, this condition occurred either mildly or
The neonate is especially capable of synthesizing and
seriously in 1 of every 50 to 100 neonates.
storing proteins. Indeed, with an adequate diet, as much
Chapter 83
Fetal and Neonatal Physiology
1049
Necessity for Iron in the Diet
If the mother has had adequate amounts of iron in her
99
diet, the liver of the infant usually has stored enough
Birth
iron to keep forming blood cells for 4 to 6 months after
98
birth. But if the mother has had insufficient iron in her
diet, severe anemia is likely to occur in the infant after
97
about 3 months of life. To prevent this possibility, early
feeding of the infant with egg yolk, which contains rea-
96
sonably large quantities of iron, or the administration of
iron in some other form is desirable by the second or
95
third month of life.
94
Vitamin C Deficiency in Infants
93
Ascorbic acid (vitamin C) is not stored in significant
0
2
4
6
8 1012 2
4
6
8 1012 14 16 18 20
quantities in the fetal tissues; yet it is required for
proper formation of cartilage, bone, and other intercel-
Hours after birth
Days after birth
lular structures of the infant. Furthermore, milk has
poor supplies of ascorbic acid, especially cow’s milk,
which has only one fourth as much as human milk. For
this reason, orange juice or other sources of ascorbic
Figure 83-7
acid are often prescribed by the third week of life.
Fall in body temperature of the neonate immediately after birth,
and instability of body temperature during the first few days of life.
Immunity
The neonate inherits much immunity from the mother
because many protein antibodies diffuse from the
mother’s blood through the placenta into the fetus.
as 90 per cent of the ingested amino acids are used for
However, the neonate does not form antibodies of its
formation of body proteins. This is a much higher per-
own to a significant extent. By the end of the first month,
centage than in adults.
the baby’s gamma globulins, which contain the anti-
bodies, have decreased to less than one half the original
Metabolic Rate and Body Temperature. The normal meta-
level, with a corresponding decrease in immunity. There-
bolic rate of the neonate in relation to body weight is
after, the baby’s own immunity system begins to form
about twice that of the adult, which accounts also for
antibodies, and the gamma globulin concentration
the twice as great cardiac output and twice as great
returns essentially to normal by the age of 12 to 20
minute respiratory volume in relation to body weight in
months.
the infant.
Despite the decrease in gamma globulins soon after
Because the body surface area is large in relation to
birth, the antibodies inherited from the mother protect
body mass, heat is readily lost from the body. As a result,
the infant for about 6 months against most major child-
the body temperature of the neonate, particularly of
hood infectious diseases, including diphtheria, measles,
premature infants, falls easily. Figure 83-7 shows that
and polio. Therefore, immunization against these
the body temperature of even a normal infant often falls
diseases before
6 months is usually unnecessary.
several degrees during the first few hours after birth but
Conversely, the inherited antibodies against whooping
returns to normal in 7 to 10 hours. Still, the body tem-
cough are normally insufficient to protect the neonate;
perature regulatory mechanisms remain poor during
therefore, for full safety, the infant requires immuniza-
the early days of life, allowing marked deviations in
tion against this disease within the first month or so of
temperature, which are also shown in Figure 83-7.
life.
Nutritional Needs During the Early Weeks of Life. At birth, a
Allergy. The newborn infant is seldom subject to allergy.
neonate is usually in complete nutritional balance, pro-
Several months later, however, when the infant’s own
vided the mother has had an adequate diet. Further-
antibodies first begin to form, extreme allergic states
more, function of the gastrointestinal system is usually
can develop, often resulting in serious eczema, gas-
more than adequate to digest and assimilate all the
trointestinal abnormalities, and even anaphylaxis. As
nutritional needs of the infant if appropriate nutrients
the child grows older and still higher degrees of immu-
are provided in the diet. However, three specific prob-
nity develop, these allergic manifestations usually dis-
lems do occur in the early nutrition of the infant.
appear. This relation of immunity to allergy is discussed
in Chapter 34.
Need for Calcium and Vitamin D
The neonate is in a stage of rapid ossification of its
bones at birth, so that a ready supply of calcium
Endocrine Problems
throughout infancy is needed. This is ordinarily supplied
adequately by the usual diet of milk. Yet absorption of
Ordinarily, the endocrine system of the infant is highly
calcium by the gastrointestinal tract is poor in the
developed at birth, and the infant seldom exhibits any
absence of vitamin D. Therefore, the vitamin D-
immediate endocrine abnormalities. However, there are
deficient infant can develop severe rickets in only a few
special instances in which the endocrinology of infancy
weeks. This is particularly true in premature babies
is important:
because their gastrointestinal tracts absorb calcium
1. If a pregnant mother bearing a female child is
even less effectively than those of normal infants.
treated with an androgenic hormone or if an
1050
Unit XIV Endocrinology and Reproduction
androgenic tumor develops during pregnancy,
under the following two headings: (1) immaturity of
the child will be born with a high degree of
certain organ systems and (2) instability of the different
masculinization of her sexual organs, thus resulting
homeostatic control systems. Because of these effects, a
in a type of hermaphroditism.
premature baby seldom lives if it is born more than 3
2.
The sex hormones secreted by the placenta and by
months before term.
the mother’s glands during pregnancy occasionally
cause the neonate’s breasts to form milk
during the first days of life. Sometimes the breasts
Immature Development of the
then become inflamed, or infectious mastitis
Premature Infant
develops.
3.
An infant born of an untreated diabetic mother will
Almost all the organ systems of the body are immature
have considerable hypertrophy and hyperfunction
in the premature infant, but some require particular
of the islets of Langerhans in the pancreas.
attention if the life of the premature baby is to be saved.
As a consequence, the infant’s blood glucose
concentration may fall to lower than 20 mg/dl
Respiration. The respiratory system is especially likely to
shortly after birth. Fortunately, however, in the
be underdeveloped in the premature infant. The vital
neonate, unlike in the adult, insulin shock or coma
capacity and the functional residual capacity of the
from this low level of blood glucose concentration
lungs are especially small in relation to the size of the
only rarely develops.
infant. Also, surfactant secretion is depressed or absent.
Maternal type II diabetes is the most common
As a consequence, respiratory distress syndrome is a
cause of large babies. Type II diabetes in the
common cause of death. Also, the low functional resid-
mother is associated with resistance to the
ual capacity in the premature infant is often associated
metabolic effects of insulin and compensatory
with periodic breathing of the Cheyne-Stokes type.
increases in plasma insulin concentration. The high
levels of insulin are believed to stimulate fetal
Gastrointestinal Function. Another major problem of the
growth factor and contribute to increased birth
premature infant is to ingest and absorb adequate food.
weight. Increased supply of glucose and other
If the infant is more than 2 months premature, the
nutrients to the fetus may also contribute to
digestive and absorptive systems are almost always
increased fetal growth. However, most of the
inadequate. The absorption of fat is so poor that the pre-
increased fetal weight is due to increased body fat;
mature infant must have a low-fat diet. Furthermore,
there is usually little increase in body length
the premature infant has unusual difficulty in absorbing
although the size of some organs may be increased
calcium and, therefore, can develop severe rickets
(organomegaly).
before the difficulty is recognized. For this reason,
In the mother with uncontrolled type I diabetes
special attention must be paid to adequate calcium and
(caused by lack of insulin secretion), fetal growth
vitamin D intake.
may be stunted because of metabolic deficits in the
mother, and growth and tissue maturation of the
Function of Other Organs. Immaturity of other organ
neonate are often impaired. Also, there is a high
systems that frequently causes serious difficulties in the
rate of intrauterine mortality, and among those
premature infant includes (1) immaturity of the liver,
fetuses that do come to term, there is still a high
which results in poor intermediary metabolism and
mortality rate. Two thirds of the infants who die
often a bleeding tendency as a result of poor formation
succumb to respiratory distress syndrome, described
of coagulation factors; (2) immaturity of the kidneys,
earlier in the chapter.
which are particularly deficient in their ability to rid the
4.
Occasionally a child is born with hypofunctional
body of acids, thereby predisposing to acidosis as well
adrenal cortices, often resulting from agenesis of
as to serious fluid balance abnormalities; (3) immaturity
the adrenal glands or exhaustion atrophy, which can
of the blood-forming mechanism of the bone marrow,
occur when the adrenal glands have been vastly
which allows rapid development of anemia; and (4)
overstimulated.
depressed formation of gamma globulin by the lym-
5.
If a pregnant woman has hyperthyroidism or is
phoid system, which often leads to serious infection.
treated with excess thyroid hormone, the infant is
likely to be born with a temporarily hyposecreting
thyroid gland. Conversely, if before pregnancy a
Instability of the Homeostatic
woman had had her thyroid gland removed, her
Control Systems in the
pituitary gland may secrete great quantities of
Premature Infant
thyrotropin during gestation, and the child might be
born with temporary hyperthyroidism.
Immaturity of the different organ systems in the pre-
6.
In a fetus lacking thyroid hormone secretion, the
mature infant creates a high degree of instability in the
bones grow poorly and there is mental retardation.
homeostatic mechanisms of the body. For instance, the
This causes the condition called cretin dwarfism,
acid-base balance can vary tremendously, particularly
discussed in Chapter 76.
when the rate of food intake varies from time to time.
Likewise, the blood protein concentration is usually low
because of immature liver development, often leading
Special Problems
to hypoproteinemic edema. And inability of the infant
to regulate its calcium ion concentration frequently
of Prematurity
brings on hypocalcemic tetany. Also, the blood glucose
All the problems in neonatal life just noted are severely
concentration can vary between the extremely wide
exacerbated in prematurity. They can be categorized
limits of 20 to more than 100 mg/dl, depending princi-
Chapter 83
Fetal and Neonatal Physiology
1051
pally on the regularity of feeding. It is no wonder, then,
with these extreme variations in the internal environ-
ment of the premature infant, that mortality is high.
70
Instability of Body Temperature. One of the particular prob-
lems of the premature infant is inability to maintain
60
normal body temperature. Its temperature tends to
approach that of its surroundings. At normal room
temperature, the infant’s temperature may stabilize in
the low 90°s or even in the 80°s F. Statistical studies
50
show that a body temperature maintained below 96°F
(35.5°C) is associated with a particularly high incidence
of death, which explains the almost mandatory use of
40
the incubator in treatment of prematurity.
Boys
Girls
30
Danger of Blindness Caused
by Excess Oxygen Therapy
in the Premature Infant
20
0
4
8 12 16 20 24 4
8
12
16
20
Because premature infants frequently develop respira-
tory distress, oxygen therapy has often been used in
Age in months
Age in years
treating prematurity. However, it has been discovered
that use of excess oxygen in treating premature infants,
especially in early prematurity, can lead to blindness.
The reason is that too much oxygen stops the growth of
Figure 83-8
new blood vessels in the retina. Then when oxygen
Average height of boys and girls from infancy to 20 years of age.
therapy is stopped, the blood vessels try to make up for
lost time and burst forth with a great mass of vessels
growing all through the vitreous humor, blocking light
from the pupil to the retina. And still later, the vessels
are replaced with a mass of fibrous tissue where the
eye’s clear vitreous humor should be.
This condition, known as retrolental fibroplasia,
causes permanent blindness. For this reason, it is par-
12
Walks alone
ticularly important to avoid treatment of premature
infants with high concentrations of respiratory oxygen.
11
Stands alone
Physiologic studies indicate that the premature infant is
10
usually safe with up to 40 per cent oxygen in the air
Walks with support
breathed, but some child physiologists believe that com-
9
Pulls up
plete safety can be achieved only at normal oxygen con-
centration in the air that is breathed.
8
Grasps
Crawls
7
Growth and Development
Sits briefly
6
of the Child
5
Rolls over
The major physiologic problems of the child beyond the
neonatal period are related to special metabolic needs
4
for growth, which have been fully covered in the sec-
Hand control
tions of this book on metabolism and endocrinology.
3
Head control
Figure 83-8 shows the changes in heights of boys and
2
girls from the time of birth until the age of 20 years.
Vocalizes
Note especially that these parallel each other almost
1
Smiles
exactly until the end of the first decade of life. Between
the ages of 11 and 13 years, the female estrogens begin
0
Suckles
to be formed and cause rapid growth in height but early
Birth
uniting of the epiphyses of the long bones at about the
14th to 16th year of life, so that growth in height then
ceases. This contrasts with the effect of testosterone in
the male, which causes extra growth at a slightly later
Figure 83-9
age—mainly between ages 13 and 17 years. The male,
Behavioral development of the infant during the first year of life.
however, undergoes much more prolonged growth
because of much delayed uniting of the epiphyses, so
that his final height is considerably greater than that of
the female.
1052
Unit XIV Endocrinology and Reproduction
Behavioral Growth
Hilaire G, Duron B: Maturation of the mammalian respira-
tory system. Physiol Rev 79:325, 1999.
Behavioral growth is principally a problem of maturity
Holemans K, Aerts L, Van Assche FA: Lifetime conse-
of the nervous system. It is extremely difficult to
quences of abnormal fetal pancreatic development. J
dissociate maturity of the anatomical structures of the
Physiol 547:11, 2003.
nervous system from maturity caused by training.
Jensen BL, Stubbe J, Madsen K, et al: The renin-angiotensin
Anatomical studies show that certain major tracts in the
system in kidney development: role of COX-2 and adrenal
central nervous system are not completely myelinated
steroids. Acta Physiol Scand 181:549, 2004.
until the end of the first year of life. For this reason,
Johnson MH: Functional brain development in humans. Nat
it is frequently stated that the nervous system is not
Rev Neurosci 2:475, 2001.
fully functional at birth. The brain cortex and its associ-
Kanwar YS, Wada J, Lin S, Danesh FR, et al: Update of extra-
ated functions, such as vision, seem to require several
cellular matrix, its receptors, and cell adhesion molecules
months after birth for final functional development to
in mammalian nephrogenesis. Am J Physiol Renal Physiol
occur.
286:F202, 2004.
At birth, the infant brain mass is only 26 per cent of
Kovacs CS, Kronenberg HM: Maternal-fetal calcium and
the adult brain mass and 55 per cent at 1 year, but it
bone metabolism during pregnancy, puerperium, and
reaches almost adult proportions by the end of the
lactation. Endocr Rev 18:832, 1997.
second year. This is also associated with closure of the
Labbok MH, Clark D, Goldman AS: Breastfeeding: main-
fontanels and sutures of the skull, which allows only 20
taining an irreplaceable immunological resource. Nat Rev
per cent additional growth of the brain beyond the first
Immunol 4:565, 2004.
2 years of life. Figure 83-9 shows a normal progress
MacLellan WR, Schneider MD: Genetic dissection of
chart for the infant during the first year of life. Com-
cardiac growth control pathways. Annu Rev Physiol
parison of this chart with the baby’s actual development
62:289, 2000.
is used for clinical assessment of mental and behavioral
Matias A, Montenegro N, Areias JC, Leite LP: Haemody-
growth.
namic evaluation of the first trimester fetus with special
emphasis on venous return. Hum Reprod Update 6:177,
2000.
References
McMurtry IF: Pre- and postnatal lung development, matu-
ration, and plasticity. Am J Physiol Lung Cell Mol Physiol
Bissonnette JM: Mechanisms regulating hypoxic respiratory
282:L341, 2002.
depression during fetal and postnatal life. Am J Physiol
Olver RE, Walters DV, M Wilson S: Developmental regula-
Regul Integr Comp Physiol 278:R1391, 2000.
tion of lung liquid transport. Annu Rev Physiol 66:77,
Chen Y, Lasaitiene D, Friberg P: The renin-angiotensin
2004.
system in kidney development. Acta Physiol Scand
Ross MG, Nijland MJ: Development of ingestive behavior.
181:529, 2004.
Am J Physiol 274:R879, 1998.
Challis JRG, Matthews SG, Gibb W, Lye SJ: Endocrine and
Shannon JM, Hyatt BA: Epithelial-mesenchymal interac-
paracrine regulation of birth at term and preterm. Endocr
tions in the developing lung. Annu Rev Physiol 66:625,
Rev 21:514, 2000.
2004.
de Castro F: Chemotropic molecules: guides for axonal
Srivastava D: Genetic assembly of the heart: implications for
pathfinding and cell migration during CNS development.
congenital heart disease. Annu Rev Physiol 63:451, 2001.
News Physiol Sci 18:130, 2003.
Wood CE, Tong H: Central nervous system regulation of
Dunne MJ, Cosgrove KE, Shepherd RM, et al: Hyperin-
reflex responses to hypotension during fetal life. Am J
sulinism in infancy: from basic science to clinical disease.
Physiol 277:R1541, 1999.
Physiol Rev 84:239, 2004.
U N I T XV
Sports Physiology
84. Sports Physiology
C H A P T E R
8
4
Sports Physiology
There are few stresses to which the body is exposed that
even nearly approach the extreme stresses of heavy
exercise. In fact, if some of the extremes of exercise
were continued for even moderately prolonged periods,
they might be lethal. Therefore, in the main, sports
physiology is a discussion of the ultimate limits to which
several of the bodily mechanisms can be stressed. To
give one simple example: In a person who has
extremely high fever approaching the level of lethality,
the body metabolism increases to about 100 per cent above normal. By compari-
son, the metabolism of the body during a marathon race may increase to 2000 per
cent above normal.
Female and Male Athletes Most of the quantitative data that are given in this chapter
are for the young male athlete, not because it is desirable to know only these values
but because it is only in male athletes that relatively complete measurements have
been made. However, for those measurements that have been made in the female
athlete, almost identical basic physiologic principles apply, except for quantitative
differences caused by differences in body size, body composition, and the presence
or absence of the male sex hormone testosterone.
In general, most quantitative values for women—such as muscle strength, pul-
monary ventilation, and cardiac output, all of which are related mainly to the muscle
mass—vary between two thirds and three quarters of the values recorded in men.
When measured in terms of strength per square centimeter of cross-sectional area,
the female muscle can achieve almost exactly the same maximal force of contrac-
tion as that of the male-between 3 and 4 kg/cm2. Therefore, most of the difference
in total muscle performance lies in the extra percentage of the male body that is
muscle, caused by endocrine differences that we discuss later.
The performance capabilities of the female versus the male athlete are illustrated
by the relative running speeds for a marathon race. In a recent comparison, the top
female performer had a running speed that was 11 per cent less than that of the top
male performer. For other events, however, women have at times held records faster
than men—for instance, for the two-way swim across the English Channel, where
the availability of extra fat seems to be an advantage for heat insulation, buoyancy,
and extra long-term energy.
Testosterone secreted by the male testes has a powerful anabolic effect in causing
greatly increased deposition of protein everywhere in the body, but especially in the
muscles. In fact, even a male who participates in very little sports activity but who
nevertheless is well endowed with testosterone will have muscles that grow about
40 per cent larger than those of a comparable female without the testosterone.
The female sex hormone estrogen probably also accounts for some of the differ-
ence between female and male performance, although not nearly so much as testos-
terone. Estrogen is known to increase the deposition of fat in the female, especially
in the breasts, hips, and subcutaneous tissue. At least partly for this reason, the
average nonathletic female has about 27 per cent body fat composition, in contrast
to the nonathletic male, who has about 15 per cent. This is a detriment to the highest
levels of athletic performance in those events in which performance depends on
speed or on ratio of total body muscle strength to body weight.
Muscles in Exercise
Strength, Power, and Endurance of Muscles
The final common determinant of success in athletic events is what the muscles can
do for you—what strength they can give when it is needed, what power they can
achieve in the performance of work, and how long they can continue their activity.
1055
1056
Unit XV Sports Physiology
The strength of a muscle is determined mainly by its
is for the next 30 minutes, because the efficiency for
size, with a maximal contractile force between 3 and
translation of muscle power output into athletic per-
4 kg/cm2 of muscle cross-sectional area. Thus, a man who
formance is often much less during rapid activity than
is well supplied with testosterone or who has enlarged
during less rapid but sustained activity. Thus, the veloc-
his muscles through an exercise training program will
ity of the 100-meter dash is only 1.75 times as great as
have correspondingly increased muscle strength.
the velocity of a 30-minute race, despite the fourfold dif-
To give an example of muscle strength, a world-class
ference in short-term versus long-term muscle power
weight lifter might have a quadriceps muscle with a
capability.
cross-sectional area as great as 150 square centimeters.
Another measure of muscle performance is
This would translate into a maximal contractile strength
endurance. This, to a great extent, depends on the nutri-
of 525 kilograms (or 1155 pounds), with all this force
tive support for the muscle—more than anything else
applied to the patellar tendon. Therefore, one can
on the amount of glycogen that has been stored in the
readily understand how it is possible for this tendon at
muscle before the period of exercise. A person on a
times to be ruptured or actually to be avulsed from its
high-carbohydrate diet stores far more glycogen in
insertion into the tibia below the knee. Also, when such
muscles than a person on either a mixed diet or a high-
forces occur in tendons that span a joint, similar forces
fat diet. Therefore, endurance is greatly enhanced by a
are applied to the surfaces of the joint or sometimes to
high-carbohydrate diet. When athletes run at speeds
ligaments spanning the joints, thus accounting for such
typical for the marathon race, their endurance (as meas-
happenings as displaced cartilages, compression frac-
ured by the time that they can sustain the race until
tures about the joint, and torn ligaments.
complete exhaustion) is approximately the following:
The holding strength of muscles is about 40 per cent
greater than the contractile strength. That is, if a muscle
is already contracted and a force then attempts to
Minutes
stretch out the muscle, as occurs when landing after a
High-carbohydrate diet
240
jump, this requires about 40 per cent more force than
Mixed diet
120
can be achieved by a shortening contraction. Therefore,
High-fat diet
85
the force of 525 kilograms calculated above for the
patellar tendon during muscle contraction becomes 735
kilograms (1617 pounds) during holding contractions.
This further compounds the problems of the tendons,
The corresponding amounts of glycogen stored in the
joints, and ligaments. It can also lead to internal tearing
muscle before the race started explain these differences.
in the muscle itself. In fact, forceful stretching of a max-
The amounts stored are approximately the following:
imally contracted muscle is one of the surest ways to
create the highest degree of muscle soreness.
Mechanical work performed by a muscle is the
g/kg Muscle
amount of force applied by the muscle multiplied by the
High-carbohydrate diet
40
distance over which the force is applied. The power of
Mixed diet
20
muscle contraction is different from muscle strength,
High-fat diet
6
because power is a measure of the total amount of
work that the muscle performs in a unit period of time.
Power is therefore determined not only by the strength
of muscle contraction but also by its distance of
Muscle Metabolic Systems
contraction and the number of times that it contracts
in Exercise
each minute. Muscle power is generally measured in
kilogram meters (kg-m) per minute. That is, a muscle
The same basic metabolic systems are present in muscle
that can lift 1 kilogram weight to a height of 1 meter or
as in other parts of the body; these are discussed in
that can move some object laterally against a force of 1
detail in Chapters
67 through 73. However, special
kilogram for a distance of 1 meter in 1 minute is said to
quantitative measures of the activities of three meta-
have a power of 1 kg-m/min. The maximal power
bolic systems are exceedingly important in understand-
achievable by all the muscles in the body of a highly
ing the limits of physical activity. These systems are (1)
trained athlete with all the muscles working together is
the phosphocreatine-creatine system, (2) the glycogen-
approximately the following:
lactic acid system, and (3) the aerobic system.
Adenosine Triphosphate. The source of energy actually
kg-m/min
used to cause muscle contraction is adenosine triphos-
phate (ATP), which has the following basic formula:
First 8 to 10 seconds
7000
Next 1 minute
4000
-
Adenosine-PO3 ~ PO3 ~ PO3
Next 30 minutes
1700
The bonds attaching the last two phosphate radicals
to the molecule, designated by the symbol ~, are high-
Thus, it is clear that a person has the capability of
energy phosphate bonds. Each of these bonds stores
extreme power surges for short periods of time, such as
7300 calories of energy per mole of ATP under standard
during a 100-meter dash that is completed entirely
conditions (and even slightly more than this under the
within 10 seconds, whereas for long-term endurance
physical conditions in the body, which is discussed in
events, the power output of the muscles is only one
detail in Chapter 67). Therefore, when one phosphate
fourth as great as during the initial power surge.
radical is removed, more than 7300 calories of energy
This does not mean that one’s athletic performance is
are released to energize the muscle contractile process.
four times as great during the initial power surge as it
Then, when the second phosphate radical is removed,
Chapter 84
Sports Physiology
1057
I. Phosphocreatine
Creatine + PO3-
ATP
Energy
II. Glycogen
Lactic acid
ADP
for muscle
contraction
III. Glucose
AMP
Fatty acids
+ O2
CO2 + H2O
Figure 84-1
Amino acids
+
Urea
Important metabolic systems
that supply energy for muscle
contraction.
still another 7300 calories become available. Removal
glycolysis, occurs without use of oxygen and, therefore,
of the first phosphate converts the ATP into adenosine
is said to be anaerobic metabolism (see Chapter 67).
diphosphate (ADP), and removal of the second converts
During glycolysis, each glucose molecule is split into two
this ADP into adenosine monophosphate (AMP).
pyruvic acid molecules, and energy is released to form
The amount of ATP present in the muscles, even in a
four ATP molecules for each original glucose molecule,
well-trained athlete, is sufficient to sustain maximal
as explained in Chapter 67. Ordinarily, the pyruvic
muscle power for only about 3 seconds, maybe enough
acid then enters the mitochondria of the muscle cells
for one half of a 50-meter dash. Therefore, except for a
and reacts with oxygen to form still many more
few seconds at a time, it is essential that new ATP be
ATP molecules. However, when there is insufficient
formed continuously, even during the performance of
oxygen for this second stage (the oxidative stage) of
short athletic events. Figure
84-1 shows the overall
glucose metabolism to occur, most of the pyruvic acid
metabolic system, demonstrating the breakdown of
then is converted into lactic acid, which diffuses out of
ATP first to ADP and then to AMP, with the release of
the muscle cells into the interstitial fluid and blood.
energy to the muscles for contraction. The left-hand side
Therefore, much of the muscle glycogen is transformed
of the figure shows the three metabolic systems that
to lactic acid, but in doing so, considerable amounts of
provide a continuous supply of ATP in the muscle fibers.
ATP are formed entirely without the consumption of
oxygen.
Another characteristic of the glycogen-lactic acid
Phosphocreatine-Creatine System
system is that it can form ATP molecules about 2.5
times as rapidly as can the oxidative mechanism of the
Phosphocreatine
(also called creatine phosphate) is
mitochondria. Therefore, when large amounts of ATP
another chemical compound that has a high-energy
are required for short to moderate periods of muscle
phosphate bond, with the following formula:
contraction, this anaerobic glycolysis mechanism can be
Creatine ~ PO3
-
used as a rapid source of energy. It is, however, only
about one half as rapid as the phosphagen system.
This can decompose to creatine and phosphate ion, as
Under optimal conditions, the glycogen-lactic acid
shown to the left in Figure 84-1, and in doing so release
system can provide 1.3 to
1.6 minutes of maximal
large amounts of energy. In fact, the high-energy phos-
muscle activity in addition to the 8 to 10 seconds pro-
phate bond of phosphocreatine has more energy than
vided by the phosphagen system, although at somewhat
the bond of ATP, 10,300 calories per mole in compari-
reduced muscle power.
son with 7300. Therefore, phosphocreatine can easily
provide enough energy to reconstitute the high-energy
Aerobic System. The aerobic system is the oxidation of
bond of ATP. Furthermore, most muscle cells have two
foodstuffs in the mitochondria to provide energy. That
to four times as much phosphocreatine as ATP.
is, as shown to the left in Figure 84-1, glucose, fatty acids,
A special characteristic of energy transfer from phos-
and amino acids from the foodstuffs—after some inter-
phocreatine to ATP is that it occurs within a small frac-
mediate processing—combine with oxygen to release
tion of a second. Therefore, all the energy stored in the
tremendous amounts of energy that are used to convert
muscle phosphocreatine is almost instantaneously avail-
AMP and ADP into ATP, as discussed in Chapter 67.
able for muscle contraction, just as is the energy stored
In comparing this aerobic mechanism of energy
in ATP.
supply with the glycogen-lactic acid system and the
The combined amounts of cell ATP and cell phos-
phosphagen system, the relative maximal rates of power
phocreatine are called the phosphagen energy system.
generation in terms of moles of ATP generation per
These together can provide maximal muscle power for
minute are the following:
8 to 10 seconds, almost enough for the 100-meter run.
Thus, the energy from the phosphagen system is used for
Moles of ATP/min
maximal short bursts of muscle power.
Phosphagen system
4
Glycogen-Lactic Acid System. The stored glycogen in
Glycogen-lactic acid system
2.5
muscle can be split into glucose and the glucose then
Aerobic system
1
used for energy. The initial stage of this process, called
1058
Unit XV
Sports Physiology
When comparing the same systems for endurance, the
oxidative metabolism of the aerobic system can be used
relative values are the following:
to reconstitute all the other systems-the ATP, the phos-
phocreatine, and the glycogen-lactic acid system.
Reconstitution of the lactic acid system means mainly
Time
the removal of the excess lactic acid that has accumu-
lated in all the fluids of the body. This is especially
Phosphagen system
8 to 10 seconds
Glycogen-lactic acid system
1.3 to 1.6 minutes
important because lactic acid causes extreme fatigue.
Aerobic system
Unlimited time (as long as
When adequate amounts of energy are available from
nutrients last)
oxidative metabolism, removal of lactic acid is achieved
in two ways: (1) A small portion of it is converted back
into pyruvic acid and then metabolized oxidatively by
all the body tissues. (2) The remaining lactic acid is
Thus, one can readily see that the phosphagen system
reconverted into glucose mainly in the liver, and the
is the one used by the muscle for power surges of a few
glucose in turn is used to replenish the glycogen stores
seconds, and the aerobic system is required for pro-
of the muscles.
longed athletic activity. In between is the glycogen-lactic
acid system, which is especially important for giving
Recovery of the Aerobic System After Exercise. Even during
extra power during such intermediate races as the 200-
the early stages of heavy exercise, a portion of one’s
to 800-meter runs.
aerobic energy capability is depleted. This results from
two effects: (1) the so-called oxygen debt and (2) deple-
What Types of Sports Use Which Energy Systems? By consid-
tion of the glycogen stores of the muscles.
ering the vigor of a sports activity and its duration, one
can estimate closely which of the energy systems is used
Oxygen Debt. The body normally contains about 2 liters
for each activity. Various approximations are presented
of stored oxygen that can be used for aerobic metabo-
in Table 84-1.
lism even without breathing any new oxygen. This
stored oxygen consists of the following: (1) 0.5 liter in
Recovery of the Muscle Metabolic Systems After Exercise. In
the air of the lungs, (2) 0.25 liter dissolved in the body
the same way that the energy from phosphocreatine can
fluids, (3) 1 liter combined with the hemoglobin of the
be used to reconstitute ATP, energy from the glycogen-
blood, and (4) 0.3 liter stored in the muscle fibers them-
lactic acid system can be used to reconstitute both phos-
selves, combined mainly with myoglobin, an oxygen-
phocreatine and ATP. And then energy
from
the
binding chemical similar to hemoglobin.
In heavy exercise, almost all this stored oxygen is used
within a minute or so for aerobic metabolism. Then,
Table 84-1
after the exercise is over, this stored oxygen must be
replenished by breathing extra amounts of oxygen over
Energy Systems Used in Various Sports
and above the normal requirements. In addition, about
9 liters more oxygen must be consumed to provide for
reconstituting both the phosphagen system and the
Phosphagen system, almost entirely
100-meter dash
lactic acid system. All this extra oxygen that must be
Jumping
“repaid,” about 11.5 liters, is called the oxygen debt.
Weight lifting
Figure 84-2 shows this principle of oxygen debt.
Diving
During the first 4 minutes of the figure, the person exer-
Football dashes
cises heavily, and the rate of oxygen uptake increases
Phosphagen and glycogen-lactic acid systems
200-meter dash
Basketball
Baseball home run
5
Ice hockey dashes
Glycogen-lactic acid system, mainly
4
400-meter dash
100-meter swim
Tennis
3
Soccer
Alactacid oxygen debt = 3.5 liters
2
Glycogen-lactic acid and aerobic systems
800-meter dash
200-meter swim
Lactic acid oxygen debt = 8 liters
1
1500-meter skating
Boxing
2000-meter rowing
0
1500-meter run
0
4
8
12
16
20
24
28
32
36
40
44
1-mile run
Minutes
400-meter swim
Aerobic system
10,000-meter skating
Figure 84-2
Cross-country skiing
Marathon run (26.2 miles, 42.2 km)
Rate of oxygen uptake by the lungs during maximal exercise for
Jogging
4 minutes and then for about 40 minutes after the exercise is over.
This figure demonstrates the principle of oxygen debt.
Chapter 84
Sports Physiology
1059
2 hours of
exercise
24
20
High-carbohydrate diet
No food
16
Fat and protein diet
12
8
4
Figure 84-3
Effect of diet on the rate of muscle
0
glycogen replenishment after pro-
0
10
20
30
40
50
longed exercise. (Redrawn from
Hours of recovery
5 days
Fox EL: Sports Physiology.
Philadelphia: Saunders College
Publishing, 1979.)
more than 15-fold. Then, even after the exercise is over,
the oxygen uptake still remains above normal, at first
very high while the body is reconstituting the phospha-
High-carbohydrate diet
gen system and repaying the stored oxygen portion of
Mixed diet
the oxygen debt, and then for another 40 minutes at a
100
High-fat diet
0
lower level while the lactic acid is removed. The early
portion of the oxygen debt is called the alactacid oxygen
debt and amounts to about 3.5 liters. The latter portion
75
25
is called the lactic acid oxygen debt and amounts to
about 8 liters.
50
50
Recovery of Muscle Glycogen. Recovery from exhaustive
muscle glycogen depletion is not a simple matter. This
often requires days, rather than the seconds, minutes, or
25
Exhaustion
75
hours required for recovery of the phosphagen and
lactic acid metabolic systems. Figure 84-3 shows this
recovery process under three conditions: first, in people
0
100
on a high-carbohydrate diet; second, in people on a
0
10
20
40
2 4
1
2
3
4
high-fat, high-protein diet; and third, in people with no
Seconds
Minutes
Hours
food. Note that on a high-carbohydrate diet, full recov-
Duration of exercise
ery occurs in about 2 days. Conversely, people on a high-
fat, high-protein diet or on no food at all show very little
recovery even after as long as 5 days. The messages of
this comparison are (1) that it is important for an athlete
Figure 84-4
to have a high-carbohydrate diet before a grueling ath-
letic event and (2) not to participate in exhaustive exer-
Effect of duration of exercise as well as type of diet on relative
cise during the 48 hours preceding the event.
percentages of carbohydrate or fat used for energy by muscles.
(Based partly on data in Fox EL: Sports Physiology. Philadelphia:
Saunders College Publishing, 1979.)
Nutrients Used During
Muscle Activity
longer than 4 to 5 hours, the glycogen stores of the
In addition to the large usage of carbohydrates by the
muscle become almost totally depleted and are of little
muscles during exercise, especially during the early
further use for energizing muscle contraction. Instead,
stages of exercise, muscles use large amounts of fat for
the muscle now depends on energy from other sources,
energy in the form of fatty acids and acetoacetic acid (see
mainly from fats.
Chapter 68), and they use to a much less extent proteins
Figure 84-4 shows the approximate relative usage of
in the form of amino acids. In fact, even under the best
carbohydrates and fat for energy during prolonged
conditions, in those endurance athletic events that last
exhaustive exercise under three dietary conditions:
1060
Unit XV
Sports Physiology
high-carbohydrate diet, mixed diet, and high-fat diet.
muscle strength increases about 30 per cent during the
Note that most of the energy is derived from carbohy-
first 6 to 8 weeks but almost plateaus after that time.
drates during the first few seconds or minutes of the
Along with this increase in strength is an approximately
exercise, but at the time of exhaustion, as much as 60 to
equal percentage increase in muscle mass, which is
85 per cent of the energy is being derived from fats,
called muscle hypertrophy.
rather than carbohydrates.
In old age, many people become so sedentary that
Not all the energy from carbohydrates comes from
their muscles atrophy tremendously. In these instances,
the stored muscle glycogen. In fact, almost as much
muscle training often increases muscle strength more
glycogen is stored in the liver as in the muscles, and this
than 100 per cent.
can be released into the blood in the form of glucose
and then taken up by the muscles as an energy source.
Muscle Hypertrophy. The average size of a person’s
In addition, glucose solutions given to an athlete to
muscles is determined to a great extent by heredity plus
drink during the course of an athletic event can provide
the level of testosterone secretion, which, in men, causes
as much as 30 to 40 per cent of the energy required
considerably larger muscles than in women. With train-
during prolonged events such as marathon races.
ing, however, the muscles can become hypertrophied
Therefore, if muscle glycogen and blood glucose are
perhaps an additional 30 to 60 per cent. Most of this
available, they are the energy nutrients of choice for
hypertrophy results from increased diameter of the
intense muscle activity. Even so, for a long-term
muscle fibers rather than increased numbers of fibers,
endurance event, one can expect fat to supply more than
but this probably is not entirely true, because a very few
50 per cent of the required energy after about the first
greatly enlarged muscle fibers are believed to split down
3 to 4 hours.
the middle along their entire length to form entirely
new fibers, thus increasing the number of fibers slightly.
The changes that occur inside the hypertrophied
Effect of Athletic Training on
muscle fibers themselves include (1) increased numbers
Muscles and Muscle Performance
of myofibrils, proportionate to the degree of hypertro-
phy; (2) up to 120 per cent increase in mitochondrial
Importance of Maximal Resistance Training. One of the car-
enzymes; (3) as much as 60 to 80 per cent increase in
dinal principles of muscle development during athletic
the components of the phosphagen metabolic system,
training is the following: Muscles that function under no
including both ATP and phosphocreatine; (4) as much
load, even if they are exercised for hours on end,
as 50 per cent increase in stored glycogen; and (5) as
increase little in strength. At the other extreme, muscles
much as 75 to 100 per cent increase in stored triglyc-
that contract at more than 50 per cent maximal force of
eride (fat). Because of all these changes, the capabilities
contraction will develop strength rapidly even if the
of both the anaerobic and the aerobic metabolic systems
contractions are performed only a few times each day.
are increased, increasing especially the maximum oxi-
Using this principle, experiments on muscle building
dation rate and efficiency of the oxidative metabolic
have shown that six nearly maximal muscle contractions
system as much as 45 per cent.
performed in three sets 3 days a week give approxi-
mately optimal increase in muscle strength, without
Fast-Twitch and Slow-Twitch Muscle Fibers. In the human
producing chronic muscle fatigue.
being, all muscles have varying percentages of fast-
The upper curve in Figure 84-5 shows the approxi-
twitch and slow-twitch muscle fibers. For instance, the
mate percentage increase in strength that can be
gastrocnemius muscle has a higher preponderance of
achieved in a previously untrained young person by this
fast-twitch fibers, which gives it the capability of force-
resistive training program, demonstrating that the
ful and rapid contraction of the type used in jumping.
In contrast, the soleus muscle has a higher preponder-
ance of slow-twitch muscle fibers and therefore is used
to a greater extent for prolonged lower leg muscle
activity.
The basic differences between the fast-twitch and the
Resistive training
slow-twitch fibers are the following:
30
1. Fast-twitch fibers are about twice as large in
25
diameter.
2. The enzymes that promote rapid release of energy
20
from the phosphagen and glycogen-lactic acid
15
energy systems are two to three times as active in
fast-twitch fibers as in slow-twitch fibers, thus
10
making the maximal power that can be achieved
5
for very short periods of time by fast-twitch fibers
No-load
training
about twice as great as that of slow-twitch fibers.
0
3. Slow-twitch fibers are mainly organized for
endurance, especially for generation of aerobic
0
2
4
6
8
10
energy. They have far more mitochondria than
Weeks of training
the fast-twitch fibers. In addition, they contain
considerably more myoglobin, a hemoglobin-like
protein that combines with oxygen within the
muscle fiber; the extra myoglobin increases the rate
Figure 84-5
of diffusion of oxygen throughout the fiber by
Approximate effect of optimal resistive exercise training on
shuttling oxygen from one molecule of myoglobin
increase in muscle strength over a training period of 10 weeks.
to the next. In addition, the enzymes of the aerobic
Chapter 84
Sports Physiology
1061
metabolic system are considerably more active in
slow-twitch fibers than in fast-twitch fibers.
4. The number of capillaries is greater in the vicinity
120
of slow-twitch fibers than in the vicinity of fast-
110
twitch fibers.
In summary, fast-twitch fibers can deliver extreme
100
amounts of power for a few seconds to a minute or
so. Conversely, slow-twitch fibers provide endurance,
80
delivering prolonged strength of contraction over many
60
minutes to hours.
40
Hereditary Differences Among Athletes for Fast-Twitch Versus
Slow-Twitch Muscle Fibers. Some people have considerably
20
Moderate
Severe
more fast-twitch than slow-twitch fibers, and others have
exercise
exercise
0
more slow-twitch fibers; this could determine to some
0
1.0
2.0
3.0
4.0
extent the athletic capabilities of different individuals.
O2 consumption (L/min)
Athletic training has not been shown to change the rel-
ative proportions of fast-twitch and slow-twitch fibers
however much an athlete might want to develop one
type of athletic prowess over another. Instead, this seems
Figure 84-6
to be determined almost entirely by genetic inheritance,
and this in turn helps determine which area of athletics
Effect of exercise on oxygen consumption and ventilatory rate.
is most suited to each person: some people appear to be
(Redrawn from Gray JS: Pulmonary Ventilation and Its Physiolog-
born to be marathoners; others are born to be sprinters
ical Regulation. Springfield, IL: Charles C Thomas, 1950.)
and jumpers. For example, the following are recorded
percentages of fast-twitch versus slow-twitch fiber in the
quadriceps muscles of different types of athletes:
Limits of Pulmonary Ventilation. How severely do we stress
our respiratory systems during exercise? This can be
answered by the following comparison for a normal
young man:
Fast-Twitch
Slow-Twitch
Marathoners
18
82
Swimmers
26
74
L/min
Average male
55
45
Weight lifters
55
45
Pulmonary ventilation at maximal exercise
100 to 110
Sprinters
63
37
Maximal breathing capacity
150 to 170
Jumpers
63
37
Thus, the maximal breathing capacity is about 50 per
Respiration in Exercise
cent greater than the actual pulmonary ventilation
during maximal exercise. This provides an element of
Although one’s respiratory ability is of relatively little
safety for athletes, giving them extra ventilation that can
concern in the performance of sprint types of athletics,
be called on in such conditions as (1) exercise at high
it is critical for maximal performance in endurance
altitudes, (2) exercise under very hot conditions, and (3)
athletics.
abnormalities in the respiratory system.
The important point is that the respiratory system is
Oxygen Consumption and Pulmonary Ventilation in Exercise.
not normally the most limiting factor in the delivery of
Normal oxygen consumption for a young man at rest is
oxygen to the muscles during maximal muscle aerobic
about 250 ml/min. However, under maximal conditions,
metabolism. We shall see shortly that the ability of the
this can be increased to approximately the following
heart to pump blood to the muscles is usually a greater
average levels:
limiting factor.
Effect of Training on VO2 Max. The abbreviation for the rate
ml/min
VO2 Max. Figure 84.7 shows the progressive effect of
Untrained average male
3600
athletic training on VO2 Max recorded in a group of sub-
Athletically trained average male
4000
jects beginning at the level of no training and then pur-
Male marathon runner
5100
suing the training program for.7 to 13 weeks. In this
study, it is surprising that the VO2 Max increased only
about 10 per cent. Furthermore, the frequency of train-
ing, whether two times or f.ve times per week, had little
Figure 84-6 shows the relation between oxygen con-
effect on th
sumption and total pulmonary ventilation at different
earlier, the VO2 Max of a marathoner is about 45 per
levels of exercise. It is clear from this figure, as would
cent greater
be expected, that there is a linear relation. Both oxygen
this greater VO2 Max of the marathoner probably is
consumption and total pulmonary ventilation increase
genetically determined; that is, those people who have
about 20-fold between the resting state and maximal
greater chest sizes in relation to body size and stronger
intensity of exercise in the well-trained athlete.
respiratory muscles select themselves to become
1062
Unit XV Sports Physiology
naturally greater diffusing capacities choose these types
of sports, or is it because something about the training
3.8
procedures increases the diffusing capacity? The answer
is not known, but it is very likely that training, particu-
3.6
larly endurance training, does play an important role.
3.4
Blood Gases During Exercise. Because of the great usage of
Training frequency
oxygen by the muscles in exercise, one would expect
3.2
= 5 days/wk
the oxygen pressure of the arterial blood to decrease
= 4 days/wk
markedly during strenuous athletics and the carbon
= 2 days/wk
dioxide pressure of the venous blood to increase far
3.0
above normal. However, this normally is not the case.
Both of these values remain nearly normal, demon-
2.8
strating the extreme ability of the respiratory system to
0
2
4
6
8
10
12
14
Weeks of training
provide adequate aeration of the blood even during
heavy exercise.
This demonstrates another important point: The
blood gases do not always have to become abnormal for
Figure 84-7
respiration to be stimulated in exercise. Instead, respira-
tion is stimulated mainly by neurogenic mechanisms
Max over a period of 7 to 13 weeks of athletic
during exercise, as discussed in Chapter 41. Part of this
Increase in VO2
training. (Redrawn from Fox EL: Sports Physiology. Philadelphia:
stimulation results from direct stimulation of the respi-
Saunders College Publishing, 1979.)
ratory center by the same nervous signals that are trans-
mitted from the brain to the muscles to cause the
exercise. An additional part is believed to result from
marathoners. However, it is also likely tha. many years
sensory signals transmitted into the respiratory center
of training increase the marathoner’s VO2
Max by
from the contracting muscles and moving joints. All this
values considerably greater than the 10 per cent that has
extra nervous stimulation of respiration is normally suf-
been recorded in short-term experiments such as that in
ficient to provide almost exactly the necessary increase
Figure 84-7.
in pulmonary ventilation required to keep the blood
respiratory gases—the oxygen and the carbon dioxide—
Oxygen Diffusing Capacity of Athletes. The oxygen diffusing
very near to normal.
capacity is a measure of the rate at which oxygen can
diffuse from the pulmonary alveoli into the blood. This
Effect of Smoking on Pulmonary Ventilation in Exercise. It is
is expressed in terms of milliliters of oxygen that will
widely known that smoking can decrease an athlete’s
diffuse each minute for each millimeter of mercury dif-
“wind.” This is true for many reasons. First, one effect
ference between alveolar partial pressure of oxygen and
of nicotine is constriction of the terminal bronchioles of
pulmonary blood oxygen pressure. That is, if the partial
the lungs, which increases the resistance of airflow into
pressure of oxygen in the alveoli is 91 mm Hg and the
and out of the lungs. Second, the irritating effects of the
oxygen pressure in the blood is 90 mm Hg, the amount
smoke itself cause increased fluid secretion into the
of oxygen that diffuses through the respiratory mem-
bronchial tree, as well as some swelling of the epithelial
brane each minute is equal to the diffusing capacity. The
linings. Third, nicotine paralyzes the cilia on the surfaces
following are measured values for different diffusing
of the respiratory epithelial cells that normally beat con-
capacities:
tinuously to remove excess fluids and foreign particles
from the respiratory passageways. As a result, much
debris accumulates in the passageways and adds further
ml/min
to the difficulty of breathing. Putting all these factors
together, even a light smoker often feels respiratory
Nonathlete at rest
23
strain during maximal exercise, and the level of per-
Nonathlete during maximal exercise
48
formance may be reduced.
Speed skaters during maximal exercise
64
Swimmers during maximal exercise
71
Much more severe are the effects of chronic smoking.
Oarsman during maximal exercise
80
There are few chronic smokers in whom some degree
of emphysema does not develop. In this disease, the fol-
lowing occur: (1) chronic bronchitis, (2) obstruction of
The most startling fact about these results is the sev-
many of the terminal bronchioles, and (3) destruction
eralfold increase in diffusing capacity between the
of many alveolar walls. In severe emphysema, as much
resting state and the state of maximal exercise. This
as four fifths of the respiratory membrane can be
results mainly from the fact that blood flow through
destroyed; then even the slightest exercise can cause
many of the pulmonary capillaries is sluggish or even
respiratory distress. In fact, many such patients cannot
dormant in the resting state, whereas in maximal exer-
even perform the simple feat of walking across the floor
cise, increased blood flow through the lungs causes
of a single room without gasping for breath.
all the pulmonary capillaries to be perfused at their
maximal rates, thus providing a far greater surface area
through which oxygen can diffuse into the pulmonary
Cardiovascular System
capillary blood.
in Exercise
It is also clear from these values that those athletes
who require greater amounts of oxygen per minute have
Muscle Blood Flow. A key requirement of cardiovascular
higher diffusing capacities. Is this because people with
function in exercise is to deliver the required oxygen
Chapter 84
Sports Physiology
1063
Therefore, a 30 per cent increase in blood pressure can
often more than double the blood flow; this multiplies
the great increase in flow already caused by the meta-
Rhythmic exercise
bolic vasodilation at least another twofold.
40
Work Output, Oxygen Consumption, and Cardiac Output During
Exercise. Figure 84-9 shows the interrelations among
work output, oxygen consumption, and cardiac output
during exercise. It is not surprising that all these are
directly related to one another, as shown by the linear
20
functions, because the muscle work output increases
oxygen consumption, and oxygen consumption in turn
dilates the muscle blood vessels, thus increasing venous
return and cardiac output. Typical cardiac outputs at
Calf
several levels of exercise are the following:
flow
0
10
16
18
Minutes
L/min
Cardiac output in young man at rest
5.5
Maximal cardiac output during exercise in young
23
untrained man
Figure 84-8
Maximal cardiac output during exercise in
30
average male marathoner
Effects of muscle exercise on blood flow in the calf of a leg dur-
ing strong rhythmical contraction. The blood flow was much less
during contraction than between contractions. (Redrawn from
Barcroft H, Dornhors AC: Blood flow through human calf during
rhythmic exercise. J Physiol 109:402, 1949.)
Thus, the normal untrained person can increase
cardiac output a little over fourfold, and the well-
trained athlete can increase output about sixfold.
and other nutrients to the exercising muscles. For this
(Individual marathoners have been clocked at cardiac
purpose, the muscle blood flow increases drastically
outputs as great as 35 to 40 L/min, seven to eight times
during exercise. Figure
84-8 shows a recording of
normal resting output.)
muscle blood flow in the calf of a person for a period of
6 minutes during moderately strong intermittent con-
Effect of Training on Heart Hypertrophy and on Cardiac Output.
tractions. Note not only the great increase in flow—
From the foregoing data, it is clear that marathoners can
about 13-fold—but also the flow decrease during each
achieve maximal cardiac outputs about 40 per cent
muscle contraction. Two points can be made from
greater than those achieved by untrained persons. This
this study:
(1) The actual contractile process itself
results mainly from the fact that the heart chambers of
temporarily decreases muscle blood flow because the
marathoners enlarge about 40 per cent; along with this
contracting skeletal muscle compresses the intramuscu-
enlargement of the chambers, the heart mass also
lar blood vessels; therefore, strong tonic muscle con-
increases 40 per cent or more. Therefore, not only do
tractions can cause rapid muscle fatigue because of lack
the skeletal muscles hypertrophy during athletic train-
of delivery of enough oxygen and other nutrients during
ing but the heart does also. However, heart enlargement
the continuous contraction.
(2) The blood flow to
and increased pumping capacity occur almost entirely
muscles during exercise increases markedly. The fol-
in the endurance types, not in the sprint types, of ath-
lowing comparison shows the maximal increase in blood
letic training.
flow that can occur in a well-trained athlete.
Even though the heart of the marathoner is consid-
erably larger than that of the normal person, resting
cardiac output is almost exactly the same as that in the
ml/100 g Muscle/min
normal person. However, this normal cardiac output is
achieved by a large stroke volume at a reduced heart
Resting blood flow
3.6
rate. Table 84-2 compares stroke volume and heart rate
Blood flow during maximal exercise
90
in the untrained person and the marathoner.
Thus, the heart-pumping effectiveness of each heart-
beat is 40 to 50 per cent greater in the highly trained
Thus, muscle blood flow can increase a maximum of
athlete than in the untrained person, but there is a cor-
about
25-fold during the most strenuous exercise.
responding decrease in heart rate at rest.
Almost one half this increase in flow results from intra-
muscular vasodilation caused by the direct effects of
Role of Stroke Volume and Heart Rate in Increasing the Cardiac
increased muscle metabolism, as explained in Chapter
Output. Figure 84-10 shows the approximate changes in
21. The remaining increase results from multiple factors,
stroke volume and heart rate as the cardiac output
the most important of which is probably the moderate
increases from its resting level of about 5.5 L/min to
increase in arterial blood pressure that occurs in exer-
30 L/min in the marathon runner. The stroke volume
cise, usually about a 30 per cent increase. The increase
increases from 105 to 162 milliliters, an increase of about
in pressure not only forces more blood through the
50 per cent, whereas the heart rate increases from 50 to
blood vessels but also stretches the walls of the arteri-
185 beats/min, an increase of 270 per cent. Therefore,
oles and further reduces the vascular resistance.
the heart rate increase accounts by far for a greater
1064
Unit XV Sports Physiology
35
Dexter 1951
30
Douglas 1922
15
Christensen 1931
4
Donald 1956
25
3
20
10
15
Figure 84-9
2
10
Relation between cardiac output
5
and work output (solid line) and
1
5
between oxygen consumption
and work output
(dashed line)
during different levels of exercise.
0
0
0
(Redrawn from Guyton AC, Jones
0
200
400
600
800
1000 1200 1400
1600
CE, Coleman TB: Circulatory
Work output during exercise (kg-meters/min)
Physiology: Cardiac Output and
Its Regulation. Philadelphia: WB
Saunders Co, 1973.)
Table 84-2
190
Comparison of Cardiac Function Between Marathoner
and Nonathlete
170
Stroke volume
165
150
Stroke Volume (ml)
Heart Rate (beats/min)
Resting
150
130
Nonathlete
75
75
Marathoner
105
50
110
135
Maximum
Nonathlete
110
195
Heart rate
90
Marathoner
162
185
120
70
105
50
0
10
15
20
25
30
proportion of the increase in cardiac output than does
Cardiac output (L/min)
the increase in stroke volume during strenuous exercise.
The stroke volume normally reaches its maximum by
the time the cardiac output has increased only halfway
to its maximum. Any further increase in cardiac output
Figure 84-10
must occur by increasing the heart rate.
Approximate stroke volume output and heart rate at different levels
of cardiac output in a marathon athlete.
Relation of Cardiovascular Performance to VO
2
Max. During
maximal exercise, both the heart rate and the stroke
volume are increased to about 95 per cent of their
maximal levels. Because the cardiac output is equal
important physiologic benefit of the marathoner’s train-
to stroke volume times heart rate, one finds that the
ing program.
cardiac output is about 90 per cent of the maximum that
the person can achieve. This is in contrast to about 65
Effect of Heart Disease and Old Age on Athletic Performance.
per cent of maximum for pulmonary ventilation. There-
Because of the critical limitation that the cardiovascu-
fore, one can readily see that the ca
lar system places on maximal performance in endurance
is normally much more limiting on VO2 Max than is the
athletics, one can readily understand that any type of
respiratory system, because oxygen utilization by the
heart disease that reduces maximal cardiac output will
body can never be more than the rate at which the car-
cause an almost corresponding decrease in achievable
diovascular system can transport oxygen to the tissues.
total body muscle power. Therefore, a person with con-
For this reason, it is frequently stated that the level
gestive heart failure frequently has difficulty achieving
of athletic performance that can be achieved by the
even the muscle power required to climb out of bed,
marathoner mainly depends on the performance capa-
much less to walk across the floor.
bility of his or her heart, because this is the most limit-
The maximal cardiac output of older people also
ing link in the delivery of adequate oxygen to the
decreases considerably—there is as much as a 50 per
exercising muscles. Therefore, the 40 per cent greater
cent decrease between age 18 and age 80. Also, there is
cardiac output that the marathoner can achieve over the
even more decrease in maximal breathing capacity. For
average untrained male is probably the single most
these reasons, as well as reduced skeletal muscle mass,
Chapter 84
Sports Physiology
1065
the maximal achievable muscle power is greatly
Body Fluids and Salt
reduced in old age.
in Exercise
As much as a 5- to 10-pound weight loss has been
Body Heat in Exercise
recorded in athletes in a period of
1 hour during
endurance athletic events under hot and humid condi-
Almost all the energy released by the body’s metabo-
tions. Essentially all this weight loss results from loss of
lism of nutrients is eventually converted into body heat.
sweat. Loss of enough sweat to decrease body weight
This applies even to the energy that causes muscle con-
only 3 per cent can significantly diminish a person’s per-
traction for the following reasons: First, the maximal
formance, and a 5 to 10 per cent rapid decrease in
efficiency for conversion of nutrient energy into muscle
weight can often be serious, leading to muscle cramps,
work, even under the best of conditions, is only 20 to 25
nausea, and other effects. Therefore, it is essential to
per cent; the remainder of the nutrient energy is con-
replace fluid as it is lost.
verted into heat during the course of the intracellular
chemical reactions. Second, almost all the energy that
Replacement of Sodium Chloride and Potassium. Sweat con-
does go into creating muscle work still becomes body
tains a large amount of sodium chloride, for which
heat because all but a small portion of this energy is
reason it has long been stated that all athletes should
used for (1) overcoming viscous resistance to the move-
take salt (sodium chloride) tablets when performing
ment of the muscles and joints, (2) overcoming the
exercise on hot and humid days. However, overuse of
friction of the blood flowing through the blood vessels,
salt tablets has often done as much harm as good. Fur-
and (3) other, similar effects—all of which convert
thermore, if an athlete becomes acclimatized to the heat
the muscle contractile energy into heat.
by progressive increase in athletic exposure over a
Now, recognizing that the oxygen consumption by the
period of 1 to 2 weeks rather than performing maximal
body can increase as much as 20-fold in the well-trained
athletic feats on the first day, the sweat glands also
athlete and that the amount of heat liberated in the
become acclimatized, so that the amount of salt lost in
body is almost exactly proportional to the oxygen con-
the sweat becomes only a small fraction of that lost
sumption (as discussed in Chapter 72), one quickly real-
before acclimatization. This sweat gland acclimatization
izes that tremendous amounts of heat are injected into
results mainly from increased aldosterone secretion by
the internal body tissues when performing endurance
the adrenal cortex. The aldosterone in turn has a direct
athletic events. Next, with a vast rate of heat flow into
effect on the sweat glands, increasing reabsorption of
the body, on a very hot and humid day so that the sweat-
sodium chloride from the sweat before the sweat itself
ing mechanism cannot eliminate the heat, an intolera-
issues forth from the sweat gland tubules onto the
ble and even lethal condition called heatstroke can
surface of the skin. Once the athlete is acclimatized,
easily develop in the athlete.
only rarely do salt supplements need to be considered
during athletic events.
Heatstroke. During endurance athletics, even under
Experience by military units exposed to heavy exer-
normal environmental conditions, the body tempera-
cise in the desert has demonstrated still another elec-
ture often rises from its normal level of 98.6° to 102° or
trolyte problem—the loss of potassium. Potassium loss
103°F (37° to 40°C). With very hot and humid condi-
results partly from the increased secretion of aldos-
tions or excess clothing, the body temperature can easily
terone during heat acclimatization, which increases the
rise to 106° to 108°F (41° to 42°C). At this level, the ele-
loss of potassium in the urine as well as in the sweat. As
vated temperature itself becomes destructive to tissue
a consequence of these findings, some of the supple-
cells, especially the brain cells. When this happens, mul-
mental fluids for athletics contain properly propor-
tiple symptoms begin to appear, including extreme
tioned amounts of potassium along with sodium, usually
weakness, exhaustion, headache, dizziness, nausea,
in the form of fruit juices.
profuse sweating, confusion, staggering gait, collapse,
and unconsciousness.
This whole complex is called heatstroke, and failure
Drugs and Athletes
to treat it immediately can lead to death. In fact, even
though the person has stopped exercising, the tempera-
Without belaboring this issue, let us list some of the
ture does not easily decrease by itself. One of the
effects of drugs in athletics.
reasons for this is that at these high temperatures, the
First, caffeine is believed by some to increase athletic
temperature-regulating mechanism itself often fails (see
performance. In one experiment on a marathon runner,
Chapter 73). A second reason is that in heatstroke, the
running time for the marathon was reduced by 7 per
very high body temperature itself approximately
cent by judicious use of caffeine in amounts similar to
doubles the rates of all intracellular chemical reactions,
those found in one to three cups of coffee. Yet experi-
thus liberating still more heat.
ments by others have failed to confirm any advantage,
The treatment of heatstroke is to reduce the body
thus leaving this issue in doubt.
temperature as rapidly as possible. The most practical
Second, use of male sex hormones (androgens) or
way to do this is to remove all clothing, maintain a spray
other anabolic steroids to increase muscle strength
of cool water on all surfaces of the body or continually
undoubtedly can increase athletic performance under
sponge the body, and blow air over the body with a fan.
some conditions, especially in women and even in men.
Experiments have shown that this treatment can reduce
However, anabolic steroids also greatly increase the risk
the temperature either as rapidly or almost as rapidly
of cardiovascular damage because they often cause
as any other procedure, although some physicians
hypertension, decreased high-density blood lipopro-
prefer total immersion of the body in water containing
teins, and increased low-density lipoproteins, all of
a mush of crushed ice if available.
which promote heart attacks and strokes.
1066
Unit XV
Sports Physiology
In men, any type of male sex hormone preparation
Booth FW, Chakravarthy MV, Spangenburg EE: Exercise
also leads to decreased testicular function, including
and gene expression: physiological regulation of the
both decreased formation of sperm and decreased
human genome through physical activity. J Physiol
secretion of the person’s own natural testosterone, with
543:399, 2002.
residual effects sometimes lasting at least for many
Chakravarthy MV, Booth FW: Eating, exercise, and “thrifty”
months and perhaps indefinitely. In a woman, even
genotypes: connecting the dots toward an evolutionary
more dire effects can occur because she is not normally
understanding of modern chronic diseases. J Appl Physiol
adapted to the male sex hormone—hair on the face, a
96:3, 2004.
bass voice, ruddy skin, and cessation of menses.
Clausen T: Effects of age and exercise training on NA+K+
Other drugs, such as amphetamines and cocaine, have
pumps in skeletal muscle. Am J Physiol Regul Integr
been reputed to increase one’s athletic performance. It
Comp Physiol 285:R720, 2003.
is equally true that overuse of these drugs can lead to
Delp MD, Laughlin MH: Regulation of skeletal muscle per-
deterioration of performance. Furthermore, experi-
fusion during exercise. Acta Physiol Scand 162:411, 1998.
ments have failed to prove the value of these drugs
Evans NA: Current concepts in anabolic-androgenic
except as a psychic stimulant. Some athletes have been
steroids. Am J Sports Med 32:534, 2004.
known to die during athletic events because of interac-
Gandevia SC: Spinal and supraspinal factors in human
tion between such drugs and the norepinephrine and
muscle fatigue. Physiol Rev 81:1725, 2001.
epinephrine released by the sympathetic nervous
Glass JD: Signalling pathways that mediate skeletal muscle
system during exercise. One of the possible causes of
hypertrophy and atropy. Nat Cell Biol 5:87, 2003.
death under these conditions is overexcitability of the
Guyton AC, Jones CE, Coleman TB: Circulatory Physiology:
heart, leading to ventricular fibrillation, which is lethal
Cardiac Output and Its Regulation, 2nd ed. Philadelphia:
within seconds.
WB Saunders Co, 1973.
Hochachka PW, Beatty CL, Burelle Y, et al: The lactate
paradox in human high-altitude physiological perform-
ance. News Physiol Sci 17:122, 2002.
Body Fitness Prolongs Life
Johnson NA, Stannard SR, Thompson MW: Muscle triglyc-
Multiple studies have now shown that people who main-
eride and glycogen in endurance exercise: implications for
tain appropriate body fitness, using judicious regimens
performance. Sports Med 34:51, 2004.
of exercise and weight control, have the additional
Kraemer WJ, Adams K, Cafarelli E, et al: American College
benefit of prolonged life. Especially between the ages of
of Sports Medicine position stand. Progression models in
50 and 70, studies have shown mortality to be three
resistance training for healthy adults. Med Sci Sports
times less in the most fit people than in the least fit.
Exerc 34:364, 2002.
But why does body fitness prolong life? The follow-
Myburgh KH: What makes an endurance athlete world-
ing are the two most evident reasons.
class? Not simply a physiological conundrum. Comp
First, body fitness and weight control greatly reduce
Biochem Physiol A Mol Integr Physiol 136:171, 2003.
cardiovascular disease. This results from (1) mainte-
Noakes TD, Peltonen JE, Rusko HK: Evidence that a central
nance of moderately lower blood pressure and (2)
governor regulates exercise performance during acute
reduced blood cholesterol and low-density lipoprotein
hypoxia and hyperoxia. J Exp Biol 204:3225, 2001.
along with increased high-density lipoprotein. As
Rennie MJ, Wackerhage H, Spangenburg EE, Booth FW:
pointed out earlier, these changes all work together to
Control of the size of the human muscle mass. Annu Rev
reduce the number of heart attacks and brain strokes.
Physiol 66:799, 2004.
Second, and perhaps equally important, the athleti-
Schnermann J: Exercise. Am J Physiol Regul Integr Comp
cally fit person has more bodily reserves to call on when
Physiol 283:R2, 2002.
he or she does become sick. For instance, an 80-year-old
Spangenburg EE, Booth FW: Molecular regulation of indi-
nonfit person may have a respiratory system that limits
vidual skeletal muscle fibre types. Acta Physiol Scand
oxygen delivery to the tissues to no more than 1 L/min;
178:413, 2003.
this means a respiratory reserve of no more than three-
Steele DS, Duke AM: Metabolic factors contributing to
fold to fourfold. However, an athletically fit old person
altered Ca2+ regulation in skeletal muscle fatigue. Acta
may have twice as much reserve. This is especially
Physiol Scand 179:39, 2003.
important in preserving life when the older person
Tanaka H, Seals DR: Dynamic exercise performance in
develops conditions such as pneumonia that can rapidly
Masters athletes: insight into the effects of primary human
require all available respiratory reserve. In addition, the
aging on physiological functional capacity. J Appl Physiol
ability to increase cardiac output in times of need (the
95:2152, 2003.
“cardiac reserve”) is often 50 per cent greater in the ath-
Tschakovsky ME, Hughson RL: Interaction of factors deter-
letically fit old person than in the nonfit person.
mining oxygen uptake at the onset of exercise. J Appl
Physiol 86:1101, 1999.
Watt MJ, Heigenhauser GJ, Spriet LL: Intramuscular tria-
cylglycerol utilization in human skeletal muscle during
References
exercise: is there a controversy? J Appl Physiol 93:1185,
2002.
Blair SN, LaMonte MJ, Nichaman MZ: The evolution of
physical activity recommendations. How much is enough?
Am J Clin Nutr 79:913S, 2004.
I
N D E X
Note: Page numbers followed by f refer to figures; page numbers followed by t refer to tables.
A
Acetyl-CoA carboxylase, 846
Acidosis (Continued)
A bands, 72, 73f, 90f
Achalasia, 783, 819
synaptic transmission and, 570
A wave, 107f, 108
Achlorhydria, 797, 820
treatment of, 398
Abdominal cavity
Acid(s). See also Acid-base balance;
Acini, pancreatic, 799
fluid accumulation in, 177, 306, 860
Acidosis; Hydrogen ions.
salivary, 793, 961-962
pressure in, 177
definition of, 383
Acne, testosterone and, 1005
Abdominal compression reflex, 213
ingestion of, 398
Acquired immunodeficiency syndrome
Abdominal muscles
ionization of, 385-386
(AIDS), T-cell destruction in, 447
during parturition, 1037
nonvolatile, 390
Acromegaly, 927, 927f, 974
in defecation, 790
strong, 384
Acrosome reaction, 1000
in respiration, 471, 472f
titratable, 394-395
Actin, 72-74, 73f, 74f
Abdominal veins, reservoir function of, 179
weak, 384
in pinocytosis, 19, 19f
ABO blood group, 451-453, 452f, 452t
Acid-base balance, 383-400
Actin filaments
Absence syndrome, 744
buffer systems in, 385-388, 387f. See also
in ameboid movement, 24
Acceleration
Buffer systems.
of cardiac muscle, 103
angular, 695-696, 695f
disorders of, 396-397, 396t. See also
of skeletal muscle, 72-73, 76-78, 76f
linear, 695
Acidosis; Alkalosis.
of smooth muscle, 93-94, 93f
Acceleratory forces
acid-base nomogram in, 399-400, 400f
Action potential. See also Membrane
centrifugal, 541-542, 542f
anion gap in, 400, 400t
potential.
linear, 542-543, 543f
diagnosis of, 398-400, 399f, 400f, 400t
cardiac muscle, 104-106, 104f, 105f. See
Acclimatization, 519
gastrointestinal tract obstruction and,
also Cardiac muscle, contraction of.
cold, 896
824-825, 824f
at sinus node, 116-118, 117f, 120f
heat, 893-894, 900, 1065
mixed, 399
atrial transmission of, 117f, 118, 118f,
high altitude. See High altitude.
potassium concentration in, 366
120f
natural, 540, 540f
isohydric principle of, 388
duration of, 105
work capacity and, 540
neonatal, 1048
excitation-contraction coupling and, 106
Accommodation
nomogram for, 399-400, 400f
hyperpolarization of, 67-68
ocular, 617-618, 617f, 649-650
normal values for, 399-400, 400f
plateau in, 67-68, 104f, 105
of pacinian corpuscle, 576
renal regulation of, 308, 390-400
refractory period of, 105-106, 105f
Acetate, blood flow and, 203
ammonia buffer system in, 393-394,
relative refractory period of, 105-106,
Acetazolamide, 403t, 404
394f
105f
Acetoacetic acid, 844, 861, 966, 966f
bicarbonate reabsorption in, 390-392,
rhythmicity of, 67-68, 67f, 117-118, 117f
Acetone, 844, 966
390f-392f, 395, 395t
through atrioventricular node, 117f,
Acetone breath, 844, 976
hydrogen ion secretion in, 390-392,
118-119, 118f, 120f
Acetyl coenzyme A, 22f, 23
391f, 392f, 395, 395t
through internodal pathways, 117f, 118,
degradation of, 833-835, 834f
in acidosis, 396, 396t
118f, 120f
fatty acid conversion of, 844-845, 845f
in alkalosis, 396-397, 396t
through Purkinje system, 117f, 119, 120f
fatty acid degradation to, 842-843, 843f
phosphate buffer system in, 392-393,
velocity of, 105
oxidation of, 843, 861
393f
ventricular transmission of, 117f,
pyruvic acid conversion to, 23, 833, 833f
quantification of, 394-395, 395t
119-120, 120f
Acetylcholine, 562-563, 562t, 730f, 731
respiratory regulation of, 388-389, 388f,
gastrointestinal smooth muscle, 772-773,
botulinum toxin effect on, 88, 88f
389f
772f
bronchial effects of, 479
Acid-base nomogram, 399-400, 400f
inspiratory, 514
coronary blood flow and, 251
Acidophils, 919, 919f, 920t
neural, 9, 61-65, 61f-64f, 564-568, 564f,
curare effect on, 88, 88f
Acidosis, 384, 390
566f, 567f. See also
destruction of, 87
ammonium ion excretion in, 394
Neurotransmitters; Synapse(s).
drug effects on, 88-89
hydrogen ion secretion in, 395
absolute refractory period of, 70
in cardiac function, 121, 148
in hemorrhagic shock, 283-284
all-or-nothing principle of, 66
in gallbladder emptying, 803
metabolic, 386, 399f, 400f
anesthetic effect on, 70
in gastrointestinal function, 775
anion gap in, 400, 400t
anions in, 64
in pancreatic secretion, 800-801
calcium reabsorption in, 372
calcium in, 64-65, 86, 86f, 560
molecular biology of, 88
diagnosis of, 398-400, 399f, 400f, 400t
cathode ray recording of, 70-71, 70f
pepsinogen secretion and, 798
hyperchloremic, 400, 400t
conduction of, 68-69, 69f
receptors for, 86-87, 86f, 87f, 96, 752
in chronic renal failure, 411f, 412
depolarization stage of, 61-62, 61f
REM sleep and, 741
in diabetes mellitus type I, 973-974, 973f
electrode excitation of, 69
secretion of, 750-751
potassium concentration in, 366
elicitation of, 69-70, 69f
in bronchiolar function, 479
renal correction of, 396, 396t
energy metabolism and, 66, 66f
in skeletal muscle function, 85-88,
oxygen-hemoglobin dissociation curve in,
frequency of, 575
86f-88f
508, 508f
generation of, 566, 566f
in smooth muscle function, 96
plasma calcium in, 371
hyperpolarization of, 67f, 68
structure of, 751
potassium secretion in, 371
inhibition of, 70
synthesis of, 751
renal tubular, 397, 413
initiation of, 65, 69
Acetylcholine channel, 48, 85-88, 86f-88f
respiratory, 386, 389, 399f, 400f
local, 69, 69f
Acetylcholine pathway, 710, 710f
diagnosis of, 398-400, 399f, 400f
plateau in, 66-67, 67f
Acetylcholinesterase, 87
hydrogen ion secretion in, 395
point of origin of, 566, 566f
drug inactivation of, 89
in diver, 547
positive feedback in, 9
Acetyl-CoA. See Acetyl coenzyme A.
renal correction of, 396, 396t
potassium in, 61-65, 61f-64f, 66
1067
1068
Index
Action potential (Continued)
Adenosine
Adrenal diabetes, 951-952
propagation of, 65-66, 65f
in blood flow, 196-197, 247, 251-252
Adrenal gland, 906f, 944, 945f. See also
receptor potential and, 574, 574f
in irreversible shock, 284
Adrenal cortex; Adrenal medulla.
recording of, 70-71, 70f
Adenosine diphosphate (ADP)
Adrenal medulla, 907t, 944, 945f
refractory period of, 70
formation of, 22, 22f
epinephrine secretion by, 207-208, 755,
repolarization stage of, 61f, 62
in glycolysis regulation, 836
756
resting, 61, 61f, 564-565, 564f
in platelet plug formation, 458
function of, 755
rhythmicity of, 67-68, 67f
oxygen utilization and, 508-509, 509f
norepinephrine secretion by, 207-208, 755,
safety factor for, 66, 70
rate-limiting activity of, 884
756
saltatory conduction of, 68-69, 69f
Adenosine monophosphate (AMP), 830
sympathetic nerve endings in, 749-750,
sodium in, 61-65, 61f-64f, 66, 87,
cyclic (cAMP)
755
87f
in adrenal steroid synthesis, 955
Adrenalin. See Epinephrine.
subthreshold, 69, 69f
second messenger activity of, 912,
Adrenergic receptors, 752, 753t
threshold for, 65, 69-70, 69f
913-914, 913f, 913t
Adrenocorticotropin/adrenocorticotropic
voltage-gated potassium channel in,
thyroid-stimulating hormone function
hormone (ACTH), 907t, 918, 919f,
62-63, 62f, 64, 64f
and, 938
920t
voltage-gated sodium channel in, 62,
Adenosine triphosphatase (ATPase), 23,
aldosterone secretion and, 950
62f, 64, 64f
835-836, 835f
corticotropin-releasing factor regulation
vs. stimulus artifact, 70f, 71
in muscle contraction, 76
of, 955
olfactory, 667-668
in renal potassium secretion, 368, 368f
cortisol secretion and, 955-957, 956f
retinal, 637-638, 637f
in sodium-potassium pump, 53, 53f
excess of, 958-959, 959f
skeletal muscle, 89-91, 90f, 91f. See also
Adenosine triphosphate (ATP), 829-830,
inhibition of, 955
Muscle (skeletal), contraction of.
830f, 881-884
structure of, 955
excitation-contraction coupling in,
cellular utilization of, 23-24, 23f, 829-830,
thyroid hormone effects on, 937
89-91, 91f
830f, 883-884, 883f
Adrenogenital syndrome, 959, 960f
initiation of, 87-88, 88f
formation of, 17, 22-24, 22f, 23f, 829-830,
Afterdischarge, 581-582, 581f
safety factor for, 88
830f, 881
Afterload, 110f, 111
T tubules in, 89, 90f
anaerobic, 836-837
Age
smooth muscle, 96-98, 97f
in citric acid cycle, 834, 834f, 836
cardiac output and, 232, 233f, 237
calcium in, 97
in fatty acid oxidation, 842-844, 843f
growth hormone and, 927
of gastrointestinal tract, 772-773
in glycolysis, 832-837, 833f
pulse pressure and, 173, 173f
plateau of, 96-97, 97f
in mitochondria, 17, 22-24, 22f, 23f,
vision and, 618
spontaneous generation of, 97
835-836, 835f
Agglutination, antigen, 444, 445
stretch excitation of, 97
in oxidative phosphorylation, 835-836,
in transfusion reactions, 452-453
Action tremor, 705, 707
835f
Agglutinins
Activator operator, 35f, 36
summary of, 836
anti-A, 452, 452t, 453, 453t
Active transport, 19, 46, 46f, 52-56
in action potential, 66
anti-B, 452, 452t, 453, 453t
adenosine triphosphate in, 882
in active transport, 882
anti-Rh, 453
co-transport, 54-55, 55f
in cardiac muscle, 251-252
Agglutinogens, 451-452, 452t
renal, 329-330, 330f, 333-334
in glandular secretion, 882
Aggressiveness, 720
sodium-amino acid, 55, 816
in glycolysis regulation, 836
Agouti-related protein, 868-870, 869t
sodium-glucose, 55, 55f, 816, 831
in irreversible shock, 284
Air. See also Nitrogen; Oxygen.
counter-transport, 54-55
in metabolism, 829-830, 830f
alveolar, 493-496
renal, 330, 330f, 333
in muscle contraction, 77, 79-80, 881-882
carbon dioxide concentration in,
in tubular reabsorption, 328-332,
in nerve conduction, 882
495-496, 495f, 496f
328f-331f
phosphocreatine interrelation with, 882
composition of, 493-494, 493t
primary, 53-54, 53f
phosphofructokinase inhibition by, 836
humidification of, 494
renal, 328f, 329
structure of, 22
oxygen concentration in, 494-495, 495f,
secondary, 54-55
Adenosine triphosphate (ATP) synthetase,
496f
renal, 329-330, 330f
23
renewal rate of, 494, 494f
through cellular sheets, 55-56, 56f
Adenyl cyclase-cAMP second messenger
atmospheric, composition of, 493-494,
Actomyosin filaments, of lymphatic
system, 913-914, 913f, 913t
493t
capillary, 193
ADH. See Antidiuretic hormone (ADH).
dead space, 495-496, 496f
Acupuncture, 603
Adipocytes (fat cells), 842
expired, 495-496, 496f
Adaptation
Adipokines, 905
Air embolism
of olfactory receptors, 668
Adipose cell lipase, 970
brain surgery and, 178
of pacinian corpuscle, 575-576, 575f
Adipose tissue, 842, 906f
submarine escape and, 550
of retinal receptors, 631-632, 632f
brown, 887-888, 896
Air hunger (dyspnea), 527, 531, 532
of sensory receptors, 575-576, 575f
fat storage in, 965
Air travel. See Aviation.
of taste receptors, 666
glucose transport to, 965
Airway obstruction, 526, 526f
of thermal receptors, 608
in athlete, 1055
in asthma, 529
pain receptors and, 599
in feeding regulation, 871
in atelectasis, 528-529, 529f
to cold, 896
lipoprotein lipase of, 841, 842
in emphysema, 526-527, 527f, 528f
to dark, 631-632, 632f
triglycerides of, 12f, 842
Airway resistance, in emphysema,
to heat, 893-894, 900, 1065
Adiposogenital syndrome, 1009, 1009f
527
to high altitude. See High altitude.
ADP. See Adenosine diphosphate (ADP).
Akinesia, 711
to light, 631-632, 632f
Adrenal cortex, 907t, 944, 945f
Alanine, 853f
Adaptive control, 9
disorders of, 957-960, 959f, 960f
glucose formation from, 857
Addisonian crisis, 958
hormones of, 944-960. See also
synthesis of, 856, 856f
Addison’s disease, 342, 957-958
Aldosterone; Androgens, adrenal;
Alarm response, 208, 757, 758
hyperkalemia in, 366, 370
Cortisol.
Albinism, 628
sodium excretion in, 363, 379
layers of, 944-945, 945f
Albino, 628
Adenine, 27, 28f, 29, 29f
tumor of, 959, 960f
Albumin, 855-857, 855f. See also Plasma
Adenohypophysis. See Pituitary gland,
zona fasciculata of, 944-945, 945f
proteins.
anterior.
zona glomerulosa of, 944, 945f
capillary diffusion of, 184, 184t
Adenoma, thyroid, 940
zona reticularis of, 945, 945f
colloid osmotic pressure and, 188
Index
1069
Albumin (Continued)
Alkalosis (Continued)
Amino acids (Continued)
fatty acid binding to, 841
respiratory, 386, 399f, 400f
cortisol effects on, 952
thyroxine binding to, 934
altitude and, 539
co-transport of, 55, 854
urinary, 317
causes of, 397
deamination of, 856, 862
Albuminuria, 317
diagnosis of, 398-400, 399f, 400f
essential, 853f, 855
Albuterol, 759
hydrogen ion secretion in, 395
extracellular, 4, 46f, 854-855
Alcohol ingestion
renal correction of, 396-397, 396t, 539
facilitated diffusion of, 50, 966, 969
headache and, 607
synaptic transmission and, 570
gastrointestinal absorption of, 854
peptic ulcers and, 821
treatment of, 398
glucose formation from, 857
Aldosterone, 907t, 946f, 947t
whole-body, 824
hydrogen bonds of, 854
ACTH effects on, 950
Allergen, 449
insulin secretion and, 969
angiotensin and, 225
Allergy, 449-450
interstitial, 294t
arterial pressure and, 225, 230f, 231,
anaphylaxis in, 285-286, 287, 449-450
intracellular, 22, 22f, 46f, 294t. See also
948-949, 948f
atopic, 449
Adenosine triphosphate (ATP).
cellular effects of, 949-950
basophils in, 436
nonessential, 855-856, 856f, 862
deficiency of, 947-948, 957
cortisol in, 954
oxidation of, 856-857
potassium balance and, 342
delayed-reaction, 449
peptide linkages of, 34-35, 808, 810, 810f,
sodium balance and, 342, 379
eosinophils in, 436
852, 881
excess of, 948-949, 948f, 959
in asthma, 529
plasma concentration of, 854-855
alkalosis and, 398
mast cells in, 436
cortisol effects on, 952
hypertension and, 223
neonatal, 1049
insulin deficiency and, 967
potassium balance and, 342
Allograft, 455
proximal tubular transport of, 330, 330f
sodium balance and, 342, 379
All-or-nothing principle, 66
renal threshold for, 854
extracellular fluid osmolarity and,
Alpha brain waves, 742, 742f, 743, 743f
RNA codons for, 31-32, 32t
362-363, 363f
Alpha receptors, 251, 752, 753t
storage of, 854-855
functions of, 947-950, 948f
Altitude. See High altitude.
transamination of, 856, 856f, 862, 877
hepatic metabolism of, 947
Alveolar macrophages, 433, 481
urinary, 413
hydrogen ion secretion and, 395
Alveolar membrane, 5.
Aminoaciduria, 413
in cardiac failure, 263-264
Alveolar pressure, 472-473, 472f
Aminopolypeptidase, 810, 810f
in extracellular sodium regulation,
Alveolar ventilation, 477-478
Aminotransferases, 856
362-363, 363f
alveolar Pco2 and, 494-495, 495f
Amitriptyline, 745
in heat acclimatization, 893, 1065
arterial Pco2 and, 521, 521f
Ammonia, 862. See also Urea.
in pregnancy, 1034
arterial Pco2 and, 519-520, 519f
Ammonia buffer system, 393-394, 394f
inhibitors of, 403t, 404
dead space and, 477-478, 478f
Ammonium chloride, in alkalosis, 398
intestinal sodium absorption and,
Pco2 and, 495-496, 495f, 517, 517f
Ammonium citrate, 467
814-815, 814f, 949
pH and, 388-389, 388f, 517, 517f
Ammonium ion, 393-394, 394f
intracellular potassium and, 366, 366t
rate of, 478, 494, 494f
Amnesia
nongenomic actions of, 950
ventilation-perfusion ratio and, 499-501,
anterograde, 737
plasma protein binding of, 947
500f
retrograde, 725, 726
potassium effects on, 950
wasted, 500-501
Amniotic fluid, 1035
potassium secretion and, 366, 369-370,
Alveolus (alveoli), 479, 479f. See also
Amorphosynthesis, 592
369f, 370f, 378, 948
Lungs.
Amphetamines, 759
protein synthesis and, 915, 949-950
air in, 493-496
exercise and, 1066
radioimmunoassay of, 916, 916f
carbon dioxide concentration in,
in obesity, 873
receptor for, 949-950
495-496, 495f, 496f
Ampulla, 693f, 694
regulation of, 950, 951f
composition of, 493-494, 493t
Amygdala, 737-738, 870
renin-angiotensin system effects on, 225,
humidification of, 494
Amylase, pancreatic, 799, 809, 809f
950, 951f
oxygen concentration in, 485, 494-495,
Amylin, 961
salivary gland effects of, 949
495f, 496f
Amyloid plaques, 746
sodium effects on, 950
renewal rate of, 494, 494f
Amyloidosis, 409
sodium reabsorption and, 225, 342, 342t,
carbon dioxide diffusion into, 504-505,
Anaerobic energy, 836-837, 882-883
378-379, 948
505f
Anal sphincters, 789
structure of, 908f
collapse of (atelectasis), 528-529, 529f
Analgesia system, of central nervous
sweat gland effects of, 949
fluid in, 487
system, 602-603, 602f
synthesis of, 945-947, 946f, 947t
gas exchange in. See Gas exchange.
Anaphase, 37f, 39
tumor-related secretion of, 223, 959
interstitial pressure effect on, 488, 488f
Anaphylaxis, 285-286, 287, 449-450
Aldosterone antagonists, 403t, 404
J receptors of, 521
slow-reacting substance of, 450, 480,
Aldosterone escape, 379, 948-949
occlusion of, 474
529
Aldosteronism. See Aldosterone, excess of.
oxygen diffusion from, 502-503, 503f
Anatomic dead space, 478
Alimentary tract, 771, 772f. See also
pressures in, 472-473, 472f, 492
Anchoring filaments, of lymphatic capillary,
Gastrointestinal tract and specific
structure of, 496, 496f, 497f
191, 192f
components.
surface tension of, 473-474
Androgens. See also Testosterone.
Alkali
type II epithelial cells of, 474
adrenal, 944, 957, 1003, 1018
definition of, 383-384
walls of, 496, 497f
tumor-related excess of, 959, 960f
ingestion of, 398
Alzheimer’s disease, 746
chemistry of, 1003, 1004f
Alkaline phosphatase, 990
Amacrine cells, 634, 635, 635f, 636
exercise and, 1065-1066
Alkalosis, 384, 390
Ambenonium, 759
ovarian, 1003
metabolic, 386, 399f, 400f
Ameboid movement, 24, 24f, 431
Androstenedione, 946f
aldosterone excess and, 949
Ameloblasts, 992
Anemia, 422, 426-427
calcium reabsorption in, 372
Amenorrhea, 938, 1023
aplastic, 426
causes of, 397-398
Amiloride, 403t, 404
blood loss, 426
diagnosis of, 398-400, 399f, 400f
Amino acids, 33-35, 34f, 810, 810f, 852-854,
bone marrow response to, 422
hydrogen ion secretion in, 395
853f. See also Protein(s).
cardiac effects of, 236-237, 236f, 427
potassium concentration in, 366
active transport of, 854
exercise and, 427
renal correction of, 396-397, 396t
blood concentration of, 854
hematocrit in, 293
plasma calcium in, 371
glucagon secretion and, 971
hemolytic, 427
1070
Index
Anemia (Continued)
Antibody (antibodies) (Continued)
Aorta
hypochromic, 421f, 425, 426
agglutination by, 444, 445
baroreceptors of, 209-212, 209f
in erythroblastosis fetalis, 454
agglutinin, 452, 452t, 453, 453t
chemoreceptors of, 211-212, 518-519,
in kidney disease, 412, 423
bivalent, 444, 444f
518f
megaloblastic, 421f, 427
classes of, 444
coarctation of, 227, 274
microcytic hypochromic, 421f, 426
complement interaction with, 445-446,
pressure in, 109, 173, 173f
pernicious, 423, 427, 797, 820, 877
445f
Aortic body, 211-212, 518, 518f
sickle cell, 421f, 424, 427
direct action of, 444
mechanism of action of, 518-519
Anencephaly, 697
for passive immunity, 449
Aortic-coronary bypass surgery, 256
Anesthesia
formation of, 442-446, 443f-445f
Aortic pressure curve, 109
cardiac arrest and, 156
in human milk, 1041
Aortic regurgitation
cardiac output and, 242, 242f
lysis by, 444, 445, 445f
circulatory dynamics in, 272-273
local, 70
mechanisms of action of, 444-446,
heart sounds in, 271f, 272
neurogenic shock and, 285
445f
pulse pressure in, 173, 173f, 174
respiratory effects of, 522
neutralization by, 444, 445
Aortic stenosis
synaptic transmission and, 570
precipitation by, 444
circulatory dynamics in, 272-273
Angina pectoris, 145, 255-256. See also
salivary, 794
congenital, 274
Myocardial infarction.
sensitizing, 449
heart sounds in, 271f, 272
Angiogenesis
specificity of, 444, 444f
pulse pressure in, 173, 173f, 174
growth factors in, 41, 200-201
structure of, 443-444, 444f
Aortic valve, 109, 109f
high altitude effects on, 539-540
Anticholinesterase drugs, 759
Aphagia, 868
Angiogenic factors, 41, 200-201
Anticoagulants
Aphasia, 721
Angiogenin, 200
clinical, 466-468
Aplysia, 724, 724f
Angioplasty, coronary, 256
endogenous, 459, 463-464
Apnea, sleep, 522-523
Angiotensin I, 223f, 224
Anticodons, 32-33, 32f
Apocrine glands. See Sweating.
Angiotensin II. See also Renin-angiotensin
Antidepressants, 745
Apoferritin, 425
system.
Antidiuretic hormone (ADH), 907t, 918,
Apolipoprotein E, 746
arteriolar effects of, 201-202
919f
Apoptosis, 40
extracellular fluid osmolarity and,
blood flow and, 202
Apotransferrin, 425, 426
362-363
functions of, 928-929
Appendix, inflammation of, 605, 605f
formation of, 223f, 224
in extracellular fluid sodium regulation,
Appetite, 734, 781, 867, 870. See also
in cardiac failure, 263
358-359, 358f, 362-363, 362f
Feeding; Food, intake of.
glomerular filtration rate and, 322, 322t,
in hemorrhagic shock, 281
brain area of, 795
325
in urine concentration, 337, 350-351, 353,
salt, 363
in arterial pressure regulation, 201-202,
356, 379
Apraxia, motor, 687
224-226, 225f, 226f, 378
in urine dilution, 337, 348-350, 349f
Aquaporins, 928
in extracellular volume control, 377-378
secretion of, 220
Aqueous humor, 623-624, 623f, 624f
in hemorrhagic shock, 281
arterial pressure and, 360, 360t
Arachnoidal villi, 765
in pressure natriuresis, 378, 378f
blood volume and, 360, 360f, 360t
Arcuate arteries, 309, 309f
in renal blood flow, 322, 322t
cardiopulmonary reflexes and, 360
Arcuate veins, 309f, 310
in renal ischemia, 413
drug effects on, 360t, 361
Arginine, 853f
in sodium excretion, 377-378
hypothalamus and, 734
Argyll Robertson pupil, 650
in sodium reabsorption, 342-343, 342t
in cardiac failure, 264
Arm
in tubular reabsorption, 342, 342t
inappropriate, 357, 379
lever system of, 82, 82f
in water reabsorption, 342-343, 342t
nausea and, 360-361
veins of, 178
thirst and, 361, 361t
plasma osmolarity and, 358-360, 358f,
Arrhenoblastoma, 1003
urine output and, 342
360t
Arrhythmias, 147-156. See also at specific
Angiotensin II antagonists, glomerular
regulation of, 358-361, 358f-360f, 360t,
arrhythmias.
filtration rate and, 325
928-929
arrest, 156
Angiotensin-converting enzyme (ACE)
thirst system integration with, 362-363,
atrial fibrillation, 155-156, 155f
inhibitors
362f
atrial flutter, 156, 156f
glomerular filtration rate and, 325
structure of, 928
atrioventricular block, 148-150, 149f
pressure natriuresis effects of, 378, 378f
synthesis of, 359-360, 359f
intraventricular block, 150, 150f
Angiotensinase, 223f, 224
vasoconstrictor effects of, 929
paroxysmal tachycardia, 151-152, 153f
Angiotensinogen, 223f, 224
water reabsorption and, 337, 338, 342t,
premature contraction, 150-151, 150f, 151f
Angular acceleration, 695-696, 695f
343, 348-349, 379, 734
sinoatrial block, 148, 148f
Angular gyrus, 716, 716f, 718
Antigen(s), 440, 441f. See also Antibody
sinus, 148, 148f
Anion gap, 400, 400t
(antibodies).
ventricular fibrillation, 152-155, 153f-155f
Anions
antibody interaction with, 442, 444-446,
Arterial blood pressure, 166-167, 166f
action potential and, 64
445f
abdominal compression reflex and, 213
blood flow and, 203
HLA, 455-456
aldosterone effects on, 225, 948-949, 948f
Annulospiral ending, 675-676
red blood cell, 451-454, 452f, 452t, 453t
angiotensin effect on, 201-202, 224-226,
Anorexia, 874
self-, 448
225f, 226f, 378
Anorexia nervosa, 874
Antigen-presenting cells, 446, 446f
antidiuretic hormone effect on, 360, 379
Anorexigenic substances, 868, 869t
Antihemophilic factor (factor VIII), 459t,
auscultation of, 175, 175f
Anovulation, 1021, 1024-1025
465
baroreceptor response to, 6-7, 209-212,
Anterior commissure, 722
Antimuscarinic drugs, 759
209f-211f, 230, 230f
Anterograde amnesia, 737
Antioncogenes, 41
blood flow and, 164, 164f. See also Blood
Anterolateral sensory pathway, 587,
Antiperistalsis, 824
flow.
595-597, 596f
Antipyrine, for total body water
blood volume and, 172, 172f, 219-220,
Antibiotics
measurement, 296
220f
in peptic ulcers, 821
Antithrombin III, 463-464
body fluid volume and, 219-220, 220f,
renal toxicity of, 406
Antithyroid drugs, 939-940
374-376, 375f
Antibody (antibodies), 432, 441, 441f. See
Anuria
capillary fluid shift and, 230f, 231
also at Immunoglobulin.
in cardiac failure, 259, 263-264
cardiac output and, 114, 114f, 219-220,
affinity constant of, 444
in kidney failure, 405, 406
220f
Index
1071
Arterial blood pressure (Continued)
Arterial blood pressure (Continued)
Artery (arteries) (Continued)
centrifugal acceleratory force effects on,
renal control of, 216-231, 230f, 308, 323,
diameter of, 167-168, 168f
542, 542f
341-342, 374-376, 375f, 376f. See also
distensibility of, 171, 173-176, 173f-176f
cerebral blood flow and, 762-763, 763f
Kidneys; Renal tubules.
function of, 161
chemoreceptor response to, 211-212, 230,
aldosterone escape and, 948-949, 948f
hardening of, 848-851, 849f. See also
230f
angiotensin in, 223-226, 224f-226f
Arteriosclerosis; Atherosclerosis;
control of, 163, 230-231, 230f
atrial reflexes and, 212
Myocardial infarction.
capillary fluid shift mechanism in, 230f,
baroreceptor reflexes and, 211
mean pressure in, 175-176, 176f
231
experimental demonstration of, 217,
nitric oxide synthesis in, 199-200
hypothalamus in, 207, 733, 733f
217f
parallel circuits of, 168-169, 169f
neural. See Arterial blood pressure,
graphical analysis of, 217-218, 218f
pressure in. See Arterial blood
nervous system control of.
peripheral resistance and, 219-220,
pressure.
renal. See Arterial blood pressure, renal
219f, 220f
pressure pulse in, 174-175, 174f
control of.
quantitation of, 217-220, 217f-220f
sympathetic innervation of, 172, 173f, 204,
stress-relaxation mechanisms in,
renal resistance and, 219
205f
230-231, 230f
renin-angiotensin system in, 223-226,
volume-pressure curves of, 172, 172f
diastolic, 162, 163f, 173, 175-176, 175f
224f-226f
Articulation, 721-722, 721f. See also
diuresis and, 216, 217-220, 217f-220f
salt intake/output and, 218-219, 218f,
Language; Speech.
during exercise, 208, 213-214, 235-236,
220, 224-226, 226f
Artificial cooling, 900
248-249, 248f
water intake/output and, 218-219, 218f,
Artificial kidney (dialysis), 414-415, 414f,
equilibrium point of, 217-218
224-226, 226f
415t
frequency distribution of, 210, 211f
renin-angiotensin system effects on,
Artificial respiration, 532, 532f
hemorrhage effect on, 224, 224f
224-226, 224f-226f
Arytenoid cartilage, 481, 481f
high. See Hypertension.
respiratory waves in, 214
Ascites, 177, 306, 381, 860
high-fidelity measurement of, 166-167,
salt sensitivity of, 229, 229f
in thiamine deficiency, 876
167f
sodium excretion and, 374-375, 375f
Ascorbic acid. See Vitamin C (ascorbic
in cardiogenic shock, 262
stress relaxation and, 230-231, 230f
acid).
in cerebral ischemia, 212-213
systemic (greater, peripheral), 162, 163f
Asparagine, 853f
in circulatory shock, 279, 279f
systolic, 162, 163f, 173, 175-176, 175f, 220
Aspartic acid, 853f
in extracellular fluid balance, 374-376,
thirst and, 361, 361t
Aspirin, peptic ulcers and, 821
375f, 376f
thyroid hormone effects on, 937
Astereognosis, 591
in hemorrhage, 224, 224f
units of, 166-167
Asthenia, 974
in hemorrhagic shock, 279-280, 279f, 280f
urine output and, 216, 323, 341-342,
Asthma, 436, 450, 480, 529
in polycythemia, 428
374-376, 375f, 376f
Astigmatism, 620, 620f
in sodium balance, 374-376, 375f
aldosterone escape and, 948-949,
Astronauts. See Space travel.
integrated regulation of, 230-231, 230f
948f
Ataxia, 706
low-pressure receptor response to, 212
quantitation of, 217-220, 217f-220f
Atelectasis, 528-529, 529f
Mayer waves of, 214, 214f
vascular resistance and, 170, 170f, 219,
Atherosclerosis, 842, 848-851, 849f. See also
mean, 175-176, 176f, 220
219f, 220, 220f
Arteriosclerosis.
measurement of, 166-167, 166f, 167f,
vasoconstrictor tone and, 206, 207f
arterial pressure in, 175
175-176, 175f, 178
vasomotor waves of, 214, 214f
blood pressure in, 175
neonatal, 1047
vasopressin effect on, 202
cholesterol in, 850
nervous system control of, 208-213, 230,
Arteriole(s), 181, 182f
coronary, 252, 256. See also Myocardial
230f, 235-236, 235f, 755
angiotensin effect on, 201-202
infarction; Myocardial ischemia.
abdominal compression reflex in, 213
antidiuretic hormone effects on, 929
in Alzheimer’s disease, 746
adrenal medullae in, 207-208
autonomic nervous system control of, 204,
in hypothyroidism, 942
atrial reflex in, 212
205f, 754t
lipoproteins in, 850
Bainbridge reflex in, 212
blood distribution to, 162f
prevention of, 850-851
baroreceptors in, 209-212, 209f-211f,
bradykinin effect on, 202
renal, 408
214, 214f
cross-sectional area of, 162
risk factors for, 850
Cushing reaction in, 213, 213f
diameter of, 168, 181
Athetosis, 709
during exercise, 208, 213-214
during exercise, 247-248
Athlete, 1055. See also Exercise.
ischemic response in, 212-213, 213f,
function of, 161
bradycardia in, 147
214, 214f
hepatic, 859
Atmospheric pressure, under water, 545,
norepinephrine in, 207
hydrogen ion effect on, 202-203
546f
parasympathetic system in, 205-206
intermittent contraction of (vasomotion),
Atony
rapidity of, 208-213, 209f-211f
182-183, 187
bladder, 313-314
reflex mechanisms in, 209-212,
nitric oxide synthesis in, 199-200
colonic, 822
209f-211f, 214, 214f
pressure pulse in, 174-175, 174f
ATP. See Adenosine triphosphate (ATP).
respiratory waves and, 214, 214f
renal, 309, 309f, 311f
Atrial fibrillation, 155-156, 155f, 156f, 273
stress and, 208
resistance of, 168
Atrial flutter, 156, 156f
sympathetic system in, 204-205, 205f,
Arteriosclerosis, 982. See also
Atrial natriuretic peptide, 907t
206-208, 206f
Atherosclerosis.
extracellular fluid volume and, 379-380
vasoconstriction and, 204-205, 205f,
diabetes mellitus and, 976
in cardiac failure, 264
206-208, 206f
pulse pressure in, 173, 173f
tubular reabsorption and, 342t, 343
vasodilation and, 208
Arteriovenous fistula, cardiac output in, 236,
Atrial paroxysmal tachycardia, 152, 152f
vasomotor center in, 206-207, 206f,
236f, 242-243, 243f, 267, 267f
Atrial pressure
212-213, 213f, 214, 214f
Artery (arteries). See also Arteriole(s);
heart rate and, 212
volume reflex in, 212
Capillary (capillaries).
left, 107f, 108, 484, 487
normal, 175, 176f
autonomic nervous system control of,
edema and, 489, 489f
peripheral vascular resistance and, 170,
754-755, 754t
right, 176
170f, 219, 219f, 220, 220f
baroreceptors of. See Baroreceptor(s).
cardiac output and, 240-243, 240f-243f.
pressure diuresis and, 375-376, 375f,
blood flow in. See Blood flow.
See also Cardiac output.
376f
collateral, 201
measurement of, 176, 179
pressure natriuresis and, 375-376, 375f
compliance of, 171-172, 173, 173f
peripheral venous pressure and, 177
renal, 320-321, 321t
cross-sectional area of, 162
Atrioventricular block, 121
1072
Index
Atrioventricular bundle, 104
Autonomic nervous system (Continued)
Beriberi, 875
action potential transmission in, 117f,
in ocular accommodation, 618, 648-649,
blood flow in, 198
118-119, 118f
648f
cardiac output in, 236, 236f, 267, 267f
left and right branches of, 117f, 118f, 119
pharmacology of, 759-760
oxygen uptake in, 530
pacemaker function in, 120-121
vasomotor center of, 205f, 206, 206f
Beta-aminoisobutyricaciduria, 413
Atrioventricular node
Autoregulation
Beta-amyloid peptide, 746
acetylcholine effects on, 121
in coarctation of aorta, 227
Beta blockers, 256
action potential transmission in, 117f,
of blood flow, 198-199, 198f, 219-220,
Beta brain waves, 742, 742f, 743, 743f
118-119, 118f, 119f
220f
Beta receptors, 251, 752, 753t
block of, 148-150, 149f
A-V node. See Atrioventricular node.
Beta-oxidation, 842-843, 843f, 861
hyperpolarization of, 121
Aviation. See also Space travel.
Betz cells, 687-688
in atrial fibrillation, 155-156
acceleratory forces in, 541-543, 542f,
Bicarbonate. See also Acid-base balance.
intrinsic rhythmical rate of, 120
543f
blood, 394
norepinephrine effects on, 122
in unpressurized airplane, 538, 538f
extracellular, 4, 7t, 46f, 294f, 294t
pacemaker function in, 120-121
mental proficiency in, 539
from carbonic acid dissociation, 511
premature contractions of, 150-151,
Axis. See Mean electrical axis.
ileal secretion of, 815
151f
Axon, 68, 68f, 555, 556f, 559, 559f. See also
interstitial, 294t
safety factor for, 121
Action potential, neural.
intestinal absorption of, 815
Atrioventricular valves, 107f, 108, 109, 109f.
Axonal streaming, 564
intestinal secretion of, 815, 817
See also Mitral valve; Tricuspid valve.
Axoneme, of cilia, 25
intracellular, 294f, 294t
sounds at, 269, 270
Axoplasm, 68, 68f
loop of Henle transport of, 334-336,
Atrium (atria), 104
Azotemia, 411f, 412. See also Kidneys,
335f
action potential transmission in, 117f, 118,
failure of.
pancreatic secretion of, 800, 800f, 801
118f, 119f, 120f
reabsorption of, 328t, 338f, 390-392,
in mitral valve lesions, 273
390f-392f, 395, 395t
pressure in, 107f, 108, 484, 487
renal formation of, 392-394, 393f, 394f
edema and, 489, 489f
B
ammonia buffer system in, 393-394,
low-pressure receptors of, 212
Bacteria, 18, 18f. See also Infection.
394f
pressure waves of, 107f, 108
colonic, 817
phosphate buffer system in, 393, 393f
primer pump function of, 107-108, 107f
Bactericidal agents
renal titration of, 392
pressure in, 176
of lysosomes, 20
salivary, 794
cardiac output and, 240-243, 240f-
of saliva, 794
urinary, 394
243f. See also Cardiac output.
of white blood cells, 432
Bicarbonate buffer system, 385-387, 386f
measurement of, 176, 179
Bainbridge reflex, 212, 233
dissociation constant in, 385-386
peripheral venous pressure and, 177
Baldness, 1005
Henderson-Hasselbalch equation in,
stretch of, 112
Baroreceptor(s), 6-7, 209-212, 757
386
volume reflex of, 212
activation of, 209-210, 209f, 210f
titration curve for, 386-387, 386f
Atrophy
anatomy of, 209, 209f
Bile, 802-805, 803f, 803t. See also Bile salts.
gastric, 820
buffer function of, 210, 211f, 230, 230f
composition of, 803, 803t
muscle, 83
denervation of, 210, 211f, 228
deficiency of, 464-465
Atropine, 759
in antidiuretic hormone secretion, 360
gallbladder storage of, 802-803
Audiogram, 661, 661f
in cardiac failure, 258-259
in fat digestion, 811
Audiometry, 660-661, 661f
in extracellular volume control, 377
secretion of, 802, 803f, 804
Auditory association cortex, 658, 659, 659f
in hemorrhagic shock, 281
Bile acids, 802, 804
lesions of, 659
oscillation of, 214, 214f
atherosclerosis and, 850-851
Auditory cortex, 657-659
postural effects on, 210
Bile ducts, 802
primary, 658-659, 659f
resetting of, 210-211
obstruction of, 864
lesions of, 660
Barrel chest, 529
Bile salts, 803, 803t, 861
pathway to, 657-658, 658f
Basal ganglia, 558, 707-712, 708f
emulsifying function of, 804, 811
sound direction and, 660
caudate circuit of, 709, 709f, 710
enterohepatic circulation of, 804
sound frequency response of, 659
in movement control, 709-710, 710f,
extracellular, 294f
sound pattern discrimination by, 659
712-713
function of, 804-805
secondary, 658, 659, 659f
lesions of, 709, 710, 710f, 711-712
in fat digestion, 804-805, 811, 812, 816
lesions of, 659
neurotransmitters of, 710-711, 710f
plasma, 294f
Auditory nerve
putamen circuit of, 708-709, 708f
tubular secretion of, 334
disorders of, 660-661
Basal metabolic rate. See Metabolic rate,
Bilirubin, 426, 862-864, 863f
stimulation of, 655-656
basal.
excretion of, 307, 803, 803t
Auditory receptive aphasia, 721
Base, 383-384
extracellular, 294f
Auerbach’s (myenteric) plexus, 774-775,
strong, 384
free vs. conjugated, 863, 864
774f, 777
weak, 384
plasma, 294f
Auscultation
Basement membrane, of glomerular
Bilirubin glucuronide, 863
of blood pressure, 175, 175f
capillary, 316, 316f
Bilirubin sulfate, 863
of heart sounds, 270-271, 270f
Basilar membrane, 652, 652f, 653, 653f
Bipolar cells, 633, 635, 635f, 636, 638f
Autacoids, in renal blood flow, 322-323, 322t
vibration of, 654-655, 654f
Bipolar disorder, 745
Autocrines, 905
Basket cells, 702
Birth, 929, 1036-1038, 1037f
Autograft, 455
Basophil(s), 421f, 429-430, 430f, 436
breathing at, 1044-1045, 1045f
Autoimmune disease, 448
activation of, 445-446
2,3-Bisphosphoglycerate, oxygen-
Autolysis, 20
concentration of, 430
hemoglobin dissociation curve effects
Autonomic nervous system, 6, 556, 748-760.
heparin production by, 464
of, 508, 508f
See also Parasympathetic nervous
Basophil erythroblast, 421f, 422
Bitemporal hemianopsia, 645
system; Sympathetic nervous system.
Bathorhodopsin, 629, 629f
Black tongue, 876
anatomy of, 748-750, 749f, 750f
B-cell growth factors, 447
Bladder. See also Kidneys; Urine.
control of, 758-759, 758f
B-cell stimulating factors, 447
anatomy of, 309, 309f, 311-312, 311f
functions of, 750-757, 753t, 754t
Bellini, duct of, 311f
atonic, 313-314
in blood flow regulation, 204-208,
Bends (decompression sickness), 548-549,
autonomic control of, 754t, 757
205f-207f
548f
cystometrography of, 312-313, 313f
Index
1073
Bladder (Continued)
Blood (Continued)
Blood flow (Continued)
filling of, 312-313, 313f
venous admixture of, 503, 503f
autoregulatory mechanisms in, 198-199,
incontinent, 313-314
viscosity of
198f, 219-220, 220f
infection of, 409
in anemia, 427
collateral circulation and, 201
innervation of, 311f, 312, 313
in polycythemia, 428
during exercise, 198, 201
micturition reflex of, 311, 313-314
vascular resistance and, 169-170, 170f
glucose in, 198
neurogenic, 314
volume of. See Blood volume.
hyperemia and, 198
sensory stretch receptors of, 313
Blood clot, 458-459, 458f, 460. See also
in brain, 199
tabetic, 314
Coagulation.
in kidneys, 199. See also Kidneys, blood
urine transport to, 312-313, 313f
intravascular, 465-466
flow of.
voluntary control of, 313
lysis of, 464
long-term, 198f, 200-201
Blastocyst, 1029, 1029f
retraction of, 458f, 460
metabolic theory of, 199
Bleeding. See also Coagulation;
Blood flow, 164-170. See also Cardiac
myogenic theory of, 199
Hemorrhage.
output; Circulation (blood).
oxygen availability and, 196-198, 196f,
in coagulation factor deficiency, 464-465
acute control of, 196-200, 196f-198f
197f, 200
in hemophilia, 465
after vascular occlusion, 198
oxygen lack theory of, 197-198, 197f
in thrombocytopenia, 465
angiotensin effect on, 201-202
tissue metabolism and, 196, 196f
in vitamin C deficiency, 878
anion effect on, 203
vascularity and, 200-201
Bleeding time, 467
arrest of, 287-288
vasodilator theory of, 196-197
Blind spot, 644
autonomic nervous system regulation of,
vitamin deficiency and, 198
Blindness
204-208, 205f-207f
magnesium effect on, 202
color, 633, 634f
autoregulation of, 198-199, 198f, 219-220,
maximum need for, 201
glaucoma and, 624-625
220f
measurement of, 164-165, 165f
in premature infant, 1051
blood pressure and, 164, 164f, 166-167,
metabolic rate and, 196, 196f
night, 629, 875
166f, 170, 170f
metabolic theory of, 199
visual cortex removal and, 644
bradykinin effect on, 202
myogenic theory of, 199
Blood. See also Plasma.
calcium ion effect on, 202
norepinephrine and, 201, 247, 251, 322,
acetoacetic acid transport in, 844
capillary, 4-5, 4f, 5f, 182-183
322t
amino acid concentration of, 854
average rate of, 183
of kidneys. See Kidneys, blood flow of.
glucagon secretion and, 971
during exercise, 246
Ohm’s law and, 164
autonomic nervous system control of,
carbon dioxide effect on, 203
oxygen deficiency and, 196-198, 197f
754t
cerebral, 199, 761-763, 762f, 763f. See also
oxygen effect on, 200
bicarbonate in, 394-395
Cerebral blood flow.
oxygen lack theory of, 197-198, 197f
carbon dioxide in, 385-386
conductance in, 167-168, 168f, 169
oxygen utilization and, 509
circulation of, 4-5, 4f. See also Circulation
control of
parabolic velocity profile of, 165-166, 165f
(blood).
acute, 196-200, 196f-198f
placental, 1035
citrate treatment of, 467
angiotensin in, 201-202
potassium ion effect on, 202
clotting of, 458-459, 458f, 460, 464. See
bradykinin in, 202
pulmonary, 244, 244f
also Coagulation.
endothelin in, 202
in atelectasis, 529, 529f
collection of, 466-467
epinephrine in, 201
rate of, 163
extracorporeal circulation of, 276
histamine in, 202
renal. See Kidneys, blood flow of.
fatty acid concentration of, 841
humoral, 201-203
resistance to. See Vascular resistance.
flow of. See Blood flow; Circulation
ions in, 202-203
Reynolds’ number for, 166
(blood).
kidney in, 216-231. See also Arterial
right atrial pressure and, 176
glucose concentration of, 839, 975, 975f
blood pressure, renal control of.
shunt, 500, 503
feeding and, 871
local, 195-203, 196f, 196t. See also
skeletal muscle, 246-249, 247f, 248f
glucagon secretion and, 971, 971f
Blood flow, local control of.
splanchnic, 777-780, 778f-780f
insulin secretion and, 968-969, 968f,
long-term, 196, 200-201
sympathetic nervous system regulation of,
969f
nervous system in, 204-214. See also
170, 170f, 204-205, 205f, 206-208,
regulation of, 971-972
Arterial blood pressure, nervous
206f, 207f
hematocrit of, 169-170, 169f, 170f
system control of.
thyroid hormone effects on, 937
hepatic cleansing of, 861
nitric oxide in, 199-200
tissue vascularity and, 200
hepatic storage of, 860
norepinephrine in, 201
tissue-specific, 195-196, 196t, 199, 233-234,
hormone clearance from, 909-910
vasopressin in, 202
233f
hormone transport in, 909
coronary, 249-256. See also Coronary
turbulent, 165, 165f, 166
O-A-B, 451-453, 452f, 452t
blood flow.
ultrasonic Doppler flowmeter
osmolar clearance of, 357
definition of, 164
measurement of, 164-165, 165f
Pco2 of, 386, 399, 399f, 400f
during exercise, 195, 198, 201, 246-249,
units of, 164
pH of, 384, 384t
247f, 248f
vasa recta, 321, 354-355, 355f
alveolar ventilation and, 389, 389f
eddy currents of, 165f, 166
vascular endothelial growth factor and,
measurement of, 398-400, 399f, 400f
electromagnetic flowmeter measurement
200-201
phosphorus in, 979
of, 164, 165f, 220
vascular resistance and, 164, 164f,
physiologic shunt of, 500, 501, 503,
endothelin effect on, 202
167-169, 168f-170f
503f
epinephrine effect on, 201
vasoconstrictor control of, 201-202
reservoirs for, 179-180, 180f
gastrointestinal, 777-780, 778f-780f
vasodilator theory of, 196-197
Rh, 453-454
glucose effect on, 198
vasodilators and, 196-197, 202
shunted, 500, 501, 503, 503f
hepatic, 860
vasopressin effect on, 202
silicone containers for, 466-467
histamine effect on, 202
velocity of, 162, 165-166, 166f
sludged, 281-282
humoral control of, 201-203
vessel diameter and, 167-168, 168f
transfusion of
hydrogen ion effect on, 202-203
Blood iron buffer, 862
blood typing for, 453, 453t
in collateral circulation, 201
Blood pressure. See Arterial blood pressure.
in erythroblastosis fetalis, 454
in polycythemia, 428
Blood transfusion
in shock, 286
laminar, 165-166, 168
blood typing for, 453, 453t
in thrombocytopenia, 465
local control of, 195-203
citrate in, 467
reactions to, 452-453, 454
adenosine in, 196-197
in erythroblastosis fetalis, 454
typing of, 451-454, 452f, 452t, 453t
angiogenesis and, 200-201
in shock, 286
1074
Index
Blood transfusion (Continued)
Body mass index, 872
Brain (Continued)
in thrombocytopenia, 465
Bohr effect, 508, 508f
injury to, 764
reactions to, 452-453, 454
Bone(s), 980-983
Cheyne-Stokes breathing in, 522
Blood types
absorption of, 982-983, 982f, 985
concussive, 767
determination of, 453, 453t
calcification of, 981-982, 985
shock and, 285
O-A-B, 451-453, 452f, 452t
calcium of, 371-372, 981, 989
interstitial fluid pressure of, 187
Rh, 453-454
compact, 981
limbic system of, 731-738, 732f. See also
Blood vessels. See also Arteriole(s); Artery
cystic disease of, 990
Limbic system.
(arteries); Capillary (capillaries);
deconditioning of, in space travel, 544
lymphatic system of, 765, 765f
Vein(s).
deposition of, 982-983, 982f, 985
metabolism of, 767
blood clotting in, 458-459, 458f. See also
estrogen effects on, 1017
negative pressure in, 178
Coagulation.
fracture of, 983
neurohormonal systems of, 730-731, 730f
compliance of, 171-172
growth hormone effects on, 923
oxygen requirement for, 767
delayed compliance of, 172, 173f
in rickets, 991
thyroid hormone effects on, 936, 937
distensibility of, 171-180
in vitamin C deficiency, 878
tumor of, 765, 899
arterial pressure and, 173-176,
organic matrix of, 981
vasomotor center of, 205f, 206-207, 206f,
173f-176f
osteoporosis of, 992
212-213
units of, 171
parathyroid hormone effects on,
Brain stem, 728, 729f
venous pressure and, 176-179,
986-987
autonomic control areas of, 758-759, 758f
177f-179f
phosphate of, 981
bulboreticular facilitatory area of, 678,
platelet plug repair of, 458
remodeling of, 982-983, 982f
728-729, 729f
stress-relaxation of, 172, 173f
sound conduction through, 652
cerebral cortex feedback to, 729
sympathetic innervation of, 204, 205f,
stress on, 983
in feeding behavior, 870
206-208, 206f
structure of, 983f
in hearing, 657-658
thromboembolism of, 465-466
tensile strength of, 981
in motor function, 678, 688, 691-692, 691f,
trauma-induced constriction of, 457
testosterone effects on, 1005-1006
697
Blood volume, 293
vitamin D effects on, 985
in pain sensation, 600-601, 600f
antidiuretic hormone secretion and, 360,
Bone fluid, 987
in taste, 665-666, 666f
360f, 360t
Bone marrow
neospinothalamic tract to, 600-601, 600f
arterial pressure and, 172, 172f, 219-220,
aplasia of, 426, 437
paleospinothalamic tract to, 600f, 601
220f
B lymphocyte preprocessing in, 441,
reticular excitatory area of, 601, 728-729,
atrial natriuretic peptide and, 379-380
448
729f
atrial reflex effect on, 212
lymphocytes in, 440
reticular inhibitory area of, 729, 729f
cardiac output and, 237, 241-242, 242f
macrophages in, 434
reticular nuclei of, 691-692, 691f
sympathetic nervous system response
red blood cell production in, 420-423,
transection of, 673
to, 377
420f, 421f
arterial pressure and, 758
distribution of, 161-162, 162f
white blood cell production in, 430-431,
positive supportive reaction and, 682
in aortic valve lesions, 273
430f, 435, 437
spastic rigidity and, 692
in cardiac failure, 259-260, 380
Botulinum toxin, end plate potential effects
vestibular nuclei of, 691-692, 691f
in liver cirrhosis, 381
of, 88, 88f
Brain tumor, 765, 899
in mitral valve lesions, 273
Botulism, swallowing in, 819
Brain waves, 740, 741-743, 742f
in nephrotic syndrome, 381
Bowel. See Colon.
Braxton Hicks contractions, 1037
in pregnancy, 381, 1035, 1035f
Bowman’s capsule
Breast(s)
in valvular disease, 273
anatomy of, 309f, 310, 310f
anatomy of, 1039f
in varicose veins, 381
hydrostatic pressure of, 318, 318f
development of, 1038-1039
increases in, 380-381
Boyle’s law, 545, 546f
estrogen effects on, 1017
mean circulatory filling pressure and, 239,
Bradycardia, 147-148, 148f
oxytocin effects on, 929
239f
Bradykinin
progesterone effects on, 1018
measurement of, 296
blood flow and, 202
Breast milk, 1041, 1041t
neonatal, 1047
gastrointestinal release of, 779
Breathing, 471-472, 472f. See also
regulation of, 375-377, 375f-377f
in asthma, 529
Respiration.
thirst and, 361, 361t
in pain, 599
Cheyne-Stokes, 522, 522f
Blood-brain barrier, 766
in renal blood flow, 322
in sleep apnea, 523
Blood-cerebrospinal fluid barrier, 766
in salivary gland secretion, 795
periodic, 522, 522f
Blue weakness, 633
mast cell release of, 436
work of, 475
Body fluids. See also Blood; Cerebrospinal
Brain. See also Brain stem; Cerebellum;
Broca’s area, 687, 687f, 717, 717f, 719
fluid (CSF); Extracellular fluid;
Cerebral cortex; Motor cortex;
loss of, 721
Interstitial fluid; Intracellular fluid;
Somatosensory cortex.
Brodmann’s areas, 589, 589f, 642
Urine; Water.
activating-driving systems of, 728-731,
Bronchial arteries, 483
compartments of, 292-293, 292f
729f, 730f
Bronchioles, 479
volume measurement of, 295-296, 295f,
brain stem in, 728-729, 729f
acetylcholine effect on, 479
296t
cerebral cortex in, 729, 729f
blood supply of, 483
corrected osmolar activity of, 294t, 298,
neurohormonal, 730-731, 730f
constriction of, 479-480, 529
351
sensory input to, 729
dilation of, 479
distribution of, 296-299, 298f
arterioles of, 763
epinephrine effect on, 479
in neonate, 1048
blood flow in. See Cerebral blood flow.
norepinephrine effect on, 479
in potential spaces, 305-306
capillaries of, 182, 763
terminal, 479
indicator-dilution measurement of,
cognitive activity of. See Learning;
Brown fat, 887-888, 896
295-296, 295f
Memory; Thinking.
Brown-Séquard syndrome, 606
measurement of, 295-296, 295f, 296t
during sleep, 739-741. See also Sleep.
Brunner’s glands, 805
osmolarity of, 294t, 298
edema of, 521, 766-767
Buccal glands, 793-795
osmotic equilibrium of, 298-299, 298f
glucose for, 767, 965
Buffer nerves, 210, 211f
osmotic pressure of, 52, 294t, 298
hypothalamus of. See Hypothalamus.
Buffer systems, 384, 385-388
retention of. See Edema.
in circulatory arrest, 287-288
ammonia, 393-394, 394f
transcellular, 292. See also Cerebrospinal
in epilepsy, 743-745, 744f
baroreceptor, 210, 211f
fluid (CSF).
inhibitory circuits of, 583
bicarbonate, 385-387, 386f
Index
1075
Buffer systems (Continued)
Calcium (Continued)
Capillary membrane (Continued)
intracellular, 387-388
reabsorption of, 342t, 343, 372, 372f
filtration across, 185-190, 302
isohydric principle of, 388
regulation of, 989-990
abnormalities in, 190
phosphate, 387, 393, 393f
sarcoplasmic reticulum release of, 90-91,
at arterial end, 189
protein, 387-388
91f, 99, 99f
at venous end, 189
respiratory, 389, 389f
testosterone effects on, 1005-1006
capillary pressure in, 185, 185f, 186-187,
Bulboreticular facilitatory area, 678,
urinary, 372
186f
728-729, 729f
Calcium-binding protein, 985
filtration coefficient in, 185-186, 190
Bulbourethral glands, 996, 997f
Calcium-calmodulin second messenger
interstitial fluid colloid osmotic
Bumetanide, 403, 403t
system, 914-915
pressure in, 185, 185f, 188-189
Bundle branch block, 139-140, 139f, 140f,
Calcium channel
interstitial fluid pressure in, 185, 185f,
141, 145
slow, of cardiac muscle, 105, 117
187-188
Burns
voltage-gated, 64-65
net filtration pressure in, 185-186, 189
shock and, 285
of neuromuscular junction, 86, 86f
net reabsorption pressure and, 189
water loss and, 291-292
of presynaptic membrane, 560
plasma colloid osmotic pressure in, 185,
Bursa of Fabricius, 440. See also
Calcium pump, 54, 64, 67
185f, 188
Lymphocyte(s), B.
in skeletal muscle contraction, 90
rate of, 186, 302
in smooth muscle contraction, 99
Starling equilibrium for, 189-190
Calcium-sodium channel, 64-65, 67, 773
glomerular, 182
Callus, 983
intercellular cleft of, 182, 182f
C
Calmodulin, 95
oxygen diffusion across, 183, 503-504,
C wave, 107f, 108
Calorie, 885
504f. See also Oxygen, transport of.
Cachexia, 874
Calorimetry, 885
permeability of, 183-184, 184t
Caffeine
Calsequestrin, 90
in hemorrhagic shock, 282f
exercise and, 1065
Camosine, 294t
plasmalemmal vesicles of, 182, 182f
synaptic transmission and, 570
Canal of Schlemm, 624, 624f
pores of, 182, 182f
Caisson disease, 548-549, 548f
Cancer, 40-42
diffusion through, 183-184, 184t
Cajal, interstitial cells of, 773
invasiveness of, 41
rate of diffusion across, 184
Calcification, metastatic, 990
prostatic, 1008
Starling equilibrium at, 189-190
Calcitonin, 907t, 988-989
Capillary (capillaries). See also Artery
vesicular channels of, 182, 182f
Calcitriol (1,25-dihydroxy vitamin D3), 308
(arteries); Vein(s).
Capillary pressure, 185-187, 185f, 186f
Calcium, 878t, 879, 879t
average function of, 183
abnormal, 190
active transport of, 54, 64-65
blood flow in, 4-5, 4f, 5f, 182-183
at arterial end, 189
arterial deposition of, 982
average rate of, 183
at venous end, 189
blood flow and, 202
during exercise, 246
functional, 186-187, 189
bone, 371-372, 981-982, 989
carbon dioxide diffusion from, 183,
isogravimetric measurement of, 186-187,
deficiency of, 371, 978, 979-980, 979f
504-505, 505f
186f
deionization of, 467
colloid osmotic pressure of, 185-186, 185f
measurement of, 186-187, 186f
dietary intake of, 371
cross-sectional area of, 162
micropipette measurement of, 186-187
excess of, 371, 978, 980, 990
fragility of, 878
pulmonary, 162-163, 163f, 484, 484f, 487,
excretion of, 371-372, 372f, 373t, 980, 985
function of, 161, 162
488, 488f
parathyroid hormone and, 987
glomerular, 309-310, 309f, 316-317, 316f,
Captopril, 225
extracellular, 46f, 294f, 294t, 978
317f, 317t
Carbachol, neuromuscular junction effects
in smooth muscle contraction, 99
high altitude effects on, 539-540
of, 88-89
normal value for, 7t, 371
intercellular cleft of, 182, 182f
Carbaminohemoglobin, 511
regulation of, 371-373, 372f, 373t
lymphatic, 190-191, 192f
Carbohydrates
fecal excretion of, 980
membrane of. See Capillary membrane.
cellular, 12f
fetal, 1043, 1043f
micropipette cannulation of, 186
depletion of, 874-875, 874f
hepatic excretion of, 862
neutrophil adherence to, 430f, 435
digestion of, 809, 809f. See also
in action potential, 64-65
oxygen diffusion from, 183, 503-504,
Carbohydrates, metabolism of.
in cardiac function, 105, 106, 114, 117-118
504f
energy content of, 865-866, 866t
in coagulation, 463, 467
peritubular, 309-310, 309f, 340, 340f,
fat synthesis from, 837-838, 844-845,
in 1,25-dihydroxycholecalciferol
341t
845f
formation, 984-985, 984f
plasmalemmal vesicles of, 182, 182f
fat-sparing effects of, 846
in gastrointestinal smooth muscle
potential space fluid exchange with,
formation of, 838
contraction, 773
305-306
Golgi apparatus formation of, 21
in memory formation, 724, 724f
pressure pulse in, 174-175, 174f
hydrolysis of, 808
in parathyroid hormone secretion, 988,
pressures in. See Capillary pressure.
intestinal absorption of, 815-816
988f
pulmonary. See Pulmonary capillaries.
metabolism of, 829-839. See also
in sinus nodal rhythmicity, 117-118
renal, 309-310, 309f
Glucose.
in skeletal muscle function, 76, 90-91, 91f
structure of, 181-182, 182f
adenosine triphosphate in, 829-830,
in smooth muscle function, 97, 99, 99f
surface area of, 182
830f
in vasoconstriction, 202
vasomotion of, 182-183, 197-198, 197f
anaerobic, 836-837, 882-883
intake of, 371
wall of, 181-182, 182f
citric acid cycle in, 833-835, 834f
interstitial, 294t, 978-979, 979f
Capillary filtration coefficient, 185-186, 190
control of, 836
intestinal absorption of, 815, 878, 980,
Capillary fluid shift, arterial pressure and,
cortisol effects on, 951-952
980f, 985
230f, 231
fat-sparing effects of, 846
parathyroid hormone and, 987
Capillary membrane
glucose in, 830-831, 831f
intracellular, 46f, 294f, 294t
carbon dioxide diffusion across, 183,
glycogen storage in, 831-832
loop of Henle transport of, 334-336,
504-505, 505f. See also Carbon
glycogenesis in, 832, 832f
335f
dioxide, transport of.
glycogenolysis in, 832
neonatal, 1049
concentration gradients across, 184
glycolysis in, 832-837, 833f-835f
parathyroid hormone effects on, 986-988,
diffusion through, 4-5, 4f, 183-184, 183f,
growth hormone and, 923
986f, 989-990
184t, 503-505, 504f, 505f
in Cushing’s syndrome, 958-959
plasma, 371-372, 372f, 978-979, 979f
concentration difference and, 184
insulin and, 831, 963-965, 964f, 969-970
calcitonin secretion and, 989
net, 184
liver in, 861
1076
Index
Carbohydrates (Continued)
Carbon monoxide
Cardiac muscle (Continued)
measurement of, 867
diffusing capacity of, 499, 499f
contraction of, 106-109, 107f. See also
oxidative phosphorylation in, 835-836,
diffusion coefficient of, 493
Cardiac output.
835f
hemoglobin binding of, 509-510, 510f
afterload and, 111
pentose phosphate pathway in,
solubility coefficient of, 492
arterial pressure and, 114, 114f
837-838, 838f
Carbon monoxide poisoning, 196, 510
calcium in, 114
respiratory quotient for, 867
Carbon monoxide-hemoglobin dissociation
efficiency of, 111
thyroid hormone and, 936
curve, 509-510, 510f
ejection period in, 108
vs. fat metabolism, 846
Carbon tetrachloride, renal toxicity of, 406
energy for, 111
of cell membrane, 14
Carbonate, in bone, 981
excitation-contraction coupling in, 106
protein-sparing activity of, 866
Carbonic acid, 391, 391f, 510-511
Frank-Starling mechanism in, 111-113,
storage of, 831-832
Carbonic anhydrase, 385, 419, 510-511, 879
112f, 113f, 233
triglyceride synthesis from, 844-845, 845f
in hydrogen ion secretion, 391, 391f
isovolumic (isometric) contraction in,
Carbon dioxide
Carbonic anhydrase inhibitors, 403t, 404
107f, 108, 110f, 111
alveolar, 495-496, 495f, 496f
Carboxypeptidase, 202
isovolumic (isometric) relaxation in,
partial pressure of, 500, 500f
Carboxypolypeptidase, 799-800, 810, 810f
107f, 108, 110f, 111
altitude effects on, 537, 538t
Carcinogens, 41
parasympathetic regulation of, 113, 113f
blood, 385-386, 399, 399f, 400f
Cardiac arrest, 156, 287-288
potassium in, 113-114
alveolar ventilation and, 517, 517f
Cardiac cycle, 106-109, 107f. See also
preload and, 111
in respiratory insufficiency, 524-525
Cardiac muscle, contraction of.
regulation of, 111-114, 112f-114f
blood flow and, 203
current flow during, 126-127, 126f, 127f
rhythmical excitation in, 116-122, 117f,
chemoreceptor response to, 518
electrocardiogram of, 107, 107f
118f, 120f
chemosensitive area stimulation by,
volume-pressure diagram of, 110-111,
strength of, 106, 114
516-517, 516f
110f
stretch and, 112
diffusing capacity of, 498-499, 499f
Cardiac failure, 258-267
sympathetic regulation of, 112-113, 113f
diffusion coefficient of, 493
acute effects of, 258-259, 259f
T tubules in, 106
diffusion of. See Carbon dioxide, transport
aldosterone in, 263-264
temperature effect on, 114
of.
anemia and, 427
venous return and, 111-113, 112f, 113f
dissociation curve of, 511, 511f
angiotensin in, 263
ventricular function curves for, 112, 112f
excess of, 528, 528f, 531-532
arteriovenous fistula and, 267, 267f
deconditioning of, in space travel, 544
extracellular, 4, 6, 7t, 388
atrial natriuretic factor in, 264
histology of, 103-104, 104f
Haldane effect on, 511-512, 512f
beriberi and, 267, 267f
hypertrophy of, 235, 255, 272-273, 276
hemoglobin reaction with, 511
blood volume in, 380, 485
cardiac output and, 235
in hydrochloric acid formation, 796f, 797
cardiac output curve in, 258, 259f,
in athlete, 1063
in respiration regulation, 516-517, 516f,
265-267, 265f-267f
intercalated discs of, 103-104, 104f
517f
cardiac reserve in, 260, 264-265, 265f
lactic acid use by, 837
in vasodilation, 203
Cheyne-Stokes breathing in, 522
metabolism of, 251-252
lipid solubility of, 47
chronic effects of, 259-260, 259f
myosin filaments of, 103
metabolic formation of, 388, 833-835,
compensated, 259f, 260, 265-266, 265f
oxygen consumption of, 253
834f, 837, 838f, 867
decompensated, 260-262, 261f, 266-267,
oxygen requirement of, 253
partial pressure of, 492, 493t
266f, 267f
parasympathetic innervation of, 205-206,
alveolar, 495-496, 495f, 500, 500f, 537,
definition of, 258
205f
538t
digitalis in, 262, 266-267
physiology of, 103-106, 104f, 105f
alveolar ventilation and, 519-520, 519f
edema in, 259-260, 263-264, 263f, 303
post-infarction recovery of, 260
cerebral blood flow and, 761, 762f
glomerular filtration rate in, 263
potassium channel of, 67-68, 67f, 117
during exercise, 521, 521f
graphical analysis of, 261, 261f, 265-267,
rupture of, 254
high altitude and, 438t, 537
265f-267f
scarring of, 255, 255f
in blood, 386, 399, 399f, 400f
high-output, 267, 267f
sympathetic innervation of, 205, 205f
in Cheyne-Stokes breathing, 522, 522f
in thiamine deficiency, 876
transverse tubules of, 106
in diver, 547
left-sided, 262, 487, 488-489
work output of, 110-111, 110f, 249-250
in extracellular fluid, 46f, 388
low-output, 262-263
Cardiac output. See also Cardiac output
in intracellular fluid, 46f
peripheral venous pressure in, 177
curve.
interstitial, 505, 505f
pulmonary edema in, 264, 488-489
abdominal compression reflex effect on,
respiration and, 517, 517f
quantitative analysis of, 265-267, 265f,
213
placental, 1031
266f
abnormalities of, 236-243, 236f, 238f-243f
plasma protein combination with, 511
red blood cell production in, 422
after myocardial infarction, 253-254, 253f,
pulmonary expiration of, 388
renin-angiotensin system in, 263
255, 259f, 260-262, 261f
release of
sympathetic nervous system in, 258-259,
age-related, 232, 233f, 237
Haldane effect and, 511-512, 512f
259f, 261, 261f, 265
arterial pressure and, 114, 114f, 219-220,
in citric acid cycle, 834-835, 834f
thyroid hormone effects on, 937
220f
in pentose phosphate pathway, 837, 838f
treatment of, 261-262, 266-267, 267f
at rest, 232
solubility coefficient of, 492
unilateral, 262
autonomic nervous system effects on,
transport of, 5, 183, 184, 510-512, 510f
Cardiac impulse, 116-120, 117f, 118f, 120f.
235-236, 235f
from peripheral tissue cells to
See also Heart, conductive system of.
blood volume and, 237, 241-243, 242f
capillaries, 504-505, 505f
Cardiac index, 232, 233f
cardiac hypertrophy and, 235
from pulmonary capillaries to alveoli,
Cardiac muscle, 103-114. See also Atrium
decrease in, 234, 234f, 235
504-505, 505f
(atria); Heart; Ventricle(s), cardiac.
definition of, 164, 232
Haldane effect and, 511-512, 512f
actin filaments of, 103
dinitrophenol effect on, 235, 235f
hemoglobin in, 511
action potential of, 104-106, 104f, 105f.
during exercise, 213-214, 232-233, 233f,
in bicarbonate form, 510-511
See also Action potential, cardiac
235-236, 248-250, 248f, 1063-1064,
in dissolved state, 510
muscle.
1064f, 1064t
pH and, 512
anatomy of, 103-104, 104f
during pregnancy, 1035
respiratory exchange ratio in, 512
as syncytium, 103-104, 104f
electromagnetic flowmeter measurement
water reaction with, 510-511
blood supply of, 249, 249f
of, 243, 243f
Carbon dioxide dissociation curve, 511-512,
conductive system of. See Heart,
heat production and, 885
511f
conductive system of.
high, 236-237, 236f
Index
1077
Cardiac outpu (Continued)
Carpopedal spasm, 979, 979f
Cellulose, 809, 817
hypereffective, 234-235, 234f
Carrier proteins, 46f, 47, 49-50, 53-54, 53f
Cementum, 992-993, 992f
hypoeffective, 234, 234f, 235
Cartilage, growth hormone effects on, 923
Central fovea, 626, 636
in anemia, 236-237, 236f, 427
Caspases, 40
Central nervous system, 6. See also Brain;
in arteriovenous fistula, 236, 236f,
Castration, 1008-1009
Spine; Spinal cord.
242-243, 243f, 267, 267f
Catalase, 16
analgesia system of, 602-603, 602f
in beriberi, 236, 236f, 267, 267f
Cataracts, 621
levels of, 557-558
in cardiac failure, 258-260, 259f
Catecholamines. See Epinephrine;
neuronal pools of, 578-583, 579f-583f. See
in hemorrhage, 237
Norepinephrine.
also Neuronal pools.
in hemorrhagic shock, 279-280, 279f, 281,
Cathode ray oscilloscope, 70-71, 70f
neurons of, 555, 556f. See also Neuron(s).
283f
Caudate circuit, 709, 709f
synapses of, 559-569. See also Synapse(s).
in hypertension, 228, 236f
Caudate nucleus, 688, 708, 708f
Central nervous system ischemic
in hyperthyroidism, 236, 236f
Caveolae, of smooth muscle, 99, 99f
response, 212-213, 213f, 214, 214f, 230,
in septic shock, 286
Celiac disease, 822
230f
in venous dilation, 237
Cell(s), 3, 11-25. See also specific cells.
in heart failure, 258-259
indicator dilution method measurement
ameboid movement of, 24, 24f
in hemorrhagic shock, 281
of, 244-245, 244f
apoptosis of, 40
Central retinal artery, 628
intrapleural pressure and, 237-238, 238f
carbohydrates of, 12
Central sleep apnea, 523
limits on, 234, 234f
cilia of, 24-25, 25f
Central venous catheter, 179
low, 236f, 237. See also Circulatory shock.
cytoplasm of, 13f, 14-17, 15f, 16f. See also
Central venous pressure, 176
measurement of, 243-245, 243f, 244f
Cytoplasm.
Centrifugal acceleratory forces, 541-542,
neonatal, 1047
diameter of, 18
542f
normal, 232, 233f, 234, 234f, 242, 242f
differentiation of, 40
Centrioles, 13f, 17, 37f, 38-39
oxygen Fick principle for, 244, 244f
endocytosis by, 19-20, 19f. See also
Centromere, 37f, 38
parasympathetic nervous system effects
Phagocytosis.
Centrosome, 39
on, 113, 113f, 234-235
enzyme regulation of, 36-37
Cephalins, 846-847, 847f. See also
pathologic alterations in, 236-237, 236f
filaments of, 17, 17f
Phospholipids.
peripheral resistance and, 234, 234f,
genetic regulation of, 35-36, 35f
Cerebellar hemispheres, 699, 699f, 704
236-237, 236f
growth factor effects on, 39
intermediate zone of, 699, 699f, 704-705,
quantitative analysis of, 237-245
growth of, 39-40
704f
cardiac output curve in, 234, 234f,
high altitude effects on, 540
lateral zones of, 699, 699f, 705-706
237-238, 238f, 241-243, 242f, 243f
ingestion by, 19-20, 19f
Cerebellar nystagmus, 707
venous return curve in, 238-243,
ions of, 11, 46f, 294f, 294t. See also specific
Cerebellum, 558, 698-707, 699f
239f-243f
ions.
afferent pathways to, 700-701, 700f
regulation of, 163, 375f, 376
life cycle of, 37-40, 37f
anatomy of, 699-700, 699f
resting, 232
lipids of, 12
anterior lobe of, 699, 699f
right atrial pressure and, 238, 239,
locomotion of, 24-25, 24f, 25f
basket cells of, 702
241-243, 242f, 243f
lysis of, 366
climbing fibers to, 701-702, 702f, 703
spinal anesthesia and, 242, 242f
membrane of, 12-14, 12f, 14f. See also Cell
corrective function of, 698-699, 703,
sympathetic nervous system effects on,
membrane.
704-705, 704f
112-113, 113f, 234-235, 242, 242f
nuclear membrane of, 17-18, 18f
damping in, 702, 705
thyroid hormone effects on, 937
nucleolus of, 12f, 13f, 18, 18f
deep nuclear cells of, 701-702, 702f
venous return and, 232-236, 234f, 235f. See
nucleus of, 17, 18, 18f, 27-30, 28f-30f
disorders of, 706-707
also Venous return.
organelles of, 13f, 14-17, 15f, 16f. See also
efferent pathways from, 701, 701f
volume-loading hypertension and,
at Cytoplasm.
extramotor predictive function of,
221-223, 221f, 222f
origin of, 18
706
Cardiac output curve, 234, 234f, 237-238,
proteins of, 11-12. See also Cell
flocculonodular lobes of, 696, 697, 699,
238f, 241-243, 242f, 243f
membrane, proteins of.
699f, 703-704
cardiac failure and, 258, 259f, 265-267,
red. See Red blood cell(s).
functional unit of, 701-702, 702f
265f-267f
reproduction of, 37-40, 37f
granule cells of, 702, 702f
external pressure effects on, 237-238, 238f
ribosomes of, 18
in motor function, 702-706, 704f, 712
in hemorrhagic shock, 281, 283f
size of, 40
lateral inhibition in, 702
normal, 241, 242f, 265, 265f
structure of, 12-18, 12f, 13f
mossy fibers to, 702, 702f
parasympathetic nervous system effect
volume of, 53-54, 53f
neuronal circuit of, 699f, 700-702, 701f,
on, 113, 113f
water of, 11
702f
post-infarction, 259f, 260
white. See White blood cell(s).
peripheral afferent pathways to, 700-701,
sympathetic nervous system effect on,
Cell membrane, 12-14, 12f, 13f
700f
113, 113f
active transport across, 46, 52-56. See also
planning function of, 706
Cardiac reserve
Active transport.
posterior lobe of, 699, 699f
after myocardial infarction, 255
carbohydrates of, 14
Purkinje cells of, 701-702, 702f, 703
definition of, 264
co-transport across, 55, 55f
stellate cells of, 702
exercise test of, 164-165, 165f
counter-transport across, 55
timing function of, 706
in cardiac failure, 260, 264-265, 265f
diffusion across, 46-52. See also Diffusion.
topographic representation in, 699-700,
in patent ductus arteriosus, 275
electrical potential of, 50, 50f. See also
699f
in valvular disease, 273-274
Membrane potential.
turn-on/turn-off output from, 702-703
Cardiac (cardiogenic) shock, 237, 254,
lipid bilayer of, 12-13, 14f, 45, 46f, 848
Cerebral blood flow, 199, 761-763
262-263
permeability of, 51, 58
arterial pressure effects on, 762-763,
Cardiac tamponade, 238, 238f, 254
pressure difference across, 50-51
763f
Cardiac veins, 249
proteins of, 13-14, 14f, 45
autoregulation of, 762-763, 763f
Cardiopulmonary resuscitation, 155
carrier, 13, 46f, 47, 49-50, 53-54, 53f
blockage of, 763
Cardiotachometer, 148, 148f
channel, 13, 46f, 47-49, 47f, 48f
carbon dioxide effect on, 761, 762f
Cardiotonic drugs, 261-262, 266-267, 267f
integral, 13, 14
circulatory arrest and, 287-288
Caries, 994
peripheral, 13, 14, 14f
hydrogen ion effect on, 761
Carotid body, 209, 209f, 211-212, 518, 518f
replenishment of, 22
in hemorrhagic shock, 280
mechanism of action of, 518-519
Cellular sheet, active transport through,
neuronal activity effect on, 762, 762f
Carotid sinus syndrome, 148
55-56, 56f
normal, 761
1078
Index
Cerebral blood flow (Continued)
Chemoreceptor trigger zone, in vomiting,
Chylomicrons, 816, 840-841
oxygen effect on, 761-762
823f, 824
Chyme, 784
regulation of, 761-763, 762f, 763f
Chemosensitive area, of respiratory center,
intestinal movement of, 786-788, 787f
vasomotor center response to, 212-213
516-517, 516f
neutralization of, 801
Cerebral blood pressure, negative
Chemotaxis, 24, 430f, 431, 435, 445
Chymotrypsin, 799-800, 810, 810f
centrifugal acceleratory force effects
Chenodeoxycholic acid, 804
Chymotrypsinogen, 799, 800
on, 542
Chest
Cigarette smoking
Cerebral cortex, 558. See also Motor cortex;
barrel, 529
in submarine, 550
Somatosensory cortex.
current flow in, 126-127, 127f
peptic ulcers and, 821
anatomy of, 714-715, 715f
Chewing, 781-782
respiratory effects of, 1062
association areas of, 716-718, 716f
Cheyne-Stokes breathing, 522, 522f
Cilia
Broca’s area of, 687, 717, 717f, 719, 721
Chief cells, 795-796, 797
movement of, 24-25, 25f
Brodmann’s areas of, 589, 589f, 642
Childbirth, 8-9, 929, 1036-1038, 1037f
of hair cells, 693-694, 693f
corticofugal signals of, 597
Chills, 899, 899f
of olfactory cells, 667, 667f
dominant hemisphere of, 718, 719
Chloride
of respiratory system, 480
face recognition area of, 717-718, 717f,
capillary diffusion of, 183-184, 184t
structure of, 25, 25f
718f
excretion of, 328t
Ciliary body, aqueous humor formation by,
functional areas of, 715-720, 715f-716f
extracellular, 4, 7t, 46f, 294f, 294t
623, 623f
horizontal fibers of, 714, 715f
in hemorrhagic shock, 283
Ciliary muscle, 617f, 618
in pain sensation, 601
in neuronal inhibition, 566, 566f, 567
autonomic nervous system control of, 753,
in vasomotor control, 207
interstitial, 294t
754t
information storage in, 557
intestinal absorption of, 815, 817
excessive contraction of, 607
limbic association area of, 716f, 717, 717f
intestinal secretion of, 815
Cingulate gyri, ablation of, 738
neurons of, 714, 715f, 725
intracellular, 46f, 294f, 294t
Circulation (blood), 4-5, 4f. See also Blood
nondominant hemisphere of, 719
loop of Henle transport of, 334-336, 335f
flow.
parieto-occipitotemporal association area
Nernst potential for, 565
blood volumes in, 161-162, 162f, 172,
of, 716, 716f
neuronal concentration of, 564f, 565
172f
prefrontal association area of, 716-717,
plasma, in chronic renal failure, 410, 410f
centrifugal acceleratory force effects on,
716f, 719-720
reabsorption of, 328t, 332-333, 332f, 338f
541-542, 542f
thalamus relation to, 714-715, 715f, 729,
salivary, 794
collapse of. See Circulatory shock.
729f
Chloride channel, 562, 566, 566f
collateral, 201
vasomotor center control by, 206f, 207
Chloride shift, 511
compliance in. See Vascular compliance.
vertical fibers of, 714, 715f
Chlorothiazide, 403t, 404
acute, 196-200, 196f-198f
Wernicke’s area of, 716, 718-719, 718f,
Cholecalciferol (vitamin D3), 983-984, 984f
humoral, 201-203
721, 721f
Cholecystokinin, 907t
kidney in, 216-231, 308. See also
Cerebral edema, 766-767
composition of, 799
Arterial blood pressure, renal
high-altitude, 541
in feeding, 870
control of.
respiratory effects of, 521
in gallbladder emptying, 776, 803-804
nervous system in, 204-214. See also
Cerebral ischemia, 212-213, 220-221, 691,
in insulin secretion, 969
Arterial blood pressure, nervous
763. See also Cerebral blood flow.
in pancreatic enzyme secretion, 800-801
system control of.
in circulatory arrest, 287-288
in stomach emptying, 786
dynamics of. See Blood flow.
Cerebrocerebellum, 705-706
Cholera, 815, 823
exercise-induced adjustments in, 247-249,
Cerebrospinal fluid (CSF), 292, 763-767
Cholesterol, 840, 847-848, 847f. See also
248f
arachnoidal absorption of, 765
Lipid(s).
extracorporeal, 276
circulation of, 764, 764f
cellular, 12, 13, 46f
fetal, 1042, 1045, 1046f
composition of, 765
dietary, 848
function of, 163
cushioning function of, 763-764
digestion of, 812
hemodynamics of. See Blood flow.
obstruction of, 766
extracellular, 46f, 294f
in anemia, 427
osmolarity of, 361
formation of, 847-848
in cardiac failure, 258-259, 259f
perivascular spaces and, 765, 765f
functions of, 848
in hypertension, 221-223, 222f
pressure of, 765-766
hepatic secretion of, 804-805
in persistent patent ductus arteriosus,
Cushing reaction and, 213, 213f
in adrenal steroid synthesis, 945, 946f,
274-275, 274f
removal of, headache and, 607
955
in polycythemia, 428
secretion of, 764-765, 764f
in bile salt formation, 804
in tetralogy of Fallot, 275-276, 275f
Cervix, stretch of, 1036-1037
in familial hypercholesterolemia, 850
micro-. See Arteriole(s); Capillary
CGS units, 167
in gallstone formation, 804-805, 805f
(capillaries).
Chain-terminating sequence, 31
plasma, 294f, 848
neonatal, 1045-1048, 1046f
Channel proteins, 45, 46f, 47-49, 47f, 48f
insulin deficiency and, 966
physical characteristics of, 161-163, 162f,
Chemicals. See also Drugs;
thyroid hormone effects on, 848, 936
162t
Neurotransmitters and specific
transport of, 841-842
pressures in, 162-163, 163f. See also
substances.
Cholesterol esterase, 799
Arterial blood pressure.
mutation and, 41
Cholic acid, 804
pulmonary, 161, 162f, 483-490. See also
pain elicitation by, 599, 604
Choline acetyltransferase, 563
Pulmonary circulation.
Chemiosmotic mechanism, 23, 835, 835f, 843
Cholinesterase, 563
splanchnic, 777-780, 778f
Chemoreceptor(s), 572, 573t
Chondroitin sulfate, 21
systemic (greater, peripheral), 161, 162f
aortic, 211-212, 518-519, 518f
Chordae tendineae, 109, 109f
theory of, 163
arterial pressure and, 211-212, 230, 230f
Chorea, 709, 711-712
vascular distensibility in, 171-172
carotid, 209, 209f, 211-212, 518-519,
Choroid, 628
volume-pressure curve and, 172, 172f
518f
Choroid plexus, 764-765, 764f
Circulation (lymph), 192-193, 192f, 193f
in acclimatization, 539
Chromatids, 38
Circulatory arrest, 287-288
in cardiac failure, 258-259
Chromatin, 17, 18f
Circulatory shock, 236f, 237, 278-288
in respiration, 516, 518-520, 518f, 519f
Chromosomes, 17, 36
anaphylactic, 285-286, 287
olfactory, 667-668, 667f
daughter, 37f, 39
arterial pressure in, 279, 279f
oscillation of, 214
replication of, 38
cardiogenic, 254, 262-263
Po2 stimulation of, 518-519, 518f, 519f
sex, 997, 1028
decreased cardiac output and, 278
taste, 665-665, 666f
Chronic obstructive lung disease, 501
decreased venous return and, 278
Index
1079
Circulatory shock (Continued)
Cochlear nerve, 652f, 655
Conductance, in blood flow, 167-168, 168f,
hemorrhagic, 279-284. See also
Cochlear nuclei, 657, 658, 658f
169
Hemorrhagic shock.
Codons, 30, 31-32, 32t
Conduction (heat), 891, 891f, 892
irreversible stage of, 279
Coenzyme A, 877. See also Acetyl coenzyme
Conductive system. See Heart, conductive
neurogenic, 285, 287
A.
system of.
nonprogressive stage of, 279
Cognition. See Memory; Thinking.
Cones, 626-628, 627f, 628f, 632-633, 633f,
plasma loss and, 284-285
Cold exposure, 900
635f. See also Rods.
progressive stage of, 279
acclimatization to, 896
absence of, 633, 634f
septic, 286
feeding and, 871
dark/light adaptation of, 631-632, 632f
stages of, 279
spermatogenesis and, 1001
discs of, 627, 628f
tissue deterioration in, 279
thyrotropin-releasing hormone secretion
inner segment of, 627, 628f
traumatic, 285
and, 896, 938-939
outer segment of, 627, 628f
treatment of, 286-287
Cold receptors, 607-609, 894-895
photochemistry of, 631, 631f
without decreased cardiac output, 278
Cold sensation, 572, 573t, 607-609, 608f
spectral sensitivities of, 632-633, 633f
Circulatory system, 4-5, 4f. See also
transmission of, 587, 595-597, 596f, 609,
synaptic body of, 627-628, 628f
Arteriole(s); Artery (arteries); Capillary
895
Conn’s syndrome, 342, 379, 959
(capillaries); Cardiac muscle; Heart;
Colitis, ulcerative, 823
hydrogen ion secretion in, 395
Vein(s).
Collagen
hypokalemia in, 366
Circus movements, 153, 153f, 273
digestion of, 810
sodium retention in, 379
Cirrhosis, 381, 855, 860
in ascorbic acid deficiency, 877
Consciousness, 723. See also Thinking.
bleeding in, 464
of interstitium, 184-185, 184f
Constipation, 607, 822
edema in, 303-304
of lung tissue, 473
Contact junctions, 96
Cisterna magna, 764
Collateral vessels, 201
Contact lenses, 620-621
Citrate
Colloid osmotic pressure. See also Osmotic
Continuous positive airway pressure, 523
anticoagulant action of, 467
pressure.
Contraception, 1024
blood flow and, 203
interstitial, 185-186, 185f, 188-189,
Contrecoup head injury, 764
in vasodilation, 203
193-194
Control systems, 6-9, 8f
phosphofructokinase inhibition by, 467
normal, 188
feed-forward, 9
Citric acid cycle (Krebs cycle), 23, 833-835,
of peritubular capillaries, 309, 309f, 310t,
gain of, 7-8
834f, 843
340, 340f
negative feedback of, 7, 8f
Clathrin, 19, 19f, 88
plasma, 185-186, 185f, 188
positive feedback of, 8-9, 8f
Clearance
Colon. See also Gastrointestinal tract.
Convection (heat), 891-892, 891f
creatinine, 344-345, 344t
absorption in, 817
Convergence
free-water, 357
atonic, 822
of light rays, 613-614, 614f, 615, 615f
inulin, 344, 344t, 345f, 347
bacteria of, 817
of nerve fibers, 580, 580f
Climacteric
bicarbonate secretion by, 815, 817
Cooling, 891-892, 891f
female, 1011, 1022, 1022f
disorders of, 822-823
artificial, 900
male, 1007-1008, 1008f
distension of, 822
Cord righting reflex, 682
Clonus, 678f, 679
feces formation in, 817
Cordotomy, for pain interruption, 602
Clot. See Blood clot; Coagulation;
haustrations of, 788-789, 788f
Cornea. See also Eye(s); Lens; Vision.
Coagulation factors.
mass movements of, 789
bulging of, 621
Clothing, body temperature and, 892
mixing movements of, 788-789, 788f
Coronary arteries, 249, 249f
Clotting time, 467
mucus secretion by, 806
blood flow in, 249-256, 250f. See also
Coagulation, 459-464
propulsive movements of, 789
Coronary blood flow.
abnormal, 465-466
sodium absorption in, 817
collateral, 201, 252-253, 252f, 260
calcium ions in, 463, 467
water absorption in, 817
embolus of, 252
endothelial cell surface factors in, 463
Colonoileal reflex, 776
epicardial, 250, 250f
extrinsic pathway of, 461-462, 461f, 463
Colony-forming unit-erythrocyte, 420, 421f
innervation of, 251
fibrin formation in, 460
Color blindness, 633, 634f
intramuscular, 250, 250f
intravascular, 465-466
Color pigments, 627, 631, 631f
occlusion of, 252-254, 253f. See also
intrinsic pathway of, 461f, 462-463
in dark/light adaptation, 631-632, 632f
Myocardial infarction.
positive feedback in, 8, 460-461
Color vision, 632-633, 633f, 638. See also
spasm of, 252
prevention of, 463-464
Cones.
subendocardial, 250, 250f
prothrombin activator in, 461-463, 461f,
Ishihara chart tests of, 633, 634f
thrombus of, 252
462f
Coma
vasodilation of, 250-251, 251
tests of, 467-468, 467f
acidotic, 973-974, 973f
Coronary artery angioplasty, 256
thrombin formation in, 459-460, 459f
hepatic, 862
Coronary artery disease, 252-255, 253f. See
vicious circle in, 460-461
Commissure, anterior, 722
also Myocardial infarction; Myocardial
Coagulation factors, 8, 459t, 461-463, 461f,
Communication, 720-722, 721f. See also
ischemia.
462f
Language; Speech.
acute occlusion in, 252-253
deficiency of, 464-465
Complement, 432, 445-446, 445f
atherosclerosis in, 252, 256. See also
hepatic formation of, 862
Complement complex, 443
Atherosclerosis.
vitamin K-related, 460, 464-465, 878
Compliance
collateral vessel formation in, 201,
Coarctation of aorta, 227, 274
lung, 473-475, 473f
252-253, 252f
Coated pits, 19, 19f, 88
thoracic, 474-475
pain in, 255-256
Cobalamin. See Vitamin B12 (cobalamin).
vascular, 171-172
treatment of, 256
Cocaine, exercise and, 1066
delayed (stress-relaxation), 94-95, 172,
Coronary blood flow, 249-256
Cochlea, 652-657
173f
adenosine and, 251-252
anatomy of, 652-653, 652f, 653f, 693, 693f
in hemorrhagic shock, 281
anatomy of, 249, 249f
basilar membrane in, 653, 653f
pulmonary, 483
autonomic nervous system effect on, 251
fluid movement in, 652-653, 653f, 654, 654f
pulse pressure and, 173, 174
control of, 250-251
impairment of, 660-661
Compressed air sickness, 548-549, 548f
epicardial, 250, 250f
organ of Corti of, 655-656, 655f
Computer, 558, 558f
normal, 249-250, 250f
resonance in, 653
Concentration gradient, in gas diffusion,
oxygen demand and, 250-251
sound transmission in, 594, 654-656, 654f,
491, 492f
subendocardial, 250, 250f
655f
Concussion, 767
Coronary sinus, 249
1080
Index
Coronary steal syndrome, 255
Creatinine
Decompression sickness, 548-549, 548f
Corpus callosum, 591, 719, 722
excretion of, 307, 328t, 333, 338f
Deconditioning, in space travel, 544
transection of, 722
in chronic renal failure, 410, 410f
Deep nuclear cells, of cerebellum, 701-702,
Corpus luteum, 1015
plasma, 294f, 344-345, 346f
702f
Corti
transport maximum and, 331
Deep-sea diving, 545-549
organ of, 652, 652f, 653f, 655-656, 655f
Creatinine clearance, 344-345, 344t
atmospheric pressures during, 545,
rods of, 655, 655f
Cretinism, 942
546f
spiral ganglion of, 655, 655f
Cricoid cartilage, 481, 481f
carbon dioxide pressures during, 547
Cortical collecting duct. See Renal tubules,
Crisis, febrile, 899, 899f
decompression after, 547-549, 548f
cortical collecting.
Crista ampullaris, 693f, 694
decompression sickness with, 548-549,
Corticofugal signals, 597
Cross-bridges
548f
Corticopontocerebellar pathway, 700,
of skeletal muscle, 72, 75, 75f, 76-77, 77f
gas compression during, 545, 546f
700f
of smooth muscle, 94
helium-oxygen mixture in, 549
Corticorubral tract, 688-689, 689f
Crossed extensor reflex, 680f, 681, 681f
nitrogen pressures during, 545-546
Corticorubrospinal system, 688-689, 689f
Cryptorchidism, 1001
oxygen pressures during, 546-547, 546f
Corticospinal (pyramidal) tract, 687-688,
Crypts of Lieberkühn, 791, 792f
Defecation, 757, 789-790, 790f, 822
688f
secretions of, 805-806, 805f
in spinal cord injury, 823
lesions of, 691
Cupula, 693f, 694
Defibrillation, 154-155, 155f
Corticosteroids, 944-960. See also
Curare, end plate potential effects of, 88, 88f
Deglutition, 782-784, 782f
Aldosterone; Cortisol.
Current of injury, cardiac, 141-145,
Deglutition center, 783
synthesis of, 945-947, 946f, 947t
142f-145f, 254
Dehydration
Corticosterone, 945, 947, 947t, 951
Cushing reaction, 213, 213f
hyperosmotic, 301t, 302
Corticotropes, 919, 920t
Cushing’s disease, 846
hypo-osmotic, 301, 301t
Corticotropin
Cushing’s syndrome, 958-959, 959f, 974
in diabetes mellitus type I, 973
in fat metabolism, 838
osteoporosis in, 992
low sodium intake and, 356
in gluconeogenesis, 838
Cyanide poisoning, 196, 530
seawater consumption and, 350
in pregnancy, 1034
Cyanocobalamin. See Vitamin B12
shock and, 285
Corticotropin-releasing hormone, 907t, 921,
(cobalamin).
threshold of drinking and, 362
921t, 955
Cyanosis, 531
Dehydroepiandrosterone, 946f
Cortisol, 907t, 946f, 947, 947t
in carbon monoxide poisoning, 510
Dehydrogenases, in citric acid cycle, 834
ACTH effects on, 955-957, 956f
in polycythemia, 428
Delta brain waves, 742-743, 742f, 743f
amino acid transport and, 952
in tetralogy of Fallot, 276
Dementia, 746
anterior pituitary effects of, 955
Cyclosporine, in organ transplantation, 456
Dendrites, 555, 556f, 559, 559f
carbohydrate metabolism and, 838,
Cysteine, 853f
decremental conduction in, 569
951-952
Cystinuria, 413
electrotonic conduction in, 568-569,
cellular action of, 954-955
Cystitis, 409
568f
cellular protein and, 952
Cystometrography, 312-313, 313f
spatial field of, 568
circadian rhythm of, 956, 956f
Cytochrome oxidase, 835, 835f
summation in, 569
deficiency of, 957-958
cyanide blockade of, 530
Dendritic cells, 446, 446f
excess of, 958-959, 959f
Cytokines, 29, 29f, 905
Denervation supersensitivity, 757
fat metabolism and, 846, 952
cachexia and, 874
Dense bars, 86, 86f
functions of, 950-957, 953f, 956f
Cytoplasm, 12f, 14-17, 15f, 16f
Dense bodies, 93, 93f
hepatic metabolism of, 947
endoplasmic reticulum of, 15, 15f, 20-21,
Dentate nucleus, 701, 701f, 706
hypothalamic effects of, 955
21f, 33, 34f
Dentin, 992, 992f
in allergic reaction, 954
filaments of, 17
Dentition. See Teeth.
in gluconeogenesis, 838
Golgi apparatus of, 15, 15f, 21-22
Deoxyadenylic acid, 27, 29f
inflammation-related secretion of,
lysosomes of, 13f, 16, 20, 20f
Deoxycorticosterone, 947t
952-954
microtubules of, 11, 13f, 17, 17f
Deoxycytidylic acid, 27, 29f
insulin secretion and, 969
mitochondria of, 12, 13f, 16-17, 16f. See
Deoxyguanylic acid, 27, 29f
liver proteins and, 952
also Mitochondrion (mitochondria).
Deoxyribonucleic acid (DNA), 27-30
obesity and, 952
peroxisomes of, 16
activator operator of, 35f, 36
plasma proteins and, 947, 952
secretory vesicles of, 13f, 16, 16f, 21, 21f
bases of, 27, 28f, 29-30, 29f, 30f
protein metabolism and, 952
transcription in, 30-35, 32f, 32t, 34f
deoxyribose of, 27, 28f, 29f
secretion of, 955-957, 956f
Cytosine, 27, 28f
mitochondrial, 17
stress-related, 952-954
Cytoskeleton, 11, 17
nucleotides of, 27, 29, 29f
structure of, 908f
Cytosol, 14
operon of, 35, 35f, 36
synthesis of, 946f, 947, 947t
phosphoric acid of, 27, 28f
Cortisol-binding globulin, 947
promoter of, 35, 35f
Cortisone, 947, 947t
proofreading of, 38
Co-transport, 54-55, 55f
D
replication of, 37-38
in renal reabsorption, 329-330, 330f,
Dark adaptation, 631-632, 632f
repressor operator of, 35-36, 35f
333-334
Dead space
Deoxyribonucleic acid (DNA) ligase, 38
sodium-amino acid, 55, 816
air in, 478, 495-496, 496f
Deoxyribonucleic acid (DNA) polymerase,
sodium-glucose, 55, 55f, 816, 831
alveolar ventilation and, 477-478, 478f,
38
Cotton wick method, for interstitial fluid
500-501
Deoxyribose, 27, 28f
pressure, 187
anatomic, 478
Deoxythymidylic acid, 27, 29f
Cough reflex, 480
measurement of, 477-478, 478f
Depression, 745
Coumarins, 466
physiologic, 478, 500-501
Depth of focus, 618-619, 618f
Countercurrent mechanism, of loop of
Deafness, 660-661, 661f
Depth perception, 621-622, 622f, 647-648
Henle, 351-352, 352f, 357-358
Deamination, 856, 862
Dermatomes, 596f, 597
Counter-transport, 54-55
Decarboxylases, in citric acid cycle, 834-835
Desmopressin, in central diabetes insipidus,
renal, 330, 330f, 333
Deceleratory forces, in parachute jumps, 543
357
Coupled reactions, 829
Decerebrate animal, 673, 682, 692, 758
Desoxycorticosterone, 945
Cramps, 604, 683
Decibel, 657, 657f
Detrusor muscle, 311, 311f
Cranial nerves, 750, 750f
Decidua, 1029
Deuteranope, 633, 634f
Creatine, 294t
Decompression, of diver, 547-549, 548f
Dexamethasone, 947, 947t
Index
1081
Dextran
Diffusion (Continued)
Drugs. See also specific drugs.
glomerular filtration of, 317, 317f
of lipid-insoluble substances, 47
antidiuretic hormone secretion and, 360t,
in shock, 286-287
of lipid-soluble substances, 47
361
a-Dextrinase, 809
of sodium, 60-61, 60f
diabetes insipidus and, 357
Diabetes, adrenal, 951-952
of water, 47, 51-52, 51f
ganglionic blocking, 760
Diabetes insipidus, 302
pressure difference and, 50-51, 50f
hepatic processing of, 862
central, 357
protein-channel, 47-49, 47f, 48f
immunosuppressive, 456
nephrogenic, 357-358, 414
simple, 46-47, 46f
in obesity treatment, 873-874
Diabetes mellitus, 972-976
Diffusion coefficient, of gases, 493, 498
leukopenia and, 437
acidosis in, 398
Diffusion potential, 57-58, 58f. See also
nicotinic, 759-760
arteriosclerosis and, 976
Membrane potential.
parasympathomimetic, 759
atherosclerosis and, 850
calculation of, 58
sympathomimetic, 759
complications of, 976
potassium, 60, 60f
synaptic transmission and, 570
diagnosis of, 975-976, 975f, 975t
Digestion, 808-817
D-tubocurarine, neuromuscular junction
hyperkalemia in, 366
carbohydrate, 808, 809, 809f
effects of, 89
in gigantism, 926
fat, 808, 811-812, 811f, 812f
Duct of Bellini, 311f
insulin overtreatment in, 767
hydrolysis in, 808-809
Ductus arteriosus, 1045, 1046f
ketosis in, 844
intracellular, 16, 20, 20f
closure of, 274, 1046-1047
obesity and, 974
protein, 808-809, 810-811, 810f
patent, 274-275, 274f
plasma glucose in, 331
Digitalis
Ductus venosus, 1045, 1046f
potassium distribution in, 366, 366t
in cardiac failure, 261-262, 266-267,
closure of, 1047
pregnancy and, 1050
267f
Duodenocolic reflex, 789, 822
protein wasting in, 967
toxicity of, 146, 146f
Duodenum. See also Gastrointestinal tract.
renal failure in, 407, 408, 408t
Dihydroepiandrosterone, 947t
bicarbonate ion absorption in, 815
respiratory quotient in, 867
Dihydrotestosterone, 1003
Brunner’s glands of, 805
type I, 972-974, 973f
1,25-Dihydroxycholecalciferol, 308, 907t,
calcium absorption in, 815
diagnosis of, 975-976, 975f, 975t
984-985, 984f
chloride absorption in, 815
treatment of, 976
Diisopropyl fluorophosphate, 89
gastric inhibition by, 785-786
type II, 972, 974-975, 974t
Dinitrophenol, cardiac output effects of,
hormone secretion by, 776, 786. See also
diagnosis of, 975-976, 975f, 975t
235, 235f
Cholecystokinin; Secretin.
treatment of, 976
Diopter, 616-617, 616f
mucus secretion by, 805
Diacylglycerol, 914, 914f
Dipalmitoylphosphatidylcholine, 474
tubular glands of, 791, 795f
Dialysis, 414-415, 414f, 415t
Dipeptidases, 810-811
Dwarfism, 924, 926
Dialyzing fluid, 415, 415t
Disse, spaces of, 859
Dynein, 25
Diapedesis
Disseminated intravascular coagulation, 286,
Dynorphin, 603
neutrophil, 430f, 431
466
Dysarthria, 707
reticulocyte, 422
Dissociation constant (K¢), 385-386
Dysbarism, 548-549, 548f
Diarrhea, 822-823
Diuresis, pressure, 216, 323, 341-342,
Dysdiadochokinesia, 707
acidosis and, 397
374-376, 375f, 376f
Dysequilibrium, 695-696. See also
infectious, 806, 815, 817
aldosterone escape and, 948-949, 948f
Equilibrium.
ion secretion in, 815
quantitation of, 217-220, 217f-220f
Dyslexia, 718, 721
peristaltic rush in, 788
Diuretics, 402-404, 403f, 403t
Dysmetria, 706
psychogenic, 823
alkalosis and, 398
Dyspnea, 527, 531, 532
water loss with, 292
loop, 403, 403t
Diastole, 106-107, 107f, 108, 110-111, 110f
osmotic, 402-403, 403t
blood flow during, 250, 250f
potassium-sparing, 337, 403t, 404
Diet. See also Food.
thiazide, 403t, 404
E
carbohydrates in, 866, 866t, 867. See also
Divergence
Ear
Carbohydrates.
of light rays, 614, 614f, 615, 615f
cochlea of. See Cochlea.
energy in, 865-866, 866t
of nerve fibers, 580, 580f
disorders of, 660-661, 661f
fats in, 866, 866t, 867. See also Fat(s).
Diving. See Deep-sea diving.
ossicular system of, 651-652, 652f
high-fat, 844, 848
DNA. See Deoxyribonucleic acid (DNA).
sound conduction in. See also Hearing.
in atherosclerosis prevention, 851
Donnan effect, 188, 293
from tympanic membrane to cochlea,
minerals in, 878-879, 878t, 879t
Dopamine, 563, 730-731, 730f, 907t
651-652, 652f
protein in, 866, 866t. See also Protein(s).
in Parkinson’s disease, 711
within cochlea, 654-655, 654f
vitamins in, 875-878, 875t. See also
in schizophrenia, 745-746
Eardrum, 651-652, 652f
specific vitamins.
Dopamine pathway, 710-711, 710f
Eclampsia, 1035-1036
Dieting, 873
Doppler flowmeter, 164-165, 165f
Ectoplasm, 17
Differentiation inducers, in stem cell
Dorsal column-medial lemniscal system,
Eddy currents, 165f, 166
development, 420
587, 588-589, 592-594
Edema, 185, 190, 302-305
Diffusing capacity
anatomy of, 588, 588f
cerebral, 766-767
of carbon dioxide, 498-499, 499f
fiber orientation in, 589
high-altitude, 541
of carbon monoxide, 499, 499f
in position sensation, 595, 595f
respiration and, 521
of oxygen, 498, 499f, 539, 1062
in two-point discrimination, 592-593,
fluid dynamics of, 376-377, 377f
Diffusion, 19, 46-52, 46f
593f
hypoproteinemic, in premature infant,
capillary, 4-5, 5f, 183-184, 183f, 184t
in vibratory sensation, 593
1050
electrical potential and, 50, 50f
lateral inhibition in, 593
in cardiac failure, 259-260, 261, 263-264,
facilitated (carrier-mediated), 46f, 47,
neuronal circuit of, 592, 592f
263f, 303
49-50, 49f, 831
Dorsal lateral geniculate nucleus, 640-641,
in cirrhosis, 303-304, 381
in renal reabsorption, 332-333, 332f
641f
in kidney disease, 303, 381, 406
interstitial, 185
Dorsal spinocerebellar tract, 700, 700f
in nephrotic syndrome, 381
membrane potential and, 57-58, 58f
Double helix, 28f, 29
in thiamine deficiency, 876
net rate of, 50-51, 50f
Dreaming, 739-740
intracellular, 302
of gases, 491-493, 492f. See also Carbon
Drinking, 361-362, 361t. See also Thirst.
optic disc, 766
dioxide, transport of; Gas exchange;
seawater, 350
pitting, 305
Oxygen, transport of.
threshold for, 362
preventive mechanisms in, 304-305, 340f
1082
Index
Edema (Continued)
Electrocardiogram (Continued)
Embryonic cells, ameboid movement of,
pulmonary, 488-489
leads for, 127-130, 127f-129f
24
high altitude, 540-541
axes of, 132, 132f
Emmetropia, 619, 619f
hypoxia in, 530-531, 531f
limb leads for, 127-129, 127f
Emotion
in aortic valve lesions, 273
axes of, 132, 132f
fainting and, 208
in cardiac failure, 262, 264, 488-489
vectorial analysis of, 133-134, 133f
limbic system in, 731-732, 732f
in mitral valve lesions, 273
mean electrical axis from, 137-140,
thyrotropin-releasing hormone secretion
in unilateral left-sided cardiac failure,
137f-140f. See also Mean electrical
and, 939
262
axis.
Emotional dyspnea, 532
Edinger-Westphal nucleus, 648, 648f, 649
monophasic ventricular action potential
Emphysema, 526-527, 527f, 528f
Effusion, 306
on, 124-125, 125f
acidosis in, 389
pericardial, 140
normal, 107, 107f, 128-129, 128f, 129f,
electrocardiogram in, 141
pleural, 140-141, 490
134-137, 135f-137f
gas exchange in, 498
Einthoven’s law, 128
P wave of, 107, 107f, 123, 124f, 136, 136f
Enamel, 992, 992f
Einthoven’s triangle, 127f, 128
pen recording system for, 126
Encephalitis, swallowing in, 819
Ejaculation, 757
precordial leads for, 130, 130f
End-diastolic pressure, 110, 111
Ejaculatory duct, 996, 997f
Q wave of, 107, 107f, 123, 124f
End-diastolic volume, 108
Ejection fraction, 108
QRS complex of, 107, 107f, 124-125, 125f.
Endocochlear potential, 656
Elastase, 810, 810f
See also QRS complex.
Endocrine system, 6, 905-916, 906f, 907t. See
Elastin, 473
R wave of, 107, 107f, 123, 124f, 134, 135f
also specific glands and hormones.
digestion of, 810
repolarization wave on, 123-125, 124f,
Endocytosis, 19-20, 19f
Electrical alternans, 150, 150f
125f
Endolymph, 656
Electrical potential, 50, 50f, 564f, 565. See
S wave of, 107, 107f, 123, 124f, 134, 135f
Endometriosis, 1025
also Membrane potential.
T wave of, 107, 107f, 123, 124-125, 124f,
Endometrium
endocochlear, 656
125f
blastocyst implantation in, 1029, 1029f
Electrical stimulation, in pain treatment,
abnormalities of, 145-146, 146f
cyclic changes of, 1018-1019, 1018f
603
vectorial analysis of, 134-136, 136f
Endoplasmic matrix, 15, 20
Electrocardiogram, 107, 107f, 123-130, 124f
time calibration in, 125-126
Endoplasmic reticulum, 15, 15f
after myocardial infarction, 140, 140f, 145,
vectorial analysis of, 131-146
agranular, 13f, 15, 15f
145f
during atrial depolarization, 136, 136f
functions of, 21, 21f
augmented unipolar limb leads for, 129f,
during depolarization, 134, 135f
functions of, 20-21, 21f
130-131
during repolarization, 134-136, 136f
granular, 13f, 15, 15f
bipolar limb leads for, 127-129, 127f
in bipolar limb leads, 133-134, 133f
functions of, 20, 21f
axes of, 132, 132f
in bundle branch block, 139-140, 139f,
lipid formation by, 21, 21f
vectorial analysis of, 133-134, 133f
140f
protein formation by, 20, 21f
chest leads for, 130, 130f
in cardiac myopathy, 140, 140f
ribosome attachment to, 33, 34f
current flow and, 126-127, 126f, 127f
in current of injury, 141-145, 142f-146f
Endoplasmic reticulum vesicles, 15, 15f
current of injury effect on, 141-145,
in myocardial infarction, 143-145, 143f,
Endorphins, 602-603, 956-957, 957f
142f-145f
144f
Endothelial glycocalyx, 463
depolarization wave on, 123-125, 124f,
in premature ventricular contractions,
Endothelin, in blood flow, 202, 322, 322t
125f
151, 151f
Endothelium-derived relaxing factor,
Einthoven’s law and, 128
in T wave abnormalities, 145-146, 146f
199-200
Einthoven’s triangle in, 127f, 128
in ventricular hypertrophy, 138-139,
Endotoxin, in hemorrhagic shock, 282
electrical axis from, 137-138, 137f
138f, 139f
End-plate potential, 87-88, 87f, 88f
heart rate on, 126
lead axes in, 132, 132f
End-stage renal disease, 407-408, 407f, 408t.
in atrial fibrillation, 155-156, 155f
mean electrical axis and, 137-140,
See also Kidneys, failure of.
in atrial flutter, 156, 156f
137f-140f
End-systolic volume, 108
in atrioventricular block, 148-150, 149f
of abnormal electrocardiogram,
Energy. See also Adenosine triphosphate
in bradycardia, 147-148, 148f
140-146, 140f, 142f-146f
(ATP).
in bundle branch block, 139-140, 139f,
of normal electrocardiogram, 134-137,
aerobic, 882
140f
135f-137f
anaerobic, 836-837, 882-883
in cardiac myopathy, 140, 140f
principles of, 131-134, 132f, 133f
daily requirement for, 885-886
in current of injury, 141-145, 142f-146f
projected vector in, 133, 133f
excess intake of, 872-874. See also
in digitalis toxicity, 146, 146f
vector direction in, 131-132, 132f
Obesity.
in electrical alternans, 150, 150f
vectorcardiogram for, 136-137, 137f
fats for, 966, 966f
in intraventricular block, 150, 150f
voltages in, 125
for food processing, 887
in mild ischemia, 145-146, 146f
Electroconvulsive therapy, 745
for physical activity, 872, 887, 887t
in myocardial infarction, 143-145, 143f,
Electroencephalography
for respiration, 475
144f
during seizures, 744, 744f
for thermogenesis, 887-888
in paroxysmal tachycardia, 151-152
during sleep, 741-743, 742f, 743f
free, 829
in pericardial effusion, 140
during wakefulness, 743, 743f
from food, 865-866, 866t
in pleural effusion, 140-141
Electrogenic pump, 59, 60f
in action potential, 66, 66f
in premature atrial contractions, 150, 150f
Electrolytes. See Chloride; Magnesium;
in active transport, 54
in premature atrioventricular
Potassium; Sodium.
kinetic, 46, 46f, 51-52
contractions, 150-151, 151f
Electromagnetic flowmeter, 164, 165f
minimum expenditure of, 886-887,
in premature contractions, 150-151, 150f,
Electromagnetic receptors, 572, 573t
887f
151f
Electromotive force, 50, 58
Enkephalins, 602-603, 602f
in premature ventricular contractions,
Electron transport chain, 835, 835f
Enteric nervous system, 753-754, 773-776,
151, 151f
Electrotonic conduction, 568-569, 568f, 635
791-792
in pulmonary emphysema, 141
Embolus
myenteric (Auerbach’s) plexus of,
in sinoatrial nodal block, 148, 148f
air, 178, 550
774-775, 774f
in T wave abnormalities, 145-146, 146f
coronary, 252
reflexes of, 775-776
in tachycardia, 147, 148f
pulmonary, 466
submucosal (Meissner’s) plexus of,
in ventricular fibrillation, 154, 154f
Embryo. See also Fetus.
774-775, 774f
in ventricular hypertrophy, 138-139, 138f,
cell differentiation in, 40
Enteritis, 806, 822-823
139f
early nutrition of, 1029-1031, 1029f-1031f
Enterochromaffin-like cells, 797
Index
1083
Enterocytes, 805-806, 805f
Erythropoietin, 907t
Exercise (Continued)
enzymes of, 809, 810-811
in chronic renal failure, 412
in obesity, 873-874
Enterogastric reflexes, 776, 785-786, 799
in red blood cell production, 422-423,
in valvular disease, 273-274
Enterokinase, 800
422f
life-prolonging effect of, 1066
Enzyme(s), 13. See also specific enzymes.
secretion of, 308
mean systemic filling pressure during, 248,
activation of, 36-37
Esophageal reflux, 783, 784
249
catalytic action of, 884, 884f
Esophagus
muscles in, 1055-1061. See also at Muscle
digestive, 782
achalasia of, 819
(skeletal).
hydrolytic, 16
disorders of, 819
adenosine triphosphate system of,
in phagocytosis, 432
dryness of, 361, 361t
1056-1057
inhibition of, 36
enlargement of, 819
aerobic system of, 1057-1058
intestinal, 806, 809, 809f
in swallowing, 782f, 783-784
anaerobic system of, 883
intracellular, 36-37
mucus secretion by, 795
blood flow of, 246, 247f, 1062-1063,
of complement system, 445-446, 445f
peristalsis of, 783
1063f
of sperm, 1000
smooth muscle of, 783
endurance of, 1056
pancreatic, 799-800, 801, 809, 809f
striated muscle of, 783
fast-twitch, 1060-1061
peptidase, 810-811, 810f
Estradiol. See also Estrogen.
glycogen recovery of, 1059, 1059f
proteolytic, 432, 1000
in female reproduction, 1016-1018, 1016f
glycogen-lactic acid system of, 1057
Enzyme-linked immunosorbent assay, for
structure of, 908f
heat production and, 885
hormone measurement, 916, 916f
Estrogen, 907t
hypertrophy of, 1060-1061, 1060f
Eosinophil(s), 421f, 429-430, 430f
blood transport of, 1016
metabolic systems of, 883, 1056-1059,
concentration of, 430
bone effects of, 1017
1057f, 1058t
cortisol effects on, 954
breast effects of, 1017
nutrients for, 1059-1060, 1059f
in parasitic infection, 436
deficiency of, 1017
phosphocreatine-creatine system of,
Eosinophil chemotactic factor, 436, 529
electrolyte effects of, 1018
1057-1059, 1058t
Ephedrine, 759
fallopian tube effects of, 1017
power of, 1056
Epididymis, 996, 997f, 999
in female reproduction, 1011, 1013f,
recovery of, 1058, 1059, 1059f
Epilepsy, 743-745
1016-1018, 1016f
resistance training for, 1060, 1060f
focal, 744-745
in spermatogenesis, 999
slow-twitch, 1060-1061
grand mal, 743-744, 743f, 744f
insulin secretion and, 969
strength of, 1056, 1060, 1060f
petit mal, 743f, 744, 744f
liver metabolism of, 1016-1017
norepinephrine secretion during, 247, 846
psychomotor, 744f, 745
metabolic rate and, 1018
oxygen consumption during, 233, 233f,
surgery in, 745
placental secretion of, 1032-1033
247, 506-507, 506f, 520, 520f,
Epinephrine, 907t
protein deposition and, 857, 1017-1018
1061-1062, 1061f, 1062f
basal secretion of, 756
skin effects of, 1018
oxygen debt in, 883, 1058-1059, 1059f
blood flow and, 201, 251, 322, 322t
synthesis of
oxygen diffusing capacity in, 498, 499f,
bronchial effects of, 479
in female, 1016
1062
coronary blood flow and, 251
in male, 1004
oxygen transport during, 507-508, 509
in bronchiolar function, 479
uterine effects of, 1017
oxygen-hemoglobin dissociation curve
in exercise, 247, 846
vaginal effects of, 1017
during, 508, 508f
in gastrointestinal function, 775
Ethacrynic acid, 403, 403t
potassium replacement in, 1065
in glycogenolysis, 832
Ethylene glycol, renal toxicity of, 406
pulmonary blood flow during, 485f,
in renal blood flow, 322, 322t
Evans blue dye, for plasma volume, 296,
486-487, 487f
intracellular potassium and, 366
296t
pulmonary oxygen uptake during, 503,
plasma fatty acids and, 970
Evaporation, 891f, 892
503f
plasma glucose and, 970
Excitation-contraction coupling
respiration during, 516, 520-521, 520f,
potassium effects of, 366
in cardiac muscle, 106
1061-1062, 1061f, 1062f
receptors for, 752
in skeletal muscle, 89-91, 90f, 91f
sodium chloride replacement in, 1065
secretion of, 207-208, 750, 751-752, 755,
Exercise, 1055-1066
sympathetic nervous system in, 247,
756
age-related changes and, 1064-1065
248-249, 248f
structure of, 751
amphetamines and, 1066
total body circulation during, 247-249,
tubular secretion of, 334
androgens and, 1065-1066
248f
Epithelium
anemia and, 427
venous return curve in, 248f, 249
active transport through, 55-56, 56f
arterial pressure during, 208, 213-214,
ventilation-perfusion ratio during, 501
in vitamin A deficiency, 875
235-236, 248-249, 248f
ventilatory rate during, 1061-1062, 1062f,
of glomerular capillary, 316, 316f
blood flow during, 246-249, 247f, 248f
1064
Eplerenone, 403t, 404
blood gases during, 1062
Exercise testing, in heart disease, 264-265
Equilibrium, 692-697
body heat during, 1065
Exhaustion atrophy, 1050
central nervous system in, 696-697,
caffeine and, 1065
Exocytosis, 20, 21, 21f, 906
696f
cardiac output during, 213-214, 232-233,
Exophthalmos, 940, 940f
exteroceptive information in, 696
233f, 235-236, 248-250, 248f,
Expiration, 471-472, 472f, 476f. See also
flocculonodular lobes in, 696, 697
1063-1064, 1064f, 1064t
Breathing; Inspiration; Respiration.
neck proprioceptors in, 696
cardiovascular system and, 1062-1065,
maximum flow of, 525-526, 525f, 526f
static, 694-695
1063f
regulation of, 515
vestibular apparatus in, 692-696,
cigarette smoking and, 1062
Expiratory compliance curve, 473, 473f
693f-695f
cocaine and, 1066
Expiratory flow, maximum, in respiratory
vestibulocerebellum in, 703-704
cramps during, 683
insufficiency, 525-526, 525f, 526f
visual information in, 696
energy expenditure during, 887, 887t
Expiratory reserve volume, 475, 476f
Equilibrium point, of arterial pressure,
epinephrine secretion during, 247, 846
Extracellular fluid, 3-4, 292f, 293
217-218
fat utilization during, 846
calcium in, 7t, 46f, 294f, 294t, 371-373,
Erection, 757
glucagon secretion and, 971
372f, 373t, 978
Erythremia, 428
growth hormone secretion and, 924, 924f
carbon dioxide in, 6, 7t
Erythroblast, 421f, 422
heart disease and, 1064-1065
composition of, 293-295, 294f, 294t
Erythroblastosis fetalis, 421f, 427, 454,
heart rate during, 1063-1064, 1064f, 1064t
corrected osmolar activity of, 294t, 298
1048
heatstroke and, 1065
hypertonic, 298f, 299-301, 300f
Erythrocytes. See Red blood cell(s).
hyperkalemia and, 366
hypertonic saline effect on, 298f, 299-301
1084
Index
Extracellular fluid (Continued)
Eye(s) (Continued)
Fanconi’s syndrome, 414
hypotonic, 298, 298f, 299, 300f
innervation of, 648-649, 648f
Farsightedness, 619, 619f
in cardiac failure, 380
intraocular fluid of, 623-625, 623f, 624f
Fastigial nucleus, 701, 701f
in nephrotic syndrome, 381
lens of, 617-618, 617f. See also Lens.
Fat(s). See also Lipid(s); Triglycerides.
in pregnancy, 381
light adaptation of, 631-632, 632f
brown, 887-888, 896
increase in, 380-381
fixation, 627f, 645-648, 646f
carbohydrate formation from, 838
ions of, 7, 7t, 46f
flicking, 646, 647f
depletion of, 874-875, 874f
magnesium in, 373
occipital cortex in, 645-646, 646f
deposition of, estrogen and, 1018
measurement of, 296, 296t
premotor area in, 687, 687f, 696
digestion of, 811-812, 811f, 812f
osmolarity of
pursuit, 647
emulsification of, 811
potassium distribution and, 366-367
saccadic, 646-647
energy content of, 865-866, 866t, 966, 966f
regulation of, 358-363
semicircular duct effect on, 696, 697
extracellular, 46f, 294f
aldosterone in, 362-363, 363f
turning, 647
formation of, 837-838, 842, 844-845, 845f
angiotensin II in, 362-363
myopic, 619-620, 619f
hydrolysis of, 808
antidiuretic hormone in, 358-361,
negative centrifugal acceleratory force
in gallbladder emptying, 803-804
359f, 360f, 360t, 362, 362f
effects on, 542
intestinal absorption of, 816
thirst in, 361-363, 361t, 362f, 363f
nystagmus of, 707
intracellular, 46f
osmotic pressure of, 294t, 298
oblique muscles of, 645, 645f
metabolism of. See also at Triglycerides.
oxygen in, 6, 7t
optics of, 617-622, 617f, 618f
bile salts and, 804-805
pH of, 46f
parasympathetic innervation of, 648, 648f
carbohydrate lack and, 846
abnormalities of, 396-397, 396t. See also
pressure of, 624-625, 624f
cortisol and, 846, 952
Acid-base balance; Acidosis;
pupil of, 618-619, 618f, 649-650
growth hormone and, 846, 922-923
Alkalosis.
pursuit movements of, 647
in diabetes mellitus type I, 973-974
potassium in, 365-371, 366f, 366t, 369f,
reading-related saccadic movements of,
insulin and, 965-966, 969-970
370f
646-647
liver in, 842, 844, 861-862
aldosterone blockade and, 370, 370f
rectus muscles of, 645, 645f
regulation of, 846
aldosterone secretion and, 369-370,
reduced, 617
thyroid hormone and, 846, 936
369f, 370f
refractive errors of, 619-621, 619f, 620f
neutral, 12f, 46f, 294f, 850. See also
normal, 370, 370f
refractive power of, 617, 617f
Lipid(s).
renal potassium secretion and, 368, 369f
repressed, 648
protein-sparing activity of, 866
sodium in
saccadic movements of, 646-647
storage of, 842, 845. See also Adipose
aldosterone effect on, 362-363, 363f
slow drift of, 646
tissue.
angiotensin II effect on, 362-363
stereopsis of, 622, 622f, 643, 647-648
insulin and, 965
antidiuretic hormone effect on, 379
sympathetic innervation of, 648-649, 648f
tissue. See Adipose tissue.
deficiency of, 363
tremor of, 646
Fat cells (adipocytes), 12, 842. See also
osmolarity from, 358
turning movements of, 647
Adipose tissue.
regulation of, 358-363, 358f, 373-374
ultraviolet light exposure of, 607
Fatigue
salt-appetite mechanism and, 363
visual acuity of, 621, 621f
neuromuscular junction, 88
transport of, 4-5, 5f
skeletal muscle, 82
volume of
synaptic, 570
abnormalities of, 301-302, 301t
Fatty acids, 840. See also Triglycerides.
angiotensin II effects on, 377-378
F
acetyl-CoA conversion to, 844-845, 845f
antidiuretic hormones and, 379
Fabricius, bursa of, 440. See also
beta-oxidation of, 842-843, 843f
arterial pressure and, 219-220, 220f
Lymphocyte(s), B.
blood transport of, 841
atrial natriuretic peptide and, 379-380
Face, trigeminal neuralgia of, 605-606
direct absorption of, 816
measurement of, 295-296, 295f, 296t
Face recognition, 717-718, 717f, 718f
epinephrine effect on, 970
regulation of, 373-381, 375f-378f
Facilitation
extracellular, 4
sodium excretion and, 373-374
neuronal, 568, 579, 579f
free, 841
sympathetic nervous system effects on,
synaptic, 557
glucose conversion to, 964-965
377
memory and, 724-725, 724f
a-glycerophosphate combination with,
Extracorporeal circulation, 276
Factor I (fibrinogen), 188, 459f, 459t, 460
845, 845f
Extrapyramidal motor system, 689
Factor II (prothrombin), 459-460, 459f, 459t
insulin and, 965
Eye(s). See also Lens; Retina; Vision.
Factor III (tissue factor), 459t, 461, 461f
intracellular, 22, 22f. See also Adenosine
accommodation of, 617-618, 617f, 649
Factor IV. See Calcium.
triphosphate (ATP).
aging of, 618
Factor V (proaccelerin), 459t
plasma, 841
astigmatic, 620, 620f
Factor VII (serum prothrombin conversion
transport of, 841
autonomic innervation of, 648-649, 648f
accelerator), 459t
Fatty streak, 849
cataracts of, 621
Factor VIII (antihemophilic factor), 459t,
Feces
cerebellar nystagmus of, 707
465
colonic movements of, 788-789, 788f
dark adaptation of, 631-632, 632f
Factor IX (plasma thromboplastin
composition of, 817
depth perception of, 647-648
component), 459t, 462f, 463
expulsion of, 757, 789-790, 790f, 822, 823
emmetropic, 619, 619f
Factor X (Stuart factor), 459t, 461f, 462,
formation of, 817
examination of
462f, 463
water in, 292, 292t
for color vision, 633, 634f
Factor XI (plasma thromboplastin
Feedback
for depth perception, 621-622, 622f
antecedent), 459t, 462f, 463
negative, 7, 8f
for field of vision, 644-645, 644f
Factor XII (Hageman factor), 459t
positive, 8-9, 8f
for visual acuity, 621, 621f
Factor XIII (fibrin-stabilizing factor), 459t,
Feedback gain, in arterial pressure
ophthalmoscopic, 622-623, 622f
460
regulation, 217-218
extraocular muscles of, 645-648, 645f-647f
Fainting, 208
Feed-forward control, 9
fixation movements of, 627f, 645-648, 646f
Fallopian tubes, 1011, 1012f
Feeding. See also Food.
flicking movements of, 646, 647f
estrogen effects on, 1017
abnormal, 872-873. See also Obesity.
fluid system of, 623-625, 623f, 624f
inflammation of, 1025
adipose tissue feedback in, 871
glaucoma of, 624-625
ovum in, 1027, 1028-1029, 1028f
aminostatic theory of, 871
Horner’s syndrome of, 650
progesterone effects on, 1018
brain stem function in, 870
hyperopic, 619, 619f
Fallot, tetralogy of, 139, 275-276, 275f
cholecystokinin and, 870
image formation in, 617
Familial hypercholesterolemia, 850
environmental temperature and, 871
Index
1085
Feeding (Continued)
Flatus, 817, 825
Functional residual capacity (Continued)
excess, 872-874
Flexor reflex, 680-681, 680f, 681f
in asthma, 529
gastrointestinal factors in, 870
Flowmeter
Furosemide, 403, 403t
ghrelin and, 870
Doppler, 164-165, 165f
glucostatic theory of, 871
electromagnetic, 164, 165f, 243, 243f
hypothalamic regulation of, 733f, 734,
Fluid(s)
867-870, 868f, 869f, 869t, 871-872
body. See Blood; Body fluids;
G
inhibition of, 868-870, 869f, 870
Cerebrospinal fluid (CSF);
G cells, 797-798. See also Gastrin.
insulin and, 870
Extracellular fluid; Interstitial fluid;
G (GTP-binding) proteins, in second
lipostatic theory of, 871
Intracellular fluid; Pleural fluid;
messenger system, 561-562, 561f, 911,
long-term regulation of, 871-872
Urine; Water.
911f, 913-914, 913f
mechanics of, 781-784, 782f, 870
dialyzing, 415, 415t
GABA (gamma-aminobutyric acid), 563
neural regulation of, 733f, 734, 867-870,
transcellular, 293. See also Cerebrospinal
GABA (gamma-aminobutyric acid)
868f, 869f, 869t
fluid(CSF).
pathway, 710-711, 710f
neurotransmitters in, 868-870, 869f, 869t
Fluorine, 879, 994
Gain, of control system, 7-8
oral receptors in, 871
9a-Fluorocortisol, 945, 947t
Galactose, intestinal absorption of, 816
peptide YY and, 870
Fluorosis, 879
Gallbladder
quantity of, 870-872
Flush, 899, 899f
bile storage in, 802-803, 803f
short-term regulation of, 870-871
Foam cells, 849
emptying of, 776, 803-804
stimulation of, 868-870, 869f
Focal epilepsy, 744-745
Galloping reflex, 683
thermogenic effect of, 887
Focal length, 614f, 615
Gallstones, 804-805, 805f
Femoral vein, thrombosis of, 466
Focal line, 614, 615
of papilla of Vater, 821-822
Fenestrae, of glomerular capillary
Focal point, 614-615, 614f
Gamma-aminobutyric acid (GABA), 563
membrane, 316, 316f
Focus, depth of, 618-619, 618f
Gamma-aminobutyric acid (GABA)
Ferritin, 425, 425f, 862
Folic acid, 875t, 877
pathway, 710-711, 710f
Fertility, 1024-1025
deficiency of, 423
Ganglia, basal. See Basal ganglia.
Fertilization, 1000, 1027-1029, 1028f
in red blood cell maturation, 423
Ganglion cells, 634, 635, 635f, 636-638
Fetal cells, in Parkinson’s disease, 711
Follicle-stimulating hormone, 907t, 918, 919f,
action potential in, 637-638, 637f
Fetus, 1042-1044. See also Neonate;
920t
color signals from, 638
Newborn.
in female reproduction, 1011, 1012-1013,
excitation of, 637-638, 637f, 638f
calcium metabolism in, 1043, 1043f
1013f
lateral inhibition and, 638, 638f
circulation of, 1042, 1045, 1046f
in male reproduction, 999, 1007
on-off response of, 637-638, 637f
erythroblastosis of, 421f, 427, 454, 1048
testosterone inhibition of, 1007
types of, 637
gastrointestinal tract of, 1043
Food. See also Carbohydrates; Fat(s);
W, 637
growth of, 1042, 1043f
Protein(s), dietary.
X, 637
iron metabolism in, 1043-1044, 1043f
appetite for, 781
Y, 637
kidneys of, 1043
chewing of, 781-782
Ganglionic blocking drugs, 760
metabolism in, 1043
colonic mixing of, 788-789
Gangrene, hyperbaric oxygen therapy in, 550
nervous system of, 1043
digestion of, 808-817. See also Digestion;
Gap junctions, 92-93, 559, 771
phosphate metabolism in, 1043, 1043f
Gastrointestinal tract.
Gas(es). See also Carbon dioxide; Nitrogen;
red blood cells of, 1042
energy in, 865-866, 866t
Oxygen.
respiratory system of, 1042-1043
free energy of oxidation of, 829
Boyle’s law of, 545, 546f
sex of, 1028
gas-forming, 825
compression of, under water, 545, 546f
vitamins in, 1044
gastric mixing of, 784
diffusing capacity of, 498-499, 499f
Fever, 898-900, 898f
gastric peristalsis of, 785-786
diffusion coefficient of, 493, 498
brain lesions and, 899
hunger for, 733f, 734, 781, 785, 867
diffusion of, 491-493, 492f. See also Gas
interleukin-1 in, 898-899
intake of, 781-784, 782f, 867-872. See also
exchange.
metabolic rate and, 887
Feeding.
concentration gradient in, 491, 492f
rheumatic, 271
intestinal mixing of, 786-787, 787f
net, 491, 492, 493
Fibrin, 459f, 460
peristalsis of, 787-788
through respiratory membrane,
antithrombin action of, 463-464
storage of
496-499, 496f, 497f, 499f
formation of, 460
in colon, 788-789, 788f
exchange of. See Gas exchange.
in platelet plug formation, 458
in stomach, 784, 784f
gastrointestinal, 817, 825
Fibrinogen (factor I), 459f, 459t, 460,
swallowing of, 782-784, 782f
in fluids, 492, 493
855-857, 855f. See also Plasma proteins.
taste of. See Taste sensation.
in tissues, 493
osmotic pressure and, 188
thermogenic effect of, 887
partial pressures of, 492. See also Carbon
Fibrinolysin, 1019
volume of, stomach emptying and, 785
dioxide, partial pressure of; Oxygen,
Fibrin-stabilizing factor (factor XIII), 458,
Foramen ovale, 1045, 1046f
partial pressure of.
459t, 460
closure of, 1046
pressures of, 491-492
Fibroblast growth factor, 200
Forced expiratory vital capacity, 526, 526f
solubility coefficient of, 492
Fight or flight reaction, 758
Forced expiratory volume during first
solubility of, 493, 498
Filaments
second (FEV1), 526, 526f
Gas bubbles, in decompression sickness,
actin
Fourth power law, 168
548-549
of cardiac muscle, 103
Fourth ventricle, 763, 764, 764f
Gas exchange, 491-501
of skeletal muscle, 72-73, 76-78, 76f
Fovea, 626, 627f, 635, 635f, 636
alveolar vs. atmospheric air composition
of smooth muscle, 93-94, 93f
Fracture, 683, 983
in, 493-496, 493t, 494f-496f
cellular, 17, 17f
Frank-Starling mechanism, 111-113, 112f,
carbon dioxide transport in, 5, 510-512,
myosin
113f, 233
510f-512f. See also Carbon dioxide,
of cardiac muscle, 103
Free energy, 829
transport of.
of skeletal muscle, 72-73, 73f, 75-78, 75f
Free-water clearance, 357
diffusion in, 491-492, 496-499, 496f, 497f,
of smooth muscle, 93-94, 93f
Freon gas, in submarine, 550
499f
proteoglycan, 184-185, 184f, 305
Fröhlich’s syndrome, 1009, 1009f
in pneumonia, 528, 528f
Filtration coefficient, 185-186, 190, 340, 341t
Frostbite, 900
net diffusion in, 491, 492, 493
Filtration fraction, 316, 319, 319f, 340, 346
Fructose, intestinal absorption of, 816
oxygen transport in, 502-510, 503f-506f,
Fitness. See Exercise.
Functional residual capacity, 475, 476-477,
508f, 509f. See also Oxygen, transport
Flagellum, of sperm, 17, 17f, 25
476f, 494, 529
of.
1086
Index
Gas exchange (Continued)
Gastrointestinal tract (Continued)
Gastrointestinal tract (Continued)
physics of, 491-493, 492f
enteric nervous system of, 773-776, 774f,
villi of, 812-813, 812f, 813f
physiologic shunt in, 500
792
blood flow of, 779, 779f
respiratory exchange ratio in, 512
hormones in, 776
vitamin B12 absorption from, 423
respiratory membrane in, 496-499, 496f,
neurotransmitters of, 775-776
Gene(s), 27-30. See also Deoxyribonucleic
497f. See also Respiratory membrane.
fetal, 1043
acid (DNA).
respiratory unit in, 496, 496f
filling of, 870
agglutinogen, 451-452, 452t
ventilation-perfusion ratio and, 499-501,
food intake regulation by, 870
in obesity, 873
500f
glands of, 791-793, 792f
mutation in, 38, 41
Gas gangrene, hyperbaric oxygen therapy
gastric, 795-799, 796f, 798f
regulation of, 35-37, 35f
in, 550
secretions of, 792-793, 792f. See also
regulatory, 36
Gastric banding surgery, 874
Gastrointestinal tract, secretions
structural, 35, 35f
Gastric bypass surgery, 874
of.
Gene segments, in lymphocyte processing,
Gastric glands, 784
stimulation of, 791-792
442
secretions of, 795-799, 795f, 796f, 798f
hormones of, 776, 779, 792. See also
Genetic code, 29-30, 29f, 30f
inhibition of, 798-799
Cholecystokinin; Gastrin; Secretin.
Ghrelin, 870
interdigestive, 799
hypothalamic input from, 868, 868f
Gigantism, 926
phases of, 798, 798f
in premature infant, 1050
Gigantocellular neurons, 730f, 731
Gastric inhibitory peptide, 776, 786, 969
iron absorption in, 426
Glaucoma, 624-625
Gastric juice. See also Hydrochloric acid.
layers of, 771-772, 772f
Global aphasia, 721
neutralization of, 820-821
lipid absorption in, 840
Globulins, 855-857. See also at
visceral pain and, 604
local intermittent constrictive
Immunoglobulin; Plasma proteins.
Gastrin, 907t
contractions of, 777
osmotic pressure and, 188
composition of, 799
lymphocytes in, 440
Globus pallidus, lesions of, 709
insulin secretion and, 969
mixing movements of, 777
Glomerular filtration, 316-320
secretion of, 776, 797-798
motility of, 771-773, 772f
capillary membrane in, 316-317, 316f,
stomach emptying and, 785
hormone effects on, 776, 937
317f, 317t
Gastritis, 819-820
mucous glands of, 791, 795
solute electrical charge in, 317, 317f
Gastrocolic reflex, 776, 789, 822
mucus of, 791, 793, 797
solute size in, 317, 317t
Gastroenteric reflex, 787
myenteric plexus of, 777
Glomerular filtration coefficient, 318, 320t
Gastroesophageal sphincter, 783
nervous system of, 753-754, 791-792
Glomerular filtration rate (GFR), 315, 316,
Gastroileal reflex, 787
nutrient absorption in, 5
317-320, 318f
Gastrointestinal tract, 771, 772f. See also
obstruction of, 285, 824-825, 824f
angiotensin II effect on, 322, 322t, 325
Colon; Small intestine; Stomach.
pain in, 603-605, 603f, 605f
autacoid effect on, 322-323, 322t
absorption in
parasympathetic nervous system effects
autoregulation of, 323-325, 323f, 324f
anatomical basis of, 812-813, 812f, 813f
on, 775, 779
creatinine clearance for, 344-345, 344t
calcium, 980, 980f, 985, 987
peristalsis of, 776-777, 777f
decrease in, 318-320, 319f, 320t, 321-322
carbohydrate, 815-816
pyloric glands of, 797
endothelin effect on, 322, 322t
electrolyte, 817
reflexes of, 775-776
epinephrine effect on, 322, 322t
fat, 816
salivary glands of, 793-795, 794t, 795f
estimation of, 343-347, 344t, 345f, 346f
fluid, 812
secretions of, 792-793, 792f
filtration coefficient in, 318, 320t
ion, 814-815, 814f
cellular mechanism of, 792-793
formula for, 317-318
iron, 426
cephalic phase of, 798, 798f
hormonal control of, 322-323, 322t
large intestine in, 817
electrolytes in, 793
in acute renal failure, 405
lymphatic system in, 192
exocytosis in, 793
in cardiac failure, 263
nutrient, 815-816
gastric, 794t, 795-799, 796f, 798f
in chronic renal failure, 409-411, 410f
phosphate, 980, 985, 987
gastric phase of, 798, 798f
in hyperkalemia, 367
protein, 816
hepatic, 794t, 802-805, 803f, 803t,
increase in, 318
small intestine in, 813-816, 814f
805f
inulin clearance for, 344, 344t, 345f, 347
sodium absorption in, 949
hormonal stimulation of, 792
macula densa feedback and, 374
vitamin B12, 423
inhibition of, 798-799
measurement of, 343-347, 344t, 345f,
water, 814, 817
intestinal phase of, 798, 798f
346f
afferent sensory fibers from, 775
large intestine, 794t, 806
nitric oxide effect on, 322, 322t
amino acid absorption from, 854
pancreatic, 794t, 799-802, 800f, 802f
norepinephrine effect on, 322, 322t
antiperistalsis of, 824
parasympathetic stimulation of, 792
normal, 323
atony of, 756
phases of, 798, 798f
plasma creatinine for, 344-345, 346f
autonomic reflexes of, 757
pyloric, 797
prostaglandin effect on, 322-323, 322t
blood flow of, 777-780, 777f-780f
saliva, 793-795, 794t
quantification of, 343-347, 344t, 345f,
blood supply to, 778, 778f
small intestine, 794t, 805-806, 805f
346f
calcium absorption from, 878
sympathetic stimulation of, 792
sodium excretion and, 374
capillaries of, 182
vesicles for, 793
sympathetic nervous system effect on,
complex glands of, 791, 792f
water in, 793
321-322
crypts of Lieberkühn of, 791, 792f
sensory nerves of, 775
tubular reabsorption and, 327-328, 328t,
digestion in, 808-817. See also Digestion.
smooth muscle of, 771-773, 772f
339
disorders of, 819-825
contraction of, 772-773, 772f
tubuloglomerular feedback and, 323-325,
esophageal, 819
intermittent contraction of, 777
324f
gastric, 819-821, 820f
resting membrane potential of, 773
Glomerular tufts, 182
in thiamine deficiency, 876
slow waves of, 772-773, 772f
Glomerulonephritis, 405-406, 405t, 407,
large intestine, 822-823
spike potentials of, 772f, 773
408-409, 408t
obstructive, 824-825, 824f
tonic contraction of, 773
autoimmune, 448
small intestine, 821-822
splanchnic circulation of, 777-780,
chronic, 409
vomiting in, 823-824, 823f
778f
edema in, 303
distention of
sympathetic nervous system effects on,
Glomerulosclerosis, 408, 408f
peristalsis and, 776
775, 779-780
Glomerulotubular balance, 323, 339, 374
thirst and, 361, 361t
syncytium of, 771-772
Glomerulus
electrolytes in, 793
tubular glands of, 791, 795f
olfactory bulb, 669
Index
1087
Glomerulus (Continued)
Glucose transporters, 967, 968f
Growth hormone, 907t, 918, 919f, 920t,
renal, 309f, 310
Glucose-1-phosphate, 832, 832f
921-927
capillaries of, 309, 309f, 316-317, 316f,
Glutamate, 564
abnormalities of, 926-927, 927f
317f, 317t
in pain sensation, 601
aging-related changes in, 927
colloid osmotic pressure of, 318-319,
Glutamic acid, 853f
amino acid transport and, 922
319f
Glutamine, 853f, 856, 856f
bone growth and, 923
hydrostatic pressure of, 319-320, 319f
renal metabolism of, 393, 394, 394f
carbohydrate utilization and, 923
Glomus cells, 519
Gluten enteropathy, 822
cartilage growth and, 923
Glossopharyngeal neuralgia, 605-606
Glycerol-3-phosphate, 842
daily variation in, 924, 924f
Glucagon, 907t, 970-971
a-Glycerophosphate, 841, 845, 845f, 846
diabetogenic effects of, 923
in glycogenolysis, 832
Glycine, 563, 853f
fat utilization and, 846, 922-923
insulin secretion and, 969
Glycinuria, 413
growth effects of, 922, 922f, 923-924
regulation of, 971
Glycocalyx, 463
growth hormone-releasing hormone
secretion of, 832, 961, 970-971
Glycogen, 12, 13f
effect on, 925-926
somatostatin inhibition of, 971
breakdown of, 832, 970
in dwarfism, 926
Glucagon-like peptide, 870
formation of, 832, 832f
in spermatogenesis, 999
Glucocorticoids, 944. See also Cortisol.
storage of, 831-832, 836, 838, 861
insulin interaction with, 923, 967, 967f,
circadian rhythm of, 956, 956f
Glycogen synthase, 964
969
functions of, 950-957, 953f, 956f
Glycogenesis, 832, 832f
ketogenic effect of, 923
in fat metabolism, 838, 846
Glycogenolysis, 832, 970
metabolic effects of, 922-923
in gluconeogenesis, 838
Glycolipids, 14
metabolic rate and, 887
in organ transplantation, 456
Glycolysis, 22-23, 832-837, 833f-835f, 882
protein synthesis and, 857, 922
in protein metabolism, 857
acetyl coenzyme A formation in, 833,
protein-sparing effect of, 922
in shock, 287
833f
regulation of, 924-926, 924f, 925f, 925t
synthesis of, 946f, 947, 947t
anaerobic, 836-837
RNA formation and, 922
Glucokinase, 831, 967, 968f
citric acid cycle in, 833-835, 834f
somatostatin effect on, 925-926
Gluconeogenesis, 838, 857
in muscle, 79, 883
tissue protein deposition and, 922
cortisol effects on, 838, 951
oxidative phosphorylation in, 835-836,
Growth hormone inhibitory hormone, 907t,
glucagon in, 970
835f
921, 921t
hepatic, 832, 832f, 861
pyruvic acid formation and, 833, 833f
glucagon inhibition and, 971
insulin effects on, 965
rate of, 836
insulin inhibition and, 971
renal, 308
Glycoproteins, 13, 14
secretion of, 961
Glucose, 830-831, 831f
Glycosuria, 413
Growth hormone-releasing hormone, 907t,
blood, 839, 975, 975f
Goblet cells, 805, 805f
921, 921t, 925-926
cortisol effects on, 951-952
Goiter, 939, 940, 941
Growth inducers, in stem cell development,
feeding and, 871
Goldblatt hypertension, 226-227, 226f
420
glucagon secretion and, 971, 971f
Goldman equation (Goldman-Hodgkin-
Guanethidine, 759
in diabetes mellitus type I, 973
Katz equation), 58
Guanine, 27, 28f, 29, 29f
insulin secretion and, 968-969, 968f,
Golgi apparatus, 13f, 15, 15f
Guanosine monophosphate, cyclic (cGMP)
969f
functions of, 21-22, 21f
in rod function, 631
regulation of, 971-972
vesicle formation by, 21, 21f
second messenger activity of, 912
renal blood flow and, 325
Golgi tendon organ, 675, 679-680, 679f
blood flow and, 198
Golgi tendon reflex, 679-680
capillary permeability to, 183-184, 184t
Gonadotropes, 919, 920t
cortisol effects on, 951
Gonadotropin-releasing hormone, 907t, 921,
H
co-transport of, 55, 55f, 816, 831
921t
H band, 73f
energy release from, 832-838, 833f-835f,
in female reproduction, 1011, 1020
Habituation, memory and, 723, 724, 736
838f. See also Adenosine
in male reproduction, 1006
Hageman factor (factor XII), 459t
triphosphate (ATP), formation of.
Grand mal epilepsy, 743-744, 743f, 744f
Hagfish, kidney of, 216
epinephrine effect on, 970
Granule cells
Hair
extracellular, 4, 7t, 46f, 294f
cerebellar, 702, 702f
estrogen effects on, 1018
facilitated diffusion of, 50, 55, 55f, 831
olfactory, 668, 670
testosterone effects on, 1005
fatty acid conversion of, 964-965
Granules, secretory, 13f, 16, 16f, 21, 21f
Hair cells
formation of. See Gluconeogenesis.
Granulocyte(s), 429-430, 430f. See also
of macula, 693-694, 693f
glucagon effect on, 970
Basophil(s); Eosinophil(s);
of organ of Corti, 652, 655-656, 655f
in brain metabolism, 767, 965
Neutrophil(s).
Hair end-organ, 586
insulin effects on, 831
in inflammation, 435
Haldane effect, 511-512, 512f
interstitial, 294t
life span of, 431
Hand skills, motor cortex and, 687, 687f,
intestinal absorption of, 816
Granulocyte colony-stimulating factor, 435,
691
intracellular, 22, 22f, 46f, 294t. See also
436f
Haustrations, 788-789, 788f
Adenosine triphosphate (ATP).
Granulocyte-monocyte colony-stimulating
Hay fever, 450
liver uptake of, 964-965
factor, 435, 436f
Head. See also Brain.
muscle storage of, 963-964
Granulosa cell tumor, 1023
contrecoup injury to, 764
muscle transport of, 964, 964f
Graves’ disease, 940
gravity-related orientation of, 693, 693f
muscle uptake of, 963
Gravitational pressure. See Hydrostatic
rotation of, 687, 687f, 694, 694f, 695-696,
phosphorylation of, 831
pressure.
695f
placental, 1031
Ground substance, 21
visual orientation of, 647
plasma, 294f
Growth, 1051-1052, 1051f
Headache, 606-607, 606f
reabsorption of, 328t, 330, 330f, 331, 331f,
fetal, 1042, 1043f
Head-down position, in shock, 287
338f
growth hormone effects on, 922, 922f,
Hearing, 651-661
urinary, 413, 975
923-924
abnormalities of, 660-661, 661f
in diabetes mellitus type I, 973
insulin and, 967, 967f
auditory cortex in, 657-659, 659f. See also
Glucose phosphatase, 830-831, 831f, 964
neonatal, 1051f, 1052
Auditory cortex.
Glucose solution, body fluid effects of,
testosterone effects on, 1005-1006
auditory nerve stimulation in, 655-656
301
thyroid hormone effects on, 936
centrifugal signals in, 660
Glucose tolerance test, 975, 975f
Growth factors, 39
decibel threshold of, 657, 657f
1088
Index
Hearing (Continued)
Heart (Continued)
Heme, 424, 424f, 425f
endocochlear potential in, 655
pressures in, 107f, 108. See also at Atrium
Hemianopsia
frequency range of, 657, 657f
(atria); Ventricle(s), cardiac.
bitemporal, 645
hair cells in, 655-656, 655f
reservoir function of, 179
homonymous, 645
impedance matching in, 651-652
sinus node of, 116-118, 117f, 120-121. See
Hemiballismus, 709
loss of, 660-661, 661f
also Sinus (sinoatrial) node.
Hemodialysis, 414-415, 414f, 415t
loudness determination in, 656-657, 657f
sounds of. See Heart sound(s).
Hemodynamics. See Blood flow; Circulation
organ of Corti in, 655-656, 655f
sympathetic innervation of, 205, 205f
(blood).
place principle in, 656
systolic stretch of, 253-254, 253f, 254
Hemoglobin, 419-420, 879
sound conduction in
thyroid hormone effects on, 937
buffer action of, 6, 419, 506f, 507
from tympanic membrane to cochlea,
valves of. See Heart valves.
carbon dioxide transport with, 511
651-652, 652f
vasomotor center control of, 206-207
carbon monoxide combination with,
within cochlea, 654-655, 654f
ventricles of. See Ventricle(s), cardiac.
509-510, 510f
sound direction in, 660
work output of, 110-111, 110f, 249-250
concentration of, 420
sound frequency in, 654-655, 654f, 656,
Heart attack. See Cardiac failure;
deficiency of, 426-427. See also Anemia.
657, 657f
Myocardial infarction.
destruction of, 426
stimulus intensity and, 594
Heart beat, 106-109, 107f
excessive deoxygenation of, 531
tests of, 660-661, 661f
Heart block, 148-150
fetal, 1030-1031, 1031f
Heart. See also at Cardiac.
first degree, 149, 149f
formation of, 424-425, 424f
atria of. See Atrium (atria).
second degree, 149, 149f
high altitude effects on, 538f, 539
atrioventricular bundle of, 104, 118-119,
third degree, 149, 149f
iron in, 424-425, 424f. See also Iron.
118f, 119f
Heart failure. See Cardiac failure.
oxygen combination with, 420, 424-425,
atrioventricular valves of, 107f, 108, 109,
Heart murmurs, 271f, 272
505-510. See also Oxygen-
109f
in patent ductus arteriosus, 275
hemoglobin dissociation curve.
autonomic nervous system control of,
Heart rate
altitude effects on, 538, 538f
205-206, 205f, 754, 754t
abnormal, 147-148, 148f
during exercise, 507-508
blood return to. See Venous return.
atrial pressure and, 212
reversibility of, 505-507, 507f
collateral circulation in, 252-253, 253f
during exercise, 1063-1064, 1064f, 1064t
saturation of, 507, 507f
conductive system of, 116-120, 117f. See
hypothalamic regulation of, 733, 733f
structure of, 424, 424f
also Action potential, cardiac
on electrocardiogram, 126
subunits of, 424, 424f
muscle.
temperature effects on, 114, 147, 148f
tubular, 455
abnormalities of, 147-156. See also
Heart sound(s), 109-110, 269-272
Hemoglobin S, 424, 427
Arrhythmias.
auscultation of, 270-271, 270f
Hemolysins, 453
acetylcholine effects on, 121
first, 110, 269-270, 270f, 271f
Hemolysis
after myocardial infarction, 254
fourth, 270, 271f
in transfusion reactions, 453, 454, 455
atrioventricular node in, 117f, 118-119,
in patent ductus arteriosus, 275
jaundice in, 454, 455, 864
118f, 120f
phonocardiogram of, 271, 271f, 272
Hemolytic disease of the newborn, 421f,
control of, 120-122
second, 110, 269-270, 270f, 271f
427, 454
electrocardiography of, 123-130. See
third, 270, 271f
Hemophilia, 465
also Electrocardiogram.
Heart valves
Hemorrhage. See also Bleeding;
internodal pathway in, 117f, 118, 118f,
congenital lesions of, 271-272
Hemorrhagic shock.
120f
circulatory dynamics of, 274-276, 274f,
arterial pressure and, 224, 224f
norepinephrine effects on, 122
275f
brain, 765
parasympathetic nervous system effects
lesions of, 271-272, 271f
renal response to, 376
on, 121
cardiac reserve and, 273-274
renin-angiotensin system response to, 224,
parasympathetic stimulation of, 121
circulatory dynamics in, 272-274
224f
Purkinje fibers in, 117f, 119, 120f, 121
exercise effects on, 273-274
sympathetic nervous system in, 172
sinus node of, 116-118, 117f, 120-121,
murmurs of, 271f, 272
Hemorrhagic shock, 279-284, 279f
120f. See also Sinus (sinoatrial)
rheumatic fever-related lesions of, 271
acidosis in, 283-284
node.
sounds of, 269-272, 270f, 271f. See also
arterial pressure in, 279-280, 279f, 280f
sympathetic nervous system effects on,
Heart sound(s).
capillary permeability in, 282
121-122
Heart-lung machines, 276
cardiac output in, 279-280, 279f, 281,
sympathetic stimulation of, 121-122
Heat, 46. See also Temperature;
283f
ventricular transmission in, 119-120,
Thermogenesis.
compensated, 280-281, 280f
120f
loss of, 890-894, 890f, 891f
endotoxin in, 282
electrocardiography of, 123-130. See also
production of, 884-885, 889-890
irreversible, 284, 284f
Electrocardiogram.
Heat acclimatization, 893-894, 900, 1065
liver damage in, 283, 283f
failure of. See Cardiac failure.
Heat receptors, 607-609, 608f
negative feedback mechanisms in, 281
flaccidity of, 114
Heat sensation, 572, 573t, 607-609, 608f
nonprogressive, 280-281, 280f
hypereffective, 234-235, 234f, 238, 238f
tissue damage and, 599, 599f
positive feedback mechanisms in,
hypertrophy of, 235, 272-273, 276
transmission of, 609
281-284, 282f
in athlete, 1063
Heatstroke, 899-900
progressive, 280f, 281-284, 282f, 283f
hypoeffective, 234, 234f, 235, 238, 238f
exercise and, 1065
sludged blood in, 281-282
in persistent patent ductus arteriosus,
Heavy metals, renal toxicity of, 406
sympathetic nervous system in, 279-280,
274-275, 274f
Helicobacter pylori, peptic ulcer and, 821
279f
in tetralogy of Fallot, 275-276, 275f
Helium
tissue necrosis in, 283, 283f
intercalated discs of, 103-104, 104f
diffusion coefficient of, 493
treatment of, 286-287
ischemic disease of, 252-255, 253f. See
solubility coefficient of, 492
vasomotor failure in, 281
also Myocardial infarction.
Helium-oxygen mixture, in diving, 549
Hemosiderin, 425, 425f
pain in, 255-256
Hematocrit, 169, 169f, 293
Hemostasis, 457-459
recovery from, 254-255, 255f
high altitude effects on, 539
clot formation in, 458-459, 458f, 459t. See
T wave in, 145-146, 146f
in polycythemia vera, 428
also Coagulation.
output of. See Cardiac output.
vascular resistance and, 169-170, 170f
platelet plug formation in, 457-458
papillary muscles of, 109
Hematopoietic stem cells, 420-422, 421f
vascular constriction in, 457
parasympathetic innervation of, 205-206,
committed, 420, 421f
Henderson-Hasselbalch equation, 386
205f
pluripotential, 420, 421f
Henle, loop of. See Loop of Henle.
Index
1089
Heparin
Homeostasis (Continued)
Hydrogen bonds, 29, 29f, 854
anticoagulant action of, 464, 466,
kidneys in, 5
Hydrogen ions
467
liver in, 5
active transport of, 54
mast cell release of, 436, 464
musculoskeletal system in, 5
alveolar ventilation and, 389, 389f,
Hepatic artery, 860, 860f
nervous system in, 5-6
519-520, 519f
Hepatic coma, 862
reproductive system in, 6
arteriolar effects of, 202-203
Hepatic vein, 859, 860, 860f
respiratory system in, 4f, 5
blood flow and, 202-203
Hepatitis, bleeding in, 464
Homonymous hemianopsia, 645
cerebral blood flow and, 761, 762f
Hepatocyte growth factor, 861
Horizontal cells, 633, 635f, 638f
chemoreceptor response to, 518
Hereditary factors, in cancer, 41
Hormone(s), 6, 905-916, 906f, 907t. See also
chemosensitive area excitation by, 516
Hering-Breuer inflation reflex, 515-516
specific glands and hormones.
counter-transport of, 55
Hering’s nerve, 209, 209f
amine, 906, 907t, 908
deficit of. See Alkalosis.
Hermaphroditism, 1049
blood concentration of, 908-909
excess of. See Acidosis.
Herpes zoster infection, 605
measurement of, 915-916, 916f
formation of, 511, 835, 835f
Hexamethonium, 242, 759, 760
blood transport of, 909
in hydrochloric acid formation, 796-797,
Hexokinase, 831
classes of, 906, 907t
796f
Hexose monophosphate, 294t
cyclical variations in, 909
in pancreatic juice secretion, 800, 800f
High altitude
enzyme-linked immunosorbent assay for,
in respiration regulation, 516-517, 516f,
acclimatization at, 519
916, 916f
517f
angiogenesis and, 539-540
feedback regulation of, 909
intercalated cell secretion of, 337, 392,
bone marrow response to, 422
gastric inhibition by, 786
392f
cellular effects of, 540
gastrointestinal, 776, 779
mitochondrial, 835-836, 835f
cerebral edema and, 541
in renal blood flow, 322-323, 322t
net renal excretion of, 394-395
hematocrit and, 539
in smooth muscle contraction, 98
normal level of, 383, 384, 384t
hemoglobin and, 539
mechanism of actions of, 910-914
regulation of, 383-400. See also Acid-base
hemoglobin-oxygen combination and,
receptors in, 910-912, 911f-913f
balance.
538, 538f
second messengers in, 912-915, 913f,
secretion of, 390-392, 390f-392f
hypoxia and, 537-539, 538f, 538t
913t, 914f, 914t
aldosterone excess and, 949
mental proficiency at, 539
metabolic clearance rate of, 909-910
disorders of, 413
mountain sickness and, 540-541
negative feedback regulation of, 909
intercalated cells in, 337, 392, 392f
native populations at, 540, 540f
peptide, 906, 907t
medullary collecting duct in, 338, 338f
oxygen diffusing capacity and, 539
positive feedback regulation of, 909
regulation of, 395, 395t
oxygen-hemoglobin dissociation curve at,
radioimmunoassay for, 915-916, 916f
sodium counter-transport and, 330,
507, 540, 540f
receptors for, 14, 910-912
330f, 333
Po2 and, 537-538, 538f, 538t
down-regulation of, 910
Hydrogen peroxide, 432
polycythemia and, 427-428
enzyme-linked, 911-912, 912f
Hydrogen-potassium ATPase transporter,
pulmonary circulation and, 539-540
G protein-linked, 911, 911f
368
pulmonary edema and, 540-541
gene activation and, 912, 913f, 915
Hydrolases, 16, 20
red blood cell effects of, 422, 539
ion channel-linked, 910-911
in hemorrhagic shock, 283
respiratory alkalosis and, 397, 539
sensitivity of, 910
Hydrolysis
High-molecular-weight kininogen
up-regulation of, 910
carbohydrate, 808, 809, 809f
(Fitzgerald factor), 459t
secretion of, 906, 908-909, 908f
fat, 808, 811-812, 811f
Hippocampal formation, 736
steroid, 906, 907t, 908
protein, 808-809, 810-811, 810f
Hippocampus
protein synthesis and, 915
Hydrostatic pressure
functions of, 736-737
storage of, 906, 908f
in interstitial fluid, 185, 185f, 304-305, 304f
hyperexcitable, 736-737
synthesis of, 906, 908
in pulmonary circulation, 485-487, 485f,
in learning, 737
Hormone response element, 912, 913f
486f, 488, 488f
in memory, 725-726
Hormone-sensitive lipase, 841, 846, 966
of capillaries, 185-187, 185f, 186
in schizophrenia, 746
Horner’s syndrome, 650
of peritubular capillaries, 309, 309f, 310t,
lesions of, 725
Human chorionic gonadotropin, 907t, 1007,
340, 340f
removal of, 737
1032, 1032f
of renal interstitium, 340-341, 341f
Hirschsprung’s disease, 822
Human chorionic somatomammotropin,
venous pressure and, 177-178, 177f
Histamine
1033
Hydroxyapatite, 981
blood flow and, 202
Human chorionic thyrotropin, 1034
ß-Hydroxybutyric acid, 844
bronchial effects of, 480
Human physiology, 3
25-Hydroxycholecalciferol, 983-984, 984f
enterochromaffin-like cell secretion of,
Human somatomammotropin, 907t
Hydroxyl ions, 432
797, 798
Hunger, 733f, 734, 781, 785, 867, 871. See
Hydroxymethylglutaryl-coenzyme A
in anaphylaxis, 450
also Feeding; Food.
reductase inhibitors (statins), 851
in asthma, 529
Hunger center, 867, 868f, 871
Hyperadrenalism, 958-959, 959f
in hay fever, 450
Hunger pangs, 785
Hyperalgesia, 599, 603f, 605
in urticaria, 450
Huntington’s disease, 711-712
Hyperbaric oxygen therapy, 550-551
Histamine shock, 285-286
Hyaline membrane disease, 474, 529, 1045
Hyperbarism, 545
Histidine, 853f
Hyaluronic acid, 21, 184-185
Hyperbilirubinemia, 1048, 1048f
Histiocytes, 433
Hyaluronidase, of sperm, 1000
Hypercalcemia, 371, 978, 980, 990
Histones, 36, 38
Hydrocephalus, 765, 766
Hypercapnia, 531-532
Hives, 450
Hydrochloric acid, 810
in pneumonia, 528, 528f
HLA antigens, 455-456
absence of, 820
Hypercholesterolemia, 850
Hollow capsule method, for interstitial fluid
formation of, 796-797, 796f
Hyperemia, 198
pressure, 187, 188
neutralization of, 820-821
Hyperestrinism, 1017
Homatropine, 759
pH of, 384, 384t
Hyperglycemia, in gigantism, 926
Homeostasis, 4-6
secretion of, 54, 796-797, 796f
Hyperinsulinism, 976
circulatory system in, 4-5, 4f, 5f
Hydrocortisone. See Cortisol.
Hyperkalemia, 365
control systems in, 6-9, 8f
Hydrogen atoms. See also Hydrogen ions.
aldosterone deficiency and, 947-948, 949
definition of, 4
oxidation of, 835-836, 835f, 843
cell lysis and, 366
endocrine system in, 6
in citric acid cycle, 23, 834, 834f
exercise-induced, 366
gastrointestinal tract in, 5
in pentose phosphate pathway, 837, 838f
in acute renal failure, 406
1090
Index
Hyperkalemia (Continued)
Hypothalamus (Continued)
Imipramine, 745
in Addison’s disease, 366, 370
control centers of, 733, 733f, 758-759,
Immune complexes, in glomerulonephritis,
in cell lysis, 366
758f
405-406
in diabetes mellitus, 366, 366t
cortisol effects on, 955
Immune hypersensitivity, 449-450. See also
in hypertension, 366
hunger center of, 867, 868f
Allergy.
Hypernatremia, 301t, 302
in anorexia, 874
Immune tolerance, 448
Hyperopia, 619, 619f
in behavior, 733, 733f, 734-736, 758f, 759
Immunity
Hyperparathyroidism, 990-991
in cardiovascular function, 733. See also
acquired (adaptive), 439-449
in chronic renal failure, 412
Arterial blood pressure, nervous
antigens in, 440, 441f
Hyperphagia, 867
system control of.
cell-mediated (T-cell), 440, 441f,
Hyperpyrexia, 899-900
in endocrine function, 734. See also
446-448, 446f-448f
Hypersensitivity, 449-450. See also Allergy.
Pituitary gland.
humoral (B-cell), 440, 441f, 443-446,
Hypertension, 220-223
in fever, 899
443f-445f. See also Antibody
aldosteronism and, 223
in gastrointestinal function, 734. See also
(antibodies).
angiotensin II in, 378, 378f
Feeding.
primary, 443, 443f
atherosclerosis and, 850
in obesity, 873
secondary, 443, 443f
cardiac output in, 228, 236f
in suckling, 734
lymphocytes in, 440-442, 441f. See also
circulatory function in, 221-223, 222f
in temperature regulation, 733-734,
Lymphocyte(s), B; Lymphocyte(s),
diuretics in, 229-230, 402-404, 403f, 403t
894-898, 894f, 895f, 897f
T.
drug treatment of, 229-230
in uterine contractility, 734
regulation of, 446-448, 447f
angiotensin II blockade in, 378, 378f
in vasodilation, 206f, 207-208
tolerance and, 448
glomerular filtration rate and, 325
in water regulation, 734
types of, 440
Goldblatt, 226-227, 226f
lateral areas of, 733, 733f, 734
cortisol effects in, 954
hyperkalemia in, 366
lesions of, 735
disorders of, 448, 449-450. See also
in Alzheimer’s disease, 746
magnocellular neurons of, 919
Allergy.
in baroreceptor nerve sectioning, 228
paraventricular nucleus of, 919
innate, 439. See also Phagocytosis.
in coarctation of aorta, 227
pituitary gland regulation by, 919-921,
neonatal, 1049
in lower animals, 228
920f, 921t
passive, 449
in pregnancy, 227-228, 1034
posterior, 733-734, 733f
Immunization, 439-440, 448-449
in renal ischemia, 227
preoptic area of, 894
Immunoglobulin(s). See also Antibody
left ventricular hypertrophy and, 138,
satiety center of, 867-868, 868f. See also
(antibodies).
138f
Feeding.
classes of, 444
life expectancy and, 220-221
supraoptic nucleus of, 919
mechanism of action of, 444-446, 444f,
neurogenic, 228
thyrotropin-releasing hormone of,
445f
nonsalt-sensitive, 229, 229f
938-939
specificity of, 444
obesity in, 228-229
tumor of, 873
structure of, 443-444, 444f
one-kidney, 226-227, 226f
vasomotor center control by, 206f, 207
Immunoglobulin E, 449-450
portal, 860
Hypothermia, 114, 895, 900
Immunoglobulin G, 444
pressure natriuresis in, 228-229
artificial, 900
Immunoglobulin M, 444
primary (essential), 228-230, 229f
Hypothyroidism, 938, 941-942, 942f,
Immunosuppression, in transplantation,
pulmonary, 527
1023
456
renal failure and, 407, 408, 408t, 412-413
Hypotonia, 691, 707
Impedance matching, 651
renin-angiotensin system in, 223-227,
Hypovolemia, shock and, 279-285. See also
Implanted electronic cardiac pacemaker,
223f-227f
Hemorrhagic shock.
150, 156
salt-sensitive, 229, 229f
Hypoxemia, in pneumonia, 528, 528f
Inanition, 867, 874
sodium intake and, 378, 378f
Hypoxia
Incontinence, 313-314
sympathetic nervous system in, 228
anaerobic energy utilization during,
Incus, 651, 652f
vasodilator drugs in, 229-230
882-883
Indicator-dilution method, 244-245, 244f,
volume-loading, 221-223, 221f, 222f
at birth, 1044
295-296, 295f
Hyperthyroidism, 940-941, 940f
at high altitude, 427-428, 537-539, 538f,
Infection
cardiac output in, 236, 236f
538t
brain, 765
muscle tremor in, 937
atmospheric, 530
diarrhea and, 806, 815, 817
Hypoadrenalism, 957-958
cardiac arrest and, 156
inflammation in, 434-436, 436f. See also
Hypocalcemia, 371, 978, 979-980, 979f
erythropoietin secretion and, 308
Inflammation.
Hypochlorhydria, 820
hypoventilation, 530, 531
resistance to. See Immunity; Phagocytosis;
Hypochlorite, 432
in anemia, 531
White blood cell(s).
Hypogastric nerves, 311f, 312
in pulmonary edema, 530-531, 531f
streptococcal, 434, 448
Hypogonadism
oxygen therapy in, 530-531, 531f
Inflammation, 434-436, 436f. See also
female, 1023
red blood cell production and, 422-423,
Macrophage(s); Neutrophil(s).
male, 1008-1009
422f, 427-428
appendiceal, 605, 605f
Hypokalemia, 365
synaptic transmission and, 570
complement in, 445-446
aldosterone excess and, 949
tissue oxygen deficiency and, 530-531,
cortisol effects in, 952-954
exercise-related, 1065
531f
edema and, 302
in aldosteronism, 370
work capacity and, 540
gastric, 819-820
in Conn’s syndrome, 366
in niacin deficiency, 876
Hyponatremia, 301-302, 301t
intestinal, 822-823
Hypoparathyroidism, 990
pancreatic, 821-822
Hypophosphatemia, 413, 991
I
stages of, 953
Hypophysis. See Pituitary gland.
I bands, 72, 73f, 90f
walling-off effect of, 434
Hypothalamic-hypophysial portal vessels,
I cells, 801
Inhibin, 1007, 1015, 1020
920-921, 920f
Idiopathic nontoxic colloid goiter, 941
Inositol triphosphate, 914, 914f
Hypothalamus, 558, 731, 732-736, 732f, 906f,
Idiopathic thrombocytopenia, 465
Insecticides, renal toxicity of, 406
907t. See also Limbic system.
Iggo dome receptors, 586, 586f
Inspiration, 471-472, 472f, 476f. See also
anterior, 733-734, 733f
Ileocecal sphincter, 774, 788, 788f
Breathing; Expiration; Respiration.
antidiuretic hormone synthesis in,
Ileocecal valve, 788, 788f
regulation of, 514-516, 515f
359-360, 359f
Ileum, bicarbonate secretion in, 815
work of, 475
Index
1091
Inspiratory capacity, 475, 476f
Interstitial fluid (Continued)
Iron (Continued)
Inspiratory compliance curve, 473, 473f
colloid osmotic pressure of, 185-186, 185f,
neonatal, 1049
Inspiratory reserve volume, 475, 476f
193-194, 294t, 298
oxygen combination with, 424-425, 424f
Instantaneous mean vector, 131, 132f
composition of, 293-295, 294f, 294t
regulation of, 426
Insulin, 907t, 961-970
corrected osmolar activity of, 294t, 298,
storage of, 425, 862
amino acid effects on, 969
351
transport of, 425-426, 425f
appetite and, 870
free, 185
Iron lung, 532, 532f
blood glucose effect on, 968-969, 968f,
gel form of, 185, 304-305, 304f
Ischemia
969f
osmolarity of, 294t, 298
cerebral, 212-213, 220-221, 763. See also
brain glucose and, 965
Po2 of, 503-504, 504f
Cerebral blood flow.
carbohydrate metabolism and, 963-965,
pressure in. See Interstitial fluid pressure.
in circulatory arrest, 287-288
964f, 969-970
protein in, 188-189, 193-194
myocardial, 252-255, 253f. See also
cholecystokinin effects on, 969
pulmonary, 487-489, 488f
Myocardial infarction.
cortisol effects on, 969
renal, 340-341, 341f, 351-352, 352f
pain in, 599, 604
deficiency of, 845, 966, 966f, 967. See also
Interstitial fluid colloid osmotic pressure,
renal, 227
Diabetes mellitus.
185-186, 185f, 188-189
visceral pain and, 604
energy storage and, 961-962
Interstitial fluid pressure, 185, 185f, 187-188,
Ishihara charts, 633, 634f
estrogen effects on, 969
304-305, 340f
Islets of Langerhans, 961, 962f
excess of, 976
cotton wick measurement of, 187
Isograft, 455
fat metabolism and, 965-966, 969-970
hollow capsule measurement of, 187, 188
Isogravimetric method, for capillary
gastric inhibitory peptide effects on,
in lung, 487-488, 488f
pressure, 186, 186f
969
lymphatic system and, 188, 192, 192f,
Isohydric principle, 388
gastrin effects on, 969
193-194
Isoleucine, 853f
glucagon effects on, 969
micropipette measurement of, 187
Isomaltase, 806
gluconeogenesis inhibition by, 965
negative, 187, 188-189, 194
Isoproterenol, 759
glucose transport and, 831
of brain, 187
Itching, 587
growth hormone interaction with, 923,
of kidney, 187
967, 967f, 969
subatmospheric, 187-188
intracellular potassium and, 366, 366t
Interstitium, 184-185, 184f. See also
plasma, 975
Interstitial fluid.
J
progesterone effects on, 969
Interventricular septal defect, 139
J point, in current of injury, 142-143, 143f
protein metabolism and, 857, 966-967
Intestinal lipase, 806
J receptors, 521
receptors for, 962-963, 963f
Intestine. See Colon; Gastrointestinal tract;
Jacksonian epilepsy, 744-745
resistance to, 972, 974-975, 974t
Small intestine.
Jaundice, 863-864
secretin effects on, 969
Intra-abdominal pressure, venous pressure
hemolytic vs. obstructive, 864
secretion of, 961, 967-969, 968t
and, 177
in erythroblastosis fetalis, 454
regulation of, 968-969, 968f, 969f
Intracellular fluid, 4, 46f, 292f, 293
in transfusion reactions, 455
somatostatin inhibition of, 971
calculation of, 296, 296t
neonatal, 1048
structure of, 962, 962f
composition of, 294f, 294t, 295
Jejunum. See also Gastrointestinal tract.
synthesis of, 962
corrected osmolar activity of, 294t, 298
bicarbonate ion absorption in, 815
thyroid hormone effects on, 937
hypertonic saline effect on, 298f, 299-301
chloride absorption in, 815
Insulinase, 962
osmotic pressure of, 294t, 298
hormone secretion by, 776. See also
Insulin-like growth factors, 923-924
pH of, 384, 384t, 387-388
Cholecystokinin.
Insulinoma, 976
phosphate buffer system of, 387
Joints, position receptors of, 594-595
Insulin-receptor substrates, 963
protein buffers of, 387-388
Junctional potential, 98
Intention tremor, 705, 707
Intrafusal fibers, 674, 674f, 675-676, 676f
Juxtaglomerular cells, 223-224, 324, 324f
Intercalated cells, 336-337, 336f
Intramyocardial pressure, 250, 250f
tumor of, 226
hydrogen ion secretion by, 54, 336, 392,
Intraocular fluid, 623-625, 623f, 624f
Juxtaglomerular complex, 324, 324f
392f
Intraocular pressure, 624-625, 624f
potassium reabsorption by, 337, 368
Intraventricular block, 150, 150f
Intercalated discs, 103-104, 104f
Intrinsic factor, 423, 797, 820, 877
Intercellular cleft, 182, 182f
Inulin, in water reabsorption evaluation, 339
K
Intercostal muscles, 471, 472f
Inulin clearance, 344, 344t, 345f, 347
Kallidin, 202, 779
Interdigestive myoelectric complexes, 776
Inulin space, 296
Kallikrein, 202
Interleukin-1
Iodide
salivary, 795
in fever, 898-899
deficiency of, 941
Keratoconus, 621
in inflammation, 435, 436f
oxidation of, 932, 932f
Kerckring, folds of (valvulae conniventes),
in lymphocyte activation, 442
thyroid suppression with, 939-940
812-813, 812f
Interleukin-2, 447
Iodide pump (iodide trapping), 932, 932f
Kernicterus, 454
Interleukin-3, 420
Iodine, 878t, 879, 879t, 931-932
Ketogenesis, 857
Interleukin-4, 447
radioactive, 941
Ketone bodies, 844, 966
Interleukin-5, 447
Ions, 11. See also specific ions.
Ketosis, 844, 966
Interleukin-6, 447
extracellular, 4, 7, 7t, 46f, 294f, 294t
growth hormone and, 923
Interleukin(s), 905
interstitial, 293-295, 294t
Kidneys. See also at Renal.
T-cell production of, 447, 447f
intracellular, 4, 46f, 294f, 294t
after myocardial infarction, 254
Internal capsule, 708
plasma, 293-295, 294t
anatomy of, 308-312, 309f-311f
Internal environment, 4
Iridocorneal angle, 623-624, 624f
arterial pressure control by, 216-231, 308,
Interneurons, 674-675, 674f
Iris, 97-98, 618-619, 618f
323, 341-342, 374-376, 375f, 376f
Internodal pathways, action potential
Iron, 878t, 879, 879t. See also Hemoglobin.
aldosterone escape and, 948-949,
transmission in, 117f, 118, 119f
atherosclerosis and, 850
948f
Interplexiform cells, 634
coordination bonds of, 425
atrial reflexes and, 212
Interposed nucleus, 701
daily loss of, 426
baroreceptor reflexes and, 211
Interstitial fluid, 4-5, 5f, 184-185, 184f, 292f,
fetal, 1043-1044, 1043f
experimental demonstration of, 217,
293
hepatic storage of, 425, 862
217f
accumulation of, 302-305, 376-377, 377f
intestinal absorption of, 426, 815
graphical analysis of, 217-218, 218f
calculation of, 296, 296t
metabolism of, 425-426, 425f
peripheral resistance and, 219, 219f
1092
Index
Kidneys (Continued)
Kidneys (Continued)
Lactic acid, 294f
quantitation of, 217-220, 217f-220f
function curve of, 217, 217f
formation of, 837
renal resistance and, 219
function of, 5, 307. See also at Renal
in hemorrhagic shock, 283-284
salt intake/output and, 218-219, 218f,
tubules.
Lactose, 809. See also Carbohydrates.
220, 224-226, 226f
quantification of, 343-347, 344t, 345f,
Lactotropes, 919, 920t
water intake/output and, 218-219, 218f,
346f
Language, 720-722, 721f. See also Cerebral
224-226, 226f
glomerular tufts of, 182
cortex.
arterioles of, reflex dilation of, 212
glucose synthesis by, 308
angular gyrus in, 718
artificial (dialysis), 223, 414-415, 414f
hilum of, 308
articulation of, 721-722, 721f
autonomic nervous system control of, 754t
in acclimatization, 539
Broca’s area in, 687, 687f, 717, 717f, 719,
autoregulation of, 321
in acid-base balance, 308, 390-400. See
721
blood flow of, 5, 199, 309-310, 309f,
also Acid-base balance, renal
comprehension of, 716, 716f, 719, 721
320-323, 321f, 321t, 323f, 405
regulation of.
visual, 716
angiotensin II and, 322, 322t
in cardiac failure, 259-260, 263-264
Wernicke’s area in, 716, 718-719, 718f,
arterial pressure and, 355
in hemorrhagic shock, 281
721, 721f
autacoid control of, 322-323, 322t
in homeostasis, 5
Large intestine. See Colon.
autoregulation of, 323-325, 323f, 324f
in premature infant, 1050
Larynx, 481, 481f
blood glucose and, 325
in volume regulation, 212
in swallowing, 782, 782f
calculation of, 346
infection of, 409
Lateral inhibition, 593, 593f
determinants of, 320-321, 321t
intercalated cells of, 54, 336-337, 336f,
Lateral olfactory area, 667f, 669
endothelin and, 322, 322t
368, 392, 392f
Lateral ventricle, 763, 764, 764f
epinephrine and, 322, 322t
interstitial fluid pressure of, 187
Law of mass action, 837
hormonal control of, 322-323, 322t
ischemia of, 405-406, 405t, 413
L-Deprenyl, 711
in cardiac failure, 259-260
medulla of, 309, 309f, 310f
L-Dopa, 711
medullary, 354-355, 355f
blood flow of, 354-355, 355f
Learning. See also Memory; Thinking.
myogenic autoregulation of, 325
countercurrent exchange in, 354-355,
dominant hemisphere in, 719
nitric oxide and, 322, 322t
355f
exercise-related ventilation and, 521,
norepinephrine and, 322, 322t
hyperosmotic fluid of, 350-355, 351t,
521f
oxygen consumption and, 320, 321f
352f-355f
habituation in, 736
prostaglandins and, 322-323, 322t
neonatal, 1048
hippocampus in, 737
protein intake and, 325
nephrons of, 310, 310f. See also Renal
reinforcement of, 736
vasa recta in, 354-355, 355f
tubules.
skill, 725-726
vasodilator effects on, 355
of hagfish, 216
Lecithins, 846-847, 847f. See also
calcitriol production by, 308
oxygen consumption by, 320, 321f
Phospholipids.
calices of, 309, 309f
phosphate buffer system of, 387
in fat digestion, 811
capillaries of, 184, 309-310, 309f
plasma flow of, 345-346, 346f
Left bundle branch, 117f, 118f, 119
capsule of, 308, 309f
pressure diuresis of, 216, 217-220,
Left bundle branch block, 139, 139f, 145
cortex of, 309, 309f, 310f
217f-220f
Left coronary artery, 249, 249f
1,25-dihydroxycholecalciferol formation
pyramids of, 309, 309f
Left ventricular hypertrophy, 272-273
in, 308, 984-985, 984f
size of, 308
Left ventricular volume curve, 107f, 108
disease of, 404-415. See also Kidneys,
sodium-loading function curves of, 229,
Left-to-right cardiac shunt, 274-275, 274f
failure of.
229f
Leg, pressures in, 177, 177f, 178-179, 178f
edema in, 303
stones of, 990
Lengthening reaction, 679
end-stage, 407-408, 407f, 408t
toxin injury to, 406
Lens. See also Eye(s); Lenses.
rickets in, 991
urine formation by. See Urine, formation
accommodation of, 617-618, 617f, 649
end-stage disease of, 407-408, 407f, 408t
of.
aging of, 618
erythropoietin of, 308, 422-423, 422f
vascular resistance of, 219, 291f
autonomic nervous system control of, 753,
failure of
weight of, 308
754t
acute, 404-406
Killer cells, 447-448, 447f, 448f
cataracts of, 621
dialysis for, 414-415, 414f, 415t
Kilocalorie, 885
depth of focus of, 618-619, 618f
in transfusion reaction, 455
Kinetic energy, 46, 46f, 51-52
refractive errors of, 619-621, 619f, 620f
intrarenal, 405-406, 405t
Kinins, 202. See also Bradykinin.
Lenses
physiologic effects of, 406
Kinocilium, 693-694, 693f
concave, 614, 614f
postrenal, 405t, 406
Klüver-Bucy syndrome, 737-738
contact, 620-621
prerenal, 405, 405t
Knee jerk, 678-679, 678f
convex, 613-614, 614f, 616, 616f
chronic, 406-413
Korotkoff sounds, 175
cylindrical, 614-615, 614f, 615f, 620
acidosis in, 398
Krause’s corpuscle, 573f
focal length of, 615, 615f
anemia and, 412
Krebs cycle, 23, 833-835, 834f, 843
image formation by, 616, 616f
causes of, 406-407, 407t
Kupffer cells, 433, 433f, 859
in astigmatism, 620, 620f
dialysis for, 414-415, 414f, 415t
Kwashiorkor, 866, 925, 925f
in hyperopia, 619, 619f
edema in, 411f, 412
Kyphosis, 927
in myopia, 619, 619f
glomerular disease and, 408-409
refractive power of, 616-617, 616f
hypertension and, 412-413
spherical, 614-615, 614f
interstitial disease and, 409
Leptin
nephron function in, 409-412, 410f,
L
obesity and, 871, 873
411f, 411t
Labeled line principle, 572-573
receptor for, 871, 911-912, 912f
osteomalacia and, 412
Labor, 8-9, 929, 1036-1038, 1037f
Leucine, 853f
progression of, 407-408, 407f, 408t
Labyrinth, 652, 692-693, 693f
Leukemia, 437
protein excretion in, 409
Lacrimal glands, 753, 754t
Leukocytes. See White blood cell(s).
uremia in, 411-412, 411f
Lactase, 806, 809, 809f
Leukopenia, 436-437
vascular lesions and, 408, 408f
Lactate, 294t
Leukorrhea, during menstruation, 1019
in transfusion reactions, 455
Lactation, 1038-1041, 1039f
Lever system, 82, 82f
fetal, 1043
milk composition in, 1041, 1041t
Leydig cell tumor, 1009
filtration fraction of, 316, 319, 319f, 340,
oxytocin in, 929, 1040-1041
Lieberkühn, crypts of, 805-806, 805f
346
prolactin in, 1039-1040
Ligands, 14
Index
1093
Light
Liver (Continued)
Lungs. See also Alveolus (alveoli);
adaptation to, 631-632, 632f
cells of, 859
Pulmonary ventilation; Respiration.
cone response to, 632-633, 633f
cholecalciferol-25-hydroxycholecalciferol
at birth, 1044-1045, 1045f
refraction of, 613-615, 614f, 615f
conversion in, 983-984, 984f
blood flow of. See Pulmonary circulation.
rod response to, 629-631, 630f
cholesterol secretion by, 804-805
blood volume of, 484-485
white, 633
cirrhosis of, 381, 464, 855, 860
cilia in, 480
Limbic association area, 716f, 717, 717f
edema in, 303-304
collagen fibers of, 473
Limbic cortex, 738
coagulation factor formation by, 862
collapse of, 528-529, 529f
Limbic system, 731-738, 732f
detoxification function of, 862
compliance of, 473-475, 473f
amygdala of, 737-738
endothelial cells of, 859
constriction of, 525-526, 526f. See also
cortex of, 738
estrogen metabolism of, 1016-1017
Respiratory insufficiency.
functional anatomy of, 731-732, 732f
fat storage in, 842
contraction of, 471-472, 472f
hypothalamus of, 732-736, 733f. See also
ferritin storage in, 862
dead space of, 478, 500-501
Hypothalamus.
gluconeogenesis in, 832, 832f, 861
edema of, 488-489
in behavior, 731-732, 733f, 734-736, 735f
glucose transport to, 965
in aortic valve lesions, 273
in olfaction, 669
glucose uptake by, 964-965
in cardiac failure, 264
lesions of, 735, 738
glycogen storage in, 831-832, 861
in mitral valve lesions, 273
memory and, 736
glycogenolysis in, 832, 861, 970
in unilateral left-sided cardiac failure,
punishment centers of, 725, 735-736
in carbohydrate metabolism, 861
262
reward centers of, 725, 735, 735f
in hemorrhagic shock, 283, 283f
elastin of, 473
tranquilizer effect on, 736
in homeostasis, 5
expansion of, 471-472, 472f
Linear acceleration, 695
in lipid metabolism, 842, 844, 861-862
expiratory reserve volume of, 475, 476f
Linear acceleratory forces, in space travel,
in premature infant, 1050
fibrosis of, 530
542-543, 543f
in protein metabolism, 862. See also
forced expiratory vital capacity of, 526,
Lingual lipase, 811
Amino acids; Protein(s).
526f
Lipase(s), 811
iron storage in, 862
forced expiratory volume of, 526, 526f
enteric, 811
Kupffer cells of, 433, 433f, 861
functional residual capacity of, 475,
in phagocytosis, 432
lipoprotein formation in, 842
476-477, 476f, 529
intestinal, 806
lipoprotein lipase of, 841, 842
gas exchange in. See Gas exchange.
lingual, 811
lobules of, 859, 860f
infection of, 527-528, 528f, 530
pancreatic, 799, 811-812, 812f
lymph flow of, 860
inspiratory capacity of, 475, 476f
tissue, 841, 842
macrophages of, 433, 433f, 861
inspiratory reserve volume of, 475,
Lipid(s), 12f
mast cells of, 464
476f
bile salt combination with, 804, 812, 816
metabolic functions of, 861-862
interstitial fluid pressure of, 487, 488,
formation of
neonatal, 1048
488f
by endoplasmic reticulum, 21, 21f
pain in, 604
irritant receptors of, 480, 521
from carbohydrates, 844-845, 845f
plasma protein formation by, 855, 862
lymph vessels of, 483
from proteins, 845
regeneration of, 39, 860-861
macrophages in, 433
metabolism of, 840-851
sinusoids of, 859, 860f
mast cells of, 464, 480
ATP formation in, 842-844, 843f
triglycerides in, 842
mucus in, 480
chylomicrons in, 840-841
urea formation in, 856, 862
oxygen poisoning of, 547
during exercise, 846
vascular system of, 179, 859-861, 860f
pain in, 604
fatty acid transport in, 841
vitamin storage in, 862, 875
reservoir function of, 179
hormonal regulation of, 846
Liver phosphorylase, 964
residual volume of, 475, 476f, 477, 529
in liver, 842, 844, 861-862
Lobotomy, prefrontal, 720
shock, 283
lipoproteins in, 841-842
Locomotion. See Movement.
stretch receptors of, 515-516
measurement of, 867
Locus ceruleus, 730, 730f
surface tension of, 474
regulation of, 846
Loop diuretics, 403, 403t
surfactant of, 474, 529, 1045
respiratory quotient for, 867
Loop of Henle, 310, 310f, 311f, 327
tidal volume of, 475, 476f, 477
vs. carbohydrate metabolism, 846
ascending limb of, 310, 310f, 351-352,
total capacity of, 476, 476f, 477
solubility of, 47
351t, 352f
tuberculosis of, 530
transport of, 840-842
in urine concentration, 351-352, 352f,
vital capacity of, 475-476, 476f
Lipid bilayer, 12-13, 14f, 45, 46f, 848
355, 356f
Lupus erythematosus, 448
diffusion through, 47
in urine dilution, 349-350, 349f
Luschka, foramen of, 764, 764f
Lipoprotein(a), 850
osmolarity of, 355, 356f
Luteinizing hormone, 907t, 918, 919f, 920t
Lipoprotein lipase, 841, 965
calcium reabsorption in, 372
in female reproduction, 1011, 1012-1013,
Lipoproteins, 841-842, 850. See also
countercurrent mechanism of, 351-352,
1013f, 1020-1021, 1020f
Cholesterol; Lipid(s).
352f, 357-358
in male reproduction, 999, 1007
b-Lipotropin, 956-957, 957f
descending limb of, 310, 310f, 351-352,
positive feedback regulation of, 909
Lissauer, tract of, 609
351t, 352f
testosterone inhibition of, 1007
Liver, 791, 859-864
in urine concentration, 352, 352f, 355,
Lymph
acetoacetic acid formation in, 844, 966,
356f
flow of, 192-193, 192f, 193f, 194
966f
osmolarity of, 355, 356f
hepatic, 860
amino acids of, 854, 952
in urine concentration, 351-352, 352f, 355,
in edema prevention, 305
anatomy of, 859, 860f
356f
in node, 433, 433f
arterioles of, 859
osmolarity in, 349-352, 349f, 352f
formation of, 191-192
B lymphocyte preprocessing in, 441
solute transport in, 334-336, 335f, 351,
Lymph nodes, 190, 191f
bile secretion by, 802-805, 802f, 803t. See
351t
lymphocytes in, 440, 442
also Bile; Bile salts.
water transport in, 334-336, 335f, 351,
macrophages in, 433, 433f
bleeding in, 464-465
351t
structure of, 433, 433f
blood flow of, 860
Low cardiac output failure. See Cardiac
Lymphatic capillaries, 190-191, 192f, 193f
blood storage in, 179, 860
shock.
pump of, 193
blood-cleansing function of, 861
Lumbar puncture, 765-766
Lymphatic system, 185, 190-194, 191f, 440
calcium excretion by, 862
Lumirhodopsin, 629, 629f
blockage of, 302-304
capillaries of, 182, 184
Lung volumes, 475-477, 475f, 476f, 477t
contraction of, 193
1094
Index
Lymphatic system (Continued)
Macrophages (Continued)
Mean systemic filling pressure
in edema prevention, 305
in spleen, 434, 434f
during exercise, 248, 249
interstitial fluid control and, 188, 192,
lymphokine effects on, 447
in cardiac failure, 259
192f, 193-194
phagocytosis by, 432, 435, 447
mean circulatory filling pressure and, 240
of brain, 765, 765f
pinocytosis by, 19
spinal anesthesia and, 242, 242f
of lung, 483
plasma protein processing by, 855
venous return curve and, 238, 239, 239f,
pumping of, 193
tissue, 431, 433, 433f, 435
240, 240f
valves of, 192-193, 193f
Macula densa, 310, 310f
Mechanoreceptors, 572, 573f, 573t
Lymphoblasts, 443
sodium chloride concentration in, 324,
adaptation of, 575-576, 575f
Lymphocyte(s), 429-430, 430f
324f, 374
signal transmission from, 588-589, 588f,
B, 440-441, 441f
Maculae, 693, 693f
595-596, 596f
activation of, 442
Magendie, foramen of, 764, 764f
Meconium, 1043
antigen interaction with, 442-446,
Magnesium, 878-879, 878t, 879t
Medial forebrain bundle, 731-732
443f-445f. See also Antibody
blood flow and, 202
Medial olfactory area, 667f, 669
(antibodies).
excretion of, 373
Median eminence, hormonal release at, 921,
antigen presentation by, 446, 446f
extracellular, 46f, 294f, 294t, 373
921t
bone marrow preprocessing of, 441
in bone, 981
Medulla, 558. See also Brain stem.
clone of, 442, 448
in vasodilation, 202
pyramids of, 687, 688f
interleukin effects on, 447
interstitial, 294t
respiratory center of, 514-516, 515f
liver preprocessing of, 441
intestinal absorption of, 815
reticular nuclei of, 691f, 692
memory, 443, 443f
intracellular, 4, 46f, 294f, 294t
vestibular nuclei of, 691f, 692, 701
preprocessing of, 440-441, 441f
loop of Henle transport of, 334-336, 335f
vomiting center of, 824
production of, 421f, 430-431, 430f, 440,
Magnet reaction, 682
Medullary collecting duct, 310, 310f. See also
447, 447f
Major basic protein, 436
Renal tubules.
concentration of, 430
Major histocompatibility complex, 446, 446f
Medullary reticulospinal tract, 692, 692f
cortisol effects on, 954
Malabsorption, in sprue, 822
Megacolon, 822
life span of, 431
Malignant nephrosclerosis, 408
Megaesophagus, 819
T, 440-441, 441f
Malleus, 651, 652f
Megakaryocytes, 429, 430f, 431
activation of, 442, 446, 446f
Malnutrition. See also Feeding; Food;
Megaloblasts, 427
clone of, 442, 448
Starvation.
Meiosis, 997, 998f
cytotoxic, 447-448, 447f, 448f
growth and, 925, 925f
Meissner’s corpuscles, 573f, 586, 693
helper, 442, 446-447, 447f
metabolic rate and, 887
Meissner’s plexus, 774-775, 774f
lymphokine production by, 447, 447f
urine concentration in, 353
Melanin, 956
memory, 446
Malocclusion, 994
in Addison’s disease, 958
preprocessing of, 440-441, 441f
Malonyl-CoA, 845, 845f
retinal, 628
production of, 430-431, 430f, 440
Maltase, 806, 809, 809f
Melanocortin, 874
regulatory, 448
Maltose, 809, 809f
Melanocytes, 956
suppressor, 447, 448
Mamillary bodies, 734
Melanocyte-stimulating hormone, 956-957,
surface receptor proteins of, 442, 446,
Mania, 745
957f
446f
Manic-depressive psychosis, 745
Melatonin, 1009-1010
thymus gland preprocessing of, 441
Marginal ulcer, 820
Membrane. See Capillary membrane; Cell
types of, 446-448, 447f
Mark time reflex, 682-683
membrane; Respiratory membrane.
tissue distribution of, 440
Mass action, law of, 837
Membrane potential, 50, 50f, 54. See also
Lymphocytic leukemia, 437
Mass discharge, 757, 758
Action potential.
Lymphokines, 442, 447, 447f, 905
Mass reflex, 684
diffusion and, 57-58, 58f
Lysine, 853f
Mast cells, 436
neural, 57-58, 58f, 65. See also Action
Lysine monohydrochloride, in alkalosis, 398
activation of, 445-446
potential, neural.
Lysis
heparin production by, 436, 464
measurement of, 58-59, 59f
antigen, 444, 445, 445f
in asthma, 480, 529
physics of, 57-58, 58f
clot, 464
of lungs, 480
resting, 59-61, 60f
red blood cell, 453, 454, 455, 864
Mastectomy, lymphatic blockage after,
physics of, 57-58, 58f
Lysoferrin, 20
302-303
recording of, 70-71, 70f
Lysosomes, 13f, 16, 20, 20f
Mastication, 781-782
resting, 59-61, 60f
cortisol effects on, 953
Maximum expiratory flow, in respiratory
of gastrointestinal smooth muscle, 773
in hemorrhagic shock, 283
insufficiency, 525-526, 525f, 526f
of neuronal soma, 564-565, 564f
Lysozyme, 20
Mayer waves, 214, 214f
of sinus nodal fiber, 117, 117f
salivary, 794
Mean arterial pressure, 175-176, 176f
potassium diffusion in, 60, 60f
Mean circulatory filling pressure, 239-240,
sodium diffusion in, 60f, 61
239f
sodium-potassium pump in, 60f, 61
mean systemic filling pressure and, 240
smooth muscle, 96
M
Mean electrical axis, 137-140
slow wave rhythm of, 97, 97f, 772-773,
Machinery murmur, 275
bundle branch block effect on, 139-140,
772f
Macrocytes, 423
139f
Membrane proteins, 13-14, 14f
Macrophages, 431
from electrocardiogram, 137-138, 137f
carrier, 46f, 47, 49-50, 53-54, 53f
alveolar, 433, 481
left bundle branch block effect on, 139,
channel, 46f, 47-49, 47f, 48f
ameboid movement of, 24, 431
139f
Membrane-stabilizing factors, 70
antigen presentation by, 446, 446f
left heart shift effect on, 138
Memory, 557, 723-727
bactericidal agents of, 432
left ventricular hypertrophy and, 138, 138f
classification of, 723-724
enzymes of, 432
right bundle branch block effect on,
codification of, 725
in bone marrow, 434
139-140, 140f
consolidation of, 725-727
in inflammation, 434-436, 436f
right heart shift effect on, 138
declarative, 723-724
in liver, 433, 433f, 859, 861
right ventricular hypertrophy and,
failure of, 726
in lungs, 433
138-139, 139f
habituation and, 723, 724
in lymph nodes, 433, 433f
Mean pulmonary filling pressure, in
hippocampus in, 726-727
in lymphocyte activation, 442
unilateral left-sided cardiac failure, 262
in Aplysia, 724, 724f
in skin, 433
Mean QRS vector, 132, 132f
intermediate long-term, 723, 724-725, 724f
Index
1095
Memory (Continued)
Methane, 825
Motion sickness, 824
long-term, 723, 725, 726
Methionine, 853f
in space travel, 544
negative, 723
Methylmercaptan, 668
Motor aphasia, 721
positive, 723
Methylprednisone, 947, 947t
Motor apraxia, 687
rehearsal and, 726, 737
Metoprolol, 759
Motor cortex, 589-590, 685, 686f, 712
short-term, 723, 724
Micelles, 804, 812, 816
Betz cells of, 687-688
skill, 724
Michaelis-Menten equation, 884
body representation in, 685-686, 686f
synaptic facilitation and, 723, 724-725, 724f
Microcirculation, 181-182, 182f. See also
Broca’s area of, 687, 687f, 717, 717f,
synaptic inhibition and, 723, 724
Arteriole(s); Capillary (capillaries).
719
synaptic transmission in, 723, 724
Microfilaments, 13f
corticorubral tract from, 688-689, 689f
Wernicke’s area in, 718, 718f
Microgravity, 543-544
corticospinal (pyramidal) tract from,
working, 720, 723
Micropipette method
687-688, 688f
Memory cells, 443, 443f, 446
for capillary pressure, 186
eye movement area of, 687, 687f
Memory sensitization, 723
for interstitial pressure, 187
hand skill area of, 687, 687f
Memory traces, 723, 736
Microtubules, 11, 13f, 17, 17f
head rotation area of, 687, 687f
Menarche, 1021-1022, 1022f
Microvilli, intestinal, 813, 813f
incoming fiber pathways to, 688
Meningitis, 607
Micturition, 309, 311-314, 311f, 313f
lesions of, 691
Menopause, 1011, 1022, 1022f
abnormalities of, 313-314
muscle representation in, 685-686, 686f
Menorrhagia, 938
Micturition reflex, 311, 313-314
premotor area of, 686, 686f
Menstrual cycle, 1012-1015, 1013f
Micturition waves, 313, 313f
primary, 685, 686f
anovulatory, 1021, 1024-1025
Migraine headache, 607
removal of, 691
endometrial changes in, 1018-1019, 1018f
Milieu intérieur, 4, 9
signal transmission from, 687-688, 688f
follicular phase of, 1013-1014, 1021
Milk ejection (milk let-down), 1040-1041
signal transmission to, 688, 690
luteal phase of, 1014-1015
Milliosmole, 297
specialized areas of, 686-687, 687f
ovulation and, 1014, 1015f, 1021
Mineralocorticoids, 944. See also
supplementary motor area of, 686, 686f
postpartum suppression of, 1040
Aldosterone.
topographical representation of, 685-686,
regulation of, 1019-1021, 1020f, 1022f
functions of, 947-950, 948f
686f
Menstruation, 1019
synthesis of, 945-947, 946f, 947t
vertical columns of, 689-690
Merkel’s discs, 586, 586f
Minerals, metabolism of, 878-879, 878t
Motor end plate, 85, 86f
Mesencephalon, 558. See also Brain stem.
Minimal change nephropathy, 317, 409
Motor neurons, 559-560, 559f, 690, 690f
Metabolic rate, 884-885
Minimal change nephrotic syndrome,
alpha, 674, 674f, 675
after meals, 887
409
anterior, 674, 674f
basal, 886-887, 887f
Minute respiratory volume, 477
dendrites of, 568-569, 568f
cold effects on, 938-939
Miosis, 649
dynamic, 690
exercise effects on, 887, 887t
Mitochondrion (mitochondria), 12, 13f,
excitation of, 564-566, 566f
fever effects on, 887
16-17, 16f
gamma, 674, 674f, 675, 676, 678
growth hormone effects on, 887
ATP formation in, 17, 22-24, 22f, 23f,
inhibition of, 566-568, 567f
malnutrition effects on, 887
835-836, 835f
pyramidal, 690
sleep effects on, 887
chemiosmotic mechanism of, 835, 835f,
rate of firing of, 569, 569f
testosterone effects on, 1006
843
resting membrane potential of, 564-565,
thyroid hormone effects on, 886, 936,
DNA of, 17
564f
936f
fatty acid metabolism in, 842-843, 843f
static, 690
blood flow and, 196, 196f
function of, 22-24, 22f, 23f
stimulation of, 690-691, 690f
epinephrine effect on, 755
matrix of, 16f, 17
vertical columns of, 689-690
estrogen and, 1018
citric acid cycle of, 833-835, 834f
Motor unit, 674
in Inuit, 896
of muscle, 74
Mountain sickness
measurement of, 885
oxidative phosphorylation in, 835-836,
acute, 540-541
neonatal, 1048-1049
835f
chronic, 541
protein effect on, 887
structure of, 16f, 17
Mouth, dryness of, thirst and, 361, 361t
Metabolic syndrome, 974
Mitosis, 17, 37-39, 37f
Movement(s)
Metabolic theory, of blood flow regulation,
colchicine prevention of, 40
ameboid, 24, 24f, 431
199
Mitotic apparatus, 37f, 38-39
amygdala in, 737
Metabolism. See also Adenosine
Mitotic spindle, 17, 39
ballistic, 705
triphosphate (ATP); Energy.
Mitral cells, of olfactory bulb, 667f, 669, 670
cellular, 24-25, 24f, 25f
brain, 767
Mitral regurgitation
ciliary, 24-25, 25f
carbohydrate, 829-839. See also
circulatory dynamics in, 273
control of. See also Muscle (skeletal),
Carbohydrates, metabolism of.
heart sounds in, 271f, 272
contraction of.
coupled reactions in, 829
Mitral valve, 109, 109f
basal ganglia in. See Basal ganglia.
during pregnancy, 1034-1035
stenosis of
brain stem in, 678, 691-692, 691f, 692f,
enzymes in, 884, 884f
circulatory dynamics in, 273
697
in leukemia, 437
heart sounds in, 271f, 272
cerebellum in. See Cerebellum.
lipid, 840-851. See also Lipid(s),
pulmonary capillary pressure in, 489
cognitive, 709, 709f
metabolism of.
pulmonary edema in, 488-489
intermediate cerebellar cortex in,
protein, 852-856. See also Protein(s),
Modiolus, 653, 655f
704-705, 704f
metabolism of.
Monoamine oxidase inhibitors, 745
motor cortex in. See Motor cortex.
rate of, 884-885, 884f
Monocyte(s), 421f, 429-430, 430f
posterior parietal cortex in, 710,
basal, 886-887, 887f, 887t. See also
concentration of, 430
710f
Metabolic rate, basal.
diapedesis of, 431
prefrontal association area in, 716-
Metaphase, 37f, 39
in inflammation, 435
717
Metarhodopsin, 629, 629f
life span of, 431
spinal cord in. See Spinal cord, motor
Metarterioles, 181, 182f
Monocyte colony-stimulating factor, 435,
system of; Spinal reflexes.
intermittent contraction of (vasomotion),
436f
spinocerebellum in, 704-705, 704f
182-183, 187, 197-198, 197f
Monocyte-macrophage cell system
summary of, 712-713
Metastatic calcification, 990
(reticuloendothelial system), 180,
vestibular apparatus in, 692-696,
Methacholine, 759
432-434, 433f, 434f
693f-695f
neuromuscular junction effects of, 88-89
Motilin, 776
vestibulocerebellum in, 703-704
1096
Index
Movement(s) (Continued)
Muscle (skeletal) (Continued)
Muscle (smooth), 92-99
disorders of
in slow fibers, 81
actin filaments of, 93-94, 93f
basal ganglia lesions and, 709, 710, 710f,
isometric, 80, 80f
action potential of, 96-98, 97f, 772-773
711-712
isotonic, 80, 80f
calmodulin of, 95
brain stem section and, 692
load and, 78, 79f
contact junctions of, 96
cerebellar lesions and, 706-707
maximum strength of, 81
contraction of, 93-99
motor cortex lesions and, 691
mechanics of, 81-82, 81f
action potential in, 96-98, 97f
spinal cord transection and, 684
molecular mechanism of, 74-78, 75f-77f
calcium in, 95, 99, 99f
finger, 678
motor unit in, 81
calmodulin in, 95
involuntary, 737
multiple fiber summation in, 81
cessation of, 95
ocular, 645-648, 646f, 647f
muscle length and, 78, 78f
chemical factors in, 98
premotor area in, 687, 687f, 696
muscle spindles and, 675-679, 676f-678f
duration of, 94
pendular nature of, 705
myosin filament in, 75-78, 75f-78f
energy for, 94
saccadic, 646-647
oxidative metabolism in, 79
force of, 94
sequential, 706
phosphocreatine in, 79
hormonal factors in, 98
timing of, 706, 709-710
ratchet theory of, 76-77, 77f
in blood flow autoregulation, 199
Mucin, 793
recovery of, 83
inhibition of, 98
Mucous glands, 791, 795. See also Mucus.
sliding filament mechanism of, 74-75, 75f
latch mechanism of, 94, 95
Mucous neck cells, 795, 796f, 797
slow fibers in, 81
myosin cross-bridges in, 94
Mucus
somatosensory feedback in, 690
myosin kinase in, 95
duodenal, 805
staircase effect (treppe) in, 81-82
myosin phosphatase in, 95
esophageal, 795
strength of, 81-82, 81f
sarcoplasmic reticulum in, 99, 99f
gastric, 797, 799
tension with, 77-78, 78f
stress-relaxation of, 94-95
gastrointestinal, 791, 793
tetanic, 246
tissue factors in, 98
large intestine, 806
tetanization of, 81, 81f
without action potentials, 97-98
pyloric, 797
tropomyosin in, 76
cramps of, 604
respiratory tract, 480
troponin in, 76
dense bodies of, 93, 93f
small intestine, 805
velocity of, 78, 79f
diffuse junction of, 96, 96f
Muramyl peptide, 740-741
voluntary, 678
hormone receptors of, 98
Murmurs, 271f, 272
walk-along theory of, 76-77, 77f
hyperpolarization of, 98
in patent ductus arteriosus, 275
work output during, 78-79
innervation of, 95-96, 96f
Muscle. See Cardiac muscle; Muscle
contracture of, 83
junctional potential of, 97-98
(skeletal); Muscle (smooth).
deconditioning of, in space travel, 544
membrane potentials in, 96
Muscle (skeletal), 72-83. See also Exercise,
denervation of, 83
multi-unit, 92, 93f, 97-98
muscles in.
energy metabolism in, 883, 1056-1059,
myosin filaments of, 93-94, 93f
action potential of, 89-91, 90f, 91f
1057f, 1058t
neuromuscular junction of, 95-96, 96f
agonist, 82
fast, 80-81
sarcoplasmic reticulum of, 99, 99f
anatomy of, 72-74, 73f, 74f
fatigue of, 82
stress-relaxation of, 94-95, 172, 173f,
antagonist, 82
glucose storage in, 963-964
281
antigravity, 678, 691f, 692
glucose transport in, 964, 964f
stretch of, 97
atrophy of, 83
glucose uptake by, 963
structure of, 93-94, 93f
blood flow in, 195, 196t
glycogen storage in, 831-832
syncytial, 92-93, 93f
during exercise, 246, 247-248, 247f,
glycolysis in, 883
types of, 92-93, 93f
1062-1063, 1063f
Golgi tendon organ of, 675, 679-680, 679f
unitary (single-unit), 92-93, 93f, 96-97,
nervous control of, 247
hyperplasia of, 83
97f
oxygen effect on, 247
hypertrophy of, 83
visceral, 92-93, 93f
oxygen-hemoglobin dissociation curve
in exercise, 1055-1061. See also Exercise,
Muscle impulse, 65-66, 65f. See also Action
of, 508, 508f
muscles in.
potential.
capillaries of, 184, 184t, 246
in poliomyelitis, 83
Muscle spindles, 595, 674, 674f, 675-679
clonus of, 679
innervation of, 556, 557f
continuous discharge of, 676
contraction of, 74-83. See also
length of, 78, 78f, 83
dynamic response of, 676
Movement(s).
lever system of, 82, 82f
gamma motor nerves to, 676
actin filament in, 75f-78f, 76-78
muscle spindles of, 674, 674f, 675-679,
in muscle stretch reflex, 676-677, 677f
adenosine triphosphatase in, 76
676f, 677f. See also Muscle spindles.
in position stabilization, 678
adenosine triphosphate in, 77, 79-80
myofibrils of, 72-74, 73f, 74f, 75-77, 76f,
in voluntary motor activity, 678
atrophy and, 83
77f, 90f
intrafusal fibers of, 674, 674f, 675, 676,
blood flow and, 246, 247f
oxygen consumption by, 247
676f
calcium in, 74, 76, 90-91, 91f
potassium release from, 366
nuclear bag fibers of, 676, 676f
cardiac output and, 213-214
reciprocal inhibition of, 681-682, 682f
nuclear chain fibers of, 676, 676f
coactivation in, 82
red, 81
receptor function of, 675-676
efficiency of, 79-80
remodeling of, 82-83
sensory innervation of, 674f, 675-676,
energetics of, 78-80
sarcolemma of, 72, 90f
676f
energy sources for, 79-80
sarcoplasm of, 74, 74f
signal averaging function of, 677, 677f
equalization of, 680
sarcoplasmic reticulum of, 74, 74f, 89-91,
static response of, 676
excitation of, 690
90f
Muscular dystrophy, swallowing in, 819
fast fibers in, 80-81
slow, 81
Muscularis mucosae, 787-788
fatigue of, 82
spasm of, 683, 691
Musculoskeletal system, 5. See also Bone(s);
force of, 78, 78f
headache and, 607
Muscle (skeletal).
force summation in, 81, 81f
pain and, 599
Mutation, 38, 41
frequency summation in, 81, 81f
stretch reflexes of, 676-678, 677f
Myasthenia gravis, 89, 448, 819
general mechanism of, 74
testosterone effects on, 1005
Mydriasis, 649
glycogen in, 79
thyroid hormone effects on, 937
Myelin sheath, 68, 68f
Golgi tendon organ and, 679-680, 679f
tone of, 82
degeneration of, 876
hyperplasia and, 83
tonic disorders of, 691
Myelogenous leukemia, 437
hypertrophy and, 83
transverse tubules of, 89-90, 90f
Myenteric (Auerbach’s) plexus, 774-775,
in fast fibers, 80-81
white, 81
774f, 777
Index
1097
Myocardial infarction, 253-255, 253f. See
Neonate (Continued)
Nervous system (Continued)
also Cardiac failure.
iron for, 1049
enteric. See Enteric nervous system.
adenosine loss with, 252
jaundice in, 1048
integrative function of, 556-557
anterior wall, 143-144, 143f, 145, 145f
liver function in, 1048
motor axis of, 556, 557f. See also
cardiac output after, 255, 259f, 260-262,
metabolic rate in, 1048-1049
Movement(s), control of; Muscle
261f
nutrition for, 1047, 1049
(skeletal), contraction of.
cardiac output in, 236f, 237
of diabetic mother, 1050
somatosensory axis of, 555, 556f. See also
conduction pathways after, 254
premature, 1050-1051
Sensory receptors; Somatic sensation.
congestive symptoms after, 254
red blood cell count in, 1047-1048,
Net filtration pressure, 185-186, 185f,
electrocardiogram after, 140, 140f, 145,
1048f
189
145f
renal function in, 1048
Net reabsorption pressure, 189
electrocardiogram in, 143-145, 143f, 144f
respiration in, 1047
Neuroendocrine system, 905. See also
injury current in, 141-145, 142f-145f, 254
thyroid disorders in, 1050
Adrenal medulla; Hypothalamus;
posterior wall, 144-145, 144f, 145f
vitamin C deficiency in, 1049
Pituitary gland.
recovery from, 254-255, 255f, 260
vitamin D in, 1049
Neurogenic dyspnea, 532
rupture after, 254
Neospinothalamic tract, 600, 600f
Neurogenic hypertension, 228
shock in, 262-263
Neostigmine, 89, 759
Neurogenic shock, 285, 287
subendocardial, 253
Nephritis, 409
Neurohypophysis. See Pituitary gland,
ventricular fibrillation after, 254
Nephrogenic diabetes insipidus, 302,
posterior.
Myocardial ischemia, 252-255, 253f. See also
357-358, 414
Neuromuscular junction. See also
Myocardial infarction.
Nephrons. See also Renal tubules.
Synapse(s).
pain in, 255-256
anatomy of, 310, 310f
skeletal muscle, 85-88
recovery from, 254-255, 255f
cortical, 310, 311f
acetylcholine secretion at, 85-88,
T wave in, 145-146, 146f
in chronic renal failure, 409-412, 410f,
86f-88f
Myofibrils, 72-74, 73f, 74f, 75-77, 76f, 77f,
411f, 411t
anatomy of, 85, 86f
90f
intercalated cells of, 336-337, 336f
drug effects at, 88-89
Myogenic theory, of blood flow regulation,
juxtamedullary, 310, 311f
fatigue of, 88
199
principal cells of, 336-337, 336f
safety factor for, 88
Myoglobin, 81
Nephrosclerosis, 408, 408f
smooth muscle, 95-96, 96f
Myopia, 619-620, 619f
Nephrotic syndrome, 303, 381, 409
acetylcholine secretion at, 96
Myosin, 72-74, 73f, 74f
Nernst equation, 50, 58
norepinephrine secretion at, 96
in pinocytosis, 19, 19f
Nernst potential, 50, 50f, 58, 565
Neuron(s), 555, 556f. See also Action
Myosin filaments
Nerve(s). See Nerve fibers; Neuromuscular
potential, neural; Synapse(s).
of cardiac muscle, 103
junction; Neuron(s); Synapse(s).
chloride channels of, 566, 566f
of skeletal muscle, 72-73, 73f, 75-78, 75f
Nerve fibers. See also Neuron(s);
chloride ion concentration of, 564f,
of smooth muscle, 93-94, 93f
Synapse(s).
565
Myosin kinase, 95, 915
action potential of, 61-65, 61f-64f. See
concentration gradients of, 564f, 565
Myosin phosphatase, 95
also Action potential, neural.
continuous discharge from, 581, 582
Myxedema, 942, 942f
anesthetic effects on, 70
electrical potential of, 564f, 565
classification of, 576-577, 577f
excitation of, 565-566, 566f
conduction velocity in, 69
excitatory postsynaptic potential of,
convergence of, 580, 580f
565-566, 566f, 567, 567f, 568
N
diameter of, 576-577, 577f
excitatory state of, 569, 569f
Naming, 716, 716f
discharge zone of, 579, 579f
excitatory stimulus of, 579, 579f
Narcosis, nitrogen, 545-546
divergence of, 580, 580f
facilitation of, 568, 579, 579f
Natriuresis, pressure, 216, 323, 341-342,
energy metabolism of, 66, 66f
fusiform, 714, 715f
374-376, 375f
facilitated zone of, 579, 579f
gigantocellular, 730f, 731
aldosterone escape and, 948-949, 948f
from sensory receptors, 576-578, 577f,
granular, 714, 715f
angiotensin II in, 378, 378f
578f
heat-sensitive, 894
in hypertension, 228-229
labeled line principle of, 572-573
hyperpolarization of, 566, 566f
Nausea, 824
membrane potential of, 57-58, 58f, 65
inhibition of, 566, 566f, 567
antidiuretic hormone secretion and,
measurement of, 58-59, 59f
inhibitory, 580, 581f
360-361
physics of, 57-58, 58f
inhibitory postsynaptic potential of, 566,
Nearsightedness, 619-620, 619f
resting, 59-61, 60f
566f, 567, 568
Neck
myelinated, 68, 68f, 577f
inhibitory state of, 569
proprioceptors of, 696
resting membrane potential of, 59-61, 60f
intracellular fluid of, 565
veins of, 179
saltatory conduction of, 68-69, 69f
intrinsic excitability of, 582
Necrosis, 40
spatial summation in, 578, 578f
motor, 559-560, 559f. See also Motor
hepatic, 283, 283f
specificity of, 572-573
neurons.
renal, 406
stimulatory field of, 579, 579f
NPY-AGRP, 868-870, 869f
tissue, 283, 283f
structure of, 68, 68f
of motor cortex, 689-690
Negative feedback, 7, 8f
temporal summation in, 578, 578f
of red nucleus, 690
Neonate. See also Fetus.
type A, 576, 577, 577f
of somatosensory cortex, 590-591, 591f
acid-base balance in, 1048
type C, 576, 577, 577f
of vasomotor center, 206
allergy in, 1049
unmyelinated, 68, 68f, 577f
parasympathetic, 750
arterial pressure in, 1047
varicosities of, 96, 96f
pools of, 578-583, 579f-583f. See also
blood volume in, 1047
Nerve gas, 89
Neuronal pools.
body temperature in, 1048-1049, 1049f
Nerve impulse, 65-66, 65f. See also Action
postganglionic, 748-749, 749f, 750,
breathing onset in, 1044-1045, 1045f
potential, neural.
759-760
calcium in, 1049
Nervous system, 5-6
postsynaptic, 557, 559
cardiac output in, 1047
action potential of. See Action potential,
excitation of, 562
circulation in, 1045-1048, 1046f
neural.
inhibition of, 562
endocrine problems in, 1049-1050
autonomic. See Autonomic nervous
ion channels of, 561, 561f, 910-911
fluid balance in, 1048
system.
receptor proteins of, 560-562, 561f
growth of, 1051f, 1052
central. See Central nervous system.
second messenger system of, 561-562,
immunity in, 1049
computer analogy to, 558, 558f
561f
1098
Index
Neuron(s) (Continued)
Nicotine, 759
Obesity (Continued)
potassium channels of, 566, 566f
neuromuscular junction effects of, 88-89
hypertrophic, 872
potassium ion concentration of, 564f,
Nicotinic acid, 876
leptin and, 871
565
Nicotinic drugs, 759-760
renal failure in, 407-408
preganglionic, 748-749, 749f, 750, 760
Night blindness, 629, 875
sympathetic nervous system in, 887-888
presynaptic, 559, 560
Nitrates, 256
treatment of, 873-874
presynaptic inhibition of, 567
Nitric oxide, 564
Obligatory urine volume, 350, 356-357
pro-opiomelanocortin, 868-870, 869f
blood flow and, 199-200, 322, 322t
Oblique muscles, of eye, 645, 645f
pyramidal, 714, 715f
Nitrogen
Obstructive sleep apnea, 523
rate of firing of, 569, 569f
diffusion coefficient of, 493
Occipital cortex, in eye movement, 645-646,
resting membrane potential of, 564-565,
excretion of, 866
646f
564f
lipid solubility of, 47
Oddi, sphincter of, 803, 803f
reverberatory circuit of, 581-582,
partial pressure of, 492, 493t
Odontoblasts, 992
581f-583f
pressures of, during deep-sea diving,
Odor blindness, 668. See also Olfactory cells;
rhythmical signal output from, 582-583,
545-546, 548-549, 548f
Smell sensation.
583f
solubility coefficient of, 492
Ohm’s law, 164, 234
serotonergic, 729, 730, 730f
Nitroglycerin, 256
Oleic acid, 840. See also Triglycerides.
sodium channels of, 566, 566f
Nociceptive reflex, 680-681, 680f
Olfactory area
sodium ion concentration of, 564f, 565
Nociceptors, 572, 573t. See also Pain.
lateral, 669
spatial summation in, 567-568, 567f
Nontropical sprue, 822
medial, 669
subthreshold, 579, 579f
Norepinephrine, 563, 907t
Olfactory bulb, 667f, 669-670
sympathetic, 748-749, 749f
basal secretion of, 756
glomeruli of, 669
temporal summation in, 568
blood flow and, 201, 247, 251, 322, 322t
Olfactory cells, 667-669. See also Smell
Neuronal pools, 579-580
bronchial effects of, 479
sensation.
afterdischarge of, 581-583, 581f, 582f
coronary blood flow and, 251
action potential of, 668
continuous discharge from, 582, 582f
in brain, 730-731, 730f
adaptation of, 668
excitation of, 579, 579f
in bronchiolar function, 479
excitation of, 667-668
inhibition of, 579
in cardiac function, 122
glomerular termination of, 667f, 669
inhibitory circuits of, 583
in depression, 745
Olfactory cilia, 667, 667f
organization of, 579, 579f
in exercise, 247, 846
Olfactory membrane, 667, 667f
reciprocal inhibition circuit of, 580, 581f
in gastrointestinal function, 775
Olfactory nerve, 667-668, 669
rhythmical discharge from, 582-583, 583f
in renal blood flow, 322, 322t
Olfactory tract, 667f, 669-670
signal convergence in, 580, 580f
in skeletal muscle blood flow, 247
Oligohydramnios, 1043
signal divergence in, 580, 580f
in vasoconstriction, 207
Oligomenorrhea, 938
stimulatory field of, 579, 579f
nerve secretion of, 96
Oliguria, 405
Neuropeptide Y (NPY), 868-870, 869t
receptors for, 96
Olivary nucleus, 657, 658f, 660, 688
Neuropeptide Y (NPY)-agouti-related
secretion of, 207-208, 750, 755, 756
Olivocerebellar fibers, 688
protein (AGRP) neuron, 868-870,
synthesis of, 751
Olivocerebellar tract, 700, 700f
869f
tubular secretion of, 334
Oncogenes, 41
Neuropeptides, 563t, 564
Nose
Oncotic pressure. See Colloid osmotic
Neurotransmitters, 559, 562-564, 562t, 563t,
filtration function of, 479f, 480-481
pressure.
905. See also specific neurotransmitters.
in vocalization, 481
Oocyte maturation-inhibiting factor,
excitatory, 561
Nuclear membrane (nuclear envelope), 12f,
1013
in feeding, 868-870, 869f, 869t
13f, 17-18, 18f
Oocytes, 1011
in obesity, 873
Nuclear pores, 18, 18f
Operon, 35-36, 35f
in retina, 635
Nucleic acid, of virus, 18
Ophthalmoscopy, 622-623, 622f
inhibitory, 561
Nucleolus, 12f, 13f, 18, 18f, 33
Opiates, endogenous, 602-603, 602f
neurohormonal, 730-731, 730f
Nucleoplasm, 12f
Opsonization, 19, 432, 445, 445f
neuropeptide, 563t, 564
Nucleotides. See Deoxyribonucleic acid
Optic chiasm, 640, 641f
of basal ganglial system, 710-711, 710f
(DNA); Ribonucleic acid (RNA).
lesions of, 645
of enteric nervous system, 775
Nucleus, 12f, 17, 18f. See also Cell(s);
Optic disc, 644
receptor binding of, 560-562, 561f
Cytoplasm.
edema of, 766
release of, 560
genes in, 27-30, 28f-30f
Optic nerve, 636-638, 640, 641f
sleep and, 740-741
Nucleus accumbens, 711
lesions of, 645
small-molecule, 562-564, 562t
Nucleus ambiguus, 515
Optics
vesicles of, 560, 560f
Nucleus retroambiguus, 515
ocular, 617-622, 617f-621f. See also
Neutralization, antigen, 444, 445
Nutrient lack theory, 197-198, 197f
Eye(s); Vision.
Neutrophil(s), 421f, 429-430, 430f
Nutrients. See also Carbohydrates; Fat(s);
physical principles of, 613-617, 614f-616f
ameboid movement of, 431
Food; Protein(s).
Optokinetic movements, 646-647
bactericidal agents of, 432
absorption of, 5
Oral contraception, 1024
concentration of, 430
diffusion of, 183-183, 183f
Orbital frontal cortex, ablation of, 738
diapedesis of, 430f, 431
Nutrition. See also Digestion; Feeding;
Orexigenic substances, 868, 869t
enzymes of, 432
Food.
Organ of Corti, 652, 652f, 653f, 655-656, 655f
in inflammation, 435-436, 436f
during pregnancy, 1034-1035
Organ transplantation, 455-456
margination of, 430f, 435
neonatal, 1047, 1049
rejection prevention in, 456
phagocytosis by, 432
Nystagmus, 707
tissue typing for, 455-456
Neutrophilia, 435
Organelles, 13f, 14-17, 15f, 16f
Newborn. See also Neonate.
Organomegaly, 1050
hemolytic disease of, 421f, 427, 454
Organum vasculosum, 929
respiratory distress syndrome of, 474, 529,
O
Orgasm
1045
O-A-B blood types, 451-453, 452f, 452t
female, 1023-1024
Niacin, 875t, 876
Oat bran, 851
male, 1002-1003
deficiency of, 198, 876
Obesity, 846, 872-874
Orlistat, 873
Nicotinamide adenine dinucleotide, 834
cortisol and, 952
Osmolality, 52, 297
Nicotinamide adenine dinucleotide
diabetes mellitus and, 974, 975
interstitial, 294t
phosphate, 837
hyperplastic, 872
intracellular, 294t
Index
1099
Osmolar clearance, 357
Ovum (Continued)
Oxygen (Continued)
Osmolarity, 52, 297-298
fertilization of, 1000-1001, 1027-1029,
from alveoli to pulmonary capillaries,
body fluid, 294t, 298
1028f
502-503, 503f
calculation of, 297-298
Oxalate, renal tubule secretion of, 334
from peripheral capillaries to tissue
cerebrospinal fluid, 361
Oxaloacetic acid, 833-834, 834f
cells, 504
extracellular fluid. See also Urine,
Oxidative phosphorylation, 835-836, 835f
from peripheral capillaries to tissue
osmolarity of.
Oxygen
fluid, 503-504, 504f
potassium distribution and, 366-367
concentration of, 485, 494-495, 495f,
hemoglobin in, 505-508, 506f, 508f. See
regulation of, 358-363
496f
also Hemoglobin; Oxygen-
aldosterone in, 362-363, 363f
partial pressure of, 500, 500f
hemoglobin dissociation curve.
angiotensin II in, 362-363
altitude effects on, 537-538, 538f, 538t
in arterial blood, 503, 503f
antidiuretic hormone in, 358-361,
dissolved oxygen and, 546, 546f
in dissolved state, 509
359f, 360f, 360t, 362, 362f
free radical production and, 547
respiratory exchange ratio in, 512
thirst in, 361-363, 361t, 362f, 363f
in unpressurized airplane, 538
utilization coefficient and, 507
interstitial fluid, 294t, 298
pure-oxygen breathing and, 538, 538f
uptake of, during exercise, 503, 503f
plasma, 294t, 298
tissue Po2 and, 546-547, 546f
utilization coefficient of, 507, 508
antidiuretic hormone secretion and,
pulmonary blood flow and, 485
utilization of, 508-509, 509f
348-349, 358-360, 358f, 360t
blood, in respiratory insufficiency, 525
adenosine diphosphate and, 508-509,
estimation of, 358
brain requirement for, 767
509f
regulation of, 358-363, 358f
chemoreceptor response to, 518-520, 518f,
blood flow and, 509
Osmole, 52, 297
519f
diffusion distance and, 509
Osmoreceptors, 358-361, 358f, 359f, 929. See
consumption of
inadequate, 530-531, 531f
also Antidiuretic hormone (ADH).
blood flow rate and, 504, 504f
intracellular Po2 and, 508-509, 509f
thirst integration with, 362-363, 362f
cardiac, 250-251, 253
tissue Po2 and, 503-504, 504f
Osmosis, 51-52, 51f, 296-299, 298f
exercise and, 233, 233f, 247, 506-507,
Oxygen debt, 883, 1058-1059, 1059f
of water, 332, 814
506f, 520, 520f, 1061-1062, 1061f,
Oxygen diffusing capacity, 498, 499f
rate of, 297
1062f
exercise and, 498, 1062
Osmotic coefficient, 297-298
renal, 320, 321f
high altitude and, 539
Osmotic diuretics, 402-403, 403t
skeletal muscle, 247
Oxygen Fick principle, for cardiac output,
Osmotic pressure, 51-52, 52f, 297-298. See
deficiency of
244, 244f
also Colloid osmotic pressure.
blood flow and, 196-198, 197f
Oxygen free radicals, in deep-sea diver, 547
calculation of, 297-298
cerebral blood flow and, 761-762
Oxygen poisoning, 547
interstitial, 294t, 298
diffusing capacity of, 498, 499f
Oxygen therapy, 530-531, 531f
intracellular, 294t
exercise and, 498, 1062
hyperbaric, 550-551
renal, 309, 309f, 310t
high altitude and, 539
in carbon monoxide poisoning, 510
diuretic effects on, 402-403, 403t
diffusion coefficient of, 493
in premature infant, 1051
Ossicles, 651-652, 652f
diffusion of. See Oxygen, transport of.
in shock, 287
ankylosis of, 661
energy equivalent of, 885
Oxygen-hemoglobin dissociation curve, 507,
Osteitis fibrosa cystica, 990
extracellular, 4, 6, 7t
507f
Osteoblasts, 982, 982f
gastrointestinal concentration of, 779
at high altitude, 507, 540, 540f
growth hormone effects on, 923
hemoglobin combination with, 420,
during exercise, 508, 508f
Osteoclasts, 982, 982f
424-425, 505-510. See also Oxygen-
shifts in, 507-508, 508f
activation of, 987
hemoglobin dissociation curve.
Oxyntic glands, 795-797, 796f
parathyroid hormone effect on, 987
altitude and, 538, 538f
Oxytocin, 907t, 918, 919f, 929
Osteocytic membrane system, 987
carbon dioxide displacement and,
in lactation, 1040-1041
Osteolysis, 986-987
511-512, 512f
secretion of, 733f, 734
Osteomalacia, 412, 991
exercise and, 507-508
structure of, 928
Osteon, 982, 983f
Haldane effect and, 511-512, 512f
uterine effects of, 1036
Osteoporosis, 992, 1017
reversibility of, 505-507, 507f
Osteoprogenitor cells, 983
saturation of, 507, 507f
Otosclerosis, 661, 661f
in space travel, 543
Ovarian cycle, 1012-1015, 1013f
iron combination with, 424-425, 424f
P
anovulatory, 1021, 1024-1025
lipid solubility of, 47
P wave, 107, 107f, 123, 124f
endometrial changes in, 1018-1019, 1018f
partial pressure of, 492, 493t
vectorial analysis of, 136, 136f
follicular phase of, 1013-1014, 1021
alveolar, 494-495, 495f, 500, 500f
Pacemaker. See also Sinus (sinoatrial) node.
luteal phase of, 1014-1015
arterial, 518, 518f
artificial, 150, 156
ovulation and, 1014, 1015f, 1021
chemoreceptor stimulation by, 518, 518f
ectopic, 120-121
postpartum suppression of, 1040
extracellular, 46f
Pacemaker waves, 97, 97f, 772-773
regulation of, 1019-1021, 1020f, 1022f
hemoglobin and, 507
Pacinian corpuscles, 573f, 587
Ovary (ovaries), 906f, 907t, 1011, 1012f. See
in cerebral tissue, 761-762
accommodation of, 576
also Reproduction, female.
interstitial, 503-504, 504f
adaptation of, 575, 575f
abnormalities of, 1023
intracellular, 46f, 504, 508-509, 509f
receptor potential of, 574-575, 574f, 575f
androgen secretion by, 1003
low, acclimatization to, 539-540
vibratory signals from, 693-694
follicular phase of, 1013-1014, 1013f
respiration and, 518-520, 518f, 519f
Paget’s disease, 989
luteal phase of, 1014-1015
tissue, 503-504, 504f, 506f, 507
PAH. See Para-aminohippuric acid (PAH).
monthly cycle of, 1012-1015, 1013f. See
alveolar Po2 and, 546-547, 546f
Pain, 598-606
also Ovarian cycle.
venous admixture and, 503, 503f
appendiceal, 605, 605f
Overhydration
placental, 1030-1031, 1031f
brain stem areas of, 601
hyperosmotic, 301t, 302
solubility coefficient of, 492
cerebral cortex in, 601
hypo-osmotic, 301-302, 301t
blood flow and, 196-198, 196f, 197f,
chemical stimuli in, 599
Overnutrition, 873. See also Obesity.
200
electrical suppression of, 603
Ovulation, 1014, 1015f, 1021
Bohr effect and, 508, 508f
elicitation of, 599
temperature and, 1025, 1025f
disorders of, 530
fast, 598, 600, 600f, 601
Ovum, 1011, 1013, 1013f
transport of, 183, 184, 502-510
flexor reflex in, 680-681, 680f, 681f
fallopian tube transport of, 1028-1029,
Bohr effect and, 508, 508f
glutamate in, 601
1028f
during exercise, 506-507, 508, 509
headache, 606-607, 606f
1100
Index
Pain (Continued)
Parasympathetic nervous system
Penis, 754t, 1002, 1002f. See also Sexual
hypersensitivity to, 603f, 605
(Continued)
function, male.
in coronary heart disease, 255-256
coronary blood flow and, 251
Pentagastrin, 799
in decompression sickness, 549
denervation effects on, 756-757, 756f
Pentolinium, 760
in gastrointestinal spasm, 604
functions of, 750-757, 754t
Pentose phosphate pathway, 837-838, 838f
in ischemia, 604
in bronchiolar function, 479
Pepsin, 797, 810, 810f
in muscle spasm, 599
in cardiac function, 113, 113f, 121,
Pepsinogen, 797, 798
in shingles, 605
205-206, 205f, 234-235, 754, 754t, 755
Peptic (chief) cells, 795-796, 797
in tic douloureux, 605-606
in gastrointestinal blood flow, 779
Peptic ulcers, 820-821, 820f
intracranial, 606-607, 606f
in gastrointestinal function, 753-754, 775,
Peptidases, 806, 810-811, 810f
ischemic stimuli in, 599
792
Peptide chain, 852. See also Amino acids;
labor, 1038
in large intestine mucus secretion, 806
Protein(s).
localization of, 601
in ocular accommodation, 618, 648-649,
Peptide linkages, 34-35, 808, 810, 810f, 852,
neospinothalamic tract for, 600, 600f
648f, 753, 754t
881
paleospinothalamic tract for, 600-601,
in respiratory function, 479-480
Peptide YY, in feeding, 870
600f
in salivary secretion, 794-795
Perforins, 447
parietal, 604-605, 605f
localized responses of, 757-578
Pericardial effusion, 140
receptors for, 598-599
of eye, 648, 648f
Pericardial fluid, 292
referred, 603-605, 603f, 605f
postganglionic neurons of, 750
Pericardium, pain in, 604
reticular formation in, 601
preganglionic neurons of, 750
Perilymph, 656
slow, 598, 600, 600f, 601
supersensitivity of, 756-757, 756f
Perimetry, 644-645, 644f
spasm in, 599
tone of, 756-757, 756f
Periodic breathing, 522, 522f
substance P in, 601
Parasympathomimetic drugs, 759
Periodontal membrane, 992-993
suppression of, 602-603, 602f
Parathyroid glands, 906f, 907t, 985-986,
Peripheral neuropathy, in diabetes mellitus
surgical interruption of, 602
986f
type I, 973
thalamus in, 601
Parathyroid hormone, 907t, 985-988
Peripheral resistance unit (PRU), 167
thermal stimuli in, 599, 608, 608f
bone effects of, 986-987
Peripheral vascular resistance, 167. See also
tissue damage in, 599
calcium regulation of, 988, 988f
Vascular resistance.
transmission of, 600-602, 600f
cAMP effects on, 987-988
arterial pressure and, 219, 219f, 220,
ureteral, 312
deficiency of, 990
220f
visceral, 603-605, 605f
disorders of, 990-992
cardiac output and, 234, 234f, 236-237,
Pain reflex, 680-681, 680f, 681f
excess of, 990-991
236f
Paleocortex, 669
extracellular calcium and, 986-988, 986f
Peripheral venous pressure, 176-177, 177f
Paleospinothalamic tract, 600-601, 600f
extracellular phosphate and, 986-988, 986f
Perisinusoidal spaces, 859
Palmitic acid, 840. See also Triglycerides.
in calcium homeostasis, 342t, 343,
Peristalsis
Pancreas, 791, 792f, 906f, 907t
371-372, 815, 989-990
colonic, 789
alpha cells of, 832
in phosphate homeostasis, 373
esophageal, 783
bicarbonate secretion by, 800, 800f, 801
in pregnancy, 1034
gastric, 784, 785-786
cells of, 961, 962f
synthesis of, 986
gastrointestinal, 776-777, 777f
enzymes of, 799-800, 801, 809, 809f
thyroid hormone effects on, 937
in vomiting, 824
glucagon secretion by, 832, 961, 970-971
Parathyroid poisoning, 990
intestinal, 787-788
insulin secretion by, 961-970. See also
Paraventricular nuclei, antidiuretic hormone
Peristaltic rush, 788
Insulin.
synthesis in, 359, 359f
Peritoneal fluid, 292
secretions of, 799-802, 800f, 802f
Paraympathectomy, 756-757
Peritoneointestinal reflex, 790
cephalic phase of, 801
Parietal (oxyntic) cells, 54, 796-797, 796f
Peritoneum, pain in, 604-605, 605f
gastric phase of, 801
hydrochloric acid secretion by, 54,
Peritonitis, muscle spasm in, 683
in protein digestion, 810, 810f
796-797, 796f, 810, 820
Peritubular capillaries, 309-310, 309f
intestinal phase of, 801
intrinsic factor secretion by, 797, 820
colloid osmotic pressure of, 309, 309f,
phases of, 801, 802f
structure of, 796, 796f
310t, 340, 340f
stimuli of, 800-801
Parietal cortex, in motor control, 710, 710f
filtration coefficient of, 340, 341t
somatostatin secretion by, 971
Parietal lobe
hydrostatic pressure of, 309, 309f, 310t,
Pancreatic amylase, 799, 809, 809f
angular gyrus of, 718
340, 340f
Pancreatic lipase, 799, 811-812, 812f
somatosensory areas of, 589, 590, 590f
Perivascular spaces, 765, 765f
Pancreatic polypeptide, 961
Parietal pain, 604-605, 605f
Pernicious anemia, 423, 427, 797, 820, 877
Pancreatitis, 800, 821-822
Parieto-occipitotemporal association area,
Peroxisomes, 16, 432
Panhypopituitarism, 926
716, 716f
Petit mal epilepsy, 743f, 744, 744f
Panting, 893-894
Parkinson’s disease, 711
pH, 384. See also Acid-base balance.
Pantothenic acid, 875t, 877
Parotid glands, 793-795, 795f
alveolar ventilation and, 388-389, 388f,
Papilla of Vater, gallstone of, 821-822
Paroxysmal tachycardia, 151-152
389f, 517, 517f, 519-520, 519f
Papillary muscles, 109, 109f
atrial, 152, 152f
carbon dioxide transport and, 512
Papilledema, 766
ventricular, 152, 152f
in respiratory insufficiency, 524
Para-aminohippuric acid (PAH)
Pars intermedia, 918, 956-957
measurement of, 398-400, 399f, 400f
clearance of, 345-346, 346f
Partial pressures, 491-492. See also Carbon
respiration and, 516-517, 516f, 517f
transport maximum in, 331
dioxide, partial pressure of; Oxygen,
blood flow and, 202-203
tubular secretion of, 334
partial pressure of.
cerebral blood flow and, 761, 762f
Parachute jumps, 543
Parturition, 8-9, 929, 1036-1038, 1037f
extracellular, 46f
Paracrines, 905
Past pointing, 706-707
abnormalities of, 396-397, 396t. See also
Paralysis
Patch clamp testing, 48-49, 48f
Acidosis; Alkalosis.
diver’s, 548-549, 548f
Patent ductus arteriosus, 274-275, 274f,
Henderson-Hasselbalch equation for,
of swallowing mechanism, 819
1046-1047
386
Paralysis agitans, 711
heart sounds in, 271f
intracellular, 46f, 384, 384t, 387-388
Parasitic infection, eosinophils in, 436
pulse pressure in, 173, 173f, 174
normal, 7t, 384, 384t
Parasympathetic nervous system. See also
Pco2. See Carbon dioxide, partial pressure
of chyme, 786
Sympathetic nervous system.
of.
oxygen-hemoglobin dissociation curve
acetylcholine secretion by, 750-751
Pellagra, 876
shifts and, 507-508, 508f
anatomy of, 750, 750f
Pelvic nerves, 311f, 312, 313, 775
urinary, 384, 384t, 392
Index
1101
Phagocytosis, 19-20, 430, 431-432
Pituitary gland (Continued)
Plasma cells, 429, 443
macrophages in, 432, 435
gonadotropins of. See Follicle-
Plasma colloid osmotic pressure, 185, 185f,
neutrophils in, 432
stimulating hormone; Luteinizing
188
Phagosome, 432
hormone.
Plasma proteins
Pharyngoesophageal sphincter, 782, 782f
growth hormone of, 921-927. See also
blood viscosity and, 169-170
Pharynx, in swallowing, 782-783, 782f
Growth hormone.
carbon dioxide reaction with, 511
Phenoxybenzamine, 759
hormones of, 907t, 918, 919f
colloid osmotic pressure and, 185-186,
Phentolamine, 759
hypothalamic regulation of, 919-921,
185f, 188-189
Phenylalanine, 853f
920f, 921t
cortisol effects on, 952
Phenylephrine, 759
hypothalamic-hypophysial portal
decrease in, 303-304
Phenylthiocarbamide, taste blindness for,
vessels of, 920-921, 920f
formation of, 855, 862
664
inhibin effects on, 1007, 1015, 1020
functions of, 855-857, 855f, 856f
Phonation, 481, 481f
neural control of, 733f, 734
in liver cirrhosis, 381
Phonocardiogram, 271, 271f, 272
thyroid-stimulating hormone of,
in nephrotic syndrome, 381
Phosphate
938-939
macrophage processing of, 855
excretion of, 372-373, 980, 985
antidiuretic hormone release by, 359-360,
tissue protein equilibrium with, 855, 855f
parathyroid hormone and, 987
359f
Plasmablasts, 443
extracellular, 46f, 978, 979
corticotropes of, 919, 920t
Plasmalemmal vesicles, 182, 182f
fetal, 1043, 1043f
fetal, 1036
Plasmin, 464
in bone, 981
gonadotropes of, 919, 920t
Plasminogen, 464
intestinal absorption of, 815, 980, 985, 987
lactotropes of, 919, 920t
Platelet(s), 421f, 429
intracellular, 4, 46f
posterior, 919, 919f, 927-929, 928f
concentration of, 430
parathyroid hormone effects on, 986-988,
antidiuretic hormone of, 927-929. See
deficiency of, 465
986f
also Antidiuretic hormone (ADH).
formation of, 430f, 431, 457
plasma, in chronic renal failure, 410, 410f
cells of, 927
functions of, 457-458
reabsorption of, 372-373
hormone secretion by, 907t
in clot retraction, 460
failure of, 413
hypothalamic regulation of, 919-921,
life span of, 431, 458
serum, in chronic renal failure, 412
920f, 921t, 927-928, 928f
membrane of, 458
urinary, 372-373
oxytocin of, 927, 929. See also Oxytocin.
Platelet plug, 457-458
Phosphate buffer system, 387, 393, 393f
somatotropes of, 919, 920t
cis-Platinum, renal toxicity of, 406
Phosphatidylinositol biphosphate, 914, 914f
thyrotropes of, 919, 920t
Pleural cavity
Phosphocreatine, 882
Pituitary stalk, 927
fluid in. See Pleural fluid.
in skeletal muscle contraction, 79
Placenta, 1029-1031, 1029f
pain in, 604
intracellular, 294t
anatomy of, 1029-1030, 1030f
Pleural fluid, 472, 489-490, 490f
Phosphodiesterase, in rod function, 631
blood flow in, 1035
effusion of, 140-141, 490
Phosphofructokinase, inhibition of, 836
carbon dioxide diffusion through, 1031
Pleural pressures, 489-490
Phosphogluconate pathway, 837-838, 838f
delivery of, 1038
cardiac output and, 237-238, 238f
Phospholipase, 799
estrogen secretion by, 1032-1033
in respiration, 472, 472f
Phospholipase C second messenger system,
hormones of, 907t
Pleural space, 489
914, 914f, 914t
human chorionic somatomammotropin
Pluripotential hematopoietic stem cells, 420,
Phospholipids, 12f, 846-847, 847f. See also
secretion by, 1033
421f, 430, 440
Lipid(s).
membrane of, 1030
Pneumonia, 527-528, 528f
digestion of, 812
nutrient diffusion through, 1031
Pneumotaxic center, 514, 515, 515f
extracellular, 46f, 294f
oxygen diffusion through, 1030-1031,
Po2. See Oxygen, partial pressure of.
formation of, 847
1031f
Podocytes, 316, 316f
functions of, 847
permeability of, 1030
Poiseuille’s law, 168, 168f
hydrolysis of, 841
progesterone secretion by, 1033
Poison ivy, 449
intracellular, 46f
waste excretion through, 1031
Polarography, 525
plasma, 294f
Plaque, atherosclerotic, 848, 849f
Poliomyelitis, swallowing in, 819
insulin deficiency and, 966
Plasma, 293. See also Blood.
Polycystic ovary syndrome, 974
transport of, 841-842
agglutinins in, 452
Polycythemia
Phosphoric acid, 27, 28f
amino acids of, 854-855. See also Plasma
blood viscosity and, 169, 169f
Phosphorus, 878t, 879, 879t
proteins.
hematocrit in, 169f, 293
blood, 979
anion gap of, 400, 400t
physiologic, 427-428
Phosphorylase, 832, 964
calcium in, 371-372, 372f
secondary, 427-428
Photopsins, 631
cholesterol of, 848
Polycythemia vera, 428, 531
Physical activity. See also Exercise.
chylomicrons of, 841
Polymenorrhea, 938
energy for, 887, 887t
composition of, 293-295, 294f, 294t
Polyneuritis, 876
obesity and, 872, 873-874
corrected osmolar activity of, 294t, 298
Polyphagia, 974
Physiologic dead space, 478, 500-501
creatinine concentration of, 344-345, 346f
Polyribosomes, 33
Physiologic shunt, 500, 501, 503, 503f
fatty acids in, 841
Pons, 558. See also Brain stem.
Physiology, 3
lipoproteins of, 841
reticular nuclei of, 691f, 692
Physostigmine, 89
loss of, shock and, 284-285
Pontine reticulospinal tract, 692, 692f
Pilocarpine, 759
measurement of, 296, 296t
Portal hypertension, 860
Piloerection, 97-98, 895
nonelectrolytes of, 294f
Portal vein, 860
Pineal gland, 906f, 1009-1010
osmolar clearance of, 357
Position sensation, 585, 594-595, 595f
Pinna, 660
osmolarity of, 294t, 298
baroreceptors and, 210
Pinocytosis, 19, 19f, 330, 813
antidiuretic hormone secretion and,
muscle spindles in, 595, 678. See also
Pituicytes, 927
358-360, 358f, 360t
Muscle spindles.
Pituitary gland, 906f
plasma sodium and, 358
transmission of, 595, 595f
anterior, 918-919, 919f
osmotic pressure of, 294t, 298. See also
Positive feedback, 8-9, 8f
ACTH secretion by, 955-957, 956f
Colloid osmotic pressure.
Positive supportive reaction, 682
blood supply to, 734
proteins of. See Plasma proteins.
Postural reflexes, 682
cells of, 919, 919f, 920t
renal flow of, 345-346, 346f
Potassium. See also Hyperkalemia;
cortisol effects on, 955
transfusion of, in shock, 286
Hypokalemia.
feedback regulation of, 939, 939f
vs. serum, 460
active transport of, 53-54, 53f, 59-60, 60f
1102
Index
Potassium (Continued)
Precapillary sphincter, 181, 182f, 197, 197f
Pressure(s) (Continued)
blood flow and, 202
intermittent contraction of (vasomotion),
leg, 177, 177f, 178-179, 178f
channel leakage of, 60, 60f
182-183, 187, 197-198, 197f
osmotic, 51-52, 52f. See also Osmotic
depletion of, 368
Precellular life forms, 18
pressure.
excretion of, 328t, 365-367, 366f, 366t, 367f
Precipitation, antigen-antibody, 444
partial, 491-492. See also Carbon dioxide,
sodium intake and, 370-371, 371f
Prednisolone, 947t
partial pressure of; Oxygen, partial
exercise-related loss of, 1065
Prednisone, 947
pressure of.
extracellular, 46f, 294f, 294t, 365-371
Preeclampsia, 227-228, 1035-1036
pleural, 237-238, 238f, 472, 472f, 489-490
aldosterone blockade and, 370, 370f
Prefrontal association area, 716-717, 716f,
portal vein, 860
aldosterone secretion and, 369-370,
719-720
pulmonary artery, 109, 162-163, 163f, 484,
369f, 370f, 950
injury to, 720
484f
cell lysis and, 366
Prefrontal cortex, in feeding behavior, 870
reabsorption, 189
disorders of. See Hyperkalemia;
Prefrontal lobes, in schizophrenia, 746
receptors for. See Baroreceptor(s).
Hypokalemia.
Prefrontal lobotomy, 720
recoil, 472f, 473
distribution of, 366-367, 366f, 366t
Pregnancy. See also Fetus.
right atrial, 176, 177, 179, 240-243,
exercise and, 366
aldosterone secretion in, 1034
240f-243f. See also Cardiac output.
myocardial infarction and, 254
anterior pituitary gland in, 1034
sensation of, 585-587
normal value for, 7t, 365, 370, 370f
blood volume during, 1035, 1035f
systolic, 110-111, 110f
osmolarity and, 366-367
blood volume in, 381
transpulmonary, 472f, 473
renal potassium secretion and, 368,
cardiac output during, 1035
tricuspid valve, 179, 179f
369-370, 369f, 370f
eclampsia in, 1035-1036
under water, 545, 546f
in acidosis, 366, 371
human chorionic gonadotropin in, 1007,
vapor, 492
in action potential, 61-65, 61f-64f, 66
1032, 1032f
venous. See Venous pressure.
in bone, 981
human chorionic somatomammotropin in,
ventricular, 107f, 108, 110-111, 110f,
in cardiac failure, 263-264
1033
483-484, 484f
in cardiac muscle function, 105, 113-114
metabolism during, 1034
Pressure difference
in diffusion potential, 57, 58f
nutrition during, 1034-1035
in blood flow, 164, 164f
in hemorrhagic shock, 283
oxytocin in, 929
in diffusion, 493
in hydrochloric acid formation, 796-797,
parathyroid hormone in, 1034
Pressure diuresis, 216, 323, 341-342,
796f
placenta in. See Placenta.
374-376, 375f, 376f
in membrane potential, 57, 58f, 59-60, 60f
placental estrogen in, 1032-1033
aldosterone escape and, 948-949, 948f
in resting membrane potential, 60, 60f
placental progesterone in, 1033
quantitation of, 217-220, 217f-220f
in vasodilation, 202
preeclampsia in, 227-228, 1035-1036
Pressure natriuresis, 216, 323, 341-342,
intake of, 367
relaxin in, 1034
374-376, 375f
interstitial, 294t
respiration during, 1035
aldosterone escape and, 948-949, 948f
intestinal absorption of, 815
thyroid hormones in, 1034
angiotensin II in, 378, 378f
intracellular, 4, 46f, 294f, 294t, 365,
toxemia of, 227-228, 1035-1036
in hypertension, 228-229
366-367, 366f, 366t
urine formation during, 1035
Pressure pulse, 173, 173f
loop of Henle transport of, 334-336, 335f
weight gain in, 1034
abnormal, 173f, 174
Nernst potential for, 565
Prekallikrein (Fletcher factor), 459t
aortic, 173, 173f
neuronal concentration of, 564f, 565
Preload, 111
damping of, 174-175, 174f
reabsorption of, 328t, 338f, 367, 367f
Prelymphatics, 190
peripheral transmission of, 174-175,
potassium depletion and, 368
Premature atrial contractions, 150, 150f
174f
renal filtration of, 328t, 367, 367f
Premature atrioventricular contractions,
Presynaptic membrane, 560
salivary, 794
150-151, 151f
Presynaptic terminals, 559-560, 559f, 560f
secretion of, 338f, 367-371, 367f
Premature contractions, 150-151, 150f,
Primordial follicle, 1013, 1013f
acidosis and, 371
151f
Principal cells, 336-337, 336f
aldosterone and, 342, 342t, 366,
atrial, 150, 150f
potassium secretion by, 367-368, 368f
369-370, 369f, 370f, 948
atrioventricular, 150-151, 151f
Procaine, 70
distal tubular flow rate and, 370-371,
ventricular, 151, 151f
Procarboxypolypeptidase, 799
371f
Prematurity, 1050-1051
Procaspases, 40
extracellular potassium concentration
Premotor area, 686, 686f
Procoagulants, 459
and, 368, 369-370, 369f, 370f
Preprohormones, 906
Proelastase, 810, 810f
principal cells in, 336-337, 336f, 337f,
Presbyopia, 618
Proerythroblasts, 421f, 423
367-368, 368f
Pressoreceptors. See Baroreceptor(s).
Progesterone, 907t
urinary, 367
Pressure(s)
blood transport of, 1016
Potassium channel, 47-48, 47f
alveolar, 472-473, 472f
breast effects of, 1018
of cardiac muscle, 67-68, 67f, 117
aortic, 109, 173, 173f
degradation of, 1017
of neuron, 566, 566f
arterial. See Arterial blood pressure.
fallopian tube effects of, 1018
of postsynaptic neuron, 561, 561f, 562
capillary, 185-187, 185f, 186f. See also
in female reproduction, 1011, 1013f, 1016,
voltage clamp testing of, 63, 63f
Capillary pressure.
1016f, 1018, 1019
voltage-gated, 62-63, 62f, 64, 64f, 66-67
cardiac, 110-111, 110f, 162. See also at
insulin secretion and, 969
activation of, 62-63, 62f
Atrium (atria); Ventricle(s),
placental secretion of, 1033
Potassium citrate, 467
cardiac.
synthesis of, 1016
Potassium-sodium leak channel, 60, 60f
cerebrospinal fluid, 213, 213f, 765-766
uterine effects of, 1018
Potassium-sparing diuretics, 337, 403t, 404
circulatory, 162-163, 163f
Prognostication, 720
Potential spaces
diastolic, 110-111, 110f
Prohormones, 906
capillary fluid exchange with, 305-306
difference in, 164, 164f
Proinsulin, 962
fluid in, 305-306
end-diastolic, 110, 111
Prolactin, 907t, 918, 919f, 920t
hydrostatic pressure of, 306
filtration, 185-186, 185f, 189
in lactation, 1039-1040, 1040f
protein of, 306
hydrostatic. See Hydrostatic pressure.
in pregnancy, 1034
Power law, 594, 594f, 657
in dural sinuses, 178
Prolactin inhibitory hormone, 921, 921t,
P-Q interval, 125
interstitial. See Interstitial fluid pressure.
1040
P-R interval, 125
intra-abdominal, 177
Prolactin-inhibiting hormone, 907t
in atrioventricular heart block, 149, 149f
intramyocardial, 250, 250f
Prolyl hydroxylase, 877
Prealbumin, 934
intraocular, 624-625, 624f
Prometaphase, 37f, 39
Index
1103
Promoter, 31, 35, 35f
Protein(s) (Continued)
Pulmonary circulation (Continued)
Proopiomelanocortin, 956-957, 957f
potential space, 306
gaseous interface with. See Respiratory
Pro-opiomelanocortin neurons, 868-870,
reabsorption of, 330
membrane.
869f
repressor, 35-36, 35f
high altitude effects on, 539-540
Prophase, 37f, 39
specific dynamic action of, 887
hydrostatic pressure gradients in, 485-487,
Propranolol, 759
starvation effects on, 857
485f, 486f, 488, 488f
Proprioceptive receptors, 594-595, 696. See
storage of, 854-855
in left-sided heart failure, 487
also Muscle spindles; Pacinian
structural, 11
left atrial pressure and, 487
corpuscles.
synthesis of, 20, 33-35, 34f, 854-855
oxygen absorption and, 244, 244f
Propriospinal fibers, 675
aldosterone and, 949-950
physiologic anatomy of, 483
Propylthiouracil, 939
chemical steps in, 33-34, 34f
pressures in, 162-163, 163f, 483-484, 484f
Prorenin, 223
estrogen and, 857, 1017-1018
shifts in, 485
Prosecretin, 801
growth hormone and, 857, 922
vascular resistance in, 167
Prosophenosia, 717
insulin and, 857, 966-967
zones of, 485-486, 486f
Prostaglandins
steroid hormones and, 915
Pulmonary edema, 488-489
in male reproduction, 999
testosterone and, 857, 1005, 1006
in aortic valve lesions, 273
in renal blood flow, 322-323, 322t
thyroid hormones and, 857, 934, 935f
in cardiac failure, 262, 264
Prostate gland, 996, 997f
tissue, 855, 855f, 857
in mitral valve lesions, 273
function of, 999
transport, 45, 816. See also Protein(s),
in patent ductus arteriosus, 275
tumors of, 1008
membrane.
in unilateral left-sided cardiac failure, 262
Prostatic fluid, 1000
triglyceride formation from, 845
Pulmonary embolism, 466
Protanope, 633, 634f
urinary, 317, 409
Pulmonary function studies, 475-477, 475f,
Protein(s)
Protein C, 463
476f, 477t
activator, 35f, 36
Protein channels, 47-49, 47f
Pulmonary membrane. See Respiratory
amino acids of, 852-854, 853f. See also
gating of, 48-49, 48f
membrane.
Amino acids.
ligand-gated, 48
Pulmonary valve, 109
carbohydrate formation from, 838
patch-clamp recording of, 48f, 49
congenital stenosis of, 138-139, 139f
carrier, 13, 45, 46f, 47, 49-50, 53-54, 53f,
selective permeability of, 47-48, 47f
Pulmonary vascular resistance, 167. See also
831
voltage-gated, 48-49, 48f
Vascular resistance.
channel, 45, 46f, 47-49, 47f, 48f
Protein kinase C, 914, 914f
Pulmonary vein, 483
chromosomal, 38
Proteinuria, 317, 409
distensibility of, 171
complete, 857
Proteoglycan, 14
pressure in, 484
daily requirement for, 857, 866
Proteoglycan filaments, of interstitium,
Pulmonary ventilation, 471-481. See also
deficiency of, 925, 925f
184-185, 184f, 305
Alveolar ventilation.
depletion of, 874-875, 874f
Proteolytic enzymes, 432, 1000
alveolar pressure in, 472-473, 472f
dietary, 866, 866t, 867
Prothrombin (factor II), 459-460, 459f,
lung compliance in, 473-474, 473f
daily requirement for, 857, 866
459t
mechanics of, 471-475, 472f
digestion of, 808-809, 810-811, 810f
Prothrombin activator, 459, 459f, 461-464,
pleural pressure in, 472, 472f
energy content of, 865-866, 866t
461f, 462f
spirometric evaluation of, 475-477, 475f,
pancreatic digestion of, 810, 810f
Prothrombin time, 467-468, 467f
476f, 477t
pepsin digestion of, 810, 810f
Protoplasm, 11-12
surface tension in, 474
renal blood flow and, 325
Psychic blindness, 870
surfactant in, 474
urine concentration and, 353
Psychogenic diarrhea, 823
thoracic cage in, 474-475
differentiation, 420
Psychomotor epilepsy, 744f, 745
transpulmonary pressure in, 472f, 473
endoplasmic reticulum formation of, 20,
Psychosis, 745-746
Pulmonary volumes, 475, 475f, 476f, 477t
21f
Pteroylglutamic acid. See Folic acid.
Pulmonary wedge pressure, 484
extracellular, 46f, 294f, 294t
Ptyalin, 793, 809, 809f
Pulp cavity, 992f, 993
functional, 11-12
Puberty
Pulse deficit, 150
hydrolysis of, 808-809, 810-811, 810f
female, 1021-1022, 1022f
Pulse pressure, 173
incomplete, 857
male, 1007-1008, 1008f
abnormal, 174, 174f
integral, 13, 14, 14f
Pudendal nerve, 311f, 312, 313, 789
age-related changes in, 173, 173f
interstitial, 188-189, 193-194, 294t
Pulmonary artery, 483
Punishment centers, 725, 735-736
in edema prevention, 305
distensibility of, 171
Pupil. See also Eye(s).
intestinal absorption of, 816
in patent ductus arteriosus, 275
Argyll Robertson, 650
intracellular, 11-12, 46f, 294f, 294t,
low-pressure receptors of, 212
autonomic nervous system control of,
387-388
Pulmonary artery pressure, 109, 162-163,
649-650, 753, 754t
cortisol effects on, 952
163f, 484, 484f
diameter of, 618-619, 618f, 632, 649-650
lymph, 192
Pulmonary capacities, 475-476, 475f, 476f,
in dark/light adaptation, 632
membrane, 13-14, 14f
477t
in Horner’s syndrome, 650
carrier, 46f, 47, 49-50, 53-54, 53f
Pulmonary capillaries
light reflex of, 649
channel, 46f, 47-49, 47f, 48f
blood flow through, 485-486, 487
Pupillary light reflex, 649
metabolism of, 852-856
diameter of, 497
Purines, 27, 28f, 29, 29f, 37
cortisol effects on, 857, 952
dynamics of, 487-489, 488f, 489f
Purkinje cells, of cerebellum, 701-702, 702f,
deamination in, 856
fluid exchange in, 487-488, 488f
703
heat production and, 885
oxygen diffusion into, 502-503, 503f
Purkinje fibers
hormonal regulation of, 857
pressures in, 162-163, 163f, 484, 484f, 487,
action potential transmission in, 117f, 119,
in Cushing’s syndrome, 958-959
488, 488f
120f, 121
insulin and, 857, 966-967
size of, 496, 497f
blockade of, 141
liver in, 862, 952
Pulmonary capillary pressure, 162-163, 163f,
in atrioventricular (A-V) block, 121
measurement of, 866
484, 484f, 487, 488, 488f
intrinsic rhythmical rate of, 120
oxidation in, 856-857
Pulmonary circulation, 483-490
Purpura, thrombocytopenic, 465
thyroid hormones and, 857, 934, 935f
alveolar oxygen and, 485
Pus, 435-436
obligatory degradation of, 857
cardiac output and, 486-487, 487f
Putamen, 688, 708, 708f
partial, 857, 866
distribution of, 162f, 485-487, 485f-487f
lesions of, 709
peripheral, 13, 14f
exercise effect on, 485f, 486-487, 487f
Putamen circuit, 708-709, 708f
plasma. See Plasma proteins.
gas exchange in. See Gas exchange.
Pyelonephritis, 409
1104
Index
Pyloric gland, 797-798. See also Gastrin.
Receptor potential, 573-576, 574f, 575f
Reflex(es) (Continued)
Pyloric pump, 785
action potential and, 574, 574f. See also
galloping, 683
Pyloric sphincter, 774, 785
Action potential, neural.
gastrocolic, 776, 789
Pylorus, in stomach emptying, 785
maximum amplitude of, 574
gastroenteric, 787
Pyramidal (corticospinal) tract, 687-688,
mechanisms of, 574
gastroileal, 787
688f
of hair cells, 656
gastrointestinal, 775-776
lesions of, 691
of pacinian corpuscle, 574-575, 574f,
Hering-Breuer, 515-516
Pyridostigmine, 759
575f
light, 649
Pyridoxine, 875t, 877
of rods, 629-630, 630f
low-pressure, 212
Pyrimidines, 27, 28f, 29, 29f, 37
of taste buds, 665
mark time, 682-683
Pyrogens, 898-899
stimulus intensity and, 574-575, 575f
mass, 684
Pyrophosphate, 981
Reciprocal inhibition circuits, 580, 581f, 680f,
micturition, 313-314
Pyruvic acid, 22, 22f
681-682, 682f, 691
myenteric, 776-777
acetyl coenzyme A conversion from, 23,
Recoil pressure, 472f, 473
peristaltic, 776-777
833, 833f
Rectum
peritoneointestinal, 790
formation of, 22-23, 833, 833f
emptying of, 789-790
postural, 682
lactic acid conversion from, 837
empyting of, 757, 790f, 822, 823
renointestinal, 790
Rectus muscles, of eye, 645, 645f
scratch, 587, 683
Red blood cell(s), 419-426
sneeze, 480
antigens of, 451-453, 452f, 452t, 453t
stretch, 676-677, 677f, 678-679, 678f
Q
carbonic anhydrase of, 419
swallowing, 783
Q wave, 107, 107f, 123, 124f
concentration of, 419
ureterorenal, 312
QRS complex, 107, 107f, 123, 124-125, 124f,
cortisol effects in, 954
vesicointestinal, 790
125f
destruction of, 180, 426
visceral, 748
bizarre patterns of, 139f, 141
diameter of, 419
volume, 212
current of injury effect on, 141-143, 142f
differentiation of, 420-422, 421f
Refraction, 613-615, 614f, 615f
high-voltage, 138f, 139f, 140
fetal, 1042
errors of, 619-621, 619f, 620f
in premature ventricular contractions,
functions of, 419
Refractive index, 613, 617, 617f
151, 151f
hemoglobin of, 419-420, 424-425, 424f.
Refractive power, 616-617, 616f
low-voltage, 140-141, 140f
See also Hemoglobin.
Regression, tissue, 20
mean electrical axis of, 137-140, 137f-140f
high altitude effects on, 422, 539
Regulatory genes, 36
prolongation of, 139f, 140f, 141
in pulmonary capillaries, 496, 497f
Regulon, 36
vectorial analysis of, 134, 135f
life span of, 426
Rehearsal, memory and, 726, 737
QRS vectorcardiogram, 137, 137f
maturation of, 423
Reissner’s membrane, 652-653, 652f, 653f
Q-T interval, 125-126
neonatal, 1047-1048, 1048f
Relaxin, 1034
packed volume of (hematocrit), 293
Renal arteries, 310
production of, 420-423, 420f, 421f
constriction of, renin secretion and,
cyanocobalamin in, 423
226-227, 226f
R
erythropoietin in, 422-423, 422f
disease of, 408, 413
R wave, 107, 107f, 123, 124f, 134, 135f
folic acid in, 423
fibromuscular hyperplasia of, 408
Radiation (heat), 891, 891f
hypoxia and, 422-423, 422f, 427-428
pressure in, 320-321, 321t
Radiation exposure
lung disease and, 422
Renal failure. See Kidneys, failure of.
in submarine, 550
regulation of, 422-423, 422f
Renal function curve, 217, 217f
mutation and, 41
sites of, 420, 420f
shift in, 218-219, 218f
Radiation therapy, leukopenia and, 437
tissue oxygenation and, 422
sodium-loading, 229, 229f
Radioimmunoassay, for hormone
vitamin B12 in, 423
Renal papillae, 309, 309f, 310
measurement, 915-916, 916f
shape of, 419, 421f
Renal pelvis, 309, 309f
Rage, 735-736, 737, 738, 758
size of, 419
Renal plasma flow, 345-346, 346f
Ramp signal, 514-515
splenic storage of, 179-180, 180f
Renal pyramids, 309, 309f
Ranitidine, in peptic ulcers, 821
testosterone effects on, 1006
Renal tubular acidosis, 397
Ranvier, node of, 68, 68f
transfusion of, 451-455, 452f, 452t, 453t
Renal tubules, 309f, 310, 310f. See also
Raphe nuclei, 730f, 731, 740
volume of, 419
Glomerulus; Kidneys; Loop of Henle.
lesions in, 740
Red nucleus, 688-689, 689f, 690, 705
ammonia buffer system in, 393-394,
Rathke’s ouch, 918
Red pulp, 180, 180f, 434
394f
Reading, 716, 716f, 719, 721f, 722
Red-green color blindness, 633
failure of, 357-358, 414
Reagins, 449
Referred pain, 603-605, 603f, 605f
in urine concentration, 348-349,
Receptor(s), 14
Reflex(es)
350-351, 355-357, 928
acetylcholine, 86-87, 86f, 87f, 752
abdominal compression, 213
osmoreceptor interaction with, 358-361,
adrenergic, 752, 753t
atrial, 212
358f-360f
aldosterone, 949-950
attenuation, 652
phosphate buffer system in, 393, 393f
enzyme-linked, 911-912, 912f, 913-914,
autonomic, 757
bicarbonate titration by, 392
913f, 913t, 914f, 914t
Bainbridge, 212, 233
connecting, 310, 310f
G protein-linked, 911, 911f
baroreceptor, 209-212, 757. See also
cortical collecting, 310, 310f, 327
hormone, 14, 910-912, 911f-913f. See also
Baroreceptor(s).
antidiuretic effects on, 928
Hormone(s), receptors for.
bladder emptying, 757, 758
bicarbonate formation in, 393-394,
in pinocytosis, 19, 19f
chemoreceptor, 211-212. See also
394f
insulin, 962-963, 963f
Chemoreceptor(s).
chloride reabsorption in, 336-337, 336f,
intracellular, 912, 913f
chewing, 781
337f
ion channel, 910-911
colonoileal, 776
hydrogen ion secretion by, 392, 392f
leptin, 911-912, 912f
cough, 480
in urine concentration, 353, 353f, 356,
low-pressure, 212
crossed extensor, 680f, 681, 681f
356f, 928
muscarinic, 752
defecation, 757, 758, 789-790, 790f, 822
in urine dilution, 349f, 350
nicotinic, 752
duodenocolic, 789
osmolarity in, 349f, 350, 356, 356f
sensory. See Sensory receptors.
enterogastric, 776, 785-786, 799
potassium secretion by, 336-337, 336f,
stretch. See Baroreceptor(s).
reverse, 799
337f, 367-371, 367f-371f
thyroid hormone, 934, 935f
flexor, 680-681, 680f, 681f
resorption by, 336-337, 336f, 337f
Index
1105
Renal tubules (Continued)
Renal tubules (Continued)
Renal tubules (Continued)
sodium reabsorption by, 336-337, 336f,
active transport in, 328-332, 328f-331f
hydrogen ion, 390-392, 390f-392f. See
337f
aldosterone effect on, 342, 342t
also Hydrogen ions, secretion of.
disorders of, 413-414
amino acid, 330, 330f, 413, 854
potassium, 367-371, 368f, 369f. See also
distal, 309f, 310, 310f, 327
angiotensin II effect on, 342-343, 342t
Potassium, secretion of.
calcium reabsorption in, 372
antidiuretic hormone effect on, 342t,
toxin effects on, 334, 406
cells of, 54, 336-337, 336f
343
Renal urinary output curve (renal function
flow rate of, potassium secretion and,
arterial pressure in, 341-342
curve), 217, 217f
370-371
atrial natriuretic peptide effect on, 342t,
shift in, 218-219, 218f
hydrogen ion secretion by, 54, 391, 391f,
343
sodium-loading, 229, 229f
392, 392f
bicarbonate, 337, 390-392, 390f-392f,
Renal vein, 310
in urine concentration, 352-353, 353f,
404
Renin, 223-226, 223f, 224f, 907t
356, 356f
calcium, 343, 372, 372f, 373t
in renal ischemia, 413
in urine dilution, 349f, 350
calculation of, 339-340, 346-347
secretion of, 308
osmolarity in, 350, 356, 356f
carbonic anhydrase inhibitor effect on,
chronic, 227
potassium secretion by, 367-371,
404
in cardiac failure, 263
367f-371f
chloride, 332-333, 332f, 336, 336f
renal artery constriction and, 226-227,
solute resorption in, 336-337, 336f, 337f
cortical collecting tubule in, 336-337,
226f
diuretic effects on, 402-403, 403t
337f, 338-339, 338f
tumor-related, 226
excretion by, 314-315, 314f
co-transport in, 329-330, 330f, 333-334
Renin-angiotensin system, 223-230, 223f,
acid-base, 390-400. See also Acid-base
counter-transport in, 330, 330f, 333
230f, 231. See also Angiotensin II.
balance, renal regulation of;
diffusion in, 332-333, 332f
aldosterone secretion and, 950, 951f
Acidosis; Alkalosis.
disorders of, 413-414
in cardiac failure, 263
calcium, 371-372, 372f, 373t, 980, 985,
distal tubule in, 336-337, 336f, 338-339,
salt retention and, 224-226, 225f, 226f
987
338f
vasoconstrictor pressure response to, 224,
clearance method quantification of,
filtration coefficient in, 340
224f
343-347, 344t, 345f, 346f
glomerular filtration rate and, 327-328,
water retention and, 224-226, 226f
magnesium, 373
328t
Renointestinal reflex, 790
phosphate, 372-373, 980, 985, 987
glomerulotubular balance in, 339
Renshaw cells, 675
potassium, 328t, 365-367, 366f, 366t,
glucose, 330, 330f, 331, 331f, 413
Repressor operator, 35-36, 35f
367, 367f
gradient-time transport in, 331-332
Reproduction, 6
rates of, 357
hormonal control of, 342-343, 342t
cell, 37-40, 37f
sodium, 307-308, 308f. See also Sodium,
increase in, 339
female, 1011-1025. See also Sexual
excretion of; Urine, concentration
interstitial pressures in, 340-341
function, female.
of.
loop of Henle in, 334-336, 335f,
abnormalities of, 1023
toxin, 307
338-339, 338f
anatomy of, 1011, 1012f
urea, 307, 353-354
medullary collecting duct in, 337-338,
estradiol in, 1016-1018, 1016f
water. See at Urine.
338f
follicle-stimulating hormone in,
inulin concentrations in, 339
net, 346-347
1012-1013, 1013f, 1020
medullary collecting duct, 309f, 310, 310f
osmosis in, 332
gonadotropin-releasing hormone in,
bicarbonate formation in, 393-394, 394f
paracellular pathway in, 329, 332, 335
1020
cells of, 337-338, 338f
parathyroid hormone effect on, 342t,
hormones in, 1011-1012
hydrogen ion secretion by, 392, 392f
343
hypothalamic-pituitary-ovarian
in urine concentration, 353, 353f,
passive mechanisms of, 332-333,
feedback system in, 1019-1021,
356-357, 356f
332f
1020f
osmolarity of, 349f, 350, 356-357, 356f
peritubular capillary hydrostatic
inhibin in, 1020
reabsorption by, 337-338, 338f
pressure in, 340, 341f, 341t
luteinizing hormone in, 1012-1013,
necrosis of, 406
phosphate, 372-373
1013f, 1020-1021, 1020f
net acid excretion by, 394-395
disorders of, 413
menstrual cycle in, 1012-1015, 1013f
potassium processing by, 367-371,
physical forces in, 339-341, 340f
progesterone in, 1016, 1016f, 1019
367f-371f. See also at Potassium.
pinocytosis in, 330
regulation of, 1019-1021, 1020f, 1022f
proximal, 309f, 310, 310f, 327
potassium, 337, 337f, 367, 367f
homeostasis and, 6
amino acid transport in, 330, 330f
protein, 330
male, 996-1010. See also Sexual function,
bicarbonate formation in, 393, 394f
proximal tubule in, 333-334, 333f, 334f,
male.
bicarbonate reabsorption in, 390-392,
338-339, 338f
abnormalities of, 1001, 1001f
390f-392f
quantification of, 343-347, 344t, 345f,
anatomy of, 996, 997f
bile salt secretion by, 334
346f
hormones in, 1003-1008, 1003f, 1004f,
calcium reabsorption in, 372
rate of, 339-340
1008f
catecholamine secretion by, 334
regulation of, 339-343, 340f, 341f, 341t,
pineal gland in, 1009-1001
cells of, 333, 333f
342t
prostate gland in, 999
chloride reabsorption in, 333-334, 334f
selectivity of, 327-328, 328t
semen in, 999-1001
glucose transport in, 330, 330f
sodium, 333-334, 333f, 334f. See also
seminal vesicles in, 999
hydrogen ion secretion by, 390-392,
Sodium, reabsorption of.
spermatogenesis in, 996-1001, 997f, 998f
391f, 392f
solvent drag in, 332
Reserpine, 759
organic acid secretion by, 334
summary of, 338, 338f
Residual body, 20, 20f
osmolarity in, 349, 349f, 355, 356f
sympathetic nervous system effect on,
Residual volume, 475, 476f, 477, 529
oxalate secretion by, 334
343
in asthma, 529
PAH secretion by, 334
transcellular pathway in, 329
Resin agents, 851
protein reabsorption by, 330
transport maximum for, 330-331, 331f,
Respiration, 471-481. See also Alveolar
sodium reabsorption in, 333-334, 333f,
332
ventilation; Pulmonary ventilation.
334f
tubular load in, 330-331, 331f, 339
arterial pressure and, 214
solute concentrations in, 334, 334f
urea, 333, 338, 351t, 353-354, 354f
artificial, 532, 532f
toxin secretion by, 334
water, 224-225, 332, 334. See also Water,
bronchi in, 479, 479f
urate secretion by, 334
reabsorption of.
bronchioles in, 479-480, 479f
water reabsorption in, 332, 334
secretion into, 314-315, 314f, 315f, 327
disorders of. See Respiratory distress
reabsorption by, 327-346, 328t
active transport in, 330
syndrome; Respiratory insufficiency.
1106
Index
Respiration (Continued)
Respiratory insufficiency (Continued)
Retinol, 875. See also Vitamin A (retinol).
energy for, 475
in chronic pulmonary emphysema,
Retrograde amnesia, 725
fetal, 1042-1043
526-527, 527f, 528f
Retrolental fibroplasia, 200, 1051
gas exchange in. See Gas exchange.
in pneumonia, 527-528, 528f
Reverberatory circuit, 581-582, 581f-583f
in acid-base balance, 388-389, 388f, 389f,
in tuberculosis, 530
Reverse enterogastric reflex, 799
396, 397
maximum expiratory flow in, 525-526,
Reward centers, 725, 735, 735f, 736
in exercise, 516, 520-521, 520f, 521f,
525f, 526f
Reynolds’ number, 166
1061-1062, 1061f, 1062f
oxygen therapy in, 530-531, 531f
Rh blood types, 453-454
in neonate, 1047
Respiratory membrane
Rh immunoglobulin globulin, in
in pregnancy, 1035
diffusing capacity of, 498-499, 499f
erythroblastosis fetalis, 454
in premature infant, 1050
gas exchange through, 496-499, 496f, 497f.
Rheumatic fever, 271, 448
lung compliance and, 473-475, 473f
See also Carbon dioxide, transport of;
Rhodopsin, 627, 629-631
muscles of, 471-472, 472f
Oxygen, transport of.
activated, 629, 630
nose in, 479f, 480-481
diffusing capacity and, 498-499, 499f
decomposition of, 629, 629f
pressure of, 472-473, 472f
rate of, 498
in dark/light adaptation, 631-632, 632f
rate of, 477
pressure difference across, 498
in rod hyperpolarization, 629-631, 630f
regulation of, 514-523
structure of, 497, 497f
re-formation of, 629, 629f
anesthesia and, 522
surface area of, 498
vitamin A and, 629, 629f
brain edema and, 521
thickness of, 498
Rhodopsin kinase, 631
carbon dioxide in, 516-517, 516f, 517f,
Respiratory quotient, 867
Riboflavin, 875t, 876
518
Respiratory rate, 477
deficiency of, 198, 876
chemical, 516-517, 516f, 517f
Respiratory system, 5, 478-481, 479f, 481f.
Ribonucleic acid (RNA), 27
chemosensitive area in, 516-517,
See also Respiration.
growth hormone effects on, 922
516f
water loss through, 292
messenger, 31-32, 32f
during exercise, 520-521, 520f, 521f
Respiratory unit (lobule), 486f, 496, 496f
nucleotides of, 30-31
hydrogen ions in, 516-517, 516f, 517f,
Reticular excitatory area, 728-729, 729f,
ribose of, 30
518
730f, 731
ribosomal, 33-35, 34f
irritant receptors in, 521
Reticular formation, in pain sensation, 601
synthesis of, 30-31, 30f
J receptors in, 521
Reticular inhibitory area, 729, 729f
transfer, 32-33
oxygen in, 517, 518-520, 518f, 519f
Reticular lamina, 655, 655f
uracil of, 30
peripheral chemoreceptors in, 518-520,
Reticular nuclei, 691-692, 691f
Ribonucleic acid (RNA) polymerase, 31,
518f, 519f
Reticular substance, 206f, 207
35
respiratory center in, 514-516, 515f
Reticulocyte, 421f, 422
Ribose, 30
voluntary, 521
Reticuloendothelial system, 180, 432-434,
Ribosomes, 13f, 15, 18, 18f
reserve of, 1066
433f, 434f
endoplasmic reticulum attachment of, 33,
speech and, 481, 481f
Retina. See also Eye(s); Vision.
34f
spirometric evaluation of, 475-477, 475f,
action potential of, 637-638, 637f
RNA of, 33-35, 34f
476f, 477t
amacrine cells of, 627f, 634, 635, 635f, 636
d-Ribulose, 837
surfactant and, 474
anatomy of, 626-628, 627f, 628f
Rickets, 988, 991
swallowing and, 783
bipolar cells of, 627f, 633, 635, 635f, 636,
Right atrial pressure, 176
thoracic compliance in, 474-475
638f
cardiac output and, 240-243, 240f-243f.
thyroid hormone effects on, 937
blood supply of, 628
See also Cardiac output.
trachea in, 478-479
central fovea of, 626, 636
measurement of, 176, 179
voluntary control of, 521
cones of, 626-628, 627f, 628f, 631, 631f,
peripheral venous pressure and, 177
Respiratory acidosis. See Acidosis,
632-633, 633f, 635f
Right bundle branch, 117f, 118f, 119
respiratory.
dark adaptation of, 631-632, 632f
Right bundle branch block, 139-140, 140f
Respiratory alkalosis. See Alkalosis,
detachment of, 628
Right coronary artery, 249, 249f
respiratory.
electrotonic conduction in, 635
Right ventricular hypertrophy, 273
Respiratory center, 514-516, 515f
fovea of, 626, 627f, 635, 635f, 636
Righting reflex, 682
acclimatization effects on, 519, 539
ganglion cells of, 627f, 634, 635, 635f,
Right-to-left cardiac shunt, 275-276, 275f
chemosensitive area of, 516-517, 516f
636-638. See also Ganglion cells.
Rigidity, decerebrate, 692
disorders of, 523
glutamate in, 635
Rigor mortis, 83
dorsal neurons of, 514-515, 515f
horizontal cells of, 627f, 633, 635f, 638f
RNA. See Ribonucleic acid (RNA).
exercise effects on, 520-521, 520f
image formation on, 617
Rod of Corti, 655, 655f
in acid-base balance, 384
interplexiform cells of, 634
Rods, 626-628, 627f, 628f, 629-631. See also
pneumotaxic center of, 515, 515f
lateral inhibition in, 635-636, 636f
Cones.
ventral neurons of, 515, 515f
layers of, 626, 627f
dark/light adaptation of, 631-632, 632f
Respiratory distress syndrome, 1045, 1050
light adaptation of, 631-632, 632f
discs of, 627, 628f
of newborn, 474, 529
neural circuitry of, 633-636, 635f, 636f
excitation of, 629-631, 630f
Respiratory exchange ratio, 512, 867
neurotransmitters of, 635
inner segment of, 627, 628f
Respiratory insufficiency, 524-532
ophthalmoscopic examination of, 622-623,
outer segment of, 627, 628f
artificial respiration in, 532, 532f
622f
potential of, 629-631, 630f
blood CO2 in, 524-525
peripheral area of, 635, 635f, 637
rhodopsin of, 629, 629f
blood pH in, 524
pigment layer of, 628
synaptic body of, 627-628, 628f
blood Po2 in, 525
retrolental fibroplasia of, 200
vitamin A in, 629
cyanosis in, 531
rods of, 626-628, 627f, 628f, 629-631,
Rubrospinal tract, 688-689, 689f, 705
dyspnea in, 532
629f-631f, 633, 635f, 638f
Ruffini’s end-organ, 573f, 586, 595
forced expiratory vital capacity in, 526,
vitamin A in, 629
Running, 695, 696. See also Exercise.
526f
W cells of, 637
forced expiratory volume in, 526, 526f
X cells of, 637
hypercapnia in, 531-532
Y cells of, 637
hypoxia in, 530-531, 531f
Retinal, 629
S
in airway obstruction, 526-527, 526f, 527f,
vitamin A interaction with, 629, 629f
S wave, 107, 107f, 123, 124f, 134, 135f
528f, 529
Retinal artery, 628
Saccades, 646-647
in asthma, 529
Retinal isomerase, 629, 629f
Saccule, 693, 693f
in atelectasis, 528-529, 529f
Retinitis pigmentosa, 645
macula of, 693, 693f, 695
Index
1107
Saliva, 792f, 793-795, 794t
Sensory receptors, 5-6, 555, 556f, 572-584
Shock
in carbohydrate digestion, 809, 809f
accommodation of, 576
anaphylactic, 285-286, 287, 449-450
Salivary glands, 791, 792f, 793-795
adaptation of, 575-576, 575f
cardiac (cardiogenic), 237, 254,
aldosterone effects on, 949
auditory, 652, 655-656, 655f
262-263
autonomic nervous system control of, 753,
differential sensitivity of, 572-573
cardiac output in, 236f, 237
754t
downregulation of, 583-584
circulatory, 236f, 237, 278-288, 899, 949.
blood supply to, 795
nerve fibers from, 576-578, 577f, 578f
See also Circulatory shock;
Salivatory nuclei, 666
olfactory, 667-668, 667f
Hemorrhagic shock.
Salpingitis, 1025
pain, 598-599
gastrointestinal blood flow in, 780
Salt. See Sodium; Sodium chloride.
phasic, 576
glucocorticoids in, 287
Saltatory conduction, 68-69, 69f
potentials of, 573-576, 574f, 575f. See also
head-down position in, 287
Sarcolemma, 72, 90f
Receptor potential.
hemorrhagic, 279-284. See also
Sarcomere, 73, 73f
predictive function of, 576
Hemorrhagic shock.
contraction of, 74-78, 75f-78f
proprioceptive, 594-595, 696. See also
histamine, 285-286
relaxed state of, 74-75, 75f
Muscle spindles; Pacinian corpuscles.
hypovolemic, 279-285. See also
Sarcoplasm, 74, 74f
rapidly adapting, 575f, 576
Hemorrhagic shock.
Sarcoplasmic reticulum, 74, 74f, 89-91, 90f
rate, 576
insulin, 976
calcium release by, 90-91, 91f, 99, 99f
signal transduction in, 573-576, 574f,
neurogenic, 285, 287
Satiety, 867
575f
oxygen therapy in, 287
Satiety center, 734, 867-868, 868f, 871
slowly adapting, 575f, 576
plasma loss-related, 284-285
Saturation diving, 549
tactile, 585-586, 586f
septic, 282, 286
Scala media, 652, 652f, 656
taste, 664-665, 665f
septicemic, 466
Scala tympani, 652, 652f, 656
thermal, 607-609, 608f
spinal, 684
Scala vestibuli, 652, 652f, 656
tonic, 576
traumatic, 285
Scar, cardiac, 255, 255f
types of, 572, 573f, 573t
treatment of, 286-287
Schistosomiasis, 436
upregulation of, 583-584
Shock lung syndrome, 283
Schizophrenia, 745-746
visual, 626-628, 627f, 628f. See also Cones;
Shunt
Schlemm, canal of, 624, 624f
Rods.
left-to-right, 274-275, 274f
Schwann cell, 68, 68f
Sensory stimulus. See also Sensory
right-to-left, 275-276, 275f
Scopolamine, 759
receptors.
physiologic, 500, 501, 503, 503f
Scotomata, 644-645
intensity of, 593-594, 594f
Sibutramine, 873
Scotopsin, 629
interpreted strength of, 594, 594f
Sickle cell anemia, 421f, 424, 427
Scratch reflex, 683
modality of, 572-573
Sigmoid colon, spasm of, 822
Scratching, 587
receptor potential and, 574-575, 574f,
Sinus arrhythmia, 148, 148f
Scuba diving, 549-550, 550f
575f
Sinus bradycardia, 147, 148f
Scurvy, 878
Septal nuclei, 669
Sinus infection, 606f, 607
Seawater, 350
Septic shock, 282, 286
Sinus (sinoatrial) nodal block, 148, 148f
diving in. See Deep-sea diving.
Serine, 853f
Sinus (sinoatrial) node, 106, 116-118, 117f,
pressure-depth relationship of, 545,
Serotonin, 564, 602, 602f, 730-731, 730f
120f
546f
in depression, 745
acetylcholine effects on, 121
Second messenger systems, 560, 561-562,
in memory formation, 724f, 725
action potential of, 117, 117f
561f
in sleep, 740
automaticity of, 116-118, 117f
adenyl cyclase-cAMP, 913-914, 913f,
mast cell release of, 436
hyperpolarization of, 118, 121
913t
Sertoli cells, 996-997
ion channels of, 117
calcium-calmodulin, 914-915
Serum, 460
norepinephrine effects on, 122
in hormonal function, 912-915, 913f, 913t,
Sex hormone-binding globulin, 1003
parasympathetic nervous system effects
914f, 914t
Sexual function. See also Reproduction.
on, 121, 148
in postsynaptic neuron, 560, 561-562,
amygdala in, 737
resting potential of, 117, 117f
561f
female, 1023-1025
rhythmicity of, 117-118, 117f, 120
phospholipase C, 914, 914f, 914t
clitoris in, 1023
self-excitation of, 117-118, 117f
Secretin, 907t
erection in, 1023
stretch of, 212, 233
composition of, 799
lubrication in, 1023
sympathetic nervous system effects on,
in bile secretion, 804
orgasm in, 1023-1024
121, 148
in gastrointestinal motility, 776
stimulation of, 1023
Sinus tachycardia, 147, 148f
in insulin secretion, 969
hypothalamus in, 734
Skin
in pancreatic secretion, 800-801
male, 1001-1003
blood flow to, 890-891, 890f
in stomach emptying, 786
abnormalities in, 1008-1009, 1009f
cholecalciferol (vitamin D3) formation in,
Secretory vesicles (granules), 13f, 16, 16f, 21,
ejaculation in, 1002-1003
983, 984f
21f
follicle-stimulating hormone and,
cholesterol of, 848
Seizures, 583, 743-745, 743f, 744f
1007
cyanosis of, 276, 428, 510, 531
hippocampal, 736-737
gonadotropin-releasing hormone and,
dermatomes of, 596f, 597
in deep-sea diver, 547
1006
estrogen effects on, 1018
Self-antigen, 441
hormones in, 1003-1008, 1003f, 1004f,
free nerve endings in, 586, 587
Semen, 999-1001
1008f
heat conduction to, 890-891, 890f, 891f
Semicircular canals (ducts), 693, 693f, 694,
inhibin and, 1007
heat loss from, 891-892, 891f
694f
lubrication in, 1002
insulating property of, 890
function of, 695-696, 695f
luteinizing hormone and, 1007
macrophages in, 433
receptors of, 576
neuronal stimulus for, 1001-1002
tactile receptors in, 585-586, 586f
Semilunar valves, 109, 109f. See also Aortic
penile erection in, 1002, 1002f
testosterone effects on, 1005
valve; Pulmonary valve.
pineal gland in, 1009-1001
urticaria of, 450
sounds at, 269, 270
stages of, 1002-1003, 1002f
yellow, 863-864
Seminal fluid, 999
testosterone and, 1003-1006, 1007. See
Sleep, 739-741
Seminal vesicles, 996, 997f, 999
also Testosterone.
alpha waves during, 742, 742f, 743, 743f
Seminiferous tubules, 996, 997f
thyroid hormone effects on, 937-938
beta waves during, 742, 742f, 743, 743f
Sensation. See Sensory receptors; Somatic
Shingles, 605
brain waves during, 740, 741-743, 742f
sensation.
Shivering, 895-896, 896f
delta waves during, 742-743, 742f
1108
Index
Sleep (Continued)
Sodium (Continued)
Sodium (Continued)
electroencephalography during, 741-743,
aldosterone disorders and, 379
carbonic anhydrase inhibitor and, 404
742f, 743f
angiotensin II in, 377-378, 378f
countercurrent mechanism in, 351-352,
metabolic rate and, 887
antidiuretic hormone and, 379
352f
neurotransmitters and, 740-741
arterial pressure and, 374-376, 375f, 379
diuretics and, 402-404, 403f, 403t
passive theory of, 740
glomerular filtration rate and, 374
gradient-time transport in, 331-332
physiologic effects of, 741
in Addison’s disease, 363, 379
in cardiac failure, 264, 380
raphe nuclei in, 740
macula densa feedback and, 374
in cirrhosis, 381
REM, 739, 740, 741
pressure natriuresis and, 374-375, 375f
in nephrotic syndrome, 381
slow-wave, 739-740
sodium intake and, 307-308, 308f,
principal cells in, 336-337, 336f, 337f
theories of, 740-741
373-374, 380
rate of, 374
theta waves during, 742, 742f
sympathetic nervous system in, 377
sodium intake and, 307-308, 308f
thyroid hormone effects on, 937
volume regulation and, 373-374
sympathetic nervous system in, 343
tractus solitarius in, 740
extracellular, 4, 46f, 294f, 294t, 358
water reabsorption and, 332
Sleep apnea, 522-523
aldosterone and, 362-363, 363f, 379
salivary, 794
Sleep spindles, 743
angiotensin II and, 362-363
Sodium bicarbonate. See also Bicarbonate;
Sleep-wakefulness cycle, 741, 743, 743f
antidiuretic hormone and, 358-359,
Bicarbonate buffer system.
Slit pores, of glomerular capillary, 316,
358f, 362-363, 362f, 379
in acidosis, 398
316f
deficiency of, 301-302, 301t, 363
Sodium-calcium channel, of cardiac muscle,
Slow wave rhythm, of smooth muscle
excess of, 301t, 302
105, 117
membrane potential, 97, 97f, 772-773,
in chronic renal failure, 410, 410f
Sodium channel, 47, 47f
772f
normal value for, 7t, 358
all or none nature of, 48-49, 48f
Slow-reacting substance of anaphylaxis, 450,
osmolarity from, 358
calcium deficit and, 65
480, 529
regulation of, 358-363, 358f
neuronal, 562, 566, 566f
Small intestine. See also Gastrointestinal
renal regulation of, 373-374
voltage clamp testing of, 63, 63f
tract.
salt appetite and, 363
voltage-gated, 48, 48f, 62, 62f, 63f, 64, 64f,
absorption in, 813-816, 814f
thirst and, 361-363, 361t, 362f, 363f
66-67
bicarbonate absorption in, 815
glomerulotubular balance of, 374
activation of, 62, 62f, 65
calcium absorption in, 815
glucose co-transport and, 330, 330f
anesthetic effect on, 70
carbohydrate absorption in, 815-816
hydrogen ion counter-transport and, 330,
calcium effect on, 65
carbohydrate digestion in, 809
330f
inactivation of, 62, 62f
chloride absorption in, 815
in bone, 981
Sodium-amino acid co-transport, 55, 816
crypts of Lieberkühn of, 791, 792f,
in cardiac muscle action potential, 105
Sodium channel blockers, 403t, 404
805-806, 805f
in diffusion potential, 57, 58f
Sodium chloride. See also Sodium.
disorders of, 821-822
in end-plate potential, 87, 87f
appetite for, 363
enzymes of, 806
in hemorrhagic shock, 283
exercise-related loss of, 1065
fat absorption in, 816
in hydrochloric acid formation, 796-797,
in seawater, 350
iron absorption in, 815
796f
macula densa concentration of, 324, 324f
magnesium absorption in, 815
in membrane potential, 57, 58f, 59-61, 60f
Sodium chloride wasting, aldosterone
mixing (segmentation) contractions of,
in neural action potential, 61-65, 61f-64f,
deficiency and, 947-948
786-787, 787f
66, 87, 87f
Sodium citrate, 467
nutrient absorption in, 815-816
in neuronal excitation, 564-566, 564f, 566f,
Sodium lactate, in acidosis, 398
phosphate absorption in, 815
567, 567f
Sodium-loading renal function curves, 229,
potassium absorption in, 815
in pancreatic juice secretion, 800, 800f
229f
propulsive movements of, 787-788
in resting membrane potential, 60-61, 60f
Sodium-potassium pump, 53-54, 53f, 59-60,
protein absorption in, 816
in sinus nodal rhythmicity, 117-118
60f
secretions of, 776, 805-806, 805f
aldosterone secretion and, 370-371,
in action potential, 66
slow waves of, 772-773, 772f, 787
371f
in resting membrane potential, 60f, 61
sodium absorption in, 814-815, 814f,
arterial pressure and, 218-219, 218f,
Sodium space, 296
949
220, 224-226, 226f
Sodium-calcium counter-transport, 55
villi of, 812-813, 812f, 813f
integrated responses to, 380-381
Sodium-glucose co-transport, 55, 55f, 816,
blood flow in, 779, 779f
potassium excretion and, 370-371, 371f
831
brush border of, 813, 813f
potassium secretion and, 370-371, 371f
Sodium-hydrogen counter-transport, 55
contractions of, 788
renal function curve for, 229, 229f
Solubility coefficient, of gas, 492
renewal of, 806
renal response to, 307-308, 308f, 356,
Solution(s)
water absorption in, 814
373-374, 380
concentration of, 51-52
Smell sensation, 667-670. See also Olfactory
variability in, 292
glucose, 301
cells.
interstitial, 294t
hyperosmotic, 299
affective nature of, 668
intestinal absorption of, 814-815, 814f, 817
hypertonic, 298f, 299-301
centrifugal signals in, 670
aldosterone effects on, 814-815, 949
hypo-osmotic, 299
gradations of, 668-669
intestinal secretion of, 815
hypotonic, 298, 298f, 299, 300f
olfactory cell stimulation in, 667-669
intracellular, 46f, 294f, 294t
isosmotic, 299
olfactory membrane in, 667, 667f
loop of Henle transport of, 334-336, 335f
isotonic, 298, 298f, 299, 300f
pathways of, 667f, 669-670
Nernst potential for, 565
osmolality of, 52, 297
threshold for, 668
neuronal concentration of, 564f, 565
osmolarity of, 297-298
types of, 668
plasma. See Sodium, extracellular.
osmotic pressure of, 297-298
Sneeze reflex, 480
reabsorption of, 328t, 332f, 333-334, 333f,
Solvent drag, 332
Snoring, 523
334f, 338f, 351t
Soma, 559, 559f
Sodium. See also Sodium chloride.
abnormalities of, 374
Somatic sensation, 585-597
active transport of, 53-54, 53f, 59-60, 60f,
active transport in, 329, 329f
classification of, 585
814, 814f
aldosterone and, 225, 342, 342t,
corticofugal signals in, 597
aldosterone secretion and, 950
378-379, 948
deep, 585
capillary diffusion of, 183-184, 184t
angiotensin II and, 224-225, 225f,
dermatomes in, 597, 598f
co-transport with, 54-55, 55f
342-343, 342t
exteroreceptive, 585
epithelial transport of, 55-56, 56f
antidiuretic hormone and, 343
mechanoreceptive. See Position sensation;
excretion of, 307-308, 308f, 328t
arterial pressure and, 341-342
Tactile sensation.
Index
1109
Somatic sensation (Continued)
Speech, 721-722, 721f. See also Cerebral
Spinal cord (Continued)
nociceptive. See Pain.
cortex; Language.
muscle spindle interaction with,
position. See Position sensation.
Broca’s area in, 687, 687f, 717,
675-679, 676f-678f. See also Muscle
range of, 594
717f
spindles.
receptors for. See Sensory receptors.
dysarthria of, 707
neurons in, 674-675, 674f. See also
sensory pathways for, 587-595
Wernicke’s area in, 716, 718-719, 718f,
Motor neurons.
anterolateral system of, 587, 595-597,
721, 721f
neospinothalamic tract of, 600-601, 600f
596f
Sperm, 997-998, 998f
paleospinothalamic tract of, 600f, 601
dorsal column-medial lemniscal system
abnormalities of, 1001, 1001f
propriospinal fibers of, 675
of, 587, 588-589, 588f, 592-594,
acrosome of, 1000
Renshaw cells of, 675
592f, 593f
capacitation of, 1000
surgical interruption of, for pain
somatosensory cortex in, 589-592,
count of, 1001
treatment, 602
589f-591f
enzymes of, 1000
tract of Lissauer of, 609
spinal cord transection and, 606, 606f
flagellum of, 17, 17f, 25
transection of, 673
stimulus intensity and, 594f
motility of, 1001
body temperature and, 898
tactile. See Tactile sensation.
physiology of, 999
Brown-Séquard syndrome in, 606, 606f
thalamus in, 596
storage of, 999
flexor reflex and, 680, 680f
thermoreceptive. See Cold sensation; Heat
Spermatocytes, 997, 998f
galloping reflex and, 683
sensation.
Spermatogenesis, 996-1001
shock and, 684
visceral, 585
abnormalities of, 1001, 1001f
stepping movements and, 682-683, 683f
Somatomedins, 923-924
follicle-stimulating hormone effect on,
Spinal nerves, 204, 748-749, 749f
Somatosensory area I, 590, 590f
1007
Spinal reflexes, 690-691
functions of, 591-592
hormonal stimulation of, 998-999
autonomic, 683-684
Somatosensory area II, 590, 590f
meiosis in, 997, 998f
crossed extensor, 680f, 681, 681f
Somatosensory association areas, 589f, 591,
regulation of, 1007
depression of, 684
592
sperm formation in, 997-998, 998f,
flexor, 680-681, 680f, 681f
Somatosensory cortex, 589-592
999
galloping, 683
area I of, 590, 590f, 591-592
temperature effects on, 1001
Golgi tendon, 679-680, 679f
area II of, 590, 590f
testosterone effect on, 1007
mark time, 682-683, 683f
body representation of, 590, 590f
Spermatogonia, 996, 997f
mass, 683-684
layers of, 590-591, 591f
Spherocytosis, hereditary, 427
pain (nociceptive), 680-681, 680f, 681f
loss of, 596
Sphincter(s)
postural, 682
vertical columns of, 591
anal, 789
reciprocal relations in, 681-682, 682f
Somatostatin, 907t, 921, 921t, 925-926
esophageal, 782f, 783, 819
righting, 682
glucagon inhibition and, 971
gastroesophageal, 783
scratch, 683
insulin inhibition and, 971
ileocecal, 774, 788, 788f
spasm and, 683
secretion of, 961
pharyngoesophageal, 782, 782f
stepping, 682-683, 683f
Somatotropes, 919, 920t
precapillary, 181-183, 182f, 187, 197-198,
stretch, 676-677, 677f, 678-679, 679f
Somatotropic hormone. See Growth
197f
withdrawal, 680-681, 680f, 681f
hormone.
pyloric, 774, 785
Spinal shock, 684
Somatotropin. See Growth hormone.
urinary, 312, 313
Spine. See also Spinal cord.
Sound
Sphincter of Oddi, 803, 803f
centrifugal acceleratory force effects on,
attenuation of, 652
Sphingomyelin, 68, 846-847, 847f. See also
542
conduction of. See also Hearing.
Phospholipids.
Spinocerebellar pathways, 700-701, 700f
from tympanic membrane to cochlea,
Spinal anesthesia, neurogenic shock and,
Spinocerebellum, 704-705, 704f
651-652, 652f
285
Spiral ganglion of Corti, 652f, 655
through bone, 652
Spinal animal, 673. See also Spinal cord,
Spirometry, 475-477, 475f, 476f, 477t
within cochlea, 594, 654-655, 654f
transection of.
abbreviations for, 476, 477t
direction of, 660
Spinal cord, 558
pulmonary capacities on, 475-476,
frequency of, 654-655, 654f, 656, 657,
alpha motor neurons of, 674
476f
657f
anterior motor neurons of, 674
pulmonary volumes on, 475, 476f
impedance matching of, 651-652
anterolateral pathway of, 587, 595-597,
Spironolactone, 403t, 404
loudness of, 656-657, 657f
596f
Splanchnic circulation, 777-780, 778f
resonance of, 653
corticorubral tract to, 688-689, 689f
Spleen
Space travel
demyelination of, 877
blood-cleansing function of, 180
artificial climates in, 543
dorsal column-medial lemniscal system
lymphocytes in, 440
deconditioning during, 544
of, 587, 588-589, 588f, 592-594, 592f,
macrophages in, 434, 434f
linear acceleratory forces in, 542-543, 543f
593f
reservoir function of, 179-180, 180f
motion sickness during, 544
gamma motor neurons of, 674
reticuloendothelial cells of, 180
weightlessness in, 543-544
gray matter of, 673-675, 674f
structure of, 180f, 434, 434f
Spaces of Disse, 859
injury to. See also Spinal cord, transection
Splenectomy, in thrombocytopenia, 465
Spasm
of.
Sports physiology, 1055-1066. See also
carpopedal, 979, 979f
defecation and, 823
Exercise.
coronary, 252
urination and, 314
Sprue, 423, 427, 822
gastrointestinal, 604
interneurons of, 674-675, 674f
Standing, baroreceptor response to, 210
muscle, 683
lateral motor system of, 687-689, 688f,
Stapedius muscle, 652
drug-induced, 89
689f, 692, 692f
Stapes, 651, 652f
headache and, 607
medial motor system of, 692, 692f,
ankylosis of, 661
motor cortex lesion and, 691
696-697, 696f
Staphylococcal infection, 434
pain and, 599
motor neurons of, 690, 690f
Starches, 809. See also Carbohydrates.
sigmoidal, 822
motor system of, 673-675, 674f, 712. See
Starling equilibrium, 189-190
vascular, 457
also Spinal reflexes.
Starvation, 874-875, 874f. See also Feeding;
Spatial summation
experimental study of, 673, 682-683,
Food.
in nerve fibers, 578, 578f
683f
hypothalamic lesions and, 735
in neurons, 567-568, 567f
Golgi tendon organ interaction with,
ketosis in, 844
in thermal sensation, 609
679-680, 679f
protein effects of, 857
1110
Index
Statins, 851
Stress response, 757, 758
Swallowing center, 783
Statoconia, 693, 693f
Stress-relaxation, smooth muscle, 94-95, 172,
Sweat, evaporation of, 892
Stearic acid, 840. See also Triglycerides.
173f
Sweating, 292, 292t, 895, 895f
Steatorrhea, 822, 991
in hemorrhagic shock, 281
absence of, 892
Stellate cells, 702
Stretch
acclimatization of, 893-894
Stem cells, 420-422, 421f, 430, 440
of right atrium, 112
aldosterone effects on, 893, 949
Stepping movements, 682-683, 683f
of sinus node, 212, 233
autonomic nervous system control of, 753,
Stercobilin, 817
of smooth muscle, 97
754t, 892-894, 893f
Stereocilia, 655, 693-694, 693f
of viscus, 604
mechanism of, 892-893, 893f
Stereopsis, 622, 622f, 643
Stretch receptors. See Baroreceptor(s).
Sympathectomy, 756-757, 756f
neuronal mechanism of, 647-648
Stretch reflex, 676-677, 677f
Sympathetic nervous system. See also
Sterility, female, 1024-1025
clinical application of, 678-679, 678f
Parasympathetic nervous system.
Steroids, adrenocortical, 944-960. See also
damping mechanism of, 677, 677f
alarm response of, 578
Aldosterone; Cortisol.
dynamic, 677
anatomy of, 204, 205f, 748-750, 749f
hepatic metabolism of, 947
neuronal circuitry of, 677, 677f
coronary blood flow and, 251
synthesis of, 945-947, 946f, 947t
static, 677
denervation effects on, 756-757, 756f
Stimulus. See Sensory stimulus.
Stria vascularis, 656
functions of, 753-755, 754t
Stokes-Adams syndrome, 121, 149-150
Stroke, 212-213, 220-221, 691, 763. See also
in adrenal medullae function, 749-750,
Stomach. See also at Gastric;
Cerebral blood flow.
755
Gastrointestinal tract.
Stroke volume, 108
in arterial function, 170, 170f, 172, 173f,
anatomy of, 784, 784f
during exercise, 1063-1064, 1064f, 1064t
754t, 755
antrum of, 784, 784f
pulse pressure and, 173
in blood flow regulation, 170, 170f,
atrophy of, 820
Stroke work output curve, 112, 112f
204-205, 205f, 206-208, 206f, 207f
body of, 784, 784f
Strychnine, synaptic transmission and, 570
in bronchiolar function, 479
carbohydrate digestion in, 809, 809f
Stuart factor (factor X), 459t, 461f, 462, 462f,
in cardiac failure, 258-259, 259f, 261, 261f,
chief cells of, 795-796, 797
463
265
constrictor waves of, 784
Stumble reflex, 682
in cardiac function, 112-113, 113f, 121,
disorders of, 819-821
Subarachnoid space, 764
205, 205f, 234-235, 242, 242f, 754, 754t
emptying of, 785-786
Subcallosal gyri, ablation of, 738
in cardiac output, 234-235, 242, 242f
enterogastric inhibition of, 785-786
Subconscious, 558
in cerebral ischemia, 212-213
fat digestion in, 811
Subcutaneous tissue
in exercise, 247, 248-249, 248f
food mixing in, 784
insulating property of, 890
in extracellular volume control, 377
food volume in, 785
interstitial fluid pressure in, 187-188
in gastrointestinal blood flow, 779-780
gastrin secretion by, 776, 785, 797-798
Sublingual glands, 793-795
in gastrointestinal function, 775, 792
hormonal inhibition of, 786
Submandibular glands, 792f, 793-795,
in glomerular filtration, 321-3322
hunger contractions of, 785
795f
in glycogenolysis, 832
hydrochloric acid secretion by, 54,
Submarines, 550
in heat conduction, 891, 895-896
796-797, 796f, 810
Submucosal (Meissner’s) plexus, 774-775,
in hemorrhage, 172
absence of, 820
774f
in hemorrhagic shock, 279-280, 279f, 281
pH of, 384, 384t
Subneural cleft, 85, 86f
in hypertension, 228
mixing function of, 784
Substance P, in pain sensation, 601
in kidney function, 377
mixing waves of, 784
Substantia gelatinosa, 600f, 601
in nonshivering thermogenesis, 887-888
motor functions of, 784-786
Substantia nigra, 730-731, 730f
in ocular function, 753, 754t
mucosa of, 820
lesions of, 709, 711
in renal sodium reabsorption, 343
mucous neck cells of, 795, 796f, 797
Subthalamic surgery, in Parkinson’s disease,
in respiration, 479
oxyntic glands of, 795-797, 796f
711
in salivary secretion, 795
pain in, 604, 605f
Subthalamus, lesions of, 709
in skeletal muscle blood flow, 247
parietal cells of, 54, 796-797, 796f
Suckling, 733f, 734
in sweating, 753, 892-894, 893f
propulsive function of, 784
Sucrase, 806, 809, 809f
in venous function, 172, 173f
protein digestion in, 810
Sucrose, 809. See also Carbohydrates.
mass discharge of, 757, 758
receptive relaxation of, 783
Sulfate, 46f, 294t
mean circulatory filling pressure and,
secretions of, 795-799, 795f, 796f, 798f
Sulfonylurea drugs, 967-968
239-240, 239f
inhibition of, 798-799
Summation
norepinephrine secretion by, 751
interdigestive, 799
in dendrites, 569
of eye, 648-649, 648f
phases of, 798, 798f
in nerve fibers, 578, 578f
postganglionic neurons of, 749, 749f
slow waves of, 772-773, 772f, 784
in neurons, 567-568, 567f
preganglionic neurons of, 748-749,
storage function of, 784
in thermal sensation, 609
749f
surface mucous cells of, 797
Sunburn, 605
stress response of, 757, 758
triglyceride digestion in, 811
Superior colliculi, 647
supersensitivity of, 756-757, 756f
tubular glands of, 791, 795f
Superoxide, 432
tone of, 756-757, 756f
ulcers of, 820-821, 820f
Supplementary motor area, 686
vasoconstrictor nerve fibers of, 206
Stones
Supraoptic nuclei, antidiuretic hormone
vasoconstrictor tone of, 206, 207f
cholesterol, 804-805, 805f
synthesis in, 359, 359f
vasomotor center of, 205f, 206, 206f
kidney, 991
Surfactant, 474
Sympathomimetic drugs, 287, 759
Strabismus, 648
deficiency of, 529, 1045
Synapse(s), 555, 556f, 559-569. See also
Streptococcal infection
Suspensory ligaments, of lens, 617-618,
Neuromuscular junction; Neuron(s).
inflammation in, 434
617f
acidosis effect on, 570
rheumatic fever and, 448
Sustentacular cells, 664-665, 665f, 667,
afterdischarge of, 581
Streptokinase, in cardiogenic shock,
667f
alkalosis effect on, 570
263
Swallowing, 782-784, 782f
anatomy of, 559-562, 559f, 560f
Stress
disorders of, 819
chemical, 559, 562-564, 562t, 563t. See also
ACTH secretion and, 955
esophageal stage of, 783-784
specific neurotransmitters.
bone, 983
pharyngeal stage of, 782-783
delay at, 570
cortisol secretion and, 952-954
respiration and, 783
downregulation at, 583-584
fat utilization during, 846
voluntary stage of, 782, 782f
drug effect on, 570
Index
1111
Synapse(s) (Continued)
Tactile sensation (Continued)
Temperature (Continued)
electrical, 559
Ruffini’s end-organ in, 573f, 586, 595
set-point in, 896-897, 897f, 899, 899f
facilitation of, 557, 723, 724, 725f
signal transmission in
skin temperature and, 889, 897, 897f
fatigue of, 570, 583-584, 583f
anterolateral pathway in, 595-596, 596f
spinal cord transection and, 898
hypoxia effect on, 570
dorsal column-medial lemniscal
sweating and, 892-894, 893f
neurotransmitters at, 562-564, 562t, 563t.
pathway in, 588-589, 588f, 592-594,
sympathetic nervous system and, 891
See also specific neurotransmitters.
593f
water effect on, 892
one-way conduction at, 559
peripheral nerve fibers in, 586-587
wind effect on, 892
signal transmission by, 557, 570
two-point, 592-593, 593f
sensation of, 607-609, 608f. See also
acidosis and, 570
Tank decompression, 549
Cold sensation; Heat sensation.
alkalosis and, 570
Tank respirator, 532, 532f
pain threshold for, 599, 599f, 608, 608f
delay of, 570
Taste aversion, 666
spermatogenesis and, 1001
drugs and, 570
Taste blindness, 664
vapor pressure and, 492
fatigue of, 570
Taste buds, 665-666, 666f
Temporal cortex, anterior, ablation of,
hypoxia and, 570
stimulus specificity of, 665
738
in memory formation, 723, 724-725,
Taste cells, 664-665, 665f
Temporal summation
724f
Taste pore, 665, 665f
in nerve fibers, 578, 578f
ion channels in, 561, 561f
Taste preference, 666
in neurons, 568
neuronal excitation in, 564-566, 564f,
Taste sensation, 663-666
Tensor tympani muscle, 651, 652
566f
adaptation of, 666
Teratoma, 1009
neuronal inhibition in, 566-568, 566f
bitter, 664, 664t
Terminal cisternae, 90f
neurotransmitters in, 562-564, 562t,
blindness for, 664
Testis (testes), 906f, 907t, 996, 997f. See also
563t
neuronal pathways of, 665-666, 666f
Reproduction, male.
one-way, 559
salty, 663, 664t
descent of, 1005
postsynaptic function in, 560-562, 561f
sour, 663, 664t
fetal, 1032
presynaptic terminals in, 560, 560f
sweet, 663-664, 664t
nondescent of, 1001
second messenger systems in, 561-562,
threshold for, 664, 664t
tumors of, 1009
561f
umami, 664
Testosterone, 907t, 1003-1008
summation in, 567-568, 567f
Tectorial membrane, 655, 655f
after puberty, 1005-1006
upregulation at, 583-584
Teeth, 992-994
bone effects of, 1005-1006
Synaptic cleft, 560, 560f
caries of, 994
chemistry of, 1003, 1004f
Synaptic delay, 570
cementum of, 992-993, 992f
degradation of, 1004
Synaptic fatigue, 583-584, 583f
dentin of, 992, 992f
electrolyte effects of, 1006
Synaptic gutter (trough), 85, 86f
enamel of, 992, 992f
excretion of, 1004
Synaptic knobs, 560
eruption of, 993
fetal, 1004-1005, 1007
Synaptic space (cleft), 85, 86f, 560, 560f
formation of, 993, 993f
functions of, 1004-1006, 1004f
Synaptic vesicles, 85, 86f, 560, 560f
functional parts of, 992-993, 992f
hair distribution and, 1005
Syncope, vasovagal, 208
malocclusion of, 994
in spermatogenesis, 998-999
Syncytium
mineral exchange in, 993-994
in testicular descent, 1005
cardiac, 103-104, 104f
pulp cavity of, 992f, 993
intracellular action of, 1006
gastrointestinal, 771-772
Telophase, 37f, 39
luteinizing hormone regulation of,
Synovial fluid, 292
Temperature. See also at Cold; Heat.
1007
Systole, 106-107, 107f, 108, 110-111, 110f
abnormalities of, 898-900, 898f
metabolic rate and, 886, 1006
coronary blood flow during, 250, 250f
after ovulation, 1025, 1025f
metabolism of, 1003-1004
Systolic stretch, 253-254, 253f, 254
behavioral control of, 892, 897-898
muscle development and, 1005
blood flow and, 890-891, 890f, 891f
protein synthesis and, 1005
cardiac function and, 114
protein tissue deposition and, 857
clothing and, 892
red blood cell effects of, 1006
T
core, 889, 890f
regulation of, 1007
T (transverse) tubules
decreasing mechanisms of, 895, 895f
secretion of, 1003, 1003f
of cardiac muscle, 106
evaporation and, 892
skin effects of, 1005
of skeletal muscles, 89-90, 90f
fat insulator system and, 890
structure of, 908f
T wave, 107, 107f, 123, 124-125, 124f, 125f
feedback gain in, 896-897
voice and, 1005
abnormalities of, 145-146, 146f
heart rate and, 147, 148f
water balance and, 1006
atrial, 125
heat conduction and, 891, 891f
Tetanization, 81, 81f
vectorial analysis of, 136, 136f
heat convection and, 891, 891f
Tetany, 7, 979-980, 979f
in premature ventricular contractions,
heat loss and, 890-894, 890f, 891f
blood flow and, 246
151, 151f
heat production and, 889-890
hypocalcemia and, 371
vectorial analysis of, 134-136, 136f
heat radiation and, 891, 891f
in rickets, 991
Tabes dorsalis, 314
high, 898-900, 898f
Tetracaine, 70
Tachycardia
hypothalamic regulation of, 733-734,
Tetraethyl ammonium ion, 760
electrocardiogram in, 147, 148f
733f, 894-898, 894f, 895f, 897f
Tetralogy of Fallot, 139, 275-276, 275f
paroxysmal, 151-152
in premature infant, 1050-1051
Thalamocortical system, 714-715, 715f
atrial, 152, 152f
increasing mechanisms of, 895-896, 895f
Thalamus, 558
ventricular, 152, 152f
local effects on, 898
anatomy of, 714-715, 715f
Tactile sensation, 585-587
low, 898f, 900
cauterization of, 602
free nerve endings in, 573f, 586
neonatal, 1048-1049, 1049f
cerebral cortex relationship to, 729, 729f
hair end-organ in, 586
neuronal regulation of, 895-896, 895f
dorsal lateral geniculate nucleus of,
Iggo dome receptors in, 586, 586f
normal, 7t, 889, 890f
640-641, 641f
in Brown-Séquard syndrome, 606
oxygen-hemoglobin dissociation curve
electrical stimulation of, 603
Meissner’s corpuscles in, 573f, 586, 699
effects of, 508, 508f
in motor function, 588
Merkel’s discs in, 586, 586f
physics of, 891-892, 891f
in pain sensation, 600-601, 600f
Pacinian corpuscles in, 573f-575f,
regulation of, 894-898, 894f, 895f, 897f
in somatic sensation, 596
574-575, 576, 587, 693-694
hypothalamus in, 733-734, 733f,
neospinothalamic tract to, 600-601, 600f
receptors for, 572, 573f, 585-586, 586f
894-898, 894f, 895f, 897f
paleospinothalamic tract to, 600f, 601
1112
Index
Theobromine, synaptic transmission and,
Thyroid-stimulating hormone (Continued)
Transcellular fluids, 293. See also
570
measurement of, 941
Cerebrospinal fluid (CSF).
Theophylline, synaptic transmission and,
regulation of, 938-939, 939f
Transcortin, 947
570
Thyroid-stimulating immunoglobulin, 940
Transcription, 30-35, 32f, 32t, 34f
Thermal receptors, 572, 573t, 607-609, 608f,
Thyrotoxicosis, 940
growth hormone effects on, 922
894-895. See also Temperature, body.
Thyrotropes, 919, 920t
partial, 36
adaptation of, 608
Thyrotropin. See Thyroid-stimulating
Transferrin, 425, 425f, 426
distribution of, 607-608
hormone.
Transforming growth factor-ß, 861
stimulation of, 608, 608f, 609
Thyrotropin-releasing hormone, 907t, 921,
Transfusion reactions, 454-455
Thermal sensation, 572, 573t, 607-609, 608f
921t
agglutination in, 452-453
tissue damage and, 599, 599f
cold effects on, 896, 938-939
hemolysis in, 453
transmission of, 609
Thyroxine, 879, 907t
Translation, 33-35, 34f. See also Protein(s),
Thermode, 894
arterial pressure and, 937
synthesis of.
Thermogenesis
basal metabolic rate and, 886, 936,
Transmitters. See Neurotransmitters.
chemical, 896
936f
Transplantation, 455-456
nonshivering, 887-888
body weight and, 936-937
rejection prevention in, 456
shivering, 895-896
carbohydrate metabolism and, 936
tissue typing for, 455-456
Theta brain waves, 742, 742f
cardiac output and, 937
Transport
Thiamine. See Vitamin B1 (thiamine).
cardiovascular effects of, 935f, 937
acetoacetic acid, 844
Thiazide diuretics, 403t, 404
cellular active transport and, 935
active. See Active transport.
Thinking, 717, 723-727. See also Cerebral
cholesterol levels and, 848, 936
carbon dioxide. See Carbon dioxide,
cortex; Memory.
excitatory effects of, 937
transport of.
corpus callosum in, 722
fat metabolism and, 846, 936
glucose, 55, 55f, 816, 831, 964, 964f
definition of, 723
formation of, 931-933, 932f
hormone, 909, 1016
holistic theory of, 723
gastrointestinal motility and, 937
iron, 425-426, 425f
prefrontal association area in, 717,
growth effects of, 935f, 936
lipid, 840-842
719-720
heart rate and, 937
oxygen. See Oxygen, transport of.
Wernicke’s area in, 718-719, 718f
in chemical thermogenesis, 896
passive. See Diffusion.
Thiocyanate, 794, 939
in fat utilization, 846
Transport vesicles, 21, 21f
Third ventricle, 763, 764, 764f
in pregnancy, 1034
Transpulmonary pressure, 472f, 473
Thirst, 361-363
measurement of, 941
Transverse tubules
central nervous system centers for, 359f,
metabolic effects of, 934-935, 935f,
of cardiac muscle, 106
361
936-937, 936f
of skeletal muscles, 89-90, 90f
osmoreceptor integration with, 362-363,
mitochondrial effects of, 935
Trauma, shock and, 285
362f
muscle effects of, 937
Tremor
stimuli for, 361-362, 361t
Na,K-ATPase effects of, 935
action (intention), 705, 707
Thirst center, 361
period of action of, 934, 934f
muscle, 937
Thoracic duct, 190, 191f, 192
protein metabolism and, 857, 915
Parkinson’s, 711
Thorax, compliance of, 474-475
protein synthesis and, 934, 935f
Treppe, 81-82
Threonine, 853f
receptor for, 934, 935f
Triamterene, 403t, 404
Thrill, in aortic stenosis, 272
respiration and, 937
Tricarboxylic acid cycle, 23, 833-835, 834f,
Thrombin, 459-460, 459f, 461
secretion of, 933-934
843
Thrombocytes. See Platelet(s).
sexual function and, 937-938
Trichinosis, 436
Thrombocytopenia, 465
sleep effects of, 937
Tricuspid valve, 109
Thrombomodulin, 463
therapeutic, 942
pressure measurement at, 179, 179f
Thrombosthenin, 457
tissue transport of, 934
Tricyclic antidepressants, 745
Thromboxane A2, 457, 458
vitamin requirements and, 936
Trigeminal neuralgia, 605-606
Thrombus, 252, 465-466
Thyroxine-binding globulin, 908, 934
Triglycerides. See also Lipid(s);
Thymine, 27, 28f, 29, 29f
Tic douloureux, 605-606
cellular, 12, 842
Thymus gland, 906f
Tickling, 587
digestion of, 811-812, 811f, 812f
T lymphocyte preprocessing in, 440, 441,
Tidal volume, 475, 476f
energy release from, 842-844, 843f,
448
Tissue factor (factor III), 459t, 461, 461f
846
Thyroglobulin, 908, 931
Tissue gel, 185
formation of, 842, 844-845, 845f
formation of, 932, 932f
Tissue plasminogen activator, 464, 466
hydrolysis of, 841, 842
organification of, 933, 933f
Tissue-type plasminogen activator, 464
lipoprotein transport of, 841-842
storage of, 933
in cardiogenic shock, 263
storage of, 842. See also Adipose tissue.
Thyroid cartilage, 481, 481f
in thrombus, 466
structure of, 840
Thyroid gland, 906f, 907t
Titin, 73-74
transport of, 840-842
adenoma of, 940
Tolerance, immune, 448
Trigone, 311, 311f
anatomy of, 931, 932f
Tongue
Triiodothyronine, 879, 907t
calcitonin secretion by, 988-989
black, 876
arterial pressure and, 937
cells of, 988
taste buds of, 664-665, 665f. See also Taste
basal metabolic rate and, 936, 936f
cold effects on, 896
sensation.
body weight and, 936-937
disorders of, 940-942, 940f, 942f
Tonic-clonic seizures, 743-744, 743f, 744f
carbohydrate metabolism and, 936
drug suppression of, 939-940
Tonometry, 624, 624f
cardiac output and, 937
hormones of. See Thyroxine;
Total lung capacity, 476, 476f
cardiovascular effects of, 935f, 937
Triiodothyronine.
Touch sensation. See Tactile sensation.
cellular active transport and, 935
neonatal, 1050
Toxemia of pregnancy, 227-228, 1035-1036
cholesterol levels and, 936
radioactive iodine destruction of, 941
Toxins, renal, 307, 406
excitatory effects of, 937
surgical removal of, 941
Trace elements, 878t, 879, 879t
fat metabolism and, 936
Thyroid hormones. See Thyroxine;
Trachea, 478-479, 479f
formation of, 932-933, 932f
Triiodothyronine.
Tractus solitarius, 666, 666f
gastrointestinal motility and, 937
Thyroid-stimulating hormone, 896, 907t, 918,
in sleep, 740
growth effects of, 935f, 936
919f, 920t, 938
in swallowing, 783
heart rate and, 937
in pregnancy, 1034
Tranquilizers, 736
metabolic effects of, 934-935, 935f,
in thermogenesis, 896
Transamination, 856, 856f, 862
936-937, 936f
Index
1113
Triiodothyronine (Continued)
Urinary tract obstruction, renal failure and,
Urine (Continued)
metabolic rate and, 886
406
water deficiency and, 350-357, 351t,
mitochondrial effects of, 935
Urination, 313-314, 313f
352f-356f
muscle effects of, 937
abnormalities of, 313-314
water excess and, 348-350, 349f
period of action of, 934
voluntary, 313
pH of, 384, 384t, 392
protein metabolism and, 857
Urine. See also Bladder; Kidneys; Renal
phosphate in, 372-373
protein synthesis and, 915, 934, 935f
tubules.
potassium in, 367
receptor for, 934
amino acids in, 413
protein in, 409
respiration and, 937
bicarbonate in, 394
titratable acid of, 394-395
secretion of, 933-934
calcium in, 372
transport of, 312-313, 313f
sexual function and, 937-938
concentration of, 350-357, 351t, 352f,
volume of, 292, 292t, 327, 328t, 350
sleep effects of, 937
353f-356f
Urobilin, 817, 863, 863f
thyroxine conversion to, 934
angiotensin II in, 377-378
Urobilinogen, 863, 863f, 864
tissue transport of, 934
antidiuretic hormone in, 348-349,
Urogenital diaphragm, 312
vitamin requirements and, 936
350-351, 356
Urticaria, 450
Trophoblasts, 1029, 1029f
arterial pressure and, 374-375, 375f
Uterus, 1011, 1012f
Tropical sprue, 822
cortical collecting tubule in, 353, 353f,
contractions of, 8-9, 733f, 734, 1036-1038
Tropomyosin, 76
356, 356f
estrogen effects on, 1017
Troponin, 76
countercurrent mechanisms in, 351-352,
involution of, 1038
Trypsin, 799-800, 810, 810f
352f, 357-358
oxytocin effects on, 1036
Trypsin inhibitor, 800
disorders of, 357-358
progesterone effects on, 1018
Trypsinogen, 799-800
distal tubule in, 352-353, 353f, 356,
stretch of, 1036
Tryptophan, 853f
356f
Utilization coefficient, of oxygen, 507, 508
Tuberculosis, 432, 530
diuretic effects on, 403
Utricle, 693, 693f
D-Tubocurarine, neuromuscular junction
in chronic renal failure, 411, 411f
macula of, 693, 693f, 695
effects of, 89
loop of Henle in, 351-352, 352f, 355,
Uvulopalatopharyngoplasty, 523
Tubular glands, 791, 795-797, 795f, 796f
356f
Tubulin, 17
medullary blood flow and, 354-355,
Tufted cells, of olfactory bulb, 669, 670
355f
Tumor necrosis factor, in inflammation, 435,
medullary collecting duct in, 353, 353f,
V
436f
356-357, 356f
V wave, 107f, 108
Two-point discrimination, 592-593, 593f
obligatory urine volume and, 350
Vagal reflex, in paroxysmal tachycardia, 152
Tympanic membrane, 651-652, 652f
osmolarity in, 350-352, 352f
Vagina, 1011, 1012f
Tyramine, 759
quantification of, 357
estrogen effects on, 1017
Tyrosine, 853f
summary of, 355-357, 356f
Vagus nerve, 750, 750f. See also
iodination of, 933, 933f
urea in, 351t, 353-354, 354f
Parasympathetic nervous system.
vasodilator effect on, 355
in cardiac function, 113, 113f, 121, 148,
volume regulation and, 373-374
152, 205, 205f
dilution of, 348-350, 349f, 356
Valine, 853f
U
after excess water ingestion, 349-350,
Valves
Ulcerative colitis, 823
349f
heart. See Heart valves.
Ulcers
collecting tubules in, 349f, 350
lymph, 191, 192-193, 192f, 193f
intestinal, 823
distal tubule in, 349f, 350
venous, 178-179, 178f
marginal, 820
diuretic effects on, 403
Valvulae conniventes (folds of Kerckring),
peptic, 820-821, 820f
in chronic renal failure, 411, 411f
812-813, 812f
Ultimobranchial glands, 988
loop of Henle in, 349-350, 349f
Van den Bergh reaction, 864
Ultrasound Doppler flowmeter, 164-165,
quantification of, 357
Vapor pressure, 492
165f
diuretic effect on, 402-404, 403f, 403t
Varicosities
Ultraviolet light, headache and, 607
elimination of. See Micturition; Urination.
axonal, 96, 96f
Unconsciousness, 767
formation of, 292, 314-325, 314f, 315f. See
venous, 178-179, 381
Uracil, 30
also Urine, concentration of; Urine,
Vas deferens, 996, 997f
Urate, renal secretion of, 334
dilution of.
Vasa recta, 310, 311f
Urea
antidiuretic hormone in, 348-349, 349f
blood flow in, 321, 354-355, 355f
capillary diffusion of, 183-184, 184t
arterial pressure and, 216, 323, 341-342,
countercurrent exchange in, 354-355, 355f
excretion of, 307, 328t, 353-354
374-376, 375f, 376f, 948-949, 948f
Vascular compliance, 171-172
extracellular, 294f
diuretic effect on, 402-404, 403f, 403t
delayed (stress-relaxation), 94-95, 172,
formation of, 856, 862
during pregnancy, 1035
173f
in urine concentration, 351t, 353-354, 354f
glomerular filtration in, 316-323,
in hemorrhagic shock, 281
interstitial, 294t
316f-320f, 320t, 323f, 323t, 342f. See
reverse, in hemorrhagic shock, 281
intracellular, 294t
also Glomerular filtration;
pulse pressure and, 173, 174
plasma, 294f
Glomerular filtration rate (GFR).
Vascular endothelial growth factor, 200-201
in chronic renal failure, 410, 410f, 411f,
hypothalamus in, 734
Vascular resistance. See also Blood flow;
412
obligatory volume for, 350, 356-357
Circulation (blood).
reabsorption of, 328t, 332-333, 332f, 338f,
renal blood flow in, 320-323, 321f, 321t,
arteriolar, 168
351t, 353-354, 354f
323f. See also Kidneys, blood flow
blood flow and, 164, 164f, 167-169,
Urea transporter AI, 353
of.
168f-170f
Uremia, 411-412, 411f
tubular processing in, 314-315, 314f,
blood pressure and, 170, 170f, 219, 219f,
Ureterorenal reflex, 312
315f. See also at Renal tubules.
220, 220f
Ureters, 312, 312f
water deficiency and, 350-357, 351t,
blood viscosity and, 169-170, 170f
Urethra, 996
352f, 353f-356f, 379
CGS units for, 167
posterior, 311, 311f, 312
water excess and, 348-350, 349f, 379
conductance and, 167-168, 168f, 169
Uric acid
glucose in, 413, 975
fourth power law and, 168
excretion of, 307
keto acids in, 976
hematocrit and, 169-170, 169f, 170f
extracellular, 294f
obligatory volume of, 350, 356-357
in parallel circuits, 168-169, 169f
in irreversible shock, 284
osmolarity of, 348
in series circuits, 168-169, 169f
Uridine diphosphate glucose, 832, 832f
free-water clearance and, 357
intrarenal, 219, 219f
1114
Index
Vascular resistance (Continued)
Vein(s) (Continued)
Ventricle(s) (Continued)
peripheral, 167, 170, 170f, 219, 219f, 220,
distensibility of, 171, 176-179, 177f-179f
function of, 107f, 108, 110-111, 110f, 121
220f
functions of, 161, 176-180, 177f, 178f
hypertrophy of, 272-273
Poiseuille’s law and, 168
neck, 179
axis deviation with, 138, 138f
pressure pulse damping and, 175
obstruction of, cardiac output and, 237
in aortic valve lesions, 272-273
pulmonary, 167
pressures in, 176-180, 177f-179f
pressures in, 107f, 108, 110-111, 110f
total, 167
reservoir function of, 171, 179-180
stroke work output curve of, 112,
units of, 167
sympathetic nervous system effects on,
112f
venous, 176-177, 177f, 240-241, 240f
172, 173f, 204, 205f
volume output curve of, 112, 112f
vessel diameter and, 167-168, 168, 168f
valves of, 178, 178f
work output of, 110-111, 110f
Vasoactive intestinal polypeptide, 774
varicose, 178-179, 381
mean electrical axis of, 137-140,
Vasoconstriction. See also Vasodilation.
volume-pressure curves of, 172, 172f
137f-140f
adrenal medullae in, 207
Venae cavae, cross-sectional area of, 162
coronary blood flow in, 250
angiotensin II in, 201-202, 223f, 224, 224f
Venous pressure, 176-180
hypertrophy of, axis deviation with,
calcium in, 202
central, 176
138, 138f
cerebral cortex in, 207
gravitational pressure and, 177-178, 177f
in mitral valve lesions, 273
cerebral ischemia and, 212-213
intra-abdominal pressure and, 177
pressures in, 483-484, 484f
endothelin in, 202
measurement of, 179, 179f
stroke work output curve of, 112,
gastrointestinal, 779-780
peripheral, 176-177, 177f
112f
hypothalamus in, 207
pulmonary, 484
volume output curve of, 112, 112f
in cerebral ischemia, 212-213
resistance and, 176-177, 177f
work output of, 110
in exercise, 247
right atrial pressure and, 177, 179, 179f
Ventricular escape, 121, 149-150
in heating, 895
Venous pump, 176, 178-179, 178f. See also
Ventricular fibrillation, 152-155, 287-288
in vessel trauma, 457
Venous return.
after myocardial infarction, 254
norepinephrine in, 207, 247
Venous resistance, 176-177, 177f, 240-241,
cardiopulmonary resuscitation in, 155
sympathetic nervous system in, 170, 170f,
240f
chain reaction mechanism of, 153-154,
204-205, 205f, 206-208, 206f, 207f,
Venous return, 111, 232-236, 234f, 235f. See
153f
754-755, 754t
also Venous return curve.
circus movements in, 153, 153f, 154
vasopressin in, 202
artificial respirator and, 532
60-cycle alternating current in, 153-154,
Vasoconstrictor tone, 206, 207f
calculation of, 240
153f
Vasodilation. See also Vasoconstriction.
decrease in, 236f, 237
electrocardiogram in, 154, 154f
anions in, 203
definition of, 232
electroshock defibrillation in, 154-155,
baroreceptors in, 210, 210f
in cardiac failure, 259
155f
bradykinin in, 202, 779
local blood flow and, 233-234
impulse division in, 153f, 154
carbon dioxide in, 203
mean circulatory filling pressure and,
re-entry in, 153, 153f
cardiac output and, 237
239-240, 239f
Ventricular function curves, 112, 112f
cold-induced, 895, 900
mean systemic filling pressure and, 238,
Ventricular paroxysmal tachycardia, 152,
during exercise, 247
239, 240, 240f
152f
emotion-related, 208
pressure gradient for, 240-241, 241f
Ventricular volume output curve, 112,
histamine in, 202
resistance to, 238, 240-241, 241f
112f
hypothalamus in, 206f, 207-208
right atrial pressure and, 238, 239,
Ventrobasal complex, 600
in thiamine deficiency, 876
241-243, 242f, 243f
Venule(s)
magnesium in, 202
vasodilation-induced reduction in, 285
blood distribution to, 162f
potassium in, 202
Venous return curve, 238-243, 239f-243f
cross-sectional area of, 162
sympathetic nervous system in, 170, 170f,
during exercise, 248f, 249
function of, 161
206f, 207-208, 754-755, 754t
in cardiac failure, 265-267, 265f, 266f
sympathetic innervation of, 204, 205f
Vasodilators
mean systemic filling pressure and, 238,
Vermis, 699, 699f, 704
in angina pectoris, 256
239, 239f, 240, 240f
topographic representation in, 699-700,
in hypertension, 229-230
normal, 238-239, 239f, 241, 242f, 265,
699f
urine concentration and, 355
265f
Vesicointestinal reflex, 790
Vasomotion, 182-183, 187, 197-198, 197f
plateau in, 239, 239f
Vesicoureteral reflux, 312, 409
Vasomotor center
right atrial pressure and, 238-239, 239f
Vestibular apparatus, 692-694, 693f
anesthesia effect on, 285
venous resistance and, 241, 241f
in angular acceleration detection,
baroreceptor effect on, 6-7, 210, 210f
Venous thrombosis, 466
695-696, 695f
chemoreceptor effect on, 212
Ventilation. See Alveolar ventilation;
in linear acceleration detection, 695
control of, 206f, 207
Pulmonary ventilation; Respiration.
in vomiting, 824
in arterial pressure control, 6-7, 212-213
Ventilation-perfusion ratio, 498, 499-501,
saccule of, 693, 693f, 694-695
in hemorrhagic shock, 281
500f
semicircular canals of, 694, 694f, 695-696,
Vasomotor tone, sudden loss of, 285
abnormalities of, 501
695f
Vasomotor waves, 214, 214f
above normal, 500-501
utricle of, 693, 693f, 694-695
Vasopressin. See Antidiuretic hormone
below normal, 500
Vestibular nerve, 694, 694f
(ADH).
during exercise, 501
Vestibular nuclei, 688, 691-692, 691f,
Vasovagal syncope, 208
in chronic obstructive lung disease, 501
696-697, 696f
Vater, papilla of, gallstone of, 821-822
in emphysema, 527
Vestibulocerebellum, 703-704
Vector, 131, 132f
in pneumonia, 528, 528f
Vibration sensation, 585-587
direction of, 131-132, 132f
normal, 500, 500f
transmission of, 588-589, 588f, 593
Vectorcardiogram, 136-137, 137f
Ventral spinocerebellar tract, 700-701, 700f
Villi
Vectorial analysis, 131-146. See also
Ventricle(s)
arachnoidal, 765
Electrocardiogram, vectorial analysis
brain, 763, 764, 764f
absorptive surface of, 812-813, 812f,
of.
cardiac, 104
813f
Vein(s)
action potential transmission in, 117f,
blood flow in, 779, 779f
arm, 178-179
119-120, 120f
brush border of, 813, 813f
blood distribution to, 162f, 254
current flow in, 126-127, 126f, 127f
contractions of, 788
collateral, 201
emptying of, 107f, 108
renewal of, 806
compliance of, 171-172
filling of, 107f, 108
Viruses, 18, 18f
cross-sectional area of, 162
function curves of, 112, 112f
in cancer, 41
Index
1115
Visceral pain, 603-605, 605f
Vitamin(s) (Continued)
Warfarin, 466
localization of, 604, 605f
requirements for, 875, 875t
Warmth receptors, 607-609, 608f, 894-895.
transmission of, 604-605
thyroid hormone effects on, 936
See also Heat sensation.
Visceral reflexes, 748
storage of, 862, 875
Wasting syndrome, 874
Vision, 613-625. See also Eye(s); Retina.
Vitamin A (retinol), 875, 875t
Water
acuity of, 621, 621f
deficiency of, 629, 875
carbon dioxide reaction with, 510-511
astigmatic, 620, 620f
hepatic storage of, 862
cellular, 11
autonomic nervous system in, 648-650,
retinal, 628, 629, 629f
deficiency of, 301t, 302
648f
Vitamin B1 (thiamine), 875, 875t
renal response to, 350-357, 351t,
binocular, 622, 622f
deficiency of, 875-876
352f-356f
color, 632-633, 633f, 638, 641f, 643, 644
blood flow and, 198, 267, 267f, 876
diffusion of, 47, 51-52, 51f, 183-184, 183f,
cones in, 631, 631f, 632-633, 633f. See also
cardiac output and, 236, 236f, 267, 267f,
814
Cones.
876
evaporation of, 891f, 892
contrast, 638, 638f, 643, 643f
gastrointestinal effects of, 876
excess of, 301-302, 301t
contrast in, 643, 643f, 644
nervous system effects of, 876
renal response to, 349-350, 349f
depth perception in, 621-622, 622f,
oxygen uptake in, 530
formation of, 835, 835f
647-648
Vitamin B2 (riboflavin), 875t, 876
free, 357
detail in, 641f, 643
deficiency of, 198, 876
gases in, 492
equilibrium and, 696
Vitamin B6 (pyridoxine), 875t, 877
gastrointestinal, 793
farsighted (hyperopia), 619, 619f
Vitamin B12 (cobalamin), 875t, 876-877
hypothalamic regulation of, 733f, 734
fields of, 644-645, 644f
absorption of, 820
insensible loss of, 291-292
form in, 641f, 643
fetal, 1044
intake/output of, 291-292, 292t
image fusion in, 647-648
hepatic storage of, 862
angiotensin II and, 361, 361t
in albinism, 628
in red blood cell production, 423
arterial pressure and, 218-219, 218f,
lateral inhibition in, 638, 638f
Vitamin C (ascorbic acid), 875t, 877-878
224-226, 226f, 361
lines and borders in, 643-644
deficiency of, 878
dry mouth and, 361, 361t
motion in, 641f, 643
neonatal, 1049
gastrointestinal factors in, 361, 361t
nearsighted (myopia), 619-620, 619f
fetal, 1044
thirst and, 361-362, 361t
neurophysiology of, 640-650
Vitamin D, 875t, 878, 983-985
threshold of drinking and, 362
accurate color pathway in, 642f, 643
deficiency of, 991
intestinal osmosis of, 814
color detection in, 644
in chronic renal failure, 412
large intestine absorption of, 817
dorsal lateral geniculate nucleus in,
fetal, 1044
large intestine secretion of, 815
640-641, 641f
formation of, 983-985, 984f
loop of Henle transport of, 334-336, 335f
fast position and motion pathway in,
functions of, 985
loss of, 291-292, 292t, 350, 362
642f, 643
hepatic storage of, 862
osmosis of, 332, 814
field of vision in, 644-645, 644f
in hypoparathyroidism, 990
reabsorption of, 339, 350-357, 351t,
image analysis in, 643-645, 643f, 644f
in intestinal calcium absorption, 985
352f-356f, 734. See also Urine,
visual cortex in, 640, 641-643, 641f, 642f.
in intestinal phosphate absorption, 985
concentration of; Urine, dilution of.
See also Visual cortex.
neonatal, 1049
angiotensin II and, 342-343, 342t,
normal (emmetropia), 619, 619f
Vitamin D-resistant rickets, 991
377-378
ocular movements in, 645-648, 645f-648f
Vitamin E, 875t, 878
antidiuretic hormone and, 337, 338,
orientation in, 643-644
deficiency of, 878
342t, 343, 348-349, 379, 734
photochemistry of, 628-632
fetal, 1044
arterial pressure and, 341-342
color pigments in, 631, 631f, 632-633,
Vitamin K, 875t, 878
in cardiac failure, 259-260
633f
deficiency of, 464-465
osmosis in, 332, 332f
regulation of, 631-632, 632f
fetal, 1044
tubular/plasma inulin evaluation of,
rhodopsin in, 629-631, 629f-631f
in coagulation, 460, 464-465
339
rods in, 629-631, 629f-631f. See also
Vitreous body, 623
retention of. See Edema.
Rods.
Vitreous humor, 623
salt, 350
stereopsis in, 622, 622f, 643, 647-648
Vocal folds (cords), 481, 481f
testosterone effects on, 1006
third-dimensional position in, 641f,
in swallowing, 782, 782f
total, 293, 295-296, 296t
643
Vocal ligament, 481, 481f
vapor pressure of, 492
Visual acuity, 621, 621f
Vocalization, 481, 481f. See also Language;
Water vapor, partial pressure of, 493t, 494
Visual area II, 641f, 642
Speech.
Weber-Fechner principle, 594
Visual cortex, 640, 641-643, 641f
Voice, testosterone effects on, 1005
Weight, body
color blobs of, 642-643, 642f
Voltage clamp testing, 63, 63f
during pregnancy, 1034
complex cells of, 644
Volume reflex, 212
excess, 872-874. See also Obesity.
hypercomplex cells of, 644
Volume-pressure relationship
loss of, 867, 874
in image fusion, 647-648
of arteries, 172, 172f
thyroid hormone effects on, 936-937
layer IV of, 642f, 643
of cardiac cycle, 110-111, 110f
Weightlessness, in space travel, 543-544
layers of, 642-643, 642f
of veins, 172, 172f
Wernicke’s aphasia, 721
primary, 641-642, 641f
of ventricles, 107f, 108
Wernicke’s area, 716, 718-719, 718f, 721,
removal of, 644
Vomiting, 823-824, 823f
721f
secondary, 641f, 642
acidosis and, 397-398
White blood cell(s), 429-431. See also
separate signals to, 640-641, 642f, 643
alkalosis and, 398
specific cell types.
simple cells of, 643-644
Vomiting center, 823-824, 823f
abnormal excess of, 437
vertical columns of, 642, 642f
Von Willebrand factor, 458
ameboid movement of, 24, 431
Visual purple. See Rhodopsin.
Von Willebrand’s disease, 465
bactericidal agents of, 432
Visual receptive aphasia, 721
chemotaxis of, 430f, 431
Vital capacity, 475-476, 476f
concentration of, 430
Vitamin(s), 875-878, 875t. See also specific
deficiency of, 436-437
vitamins.
W
diapedesis of, 430f, 431
calcium absorption and, 815
W ganglion cells, 637
life span of, 431
deficiency of, 875
Wakefulness, 558, 741. See also Sleep.
lymphocytic lineage of, 430, 430f
fetal, 1044
electroencephalography during, 743, 743f
myelocytic lineage of, 430, 430f
hepatic storage of, 862
Walking movements, 682-683, 683f
phagocytosis by, 431-432
1116
Index
White blood cell(s) (Continued)
X
Z
production of, 430-431, 430f
X ganglion cells, 637
Z disc, 72-73, 73f, 74-75, 75f, 90f
types of, 429-430, 430f
Xenograft, 455
Zero pressure reference level, 179
White pulp, 180, 180f
Zero reference level, in current of injury,
Withdrawal reflexes, 680-681, 680f, 681f
142-143, 143f
Word blindness, 718, 721
Zinc, 879, 879t
Word deafness, 721
Y
Work, skeletal muscle contraction and,
Y ganglion cells, 637
78-79
Work capacity, at high altitude, 540